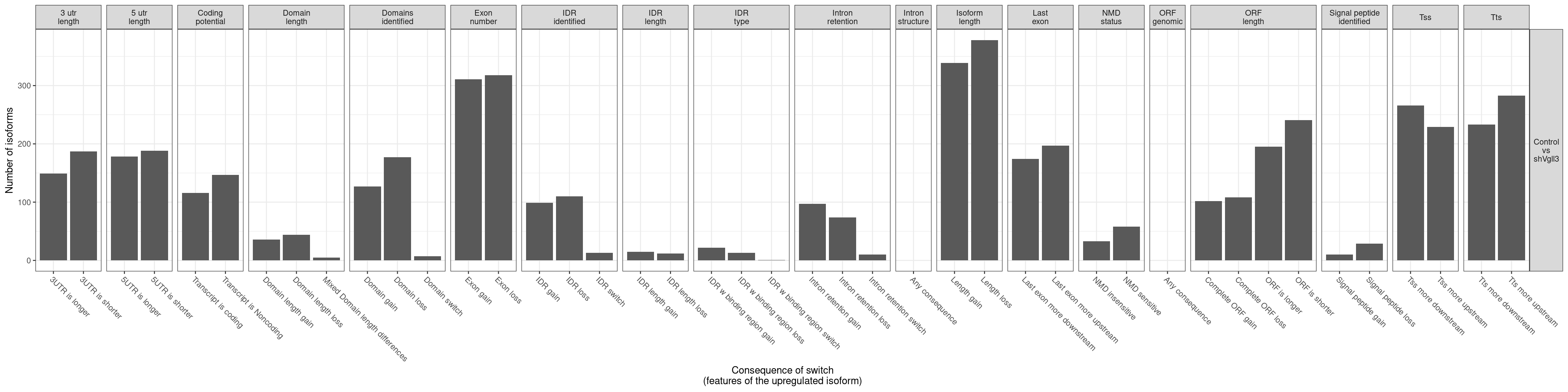
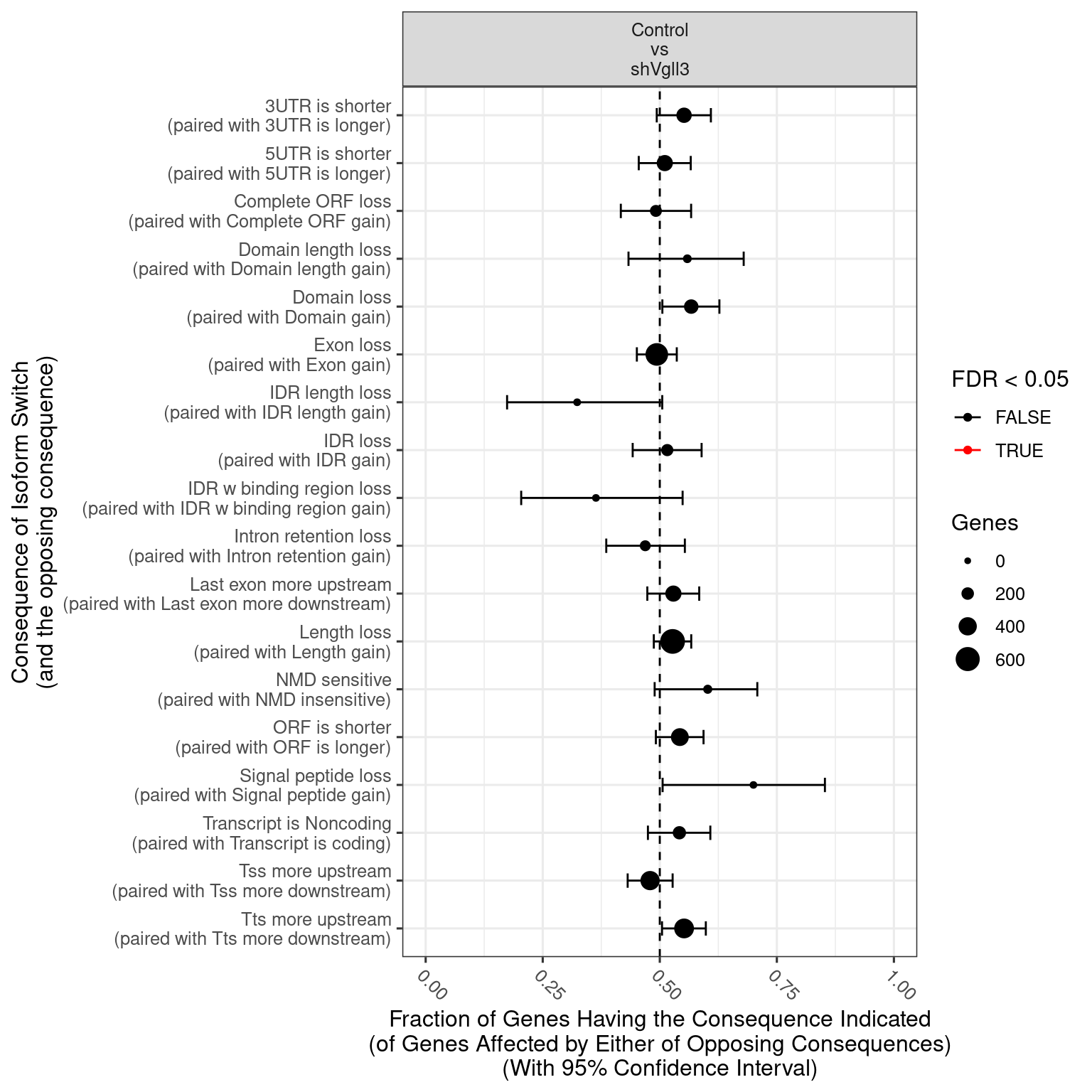
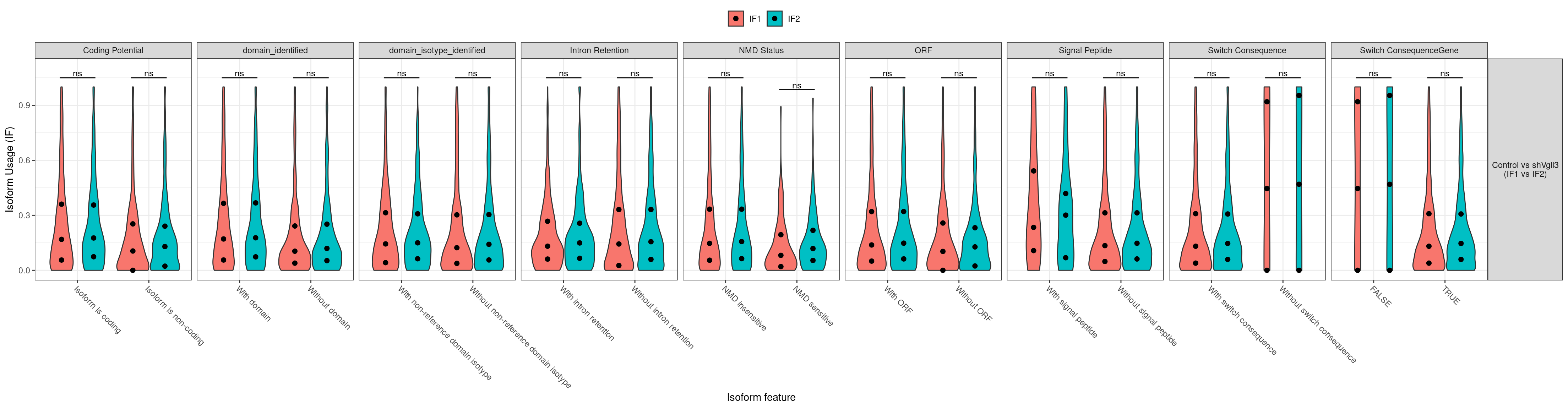
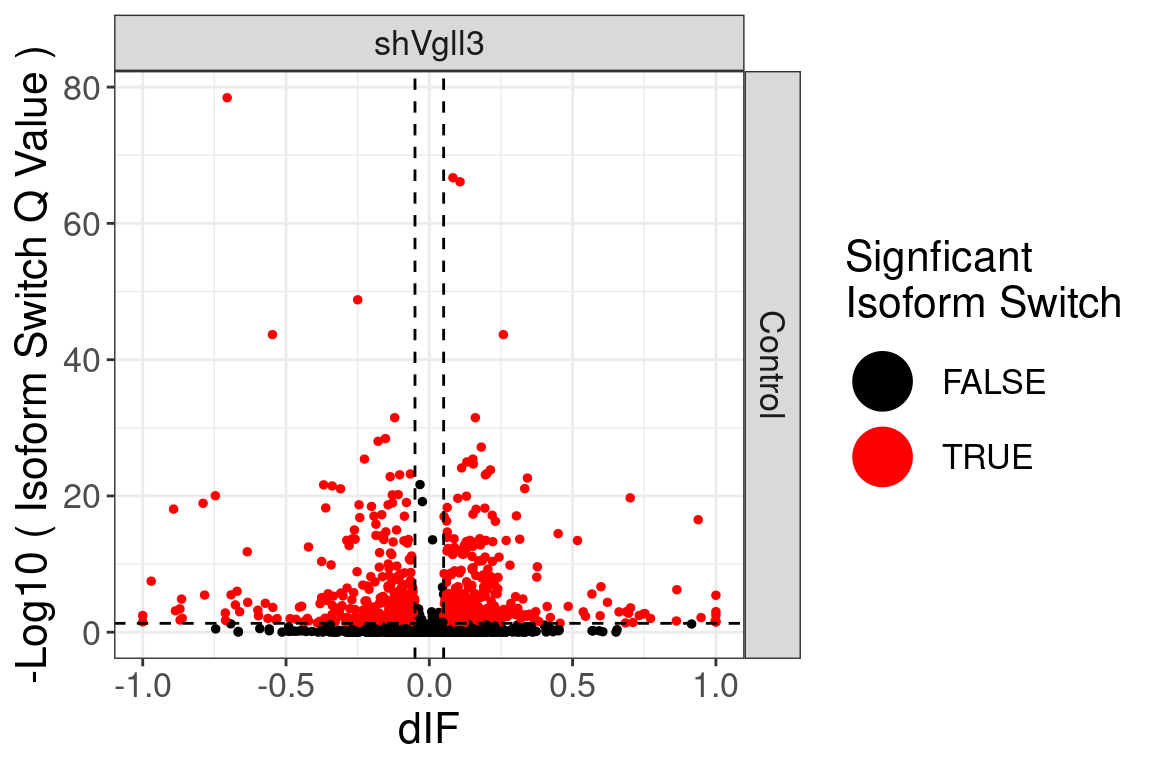
| ENSMUSG00000000031 |
14955 |
H19 |
lncRNA |
|
|
|
|
|
|
|
1709450_H19 gene encodes a non-protein coding transcript that is preferentially expressed from the maternal allele. 11805093_Unregulated expression of the imprinted genes H19 and Igf2r in mouse uniparental fetuses 11861904_gene silencing at the H19 imprinting control region 11874916_These novel sequences interact with the ICR at H19 and the epigenetically regulated silencer at differentially methylated region 1 (DMR1) of Igf2. 11884038_Mouse parthenogenetic embryos with monoallelic H19 expression can develop to day 17.5 of gestation. 12029086_A dyad oct-binding sequence functions as a maintenance sequence for the unmethylated state within the imprinted control region 12475990_Loss of Igf2 imprinting in monoclonal mouse hepatic tumor cells is not associated with abnormal methylation patterns for this protein's and Kvlqt1 differentially methylated regions. 12629040_Mutations lead to loss of CTCF binding and Igf2 imprinting only when passaged through the female germline. 14585991_DNA methylation state of the H19/Igf2 imprinting control region (ICR) of the paternal and maternal alleles 14716017_results suggest that CTCF protects the H19 differentially methylated domain (DMD) from de novo methylation during oocyte growth and is required for normal preimplantation development 15060168_Data show that a mutation in CTCF protein binding site 4 in the imprinting control region upstream of the H19 gene leads to robust activation of the maternal Igf2 allele. 15082756_Data demonstrate that the differentially methylated region (DMR) upstream of the H19 gene can mark its parental origin by a mechanism independent of its own DNA methylation. 15094229_H19 gene expression was dynamically regulated in adult hepatocytes in close association with their proliferation 15199123_targeted deletion of the direct repeat element near a differentially methylated region upstream of H19 had no effect on imprinting within the Igf2-H19 region 15485926_the presence of FoxA proteins is not sufficient for binding but that additional mechanisms must govern the accessibility of FoxA proteins to their cognate binding sites within the H19 E1 enhancer 15543560_expression profile of Igf2 and H19 indicated that the thymus-specific changes of Igf2 and H19 expression were coherent to the hypomethylation of the ICR in the thymus 15870260_sites within the imprinting control center of the H19 and Igf2 genes bind Ku70/80 in a sequence-specific manner and with higher affinity than previously reported binding sites 15899898_hyperhomocysteinemia produces tissue-specific changes in H19 DMD methylation and increased vascular expression of H19 in adult mice 16117632_Drosophila Su(Hw) regulates an evolutionarily conserved silencer from the mouse H19 imprinting control region. 16359639_Our data proved that the changes of the H19 gene methylation pattern interfered by X-ray irradiation could be transmitted and maintained in the first-generation offspring. 16449639_differentially methylated domain exhibits positive transcriptional activity and is required for H19 expression in blastocysts and full activation of H19 during subsequent development 16575185_Imprinting-Control Regionis a bi-functional regulatory element involved not only in H19 imprinting but also in 'formatting' the higher-order chromatin structure for proper tissue-specific expression of both H19 and Igf2 genes. 16575186_The H19/Igf2 imprinting control region (ICR) is a DNA methylation-dependent chromatin insulator in somatic cells. 16928784_addition to being essential for a functional insulator, the CTCF repeats facilitate initiation of H19 expression in the early embryo 17708683_Chromatin immunoprecipitation shows that MBD3 is bound to the H19 differentially methylated domain. 18413996_The paternal imprinted genes, H19 and Igf-2r, had lower methylation levels in heat-stressed embryos, whereas the maternal imprinted genes, Peg3 and Peg1, had similar methylation pattern in heat-stressed embryos and in control embryos. 18459101_The region of the genome containing the imprinted genes insulin-like growth factor 2 (Igf2) and H19, both of which display oncogenic functions, may be particularly sensitive to environmental influences 18617529_The endodermal enhancers contact the maternal H19 and the paternal Igf2 genes. 18632606_CTCFL/BORIS is a methylation-independent DNA-binding protein that preferentially binds to the paternal H19 differentially methylated region 18719115_the H19 locus controls the size of experimental teratocarcinomas, the number of polyps in the Apc murine model of colorectal cancer and the timing of appearance of SV40-induced hepatocarcinomas. It clearly displays a tumor suppressor effect in mice 18724775_study aimed to reveal alterations in expression and methylation levels of the growth-related imprinted genes H19 and Igf2 in fetuses of diabetic mice 19074953_Results show how methylation-dependent repression of the Igf2/H19 imprinting control region from a distance varies with CpG number, density and surrounding sequence. 19524295_placentas from H19(-/-) mice are heavier, have higher number of glycogen cells, and contain larger glycogen concentrations than those of H19(+/+) mice 19533039_These data suggest that multipotent germline stem cells are epigenetically stable for the H19 gene during in vitro modifications. 19762426_Study proposes that the H19 gene participates as a trans regulator in the fine-tuning of this imprinted gene network in the mouse embryo. 19956719_Knockout of Sdhd in the mouse does not result in a disease phenotype; H19 may not be an initiator of PGL/PC tumorigenesis 20038532_The 2.4-kb H19 imprinting control region insertion is sufficient to transmit parent-of-origin-dependent DNA methylation patterns independent of its methylation status in sperm. 20062522_Maternal imprinting control region of Igf2/H19 substantially enhanced normal growth rate and survival in mice. 20720167_Data indicate that regulation of H19 transcription in blastocysts is controlled, at least in part, by Mta2. 20838620_Complete biallelic insulation at the H19/Igf2 imprinting control region position results in fetal growth retardation and perinatal lethality. 21047779_Phosphatidylinositol 3-kinase (PI3K) signaling via glycogen synthase kinase-3 (Gsk-3) regulates DNA methylation of imprinted loci Dnmt3a2, and misexpression of Igf2, H19, and Igf2r 21124827_CTCF-dependent allele-specific chromatin composition imposes a maternal allele-specific delay on de novo methylation imprint establishment at the H19/Igf2 imprinting control region in prospermatogonia. 21478171_Data reveal a novel imprinted domain in the Igf2/H19 locus and leads to a proposed model for chromatin folding of this locus on the paternal chromosome. 21793658_H19 expression was highly upregulated in parthenogenetic embryonic stem cells (ESCs) with more than 6-fold overexpression compared with biparental ESCs. 22415163_The CTCF-binding sites within the H19 imprinting control region (ICR) promote and antagonize the formation of extensive local chromatin loops. 22447856_The expression of Igf2 and H19 genes in the offsping of mice with gestational diabetes mellitus are reported. 22662250_our experiments indicate that a large excess of the H19 transcript can counteract 91H-mediated Igf2 activation 22684254_The controlled release of miR-675 from H19 may also allow rapid inhibition of placental cell proliferation in response to cellular stress or oncogenic signals. 23047867_Polyploidy might therefore serve as a protective mechanism aimed at reducing malignant transformation through the involvement of the H19 regulatory long noncoding RNA. 23216893_Cerebellar DNA methylation across all four CTCF binding sites spanning the murine Igf2/H19 ICR in an outbred population , was quantified. 23221482_The goal of this study is to determine the relationship of tissue S-adenosylmethionine and S-Adenosylhomocysteine concentrations to allele-specific methylation and expression of genomically imprinted H19/Igf2. 23390160_IVF causes alterations in mouse growth during the postnatal periods that may be associated with alterations in Igf2/H19 expression 23406902_Myod and H19-Igf2 locus interactions are required for diaphragm formation. 23437185_The female-biased expression of H19 and Igf2 specifically in the eye may contribute to our understanding of sex differences in normal as well as abnormal eye physiology and processes 23486912_Loss of imprinting of H19 occurs in the primary trophoblast giant cell-containing section of peri-implantation mouse embryos. 23743205_Data indicate that the H19 could induce the Cdx2 even under suppressive stress by overexpressed Oct3/4. 24055342_Identify this H19 lncRNA as an important regulator of the major let-7 family of microRNAs. 24141783_Yap1 and H19 are overexpressed and function downstream of Hedgehog signaling in osteosarcoma. 24297921_H19 lncRNA controls gene expression of the Imprinted Gene Network by recruiting MBD1. 24324735_Using a mouse with mutations in the Oct4 binding sites, maternally transmitted mutant imprinting control region were found to acquire partial methylation in somatic tissues, but there was little effect on imprinted expression of H19 and Igf2. 24532688_the H19 long noncoding RNA has a critical trans-regulatory function in skeletal muscle differentiation and regeneration that is mediated by the microRNAs encoded within H19 24703882_Oncofetal H19 RNA promotes tumor metastasis. 24894171_Knockdown of H19 enhances differentiation capacity to epidermis of parthenogenetic embryonic stem cells. 25064401_Oocytes vitrification could lead to the loss of DNA methylation of imprinted genes (H19, Peg3, and Snrpn) in mouse blastocysts, which is mainly caused by the reductions of DNMTs after vitrification of oocytes. 25257147_analysis of modifications of H3 histone in H19 and MEST imprinted genes in blastocysts produced in vitro from non-vitrified and vitrified two-cell embryos 25399420_H19 acts as an upstream regulator of let-7 in skeletal muscle cells. 26417043_De novo DNA methylation through the 5'-segment of the H19 ICR maintains its imprint during early embryogenesis. 26687445_H19 knockdown activates SAHH, leading to increased DNMT3B-mediated methylation of an lncRNA-encoding gene Nctc1 within the Igf2-H19-Nctc1 locus. 26801864_H19 may inhibit the proliferation of fetal liver cells by blocking the interaction between heterogeneous nuclear ribonucleoprotein (hnRNP) U and actin, which results in gene transcriptional repression. 26980793_the function of H19 in adult muscle, was investigated. 27062045_melatonin antagonized premature senescence of cardiac progenitor cells via H19/miR-675/USP10 pathway, which provides new insights into pharmacological actions and potential applications of melatonin on the senescence of cardiac progenitor cells. 27084844_Reveal a novel function of H19-miR-675 axis targeting CaMKIIdelta as a negative regulator of cardiac hypertrophy, suggesting its potential therapeutic role in cardiac diseases. 27129775_CLIM interacts with estrogen receptor alpha at the H19 locus, potentially explaining the higher expression of H19 in female than male corneas. 27613097_H19 has a promotive effect on BLM-induced idiopathic pulmonary fibrosis, and demonstrated the mechanism by which H19/miR-29b interaction exerts its effect on regulating pulmonary fibrosis 27687727_results demonstrate that the H19-Igf2 axis is negatively regulated by CTCF-PHB1 cooperation and that H19 is involved in modulating the growth-suppressive effect of PHB1 in the liver. 27813675_overexpression of H19 stimulates StAR expression by antagonizing let-7, which inhibits StAR at the post-transcriptional level 27824289_Our study also identified the imprinting control region (ICR) of H19 as a genomic target. According to the results, PEG3 binds to a specific sequence motif located between the 3(rd) and 4(th) CTCF binding sites of the H19-ICR. PEG3 also binds to the active maternal allele of the H19-ICR. 27874091_HMGA1P7 mRNA sustains the H19 and Igf2 overexpression by acting as miRNA decoy. 28367967_These results demonstrate that the G-quadruplex structure immediately downstream of the transcription start site functions as a novel regulatory element for H19 gene expression. 28407375_activation of hepatic H19RNA promoted cholestatic liver fibrosis through the ZEB1/EpCAM signaling pathway 28753062_Long noncoding RNA H19 mediates CTGF expression in cardiac fibrosis. 28814771_our results identify H19 as a critical regulator of hepatic gluconeogenesis during diabetes and provide new insights into the roles of lncRNAs in metabolism. 28943313_The lethal Ogden syndrome-associated mutation of Naa10p disrupts its binding to the imprinting control region of H19 and Dnmt1 recruitment. 29099871_H19 may be important for normal liver development, and understanding how the liver matures will assist in predicting drug efficacy and toxicity in pediatric populations 29140550_Study unveils an H19/PTBP1/sterol regulatory element-binding protein 1 feedforward amplifying signaling pathway to exacerbate the development of fatty liver 29172088_Expression of H19 was up-regulated in ox-LDL-treated Raw264.7 cells. H19 shRNA decreased the level of pro-inflammatory factors (TNF-alpha, IL-1beta)/CD68+ cells and increased the level of anti-inflammatory factors (IL-4, IL-10)/CD163+ cells compared with the control group. Silencing H19 inhibits the adipogenesis and inflammation response in ox-LDL-treated Raw264.7 cells by up-regulating miR-130b. 29244185_Loss of imprinting at the Igf2/H19 locus disrupts in vitro differentiation of primary myoblasts. 29425397_Cholangiocyte-derived exosomal-H19 plays a critical role in cholestatic liver injury. Serum exosomal H19 represents a noninvasive biomarker and potential therapeutic target for cholestatic diseases 29621481_The level of lncRNA H19 is increased in inflamed intestinal tissues from mice and patients. The inflammatory cytokine IL22 induces expression of H19 in IECs, which is required for intestinal epithelial proliferation and mucosal healing. H19 lncRNA appears to inhibit p53 protein and microRNA 34a and let-7 to promote proliferation of IECs and epithelial regeneration. 29663375_Long noncoding RNA H19 mediates LCoR to regulate the balance between osteogenic and adipogenic differentiation of mBMSCs in mice through sponging miR-188. 29669788_The long noncoding RNA H19 is a novel regulator of smooth muscle cell survival in abdominal aortic aneurysm development and progression. 29762823_H19, as a molecular marker for promoting fracture healing, promote the proliferation of osteocytes by inhibiting the expression of p53. 30014520_TGF-beta signaling in tumor-initiating hepatocytes inhibits H19 expression by Sox2, and this signaling axis is crucial for inhibition of hepatocellular carcinoma development. 30078183_our findings provide the first evidence supporting the role of H19 in IECs of DM mice, and miR-141-3p targets not only protein-coding genes but also the lncRNA H19. 30107531_lncRNA H19 is repressed during aging and controls endothelial cell senescence, proliferation, inflammatory activation and angiogenic sprouting by inhibiting STAT3 activation. 30110641_RNA pol III-mediated transcription positively regulates long non-coding RNA H19 and Wnt6 expression, established adipogenesis inhibitors. 30137188_Long non-coding RNA H19 (H19) regulated the proliferation and migration of cardiac progenitor cells (CPCs) through mediating the expression of sirtuin 1 (Sirt1) which is a target of miR-200a-3p under hypoxia. 30137224_AMH's inhibitory actions are regulated at least in part by H19, likely via let-7, marking this ncRNA pair as important regulators of the establishment and maintenance of the follicular pool. 30190464_H19 controls brown adipogenesis and brown adipose tissue oxidative metabolism.H19 increases brown adipose tissue metabolism and prevents obesity. 30362572_a novel H19/miR-148a/USP4 axis which promoted liver fibrosis via TGF-beta pathway in both hepatic stellate cells and hepatocytes, is reported. 30465059_H19 regulates FoxO1 transcript and protein levels. H19 inhibition increases p53 occupancy on the FoxO1 promoter that promotes FoxO1 transcription. H19 inhibition in vivo induces hyperglycemia and impairs glucose, insulin, and pyruvate tolerance. In vivo H19 inhibition increases the hepatic transcript levels of gluconeogenic genes and FoxO1. 30496976_these data indicate that H19 inhibition protects the heart against myocardial I/R injury, which may be partly attributed to regulation of the miR-675/PPARalpha axis. 30633332_H19 and Foxc2 synergistically promoted osteogenic differentiation of bone marrow mesenchymal stem cells via Wnt-beta-catenin pathway 30639035_H19 deficiency increased the activity of S-adenosylhomocysteine hydrolase, a regulator of DNA methylation, which partially contributed to the observed hematopoietic defect. 30778047_H19 promoted let-7 expression by decreasing PTBP1's expression level and its binding to the let-7 precursors in cholestasis. 30922932_noncoding-RNA-mediated regulation of STAR may play an important role in the regulation of steroid hormone production. 31127201_LncRNA H19 initiates microglial pyroptosis and neuronal death in retinal ischemia/reperfusion injury. 31152924_IGFBPrP1 promoted autophagy and contributed to hepatic stellate cell activation via mutual regulation between H19 and the PI3K/AKT/mTOR pathway. 31299871_Targeted inhibition of long non-coding RNA H19 blocks anaplastic thyroid carcinoma growth and metastasis in a cell line and in vivo xenografts. 31515683_The loss of methylation of H19 ICR caused by ARTs occurs between the 8-cell and the morula stages. 31556166_n vitro fertilization placenta overgrowth in mice is associated with downregulation of the paternal imprinting gene H19. 31588235_Hypoxia-induced H19/YB-1 cascade modulates cardiac remodeling after infarction. 31609547_H19 is not hypomethylated or upregulated with age or sex in the aortic valves of mice. 31809000_Long non-coding RNA (lncRNA) H19 induces hepatic steatosis through activating MLXIPL and mTORC1 networks in hepatocytes. 31862317_Long Noncoding RNA H19 Impairs the Intestinal Barrier by Suppressing Autophagy and Lowering Paneth and Goblet Cell Function. 31940841_Cholangiocyte-Derived Exosomal lncRNA H19 Promotes Macrophage Activation and Hepatic Inflammation under Cholestatic Conditions. 32084395_Ghrelin protects against obesity-induced myocardial injury by regulating the lncRNA H19/miR-29a/IGF-1 signalling axis. 32281297_Inhibition of LncRNAH19 has the effect of anti-tumour and enhancing sensitivity to Gefitinib and Chemotherapy in Non-small-cell lung cancer in vivo. 32301281_Epidermal growth factor regulation by autophagy-mediated lncRNA H19 in murine intestinal tract after severe burn. 32304991_LncRNA-H19 induces hepatic stellate cell activation via upregulating alcohol dehydrogenase III-mediated retinoic acid signals. 32308025_The Role of LncRNA H19 in MAPK Signaling Pathway Implicated in the Progression of Bronchopulmonary Dysplasia. 32335212_LncRNA H19 Aggravates Cerebral Ischemia/Reperfusion Injury by Functioning as a ceRNA for miR-19a-3p to Target PTEN. 32356199_LncRNA H19 Attenuates Apoptosis in MPTP-Induced Parkinson's Disease Through Regulating miR-585-3p/PIK3R3. 32445757_these results provided the first mechanistic evidence that hepatic stellate cell activation-induced lipid droplet disappearance in liver fibrosis is associated with the lncRNA-H19/AMPKalpha pathway 32603313_Long noncoding RNA H19X is a key mediator of TGF-beta-driven fibrosis. 32649928_Pleiotropic cardiac functions controlled by ischemia-induced lncRNA H19. 32945413_Downregulation of lncRNA H19 alleviates atherosclerosis through inducing the apoptosis of vascular smooth muscle cells. 33093654_The pro-oncogenic effect of the lncRNA H19 in the development of chronic inflammation-mediated hepatocellular carcinoma. 33115498_LncRNAH19 improves insulin resistance in skeletal muscle by regulating heterogeneous nuclear ribonucleoprotein A1. 33138822_Long non-coding RNA H19 deficiency ameliorates bleomycin-induced pulmonary inflammation and fibrosis. 33174326_Long noncoding RNA H19 acts as a miR-29b sponge to promote wound healing in diabetic foot ulcer. 33205379_LncRNA H19 Regulates Lipopolysaccharide (LPS)-Induced Apoptosis and Inflammation of BV2 Microglia Cells Through Targeting miR-325-3p/NEUROD4 Axis. 33230843_Long noncoding RNA H19 act as a competing endogenous RNA of Let-7g to facilitate IEC-6 cell migration and proliferation via regulating EGF. 33285606_Mechanism of lncRNA H19 in Regulating Pulmonary Injury in Hyperoxia-Induced Bronchopulmonary Dysplasia Newborn Mice. 33313940_Long noncoding RNA H19 inhibition ameliorates oxygenglucose deprivationinduced cell apoptosis and inflammatory cytokine expression by regulating the microRNA29b/SIRT1/PGC1alpha axis. 33615520_H19 Promotes HCC Bone Metastasis Through Reducing Osteoprotegerin Expression in a Protein Phosphatase 1 Catalytic Subunit Alpha/p38 Mitogen-Activated Protein Kinase-Dependent Manner and Sponging microRNA 200b-3p. 33758383_6-Gingerol relieves myocardial ischaemia/reperfusion injury by regulating lncRNA H19/miR-143/ATG7 signaling axis-mediated autophagy. 33811969_Paternal obesity impairs hepatic gluconeogenesis of offspring by altering Igf2/H19 DNA methylation. 34335960_Exosomal lncRNA-H19 promotes osteogenesis and angiogenesis through mediating Angpt1/Tie2-NO signaling in CBS-heterozygous mice. 34392794_Effects of long noncoding RNA H19 on cementoblast differentiation, mineralisation, and proliferation. 34402430_Cardiac pathologies in mouse loss of imprinting models are due to misexpression of H19 long noncoding RNA. 34454586_Functional significance of gain-of-function H19 lncRNA in skeletal muscle differentiation and anti-obesity effects. 34670662_[lncRNA-H19 participates in the progression of synovial inflammation in collagen induced arthritis(CIA) mice by promoting the activity of fibroblast-like synovial cells]. 34818249_The Long Noncoding RNA-H19 Mediates the Progression of Fibrosis from Acute Kidney Injury to Chronic Kidney Disease by Regulating the miR-196a/Wnt/beta-Catenin Signaling. 35008427_Paternal Exercise Improves the Metabolic Health of Offspring via Epigenetic Modulation of the Germline. 35139769_Long noncoding RNA H19 suppresses cardiac hypertrophy through the MicroRNA-145-3p/SMAD4 axis. 35243752_Long non-coding RNA H19 as a biomarker for hepatocellular carcinoma. 35432202_Hepatic IGF2/H19 Epigenetic Alteration Induced Glucose Intolerance in Gestational Diabetes Mellitus Offspring via FoxO1 Mediation. 35437883_FGF21 alleviates pulmonary hypertension by inhibiting mTORC1/EIF4EBP1 pathway via H19. 35438054_LncRNA H19 acts as miR-301a-3p sponge to alleviate lung injury in mice with sepsis by regulating Adcy1. 35483327_Mechanism of lncRNA-H19 in Intestinal Injury of Mice with Ulcerative Colitis. 35538041_LncRNA H19 inhibits oxidative stress injury of cochlear hair cells by regulating miR-653-5p/SIRT1 axis. 35793244_Long non-coding RNA H19 contributes to spinal cord ischemia/reperfusion injury through increasing neuronal pyroptosis by miR-181a-5p/HMGB1 axis. 35842608_H19 inhibition increases HDAC6 and regulates IRS1 levels and insulin signaling in the skeletal muscle during diabetes. 36010635_Long Non-Coding RNA H19 Prevents Lens Fibrosis through Maintaining Lens Epithelial Cell Phenotypes. 36180862_LncRNA H19 alleviates sepsis-induced acute lung injury by regulating the miR-107/TGFBR3 axis. 36314132_RNA-Binding Protein LIN28a Regulates New Myocyte Formation in the Heart Through Long Noncoding RNA-H19. 36326553_LncRNA H19 via miR-29a-3p is involved in lung inflammation and pulmonary fibrosis induced by neodymium oxide. 36459790_Long noncoding RNA H19 synergizes with STAT1 to regulate SNX10 in rheumatoid arthritis. |
|
|
1211.984619 |
3.2244611110 |
1.689058 |
0.12586668 |
173.928602 |
0.00000000000000000000000000000000000000102606855027680597648161003750304085829063360164941818941484753666405303907323086613354981073197992971247904936404893305734731256961822509765625000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000001175118508106489403929950702311817033715390613711573955209413219601609616582242282745235545415082162312714331164897885173559188842773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1860.98082291153 |
162.565704126867 |
577.144756555391 |
38.5592681724131 |
| ENSMUSG00000000037 |
107815 |
Scml2 |
protein_coding |
A0A571BDL9 |
Human_homologues FUNCTION: Putative Polycomb group (PcG) protein. PcG proteins act by forming multiprotein complexes, which are required to maintain the transcriptionally repressive state of homeotic genes throughout development (By similarity). {ECO:0000250}. |
Proteomics identification;Reference proteome |
|
|
|
chromatin [GO:0000785]; nucleus [GO:0005634]; PcG protein complex [GO:0031519]; XY body [GO:0001741]; chromatin binding [GO:0003682]; histone binding [GO:0042393]; anatomical structure morphogenesis [GO:0009653]; apoptotic process [GO:0006915]; histone H2A-K119 monoubiquitination [GO:0036353]; negative regulation of DNA-templated transcription [GO:0045892]; protein localization [GO:0008104]; protein localization to chromosome [GO:0034502] |
25634095_Our results demonstrate that the SCML2/USP7 complex constitutes a novel molecular pathway in modulating the epigenetic state of sex chromosomes during male meiosis 25634095_Scml2 is specifically expressed in germ cells. SCML2 localizes to the XY body during male meiosis. SCML2 is associated with phosphorylated H2AX and USP7, a deubiquitinase. Loss of SCML2 in mice causes defective spermatogenesis, resulting in sharply reduced sperm production. In the absence of SCML2, USP7 fails to accumulate on the XY body, whereas H2A monoubiquitination is dramatically augmented in the XY chromatin. 29462142_RNF8 and SCML2 cooperate to regulate ubiquitination and H3K27 for gene activation on sex chromosomes. 29686098_SCML2-dependent H3K27me3 in the male germline prepares the expression of developmental regulator and somatic genes in embryonic development. 30097555_SCML2 accumulates on pericentromeric heterochromatin (PCH) in male germ cells, where it suppresses PRC1-mediated monoubiquitylation of histone H2A at Lysine 119 (H2AK119ub) and promotes deposition of PRC2-mediated H3K27me3 during meiosis. 30137219_These results suggest that the rapid evolution of SCML2 is due to reproductive adaptation, which has promoted species-specific gene expression programs in spermatogenesis. 32895557_Super-enhancer switching drives a burst in gene expression at the mitosis-to-meiosis transition. |
ENSG00000102098 |
SCML2 |
225.072857 |
2.0276000402 |
1.019773 |
0.35747111 |
7.320509 |
0.00681721192341575802503550463029569073114544153213500976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0241937959034177127448650423957587918266654014587402343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
298.582800987211 |
74.7422308166444 |
147.259220292655 |
27.612404799053 |
| ENSMUSG00000000093 |
21385 |
Tbx2 |
protein_coding |
Q60707 |
FUNCTION: Transcription factor which acts as a transcriptional repressor (PubMed:22186728, PubMed:11867218, PubMed:18025091, PubMed:12023302). May also function as a transcriptional activator (PubMed:26486273, PubMed:22186728, PubMed:11867218). Binds to the palindromic T site 5'-TTCACACCTAGGTGTGAA-3' DNA sequence, or a half-site, which are present in the regulatory region of several genes (PubMed:9710594, PubMed:26971330, PubMed:12023302, PubMed:33731112, PubMed:27720610). Required for cardiac atrioventricular canal formation (PubMed:15459098). May cooperate with NKX2.5 to negatively modulate expression of NPPA/ANF in the atrioventricular canal (PubMed:12023302). May play a role as a positive regulator of TGFB2 expression, perhaps acting in concert with GATA4 in the developing outflow tract myocardium (PubMed:22186728). Plays a role in limb pattern formation (PubMed:15459098). Acts as a transcriptional repressor of ADAM10 gene expression, perhaps in concert with histone deacetylase HDAC1 as cofactor (PubMed:30599067). Involved in branching morphogenesis in both developing lungs and adult mammary glands, via negative modulation of target genes; acting redundantly with TBX3 (PubMed:27720610, PubMed:16222716). Required, together with TBX3, to maintain cell proliferation in the embryonic lung mesenchyme; perhaps acting downstream of SHH, BMP and TGFbeta signaling (PubMed:27720610). Involved in modulating early inner ear development, acting independently of, and also redundantly with TBX3, in different subregions of the developing ear (PubMed:33795231). Acts as a negative regulator of PML function in cellular senescence (By similarity). Acts as a negative regulator of expression of CDKN1A/p21, IL33 and CCN4; repression of CDKN1A is enhanced in response to UV-induced stress, perhaps as a result of phosphorylation by p38 MAPK (PubMed:18025091, PubMed:33731112). Negatively modulates expression of CDKN2A/p19ARF and CDH1/E-cadherin (By similarity). Plays a role in induction of the epithelial-mesenchymal transition (EMT) (By similarity). Plays a role in melanocyte proliferation, perhaps via regulation of cyclin CCND1 (PubMed:26486273). Involved in melanogenesis, acting via negative modulation of expression of DHICA oxidase/TYRP1 and P protein/OCA2 (PubMed:26971330, PubMed:9710594). Involved in regulating retinal pigment epithelium (RPE) cell proliferation, perhaps via negatively modulating transcription of the transcription factor CEBPD (PubMed:28910203). {ECO:0000250|UniProtKB:Q13207, ECO:0000269|PubMed:11867218, ECO:0000269|PubMed:12023302, ECO:0000269|PubMed:15459098, ECO:0000269|PubMed:16222716, ECO:0000269|PubMed:18025091, ECO:0000269|PubMed:22186728, ECO:0000269|PubMed:26486273, ECO:0000269|PubMed:26971330, ECO:0000269|PubMed:27720610, ECO:0000269|PubMed:28910203, ECO:0000269|PubMed:30599067, ECO:0000269|PubMed:33731112, ECO:0000269|PubMed:33795231, ECO:0000269|PubMed:9710594}. |
Developmental protein;DNA-binding;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:21385; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; DNA binding [GO:0003677]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; histone deacetylase binding [GO:0042826]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; aorta morphogenesis [GO:0035909]; apoptotic process [GO:0006915]; atrioventricular canal development [GO:0036302]; atrioventricular canal morphogenesis [GO:1905222]; cardiac jelly development [GO:1905072]; cardiac muscle cell myoblast differentiation [GO:0060379]; cardiac muscle tissue development [GO:0048738]; cell fate specification [GO:0001708]; cellular senescence [GO:0090398]; cochlea morphogenesis [GO:0090103]; developmental growth involved in morphogenesis [GO:0060560]; embryonic camera-type eye morphogenesis [GO:0048596]; embryonic digit morphogenesis [GO:0042733]; embryonic heart tube development [GO:0035050]; endocardial cushion formation [GO:0003272]; endocardial cushion morphogenesis [GO:0003203]; epithelial tube branching involved in lung morphogenesis [GO:0060441]; fibroblast growth factor receptor signaling pathway [GO:0008543]; heart looping [GO:0001947]; heart morphogenesis [GO:0003007]; mammary placode formation [GO:0060596]; melanocyte proliferation [GO:0097325]; mesenchymal cell proliferation involved in lung development [GO:0060916]; muscle cell fate determination [GO:0007521]; negative regulation of cardiac chamber formation [GO:1901211]; negative regulation of cellular senescence [GO:2000773]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of heart looping [GO:1901208]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neurogenesis [GO:0022008]; Notch signaling pathway [GO:0007219]; outflow tract morphogenesis [GO:0003151]; outflow tract septum morphogenesis [GO:0003148]; pharynx development [GO:0060465]; pigment metabolic process involved in pigmentation [GO:0043474]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cell cycle G1/S phase transition [GO:1902808]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of heart contraction [GO:0008016]; regulation of transcription by RNA polymerase II [GO:0006357]; response to retinoic acid [GO:0032526]; roof of mouth development [GO:0060021]; smooth muscle cell differentiation [GO:0051145]; ureteric peristalsis [GO:0072105] |
12023302_Cooperative action of Tbx2 and Nkx2.5 inhibits ANF expression in the atrioventricular canal thus suppressing myocardial chamber formation 15042700_T-box transcription factor Tbx2 represses differentiation and formation of the cardiac chambers. 15093729_Tbx2 is an immediate-early gene target in retinoic acid-treated B16 murine melanoma cells. 15459098_down-regulates Cdkn's, but loss of function doesn't result in increased levels of p19(ARF), p16(INK4a), p15(INK4b) or p21 15591049_SRF embryonic cardiac gene expression is dependent upon the SRF 3'-UTR enhancer, Tbx2, Tbx5, and TIP60 histone acetyltransferase activity. 15843407_Tbx2 directly represses Nmyc1 in outflow tract and atrioventricular canal of the developing heart, resulting in relatively low proliferation. 15901664_Tbx20 may ensure progression to a multi-chambered heart by repressing Tbx2 in the myocardial precursor cells of the linear heart tube destined to form the chambers 16222716_Tbx3, the ulnar-mammary syndrome gene, and Tbx2 interact in mammary gland development through a p19Arf/p53-independent pathway. 16730707_Results are consistent with Tbx2 playing a role in cell cycle progression and organization of subnuclear compartments. 16912158_Inactivation of TBX2 using a dominant-negative TBX2 resulted in up-regulation of p19(ARF) in SAFB1 knockout mouse embryo fibroblasts. 16958122_Tbx2 expression is observed during somitogenesis beginning at E9.5, both in the posterior half of prospective somites and in a progressively restricted pattern in recently formed somites. 17259303_Hesr1 and Hesr2 play crucial roles in AV boundary formation through the suppression of Tbx2. 18025091_the ability of Tbx2 to repress the p21 gene is enhanced in response to a stress-induced senescence pathway, which leads to a better understanding of the regulation of the anti-senescence function of Tbx2. 18285513_Msx1 and Msx2 can function in concert with the TBX2/3 to suppress Cx43 and other working myocardial genes. 18534921_A comparison of the expression of Tbx2 and Tbx3 in the developing hypothalamic-pituitary axis. 18936168_Results show that depending on the level of pocket proteins, loss of adhesion induces G(1) and G(2) arrest, which could be alleviated by overexpression of the TBX2 oncogene. 19423846_Tbx2 temporally suppresses differentiation and proliferation of primary myocardial cells. A subset of these Tbx2Cre-marked cells switch off expression of Tbx2, which allows them to differentiate into chamber myocardium and to initiate proliferation. 19563797_Tbx20 within the OFT, indicating complex and region-specific transcriptional cross-regulation between the two T-box genes. 19576202_Tbx2 is required for proper optic cup formation and plays a critical early role in regulating regional retinal growth and the acquisition of shape during optic vesicle invagination 19661464_Tbx20 interacts with smads to confine tbx2 expression to the atrioventricular canal. 19769959_Mice homozygous mutant for Tbx2 and mice double heterozygous for Tbx2 and Tbx3 exhibit a cleft palate phenotype arguing for an important contribution of Tbx2 and Tbx3 to palatogenesis 19846762_Tbx2, a central intermediary of Bmp-Smad signaling, has a central part in directing Has2 and Tgfbeta2 expression, facilitating endocardial cushions formation. 19960541_This review presents a state of the art overview of the role and regulation of Tbx2 in early embryonic development and in cancer[review] 20941780_Study identify that the homeobox gene Nkx2-5 is required for early ventral restriction of Slit3 and that the T-box transcription factor Tbx2 mediates repression of Slit3 in nonchamber myocardium. 21266775_Data show that myocardium-specific inactivation of T-box 2 (Tbx2) leads to the formation of fast-conducting accessory pathways, malformation of the annulus fibrosus, and ventricular preexcitation in mice. 21867776_Tbx2 and Tbx3 are both expressed in the pancreatic mesenchyme throughout development 22130515_Tbx2 and Tbx3 trigger development of the endocardial cushions through a regulatory feed-forward loop with Bmp2, thus providing a mechanism for the co-localization and coordination of these important processes in heart development. 22186728_The Tbx2-Tgfbeta2 cascade is one of the key pathways involved in inducing the transposition of the great arteries phenotype. 22223620_This study lays the foundation for investigation of functional requirements for Tbx2 subfamily genes in development of the mammalian reproductive system. 22844464_an unanticipated link between TBX2 deregulation in cancer and the acquisition of EMT and invasive features of epithelial tumor cells. 23020925_The identification of TBX2 as a target for PAX3 provides a key insight into how PAX3 may contribute to melanoma evolution. 23341776_Tbx2-mediated regulation of Cdkn1a and Cdkn1b represents a crucial node in the network integrating patterning information and cell cycle regulation that underlies growth, differentiation, and branching morphogenesis of this organ. 23633963_We show that Tbx2 directly represses Grem1 in distal regions of the posterior limb mesenchyme allowing Bone morphogenetic protein (Bmp) signaling to abrogate Fgf4/9/17 expression in the overlying epithelium. 23674600_formation of the neurohypophysis requires Tbx3 and Tbx2 to sequester the SRY box-containing transcription factor Sox2 away from a Shh forebrain enhancer (SBE2) 23720046_Msx1 and Tbx2 antagonistically regulate Bmp4 expression during the bud-to-cap stage transition in tooth development. 26971330_Standard chromatin immunoprecipitation and reporter assays suggest that TBX2 represses Oca2 at least in part directly. Hence, the results suggest that TBX2 may act as a nexus linking cell proliferation and melanogenesis 27253890_Tbx18 does not function redundantly with Tbx2 or Tbx20 in epicardial development. 27720610_Tbx2 and Tbx3 function downstream of Shh to maintain pro-proliferative mesenchymal Wnt signaling. 28910203_TBX2 is expressed in RPE cells both in vivo and in vitro. Specific knockdown of TBX2 in the human RPE cell line ARPE-19 leads to an accumulation of cells at G1. 30478225_TBX2 and TBX3 transcription factors have novel and required roles in regulating patterning and differentiation in the murine ureter. 31870435_The expression of TBX2 in the developing lung shows a role for TBX2 in maintaining the precursor state of pulmonary mesenchymal progenitor cells. 33410138_A role of Hey2 transcription factor for right ventricle development through regulation of Tbx2-Mycn pathway during cardiac morphogenesis. 33731112_Combined genomic and proteomic approaches reveal DNA binding sites and interaction partners of TBX2 in the developing lung. 33795231_Regulation of otocyst patterning by Tbx2 and Tbx3 is required for inner ear morphogenesis in the mouse. 35506352_Complex functional redundancy of Tbx2 and Tbx3 in mouse limb development. 35508658_Tbx2 is a master regulator of inner versus outer hair cell differentiation. 36494345_TBX2 specifies and maintains inner hair and supporting cell fate in the Organ of Corti. |
ENSG00000121068 |
TBX2 |
418.575901 |
0.4751570756 |
-1.073524 |
0.14822207 |
51.790448 |
0.00000000000061751513542025680122573141199841580009730762634845291358942631632089614868164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000126254906613709422192148959165524573031863520000683820398990064859390258789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
271.894210763695 |
27.983501904685 |
572.219640100548 |
42.7842998536767 |
| ENSMUSG00000000120 |
18053 |
Ngfr |
protein_coding |
Q9Z0W1 |
FUNCTION: Low affinity neurotrophin receptor which can bind to mature NGF, BDNF, NTF3, and NTF4 (PubMed:11559852, PubMed:1317267). Forms a heterodimeric receptor with SORCS2 that binds the precursor forms of NGF (proNGF), BDNF (proBDNF) and NTF3 (proNT3) with high affinity, and has much lower affinity for mature NGF and BDNF (PubMed:22155786, PubMed:24908487, PubMed:27457814). Plays an important role in differentiation and survival of specific neuronal populations during development (PubMed:1317267, PubMed:11559852). Can mediate cell survival as well as cell death of neural cells (PubMed:1317267, PubMed:11559852, PubMed:24908487). The heterodimeric receptor formed with SORCS2 plays a role in proBDNF-dependent synaptic plasticity, in hippocampal long term depression (LTD) and long term potentiation (LTP) (PubMed:27457814). Plays a role in the inactivation of RHOA (By similarity). Plays a role in the regulation of the translocation of GLUT4 to the cell surface in adipocytes and skeletal muscle cells in response to insulin, probably by regulating RAB31 activity, and thereby contributes to the regulation of insulin-dependent glucose uptake (PubMed:22460790). Necessary for the circadian oscillation of the clock genes BMAL1, PER1, PER2 and NR1D1 in the suprachiasmatic nucleus (SCN) of the brain and in liver and of the genes involved in glucose and lipid metabolism in the liver (PubMed:23785138). {ECO:0000250, ECO:0000250|UniProtKB:P08138, ECO:0000269|PubMed:11559852, ECO:0000269|PubMed:1317267, ECO:0000269|PubMed:22155786, ECO:0000269|PubMed:22460790, ECO:0000269|PubMed:23785138, ECO:0000269|PubMed:24908487, ECO:0000269|PubMed:27457814}.; FUNCTION: (Microbial infection) Cell surface receptor for rabies virus glycoprotein Gs. {ECO:0000269|PubMed:9857182}.; FUNCTION: [Isoform 2]: Does not bind NGF, BDNF, NTF3, and NTF4. {ECO:0000269|PubMed:11559852}. |
Alternative splicing;Apoptosis;Biological rhythms;Cell membrane;Cell projection;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Membrane;Neurogenesis;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:18053; |
cell surface [GO:0009986]; cell-cell junction [GO:0005911]; clathrin-coated endocytic vesicle [GO:0045334]; coated vesicle [GO:0030135]; cytoplasm [GO:0005737]; dendrite membrane [GO:0032590]; dendritic spine [GO:0043197]; external side of plasma membrane [GO:0009897]; Golgi apparatus [GO:0005794]; growth cone [GO:0030426]; membrane raft [GO:0045121]; mitochondrion [GO:0005739]; neuromuscular junction [GO:0031594]; neuronal cell body membrane [GO:0032809]; nuclear envelope [GO:0005635]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; presynaptic membrane [GO:0042734]; rough endoplasmic reticulum [GO:0005791]; amyloid-beta binding [GO:0001540]; calmodulin binding [GO:0005516]; coreceptor activity [GO:0015026]; death receptor activity [GO:0005035]; identical protein binding [GO:0042802]; nerve growth factor binding [GO:0048406]; neurotrophin binding [GO:0043121]; neurotrophin TRKA receptor binding [GO:0005168]; preprotein binding [GO:0070678]; protein-containing complex binding [GO:0044877]; small GTPase binding [GO:0031267]; ubiquitin protein ligase binding [GO:0031625]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; axon guidance [GO:0007411]; cellular glucose homeostasis [GO:0001678]; cellular response to amyloid-beta [GO:1904646]; cellular response to oxidative stress [GO:0034599]; central nervous system development [GO:0007417]; circadian regulation of gene expression [GO:0032922]; circadian rhythm [GO:0007623]; detection of temperature stimulus [GO:0016048]; dorsal aorta development [GO:0035907]; fibroblast growth factor receptor signaling pathway [GO:0008543]; fibroblast proliferation [GO:0048144]; glucose homeostasis [GO:0042593]; hair follicle development [GO:0001942]; hair follicle morphogenesis [GO:0031069]; intracellular protein transport [GO:0006886]; negative regulation of angiogenesis [GO:0016525]; negative regulation of apoptotic process [GO:0043066]; negative regulation of blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:1903588]; negative regulation of cell migration [GO:0030336]; negative regulation of dendritic spine development [GO:0061000]; negative regulation of fibroblast growth factor receptor signaling pathway [GO:0040037]; negative regulation of hair follicle development [GO:0051799]; negative regulation of mitochondrial depolarization [GO:0051902]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of neuron projection development [GO:0010977]; nerve development [GO:0021675]; neuron apoptotic process [GO:0051402]; odontogenesis of dentin-containing tooth [GO:0042475]; positive regulation of apoptotic process [GO:0043065]; positive regulation of apoptotic signaling pathway [GO:2001235]; positive regulation of excitatory postsynaptic potential [GO:2000463]; positive regulation of fibroblast proliferation [GO:0048146]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of miRNA transcription [GO:1902895]; positive regulation of myelination [GO:0031643]; positive regulation of neural precursor cell proliferation [GO:2000179]; positive regulation of neuron death [GO:1901216]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of neuron projection development [GO:0010976]; positive regulation of odontogenesis of dentin-containing tooth [GO:0042488]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein localization to nucleus [GO:1900182]; positive regulation of Rho protein signal transduction [GO:0035025]; positive regulation of synaptic transmission, cholinergic [GO:0032224]; positive regulation of synaptic transmission, glutamatergic [GO:0051968]; presynaptic modulation of chemical synaptic transmission [GO:0099171]; regulation of cell death [GO:0010941]; regulation of gene expression [GO:0010468]; regulation of reactive oxygen species metabolic process [GO:2000377]; response to wounding [GO:0009611]; Rho protein signal transduction [GO:0007266]; sensory perception of pain [GO:0019233]; signal transduction [GO:0007165]; skin development [GO:0043588] |
11756426_Our data suggest that p75(NTR) may be present as a preformed trimer that binds amyloid beta to induce receptor activation, and support the hypothesis that p75(NTR) activation by amyloid beta is causally related to Alzheimer's disease 11771768_The p75 neurotrophin receptor, expressed in lumbar motor neurons in symptomatic SOD1-G93A transgenic mice, may play a death-signalling role in the pathogenesis of motor neuron degeneration. 11777929_Nerve growth factor-induced p75-mediated death of cultured hippocampal neurons is age-dependent and transduced through ceramide 11872939_P75 was not detected either in retinal ganglion cells or TUNEL-positive cells; Muller cells were p75 immunopositive. It is unlikely that p75 induces apoptosis of rat retinal ganglion cells. 12163480_Rabies virus glycoprotein (RVG) is a trimeric ligand for the N-terminal cysteine-rich domain of this protein 12204258_NRAGE's expression is not restricted solely to areas of p75(NTR) expression 12231242_The mechanisms governing cell survival in facial motoneurons following axotomy are likely the result of interactions between p75NTR, neurotrophins, and aberrant sprouting. 12414813_diverse MAGE family members can interact with the p75 receptor 12422217_P75 interacts with the Nogo receptor as a co-receptor for Nogo, MAG (myelin-associated glycoprotein) and OMgp 12424382_a positive regulator of myelination; enhancement of myelin formation by endogenous brain-derived neurotrophic factor is mediated by the p75NTR receptor 12573711_In sod-1 transgenic mice, spinal motoneurons, which normally do not contain the neurotrophin receptor p75 receptor, express this receptor during the progress of the disease. 12595234_p75NTR is at highest levels in cerebellar areas where foliation is altered in BDNF and NT3 mutants. Mutation of p75NTR in BDNF heterozygotes results in defects in foliation and in Purkinje cell morphologic development. 12655507_Lack of the p75 receptor in knockout mice results in neuronal dorsal root ganglia cells that are resistant to nerve injury, pointing to a role for the p75 receptor in apoptosis. 12675131_Activation of TrkA receptors may overcome p75 receptor-mediated inhibitory effects on pyruvate dehydrogenase expression in cholinergic cells, but make expression of choline acetyltransferase and cell differentiation more susceptible to beta-amyloid. 12722832_Apoptosis of Schwann cells in axotomized peripheral nerve is mediated predominantly through p75(NTR) signaling and initiated via endogenously produced NGF. 12794303_Lesion volume and neuronal cell counts between p75NTR null and wild-type mice were similar after chemical striatal lesions, indicating that expression of p75NTR within reactive astrocytes is not a key factor in preventing neuronal cell death. 12860969_Blocking overactivation of Rho GTP-Binding protein after spinal cord injuries protects cells from p75NTR-dependent apoptosis. 14506941_p75NTR is not directly involved in the mechanism leading to motoneuron degeneration. 14572455_The p75NTR receptor plays an important regulatory role in early neuroglial and immune activation. 14593116_necdin and MAGE-G1 target both E2F1 and p75 to regulate cell viability during brain development. 14638693_p75NTR shedding is abrogated in cells isolated from adam17-/- mice 14724254_Regeneration of the injured spinal cord is not improved in p75NTR knockout mice. 14751775_In dorsal root ganglia neurons with blocked p75NTR, high-dose NGF inhibited neurite outgrowth, implying that p75NTR is not involved in suppression of neurite outgrowth. 14985432_p75NTRExonIV-/- mice express a p75NTR gene product that encodes a truncated protein; aspects of the p75NTRExonIV-/- phenotype may reflect a gain-of-function mutation 15015775_p75NTR activation has diverse effects on neuroblastomas. 15126035_The co-occurrence in p75NTR null mice of a 35% reduction in geniculate ganglion taste neurons and a shortfall of taste buds is consistent with gustatory innervation prompting taste receptor cell differentiation. 15128850_Neurotrophins regulate filopodial dynamics by depressing the activation of RhoA that occurs through p75NTR signaling 15365620_In experimental diabetes the p75 neurotrophin receptor is involved in neuronal dorsal root ganglions cell body shrinkage without loss of neuronal DRG cells. 15496460_NGF controls dendrite development in hippocampal neurons by binding to p75NTR and modulating the cellular targets of Notch 15558233_Plasma p75(NTR) immunoreactivity may serve as an early indicator of neuronal dysfunction or damage in diabetes. 15668238_These results establish neurotrophin receptor interacting factor (NRIF) as an essential component of the p75 apoptotic pathway. 15754321_p75(NTR) mediates survival of myoblasts prior to differentiation 15866043_neither BDNF nor NT-3 utilizes p75 for anterograde transport in adult rodent retinal ganglion cells 15883381_The neurotrophin receptor p75NTR modulates long-term depression and regulates the expression of AMPA receptor subunits in the hippocampus. 15966860_results indicate that the p75NTR-ceramide signalling pathway activates the rate of Abeta generation in an age-dependent fashion 16006137_P75 receptor is required for both resting and NGF-induced Ncam1 expression. Activation of TrkA receptors alone does not upregulate Ncam1. The normal Ncam1 expression depends on the relative ratio of TrkA and p75 receptors. 16025106_results show a crucial role for proBDNF-p75(NTR) signaling in long-term depression and its potential mechanism 16281908_intracellular apoptosis pathways in adult oligodendrocytes do not require p75(NTR) activated signal transduction in the absence of T-lymphocytes and T-lymphocyte derived cytokines 16343477_p75(NTR) and TrkA are involved in the regulation of mouse RGC number in the early phase of retinal development. 16356734_Data suggest that p75 plays a role in the actions of nerve growth factor and neurotrophin-3, but does not affect actions of brain-derived neurotrophic factor. 16402023_In contrast to its role in light-induced photoreceptor degeneration, p75NTR is not essential for apoptosis in the rd mouse. 16539652_Our results reveal a hitherto unknown contribution of NGF/p75NTR to dendritic and synaptic plasticity through Neurogenin 3 signalling. 16555252_Loss of p75NTR expression accompanies maligant progression in TAg-RB retinoblastoma mouse model and is corellated with loss activated caspase-3 expression. 16564506_neurotrophin receptor p75 may play a significant role in the maintenance of cochlear function, and that mice carrying a mutation in the p75 gene could be a good animal model of early onset progressive hearing loss. 16723502_In cultured hippocampal neurons, the capacity of Nerve Growth Factor to promote axon elongation is mostly mediated by p75NTR, and the activation of this pathway leads to the inactivation of GSK-3beta. 16762005_Although activated hepatic stellate cells may produce the factors that regulate liver regeneration, the de novo NGF production by regenerating hepatocytes may induce the death of HSCs via p75NTR, leading to termination of hepatic regeneration. 16887237_In p75NTR-expressing cells, p75NTR regulates cholesterol synthesis through regulation of HMG-CoAred and 7dhcred expression. Thus, least some adult neurons retain the ability to synthesize cholesterol. 17161916_The data show that increased expression of the TNF-p75 receptor following induction of focal cerebral ischemia in mice can be attributed to expression in activated microglial cells and blood-borne macrophages. 17287525_Conserved elements were found in the 25 kb upstream sequences of the mouse p75NTR gene. Only 1 of these, a proximal region rich in Sp1 sites, responds to changes in hypo-osmolarity. 17395831_results identify signaling from p75NTR to Rho as a mechanism for the regulation of hepatic stellate cell differentiation to regeneration-promoting cells that support hepatocyte proliferation in the diseased liver 17576803_p75(NTR)is up-regulated after tissue injury, blocks fibrinolysis by down-regulating the serine protease, tissue plasminogen activator, and up-regulating PAI-1. 17706365_We conclude that the p75(NTR) receptor is integral to neuronal cell survival and endogenous reparative mechanisms after compressive/contusive SCI. 17716721_p75NTR plays a dual role by promoting hyperreactivity of sensory nerves and airway inflammation. 17904861_Our findings support a role for TACE in the proteolytic cleavage of p75NTR and light-induced apoptosis. 17916431_Taken together, the results elucidate a novel mechanism whereby Egr proteins can directly modulate p75(NTR) expression and signaling in vivo. 17928338_We conclude that although p75(NTR) is a receptor for soluble RVG in transfected cells of heterologous expression systems, an RVG-p75(NTR) interaction is not necessary for RV infection of primary neurons. 17964555_pro-NGF-induced, p75NTR-sortilin-mediated neuronal death as a critical aspect of nerve injury-induced death in the dorsal root ganglion 18006062_TLR4 signaling can induce expression of NGF and p75 (NTR) on DCs via activation of p38 MAPK and NF-kappaB pathways, suggesting that NGF may be involved in the pathogenesis of inflammatory diseases. 18006858_Tumor necrosis factor signaling via p75 neurotrophin receptor reduces vein graft neointimal hyperplasia through mechanisms involving reduction of adhesion molecule expression and endothelial cell apoptosi 18032650_The p75 receptor is required only for neurotrophin-dependent expansion of the GABAergic, not the cholinergic, population. 18032673_p75(NTR) is an important functional modulator of Sema3A activity; spinal ganglia neurons are more sensitive to Sema3A growth cone collapse activity in the absence of p75NTR. 18055214_Thus p75(NTR) activates neuronal death pathways in conditions where the balance of normal RIP is shifted toward extracellular domain cleavage due to increased metalloprotease activity, decreased TrkA activity or compromised gamma-secretase activity. 18182498_Nogo receptor antagonizes p75NTR-dependent motor neuron death 18189308_p75(NTR) expression in micturition reflexes is present constitutively and modified by bladder inflammation 18196575_p75(NTR) suppresses progression of TAg-RB murine retinoblastoma and is a potential target for therapy. 18349525_Immunostaining for p75 was noted in articular chondrocytes. 18371392_Report neurotrophin receptor-mediated death of misspecified neurons generated from embryonic stem cells lacking Pax6. 18373618_The data demonstrate TNFR-mediated apoptotic death of dentate granule neurons utilizing both TNFRs and suggest a TNFp75R-mediated apoptosis in the absence of normal TNFp55R activity. 18374455_results suggest that overstimulation of p75(NTR)-positive progenitors during early life might result in depletion of the stem cell pool and thus a more rapid decline in basal neurogenesis 18400893_Neuronal death correlated with Abeta(1-42) peptide-stimulated accumulation of the death-inducing p75(NTR) C-terminal fragment generated by extracellular metalloprotease cleavage of full-length p75(NTR). 18430612_Thus, p75 regulates both the growth and stability of cardiac sympathetic fibers. 18602709_Involvement of NF-kappaB signaling pathway in p75(NTR)-mediated apoptosis may partially account for the PrP106-126-induced neurotoxicity in N2a cells. 18663291_P75(NTR) has a remarkable influence on spiral ganglion neurite growth behavior, but p75(NTR) does not seem to be essential for the development of basic hearing function in the first 3 postnatal weeks 18689502_Data suggest that intact p75(NTR) signaling may play a role in modulating pulmonary vasoconstriction induced by endothelin-1 and by active store depletion. 18709654_Pro-NGF, as well as mature NGF, binding to p75(NTR) plays an important role in inflammation-induced hyperalgesia. Interference in the binding may provide a therapeutic approach for the treatment of inflammatory pain. 18717734_Mice lacking the p75 neurotrophin receptor have 25% fewer neuroblasts and 50% fewer newborn neurons in the dentate gyrus, coincident with increased cell death and a smaller granular cell layer and dentate gyrus, than those of p75(NTR+/+) mice. 18772898_NRAGE plays an important role in apoptotic-signaling in vivo. 19026664_p75NTR plays an important role in the regeneration of injured peripheral nerves. 19046947_This study provides the first comparative proteomic assessment of sympathetic ganglia and sheds new light onto the phenotypic changes that occur as a consequence of a loss of p75NTR expression in adult mice. 19072833_Whereas fibrosis develops in the absence of p75(NTR) signaling, the dominant effects of loss of p75(NTR) ligand-mediated events are the retardation of liver fibrosis resolution via regulation of hepatic myofibroblast proliferation and apoptosis. 19114089_The findings suggest that p75NTR activity plays a critical role in the development of opioid analgesic tolerance but not in the induction or the expression of opioid physical dependence. 19193386_bone marrow stromal cells treatment benefits on experimental autoimmune encephalomyelitis mice may be fostered by decreasing the cellular expression of proNGF and p75, thereby reducing oligodendrocyte death. 19210757_These results illustrate the selective expression of p75 in basal pterygium, conjunctival and limbal epithelium, while staining was absent in adult and foetal central cornea. 19291307_Data show that elevated P75NTR expression causes death of engrailed-deficient midbrain dopaminergic neurons by Erk1/2 suppression. 19360854_results suggest that the effects of p75(NTR) on hippocampal function are likely to be mediated indirectly, by its actions on basal forebrain cholinergic neurons 19376199_data suggest that in the brain of mdx mice, an upregulation of the nerve growth factor-nerve growth factor receptors (TrkA and p75) system might be involved directly in the angiogenic response taking place in muscular dystrophy 19389705_the relative level of proNGF receptors determines whether NGF precursor exhibits neurotrophic or apoptotic activity 19403809_Signaling through the p75 pathway causes sympathetic neurons to fire in a phasic pattern showing marked accommodation. 19416837_p75 reduces beta-amyloid-induced sympathetic innervation deficits in an Alzheimer's disease mouse model 19508690_We found that the expression level of p75 neurotrophin receptor mRNA and protein was strongly reduced in developing sciatic nerves in MAL-overexpressing mice. 19553472_Results demonstrate that NGF acts through its low-affinity receptor p75(NTR) in a developmentally regulated signaling pathway necessary to myogenic differentiation and muscle repair in vivo. 19720059_p75NTR is expressed in cells endowed with wide pluripotency. 19824062_CGRP and p75 NTR in L5 dorsal root ganglia neurons was upregulated in injured nerves; however, p75 NTR inhibitory antibody decreased CGRP and p75 NTR expression. 20047901_p75NTR-deficient Schwann cells enhance neurotrophin-dependent regeneration in vitro. Schwann cell p75NTR restricts neurotrophin availability to the extent that it prevents spontaneous sensory axon regeneration into the spinal cord. 20190098_The lack of p75(NTR) resulted heterogeneous ventricular sympathetic innervation, altered beta-adrenergic receptor expression, and rhythm instability in deficient mice. 20348920_The data of this study indicated that p75NTR maintains the specificity of neural connectivity by preventing inappropriate sprouting onto myelinated tracts and provide a physiological explanation for myelin inhibition after neural injury. 20421303_Results demonstrate that p75NTR-mediated activation of JNK3 is required for up-regulation of TACE, which promotes receptor proteolysis, leading to prolonged activation of JNK3 and subsequent apoptosis in sympathetic neurons. 20466052_Data do not support a role of p75NTR as a death inducer in primary auditory neurons (PAN) but show its crucial role in protecting PANs. 20530577_Data suggest that generation of the soluble p75NTR intracellular domain by Trk-induced cleavage plays a fundamental role in Trk-dependent signaling events. 20566851_Data show that different streams of AD neurodegeneration are related in complex ways to TrkA versus p75NTR signaling. 20584617_these data suggest that in addition to its role as a co-receptor, p75NTR may provide an obligate parallel neurotrophin-activated inhibitory pathway that broadly sensitizes neurons to inhibitory cues. 20692240_p75NTR contributes to N-methyl-D-aspartate receptor-mediated apoptosis in the neonatal brain; an increase in lesion size in female animals after excitotoxic brain injury suggests that in females p75NTR may play a dual role. 20696380_The p75 neurotrophin receptor restricts postsynaptic density (PSD) formation, suggesting important roles for antagonistic NGF-TrkA and p75 signaling pathways during retrograde control of synapse establishment. 20875398_nearly all hippocampal dentate granule cells of adult wild-type and transgenic Alzheimer's disease-model mice localize the p75 neurotrophin receptor along with the expected SSTR3 to their primary cilia. 20955473_Data show that TrkB.T1 regulates the function/structure of mature pyramidal neurons, and that the ratio of expression levels of p75(NTR) and TrkB.T1 plays a role in modulating dendritic architecture and synaptic plasticity in the adult hippocampus. 20961458_p75NTR is expressed by adult dentate progenitor cells and is an important receptor promoting the proliferation/maturation of neurons and glia. 21034655_p75(NTR)-dependent JNK-caspase-3 pathway is involved in DRG B-cell loss after nerve injury and JNK is not the unique upstream of c-jun activation. 21105874_A stress-induced decline in the monosaccharide O-linked N-acetylglucosamine content of Sp1 drives expression of p75NTR. 21142793_CD271/p75(NTR) inhibits the differentiation of mesenchymal stem cells into osteogenic, adipogenic, chondrogenic, and myogenic lineages. 21385399_protein levels of NGF and its receptors in uteri and trkA mRNA levels in DRG were higher in adenomyosis mice compared with those in controls, and these levels increased with the severity of disease 21402589_Nephrin directly interacts with p75NTR and reveals an important role for nephrin in murine cardiac development by permitting survival of cardiovascular progenitor cells. 21646052_Norepinephrine content is elevated in the base of the p75NTR-deficient ventricle compared to wildtype, but levels are identical below the site of occlusion. 21881600_The p75 has a critical role in axon growth inhibition in specific neuronal tracts. 21904642_Expression of intracellular domain of p75NTR can enhance or attenuate oxidative induction of apoptosis. Variability of the effects of p75ICD is not related to variability of the effects of its Chopper domain. 21922519_The data of this study did not support an apoptotic role but indicate a broad antineurotrophic role of p75(NTR) in the cholinergic basal forebrain. 21978521_The results of this study suggest that loss of p75(NTR) leads to changes in hippocampal cholinergic signaling, which may be involved in regulation of stress-enabled hippocampal LTD and in modulating behaviors related to stress and anxiety. 22055192_Oxygen-dependent cleavage of the p75 neurotrophin receptor triggers stabilization of HIF-1alpha to control hypoxic response. 22183269_p75NTR may not play a main role in Abeta internalization at the concentrations tested but is responsible for Abeta induced toxicity in primary neurons 22198221_Propofol induces apoptosis in developing neurons in vivo and in vitro, implicating a role for p75(NTR) and the downstream effector RhoA kinase. 22460790_These results identify p75(NTR) as a unique player in glucose metabolism. 22503986_the results show that specific binding of NGF to p75(NTR) mediates neuronal cell survival. 22520925_Letter: NGF-p75 and neuropsin/KLK8 pathways may cooperate in regulation of epidermal homeostasis in inflamed skin. 23065828_p75(NTR) inhibits the healing of ischemic lower limb wounds in diabetes mellitus and promotes ST2 expression. 23105113_although the intracellular domain of sortilin does not contribute to p75(NTR) binding, it does regulate the rates of p75(NTR) cleavage, which is required to mediate pro-neurotrophin-stimulated cell death. 23260665_Dissociation of c-Jun kinase (JNK) and caspase-3 activities indicated that JNK is necessary but not sufficient for p75(NTR)-mediated cell death. 23307658_This study demonistrated that p75 plays an important role in regulating the firing-to-silence transition during the phasic firing period of the spontaneous firing of Purkinje cells. 23322532_an absence of p75NTR by sensory afferents leads to an increase of terminal arborization in certain NGF-rich peripheral tissues 23471969_the juxtamembrane region of p75(ICD) acts to cause a conformational change within the extracellular domain of TrkA. 23559013_A DR6/p75(NTR) complex is responsible for beta-amyloid-induced cortical neuron death. 23633509_proNGF selectively promotes the growth of neurites from a subset of NGF-responsive neurons by a p75(NTR)-dependent mechanism during postnatal development when the axons of these neurons are ramifying within their targets in vivo 23642664_Integrin beta-1(+) and p75NTR(+) cells play an important role in wound healing process 23785138_transcriptional activation of p75(NTR) is under circadian regulation in the nervous system and peripheral tissues 23838122_Results suggest that the proNGF/p75NTR signal plays a key role in the regulation of NSCs' behavior. 23940017_Tests the hypotheses that pharmacologic modulation of p75(NTR) signaling will promote neural progenitor survival and proliferation, and improve outcomes of traumatic brain injury (TBI). 23959707_The data illustrates that lack of the individual components of the proBDNF/BDNF signaling complex has unexpected and often opposing effects on specific behavioral traits, suggesting that ligands and receptors may participate in separate signaling cascades independent of each other. 24013014_The p75 neurotrophin receptor is required for sympathetic denervation of peri-infarct myocardium. 24190996_p75NTR is expressed most strongly in the peripheral nerve, where it is important for nerve development. 24224958_This study aimed to examine the roles of mature neurotrophin-3 (NT3), pro-NT3 and p75 neurotrophin receptor (P75(NTR)) in photoreceptor degeneration 24599786_p75NTRE knockout mice display an enhanced volume of the dentate gyrus (DG). However, adult neurogenesis within the adult DG was unaffected in both adult and aged knockout mice. Specific alterations in behavior was also observed in knockout mice. 24608438_p75NTR-mediated nerve loss may contribute to the impaired glucose counterregulation seen in type 1 diabetes. 24685135_p75(NTR) governs neuronal polarity, determining pattern and assembly of neuronal circuits in adult hippocampus and cortical development. 24939843_data suggest a novel mechanism linking oxidative stress to ligand-independent cleavage of p75(NTR), resulting in axonal fragmentation and neuronal death 24961238_p75 regulates gustatory neuron axon branching and tongue innervation patterns during taste system development. 25165859_Rabies virus may employ p75NTR-dependent transport as a fast mechanism to facilitate movement to the central nervous system 25180603_p75(NTR) mediates synaptic, learning, and memory dysfunction in Huntington's disease 25253694_p75 plays a role in maintaining normalcy of Purkinje cell firing in the cerebellum in part by activating Rac1 in synaptic compartments and modulating SK channels 25443284_Its reduction ameliorates cognitive deficits in Alzheimer disease. 25662808_uterine BDNF and NGFR are regulated by estradiol 25720379_The activities of multifunctional receptors, such as p75(NTR), can be tuned into narrower activity profiles by cell-type-specific differences in intracellular processes, such as proteolytic cleavage, leading to very different biological outcomes. 25879397_Genetic mutation of the neurotrophin interaction site of p75NTR strongly limits post-traumatic brain cell death in mice. 25898755_mice. Thus, immunization with both fragments of p75 receptor is believed to be an effective tool for the development of new drugs against Alzheimer's disease. 26066836_In E-Reeler retinas, p75(NTR) was increased in both retinal bipolar cells and retinal ganglion cells. These cells overexpress NGF and p75(NTR) as a protective endogenous response to Reelin deprivation. 26073142_Induction of colonic inflammation confirms local increases in NGF mRNA/protein levels, which coincide with myenteric neurons. 26134656_The results of this study proposed that atenuating EC network performance, the proBDNF-p75NTR signaling cascade reduces the probability of epileptogenesis. 26640617_the present studies demonstrate that neither induced expression of p75ICD or its active fragments nor overexpression of p75NTR results in altered expression or activity of Complex II. 26663679_75NTR is involved in downstream signaling events that mediate BMP7-induced dendritic growth in sympathetic neurons. 26700963_Studies indicate that neurotrophin receptor p75(NTR) mediates Huntington's disease-associated synaptic and memory dysfunction. 26947127_Results suggest a role for p75 in augmenting survival signaling in the striata of Hdh+/Q175 mice, and that the loss of this early pro-survival signaling leads to earlier onset of striatal dysfunction/degeneration 26984409_Identify a novel signaling pathway in hepatocytes triggered by ligand-activated p75NTR that via p38 MAPK and caspase-3 mediate the activation of SREBP2. This pathway may regulate LDLRs and lipid uptake particularly after injury or during tissue inflammation accompanied by an increased production of growth factors, including NGF and pro-NGF. 26987954_Study showed that BDNF signaling through its low-affinity receptor p75 is compromised in the forebrain of En2-/- mice; BDNF signaling deficits might be involved in the neurodevelopmental alterations observed in the En2-/- forebrain 27194337_Neuronal death induced by p75NTR requires Cys259. 27348017_Results suggest that p75 regulates gene and protein expression that limits the distal atrophy after sciatic nerve injury, thereby regulating axonal growth and functional recovery. 27434667_Here the authors demonstrate a novel function for p75(NTR) in regulating proper cell cycle exit of neuronal progenitors in the developing rat and mouse external granule layer, which is stimulated by proneurotrophin-3. In the absence of p75(NTR), cerebellar granule cell progenitors continue to proliferate beyond their normal period, resulting in a larger cerebellum that persists into adulthood, with consequent motor defici 27498053_Neurotoxic effect of gp120 is mediated by p75NTR. 27683113_Data from murine model indicate that NGFR plays an important role in the pathogenesis and progression of oral squamous cell carcinoma via regulation of ESM1. 27746130_Peri-synaptic glia uses p75NTR to clear proBDNF secreted from neurons. Glial p75NTR is essential for long term potentiation maintenance. 27775873_NRADD deletion results in an increase in the proportion of dorsal root ganglion neurons expressing p75NTR 28127619_Dermal CD271+ cells are closely associated with wound healing. 28266720_deletion of p75NTR in mice leads to physiological and morphological changes in the amygdala and altered behavior that is linked to the limbic system 28522736_We show that myelin-associated glycoprotein or CNS myelin, in general, inhibit rodent Schwann cell migration and induce their death via cleavage of the neurotrophin receptor p75. 28869759_Results suggest that the colocalization of beta-site amyloid precursor protein cleaving enzyme-1 and amyloid precursor protein is increased in the presence of both amyloid beta and p75 in cortical neurons. 29045838_p75 is required for the development of the nonpeptidergic nociceptor lineage by fine-tuning Ret-mediated trophic support. 29150821_These findings shed light on the role of Cd271 in wound healing and may provide new therapeutic approaches for curing burn wounds of the skin. 29166838_antibodies raised against NGF, TrkA, and p75 (also known as CD271) were used to explore the expression of these antigens in the non-decalcified young mouse femur. 29569362_p75NTR-/- mice display elevated cell death during typical windows of naturally occurring cell death in TrkA, TrkB, and TrkC expressing sensory neurons, resulting in substantially lower numbers of sensory neurons postnatally. p75NTR-dependent survival within the typical period of developmental cell death further supports a role for p75NTR in neurotrophic signaling sensitization. 29584618_These results unveil a dual role of proNGF/p75 signaling in bladder function under pathological conditions with a CNS effect overriding the peripheral one. 29604364_study suggests a detrimental role of hippocampal p75NTR up-regulation in synaptic and cognitive impairments after sepsis. Data uncovers a new role of p75NTR and supports the possibility of treating synaptic and memory impairments in sepsis-associated encephalopathy by pharmacological modulation of p75NTR function. 29860333_this study assessed the feasibility of utilizing PET imaging with the TSPO tracer, [18F]PBR06, to detect activated microglia in two HD mouse models and to monitor response to treatment with LM11A-31, a p75NTR ligand known to reduce neuroinflammation in HD mice. 29867188_Results support the pathological role of p75NTR and proNGF in tau pathology and indicate that genetic or pharmacological (e.g., p75ECD) modulation of p75NTR may be a valid therapeutic approach in clinical application for frontotemporal lobar degeneration-tau and perhaps other tauopathies. 30044969_In CGNs lacking RIP2, more TRAF6 is associated with p75(NTR), leading to increased JNK-dependent apoptosis. 30092219_The p75NTR enhances HIF1A/HIF2A-dependent signaling in glioma. 30242146_Study shows that removing the p75 neurotrophin receptor from cholinergic basal forebrain neurons induces a significant impairment in fear extinction consolidation. 30374941_the contribution of cholinergic basal forebrain neuronal p75(NTR) to the progression of Alzheimer's disease (AD) by removing p75(NTR) from cholinergic neurons in the APP/PS1 familial AD mouse strain. 30597659_Neurotrophin signaling via the p75 nerve growth factor receptor alters innate immune cell behavior and contributes to cell function and neuronal structure in Toxoplasma infection-induced neuroinflammation. 30936240_p75NTR expression in cortical inhibitory parvalbumin cells decreases between the 2d and 4th postnatal week when synapses are form. p75NTR expression modulates the timing of maturation of PV cell connectivity in adolescent cortex.PV cells still express p75NTR in adult mouse cortex of both sexes and its activation is sufficient to destabilize PV cell connectivity and restore cortical plasticity after monocular deprivation. 31073629_the p75 neurotrophin receptor can be modulated using LM11A-31, which prevents diabetes-induced retinal vascular permeability in mice via inhibition of inflammation and the RhoA kinase pathway 31085607_Male mice lacking p75NTR were resistant to the detrimental effects of sleep deprivation on hippocampal plasticity. Sleep deprivation increased p75NTR expression and its interaction with phosphodiesterase. 31108130_Exercise significantly increased BDNF expression and decreased the expression of p75 in both SAMR1 and SAMP8 strains of mice. 31296846_p75NTR signaling influences a network of genes involved in lipid metabolism in liver and hepatocyte cells. 31488566_The phenotype of this strain demonstrates that p75(NTR) regulates multiple aspects of brain development. 31514753_The p75NTR neurotrophin receptor is required to organize the mature neuromuscular synapse by regulating synaptic vesicle availability. 31515449_The wild type (wt) p75(NTR) and mutated (mut) p75(NTR) differ in their partitioning in cholesterol-rich membrane regions upon nerve growth factor (NGF) stimulation: this is the origin of the ability of wt p75(NTR), but not of mut p75(NTR), to mediate immature p75 neurotrophin (proNT)-induced apoptosis. 31628183_p75NTR governs the formation and growth of spheroids. 31922317_P75NTR not only promotes osteogenic differentiation. 31995032_The p75 neurotrophin receptor in AgRP neurons is necessary for homeostatic feeding and food anticipation. 31996124_NGF (-198C > T, Ala35Val) and p75(NTR) (Ser205Leu) gene mutations are associated with liver funct |
ENSG00000064300 |
NGFR |
47.281368 |
0.0207051613 |
-5.593866 |
0.74581894 |
71.451288 |
0.00000000000000002841882561881921021871303933954747118548762914859266703881957027988391928374767303466796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000008980820577299880735822293346952424589483345322357688012004928168607875704765319824218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.04282954099531 |
1.10373735096235 |
98.6628168060461 |
24.3342440347042 |
| ENSMUSG00000000126 |
216795 |
Wnt9a |
protein_coding |
Q8R5M2 |
FUNCTION: Ligand for members of the frizzled family of seven transmembrane receptors (Probable). Functions in the canonical Wnt/beta-catenin signaling pathway (By similarity). Required for normal timing of IHH expression during embryonic bone development, normal chondrocyte maturation and for normal bone mineralization during embryonic bone development (PubMed:16818445). Plays a redundant role in maintaining joint integrity (PubMed:16818445). {ECO:0000250|UniProtKB:O42280, ECO:0000269|PubMed:16818445, ECO:0000305}. |
Developmental protein;Disulfide bond;Extracellular matrix;Glycoprotein;Lipoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:216795; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; frizzled binding [GO:0005109]; receptor ligand activity [GO:0048018]; anatomical structure development [GO:0048856]; canonical Wnt signaling pathway [GO:0060070]; cartilage development [GO:0051216]; cell fate commitment [GO:0045165]; chondrocyte differentiation [GO:0002062]; embryonic forelimb morphogenesis [GO:0035115]; embryonic skeletal system morphogenesis [GO:0048704]; epithelial cell differentiation [GO:0030855]; negative regulation of cartilage development [GO:0061037]; negative regulation of cell death [GO:0060548]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of chondrocyte differentiation [GO:0032331]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043154]; neuron differentiation [GO:0030182]; positive regulation of epithelial cell differentiation [GO:0030858]; positive regulation of smoothened signaling pathway [GO:0045880]; smoothened signaling pathway [GO:0007224] |
16818445_Embryos double-heterozygous for Wnt9a and beta-catenin show reduced Ihh expression. 20083111_The mRNA levels of Wnt3, 6, and 9a were significantly upregulated in the intestinal epithelial cells by Salmonella. 24778176_These studies reveal how carrageenan exposure can lead to transcriptional events in colonic epithelial cells through decline in arylsulfatase B activity, with subsequent impact on C4S, galectin-3, Sp1, and Wnt9A 26771085_Wnt9a is expressed in the embryonic pancreas and that its deficiency enhances activation of the endocrine transcriptional program and increases the number of endocrine cells at birth. 29440280_Tubular cells of several mouse models of chronic kidney disease expressed high levels of Wnt9a that colocalized with the senescence-related protein p16(INK4A). Ectopic expression of Wnt9a after ischemia-reperfusion injury induced beta-catenin activation and exacerbated renal fibrosis. This was inhibited by shRNA. 29869452_The synergistic effects of TGFbeta2, WNT9a, and FGFR4 were responsible for attenuating of the differentiation of aging satellite cells during skeletal muscle development. 30801848_Wnt9a genetically interacts with Ror1 and Ror2. Wnt9a-/- single mutants display a mild shortening of the long bones. Wnt9a-/- Ror double mutants showed an aggravated skeletal dysplasia phenotype. 32800757_Pdgfra regulates multipotent cell differentiation towards chondrocytes via inhibiting Wnt9a/beta-catenin pathway during chondrocranial cartilage development. 33990546_Loss of the WNT9a ligand aggravates the rheumatoid arthritis-like symptoms in hTNF transgenic mice. 35560108_Mice Lacking Wnt9a or Wnt4 Are Prone to Develop Spontaneous Osteoarthritis With Age and Display Alteration in Either the Trabecular or Cortical Bone Compartment. |
ENSG00000143816 |
WNT9A |
152.739605 |
0.3415128153 |
-1.549988 |
0.21700394 |
50.250943 |
0.00000000000135289668108355284665145473430939164656561235489107275498099625110626220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000264535587243448970788045795981269768006038933805257329368032515048980712890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
78.5931247994597 |
11.8662648748684 |
230.132285757831 |
24.4264232860236 |
| ENSMUSG00000000202 |
72014 |
Btbd17 |
protein_coding |
Q9DB72 |
|
Glycoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:72014; |
extracellular region [GO:0005576] |
|
ENSG00000204347 |
BTBD17 |
26.830906 |
4.0692687092 |
2.024770 |
0.64371924 |
9.561622 |
0.00198687496796304870899652073035213106777518987655639648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0084560155104814634097198933204708737321197986602783203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.0656439939093 |
21.7041082239824 |
11.5661184741334 |
4.18631220435892 |
| ENSMUSG00000000276 |
56077 |
Dgke |
protein_coding |
Q9R1C6 |
FUNCTION: Membrane-bound diacylglycerol kinase that converts diacylglycerol/DAG into phosphatidic acid/phosphatidate/PA and regulates the respective levels of these two bioactive lipids (PubMed:11287665). Thereby, acts as a central switch between the signaling pathways activated by these second messengers with different cellular targets and opposite effects in numerous biological processes (PubMed:11287665). Also plays an important role in the biosynthesis of complex lipids (By similarity). Displays specificity for diacylglycerol substrates with an arachidonoyl acyl chain at the sn-2 position, with the highest activity toward 1-octadecanoyl-2-(5Z,8Z,11Z,14Z-eicosatetraenoyl)-sn-glycerol the main diacylglycerol intermediate within the phosphatidylinositol turnover cycle (PubMed:11287665, PubMed:19744926). Can also phosphorylate diacylglycerol substrates with a linoleoyl acyl chain at the sn-2 position but much less efficiently (By similarity). {ECO:0000250|UniProtKB:P52429, ECO:0000269|PubMed:11287665, ECO:0000269|PubMed:19744926}. |
ATP-binding;Cytoplasm;Kinase;Lipid metabolism;Membrane;Metal-binding;Nucleotide-binding;Reference proteome;Repeat;Transferase;Transmembrane;Transmembrane helix;Zinc;Zinc-finger |
PATHWAY: Lipid metabolism; glycerolipid metabolism. {ECO:0000269|PubMed:11287665, ECO:0000269|PubMed:19744926}. |
|
mmu:56077; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; nucleoplasm [GO:0005654]; ATP binding [GO:0005524]; diacylglycerol kinase activity [GO:0004143]; kinase activity [GO:0016301]; metal ion binding [GO:0046872]; diacylglycerol metabolic process [GO:0046339]; intracellular signal transduction [GO:0035556]; lipid phosphorylation [GO:0046834]; modulation of chemical synaptic transmission [GO:0050804]; phosphatidic acid biosynthetic process [GO:0006654]; phosphatidylinositol biosynthetic process [GO:0006661]; protein kinase C-activating G protein-coupled receptor signaling pathway [GO:0007205] |
16137646_A regulatory role is suggested for DGK epsilon, COX-2, and catecholamine signaling during kindling epileptogenesis. 18702510_DGKepsilon plays a significant role in determining the enrichment of GPInsP n with 20:4 and that there is a pathway for the selective translocation of arachidonoyl phosphatidic acid from the plasma membrane to the endoplasmic reticulum 22511757_Inhibition of lipid signaling enzyme diacylglycerol kinase epsilon attenuates mutant huntingtin toxicity. 26887830_Our results unveil an unexpected role of Dgke in the induction of cyclooxygenase-2 and in the regulation of glomerular prostanoids synthesis under stress 28246337_DGKepsilon plays a role in glucose and energy homeostasis by modulating lipid metabolism in skeletal muscle 31607681_Mice lacking DGKepsilon show increased beige adipogenesis in visceral white adipose tissue after long-term high fat diet in a COX-2- dependent manner. 32291315_Diacylglycerol kinase zeta promotes actin cytoskeleton remodeling and mechanical forces at the B cell immune synapse. 33986189_Loss of diacylglycerol kinase epsilon causes thrombotic microangiopathy by impairing endothelial VEGFA signaling. 35616094_Diacylglycerol kinase epsilon protects against renal ischemia/reperfusion injury in mice through Kruppel-like factor 15/klotho pathway. |
ENSG00000153933 |
DGKE |
194.800382 |
0.2712019958 |
-1.882560 |
0.45624914 |
15.730976 |
0.00007301858094134145783001693663649689369776751846075057983398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004477532306354722077372521038540753579582087695598602294921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
87.691050431042 |
28.1966794270856 |
323.342201762823 |
77.3755136705129 |
| ENSMUSG00000000392 |
14089 |
Fap |
protein_coding |
P97321 |
FUNCTION: Cell surface glycoprotein serine protease that participates in extracellular matrix degradation and involved in many cellular processes including tissue remodeling, fibrosis, wound healing, inflammation and tumor growth. Both plasma membrane and soluble forms exhibit post-proline cleaving endopeptidase activity, with a marked preference for Ala/Ser-Gly-Pro-Ser/Asn/Ala consensus sequences, on substrate such as alpha-2-antiplasmin SERPINF2 and SPRY2. Degrade also gelatin, heat-denatured type I collagen, but not native collagen type I and IV, vibronectin, tenascin, laminin, fibronectin, fibrin or casein. Also has dipeptidyl peptidase activity, exhibiting the ability to hydrolyze the prolyl bond two residues from the N-terminus of synthetic dipeptide substrates provided that the penultimate residue is proline, with a preference for Ala-Pro, Ile-Pro, Gly-Pro, Arg-Pro and Pro-Pro. Natural neuropeptide hormones for dipeptidyl peptidase are the neuropeptide Y (NPY), peptide YY (PYY), substance P (TAC1) and brain natriuretic peptide 32 (NPPB). The plasma membrane form, in association with either DPP4, PLAUR or integrins, is involved in the pericellular proteolysis of the extracellular matrix (ECM), and hence promotes cell adhesion, migration and invasion through the ECM. Plays a role in tissue remodeling during development and wound healing. Participates in the cell invasiveness towards the ECM in malignant melanoma cancers. Enhances tumor growth progression by increasing angiogenesis, collagen fiber degradation and apoptosis and by reducing antitumor response of the immune system. Promotes glioma cell invasion through the brain parenchyma by degrading the proteoglycan brevican. Acts as a tumor suppressor in melanocytic cells through regulation of cell proliferation and survival in a serine protease activity-independent manner. {ECO:0000269|PubMed:10629066, ECO:0000269|PubMed:11330865, ECO:0000269|PubMed:15133496, ECO:0000269|PubMed:21051638, ECO:0000269|PubMed:23710635, ECO:0000269|PubMed:24371721, ECO:0000269|PubMed:9688278}. |
Alternative splicing;Angiogenesis;Apoptosis;Cell adhesion;Cell junction;Cell membrane;Cell projection;Cleavage on pair of basic residues;Disulfide bond;Glycoprotein;Hydrolase;Membrane;Protease;Reference proteome;Secreted;Serine protease;Signal-anchor;Transmembrane;Transmembrane helix |
|
This gene belongs to the serine protease family. The encoded protein is an inducible cell-surface bound glycoprotein specifically expressed in tumor-associated fibroblasts and pericytes of epithelial tumors and has protease and gelatinase activity. The protein plays a role in remodeling of the extracellular matrix (ECM) and may affect tumorigenesis and tissue repair. Alternately spliced transcript variants of this gene are described in the literature (PMID 9139873), but the full-length sequence of these variants is not available. [provided by RefSeq, Apr 2013]. |
mmu:14089; |
anchoring junction [GO:0070161]; apical part of cell [GO:0045177]; basal part of cell [GO:0045178]; cell surface [GO:0009986]; extracellular space [GO:0005615]; lamellipodium [GO:0030027]; lamellipodium membrane [GO:0031258]; peptidase complex [GO:1905368]; plasma membrane [GO:0005886]; ruffle membrane [GO:0032587]; dipeptidyl-peptidase activity [GO:0008239]; endopeptidase activity [GO:0004175]; identical protein binding [GO:0042802]; integrin binding [GO:0005178]; peptidase activity [GO:0008233]; protease binding [GO:0002020]; protein homodimerization activity [GO:0042803]; serine-type endopeptidase activity [GO:0004252]; serine-type peptidase activity [GO:0008236]; angiogenesis [GO:0001525]; cell adhesion [GO:0007155]; endothelial cell migration [GO:0043542]; melanocyte apoptotic process [GO:1902362]; melanocyte proliferation [GO:0097325]; negative regulation of cell proliferation involved in contact inhibition [GO:0060244]; negative regulation of extracellular matrix disassembly [GO:0010716]; negative regulation of extracellular matrix organization [GO:1903054]; positive regulation of execution phase of apoptosis [GO:1900119]; proteolysis [GO:0006508]; proteolysis involved in protein catabolic process [GO:0051603]; regulation of cell cycle [GO:0051726]; regulation of collagen catabolic process [GO:0010710] |
15767544_the enzymatic activity of fibroblast activation protein plays an important role in the promotion of tumor growth 18823010_seprase may have a role in promotion of an invasive phenotype by collagenous matrices in ovarian tumor cells 19920354_FAP depletion inhibits tumor cell proliferation indirectly, increases accumulation of collagen, decreases myofibroblast content, and decreases blood vessel density in tumors. 20225612_FAP may be a potentially useful marker for wound age determination. 20804499_Results suggest that FAP, a product preferentially expressed by TAF, could function as an effective tumor rejection antigen. 21604185_Authors conclude that the proteolytic activity of FAP participates in matrix degradation, but other functions of the protein stimulate increased tumor growth. 21668992_In NIH 3T3 cells overexpressing recombinant mouse FAP, FAP enzymatic activity during matrix production is important for the topographical organization of the ECM fibers. 23712428_The FAP(+) stromal cell may have roles in two adverse consequences of cancer. 25994578_A transgenic mouse model for pulmonary fibrosis was generated. After bleomycin induction, luciferase cDNA under the control of the FAPa promoter presents strong luminescence in the lungs especially; the expression level reflects the degree of the disease. 26305550_Data indicate that indolamine-2,3-dioxygenase (IDO) and Fibroblast activation protein alpha (FAPalpha) were detectable in B16 melanoma tumor-bearing mice. 26663085_In two different models of pulmonary fibrosis, intratracheal bleomycin instillation and thoracic irradiation, the study finds increased mortality and increased lung fibrosis in FAP-deficient mice compared with wild-type mice. 26962859_Mouse FGF-21, however, lacks the FAP cleavage site and is not cleaved by FAP. 27216177_FAP-STAT3-CCL2 signaling in Cancer-associated fibroblasts (CAF) was sufficient to program an inflammatory component of the tumor microenvironment, which may have particular significance in desmoplasia-associated cancers. 27545090_this study shows that FAPalpha-targeted modified vaccinia ankara boosting in combination with cyclophosphamide is an effective approach to improving specific anti-tumor immune responses through overcoming immunosuppression 27590504_Adora2B stimulation promotes FGF2 and CXCL12 expression in FAP-positive melanoma-associated fibroblasts, contributing to the creation of a tumor-promoting microenvironment. 28004985_FAP-vaccinated mice also treated with Cyclophosphamide chemotherapy showed a marked suppression of tumor growth (inhibition ratio =80%) and a prolongation of survival time. 28158223_There was no evidence of compensatory upregulation of other DPP4 family members in influenza-infected FAP-deficient mice. FAP appears to be dispensable in anti-influenza adaptive immunity. 28302482_Taken together, our study suggested that high FAP expression in CAFs is one reason leading to immune checkpoint blockades resistance in CRC patients and FAP is an optional target for reversing immune checkpoint blockades resistance. 29361119_the uptake of the tracer in the joints reflects increased FAP expression during subclinical synovitis and that these joints will show symptoms of inflammation upon disease progression 29410133_The findings provide a novel regulation mechanism on FAPalpha expression and highlight the role of snail/FAPalpha axis as a novel target for melanoma treatment. 30257879_Study identified many potential FAP substrates including ECM and immunoregulatory proteins, several of which were confirmed to have implications for the role of FAP in health and disease. 31324739_Embryonic FAP(+) lymphoid tissue organizer cells generate the reticular network of adult lymph nodes. 31325484_Data demonstrated a novel role for fibroblast activation protein in regulating weight gain and adipocyte hypertrophy. FAP expression by pre-adipocytes provides a cell-intrinsic restriction on adipogenic differentiation, and FAP-mediated collagen remodeling alters lipid metabolism in mature adipocytes. Its loss impairs collagen accumulation, suggesting a novel link between matrix degradation and fibrillogenesis. 32084369_d, these data suggest that Fap restrains the progression of atherosclerosis and may contribute to the sexually dimorphic susceptibility to atherosclerosis by regulating the balance between inflammation and fibrosis 32402085_Deletion of fibroblast activation protein provides atheroprotection. 33053358_Identification of Fibroblast Activation Protein as an Osteogenic Suppressor and Anti-osteoporosis Drug Target. 33667270_Genetic ablation of fibroblast activation protein alpha attenuates left ventricular dilation after myocardial infarction. 34335962_Molecular imaging of fibroblast activation protein after myocardial infarction using the novel radiotracer [(68)Ga]MHLL1. 35011496_Design, Synthesis and Preclinical Assessment of (99m)Tc-iFAP for In Vivo Fibroblast Activation Protein (FAP) Imaging. 35951441_Targeting FAPalpha-expressing hepatic stellate cells overcomes resistance to antiangiogenics in colorectal cancer liver metastasis models. |
ENSG00000078098 |
FAP |
29.809561 |
0.4172865384 |
-1.260890 |
0.46319756 |
7.298432 |
0.00690148040228012217556230467607747414149343967437744140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0244586667025432323574474224869845784269273281097412109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
17.8956469301157 |
5.84487185883654 |
42.8857518357595 |
9.93692453529811 |
| ENSMUSG00000000440 |
19016 |
Pparg |
protein_coding |
Q6GU14 |
FUNCTION: Nuclear receptor that binds peroxisome proliferators such as hypolipidemic drugs and fatty acids. Once activated by a ligand, the nuclear receptor binds to DNA specific PPAR response elements (PPRE) and modulates the transcription of its target genes, such as acyl-CoA oxidase. It therefore controls the peroxisomal beta-oxidation pathway of fatty acids. Key regulator of adipocyte differentiation and glucose homeostasis. May play a role in the regulation of circadian rhythm. {ECO:0000256|RuleBase:RU364110}. |
Activator;Biological rhythms;Cytoplasm;DNA-binding;Metal-binding;Nucleus;Phosphoprotein;Proteomics identification;Receptor;Reference proteome;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
This gene encodes a nuclear receptor protein belonging to the peroxisome proliferator-activated receptor (Ppar) family. The encoded protein is a ligand-activated transcription factor that is involved in the regulation of adipocyte differentiation and glucose homeostasis. The encoded protein forms a heterodimer with retinoid X receptors and binds to DNA motifs termed 'peroxisome proliferator response elements' to either activate or inhibit gene expression. Mice lacking the encoded protein die at an embryonic stage due to severe defects in placental vascularization. When the embryos lacking this gene are supplemented with healthy placentas, the mutants survive to term, but succumb to lipodystrophy and multiple hemorrhages. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Apr 2015]. |
mmu:19016; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; perinuclear region of cytoplasm [GO:0048471]; nuclear estrogen receptor binding [GO:0030331]; nuclear receptor activity [GO:0004879]; protein phosphatase binding [GO:0019903]; transcription cis-regulatory region binding [GO:0000976]; transcription coactivator binding [GO:0001223]; zinc ion binding [GO:0008270]; animal organ regeneration [GO:0031100]; cellular response to hyperoxia [GO:0071455]; cellular response to hypoxia [GO:0071456]; cellular response to prostaglandin E stimulus [GO:0071380]; cellular response to retinoic acid [GO:0071300]; cellular response to vitamin E [GO:0071306]; fatty acid oxidation [GO:0019395]; heart development [GO:0007507]; negative regulation of acute inflammatory response [GO:0002674]; negative regulation of cardiac muscle hypertrophy in response to stress [GO:1903243]; negative regulation of cell growth [GO:0030308]; negative regulation of collagen biosynthetic process [GO:0032966]; negative regulation of connective tissue replacement involved in inflammatory response wound healing [GO:1904597]; negative regulation of gene expression [GO:0010629]; negative regulation of lipid storage [GO:0010888]; negative regulation of pancreatic stellate cell proliferation [GO:2000230]; negative regulation of smooth muscle cell proliferation [GO:0048662]; negative regulation of telomerase activity [GO:0051974]; positive regulation of adiponectin secretion [GO:0070165]; positive regulation of apoptotic process [GO:0043065]; positive regulation of fat cell differentiation [GO:0045600]; positive regulation of fatty acid oxidation [GO:0046321]; positive regulation of oligodendrocyte differentiation [GO:0048714]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of transcription by RNA polymerase II [GO:0045944]; response to caffeine [GO:0031000]; response to cold [GO:0009409]; response to estrogen [GO:0043627]; response to immobilization stress [GO:0035902]; response to mechanical stimulus [GO:0009612]; response to metformin [GO:1901558]; response to starvation [GO:0042594]; response to vitamin A [GO:0033189]; response to xenobiotic stimulus [GO:0009410]; rhythmic process [GO:0048511] |
11792850_A mutation in the peroxisome proliferator-activated receptor gamma-binding site in the gene for the cytosolic form of phosphoenolpyruvate carboxykinase reduces adipose tissue size and fat content in mice. 11877444_T0070907, a selective ligand for peroxisome proliferator-activated receptor gamma, functions as an antagonist of biochemical and cellular activities 11884400_PPARgamma requirement for the establishment and physiology of different cell types 11903058_Nuclear receptor corepressor-dependent repression of peroxisome-proliferator-activated receptor delta-mediated transactivation 11909955_Conditional disruption of the peroxisome proliferator-activated receptor gamma gene in mice results in lowered expression of ABCA1, ABCG1, and apoE in macrophages and reduced cholesterol efflux. 11923467_Selective intranuclear redistribution of PPAR isoforms by retinoid X receptor alpha 11996104_Ligands of peroxisome proliferator-activated receptor-gamma increase the generation of vascular endothelial growth factor in vascular smooth muscle cells and in macrophages 12015306_PPRE in intron 1 of the ACBP gene is a bona fide PPARgamma-response element. 12037571_Trap220(-/-) fibroblasts are refractory to PPAR gamma(2)-stimulated adipogenesis. (PPAR gamma 2) 12056591_Peroxisome proliferator-activated receptor alpha,gamma coagonist LY465608 inhibits macrophage activation and atherosclerosis in apolipoprotein E knockout mice 12059785_Genomic structure and tissue distribution of PPARgamma 12086932_role in maintenance of the characteristics of mature 3T3-L1 adipocytes 12147237_transient expression exhibited by serum-free differentiation of 3T3-L1 preadipocytes 12172784_This protein was found in immature adipocytes which differentiated from chondrocytes in vitro in response to chick serum. 12200443_formal genetic analysis to assess the specific role of each individual isoform in adipogenesis 12270934_role of expression in promotion of adipogenesis via activation of MEK/ERK signaling 12370429_mice with preexisting damage to Apc, a regulator of beta-catenin, develop colon tumors in a manner insensitive to the status of PPARgamma 12401792_results suggest that a high level of PPARgamma in mouse liver is sufficient for the induction of adipogenic transformation of hepatocytes with adipose tissue-specific gene expression and lipid accumulation 12468449_PPAR-gamma activation suppresses induction of Egr-1 and its inflammatory gene targets in a ischemia reperfusion injury model. 12471115_PPAR gamma agonists rapidly induce apoptosis and a profound increase in NF-kappa B DNA binding in primary B cells from the bone marrow and in a B cell line, BU-11, representing the pro/pre-B cell stage. 12589053_PPARgamma is regulated by Smad1 and p38 kinase during bone morphogenetic protein 2-induced adipogenesis 12664620_Apoptosis of mouse T cells in induced by this receptor. 12704187_the activation of PPARgamma inhibits osteocalcin expression both by suppressing the expression of Runx2 and by interfering with the transactivation ability of Runx2 12729899_Regulation of phosphodiesterase 3B gene expression by peroxisome proliferator-activated receptor gamma. 12730300_PPARG has a role in regulating glucose and lipid uptake and oxidation and preadipocyte differentiation 12730867_commensal intestinal flora affects the expression of PPAR gamma and that PPAR gamma expression is considerably impaired in ulcerative colitis 12734379_PPAR-gamma activation inhibits the migration of dendritic cells from the airway mucosa to the thoracic lymph nodes and also profoundly reduces the priming of antigen-specific T lymphocytes in the draining lymph nodes. 12754280_trans10, cis12 CLA isomer prevents lipid accumulation in adipocytes by acting as a PPARgamma modulator 12759335_Transcriptional activation of the beta-carotene 15, 15'-monooxygenase gene by peroxisome proliferator-activated receptor (beta-carotene 15, 15'-monooxygenase). 12777391_PPARgamma represses transcriptional activity of the GLUT4 promoter via direct and specific binding of PPARgamma/retinoid X receptor-alpha to the GLUT4 promoter. 12805374_liver PPAR gamma regulates triglyceride homeostasis, contributing to hepatic steatosis, but protecting other tissues from triglyceride accumulation and insulin resistance 12821117_PPAR-gamma plays a negative role in pancreatic beta cells 12829715_Peroxisome proliferator-activated receptor gamma and ligands inhibit surfactant protein B gene expression in the lung 12833141_the effects of PPARgamma on cell proliferation and apoptosis are dependent on the presence of an RB-HDAC3 complex 12851309_Data suggest that that the agouti/melanocortin signaling pathway regulates levels of peroxisome proliferator activated receptor gamma in adipocytes. 12878168_Activation of PPARdelta by long chain fatty acids impairs expression of determination factor MyoD1 and alpha-actin, abolishes the development of multinucleated myotubes, and in parallel induces expression of PPARgamma gene, regulator of adipogenesis. 12878983_ACtivation of this receptor inhibits differentiation of preosteoblasts in vitro. 12887689_Our results indicate for the first time that PPARgamma expression can be modulated by the cAMP signalling pathway, and suggest the anti-inflammatory effects of noradrenaline on brain cells may be partly mediated by increasing PPARgamma 12900517_Data show that peroxisome proliferator-activated receptors alpha and gamma decrease airway hyperresponsiveness, lung inflammation, eosinophilia, cytokine production, GATA-3 expression, and serum IgE in a murine model of human asthma. 12917338_The inhibition of PPAR gamma function by cyclin D1 is a new mechanism of signal transduction cross talk between PPAR gamma ligands and mitogenic signals that induce cyclin D1. 12925701_Loss of muscle-expressed pparg in the mouse leads to adiposity and insulin resistance. 12925701_PPARgamma has a role in maintenance of normal adiposity, whole-body insulin sensitivity, and hepatic insulin action 12960963_Data show that neither hemizygous deletion of peroxisome proliferator-activated receptor (PPAR)-gamma nor complete ablation of Ppara influenced the development of prostate cancer in this experimental context. 12966085_PPARgamma and Foxo1 together may play a role in insulin action and the insulinomimetic properties of PPARgamma ligands 12975479_findings show that 15-deoxy-delta12,14-PGJ2 is not the endogenous mediator of PPARgamma-dependent adipocyte activation 14500358_Mutant thyroid hormone receptor beta represses the expression and transcriptional activity of peroxisome proliferator-activated receptor gamma during thyroid carcinogenesis. 14517292_PPAR gamma function has a critical role in beta-cell proliferation 14603033_PPAR gamma is the master regulator of adipogenesis in vivo. Lipid and glucose homeostasis can be relatively well maintained in the absence of white adipose tissue. 14614508_mice deficient in CREB activity have a fatty liver phenotype and display elevated expression of the nuclear hormone receptor PPAR-gamma, a key regulator of lipogenic genes 14617516_important immunomodulatory role for PPAR-gamma in alveolar macrophage responses. 14625542_role for muscle PPAR-gamma in the maintenance of skeletal muscle insulin action and the etiology of insulin resistance 14634082_PPAR gamma is a critical physiological regulator of central nervous system inflammation and demyelination in experimental allergic encephalomyelitis. 14646597_inhibition of 3T3-L1 cells differentiation by TNF-alpha is partly implemented through NF-kappaB and one of its downstream effectors is PPARgamma 14660788_Deletion of PPARgamma in adipose tissue resulted in marked adipocyte hypocellularity and hypertrophy, elevated levels of plasma free fatty acids and triglyceride, and decreased levels of plasma leptin and ACRP30. 14688261_progesterone-induced synthesis of the 12/15-lipoxygenase-derived lipid mediators activates PPARgamma and its downstream gene networks, which in turn function as critical regulators of implantation in the mouse 14704148_PPARgamma regulates expression of the perilipin gene in adipocytes 14726448_propose that peroxisome proliferator-activated receptor gamma(PPAR)plays a significant role in the transcriptional regulation of murine perilipin gene via the PPAR response element in its promoter 14764597_PPARgamma2 activation inhibited cyclin D1 transcription in hepatocytes via CREB-dependent and beta-catenin-independent pathways 14988019_An RNA interfering approach was used to study the Pparg pathway using lentiviral vector expressing microRNA. 15016406_treatment with IL-1beta or Lipopolysaccharides, but not with IL-6, inhibited the differentiation of brown adipocytes, which was accompanied by a 2.5-10-fold decrease in PPARgamma mRNA 15037548_once an initiating event has taken place, increased PPAR gamma signaling serves as a tumor promoter in the mammary gland 15054141_39 (of 12,488 tested) genes are upregulated by PPARgamma, in aortic mRNA isolated from mice treated for 21 days with a PPARgamma agonist (rosiglitazone). Some of these are implicated in inflammation and hypertension. 15060163_Results suggest that liver X receptors play a role in adipocyte differentiation by regulation of lipogenesis and expression of adipocyte-specific genes, such as peroxisome proliferator-activated receptor-gamma. 15070754_PPARgamma is essential for the in vivo survival of mature adipocytes,in transgenic mice. 15073165_peroxisome proliferator-activated receptor gamma and sterol regulatory element-binding protein have roles in the regulation of adipogenesis and regulate INSIG-1 15111493_Transcription of the human perilipin gene is stimulated by peroxisome proliferator-activated receptor-gamma (PPAR-gamma) through a DR-1 type PPRE. 15123608_Results suggest that lipin expression is required prior to peroxisome proliferator-activated receptor (PPAR)gamma during adipocyte differentiation. 15123625_These results suggest that the PPARgamma-dependent transactivation pathway seems to be modulated by SUMO-1 modification and may serve as a novel target for apoptosis-induction therapy in cancer cells. 15157741_specificity protein-1, PPARgamma, and pituitary protein transcription factor-1 have roles in transcriptional regulation of the murine CORS-26 promoter 15158467_C/EBPbeta, C/EBPalpha, and PPARgamma participate in a cascade during adipogenesis 15164767_role in palmitic acid transport and signaling in E/H-FABP knockout mice 15174093_Paprg expresssion is decreased in cftr-/- mice. 15187132_deomstrate that PPARgamma is expressed in both murine CD4 and CD8 cells, and that its ligands directly decrease IFN-gamma expression by T cell lines, and that PPARgamma acts on the IFN-gamma promoter by interfering with c-Jun activation 15231871_Role of PPARgamma in growth hormone signaling, which might contribute to the metabolic disorder affecting insulin signaling in PPARgamma mutant mice. 15246971_data indicate that an endogenous pathway associated with PPARgamma plays an important role in the pathogenesis of ischemia-reperfusion-associated injury in the stomach 15247146_both PPARbeta and PPARgamma isoforms are required to facilitate maximal lipid accumulation and differentiation during adipogenesis. 15249658_Data demonstrate that peroxisome proliferator-activated receptor gamma 2 deficiency impairs the development of adipose tissue and insulin sensitivity. 15254591_These results provide genetic evidence for a critical role for peroxisome proliferator-activated receptor gamma in blood pressure regulation that is not dependent on altered insulin sensitivity. 15284209_TNFalpha suppresses PPARgamma2 gene transcription by the inhibition of C/EBPdelta expression and its DNA binding during the early stage of adipocyte differentiation 15304360_Anti-inflammatory action of capsaicin may be mediated by PPARgamma activation in LPS-stimulated macrophage RAW 264.7 cells. 15308623_GSKbeta is a key regulator of the balance between beta-catenin and PPARgamma activity and activation of canonical Wnt signaling downstream of PPARgamma blocks expression of a select subset of adipogenic genes 15359271_antagonizing PPARgamma activity in fat could be an effective way to combat osteoporosis 15504964_PPAR-gamma agonist can restore impaired insulin secretion under conditions of islet fat accumulation in ppargamma deficient mice. 15572375_PPARgamma exhibits promiscuity in binding on a PPAR response elements, suggesting that the definition for PPARgamma may need to be expanded. 15574741_Inducing expression or activating PPARgamma using thiazolinediones decreases amyloid-beta (Abeta) levels in conditioned medium. The Abeta clearance mechanism is induced by PPARgamma activation in primary murine-mixed glia and cortical neuronal cultures. 15578089_Results reveal receptor-specific mechanisms by which PPARs alpha, beta/delta, and gamma influence macrophage cholesterol homeostasis. 15611480_Since PPARgamma is a master regulator of adipocyte differentiation, we assessed the effect of organotin compounds on preadipocyte 3T3-L1 cells. 15664998_KLF15 plays an essential role in adipogenesis in 3T3-L1 cells through its regulation of PPAR gamma expression 15670761_Taken together, these results strongly suggest that PPAR-gamma ligands up-regulate FGF-2-stimulated VEGF release resulting from amplifying activation of SAPK/JNK in osteoblasts. 15673477_Review. In vitro studies suggest that PPAR gamma is a master regulator of adipogenesis. In vivo findings from use of PPAR gamma agonists & knockout studies in mice are reviewed. 15681609_modulation of adipocyte PPARgamma target genes by TZDs involves the dissociation of corepressors by direct and indirect mechanisms 15694373_These results indicate that glimepiride has the potential to induce PPAR gamma activity, thereby improving insulin resistance. 15713663_cyclin D1 has a role in regulation of PPARgamma-mediated adipocyte differentiation through recruitment of HDACs to regulate PPAR response element local chromatin structure and PPARgamma function 15716267_mice with a PPARgamma L466A knock-in mutation exhibit symptoms of metabolic syndrome 15778364_CD40 engagement with CD40L blunted the ability of PPARg agonists to induce apoptosis of B lymphocytes and prevented both the inhibition of NF-kappaB mobilization and the PPARg agonist-induced increase in IkappaBalpha and IkappaBbeta protein levels 15799829_PPAR-gamma has a role in inhibiting angiogenesis in non-small cell lung cancer 15831367_Beneficial role for PPARgamma-specific ligands in the treatment of hypertension. Therapeutic strategies targeting endothelial cells may prove useful. 15833818_essential role for PPARgamma in the development of adipose tissues and induction of hepatic PPARgamma may stimulate an increase in glucose disposal by the liver. 15857827_analysis of gammaORF4, a peroxisome proliferator-activated receptor gamma isoform with dominant negative activity generated by alternative splicing 15867269_Potent preventive effect of (n-3) PUFA on HER-2/neu-mediated mammary carcinogenesis, without interaction with a synthetic PPARgamma activator. 15897255_ppar-gamma mutants appeared deficient in spatial working memory as tested in spontaneous alternation in the Y-maze and delayed nonmatch to place 15905321_Mechanism contributing to diabetic bone loss: increased marrow adiposity in type 1 diabetes: increased expression of Ppaarg. 15917242_PPARgamma binds to the functional PPAR-response element of the PC promoter. Impaired in vitro differentiation of white adipocytes of PPARgamma2 knock-out mice was also associated with a marked reduction of PC mRNA 15917308_PPARgamma agonists reduce oval cell proliferation and fibrosis during chronic liver injury and may be useful in the prevention of hepatocellular carcinoma. 15919792_These findings suggest that PPARgamma2 is required for the maintenance of normal insulin sensitivity in mice but also raises the intriguing notion that PPARgamma2 may be necessary for the adverse effects of a high-fat diet on carbohydrate metabolism. 15947238_macrophage PPARgamma deficiency increases atherosclerosis under conditions of mild and severe hypercholesterolemia, indicating an antiatherogenic role for PPARgamma 15956187_there is a PPARgamma-dependent pathway in regulation of sodium transport in the collecting duct that underlies TZD-induced fluid retention 16023420_data demonstrate that activation of PPARgamma signaling by either addition of PPARgamma ligands or overexpression of PPARgamma in growth plate chondrocytes inhibits TR-mediated gene transcription 16039966_Whereas genetically reduced PPARgamma activity in adipose tissue leads to reduction of total fat mass in humans and in mice, mutations in the ligand-binding domain of PPARgamma cause abnormal body fat distributions. Review. 16106032_EBF-1 and PPARgamma2 induce adipocyte differentiation with comparable kinetics and efficiency. 16125673_CREB-PPARgamma signaling pathway may be involved in the high fat diet-induced liver steatosis 16127159_Inflammation-triggered cell growth and emphysema during lysosomal acid lipase deficiency are partially caused by peroxisome proliferator-activated receptor-gamma inactivation. 16127449_identification of a molecular pathway by which PPAR-gamma represses the transcriptional activation of inflammatory response genes in mouse macrophages 16150867_PDK1 can function as a PPARgamma1 coactivator independently of its catalytic activity and establishes an important mechanistic link between adipocyte differentiation and the insulin signaling pathway. 16150927_findings suggest that a protective role of PPARgamma in the pathogenesis of the asthma is partly mediated through an IL-10-dependent mechanism 16179348_peroxisomal proliferator activated receptor gamma and retinoic acid X receptor agonists lower glucose through distinct mechanisms 16213226_Disruption of cdk4 or activating mutations in cdk4 in primary mouse embryonic fibroblasts results in reduced and increased adipogenic potential, respectively, of these cells. cdk4 also participates in adipocyte function through activation of PPARgamma. 16246044_Models have confirmed a key role for PPAR-gamma in the metabolic syndrome; however, they challenge the concept that insulin resistance is the main factor linking the clinical manifestations of the metabolic syndrome. (review) 16253638_The opposite hepatic outcomes in Liver mt PPARgamma Tg as a result of fasting and high-fat feeding may indicate distinct roles of PPARalpha and PPARgamma in 2 different types of nutritionally provoked steatosis 16260612_cyclin D3 is an important factor governing adipogenesis and obesity through activation of peroxisome proliferator-activated receptor gamma 16303334_Reduced PPARgamma expression/activity leads to increased expression of induced nitric oxide synthase (iNOS) and overproduction of nitric oxide observed in MRL/lpr mice. 16306350_PPARalpha activation prevents inflammation in white adiopse tissue and dual activation of PPARalpha and -gamma enhances the action of adiponectin. 16306360_PPARgamma modulation is a new approach that, based on selective receptor-cofactor interactions and target gene regulation, should result in potent insulin sensitization in the absence of PPARgamma-mediated adverse effects. 16314832_PPARgamma insufficiency promotes follicular thyroid carcinogenesis via activation of the nuclear factor-kappaB signaling pathway 16326714_adiponectin is an important contributor to PPARgamma-mediated improvements in glucose tolerance through mechanisms that involve the activation of the AMPK pathway 16341123_Recognition of AC by macrophages suffices to attenuate the early phase of ROS formation that is attributed to PPARgamma activation. 16357043_PPARalpha and PPARalpha-signaling are activated in liver by chronic high-fat feeding and PPARgamma may compensate for PPARalpha in PPARalpha null mice on high-fat diets 16364246_summary, PPARgamma2 is increased in hepatocytes by oleic acid and insulin. 16376852_glutathione S-transferase zeta/maleylacetoacetate isomerase is induced by C/EBPalpha and dependent on PPAR gamma 16403437_All these results suggest that ADRP is up-regulated in liver steatosis in human and mice, and that high fat diet increases expression of ADRP through PPARgamma activation, followed by induction of liver steatosis. 16407166_protective mechanism of nonsteroidal anti-inflammatory drugs in Alzheimer's disease involves activation of PPARgamma and decreased beta-site amyloid precursor protein cleaving enzyme gene transcription 16431920_C/EBP beta activates a single unified pathway of adipogenesis involving its stimulation of PPARgamma expression, which then activates C/EBP alpha expression by dislodging HDAC1 from the promoter for degradation in the proteasome 16457814_All together, data presented suggest that PPARgamma participates in regulation of STAT5A expression. 16461467_These results reveal a MAP4K4/NIK-dependent signaling pathway that potently inhibits PPARgamma-responsive gene expression. 16484339_PPARgamma-cdk9 interaction results in increased transcriptional activity of PPARgamma and therefore increased adipogenesis. 16499711_mechanical loading of osteoblasts results in expression 16524877_PPARgamma regulates the development of the central nervous system during early embryogenesis via control of neural stem cell proliferation 16527895_purified tumor-associated macrophages display a similar M2 phenotype and are suppressive for antitumor CTLs, via a mechanism that can be almost completely reversed by PPARgamma ligands 16547277_demonstrate the inhibition of proinflammatory cytokine-induced NF-kappaB transactivation in central nervous system glial cells by IL-4 via PPAR-gamma activation 16556603_cross-regulation of CD36 by PXR and PPARgamma suggests that this fatty acid transporter may function as a common target of orphan nuclear receptors in their regulation of lipid homeostasis 16567512_Fish oil regulates adiponectin secretion through a PPARgamma-dependent manner in epididymal fat. 16574647_the trophic properties of gastrin in colorectal cancer may be mediated in part by transactivation of the EGFR and phosphorylation of ERK1/2, leading to degradation of PPARgamma protein and a decrease in PPARgamma activation 16580992_The expression of PPARgamma2 is induced by Shn-2 entered the nucleus upon BMP-2 stimulation and, in cooperation with Smad1/4 and C/EBPalpha. 16675498_These findings suggest that muscle denervation-induced fatty degeneration may be mediated through C/EBPalpha and PPARgamma2. 16720732_Respiratory epithelial cell PPARgamma is necessary for normal lung structure and function. 16828750_PPARgamma-siRNA downregulated the expression of aP2 mRNA and lipid accumulation, whereas it upregulated the expression of osteocalcin and calcium deposition. 16858678_Intestinal PPARgamma deficiency enhanced tumorigenesis irrespective of gender. Our results suggest that PPARgamma functions as a tumor resistance factor in the mouse intestine. 16873730_GM-CSF deficiency increases atherosclerosis under hypercholesterolemic conditions, indicating antiatherogenic role for GM-CSF. Protective role may be mediated by PPAR-gamma and ABCA1, molecules that affect cholesterol homeostasis and inflammation. 16876120_PPARgamma activation represses the expression of the resistin gene by modulating Sp1 activity 16921131_Epithelial PPAR gamma is necessary for proper lung maturation and response to injury. 16951334_Cell survival promotion is a novel activity of PPAR gamma, which may modulate cytokine withdrawal-induced activated T cell death. 16966031_The expression of PPAR-gamma inversely correlated with pancreatitis severity and expression of the proinflammatory cytokines, interleukin-6, and tumor necrosis factor-alpha. 16971390_hepatic steatosis in apoB/BATless mice is associated with elevated rates of hepatic lipogenesis that are linked directly to increased hepatic expression of PPARgamma2 16972249_Results show that inhibition of PPARgamma by BADGE prevents type I diabetic bone marrow adiposity but not bone loss. 16980437_Whole animal impairment of PPARgamma alters the cellular composition of the adipose organ to a more 'white' adipose phenotype. 16981222_LPC promotes cholesterol efflux from macrophage foam cells via a PPARgamma-LXRalpha-ABCA1-dependent pathway 17003330_when expressed on a hyperphagic ob/ob background, the P465L PPARgamma mutant grossly exacerbates the insulin resistance and metabolic disturbances associated with leptin deficiency 17008383_metabolism of a parent compound, beta-carotene, may alternatively activate (9-cisRA) or inhibit (beta-apo-14'-carotenal) specific RXR and PPAR responses 17015710_role of PPARg in TDI-induced asthma includes increasing numbers of inflammatory cells of the airways, airway hyperresponsiveness, increased levels of Th2 cytokines, adhesion molecules, chemokines, TGF-beta1, and NF-kappaB in nuclear protein extracts. 17019405_These results suggest that PPARgamma has a significant role in the expression of behavioral sensitization to METH in mice. 17095756_Data suggest that PPAR-gamma regulates recruitment of macrophages to inflammatory foci in the colon. 17143554_Our data demonstrate that, although having anti-fibrotic properties in rats, PPARgamma agonists do not prevent activation of HSCs in vitro, nor hepatic fibrogenesis in vivo in mice. 17174269_These data suggest a new role for emodin as a PPARgamma agonist in 3T3-L1 cells. 17214992_proliferator-activated receptor alpha/gamma have roles in preventing atherosclerosis in female low density lipoprotein receptor deficient mice 17218271_These studies define parallel but functionally distinct pathways that are utilized by PPARgamma and LXRs to differentially regulate complex programs of gene expression that control immunity and homeostasis. 17239574_PPARgamma is upregulated during the resolution phase of wound repair 17240171_Antidiabetic peroxisome proliferator-activated receptor gamma (PPARgamma) agonists inhibited expression of 3T3-L1 adipocyte alpha1-AGP in a concentration- and time-dependent manner via an apparent PPARgamma-mediated mechanism.[Alpha1 acid glycoprotein] 17276354_XOR is a novel regulator of adipogenesis and of PPARgamma activity and essential for the regulation of fat accretion. 17301838_As keratinocyte-selective peroxisome proliferator-activated receptor gamma (PPARgamma) ablation had similar effects, RXRalpha/PPARgamma heterodimers most probably mediate epidermal tumor suppression. 17304352_PPARgamma is required to maintain normal adiposity and insulin sensitivity in adult mice. 17389706_Endogenous PPAR-gamma may play a role in suppression of the inflammatory pathway in 3T3-L1 cells. 17394061_Modulation of PPAR-gamma RNA expression may be implicated in the protective effect of YHK in experimental NASH, limiting hepatocyte lipid accumulation. 17462989_Cidea is a novel target gene for both PPARalpha and -gamma in the liver where these two transcription factors utilize the same PPRE region for dual regulation 17463056_vivo activation of PPARgamma modulates placental morphology and fatty acid accumulation 17463185_These data show that the PPAR-gamma in the pancreatic epithelia and islets is important in suppressing inflammation and is required for the anti-inflammatory effects of thiazolidinediones in acute pancreatitis. 17468515_FUS-DDIT3 contributes to differentiation arrest acting at a point in the adipocyte differentiation process after induction of peroxisome proliferator-activated receptor gamma expression. 17484888_increased HBx expression causes lipid accumulation in hepatic cells mediated by sterol regulatory element binding protein 1 and PPARgamma, which could be a putative molecular mechanism mediating the pathophysiology of HBV infection. 17515919_PPARgamma is required for maturation of alternatively activated macrophages 17519361_PPAR-gamma is induced on differentiation of colonic epithelial cells and that this receptor stimulates differentiated function in epithelial cells throughout the colon. 17521607_These results establish the importance of salt bridges between these residues in the heterodimerization of nuclear receptors, and offer a technical approach to generating functional NR mutants with directed heterodimerization specificity. 17536062_support the utility of dual PPARalpha/gamma agonists in treating type 2 diabetes and diabetic nephropathy 17540578_These results suggest that 10t-12c CLA modulates the phagocytic activity of RAW macrophages by upregulating TNF-alpha expression via a PPARgamma-dependent pathway. 17543202_Data show that activation of peroxisome proliferator activated receptor-gamma ameliorates the mitochondrial translocation of NOX-4 and matrix metalloproteinase-9 activation. 17587675_Both GR and GRP extracts enhanced insulin-stimulated glucose uptake through peroxisome proliferation-activated receptor (PPAR)-gamma activation in 3T3-L1 adipocytes 17625041_PPARgamma gene introduction suppressed monocyte/macrophage invasion, the generation of myofibroblasts, and the upregulation of cytokines/growth factors/MMPs in a healing cornea. PPARgamma overexpression may be an effecive treatment of ocular burns. 17641685_unexpected essential role for PPARbeta in constraining tumor endothelial cell proliferation to allow for the formation of functional tumor microvessels 17652179_PPAR gamma deficiency leads to increased expression of lipid oxidation enzymes in the lactating mammary gland. Metabolomic profiling detects increased levels of oxidized free fatty acids in the pups nursed by PPAR gamma-deficient mothers. 17678635_Cardiac peroxisome proliferator-activated receptor gamma is essential in protecting cardiomyocytes from oxidative damage. 17699161_PPARg has a critical role in maintaining adipose tissue integrity as a prerequisite for systemic energy balance 17713572_PPARbeta/delta in the epidermis: (1) is required for permeability barrier homeostasis; (2) regulates keratinocyte proliferation; and (3) modulates cutaneous inflammation 17719655_We therefore propose that PPARgamma in brain EC may be exploited to target detrimental EC-T cell interactions under inflammatory conditions. 17823655_PPARgamma1-transgenic mice developed a dilated cardiomyopathy associated with increased lipid and glycogen stores, distorted architecture of the mitochondrial inner matrix, and disrupted cristae. 17848638_PPARgamma positively regulates ATGL mRNA and protein expression in mature adipocytes in vitro and in adipose tissue in vivo, suggesting a role for ATGL in mediating PPARgamma's effects on lipid metabolism. 17888405_We demonstrated that Wnt3a conditioned medium suppresses C/EBPbeta/delta-induced adipogenesis of 3T3-L1 cells by inhibiting PPARgamma induction. 17936399_inhibitory role in hepatic I/R injury 17947702_Pyk-2 tyrosine kinase-ERK1/2 pathway is in part responsible for the adrenomedullin and its binding protein-mediated induction of PPAR-gamma and the anti-inflammatory effect 17954559_inhibition of SIRT1 in adipocytes induces the same insulin-sensitizing action as Peroxisome proliferator-activated receptor gamma ligands 17963725_Selective modulation of PPARgamma by TZDs resulted in distinct expression profiles and transcription kinetics which may be due to differential promoter activation and chromatin remodeling of target genes. 17965419_PPARgamma supports survival in neurons in part through a mechanism involving increased expression of Bcl-2. 18032385_Inhibition of adipogenesis caused by SCD2 depletion was associated with a decrease in PPAR-gamma mRNA and protein, whereas in mature adipocytes loss of SCD2 diminished PPARgamma protein levels, with little change in mRNA levels. 18039840_there is a conditional requirement for MED1 and that a direct interaction between PPARgamma and Mediator through MED1 is not essential either for PPARgamma-stimulated adipogenesis or for PPARgamma target gene expression in fibroblasts 18056926_the t-10, c-12 CLA isomer appears to alter adipocyte adiponectin metabolism through PPARgamma-dependent and PPARgamma-independent mechanisms. 18059282_PPAR-gamma and its ligands have a previously unrecognized role in promoting osteoclast differentiation and bone resorption 18060855_The results indicate that auraptene activates PPARgamma in adipocytes to control adipocytekines such as adiponectin and MCP-1. 18073138_Collectively, our findings suggest that PPARgamma plays a crucial role in the expression of catalase in adipocytes. 18085707_PPARgamma-deficient mice had more liver injury after ischemia/reperfusion than their wild-type |
ENSG00000132170 |
PPARG |
16.312628 |
5.1693970146 |
2.369996 |
0.66459117 |
12.194612 |
0.00047927733660552368586543758866014286468271166086196899414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0024149362855360027337037198691405137651599943637847900390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.9881934267462 |
12.8767628166363 |
5.22076237330876 |
1.95120696803493 |
| ENSMUSG00000000489 |
18591 |
Pdgfb |
protein_coding |
A0A0R4IZW4 |
Human_homologues FUNCTION: Growth factor that plays an essential role in the regulation of embryonic development, cell proliferation, cell migration, survival and chemotaxis. Potent mitogen for cells of mesenchymal origin (PubMed:26599395). Required for normal proliferation and recruitment of pericytes and vascular smooth muscle cells in the central nervous system, skin, lung, heart and placenta. Required for normal blood vessel development, and for normal development of kidney glomeruli. Plays an important role in wound healing. Signaling is modulated by the formation of heterodimers with PDGFA (By similarity). {ECO:0000250|UniProtKB:P31240, ECO:0000269|PubMed:26599395}. |
Disulfide bond;Growth factor;Mitogen;Proteomics identification;Reference proteome;Secreted;Signal |
|
|
mmu:18591; |
cell surface [GO:0009986]; extracellular region [GO:0005576]; membrane [GO:0016020]; platelet-derived growth factor complex [GO:1990265]; chemoattractant activity [GO:0042056]; collagen binding [GO:0005518]; growth factor activity [GO:0008083]; platelet-derived growth factor binding [GO:0048407]; platelet-derived growth factor receptor binding [GO:0005161]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; superoxide-generating NADPH oxidase activator activity [GO:0016176]; activation of protein kinase B activity [GO:0032148]; cellular response to growth factor stimulus [GO:0071363]; interleukin-18-mediated signaling pathway [GO:0035655]; monocyte chemotaxis [GO:0002548]; negative regulation of gene expression [GO:0010629]; negative regulation of miRNA transcription [GO:1902894]; negative regulation of phosphatidylinositol biosynthetic process [GO:0010512]; negative regulation of platelet activation [GO:0010544]; negative regulation of protein binding [GO:0032091]; negative regulation of vascular associated smooth muscle cell differentiation [GO:1905064]; peptidyl-serine phosphorylation [GO:0018105]; peptidyl-tyrosine phosphorylation [GO:0018108]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of calcium ion import [GO:0090280]; positive regulation of cell division [GO:0051781]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of chemotaxis [GO:0050921]; positive regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0045737]; positive regulation of DNA biosynthetic process [GO:2000573]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of fibroblast proliferation [GO:0048146]; positive regulation of gene expression [GO:0010628]; positive regulation of glomerular mesangial cell proliferation [GO:0072126]; positive regulation of hyaluronan biosynthetic process [GO:1900127]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of metanephric mesenchymal cell migration by platelet-derived growth factor receptor-beta signaling pathway [GO:0035793]; positive regulation of miRNA transcription [GO:1902895]; positive regulation of mitotic nuclear division [GO:0045840]; positive regulation of phosphatidylinositol 3-kinase activity [GO:0043552]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of protein autophosphorylation [GO:0031954]; positive regulation of protein tyrosine kinase activity [GO:0061098]; positive regulation of reactive oxygen species metabolic process [GO:2000379]; positive regulation of vascular associated smooth muscle cell dedifferentiation [GO:1905176]; positive regulation of vascular associated smooth muscle cell migration [GO:1904754]; positive regulation of vascular associated smooth muscle cell proliferation [GO:1904707]; protein kinase C signaling [GO:0070528]; reactive oxygen species metabolic process [GO:0072593]; response to wounding [GO:0009611] |
11811556_results show that osteoclasts regulate osteoblasts directly and suggest that PDGF BB is a key factor in bone remodeling 11855867_PDGF-B chain transformation of NIH3T3 cells requires activation of NF-kappaB 12169633_endothelium-restricted ablation of PDGF-B generates viable mice with variation in the density of pericytes throughout the CNS; data suggest that a reduction of the pericyte density is sufficient to cause retinopathy in mice 12368212_Blockade transiently delays but does not prevent fibrous cap formation in ApoE null mice 12528182_PDGF is an important effector molecule in postnatal retinal development. 12897053_retention of PDGF-B in microvessels is essential for proper recruitment and organization of pericytes and for renal and retinal function in adult mice 14522834_PDGF system is highly expressed in the epididymis and suggest that PDGF could be involved in the maintenance of morphological structure and functional control of this organ. 14561699_Endothelial and nonendothelial sources of PDGF-B regulate pericyte recruitment and influence vascular pattern formation in tumors. 14644167_PDGF-BB stimulation of fibroblast-collagen interactions is mediated by the alphavbeta3 integrin when beta1 integrin function is impaired. 14963008_In vitro cultures supporting the spontaneous generation of functional cardiac myocytes from murine BMCs demonstrated induced expression of PDGF)-A and -B. PDGF-AB enhances the generation of BMC-derived cardiac myocytes in mouse hearts. 14988091_Imatinib reduces PDGFB by treating the atherosclerosis or by an autocrine loop linking PDGF signal transduction to PDGF expression. PDGF plays a central role in the complex interplay of cytokines & growth factors leading to a vicious circle. 14996938_expression and potential role of platelet-derived growth factor-BB (PDGF-BB) and its receptor, PDGF receptor beta (PDGFR-beta), during testis cord formation 15735680_PDGF-B provides a key switch from cell death to survival induced by TNF 15838875_syngenic mice deficient of PDGF-B leading to a defect in the recruitment of perivascular cells 15907784_PDGF-BB induces phosphorylation of alphaB-crystallin via p38 MAP kinase during myocardial infarction 16127167_Our studies identify two independent negative immune regulatory pathways-PDGF-B and SOCS-that may help limit lesion expansion. 16641085_PDGF-BB and TGF-beta1 binding to intact alpha2M is specific, involving a defined region of the alpha2M subunit 16723717_platelet-derived growth factor B has a role in anti-vascular endothelial growth factor therapy in multiple models of ocular neovascularization 16842882_PDGF-B overexpression causes liver fibrosis without significantly upregulating TGF-beta1, suggesting a TGF-beta-independent mechanism. 17438529_PDGFB-induced activation of the mitogen-activated protein kinase pathway via extracellular signal-regulated kinase was involved in the initiation of low-grade oligodendrogliomas 17545544_KIT and PDGFRbeta gatekeeper mutations are involved in tumor progression and are resistant to imatinib, but are inhibited by sorafenib 17724602_The trichloroacetic acid-treated epidermis acts as a major source of growth factors, including PDGF-B, before undergoing full necrosis. 18180311_OPN and PDGF may contribute to inflammation-associated fibrosis 18213589_PDGF-B signaling plays an important role in developing atrioventricular valves, coronaries, and cardiac innervation. 18441206_Statins restore ischemic limb blood flow in diabetic microangiopathy via eNOS/NO upregulation but not via PDGF-BB expression. 18765673_the Nck adapters are needed for signaling to Rho GTPases and actin dynamics downstream of the PDGFbeta receptor 18922925_Spinal glioma: platelet-derived growth factor B-mediated oncogenesis in the spinal cord. 19048116_PDGF-B is required to overcome cell-cell contact inhibition and to confer in vivo infiltrating potential on tumor cells 19146952_Stimulation of cultured cells with PDGF-BB induces tyrosine phosphorylation & activation of DLK by a Src-dependent mechanism. DLK may have a role in PDGF signaling. 19165863_Comparison of microarray data with published gene expression data sets for different murine neural cell types revealed a correlation between tumor cells and oligodendrocyte progenitor cells, confirming PDGF-B-induced gliomas are pure oligodendrogliomas 19219070_Sox5 can suppress platelet-derived growth factor B-induced glioma development in Ink4a-deficient mice through induction of acute cellular senescence. 19605488_analysis of specific features of BPV E5 important for its ability to activate the PDGF beta receptor and transform mouse fibroblasts 19734356_VEGF-C regulates lymphangiogenesis and capillary stability by regulation of PDGF-B. 19896531_study to examine the potential role of PDGF-B in an experimental model of peritoneal fibrosis 19998489_PDGF protects neurons from H2O2-induced cytotoxicity, and that PI-3K/Akt and MAPK pathways are involved in the protective effects; PDGF-BB exerted more potent neuroprotective effects than PDGF-AA 20035058_Failure of laminin alpha4-mediated down-regulation of PDGF activity contributes to the progressive renal lesions in this animal model. 20133221_The COL1A1-PDGFB fusion gene is the most characteristic cytogenic anomaly in DFSP. 20452974_PDGF has a role in induction of the synaptic plasticity gene Arc/Arg3.1 20506153_Overexpression of platelet-derived growth factor-B accelerates liver cancer development in chemically induced liver carcinogenesis. 20643125_a tightly regulated expression of PDGF during embryogenesis is required for normal brain development and function in mice. 20687910_identified tumor cell-secreted platelet-derived growth factor-B (PDGF-B) as a crucial factor controlling the differentiation and recruitment processes through an interaction with neuropilin-1 (NRP-1) in mesenchymal stem cells 20939912_PDGF-B-driven gliomagenesis can occur in the absence of the proteoglycan NG2 21200430_PDGF-BB stimulates cultured cardiomyocytes to synthesize the extracellular matrix component hyaluronan via HAS2 21216974_PDGF-BB induces ASM hyperplasia and changes in lung mechanics in mice 21673305_this is the first study to connect PDGF-B-induced changes in hepatic sinusoidal transport to changes in insulin action, demonstrating a link between PDGF-B signaling and insulin sensitivity. 21677873_Data show that PDGF-B depletion by RNAi caused GICs to lose their tumor-initiating capacity. 21978952_TGFbeta and PDGF-B have important roles in modulating myofibroblast generation in the mouse cornea after haze-associated injury. 22138754_define a previously unidentified mechanism of PDGF-BB-induced tumor angiogenesis and provide compelling evidence for the key role of EPO in mediating PDGF-BB-induced angiogenesis, tumor growth and hematopoiesis 22264326_These results suggest that PDGF functions as a positive regulator in the final differentiation phase of tongue muscle cells in mouse embryos. 22387002_PDGF-B signaling in trophoblasts was identified as a key component of the unique placental hematopoietic microenvironment that protects hematopoietic stem cells from premature differentiation. 22880002_Development of the retina is severely disturbed in transgenic mice in which PDGF-B was over-expressed . 22885106_Lysophosphatidic acid signals through LPA2 receptors and Galphaq proteins of cultured proximal tubule cells to transactivate latent TGF-beta in a Rho/Rho-kinase and alphavbeta6 integrin-dependent manner. 23477786_endothelium, via Dll4 and PDGF-BB, induces a fate switch in adjacent skeletal myoblasts 23831851_Tumor PDGF-BB expression levels determine dual effects of anti-PDGF drugs on vascular remodelling and metastasis. 23913003_Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. 24423492_The data indicate a dual role of Lhx2 during EMT and tumor progression: by inducing the expression of PDGF-B, Lhx2 provokes an autocrine PDGF-B/PDGFRbeta loop required for cell migration, invasion and metastatic dissemination 24472833_Our findings demonstrate, for the first time, that Shh is involved in PDGF-BB-induced smooth muscle cell migration and recruitment of mural cells into neovessels 24801603_High PDGFB expression is associated with liver fibrosis. 24860149_The signaling pathways of VEGF and PDGF are crucial mediators for determining proliferation of endothelial cells and oligodendrocytes in the neurohypophysis of adult mice. 25164676_These findings thus implicate a novel role of PDGF-BB in the migration of pericytes, resulting in loss of pericyte coverage from the endothelium with a subsequent breach of the blood-brain barrier 25173753_Platelets produce PDGFB to activate stellate cells and promote fibrosis in MDR2-null mice. 25282358_The endogenous levels of PDGF-BB, increases CD31(hi)endomucin(hi) vessel number and stimulates bone formation in ovariectomy mice. 25302990_Increased expression of STAT5b + PDGFB led to increased expression of downstream STAT5b targets. 25330836_Results suggest that regional Pax3 expression not only marks a novel subset of High-grade Brainstem Glioma but also contributes to PDGF-B-induced brainstem gliomagenesis 26599395_loss-of-function mutations in PDGFB or PDGFRB cause Primary Familial Brain Calcification. 27725190_Mechanical strain stimulates vasculogenesis of embryonic stem cells by the intracellular messengers ROS, NO and calcium as well as by upregulation of angiogenesis guidance molecules and the angiogenic growth factors VEGF, FGF-2 and PDGF-BB. 27816607_data suggest that the stronger fibrotic effect generated by Pdgfa overexpression was mediated by Pdgfralpha in cardiac interstitial mesenchymal cells 28122789_PDGF-B-PDGFRbeta signaling plays a significant role in the development of adipose tissue neovascularization. 28524773_Myeloid cell-derived PDGF contributes to vascular neotissue formation by regulating macrophage apoptosis, smooth muscle cell proliferation and extracellular matrix deposition. 28699690_microRNA let-7g suppresses PDGF-induced conversion of vascular smooth muscle cell into the atherosclerotic phenotype. 28836293_The PDGFB-overexpressing murine tumors closely cluster with human proneural and mesenchymal subtypes, as well as PDGFRA-amplified at both the RNA and protein expression levels. These models can be generated in fully immunocompetent mixed or C57BL/6 genetic background mice, and therefore can easily be incorporated into preclinical studies for cancer cell-specific or immune cell-targeting drug discovery studies. 28851707_These results demonstrate the requirement of lymphatic endothelial cells-autonomous PDGFB expression and retention for smooth muscle cell recruitment to lymphatic vessels. 29863873_the importance of PDGF-B regulatory network in prostate cancer progression. 29869048_The effect of loud sound on pericytes is mediated by upregulation of PDGF-BB. Normal functioning pericytes are required for vascular stability. 30014172_PDGF-BB co-expression prevents VEGF-induced aberrant angiogenesis by modulating VEGF-R2 signaling and endothelial proliferation, thereby limiting the degree of circumferential enlargement and enabling efficient completion of vascular splitting into normal capillary networks despite high VEGF doses. 30853660_These results collectively suggest that GIT1 is a critical participant in formation of the CD31(hi)Emcn(hi) vessel subtype, highlighting a novel biologic function of this scaffold protein in preosteoclasts. 30865886_PDGF-B is required for maturation of the glymphatic system. 31092253_An increase in PDGF-B directly enhances blood brain barrier permeability by decreasing expression of tight junction proteins. 31959796_Pro-inflammatory macrophages coupled with glycolysis remodel adipose vasculature by producing platelet-derived growth factor-B in obesity. 32078184_Glucocorticoids Disrupt Skeletal Angiogenesis Through Transrepression of NF-kappaB-Mediated Preosteoclast Pdgfb Transcription in Young Mice. 32258116_Upregulation of PDGF Mediates Robust Liver Regeneration after Nanosecond Pulsed Electric Field Ablation by Promoting the HGF/c-Met Pathway. 32587457_A systems genetics approach to revealing the Pdgfb molecular network of the retina. 33168840_Genetic analyses in mouse fibroblast and melanoma cells demonstrate novel roles for PDGF-AB ligand and PDGF receptor alpha. 34689641_Adult-induced genetic ablation distinguishes PDGFB roles in blood-brain barrier maintenance and development. 34831221_Regulation of Transplanted Cell Homing by FGF1 and PDGFB after Doxorubicin Myocardial Injury. 35863346_Microglia-derived PDGFB promotes neuronal potassium currents to suppress basal sympathetic tonicity and limit hypertension. 36351762_The Role of Platelet-Derived Growth Factor in Focal Segmental Glomerulosclerosis. 36521377_PDGF-B secreted from skeletal muscle enhances myoblast proliferation and myotube maturation via activation of the PDGFR signaling cascade. 36529087_Lin28b delays vasculature aging by reducing platelet-derived growth factor-beta resistance in senescent vascular smooth muscle cells. |
ENSG00000100311 |
PDGFB |
52.067586 |
0.4348110059 |
-1.201540 |
0.40228710 |
8.597801 |
0.00336569194726828429392595509739294357132166624069213867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0132129432321625005924969897819210018496960401535034179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.8069163379652 |
9.2670282455056 |
75.4509796106137 |
15.3157242985612 |
| ENSMUSG00000000628 |
15277 |
Hk2 |
protein_coding |
O08528 |
FUNCTION: Catalyzes the phosphorylation of hexose, such as D-glucose and D-fructose, to hexose 6-phosphate (D-glucose 6-phosphate and D-fructose 6-phosphate, respectively) (By similarity). Mediates the initial step of glycolysis by catalyzing phosphorylation of D-glucose to D-glucose 6-phosphate (By similarity). Plays a key role in maintaining the integrity of the outer mitochondrial membrane by preventing the release of apoptogenic molecules from the intermembrane space and subsequent apoptosis (PubMed:18350175). {ECO:0000250|UniProtKB:P27881, ECO:0000269|PubMed:18350175}. |
Acetylation;Allosteric enzyme;ATP-binding;Cytoplasm;Glycolysis;Kinase;Membrane;Mitochondrion;Mitochondrion outer membrane;Nucleotide-binding;Reference proteome;Repeat;Transferase |
PATHWAY: Carbohydrate metabolism; hexose metabolism. {ECO:0000250|UniProtKB:P52789}.; PATHWAY: Carbohydrate degradation; glycolysis; D-glyceraldehyde 3-phosphate and glycerone phosphate from D-glucose: step 1/4. {ECO:0000250|UniProtKB:P52789}. |
|
mmu:15277; |
centrosome [GO:0005813]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; plasma membrane [GO:0005886]; sarcoplasmic reticulum [GO:0016529]; ATP binding [GO:0005524]; fructokinase activity [GO:0008865]; glucokinase activity [GO:0004340]; glucose binding [GO:0005536]; hexokinase activity [GO:0004396]; apoptotic mitochondrial changes [GO:0008637]; carbohydrate phosphorylation [GO:0046835]; cellular glucose homeostasis [GO:0001678]; cellular response to leukemia inhibitory factor [GO:1990830]; establishment of protein localization to mitochondrion [GO:0072655]; fructose 6-phosphate metabolic process [GO:0006002]; glucose 6-phosphate metabolic process [GO:0051156]; glucose metabolic process [GO:0006006]; glycolytic process [GO:0006096]; lactation [GO:0007595]; maintenance of protein location in mitochondrion [GO:0072656]; negative regulation of mitochondrial membrane permeability [GO:0035795]; negative regulation of reactive oxygen species metabolic process [GO:2000378]; positive regulation of angiogenesis [GO:0045766]; positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization [GO:1904925]; regulation of glucose import [GO:0046324]; response to hypoxia [GO:0001666]; response to ischemia [GO:0002931] |
11782486_induction of expression and activity by HB-EGF 12865258_muscle glucose uptake is impaired by reductions in hexokinase II activity during exercise 15070811_IL-1beta increased HK activity in both a time- and concentration-dependent manner in glomerular mesangium cells 15627654_sterol regulatory element binding factor 1 plays a major role in the response of FAS and hexokinase 2 genes to nutritional regulation 15878951_adaptations that increase Hexokinase II protein content and/or functional activity such as regular exercise contribute to increased muscular endurance 16327801_IRS2, HK2 and INSIG1 are direct targets of TFE3 17639019_Impairments in whole-body metabolism and muscle glucose uptake due to high-fat feeding and partial HK II knockout mice 18165236_Between these two strategies to activate glycogen deposition in the absence of GK, embryonic livers choose to express massive levels of HKI and HKII. 18535403_hexokinase II gene inhibits human colon cancer LoVo cell growth 19049977_results point to HK-I and HK-II as promoting tumor cell survival through binding to VDAC1, thereby inhibiting cytochrome c release and apoptotic cell death. 19140804_GAPDH and HKII reciprocally interact with GLUT4 and suggest that these interactions regulate GLUT4 intrinsic activity in response to insulin 19620286_mTORC1 hyperactivity contributes to the apoptotic resistance of serum-deprived Tsc1/2-deficient cells in part by increasing the growth factor-independent expression of hexokinase II (HKII) and GLUT1. 20182739_The data suggest for the first time that HKII mediates skeletal muscle ischemia-reperfusion injury in the intact male animal. 20200205_IL-7 controls glucose utilization by regulating the gene expression of HXKII, suggesting a mechanism by which IL-7 supports bioenergetics that control cell fate decisions in lymphocytes. 20453422_Interleukin 6 enhances glycolysis through expression of the glycolytic enzymes hexokinase 2 and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-3. 21071708_reduction in HKII levels causes altered remodeling of the heart in I/R by increasing cell death and fibrosis and reducing angiogenesis and that mitochondrial binding is needed for protection of cardiomyocytes. 21430642_Hexokinase regulates Bax-mediated mitochondrial membrane injury following ischemic stress. 21504994_In a cardiac low-flow ischaemia-reperfusion model, resulting in only mild injury, standard chow or reduced HKII does not significantly affect ischaemia-reperfusion damage. 21527739_The association of hexokinase II with mitochondria is essential for the protective effects of ischemic preconditioning and normal cardiac function through maintenance of mitochondrial potential. 21554247_UCP3 was required for the HKII-mediated decrease in mitochondrial ROS emission 22334075_PPARgamma contributes to PKM2 and HK2 expression in fatty liver 22517678_HKII mitochondrial binding is also important for the hypertrophic effects, as HKII dissociation from the mitochondria resulted in de novo hypertrophy. 22593586_We identified miR-143 as an essential regulator of cancer glycolysis via targeting HK2. 22723631_Reduced muscle HKII protein content results in impaired muscle functionality during recovery from ischemic-reperfusion injury. 23836898_phosphorylation-mediated regulation of mitochondrial HK-II would be a critical component of the protective effect of Akt. 24190878_Suggest that HK2 attenuates cardiac hypertrophy by decreasing ROS accumulation via increased pentose phosphate pathway flux. 25172909_Both HK2 mRNA and protein were increased under hypoxia, which is accompanied by an increase of glucose uptake and production of lactate. 25176644_HK2 is upregulated in prostate cancer cells harboring Pten/p53 mutations. HK2 is required for Pten-/p53-deficiency-driven prostate tumor growth in vivo. 25304950_A lack of effect on gene expression, changes in the protein expression patterns of the key genes GLUT1/SLC2A1 and HK2 were observed after radiation treatment. 25904018_Hexokinase expression is highly enriched in neurons compared to astrocytes. 25948682_Hexokinase II (HKII) binding to the mitochondria is decreased in muscle from high fat diet-fed SIRT3 KO mice. 28288978_microcirculatory ischemia/reperfusion injury can be attributed to Mff-dependent mitochondrial fission via voltage-dependent anion channel 1/hexokinase 2-mediated mitochondrial permeability transition pore opening and mitochondrial reactive oxygen species/cardiolipin involved cyt-c release. 28398674_Studied melatonin's role in microvascular ischemia/reperfusion injury; found melatonin plays a protective role in mitochondrial fission-VDAC1-HK2-mPTP-mitophagy axis signal pathway suppression. 28478227_In high-fat diet fed mice, oral administration of mangiferin induced Akt phosphorylation, increased HK-II binding to mitochondria and resultantly protected vessel endothelial function, demonstrating its protective effect on endothelial integrity in vivo. 28634116_Kaempferol inhibited mitochondrial fission and maintained mitochondrial HK-II through activation of Akt, and thereby protected neurons from succinate-mediated ischemi injury. 28641785_changes in mitochondrial HKII modestly affect cardiac oxygen consumption and energy substrate metabolism 28986541_deletion of CypD is associated with an increased mtHK activity. 29114069_BAG3 directly stabilizes hexokinase 2 mRNA and promotes aerobic glycolysis in pancreatic cancer cells. 29352298_CD4 T cell mediated immuno-inflammatory responses to a virus infection were similar between WT and HK2 KO animals. The observations that the expression of HK2 appears non-essential for CD4 T cell responses against virus infections is of interest since it suggests that targeting HK2 for cancer therapy may not have untoward effects on CD4 T cell mediated immune response against virus infections. 29513568_HK2 overexpression in coronary endothelial cells might decrease EC apoptosis, increase capillary density, and help reverse coronary microvascular disease in diabetes. 30129112_The HOTTIP overexpression attenuated OGD-induced neuronal injury and imbalanced glycolytic metabolism by sponging miR-143, resulting in the de-repression of its endogenous target HK-2. Taken together, these findings improve understanding of the pathogenesis of ischemic stroke. 30191948_This study aims to explore the functions of microRNA-384-3p (miR-384-3p) in retinal neovascularization by targeting hexokinase 2 (HK2) in mice with Diabetic retinopathy 30871777_Fubp1 upregulates the mRNA levels of two hexokinase genes, Hk1 and Hk2. We suggest that Fubp1 contributes to cell survival through supporting lactate-Akt-mTOR axis. 31848318_Hexokinase 2 couples glycolysis with the profibrotic actions of TGF-beta. 32499533_Hexokinase 2 is dispensable for photoreceptor development but is required for survival during aging and outer retinal stress. 33031600_T cell immunoglobulin and mucin domain protein 3 inhibits glycolysis in RAW 264.7 macrophages through Hexokinase 2. 33082308_Selective knockdown of hexokinase 2 in rods leads to age-related photoreceptor degeneration and retinal metabolic remodeling. 33525649_Effect of Increased IL-1beta on Expression of HK in Alzheimer's Disease. 34098747_Neuronal Pentraxin 1 Promotes Hypoxic-Ischemic Neuronal Injury by Impairing Mitochondrial Biogenesis via Interactions With Active Bax[6A7] and Mitochondrial Hexokinase II. 34585393_Hexokinase 2-mediated glycolysis promotes receptor activator of NF-kappaB ligand expression in Porphyromonas gingivalis lipopolysaccharide-treated osteoblasts. 34847376_Microbial regulation of hexokinase 2 links mitochondrial metabolism and cell death in colitis. 34983946_The transcription factor KLF14 regulates macrophage glycolysis and immune function by inhibiting HK2 in sepsis. 35470850_Downregulating HK2 inhibits proliferation of endometrial stromal cells through a noncanonical pathway involving phosphorylation of signal transducer and activator of transcription 1 in endometriosis. 35975322_A non-metabolic function of hexokinase 2 in small cell lung cancer: promotes cancer cell stemness by increasing USP11-mediated CD133 stability. 36203054_Microglial hexokinase 2 deficiency increases ATP generation through lipid metabolism leading to beta-amyloid clearance. 36536134_Dual roles of hexokinase 2 in shaping microglial function by gating glycolytic flux and mitochondrial activity. 36787364_Mitochondrial control of microglial phagocytosis by the translocator protein and hexokinase 2 in Alzheimer's disease. |
ENSG00000159399 |
HK2 |
1324.279956 |
2.0532612113 |
1.037917 |
0.10653845 |
93.942371 |
0.00000000000000000000032484540975041761488551561887310867305249393908111511785079175002044493680841696914285421371459960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000146826269475322275716565700595620310751940008157852922258436445535068060053163208067417144775390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1769.58452890275 |
130.797002958295 |
861.840918794147 |
48.4355441916868 |
| ENSMUSG00000000731 |
11634 |
Aire |
protein_coding |
Q9Z0E3 |
FUNCTION: Transcription factor playing an essential role to promote self-tolerance in the thymus by regulating the expression of a wide array of self-antigens that have the commonality of being tissue-restricted in their expression pattern in the periphery, called tissue restricted antigens (TRA) (Probable). Binds to G-doublets in an A/T-rich environment; the preferred motif is a tandem repeat of 5'-. ATTGGTTA-3' combined with a 5'-TTATTA-3' box. Binds to nucleosomes (By similarity). Binds to chromatin and interacts selectively with histone H3 that is not methylated at 'Lys-4', not phosphorylated at 'Thr-3' and not methylated at 'Arg-2'. Functions as a sensor of histone H3 modifications that are important for the epigenetic regulation of gene expression. Mainly expressed by medullary thymic epithelial cells (mTECs), induces the expression of thousands of tissue-restricted proteins, which are presented on major histocompatibility complex class I (MHC-I) and MHC-II molecules to developing T-cells percolating through the thymic medulla (By similarity). Also induces self-tolerance through other mechanisms such as the regulation of the mTEC differentiation program (PubMed:19015306). Controls the medullary accumulation of thymic dendritic cells and the development of regulatory T-cell through the regulation of XCL1 expression (PubMed:21300913). Regulates the production of CCR4 and CCR7 ligands in medullary thymic epithelial cells and alters the coordinated maturation and migration of thymocytes (PubMed:19923453). In thimic B-cells, allows the presentation of licensing-dependent endogenous self-anitgen for negative selection (PubMed:26070482). In secondary lymphoid organs, induces functional inactivation of CD4(+) T-cells. Expressed by a distinct bone marrow-derived population, induces self-tolerance through a mechanism that does not require regulatory T-cells and is resitant to innate inflammatory stimuli (PubMed:23993652). {ECO:0000250|UniProtKB:O43918, ECO:0000269|PubMed:19015306, ECO:0000269|PubMed:19923453, ECO:0000269|PubMed:21300913, ECO:0000269|PubMed:23993652, ECO:0000269|PubMed:26070482, ECO:0000305|PubMed:19302042}. |
Activator;Alternative splicing;Cytoplasm;DNA-binding;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:11634; |
cytoplasm [GO:0005737]; female germ cell nucleus [GO:0001674]; male germ cell nucleus [GO:0001673]; nuclear body [GO:0016604]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; histone binding [GO:0042393]; identical protein binding [GO:0042802]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; translation regulator activity [GO:0045182]; zinc ion binding [GO:0008270]; central tolerance induction to self antigen [GO:0002509]; humoral immune response [GO:0006959]; negative thymic T cell selection [GO:0045060]; peripheral T cell tolerance induction [GO:0002458]; positive regulation of chemokine production [GO:0032722]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of thymocyte migration [GO:2000410]; thymus epithelium morphogenesis [GO:0097536]; transcription by RNA polymerase II [GO:0006366] |
11854172_Maintains immune system homeostasis. Deficiency causes autoimmune features. 12376594_Regulates expression of many self antigens in the thymus. Deficiency leads to autoimmunity. Transfer of deficient thymic tissue can transfer the autoimmune disease. 12376594_role in regulating autoimmunity by promoting the ectopic expression of peripheral tissue- restricted antigens in medullary epithelial cells of the thymus 12612579_Aire regulates negative selection of organ-specific T cells 15492124_Gene dosage--limiting role of Aire in thymic expression, clonal deletion, and organ-specific autoimmunity. 15699112_The mechanisms underlying the breakdown of self-tolerance in Aire-deficient mice are identified together with strain-dependent target-organ specificity of the autoimmune disease. 16111640_Aire exerts its tolerance-promoting function by negative selection of T effector cells. 16172259_Genetic background strongly influences the pattern of organs that were targeted and the severity of the targeting in AIRE deficiency. 16628255_Aire may regulate the survival of autoreactive T cells beyond transcriptional control of self-protein expression in the thymus 16709926_propose that Aire, in addition to its function in the thymus, also has a peripheral regulatory role by controlling the development of antigen-presenting cells and marginal zone B-cell activation 16785524_Ectopic expression of type II collagen (CII)in medullary thymic epithelial cells and the corresponding central tolerance to CII are AIRE independent. 16982213_Aire deficient antigen presenting cells have a defect receptor mediated internalization of Candida which affects kinase activation, likely altering the innate Candida immune response 17039258_NF-kappaB2 downstream of lymphotoxin beta receptor plays an important role in the regulation of central tolerance in an Aire-dependent manner 17116738_The eye disease that spontaneously develops in Aire-deficient mice is targeted against a single antigen whose expression is controlled within the thymus by Aire. 17360567_transcription of the Mucin 6 gene in thymic medullary epithelial cells is indeed Aire-dependent 17513766_Aire functions as a transcription factor and directly regulates the expression of a large number of, but not all, autoantigens, providing a molecular basis for the defect in negative selection in Aire-deficient mice. 17599412_The data underline a direct role for Aire in tissue-restricted antigens expression and suggest that modulation of Aire has a potential to control central tolerance and autoimmunity. 17908938_Aire does not have a direct impact on the division of thymic epithelial cellss in vitro or in vivo but, rather, induces their apoptosis. 17938200_These findings reveal critical steps by which AIRE regulates the transcription of genes that control central tolerance in the thymus. 17947641_was minimal overlap between the sets of genes controlled by Aire and lymphotoxin 17974569_Combined homology modeling and in vitro data now show how APECED mutations influence the activity of this transcriptional regulator. 17984055_Aire deficiency was found to cause severe blockage at the transition from single positive 3 to single positive 4 thymocytes 17991771_aire(-/-) mice exhibited autoimmunity in all organs normally targeted in this model, indicating that microbial conditioning is not required for activation of autoreactive T cells relevant to polyendocrinopathy-candidiasis-ectodermal dystrophy 18209027_Aire mRNA is expressed in the testis;Aire protein is expressed only sporadically in spermatogonia and spermatocytes 18322189_Aire-positive cells are specifically localized to the perinuclear region of thymic epithelium or, more precisely, to the cortico-medulla junction and medulla, correlating with the site of negative selection. 18350550_support a model where the initial differentiation of Aire+ medullary thymic epithelial cells involves receptor activator of NF-kappaB (RANK)-RANKL interactions with lymphoid tissue inducer cells 18414681_These results may demonstrate a mechanism by which autoimmune predisposition to phenotypes distinct from APS 1 can be mediated in a dominant-negative fashion by Aire. 18566406_absence of Ltbetar has no effect on the expression of Aire and Aire-dependent tissue-specific Ags 18687966_study reports the identification of extrathymic Aire-expressing cells (eTACs) resident within the secondary lymphoid organs; these cells are capable of interacting with and deleting naive autoreactive T cells 18755889_B cells must be present for Aire-deficient mice to develop fulminant infiltrates. 18780794_Aire promotes ectopic gene expression by a generic mechanism that does not depend on any particular characteristics or transcription mechanisms operating in medullary epithelial cells 18828139_study concludes that Aire has little or no effect on regulatory T cell development in general and NKT cells in particular 18832676_Aire-mediated apoptosis are postmitotic MTEC that have not yet completed their terminal differentiation program. 18836079_findings support the 'terminal differentiation' model of Aire function: as MECs mature, they transcribe more PTA genes, culminating in a cell population that is both capable of presenting antigens and can draw on a large repertoire of antigens to present. 18838677_greater variability in Aire-controlled ectopic gene expression than in Aire-independent transcripts 18840680_Aire's activity as a histone-binding module mediates the thymic display of peripheral tissue antigens that promotes self-tolerance and prevents organ-specific autoimmunity. 19011083_results suggest a cell-intrinsic role for AIRE in peripheral dendritic cells by regulating IFN-gamma-receptor signaling and point toward complementary mechanisms by which AIRE is involved in maintaining tolerance 19015306_Data suggest that Aire controls the differentiation of medullary thymic epithelial cells (mTECs), thereby organizing the mTEC integrity that enables tissue-restricted antigen expression from terminally differentiated mTECs in the thymic microenvironment. 19077444_Aire and Foxp3 are present in the particular TNC microenvironment which has previously been shown to support thymic selection. The differential localization of these two markers suggests a role for TNC in nTreg development. 19148669_the development/maintenance of thymic metallophilic macrophages is executed via LT beta R circumventing the Aire transcription factor 19234163_NZB mice demonstrate a complex thymic phenotype, including a lack of the autoimmune regulator (Aire), early defects in thymic epithelial cell expansion, and evidence for altered NF-kappaB2 signaling. 19261265_gammadelta T cells develop independently of Aire and are unlikely to have a significant pathogenetic or protective role in autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy. 19264867_Varying intrathymic autoantigen expression may modulate thyroid autoimmunity. This suggests that Aire deficiency may contribute more to developing Hashimoto's thyroiditis than Graves' disease. 19265170_Many of the orthologous genes putatively controlled by autoimmune regulator AIRE in human thymus, which then become autoantigens in autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy (APECED), are also controlled by Aire in mice. 19365590_Specific immune events that predict squamous metaplasia of the cornea in Aire deficiency, were determined. 19479827_Aire-dependent expression of type II collagen occurs in medullary thymic epithelial cells, implying that there is central tolerance to self antigens found in articular cartilage. 19487417_Data suggets that Aire expression during the perinatal period is both necessary and sufficient to induce long-lasting tolerance and avoid autoimmunity. 19651488_protein deficiency leads to impaired iNKT cell development 19721455_show that Aire deficiency affects all medullary thymic epithelial cell types inducing a general change in the thymic medulla. 19923453_Aire regulates the production of chemokine CCR4 and CCR7 ligands in medullary thymic epithelial cells and alters the coordinated maturation and migration of thymocytes. 20085707_To study how Aire targets and induces peripheral-tissue self-antigen study identified a set of proteins that associate with Aire fall into 4 major functional classes: nuclear transport, chromatin binding/structure, transcription and pre-mRNA processing. 20226168_These results suggest that transcription factors such as Aire and Deaf1, which exert global transcriptional regulatory functions, may play important roles in self-renewal of ESCs and maintaining ESC in a transcriptionally hyperactive state. 20237294_Aire-deficient non-obese diabetic (NOD) mice at 5 weeks of age demonstrate clinical signs of aqueous-deficient keratoconjunctivitis sicca, consistent with human Sjogren's syndrome. 20308365_Lymph node-resident lymphatic endothelial cells express multiple peripheral tissue antigens independent of the autoimmune regulator (Aire). 20404099_Medullary thymic epithelial cells express Aire protein and are in close contact with thymic dendritic cellsl 20414703_Down-regulation of Aire is associated with autoimmune type 1 diabetes mellitus. 20483761_positive selection and self-reactivity of the global Treg repertoire are not controlled by Aire-dependent tissue-specific antigens 20615959_Aire's histone H3-binding module is necessary for Aire-mediated regulation of gene expression and central tolerance induction. 20668237_Data show that intrinsic regulation of peripheral T cells by CBL-B serves a uniquely critical role as a failsafe against clinical onset of autoimmune disease in AIRE deficiency. 21035305_demonstrated that Aire KO mice have a significant and reproducible lower frequency of CD4+Foxp3+ cells and a higher expression of Th17 markers in their thymus, compared to wild type (WT) mice 21048786_Aire has an important role in Dsg3 expression as well as in selection of T cells that help B cells to produce anti-Dsg3 IgG in thymus. 21300913_XCL1-mediated accumulation of dendritic cells in the thymic medulla contributes to naturally occurring regulatory T cell development and is regulated by Aire. 21352256_these results support the importance of the peripheral AIRE expression in the maintenance of immunological tolerance. 21505196_Aire controls thymic negative selection by regulating the indirect presentation of mTEC-derived antigens by thymic dendritic cells. mTECs can mediate tolerance by direct presentation of Aire-regulated antigens to both CD4 and CD8 T cells. 21628060_Aire interacted with the TLR1, TLR3, and TLR8 promoters and increased the luciferase transcriptional activity of these promoters in RAW264.7 cells. 21683072_Aire cells are derived from mTECs and exhibit characteristic natures of antigen presenting cells, incl. several distinct abilities: to express a variety of peripheral tissue-specific antigens and to produce immunoproteasome and immunological synapses 21746930_CD8(+)CD28(low) regulatory T lymphocytes from AIRE-deficient mice are transcriptionally and phenotypically normal and exert efficient suppression of in vitro immune responses, but completely fail to prevent experimental colitis in vivo. 21819345_this study identifies a correlation between the presence of AIRE and proteasomal breakdown of cyclin B2, which leads us to speculate that cyclin B2 could be a target of AIRE's E3-ubiquitin ligase activity 21952165_Aire represents an early molecular switch imposing a suppressive MSC phenotype via regulation of Eta-1. 22036612_a positive correlation between AIRE expression and histone H3 lysine 4 trimethylation, an active chromatin mark, was found in the AIRE promoter in human and mouse thymic epithelial cells 22203960_Aire unleashes stalled RNA polymerase to induce ectopic gene expression in thymic epithelial cells. 22219212_results suggest that immune-mediated ovarian follicular depletion is a mechanism of infertility in Aire(-/-) mice 22239103_our results suggest that DNA-PKcs may interact with Aire to promote the expression of Toll-like receptors in RAW264.7 cells. 22425250_demonstrated a Rank-mediated reciprocal link between dendritic epidermal T cell progenitors and Aire(+) medullary thymic epithelial cell maturation 22447927_polymorphisms within the NOD Ica1 core promoter may determine AIRE-mediated down-regulation of ICA69 expression in medullary thymic epithelial cells. 22490868_Mutant Aire-mediated central tolerance to myelin protein zero initiates an autoimmune T helper cell type (Th)1 effector response toward peripheral nerves. 22540148_Aire promotes the self-renewal of embryonic stem cells through the activation of Lin28 and the inhibition of the let-7 microRNA family. 22564670_transcriptional activity determines expression profile of peripheral tissue antigen genes in medullary thymic epithelial cells 22883565_Aire can play a role as a controller of transcription of miRNAs located within genomic regions encompassing ORF and/or mRNA genes. 23041971_These findings suggest that the capacity of Aire to regulate expression of a huge array of tissue-restricted antigens relies solely on an unconventional transcriptional mechanism, without intermediary transcription factors. 23116172_These results indicate a unique DNA methylation profile that is independent of Aire in medullary thymic epithelial cells. 23265639_These findings add to the understanding of how specific DC subtypes regulate the early responses during T cell-dependent antibody responses within the spleen and further define the role of AIRE in the periphery as regulator of self-antigen expression and lymphocyte migration. 23319629_Data indicate that Aire requires its PHD2 domain to effectively impose immunological tolerance. 23370329_Aire deficiency promotes TRP-1-specific immune rejection of melanoma. 23456700_[review] The autoimmune regulator Aire is a critical transcription factor for generating central tolerance in the thymus and is highly conserved between human and mouse AIRE proteins. 23471412_Aire-mediated expression of peripheral tissue antigens drives the thymic development of a subset of organ-specific T(regs), which are likely coopted by tumors developing within the associated organ. 23487421_Blockade of B7-1/B7-2 in NOD.AireGW/+ mice results in fulminant, early-onset autoimmune peripheral neuropathy. 23589212_These findings reveal a mutual interdependence of miRNA and Aire in the regulation of promiscuous gene expression in purified mouse and human thymic epithelial cells 23849800_There is an age-related association between Aire and regulatory T cells (Tregs) in the susceptibility to experimental autoimmune encephalomyelitis. 23850677_Aire can facilitate early chondrocyte differentiation by expression of Bmp2 through altering the histone modification status of the promoter region of Bmp2. 24434558_Aire-containing complexes include 7SK RNA, the latter interaction disrupted by HNRNPL knockdown, suggesting that HNRNPL may partake in delivering inactive P-TEFb to Aire 24464130_We demonstrated the importance of Aire's interaction with the ATF7ip-MBD1 protein complex in maintaining central tolerance 24516201_Results indicate the roles of Aire in the differentiation program of medullary thymic epithelial cells (AEL-mTECs). 25157574_Aire expression in thymic epithelial cells was decreased in germ-free mice without commensal bacteria. 25220213_The results reveal a cooperation between medullary thymic epithelial cells and CD8alpha+ dendritic cells for presentation of Aire-induced self-antigens to developing thymocytes. 25689230_subfertility in Aire(-/-) mice is dependent on a functional adaptive immune system thus confirming its autoimmune aetiology. 25791085_Aire promotes the perinatal generation of a distinct compartment of Foxp3(+)CD4(+) regulatory T (Treg) cells, which stably persists in adult mice. 26006015_Data show that sirtuin 1 (Sirt-1) mediated deacetylation of Aire protein was required for the transcriptional-transactivational potential of Aire. 26034170_AIRE-dependent versus AIRE-independent tissue-restricted antigens project nonredundant representations of peripheral tissues in the thymus. 26070482_expressed in thymic but not peripheral B cells 26112418_Our results identify Aire as an important regulator of peripheral T cell homeostasis in gastrointestinal tissues. Given a suitable trigger the absence of peripheral Aire leads to dysregulated T cell proliferation and disease. 26168014_Data indicate that keratin 17 (k17) regulates autoimmune regulator Aire to promote skin tumorigenesis. 26216992_Aire's collaboration with the bromodomain-containing protein, Brd4, uncovered correspondence between those genes induced by Aire and those inhibited by a small-molecule bromodomain blocker. 26237550_Aire increased the diversity of the thymic epithelial cell transcriptome and seemed to target genes expressed in a minority of cells, and increased expression of its target genes. 26453754_Aire's unique property of generating a self-tolerant T cell repertoire is functional only in medullary thymic epithelial cells. 26527800_Data indicate a highly conserved noncoding DNA element that is essential for Autoimmune regulator (Aire) gene expression. 26548750_Aire may have a role in inducing Th1 and Th17 differentiation by upregulating cytokine expression in dendritic cells. 26579596_Molecular characterization of the functional domains of Aire has revealed multiple binding partners that assist Aire's function in altering gene transcription and chromatin remodeling. 26729097_Aire-expressing dendritic cells inhibited TCR signaling pathways and decreased quantity of CD4+IFN-gamma+ autoreactive T cells. Aire plays role in maintenance of peripheral immune tolerance. 26999605_results indicate that in females, estrogen induces epigenetic changes in the AIRE gene, leading to reduced AIRE expression under a threshold that increases female susceptibility to autoimmune diseases. 27072778_androgen control of an intrathymic Aire-mediated tolerance mechanism contributes to gender differences in autoimmunity. 27130899_this study shows that Aire enforces immune tolerance by ensuring that distinct autoreactive T cell specificities differentiate into the Treg cell lineage 27281783_Results indicated that AIRE influenced gene expression in germ cell 1 spermatogonial (GC1-spg) cells by acting at multiple levels, including transcription, translation, RNA processing, protein transport, protein localization, and protein degradation, thus setting the foundation in understanding the functional role of AIRE in germ cell biology. 27365398_AIRE, which is phosphorylated on two specific residues near its N terminus, then binds to the F-box protein 3 (FBXO3) E3 ubiquitin ligase. In turn, this SCF(FBXO3) (SKP1-CUL1-F box) complex ubiquitylates AIRE, increases its binding to the positive transcription elongation factor b (P-TEFb), and potentiates its transcriptional activity. 27401343_findings identified two novel stages in the differentiation program of Aire(+) Medullary thymic epithelial cells. 27505711_This finding is the first direct evidence that Aire also plays a role in controlling medullary thymic epithelial cells-thymocyte adhesion. 27563821_allelic variants of one AIRE SNP may differentially shape the melanoma antigen-specific T cell repertoire potentially influencing susceptibility to melanoma 27571405_A mutation in LYN, an inhibitory protein tyrosine kinase that is implicated in systemic autoimmunity, combines with an Aire mutation to provoke organ-specific autoimmunity. 27851927_Aire exerts multi-faceted autoimmune control that extends to a population of innate-like T cells. 27916941_we found that Aire up-regulated TLR3 expression and modulated the downstream cytokine expression and nuclear factor-kappaB (NF-kappaB) of the TLR3 signaling pathway. 27941786_this study provides insight into the complex molecular mechanisms that control the medullary thymic epithelial cells-specific expression of Aire 28135252_this study shows that Aire and some of its partners, notably those implicated in the DNA-damage response, preferentially localize to and activate long chromatin stretches that are overloaded with transcriptional regulators, known as super-enhancers 28742026_Aire associates with spindle-associated proteins in mouse embryonic stem cells. Loss of function analysis revealed that Aire was important for centrosome number regulation and spindle pole integrity specifically in mouse embryonic stem cells. 29021375_the maternal microbiome influences Treg differentiation in the thymus of offspring through GPR41-mediated Aire expression 29101311_In an insulin-promoter model, lack of Aire in medullary thymic epithelial cells, but not in BM-derived APCs, impaired clonal deletion and Treg production. In an Aire-promoter Tg model, both mTECs and BM-derived APCs independently induced clonal deletion without Aire. Treg production was impaired by the lack of Aire in mTECs, but not in BM-derived APCs, suggesting an essential but not universal role for Aire in tolerance. 29193031_The Aire fl/fl strain is a suitable model to scrutinize the cell specific function of Aire in distinct biological processes. Medullary thymic epithelial cell (mTEC)-restricted expression of autoimmune regulator (Aire) is essential for establishment of immune tolerance. Aire was also shown to be expressed in cells of hematopietic and reproductive lineages. 29285761_This study re-defines requirements for late stage intrathymic alphabetaT-cell development, and demonstrates that Aire controls a CCR6-CCL20 axis that determines the developmental makeup of the intrathymic regulatory T-cell pool. 29297233_The results demonstrate that Aire expressed by bone marrow-derived dendritic cells plays an important role in the maintenance of homeostasis by regulating TRA expression and the differentiation of T cell subsets. 29335648_Together, Brg1 and Aire fine-tune the expression of tissue-specific genes at levels that prevent toxicity yet promote immune tolerance 29753741_Two-week hindlimb unloading did not alter percentage of CD4+CD8+ cells in thymocytes. Hindlimb unloading reduced cellularity of thymic epithelial cells expressing Aire. 29867946_Aire plays a role in the process of medullary thymic epithelial cell-thymocyte adhesion 30453645_At 5 weeks of age (wk), Aire-/- mice show minimal tissue dysfunction and destruction compared to 7 wk Aire-/-, which exhibit severe dry eye, poor tear secretion, extensive lymphocytic infiltration, reduced functional innervation, and increased vascularization. 31023489_The results showed that Aire deficiency caused increased number of TFH cells, both in vivo and in vitro. Further studies showed that Aire deficiency promoted TFH differentiation through the upregulation of ICOSL and IL-6 in DCs. Thus Aire could suppress the expression of ICOSL and IL-6 to inhibit TFH cell differentiation. 31942786_CARD domain of AIRE enhances caspase-3 expression through possible direct DNA binding and triggers non-apoptotic downstream signaling in cultured spermatogonial cells. 32242017_Aire utilizes its caspase activation recruitment domain (CARD) to form filamentous homo-multimers in vitro, and this assembly mediates foci formation and transcriptional activity. 32547551_Aire Gene Influences the Length of the 3' UTR of mRNAs in Medullary Thymic Epithelial Cells. 32588888_Germline PTEN mutations are associated with a skewed peripheral immune repertoire in humans and mice. 32845012_A novel method to identify Post-Aire stages of medullary thymic epithelial cell differentiation. 34126085_Multiple Lesions Contribute to Infertility in Males Lacking Autoimmune Regulator. 34272228_Extrathymic Aire-expressing cells support maternal-fetal tolerance. 34293711_AIRE-overexpressing BMDCs suppress TFH cells through ICOSL to prevent and attenuate autoimmune diabetes in NOD mice. 34477806_Mechanistic dissection of dominant AIRE mutations in mouse models reveals AIRE autoregulation. 34518235_Aire regulates chromatin looping by evicting CTCF from domain boundaries and favoring accumulation of cohesin on superenhancers. 34700158_Autoimmune regulator act in synergism with thymocyte adhesion in the control of lncRNAs in medullary thymic epithelial cells. 34710738_Attenuate ICOSL and IL-27 in Aire-overexpressing DC2.4 cells suppress TFH cell differentiation. 34767455_Single-cell multiomics defines tolerogenic extrathymic Aire-expressing populations with unique homology to thymic epithelium. 34930780_Aire Controls Heterogeneity of Medullary Thymic Epithelial Cells for the Expression of Self-Antigens. 35058362_Pathogenic TNF-alpha drives peripheral nerve inflammation in an Aire-deficient model of autoimmunity. 35061506_The acetyltransferase KAT7 is required for thymic epithelial cell expansion, expression of AIRE target genes, and thymic tolerance. 35172142_Aire suppresses CTLA-4 expression from the thymic stroma to control autoimmunity. 35331137_The absence of the autoimmune regulator gene (AIRE) impairs the three-dimensional structure of medullary thymic epithelial cell spheroids. 35394861_PD-1 cooperates with AIRE-mediated tolerance to prevent lethal autoimmune disease. 36409920_CTLA-4 on thymic epithelial cells complements Aire for T cell central tolerance. |
ENSG00000160224 |
AIRE |
62.653537 |
7.3102324086 |
2.869917 |
0.44570303 |
39.542045 |
0.00000000032107167401478816003247290969239719632399854276627593208104372024536132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000048040901164963191645456704699165828564844105130759999155998229980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
114.898253678742 |
36.0637591936668 |
15.7174556508331 |
3.96504861328281 |
| ENSMUSG00000000732 |
50723 |
Icosl |
protein_coding |
Q9JHJ8 |
FUNCTION: Ligand for the T-cell-specific cell surface receptor ICOS. Acts as a costimulatory signal for T-cell proliferation and cytokine secretion; induces also B-cell proliferation and differentiation into plasma cells. Could play an important role in mediating local tissue responses to inflammatory conditions, as well as in modulating the secondary immune response by co-stimulating memory T-cell function. During pregnancy, may function to skew the cytokine of maternal T-cells toward immunoprotective Th2 phenotype. |
Adaptive immunity;Alternative splicing;B-cell activation;Cell membrane;Disulfide bond;Glycoprotein;Immunity;Immunoglobulin domain;Membrane;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:50723; |
cytoplasmic ribonucleoprotein granule [GO:0036464]; external side of plasma membrane [GO:0009897]; membrane [GO:0016020]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; signaling receptor binding [GO:0005102]; isotype switching [GO:0045190]; positive regulation of activated T cell proliferation [GO:0042104]; positive regulation of interleukin-4 production [GO:0032753]; regulation of cytokine production [GO:0001817]; signal transduction [GO:0007165]; T cell activation [GO:0042110]; T cell receptor signaling pathway [GO:0050852] |
11418641_This molecule acts as a costimulatory counterreceptor by augmenting the recall responses of CD8+ T cells. 12145647_These studies demonstrate that T(R) cells and the ICOS-ICOS-ligand signaling pathway are critically involved in respiratory tolerance and in downregulating pulmonary inflammation in asthma. 12370365_The involvement of the B7h-Icos costimulatory pathway has been demonstrated in the development and progression of collagen-induced arthritis. 12456022_ICOS-L is a ligand for ICOS and plays an important functional role in the activation of memory T cells by endothelial cells [review] 12594252_While interactions of B7RP-1 with ICOS have no effect on the migration of T cells into B cell follicles, they are required for Th1 and Th2 cells to support fulminant B cell responses in vivo. 12714510_B7-H2 on B cells is important for recruiting T-cell help via its interaction with ICOS and plays a critical role in costimulating humoral immune responses. 12853164_data suggest that ICOS/B7RP-1 interactions may not affect the organogenesis, but involve in the functional development of Peyer's patches 12865411_the ICOS-B7h pathway has roles in regulating alloimmune responses in vivo 14615582_Th cells, when activated with B7h-deficient APC, exhibited reduced proliferation and IL-2 production. 15636609_ICOS-B7h has a role in transplantation tolerance to MHC class II mismatched skin grafts 15728513_Experiments with mice genetically engineered for the absence of B7-related protein 1 (B7RP-1KO) demonstrate that efficient T helper 2 (TH2) cell sensitization and effector function occur in the absence of the ICOS-B7RP-1 costimulatory pathway. 15880041_ICOS-B7h pathway is critical in the activation of effector/memory T cells that are necessary for the progression of chronic rejection. 16081804_The B7RP-1/ICOS interaction plays an essential role in the development of CXCR-positive helper T cells in vivo, which preferentially migrate to the germinal center B cell zone where they provide cognate help to B cells. 16540559_ICOSL is upregulated in nephritic glomeruli, where it locally reduces accumulation of T cells and macrophages and attenuates renal injury. 16621988_critical role played by ICOS-L-expressing and interleukin 10-producing dendritic cells from Chlamydia-infected mice in the infection-mediated inhibition of allergic responses 16887997_B7h shedding defines a novel immunoregulatory mechanism by which toll-like receptor (TLR)7/8 and TLR9 signals can enhance T cell help during antibody responses. 17039566_These results suggest that the ICOS/B7RP-1 interaction plays a critical role in the pathogenesis of uveitis. 17975322_The low costimulatory molecule expression (PD-L1, ICOS-L and CD40) may explain the suboptimal T cell activation by renal proximal tubular epithelial cells. 18294651_The generated a 3T3 cellular library retrovirally expressing mutants of the murine costimulatory B7h gene. Screening of this unbiased cellular library identified six residues of murine B7h that are critical for binding to the ICOS receptor. 18390703_a novel mechanism for the regulation of ICOSL expression in vivo and suggest that the ICOS costimulatory pathway is subject to negative feedback regulation by ICOSL down-regulation in response to ICOS expression. 19155489_Levels of follicular T helper cells and germinal center B cells in two different models of autoimmunity are shown to be dependent on the maintenance of the inducible T-cell co-stimulator (ICOS)/B7-related protein-1 (B7RP-1) pathway. 19208362_These findings implicate ICOSL-induced IL-10, but not IDO in the regulation of BM-derived pDC function. 19249753_compared T cell responses of WT, CD28 knockout (KO), B7-H2 KO, and CD28-B7-H2 double KO (DKO) mice in a model of delayed-type hypersensitivity 20190137_The data suggest that ICOSL plays a significant role in immunoregulation and protective immunity against Chlamydia infections 21364749_A role for ICOS costimulation in the maintenance of effector memory but not central memory CD4 T cells. 21402623_expression is essential for allergic airway hyperresponsiveness 21636296_demonstrate that inducible costimulator (ICOS) provides a critical early signal to induce the transcription factor Bcl6, and Bcl6 then induces CXCR5, the canonical feature of follicular helper CD4(+) T cells 21768353_Findings suggest that the noncanonical NF-kappaB pathway regulates the development of Tfh cells by mediating ICOSL gene expression in B cells. 21958057_Results indicate that ICOSLG blockade may enhance cytotoxity in allogeneic mixed lymphocyte-hematologic neoplasm cell reactions. 22154192_Induction of IL-10 producing regulatory T cells mainly relied on the inducible costimulator ligand (ICOSL)/ICOS axis 22172879_ICOS/B7h signal plays an important role in direct allorecognition, eliciting allogeneic responses in vitro. 22686515_lineage-specific transgenic expression on dendritic and plasma cells generates different outcome upon ICOS costimulation 22732700_Data suggest that knockdown of Icosl in mouse leukemic cells may significantly enhance graft versus leukemia effect after allogeneic bone marrow transplantation. 23498793_In cardiac transplantation apoptosis of CD8(+) T cells in recipient draining lymph nodes was enhanced by pretreatment with donor specific transfusion and impaired ICOS/B7h allorecognition. 23578385_ICOS-L maintains tolerance at the fetomaternal interface. 24449576_Data indicate that the signals provided by ICOSL-expressing B cells to Teff cells and Tfh cells are necessary for the development of arthritis. 24610013_Findings indicate the separable roles of delivery of antigens and ICOS-L by cognate B cells for follicular Th (Tfh) cell maturation and function. 24729612_the B7h-ICOS interaction may modulate the spread of cancer metastases 25002484_The need for additional immune suppression in the intestine reflects commensal microbe-driven T-cell activation through the accessory costimulation molecules ICOSL and OX40L in B7 deprived mice. 25108021_the costimulatory ligand ICOS ligand (ICOSL) is selectively downregulated on the surface of B cells in an ADAM17-specific manner, although it is not proteolitically processed by recombinant ADAM17 in vitro. 25317561_ICOSL is a molecular linkage between T-B interactional dynamics and positive selection for high-affinity bone-marrow plasma cell formation; study reveals a pathway by which follicular T-helper cells control the quality of long-lived humoral immunity 25552357_restoring control of the T follicular helper-germinal center B-cell axis by blocking the ICOS-ICOSL pathway reduced the development of atherosclerosis and the formation of tertiary lymphoid organs. 25590646_There was a clearly positive correlation between the presence of IL-17-producing cells and ICOS expression in ICOSL KO mice; it also showed that Th17 was involved in the pathological tissue remodeling in liver fibrosis induced by schistosomiasis. 25769613_ICOS-ICOS-L interaction promoted cytokine production and survival in type 2 innate lymphoid cells through STAT5 signaling in asthma. 25786178_Selective ablation of ICOSL in CD11c+ cells, but not in B cells, dramatically ameliorates kidney and lung inflammation in lupus-prone transgenic mice. 27138044_this study shows that sICOSL in the serum of healthy donors increases in an age-dependent manner and that the matrix metalloproteinase inhibitor (MMPI) could suppress sICOSL production 27183569_study concludse that lymphopenia-driven autoimmunity and the development of Tfh17 cells in Rasgrp1-deficient mice occur independently of CD275(B7-H2) 27798154_results showed that monocyte-derived osteoclast (OC)-like cells (MDOCs) express B7h during their differentiation, and that B7h triggering reversibly inhibits OC differentiation and function both in vitro and in vivo 28002569_The expression of ICOSL in the cornea and the ICOS-mediated induction of Foxp3+ CD4+ regulatory T cells may contribute to successful corneal allograft survival. 28814605_study confirms the importance of ICOSL shedding in ICOS/ICOSL function and expression and it identifies ADAM10 as the most important sheddase for controlling ICOSL levels 30747722_work identified a potentially novel role for ICOSL, which serves as an endogenous alphavbeta3-selective antagonist to maintain glomerular filtration. 33753483_ICOS ligand and IL-10 synergize to promote host-microbiota mutualism. 34710738_Attenuate ICOSL and IL-27 in Aire-overexpressing DC2.4 cells suppress TFH cell differentiation. |
ENSG00000160223 |
ICOSLG |
17.844536 |
3.2297487320 |
1.691422 |
0.60999730 |
7.654947 |
0.00566168634235126363302148533307445177342742681503295898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0206708548338193805193885310700352420099079608917236328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.1195388881731 |
11.8113514829405 |
8.39679527370093 |
2.91911204976671 |
| ENSMUSG00000000739 |
57429 |
Sult5a1 |
protein_coding |
Q3UQP7 |
Human_homologues FUNCTION: Sulfotransferase that utilizes 3'-phospho-5'-adenylyl sulfate (PAPS) as sulfonate donor to catalyze the sulfate conjugation of a wide variety of acceptor molecules bearing a hydroxyl or an amine groupe. Sulfonation increases the water solubility of most compounds, and therefore their renal excretion, but it can also result in bioactivation to form active metabolites. Displays broad substrate specificity for small phenolic compounds. Plays an important role in the sulfonation of endogenous molecules such as steroid hormones and 3,3'-diiodothyronin (PubMed:16221673, PubMed:12471039, PubMed:22069470, PubMed:21723874, PubMed:10199779, PubMed:7834621). Mediates the sulfate conjugation of a variety of xenobiotics, including the drugs acetaminophen and minoxidil (By similarity). Mediates also the metabolic activation of carcinogenic N-hydroxyarylamines leading to highly reactive intermediates capable of forming DNA adducts, potentially resulting in mutagenesis (PubMed:7834621). May play a role in gut microbiota-host metabolic interaction. O-sulfonates 4-ethylphenol (4-EP), a dietary tyrosine-derived metabolite produced by gut bacteria. The product 4-EPS crosses the blood-brain barrier and may negatively regulate oligodendrocyte maturation and myelination, affecting the functional connectivity of different brain regions associated with the limbic system. {ECO:0000250|UniProtKB:P17988, ECO:0000269|PubMed:10199779, ECO:0000269|PubMed:12471039, ECO:0000269|PubMed:16221673, ECO:0000269|PubMed:21723874, ECO:0000269|PubMed:22069470, ECO:0000269|PubMed:35165440, ECO:0000269|PubMed:7834621}.; + ;FUNCTION: Sulfotransferase that utilizes 3'-phospho-5'-adenylyl sulfate (PAPS) as sulfonate donor to catalyze the sulfate conjugation of catecholamines, phenolic drugs and neurotransmitters. Is also responsible for the sulfonation and activation of minoxidil. Mediates the metabolic activation of carcinogenic N-hydroxyarylamines to DNA binding products and could so participate as modulating factor of cancer risk.; + ;FUNCTION: Sulfotransferase that utilizes 3'-phospho-5'-adenylyl sulfate (PAPS) as sulfonate donor to catalyze the sulfate conjugation of phenolic monoamines (neurotransmitters such as dopamine, norepinephrine and serotonin) and phenolic and catechol drugs.; + ;FUNCTION: Sulfotransferase that utilizes 3'-phospho-5'-adenylyl sulfate (PAPS) as sulfonate donor to catalyze the sulfate conjugation of phenolic monoamines (neurotransmitters such as dopamine, norepinephrine and serotonin) and phenolic and catechol drugs. |
Proteomics identification;Reference proteome;Transferase |
Human_homologues NA; + ;NA; + ;NA; + ;NA |
|
|
cytoplasm [GO:0005737]; sulfotransferase activity [GO:0008146]; sulfation [GO:0051923] |
Human_homologues 11156380_Observational study of gene-disease association. (HuGE Navigator) 11207031_Observational study of genotype prevalence. (HuGE Navigator) 11219777_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 11692076_Observational study of gene-disease association. (HuGE Navigator) 11692076_The high activity SULT1A1*1 allozyme protects against dietary and/or environmental chemicals involved in the pathogenesis of colorectal cancer. 11804685_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 11804685_genetic polymorphism in SULT1A1 gene may be associated with increased lung cancer risk 12162852_7-OH-flavone sulfotransferase followed Michaelis-Menten kinetics 12165038_Observational study of gene-disease association. (HuGE Navigator) 12165038_The SULT1A1 R213H polymorphism is not linked with colorectal cancer in an elderly Australian population. 12402313_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 12419790_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 12419790_There is an an association between genotype and survival of breast cancer patients receiving tamoxifen therapy 12419832_Observational study of gene-disease association. (HuGE Navigator) 12455060_Observational study of gene-disease association. (HuGE Navigator) 12455060_SULT1A1 his(213) allele is important in the development of esophageal cancer in men. 12468438_Observational study of gene-disease association. (HuGE Navigator) 12469224_Observational study of gene-disease association. (HuGE Navigator) 12469224_the relationship among the polymorphisms of this enzyme and SULT1A2 in different types of cancers in Taiwanese 12471039_the crystal structure of SULT1A1 provides the molecular basis for substrate inhibition and how the enzyme sulfonates a wide variety of lipophilic compounds 12725421_Observational study of gene-disease association. (HuGE Navigator) 12761191_the differential substrate specificity of the two enzymes M-PST and P-PST for the thirteen drug compounds tested 12814450_Observational study of genotype prevalence. (HuGE Navigator) 12867416_study of arginine residues in the active site of human phenol sulfotransferase 12867492_P. 1038 '...clearly expressed...SULT1A1...' P. 1040 '...SULT1A1...which belongs to phase II metabolism,...significantly expressed in HepG2.' 14520706_Observational study of gene-environment interaction. (HuGE Navigator) 14618622_Observational study of gene-environment interaction. (HuGE Navigator) 14618622_We conclude that smoking increases risk of colorectal adenomas and that SULT1A1 and NAT2 only modestly modify this association. 14642079_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 14643027_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 14643027_Sulfotransferase 1A1 polymorphism is associated with bladder cancer 14648207_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 14648207_SULT1A1 slow acetylator genotype might modulate the effect of carcinogenic arylamines contained in tobacco smoke, and has a tendency to present a higher risk for highly differentiated tumors among heavy-smokers. 14688021_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 14688021_SULT1A1 polymorphism is associated with susceptibility to lung cancer in relation to tobacco smoking 14871892_In SULT1A1, substrate inhibition occurs with pNP as the substrate but not with dopamine; residue Phe-247 of SULT1A1, which interacts with both p-nitrophenol molecules in the active site, is important for substrate inhibition 14973106_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 15090717_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 15093672_Observational study of gene-disease association. (HuGE Navigator) 15093672_SULT1A1 His allele was positively associated with the risk of breast cancer in Chinese women 15122594_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 15318931_Observational study of gene-disease association. (HuGE Navigator) 15377847_Observational study of gene-disease association. (HuGE Navigator) 15383623_SULT1A1 promoter was shown to be dependent on the presence of Sp1 and Ets transcription factor binding sites. 15455371_Observational study of gene-disease association. (HuGE Navigator) 15516026_Observational study of gene-disease association. (HuGE Navigator) 15604994_Observational study of gene-disease association. (HuGE Navigator) 15632378_Observational study of gene-environment interaction and pharmacogenomic / toxicogenomic. (HuGE Navigator) 15743503_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 15894657_No significant association found for breast cancer risk. 15894657_Observational study of gene-disease association. (HuGE Navigator) 15949571_Observational study of gene-disease association. (HuGE Navigator) 15952058_Observational study of gene-disease association, gene-gene interaction, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 15970794_Observational study of gene-disease association. (HuGE Navigator) 15987423_Observational study of gene-disease association, gene-gene interaction, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 16006997_Observational study of gene-disease association. (HuGE Navigator) 16080486_Observational study of gene-disease association. (HuGE Navigator) 16103451_Observational study of gene-disease association. (HuGE Navigator) 16133548_Observational study of genotype prevalence. (HuGE Navigator) 16133548_SULT1A1*2 and SULT1A2*2 are the major allelic variants in the Korean population. 16137826_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 16141802_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 16175316_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 16175316_Postmenopausal women carrying the variant SULT1A1 His allele may be more susceptible to estrogen-induced carcinogenesis in mammary tissue. 16221673_x-ray crystallographic structure of human SULT1A1 with estradiol 16232327_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 16272171_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 16280036_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 16284375_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 16317586_Observational study of genotype prevalence. (HuGE Navigator) 16317586_SULT1A1 genotype did not correlate with any prognostic or predictive markers associated with breast cancer. 16328031_Progressive SULT1A1 methylation within the promoter area of the gene occurs during breast carcinogenesis. 16380991_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 16395669_Observational study of gene-disease association. (HuGE Navigator) 16402077_Observational study of gene-disease association. (HuGE Navigator) 16418064_Sulfation of resveratrol in human liver: evidence of a major role for the sulfotransferases SULT1A1. 16425401_Observational study of gene-disease association. (HuGE Navigator) 16504378_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 16575574_Observational study of gene-disease association. (HuGE Navigator) 16637266_Observational study of gene-disease association. (HuGE Navigator) 16875543_Observational study of gene-disease association. (HuGE Navigator) 16926176_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 16985032_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 16985250_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 16985250_The SULT1A1*2 allele and long-term use of estrogen replacement therapy were associated with statistically significantly higher risk of endometrial cancer 17013894_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17074589_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17074589_Results indicate that the SULT1A1 genotype may play an important role in the risk of developing lung cancer, especially in cigarette smokers. 17189289_The presence of SULT1A1 gene deletions and duplications, representing an additional source of variability in the metabolic activity of this enzyme, is demonstrated. 17244352_Clinical trial of gene-environment interaction and pharmacogenomic / toxicogenomic. (HuGE Navigator) 17274372_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17293380_Apigenin, epicatechin, and resveratrol exhibited SULT1a1 allele-specific variation in sulfation, with SULT1A1*1 and *3 acting as normal-activity allozymes and SULT1A*2 as low-activity allozyme. 17372239_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 17372243_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 17479406_Observational study of gene-disease association. (HuGE Navigator) 17603900_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 17605044_Observational study of gene-disease association. (HuGE Navigator) 17605044_SULT1A1 polymorphism is associated with primary brain tumors 17619904_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17619904_SULT1A1 polymorphism is associated with urothelial cancer 17912498_Increased generation of urinary 8-hydroxy-2'-deoxyguanosine was found in betel-quid chewers with SULT1A1 and GSTP1 genotypes that affect susceptibility to DNA damage. 17912498_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 17947222_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17996038_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 18006944_Observational study of gene-disease association. (HuGE Navigator) 18259693_Observational study of gene-disease association. (HuGE Navigator) 18259693_SULT1A1/SULT1A2 gene complex showed suggestive haplotypic association in the family-based cardiovascular disease study, with the greatest increase in risk conferred by the SULT1A2 235T allele 18264785_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18318428_Observational study of gene-disease association. (HuGE Navigator) 18318428_a decreased single nucleotide polymorphism of CYP1A1 and an increased single nucleotide polymorphism for SULT1A1 and SULT1E1 genes may be risk factors for endometrial cancer in Caucasians. 18365755_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18365755_The aim of the study was to investigate NAT1, NAT2, GSTM1, GSTT1, GSTP1, SULT1A1, XRCC1, XRCC3 and XPD genetic polymorphisms, coffee consumption and risk of bladder cancer (BC) through a hospital-based case-control study. 18368507_Observational study of gene-disease association. (HuGE Navigator) 18368507_The study showed a null association of SULT1A1 polymorphism with familial prostate cancer risk in the Japanese population. 18447907_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18497059_Observational study of gene-disease association. (HuGE Navigator) 18497059_SULT1A1 polymorphism is associated with ovarian cancer 18499698_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18499698_The CYP1A2 and NAT1 but not SULT1A1 and NAT2 genotypes showed significant interactions with heavy smoking in women not men 18632753_Observational study of gene-disease association. (HuGE Navigator) 18794456_SULT1A1 mRNA is expressed in human skin, at similar levels in men and women. SULT1A1 levels are not altered by topical 17-beta-estradiol treatment. 18854828_Observational study and meta-analysis of gene-disease association. (HuGE Navigator) 18854828_The SULT1A1*2 revealed contrasting risk association for upper aerodigestive tract cancers and conferred significant increased risk of breast cancer to Asian but not Causcasian women in patients with multiple tobacco-related cancers 18990750_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18990750_Red meat intake was associated with increased colorectal cancer regardless of genotype, but the SULT1A1-638G>A variant modified the association between meat doneness and cancer risk. 19120511_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 19120511_SULT1A1 Arg213His polymorphism is associated with the development of urothelial cancer, especially among cigarette smokers exposed to hazardous chemicals. 19126640_Observational study of gene-disease association. (HuGE Navigator) 19126640_SULT1A1 gene is a risk modifier on environmental carcinogen in OSCC and the association of SULT1A1 haplotypes with the risk of OSCC might be modified by betel quid chewing. 19237513_he importance of human SULT1A1 as a homodimer was to maintain its structural stability, and the change of secondary structure was responsible for alternating its quaternary structure 19307236_Observational study of gene-disease association. (HuGE Navigator) 19322015_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 19322015_SULT1A1 genetic polymorphisms might modify the arsenic methylation profile and urothelial carcinoma progression. 19339270_Observational study of gene-disease association. (HuGE Navigator) 19343046_Observational study of gene-disease association. (HuGE Navigator) 19350537_Observational study of gene-disease association. (HuGE Navigator) 19350537_The variant SULT1A1 genotype (SULT1A1*1/SULT1A1*2 or SULT1A1*2/SULT1A1*2) was associated with a significantly increased lung cancer risk in cases. 19424794_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 19484729_human CYP2E1 and SULT1A1 activate an endogenous cellular molecule or a medium component to become mutagenic 19575027_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 19706609_para-Nitrophenyl sulfate activation of human sulfotransferase 1A1 is consistent with intercepting the E[middle dot]PAP complex and reformation of E[middle dot]PAPS. 19776291_Observational study of gene-disease association. (HuGE Navigator) 19789190_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 19863350_Observational study of gene-disease association. (HuGE Navigator) 19904771_study demonstrates that the loss of SULT1A1 appears to be a characteristic molecular signature of hepatocellular carcinoma. 19906068_The interaction between SULT1A1 and CYP1A2 can play an important role in hepatocarcinogenesis in the Chinese population. 19949855_Meta-analysis did not find a significant general relationship between SULT1A1 R213H polymorphism & the risk of breast cancer, but ethnic population analysis revealed a significantly increased breast cancer risk for HH allele carriers among Asians. 19949855_Meta-analysis of gene-disease association. (HuGE Navigator) 19956635_Uncategorized study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20032816_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 20142249_Observational study of gene-disease association. (HuGE Navigator) 20192879_Observational study of genetic testing. (HuGE Navigator) 20204402_Observational study of gene-disease association. (HuGE Navigator) 20214802_Observational study of gene-disease association. (HuGE Navigator) 20309015_Clinical trial of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20417180_the structures of SULT1A2 and an allozyme of SULT1A1, SULT1A1 *3, bound with 3'-phosphoadenosine 5'-phosphate at 2.4 and 2.3A resolution, respectively, were determined. 20437850_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 20505544_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 20505990_Meta-analysis of gene-disease association. (HuGE Navigator) 20528568_Observational study of gene-disease association. (HuGE Navigator) 20529763_Observational study of gene-disease association. (HuGE Navigator) 20565970_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20620409_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 20628863_Only NAT1 showed a significant lower DNA methylation rate in the control group than in the tamoxifen-resistant breast cancer group, and no significant difference in methylation was found in COMT, CYP1A1, CYP2D6, and SULT1A1 genes. 20634891_Observational study of gene-disease association. (HuGE Navigator) 20663177_Meta-analysis of gene-disease association. (HuGE Navigator) 20663177_Polymorphism of SULT1A1 Arg213His is associated with breast cancer. 20734064_Observational study of gene-disease association. (HuGE Navigator) 20881232_Observational study of gene-disease association. (HuGE Navigator) 20881232_SULT1A1 1/2 does not contribute to the variation in SULT1A1 enzymatic activity when the 3'-UTR SNPs are included in the statistical model 20936502_Observational study of genotype prevalence. (HuGE Navigator) 20936502_the statistical hypothesis that SULT1A1 and SULT1A2 alleles are independently distributed was rejected; a strongly positive linkage was detected between SULT1A1*2 and SULT1A2*2 alleles in Turkey population 21072184_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 21111704_mechanism of SULT1A1-catalyzed sulfation of adenosine 3',5'-diphosphate by para-nitrophenyl sulfate 21670965_SULT1A1 Arg213His polymorphism, ethnicity, and smoking may modulate environment-related cancer risk. 21695180_This meta-analysis demonstrates that there is no association between the SULT1A1 R213H polymorphism and colorectal cancer. 21977969_Women in Siberia with SNPS in CYP1A1 gene, in CYP1A2 gene,and in the SULT1A1 gene have an increased risk of development of breast cancer . 22011087_SULT1A1 Arg213His polymorphism is associated with breast cancer. 22081606_Stp1 is important for appropriate regulation of Stk1 function, hemolysin activity, autolysis, and GBS virulence 22524828_Arg213His polymorphism is not associated with lung cancer. 22678655_discussion of possible association between lower-activity of SULT1A1 with sudden cardiac death (both holiday sudden cardiac death in older adults and sudden infant death syndrome) [REVIEW] 22708928_We observed a previously unreported association between the SULT1A1 rs9282861 genotype and overall survival of breast cancer patients treated with adjuvant chemotherapy or tamoxifen. 23080433_The functional significance of the single nucleotide polymorphisms/haplotypes located upstream of the SULT1A1 start codon. 23157889_SULT1A1 variant allele increases breast cancer risk among subjects who were exposed to high smoked meat intake. 23358261_resequenced the SULT1A2 and SULT1A2 genes and identified 51 variations in ethnic Koreans, including 9 that were previously unknown. Allele frequencies, haplotype structures, LD blocks, and haplotype-tagging SNPs were determined. 23711090_Association between BRCA2 mutation and SULT1A1 gene deletion in male breast cancer emerged. 24010997_The SULT1A1 genetic variability is associated with cancer risk and response to therapy (review). 24039991_Data indicate that protein-ligand interaction energy by using docking Quantitative Structure-Activity Relationships(QSAR) models showed accuracy of 67.28%, 78.00% and 75.46%, for the isoforms SULT1A1, SULT1A3 and SULT1E1, respectively. 24307569_The SNP rs9282861 with GG genotype of SULT1A1 was associated with an elevated risk of total neural tube defects. 24763827_The results of this meta-analysis indicate that the SULT1A1 Arg213His polymorphism is associated with the risk bladder cancer under a recessive model. 25103078_The present study provided epidemiological evidence for a significantly increased risk of UCB in ever smokers with the Ala/Ala genotype of the GSTO1 gene and the Arg/Arg genotype of the SULT1A1 gene. 25194687_that SULT1A1 Arg213His polymorphism is associated with bladder cancer risk. 25225888_the SULT1A1 Arg213His polymorphism may contribute UADT cancer risk, but didn't show any association with breast cancer. 25314023_3'-Phosphoadenosine 5'-phosphosulfate binds antisynergistically to the subunits of the SULT1A1 dimer. 25370010_SULT1A1 is responsible for bioactivation of food genotoxicants 5-hydroxymethylfurfural and furfuryl alcohol. 25385181_Results indicate that SULT1A1 Arg(213)His may act as a low-penetrance risk allele for developing MBC and could be associated with a specific tumor subtype associated with HER2 overexpression. 25654087_The aim of the study was to assess whether selected single nucleotide polymorphisms of CYP1A1 and 2E1, GSTM1, GSTT1, and SULT1A1 influence susceptibility towards hepatocellular carcinoma. 25771868_Sulfo-conjugation of the multi-hydroxylated metabolites of benzene by human SULT1A1 may represent an important detoxifying pathway. 25819444_Data suggest that the substrate specificities of SULT1E1 and SULT1A1*1 include metabolites of tamoxifen (endoxifen, 4-hydroxytamoxifen, and N-desmethyltamoxifen); these metabolites are weak inhibitors of sulfation of estradiol by SULT1E1/SULT1A1*1. 26022216_Higher SULT1A1 levels were observed in rats as well as in humans exposed to high altitude, when compared to sea-level controls 26067475_A systematic analysis showed that three of the twelve human SULTs, SULT1A1, SULT1A3 and SULT1C4, displayed the strongest sulphating activity towards acetaminophen. 26169578_It metabolizes breast cancer drugs like afimoxifene and endoxifen by sulfation. 26340710_Sulfotransferase 1A1 Substrate Selectivity: A Molecular Clamp Mechanism. 26455829_The polymorphism of SULT1A1*2 is not associated with esophageal squamous cell carcinoma risk. 26906565_Cytosolic sulfotransferase 1A1 regulates HIV-1 minus-strand DNA elongation in primary human monocyte-derived macrophages. 27207664_We conclude from these studies that SULT1A1 is involved in the bioactivation of AA-I through the sulfonation of AL-I-NOH, contributing significantly to the toxicities of AA observed in vivo. 27221864_SULT1A1 high Copy Number Variation on high Estrogen Concentration and Tamoxifen-Associated Adverse Drug Reactions in Premenopausal Thai Breast Cancer. 27300114_SULT1A1 gene copy number affected the minor allele frequency for each single nucleotide polymorphisms tested. Before administration of exogenous hormones, increasing number of G alleles at rs9282861 was associated with earlier age at menopause, lower frequency of night sweats, and less severe insomnia. Variability in onset of menopause and symptoms before initiation of hormone therapy is partly due to SULT1A1 variation. 27356022_Epigallocatechin gallate (EGCG) displays high affinity and specificity for SULT1A1. The allosteric network is shown to involve 14 distinct complexes. ECGG binds both the allosteric site and, relatively weakly, the active site of SULT1A1. 28160022_SULT1A1 role in the metabolic detoxification of heterocyclic aromatic amines 28326452_The importance of SULT1A1 genotype for hepatic methyleugenol DNA adducts in humans, and a strong impact of SULT1A1 copy number variations on SULT1A1 hepatic phenotype. 28523759_Silencing the SULT1A1 gene led to changes in resveratrol metabolism, with higher intracellular accumulation of the nonmetabolized resveratrol. 28589969_the NQO1 Pro187Ser or SULT1A1 Arg213His polymorphism combination with smoking significantly confer susceptibility to BC. [META-ANALYSIS] 28867356_SULT1A1 copy number variation was negatively correlated with estrone-sulfate to estrone ratio predominantly in males (E1S/E1; p=0.03, r=-0.21) and may be associated with increased risk for common allergies. 28887105_No significant difference was observed in the RNA levels of CYP1A1 and SULT1A1 between the two groups. The frequency of expression of the CYP17 T/C variant tended to be higher and the A allele of COMT polymorphism together with down-regulation of its mRNA expression may be more frequent in Chinese women with idiopathic POI 29110586_This study demonstrates that the presence of His allele and Gln allele in case of SULT1A1 rs9282861 and XRCC1 rs25487, respectively, involve in lung cancer prognosis in Bangladeshi population. 29233949_SULT1A1 Arg213His (rs9282861) polymorphism might be associated with breast cancer risk, especially among Asian population. 29790428_Different ethnic and racial populations have varying degrees of SULT1A1-mediated sulfation activity. 29908303_Suggest SULT1A1/1A2 play a central role in furfuryl alcohol bioactivation and the formation of hemoglobin adducts. 30120701_Patients were categorized in three groups depending on the decreased SULT1A1 activity due to rs6839 and rs1042157: low activity group (rs6839 (GG) and rs1042157 (TT)); high activity group (rs6839 (AA) and rs1042157 (CC)); and medium activity group (all the other combinations of rs6839 and rs1042157). Associations between SULT1A1 phenotypes and clinical outcome (RFS) were explored 31671219_Prediagnostic circulating inflammation biomarkers and esophageal squamous cell carcinoma: A case-cohort study in Japan. 31835852_These findings highlight the key insights of structural consequences caused by R213H mutation, which would enrich the understanding regarding the role of SULT1A1 mutation in cancer development. 32152050_Interaction of the Brain-Selective Sulfotransferase SULT4A1 with Other Cytosolic Sulfotransferases: Effects on Protein Expression and Function. 33049293_Evaluation of a conserved tryptophanyl residue in donor substrate binding and catalysis by a phenol sulfotransferase (SULT1A1). 33297275_Investigating the molecular mechanism of hydroxylated bromdiphenyl ethers to inhibit the thyroid hormone sulfotransferase SULT1A1. 34162941_Insights into the substrate binding mechanism of SULT1A1 through molecular dynamics with excited normal modes simulations.; + ;11207031_Observational study of genotype prevalence. (HuGE Navigator) 12373301_Observational study of gene-disease association. (HuGE Navigator) 12373301_SULT1A2 polymorphisms in 230 Taiwanese breast cancer patients 12419832_Observational study of gene-disease association. (HuGE Navigator) 12469224_Observational study of gene-disease association. (HuGE Navigator) 12469224_the relationship among the polymorphisms of this enzyme and SULT1A1 in different types of cancers in Taiwanese 16133548_Observational study of genotype prevalence. (HuGE Navigator) 16133548_SULT1A1*2 and SULT1A2*2 are the major allelic variants in the Korean population 18259693_Observational study of gene-disease association. (HuGE Navigator) 18259693_SULT1A1/SULT1A2 gene complex showed suggestive haplotypic association in the family-based cardiovascular disease study, with the greatest increase in risk conferred by the SULT1A2 235T allele 18632753_Observational study of gene-disease association. (HuGE Navigator) 18992148_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 19343046_Observational study of gene-disease association. (HuGE Navigator) 19692168_Observational study of gene-disease association. (HuGE Navigator) 20214802_Observational study of gene-disease association. (HuGE Navigator) 20417180_the structures of SULT1A2 and an allozyme of SULT1A1, SULT1A1 *3, bound with 3'-phosphoadenosine 5'-phosphate at 2.4 and 2.3A resolution, respectively, were determined. 20529763_Observational study of gene-disease association. (HuGE Navigator) 20936502_Observational study of genotype prevalence. (HuGE Navigator) 20936502_the statistical hypothesis that SULT1A1 and SULT1A2 alleles are independently distributed was rejected; a strongly positive linkage was detected between SULT1A1*2 and SULT1A2*2 alleles in Turkey population 23358261_resequenced the SULT1A2 and SULT1A2 genes and identified 51 variations in ethnic Koreans, including 9 that were previously unknown. Allele frequencies, haplotype structures, LD blocks, and haplotype-tagging SNPs were determined. 23922954_SULT1A2 genotype also seems to play an important role in maintaining optimal levels of both 4OH-TAM and endoxifen. 26169578_It metabolizes breast cancer drugs like afimoxifene and endoxifen by sulfation. 26455829_The polymorphism of SULT1A2*2 is not associated with esophageal squamous cell carcinoma risk. 29908303_Suggest SULT1A1/1A2 play a central role in furfuryl alcohol bioactivation and the formation of hemoglobin adducts.; + ;12228221_M-PST is involved in protective and detoxification mechanisms that may operate in neurodegenerative processes in the brain 12424257_structure-function relationships in the stereospecific and manganese-dependent 3,4-dihydroxyphenylalanine/tyrosine-sulfating activity of human monoamine-form phenol sulfotransferase, SULT1A3 12761191_the differential substrate specificity of the two enzymes M-PST and P-PST for the thirteen drug compounds tested 14622112_Eight single nucleotide polymorphisms were observed in African American[AA] and five in Caucasian-American subjects, including one (Lys234Asn) that was observed only in AA subjects with an allele frequency of 4.2% 14871892_For SULT1A3, substrate inhibition is found for dopamine but not with pNP. Based on modeling and kinetic studies, it is proposed that substrate inhibition by dopamine in SULT1A3 is caused by binding of two dopamine molecules in the active site. 15358107_2 SULT1A3 genes were present on chromosome 16 in all DNA samples studied. 16083857_X-ray crystallographic analysis of SULT1A3 complexed with dopamine and 3'-phosphoadenosine 5'-phosphate 17002600_Of the 11 human cytosol sulfotransferase enzymes tested, only SULT1A3 displayed sulfating activity toward nitrotyrosine. The pH-dependence and kinetic constants of SULT1A3 with nitrotyrosine or dopamine as substrate were determined. 17963788_data from this study shows that the human SULT1A3 gene is inducible by glucocorticoids through a glucocorticoid receptor-mediated mechanism and the glucocorticoid response element at position (-1211 to -1193) is necessary for this induction. 19447296_Concerted action of cytosolic sulfotransferase, SULT1A3, and catechol-O-methyltransferase in the metabolism of dopamine in SK-N-MC human neuroblastoma cells. SULT1A3 is the major enzyme responsible for the sulfation of both dopamine and 3-methyldopamine. 21168432_SULT1A3 has a role in the inactivation of excess chlorotyrosine and nitrotyrosine during inflammation in lung endothelial and epithelial cells. 22352375_Studies indicate that SULT1A3 is the main isoform responsible for flavonoid sulfonation in the Caco-2 cells. 22949271_The expression of ALDOA and/or SULT1A3 is significantly higher. 24039991_Data indicate that protein-ligand interaction energy by using docking Quantitative Structure-Activity Relationships(QSAR) models showed accuracy of 67.28%, 78.00% and 75.46%, for the isoforms SULT1A1, SULT1A3 and SULT1E1, |
ENSG00000196502+ENSG00000197165+ENSG00000261052+ENSG00000213648 |
SULT1A1+SULT1A2+SULT1A3+SULT1A4 |
42.687927 |
3.3480232534 |
1.743310 |
0.45068338 |
14.141457 |
0.00016956538025177437015687809118702489286079071462154388427734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0009582679053528873371059493280199603759683668613433837890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
66.9630364521581 |
21.2604728928905 |
20.0007680304743 |
4.92743861451065 |
| ENSMUSG00000001053 |
212706 |
N4bp3 |
protein_coding |
Q8C7U1 |
FUNCTION: Plays a positive role in the antiviral innate immune signaling pathway. Mechanistically, interacts with MAVS and functions as a positive regulator to promote 'Lys-63'-linked polyubiquitination of MAVS and thus strengthens the interaction between MAVS and TRAF2 (By similarity). Also plays a role in axon and dendrite arborization during cranial nerve development. May also be important for neural crest migration and early development of other anterior structures including eye, brain and cranial cartilage (By similarity). {ECO:0000250|UniProtKB:A0A1L8GXY6, ECO:0000250|UniProtKB:O15049}. |
Cell projection;Coiled coil;Cytoplasmic vesicle;Developmental protein;Neurogenesis;Phosphoprotein;Reference proteome |
|
|
mmu:212706; |
axon [GO:0030424]; cytoplasmic vesicle [GO:0031410]; dendrite [GO:0030425]; nervous system development [GO:0007399] |
Human_homologues 29343084_N4BP3 was associated with the survival time of cervical squamous cell carcinoma patients. mir-451 and mir-486 were related to tumor differentiation stage. 35848906_N4BP3 promotes angiogenesis in hepatocellular carcinoma by binding with KAT2B. 36162713_N4BP3 promotes breast cancer metastasis via NEDD4-mediated E-cadherin ubiquitination and degradation. |
ENSG00000145911 |
N4BP3 |
66.608305 |
0.3314838294 |
-1.592990 |
0.32654217 |
23.514365 |
0.00000123984599845375424247400763383408772710936318617314100265502929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000108849188751859276809777488104202802787767723202705383300781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.4751151945283 |
7.41339813098019 |
97.9689273278425 |
15.7942514511339 |
| ENSMUSG00000001103 |
18292 |
Sebox |
protein_coding |
P70368 |
FUNCTION: Probable transcription factor involved in the control of specification of mesoderm and endoderm. {ECO:0000250}. |
Developmental protein;Differentiation;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:18292; |
nucleus [GO:0005634]; DNA binding [GO:0003677]; embryo development ending in birth or egg hatching [GO:0009792]; oogenesis [GO:0048477] |
18753614_Sebox is a new addition to maternal effect genes that produced and stored in oocytes and function in preimplantation embryo 25679966_Sebox is important in preparing oocytes for embryonic development by orchestrating the expression of other important MEGs |
ENSG00000274529 |
SEBOX |
84.854622 |
6.2488855235 |
2.643599 |
0.31220337 |
71.626470 |
0.00000000000000002600446665589200115168548298275932146439080591487306298725812325756123755127191543579101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000008303983985378343176723059583986048518644706062548366265474442116101272404193878173828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
146.929899473547 |
32.0448054764194 |
23.5129766613872 |
4.22889662992483 |
| ENSMUSG00000001211 |
28169 |
Agpat3 |
protein_coding |
Q9D517 |
FUNCTION: Converts 1-acyl-sn-glycerol-3-phosphate (lysophosphatidic acid or LPA) into 1,2-diacyl-sn-glycerol-3-phosphate (phosphatidic acid or PA) by incorporating an acyl moiety at the sn-2 position of the glycerol backbone (PubMed:15367102). Acts on LPA containing saturated or unsaturated fatty acids C16:0-C20:4 at the sn-1 position using C18:1, C20:4 or C18:2-CoA as the acyl donor (By similarity). Also acts on lysophosphatidylcholine, lysophosphatidylinositol and lysophosphatidylserine using C18:1 or C20:4-CoA (By similarity). Has a preference for arachidonoyl-CoA as a donor (PubMed:19114731). Has also a modest lysophosphatidylinositol acyltransferase (LPIAT) activity, converts lysophosphatidylinositol (LPI) into phosphatidylinositol (PubMed:19114731). {ECO:0000250|UniProtKB:Q9NRZ7, ECO:0000269|PubMed:15367102, ECO:0000269|PubMed:19114731}. |
Acyltransferase;Direct protein sequencing;Endoplasmic reticulum;Lipid biosynthesis;Lipid metabolism;Membrane;Nucleus;Phospholipid biosynthesis;Phospholipid metabolism;Reference proteome;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Phospholipid metabolism; CDP-diacylglycerol biosynthesis; CDP-diacylglycerol from sn-glycerol 3-phosphate: step 2/3. |
|
mmu:28169; |
endomembrane system [GO:0012505]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; nuclear envelope [GO:0005635]; 1-acylglycerol-3-phosphate O-acyltransferase activity [GO:0003841]; acyltransferase activity [GO:0016746]; lysophosphatidic acid acyltransferase activity [GO:0042171]; CDP-diacylglycerol biosynthetic process [GO:0016024] |
16150824_mAGPAT 1, 2, and 3 mRNA levels increased rapidly in mouse skin after acute permeability barrier disruption 21968070_The induction of LPAAT3 during germ cell development critically contributes to the accumulation of PUFAs in testicular phospholipids, thereby possibly affecting sperm cell production. 28578315_Data suggest that LPAAT3 is required for producing docosahexaenoic acid- (DHA)-containing phospholipids in various tissues, such as the testes and retina; LPAAT3-knockout mice exhibit severe male infertility with abnormal sperm morphology; lack of LPAAT3 causes drastic reduction of DHA-containing phospholipids in spermatids with excess cytoplasm around the sperm head. 28578316_Data suggest that PL-DHA (docosahexaenoic acid-containing phospholipids) contribute to visual function by maintaining disc shape in photoreceptor cells; PL-DHA disappears from outer segment in LPAAT3-knockout mice; LPAAT3-knockout mice exhibit abnormalities in retinal layers, disordered disc morphology in photoreceptor cells, and impaired visual function. 29284664_It identified lysophosphatidic acid acyltransferase (LPAAT)3 as an enzyme that was upregulated in myoblasts during in vitro differentiation and selectively incorporated DHA intophosphatidylcholine and phosphatidylethanolamine. |
ENSG00000160216 |
AGPAT3 |
429.539492 |
0.4587209647 |
-1.124311 |
0.12839996 |
76.145330 |
0.00000000000000000263537696099971092786556124365615461354359110851390025909157799333115690387785434722900390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000912319587953354395499585648214634054481443333015772134686471872555557638406753540039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
273.734070057654 |
24.3945263074146 |
596.733289078773 |
38.1301264405009 |
| ENSMUSG00000001240 |
54409 |
Ramp2 |
protein_coding |
Q9WUP0 |
FUNCTION: Transports the calcitonin gene-related peptide type 1 receptor (CALCRL) to the plasma membrane. Acts as a receptor for adrenomedullin (AM) together with CALCRL. {ECO:0000269|PubMed:10854696}. |
Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:54409; |
adrenomedullin receptor complex [GO:1903143]; amylin receptor complex 2 [GO:0150057]; cell surface [GO:0009986]; cytoplasm [GO:0005737]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; adrenomedullin binding [GO:1990409]; adrenomedullin receptor activity [GO:0001605]; amylin receptor activity [GO:0097643]; coreceptor activity [GO:0015026]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; adherens junction assembly [GO:0034333]; adrenomedullin receptor signaling pathway [GO:1990410]; amylin receptor signaling pathway [GO:0097647]; angiogenesis [GO:0001525]; basement membrane assembly [GO:0070831]; bicellular tight junction assembly [GO:0070830]; calcium ion transport [GO:0006816]; cellular response to hormone stimulus [GO:0032870]; cellular response to vascular endothelial growth factor stimulus [GO:0035924]; G protein-coupled receptor signaling pathway [GO:0007186]; heart development [GO:0007507]; intracellular protein transport [GO:0006886]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of vascular permeability [GO:0043116]; positive regulation of angiogenesis [GO:0045766]; positive regulation of gene expression [GO:0010628]; positive regulation of protein binding [GO:0032092]; positive regulation of vasculogenesis [GO:2001214]; protein localization to plasma membrane [GO:0072659]; protein transport [GO:0015031]; receptor internalization [GO:0031623]; regulation of blood pressure [GO:0008217]; regulation of G protein-coupled receptor signaling pathway [GO:0008277]; sprouting angiogenesis [GO:0002040]; vascular associated smooth muscle cell development [GO:0097084]; vasculogenesis [GO:0001570] |
11854283_role in cell surface expression of CRLR/RAMP heterodimeric receptors 15691893_expressed in osteoclast-like cells 16373602_RAMP2 plays a key role in the sensitivity and potency of AM-induced hypotensive responses via the AM1 receptor. 17470425_RAMP2 and RAMP3 have distinct physiological functions throughout embryogenesis, adulthood, and old age 18097473_RAMP2 is a key determinant of the effects of AM on the vasculature and is essential for angiogenesis and vascular integrity. 19858409_the vascular smooth muscle AM1 receptor RAMP2 acts at a local level to protect against hypertension-induced vascular hypertrophy and inflammation 21566080_this work reveals an essential role for RAMP2 in endocrine physiology and provides the first in vivo evidence for a physiological role of RAMP2 beyond that of AM/CLR signaling 21782867_Adrenomedullin-RAMP2 system is the potential target for the induction of liver sinusoidal endothelial cells. 22563471_Data show that mechanical ventilation reduced the expression of receptor activity-modifying protein RAMP3, but not of intermedin (IMD), calcitonin receptor-like receptor (CRLR), and RAMP1 and RAMP2. 23297372_These data demonstrate that the adrenomedullin-RAMP2 system is essential for cardiac metabolism and homeostasis. 23355623_The AM-RAMP2 system is a key determinant of vascular integrity and homeostasis from prenatal stages through adulthood. 24813793_It is not only an angiogenesis factor, but maintains hemostasis of blood vessels and organs. 25252154_These results indicate the AM-RAMP2 system works to protect nerve cells from both acute and chronic cerebral ischemia by maintaining cerebral blood flow suppressing oxidative stress, and in the case of chronic ischemia, enhancing capillary growth. 25264174_RAMP2 is essential for early vascular development and knockout-mice die in utero. 27307317_the Adrenomedullin-RAMP2 system regulates vascular integrity, whereas RAMP2 deletion promotes vascular permeability 27940069_the cardiovascular response of Ramp1(-/-), Ramp2(+/-), Ramp3(-/-), Ramp1(-/-)/Ramp3(-/-) double-knockout (dKO), and Calcrl(+/-) mice to AM and CGRP were compared to wildtype mice.These results suggest that the hypotensive effect of AM is primarily mediated through the CLR/RAMP1 heterodimer, but that AM signaling via CLR/RAMP2 and CLR/RAMP3 also contributes to some hypotensive action. 28324104_The AM-RAMP2 system exerts crucial vasoprotective effects after vascular injury and could be a therapeutic target for the treatment of vascular diseases. 28727763_Results provide in vivo evidence of a role for RAMP2 in placental development distinct from the RAMP2-calcitonin receptor-like receptor/adrenomedullin signaling paradigm; decreases Pthr1 expression and causes a blunted response to systemic parathyroid hormone; and identify additional pathways underlying the endocrine and fertility defects of the previously characterized Ramp2 heterozygous adult females. 30658846_Development of a Novel Model of Central Retinal Vascular Occlusion and the Therapeutic Potential of the Adrenomedullin-Receptor Activity-Modifying Protein 2 System. 33385343_Adrenomedullin-Receptor Activity-Modifying Protein 2 System Ameliorates Subretinal Fibrosis by Suppressing Epithelial-Mesenchymal Transition in Age-Related Macular Degeneration. 33545715_Adrenomedullin-RAMP2 and -RAMP3 Systems Regulate Cardiac Homeostasis during Cardiovascular Stress. 33955458_Adrenomedullin Ameliorates Pulmonary Fibrosis by Regulating TGF-ss-Smads Signaling and Myofibroblast Differentiation. |
ENSG00000131477 |
RAMP2 |
160.206848 |
2.2468138063 |
1.167881 |
0.20852212 |
30.951001 |
0.00000002646255366976858195244835564481633571887186917592771351337432861328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003005798788202200032154708976561696687213043333031237125396728515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
220.046590566728 |
31.9156222055837 |
97.9371721652517 |
10.9323262430556 |
| ENSMUSG00000001270 |
12709 |
Ckb |
protein_coding |
Q04447 |
FUNCTION: Reversibly catalyzes the transfer of phosphate between ATP and various phosphogens (e.g. creatine phosphate) (PubMed:33597756). Creatine kinase isoenzymes play a central role in energy transduction in tissues with large, fluctuating energy demands, such as skeletal muscle, heart, brain and spermatozoa (Probable). Acts as a key regulator of adaptive thermogenesis as part of the futile creatine cycle: localizes to the mitochondria of thermogenic fat cells and acts by mediating phosphorylation of creatine to initiate a futile cycle of creatine phosphorylation and dephosphorylation (PubMed:33597756). During the futile creatine cycle, creatine and N-phosphocreatine are in a futile cycle, which dissipates the high energy charge of N-phosphocreatine as heat without performing any mechanical or chemical work (PubMed:33597756). {ECO:0000269|PubMed:33597756, ECO:0000305}. |
ATP-binding;Cytoplasm;Direct protein sequencing;Kinase;Mitochondrion;Nitration;Nucleotide-binding;Phosphoprotein;Reference proteome;Transferase |
|
|
mmu:12709; |
cytosol [GO:0005829]; dendrite [GO:0030425]; extracellular space [GO:0005615]; mitochondrion [GO:0005739]; myelin sheath [GO:0043209]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; ATP binding [GO:0005524]; creatine kinase activity [GO:0004111]; kinase activity [GO:0016301]; ubiquitin protein ligase binding [GO:0031625]; cellular chloride ion homeostasis [GO:0030644]; cellular response to estrogen stimulus [GO:0071391]; cellular response to wortmannin [GO:1904568]; cerebellum development [GO:0021549]; phosphocreatine biosynthetic process [GO:0046314]; phosphorylation [GO:0016310] |
11892798_The comparison of protein patterns obtained with neural and non-neural derivatives of EC cell lines indicates changes are specific for the neural pathway, including the accumulation of the brain isozyme of creatine phosphokinase. 12526893_In rat cerebrum and cerebellum, as in rat heart, at all ages the several cCK isoenzymes do participate in a gender-specific manner, in favor of females, in diverse functions of the different cell compartments of glial and neuronal cells. 15245487_CK-B was expressed selective to astrocytes among glial populations, and was exclusive to inhibitory neurons among neuronal populations. 15639173_Brain type and mitochondrial ubiquitous CK double knockout mice have a reduced body weight, and demonstrate a severely impaired spatial learning in both a dry and a wet maze, lower nestbuilding activity and diminished acoustic startle reflex responses. 18724377_Brain-type creatine kinase has a crucial role in osteoclast-mediated bone resorption. 19333390_cytoskeletal dynamics during cell motility is coupled to on-site availability of ATP generated by CKB 19419668_Here, we report on the coupling of CK activity to body temperature rhythm and adaptive thermoregulation 21403395_reduced expression of brain creatine kinase in the cochlea of Huntington disease mice is associated with hearing impairment 23416527_negative regulation of CKB by mHTT is a key event in the pathogenesis of HD and contributes to the neuronal dysfunction associated with HD. 23891845_Hypothalamic plasticity of neuropeptide Y is lacking in brain-type creatine kinase double knockout mice with defective thermoregulation 24727412_BCK phosphorylation at Ser6 did not affect its enzymatic activity, but led to the appearance of the phosphorylated enzyme at the endoplasmic reticulum (ER) 24955845_Loss of normal CKB structure and function contributes to the mechanisms by which isoaspartate accumulation leads to central nervous system dysfunction in the PIMT-Knockout mouse. 25810257_CKB plays a key role in myotube formation by limiting myoblast fusion during myogenesis. 33547244_Inositol hexakisphosphate kinase-2 determines cellular energy dynamics by regulating creatine kinase-B. 33597756_Creatine kinase B controls futile creatine cycling in thermogenic fat. |
ENSG00000166165 |
CKB |
193.771008 |
2.0829599297 |
1.058635 |
0.18684080 |
31.978880 |
0.00000001558578853100259550172192288437778007192946461145766079425811767578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001821970306248899043764305971312977838749702641507610678672790527343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
267.00625404401 |
34.6762366748385 |
128.185977191421 |
12.8416596694756 |
| ENSMUSG00000001300 |
13642 |
Efnb2 |
protein_coding |
P52800 |
FUNCTION: Cell surface transmembrane ligand for Eph receptors, a family of receptor tyrosine kinases which are crucial for migration, repulsion and adhesion during neuronal, vascular and epithelial development. Binds promiscuously Eph receptors residing on adjacent cells, leading to contact-dependent bidirectional signaling into neighboring cells. The signaling pathway downstream of the receptor is referred to as forward signaling while the signaling pathway downstream of the ephrin ligand is referred to as reverse signaling. Binds to receptor tyrosine kinase including EPHA4, EPHA3 and EPHB4. Together with EPHB4 plays a central role in heart morphogenesis and angiogenesis through regulation of cell adhesion and cell migration. EPHB4-mediated forward signaling controls cellular repulsion and segregation from EFNB2-expressing cells. May play a role in constraining the orientation of longitudinally projecting axons. {ECO:0000269|PubMed:10704386}. |
3D-structure;Angiogenesis;Cell junction;Cell membrane;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Membrane;Methylation;Neurogenesis;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:13642; |
adherens junction [GO:0005912]; dendrite [GO:0030425]; glutamatergic synapse [GO:0098978]; plasma membrane [GO:0005886]; postsynaptic density membrane [GO:0098839]; presynaptic membrane [GO:0042734]; Schaffer collateral - CA1 synapse [GO:0098685]; ephrin receptor binding [GO:0046875]; signaling receptor binding [GO:0005102]; adherens junction organization [GO:0034332]; animal organ morphogenesis [GO:0009887]; axon guidance [GO:0007411]; blood vessel morphogenesis [GO:0048514]; cell adhesion [GO:0007155]; cell migration involved in sprouting angiogenesis [GO:0002042]; cellular response to lipopolysaccharide [GO:0071222]; ephrin receptor signaling pathway [GO:0048013]; keratinocyte proliferation [GO:0043616]; lymph vessel development [GO:0001945]; negative regulation of keratinocyte proliferation [GO:0010839]; negative regulation of neuron projection development [GO:0010977]; nephric duct morphogenesis [GO:0072178]; positive regulation of aorta morphogenesis [GO:1903849]; positive regulation of cardiac muscle cell differentiation [GO:2000727]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of neuron death [GO:1901216]; presynapse assembly [GO:0099054]; regulation of chemotaxis [GO:0050920]; regulation of postsynaptic membrane neurotransmitter receptor levels [GO:0099072]; regulation of postsynaptic neurotransmitter receptor internalization [GO:0099149]; T cell costimulation [GO:0031295]; venous blood vessel morphogenesis [GO:0048845] |
11780069_crystal structure of the complex formed between EphB2 and ephrin-B2, determined at 2.7 A resolution 11880349_a vascular-specific deletion of ephrinB2 results in angiogenic remodeling defects 12149214_EphB/ephrin-B2 signaling between endothelial cells and surrounding mesenchymal cells plays an essential role in vasculogenesis, angiogenesis, and vessel maturation 12217323_in Delta-1 mutant mice neural crest cells moved through both rostral and caudal halves of the somites in mutants, consistent with the finding that ephrinB2 levels are significantly reduced in the caudal-half somites. 12588758_Ephrin-B2 forward signaling differentially affects cell adhesion and migration between arterial and venous endothelial cells. 12684176_Organ of Corti and spiral ganglion showed strong expression of ephrin-A3, ephrin-B2 and ephrin-B3. In lateral wall, ephrin-A3 and ephrin-B2 were strongly expressed. 12816988_Solid-phase EFNB2 costimulates T cells in the presence of suboptimal T cell receptor ligation; such costimulation leads to enhanced T cell proliferation and IFN-gamma production. 12971893_Ephrin-B2 is expressed at the mouse chiasm midline as the ipsilateral projection is generated and is selectively inhibitory to axons from ventrotemporal (VT) retina, where ipsilaterally projecting retinal ganglion cells reside. 15223333_ephrin-B2 functions in the early embryo as an instructive ligand to stimulate EphB4 receptor forward signaling during angiogenic remodeling, and later as a receptor to transduce reverse signals involved in cardiac valve maturation and axon pathfinding. 15223334_EphB2 and ephrin-B2 are coexpressed at the midline in the fusing urethral/cloacal endoderm and underlying lateral mesoderm of the urorectal septum that migrates toward the caudal midline as the cloaca septates. 15687262_Essential regulator of lymphatic development, interactions with PDZ domain effectors are required. 15851594_ephrin-B2 is exclusively expressed and significantly upregulated in arterial vasculature after the initial angiogenic responses in tissue ischemia; an ephrin-B2-rich environment was shown to induce neovascularization mainly through venous angiogenesis 16211213_ephrin-B2 can act as a major modulator of cell migration and matrix interactions of melanoma cells, which possibly contributes to the expansion and metastatic spread of melanoma in vivo 16430858_Dll4 stimulation but not Jagged1 markedly induced ephrinB2 expression 16890539_Here it is shown that osteoclasts express the NFATc1 target gene Efnb2 (encoding ephrinB2), while osteoblasts express the receptor EphB4, along with other ephrin-Eph family members. 16951162_DDL4/Notch4 and ephrin B2 pathways downstream of VEGF plays a key role in the abnormal remodeling of tumor vessels. 17158005_cytoplasmic domain mutations that interfere with EphB2 forward signaling or ephrin-B2 reverse signaling exhibit a hyperactive circling (waltzing) locomotion associated with a decreased amount of endolymph fluid that normally fills the vestibular labyrinth 17223098_Ephrin-B2 forward signaling regulates somite patterning and neural crest cell development. 17945206_ephrin-B2 is expressed in areas with high levels of synaptic plasticity, such as the cerebral cortex, hippocampus and cerebellum. However, at the cellular level, ephrin-B2 was localized to neuronal cell bodies. 17948875_These differences in cell number and distribution suggest that EphA4 and ephrin-B2 signaling influence auditory activation patterns. 17967521_Characterizes neural activation in EphA4/ephrin-B2 protein deficient mice in the auditory brainstem response. 17972914_EphrinB2 overexpression results in increased NR2B tyrosine phosphorylation, which is essential for caMEK1-induced epilepsy in vivo, since conditional inactivation of ephrinB2 greatly reduces seizure frequency in caMEK1 transgenic mice 18445690_expression of ephrinB2 in adult endothelial cells is up-regulated during arterial remodeling and controlled by cyclic stretch 18463357_Patterns of endometrial EFNB2/EPHB4 expression during the period of spiral arterial modification. 18571496_These results implicate that EphB4/ephrinB2 is an important signaling pathway in ischemic heart disease and its modulation may induce therapeutic angiogenesis. 18627264_The data raise the possibility that PTH or PTHrP might regulate ephrinB2 to act in a paracrine or autocrine manner on EphB4 or EphB2 in the osteoblast, contributing as a local event to the anabolic action of PTH or PTHrP. 18651661_lung alveolar formation relies on endothelial ephrinB2 function 18713744_MMP7 and MMP9-mediated cleavage of EphB2 is induced by receptor-ligand interactions at the cell surface and that this event triggers cell-repulsive responses 18950979_Ex vivo gene delivery of ephrin-B2 induces development of functional collateral vessels in a rabbit model of hind limb ischemia. 19032981_Strong evidence is provided for specific and co-operative roles of EphB2 and EphB3 in palate development. 19113915_beta-arrestin2 inhibits osteoclastogenesis in vitro, which resulted in decreased bone resorption in vivo by regulating RANKL/OPG production and ephrins mRNAs. 19218547_ephrinB2 plays a crucial role in nonproductive angiogenesis caused by DLL4 blockade for tumors 19268698_Study reports cloning and characterization of promoter of ephrin-B2 and its regulatory elements; the 180 bp region located upstream of the transcription start site has a high GC content and contains a TATA-box vital for basal transcriptional activity. 19411631_A role for EphrinB signaling in orchestrating pericyte/endothelial cell assembly, suggest that therapeutic targeting of EphrinB may prove useful for disrupting angiogenesis when it contributes to disease. 19571816_The transient expression of ephrin B2 in perinatal dermal mesenchymal cells plays a role in adjusting the activity of the mesenchymal microenvironment that regulates proliferation of keratinocytes. 19639173_mutated ephrin-B2 expression significantly accelerated mammary tumour growth and imposed a metastatic phenotype 19797707_When ES cell-derived VEGFR2(+) cells were exposed to shear stress, expression of the arterial EC marker protein ephrinB2 increased dose-dependently. 19843150_Abolishing ephrinB2 function led to severe interference with the architecture and functioning of the mammary gland at lactation. 19883626_These results indicate that hypoxia drives arterial differentiation by increasing ephrinB2 expression in endothelial cells through Sp1 activation. 20101599_Results suggest that formation of arteriovenous malformations in Notch1 gain of function mutants and ephrinB/EphB pathway loss of function mutant embryos occurs by different mechanisms. 20445537_results show that full VEGFR3 signalling is coupled to receptor internalization; ephrin-B2 is a key regulator of this process and thereby controls angiogenic and lymphangiogenic growth 20445540_ephrin-B2 reverse signalling involving PDZ interactions regulates endothelial tip cell guidance to control angiogenic sprouting and branching in physiological and pathological angiogenesis 20504947_These data suggest the relevance of a nonactivated EphB2 for regulating T cell progenitor migration and its modulation upon ephrin-B engagement. 20616004_Result implies a NC-derived cell-specific role of EphB-ephrin-B2 interactions in the collective migration of the thymic rudiment during organogenesis. 20629048_demonstrate that bath application of ephrin-B2 induces rapid and sustained growth cone collapse and axon retraction in ventrotemporal retinal ganglion cell axons 20649938_Data show that ephrinB4 can promote the growth of melanomas expressing the ephrin-B2 ligand by stimulating proliferation, survival and angiogenesis. 20660266_EphB2-ephrin-B2 reverse signaling is required to prevent the formation of ipsilateral VCN-MNTB projections and that this signaling operates non-cell autonomously. 20886601_These findings suggest the involvement of cell-cell EphA4 and ephrin-B2 signaling in establishing order in the developing inferior colliculus 21059214_Presynaptic ephrin-B2 expression plays an important role in regulating inflammatory pain through the regulation of synaptic plasticity in the dorsal horn and is also involved in the pathogenesis of some types of neuropathic pain. 21127290_There may be a link between vascular ephrinB2 expression and the proinflammatory activation of monocytes that may contribute to the pathogenesis of arteriosclerosis. 21539827_Both EphB2/EphB3 forward signaling and ephrin-B2 reverse signaling were shown to be required for midline fusion of the palate. 21847105_Dorsal and ventral-temporal axons require ephrin-B2 reverse signalling. 21976681_Efnb1 and Efnb2 proteins regulate thymocyte development, peripheral T cell differentiation, and antiviral immune responses and are essential for interleukin-6 (IL-6) signaling. 22069310_a novel function of Efnb1 and Efnb2 in stabilizing IL-7Ralpha expression at the post-translational level 22101302_The results of this study suggested that rapid accumulation of EphrinB2 in hippocampal CA1 neurons is involved in the behavioural and cellular modifications induced by contextual fear conditioning. 22622516_in an in vitro model of HHT2, loss of Alk1 blocks BMP9 signaling, resulting in reduced EphrinB2 expression, enhanced VEGFR2 expression, and misregulated EC sprouting and anastomosis 22673212_results suggest the involvement of EFNB2 in thymocyte development; however, heavy redundancy among Eph/EFN family members prevents the occurrence of detrimental phenotypes in the T cell compartment caused by T cell-specific EFNB2 gene null mutation 22983209_Ephrin-B2(+) astrocytes therefore promote neuronal differentiation of adult NSCs through juxtacrine signaling. 23042409_These findings suggest the importance of these ephrin-b2 in establishing functional auditory circuits prior to experience. 23106883_ephrin-B2 could be one of the promoters to upregulate gliosis following brain injury. 23129351_a role for ephrin-B2 in the development and healing of bone through activation of osteoblast-specific gene expression. 23146940_EphB2 and EphB3 are involved in the control of thymic epithelial cells (TEC) survival and that the absence of these molecules causes increased apoptotic TEC. 23165727_These data indicate that ephrinB2/EphB4 signaling within the osteoblast lineage is required for late stages of osteoblast differentiation. 23492730_PDZ domain-dependent ephrinB2 reverse signaling protects against PTC rarefaction by regulating angiogenesis and vascular stability during kidney injury 23518227_B2 expressed on astrocytes inhibited axonal growth 23623938_We confirmed that ephrin-B2 protein causes co-clustering of EphB2 and glutamate receptors in spinal cord neurons. 23742148_Ephrin-B2 reverse signaling mediates distal alveolar formation. 24298057_ephrin-B2 is an important regulator of PDGFRbeta endocytosis and thereby acts as a molecular switch controlling the downstream signaling activity of this receptor 24477991_These results suggest that ephrin-B2 forward signaling through EphA4 is required for the precise control of cortical neuron migration. 24583262_Data indicate that conditional inactivation of Efnb2 in early-stage embryonic ear tissues disrupted cell proliferation, cell survival, and epithelial folding at the origin of the endolymphatic epithelium. 24673722_Efnb2 conditional knockout mice were defective in acute arterial dilation. Vasodilation was impaired in cremaster arterioles in response to either increased flow or ACh, and in the carotid arteries in response to increased flow. 25283993_cell-cell interactions with endothelial cells enforce quiescence and promote stem cell identity. Mechanistically, endothelial ephrinB2 and Jagged1 mediate these effects 25299585_Efnb2 expression was attenuated in Pou3f4 hemizygous null mutants relative to control. 25417117_Conditioned deletion of ephrinB1 and/or ephrinB2 in either thymocytes or thymic epithelial cells alters the organization of thymic medulla and favors the appearance of thymic epithelial cysts. 25480420_ephrin-B2 signaling has an important role in the formation of pituitary stem/progenitor cell niches and in pituitary organogenesis. 25779027_Efnb1 and Efnb2 in T cells are essential for pathogenic antibody production and for T cell migration to the inflamed paws in mice with collagen-induced arthritis. 25807892_EfnB2 is an essential regulator of endothelial cell death and vessel pruning. This regulation depends upon phosphotyrosine-EfnB2 signaling repressing JNK3 activity via STAT1. 25865237_Mice bearing genetically altered cytoplasmic region of ephrinB2 have significantly altered EphB4-dependent forward signaling. 25923646_disruption of cochlear signalling results in supporting cell translocation into hair cell layers 26385750_Neural crest defects in ephrin-B2 mutant mice are non-autonomous and originate from defects in the vasculature. 26503288_our study uncovered a novel cell autonomous role for ephrinB2 in lateral motor column motoneurons 26633881_The Netrin-1 and ephrin-B2 synergistic growth cone responses involve the potentiation of Src family kinase signaling, a common effector of both pathways. 26755702_the role of ephrin B2 in endochondral ossification using Osx1Cre-targeted gene deletion, was investigated. 26980243_in vivo study demonstrated, for the first time, that EFNB2 is essential for normal long bone growth and development 27039370_that expression of EFNB1 and EFNB2 is implicated in Th cell differentiation and migration to inflammatory sites in both EAE and MS 27060907_The Eph/ephrin-B2-mediated heterotypic or homotypic cell interactions between thymocytes and thymic epithelial cells (TECs), or between TECs and themselves, contribute to the early maturation of MTS20(+) TECs. 27250641_Results show that EfnB2 plays a previously unrecognized role as a principal regulator of hematopoiesis from the dorsal aorta and embryonic stem cells by controlling the emergence of hemogenic endothelial cells. During vascular development, EfnB2 is essential for the proper sorting of arterial and venous-fated endothelium into distinct arterial and venous vascular beds. 27344203_During urogenital development, different modes of Eph/Ephrin signaling occur at several sites with EphrinB2 and EphrinA5 acting in concert. 27530629_Male Efnb2 knockout mice presented reduced blood pressure. Region from aa 313 to aa 331 of EFNB2 was essential for reverse signaling regulating VSMC contractility. 27551053_Control of abscission requires Eph kinase activity, and Src and citron kinase (CitK) are downstream effectors in the Eph-induced signal transduction cascade 28254815_Suggest role for ephrinB2/EphB4 signaling in the maintenance of neurovascular homeostasis and show that EphrinB2 activation promotes vascular repair mechanisms and reduces brain swelling after mild cerebral ischemia. 28595615_Data reveal that ephrinB2 and ephrinB3 signaling is required to control progenitor identities in the ventral spinal cord. 28732675_As endothelial progenitor cells (EPCs)derived from mouse bone marrow were cultured on substrates of increasing stiffness, the mRNA and protein levels of the specific arterial endothelial cell marker ephrinB2 were found to increase, while the expression of the venous marker EphB4 decreased 28947178_an autonomous receptor-like role for ephrin-B reverse signaling in the tangential migration of interneurons into the neocortex using ephrin-B (EfnB1/B2/B3) conditional triple mutant (TM(lz)) mice, is reported. 28978486_GRIP1 binds to apoER2 and EphrinB2 to induce activity-dependent AMPA receptor insertion at the synapse. 29444449_Results show that early but not late post conditioning hippocampal protein synthesis is necessary for the formation of remote memory and provide the first evidence of a possible involvement of EphrinB2 in neuronal plasticity in the anterior cingulate cortex. 30015911_Production of inflammatory cytokines was inhibited by Ti particles through bidirectional signals. Addition of ephB4Fc inhibited the osteoclastmediated formation of Ti particles via bidirectional ephrinB2/ephB4 signaling. Activation of this bidirectional signaling pathway may be a potential clinical treatment for osteolysis surrounding prostheses. 30075851_Spatiomechanical Modulation of EphB4-Ephrin-B2 Signaling in Neural Stem Cell Differentiation 31366886_secondary mineralization involves EphrinB2-RhoA-limited autophagy in osteocytes, and disruption leads to a bone fragility independent of bone mass. 31782728_we have used mouse genetics, imaging and cell biology approaches to investigate how homeostasis in the adult heart is controlled by endothelial EphB4 and its ligand ephrin-B2, which are known regulators of vascular morphogenesis and arteriovenous differentiation during development. 31830612_A novel long non-coding RNA, Leat1, causes reduced anogenital distance and fertility in female mice. 31868584_EphrinB2 regulates VEGFR2 during dendritogenesis and hippocampal circuitry development. 32585189_Increased Ephrin-B2 expression in pericytes contributes to retinal vascular death in rodents. 32897857_EphrinB2-EphB4 signalling provides Rho-mediated homeostatic control of lymphatic endothelial cell junction integrity. 33039109_Activation of EphrinB2 Signaling Promotes Adaptive Venous Remodeling in Murine Arteriovenous Fistulae. 33789115_EphrinB2 and GRIP1 stabilize mushroom spines during denervation-induced homeostatic plasticity. 34281570_RNA-seq co-expression network analysis reveals anxiolytic behavior of mice with Efnb2 knockout in parvalbumin+ neurons. 35725568_EphB4 and ephrinB2 act in opposition in the head and neck tumor microenvironment. 36029930_Histone deacetylase 4 inhibition ameliorates the social deficits induced by Ephrin-B2 mutation. 36376513_Adaptive activation of EFNB2/EPHB4 axis promotes post-metastatic growth of colorectal cancer liver metastases by LDLR-mediated cholesterol uptake. |
ENSG00000125266 |
EFNB2 |
73.988866 |
4.1412720076 |
2.050074 |
0.39054751 |
26.080864 |
0.00000032741295908411416198732367573154267148538565379567444324493408203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000031484559297785521530675841017643534769376856274902820587158203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
118.357453517727 |
32.4329457730608 |
28.5799757421876 |
6.10975363958943 |
| ENSMUSG00000001333 |
68828 |
Sync |
protein_coding |
Q9EPM5 |
FUNCTION: Atypical type III intermediate filament (IF) protein that may play a supportive role in the efficient coupling of mechanical stress between the myofibril and fiber exterior. May facilitate lateral force transmission during skeletal muscle contraction. Does not form homofilaments nor heterofilaments with other IF proteins. {ECO:0000269|PubMed:11694502, ECO:0000269|PubMed:18367591}. |
Alternative splicing;Coiled coil;Cytoplasm;Intermediate filament;Phosphoprotein;Reference proteome |
|
|
mmu:68828; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; intermediate filament [GO:0005882]; neuromuscular junction [GO:0031594]; perinuclear region of cytoplasm [GO:0048471]; sarcolemma [GO:0042383]; Z disc [GO:0030018]; intermediate filament-based process [GO:0045103] |
17629480_The data show that absence of alpha-dystrobrevin or desmin leads to dynamic changes in syncoilin that may compensate for, or participate in, different muscle myopathies. 18367591_Syncoilin may play a supportive role for desmin in the efficient coupling of mechanical stress between the myofibril and muscle fiber exterior. 18594912_A second viable syncoilin knockout model and highlights the importance of further investigations to determine the role of syncoilin in skeletal muscle. 19070665_Despite lacking an apparent structural role in striated muscle, the three syncoilin isoforms are differentially and strongly upregulated during muscle regeneration and atrophy in C57BL/6 mice, possibly suggesting an atypical role for syncoilin in muscle. 20587592_A neuronal analysis of the syncoilin-null mouse (Sync(-/-)) revealed a reduced ability in accelerating treadmill and rotarod tests. 24043511_in the liver, syncoilin is predominantly expressed by activated Hepatic stellate cells and displays very low-expression levels in other liver cell types, making it a good marker of activated Hepatic stellate cells. 34052184_Single-cell brain atlas of Parkinson's disease mouse model. |
ENSG00000162520 |
SYNC |
28.692529 |
0.4109058086 |
-1.283120 |
0.46717717 |
7.514761 |
0.00611954074448318053464079824266264040488749742507934570312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0220778883514845571112150679482510895468294620513916015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.205449970728 |
5.3398850458212 |
39.4383569903563 |
9.16923845400597 |
| ENSMUSG00000001349 |
12797 |
Cnn1 |
protein_coding |
Q08091 |
FUNCTION: Thin filament-associated protein that is implicated in the regulation and modulation of smooth muscle contraction. It is capable of binding to actin, calmodulin and tropomyosin. The interaction of calponin with actin inhibits the actomyosin Mg-ATPase activity. |
Actin-binding;Alternative splicing;Calmodulin-binding;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:12797; |
cytoskeleton [GO:0005856]; actin binding [GO:0003779]; calmodulin binding [GO:0005516]; actomyosin structure organization [GO:0031032]; negative regulation of vascular associated smooth muscle cell proliferation [GO:1904706] |
12576725_Mutated mice showed increased locomotor activity, as well as increased intake of food and water, associated with the decreased number of neurons in the paraventricular nucleus of the hypothalamus (PVN) 12716472_CNh1 plays a role as tumor suppressor in SR-3Y1 mainly by decreasing VEGF expression and angiogenesis in vivo and partially through reducing cellular proliferative potential and cell motility 16973711_Up-regulation of h1-calponin in SM-B null mice may be necessary to maintain reduced level of cross-bridge cycling over time in absence of SM-B myosin and play important role in regulating smooth muscle contraction. 21463585_The structure of this functionally important region of calponin and its interaction with actin and calmodulin at amino-acid resolution, was mapped. 21817093_A single intronic CArG element is necessary but insufficient for proper CNN1 expression in vivo. CNN1 overexpression antagonizes arterial injury-induced neointimal formation. 22551221_data suggest that in mice myometrium, phosphorylation of h1 calponin induced by the PKC-epsilon might facilitate the contraction of uterine in labor and regulate pregnant myometrial contractility. 23044709_These findings reveal a novel function of CNN1 in maintaining bone homeostasis by coupling bone formation to bone resorption 28457943_Actin cytoskeleton depolymerization inhibites BMP2 signaling via blocking of Smad by dephosphorylated CNN1 in osteoblast cells under simulated microgravity. 30707993_Cnn1(-/-) and Cnn2(-/-) double knockout aortic smooth muscle exhibits faster relaxation than that of wild type control. |
ENSG00000130176 |
CNN1 |
63.392468 |
96.6400896074 |
6.594550 |
0.74278163 |
129.190781 |
0.00000000000000000000000000000616032619289975762215507823701303140670306518145046458794058346049921326541027200712763445267228235024958848953247070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000004645251909418272070004020619118139432061391215736081066246250539457115331324482676578213613538537174463272094726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
125.996638597007 |
67.7744193440543 |
1.30377195539486 |
0.66434376727921 |
| ENSMUSG00000001403 |
68612 |
Ube2c |
protein_coding |
Q9D1C1 |
FUNCTION: Accepts ubiquitin from the E1 complex and catalyzes its covalent attachment to other proteins. In vitro catalyzes 'Lys-11'- and 'Lys-48'-linked polyubiquitination. Acts as an essential factor of the anaphase promoting complex/cyclosome (APC/C), a cell cycle-regulated ubiquitin ligase that controls progression through mitosis. Acts by initiating 'Lys-11'-linked polyubiquitin chains on APC/C substrates, leading to the degradation of APC/C substrates by the proteasome and promoting mitotic exit. {ECO:0000250|UniProtKB:O00762}. |
Acetylation;ATP-binding;Cell cycle;Cell division;Mitosis;Nucleotide-binding;Phosphoprotein;Reference proteome;Transferase;Ubl conjugation;Ubl conjugation pathway |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000255|PROSITE-ProRule:PRU00388}. |
|
mmu:68612; |
anaphase-promoting complex [GO:0005680]; cytosol [GO:0005829]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; ubiquitin ligase complex [GO:0000151]; ATP binding [GO:0005524]; ubiquitin conjugating enzyme activity [GO:0061631]; ubiquitin-like protein ligase binding [GO:0044389]; ubiquitin-protein transferase activity [GO:0004842]; anaphase-promoting complex-dependent catabolic process [GO:0031145]; cell division [GO:0051301]; exit from mitosis [GO:0010458]; free ubiquitin chain polymerization [GO:0010994]; positive regulation of exit from mitosis [GO:0031536]; protein K11-linked ubiquitination [GO:0070979]; protein K48-linked ubiquitination [GO:0070936]; protein polyubiquitination [GO:0000209]; protein ubiquitination [GO:0016567]; regulation of mitotic metaphase/anaphase transition [GO:0030071]; ubiquitin-dependent protein catabolic process [GO:0006511] |
20065091_results identify UbcH10 as a prominent protooncogene that causes whole chromosome instability and tumor formation over a wide gradient of overexpression levels 25376843_UbcH10 overexpression increases carcinogenesis and blocks ALLN susceptibility in colorectal cancer. 26207029_alongside their canonical function in protein degradation, Ube2C and -S also control the extrusion of the first polar body. 35671859_Ube2c-inhibition alleviated amyloid pathology and memory deficits in APP/PS1 mice model of AD. |
ENSG00000175063 |
UBE2C |
1076.802113 |
2.3104251257 |
1.208158 |
0.08963192 |
179.830419 |
0.00000000000000000000000000000000000000005277725805942378812511367337197741525792210034349113294753917959400226274018484827481384794936128683658094207231670225155539810657501220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000062318077113266908008949822264198215273289470818182728923412937960169222129629650848769527492653408275863347043355133791919797658920288085937500000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1519.47058388762 |
94.4625815011744 |
657.658439998034 |
31.5637333293546 |
| ENSMUSG00000001435 |
12822 |
Col18a1 |
protein_coding |
P39061 |
FUNCTION: Probably plays a major role in determining the retinal structure as well as in the closure of the neural tube. {ECO:0000250|UniProtKB:P39060}.; FUNCTION: [Non-collagenous domain 1]: May regulate extracellular matrix-dependent motility and morphogenesis of endothelial and non-endothelial cells; the function requires homotrimerization and implicates MAPK signaling. {ECO:0000250|UniProtKB:P39060}.; FUNCTION: [Endostatin]: Potently inhibits endothelial cell proliferation and angiogenesis. May inhibit angiogenesis by binding to the heparan sulfate proteoglycans involved in growth factor signaling (PubMed:9008168). Inhibits VEGFA isoform VEGF165-induced endothelial cell proliferation and migration. Seems to inhibit VEGFA-mediated signaling by blocking the interaction of VEGFA to its receptor KDR/VEGFR2 (PubMed:12029087). Modulates endothelial cell migration in an integrin-dependent manner implicating integrin ITGA5:ITGB1 and to a lesser extent ITGAV:ITGB3 and ITGAV:ITGB5 (PubMed:11158588). May negatively regulate the activity of homotrimeric non-collagenous domain 1 (By similarity). {ECO:0000250|UniProtKB:P39060, ECO:0000269|PubMed:11158588, ECO:0000269|PubMed:9008168}. |
3D-structure;Alternative promoter usage;Alternative splicing;Basement membrane;Cell adhesion;Collagen;Direct protein sequencing;Disulfide bond;Extracellular matrix;Glycoprotein;Hydroxylation;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal;Zinc |
|
|
mmu:12822; |
basement membrane [GO:0005604]; collagen trimer [GO:0005581]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; extracellular matrix structural constituent [GO:0005201]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; angiogenesis [GO:0001525]; blood vessel endothelial cell migration [GO:0043534]; cell adhesion [GO:0007155]; endothelial cell apoptotic process [GO:0072577]; endothelial cell morphogenesis [GO:0001886]; extracellular matrix organization [GO:0030198]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of endothelial cell apoptotic process [GO:2000353]; positive regulation of vascular endothelial cell proliferation [GO:1905564]; vascular endothelial cell proliferation [GO:0101023] |
11927538_Lack of collagen XVIII/endostatin results in eye abnormalities. 11991745_Endostatin gene transfer inhibits joint angiogenesis and pannus formation in inflammatory arthritis 12673115_Endostatin expression effectively inhibits mammary neoplasms growth in nude mice 14525950_Col18a1 knockout mice have iris and ciliary body defects. 14614990_collagens XV and XVIII have distinct roles as revealed by double knockout mice 14685281_Col18a1 is essential for vision and retinal pigment epithelial function. 14973128_there is a putative integrin-binding sequence with anti-migratory activity within endostatin 15296943_Collagen XVIII and enamelysin were co-localized in the developing enamel matrix and stratum intermedium and in the enamel-like tumor matrix of odontogenic tumors. 15313955_Studies in knockout mice identify a previously unrecognized function for collagen XVIII in vascular permeability. Loss of collagen XVIII enhances angiogenesis & vascular permeability in atherosclerosis by distinct gene-dose-dependent mechanisms. 15614762_collagen XVIII's C-terminal endostatin domain harbors a prominent tissue-binding site, which can bind in the absence of heparan sulfates in situ 15632014_In knockout mice, a widening of the epidermal basement membrane (BM) zone of the skin was observed in electron microscopy. 15694132_angiostatic function is heparan sulfate-dependent, and in situ-binding of endostatin to endothelial cells is increased by heparan sulfates 15714516_The p.D104N allele was found in trans with a pathogenic frameshift mutation in a healthy carrier of Knobloch syndrome, showing it is not very harmful. It may be a modifier or at-risk allele. 16170784_Collagen XVIII is localized in various basement membranes and is highly expressed throughout the connective tissue core of the endocardial cushions and embryonic atrioventricular valve leaflets; it may have a role in cardiac valve morphogenesis. 16465514_Exposure to hypoxia resulted in organ-specific, long-term increase of 24 & 26 kDa endostatin fragments in mouse aorta & lung. 16574906_Proteolytic matrix fragment endostatin(ES) is prominent vasorelaxing agent. Because ES is constantly released into the blood, it is novel regulator of blood pressure and represents an interesting pharmacological target. 17525255_Data show that upon experimental and human renal ischemia/reperfusion, microvascular basement membrane (BM) heparan sulfate proteoglycans (collagen 18alpha1, agrin and perlecan) are modified to bind L-selectin and monocyte chemoattractant protein-1. 17574579_following resection of cirrhotic liver, regeneration capacity is depressed and high endostatin levels are independent of hepatic regeneration 17727259_Results show that endogenous endostatin and its related peptides are widely distributed in all in vivo tumor types tested, but not in most of the cultured tumor cell lines. 17803469_Local synthesis of a 30 kDa endostatin-related fragment following acute renal failure. Role in the modulation of renal capillary density. 18006826_Endostatin binding to ovarian cancer cells inhibits peritoneal attachment and dissemination 18054814_D104N polymorphism of the COL18A gene may be an important inherited determinant of the LC susceptibility. 18089781_Inhibition of tumor angiogenesis by endostatin at an early stage in skin tumor development and an inhibition of lymph node metastasis. 18455382_study shows that a lack of collagen XVIII accelerates wound vascularisation and the rate of cutaneous wound healing whereas an excess of endostatin leads to delayed wound healing 18840438_These data indicate that collagen XVIII may play an important role in corneal reinnervation after wounding. 19513570_Report effects of endostatin production on oncogenicity and metastatic activity of HPV16-transformed mouse cells: role of interleukin 1alpha. 19564642_Women with early-onset, but not late-onset preeclampsia have higher levels of endostatin than healthy pregnant women in corresponding lengths of gestation. 19763371_In vivo, Collagen XVIII/endostatin does not play an indispensable role in skeletal development 20110665_the local synthesis of a 28-kDa ES-related fragment following acute kidney injury and suggest its role in endothelium survival 21085708_Data show that Col18-deficiency causes hypertriglyceridemia in mice. 21193414_Lack of collagen XVIII long isoforms affects kidney podocytes, whereas the short form is needed in the proximal tubular basement membrane 22733742_Endostatin lowers blood pressure via nitric oxide and prevents hypertension associated with VEGF inhibition 23463096_Data indicate that delivery by attenuated Salmonella of the combination therapeutic plasmid simultaneously knocked down the expression of Stat3 and resulted in over-expression of Endostatin, which synergistically inhibited prostate cancer growth. 23580202_Type XVIII collagen is an important functional component of the liver matrix microenvironment and is crucial for hepatocyte survival during injury and stress. 23720153_The results of the present study indicate endostatin promotes soft callus formation but inhibits callus remodeling during fracture healing most probably by an inhibition of angiogenesis. 24135756_An excess of the collagen XVIII Tsp-1 domain is deleterious in the eye, possibly by impairing certain functions of the full-length molecule. 24727495_There are correlations between the elevation of endostatin levels and parallel microvascular rarefaction and induction of renal fibrosis in aging mice. 25024173_Specific collagen XVIII isoforms promote adipose tissue accrual via mechanisms determining 25188209_Basement membrane zone collagens XV and XVIII mediate leukocyte influx in renal ischemia-reperfusion. 27125427_This study examined the staining levels of proteasomal and autophagy markers in the RPE of different ages of the Col18a1 (-/-) mice. Eyes from 3, 6-7, 10-13 and 18 months old mice were enucleated and embedded in paraffin according to the routine protocol. 27165830_Kidney fibrosis in aging may represent a natural outcome of upregulated endostatin (EST) and transglutaminase 2 (TG2), but more likely it appears to be a result of cumulative stresses occurring on the background of synergistically acting geronic (aging) proteins, EST and TG2. 31015748_Collagen XVIII plays a role in the formation of the dermal-epidermal junction during re-epithelialization. 32449518_Lack of collagen XVIII leads to lipodystrophy and perturbs hepatic glucose and lipid homeostasis. |
ENSG00000182871 |
COL18A1 |
822.034387 |
2.2331914524 |
1.159107 |
0.12323242 |
86.763921 |
0.00000000000000000001222800324615900536925609814810452168522098857176585545410499311635987851332174614071846008300781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000004888633738726875506154376965304096225234514054511236486738612327940245450008660554885864257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1131.16531081828 |
96.7396523746564 |
506.524109066178 |
33.0322135045113 |
| ENSMUSG00000001494 |
74499 |
Sost |
protein_coding |
Q99P68 |
FUNCTION: Negative regulator of bone growth that acts through inhibition of Wnt signaling and bone formation. {ECO:0000250, ECO:0000269|PubMed:19419300}. |
3D-structure;Disulfide bond;Extracellular matrix;Glycoprotein;Heparin-binding;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:74499; |
extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; protein-containing complex [GO:0032991]; BMP binding [GO:0036122]; carbohydrate binding [GO:0030246]; DNA-binding transcription factor binding [GO:0140297]; heparin binding [GO:0008201]; molecular function inhibitor activity [GO:0140678]; BMP signaling pathway [GO:0030509]; canonical Wnt signaling pathway [GO:0060070]; cellular response to parathyroid hormone stimulus [GO:0071374]; localization [GO:0051179]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of ossification [GO:0030279]; negative regulation of protein-containing complex assembly [GO:0031333]; negative regulation of Wnt signaling pathway [GO:0030178]; ossification [GO:0001503]; positive regulation of DNA-templated transcription [GO:0045893]; response to mechanical stimulus [GO:0009612] |
12702725_effects of sclerostin on the activity of BMP2, BMP4, BMP6, and BMP7 and role in bone remodeling 15199066_noggin and sclerostin form a mutually inhibitory complex that fine-tunes of BMP activity in bone homeostasis 15217980_Expression and regulation of sclerostin under the control of bone morphogenetic protein were closely associated with those of osterix in vivo and in vitro. (Bone morphogenetic protein 2) 15545262_Sclerostin inhibits Wnt-3a-induced C3H10T1/2 cell differentiation indirectly in a path mediated by bone morphogenetic proteins 15946907_SOST regulation may play a role in mediating PTH action in bone 16081646_Suppression of Sost by PTH represents a novel mechanism for hormonal control of osteoblastogenesis mediated by osteocytes. 17002572_functional interactions between Sost or Wise and LRP5 have the potential to regulate bone deposition by modulating Wnt signaling 17032150_Bone morphogenetic protein-induced ectopic bone formation in calf muscle of mice was prevented by co-expression of sclerostin in vivo. 17185821_Mechanical stimulation in vivo reduces osteocyte expression of sclerostin. 17185824_PTHR1 signaling in osteocytes is sufficient for inhibition of Sost expression and leads to a concomitant increase in bone mass. 17195180_SOST expression is restricted to the great arteries during embryonic and neonatal cardiovascular development. 17696759_SOST expression in osteocytes of adult bone and its inhibition by PTH is mediated by MEF2A, C, and D transcription factors controlling the SOST bone enhancer. 18089564_Modulation of sclerostin levels appears to be a finely tuned mechanism by which osteocytes coordinate regional and local osteogenesis in response to increased mechanical stimulation, perhaps via releasing the local inhibition of Wnt/Lrp5 signaling 18269310_sclerostin is a key negative regulator of a powerful, evolutionarily conserved bone formation pathway that acts on both trabecular and cortical bone 18521528_DKK1 and sclerostin are independent, and not synergistic, regulators of LRP5 signaling and that the function of each is impaired by HBM-LRP5 mutations 18927151_BMPR1A signaling in osteoblasts restrain endogenous bone mass directly by upregulating osteoclastogenesis through the RANKL-OPG pathway, or indirectly by downregulating canonical Wnt signaling through sclerostin 19075223_S1P induces osteoblast precursor recruitment and promotes mature cell survival. Wnt10b and BMP6 also were significantly increased in mature osteoclasts, whereas sclerostin levels decreased during differentiation. 19166819_The NMR structure of murine Sclerostin was presnted providing the first insights how Sclerostin might bind to LRP5. 19419300_Sclerostin played an essential role in mediating bone response to mechanical unloading, likely through Wnt/beta-catenin signaling. 19837663_the matricellular periostin protein is required for Sost inhibition and thereby plays an important role in the determination of bone mass and microstructural in response to loading. 19874086_BMPRIA in osteoblasts negatively regulates endogenous bone mass and Wnt/beta-catenin signaling and that this regulation may be mediated by the activities of Sost and Dkk1 19936252_Lrp4 is a novel osteoblast expressed Dkk1 and sclerostin receptor with a physiological role in the regulation of bone growth and turnover 20816666_these findings support our hypothesis that Sost is a direct target of Osx. 20952383_when both proteins are expressed in the same cell, sclerostin can antagonize BMP signaling directly by inhibiting BMP7 secretion 21079361_distribution of the osteocytic lacunar-canalicular system and osteocyte-secreting molecules--dentin matrix protein 1 and sclerostin--in the epiphyses and cortical bones in osteoprotegerin deficiency 21312267_Sclerostin acts through regulation of the PHEX/MEPE axis at the preosteocyte stage and serves as a master regulator of physiologic bone mineralization. 21329818_Sclerostin expressed in secretory odontoblasts located with ameloblasts of fetal mouse tooth germ; seen in fetal/adult osteocytes in jaw bone. suggest sclerostin is 1 of important regulatory factors of differentiated odontoblast function 21890008_Fractures in mice lacking sclerostin showed accelerated bridging, greater callus maturation, and increased bone formation and strength in the callus. 21945937_These results suggest that the Acvr1-deficiency can increase bone mass by activating Wnt signaling in which both Sost and Dkk1 expression levels are diminished. 21952235_role for Sirt1 in bone as a regulator of bone mass and a repressor of sclerostin 22075208_Data suggest that downregulation of Sost/sclerostin in osteocytes is an obligatory step in the mechanotransduction cascade that activates Wnt signaling and directs osteogenesis to where bone is structurally needed. 22434688_Sost has a novel role in the regulation of bone marrow environments that support B cells 22560000_sclerostin was not identified in the initial stages of cementum development, its biological significance seems to be restricted to cementum homeostasis and possibly to regenerative processes 22735261_these results suggest that TNF-alpha originating from T cells may be at least in part responsible for stimulating the sclerostin expression observed in an estrogen-deficient condition. 22768243_These findings indicate that the osteocyte network is required for the upregulation of Rankl in osteoblasts and Sost in osteocytes in the unloaded condition. 22886088_The absence of the Sost-specific long-range regulatory element ECR5 causes van Buchem disease in rodents, and that Mef2C is the main transcription factor responsible. 23044658_results demonstrate that ovariectomy has variable effects on sclerostin mRNA and protein in mice, which are dependent on the bones examined and the time after surgery 23171658_Data indicate that both Dickkopf-1 (DKK1) and sclerostin (SOST) were downregulated in proteoglycan-induced spondylitis (PGISp)-affected mouse spines. 23233270_Genetic absence of sclerostin does not alter the normal development of age-dependent osteoarthritis in mice. 23308287_Physical cues maintain bone homeostasis through the TGFbeta pathway to regulate Sclerostin expression and the deposition of new bone. 23362266_Estrogen receptor alpha mediates proliferation of osteoblastic cells stimulated by estrogen and mechanical strain, but their acute down-regulation of the Wnt antagonist Sost is mediated by estrogen receptor beta 23494985_osteoclasts from aged mice also produce sclerostin in quantities that may contribute to the age-related impairment in bone formation. 23530237_sclerostin not only alters bone mineralization, but also influences mineral metabolism by altering concentrations of hormones that regulate mineral accretion, including FGF23 23638027_Wnt3a-induced transcriptional responses and induction of alkaline phosphatase activity, an early marker of osteoblast differentiation, require the Wnt co-receptors LRP5 and LRP6 23737439_sclerostin upregulated osteocyte expression of carbonic anhydrase 2. 23776575_The findings support our hypothesis that HIF-1alpha activates Sost expression. 23782749_Sclerostin appears to be abundantly synthesized in regular osteocytic lacunar-canalicular system (OLCS) of cortical bone, but less produced in irregular OLCS as seen in primary trabecules. 23994639_Sost and its paralog Sostdc1 coordinate digit number in a Gli3-dependent manner. 24211207_these data indicate that enhanced beta-catenin signaling is present in Sost(-/-) mice that demonstrate accelerated healing of bone defects, suggesting that modulation of beta-catenin signaling in bone could be used to promote fracture repair. 24225945_In the absence of LRP5, the anabolic effects of SOST depletion can occur via other receptors (such as LRP4/6) 24312339_importance of the flexible loop and the cystine-knot for Wnt-signaling inhibition 24432364_Data indicate that sclerostin blockade restored bone mineral density (BMD) and bone volume fraction at all assessed sites but was unable to repair focal erosions. 24446199_These results support a model in which, in the context of obesity or other inflammatory diseases that increase the production of TNF-alpha, TNF-alpha upregulates the expression of sclerostin through NF-kappaB signaling pathway, thus contributing to bone loss. 24699186_results suggest that the lack of sclerostin mainly alters the bone and cementum phenotypes rather than producing abnormalities in tooth structures such as dentin 24753092_the altered bone composition contributes to the increased bone strength of patients with sclerostin deficiency. 24789067_Findings strongly suggest that Wise and Sost are key modulators of bone development through the ability of their encoded proteins to interact with Lrp5 and control the balance or levels of Wnt signaling. 24821585_long-term Sclerostin deficiency inhibits the bone loss normally induced with decreased mechanical load, but it can augment the increase in bone formation with increased load. 24876121_Evidence is accumulating that sclerostin directly or indirectly reduces renal calcium reabsorption [review]. 24949665_Ovariectomy resulted in a substantial decrease in skeletal Sirt1 expression accompanied by an increase in sclerostin. 25046534_Estrogen replacement treatment in ovariectomized ER beta KO mice caused a significant increase in Col2 expression, no change in ER alpha expression, and a significant increase in Sost expression. 25056116_Data indicate that low-dose parathyroid hormone (PTH) decreased the expression of the myocyte enhancer factor 2C (Mef2c) transcription factor, resulting in decreased sclerostin (Sost) expression in osteoblasts/osteocytes. 25271055_increased sclerostin production achieved by HDAC5 shRNA is abrogated by simultaneous knockdown of MEF2C, indicating that MEF2C is a major target of HDAC5 in osteocytes 25404300_Data indicate that LRP4 (low-density lipoprotein receptor-related protein 4) deficiency in bone dramatically elevated serum sclerostin levels whereas bone expression of Sost encoding for sclerostin was unaltered. 25445453_bony union was not altered by Sclerostin deficiency in externally-fixed closed tibial fractures, but fibrocartilage removal was enhanced and the resultant united bony calluses had increased bone fraction and increased strength. 25640331_Sclerostin inhibits bone formation through Lrp5 interaction. 25660312_sclerostin is regulated by glutathione, N-acetylcysteine and lipoic acid in osteocytes in a process involving JNK and ERK1/2 25721666_Advanced glycation end products increased sclerostin as well as apoptosis, and decreased RANKL in osteocytes. 25757567_deleting the Sost gene (a potent inhibitor of WNT signaling) or blocking sclerostin function by using the mAb in a periodontitis model significantly restores bone and periodontal ligament defects 25808845_These in vivo data support in vitro studies regarding the mechanism of HBM-causing mutations, and imply that HBM LRP5 receptors differ in their relative sensitivity to inhibition by SOST and DKK1. 25847683_Data (including data from studies in knockout/transgenic mice) suggest that Lrp6 (lipoprotein receptor-related protein 6) is required for suppression of Sost expression by parathyroid hormone (here, human PTH peptide 1-34). 25953900_Simulated microgravity induces an autonomous up-regulation of sclerostin. 26218891_thyroid hormone-induced changes in bone remodeling are associated with a divergent regulation of DKK1 and sclerostin 26337453_SOST gene is involved in the regulation of renal interstitial fibrosis (RIF) progression. In obstructive kidney injury, SOST gene deletion would probably enhance renal fibrogenic response and promote the progression of RIF. 26361013_Analysis of SOST expression using large minigenes reveals the MEF2C binding site in the evolutionarily conserved region (ECR5) enhancer mediates forskolin, but not 1,25-dihydroxyvitamin D3 or TGFbeta1 responsiveness. 26456319_Data show that the phenotype of Notch activation in osteocytes was prevented in matrix protein 1 (Dmp1)-Cre;Rosa(Notch) mice hemizygous for the Dmp1-sclerostin (SOST) transgene. 26553151_Results found that sclerostin enhances adipocyte differentiation in 3T3-L1 cells and reduced TAZ-responsive transcriptional activity and TAZ-responsive gene expression, indicating a role for TAZ as a regulator of adipogenesis by sclerostin. 26608966_Sclerostin depletion enhances tibial fracture healing. 26713363_These findings indicated that AMPK regulated RANKL and sclerostin expression through the mevalonate pathway in osteocytes. 26763740_humanized Multiple Myeloma xenograft mouse model bearing human MM cells (NOD-SCID.CB17 male mice injected intravenously with 2.5 million of MM1.S-Luc-GFP cells) demonstrated significantly higher concentrations of mouse-derived sclerostin, suggesting a microenvironmental source of sclerostin. 26845353_Our results suggested that sclerostin could be expressed in the liver and sustained successfully at high levels in the blood by using the PhiC31 integrase system, leading to trabecular bone loss. 27089204_chronic TNFalpha (tumor necrosis factor alpha)-dependent arthritis, fibroblast-like synoviocytes constitute a major source of sclerostin and that either the lack of sclerostin or its antibody-mediated inhibition leads to an acceleration of rheumatoid arthritis (RA)-like disease. 27163932_Protection From Glucocorticoid-Induced Osteoporosis by Anti-Catabolic Signaling in the Absence of Sost/Sclerostin 27349550_removal of sclerostin appears to modestly protect the alveolar bone from resorption in this experimental setting 27402532_loss of BMP signaling specifically in osteocytes dramatically increases bone mass presumably through simultaneous inhibition of RANKL and SOST, leading to osteoclast inhibition and Wnt activation together. 27528549_complete absence of sclerostin has only minor effects on chronic kidney disease-induced bone loss in mice. 27653320_These results show that osteocytes and/or osteoblasts secrete factors regulating beige adipogenesis, at least in part, through the Wnt-signaling inhibitor sclerostin. 28062506_Sclerostin inhibits angiotensin II-induced aortic aneurysm and atherosclerosis via wnt signaling pathway inhibition. 28081119_These data suggest that sclerostin plays an important role in the bone remodeling of tooth movement. 28460416_Osteocyte-derived molecule sclerostin drives bone marrow adipogenesis. 28543818_osteoclast-derived LIF regulates bone turnover through sclerostin expression. 28571484_In mice, sclerostin deficiency hastened reparative dentinogenesis after pulp injury, suggesting that the inhibition of sclerostin may constitute a promising therapeutic strategy for improving the healing of damaged pulps. 28794403_our study provided histological evidences that sclerostin tends to be secreted in osteocytes of remodeled mature bone, while FGF23 would be differently synthesized in osteoblasts and osteocytes according to the developmental stages 28842678_Sost deficiency led to a greater cortical bone formation response to mechanical loading and altered gene expression 29024567_Findings establish mechanical loading-induced attenuation of sclerostin expression and elevation of bone formation along the subchondral bone plate surface as the major mechanisms characterizing subchondral bone phenotypes associated with severe late-stage osteoarthritis in mice 29149200_In vivo muCT analysis of cortical bone at age 1 and 3 months confirmed increased thickness in Sost-/-mice, but revealed no cortical abnormalities in single Gja1+/-or Sost+/-mice 29162742_A microtubule-dependent mechanotransduction pathway that linked fluid shear stress to reactive oxygen species and calcium (Ca2+) signals that led to a reduction in sclerostin abundance in cultured osteocytes. 29229807_Since adipocytes do not produce sclerostin, these findings suggest an unexplored endocrine function for sclerostin that facilitates communication between the skeleton and adipose tissue. 29351359_Overexpression of CYR61/CCN1 can suppress Sost expression in both control and Cyr61 knockout cells, and blocking Sost with siRNA can rescue Wnt responsiveness in Cyr61 knockout cells in vitro. 29377313_SOST(transgenic) had approximately 2-fold less MMP activation than WT or Sost(-/-) , and this was supported by a significant reduction in MMP2/3 protein levels. 29734465_lack of Dkk1 in cells of the osteoblastic lineage leads to high bone mass with increased bone formation, despite increased levels of sclerostin. 29750826_several cell types within the Prx1-osteoprogenitor-derived lineages contribute significant amounts of sclerostin protein to the paracrine pool of Sost in bone. 29845410_In this study we aimed to explore the regulatory interaction between DKK1 and sclerostin within the bone compartment 29967369_Oxygen sensing by PHD2 in osteocytes negatively regulates bone mass through epigenetic regulation of sclerostin and targeting PHD2 elicits an osteo-anabolic response in osteoporotic mouse models. 30041240_The SOST gene inhibited the expression of COL1, OCN, and OPN, reduced the activity of alkaline phosphatase, and increased the expression of LPL and PPARgamma. 30071108_Study demonstrated that sclerostin is expressed in chondrocytes and upregulates the early stage of chondrogenic differentiation in vitro. Furthermore, sclerostin downregulates the expression of markers associated with the late stage of chondrogenic differentiation. 30266511_Gsalpha signaling in osteocytes leads to osteopenia driven, at least in part, by increased secretion of sclerostin. 30689868_our study elucidated the timing of development of bone and muscle abnormalities. The bone abnormalities in dKO-Hom mice are correlated with lower serum RANKL and higher SOST levels that resulted in dysregulation of osteogenesis and osteoclastogenesis and bone loss 30735798_Advanced glycation end-product 2 and Porphyromonas gingivalis lipopolysaccharide influence bone metabolism and inflammation through the regulation of sclerostin expression. 31088250_Results found that Sost expression is regulated by TSC1 which promotes its production. 31292298_Study in knockout mice provides evidence that bone 1alpha-hydroxylase Cyp27b1 may play a central role in the regulation of sclerostin, BMP2, and FGF23 in chronic kidney disease. Increased osteocytic 1,25(OH)2D production, triggered by renal malfunction, may represent a 'primary defensive response' to protect the organism from ectopic calcification by increasing sclerostin and suppressing BMP2 production. 31501981_Sclerostin, secreted by osteocytes, played a role in bone loss during lactation and also in the recovery of trabecular bone compartment by activating bone formation after weaning. 31505764_Twist1 Inactivation in Dmp1-Expressing Cells Increases Bone Mass but Does Not Affect the Anabolic Response to Sclerostin Neutralization. 31708103_Mechanically stretched osteocytic IDG-SW3 cells exhibit decreased Sost expression. Mechanical stretch induces Sost suppression via Piezo1-Akt pathway in osteocytes. 31828128_SOST KO mice with osteoarthritis (OA) exhibited a higher BMD and BV/TV ratio, as well as a higher rate of bone remodeling and TRAP-positive cell number, when compared to the WT counterparts, but the difference was not significant between the sham-operation groups. It was concluded that loss of sclerostin aggravates knee OA in mice by promoting subchondral bone sclerosis and increasing catabolic activity of cartilage 31970653_The Effects of Sclerostin on the Immune System. 31975377_Sclerostin antibody treatment rescues the osteopenic bone phenotype of TGFbeta inducible early gene-1 knockout female mice. 32134561_Reduction of SOST gene promotes bone formation through the Wnt/beta-catenin signalling pathway and compensates particle-induced osteolysis. 32792620_Sclerostin expression in trabecular bone is downregulated by osteoclasts. 32976505_Sclerostin inhibits interleukin-1beta-induced late stage chondrogenic differentiation through downregulation of Wnt/beta-catenin signaling pathway. 33078679_Molecular Basis for Craniofacial Phenotypes Caused by Sclerostin Deletion. 33148174_Scl-Ab reverts pro-osteoclastogenic signalling and resorption in estrogen deficient osteocytes. 33339872_Cortical bone adaptation to a moderate level of mechanical loading in male Sost deficient mice. 33479403_Role of osteogenic Dickkopf-1 in bone remodeling and bone healing in mice with type I diabetes mellitus. 33767359_Kindlin-2 mediates mechanotransduction in bone by regulating expression of Sclerostin in osteocytes. 33907818_Advanced oxidation protein products aggravate agerelated bone loss by increasing sclerostin expression in osteocytes via ROSdependent downregulation of Sirt1. 34421825_Decreased Sclerostin Secretion in Humans and Mice With Nonalcoholic Fatty Liver Disease. 34502021_Sclerostin Depletion Induces Inflammation in the Bone Marrow of Mice. 34606641_Bone-derived sclerostin and Wnt/beta-catenin signaling regulate PDGFRalpha(+) adipoprogenitor cell differentiation. 35099616_Competitive blocking of LRP4-sclerostin binding interface strongly promotes bone anabolic functions. 35169297_Regulation of sclerostin by the SIRT1 stabilization pathway in osteocytes. 35278242_Suppression of Sost/Sclerostin and Dickkopf-1 Augment Intervertebral Disc Structure in Mice. 36087347_Sclerostin aggravates insulin signaling in skeletal muscle and hepatic steatosis via upregulation of ER stress by mTOR-mediated inhibition of autophagy under hyperlipidemic conditions. |
ENSG00000167941 |
SOST |
34.285123 |
0.0099909713 |
-6.645159 |
1.12523716 |
69.797514 |
0.00000000000000006571564694005927509118176275304914290066880176678296154868519352021394297480583190917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000019900213403399261756579861469965555934136356049102278831242074375040829181671142578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.600654470971988 |
0.517448022873185 |
60.1197274588445 |
15.7697972407989 |
| ENSMUSG00000001508 |
20391 |
Sgca |
protein_coding |
P82350 |
FUNCTION: Component of the sarcoglycan complex, a subcomplex of the dystrophin-glycoprotein complex which forms a link between the F-actin cytoskeleton and the extracellular matrix. |
Cell membrane;Cytoplasm;Cytoskeleton;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the sarcoglycan alpha/epsilon family of transmembrane proteins. The encoded protein is part of the dystrophin-glycoprotein complex which links the extracellular matrix to the cytoskeleton in muscle fibers. Disruption of this gene results in progressive muscular dystrophy and is associated with the development of embryonal rhabdomysarcoma. [provided by RefSeq, Dec 2012]. |
mmu:20391; |
cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; dystroglycan complex [GO:0016011]; membrane raft [GO:0045121]; plasma membrane [GO:0005886]; sarcoglycan complex [GO:0016012]; sarcolemma [GO:0042383]; calcium ion binding [GO:0005509]; membrane organization [GO:0061024]; response to denervation involved in regulation of muscle adaptation [GO:0014894]; skeletal muscle tissue regeneration [GO:0043403] |
12620894_These findings suggest that the sarcoglycan complex serves a mechanical function in the diaphragm by contributing to muscle passive stiffness and to the modulation of the contractile properties of the muscle. 15886330_Muscle masses were 40-100% larger for Sgca-null compared with age- and gender-matched wild-type mice, with the majority of the increased muscle mass for Sgca-null mice attributable to greater connective tissue and water contents 15990925_Diaphragm from Sgca-null mouse presents a clear dystrophic phenotype, with necrosis, regeneration, fibre hypertrophy and splitting, excess of collagen and fatty infiltration. 16002556_Deficiency of Sgca differently affects fast- and slow-twitch skeletal muscles. 16797743_The alpha-SG promoter is activated by MyoD, which interacts with TFIID and TFIIB in a protein complex differentially located at the distal promoter and around the proximal promoter during myogenic cell differentiation. 18078839_identified a negative regulatory element in the alpha-SG distal promoter including two conserved E-boxes (E1 and E2), which interact with MyoD 18252746_Contrary to expectations, mice homozygous for the H77C-encoding allele expressed both this mutant alpha-sarcoglycan and the other components of the sarcoglycan-sarcospan complex in striated muscle, and did not develop muscular dystrophy. 19729026_Sox9 and Smad3 are responsible for preventing precocious activation of alpha-SG gene expression during myogenic differentiation. 20019182_Absence of members of the dystrophin-associated glycoprotein complex constitutes a permissive environment for spontaneous development of embryonal rhabdomyosarcoma associated with mutation of p53 and mutation or altered splicing of Mdm2. 21890494_Sgca-;Sgce-null mouse shows a complete loss of residual sarcoglycans and a strong reduction in both dystrophin and dystroglycan. 21903674_Impaired proliferation of Sgca-null myogenic precursors was confirmed by single fiber analysis and this difference correlated with Sgca expression during myogenic progenitor cell proliferation. 23387296_Loss of sarcoglycan is associated with loss of miRNA669a and myopathy. 25091331_The differential expression of two alpha-SG mRNAs during mouse embryonic development may be a consequence of the differential regulation of both promoters by myogenic and cardiogenic factors. 30448410_The Danger Signal Extracellular ATP Is Involved in the Immunomediated Damage of alpha-Sarcoglycan-Deficient Muscular Dystrophy. |
ENSG00000108823 |
SGCA |
196.492702 |
0.0877386161 |
-3.510644 |
0.27597388 |
153.742695 |
0.00000000000000000000000000000000002636091251492889744864376858434484110473301831293618493180588589714705764479193551241991192411739941547921262099407613277435302734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000024785766631320801929269704757408504935213256284041995371531611255578101798823622683239260855447838594045606441795825958251953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.6616395768595 |
6.28603534347586 |
372.260710553904 |
46.5743335006921 |
| ENSMUSG00000001510 |
13393 |
Dlx3 |
protein_coding |
Q64205 |
FUNCTION: Likely to play a regulatory role in the development of the ventral forebrain. May play a role in craniofacial patterning and morphogenesis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome |
|
|
mmu:13393; |
nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; blood vessel development [GO:0001568]; BMP signaling pathway [GO:0030509]; cell differentiation [GO:0030154]; epithelial cell differentiation [GO:0030855]; gene expression [GO:0010467]; hair cell differentiation [GO:0035315]; hair cycle [GO:0042633]; hair follicle cell proliferation [GO:0071335]; hair follicle development [GO:0001942]; hair follicle morphogenesis [GO:0031069]; odontoblast differentiation [GO:0071895]; odontogenesis of dentin-containing tooth [GO:0042475]; placenta development [GO:0001890]; regulation of transcription by RNA polymerase II [GO:0006357]; Wnt signaling pathway [GO:0016055] |
11773066_AP-2 gamma and Dlx 3, together with an additional transcription factor(s) that are conserved between humans and mice, are required for trophoblast-specific expression of 3 beta-HSD VI. 11788714_Bone morphogenetic protein-2 (BMP-2) transactivates Dlx3 through Smad1 and Smad4: alternative mode for Dlx3 induction in mouse keratinocytes 11792834_Genomic structure and functional control of the Dlx3-7 bigene cluster 12642674_Gene expression regulation of this protein in visceral arches is evolutionarily conserved. 15456894_Results describe gene regulation by Dlx3 in relation to that of Msx2 and Dlx5 during osteoblast differentiation. 17060321_DLX3 is one of 3 homeodomain proteins that provide a key series of molecular switches that regulate expression of Runx2 throughout bone formation. 17110422_Dlx3 is sufficient to directly modulate expression of the Placental growth factor gene promoter in placental cells. 17950683_DLX3 mutation also accelerates the differentiation of osteoprogenitor cells to osteoblasts at later stages of osteogenesis 18684741_Dlx3 is a crucial regulator of hair follicle differentiation and cycling. 18996110_These data suggest a novel role for this DLX-3 mutation in osteoclast differentiation and bone resorption. 20510228_Mutant Dlx3 disrupts odontoblast polarization and dentin formation. 21709238_Dlx3 ablation in epidermis is linked to altered epidermal differentiation, barrier development, and IL-17-associated skin inflammation. 22351765_The transcription factor Dlx3 is essential in dentin formation by directly regulating a crucial matrix protein. 22442153_regulation of Dlx3 by HR affects the IRS keratin expression, thus modulating the formation of IRS of hair follicle. 22886599_used a conditional knockout approach to analyze the effects of neural crest deletion of Dlx3 on craniofacial bones development 24647893_we provide a novel insight that BMP-2-induced Dlx3 expression is regulated by p38/Smad5 signaling pathway in osteoblasts. 24924519_Results suggest that Dlx3 is a novel target of PKA, and that PKA mediates BMP signaling during osteoblast differentiation, at least in part, by phosphorylating Dlx3 and modulating its stability and function. 24948010_The DLX3 regulates transcription factors crucial for bone formation and DLX3 attenuates bone mass accrual to support bone homeostasis by osteogenic gene pathway regulation. 25120002_We show that the deletion of Dlx3 in epidermis and isthums/infundibulum area leads to hair shaft differentiation but not hair growth. 25815730_our data show that DLX3 promotes the expression of the EMP genes Amelx, Enam, Klk4, and Odam in amelogenesis. 26674964_ER-alpha regulates the osteoblast differentiation through modulation of Dlx3 expression and/or interaction with Dlx3. 26836061_indicate that Distal-less-3 negatively regulates osteoclastic differentiation through microRNA-124 27760456_DLX3 is indispensable for the regulation of ion transporters and carbonic anhydrases during the maturation stage of amelogenesis, exerting a crucial regulatory function on pH oscillations during enamel mineralization. 29246798_DLX3 is a critical regulator of STAT3 signaling network that maintains skin homeostasis. 29745813_DLX3 regulates the expression of some components of myosin II complexes potentially involved in driving the movement of ameloblasts that leads to enamel rod decussation. 35220830_Dlx3 Ubiquitination by Nuclear Mdm2 Is Essential for Dentinogenesis in Mice. |
ENSG00000064195 |
DLX3 |
1101.626010 |
0.4735912826 |
-1.078286 |
0.09742387 |
120.848686 |
0.00000000000000000000000000041241848392954008444027195417534532989535110576194040561060882214459719975231666300174993011751212179660797119140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000279198148765100169779147383214234050644375979233392652184307323044417173396603004675853298977017402648925781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
707.779108783297 |
47.8320055602301 |
1494.49353217785 |
73.6923094880148 |
| ENSMUSG00000001517 |
14235 |
Foxm1 |
protein_coding |
Q6P1H7 |
Human_homologues FUNCTION: Transcription factor regulating the expression of cell cycle genes essential for DNA replication and mitosis (PubMed:19160488, PubMed:20360045). Plays a role in the control of cell proliferation (PubMed:19160488). Also plays a role in DNA break repair, participating in the DNA damage checkpoint response (PubMed:17101782). Promotes transcription of PHB2 (PubMed:33754036). {ECO:0000269|PubMed:17101782, ECO:0000269|PubMed:19160488, ECO:0000269|PubMed:20360045, ECO:0000269|PubMed:33754036}. |
DNA-binding;Nucleus;Proteomics identification;Reference proteome |
|
|
mmu:14235; |
nucleus [GO:0005634]; DNA-binding transcription factor activity [GO:0003700]; protein kinase binding [GO:0019901]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; DNA damage response, signal transduction by p53 class mediator resulting in transcription of p21 class mediator [GO:0006978]; G2/M transition of mitotic cell cycle [GO:0000086]; negative regulation of stress-activated MAPK cascade [GO:0032873]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of double-strand break repair [GO:2000781]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of cell cycle [GO:0051726]; regulation of Ras protein signal transduction [GO:0046578]; regulation of reactive oxygen species metabolic process [GO:2000377] |
12221098_restoring FoxM1B expression in old-aged mice caused elevated levels of Cyclin B1, Cyclin B2, Cdc25B, Cdk1, and p55CDC mRNA as well as stimulating Cdc25B nuclear localization during liver regeneration, all of which are required for mitosis 12482952_Foxm1b transcription factor regulates expression of cell cycle proteins essential for hepatocyte entry into DNA replication and mitosis. 12867420_increasing FoxM1B levels is an effective means to stimulate cellular proliferation during aging and in lung diseases such as emphysema 13679865_Sustained hepatic expression of Foxm1 in transgenic mice increases cell proliferation rates in preneoplastic and early neoplastic lesions, and has minimal effects on hepatocellular carcinoma development. 14647066_Growth hormone stimulates proliferation of old-aged regenerating liver through forkhead box m1b. 15531365_Foxm1 is essential for hepatoblast mitosis and development of intrahepatic bile ducts and vessels during liver morphogenesis. 15817462_development of mouse lungs depends on the Foxm1 transcription factor, which regulates expression of genes essential for mesenchyme proliferation, extracellular matrix remodeling, and vasculogenesis 16556734_Foxm1 is essential to maintain normal beta-cell mass and regulate postnatal beta-cell turnover. 17101782_These results identify a novel role for FoxM1 in the transcriptional response during DNA damage/checkpoint signaling and show a novel mechanism by which Chk2 protein regulates expression of DNA repair enzymes. 17173139_To pharmacologically reduce FoxM1 activity in hepatocellular carcinoma, we subjected these mice to daily injections of a cell-penetrating ARF(26-44) peptide inhibitor of FoxM1 function. 17366632_Foxm1 regulates expression of genes essential for the proliferation of cardiomyocytes during heart development. 17891172_The N-terminal domain of FoxM1 interacts with the C-terminal half of the transcription factor to attenuate its transcriptional activity. 17893320_These findings indicate that the functions of FoxM1 in Shh-induced neuroproliferation are restricted to the regulation of the G2/M transition in cerebellar granule neuron precursors. 17984092_Results show that FoxM1 promotes cell cycle progression by down-regulating p27(Kip1) through multiple mechanisms. 18345025_Transgenic expression of the forkhead box M1 transcription factor induces formation of lung tumors. 18393292_TIS21 negatively regulated hepatocarcinogenesis in part by disruption of the FoxM1-cyclin B1 regulatory loop, thereby inhibiting proliferation of transformed cells developed in mouse and human livers. 18408007_FoxM1c counteracts oxidative stress-induced senescence and stimulates Bmi-1 expression 18524773_JNK1 is a critical transcriptional target of FoxM1 that contributes to FoxM1-regulated cell cycle progression, tumor cell migration, invasiveness, and anchorage-independent growth 18798339_c-Myc and FoxM1 mediated proliferative programs are key mediators of TCPOBOP-CAR induced direct liver hyperplasia. 18803232_chromatin mediated gene silencing in IKKbetaca's short term repressive effects on E2F and FoxM1 target gene expression 19033457_Foxm1 is critical for surfactant homeostasis and lung maturation before birth and is required for adaptation to air breathing 19672312_Expression of Foxm1 in respiratory epithelial cells is critical for lung cancer formation and TOPO-2alpha expression in vivo, suggesting that Foxm1 is a promising target for anti-tumor therapy 19696738_Induction of Foxm1 by oncogenic Ras requires reactive oxygen species (ROS). Elevated Foxm1, in turn, downregulates ROS levels by stimulating expression of ROS scavenger genes. 19833884_FoxM1 functions downstream of placental lactogen to mediate its effect on beta-cell proliferatoin. 19835856_Depletion of Foxm1 in cultured SMC caused G(2) arrest and decreased numbers of cells undergoing mitosis. Foxm1-deficiency in vitro and in vivo was associated with reduced expression of cell cycle regulatory genes 19998409_Our results have revealed Foxm1b as the first known direct FXR target gene involved in cell cycle regulation and have demonstrated that defective activation of FXR could be an intrinsic defect in aging regenerating livers 20023695_Studies identified FOXM1 as a key downstream target of Ras-MKK3-p38 regulated in vitro cellular invasion. 20111592_In the absence of IGF-I, nuclear levels of MEF2 were diminished, indicating less transcriptional MEF2 activity. By contrast, there was an increase in the nuclear accumulation of FoxM1 20169079_Data show that regulation of cell cycle genes by FoxM1 is stage- and context-dependent. 20660304_FoxM1 stimulated mouse and human beta-cell proliferation by activating many cell cycle phases. 20660612_FoxM1 is required for reannealing of endothelial adherens junctions in order to form a restrictive endothelial barrier through transcriptional control of beta-catenin expression. 20702419_To study whether Foxm1 participates in the maintenance of pluripotency of stem cells, Foxm1 expression was knockdown in P19 cells and identified that Oct4 is regulated directly by Foxm1. 20816795_Foxm1 expression was detected in epithelial cells of conducting and peripheral airways and changed dynamically with lung maturation. 20837707_Data show that Foxm1 is critical for liver repair and is required for the recruitment of monocytes to the injured liver. 20887555_Checkpoint kinase 1 and p21 are direct transcription targets of FoxM1 and FoxM1 participates in transcriptional responses to stress in normal cells. 21199796_results suggest that endothelial-specific expression of FoxM1 limits lung inflammation and canonical Wnt signaling in lung epithelial cells, thereby restricting lung tumorigenesis 21270518_FoxM1 has a role in embryonic development, organ injury and cancer formation. [review] 21705713_CAR-mediated hepatocyte proliferation was dependent on estrogen signaling and associated with enhanced expression of FoxM1. 21708928_identified FoxM1 in type II alveolar epithelial cells as the key regulator of alveolar epithelial barrier repair through its ability to control proliferation and trans-differentiation into type I cells 21712406_This review discusses the evidence for a master regulatory role of FoxM1 in tumor metastasis. 21779394_Results indicate that Foxm1 expression in cardiomyocytes is critical for proper heart development and required for cardiomyocyte proliferation and myocardial growth. 21993994_AKT and N-Ras coactivation in the mouse liver promotes rapid carcinogenesis by way of mTOR, FOXM1, and c-Myc pathways. 22139074_Data show that expression of Forkhead Box m1 (Foxm1) transcription factor in macrophages is required for pulmonary inflammation, recruitment of macrophages into tumor sites and lung tumor growth. 22513242_Our data suggest that B-Myb is required as a pioneer factor to enable FoxM1 binding to G2/M gene promoters and explains how these transcription factors may collaborate to induce mitosis. 22813746_This study identifies FoxM1 as a critical regulator of mammary differentiation with significant implications for the development of aggressive breast cancers. 22826436_Foxm1 is required for Kras signaling in distal lung epithelium and provides a mechanism integrating Kras and canonical Wnt/beta-catenin signaling during lung development. 22885335_Foxm1 is required for differentiation and maintenance of epithelial cells lining conducting airways. 22982677_downregulation of FoxM1 is required for contact inhibition by regulating expression of cyclin A and polo-like kinase 1. 23144938_Foxm1 regulates genes critical for age-induced cardiomyocyte hypertrophy and cardiac fibrosis. 23149934_Pulmonary FOXM1 expression is increased after allergen stimulation. Deletion of FOXM1 from myeloid cells reduced pulmonary inflammation and airway resistance after allergen challenge. FOXM1 deletion decreased goblet cell metaplasia in airway epithelium. 23185540_FoxM1 endothelial cell expression promotes endothelial repair following experimentally induced lung injury and sepsis. 23192351_FoxM1 is regulated by HSF1 and promotes cell cycle progression and cancer cell survival under heat stress conditions. 23576572_Results show that the miR-370-FoxM1 pathway is involved in the progress of gastritis toward gastric cancer induced by H. pylori infection. 23665201_RNA interference-mediated knockdown of FOXM1 further elevates intracellular reactive oxygen species (ROS) levels and increases sensitivity of cancer cells to ROS-mediated cell death. 23804320_These findings highlight the protective effects of ERK/FOXM1 pathway against UVB-induced cell senescence, suggesting a potential protecting strategy for treating skin aging by LPLI 24213573_Data indicate that forkhead box M1 protein (FOXM1) is required for K-Ras-mediated lung tumorigenesis. 24453338_this study provides evidence of linkages among the cell cycle regulator Foxm1, interkinetic nuclear migration, and adult behavior 25073103_FoxM1 has a role in promoting beta-cell survival 25254494_SPDEF inhibits prostate carcinogenesis by disrupting a positive feedback loop in regulation of the Foxm1 oncogene. 25275225_Foxm1 influences pulmonary inflammatory responses to hyperoxia, inhibiting neutrophil-derived enzymes and enhancing monocytic responses that limit alveolar injury and remodeling in neonatal lungs. 25400737_FoxM1 might play an important role during the process of mouse embryo implantation. 25426548_Both gain-of-function and loss-of-function TP53 mutations contribute to overexpression of FoxM1 in high-grade serous ovarian cancer. 25477198_enrichment of FOXM1, as with other oncoproteins, may cause increased persistent DNA lesions and/or senescence in normal murine hepatocytes 26022336_EGF promotes FoxM1 expression through the ERK signal pathway 26045783_FoxM1 expression correlates with proliferation, invasion and migration in mouse hepatocellular carcinoma cell lines. 26075743_null mice have reductions in both the somatotrope and gonadotrope cell populations. 26080894_MicroRNA-802 suppresses breast cancer proliferation through down-regulation of FoxM1. 26147687_Data indicate transcription factor Foxm1 as a critical regulator of the quiescence and self-renewal of hematopoietic stem cells (HSCs). 26251404_activated FoxM1 increases beta-cell replication while simultaneously enhancing insulin secretion and improving glucose homeostasis, making FoxM1 an attractive therapeutic target for diabetes. 26350477_we suggest that proper regional decidualization and polyploidy development requires FoxM1 signaling downstream of Hoxa10 and cyclin D3. 26387954_FOXM1 and CENPF are master regulators of prostate cancer malignancy, and can serve as drug response markers for antineoplastic drugs efficiency. 26839042_FoxM1 induction in the pulmonary vasculature was inhibited by a p110gamma-selective inhibitor and in Pik3cg(-/-) mice after LPS challenge. Defective vascular repair in Pik3cg-/- mice results from impaired FoxM1 expression 26869074_Interactions between the Wnt/beta-catenin and the Kras/ERK/Foxm1 pathways are essential to restrict SOX9 expression in basal cells during pulmonary branching morphogenesis 28249813_YAP cooperates with FOXM1 to contribute to chromosome instability in hepatocellular carcinoma. 28286049_These data implicate the insulin-FoxM1/PLK1/CENP-A pathway-regulated mitotic cell-cycle progression as an essential component in the beta cell adaptation to delay and/or prevent progression to diabetes. 28420758_RCM-1 blocked the nuclear localization and increased the proteasomal degradation of Forkhead box M1 (FOXM1), a transcription factor critical for the differentiation of goblet cells from airway progenitor cells. 29158087_Upregulated ROS induced by FABP4 was of significance in activating FoxM1 leading to airway inflammation and epithelial barrier dysfunction. 29208957_Vagal signals activate the forkhead box M1 (FoxM1) pathway in beta-cells, resulting in compensatory beta-cell proliferation. Inducible beta-cell-specific FoxM1 deficiency also blocks compensatory beta-cell proliferation. 29476172_Foxm1, which proved to be directly regulated by Gli and Nanog, controls a pro-stemness microRNA network in neural stem cells. 29664678_Signaling via Interaction with Endothelial and Smooth Muscle Cells Mediates Vascular Remodeling and Pulmonary Hypertension 29733296_results identify FOXM1 as a driver of lung fibroblast activation and underscore the therapeutic potential of targeting FOXM1 for pulmonary fibrosis. 29950315_Disrupting LXRalpha phosphorylation promotes FoxM1 expression and modulates atherosclerosis by inducing macrophage proliferation 30092353_this study showed that in hypertensive human lungs or mouse lungs, FoxM1 levels were elevated. 30546054_vagal signal-mediated interleukin-6 production in hepatic macrophages upregulates hepatocyte FoxM1, leading to liver regeneration and assures survival. 31121134_Endothelial Hypoxia-Inducible Factor-1alpha Is Required for Vascular Repair and Resolution of Inflammatory Lung Injury through Forkhead Box Protein M1. 31669262_Forkhead Box M1 Transcription Factor Drives Liver Inflammation Linking to Hepatocarcinogenesis in Mice. 31740799_Foxm1 deletion suppressed the capacity of arthritis-associated osteoclastogenic macrophages to differentiate into osteoclasts in vitro and in vivo 32066721_Foxm1 is required for survival, quiescence and self-renewal of MLL-AF9 (MA9)-transformed leukemia stem cells (LSCs) in vivo. Foxm1 upregulation activates the Wnt/beta-catenin signaling pathways by directly binding to beta-catenin and stabilizing beta-catenin protein through inhibiting its degradation, thereby preserving LSC quiescence, and promoting LSC self-renewal in MLL-rearranged acute myeloid leukemia. 32271749_Macrophage-specific deletion of Foxm1 exacerbated pulmonary fibrosis. FOXM1 directly activated Dusp1 promoter. 32348194_FOXM1 nuclear transcription factor translocates into mitochondria and inhibits oxidative phosphorylation. 32475644_Overexpression of FoxM1 promotes differentiation of bone marrow mesenchymal stem cells into alveolar type II cells through activating Wnt/beta-catenin signalling. 32813289_Glycogen synthase kinase-3beta inhibits tubular regeneration in acute kidney injury by a FoxM1-dependent mechanism. 32915464_Loss of aryl hydrocarbon receptor potentiates FoxM1 signaling to enhance self-renewal of colonic stem and progenitor cells. 33323968_Regucalcin promotes dormancy of prostate cancer. 33434361_FoxM1 promotes Wnt/beta-catenin pathway activation and renal fibrosis via transcriptionally regulating multi-Wnts expressions. 33804745_Endothelial Autocrine Signaling through CXCL12/CXCR4/FoxM1 Axis Contributes to Severe Pulmonary Arterial Hypertension. 34427853_Involvement of miR-214-3p/FOXM1 Axis During the Progression of Psoriasis. 34497269_PRMT7 targets of Foxm1 controls alveolar myofibroblast proliferation and differentiation during alveologenesis. 34626587_FOXM1-activated SIRT4 inhibits NF-kappaB signaling and NLRP3 inflammasome to alleviate kidney injury and podocyte pyroptosis in diabetic nephropathy. 34808333_FOXM1-mediated activation of phospholipase D1 promotes lipid droplet accumulation and reduces ROS to support paclitaxel resistance in metastatic cancer cells. 34841254_FoxM1 insufficiency hyperactivates Ect2-RhoA-mDia1 signaling to drive cancer. 35583996_Transcriptional Repression by FoxM1 Suppresses Tumor Differentiation and Promotes Metastasis of Breast Cancer. 35910798_Microbial metabolite restricts 5-fluorouracil-resistant colonic tumor progression by sensitizing drug transporters via regulation of FOXO3-FOXM1 axis. 36788588_Overexpression of FoxM1 optimizes the therapeutic effect of bone marrow mesenchymal stem cells on acute respiratory distress syndrome. |
ENSG00000111206 |
FOXM1 |
1665.372744 |
2.2570827781 |
1.174459 |
0.09255852 |
158.961168 |
0.00000000000000000000000000000000000190820513020470608762326714762276971592964249377484124467603150328315477363592279312839350609038585604437798792787361890077590942382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000001899724218514907417658405605563963149552501568482347539723665853091218067636985288806921899282253995977498561842367053031921386718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2338.64822716375 |
150.142843361733 |
1036.1375532461 |
50.8220932698168 |
| ENSMUSG00000001542 |
192657 |
Ell2 |
protein_coding |
Q3UKU1 |
FUNCTION: Elongation factor component of the super elongation complex (SEC), a complex required to increase the catalytic rate of RNA polymerase II transcription by suppressing transient pausing by the polymerase at multiple sites along the DNA. Component of the little elongation complex (LEC), a complex required to regulate small nuclear RNA (snRNA) gene transcription by RNA polymerase II and III (By similarity). Plays a role in immunoglobulin secretion in plasma cells: directs efficient alternative mRNA processing, influencing both proximal poly(A) site choice and exon skipping, as well as immunoglobulin heavy chain (IgH) alternative processing. Probably acts by regulating histone modifications accompanying transition from membrane-specific to secretory IgH mRNA expression. {ECO:0000250, ECO:0000269|PubMed:19749764, ECO:0000269|PubMed:21832080}. |
Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:192657; |
nucleoplasm [GO:0005654]; transcription elongation factor complex [GO:0008023]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; positive regulation of transcription elongation by RNA polymerase II [GO:0032968]; snRNA transcription by RNA polymerase II [GO:0042795]; transcription elongation by RNA polymerase II promoter [GO:0006368] |
19749764_ELL2 enhanced both polyadenylation and exon skipping with the gene encoding the immunoglobulin heavy-chain complex (Igh) and reporter constructs 22991471_The splicing factor hnRNPLL and the transcription elongation factor ELL2 modulate the ratio of secred versus membrane-encoding Ighg2b transcripts at the expense of the secreted isoform. 25238757_Deletion of the Ell2 gene in mice results in loss of Ig secretion from plasma cells and reduced expression of a number of other genes involved in secretion and the unfolded protein response like XBP1, OcaB (BOB1, Pou2af1), cyclin B2 and Ig light chain 25238757_ELL2 is important for many aspects of Ab secretion, XBP1 expression, and the unfolded protein response. 28870994_These results suggest that ELL2 and its pathway genes likely play an important role in the development and progression of prostate cancer. |
ENSG00000118985 |
ELL2 |
360.551489 |
0.2943254266 |
-1.764516 |
0.25209822 |
46.447910 |
0.00000000000940866734703279108178951813256534787710538836336127133108675479888916015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001674215198948638712124041357420974052150342004097183234989643096923828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
162.021544845618 |
28.4561353579617 |
550.484362553312 |
70.7680525773977 |
| ENSMUSG00000001552 |
16480 |
Jup |
protein_coding |
Q02257 |
FUNCTION: Common junctional plaque protein. The membrane-associated plaques are architectural elements in an important strategic position to influence the arrangement and function of both the cytoskeleton and the cells within the tissue. The presence of plakoglobin in both the desmosomes and in the intermediate junctions suggests that it plays a central role in the structure and function of submembranous plaques. Acts as a substrate for VE-PTP and is required by it to stimulate VE-cadherin function in endothelial cells. Can replace beta-catenin in E-cadherin/catenin adhesion complexes which are proposed to couple cadherins to the actin cytoskeleton. {ECO:0000269|PubMed:19015309}. |
Acetylation;Cell adhesion;Cell junction;Cytoplasm;Cytoskeleton;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:16480; |
actin cytoskeleton [GO:0015629]; adherens junction [GO:0005912]; apicolateral plasma membrane [GO:0016327]; catenin complex [GO:0016342]; cell-cell junction [GO:0005911]; cornified envelope [GO:0001533]; cytoplasm [GO:0005737]; cytoplasmic side of plasma membrane [GO:0009898]; cytosol [GO:0005829]; desmosome [GO:0030057]; fascia adherens [GO:0005916]; gamma-catenin-TCF7L2 complex [GO:0071665]; intercalated disc [GO:0014704]; intermediate filament [GO:0005882]; lateral plasma membrane [GO:0016328]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protein-DNA complex [GO:0032993]; Z disc [GO:0030018]; alpha-catenin binding [GO:0045294]; cadherin binding [GO:0045296]; cell adhesion molecule binding [GO:0050839]; cytoskeletal protein-membrane anchor activity [GO:0106006]; DNA-binding transcription factor binding [GO:0140297]; protein kinase binding [GO:0019901]; protein phosphatase binding [GO:0019903]; protein tyrosine kinase binding [GO:1990782]; protein-containing complex binding [GO:0044877]; transcription coactivator activity [GO:0003713]; bundle of His cell-Purkinje myocyte adhesion involved in cell communication [GO:0086073]; canonical Wnt signaling pathway [GO:0060070]; cell adhesion [GO:0007155]; cell migration [GO:0016477]; cell-cell adhesion [GO:0098609]; cellular response to indole-3-methanol [GO:0071681]; desmosome assembly [GO:0002159]; detection of mechanical stimulus [GO:0050982]; negative regulation of blood vessel endothelial cell migration [GO:0043537]; positive regulation of angiogenesis [GO:0045766]; positive regulation of cell-matrix adhesion [GO:0001954]; positive regulation of DNA-binding transcription factor activity [GO:0051091]; positive regulation of protein import into nucleus [GO:0042307]; positive regulation of transcription by RNA polymerase II [GO:0045944]; protein localization to plasma membrane [GO:0072659]; regulation of cell population proliferation [GO:0042127]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of ventricular cardiac muscle cell action potential [GO:0098911]; skin development [GO:0043588] |
15805189_PG strengthens adhesion and suppresses motility in mouse keratinocytes, through both intercellular adhesion-dependent and -independent mechanisms 16183992_plakoglobin-dependent adhesion includes desmosome-specific functions in assembly and regulation by epidermal growth factor receptor 16357441_These data confirmed that beta-catenin or plakoglobin is required for alpha-catenin to form complex with E-cadherin,It is also reqd for the stable expression and cell surface localization of E-cadherin. 16510446_O-glycosylation functions to regulate the post-translational stability of plakoglobin and keratinocyte cell-cell adhesion 16823493_Data show suppression of desmoplakin expression leads to nuclear localization of plakoglobin and a 2-fold reduction in canonical Wnt/beta-catenin signaling through Tcf/Lef1 transcription factors. 16871158_Plakoglobin is a potent modulator of epithelial homeostasis via its role as a key suppressor of c-Myc in the skin. 16936006_The results suggest that upregulation of gamma-catenin can compensate for the loss of beta-catenin in cardiomyocytes to maintain normal cardiac structure and function. 17030684_Heterozygous plakoglobin deficiency provokes arrhythmogenic right ventricular cardiomyopathy. 17110936_Plakoglobin may potentiate death in cells damaged by apoptotic stimuli, perhaps limiting the potential for the propagation of mutations and cellular transformation. 17518986_PG is a principle effector of the PVIgG-induced signals downstream of c-Myc that disrupt the desmosomal plaque at the plasma membrane 17855627_results exclude an essential role for canonical Wnt signaling (as mediated by beta- and/or gamma-catenin) during hematopoiesis and lymphopoiesis 17906078_hematopoietic cells can transduce canonical Wnt signals in the combined absence of beta- and gamma-catenin 17984222_plakoglobin is insufficient for LEF/TCF-dependent transcriptional activation by Wnt-3a in F9 cells 18496566_Plakoglobin (PG) is a major component of the intracellular plaque that serves to connect transmembrane elements to the cytoskeleton. 19015309_Data show that leukocytes interacting with endothelial cells rapidly dissociate VE-PTP from VE-cadherin, weakening endothelial cell contacts via a mechanism that requires plakoglobin but not beta-catenin. 20876660_Data propose a novel role for PG in regulating cell motility through distinct ECM-Src and RhoGTPase-dependent pathways, influenced in part by PG-dependent regulation of FN mRNA stability. 21245375_Plakoglobin (JUP) knockout mice exhibited progressive loss of cardiac myocytes, extensive inflammatory infiltration, fibrous tissue replacement, and cardiac dysfunction similar to those of arrhythmogenic right ventricular cardiomyopathy patients. 21703193_While gamma-catenin gene expression remained unaltered, an increase in serine- and threonine-phosphorylated, but not tyrosine-phosphorylated gamma-catenin was observed in beta-catenin knockout livers. 21880664_Ventricular arrhythmias were associated with progressive cardiomyopathy and fibrosis in Jup mutant hearts. 22021931_Nuclear plakoglobin is essential for differentiation of cardiac progenitor cells to adipocytes in arrhythmogenic right ventricular cardiomyopathy. 22036570_The cadherin-catenin signaling pathway was inactivated in spinal motor neurons to assess the significance of motor neuron position in motor circuit assembly; genetic inactivation of both beta- and gamma-catenin or N-cadherin, disrupts motor neuron positioning. 22252313_Loss of Ncad-binding proteins beta-catenin and plakoglobin in the heart leads to gap junction remodeling and abrupt onset of spontaneous lethal ventricular arrhythmia in double knockout mice. 22315228_Lack of plakoglobin in epidermis leads to keratoderma. 22522917_The data suggest novel function(s) for plakoglobin (PG) beyond the heart and define a critical threshold of PG expression that is necessary for postnatal survival. 22632416_decreased plakoglobin expression increases the invasive behavior of breast cancer cells; this is the first demonstration of a functional role for plakoglobin/gamma-catenin in the metastatic process 23652796_plakoglobin (Pg) in conjunction with lymphoid enhancer-binding factor 1 (Lef-1) differentially regulates the proximal promoters of Dsc2 and Dsc3 23724138_our study reveals a function for gamma-catenin in the regulation of mESC differentiation and has implications for human cancers in which gamma-catenin is mutated and/or aberrantly expressed. 24994937_EMMPRIN ensures proper actomyosin-driven maturation of competent endothelial junctions by forming a molecular complex with gamma-catenin (also known as junction plakoglobin) and Nm23 (also known as NME1), a nucleoside diphosphate kinase 25705887_In 2 knockin mouse models, endogenous Jup was engineered to express the Naxos-associated form of plakoglobin. Insufficiency of the truncated Naxos plakoglobin, rather than gain of function, accounted for cardiac disease manifestations. 26485505_Mice with hepatic loss of the desmosomal protein gamma-Catenin are prone to cholestatic injury and chemical carcinogenesis. 26676851_Reduced plakoglobin staining in intercalated discs of heart tissue from human arrhythmogenic right ventricular cardiomyopathy (ARVC) patients and in a murine ARVC model is caused by alterations in epitope accessibility and not by protein relocalization 29802338_essential to the maintenance of the structure of the corneal epithelium and its wound healing 30991025_gamma-catenin signaling is involved in the initiation and maintenance of BCR-ABL1(+) B-cell acute lymphoblastic leukemia. 31425296_FLG and DST support melanoma cell growth in vitro and in vivo. Growth effects of JUP were only evident in vivo, and may be mediated, in part, by enhancing angiogenesis. In addition, growth-promoting effects of FLG and DST in vitro suggest that these genes may also support melanoma cell proliferation through angiogenesis-independent pathways. 32820720_beta-catenin and gamma-catenin are dispensable for T lymphocytes and AML leukemic stem cells. 35054841_Premature Termination Codon in 5' Region of Desmoplakin and Plakoglobin Genes May Escape Nonsense-Mediated Decay through the Reinitiation of Translation. 36577382_CYLD deubiquitinates plakoglobin to promote Cx43 membrane targeting and gap junction assembly in the heart. |
ENSG00000173801 |
JUP |
169.207885 |
0.4380347380 |
-1.190883 |
0.32339085 |
13.136351 |
0.00028962134423941138759717173201124751358292996883392333984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0015457296129834316184697984297713446721900254487991333007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
104.451064898312 |
23.6100525757361 |
238.453839025269 |
39.4667431329038 |
| ENSMUSG00000001655 |
15422 |
Hoxc13 |
protein_coding |
P50207 |
FUNCTION: Transcription factor which plays a role in hair follicle differentiation. Regulates FOXQ1 expression and that of other hair-specific genes. {ECO:0000269|PubMed:11290294, ECO:0000269|PubMed:16835220}. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15422; |
nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; anatomical structure morphogenesis [GO:0009653]; anterior/posterior pattern specification [GO:0009952]; hair follicle development [GO:0001942]; nail development [GO:0035878]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; tongue morphogenesis [GO:0043587] |
15385554_Krtap16 is a hair keratin-associated protein gene complex regulated by Hoxc13 16382673_We provide evidence for specific interaction of Hoxc13 with at least one cognate binding site found in the Crisp1 promoter region. 16835220_satin hair mutant gene Foxq1 is among multiple and functionally diverse regulatory targets for Hoxc13 during hair follicle differentiation 21191399_data support a regulatory model of keratinocyte differentiation in which HOXC13-dependent activation of Foxn1 is part of a regulatory cascade controlling the expression of hair and nail terminal differentiation markers 25691658_YAP regulates the expression of Hoxa1 and Hoxc13 in oral and dental epithelial tissues as well as in the epidermis of skin during embryonic and adult stages. 26321246_a potential regulatory relationship between the sterol O-acetyltransferase 1 gene and Hoxc13 gene 26553656_data strongly suggest that Hoxc13 is a novel and crucial regulator of the hair cycle. 27506447_Here we demonstrate that the transcription factors SPI1 (PU.1) and HOXC13 synergistically regulate Zfp521 expression, and identify the regions of the Zfp521 promoter required for this transcriptional activity. We also show that SPI1 and HOXC13 activate Zfp521 in a dose-dependent manner. |
ENSG00000123364 |
HOXC13 |
95.265706 |
7.7599547346 |
2.956048 |
0.31658416 |
87.413849 |
0.00000000000000000000880319086946567187998062583796287576893251577723738002744478803318628479246399365365505218505859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000003538000087696599357909158229111348350489759444629150669445194488105244090547785162925720214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
169.744328687249 |
37.5482594139509 |
21.874396757836 |
4.02021055007566 |
| ENSMUSG00000001864 |
108897 |
Aif1l |
protein_coding |
Q9EQX4 |
FUNCTION: Actin-binding protein that promotes actin bundling. May neither bind calcium nor depend on calcium for function (By similarity). {ECO:0000250}. |
Acetylation;Actin-binding;Calcium;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:108897; |
actin cytoskeleton [GO:0015629]; actin filament [GO:0005884]; cytoplasm [GO:0005737]; focal adhesion [GO:0005925]; protein-containing complex [GO:0032991]; ruffle membrane [GO:0032587]; actin filament binding [GO:0051015]; calcium ion binding [GO:0005509]; actin filament bundle assembly [GO:0051017]; ruffle assembly [GO:0097178] |
Human_homologues 18699778_Structure of human Iba2 and its homodimerization properties, F-actin cross-linking activity, cellular localization and recruitment upon bacterial invasion in comparison with Iba1. 20237496_Observational study of gene-disease association. (HuGE Navigator) 29726059_We did not find the AIF1L-ETV6 and ABL1-AIF1L fusions in other ETV6-ABL1-positive ALL. Nevertheless, functional studies would be needed to establish the biological role of AIF1L-ETV6 and ABL1-AIF1L and to determine whether they contribute to leukemogenesis and/or to the final leukemia phenotype. 30001384_these findings indicate an involvement of AIF1L in the stabilization of podocyte morphology by titrating actomyosin contractility and membrane dynamics. |
ENSG00000126878 |
AIF1L |
549.434118 |
5.3638219317 |
2.423261 |
0.23183311 |
99.153680 |
0.00000000000000000000002336483983383625087513055037258337641246029441101630365371051101588961707022917835274711251258850097656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000011443512551414590358985588876706427661245851690657515938400780319827987341341213323175907135009765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
910.173830220884 |
146.890179778146 |
169.687555218079 |
20.9106396650828 |
| ENSMUSG00000001870 |
268977 |
Ltbp1 |
protein_coding |
Q8CG19 |
FUNCTION: Key regulator of transforming growth factor beta (TGFB1, TGFB2 and TGFB3) that controls TGF-beta activation by maintaining it in a latent state during storage in extracellular space. Associates specifically via disulfide bonds with the Latency-associated peptide (LAP), which is the regulatory chain of TGF-beta, and regulates integrin-dependent activation of TGF-beta. Outcompeted by LRRC32/GARP for binding to LAP regulatory chain of TGF-beta. {ECO:0000250|UniProtKB:Q14766}. |
Alternative splicing;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Growth factor binding;Hydroxylation;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:268977; |
collagen-containing extracellular matrix [GO:0062023]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; large latent transforming growth factor-beta complex [GO:0038045]; microfibril [GO:0001527]; mitochondrion [GO:0005739]; neuronal cell body [GO:0043025]; perinuclear region of cytoplasm [GO:0048471]; protein-containing complex [GO:0032991]; calcium ion binding [GO:0005509]; microfibril binding [GO:0050436]; transforming growth factor beta binding [GO:0050431]; aorta development [GO:0035904]; coronary vasculature development [GO:0060976]; positive regulation of collagen biosynthetic process [GO:0032967]; regulation of transforming growth factor beta activation [GO:1901388]; sequestering of TGFbeta in extracellular matrix [GO:0035583]; transforming growth factor beta receptor signaling pathway [GO:0007179]; ventricular septum development [GO:0003281] |
12962157_IGFBP-3 can bind to LTBP-1 and provide a potential mechanism whereby IGFBP-3 can interact with the TGF-beta system 15677465_fibronectin has a role in LTBP1 assembly in the ECM and in regulation of TGF beta via LTBP1 interactions 16260650_integrin alphavbeta6-mediated activation of latent TGF-beta complexes containing LTBP-1 requires fibronectin 17015622_Cleavage of LTBP1 by BMP1 is involved in TGFbeta1 activation. 17804598_Data show that the lack of Ltbp1L in the extracellular matrix of the septating cardiac outflow tract and associated vessels results in altered gene expression and function of cardiac neural crest cells and decreased Tgf-beta activity in the OFT. 17804598_The long form of LTBP1 (LTBP-1L) is essential for proper heart development. 17950478_LTBP-1 has essential functions in the control of TGF-beta activation 18508077_AhR regulates Ltbp-1 transcription by a mechanism involving recruitment of co-activators such as CREB1 and co-repressors such as HDAC2 to the Ltbp-1 promoter. 18516051_Blocking Tgfb-Smad2/3 innate immune signaling mitigates Alzheimer-like pathology. 18672106_When TGF-beta activity would inhibit further maturation and mineralization, LTBP-1 containing large latent TGF-beta1 complexes that accumulate into the ECM and represent targets for osteoclast mediated release and activation of TGF-beta in bone tissue. 19022904_Data suggest that association of latent TGF-beta binding protein (LTBP) with the latent TGF-beta complex is important for proper TGF-beta1 function. 20565961_Shows that Latent Tgfbeta Binding protein 1 (LTBP1), which is a key extracellular modulator of Tgfbeta ligand bioavailability, is coexpressed with Tgfbetas in the early limb bud. 21181942_Ltbp1L is a major regulator of Tgf-beta activity during valvulogenesis since its absence results in a perturbed Tgf-beta pathway that causes all Ltbp1L(-/-) valvular defects. 21422469_requirement for TGF-beta signaling into T cells to achieve peripheral CD8 but not CD4 T-cell tolerance 22036565_Study reports that master transcription factors also are responsible for directing the gene targets of TGF-beta signaling and thus determine the cell-type-specific effects of TGF-beta signaling. 22045186_results suggest a role for VEGFA-driven tumour growth by TGF-beta signalling inhibition via paracrine mechanisms in vivo 23183523_miR-133a mediates TGF-beta-dependent derepression of collagen synthesis in hepatic stellate cells during liver fibrosis. 23462118_High transforming Growth Factor beta1 expression is associated with myelofibrosis. 23645881_Induction of both Th17-producing cells and Tregs is caused preferentially by Tregs expressing the latent TGF-beta1/GARP complex on their cell surface rather than by secreted latent TGF-beta1. 23804438_a detailed computational model for TGF-beta signalling that incorporates elements of previous models together with crosstalking between Smad1/5/8 and Smad2/3 channels through a negative feedback loop dependent on Smad7. 24262428_Ltbp1 functions in a mechanosensory capacity to establish and maintain mammary gland ductal luminal cell fate, support and detect ductal distension, trigger irreversible involution, and facilitate nipple sphincter function. 25805620_Data show the complete knockout of both isoforms of LTBP-1 leads to defective cardiovascular development and subsequent perinatal lethality supporting the idea that LTBP-1 is essential in the development of the great arteries and cardiac cushions. 27956181_LTBP1 has a role in female reproduction 31343988_Fibulin-1c regulates transforming growth factor-beta activation in pulmonary tissue fibrosis. |
ENSG00000049323 |
LTBP1 |
2346.545420 |
0.2129381405 |
-2.231494 |
0.70932025 |
9.075245 |
0.00259092830313050652771278414832067937823012471199035644531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0106202959938869415795759110210383369121700525283813476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
882.743815741448 |
451.71118509503 |
4145.54111235594 |
1559.35707165759 |
| ENSMUSG00000001942 |
22619 |
Siae |
protein_coding |
P70665 |
FUNCTION: Catalyzes the removal of O-acetyl ester groups from position 9 of the parent sialic acid, N-acetylneuraminic acid. |
Alternative splicing;Cytoplasm;Disulfide bond;Glycoprotein;Hydrolase;Lysosome;Reference proteome;Serine esterase;Signal |
|
|
mmu:22619; |
lysosome [GO:0005764]; sialate 4-O-acetylesterase activity [GO:0106331]; sialate 9-O-acetylesterase activity [GO:0106330]; sialate O-acetylesterase activity [GO:0001681]; carbohydrate metabolic process [GO:0005975]; regulation of immune system process [GO:0002682] |
19103880_Sialate:O-acetyl esterase is a catalytic regulator of B cell signaling and plays an essential role in the maintenance of immunological tolerance in the B lineage. |
ENSG00000110013 |
SIAE |
1035.273106 |
4.2293812690 |
2.080447 |
0.64495707 |
8.710853 |
0.00316321237563515567439709208485965064028277993202209472656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0125213230004352117641364117162083857692778110504150390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1725.11982238504 |
797.161423923807 |
407.889408090638 |
139.051599678898 |
| ENSMUSG00000002020 |
16997 |
Ltbp2 |
protein_coding |
E9Q1D6 |
Human_homologues FUNCTION: May play an integral structural role in elastic-fiber architectural organization and/or assembly. {ECO:0000303|PubMed:10743502, ECO:0000303|PubMed:11104663}. |
Disulfide bond;EGF-like domain;Extracellular matrix;Proteomics identification;Reference proteome;Repeat;Secreted;Signal |
|
|
|
calcium ion binding [GO:0005509] |
17581631_These results suggest a novel regulatory mechanism of elastic fiber assembly in which LTBP-2 regulates targeting of DANCE on suitable microfibrils to form elastic fibers. 21700711_Latent transforming growth factor beta-binding proteins-2 and -3 inhibit the proprotein convertase 5/6A. 24867584_perlecan HS was not essential for latent transforming growth factor-beta-1 binding protein-2 deposition 24908666_LTBP-2 is an essential component for the formation of microfibril bundles in ciliary zonules. 28252045_These data suggest that LTBP-2 and -4 have critical overlapping functions in forming the robust structure of microfibrils in vitro and in vivo. 33039488_Latent-transforming growth factor beta-binding protein-2 (LTBP-2) is required for longevity but not for development of zonular fibers. |
ENSG00000119681 |
LTBP2 |
700.421430 |
0.4528223083 |
-1.142983 |
0.12056795 |
88.670536 |
0.00000000000000000000466359993125141137964647204223690094639849520667453695399549268296723880666831973940134048461914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001919890652778959317956693499180803577502175120660641606529228653243990265764296054840087890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
426.068266655009 |
35.6485571210772 |
940.917129861997 |
57.8373507181549 |
| ENSMUSG00000002055 |
54141 |
Spag5 |
protein_coding |
Q7TME2 |
FUNCTION: Essential component of the mitotic spindle required for normal chromosome segregation and progression into anaphase. Required for chromosome alignment, normal timing of sister chromatid segregation, and maintenance of spindle pole architecture. In complex with SKAP, promotes stable microtubule-kinetochore attachments. May contribute to the regulation of separase activity. May regulate AURKA localization to mitotic spindle, but not to centrosomes and CCNB1 localization to both mitotic spindle and centrosomes. Involved in centriole duplication. Required for CDK5RAP22, CEP152, WDR62 and CEP63 centrosomal localization and promotes the centrosomal localization of CDK2. In non-mitotic cells, upon stress induction, inhibits mammalian target of rapamycin complex 1 (mTORC1) association and recruits the mTORC1 component RPTOR to stress granules (SGs), thereby preventing mTORC1 hyperactivation-induced apoptosis. May enhance GSK3B-mediated phosphorylation of other substrates, such as MAPT/TAU (By similarity). {ECO:0000250|UniProtKB:Q96R06}. |
Cell cycle;Cell division;Centromere;Chromosome;Coiled coil;Cytoplasm;Cytoskeleton;Kinetochore;Microtubule;Mitosis;Phosphoprotein;Reference proteome |
|
|
mmu:54141; |
centriolar satellite [GO:0034451]; ciliary basal body [GO:0036064]; ciliary rootlet [GO:0035253]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; kinetochore [GO:0000776]; microtubule plus-end [GO:0035371]; midbody [GO:0030496]; mitotic spindle [GO:0072686]; mitotic spindle pole [GO:0097431]; nuclear body [GO:0016604]; spindle [GO:0005819]; microtubule binding [GO:0008017]; cell division [GO:0051301]; chromosome segregation [GO:0007059]; establishment of spindle orientation [GO:0051294]; mitotic sister chromatid segregation [GO:0000070]; positive regulation of intracellular transport [GO:0032388]; positive regulation of spindle assembly [GO:1905832]; protein localization to centrosome [GO:0071539]; regulation of attachment of spindle microtubules to kinetochore [GO:0051988]; regulation of metaphase plate congression [GO:0090235]; spindle organization [GO:0007051] |
12893248_cloning and genomic structure of mastrin; mastrin protein was demonstrated to localize to mitotic spindles during mitosis[masatrin] 19806032_Loss-of-function of Astrin by RNAi and overexpression of the coiled-coil domain results in spindle disorganization, chromosome misalignment and meiosis progression arrest. |
ENSG00000076382 |
SPAG5 |
986.747348 |
2.1170580554 |
1.082061 |
0.14606272 |
53.571950 |
0.00000000000024929348590783644082543539550619065588285203083351859731919830664992332458496093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000053709170825292291935695330601815121202707858927283268712926656007766723632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
1335.64125511894 |
135.455409719026 |
630.894959028103 |
48.3325490878731 |
| ENSMUSG00000002105 |
68427 |
Slc39a13 |
protein_coding |
Q8BZH0 |
FUNCTION: Acts as a zinc-influx transporter. {ECO:0000250}. |
Alternative splicing;Golgi apparatus;Ion transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport;Zinc;Zinc transport |
|
|
mmu:68427; |
endoplasmic reticulum [GO:0005783]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; membrane [GO:0016020]; perinuclear region of cytoplasm [GO:0048471]; protein homodimerization activity [GO:0042803]; zinc ion transmembrane transporter activity [GO:0005385]; cellular zinc ion homeostasis [GO:0006882]; connective tissue development [GO:0061448]; zinc ion transmembrane transport [GO:0071577] |
18985159_The Slc39a13-KO mouse represents a novel animal model linking zinc metabolism, BMP/TGF-beta signaling and connective tissue dysfunction. 26050750_This study concluded that skin fragility due to defective ZIP13 protein may be attributable to impaired extracellular matrix synthesis accompanied by abnormal peripheral TGF-beta homeostasis. 28854265_Both gain- and loss-of-function studies showed that an accumulation of the CCAAT/enhancer binding protein-beta (C/EBP-beta) protein, which cooperates with dominant transcriptional co-regulator PR domain containing 16 (PRDM16) to determine brown/beige adipocyte lineage, is essential for the enhanced adipocyte browning caused by the loss of ZIP13 31412620_we have clarified the functional differences between ZIP7 and ZIP13 and show that their different regulation of cellular zinc homeostasis is critical for dermal development and homeostasis. 31636298_Loss of the dermis zinc transporter ZIP13 promotes the mildness of fibrosarcoma by inhibiting autophagy. 33307093_Downregulation of the zinc transporter SLC39A13 (ZIP13) is responsible for the activation of CaMKII at reperfusion and leads to myocardial ischemia/reperfusion injury in mouse hearts. |
ENSG00000165915 |
SLC39A13 |
3215.853363 |
0.4711004383 |
-1.085893 |
0.05656110 |
364.716344 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000264625666762033111038213081123168160709802650801440134211794174762342354417816948455742336485548760738108684659672709950049646605988373783878757067596226028565822352989028446478593530 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000009831166234437287833540245237393466666830312174677072177573599866373021042548499576013643895989228898441140714930843396978196352785921711560861076571032795415871091222939531226578793 |
Yes |
No |
2047.64038280945 |
80.2986559635855 |
4346.50494082564 |
124.294481463303 |
| ENSMUSG00000002804 |
66174 |
Nudt14 |
protein_coding |
Q9D142 |
FUNCTION: Hydrolyzes UDP-glucose to glucose 1-phosphate and UMP and ADP-ribose to ribose 5-phosphate and AMP. The physiological substrate is probably UDP-glucose. Poor activity on other substrates such as ADP-glucose, CDP-glucose, GDP-glucose and GDP-mannose (By similarity). {ECO:0000250}. |
Cytoplasm;Hydrolase;Magnesium;Reference proteome |
|
|
mmu:66174; |
cytoplasm [GO:0005737]; ADP-ribose diphosphatase activity [GO:0047631]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; UDP-sugar diphosphatase activity [GO:0008768]; nucleoside phosphate metabolic process [GO:0006753]; ribose phosphate metabolic process [GO:0019693] |
19896456_Nudt14 catalyzed the conversion of [beta-32P]UDP-glucose to [32P]glucose-1-P and UMP, confirming that it hydrolyzed the pyrophosphate of the nucleoside diphosphate sugar to generate glucose-1-P and UMP. |
ENSG00000183828 |
NUDT14 |
108.736237 |
2.4318501533 |
1.282054 |
0.24454591 |
27.200979 |
0.00000018336578147438533038977599015639796675714023876935243606567382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000018472404652234375593031039108904423073909129016101360321044921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
158.757472504741 |
27.0394453276014 |
65.2825883579082 |
8.4694594579563 |
| ENSMUSG00000002831 |
57435 |
Plin4 |
protein_coding |
O88492 |
FUNCTION: May play a role in triacylglycerol packaging into adipocytes. May function as a coat protein involved in the biogenesis of lipid droplets. {ECO:0000269|PubMed:12840023, ECO:0000269|PubMed:15731108}. |
Alternative splicing;Cell membrane;Cytoplasm;Lipid droplet;Membrane;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:57435; |
cytosol [GO:0005829]; lipid droplet [GO:0005811]; plasma membrane [GO:0005886] |
12840023_Adipocyte protein S3-12 coats nascent lipid droplets. 15111493_Direct regulation of perilipin and S3-12 by PPAR-gamma may to be important mediator of prolonged treatment with PPAR-gamma activators: insulin sensitization, fatty acid trapping in adipose tissue, reduced basal adipose lipolysis, and weight gain. 15731108_the non-lipid droplet pools of S3-12, adipophilin, and TIP47 constitute a ready reservoir of coat proteins to permit rapid packaging of newly synthesized triacylglycerol and to maximize energy storage during nutrient excess 23423172_Loss of Plin4 has no effect on body weight or composition or on adipose mass or development. Triacylglycerol (TAG) content in the heart is markedly reduced. The heart of Plin4(-/-) mice displays reduced Plin5 mRNA and protein levels. 33242187_Perilipin 4 Protein: an Impending Target for Amyotrophic Lateral Sclerosis. |
ENSG00000167676 |
PLIN4 |
188.556298 |
2.7094084472 |
1.437978 |
0.21664339 |
43.301101 |
0.00000000004693194310366349914587943405719068588194398117252603697124868631362915039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000007753442053741892627554563155033596349996471985832613427191972732543945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
267.089618302701 |
40.2586203417623 |
98.5785729659988 |
11.588477393382 |
| ENSMUSG00000002885 |
26364 |
Adgre5 |
protein_coding |
A0A0D9SEG8 |
Human_homologues FUNCTION: Receptor potentially involved in both adhesion and signaling processes early after leukocyte activation. Plays an essential role in leukocyte migration. {ECO:0000250|UniProtKB:Q9Z0M6}. |
Cell membrane;Disulfide bond;EGF-like domain;G-protein coupled receptor;Glycoprotein;Membrane;Proteomics identification;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:26364; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; G protein-coupled receptor activity [GO:0004930]; cell surface receptor signaling pathway [GO:0007166] |
14707087_In vivo findings in experimental colitis and pneumococcal pneumonia indicate an essential role for CD97 in the migration of neutrophils. 17158902_CD97 plays a role in peripheral granulocyte homeostasis by functioning in an adhesive capacity. 18326527_Mobilization was absent and neutrophils were reduced in mice receiving CD97 mAb plus interleukin-8, while granulocyte-colony stimulating factor-induced mobilization remained unaltered by anti-CD97. CD97 has a role in IL-8 induced HSC/HPC mobilization. 18603564_In BALB/c mice, CD97 expression can be applied to almost completely separate colony-forming cells and cells exhibiting radioprotective capacity. 18941248_Despite the broad expression pattern of wild-type murine CD97, the successfully deleted gene Cd97-deficient mouse that is created has no overt phenotype, except for a mild granulocytosis that is normal in the absence of CD97. 20131275_Insight into a role for CD55 interaction with CD97 in the pathogenesis of rheumatoid arthritis suggest that therapeutic strategies that disrupt CD55/CD97 may be clinically beneficial. 23447688_CD55 downregulates CD97 surface expression on circulating leukocytes by a process that requires physical forces. 23676664_Intestinal enlargement by CD97 requires its seven-span transmembrane/cytoplasmic C-terminal fragment. 24949957_We conclude that CD97 is located in the SR and at the peripheral sarcolemma of human and murine skeletal muscle, where its absence affects the structure of the SR without impairing skeletal muscle function 26663852_These findings demonstrate that CD97 is a positive regulator of osteoclast-like cell differentiation, a mechanism that influences bone homeostasis. However, the presence of CD97 may be essential to suppress the initial osteoclastogenesis that occurs in response to acute and local inflammatory stimuli. 34298973_How to Obtain a Mega-Intestine with Normal Morphology: In Silico Modelling of Postnatal Intestinal Growth in a Cd97-Transgenic Mouse. 34314469_CD97 stabilises the immunological synapse between dendritic cells and T cells and is targeted for degradation by the Salmonella effector SteD. 35143305_CD97 promotes spleen dendritic cell homeostasis through the mechanosensing of red blood cells. |
ENSG00000123146 |
ADGRE5 |
126.082052 |
0.4684153233 |
-1.094140 |
0.31082448 |
12.163747 |
0.00048727388319455821339212797660422893386567011475563049316406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0024479405635948253347478065222730947425588965415954589843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
77.5203133490082 |
16.8310347565921 |
165.494827973536 |
26.2967786916047 |
| ENSMUSG00000002980 |
57278 |
Bcam |
protein_coding |
Q9R069 |
FUNCTION: Laminin alpha-5 receptor. May mediate intracellular signaling (By similarity). {ECO:0000250}. |
Cell adhesion;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:57278; |
cell surface [GO:0009986]; plasma membrane [GO:0005886]; laminin binding [GO:0043236]; laminin receptor activity [GO:0005055]; protein C-terminus binding [GO:0008022]; cell adhesion [GO:0007155]; cell-matrix adhesion [GO:0007160] |
18032551_Lu/BCAM, as a laminin receptor, is involved in vivo in the maintenance of normal basement membrane organization in the kidney and intestine. 24036115_Lutheran (Lu), also known as basal cell adhesion molecule (B-CAM), competes with integrins for binding to laminin alpha5, a subunit of LM-511, a major component of basement membranes. 24429403_Lu/BCAM may have a critical role in facilitating the accumulation of monocytes and macrophages, thereby exacerbating renal injury in crescentic glomerulonephritis. 30059007_Lutheran (Lu)/Basal cell adhesion molecule (BCAM) regulates the morphogenesis of DR depending on liver disease models. |
ENSG00000187244 |
BCAM |
11.491668 |
0.0768057219 |
-3.702642 |
0.96116396 |
15.962711 |
0.00006460252137593014334221441785643946786876767873764038085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004019712441168986696848897111067344667389988899230957031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.63422213627297 |
1.1711143532398 |
21.277348816069 |
8.50379356733765 |
| ENSMUSG00000003070 |
13637 |
Efna2 |
protein_coding |
P52801 |
FUNCTION: Cell surface GPI-bound ligand for Eph receptors, a family of receptor tyrosine kinases which are crucial for migration, repulsion and adhesion during neuronal, vascular and epithelial development. Binds promiscuously Eph receptors residing on adjacent cells, leading to contact-dependent bidirectional signaling into neighboring cells. The signaling pathway downstream of the receptor is referred to as forward signaling while the signaling pathway downstream of the ephrin ligand is referred to as reverse signaling. With the EPHA2 receptor may play a role in bone remodeling through regulation of osteoclastogenesis and osteoblastogenesis. {ECO:0000269|PubMed:19299512, ECO:0000269|PubMed:9053851}. |
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:13637; |
neuromuscular junction [GO:0031594]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; ephrin receptor binding [GO:0046875]; axon guidance [GO:0007411]; bone remodeling [GO:0046849]; ephrin receptor signaling pathway [GO:0048013]; olfactory bulb development [GO:0021772]; osteoclast differentiation [GO:0030316] |
14988728_Tiam1 is required for neurite outgrowth induced by both ephrin-B1-mediated reverse signaling and EphA2-mediated forward signaling 15710390_ephrin-A2 expression was observed in synapses during reactive synaptogenesis 15713841_identification of ephrin-A2 and EphA7 as negative regulators of progenitor cell proliferation reveals a novel mechanism to control cell numbers in the brain 16025107_In mice deficient for ephrin-A2, A3 and A5, eye-specific inputs segregated but shape and location of eye-specific layers were profoundly disrupted. Ephrin-As and neural activity act together to control patterning of eye-specific retinogeniculate layers. 18632942_Misalignment of ipsilateral and contralateral visual information in the superior colliculus results in inappropriate terminations in ephrin-A2/A5(-/-) double knock-out mice and suggests a role for ephrin-As in integrating convergent visual inputs. 19299512_Bidirectional ephrinA2-EphA2 signaling regulates bone remodeling at the initiation phase. 20100519_The results of this study suggested that the misexpression of ephrin-A2 may lead to abnormal connectivity in regions known for their involvement in reversal learning and perseverative behaviours. 22247258_Implicate EphA2 as a novel proinflammatory mediator and potential regulator of atherosclerotic plaque development. 22903544_EphA2-mediated mesenchymal-amoeboid transition induced by endothelial progenitor cells enhances metastatic spread due to cancer-associated fibroblasts. 24094103_ephrin-A2 regulates experience-dependent, NMDA receptor-mediated synaptic pruning through glial glutamate transport during maturation of the mouse cortex. 24381199_In EphA4 mutant mice, as well as after ephrin A2 knockdown using in utero electroporation, we found delayed interneuron migration at embryonic stages. 24457350_At high frequencies, the ephrin-A2A5(-/-) mice exhibited thresholds that were significantly lower than in wild-type mice by approximately 20 dB, suggesting ephrin-A2 and ephrin-A5 may have frequency-specific effects on the auditory system. 25200515_The results of this study suggested that majority of the changes observed ephrin-A2 and ephrin-A5 KO mice appear to be mediated by the effects on motor neurons and their muscle targets, rather than changes in auditory sensitivity. 25243788_Ephrin-A2 and Ephrin-A5 are important for the functional development of cutaneous innervation. 25269910_Ephrin-A2 and -A3 are negative regulators of the proliferative and neurogenic potentials of Muller cells. 25281279_The ephrin-A2/-A3 DKO mice have utility as a novel ASD model with an emphasis on sensory abnormalities and restricted, repetitive behavioral symptoms. 28924206_ephrin-A2 participates in the creation of excitatory neurons by inhibiting apical progenitor proliferation in the ventricular zone, with the disruption of ephrin-A2 signaling in these cells recapitulating the abnormal neocortex observed in the knockout. 29464193_Study showed that in comparison with wild-type mice revealed that ephrin-A2A5-/- mice had reduced accuracy and response rates, suggesting a goal-directed behavioral deficit, which was improved by low intensity repetitive transcranial magnetic stimulation. 30482614_The loss of Ephrin-A2 and A5 ligands did not impact on the rate of wound closure or re-innervation after injury. However, changes in the gross morphology of the healed scar and in collagen histology of the scar dermis were observed in transgenic mice. Therefore Ephrin-A2 and A5 ligands may play an important role in final scar appearance associated with collagen deposition and structure. 33181182_Concurrent LI-rTMS induces changes in c-Fos expression but not behavior during a progressive ratio task with adult ephrin-A2A5(-/-) mice. |
ENSG00000099617 |
EFNA2 |
116.168495 |
0.3609887192 |
-1.469974 |
0.24199887 |
36.451232 |
0.00000000156534181424746707521184230655622235395263430746126687154173851013183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000214611039735530335103643481773488810304684193397406488656997680664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
62.1521935806401 |
10.4744199224763 |
172.172121379267 |
20.4104814701201 |
| ENSMUSG00000003134 |
54610 |
Tbc1d8 |
protein_coding |
Q9Z1A9 |
FUNCTION: May act as a GTPase-activating protein for Rab family protein(s). |
GTPase activation;Reference proteome;Repeat |
|
|
mmu:54610; |
GTPase activator activity [GO:0005096]; activation of GTPase activity [GO:0090630] |
Human_homologues 17686842_isolated a TBC Rab-GAP cellular binding partner interacting with the N terminus of HCV NS5A; depleting the expression of this partner severely impairs HCV RNA replication with no obvious effect on cell viability 20548944_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 30809301_TBC1D8 drives ovarian cancer tumorigenesis and metabolic reprogramming and serves as an independent prognosis factor for ovarian cancer patients |
ENSG00000204634 |
TBC1D8 |
28.039067 |
3.3277712575 |
1.734556 |
0.63651015 |
7.213612 |
0.00723527254735370928956372793550144706387072801589965820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0255051310903243941274887163217499619349837303161621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.9221699877311 |
19.5574231055141 |
12.8981731814621 |
4.55782625397713 |
| ENSMUSG00000003153 |
20527 |
Slc2a3 |
protein_coding |
P32037 |
FUNCTION: Facilitative glucose transporter that can also mediate the uptake of various other monosaccharides across the cell membrane. Mediates the uptake of glucose, 2-deoxyglucose, galactose, mannose, xylose and fucose, and probably also dehydroascorbate. Does not mediate fructose transport. {ECO:0000250|UniProtKB:P11169}. |
Cell membrane;Cell projection;Direct protein sequencing;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Sugar transport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:20527; |
acrosomal membrane [GO:0002080]; caveola [GO:0005901]; cell projection [GO:0042995]; cytoplasm [GO:0005737]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; synaptic membrane [GO:0097060]; synaptic vesicle membrane [GO:0030672]; D-glucose transmembrane transporter activity [GO:0055056]; dehydroascorbic acid transmembrane transporter activity [GO:0033300]; galactose transmembrane transporter activity [GO:0005354]; galactoside binding [GO:0016936]; glucose binding [GO:0005536]; glucose transmembrane transporter activity [GO:0005355]; hexose transmembrane transporter activity [GO:0015149]; kinase binding [GO:0019900]; monosaccharide transmembrane transporter activity [GO:0015145]; xylose binding [GO:0033222]; dehydroascorbic acid transport [GO:0070837]; galactose transmembrane transport [GO:0015757]; glucose import [GO:0046323]; glucose transmembrane transport [GO:1904659]; monosaccharide transmembrane transport [GO:0015749]; negative regulation of apoptotic process [GO:0043066] |
15054091_phosphorylated CREB and Sp3 induce GLUT 3 expression in response to development/cell differentiation and neurotransmission 16365304_crosstalk between an imprinted growth demand gene (Igf2) and placental supply transporter genes (Slc38a4, Slc38a2, and Slc2a3) may be a component of the genetic control of nutrient supply and demand during mammalian development 17213475_Trophectodermal deficiency of GLUT3 adversely affects in vivo embryonic development in th mouse. 17983831_Chondrocytes have the capacity to detect and respond to low oxygen availability with changes in expression of oxygen-regulated genes. Hypoxia regulation of facilitated GLUT-1 and -3 was mediated by HIF-1alpha. 18445753_Male GLUT3(+/-) mice expressed decreased insulin-responsive GLUT4 in white adipose tissue and skeletal muscle sarcolemma. Authors conclude that the GLUT3(+/-) male mice develop adult-onset adiposity with insulin resistance. 18780771_A single allele of Slc2a3 is sufficient for maintenance of neuronal energy supply, motor abilities, learning and memory, and feeding behavior. 18948350_GLUT3 is essential for the development of early post-implanted embryos. 19327547_upregulated in activated T cells; associated with increase in Vitamin C uptake, mostly in oxidized form 19506559_This is the first description of GLUT3 deficiency that forms a possible novel genetic mechanism for pervasive developmental disorders, such as the neuropsychiatric autism spectrum disorders 19550108_Both transcripts and protein synthesis of the glucose transporters Glut1 and Glut3 were reduced in blastocysts cultured in the presence of either 25 or 55 mM glucose 19841136_GLUT1 and -3 are mislocalized from the apical plasma membrane in embryonic epithelia 21316350_This study demonistrated that Glut3 haploinsufficiency does not impair brain glucose uptake or utilization. 22383310_Hesperetin and hesperidin downregulate Akt, GLUT3, and GLUT4 of the insulin signaling pathway in Abeta1-42-induced Neuro-2A cells. 22700768_In glut3(+/-) mice, a key role of placental Glut3 in mediating transplacental and intraplacental glucose transport was established. 23193055_These observations collectively support a temporal contribution by transcription toward ensuring adequate tissue-specific, developmental (placenta and embryonic brain), and postnatal hypoxic brain GLUT3 expression. 23321477_Data suggest that co-activation of CREST (calcium-responsive transactivator) and CBP (CREB-binding protein) enhances signaling between p-Creb/AP-1 and p-HIF-1/HRE resulting in up-regulation of Glut3 gene; here, stimulus was cell hypoxia. 23493374_Recruitment of Creb1-Mecp2 by glut3-(m)CpG contributes towards transactivation, formulating an escape from (m)CpG-induced gene suppression, and thereby promoting developmental neuronal glut3 gene transcription and expression. 23615220_knock-down of glucose transporter 3 in embryonic stem cells impaired the beating function of ESC-derived cardiomyocytes, suggesting its potential role in mediating stem cell differentiation 24243341_GLUT3 levels in the brains of scrapie-infected animals was significantly downregulated. 24370435_Placental endoplasmic reticulum stress by administration of Tun causes downregulation of Slc2a1(GLUT1) and upregulation of Slc2a3(GLUT3) mRNA expression. 25526803_Results define Glut3 to be a rab11-dependent trafficking cargo and suggest that impaired Glut3 trafficking arising from rab11 dysfunction underlies the glucose hypometabolism observed in Huntington's disease 25578782_Glut3 is a downstream target of mTORC1, and it is critical for oncogenic mTORC1-mediated aerobic glycolysis and tumorigenesis. 26339590_Morphological changes and GLUT1, GLUT3, and GLUT4 expression were evaluated in placentas by immunohistochemical and image analysis and correlated with iAs and arsenical species concentration, which were quantified by atomic absorption spectroscopy 28471544_Neonatal hypothyroridism downregulates the expressions of GLUT3 and GLUT8 in the testis of perpubertal mice. 28663252_GLUT3-mediated glucose utilization and glycogenolysis in platelets promotes alpha-granule release, platelet activation, and postactivation functions. 29582588_Study found no differences in mRNA or protein levels of neuronal monocarboxylate transporters (MCTs). Functional analyses revealed that neuronal MCT2 had high catalytic efficiency in Huntington's disease (HD) cells. Ascorbic acid did not stimulate lactate uptake in HD cells; and was unable to inhibit glucose transport in HD cells because they exhibit decreased expression of the neuronal glucose transporter GLUT3. 29614685_1) that decrease of GLUT3 is associated with the reduction of protein O-GlcNAcylation in Alzheimer's disease (AD)brain, 2) that GLUT3 level is negatively correlated with calpain I activation in human brain, 3) that calpain I proteolyzes GLUT3 at the N-terminus in vitro, and 4) that activation of calpain I is negatively correlated with protein O-GlcNAcylation in AD brain. 30232223_Lack of Glut3 during early postnatal life, results in a reduction in dendritic spines and brain size, along with a shortened lifespan. 31561359_Regular Exercise Enhances Cognitive Function and Intracephalic GLUT Expression in Alzheimer's Disease Model Mice. 31817208_GLUT3 is essential for breast cancer cell survival in the brain. 31882430_Diabetes-induced hyperglycemia reduces the capability of ovarian follicle cells by down-regulating Glut3 expression. 31884568_Sevoflurane Induces Learning and Memory Impairment in Young Mice Through a Reduction in Neuronal Glucose Transporter 3. 33368883_CD205(+) polymorphonuclear myeloid-derived suppressor cells suppress antitumor immunity by overexpressing GLUT3. 33482226_Adult glut3 homozygous null mice survive to demonstrate neural excitability and altered neurobehavioral responses reminiscent of neurodevelopmental disorders. 33586490_High NOV/CCN3 expression during high-fat diet pregnancy in mice affects GLUT3 expression and the mTOR pathway. 33774619_Developing Brain Glucose Transporters, Serotonin, Serotonin Transporter, and Oxytocin Receptor Expression in Response to Early-Life Hypocaloric and Hypercaloric Dietary, and Air Pollutant Exposures. 34035344_Analysis of glucose metabolism by (18)F-FDG-PET imaging and glucose transporter expression in a mouse model of intracerebral hemorrhage. 35077394_Recurrent moderate hypoglycemia accelerates the progression of Alzheimer's disease through impairment of the TRPC6/GLUT3 pathway. 35316657_The glucose transporter GLUT3 controls T helper 17 cell responses through glycolytic-epigenetic reprogramming. |
|
|
234.024340 |
5.9967311744 |
2.584176 |
0.32407440 |
58.016543 |
0.00000000000002599227380736352343887739093182888353575831982822386123643809696659445762634277343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000006225067840153476874909692840349480929835329634514096142083872109651565551757812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
367.841848262168 |
83.3254297269206 |
61.3403932186284 |
10.8594262881429 |
| ENSMUSG00000003283 |
15162 |
Hck |
protein_coding |
P08103 |
FUNCTION: Non-receptor tyrosine-protein kinase found in hematopoietic cells that transmits signals from cell surface receptors and plays an important role in the regulation of innate immune responses, including neutrophil, monocyte, macrophage and mast cell functions, phagocytosis, cell survival and proliferation, cell adhesion and migration. Acts downstream of receptors that bind the Fc region of immunoglobulins, such as FCGR1A and FCGR2A, but also CSF3R, PLAUR, the receptors for IFNG, IL2, IL6 and IL8, and integrins, such as ITGB1 and ITGB2. During the phagocytic process, mediates mobilization of secretory lysosomes, degranulation, and activation of NADPH oxidase to bring about the respiratory burst. Plays a role in the release of inflammatory molecules. Promotes reorganization of the actin cytoskeleton and actin polymerization, formation of podosomes and cell protrusions. Inhibits TP73-mediated transcription activation and TP73-mediated apoptosis. Phosphorylates CBL in response to activation of immunoglobulin gamma Fc region receptors. Phosphorylates ADAM15, BCR, ELMO1, FCGR2A, GAB1, GAB2, RAPGEF1, STAT5B, TP73, VAV1 and WAS (By similarity). {ECO:0000250, ECO:0000269|PubMed:10547366, ECO:0000269|PubMed:10799548, ECO:0000269|PubMed:12235133, ECO:0000269|PubMed:14551197, ECO:0000269|PubMed:16809022, ECO:0000269|PubMed:17513616, ECO:0000269|PubMed:18246197, ECO:0000269|PubMed:18625913, ECO:0000269|PubMed:19897576, ECO:0000269|PubMed:19903482, ECO:0000269|PubMed:8125254, ECO:0000269|PubMed:9400828, ECO:0000269|PubMed:9916742}. |
Alternative initiation;ATP-binding;Cell junction;Cell membrane;Cell projection;Cytoplasm;Cytoplasmic vesicle;Cytoskeleton;Exocytosis;Golgi apparatus;Immunity;Inflammatory response;Innate immunity;Kinase;Lipoprotein;Lysosome;Membrane;Myristate;Nucleotide-binding;Nucleus;Palmitate;Phagocytosis;Phosphoprotein;Proto-oncogene;Reference proteome;SH2 domain;SH3 domain;Transferase;Tyrosine-protein kinase;Ubl conjugation |
|
The protein encoded by this gene is a member of the Src family of tyrosine kinases. This protein is primarily hemopoietic, particularly in cells of the myeloid and B-lymphoid lineages. It may play a role in the innate immune response and the STAT5 signaling pathway. Alternative translation initiation site usage, including a non-AUG (CUG) codon, results in the production of two different isoforms, that have different subcellular localization. [provided by RefSeq, Feb 2010]. |
mmu:15162; |
actin filament [GO:0005884]; caveola [GO:0005901]; cell projection [GO:0042995]; cytosol [GO:0005829]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; focal adhesion [GO:0005925]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; lysosome [GO:0005764]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; transport vesicle [GO:0030133]; ATP binding [GO:0005524]; lipid binding [GO:0008289]; non-membrane spanning protein tyrosine kinase activity [GO:0004715]; phosphotyrosine residue binding [GO:0001784]; protein tyrosine kinase activity [GO:0004713]; signaling receptor binding [GO:0005102]; cell differentiation [GO:0030154]; defense response to Gram-positive bacterium [GO:0050830]; exocytosis [GO:0006887]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; localization [GO:0051179]; negative regulation of apoptotic process [GO:0043066]; peptidyl-tyrosine phosphorylation [GO:0018108]; phagocytosis [GO:0006909]; positive regulation of actin cytoskeleton reorganization [GO:2000251]; positive regulation of cell population proliferation [GO:0008284]; protein autophosphorylation [GO:0046777]; regulation of cell shape [GO:0008360]; regulation of DNA-binding transcription factor activity [GO:0051090]; regulation of phagocytosis [GO:0050764]; regulation of podosome assembly [GO:0071801]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
1875927_Alternate protein isoforms arise through the use of a non-AUG (CUG), and a downstream in-frame AUG, translation initiation codons, in mouse and human. 12208875_enhanced innate immune response in Hck(F/F) mice thereby skewing innate immunity from a reversible physiological host defense response to one causing irreversible tissue damage 12411494_BCR/ABL SH3 and SH2 domains interact with hematopoietic cell kinase (Hck), leading to the stimulation of Hck catalytic activity 15723811_Hck and Fgr function as negative regulators of myeloid cell chemokine signaling by maintaining the tonic phosphorylation of PIR- 16785558_Hck plays no role in chemoattractant-induced, inside-out beta 2 integrin activation of neutrophils but regulates their outside-in, signaling-dependent sustained adhesion. 17500055_de novo actin polymerization process was functionally dependent on the kinase activity of Hck, WASp, the Arp2/3 complex, and Cdc42 but not Rac or Rho. 17513616_Lyn activity is elevated in hck(-/-) cells, leading to increased phosphorylation of several negative regulators 18246197_a myeloproliferative transformation-sensitive signaling pathway, composed of Lyn/Hck, SHIP, autocrine/paracrine cytokines, and Stat5, that regulates HSC differentiation and M2 macrophage programming. 19234535_Hck functions in neutrophils to be realized, at least in part, via its interaction with mDia1 and WASp, and identifies the mDia1/Hck/WASp axis as a cytoskeletal signaling interface. 19897576_spatial organization of podosomes as large rosettes, proteolytic degradation of extracellular matrix, and 3D migration appeared to be functionally linked and regulated by Hck in macrophages 20181660_Taken together, PKR and Hck were critical for DON-induced ribosomal recruitment of p38, its subsequent phosphorylation, and, ultimately, p38-driven proinflammatory cytokine expression. 20488787_Nef participates in HIV-1-induced multinucleated giant cells formation via a p61Hck- and lysosomal enzyme-dependent pathway 21220696_Despite possessing enhanced killing, alveolar macrophage Hck/Fgr/Lyn-deficient ('triple-knockout') mice do not demonstrate enhanced inflammatory responses to Pneumocystis murina. 23742809_Hck is one of the very few effectors of preosteoclast recruitment described to date and thereby plays a critical role in bone remodeling. 24482227_tyrosine phosphorylation of WASP by Hck is required for proper macrophage functions 25225462_The Src family kinases Hck, Fgr, and Lyn are critical for the generation of the in vivo inflammatory environment without a direct role in leukocyte recruitment. 26068045_Hck is not only progressively overexpressed in atherosclerosis, but it also controls critical molecular processes in monocyte influx, blood monocyte subset balance, macrophage accumulation, and the maintenance of atherosclerotic lesion stability. 26232427_Data indicate that Src-Family Kinases Hck and Fgr regulate cytokine secretion by macrophages. 27358479_the Hck, Fgr and Lyn kinases are also necessary for amastigote uptake by macrophages. Src-mediated Arg activation is required for efficient uptake. 27927780_Data suggest that Hck is a key mediator of renal fibrosis 28399411_Hematopoietic Hck promotes tumorigenesis. 29962266_These results suggest that the estrogen-autophagy-STAT3-HCK axis participates in the differentiation of PTGS2(high) IFNG(low) PRF1(low) GZMB(low) FCGR3(-) NK cells in ectopic lesion microenvironment. 31065022_Hemopoietic Cell Kinase amplification with Protein Tyrosine Phosphatase Receptor T depletion leads to polycythemia, aberrant marrow erythoid maturation, and splenomegaly. 31992566_Inhibition of the SRC Kinase HCK Impairs STAT3-Dependent Gastric Tumor Growth in Mice. 32146679_Genetic Ablation of Hematopoietic Cell Kinase Accelerates Alzheimer's Disease-Like Neuropathology in Tg2576 Mice. 32290615_Expression of a Constitutively Active Form of Hck in Chondrocytes Activates Wnt and Hedgehog Signaling Pathways, and Induces Chondrocyte Proliferation in Mice. 34363435_Hematopoietic cell kinase enhances osteosarcoma development via the MEK/ERK pathway. 36223746_Inhibition of HCK in myeloid cells restricts pancreatic tumor growth and metastasis. |
ENSG00000101336 |
HCK |
11.308554 |
6.5512556318 |
2.711771 |
0.82996570 |
10.908642 |
0.00095716675898750984852886292131302070629317313432693481445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0044477010594562993439637921255780383944511413574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.6733399351944 |
11.3449921636153 |
2.85034518336897 |
1.4366085848576 |
| ENSMUSG00000003352 |
12297 |
Cacnb3 |
protein_coding |
P54285 |
FUNCTION: Regulatory subunit of the voltage-gated calcium channel that gives rise to L-type calcium currents (PubMed:24751537). Increases CACNA1B peak calcium current and shifts the voltage dependencies of channel activation and inactivation (By similarity). Increases CACNA1C peak calcium current and shifts the voltage dependencies of channel activation and inactivation (By similarity). {ECO:0000250|UniProtKB:P54287, ECO:0000250|UniProtKB:Q9MZL3, ECO:0000269|PubMed:24751537}. |
Alternative splicing;Calcium;Calcium channel;Calcium transport;Cytoplasm;Ion channel;Ion transport;Phosphoprotein;Reference proteome;SH3 domain;Transport;Voltage-gated channel |
|
|
mmu:12297; |
apical plasma membrane [GO:0016324]; cytoplasm [GO:0005737]; L-type voltage-gated calcium channel complex [GO:1990454]; membrane [GO:0016020]; plasma membrane [GO:0005886]; synapse [GO:0045202]; voltage-gated calcium channel complex [GO:0005891]; calcium channel regulator activity [GO:0005246]; high voltage-gated calcium channel activity [GO:0008331]; protein kinase binding [GO:0019901]; voltage-gated calcium channel activity [GO:0005245]; calcium ion transmembrane transport [GO:0070588]; calcium ion transmembrane transport via high voltage-gated calcium channel [GO:0061577]; calcium ion transport [GO:0006816]; calcium ion transport into cytosol [GO:0060402]; chemical synaptic transmission [GO:0007268]; detection of mechanical stimulus involved in sensory perception of pain [GO:0050966]; negative regulation of detection of mechanical stimulus involved in sensory perception of touch [GO:1905788]; negative regulation of voltage-gated calcium channel activity [GO:1901386]; neuromuscular junction development [GO:0007528]; positive regulation of excitatory postsynaptic potential [GO:2000463]; positive regulation of high voltage-gated calcium channel activity [GO:1901843]; positive regulation of protein targeting to membrane [GO:0090314]; positive regulation of voltage-gated calcium channel activity [GO:1901387]; protein localization to plasma membrane [GO:0072659]; regulation of membrane hyperpolarization [GO:1902630]; regulation of membrane repolarization during action potential [GO:0098903]; regulation of voltage-gated calcium channel activity [GO:1901385]; T cell receptor signaling pathway [GO:0050852] |
12054671_Mice with cardiac-specific sequestration of the beta-subunit of the L-type calcium channel 15479643_Data show that the calcium channel beta3 subunit negatively modulates inositol triphosphate-induced calcium release. 16822546_Down-regulation of Cacnb3 expression does not rescue placental hyperplasia in a model of interspecies hybrid placentas, indicaating that upregulation is a downstream event. 17335778_the involvement of voltage-dependent Ca2+ channels and importance of the beta3 subunit in olfactory signal transduction 17580090_Ca(V)beta(3) has only subtle effects on L-type Ca(2+) currents in ileum smooth muscle. 17588550_This study showed the importance of voltage-dependent calcium channels and the key role of the beta3 subunit in memory formation, nociceptive sensory transduction, and various neurological signal transduction pathways. 18339621_These results suggest a possibility that, unrelated to voltage-dependent Ca(2+) channels regulation, Ca(v)beta3 negatively regulates the NMDAR activity in the hippocampus. 18628210_Modified sympathetic nerve system activity with overexpression of the voltage-dependent calcium channel beta3 subunit.( 19755851_neither Ca(V)beta3 nor Ca(V)beta4 are indispensable for hair cell Ca(2+) currents but contribute to the overall current properties 19767875_In transgenic mice lacking the calcium channel beta3 subunit, there are compensatory changes of calcium channel abundance leading to adaptation of alternate calcium transport mechanisms. 19838200_Deficiency in beta3 regulatory subunit of voltage-gated calcium channels resulted in a profound survival defect of CD8(+) T cells. 20144588_these results indicate the importance of the beta3 subunit in determining calcium channel characteristics and catecholamine release in adrenal chromaffin cells. 21040760_Differential gene expression analysis identifies murine Cacnb3 as strongly upregulated in distinct dendritic cell populations upon stimulation 22737983_During development Cavbeta3 protein expression in the cerebellum peaks at postnatal days 15 and 21, and has diminished drastically by day 60, and in the hippocampus increases with age throughout all subfields. 29386119_our results indicate that beta3, unrelated to its role as a Ca2+ channel subunit, specifically modulates the cell's IP3 sensitivity and thereby efficiently tunes agonist-induced cytoplasmic Ca2+ signals and cellular functions. |
ENSG00000167535 |
CACNB3 |
1479.901504 |
0.4543006489 |
-1.138281 |
0.07760979 |
212.780064 |
0.00000000000000000000000000000000000000000000000339901581244673853775406231009591206296343204146006286350719441153817185152136715683093636792788369723250689465511117817557440456965878183837048709392547607421875000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000005283041719917216247858625495508299494146589595150631972485033288177646638467617281270839882029930618557638461774172197044086018991038145031780004501342773437500000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
913.447587543982 |
49.1626210376784 |
2010.66758286864 |
78.5897458878247 |
| ENSMUSG00000003411 |
69908 |
Rab3b |
protein_coding |
Q9CZT8 |
FUNCTION: Protein transport. Probably involved in vesicular traffic (By similarity). {ECO:0000250}. |
Acetylation;Cell membrane;Direct protein sequencing;Golgi apparatus;GTP-binding;Lipoprotein;Membrane;Methylation;Nucleotide-binding;Phosphoprotein;Prenylation;Protein transport;Reference proteome;Transport |
|
|
mmu:69908; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; dopaminergic synapse [GO:0098691]; endosome [GO:0005768]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; synaptic vesicle [GO:0008021]; synaptic vesicle membrane [GO:0030672]; vesicle [GO:0031982]; GDP binding [GO:0019003]; GTP binding [GO:0005525]; GTP-dependent protein binding [GO:0030742]; GTPase activity [GO:0003924]; myosin V binding [GO:0031489]; antigen processing and presentation [GO:0019882]; peptidyl-cysteine methylation [GO:0018125]; positive regulation of dopamine uptake involved in synaptic transmission [GO:0051586]; protein localization to plasma membrane [GO:0072659]; protein secretion [GO:0009306]; regulation of exocytosis [GO:0017157]; regulation of synaptic vesicle cycle [GO:0098693]; regulation of vesicle size [GO:0097494]; vesicle docking involved in exocytosis [GO:0006904] |
16436611_Rab3-induced modifications to primed vesicles causes a transient increase in the transduction efficacy of synaptic action potential trains and optimizes the encoding of synaptic information at an intermediate spike frequency range. 18396146_Gas8 was specifically interacted with the GTP-bound form of Rab3B and co-localized with Rab3B at the Golgi in NIH 3T3 cells 19717423_Rab3b/3c-positive recycling vesicles are involved in and may constitute one of the recycling compartments in exogenous antigen cross-presentation. 21844341_Presynaptic forms of long-term plasticity at excitatory and inhibitory synapses generally are mediated by a common Rab3/RIM-dependent pathway, with various types of synapses using distinct Rab3 isoforms. |
ENSG00000169213 |
RAB3B |
37.111514 |
3.3383694256 |
1.739144 |
0.43427091 |
15.889610 |
0.00006714633225265703839104020067551914507930632680654525756835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004159304322376868967396645881962058410863392055034637451171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
54.5105197155771 |
16.6573280117715 |
16.3284863856279 |
3.98096502759793 |
| ENSMUSG00000003526 |
19125 |
Prodh |
protein_coding |
Q9WU79 |
FUNCTION: Converts proline to delta-1-pyrroline-5-carboxylate. |
Acetylation;FAD;Flavoprotein;Mitochondrion;Oxidoreductase;Proline metabolism;Reference proteome;Transit peptide |
PATHWAY: Amino-acid degradation; L-proline degradation into L-glutamate; L-glutamate from L-proline: step 1/2. |
|
mmu:19125; |
intracellular membrane-bounded organelle [GO:0043231]; mitochondrial inner membrane [GO:0005743]; mitochondrial matrix [GO:0005759]; mitochondrion [GO:0005739]; nucleoplasm [GO:0005654]; amino acid binding [GO:0016597]; FAD binding [GO:0071949]; proline dehydrogenase activity [GO:0004657]; positive regulation of cell death [GO:0010942]; proline catabolic process to glutamate [GO:0010133] |
26660760_PRODH/POX and succinate regulated mitochondrial respiration. 27705802_Here, we show that Prodh-deficient mice with elevated CNS L-proline display specific deficits in high-frequency GABA-ergic transmission and gamma-band oscillations. We find that L-proline is a GABA-mimetic and can act at multiple GABA-ergic targets 28685754_PRODH1-mediated proline metabolism promotes pancreatic ductal adenocarcinoma growth. |
ENSG00000277196+ENSG00000100033 |
PRODH+PRODH |
37.968388 |
2.4465460647 |
1.290746 |
0.45012873 |
7.982316 |
0.00472364833576385725194546338912005012389272451400756835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0177261915374119435884203710429574130102992057800292968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.9631979308508 |
16.7942229049545 |
21.6481507113092 |
5.23158209700944 |
| ENSMUSG00000003721 |
72999 |
Insig2 |
protein_coding |
Q91WG1 |
FUNCTION: Oxysterol-binding protein that mediates feedback control of cholesterol synthesis by controlling both endoplasmic reticulum to Golgi transport of SCAP and degradation of HMGCR (PubMed:12242332, PubMed:12624180, PubMed:16100574). Acts as a negative regulator of cholesterol biosynthesis by mediating the retention of the SCAP-SREBP complex in the endoplasmic reticulum, thereby blocking the processing of sterol regulatory element-binding proteins (SREBPs) SREBF1/SREBP1 and SREBF2/SREBP2 (PubMed:16100574). Binds oxysterol, including 22-hydroxycholesterol, 24-hydroxycholesterol, 25-hydroxycholesterol and 27-hydroxycholesterol, regulating interaction with SCAP and retention of the SCAP-SREBP complex in the endoplasmic reticulum (By similarity). In presence of oxysterol, interacts with SCAP, retaining the SCAP-SREBP complex in the endoplasmic reticulum, thereby preventing SCAP from escorting SREBF1/SREBP1 and SREBF2/SREBP2 to the Golgi (By similarity). Sterol deprivation or phosphorylation by PCK1 reduce oxysterol-binding, disrupting the interaction between INSIG2 and SCAP, thereby promoting Golgi transport of the SCAP-SREBP complex, followed by processing and nuclear translocation of SREBF1/SREBP1 and SREBF2/SREBP2 (By similarity). Also regulates cholesterol synthesis by regulating degradation of HMGCR: initiates the sterol-mediated ubiquitin-mediated endoplasmic reticulum-associated degradation (ERAD) of HMGCR via recruitment of the reductase to the ubiquitin ligase RNF139 (By similarity). {ECO:0000250|UniProtKB:Q9Y5U4, ECO:0000269|PubMed:12242332, ECO:0000269|PubMed:12624180, ECO:0000269|PubMed:16100574}. |
Cholesterol metabolism;Endoplasmic reticulum;Lipid metabolism;Lipid-binding;Membrane;Oxidation;Phosphoprotein;Reference proteome;Steroid metabolism;Sterol metabolism;Thioester bond;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:72999; |
endoplasmic reticulum [GO:0005783]; SREBP-SCAP-Insig complex [GO:0032937]; oxysterol binding [GO:0008142]; cellular response to insulin stimulus [GO:0032869]; cholesterol biosynthetic process [GO:0006695]; cholesterol metabolic process [GO:0008203]; cranial suture morphogenesis [GO:0060363]; inner ear morphogenesis [GO:0042472]; middle ear morphogenesis [GO:0042474]; negative regulation of fatty acid biosynthetic process [GO:0045717]; negative regulation of steroid biosynthetic process [GO:0010894]; response to fatty acid [GO:0070542]; response to sterol depletion [GO:0006991]; roof of mouth development [GO:0060021]; SREBP signaling pathway [GO:0032933]; SREBP-SCAP complex retention in endoplasmic reticulum [GO:0036316]; sterol biosynthetic process [GO:0016126]; triglyceride metabolic process [GO:0006641] |
12624180_Liver-specific mRNA for Insig-2 down-regulated by insulin: implications for fatty acid synthesis. 15528275_a vitamin D response element in the murine Insig-2 promoter may have a role in the differentiation of 3T3-L1 preadipocytes 16100574_Results define Insig proteins 1 and 2 as essential elements of the feedback inhibition system of cholesterol synthesis in mouse liver. 16126366_Insig2 identified as a strong candidate susceptibility gene for total plasma cholesterol levelsin inbred mice strains 16955138_Treatment of pregnant mice with the HMG-CoA reductase inhibitor lovastatin reduced sterol synthesis in Insig2-knockout embryos and reduced the pre-cholesterol intermediates ameliorating the clefting. 17440045_A critical gene involved in cholesterol homeostasis, Insig-2, was induced when mice or cultured cells were treated with FXRalpha agonists or infected with constitutively active FXRalpha. 19110480_Peroxisome deficiency causes a complex phenotype because of hepatic SREBP-1c,-2/Insig-2a,b dysregulation associated with endoplasmic reticulum stress 19360322_Silibinin inhibits adipocyte differentiation through a potential up-regulation of insig-1 and insig-2 at an early phase in adipocyte differentiation. 19721697_Data show that REV-ERBalpha participates in the circadian modulation of SREBP activity, and the expression of SREBP target genes involved in cholesterol and lipid metabolism via the cyclic transcription of Insig2. 20090767_Insig deficiency in skin causes accumulation of cholesterol precursors, and this impairs normal hair development 24337570_the essential role of Insig1,2 proteins in the sterol homeostasis of enterocytes. 27568546_INSIG2 is a negative regulator of SREBP, and acute glucocorticoid treatment decreased active SREBP during refeeding or in livers of Ob/Ob mice, both systems of elevated SREBP-1c-driven lipogenesis. 28416613_These results indicate that HIF-mediated induction of Insig-2 and degradation of HMGCR are physiologically relevant events that guard against wasteful oxygen consumption and inappropriate cell growth during hypoxia. 28855656_These results suggest that PPARalpha is a trans-acting factor that enhances Insig2a gene expression, thereby suppressing SREBP-1c processing during fasting. 29679236_CRTC2 modulates hepatic SREBP1c cleavage by controlling Insig2a expression during fasting 29899135_Intracellular biosynthesis of lipids and cholesterol by Scap and Insig in mesenchymal cells regulates long bone growth and chondrocyte homeostasis. 31953408_Insig-2 is an ER membrane protein negatively controlling lipid biosynthesis. The stabilized Insig-2 downregulates lipogenesis through inhibiting the SREBP pathway, helping to channel the carbon flux to ATP generation and protecting myotubes from lipid over-accumulation. 34974159_CD36 promotes de novo lipogenesis in hepatocytes through INSIG2-dependent SREBP1 processing. |
ENSG00000125629 |
INSIG2 |
1823.647547 |
0.4085097840 |
-1.291557 |
0.12594861 |
102.409720 |
0.00000000000000000000000451480860498882595782341781784052972463204275145488160917410383823900055233480088645592331886291503906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000002339100839156115387130219216938092432930205757787676857883366228485488846899897907860577106475830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1044.1314888377 |
91.2694749268983 |
2555.95221858756 |
165.448774907625 |
| ENSMUSG00000003779 |
19348 |
Kif20a |
protein_coding |
P97329 |
FUNCTION: Mitotic kinesin required for chromosome passenger complex (CPC)-mediated cytokinesis. Following phosphorylation by PLK1, involved in recruitment of PLK1 to the central spindle (By similarity). Interacts with guanosine triphosphate (GTP)-bound forms of RAB6A and RAB6B. May act as a motor required for the retrograde RAB6 regulated transport of Golgi membranes and associated vesicles along microtubules. Has a microtubule plus end-directed motility. {ECO:0000250, ECO:0000269|PubMed:9438855}. |
3D-structure;Acetylation;ATP-binding;Coiled coil;Cytoplasm;Cytoskeleton;Golgi apparatus;Microtubule;Motor protein;Nucleotide-binding;Phosphoprotein;Protein transport;Reference proteome;Transport |
|
|
mmu:19348; |
Golgi apparatus [GO:0005794]; intercellular bridge [GO:0045171]; kinesin complex [GO:0005871]; microtubule [GO:0005874]; midbody [GO:0030496]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; protein kinase binding [GO:0019901]; microtubule bundle formation [GO:0001578]; microtubule-based movement [GO:0007018]; midbody abscission [GO:0061952]; mitotic cytokinesis [GO:0000281]; protein transport [GO:0015031]; regulation of cytokinesis [GO:0032465] |
30006548_It has been reported that mitotic kinesin KIF20A interacts with RGS3 and plays a crucial role in controlling the division modes of neural progenitor cells during cortical neurogenesis. 31813992_SEPT7 Interacts with KIF20A and Regulates the Proliferative State of Neural Progenitor Cells During Cortical Development. 31884069_KIF20A binds the microtubules to organize the central spindle assembly during spermatogenesis. 33976373_Depletion of kinesin motor KIF20A to target cell fate control suppresses medulloblastoma tumour growth. |
ENSG00000112984 |
KIF20A |
1568.708091 |
2.2419485233 |
1.164753 |
0.07671544 |
227.408886 |
0.00000000000000000000000000000000000000000000000000218995382398325223681069971098558312413085186850779504537334106448595281904780780902231316064316514920168620884658106027826048934660507772065329845645464956760406494140625000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000003924397252577987628542064051694215059512795765887682552470393842761028800542471960607655958080105586044664415396529256297537477382597614905535010620951652526855468750000000000000000000000000000000000000000000000 |
Yes |
No |
2197.2585251194 |
116.894490412831 |
980.066447680765 |
40.1660222695309 |
| ENSMUSG00000004508 |
14389 |
Gab2 |
protein_coding |
Q3ZB57 |
Human_homologues FUNCTION: Adapter protein which acts downstream of several membrane receptors including cytokine, antigen, hormone, cell matrix and growth factor receptors to regulate multiple signaling pathways. Regulates osteoclast differentiation mediating the TNFRSF11A/RANK signaling. In allergic response, it plays a role in mast cells activation and degranulation through PI-3-kinase regulation. Also involved in the regulation of cell proliferation and hematopoiesis. {ECO:0000269|PubMed:15750601, ECO:0000269|PubMed:19172738}. |
Proteomics identification;Receptor;Reference proteome |
|
|
|
cytoplasm [GO:0005737]; plasma membrane [GO:0005886]; transmembrane receptor protein tyrosine kinase adaptor activity [GO:0005068]; osteoclast differentiation [GO:0030316]; positive regulation of cell population proliferation [GO:0008284] |
11701952_comparative FISH mapping of Gab1 and Gab2 genes in human, mouse and rat 11830491_Transfected Gab2 is a limiting signaling component for Erk MAP kinase activation and terminal differentiation of K562 CML cells. 11861309_Requirement of Gab2 for mast cell development and KitL/c-Kit signaling. 11895767_role in beta1-integrin signaling pathway hematooietic stem cell adhesion and migration 12058075_distinct recruitment and function in Met receptor-mediated epithelial morphogenesis 12640133_Gads/Grb2-mediated LAT association is critical for the inhibitory function of Gab2 12821647_Gab2 is recruited to the nascent phagosome, where de novo PI3K lipid production occurs. Gab2 recruitment requires the pleckstrin homology domain of Gab2. 14656892_data propose that Lyn, not Jak2, phosphorylates Gab2 and that maximal phosphorylation of Gab2 requires Y764, a Grb2-binding site. 15750601_Inactivation of the gene encoding Gab2 in mice results in osteopetrosis and decreased bone resorption as a result of defective osteoclast differentiation. 16009726_Gab2 is an important player in neural differentiation, partly by acting downstream of bFGF to mediate survival through phosphoinositide 3 kinase-AKT 16275030_These findings suggest that once Gab-2 is tyrosine phosphorylated its pleckstrin homology (PH) domain is required for the optimal stabilisation of the signalsome, enabling full activation of downstream signals. 16369543_GAB2 may play a role in development of mammary cancer 16456001_Grb2 SH3 binding sites play a critical role in bringing Gab2 to FcepsilonRI, whereupon Gab2 becomes tyrosyl-phosphorylated in a Syk-dependent fashion 16873377_Gab2 via Shp-2 is critical for transmitting signals from Kit Tyr(567) to activate the Rac/JNK pathway controlling mast cell proliferation 17053833_in osteoclasts, phospholipase C, gamma 2 formed a complex with the regulatory adapter molecule GAB2, was required for GAB2 phosphorylation, and modulated GAB2 recruitment to receptor activator of NF-kappaB 17310989_Gab2(-/-) mammary tumor cancer cells exhibited decreased migration and impaired Erk activation. 17323176_Gab2 plays opposite roles in osteoclastogenesis during the phases of skeletal development and maintenance. 17353274_Data demonstrate that Stat3 is required for the erythropoietin-independent growth of Friend virus-infected cells and that the activation of Stat3 by Sf-Stk is mediated by a novel Stat3 binding site in Gab2. 17374739_Gab2 adapter function is intrinsically required for hematopoietic cell response to early-acting cytokines, resulting in defective hematopoiesis in Gab2-deficient mice. 17571162_in the NRG-1beta/ErbB signaling, Gab1 and Gab2 of the myocardium are essential for both maintenance of myocardial function and stabilization of cardiac capillary and endocardial endothelium in the postnatal heart 18586679_Cn dephosphorylates Gab2, resulting in c-fos activation and cell proliferation. We also report that there is a direct interaction between Cn and Gab2 upon IL-3 stimulation, and Akt can regulate this interaction 18697750_activation of PI3K by c-Kit is dependent both on the direct PI3K-binding site in c-Kit and on the phosphorylation of Gab2. 18950707_overexpression of wild-type growth factor receptor bound protein 2 in A20 cells suppressed Fas-mediated apoptosis, and enhanced B-cell mediated rescue from Fas-induced cell death. 19233262_These results underscore the non-redundant and essential roles of Gab1 and Gab2 in endothelial cells, and suggest major contributions of these proteins during in vivo angiogenesis. 20161778_Data indicate that fetal liver cells isolated from homozygous STAT5 mutant mice lacking Gab2 showed significant reduction in HSC number and survival. 20335178_The limited contribution of Fyn and Gab2 to the high affinity IgE receptor signaling in mast cells. 21930791_Our data provide genetic and biochemical evidence that CSF-1R, through Gab2, utilizes different effectors at different stages of MNP development to promote their expansion 22859374_These results define a novel role for Gab2 in mediating mucin gene expression and GCH; these findings have important implications for the pathogenesis and therapy of airway inflammatory diseases. 24284065_These results establish the Frs2alpha-Shp2 complex as the key mediator of FGF signaling in lens development 24805943_Down-regulation of Gab2 has a protective function during M. tuberculosis infection, revealing a potential negative regulatory role for Gab2 in immunity to TB. 26773044_Given that GAB2 is dispensable for normal hematopoiesis, GAB2 and its effectors PI3K and SHP2 represent promising targets for therapy in Ph(+)hematologic neoplasms 27125306_we demonstrated for the first time that Gab2 deficiency has a profound effect on the course of disease in an in vivo chronic myeloid leukemia model 27282405_Gab2 may be a disease-associated protein that is induced by pathogenic factors to amplify and coordinate multifactor-induced signals to govern disease development in the liver. 27840422_High Gab2 expression is associated with myeloproliferative neoplasm. 28687632_Data, including data from studies in transgenic/knockout mice, suggest expression of Gab1/Gab2 is up-regulated in activated macrophages in pulmonary fibrosis; both Gab1/Gab2 are recruited to Il4r, synergistically enhancing downstream signal amplification. (Gab1 = growth factor receptor bound protein 2-associated protein 1; Gab2 = growth factor receptor bound protein 2-associated protein 2; Il4r = interleukin-4 receptor) 28722233_The results of this study showed that the reactive Schwann cells exhibit migratory features dependent on the expression of a scaffolding oncoprotein Grb2-associated binder-2 (Gab2), which was transcriptionally induced by neuregulin 1-ErbB2 signaling following nerve injury. 30816308_Unique properties of PTEN-L contribute to neuroprotection in response to ischemic-like stress. 30936879_Gab2 and Gab3 Redundantly Suppress Colitis by Modulating Macrophage and CD8(+) T-Cell Activation. 32437087_G-CSF-dependent neutrophil differentiation requires downregulation of MAPK activities through the Gab2 signaling pathway. 33637697_Gab2 deficiency suppresses high-fat diet-induced obesity by reducing adipose tissue inflammation and increasing brown adipose function in mice. 33827252_Gab2 (Grb2-Associated Binder2) Plays a Crucial Role in Inflammatory Signaling and Endothelial Dysfunction. 34146905_Scaffolding protein Gab2 is involved in postnatal development and lipopolysaccharide-induced activation of microglia in the mouse brain. 34903841_Gab2 deficiency prevents Flt3-ITD driven acute myeloid leukemia in vivo. 35411035_Liver-specific overexpression of Gab2 accelerates hepatocellular carcinoma progression by activating immunosuppression of myeloid-derived suppressor cells. 35895897_The Gab2-MALT1 axis regulates thromboinflammation and deep vein thrombosis. 36007472_Inhibition of GAB2 expression has a protective effect on osteoarthritis:An in vitro and in vivo study. |
ENSG00000033327 |
GAB2 |
61.197870 |
0.0400806030 |
-4.640952 |
0.50723418 |
100.390627 |
0.00000000000000000000001251187695417155487505467956918212556254624649542385071557528352335919574755962457857094705104827880859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000006228134306076507420071160569751769877244046008588377051189968400257690461785387014970183372497558593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.71850484464547 |
1.69335992336978 |
117.725395596317 |
21.339303221439 |
| ENSMUSG00000004530 |
23790 |
Coro1c |
protein_coding |
Q9WUM4 |
FUNCTION: Plays a role in directed cell migration by regulating the activation and subcellular location of RAC1 (PubMed:25074804, PubMed:25925950). Increases the presence of activated RAC1 at the leading edge of migrating cells (PubMed:25074804, PubMed:25925950). Required for normal organization of the cytoskeleton, including the actin cytoskeleton, microtubules and the vimentin intermediate filaments (PubMed:27178841). Required for normal cell proliferation, cell migration, and normal formation of lamellipodia (PubMed:27178841). Plays a role in endoplasmic reticulum-associated endosome fission: localizes to endosome membrane tubules and promotes recruitment of TMCC1, leading to recruitment of the endoplasmic reticulum to endosome tubules for fission. Endosome membrane fission of early and late endosomes is essential to separate regions destined for lysosomal degradation from carriers to be recycled to the plasma membrane (By similarity). Required for normal distribution of mitochondria within cells (PubMed:27178841). {ECO:0000250|UniProtKB:Q9ULV4, ECO:0000269|PubMed:25074804, ECO:0000269|PubMed:25925950, ECO:0000269|PubMed:27178841}. |
Acetylation;Actin-binding;Cell membrane;Cell projection;Coiled coil;Cytoplasm;Cytoskeleton;Endosome;Membrane;Reference proteome;Repeat;WD repeat |
|
|
mmu:23790; |
actin cytoskeleton [GO:0015629]; cell cortex [GO:0005938]; cytoskeleton [GO:0005856]; endosome membrane [GO:0010008]; flotillin complex [GO:0016600]; lamellipodium [GO:0030027]; lateral plasma membrane [GO:0016328]; ruffle membrane [GO:0032587]; vesicle [GO:0031982]; actin filament binding [GO:0051015]; small GTPase binding [GO:0031267]; actin cortical patch assembly [GO:0000147]; actin cytoskeleton organization [GO:0030036]; actin filament organization [GO:0007015]; activation of GTPase activity [GO:0090630]; cell migration [GO:0016477]; corpus callosum development [GO:0022038]; endosomal transport [GO:0016197]; endosome fission [GO:0140285]; endosome membrane tubulation [GO:0097750]; establishment of protein localization [GO:0045184]; membrane fission [GO:0090148]; negative regulation of epithelial cell migration [GO:0010633]; negative regulation of focal adhesion assembly [GO:0051895]; negative regulation of protein kinase activity by regulation of protein phosphorylation [GO:0044387]; negative regulation of protein phosphorylation [GO:0001933]; negative regulation of substrate adhesion-dependent cell spreading [GO:1900025]; neural crest cell migration [GO:0001755]; phagocytosis [GO:0006909]; positive regulation of lamellipodium morphogenesis [GO:2000394]; regulation of epithelial cell migration [GO:0010632]; regulation of fibroblast migration [GO:0010762]; regulation of focal adhesion assembly [GO:0051893]; regulation of protein phosphorylation [GO:0001932]; regulation of ruffle assembly [GO:1900027]; regulation of substrate adhesion-dependent cell spreading [GO:1900024]; ventricular system development [GO:0021591] |
15813925_Coronin 3 is abundantly expressed in the adult CNS. All murine brain areas express coronin 3 during embryogenesis and the first postnatal stages. 18768935_Results indicate that interaction of GDP-Rab27a and coronin 3 is important in stimulus-endocytosis coupling, and that GTP- and GDP-Rab27a regulates insulin membrane recycling at the distinct stages. 20362548_These results indicate that glucose recruits coronin 3 near the plasma membrane, and that it regulates the retrograde transport of the secretory membrane in pancreatic beta-cells. 22364218_Coronin 1C displays highly co-operative binding to actin filaments. We highlight a novel mode of coronin regulation, which has implications for how coronins orchestrate cytoskeletal dynamics. 25925950_Coronin-1C Protein and Caveolin Protein Provide Constitutive and Inducible Mechanisms of Rac1 Protein 31677819_The results imply that CRN2 represents a pro-invasive effector within the tumour cell microenvironment of glioblastoma multiforme. 32686704_Coronin 1C inhibits melanoma metastasis through regulation of MT1-MMP-containing extracellular vesicle secretion. 35657370_Coro1B and Coro1C regulate lamellipodia dynamics and cell motility by tuning branched actin turnover. |
ENSG00000110880 |
CORO1C |
1091.119100 |
0.4485076750 |
-1.156795 |
0.40419693 |
7.934101 |
0.00485117123845395844389249973005462379660457372665405273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0181199216047402376916775068593778996728360652923583984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
715.031975235132 |
202.960131991258 |
1594.24691040306 |
336.514616626917 |
| ENSMUSG00000004633 |
69993 |
Chn2 |
protein_coding |
Q80XD1 |
FUNCTION: GTPase-activating protein for p21-rac. {ECO:0000250}. |
Alternative splicing;GTPase activation;Membrane;Metal-binding;Reference proteome;Zinc;Zinc-finger |
|
|
mmu:69993; |
membrane [GO:0016020]; synapse [GO:0045202]; GTPase activator activity [GO:0005096]; metal ion binding [GO:0046872]; positive regulation of GTPase activity [GO:0043547]; regulation of GTPase activity [GO:0043087]; signal transduction [GO:0007165] |
20335173_the Rac-GAP beta2-chimaerin is negatively regulated by protein kinase Cdelta-mediated phosphorylation 22726444_Study demonstrates that Sema3F-dependent inhibition of Rac1 by the Rac-GAP beta2Chn is required for hippocampal infrapyramidal tract pruning. 27058424_our data redefine the role of beta2-chimaerin as tumor suppressor and provide the first in vivo evidence of a dual function in breast cancer, suppressing tumor initiation but favoring tumor progression. 31209021_essential for neural stem cell homeostasis in mouse adult neurogenesis |
ENSG00000106069 |
CHN2 |
155.387616 |
2.5449691345 |
1.347648 |
0.27275443 |
23.564814 |
0.00000120775975501110875235115556247667001343870651908218860626220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000106277276651237487551607957536603521475626621395349502563476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
229.531957252936 |
43.6540502651159 |
90.1904679860357 |
13.0992500096167 |
| ENSMUSG00000004661 |
56380 |
Arid3b |
protein_coding |
Q9Z1N7 |
FUNCTION: Transcription factor involved in the production of cranial mesenchymal tissues. Favors nuclear targeting of ARID3A. {ECO:0000269|PubMed:16530748}. |
Acetylation;Alternative splicing;Developmental protein;DNA-binding;Methylation;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:56380; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
16530748_indispensable for production of cranial mesenchymal tissues 16951138_ARID3B seems to play a key role in the malignant transformation of neuroblastoma and may serve as a marker of malignancy and also as a potential target for cancer therapy of stage IV neuroblastoma. 21307092_Arid3b is involved in the regulation of cell motility and rearrangements that lead to apical ectodermal ridge maturation. 22751132_Epigenetic regulation of the neuroblastoma genes, Arid3b and Mycn. 24704276_The different patterns of ARID3B in normal tissues translate into different roles for ARID3B in carcinomas. 27537840_critical for B lymphocyte development |
ENSG00000179361 |
ARID3B |
67.638950 |
0.4780140580 |
-1.064875 |
0.36726698 |
8.066479 |
0.00450916635589870568728843025496644258964806795120239257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0170304046449415054376164135874205385334789752960205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.1641126652825 |
10.8500323397616 |
88.2068465551868 |
16.5726566010808 |
| ENSMUSG00000004698 |
79221 |
Hdac9 |
protein_coding |
A0A1B0GSQ5 |
FUNCTION: Responsible for the deacetylation of lysine residues on the N-terminal part of the core histones (H2A, H2B, H3 and H4). Histone deacetylation gives a tag for epigenetic repression and plays an important role in transcriptional regulation, cell cycle progression and developmental events. {ECO:0000256|PIRNR:PIRNR037911}. |
Chromatin regulator;Hydrolase;Metal-binding;Proteomics identification;Reference proteome;Repressor;Transcription;Transcription regulation;Zinc |
|
|
|
cytoplasm [GO:0005737]; histone deacetylase complex [GO:0000118]; transcription regulator complex [GO:0005667]; histone deacetylase binding [GO:0042826]; histone H4K16 deacetylase activity [GO:0034739]; metal ion binding [GO:0046872]; protein kinase C binding [GO:0005080]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; cellular response to insulin stimulus [GO:0032869]; cholesterol homeostasis [GO:0042632]; chromatin organization [GO:0006325]; histone H3 deacetylation [GO:0070932]; histone H4-K16 deacetylation [GO:1990678]; negative regulation of cytokine production [GO:0001818]; negative regulation of lipoprotein lipase activity [GO:0051005]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuron differentiation [GO:0030182]; peptidyl-lysine deacetylation [GO:0034983]; positive regulation of cell migration involved in sprouting angiogenesis [GO:0090050]; response to amphetamine [GO:0001975] |
12242305_MITR, HDAC4, and HDAC5 associate with heterochromatin protein 1 (HP1), an adaptor protein that recognizes methylated lysines within histone tails and mediates transcriptional repression by recruiting histone methyltransferase 15737621_These results suggest that JMJ represses alphaMHC expression via protein-protein interaction with MEF2A. 17101791_The transcriptional repressor HDAC9 forms a negative-feedback loop in the transcriptional circuitry of muscle differentiation. 18480421_HDRP acts as a negative regulator of the Shh pathway and the absence of HDRP results in hyper-activation of this pathway resulting in polydactyly 19412888_HDAC4, HDAC5, HDAC7 and HDAC9 were found associated with cardiac development. 19879272_Inhibition of HDAC9 increases T regulatory cell function and prevents colitis in mice. 20525066_findings indicate that chromatin remodeling with nucleocytoplasmic translocation of HDAC9 regulates activity-dependent gene expression and dendritic growth in developing cortical neurons 20947501_Histone deacetylase 9 (HDAC9) regulates the functions of the ATDC (TRIM29) protein 21680747_findings provide new insights into mechanisms of adipogenic differentiation and document a critical regulatory role for HDAC9 in adipogenic differentiation through a deacetylase-independent mechanism 21708950_HDAC9 acts as an epigenetic switch in effector T cell-mediated systemic autoimmunity. 21953612_investigation of role of class IIa HDAC isoenzymes in pancreas development: Data suggest HDAC4, -5, and -9 are key regulators controlling the pancreatic beta-/delta-cell lineage. 22226696_HDAC9 is responsible for repressing ChAT gene expression in NG108-15 neuronal cells and thus plays an important role in cholinergic differentiation. 22715468_Data indicate that although histone deacetylases HDAC6, HDAC9, and Sirt1 all deacetylated Foxp3, each protein had different effects on transcription factors that control expression of the gene encoding Foxp3. 23288173_HDAC9 promotes angiogenesis and transcriptionally represses the endothelial cell miR-17-92 cluster. 23297420_Dephosphorylation at a conserved SP motif governs cAMP sensitivity and nuclear localization of class IIa histone deacetylases HDAC4, 5 and 9 24101673_Genetic ablation of HDAC9 improves adipogenic differentiation and systemic metabolic state during a high-fat diet, resulting in diminished weight gain, improved glucose tolerance and insulin sensitivity, and reduced hepatosteatosis. 25035344_HDAC9 deletion decreased atherosclerosis in LDLr(-/-) mice with minimal effect on plasma lipids. Macrophage HDAC9 upregulation is atherogenic via suppression of cholesterol efflux and generation of alternatively activated macrophages in atherosclerosis. 25388417_Compared with HDAC9(+/+)ApoE(-/-) mice, HDAC9(-/-)ApoE(-/-) mice exhibited markedly reduced lesion sizes throughout atherosclerotic aortas and significantly less advanced lesions. 25793404_HDAC9 is a novel, important and physiologically relevant modulator of bone remodeling and skeletal homeostasis. 25873389_Class IIa HDAC9 interact with Class IIb HDAC6 to modulate cell movement and survival in GnRH neurons 26483211_Dach2 and Hdac9 mediate the effects of muscle activity on muscle reinnervation; Myog and Gdf5 appear to stimulate muscle reinnervation through parallel pathways 27033599_Xenograft study in nude mice showed that downregulation of HDAC9 inhibited tumor growth and development in vivo. 27250566_decline in HDAC9c expression over time was accompanied by increased EZH2 expression. 27633396_HDAC9 may be involved in the process of diabetic nephropathy. 28855441_miR-182 upregulates LPL expression, promotes lipid accumulation in atherosclerotic lesions, and increases proinflammatory cytokine secretion, likely through targetingHDAC9, leading to an acceleration of atherogenesis in ApoE-KO mice. 29520069_HDAC9, in cooperation with BRG1 and MALAT1, mediates a critical epigenetic pathway responsible for vascular smooth muscle cells dysfunction. 30031609_HDAC9 depletion resulted in a decreased infarct volume and an improved neurological function in mice after ischemic reperfusion (I/R) injury. 30679521_All our findings revealed that STAT3-inducetd upregulation of lncRNA MEG3 controls cardiac hypertrophy by regulating miR-362-5p/HDAC9 axis. 31320610_Histone deacetylases 9 (HDAC9), a member of the histone deacetylase family, was significantly increased in C2C12 cells under hypoxic conditions, thereby inhibiting intracellular autophagy levels by directly binding to the promoter regions of Atg7, Beclin1, and LC3. 32546048_Histone Deacetylase 9 Activates IKK to Regulate Atherosclerotic Plaque Vulnerability. 32554611_HDAC9 deficiency promotes tumor progression by decreasing the CD8(+) dendritic cell infiltration of the tumor microenvironment. 32620134_Impaired autophagy triggered by HDAC9 in mesenchymal stem cells accelerates bone mass loss. 32702412_Hdac9 inhibits medial artery calcification through down-regulation of Osterix. 34145738_Histone deacetylase 9 deficiency exaggerates uterine M2 macrophage polarization. 34290714_A Biological Circuit Involving Mef2c, Mef2d, and Hdac9 Controls the Immunosuppressive Functions of CD4+Foxp3+ T-Regulatory Cells. 34338228_Histone deacetylase 9 promotes endothelial-mesenchymal transition and an unfavorable atherosclerotic plaque phenotype. 36078104_Sex-Dependent Role of Adipose Tissue HDAC9 in Diet-Induced Obesity and Metabolic Dysfunction. |
ENSG00000048052 |
HDAC9 |
89.678442 |
0.1301641972 |
-2.941595 |
0.60497198 |
19.008282 |
0.00001301522802038606228759633903768744289664027746766805648803710937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000944937813186465724521467812735409097513183951377868652343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.459103587657 |
8.83286057819956 |
157.179193937087 |
48.3569575192327 |
| ENSMUSG00000004709 |
18106 |
Cd244a |
protein_coding |
Q07763 |
FUNCTION: Heterophilic receptor of the signaling lymphocytic activation molecule (SLAM) family; its ligand is CD48. SLAM receptors triggered by homo- or heterotypic cell-cell interactions are modulating the activation and differentiation of a wide variety of immune cells and thus are involved in the regulation and interconnection of both innate and adaptive immune response. Activities are controlled by presence or absence of small cytoplasmic adapter proteins, SH2D1A/SAP and/or SH2D1B/EAT-2. Acts as activating natural killer (NK) cell receptor (PubMed:8326140, PubMed:12734329, PubMed:19648922, PubMed:20962259). Activating function implicates association with SH2D1A and FYN. Downstreaming signaling involves predominantly VAV1, and, to a lesser degree, INPP5D/SHIP1 and CBL. Signal attenuation in the absence of SH2D1A is proposed to be dependent on INPP5D and to a lesser extent PTPN6/SHP-1 and PTPN11/SHP-2. Stimulates NK cell cytotoxicity, production of IFN-gamma and granule exocytosis (PubMed:8326140, PubMed:15169881, PubMed:15998796, PubMed:22683124). Optimal expansion and activation of NK cells seems to be dependent on the engagement of CD244 with CD48 expressed on neighboring NK cells (PubMed:15905190). Regulation of NK cell activity by adapters Sh2d1b and Sh2d1b2 is reported conflictingly (PubMed:16127454, PubMed:16425036). Acts as costimulator in NK activation by enhancing signals by other NK receptors such as NCR3 and NCR1. At early stages of NK cell differentiation may function as an inhibitory receptor possibly ensuring the self-tolerance of developing NK cells (By similarity). Involved in the regulation of CD8(+) T-cell proliferation; expression on activated T-cells and binding to CD488 provides costimulatory-like function for neighboring T-cells (PubMed:11739483). Inhibits inflammatory responses in dendritic cells (DCs) (PubMed:25643613). {ECO:0000250|UniProtKB:Q9BZW8, ECO:0000269|PubMed:11739483, ECO:0000269|PubMed:12734329, ECO:0000269|PubMed:15169881, ECO:0000269|PubMed:15998796, ECO:0000269|PubMed:19648922, ECO:0000269|PubMed:20962259, ECO:0000269|PubMed:22683124, ECO:0000269|PubMed:8326140, ECO:0000305|PubMed:16127454, ECO:0000305|PubMed:16425036}. |
3D-structure;Adaptive immunity;Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Immunity;Immunoglobulin domain;Innate immunity;Membrane;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:18106; |
external side of plasma membrane [GO:0009897]; MHC class I protein binding [GO:0042288]; adaptive immune response [GO:0002250]; innate immune response [GO:0045087]; myeloid dendritic cell activation [GO:0001773]; natural killer cell activation involved in immune response [GO:0002323]; positive regulation of CD8-positive, alpha-beta T cell proliferation [GO:2000566]; positive regulation of granzyme B production [GO:0071663]; positive regulation of inositol phosphate biosynthetic process [GO:0060732]; positive regulation of interleukin-8 production [GO:0032757]; positive regulation of natural killer cell proliferation [GO:0032819]; positive regulation of type II interferon production [GO:0032729] |
11739483_Regulation of CD8(+) T cell proliferation 11815370_Increased blood flow observed in response to topical application of capsaicin involves activation of NK 1 receptors. 12077228_Due to a CxC cysteine motif found in the transmembrane region, 2B4 localizes in glycolipid-enriched microdomains of NK and T cell plasma membranes, where it associates with LAT. 12734329_2B4 represents a novel T cell costimulator that enhances MHC-restricted cytolysis by interacting with CD48+ neighboring T cells. 14666660_Activation of 2B4 reduces metastasis of B16F10 melanoma cells and this anti-tumor effect involves both cytolytic function and cytokine production. 15123744_a novel role for 2B4 as a non-major histocompatibility complex binding negative regulator of NK cells 15356144_2B4 engagement by its counterreceptor CD48 inhibits NK cell cytotoxicity, a function that correlates with increased 2B4 long isoform level expression over 2B4 short isoform. 15634901_Male 2B4-deficient mice show enhanced ability to reject CD48+ melanoma cells. However, female 2B4-deficient mice poorly rejected both CD48+ and CD48- melanoma cells, revealing a gender-specific and CD48-independent defect in mice lacking 2B4. 15778370_Analysis of the ligand-receptor pairs involved in induction of germline transcripts (Igamma2a) necessary for switch recombination revealed that expression of CD48 on B cells and ligands CD2 and CD244 on NK cells are able to stimulate Igamma2a 15870174_compare the relative contributions of MHC class I and CD48 to NK cell self-tolerance; and CD244 regulates NK cell tolerance to normal cells in vivo; evidence for MHC-independent NK self-recognition in a bone marrow rejection assay 15989959_Cell surface receptors of the SLAM family, including CD150, CD244, and CD48, were differentially expressed among functionally distinct hematopoietic progenitor cells. 16081768_Review of recent studies suggests an important role for interactions between 2B4 and CD48 in the course of T cell activation and proliferation 16493031_Expression of CD244 on natural killer (NK) cells is absolutely required for the interaction between CD48 on B cells and CD244 on NK cells, which results in induction of IL-13 mRNA expression. 16621032_REVIEW: different functions of the murine and human 2B4 (CD244) receptor on NK cells 17599905_the mechanism of signal transduction by CD244 is to regulate FYN kinase recruitment and/or activity and the outcome of CD48/CD244 interactions is determined by which other receptors are engaged. 17981603_2B4, NTB-A and CRACC have roles in the regulation of Natural Killer cell function [review] 18056340_In SHIP-/- NK cells there is a pronounced bias toward the 2B4 receptor long isoform. 18523281_Studies under identical and controlled conditions reveal that both human and mouse 2B4 can activate or inhibit natural killer (NK) cells. 19586919_CD244 inhibition and activation depends on CD2 and phospholipase C-gamma1 20363967_The interaction of SHIP, 2B4 activating receptor, and the major histocompatibility complex influence the regulation of natural killer (NK) cell receptor repertoire, cytolytic function, and interferon-gamma production. 20647560_CD48 on surrounding NK or non-NK cells serves primarily as a ligand to stimulate 2B4 on the adjacent NK cells in mice 21221603_Ox-LDL significantly promoted the interaction of natural killer cells and dendritic cells via CD48-2B4 contact-dependent mechanisms. 21622868_Slamf4 has a natural killer (NK) cell-independent negative regulatory function in vivo and can contribute to tolerance to chromatin in murine models of systemic lupus erythematosus. 22683123_In the absence of SAP, signaling through the SLAM family members Ly108 and 2B4 resulted in increased recruitment of the SHP-1 phosphatase, associated with altered SHP-1 localization and decreased activation of Src kinases at the synapse. 24493803_blockade of CD28 signals in the presence of preserved CTLA-4 signals results in the unique up-regulation of 2B4 on primary CD8(+) effectors, and that this 2B4 expression plays a critical functional role in controlling antigen-specific T8 cell responses 24610736_NKG2D ligation relieves 2B4-mediated NK-cell self-tolerance in mice. 24999594_CD244 (2B4) has a regulatory effect on mast cells and eosinophils in murine allergic inflammation. 25142868_Molecular dynamic simulation explains unbinding and unfolding of CD48-2B4 protein complexes. 25643613_DCs from CD244(-/-) mice elicited increased NK cell activation in vitro. These data add CD244 to a growing list of immuno-modulatory receptors found on DCs. 25678452_Signaling via SLAMF4 controls expansion of cytotoxic CD8alphabeta(+) IELs, which regulate the reversible depletion of lamina propria phagocytes and inflammation in the small intestine. 26232435_Data show that enforced expression of inhibitor of differentiation protein Id3 specifically increased the persistence of Cd244 protein 2B4(+) virus-specific CD8(+) T cells. 26323020_2B4 dampens T-independent B cell responses. 28341747_SLAMF4 is a marker of intestinal immune cells which contributes to the protection against enteric pathogens and whose expression is dependent on the presence of the gut microbiota 28768726_2B4 expression is increased on CD4+ T cells in septic animals 30012849_these data indicate that 2B4 signals function to alter T cell glucose metabolism, thereby limiting the proliferation and accumulation of CD8(+) T cells. 30624806_Phenotypic analysis revealed an increased frequency of murine EBV homolog-specific CD8(+) T cells expressing the 2B4 coinhibitory receptor in septic animals compared with sham controls. 31045575_Increased attrition of memory T cells during sepsis requires 2B4. 31723059_2B4 but not PD-1 blockade improves mortality in septic animals with preexisting malignancy. 35061505_SLAMF3 and SLAMF4 are immune checkpoints that constrain macrophage phagocytosis of hematopoietic tumors. 36153598_A novel 2B4 receptor leads to worse pregnancy outcomes by facilitating TNF-alpha and IFN-gamma production in dNK cells during Toxoplasma gondii infection. |
ENSG00000122223 |
CD244 |
79.038649 |
0.4292800969 |
-1.220009 |
0.29012282 |
17.571348 |
0.00002767265369371832526169258104076220661227125674486160278320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001877549492484265178295771425709403956716414541006088256835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
46.0935437656963 |
9.33191301534657 |
107.37405274569 |
15.6621028952887 |
| ENSMUSG00000004864 |
26415 |
Mapk13 |
protein_coding |
Q9Z1B7 |
FUNCTION: Serine/threonine kinase which acts as an essential component of the MAP kinase signal transduction pathway. MAPK13 is one of the four p38 MAPKs which play an important role in the cascades of cellular responses evoked by extracellular stimuli such as pro-inflammatory cytokines or physical stress leading to direct activation of transcription factors such as ELK1 and ATF2. Accordingly, p38 MAPKs phosphorylate a broad range of proteins and it has been estimated that they may have approximately 200 to 300 substrates each. MAPK13 is one of the less studied p38 MAPK isoforms. Some of the targets are downstream kinases such as MAPKAPK2, which are activated through phosphorylation and further phosphorylate additional targets. Plays a role in the regulation of protein translation by phosphorylating and inactivating EEF2K. Involved in cytoskeletal remodeling through phosphorylation of MAPT and STMN1. Mediates UV irradiation induced up-regulation of the gene expression of CXCL14. Plays an important role in the regulation of epidermal keratinocyte differentiation, apoptosis and skin tumor development. Phosphorylates the transcriptional activator MYB in response to stress which leads to rapid MYB degradation via a proteasome-dependent pathway. MAPK13 also phosphorylates and down-regulates PRKD1 during regulation of insulin secretion in pancreatic beta cells. {ECO:0000269|PubMed:19135240, ECO:0000269|PubMed:21558321}. |
ATP-binding;Cell cycle;Kinase;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Stress response;Transcription;Transcription regulation;Transferase |
|
|
mmu:26415; |
cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; ATP binding [GO:0005524]; MAP kinase activity [GO:0004707]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; cell cycle [GO:0007049]; cellular response to anisomycin [GO:0072740]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to interleukin-1 [GO:0071347]; cellular response to sodium arsenite [GO:1903936]; cellular response to sorbitol [GO:0072709]; cellular response to UV [GO:0034644]; intracellular signal transduction [GO:0035556]; peptidyl-serine phosphorylation [GO:0018105]; positive regulation of interleukin-6 production [GO:0032755]; protein phosphorylation [GO:0006468]; response to osmotic stress [GO:0006970]; stress-activated MAPK cascade [GO:0051403] |
11846446_Overexpression of the type II transforming growth factor-beta receptor inhibits fibroblasts proliferation and activates extracellular signal regulated kinase and c-Jun N-terminal kinase 12374793_MKK3 activation is required for subsequent p38alpha and p38delta MAPK activation and collagen stimulation by TGF-beta1 12482858_p38 has a role in mediating induction of VEGF mRNA expression by sodium arsenite 15143069_data suggest role for mitogen-activated protein kinase (MAPK) kinase 3 (MKK3)-p38alpha and p38delta MAPK pathway in mediating vascular endothelial growth factor VEGF164 isoform-specific stimulation by transforming growth factor-beta1 in mesangial cells 16644870_sapk4 activation mediates apoptosis and neurodegeneration in infantile neuronal ceroid lipofuscinosis 18641461_This study constitutes the first analysis addressing the functionality of p38beta, p38gamma and p38delta in satellite cell-dependent adult muscle regeneration and growth. 19135240_Deletion of p38delta results in activation of protein kinase D (PKD), which has been identified as a regulator of stimulated insulin exocytosis; p38delta catalyzes an inhibitory phosphorylation of PKD1, thereby attenuating stimulated insulin secretion. 19458068_An in vivo role for p38delta in promoting cell proliferation and tumor development in epidermis. 19539422_Mouse embryonic stem cells lacking p38delta can differentiate to endothelial cells, smooth muscle cells, and epithelial cells. 21336470_Demonstrate roles of the p38alpha/beta/delta isoforms in the regulation of 12-O-tetradecanoylphorbol 13-acetate-induced skin inflammation. 21558321_results indicate that p38gamma and p38delta have a role in the suppression of tumor development 22733747_p38gamma and p38delta kinases regulate the Toll-like receptor 4 (TLR4)-induced cytokine production by controlling ERK1/2 protein kinase pathway activation 23129748_Aberrant p38delta-PKD1 signaling in neutrophils may underlie development of acute lung injury and life-threatening acute respiratory distress syndrome. 23639288_p27 inhibition of COX-2 expression was specifically due to phosphorylation of transcription factor cAMP response element binding (CREB) phosphorylation mediated by p38beta and p38delta. 23798682_GSK3alpha, GSK3beta, and MAPK13 were found to be the most active tau kinases, phosphorylating tau at all four epitopes. 24782184_p38gamma and p38delta are crucial regulators of inflammatory joint destruction in collagen-induced arthritis. 25217523_Together, our results establish that p38gamma and p38delta are central to colitis-associated colon cancer formation through regulation of hematopoietic cell response to injury, and validate p38gamma and p38kappa as potential targets for cancer therapy. 25753147_Results reveal that p38delta contributes both cell-autonomous and paracrine effects during the course of skin tumor formation. 26795633_p38gamma and p38delta control heart growth by modulating mTOR pathway through DEPTOR phosphorylation and subsequent degradation. 26843485_p38gamma and p38delta reprogram liver metabolism by modulating neutrophil infiltration and provide a potential target for NAFLD therapy 28783172_Study proposes MAPK p38delta protein as a key factor in breast cancer. 29979672_These observations indicate that p38alpha probably blocks brown adipose tissue thermogenesis through p38delta inhibition. 30934690_cell-specific p38delta targeting modifies susceptibility to chemical skin carcinogenesis in a context-, stage-, and sex-specific manner. 32822209_p38delta genetic ablation protects female mice from anthracycline cardiotoxicity. 34758018_p38gamma and p38delta regulate postnatal cardiac metabolism through glycogen synthase 1. |
ENSG00000156711 |
MAPK13 |
7.255994 |
9.9700856496 |
3.317606 |
1.15136727 |
8.545258 |
0.00346423434579616563652737504241940769134089350700378417968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0135222782491063470838854598810030438471585512161254882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.5801215365329 |
11.1399787235442 |
1.2617867066246 |
0.952701554708809 |
| ENSMUSG00000004892 |
12032 |
Bcan |
protein_coding |
Q61361 |
FUNCTION: May play a role in the terminally differentiating and the adult nervous system during postnatal development. Could stabilize interactions between hyaluronan (HA) and brain proteoglycans. |
Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Hyaluronic acid;Immunoglobulin domain;Lectin;Phosphoprotein;Proteoglycan;Reference proteome;Repeat;Secreted;Signal;Sushi |
|
|
mmu:12032; |
axon [GO:0030424]; axon initial segment [GO:0043194]; cell surface [GO:0009986]; dendrite [GO:0030425]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; node of Ranvier [GO:0033268]; perineuronal net [GO:0072534]; perisynaptic extracellular matrix [GO:0098966]; synapse [GO:0045202]; calcium ion binding [GO:0005509]; carbohydrate binding [GO:0030246]; hyaluronic acid binding [GO:0005540]; cell-matrix adhesion [GO:0007160]; central nervous system development [GO:0007417]; hippocampus development [GO:0021766]; neuron projection extension [GO:1990138]; skeletal system development [GO:0001501]; synapse maturation [GO:0060074] |
12370289_Brevican is not essential for learning and memory, but plays a role in long-term potentiation in mice 18221525_Brevican and the ADAMTS-cleaved fragment of brevican were most broadly distributed in neuropil in the brain. 18611854_The proteoglycan brevican binds to fibronectin after proteolytic cleavage and promotes glioma cell motility 19141078_This study show that brevican plays a crucial role in determining the specialization of the hyaluronan-binding nodal matrix assemblies in large diameter nodes. 19683574_Results indicate that reactive gliosis differs between the spinal cord dorsal root entry zone and dorsal column and that Brevican and Versican may participate in the inhibitory properties of the dorsal root entry zone. 20180882_defective brevican processing observed in amyloid beta-bearing mice may contribute to deficient neural plasticity 22121037_these results imply that Bral2-brevican interaction may play a key role in synaptic stabilization and the structural integrity of brain perineuronal nets 23637166_This study provided evidence that the brevican modulates synaptogenesis and long-term synapse stability. The mutant neurons display reduced frequencies of mEPSCs and mIPSCs. 26223835_The extracellular matrix molecule brevican is an integral component of the machinery mediating fast synaptic transmission at the calyx of Held 26711251_This study found that mice with reduced brevican levels show enhanced locomotor activity to acute cocaine and a progressive increase in expression of cocaine CPP during forced abstinence. 28512860_The synaptic plasticity occurs in the hippocampus of freely behaving mice that lack brevican, the informational content stored by synaptic plasticity is not the same. 28695888_intrachromosomal deletion generating a fusion between BCAN and NTRK1 drives the formation of aggressive high-grade gliomas and confers sensitivity to entrectinib 30253762_Genetic deletion of brevican, one of the main components, resulted in a massive degradation of extracellular matrix baskets at inner hair cells, a significant impairment in spatial coupling of pre- and postsynaptic elements and mild impairment of hearing. 31217331_Brevican expression in the auditory cortex is upregulated during consolidation of long-term memories. 31558805_Elimination of the four extracellular matrix molecules tenascin-C, tenascin-R, brevican and neurocan alters the ratio of excitatory and inhibitory synapses. |
ENSG00000132692 |
BCAN |
14.119956 |
0.1571038857 |
-2.670209 |
0.80038097 |
10.680179 |
0.00108289562261329290471234543247192050330340862274169921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0049622942610245716552053174552838754607364535331726074218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.2492362165198 |
1.8965197540457 |
20.6820869031614 |
8.10816604535369 |
| ENSMUSG00000005397 |
18073 |
Nid1 |
protein_coding |
P10493 |
FUNCTION: Sulfated glycoprotein widely distributed in basement membranes and tightly associated with laminin. Also binds to collagen IV and perlecan. It probably has a role in cell-extracellular matrix interactions. |
3D-structure;Basement membrane;Calcium;Cell adhesion;Direct protein sequencing;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Reference proteome;Repeat;Secreted;Signal;Sulfation |
|
|
mmu:18073; |
basement membrane [GO:0005604]; cell periphery [GO:0071944]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; plasma membrane [GO:0005886]; protein complex involved in cell-matrix adhesion [GO:0098637]; calcium ion binding [GO:0005509]; collagen binding [GO:0005518]; extracellular matrix binding [GO:0050840]; laminin binding [GO:0043236]; laminin-1 binding [GO:0043237]; proteoglycan binding [GO:0043394]; cell-matrix adhesion [GO:0007160]; extracellular matrix organization [GO:0030198]; glomerular basement membrane development [GO:0032836]; positive regulation of cell adhesion [GO:0045785]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of integrin-mediated signaling pathway [GO:2001046]; positive regulation of muscle cell differentiation [GO:0051149]; regulation of basement membrane organization [GO:0110011] |
11733994_short arm region of laminin-5 gamma2 chain: structure, mechanism of processing and binding to heparin, nidogen-1, fibulin-1 and fibulin-2 12015298_Specific ablation of the nidogen-binding site in the laminin gamma1 chain interferes with kidney and lung development. 12243745_These findings revealed extensive coregulation of nidogen-1 and -2 expression and much more complementary functions of the two nidogens than previously recognized. 12475645_Three- to sevenfold increase in nidogen-2 was observed in heart and muscle of mice with nidogen-1 deficiency. Nidogen-2 compensation hypothesis to explain limited phenotype observed following elimination of nidogen-1 gene. 12931195_structure of the interacting domains reveals a six-bladed Tyr-Trp-Thr-Asp (YWTD) beta-propeller domain in nidogen bound to laminin epidermal-growth-factor-like (LE) modules III3-5 in laminin (LE3-5) 15777791_Expression may promote formation of sinusoids after partial hepatectomy. 15824114_Laminin gamma3 chain, located in murine basement membranes, binds to nidogen 16024816_Nid1 is essential for late stages of lung development and for maintenance and/or integrity of cardiac tissue 17008882_nidogen-2 is equivalent to nidogen-1 in skin-organotypic coculture, and both can promote the development of a functional basement membrane zone 17023412_nidogen-1 and -2 have roles in development of the ectodermal basement membrane of the limb bud 17177854_expression of the entactin-1 dramatically declined in myogenesis 17616934_The lack of nidogen-1, nidogen-2, or both nidogens, plays no crucial role in the occurrence and localization of laminin-111, collagen type IV, and perlecan in murine tubular renal basement membranes. 18356808_In capillaries, basement membrane formation is severely impaired in the absence of nidogen-1 and -2, showing an irregular, patchy distribution and a dramatically reduced deposition of collagen IV, perlecan, and particularly laminin-411. 19530222_This study demonstrates that nidogen-1 is essential for maintenance of normal hippocampal excitability and synaptic plasticity. 19766719_Results suggest a non-structural role for nidogen 1 in wound repair. 21084308_Basement membrane deposition of nidogen 1 but not nidogen 2 requires the nidogen binding module of the laminin gamma1 chain. 22260998_show that loss of nidogen 2, but not of nidogen 1, promotes lung metastasis of melanoma cells. Histological and ultrastructural analysis of nidogen 1- and 2-deficient lungs did not reveal differences in morphology and ultrastructure of basement membrane 22623588_finding demonstrates that nidogens, although homologous proteins, exert their effect on BM assembly through different binding domains, which may in turn result in alterations of BM structure and functions 23948589_The C-terminal domains of mouse laminin gamma 1 and nidogen 1 mediate interactions complex formation. 24280453_nidogen-1 is essential to provide an anti-angiogenic environment of differentiated vessels. 25387007_Analysis of nidogen-1 and laminin-gamma1 interaction reveals multiple binding modes. 25957482_These studies provide strong evidence for a pathophysiological link between meprin beta and urinary excretion of cleaved nidogen-1 during cisplatin-induced acute kidney injury. 30893599_Nidogen-1 Contributes to the Interaction Network Involved in Pro-B Cell Retention in the Peri-sinusoidal Hematopoietic Stem Cell Niche. 35963565_Obesity impairs skeletal muscle repair through NID-1 mediated extracellular matrix remodeling by mesenchymal progenitors. |
ENSG00000116962 |
NID1 |
591.603807 |
3.2248496908 |
1.689232 |
0.13961787 |
142.488461 |
0.00000000000000000000000000000000760462379001756382982554424119299041350490680219100380034876710325799532261546865685285928604386640472512226551771163940429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000006695585524251302477975188903119023035123608849728998689134509831664822826462207675315974064744750648969784379005432128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
876.880995747863 |
84.9933222608469 |
271.913757175892 |
20.4967260137892 |
| ENSMUSG00000005465 |
50931 |
Il27ra |
protein_coding |
O70394 |
FUNCTION: Receptor for IL27. Requires IL6ST/GP130 to mediate signal transduction in response to IL27. This signaling system acts through STAT3 and STAT1. Involved in the regulation of Th1-type immune responses. Also appears to be involved in innate defense mechanisms. |
3D-structure;Glycoprotein;Membrane;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:50931; |
cytoplasm [GO:0005737]; external side of plasma membrane [GO:0009897]; extracellular region [GO:0005576]; interleukin-12 receptor complex [GO:0042022]; interleukin-23 receptor complex [GO:0072536]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; cytokine binding [GO:0019955]; cytokine receptor activity [GO:0004896]; interleukin-27 receptor activity [GO:0045509]; cytokine-mediated signaling pathway [GO:0019221]; defense response to Gram-positive bacterium [GO:0050830]; negative regulation of cellular extravasation [GO:0002692]; negative regulation of interleukin-17 production [GO:0032700]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of T cell extravasation [GO:2000408]; negative regulation of T-helper 17 type immune response [GO:2000317]; negative regulation of tumor necrosis factor production [GO:0032720]; negative regulation of type 2 immune response [GO:0002829]; positive regulation of activated T cell proliferation [GO:0042104]; positive regulation of T-helper 1 type immune response [GO:0002827]; positive regulation of type II interferon production [GO:0032729]; regulation of isotype switching to IgG isotypes [GO:0048302] |
15004160_A novel role is revealed for WSX-1 as an inhibitory regulator of cytokine production and inflammation in Con A-induced hepatitis. 15067040_The requirement for WSX-1 signaling in T helper type 1 cell differentiation is restricted to conditions in which IL-4 is produced. 15187144_demonstrate an important role for WSX-1 signaling in the promotion of type 1 responses and chronic gastrointestinal nematode infection 15494513_A novel role is identified for IL-27/WSX-1 in limiting innate and adaptive components of type 2 immunity at mucosal sites. 15585875_IL-27R signaling is dispensable for the induction of interferon (IFN)-gamma producing antigen-specific CD4 T cells during experimental pulmonary tuberculosis. 15850576_WSX-1 mRNA is markedly up-regulated in inflammatory cells in the CNS 15944269_Surface expression of the IL-27 receptor is low on naive T cells; the highest levels are found on effector and memory CD4+ and CD8+ T cells. 16081811_WSX-1 plays an important role in the down-regulation of airway hyper-reactivity and lung inflammation during the development of allergic asthma through its suppressive effect on cytokine production. 18178806_Whereas IFN-gammaR signals are dispensable for the T-bet-dependent IFN-gamma production, direct IL-27Ralpha signals are critical. 19354069_In contrast to the immunosuppressive role observed in the asthma model, IL-27/WSX-1 topically plays an exacerbating role in the pathogenesis of allergic rhinitis, presumably through differential expression of chemokines. 19549909_Results reveal an interleukin 27-independent function of WSX1 sensitizing natural killer cell-mediated antitumor surveillance. 19703422_an alternatively spliced WSX-1 isoform enables humanine to rescue Alzheimer's disease relevant neuronal death. 20631310_Findings demonstrate the essential role of IL-27R signaling in regulating effector T cell function during malaria infection and reveal a novel pathway that might be amenable to manipulation by drugs or vaccines. 21098093_Data show that the deletion of Il27ra ameliorates pathology in the pristane-induced SLE model. 21281790_In Th1-dependent autoinflammatory lesions, IL-27Ralpha has a biphasic role in vivo, initially pathogenic, but ultimately playing a protective role by regulating immune responses and attenuating disease. 22927332_These findings establish a novel antiatherogenic role for IL-27 receptor signaling, which acts to suppress the production of proinflammatory cytokines and chemokines and to curb the recruitment of inflammatory myeloid cells into atherosclerotic aortas. 23536628_Diminution of the Th1 response in infected Il27ra mice in vivo by neutralization of IL-12p40 attenuated CCR5 expression. 23993651_The loss of IL-27R signaling across the whole mouse resulted in reduced viral control and a slight increase in the number of virus-specific CD4 T cells at early stages of viral infection. 24244314_CCL4 and CCL5 expression was higher in livers of infected WSX-1(-/-) mice than infected WT mice, and hepatic CD4 T cells from WSX-1(-/-) mice expressed higher levels of CCR5 than cells from WT mice 24726498_IL-27R-mediated regulation of IL-17 controls the development of respiratory syncytial virus-associated pathogenesis. 26282876_The significant expression of IL-27 in TB and the negative influence of IL-27R on T cell function demonstrate the pathway by which this cytokine/receptor pair is detrimental in TB. 26459508_IL-10 production by CD4(+) YFP(+) T cells is controlled systemically during malaria infection through IL-27 receptor signaling that is supported after CD4(+) T cell priming by ICOS signaling. 26637970_The combination of mutant p53 & IL27RA(-/-) causes spontaneous liver inflammation, steatosis, and fibrosis, whereas either gene alone has no effects on the liver. 28536468_show that IL-27R signaling controls endothelial cells activation and myeloid cell recruitment at early and advanced stages of atherosclerosis. In the absence of IL-27R myeloid cells become hyperactivated, produce pro-inflammatory cytokines and act as more potent antigen presenting cells. 28874534_IL-27 signaling in Foxp3(+) Tregs essential for Tregs to control autoimmune inflammation in the central nervous system 29140433_IL-27 production was elevated during Clostridium difficile infection in humans and mice. Infected WSX-1-/- mice experienced increased weight loss, enhanced colonic histology damage, less Clostridium difficile clearance, and decreased survival compared to wild-type controls during Clostridium difficile infection. 29494633_Study reports a high-incidence, low-variation spontaneous mouse model of spondyloarthropathy that delineates how the combination of inflammatory cytokine interleukin-27 (IL-27) signaling deficiency and mitogenic signaling (mutant p53R172H) in vivo, leads to bone loss in the vertebral bodies and ossification of the cartilage in the intervertebral discs. 29728641_this paper shows that the increased protection and pathology in Mycobacterium tuberculosis-infected IL-27R-alpha-deficient mice is supported by IL-17A and is associated with the IL-17A-induced expansion of multifunctional T cells 29779048_data suggested that IL-27/IL-27R expression induced by Toxoplasma gondii infection may regulate mast cell-mediated immune response during acute ocular toxoplasmosis in mouse model 32075272_IL-27Ralpha: A Novel Molecular Imaging Marker for Allograft Rejection. 32305041_Aging-induced IL27Ra signaling impairs hematopoietic stem cells. 32307922_Impaired Interleukin-27-Mediated Control of CD4+ T Cell Function Impact on Ectopic Lymphoid Structure Formation in Patients With Sjogren's Syndrome. 32522836_Loss of IL-27Ralpha Results in Enhanced Tubulointerstitial Fibrosis Associated with Elevated Th17 Responses. 32761753_IL-27 Ralpha(+) cells promoted allorejection via enhancing STAT1/3/5 phosphorylation. 33863788_IL-27/IL-27R Mediates Protective Immunity against Chlamydial Infection by Suppressing Excessive Th17 Responses and Reducing Neutrophil Inflammation. 34299138_Deficiency of IL-27 Signaling Exacerbates Experimental Autoimmune Uveitis with Elevated Uveitogenic Th1 and Th17 Responses. |
ENSG00000104998 |
IL27RA |
19.668174 |
5.6112677869 |
2.488327 |
0.65236172 |
14.467101 |
0.00014262894804462016126907208946050786835257895290851593017578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008216808383569040603183752935478878498543053865432739257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.6138139159541 |
15.7228804473052 |
5.99041343107566 |
2.26598590624768 |
| ENSMUSG00000005474 |
59310 |
Myl10 |
protein_coding |
Q62082 |
|
Alternative splicing;Calcium;Metal-binding;Reference proteome;Repeat |
|
|
mmu:59310; |
cytoplasm [GO:0005737]; mitochondrion [GO:0005739]; calcium ion binding [GO:0005509] |
Human_homologues 20877624_Observational study of gene-disease association. (HuGE Navigator) |
ENSG00000106436 |
MYL10 |
35.983260 |
4.6187738819 |
2.207510 |
0.49214536 |
19.940112 |
0.00000799061343509253064185983428391324423500918783247470855712890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000603136887231563086639654636122997999336803331971168518066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
61.9740325314733 |
21.5535722150883 |
13.4178537672471 |
3.75201395583141 |
| ENSMUSG00000005583 |
17260 |
Mef2c |
protein_coding |
Q8CFN5 |
FUNCTION: Transcription activator which binds specifically to the MEF2 element present in the regulatory regions of many muscle-specific genes. Controls cardiac morphogenesis and myogenesis, and is also involved in vascular development. Enhances transcriptional activation mediated by SOX18 (PubMed:11554755). May also be involved in neurogenesis and in the development of cortical architecture. Isoforms that lack the repressor domain are more active than isoform 1 (By similarity). Plays an essential role in hippocampal-dependent learning and memory by suppressing the number of excitatory synapses and thus regulating basal and evoked synaptic transmission. Crucial for normal neuronal development, distribution, and electrical activity in the neocortex. Necessary for proper development of megakaryocytes and platelets and for bone marrow B-lymphopoiesis. Required for B-cell survival and proliferation in response to BCR stimulation, efficient IgG1 antibody responses to T-cell-dependent antigens and for normal induction of germinal center B-cells. {ECO:0000250|UniProtKB:Q06413, ECO:0000269|PubMed:11554755, ECO:0000269|PubMed:18086704, ECO:0000269|PubMed:18438409, ECO:0000269|PubMed:18599437, ECO:0000269|PubMed:18599438, ECO:0000269|PubMed:19211936, ECO:0000269|PubMed:9162005, ECO:0000269|PubMed:9778514}. |
3D-structure;Acetylation;Activator;Alternative splicing;Apoptosis;Cytoplasm;Developmental protein;Differentiation;DNA-binding;Isopeptide bond;Neurogenesis;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:17260; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; nuclear speck [GO:0016607]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; postsynapse [GO:0098794]; protein-containing complex [GO:0032991]; sarcomere [GO:0030017]; sarcoplasm [GO:0016528]; chromatin binding [GO:0003682]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; histone deacetylase binding [GO:0042826]; HMG box domain binding [GO:0071837]; minor groove of adenine-thymine-rich DNA binding [GO:0003680]; protein heterodimerization activity [GO:0046982]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; apoptotic process [GO:0006915]; B cell homeostasis [GO:0001782]; B cell proliferation [GO:0042100]; B cell receptor signaling pathway [GO:0050853]; blood vessel development [GO:0001568]; blood vessel remodeling [GO:0001974]; cardiac muscle cell differentiation [GO:0055007]; cardiac muscle hypertrophy in response to stress [GO:0014898]; cardiac ventricle formation [GO:0003211]; cartilage morphogenesis [GO:0060536]; cell differentiation [GO:0030154]; cell fate commitment [GO:0045165]; cell morphogenesis involved in neuron differentiation [GO:0048667]; cell proliferation in bone marrow [GO:0071838]; cellular response to calcium ion [GO:0071277]; cellular response to fluid shear stress [GO:0071498]; cellular response to growth factor stimulus [GO:0071363]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to parathyroid hormone stimulus [GO:0071374]; cellular response to retinoic acid [GO:0071300]; cellular response to transforming growth factor beta stimulus [GO:0071560]; cellular response to trichostatin A [GO:0035984]; cellular response to xenobiotic stimulus [GO:0071466]; chondrocyte differentiation [GO:0002062]; embryonic heart tube development [GO:0035050]; embryonic skeletal system morphogenesis [GO:0048704]; embryonic viscerocranium morphogenesis [GO:0048703]; endochondral ossification [GO:0001958]; epithelial cell proliferation involved in renal tubule morphogenesis [GO:2001013]; excitatory postsynaptic potential [GO:0060079]; gene expression [GO:0010467]; germinal center formation [GO:0002467]; glomerulus morphogenesis [GO:0072102]; heart development [GO:0007507]; heart looping [GO:0001947]; humoral immune response [GO:0006959]; learning or memory [GO:0007611]; MAPK cascade [GO:0000165]; melanocyte differentiation [GO:0030318]; monocyte differentiation [GO:0030224]; muscle cell fate determination [GO:0007521]; negative regulation of blood vessel endothelial cell migration [GO:0043537]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of gene expression [GO:0010629]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of ossification [GO:0030279]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of vascular associated smooth muscle cell migration [GO:1904753]; negative regulation of vascular associated smooth muscle cell proliferation [GO:1904706]; negative regulation of vascular endothelial cell proliferation [GO:1905563]; nephron tubule epithelial cell differentiation [GO:0072160]; neural crest cell differentiation [GO:0014033]; neuron development [GO:0048666]; neuron differentiation [GO:0030182]; neuron migration [GO:0001764]; osteoblast differentiation [GO:0001649]; outflow tract morphogenesis [GO:0003151]; platelet formation [GO:0030220]; positive regulation of alkaline phosphatase activity [GO:0010694]; positive regulation of B cell proliferation [GO:0030890]; positive regulation of behavioral fear response [GO:2000987]; positive regulation of bone mineralization [GO:0030501]; positive regulation of cardiac muscle cell differentiation [GO:2000727]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cardiac muscle hypertrophy [GO:0010613]; positive regulation of cell proliferation in bone marrow [GO:0071864]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of macrophage apoptotic process [GO:2000111]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of myoblast differentiation [GO:0045663]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of osteoblast differentiation [GO:0045669]; positive regulation of skeletal muscle cell differentiation [GO:2001016]; positive regulation of skeletal muscle tissue development [GO:0048643]; positive regulation of transcription by RNA polymerase II [GO:0045944]; primary heart field specification [GO:0003138]; regulation of AMPA receptor activity [GO:2000311]; regulation of dendritic spine development [GO:0060998]; regulation of DNA-templated transcription [GO:0006355]; regulation of germinal center formation [GO:0002634]; regulation of megakaryocyte differentiation [GO:0045652]; regulation of neuron apoptotic process [GO:0043523]; regulation of neurotransmitter secretion [GO:0046928]; regulation of NMDA receptor activity [GO:2000310]; regulation of sarcomere organization [GO:0060297]; regulation of synapse assembly [GO:0051963]; regulation of synaptic activity [GO:0060025]; regulation of synaptic plasticity [GO:0048167]; regulation of synaptic transmission, glutamatergic [GO:0051966]; regulation of transcription by RNA polymerase II [GO:0006357]; renal tubule morphogenesis [GO:0061333]; response to ischemia [GO:0002931]; roof of mouth development [GO:0060021]; secondary heart field specification [GO:0003139]; semaphorin-plexin signaling pathway involved in axon guidance [GO:1902287]; sinoatrial valve morphogenesis [GO:0003185]; skeletal muscle cell differentiation [GO:0035914]; skeletal muscle tissue development [GO:0007519]; smooth muscle cell differentiation [GO:0051145]; sympathetic neuron axon guidance [GO:0097492]; transdifferentiation [GO:0060290]; ventricular cardiac muscle cell differentiation [GO:0055012] |
12130539_cyclin D-cdk4 activity modulates the subnuclear localization and interaction of MEF2C with SRC-family coactivators, GRIP-1, during skeletal muscle differentiation 12694204_Calcineurein regulates skeletal muscle differentiation by activating MEF2 and MyoD transcription factors. 12857748_MEF2 activation of the troponin I slow enhancer is repressed by hMusTRD1alpha1 14515274_MEKK2-dependent role of MEF2C in induction of c-Jun expression in mast cells activated through FcepsilonRI. 14550531_mef2c is a direct transcriptional target of the MyoD family in vivo via an essential E box in the skeletal muscle enhancer of mef2c 15253934_First direct transcriptional target of ISL1 in the anterior heart field. 15340086_MEF2C is expressed exclusively as gamma- isoforms in heart tissue and predominantly as gamma- in other adult tissues and in differentiating myocytes. 15546868_Mirk is induced within the initial 24 h of myogenic differentiation and enables MEF2 to transcribe the myogenin gene by decreasing the nuclear accumulation of class II HDACs 15834131_A conserved pattern of alternative splicing in vertebrate MEF2 (myocyte enhancer factor 2) genes generates an acidic activation domain in MEF2 proteins selectively in tissues where MEF2 target genes are highly expressed. (MEF2) 15890826_BOP, a regulator of right ventricular heart development, is an essential downstream effector gene of MEF2C in the developing heart. 15998798_Our study illustrates the existence of a positive feedback mechanism that ensures an adequate supply of releasable Ca(2+) is maintained within the cell for activation of calcineurin and, subsequently, for proper functioning of MEF2C. 16510869_examination of coactivation of MEF2C transcription along with notch 16680724_There is a fundamental role of MEF2C in ventricular cardiomyocyte differentiation and apportioning of cells between inflow and outflow precursors in the primary heart field. 16775627_The increase of MEF2C protein stability is a key factor in the AICD of macrophage. 17003108_Results show that MEF2C, or genes containing MEF2 DNA-binding sites, is required for the efficient differentiation of cardiomyoblasts into cardiomyocytes. 17101791_HDAC9 gene is a direct transcriptional target of MEF2 in vitro and in vivo 17336904_These findings reveal unexpected commonalities in the mechanisms governing muscle, cardiovascular, and bone development with respect to their regulation by MEF2 and class II HDACs. 17420000_The studies presented here define a feed-forward transcriptional circuit between the MADS-box transcription factor MEF2C and the homeodomain transcription factors Dlx5 and Dlx6 in craniofacial development. 17696759_SOST expression in osteocytes of adult bone and its inhibition by PTH is mediated by MEF2A, C, and D transcription factors controlling the SOST bone enhancer. 17875930_These results reveal a key role for Mef2c in maintenance of sarcomere integrity and postnatal maturation of skeletal muscle. 18438409_Mef2c is required for B cell proliferation and survival in response to antigen receptor stimulation, and regulates T cell-dependent and T cell-independent antibody responses. 18579729_MEF2C-directed neuronal progenitor cells transplanted into a mouse model of cerebral ischemia can successfully differentiate into functioning neurons and ameliorate stroke-induced behavioral deficits. 18599437_These data support a crucial role for MEF2C in programming early neuronal differentiation and proper distribution within the layers of the neocortex. 18599438_MEF2C limits excessive synapse formation during activity-dependent refinement of synaptic connectivity and thus facilitates hippocampal-dependent learning and memory. 18718538_The current results reveal a signaling cascade that drives the expression profiles of ColQ in responding to activity challenge via acetylcholine and myocyte enhancer factor 2 in cultured myotubes. 18849322_Chromatin immunoprecipitation experiments revealed that Mef2C, Tbx5, and titin are direct target genes of HIF1alpha in vivo. Thus, hypoxia signaling controls cardiac development through HIF1alpha-mediated transcriptional regulation 18946175_GSK-3 beta and SIK1 may play important roles in the regulation of PGC-1alpha gene expression by inactivating HDAC5 followed by activation of MEF2C. 18955699_MEF2C regulates proliferation in response to B-cell receptor stimulation via the p38 MAPK pathway. 19035347_genetic interactions between Nkx2.5 and Mef2c are necessary for ventricle formation 19129464_Myostatin regulates fiber-type composition of skeletal muscle by regulating MEF2 and MyoD gene expression. 19169261_Mef2c is a crucial component of the transcriptional network that regulates cell fate in multipotent progenitors. 19211936_Mef2C is an integral member of hematopoietic transcription factors with distinct upstream regulatory mechanisms and functional requirements in megakaryocyte and B-lymphoid lineages 19321441_MEF2 and MITF function cooperatively with NFATc1 to transactivate the V-ATPase d2 promoter during RANKL-induced osteoclastogenesis. 19704004_Knockdown of PP2A inhibits forskolin-induced nuclear translocation of HDAC4 and attenuates the ability of this signaling molecule to repress collagen X expression in chondrocytes. 19893013_a novel gender-specific pathway of cardioprotection mediated by ERalpha and its regulation by MEF2 and class II HDACs. 19914232_Foxj3 is an important regulator of myofiber identity and muscle regeneration through the transcriptional activation of the Mef2c gene. 19926876_Data show that LAP2alpha knockout caused systolic dysfunction in young mice, sporadic fibrosis in old animals, and disregulation of cardiac GATA4 and MEF2c, while dstrophin deficiency in Mdx mice resulted in a transient rescue of Lap2alpha(-/-) phenotype. 20023039_Inhibition of MEF2 during pressure overload increases reactive oxygen species induced apoptosis in cardiomyocytes 20041152_MEF2C depletion was accompanied by defective activation of S6K in response to pressure overload. 20111592_E18.5 Igf1(+/+) mouse auditory ganglion neurons showed intense MEF2A and MEF2D nuclear staining and MEF2A was also evident in the organ of Corti 20141540_Fox-1, expressed specifically in the neural cell stage, promoted Mef2c exon beta inclusion via the GCAUG. 20197093_Sustained depolarization of co-cultures induced a profound pruning of glutamatergic synapses and spines in striatopallidal neurons that was dependent upom MEF2. 20334626_Study found that most MRFs, except for myogenin and Mef2C significantly increased M-cadherin promoter activity, but had no effect on that of N-cadherin. 20412016_Data show that p38 MAPK activity specifically inhibits neuronal differentiation, and that p38 MAPK achieves these effects by activating the transcription factor MEF2C, which in turn directly regulates BMP-2 expression. 20709755_RNA-binding protein Muscleblind-like 3 (MBNL3) disrupts myocyte enhancer factor 2 (Mef2) {beta}-exon splicing 20736237_miR-27b displays an overt myocardial expression during heart development and the transcription factor-encoding gene Mef2c is an miR-27b target 20801874_a novel role for Pin1 as a regulator of muscle terminal differentiation and suggest that Pin1-mediated repression of MEF2C function could contribute to this function. 20923879_data dissociate myocyte hypertrophy, a consistent response in HCM, from heterogeneous Mef2 activation and reexpression of a fetal gene program 21556048_Transcripts of MEF2C increase on days 2-5 and 9, representing cardiac and skeletal muscle induction in P19 embryonal carcinoma cells, respectively. 21610032_SOX10 and MEF2C physically interact and function cooperatively to activate the Mef2c gene in a feed-forward transcriptional circuit, suggesting that MEF2C might serve as a potentiator of the transcriptional pathways affected in Waardenburg syndromes 21652706_knockdown of bone-specific transcription factors, Runx2 and osterix by shRNAi knockdown of Mef2c, suggests that Mef2c lies upstream of these two important factors in the cascade of gene expression in osteoblasts. 21689744_MEF2 transactivation activity is inhibited by CaMK IV-mediated prohibitin 2 phosphorylation. 21715356_Mef2c may act as a novel transcription factor involved in the development and the maintenance of photoreceptor cells 21741404_[review] The mechanisms of regulation of MEF2 function by well-known neurotoxins and their implications in disease models are reviewed. 21842419_Significant gene expression changes due to overexpression of Mef2c isoforms in both an isoforms-dependent and -independent manner. 21849497_MEF2C is a transcriptional regulator of homeostasis in rod photoreceptor cells 22135324_PPARbeta/delta, but not PPARalpha, interacts with the exercise-inducible kinase AMP-activated protein kinase (AMPK) to synergistically activate Ldhb gene transcription by cooperating with myocyte enhancer factor 2A (MEF2A) in a PPARbeta/delta ligand-independent manner 22275376_a novel signaling cascade that links RhoA-mediated calcium sensitivity to MEF2-dependent myocardin expression in VSMCs through a mechanism involving p38 MAPK, PP1alpha, and CPI-17. 22311635_Forced expression of MEF2C in transitional B cells promoted cell survival, proliferation, and upregulation of pro-survival genes. 22342668_MEF2C could modulate and restrain myogenesis by myostatin activation and myostatin-dependent gene processing in porcine. 22361433_Gene and protein expression of MEF2C was significantly downregulated in skeletal muscle from Colon 26 carcinoma-bearing mice. 22367967_We show that Tbx1 occupies conserved regulatory regions of the Mef2c locus, suggesting a direct effect on Mef2c transcription 22496871_The data highlighted the key in vivo role of MEF2C isoform in the brain and suggest that MEF2A and MEF2D have only subtle roles in regulating hippocampal synaptic function. 22521302_MEF2C may play an antiangiogenic role in retinal endothelial cells under stress conditions, and modulation of MEF2C may prevent pathologic retinal neovascularization. 22581928_Induction of Gata4/Mef2c/Tbx5 overexpression in fibroblasts is inefficient at inducing molecular and electrophysiological phenotypes of mature cardiomyocytes. 22764880_MEF2A--but not MEF2C or MEF2D--is modified by ubiquitination in dopaminergic neuronal cell line SN4741 cells. 22886088_the absence of the Sost-specific long-range regulatory element ECR5 causes van Buchem disease in rodents, and that Mef2C is the main transcription factor responsible. 22931955_The gene transfer of three cardiac transcription factors, Gata4, Mef2c, and Tbx5 (GMT), induced cardiomyocyte-like cells in infarcted hearts in mice. 22956541_Foxk1 promotes muscle progenitor cell proliferation by repressing Foxo4 transcriptional activity and inhibits myogenic differentiation by repressing Mef2 activity. 22973001_Together, these data show that individual members of the MEF2 family of transcription factors differentially regulate the expression of Bdnf. 22986367_These findings establish Mef2 transcription factors as relevant molecular actors involved in the mechanisms by which Sirt1 controls heart functions. 23055528_Mef2c induces microRNAs miR-1 and miR-206, which directly down-regulate Notch3 and allow differentiation to proceed. 23087187_Chromatin immunoprecipitation analysis confirmed Mef2c binding to the promoters of these genes indicating a direct link between the presence (or absence) of Mef2c and altered transcriptional control in mature B-cells. 23143398_Results point to Mef2 and miR-133 as central upstream regulators of Prdm16 and hence of brown adipogenesis in response to cold exposure in BAT and SAT. 23328440_while loss of delta-catenin does not alter the expression levels of endogenous Mef2, expression of Mef2 in neurons that are knocked down for delta-catenin promotes spine elimination. 23389741_MEF2C deletion were statistically less likely to have epilepsy. 23598283_testosterone induced cardiomyogenesis, at least in part, by recruiting the AR receptor to the regulatory regions of the MEF2C and HCN4 genes. 23603510_Apelin-APJ signaling plays a novel role as a potent regulator of endothelial MEF2 function in the developing cardiovascular system. 23704920_Overexpression of transcription factors MYOCD and SRF alone or in conjunction with Mesp1 and SMARCD3 enhanced the basal but necessary cardio-inducing effect of the previously reported GATA4, TBX5, and MEF2C. 24043307_Activation of MEF2 reverses the oncogenic properties of cells expressing HDAC4. 24147056_The data suggested that Islet-1 might function as an assistant factor that was involved in the regulation of histone acetylation and Mef2c expression via assisting p300 on specifically targeting the promoter of Mef2c. 24495128_TGF-beta1-induced Mmp20 expression in ALCs was regulated through MEF2-binding site on Mmp20 promoter and thus mediated by MEF2C signaling pathway. 24591619_Combined deletion of the Mef2a, c, and d genes results in a blockade to muscle regeneration. 24692107_Dlx5 and Mef2 directly bound to the runx2 enhancer, and the binding sites were required for the osteoblast-specific expression 24750843_The mRNA expression of Mef2c varies during cardiogenesis in mice, which indicates that Mef2c plays an important role in the process of cardiac development. 24811170_SRC-2 is critical to transcriptional control modulated by MEF2, GATA-4, and Tbx5, thereby enhancing gene expression associated with cardiac growth. 24891619_MEF2C is a key effector of the myogenesis program promoted by AUF1. 25056116_Data indicate that low-dose parathyroid hormone (PTH) decreased the expression of the myocyte enhancer factor 2C (Mef2c) transcription factor, resulting in decreased sclerostin (Sost) expression in osteoblasts/osteocytes. 25271055_increased sclerostin production achieved by HDAC5 shRNA is abrogated by simultaneous knockdown of MEF2C, indicating that MEF2C is a major target of HDAC5 in osteocytes 25416778_Whereas MEF2A is absolutely required for proper myoblast differentiation, MEF2B, -C, and -D were found to be dispensable for this process. 25474999_Myocyte enhancer factor 2 C overexpression by adenovirus significantly repressed TNF-alpha induction of pro-inflammatory molecules, activation of NF-kappaB, and leukocyte adhesion to endothelial cells 25763715_this is a novel mechanism of H2S-mediated activation of MEF2C and induction of miR-133a and inhibition of hypertrophy in HHcy cardiomyocytes. 25774500_In skeletal muscle, transcription of Lmod3 was controlled by the transcription factors SRF and MEF2. 25826570_The RBM4-MEF2C-miR-1 network constitutes a novel mechanism which programs the gene expression profile toward the development of brown adipocytes. 26019261_Our results identify novel target genes for MEF2 and define MEF2 as an important regulator of Leydig cell function and male reproduction. 26160899_Endothelin signaling activates Mef2c expression in the neural crest through a MEF2C-dependent positive-feedback transcriptional pathway. 26196305_our data indicate that robust hypertrophic MEF2 activation in the heart in vivo requires a background of adiponectin signaling and that adiponectin signaling in primary isolated CM directly enhances MEF2 activity through activation of p38 MAPK 26361013_Analysis of SOST expression using large minigenes reveals the MEF2C binding site in the evolutionarily conserved region (ECR5) enhancer mediates forskolin, but not 1,25-dihydroxyvitamin D3 or TGFbeta1 responsiveness. 26449528_Data show that three transcriptional factors Gata4, Mef2c, and Tbx5 (abbreviated as GMT) significantly improved murine embryonic stem cells (ESCs) differentiated into cardiomyocytes. 26459759_SmarcA4 is required for synapse development and myocyte enhancer factor 2-mediated synapse remodeling 26642739_Critical role for MEF2C in the regulation of spine numbers with a dissociation of learning and memory, synaptic plasticity, and measures of autism-related behaviors in postnatal mouse brain. 26647388_Ca(2+) signaling pathway increases Nr4a1 expression in MA-10 Leydig cells, at least in part, by enhancing the recruitment of coactivator most likely through the MEF2, AP1, and CREB transcription factors thus demonstrating an important interplay between the Ca(2+) and cAMP pathways in regulating Nr4a1 expression. 26660426_These findings uncover a novel role for Mef2c/d in coordinating the transcriptional network that promotes early B-cell development. 26748576_results identify a novel cooperation between MEF2 factors and NR2F2 in the expression of the Akr1c14 gene 26788505_We conclude that ILK negatively and independently of PI3K regulated MEF2C phosphorylation activity and MCK mRNA expression in C2C12 cells 26811383_MEF2C has a role in regulating outflow tract alignment and transcriptional control of Tdgf1 27292777_Results show that MEF2C interacts with the N-terminal pre-LIM region of nTRIP6 in proliferating myoblasts. 27427476_In cardiomyocytes exposed to biomechanical stimulation, FAK accumulates in the nucleus, binds to and upregulates the transcriptional activity of MEF2c through an interaction with the FAK focal adhesion targeting (FAT) domain. 27507714_MEF2C protects B lymphopoiesis during stress by ensuring proper expression of genes that encode DNA repair and B-cell factors. 27595386_that Foxp2-Mef2C signaling is critical to corticostriatal circuit formation 27612437_the expression and the phosphorylation of MEF2Calpha1 are critically required to sustain the adult myogenesis. 27743478_lf5 ChIP-seq revealed that Klf5 binding overlaps that of MyoD and Mef2, and Klf5 physically associates with both MyoD and Mef2. In addition, MyoD recruitment was greatly reduced in the absence of Klf5. These results indicate that Klf5 is an essential regulator of skeletal muscle differentiation, acting in concert with myogenic transcription factors such as MyoD and Mef2. 27779093_The authors show here that conditional embryonic deletion of Mef2c in cortical and hippocampal excitatory neurons (Emx1-lineage) produces a dramatic reduction in cortical network activity in vivo, due in part to a dramatic increase in inhibitory and a decrease in excitatory synaptic transmission. Perturbing MEF2C function in neocortex can produce autistic- and intellectual disability-like behaviors in mice. 27796324_MEF2C is a novel target of miR-214-3p in myocardial hypertrophy, and enhancement of miR-214-3p expression may be protective against myocardial hypertrophy. 27901468_Here, the authors show that loss of Fxn in the nervous system in mice also activates an iron/sphingolipid/PDK1/Mef2 pathway, indicating that the mechanism is evolutionarily conserved. 27989458_Postnatal, postsynaptic deletion of Mef2c in a sparse population of L2/3 neurons suppressed development of excitatory synaptic connections from all local input pathways tested. In the same cell population, Mef2c deletion promoted the strength of excitatory inputs originating from contralateral neocortex. Both the synapse promoting and synapse suppressing effects of Mef2c deletion required normal whisking experience. 28017720_Mef2c is highly expressed in the retina where it modulates photoreceptor-specific gene expression 28025327_In Fmr1 KO neurons, Mdm2 is hyperphosphorylated, nuclear localized basally, and unaffected by MEF2 activation, which our data suggest due to an enhanced interaction with Eukaryotic Elongation Factor 1alpha (EF1alpha), whose protein levels are elevated in Fmr1 KO. Expression of a dephosphomimetic of Mdm2 rescues PSD-95 ubiquitination, degradation and synapse elimination in Fmr1 KO neurons. 28115742_This study demonstrated that using multidimensional integrative approaches, we identify MEF2C TF as promising therapeutic target for schizophrenia and other psychiatric disease associated with impaired cognition. 28332716_HDAC5 emerges as a cellular conductor of MEF2C and M6a activity and is regulated by miR-124 and miR-9 to control neurite development. 28351867_two MEF2 sites in the enhancer function cooperatively due to bridging of the MEF2C-bound sites by the SAP domain-containing co-activator protein myocardin 28473437_Endothelial Mef2c regulates the endothelial actin cytoskeleton and inhibits smooth muscle cell migration into the intima. 28607167_Our results elucidate the specific role of the transcription factors CREB, SRF, and MEF2 in the depression and potentiation components of ODP in vivo, therefore better informing future attempts to find therapeutic targets for diseases where activity-dependent plasticity is disrupted. 28959042_Immune challenge in mice lacking Mef2C in microglia results in an exaggerated microglial response and has an adverse effect on mice behaviour. 28973134_MEF2C is necessary for Mmp13 gene expression at the transcriptional level and participates in PTH-stimulated Mmp13 gene expression by increased binding to c-FOS at the AP-1 site in the Mmp13 promoter. 29133852_results suggest that MEF2C haploinsufficiency leads to abnormal brain development, E/I imbalance, and neurobehavioral dysfunction, which may be mitigated by pharmacological intervention 29257325_Enrichment of induced cardiomyocytes derived from mouse fibroblasts can be achieved by reprogramming with cardiac transcription factors, Gata4, MEF2c, Tbx5, and Hand2. 29505923_MiR-204-5p inhibits myoblast differentiation by targeting MEF2C and ERRgamma. 29981832_A large number of Mef2c targets overlapped with genes down-regulated by Wnt16 and Mef2c itself was transcriptionally repressed by Wnt16 suggesting that Mef2c plays a role in Wnt16-mediated transcriptional regulation. 30106596_MEF2 (myocyte enhancer factor 2) is a transcription factor that regulates multiple genes that are important during cardiovascular development and for vascular homeostasis. 30166330_Tbx1 regulates the Mef2c anterior heart field enhancer by inducing histone deacetylation. 30276662_These results reveal the specific expression and functional relevance of Mef2c in developing Purkinje cells. 30305329_Mef2c degradation was mediated by Skp2 E3 ligase 30511745_Inhibition of MEF2c and MyoD expression in the SM of lipin1(Myf5cKO) mice. 30521808_Deletion of Mef2c in the early myocardial lineage using Nkx2-5(Cre) resulted in cardiac and vascular abnormalities that were indistinguishable from the defects in the global Mef2c knockout. In contrast, early deletion of Mef2c in the vascular endothelium using an Etv2::Cre line active prior to the onset of Mef2c expression resulted in viable offspring that were indistinguishable from wild type controls. 31346235_Orai1 inhibitor STIM2beta regulates myogenesis by controlling SOCE dependent transcriptional factors. 31614133_The Ca(2+)-CaMKII-MEF2C signaling pathway is the key factor involved in regulating CAIII expression in skeletal muscle. 31628386_Stoichiometric optimization of Gata4, Hand2, Mef2c, and Tbx5 expression for contractile cardiomyocyte reprogramming. 31915320_Direct Cardiac Reprogramming for Cardiovascular Regeneration and Differentiation. 31930767_Downregulation of circulating miR 802-5p and miR 194-5p and upregulation of brain MEF2C along breast cancer brain metastasization. 31979018_Isoform Specific Effects of Mef2C during Direct Cardiac Reprogramming. 32132110_MEF2c-Dependent Downregulation of Myocilin Mediates Cancer-Induced Muscle Wasting and Associates with Cachexia in Patients with Cancer. 32418612_MEF2C Hypofunction in Neuronal and Neuroimmune Populations Produces MEF2C Haploinsufficiency Syndrome-like Behaviors in Mice. 32736694_O-GlcNAcylation of Mef2c regulates myoblast differentiation. 32757160_Decreased Myocyte Enhancer Factor 2 Levels in the Hippocampus of Huntington's Disease Mice Are Related to Cognitive Dysfunction. 32851972_An essential role for MEF2C in the cortical response to loss of sleep in mice. 32975584_Genes influenced by MEF2C contribute to neurodevelopmental disease via gene expression changes that affect multiple types of cortical excitatory neurons. 33249116_Cholecystokinin peptide signaling is regulated by a TBX5-MEF2 axis in the heart. 33315889_MEF2 transcription factors differentially contribute to retinal ganglion cell loss after optic nerve injury. 33359123_Adiponectin promotes myogenic differentiation via a Mef2C-AdipoR1 positive feedback loop. 33452355_Myocyte-specific enhancer factor 2c triggers transdifferentiation of adipose tissue-derived stromal cells into spontaneously beating cardiomyocyte-like cells. 34029137_Overexpression of Gata4, Mef2c, and Tbx5 Generates Induced Cardiomyocytes Via Direct Reprogramming and Rare Fusion in the Heart. 34290714_A Biological Circuit Involving Mef2c, Mef2d, and Hdac9 Controls the Immunosuppressive Functions of CD4+Foxp3+ T-Regulatory Cells. 34779502_MicroRNA190b expression predicts a good prognosis and attenuates the malignant progression of pancreatic cancer by targeting MEF2C and TCF4. 35130621_MEF2C ameliorates learning, memory, and molecular pathological changes in Alzheimer's disease in vivo and in vitro 35194174_MEF2C promotes M1 macrophage polarization and Th1 responses. 35536646_MEF2C opposes Notch in lymphoid lineage decision and drives leukemia in the thymus. 35729843_MEF2 intervened LPS-induced acute lung injury by binding to KLF2 promoter and modulating macrophage phenotype. 36293168_PGC-1alpha and MEF2 Regulate the Transcription of the Carnitine Transporter OCTN2 Gene in C2C12 Cells and in Mouse Skeletal Muscle. 36610028_Actin-related protein 5 suppresses the cooperative activation of cardiac gene transcription by myocardin and MEF2. |
ENSG00000081189 |
MEF2C |
2034.707494 |
0.1604413005 |
-2.639883 |
0.09739510 |
685.484049 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000042888432055268397323553946679565591646373992681740921865374141617286123653462548590084729222044458574021645558411 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000003629314428143601262022825440586303888820881828215778799982911212734847937000077603622398254236534323878352611568 |
Yes |
No |
543.983461613617 |
36.7517572883165 |
3390.54507660091 |
162.435792308372 |
| ENSMUSG00000005836 |
14465 |
Gata6 |
protein_coding |
Q61169 |
FUNCTION: Transcriptional activator that regulates SEMA3C and PLXNA2 (PubMed:19666519). May regulate genes that protect epithelial cells from bacterial infection (By similarity). Involved in gene regulation specifically in the gastric epithelium (By similarity). Involved in bone morphogenetic protein (BMP)-mediated cardiac-specific gene expression (PubMed:15329343). Binds to BMP response element (BMPRE) DNA sequences within cardiac activating regions (PubMed:15329343). {ECO:0000250|UniProtKB:Q92908, ECO:0000269|PubMed:15329343, ECO:0000269|PubMed:19666519}. |
Activator;Alternative initiation;DNA-binding;Isopeptide bond;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:14465; |
nuclear membrane [GO:0031965]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; chromatin binding [GO:0003682]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; DNA binding [GO:0003677]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; double-stranded DNA binding [GO:0003690]; NFAT protein binding [GO:0051525]; protein kinase binding [GO:0019901]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; transcription coactivator binding [GO:0001223]; zinc ion binding [GO:0008270]; animal organ formation [GO:0048645]; cardiac muscle cell differentiation [GO:0055007]; cardiac muscle cell proliferation [GO:0060038]; cardiac muscle hypertrophy in response to stress [GO:0014898]; cardiac muscle tissue development [GO:0048738]; cardiac vascular smooth muscle cell differentiation [GO:0060947]; cell fate commitment [GO:0045165]; cellular response to BMP stimulus [GO:0071773]; cellular response to gonadotropin stimulus [GO:0071371]; cellular response to hypoxia [GO:0071456]; club cell differentiation [GO:0060486]; endodermal cell differentiation [GO:0035987]; endodermal cell fate determination [GO:0007493]; epithelial cell differentiation [GO:0030855]; G1 to G0 transition involved in cell differentiation [GO:0070315]; gene expression [GO:0010467]; heart contraction [GO:0060047]; in utero embryonic development [GO:0001701]; intestinal epithelial cell differentiation [GO:0060575]; liver development [GO:0001889]; lung saccule development [GO:0060430]; male gonad development [GO:0008584]; negative regulation of apoptotic process [GO:0043066]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of miRNA transcription [GO:1902894]; negative regulation of sebum secreting cell proliferation [GO:1904003]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of transforming growth factor beta1 production [GO:0032911]; negative regulation of transforming growth factor beta2 production [GO:0032912]; odontogenesis of dentin-containing tooth [GO:0042475]; outflow tract septum morphogenesis [GO:0003148]; pancreas development [GO:0031016]; pancreatic A cell differentiation [GO:0003310]; phospholipid metabolic process [GO:0006644]; positive regulation of angiogenesis [GO:0045766]; positive regulation of apoptotic process [GO:0043065]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of cardiac muscle cell apoptotic process [GO:0010666]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cardiac muscle myoblast proliferation [GO:0110024]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of transforming growth factor beta activation [GO:1901390]; regulation of antimicrobial humoral response [GO:0002759]; regulation of cell differentiation [GO:0045595]; regulation of gene expression [GO:0010468]; response to cAMP [GO:0051591]; response to estrogen [GO:0043627]; response to growth factor [GO:0070848]; response to retinoic acid [GO:0032526]; response to toxic substance [GO:0009636]; response to xenobiotic stimulus [GO:0009410]; sebaceous gland cell differentiation [GO:0001949]; sinoatrial node development [GO:0003163]; skin epidermis development [GO:0098773]; smooth muscle cell differentiation [GO:0051145]; stem cell differentiation [GO:0048863]; tube morphogenesis [GO:0035239]; type B pancreatic cell differentiation [GO:0003309]; type II pneumocyte differentiation [GO:0060510] |
11914369_role in regulating WNT7b promoter 11937486_Differentiation of embryonic stem cells is induced by GATA factors 11959831_GATA6 regulates differentiation of distal lung epithelium. 15542856_is required for the induction of Gata-4 and Gata-6, and the interplay among these transcription factors plays a crucial role in parietal endoderm differentiation 15550397_GATA-6 can act as a positive or negative regulator of smooth muscle-specific gene expression 15591218_GATA-6 regulates a set of genes associated with vascular smooth muscle synthetic functions. GATA-6 regulates multiple genes involved in cell-cell signaling & cell-matrix interactions, as well as endothelin-1 & the AT1a receptor genes. 15767668_GATA6 alone is available for liver bud growth and commitment of the endoderm to a hepatic cell fate 16137232_expression of GATA-4 and GATA-6 is up-regulated prior to the transcriptional activation of Nkx 2.5 during cardiogenesis 16162334_GATA-6 is required to sense morphogenic (retinoic acid) signal; collaboration between GATA-6 and GATA-4, or GATA-6 and GATA-5 is involved in the perception of differentiation cues by embryonic stem cells in their determination of endoderm lineage 16199866_Expression of LMCD1/dyxin is remarkably similar to that of GATA6, with high-level expression observed in distal airway epithelium of the lung, vascular smooth muscle, and myocardium. 16199874_Loss of Hey1/2 leads to elevated GATA4/6 and ANF mRNA levels in embryoid bodies, while forced expression of Hey factors strongly represses expression of the GATA4 and GATA6 promoter. 16510470_Data show that Hop is expressed in airway epithelium coincident with HDAC2, and acts downstream of Nkx2.1 and GATA6, to negatively regulate surfactant protein expression. 16557299_GATA-6 regulates semaphorin 3C and is required in cardiac neural crest for cardiovascular morphogenesis 16621466_Upregulation by GATA6 in differentiating embryonal carcinoma cells is mediated by the direct binding of GATA-factor(s) to the Wnt2 promoter, suggesting Wnt2 is an immediate downstream target of GATA-factor regulation during early cardiogenesis. 16847256_GATA4 and GATA6 have roles in the developing cardiovascular system 16887115_At e17.5, transgenic Gata6-Engrailed embryos exhibit two distinct phenotypes: a complete absence of pancreas or a reduction in pancreatic tissue. This study suggests that Gata6 is an important regulator of pancreas specification. 17142311_Lungs from embryos carrying a missense mutation in Gata6 were structurally normal, while lungs from embryos carrying mutations of either Gata4 or of both Gata4 and Gata6 had a structural phenotype that matched the Fog2 mutant phenotype. 17605826_These findings indicate that Gata transcription factors 4 and 6 are sufficient for the derivation and propagation of XEN-like extra-embryonic endoderm cells from embryonic stem cells. 17615266_Nanog can directly repress Gata6 expression through its binding to the proximal promoter region of the Gata6 gene. 18191120_The G-protein coupled receptor is a target of GATA-6 regulation in differentiating embryonal carcinoma cells and that the expression domains of the two genes overlap within precartilaginous condensations. 18212046_GATA6 is expressed in Sertoli cells but does not bind to Rhox5 proximal promoter. 18400219_GATA4 or GATA6 are essential for expression of the network of transcription factors that regulate the onset of cardiac myocyte gene expression during mammalian development. 18536717_Gata6-regulated Wnt signaling controls the balance between progenitor expansion and epithelial differentiation required for both lung development and regeneration. 18816845_GATA6 is essential for the formation of primitive endoderm 18832096_GATA4/6 are responsible for activation of P450c17 gene expression at this early stage of embryonic development. 19084512_These findings highlight the unique genetic interactions of Gata4 and Gata6 with Tbx5 for normal cardiac morphogenesis in vivo. 19666519_mutations in GATA6 are genetic causes of congenital heart diseases involving outflow tract defects, as a result of the disruption of the direct regulation of semaphorin 3C-plexin A2 signaling 20159597_Wnt2 works in a feedforward transcriptional loop with Gata6 to regulate posterior cardiac development. 20531244_Results suggest that alterations in gene expression of Gata6, Tspan8, S100a8, and Lmo2 may act via novel pathways that play functionally important roles in Men1-associated tumor progression. 20705924_GATA-6 is both necessary and sufficient for regulating the cardiac hypertrophic response and differentiated gene expression, both alone and in coordination with GATA-4 20925113_The results suggest that GATA6 is downstream of Grb2 in the inductive signaling pathway and the expression of GATA6 is sufficient to compensate for the defects caused by Grb2 deficiency in the development of the primitive and extraembryonic endoderm. 21126317_Muc5b is a target gene of transcription factors (TTF-1, GATA-6) involved in lung differentiation programs during development and carcinogenesis. TTF-1 is a strong repressor of Muc5b. 21127043_GATA6 plays a crucial role for endothelial cell function and survival, at least in part, by suppressing autocrine TGFbeta expression and ALK5-dependent signaling. 21262227_GATA transcription factors are required for crypt cell proliferation, secretory cell differentiation, and absorptive enterocyte gene expression in the small intestinal epithelium 21514437_GATA-6 acts as a transcriptional repressor of CAV1 gene expression in partial bladder outlet obstruction-induced bladder wall smooth muscle hypertrophy 21839733_compound Gata4/Gata5 and Gata5/Gata6 mutants die embryonically or perinatally due to severe congenital heart defects 21908788_Reveal a previously uncharacterized phenotypic heterogeneity in the myocardium and suggest that decreased gap junctional coupling leads to induction of a signaling pathway that utilizes a unique GATA6 enhancer. 21954113_Post-transcriptional repression of Gata6 expression by Unr contributes to the stabilization of the embryonic stem cells pluripotent state. 22434075_These results demonstrate that GATA-4 and GATA-6 are needed for normal ovarian function. 22607194_GATA6 and FOXA2 bind the Wnt6 promoter and regulate its activity, leading to primitive endoderm formation. 22713603_Bmi1 overlaps with the nascent Gata6 and Nanog protein from the eight-cell stage onward before it preferentially cosegregates with Gata6 in primitive endoderm progenitors 22733991_In the colon, Gata6 deletion resulted in shorter, wider crypts, a decrease in proliferation, and a delayed crypt-to-surface epithelial migration rate. 22855527_Results suggest that GATA-6 promotes cell survival by regulating endoderm expression of BMP-2. 23002247_Gata6 is required for complete acinar differentiation and maintenance of the exocrine pancreas in adult mice. 23006325_Loss of Gata6 in the pancreas caused mild pancreatic defects. Loss of both Gata4 & Gata6 caused pancreatic agenesis, disrupted progenitor cell proliferation, disturbed branching morphogenesis, & failed to induce CPA1+/NEUROG3+ progenitor cells. 23006330_Double Gata4/Gata6 mutants failed to develop pancreata, died shortly after birth, & displayed hyperglycemia. Loss of only 1 Gata6 allele on a Gata4 conditional knockout background severely reduced pancreatic mass. 23275439_The study characterized the CPI-17 promoter and identified binding sites for GATA-6 and nuclear factor kappa B (NF-kappaB). 23471215_GATA6 regulates the differentiation of steroidogenic progenitors into adrenocortical cells. 24064357_Gene expression and cellular function in granulosa cells of mice with knockouts at GATA4, GATA6, or GATA 4/6 are reported. 24391969_GATA-4 and GATA-6 play a dosage-dependent and redundant role in programming cardiac hypertrophy. 24415953_conditional loss of Shh in limb buds lacking GATA6 prevents development of hindlimb polydactyly in these compound mutant embryos. 24662306_Piceatannol inhibits the development of cardiac hypertrophy through regulation of the expression and activity of transcriptional factor GATA6. 24762537_Selective deficiency of Gata6 in myeloid cells caused substantial alterations in the transcriptome of peritoneal macrophages. Gata6 deficiency also resulted in dysregulated peritoneal macrophage proliferative renewal during homeostasis and in response to inflammation, which was associated with delays in the resolution of inflammation. 24792964_Study identified retinoic acid as a signal that induces tissue-specific localization and functional polarization of peritoneal macrophages through the reversible induction of transcription factor GATA6. 24835466_Gata6 mutants exhibit a complete absence of primitive endoderm and GATA6 levels regulate the timing and speed of lineage commitment within the inner cell mass. 24899573_GATA4 and GATA6 proteins are fundamental regulators of granulosa cell differentiation and proliferation, and consequently of proper follicular assembly during normal ovarian development and function. 24929016_GATA4 and GATA6 play an essential role in maintaining proper intestinal epithelial structure and in regulating intestinal epithelial cytodifferentiation. 24952462_GATA6 creates an environment permissive for CRC initiation by lowering the threshold of BMP signalling required for tumour stem cell expansion 24970044_Gata6 is expressed in proximal airway epithelium throughout lung development. 25015078_These results establish SHP as a novel antihypertrophic regulator that acts by interfering with GATA6 signaling. 25024137_Gata6 regulates differentiation, metabolism, and survival of resident peritoneal macrophages through aspartoacylase. 25127677_In KRAS-mutant mice, nicotine promotes pancreatic carcinogenesis and tumor development via down-regulation of Gata6 to induce acinar cell dedifferentiation. 25209243_In the absence of Gata6, all inner-cell-mass cells adopt an epiblast identity. 25294942_GATA transcription factors are repressors of hedgehog signaling, and NKX3.2 maintains the ability of sclerotomal cells to express SHH transcriptional targets in the presence of BMP signals by repressing the induction of Gata4/5/6 25495347_GATA6 likely regulates enteroendocrine cell differentiation cell autonomously whereas GATA4 affects this population indirectly 25596178_In the pancreas, Gata6 acts as a tumour suppressor by enforcing acinar cell differentiation, by directly and indirectly repressing ectopic differentiation programmes, and by regulating crucial cancer-related gene expression pathways. 25613430_GATA6 contributes to the development and postnatal function of the murine atrioventricular node by promoting cell-cycle exit of specified cardiomyocytes toward a conduction system lineage. 25668066_Combined loss of GATA4 and GATA6 leads to a lack of testis functionality, with a loss of normal steroidogenic testis function, concomitant with an expansion of the adrenal-like cell population in postnatal conditional double mutant testes. 25933105_Mice with double deletion of Gata4 and Gata6 lack identifiable adrenal glands. Inactivation of Gata6 alone drastically reduced the adrenal size and corticosterone production in the adult animals. 25969542_data indicated that GATA-6 and Akt2 are involved in the mTORC1-mediated regulation of VSMC proliferation and differentiation. 26023924_The role of GATA4 in the regulation of cardiomyocyte maturation in the newborn heart and in maintaining diastolic function in the adult heart. 26109048_Gata6 is a versatile and potent reprograming factor that can act alone to drive a cell fate switch from diverse cell types 26505174_Our results, for the first time, portray a pivotal role of GATA6 in regulating metastatic behaviors of breast cancer cells 26510866_GATA4 and GATA6 are essential for female fertility, whereas targeting either factor alone causes subfertility. GATA4 and GATA6 are also required for the expression of the receptors for prolactin and luteinizing hormone. 26932670_GATA4/6-mediated inhibition of hedgehog signaling is a major mechanism regulating pancreatic endoderm specification during patterning of the gut tube 27289030_these findings indicated that the downregulation of GATA-6 effectively inhibited airway inflammation and reversed airway remodeling via Cav-1 27601327_Findings indicate that the Gata6 promoter is activated by Esrrb in association with Ncoa3, and Dax1 inhibited activities of Esrrb and Ncoa3, resulting maintenance of the undifferentiated status of embryonic stem (ES) cells. 27908934_The authors find that Gata6 protects against DNA damage associated with proliferation, thus preventing cell cycle arrest and apoptosis. 28504705_During wound healing differentiated Gata6(+) cells migrate from the sebaceous duct into the interfollicular epidermis and dedifferentiate, acquiring the ability to undergo long-term self-renewal and differentiate into a much wider range of epidermal lineages than in undamaged tissue. 28710293_GATA4/6 are key regulators of steroidogenesis and testicular somatic cell survival. 28952437_These findings identify Gata6-directed differentiation of neural crest to vascular smooth muscle cells as an essential mechanism that specifies the aortic tree, and provide a new framework for how mutations in GATA6 lead to congenital heart disorders in humans. 29197504_results support a model in which Gata6 contributes to repression of Isl1 expression in the anterior of nascent hindlimb buds. 29263149_Data (including data from studies in knockout mice and transgenic mice) suggest that pancreatic islets in adult mice deficient in GATA6 activity exhibit decreased insulin content and impaired insulin secretion. Gata6-deficient mice are apparently normal at birth; as they age they develop glucose intolerance. 29335067_Gata6 could be utilized as a biomarker to select the best Pluripotent stem cell (PSC) lines to produce PSC-derived cardiomyocytes. 29339785_The long non-coding antisense transcript of GATA6 (GATA6-AS) interacts with the epigenetic regulator LOXL2 to regulate endothelial gene expression via changes in histone methylation. A number of angiogenesis- and hypoxia-related genes were downregulated by GATA6-AS silencing. Silencing of GATA6-AS diminishes TGF-beta2-induced endothelial-mesenchymal transition in vitro and promotes formation of blood vessels in mice. 29445086_Etv5 can positively regulate primitive endoderm specification through regulating Gata6 and negatively regulate epiblast specification by inhibiting Fgf5 expression. 29454706_Phosphorylation of GATA6 by Erk1/2 compels exit from pluripotent state, and the phosphorylation propels a GATA6 positive feedback regulatory circuit to compel primitive endoderm differentiation 29567669_GATA6 haploinsufficiency leads to right-left type bicuspid aortic valve by disrupting valve remodeling and extracellular matrix composition. 30033638_Altogether, these findings support a role for Gata6 in maintaining proper upper hair follicle morphology. 30567484_role of TCF7L2 in regulation of vascular smooth muscle cell plasticity and protection against arterial disease. 31120883_The results suggested a second heart field regulatory network comprising of Gata4, Gata6 and SHh-signaling for outflow tract development. 32157212_Tumor progression and chromatin landscape of lung cancer are regulated by the lineage factor GATA6. 32907838_EZH2 Regulates Pancreatic Cancer Subtype Identity and Tumor Progression via Transcriptional Repression of GATA6. 33443158_GATA6 is a regulator of sinus node development and heart rhythm. 33479343_A genome-wide transcriptomic analysis of embryos fathered by obese males in a murine model of diet-induced obesity. 33846140_A GATA6-centred gene regulatory network involving HNFs and DeltaNp63 controls plasticity and immune escape in pancreatic cancer. 33876316_Fibroblast GATA-4 and GATA-6 promote myocardial adaptation to pressure overload by enhancing cardiac angiogenesis. 34332615_Variants in a cis-regulatory element of TBX1 in conotruncal heart defect patients impair GATA6-mediated transactivation. 34374415_GATA6 Deficiency Leads to Epithelial Barrier Dysfunction and Enhances Susceptibility to Gut Inflammation. 34911964_Directly recruited GATA6 + peritoneal cavity macrophages contribute to the repair of intestinal serosal injury. |
ENSG00000141448 |
GATA6 |
24.554281 |
5.4101411780 |
2.435666 |
0.56796125 |
18.065093 |
0.00002134795965719802791492505644743715720323962159454822540283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001483449459390695209046878710879013851808849722146987915039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.9263337312339 |
16.1274001706693 |
7.37990607219102 |
2.43358871039052 |
| ENSMUSG00000006204 |
71395 |
Cdcp3 |
protein_coding |
Q8BZE1 |
|
Disulfide bond;Glycoprotein;Reference proteome;Repeat;Signal |
|
|
mmu:71395; |
membrane [GO:0016020]; scavenger receptor activity [GO:0005044] |
|
|
|
28.006626 |
0.0193211059 |
-5.693679 |
0.99590134 |
41.980313 |
0.00000000009219698504355233885718158678214710285980793003091093851253390312194824218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000014768642706422470179554641165828310356289421179099008440971374511718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.983649027712413 |
0.734246594314605 |
50.910596499981 |
15.9565542597436 |
| ENSMUSG00000006221 |
29818 |
Hspb7 |
protein_coding |
P35385 |
|
Chaperone;Cytoplasm;Direct protein sequencing;Nucleus;Reference proteome;Stress response |
|
|
mmu:29818; |
actin cytoskeleton [GO:0015629]; aggresome [GO:0016235]; Cajal body [GO:0015030]; cytoplasm [GO:0005737]; mitochondrion [GO:0005739]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; filamin binding [GO:0031005]; heart development [GO:0007507]; response to heat [GO:0009408] |
20871982_These results imply that HspB2 and HspB7 might be up-regulated by ethanol during the short-term differentiation of neural stem cells. 22785082_HSPB7 is a potential early biomarker after MI and serves as an independent risk factor of ACS in patients with acute chest pain. 26929074_this paper shows that skeletal-muscle-specific ablation of the HspB7 causes postnatal death owing to a respiration defect, with progressive myopathy phenotypes in the diaphragm 27632998_these data indicate that MEF2 and AP-1 confer antagonistic regulation of Hspb7 gene expression in skeletal muscle, with implications for autophagy and muscle atrophy. 28827800_our findings characterize HSPB7 as an intercalated disc protein and suggest it has an essential role in maintaining intercalated disc integrity and conduction function in the adult heart. 29078393_The authors found that deleting HSPB7 resulted in uncontrolled elongation of actin filaments and the formation of atypical actin filament bundles in cardiomyocytes. |
ENSG00000173641 |
HSPB7 |
253.765537 |
0.2196159025 |
-2.186946 |
0.20275025 |
112.867736 |
0.00000000000000000000000002306596437861374651624182025255272657375605480608860213993516879067713094136893303698343515861779451370239257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000013724248805275179593293475016166549692895589663396212618248011690103016491093512740917503833770751953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
90.2812458882343 |
12.7295493514519 |
411.087015412937 |
40.3867753169228 |
| ENSMUSG00000006342 |
71733 |
Susd2 |
protein_coding |
Q9DBX3 |
FUNCTION: May be a cytokine receptor for GPR15LG. May be a tumor suppressor; together with GPR15LG has a growth inhibitory effect on colon cancer cells which includes G1 cell cycle arrest (By similarity). May play a role in breast tumorigenesis (PubMed:23131994). {ECO:0000250|UniProtKB:Q9UGT4, ECO:0000269|PubMed:23131994}. |
3D-structure;Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Sushi;Transmembrane;Transmembrane helix;Tumor suppressor |
|
|
mmu:71733; |
extracellular space [GO:0005615]; plasma membrane [GO:0005886]; negative regulation of cell cycle G1/S phase transition [GO:1902807]; negative regulation of cell division [GO:0051782] |
36266363_SUSD2 suppresses CD8+ T cell antitumor immunity by targeting IL-2 receptor signaling. |
ENSG00000099994 |
SUSD2 |
29.428082 |
2.4736417587 |
1.306637 |
0.49393726 |
6.792288 |
0.00915524715639815314660587830530857900157570838928222656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0310585132931529318833785424658344709314405918121337890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.5432296905865 |
13.4554109681984 |
15.5815730210144 |
4.26223808416402 |
| ENSMUSG00000006362 |
12398 |
Cbfa2t3 |
protein_coding |
O54972 |
FUNCTION: Transcriptional corepressor which facilitates transcriptional repression via its association with DNA-binding transcription factors and recruitment of other corepressors and histone-modifying enzymes. Can repress the expression of MMP7 in a ZBTB33-dependent manner. Reduces the protein levels and stability of the transcriptinal regulator HIF1A; interacts with EGLN1 and promotes the HIF1A prolyl hydroxylation-dependent ubiquitination and proteasomal degradation pathway. Contributes to inhibition of glycolysis and stimulation of mitochondrial respiration by down-regulating the expression of glycolytic genes including PFKFB3, PFKFB4, PDK1, PFKP, LDHA and HK1 which are direct targets of HIF1A (By similarity). Regulates the proliferation and the differentiation of erythroid progenitors by repressing the expression of TAL1 target genes (PubMed:16407974). Plays a role in granulocyte differentiation (PubMed:15231665). {ECO:0000250|UniProtKB:O75081, ECO:0000269|PubMed:11533236, ECO:0000269|PubMed:15231665, ECO:0000269|PubMed:16407974}. |
Alternative splicing;Coiled coil;Differentiation;Golgi apparatus;Membrane;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repressor;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:12398; |
Golgi membrane [GO:0000139]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; metal ion binding [GO:0046872]; transcription corepressor activity [GO:0003714]; granulocyte differentiation [GO:0030851]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of glycolytic process [GO:0045820]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032436]; regulation of aerobic respiration [GO:1903715]; response to hypoxia [GO:0001666] |
16287841_association with T-cell acute lymphocytic leukemia 1, ETO-2 represses gene expression in the early stages of erythroid differentiation 16407974_The dynamics of ETO2 recruitment within nuclear complexes couple cell proliferation to cell differentiation and determine the onset of terminal erythroid maturation. 18039847_MTG8/ETO and Mtg16 (also known as ETO2) associated with TCF4 18625887_These data suggest that ETO2 directly represses inappropriate early expression of a subset of terminally expressed megakaryocyte genes by binding to GATA1 and SCL. 18710942_the transcriptional corepressor Mtg16 (myeloid translocation gene on chromosome 16), which is targeted by t(16;21) in acute myeloid leukemia, is required for hematopoietic progenitor cell fate decisions and for early progenitor cell proliferation. 19799863_Eto2 and Mtgr1 as authentic interaction partners of Tal1 and suggest they act as heteromeric corepressors of this bHLH transcription factor during erythroid differentiation. 20123979_Data suggest that MTG16 interfaces with critical components of the Notch transcription complex to affect Notch-dependent lineage allocation in hematopoiesis. 21536648_Mtg16 integrates the activities of signaling pathways and nuclear factors in the establishment of T-cell fate specification. 22833394_MTG16 is critical for colonocyte survival and regeneration in response to intestinal injury and provide evidence that this transcriptional corepressor regulates inflammatory recruitment in response to injury. 24980046_Mtg16 promotes plasmacytoid dendritic cell differentiation and restricts classical dendritic cell development in part by repressing Id2. 25573176_MTG16 loss promotes radioresistance and impacts intestinal stem cell function, possibly due to shifting cellular response away from DNA damage-induced apoptosis and towards DNA repair after injury. 25957903_gene deficient mice have decreased levels of systemic monocytes and infarct-associated macrophages and display compromised tissue healing and post-MI heart failure 30560907_Study reveals a complex pattern of gene regulation by SCL, ETO2 and RYBP. Genome-wide analyses shows locus-specific binding of ETO2 and RYBP on SCL's 778 activated and repressed genes. 31015254_The pluripotency regulator PRDM14 requires the master hematopoietic regulator CBFA2T3 to initiate leukemia in progenitor cells, demonstrating an oncogenic role for CBFA2T3 and providing an avenue for targeting cancer-initiating cells. 32960220_Embryonic erythropoiesis and hemoglobin switching require transcriptional repressor ETO2 to modulate chromatin organization. 35503250_MTG16 regulates colonic epithelial differentiation, colitis, and tumorigenesis by repressing E protein transcription factors. |
ENSG00000129993 |
CBFA2T3 |
28.798808 |
0.0839536759 |
-3.574263 |
0.62132288 |
32.904919 |
0.00000000967780848100870328120040749463048879164261961705051362514495849609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001172731732559463438066610667040445825648475874913856387138366699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.17084886785981 |
1.85229588703703 |
49.6803602900171 |
13.6937313176786 |
| ENSMUSG00000006398 |
107995 |
Cdc20 |
protein_coding |
Q9JJ66 |
FUNCTION: Required for full ubiquitin ligase activity of the anaphase promoting complex/cyclosome (APC/C) and may confer substrate specificity upon the complex. Is regulated by MAD2L1: in metaphase the MAD2L1-CDC20-APC/C ternary complex is inactive and in anaphase the CDC20-APC/C binary complex is active in degrading substrates. The CDC20-APC/C complex positively regulates the formation of synaptic vesicle clustering at active zone to the presynaptic membrane in postmitotic neurons. CDC20-APC/C-induced degradation of NEUROD2 induces presynaptic differentiation. {ECO:0000269|PubMed:19900895}. |
Acetylation;Cell cycle;Cell division;Cytoplasm;Cytoskeleton;Differentiation;Direct protein sequencing;Isopeptide bond;Mitosis;Neurogenesis;Phosphoprotein;Reference proteome;Repeat;Ubl conjugation;Ubl conjugation pathway;WD repeat |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:107995; |
anaphase-promoting complex [GO:0005680]; centrosome [GO:0005813]; cytosol [GO:0005829]; mitotic checkpoint complex [GO:0033597]; nucleoplasm [GO:0005654]; perinuclear region of cytoplasm [GO:0048471]; protein-containing complex [GO:0032991]; spindle pole [GO:0000922]; anaphase-promoting complex binding [GO:0010997]; enzyme binding [GO:0019899]; histone deacetylase binding [GO:0042826]; protein C-terminus binding [GO:0008022]; ubiquitin ligase activator activity [GO:1990757]; anaphase-promoting complex-dependent catabolic process [GO:0031145]; cell differentiation [GO:0030154]; cell division [GO:0051301]; mitotic sister chromatid cohesion [GO:0007064]; mitotic spindle assembly [GO:0090307]; mitotic spindle assembly checkpoint signaling [GO:0007094]; negative regulation of ubiquitin-protein transferase activity [GO:0051444]; nervous system development [GO:0007399]; positive regulation of anaphase-promoting complex-dependent catabolic process [GO:1905786]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of synapse maturation [GO:0090129]; positive regulation of synaptic plasticity [GO:0031915]; positive regulation of ubiquitin protein ligase activity [GO:1904668]; protein ubiquitination [GO:0016567]; regulation of dendrite development [GO:0050773]; regulation of meiotic nuclear division [GO:0040020] |
12710943_This cell division cycle protein is a key regulator of the cell cycle. 15280373_The seven tandem WD motifs of Cdc20 they are required for speriolin binding and for localization of Cdc20 to the centrosomes and nucleus, suggesting that speriolin might regulate or stabilize the folding of Cdc20 during meiosis in spermatogenic cells 16600213_Data suggest that Mad2 and BubR1 must cooperate to inhibit Cdc20 activity. 16878123_Cdc20 is degraded through two independent degradation signals (degrons), the KEN box and a newly described CRY box. 17325031_Cdc20 and securin double mutant embryos could not maintain the metaphase arrest, suggesting a role of securin in preventing mitotic exit 17372985_Expression of the cdc20 gene is down-regulated by zif268 in neuronal cells; altered expression of proteasome-regulatory genes following zif268 induction may be a key component of long-lasting CNS plasticity. 18000406_Cdc20 is required for the anaphase onset of the first meiosis but not the second meiosis in mouse oocytes 18059335_findings suggest a novel function of HSF1 frequently overexpressed in cancer cells, to inhibit APC/C activity by interacting with Cdc20, and to result in aneuploidy development and genomic instability 19154723_Data show that the BubR1 N terminus binds to Cdc20 to inhibit APC/C activity in interphase, thereby allowing accumulation of cyclin B in G(2) phase prior to mitosis onset. 19528295_Results indicate that the spindle assembly checkpoint-mediated inhibition of Cdc20 is an important tumor-suppressing mechanism. 20053638_Results indicate that Cdc20 also contributes to post-anaphase activation of the APC/C. 20941357_The physiologically effective threshold level of Cdc20 is high for female meiosis I. 20956380_Cdc20 hypomorphism causes chromatin bridging and chromosome misalignment, revealing a requirement for Cdc20 in efficient sister chromosome separation and chromosome-microtubule attachment. 29719258_PPM1K Regulates Hematopoiesis and Leukemogenesis through CDC20-Mediated Ubiquitination of MEIS1 and p21. 30613277_Enforced expression of CDC20 in cardiomyocytes and in the heart aggravated the hypertrophic response. Furthermore, the authors found that CDC20 directly targeted LC3, a key regulator of autophagy, and promoted LC3 ubiquitination and degradation by the proteasome, which inhibited autophagy leading to cardiac hypertrophy. 32458533_CDC20 inhibitor Apcin inhibits embryo implantation in vivo and in vitro. 32666501_Biallelic mutations in CDC20 cause female infertility characterized by abnormalities in oocyte maturation and early embryonic development. 34298015_Oscillation of Cdc20-APC/C-mediated CAMDI stability is critical for cortical neuron migration. 34382737_CDC20 promotes bone formation via APC/C dependent ubiquitination and degradation of p65. 35660272_Conditional knockout of Cdc20 attenuates osteogenesis in craniofacial bones. 35913887_Germline Missense Variants in CDC20 Result in Aberrant Mitotic Progression and Familial Cancer. 36354207_Septin 9 controls CCNB1 stabilization via APC/C[CDC20] during meiotic metaphase I/anaphase I transition in mouse oocytes. |
ENSG00000117399 |
CDC20 |
561.417271 |
2.2628409844 |
1.178135 |
0.12531725 |
87.065752 |
0.00000000000000000001049726731242642407961187470858870089776080448428563493610925216881213373198988847434520721435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000004207746729023138760503358289606192563992277127127468461242898456475813873112201690673828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
782.155174161924 |
68.0260499218706 |
345.651850731229 |
23.1083741933318 |
| ENSMUSG00000006538 |
16147 |
Ihh |
protein_coding |
P97812 |
FUNCTION: [Indian hedgehog protein]: The C-terminal part of the indian hedgehog protein precursor displays an autoproteolysis and a cholesterol transferase activity (By similarity). Both activities result in the cleavage of the full-length protein into two parts followed by the covalent attachment of a cholesterol moiety to the C-terminal of the newly generated N-product (By similarity). Both activities occur in the reticulum endoplasmic (By similarity). Plays a role in hedgehog paracrine signaling. Associated with the very-low-density lipoprotein (VLDL) particles to function as a circulating morphogen for endothelial cell integrity maintenance (By similarity). {ECO:0000250|UniProtKB:Q14623, ECO:0000250|UniProtKB:Q62226}.; FUNCTION: [Indian hedgehog protein N-product]: The dually lipidated indian hedgehog protein N-product is a morphogen which is essential for a variety of patterning events during development. Binds to the patched (PTCH1) receptor, which functions in association with smoothened (SMO), to activate the transcription of target genes (By similarity). Plays a role in morphogenesis of the skeleton by coordinating growth and differentiation of the endochondral skeleton (PubMed:10465785, PubMed:10631175). Positively regulates PTHLH during endochondral bone formation preventing chondrocyte hypertrophy. In contrast, Ihh is necessary for normal chondrocyte proliferation in a PTHLH-independent pathway (PubMed:10631175). {ECO:0000250|UniProtKB:Q15465, ECO:0000250|UniProtKB:Q62226, ECO:0000269|PubMed:10465785, ECO:0000269|PubMed:10631175}. |
Autocatalytic cleavage;Calcium;Cell membrane;Developmental protein;Endoplasmic reticulum;Glycoprotein;Golgi apparatus;Hydrolase;Lipoprotein;Membrane;Metal-binding;Palmitate;Protease;Reference proteome;Secreted;Signal;Transferase;Zinc |
|
|
mmu:16147; |
endoplasmic reticulum membrane [GO:0005789]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi membrane [GO:0000139]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cholesterol-protein transferase activity [GO:0140853]; patched binding [GO:0005113]; peptidase activity [GO:0008233]; very-low-density lipoprotein particle binding [GO:0034189]; bone resorption [GO:0045453]; branching involved in blood vessel morphogenesis [GO:0001569]; camera-type eye development [GO:0043010]; camera-type eye photoreceptor cell fate commitment [GO:0060220]; cartilage development [GO:0051216]; cell fate specification [GO:0001708]; cell maturation [GO:0048469]; cell population proliferation [GO:0008283]; cell-cell signaling [GO:0007267]; chondrocyte differentiation [GO:0002062]; chondrocyte differentiation involved in endochondral bone morphogenesis [GO:0003413]; chondrocyte proliferation [GO:0035988]; embryonic camera-type eye morphogenesis [GO:0048596]; embryonic digestive tract morphogenesis [GO:0048557]; embryonic digit morphogenesis [GO:0042733]; embryonic pattern specification [GO:0009880]; embryonic skeletal joint development [GO:0072498]; endochondral bone morphogenesis [GO:0060350]; epithelial cell morphogenesis [GO:0003382]; epithelial cell-cell adhesion [GO:0090136]; head morphogenesis [GO:0060323]; heart looping [GO:0001947]; in utero embryonic development [GO:0001701]; intein-mediated protein splicing [GO:0016539]; liver regeneration [GO:0097421]; maternal process involved in female pregnancy [GO:0060135]; morphogenesis of a branching structure [GO:0001763]; multicellular organism growth [GO:0035264]; negative regulation of alpha-beta T cell differentiation [GO:0046639]; negative regulation of apoptotic process [GO:0043066]; negative regulation of chondrocyte differentiation [GO:0032331]; negative regulation of eye pigmentation [GO:0048074]; negative regulation of immature T cell proliferation in thymus [GO:0033088]; negative regulation of T cell differentiation in thymus [GO:0033085]; neuron development [GO:0048666]; ossification [GO:0001503]; osteoblast differentiation [GO:0001649]; pancreas development [GO:0031016]; pattern specification process [GO:0007389]; positive regulation of alpha-beta T cell differentiation [GO:0046638]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of collagen biosynthetic process [GO:0032967]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of mesenchymal cell proliferation [GO:0002053]; positive regulation of smoothened signaling pathway [GO:0045880]; positive regulation of T cell differentiation in thymus [GO:0033089]; positive regulation of transcription by RNA polymerase II [GO:0045944]; protein autoprocessing [GO:0016540]; proteoglycan metabolic process [GO:0006029]; regulation of gene expression [GO:0010468]; regulation of growth [GO:0040008]; response to estradiol [GO:0032355]; response to mechanical stimulus [GO:0009612]; retinal pigment epithelium development [GO:0003406]; self proteolysis [GO:0097264]; skeletal system development [GO:0001501]; smooth muscle tissue development [GO:0048745]; smoothened signaling pathway [GO:0007224]; somite development [GO:0061053]; vasculature development [GO:0001944]; vitelline membrane formation [GO:0030704] |
11977981_Indian hedgehog as a progesterone-responsive factor mediating epithelial-mesenchymal interactions in the mouse uterus. 11984876_Data suggest that Grg5 modulates the activities of transcription factors in vivo to affect Ihh expression and the function of long bone growth plates. 12050144_Indian hedgehog control differentiation of growth plate chondrocytes at multiple steps. 12217324_neither Shh nor Ihh is required for mammary gland morphogenesis and functional differentiation 12351698_Our results suggested that Ihh is regulated by P4, and the Ihh signaling axis may play a role in the preparation of the uterus for implantation during the periimplantation period. 12361605_FGF signaling interacts with the Ihh pathway in regulating chondrocyte proliferation and hypertrophic differentiation. 12435628_Hh signaling is essential for organizing ventral cell pattern, possibly through the control of differential cell affinities. 12917489_model of the interactions between beta-catenin and hedgehog signaling in the epidermis in which SHH promotes proliferation of progenitors of the hair lineages whereas IHH stimulates proliferation of sebocyte precursors 14981086_results suggest expression of the Ihh gene, which contains multiple Smad binding sites, may be finely regulated by a gradient of BMP concentrations. 15576404_Hh and Wnt signaling have roles in the regulation of osteoblast development 15689378_Indian hedgehog synchronizes skeletal angiogenesis and perichondrial maturation with cartilage development 15766752_Data suggest that tight control of Hedgehog pathway (Indian hedgehog and Sonic hedgehog) activity throughout embryonic development ensures proper pancreas organogenesis. 15771625_Upregulation of Ihh mRNA expression in the developing growth plate. 15878962_Prominent role of hedgehog signaling in the communication between granulosa cells and developing theca cells. 15951842_Mosaic ablation of the PTH/PTHrP receptor in the growth plate caused upregulation of Ihh action, PTHrP upregulation, acceleration of periarticular chondrocyte differentiation, and elongation of the columnar region. 16141219_Ihh signal determines the osteoblast versus chondrocyte fate in the mesenchymal progenitors; Gli3 is a critical effector for Ihh activity in the developing skeleton. 16284117_the Ihh/Gli3 system regulates two distinct steps of chondrocyte differentiation: (1) repression of the switch from distal into columnar chondrocytes; (2) the transition from proliferating into hypertrophic chondrocytes 16707121_These studies showed that genetic loss of Shh is accompanied by an up-regulation of Indian Hedgehog (Ihh) and maintenance of hedgehog pathway activity. 16818445_Embryos double-heterozygous for Wnt9a and beta-catenin show reduced Ihh expression. 16905129_Here, we report that disruption of both IGF and Ihh signaling resulted in additive reduction in the size of the embryonic skeleton.Thus, IGF and Ihh signaling appear to control the growth of the skeleton in parallel pathways. 16935278_The cranial base appears to be a skeletal structure in which growth and ossification patterns along its antero-posterior axis are orchestrated by both Ihh and Shh. 16951680_Ihh is an essential mediator of progesterone action in the uterus, and expression of this factor is critical in mediating the communication between the uterine epithelium and stroma required for embryo implantation 17028112_implicate the hedgehog signaling pathway in the pathogenesis of the placental defects in NSDHL deficiency 17191253_Temporomandibular joint formation and condyle growth require Indian hedgehog signaling. 17257418_proliferative changes in the intestinal mucosa were accompanied by increased expression of Indian hedgehog and activation of hedgehog signals 17343742_In a prostate cancer model, selectively increased Ihh expression accompanies increased progenitor cell markers & decreased terminal differentiation markers. Ihh may mark a progenitor cell population involved in tumor development. 17360475_the subset of MM cells that manifests Hh pathway activity is markedly concentrated within the tumor stem cell compartment 17392501_results demonstrate a functional Indian hedgehog(HH) pathway in the follicle and identify granulosa cells as at least one of the potential targets of HH signaling 17409191_Indian Hedgehog produced by postnatal chondrocytes is essential for maintaining a growth plate and trabecular bone. 17442891_These results suggest that Ihh regulates osteoblast differentiation of mesenchymal cells through up-regulation of the expression and function of Runx2 by Gli2. 17660199_Evc is an intracellular component of the hedgehog signal transduction pathway that is required for normal transcriptional activation of Ihh target genes. 17670790_Both Indian hedgehog (IHH) and parathyroid hormone-related protein (PTHrP, PTHLH) negatively regulate the transition from preosteoblastic progenitors to osteoblasts. 18059015_Indian hedgehog, expressed in pre-hypertrophic cells and collagen type X, expressed in hypertrophic chondrocytes, were both significantly inhibited after incubation with isoproterenol 18083924_Ihh is a critical and possibly direct regulator of joint development. In its absence, distribution and function of Gdf5-expressing interzone-associated cells are abnormal 18183793_Leptin may affect chondrocyte proliferation and differentiation by activating the PTHrP/Ihh growth-restraining feedback loop in the postnatal growth plate. 18239353_LIF, which has a crucial role in E2 action for initiation of implantation, causes transient induction of Pgr mRNA and subsequent upregulation of Ihh mRNA, which mediates progesterone-Pgr actions for successful implantation 18272352_Polydactyly in the mouse mutant Doublefoot involves altered Gli3 processing and is caused by a large deletion in cis to Indian hedgehog 18292212_These studies support a model in which hedgehog activates elaboration and differentiation of mammary progenitors via differential TP63 promoter selection and forfeiture of self-renewing capacity. 18434416_Results revealed a novel role of Ihh signaling in promoting chondrocyte hypertrophy independently of PTHrP, which is particularly important in postnatal cartilage development and homeostasis. 18582859_This study identifies Ihh as a novel choroid-derived signal that regulates retinal pigment epithelium, sclera and neural retina development. 18682398_Msx2 promotes the maturation of chondrocytes, at least in part, through up-regulating Ihh expression 19035497_the IHH-parathyroid hormone-related protein axis participates in the maintenance of articular cartilage 19109233_differentiation from double-negative to double-positive (DP) cell, and hence the size of the DP population, is dependent on the concentration of Ihh in the thymus 19252479_a Brachydactyly type A1 mutation (E95K) in Ihh impairs the interaction of IHH with PTCH1 and HIP1 19369399_CSPGs plays a role in modulating Ihh signaling in the developing growth plate, and highlights the importance of carbohydrate sulfation in regulating growth factor signaling. 19443245_Ihh knockout hematopoietic stem cells and progenitor cell populations are produced in normal numbers in vivo, but terminal erythroid differentiation is defective leading to fatal anemia in mid gestation in many Ihh knockout embryos. 19501129_the decreased level of VEGF and Indian hedgehog production in VE devoid G4N and G6N EBs attenuates definitive hematopoietic progenitor cell expansion. 19590577_The Gpc3-null mice show increased levels of Ihh protein and signalling, but similar levels of Ihh messenger RNA. 19761883_Ihh has both PTHrP-dependent and -independent functions during postnatal endochondral bone development. 19906842_Atf4 as a novel transcriptional activator of Indian hedgehog in chondrocytes that paces longitudinal bone growth 19906844_Gli2 and Gli3 collectively mediate all major aspects of Ihh function during endochondral skeletal development 20007344_Hedgehog signaling may be important stimulus for renal cyst formation. 20024692_Sostdc1, Penk1 and Igfbp5 were identified as possible new IHH signaling targets that may contribute to the molecular mechanism for BDA1. 20056671_Ihh regulates embryo implantation by having an impact on the cell cycle, EGF signaling, and E2 signaling. 20200412_continuous Ihh action is required for completion of post-natal temporomandibular joint growth and organization 20430747_Hh proteins act as paracrine mitogens to promote the expansion of adjacent mesenchymal progenitors, including those of the smooth muscle compartment. 20542037_Ihh regulates intestinal stem cell self-renewal and differentiation. Intestinal epithelial Ihh signals to mesenchymal cells to regulate formation and proliferation of mesenchymal cells, which in turn affect epithelial proliferation and differentiation. 20660756_Indian hedgehog and WNT/beta-catenin signaling, inhibited by receptor tyrosine kinase-like orphan receptor 2 (ROR2) in distal limb mesenchyme, are acting upstream of BMP signaling in the phalanx-forming region 20682322_Loss of Ihh initiates several events that are characteristic of an intestinal wound repair response. Prolonged loss resulted in progressive inflammation, mucosal damage, and the development of intestinal fibrosis. 21297010_Ihh signaling is essential for symphyseal cartilage development and anterior mandibular growth 21312270_IGF-1R in chondrocytes controls cell growth, survival, and differentiation in embryonic and postnatal growth plates in part by suppression of Pthrp expression. 21364421_The hedgehog pathway is activated during palatal fusion, and deletion of Indian hedgehog leads to diminished ossification of the secondary hard palate. 21557453_study defines Ihh as an important positive regulator of cranial bone ossification 21683695_Loss of muscle mass in Ihh null mouse embryos is accompanied by a dramatic increase in myoblast apoptosis by a loss of p21 protein 22155527_Indian hedgehog requires additional effectors besides Runx2 to induce osteoblast differentiation. 22190639_The data indicated that Atf4 in chondrocytes is required for normal Ihh expression and for its paracrine effect on osteoblast differentiation. 22879594_GABA(B)R1 positively regulates chondrogenesis through a mechanism relevant to the acceleration of nuclear translocation of ATF4 for Ihh expression in chondrocytes 23383321_Ihh signaling plays an important role in chondrocyte proliferation and differentiation via interactions with Runx2 and Runx3. 23665349_Data suggest that GPC3 down-regulates hepatocyte proliferation by binding to hedgehog (HH) and down-regulating the HH signaling pathway and binding with CD81, thus making it unavailable to bind to Hhex and causing its nuclear translocation. 23955340_Data indicate that the centrosomal protein DZIP1 interacts with GLI3 transcription factor in the cytoplasm and regulates Hedgehog signaling and ciliogenesis. 24149372_Sirt6 regulates postnatal growth plate differentiation and proliferation via Ihh signaling. 24428864_findings support the protective role of Ihh deletion in surgically induced osteoarthritis 24753031_Thyroid hormone regulates the secondary ossification center initiation and progression via differentiating chondrocytes into bone matrix-producing osteoblasts by stimulating Ihh and Osx expression in chondrocytes. 24786088_Increased Ihh is associated with the severity of osteoarthritis cartilage damage. 24821091_Cbfbeta functions in upregulating Ihh expression to promoter chondrocyte proliferation and osteoblast differentiation, and inhibiting PPR expression to enhance chondrocyte differentiation. 24844414_Indian Hedgehog, a critical modulator in osteoarthritis, could be a potential therapeutic target for attenuating cartilage degeneration disease. 24859162_Ihh signaling mediates gastrin-induced proliferation of epithelial cells in stomachs of adult mice. 25028519_Data indicate Indian hedgehog (Ihh) induces collagen type X COL10A1 expression and promotes its transcription through Runx2/Smad1 interactions on a specific promoter region. 25105964_C/EBPbeta and RUNX2 cooperatively stimulate expression of Ihh through direct interactions with a C/EBPbeta binding element, which further promotes hypertrophic differentiation of chondrocytes during the chondrocyte differentiation process. 25307863_Apc mutant epithelial cells secrete IHH to maintain an intestinal stromal phenotype that is required for adenoma development in mice. 25842978_increased expression in cell bodies of cutaneous sensory nerves of dorsal root ganglia as compared with skin epithelia 25917826_In organogenesis of the ovary, production of Dhh/Ihh in granulosa cells requires growth differentiation factor 9 from the oocyte. 26094741_Ihh has an important role in regulating limb mesenchymal cell differentiation 26553654_findings thus demonstrate that augmented Ihh signaling is detrimental to craniofacial development, and that finely tuned Ihh signaling is critical for temporomandibular joint formation. 26620087_Ihh and PTH1R signaling in limb mesenchyme are both essential to regulate proper development of digit structures, although they appear to use different mechanisms. 26918743_Ihh expression was downregulated in femur epiphyses of Hand1-overexpressing mice. Hand1 downregulated Ihh gene expression in vitro by inhibiting Runx2 transactivation of the Ihh proximal promoter. 26963384_The Annexin a2 Promotes Development in Arthritis through Neovascularization by Amplification Hedgehog Pathway. 27666725_An accelerated hypertrophic differentiation caused by a disturbed Ihh-PTHrP signaling pathway may lead to a higher bone mineral density in the vertebral bodies of newborn Col IX -/- mice and, as a result, to the early onset of disc degeneration. 27930311_loss of Spop, but not Spopl, disrupts chondrocyte hypertrophy and osteoblast differentiation in the mouse, suggesting the requirement for Spop-mediated protein degradation in mouse skeletal development; overexpressed Spop targets both Gli3FL and Gli3R for ubiquitination and degradation and Spop is an important positive regulator of Ihh signaling and skeletal development 28696225_GPC6 stimulates Hh signaling by binding to Hh and Ptc1 at the cilium and increasing the interaction of the receptor and ligand to promote the growth of developing long bones. 28749439_melatonin not only elevated progesterone (P) secretion, but also upregulated expressions of StAR and Cyp11a1 and also had an increased Ihh expression in endometrium. 28846100_Ihh is regulated by at least 9 enhancers with individual tissue specificities in the digit anlagen, growth plates, skull sutures and fingertips. Consecutive deletions, resulting in growth defects of the skull and long bones, showed that these enhancers function in an additive manner. Duplications caused dose-dependent upregulation and misexpression of Ihh, abnormal phalanges, fusion of sutures and syndactyly. 29244023_This work reveals the molecular function of Hedgehog signaling in extrinsic sheath tissues for tendon repair. 29545125_Variations in Ihh signaling activity contribute to the severity of the osteochondroma phenotype by affecting both, osteochondroma size and number, in distinct manners. 29771958_Noggin null mutants show increased Indian hedgehog (Ihh) expression within cartilage condensations that leads to aberrant extension of IHH downstream signaling into the interdigital mesenchyme. A converse phenotype with increased apoptosis and reduced cell proliferation was found in the interdigital mesenchyme of Ihh mutant embryos, suggesting IHH's unrecognized physiological role for IHH in interdigital webbing biology. 30032718_To define the function of Ihh in the mesenchymal cell's early stages of chondrogenesis, we specifically delete Ihh in Prx1-expressed mesenchyme cells at E9.5 using Prx1-Cre;Ihh(fl/fl);Rosa26(-ZsGreen1) mice. We found that deleting Ihh in the mesenchyme cells results in an early and quick ossification of the intermediate cartilage scaffold, causing the growth plate and phalange joint absence, short limbs, and dwarfishness 30470823_a mechanistic understanding for how Ihh signaling promotes HCC tumorigenesis specifically in obese mice 31382618_We demonstrated that the terminal differentiation process in deep zone chondrocytes promoted by FFSS and UAC was mediated by the enhanced Ihh signaling and declined PTH1R expression 31847127_The Roles of Indian Hedgehog Signaling in TMJ Formation. 32108165_Stromal Hedgehog pathway activation by IHH suppresses lung adenocarcinoma growth and metastasis by limiting reactive oxygen species. 32223017_Indian hedgehog in craniofacial neural crest cells links to skeletal malocclusion by regulating associated cartilage formation and gene expression. 32705199_MicroRNA-1 promotes cartilage matrix synthesis and regulates chondrocyte differentiation via post-transcriptional suppression of Ihh expression. |
ENSG00000163501 |
IHH |
40.932834 |
0.2126508856 |
-2.233441 |
0.42404923 |
27.588172 |
0.00000015009383246295066812929866213038865296880430832970887422561645507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000015385122853806624902277570485220614671106886817142367362976074218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.3964712106375 |
4.29272722238608 |
67.700029413528 |
13.5156072259463 |
| ENSMUSG00000006542 |
241113 |
Prkag3 |
protein_coding |
Q8BGM7 |
FUNCTION: AMP/ATP-binding subunit of AMP-activated protein kinase (AMPK), an energy sensor protein kinase that plays a key role in regulating cellular energy metabolism. In response to reduction of intracellular ATP levels, AMPK activates energy-producing pathways and inhibits energy-consuming processes: inhibits protein, carbohydrate and lipid biosynthesis, as well as cell growth and proliferation. AMPK acts via direct phosphorylation of metabolic enzymes, and by longer-term effects via phosphorylation of transcription regulators. AMPK also acts as a regulator of cellular polarity by remodeling the actin cytoskeleton; probably by indirectly activating myosin. The AMPK gamma3 subunit is a non-catalytic subunit with a regulatory role in muscle energy metabolism. It mediates binding to AMP, ADP and ATP, leading to AMPK activation or inhibition: AMP-binding results in allosteric activation of alpha catalytic subunit (PRKAA1 or PRKAA2) both by inducing phosphorylation and preventing dephosphorylation of catalytic subunits. ADP also stimulates phosphorylation, without stimulating already phosphorylated catalytic subunit. ATP promotes dephosphorylation of catalytic subunit, rendering the AMPK enzyme inactive. {ECO:0000250|UniProtKB:Q9UGI9}. |
Alternative splicing;ATP-binding;CBS domain;Fatty acid biosynthesis;Fatty acid metabolism;Lipid biosynthesis;Lipid metabolism;Nucleotide-binding;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:241113; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleotide-activated protein kinase complex [GO:0031588]; nucleus [GO:0005634]; AMP binding [GO:0016208]; AMP-activated protein kinase activity [GO:0004679]; ATP binding [GO:0005524]; protein kinase binding [GO:0019901]; protein kinase regulator activity [GO:0019887]; cellular response to glucose starvation [GO:0042149]; cellular response to nutrient levels [GO:0031669]; fatty acid biosynthetic process [GO:0006633]; glycogen biosynthetic process [GO:0005978]; glycolytic process [GO:0006096]; protein phosphorylation [GO:0006468]; regulation of catalytic activity [GO:0050790]; response to muscle activity involved in regulation of muscle adaptation [GO:0014873] |
14512293_Gamma3 subunit of AMPK is highly expressed in fast-twitch glycolytic skeletal muscle, and wild-type gamma3 functions in the regulation of alpha1 catalytic activity, but it is not associated with changes in muscle glycogen concentrations 14559719_the observation that the AMPKgamma3 isoform is expressed primarily in white skeletal muscle, in which it is the predominant gamma-isoform, strongly suggests that gamma3 has a key role in this tissue 15857891_functional studies of the protein kinase, AMP-activated, gamma 3 (AMPK3) isoform further support the close connection between glycogen content and exercise performance in skeletal muscle 16237515_AMPKg3 is involved in the coordinated transcription of genes critical for lipid and glucose metabolism in white glycolytic skeletal muscle. 16410251_analysis of transcriptional regulation in skeletal muscle of AMP-activated protein kinase gamma3 R225Q transgenic and knock-out mice 16638825_Expression of either wild type or mutant (R225Q) gamma3-subunit of AMPK results in increased glycogen concentrations in muscle, but the mechanisms underlying this alteration appear to be different. 18728219_AMPK heterotrimeric complexes containing the AMPKgamma(3) subunit may play a specific role in linking circadian oscillators and energy metabolism in skeletal muscle. 18818284_Basal and A-769662-mediated IL-6 release provide evidence for a role of AMPK in the regulation of IL-6 release from oxidative skeletal muscle. 18838377_AMPK gamma3 subunit is associated with mitochondrial biogenesis in glycolytic skeletal muscle, concomitant with increased expression of the co-activator PGC-1alpha and several transcription factors that regulate mitochondrial proteins 20023137_subunit R225Q mutation effect on growth performance with normal or high-energy diet 20197054_AMPK acts as the primordial trigger for fasting- and exercise-induced adaptations in skeletal muscle and that activation of SIRT1 and its downstream signaling pathways are improperly triggered in AMPK-deficient states. 21798999_gamma3-AMPK down-regulation is a rapid physiological muscle response observed in mouse after running exercise or fasting, two situations leading to PPARbeta activation. 26758685_The AMPKgamma3 isoform is dispensable for functional overload-induced muscle growth. 27797909_Data suggest that the AMPK-TBC1D4 signaling axis is likely mediating the improved muscle insulin sensitivity after contraction/exercise and illuminates an important and physiologically relevant role of AMPK in skeletal muscle. 33798773_Compound- and fiber type-selective requirement of AMPKgamma3 for insulin-independent glucose uptake in skeletal muscle. |
ENSG00000115592 |
PRKAG3 |
84.184834 |
2.3399126925 |
1.226455 |
0.27461196 |
19.812214 |
0.00000854350053965826230584299644243984062086383346468210220336914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000641057143941254367700693928000532650912646204233169555664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
120.88969413729 |
23.15013712056 |
51.6641900894818 |
7.67804848422289 |
| ENSMUSG00000007655 |
12389 |
Cav1 |
protein_coding |
P49817 |
FUNCTION: May act as a scaffolding protein within caveolar membranes (By similarity). Forms a stable heterooligomeric complex with CAV2 that targets to lipid rafts and drives caveolae formation. Mediates the recruitment of CAVIN proteins (CAVIN1/2/3/4) to the caveolae (PubMed:19546242). Interacts directly with G-protein alpha subunits and can functionally regulate their activity (By similarity). Involved in the costimulatory signal essential for T-cell receptor (TCR)-mediated T-cell activation. Its binding to DPP4 induces T-cell proliferation and NF-kappa-B activation in a T-cell receptor/CD3-dependent manner (By similarity). Recruits CTNNB1 to caveolar membranes and may regulate CTNNB1-mediated signaling through the Wnt pathway (PubMed:10816572). Negatively regulates TGFB1-mediated activation of SMAD2/3 by mediating the internalization of TGFBR1 from membrane rafts leading to its subsequent degradation (By similarity). {ECO:0000250|UniProtKB:Q03135, ECO:0000269|PubMed:10816572, ECO:0000269|PubMed:19546242}. |
Acetylation;Alternative initiation;Cell membrane;Direct protein sequencing;Golgi apparatus;Isopeptide bond;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:12389; |
acrosomal membrane [GO:0002080]; apical plasma membrane [GO:0016324]; basal plasma membrane [GO:0009925]; basolateral plasma membrane [GO:0016323]; caveola [GO:0005901]; caveolar macromolecular signaling complex [GO:0002095]; cell cortex [GO:0005938]; cell surface [GO:0009986]; cilium [GO:0005929]; cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; cytosol [GO:0005829]; endoplasmic reticulum [GO:0005783]; endosome [GO:0005768]; focal adhesion [GO:0005925]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; lipid droplet [GO:0005811]; membrane [GO:0016020]; membrane raft [GO:0045121]; mitochondrion [GO:0005739]; perinuclear region of cytoplasm [GO:0048471]; peroxisomal membrane [GO:0005778]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; ATPase binding [GO:0051117]; enzyme binding [GO:0019899]; identical protein binding [GO:0042802]; inward rectifier potassium channel inhibitor activity [GO:0070320]; kinase binding [GO:0019900]; molecular adaptor activity [GO:0060090]; nitric-oxide synthase binding [GO:0050998]; peptidase activator activity [GO:0016504]; protein heterodimerization activity [GO:0046982]; protein kinase binding [GO:0019901]; protein-containing complex binding [GO:0044877]; protein-macromolecule adaptor activity [GO:0030674]; receptor serine/threonine kinase binding [GO:0033612]; signaling receptor binding [GO:0005102]; small GTPase binding [GO:0031267]; SNARE binding [GO:0000149]; syntaxin binding [GO:0019905]; transmembrane transporter binding [GO:0044325]; angiogenesis [GO:0001525]; angiotensin-activated signaling pathway involved in heart process [GO:0086098]; apoptotic signaling pathway [GO:0097190]; basement membrane organization [GO:0071711]; calcium ion homeostasis [GO:0055074]; calcium ion transport [GO:0006816]; caveola assembly [GO:0070836]; caveolin-mediated endocytosis [GO:0072584]; cell differentiation [GO:0030154]; cell population proliferation [GO:0008283]; cellular calcium ion homeostasis [GO:0006874]; cellular nitric oxide homeostasis [GO:0033484]; cellular response to exogenous dsRNA [GO:0071360]; cellular response to hyperoxia [GO:0071455]; cellular response to mechanical stimulus [GO:0071260]; cellular response to peptide hormone stimulus [GO:0071375]; cellular response to transforming growth factor beta stimulus [GO:0071560]; cellular senescence [GO:0090398]; cholesterol efflux [GO:0033344]; cholesterol homeostasis [GO:0042632]; cytokine-mediated signaling pathway [GO:0019221]; endocytosis [GO:0006897]; endothelial cell proliferation [GO:0001935]; establishment of localization in cell [GO:0051649]; fibroblast proliferation [GO:0048144]; glandular epithelial cell differentiation [GO:0002067]; insulin receptor internalization [GO:0038016]; lactation [GO:0007595]; lipid storage [GO:0019915]; maintenance of protein location in cell [GO:0032507]; mammary gland development [GO:0030879]; mammary gland involution [GO:0060056]; MAPK cascade [GO:0000165]; membrane depolarization [GO:0051899]; microtubule polymerization [GO:0046785]; muscle cell cellular homeostasis [GO:0046716]; negative regulation of anoikis [GO:2000811]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of calcium ion import across plasma membrane [GO:1905949]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cation channel activity [GO:2001258]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cytokine-mediated signaling pathway [GO:0001960]; negative regulation of endothelial cell proliferation [GO:0001937]; negative regulation of epithelial cell differentiation [GO:0030857]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; negative regulation of fibroblast proliferation [GO:0048147]; negative regulation of inward rectifier potassium channel activity [GO:1903609]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of MAPK cascade [GO:0043409]; negative regulation of muscle cell apoptotic process [GO:0010656]; negative regulation of necroptotic process [GO:0060546]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of nitric oxide biosynthetic process [GO:0045019]; negative regulation of nitric-oxide synthase activity [GO:0051001]; negative regulation of peptidyl-serine phosphorylation [GO:0033137]; negative regulation of peptidyl-tyrosine autophosphorylation [GO:1900085]; negative regulation of pinocytosis [GO:0048550]; negative regulation of potassium ion transmembrane transport [GO:1901380]; negative regulation of protein binding [GO:0032091]; negative regulation of protein tyrosine kinase activity [GO:0061099]; negative regulation of protein ubiquitination [GO:0031397]; negative regulation of receptor signaling pathway via JAK-STAT [GO:0046426]; negative regulation of signal transduction [GO:0009968]; negative regulation of smooth muscle cell migration [GO:0014912]; negative regulation of smooth muscle cell proliferation [GO:0048662]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512]; negative regulation of tyrosine phosphorylation of STAT protein [GO:0042532]; negative regulation of vascular associated smooth muscle cell proliferation [GO:1904706]; nitric oxide biosynthetic process [GO:0006809]; positive regulation of calcium ion transport into cytosol [GO:0010524]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; positive regulation of catalytic activity [GO:0043085]; positive regulation of cell adhesion molecule production [GO:0060355]; positive regulation of cell migration [GO:0030335]; positive regulation of cholesterol efflux [GO:0010875]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of endocytosis [GO:0045807]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of ER-associated ubiquitin-dependent protein catabolic process [GO:1903071]; positive regulation of extrinsic apoptotic signaling pathway [GO:2001238]; positive regulation of gap junction assembly [GO:1903598]; positive regulation of gene expression [GO:0010628]; positive regulation of intrinsic apoptotic signaling pathway [GO:2001244]; positive regulation of microtubule polymerization [GO:0031116]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of protein binding [GO:0032092]; positive regulation of protein ubiquitination [GO:0031398]; positive regulation of signal transduction [GO:0009967]; positive regulation of toll-like receptor 3 signaling pathway [GO:0034141]; positive regulation of vasoconstriction [GO:0045907]; post-transcriptional regulation of gene expression [GO:0010608]; protein localization [GO:0008104]; protein transport [GO:0015031]; receptor internalization [GO:0031623]; receptor internalization involved in canonical Wnt signaling pathway [GO:2000286]; receptor-mediated endocytosis of virus by host cell [GO:0019065]; regulation of blood coagulation [GO:0030193]; regulation of cell communication by electrical coupling involved in cardiac conduction [GO:1901844]; regulation of cytosolic calcium ion concentration [GO:0051480]; regulation of entry of bacterium into host cell [GO:2000535]; regulation of fatty acid metabolic process [GO:0019217]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of membrane repolarization during action potential [GO:0098903]; regulation of peptidase activity [GO:0052547]; regulation of ruffle assembly [GO:1900027]; regulation of smooth muscle contraction [GO:0006940]; regulation of the force of heart contraction [GO:0002026]; regulation of the force of heart contraction by chemical signal [GO:0003057]; regulation of ventricular cardiac muscle cell action potential [GO:0098911]; response to bacterium [GO:0009617]; response to calcium ion [GO:0051592]; response to estrogen [GO:0043627]; response to hypoxia [GO:0001666]; response to ischemia [GO:0002931]; response to mechanical stimulus [GO:0009612]; response to progesterone [GO:0032570]; skeletal muscle tissue development [GO:0007519]; T cell costimulation [GO:0031295]; triglyceride metabolic process [GO:0006641]; tyrosine phosphorylation of STAT protein [GO:0007260]; vasculogenesis [GO:0001570]; vasoconstriction [GO:0042310]; wound healing [GO:0042060] |
11498544_Caveolin-1 and caveolae play a fundamental role in organizing multiple signaling pathways in the cell. 11739396_Our results show for the first time a clear role for caveolins in systemic lipid homeostasis in vivo and place caveolin-1/caveolae as major factors in hyperlipidemias and obesity 11845324_CAV1 was found in liver endothelial cells and in Kupffere cells (liver non-parenchymal cells). 11857686_In astrocytes, the process of transformation is not sufficient to reduce levels of caveolin-1;further,caveolin-1 expression is not sufficient to prevent the acquisition of a transformed phenotype. 11980479_Connexin family members target to lipid raft domains and interact with caveolin-1. 11997523_Intracellular retention of glycosylphosphatidyl inositol-linked proteins in caveolin-deficient cells. 12036959_phosphorylation of tyrosine by insulin receptor 12057923_Dual ablation of both Cav-1 and Cav-3 genes in mice leads to a pleiotropic defect in caveolae formation and severe cardiomyopathy. 12134086_Results indicate a central role for caveolin-1 in promoting cellular senescence and suggest the hypothesis that premature senescence may represent a tumor suppressor function mediated by caveolin-1 in vivo. 12135605_IGF-I induces caveolin 1 tyrosine phosphorylation and translocation in the lipid rafts. 12167625_caveolin-1 plays a dual regulatory role in controlling microvascular permeability: as a structural protein required for caveolae and as a tonic inhibitor of eNOS activity 12177436_Defects cause dilated cardiomyopathy and pulmonary hypertension in knockout mice 12203725_caveolin-1 is expressed in developing mouse ovary and is involved in lipid regulation and signal transduction 12368209_data clearly implicate loss of functional Cav-1 in the pathogenesis of mammary epithelial cell hyperplasia, and suggest that Cav-1-null mice represent a novel animal model to study premalignant mammary disease 12388746_Data show that caveolin-1 expression blocks prolactin-induced activation of a STAT5a (signal transducer and activator of transcription)-responsive luciferase reporter in mammary epithelial cells. 12496259_The adipocyte plasma membrane caveolin functional/structural organization is necessary for the efficient endocytosis of GLUT4. 12531427_expression of c-Abl is required for oxidative stress-induced caveolin-1 phosphorylation. 12631721_Results show that loss of caveolin-1 gene expression dramatically accelerates the development of these multifocal dysplastic mammary lesions. 12651616_Disruption of this gene impairs renal calcium reabsorption and leads to hypercalciuria and urolithiasis. 12660144_caveolin-1 acts as a molecular chaperone that is necessary for the proper stabilization of the insulin receptor in adipocytes in vivo 12692121_Caveolin-1 and caveolin-3 act as a molecular chaperone rather than a plasma membrane scaffold for AT1-R through the exocytic pathway. 12759258_Results provide the first genetic evidence that caveolin-1 indeed functions as a tumor suppressor gene in vivo. 12759260_Results provide novel evidence that Cav-1 and caveolae play an important positive role in the process of pathological angiogenesis in vivo. 12777465_increased levels of caveolin-1 characterize the apoptotic phenotype of macrophages and may be involved in the efficient externalization of PS at the surface of the apoptotic cells 12921776_Neutral ceramidase secreted by endothelial cells is released in part associated with caveolin-1. 14561756_Caveolin-1, by inhibiting cPLA2, may interfere with synaptic facilitation and long term potentiation formation in the hippocampus. 14563650_Toassess the in vivo role of Cav-1 in atherosclerosis, we interbred Cav-1-/- mice with ApoE-/- mice. Loss of Cav-1 caused a doubling of non-HDL plasma cholesterol in ApoE-/- background, but loss of Cav-1 was y protective against aortic atheromas. 14647059_caveolins may play an important role in regulating hepatic BS and cholesterol metabolism. 14690422_This is the first study to demonstrate dramatic effects of loss of Cav-1 gene expression and caveolae organelles on the long-term survival of an organism; loss of both Cav-1 alleles is required to mediate a reduction in life span. 14962949_In IDDM, there is an increase of caveolin-1 expression that leads to eNOS activity inhibition reducing NO production & promoting & stabilizing the insulin receptor in its active conformation, a body-adaptive response to a reduction in insulin production. 15020298_Caveolin-1 alpha preferentially sensitizes L929 cells to TNF-alpha through the activation of a PI3K/Akt signaling pathway. 15044451_Cav-1 cooperates with the tumor suppressor INK4a genetic locus to prevent cell proliferation and oncogene-induced tumorigenesis 15111495_Caveolin-1 important for efficient lipid droplet formation. Caveolin-1 null embryonic fibroblasts stably transfected with perilipin accumulated approximately 4.5-fold less lipid than perilipin-transfected wild-type cells. 15138262_caveolin-1 gene transcription is differentially regulated in lung type I epithelial and endothelial cell lines by ETS proteins 15182174_sterol carrier protein-2 selectively interacted with caveolin-1, both within the cytoplasm and at the plasma membrane 15205364_Caveolin-1 expression is critical for vascular endothelial growth factor-induced ischemic hindlimb collateralization and nitric oxide-mediated angiogenesis. 15222744_caveolin-1 has a role in preventing neointima formation during vascular injury 15355971_Cav-1 has a role in mammary tumorigenesis, cell invasiveness and matrix metalloproteinase (MMP-2/9) secretion 15380628_the loss of caveolin-1 is associated with disruption of M3 muscarinic cholinergic activity in the bladder 15509552_prion replication triggers an abnormal localization of caveolin 1 and synaptophysin, which in turn may alter neuronal function 15613480_PS1 and PS2 affect caveolin 1 trafficking in an indirect manner and are required for caveolae formation by controlling transport of intracellular caveolin 1 to the plasma membrane. 15686485_Caveolin-1 plays an active role in the regulation of injury-induced synaptic and terminal remodeling in the adult central nervous system. 15691837_Results demonstrate that a branched signaling pathway involving MEK, ERK, PKCepsilon, PKCalpha, and caveolin-1 regulates collagen expression in normal lung tissue and is perturbed during fibrosis. 15699039_Results suggest that insulin receptor expression and ligand-dependent signaling are independent of caveolin-1 expression. 15734843_Molecular genetic evidence that caveolin-1 plays a critical functional and structural role in the modulation of thermogenesis via an effect on lipid mobilization is presented. 15748889_ACTH induces caveolin-1 phosphorylation and podosome assembly in Y1 cells and supports the view that the morphological and functional responses to PKA activation in steroidogenic cells are related to cytoskeleton dynamics. 15802273_Cav-1 functions as a tumor promoter during prostate carcinogenesis, rather than as a tumor suppressor 15919660_CAV1 is not required for cholesterol absorption or ezetimibe sensitivity in the mouse 16113676_Lipid raft internalization is mediated by dynamin-2 and caveolin-1. Internalization requires phosphorylation of caveolin-1 on Tyr 14. 16166342_cav1 knockout affects nitric oxide function in both longitudinal and circular muscle, but its effects in circular muscle differ significantly 16183667_results demonstrate the essential requirement of caveolin-1 in mediating the formation of caveolae in endothelial cells in vivo and in negatively regulating interendothelial junction permeability 16205123_Impaired macrophage phagocytosis in Cav-1 knockout mice has implications for altered innate immunity against pathogens, the regulation of inflammatory responses, and the development of autoimmune disease. 16260622_caveolin-1 does not play a major role in lipid raft-mediated biosynthetic membrane trafficking 16294019_Loss of Caveolin-1 (Cav-1) induces the accumulation of mammary stem cells, and that this event may be an initiating factor during mammary tumorigenesis. 16294037_Ablation of Cav-1 gene expression induces an abnormal amplification of crypt stem cells, resulting in increased susceptibility to gamma-radiation. 16299308_Caveolin-1 is up-regulated in response to LPS. 16319533_acute fatal T. cruzi myocarditis is accompanied by changes in cell cycle proteins such as the cyclins and caveolin and that the upregulation of the endothelin pathway may be important in the myocardial abnormalities and mortality observed 16357362_Caveolin-1 acts as a potent immunomodulatory effector molecule in immune cells and that the regulation of LPS-induced cytokine production by caveolin-1 involves the MKK3/p38 MAPK pathway. 16380094_Findings in cav-1 knockout mice hemodynamic measurements suggest a causative role of a sustained nitrosative stress in the development of pronounced cardiac impairment. 16405917_These novel findings provide insight into possible signaling mechanisms of Nf1 and suggest that together Nf1 and Cav-1 may coordinately regulate cell growth and differentiation. 16415072_Cav-1 ablation increased the endothelin-1 induced Ca(2+) response in smooth muscle cells by altering the type and expression level of the endothelin receptor 16416023_Protein kinase C mediated endothelial barrier permeability regulation depends on caveolin-1 16418573_Study shows that loss of Cav-1 in mammary epithelial cells induces the baseline constitutive hyper-activation of STAT5a signaling, which normally controls the temporal progression of lactogenesis in the mammary gland. 16423895_colocalization of caveolin-1 and GLUT4 is not an absolute requirement for muscle glucose metabolism but reduction in GLUT4 could be contributing to the insulin resistance observed in ERalpha(-/-) mice 16428350_cav-1 is required for physiological Ca(2+) spark and transient K(Ca) current regulation in cerebral artery smooth muscle cells 16601228_Critical role of caveolin in ensuring caveolae-mediated endocytosis of CXCR4, regulating both SDF-1-mediated mobilization and peripheral homing of progenitor cells in response to ischemia. 16603689_caveolin-1 plays an important role in the regulation of intracellular cholesterol homeostasis 16670766_caveolae/Cav-1 may act as sensors of altered shear stress [commentary] 16670769_endothelial Cav-1 and caveolae are necessary for both rapid and long-term mechanotransduction in intact blood vessels 16684506_These results suggest that caveolin-1 facilities the lymphatic metastasis ability of mouse hepatocarcinoma cells via regulation tumor cell growth and VEGFA expression. 16702020_Cav-1 expression leads to an increased proportion of highly glycosylated-CD147 relative to lowly glycosylated-CD147, increased production of MMP-11 and a higher invasive capability in hepatocarcinoma cells 16702023_data provide evidence that caveolin-1 is necessary to target FAT/CD36 to the plasma membrane; caveolin-1 may influence fatty acid uptake by regulating surface availability of FAT/CD36 16708022_From these results, we suggest that the up-regulated caveolin-1 in neuronal cells can inhibit the bFGF signaling pathway from small GTPases to PAK1 by directly binding to PAK1. 16714282_Caveolin-1 has a role as a novel Cdc42 guanine nucleotide dissociation inhibitor in pancreatic beta-cells 16723714_Cav-1-deficient mammary acini displayed increased ERalpha levels and enhanced sensitivity toward estrogen-stimulated growth, with specific up-regulation of cyclin D1 16750274_cav-1 is involved in maintaining cortico-striato-pallido-thalamo-pontine pathways associated with motor control 16782699_The results of the present study show that the function of beta-adrenoceptors is reduced in cav1(-/-) small intestine likely owing to reduced PKA activity. 16844343_p38alpha can negatively modulate Akt activity, independently of PI3K, by regulating the interaction between caveolin-1 and PP2A through a mechanism dependent on cell attachment. 16904002_caveolae and caveolins are integral membrane components in basal and ciliated epithelial cellsin rats, mice, and humans, indicating a crucial role in these cell types; in addition to their physiological role, they may be involved in airway infection 16931572_The interaction of the GLP-1R with caveolin-1 regulates subcellular localization, trafficking, and signaling activity. 16973879_caveolin-1 plays a crucial role in the mechanisms that coordinate lipid metabolism with the proliferative response occurring in the liver after cellular injury 16982844_Caveolin-1-deficient mice are sensitive to S. enterica serovar Typhimurium. 17023578_caveolin-1 loss is a critical step in CCL2-induced modulation of brain microvascular endothelial cell junctional protein expression and integrity, and maypossibly serve a crucial role in regulating inflammation at the blood-brain barrier 17071600_Loss of Cav-1 dramatically exacerbated mammary lobulo-alveolar hyperplasia in cyclin D1 transgenic mice, whereas overexpression of Cav-1 caused reversion of this phenotype. 17098833_Cav-1 ablation reduces pressure-induced depolarization and depolarization-induced Ca(2+) influx, and these effects combine to produce a diminished arterial wall [Ca(2+)](i) elevation and constriction. 17102036_Data show that protein kinase C-mediated contraction and Rho activation but not calcium sensitization are increased in smooth muscle following genetic ablation of caveolin-1. 17200204_skeletal muscle fibers from male Cav-1(-/-) mice show striking abnormalities, such as tubular aggregates, mitochondrial proliferation/aggregation, and increased numbers of M-cadherin-positive satellite cells 17202321_Thyrocytes of caveolin-1 knockout mice display enhanced proliferation. 17291449_Cholesterol levels thus regulate fatty acid uptake by adipocytes via a pathway that does not involve altered cell surface localization of caveolin-1 or CD36. 17293479_Cav-1 knockout (KO) mice show a marked increase of cerebral volume of infarction, as compared with wild-type and Cav-2 KO mice. 17317261_These results suggest that Cav-1 may play an important role in the normal development of immune responses in Trypanosoma cruzi infection. 17348685_These results suggest a functional interaction between VEGFR-3 and caveolin-1 to modulate endothelial cell activation during angiogenesis. 17390339_Transcriptional regulation of caveolin-1 differs markedly between lung epithelial and endothelial cell lines. 17416589_Cav-1 is required for calcium entry in vascular endothelial cells 17418092_This study clearly suggests that Cav-3 expression is causally linked to the maturation of muscle phenotype and it is tightly regulated by hypertrophic and atrophic stimuli. 17446044_Cav1 phosphorylation is required for stretch-induced Egfr and Akt activation in mesangial cells. 17448744_in MC3T3-E1 cells caveolin-1 was enriched in the matrix vesicle fraction as well as the caveolar membrane fraction, and the amount of caveolin-1 in the matrix vesicle fraction increased as differentiation progressed 17469800_The formation of LMW-PTP/caveolin-1 complexes is accompanied by modulation of the enzyme activities, and the inhibitory effect elicited against IF1 is stronger than that against IF2 17478448_These data suggested that loss of caveolin-1 is associated with abnormal re-epithelialization in lung fibrosis. 17498208_There is an interaction between P2X7 protein and Caveolin-1 in lipid rafts. 17517963_Establishes cell polarity during directional migration through coordination of the signaling of Src kinase and Rho GTPases. 17543885_expression of Cav-1 leads to the decreased cellular responsiveness to TGF-beta through down-regulating TbetaRII gene expression 17550335_caveolin-1 helps to maintain a less differentiated state of osteoblast progenitor cells, and the absence of caveolin-1 causes bone to mature more rapidly 17609206_EMP2 regulates caveolin-1 transcription and more substantially its protein levels. 17626133_nitric oxide affects blood pressure more in Cav-1 knockout than in wild type mice 17699617_insulin receptor form complexes with Cav1 and ganglioside GM3 independently 17715130_caveolin-1 contributes to the trafficking of EAAC1 on and off the plasma membrane 17786030_These results provide the first molecular genetic evidence that Cav-1 functions upstream of FASN during prostate cancer progression. 17893196_provide clear physiological evidence supporting the important role of cell type-specific Cav-1 expression governing multiple phenotypes in the vasculature, heart, and lung 17905724_Caveolin-1, perhaps via its ability to scaffold key signaling components, is essential for NMDAR localization to neuronal membrane rafts, NMDAR/Src tyrosine kinase family/ERK signaling, and protection of neurons from ischemic injury and cell death. 17916598_Caveolin-1 therefore has the potential to be a marker of tumor vasculature maturity that may help adjusting anticancer therapies. 17979883_MMP-2 is closely associated with caveolins at fibroblasts (FB) and capillary endothelial cells as well as at cardiomyocytes. Also, MMP-2 is closely associated with FAK at FB and c-Kit at interstitial Cajal-like cells. 17982011_Caveolin-1/caveolin-3 heterooligomeric complexes may play a key role in doxorubicin-induced cardiotoxicity in the atria. 17988240_Concurrently, caveolins 1 and 2, and estrogen receptor beta were down-regulated in differentiated GN11, whereas estrogen receptor alpha was unaffected by differentiation 17989209_Caveolin 1 contributes to low basal ERK1/2 and Akt activity and is required for Akt-dependent signals in response to shear stress (flow) but is not essential for trophic effects of stretch (pressure) in the vascular wall. 17993589_Caveolin-1 expression in polymorphonuclear neutrophils (PMNs) plays a key role in mediating PMN activation, adhesion, and transendothelial migration and in PMN activation-induced lung inflammation and vascular injury. 18056268_modulation of TGF-beta signaling by IL-4, via cav1, suggesting novel therapeutic targets for controlling the effects of TGF-beta and thereby ameliorating pathological airway remodeling. 18083242_Our data confirm a distinct behavioural phenotype in CavKO mice and indicate a selective alteration in central cholinergic function. 18096699_Caveolin-1 or Caveolin-3, but not specific caveolin mutants, inhibit endocytosis of dysferlin through a clathrin-independent pathway colocalizing with internalized glycosylphosphatidylinositol-anchored proteins. 18178722_endothelial Caveolin-1, likely through an effect on eNOS activity, plays a prominent role in the regulation of vascular function during substantial changes in dietary sodium intake 18191037_Our results demonstrate a significant role of Cav-1 in HDL-cholesterol metabolism and may reflect a variety of Cav-1 functions including modulation of ACAT activity and SR-BI function. 18192592_caveolin-1 null mice have elevated pulmonary vascular resistance that is attributed to remodeling of pulmonary precapillary vessels 18203815_low caveolin-1 levels in lungs that are fibrotic cause their overexpression of collagen, tenascin-C, and alpha actin 18207479_Caveolin-1, a transmembrane protein that negatively regulates PI3K/PKB and p44/42 MAPK signaling, was downregulated in alveolar macrophages from WT mice after ozone exposure. 18268148_caveolin-1 is required for EDHF-related relaxation by modulating membrane location and activity of TRPV4 channels and connexins, which are both implicated at different steps in the EDHF-signaling pathway 18323531_cav-1 deletion protects against hyperoxia-induced lung injury, involving in part the modulation of the HO-1-cav-1 interaction, and the enhanced induction of HO-1 through a p38 MAPK-mediated pathway. 18328807_These findings provide new insights about the potential role of caveolin-1 in the regulation of oxLDL-induced apoptosis in vascular cells and its contribution to the instability of the plaque. 18347068_Thus, transmembrane crosstalk between the galectin lattice and pY14Cav1 promotes FA turnover by stabilizing FAK within FAs defining previously unknown, interdependent roles for galectin-3 and pY14Cav1 in tumor cell migration. 18349137_Control of blood pressure variability in caveolin-1-deficient mice: role of nitric oxide identified in vivo through spectral analysis. 18358473_Loss of cav-1 function in stromal cells of various organs leads to a disorganized stromal compartment indirectly promoting abnormal growth and differentiation of adjacent epithelium. 18400048_The activation of nNOS, localized in smooth muscle caveolae, by calcium entering through L-type calcium channels triggers nitric oxide production which modulates muscle contraction by a cGMP-dependent mechanism. 18407780_The expression and localization of the P2X(4)R, was analyzed and the interaction between Cav-1 and both P2X(4)R and P2X(7)R in the mouse alveolar epithelial cell line, was characterized. 18417152_Dysfunctional eNOS is of central importance in the genesis of the cardiopulmonary phenotype of cav-1(-/-). 18508910_caveolin-1 plays an important role in the regulation of lipoprotein metabolism by controlling their plasma levels as well as their lipid composition. 18511851_A large component of pulmonary vascular hyperpermeability induced by activation of PMNs adherent to the vessel wall is dependent on signaling via caveolin-1. 18577581_Caveolin-1-dependent beta1 integrin and fibronectin endocytosis plays a critical role in fibronectin matrix turnover. 18583416_These data posit caveolin-1 as a component of the IGF/PI-3K/Akt signalling modulus regulating cellular proliferation with implications for diseases, including cancers, which have altered caveolin expression 18618588_Data show that expression of caveolin-1 in Hepa1-6 cells enhanced cell transformation capability both in vitro and in vivo and prevented actinomycin D-induced apoptosis via the activation of survivin-mediated survival pathway. 18635971_These findings suggest that FASN and Cav-1 physically and functionally interact in PCa cells. 18694845_role estradiol-17beta-induced proliferation of mouse embryonic stem cells; involvement of Src, PI3K/Akt, and MAPKs pathways 18703018_These results demonstrate that Cav-1 plays a critical role for glucose and sulfonylurea-stimulated insulin secretion 18719368_Targeted loss of Cav1 produces a unique model of cardiac hypertrophy with normal substrate utilization and expression of genes involved in energy metabolism. 18725205_These findings establish caveolin-1 as an important regulator of downstream signaling and membrane targeting of BMPRII in vascular smooth muscle cells. 18801924_Cav-1/Cysteine-rich 61 (Cyr61) interaction via integrins represents a novel pathway of Cyr61 signaling involving cav-1-dependent processes, which play a critical role in regulating hyperoxia-induced cell death 18820136_Cav1 knockout mice bladder displays significant contractile dysfunction. 18849439_the important role played by cav1, as part of the TGF-beta signaling pathway, in the regulation of the pulmonary ECM. 18951881_These findings suggest that eNOD plays a crucial role in the evolution of heart failure evident in caveolin-1 knockout mice. 19015640_Caveolin-1 was upregulated and beta-catenin was recruited to the plasma membrane when xCT was deficient, which were followed by the inhibition of beta-catenin transcriptional activity. 19038362_Reviews recent studies in adipocytes, the specialized cell type for fatty acid storage, which suggest a role for caveolins in the formation, maintenance or mobilization of lipid droplet stores. 19052258_Caveolin-1 scaffold domain interacts with TRPC1 and IP3R3 to regulate Ca2+ store release-induced Ca2+ entry in endothelial cells. 19068211_important role of caveolin-1 in penile erection 19074371_study identified that Cav1 does not contribute to detergent-resistant membranes &nondetergent raft domains but contributes to cell architecture-dependent raft domains in adherent cells; data suggests Cav1 plays role in lipid homeostasis 19103597_Caveolin-1 expression is required for the development of pulmonary emphysema through activation of the ATM-p53-p21 pathway. 19104007_GTPCH I is targeted to caveolae microdomains in vascular endothelial cells, and tetrahydrobiopterin production occurs in proximity to endothelial NO synthase. The regulation of GTPCH I activity involves the caveolar coat protein, caveolin-1. 19111727_Endothelial Cav-1 mediates angiogenesis in experimental colitis. 19164603_neutrophils traverse human dermal microvascular endothelial cells using a transcellular or paracellular route. Caveolin-1 expression favors the transcellular path 19166483_Data suggest that reduction of extracellular calcium associated with caveolae/caveolin-1 in myenteric plexus, the state of L-type Ca(2+) channels or an increase in the distance between caveolae and SR affects intestinal calcium handling. 19175701_Results describe the ultrastructure of interstitial Cajal-like cells (ICLC) in normal mammalian myocardium versus caveolin-1 null mice. 19265160_Cav-1 binds to Toll-like receptor 4 and inhibits lipopolysaccharide-induced proinflammatory cytokine production in murine macrophages. 19266163_galectin-1 increased DNA synthesis in mouse ES cells via Src, caveolin-1 Akt, and mTOR signaling pathways. 19286607_Akt-mediated transactivation of the S1P1 receptor in caveolin-enriched microdomains regulates endothelial barrier enhancement by oxidized phospholipids. 19309260_Data suggest that NOX1-derived reactive oxygen species regulate cell-surface expression of AT1R through mechanisms including caveolin phosphorylation. 19342371_Cav-1(-/-) null mice are a new preclinical model for studying the molecular paradigm of estrogen hypersensitivity and the development of estrogen-dependent ductal carcinoma in situ lesions. 19406948_Caveolin-1 loss of function accelerates glucose transporter 4 and insulin receptor degradation in 3T3-L1 adipocytes. 19411449_stromal caveolin-1 expression may be a potential therapeutic target and a valuable prognostic indicator of breast cancer progression. 19435853_A physiological function of caveolin-1 in the thyroid gland. 19438805_Caveolin-1 regulation of dynamin-dependent, raft-mediated endocytosis of cholera toxin-B subunit occurs independently of caveolae 19474194_It is concluded that in caveolin-1 KO mice the glomerular permeability was not increased, despite the presence of glomerular hyperfiltration. 19477952_Caveolin-1 and caveolae play a paradoxical role in regulating VEGF-induced ERK2/1 activation and in vitro angiogenesis 19487814_loss of caveolin-1 leads to hyperactive eNOS and subsequent tyrosine nitration-dependent impairment of PKG activity, which results in pulmonary hypertension. 19583953_The loss of Cav-1 reduces LDL infiltration into the artery wall, promotes nitric oxide production, and reduces the expression of leukocyte adhesion molecules, effects completely reversed in transgenic mice. 19620284_These results provide insight into the mechanisms regulating growth of cancer cells, which frequently lose Cav1 function. 19625610_EGF-induced DNA synthesis and cell migration are mediated by caveolin-1, which is activated by Src, FAK, PI3K/Akt, ERK, and MMP-2 signals in mouse ES cells. 19675140_Cav1 knockout and wild-type C57BL/6 mice are rendered diabetic with streptozotocin, and changes in motor nerve conduction velocity (MNCV), mechanical and thermal hypoalgesia and nerve fiber density are observed. 19696145_Caveolin-1 and lipid microdomains regulate Galpha(s) trafficking and signaling in a C6 glioma cell model system. 19713536_Phosphorylation of caveolin-1 regulates oxidant-induced pulmonary vascular permeability via paracellular and transcellular pathways. 19729487_Caveolin-1 is not necessary for vesiculo-vacuolar organelle (VVO) structure but may have important roles in regulating VVO function in acute vascular hyperpermeability and angiogenesis. 19729954_PPARgamma1-induced caveolin-1 enhances cholesterol efflux and attenuates atherosclerosis in apolipoprotein E-deficient mice. 19730165_LPS induced Cav1 overexpression in liver sinusoidal endothelial cells; KO of Cav1 increased basal eNOS activity and partially restored ET-1-mediated eNOS stimulation, eNOS translocation to plasma membrane and NO production in LSECs after the LPS treatment 19737975_Adult male PBcav-1 mice showed significantly increased prostatic wet weight & higher incidence of epithelial hyperplasia compared with nontransgenic littermates. Increased immunostaining for cav-1, etc. occurred in PBcav-1 hyperplastic prostatic lesions. 19747167_Results suggest that cav-1 acts as a molecular chaperone or scaffolding molecule in cholesterol-rich lipid rafts that may be necessary for the proper stabilization and activation of PDE3B in response to CL and insulin. 19767411_Deletion of cav-1 protects hyperoxia-induced apoptosis via modulation of survivin expression. 19854179_Data show that amino acids K47-K57 of Cav-1 are a highly conserved sequence in Cav-1 and Cav-3 proteins, and that expression of either K47-K57 deletion Cav-1 mutant or wild-type Cav-2 that lacks this sequence exhibits a non-polarized distribution pattern. 19891976_In capillaries in the brain striatum, expression of P-gp and caveolins was higher and lower, respectively, in mice with type 2 diabetes compared with non-diabetic mice 19923909_The subventricular zone of caveolin knockout mouse brains displays an increased proliferation of adult neural stem cells. 19949109_an important contribution of Cav-1 to innate host immunity to P. aeruginosa infection 19965594_Caveolin-deficient adipocytes can form only small lipid droplets, suggesting that the caveolin-lipid droplet pool might be involved in lipid droplet size regulation. 19966340_study investigated the contribution of Cav-1 to early TGF-beta signaling in regenerating liver after partial hepatectomy; data might help to unravel why Cav-1-deficient mice exhibit an accelerated liver regeneration after partial hepatectomy 20004673_Upregulation of caveolin-1 expression is associated with structural modifications of endothelial cells in diabetic lung. 20004972_In yolk sac, caveolin-1 is restricted to endothelial cells & smooth muscle cells of vitelline vasculature & to mesothelial cells. In placenta, caveolin-1 is expressed in endothelial cells but not in any of the three trophoblast layers. 20097717_may have a deleterious role in mediating cardiovascular damage, in part, by modulating signaling via the mneralocorticoid receptor/aldosterone pathway 20099006_Data demonstrate that over-expression of caveolin-1 aggravates the AT-1 injury induced by LPS, involving in modulation of the cPLA2 mediated by the cPLA2/p38 MAPK pathway. 20130741_Caveolin-1 is present in mouse ventricular cardiac myocytes as well as endothelial cells. 20141598_Comparison of fenestrations in wild-type and knockout mice did not reveal any differences on either scanning or transmission electron microscopy. 20167851_Cav1-related molecules have been detected at the mRNA or protein level in various nonexcitable tissues 20211253_caveolae is involved upstream in c-Src-MAPKs activation by 1alpha,25(OH)2D3 and that VDR and cav-1 participate in the rapid signaling elicited by the hormone 20299334_In obesity, a reduced expression of Cav-1 leads to greater contribution of the BK(Ca) channel to EDHF-mediated response, which seems essential for maintained coronary dilation. 20304961_eNOS activation secondary to loss of Cav1 signals dampening of the innate immune response to lipopolysaccharide through IRAK4 nitration and the resultant impairment of kinase activity, and consequently mitigates inflammatory lung injury 20345844_Decreased Cav-1 expression in fibrotic diseases likely leads to increased deposition of IGFBP-5 in the extracellular matrix. 20351069_Caveolin-1-dependent occludin endocytosis is required for TNF-induced tight junction regulation in vivo. 20363891_In the absence of cav-1, mineralocorticoid receptor activation plays a beneficial role in regulating eNOS expression/activity and, consequently, the vascular function during high salt diet. 20368353_The ERK/PPARgamma axis plays a key role in regulating integrin alpha1beta1-dependent Cav-1 expression and consequent EGFR-mediated reactive oxygen species production. 20381543_Data show that caveolin-1 ablation affected neither the G-protein coupled LPA receptor signaling to Erk1/2, nor the receptor tyrosine kinases PDGF- or EGF-receptor signaling to Erk1/2. 20399205_caveolin-1 overexpression yields defective par |
ENSG00000105974 |
CAV1 |
2115.281380 |
2.0381065158 |
1.027229 |
0.08308700 |
151.202018 |
0.00000000000000000000000000000000009467471932849079899394432885460234499904559124916672269896299760759026555207513948773538408829053558690702629974111914634704589843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000086352414659375557028731521692284846717787362548068997112711154974631272898879560788194115206195533573918510228395462036132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2801.59007645252 |
161.437038788031 |
1374.60434711433 |
60.0806134963393 |
| ENSMUSG00000008206 |
67260 |
Cers4 |
protein_coding |
Q9D6J1 |
FUNCTION: Ceramide synthase that catalyzes formation of ceramide from sphinganine and acyl-CoA substrates, with high selectivity toward long and very-long chains (C18:0-C22:0) as acyl donor. {ECO:0000269|PubMed:12912983, ECO:0000269|PubMed:15823095}. |
Endoplasmic reticulum;Glycoprotein;Lipid biosynthesis;Lipid metabolism;Membrane;Phosphoprotein;Reference proteome;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Lipid metabolism; sphingolipid metabolism. {ECO:0000269|PubMed:12912983, ECO:0000269|PubMed:15823095}. |
|
mmu:67260; |
endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; DNA binding [GO:0003677]; N-acyltransferase activity [GO:0016410]; sphingosine N-acyltransferase activity [GO:0050291]; ceramide biosynthetic process [GO:0046513]; sphingolipid biosynthetic process [GO:0030148] |
12912983_Lag1p family members trh1 and trh4 regulate (dihydro)ceramide synthases responsible for production of sphingolipids containing different fatty acids 23936132_Data indicate that oxidized phospholipids (OxPLs)-induced ceramide synthases (CerS1-Cers6) activity in macrophages is responsible for the accumulation of ceramide. 24738593_Loss of CerS4 protein leads to an altered lipid composition of the sebum, which is more solidified and therefore might cause progressive hair loss due to physical blocking of the hair canal. 25705848_CerS4-directed epidermal ceramide composition is essential to control hair follicle stem and progenitor cell behavior potentially through its regulation of BMP and Wnt signaling 26887952_These results suggest that the phosphorylation of ceramide synthases may be a key regulatory point in the control of the distribution and levels of sphingolipids of various acyl-chain lengths. 35163788_T-Cell-Specific CerS4 Depletion Prolonged Inflammation and Enhanced Tumor Burden in the AOM/DSS-Induced CAC Model. |
ENSG00000090661 |
CERS4 |
183.522228 |
3.3436308669 |
1.741416 |
0.39684103 |
18.252940 |
0.00001934271231214770400317314036353621986563666723668575286865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001357733612620432321269059450230543006910011172294616699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
283.190786264091 |
78.8831202619645 |
84.6955891774456 |
17.7679635365644 |
| ENSMUSG00000009633 |
14373 |
G0s2 |
protein_coding |
Q61585 |
FUNCTION: Promotes apoptosis by binding to BCL2, hence preventing the formation of protective BCL2-BAX heterodimers. {ECO:0000250}. |
Apoptosis;Mitochondrion;Reference proteome |
|
|
mmu:14373; |
mitochondrion [GO:0005739]; extrinsic apoptotic signaling pathway [GO:0097191]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of extrinsic apoptotic signaling pathway [GO:2001238] |
16086669_A novel putative target gene of PPARalpha, G0S2 (G0/G1 switch gene 2) was identified and characterized. 20197052_overexpression of G0S2 diminishes the rate of lipolysis in both adipocytes and adipose tissue explants 21989031_Lipolysis is differently regulated between perigonadal and mesenteric adipocytes, and these depot-specific differences might be explained by altered regulation of G0S2 and/or perilipin. 22039468_minimal fragment of ATGL could still be activated and inhibited by CGI-58 and G0S2, respectively 22693613_a novel model of HSC quiescence in which elevated G0S2 expression can sequester nucleolin in the cytosol, precluding its pro-proliferation functions in the nucleolus 22891293_Data indicate that downregulation of G0S2 in adipose tissue could represent one of the underlying causes leading to increased lipolysis in the insulin-resistant state. 24302733_fat-specific G0S2 overexpression uncouples adiposity from insulin sensitivity and overall metabolic health through inhibiting adipose lipolysis and decreasing circulating fatty acids. 24556704_Roles for G0S2 were found in lactation, energy balance, and thermogenesis. This study provides a basis for tumor suppressive effects of G0S2 by regulating lipolysis. 24583640_G0s2 has an important role in adipogenesis and accumulation of triacylglycerol. 24993166_Proteasomal degradation of PPAR-gamma and the reduction of g0s2 content are permissive for prolonged TNF-alpha induced lipolysis. 25381555_G0s2 is a physiological regulator of adiposity and energy metabolism and is a potential target in the treatment of obesity and insulin resistance. 25666096_G0S2 expression in naive CD8(+) T cells decreased immediately after T-cell receptor activation downstream of various signal transduction pathways. G0S2 inhibits energy production by oxidative phosphorylation to fine-tune proliferation in homeostasis. 26654706_findings do not rule out the possibility that G0S2 may be playing a role in other forms of leukemia, but clearly show that the commonly used Emu-Myc transgenic is not the correct model to conduct studies on G0s2 26837760_G0S2 silencing mediates MYC-induced oncogenesis in other malignancies 27248498_G0S2 protein but not mRNA levels were reduced in the adipose tissue of ATGL-deficient mice, corroborating the involvement of ATGL in the stabilization of G0S2 28910567_ER+ breast cancer cells with restored G0S2 expression had a relative increased sensitivity to tamoxifen 31271806_G0S2 is regulated by the PERK-eIF2alpha-ATF4 branch of the UPR and mediates ER stress-induced hepatic steatosis. |
ENSG00000123689 |
G0S2 |
36.264302 |
2.8473307106 |
1.509610 |
0.44168050 |
11.421862 |
0.00072584908054134462604412014741228631464764475822448730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0034647863349438296973192841221589333144947886466979980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
54.3247518598736 |
16.892526089092 |
19.0791858696593 |
4.63879477323716 |
| ENSMUSG00000009654 |
18302 |
Oit3 |
protein_coding |
Q8R4V5 |
FUNCTION: May be involved in hepatocellular function and development. {ECO:0000250}. |
Alternative splicing;Calcium;Disulfide bond;EGF-like domain;Glycoprotein;Nucleus;Reference proteome;Signal |
|
|
mmu:18302; |
cell surface [GO:0009986]; extracellular space [GO:0005615]; nuclear envelope [GO:0005635]; calcium ion binding [GO:0005509]; extracellular matrix structural constituent [GO:0005201]; urate homeostasis [GO:1903118] |
15346761_mouse LZP localizes mostly on nuclear envelope, and at the same time, it can be secreted into blood in a truncated form. 18830570_The finding that LZP might act as a new partner of THP would provide novel insights into renal functions related to THP and LZP, such as the urothelial permeability barrier and the host defense against the adhesion of pathogens. 22306318_Data suggest that OIT3 could maintain urate homeostasis by regulating the excretion and reabsorption of uric acid in renal tubule via cooperating with THP. 33591966_LZP is required for hepatic triacylglycerol transportation through maintaining apolipoprotein B stability. |
ENSG00000138315 |
OIT3 |
16.803786 |
4.2743700816 |
2.095712 |
0.66613029 |
9.861592 |
0.00168765011824078517595204562695698768948204815387725830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0073467523866943811733376534789385914336889982223510742187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
28.3311686427499 |
13.5510275425968 |
6.6281506050434 |
2.51374632965471 |
| ENSMUSG00000010086 |
22671 |
Rnf112 |
protein_coding |
Q96DY5 |
FUNCTION: E3 ubiquitin-protein ligase that plays an important role in neuronal differentiation, including neurogenesis and gliogenesis, during brain development. During embryonic development initiates neuronal differentiation by inducing cell cycle arrest at the G0/G1 phase through up-regulation of cell-cycle regulatory proteins (PubMed:21566658, PubMed:28684796). Plays a role not only in the fetal period during the development of the nervous system, but also in the adult brain, where it is involved in the maintenance of neural functions and protection of the nervous tissue cells from oxidative stress-induced damage (PubMed:27918959, PubMed:26792191, PubMed:26951452). Exhibits GTPase and E3 ubiquitin-protein ligase activities. Regulates dendritic spine density and synaptic neurotransmission; its ability to hydrolyze GTP is involved in the maintenance of dendritic spine density (PubMed:26212327). {ECO:0000269|PubMed:21566658, ECO:0000269|PubMed:26212327, ECO:0000269|PubMed:26792191, ECO:0000269|PubMed:26951452, ECO:0000269|PubMed:27918959, ECO:0000269|PubMed:28684796}. |
Alternative splicing;Cell projection;Cytoplasm;Cytoplasmic vesicle;Endosome;GTP-binding;Membrane;Metal-binding;Neurogenesis;Nucleotide-binding;Nucleus;Reference proteome;Synapse;Transferase;Transmembrane;Transmembrane helix;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000305}. |
|
mmu:22671; |
cell body [GO:0044297]; cytoplasm [GO:0005737]; endosome [GO:0005768]; extrinsic component of membrane [GO:0019898]; neuron projection [GO:0043005]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perikaryon [GO:0043204]; postsynaptic density [GO:0014069]; synaptic vesicle [GO:0008021]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; metal ion binding [GO:0046872]; protein self-association [GO:0043621]; ubiquitin protein ligase activity [GO:0061630]; cell death in response to hydrogen peroxide [GO:0036474]; cell death in response to oxidative stress [GO:0036473]; embryonic brain development [GO:1990403]; endoplasmic reticulum organization [GO:0007029]; G1 to G0 transition involved in cell differentiation [GO:0070315]; neuron differentiation [GO:0030182]; positive regulation of glial cell differentiation [GO:0045687]; positive regulation of neuron differentiation [GO:0045666]; protein autoubiquitination [GO:0051865]; protein homooligomerization [GO:0051260]; regulation of cell cycle [GO:0051726]; response to hydroperoxide [GO:0033194] |
21566658_Induction of the Znf179 gene is associated with p35 expression and p27 protein accumulation, which lead to cell cycle arrest in the G0/G1 phase, and is critical for neuronal differentiation of P19 cells. 24788683_This study provides the first evidence that ZNF179, PLZF, IGFBP3, and BIK contributed to the novel CEBPD-induced antiapoptotic feature of astrocytes. 26212327_identify neurolastin as a dynamin family GTPase that affects endosome size and spine density. 26792191_Study focused on sigma-1 receptor (Sig-1R), a chaperone at endoplasmic reticulum, and investigated its protective functions; results reveal that zinc finger protein 179 plays a novel role in neuroprotection. 26951452_Rnf112 protein is expressed in most brain areas and mainly in neurons; deletion of both Rnf112 alleles resulted in embryonic lethality associated with defects in embryonic vasculature; results provide evidence for the important functions of Rnf112 in embryo development and in adult brain 27918959_the results obtained in this study show that Znf179 autoregulation through Sp1-dependent mechanism plays an important role in neuroprotection, and NGF-induced Sp1 signaling may help attenuate more extensive (ROS-induced) damage following brain injury 30404641_Here the authors unveil the important role for the novel E3 ligase Znf179 in TDP-43-mediated neuropathy, and provide a potential therapeutic strategy for combating ALS/ FTLD-U neurodegenerative pathologies. 30454900_Rnf112 expression was highly involved in the progression of intracerebral hemorrhage. 33333713_Puralpha regulates the induction of Znf179 transcription during neuronal differentiation. |
ENSG00000128482 |
RNF112 |
290.185375 |
0.3389554948 |
-1.560832 |
0.17397083 |
78.855748 |
0.00000000000000000066814756133836141004805581418278440086888563345136893278919387739733792841434478759765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000242314848912045738044094908610289809381782523065029799624880979536101222038269042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
147.616186304445 |
17.8438231376708 |
435.503151761507 |
37.5646311994612 |
| ENSMUSG00000010307 |
67893 |
Tmem86a |
protein_coding |
Q9D8N3 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:67893; |
membrane [GO:0016020]; alkenylglycerophosphocholine hydrolase activity [GO:0047408]; alkenylglycerophosphoethanolamine hydrolase activity [GO:0047409] |
35835749_Adipocyte lysoplasmalogenase TMEM86A regulates plasmalogen homeostasis and protein kinase A-dependent energy metabolism. |
ENSG00000151117 |
TMEM86A |
2323.836114 |
0.4533157545 |
-1.141412 |
0.07507822 |
227.077267 |
0.00000000000000000000000000000000000000000000000000258677906036229920014841730484478039563919099832770711882123034218226857935962924961948745069831917364543553106512579983449861935340674534700156073085963726043701171875000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000004528944672119372384533421739502427061044312395818755826551148718966666774202826936126274052362072834843487588208643973184622938399357394700928125530481338500976562500000000000000000000000000000000000000000000000 |
Yes |
No |
1434.02975189316 |
74.6609605051621 |
3163.42359060921 |
120.701318323162 |
| ENSMUSG00000010342 |
83560 |
Tex14 |
protein_coding |
Q7M6U3 |
FUNCTION: Required both for the formation of intercellular bridges during meiosis and for kinetochore-microtubule attachment during mitosis. Intercellular bridges are evolutionarily conserved structures that connect differentiating germ cells and are required for spermatogenesis and male fertility. Acts by promoting the conversion of midbodies into intercellular bridges via its interaction with CEP55: interaction with CEP55 inhibits the interaction between CEP55 and PDCD6IP/ALIX and TSG101, blocking cell abscission and leading to transform midbodies into intercellular bridges. Also plays a role during mitosis: recruited to kinetochores by PLK1 during early mitosis and regulates the maturation of the outer kinetochores and microtubule attachment. Has no protein kinase activity in vitro. {ECO:0000269|PubMed:16549803, ECO:0000269|PubMed:19020301, ECO:0000269|PubMed:20176808, ECO:0000269|PubMed:22405274}. |
Alternative splicing;ANK repeat;ATP-binding;Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Kinetochore;Mitosis;Nucleotide-binding;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:83560; |
cytoplasm [GO:0005737]; intercellular bridge [GO:0045171]; kinetochore [GO:0000776]; midbody [GO:0030496]; ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; protein kinase binding [GO:0019901]; attachment of spindle microtubules to kinetochore [GO:0008608]; cell division [GO:0051301]; cellular response to leukemia inhibitory factor [GO:1990830]; intercellular bridge organization [GO:0043063]; male meiotic nuclear division [GO:0007140]; mitotic sister chromatid separation [GO:0051306]; mitotic spindle assembly checkpoint signaling [GO:0007094]; negative regulation of cytokinesis [GO:0032466]; negative regulation of protein binding [GO:0032091]; protein phosphorylation [GO:0006468] |
11279525_Tex14 is specifically expressed in testis in a cDNA subtraction screen 12711554_Predicted to be localized to the nucleus, suggesting a key role in regulating gene expression during mammalian spermatogenesis. 16118233_Tex14 is expressed throughout spermatogenesis 16549803_TEX14 is required for intercellular bridges in vertebrate germ cells, and these studies provide evidence that the intercellular bridge is essential for spermatogenesis and fertility 19020301_Despite the absence of embryonic intercellular bridges in the Tex14-null mice, male mice initiate spermatogenesis, and female mice are fertile. But fewer oocytes were present in Tex14-null neonatal ovaries. 20176808_TEX14 prevents the completion of cytokinesis by altering the destiny of CEP55 from a nidus for abscission to an integral component of the intercellular bridge. 21364893_RBM44 interacts with itself and TEX14 using yeast two-hybrid, mammalian two-hybrid, and immunoprecipitation. 26392564_Data indicate that testis-expressed gene 14 (TEX14) is expressed in germ cells. 34156079_Altered germline cyst formation and oogenesis in Tex14 mutant mice. |
ENSG00000121101 |
TEX14 |
34.490821 |
0.2012822315 |
-2.312708 |
0.50755622 |
19.482983 |
0.00001014999060029043236280492379375317568701575510203838348388671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000751602609740514702356753429235425301158102229237556457519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.7997704450209 |
4.23745745894961 |
58.6230108633558 |
14.5609017675838 |
| ENSMUSG00000010476 |
13593 |
Ebf3 |
protein_coding |
O08791 |
FUNCTION: Transcriptional activator (PubMed:9151732). Recognizes variations of the palindromic sequence 5'-ATTCCCNNGGGAATT-3' (By similarity). {ECO:0000250|UniProtKB:Q07802, ECO:0000269|PubMed:9151732}. |
Activator;Alternative splicing;Developmental protein;DNA-binding;Metal-binding;Nucleus;Reference proteome;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:13593; |
nucleus [GO:0005634]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
22421355_Ebf2 and Ebf3, singly or together, control the migration of Cajal-Retzius cells arising in the cortical hem. These findings provide evidence that Ebfs directly regulate Cajal-Retzius cell development 24786561_Ebf3 is a new regulator of terminal muscle differentiation in the diaphragm, and Ebf factors cooperate with MyoD in the induction of muscle-specific genes 25192643_The regulation of Ebf3 expression by miR218 controls the terminal differentiation of dopaminergic neurons. 29113800_Our findings highlight the critical role of Ebf3 in multipolar-to-bipolar transition via positive feedback regulation of NeuroD1 in the developing neocortex. 29258872_data show that Prdm13 is not required for amacrine fate as a class, but is essential for the formation of Ebf3+ amacrine cell subtypes. Rather than driving subtype identity, Prdm13 may act by restricting competing fate programs to maintain identity and survival. 29563184_HSC cellular niches express Ebf3 that is required to create HSC niches, to inhibit their osteoblast differentiation, and to maintain spaces for HSCs. 29869132_The caudo-ventral pallium is a novel pallial domain expressing Gdf10 and generating Ebf3-positive neurons of the medial amygdala. |
ENSG00000108001 |
EBF3 |
58.893543 |
5.5246365205 |
2.465880 |
0.36815841 |
43.435707 |
0.00000000004381200512599465403773227985671670330070082144402476842515170574188232421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000007277475049936211401332408314497190299219298026400792878121137619018554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
103.777036373597 |
26.7710168876986 |
18.7844097957858 |
3.8259117741278 |
| ENSMUSG00000012126 |
67586 |
Ubxn11 |
protein_coding |
Q9D572 |
FUNCTION: May be involved in the reorganization of actin cytoskeleton mediated by RND1, RND2 and RND3. Promotes RHOA activation mediated by GNA12 and GNA13 (By similarity). {ECO:0000250}. |
Alternative splicing;Coiled coil;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:67586; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; ubiquitin binding [GO:0043130]; proteasome-mediated ubiquitin-dependent protein catabolic process [GO:0043161] |
Human_homologues 11940653_Socius is a novel Rnd GTPase-interacting protein involved in disassembly of actin stress fibers 16202387_The rat homolog of UBXD5 promotes RhoA activation through G12/13 heterotrimeric G proteins 18974390_Both CD4+ and CD8+ T-cell responses against COA-1 can occur in colorectal cancer patients with metastatic disease |
ENSG00000158062 |
UBXN11 |
91.383130 |
2.8339463301 |
1.502812 |
0.28162499 |
28.278806 |
0.00000010503979140728166026209553212539260513835870369803160429000854492187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000011003893416201611681390750210884910131881042616441845893859863281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
131.889559299344 |
25.9096636799926 |
46.5391873870017 |
7.15897801824751 |
| ENSMUSG00000012396 |
71950 |
Nanog |
protein_coding |
Q80Z64 |
FUNCTION: Transcription regulator involved in inner cell mass and embryonic stem (ES) cells proliferation and self-renewal (PubMed:25825768). Imposes pluripotency on ES cells and prevents their differentiation towards extraembryonic endoderm and trophectoderm lineages. Blocks bone morphogenetic protein-induced mesoderm differentiation of ES cells by physically interacting with SMAD1 and interfering with the recruitment of coactivators to the active SMAD transcriptional complexes. Acts as a transcriptional activator or repressor. Binds optimally to the DNA consensus sequence 5'-TAAT[GT][GT]-3' or 5'-[CG][GA][CG]C[GC]ATTAN[GC]-3'. Binds to the POU5F1/OCT4 promoter (By similarity). Able to autorepress its expression in differentiating (ES) cells: binds to its own promoter following interaction with ZNF281/ZFP281, leading to recruitment of the NuRD complex and subsequent repression of expression. When overexpressed, promotes cells to enter into S phase and proliferation. {ECO:0000250|UniProtKB:Q9H9S0, ECO:0000269|PubMed:12787504, ECO:0000269|PubMed:12787505, ECO:0000269|PubMed:14728807, ECO:0000269|PubMed:15502159, ECO:0000269|PubMed:16518401, ECO:0000269|PubMed:16791199, ECO:0000269|PubMed:16801560, ECO:0000269|PubMed:21915945, ECO:0000269|PubMed:22988117, ECO:0000269|PubMed:25825768}. |
3D-structure;Activator;Alternative splicing;Developmental protein;Direct protein sequencing;DNA-binding;Homeobox;Nucleus;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation |
Human_homologues NA; + ;NA |
The protein encoded by this gene is a DNA binding homeobox transcription factor involved in embryonic stem (ES) cell proliferation, renewal, and pluripotency. The encoded protein can block ES cell differentiation and can also autorepress its own expression in differentiating cells. Several transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Sep 2015]. |
mmu:71950; |
chromatin [GO:0000785]; intracellular membrane-bounded organelle [GO:0043231]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; identical protein binding [GO:0042802]; lysine-acetylated histone binding [GO:0070577]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II sequence-specific DNA-binding transcription factor recruiting activity [GO:0001010]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; transcription factor binding [GO:0008134]; BMP signaling pathway [GO:0030509]; cell dedifferentiation [GO:0043697]; cellular response to erythropoietin [GO:0036018]; cellular response to rapamycin [GO:0072752]; embryonic pattern specification [GO:0009880]; endodermal cell fate specification [GO:0001714]; gene expression [GO:0010467]; gonad development [GO:0008406]; mesodermal cell fate commitment [GO:0001710]; multidimensional cell growth [GO:0009825]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of cell fate commitment [GO:0010454]; negative regulation of endodermal cell fate specification [GO:0042664]; negative regulation of stem cell differentiation [GO:2000737]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of mitotic cell cycle [GO:0045931]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of stem cell division [GO:2000035]; regulation of transcription by RNA polymerase II [GO:0006357]; response to organic substance [GO:0010033]; response to retinoic acid [GO:0032526]; somatic stem cell population maintenance [GO:0035019]; stem cell differentiation [GO:0048863]; stem cell division [GO:0017145]; stem cell population maintenance [GO:0019827]; tissue regeneration [GO:0042246] |
12787505_Nanog is a homeodomain protein whose enhanced expression confers constitutive self-renewal on ES cells. Forced expression of Nanog liberates ES cells from the requirement for gp130 stimulation. 15108323_mouse and human Nanog and Nanog2 genes may have a common role in the maintenance of pluripotency in both species 15233988_Two processed NANOG pseudogenes are identified in the complete mouse genome. 15494369_aggregation-based cell-fate specification during early embryonic development happens when downregulation of Nanog plays a crucial role. 15582778_The labile undifferentiated state was fully rescued by constitutive expression of exogenous Nanog. Thus, the activity of Nanog is tightly correlated with an undifferentiated state of cells even in nuclear reprogrammed somatic cells. 15619621_The decreased inhibition of Nanog expression in p53(S315A) ESCs during differentiation is due to an impaired recruitment of the co-repressor mSin3a to the Nanog promoter. 15860457_nanog is transcriptionally regulated by OCT4 and SOX2 15916719_Results describe the isolation, sequencing, and characterization of the Nanog promoter. 15939376_NANOG is expressed in proliferating germ cells, in which nuclear reprogramming is progressing 16259959_This suggests that Nanog gene might function in a similar fashion in mature cells as in ES cells. In addition, it may provide an approach for in vitro cell expansion. 16313224_the kidney has its own cells expressing NANOG, and loss of NANOG expression occurs in an age-dependent manner 16456133_transient inhibition of OCT4 or Nanog by RNAi compounds is sufficient to induce differentiation toward extraembryonic lineages, which supports the model that these transcription factors function in a dose-dependent manner to influence cell fate 16518401_Oct4 and Nanog control a cascade of pathways that are intricately connected to govern pluripotency, self-renewal, genome surveillance and cell fate determination 16541131_identified the role of two putative Sp1 binding sites located in the Nanog gene 5'-flanking region in regulation of murine Nanog gene transcription 16714766_Nanog can regulate the expression of Rex-1 by binding to its promoter. 16773043_These data suggest that Nanog is required to sustain the proper undifferentiated status of F9 cells, and the C-terminal domain of Nanog transduces the most effects in repressing primitive endoderm and parietal endoderm differentiation in F9 cells. 16791199_in fusions between embryonic stem (ES) cells and neural stem (NS) cells, Nanog stimulates pluripotent gene activation and enables an up to 200-fold increase in the recovery of hybrid colonies, all of which show ES cell characteristics 16801560_Nanog blocks bone morphogenetic protein-induced mesoderm differentiation of ES cells by physically interacting with Smad1 and interfering with the recruitment of coactivators to the active Smad transcriptional complexes 16845474_retrotransposed copies of Nanog, NanogPc & NanogPd, identified on mouse Chromosomes 4 & 7. possibility that expression of Nanog could be attributed in error to novel cellular sources is suggested by high homology among Nanog, NanogPc, & NanogPd. 16894029_Tcf3-mediated control of Nanog levels allows stem cells to balance the creation of lineage-committed and undifferentiated cells 16908534_growth factor receptor bound protein 2/Mitogen-Activated Protein Kinase Kinases pathway primarily mediates Nanog gene repression upon embryonic stem cell differentiation into primitive endoderm 17093407_exploration of the protein network in which Nanog operates in mouse ES cells 17204467_PI3K-dependent regulation of ES cell self-renewal is mediated, at least in part, by the ability of PI3K signaling to maintain Nanog expression. Regulation of GSK-3 activity by PI3Ks appears to play a key role in this process. 17211451_Elevated levels of Nanog can maintain mouse embryonic stem cell self-renewal and pluripotency independently of extrinsic signalling molecules. 17352742_there is a tissue-dependent and differentially methylated region (T-DMR) in the Nanog up-stream region 17360937_recipients of Nanog-transduced hematopoietic stem cells invariantly develop a unique disorder typified by an atrophic thymus occupied by Nanog-expressing gammadeltaT-cell receptor-positive cells. 17518637_Nanog marker was assessed using flow cytometry analysis and gene expression profiling. 17615266_Nanog can directly repress Gata6 expression through its binding to the proximal promoter region of the Gata6 gene. Overexpression of Nanog reduces heterogeneity during embryonic stem cell maintenance. 17938196_These results suggest that Dnmt3a and Dnmt3b form a complex through direct contact in living cells and cooperate in the methylation of the promoters of Oct4 and Nanog during cell differentiation. 18086680_the CD2 domain of Nanog is a unique transactivator that utilizes aromatic residues to confer specific activity absolutely required for ES self-renewal. 18097409_Transient downregulation of Nanog appears to predispose embryonic stem cells towards differentiation but does not mark commitment 18177668_the structural and functional evidence establish Nanog as a distant member of a Q50-type homeodomain protein despite having considerable variation at the sequence level. 18199879_Mouse primordial germ cells (PGCs), from which pluripotent embryonic germ cells (EGCs) are derived, express POU5F1, NANOG, and SOX2. 18223644_Nanog represses the pro-differentiation activities of NFkappaB and cooperates with Stat3 to maintain pluripotency. 18264089_Klf2, Klf4 and Klf5 integrate into the Nanog transcriptional network to specify gene expression that is unique to ES cells 18290762_deletion of the tryptophan-repeat region eliminates the capacity of Nanog to direct LIF (leukaemia inhibitory factor)-independent self-renewal 18371449_SIRT1 makes mES cells sensitive to ROS and inhibits p53-mediated suppression of Nanog expression. 18425773_These results suggest that Netrin-1 may facilitate functional differentiation of mammary epithelial cells and possibly affect the expression of Nanog and/or Cripto-1 in multipotent cells that may reside in the mammary gland. 18436640_Nanog-Nanog homodimerization is a critical aspect of its function promoting stem cell pluripotency. 18442017_Genome-wide molecular changes upon silencing Nanog in embryonic stem cells were identified by using microarray technology. 18453147_regulates early stage of body patterning in mammals. 18454139_Nanog and Oct4 associate with unique repressor complexes on their target genes to control ES cell fate 18522731_most reliable sets of direct target genes for key pluripotency genes - Pou5f1, Sox2, and Nanog have been identified, and they predominantly function as activators of downstream gene expression. 18584034_In ES cells the normally active Nanog promoter is silenced when targeted by de novo methylation. 18653541_Maintenance of the undifferentiated state by Klf5 is, at least in part, due to the control of Nanog and Oct3/4 transcription, because Klf5 directly binds to the promoters of these genes and regulates their transcription. 18662995_Esrrb is recruited near the Oct-Sox element in the Nanog proximal promoter, where it positively regulates Nanog expression. 18687992_Zfp143 regulates Nanog expression through modulation of Oct4 binding. 18802003_study concludes that the three main genetic factors underlying pluripotency, Nanog, Oct3/4, and Sox2 cooperate to repress Xist and thus couple X inactivation reprogramming to the control of pluripotency during embryogenesis 18806776_demonstration of the existence of many naturally occurring miRNA targets in gene's amino acid coding sequence 18957414_Esrrb coordinates with Nanog and Oct4 to activate the internal machinery of ES cells 18983963_expression is a marker of self-renewal bias in embryonic and adult stem cell populations 19036726_Nanog and Med12 function in concert to regulate Nanog target genes and identify a novel role for Med12 in embryonic stem cell regulation. 19139101_Sox2 associates with mSin3A-HDAC complex members both in vitro and in vivo, similar to the interactions found between Oct4/Nanog and the mSin3A-HDAC complex. Knockdown of mSin3A-HDAC complex members or HDAC inhibitor treatment reduces Nanog expression 19150979_p300 plays an important role in the differentiation process of embryonic stem cells and provide the first evidence for the involvement of p300 in regulating Nanog expression during differentiation 19171902_study demonstrates a TLR4-dependent mechanism of synergistic liver disease by HCV and alcohol and an obligatory role for Nanog, a TLR4 downstream gene, in HCV-induced liver oncogenesis enhanced by alcohol 19366700_Nanog interacts with Nac1 through WNAAP to regulate the cell cycle of stem cells via the ERas/phosphatidylinositol 3-kinase/Akt pathway, but not pluripotency 19400741_Nogo regulates Nanog expression through stat3 signal pathway in embryonic stem cells. 19403798_Simultaneous overexpression of Oct4 and Nanog abrogates terminal myogenesis. 19544422_In CARM1 overexpressing ES cells, histone H3 arginine methylation is also at the Nanog promoter to which CARM1 now associates. 19564890_Nanog and Cdx2 bind to and repress each other's promoters in embryo and placenta development 19582141_transcriptional fluctuations in Nanog expression (i.e., low vs. high Nanog expression) are essential elements of pluripotent state; Nanog, Sox2, and Oct4 act as network to promote/maintain transcriptional noise to disrupt differentiation signals 19701907_NIH3T3 cells expressed with mouse Oct4 (mOct4), mouse Nanog (mNanog) separately as well as together, the specific knockdown of mFAK1 inhibited morphological transformation of the cells, and their invasion activity. 19703398_These findings suggest that Nanog choreographs synthesis of the naive epiblast ground state in the embryo and that this function is recapitulated in the culmination of somatic cell reprogramming. 19785992_these results suggested that Hand2 bound to the promoter of nanog, a gene required for embryonic stem cells self-renewal, and suppressed nanog expression after Hand2 induction. 19845468_Data show that knockdown of Trp53 and Pten independently results in significantly higher expression levels of Nanog, and suggest that TRP53- and PTEN-mediated repression is important for the insulation of male germ cells from pluripotency. 19906868_Conditional knockdown of Nanog induces apoptotic cell death in mouse migrating primordial germ cells. 19933152_Satb1 and Satb2 bind the Nanog locus in vivo and the balance of Satb1 and Satb2 contributes to the plasticity of Nanog expression and ES cell pluripotency 20026622_identified four embryonic stem cell-expressed, conserved lncRNA-encoding genes residing proximally to active genomic binding sites of Oct4 and Nanog 20071344_Klf4 functions upstream of Nanog in ES cell self-renewal and in preventing ES cell differentiation 20103622_Data show that a significant number of the genes contained binding sites for the stem cell transcription factors Oct4 and Nanog. 20435031_Nanog is required for primitive endoderm formation through a non-cell autonomous mechanism 20539761_findings of the current study strongly suggest a conserved role for NANOG in meiotic and post-meiotic stages of male germ cell development 20574542_mechanisms and feedback regulations to describe the observed variation of the Nanog levels in mouse embryonic stem cells 20581804_downstream effectors of Hedgehog activity, Gli1 and Gli2, bind to Nanog-specific cis-regulatory sequences both in mouse and human stem cells. 20629982_Embryonic stem cells (ESCs) have a distinctive epigenome, which includes their genome-wide DNA methylation modification status, as represented by the ESC-specific hypomethylation of tissue-dependent and differentially methylated regions (T-DMRs) of Nanog. 20661723_Nanog expression is differentially regulated by the PI3K/Akt pathway during retinoic acid-induced cell differentiation. 20872845_Data demonstrate an important pathway for regulation of Nanog expression in pluripotent ESCs through direct activation by Zic3. 20937355_These data indicate that LRH-1 and Nanog cooperate to regulate Dax1 expression in mouse embryonic stem cells. 20962578_Data demonstrate that T cells retain responsiveness to a LIF/Nanog axis and that this axis is linked to MARCH-7, a RINGv E3 ligase that regulates the LIF-receptor. 21076177_Data show that Oct4, Sox2 and Nanog individually form complexes with nucleophosmin (Npm1) to control embryonic stem (ES) cell fate determination. 21187909_Analyzed genome-wide histone acetylation patterns and gene expression profiles measured over the first five days of cell differentiation triggered by silencing Nanog, a key transcription factor in ESC regulation. 21194951_It was shown that 2i/LIF treatment in clonal lines of pre-iPS cells results in the activation of endogenous Nanog and rapid downregulation of retroviral Oct4 expression. 21304588_the mechanism by which Pten null embryonic stem cells emerge in vitro and cause tumors in vivo is through increased survival and self-renewal, due to failed repression of the transcription factor Nanog. 21490431_Ezh2 directly regulates the epigenetic status of the Nanog promoter affecting the balance of Nanog expression in pluripotent stem cells, and therefore the equilibrium between self-renewal and differentiation. 21497086_Geminin antagonizes the chromatin-remodeling protein Brg1 to maintain expression of Oct4, Sox2, and Nanog. The results define a pluripotency pathway by which suppressed anaphase-promoting complex/cyclosome activity protects geminin from degradation in G1. 21499299_NANOG promotes cancer stem cell characteristics and prostate cancer resistance to androgen deprivation 21609209_Data show that differing levels of Oct4, Nanog, and c-myc proteins among the individual cells of embryonic stem cells (mESCs) colonies and fluctuations in these levels do not disturb mESC pluripotency. 21915945_Data demonstrated that Zfp281 is required for Nanog binding to its own promoter, suggesting that Nanog-associated repressive complex(es) involving Zfp281 may fine-tune Nanog expression for pluripotency of ESCs. 21969378_Alternative splicing produces Nanog protein variants with different capacities for self-renewal and pluripotency in embryonic stem cells 22013995_Id1 regulates self-renewal of embryonic stem cells by regulating levels of Nanog and Brachyury. 22028025_Reprogramming capacity of Nanog is functionally conserved in vertebrates and resides in a unique homeodomain. 22142851_these data show that Nanog is expressed during surgically induced marrow bone formation and is functionally involved in post natal marrow stromal cell maintenance and differentiation. 22172669_in Nanog null embryos, Gata6 expression is maintained, showing that initiation of the primitive endoderm program is Nanog independent 22315219_CIBZ post-transcriptionally regulates the expression of Nanog. 22327294_tight regulation of Nanog dose at the chromosome level is necessary for the acquisition of ground-state pluripotency during development 22337995_Nanog expression enhanced the stem-like features of tumor cells and protected them from killing by tumor-reactive CTLs 22388986_Testicular Nanog expression marked a radioresistant spermatogonial subpopulation, supporting its stem cell nature. 22438569_Germ cell proliferation and expression of the pluripotency factor Nanog at a specific time point, E15.5, were directly related with increased risk of testicular teratomas. 22683858_The findings imply that mechanical force plays a role in regulating Nanog expression in ES cells through the actin cytoskeleton-PI3K-Akt signaling. 22745796_Data found that SUMOylation of Oct4 and Sox2 regulated Nanog in an opposing manner. 22787153_FGFR2 induces rapid but reversible Nanog repression within ES cells. 22795133_Oct4 and Nanog directly regulate Dnmt1 to maintain self-renewal and undifferentiated state in mesenchymal stem cells 22969005_The distribution of Oct4/Nanog ratios can be used as quantifiers to distinguish between three subpopulations in an mESC culture: pluripotent, lineage-primed, and differentiating cells. 22988117_Nanog is subjected to a negative autoregulatory mechanism in embryonic stem cells requiring the coordinated action of Zpf281. 23054952_the existence of three stable steady states for Nanog levels, which are the same in all the different conditions of the cell-culture medium. 23071603_the possibly novel function for mNanog in stimulating the endogenous gene network in Xenopus mesoderm formation. 23132827_Gata4 blocks somatic cell reprogramming by limiting the expression of Nanog. Gata4 binds to a conserved distal enhancer of Nanog and impedes Nanog induction during reprogramming. 23178592_Nanog autorepression modulates heterogeneous Nanog gene expression in mouse embryonic stem cells. 23200040_Naive embryonic stem cells decondense their chromatin in the course of downregulating the pluripotency marker Nanog before they initiate lineage commitment. 23213213_OAC1 increases transcription of the Oct4-Nanog-Sox2 triad and Tet1, a gene known to be involved in DNA demethylation 23295861_Oct4, Sall4, and Nanog form a robust and integrated network to govern mammalian pre-implantation development. 23329853_Nanog has a role in supressing cell migration. 23333380_These findings support that Nanog can replace the Oct4 for the somatic cell reprogramming and underlie the mechanisms of Nanog in reprogramming process. 23470161_Cells exposed to ethanol follow an altered differentiation pathway compared with a higher expression of Sox2, Oct4, and Nanog. 23649762_RNA-binding protein Rbm47 binds to Nanog in mouse embryonic stem cells. 23665121_This network was rearranged during differentiation and restored in induced pluripotent stem cells. A large fraction of Nanog-interacting loci were bound by Mediator or cohesin in pluripotent cells. 23770853_findings demonstrate the involvement of Nanog in breast cancer metastasis, and provide the basis for the reported correlation between Nanog expression and poor prognosis of human breast cancer patients 23892456_A direct physical interaction between Nanog and Sox2 regulates embryonic stem cell self-renewal. 23963652_Data suggest a role for Vc in Nanog regulation networks and reveal a novel role for STAT2 in regulating Nanog expression. 24059348_Transcriptional reprogramming and chromatin remodeling accompanies Oct4 and Nanog silencing in mouse trophoblast lineage. 24076654_Nanog enhances miR-17/92 cluster expression that in turn targets Trp53inp1, thus contributing to release neural stem cell self-renewal constraints. 24227785_Nanogwas selectively induced in hypoxic areas of B16-F10 tumors & correlated with the acquisition of stem cell-like properties in tumor cells. Nanog regulated TGF-beta1 under hypoxia by directly binding the TGF-beta1 proximal promoter. 24333425_These data show that in the absence of DNA damage, p53 is required for the maintenance of mouse ESC self-renewal by regulating Nanog expression. 24461999_we find that Nanog is dispensable for induced pluripotent stem cells formation under optimized culture conditions 24462001_we propose that mechanisms of reprogramming of a differentiated cell back to a naive pluripotent identity are linked, rather than independent, and are centered on a small number of genes, including NANOG. 24466030_SHP-1 participated in regulation of Nanog expression. 24472544_Tbx3+2a could directly interact with Nanog while Tbx3 couldn't, indicating the requirement of the 2a domain in Nanog binding 24527385_Nanog may be an important mediator of reprogramming, it is not required for establishing pluripotency in the mouse, even under standard conditions. 24681518_Nanog protein promotes pluripotency by inhibiting endodermal specification. 24695638_observed cell cycle-regulation of Nanog expression, and not Oct4, reaching high levels prior to G1/S transition, finely mirroring Cyclin E1 fluctuations 24803390_Foxa1 recruited Grg3 to the Nanog promoter -2kb upstream region and switched the promoter to an inactive chromatin status represented by typical modifications in histone H3. 24836291_Nuclear transfer alone affords no immediate transition from a somatic to an Oct4- and Nanog-positive gene expression pattern, unless DNA replication is also in place within the mouse ooplasm. 24929238_Nanog co-regulated by Nodal/Smad2 and Oct4 is required for pluripotency in developing mouse epiblast. 24979572_NANOG is selectively expressed in stratified epithelia, most notably in the oesophagus where the Nanog promoter is hypomethylated. 25005472_stochastic NANOG fluctuations are widespread in mouse embryonic stem cells. 25146928_BRD4 associates with BRG1, a key regulator of embryonic stem cell self-renewal and pluripotency, in the Nanog regulatory regions to regulate Nanog expression. 25173648_Despite downregulation of tumor suppressors and increased proliferation, ectopic Nanog expression did not lead to tumor formation. 25240402_Nanog functions in conjunction with mesenchymal factors Snai1 and Snai2 in the transcriptional regulation of pluripotency-associated genes and miRNAs during the Nanog-driven reprogramming process. 25284313_Dax1 and Nanog have roles in stabilizing mouse embryonic stem cells and induced pluripotency 25377420_Data position Cited2 within the pluripotency gene regulatory network by defining Nanog, Tbx3, and Klf4 as its direct targets and demonstrate that the defects caused by Cited2 depletion are, at least in part, rescued by Nanog constitutive expression. 25410303_Stochastic promoter activation significantly affects the Nanog expression variability in mouse embryonic stem cells. 25445595_Taken together, our results suggest that Nanog positively regulates Zfp57 expression in multiple types of cells. 25456834_prolonged expression upon differentiation in METTL3-depleted embryonic stem cells 25522312_Forced expression of Nanog (and Esrrb) resulted in the dispensability of nucleostemin expression in embryonic stem cells. 25743411_Myst2-mediated histone acetylation may be required for recruitment of Oct4 to the Nanog promoter 26296469_immunoprecipitation. Our data provide new insights into the expanded regulatory circuitry that coordinates Nanog expression. 26411691_Tgif1 counterbalances the activity of core pluripotency factors, Oct4, Sox2, and Nanog, in mouse embryonic stem cells. 26549484_The cellular reprogramming-promoting function of mitoflashes occurs via the upregulation of Nanog expression. 26582088_TLR4 signaling via NANOG cooperates with STAT3 to activate Twist1 and promote formation of tumor-initiating stem-like cells in livers of mice. 26642061_Pluripotency transcription factors Nanog and Oct4 positively modulate Sod2 gene transcription in induced pluripotent stem cells. 26660234_1After the 32-cell stage, when embryos form the blastocyst cavity, Nanog expression was upregulated mainly in ICM cells while it was repressed in the future primitive endoderm lineage in an FGF signaling-dependent manner in the later stages 26724859_NANOG represses mitochondrial oxidative phosphorylation genes, as well as reactive oxygen species generation, and activates fatty acid oxidation to support tumor initiating cell self-renewal and drug resistance. 26733157_Nanog is able to transform normal somatic cells into tumor cells. 26751055_NANOG alone induces germ cells in primed epiblast in vitro by activation of enhancers 26760119_The different embryonic stem cells Nanog-expressing populations differ in their differentiation propensities. Nanog upregulation is not well correlated to Oct4, Sox2 and Klf4 expression, or cell cycle phase. 26795560_Nanog enhances proliferation of fibroblasts through transcriptional regulation of cell cycle inhibitor p27 gene. 26797124_NANOG binds to GLI1 and GLI3 proteins and represses Hedgehog-mediated transcription. 26975336_Data indicate that Nanog homeobox protein is important for the effects of reprogramming media. 27125671_NANOG as a key regulator connecting the pluripotency network with constitutive heterochromatin organization in mouse embryonic stem cells. 27146218_Nanog, Oct4 and Tet1 interplay in establishing stem cell pluripotency has been described. 27226076_In mouse embryonic stem cells, Cops2 binds to Nanog protein and prevent the degradation of Nanog by the proteasome. 27292645_Otx2-mediated Nanog regulation contributes to the integrity of the embryonic stem cell state and cell lineage specification in preimplantation development. 27622636_the epigenetic status of Oct4 and Nanog genes confirm that pre-induced pluripotent stem cells belong to an RVTg+OCT4+NANOG- state 27773674_Maintenance of pluripotency is regulated by a network of transcription factors coordinated by Oct4, Sox2, and Nanog; Trim24 significantly improved efficiency of cellular reprogramming, demonstrating its direct functionality in establishing pluripotency. 27886188_USP21 interacts with, deubiquitinates and stabilizes Nanog, and therefore maintains the protein level of Nanog in mouse embryonic stem cells to maintain stemness. 27939294_Here the authors couple mutagenesis with functional and dimerization assays to show that the number of tryptophans within the tryptophan repeat is linked to the strength of homodimerization, Sox2 heterodimerization and self-renewal activity. 28093523_studies highlight a new hypoxia-induced pathway in which NANOG activates BNIP3L expression, contributing to autophagy induction in hypoxic tumor cells and their resistance to killing by CTL 28199843_Members of the SIN3A/HDAC2 corepressor complex are enriched in an extended NANOG interactome. 28253866_In mouse model of acute uterine injury, Nanog homebox (NANOG) expression reached a peak at 6 h, while sex-determining region Y-box2 (SOX2) and octamer-binding protein 4 (OCT4) peaked later at 12 h after lipopolysaccharide (LPS) treatment. 28436127_Tip110 deletion impaired embryonic and stem cell development involving downregulation of stem cell factors Nanog, Oct4, and Sox2. 28457890_Loss of phosphorylation enhances NANOG activity in reprogramming. Mutation S65A alone has the most significant impact on increasing NANOG reprogramming capacity. 28636920_Genetic reporters can compromise the behavior of important pluripotency-sustaining positive feedback loops, and induce a bifurcation in the underlying dynamics that gives rise to heterogeneous Nanog expression patterns in reporter cell lines that are not representative of the wild-type; findings help explain the range of published observations of Nanog variability and highlight the problem of measurement in live cells. 28961274_Functional studies revealed that knockdown of Nanog eliminated the mESC self-renewal-promoting ability of Sp5. Finally, we demonstrated that the self-renewal-promoting function of Sp5 was largely dependent on its zinc finger domains 29079783_Nanog-driven cell-reprogramming and self-renewal maintenance in Ptch1 (+/-) granule cell precursors after radiation injury 29245050_myoblasts that were primed by NANOG maintained their differentiation capacity for 20days after NANOG withdrawal 29476172_Foxm1, which proved to be directly regulated by Gli and Nanog, controls a pro-stemness microRNA network in neural stem cells. 29631477_Reprogramming was also achieved with Nanog alone in a p53-deficient GSC line. 29741478_SRSF3 binds to the core pluripotency transcription factor Nanog mRNA to facilitate its nucleo-cytoplasmic export independent of splicing. In the absence of SRSF3 binding, Nanog mRNA is sequestered in the nucleus and protein levels are severely downregulated. 29933066_Sod1 mRNA levels were reduced when Oct4, Sox2 and Nanog were down-regulated by a shRNA approach in mESCs. 30654938_Hmga2 transcriptionally up-regulates expression of stem cell factors, Sox2 and Nanog. They were co-localized with Hmga2 in the germs, but differentially distributed at enamel organ and mesenchyme in Hmga2-/- mice. 30790630_Fluorescence correlation spectroscopy analyses suggest a more compact chromatin organization in K6b-/- cells and impaired interactions of Oct4 and Nanog with chromatin. 30814124_Nanog expression in adults prevents the maturation of erythroid cells 30846691_Nanog deploys multiple distinct mechanisms to enhance embryonic stem cell self-renewal. In the presence of LIF, which fosters self-renewal, Nanog rewires the pluripotency network by promoting chromatin accessibility and binding of other pluripotency factors to thousands of enhancers. 31051141_Targeting dCas9 to an Oct4 site in the Nanog promoter displaced Oct4 from this site, reduced Nanog expression, and slowed division. In contrast, disrupting the Oct4 binding site adjacent to Pax6 upregulated Pax6 transcription and disrupting Nanog binding its own promoter upregulated its transcription 31204172_Nanog supports pluripotency in naive cells, while Nodal supports pluripotency in primed cells, but the handover from Nanog to Nodal does not proceed seamlessly, opening up the risk of aberrant differentiation if FGF is activated before Nodal. Here, we report that Id1 acts as a sensor to detect delays in Nodal activation after the downregulation of Nanog. 31228531_Low intensity pulsed ultrasound (LIPUS) maintains osteogenic potency by the increased expression and stability of Nanog through spleen tyrosine kinase (Syk) activation. 31262352_The results suggest that Nanog represses Gsr gene expression in embryonic stem cells, evidencing a role of this crucial pluripotency transcription factor in preservation of redox homeostasis in stem cells. 31279504_Compression force, which is generated by the 3D-SAC, promotes colony growth and expression of Nanog and Oct4 in mESCs and blastocyst development of mouse embryos. 31582397_Study shows that TET2 binds to the transcription factor NANOG suggesting that TET2 and NANOG co-localize on chromatin to regulate enhancers associated with naive pluripotency genes. 31891567_essential role in the activation of pulmonary fibroblasts 32145514_Pluripotency factor NANOG promotes germ cell maintenance in vitro without triggering dedifferentiation of spermatogonial stem cells. 32640900_Low-Level Nanog Expression in the Regulation of Quiescent Endothelium. 32762616_LC3B upregulation by NANOG promotes immune resistance and stem-like property through hyperactivation of EGFR signaling in immune-refractory tumor cells. 32991228_Long noncoding RNA lnc-NAP sponges mmu-miR-139-5p to modulate Nanog functions in mouse ESCs and embryos. 33058869_TET1 Interacts Directly with NANOG via Independent Domains Containing Hydrophobic and Aromatic Residues. 33107035_Phosphorylation of NANOG by casein kinase I regulates embryonic stem cell self-renewal. 33334884_Genome editing demonstrates that the -5 kb Nanog enhancer regulates Nanog expression by modulating RNAPII initiation and/or recruitment. 33503260_BACH1 recruits NANOG and histone H3 lysine 4 methyltransferase MLL/SET1 complexes to regulate enhancer-promoter activity and maintains pluripotency. 33665763_Effect of Nanog overexpression on the metastatic potential of a mouse melanoma cell line B16-BL6. 35976266_H3K9 tri-methylation at Nanog times differentiation commitment and enables the acquisition of primitive endoderm fate. |
ENSG00000255192+ENSG00000111704 |
NANOGP8+NANOG |
51.671916 |
5.6069305840 |
2.487211 |
0.46067248 |
28.323247 |
0.00000010265575326055222507794413306508873873212905891705304384231567382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000010776377902582573119330327723885964985583996167406439781188964843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
86.9032880994339 |
28.2233930672348 |
15.4992623499249 |
4.02010565572093 |
| ENSMUSG00000012428 |
117167 |
Steap4 |
protein_coding |
Q923B6 |
FUNCTION: Integral membrane protein that functions as NADPH-dependent ferric-chelate reductase, using NADPH from one side of the membrane to reduce a Fe(3+) chelate that is bound on the other side of the membrane (PubMed:16609065). Mediates sequential transmembrane electron transfer from NADPH to FAD and onto heme, and finally to the Fe(3+) chelate (By similarity). Can also reduce Cu(2+) to Cu(1+) (PubMed:16609065). Plays a role in systemic metabolic homeostasis, integrating inflammatory and metabolic responses (PubMed:17482547). Associated with obesity and insulin-resistance (By similarity). Involved in inflammatory arthritis, through the regulation of inflammatory cytokines (PubMed:19660107). Inhibits anchorage-independent cell proliferation (By similarity). {ECO:0000250|UniProtKB:Q687X5, ECO:0000269|PubMed:16609065, ECO:0000269|PubMed:17482547, ECO:0000269|PubMed:19660107}. |
Cell membrane;Copper;Endosome;FAD;Flavoprotein;Golgi apparatus;Heme;Ion transport;Iron;Iron transport;Membrane;Metal-binding;NADP;Oxidoreductase;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:117167; |
early endosome membrane [GO:0031901]; endosome [GO:0005768]; Golgi membrane [GO:0000139]; membrane [GO:0016020]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; cupric reductase activity [GO:0008823]; electron transfer activity [GO:0009055]; FAD binding [GO:0071949]; ferric-chelate reductase (NADPH) activity [GO:0052851]; heme binding [GO:0020037]; metal ion binding [GO:0046872]; copper ion import [GO:0015677]; fat cell differentiation [GO:0045444]; iron import into cell [GO:0033212]; iron ion import across cell outer membrane [GO:0098706]; protein homotrimerization [GO:0070207] |
14988015_Tiarp is stimulated by tumor necrosis factor-alpha in adipocytes. 17482547_Study concludes that STAMP2 participates in integrating inflammatory and metabolic responses and thus plays a key role in systemic metabolic homeostasis. 19289123_TIARP/STAMP2 is highly upregulated in 3T3-L1 cells and hMSC-Ad by IL-1beta and might, therefore, modulate proinflammatory and insulin resistance-inducing effects of IL-1beta. 20304921_analysis of the regulation of hepatic six transmembrane epithelial antigen of prostate 4 (STEAP4) expression by STAT3 and CCAAT/enhancer-binding protein alpha 22704678_Stamp2 is detected in mouse and human atherosclerotic plaques, and its deficiency promotes atherosclerosis in mice. 22886597_TIARP acts as a negative regulator of arthritis by suppressing IL-6 production, its signaling and TNFalpha-induced NF-kappaB signaling, resulting in enhanced apoptosis in macrophages. 23874564_Stamp1 and Stamp2 play critical roles in adipogenesis, but through different mechanisms. [Stamp2, mouse] 23990467_that Steap4, a member of the six-transmembrane epithelial antigen of prostate (Steap) family proteins, is an endosomal ferrireductase with a critical role in cellular iron utilization in osteoclasts. 24236066_STAMP2 gene overexpression may improve insulin resistance via regulating macrophage polarization in visceral and brown adipose tissues. 24467451_STAMP2 overexpression could exert a protective effect on diabetic atherosclerosis by reducing IR and diminishing macrophage apoptosis 25646886_hepatic STAMP2 plays a pivotal role in preventing HFD-induced NAFLD and that STAMP2 overexpression improves hepatic steatosis and insulin resistance in NAFLD. 25817898_Data show that high glucose increased protein-protein interaction between Steap4 and S100B in mesangial (MES13) cells. 26018251_related to islet dysfunction during the development of type 2 diabetes 28631352_Overexpressing STAMP2 attenuates adipose tissue angiogenesis and insulin resistance in diabetic ApoE(-/-) /LDLR(-/-) mouse via a PPARgamma/CD36 pathway. 29078383_STEAP4 and mitochondrial iron increased tumor number and burden in a CAC model. 30710574_data suggest a clear link between ER stress and quantitative and functional STAMP2-deficiency 32060280_IL-17-induced STEAP4-dependent cellular copper uptake is critical for colon tumor formation in a murine model of colitis-associated tumorigenesis and STEAP4 expression correlates with IL-17 level and XIAP activation in human colon cancer. 32721044_Hepatic STAMP2 mediates recombinant FGF21-induced improvement of hepatic iron overload in nonalcoholic fatty liver disease. 33188479_The six-transmembrane protein Stamp2 ameliorates pulmonary vascular remodeling and pulmonary hypertension in mice. 33879167_STEAP4 expression in CNS resident cells promotes Th17 cell-induced autoimmune encephalomyelitis. 35227772_sSTEAP4 regulates cellular homeostasis and improves high-fat-diet-caused oxidative stress in hepatocytes. |
ENSG00000127954 |
STEAP4 |
158.026071 |
0.3985579466 |
-1.327139 |
0.23096169 |
32.772006 |
0.00000001036265150552456358564356648675405803139426552661461755633354187011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001247777926736364824609180380898809659129256033338606357574462890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
90.0494144629014 |
14.4777080020289 |
225.938073089251 |
26.0450465771027 |
| ENSMUSG00000012443 |
16551 |
Kif11 |
protein_coding |
Q6P9P6 |
FUNCTION: Motor protein required for establishing a bipolar spindle during mitosis. Required in non-mitotic cells for transport of secretory proteins from the Golgi complex to the cell surface. {ECO:0000250|UniProtKB:P52732}. |
Acetylation;ATP-binding;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Isopeptide bond;Microtubule;Mitosis;Motor protein;Nucleotide-binding;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:16551; |
cytosol [GO:0005829]; microtubule [GO:0005874]; mitotic spindle [GO:0072686]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; spindle [GO:0005819]; spindle microtubule [GO:0005876]; spindle pole [GO:0000922]; ATP binding [GO:0005524]; microtubule binding [GO:0008017]; plus-end-directed microtubule motor activity [GO:0008574]; protein kinase binding [GO:0019901]; cell division [GO:0051301]; microtubule-based movement [GO:0007018]; mitotic centrosome separation [GO:0007100]; mitotic sister chromatid segregation [GO:0000070]; mitotic spindle assembly [GO:0090307]; regulation of mitotic centrosome separation [GO:0046602]; spindle assembly [GO:0051225]; spindle elongation [GO:0051231]; spindle organization [GO:0007051] |
17449012_Observations further support the conclusion that Eg5 is essential for cell division early in mouse development. 17974955_Results indicate that Eg5 overexpression disrupts the unique balance of forces associated with normal spindle assembly and function, and thereby leads to the development of spindle defects, genetic instability, and tumors. 18474226_Eg5 is essential during early mouse development and cannot be compensated by another molecular motor. 22337772_observations explain the poleward accumulation of Eg5 in early mitosis and its redistribution in anaphase. Inhibition of dynein blocked Eg5 movement on microtubules, whereas depletion of the Eg5-binding protein TPX2 resulted in end-directed Eg5 movement. 22552964_These results show that intratumoral electro-transfer of siRNA is feasible and kinesin spindle protein (KSP)-specific siRNA may provide a novel strategy for therapeutic intervention. 24334276_incorporation of photochromic molecules into the key region of loop L5 facilitates the photocontrol of the function of kinesin Eg5. 24451491_These results suggest that 4-(N-(2-(N-acetylcysteine-S-yl) acetyl) amino)-4'- (N-(2-(N-(triphenylmethyl)amino)acetyl)amino)azobenzene could be used as photochromic inhibitor of Eg5 to achieve photocontrol of living cells. 24607899_Finally, when kinesin spindle protein (KSP) siRNA was encapsulated in lipid nanoparticles containing a modest amount of PEG, the proliferation of endothelial cells was inhibited due to the efficient knock down of KSP mRNA. 24636920_Its inhibition reduces neutrotrophin and transmitter receptor function. 25588836_Disruption of the in vivo interaction of KIF11 with ZBP1 delocalizes beta-actin mRNA and affects cell migration. 26957206_Data suggest that amyloid-beta (here, Abeta 1-42) induces acute and chronic synaptic dysfunction in part through inhibition of Eg5 (kinesin-5) in hippocampal neurons. 27117404_Fbxo30-Eg5 interaction as a critical checkpoint in mammopoiesis. 27240320_that Pten, through the Dlg1-binding ability of its PDZ-binding domain , accumulates phosphorylated Eg5 at duplicated centrosomes to establish symmetrical bipolar spindles that properly segregate chromosomes 27336410_Data show that the STIM1 Ksp (Eg5)-cre knockout mice produced more urine compared to control. 29899413_NEK7 regulates dendrite morphogenesis in vitro and in vivo. NEK7 kinase activity is required for dendrite growth and branching, as well as spine formation and morphology. NEK7 regulates these processes in part through phosphorylation of the kinesin Eg5/KIF11, promoting its accumulation on microtubules in distal dendrites. 30479371_Kif11 regulates excitatory synaptic transmission. Kif11 knockdown enhanced dendritic arborization, synapse number, expression of synaptophysin and Piccolo in vitro. 31424977_KIF11 (kinesin-5/Eg5), which functions in spindle formation during mitosis and meiosis in oocytes and early embryos, is expressed in premeiotic germ cells (spermatogonia and spermatocytes). 31904480_TRIM8 interacts with KIF11 and KIFC1 and controls bipolar spindle formation and chromosomal stability. 31993640_A mouse model for kinesin family member 11 (Kif11)-associated familial exudative vitreoretinopathy. 36017147_KIF11 Is a Promising Therapeutic Target for Thyroid Cancer Treatment. |
ENSG00000138160 |
KIF11 |
3207.882891 |
2.2006146209 |
1.137907 |
0.07485186 |
227.167364 |
0.00000000000000000000000000000000000000000000000000247234746179101490304926223712490077594825175900622125042989932975529856103516257390811520520585567748040678662698148284899884508970802077243433814146555960178375244140625000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000004378929830000085670033997022857440025064952927507078336093056960061127399960409126758426555060483738089010264633554921488538258511624690072494558990001678466796875000000000000000000000000000000000000000000000000 |
Yes |
No |
4405.15194018137 |
228.656607321186 |
2001.78254668131 |
78.9151197793644 |
| ENSMUSG00000013089 |
104156 |
Etv5 |
protein_coding |
Q9CXC9 |
FUNCTION: Binds to DNA sequences containing the consensus nucleotide core sequence 5'-GGAA.-3'. {ECO:0000250|UniProtKB:P41161}. |
DNA-binding;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:104156; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; synapse [GO:0045202]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; cell differentiation [GO:0030154]; cellular response to oxidative stress [GO:0034599]; locomotory behavior [GO:0007626]; male germ-line stem cell asymmetric division [GO:0048133]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuromuscular synaptic transmission [GO:0007274]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of glial cell proliferation [GO:0060252]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of branching involved in mammary gland duct morphogenesis [GO:0060762]; regulation of synapse organization [GO:0050807]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal muscle acetylcholine-gated channel clustering [GO:0071340] |
12871699_ERM is a transcription factor in the ets family, within which it forms the PEA3 group. 12941618_expression of Erm is initially restricted to the distal buds of the developing mouse lung 15138262_ETS-1, PEA3, and ERM have roles in regulation of caveolin-1 transcription in epithelial cells 16107850_Mice with targeted disruption of ERM have a loss of maintenance of spermatogonial stem cell self-renewal without a block in normal spermatogenic differentiation and thus have progressive germ-cell depletion and a Sertoli-cell-only syndrome 16613858_Erm is involved in SP-C regulation, which results from an interaction with TTF-1 17574550_Results identify common and unique pathways that regulate Etv5 and Gdnf mRNA in Sertoli cells. 17597063_GDNF up-regulation of Bcl6b, Erm, and Lhx1 expression through Src family kinase signaling is a key component of the intracellular mechanism for spermatogonial stem cells self-renewal. 17638886_ERM/ETV5 up-regulation activates MMP-2 and thus has a role during myometrial infiltration in endometrial cancer 17785180_Together, our findings define Erm as an upstream regulator of a transcriptional program selective to subsynaptic nuclei at the NMJ and underscore the importance of transcriptional control of local synaptic protein accumulation. 17911411_The blood-testis barrier (Sertoli-Sertoli tight junctional complex) is abnormal in the Etv5(-/-) mouse. 18032421_Etv5(-/-) mice are infertile even when sperm is produced. 18492810_The results show that Etv4 and Etv5 are expressed in granulosa and cumulus cells during folliculogenesis and ovulation, suggesting that they influence cellular events in the ovary by regulating downstream genes such as Ptgs2. 18827017_Reduced tumorigenesis in mouse mammary cancer cells following inhibition of Pea3- or Erm-dependent transcription. 19301256_FGF2 stimulates SDF-1 expression through the Erm transcription factor in Sertoli cells. 19369650_results indicate that ETV5 expression begins in fetal Sertoli cells and neonatal gonocytes of the germ cell lineage; loss of ETV5 results in decreased RET expression with possible inhibition of downstream GDNF/RET/GFRA1 signaling 19386268_FGF activity in the limb is not only responsible for maintaining posterior-specific Shh expression, but it also acts via Etvs to prevent inappropriate anterior expansion of Shh. 19386269_ETV4 and ETV5 act downstream of FGF signaling to inhibit Shh expression in the anterior limb bud. 19545444_endogenous Ets factors, including Erm, have tumor-restricting roles in the intestinal epithelium 19898483_Etv5 is positively regulated by Ret signaling in the ureteric bud tips 20610757_This study suggested that the etv5 do not regualte the dopamine pathway in the mouse midbrain. 20799334_Data show for the first time that Sertoli cells are chemoattractive for stem/progenitor spermatogonia, and that production of specific chemokines is regulated by ETV5. 21478265_Results suggest that Etv5 is important for the developmental competence of germ cells and the regulation of responses to steroid hormones in female mice. 21768389_Data show that miR-21 is regulated by the transcription factor ETV5, known to be critical for SSC self-renewal. 21816850_found to be a critical downstream regulator of glial cell line-derived neurotrophic factor-signaling that mediated the expression of several known spermatogonial stem cell self-renewal related genes 22491947_Mouse germline stem (GS) cell transfected with an activated form of Map2k1 have upregulated Etv5 and Bcl6b gene expression. 24204802_A missense mutation in the transcription factor ETV5 leads to sterility, increased embryonic and perinatal death, postnatal growth restriction, renal asymmetry and polydactyly in the mouse. 24486067_The transcription factors Etv5 and STAt3 control TH17 cell development and allergic airway inflammation. 25683353_Noise-decreased levels of ROCK2 results in reduction of phosphorylation of ERM that leads to depolymerization of actin filaments 25813907_The data of this study suggested that the obesity-associated transcription factor ETV5 normally acts to diminish circulating glucocorticoids. 26184908_Further investigations identified Pak1-ERM as a downstream signaling cascade upon Rac1 activation in the luminal epithelium necessary for uterine receptivity...Rac1 via P38 MAPK signaling ensures timely epithelial apoptotic death at postimplantation. 26224636_ETS-related transcription factors ETV4 and ETV5 are involved in proliferation and induction of differentiation-associated genes in embryonic stem (ES) cells. 27335464_substrate that mediates RFWD2 function in lung branching 27496971_findings demonstrate that ETV5 promotes IL-9 production in Th9 cells by binding and recruiting histone acetyltransferases to the Il9 locus at sites distinct from PU.1. data define a role for ETV5 in Th9 development and extend the paradigm of related transcription factors having complementary functions during differentiation 27498191_SOX9 regulation of ETV5 contributes to the control of male fertility 27822884_bioinformatic analysis, real-time PCR, and luciferase assay validated that AK015322 serves as a decoy of microRNA-19b-3p (miR-19b-3p), antagonizes its function, and attenuates the repression of its endogenous target transcriptional factor Ets-variant 5 (ETV5) which was a pivotal gene for spermatogonial stem cell self-renewal 27909004_This study demonstrates that Etv4 and Etv5 transcription factors are expressed in hippocampal pyramidal neurons during the main period of dendritogenesis; reveals a novel role for Etv4 and Etv5 transcription factors for hippocampal circuit development and the coordination of synaptic maturation; and indicates that both factors are essential in the transcriptional program linking BDNF signaling to dendrite development. 28100679_this study shows that Etv5 plays a crucial role in regulating IL-10 production in Th2 cells by facilitating the binding of IL-10-inducing transcription factors at the Il10 locus 28351980_Etv5 is expressed alveolar type II cells in adult lung.Etv5 is a critical output of Ras signaling in alveolar type II cells, contributing to both lung homeostasis and tumor initiation. 28855737_CIC-ETV5 axis is critical for regulation of follicular helper T cell development 29445086_Etv5 can positively regulate primitive endoderm specification through regulating Gata6 and negatively regulate epiblast specification by inhibiting Fgf5 expression. 29532564_ETV5 regulates ductal morphogenesis with Sox9 and is critical for regeneration from pancreatitis 29787563_High Etv5 expression is associated with glioma. 30735697_Study found that ETV5 increased acyl ghrelin but not its total production; through the enhanced expression of GOAT enzyme. ETV5 transactivated GOAT promoter, leading to the subsequent increase of its expression. mTORC1 signaling inhibited the expression of ETV5 which deficiency attenuated the effect of mTORC1 signaling on GOAT/ghrelin. Therefore, ETV5 is a new transcription factor regulating GOAT expression. 30810242_Derepression of Etv5 increases Hobit expression in Cic -deficient CD8+ T cells and consequently enhances CD8+ T memory cell formation in liver, thereby leading to liver injury. 32043969_Etv transcription factors functionally diverge from their upstream FGF signaling in lens development. 33093014_ETV5 Regulates Hepatic Fatty Acid Metabolism Through PPAR Signaling Pathway. 33159233_Etv5 safeguards trophoblast stem cells differentiation from mouse EPSCs by regulating fibroblast growth factor receptor 2. 33298570_Establishment of Etv5 gene knockout mice as a recipient model for spermatogonial stem cell transplantation. 34716308_Chromatin accessibility analysis identifies the transcription factor ETV5 as a suppressor of adipose tissue macrophage activation in obesity. |
ENSG00000244405 |
ETV5 |
120.928989 |
0.4196303359 |
-1.252809 |
0.25261647 |
24.460948 |
0.00000075831380901115904233920125138146417498319351579993963241577148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000069248416899628148277038609814670877540265792049467563629150390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
72.4339889640008 |
12.7481309778205 |
172.613804990432 |
21.716543443375 |
| ENSMUSG00000013367 |
210094 |
Iglon5 |
protein_coding |
Q8HW98 |
|
Disulfide bond;Glycoprotein;Immunoglobulin domain;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:210094; |
extracellular region [GO:0005576] |
Human_homologues 24703753_The target antigen of patients with rapid-eye-movement parasomnia who produce antibodies (mainly IgG4) against IgLON5 is a neuronal cell adhesion molecule that suggests a tauopathy 30956130_Findings report 89 unpublished cell-surface ligand-receptor pairs and describe structural models of trans interactions of cell adhesion molecules, neuronal (IgLON5), showing that their structures are compatible with a model of interaction across the synaptic cleft. 33531378_Anti-IgLON5 Disease: A New Bulbar-Onset Motor Neuron Mimic Syndrome. |
ENSG00000142549 |
IGLON5 |
62.257562 |
5.6553956388 |
2.499628 |
0.39369755 |
39.012307 |
0.00000000042114229032157475715702431748560345681209327040050993673503398895263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000062039065436926755231256468234735806932533819235686678439378738403320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
97.7480164397404 |
27.0067958716895 |
17.2840279766531 |
3.90260816761856 |
| ENSMUSG00000013415 |
140486 |
Igf2bp1 |
protein_coding |
O88477 |
FUNCTION: RNA-binding factor that recruits target transcripts to cytoplasmic protein-RNA complexes (mRNPs). This transcript 'caging' into mRNPs allows mRNA transport and transient storage. It also modulates the rate and location at which target transcripts encounter the translational apparatus and shields them from endonuclease attacks or microRNA-mediated degradation. Preferentially binds to N6-methyladenosine (m6A)-containing mRNAs and increases their stability (By similarity). Regulates localized beta-actin/ACTB mRNA translation, a crucial process for cell polarity, cell migration and neurite outgrowth. Co-transcriptionally associates with the ACTB mRNA in the nucleus. This binding involves a conserved 54-nucleotide element in the ACTB mRNA 3'-UTR, known as the 'zipcode'. The RNP thus formed is exported to the cytoplasm, binds to a motor protein and is transported along the cytoskeleton to the cell periphery. During transport, prevents ACTB mRNA from being translated into protein. When the RNP complex reaches its destination near the plasma membrane, IGF2BP1 is phosphorylated. This releases the mRNA, allowing ribosomal 40S and 60S subunits to assemble and initiate ACTB protein synthesis. Monomeric ACTB then assembles into the subcortical actin cytoskeleton (By similarity). During neuronal development, key regulator of neurite outgrowth, growth cone guidance and neuronal cell migration, presumably through the spatiotemporal fine tuning of protein synthesis, such as that of ACTB (By similarity). May regulate mRNA transport to activated synapses (By similarity). Binds to the 3'-UTR of CD44 mRNA and stabilizes it, hence promotes cell adhesion and invadopodia formation in cancer cells (By similarity). Binds to the oncofetal H19 transcript and regulates its localization (By similarity). Binds to and stabilizes BTRC/FBW1A mRNA (By similarity). Binds to the adenine-rich autoregulatory sequence (ARS) located in PABPC1 mRNA and represses its translation. PABPC1 mRNA-binding is stimulated by PABPC1 protein. Prevents BTRC/FBW1A mRNA degradation by disrupting microRNA-dependent interaction with AGO2 (By similarity). During cellular stress, such as oxidative stress or heat shock, stabilizes target mRNAs that are recruited to stress granules, including CD44, IGF2, MAPK4, MYC, PTEN, RAPGEF2 and RPS6KA5 transcripts (By similarity). Interacts with GAP43 transcript and transports it to axons. Binds to the 3'-UTR of IGF2 mRNA by a mechanism of cooperative and sequential dimerization and regulates IGF2 mRNA subcellular localization and translation. Binds to MYC mRNA, in the coding region instability determinant (CRD) of the open reading frame (ORF), hence prevents MYC cleavage by endonucleases and possibly microRNA targeting to MYC-CRD. Binding to MYC mRNA is enhanced by m6A-modification of the CRD (By similarity). Binds to and stabilizes ABCB1/MDR-1 mRNA. Binds to the neuron-specific TAU mRNA and regulates its localization. Plays a direct role in the transport and translation of transcripts required for axonal regeneration in adult sensory neurons. During interstinal wound repair, interacts with and stabilizes PTGS2 transcript. PTGS2 mRNA stabilization may be crucial for colonic mucosal wound healing. {ECO:0000250, ECO:0000250|UniProtKB:Q9NZI8, ECO:0000269|PubMed:15355996, ECO:0000269|PubMed:17264115, ECO:0000269|PubMed:21964071, ECO:0000269|PubMed:22465430}. |
Cell projection;Cytoplasm;Direct protein sequencing;mRNA transport;Nucleus;Phosphoprotein;Reference proteome;Repeat;RNA-binding;Synapse;Translation regulation;Transport |
|
|
mmu:140486; |
axon [GO:0030424]; CRD-mediated mRNA stability complex [GO:0070937]; cytoplasm [GO:0005737]; cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; filopodium [GO:0030175]; growth cone [GO:0030426]; lamellipodium [GO:0030027]; neuronal cell body [GO:0043025]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; P-body [GO:0000932]; perinuclear region of cytoplasm [GO:0048471]; ribonucleoprotein complex [GO:1990904]; mRNA 3'-UTR binding [GO:0003730]; mRNA 5'-UTR binding [GO:0048027]; mRNA binding [GO:0003729]; N6-methyladenosine-containing RNA binding [GO:1990247]; translation regulator activity [GO:0045182]; CRD-mediated mRNA stabilization [GO:0070934]; dendrite arborization [GO:0140059]; mRNA transport [GO:0051028]; negative regulation of nuclear-transcribed mRNA catabolic process, deadenylation-dependent decay [GO:1900152]; negative regulation of translation [GO:0017148]; nervous system development [GO:0007399]; neuronal stem cell population maintenance [GO:0097150]; pallium cell proliferation in forebrain [GO:0022013]; positive regulation of cytoplasmic translation [GO:2000767]; regulation of gene expression [GO:0010468]; regulation of mRNA stability involved in response to stress [GO:0010610]; regulation of RNA metabolic process [GO:0051252]; RNA localization [GO:0006403] |
14729626_WAP-CRD-BP mice are a novel model for mammary neoplasia and might provide insights into human breast cancer biology. 15086518_IMP-1 associates with HuD and G3BP-1 proteins in an RNA-dependent manner and binds directly to tau mRNA. We also show an RNA-dependent association between G3BP-1 and HuD proteins 21209918_IMP1 plays a role in regulating the packaging of MLV genomic RNA and can be used for improving production of retroviral vectors 21734271_This study demonstrate that ZBP1 and the phosphorylation of ZBP1 are required for attractive guidance of cortical neurons, and appropriate filopodial dynamics. 24704827_Results identify IGF2BP1 as a critical translational regulator of cIAP1-mediated apoptotic resistance in rhabdomyosarcoma. 25861986_High CRD-BP expression is associated with Breast Tumor. 26194191_The tumor-suppressive role of stromal IMP1 and its ability to modulate protumorigenic factors suggest that IMP1 status is important for the initiation and growth of epithelial tumors 28073938_This study showed that Zipcode binding protein 1 (ZBP1) is an mRNA-binding protein that regulates the local translation of proteins, including actin, in the growth cone and is required for Shh-mediated axon guidance. 32179813_microRNA-670 modulates Igf2bp1 expression to regulate RNA methylation in parthenogenetic mouse embryonic development. 32385106_The mRNA-binding protein IGF2BP1 maintains intestinal barrier function by up-regulating occludin expression. 33268291_Longitudinal epitranscriptome profiling reveals the crucial role of N(6)-methyladenosine methylation in porcine prenatal skeletal muscle development. 35179253_Silencing of IGF2BP1 restrains ox-LDL-induced lipid accumulation and inflammation by reducing RUNX1 expression and promoting autophagy in macrophages. 36067310_Tagged actin mRNA dysregulation in IGF2BP1[Formula: see text] mice. 36319624_Downregulation of METTL14 improves postmenopausal osteoporosis via IGF2BP1 dependent posttranscriptional silencing of SMAD1. 36632449_Inhibition of IGF2BP1 attenuates renal injury and inflammation by alleviating m6A modifications and E2F1/MIF pathway. |
ENSG00000159217 |
IGF2BP1 |
926.983673 |
7.2648266499 |
2.860928 |
0.96995958 |
7.543104 |
0.00602401758198455473253263292576775711495429277420043945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0218051687492120012390461880613656830973923206329345703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1583.2003081104 |
1146.4470706753 |
217.926783996794 |
114.343238309216 |
| ENSMUSG00000013483 |
170720 |
Card14 |
protein_coding |
Q99KF0 |
FUNCTION: Acts as a scaffolding protein that can activate the inflammatory transcription factor NF-kappa-B and p38/JNK MAP kinase signaling pathways. Forms a signaling complex with BCL10 and MALT1, and activates MALT1 proteolytic activity and inflammatory gene expression. MALT1 is indispensable for CARD14-induced activation of NF-kappa-B and p38/JNK MAP kinases. May play a role in signaling mediated by TRAF2, TRAF3 and TRAF6 and protects cells against apoptosis. {ECO:0000250|UniProtKB:Q9BXL6}. |
Apoptosis;Coiled coil;Cytoplasm;Phosphoprotein;Reference proteome |
|
|
mmu:170720; |
aggresome [GO:0016235]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; CARD domain binding [GO:0050700]; apoptotic process [GO:0006915]; negative regulation of apoptotic process [GO:0043066]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of response to stimulus [GO:0048584]; tumor necrosis factor-mediated signaling pathway [GO:0033209] |
29150564_in experimental models of psoriasis induced by either imiquimod cream or recombinant IL-23 injection, the psoriasiform skin inflammation was abrogated in Card14(-/-) mice 29689250_hyperactivation of CARD14 alone is sufficient to orchestrate the complex immunopathogenesis that drives T helper type 17-mediated psoriasis skin disease in vivo. 29980436_Caspase recruitment domain family member 14 (Card14)(E138A/+) and Card14(DeltaQ136/+) mice develop spontaneous psoriasis-like skin inflammation, resulted from constitutively activated CARMA2 via self-aggregation leading to the enhanced activation of the IL-23-IL-17A cytokine axis. Card14(-/-) mice display attenuated skin inflammation in the imiquimod-induced psoriasis model due to impaired IL-17A signaling in keratinoc... 32597759_CARD14(E138A) signalling in keratinocytes induces TNF-dependent skin and systemic inflammation. |
ENSG00000141527 |
CARD14 |
9.346653 |
9.9321906303 |
3.312112 |
1.11440642 |
8.827264 |
0.00296762977724770397186526693644736951682716608047485351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0118892521743916412119146386316970165353268384933471679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.1436761310086 |
16.3025437312697 |
1.92743744492824 |
1.33002147377988 |
| ENSMUSG00000013846 |
20442 |
St3gal1 |
protein_coding |
P54751 |
FUNCTION: A beta-galactoside alpha2-3 sialyltransferase involved in terminal sialylation of glycoproteins and glycolipids (PubMed:8375377, PubMed:9184827). Catalyzes the transfer of sialic acid (N-acetyl-neuraminic acid; Neu5Ac) from the nucleotide sugar donor CMP-Neu5Ac onto acceptor Galbeta-(1->3)-GalNAc-terminated glycoconjugates through an alpha2-3 linkage (PubMed:8144500, PubMed:8375377, PubMed:9184827, PubMed:10755614). Adds sialic acid to the core 1 O-glycan, Galbeta-(1->3)-GalNAc-O-Ser/Thr, which is a major structure of mucin-type O-glycans. As part of a homeostatic mechanism that regulates CD8-positive T cell numbers, sialylates core 1 O-glycans of T cell glycoproteins, SPN/CD43 and PTPRC/CD45. Prevents premature apoptosis of thymic CD8-positive T cells prior to peripheral emigration, whereas in the secondary lymphoid organs controls the survival of CD8-positive memory T cells generated following a successful immune response (PubMed:10755614). Transfers sialic acid to asialofetuin, presumably onto Galbeta-(1->3)-GalNAc-O-Ser (PubMed:8375377). Sialylates GM1a, GA1 and GD1b gangliosides to form GD1a, GM1b and GT1b, respectively (PubMed:8144500, PubMed:8375377, PubMed:9184827). {ECO:0000269|PubMed:10755614, ECO:0000269|PubMed:8144500, ECO:0000269|PubMed:8375377, ECO:0000269|PubMed:9184827}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lipid metabolism;Membrane;Reference proteome;Secreted;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000305|PubMed:10755614}.; PATHWAY: Glycolipid biosynthesis. {ECO:0000305|PubMed:8375377}. |
|
mmu:20442; |
extracellular region [GO:0005576]; Golgi medial cisterna membrane [GO:1990675]; Golgi trans cisterna membrane [GO:1990676]; membrane [GO:0016020]; trans-Golgi network membrane [GO:0032588]; beta-galactoside (CMP) alpha-2,3-sialyltransferase activity [GO:0003836]; monosialoganglioside sialyltransferase activity [GO:0047288]; ganglioside biosynthetic process via lactosylceramide [GO:0010706]; memory B cell differentiation [GO:0002319]; N-acetylneuraminate metabolic process [GO:0006054]; negative regulation of activated CD8-positive, alpha-beta T cell apoptotic process [GO:1905403]; protein glycosylation [GO:0006486]; protein N-linked glycosylation [GO:0006487]; protein phosphorylation [GO:0006468]; protein sialylation [GO:1990743]; sialylation [GO:0097503] |
10755614_St3gal-1 functions to protect CD8 T cells from apoptosis 12068010_tissue-specific alterations in sialylation and sialic acid 9-O-acetylation in knockout mice 12355428_ST3Gal-I mRNA is extensively expressed during Th2, but not Th1 differentiation; suggests that lack of ST3Gal-I expression in Th1 cells allows the formation of surface core2 O-glycans and supports their interactions with endothelial selectins 12393657_was not found to play a role in regulating synthesis of selectin ligands nor in vitro rolling of marrow-derived neutrophils on E- or P-selectins presented by Chinese hamster ovary cells 16751387_STsGal-I deficiency shows no discernible effect on the pattern of CD8 noncognate binding during thymocyte development and does not impact susceptibility of thymocytes to CD8-induced apoptosis. 20653283_tryptophan and cysteine residues conserved in ST3Gal I are critical for its activity 26552809_ST3GAL-1 is an independent adverse prognostic factor for recurrence and survival of patients with clear cell renal cell carcinoma |
ENSG00000008513 |
ST3GAL1 |
777.420193 |
0.4629424693 |
-1.111095 |
0.12689386 |
75.801617 |
0.00000000000000000313644178526690476023660069519933688270162097837061819516346616865121177397668361663818359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000001075997325972646139071136722144267273129272501032069331294849234836874529719352722167968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
474.505115357571 |
41.7894640837988 |
1024.97642119957 |
65.9923934109395 |
| ENSMUSG00000014603 |
11694 |
Alx3 |
protein_coding |
O70137 |
FUNCTION: Transcriptional regulator with a possible role in patterning of mesoderm during development. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
This gene belongs to Group 1 of aristaless-like genes, which are characterized by the presence of an aristaless domain and a conserved paired-like homeodomain. The encoded protein acts as a transcriptional regulator. The protein plays a role in the development of craniofacial and appendicular skeleton and may have a role in pancreatic function. [provided by RefSeq, Apr 2013]. |
mmu:11694; |
nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific double-stranded DNA binding [GO:1990837]; embryonic forelimb morphogenesis [GO:0035115]; embryonic hindlimb morphogenesis [GO:0035116]; embryonic skeletal system morphogenesis [GO:0048704]; pattern specification process [GO:0007389]; regulation of apoptotic process [GO:0042981]; regulation of DNA-templated transcription [GO:0006355]; regulation of transcription by RNA polymerase II [GO:0006357] |
20534379_Alx3-deficient mice exhibit folic acid-resistant craniofacial midline and neural tube closure defects. 21104068_ALX3 participates in the regulation of the expression of essential genes for the function of pancreatic islets, and its deficiency alters the regulation of glucose homeostasis in vivo. 21350340_ALX3 could be considered as a candidate gene for the etiopathogenesis of diabetes or its complications during embryonic or fetal development is discussed 23181698_Alx3 promoter activity in mesenchymal and pancreatic cells is regulatd by USF transcription factors and Twist1 23825702_results indicate that Alx3 expression is enhanced by BMP-2 via the BMP receptors mediated-Smad signaling and that Alx3 is a positive regulator of osteoblast differentiation induced by BMP-2 25040025_Pdx1, USF1 and USF2 co-ordinately regulate Alx3 gene expression in pancreatic beta-cells. 26739814_Glucose-stimulated expression of ALX3 in alpha cells provides a regulatory mechanism for the downregulation of glucagon gene expression by interfering with PAX6-mediated transactivation on the glucagon G1 promoter element. 28341857_Study shows that ALX3 deficiency provides a novel molecular mechanism for developmental defects arising from maternal hyperglycaemia. 31114073_Alx3, a transcription factor of the homeobox gene family, was pivotal for prenatal tooth development but subsided in the adult.Alx3 promoted stroma regeneration via VEGF signaling. |
ENSG00000156150 |
ALX3 |
104.691205 |
2.4197207708 |
1.274841 |
0.28064639 |
20.512800 |
0.00000592338046682768161493208916579256140266807051375508308410644531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000457762208375034186191680773969636675246874801814556121826171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
144.26123751389 |
28.2403575210966 |
59.6189606894315 |
9.06549366800349 |
| ENSMUSG00000014668 |
231600 |
Chfr |
protein_coding |
Q810L3 |
FUNCTION: E3 ubiquitin-protein ligase that functions in the antephase checkpoint by actively delaying passage into mitosis in response to microtubule poisons. Acts in early prophase before chromosome condensation, when the centrosome move apart from each other along the periphery of the nucleus. Probably involved in signaling the presence of mitotic stress caused by microtubule poisons by mediating the 'Lys-48'-linked ubiquitination of target proteins, leading to their degradation by the proteasome. Promotes the ubiquitination and subsequent degradation of AURKA and PLK1. Probably acts as a tumor suppressor, possibly by mediating the polyubiquitination of HDAC1, leading to its degradation. May also promote the formation of 'Lys-63'-linked polyubiquitin chains and functions with the specific ubiquitin-conjugating UBC13-MMS2 (UBE2N-UBE2V2) heterodimer. Substrates that are polyubiquitinated at 'Lys-63' are usually not targeted for degradation, but are rather involved in signaling cellular stress (By similarity). {ECO:0000250|UniProtKB:Q96EP1, ECO:0000269|PubMed:15793587}. |
ADP-ribosylation;Alternative splicing;Cell cycle;Cell division;Metal-binding;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Transferase;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:231600; |
nucleus [GO:0005634]; PML body [GO:0016605]; metal ion binding [GO:0046872]; nucleotide binding [GO:0000166]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin-protein transferase activity [GO:0004842]; cell division [GO:0051301]; meiotic spindle checkpoint signaling [GO:0044779]; mitotic cell cycle [GO:0000278]; mitotic G2/M transition checkpoint [GO:0044818]; modification-dependent protein catabolic process [GO:0019941]; positive regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032436]; positive regulation of protein ubiquitination [GO:0031398]; protein destabilization [GO:0031648]; protein polyubiquitination [GO:0000209]; protein ubiquitination [GO:0016567]; ubiquitin-dependent protein catabolic process [GO:0006511] |
15467728_PML bodies control the distribution, dynamics and function of CHFR 15793587_Downregulation of Chfr contributes to tumorigenesis by controlling the expression levels of Aurora A. 19690386_Mice with deficiencies in both Chfr and Mlh1 (the gene that encodes the DNA mismatch-repair protein Mlh1) displayed dramatically higher incidence of spontaneous tumors relative to mice deficient for only one of these genes. 21245198_Stil protein regulates centrosome integrity and mitosis through suppression of Chfr. 21706008_RNF8 and Chfr, function together to activate ATM and maintain genomic stability in vivo. 22285184_CHFR interacts with BRG1, SNF5, and BAF60a of the SWI/SNF-like BAF complex and ubiquitinates them to target for degradation through a proteasome-mediated pathway; SRG3 stabilizes these components by blocking their interaction with CHFR. 22337872_The interaction between CHFR and PARP-1 plays an important role in cell cycle regulation and cancer therapeutic strategies. 26566864_CHFR is important for the survival of male premeiotic germ cells, which is likely through maintaining genomic stability in spermatogonial stem cells. |
ENSG00000072609 |
CHFR |
599.103999 |
0.4944986526 |
-1.015962 |
0.13983790 |
52.344409 |
0.00000000000046572262783241167825451021931916855919365849647562072277651168406009674072265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000097043598729730439809540100624353964244711567843637567420955747365951538085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
407.769067144032 |
39.5862581431511 |
824.611078295667 |
58.9109284410661 |
| ENSMUSG00000014905 |
27362 |
Dnajb9 |
protein_coding |
Q9QYI6 |
FUNCTION: Co-chaperone for Hsp70 protein HSPA5/BiP that acts as a key repressor of the ERN1/IRE1-mediated unfolded protein response (UPR) (By similarity). J domain-containing co-chaperones stimulate the ATPase activity of Hsp70 proteins and are required for efficient substrate recognition by Hsp70 proteins (PubMed:11836248). In the unstressed endoplasmic reticulum, interacts with the luminal region of ERN1/IRE1 and selectively recruits HSPA5/BiP: HSPA5/BiP disrupts the dimerization of the active ERN1/IRE1 luminal region, thereby inactivating ERN1/IRE1 (By similarity). Also involved in endoplasmic reticulum-associated degradation (ERAD) of misfolded proteins (PubMed:22267725). Required for survival of B-cell progenitors and normal antibody production (PubMed:25222125). {ECO:0000250|UniProtKB:G3H0N9, ECO:0000269|PubMed:11836248, ECO:0000269|PubMed:22267725, ECO:0000269|PubMed:25222125}. |
Chaperone;Endoplasmic reticulum;Phosphoprotein;Reference proteome;Signal;Unfolded protein response |
|
|
mmu:27362; |
cytoplasm [GO:0005737]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum lumen [GO:0005788]; nucleolus [GO:0005730]; chaperone binding [GO:0051087]; Hsp70 protein binding [GO:0030544]; misfolded protein binding [GO:0051787]; B cell differentiation [GO:0030183]; negative regulation of IRE1-mediated unfolded protein response [GO:1903895]; positive regulation of immunoglobulin production [GO:0002639]; response to endoplasmic reticulum stress [GO:0034976]; response to unfolded protein [GO:0006986]; ubiquitin-dependent ERAD pathway [GO:0030433] |
11836248_DnaJ homolog, which stimulates ATPase activity of BiP and is induced by endoplasmic reticulum stress 22267725_directly link ERdj4 to the ERAD machinery and suggest a model in which ERjd4 could help recruit clients from throughout the ER to ERAD sites 24336520_Deficiency of the BiP cochaperone ERdj4 causes constitutive endoplasmic reticulum stress and metabolic defects. 25146923_these results demonstrate that DNAJB9 is a downstream target of p53 that belongs to the group of negative feedback regulators of p53. 25222125_ERdj4 gene trap mice showed non-specific hypergammaglobulinemia and gene trap B cells exhibited increased proliferation, survival and isotype switching in response to LPS stimulation. |
ENSG00000128590 |
DNAJB9 |
510.426419 |
0.4637095274 |
-1.108707 |
0.11791260 |
87.886508 |
0.00000000000000000000693199058674815117600769553547977052902608290109832885365556687240484734502388164401054382324218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000002815682149795942559555593558886485599585120746870143268805741776361628581071272492408752441406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
317.813232575978 |
26.0040545493219 |
685.371366693023 |
40.4009420375297 |
| ENSMUSG00000015053 |
14461 |
Gata2 |
protein_coding |
O09100 |
FUNCTION: Transcriptional activator which regulates endothelin-1 gene expression in endothelial cells. Binds to the consensus sequence 5'-AGATAG-3'. |
Activator;DNA-binding;Isopeptide bond;Metal-binding;Methylation;Nucleus;Phagocytosis;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:14461; |
cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; C2H2 zinc finger domain binding [GO:0070742]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription coactivator binding [GO:0001223]; transcription coregulator binding [GO:0001221]; zinc ion binding [GO:0008270]; angiogenesis [GO:0001525]; brown fat cell differentiation [GO:0050873]; cell differentiation in hindbrain [GO:0021533]; cell fate commitment [GO:0045165]; cell fate determination [GO:0001709]; central nervous system neuron development [GO:0021954]; cochlea development [GO:0090102]; commitment of neuronal cell to specific neuron type in forebrain [GO:0021902]; definitive hemopoiesis [GO:0060216]; embryonic placenta development [GO:0001892]; eosinophil fate commitment [GO:0035854]; GABAergic neuron differentiation [GO:0097154]; glandular epithelial cell differentiation [GO:0002067]; glandular epithelial cell maturation [GO:0002071]; hematopoietic progenitor cell differentiation [GO:0002244]; hematopoietic stem cell homeostasis [GO:0061484]; homeostasis of number of cells within a tissue [GO:0048873]; inner ear morphogenesis [GO:0042472]; myeloid cell differentiation [GO:0030099]; negative regulation of brown fat cell differentiation [GO:1903444]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of fat cell proliferation [GO:0070345]; negative regulation of gene expression [GO:0010629]; negative regulation of hematopoietic progenitor cell differentiation [GO:1901533]; negative regulation of macrophage differentiation [GO:0045650]; negative regulation of myeloid cell differentiation [GO:0045638]; negative regulation of neural precursor cell proliferation [GO:2000178]; negative regulation of neuroblast proliferation [GO:0007406]; negative regulation of Notch signaling pathway [GO:0045746]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural precursor cell proliferation [GO:0061351]; neuroblast proliferation [GO:0007405]; neuroendocrine cell differentiation [GO:0061101]; neuron differentiation [GO:0030182]; neuron fate commitment [GO:0048663]; neuron migration [GO:0001764]; phagocytosis [GO:0006909]; pituitary gland development [GO:0021983]; positive regulation of angiogenesis [GO:0045766]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:1903589]; positive regulation of cell migration involved in sprouting angiogenesis [GO:0090050]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of erythrocyte differentiation [GO:0045648]; positive regulation of gene expression [GO:0010628]; positive regulation of mast cell degranulation [GO:0043306]; positive regulation of megakaryocyte differentiation [GO:0045654]; positive regulation of miRNA transcription [GO:1902895]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of phagocytosis [GO:0050766]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of forebrain neuron differentiation [GO:2000977]; regulation of histone acetylation [GO:0035065]; regulation of primitive erythrocyte differentiation [GO:0010725]; response to lipid [GO:0033993]; semicircular canal development [GO:0060872]; somatic stem cell population maintenance [GO:0035019]; thyroid-stimulating hormone-secreting cell differentiation [GO:0060129]; transcription by RNA polymerase II [GO:0006366]; urogenital system development [GO:0001655]; vascular wound healing [GO:0061042]; ventral spinal cord interneuron differentiation [GO:0021514] |
11804789_Gata-2 transcripts accumulate rapidly in the fetal ovary from 11.5 days post coitum (dpc) onwards, but are not detected in the fetal testis throughout the period studied (10.5-15.5 dpc). Ovarian expression of Gata-2 ceases by 15.5 dpc 11956331_transcription factor GATA-2 plays a significant role in the expression of the skeletal muscle L-type Ca(2+) channel alpha(1S). 12041667_Gata1, Gata2, and Gata3 bind to the TGATA site of the mEpo promoter and negatively regulates mEpo gene expression. 12045236_Essential and instructive roles of GATA factors in eosinophil development. 12065417_Results suggest that GATA-2, Fli-1 and Elf-1 are key components of an enhanceosome responsible for activating SCL transcription and establishing the transcriptional programme required for HSC formation. 12065419_role in the development and differentiation of hematopoietic progenitors; conditional activation of a GATA-2/ER chimera produced opposite effects to those observed with conditional GATA-2 expression 12077323_FOG-1 requires an interaction with either GATA-1 or -2 as part of its essential role in early megakaryopoiesis. 12385825_multiple domains of Pit-1 are required for full synergy with GATA-2 and sequences between the two binding sites contribute to co-occupancy with both factors on the proximal TSH beta promoter 12393444_regulation of cytokine-dependent growth of hematopoietic cells through accumulation of p21(WAF1) and p27(Kip1) proteins 12689939_that GATA-2 is expressed in the precursor of hematopoietic cells and works as a gatekeeper to preserve their immaturity. A reduction of GATA-2 expression or activity is required for the differentiation of precursors to hematopoietic cells. 12857954_GATA-1 instigates GATA-2 repression by means of disruption of positive autoregulation, followed by establishment of a domain-wide repressive chromatin structure. 14504093_Either GATA-1 or GATA-2 is required to initiate blood formation in the embryo 15215894_Globin gene activation during hematopoiesis is driven by protein complexes nucleated by Gata-1 and Gata-2. 15254248_there is functional cross talk between RA and GATA-2-dependent pathways 15329349_data establish Gata2 and Gata3 as essential members of the transcription factor network controlling sympathetic neuron development 15466621_GATA-2 plays a role in the production and expansion of hematopoietic stem cells (HSCs) in the aorta-gonad-mesonephros and the proliferation of HSCs in the adult bone marrow. 15494394_GATA-2 transcription is regulated via the collective actions of complexes assembled at the -2.8- and -1.8-kb regions 15499560_Gata2 is predominantly expressed in the dorsal vestibular system in developing inner ear 15542836_Gata2 enhancers specify temporally and tissue-specific patterning in the developing urogenital system 15673499_GATA1 and GATA2 modulates cytokine signals through protein-protein interactions, and inhibiting STAT3, thereby regulating the growth and survival of hematopoietic cells 15689374_activation of Gata2 expression by Notch1/RBPjkappa is a crucial event for the onset of definitive hematopoiesis in the embryo 15811962_GATA-2 gene dose regulates adult stem-cell homeostasis by affecting select aspects of stem cell function. 15889140_Evi1 directly binds to GATA-2 promoter as an enhancer. GATA-2 is presumably one of critical targets for Evi1 and that transcription factors regulate the hematopoietic stem cell pool hierarchically. 16055713_the Gata2 gene first exon promoter is regulated by a GATA factor and selectively marks putative hematopoietic/endothelial precursor cells within the para-aortic splanchnopleura 16082385_GATA2 functions to suppress brown adipocyte differentiation, whereas reduction of GATA2 promotes brown adipogenesis. 16151640_high expression of GATA-2 resulted in cell death and elevated alpha globin levels in FL5.12 cells; findings suggest a role of GATA-2 in apoptosis besides its function in maintenance and proliferation of immature hematopoietic progenitors 16396960_TSHbeta gene is maximally activated by a combination of TRAP220, Pit-1, GATA-2 that act via both protein-DNA and protein-protein interactions. 16461905_A Gata2 promoter directs activity in all hematopoietic stem cells. 16543408_Gata2-deficient mice exhibit elevated levels of Gata3 transcripts in the pituitary gland, suggesting that GATA3 can compensate for GATA2. 16621992_support a role for GATA-2 in allowing Dll-1 to restrict non-T cell lineage differentiation outcomes 16628252_GATA and Ikaros proteins coordinately fine-tune accessibility at the Il4 locus during development to variably regulate IL-4 expression 16672344_Gata2 can operate independently of neuronal differentiation 17166922_GATA2 plays specific, contextual roles in the generation of Flk-1+ mesoderm, the Flk-1+Scl+ hemangioblast, primitive erythroid and endothelial cells. 17255359_GATA2 phosphorylation by Cdk/cyclin systems is responsible for the cell-cycle-dependent regulation of GATA2 expression, which may control hematopoietic-cell proliferation and survival. 17327407_Gata1, -2, and -3 transgenes driven by Gata1 regulatory elements rescue the Gata1-null phenotype 17395646_GATA-2 is expressed early in lymphatic, cardiac and blood vascular endothelial cells, and the pan-endothelium-specific expression of Gata2 is controlled by a discrete intronic enhancer. 17823307_the intronic Runx1 enhancer targets all definitive HSCs in the mouse embryo, suggesting that it functions as a crucial cis-regulatory element that integrates the Gata, Ets, and SCL transcriptional networks to initiate HSC generation. 17950250_GATA-2 maintains a hematopoietic microenvironment by regulating the differentiation of adipocytes. 17962413_This hematopoietic stem cell enhancer ia bound by transription factors in stem cells and in hemangioblasts. 18154321_Differential GATA-1 and GATA-2 factor stability is an important determinant of chromatin target site occupancy and therefore the establishment of genetic networks that control hematopoiesis. 18202227_GATA-2 and GATA-3 bind to the GATA box in distal tubular cells, and are likely to repress constitutively ectopic Epo gene expression in these cells. 18212046_Gata2 (but not GATA3 or GATA5) mRNA was significantly expressed in Sertoli cells. Gata2 transcripts were present in both the Sertoli and interstitial cell fractions. GATA2 is present in Sertoli, Leydig, and germ cells. 18233958_Bmp4 mRNA is reduced approximately twofold in Gata2(fGN)/(/fGN) mice (as in Bmp4 null heterozygotes), and that GATA-2 trans-activates a Bmp4 first intron element-directed reporter plasmid in co-transfection assays. 18243117_In immature cells, a distal enhancer bound by GATA-2 is in physical proximity with the active Kit promoter. Upon cell maturation, GATA-1 displaces GATA-2 and triggers a loss of the enhancer/promoter interaction. 18246306_CREB-binding proteins is a coactivator of GATA-2 and GATA-1 18840712_GATA-2 regulates myeloid progenitor function through HES-1. 19036901_Site-directed mutagenesis and chromatin immunoprecipitation assays showed that progestin responsiveness is mediated through a composite element in the first intron, to which the PR binds concomitantly with GATA-2. 19036967_Our findings identify a direct transcriptional interaction between Gata-2 and FEV and a unique marker for new insight into FEV/Pet-1 function in 5-HT neuron development. 19088086_Gata2 regulates GABAergic neuron development in the midbrain. Without Gata2, all the precursors in the embryonic midbrain fail to activate GABAergic neuron-specific gene expression and instead switch to a glutamatergic phenotype. 19106099_GATA3/GATA2 switch is an important mechanism for the transcriptional regulation of other trophoblast-specific genes. 19120268_Lowered expressions of the NF-kappaB family members in dendritic cells from NOD mice are associated with a reduced expression of GATA-2. 19136559_peroxisome proliferator-activated receptor-gamma-independent down-regulation of GATA2 prevents reversion of mature adipocytes after PPARgamma depletion 19371385_The 2H and 5H domains have the independent ability to activate Gata2 gene expression in the midbrain superior colliculus, whereas the distal-5H domain is additionally capable of activating Gata2 transcription in the inferior colliculus. 19491391_PU.1/Sfpi1 expression is regulated by sequential binding of GATA-2 and GATA-1 to highly conserved sequences. A GATA-2 to GATA-1 switch at PU.1/Sfpi1 coincides with histone deacetylation and gene repression. 19528235_Results suggest that increased c-Jun expression due to mutant Shp2-induced Ras hyperactivation, and reduced GATA2 expression promote aberrant monocytic differentiation induced by activating PTPN11 mutants. 19620289_GATA-2 contributes to cell cycle progression and the maintenance of megakaryocyte identity of GATA-1-deficient cells. 20652952_Data show that Gata2 is required from E14.5-E15.5 onward for vestibular morphogenesis, as without Gata2 the semicircular ducts fail to grow to their normal size and surrounding mesenchymal cells are not removed to generate the perilymphatic space. 20808444_the expression of the VE-statin/egfl7 gene in endothelial cells and the specific importance of ETS and GATA factors 20833840_GATA2 and Sp1 play crucial roles in expression of the c-kit gene in mast cells 20838598_Cis element regulation of the critical hematopoietic factor Gata2, which is expressed in early precursors and repressed as GATA-1 levels rise during terminal differentiation, was analyzed. 21321116_Dll1/Notch signaling reduces GATA-1 and GATA-2 expression in bone marrow-derived mast cells. 21472823_expression of Gata2, Mash1, and Foxa2 synergizes to induce the serotonergic neuron phenotype during in vitro differentiation of embryonic stem cells 21947280_PPARgamma promotes osteoclast progenitors by activating GATA2 transcription 22383799_GATA factors GATA-1 and GATA-2 bind overlapping sets of genes, often at distinct sites, as a means to differentially regulate target gene expression and to regulate the balance between proliferation and differentiation. 22476030_Study reports that GATA2 protein is expressed in the uterine luminal and glandular epithelium pre-implantation, spatio-temporally co-localizing with that of the progesterone receptor. 22493062_GATA-2 expression in the caudal urogenital mesenchyme as directed by the UG4 enhancer is crucial for proper development of the urinary tract and that its regulation of BMP4 expression is a critical aspect of this function 22499991_Injury-induced repression of miR-200b turned on wound angiogenesis. In mice with diabetes mellitus,excessive tumor necrosis factor-alpha induced miR-200b blunting proangiogenic functions of GATA2 and VEGFR2. 22541434_In a Kras-driven NSCLC mouse model, Gata2 loss dramatically reduced tumor development. Furthermore, Gata2 deletion in established Kras mutant tumors induced striking regression 22988301_Gata2 mRNA level was significantly increased while Gata1 was maintained at low levels during the differentiation of mast cells derived from mouse bone marrow. 22991444_results suggest a role for Gata2 as a selector gene of both GABAergic neuron neurotransmitter and prosomere subtype identities in the developing diencephalon 22996659_The composite E-box/GATA-motif element disrupted in a human immunodeficiency is essential for establishment of the murine hematopoietic stem/progenitor cell compartment in the fetal liver and for essential vascular processes. 22996665_The vascular-endothelial enhancer regulates GATA-2 expression in fetal liver & adult BM HSCs. GATA-2 function is required for proper lymphatic vascular development during embryogenesis. 23065276_Methylated C bases in the C/EBPbeta promoter relate to expression of the C/EBPbeta gene, and that its demethylation is linked with GATA-2 protein association. 23322776_SON protein regulates GATA-2 through transcriptional control of the microRNA 23a~27a~24-2 cluster 23592430_Progesterone regulates Ednra expression by synergizing with Gata2 activity. 23732910_This study demonstrates that Ikaros directly interacts with GATA1, GATA2, and GATA3 as well as Cdk9/P-TEFb through specific protein domains. 23840778_Gata2 mRNA levels were almost completely rescued by expression of an erythroid lineage restricted ROSA26-promotor based GATA2 transgene 23892628_GATA2 and Lmo2 cooperatively regulate VEGF-induced angiogenesis and lymphangiogenesis via NRP2. 23911012_preceding binding of GATA2 helps the following binding of GATA1 and thereby secures smooth expression of the transient-phase genes. 23940812_Results of this study show that LPS induction of il-1beta gene expression in macrophages is mediated by GATA-2 via activation of TLR4, MyD88, and MAPKs. 24297994_a Gata2 cis-element (+9.5) is required for hematopoietic stem cell generation in the mammalian embryo 24297996_GATA2 is essential for hematopoietic stem cell survival 24583263_Gata2 functions as a cofactor of Etv2 in the transcriptional regulation of mesodermal progenitors during embryogenesis. 24615013_GATA2 plays a more important role than GATA1 in the regulation of most mast cell-specific genes. 24616499_established a Gata2 repression/reactivation assay, which revealed unique +9.5 site activity to mediate GATA factor-dependent chromatin structural transitions 24636993_GATA2 regulates the Aqp2 gene expression in collecting duct cells and contributes to the maintenance of the body water homeostasis 24807155_GATA2 is epigenetically repressed in human and mouse lung tumors and its further inhibition is not a valid therapeutic strategy for KRAS mutant lung cancer. 25150255_GATA2 plays an important role in regulating the differentiation potential of BM-MSC and contributes to hematopoietic supporting capacity. 25305367_Data indicate that peroxisome-proliferator activated receptor gamma (PPARgamma) promoter harbors leptin response region that includes a GATA binding protein-2 (GATA-2) binding site. 25369423_Results provide new insights into the selective roles of Gata2 within the genetic model of V2 interneurons development and in particular on the hierarchy between V2-transcription factors 25398936_we demonstrate that GATA2 acts downstream of IRF8 in regulating basophil and mast cell development 25670854_Ubiquitin-dependent degradation of GATA 2 is promoted by Fbw7, is cyclin B-CDK1-mediated Thr176 phosphorylation-dependent, and influences hematopoietic cell differentiation. 25801432_Our study reveals that the STAT5-GATA2 pathway is critical for basophil and mast cell differentiation and maintenance. 25848090_These results suggest that Setd8 is an important regulator of erythroid maturation that works in part through repression of Gata2 expression. 25855601_these results indicate that GATA2 maintains cellular identity in a subpopulation of mast cells, whereas it plays a fundamental role as a positive regulator of mast cell-specific genes throughout development of this cell lineage. 25870111_FLI1 and GATA2 are upstream regulators of SMAD1 and SMAD5 expression in endothelial cells. 26124638_LSD1 mediates erythroid differentiation, via epigenetic modification of the GATA-2 locus. 26204127_Sox17 inhibition of Runx1 and Gata2 maintains endothelial fate in endothelial-to-haematopoietic transition 26214525_Conditional Gata2 deletion in mice revealed that GATA2 is required for both development and maintenance of lymphovenous and lymphatic vessel valves. 26660446_we identified important roles for the GATA-2 C-ZnF in bone marrow hematopoiesis via control of c-Kit expression and HSC/HSPC survival. 26812161_Gata2 as a crucial rheostat of mesenchymal stem cell fate to control osteoblast and adipocyte lineage development. 26876150_Data show that transforming growth factor beta (TGFbeta)-induced changes in Gata2 transcription factor and cyclin-dependent kinase inhibitor 1C (P57) expression in hematopoietic progenitors are conveyed through Smad signaling via Smad4 protein. 27131743_These results demonstrated that an enforced expression of Gata2 in late HECs of differentiated ESCs efficiently promotes megakaryopoiesis followed by platelet production. This study provides valuable information for ex vivo platelet production from human pluripotent stem cells in future. 27259979_results suggest that GATA2 plays an important role in cell-fate specification toward the myeloid vs T-lymphocyte lineage by regulating lineage-specific transcription factors in DC progenitors, thereby contributing to DC differentiation. 27443289_High GATA2 expression is associated with leukemia. 27783953_Gata2 regulates a key regulatory network of gene expression for progesterone signaling at the early pregnancy stage. 27789623_both Gata2 and Gata3 are redundantly required for differentiation of the serotonergic and glutamatergic neurons of the dorsal raphe 28232602_impairment of trophoblast-specific GATA2/GATA3 function could lead to early pregnancy failure 28334937_Dynamically and epigenetically coordinated GATA2/ETS1SOX7 transcription factor expression is indispensable for endothelial cell differentiation. 28569748_endothelial SENP1-mediated SUMOylation drives graft arteriosclerosis by regulating the synergistic effect of GATA2 and NF-kappaB and consequent endothelial dysfunction. 28630119_Gata2 heterozygous deletion confers selective advantage to EVI1-expressing leukemia cell expansion in recipient mice 28807932_Results show that GATA2 exerts a pro-inflammatory function in injured kidney collecting duct cells, by upregulating inflammatory cytokine gene expression thereby contributes to disease progression. 28866324_GATA2 could affect the function of BM-MSCs in vivo, presumably by regulating the expression of extracellular signals. 28943250_We demonstrate that overexpression of Gfi1b, c-Fos, and Gata2, previously reported to transdifferentiate fibroblasts into hematopoietic progenitors in vitro, can induce long-term HSC formation in vivo. 29277702_GATA2 is required for connective tissue mast cell differentiation. GATA2 and MITF regulate IgE-mediated anaphylaxis. 29581184_By regulating bone trabecularization, GATA2 expression in the osteogenic lineage may contribute to the anatomical and cellular microenvironment of the hematopoietic stem cell niche required for hematopoiesis. 29666442_Analyses of mouse models demonstrate a cell-autonomous function of GATA2 in regulating lymphatic endothelial cell (LEC) responsiveness to VEGF-C and in controlling LEC migration and sprouting in vivo. 30044048_NR4A1 retards adipocyte differentiation or maturation via enhancing GATA2 and p53 expression. 30088690_Enforced expression of Gata2 in embryonic stem cells-derived hemogenic endothelial cells enhances erythroid and megakaryocyte differentiation and inhibits macrophage differentiation. 30615908_Study showed that the GATA2 repression during adipocyte differentiation is mediated by glucocorticoid receptor (GR) at a transcriptional level in 3T3-L1 cells. Several lines of evidence suggest that direct DNA binding of GR may not be necessary for the Gata2 repression but by GR-reducing histone acetylation in the early phase of adipogenesis. 30620726_Single-nucleotide human disease mutation inactivates a blood-regenerative GATA2 enhancer. 30916783_Study identified GATA2 as a feedback inhibitor of retinoic acid signalling important for precise onset of ureteric smooth muscle cell differentiation. 31378673_GATA2 acts as a critical regulator of normal and leukemic stem cells and mediates transcriptional networks that may be exploited therapeutically to target key facets of leukemic stem cells behavior in acute myeloid leukemia. 31501274_Results suggest that PU.1 and LDB1 play central roles in the formation of active chromatin structure whereas GATA2 directly activates the Ms4a2 promoter. 31624376_Gata2 deficiency delays leukemogenesis while contributing to aggressive leukemia phenotype in Cbfb-MYH11 knockin mice. 32330245_EVI1 and GATA2 misexpression induced by inv(3)(q21q26) contribute to megakaryocyte-lineage skewing and leukemogenesis. 32649900_A Novel GATA2 Protein Reporter Mouse Reveals Hematopoietic Progenitor Cell Types. 33193444_Efferocytic Defects in Early Atherosclerosis Are Driven by GATA2 Overexpression in Macrophages. 33479210_GATA2 regulates mast cell identity and responsiveness to antigenic stimulation by promoting chromatin remodeling at super-enhancers. 33547048_E-cadherin is regulated by GATA-2 and marks the early commitment of mouse hematopoietic progenitors to the basophil and mast cell fates. 33864419_GATA2 participates in the recanalization of lymphatic vessels after surgical lymph node extirpation. 34078881_Gata2-L359V impairs primitive and definitive hematopoiesis and blocks cell differentiation in murine chronic myelogenous leukemia model. 34125173_Tumor suppressor function of Gata2 in acute promyelocytic leukemia. 34492681_Redundant mechanisms driven independently by RUNX1 and GATA2 for hematopoietic development. 34741809_Cell-cycle dependent GATA2 subcellular localization in mouse 2-cell embryos. 35181392_Specific effects of somatic GATA2 zinc finger mutations on erythroid differentiation. 35440757_Heterozygous variants in GATA2 contribute to DCML deficiency in mice by disrupting tandem protein binding. 35643321_Transcription factor GATA2 may potentiate follicle-stimulating hormone production in mice via induction of the BMP antagonist gremlin in gonadotrope cells. 36444820_GATA2 promotes oxidative stress to aggravate renal ischemia-reperfusion injury by up-regulating Redd1. |
ENSG00000179348 |
GATA2 |
56.153640 |
2.9341940557 |
1.552964 |
0.37086370 |
17.219986 |
0.00003329155489234536004217179794295589090324938297271728515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002223134432793531355815330741165780636947602033615112304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
89.3101533140896 |
23.2120378058624 |
30.4377118964116 |
6.17421485617355 |
| ENSMUSG00000015222 |
17756 |
Map2 |
protein_coding |
P20357 |
FUNCTION: The exact function of MAP2 is unknown but MAPs may stabilize the microtubules against depolymerization. They also seem to have a stiffening effect on microtubules. |
Calmodulin-binding;Cell projection;Cytoplasm;Cytoskeleton;Direct protein sequencing;Microtubule;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:17756; |
actin cytoskeleton [GO:0015629]; apical dendrite [GO:0097440]; apical distal dendrite [GO:0150014]; axon hillock [GO:0043203]; axon initial segment [GO:0043194]; basal dendrite [GO:0097441]; CA3 pyramidal cell dendrite [GO:0097442]; cell body [GO:0044297]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; dendrite cytoplasm [GO:0032839]; dendritic branch [GO:0044307]; dendritic filopodium [GO:1902737]; dendritic growth cone [GO:0044294]; dendritic shaft [GO:0043198]; distal dendrite [GO:0150002]; glutamatergic synapse [GO:0098978]; microtubule [GO:0005874]; microtubule associated complex [GO:0005875]; microtubule cytoskeleton [GO:0015630]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; nuclear periphery [GO:0034399]; postsynapse [GO:0098794]; postsynaptic density [GO:0014069]; primary dendrite [GO:0150001]; protein-containing complex [GO:0032991]; proximal dendrite [GO:1990635]; proximal neuron projection [GO:1990769]; rough endoplasmic reticulum [GO:0005791]; ruffle membrane [GO:0032587]; smooth endoplasmic reticulum [GO:0005790]; actin binding [GO:0003779]; calmodulin binding [GO:0005516]; cytoskeletal regulatory protein binding [GO:0005519]; dystroglycan binding [GO:0002162]; microtubule binding [GO:0008017]; protein kinase binding [GO:0019901]; axonogenesis [GO:0007409]; cellular response to organic substance [GO:0071310]; dendrite development [GO:0016358]; establishment of cell polarity [GO:0030010]; microtubule bundle formation [GO:0001578]; microtubule cytoskeleton organization [GO:0000226]; negative regulation of axon extension [GO:0030517]; negative regulation of microtubule binding [GO:1904527]; negative regulation of microtubule depolymerization [GO:0007026]; negative regulation of microtubule polymerization [GO:0031115]; neuron projection development [GO:0031175]; positive regulation of anterograde dense core granule transport [GO:1901953]; positive regulation of anterograde synaptic vesicle transport [GO:1903744]; regulation of organelle transport along microtubule [GO:1902513]; regulation of protein localization [GO:0032880] |
11829530_Nitric oxide affects the phosphorylation state of microtubule-associated protein 2 in brain. 11891784_Participation of structural microtubule-associated proteins (MAPs) in the development of neuronal polarity. 12763072_PKA binding to MAP2 and MAP2 phosphorylation is essential for the selective development of contextual memory. 14588114_Multiple brain regions exhibit potential for endogenous recovery and/or reorganization of MAP2 after injury of moderate traumatic severity. 15306236_As young adults, S100B transgenic mice exhibit significantly greater MAP-2-immunoreactivity in the hippocampus, however in older adults, less MAP-2 staining is exhibited. 16000625_JNK1 phosphorylation of MAP2 plays an important role in defining dendritic architecture in the brain. 16257389_We also propose MAP2-deficient mice as an ideal system for identifying protein phosphatases essential for dendritic functions. 16330151_MAP2 was increased in old females compared with both young and middle-age females with no significant difference in males, suggesting it may constitute a compensatory response to age-related functional decline in mice. 16901895_TTLL7 is a mammalian beta-tubulin polyglutamylase required for growth of MAP2-positive neurites 17984326_v-KIND mediates a signaling pathway that links Ras and MAP2 to control dendrite growth 18716213_AKAP150 and Mtap2 put TREK channels at the center of a complex protein network that finely tunes channel trafficking, addressing, and regulation. 20100193_Increased MAP2 expression in malignant melanoma cells significantly inhibits tumor cell growth and neoplasm invasiveness, and induces apoptosis and cell cycle arrest. 20103640_Data show that inhibition of ZNF238 (RP58) expression in mouse GNPs decreases the expression of the neuronal differentiation markers MAP2 and NeuN and downregulates the expression of the cell cycle arrest protein p27, a regulator of GNP differentiation. 21385318_results demonstrate the structural and functional determinant underlying the v-KIND-MAP2 interaction that controls dendrite arborization patterns 23372648_Accumulation of intraneuronal beta-amyloid 42 peptides is associated with early changes in microtubule-associated protein 2 in neurites and synapses. 24965600_MAP2c levels in different brain regions of aging mice were positively correlated with neuropsin expression. 31452242_Loss of tau and Fyn reduces compensatory effects of MAP2 for tau and reveals a Fyn-independent effect of tau on calcium. 33526823_MAP2 is differentially phosphorylated in schizophrenia, altering its function. |
ENSG00000078018 |
MAP2 |
157.537298 |
0.4924028962 |
-1.022089 |
0.28543251 |
12.651537 |
0.00037525696085926441404947984104012448369758203625679016113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0019388731226536600436849422379737006849609315395355224609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
104.596266443365 |
20.8293005125556 |
212.420087779526 |
31.0036909583868 |
| ENSMUSG00000015354 |
76477 |
Pcolce2 |
protein_coding |
Q8R4W6 |
FUNCTION: Binds to the C-terminal propeptide of types I and II procollagens and may enhance the cleavage of that propeptide by BMP1. {ECO:0000250}. |
Disulfide bond;Glycoprotein;Heparin-binding;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:76477; |
collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; collagen binding [GO:0005518]; heparin binding [GO:0008201]; peptidase activator activity [GO:0016504]; cellular response to leukemia inhibitory factor [GO:1990830] |
12063410_Assignment of the mouse Pcolce2 gene, which encodes procollagen C-proteinase enhancer protein 2, to chromosome 9. 12393877_PCPE2 is shown to be a glycoprotein that differs markedly in the nature of its glycosylation from that of PCPE1 21771977_the role of the PCPE2 protein in an in vivo model 22610170_It was shown that PCOLCE2 is required for efficient procollagen processing and deposition of fibrillar collagen in the pressure-overloaded myocardium. 25947382_PCPE2 was shown to enhance SR-BI function by increasing the rate of HDL-associated cholesteryl ester uptake, possibly by optimizing SR-BI localization and/or conformation. 34551590_Pcpe2, a Novel Extracellular Matrix Protein, Regulates Adipocyte SR-BI-Mediated High-Density Lipoprotein Uptake. |
ENSG00000163710 |
PCOLCE2 |
277.869985 |
0.1113644101 |
-3.166640 |
0.26947321 |
124.384679 |
0.00000000000000000000000000006939732237395373314859779421885035585931548621543300063442379500875976597188202199539830417052144184708595275878906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000049165582065119225187121223189810219281888096291958833495460544084559113744287150993272916821297258138656616210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
55.4252622252292 |
10.4128877918688 |
497.692774151785 |
65.108871315917 |
| ENSMUSG00000015355 |
12506 |
Cd48 |
protein_coding |
P18181 |
FUNCTION: Glycosylphosphatidylinositol (GPI)-anchored cell surface glycoprotein that interacts via its N-terminal immunoglobulin domain with cell surface receptors including 2B4/CD244 or CD2 to regulate immune cell function and activation (PubMed:9881969, PubMed:21278219). Participates in T-cell signaling transduction by associating with CD2 and efficiently bringing the Src family protein kinase LCK and LAT to the TCR/CD3 complex. In turn, promotes LCK phosphorylation and subsequent activation (PubMed:9881969). Induces the phosphorylation of the cytoplasmic immunoreceptortyrosine switch motifs (ITSMs) of CD244 initiating a series of signaling events that leads to the generation of the immunological synapse and the directed release of cytolytic granules containing perforin and granzymes by T-lymphocytes and NK-cells (By similarity) (PubMed:9881969, PubMed:21278219). {ECO:0000250|UniProtKB:P09326, ECO:0000269|PubMed:21278219, ECO:0000269|PubMed:9881969}. |
3D-structure;Cell membrane;Direct protein sequencing;Disulfide bond;Glycoprotein;GPI-anchor;Immunoglobulin domain;Lipoprotein;Membrane;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:12506; |
external side of plasma membrane [GO:0009897]; extracellular region [GO:0005576]; membrane raft [GO:0045121]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; mast cell activation [GO:0045576]; signal transduction [GO:0007165]; T cell activation [GO:0042110] |
12356317_molecular mechanism of heterophilic adhesion between the murine T-cell adhesion glycoprotein CD2 and its ligand CD48 12734329_CD48 interactions with NK cell receptor 2B4 among neighboring T cells during T cell priming augment CTL lysis of their specific targets. 15778370_Analysis of the ligand-receptor pairs involved in induction of germline transcripts (Igamma2a) necessary for switch recombination revealed that expression of CD48 on B cells and ligands CD2 and CD244 on NK cells are able to stimulate Igamma2a 15870174_compare the relative contributions of MHC class I and CD48 to NK cell self-tolerance; and CD244 regulates NK cell tolerance to normal cells in vivo; evidence for MHC-independent NK self-recognition in a bone marrow rejection assay 15989959_Cell surface receptors of the SLAM family, including CD150, CD244, and CD48, were differentially expressed among functionally distinct hematopoietic progenitor cells. 16081768_Review of recent studies suggests an important role for interactions between 2B4 and CD48 in the course of T cell activation and proliferation 16472597_T cell-induced experimental colitis is ameliorated only when CD48 is absent from both T cells and antigen-presenting cells. 17599905_the mechanism of signal transduction by CD244 is to regulate FYN kinase recruitment and/or activity and the outcome of CD48/CD244 interactions is determined by which other receptors are engaged. 17884794_CD137 and CD28 can promote bcl-2 expression at both mRNA and protein level in T cells. 18826951_nanoscale increases in CD2-CD48-mediated intermembrane spacing decrease adhesion and reorganize the immunological synapse 20647560_CD48 on surrounding NK or non-NK cells serves primarily as a ligand to stimulate 2B4 on the adjacent NK cells in mice 21221603_Ox-LDL significantly promoted the interaction of natural killer cells and dendritic cells via CD48-2B4 contact-dependent mechanisms. 21561736_B6-specific background genes modulate the effect of CD48 on lupus nephritis in a recessive manner 21576698_CD48 plays a vital role as an environmental sensor for regulating hematopoietic stem cells and progenitor cell numbers and inhibiting tumor development 24056518_ORMDL3 promotes eosinophil trafficking and activation via regulation of CD18, CD49d, and CD48. 25142868_Molecular dynamic simulation explains unbinding and unfolding of CD48-2B4 protein complexes. 25255823_These data demonstrate the important role of CD48 in SA/exotoxins-eosinophil activating interactions that can take place during allergic responses and indicate CD48 as a novel therapeutic target for allergy and especially of AD. 36520044_Boosting of tau protein aggregation by CD40 and CD48 gene expression in Alzheimer's disease. |
ENSG00000117091 |
CD48 |
26.926641 |
3.9839086831 |
1.994185 |
0.52343777 |
14.495555 |
0.00014049068957551624097813969260784006110043264925479888916015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008118187342997963814802964144234920240705832839012145996093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.9735101635311 |
16.3067892939586 |
11.0377806475711 |
3.27078194794039 |
| ENSMUSG00000015501 |
15273 |
Hivep2 |
protein_coding |
Q3UHF7 |
FUNCTION: Specifically binds to the DNA sequence 5'-GGGACTTTCC-3' which is found in the enhancer elements of numerous viral promoters such as those of SV40, CMV, or HIV1. In addition, related sequences are found in the enhancer elements of a number of cellular promoters, including those of the class I MHC, interleukin-2 receptor, somatostatin receptor II, and interferon-beta genes. It may act in T-cell activation (By similarity). {ECO:0000250, ECO:0000269|PubMed:10207097}. |
DNA-binding;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:15273; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; DNA-templated transcription [GO:0006351]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; signal transduction [GO:0007165] |
15699073_Data suggest that Schnurri-2 plays a crucial role in the control of T helper type 2 cell differentiation by regulating NF-kappaB function. 16836985_Shn-2 plays a critical role in locomotion and anxiety-like behavior. Stress-induced corticosterone levels were significantly higher in Shn-2(-/-) mice compared to wild-type controls. 17404274_Shn-2 appears to control the generation of memory Th1/Th2 cells through a change in their susceptibility to cell death. 17493960_Shn-2 in Th2 cells plays an important role as a negative regulator in allergic airway inflammation. 19448624_These results newly identify Schnurri-2 and CLIC4 as modifiers of TGF-beta signalling through their stabilization of p-Smad2 and 3 in the nucleus. 20404140_report that mice bearing parallel null mutations in the adapter proteins Schnurri2 (Shn2) and Schnurri3 (Shn3) exhibit defects in patterning of the axial skeleton during embryogenesis. 20795536_Results indicate that Schnurri-2-mediated repression of NF-kappaB is required for cell survival and the successful generation of memory Th1/Th2 cells. 21069746_Shn2-deficient mice maintain bone mass at the levels comparable to wild-type sham mice even after ovariectomy-induced bone loss. 21475200_repression of T cell receptor-induced death pathways is critical for proper interpretation of positive selecting signals in vivo; identification of schnurri-2 (Shn2; or Hivep2) as a crucial death dampener 21936769_Shn-2 plays an important role in the activation and function of NK cells and the development of T cell lymphoma in vivo. 23389689_Schnurri-2 knockout mice have behavioral abnormalities that resemble those of schizophrenics. 29233179_Shn2 knockout mice serve as a unique tool for investigating morphological abnormalities of subcellular-scale structures in schizophrenia, intellectual disability, and its related disorders. 30285890_the Shn2 deficiency attenuates bidirectional short-term synaptic plasticity at the medial perforant path-granule cell synapse. 35218885_Schnurri-2 Promotes the Expression of Excitatory Glutamate Receptors and Contributes to Neuropathic Pain. |
ENSG00000010818 |
HIVEP2 |
567.333347 |
3.5188085402 |
1.815087 |
0.66699439 |
6.981188 |
0.00823709259006657687485208896305266534909605979919433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0284152172116622324382628761441083042882382869720458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
864.241623703084 |
413.947648864176 |
245.606322091082 |
86.6873544318041 |
| ENSMUSG00000015533 |
16398 |
Itga2 |
protein_coding |
Q62469 |
FUNCTION: Integrin alpha-2/beta-1 is a collagen receptor, being responsible for adhesion of platelets and other cells to collagens, modulation of collagen and collagenase gene expression, force generation and organization of newly synthesized extracellular matrix. It is also a receptor for laminins, collagen C-propeptides and E-cadherin. Mice homozygous for a null mutation in the alpha-2 die very early in embryogenesis. |
Calcium;Cell adhesion;Disulfide bond;Glycoprotein;Integrin;Magnesium;Membrane;Metal-binding;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:16398; |
axon [GO:0030424]; axon terminus [GO:0043679]; basal part of cell [GO:0045178]; cell projection [GO:0042995]; cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; focal adhesion [GO:0005925]; integrin alpha2-beta1 complex [GO:0034666]; integrin complex [GO:0008305]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; presynapse [GO:0098793]; amyloid-beta binding [GO:0001540]; collagen binding [GO:0005518]; collagen binding involved in cell-matrix adhesion [GO:0098639]; collagen receptor activity [GO:0038064]; integrin binding [GO:0005178]; laminin binding [GO:0043236]; metal ion binding [GO:0046872]; protein-containing complex binding [GO:0044877]; cell adhesion mediated by integrin [GO:0033627]; cell population proliferation [GO:0008283]; cell-cell adhesion [GO:0098609]; cell-matrix adhesion [GO:0007160]; cell-substrate adhesion [GO:0031589]; cellular response to estradiol stimulus [GO:0071392]; cellular response to mechanical stimulus [GO:0071260]; collagen-activated signaling pathway [GO:0038065]; detection of mechanical stimulus involved in sensory perception of pain [GO:0050966]; establishment of protein localization [GO:0045184]; female pregnancy [GO:0007565]; focal adhesion assembly [GO:0048041]; hepatocyte differentiation [GO:0070365]; hypotonic response [GO:0006971]; integrin-mediated signaling pathway [GO:0007229]; mammary gland development [GO:0030879]; mesodermal cell differentiation [GO:0048333]; positive regulation of alkaline phosphatase activity [GO:0010694]; positive regulation of cell adhesion [GO:0045785]; positive regulation of cell projection organization [GO:0031346]; positive regulation of collagen binding [GO:0033343]; positive regulation of collagen biosynthetic process [GO:0032967]; positive regulation of DNA binding [GO:0043388]; positive regulation of epithelial cell migration [GO:0010634]; positive regulation of inflammatory response [GO:0050729]; positive regulation of leukocyte migration [GO:0002687]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of positive chemotaxis [GO:0050927]; positive regulation of smooth muscle cell migration [GO:0014911]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of smooth muscle contraction [GO:0045987]; positive regulation of translation [GO:0045727]; positive regulation of transmission of nerve impulse [GO:0051971]; response to amine [GO:0014075]; response to hypoxia [GO:0001666]; response to L-ascorbic acid [GO:0033591]; response to muscle activity [GO:0014850]; response to parathyroid hormone [GO:0071107]; response to xenobiotic stimulus [GO:0009410]; skin morphogenesis [GO:0043589]; substrate-dependent cell migration [GO:0006929]; wound healing [GO:0042060] |
12960301_Integrin alpha 2 is rapidly induced and expressed on effector CD8+ T cells; its blockade inhibits adoptive transfer of antigen-specific delayed-type hypersensitivity responses. 14645004_that alpha 2 beta 1 integrin provides a costimulatory function required for mast cell activation and cytokine production in response to infection. 14739220_platelet aggregation response to collagen correlates with alpha(2) expression; alpha(2)beta(1) participates in signaling during platelet activation 15132990_VEGF-A induced alpha1 & alpha2 integrins, promoting lymphatic endothelial tube formation & haptotactic migration. Lineage-specific integrin receptor expression contribute to the distinct dynamics of wound-associated angiogenesis & lymphangiogenesis. 15227729_The distribution of GPIa (C807T/G873A) and HPA-3 genotypes did not differ significantly between normal subjects and coronary artery disease subjects 15886326_studies establish platelet-collagen responses under physiologic flow as the consequence of a close partnership between 2 structurally distinct receptors, glycoprotein VI and integrin alpha2beta1 16166590_alpha2beta1 integrin is a novel receptor for multiple collectins and the C1q complement protein 16373693_high level expression of alpha2 selectively marks a subset of primitive hematopoietic cells which retains multilineage reconstitution potential but exhibits reduced self-renewal capacity 16455949_decreased surface alpha2beta1 results from decreased Itga2 pre-mRNA splicing regulated by hnRNP L and depends on CA repeat length at a specific site in intron 1 16636073_Results describe the role of alpha2beta1 integrin in the interactions of primary fibroblasts and keratinocytes with the extracellular matrix. 16818752_Although the inflammatory properties of the CD49b-expressing subset of memory T helper cells are protective against intracellular bacterial infection, they are associated with immunopathology in acute viral infection. 16882656_extracellular matrix fragments produced by apoptotic EC initiate a state of resistance to apoptosis in fibroblasts via an alpha2beta1 integrin/SFK (Src and Fyn)/phosphatidylinositol 3-kinase (PI3K)-dependent pathway 16977325_Ablation of alpha2beta1 resulted in strong enhancement of neovascularization of granulation tissue and sponge implants. Alpha2beta1-null fibroblasts are hampered in generating mechanical forces. 17032352_Integrins may play an important role in lamellipodia formation. 17068473_Ligation negatively regulates expression of genes including matrix metalloproteinases. Deletion of alpha2beta1 integrin resulted in dramatic increase in neoangiogenesis in wound microenvironment. (Alpha2beta1 integrin) 17638891_CD44+alpha2beta1+ cell population is enriched in tumor-initiatin prostate cancer cells 18000613_integrin alpha2beta1 plays a role in stabilizing murine thrombi, likely by enhancing GPVI activation and thromboxane A(2) release. 18198349_crosstalk between c-met and the alpha2beta1 integrin may contribute to mast-cell activation in autoimmune and inflammatory disorders. 18436621_Cell-surface expression of the alpha2beta1-integrin plays a role in the mechanism that confers cholangiocyte susceptibility to rotavirus infection 18802064_The CD49b-positive CD8 T cell population is a far more potent antitumor effector cell population than nonfractionated CD8 or CD49b-negative CD8 T cells; expression of CD49b (VLA-2) may serve as a surrogate marker for successful tumor vaccination. 18990704_analysis of a prokaryotic Scl1 collagen sequence motif that mediates binding to human collagen receptors, integrins alpha2beta1 and alpha11beta1 19192113_Rap1b is required for maximal GPVI-induced but not ADP-induced activation of alpha2beta1 in murine platelets. 19258597_Ligand-dependent removal of activated alpha2 receptors from the cell surface negatively regulates integrin function in hematopoietic cells during migration between an adhesive environment (bone marrow) & a nonadhesive one (circulating blood). 19321657_Suggest that the recruitment of peripheral CXCR4-expressing ITGA2(+)ISG20(+) NK cells into pregnant uteri may be important in the regulation of feto-maternal tolerance. 19411307_alpha(2)-, alpha(3)-, and beta(1)-integrins and E-cadherin expression in normal human lung and bronchopulmonary sequestration and congenital cystic adenomatoid malformation were evaluated using Western blot and immunohistochemistry. 19463772_beta2 integrins, although required for inflammatory migration in acute asthma, are dispensable for structural remodeling in chronic asthma 19668337_MMP7 shedding of syndecan-1 facilitates wound closure by causing the alpha(2)beta(1) integrin to assume a less active conformation thereby removing restrictions to migration 19789387_Integrin alpha2beta1 is required for endorepellin induction of tyrosine phosphatase activity. 19815553_enhance adhesion through alpha2beta1 integrin to collagens via a PLC-dependent priming mechanism. 19840869_transgenic mice have altered keratinocyte differentiation and proliferation, which results in chronic immune cell activation 21134100_Results suggest a novel mechanism for pathogen entry into host cells as well as a new function for C1q- alpha2beta1 integrin interactions. 21135504_alpha2beta1 integrin suppresses metastasis in a clinically relevant spontaneous mouse model of breast cancer 21344393_Collagen I stimulates the self-renewal of embryonic stem cells mediated by Bmi-1 through alpha2beta1 integrin-dependent ILK, Notch, Gli-1, and DDR1-dependent Ras, PI3K/Akt, and ERK. 21558389_PHLDA1 expression marks the putative epithelial stem cells, downregulates ITGA2 and ITGA6, and contributes to intestinal tumorigenesis 21897373_show that PilA binds collagen, which promotes GBS interaction with the alphabeta integrin resulting in activation of host chemokine expression and neutrophil recruitment during infection 22046385_alpha2beta1 integrin expression by HPV-transformed keratinocytes modulates squamous carcinoma growth and progression 22083543_The loss of alpha2betaA1 integrin suppresses joint inflammation and cartilage destruction in mouse models of rheumatoid arthritis. 22103309_ADAP plays a role in mediating platelet activation via the collagen-binding integrin alpha(2) beta(1). 22210772_Circulating IGF-1 levels in the sera of mice lacking either alpha2beta1 or alpha11beta1 or both integrins were sharply reduced. 22318609_beta-arr1 has a role in promoting thrombus formation, in part through its participation in alphaIIbbeta3 signalling, but neither beta-arr1 or beta-arr2 have a role in agonist-induced platelet activation and P2Y receptors desensitisation 23165626_Expression of integrin alpha2 receptor in human cord blood CD34+CD38-CD90+ stem cells determines its long-term engrafting in NOD/SCID-IL2Rgamma(c) null mice. 23226541_These data show for the first time that decorin has an impact on the biology of alpha2beta1 integrin and the vimentin intermediate filament system. 23359821_observed a significant reduction in mean platelet volume, which is consistent with the reported role of alpha2beta1 in MK maturation and proplatelet formation in vivo 23385480_Blocking integrin alpha2beta1 and ERK 1/2 pathways alleviated the effects of AMBN on osteoclast differentiation. 23448258_TNF-alpha stimulated cerebral pericyte remodeling in an alpha2 integrin-dependent manner. 23680479_the absence of integrin alpha2beta1 alleviates the effects of age related bone degradation through over-expression of collagen type I. 23897120_required for the migration of memory CD4 T-cell precursors into their survival niches of the bone marrow 24244022_alpha2beta1 integrin is expressed on synovial Th17 cells from CIA mice. Its neutralization reduced inflammation, cartilage degradation, bone erosion, RANKL & IL-17 levels, & the numbers of osteoclasts and TH17 cells. 24480069_the collagen receptors DDR1 and integrin a2b1 contribute to regulate GBM-maturation and to control matrix accumulation. 24917135_alpha2beta1 integrin contributes to the pathogenesis of retinopathy. We describe a newly identified role for alpha2beta1 integrin in mediating hypoxia-induced Muller cell VEGF-A production. 24939421_Synthesis and modeling of linear and cyclic KTS-containing peptides is described that leads to their use as inhibitors of integrin alpha1beta1/alpha2beta1-mediated adhesion and other antiangiogenetic activity in human serum. 25352128_alphaIIbbeta3 regulates Syk through an ITAM-independent pathway in mice and provide novel insight into the course of events underlying Syk activation and hemITAM phosphorylation by CLEC-2 25375377_IFN-gamma induces aberrant CD49b+ NK cell recruitment and pregnancy failure through regulating CX3CL1. 25808553_LG3, through interactions with alpha2beta1 integrins on recipient-derived cells leading to activation of ERK1/2 and increased migration, favors myointimal thickening 26051322_HRG binds to alpha2 integrin through low-affinity interactions in a heparin sulfate-independent manner, thereby blocking endothelial cells adhesion to collagen I. 26196244_Ceacam1 is essential for normal integrin aIIbb3-mediated platelet function; the disruption of mouse Ceacam1 induced moderate integrin aIIbb3-mediated functional defects. 26556301_A sulfonamide-type small-molecule inhibitor of alpha2beta1 integrin decreased average platelet adhesion in individuals with the C/T807T genotype but not in those harboring C807C. 28251149_low expression of alpha2beta1 integrin increased protein kinase B (AKT) activation and alpha-smooth muscle actin (alpha-SMA) expression. This occurred due to the instability of phosphatase and tensin homolog (PTEN) in myofibroblasts on fibrillar collagen. 28389839_P15 does not directly activate integrins by binding, but does upregulate integrin signaling to enhance differentiation of both osteoblasts and chondrocytes to increase both intramembranous and endochondral bone formation. 29333090_Study in a mouse model of collagen-induced arthritis identified the integrin alpha2 gene (Itga2) as an additional and novel biomarker for rheumatoid arthritis. 30355617_CD49b defines functionally mature Treg cells that survey skin and vascular tissues. 32325713_Increased Collagen Turnover Impairs Tendon Microstructure and Stability in Integrin alpha2beta1-Deficient Mice. 32692932_Type I Collagen Signaling Regulates Opposing Fibrotic Pathways through alpha2beta1 Integrin. 32998468_The Commensal Microbiota Enhances ADP-Triggered Integrin alphaIIbbeta3 Activation and von Willebrand Factor-Mediated Platelet Deposition to Type I Collagen. 35182768_Characterization of Glycoproteoforms of Integrins alpha2 and beta1 in Megakaryocytes in the Occurrence of JAK2V617F Mutation-Induced Primary Myelofibrosis. |
ENSG00000164171 |
ITGA2 |
19.717818 |
0.3139395119 |
-1.671441 |
0.62271138 |
7.135748 |
0.00755621106660182405984071962734560656826943159103393554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0264224533899171214190726431070288526825606822967529296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.50957951548058 |
3.78811190213693 |
27.1057932873958 |
8.38903700832036 |
| ENSMUSG00000015647 |
16776 |
Lama5 |
protein_coding |
Q61001 |
FUNCTION: Binding to cells via a high affinity receptor, laminin is thought to mediate the attachment, migration and organization of cells into tissues during embryonic development by interacting with other extracellular matrix components. Alpha-5 may be the major laminin alpha chain of adult epithelial and/or endothelial basal laminae. |
3D-structure;Basement membrane;Cell adhesion;Coiled coil;Direct protein sequencing;Disulfide bond;Extracellular matrix;Glycoprotein;Laminin EGF-like domain;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:16776; |
basement membrane [GO:0005604]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular matrix of synaptic cleft [GO:0098965]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; glutamatergic synapse [GO:0098978]; laminin-10 complex [GO:0043259]; laminin-5 complex [GO:0005610]; neuromuscular junction [GO:0031594]; synaptic cleft [GO:0043083]; extracellular matrix structural constituent [GO:0005201]; integrin binding [GO:0005178]; animal organ morphogenesis [GO:0009887]; axon guidance [GO:0007411]; branching involved in salivary gland morphogenesis [GO:0060445]; branching involved in ureteric bud morphogenesis [GO:0001658]; branching morphogenesis of an epithelial tube [GO:0048754]; cell migration [GO:0016477]; cell-cell adhesion [GO:0098609]; cilium assembly [GO:0060271]; hair follicle development [GO:0001942]; integrin-mediated signaling pathway [GO:0007229]; kidney development [GO:0001822]; lung development [GO:0030324]; morphogenesis of a polarized epithelium [GO:0001738]; morphogenesis of embryonic epithelium [GO:0016331]; muscle organ development [GO:0007517]; neural crest cell migration [GO:0001755]; odontogenesis of dentin-containing tooth [GO:0042475]; postsynapse organization [GO:0099173]; protein localization to plasma membrane [GO:0072659]; regulation of cell adhesion [GO:0030155]; regulation of cell migration [GO:0030334]; regulation of embryonic development [GO:0045995]; regulation of epithelial cell proliferation [GO:0050678]; substrate adhesion-dependent cell spreading [GO:0034446]; tissue development [GO:0009888]; trunk neural crest cell migration [GO:0036484] |
12051813_role in lobar septation and visceral pleural basement membrane formation in developing mouse lung 12061820_several peptides of Laminin alpha 5 had cell attachment activity and the ability to promote neurite outgrowth. 12244066_Results demonstrate that the alpha5 LG3 module is essential for the Lutheran blood group glycoprotein (Lu) binding to laminin alpha5. 12519075_Beta1 integrin and alpha-dystroglycan binding sites are localized to different laminin-G-domain-like (LG) modules within the G domain of this protein. 12631063_The absence of a alpha3(IV), alpha4(IV), alpha5(IV) network may stimulate reexpression of laminin-1 by Alport mouse endothelial cells and podocytes 12743034_Data suggest that laminin-10 (alpha5beta1gamma1) is required for hair follicle development and report the first use of exogenous protein to correct a cutaneous developmental defect. 12921739_the laminin alpha5 chain is essential for normal development of the intestinal smooth muscle and point to possible mesenchyme-derived compensation to promote normal intestinal morphogenesis when laminin alpha5 is absent. 14568553_crucial role for the LG4 module of laminin-alpha5 in epithelial morphogenesis that is distinct from that of the laminin-alpha1 LG4. 15661925_Cryptic matrix epitope AQARSAASKVKVSMKF of laminin alpha 5 induces diverse inflammatory cell activities, including chemotaxis and expression of proteinases, cytokines, and cytokine receptors. 15936333_Epithelial laminin alpha5 is necessary for murine lung development through signaling pathways that promote alveolar epithelial cell differentiation and VEGF expression. 16316641_Laminin alpha5, a gene up-regulated during neural crest induction, is localized in regions of newly formed cranial and trunk neural folds and adjacent neural crest migratory pathways in a manner largely conserved between chick and mouse. 16365040_alpha5 is required for proliferation and polarity of basal epithelial cells; the interaction between laminin-10/11-integrin alpha6beta4 and the PI3-kinase-Cdc42/Rac pathways may play a role in determining the size and shape of tooth germ 16790509_A new mouse PKD model has been produced through insertion of a PGKneo cassette in an intron of the gene that encodes laminin alpha5 (Lama5). Lama5neo/neo mice exhibit PKD, proteinuria, and death from renal failure by 4 wk of age. 17000025_The major contributions of laminin-5 to the resistance of the epidermis against frictional stress but also for basement membrane regeneration and repair of damaged skin are reflected by the phenotype of Herlitz junctional epidermolysis bullosa. 17599968_podocyte foot process differentiation may require direct exposure to laminin alpha5 17601529_laminin alpha5 controls submandibular gland epithelial morphogenesis through beta1 integrin signaling by regulating FGFR expression, which also reciprocally regulates the expression of Lama5 17699809_Upregulation of Lama5 transcription and concentration of laminin alpha1 and alpha5 within Alport glomerular basement membrane thickenings contribute to abnormal permeabilities. 18628307_Laminin alpha 5 has a crucial role in establishing and maintaining the specific mucosal pattern of the mouse small intestine 18794334_These results show that laminins alpha4, alpha5, and beta2 play an autocrine role in coordinating post- with presynaptic maturation. 19396173_Laminin alpha5 directly inhibits integrin alpha(6)beta(1)-mediated migration of T lymphocytes through laminin alpha4. 20226633_Downregulation of laminin-511 is important for hair regression. 21067603_Data provides support for a primary role of laminin-511 and integrin beta-1 promoting hair follicle epithelial downgrowth without affecting dermal primary cilia and Shh target gene induction. 21311558_crystal structure of laminin alpha5LN-LE1-2 fragment shows the LN domain is a beta-jelly roll with several elaborate insertions; surface loop is required for stable ternary association with the beta- and gamma-short arms in the laminin network 22391228_most of the integrin-mediated cell adhesive peptides are located in the loop regions in the G domains, suggesting that structure is important for the integrin specific recognition 22621685_amino acid residues within the short arm region of alpha5 that were critical for neurite outgrowth activity 22666383_mechanistic role of laminin-511 in tissue homeostasis 22761447_alpha1- and alpha5-containing laminins differentially regulate the sequential events to form epithelial tissues via beta1 integrin signals. 22911783_These results suggest that laminin trimers containing alpha5 that are synthesized and secreted by endothelium or by trophoblasts are capable of integrating into the basement membrane and promoting normal vascularization of the placenta. 23529140_Data suggest that the proper expression of laminin-332 (Lamb3) and laminin-511 (lama5) may regulate appropriate hair morphogenesis. 23755407_Laminin alpha 5 (Lama5) mRNA expressions in the lung of newborn in the nicotine treated Exp D1 decreased by 0.63 fold. 24036115_Lutheran (Lu), also known as basal cell adhesion molecule (B-CAM), competes with integrins for binding to laminin alpha5, a subunit of LM-511, a major component of basement membranes. 24691446_Laminins affect T cell trafficking and allograft fate. 27064881_According to the results, it seems that maternal nicotine exposure may induce abnormal laminin alpha 5 expression which may cause defects in kidney function during life time. 27208857_Using standard histological analyses and electron microscopy, we characterized the morphology of the external genitalia in Lama5 knockout (LAMA5-KO) mouse embryos during both androgen-independent genital tubercle development and androgen-mediated sexual differentiation 27234307_the overriding effect of laminin 511 in the skin is inhibition of basal keratinocyte proliferation and migration. 27940654_The authors show that the integral endothelial basement membrane component, laminin 511 (laminin alpha5), is central to shear detection and mechanotransduction and its elimination at this site results in ablation of dilation in response to increased shear stress. 28831020_Study found that the extracellular matrix protein laminin-511 (LM511) promoted the survival and differentiation of dopaminergic neurons in the midbrain neurons. LM511 bound to integrin alpha3beta1 and activated the transcriptional cofactor YAP. 29906214_Immunofluorescence microscopy revealed that laminin-511, -332, and collagen type IV proteins were also significantly increased in psoriasis-like skin lesions of Imiquimod-treated mice. 30273664_Laminin511 controls the differentiation of melanocytes by regulating their migration from the epidermis into hair follicles (HFs) and by activating CXCR4 expression on melanocytes required for their recruitment into HFs in an SDF1-dependent manner. 30777135_Mural cell-derived laminin-alpha5 plays a detrimental role in ischemic stroke. 31343575_In diverse models, laminins alpha-4 (LAMA4) and alpha-5 (LAMA5) were differentially regulated. Immunity was associated with decreased LAMA4:LAMA5 ratio, while tolerance was associated with an increased ratio. LAMA4inhibited CD4+ T-cell proliferation and Th1, Th2, and Th17 polarization but favored Treg induction. LAMA4 and LAMA5 are coinhibitory and costimulatory ligands for human and mouse CD4 T cells, respectively. 31882770_Laminin alpha2, alpha4, and alpha5 Chains Positively Regulate Migration and Survival of Oligodendrocyte Precursor Cells. 32017712_The lymph node stromal laminin alpha5 shapes alloimmunity. 33629734_Canonical Wnt signaling regulates branching morphogenesis of submandibular gland by modulating levels of lama5. 34128985_Laminin alpha 5 regulates mammary gland remodeling through luminal cell differentiation and Wnt4-mediated epithelial crosstalk. 34161758_Intra-vessel heterogeneity establishes enhanced sites of macromolecular leakage downstream of laminin alpha5. 34605523_Laminin-511 and recombinant vitronectin supplementation enables human pluripotent stem cell culture and differentiation on conventional tissue culture polystyrene surfaces in xeno-free conditions. 35008625_Regulation and Function of Laminin A5 during Mouse and Human Decidualization. |
ENSG00000130702 |
LAMA5 |
86.265395 |
0.2874732593 |
-1.798500 |
0.51962081 |
10.291272 |
0.00133660929874587688231946103201153164263814687728881835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0059722008913162790214035346991749975131824612617492675781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
35.8224707531125 |
13.1830858412338 |
124.611488529024 |
33.4286774803398 |
| ENSMUSG00000015709 |
11864 |
Arnt2 |
protein_coding |
Q61324 |
FUNCTION: Transcription factor that plays a role in the development of the hypothalamo-pituitary axis, postnatal brain growth, and visual and renal function. Specifically recognizes the xenobiotic response element (XRE). {ECO:0000250|UniProtKB:Q9HBZ2}. |
Alternative splicing;DNA-binding;Methylation;Nucleus;Reference proteome;Repeat;Transcription;Transcription regulation |
|
|
mmu:11864; |
aryl hydrocarbon receptor complex [GO:0034751]; cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; aryl hydrocarbon receptor binding [GO:0017162]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; protein heterodimerization activity [GO:0046982]; protein-containing complex binding [GO:0044877]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; brain development [GO:0007420]; central nervous system development [GO:0007417]; in utero embryonic development [GO:0001701]; negative regulation of apoptotic process [GO:0043066]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of transcription by RNA polymerase II [GO:0006357]; response to hypoxia [GO:0001666]; response to toxic substance [GO:0009636]; response to xenobiotic stimulus [GO:0009410]; signal transduction [GO:0007165] |
15466261_data suggest that ARNT2/HIF-2beta is required at high levels during nephrogenesis in distal tubules and later exclusively in thick ascending limb 17023418_The differential binding of AhR by Arnt and Arnt2 can be ascribed to a single His/Pro amino acid difference in the PASB region of Arnt and Arnt2. 19074369_Cap-analysis of gene expression (CAGE) data points to Arnt2 as a key regulator in hippocampus. 30231889_The data of this study support ARNT2 as a neuronal transcription factor whose sustained expression is linked to neuronal and axonal health, protection that may primarily be driven through its partnering with Npas4 to influence BDNF expression. 30563851_These findings establish a requirement for ARNT2-dependent genes in the maintenance of the homeostatic feeding response, necessary for prevention of obesity and obesity-related diseases. 30846309_In the absence of synaptic excitation, ARNT2 recruits the NCoR2 co-repressor complex to suppress neuronal activity-dependent regulatory elements and maintain low basal levels of inducible genes. in response to heightened neuronal activity, ARNT2 recruits NPAS4 to activity-dependent regulatory elements to induce transcription and thereby increase somatic inhibitory input. |
ENSG00000172379 |
ARNT2 |
73.255770 |
0.1887696464 |
-2.405301 |
0.57579107 |
16.064885 |
0.00006120856353459329878517747314248254042468033730983734130859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003825723593594276546007992845233047773945145308971405029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.0156909742085 |
9.02176871508005 |
116.627283014729 |
34.639551090512 |
| ENSMUSG00000015766 |
13860 |
Eps8 |
protein_coding |
Q08509 |
FUNCTION: Signaling adapter that controls various cellular protrusions by regulating actin cytoskeleton dynamics and architecture. Depending on its association with other signal transducers, can regulate different processes. Together with SOS1 and ABI1, forms a trimeric complex that participates in transduction of signals from Ras to Rac by activating the Rac-specific guanine nucleotide exchange factor (GEF) activity. Acts as a direct regulator of actin dynamics by binding actin filaments and has both barbed-end actin filament capping and actin bundling activities depending on the context. Displays barbed-end actin capping activity when associated with ABI1, thereby regulating actin-based motility process: capping activity is auto-inhibited and inhibition is relieved upon ABI1 interaction. Also shows actin bundling activity when associated with BAIAP2, enhancing BAIAP2-dependent membrane extensions and promoting filopodial protrusions. Involved in the regulation of processes such as axonal filopodia growth, stereocilia length, dendritic cell migration and cancer cell migration and invasion. Acts as a regulator of axonal filopodia formation in neurons: in the absence of neurotrophic factors, negatively regulates axonal filopodia formation via actin-capping activity. In contrast, it is phosphorylated in the presence of BDNF leading to inhibition of its actin-capping activity and stimulation of filopodia formation. Component of a complex with WHRN and MYO15A that localizes at stereocilia tips and is required for elongation of the stereocilia actin core. Indirectly involved in cell cycle progression; its degradation following ubiquitination being required during G2 phase to promote cell shape changes. {ECO:0000269|PubMed:10499589, ECO:0000269|PubMed:11524436, ECO:0000269|PubMed:15558031, ECO:0000269|PubMed:17115031, ECO:0000269|PubMed:19564905, ECO:0000269|PubMed:20532239, ECO:0000269|PubMed:21236676, ECO:0000269|PubMed:21835647, ECO:0000269|PubMed:8404850}. |
3D-structure;Actin-binding;Cell membrane;Cell projection;Cytoplasm;Membrane;Phosphoprotein;Reference proteome;SH3 domain;Synapse;Synaptosome;Ubl conjugation |
|
|
mmu:13860; |
brush border [GO:0005903]; cell cortex [GO:0005938]; cytosol [GO:0005829]; glutamatergic synapse [GO:0098978]; growth cone [GO:0030426]; NMDA selective glutamate receptor complex [GO:0017146]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; ruffle membrane [GO:0032587]; stereocilium [GO:0032420]; stereocilium bundle [GO:0032421]; stereocilium tip [GO:0032426]; synapse [GO:0045202]; actin binding [GO:0003779]; small GTPase binding [GO:0031267]; actin crosslink formation [GO:0051764]; actin cytoskeleton reorganization [GO:0031532]; actin filament bundle assembly [GO:0051017]; actin polymerization-dependent cell motility [GO:0070358]; adult locomotory behavior [GO:0008344]; barbed-end actin filament capping [GO:0051016]; behavioral response to ethanol [GO:0048149]; cellular response to leukemia inhibitory factor [GO:1990830]; dendritic cell migration [GO:0036336]; exit from mitosis [GO:0010458]; positive regulation of ruffle assembly [GO:1900029]; Rac protein signal transduction [GO:0016601]; regulation of actin filament length [GO:0030832]; regulation of cell shape [GO:0008360]; regulation of postsynaptic membrane neurotransmitter receptor levels [GO:0099072]; regulation of Rho protein signal transduction [GO:0035023]; Rho protein signal transduction [GO:0007266] |
12515821_Eps8 is recruited into a multimolecular signaling complex by Abi1 14699156_results highlight a critical role for p97(Eps8) in trichostatin A-exerted growth inhibition of v-Src-transformed cells 15558031_multifaceted role for this complex in actin dynamics, possibly through the participation in alternative larger complexes. 16225669_Eps8 plays multiple roles in modulating actin filament organization, possibly through its interaction with distinct sets of actin regulatory complexes 16773716_Expression of eps 8 and tgfbeta 1i4 (signal molecules of epidermal growth factor [EGF] or transforming growth factor-beta [TGF-beta]) showed upregulation. TGF-beta and EGF signal pathways are indicated in O. viverrini-driven cell proliferation. 17018287_Results report that mice lacking Eps8, a regulator of actin dynamics, are resistant to some acute intoxicating effects of ethanol and show increased ethanol consumption. 20209148_Eps8 has a role in preventing intestinal defects and improved metabolic status in mice 20418908_IRSp53, through its interaction with Eps8, not only affects cell migration but also dictates cellular growth in cancer cells. 21236676_It was concluded that MyoXVa, whirlin, and Eps8 are integral components of the stereocilia tip complex, where Eps8 is a central actin-regulatory element for elongation of the stereocilia actin core. 21357683_critical role for JNK2 and EPS8 in receptor tyrosine kinase signaling and trafficking to convey distinctly different effects on cell migration. 21526224_epidermal growth factor receptor pathway substrate 8 is critical in coordinating the development and functionality of mammalian auditory hair cells 21835647_identified Eps8 as a unique actin capping protein specifically required for Dendritic cell migration 23685357_the key role of Eps8 actin-capping activity in spine morphogenesis and plasticity and indicate that reductions in actin-capping proteins may characterize forms of intellectual disabilities associated with spine defects. 24475274_The actin-binding proteins eps8 and gelsolin have complementary roles in regulating the growth and stability of mechanosensory hair bundles of mammalian cochlear outer hair cells. 24533597_These results reveal a previously unknown cell type-specific expression pattern of endogenous Eps8 protein in the mouse hippocampus. 26252776_These findings demonstrate a novel role for Wnt-Dvl1 signalling through Eps8 in the regulation of axonal remodeling. 26668327_It shows that EPS8, a signaling adapter regulating actin dynamics, is a novel partner of VE-cadherin and is able to modulate YAP activity. 27132230_Absence of Eps8 produces a weaker phenotype in vestibular hair cells compared to cochlear inner hair cells, since it affects the hair bundle morphology but not the basolateral membrane currents. This difference is likely to explain the absence of obvious vestibular dysfunction in Eps8 knockout mice. 27450093_Results implicate N-methyl-d-aspartate receptor hyperfunction in the cognitive deficits observed in Epidermal growth factor receptor substrate 8 (Eps8) knockout mice and demonstrate a novel role for Eps8 in regulating hippocampal long-term synaptic plasticity and cognitive function. 30314980_Scrutinized the motility of Hnrnpab(-/-) cells and confirmed that the decreases in both cell motility and Eps8 are restored by ectopically coexpressing both alternatively spliced Hnrnpab isoforms, therefore these variants are surprisingly nonredundant for cell motility. 32595041_EPS8 regulates an NLRP3 inflammasome-independent caspase-1 activation pathway in monosodium urate crystal-treated RAW264.7 macrophages. 36395759_Chlamydia repurposes the actin-binding protein EPS8 to disassemble epithelial tight junctions and promote infection. |
ENSG00000151491 |
EPS8 |
630.187909 |
2.5845300782 |
1.369902 |
0.13514310 |
100.747736 |
0.00000000000000000000001044770376816143844794148371399431392023350988589195422318842447137465789808175031794235110282897949218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000005252126197908746671234157080003311779556732114543545003945831592395698805830761557444930076599121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
902.850773105268 |
84.6974363803768 |
349.328793162786 |
25.1676415848773 |
| ENSMUSG00000015950 |
17969 |
Ncf1 |
protein_coding |
Q09014 |
FUNCTION: NCF2, NCF1, and a membrane bound cytochrome b558 are required for activation of the latent NADPH oxidase (necessary for superoxide production). {ECO:0000250|UniProtKB:P14598, ECO:0000269|PubMed:9490028}. |
Cytoplasm;Lipid-binding;Membrane;Phosphoprotein;Reference proteome;Repeat;SH3 domain |
|
|
mmu:17969; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; dendrite [GO:0030425]; extrinsic component of membrane [GO:0019898]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; NADPH oxidase complex [GO:0043020]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; rough endoplasmic reticulum [GO:0005791]; phosphatidylinositol binding [GO:0035091]; phosphatidylinositol-3,4-bisphosphate binding [GO:0043325]; SH3 domain binding [GO:0017124]; superoxide-generating NAD(P)H oxidase activity [GO:0016175]; superoxide-generating NADPH oxidase activator activity [GO:0016176]; apoptotic process [GO:0006915]; cellular defense response [GO:0006968]; defense response to bacterium [GO:0042742]; defense response to fungus [GO:0050832]; defense response to Gram-positive bacterium [GO:0050830]; epithelial cell proliferation [GO:0050673]; hydrogen peroxide biosynthetic process [GO:0050665]; inflammatory response [GO:0006954]; leukocyte mediated cytotoxicity [GO:0001909]; leukotriene metabolic process [GO:0006691]; NADP catabolic process [GO:0006742]; neutrophil-mediated killing of fungus [GO:0070947]; neutrophil-mediated killing of gram-positive bacterium [GO:0070946]; protein targeting to membrane [GO:0006612]; reactive oxygen species biosynthetic process [GO:1903409]; regulation of respiratory burst involved in inflammatory response [GO:0060264]; respiratory burst [GO:0045730]; respiratory burst involved in defense response [GO:0002679]; response to bacterium [GO:0009617]; response to yeast [GO:0001878]; superoxide anion generation [GO:0042554] |
11937554_Impairment in bacterial clearance in gp47phox-deficient mice results in increased chemokine generation and lung neutrophil infiltration and demonstrates the important bactericidal role of NADPH oxidase-derived superoxide generation. 12364355_Role of p47(phox) in vascular oxidative stress and hypertension caused by angiotensin II 12560337_angiotensin II-stimulated endothelial NADPH oxidase activity is regulated through serine phosphorylation of p47(phox) and its enhanced binding to p22(phox). 12688546_Wild-type and p47(phox) knockout mice were subjected to hemorrhagic shock to test the hypothesis that NADPH oxidase plays a role in producing hepatocellular injury after hemorrhagic shock and resuscitation via NADPH oxidase-mediated oxidative stress. 12958309_over-expressing p47phox in p47-/- mice restored oscillatory shear stress -induced O2- production and monocyte adhesion 14507659_Arthritis elicited in both p47phox(-/-) and gp91(-/-) mice showed more severe joint inflammation, which developed into a granulomatous synovitis. 14734763_The NADPH oxidase-dependent respiratory burst stimulated by Pseudomonas infection contributes to host defense by modulating redox-dependent signaling through the TLR4-dependent NF-kappa B pathway. 14993144_p47phox plays a complex dual role in the vasculature. It inhibits basal NADPH oxidase activity but is critical for Ang II-induced vascular dysfunction via activation of NADPH oxidase. 15033459_PI3K regulates the phosphorylation of NADPH oxidase component p47(phox) by controlling diacylglycerol-dependent PKCs but not Akt 15123520_Mouse peritoneal macrophages & aortic segments from aldosterone-treated mice show increased superoxide anion formation. This effect was probably mediated by NADPH oxidase activation. Increased translocation of p47phox to the cell membrane was observed. 15310853_These results show that Ncf1, a gene important for oxidative burst, regulates the susceptibility and severity of both arthritis and encephalomyelitis and modulates, directly or indirectly, the level of T cell-dependent autoimmune responses. 15670768_Immunohistochemical analyses revealed a co-localization of MMP-2 with p47phox, ANG II, AT1-receptor, and alpha-SMC actin. 15936744_Summarizing relevant animal arthritis models, this review finds the Ncf1 gene to be responsible for the severity of pristane induced arthritis in mice. 16109921_shear stress induces vascular NADPH oxidase comprising p47phox but not gp91phox. 16127156_Ncf1 is not a candidate gene for regulation of Lyme arthritis and reveal Lyme arthritis to be independent of NADPH oxidase activity, distinguishing it from other models of rheumatoid arthritis. 16679392_Together, our data strongly suggest that endothelial NADPH oxidase-derived ROS play a critical role in Ang-1-induced angiogenesis. 16714332_These data show the role of NADPH oxidase-derived oxidant signaling in the mechanism of NF-kappaB activation and ICAM-1 expression in mouse lung endothelial cells. 16769910_BMP4 did not cause hypertension in p47(phox)-deficient mice.There is an essential role for p47phox-dependent NADPH oxidases in BMP4-induced endothelial dysfunction. 16864727_Deletion of p47phox attenuates Ang II-induced AAA formation in apoE-/- mice, suggesting that NADPH oxidase plays a critical role in AAA formation in this model. 17167081_Myelin phagocytosis induces the generation of reactive oxygen species via p47-PHOX, and by this mechanism a marked downregulation of microglial inflammatory responses occurs. 17220182_important role of NADPH oxidase and its subunit p47(phox) in modulating Akt and ERK1/2 activation, angiogenic growth factor expression, and angiogenesis in myocardium undergoing ischemia reperfusion 17310103_The findings indicate that hydrophobic, proapoptotic bile salts induce hepatocyte shrinkage largely through NADPH oxidase-derived oxidative stress. 17332431_These results suggest a pivotal role of NAD(P)H oxidase activation and its subunit p47(phox) for left ventricular remodeling/dysfunction and survival after myocardial infarction(MI). 17352382_The data suggest that astrocyte swelling triggers a p47(phox)-dependent NADPH oxidase-catalyzed ROS production. 17872466_Ets-1 functions as a critical downstream transcriptional mediator of Ang II reactive oxygen species generation by regulating the expression of NAD(P)H oxidase subunits such as p47(phox) 18036262_Mycobacterium tuberculosis actively induces the pro-inflammatory response in microglia through NADPH oxidase-dependent ROS generation and p47phox activation. 18070887_Phosphatidylinositol 3-kinase-dependent membrane recruitment of Rac-1 and p47phox is critical for alpha-platelet-derived growth factor receptor-induced production of reactive oxygen species. 18182394_collagen synthesis in cardiac fibroblasts involves a facilitative interaction between TGFbeta(1)-NADPH oxidase and LOX-1 18192840_Enhanced mechanical stretch in renovascular hypertension induces EPC mobilization in a p47phox-dependent manner, involving bone marrow SDF-1 and MMP-9 which may contribute to compensatory vascular adaptation in renovascular hypertension. 18206127_A2AR through MAPK signaling regulates p47phox phosphorylation and cardiac ROS production by NADPH oxidase 18425380_inhibiting NADPH oxidase activity promotes cell differentiation of B16 melanoma cells 18436804_Urokinase plasminogen activator upregulates paraoxonase 2 expression in macrophages via an NADPH oxidase-dependent mechanism. 18566430_binding of the ternary complex to phagosomes can be temporally regulated by switching between adaptor proteins that have PX domains with distinct lipid-binding specificities 18638544_acute hypoxia may distinctively activate Nox to increase [ROS](i) through the mitochondrial ROS-PKCepsilon signaling axis, providing a positive feedback mechanism to contribute to the hypoxic increase in [ROS](i) and [Ca(2+)](i) 18779779_Obesity increases neointimal formation via a mechanism involving p47(phox)-TLRs signaling, suggesting that the NADPH oxidase may represent a potential novel therapeutic target for the treatment of obesity-associated vascular inflammation and dysfunction 18787174_These results indicate that NADPH oxidase-generated reactive oxygen species specifically from bone marrow-derived cells contributes importantly to lung ischemia-reperfusion injury. 18806761_cultured p47(phox-/-) CD8(+) lymphocytes with the induction of proapoptotic Bim and Puma expression, increased mitochondrial outer membrane permeabilization and depressed Bcl-2 expression. 19052348_mutation leading to decreased oxidative burst capacity, is associated with increased susceptibility to arthritis 19103777_NADPH phagocyte oxidase appears to play a crucial role in the neutrophil-mediated host defense against A. baumannii 19124074_genetic deficiency is associated with enhanced gene expression of inflammatory mediators and increased neutrophil recruitment to lung and heart 19168729_A p47(phox) and Src kinase activation of peroxide production by Nox2 appears to be an important contributor to vascular contractile mechanisms mediated through activation of protein kinase C. 19249346_mediates phosphorylation of IKK during sepsis-induced myocardial dysfunction 19299716_an important role for Ncf1 in the fine tuning of the TLR9/MyD88 pathway 19411313_p47(phox) deletion may protect postnatal type II alveolar epithelial proliferation from hyperoxia through effects on epithelial as well as phagocyte-generated superoxide. 19439231_involved in insulin-stimulated O(2)(-) production in macrophages 19553535_a deficient ROS production, due to a spontaneous mutation in Ncf1, leads to increased autoantibody production and expansion of IL-33R-expressing T cells, impaired T cell tolerance toward tissue-specific CII, and severe arthritis 19608974_p40(phox) is prephosphorylated in resting endothelial cells and can compensate p47(phox) in keeping basal ROS production. 19833721_These results suggest that hyperoxia induces caveolin-1-dependent, c-Abl-mediated dynamin 2 phosphorylation required for recruitment of p47(phox) to caveolin-enriched microdomains and subsequent ROS production in lung endothelium. 19942291_Lipopolysaccharide-induced src kinase and PI3 kinase (PI3K) facilitates the activation of p47(phox), the regulatory subunit of NADPH oxidase. 20195241_renal expression of Nox-2, p22(phox), and p47(phox), components of NADPH oxidase, are upregulated in GSD-Ia mice compared with controls 20300512_NADPH oxidase-deficient p47(phox-/-) mice and gp91(phox)-deficient mice develop exaggerated and progressive lung inflammation, augmented NF-kappaB activation, and elevated downstream pro-inflammatory cytokines compared to wildtype mice 20370567_HNE increased membrane translocation of p47phox promoting NADPH oxidase activity in peritoneal macrophages and J774A.1 cells. 20493939_increased superoxide formation by upregulated NADPH oxidase contributes significantly to age-dependent alterations in calcium handling and contractility of murine ventricular myocytes 20690191_NOX in both BM-derived and non-BM-derived cells contributes to liver fibrosis. NOX does not play a role in experimental steatosis and the generation of ROS in hepatocytes, but exerts a key role in fibrosis. 20702812_Endothelium-dependent VEGF regulation of coronary vascular tone may require NADPH oxidase-derived ROS to activate PI3-kinase-Akt-endothelial NO synthase axis. 20817944_a differential and agonist-dependent role of the p47(phox) PX domain for neutrophil NADPH oxidase activation. 20828610_Quartz treatment of cocultures containing wild-type neutrophils, but not p47(phox-/-) neutrophils, caused increased oxidative DNA damage in epithelial cells. 20859296_NADPH oxidase has pivotal roles in medium spiny neuron injury after transient global cerebral ischemia and could be therapeutic target for brain ischemia. 20864666_Early-life exposure to high levels of PM(2.5) is a risk factor for subsequent development of insulin resistance, adiposity, and inflammation. Reactive oxygen species generation by NADPH oxidase appears to mediate this risk. 21068125_p47phox in mice plays a role in homocysteine enhancement of cell proliferation in vascular smooth muscle cells. 21253614_Ncf1-deficient Tregs suppressed Ncf1-deficient Teff very poorly compared to wild type controls. 21257963_Neutrophil NADPH oxidase activation by Aspergillus fumigatus hyphal reactive oxygen species (ROS) responses is dependent on class IA phosphoinositide 3-kinase (PI3K)beta and PI3Kdelta catalytic subunits. 21285291_In the absence of ACE2, biomechanical stress triggers activation of the myocardial NAPDH oxidase system with a critical role of the p47(phox) subunit leading to severe adverse myocardial remodelling and dysfunction. 21435178_Treatment with METH induced a time-dependent increase in phosphorylation of NOX subunit p47, followed by its binding with gp91 and p22, and the formation of an active NOX complex. 21956105_cooperation of p40(phox) with p47(phox) for Nox2-based NADPH oxidase activation during Fcgamma receptor (FcgammaR)-mediated phagocytosis 22155727_The enzyme responsible for C1P-stimulated ROS generation in bone marrow-derived macrophages is NADPH oxidase, and that this enzyme is downstream of PKC-alpha and cPLA(2)-alpha in this pathway. 22163282_NADPH oxidase in antimicrobial host defense against A. fumigatus and B. cepacia, whereas the proteases neutrophil elastase, cathepsin G, and lysosomal cysteine protease cathepsin C/ dipeptidyl peptidase I are dispensable 22184329_p47(phox) is required to enhance renal NADPH oxidase activity and basal afferent arteriolar myogenic and angiotensin II contractions. 22219181_MLCK is essential for the translocation and association of cortactin and p47phox. 22376231_Total body deficiency in p47phox subunit of NADPH oxidase complex protects mice from alcohol-induced liver steatosis 22460559_Phosphorylation of p47(phox) at different serine sites plays distinct roles in endothelial cell response to TNFalpha stimulation. 22653270_Deletion of p47 (phox) attenuates diabetes-induced glomerular injury and beta cell dysfunction in the Akita mouse. 22787054_The results of this study suggested that repeated stress promotes depressive behavior through the upregulation of NADPH oxidasesubunit (47 kD) and the resultant metabolic oxidative stres. 22914323_A critical role for p47(phox) in a dynamic interplay between PP2A and FOXO3a that regulates pro-apoptotic Bim transcription in CD8(+) memory lymphocytes during infection. 23271700_the p47phox protein variant expressed in Ncf1 mice is completely defective in activating the NOX2 complex to produce ROS 23482812_NOX2 is an important regulator of metabolic homeostasis and diet-induced obesity. 23553616_Report novel role of p47(phox) subunit as a regulator of cortactin and adaptive cytoskeletal remodeling, leading to a paradoxically enhanced susceptibility to biomechanical stress and heart failure. 23639811_It was concluded that c-Src and NADPH oxidase components are necessary for redox-mediated production of TNF-alpha following liver ischemia-reperfusion and that hepatocytes play an important role in this process. 23846495_CyPA is an important regulator for AngII-induced ROS generation in VSMC through interaction with p47phox and cell cytoskeleton, which enhances the translocation of p47phox to caveolae. 23922763_Myeloid-derived suppressor cells accumulation and immunosuppression in syngeneic epithelial ovarian cancer is NADPH oxidase-independent. 23948215_data demonstrate that, depending upon modality, deletion of p47phox and nNOS genes have dissimilar, similar or additive effects; findings provide evidence that NOX and nNOS redox signaling cascades interact in the brain to affect cognitive function and social behavior 23965970_P47phox membrane translocation in the atherosclerotic aorta is blocked by apocynin treatment thereby reducing superoxide levels and lesion size. 24358335_Mice lacking NCF1 exhibit reduced growth of implanted melanoma and carcinoma tumors. 24623132_These results identify TNFR1/Phox interaction as a key event in the pathogenesis of silicosis that prevents mtROS formation and reduces macrophage apoptosis. 24633549_Nox2 and p47(phox) modulate compensatory growth of primary collateral arteries. 24778444_I-A(b) mice that were deficient in the p47(phox) or gp91(phox) subunits of NOX2 were partially protected from MOG-induced experimental autoimmune encephalomyelitis and displayed compromised reactivation of MOG-specific CD4(+) T cells in the CNS 24970888_p47(phox) C-terminal tail plays a key role in stabilizing intramolecular interactions at rest. Ser-379 phosphorylation is a molecular switch which initiates p47(phox) conformational changes and NADPH oxidase-dependent superoxide production by cells. 25287330_p47(phox-/-) BMDM has the markedly reduced expression of miR-451 compared to WT BMDM, without other significant differences. 25358739_Data indicate that cytosol-to-membrane translocation of the Nox regulatory subunit p47(phox) and the small GTPase Rac1/2 was increased in vessels from db/db mice compared with db/+ mice, an effect blunted by atorvastatin. 25474113_NOX2/ROS activity in macrophages signals the development of antigen-specific CD8(+)T cell response. 25565313_p47(phox)-dependent NADPH oxidases are a major glomerular source of reactive oxygen species, contributing to kidney injury 25628043_p47phox plays a critical role in the regulation of adventitial fibroblast proliferation and migration and may be a new therapeutic target for neointimal hyperplasia 25922068_p47phox-Nox2-dependent physiological reactive oxygen species signaling suppresses inflammation in aging. 26021615_The Park7 binds to p47(phox), a subunit of the NADPH oxidase, to promote NADPH oxidase-dependent production of ROS. 26036345_The present study provides evidence that augmented EGFRtk impairs vascular function by NADPH oxidase-dependent mechanism. Therefore, EGFRtk and oxidative stress should be potential targets to treat vascular dysfunction in TD2. 26209274_These findings suggest that a p47(phox)-dependent NAD(P)H oxidase mediates the increase in diaphragm oxidants and contractile dysfunction in chronic heart failure. 26269022_Data (including data from studies in transgenic mice) suggest that ability to generate Ncf1-derived superoxide, specifically in CD4-positive T-lymphocytes, is key for development of autoimmune responses in type 1 diabetes. 26297224_p47phox mRNA expression was increased in diet-induced obese (DIO) mice. p47phox suppresses collateral vessel growth after femoral artery ligation in DIO mice. 26528554_data partly explains the discrepancy of the phenotypes reported earlier utilizing the Ncf1m1J mice or Ncf1 knockout mice. 26645958_The authors demonstrated a significant role of the host NADPH oxidase in promoting chronic inflammatory pathology in the oviduct following chlamydial infection. 27044504_Colitis susceptibility in p47(phox-/-) mice is mediated by the microbiome. 27829407_Mice bearing point mutation in the Ncf1-gene develop fewer osteoclasts after ovariectomy (ovx) than wild-type mice. However, irrespective of genotype, bone mineral density decreases after ovx, indicating that a compensatory mechanism retains bone degradation after ovx. 27912196_we used the Ang II infused hph-1 mice to examine the roles of NOX isoforms in the development of AAA. We generated double mutants of hph-1-NOX1, hph-1-NOX2, hph-1-p47phox, and hph-1-NOX4 28467721_Results show that Ncf1 is not needed during juvenility to protect from autoimmune arthritis later on, but conditional Ncf1 suppresses arthritis even if activated after the disease priming. The study presents in vivo evidence that Ncf1/reactive oxygen species-regulated mechanisms are different in priming than in the effector phase. 28751937_Myricitrin attenuated the generation of intracellular ROS by inhibiting the assembly of components of the gp91(phox) and p47(phox). Suppression of ROS generation using NAC or apocynin or by silencing gp91(phox) and p47(phox) all demonstrated that decreasing the level of ROS inhibited the LPS-induced inflammatory response. 28939422_p47phox, but not p67phox or p40phox, binds to and activates Nrf2, enhancing the function of Nrf2 in suppressing inflammation. 29195137_ROS production stimulated by NoxO1 and p47phox limit endothelium-dependent relaxation and maintain blood pressure in mice. However, NoxO1 and p47phox cannot substitute each other despite their similar effect on vascular function. Deletion of NoxO1 induced an anti-inflammatory phenotype, whereas p47phox deletion rather elicited a hyper-inflammatory response. 29499563_we found that ROS formation and expression of p47(phox) and p67(phox), subunits of NADPH oxidase, were increased in AppTg mice but attenuated in AppTg/Cebpd(-/-) mice. Cebpd can up-regulate p47(phox) and p67(phox) transcription via a direct binding on their promoters, which results in an increase in intracellular oxidative stress 29695496_oxLDL-mediated cellular senescence is associated with increased p47phox recruitment to caveolae and its binding to Cav1. 30076526_AT2R, via PP2A-mediated inhibition of PKC, prevents the NOX activation, reactive oxygen species generation, and subsequent pro-inflammatory activation of microglia. 31118934_Neutrophil Cytosolic Factor 1 in Dendritic Cells Promotes Autoreactive CD8(+) T Cell Activation via Cross-Presentation in Type 1 Diabetes. 31350760_The results suggest that Ncf1 is an essential regulator of innate lymphoid cells transdifferentiation, and it may mediate this process in a redox-dependent manner through IL1B production in the inflamed joint. 31442911_we propose that SRF integrates transcriptional activation of NCF1/NCF2 and ROS production to promote liver fibrosis. 32276661_Independent and inter-dependent immunoregulatory effects of NCF1 and NOS2 in experimental autoimmune encephalomyelitis. 33139577_Akt3 induces oxidative stress and DNA damage by activating the NADPH oxidase via phosphorylation of p47(phox). 33545139_Natural Loss-of-Function Mutations in Qa2 and NCF1 Cause the Spread of Mannan-Induced Psoriasis. 34085299_The atypical small GTPase GEM/Kir is a negative regulator of the NADPH oxidase and NETs production through macroautophagy. 34970267_Ncf1 Governs Immune Niches in the Lung to Mediate Pulmonary Inflammation in Mice. 35455983_Exercise Affects the Formation and Recovery of Alcoholic Liver Disease through the IL-6-p47(phox) Oxidative-Stress Axis. 35788118_The NCF1 variant p.R90H aggravates autoimmunity by facilitating the activation of plasmacytoid dendritic cells. 35963953_Two major genes associated with autoimmune arthritis, Ncf1 and Fcgr2b, additively protect mice by strengthening T cell tolerance. |
ENSG00000158517 |
NCF1 |
1387.818848 |
0.2321048432 |
-2.107151 |
0.12406830 |
273.350628 |
0.00000000000000000000000000000000000000000000000000000000000021118882701999887967862137357860797850785089381598407646942541256328118795612166252095028217237817480651100779572472944017811254285406933041881286997596724186454508753740810789167881011962890625000000000 |
0.0000000000000000000000000000000000000000000000000000000000502629408307597312217420852482864317890532507537051165629551578098631392476784950781753999727440952270157589942550054197466618865541519285848929242399574235378167941235005855560302734375000000000000000 |
Yes |
No |
518.383769150097 |
44.6347267503543 |
2233.40350028674 |
139.538931362263 |
| ENSMUSG00000015957 |
22411 |
Wnt11 |
protein_coding |
P48615 |
FUNCTION: Ligand for members of the frizzled family of seven transmembrane receptors. Probable developmental protein. May be a signaling molecule which affects the development of discrete regions of tissues. Is likely to signal over only few cell diameters. |
Developmental protein;Disulfide bond;Extracellular matrix;Glycoprotein;Lipoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:22411; |
cytoplasm [GO:0005737]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; frizzled binding [GO:0005109]; GTPase activator activity [GO:0005096]; protein kinase activator activity [GO:0030295]; signaling receptor binding [GO:0005102]; adrenal gland development [GO:0030325]; animal organ morphogenesis [GO:0009887]; apoptotic process [GO:0006915]; artery morphogenesis [GO:0048844]; atrial septum development [GO:0003283]; bicellular tight junction assembly [GO:0070830]; bone mineralization [GO:0030282]; canonical Wnt signaling pathway [GO:0060070]; cell fate commitment [GO:0045165]; cell-cell signaling [GO:0007267]; cellular response to mechanical stimulus [GO:0071260]; cloacal septation [GO:0060197]; convergent extension involved in axis elongation [GO:0060028]; embryonic skeletal system development [GO:0048706]; epithelial cell apoptotic process [GO:1904019]; epithelial to mesenchymal transition [GO:0001837]; kidney development [GO:0001822]; lung-associated mesenchyme development [GO:0060484]; maintenance of epithelial cell apical/basal polarity [GO:0045199]; mesenchymal cell proliferation [GO:0010463]; mesonephric duct development [GO:0072177]; negative regulation of apoptotic process [GO:0043066]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cell death [GO:0060548]; negative regulation of cell growth [GO:0030308]; negative regulation of cell migration [GO:0030336]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of fibroblast growth factor production [GO:0090272]; negative regulation of mesenchymal cell proliferation [GO:0072201]; negative regulation of stem cell proliferation [GO:2000647]; neuroendocrine cell differentiation [GO:0061101]; neuron differentiation [GO:0030182]; non-canonical Wnt signaling pathway [GO:0035567]; notochord morphogenesis [GO:0048570]; osteoblast differentiation [GO:0001649]; outflow tract morphogenesis [GO:0003151]; paraxial mesoderm formation [GO:0048341]; planar cell polarity pathway involved in axis elongation [GO:0003402]; planar cell polarity pathway involved in gastrula mediolateral intercalation [GO:0060775]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cell migration [GO:0030335]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of epithelial cell apoptotic process [GO:1904037]; positive regulation of gene expression [GO:0010628]; positive regulation of GTPase activity [GO:0043547]; positive regulation of heart induction by negative regulation of canonical Wnt signaling pathway [GO:0090082]; positive regulation of protein kinase C signaling [GO:0090037]; positive regulation of stress fiber assembly [GO:0051496]; positive regulation of transforming growth factor beta2 production [GO:0032915]; primary heart field specification [GO:0003138]; protein localization to cell surface [GO:0034394]; protein phosphorylation [GO:0006468]; response to nutrient levels [GO:0031667]; roof of mouth development [GO:0060021]; secondary heart field specification [GO:0003139]; secondary palate development [GO:0062009]; signal transduction [GO:0007165]; somite development [GO:0061053]; stem cell proliferation [GO:0072089]; ureteric bud morphogenesis [GO:0060675]; ventricular septum development [GO:0003281]; ventricular septum morphogenesis [GO:0060412] |
12783789_Wnt11 and Ret/Gdnf cooperate in coordinating ureteric branching by maintaining a balance of Wnt11-expressing ureteric epithelium and Gdnf-expressing mesenchyme in developing kidney 15067007_Wnt11-mediated suppression of canonical signaling exists in vivo 15201220_epithelial Wnt11, mesenchymal Gdnf and stromal Fgf7 signalling is coordinated by sprouty proteins during kidney development, which regulates ureteric branching 15541384_We propose that Wnt11 plays an important role for cardiac development by embryoid bodies, and may be a key regulator of cardiac muscle cell proliferation and differentiation during heart development. 16426441_Expression levels of the beta-catenin-independent Wnts, Wnt-4, -5a and -11 were up-regulated transiently during cutaneous wound healing. 17154359_Within fetal liver kinase (Flk)1-positive cells, Wnt11 may play a critical role in Flk1-positive cell fate determination, at least partially by modulating canonical Wnt/beta-catenin signalling. 17522159_initiation of metanephric kidney development requires the reduction of BMP4 activity by the antagonist gremlin 1 in the mesenchyme, which in turn enables ureteric bud outgrowth and establishment of autoregulatory GDNF/WNT11 feedback signalling 17767158_We have found that Wnt11 is a direct target of a canonical beta-catenin pathway in developing heart and that Wnt11 mutants show cardiac outflow tract defects.Wnt signaling requires ATF/CREB for signalling. 18191119_Negative feedback of fibroblast factors triggers excessive cell proliferation then inhibits the expression of Fgfr1b and activates the expression of Wnt11 to fuse each palate. 18650792_AT1R-mediated inhibition of the Spry1 gene increases c-Ret tyrosine kinase activity leading to upregulation of its downstream target Wnt11, which induces GDNF in adjacent mesenchyme causing focal bursts of ureteric bud tip cell proliferation 19213727_Wnt11 signals through beta-catenin, activating Rspo2 expression, which is then required for Wnt11-mediated osteoblast maturation. 19622544_Wnt-11 signalling serves as a critical cell adhesion cue for the organization of the cardiomyocytes in the developing ventricular wall, which is essential for the establishment of a functional heart. 19805103_Data show that the higher expression of WNT5a in smaller EBs enhanced endothelial cell differentiation, and in contrast, the increased expression of WNT11 enhanced cardiogenesis. 19809516_GLI3 repressor controls nephron number by regulating ureteric tip cell expression of Wnt11 and Ret 21041481_noncanonical Wnt (Wnt11) enhanced cardiomyocyte differentiation while preventing stabilization of the beta-catenin protein, suggesting active repression of canonical Wnt signals 21463175_Transplantation of MSC(Wnt11) improved cardiac function. The release of Wnt11 and other factors from transplanted MSC(Wnt11) is more likely responsible for protection of native CM at risk. 21903761_Wnt11 is involved in the protection of the host intestinal cells by blocking the invasion of pathogenic bacteria, suppressing inflammation, and inhibiting apoptosis. 22556418_all the TGF-beta, Wnt11, and JNK targets were activated in a unilateral ureteral obstruction (UUO) model of renal fibrosis in vivo. 22715413_Notch1-induced WISP-1 expression appeared to be Wnt11-dependent, but Wnt1-independent 22829700_demonstrates that the combination of Wnt11 and BMP-2 effectively promotes cardiomyogenic differentiation of BM-MSCs in vitro. The synergistic effect of Wnt11 and BMP-2 on the cardiomyogenic differentiation of BM-MSCs is further enhanced in myocardium 23613470_The apical and basolateral secretion of Wnt11 and Wnt3a in polarized epithelial cells is regulated by different mechanisms. 25312921_Studied groups of embryoid bodies (EBs) with different starting numbers of ESCs & found differential gene expression patterns for Wnt5a & Wnt11. Wnt11 inc'd the percentage of beating EBs by upregulating expression of cardiac-specific genes. 25448697_These results provide formal genetic proof that the majority of the endocardium and myocardium diverge by mid-gastrulation in the mouse, and suggest a tight spatial and temporal control of Wnt11 expression in the myocardial lineage. 25482987_Wnt5a/Wnt11 inhibit beta-catenin to promote SHF development through Caspase-dependent Akt degradation 25713299_a mechanistic link between E-cadherin loss and subsequent control of Rho-driven anoikis resistance through p120- and Kaiso-dependent expression of Wnt11, is reported. 25813538_Data show that Wnt5a and Wnt11 are required for proper patterning of the neural tube and somites by regulating notochord formation. 25959411_under tensile stress, miR-154-5p negatively regulates ADSCs osteogenic differentiation through the Wnt/PCP pathway by directly targeting Wnt11 27582005_Results provide evidence that Wnt11 is involved in the organization of kidney tubules through the planar cell polarity pathway taking part in fine-tuning of nephrogenesis. 28348167_Wnt4 and Wnt11 cooperatively contribute to mammalian neuromuscular junction formation. 28669819_These studies revealed a previously unappreciated role for WNT11 for dorsal mesenchymal protrusion (DMP) formation and distinct tissue-specific requirements for WNT11 in outflow tract and DMP development. 30516471_Wnt11 directs nephron progenitor polarity and motile behavior ultimately determining nephron endowment. 30651543_Study utilizing mouse kidney cultures and time-lapse imaging provided quantitative experimental support that a ligand-receptor-based Turing mechanism, implemented via GDNF-RET signaling, specifies the areas of outgrowth during kidney branching morphogenesis. The positive feedback between GDNF and WNT11 enables the dense packing of the ureteric buds. 33082334_WNT11-FZD7-DAAM1 signalling supports tumour initiating abilities and melanoma amoeboid invasion. 36098469_Wnt11 in regulation of physiological and pathological cardiac growth. |
ENSG00000085741 |
WNT11 |
16.532695 |
3.7300530120 |
1.899196 |
0.67674890 |
7.444484 |
0.00636311444917684893712861438075378828216344118118286132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0227839584602401896973322692474539508111774921417236328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.4857216299073 |
12.3982939115669 |
6.83253603853614 |
2.58917570471344 |
| ENSMUSG00000016024 |
16803 |
Lbp |
protein_coding |
Q61805 |
FUNCTION: Plays a role in the innate immune response. Binds to the lipid A moiety of bacterial lipopolysaccharides (LPS), a glycolipid present in the outer membrane of all Gram-negative bacteria (PubMed:9144073). Acts as an affinity enhancer for CD14, facilitating its association with LPS (By similarity). Promotes the release of cytokines in response to bacterial lipopolysaccharide (PubMed:24380872). {ECO:0000250|UniProtKB:P18428, ECO:0000269|PubMed:24380872, ECO:0000269|PubMed:9144073}. |
3D-structure;Antibiotic;Antimicrobial;Disulfide bond;Glycoprotein;Immunity;Innate immunity;Lipid transport;Membrane;Reference proteome;Secreted;Signal;Transport |
|
|
mmu:16803; |
cell surface [GO:0009986]; extracellular space [GO:0005615]; membrane [GO:0016020]; coreceptor activity [GO:0015026]; lipopeptide binding [GO:0071723]; lipopolysaccharide binding [GO:0001530]; lipoteichoic acid binding [GO:0070891]; signaling receptor binding [GO:0005102]; acute-phase response [GO:0006953]; cell surface pattern recognition receptor signaling pathway [GO:0002752]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to lipoteichoic acid [GO:0071223]; defense response to Gram-negative bacterium [GO:0050829]; defense response to Gram-positive bacterium [GO:0050830]; detection of molecule of bacterial origin [GO:0032490]; innate immune response [GO:0045087]; leukocyte chemotaxis involved in inflammatory response [GO:0002232]; lipopolysaccharide transport [GO:0015920]; lipopolysaccharide-mediated signaling pathway [GO:0031663]; macromolecule localization [GO:0033036]; macrophage activation involved in immune response [GO:0002281]; negative regulation of tumor necrosis factor production [GO:0032720]; neutrophil chemotaxis [GO:0030593]; opsonization [GO:0008228]; positive regulation of chemokine production [GO:0032722]; positive regulation of cytolysis [GO:0045919]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of interleukin-8 production [GO:0032757]; positive regulation of macrophage activation [GO:0043032]; positive regulation of neutrophil chemotaxis [GO:0090023]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of respiratory burst involved in inflammatory response [GO:0060265]; positive regulation of toll-like receptor 4 signaling pathway [GO:0034145]; positive regulation of tumor necrosis factor production [GO:0032760]; regulation of membrane permeability [GO:0090559]; response to lipopolysaccharide [GO:0032496] |
11466385_Innate defense against lethal peritoneal infection with Salmonella requires the direct proinflammatory involvement of LPS-binding protein (LBP), a major nonredundant function of LBP in this model. 11884468_LBP plays an important role in early alcohol-induced liver injury by enhancing LPS-induced signal transduction, most likely in Kupffer cells. 12055258_LBP plays an important role in resistance to lethal Salmonella peritonitis (but not to oral or intravenous infection) by facilitating the role of neutrophils in mediating acute inflammation. 12353926_role LBP plays in local pulmonary immune defenses to bacterial challenge 12370383_In the case of intraperitoneal infection of LBP-deficient mice with Salmonella typhimurium, no local inflammatory response is evident, neutrophil influx is delayed, and the mice succumb to the infection. 12932360_plays an essential role in the innate immune response to Gram-positive pneumococci 14638760_Locally produced LBP is an essential component of an effective innate immune response to E. coli peritonitis. 15356561_LBP is one of the critical molecules regulating the acute and chronic airway response to inhaled lipopolysaccharides (LPS). 15619225_LBP knockout mice are protected from toxicity with a decrease in hepatic necrosis following acetaminophen challenge. This suggests a novel role for LBP in modulating acetaminophen-induced liver injury. 15993364_LBP does not contribute to an effective host response in Mycobacterium tuberculosis infection in mice. 16163057_LBP per se possesses a protective effect on acute necrotizing pancreatitis 16614372_In conclusion, hemorrhagic shock and resuscitation to mice cause severe, LBP-mediated hepatocellular damage. 18559343_Oxidized phospholipid inhibition of toll-like receptor (TLR) signaling is restricted to TLR2 and TLR4: roles for CD14, LPS-binding protein, and MD2 as targets for specificity of inhibition. 18948440_LPS-binding protein mediates LPS-induced liver injury and mortality in the setting of biliary obstruction. 19022463_Marked concomitant increases in plasma LBP and endotoxin-like activity following murine trypanosome infection might play an important role in the pathogenesis of trypanosomosis 19234712_Paneth cells express LBP together with other proteins acting in the innate immune response of the gut, such as lysozyme, defensins and intelectin. 20833181_The amino acid sequence for two mimic epitopes of the inflammatory site of LBP were determined to be WKAQKRFMKKSG and LKTRKLFWKYKD. 21993208_findings describe a novel role for LBP in normal hippocampal development and raise the possibility that some of the behavioral sequelae of early life stress are mediated by reduced expression of LBP during a critical period of neurodevelopment. 24664302_Differential expression of LBP and TGFB1, along with other genes, in different mesenchymal stromal cells preparations, produces the variable responses to external stimuli. 24671012_The LBP gene is a macrophage-specific LXR target that promotes foam cell survival and atherogenesis. 26201685_LBP is a proinflammatory soluble adipokine that acts as a brake for adipogenesis, strengthening the negative effects of palmitate and LPS on adipocyte differentiation. 27404046_LBP is a critical factor in the development of non-alcoholic fatty liver disease. 36410673_Liver lipopolysaccharide binding protein prevents hepatic inflammation in physiological and pathological non-obesogenic conditions. |
ENSG00000129988 |
LBP |
98.982267 |
2.3507816955 |
1.233141 |
0.33479453 |
12.807471 |
0.00034523788045368036933244204611526129156118258833885192871093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0018027642766782511589418458441969050909392535686492919921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
128.418743016913 |
30.0692953335052 |
54.6281023298819 |
9.74002405560297 |
| ENSMUSG00000016179 |
215303 |
Camk1g |
protein_coding |
Q91VB2 |
FUNCTION: Calcium/calmodulin-dependent protein kinase belonging to a proposed calcium-triggered signaling cascade. In vitro phosphorylates transcription factor CREB1. |
Allosteric enzyme;ATP-binding;Calmodulin-binding;Cell membrane;Cytoplasm;Golgi apparatus;Kinase;Lipoprotein;Membrane;Nucleotide-binding;Prenylation;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:215303; |
calcium- and calmodulin-dependent protein kinase complex [GO:0005954]; cytoplasm [GO:0005737]; endomembrane system [GO:0012505]; Golgi membrane [GO:0000139]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; ATP binding [GO:0005524]; calmodulin binding [GO:0005516]; calmodulin-dependent protein kinase activity [GO:0004683]; protein serine kinase activity [GO:0106310]; peptidyl-serine phosphorylation [GO:0018105]; protein phosphorylation [GO:0006468] |
12637513_CLICK-III/CaMKIgamma is a novel membrane-anchored neuronal Ca2+/calmodulin-dependent protein kinase (CaMK). 17553424_CL3 may represent a key element in the Ca(2+)-dependent and lipid-raft-delineated switch that turns on extrinsic activity-regulated dendrite formation in developing cortical neurons. 20729199_Calmodulin suppresses synaptotagmin-2 transcription in cortical neurons |
ENSG00000008118 |
CAMK1G |
40.214498 |
0.2994681737 |
-1.739525 |
0.45690191 |
14.310670 |
0.00015498391252436290654523332044334438251098617911338806152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008848256954914152181551623144173390755895525217056274414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.00753608627 |
5.79880490161652 |
60.1317190440795 |
13.5525739475818 |
| ENSMUSG00000016194 |
15483 |
Hsd11b1 |
protein_coding |
P50172 |
FUNCTION: Controls the reversible conversion of biologically active glucocorticoids such as 11-dehydrocorticosterone to corticosterone in the presence of NADP(H) (PubMed:9405715, PubMed:23415904, PubMed:30902677). Participates in the corticosteroid receptor-mediated anti-inflammatory response, as well as metabolic and homeostatic processes (PubMed:9405715). Bidirectional in vitro, predominantly functions as a reductase in vivo, thereby increasing the concentration of active glucocorticoids (PubMed:23415904). It has broad substrate specificity, besides glucocorticoids, it accepts other steroid and sterol substrates (PubMed:23415904). Interconverts 7-oxo- and 7-hydroxy-neurosteroids such as 7-oxopregnenolone and 7beta-hydroxypregnenolone, 7-oxodehydroepiandrosterone (3beta-hydroxy-5-androstene-7,17-dione) and 7beta-hydroxydehydroepiandrosterone (3beta,7beta-dihydroxyandrost-5-en-17-one), among others (By similarity). Catalyzes the stereo-specific conversion of the major dietary oxysterol, 7-ketocholesterol (7-oxocholesterol), into the more polar 7-beta-hydroxycholesterol metabolite (PubMed:23415904). 7-oxocholesterol is one of the most important oxysterols, it participates in several events such as induction of apoptosis, accumulation in atherosclerotic lesions, lipid peroxidation, and induction of foam cell formation (By similarity). Mediates the 7-oxo reduction of 7-oxolithocholate mainly to chenodeoxycholate, and to a lesser extent to ursodeoxycholate, both in its free form and when conjugated to glycine or taurine, providing a link between glucocorticoid activation and bile acid metabolism (By similarity). Catalyzes the synthesis of 7-beta-25-dihydroxycholesterol from 7-oxo-25-hydroxycholesterol in vitro, which acts as ligand for the G-protein-coupled receptor (GPCR) Epstein-Barr virus-induced gene 2 (EBI2) and may thereby regulate immune cell migration (PubMed:30902677). {ECO:0000250|UniProtKB:P28845, ECO:0000269|PubMed:23415904, ECO:0000269|PubMed:30902677, ECO:0000269|PubMed:9405715, ECO:0000303|PubMed:23415904, ECO:0000303|PubMed:9405715}. |
3D-structure;Endoplasmic reticulum;Glycoprotein;Lipid metabolism;Membrane;NADP;Oxidoreductase;Reference proteome;Signal-anchor;Steroid metabolism;Transmembrane;Transmembrane helix |
PATHWAY: Steroid metabolism. {ECO:0000305}. |
|
mmu:15483; |
apical part of cell [GO:0045177]; cytoplasm [GO:0005737]; endoplasmic reticulum membrane [GO:0005789]; intracellular membrane-bounded organelle [GO:0043231]; nuclear membrane [GO:0031965]; rough endoplasmic reticulum [GO:0005791]; 11-beta-hydroxysteroid dehydrogenase (NADP+) activity [GO:0070524]; 7-beta-hydroxysteroid dehydrogenase (NADP+) activity [GO:0047022]; cortisol dehydrogenase activity [GO:0102196]; NADP binding [GO:0050661]; oxidoreductase activity [GO:0016491]; protein homodimerization activity [GO:0042803]; steroid binding [GO:0005496]; glucocorticoid biosynthetic process [GO:0006704]; glucocorticoid catabolic process [GO:0006713]; lung development [GO:0030324]; mineralocorticoid metabolic process [GO:0008212]; regulation of pentose-phosphate shunt [GO:0043456]; steroid catabolic process [GO:0006706] |
11841241_Human liver 11 beta-HSD 1 exhibits Michaelis-Menten kinetics with cortisol and corticosterone (11 beta-dehydrogenation activity) but cooperative kinetics with cortisone and dehydrocorticosterone (11-oxoreducing activity). 12145154_Liver X receptors downregulate expression and activity 12436336_Selective inhibition of this enzyme decreases blood glucose concentrations in hyperglycemic mice. 12444044_study demonstrates for the first time remarkable spatial and temporal patterns of expression of 11beta-hydroxysteroid dehydrogenase 1 and 11beta-hydroxysteroid dehydrogenase 2 and glucocorticoid receptor in the murine placenta and uterus 14652720_Possible role of 11beta-HSD1 for development of the metabolic syndrome, raising the possibility of novel therapeutic options for the treatment of Type 2 diabetes by inhibition or down-regulation of 11beta-HSD1 activity. (review) 14715714_11betaHSD1 amplification of intracellular glucocorticoid actions in mice may be required for bone marrow adipocyte formation, but not for bone formation. 15044372_11beta-HSD-1 activity highest in subcutaneous fat, followed by epididymal fat, with lowest activity in mesenteric viscera. 15047607_data provide the first in vivo evidence that adipose 11 beta-HSD-1 deficiency beneficially alters adipose tissue distribution and function, complementing the reported effects of hepatic 11 beta-HSD-1 deficiency or inhibition 15095019_Data report on the in vitro oxysterol-metabolizing properties of human and rodent 11beta-hydroxysteroid dehydrogenase. 15145446_study demonstrates that tissue- and developmentally-stage specific expression of 11beta-HSD1 and 11beta-HSD2 as well as GR occurs in the developing mouse embryo 15277385_Cortisol loses a deuterium during conversion to cortisone, which in turn generates cortisol via 11beta-HSD type 1. 15586384_Increased levels of 11beta-HSD1 are associated with impaired glucose-stimulated insulin secretion in beta cells. 15616008_Activation of expression within the liver may contribute to the development of type 2 diabetes in db/db mice. 15634910_Presence of a functional hydroxysteroid dehydrogenase type I provides lymphocytes with a novel intracrine regulatory mechanism that could influence such processes as lymphocyte development, effector function, and susceptibility to apoptosis. 16093320_Preventing local regeneration of glucocorticoids by 11beta-hydroxysteroid dehydrogenase type 1 enhances angiogenesis. 16356929_In hexose-6-phosphate dehydrogenase knockout mice, the reaction direction of 11 beta-hydroxysteroid dehydrogenase type 1 is altered. 16612591_11beta-hydroxysteroid dehydrogenase type-1 inhibitor BVT.2733 lowers blood glucose and insulin in mutant mouse models of obesity and diabetes. 16621520_Adult thymic expression of 11beta-HSD1 mRNA and protein amounts to approximately 6-7% of those observed in liver, and the enzyme is expressed in the medullary thymocytes and thymocytes present at the corticomedullary junction. 16763061_data suggest that 11 beta-Hydroxysteroid dehydrogenase type 1 (11 beta-HSD1) in brain plays a role in the adaptive restraint of excess fat intake 17008390_increased generation of glucocorticoids by 11betaHSD1 in the murine vessel wall is unlikely to contribute to feedback regulation of inflammation 17311922_temporal regulation of 11beta-HSD1 expression is tightly controlled in 3T3-L1 cells, so as to mediate the initiation of differentiation by inactive glucocorticoids 17588937_cellular trafficking of G6P directly regulates 11beta-HSD1 reductase activity and provides a novel link between glucose metabolism and function of the hypothalamo-pituitary-adrenal axis. 17640483_11 beta-HSD1 may be an important candidate mediator of obesity and obesity-induced insulin resistance. 17702848_Ceramide- and AMPK-mediated signaling pathways augment the expression and activity of 11beta-HSD1 in preadipocytes by way of C/EBPbeta. 17912154_Transgenic overexpression of 11beta-HSD1 in either adipose tissue or the liver in mice causes components of the metabolic syndrome, while transgenic deletion of 11beta-HSD1 prevents adverse metabolic complications of obesity. Review. 17947710_TGF-beta protein expression was increased in unstimulated CD11b(-) cells residing in the BM and spleen of 11betaHSD1(-/-) mice 18073273_results indicate that 11beta-HSD1 plays a significant functional role in the initiation of 3T3-L1 adipogenesis and provide new mechanistic insights into the role of 11beta-HSD1 in the development of obesity and related diseases 18174284_11beta-HSD1 reductase activity is augmented in mouse mesenteric preadipocytes where it promotes preadipocyte differentiation and contributes to visceral fat accumulation in obesity. 18467433_Insulin and dexamethasone augmented 11beta-HSD1 activity and expression in rat white adipose tissue in vivo. 18573267_Variable expression of HSD11B1 in vascular endothelial cells is reported. 18755798_Data show that the activity and reaction direction of adipose 11beta-HSD1 is altered under conditions of oxysterol excess, and could impact upon the pathophysiology of obesity and its complications. 18778749_A NADPH/NADP(+) ratio of ten or higher was required for efficient microsomal 11beta-HSD1 reductase activity. 18779947_Local regeneration of glucocorticoid via 11beta-HSD1 within alpha cells regulates glucagon secretion and may act in a paracrine manner to limit insulin secretion from beta cells. 19010388_The impact of H6PDH on the modulation of 11beta-HSD1-dependent interconversion of cortisone and cortisol by inhibitors and alternative substrates, was investigated. 19675138_Selective 11beta-HSD1 inhibition decreases lipogenic and lipolytic gene expression that may represent an important mechanism underpinning their insulin-sensitizing action. 20045394_IGF-I tissue-specifically regulates Hsd11b1 and Hsd11b2 expression. 20495186_Inflammatory cell recruitment after myocardial infarction is modified in mice deficient in 11bHSD1 and this provides an enhanced stimulus for angiogenesis in the healing infarcts of these mice. 20544861_We propose that 11 beta HSD-1 may provide microglia with an intrinsic mechanism to autoregulate and inhibit proinflammatory mediator production through CORT formation. 21106871_These data are consistent with an 11beta-HSD1-independent function for H6PDH in which sarcoplasmic reticulum G6P metabolism and NAD/NADH redox status are important for maintaining muscle homeostasis. 21198436_Demonstrated that remarkable pharmacokinetic differences existed between high-fat diet-induced obese and lean mice for HSD11B1 inhibitors. 21215738_tissue- and differentiation state-specific promoter usage in expression of human HSD11B 21304964_11beta-HSD1 controls glucocorticoid-induced protein degradation in human and murine skeletal muscle via regulation of the E3 ubiquitin ligases Atrogin-1 and MuRF-1. 21411659_11beta-HSD1 deficiency in aging mice leads to lower intracellular GC concentrations in brain, particularly in the hippocampus, which activate predominantly MRs to enhance memory 22033707_High-fat diet accelerated aging in the liver of SAMP10 mice by increasing liver mRNA level for Hsd11b1, increasing insulin secretion, and promoting lipid accumulation in the liver. 22067318_11beta-HSD1-deficient mice showed greater numbers of inflammatory cells in pleural lavages in carrageenan-induced pleurisy with lung pathology consistent with slower resolution, suggesting that 11beta-HSD1 limits acute inflammation. 22158867_adipose tissues of 11betaHSD1(-/-) mice exhibit attenuated hypoxia, induction of hypoxia-inducible factor (HIF-1alpha) activation of the TGF-beta/Smad3/alpha-smooth muscle actin (alpha-SMA) signaling pathway, and fibrogenesis 22209663_knockdown of 11beta-HSD1 can protect against several western type diet-induced metabolic abnormalities. These effects are, at least in part, mediated by increases in the oxidative capacity of brown adipose tissue. 22315313_Optimal elevation of beta-cell 11beta-HSD1 represents a novel biological mechanism supporting compensatory insulin hypersecretion rather than exacerbating metabolic disease. 22357964_Reduced intra-adipose glucocorticoid regeneration by 11beta-HSD1 is a novel mechanism that contributes to the metabolic efficacy of salicylates. 22447855_These studies delineate a novel pathway that links hormonal and circadian signals to gluconeogenesis and glucose homeostasis. 22555437_mice with liver-specific disruption of 11beta-hydroxysteroid dehydrogenase type 1 lack significant metabolic abnormalities 22692684_Ifi202b modulates fat accumulation through expression of adipogenic genes such as 11beta-Hsd1. 22768329_11beta-HSD1 may be a very promising therapeutic target for obesity and associated disease. 22940536_11beta-HSD1 can inter-convert 7-KC and 7betaOHC in the arterial wall 23170035_11beta-HSD1 inhibition reduces atherosclerosis in mice by altering proinflammatory gene expression in the vasculature. 23197361_Our results indicate that 11beta-HSD1 acts as a vital regulator that controls the expression of genes related to brown fat function and as such may become a potential target in preventing obesity 23267038_Specific reduction of G6PT may contribute to downregulation of hepatic 11beta-HSD1 in diabetic mice. 23349944_These findings suggest that 11b-HSD1-generated glucocorticoids may tonically restrain mast cell degranulation, potentially influencing allergic, anaphylactic and inflammatory responses. 23362866_11beta-HSD1 expression was decreased in TPA-treated hyperproliferative skin. 23383297_11beta-hydroxysteroid dehydrogenase type 1 gene knockout attenuates atherosclerosis and in vivo foam cell formation in hyperlipidemic apoE knockout mice. 23512810_11beta-HSD1 expression is down-regulated during adipose tissue expansion in diet-induced obese mice. 23933573_Oxidoreduction of 7-oxolithocholic acid and its conjugated metabolites are catalyzed by 11beta-HSD1. 24169553_These data challenge the concept that inhibition of 11beta-HSD1 is likely to produce a 'step-change' treatment for diabetes and/or obesity. 24413279_TNFalpha-mediated Hsd11b1 binding of NF-kappaB p65 is associated with suppression of 11beta-HSD1 in muscle cells. 24511146_The possibility of a vicious circle of glucocorticoids signals via 11beta-HSD1 up-regulation in adipose tissues, contributing to deterioration of glucose metabolism to result in diabetes. 24569863_Elevated hepatic 11betaHSD1 is an important contributor to early insulin resistance and dyslipidemia in uremia. 24759003_Data suggest that Gh (growth hormone) represses H6pd (hexose-6-phosphate dehydrogenase) through locally produced Igf1 (insulin-like growth factor 1); Gh directly represses Hsd11b1 mRNA rather than acting via Igf1 receptor. 24889609_11beta-HSD1, particularly in adipose tissue, is key to the development of the adverse metabolic profile associated with circulating glucocorticoid excess 25109766_Dynamic increases in hippocampal 11b-HSD1 regenerated CORT levels during learning and retrieval play a key role in age- and stress-associated impairments of spatial memory. 25389364_Elevated adipose 11beta-HSD1 expression may contribute to glucocorticoid-induced insulin resistance and adipolysis. 25614240_Data indicate that 11beta-HSD1 activity contributes substantially to diurnal and stress-induced increases in hippocampal CORT levels. 25708481_elevated in livers of offspring of restraint-stressed mothers 25869616_Data suggest that Hsd11b1 expression is up-regulated in white adipose tissues of obese mice; this induction of Hsd11b1 appears to reduce mitochondrial respiration and adiponectin synthesis in hypertrophic adipocytes. 26268211_Nr3c2 has a role in mouse skin development in a process that involves HSD11B1/HSD11B2 26305481_Our results demonstrate an inhibitory effect of IGF-I on 11beta-HSD1 expression and activity within the pancreatic islets, which may mediate part of the IGF-I effects on cell proliferation, survival and insulin secretion 26465199_Data (including data from knockout mice studies) suggest expression of 11bHSD1 (in cardiomyocytes, vascular smooth muscle, other cells) promotes infarct expansion/ventricular remodeling after myocardial infarction & limits angiogenesis/infarct repair. 26563879_These findings suggest that 11beta-HSD1 may contribute to the decline in Npas4 and Arc mRNA levels associated with memory impairment during ageing 26700460_Loss of 11beta-HSD1 is associated with hepatocellular carcinoma. 27070821_these data suggest that corticosterone activation by 11beta-HSD1 in keratinocytes suppresses hapten-induced irritant dermatitis through suppression of expression of cytokines 27384305_11beta-HSD1-deficient mice are not protected from metabolic disease or hepatosteatosis in the face of a nonalcoholic fatty liver disease-inducing diet. 27578244_11beta-Hydroxysteroid dehydrogenase type 1 within muscle protects against the adverse effects of local inflammation. 27841334_study demonstrates that glucocorticoid-induced insulin resistance was dependent on 11beta-HSD1, resulting in the critical activation of JNK signaling in adipocytes. 27859809_findings indicate that 11b-HSD1 regenerated glucocorticoids in the forebrain and decreased levels of Brain Derived Neurotrophic Factor in the hippocampus play a role in spatial memory deficits in aged wild-type mice. 28237720_The present study shows the temporal localisation of 11beta-HSD1 in uterus, highlighting its importance in the timing of gestation and suggesting its contribution in the myometrium contraction. 28254522_11beta-HSD1 protein expression after caloric restriction was significantly up-regulated, while no difference was detected after re-feeding. Interestingly, upregulation of protein after CR (1.4-fold) was lower than the increase in enzymatic activity (2.6-fold). 28368470_Brown adipose tissue (BAT) from aged 11beta-HSD1 KO mice showed elevated UCP1 protein and mitochondrial content, and a favorable profile of BAT function. 28522730_These data identify suppression of CXCL2 and CXCL5 chemoattractant expression by 11beta-HSD1 as a novel mechanism with potential for regulation of neutrophil recruitment to the injured myocardium, and cardiac fibroblasts as a key site for intracellular glucocorticoid regeneration during acute inflammation following myocardial injury. 28663379_exercise training increases pulmonary expression of 11beta-HSD1, thus contributing to local glucocorticoid activation and suppression of pulmonary inflammation in obese mice 28676523_11beta-HSD1 deficiency in myeloid phagocytes promotes angiogenesis. 28749080_We envisage these data and models finding utility when investigating the muscle-specific functions of the 11beta-HSD1/G6PT/H6PDH triad. 29062028_These data suggest that 11beta-HSD1 is not crucial for survival of experimental malaria, but alters its progression in a parasite strain-specific manner. 29891135_tis study shows that 11 beta-hydroxysteroid dehydrogenase type 1 regulates synovitis, joint destruction, and systemic bone loss in chronic polyarthritis 30902677_The formation of 7-keto,25-hydroxycholesterol from 7-ketocholesterol by cholesterol 25-hydroxylase (CH25H) and further stereospecific oxoreduction to 7beta,25-dihydroxycholesterol by human and mouse 11-beta-HSD1 could be demonstrated. 34072239_Increased Expression of 11beta-Hydroxysteroid Dehydrogenase Type 1 Contributes to Epidermal Permeability Barrier Dysfunction in Aged Skin. 34490902_Transient expansion of the expression region of Hsd11b1, encoding 11beta-hydroxysteroid dehydrogenase type 1, in the developing mouse neocortex. 35637957_The GR-gp78 Pathway is involved in Hepatic Lipid Accumulation Induced by Overexpression of 11beta-HSD1. 36205523_11beta-HSD1 contributes to age-related metabolic decline in male mice. |
ENSG00000117594 |
HSD11B1 |
56.677949 |
0.4812025038 |
-1.055284 |
0.34022219 |
9.590741 |
0.00195561031029950352713719929909075290197506546974182128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0083462752161619600810293917447779676876962184906005859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
35.990706537771 |
8.56636229279083 |
74.7932653089836 |
12.7931113428982 |
| ENSMUSG00000016346 |
16536 |
Kcnq2 |
protein_coding |
B7ZBV3 |
Human_homologues FUNCTION: Associates with KCNQ3 to form a potassium channel with essentially identical properties to the channel underlying the native M-current, a slowly activating and deactivating potassium conductance which plays a critical role in determining the subthreshold electrical excitability of neurons as well as the responsiveness to synaptic inputs. Therefore, it is important in the regulation of neuronal excitability. KCNQ2/KCNQ3 current is blocked by linopirdine and XE991, and activated by the anticonvulsant retigabine (PubMed:9836639, PubMed:11572947, PubMed:14534157, PubMed:12742592, PubMed:17872363). As the native M-channel, the potassium channel composed of KCNQ2 and KCNQ3 is also suppressed by activation of the muscarinic acetylcholine receptor CHRM1 (PubMed:10684873). KCNQ2-KCNQ3 channel is selectively permeable to other cations besides potassium, in decreasing order of affinity K(+) > Rb(+) > Cs(+) > Na(+). Associates with Na(+)-coupled myo-inositol symporter SLC5A3 forming a coregulatory complex that alters ion selectivity, increasing Na(+) and Cs(+) permeation relative to K(+) permeation. {ECO:0000269|PubMed:10684873, ECO:0000269|PubMed:11572947, ECO:0000269|PubMed:12742592, ECO:0000269|PubMed:14534157, ECO:0000269|PubMed:17872363, ECO:0000269|PubMed:25740509, ECO:0000269|PubMed:28793216, ECO:0000269|PubMed:9836639}. |
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
Human_homologues hsa:3785; |
voltage-gated potassium channel complex [GO:0008076]; voltage-gated potassium channel activity [GO:0005249]; regulation of ion transmembrane transport [GO:0034765] |
15608631_Suppression of neuronal KCNQ2 current in mice is associated with spontaneous seizures, behavioral hyperactivity and morphological changes in hippocampus. 16481438_Genomic deletions affecting the C terminus of KCNQ2 have been identified in human families with benign familial neonatal convulsions. 16500630_In the adult mouse brain, a predominantly axonal staining pattern was found, most observed in the caudoputamen, the alveus and the mossy fiber pathway of the hippocampus. The hippocampal staining pattern of adult mice was not observed before P8. 18483067_This study evaluated a model of benign familial neonatal convulsions in transgenic mice with a knockin mutation for KCNQ2 and KCNQ3 mutations. 18827480_expression of Kcnq2 is in the modiolus and organ of Corti, while Kcnq3 expression was also detected in the cochlear lateral wall and both in spiral ganglion neurons 19060215_KCNQ2 and KCNQ3 channels contribute to the apamin-insensitive afterhyperpolarization. 20132415_Data show that in early pregnant mouse myometrium, the relative abundance of mRNA expression was KCNQ3 > KCNQ4 > KCNQ5 > KCNQ1 > KCNQ2. 20885443_These findings provide novel evidence that KCNQ2/3 channels could be an important regulator in neuronal apoptosis. 20962009_These findings demonstrate that spectrin cytoskeleton finely regulates ion channel distribution and implicates KCNQ2/3 subunits in axonal excitability and in myokymia etiology. 21838927_Accumulation of Kv7 channels in afferent fibers may increase M-type currents which then acquired a more important role at regulating fiber excitability. 23259949_Results show that in the same protein complex in which PKA augments L currents, AKAP79/150 directs calcineurin to activate NFAT and initiate a longer-term feedback loop that upregulates M-channel expression, countering increased neuronal excitability. 23650395_Retigabine is more effective on KCNQ3 than KCNQ2, whereas ZnPy is more effective on KCNQ2 with no detectable effect on KCNQ3. 23716673_Data show that a reduction in Kv7.2/3 channel activity is essential for tinnitus induction and for the tinnitus-specific hyperactivity. 23977150_Reduced M-current in the superior cervical ganglion neurons of Kcnq2 truncation mutation heterozygotic mice. 26312501_Resilience to tinnitus is developed in mice that show a re-emergence of KCNQ2/3 channel activity and a reduction in HCN channel activity. 26733196_Mechanosensitivity of Skin Down-hair mechanoeceptors is increased in Kcnq3-/- and in Kcnq2+/-/Kcnq3-/- Mutant Mice. 27445338_USP36 actions extend beyond TrkA because the presence of USP36 interferes with Nedd4-2-dependent Kv7.2/3 channel regulation. 28100740_Kcnq2 ablation leads to increased neuronal excitability of neocortex layer 2/3 pyramidal neurons. 28575529_The data of this study showed that, in Kcnq2 mutant slices, burst activity was modulated by GABAA receptor blockade. 30382937_decreases in interneuron KCNQ2/3 activity remodels excitatory networks, providing a new function for these channels. 30952836_The ionic mechanism driving increased excitability involves alterations of the expression levels of HCN and KCNQ channel genes leading to a complex dynamic of H-current and M-current activation. Moreover, there is a negative correlation between the disease onset and disease progression, which correlates with a decrease in the expression level of HCN and KCNQ channels. 31283873_Heterozygous loss of epilepsy gene KCNQ2 alters social, repetitive and exploratory behaviors. 31621989_Functional responses of the hippocampus to hyperexcitability depend on directed, neuron-specific KCNQ2 K(+) channel plasticity. 33512443_Impaired Kv7 channel activity in the central amygdala contributes to elevated sympathetic outflow in hypertension. 33768249_Kcnq2/Kv7.2 controls the threshold and bi-hemispheric symmetry of cortical spreading depolarization. 34509475_The SUMO-specific protease SENP2 plays an essential role in the regulation of Kv7.2 and Kv7.3 potassium channels. 34785595_Posttranscriptional modulation of KCNQ2 gene expression by the miR-106b microRNA family. 34878647_Muscarinic signaling regulates voltage-gated potassium channel KCNQ2 phosphorylation in the nucleus accumbens via protein kinase C for aversive learning. 34911751_Spontaneous seizure and memory loss in mice expressing an epileptic encephalopathy variant in the calmodulin-binding domain of Kv7.2. 35320039_KCNQ2 and KCNQ5 form heteromeric channels independent of KCNQ3. 35389519_Time-limited alterations in cortical activity of a knock-in mouse model of KCNQ2-related developmental and epileptic encephalopathy. 35892317_Genetic interaction between Scn8a and potassium channel genes Kcna1 and Kcnq2. 36047730_Mouse models of Kcnq2 dysfunction. 36070693_Dopamine drives neuronal excitability via KCNQ channel phosphorylation for reward behavior. 36113748_Epilepsy phenotype and response to KCNQ openers in mice harboring the Kcnq2 R207W voltage-sensor mutation. |
ENSG00000075043 |
KCNQ2 |
9.531242 |
0.0706218312 |
-3.823742 |
1.03639801 |
13.354127 |
0.00025785449283084884526326407971907883620588108897209167480468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0013910025113834741019441798570710489002522081136703491210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.41224133754193 |
1.10405994320109 |
19.9972347506028 |
9.16237859398487 |
| ENSMUSG00000016356 |
73368 |
Col20a1 |
protein_coding |
Q923P0 |
FUNCTION: Probable collagen protein. |
Alternative splicing;Collagen;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:73368; |
collagen trimer [GO:0005581]; collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615] |
Human_homologues 29934816_Our study proposes COL20A1 gene as another potential candidate gene for striate palmoplantar keratoderma (PPKS)which expand the spectrum of collagen proteins in the pathogenicity of Palmoplantar keratoderma. 35456962_Type XX Collagen Is Elevated in Circulation of Patients with Solid Tumors. |
ENSG00000101203 |
COL20A1 |
263.766622 |
2.9545733253 |
1.562950 |
0.18308339 |
71.227989 |
0.00000000000000003182413291255458153628147507695819322308848159592056803468551606783876195549964904785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000009953700051828159900286053104799690088124776172345820057785203971434384584426879882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
399.190885676229 |
50.7948981535639 |
135.10948679532 |
13.3974601857989 |
| ENSMUSG00000016624 |
271305 |
Phf21b |
protein_coding |
Q8C966 |
|
Coiled coil;Metal-binding;Reference proteome;Zinc;Zinc-finger |
|
|
mmu:271305; |
metal ion binding [GO:0046872] |
32820037_Phf21b imprints the spatiotemporal epigenetic switch essential for neural stem cell differentiation. 35866480_The epigenetic reader PHF21B modulates murine social memory and synaptic plasticity-related genes. |
ENSG00000056487 |
PHF21B |
114.971394 |
2.8986064158 |
1.535359 |
0.39800296 |
13.988505 |
0.00018393169880228236773463124098526577654411084949970245361328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010330559130370077156046981770032289205119013786315917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
170.970386134645 |
47.7669535368922 |
58.9836499378485 |
12.4716164659726 |
| ENSMUSG00000016918 |
240725 |
Sulf1 |
protein_coding |
Q8K007 |
FUNCTION: Exhibits arylsulfatase activity and highly specific endoglucosamine-6-sulfatase activity. It can remove sulfate from the C-6 position of glucosamine within specific subregions of intact heparin. Diminishes HSPG (heparan sulfate proteoglycans) sulfation, inhibits signaling by heparin-dependent growth factors, diminishes proliferation, and facilitates apoptosis in response to exogenous stimulation (By similarity). {ECO:0000250}. |
Apoptosis;Calcium;Endoplasmic reticulum;Glycoprotein;Golgi apparatus;Hydrolase;Metal-binding;Reference proteome;Signal |
|
|
mmu:240725; |
cell surface [GO:0009986]; endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; Golgi stack [GO:0005795]; membrane raft [GO:0045121]; plasma membrane [GO:0005886]; arylsulfatase activity [GO:0004065]; calcium ion binding [GO:0005509]; glycosaminoglycan binding [GO:0005539]; N-acetylglucosamine-6-sulfatase activity [GO:0008449]; apoptotic process [GO:0006915]; bone development [GO:0060348]; cartilage condensation [GO:0001502]; cartilage development [GO:0051216]; cell adhesion [GO:0007155]; chondrocyte development [GO:0002063]; embryonic skeletal system development [GO:0048706]; esophagus smooth muscle contraction [GO:0014846]; glial cell-derived neurotrophic factor receptor signaling pathway [GO:0035860]; glomerular basement membrane development [GO:0032836]; glomerular filtration [GO:0003094]; heparan sulfate proteoglycan metabolic process [GO:0030201]; innervation [GO:0060384]; kidney development [GO:0001822]; limb joint morphogenesis [GO:0036022]; negative regulation of angiogenesis [GO:0016525]; negative regulation of cell migration [GO:0030336]; negative regulation of endothelial cell proliferation [GO:0001937]; negative regulation of fibroblast growth factor receptor signaling pathway [GO:0040037]; negative regulation of prostatic bud formation [GO:0060686]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of vascular endothelial growth factor production [GO:0010575]; positive regulation of Wnt signaling pathway [GO:0030177]; regulation of fibroblast growth factor receptor signaling pathway [GO:0040036]; sulfur compound metabolic process [GO:0006790]; vascular endothelial growth factor receptor signaling pathway [GO:0048010] |
12368295_Sulfs are extracellular endosulfatases with strong potential for modulating the interactions of heparan sulfate proteoglycans in the extracellular microenvironment 17116694_Sulf2 may have roles in several tissues but that there is compensation by and/or redundancy with Sulf1. 17593974_results show that Sulf1 and Sulf2 play overlapping yet critical roles in mouse development and are redundant and essential for neonatal survival 17720696_SULF1 is expressed in GDNF-expressing esophageal muscle and SULF2 in innervating neurons, establishing their direct functions in esophageal innervation. 17920055_Results establish Sulfs 1 and 2 as essential regulators of heparan sulfate-dependent growth factor signaling in the adult muscle stem cell niche. 18213582_Redundant function of the heparan sulfate 6-O-endosulfatases Sulf1 and Sulf2 in fine-tuning rather than regulating the development of skeletal structures. 18503048_Sulf1 is a TGF-beta1-responsive gene both in vitro and in vivo and may function as a negative regulator of TGF-beta1-induced fibrogenesis 18687675_Sulf1 and Sulf2 are two heparan sulfate 6-O-endosulfatases that regulate the activity of multiple growth factors, such as fibroblast growth factor and Wnt, and are essential for mammalian development and survival 20394677_These results reveal that Sulf1 and Sulf2 fulfil non-redundant functions in vivo in the development and maintenance of the murine nervous system. 20410206_The data identify sulfatase 1 as an inhibitor of prostatic branching morphogenesis and growth factor signaling that is down-regulated as part of the normal response to androgen action in the male urogenital sinus. 20442248_reduction in protein levels promotes satellite cell proliferation 20855470_Sulf-1 and Sulf-2 are essential regulators of GDNF signaling in the SSC niche and direct downstream targets of Sertoli cell-specific transcriptional factor Wilm's tumor 1. 22298771_Sulf1 and Sulf2 differentially contribute to the generation of organ-specific sulfation patterns of heparan sulfate. 23074159_Distinct expression patterns of Sulf1 and Hs6st1 spatially regulate heparan sulfate sulfation during prostate development. 23238718_Our study provides additional insights into the temporal control of Olig2 progenitor cell fate change by the identification of Sulf1 as an extracellular timing signal in the ventral spinal cord. 23950901_Sulf1/, but not Sulf2/, mice, exhibited a marked delay in healing of corneal epithelial wounds. 25370455_Expression of the heparan sulfate 6-O-endosulfatases, Sulf1 and Sulf2, in the avian and mammalian inner ear suggests a role for sulfation during inner ear development. 25448158_It is proposed that Sulf1, Sulf2 and ErbB1 are involved in the inhibition of neurite outgrowth and may regulate structural plasticity and regeneration in the nervous system. 25503294_Our study proposes a novel role of SULF1 in hepatocellular carcinoma tumor progression through augmentation of the TGF-beta pathway, thus defining SULF1 as a potential biomarker for tumor progression and a novel target for drug development for HCC. 26764203_Sulf1 and Sulf2 play indispensable roles to maintain glomerular integrity and protective roles in diabetic nephropathy, probably by growth factor modulation. 27626699_our findings show that Sulf-1 is an important tumor suppressor gene in hepatocellular carcinoma (HCC), and its over expression downregulates Msln and results in a decrease in HCC cell proliferation, migration, invasion, and lymphatic metastasis. 28490013_Shh signaling requires the coordinated activity of Sulf1 and Sulf2 in order to reach that threshold in the mouse ventral spinal cord 29062064_Loss of SULF1 expression is associated with corticospinal tract defect. 30924907_Sulf-1 might play a critical role from development to maintenance of intervertebral disc homeostasis by regulating collagen expression 34324645_Regulation of fractone heparan sulfate composition in young and aged subventricular zone neurogenic niches. |
ENSG00000137573 |
SULF1 |
63.140455 |
2.0705836942 |
1.050038 |
0.31615228 |
11.026867 |
0.00089800796426442637283155123739675218530464917421340942382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0041997105654515637720991527714886615285649895668029785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
80.3941242236621 |
17.7589184562681 |
38.8267928740393 |
6.69343517437158 |
| ENSMUSG00000017057 |
16164 |
Il13ra1 |
protein_coding |
O09030 |
FUNCTION: Binds with low affinity to interleukin-13 (IL13). Together with IL4RA can form a functional receptor for IL13. Also serves as an alternate accessory protein to the common cytokine receptor gamma chain for interleukin-4 (IL4) signaling, but cannot replace the function of IL2RG in allowing enhanced interleukin-2 (IL2) binding activity (By similarity). {ECO:0000250}. |
Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a transmembrane protein that complexes with interleukin 4 receptor alpha (IL4RA) to form a functional receptor for interleukin-13. Signalling through this pathway mediates allergy response and occurs during bronchial asthma. [provided by RefSeq, May 2015]. |
mmu:16164; |
external side of plasma membrane [GO:0009897]; receptor complex [GO:0043235]; ciliary neurotrophic factor receptor binding [GO:0005127]; cytokine binding [GO:0019955]; cytokine receptor activity [GO:0004896]; interleukin-13 receptor activity [GO:0016515]; leukemia inhibitory factor receptor activity [GO:0004923]; cytokine-mediated signaling pathway [GO:0019221]; oncostatin-M-mediated signaling pathway [GO:0038165]; positive regulation of cell population proliferation [GO:0008284] |
18066066_IL-13R alpha 1 is required for allergen-induced airway hyperreactivity and mucus production but not for alternative macrophage activation. 18066066_Il13ra1-/- mice showed less mortality after infection with Schistosoma mansoni and more susceptibility to Nippostrongylus brasiliensis. IL-13Ralpha1 was essential for allergen-induced airway hyperreactivity and mucus hypersecretion. 18480254_IL-13 and IL-4 via IL-13 receptor alpha1 and the type II IL-4 receptor have direct roles in asthma pathogenesis 18852293_Differential expression of IL-4Ralpha, IL-13Ralpha1, and gammac regulates relative cytokine sensitivity. 20383033_peaks of upregulation in allergic bronchial asthma at 6 hours and 3-12 hours after the last antigen inhalation 21303908_a central role for the IL-13/IL-13Ralpha1 pathway in the regulation of intestinal epithelial cell Cl(-) secretion via up-regulation of cystic fibrosis transmembrane conductance regulator 21617187_The loss of IL-13Ralpha1 on islet-reactive T cells may be a biomarker for fading regional immune regulation and progression to overt diabetes. 21957151_IL-13Ralpha1 is the key receptor mediating airway hypersensitivity responses, mucus production, and transforming growth factor (TGF)-beta induction in response to aeroallergens. 22829596_The extracellular and transmembrane domains of the gammaC and interleukin (IL)-13 receptor alpha1 chains, not their cytoplasmic domains, dictate the nature of signaling responses to IL-4 and IL-13 23169588_Expression of IL-13Ralpha1 on dopaminergic neurons can increase their susceptibility to oxidative stress-mediated damage, thereby contributing to their preferential loss. 23650613_in neonatal mice, IL-12 uses IL-12Rbeta2 to counter IL-13Ralpha1 expression in addition to promoting Th1 differentiation 24281978_IL-13Ralpha1 is a surface marker for M2 macrophages influencing their differentiation and function. 24841514_CAR T cells killed IL13Ra1- and/or IL13Ra2-positive cells in contrast to IL13Ra1- and IL13Ra2-negative controls 26153764_IL-13Ralpha1 has a protective role in bleomycin-induced pulmonary injury and repair. 26160172_These data establish for the first time a molecular mechanism by which Mac-1 regulates the signaling activity of IL-13 in macrophages. 26810038_These results show that activation of IL-13Ralpha1 is critical for key aspects of the immune and functional responses to Hb2 infection that facilitate expulsion. 28427412_Chronic stress significantly reduced the number of substantia nigra dopaminergic neurons in Il13a1 knock-out mice. 28646042_findings indicate that NOD mice lacking the IL-4Ralpha/IL-13Ralpha1 heteroreceptor (HR) display resistance to Type 1 Diabetes as the rise in blood glucose level and islet inflammation were significantly delayed in these HR-deficient relative to HR-sufficient mice 28874358_Delivery of sIL13ralpha1 to mice by either gene transfer or recombinant protein decreases blood glucose levels. Surprisingly, the glucose-lowering effect of sIL13ralpha1 was preserved in mice lacking IL-13, demonstrating that IL-13 was not required for the effect. In contrast, deletion of IL-4 in mice eliminated the hypoglycemic effect of sIL13ralpha1. 29929727_IL-13 receptor alpha 1 (IL-13Ralpha1) dendritic cell are able to take up antigen from the peritoneum and carry it to the the thymus. 32276028_Two single nucleotide polymorphisms in IL13 and IL13RA1 from individuals with idiopathic Parkinson's disease increase cellular susceptibility to oxidative stress. 33419470_Expression of IL4Ralpha and IL13Ralpha1 are associated with poor prognosis of soft-tissue sarcoma of the extremities, superficial trunk, and retroperitoneum. 34808240_IL-13Ralpha1 Suppresses Tumor Progression in Two-Stage Skin Carcinogenesis Model by Regulating Regulatory T Cells. 35438841_Interleukin-13 Receptor alpha1-Mediated Signaling Regulates Age-Associated/Autoimmune B Cell Expansion and Lupus Pathogenesis. |
ENSG00000131724 |
IL13RA1 |
395.022184 |
2.0997234806 |
1.070199 |
0.13236538 |
64.845905 |
0.00000000000000080990231211818548820508617307444518026768835597742346443794758670264855027198791503906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000221479928513181351763572714014165252296214431160237268159107770770788192749023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
533.322079657268 |
49.0002823152857 |
253.996340272651 |
18.0852802620922 |
| ENSMUSG00000017344 |
22370 |
Vtn |
protein_coding |
P29788 |
FUNCTION: Vitronectin is a cell adhesion and spreading factor found in serum and tissues. Vitronectin interact with glycosaminoglycans and proteoglycans. Is recognized by certain members of the integrin family and serves as a cell-to-substrate adhesion molecule. Inhibitor of the membrane-damaging effect of the terminal cytolytic complement pathway. |
Cell adhesion;Direct protein sequencing;Disulfide bond;Glycoprotein;Heparin-binding;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal;Sulfation |
|
|
mmu:22370; |
basement membrane [GO:0005604]; collagen-containing extracellular matrix [GO:0062023]; cytoplasm [GO:0005737]; endoplasmic reticulum [GO:0005783]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; Golgi lumen [GO:0005796]; intracellular membrane-bounded organelle [GO:0043231]; peptidase inhibitor complex [GO:1904090]; protein complex involved in cell-matrix adhesion [GO:0098637]; rough endoplasmic reticulum lumen [GO:0048237]; collagen binding [GO:0005518]; extracellular matrix binding [GO:0050840]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; integrin binding [GO:0005178]; polysaccharide binding [GO:0030247]; scavenger receptor activity [GO:0005044]; cell adhesion mediated by integrin [GO:0033627]; cell migration [GO:0016477]; cell-matrix adhesion [GO:0007160]; endodermal cell differentiation [GO:0035987]; extracellular matrix organization [GO:0030198]; immune response [GO:0006955]; liver regeneration [GO:0097421]; negative regulation of endopeptidase activity [GO:0010951]; negative regulation of fibrinolysis [GO:0051918]; oligodendrocyte differentiation [GO:0048709]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of protein binding [GO:0032092]; positive regulation of receptor-mediated endocytosis [GO:0048260]; positive regulation of smooth muscle cell migration [GO:0014911]; protein polymerization [GO:0051258]; regulation of cell adhesion [GO:0030155]; smooth muscle cell-matrix adhesion [GO:0061302] |
12067901_Endogenous vitronectin promotes neointima formation in carotid arteries. 12393886_regulation of cell spreading on vitronectin by ERK1 association with alphaVbeta3 integrin 12482822_In a carotid artery ligation model, vitronectin (VN) knockout mice generate a larger intima than wild-type as a result of more extensive smooth muscle cell (SMC)proliferation, demonstrating that VN inhibits thrombin-mediated SMC proliferation. 12562917_vitronectin can selectively regulate inactivating potassium current in embryonic hippocampal neurons 12603314_competition with plasminogen activator inhibitor type-1 for alphavbeta3 binding inhibits insulin signaling 15733060_in contrast to released platelet granule Vn which enhances platelet aggregation, plasma Vn inhibits platelet aggregation 16479252_There is an increase of vitronectin and integrin alphav in the retina and the optic nerve after optic nerve injury. Induction of Vitronectin and integrin alphav may have an important role in the regeneration of adult mammalian retinal ganglion cells. 17504807_PrP(C) plays an important role in axonal growth, and this function may be rescued in PrP(C)-knockout animals by integrin compensatory mechanisms. 17982099_a novel vitronectin-dependent mechanism contributing to the development of acute lung injury. 19574558_VN is a key determinant of the antiproliferative effect of PAI-1 overexpression. 19760599_evaluated the temporal expression pattern of STI1 and Vn and their colocalization with their ligand PrPC, demonstrated that PrPC, STI1, and Vn are coexpressed in most of the embryonic tissues evaluated. 20492459_Plasminogen activator inhibitor-1 and vitronectin expression level and stoichiometry regulate vascular smooth muscle cell migration through physiological collagen matrices 21270094_Suggest that despite significant interstitial Vtn deposition in the ureteral obstruction model of chronic kidney disease, its fibrogenic role is either nonessential or redundant. 21734232_the vitronectin-binding capacity of PAI-1 was the primary determinant required for its ability to exacerbate lung scarring induced by intratracheal bleomycin administration 21764994_These results demonstrate that vitronectin exerts a deleterious role independently from complement, by directing PAI-dependent fibrinolysis in the glomerular compartment. 22281987_ability of vitronectin to enhance neutrophil viability was dependent on activation of phosphatidylinositol 3-kinase and extracellular signal-regulated kinase 1/2 kinases, but not on the p38 kinase 22606350_VN plays a previously unrecognized role in regulating endothelial permeability via conformational- and integrin-dependent effects on VE-cadherin trafficking. 23019340_the extracellular matrix molecule VN and its neuronal receptor TLCN play a pivotal role in the phosphorylation of ezrin/radixin/moesin proteins and the formation of phagocytic cup-like structures on neuronal dendrites 23345331_Vitronectin inhibits efferocytosis through interactions with apoptotic cells as well as with macrophages. 24509439_These data suggest that adhesion to vitronectin allows for the uncoupling of fibronectin fibril formation from downstream signaling events by reducing alpha5beta1 integrin activation and fibronectin fibril extension. 24603119_Report role of vitronectin in the regulation of angiogenesis induced by VEGF. 25799354_PAI-1, through its interaction with Vn, exerts multiple deleterious mechanisms to induce AKI. 26640242_In vtn knockout mice, neuronal differentiation in the cerebellum was suppressed compared with wild-type mice. 26648042_Elevated levels of TIMP3 and vitronectin, acting downstream of Notch3(ECD) deposition, play a role in CADASIL, producing divergent influences on early CBF deficits and later white matter lesions. 27106291_the TGF-beta-PAI-1-vitronectin-integrin axis may represent a critical convergence of factors in determining the alveolar epithelial cell apoptotic response during lung injury. 27798934_this study demonstrates in a mouse infection model that Vn reduces complement-mediated killing of Yersinia enterocolitica O:9 E40 and, thus, improves bacterial survival 29028290_The effects of genetic alterations in PAI-1 expression, pharmacologic PAI-1 inhibition and recombinant PAI-1 on smooth muscle cell vitronectin (VN) expression were studied, and vascular VN expression in wild-type and PAI-1-deficient mice was assessed. 29222114_blood-derived vitronectin (VTN) rapidly and potently activates leukemia inhibitory factor (LIF) and pro-inflammatory interleukin 6 (IL-6) in vitro and after vascular injury in the brain. 29661186_VTN had a novel therapeutic potential for the treatment of Radiation-induced lung toxicity. 30937689_Vitronectin regulates the multipolar-to-bipolar morphological transition of nitrites via alphavbeta5 integrin. 31678139_these data suggest that leakage of VTN into the subventricular zone reduces the neurogenic response to stroke in female mice 32312218_Blood Vitronectin Induces Detrimental Brain Interleukin-6 and Correlates With Outcomes After Stroke Only in Female Mice. |
ENSG00000109072 |
VTN |
67.554511 |
3.2415857469 |
1.696700 |
0.32529187 |
27.057809 |
0.00000019746090406194351243289333928260242956298498029354959726333618164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000019722783545387038364336742407578739744167251046746969223022460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
106.198426184894 |
24.1485123597039 |
32.761257753883 |
5.87304125290366 |
| ENSMUSG00000017400 |
217154 |
Stac2 |
protein_coding |
Q8R1B0 |
FUNCTION: Plays a redundant role in promoting the expression of calcium channel CACNA1S at the cell membrane, and thereby contributes to increased channel activity (PubMed:29467163). Slows down the inactivation rate of the calcium channel CACNA1C (PubMed:25548159, PubMed:29363593). {ECO:0000269|PubMed:25548159, ECO:0000269|PubMed:29363593, ECO:0000269|PubMed:29467163}. |
Cell membrane;Cytoplasm;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Repeat;SH3 domain;Zinc;Zinc-finger |
|
|
mmu:217154; |
cytosol [GO:0005829]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; sarcolemma [GO:0042383]; metal ion binding [GO:0046872]; positive regulation of protein localization to plasma membrane [GO:1903078]; positive regulation of voltage-gated calcium channel activity [GO:1901387]; skeletal muscle contraction [GO:0003009] |
20736085_found STAC1 and STAC2 expressed in mutually exclusive fashion in adult DRG neurons; STAC1 marks peptidergic nociceptive neurons; STAC2 is expressed in subset of nonpeptidergic nociceptors, all trkB+ neurons and a subpopulation of proprioceptive neurons 29348675_STAC2 physically interacts with RANK and inhibits the formation of the RANK signaling complex, which contains Grb-2-associated binder 2 (Gab2) and phospholipase Cgamma2 (PLCgamma2), thus leading to the suppression of RANK-mediated NF-kappaB and MAPK activation. |
ENSG00000141750 |
STAC2 |
396.356862 |
3.9370850552 |
1.977128 |
0.15356232 |
162.292465 |
0.00000000000000000000000000000000000035710328858254313296941169956567747985613909506538535082711421875702025403059831913387702200662898732730354822706431150436401367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000365060220918744732188011567169154394853020183903576104818951532938118931838763425472071466951207163731396576622501015663146972656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
634.204392939655 |
67.6330734295379 |
161.084757898413 |
13.6311506036979 |
| ENSMUSG00000017692 |
246104 |
Rhbdl3 |
protein_coding |
P58873 |
FUNCTION: May be involved in regulated intramembrane proteolysis and the subsequent release of functional polypeptides from their membrane anchors. {ECO:0000250}. |
Hydrolase;Membrane;Protease;Reference proteome;Repeat;Serine protease;Transmembrane;Transmembrane helix |
|
|
mmu:246104; |
membrane [GO:0016020]; calcium ion binding [GO:0005509]; serine-type endopeptidase activity [GO:0004252]; proteolysis [GO:0006508] |
11900977_Cloning and expression of Ventrhoid 27264103_Here the authors show that the mammalian rhomboid protease RHBDL4 (also known as Rhbdd1) promotes trafficking of several membrane proteins, including the EGFR ligand TGFalpha, from the endoplasmic reticulum (ER) to the Golgi apparatus, thereby triggering their secretion by extracellular microvesicles. |
ENSG00000141314 |
RHBDL3 |
176.773560 |
0.4834456388 |
-1.048574 |
0.23561974 |
19.395838 |
0.00001062383196466640417358808651249191257193160708993673324584960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000784402367841971227000255617589630219299579039216041564941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
116.339062818192 |
19.0849776603713 |
240.6455938159 |
28.571506153616 |
| ENSMUSG00000017724 |
18612 |
Etv4 |
protein_coding |
P28322 |
FUNCTION: Transcriptional activator (PubMed:1547944). May play a role in keratinocyte differentiation (By similarity). {ECO:0000250|UniProtKB:P43268, ECO:0000269|PubMed:1547944}. |
Activator;Alternative splicing;DNA-binding;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:18612; |
chromosome [GO:0005694]; nucleolus [GO:0005730]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; branching involved in mammary gland duct morphogenesis [GO:0060444]; cell differentiation [GO:0030154]; motor neuron axon guidance [GO:0008045]; negative regulation of mammary gland epithelial cell proliferation [GO:0033600]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of keratinocyte differentiation [GO:0045618]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; stem cell differentiation [GO:0048863]; transcription by RNA polymerase II [GO:0006366] |
12372283_Induction of Pea3 gene expression by peripheral signals is required to coordinate the central position and terminal arborization of specific sets of spinal motor neurons. 12372284_PEA3 expression in developing motor neurons is regulated by GDNF signals from the limb bud. 12527001_motoneurons innervating the cutaneous maximus muscle, which are normally PEA3(+), fail to develop normally so that postnatally the muscle is innervated by few motoneurons and is severely atrophic 12871699_Pea3 is expressed in tissues undergoing branching morphogenesis and promotes formation of duct-like structures by mammary epithelial cells in vitro. 12941618_Pea3 subfamily members play a role in epithelial-mesenchymal interactions during lung organogenesis. 12948444_pea3 is induced by GDNF in a subset of the motor neurons. Signaling by the HGF receptor (met), is required for rostral expansion of the pea3 domain, while pea3 expression is independent of met function. 14990565_Beta-catenin, Lef-1, Ets transcription factors, and the AP-1 protein c-Jun each weakly enhanced luciferase expression from an OPN promoter. 15138262_ETS-1, PEA3, and ERM have roles in regulation of caveolin-1 transcription in epithelial cells 17190606_We provide genetic evidence that the induction of the ETS transcription factor Pea3 by GDNF is essential in two cervical MN pools to control dendrite patterning and selectivity of IaPA connectivity. 17654714_precocious axon growth into transplanted older donor limbs, which prematurely exposes neurons to limb-derived signals, does not accelerate the onset of expression of Er81 or Pea3 18492810_The results show that Etv4 and Etv5 are expressed in granulosa and cumulus cells during folliculogenesis and ovulation, suggesting that they influence cellular events in the ovary by regulating downstream genes such as Ptgs2. 18827017_Reduced tumorigenesis in mouse mammary cancer cells following inhibition of Pea3- or Erm-dependent transcription. 18977342_Pea3 functions in multipotential progenitors to regulate their lineage-specific differentiation potential. 19307308_Sumoylation of PEA3 plays a positive role in PEA3-mediated transcriptional activation and the ERK MAP kinase pathway cooperates with rather than antagonizes this process. 19384958_Fgf4 positively regulated Pea3, Sprouty1, and 2 expression in chick limb mesenchyme. 19386268_FGF activity in the limb is not only responsible for maintaining posterior-specific Shh expression, but it also acts via Etvs to prevent inappropriate anterior expansion of Shh. 19386269_ETV4 and ETV5 act downstream of FGF signaling to inhibit Shh expression in the anterior limb bud. 19898483_Etv4 is positively regulated by Ret signaling in the ureteric bud tips. 20107508_study provides evidence for a protumorigenic role of PEA3 factors in breast neoplasia, and supports targeting the PEA3 transcription factor family in breast cancer 21613609_Smad2 and PEA3 regulate RGC-32 transcription which is essential for smooth muscle cell differentiation from neural crest cells. 22982417_PEA3-null fibroblasts exhibit impaired c-src activation and motility defects. 23918374_oncogenic Etv4 promotes prostate cancer metastasis in response to coactivation of PI3-kinase and Ras signaling pathways in a genetically engineered model of highly penetrant, metastatic prostate cancer 23989931_Study reveals molecular insight into how the Ets family transcription factor Pea3 favors EMT and contributes to tumorigenesis via a negative regulatory loop with Cyclin D2, a new Pea3 target gene. 24260201_Study hypothesized that PEA3 might play an essential role in the activation of the FAK gene during tumor metastasis. 26215568_paracrine signaling via fibroblast growth factor 2 (Fgf2) and Mapk between these diverged tumor subclones causes enhanced expression of the Pea3 transcription factor, resulting in metastatic dissemination of the neuroendocrine tumor subclones. 26224636_ETS-related transcription factors ETV4 and ETV5 are involved in proliferation and induction of differentiation-associated genes in embryonic stem (ES) cells. 27909004_This study demonstrates that Etv4 and Etv5 transcription factors are expressed in hippocampal pyramidal neurons during the main period of dendritogenesis; reveals a novel role for Etv4 and Etv5 transcription factors for hippocampal circuit development and the coordination of synaptic maturation; and indicates that both factors are essential in the transcriptional program linking BDNF signaling to dendrite development. 28373072_Our results will help understand the mechanism of ETV4 overexpression in CRC patients and provide a clue to search new therapeutic target to treat the related tumors in clinical practice. 28949289_The E26 transformation-specific transcription factor, ETV4, which is induced by fibroblast growth factor signalling and acts as a repressor of ZRS activity, interacts with the histone deacetylase HDAC2 and ensures that the poised ZRS remains transcriptionally inactive. 32043969_Etv transcription factors functionally diverge from their upstream FGF signaling in lens development. 33010000_Amniotic fluid mesenchymal stem cells repair mouse corneal cold injury by promoting mRNA N4-acetylcytidine modification and ETV4/JUN/CCND2 signal axis activation. 35904071_Etv4 regulates nociception by controlling peptidergic sensory neuron development and peripheral tissue innervation. 36301850_Transcription factor gene Pea3 regulates erectile function during copulation in mice. |
ENSG00000175832 |
ETV4 |
213.935073 |
0.3897504030 |
-1.359378 |
0.18958024 |
50.531269 |
0.00000000000117281236388608659840680449562731602736707914313996070632128976285457611083984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000231702696844524939611910419705456199226945024349788582185283303260803222656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
120.009013689672 |
15.81545017734 |
307.912481317364 |
29.0504176225949 |
| ENSMUSG00000017737 |
17395 |
Mmp9 |
protein_coding |
P41245 |
FUNCTION: Matrix metalloproteinase that plays an essential role in local proteolysis of the extracellular matrix and in leukocyte migration (By similarity). Could play a role in bone osteoclastic resorption (PubMed:8132709). Cleaves KiSS1 at a Gly-|-Leu bond (By similarity). Cleaves NINJ1 to generate the Secreted ninjurin-1 form (PubMed:23142597, PubMed:32883094). Cleaves type IV and type V collagen into large C-terminal three quarter fragments and shorter N-terminal one quarter fragments. Degrades fibronectin but not laminin or Pz-peptide (By similarity). {ECO:0000250|UniProtKB:P14780, ECO:0000269|PubMed:23142597, ECO:0000269|PubMed:32883094, ECO:0000269|PubMed:8132709}. |
Calcium;Collagen degradation;Disulfide bond;Extracellular matrix;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Repeat;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes a member of the matrix metalloproteinase family of extracellular matrix-degrading enzymes that are involved in tissue remodeling, wound repair, progression of atherosclerosis and tumor invasion. The encoded preproprotein undergoes proteolytic processing to generate a mature, zinc-dependent endopeptidase enzyme that degrades collagens of type IV, V and XI, and elastin. Mice lacking the encoded protein exhibit an abnormal pattern of skeletal growth plate vascularization and ossification, reduced keratinocyte hyperproliferation at all neoplastic stages, a decreased incidence of invasive tumors, and resistance to experimental autoimmune encephalomyelitis. [provided by RefSeq, Feb 2016]. |
mmu:17395; |
collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; protein-containing complex [GO:0032991]; endopeptidase activity [GO:0004175]; fibronectin binding [GO:0001968]; identical protein binding [GO:0042802]; metalloendopeptidase activity [GO:0004222]; metallopeptidase activity [GO:0008237]; peptidase activity [GO:0008233]; protein-containing complex binding [GO:0044877]; serine-type endopeptidase activity [GO:0004252]; zinc ion binding [GO:0008270]; aging [GO:0007568]; apoptotic process [GO:0006915]; cell migration [GO:0016477]; cellular response to cadmium ion [GO:0071276]; cellular response to cell-matrix adhesion [GO:0071460]; cellular response to reactive oxygen species [GO:0034614]; cellular response to UV-A [GO:0071492]; collagen catabolic process [GO:0030574]; embryo implantation [GO:0007566]; endodermal cell differentiation [GO:0035987]; extracellular matrix organization [GO:0030198]; heart development [GO:0007507]; macrophage chemotaxis [GO:0048246]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cation channel activity [GO:2001258]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic signaling pathway [GO:2001268]; negative regulation of epithelial cell differentiation involved in kidney development [GO:2000697]; negative regulation of fibroblast proliferation [GO:0048147]; negative regulation of intrinsic apoptotic signaling pathway [GO:2001243]; positive regulation of angiogenesis [GO:0045766]; positive regulation of apoptotic process [GO:0043065]; positive regulation of DNA binding [GO:0043388]; positive regulation of epidermal growth factor receptor signaling pathway [GO:0045742]; positive regulation of keratinocyte migration [GO:0051549]; positive regulation of leukocyte migration [GO:0002687]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of receptor binding [GO:1900122]; positive regulation of release of cytochrome c from mitochondria [GO:0090200]; positive regulation of synaptic plasticity [GO:0031915]; positive regulation of vascular associated smooth muscle cell proliferation [GO:1904707]; protein catabolic process [GO:0030163]; proteolysis [GO:0006508]; response to amyloid-beta [GO:1904645]; response to oxidative stress [GO:0006979]; response to xenobiotic stimulus [GO:0009410]; skeletal system development [GO:0001501]; tissue remodeling [GO:0048771] |
11819823_Secretion of MMPs which was associated with metastatic ability of Hca-F and Hca-P tumor cells depends on the environment of lymph nodes 11827968_Exposure of cryptic domains in the alpha 1-chain of laminin-1 by elastase stimulates macrophages urokinase and matrix metalloproteinase-9 expression. 11985514_MMP-9 activity in the mouse spinal cord correlates with the severity of experimental autoimmune encephalomyelitis 11994507_Neutrophils from MMP-9-deficient mice show no defect in transendothelial migration 12049638_involved in adipose conversion of 3T3-L1 preadipocytes 12062105_In MMP-9-/- mice, release of sKitL and hematopoietic stem cell motility are impaired, resulting in failure of hematopoietic recovery and increased mortality, while exogenous sKitL restores hematopoiesis and survival after bone marrow ablation. 12163012_Results demonstrate that TGF-beta1 stimulates matrix metalloproteinase-9 production and promoter activity in a process that depends of the activation of the Ras-ERK1,2 MAP kinase pathway. 12163374_a key role in the development of airway inflammation after allergen exposure 12183632_findings suggest a potential extracellular proteolysis pathway to neuronal cell death in which S-nitrosylation activates MMPs, and further oxidation results in a stable posttranslational modification with pathological activity 12189240_the role of matrix metalloproteinases-9 in IL-13-induced inflammation and remodeling in the lung 12193736_MMP-9 enhances the development of antibody-induced arthritis, a mouse model of rheumatoid arthritis. 12237890_Upregulation of matrix metalloproteinase-9 in murine 5T33 multiple myeloma cells by interaction with bone marrow endothelial cells. 12354772_GelB activity plays a role in ganglion cell death and degradation of laminin. 12359218_evidence for the role of promoter methylation in the regulation of gene expression 12359984_Chymase may play a role in heart remodeling by increasing Ang II formation and activating MMP-9, and the regulation of collagen I gene expression. 12368198_contributes to choroidal neovascularization 12368215_enzyme-dependent exposure of a cryptic migratory control site in collagen is required before retinal angiogenesis 12384502_The hemopexin domain is a novel gelatin binding domain and acts as an antagonist 12386280_Role of matrix metalloproteinases in kidney development and glomerulopathy. MMP9 is required for nephron mass formation and renal function in elderly mice and has a novel protective effect on the development of fibrin-induced glomerular lesions. 12393846_MMP-9 is involved in angiogenesis caused by HSV. 12438450_A marked decrease in matrix metalloproteinase-9 (MMP-9) induction was found in the obstructed kidneys of tPA(-/-) mice. 12490006_After traumatic brain injury, deleterious MMP-9 upregulation is mediated by ERK MAPK upstream signaling pathways. 12531895_activation of Liver X receptor represses MMP-9 expression in macrophages 12557216_MRL's break down basement membrane, form blastema, and close ear wounds. Inflammatory response with neutrophils and macrophages seen in injured ear. Cells positive for MMP-2 and MMP-9. 12663332_Membrane-bound MMP-9 on PMN may play pathogenetic roles in inflammatory lung diseases 12672030_TN-C induces MMP-9 expression directly and by collaboration with TGF-beta in breast cancer progression 12673046_Liver injury induces matrix metalloproteinase-9 expression in the liver and is accompanied by the recruitment of hematopoietic progenitor cells from bone marrow into the circulation. 12735482_MMP-9 expression is involved in vascular alterations seen in the wall of thoracic aorta tissue of magnesium-deficient mice. 12816956_MMP9 expression in endothelial cells is differentially regulated by shear stress via activation of c-Myc 12842442_primed polymorphonuclear leukocytes, along with constitutively expressed membrane type 1-matrix metalloproteinase, may activate matrix metalloproteinases-2 and -9 in limb ischemia 12847275_The specific absence of MMP-9 activity in MMP-9-deficient mice inhibits the development of allergic airway inflammation by impairing the recruitment of dendritic cells (DCs) into the airways and the local production of DC-derived proallergic chemokines. 12928773_An association between increased MMP-9 activity and endocardial endothelial apoptosis in diabetic mice. Genetic ablation of MMP-9 correlated with amelioration of endocardial endothelial dysfunction and apoptosis. 12929930_SDF-1 upregulated MMP-9 in various primary murine OC precursor cells 12950084_The gene expression and protein distribution of MMP-9 was analyzed during molar development. 12960961_Data show that tissue plasminogen activator upregulates matrix metalloproteinase-9 in cell culture and in vivo, and that this is mediated by the low-density lipoprotein receptor-related protein. 12963732_GnRH challenge of alphaT3-1 cells furthered the release of active MMP2 and MMP9 that were required GnRH induction of EGFR and Src, Ras, and ERK activation 13679081_MMP-9 expression may play a role in vascular remodeling during in vitro aging 14507973_Early expression and activation of MMP-9 by reactive oxygen species may be involved in early blood-brain barrier disruption and progressive striatal damage after 3-nitropropionic acid treatment 14530359_During growth of the D1-7,12-dimethylbenzanthracene-3 mammary tumor in BALB/c mice, there is progressive up-regulation of MMP-9 in splenic T cells at both the transcriptional and translational levels. 14551157_MMP-9 was necessary for SMC organization of collagen. Deficiency caused a 50% reduction of SMC attachment to gelatin; SMCs may use MMP-9 as a bridge between the cell surface & matrix. CD44 assists in attachment & utilization of MMP-9 by SMCs. 14561647_ubiquitin proteasome pathway effect on embryo implantation is achieved at least in part by regulating MMP-2 and MMP-9 mRNA expression and the gelatinolytic activity of MMP-9. 14563686_MMP-9 promotes choroidal neovascularization. 14572461_MMP-9 is involved in the control of granule cell migration and apoptosis in the developing cerebellum; its expression is spatiotemporally related to cerebellar granule cell migration during postnatal development. 14630814_Endogenous TTPA, through the enhancement of MMP 9 expression and proteolytic activation, plays an essential role in the pathogenesis of heparin-produced cerebral hemorrhage. 14657171_These results emphasize for the first time that upregulation of MMP activity can be important for facilitating regeneration from some types of CNS injury 14670843_Angiogenesis triggered by tissue ischemia requires MMP-9, which may be involved in capillary branching, a potential novel role for this MMP. 14729404_Data suggest that overexpression of matrix metalloproteinase (MMP)-2 and MMP-9 may lead to excessive matrix degradation in the early phase of myocardial infarction, impairing healing and remodeling which in turn causes cardiac rupture. 14764732_MMP-9 is critically involved in the recruitment of eosinophils and T-helper type 2 (Th2) cells to the lung following allergen challenge and plays an important role in the development of Th2 responses to allergen. 14967734_MMP-9 seems to play a key role in the early stages of hypertensive vascular remodeling. 14993123_This study is the first to provide direct genetic evidence for a significant involvement of MMP-9 in atherosclerotic plaque growth. MMP-9 deficiency significantly protected against transmedial elastin degradation & ectasia in the atherosclerotic media. 15047130_Limb ischemia causes activation of MMP-2 and -9, which is eliminated by neutrophil depletion. Neutrophils may be an important activator of MMP-2 and the suppliers of MMP-9 in the ischemic hindlimb. 15050894_Phex may control mineralization and removal of hypertrophic chondrocytes and cartilage matrix in growth plate by regulating the synthesis and deposition of certain bone matrix proteins and proteases such as MMP-9 15059974_MMP9 is broadly implicated in the resolution of allergic inflammation, an essential protective mechanism that overlaps with a more limited role played by MMP2 15075248_despite major increases in expression, MMP-2 and MMP-9 are not essential for microvascular remodeling in M. pulmonis-induced chronic airway inflammation. 15075439_MMP-9 is induced after MPTP application in mice and pharmacologic inhibition of MMPs protects against MPTP neurotoxicity. 15156560_data indicates that sGITR-induced activation of PKCdelta and PLD increases MMP-9 secretions in macrophages 15203321_Matrix metalloproteinase-9 production and activation is induced through activation of the Fcepsilon receptor I in mast cells. 15247285_osteopontin-induced MAPK/IkappaBalpha kinase-dependent nuclear factor kappaB-mediated promatrix metalloproteinase-9 activation is regulated by nuclear factor-inducing kinase 15265790_a mechanism for ROS function in Vascular cell adhesion molecule 1 (VCAM-1) activation of endothelial cell MMP2 and MMP9 during VCAM-1-dependent lymphocyte migration 15355971_Cav-1 has a role in mammary tumorigenesis, cell invasiveness and matrix metalloproteinase (MMP-2/9) secretion 15364410_A statistically significant elevation of MMP9 gelatinolytic activity is observed at days 21 and 180 during experimental herpes simplex virus encephalitis, which may contribute to ongoing brain damage. 15390331_Total MMP activity in culture supernatants was greater after preservation in wild-type than in MMP9 knockout mice. 15449178_results show that MMP-9 was associated with eosinophilic meningitis caused by Angiostrongylus cantonensis, and that the enzyme may be a useful marker for angiostrongyliasis meningitis 15474357_Predominantly upregulated in neurons. Significantly different to shams at 24- and 72-h reperfusion after ischemia. Role for matrix metalloproteinases in contributing to neuronal injury after ischemia. 15474489_MMP-9 activity modulated by HA through NF-kappaB signaling pathway may be involved in the aggressive behavior of lymphoma 15502710_Urine from mice whose livers were inhibited from regenerating contained increased levels of the gelatinases MMP-2 and MMP-9. 15522913_An increase in MMP-9 gene expression and activity contribute to early onset and severe experimental autoimmune encephalomyelitis in MMP-2 knockout mice. 15529013_a deficiency of MMP-9 gene in mutant mice increases collagenase-induced hemorrhage and the resulting brain injury 15545515_Both MMP2 and MMP-9 play some role in elastin calcification and are required to work in concert for initiation and progression of elastin calcification and degeneration in a CaCl(2) aortic injury model. 15631996_Deficiency of MMP-9 has an intermediate effect on pressure overload-induced Left ventricular hypertrophy and in heart failure. 15632000_Increased MMP-9 activity on the ocular surface in response to dryness disrupts corneal epithelial barrier function. 15634906_MMP-9 expression in macrophages mediates degradation of collagen type IV and facilitates macrophage migration from the thymus after clearance of apoptotic thymocytes. 15645290_results suggest that MMP-9 may be associated with the inflammatory reaction to larval toxocariasis during early migration 15652414_Pro-MMP-9 levels are elevated in sera and extracts of damaged nerve and muscle following nerve crush, while active MMP-9 is only elevated in the sera of axotomized mice. 15654515_tumor necrosis factor-alpha and interferon-gamma may promote the MMP-9 activity in local inflammatory cells 15728252_Alpha3beta1 integrin regulates MMP-9 mRNA stability in immortalized keratinocytes 15734845_Role of MMP2 and MMP9 in the formation and function of pancreatic islets in double knockout mice. 15746435_MMP-9 is derived from resident arterial cells and is required for early atherosclerotic plaque development and cellular accumulation in apoE-/- mice 15764676_Leukocytes, most likely neutrophils, are key cellular source of MMP-9, which promotes leukocyte recruitment, causes BBB breakdown secondary to microvascular basal lamina proteolysis, and contributes to neuronal injury after transient focal stroke. 15772259_MMP9 activity in the preimplantation uterus originates in proMMP9-bearing neutrophils attracted by seminal plasma. 15800063_gal3 is a downstream regulator of Mmp9 function during endochondral bone formation. 15841177_Data show that a plasminogen cascade synergizes with MMP-9/gelatinase B in vivo during dermal-epidermal separation in an experimental model of bullous pemphigoid. 15848174_NF-kappaB-dependent MMP-9 plays a key role in Platelet-activating factor-induced angiogenesis 15862156_MMP-2 and MMP-9 expression by TRAP positive mononuclear and multinucleated cells are involved in articular cartilage destruction in CIA. 15947259_focal VEGF and angiopoietin-2 hyperstimulation in the mouse brain increases MMP-9 activity and decreases ZO-1 protein 15969627_From studies with knockout mice, MMP-9 is not required for erythropoietin + granulocyte colony sstimulatin factor stem cell mobilization. 16000631_MMP-9 degrades the extracellular matrix protein laminin and that this degradation induces neuronal apoptosis in a transient focal cerebral ischemia model in mice. 16051896_tPA upregulates brain MMP-9 levels in stroke in vivo 16054858_These data suggest that elevated leukocyte matrix metalloproteinase-9 may contribute to the pathogenesis of type I diabetes and its associated complications. 16081621_The results suggest that impaired cardiac function in volume overload is associated with EE apoptosis, cardiac remodeling, and endothelium-myocytes uncoupling in response to MMP-9 activation. 16126817_MMP-9 deletion stimulated, rather than impaired, neovascularization in remodeling myocardium. 16159824_MMP-9 gene deletion attenuated thoracic aortic aneurysm formation despite an increase in the zymographic levels of MMP-2. These unique findings suggest that an interaction between these 2 MMPs is necessary to facilitate TAA progression. 16166639_Runx2 mediates transactivation of the MMP9 promoter in osteoblasts 16244581_study demonstrated that the growth of primary tumor is distinctly accompanied by an increase of tumor hypoxia level which positively correlates both with the activity of MMP-2 and -9 in primary tumor and metastatic efficiency 16297636_These data suggest that TNFalpha lies upstream of MMP-9 in the pathway of macrophage recruitment to injured peripheral nerve. 16303771_tPA acts as a cytokine that binds to the cell membrane receptor LRP-1, induces its tyrosine phosphorylation, and triggers intracellular signal transduction to induce MMP9 gene expression 16306430_differential effect of MMP-9 at various stages of plaque progression in atherogenesis 16338931_cyclooxygenase-2-dependent extracellular matrix-induced matrix metalloproteinase-9 expression by macrophages is blocked through prostaglandin E2 receptors 16344067_Data suggest that MMP-9 expressed by epithelial cells may play an important role in the development of colitis by modulating cell-matrix interaction and wound healing. 16374516_Macrophage proteolytic activity can induce acute plaque disruption and highlight MMP-9 as a potential therapeutic target for stabilizing rupture-prone plaques. 16458990_MMP-9 contributes to the pathogenesis of brain extravasation and edema in in fulminant hepatic failure mice. 16481424_The importance of MMP-9 function in modulating hippocampal synaptic physiology and plasticity is underscored by behavioral impairments in hippocampal-dependent memory displayed by MMP-9 null-mutant mice. 16495447_These results reveal a novel function for MMP-9 and -12 in developmental myelination likely through regulating IGF-1 bioavailability. 16517742_MMP-9 is an essential component of an effective host response to E. coli peritonitis 16521043_Mmp9 transcripts were clustered in the inner layer of perichondral mesenchyme, juxtaposed with the terminally differentiated hypertrophic chondrocytes. Later Mmp9-expressing cells were found at the sites of chondrocyte apoptosis. 16530081_Involved in neutrophil infiltration in the course of zymosan-induced experimental peritonitis. 16534014_Estradiol preserves the integrity of ischemic tissue by augmenting the mobilization and incorporation of BM-derived EPCs into sites of neovascularization by eNOS-mediated augmentation of MMP-9 expression in the BM. 16551680_MMP-9 is involved in the early recruitment cascades of neutrophils and CD4+ T cells, promotes neutrophil and T cell transmigration during hepatic ischemia/reperfusion, and is required for motility of interstitially migrating leukocytes. 16607611_Tbeta4 increased several metalloproteinases, including MMP-2 and -9 expression by several-fold over control on day 2 after wounding. 16619037_first demonstration that Cited2 functions as a Smad3/p300-interacting transcriptional co-activator in modulating the expression of MMP9, which could affect tumor cell invasion mediated by TGF-beta 16636068_Low GM1-expressing Lewis lung cancer cell lines showed increased proliferation and invasion, enrichment in the GEM/rafts, and increased secretion of MMP-9 16636500_Pro-MMP-9 (in the aorta), neutrophil gelatinase-associated lipocalin/MMP-9 complex (NGAL-MMP-9, in plasma), and active-MMP-9 protein levels were more abundant in Recs1 KO mice throughout adulthood compared with WT mice. 16650610_Overall, these data point to a specific pathogenetic role of matrix-metalloproteinase-2 and matrix-metalloproteinase-9 in neurological dysfunctions associated with Duchenne muscular dystrophy. 16691505_NIK acts as crucial regulator in MMP-9 activation thereby controlling melanoma cell motility and chemoinvasion 16697230_Deficiency in MMP-9 increases the degree of joint inflammation due to decreased bacterial clearance in Staphylococcus aureus-triggered septic arthritis. 16709900_OPN provides a paracrine signal that augments vascular pro-MMP9 activity, mediated in part via superoxide generation and oxylipid formation 16780951_NF-kappaB-dependent expression of MMP-9 in response to CpG-oligodeoxynucleotides plays an important role in the recruitment of immune cells. 16785994_use in targeting c-jun protein may inhibit cutaneous carcinoma 16787929_MMP-9 can degrade amyloid beta fibril and may contribute to ongoing clearance of plaques from amyloid-laden brains 16816114_treatment with antioxidants reversed the decreased matrix metalloproteinase-9 (MMP-9) expression in response to TNF-alpha in vascular smooth muscle cells. 16825657_Results demonstrate that matrix metalloproteinases-9 may play a role in mediating selected aspects of allergen-induced airway remodeling, but does not play a significant role in mucus expression, smooth muscle thickness, or airway responsiveness. 16835221_Data sh ow that transgelin (SM22) is a novel repressor of 92-kDa type IV collagenase (MMP-9) expression. 16880329_Loss of uPA or MMP activity reduces the cardiac inflammatory response after CVB3 infection, thereby protecting against cardiac injury, dilatation, and failure during CVB3-induced myocarditis. 16887049_MMP-2, MMP-9 and EMMPRIN may play an important role in the development of hyperoxia induced lung injury. 16891410_Data describe two inflammatory cell types that are major sources of MMP-9 in the angiogenic stages of pancreatic islet carcinogenesis that unfold in RIP1-Tag2 transgenic mice. 16942752_MMP-9 activity is suppressed by GD1a as determined by gelatin zymography 16951375_Expression of MMP-9 may partially modulate the development of colitis in MMP-2-deficient mice. 16967517_Collectively, these data suggest that E(2) regulates uterine MMP-9 expression and activity in vivo via a complex mechanism. This estrogen regulation of MMP-9 activity may play an important role in uterine tissue remodeling. 16979597_Thus, MMP-9 gene deletion specifically increased resistance artery dilation endothelium-dependent and eNOS expression. 16982845_MMP-9 is required for recruitment of macrophages and tissue remodeling to allow for the formation of tight, well-organized granulomas. 17010584_MMP-9 expression in the remodeling phase of wound healing in nude mice could be a major component of their ability for scar-free healing 17021183_Thrombin and MMP-9 collaborate to promote neuronal death in culture and in intracerebral hemorrhage (ICH). 17065436_MMP-9 may contribute to extracellular brain amyloid-beta peptide (Abeta) clearance by promoting Abeta catabolism. 17071581_These results directly link the inflammatory response in the heart and extracellular matrix remodeling by the matrix metalloproteinases released by the cardiomyocytes. 17072979_We conclude that dextran sodium sulphate induced colitis is markedly attenuated in animals lacking MMP-9. 17082650_An important effect of matrix metalloproteinase 9 (MMP9) is to differentially modify chemotactic bioactivity through proteolytic processing of proteins present in the allergic airway, enhancing the clearance of lung inflammatory cells. 17136358_These results indicate that the degradation of Meckel's cartilage involves an activation cascade of MMPs that differs from that in endochondral bone formation. 17151782_DPC4 regulates MMP9 and may inhibit the proliferation of colon cancer cell by restraining growth and inducing apoptosis 17187368_These data indicate that MMP-9, the expression of which was regulated by NF-kappaB and AP-1, plays a critical role in PAF-induced enhancement of pulmonary melanoma metastasis. 17202364_MMP-9 activity plays a central role in modulating the clinical course and severity of respiratory tularemia 17235437_Matrix metalloproteinases (MMPs) such as MMP-3 and MMP-9 were up-regulated, as further revealed by the reverse transcriptase-polymerase chain reaction (RT-PCR) and immunohistochemistry assays. 17276429_These observations suggest that phosphatidic acid may play a role in MMP-9 regulation through ERKs/TNF-alpha/TNFRs-dependent signaling pathway. 17301159_This study demonstrates that excessive activation of MMP-9 is deleterious to the immature brain, which is associated with the degree of BBB leakage and inflammation. In contrast, apoptosis does not appear to be a major contributing factor. 17311292_This study demonstrated that proteinase expression, particularly of MMP-9, is modulated by pemphigus vulgaris (PV) serum and associated with PV acantholysis. 17316137_MIF activates the MEK-ERK MAP kinase pathway to induce MMP-9 expression by murine macrophages 17382908_expression of the Neu3 gene led to the inhibition of TNF-alpha-induced matrix metalloproteinase-9 (MMP-9) expression in VSMC. Neu3 gene expression strongly decreased MMP-9 promoter activity in response to TNF-alpha. 17464186_These data demonstrate that CpG oligodeoxynucleotides triggers MMP-9 expression via TLR-9 dependent ERK and p38 MAPK activation followed by NF-kappaB activation. 17484881_MMP-9 regulates goblet cell differentiation in colon. Temporal expression of MMP-9 inversely correlated with MUC-2 expression during maturation of goblet cells. 17508356_Actin polymerization modulates MMP9 activation. 17515450_Absence of MMP-9 activity results in vessel stiffness and increased pulse pressure. MMP-9 activation is associated with beneficial role early on in hypertension by preserving vessel compliance and alleviating blood pressure increase. 17540003_These data suggest that the calcium influx-triggered phospholipase D and acidic sphingomyelinase pathways of acidic extracellular pH induced matrix metalloproteinase-9 expression, at least in part, through nuclear factor-kappaB activation. 17543202_Data show that activation of peroxisome proliferator activated receptor-gamma ameliorates the mitochondrial translocation of NOX-4 and matrix metalloproteinase-9 activation. 17552869_Targeted deletion of MMP-9 attenuates myocardial contractile dysfunction in heart failure. 17561941_PMN-driven neurotoxicity is dependent on MMPs, ROS, and TNF-alpha; these factors regulate PMN release of these soluble factors or interact with one another to mediate PMN-driven neurotoxicity. 17582780_In macrophages, raloxifene significantly reduced MMP-9 secretion in a concentration-dependent manner, without affecting MMP-9 activation. 17588309_Our study supports that the down-regulation of MMP-9 by agmatine runs parallel to the up-regulation of eNOS and the maintenance of functional NO release. 17590163_co-ordinated post-translational modifications of p65 and histone H3 involving phosphorylation and acetylation drive COX-2-dependent transcriptional activation of the MMP-9 gene in response to challenge of macrophages with M. avium. 17596135_plasminogen and MMP-3 are relatively more important than MMP-9 for the increased ICB induced by t-PA treatment of ischemic stroke 17633135_Ang II and AT1-R and AT2-R differentially affect the collagenase and gelatinase MMPs -2 and -9 activities released by cardiac fibroblasts. 17641224_Results show upregulation of MMP-2 and -9 during thoracic aortic aneurysm (TAA) formation in Marfan syndrome. Resulting elastic fiber degeneration with deterioration of the aortic contraction and mechanical properties may explain the pathogenesis of TAA. 17653976_In mouse osteosarcoma cells, MMP-9 is inversely regulated by GD1a at the transcriptional level. 17673717_Data show that vascular endothelial growth factor-induced intracerebral hemorrhage (ICH) is associated with increased MMP-9 expression, and that suppression of MMP-9 by minocycline or pyrrolidine dithiocarbamate attenuates ICH. 17681721_Environmental pollutants affect extracellular matrix turnover by modulating MMP-2/MMP-9/MMP-12. 17682049_Study defines a physiological mechanism for the proteolytic processing of ICAM-5 by MMP-2 and MMP-9 and the importance of the its cleavage on regulation of dendritic spine development. 17686468_The results indicated an increase in MMP-9 activity at sites of BBB disruption exhibiting leukocyte infiltration. 17702637_Findings suggest that increased activity of MMP9 secreted by alveolar macrophages in the lung microenvironment may have an antifibrotic effect and provide a potential mechanism involving IGFBP3 degradation. 17709990_CD147 expression on tumor cells seems not to be crucial for MMP-2, MMP-9 and MT1-MMP induction on tumor-associated stromal cells 17762853_role for alpha3beta1 in augmenting TGFbeta-mediated induction of MMP-9 in immortalized or transformed keratinocytes during skin carcinogenesis 17805326_Although aggressive forms of lymphoma constitutively produce MMP-9, tumor growth and dissemination can occur in total absence of MMP-9. 17826846_findings indicate that resident tissue leukocytes, and among them especially macrophages, constitute an important source of MMP-9 during acute peritoneal inflammation 17880014_In liver ischemia/reperfusion injury, MMP-9 functions in leukocyte recruitment and activation leading to liver damage. 17897957_TAK1-mediated pathways are involved in TNF-alpha-induced MMP-9 production in skeletal muscle cells 17911518_MMP-9 may play a role in recanalization of arteries after coil occlusion and in recurrences after sponge embolization of aneurysms. 17925490_Dendritic cells matured within inflammatory sites require both CCR7 and PGE2-induced MMP-9 for their directional migration to draining lymph nodes 17942699_These results suggest that NO regulation of MMP-9 secreted from macrophages may occur chemically by reactive nitrogen species-mediated protein modification, biologically through soluble guanylyl-cyclase. 18003950_Expression of matrix metalloproteases with elastase activity (matrix metalloproteinase [MMP] 2, MMP9, and MMP12) and their inhibitors in the vaginal wall of nonpregnant, pregnant, and postpartum mice. 18006877_Acrolein, a constituent of cigarette smoke and an endogenous mediator of oxidative stress, increases Mmp9 transcripts, protein, and activity in Chronic obstructive pulmonary disease (COPD). 18007984_Mmp9 limits O(3)-induced airway injury possibly via posttranscriptional effects on proinflammatory CXC chemokines including KC and MIP-2. 18023394_MMP-9 is involved in pathogenesis of urogenital Chlamydia muridarum infection in mice possibly by amplifying inflammatory responses. 18030366_a major role for host-derived MMP-9 in angiogenesis and in the growth of human pancreatic cancer in the pancreas of nude mice. 18065684_A novel high-throughput screening system identifies a small molecule repressive for Mmp9. 18069628_The combined use of doxycycline and endostatin in melanoma can inhibit the expression of MMP2 and MMP9. 18097625_These results suggest that the tumor burden may be activating various signaling pathways within splenic T lymphocytes to upregulate MMP-9 expression. 18164515_The spatial and temporal association between increased EMMPRIN expression and elevated MMP-9 levels suggest that EMMPRIN may modulate MMP-9 activity, and participate in neurovascular remodeling after ischemic stroke. 18181170_The molecular mechanisms by which P450 epoxygenase gene transfection or epoxyeicosatrienoic acids supplementation attenuate homocysteine (Hcy)-induced MMP-9 activation, is determined. 18192840_Enhanced mechanical stretch in renovascular hypertension induces EPC mobilization in a p47phox-dependent manner, involving bone marrow SDF-1 and MMP-9 which may contribute to compensatory vascular adaptation in renovascular hypertension. 18222481_Urinary MMP-9 activity was elevated during hepatic regeneration, and decreased on postoperative day 8 when the liver returned to its preoperative mass 18223162_MMP-9 protects against ventilator-induced lung injury by decreasing alveolar neutrophilic infiltration, probably by modulation of the cytokine response in the air spaces. 18243551_blood nerve barrier disruption and MMP-9 induction are differentially regulated in the peripheral and central nervous systems after injuries 18264108_After nerve injury, MMP-9 induces neuropathic pain through interleukin-1beta cleavage and microglial activation at early times, whereas MMP-2 maintains neuropathic pain through interleukin-1beta cleavage and astrocyte activation at later times 18298469_the role of melatonin in arresting peritoneal endometriosis in mice and a novel marker, expression ratio of proMMP-9 versus TIMP-1, was identified for assessing severity and progression of endometriosis. 18311803_TNFalpha up-regulates expression of MMP-9, an important proteinase responsible for extracellular matrix breakdown, leading to coronary artery damage in this model of Kawasaki disease. 18328424_Tumors were unable to grow in MMP-9 knockout mice, but tumor growth could be restored by transplantation of wild-type bone marrow cells. 18328425_MMP-9 activity of bone marrow-derived CD45+ cells is essential and sufficient to initiate glioblastoma angiogenesis by increasing VEGF bioavailability. 18332222_Our work suggests that a synaptic pool of MMP-9 is critical for the sequence of events that underlie the development of seizures in animal models of TLE. 18332263_During acute CVB3 infection, MMP-9 appears necessary to halt virus propagation in the heart, promote proper immune infiltration and remodeling, and preserve cardiac output 18337830_Galardin-sensitive metalloproteinases (MMP-3, MMP-9, MMP-13) mediate compensatory fibrinolysis to facilitate wound healing in the absence of plasminogen. 18339771_Both hematopoietic and nonhematopoietic cells contributed to MMP-9 secretion upon poly-(1,6)-beta-d-glucopyranosyl-(1,3)-beta-d-glucopyranose beta-glucan treatment. 18364034_MMP9 and MMP2 were upregulated in astrocyte processes in noradrenaline treated supraoptic hypothalamic nucleus. 18403633_These findings point to a complex cross-talk between VEGF and MMPs in the progression of ovarian tumor and suggest the possibility of using VEGF inhibitors to affect MMP-dependent tumor invasion. 18413157_expression of matrix metalloproteinase-9 (MMP-9) in the kidney of mice with diabetic nephropathy was enhanced, and MMP-9 might be involved in the development of diabetic nephropathy 18445772_SPARC and MMP9 interact to regulate many stages of pancreatic tumor progression including ECM deposition, angiogenesis and metastasis. 18458097_L-arginine decreases the activity of metalloproteinase (MMP)-2 and MMP-9, which are transcriptionally activated by NF-kappaB. 18464248_Stronger Mmp-9 and -13 expression was detected in fracture calluses at day 7 in the juvenile compared to the middle-aged and elderly mice. 18474440_study shows ras/myc SFME cells under inflammatory & infectious conditions increase iNOS mRNA expression, NO production & MMP-9 expression; data also show that simulated inflammatory, infectious & hypoxic conditions i |
ENSG00000100985 |
MMP9 |
51.343849 |
4.5769867937 |
2.194398 |
0.39651756 |
30.186885 |
0.00000003923536951526694560201102010901785810403907817089930176734924316406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000004349586233308196293149429888236223717967732227407395839691162109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
85.598386996049 |
23.8236069823681 |
18.7019082324058 |
4.14262544843948 |
| ENSMUSG00000017969 |
19223 |
Ptgis |
protein_coding |
O35074 |
FUNCTION: Catalyzes the biosynthesis and metabolism of eicosanoids. Catalyzes the isomerization of prostaglandin H2 to prostacyclin (= prostaglandin I2), a potent mediator of vasodilation and inhibitor of platelet aggregation. Additionally, displays dehydratase activity, toward hydroperoxyeicosatetraenoates (HPETEs), especially toward (15S)-hydroperoxy-(5Z,8Z,11Z,13E)-eicosatetraenoate (15(S)-HPETE). {ECO:0000250|UniProtKB:Q16647}. |
Endoplasmic reticulum;Fatty acid biosynthesis;Fatty acid metabolism;Heme;Iron;Isomerase;Lipid biosynthesis;Lipid metabolism;Lyase;Membrane;Metal-binding;Prostaglandin biosynthesis;Prostaglandin metabolism;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:19223; |
caveola [GO:0005901]; cytoplasm [GO:0005737]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; extracellular space [GO:0005615]; nucleus [GO:0005634]; heme binding [GO:0020037]; hydroperoxy icosatetraenoate dehydratase activity [GO:0106256]; iron ion binding [GO:0005506]; monooxygenase activity [GO:0004497]; oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen [GO:0016705]; prostaglandin-I synthase activity [GO:0008116]; apoptotic signaling pathway [GO:0097190]; cellular response to hypoxia [GO:0071456]; cellular response to interleukin-1 [GO:0071347]; cellular response to interleukin-6 [GO:0071354]; decidualization [GO:0046697]; embryo implantation [GO:0007566]; icosanoid metabolic process [GO:0006690]; negative regulation of inflammatory response [GO:0050728]; negative regulation of NF-kappaB transcription factor activity [GO:0032088]; negative regulation of nitric oxide biosynthetic process [GO:0045019]; positive regulation of angiogenesis [GO:0045766]; positive regulation of execution phase of apoptosis [GO:1900119]; positive regulation of peroxisome proliferator activated receptor signaling pathway [GO:0035360]; prostaglandin biosynthetic process [GO:0001516]; response to hypoxia [GO:0001666] |
11782360_Gene transfer of thromboxane A(2) synthase and prostaglandin I(2) synthase antithetically altered tumor angiogenesis and tumor growth. 12664598_an anlysis of the transcription regulation of this gene 16081819_The two major genetic loci Pgis1 and Pgis2 of murine spondylitis are homologous to chromosome regions in human genome, which control ankylosing spondylitis in human patients. 16186418_CD40 ligand might contribute to the initiation and progression of atherosclerosis by increasing activity of prostacyclin synthase. 17065353_Sepiapterin not only reversed the effects of high glucose on both angiotensin II-induced relaxation and PGI(2) release but also abolished high-glucose-enhanced PGIS nitration. 17120018_Alpha(2)-adrenoreceptor activation is required for coupling heart rate to central inspiratory drive during acute hypoxia, and PGI(2) is required to enhance the inhibition of sympathoactivation. 17187424_The results provide strong evidence that the protective effects of COX-2 within the liver are mediated through the production of PGE(2) and PGI(2), which exert anti-inflammatory functions. 17652370_Endothelial PGI synthase-mediated prostacyclin contributes to renal protection against endotoxemia-related acute kidney injury. 17947695_identify PGI(2)-prostaglandin I2 receptor as an important pathway for inhibiting allergic pulmonary inflammation by controlling recruitment of CD4(+) Th2 cells into the inflammatory site 18514659_COX-1 contributes to the enhanced formation of both PGI(2) and TXA(2) in atherosclerosis, and to the development of the disease 18790018_The results establish a role for the IP receptor in protecting pyramidal hippocampal neurons after this global ischemic model and suggest that IP receptor agonists could be developed to prevent delayed pyramidal neuronal cell death. 19138979_PGIS/ IP transgenic animals with lung-specific PPARgamma overexpression also developed fewer lung tumors. 22865892_Cyclooxygenase-2-derived prostacyclin regulates arterial thrombus formation by suppressing tissue factor in a sirtuin-1-dependent-manner. 23042236_Taken together, our in vitro and in vivo findings show that prostacyclin is secreted from neovessels formed as a result of CNS inflammation 26611322_Prostacyclin synthase and prostaglandin E synthase-1 cooperatively exacerbate inflammatory reactions but have opposing effects on carcinogenesis. 33582554_Coordinated action of microsomal prostaglandin E synthase-1 and prostacyclin synthase on contact hypersensitivity. |
ENSG00000124212 |
PTGIS |
445.203641 |
0.4133006040 |
-1.274737 |
0.13922547 |
82.731814 |
0.00000000000000000009397648103948448087904209781300260378619109524523205488090449311044949354254640638828277587890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000036147721191753223905949688806319685516127402907912615631635944168920104857534170150756835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
256.313831171566 |
24.7736551153089 |
620.163214576095 |
43.1905370088464 |
| ENSMUSG00000018126 |
207495 |
Baiap2l2 |
protein_coding |
Q80Y61 |
FUNCTION: Phosphoinositides-binding protein that induces the formation of planar or gently curved membrane structures. Binds to phosphoinositides, including to phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P2) headgroups. There seems to be no clear preference for a specific phosphoinositide. {ECO:0000269|PubMed:21743456}. |
3D-structure;Alternative splicing;Cell junction;Cell membrane;Cytoplasmic vesicle;Lipid-binding;Membrane;Phosphoprotein;Reference proteome;SH3 domain |
|
|
mmu:207495; |
cell-cell contact zone [GO:0044291]; clathrin complex [GO:0071439]; cytoplasmic vesicle membrane [GO:0030659]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; vesicle membrane [GO:0012506]; identical protein binding [GO:0042802]; phospholipid binding [GO:0005543]; actin crosslink formation [GO:0051764]; actin filament bundle assembly [GO:0051017]; membrane organization [GO:0061024]; plasma membrane organization [GO:0007009]; positive regulation of actin cytoskeleton reorganization [GO:2000251]; positive regulation of actin filament polymerization [GO:0030838] |
21743456_Pinkbar, is specifically expressed in intestinal epithelial cells, where it localizes to Rab13-positive vesicles and to the plasma membrane at intercellular junctions. 33151556_Loss of Baiap2l2 destabilizes the transducing stereocilia of cochlear hair cells and leads to deafness. 34346063_BAIAP2L2 is required for the maintenance of mechanotransducing stereocilia of cochlear hair cells. 35044843_Ca(2+) entry through mechanotransduction channels localizes BAIAP2L2 to stereocilia tips. 36520027_HNF1beta-associated cyst development and electrolyte disturbances are not explained by BAIAP2L2 expression. |
ENSG00000128298 |
BAIAP2L2 |
37.748528 |
0.0397014513 |
-4.654664 |
0.65551041 |
54.329256 |
0.00000000000016955853538033337336919884626991609962711957115821803654398536309599876403808593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000037054743341653340900497747458523973857619226990323113568592816591262817382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.55833327389363 |
1.20282889450088 |
64.4392884210222 |
16.6233137750514 |
| ENSMUSG00000018362 |
16647 |
Kpna2 |
protein_coding |
P52293 |
FUNCTION: Functions in nuclear protein import as an adapter protein for nuclear receptor KPNB1. Binds specifically and directly to substrates containing either a simple or bipartite NLS motif. Docking of the importin/substrate complex to the nuclear pore complex (NPC) is mediated by KPNB1 through binding to nucleoporin FxFG repeats and the complex is subsequently translocated through the pore by an energy requiring, Ran-dependent mechanism. At the nucleoplasmic side of the NPC, Ran binds to importin-beta and the three components separate and importin-alpha and -beta are re-exported from the nucleus to the cytoplasm where GTP hydrolysis releases Ran from importin. The directionality of nuclear import is thought to be conferred by an asymmetric distribution of the GTP- and GDP-bound forms of Ran between the cytoplasm and nucleus. |
3D-structure;Acetylation;Cytoplasm;Nucleus;Phosphoprotein;Protein transport;Reference proteome;Repeat;Transport |
|
|
mmu:16647; |
cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; glutamatergic synapse [GO:0098978]; host cell [GO:0043657]; NLS-dependent protein nuclear import complex [GO:0042564]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; postsynaptic density [GO:0014069]; DNA-binding transcription factor binding [GO:0140297]; histone deacetylase binding [GO:0042826]; nuclear import signal receptor activity [GO:0061608]; nuclear localization sequence binding [GO:0008139]; entry of viral genome into host nucleus through nuclear pore complex via importin [GO:0075506]; NLS-bearing protein import into nucleus [GO:0006607]; positive regulation of viral life cycle [GO:1903902]; postsynapse to nucleus signaling pathway [GO:0099527]; protein import into nucleus [GO:0006606] |
11988093_Karyopherin alpha2: a control step of glucose-sensitive gene expression in hepatic cells 15689618_Results suggest that the importin alpha/beta system is involved in nuclear entry of mammalian clock components Cry2 and Per2, which is indispensable to transcriptional oscillation of clock genes 20406804_Study identified the sequences KKKRR, KKKRK, and KKRKK as the optimal sequences for binding to this site for mouse importin-alpha2, human importin-alpha1, and human importin-alpha5, respectively. 20818336_Data show that MRTF-A contains an unusually long bipartite nuclear localisation signal embedded within the RPEL domain, that uses the importin (Imp)alpha/beta-dependent import pathway, and that import is inhibited by G-actin. 21664354_The results suggest that Klf2 and Klf4 function redundantly to drive high level of Kpna2 expression in ES cells. 21741962_knockdown of KPNA2 decreased myotube growth. 21840941_Overexpression of a dominant-negative IMPalpha2 isoform, when assessed against adjacent untransfected or IMPalpha2 transfected cells, led to both a significant reduction in endogenous Oct3/4 protein levels and inhibition of Oct3/4 nuclear localization. 21900684_Chrp, Arip3, and Hop2 are binding partners of IMPalpha2 in the developmental context of germ line development. 21964068_results collectively reveal that nuclear-localized importin alpha2 influences gene expression and contributes directly to cell fate outcomes including non-apoptotic cell death. 22510057_importin alpha binds to Nup153 23906064_study reports a cell-fate determination mechanism in which importin alpha2 negatively regulates the nuclear import of certain transcription factors to maintain embryonic stem cell properties 24930886_a significant correlation of KPNA2 expression and tumour aggressiveness in a large variety of other solid tumour entities 25389311_constitutive expression of Kpna2 during the differentiation culture of ESCs significantly impairs clock development, and KPNA2 facilitates cytoplasmic localization of PER1/ 25694451_Specific interaction with the nuclear transporter importin alpha2 can modulate paraspeckle protein 1 delivery to nuclear paraspeckles. 26456934_Structure of importin-alpha bound to a non-classical nuclear localization signal of the influenza A virus nucleoprotein has been reported. 26553592_provided support for a link between autophagy and epithelial-to-mesenchymal (-like) transition status in WT TP53 glioblastoma cells and provided evidence for the signaling pathway (MIR517C-KPNA2-cytoplasmic TP53) involved in attenuating autophagy 26844761_mTORC1 positively regulated the importer protein KPNA2, which participated in glycolysis regulation downstream of mTORC1 in a HIF1alpha-independent manner, indicating that mTORC1 regulates glycolysis through multiple ways. 27107455_Results demonstrated that radiation-induced dying colorectal cancer cells released considerable amounts of KPNA2 that induce the maturation and activation of DCs for synergistic antitumor effect of radiation. 31179341_upregulation of KPNA2 seemed to be correlated with progression of the degree of atypia in pancreatic ductal cells in aged diabetic C414A-mutant-CRY1 transgenic mice 31597697_NLRP3 interacted with Kpna2 and translocated to the nucleus from the cytoplasm under regulatory T cells-polarizing conditions. 32130408_USP22 promotes IRF3 nuclear translocation and antiviral responses by deubiquitinating the importin protein KPNA2. 34519142_Importin alpha2 association with chromatin: Direct DNA binding via a novel DNA-binding domain. 36578083_KPNA2 promotes angiogenesis by regulating STAT3 phosphorylation. |
ENSG00000182481 |
KPNA2 |
3319.829167 |
2.0814226356 |
1.057570 |
0.05823575 |
325.232194 |
0.00000000000000000000000000000000000000000000000000000000000000000000000105013343850343963100205893784768080596432468334560130772994674546647373685719078998554969782210449283601903848774544218571975905945434918948457977430824287024511026393089300362028437607619935 |
0.0000000000000000000000000000000000000000000000000000000000000000000003403326071337104942884468119468130161125833956909264768212656598327394050589338123105915943295315855405059962147818500855933932045865392218673113372121204815769663653388815184597238072683467 |
Yes |
No |
4512.56162314524 |
182.203285714249 |
2168.01794402484 |
66.8958932620101 |
| ENSMUSG00000018509 |
73139 |
Cenpv |
protein_coding |
Q9CXS4 |
FUNCTION: Required for distribution of pericentromeric heterochromatin in interphase nuclei and for centromere formation and organization, chromosome alignment and cytokinesis. {ECO:0000250}. |
Alternative splicing;Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Cytoskeleton;Kinetochore;Metal-binding;Methylation;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Zinc |
|
|
mmu:73139; |
cytosol [GO:0005829]; kinetochore [GO:0000776]; microtubule cytoskeleton [GO:0015630]; midbody [GO:0030496]; nuclear membrane [GO:0031965]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle midzone [GO:0051233]; carbon-sulfur lyase activity [GO:0016846]; metal ion binding [GO:0046872]; ameboidal-type cell migration [GO:0001667]; cell cycle [GO:0007049]; cell division [GO:0051301]; centromere complex assembly [GO:0034508]; pericentric heterochromatin formation [GO:0031508]; positive regulation of cytokinesis [GO:0032467]; regulation of chromosome organization [GO:0033044] |
Human_homologues 18772885_Results suggest that CENP-V (nuclear protein p30) is required for centromere organization, chromosome alignment and cytokinesis in human cells |
ENSG00000166582 |
CENPV |
250.628668 |
0.3501951372 |
-1.513769 |
0.18177992 |
68.642735 |
0.00000000000000011801871135566787203972747015993459779434420723210480419496093418274540454149246215820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000034805508700422971109321582083293995938451815402858713355271902401000261306762695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
130.191393176581 |
16.4475865578238 |
371.768135421986 |
33.4300834392412 |
| ENSMUSG00000018752 |
619441 |
Tnfsfm13 |
protein_coding |
B0QZJ0 |
|
Cytokine;Reference proteome |
|
This gene represents read-through transcription between neighboring genes encoding tumor necrosis factor family members. This results a hybrid protein composed of the cytoplasmic and transmembrane domains of family member 12 fused to the C-terminal domain of family member 13. The homologous fusion protein in human is known to be membrane-anchored and presents the receptor-binding domain of family member 13 at the cell surface. It stimulates cycling in T- and B-lymphoma cell lines. Alternative splicing of this mouse gene results in multiple transcript variants. [provided by RefSeq, Oct 2010]. |
|
extracellular space [GO:0005615]; membrane [GO:0016020]; cytokine activity [GO:0005125]; receptor ligand activity [GO:0048018]; tumor necrosis factor receptor binding [GO:0005164]; immune response [GO:0006955]; positive regulation of isotype switching to IgA isotypes [GO:0048298] |
30337376_the first and physiologically relevant role for TWE-PRIL is reported and suggests that it mediates reverse signalling. |
|
|
114.887574 |
2.6075226486 |
1.382680 |
0.31910111 |
18.473186 |
0.00001723116647049349918550779914294679429076495580375194549560546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001222473813127885515841808405923529790015891194343566894531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
174.191103781676 |
38.8432102484971 |
66.8032946416984 |
11.3723991890483 |
| ENSMUSG00000018819 |
16985 |
Lsp1 |
protein_coding |
P19973 |
FUNCTION: May play a role in mediating neutrophil activation and chemotaxis. {ECO:0000269|PubMed:17481585}. |
Acetylation;Alternative splicing;Cell membrane;Direct protein sequencing;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:16985; |
plasma membrane [GO:0005886]; actin binding [GO:0003779]; apoptotic process [GO:0006915]; cellular response to interleukin-7 [GO:0098761]; chemotaxis [GO:0006935]; cytoskeleton organization [GO:0007010]; defense response [GO:0006952]; signal transduction [GO:0007165] |
12077272_Lsp1 negatively regulates fMLP-induced polarization and chemotaxis of neutrophils during inflammation through its function on adhesion via specific integrins, such as Mac-1 (CD11b/CD18). 17003510_The results show a complex function of LSP1 in regulating leukocyte recruitment to inflamed sites. 17481585_MK2-regulated LSP1 phosphorylation is involved in stabilization of F-actin polarization during neutrophil chemotaxis. 18040084_These results demonstrate that the absence of LSP1 promotes healing of skin wounds. 18580965_the absence of LSP1 promotes fibrosis in the skin 21030556_Data demonstrate that endothelial LSP1 is recruited to the cytoskeleton in inflammation and plays an important role in forming endothelial domes thereby regulating neutrophil transendothelial migration. 22683630_LSP1 plays an important role in leukocyte recruitment induced by both KC and MIP-2. 23446150_TRAF1 and LSP1 cooperate downstream of 4-1BB to activate ERK signaling and down-modulate the levels of Bim leading to enhanced T cell survival. 23447036_Endothelial LSP1 participates in leukocyte adhesion in vitro, and leukocyte adhesion, through ICAM-1, fosters the activation of endothelial LSP1. 25234543_functions as a negative regulator of proliferation and migration in liver 25717183_the LSP1-myosin1e bimolecular complex plays a pivotal role in the regulation of actin cytoskeleton remodeling during Fc gamma receptor-driven phagocytosis. 26238489_Data indicate that in lymphocyte specific 1 protein (LSP1)-deficient mice, PECAM-1 expression was reduced in endothelial cells, but not in neutrophils. 26554018_the importance of LSP1 Copy number variations and LSP1 insufficiency in the pathogenesis of rheumatoid arthritis, is reported. 27555448_Regulation of O-GlcNAcylation and phosphorylation of lymphocyte-specific protein-1 (Lsp1) is a key switch that triggers apoptosis in activated B cells. O-GlcNAcylation at S209 of Lsp1 is a prerequisite for the recruitment of its kinase, PKC-beta1, to induce S243 phosphorylation. 33470457_LSP1-myosin1e bimolecular complex regulates focal adhesion dynamics and cell migration. 35390317_Lymphocyte-Specific Protein-1 Suppresses Xenobiotic-Induced Constitutive Androstane Receptor and Subsequent Yes-Associated Protein-Activated Hepatocyte Proliferation. |
ENSG00000130592 |
LSP1 |
640.294348 |
0.3917653178 |
-1.351938 |
0.10934617 |
151.326617 |
0.00000000000000000000000000000000008892032114497615552141770769706530065712502794216628689165716498588441759079615505806810362918191081860186386620625853538513183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000081592429619293780466457950718310904964989414254436087799527436795788472769765732909783007009707489487482234835624694824218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
341.16103023178 |
25.8823784954442 |
870.830098414432 |
47.3543222972063 |
| ENSMUSG00000018983 |
242705 |
E2f2 |
protein_coding |
P56931 |
FUNCTION: Transcription activator that binds DNA cooperatively with DP proteins through the E2 recognition site, 5'-TTTC[CG]CGC-3' found in the promoter region of a number of genes whose products are involved in cell cycle regulation or in DNA replication. The DRTF1/E2F complex functions in the control of cell-cycle progression from g1 to s phase. E2F2 binds specifically to RB1 in a cell-cycle dependent manner. |
Activator;Cell cycle;DNA-binding;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:242705; |
nucleus [GO:0005634]; Rb-E2F complex [GO:0035189]; RNA polymerase II transcription regulator complex [GO:0090575]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; protein dimerization activity [GO:0046983]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; intrinsic apoptotic signaling pathway by p53 class mediator [GO:0072332]; lens fiber cell apoptotic process [GO:1990086]; negative regulation of DNA-binding transcription factor activity [GO:0043433]; negative regulation of sprouting angiogenesis [GO:1903671]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of cell cycle [GO:0051726]; regulation of DNA-templated transcription [GO:0006355]; regulation of transcription by RNA polymerase II [GO:0006357]; regulation of transcription involved in G1/S transition of mitotic cell cycle [GO:0000083] |
11713289_E2F1 and E2F2 determine thresholds for antigen-induced T-cell proliferation and suppress tumorigenesis 11719808_The E2F1-3 transcription factors are essential for cellular proliferation 11754817_Mutation of E2F2 in mice causes enhanced T lymphocyte proliferation, leading to the development of autoimmunity 12724419_E2F1 and E2F2 play essential and redundant roles in the proper coordination of cell cycle progression with differentiation which is necessary for efficient hematopoiesis 14566047_role in postnatal pancreas development and homeostatic maintenance 14684733_E2F2 promotes unscheduled cell division and oncogenic transformation of thymic epithelial cells 15146237_E2F1/E2F2 compound-mutant mice develop nonautoimmune insulin-deficient diabetes and exocrine pancreatic dysfunction 16002659_A role is indicated for E2F2 in promoting T cell proliferation in response to T cell receptor (TCR) stimuli in the context of homeostatic signaling in vivo. 17008321_the negative regulation of the p53-p21(CIP1) axis by the E2F1-3 factors is critical for cell cycle progression and cellular transformation 17167174_a critical function of the E2F1, E2F2, and E2F3 activators is in the control of a p53-dependent axis that indirectly regulates E2F-mediated transcriptional repression and cellular proliferation 17855529_T-antigen-induced intestinal proliferation is reduced in mice lacking either E2F2 alone or both E2F2 and E2F3a 17881568_E2F2 has a tumor suppressor function in Myc-induced T cell lymphomagenesis 17923680_These results implicate deregulated E2f-2 in the cell cycle defects observed in Rb null erythroblasts and reveal a novel role for E2f-2 during terminal red blood cell differentiation. 18322175_E2F4 is important in early stages of commitment to the lymphoid lineage. 18852285_Results suggest a model in which Erythroid Kruppel-like factor-dependent activation and modification of the E2f2 locus is required for cell cycle progression preceding terminal erythroid differentiation. 19066456_E2F2 Transcription Factor functions to transcriptionally repress cell cycle genes to establish the G(0) state. 19142862_E2F2 and E2F3 had positive effects on the promoter activity of fibrillin-2 in chondrocyte differentiation 19457859_Direct regulation of the E2f2 enhancer is a generic mechanism by which many Kruppel-like factors regulate proliferation and differentiation. 19513100_GATA-1, the master transcription factor of erythropoiesis, forms a tricomplex with the retinoblastoma protein (pRb) and E2F-2. 19749980_The absence of E2F2 substantially accelerates Myc-induced lymphomagenesis. 19752322_results identify a cell-cycle-independent mechanism by which E2F2 regulates endothelial function, arterial contractility, and blood pressure 19798698_findings demonstrate that E2F2 can function as a tumor suppressor in epithelial tissues, perhaps by limiting proliferation in response to Myc 20016601_loss of activating E2fs caused downregulation of the p53 deacetylase Sirt1, p53 hyperacetylation and elevated apoptosis, establishing a novel E2f-Sirt1-p53 survival axis in vivo 20016602_In differentiating cells E2f1-3 function in a complex with Rb as repressors to silence E2f targets and facilitate exit from the cell cycle 20187704_we have examined the effect that a simultaneous inactivation of the E2F2 gene and over-expression of the Bcl-2 gene in B cells has on lymphoid homeostasis and autoimmunity 20404092_Control of miRNAs by E2F is likely to play multiple roles in cell proliferation and in proliferative diseases such as cancer. 20573986_loss of E2F2 results in a deregulated Aryl-hydrocarbon-receptor pathway 20676136_Findings show that rapid proliferation of E2F1/E2F2 compound mutant cultures is temporally followed by induction of a DNA damage response and the implementation of a p21(CIP1)-dependent senescence. 21115501_functions for E2f1-3 at distinct stages of myeloid development in vivo, first as repressors in cell survival and then as activators in cell proliferation. 21185283_E2F1-3 in mediating transcriptional repression by retinoblastoma tumor suppressor during cell cycle exit and point to a critical role for their repressive functions in cell survival. 21527726_these results indicate that the transcriptional activity of E2F2 contributes to promote adult hepatocyte proliferation and liver regeneration. 23558950_E2f2 induces cone photoreceptor apoptosis independent of E2f1 and E2f3 in mice. 23603666_E2F2 impairs, and endothelial E2F3 promotes, the angiogenic response to peripheral ischemic injury through corresponding changes in EC cell-cycle progression. 23799044_E2F3 promotes while E2F2 suppresses ischemic cardiac repair through corresponding changes in endothelial cell proliferation. 25088905_showed that the D326V missense pRb bound to E2F1 but failed to interact with E2F2/3 25396754_E2F2 activity sustains the hepatic homeostasis of major membrane glycerolipid components while it is dispensable for storage glycerolipid balance. 25656653_A role for E2F1 and E2F2 as suppressors of replicative stress in differentiating cells, and the existence of a robust E2F-p53 regulatory axis in tissue homeostasis enabling and tumorigenesis preventing is shown. 25892555_E2F2 accumulates at sites of oxidative and UV-induced DNA damage, and interact with gammaH2AX DNA repair factor. 26192440_Rb-deficient cells hijack and redeploy Myc and E2f3 from an S-G2 program essential for normal cell cycles to a G1-S program that re-engages ectopic cell cycles, exposing an unanticipated addiction of Rb-null cells on Myc. 26474282_E2F2 loss results in increased lung metastasis in breast cancer, potentially functioning through a PTPRD dependent mechanism. 26505089_Spinal cord injury-induced activation of E2F1-2 mediates cell cycle activation, contributing to gliopathy and neuronal/tissue loss associated with motor impairments and post-traumatic hyperesthesia. 27795297_Findings implicate E2F-2 in regulating the expression of mitotic kinases that are adapted to perform specialized functions in nuclear condensation and enucleation of maturing erythroblasts. 28558060_Genomic structure, expression pattern, and functional characterization of transcription factor E2F-2 from black tiger 30401820_E2F1 and/or E2F2 likely play only a modest role in neuronal cell loss after brain trauma. 30679264_E2F2 and CCND2 were identified as the targets of let-7i-5p, mediating its effect in regulating the cell cycle of cardiomyocyte. 31827131_Defined factors to reactivate cell cycle activity in adult mouse cardiomyocytes. 35008734_E2f2 Attenuates Apoptosis of Activated T Lymphocytes and Protects from Immune-Mediated Injury through Repression of Fas and FasL. 36291740_Molecular Regulation of Heme Oxygenase-1 Expression by E2F Transcription Factor 2 in Lung Fibroblast Cells: Relevance to Idiopathic Pulmonary Fibrosis. |
ENSG00000007968 |
E2F2 |
163.404114 |
2.3299039116 |
1.220270 |
0.21632122 |
31.421530 |
0.00000002076653366736203236844034646872009242102308235189411789178848266601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000002383691340024555136849508316099210958327603293582797050476074218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
223.089416339305 |
33.5760561173622 |
95.7504793329027 |
11.0956051189049 |
| ENSMUSG00000019230 |
16876 |
Lhx9 |
protein_coding |
Q9WUH2 |
FUNCTION: Involved in gonadal development. {ECO:0000269|PubMed:10706291}. |
Alternative splicing;DNA-binding;Homeobox;LIM domain;Metal-binding;Nucleus;Reference proteome;Repeat;Zinc |
|
|
mmu:16876; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific double-stranded DNA binding [GO:1990837]; cell population proliferation [GO:0008283]; dorsal spinal cord interneuron anterior axon guidance [GO:0097380]; female gonad development [GO:0008585]; gonad morphogenesis [GO:0035262]; male gonad development [GO:0008584]; negative regulation of DNA-templated transcription [GO:0045892]; neuron differentiation [GO:0030182]; regulation of transcription by RNA polymerase II [GO:0006357] |
12130543_a pathway in which the products of the Wt1 and Lhx9 genes activate expression of Sf1 and thus mediate early gonadogenesis 18577233_the Lhx9 gene represents the first direct target of the GATA4/FOG2 repressor complex in cardiac development. 18701067_Lhx2/9 regulate directly the expression of Rig-1. And expressed by a set of commissural relay neurons and are required for the this neuron axon projection. 18942141_The second wave of expression at E12.5 includes Lhx3, 5, 9, Isl2, and Lmx1b in the differentiating sensory epithelia with cellular specificities. 23136391_Gbx2 regulates thalamocortical axon guidance by modifying the LIM and Robo codes. 24647753_Study has revealed that Lhx9 plays a role in the development of the pineal gland and in maintenance of the brain structure. 25112520_Lhx9-GCE mouse line could serve as a beneficial tool for lineage tracing and gene manipulation experiments. 26220830_Authors examined the expression pattern for the three alternative splice forms of Lhx9; Lhx9alpha, Lhx9beta and Lhx9c during early limb development. 28456934_Lhx9 is a new molecular marker that defines a subset of amacrine cells and show that it is necessary for the development of the Nitric Oxide-Synthesizing Amacrine Cell subtype of amacrine cells. 29565736_that Smurf1 promotes Lhx9 ubiquitylation and is involved in testosterone production in Leydig cells directly 33515280_Development of the mouse anterior amygdalar radial unit marked by Lhx9-expression. 35247020_Alternative LIM homeodomain splice variants are dynamically regulated at key developmental steps in vertebrates. |
ENSG00000143355 |
LHX9 |
22.649698 |
3.4334369496 |
1.779653 |
0.55739742 |
10.022468 |
0.00154642130557916404956508849011243000859394669532775878906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0067980055776570924921942129515173292020335793495178222656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.6237387729384 |
12.9203897690149 |
9.50177307804453 |
2.94479840379082 |
| ENSMUSG00000019278 |
13479 |
Dpep1 |
protein_coding |
P31428 |
FUNCTION: Hydrolyzes a wide range of dipeptides including the conversion of leukotriene D4 to leukotriene E4 (PubMed:12738806, PubMed:31442408, PubMed:9560193). Hydrolyzes cystinyl-bis-glycine (cys-bis-gly) formed during glutathione degradation (PubMed:12738806, PubMed:9560193). Possesses also beta lactamase activity and hydrolytically inactivates beta-lactam antibiotics (PubMed:12738806). {ECO:0000269|PubMed:12738806, ECO:0000269|PubMed:31442408, ECO:0000269|PubMed:9560193}.; FUNCTION: Independently of its dipeptidase activity, acts as an adhesion receptor for neutrophil recruitment from bloodstream into inflamed lungs and liver. {ECO:0000269|PubMed:31442408}. |
Cell membrane;Cell projection;Dipeptidase;Disulfide bond;Glycoprotein;GPI-anchor;Hydrolase;Lipid metabolism;Lipoprotein;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Signal;Zinc |
|
|
mmu:13479; |
apical part of cell [GO:0045177]; apical plasma membrane [GO:0016324]; cell junction [GO:0030054]; extracellular space [GO:0005615]; microvillus membrane [GO:0031528]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; beta-lactamase activity [GO:0008800]; cysteine-type endopeptidase inhibitor activity involved in apoptotic process [GO:0043027]; dipeptidase activity [GO:0016805]; GPI anchor binding [GO:0034235]; metallodipeptidase activity [GO:0070573]; modified amino acid binding [GO:0072341]; zinc ion binding [GO:0008270]; antibiotic metabolic process [GO:0016999]; cellular response to calcium ion [GO:0071277]; cellular response to nitric oxide [GO:0071732]; cellular response to xenobiotic stimulus [GO:0071466]; glutathione catabolic process [GO:0006751]; homocysteine metabolic process [GO:0050667]; inflammatory response [GO:0006954]; leukotriene D4 catabolic process [GO:1901749]; leukotriene metabolic process [GO:0006691]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cell migration [GO:0030336]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043154]; neutrophil chemotaxis [GO:0030593]; proteolysis [GO:0006508] |
29863495_Contrast-induced acute kidney injury is a multistep process that involves immune surveillance by resident and infiltrating renal phagocytes, Nlrp3-dependent inflammation, and the tubular reabsorption of contrast via dipeptidase-1. 31442408_Genetic ablation or functional peptide blocking of DPEP1 significantly reduced neutrophil recruitment to the lungs and liver and provided improved survival in models of endotoxemia. 34426578_A single genetic locus controls both expression of DPEP1/CHMP1A and kidney disease development via ferroptosis. 35108057_Dipeptidase-1 governs renal inflammation during ischemia reperfusion injury. |
ENSG00000015413 |
DPEP1 |
40.125122 |
0.1784460139 |
-2.486440 |
0.49370873 |
24.647127 |
0.00000068847331190502105116888831651267288691542489686980843544006347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000063135614009255153667062032685208805560250766575336456298828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.6490780401109 |
4.06472956557995 |
65.2806850835654 |
15.6575820577208 |
| ENSMUSG00000019326 |
11754 |
Aoc3 |
protein_coding |
O70423 |
FUNCTION: Cell adhesion protein that participates in lymphocyte recirculation by mediating the binding of lymphocytes to peripheral lymph node vascular endothelial cells in an L-selectin-independent fashion. Has a monoamine oxidase activity. May play a role in adipogenesis (By similarity). {ECO:0000250}. |
Alternative splicing;Calcium;Cell adhesion;Copper;Disulfide bond;Glycoprotein;Membrane;Metal-binding;Oxidoreductase;Reference proteome;Signal-anchor;TPQ;Transmembrane;Transmembrane helix |
|
|
mmu:11754; |
cell surface [GO:0009986]; cytoplasm [GO:0005737]; early endosome [GO:0005769]; endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; microvillus [GO:0005902]; plasma membrane [GO:0005886]; aminoacetone:oxygen oxidoreductase(deaminating) activity [GO:0052594]; calcium ion binding [GO:0005509]; copper ion binding [GO:0005507]; identical protein binding [GO:0042802]; phenethylamine:oxygen oxidoreductase (deaminating) activity [GO:0052596]; primary amine oxidase activity [GO:0008131]; protein heterodimerization activity [GO:0046982]; quinone binding [GO:0048038]; tryptamine:oxygen oxidoreductase (deaminating) activity [GO:0052593]; amine metabolic process [GO:0009308]; cell adhesion [GO:0007155]; eating behavior [GO:0042755]; leukocyte migration involved in inflammatory response [GO:0002523]; negative regulation of primary amine oxidase activity [GO:1902283]; positive regulation of acute inflammatory response [GO:0002675]; positive regulation of glucose transmembrane transport [GO:0010828]; positive regulation of leukocyte migration [GO:0002687]; regulation of blood pressure [GO:0008217]; response to antibiotic [GO:0046677]; response to immobilization stress [GO:0035902] |
12242458_Enzyme is in involved in deamination in atherogenesis in KKAy diabetic mice fed with high cholesterol diet. 12606817_the increased enzyme activity observed in diabetes is not a result of increased SSAO gene transcription. It is controlled by posttranslational modifications of the protein, and the catalytic activity controls the gene expression 12700900_vascular adhesion protein-1 expression may contribute to the functional heterogeneity of endothelial cells within the lung to create distinct sites for the recruitment of inflammatory cells 14968297_Release of this enzyme is regulated by TNF-alpha and insulin and, in adipose cells could contribute to the atherogenesis and vascular dysfunction associated with diabetes and obesity. 15178639_The vascular endothelium is the major source of circulating VAP-1 protein and semicarbazide-sensitive amine oxidase activity. 15743791_VAP-1 mediates leukocyte trafficking to sites of inflammation and thus is a potential target for anti-inflammatory therapies 16111634_T helper type 2 cells use Vap-1 to roll and adhere in the inflamed liver microcirculation 17393060_These data demonstrate the potential clinical benefit of small molecule anti-SSAO therapy in this mouse multiple sclerosis model. 17406965_The effects of exogenous or biogenic amines on glucose transport are not receptor-mediated but are oxidation-dependent. The major SSAO form expressed in mouse adipocytes is encoded by the AOC3 gene. 17947691_VAP-1-deficient mice have mild deviations in the mucosal immune system 18330481_The atheroprotective effect of LXR agonist T0901317 is related to the inhibition of SSAO gene expression and its activity. 18835815_LOX binds to mature TGF-beta1 and enzymatically regulates its signaling in bone via amine oxidase activity, and thus may play an important role in bone maintenance and remodeling 18991279_VAP-1 regulates the inflammatory response in ischemia-reperfusion injury and suggest that blockade of VAP-1 may have therapeutic value. 20012150_histamine moderately activated lipolysis in adipocytes in AOC3 knockout mice 20877383_VAP-1 inhibition provided antiinflammation effect by reducing adhesion molecule expression and immune cell infiltration after intracerebral hemorrhage. 21298297_AOC3 expression is increased in perigonadal and subcutaneous adipose tissue of diabetic db-/- obese mice. 21435467_VAP-1-mediated M2 macrophage infiltration underlies IL-1beta- but not VEGF-A-induced lymph- and angiogenesis 21512782_This review examines the biological functions of VAP-1, especially the role that this molecule might play in the establishment and progression of chronic liver disease. 22238597_The demonstrated AOC3 turnover of primary amines that are non-native to human tissue suggests possible roles for the adipocyte enzyme in subcutaneous bacterial infiltration and obesity. 22480418_Long-term activation of semicarbazide-sensitive amine oxidase by benzylamine lowers circulating levels of uric acid in diabetic conditions. 22547093_The antihyperglycemic effect of benzylamine is abolished by AOC3 gene knockout in mice. 23407548_The results of the study establish VCAM-1 and VAP-1 as mediators of myeloid cell recruitment in metastasis and identify VAP-1 as a potential target for therapeutic intervention to combat early metastasis. 25116373_these results suggest a largely redundant function for AOC3/VAP-1 in allergic inflammatory responses of the airways. 25562318_these results link the amine oxidase activity of VAP-1 with hepatic inflammation and fibrosis and suggest that targeting VAP-1 has therapeutic potential for NAFLD and other chronic fibrotic liver diseases. 25889951_Suggest inhibiting VAP-1/SSAO enzymatic function, by PXS-4728A, as a novel therapeutic approach in lung diseases that are characterized by neutrophilic pattern of inflammation. 27043821_data indicate that SSAO inactivation in vivo stabilizes the established plaques mainly via inducing the switch of SMCs from a contractile to a synthetic phenotype 27731409_VAP-1 was expressed on endothelial cells lining inflamed atherosclerotic lesions; normal vessel walls in aortas of C57BL/6N control mice were VAP-1-negative. 28251930_This study showed that the oxidase activity of AOC3 contributes to the development of lung fibrosis mainly by regulating the accumulation of pathogenic leukocyte subtypes, which drive the fibrotic response. 28318627_VAP-1 plays a critical role in the pathophysiology of renal ischemia/reperfusion injury by enhancement of neutrophil infiltration generating a local hydrogen peroxide gradient. 29166944_Data indicate strong-to-moderate expression of vascular adhesion protein-1 (VAP-1) in the synovium of Borrelia burgdorferi-infected mice, while only weak expression of VAP-1 was detected in uninfected mice. 29490166_The blockade of VAP-1 could be a promising medical modality to combat the acute and chronic variants. 30199530_The results establish a role for Copper homeostasis in adipocyte metabolism and identify SSAO as a regulator of energy utilization processes in adipocytes. 32410850_Comparison of Inhibitor and Substrate Selectivity between Rodent and Human Vascular Adhesion Protein-1. 32712883_Obesity of mice lacking VAP-1/SSAO by Aoc3 gene deletion is reproduced in mice expressing a mutated vascular adhesion protein-1 (VAP-1) devoid of amine oxidase activity. |
ENSG00000131471 |
AOC3 |
161.810664 |
4.0539835747 |
2.019340 |
0.25052757 |
62.883665 |
0.00000000000000219283308311076294829147726859655933892337387712545559992349808453582227230072021484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000577876012490365676101934125821759967521051154504263536182406824082136154174804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
258.186725885037 |
45.0604384404654 |
63.6871662463832 |
8.77360546557685 |
| ENSMUSG00000019773 |
67141 |
Fbxo5 |
protein_coding |
Q7TSG3 |
FUNCTION: Regulator of APC activity during mitotic and meiotic cell cycle (PubMed:17190794, PubMed:15526037, PubMed:16809773). During mitotic cell cycle plays a role as both substrate and inhibitor of APC-FZR1 complex (PubMed:16809773). During G1 phase, plays a role as substrate of APC-FZR1 complex E3 ligase. Then switches as an inhibitor of APC-FZR1 complex during S and G2 leading to cell-cycle commitment. As APC inhibitor, prevents the degradation of APC substrates at multiple levels: by interacting with APC and blocking access of APC substrates to the D-box co-receptor, formed by FZR1 and ANAPC10; by suppressing ubiquitin ligation and chain elongation by APC by preventing the UBE2C and UBE2S activities. Plays a role in genome integrity preservation by coordinating DNA replication with mitosis through APC inhibition in interphase to stabilize CCNA2 and GMNN in order to promote mitosis and prevent rereplication and DNA damage-induced cellular senescence (By similarity). During oocyte maturation, plays a role in meiosis through inactivation of APC-FZR1 complex. Inhibits APC through RPS6KA2 interaction that increases FBXO5 affiniy for CDC20 leading to the metaphase arrest of the second meiotic division before fertilization (PubMed:15526037). Controls entry into the first meiotic division through inactivation of APC-FZR1 complex (PubMed:17190794). Promotes migration and osteogenic differentiation of mesenchymal stem cells (By similarity). {ECO:0000250|UniProtKB:Q9UKT4, ECO:0000269|PubMed:15526037, ECO:0000269|PubMed:16809773, ECO:0000269|PubMed:17190794}. |
Cell cycle;Cell division;Cytoplasm;Cytoskeleton;Metal-binding;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:67141; |
cytoplasm [GO:0005737]; meiotic spindle [GO:0072687]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; anaphase-promoting complex binding [GO:0010997]; metal ion binding [GO:0046872]; molecular function inhibitor activity [GO:0140678]; protein kinase binding [GO:0019901]; ubiquitin ligase inhibitor activity [GO:1990948]; cell division [GO:0051301]; microtubule polymerization [GO:0046785]; negative regulation of cellular senescence [GO:2000773]; negative regulation of DNA endoreduplication [GO:0032876]; negative regulation of meiotic nuclear division [GO:0045835]; negative regulation of mitotic metaphase/anaphase transition [GO:0045841]; negative regulation of response to DNA damage stimulus [GO:2001021]; negative regulation of ubiquitin protein ligase activity [GO:1904667]; negative regulation of ubiquitin-protein transferase activity [GO:0051444]; oocyte maturation [GO:0001556]; positive regulation of biomineral tissue development [GO:0070169]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of G2/M transition of mitotic cell cycle [GO:0010971]; positive regulation of meiosis I [GO:0060903]; positive regulation of mesenchymal stem cell migration [GO:1905322]; positive regulation of osteoblast differentiation [GO:0045669]; protein ubiquitination [GO:0016567]; regulation of DNA replication [GO:0006275]; regulation of meiotic nuclear division [GO:0040020]; regulation of mitotic cell cycle [GO:0007346]; regulation of mitotic nuclear division [GO:0007088]; spindle assembly [GO:0051225]; spindle assembly involved in female meiosis I [GO:0007057]; vesicle organization [GO:0016050] |
Human_homologues 11988738_These data suggest that E2F can activate both transcription of cyclin A and the hEmi1-dependent stabilization of APC(Cdh1) targets, such as cyclin A, to promote S phase entry. 15469984_Plk1 activates the anaphase promoting complex by directing the SCF-dependent destruction of Emi1 in prophase 16861914_loss of pRb repression of E2F-mediated transcription causing misregulation of Emi1 and APC/C substrates results in the generation of tetraploidy and proliferation of genomically unstable cells in the absence of normal p53 function 16921029_Emi1 associates in a complex with the anaphase-promoting complex/cyclosome (APC/C) and Cdh1; Emi1 binds to the APC/C via a conserved C-terminal destruction (D)-box and can compete for APC/C-substrate interaction. 17190794_bservations reveal a novel mechanism for the control of entry into the first meiotic division: an Emi1-dependent inhibition of APC(Cdh1). 17234884_These data suggest that Emi1 plays a critical role in preserving genome integrity by blocking rereplication, revealing a previously unrecognized function of this inhibitor of anaphase-promoting complex/cyclosome. 17485488_Emi1 plays a crucial role in the cell cycle to couple DNA replication with mitosis 17609108_critical spindle pole-associated mechanism, called the END (Emi1/NuMA/dynein-dynactin) network, spatially restricts APC/C activity in early mitosis 18204430_We investigated Emi1 protein expression in ovarian neoplasms 19211842_Emi1 down-regulation and APC activation leads to stable p21-dependent G2 arrest after DNA damage. 19822658_Results underscore the crucial role of cyclin A2-CDK2 in regulating the PLK1-SCF(beta-TrCP1)-EMI1-APC/C axis and CDC6 to trigger genome reduplication after the activity of CDK1 is suppressed. 20641033_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 20717963_Results suggest that Bcr-Abl increases Emi1 phosphorylation and stability to prevent Skp2 protein degradation via APC/Cdh1-induced ubiquitination and to enhance proliferation of CML cells. 20800603_Observational study of gene-disease association. (HuGE Navigator) 21454540_the ability of Emi1 to inhibit APC/C is negatively regulated by CDKs 22995332_Emi1 participates in human hepatocellular carcinoma (HCC) cell proliferation and that progression is controlled by anaphase-promoting complex/cyclosome (APC/C) inhibition, which stabilized Skp2 and enabled p27(kip1) degradation. 23202783_Emi1 expression (>5%) was seen in 23.3% of ovarian clear cell carcinoma, and associated with high FIGO grades and poor overall survival 23645673_Emi1 depletion enhances the sensitivity of cancer cells to doxorubicin and x-ray irradiation. 23708605_The C-terminal domain inhibits multiple APC/C(CDH1) functions. The intrinsically disordered D-box, linker & tail elements, & a structured Zn-binding domain synergistically block the substrate-binding site & inhibit ubiquitin-chain elongation. 24074588_Human papillomavirus type 16 E7 expression causes increased EMI1 mRNA expression and also inhibits EMI1 degradation. 24277465_Examined eoffect of Emi1 over-expression on Skp2 expression in breast cancer. Found expression of Emi1 was positively related with Skp2 expression; Emi1 expression correlated significantly with histologic grade. Skp2 expression obtained similar results. 26288249_Results from a study on gene variability markers in early-stage human embryos shows that FBXO5 is a putative variability marker for the 3-day, 8-cell embryo stage. 27065322_The fact that Emi1 overexpression promotes chromosome instability (CIN) and the formation of solid cancers in vivo indicates that Emi1 overexpression actively drives solid tumorigenesis. These novel mechanistic insights have important clinical implications. 29850565_Both isoforms of FBXO5 promoted the migration and osteogenic differentiation potential of human periodontal ligament stem cells. 29875408_demonstration using human cell models that cell-cycle commitment is mediated by an EMI1-APC/C(CDH1) dual-negative feedback switch, in which EMI1 is both a substrate and an inhibitor of APC/C(CDH1) 30341246_Higher mRNA expression levels of FBXO1, FBXO31, SKP2, and FBXO5 were significantly associated with worse prognosis for breast cancer patients. (Review) 30554948_A subset of BRCA1-deficient triple-negative breast cancer cells develop resistance to PARP inhibitors by downregulating EMI1 and restoring RAD51-dependent HRR. Reconstitution of EMI1 expression reestablishes PARPi sensitivity both in cellular systems and in an orthotopic mouse model. 31036696_We identified hyperphosphorylation of CDH1, but not pseudosubstrate inhibition by early mitotic inhibitor 1 (EMI1), as a major mechanism driving attenuated APC/C(CDH1) activity in the G1-phase of the cell cycle in cancer stem-like cells (CSC). 33412559_Modulation of Early Mitotic Inhibitor 1 (EMI1) depletion on the sensitivity of PARP inhibitors in BRCA1 mutated triple-negative breast cancer cells. 33785736_PUMA facilitates EMI1-promoted cytoplasmic Rad51 ubiquitination and inhibits DNA repair in stem and progenitor cells. 35720327_Prognostic Significance and Immunological Role of FBXO5 in Human Cancers: A Systematic Pan-Cancer Analysis. |
ENSG00000112029 |
FBXO5 |
549.227927 |
2.1329355119 |
1.092840 |
0.12200419 |
79.793933 |
0.00000000000000000041556681271340695502670576424007049088522094565129483961785084034090687055140733718872070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000153638681826471214475425691467017342775313883232500902775718998327647568657994270324707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
745.685278790497 |
63.1354519584131 |
349.605168385963 |
22.7923847978074 |
| ENSMUSG00000019843 |
14360 |
Fyn |
protein_coding |
P39688 |
FUNCTION: Non-receptor tyrosine-protein kinase that plays a role in many biological processes including regulation of cell growth and survival, cell adhesion, integrin-mediated signaling, cytoskeletal remodeling, cell motility, immune response and axon guidance. Inactive FYN is phosphorylated on its C-terminal tail within the catalytic domain. Following activation by PKA, the protein subsequently associates with PTK2/FAK1, allowing PTK2/FAK1 phosphorylation, activation and targeting to focal adhesions. Involved in the regulation of cell adhesion and motility through phosphorylation of CTNNB1 (beta-catenin) and CTNND1 (delta-catenin). Regulates cytoskeletal remodeling by phosphorylating several proteins including the actin regulator WAS and the microtubule-associated proteins MAP2 and MAPT. Promotes cell survival by phosphorylating AGAP2/PIKE-A and preventing its apoptotic cleavage. Participates in signal transduction pathways that regulate the integrity of the glomerular slit diaphragm (an essential part of the glomerular filter of the kidney) by phosphorylating several slit diaphragm components including NPHS1, KIRREL1 and TRPC6. Plays a role in neural processes by phosphorylating DPYSL2, a multifunctional adapter protein within the central nervous system, ARHGAP32, a regulator for Rho family GTPases implicated in various neural functions, and SNCA, a small pre-synaptic protein. Participates in the downstream signaling pathways that lead to T-cell differentiation and proliferation following T-cell receptor (TCR) stimulation. Phosphorylates PTK2B/PYK2 in response to T-cell receptor activation. Also participates in negative feedback regulation of TCR signaling through phosphorylation of PAG1, thereby promoting interaction between PAG1 and CSK and recruitment of CSK to lipid rafts. CSK maintains LCK and FYN in an inactive form. Promotes CD28-induced phosphorylation of VAV1. In mast cells, phosphorylates CLNK after activation of immunoglobulin epsilon receptor signaling (PubMed:12681493). {ECO:0000250|UniProtKB:P06241, ECO:0000269|PubMed:12218089, ECO:0000269|PubMed:12640114, ECO:0000269|PubMed:12681493, ECO:0000269|PubMed:14999081, ECO:0000269|PubMed:8007959}. |
3D-structure;Adaptive immunity;Alternative splicing;ATP-binding;Cell membrane;Cytoplasm;Developmental protein;Immunity;Kinase;Lipoprotein;Manganese;Membrane;Metal-binding;Myristate;Nucleotide-binding;Nucleus;Palmitate;Phosphoprotein;Proto-oncogene;Reference proteome;SH2 domain;SH3 domain;Transferase;Tyrosine-protein kinase |
|
|
mmu:14360; |
actin filament [GO:0005884]; cell body [GO:0044297]; cell periphery [GO:0071944]; cytosol [GO:0005829]; dendrite [GO:0030425]; endosome [GO:0005768]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; glial cell projection [GO:0097386]; glutamatergic synapse [GO:0098978]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; membrane raft [GO:0045121]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; perikaryon [GO:0043204]; perinuclear endoplasmic reticulum [GO:0097038]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic density, intracellular component [GO:0099092]; Schaffer collateral - CA1 synapse [GO:0098685]; alpha-tubulin binding [GO:0043014]; ATP binding [GO:0005524]; CD4 receptor binding [GO:0042609]; CD8 receptor binding [GO:0042610]; disordered domain specific binding [GO:0097718]; enzyme binding [GO:0019899]; ephrin receptor binding [GO:0046875]; G protein-coupled receptor binding [GO:0001664]; growth factor receptor binding [GO:0070851]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; non-membrane spanning protein tyrosine kinase activity [GO:0004715]; peptide hormone receptor binding [GO:0051428]; phosphatidylinositol 3-kinase binding [GO:0043548]; phospholipase activator activity [GO:0016004]; phospholipase binding [GO:0043274]; protein kinase activity [GO:0004672]; protein tyrosine kinase activity [GO:0004713]; protein-containing complex binding [GO:0044877]; scaffold protein binding [GO:0097110]; signaling receptor binding [GO:0005102]; T cell receptor binding [GO:0042608]; tau protein binding [GO:0048156]; transmembrane transporter binding [GO:0044325]; tubulin binding [GO:0015631]; type 5 metabotropic glutamate receptor binding [GO:0031802]; activated T cell proliferation [GO:0050798]; adaptive immune response [GO:0002250]; cell differentiation [GO:0030154]; cell surface receptor signaling pathway [GO:0007166]; cellular response to amyloid-beta [GO:1904646]; cellular response to glycine [GO:1905430]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to L-glutamate [GO:1905232]; cellular response to platelet-derived growth factor stimulus [GO:0036120]; cellular response to transforming growth factor beta stimulus [GO:0071560]; dendrite morphogenesis [GO:0048813]; detection of mechanical stimulus involved in sensory perception of pain [GO:0050966]; forebrain development [GO:0030900]; heart process [GO:0003015]; I-kappaB kinase/NF-kappaB signaling [GO:0007249]; innate immune response [GO:0045087]; intracellular signal transduction [GO:0035556]; modulation of chemical synaptic transmission [GO:0050804]; myelination [GO:0042552]; negative regulation of dendritic spine maintenance [GO:1902951]; negative regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001240]; negative regulation of gene expression [GO:0010629]; negative regulation of hydrogen peroxide biosynthetic process [GO:0010730]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of oxidative stress-induced cell death [GO:1903202]; negative regulation of protein catabolic process [GO:0042177]; negative regulation of protein ubiquitination [GO:0031397]; neuron migration [GO:0001764]; neuron projection development [GO:0031175]; peptidyl-tyrosine phosphorylation [GO:0018108]; positive regulation of cysteine-type endopeptidase activity [GO:2001056]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of neuron death [GO:1901216]; positive regulation of neuron projection development [GO:0010976]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of protein localization to nucleus [GO:1900182]; positive regulation of protein targeting to membrane [GO:0090314]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531]; protein autophosphorylation [GO:0046777]; protein catabolic process [GO:0030163]; protein phosphorylation [GO:0006468]; protein ubiquitination [GO:0016567]; regulation of calcium ion import across plasma membrane [GO:1905664]; regulation of cell shape [GO:0008360]; regulation of glutamate receptor signaling pathway [GO:1900449]; regulation of peptidyl-tyrosine phosphorylation [GO:0050730]; response to amyloid-beta [GO:1904645]; response to ethanol [GO:0045471]; response to singlet oxygen [GO:0000304]; T cell receptor signaling pathway [GO:0050852]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
11106751_PKC-eta/Fyn-dependent pathway leading to keratinocyte growth arrest and differentiation 11489976_Fyn mediates inhibitory signals against the production of Th2 cytokines and down-regulates the differentiation of naive CD4+ T cells into the Th2 subset. 11676481_Src kinases Fyn and Lck facilitate the accumulation of phosphorylated CTLA-4 and its association with PI-3 kinase in intracellular compartments of T-cells. 11751285_Fyn-mediated signal transduction in the Sertoli cells may affect the survival and differentiation of germ cells during spermatogenesis 11834300_Female homozygous Fyn-deficient mice showed an enhancement of the startle amplitude under bright light but heterozygotes and wild-types did not. This is related to enhanced serotonergic and dopaminergic activity in the prefrontal cortex and limbic system. 11870621_The repertoire of inhibitory Ly49 receptors may be formed under the influenced of Fyn-dependent NK cell activation as well as the respective MHC class I environment. 12218089_Fyn demonstrates its kinase activity in membrane rafts and plays a critical role in Csk-binding protein (Cbp) phosphorylation, Cbp-Csk interaction, and Csk kinase activity. 12218114_Long term survival of naive T cells requires a combination of signals transduced by Src family kinase member Fyn (and Lck) and signals through the IL-7 receptor. 12359721_role of signaling in maximal hypertonic activation of the osmotic response element-binding protein/tonicity-responsive enhancer-binding protein 12372285_Fyn mediates neuronal guidance regulated by Sema3A signaling. 12374739_the mechanism by which tr-kit triggers resumption of meiosis of mouse eggs requires a functional interaction with Fyn and phosphorylation of PLCgamma1 12399115_Fyn can modulate alcohol consumption and prevent behavioural changes during alcohol withdrawal, possibly via phosphorylation of NR2B. 12526739_Reelin activates Fyn to phosphorylate and downregulate Dab1 during brain development 12545173_described a signalling mechanism by which SAP is able to physically and functionally link certain immune cell receptors to the Src-related PTK FynT 12668668_Fyn catalyzes Nephrin phosphorylation in podocyte detergent-resistant membrane fractions. 12732664_regulation through translocation of activated Lck into lipid rafts 12921535_Fyn is required for stress-induced caveolin phosphorylation. 13129922_Fyn uses cortactin as a specific substrate and cooperative effector in integrin-mediated signaling processes regulating metastatic potential 13129934_Fyn has a role in phosphorylating PSD-93, which is also involved in fyn-mediated modification of NMDA receptor function 15034040_Rsults characterize the spatial and temporal roles of Fyn and Lck in T cell receptor-proximal signaling and underscore the importance of lipid rafts in coordinating the interaction of these kinases either directly or through intermediates. 15049519_Expression of 20 genes including a substantial number of myelin-associated genes was strongly repressed in fyn-deficient mice 15073522_Fyn was intensely expressed in the leading process of migratory cortical neurons generated in the later stage. These findings strongly suggest that Fyn is required for the migration of later-generated neurons. 15210764_Fyn may substitute (inefficiently) for Lyn in signal initiation in Lyn(-/-) BMMCs. 15220932_The interaction with host c-Fyn is a pivotal link between bacterial Tir and the cellular Nck-WASP-Arp2/3 cascade, and shows a tractable experimental system in which to dissect tyrosine kinase signalling. 15465914_When signaling of naive T cells is restricted to the T cell antigen receptor, Fyn plays an essential role by positive regulation of Lck activity. 15528192_Fyn plays critical roles in promoting accelerated myelin basic protein (MBP) expression during myelinogenesis in a MBP isoform-preferential manner 15537652_Fyn kinase plays a key role in the UVB-induced phosphorylation of histone H3 at serine 10 15539156_SAP is necessary for T(H)2 cytokine regulation primarily as a result of its capacity to recruit FynT. 15623578_Data show that the NCAM140 isoform directly interacts with the intracellular domain of the receptor-like protein tyrosine phosphatase RPTPalpha, a known activator of p59fyn. 15713745_PEDF downregulates Fyn through Fes, resulting in inhibition of FGF-2-induced capillary morphogenesis of endothelial cells 15813945_The impairment of tone-dependent conditioned freezing in Fyn-TG mice is caused by disruption of the NR2B-containing NMDA receptor function. 15902435_negatively regulates the development of experimental autoimmune uveoretinitis 16101815_Fyn is involved in fasting-induced thymic involution. 16115884_the Fyn-tau interaction has a role in neurodegeneration 16162939_Src and Fyn are implicated in Reelin-dependent tyrosine phosphorylation, impairing formation of the Purkinje plate. 16316995_propose a cooperative role between Fyn and Lyn kinases in the activation of SphK1 and Sphk2, which contributes to mast cell responses 16337776_Muscarinic M2 receptor (M2R)-mediated transactivation of EGFR specifically involves Fyn but not c-Src or Yes. 16339530_PTPalpha is a component of the complex Src family tyrosine kinase regulatory network in thymocytes and is required to suppress Fyn activity in unstimulated cells in a manner that is not compensated for by the major T cell PTP and SFK regulator, CD45. 16407246_the dopamine D(2) receptor antagonist induces striatal Fyn activation and the subsequent tyrosine phosphorylation of NR2B alters striatal neuronal activity 16442778_Data suggest that recruitment of both Shp2 and Fyn to juxtamembrane sites in c-Kit results in Shp2 phosphorylation, downstream signaling to p38 mitogen-activated protein kinase, and enhanced chemotaxis of mast cells. 16540575_Fyn tyrosine kinase plays a pivotal role in regulating spine morphogenesis. Fyn is involved in Sema3A-induced clustering of PSD-95. 16547257_Fyn to be a key regulator of tubulin cytoskeleton reorganization in T cells 16628251_CD2AP, Fyn, and synaptopodin have roles in development of focal segmental glomerulosclerosis in a mouse model 16723512_Data show that in response to angiotensin II, the ability of ERK1/2 to remain within the cytoplasm or translocate into the nucleus is controlled by c-Src/Yes/Fyn or heterotrimeric G protein/PKCzeta signaling, respectively. 16751367_Fyn kinase is involved in the regulation of binding properties of gamma-tubulin and/or its associated proteins, and thus modulates microtubule nucleation in activated bone marrow-derived mast cells. 16765915_Fyn phosphorylates and binds to beta-adducin at Y489, resulting in translocation of beta-adducin to the Fyn-enriched regions in the plasma membrane 16841086_Fyn is essential for phosphorylating PIKE-A and protects it from apoptotic cleavage. 16847311_Data show that the interaction of the FynT SH3 domain with SLAM-SAP is strictly inducible, and is dependent on engagement of SLAM by extracellular ligands. 16880206_Fyn-calcineurin-NFATc2 and MEK2-ERK1/2 are two independent signaling pathways that cooperate to control T cell receptor-mediated ICOS induction 16882656_extracellular matrix fragments produced by apoptotic EC initiate a state of resistance to apoptosis in fibroblasts via an alpha2beta1 integrin/SFK (Src and Fyn)/phosphatidylinositol 3-kinase (PI3K)-dependent pathway 16908977_The experimental model suggests that a deficiency for Fyn is detrimental in prion diseases, although it has no major effect on the clinical course of an experimental prion infection of the CNS. 16966372_Fyn stimulates the ERK/MAPK pathway in primary T cells but has little influence on the mobilization of Ca2+. Our result suggest that Fyn activates ERK via a different upstream signaling route. 17045972_BDNF-dependent phosphorylation was abolished in Fyn knockout mice. 17046829_Srcasm is a novel regulator of Fyn promoting kinase down-regulation in a phosphorylation-dependent manner 17202343_distinct SLAM-associated protein(SAP) signaling pathway regulates follicular helper CD4 T cell differentiation, separate from the SLAM-SAP-Fyn signaling pathway regulating Th1/Th2 differentiation 17277164_the dominance of the Fyn/p38 MAPK pathway in driving IL-4 production. 17290413_These results suggest that the restoration of FcRgamma/Fyn signaling represents a new approach for the treatment of demyelinating diseases. 17460065_NRG1 stimulation of cells expressing ErbB4 and Fyn leads to the association of Fyn with ErbB4 and consequent activation 17475794_Lipid raft localization of protein-tyrosine kinase TrkB receptors is regulated by Fyn and represents an important factor in determining brain-derived neurotrophic factor (BDNF) signaling in neurons. 17488639_The loss of Fyn markedly improves peripheral tissue insulin sensitivity by relieving a selective negative modulation of AMP kinase activity in adipose tissue and skeletal muscle. 17521824_Transient transfection of Fyn hardly affected multiplication and specialization of murine embryonic stem (ES) cells. Fyn overexpression in ES cells resulted in altered sphere formation capability in all clones stably expressing Fyn. 17599905_the mechanism of signal transduction by CD244 is to regulate FYN kinase recruitment and/or activity and the outcome of CD48/CD244 interactions is determined by which other receptors are engaged. 17623777_Data imply that distinct modes of spatial activation and membrane delivery, under the control of specific acylation attachment sequences and endosome sub-type requirements, define distinct properties of the three SFKs (Src family kinases). 17845201_Fyn, PI-3 kinase, and Akt are required for the activation of T cells by cross-linking Qa-2. 17878361_demonstrate a role for Fyn in modulating the response to Ag in primary T cells 18089558_Fyn, due in part to its effects on Dab1, regulates the phosphorylation, trafficking, and processing of APP and apoEr2. 18095272_Fyn is a target of (-)EGCG in the inhibition of JB6 C141 cell transformation. 18096814_our data from fyn(-/-) transgenic mice show that accumulation of Abeta on the neuronal surface was not sufficient to cause cell death but that fyn is required for both the redistribution of Abeta and subsequent cell death. 18301737_optimal association of GAMMAHV.M2 and the Vav1 and Fyn proteins requires a second structural motif encompassing two tyrosine residues (Tyr120 and 129) 18403215_Fyn is essential for normal brain development suggesting that defects in oligodendrocyte development cause degeneration of cortical axons and subsequent hydrocephalus in fyn-deficient mice 18430780_Fyn and Src have distinct requirements for interaction with alphaIIbbeta3; and, consequently, the two src-family kinases can mediate different functional responses. 18482989_Ly108-triggered protein tyrosine phosphorylation was due to the capacity of SAP to recruit FynT 18490510_Active Fyn phosphorylates hnRNP A2 and stimulates translation of an myelin basic protein (MBP) A2RE (A2 response element) -containing reporter construct. 18632659_myricetin exerts potent chemopreventive activity mainly by targeting Fyn in skin carcinogenesis 18636972_Results suggest that Fyn and PI3K might take part in the modulation of membrane-associated gamma-tubulin activities. 18647647_These results show that, in MA-10 cells, the hLHR activates Fyn through an arrestin-3-dependent pathway and that this pathway is a mediator of the hLHR-provoked release of EGF-like growth factors. 18660530_Lck-dependent Fyn activation is predicated by proximity-mediated transphosphorylation of the Fyn kinase domain, and targeting kinase-active Lck to LR is dependent on the C-terminal sequence QPQP. 18684920_Signaling lymphocytic activation molecule-associated protein (SAP)-Fyn interaction during natural killer (NK)T cell ontogeny and function is critical for efficient NK T cell development in vitro and in vivo. 18691323_These findings strongly suggest that activation of the Fyn signaling pathway is involved in hippocampus-dependent formation of contextual fear memory. 18832694_ablation of Fyn expression does not alter most Ag-driven CD4(+) T cell responses, with the exception of cytokine production, which under some circumstances is enhanced in Fyn-deficient CD4(+) T cells. 18854310_LAR regulates Fyn/JAK2/STAT3 and Fyn/p38 MAPK pathways involved in ROS-induced apoptosis 18948260_Activation of c-Src and Fyn kinases by protein-tyrosine phosphatase RPTPalpha is substrate-specific and compatible with lipid raft localization. 19000718_Fyn-catalyzed Tyr-phosphorylation of Grp94 is an event required to promote the chaperone export from the endoplasmic reticulum occurring in the early phase of myoblast differentiation. 19016778_The authors show that clustering transfers Tir into cholesterol-rich detergent-resistant microdomains (DRMs), a signal prompting transient c-Fyn accumulation at bacterial adhesion sites. 19118543_Results demonstrate a significant role for Fyn kinase activity in meiotic maturation. 19294646_Our data suggest that betaAP acutely affects proper hippocampal function through a Fyn-dependent mechanism. We propose that such alteration might be related to the cognitive impairment observed, at least, during the early phases of Alzheimer's disease. 19363790_Demonstrated suppression of Fyn kinase signaling prior to fertilization caused disruption of the functional polarity of the oocyte. 19403835_Fyn is expressed in spinal lateral motor neurons of mouse embryo at the time of limb trajectory selection. 19776266_findings suggest that a normal role for CNS laminin is to promote the development of oligodendrocyte progenitors into myelin-forming oligodendrocytes via modulation of Fyn regulatory molecules 19812040_Findings demonstrate that PTPalpha is a critical regulator of Fyn activation and of specific Fyn signaling events during differentiation, and is essential for promoting OPC differentiation and central nervous system myelination. 19888448_in the thymus, Fyn acts as a tyrosine kinase that transduces the leptin signal independently of JAK2 activation, and mediates some of the immunomodulatory effects of leptin in this tissue 19904769_Data indicate that Fyn was not required for the induction of central tolerance by negative selection, the adaptor protein role of c-Cbl, or the normal development and function of Treg. 19917692_maximal TCR-induced rpS6 phosphorylation in CD8 T cells requires both Lck and Fyn activity 19917775_Gbeta1-mediated Fyn activation integrates FAK with AJ, preventing persistent endothelial barrier leakiness. 20043875_Results indicate Fyn kinase mediates the receptor-dependent activation of TRPC channels that contribute to degranulation in FcepsilonRI-stimulated mast cells. 20126650_results indicate that the cellular levels of Lck and Fyn play a role in regulating the strength of the gammadeltaTCR signaling response at different stages in the life of the gammadelta T cell 20142099_Fyn kinase directly phosphorylated LKB1 on tyrosine 261 and 365 residues, and mutations of these sites resulted in LKB1 export into the cytoplasm and increased AMP-dependent protein kinase phosphorylation. 20233604_In summary, we demonstrate Fyn as one of the SFKs that binds to and phosphorylates the gamma2 subunit of the GABA(A) receptor. 20335178_The limited contribution of Fyn and Gab2 to the high affinity IgE receptor signaling in mast cells. 20372074_Fyn takes part in regulating several key signaling pathways leading to the exit from germinal vesicle and metaphase of the first meiotic division and in the ingression of the cleavage furrow during RMI in mammalian oocytes. 20398064_Results suggest that tyrosine phosphorylation of R3 subtype receptor-type protein tyrosine phosphatases promotes their complex formations with Grb2 or Fyn and thus participates in the regulation of cell morphology. 20495068_PLZF acts independently of SAP- and Fyn-mediated signaling pathways. 20506281_Data demonstrate that CRMP1 tyrosine 504 is a primary target of the Src family of tyrosine kinases (SFKs), specifically Fyn. 20544909_The integrity of FYN-dependent lipid rafts is required for local immune system activation within denervated muscle, and lipid rafts are implicated in orchestrating muscle-immune-cell cross-talk 20591331_Fyn-null female mice exhibit reduced fertility that appears to result from actin cytoskeletal defects rather than calcium signalling. 20717919_Fyn is a pro-resorptive Src family kinase, which exerts its effects by prompting proliferation and differentiation while attenuating apoptosis of osteoclast lineage cells. 20814235_data indicate that glucocorticoids promote autophagy by inhibiting Fyn- and IP(3)-dependent calcium signals. 20841362_A continuous colocalization of Fyn and F-actin during meiosis and imply a role for the SFKs, in general, and for Fyn, in particular, in regulating pathways that involve actin cytoskeleton, during ingression of the meiotic and mitotic cleavage furrows. 20850416_these results show that bone marrow-derived mast cells are able to release VEGF under hypoxic conditions through a tetanus toxin-sensitive mechanism, promoted by free radicals-dependent Fyn kinase activation. 21054385_cAMP-dependent protein kinase (PKA)/Fyn/GluN2-containing NMDA receptor GluN2BR signaling plays a critical role in triggering GluN2BR hyperfunction and pain hypersensitivity during inflammation. 21097520_The preinduction regulation of Nrf2 is controlled by the nuclear export of Fyn, allowing for activation of defensive gene expression 21228179_Abeta, tau, and Fyn jointly impair synaptic and network function and disrupting the copathogenic relationship between these factors could be of therapeutic benefit. 21592972_Negative regulation of Gq-mediated pathways in platelets by G(12/13) pathways through Fyn kinase 21705469_the WASP N-terminal domain plays a pivotal role in the TCR signaling cascade by binding to Fyn. 21741124_loss of Fyn at early stages of disease increases soluble Abeta accumulation and worsens spatial learning in the absence of changes in tau phosphorylat in Alzheimer's disease. 21757751_CD5 glycoprotein-mediated T cell inhibition dependent on inhibitory phosphorylation of Fyn kinase 21871204_Post-translational modification of Fyn determines its localization: localization of active Fyn to spindle poles is important in metaphase; localization of Fyn at cell membrane near cleavage furrow is important in furrow ingression during cytokinesis. 21872217_The data of this study indicated that Fyn differentially affects spine density and morphology in the cortex and hippocampus and has age-dependent effects on dendritic spine formation and spine morphology in vivo 21881001_TM4SF10, possibly through ADAP, may regulate Fyn activity 21918125_These findings are consistent with a model in which FYN kinase plays an important role in proper shaping of the head and acrosome within the testis and possibly an additional role in the sperm acrosome reaction. 22057277_hyperosmotic cholestasis is triggered by a NADPH oxidase-driven reactive oxygen species formation that mediates Fyn-dependent retrieval of the Mrp2 and Bsep from the canalicular membrane, which may involve an increased cortactin phosphorylation. 22189847_Fyn is not required for the pathogenesis of Bcr-Abl-mediated leukemias. 22386537_mechanism underlying impaired TCR signaling and subsequent memory cell anergy must involve a Fyn signaling pathway given that the suppression of Fyn activity restores CD3/ZAP-70 complex formation and TCR proximal signaling 22387224_Fyn relays RANK/RANKL signal to the costimulatory DAP12/FcRgamma-mediated signals. 22437915_our results demonstrate that Fyn kinase downregulates nonmuscle myosin II activity, thus promoting oligodendrocyte morphological differentiation. 22450804_Lyn but not Fyn kinase controls IgG-mediated systemic anaphylaxis. 22539787_Fyn is identified as a possible novel regulator of the developmental balance between T helper (Th)17 type and regulatory T (TREG) cell subsets. 22564682_Dampening of Fyn kinase activity also inhibited RhoA activation and microtubule nucleation, key regulators of mast cell degranulation 22745922_Mouse skeletal muscle fiber-type-specific macroautophagy and muscle wasting are regulated by a Fyn/STAT3/Vps34 signaling pathway. 23012656_Phosphorylation of cytohesin-1 by Fyn is required for initiation of myelination and the extent of myelination during development. 23127881_Our findings suggest a role for Fyn kinase in neuronal death after neonatal hypoxic-ischemic injury 23284724_Overexpression and activity of Fyn mediate proliferation and blastic features of chronic myelogenous leukemia. 23321073_Data from knockout mice suggest that Fyn is important regulator of whole-body integrative metabolism that coordinates adipose tissue expansion, inflammation, and insulin sensitivity in states of nutrient excess (i.e., diet-induced obesity). 23438599_Fyn regulates the activity of the adipogenic transcription factor signal transducer and activator of transcription 5a (STAT5a) through enhancing its interaction with the GTPase phosphoinositide 3-kinase enhancer A (PIKE-A). 23593269_Fyn kinase is required for optimal humoral responses 23967147_CD36 and Fyn kinase mediate malaria-induced lung endothelial barrier dysfunction in mice infected with Plasmodium berghei. 24036998_Signaling by Fyn-ADAP via the Carma1-Bcl-10-MAP3K7 signalosome exclusively regulates inflammatory cytokine production in NK cells. 24045941_SAP plays an essential role in CIA because of Fyn-independent and Fyn-dependent effects on TFH cells and, possibly, other T cell types. 24312371_The normal levels of dihydroxyacetone phosphate and glyceraldehyde-3-phosphate observed in the FynKO liver extracts suggested normal triose kinase function 24312422_identify Fyn-centric gene networks influencing variance in ethanol LORR. Such networks may also inform future design of pharmacotherapies for the treatment and prevention of alcohol use disorders 24912779_The SH2 domain of Fyn plays an essential role in Reelin signaling transduction, and an SH2 domain mutant disturbs neuronal migration in a dose-dependent manner. 25106729_These results suggest the critical physiological role of TRL4-Fyn interaction in the modulation of PAI-1-tPA axis in astrocytes during neuroinflammatory responses such as ischemia/reperfusion injuries. 25251774_The aspartic acid of Fyn at 390 is indispensable for the radial migration. 25340851_total levels of ApoER2 and surface levels of ApoER2 are increased by active Fyn 25877924_The expression of Fyn is protective in acute chemical-induced colitis and promotes recovery from disease. 25951993_A synergistic regulation of Fyn signaling was found in corpus striatum regulated by protein tyrosine phosphate. 25967238_Fyn, but not Lyn, was required for complete Pyk2 phosphorylation by thrombin. 26157004_This study demonstrate that both the tyrosine kinase Fyn and the serine/threonine kinase PKCdelta are differentially expressed in microglia and astrocytes. 26365631_Fyn facilitates mitotic spindle formation through the increase in microtubule polymerization, resulting in the acceleration of M-phase progression. 26449489_Results identified Fyn kinase as a molecular mediator of BDNF signaling that activates Erk1/2 and promotes oligodendrocyte myelination 26459603_Thy-1 facilitates the recruitment of membrane raft-residing signaling molecules such as Fyn kinase and the SFK regulator, Csk binding protein (Cbp), to focal adhesions via its direct coupling of integrins and raft domains. 26686094_Fyn acts as a regulatory nexus between solar UV, ROS and signal transduction during skin carcinogenesis. 26801084_MOBP synthesis is stimulated by Fyn activity. 26808221_Fyn deficiency alters retinal morphology and adhesion properties of Muller cells. Fyn deficiency parallels a decreased optokinetic response to visual stimuli in vivo. 26881253_Fyn mediates high glucose-induced actin cytoskeleton remodeling of podocytes via promoting ROCK activation and paxillin phosphorylation 26892111_Fyn expression was elevated in abortion-prone mice 27146985_The results of this study suggested that Fyn-mediated NR2B signaling plays a critical role in regulation of prediabetic neuropathy and that the increased expression/function of NR2B subunit-containing NMDARs may contribute to the progression of neuropathy in type 2 diabetes 27183589_data show that Fyn kinase is activated after TLR4 triggering and exerts an important negative control on LPS-dependent TNF production in mast cells controlling the inactivation of PP2Ac and activation of PKCalpha/beta necessary for the secretion of TNF by VAMP3(+) carriers 27525436_FYN is activated by oxidative stress and serves as a negative feedback regulator of NOX4 in cardiomyocytes during cardiac remodeling 27616741_loss of Fyn in the kidney prevents unilateral ureteral obstruction-induced tubulointerstitial fibrosis, mediated by a reduction in STAT3 phosphorylation 27859601_The results of this study suggested the existence of a potential DEP-1-Fyn axis in the regulation of microglial functions. 28363782_This study reported that suppression of Fyn expression in mouse cerebral cortex led to migration defects of both early-born and late-born neurons. 28368000_Fyn and Lyn as important factors that promote Plasmacytoid dendritic cell responses. 28440917_ATF3 inhibit the expression and release of TNF-alpha, IL-1beta, IL-6, and IL-18 induced by Mycoplasma pneumonia in vitro and in vivo, which is associated with its negative regulation of Egr-1/Fyn signaling pathway. 28533542_Our findings suggest a mechanism, by which a decrease in miR-125a-3p during oocyte maturation facilitates GVBD by allowing Fyn up-regulation and the resulting stabilization of the interaction between actin and A-type lamins. 28539383_In summary,MAGI-2 and Fyn protect dendrin from Nedd4-2-mediated ubiquitination and from nuclear translocation, thereby maintaining the physiologic homeostasis of podocytes. 28586109_results show that Fyn kinase is an important positive effector of TGF-beta-induced chemotaxis through the control of phosphatase PP2A activity and this is relevant to pathological processes that are related to TGF-beta-dependent mast cell migration 28637841_We show that PTP-3, a LAR homolog in Caenorhabditis elegans, participates in Sema2A-regulated axon guidance. PTPdelta, a member of vertebrate LAR class PTPs, is involved in Sema3A-regulated cortical dendritic growth. In Sema3A signaling, PTPdelta activates Fyn and Src kinases by dephosphorylating their C-terminal Tyr residues. 28840468_Fyn has a critical role in the molecular pathways affected during the development of L-DOPA-induced dyskinesia. 28864542_Somatodendritic accumulation of Tau in Alzheimer's disease is promoted by Fyn-mediated local protein translation. 29180007_These results suggested that Fyn has a regulatory role in iNOS expression in astrocytes during neuroinflammatory responses. 29353218_these results suggest that Nrf2 plays a central role in the prevention of Ang II-induced cardiomyopathy, and SFN prevents Ang II-induced cardiomyopathy partially via the Akt/GSK-3beta/Fyn-mediated Nrf2 activation. 29661967_Our results suggest that Fyn-mediated regulation of the cell-cell adhesion of neuroblasts is critical for their detachment from chains in the postnatal brain 30252956_Fyn(-/-) mice exhibit reduced Tyr-phosphorylation of EPO-R and decreased STAT5-activity. Fyn(-/-) mice responded poorly to stress erythropoiesis. Absence of Fyn resulted in persistent-activation of Nrf2, accumulation of nonfunctional proteins, ROS-induced over-activation of Jak2-Akt-mTOR-pathway, and repression of autophagy with perturbation of lysosomal-clearance. 30658076_MiR-125a-3p exerts anti-fibrotic effects in silica-induced pulmonary fibrosis by regulating Fyn/STAT3 signaling. 31036561_In vivo experiments using A53T and viral-alphaSyn overexpression mouse models as well as human Parkinson's disease neuropathological results further confirm the role of Fyn in NLRP3 inflammasome activation. 31237563_Tau controls the nanoscale organization of Fyn in dendrites, with the pathological Tau P301L mutation potentially contributing to synaptic dysfunction by promoting aberrant Fyn nanoclustering in spines. 31452242_Loss of tau and Fyn reduces compensatory effects of MAP2 for tau and reveals a Fyn-independent effect of tau on calcium. 31609430_Nyap1 Regulates Multipolar-Bipolar Transition and Morphology of Migrating Neurons by Fyn Phosphorylation during Corticogenesis. 31950181_Fyn tyrosine kinase, a downstream target of receptor tyrosine kinases, modulates antiglioma immune responses. 31994829_Fyn-mediated phosphorylation of Pyk2 promotes its activation and dissociation downstream of gonadotropin-releasing hormone receptor. 32565338_Fyn kinase mediates pro-inflammatory response in a mouse model of endotoxemia: Relevance to translational research. 32665013_Fyn depletion ameliorates tau(P301L)-induced neuropathology. 32814048_Fyn Kinase Controls Tau Aggregation In Vivo. 32863204_Fyn specifically Regulates the activity of red cell glucose-6-phosphate-dehydrogenase. 33380425_Disturbances in PP2A methylation and one-carbon metabolism compromise Fyn distribution, neuritogenesis, and APP regulation. 34105991_Differential effects of the Src family tyrosine kinases Yes and Fyn on lipopolysaccharide-induced lung injury in ice. 35353146_DHF-7 Ameliorates Behavioral Disorders and White Matter Lesions by Regulating BDNF and Fyn in a Mouse Model of Schizophrenia Induced by Cuprizone and MK-801. 35710078_Fyn-kinase and caveolin-1 in the alveolar epithelial junctional adherence complex contribute to the early stages of pulmonary fibrosis. 36016954_FYN regulates cell adhesion at the blood-testis barrier and the apical ectoplasmic specialization via its effect on Arp3 in the mouse testis. |
ENSG00000010810 |
FYN |
864.826401 |
0.4761435063 |
-1.070532 |
0.09432001 |
127.970145 |
0.00000000000000000000000000001139441213997213747880853474468909092833435937473131632292300207768071163325724823311979605477972654625773429870605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000008507827731179196881008056443917446617676588305345706873816817685638358610689113703529073973186314105987548828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
559.282938028511 |
36.5906580604908 |
1174.61002959151 |
55.7696757290146 |
| ENSMUSG00000019851 |
64058 |
Perp |
protein_coding |
Q9JK95 |
FUNCTION: Component of intercellular desmosome junctions. Plays a role in stratified epithelial integrity and cell-cell adhesion by promoting desmosome assembly. Plays a role as an effector in the TP53-dependent apoptotic pathway. {ECO:0000269|PubMed:15797384}.; FUNCTION: Plays a role as an effector in the TP53-dependent apoptotic pathway. {ECO:0000269|PubMed:10733530}. |
Apoptosis;Cell adhesion;Cell junction;Cell membrane;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:64058; |
cell-cell junction [GO:0005911]; desmosome [GO:0030057]; Golgi apparatus [GO:0005794]; mitochondrion [GO:0005739]; plasma membrane [GO:0005886]; activation of cysteine-type endopeptidase activity [GO:0097202]; amelogenesis [GO:0097186]; cell-cell adhesion [GO:0098609]; desmosome organization [GO:0002934]; heterotypic cell-cell adhesion [GO:0034113]; intrinsic apoptotic signaling pathway by p53 class mediator [GO:0072332]; Notch signaling pathway [GO:0007219]; positive regulation of proteolysis [GO:0045862] |
10733530_A paper describing the identification of Perp (p53 Apoptosis Effector Related to PMP22). Perp was found as a novel target gene of the p53 tumor suppressor, which is selectively induced during apoptosis compared to cell cycle arrest. 15192153_In contrast to p53 shRNAs, PUMA shRNAs were unable to cooperate with oncogenic ras in transformation 15797384_Perp is a key effector in the p63 developmental program. 16061634_Perp is required for efficient skin carcinogenesis and suggest a role for intact cell-cell adhesion in supporting tumor development in these settings. 16417219_Perp's requirement for both viability and epithelial integrity reflects a role in the stratified epithelial compartment. 16485031_Perp is requered throughout adult life for proper epithelial homeostasis and function. 19158346_PERP, a p53 proapoptotic target, mediates apoptotic cell death in renal ischemia. 19158843_pemphigus vulgaris autoantibodies disrupt Perp expression at the membrane and trigger its internalization along with DSG3 into the endosomal pathway, where it is ultimately targeted to the lysosome for degradation 20975948_Studies suggest that Perp-deficiency promotes cancer by enhancing cell survival, desmosome loss, and inflammation, and they highlight a fundamental role for Perp and desmosomes in tumor suppression. 21285247_Data show that PERP is necessary for the integrity of the ameloblast-SI interface and that a lack of Perp causes downregulation of genes that are required for proper enamel formation. 22515648_an important role for Perp in normal mammary tissue function and in mammary cancer suppression 24865245_Perp might play important roles in the formation and integration of stellate reticulum, dental lamina structure and enamel formation 29740445_Perp deletion promoted Th17 responses and exacerbated the development and severity of experimental autoimmune encephalomyelitis. 31898316_Confirming the recessive inheritance of PERP-related erythrokeratoderma. |
ENSG00000112378 |
PERP |
7.513827 |
0.0240400856 |
-5.378414 |
1.54694418 |
13.327303 |
0.00026156939389815391048405013307842637004796415567398071289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0014088490126791656555094167657671277993358671665191650390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.367724959129053 |
0.474317668063576 |
15.2963248416212 |
8.76823030119815 |
| ENSMUSG00000019889 |
19272 |
Ptprk |
protein_coding |
P35822 |
FUNCTION: Regulation of processes involving cell contact and adhesion such as growth control, tumor invasion, and metastasis. Negative regulator of EGFR signaling pathway. Forms complexes with beta-catenin and gamma-catenin/plakoglobin. Beta-catenin may be a substrate for the catalytic activity of PTPRK/PTP-kappa. |
Disulfide bond;Glycoprotein;Hydrolase;Immunoglobulin domain;Membrane;Phosphoprotein;Protein phosphatase;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:19272; |
axon [GO:0030424]; cell junction [GO:0030054]; cell surface [GO:0009986]; cell-cell junction [GO:0005911]; dendrite [GO:0030425]; intracellular membrane-bounded organelle [GO:0043231]; leading edge membrane [GO:0031256]; neuronal cell body [GO:0043025]; photoreceptor outer segment [GO:0001750]; plasma membrane [GO:0005886]; beta-catenin binding [GO:0008013]; gamma-catenin binding [GO:0045295]; protein kinase binding [GO:0019901]; protein tyrosine phosphatase activity [GO:0004725]; cell adhesion [GO:0007155]; cell migration [GO:0016477]; cellular response to reactive oxygen species [GO:0034614]; cellular response to UV [GO:0034644]; focal adhesion assembly [GO:0048041]; negative regulation of cell cycle [GO:0045786]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of keratinocyte proliferation [GO:0010839]; neuron projection development [GO:0031175]; protein dephosphorylation [GO:0006470]; protein localization to cell surface [GO:0034394]; signal transduction [GO:0007165]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
21690299_RPTP-kappa is proteolytically processed to isoforms that have opposing effects on beta-catenin activity. 30185827_Study show that RAS at the Golgi is selectively activated by apoptogenic stimuli and antagonizes cell survival by suppressing ERK activity through the induction of PTPRkappa, which targets CRAF for dephosphorylation. RAS oncogenic potential is strictly dependent on its sublocalization, with Golgi complex-located RAS antagonizing tumor development. |
ENSG00000152894 |
PTPRK |
178.718896 |
0.1436160976 |
-2.799711 |
0.26401758 |
104.827671 |
0.00000000000000000000000133233679221990668554737197562208776712020848107218392656144323319463568244813700403028633445501327514648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000709585804863413284171376078223492994209272955530131159787273278927166586527164326980710029602050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.8398319288609 |
8.80378347608978 |
333.109120266938 |
42.5449911942779 |
| ENSMUSG00000019944 |
69288 |
Rhobtb1 |
protein_coding |
Q9DAK3 |
|
Direct protein sequencing;GTP-binding;Nucleotide-binding;Reference proteome;Repeat |
|
|
mmu:69288; |
cell cortex [GO:0005938]; cell projection [GO:0042995]; cytoplasmic vesicle [GO:0031410]; cytoskeleton [GO:0005856]; plasma membrane [GO:0005886]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; protein kinase binding [GO:0019901]; actin filament organization [GO:0007015]; cortical cytoskeleton organization [GO:0030865]; engulfment of apoptotic cell [GO:0043652]; establishment or maintenance of cell polarity [GO:0007163]; regulation of actin cytoskeleton organization [GO:0032956]; regulation of cell shape [GO:0008360]; small GTPase mediated signal transduction [GO:0007264] |
30896450_RhoBTB1 protected from hypertension, vascular smooth muscle dysfunction, and arterial stiffness in at least two models of hypertension. |
ENSG00000072422 |
RHOBTB1 |
762.993192 |
0.3308937044 |
-1.595560 |
0.16576303 |
89.600495 |
0.00000000000000000000291451192671146030666975050007417076089575828420230883862920845395194646698655560612678527832031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001219611144716180442574780312248835412557971140605535677152448936766404585796408355236053466796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
381.249517218484 |
43.9013212389485 |
1152.18123553996 |
97.4193598458529 |
| ENSMUSG00000019992 |
71804 |
Mtfr2 |
protein_coding |
Q8VED8 |
FUNCTION: May play a role in mitochondrial aerobic respiration essentially in the testis. Can also promote mitochondrial fission. {ECO:0000269|PubMed:20568109}. |
Mitochondrion;Phosphoprotein;Reference proteome |
|
|
mmu:71804; |
mitochondrion [GO:0005739]; aerobic respiration [GO:0009060]; mitochondrial fission [GO:0000266]; mitochondrion organization [GO:0007005] |
20568109_The nuclear genes Mtfr1 and Dufd1 regulate mitochondrial dynamic and cellular respiration and may affect male fertility. |
ENSG00000146410 |
MTFR2 |
110.160239 |
2.2645075793 |
1.179197 |
0.26606360 |
19.550316 |
0.00000979845473618919624058439671321352193444909062236547470092773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000727694112830979316980037352458055011084070429205894470214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
145.031747003639 |
26.8988077044779 |
64.0456001688819 |
9.22226719966005 |
| ENSMUSG00000019997 |
14219 |
Ccn2 |
protein_coding |
P29268 |
FUNCTION: Major connective tissue mitoattractant secreted by vascular endothelial cells. Promotes proliferation and differentiation of chondrocytes (By similarity). Mediates heparin- and divalent cation-dependent cell adhesion in many cell types including fibroblasts, myofibroblasts, endothelial and epithelial cells (By similarity). Enhances fibroblast growth factor-induced DNA synthesis (By similarity). {ECO:0000250|UniProtKB:P29279, ECO:0000269|PubMed:10082563, ECO:0000269|PubMed:9184077}. |
Cell adhesion;Disulfide bond;DNA synthesis;Extracellular matrix;Heparin-binding;Reference proteome;Secreted;Signal |
|
|
mmu:14219; |
cell cortex [GO:0005938]; cis-Golgi network [GO:0005801]; cytosol [GO:0005829]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; perinuclear region of cytoplasm [GO:0048471]; fibronectin binding [GO:0001968]; growth factor activity [GO:0008083]; heparin binding [GO:0008201]; integrin binding [GO:0005178]; protein C-terminus binding [GO:0008022]; angiogenesis [GO:0001525]; calcium ion transmembrane import into cytosol [GO:0097553]; cartilage condensation [GO:0001502]; cell adhesion [GO:0007155]; cell migration [GO:0016477]; cell-matrix adhesion [GO:0007160]; chondrocyte differentiation [GO:0002062]; chondrocyte proliferation [GO:0035988]; connective tissue development [GO:0061448]; DNA biosynthetic process [GO:0071897]; extracellular matrix constituent secretion [GO:0070278]; fibroblast growth factor receptor signaling pathway [GO:0008543]; integrin-mediated signaling pathway [GO:0007229]; intracellular signal transduction [GO:0035556]; lung development [GO:0030324]; negative regulation of cell death [GO:0060548]; negative regulation of gene expression [GO:0010629]; ossification [GO:0001503]; positive regulation of cardiac muscle contraction [GO:0060452]; positive regulation of cell activation [GO:0050867]; positive regulation of cell death [GO:0010942]; positive regulation of cell differentiation [GO:0045597]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of collagen biosynthetic process [GO:0032967]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of G0 to G1 transition [GO:0070318]; positive regulation of gene expression [GO:0010628]; positive regulation of JNK cascade [GO:0046330]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of stress fiber assembly [GO:0051496]; reactive oxygen species metabolic process [GO:0072593]; regulation of chondrocyte differentiation [GO:0032330]; signal transduction [GO:0007165]; tissue homeostasis [GO:0001894] |
12036876_Cyr61 and CTGF are expressed in advanced atherosclerotic lesions of apoE-deficient mice. 12112457_CTGF is expressed during odontogenesis in mice at specific sites and times 12297288_ErbB4 is expressed in chondrocytes and may play some roles in chondrocytic growth and differentiation along with CTGF/Hcs24. 12571253_Gene expression regulation requires tgf-beta2 in fibroblasts. 12712324_connective tissue growth factor performs a dual role: a continuous function in the cardiovascular system, bone and cartilage-associated mesenchyme and maturing layer VII neurons 12842083_Overexpression of connective tissue growth factor is associated with breast cancer metastasis to bone 14633130_increased expression of fisp-12 in response to TGF-beta1 treatment and during epithelial to mesenchymal transition suggests that this gene may offer a potential therapeutic target against tubulointerstitial fibrosis 15256388_CTGF is an important mediator in the pathogenesis of pulmonary fibrosis in that it provides a local microenvironment in the lung that causes individual susceptibility to disease 15305294_CTGF may aid chondrogenesis by acting down-stream of transforming growth factor-beta and stimulating cell-cell interactions and expression of condensation-associated genes. 15315937_hypoxia response elements present upstream of Ctgf protein enable direct interaction of Hif-1 alpha transcription factor with the Ctgf promoter, resulting in increased transcription of Ctgf mRNA 15376321_The expression of Ctgf mRNA was mapped during mouse heart development, postnatal adult life, and after experimental myocardial infarction. 15496414_CTGF is regulated by Wnt and bone morphogenetic proteins signaling in osteoblast differentiation of mesenchymal stem cells 15574513_tubular CTGF acts as a downstream mediator of the profibrotic effects of TGF-beta1 in the remnant kidney 15699040_AngII regulates CTGF expression in cardiomyocytes through a PKC activation-mediated pathway 15862142_CTGF plays a role in pulmonary fibrosis; detection of CTGF may be an early and sensitive marker for evaluating the occurrence and development of pulmonary fibrosis 15980944_CTGF plays an important role in mediating renal interstitial fibrogenesis in response to high glucose and, as such, is a reasonable target for anti-fibrotic therapy. 15981023_After 15 days of development (E15), the expression of ctgf was detected in the zone of hypertrophy and provisional calcification, in which ossification proceeds toward the epiphysis during skeletal development. 16019426_Gene expression of Ctgf was studied in the adult mouse. 16186174_CTGF expression in the brain might have a role in Alzheimer's disease clinical progression and neuropathology 16343439_Taken together, these data indicate that the CTGF gene is a target of c-Maf and Lc-Maf in cartilage development. 16380465_study suggests a pathogenic role of Connective tissue growth factor (CTGF)in the development of diabetic nephropathy 16611331_Connective tissue growth factor (CTGF), which is induced by TGFbeta, is expressed only iin heart transplantations with chronic rejection. 16767377_Upregulation of CTGF expression was confirmed in RPE cells by oxidative stress in vitro and in vivo. CTGF might be one of the factors that induces the changes observed in aging retinas. 16889607_CTGF has potential as a biomarker of CAN, and also a therapeutic target in managing graft fibrosis. 17111364_Thus, CTGF is a downstream mediator of TGF-beta1-induced MC condensation through its effects on cell proliferation and migration. CTGF is also involved in up-regulating FN and suppressing Sox9 expression during TGF-beta1 induced MC condensation. 17197570_These results demonstrate that TGF-beta1 induces CTGF expression in mouse embryonic lung explants, that CTGF inhibits branching morphogenesis, and that CTGF expression is mediated by the TbetaRI/ALK-5-dependent Smad2 signaling pathway. 17215322_In endothelial cells CTGF gene transcription is controlled by alteration is cytoskeletal actin and serum response factor. 17350707_based on an acute CVB3 model of myocarditis, CTGF may mediate the development of fibrosis after viral myocarditis, and the cells expressing CTGF change during the course of viral myocarditis 17541935_Glucocorticoid induced expression of Ctgf contributes to lactogenic differentiation of mouse mammary epithelial cells. 17554073_Data show that specific down-regulation of connective tissue growth factor attenuates progression of nephropathy in mouse models of type 1 and type 2 diabetes. 17625227_Downregulation of CTGF levels does not affect neovascularization, indicating distinct roles of VEGF and CTGF in angiogenesis and fibrosis in eye conditions. 17657819_Smad7 overexpression in cultured hepatocytes abrogated TGF-beta-dependent and intrinsic CTGF expression. 17765657_We demonstrate that infection of epithelial cells with attenuated Yersinia enterocolitica lacking the virulence plasmid pYV leads to the expression of CYR61 and CTGF. 17846733_The significantly enhanced transcript levels of TGF-beta, CTGF, and procollagen type I in cultivated CVB3-infected primary cardiac fibroblasts substantiate the role of fibroblasts as a relevant cell population in cardiac remodeling processes 17875658_These findings suggest that the CCN2/CTGF is involved in establishing epiphyseal vascularization and remodeling, which eventually determines the secondary ossification center in the developing epiphysial cartilage. 17890868_Oxidative stress, collagen production, TGF-beta(1), connective tissue growth factor, ECM turnover are part of the comprehensive adaptation to nephron loss. 18075496_Overexpression of CTGF in podocytes is sufficient to exacerbate proteinuria and mesangial expansion through a functional impairment and loss of podocytes. 18201696_TGF-beta-induced activation of the Smad pathways, including Smad2 phosphorylation, Smad2/3 and Smad4 nuclear accumulation and Smad-dependent transcriptional responses, were unaffected by loss of CCN2. 18213577_The absence of Ctgf and/or its protein product, CTGF, may induce pulmonary hypoplasia by both disrupting basic lung developmental processes and restricting thoracic expansion. 18276598_FAK/Src signaling is a critical feature permitting chondrogenic differentiation and that CCN2 operates downstream of this loss to promote chondrogenesis. 18287562_the PARP-1/PARP-1 binding element complex functions as a nonspecific, fundamental enhancer of both basal and induced CCN2 gene transcription in tubular epithelial cells 18319604_In mouse fibroblasts, SGK1 transcript levels were significantly increased after angiotensin II treatment; both transcript and protein connective tissue growth factor (CTGF) increased 18474939_We conclude that CTGF expression is necessary for diabetes-induced BL thickening and suggest that reduction of CTGF levels may be protective against the development of diabetic retinopathy. 18535099_CTGF overexpression in vivo causes osteopenia, secondary to decreased bone formation, possibly by antagonizing BMP, Wnt, and IGF-I signaling and activity. 18596209_Connective tissue growth factor inhibits adipocyte differentiation. 18597638_CCN2 induces chondrocyte differentiation by regulating the expression of chondrocyte-associated genes but that these effects are counteracted by CCN3. 18632843_upregulation of CTGF inhibits BMP-7 signal transduction in the diabetic kidney and contributes to altered gene transcription, reduced MMP activity, glomerular basement membrane thickening and albuminuria, all of which are hallmarks of diabetic nephropathy 18835464_These results suggest that CCN2 regulates the expression of VEGF at a transcriptional level by promoting HIF-1alpha activity. 19096030_miR-133 and miR-30 regulate connective tissue growth factor: implications for a role of microRNAs in myocardial matrix remodeling. 19111553_CTGF could be a mediator of the profibrotic effects of ET-1 in cardiomyocytes. CTGF inhibitors should be considered in setting a comprehensive pharmacological approach towards ET-1 induced cardiovascular diseases 19131512_Unlike CTGF null embryos, CTGF heterozygotes survive past birth and exhibit a range of islet phenotypes, including an intermingling of islet cell types, increased number of glucagon(+) cells, and beta-cell hypertrophy. 19194549_hypoxia induces CTGF mRNA expression via a TGF-beta1-independent mechanism 19276073_activity of MMP-2 can play an important role in the initial steps of fibrosis and shows that FN levels can regulate the cellular response to CTGF. 19332661_Gene expression of Ctgf induced by cigarette smoke during cutaneous wound healing is strain dependent. 19385047_Suppression of TNF-alpha on TGF-beta-induced CTGF expression is due to the competition of p300 by p65 and Smad4. 19407155_CTGF mRNA expression was detected at 2 hours in osteocytes on the pressure side, followed by apoptosis at 6 hours after tooth movement in mice. 19423687_CTGF contributes to the recruitment of inflammatory cells in the kidney by activating the NF-kappaB pathway. 19498055_TAZ up-regulated Ctgf expression not only by reinforcing transforming growth factor-beta/smad signals, but also by interfering in the more proximal Ctgf promoter region (from bp -123 to -76), defined as the TAZ response element. 19500499_Data show that losartan can inhibit high glucose-induced CTGF expression in mouse mesangial cells, and the mechanisms maybe involve the interruption of ERK1/2 MAPK pathway. 19541844_These data demonstrate that overexpression of CTGF disrupts alveologenesis and capillary formation, and induces fibrosis during the critical period of alveolar development. 19565505_A direct profibrotic effect of CTGF and its contribution to lung fibrosis through transcriptional activation of Col1a2 has been demonstrated. 19657322_Transforming growth factor-beta2 upregulates sphingosine kinase-1 activity, which in turn attenuates the fibrotic response to TGF-beta2 by impeding CTGF expression. 19698122_THBS1, Cyr61 and CTGF were all over-expressed in esophageal squamous cell carcinoma. 19707545_data suggest that CTGF itself does not induce cardiac fibrosis. Moreover, it is involved in hypertrophy induction and cellular remodeling depending on the cardiac stress stimulus 19817698_CTGF can be upregulated by TGF-beta1 and appears to play an important role in the development of pulmonary fibrosis 19959709_Advanced glycation end-products induce tubular CTGF via TGF-beta-independent Smad3 signaling. 20018872_Exposure to low oxygen tension or treatment with the hypoxia-mimetic dimethyloxalyl glycine (DMOG) stabilized HIF-1alpha and up-regulated CTGF in human umbilical vein endothelial cells and in a murine microvascular endothelial cell line. 20053791_Conclude that forskolin stimulates CREB-mediated trans-activation of the Dot1 gene, which leads to hypermethylation of histone H3K79 at the CTGF promoter, and inhibition of CTGF transcription. 20213804_selective expression of CTGF in fibroblasts alone causes tissue fibrosis in vivo through specific signaling pathways 20235153_CTGF enhances NFAT signaling through the induction of cGKII and the phosphorylation of GSK3beta 20333347_These data suggest that the reduced expression of Hic-5 and concomitant reduced CTGF promoter activity may contribute to the anti-angiogenic effects of histone deacetylase inhibitors. 20406330_findings suggest CCN2 regulates integrin expression in primary culture hepatic stellate cells (HSC) and supports HSC adhesion via its binding of cell surface integrin alpha(5)beta(1) 20430035_CCN2 is pro-hypertrophic and -fibrotic, whereas CCN5 is anti-hypertrophic and -fibrotic. 20497571_enhances lactogenic differentiation of mammary epithelial cells 20534727_CTGF is necessary for normal skeletal development but to a lesser extent for postnatal skeletal homeostasis. 20571031_Differential occupancy of the TCF x LEF x Sox9 site by Sox9 versus beta-catenin restricts high levels of Ccn2 expression to hypertrophic chondrocytes. 20585662_Connective tissue growth factor (CTGF) was identified as a major regulator of osteogenesis associated with RhoA mediated cytoskeletal tension 20721934_CCN2 expressed during osteoclastogenesis promotes osteoclast formation via induction of and interaction with DC-STAMP 20877562_Ctgf expression reduced to 30% of wild type is survivable, while expression nine fold higher than wild type leads to embryonic lethality. 20936632_these results indicate that CCN2 is essential for bleomycin-induced skin fibrosis, likely due to a defect in myofibroblast recruitment. 20946117_CTGF downregulated the expression of E-cadherin through activation of the nuclear factor-kappaappa B (NF-kappaB) pathway. 21121985_CTGF may be a key mediator of early and persistent fibrosis in dystrophic cardiomyopathy. 21186275_This report provides novel evidence that CTGF confers cardioprotection by salvage phosphokinase signaling leading to inhibition of glycogen synthase kinase 3 beta, activation of phospho-SMAD2, and reprogramming of gene expression. 21239535_Overexpression of CTGF in alveolar type II epithelial cells results in lung pathology similar to those observed in infants with severe bronchopulmonary dysplasia. 21382976_Rad GTPase inhibits cardiac fibrosis through connective tissue growth factor in failing hearts. 21454550_CTGF specifically induced the synthesis of decorin, suggesting a mechanism of autoregulation. 21602138_CTGF is an important mediator of high glucose-induced podocyte damage and decreases the protein level of podocalyxin by the ERK1/2 pathway. 21654839_Overexpression of PTEN reduces the overexpression of type I collagen and CCN2 by dSSc fibroblasts 21747166_p53 induces CTGF expression and promotes liver fibrosis 21826667_CTGF acts downstream of muscle injury and also contributes directly to the deterioration of skeletal muscle phenotype and function in skeletal muscle dystrophy. 21868453_These findings demonstrate that CTGF is a downstream mediator in TGF-beta and progesterone signaling cascades and is necessary for normal follicle development and ovulation. 21876171_Loss of CTGF from either endothelial cells or beta cells results in decreased embryonic beta-cell proliferation. 21913214_CTGF and ILK were involved in high glucose-induced phenotypic alterations of podocytes. ILK acted as a downstream kinase of CTGF and high glucose-induced ILK expression might occur through CTGF-dependent and -independent pathways. 21925146_these findings support the chemoattractive role of CCN2 for P19 cells, but not its adhesive role when compared to laminin or fibronectin. 21969300_Ctgf is important for vessel growth during early retinal development and promotes the fibrovascular reaction in murine retinal ischemia after laser injury. 21978691_SPARC and connective tissue growth factor appeared to be involved in the same biological pathway 22321187_The increase of CTGF expression was associated with the severity of myocardial fibrosis in viral myocarditis. 22363445_CCN2 is required for the normal production of vascular basement membranes. 22532143_It conclude that in the murine atria, CCN1 and CCN2 proteins are expressed constitutively. In chronic heart failure, CCN proteins tend to be upregulated, which may be related to the action of angiotensin II. 22542845_Connective tissue growth factor causes glaucoma by modifying the actin cytoskeleton of the trabecular meshwork. 22563064_SRF plays a key role in the modulation of cardiac fibrosis through repression of cardiomyocyte CTGF expression in a paracrine fashion. 22611085_These results provided mechanistic insight into the angiogenic action of CCN2/CTGF and demonstrated the therapeutic potential of dominant-negative CCN2/CTGF mutants for antiangiogenesis. 22749815_The MAPKs/TGFb1/TRAF6 pathway is an important signaling pathway in AngII-induced CTGF expression. 22885106_Lysophosphatidic acid signals through LPA2 receptors and Galphaq proteins of cultured proximal tubule cells to transactivate latent TGF-beta in a Rho/Rho-kinase and alphavbeta6 integrin-dependent manner. 22913183_CTGF but not TGFb1 mRNA expression in the liver is significantly related to the duration of infection in schistosomal hepatic fibrosis. 22993071_Bladder remodeling corresponding to experimental autoimmune encephalomyelitis severity may be due to enhanced expression of CTGF and increased growth of connective tissue. 23048035_CTGF regulates retinal neovascularization through p53 protein-dependent transactivation of the MMP-2 gene. 23073844_The skeletal site-specific role of connective tissue growth factor in prenatal osteogenesis. 23199377_Hemizygous deletion of CTGF/CCN2 does not suffice to prevent fibrosis of the severely injured kidney. 23212098_Subcutaneously implanted, MSC-loaded polyvinyl alcohol (PVA) sponges and stented excisional wounds were used as wound models to study the dynamics of CTGF expression. 23284892_Increased myocardial CTGF activities after MI are associated with attenuation of LV remodeling and improved LV function mediated by attenuation of inflammatory responses and inhibition of apoptosis. 23287510_CTGF contributes to fibrocyte proliferation in the myocardium and enhances fibrocyte differentiation into a myofibroblast phenotype responsible for extracellular matrix deposition. 23322166_LPA1-induced cytoskeleton reorganization makes a critically important contribution to the profibrotic activities of LPA by driving MRTF-dependent CTGF expression, which, in turn, drives fibroblast proliferation 23414741_ET1 contributes to AF-dependent atrial fibrosis by synergistic activity with AngII to stimulate SGK1 expression and enhance phosphorylation of the SGK1 protein which, in turn, induces CTGF. 23452880_CTGF attenuates cardiac hypertrophy upon chronic pressure-overload due to inhibition of signaling mechanisms that promote pathologic myocardial hypertrophy. 23525012_promotes epithelium-to-mesenchyma transition morphological changes in fibroblasts 23530034_findings reveal a negative feedback loop between CCN2 and HIF-1alpha in NP cells and demonstrate a role for CCN2 in maintaining matrix homeostasis in this tissue. 23555635_Cartilage-specific over-expression of CCN2 stimulates insulin-like growth factor expression and bone growth. 23564449_hnRNP-K regulates extracellular matrix, cell motility, and angiogenesis pathways. Involvement of the selected genes (Cck, Mmp-3, Ptgs2, and Ctgf) and pathways was validated by gene-specific expression analysis 23571711_CCN-2 plays a role in adipocyte differentiation in vivo and thus in the pathogenesis of obesity linked with insulin resistance. 23673415_ACE inhibition might improve muscle strength and decrease fibrosis by diminishing specifically CTGF expression and activity without affecting TGF-beta1 signaling. 23741006_The data suggest an important role of CTGF in mesenchymal stromal cells differentiation into adipocytes and of leptin in homing and progression of leukemia. 23756156_CCN2 plays a key role in extracellular matrix gene expression in severe hypertrophic cardiomyopathy and heart failure. 23778361_this study uncovers a novel mechanism controlling beta-adrenergic receptor responsiveness in cardiomyocytes involving connective tissue growth factor mediated regulation of GRK5. 23816882_Ctgf is a downstream target of the Smad-dependent TGF-beta signaling pathway in the developing palate. 23839921_Impaired intervertebral disc development and premature disc degeneration develops in mice with notochord-specific deletion of CCN2. 23868935_suggest a critical role for LRP1 in maintaining the integrity of vessels by regulating protease activity as well as matrix deposition by modulating HtrA1 and connective tissue growth factor protein levels. 23904456_Reducing CTGF/CCN2 slows down mdx muscle dystrophy and improves cell therapy. 23950936_genetic variability in CTGF expression directly modulates the severity of diabetic nephropathy. 23951098_CCN family member 2/connective tissue growth factor (CCN2/CTGF) has anti-aging effects that protect articular cartilage from age-related degenerative changes. 23983074_Loss of CCN2 resulted in resistance to the increases in collagen production and myofibroblast recruitment that are caused by loss of PTEN. 23985903_show that ROS accumulation correlates with media layer degeneration and increased connective tissue growth factor (CTGF) expression 23993699_This study showed that the odorant-specific recruitment of distinct glomeruli resulted in enhanced local CTGF expression levels in the activated glomeruli. Collectively our data reveal a molecular 24122827_Exosomal transfer of miR-214 is a paradigm for the regulation of CCN2-dependent fibrogenesis and identifies fibrotic pathways as targets of intercellular regulation by exosomal miRs. 24152728_CCN2 expression by dermal papillae cells is a physiologically relevant suppressor of hair follicle formation by destabilization of beta-catenin and CCN2 normally acts to maintain stem cell quiescence. 24155087_application of exogenous CCN2 protein caused ERK1/2 activation, and the neutralizing CCN2 antibody inhibited loading-induced ERK1/2 activation. 24161906_Loss of ACE2 exacerbates AngII-mediated inflammation, myocardial injury and dysfunction in ACE2-deficient hearts via activation of the CTGF-FKN-ERK and MMP signaling. 24217924_CTGF is involved in structural retinal vascular changes in diabetic rodents. 24288211_These findings uncover a novel functional role of CCN2 as a metabolic supporter in the growth-plate chondrocytes, which is required for skeletogenesis in mammals. 24380865_The PDZ-binding motif of YAP is critical for YAP-mediated oncogenesis, and that this effect is mediated by YAP's co-activation of TEAD-mediated CTGF transcription. 24464932_In a cardiomyocyte cell line, CTGF was secreted in response to cell stretching on a flexible membrane base (a cellular phenotype of cardiac diastolic dysfunction). 24508728_Deletion of CTGF had an attenuated fibrotic response to bleomycin. 24627144_Connective tissue growth factor is expressed in the naive cornea, lens, iris, and retina, and is expressed immediately after epithelial injury. Loss of CTGF impairs efficient re-epithelialization of corneal wounds. 24688123_Data indicate that connective tissue growth factor (CTGF) is expressed in cardiomyocytes and cardiac Fibroblasts. 24727816_This study provides a better understanding of the functions of connective tissue growth factor within the bone marrow, showing the dual regulatory role of the growth factor in skeletogenesis and in stage-specific B lymphopoiesis. 24754049_Demonstrate a novel role for CTGF/CCN2 during pancreatitis and its ability to modulate pro-inflammatory pathways. 24792956_Ctgf is a target of Notch canonical signaling in osteoblasts, and may act in concert with Notch to regulate skeletal homeostasis 24882759_CCN2 is packaged into secreted, nano-sized exosomes that mediate its intercellular transfer between hepatic stellate cells. 24910328_MRTF-A promotes microvessel growth (via CCN1) and maturation (via CCN2), thereby enabling functional improvement of ischaemic muscle tissue 25027872_Transverse aortic constriction induced the cardiac expression of profibrotic connective tissue growth factor and attenuated the expression of SERCA2a (sarcoplasmic reticulum Ca2+-ATPase) in knockout mice 25108224_Descriptive Statement Elimination of TGF-betaIIR is not sufficient to completely prevent liver fibrosis. TGF-beta-independent mechanism of type I collagen production and suggest connective tissue growth factor as its potent mediator. 25203810_functional connection between CTGF and integrin alphavbeta6 during hepatic progenitor/oval cell activation and associated fibrosis 25457224_CTGF exerts profibrotic action in myoblasts via the up-regulation of sphingosine kinase-1/S1P3 signaling axis in TGF-beta dependent manner. 25464947_CTGF reporter mice selectively identify a subpopulation of bone marrow mesenchymal progenitor cells that reside in the trabecular bone region. 25554597_findings suggest CCN2 as a candidate of the fourth factor in the RANK/RANKL/OPG system for osteoclastogenesis, which regulates OPG and RANK via direct interaction 25580742_CTGF overexpression induces epithelial to mesenchymal transition in mouse primary AT II cells, which is mediated by ILK 25644104_Report showed that loss of PTEN expression resulted in collagen deposition in the lung in a CCN2-dependent manner supporting the notion that alterations in PTEN expression contribute to fibrogenesis and that CCN2 mediates collagen production in fibrosis. 25712640_CCN genes are activated in heart failure. 25870108_Neither heart-specific Ctgf deletion nor CTGF overexpression altered cardiac remodeling and function with aging or after multiple acute stress stimuli. 25955164_knockdown of MRTF-A synthesis abolishes the systemic sclerosis myofibroblast enhanced basal contractility and synthesis of type I collagen and inhibits the matricellular profibrotic protein, connective tissue growth factor CCN2/CTGF 26168233_CCN2 up regulation by tumor stromal cells is responsible for the melanoma metastasis. 26171399_Data suggest that, in mice with type 1 diabetes mellitus and diabetic nephropathy, up-regulation of urinary CTGF level is independently associated with biomarkers of proximal and distal tubular dysfunction and pathology. 26257513_CTGF siRNA ameliorates tubular cell apoptosis and tubulointerstitial fibrosis in obstructed mouse kidneys in a Sirt1-independent manner. 26364758_Under physiological conditions, CCN2 may be regulating the levels of free amino acids in the extracellular matrix of cartilage. 26392029_In cultured neonatal cardiac fibroblasts, p63RhoGEF regulated the angiotensin II (Ang II)-dependent RhoA activation, the activation of the serum response factor, and the expression and secretion of CTGF. 26410398_CTGF knockout does not affect cardiac hypertrophy and fibrosis formation upon chronic pressure overload. 26440278_these findings reveal a key role of the SRF/CTGF/miR-133a axis in the regulation of cardiac fibrosis 26447680_Data suggest that PTEN phosphatase (PTEN) regulates renal extracellular matrix production via activated protein kinase B (Akt) and increased connective tissue growth factor (CTGF) in diabetes mellitus. 26549358_CTGF inhibition may benefit patients with dilated cardiomyopathy. 26554857_Down-regulation of CTGF is effective in inhibiting postoperative scarring in vivo. This suggests that RNAi with CTGF siRNA may potentially pave the road for a novel therapeutic strategy to improve glaucoma surgery results. 26686381_These results suggest that TGF-beta1 and CTGF may be involved in the process of denervated skeletal muscle fibrosis. 26743816_These data indicate that LPA increases CCN2 expression through the activation of PKC and PKA. Thus, the regulatory functions of the PKC and PKA pathways are implicated in the LPA-induced increase in CCN2 expression 26775556_these results suggested that loss of miR-30c may contribute to the pathogenesis of diabetic nephropathy (DN) by inhibiting target CTGF expression; replenishing miR-30c may ameliorate renal structure and function by reducing renal fibrosis in DN. 26825658_These results indicate that tensile force induces in vivo gene expression associated with vascularization early in tensile-force-induced sutural bone formation. Moreover, the early induction of Vegf gene expression is regulated by CTGF and ROCK2. 26984259_CTGF and beta-catenin are involved in the epithelial-mesenchymal transition of podocytes in diabetes. CTGF mediates high-glucose induced EMT through activation of beta-catenin in podocytes. 26991260_beta-catenin signaling inhibition protects against CTGF-induced alveolar and vascular pathology in neonatal mouse lung 27126736_RhoA determines lineage fate of mesenchymal stem cells by modulating CTGF-VEGF complex in extracellular matrix. 27131347_these findings show that cardiac ERK1/2 activity is modulated in part by TGF-b/Smad signaling, leading to altered activation of CTGF/CCN2 to mediate fibrosis and alter cardiac function. This identifies a novel mechanism in the development of LMNA cardiomyopathy. 27179776_CTGF is overexpressed in gastric cancer and adjacent tissue compared to normal gastric tissue. Gastrin induces expression of CTGF in gastric epithelial cells. 27436052_Ctgf is the direct target gene of SOX9 in chondrocytes and nucleus pulposus cells. 27558559_In summary, our data suggest that in obstructive nephropathy atrophy increases and fibrosis decreases with age and that this relates to increased BMP signaling, most likely due to higher BMP6 and lower CTGF expression. 27662798_CCN2 suppression by miR-199a-5p accounts, in part, for low-level fibrogenic gene expression in quiescent hepatic stellate cells (HSCs) and causes dampened gene expression in activated HSCs after horizontal transfer of miR-199a-5p in exosomes from quiescent HSCs. 27927603_LPA-LPA1 signaling initiates profibrotic epithelial cell fibroblast communication mediated by epithelial cell derived connective tissue growth factor. 28079129_miR-26b-5p interacted with 3'UTRs of Col1a2 and CTGF, and circ_000203 could block the interactions of miR-26b-5p and 3'UTRs of Col1a2 and CTGF. 28183733_the mechanistic target of rapamycin (mTOR) pathway in neurons regulates CTGF production and secretion, revealing a paracrine mechanism by which neuronal signaling regulates oligodendrocyte maturation and myelination in tuberous sclerosis complex (TSC). 28191821_These results reveal a novel mechanism of macrophage migration into glomeruli with nephritis mediated by connective tissue growth factor derived from mesangial cells. 28287886_Overexpression of CTGF in cardiomyocytes attenuates left ventricular hypertrophy in angiotensin II induced hypertension. 28331224_These results indicate that the hepatocytic expression of TGF-beta and CTGF is mediated by Wnt signalling in Schistosoma japonicum infection. 28370212_this study suggested that CTGF antibody protected podocytes against injury in DN mice by reducing beta-catenin overexpression and preventing podocyte EMT, which might provide new insight into the mechanism of CTGF inhibition in the treatment of DN. 28460577_CTGF and BMP2 are induced following myocardial ischemia in mice and humans. 28545716_As shown in mouse model of kidney fibrosis CTGF is significantly involved in fibrosis-associated renal lymphangiogenesis through regulation of, and direct interaction with, VEGF-C. 28562688_Notch1 haploinsufficiency decreased the expression of Ctgf in the aorta and in vitro cell culture system. In vitro studies on SMCs using the Notch1 intracellular domain (NICD) plasmid, dominant negative mastermind-like (dnMAML), or specific siRNA suggest that Notch1, not Notch3, directly modulates the expression of CTGF 28710437_Inhibition of CTGF ameliorates peritoneal fibrosis through suppression of fibroblast and myofibroblast accumulation and angiogenesis. 28753062_CTGF role in cardiac fibrosis. Long noncoding RNA H19 mediates CTGF expression. 28782260_The data demonstrate that MMP13 and CTGF play a crucial role in modulation of fibrogenic mediators while promoting hepatic fibrogenesis. 28825193_p-SMAD2/3 and p-ERK1/2 might play a regulatory role in TGF-beta1 induced CTGF exp p-SMAD2/3 and p-ERK1/2 might play a regulatory role in TGF-beta1 induced CTGF expression during tooth development. 29111856_Data (including data from studies using transgenic mice) suggest that Ctgf secreted from vascular endothelium in pancreas plays critical role in up-regulation of insulin secretion in pancreatic beta-cells during pregnancy; here, pregnant mice with global Ctgf haplo-insufficiency (heterozygous for loss-of-function mutation) (a) exhibit impairment in maternal beta-cell proliferation and (b) develop gestational diabetes. 29160908_EGFR regulates CCN2 fibrotic signalling in the kidney. 29165602_High CTGF expression is associated with peritoneal fibrosis. 29575960_early myocardial CTGF mRNA expression (six hours) after Ang-II exposure is likely dependent on latent TGF-beta activation via the canonical Smad-dependent pathway in resident cardiac cells. 29683732_miR-18a was decreased during lung fibrosis in vitro and in vivo, as well as in patients with IPF. Moreover, knockdown of miR-18a led to fibrogenesis in lung fibroblasts, whereas enhanced expression of miR-18a attenuated TGF-beta1-induced lung fibrosis by directly targeting the regulation of connecting tissue growth factor. 29860398_we also found that CTGF/CCN2 is expressed in astrocytes and neurons, predominantly in dorsal areas of spinal cord from |
ENSG00000118523 |
CCN2 |
412.453678 |
0.1824490638 |
-2.454434 |
0.16181089 |
223.339885 |
0.00000000000000000000000000000000000000000000000001690027536703011186223481155348239104965875815804162190709458944495478128610944431823809672962325744239222648251230690722286776825344922770000266609713435173034667968750000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000028602777154511408111256187279116730893634975893640983660110220744522044019845316339027843422737796670479174253576057211125027723852554117911495268344879150390625000000000000000000000000000000000000000000000000000 |
Yes |
No |
126.610842727735 |
14.2303065300344 |
693.951725992318 |
53.228553481594 |
| ENSMUSG00000020019 |
57764 |
Ntn4 |
protein_coding |
Q9JI33 |
FUNCTION: May play an important role in neural, kidney and vascular development. Promotes neurite elongation from olfactory bulb explants. {ECO:0000269|PubMed:11038171}. |
3D-structure;Basement membrane;Disulfide bond;Extracellular matrix;Glycoprotein;Laminin EGF-like domain;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:57764; |
basement membrane [GO:0005604]; extracellular region [GO:0005576]; laminin complex [GO:0043256]; plasma membrane [GO:0005886]; laminin-1 binding [GO:0043237]; animal organ morphogenesis [GO:0009887]; basement membrane assembly [GO:0070831]; cell migration [GO:0016477]; neuron remodeling [GO:0016322]; regulation of branching involved in salivary gland morphogenesis by extracellular matrix-epithelial cell signaling [GO:0060668]; substrate adhesion-dependent cell spreading [GO:0034446]; tissue development [GO:0009888] |
17588941_netrin-4 is a component of basement membranes and is integrated into the laminin polymer via interactions with the laminin gamma1 andgamma3 short arms. 18719102_Netrin-4 significantly reduced pathological angiogenesis in Matrigel and laser-induced choroidal neovascularization models. Netrin-4, neogenin, and Unc5B receptor expression was up-regulated in choroidal neovessel EC after laser injury 18985053_Netrin-4 may improve poststroke functional recovery by enhancing blood vessel proliferation 19193855_netrin-4 is produced by selected GFAP-positive astrocytes positioned close to newborn neurons migrating in the anterior part of the rostral migratory stream and within the olfactory bulb 20407033_In vivo, Netrin-4 induces growth of lymphatic and blood vessels in the skin of transgenic mice. 21406174_NT-4 overexpression decreases tumor growth and carcinomatosis, probably via an antiangiogenic effect 21799154_The alpha6beta1 integrin is a netrin-4 receptor in endothelial cells. 23124703_Loss of Netrin-4 is associated with metastatic colorectal tumor progression. 24846137_Vascular endothelial cells interact with bone in suppressing bone through netrin-4. 25853509_These results indicate that netrin-4 shed new light on its potential roles in treatmenting for angiogenic diseases that affect the ocular surface, as well as other tissues 26732856_Our results indicate a role for netrin-4 as an angiogenesis modulating factor in O2-dependent vascular homeostasis while being less important during normal retinal developmental angiogenesis or during inflammatory neovascularization. 27901020_netrin-4 disrupts laminin networks and basement membranes through high-affinity binding to the laminin gamma-1 chain. 28253401_Netrin-4 acted as an antiangiogenic factor in the cornea, with which the healthy cornea is enriched via its expression by corneal epithelial cells. 28882944_we found that Ntn4 promotes differentiation and migration of osteoblasts. 33923095_Anti-Inflammatory Role of Netrin-4 in Diabetic Retinopathy. 35687692_The risk variant rs11836367 contributes to breast cancer onset and metastasis by attenuating Wnt signaling via regulating NTN4 expression. |
ENSG00000074527 |
NTN4 |
19.397223 |
0.0461800448 |
-4.436587 |
0.88159704 |
28.550062 |
0.00000009130689116652129078015363936732251559647011163178831338882446289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000009664951815486117731821566825822777957455400610342621803283691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.59004843738107 |
1.03325117729922 |
34.4315048965466 |
11.584896002472 |
| ENSMUSG00000020121 |
117600 |
Srgap1 |
protein_coding |
Q91Z69 |
FUNCTION: GTPase-activating protein for RhoA and Cdc42 small GTPases. Together with CDC42 seems to be involved in the pathway mediating the repulsive signaling of Robo and Slit proteins in neuronal migration. SLIT2, probably through interaction with ROBO1, increases the interaction of SRGAP1 with ROBO1 and inactivates CDC42 (By similarity). {ECO:0000250}. |
3D-structure;Coiled coil;GTPase activation;Phosphoprotein;Reference proteome;SH3 domain |
|
|
mmu:117600; |
cytoplasm [GO:0005737]; GTPase activator activity [GO:0005096]; small GTPase binding [GO:0031267]; cell migration [GO:0016477]; negative regulation of cell migration [GO:0030336]; Rho protein signal transduction [GO:0007266] |
19137586_Data show that srGAP3 is expressed in ventricular zones of neurogenesis in many different tissues of the central nervous system. 22271578_srGAP1 and srGAP3 are largely expressed in subpopulations of dorsal root ganglion neurons following sciatic nerve lesion. 26026792_Mutations of the SLIT2-ROBO2 pathway genes SLIT2 and SRGAP1 confer risk for congenital anomalies of the kidney and urinary tract 33514561_SRGAP1 Controls Small Rho GTPases To Regulate Podocyte Foot Process Maintenance. |
ENSG00000196935 |
SRGAP1 |
227.640778 |
0.1590827534 |
-2.652151 |
0.25501770 |
103.344365 |
0.00000000000000000000000281674573111969645648815968601367241912158889660867593453575744044149742340010789121151901781558990478515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000001479471412979835076709096789535026334717271978767945267294525377166358737213158747181296348571777343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
58.7087423371601 |
10.4317665292605 |
369.045299261553 |
45.1386659303948 |
| ENSMUSG00000020140 |
14160 |
Lgr5 |
protein_coding |
Q9Z1P4 |
FUNCTION: Receptor for R-spondins that potentiates the canonical Wnt signaling pathway and acts as a stem cell marker of the intestinal epithelium and the hair follicle. Upon binding to R-spondins (RSPO1, RSPO2, RSPO3 or RSPO4), associates with phosphorylated LRP6 and frizzled receptors that are activated by extracellular Wnt receptors, triggering the canonical Wnt signaling pathway to increase expression of target genes. In contrast to classical G-protein coupled receptors, does not activate heterotrimeric G-proteins to transduce the signal. Involved in the development and/or maintenance of the adult intestinal stem cells during postembryonic development. {ECO:0000269|PubMed:21727895}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Golgi apparatus;Leucine-rich repeat;Membrane;Receptor;Reference proteome;Repeat;Signal;Transducer;Transmembrane;Transmembrane helix |
|
The protein encoded by this gene is a leucine-rich repeat-containing receptor (LGR) and member of the G protein-coupled, 7-transmembrane receptor (GPCR) superfamily. The encoded protein is a receptor for R-spondins and is involved in the canonical Wnt signaling pathway. This protein plays a role in the formation and maintenance of adult intestinal stem cells during postembryonic development. [provided by RefSeq, Sep 2015]. |
mmu:14160; |
Golgi apparatus [GO:0005794]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; trans-Golgi network membrane [GO:0032588]; protein-hormone receptor activity [GO:0016500]; transmembrane signaling receptor activity [GO:0004888]; epithelial cell proliferation involved in renal tubule morphogenesis [GO:2001013]; G protein-coupled receptor signaling pathway [GO:0007186]; hair follicle development [GO:0001942]; inner ear development [GO:0048839]; oocyte differentiation [GO:0009994]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; regulation of cell population proliferation [GO:0042127] |
15509778_physiological role of the LGR5 signaling system during embryonic development 17934449_using two knock-in alleles, exclusive expression of Lgr5 in cycling columnar cells at the crypt base of the intesine is revealed 18849992_Lgr5(+) cells comprise an actively proliferating and multipotent stem cell population able to give rise to new hair follicles and maintain all cell lineages of the hair follicle over long periods of time. 19136944_eQTL analysis of normal skin suggested allelic variations in Lgr5 influence a correlated expression network of follicle keratins and transcription factors 19197002_Lgr5 has been recently identified as a novel stem cell marker of the intestinal epithelium and the hair follicle[REVIEW] 19329995_intestinal crypt-villus units are self-organizing structures, which can be built from a single Lgr5(+) stem cell in the absence of a non-epithelial cellular niche 19394326_Data identify LGR5 as a negative regulator of the Wnt pathway in the developing mouse intestine. 19478326_lineage tracing during a 14-month period revealed that Lgr5(+ve) cells located in the bulge of the hair follicle sustained multiple rounds of hair growth 19797400_Lgr5 is highly expressed in the uterine epithelium of immature mice and is dramatically down-regulated after the mice resume estrous cycles. 19806668_Lgr5 is a marker of incisor stem cells but is not regulated by Wnt signaling in the incisor. 20085740_In neonatal stomach, Lgr5 was expressed at the base of prospective corpus and pyloric glands, whereas expression in the adult was predominantly restricted to the base of mature pyloric glands. 21113151_Lgr5 stem cells compete for essential niche signals provided by a specialized daughter cell, the Paneth cell 21472479_Lgr5 and Axin2 differential expression among cell populations highlights the dynamic but complex distribution of Wnt-activated cells in and around the embryonic and postnatal cochlea. 21727895_Lgr5 homologues are facultative Wnt receptor components that mediate Wnt signal enhancement by soluble R-spondin proteins 21927002_Bmi1-expressing stem cells may represent both a reserve stem cell pool in case of injury to the small intestine epithelium and a source for replenishment of the Lgr5-expressing cells under non-pathological conditions 21997934_Lgr5 expression is Myb-dependent. 22002312_Lgr5 has been identified as a marker of Wnt-regulated adult stem cell populations in the hair-follicle, intestine and stomach. A closely-related protein, Lgr6 marks adult stem cells responsible for fueling the renewal of the sebaceous gland and skin.[Review] 22028024_Lhx2 differentially regulates Sox9, Tcf4 and Lgr5 in hair follicle stem cells to promote epidermal regeneration after injury. 22190486_Lgr5 is a marker for mitotically active crypt intestinal stem cells that exhibit exquisite sensitivity to canonical Wnt modulation, contribute robustly to homeostatic regeneration, and are quantitatively ablated by irradiation. 22198711_While chief cells with Lgr5 transcriptional activity are present along the lesser curvature of the gastric oxyntic mucosa, they are not responsible for production of metaplasia. 22355124_Lgr5 receptor-expressing intestinal stem cells function in the absence of Paneth cells 22355691_In SACK-expanded hair follicle stem cells, nuclear Lgr5 is expressed in both sisters during symmetric self-renewal divisions, which are associated with random chromosome segregation. 22723265_CD24-based sorting in wild-type mice isolates a colonic epithelial fraction highly enriched in actively cycling and quiescent Lgr5 positive putative colonic epithelial stem cells. 22787049_This study showed here that Lgr5, a marker for adult stem cells, was expressed in a subset of supporting cells in the newborn and adult murine cochlea 22926799_There is a stage-specific contribution of LGR5-expressing cells to luminal and basal epithelial lineages during postnatal mammary gland development in mice. 22945646_large subset of the Lgr5(+) stem cell progeny, expressing K15 and P-cadherin, is aberrantly mobilized to the upper region of hair follicles and the epidermis 22999937_Lgr5 expression remained restricted to cell clusters within developing nephrons in the cortex until postnatal day 7, when expression was permanently silenced. 23142137_TROY is up-regulated in human colorectal cancer cell lines and in intestinal tumors in mice. It functions as a negative modulator of the Wnt pathway in LGR5-positive stem cells. 23167464_Stem cell marker Lgr5 protein was found in clusters of epithelial cells of periodontal ligament in aging mice, suggesting a maintenance role for epithelial stem cells throughout the life cycle. 23352663_The outstanding regenerative potential of a rare population of Lgr5-expressing (Lgr5(+)) mammary epithelial cells. 23354049_Lgr5 is not expressed in healthy adult liver; however, small Lgr5(+) cells appear near bile ducts upon liver damage, coinciding with robust activation of Wnt signalling 23423968_The expression of Lgr5 protein is not a prerequisite for proper thymus organogenesis. 23475985_Lgr5 can be considered a universal SSC marker. 23553959_The results suggested that CD61 signaling is required for the proper growth of the cervical loop and for the promotion of the proliferation of pre-ameloblastic cells through Lgr5. 23824276_demonstrates that Lgr5 cells give rise to both anterior and posterior taste buds during development, and are capable of regenerating posterior taste buds after injury induced by glossopharyngeal nerve transection 23918377_Wnt/beta-catenin can drive Lgr5-positive cells to act as hair cell progenitors, even after their exit from the cell cycle and apparent establishment of cell fate. 24340024_Lgr5(+) cells are seen in a large number of gastric tumors ; their frequent basal arrangements and coexpression of ISC markers support the idea that Lgr5(+) cells act as stem cells during the early stage of intestinal-type gastric tumorigenesis. 24469993_LGR5 is critical for corpora lutea function and successful pregnancy in mice 24616052_The members of the R-spondin family are known as activators of Wnt signaling, and Lgr4, Lgr5, and Lgr6 have been identified as receptors for R-spondins. 24672754_These data suggest that the neonatal mammalian cochlea has some capacity for hair cell regeneration following damage alone and that Lgr5-positive cells act as hair cell progenitors in the cochlea. 24680895_Lgr4KO and Lgr4/5KO results in complete loss of intestinal stem cells in embryos, impairs proliferation of developing kidney and skin epithelium, and depletes Lgr5+follicular epithelium and impairs hair follicle development. 24920630_This report that Lgr5, a recently discovered adult stem cell marker, is exclusively expressed in GBCs in neonatal and adult mice 24997521_Using in vivo lineage tracing, identifed embryonic and neonate Lgr5(+) populations as stem/progenitor cells contributing to the development of the ovary surface epithelium cell lineage, as well as epithelia of the mesovarian ligament and oviduct/fimbria. 25158167_LGR5 is required for correct timing of germ cell differentiation and is found in cells that give rise to adult cortical granulosa cells. 25368147_Organoids that continue to express Lgr5 are multipotent.all three Lgr transcripts Lgr4-Lgr6 were detected in sorted Lgr5+ cells from posterior tongue and in sorted Lgr6+ cells from either posterior or anterior tongue. 25609789_Positioning within the niche and levels of LGR5 expression discriminate intestinal intestinal stem and progenitor cells with different Wnt/beta-catenin signaling activity. 25725293_H pylori colonize and manipulate the progenitor and Lgr5(+)stem cell compartments, which alters turnover kinetics and glandular hyperplasia. 25968904_Myh9 mediates colitis-induced epithelium injury by restricting Lgr5+ stem cells 25990970_The generation of new retinal cells, including retinal neurons and Muller glia from Lgr5-expressed amacrine cells, begins in early adulthood and continues as the animal ages. 26362184_Lgr6 and Bmi1 have roles in stem cells in the acral epithelium that participate in the long-term maintenance of sweat glands, ducts, and interadnexal epidermis, while Lgr5 has a role in rapid cycling and maintance of sweat glands only 26505972_Endogenous hyaluronic acid increases Lgr5(+) stem cell proliferation, crypt fission, and intestinal lengthening and that these effects are dependent on signaling through CD44 and TLR4. 26582901_Report targeting LGR5+ tumor stem cells with an antibody-drug conjugate as effective treatment for colon cancer. 26831517_Genetic fate-mapping of Alkaline phosphate intestinal(+) cells before or during targeted ablation of Lgr5-expressing stem cells generated numerous long-lived crypt-villus 'ribbons,' indicative of dedifferentiation of enterocyte precursors into Lgr5(+) stems. 26865184_Our studies show for the first time that Lgr5 is expressed in the NC cells at embryonic day 9.5 (E9.5) and is maintained during embryonic development and postnataly in the stroma of the ventral tongue, and some areas of the oral mucosa and that Lgr5+ cells participate in the maintenance of the stroma. 27088858_RSPO-LGR4/5-ZNRF3/RNF43 module controls metabolic liver zonation and is a hepatic growth/size rheostat during development, homeostasis and regeneration. 27195676_Arrb1 reduced the chemotherapy-induced Lgr5 stem cell apoptosis by inhibiting endoplasmic reticulum stress-mediated mitochondrial apoptotic signaling. 27328992_Results show that Lgr5 is expressed at the neuromuscular junction (NMJ) and is a receptor for Rspo2. 27348469_Data show that both the number and relative position of Paneth cells and Lgr5+ cells are important for fission. 27524622_Single-cell transcript profiles reveal multilineage priming in early progenitors derived from Lgr5-expressing intestinal stem cells. 27573849_sorted Reg4(+) DCS cells promote organoid formation of single Lgr5(+) colon stem cells. DCS cells can be massively produced from Lgr5(+) colon stem cells in vitro by combined Notch inhibition and Wnt activation 27773817_adult stem cell marker leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5) is expressed in the mice nasal respiratory epithelium. 28202528_Employing Lgr5-lacZ transgenic mice and Lgr5 in situ hybridization, authors found colonic epithelial stem cells (CESC) markedly more radioresistant in vivo than small intestinal crypt base columnar stem cells. 28228258_The Lgr5-expressing cells differentiate into hair cells in high yield. 28285904_Mex3a defines a reserve-like intestinal stem cell population within the Lgr5+ compartment. 28358093_role for Lgr5(+) cancer stem cells in primary and metastatic colon cancer 28553937_Stem cell plasticity enables hair regeneration following Lgr5(+) cell loss. 28581476_Lgr5(+) chief cells are involved in maintaining the homeostatic stem cell pool. Define Lgr5(+) chief cells as a major cell-of-origin of gastric cancer. 28880275_LGR5 promotes cancer stem cell traits and chemoresistance in cervical cancer. 30465020_NHE8 deficiency resulted in increased Lgr5 expression in the colon. 30503340_in the case of intestinal organoids from Lgr5-EGFP-IRES-CreERT2 transgenic mice, in vitro expansion of Lgr5 expression is limited in a culture condition supplemented with essential proteins 30553965_PERK knock-down could significantly diminish the number and proliferation of Lgr5-positive cells, induce the low expression of several proliferation markers and the high expression of several differentiation markers in Lgr5-positive cells, enhance the apoptotic Lgr5-positive cells, and reduce the Akt expression in intestinal Lgr5-positive stem cells 30713072_At 4 to 5 weeks of fetal age, the LGR5(+) population appears to emerge specifically in the proximal outflow tract 30725351_An enhancement in Lgr5(+ ve) crypt stem cells. 30991839_MSC(Lgr5) exhibited enhanced therapeutic efficacy for fracture healing. 31142540_Lgr5 may have a role in embryonic liver development and Lgr5(+) hepatoblasts can be clonally expanded in vitro into embryonic liver organoids 31580946_The vanillin derivative VND3207 protects intestine against radiation injury by modulating p53/NOXA signaling pathway and restoring the balance of gut microbiota. 31772170_Lgr5 expression persists through endometrial development.Lgr5 is broadly expressed in the uterine epithelium during embryogenesis, but becomes largely restricted to the tips of developing glands after birth. 31905853_PP2A Deficiency Enhances Carcinogenesis of Lgr5(+) Intestinal Stem Cells Both in Organoids and In Vivo. 32213541_Loss of Apc Rapidly Impairs DNA Methylation Programs and Cell Fate Decisions in Lgr5(+) Intestinal Stem Cells. 32321913_The small intestinal stroma and Lgr5+ villus tip telocytes are regulators of the epithelial spatial expression programs along the villus axis. 32327656_Mouse liver tumors contain the enrichment of LGR5 expressing cells. Isolated LGR5 expressing cells from mouse liver tumors are superior in initiating organoids and forming tumors upon engraftment, featuring candidate tumor-initiating cells. These cells are resistant to conventional treatment including sorafenib and 5-FU. LGR5 lineage ablation inhibits organoid initiation and tumor growth. 32464345_Lgr5-positive endothelial progenitor cells occupy a tumor and injury prone niche in the kidney vasa recta. 33066957_Interleukin-10 expands transit-amplifying cells while depleting Lgr5(+) stem cells via inhibition of Wnt and notch signaling. 33357435_Lgr5 Marks Adult Progenitor Cells Contributing to Skeletal Muscle Regeneration and Sarcoma Formation. 33767594_PTEN-mediated AKT/beta-catenin signaling enhances the proliferation and expansion of Lgr5+ hepatocytes. 34108617_alphaSMA(+) fibroblasts suppress Lgr5(+) cancer stem cells and restrain colorectal cancer progression. 34217895_ANKRD22 Drives Rapid Proliferation of Lgr5(+) Cells and Acts as a Promising Therapeutic Target in Gastric Mucosal Injury. 34260310_Persistence of Lgr5+ colonic epithelial stem cells in mouse models of inflammatory bowel disease. 35081371_IL-17RA-signaling in Lgr5(+) intestinal stem cells induces expression of transcription factor ATOH1 to promote secretory cell lineage commitment. 35729700_Study on Lgr5 expression in pancreas tissues and organoids by lineage tracing. 36410420_Loss of Hepatic Leucine-Rich Repeat-Containing G-Protein Coupled Receptors 4 and 5 Promotes Nonalcoholic Fatty Liver Disease. |
ENSG00000139292 |
LGR5 |
13.579447 |
0.1392455664 |
-2.844297 |
0.80153466 |
12.954081 |
0.00031922481048073060236336395689704659162089228630065917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016820093721157838677349172229469331796281039714813232421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.29925317329771 |
1.92876922637947 |
23.6937753748701 |
8.41512803761132 |
| ENSMUSG00000020151 |
19279 |
Ptprr |
protein_coding |
Q62132 |
FUNCTION: Sequesters mitogen-activated protein kinases (MAPKs) such as MAPK1, MAPK3 and MAPK14 in the cytoplasm in an inactive form. The MAPKs bind to a dephosphorylated kinase interacting motif, phosphorylation of which by the protein kinase A complex releases the MAPKs for activation and translocation into the nucleus. Isoform gamma may have a role in patterning and cellular proliferation of skeletal elements in the precartilaginous/cartilaginous skeleton. {ECO:0000269|PubMed:10601328, ECO:0000269|PubMed:10949045}. |
3D-structure;Alternative splicing;Cell membrane;Cytoplasm;Glycoprotein;Hydrolase;Membrane;Phosphoprotein;Protein phosphatase;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:19279; |
cell junction [GO:0030054]; cytosol [GO:0005829]; plasma membrane [GO:0005886]; protein kinase binding [GO:0019901]; protein tyrosine phosphatase activity [GO:0004725]; ERBB2 signaling pathway [GO:0038128]; in utero embryonic development [GO:0001701]; negative regulation of epithelial cell migration [GO:0010633]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; peptidyl-tyrosine dephosphorylation [GO:0035335]; protein dephosphorylation [GO:0006470]; signal transduction [GO:0007165] |
15461663_Multiple isoforms through the use of distinct promoters, alternative splicing and differential translation starts. 16148006_The ERK2 docking groove is more restrictive and selective for its tyrosine phosphatase inactivators than for MEK1/2 and indicate that distinct ERK2 residues modulate the docking interactions with activating and inactivating effectors. 17147696_PTPBR7, was subject to N-terminal proteolytic processing, in between amino acid position 136 and 137, resulting in an additional, 65-kDa transmembrane PTPRR isoform. 17266727_Ptprr-/- mice displayed ataxia characterized by defects in fine motor coordination and balance skills. 18068678_PTP-SL, like PTPBR7, is a single membrane-spanning receptor-type protein tyrosine phosphatases. 21850207_Results support the suggested link between PTPRR isoforms and cerebellar calcium ion homeostasis, and suggest an additional role in the process of cell-cell adhesion 24556203_The data of this study disclosed a distinct role for PTPRR in recognition memory and exploration of a new environment. 25571783_These findings indicate an important role for PTPRR in maintaining low basal mitogen-activated protein kinase activity in Purkinje cells. |
ENSG00000153233 |
PTPRR |
74.554321 |
0.2974370158 |
-1.749344 |
0.36815324 |
22.058572 |
0.00000264456105945754172247149325902793037812443799339234828948974609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000218093958081522897192710414016048048324591945856809616088867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.6753491319477 |
8.42903018748307 |
109.85636418059 |
20.0991062996177 |
| ENSMUSG00000020182 |
13195 |
Ddc |
protein_coding |
O88533 |
FUNCTION: Catalyzes the decarboxylation of L-3,4-dihydroxyphenylalanine (DOPA) to dopamine and L-5-hydroxytryptophan to serotonin. {ECO:0000250|UniProtKB:P80041}. |
Acetylation;Catecholamine biosynthesis;Decarboxylase;Lyase;Pyridoxal phosphate;Reference proteome;Repeat |
PATHWAY: Catecholamine biosynthesis; dopamine biosynthesis; dopamine from L-tyrosine: step 2/2. {ECO:0000250|UniProtKB:P80041}. |
|
mmu:13195; |
axon [GO:0030424]; cytoplasm [GO:0005737]; neuronal cell body [GO:0043025]; synaptic vesicle [GO:0008021]; 5-hydroxy-L-tryptophan decarboxylase activity [GO:0036467]; amino acid binding [GO:0016597]; aromatic-L-amino-acid decarboxylase activity [GO:0004058]; carboxy-lyase activity [GO:0016831]; enzyme binding [GO:0019899]; L-dopa decarboxylase activity [GO:0036468]; protein domain specific binding [GO:0019904]; pyridoxal phosphate binding [GO:0030170]; aminergic neurotransmitter loading into synaptic vesicle [GO:0015842]; amino acid metabolic process [GO:0006520]; catecholamine metabolic process [GO:0006584]; cellular response to alkaloid [GO:0071312]; cellular response to growth factor stimulus [GO:0071363]; cellular response to xenobiotic stimulus [GO:0071466]; circadian rhythm [GO:0007623]; dopamine biosynthetic process [GO:0042416]; isoquinoline alkaloid metabolic process [GO:0033076]; multicellular organism aging [GO:0010259]; phytoalexin metabolic process [GO:0052314]; response to pyrethroid [GO:0046684]; response to toxic substance [GO:0009636]; serotonin biosynthetic process [GO:0042427] |
15207355_Data suggest a possible participation of aromatic L-amino acid decarboxylase (AADC) in the regulation of catecholamine biosynthesis in stellate ganglia of repeatedly stressed mice. 17211625_Results suggest that AADC expression begins at early stages of taste bud cell differentiation, and biogenic amines may act on taste bud differentiation of tongue epithelial cells, and further may regulate innervation of taste bud progenitor cells. 17967881_analysis of Ddc redefines the imprinted Grb10 domain on mouse proximal chromosome 11 and identifies Ddc_exon1a as the first example of a heart-specific imprinted gene 18310457_the fact that renal DDC inversely correlated with renal sodium reabsorption suggest that renal and intestinal gender dimorphism in DDC could be related to sex-related differences in sodium balance observed between males and females 20456015_evidence for PKGIalpha-dependent phosphorylation and activation of neuronal AAAD in vitro, and introduce AAAD as a putative PKGIalpha substrate. 21322090_Mash1 plays an important role in regulating the expression of AADC in type III cells in taste buds. 22829864_The data show that endothelial AADC plays an important role in cardiac synthesis of 5-HT and possibly in 5-HT-dependent regulation of nitric oxide generation. 27658936_We found that only the modified U1 snRNA (IVS-AAA) that completely matched both the intronic and exonic U1 binding sequences of the mutated DDC gene could correct splicing errors of either the mutated human DDC minigene or the mouse artificial splicing construct in vitro 32522342_Metastasis-associated protein 1 (MTA1) regulates the catecholamine production homeostasis via transcriptional repression of aromatic l-amino acid decarboxylase (Aadc) in the interstitial cells of Cajal of mouse prostate. |
ENSG00000132437 |
DDC |
166.129619 |
3.5686828821 |
1.835392 |
0.23307064 |
60.145014 |
0.00000000000000881198227081037042883377291251324348092717215347180470530474849510937929153442382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000002207633453108282438609658291048911378062595545213042669274727813899517059326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
254.044735074494 |
41.2202580678653 |
71.1872540835163 |
9.12896966412185 |
| ENSMUSG00000020205 |
21664 |
Phlda1 |
protein_coding |
Q62392 |
FUNCTION: Seems to be involved in regulation of apoptosis. May be involved in detachment-mediated programmed cell death. May mediate apoptosis during neuronal development. May be involved in regulation of anti-apoptotic effects of IGF1. Required for TCR-induced apoptosis and expression of TNFRSF6/FAS in a T-cell hybridoma cell line. May be involved in translational regulation. {ECO:0000269|PubMed:15037619, ECO:0000269|PubMed:8673705}. |
Apoptosis;Cytoplasm;Cytoplasmic vesicle;Nucleus;Reference proteome;Repeat |
|
|
mmu:21664; |
cytoplasmic vesicle [GO:0031410]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; phosphatidylinositol phosphate binding [GO:1901981]; apoptotic process [GO:0006915]; FasL biosynthetic process [GO:0045210]; positive regulation of apoptotic process [GO:0043065] |
12738777_TDAG51 is induced by homocysteine, promotes detachment-mediated PCD, and contributes to the development of atherosclerosis observed in hyperhomocysteinemia 21558389_PHLDA1 expression marks the putative epithelial stem cells, downregulates ITGA2 and ITGA6, and contributes to intestinal tumorigenesis 22961087_TDAG51 is involved in energy homeostasis at least in part by regulating lipogenesis in liver and white adipose tissue. 23063459_Our results suggest that the balance between the cell survival and death signals mediated by HSP70 and TDAG51, respectively, may be disturbed by the altered expression of HSF1 during the progression of disease in this amyotropic lateral sclerosis model. 23928855_TDAG51 has a protective role in oxidative stress-induced cell death in mouse embryonic fibroblasts. 26239656_The JAK2-ERK1/2-STAT3 pathway is an important signaling pathway for regulation of PHLDA1 expression. 26873343_These findings suggested that TDAG51 up-regulation is a dependent event during LPS-mediated proliferation and cell cycle progression, and which increase our understanding of the interaction mechanism between LPS and macrophages. 27960185_PHLDA1 plays a critical role in the development of progressive lung contusion and subsequent inflammation. 29127005_The data presented herein demonstrate TDAG51 expression to be upregulated in damaged skeletal muscle and its absence attenuates the early phases of muscle regeneration. 31251738_These findings indicate that TDAG51 plays a protective role against maternal care defects and depressive-like behavior after parturition. Thus, TDAG51 is a maternal care-associated gene that functions as a crucial regulator of maternal and depressive-like behavior after parturition. 32434409_TDAG51 (T-Cell Death-Associated Gene 51) Is a Key Modulator of Vascular Calcification and Osteogenic Transdifferentiation of Arterial Smooth Muscle Cells. 33335079_T-Cell Death Associated Gene 51 Is a Novel Negative Regulator of PPARgamma That Inhibits PPARgamma-RXRalpha Heterodimer Formation in Adipogenesis 33839577_miR-194 ameliorates hepatic ischemia/reperfusion injury via targeting PHLDA1 in a TRAF6-dependent manner. 34625532_TDAG51 induces renal interstitial fibrosis through modulation of TGF-beta receptor 1 in chronic kidney disease. 35175494_TDAG51-Deficiency Podocytes are Protected from High-Glucose-Induced Damage Through Nrf2 Activation via the AKT-GSK-3beta Pathway. 35237256_PHLDA1 Suppresses TLR4-Triggered Proinflammatory Cytokine Production by Interaction With Tollip. 36681251_PHLDA1 knockdown alleviates mitochondrial dysfunction and endoplasmic reticulum stress-induced neuronal apoptosis via activating PPARgamma in cerebral ischemia-reperfusion injury. |
ENSG00000139289 |
PHLDA1 |
379.194468 |
0.4491716435 |
-1.154661 |
0.16137415 |
50.256663 |
0.00000000000134895933939826536533469535487902299310432330248232801750418730080127716064453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000264104738531033163485757835008444654491255310091446517617441713809967041015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
235.32874597348 |
26.3778656329211 |
523.917191525977 |
42.7522966829387 |
| ENSMUSG00000020218 |
24117 |
Wif1 |
protein_coding |
Q9WUA1 |
FUNCTION: Binds to WNT proteins and inhibits their activities. May be involved in mesoderm segmentation. |
Developmental protein;Disulfide bond;EGF-like domain;Glycoprotein;Reference proteome;Repeat;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:24117; |
cell surface [GO:0009986]; extracellular region [GO:0005576]; signaling receptor binding [GO:0005102]; Wnt-protein binding [GO:0017147]; anatomical structure development [GO:0048856]; negative regulation of Wnt signaling pathway [GO:0030178]; positive regulation of fat cell differentiation [GO:0045600]; Wnt signaling pathway [GO:0016055] |
15555925_WIF-1 protein inhibits rod production, and anti-WIF-1 antibodies increase rod production; in contrast, Wnt4 promotes rod production. 17964517_Dkk-1/Wnt/beta-catenin cascade may mediate the proliferation and migration of hepatocellular carcinoma cells during the metastasis process. 18586116_Wif1 mRNA can be detected as early as the developmental stage E11, and expression persists to adulthood. 19185060_Data provided a full expression pattern for Wif1 in limb development, for which no limb expression had been documented so far. 19254785_Results indicate that WIF-1 plays as a negative regulator of osteoblastic differentiation in mouse mesenchymal C3H10T1/2 cells in vitro. 19307728_targeted deletion of Wif1 accelerated development of radiation-induced osteosarcomas in vivo 19755491_These results identify Wif-1 as a novel extracellular Wnt modulator in cartilage biology. 21179454_wif1 is a modulator of cardiomyocyte differentiation 21270055_a novel regulatory loop of Bmp4-Smad1-Wif1-Wnt/beta-catenin in coordinating BMP and Wnt pathways to control fetal lung development 21652676_Osteoblastic Wif1 overexpression disrupts stem cell quiescence, leading to a loss of self-renewal potential. 23087175_Wnt inhibitory factor 1 (Wif1) is regulated by androgens and enhances androgen-dependent prostate development 23784913_These data suggest that WIF-1 may take part in the fine-tuning of cartilage and bone turnover, promoting the balance of cartilage versus bone anabolism. 23921644_It is anticipated that our findings will contribute to expansion of our understanding of WIF1 biological function on heart development and possible modes of treatment of heart diseases 24362065_WIF1 secretion by the Mullerian duct mesenchyme plays a role in Mullerian duct regression in fetal males 24632949_Dysregulation of this endodermal Shh-Wif1-b-catenin signaling axis contributes to ARM pathogenesis. 25486432_EZH2-induced downregulation of WIF1 expression may partially regulate Wnt/beta-catenin-dependent crypt hyperplasia in response to citrobacter rodentium infection 25571841_results demonstrate that Wif1 is not targeted for silencing by DNA methylation in OS. Instead, the reduced expression of Wif1 in OS cells is in context with their stage in differentiation 25893927_Wif1 localizes to the enamel knot in which Wif1 regulates apoptosis by mediating and regulating Wnt-beta-catenin signaling. Thus, Wif1 plays an essential role in tooth development. 25918249_WIF1 has a role in breast neoplasms: its inhibition significantly relieves the cancer stem cell-limiting effects of dietary compound isoliquiritigenin 31987884_WIF1 Suppresses the Generation of Suprabasal Cells in Acanthotic Skin and Growth of Basal Cell Carcinomas upon Forced Overexpression. 35080047_Wnt inhibitory 1 ameliorates neovascularization and attenuates photoreceptor injury in an oxygen-induced retinopathy mouse model. |
ENSG00000156076 |
WIF1 |
4281.154096 |
0.3140815718 |
-1.670789 |
0.07044179 |
542.364844 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000576723620417116732847805181840329114357547058822516848278752821998825185577191991524048031598443329379299814867745718320459198929687998956224464 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000043923270930967609449137112619374335630988070241294504321502717482774089513095284817093997572013439412019047050200359724253205661978455329722041 |
Yes |
No |
2040.74700784901 |
99.6821907139854 |
6497.506351814 |
232.292037017738 |
| ENSMUSG00000020227 |
73914 |
Irak3 |
protein_coding |
Q8K4B2 |
FUNCTION: Putative inactive protein kinase which regulates signaling downstream of immune receptors including IL1R and Toll-like receptors (PubMed:12150927, PubMed:12054681, PubMed:29686383). Inhibits dissociation of IRAK1 and IRAK4 from the Toll-like receptor signaling complex by either inhibiting the phosphorylation of IRAK1 and IRAK4 or stabilizing the receptor complex (PubMed:12150927, PubMed:12054681). Upon IL33-induced lung inflammation, positively regulates expression of IL6, CSF3, CXCL2 and CCL5 mRNAs in dendritic cells (PubMed:29686383). {ECO:0000269|PubMed:12054681, ECO:0000269|PubMed:12150927, ECO:0000269|PubMed:29686383}. |
ATP-binding;Cytoplasm;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:73914; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; ATP binding [GO:0005524]; magnesium ion binding [GO:0000287]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; protein kinase activity [GO:0004672]; protein kinase binding [GO:0019901]; protein serine/threonine kinase activity [GO:0004674]; cytokine-mediated signaling pathway [GO:0019221]; interleukin-1-mediated signaling pathway [GO:0070498]; intracellular signal transduction [GO:0035556]; negative regulation of cytokine-mediated signaling pathway [GO:0001960]; negative regulation of innate immune response [GO:0045824]; negative regulation of interleukin-12 production [GO:0032695]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of macrophage cytokine production [GO:0010936]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of NF-kappaB transcription factor activity [GO:0032088]; negative regulation of protein catabolic process [GO:0042177]; negative regulation of protein-containing complex disassembly [GO:0043242]; negative regulation of toll-like receptor signaling pathway [GO:0034122]; negative regulation of tumor necrosis factor production [GO:0032720]; positive regulation of cytokine production [GO:0001819]; positive regulation of macrophage tolerance induction [GO:0010933]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; protein autophosphorylation [GO:0046777]; protein phosphorylation [GO:0006468]; regulation of protein-containing complex disassembly [GO:0043244]; response to exogenous dsRNA [GO:0043330]; response to interleukin-1 [GO:0070555]; response to lipopolysaccharide [GO:0032496]; response to peptidoglycan [GO:0032494]; response to virus [GO:0009615] |
12150927_Endotoxin tolerance was significantly reduced in macrophages from IRAK-M knockout mice. IRAK-M regulates TLR signaling and innate immune homeostasis. 14660668_induction of IRAK-M and inhibition of kinase activity of IRAK-1 are crucial to PGN-induced tolerance in macrophages 15809356_IRAK-M is a key regulator of the bone loss that is due to osteoclastic resorption of bone. 16263713_IRAK-M induction negatively regulates Toll-like receptor-dependent interleukin-12 p40 production in macrophages in the presence of Mycobacterium tuberculosis lipoarabinomannan 17379480_These findings indicate that IRAK-M selectively attenuates p38 activation and inhibits innate immunity through stabilizing MKP-1. 17477969_IRAK-M is directly involved in the regulation of both innate and adaptive immune signaling processes, and deletion of IRAK-M enhances host anti-tumor immune response. 18378686_reduced expression of IRAK-1 and increased expression of IRAK-M after CpG DNA pretreatment resulted in the hyporesponsiveness of macrophages that leads to the protection of mice from hepatic injury and death caused by CpG DNA/D-GalN. 19225984_Tolerance to intraluminally administered LPS in the lymphocyte recruitment process was induced by enterobacteria, possibly via the induction of IRAK-M and TGF-beta. 19251253_The expression of IRAK-M as a negative regulator of TLR7 signaling was markedly augmented in immune tolerant macrophage-like cells while the interleukin-1 receptor-associated kinase (IRAK)-1 functioned normally. 19414798_IRAK-M is a major mediator of globular adiponectin-induced endotoxin tolerance in primary macrophages 19596984_TREM-1 activation beneficially impacts pulmonary IRAK-M expression 19809574_these findings suggest that IRAK-M negatively regulates the alternative NFkappaB pathway in a ligand-specific manner. 20042589_IRAK-M is critical to preventing deleterious neutrophil-dependent lung injury during influenza infection of the respiratory tract. 20439918_These data suggest that the absence of IRAK-M in the hematopoietic compartment post-bone marrow transplantation enhances pulmonary host defense and mitigates alveolar macrophage sensitivity to the inhibitory effects of PGE(2 20585389_findings indicate that systemic sepsis induces epigenetic silencing of cytokine gene expression in lung macrophages, and IRAK-M appears to be a critical mediator of this response 20817880_manipulation of IRAK-M levels can increase the potency of DC vaccines by enhancing their Ag-presenting function, migration, and longevity 20848470_Loss of IRAK-M is associated with colitis. 21098228_IRAK-M is bound to caspase (CASP)-6 in a complex located near the plasma membrane in resting cells, and the components of this complex redistribute to nucleus and cytoplasm after neutrophil stimulation. 21257958_By facilitating IRAK-M expression, DNAX-activation protein (DAP)12 functions to negatively regulate lipopolysaccharide-induced liver dendritic cell maturation. 21690387_Role of IL-1 receptor-associated kinase-M (IRAK-M) in priming of immune and inflammatory responses by nitrogen bisphosphonates. 21875872_These data identify a previously unknown function of IRAK-M :suppression of TLR7-mediated autoimmunity-and mutant IRAK-M as a previously unknown genetic risk for murine SLE. 21934070_Endotoxin tolerization in vivo blocked TLR4-driven IRAK4 phosphorylation and activation in macrophages, while increasing expression of IRAK-M, SHIP-1, A20 mRNA, and A20 protein. 21987497_The upregulation of IRAK-M in macrophages is involved in the local immunosuppression around implants, and may contribute to septic and aseptic implant loosening. 22472665_Along with endotoxin, bacterial sonicate is able to induce refractory tolerance in BM-DCs, and IRAK-M plays a role in modulating cell surface expression of MHC class II and CD80 and release of IL-10 during this tolerance. 22492852_These data suggest that IRAK-M impairs host defense during pneumococcal pneumonia at the primary site of infection at least in part by inhibiting the early immune response. 22729155_IRAK-M impairs host defense during pneumonia caused by a common gram-negative respiratory pathogen. 22941946_debris-induced IRAK-M decreases foreign body reactions, but at the same time, the over-expression of IRAK-M may also be detrimental on local intrusion of PAMPs or bacteria 23376919_IRAK-M mediates TLR7-induced MEKK3-dependent second wave NFjB activation to produce inhibitory molecules 23437317_IRAK-M plays an important role in alcohol-induced liver injury and IRAK-M negatively regulates the innate and possibly the adaptive immune response in the liver reacting to acute insult by alcohol. 23567778_Altered gut microbiota promotes colitis-associated cancer in IL-1 receptor-associated kinase M-deficient mice. 23872113_novel findings provide new insights into the understanding of negative regulatory mechanisms of the TLR4 signaling pathway. 24172845_Our study identifies the DAP12/IRAK-M/IL-10 to be a novel molecular pathway in APCs exploited by mycobacterial pathogens, allowing infection a foothold in the lung. 24696448_This study illustrates how the modulation of innate immune pathways through IRAK-M influences the development of autoimmune diabetes. 25595781_These data indicate expression of IRAK-M skews lung macrophages toward an alternatively activated profibrotic phenotype, which promotes collagen production, leading to the progression of experimental pulmonary fibrosis. 25603827_These data demonstrate LTi cells are present in the stomach and promote lymphoid follicle formation in response to infection, but are limited by IRAK-M expression. 26140693_the results suggest that IRAK-M may be targeted by L. donovani to inhibit TLR-mediated proinflammatory response late during in vitro infection. 26218271_IL-7 reduced IRAK-M expression and attenuated immune tolerance induced by either LPS or CpGA 26542797_IRAK-M may also contribute to myofibroblast conversion. 27150039_Data show that interleukin-1 receptor-associated kinase 3 (IRAK-M) is responsible for regulation of microbial colonization of tumors and STAT3 protein stability in tumor cells, leading to tumor cell proliferation. 27939424_IRAK-M functions to modulate inflammatory signaling pathways and is critical in maintaining immune system homeostasis in the gut. However, increased IRAK-M is associated with increased disease pathogenesis and increased cancer severity in human patients. 28011933_polycomb recessive complex 2 repressed the IRAK-M promoter, allowing low levels of expression; following LPS stimulation, the IRAK-M promoter is derepressed, and transcription is induced to allow its expression. 28120642_this study shows that following Pseudomonas aeruginosa infection, IRAK-M knock-out mice have enhanced lung neutrophilic inflammation and reduced bactertial load 28665693_IRAK-M plays a crucial role in the regulation of allergic airway inflammation by modifying the function of airway epithelia 28701510_Taken together, these results strongly support a role for IRAK-M in renal injury and identify IRAK-M as a possible modulator in driving an alternatively activated profibrotic macrophage phenotype in unilateral ureteral obstruction-induced chronic kidney disease. 28713897_IRAK3 methylation may be a predictive factor in the transition from colitis to cancer. 29686383_Study using IL-33-induced type 2 immunity signaling in a combination of cell lines, mouse models, and primary cells from mouse models reveal critical roles of IRAK-M and PIN1 in IL-33-induced type 2 immunity. Upon IL-33-induced inflammation, activated PIN1 binds to and catalyzes cis-trans isomerization of phosphorylated IRAK-M, inducing IRAK-M stabilization and nuclear translocation. 30131810_we identify a Blimp-1-dependent pathway that rapidly facilitates IFN-I production by relieving interleukin-1 receptor-associated kinase M, encoded by Irak3, in pDCs. 30372424_TLR stimulation led to IRAKM-caspase-8-ASC complex formation, resulting in the activation of caspase-8 and IL-1beta release in microglia. 30617222_Findings demonstrated a distinctive role of IRAK-M in maintaining chronic Th2 airway inflammation and indicated a complex role for IRAK-M in the initiation and progression of experimental allergic asthma. 31036429_the data indicates that IRAK-M significantly improves experimental autoimmune encephalomyelitis onset through down-regulation of the TLR4-MyD88 signaling pathway, which finally leads to differentiation of M2 phenotype in the microglia. 31607540_Neutrophils Deficient in Innate Suppressor IRAK-M Enhances Anti-tumor Immune Responses. 32253243_IRAK-M Regulates Monocyte Trafficking to the Lungs in Response to Bleomycin Challenge. 34031144_TRAM-Related TLR4 Pathway Antagonized by IRAK-M Mediates the Expression of Adhesion/Coactivating Molecules on Low-Grade Inflammatory Monocytes. 35139396_IRAK-M deletion aggravates acute inflammatory response and mitochondrial respiratory dysfunction following myocardial infarction: A bioinformatics analysis. 36765034_IRAK-M suppresses the activation of microglial NLRP3 inflammasome and GSDMD-mediated pyroptosis through inhibiting IRAK1 phosphorylation during experimental autoimmune encephalomyelitis. |
ENSG00000090376 |
IRAK3 |
69.091303 |
0.4330878949 |
-1.207268 |
0.36803057 |
10.671757 |
0.00108783728878722495483910570612806623103097081184387207031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0049812044392309020104936401196482620434835553169250488281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.8981882954526 |
10.2887517278735 |
92.1249214382801 |
17.164665640769 |
| ENSMUSG00000020262 |
110532 |
Adarb1 |
protein_coding |
Q91ZS8 |
FUNCTION: Catalyzes the hydrolytic deamination of adenosine to inosine in double-stranded RNA (dsRNA) referred to as A-to-I RNA editing. This may affect gene expression and function in a number of ways that include mRNA translation by changing codons and hence the amino acid sequence of proteins; pre-mRNA splicing by altering splice site recognition sequences; RNA stability by changing sequences involved in nuclease recognition; genetic stability in the case of RNA virus genomes by changing sequences during viral RNA replication; and RNA structure-dependent activities such as microRNA production or targeting or protein-RNA interactions. Can edit both viral and cellular RNAs and can edit RNAs at multiple sites (hyper-editing) or at specific sites (site-specific editing). Its cellular RNA substrates include: bladder cancer-associated protein (BLCAP), neurotransmitter receptors for glutamate (GRIA2 and GRIK2) and serotonin (HTR2C), GABA receptor (GABRA3) and potassium voltage-gated channel (KCNA1). Site-specific RNA editing of transcripts encoding these proteins results in amino acid substitutions which consequently alter their functional activities. Edits GRIA2 at both the Q/R and R/G sites efficiently but converts the adenosine in hotspot1 much less efficiently. Can inhibit cell proliferation and migration and can stimulate exocytosis. {ECO:0000269|PubMed:17369310}. |
3D-structure;Alternative splicing;Hydrolase;Metal-binding;mRNA processing;Nucleus;Phosphoprotein;Reference proteome;Repeat;RNA-binding;Zinc |
|
This gene encodes a double-stranded-RNA-specific adenosine deaminase that is involved in editing pre-mRNAs by site-specific conversion of adenosine (A) to inosine (I). Substrates for this enzyme include ionotropic glutamate receptors (GluR2-6) and serotonin receptor (5HT2C). Studies in rodents have shown that this protein can modify its own pre-mRNA by A->I editing to create a novel acceptor splice site, alternative splicing to which results in down regulation of its protein expression. Additional splicing events result in transcript variants encoding different isoforms. [provided by RefSeq, Jul 2008]. |
mmu:110532; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; synapse [GO:0045202]; double-stranded RNA adenosine deaminase activity [GO:0003726]; double-stranded RNA binding [GO:0003725]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; RNA binding [GO:0003723]; tRNA-specific adenosine deaminase activity [GO:0008251]; adenosine to inosine editing [GO:0006382]; base conversion or substitution editing [GO:0016553]; facial nerve morphogenesis [GO:0021610]; hypoglossal nerve morphogenesis [GO:0021618]; innervation [GO:0060384]; motor behavior [GO:0061744]; motor neuron apoptotic process [GO:0097049]; mRNA modification [GO:0016556]; mRNA processing [GO:0006397]; multicellular organism growth [GO:0035264]; muscle tissue morphogenesis [GO:0060415]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of protein kinase activity by regulation of protein phosphorylation [GO:0044387]; neuromuscular process controlling posture [GO:0050884]; neuromuscular synaptic transmission [GO:0007274]; positive regulation of mRNA processing [GO:0050685]; positive regulation of viral genome replication [GO:0045070]; regulation of cell cycle [GO:0051726]; RNA processing [GO:0006396]; spinal cord ventral commissure morphogenesis [GO:0021965] |
14612560_the nucleolar localization of ADAR2 represents an important mechanism by which RNA editing can be modulated by the sequestration of enzymatic activity from potential RNA substrates in the nucleoplasm 15939761_Through the subcellular targeting of minigenes that contain natural editing sites, we show that ADAR2- but not ADAR1-mediated RNA editing occurs in the nucleolus. 16369484_mature miRNA-142 expression levels increased substantially in ADAR1 null or ADAR2 null mice 16382140_ADAR2 autoediting is a key regulator of ADAR2 protein expression and activity in vivo. 16956888_RNA editing by ADAR2 in pancreatic islets and beta-cells is metabolically regulated by nutritional and energy status 17567573_ADAR2 may possess additional biological activities beyond those required for the site-selective deamination of adenosine or may interfere with the actions of other double-stranded RNA-specific binding proteins in the cell 18430892_Possible target preferences of ADAR1 and ADAR2 were examined by determining the editing status of transcripts derived from mice deficient in either enzyme. 19156214_analysis of a splice variant that extends the open reading frame of ADAR2 by 49 amino acids through the utilization of an exon located 18 kilobases upstream of the previously annotated first coding exon and driven by a candidate alternative promoter 19325227_ADAR1 and ADAR2 are broadly distributed in most regions of the mouse forebrain by P0. 19361536_ADAR2 transgenic mice represent a novel rodent model of endogenous behavioral despair and anxiety accompanied by elevated hypothalamo-pituitary adrenal axis activity. 20651031_Activation of phospholipase C and alternative splicing of pre-mRNA encoding the editing enzymes ADAR1 and ADAR2 have both overlapping and specific roles in modulating 5-HT2CR editing phenotypes. 20826656_The loss of ADAR2 activity causes AMPA receptor-mediated death of motor neurons in mice disease model(ALS). 21467037_Requirement of the RNA-editing enzyme ADAR2 for normal physiology in mice. 21615684_These results altogether suggest that altered 5HT(2C) R function could be contributing to enhanced depression-like behavior of ADAR2 transgenic mice and further implicate ADAR2 as a contributing factor in cases of affective disorder. 21632755_These results provide evidence that ADAR1, and not ADAR2, is the deaminase responsible for protection against virus-induced cytopathic effects, and that ADAR de fi ciency does not adversely affect PyV growth. 21788117_Through these gene knockout animal models we demonstrated for the first time that ADAR1 is an essential molecule for skin integrity. 21847096_ADAR2 protein levels and catalytic activity are coordinately regulated in a positive manner by Pin1 and negatively by WWP2 and this may have downstream effects on the function of glutamate receptor 2. 22310477_miRNA abundance and miRNA processing are mainly influenced by ADARB1; an additional loss of ADAR has only a minor effect on expression of mature miRNAs. 22916266_Study show co-occurrence of TDP-43 mislocalization with reduced activity of an RNA editing enzyme, ADAR2, in aged mouse motor neurons. 23139803_The JNK1 pathway may be functionally linked to the nutrient-sensing actions of ADAR2-mediated RNA editing in professional secretory cells. 23323881_Goal-oriented overeating behavior of ADAR2 transgenic mice is associated with altered feeding, reward-related mRNAs and hyperactive brain mesolimbic region. 24025532_Analysis of editing in the filamin A encoding mRNA shows very high editing levels outside the nervous system; further shows FLNA editing is mainly achieved by ADAR2 but that in some cases ADAR1 can efficiently compensate for ADAR2. 24440630_Results suggest that ADAR2-reduction is associated with progressive deterioration of nuclear architecture, resulting in vacuolated nuclei due to a Ca2+-permeable AMPA receptor-mediated mechanism 24443933_altered RNA editing efficiency of AMPA receptors due to down-regulation of ADAR2 has a possible role in the pathophysiology of mental disorders. 24608178_chronically elevated intraocular pressure in adult mice reduces expression of the ADAR2 enzyme. 25260591_This study clearly demonstrates that ADAR2 influences miRNA abundance and targeting at several levels in the mouse brain. 25582055_ADAR2 is a key factor for maintaining edited-miRNA population and balancing the expression of several essential miRNAs involved in cancer. 26383032_After fear conditioning protocol, mRNA expression of ADAR2 increased in amygdala and hippocampus, and those mice had a learning-dependent increase in the alternatively spliced inactive form of ADAR2 in the amygdala. 26669816_Results show that ADAR2 is important in site-specific changes of protein coding sequences but has relatively modest effects on gene expression and splicing in the adult mouse frontal cortex. 26768488_Thus, Adarb1 and Adarb2 have nearly exclusive expression within brain tissue, while Adar has more appreciable expression across multiple tissues. 28126736_that ADAR2 is a mediator of injury-induced tactile allodynia and thus a potential therapeutic target for the treatment of neuropathic pain 28395340_ADARB1 mediates adenosine to inosine RNA editing in the testis, and these editing events are dispensable for male fertility in an inbred mouse strain in the lab. 28640668_FMRP inhibits ADAR2 activity, absence of FMRP results in defects of RNA editing of neuronal mRNAs in the mouse model of Fragile X Syndrome. 29694894_PKCzeta/ADAR2 axis is a critical regulator of colorectal cancer metastases through modulation of miR-200 levels. 30287844_ADAR2 plays a crucial role for light-induced phase-shift of the circadian clock. 30945056_that the mislocalization of ADAR2 in C9orf72 mediated amyotrophic lateral sclerosis/frontotemporal dementia is responsible for the alteration of RNA processing events that may impact vast cellular functions 31815657_ADAR1 and ADAR2 are non-redundant and do not compensate for each other's essential functions in vivo. 36221913_Heterogeneous RNA editing and influence of ADAR2 on mesothelioma chemoresistance and the tumor microenvironment. 36527980_Pathological features of glial cells and motor neurons in the anterior horn of the spinal cord in sporadic ALS using ADAR2 conditional knockout mice. 36615779_Exercise-Induced ADAR2 Protects against Nonalcoholic Fatty Liver Disease through miR-34a. |
ENSG00000197381 |
ADARB1 |
599.217641 |
0.2944781755 |
-1.763767 |
0.16045437 |
115.977477 |
0.00000000000000000000000000480725168272225164813799828216030607196309196752306583716916611348800086531141140255840582540258765220642089843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000002988737046172462502808233954302834849163945824839628298694538816450243234879025067129987291991710662841796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
273.319377856313 |
30.4608763208421 |
928.148163845523 |
74.8794294136306 |
| ENSMUSG00000020286 |
67358 |
1700093K21Rik |
protein_coding |
Q810S2 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:67358; |
membrane [GO:0016020] |
24549580_The 1700093K21Rik gene and flanking regions are correlated with reproductive isolation in the house mouse. |
ENSG00000237651 |
C2orf74 |
17.096784 |
0.1319683812 |
-2.921736 |
0.76874781 |
13.939585 |
0.00018878065231027742697754445710955906179151497781276702880859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010567831297280947441125098507086477184202522039413452148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.88562347744169 |
2.16985581714322 |
29.44359430475 |
10.7797822239607 |
| ENSMUSG00000020300 |
67579 |
Cpeb4 |
protein_coding |
Q7TN98 |
FUNCTION: Sequence-specific RNA-binding protein that binds to the cytoplasmic polyadenylation element (CPE), an uridine-rich sequence element (consensus sequence 5'-UUUUUAU-3') within the mRNA 3'-UTR (PubMed:17024188). RNA binding results in a clear conformational change analogous to the Venus fly trap mechanism (By similarity). Regulates activation of unfolded protein response (UPR) in the process of adaptation to ER stress in liver, by maintaining translation of CPE-regulated mRNAs in conditions in which global protein synthesis is inhibited (PubMed:28092655). Required for cell cycle progression, specifically for cytokinesis and chromosomal segregation (By similarity). Plays a role as an oncogene promoting tumor growth and progression by positively regulating translation of t-plasminogen activator/PLAT (PubMed:22138752). Stimulates proliferation of melanocytes (By similarity). In contrast to CPEB1 and CPEB3, does not play role in synaptic plasticity, learning and memory (PubMed:24386439). {ECO:0000250|UniProtKB:Q17RY0, ECO:0000269|PubMed:17024188, ECO:0000269|PubMed:22138752, ECO:0000269|PubMed:24386439, ECO:0000269|PubMed:28092655}. |
Alternative splicing;Cell projection;Cytoplasm;Endoplasmic reticulum;Metal-binding;Phosphoprotein;Reference proteome;Repeat;RNA-binding;Synapse;Zinc |
|
|
mmu:67579; |
cytoplasm [GO:0005737]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; endoplasmic reticulum [GO:0005783]; growth cone [GO:0030426]; messenger ribonucleoprotein complex [GO:1990124]; neuron projection [GO:0043005]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; postsynapse [GO:0098794]; postsynaptic density [GO:0014069]; synapse [GO:0045202]; metal ion binding [GO:0046872]; mRNA 3'-UTR binding [GO:0003730]; mRNA regulatory element binding translation repressor activity [GO:0000900]; ribosome binding [GO:0043022]; RNA binding [GO:0003723]; translation factor activity, RNA binding [GO:0008135]; cellular response to amino acid stimulus [GO:0071230]; cellular response to decreased oxygen levels [GO:0036294]; cellular response to glucose starvation [GO:0042149]; ionotropic glutamate receptor signaling pathway [GO:0035235]; negative regulation of cytoplasmic translation [GO:2000766]; negative regulation of neuron apoptotic process [GO:0043524]; response to ischemia [GO:0002931] |
12871996_CPEB4 is expressed in the principal cell layers of the hippocampus 24386439_Unlike its related family members, CPEB1 and CPEB3, CPEB4 seems to be dispensable for hippocampus-dependent plasticity, learning and memory. 25220394_Cpeb4 is induced by Gata1 and Tal1, and is essential for terminal erythropoiesis. 26627607_Identify a mechanism of VEGF overexpression in liver and mesentery that promotes pathologic, but not physiologic, angiogenesis, via sequential and nonredundant functions of CPEB1 and CPEB4. 27381259_Results show that Cpeb4 is highly expressed in the nervous system including spinal motor neurons. the CPEB4-unstructured low complexity domains (LCD) impairs neuron development that results in reduced motor axon branching and diminished NMJ formation in embryos and uncoordinated movement in neonatal mice. 28092655_combined transcriptional and translational Cpeb4 regulation generates a 'circadian mediator', which coordinates hepatic UPR activity with periods of high ER-protein-folding demand 29166615_that cytoplasmic polyadenylation element-binding protein 4 (CPEB4) acts as a survival factor exclusively for early postnatal granule cells 32169429_CPEB4 Increases Expression of PFKFB3 to Induce Glycolysis and Activate Mouse and Human Hepatic Stellate Cells, Promoting Liver Fibrosis. 32517870_The RNA-binding protein Cpeb4 is a novel positive regulator of osteoclast differentiation. 32594159_Polyadenylation of mRNA as a novel regulatory mechanism of gene expression in temporal lobe epilepsy. 35909199_mmu-microRNA-92a-3p attenuates pulmonary fibrosis by modulating Cpeb4-mediated Smad2/3 signaling pathway. |
ENSG00000113742 |
CPEB4 |
135.546268 |
0.2723169338 |
-1.876641 |
0.29349488 |
40.059127 |
0.00000000024639080275327864161412219055913037230043372005638957489281892776489257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000037455336402574257121183705344057146557901205596863292157649993896484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.0552094785076 |
13.1211509188746 |
235.223012330259 |
33.7315227396132 |
| ENSMUSG00000020303 |
20856 |
Stc2 |
protein_coding |
O88452 |
FUNCTION: Has an anti-hypocalcemic action on calcium and phosphate homeostasis. |
Disulfide bond;Glycoprotein;Hormone;Reference proteome;Secreted;Signal |
|
|
mmu:20856; |
endoplasmic reticulum [GO:0005783]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; perinuclear region of cytoplasm [GO:0048471]; enzyme binding [GO:0019899]; heme binding [GO:0020037]; hormone activity [GO:0005179]; protein homodimerization activity [GO:0042803]; calcium ion homeostasis [GO:0055074]; cellular calcium ion homeostasis [GO:0006874]; cellular response to hypoxia [GO:0071456]; decidualization [GO:0046697]; embryo implantation [GO:0007566]; endoplasmic reticulum unfolded protein response [GO:0030968]; negative regulation of gene expression [GO:0010629]; negative regulation of multicellular organism growth [GO:0040015]; regulation of hormone biosynthetic process [GO:0046885]; regulation of sodium-dependent phosphate transport [GO:2000118]; regulation of store-operated calcium entry [GO:2001256]; response to endoplasmic reticulum stress [GO:0034976]; response to oxidative stress [GO:0006979]; response to peptide hormone [GO:0043434]; response to vitamin D [GO:0033280] |
15485913_induced STC2 expression is an essential feature of survival component of the unfolded-protein response 18258678_The murine stanniocalcin 2 gene is a negative regulator of postnatal growth. 21545732_STC2 is linked to PERK signalling in acinar cells and has a role in limiting damage during pancreatic injury. 21746875_These results define a novel molecular function for STC2 as a negative modulator of Store-Operated Calcium Entry and provide the first direct evidence for the regulation of Ca2+ homeostasis by mammalian STC2. 22285620_These results suggest that the up-regulation of STC2 gene expression resulting from abnormal alpha-klotho-Fgf23 signaling may contribute to limitation of ectopic calcification. 23269473_The Stc2 promoter contains multiple putative xenobiotic response elements. 25533459_Stanniocalcin-2 inhibits proteolytic release of IGF and its ability to cause growth retardation upon transgenic overexpression in mice depends on its proteinase inhibitory function. 26983002_STC2 is involved in regulating PAPP-A activity during the development of atherosclerosis 28696214_results in MTA2 recruitment to the Stc2 promoter, concomitant with agonist-specific epigenetic modifications targeting histone H4 lysine acetylation 28990324_STC2, is a possible early marker during development of diabetes mellitus. 29748538_these findings newly identify STC2 as the first stanniocalcin responsible for mediating the immunomodulatory effects of MSCs on allogeneic T cells and STC2 contribute to MSC-based treatment for ACD mainly via reducing the CD8(+) Tc1 cells. 33011858_In Vivo Gene Delivery of STC2 Promotes Axon Regeneration in Sciatic Nerves. 34111662_Transcriptome-wide profiling discover: PM2.5 aggravates airway dysfunction through epithelial barrier damage regulated by Stanniocalcin 2 in an OVA-induced model. |
ENSG00000113739 |
STC2 |
16.683015 |
0.0550267679 |
-4.183723 |
0.82163486 |
24.499360 |
0.00000074334515640770570389753461523763000684539292706176638603210449218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000067962985728704516659014894797152805949735920876264572143554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.03378534873914 |
1.22190562679169 |
36.9599274066489 |
13.8219613577894 |
| ENSMUSG00000020330 |
15366 |
Hmmr |
protein_coding |
Q00547 |
FUNCTION: Receptor for hyaluronic acid (HA) (PubMed:1376732). Involved in cell motility (PubMed:1376732). When hyaluronan binds to HMMR, the phosphorylation of a number of proteins, including the PTK2/FAK1 occurs. May also be involved in cellular transformation and metastasis formation, and in regulating extracellular-regulated kinase (ERK) activity. May act as a regulator of adipogenesis (PubMed:22666460). {ECO:0000269|PubMed:1376732, ECO:0000269|PubMed:22666460}. |
Alternative splicing;Cytoplasm;Cytoskeleton;Glycoprotein;Hyaluronic acid;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:15366; |
cell surface [GO:0009986]; centrosome [GO:0005813]; cytosol [GO:0005829]; microtubule cytoskeleton [GO:0015630]; spindle [GO:0005819]; cargo receptor activity [GO:0038024]; hyaluronic acid binding [GO:0005540]; hyaluronan catabolic process [GO:0030214]; receptor-mediated endocytosis [GO:0006898] |
14534534_Gene deletion of Rhamm attenuates the formation of aggressive fibromatosis. 15596723_CD44 and RHAMM are molecularly redundant and have roles in a model of arthritis and inflammation 15809329_IHABP/RHAMM mediates cytoplasmic anchoring of the transcription factor Bach1 18174258_Androgen receptor regulates CD168 expression and signaling in prostate cancer 20558733_intracellular RHAMM(Delta163) functions as an adaptor protein to control microtubule polymerization during interphase and mitosis as a result of localizing ERK1/2-MEK1 complexes to their tubulin-associated substrates 21940500_RHAMM isoform B promotes liver metastasis in a mouse model of multistep tumorigenesis. 21947782_Overexpressing RHAMM was located intracellular and activated ERK1/2. 22418440_Results suggest that the CD44 and RHAMM receptors function on membrane lipid rafts during Cryptococcus neoformans invasion. 23391595_This study demonistrated that Rhamm expression in adult mouse subventricular zone and rostral migratory stream and in ischemic cortex. 23768790_Hyaluronon acid activation of RHAMM significantly impacts smooth muscle cell-ECM adhesive interactions and contributes to constrictive artery wall remodeling in mice. 24019927_RHAMM is not found on the cell-surface of embryonic stem cells, but it is required to maintain pluripotency and its dominant mechanism of action is through the modulation of signal transduction pathways at microtubules 25750434_RHAMM localises at the mitotic spindle of ovarian granulosa cells via its C-terminus, deletion of which impairs proper spindle orientation and folliculogenesis. 28994651_These data identify an essential role for HMMR in the PLK1-dependent regulatory pathway that orients progenitor cell division and supports neural development. 30098420_Study provides evidence that RHAMM is a critical component of the inflammatory response, respiratory distress and fibrosis after acute lung injury. 30130585_Results showed, for the first time, the distribution and activity of RHAMM in the context of skeletal muscle, and established that CD44 and RHAMM activation promote myogenic and connective tissue migration and proliferation. 32165498_Cell-specific expression of the transcriptional regulator RHAMM provides a timing mechanism that controls appropriate wound re-epithelialization. 33061798_Oleate acid-stimulated HMMR expression by CEBPalpha is associated with nonalcoholic steatohepatitis and hepatocellular carcinoma. 34044069_High levels of truncated RHAMM cooperate with dysfunctional p53 to accelerate the progression of pancreatic cancer. |
ENSG00000072571 |
HMMR |
1368.842385 |
2.3648124968 |
1.241726 |
0.07951724 |
240.516012 |
0.00000000000000000000000000000000000000000000000000000303523580511988530361252469676048572277907743347543516718627755261111924208393840674281442253612675105756350797044513341951953416713639623270015732714455225504934787750244140625000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000006004248283582609405887937024002172016484152058445233324041858526659977656103517086109761255932749663959105944435927145085541930570605106964876540587283670902252197265625000000000000000000000000000000000000000 |
Yes |
No |
1912.67384564897 |
105.474512761338 |
808.805707940916 |
34.5316047722424 |
| ENSMUSG00000020331 |
15166 |
Hcn2 |
protein_coding |
O88703 |
FUNCTION: Hyperpolarization-activated ion channel exhibiting weak selectivity for potassium over sodium ions. Contributes to the native pacemaker currents in heart (If) and in neurons (Ih). Can also transport ammonium in the distal nephron. Produces a large instantaneous current. {ECO:0000269|PubMed:10962006, ECO:0000269|PubMed:11096117, ECO:0000269|PubMed:11741901, ECO:0000269|PubMed:12034718, ECO:0000269|PubMed:12193608, ECO:0000269|PubMed:12968185, ECO:0000269|PubMed:17562314, ECO:0000269|PubMed:23103389}. |
3D-structure;cAMP;cAMP-binding;Cell membrane;Glycoprotein;Ion channel;Ion transport;Ligand-gated ion channel;Membrane;Methylation;Nucleotide-binding;Phosphoprotein;Potassium;Potassium channel;Potassium transport;Reference proteome;Sodium;Sodium channel;Sodium transport;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
|
mmu:15166; |
axon [GO:0030424]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; dendrite membrane [GO:0032590]; dendritic shaft [GO:0043198]; HCN channel complex [GO:0098855]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; somatodendritic compartment [GO:0036477]; synapse [GO:0045202]; cAMP binding [GO:0030552]; identical protein binding [GO:0042802]; intracellular cAMP-activated cation channel activity [GO:0005222]; molecular adaptor activity [GO:0060090]; PDZ domain binding [GO:0030165]; protein-containing complex binding [GO:0044877]; voltage-gated potassium channel activity [GO:0005249]; voltage-gated sodium channel activity [GO:0005248]; cellular response to cAMP [GO:0071320]; cellular response to cGMP [GO:0071321]; potassium ion import across plasma membrane [GO:1990573]; potassium ion transmembrane transport [GO:0071805]; regulation of cation channel activity [GO:2001257]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of membrane depolarization [GO:0003254]; regulation of membrane potential [GO:0042391]; regulation of SA node cell action potential [GO:0098907]; sodium ion import across plasma membrane [GO:0098719]; sodium ion transmembrane transport [GO:0035725] |
11741901_Here, we report that the mouse HCN2 pacemaker channel isoform also produced a large instantaneous current (I(inst(HCN2))) in addition to the well characterized, slowly activating I(h). 12121985_gating mechanisms in mouse HCN2 pacemaker channel and sensitivity to voltage, mutations and cations 12193608_a conserved domain in HCN2 is the critical NH(2)-terminal determinant of subunit co-assembly and trafficking of pacemaker channels 12433951_Major contributions of HCN1 and HCN2 channel isoforms to hyperpolarization-activated, non-selective cation current. HL-1 cells display a hyperpolarization-activated current which might contribute to spontaneous contractile activity of these cells. 12514127_Results define the physiological role of the hyperpolarization-activated cation (HCN2) channel subunit as a major determinant of membrane resting potential that is required for regular cardiac and neuronal rhythmicity. 12562911_Gating and permeation of HCN channels are coupled, and that modulation of this 'pore-to-gate' coupling by external K+ is isoform-specific. 12968185_X-ray crystallographic structures of C-terminal fragment bound to cAMP or cGMP, identify a tetramerization domain and the mechanism for cyclic nucleotide specificity, and suggest a model for ligand-dependent channel modulation 14726518_a specific electrostatic interaction between Arg-339 of the S4-S5 linker and Asp-443 of the C-linker stabilizes the closed state and thus participates in the coupling of voltage sensing and activation gating in HCN channels. 15292205_In filamin- cells the gating kinetics of HCN1 were strongly accelerated relative to filamin+ cells. 15525777_These studies show that HCN1 and HCN2 channels in GABAergic GP neurons are key determinants of the regularity and rate of pacemaking as well as striatal resetting of this activity. 15644313_Data indicate that Arg-405 is a key component of a domain that allosterically couples chloride binding with hyperpolarization-activated cyclic nucleotide-gated channel (HCN1 and 2)activation. 15687126_Single-channel properties of cells cotransfected with mHCN2 & hHCN4 were distinct from cells expressing HCN2 or HCN4 alone. Different HCN isoforms influence current properties of a single HCN channel complex, providing for HCN heteromerization. 16177047_Somatic voltage-clamp experiments in slices of prelimbic and infralimbic cortex reveal the presence of rapidly activating and deactivating cationic currents attributable to HCN1/HCN2 channels 16500960_We propose that the cAMP-triggered large-scale oscillation plays an important role for the channel's function, being coupled to a motion of the C-linker which, in turn, modulates the gating of the channel. 16542642_Black Swiss mice have a single polymorphism in exon 2 within the Hcn2 gene, but we conclude that juvenile audiogenic seizures in Black Swiss mice are unlikely to be due to abnormalities of HCN2 channel function. 16760342_TASK3 and HCN2 channels stabilize the membrane potential by a mutual functional interactions. 16777944_With computer modelling, we show that in channels with relatively slow opening kinetics and fast mode-shift transitions, such as HCN2 and HCN4 channels, the mode shift effects are not readily observable, except in the tail kinetics. 16923750_Mouse HCN2 was injected into canine left bundle branch block increasing basal heart rate. 17074978_Thus, both native and recombinant HCN channels are regulated by PI(4,5)P(2). 17261842_A closed-open transition that is inherently voltage independent is subject to dual allosteric regulation by voltage sensor movement and cAMP binding. 17553794_HCN2-HCN4 heteromeric channels form in native tissue to make functional pacemaker channels 17562313_Using computer simulation and mutagenesis, ligand contacts important for the regulation of HCN2 by cyclic nucleotides were identified. 17578902_A site-directed mutagenesis approach was used to identify specific residues of the mouse HCN2 channel pore that interact with ZD7288. 18033814_Current through HCN2 and HCN3 channels confers regular and faster rhythmicity to mouse embryonic stem cells, which mirrors the developing embryonic mouse heart, and contributes to modulation of rhythmicity by autonomic stimulation. 18252758_Provide direct evidence for calcium conductance of HCN2 channels. 18579800_Results suggest that intracellular Mg(2+) acts as a voltage-dependent pore blocker and, therefore, reduces outward currents through HCN channels. 18768480_RPTPalpha plays a critical role in HCN channel function via tyrosine dephosphorylation 19150498_we characterized a novel spontaneous mutant, apathetic (ap/ap); identified ap mutation as a 4 base pair insertion in the coding region of Hcn2; ap/ap mice exhibited ataxia, generalized spike-wave absence seizures & rare generalized tonic-clonic seizures 19171659_In vitro characterization of HCN channel kinetics and frequency dependence in myocytes predicts biological pacemaker functionality. 19269964_Alanine/valine scan of the distal pore of the HCN2 isoform show that effects of mutations on channel opening and on the steepness of the response of the channel to voltage are mixed and smaller than those in Shaker channels. 19421833_we describe the transcription profiles of the four HCN genes from embryonic stage E9.5 dpc to postnatal day 120 in the mouse. 19574228_functional myocardial I(f) channels reflect the heteromeric assembly of HCN2 and HCN4 subunits and further that the HCN4 subunit underlies the cAMP-mediated regulation of cardiac I(f) channels. 20130205_The differences in evolution and fucntion of HCN1 and HCN2 are reported. 20140458_The expression pattern of HCN isoforms in the olfactory bulb of mice, is described. 20623157_HCN2 channels in inhibitory interneurons modulate synaptic plasticity in the perforant path by facilitating the GABAergic output onto pyramidal neurons. 21076425_results show that rendering rodents parkinsonian by DA depletion induces the downregulation of HCN channel expression in GPe neurons, leading to a loss of their characteristic autonomous pacemaking. 21347269_cGKII and HCN2 interact and colocalize with each other 21354395_new insights into the cAMP-dependent gating in HCN channel and the interpretation of protein allostery for general ligand-gated channels and receptors 21903816_findings show neuropathic pain is initiated by HCN2-driven action potential firing in Na(V)1.8-expressing nociceptors 21945247_HCN2 shows only a confined expression to individual pacemaker cells. 22279546_Data show that HCN1 and HCN2 channels are expressed at distinct retinal sites and serve different functions. 22652004_Suggest that the strongly increased HCN channel activity in hypertrophied myocytes prolongs the repolarization of the ventricular action potential and thereby may increase the arrhythmogenic potential. 22662315_Dendritic expression of the h-channel subunit HCN1, but not HCN2, was higher in the CA1 region of fmr1(-/y) mice. 22880098_Loss of KCNE2 leads to downregulation of HCN channel function associated with increased excitability in neurons in the cortico-thalamo-cortical loop. 22948144_Data indicate that HCN1 and HCN2 form alternate ternary protein complexes with hair-cell stereociliary proteins. 23048023_Leucine zipper motif essential for gating of HCN1 and HCN2. 23525108_cAMP-dependent changes in the structure of the C-helix of a C-terminal fragment of HCN2 channels 23609807_HCN2 channel expression increases significantly during pregnancy, accelerating the heart rate. 24085296_fisetin is a partial agonist for HCN2 channels. 25042871_This study demonstrated that Increased expression of hyperpolarization-activated cyclic nucleotide-gated (HCN) channels in reactive astrocytes following ischemia. 25562801_HCN2 channel activity reduces the repolarization reserve of the ventricular action potential and increases ectopic automaticity under pathological conditions such as excessive beta-adrenergic stimulation. 25761792_HCN1, HCN2, and HCN4 subunits may have distinct physiological roles in the developing hippocampus. 26559974_the allosteric mechanism of different cyclic nucleotides on the cyclic nucleotide-binding domain of HCN2 and on channel gating, was investigated. 27137910_Mouse HCN2-expressing human mesenchymal stem cells are relevant biological models for the generation of biological pacemakers. 27568927_Data support a model in which the degree of facilitation of opening in the full-length HCN2 channel by a given ligand depends upon its ability to promote oligomerization of the C-linker/cyclic nucleotide-binding domain in solution. 28767511_The data of this study strongly suggest that cAMP acts by PKA-mediated regulation of HCN2 channel activity and thereby generates nociceptive sensitization. 28954930_the increased intracellular cAMP drives diabetes-associated pain by facilitating HCN2 activation and consequently promoting repetitive firing in primary nociceptive nerve fibers. 29256865_Together, these results reveal a critical role for HCN2 in regulating ventral tegmental area dopamine neuronal activity and depressive-related behaviors. 29466436_loss via insertion mutation leads to delayed gastrointestinal motility and reduced energy intake 29519998_rescues nicotine-exposed Xenopus embryos, restoring the endogenous bioelectric prepatterns necessary for brain patterning, resulting in normal brain morphology 29980633_A mutant channel containing just five mutations (M485I, G497D, S514T, V562A, and S563G) switches cAMP sensitivity of full-length HCN2 to that of HCN1 channels. 30877792_Results establish that thalamic HCN4 channels are essential for the production of rhythmic intrathalamic oscillations and determine regular thalamocortical oscillatory activity during alert states. 31045576_Abolishing cAMP sensitivity in HCN2 pacemaker channels induces generalized seizures. 31087220_HCN2 channel was constitutively activated at the hyperpolarized RMP. 31496517_Computational modeling indicated that HCN2 channels control conduction velocity primarily by altering the resting membrane potential and are associated with significant metabolic costs. 31503201_Incorporation of one N-glycosylation-deficient subunit within a tetramer of HCN2 channel is tolerated. 31615893_Docking experiments suggested a pivotal role of Arg-635 at the entrance of the binding pocket in HCN2, either causing stabilizing cation-pi interactions with the aromatic ring in N (6)-Phe-cAMP or N (6)-Bn-cAMP or a steric clash with the aromatic ring in N (6)-Bnz-cAMP. 32184125_Pdpk1 (PDK1) and AKT are involved in the regulation of HCN2 and HCN4 mRNA transcription, protein expression, HCN current density, and cell membrane location 32341127_The HCN domain is required for HCN channel cell-surface expression and couples voltage- and cAMP-dependent gating mechanisms. 33181864_HCN channels in the mammalian cochlea: Expression pattern, subcellular location, and age-dependent changes. 34206649_Loss of HCN2 in Dorsal Hippocampus of Young Adult Mice Induces Specific Apoptosis of the CA1 Pyramidal Neuron Layer. 34948328_The Role of Prostaglandin E1 as a Pain Mediator through Facilitation of Hyperpolarization-Activated Cyclic Nucleotide-Gated Channel 2 via the EP2 Receptor in Trigeminal Ganglion Neurons of Mice. |
ENSG00000099822 |
HCN2 |
109.002696 |
0.4614229432 |
-1.115838 |
0.25386544 |
19.228269 |
0.00001159830775472274732576169475883887116651749238371849060058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000848944852090037982081383027832544030388817191123962402343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
68.8665016521548 |
12.1808056051978 |
149.248108859862 |
18.929684756787 |
| ENSMUSG00000020388 |
30794 |
Pdlim4 |
protein_coding |
P70271 |
FUNCTION: Suppresses SRC activation by recognizing and binding to active SRC and facilitating PTPN13-mediated dephosphorylation of SRC 'Tyr-419' leading to its inactivation. Inactivated SRC dissociates from this protein allowing the initiation of a new SRC inactivation cycle. Involved in reorganization of the actin cytoskeleton (By similarity). In nonmuscle cells, binds to ACTN1 (alpha-actinin-1), increases the affinity of ACTN1 to F-actin (filamentous actin), and promotes formation of actin stress fibers. Involved in regulation of the synaptic AMPA receptor transport in dendritic spines of hippocampal pyramidal neurons directing the receptors toward an insertion at the postsynaptic membrane. Links endosomal surface-internalized GRIA1-containing AMPA receptors to the alpha-actinin/actin cytoskeleton. Increases AMPA receptor-mediated excitatory postsynaptic currents in neurons (By similarity). {ECO:0000250|UniProtKB:P36202, ECO:0000250|UniProtKB:P50479}. |
Cell projection;Cytoplasm;Cytoskeleton;Endosome;LIM domain;Membrane;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Synapse;Synaptosome;Zinc |
|
|
mmu:30794; |
actin cytoskeleton [GO:0015629]; adherens junction [GO:0005912]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; dendritic spine [GO:0043197]; early endosome lumen [GO:0031905]; early endosome membrane [GO:0031901]; filamentous actin [GO:0031941]; lamellipodium [GO:0030027]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; postsynaptic membrane [GO:0045211]; recycling endosome lumen [GO:0034777]; recycling endosome membrane [GO:0055038]; stress fiber [GO:0001725]; Z disc [GO:0030018]; actin binding [GO:0003779]; actinin binding [GO:0042805]; alpha-actinin binding [GO:0051393]; metal ion binding [GO:0046872]; muscle alpha-actinin binding [GO:0051371]; protein homodimerization activity [GO:0042803]; protein phosphatase binding [GO:0019903]; actin cytoskeleton organization [GO:0030036]; actin cytoskeleton reorganization [GO:0031532]; excitatory chemical synaptic transmission [GO:0098976]; heart development [GO:0007507]; muscle structure development [GO:0061061]; positive regulation of stress fiber assembly [GO:0051496] |
15663004_RIL C-terminus proper, but not the RIL LIM domain, can interact with protein tyrosine phosphatase BL 19212833_PDLIM4 may function as a tumor suppressor, involved in the control of cell proliferation by associating with actin in prostate cancer cells. 31287961_Pdlim4 is required for dendritic cell migration via CCR7-JNK, dendrite formation, and subsequent development of functional T-cell responses. 31393857_findings have identified Pdlim4 as a JMJD3 target gene that affects T-cell trafficking by cooperating with S1P1, and provided insights into the molecular mechanisms by which JMJD3 regulates genes involved in T cell trafficking. |
ENSG00000131435 |
PDLIM4 |
84.967606 |
0.4375352904 |
-1.192529 |
0.29351005 |
16.459303 |
0.00004970566187317245257764469767991499793424736708402633666992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003191891406628005224688227059459677548147737979888916015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
50.4816050510453 |
10.3412583352219 |
115.377219077704 |
16.9334740269983 |
| ENSMUSG00000020493 |
270906 |
Prr11 |
protein_coding |
Q8BHE0 |
FUNCTION: Plays a critical role in cell cycle progression. {ECO:0000250|UniProtKB:Q96HE9}. |
Cytoplasm;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:270906; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; regulation of cell cycle [GO:0051726] |
35980531_Identification of proline-rich protein 11 as a major regulator in mouse spermatogonia maintenance via an increase in BMI1 protein stability. |
ENSG00000068489 |
PRR11 |
647.971353 |
2.2312833454 |
1.157874 |
0.10566797 |
118.992223 |
0.00000000000000000000000000105140925199224537230498673421868411210259730775264791771268511358007358711189049849110688228392973542213439941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000687341876667205245115674512459312641244013721745017290855016100817559236701992375628833542577922344207763671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
881.540742817085 |
64.6248348719866 |
395.082383696111 |
22.5214247121511 |
| ENSMUSG00000020534 |
20425 |
Shmt1 |
protein_coding |
P50431 |
FUNCTION: Interconversion of serine and glycine. {ECO:0000250|UniProtKB:P34896}. |
3D-structure;Cytoplasm;Direct protein sequencing;One-carbon metabolism;Pyridoxal phosphate;Reference proteome;Transferase |
PATHWAY: One-carbon metabolism; tetrahydrofolate interconversion. {ECO:0000250|UniProtKB:P34896}. |
|
mmu:20425; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; mitochondrion [GO:0005739]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; amino acid binding [GO:0016597]; glycine hydroxymethyltransferase activity [GO:0004372]; identical protein binding [GO:0042802]; L-allo-threonine aldolase activity [GO:0008732]; mRNA 5'-UTR binding [GO:0048027]; mRNA regulatory element binding translation repressor activity [GO:0000900]; protein homodimerization activity [GO:0042803]; pyridoxal phosphate binding [GO:0030170]; serine binding [GO:0070905]; small molecule binding [GO:0036094]; zinc ion binding [GO:0008270]; cellular response to leukemia inhibitory factor [GO:1990830]; cellular response to tetrahydrofolate [GO:1904482]; dTMP biosynthetic process [GO:0006231]; folic acid metabolic process [GO:0046655]; glycine biosynthetic process [GO:0006545]; glycine biosynthetic process from serine [GO:0019264]; glycine metabolic process [GO:0006544]; L-serine catabolic process [GO:0006565]; L-serine metabolic process [GO:0006563]; negative regulation of translation [GO:0017148]; one-carbon metabolic process [GO:0006730]; protein homotetramerization [GO:0051289]; purine nucleobase biosynthetic process [GO:0009113]; tetrahydrofolate interconversion [GO:0035999]; tetrahydrofolate metabolic process [GO:0046653] |
18644786_mitochondrial SHMT-derived one-carbon units are essential for folate-mediated one-carbon metabolism in the cytoplasm 19513116_SHMT1 and SHMT2 are functionally redundant in nuclear de novo thymidylate biosynthesis 20977478_Shmt1 (SHMT1), but not Srr, is likely to be one of the genetic components regulating prepulse inhibition in mice and possibly relevant to human schizophrenia. 21226315_It is one of the genetic components regulating PPI in mice and is relevant to schizophrenia susceptibility in humans. (review) 21346092_These results provide evidence that disruption of Shmt1 expression causes NTDs by impairing thymidylate biosynthesis and shows that changes in the expression of genes that encode folate-dependent enzymes may be key determinates of NTD risk. 21406397_Data show that Shmt1 hemizygosity was associated with increased risk for intestinal cancer in Apc(min)(/+) mice through a gene-by-diet interaction. 22057276_SHMT1 and TYMS localization to the nucleus is essential to prevent uracil accumulation in DNA 23700346_Shmt1 and dietary folate deficiency influence metabolic markers of homocysteine remethylation in mice. |
ENSG00000176974 |
SHMT1 |
366.463241 |
2.6219759421 |
1.390654 |
0.17195551 |
63.199851 |
0.00000000000000186762557951066361012123353182996731822572834651186024501612337189726531505584716796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000494742136123590032125029231360263887520845038636707613477483391761779785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
529.497949938899 |
63.2606502787508 |
201.946151160911 |
18.3110273385836 |
| ENSMUSG00000020541 |
71943 |
Tom1l1 |
protein_coding |
Q923U0 |
FUNCTION: Probable adapter protein involved in signaling pathways. Interacts with the SH2 and SH3 domains of various signaling proteins when it is phosphorylated. May promote FYN activation, possibly by disrupting intramolecular SH3-dependent interactions. {ECO:0000269|PubMed:11711534}. |
Alternative splicing;Cytoplasm;Endosome;Golgi apparatus;Membrane;Phosphoprotein;Protein transport;Reference proteome;SH3-binding;Transport |
|
|
Human_homologues hsa:10040; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; endosome [GO:0005768]; endosome membrane [GO:0010008]; Golgi stack [GO:0005795]; membrane [GO:0016020]; clathrin binding [GO:0030276]; phosphatidylinositol binding [GO:0035091]; protein kinase activator activity [GO:0030295]; protein kinase binding [GO:0019901]; SH3 domain binding [GO:0017124]; ubiquitin binding [GO:0043130]; activation of protein kinase activity [GO:0032147]; negative regulation of mitotic nuclear division [GO:0045839]; positive regulation of protein autophosphorylation [GO:0031954]; protein transport [GO:0015031]; signal transduction [GO:0007165]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
16479011_Tom1L1 defines a novel mechanism for regulation of Src family of protein-tyrosine kinases mitogenic signaling induced by growth factors. 17046829_Srcasm may act as a molecular 'rheostat' for activated Src family tyrosine kinases 17785434_Tom1L1-clathrin heavy chain (CHC) complex negatively regulates the level of SFK in caveolae needed for the induction of DNA synthesis. |
ENSG00000141198 |
TOM1L1 |
148.188450 |
3.1852312708 |
1.671398 |
0.22884601 |
52.257638 |
0.00000000000048676235752497056087138814363410363580866174748607022593205329030752182006835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000101289128822682388878036840922212838498467357695176360721234232187271118164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
229.162017911038 |
36.5032202332224 |
71.9451739693143 |
8.85134679309733 |
| ENSMUSG00000020577 |
66109 |
Tspan13 |
protein_coding |
Q9D8C2 |
|
Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:66109; |
plasma membrane [GO:0005886]; calcium channel regulator activity [GO:0005246]; regulation of calcium ion transmembrane transport [GO:1903169] |
17965585_Down-regulated in pre-osteoclasts during cell differentiation. |
ENSG00000106537 |
TSPAN13 |
12.530661 |
0.0637271260 |
-3.971949 |
1.15143927 |
10.889227 |
0.00096725169642011745579912584958037768956273794174194335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0044836207668506478790648728249834675807505846023559570312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.45510643628691 |
1.28862627270815 |
22.8333918109379 |
11.9964919785461 |
| ENSMUSG00000020589 |
76820 |
Cyria |
protein_coding |
Q8BHZ0 |
FUNCTION: May negatively regulate RAC1 signaling and RAC1-driven cytoskeletal remodeling. May regulate chemotaxis, cell migration and epithelial polarization by controlling the polarity, plasticity, duration and extent of protrusions. {ECO:0000250|UniProtKB:Q9H0Q0}. |
Lipoprotein;Membrane;Reference proteome |
|
|
mmu:76820; |
membrane [GO:0016020]; small GTPase binding [GO:0031267]; regulation of actin filament polymerization [GO:0030833] |
Human_homologues 29949196_confirmed that three genes (rs7922405 of MGMT, rs73039426 of RHPN2, and rs7552 of FAM49A) may contribute to nonsyndromic orofacial clefts in Chinese populations 34165494_CYRI-A limits invasive migration through macropinosome formation and integrin uptake regulation. |
ENSG00000197872 |
CYRIA |
84.525018 |
8.4615665498 |
3.080925 |
0.65244654 |
18.859033 |
0.00001407430022292157040068871776128389683435671031475067138671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001015055591834949569448806605542756642535096034407615661621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
151.575898858225 |
70.9095504027916 |
17.9134558554042 |
6.6252652876014 |
| ENSMUSG00000020646 |
67216 |
Mboat2 |
protein_coding |
Q8R3I2 |
FUNCTION: Acyltransferase which catalyzes the transfert of an acyl group from an acyl-CoA to a lysophospholipid leading to the production of a phospholipid and participates in the reacylation step of the phospholipid remodeling pathway also known as the Lands cycle (PubMed:18287005, PubMed:29196633). Catalyzes the acylation of lysophosphatidylcholine (1-acyl-sn-glycero-3-phosphocholine or LPC) and to a lesser extend lysophosphatidylethanolamine (1-acyl-sn-glycero-3-phosphoethanolamine or LPE) (PubMed:18287005, PubMed:29196633). Does not acylates lysophosphatidic acid (LPA) and lysophosphatidylserine (PubMed:18287005). Prefers oleoyl-CoA as the acyl donor (PubMed:18287005, PubMed:29196633). May be involved in chondrocyte differentiation (PubMed:29196633). {ECO:0000269|PubMed:18287005, ECO:0000269|PubMed:29196633}. |
Acyltransferase;Alternative splicing;Endoplasmic reticulum;Lipid biosynthesis;Lipid metabolism;Membrane;Phospholipid biosynthesis;Phospholipid metabolism;Reference proteome;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Lipid metabolism; phospholipid metabolism. {ECO:0000269|PubMed:18287005}. |
|
mmu:67216; |
endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; 1-acylglycerophosphocholine O-acyltransferase activity [GO:0047184]; 1-acylglycerophosphoethanolamine O-acyltransferase activity [GO:0106262]; 1-acylglycerophosphoserine O-acyltransferase activity [GO:0106263]; acyltransferase activity [GO:0016746]; lipid modification [GO:0030258]; phosphatidylcholine acyl-chain remodeling [GO:0036151]; phosphatidylethanolamine acyl-chain remodeling [GO:0036152]; phosphatidylserine acyl-chain remodeling [GO:0036150]; phospholipid biosynthetic process [GO:0008654]; regulation of chondrocyte differentiation [GO:0032330] |
Human_homologues 33832516_Circ-MBOAT2 knockdown represses tumor progression and glutamine catabolism by miR-433-3p/GOT1 axis in pancreatic cancer. 36635270_CircRNA MBOAT2 promotes intrahepatic cholangiocarcinoma progression and lipid metabolism reprogramming by stabilizing PTBP1 to facilitate FASN mRNA cytoplasmic export. |
ENSG00000143797 |
MBOAT2 |
110.254045 |
3.6818764930 |
1.880441 |
0.26497051 |
49.886039 |
0.00000000000162939941837067990339466049694055444049985148513570720751886256039142608642578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000316569029854874904191920022626628109657653808994837163481861352920532226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
179.960432785022 |
33.2383088603637 |
48.8773681376717 |
7.14432889077769 |
| ENSMUSG00000020758 |
192897 |
Itgb4 |
protein_coding |
A2A863 |
FUNCTION: Integrin alpha-6/beta-4 is a receptor for laminin. It plays a critical structural role in the hemidesmosome of epithelial cells. Is required for the regulation of keratinocyte polarity and motility. ITGA6:ITGB4 binds to NRG1 (via EGF domain) and this binding is essential for NRG1-ERBB signaling. ITGA6:ITGB4 binds to IGF1 and this binding is essential for IGF1 signaling. ITGA6:ITGB4 binds to IGF2 and this binding is essential for IGF2 signaling. {ECO:0000250|UniProtKB:P16144}. |
Alternative splicing;Cell adhesion;Cell junction;Cell membrane;Disulfide bond;Glycoprotein;Integrin;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
Integrins are heterodimers comprised of alpha and beta subunits, that are noncovalently associated transmembrane glycoprotein receptors. Different combinations of alpha and beta polypeptides form complexes that vary in their ligand-binding specificities. Integrins mediate cell-matrix or cell-cell adhesion, and transduced signals that regulate gene expression and cell growth. This gene encodes the integrin beta 4 subunit, a receptor for the laminins. This subunit tends to associate with alpha 6 subunit and is likely to play a pivotal role in the biology of invasive carcinoma. Multiple alternatively spliced transcript variants encoding distinct isoforms have been found for this gene. [provided by RefSeq, Jul 2008]. |
|
basal plasma membrane [GO:0009925]; basement membrane [GO:0005604]; cell cortex [GO:0005938]; cell junction [GO:0030054]; cell leading edge [GO:0031252]; cell surface [GO:0009986]; cytoplasm [GO:0005737]; focal adhesion [GO:0005925]; glutamatergic synapse [GO:0098978]; hemidesmosome [GO:0030056]; integrin complex [GO:0008305]; membrane [GO:0016020]; nuclear membrane [GO:0031965]; nucleolus [GO:0005730]; plasma membrane [GO:0005886]; postsynaptic membrane [GO:0045211]; receptor complex [GO:0043235]; G protein-coupled receptor binding [GO:0001664]; insulin-like growth factor I binding [GO:0031994]; integrin binding [GO:0005178]; neuregulin binding [GO:0038132]; autophagy [GO:0006914]; cell adhesion [GO:0007155]; cell adhesion mediated by integrin [GO:0033627]; cell migration [GO:0016477]; cell motility [GO:0048870]; cell-matrix adhesion [GO:0007160]; filopodium assembly [GO:0046847]; hemidesmosome assembly [GO:0031581]; integrin-mediated signaling pathway [GO:0007229]; mesodermal cell differentiation [GO:0048333]; myelination in peripheral nervous system [GO:0022011]; nail development [GO:0035878]; peripheral nervous system myelin formation [GO:0032290]; response to wounding [GO:0009611]; skin morphogenesis [GO:0043589]; trophoblast cell migration [GO:0061450] |
12070014_Expression of the beta 4 subunit of the alpha 6 beta 4 integrin induces monocytic differentiation of 32D/v-Abl transformed cells. alpha 6 beta 4 integrin promotes growth arrest and differentiation by modulating Abl kinases and p73 protein pathway(s). 15161909_alpha(6)beta(4) has a generic influence on the invasion of carcinoma cells that is not specific to Met 15731010_integrin alpha6beta4 is not required for postnatal epidermal differentiation, proliferation and survival, provided that cell adhesion is not compromised 15988021_Integrin beta4 signaling promotes epidermal growth and wound healing through an effect on nuclear translocation of NF-kappaB and mitogen-activated protein kinases. 16268053_both beta1 & beta4 integrins may have a role in implantation since immunoreactivity was seen on apical membrane of surface epithelium on day 4 when implantation occurred 16365040_alpha5 is required for proliferation and polarity of basal epithelial cells; the interaction between laminin-10/11-integrin alpha6beta4 and the PI3-kinase-Cdc42/Rac pathways may play a role in determining the size and shape of tooth germ 16436605_PMP22 is a binding partner in the integrin alpha6beta4/laminin complex. The levels of beta4 integrin are severely reduced in nerves of PMP22-deficient mice. 16901783_These results indicate that beta 4 integrin promotes tumor progression by amplifying ErbB2 signaling and identify beta 4 as a potential target for molecular therapy of breast cancer. 17699601_alpha6beta4 can either mediate an adhesion-independent tumor-suppressive effect or act as a tumor promotor 18006359_Data suggest that integrin beta4 is a key factor in neuron survival and apoptosis and that this integrin subunit might perform its action through regulating NADPH oxidase and the level of reactive oxygen species in neuronal survival and apoptosis. 18940803_ZKSCAN3 regulates the expression of several genes favoring tumor progression including integrin beta 4 and VEGF 19087978_Integrin beta-4 signaling plays a key role in mouse embryogenesis. 19193855_Combinatorial selection based on phage display technology revealed a previously unrecognized complex between the soluble protein netrin-4 and laminin gamma1 subunit that activates an alpha6beta1 integrin-mediated signaling pathway in neural stem cells 19419971_novel model for the disassembly of hemidesmosomes where both binding of calmodulin to plectin 1a and phosphorylation of integrin beta4 by protein kinases, are required for disruption of the integrin alpha6beta4-plectin complex 19549682_report regulation of beta 4-integrin expression by epigenetic modifications in the mammary gland and during the epithelial-to-mesenchymal transition. 20509141_Show increased integrin b4 in the endothelium of mouse thoracic aortas during natural aging and atherosclerosis. These data suggest that integrin b4 might be a potential target for therapy of cardiovascular diseases. 20544851_cerebral hypoxia promotes first an endothelial response followed by astrocyte activation, proliferation, and reorganization of astrocyte end-feet, which correlates with increased expression of adhesion molecules alpha 6 beta 4 integrin and dystroglycan 20870721_EGF-induced MAPK signaling inhibits hemidesmosome formation through phosphorylation of the integrin beta 4. 20937830_CD151 controls Met-dependent neoplastic growth by enhancing receptor signaling through beta4 integrin-mediated pathways, independent of cell-substrate adhesion 21556948_Integrin beta4 signaling promotes mammary tumor cell adhesion to brain microvascular endothelium by inducing ErbB2-mediated secretion of VEGF 21701069_Data show that a previously unrecognized subpopulation of mouse alveolar epithelial cells expressing the laminin receptor alpha6beta4, but little or no pro-surfactant C, is endowed with regenerative potential. 21750188_Data show that beta4 integrin associates with MMP9 and that its ectodomain is a target for cleavage by MMP9 in vivo under pathological conditions. 22205679_The expression of beta4 integrin in interstitial, vessel-associated cells with myogenic activity within adult skeletal muscle. 22464766_aberrant expression of alpha6beta4 integrin in post-mitotic epidermal keratinocytes stimulates a pro-tumorigenic skin microenvironment 22995187_Deficiency of integrin alpha(6)beta(4) is modestly but significantly suppressed reporter-gene transduction by papillomaviruses in conditional integrin beta(4) knockout mice. 23348745_Targeted deletion of the signaling domain of beta4 inhibited prostate tumor growth and progression in response to loss of p53 and Rb function in a model of prostate cancer (PB-TAg mice). 23471230_Lack of endothelial beta4 integrin had no effect on vascular development, integrity, or endothelial proliferation 23878399_Findings indicate a function for Id2 in regulating Snai1-mediated repression of integrin beta4. 24007983_Both annexin A7 and integrin beta4 were essential for small molecule, 6-amino-2, 3-dihydro-3-hydroxymethyl-1, 4-benzoxazine-induced autophagy. 24505282_Col17a1/ keratinocytes exhibited increased spreading on laminin 332 and accelerated, but less directed cell motility. These effects were accompanied by increased expression of the integrin subunits beta4 and beta1. 25326539_Ectopic induction of Nur77 in endothelial cells is sufficient to improve skin wound healing. Furthermore, TR3/Nur77 regulates the expression of integrin beta4 by targeting its promoter activity. 25637531_Up-regulation of integrin beta4 and TRPV1 in migrating keratinocytes. 25668239_keratins have a role in controlling beta4-integrin endocytosis involving a plectin-Erk1/2-dependent mechanism relevant for epidermal differentiation and pathogenesis 26432258_Integrin beta4 contributes to mammary gland development by sustaining PTHrP expression and enabling PTHrP signaling. 27881869_he potential effect of Ntn-1 on angiogenesis is further verified by the mouse hindlimb ischemia model, where the pre-activation of hUCB-MSCs with Ntn-1 significantly improved vascular regeneration. These results demonstrate that Ntn-1 plays an important role in the tissue regeneration process of hUCB-MSC via the lipid raft-mediated Inalpha6beta4 signaling pathway. 28708243_MARVELD1 mediated the invasion of trophoblast cells via regulation of the expression of integrin beta4 during placenta development. 29315582_These data indicate that integrin alpha6beta4 and PMP22 cooperate to organize the internode and to allow proper propagation of the axonal signal along the axons. 29393977_ITGB4 deficiency leads to severe allergy-induced airway inflammation and airway hyper-responsiveness. 30170608_The results of this study suggested that ITGB4 knockout induced chronic inflammation mediated the comorbidity of asthma and bipolar disorder. 31586558_Study reports that alveolar progenitor cells in the mouse mammary gland express alpha6 beta4 integrin based on analysis of single cell RNA-Seq data. Subsequent experiments using a mouse mammary epithelial cell line confirmed this finding and revealed that beta4 is essential for maintaining progenitor function as assessed by serial passage mammosphere assays. 31970850_Airway epithelial ITGB4 deficiency in early life mediates pulmonary spontaneous inflammation and enhanced allergic immune response. 32608482_Airway epithelial integrin beta4 suppresses allergic inflammation by decreasing CCL17 production. 32618381_DNA methylation down-regulates integrin beta4 expression in asthmatic airway epithelial cells. 34018612_Airway epithelial integrin beta4-deficiency exacerbates lipopolysaccharide-induced acute lung injury. 35173551_Integrin-beta4 regulates the dynamic changes of phenotypic characteristics in association with epithelial-mesenchymal transition (EMT) and RhoA activity in airway epithelial cells during injury and repair. 35394541_KRAS Mutants Upregulate Integrin beta4 to Promote Invasion and Metastasis in Colorectal Cancer. 36243221_Airway epithelial ITGB4 deficiency induces airway remodeling in a mouse model. 36282138_ITGB4 deficiency induces DNA damage by downregulating HDAC1 in airway epithelial cells under stress stimulation. 36293483_Lack of NPR1 Increases Vascular Endothelial Adhesion through Induction of Integrin Beta 4. |
ENSG00000132470 |
ITGB4 |
214.415431 |
2.9952026351 |
1.582654 |
0.23228463 |
45.430604 |
0.00000000001581438704068477575024797390269588187594607120445289183408021926879882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000002762439717932459597830469314632458738278941723365278448909521102905273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
321.695259434025 |
52.019405405219 |
107.403504412057 |
13.4148515946578 |
| ENSMUSG00000020774 |
11484 |
Aspa |
protein_coding |
Q8R3P0 |
FUNCTION: Catalyzes the deacetylation of N-acetylaspartic acid (NAA) to produce acetate and L-aspartate. NAA occurs in high concentration in brain and its hydrolysis NAA plays a significant part in the maintenance of intact white matter (By similarity). {ECO:0000250}. |
Cytoplasm;Direct protein sequencing;Hydrolase;Metal-binding;Nucleus;Reference proteome;Zinc |
|
This gene encodes an enzyme that deacteylates N-acetyl-L-aspartic acid (NAA) in the brain to yield acetate and L-aspartate. In humans, alterations in neuronal NAA concentration are associated with many neurodegenerative diseases (decrease associated with epilepsy, multiple sclerosis, myotrophic lateral sclerosis, and Alzheimer's disease; increase associated with Canavan disease). In mouse, mutations in this gene, which cause accumulation of NAA, result in demyelination and spongy degeneration in the CNS and serve as a pathophysiological model for Canavan disease. [provided by RefSeq, Dec 2012]. |
mmu:11484; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleus [GO:0005634]; aminoacylase activity [GO:0004046]; aspartoacylase activity [GO:0019807]; hydrolase activity, acting on carbon-nitrogen (but not peptide) bonds, in linear amides [GO:0016811]; hydrolase activity, acting on ester bonds [GO:0016788]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; acetate metabolic process [GO:0006083]; central nervous system myelination [GO:0022010]; positive regulation of oligodendrocyte differentiation [GO:0048714] |
15686967_ASPA gene defect in the mouse leads to spinal cord pathology; these changes may be involved in the cause of the physiological/behavioral abnormalities seen in the knockout mouse. 16707098_normal hydrolysis of N-acetylaspartic acid is affected by aspartoacylase activity seen in the type 2 diabetes model mouse 17254025_we now present immunoblot, proteomic, and biochemical evidence that the membrane-bound form of ASPA is intrinsic to purified myelin membranes. 18987190_Nur7( Q193X) is a nonsense mutation in the mouse aspartoacylase gene that causes spongy degeneration of the CNS. 19739253_Death of OLs, neurons, and astrocytes was identified in every region of the aspartoacylase KO brain. These findings support involvement of ASPA in CNS development and function. 20637282_Aspa defects were associated with increased levels of acetylated histone H3, decreased chromatin compaction and increased GFAP protein, a marker for astrogliosis. Early in postnatal development ASPA defic affects oligodendrocyte maturation and myelination 21625469_Intracellular vacuolisation in astrocytes coincides with axonal swellings in cerebellum and brain stem of aspa(lacZ/lacZ) mutants indicating that astroglia may act as an osmolyte buffer in the aspa-deficient CNS 22617649_aspartoacylase is proposed here to support lipid synthesis and energy metabolism via the provision of substrate for both cellular processes during early postnatal development 25024137_Gata6 regulates differentiation, metabolism, and survival of resident peritoneal macrophages through aspartoacylase. 27717881_This study demonstrated that N-acetylaspartate supports the energetic demands of developmental myelination via oligodendroglial aspartoacylase. |
ENSG00000108381 |
ASPA |
34.968801 |
0.4150248175 |
-1.268730 |
0.45741291 |
7.527153 |
0.00607758652021595565523304216526412346865981817245483398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0219733676420435404541553481294613447971642017364501953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.327470844795 |
6.87583024463719 |
51.3884229238095 |
11.7962636159662 |
| ENSMUSG00000020788 |
53313 |
Atp2a3 |
protein_coding |
Q64518 |
FUNCTION: This magnesium-dependent enzyme catalyzes the hydrolysis of ATP coupled with the transport of calcium. Transports calcium ions from the cytosol into the sarcoplasmic/endoplasmic reticulum lumen. Contributes to calcium sequestration involved in muscular excitation/contraction. {ECO:0000250|UniProtKB:Q93084}. |
Acetylation;Alternative splicing;ATP-binding;Calcium;Calcium transport;Endoplasmic reticulum;Ion transport;Magnesium;Membrane;Metal-binding;Nucleotide-binding;Phosphoprotein;Reference proteome;Sarcoplasmic reticulum;Translocase;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:53313; |
endoplasmic reticulum [GO:0005783]; membrane [GO:0016020]; organelle membrane [GO:0031090]; sarcoplasmic reticulum [GO:0016529]; sarcoplasmic reticulum membrane [GO:0033017]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; calcium ion transmembrane transporter activity [GO:0015085]; calcium-dependent ATPase activity [GO:0030899]; cysteine-type endopeptidase activator activity involved in apoptotic process [GO:0008656]; metal ion binding [GO:0046872]; P-type calcium transporter activity [GO:0005388]; P-type ion transporter activity [GO:0015662]; transmembrane transporter binding [GO:0044325]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; calcium ion transmembrane transport [GO:0070588]; calcium ion transport from cytosol to endoplasmic reticulum [GO:1903515]; cellular calcium ion homeostasis [GO:0006874]; endoplasmic reticulum calcium ion homeostasis [GO:0032469]; intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress [GO:0070059]; ion transmembrane transport [GO:0034220]; negative regulation of receptor binding [GO:1900121] |
12119294_identification of promoter region and binding site for Ets-1 in mouse SERCA3 12401716_Ablation does not impair insulin secretion but suggests distinct roles of different sarcoendoplasmic reticulum Ca(2+) pumps for Ca(2+) homeostasis in pancreatic beta-cells 12524443_regulation in insulin-secreting beta-cells by insulin receptor substrate 1 15161756_IRS-1 modulation of insulin secretion is associated with Ca(2+) signaling and expression of SERCA-2b and -3 genes in pancreatic islets. Direct link between insulin resistance and defective insulin secretion. 21885870_This study evaluated the role of SERCA2b and SERCA3 in the control of the free calcium concentration in the endoplasmic reticulum and the role of SERCA3 in the control of insulin secretion. 25027872_ANP counter-regulates cardiac MR activation in hypertensive heart disease. An imbalance in cardiac ANP/GC-A (inhibition) and aldosterone/MR signaling (augmentation) favors adverse cardiac remodeling in chronic pressure overload. 27301859_SERCA3-dependent Ca(2+) stores control a specific ADP secretion pathway. 32588751_NAADP/SERCA3-Dependent Ca(2+) Stores Pathway Specifically Controls Early Autocrine ADP Secretion Potentiating Platelet Activation. |
ENSG00000074370 |
ATP2A3 |
96.393721 |
5.6831197170 |
2.506683 |
0.29462095 |
71.061717 |
0.00000000000000003462234142455387770159975772246583832174386709799353278871336669908487237989902496337890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000010806711159401735604386570157583681786837577053911330082769381988327950239181518554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
162.556892538397 |
33.4278871065345 |
28.6034608865818 |
4.78831892387274 |
| ENSMUSG00000020793 |
14428 |
Galr2 |
protein_coding |
O88854 |
FUNCTION: Receptor for the hormone galanin, GALP and spexin-1. The activity of this receptor is mediated by G proteins that activate the phospholipase C/protein kinase C pathway (via G(q)) and that inhibit adenylyl cyclase (via G(i)). {ECO:0000250|UniProtKB:O43603, ECO:0000269|PubMed:9832122}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:14428; |
cilium [GO:0005929]; plasma membrane [GO:0005886]; G protein-coupled peptide receptor activity [GO:0008528]; galanin receptor activity [GO:0004966]; neuropeptide binding [GO:0042923]; peptide hormone binding [GO:0017046]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; adenylate cyclase-modulating G protein-coupled receptor signaling pathway [GO:0007188]; G protein-coupled receptor signaling pathway [GO:0007186]; galanin-activated signaling pathway [GO:0090663]; inositol phosphate metabolic process [GO:0043647]; negative regulation of adenylate cyclase activity [GO:0007194]; neuron projection development [GO:0031175]; neuropeptide signaling pathway [GO:0007218]; phosphatidylinositol metabolic process [GO:0046488]; phospholipase C-activating G protein-coupled receptor signaling pathway [GO:0007200]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of large conductance calcium-activated potassium channel activity [GO:1902608]; positive regulation of transcription by RNA polymerase II [GO:0045944] |
15899880_To gain insight into the possible physiological significance of GALR2, phenotypic analysis of mice deficient in GALR2 was determined. 17076662_lack of GalR2 activation in galanin knockout and GalR2-MUT animals is responsible for the observed developmental deficits in the dorsal root ganglia 17257664_the discovery of an anxiogenic phenotype specific to the elevated plus-maze highlights the potential therapeutic efficacy of targeting GalR2 receptors in treating anxiety disorders 17263796_These findings imply that the rise in endogenous galanin observed either after brain injury or in various disease states is an adaptive response that reduces apoptosis by the activation of GalR2, and hence Akt and ERK. 17627995_Galr2 receptors stimulate vagal afferent mechanosensitivity in the gastro-esophageal afferent preparation. 18554714_The present findings are consistent with a predicted antidepressant-like effect of GalR2 signaling, suggesting that GalR2 might be a valid drug target for depressive disorders. 19127771_The result indicate that galanin and its receptors (GalR1 and GalR2) might regulate neurite extension in differentiating neural stem cells and even participate in the development of the nervous system. 21496293_High level of endogenous galanin in injured primary afferents activates peripheral GalR2, which leads to an increase in C-fibre mechanical activation thresholds and a marked reduction in evoked and ongoing nociceptive responses. 21672612_Three age groups of the Galr2 overexpressing mice demonstrated decreased levels of immobility in the forced swim test, indicative of antidepressive-like behavior and/or increased stress resistance. 23639882_In chronic restraint stress, the expression of GalR2 is increased in the nucleus accumbens following antidepressant administration. 29183756_these results identify that GALR2 is a regulator of insulin resistance and activation of GALR2 represents a promising strategy against obesity-induced insulin resistance. 30298565_The data of this study support a specific role of GAL2-R but not of GAL3-R in mediating the anticonvulsive actions of endogenous galanin. 31730796_Palmitate and Nitric Oxide Regulate the Expression of Spexin and Galanin Receptors 2 and 3 in Hypothalamic Neurons. 31953035_These data demonstrate that Galanin (Gal) contributes not only to cholangiocyte proliferation but also to liver fibrogenesis via the coordinate activation of GalR1 in cholangiocytes and GalR2 in hepatic stellate cells (HSCs). 32231393_molecular structures that confer ligand selectivity for galanin type II and III receptors 33915732_Involvement of Neuropeptide Galanin Receptors 2 and 3 in Learning, Memory and Anxiety in Aging Mice. |
ENSG00000182687 |
GALR2 |
39.477843 |
2.0534692943 |
1.038063 |
0.40589666 |
6.461243 |
0.01102525070416040392506928924376552458852529525756835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0363892998322364671914819211906433338299393653869628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
49.8834777854504 |
14.2204093800126 |
24.2922930107664 |
5.38644217994703 |
| ENSMUSG00000020798 |
77577 |
Spns3 |
protein_coding |
Q9D232 |
FUNCTION: Sphingolipid transporter. {ECO:0000250}. |
Alternative splicing;Lipid transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:77577; |
membrane [GO:0016020]; transmembrane transporter activity [GO:0022857]; lipid transport [GO:0006869] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 34608834_Bioinformatics-based identification of SPNS3 (Spinster homolog 3) as a prognostic biomarker of apoptosis resistance in acute myeloid leukemia. 35249471_Upregulation of FHL1, SPNS3, and MPZL2 predicts poor prognosis in pediatric acute myeloid leukemia patients with FLT3-ITD mutation. |
ENSG00000182557 |
SPNS3 |
55.651008 |
0.3005464681 |
-1.734340 |
0.40748829 |
17.586778 |
0.00002744902248521603516139347189728425746579887345433235168457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001865700626929097261791357098559274163562804460525512695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.1674355466076 |
7.48964058208974 |
87.0661888405964 |
17.6243815600953 |
| ENSMUSG00000020805 |
237831 |
Slc13a5 |
protein_coding |
Q67BT3 |
FUNCTION: High-affinity sodium/citrate cotransporter that mediates citrate entry into cells, which is a critical participant of biochemical pathways (PubMed:35448538, PubMed:26324167, PubMed:14656221). May function in various metabolic processes in which citrate has a critical role such as energy production (Krebs cycle), fatty acid synthesis, cholesterol synthesis, glycolysis, and gluconeogenesis (PubMed:12826022). Transports citrate into the cell in a Na(+)-dependent manner, recognizing the trivalent form of citrate (physiological pH) rather than the divalent form (PubMed:12826022, PubMed:14656221, PubMed:26324167). Can recognizes succinate as a substrate, but its affinity for succinate is several fold lower than for citrate (PubMed:26324167, PubMed:14656221). The stoichiometry is probably 4 Na(+) for each carboxylate, irrespective of whether the translocated substrate is divalent or trivalent, rendering the process electrogenic (PubMed:26324167, PubMed:14656221). Involved in the regulation of citrate levels in the brain (PubMed:32682952). {ECO:0000269|PubMed:12826022, ECO:0000269|PubMed:14656221, ECO:0000269|PubMed:26324167, ECO:0000269|PubMed:32682952, ECO:0000269|PubMed:35448538}. |
Cell membrane;Glycoprotein;Ion transport;Membrane;Reference proteome;Sodium;Sodium transport;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:237831; |
basolateral plasma membrane [GO:0016323]; cytosol [GO:0005829]; membrane [GO:0016020]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; citrate transmembrane transporter activity [GO:0015137]; identical protein binding [GO:0042802]; organic acid:sodium symporter activity [GO:0005343]; sodium:dicarboxylate symporter activity [GO:0017153]; succinate transmembrane transporter activity [GO:0015141]; tricarboxylic acid transmembrane transporter activity [GO:0015142]; alpha-ketoglutarate transport [GO:0015742]; anion transmembrane transport [GO:0098656]; cellular response to lithium ion [GO:0071285]; citrate transport [GO:0015746]; fumarate transport [GO:0015741]; oxaloacetate transport [GO:0015729]; succinate transport [GO:0015744]; tricarboxylic acid transport [GO:0006842] |
14656221_Molecular cloning and transport properties of NaCT. 16516867_Na+ -coupled di/tricarboxylate transport system expressed in neurons as NaC2/NaCT, which can transport the tricarboxylate citrate as well as dicarboxylates such as succinate, alpha-ketoglutarate, and malate. 21803289_Our studies demonstrate a profound effect of mIndy on mammalian energy metabolism. 28406943_The findings revealed the potentially important role of citrate and Slc13a5 in the development and function of teeth and bone. 32682952_Disruption of the sodium-dependent citrate transporter SLC13A5 in mice causes alterations in brain citrate levels and neuronal network excitability in the hippocampus. 32808644_Nervous System Deletion of Mammalian INDY in Mice Mimics Dietary Restriction-Induced Memory Enhancement. 33491666_The longevity gene mIndy (I'm Not Dead, Yet) affects blood pressure through sympathoadrenal mechanisms. 33544126_Consequences of NaCT/SLC13A5/mINDY deficiency: good versus evil, separated only by the blood-brain barrier. |
ENSG00000141485 |
SLC13A5 |
216.528065 |
0.2291415763 |
-2.125689 |
0.20190593 |
107.947927 |
0.00000000000000000000000027593345476564753328999710555922617291319454370428990070115900798755625977776162471855059266090393066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000153956717325653593540158574195529110955913658658166578627112474307592204780803513131104409694671630859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
82.2078993198204 |
11.5426319156754 |
358.764658345637 |
34.5738084152942 |
| ENSMUSG00000020866 |
12291 |
Cacna1g |
protein_coding |
Q5SUG2 |
|
Coiled coil;Membrane;Proteomics identification;Reference proteome;Repeat;Transmembrane;Transmembrane helix |
|
|
|
cell body [GO:0044297]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; membrane [GO:0016020]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; synapse [GO:0045202]; voltage-gated calcium channel complex [GO:0005891]; voltage-gated sodium channel complex [GO:0001518]; low voltage-gated calcium channel activity [GO:0008332]; scaffold protein binding [GO:0097110]; voltage-gated calcium channel activity [GO:0005245]; voltage-gated sodium channel activity [GO:0005248]; action potential [GO:0001508]; artery smooth muscle contraction [GO:0014824]; calcium ion import [GO:0070509]; calcium ion transmembrane transport [GO:0070588]; calcium ion transport into cytosol [GO:0060402]; chemical synaptic transmission [GO:0007268]; membrane depolarization during action potential [GO:0086010]; neuronal action potential [GO:0019228]; positive regulation of calcium ion-dependent exocytosis [GO:0045956]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; regulation of atrial cardiac muscle cell membrane depolarization [GO:0060371]; regulation of calcium ion transport [GO:0051924]; regulation of heart rate [GO:0002027]; regulation of membrane potential [GO:0042391]; response to nickel cation [GO:0010045] |
14526084_results indicate T-type Ca2+ channels underlie an antinociceptive mechanism operating in the thalamus and support the idea that burst firing plays a critical role in sensory gating in the thalamus 15175395_Alpha1G T-type Ca2+ channels play a critical role in the genesis of spontaneous absence seizures resulting from hypofunctioning P/Q-type channels, but the augmentation of thalamic T-type Ca2+ currents is not essential in the genesis of absence seizures 15601764_the alpha1(G)-subunit of T-type calcium channels plays a critical role in the genesis of thalamocortical oscillations and contributes to the modulation of sleep states and the transition between NREM sleep and wake states 15677322_Sleep was not disturbed when Cav3.1 was deleted from cortical pyramidal neurons(Cav3.1 calcium channel) 16282520_Cacnalpha(1G) protects cells from Cd(2+) exposure by limiting Cd(2+) uptake. 16690884_CaV3.1/T-type Ca2+ channels contribute to sinoatrial node pacemaker activity and atrioventricular conduction. 16883604_These results suggest that muscle T-channel downregulation by TGF-beta1 and BMP-2 may be mediated by reduced transcription rather than through post-transcriptional modifications of CaV3.1 channels. 18414012_These results reveal the importance of alpha(1G) T-type Ca2+ channels in the development of neuropathic pain. 18753322_we have isolated and sequenced the full-length complementary DNA of a distinct isoform of Cav3.1 in the mouse inner ear 19480703_Six novel alternative splice sites within the coding region of the Cacna1g gene were identified. 19657020_Our data identify a new fast calcium signaling pathway in Purkinje cell dendritic spines triggered by short burst of parallel fiber inputs and mediated by Ca(v)3.1 T-type calcium channels and mGluR1s. 19920353_alpha1G transgenic mice showed no cardiac pathology despite large increases in Ca2+ influx, and they were even partially resistant to pressure overload-, isoproterenol-, and exercise-induced cardiac hypertrophy 20371816_Expression of Cacna1g is high in the thalamus and is activated by Lef1 and beta-catenin. 20435690_Results reveal an intracellular WPb-independent P-selectin pool in pulmonary capillary endothelium, where the regulated P-selectin surface expression is triggered by Ca(2+) transients evoked through activation of the alpha(1G) T-type channel. 20457833_The Ca(V)3.1 T-type Ca(2+) channel encoded by alpha(1G) subtype is the dominant Ca(V) in mouse preadipocytes and may play a role in regulating preadipocyte proliferation, a key step in adipose tissue development. 20547676_Suggest that both Cav2.1 P/Q- and Cav3.1 T-type calcium channels are required for the dynamic control of neuronal oscillations in the inferior olive neurons. 20682748_Results indicate that alpha1G T-type channels are critical for the opioidergic descending analgesia system in the PAG. 20827487_We hypothesize that cysteines in the large extracellular loop of Ca(V)3.1 form bridges within the loop and/or neighboring channel segments that are essential for channel gating. 21079360_Cav3.2 may contribute to the development of the outflow tract from the secondary heart field in the embryonic heart, whereas Cav3.1 may be involved in the development of the cardiac conduction-system together with Cav3.2. 21621520_this study suggests that the CaV3.1 T-type Ca2+ channel plays a role in modulating motor function under pathological condition. 21622683_Elimination of Cav3.1 expression leads to impaired cardiac function and enhanced arrhythmia vulnerability post-myocardial infarction. 22886848_Ca(v)3.1 is required for vascular smooth muscle proliferation during neointimal formation, and blocking of Ca(v)3.1 may be beneficial for preventing restenosis. 22933804_Activation of Cav3.1 T-type channel subunit during wake-like states is a major determinant for single and multiple spike occurrence during tonic firing and for the robustness of the thalamocortical transfer of sensory inputs. 23356724_CaV 3.1 channels are important for the myogenic tone at low arterial pressure, which is potentially relevant under resting conditions in vivo mice. 23868804_This study reported on the cloning and characterization of a proximal promoter region and initiated the analysis of transcription factors that control CaV 3.1 channel expression using the murine Cacna1g gene as a model. 23884198_Suggest that T-type Ca(2+) channels play an important role in infranodal escape automaticity. 24285899_Ca(v)3.1-dependent synaptic depression at thalamocortical projections contributes to mechanisms of forward suppression in the auditory cortex 25825486_CaV3.1 and CaV3.2 are substrates for EHD3-dependent protein trafficking in heart 26111648_Results suggest that alpha1G T-type calcium channel plays a modulatory role in the duration and frequency of hippocampal seizures as well as the epileptogenicity of kainic acid-induced temporal lobe epilepsy in mice, mostly during acute periods 26390982_GluA4 subunits are required to produce an EPSC-triggerable postsynaptic action potentials after the presynaptic action potential, while Cav3.1 expression is needed to establish the driver function of L5B-POm synapses at hyperpolarized membrane potentials 26858455_Cross-frequency coupling between low-frequency and gamma rhythms was pronounced in wild-type but not in CaV3.1 knockouts, suggesting that the presence of CaV3.1 channels is a key element in the pathophysiology of trigeminal neuropathic pain. 27037192_Mice deficient for CaV3.1 were resistant to the induction of experimental autoimmune encephalomyelitis and had reduced productions of the granulocyte-macrophage colony-stimulating factor by central nervous system-infiltrating Th1 and Th17 cells. 27112236_These results provide support for Cacna1g as an epilepsy modifier gene and suggest that modulation of Cav3.1 may be an effective therapeutic strategy. 27208094_Localized Cav3.1 knockdown in the medial septum selectively enhanced object exploration, whereas the null mutant (KO) mice showed enhanced-object exploration as well as open-field exploration. 27353765_Membrane-protein extraction and use of an intracellular protein-transport inhibitor showed that GDF-15 promoted CaV3.1 and CaV3.3 alpha-subunit expression by trafficking to the membrane. 28438566_Data, including data from studies using knockout mice, suggest that acetylcholine- (Ach-)induced vasorelaxation/vasodilatation of intrapulmonary arteries is reduced in pulmonary hypertension, but is still dependent on Cav3.1 activity; thus, Cav3.1 contributes to intrapulmonary vascular reactivity by controlling calcium signaling in arterial endothelium and Ach-induced vasorelaxation/vasodilatation. 28806761_Cacna1g exclusively expressed in serosal PDGFRalpha+ cells is a new pathological marker for gastrointestinal diseases. 29082441_Cav3.1 expression was significantly reduced in aortae from aged compared to young WT mice. The level of phosphorylated eNOS was significantly increased in aortae from aged Cav3.1-/- mice. Cav3.1-deficient mice develop less age-dependent endothelial dysfunction. 29274077_Loss of Cav3.1 makes the beating rates of the heart in vivo and perfused hearts ex vivo, as well as sinoatrial node cells, less sensitive to beta-adrenergic stimulation. 29578032_Results confirm the idea that T-channels critically support rebound burst firing following periods of membrane hyperpolarization, similar to those that occur during inhibitory synaptic potentials in the ventral tegmental area. Behavioral results indicate that deletion of CaV3.1 channels disrupts the hyperlocomotion. 29913145_FMOD affected the expressions of the Cav1.1 and Cav3.1 genes. FMOD regulates calcium channel activity. The mRNA expressions of Cav1.1 and Cav3.1 increased during muscle regeneration. 30715245_Alterations in Oscillatory Behavior of Central Medial Thalamic Neurons Demonstrate a Key Role of CaV3.1 Isoform of T-Channels During Isoflurane-Induced Anesthesia. 31042060_T-type channels Cav3.1and Cav3.2 do not contribute to baseline blood pressure levels and ANG II-induced hypertension. Cav3.1, but not Cav3.2, contributes to aldosterone secretion. Aldosterone promotes cardiac hypertrophy during hypertension. 31800012_Adult loss of Cacna1a in mice recapitulates childhood absence epilepsy by distinct thalamic bursting mechanisms. 33144668_Concomitant genetic ablation of L-type Cav1.3 (alpha1D) and T-type Cav3.1 (alpha1G) Ca(2+) channels disrupts heart automaticity. 33547341_Homeostatic plasticity and burst activity are mediated by hyperpolarization-activated cation currents and T-type calcium channels in neuronal cultures. 34403092_Downregulation of Cav3.1 T-type Calcium Channel Expression in Age-related Hearing Loss Model. |
ENSG00000006283 |
CACNA1G |
2940.002177 |
2.0742343332 |
1.052579 |
0.07373792 |
200.156526 |
0.00000000000000000000000000000000000000000000193052113654169189543905364939259204542535519388954132292071723663392732639836198760866405968127258294007597671235795105948440664178633596748113632202148437500000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000002748196070261967475405365179779620757447782136667118417284703348000952521843519111800098779490929775359967836018820763754888503171969205141067504882812500000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3948.81365745299 |
201.916602947812 |
1903.74520093216 |
73.7548650988278 |
| ENSMUSG00000020875 |
15417 |
Hoxb9 |
protein_coding |
P20615 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:15417; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; RNA polymerase II transcription regulator complex [GO:0090575]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; anterior/posterior pattern specification [GO:0009952]; cell chemotaxis [GO:0060326]; embryonic skeletal system development [GO:0048706]; embryonic skeletal system morphogenesis [GO:0048704]; mammary gland development [GO:0030879]; positive regulation of transcription by RNA polymerase II [GO:0045944]; proximal/distal pattern formation [GO:0009954]; regulation of transcription by RNA polymerase II [GO:0006357] |
30314948_HOXB9 can suppress both phosphorylated-Akt expression and NF-kappaB activity. The latter phenomenon affects Snail protein expression and the inhibition of gastric carcinoma proliferation. 31504243_Homeobox B9 integrates bone morphogenic protein 4 with inflammation at atheroprone sites. 33214683_HOX genes promote cell proliferation and are potential therapeutic targets in adrenocortical tumours. 34508900_WT1 regulates HOXB9 gene expression in a bidirectional way. |
ENSG00000170689 |
HOXB9 |
31.417100 |
0.0291333672 |
-5.101184 |
0.82032579 |
48.376991 |
0.00000000000351670285309347795725156991155395315874071515693799483415205031633377075195312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000655821764797064233639659359343851950663317751377690001390874385833740234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.69860828622458 |
1.01873238553575 |
58.3045644023245 |
16.4044868040519 |
| ENSMUSG00000020897 |
20877 |
Aurkb |
protein_coding |
O70126 |
FUNCTION: Serine/threonine-protein kinase component of the chromosomal passenger complex (CPC), a complex that acts as a key regulator of mitosis (By similarity). The CPC complex has essential functions at the centromere in ensuring correct chromosome alignment and segregation and is required for chromatin-induced microtubule stabilization and spindle assembly (By similarity). Involved in the bipolar attachment of spindle microtubules to kinetochores and is a key regulator for the onset of cytokinesis during mitosis (By similarity). Required for central/midzone spindle assembly and cleavage furrow formation (By similarity). Key component of the cytokinesis checkpoint, a process required to delay abscission to prevent both premature resolution of intercellular chromosome bridges and accumulation of DNA damage: phosphorylates CHMP4C, leading to retain abscission-competent VPS4 (VPS4A and/or VPS4B) at the midbody ring until abscission checkpoint signaling is terminated at late cytokinesis (By similarity). AURKB phosphorylates the CPC complex subunits BIRC5/survivin, CDCA8/borealin and INCENP (By similarity). Phosphorylation of INCENP leads to increased AURKB activity (By similarity). Other known AURKB substrates involved in centromeric functions and mitosis are CENPA, DES/desmin, GPAF, KIF2C, NSUN2, RACGAP1, SEPTIN1, VIM/vimentin, HASPIN, and histone H3 (By similarity). A positive feedback loop involving HASPIN and AURKB contributes to localization of CPC to centromeres (By similarity). Phosphorylation of VIM controls vimentin filament segregation in cytokinetic process, whereas histone H3 is phosphorylated at 'Ser-10' and 'Ser-28' during mitosis (H3S10ph and H3S28ph, respectively) (PubMed:11784863). A positive feedback between HASPIN and AURKB contributes to CPC localization (By similarity). AURKB is also required for kinetochore localization of BUB1 and SGO1 (By similarity). Phosphorylation of p53/TP53 negatively regulates its transcriptional activity (By similarity). Key regulator of active promoters in resting B- and T-lymphocytes: acts by mediating phosphorylation of H3S28ph at active promoters in resting B-cells, inhibiting RNF2/RING1B-mediated ubiquitination of histone H2A and enhancing binding and activity of the USP16 deubiquitinase at transcribed genes (PubMed:24034696). Acts as an inhibitor of CGAS during mitosis: catalyzes phosphorylation of the N-terminus of CGAS during the G2-M transition, blocking CGAS liquid phase separation and activation, and thereby preventing CGAS-induced autoimmunity (By similarity). Phosphorylates KRT5 during anaphase and telophase (PubMed:29518391). {ECO:0000250|UniProtKB:Q96GD4, ECO:0000269|PubMed:11784863, ECO:0000269|PubMed:24034696, ECO:0000269|PubMed:29518391}. |
Acetylation;ATP-binding;Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Cytoskeleton;Kinase;Kinetochore;Mitosis;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase;Ubl conjugation |
|
This gene encodes a member of the aurora kinase subfamily of serine/threonine kinases. The genes encoding the other two members of this subfamily are located on chromosomes 2 and 7. These kinases participate in the regulation of alignment and segregation of chromosomes during mitosis and meiosis through association with microtubules. [provided by RefSeq, Sep 2015]. |
mmu:20877; |
chromocenter [GO:0010369]; chromosome [GO:0005694]; chromosome passenger complex [GO:0032133]; chromosome, centromeric region [GO:0000775]; condensed chromosome, centromeric region [GO:0000779]; cytoplasm [GO:0005737]; kinetochore [GO:0000776]; microtubule cytoskeleton [GO:0015630]; midbody [GO:0030496]; mitotic spindle midzone [GO:1990023]; mitotic spindle pole [GO:0097431]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; spindle microtubule [GO:0005876]; spindle midzone [GO:0051233]; spindle pole centrosome [GO:0031616]; ATP binding [GO:0005524]; histone H3S28 kinase activity [GO:0044022]; histone serine kinase activity [GO:0035174]; kinase binding [GO:0019900]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; abscission [GO:0009838]; cellular response to UV [GO:0034644]; cleavage furrow formation [GO:0036089]; mitotic cell cycle [GO:0000278]; mitotic cytokinesis [GO:0000281]; mitotic cytokinesis checkpoint signaling [GO:0044878]; mitotic spindle midzone assembly [GO:0051256]; mitotic spindle organization [GO:0007052]; negative regulation of B cell apoptotic process [GO:0002903]; negative regulation of cytokinesis [GO:0032466]; negative regulation of protein binding [GO:0032091]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of attachment of mitotic spindle microtubules to kinetochore [GO:1902425]; positive regulation of cytokinesis [GO:0032467]; positive regulation of lateral attachment of mitotic spindle microtubules to kinetochore [GO:1905116]; positive regulation of mitotic cell cycle spindle assembly checkpoint [GO:0090267]; positive regulation of mitotic cytokinesis [GO:1903490]; positive regulation of mitotic sister chromatid segregation [GO:0062033]; positive regulation of mitotic sister chromatid separation [GO:1901970]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of telomerase activity [GO:0051973]; positive regulation of telomere capping [GO:1904355]; positive regulation of telomere maintenance via telomerase [GO:0032212]; post-translational protein modification [GO:0043687]; protein localization to kinetochore [GO:0034501]; protein phosphorylation [GO:0006468]; regulation of cytokinesis [GO:0032465]; spindle organization [GO:0007051] |
12446714_downregulation in response to genotoxic stress and interaction of E2F 12584241_in spermatogonia, aurora-B first appears at chromocentres in late diplotene before the initial phosphorylation of histone H3. 14751927_found normal expression and localization of Aurora-B during prophase in polyploidizing bone marrow megakaryocytes. At late anaphase, however, Aurora-B/AIM-1 is absent or mislocalized. 15130946_results suggest that endomitotic MKs appropriately express functional Aurora-B kinase and related proteins in early anaphase, making a simple deficiency of this protein an unlikely explanation for polyploidy in this cell type. 16386730_INCENP recruits Aurora-C (or some other factor(s) recruit INCENP and Aurora-C) to meiotic chromosomes, while Aurora-C may either work alone or cooperate with Aurora-B to regulate chromosome segregation during male meiosis 16741559_the perikinetochoric rings of MCAK and Aurora-B define a novel transient centromere domain at least in mouse chromosomes during meiosis. 17192404_Aurora-B and Aurora-C serve specialized functions in mammalian spermatogenesis. 17948062_Aurora B kinase has a role in marking silent chromatin independently of the cell cycle, and targeting of Aurora B-mediated phosphorylation of H3 S10 to repressed genes could be a mechanism for epigenetic silencing of gene expression. 18353803_AURK may be an oocyte histone-H3 kinase capable of regulating chromatin remodeling throughout oocyte meiosis. 19332642_sustained expression of Aurora-B induces tetraploidy, which, in turn, facilitates genomic instability and tumor development in a xenograft model 19565641_Results show over-expression of AURKB, but not AURKA or AURKC, rescues the chromosome alignment defect suggesting that AURKB is the primary Aurora kinase responsible for regulating chromosome dynamics during meiosis in mouse oocytes. 19778480_Changes in activity of AURKB may increase risks for chromosome non-disjunction and aneuploidy in mammalian oocytes. 21613325_Data show that aurora B is dispensable for CPC function during early cell divisions and aurora B-null embryos are normally implanted. 21646403_Results establish that Bub1 has oncogenic properties and suggest that Aurora B is a critical target through which overexpressed Bub1 drives aneuploidization and tumorigenesis. 22314491_Results indicated that the activity of AURKB was required for regulating multiple stages of mitotic progression in the early development of mouse zygotes and was correlated with the activation of the MAPK pathway. 22767597_the BRAF/ERK axis controls Aurora B expression at the transcriptional level, likely through the transcription factor FOXM1. 22778418_These findings suggest a model for the presence of AURKC in oocytes: that AURKC compensates for loss of AURKB through differences in both message recruitment and protein stability. 22858681_These results provide a scientific rationale for investigating ABK inhibitors as a treatment for intestinal cancer 23370391_Calmodulin protects Aurora B on the midbody to regulate the fidelity of cytokinesis. 23428904_Aurora B represses p21(Cip1), preventing delayed DNA replication, Cdk inhibition and premature mitotic exit. 23928698_FBXL2 mediates Aurora B ubiquitination and degradation within the midbody, which is sufficient to induce mitotic arrest and apoptosis. 23948252_Inhibitions of Aurora B and Cyclin-dependent kinase 1 activity in vertebrate cells also have opposite effects on the timing of abscission. 24034696_Aurora B and Ring1B co-occupy active promoters in resting lymphocytes. 24141881_reduced accumulation of Aurora B at the inner centromere region in cells lacking Pds5B impairs its error correction function, promoting chromosome mis-segregation and aneuploidy 24586209_Using this mutant we show for the first time that AURKC has functions that do not overlap with AURKB. These functions include regulating localized CPC activity and regulating chromosome alignment and K-MT attachments at metaphase of meiosis I (Met I). 25057018_Chromsome stability is on of the tumor-suppressive functions of ARF as well as regulation of Aurora B expression in tumors. 26240282_Overexpression of Aurora B also results in a reduced DNA damage response and decreased levels of the p53 target p21(Cip1) in vitro and in vivo. 26880562_Aurkb phosphorylates Oct4(S229) during G2/M phase, leading to the dissociation of Oct4 from chromatin, whereas PP1 binds Oct4 and dephosphorylates Oct4(S229) during M/G1 transition, which resets Oct4-driven transcription for pluripotency and the cell cycle. 27389782_The results propose a model in which Aurora B-mediated H2AX-phosphorylated serine 121 probably provide a platform for Aurora B autoactivation circuitry at centromeres and thus play a pivotal role in proper chromosome segregation. 27849008_Data show that phosphorylated Ku70 S155 interacts with the Aurora B kinase and leads to inhibition of its activity. 28359584_The high sequence similarity among the AURK family members has made discerning the individual kinase functions in meiosis challenging. Technical limitations in specifically targeting AURKB or AURKC using small-molecule inhibitors and compensatory abilities in single-knockout animals add to this challenge...proper regulation of AURKA expression is crucial for spindle formation in meiosis 29040668_Loss of AURKB function affects TERF1 telomere binding and results in aberrant telomere structure. 30415701_in the absence of AURKC, AURKA localizes to chromosomes in a CPC-dependent manner. These data suggest that AURKC prevents AURKA from localizing to chromosomes by competing for CPC binding. This competition is important for adequate spindle length to support meiosis I. There is also a unique requirement for AURKB to negatively regulate AURKC to prevent aneuploidy. 31264311_Aurora B causes mitotic arrest and participates in spindle assembly checkpoint via Mad2 and H3S10P, which is required for self-correction of aneuploidies 31383943_Salivary glands require Aurora Kinase B for regeneration after transient innate immune-mediated injury. 33172986_Aurora B and C kinases regulate chromosome desynapsis and segregation during mouse and human spermatogenesis. 33854138_High-throughput kinase inhibitor screening reveals roles for Aurora and Nuak kinases in neurite initiation and dendritic branching. 34047289_Adhesion-growth factor crosstalk regulates AURKB activation and ERK signalling in re-adherent fibroblasts. 34121118_A functional genetic screen identifies aurora kinase b as an essential regulator of Sox9-positive mouse embryonic lung progenitor cells. 34613789_CNOT3 interacts with the Aurora B and MAPK/ERK kinases to promote survival of differentiating mesendodermal progenitor cells. 34636397_Aurora kinase B inhibits aurora kinase A to control maternal mRNA translation in mouse oocytes. 34704342_Age-dependent integrity of the meiotic spindle assembly checkpoint in females requires Aurora kinase B. 35694956_Haspin participates in AURKB recruitment to centromeres and contributes to chromosome congression in male mouse meiosis. |
ENSG00000178999 |
AURKB |
953.101452 |
2.0659655541 |
1.046816 |
0.10237460 |
103.357680 |
0.00000000000000000000000279787892686498499054284013328427347840249699787542243133611793277047696815174049334018491208553314208984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000001474646775571191909351678556251739391045375077830883685661245239423289277169715205673128366470336914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1294.61763902314 |
91.944045110909 |
626.64047637593 |
34.0424407470508 |
| ENSMUSG00000020961 |
108800 |
Ston2 |
protein_coding |
Q8BZ60 |
FUNCTION: Adapter protein involved in endocytic machinery. Involved in the synaptic vesicle recycling. May facilitate clathrin-coated vesicle uncoating (By similarity). {ECO:0000250}. |
Cytoplasm;Endocytosis;Membrane;Phosphoprotein;Reference proteome;Repeat;Synapse;Synaptosome;Ubl conjugation |
|
|
mmu:108800; |
clathrin-coated vesicle [GO:0030136]; cytoplasmic vesicle [GO:0031410]; cytosol [GO:0005829]; membrane [GO:0016020]; neuron projection [GO:0043005]; nucleolus [GO:0005730]; presynapse [GO:0098793]; synaptic vesicle [GO:0008021]; clathrin adaptor activity [GO:0035615]; chemical synaptic transmission [GO:0007268]; hematopoietic progenitor cell differentiation [GO:0002244]; regulation of endocytosis [GO:0030100]; synaptic vesicle endocytosis [GO:0048488]; vesicle-mediated transport [GO:0016192] |
23345427_Loss of Stn2 leads to selective missorting of synaptotagmin 1 to the neuronal surface, an elevated synaptic vesicle (SV) pool size, and accelerated SV protein endocytosis. |
ENSG00000140022 |
STON2 |
102.726957 |
0.3896557283 |
-1.359728 |
0.50620076 |
6.997886 |
0.00816060446988424101344428152060572756454348564147949218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0282102569809912599729795346092942054383456707000732421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
60.1301541890288 |
21.5335358665883 |
154.316104750887 |
40.5885943413711 |
| ENSMUSG00000021125 |
11847 |
Arg2 |
protein_coding |
O08691 |
FUNCTION: May play a role in the regulation of extra-urea cycle arginine metabolism and also in down-regulation of nitric oxide synthesis. Extrahepatic arginase functions to regulate L-arginine bioavailability to nitric oxid synthase (NOS). Arginine metabolism is a critical regulator of innate and adaptive immune responses. Seems to be involved in negative regulation of the survival capacity of activated CD4(+) and CD8(+) T cells (PubMed:27745970, PubMed:25009204). May suppress inflammation-related signaling in asthmatic airway epithelium (PubMed:27214549). May contribute to the immune evasion of H.pylori by restricting M1 macrophage activation and polyamine metabolism (PubMed:27074721). May play a role in promoting prenatal immune suppression (By similarity). Regulates RPS6KB1 signaling, which promotes endothelial cell senescence and inflammation and implicates NOS3/eNOS dysfunction (PubMed:22928666). Can inhibit endothelial autophagy independently of its enzymatic activity implicating mTORC2 signaling (PubMed:25484082). Involved in vascular smooth muscle cell senescence and apoptosis independently of its enzymatic activity (By similarity). {ECO:0000250|UniProtKB:P78540, ECO:0000269|PubMed:22928666, ECO:0000269|PubMed:25009204, ECO:0000269|PubMed:25484082, ECO:0000269|PubMed:27074721, ECO:0000269|PubMed:27214549, ECO:0000269|PubMed:27745970}. |
Adaptive immunity;Arginine metabolism;Direct protein sequencing;Hydrolase;Immunity;Innate immunity;Manganese;Metal-binding;Mitochondrion;Reference proteome;Transit peptide;Urea cycle |
PATHWAY: Nitrogen metabolism; urea cycle; L-ornithine and urea from L-arginine: step 1/1. {ECO:0000250|UniProtKB:P05089}. |
|
mmu:11847; |
cytoplasm [GO:0005737]; mitochondrion [GO:0005739]; arginase activity [GO:0004053]; manganese ion binding [GO:0030145]; nitric-oxide synthase binding [GO:0050998]; adaptive immune response [GO:0002250]; arginine catabolic process to ornithine [GO:0019547]; arginine metabolic process [GO:0006525]; innate immune response [GO:0045087]; negative regulation of activated CD8-positive, alpha-beta T cell apoptotic process [GO:1905403]; negative regulation of CD4-positive, alpha-beta T cell proliferation [GO:2000562]; negative regulation of chemokine (C-C motif) ligand 4 production [GO:0071644]; negative regulation of chemokine (C-C motif) ligand 5 production [GO:0071650]; negative regulation of defense response to bacterium [GO:1900425]; negative regulation of interleukin-13 production [GO:0032696]; negative regulation of interleukin-17 production [GO:0032700]; negative regulation of macrophage inflammatory protein 1 alpha production [GO:0071641]; negative regulation of nitric-oxide synthase activity [GO:0051001]; negative regulation of striated muscle contraction [GO:0045988]; negative regulation of tumor necrosis factor production [GO:0032720]; negative regulation of type 2 immune response [GO:0002829]; positive regulation of cellular senescence [GO:2000774]; regulation of interleukin-1 beta production [GO:0032651]; regulation of L-arginine import across plasma membrane [GO:1905541]; regulation of reactive oxygen species biosynthetic process [GO:1903426]; striated muscle contraction [GO:0006941]; urea cycle [GO:0000050]; ureteric bud development [GO:0001657] |
12049781_Arginase II expression in mouse embryonic development 12679322_induced in bleomycin-induced pulmonary fibrosis in mice. 12813022_Gene expression of arginase I and II in pathogenesis of asthma. 14675208_Upon injury, arginase-1 and -2 mRNA, protein, and activity were strongly induced reaching a maximum between day 3 and day 7 postwounding in diabetic mice 15128819_Arginase II mRNA levels and enzyme activity in macrophages from Trypanosoma brucei brucei-infected mice are higher in susceptible BALB/c mice compared with resistant C57BL/6 mice. 15539552_Variations of the levels of OAT protein and arginase II mRNA and protein were strongly correlated with testosteronemia. 15616821_AII was expressed in the cortex and the outer stripe of outer medulla, urea was produced mainly in the proximal tubules, the 38-kDa AII protein was more abundant in the mitochondria than the cytosol, and AII was three-fold higher in females than males 16794538_upregulation of arginase activity by Th2 cytokines during xenograft rejection limits the bioavailability of L-arginine for the inducible nitric oxide synthase 16943198_Arg2 is an anti-inflammatory target of liver X receptor in macrophages. 17442735_Neither the overexpression nor the deficiency of MKP-1 had any substantial effect on the expression of the arginases. 17686999_Results suggest that endogenous arginase-derived ornithine may not directly contribute to polyamine homeostasis. 17827260_Endothelial cell mitochondrial arginase II regulates NO production, vascular endothelial function, and vascular stiffness by modulating NOS-3 activity. 17890324_These results suggested that an increase in arginase may lead to decreased availability of arginine for nitric oxide synthase and may contribute to the remodeling of the lung. 18309100_arginase II plays a critical role in the pathophysiology of cholesterol-mediated endothelial dysfunction 18663728_Lack of arginase II in the TRAMP model seems to speed up the tumor progression process leading to a more aadvanced cancer state in these mice. 19027033_NOS2 knockout mice are more sensitive to ovalbumin-induced airway inflammation and these findings are strongly correlated with increased expression of arginase I/II. 19590038_these data provide the first evidence that arginase is involved in the pathogenesis of an ocular disease. 20097867_Studies implicate Arg2 in the immune evasion of H. pylori by causing intracellular depletion of l-arginine and thus reduction of NO-dependent bactericidal activity. 20372170_arginase II deficiency leads to a better CBF recovery after TBI and implicates arginase II in hemodynamic processes. 20978229_Data suggest that arginase II present in the small intestinal mucosa has the role of providing ornithine for citrulline synthesis. 21130456_Delineate a clearer path from OxLDL through the endothelial cell LOX-1 receptor, RhoA, and ROCK, to the activation of arginase II, downregulation of NO, and vascular dysfunction in atherosclerosis. 21151916_a high fat diet affects arginase activities and global arginine bioavailability in wild-type and ApoE-deficient mice 21296975_Helicobacter infection induces Arg2 (but not Arg1) in gastric lamina propria macrophages. 21811615_data implicate Arg2 in neurodegeneration during retinopathy of prematurity; deletion significantly improves neuronal survival and function, possibly through the regulation of mitochondrial membrane permeability mediated apoptosis during retinal ischemia 21876618_Data suggest that the in vivo source for the essential urea utilized by H. pylori urease is neither bacterial arginase nor host arginase II; instead, either residual host arginase I or agmatinase is probably responsible. 21926276_In diabetes models, kidney Arg2 increases as diabetes develops (but not in diabetic Arg2-deficient mice). Increase in kidney arginase activity is associated with loss of renal function; such function loss is not seen in diabetic Arg2-deficient mice. 22288373_inhibition of Arg2 increases L-arginine levels, diminishes ROS production and ameliorates nitrate tolerance 22854495_Arginase II is upregulated in aortic vessels of aged mice and contributes to decreased nitric oxide generation and increased reactive oxygen species production via endothelial nitric oxide synthase uncoupling. 22928666_A novel mechanism has been uncovered for mutual positive regulation between S6K1 and Arg-II in endothelial inflammation and aging. 23125252_IL-13 contributes to the development of pulmonary hypertension via an IL-13receptor alpha2-arginase 2-dependent pathway. 23443634_Studied thoracic aorta from ApoE-/- mice, and found the high-cholesterol diet (HCD) induced an increase in arginase activity, a decrease in NO release and an increase in reactive oxygen species generation that was reversed by treatment with PG. 23566117_ROCK 2, p38 MAPK and arginase play key roles in diabetes-induced impairment of corpora cavernosa relaxation. 23760286_Arginase inhibition mediates renal tissue protection in diabetic nephropathy by an eNOS-dependent mechanism and has an eNOS-independent effect on kidney macrophage recruitment. 23832324_Arg-II promotes mitochondrial dysfunction leading to VSMC senescence/apoptosis through complex positive crosstalk among S6K1-JNK, ERK, p66Shc, and p53, contributing to atherosclerotic vulnerability phenotype. 24556690_Suggest that retinal arginase is involved in the hyperoxia-induced neuronal degeneration in the OIR model, through the regulation of polyamine metabolism. 24833798_HDAC2 is a critical regulator of Arg2 expression and thereby endothelial nitric oxide and endothelial function. 24903103_OxLDL triggers retrograde translocation of arginase2 in aortic endothelial cells via ROCK and mitochondrial processing peptidase. 25009204_miR155-induced repression of Arg2 expression is critical for the ability of dendritic cells to drive T cell activation by controlling arginine availability in the extracellular environment. 25034973_high fat diet enhanced arginase-II expression/activity and p38mapk activity, which was associated with eNOS-uncoupling as revealed by decreased nitric oxide 25205225_Arg1 expression is decreased, and Arg2 expression is increased in the newborn congenital obstructive nephropathy and in the mouse model. 25375125_Arginase 2 deletion prevents hyperoxia-induced retinal vascular injury by preventing NOS uncoupling resulting in decreased reactive oxygen species formation and increased nitric oxide bioavailability. 25451938_ERK2 and p38 regulate arginase II induction in LPS-stimulated macrophages, but iNOS induction by LPS is dependent on p38 activation 25484082_arginase 2 impairs endothelial autophagy independently of the L-arginine ureahydrolase activity through activation of RPS6KB1 and inhibition of PRKAA, which is implicated in atherogenesis 25635535_Arg-II, p38, and S6K1 form a positive circuit which regulates endothelial senescence and cardiovascular aging. 27074721_the finding of this study is that ARG2 contributes to the immune evasion of H. pylori by restricting M1 macrophage activation and polyamine metabolism. 27406916_The possibility that the Gas6-Mer-PI3K/Akt-STAT1-LXR-Arg2 pathway plays an essential role for resolving inflammatory response in acute lung injury. 27757983_Arginase II expression is reduced in aortic endothelial cells and macrophages following PARP-1 knockout. 27882947_This study shows for the first time that neurovascular injury after retinal ischemia/reperfusion is mediated through increased expression of Arginase 2. 28356309_Arg2 KO female (not male) mice are protected from age-associated glucose intolerance and reveal greater glucose induced-insulin release, larger islet size and beta-cell mass, and more proliferative and less apoptotic beta-cells compared with the age-matched WT controls. Arg2 is mainly expressed in acinar cells and is upregulated with aging, which enhances p38 MAPK activation and release of TNF-alpha. 28446459_diabetes-dependent increase in renal arginase-2 expression also requires arginase-1 expression in macrophages 28808255_ARG2 which hydrolyzes arginine into ornithine and urea, is induced upon obesity, and silencing or loss of ARG2 markedly suppresses pancreatic ductal adenocarcinoma. 29266319_Arginase and alpha-smooth muscle actin induction after hyperoxic exposure in a mouse model of bronchopulmonary dysplasia 29472548_The senescent vascular smooth muscle cells showed elevated endogenous Arg-II and silencing Myo1b prevents Arg-II-mediated lysosomal positioning, dissociation of TSC from lysosome, mTORC1 activation and cell apoptosis. 29574762_Arg II was identified as a factor promoting EAE likely via an Arg I-independent mechanism; Arg II may promote EAE by enhancing dendritic cells production of Th17-differentiating cytokines; specific inhibition of Arg II could be a potential therapy for multiple sclerosis 29718707_in vivo and in vitro experiments demonstrate that Arg-II negatively regulates AQP2 and the urine-concentrating capability in kidneys via a mechanism that is not associated with the modulation of the cAMP pathway. 30371203_These data demonstrate a novel function of arginase II in regulation of Ca(2+)-dependent eNOS phosphorylation. This novel mechanism drives arginase activation, mitochondrial dysfunction, endothelial dysfunction, and atherogenesis 30962478_Arg2 is a critical effector of the hepatic glucose fasting response. 31155612_Arginase II activity-dependent production of spermine augments Ca(2+) transition from the cytosol to the mitochondria in a mitochondrial p32 protein -dependent manner and regulates CaMKII-dependent constriction in vascular smooth muscle cells. 31280447_The current study was undertaken to investigate the role of Arginase 2 in Multiple sclerosis-mediated retinal neuronal damage and degeneration. 31852848_Regulatory T cells use arginase 2 to enhance their metabolic fitness in tissues. 31996482_Arginine metabolic control of airway inflammation. 32072766_Renal tubular arginase-2 participates in the formation of the corticomedullary urea gradient and attenuates kidney damage in ischemia-reperfusion injury in mice. 32739205_Arginase 2 is a mediator of ischemia-reperfusion injury in the kidney through regulation of nitrosative stress. 33674584_Mitochondrial arginase-2 is essential for IL-10 metabolic reprogramming of inflammatory macrophages. 33735314_Deletion of arginase 2 attenuates neuroinflammation in an experimental model of optic neuritis. 34139341_Endothelial arginase 2 mediates retinal ischemia/reperfusion injury by inducing mitochondrial dysfunction. 36139052_Role of Arginase-II in Podocyte Injury under Hypoxic Conditions. |
ENSG00000081181 |
ARG2 |
7.192458 |
0.0575791877 |
-4.118309 |
1.41191784 |
8.652824 |
0.00326554853831064915634097012286929384572431445121765136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0128828892347961181108484396418134565465152263641357421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.593210881472108 |
0.677771543256199 |
10.3025225834558 |
6.42300492104281 |
| ENSMUSG00000021175 |
217946 |
Cdca7l |
protein_coding |
Q922M5 |
FUNCTION: Plays a role in transcriptional regulation as a repressor that inhibits monoamine oxidase A (MAOA) activity and gene expression by binding to the promoter. Plays an important oncogenic role in mediating the full transforming effect of MYC in medulloblastoma cells (By similarity). Involved in apoptotic signaling pathways; May act downstream of P38-kinase and BCL-2, but upstream of CASP3/caspase-3 as well as CCND1/cyclin D1 and E2F1. {ECO:0000250, ECO:0000269|PubMed:16829576}. |
Cytoplasm;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Repressor;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:217946; |
cytosol [GO:0005829]; fibrillar center [GO:0001650]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; positive regulation of cell population proliferation [GO:0008284]; regulation of DNA-templated transcription [GO:0006355] |
15994933_Mouse JPO2/RAM2 (Cdca7l) interacts with cMyc. |
ENSG00000164649 |
CDCA7L |
141.313617 |
2.4542695946 |
1.295294 |
0.21385639 |
36.455296 |
0.00000000156208132766113707490906654555074331569564094479574123397469520568847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000214550250522402546676366762688770162270657237968407571315765380859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
200.078394172454 |
29.7671461081648 |
81.5225819581801 |
9.49282507753936 |
| ENSMUSG00000021182 |
68339 |
Ccdc88c |
protein_coding |
Q6VGS5 |
FUNCTION: Required for activation of guanine nucleotide-binding proteins (G-proteins) during non-canonical Wnt signaling. Binds to ligand-activated Wnt receptor FZD7, displacing DVL1 from the FZD7 receptor and leading to inhibition of canonical Wnt signaling. Acts as a non-receptor guanine nucleotide exchange factor by also binding to guanine nucleotide-binding protein G(i) alpha (Gi-alpha) subunits, leading to their activation. Binding to Gi-alpha subunits displaces the beta and gamma subunits from the heterotrimeric G-protein complex, triggering non-canonical Wnt responses such as activation of RAC1 and PI3K-AKT signaling. Promotes apical constriction of cells via ARHGEF18. {ECO:0000250|UniProtKB:Q9P219}. |
Alternative splicing;Cell junction;Coiled coil;Cytoplasm;Guanine-nucleotide releasing factor;Phosphoprotein;Reference proteome;Wnt signaling pathway |
|
|
mmu:68339; |
anchoring junction [GO:0070161]; cell junction [GO:0030054]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; dynein light intermediate chain binding [GO:0051959]; frizzled binding [GO:0005109]; G-protein alpha-subunit binding [GO:0001965]; guanyl-nucleotide exchange factor activity [GO:0005085]; identical protein binding [GO:0042802]; microtubule binding [GO:0008017]; PDZ domain binding [GO:0030165]; protein self-association [GO:0043621]; apical constriction [GO:0003383]; cytoplasmic microtubule organization [GO:0031122]; cytoskeleton-dependent intracellular transport [GO:0030705]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; non-canonical Wnt signaling pathway [GO:0035567]; protein destabilization [GO:0031648]; regulation of protein phosphorylation [GO:0001932]; small GTPase mediated signal transduction [GO:0007264]; stress-activated protein kinase signaling cascade [GO:0031098] |
14750955_Identification of Daple, a Dvl-binding protein, that suppresses Wnt signalling pathway. 22643886_Findings indicate that Daple interacts with Dishevelled to direct the Dishevelled/protein kinase lambda protein complex to activate Rac, which in turn mediates the non-canonical Wnt signalling pathway required for cell migration. 29229865_results suggest that the primary cilium or an associated structure influences the domain of cell-intrinsic signals that shape the hair bundle. Daple is therefore essential to orient and pattern sensory hair bundles. 32805026_Wnts regulate planar cell polarity via heterotrimeric G protein and PI3K signaling. 33929515_Planar cell polarity induces local microtubule bundling for coordinated ciliary beating. 34642354_Daple deficiency causes hearing loss in adult mice by inducing defects in cochlear stereocilia and apical microtubules. |
ENSG00000015133 |
CCDC88C |
79.296454 |
0.4166753299 |
-1.263004 |
0.37844356 |
10.957014 |
0.00093249954204169249998707114102103332697879523038864135742187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0043450085727681428404700625378609402105212211608886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.6984692977483 |
11.5947685898579 |
104.874145798925 |
19.9208158524452 |
| ENSMUSG00000021213 |
27384 |
Akr1c13 |
protein_coding |
Q8VC28 |
FUNCTION: Catalyzes the dehydrogenation of 17-beta-hydroxysteroids. May also exhibit significant activity with a variety of cyclic and alicyclic alcohols. Uses both NAD and NADP, but the activity is much greater with NAD than with NADP (By similarity). {ECO:0000250}. |
3D-structure;Direct protein sequencing;NAD;NADP;Oxidoreductase;Reference proteome |
|
|
mmu:27384; |
cytosol [GO:0005829]; alditol:NADP+ 1-oxidoreductase activity [GO:0004032]; aldo-keto reductase (NADP) activity [GO:0004033]; androsterone dehydrogenase activity [GO:0047023]; chlordecone reductase activity [GO:0047743]; ketosteroid monooxygenase activity [GO:0047086]; oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor [GO:0016655]; retinal dehydrogenase activity [GO:0001758]; steroid dehydrogenase activity [GO:0016229]; steroid metabolic process [GO:0008202]; xenobiotic metabolic process [GO:0006805] |
17888864_analysis of rat and mouse NAD+-dependent 3alpha/17beta/20alpha-hydroxysteroid dehydrogenases. 28329705_Akr1c13 functions as a cytosolic sensor for bacterial cyclic dinucleotides, shaping inflammatory gene activation 29764944_Listeria monocytogenes secretion of c-di-AMP inhibits RECON ( Akr1c13) activity, drives augmented NF-kappaB activation and nitric oxide production, and ultimately enhances intercellular spread. |
|
|
11.420907 |
0.0713499626 |
-3.808944 |
1.06785746 |
12.445032 |
0.00041910504991655344111944891949406155617907643318176269531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0021414988662626440081571121964998383191414177417755126953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.72571483654818 |
1.39721718450119 |
24.1866256719426 |
11.5694075892235 |
| ENSMUSG00000021285 |
21981 |
Ppp1r13b |
protein_coding |
Q62415 |
FUNCTION: Regulator that plays a central role in regulation of apoptosis via its interaction with p53/TP53 (By similarity). Regulates TP53 by enhancing the DNA binding and transactivation function of TP53 on the promoters of proapoptotic genes in vivo (By similarity). {ECO:0000250, ECO:0000250|UniProtKB:Q96KQ4}. |
ANK repeat;Apoptosis;Cytoplasm;Methylation;Nucleus;Phosphoprotein;Reference proteome;Repeat;SH3 domain |
|
|
mmu:21981; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; p53 binding [GO:0002039]; intrinsic apoptotic signaling pathway by p53 class mediator [GO:0072332]; negative regulation of cell cycle [GO:0045786]; positive regulation of neuron death [GO:1901216] |
18304521_Aspp1 plays a crucial role in the initial assembly and function of lymphatic vessels during mouse development in a p53-independent manner. 23365256_Our study demonstrates a novel role for ASPP1 and ASPP2 in the death of retinal ganglion cells. 26119234_After genotoxic stress, Aspp1 promotes hematopoietic stem cell (HSC) cycling and induces p53-dependent apoptosis in cells with persistent DNA damage foci. Aspp1 also attenuates HSC self-renewal and accumulation of DNA damage in p53 null HSCs. 36656967_Interdependent Nuclear Co-Trafficking of ASPP1 and p53 Aggravates Cardiac Ischemia/Reperfusion Injury. |
ENSG00000088808 |
PPP1R13B |
34.082694 |
0.3267993054 |
-1.613523 |
0.44294177 |
13.162693 |
0.00028557819496981035230220857812355461646802723407745361328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0015273620315239295076176206933382673014421015977859497070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.5997512946669 |
4.86509686152557 |
47.7349585416995 |
10.3862671140817 |
| ENSMUSG00000021303 |
14706 |
Gng4 |
protein_coding |
P50153 |
FUNCTION: Guanine nucleotide-binding proteins (G proteins) are involved as a modulator or transducer in various transmembrane signaling systems. The beta and gamma chains are required for the GTPase activity, for replacement of GDP by GTP, and for G protein-effector interaction. {ECO:0000305}. |
Cell membrane;Lipoprotein;Membrane;Methylation;Prenylation;Reference proteome;Transducer |
|
This gene encodes the gamma subunit of the heterotrimeric G-proteins that are comprised of alpha, beta and gamma subunits. Upon activation by G protein-coupled receptors, the beta-gamma heterodimer dissociates from the alpha subunit to activate downstream signaling events. Alternate splicing results in multiple transcript variants. [provided by RefSeq, Dec 2014]. |
mmu:14706; |
heterotrimeric G-protein complex [GO:0005834]; G-protein beta-subunit binding [GO:0031681]; GTPase activity [GO:0003924]; G protein-coupled receptor signaling pathway [GO:0007186]; negative regulation of cell growth [GO:0030308] |
Human_homologues 29338302_Common genetic variation in GNG4 and KCNQ2 was associated with cognitive decline. In human brain tissue data sets, both GNG4 and KCNQ2 show higher expression in hippocampus relative to other brain regions. 33854208_G-protein subunit gamma-4 expression has potential for detection, prediction and therapeutic targeting in liver metastasis of gastric cancer. 34275892_Identifying GNG4 might play an important role in colorectal cancer TMB. 35456499_G-Protein Subunit Gamma 4 as a Potential Biomarker for Predicting the Response of Chemotherapy and Immunotherapy in Bladder Cancer. |
ENSG00000168243 |
GNG4 |
19.810137 |
0.0486037985 |
-4.362787 |
0.95997297 |
19.757984 |
0.00000878937753332045437315144659207177824100654106587171554565429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000657240051976127492459542334124478202284080907702445983886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.11476887185943 |
1.51333590636998 |
43.5103620885474 |
17.615231422149 |
| ENSMUSG00000021379 |
15904 |
Id4 |
protein_coding |
P41139 |
FUNCTION: Transcriptional regulator (lacking a basic DNA binding domain) which negatively regulates the basic helix-loop-helix (bHLH) transcription factors by forming heterodimers and inhibiting their DNA binding and transcriptional activity. Implicated in regulating a variety of cellular processes, including cellular growth, senescence, differentiation, apoptosis, angiogenesis, and neoplastic transformation. |
Nucleus;Reference proteome;Repressor;Transcription;Transcription regulation |
|
|
mmu:15904; |
cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein dimerization activity [GO:0046983]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; transcription regulator inhibitor activity [GO:0140416]; astrocyte differentiation [GO:0048708]; brain development [GO:0007420]; cell differentiation [GO:0030154]; cell population proliferation [GO:0008283]; central nervous system myelination [GO:0022010]; cerebral cortex neuron differentiation [GO:0021895]; circadian regulation of gene expression [GO:0032922]; circadian rhythm [GO:0007623]; fat cell differentiation [GO:0045444]; G1/S transition of mitotic cell cycle [GO:0000082]; hippocampus development [GO:0021766]; negative regulation of astrocyte differentiation [GO:0048712]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of oligodendrocyte differentiation [GO:0048715]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuroblast proliferation [GO:0007405]; neuron differentiation [GO:0030182]; oligodendrocyte differentiation [GO:0048709]; osteoblast differentiation [GO:0001649]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of gene expression [GO:0010628]; positive regulation of neuroblast proliferation [GO:0002052]; positive regulation of transcription by RNA polymerase II [GO:0045944]; prostate gland epithelium morphogenesis [GO:0060740]; prostate gland morphogenesis [GO:0060512]; prostate gland stromal morphogenesis [GO:0060741]; protein localization [GO:0008104]; seminal vesicle morphogenesis [GO:0061682] |
15469968_Id4 has a unique and complex function in regulating neural stem cell proliferation and differentiation 15882580_Id4 has a critical role in regulating neural precursor cell proliferation and differentiation 16682435_ID proteins (ID1, ID2, ID3 and ID4) were significantly increased in Mecp2-deficient Rett syndrome brain; ID genes are ideal targets for MeCP2 regulation of neuronal maturation that may explain the molecular pathogenesis of Rett syndrome 16862533_these data support the role of Id4 as oligodendrocyte differentiation inhibitor 19777566_ta indicate that Zik1 and Gja9 demonstrated cancer-specific aberrant DNA methylation, whereas, Cdkn2a/p16, Igfbp3, Mgmt, Id4, and Cxcr4 were methylated in both the AOM tumors and normal colon mucosa. 20460371_ID4 has a role in adipocyte differentiation and the implications of this regulation for adipose tissue formation 20628571_Id4 was identified as a key transcription factors enriched in the early phase of differentiation and affecting the differentiation of both osteoblasts and adipocyte cell types. 21098398_Study confirms the importance of the silencing of ID4 in murine and human CLL pathogenesis. 21145943_High expression levels of Id1-4 proteins may play important roles in regulating retinal progenitor cell proliferation and differentiation. 22041901_CpG-rich islands within the Id2 and Id4 genes were bound by PRMT5 and were hypomethylated in PRMT5-deficient cells, suggesting that PRMT5 plays a role in gene silencing during glial cell 22069192_ID4 promotes mammary gland development by suppressing p38MAPK activity. 23786676_Id4 regulates NKX3.1, Sox9 and PTEN. 25038044_Id4 is a novel regulator of estrogen signaling, where Id4 restrains ERalpha expression in the basal and luminal cellular compartments of the mammary gland and regulates estrogen biosynthesis in the ovary 25813983_ID4 marks a stem cell-enriched subset of the mammary basal cell population. ID4 maintains the mammary stem cell pool by suppressing key factors required for luminal differentiation. 26621350_results indicate that Id4 marks spermatogonial stem cells in the mouse testis. 28087628_ID4 has a role in stem cell or progenitor capacity in spermatogonia and dictates the interface of transition between the different functional states 30884976_Id4 is a transcriptional regulator critically involved in the differentiation of OPN-type osteoblasts. JNK-Id4 signaling axis is crucial in the control of OPN and OCN expression during osteoblastic differentiation. 31390563_Id4 Downstream of Notch2 Maintains Neural Stem Cell Quiescence in the Adult Hippocampus. 31550547_BMP9 prevents induction of osteopontin in JNK-inactivated osteoblasts via Hey1-Id4 interaction. 31552825_Id4 and other Id proteins promote elimination of Ascl1 protein in neural stem cells culture. Id4 sequesters Ascl1 heterodimerization partner E47, promoting Ascl1 protein degradation and stem cell quiescence. 34482420_DNA methylation regulates the expression of the negative transcriptional regulators ID2 and ID4 during OPC differentiation. 34502541_Targeted Disruption of the Inhibitor of DNA Binding 4 (Id4) Gene Alters Photic Entrainment of the Circadian Clock. 36263050_miR-210-3p protects against osteoarthritis through inhibiting subchondral angiogenesis by targeting the expression of TGFBR1 and ID4. 36535369_Id4 modulates salivary gland homeostasis and its expression is downregulated in IgG4-related disease via miR-486-5p. |
ENSG00000172201 |
ID4 |
403.641991 |
0.4140752821 |
-1.272035 |
0.13405023 |
89.125097 |
0.00000000000000000000370619317044380573136437142460096211508824473245782351877707777587644955019641201943159103393554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001538221645019074921590349718930117161662557826543468999722086287818001437699422240257263183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
235.784946654657 |
21.9398590983605 |
569.425311880525 |
37.8818754595022 |
| ENSMUSG00000021388 |
66695 |
Aspn |
protein_coding |
Q99MQ4 |
FUNCTION: Binds calcium and plays a role in osteoblast-driven collagen biomineralization activity (By similarity). Critical regulator of TGF-beta in articular cartilage and plays an essential role in cartilage homeostasis and osteoarthritis (OA) pathogenesis. Negatively regulates chondrogenesis in the articular cartilage by blocking the TGF-beta/receptor interaction on the cell surface and inhibiting the canonical TGF-beta/Smad signal. Negatively regulates periodontal ligament (PDL) differentiation and mineralization to ensure that the PDL is not ossified and to maintain homeostasis of the tooth-supporting system. Inhibits BMP2-induced cytodifferentiation of PDL cells by preventing its binding to BMPR1B/BMP type-1B receptor, resulting in inhibition of BMP-dependent activation of SMAD proteins. Inhibits the interaction between TGFB1 and TGF-beta receptor type II in the presence of heparin/heparan sulfate in vitro. {ECO:0000250, ECO:0000269|PubMed:17522060, ECO:0000269|PubMed:17827158, ECO:0000269|PubMed:18407830, ECO:0000269|PubMed:20052601}. |
Biomineralization;Calcium;Disulfide bond;Extracellular matrix;Glycoprotein;Leucine-rich repeat;Reference proteome;Repeat;Secreted;Signal |
|
This gene encodes a member of the small leucine-rich proteoglycan family. The encoded protein is an extracellular matrix protein that modulates the transforming growth factor-beta signaling pathway, regulating cartilage matrix gene expression and cartilage formation. The protein plays a role in the pathology of osteoarthritis. Alternative splicing results in multiple transcript variants.[provided by RefSeq, Feb 2010]. |
mmu:66695; |
cell projection [GO:0042995]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; calcium ion binding [GO:0005509]; bone mineralization [GO:0030282]; negative regulation of tooth mineralization [GO:0070171]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512]; response to fluoride [GO:1902617] |
17522060_PLAP-1/asporin plays a specific role in the periodontal ligament as a negative regulator of cytodifferentiation and mineralization by regulating BMP-2 activity to prevent the periodontal ligament from developing non-physiological mineralization 17804408_The findings indicate that TGF-beta1 induces ASPN through Smad3 but that this induction is indirect. 18407830_Leucine-rich repeat motif 5 (LRR5) of PLAP-1/asporin, plays an important role in the PLAP-1/asporin-BMP-2 interaction. 20052601_Findings suggest that asporin is one of the important cartilage matrix proteins that binds to the ECM and TGF-beta1 and thereby modulates interactions between TGF-beta and its signaling receptors. 26239644_positively regulates FGF-2 activity through a direct interaction 26399972_inhibition of TLR2- and TLR4-induced inflammatory responses and binding abilities to TLR2 and TLR4 28859141_Asporin deficiency changes skin glycosaminoglycan composition, and decorin and biglycan content, which may explain the changes in skin mechanical properties. 30511172_PLAP-1 gets involved in the development of osseous eruption canal. 31123087_Data establish a role for ASPN as a critical MSC factor that extensively affects the tumor microenvironment and induces metastatic progression. 33654143_Mice lacking PLAP-1/asporin counteracts high fat diet-induced metabolic disorder and alveolar bone loss by controlling adipose tissue expansion. 34705621_Asporin Promotes TGF-beta-induced Lung Myofibroblast Differentiation by Facilitating Rab11-Dependent Recycling of TbetaRI. 35470068_Asporin, an extracellular matrix protein, is a beneficial regulator of cardiac remodeling. 36245218_Plap-1 lineage tracing and single-cell transcriptomics reveal cellular dynamics in the periodontal ligament. 36445954_Periodontal ligament-associated protein-1 engages in teeth overeruption and periodontal fiber disorder following occlusal hypofunction. |
ENSG00000106819 |
ASPN |
15601.206242 |
0.3952050885 |
-1.339327 |
0.07135129 |
341.535369 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000029545863072980219125641723372766960638931631940204555554355868758556525703586002845650730910668448700614089156934559437415069559228517615548420574209845859371841007190039253602614071920335 |
0.0000000000000000000000000000000000000000000000000000000000000000000000001046610665878220274752566386168671640303762675479308058094988662623356256700154239170487890403813426701769775892391001915790691249110220778632752571467913408444520673981315981971944827078 |
Yes |
No |
8771.62806926951 |
433.994786428819 |
22195.1293742904 |
818.236706213436 |
| ENSMUSG00000021476 |
56541 |
Habp4 |
protein_coding |
E9QKB2 |
Human_homologues FUNCTION: RNA-binding protein that plays a role in the regulation of transcription, pre-mRNA splicing and mRNA translation (PubMed:14699138, PubMed:16455055, PubMed:19523114, PubMed:21771594). Negatively regulates DNA-binding activity of the transcription factor MEF2C in myocardial cells in response to mechanical stress (By similarity). Plays a role in pre-mRNA splicing regulation (PubMed:19523114). Binds (via C-terminus) to poly(U) RNA (PubMed:19523114). Involved in mRNA translation regulation, probably at the initiation step (PubMed:21771594). Seems to play a role in PML-nuclear bodies formation (PubMed:28695742). {ECO:0000250|UniProtKB:A1L1K8, ECO:0000269|PubMed:14699138, ECO:0000269|PubMed:16455055, ECO:0000269|PubMed:19523114, ECO:0000269|PubMed:21771594, ECO:0000269|PubMed:28695742}. |
Cytoplasm;Nucleus;Proteomics identification;Reference proteome;RNA-binding |
|
|
mmu:56541; |
Cajal body [GO:0015030]; cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; Gemini of coiled bodies [GO:0097504]; nuclear membrane [GO:0031965]; nuclear speck [GO:0016607]; nucleolus [GO:0005730]; sarcomere [GO:0030017]; RNA binding [GO:0003723]; SUMO binding [GO:0032183]; cellular response to mechanical stimulus [GO:0071260]; negative regulation of DNA binding [GO:0043392]; PML body organization [GO:0030578]; positive regulation of RNA splicing [GO:0033120]; positive regulation of translational initiation [GO:0045948] |
Human_homologues 12505151_interacts with the C-terminal region of the human chromatin-remodeling factor CHD-3 14699138_association of Ki-1/57 with the RACK1/PKC pathway and may be important for the regulation of its cellular functions 15862299_MEF2C DNA-binding activity is inhibited through its interaction with Ki-1/57. 16879614_Has two conserved Gly/Arg-rich motif clusters (RGG/RXR box, where X is any amino acid) that may be substrates for arginine-methylation by PRMT1. 18788774_Data suggest that Ki-1/57 has characteristics of an intrinsically unstructured protein, which may explain its functional plasticity. 19523114_Findings suggest that Ki-1/57 is probably involved in cellular events related to RNA functions, such as pre-mRNA splicing. 20436279_Ki-1/57 is localized to several subnuclear domains, all of which have been described to splicing and other RNA processing events. 20551049_Observational study of gene-disease association. (HuGE Navigator) 21771594_Ki-1/57 may be involved in translational regulation 28695742_Ki-1-57 is a target of sumoylation and affects pml nuclear body formation. |
ENSG00000130956 |
HABP4 |
1382.414493 |
0.4573965642 |
-1.128483 |
0.07912585 |
201.274611 |
0.00000000000000000000000000000000000000000000110074695738781279498051012501600309906471659751299205910278222265796998824520474920134653136504586702629326352758862284098029249435057863593101501464843750000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000001581752608955770404280333042287978383249453839810724182476056925209654074912504132006431563013472613917038495307335055350961283693322911858558654785156250000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
871.576646879205 |
47.8263373392248 |
1905.51638376521 |
75.841158973724 |
| ENSMUSG00000021567 |
72293 |
Nkd2 |
protein_coding |
Q8VE28 |
FUNCTION: Cell autonomous antagonist of the canonical Wnt signaling pathway. May activate a second Wnt signaling pathway that controls planar cell polarity. Required for processing of TGFA and for targeting of TGFA to the basolateral membrane of polarized epithelial cells (By similarity). {ECO:0000250}. |
Alternative splicing;Calcium;Cell membrane;Cytoplasm;Cytoplasmic vesicle;Exocytosis;Lipoprotein;Membrane;Metal-binding;Myristate;Reference proteome;Transport;Ubl conjugation;Wnt signaling pathway |
|
|
mmu:72293; |
basolateral plasma membrane [GO:0016323]; cell periphery [GO:0071944]; cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; exocytic vesicle [GO:0070382]; lateral plasma membrane [GO:0016328]; plasma membrane [GO:0005886]; ATPase binding [GO:0051117]; calcium ion binding [GO:0005509]; growth factor binding [GO:0019838]; molecular adaptor activity [GO:0060090]; myosin heavy chain binding [GO:0032036]; ubiquitin protein ligase binding [GO:0031625]; exocytosis [GO:0006887]; Golgi vesicle fusion to target membrane [GO:0048210]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of Wnt signaling pathway [GO:0030178]; positive regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032436]; positive regulation of protein localization to plasma membrane [GO:1903078]; positive regulation of protein processing [GO:0010954]; protein localization to plasma membrane [GO:0072659]; Wnt signaling pathway [GO:0016055] |
17094106_Mouse naked cuticle 2 (mNkd2) is a direct transcriptional target of Hoxc8 in vivo. 25579177_studies provide compelling evidence that downregulation of NKD2 expression and alterations in associated regulated pathways have a significant role in driving OS tumor growth and metastasis |
ENSG00000145506 |
NKD2 |
59.118648 |
0.4650260892 |
-1.104616 |
0.40153859 |
7.496080 |
0.00618334436309314703500428223037488351110368967056274414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0222606242823528283070189104364544618874788284301757812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.2159893587186 |
10.4924043000558 |
80.0298955732567 |
16.2899158961328 |
| ENSMUSG00000021575 |
11624 |
Ahrr |
protein_coding |
A0A0R4J1D3 |
Human_homologues FUNCTION: Mediates dioxin toxicity and is involved in regulation of cell growth and differentiation. Represses the transcription activity of AHR by competing with this transcription factor for heterodimer formation with the ARNT and subsequently binding to the xenobiotic response element (XRE) sequence present in the promoter regulatory region of variety of genes. Represses CYP1A1 by binding the XRE sequence and recruiting ANKRA2, HDAC4 and/or HDAC5. Autoregulates its expression by associating with its own XRE site. {ECO:0000269|PubMed:17890447, ECO:0000269|PubMed:18172554}. |
Reference proteome |
|
This gene encodes a protein that represses aryl hydrocarbon receptor-dependent signaling. The encoded protein competes with the aryl hydrocarbon receptor transcription factor for heterodimerization with the aryl hydrocarbon receptor nuclear translocator protein and binding to xenobiotic response element (XRE) sequence in many genes. This protein is implicated in the regulation of cell growth and differentiation as well as mediating dioxin toxicity. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Nov 2015]. |
|
|
16205913_AhRR mRNA expression pattern in untreated C57BL/6 mice varies across tissues with high levels in hearts and brains 24806346_Immortalized TiPARP(-/-) mouse embryonic fibroblasts (MEFs) and AHRR(-/-) MEFs exhibited enhanced AHR transactivation. 25265996_Suggest AHRR mediates dioxin inhibition of lactation by blocking transcription of beta-casein. 27184933_These findings highlight the physiologic importance of cell-type specific balancing of AhR/AhRR expression in response to microbial, nutritional and other environmental stimuli. Specifically, AhRR contributes to the maintenance of colonic intraepithelial lymphocytes and prevents excessive IL-1beta production and Th17/Tc17 differentiation. In contrast, the AhRR enhances IFN-gamma-production by effector T cells in the in... 29432714_Since AhRR knockdown increases the expression and activity of peroxisome proliferator-activated receptor gamma (PPARgamma), AhRR negatively regulates PPARgamma during adipogenesis 32213963_AHR Signaling Dampens Inflammatory Signature in Neonatal Skin gammadelta T Cells. |
ENSG00000063438 |
AHRR |
83.715930 |
3.0077575523 |
1.588688 |
0.30598848 |
26.738115 |
0.00000023298028315411267499960353433846593773637323465663939714431762695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000022865693769352092156333516870247990482312161475419998168945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
126.247830745018 |
26.9777464900939 |
41.9740715632158 |
7.03269923722835 |
| ENSMUSG00000021611 |
21752 |
Tert |
protein_coding |
O70372 |
FUNCTION: Telomerase is a ribonucleoprotein enzyme essential for the replication of chromosome termini in most eukaryotes. Active in progenitor and cancer cells. Inactive, or very low activity, in normal somatic cells. Catalytic component of the teleromerase holoenzyme complex whose main activity is the elongation of telomeres by acting as a reverse transcriptase that adds simple sequence repeats to chromosome ends by copying a template sequence within the RNA component of the enzyme. Catalyzes the RNA-dependent extension of 3'-chromosomal termini with the 6-nucleotide telomeric repeat unit, 5'-TTAGGG-3'. The catalytic cycle involves primer binding, primer extension and release of product once the template boundary has been reached or nascent product translocation followed by further extension. More active on substrates containing 2 or 3 telomeric repeats. Telomerase activity is regulated by a number of factors including telomerase complex-associated proteins, chaperones and polypeptide modifiers. Modulates Wnt signaling. Plays important roles in aging and antiapoptosis (By similarity). {ECO:0000250, ECO:0000269|PubMed:17130244, ECO:0000269|PubMed:19571879, ECO:0000269|PubMed:9582020}. |
Chromosome;Cytoplasm;DNA-binding;Magnesium;Metal-binding;Nucleotidyltransferase;Nucleus;Phosphoprotein;Reference proteome;Ribonucleoprotein;RNA-directed DNA polymerase;Telomere;Transferase;Ubl conjugation |
|
|
mmu:21752; |
chromosome, telomeric region [GO:0000781]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; mitochondrial nucleoid [GO:0042645]; mitochondrion [GO:0005739]; nuclear speck [GO:0016607]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; PML body [GO:0016605]; RNA-directed RNA polymerase complex [GO:0031379]; telomerase catalytic core complex [GO:0000333]; telomerase holoenzyme complex [GO:0005697]; TERT-RMRP complex [GO:1990572]; chaperone binding [GO:0051087]; DNA binding [GO:0003677]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; protein C-terminus binding [GO:0008022]; protein homodimerization activity [GO:0042803]; protein N-terminus binding [GO:0047485]; RNA binding [GO:0003723]; RNA-dependent RNA polymerase activity [GO:0003968]; RNA-directed DNA polymerase activity [GO:0003964]; telomerase activity [GO:0003720]; telomerase RNA binding [GO:0070034]; telomerase RNA reverse transcriptase activity [GO:0003721]; telomeric DNA binding [GO:0042162]; template-free RNA nucleotidyltransferase [GO:0098680]; transcription coactivator binding [GO:0001223]; tRNA binding [GO:0000049]; cellular response to hypoxia [GO:0071456]; DNA biosynthetic process [GO:0071897]; DNA strand elongation [GO:0022616]; establishment of protein localization to telomere [GO:0070200]; mitochondrion organization [GO:0007005]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cellular senescence [GO:2000773]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001240]; negative regulation of gene expression [GO:0010629]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of neuron apoptotic process [GO:0043524]; positive regulation of angiogenesis [GO:0045766]; positive regulation of G1/S transition of mitotic cell cycle [GO:1900087]; positive regulation of glucose import [GO:0046326]; positive regulation of hair cycle [GO:0042635]; positive regulation of miRNA transcription [GO:1902895]; positive regulation of nitric-oxide synthase activity [GO:0051000]; positive regulation of protein binding [GO:0032092]; positive regulation of protein localization to nucleolus [GO:1904751]; positive regulation of stem cell proliferation [GO:2000648]; positive regulation of transdifferentiation [GO:1903620]; positive regulation of vascular associated smooth muscle cell migration [GO:1904754]; positive regulation of vascular associated smooth muscle cell proliferation [GO:1904707]; positive regulation of Wnt signaling pathway [GO:0030177]; regulation of protein stability [GO:0031647]; replicative senescence [GO:0090399]; response to cadmium ion [GO:0046686]; RNA-templated DNA biosynthetic process [GO:0006278]; RNA-templated transcription [GO:0001172]; siRNA processing [GO:0030422]; siRNA transcription [GO:0140745]; telomere maintenance via telomerase [GO:0007004] |
11788723_presence of the P6.1 stem-loop in telomerase RNA and its importance for the assembly and enzymatic activity of the telomerase complex 11904422_Preferential maintenance of critically short telomeres in mammalian cells heterozygous for mTert 11929765_Under steady-state conditions, the levels and functionality of hematopoietic-committed or multipotent progenitors were not affected by telomerase deficiency. 11950883_data show that Kitl induces telomerase activity in mitotic spermatogonia and increases the mRNA levels of both the catalytic subunit form and the telomerase RNA template 12037663_role of Ets-mediate transactivation in activation of telomerase by EGF 12242304_Cooperation between p53 mutation and high telomerase transgenic expression in spontaneous cancer development. 12461078_one role for telomerase in T cells may be to renew or extend replicative potential via the rejuvenation of telomere length. 12663456_One role for telomerase in the HSC is to partially counter the rate of telomere shortening during division of HSCs, thereby preventing premature loss of telomere function and providing added replicative capacity. 14612409_exogenous wtBRCA1 strongly suppressed TERT promoter activity in various cell lines 14960598_TERT is inducible in postmitotic neurons after ischemic brain injury and prevents NMDA neurotoxicity through shift of the cytosolic free Ca2+ into the mitochondria, and thus plays a protective role in ameliorating ischemic neuronal cell death 15079066_limiting TERT levels play a key role in the maintenance of genome integrity, with important ramifications for the maintenance of short telomeres in human cancer and aging 15121848_Lck-Tert mouse lines showed higher incidences of spontaneous T-cell lymphoma than the corresponding age-matched wild-type controls, indicating that constitutive expression of Tert promotes lymphoma. 15184392_telomerase activity is regulated by glutathione 15282303_Data show that telomerase RNA template (TR) and telomerase reverse transcriptase (TERT) are essential for telomere maintenance and elongation but gene copy number and transcriptional regulation of TR, but not TERT, are limiting for telomerase activity. 15608677_In knockout mice, negative impact of telomere shortening on organ homeostasis and organismal survival can surpass the beneficial effects of telomere shortening on suppression of tumor growth in the setting of chronic organ damage. 15687103_Ectopic mTERT expression protects embryonic stem cells against cell death during differentiation. 15860505_Telomere dysfunction induces a robust compensatory response to rescue impaired germ cell function through the induction of survival signals. 15919739_the telomerase activity of cells in various stages of differentiation was unaffected by aging and, notably, remained high in the alpha6-integrin-positive side Population 16037417_findings show that telomere length, as well as the catalytic component of telomerase, Tert, are critical determinants in the mobilization of epidermal stem cells 16131840_high rate of genomic rearrangements in telomerase deficient ES cells may reflect the cultured cells' gained ability to protect chromosome ends with eroded telomeres allowing them to escape 'end crisis' 16135795_there was no effect on mTERT expression or mTERT promoter activity by AP-1 overexpression in mouse fibroblasts. (TERT) 16344462_GC-box-mediated, human-specific mechanism for TERT repression is impaired in human cancers 16457732_The 1 kb promoter upstream of the initiating ATG codon of mTert contains all the regulatory elements to control telomerase expression in ES cells during in vitro loss of pluripotency. 16501597_Abrogates the growth-inhibitory effect of transforming growth factor-beta in embryonic fibroblasts. 16831983_Mice lacking telomerase activity show hypertension as a result of an increase in plasma ET-1 levels, which is a consequence of ECE-1 overexpression. A direct link between telomerase activity and hypertension is reported. 16847266_telomerase activity controls the glycolytic pathway, potentially altering the energy state of tumor cells and thereby modulating tyrosinase activity and melanin production 16880523_These results highlight telomerase as a mediator of Myc-induced papillomatosis and suggest telomerase as a putative therapeutic target for Myc-dependent lesions. 16979586_These results show for the first time that IP6 represses telomerase activity in prostate cancer cells by posttranslational modification of TERT via the deactivation of Akt and PKCalpha. 17130244_Murine telomere homeostasis or genetic stability does not depend on mPif1. 17145779_DNA-dependent protein kinase deficiency does not reduce apoptosis, tissue atrophy, or p53 activation in late-generation mTerc(-/-) tissues but rather moderately exacerbates germ cell apoptosis and testicular degeneration 17433324_telomerase limits the accumulation of telomere dysfunction, the evolution of excessive aneuploidy, and the activation of p53-independent checkpoints suppressing hepatocarcinogenesis. 17456801_Large defects of type I allergic response in Tert knockout mice. 18008008_TERT induction is associated with increased survival of lung fibroblasts, which favors the development of fibrosis instead of injury resolution. 18067891_Thus, mouse cells, in contrast to human cells may not tolerate telomerase inhibition by introduction of mTERT-DN, independently of telomere length. 18208333_TERT promotes epithelial proliferation through transcriptional control of a Myc- and Wnt-related developmental program. 18223679_telomerase activity-independent TERT function may contribute to cancer development and aging independently of telomere lengthening. 18256293_telomerase can potentiate the transition between pluripotent stem cell and committed neuron in both embryonal carcinoma and embryonal stem cells 18285708_VEGF, hTERT and Bcl-xl have roles in laryngeal squamous carcinoma 18708758_there is a synergistic effect between TERT siRNA and cisplatin in the suppression of HCC progression 18729084_hTERTp-lacZ transgenic mouse to analyze mouse Tert gene expression & human TERT promoter during testis development; hTERT promoter activity increased in puberty & was highest in adult; mouse Tert expression was high in testis from earliest time point 18818403_PPARalpha trans-repression involved the recruitment of the retinoblastoma (RB) family proteins p107 and p130 to the TERT promoter resulting in impaired E2F-1 binding, an effect that was dependent on p16. 18936156_These results indicate an essential role for Pot1b in the maintenance of genome integrity and the long-term viability of proliferative tissues in the setting of telomerase deficiency. 18936784_Gene expression analysis showed that, in the absence of estrogen, telomerase reverse transcriptase (TERT) gene expression is reduced in association with compromised cell proliferation in the adrenal gland cortex and adrenal atrophy. 19013273_Telomerase reverse transcriptase delays aging in cancer-resistant mice 19080380_Selenium and benzene can upregulate the telomerase activity in mouse lymphocytes in vivo. 19265030_Mitochondrial telomerase reverse transcriptase binds to and protects mitochondrial DNA and function from damage. 19270068_modest mTERT expression was detected in terminally differentiated and post-mitotic cells. 19571879_The telomerase protein component TERT (telomerase reverse transcriptase) interacts with BRG1, a SWI/SNF-related chromatin remodelling protein, and activates Wnt-dependent reporters in cultured cells and in vivo 19575911_TERT was up-regulated in a mouse model of oxygen-induced retinopathy. 19685288_results suggest that TERT is constitutively expressed in the hippocampus and the olfactory bulbs, and that it is important for regulating normal brain functions. 19841238_Telomerase reverse transcriptase-dependent telomere equilibration mitigates tissue dysfunction in mTert heterozygotes 19850716_mTERT does not act as a transcription factor or play a role in the DNA damage response. 19903789_deregulated GSK3beta sustains gastrointestinal cancer cells survival through modulation of hTERT and telomerase 20157588_DeltaNp63alpha regulates mTERT activity at the transcriptional level via the mTERT promotor site, and also through the induction of spliced mRNA mTERT isoforms. 20354136_the hTERT promoter was strongly activated in discrete steps, revealing a critical difference in human and mouse cell reprogramming 20486780_Tumor volumes significantly decreased, and appearances of tumor formation in mice were delayed in the TERT-knockdown embryonic stem cells. 20643931_Direct shRNA-mediated knockdown of Hif1alpha expression confirmed that suppression of hypoxia inducible factor 1 alpha levels was accompanied by a reduction in both Tert mRNA and telomerase activity levels. 20864668_Vascular injury induces de novo expression of TERT in intimal smooth muscle cells via activation of nuclear factor kappa B and upregulation of c-Myc,leading to hyperplasia. 20937271_Hypertonic stress activates mTERT transcription via the activation and recruitment of NFAT5 to the mTERT promoter. 21088250_TERT deficiency in bone marrow-derived macrophages attenuates Ang II-induced abdominal aortic aneurysms formation in mice. These results point to a previously unrecognized role of TERT in the pathogenesis of abdominal aortic aneurysms. 21106948_TERT expression prevents macrophage senescence and may have important implications for the development of atherosclerosis. 21113150_Telomerase reactivation extends telomeres, reduces DNA damage signalling and associated cellular checkpoint responses, allows resumption of proliferation in quiescent cultures, and eliminates degenerative phenotypes across multiple organs 21173232_mTert(+) cells give rise to all differentiated intestinal cell types, persist long term, and contribute to the regenerative response following injury. 21195081_Results suggest that oxidant stress might affect myocardial telomerase activity and telomere-associated proteins. 21297001_Atm and Tert doubly deficient mice demonstrated increased progression of aging and had shorter lifespans than Atm-null mice, while Tert alone was insufficient to affect lifespan. 21300782_PITX1 suppresses TERT transcription through direct binding to the TERT promoter, which ultimately regulates telomerase activity 21364961_Pancreatic regeneration is limited in the context of telomere dysfunction without evidence for p53 checkpoint activation. 21396644_Pioglitazone treatment up-regulates telomerase activity, telomere-stabilizing proteins and reduces senescence markers in vascular cells. 21464209_Results indicate that both telomerase reverse transcriptase and telomerase RNA are haploinsufficient and that their deficiency leads to telomere shortening, which limits tissue renewal. 21672962_The results of this study provide the first experimental evidence that telomere shortening, despite impairing adult neurogenesis and maintenance of post-mitotic neurons, can slow down the progression of amyloid plaque pathology in Alzheimer's disease. 21730156_a complex composed of TERT, BRG1, and NS/GNL3L maintains the function of tumor initiating cells 21730353_Primitive hematopoietic Tert(-/-) cells lacking telomerase activity exhibit signs of enhanced DNA damage. 21865469_Hippocampal telomerase is involved in the modulation of depression-related behaviors, possibly by regulating adult neurogenesis. 22138751_Silencing transgenic TERT expression or inhibiting Wnt signaling through systemic expression of the Wnt inhibitor Dkk1 in either TERT transgenic mice or in a mouse model of HIVAN results in marked normalization of podocytes 22341455_Data provide evidence that telomere dysfunction plays a critical role in prostate cancer initiation and progression, permitting acquisition of and selection for cancer-relevant genomic events upon telomerase reactivation. 22408137_Essential role for telomerase in chronic myeloid leukemia induced by BCR-ABL in mice. 22533433_telomerase plays a role in the aging of nondividing cells 22633954_Data show that AUF1 binds and strongly activates the transcription promoter for telomerase catalytic subunit Tert. 22723415_findings show beta-catenin regulates Tert expression through interaction with Klf4, a core component of the pluripotency transcriptional network; beta-Catenin binds to the Tert promoter in a mouse intestinal tumor model 22855157_mTR stability and telomerase activity produced by the Dkc1 A353V mutation in dyskeratosis congenita are rescued by a peptide from the dyskerin TruB domain. 22884695_Overexpression of TERT in mesenchmal stem cells resulted in increased cell proliferation. 22919071_Study identified the cells responsible for cardiac telomerase activity, demonstrates a significant diminution with age. 23055531_mir498 has a role in 1,25-Dihydroxyvitamin D3 suppression of telomerase expression and cancer growth 23056318_prepared two mutant forms of the PhSurv-PhTERT tandem with two or four Sp1 sites removed from the distal 'long' PhSurv promoter 23349740_calorie restriction attenuates telomere erosion associated to aging and that synergizes with TERT over-expression in increasing 'health span' and extending mouse longevity 23526226_TERT expression was up-regulated by a histone deacetylase inhibitor, while the induction of TERT in lung fibroblasts was associated with the binding of acetylated histone H3K9 to the TERT promoter region. 24119029_These findings establish a functional link between endoplasmic reticulum stress and telomerase. 24254728_Once HIV production had reached a peak (7 dpi), the telomerase activity decreased, showing levels similar to those of noninfected cells 24401839_TERT, combined with beta-catenin and BRG1, serves as a transcriptional complex, which binds the FAS ligand (FASL) promoter to upregulate FASL expression, leading to an elevated immunomodulatory function. 24733392_These data indicate that TERT plays an extratelomeric role in the reprogramming process, but its function is dispensable. 25032857_The overexpression of Zfp637 markedly increases mTERT expression and telomerase activity, maintains telomere length, and inhibits H2O2 and D-galactose-induced senescence. 25038516_The lack of telomerase produce specific phenotypes, some of which can be prevented by dietary restriction. 25118183_Telomerase may direct Pol I transcription in oncogenic and regenerative hyperproliferation. 25127175_Pharmacologically relevant doses of atorvastatin resulted in a 6-fold increase of telomerase activity in mouse PBMCs and CD4 T cells. Transgenic GFP-mTert reporter mice had 30% to 15% less telomerase-positive lymphocytes during the first 5 months of age. 25519492_Tert expression confers cardioprotection in the adult mouse heart after acute myocardial infarction. 25550449_Telomerase exerts telomere-independent effects on pulmonary artery smooth muscle cell growth in pulmonary hypertension. 25586649_Studies indicate that reverse transcriptase (RT) enzyme highly expressed in mouse embryos and mouse and human cancer cells and repressed in somatic differentiated healthy cells. 25893605_TERT is a regulator of MYC stability in cancer. Reactivation of TERT, a direct transcriptional MYC target in tumors, provides a feed-forward mechanism to potentiate MYC-dependent oncogenesis. 26082434_data suggest that S1P binding to hTERT allosterically mimicks phosphorylation, promoting telomerase stability and hence telomere maintenance, cell proliferation, and tumor growth. 26258591_miR-512-5p suppresses tumor growth by targeting TERT in telomerase positive head and neck squamous cell carcinoma in vitro and in vivo. 26374457_Results suggest that in mature Purkinje neurons, TERT is present both in the nucleus and in mitochondria, where it may participate in adaptive responses of the neurons to excitotoxic and radiation stress 26390028_miR-195 overexpressed in old mesenchymal stem cells (OMSCs) induces stem cell senescence deteriorating their regenerative ability by directly deactivating telomerase reverse transcriptase (Tert), and abrogation of miR-195 can reverse stem cell aging. 26518879_Telomerase deficiency triggers alveolar stem cell replicative senescence-associated low-grade inflammation. 26555817_findings identified a key role for TERT in fibroblast proliferation and survival essential for pulmonary fibrosis 26584619_high telomerase expression is a fundamental characteristic of germline stem cells. 26725521_TERT switches macrophages towards M1 phenotype by regulating NF-kappaB signaling, but has limited effect on M2 macrophages polarization in vitro. 26740067_The results suggest that the mouse endometrial epithelium and vasculature are foci of stem/progenitor activity expressing mTert. 26787169_This study reports the characterisation of two novel mouse TERT splice variants, Ins-i1[1-102] (Insi1 for short) and Del-e12[1-40] (Dele12 for short) that have not been previously described. Insi1 represents an in-frame insertion of nucleotides 1-102 from intron 1, encoding a 34 amino acid insertion at amino acid 73. 26903137_TERT may promote gastric cancer metastasis through the TERT-miR-29a-ITGB1 regulatory pathway. 26903545_These findings indicate that telomerase gene therapy represents a novel therapeutic strategy to treat aplastic anemia provoked or associated with short telomeres. 27148686_Nrf2-driven TERT regulates pentose phosphate pathway in glioblastoma. 27300262_Reactivation of Tert in the hippocampus was sufficient to normalize the depressive but not the aggressive behaviors of Tert(-/-) mice. Conversely, re-expression of Tert in the medial prefrontal cortex (mPFC) reversed the aggressive but not the depressive behavior of Tert(-/-) mice. 27545609_these findings support a model in which gain of TERT function modulates mTORC1 activity and induces autophagy. 27633795_Regarding extratelomeric activities, our results showed a decrease of 64, 38 and 25% in the transcription of c-Myc, Cyc-D1 and TERT, respectively (p |
ENSG00000164362 |
TERT |
36.760016 |
12.2401433217 |
3.613549 |
0.81321306 |
17.583168 |
0.00002750118101078915153179758890100714552318095229566097259521484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001867579086742489238685827057651067661936394870281219482421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
65.8249225584949 |
39.1002490657874 |
5.37779018009395 |
2.55596202414985 |
| ENSMUSG00000021684 |
218461 |
Pde8b |
protein_coding |
E9Q4S1 |
FUNCTION: Hydrolyzes the second messenger cAMP, which is a key regulator of many important physiological processes. May be involved in specific signaling in the thyroid gland (By similarity). {ECO:0000250}. |
cAMP;Hydrolase;Metal-binding;Phosphoprotein;Reference proteome |
PATHWAY: Purine metabolism; 3',5'-cyclic AMP degradation; AMP from 3',5'-cyclic AMP: step 1/1. |
|
|
3',5'-cyclic-AMP phosphodiesterase activity [GO:0004115]; 3',5'-cyclic-nucleotide phosphodiesterase activity [GO:0004114]; metal ion binding [GO:0046872]; behavioral fear response [GO:0001662]; cAMP catabolic process [GO:0006198]; negative regulation of insulin secretion [GO:0046676]; negative regulation of insulin secretion involved in cellular response to glucose stimulus [GO:0061179]; negative regulation of steroid hormone biosynthetic process [GO:0090032]; neuromuscular process controlling balance [GO:0050885]; operant conditioning [GO:0035106]; signal transduction [GO:0007165]; visual learning [GO:0008542] |
21187369_Phosphodiesterase 8B is a major regulator of pools of cyclic AMP that promote steroidogenesis. 22232524_findings suggest that both PDE8A and PDE8B play essential roles to maintain low cAMP levels, thereby suppressing resting steroidogenesis by keeping CEH/HSL inactive and StAR protein expression low 22925203_Pde8b knockout mice demonstrate improvement in select behaviors, motor-coordination, attenuation of age-induced motor coordination decline and increased anxiety levels. 33338547_Pde8b haploinsufficiency in mice is associated with modest adrenal defects, impaired steroidogenesis, and male infertility, unaltered by concurrent PKA or Wnt activation. |
ENSG00000113231 |
PDE8B |
60.232625 |
3.2165614239 |
1.685519 |
0.33469240 |
24.863834 |
0.00000061525908710887315664138201565247499047472956590354442596435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000056866665138606524787577393975634976186483982019126415252685546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
91.0935043400735 |
21.3229537698812 |
28.3201507251135 |
5.15076654511557 |
| ENSMUSG00000021699 |
238871 |
Pde4d |
protein_coding |
Q01063 |
FUNCTION: Hydrolyzes the second messenger cAMP, which is a key regulator of many important physiological processes. {ECO:0000250}. |
Alternative splicing;cAMP;Cell membrane;Cytoplasm;Cytoskeleton;Hydrolase;Isopeptide bond;Magnesium;Manganese;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Ubl conjugation;Zinc |
PATHWAY: Purine metabolism; 3',5'-cyclic AMP degradation; AMP from 3',5'-cyclic AMP: step 1/1. |
|
mmu:238871; |
apical plasma membrane [GO:0016324]; calcium channel complex [GO:0034704]; centrosome [GO:0005813]; cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; myofibril [GO:0030016]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; potassium channel complex [GO:0034705]; protein-containing complex [GO:0032991]; voltage-gated calcium channel complex [GO:0005891]; 3',5'-cyclic-AMP phosphodiesterase activity [GO:0004115]; 3',5'-cyclic-nucleotide phosphodiesterase activity [GO:0004114]; ATPase binding [GO:0051117]; beta-2 adrenergic receptor binding [GO:0031698]; cAMP binding [GO:0030552]; cyclic-nucleotide phosphodiesterase activity [GO:0004112]; enzyme binding [GO:0019899]; heterocyclic compound binding [GO:1901363]; metal ion binding [GO:0046872]; scaffold protein binding [GO:0097110]; transmembrane transporter binding [GO:0044325]; adrenergic receptor signaling pathway [GO:0071875]; aging [GO:0007568]; cAMP catabolic process [GO:0006198]; cellular response to cAMP [GO:0071320]; cellular response to epinephrine stimulus [GO:0071872]; cellular response to lipopolysaccharide [GO:0071222]; establishment of endothelial barrier [GO:0061028]; memory [GO:0007613]; multicellular organism growth [GO:0035264]; negative regulation of cAMP-mediated signaling [GO:0043951]; negative regulation of heart contraction [GO:0045822]; negative regulation of peptidyl-serine phosphorylation [GO:0033137]; negative regulation of relaxation of cardiac muscle [GO:1901898]; neutrophil chemotaxis [GO:0030593]; neutrophil migration [GO:1990266]; positive regulation of cell-cell adhesion [GO:0022409]; positive regulation of interleukin-2 production [GO:0032743]; positive regulation of interleukin-5 production [GO:0032754]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of smooth muscle cell migration [GO:0014911]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of type II interferon production [GO:0032729]; protein-containing complex assembly [GO:0065003]; regulation of cAMP-mediated signaling [GO:0043949]; regulation of cardiac muscle cell contraction [GO:0086004]; regulation of cell communication by electrical coupling involved in cardiac conduction [GO:1901844]; regulation of G protein-coupled receptor signaling pathway [GO:0008277]; regulation of heart rate [GO:0002027]; regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum [GO:0010880]; regulation of ryanodine-sensitive calcium-release channel activity [GO:0060314]; regulation of signaling receptor activity [GO:0010469]; signal transduction [GO:0007165]; smooth muscle contraction [GO:0006939]; T cell receptor signaling pathway [GO:0050852] |
12834813_Reported here the cloning and characterization of a novel PDE4D isoform, PDE4D7. 14519662_PDE4D plays a critical role in the control of trachea smooth muscle contraction. 15644445_results suggest PDE4D is an integral component of the beta2AR signaling complex and illustrate a mechanism for fine-tuned betaAR subtype signaling specificity and intensity in the cardiac system 18006274_Deletion study confirmed that the N-terminus of PDE4D1 is necessary for nuclear targeting. In addition, we showed that the unique N-terminus contains nuclear localization signal sequence. 18274640_PDE4 inhibitors suppressed the pannus-like inflammation by inhibition of cytokine production by macrophages and synovial fibroblast proliferation 18301971_These findings suggest that PDE4D1, -2, and -9 play some roles in bone formation. 18508768_PDE4D ablation affected the kinetics of beta2adrenergic receptor desensitization as well as the interaction of the receptor with Galphai indicaating that PDE4D plays a major role in shaping the receptor signal. 18702734_Data suggest that the phosphodiesterase 4D isoform may be an important mediator of long-term memory formation. 18804162_Phosphodiesterase 4D11 isoform isoform is ubiquitously expressed in different mouse tissues, particularly in the brain, liver and spleen. The expression levels increased in the cerebellum, but decreased in the hippocampus with progressive age 19218243_PDE4D3, like protein kinase A and protein phosphatase 1, is recruited to the I(Ks) channel via AKAP-9 and contributes to its critical regulation by cAMP. 19401450_PDE4D functions as a proliferation promoting factor in prostate cancer. 19596983_increased PDE4D5 and muscarinic receptor expression is associated with airway hyperreactivity due to maternal exposure to secondhand cigarette smoke 19801680_Differential association of phosphodiesterase 4D isoforms with beta2-adrenoceptor in cardiac myocytes 20647544_Results identify a novel signaling process governing G-protein-coupled cAMP signal transduction-opposing actions of the phosphatase calcineurin and the CK1/GSK3beta protein kinases on the phosphodegron-dependent degradation of PDE4D. 20705779_Activation of phosphodiesterase 4D constrains myocardial contractility via a ss2 adrenergic receptor-coupled phosphoinositide 3-kinase mechanism. 20837708_results suggest a novel regulatory mechanism for mTORC1 in which the cAMP-determined dynamic interaction between Rheb and PDE4D provides a key, unique regulatory event, and propose a new role for PDE4 as a molecular transducer for cAMP signaling. 21209202_The present results suggest that PDE4D, in particular long-form PDE4D, plays a critical role in the mediation of memory and hippocampal neurogenesis, which are mediated by cAMP/CREB signaling. 21288894_Findings demonstrate that PDE4D and PDE4B have specialized functions in mouse embryonic fibroblasts with PDE4B controlling cAMP in a discrete subdomain near the plasma membrane. 21670503_Results identify PDE4B in the CaV1.2 complex as a critical regulator of ICa during beta-AR stimulation and suggest that distinct PDE4 subtypes are important for normal regulation of calcium-induced calcium release in cardiomyocytes. 21903937_Phosphodiesterase 4D regulates baseline sarcoplasmic reticulum Ca2+ release and cardiac contractility, independently of L-type Ca2+ current. 22952227_Beta-arrestin-2 scaffolding of phosphodiesterase PDE4D5 to the plasma membrane was required for TRPV1 desensitization. 23258544_Calcineurin regulates degradation of phosphodiesterase 3B, in addition to phosphodiesterase 4D. 23587879_the loss of function of either DISC1 or ATF4 increases PDE4D9 transcription, and the association of DISC1 with the PDE4D9 locus requires ATF4 24220334_these results suggest that Pde4d might serve specific biological functions in regulating the development process of the mouse embryo, and that CpG methylation of the Pde4d promoter may play an important role in regulating Pde4d at a transcriptional level. 24944202_Cytokine-induced iNOS and ERK1/2 inhibit adenylyl cyclase type 5/6 activity and stimulate phosphodiesterase 4D5 activity in intestinal longitudinal smooth muscle, contributing to colonic dysmotility during inflammation. 25882071_The authors demonstrate that the Attention Deficit Disorder with Hyperactivity phenotype depends on a dysregulation of CREB signaling exerted by a kinase-independent PI3Kgamma-PDE4D interaction in the noradrenergic neurons of the locus coeruleus. 25918234_SIK1 serves as a part of a self-regulatory circuit to modulate insulin secretion from pancreatic beta-cells by controlling cAMP concentration through modulation of PDE4D activity. 27100201_Results indicate that the cyclic AMP (cAMP) increase is caused by the attenuation of phosphodiesterase (PDE) activity adrenal cortex-derived Y-1 cell line. 27595237_In vivo knockdown of PDE4D5 inhibited inflammation at athero-prone sites 28235598_These results indicate that Phosphodiesterase-4D overexpression may be involved in the development of surgery-induced cognitive dysfunction in mice. 28455289_The effect of dobutamine as a positive inotrope is impaired in mice with cecal ligation and puncture-induced sepsis without changes in cardiac beta1-adrenoceptor signaling as a result of cAMP breakdown achieved by upregulated phosphodiesterase 4D 29123207_In vivo, the atropine-dependent prolongation of heart rate increase was blunted in PDE4D but not in wildtype or PDE4B knockout mice. The authors propose that inhibition of PDE4 by atropine accounts, at least in part, for the induction of tachycardia and the arrhythmogenic potency of this drug. 29497131_Long-term memory is enhanced by retrosplenial and hippocampal knockdown (KD) of the cAMP specific phosphodiesterase Pde4d. 31408443_Proteomics identified the PP2A regulatory subunit B55alpha as the factor recruiting PP2A to PDE4D5. The B55alpha-PP2A complex localized to adhesions and directly dephosphorylated PDE4D5. 32249206_Upregulation of miR-219a-5p Decreases Cerebral Ischemia/Reperfusion Injury In Vitro by Targeting Pde4d. 32784895_Dominant-Negative Attenuation of cAMP-Selective Phosphodiesterase PDE4D Action Affects Learning and Behavior. 35999453_Phosphodiesterase 4D contributes to angiotensin II-induced abdominal aortic aneurysm through smooth muscle cell apoptosis. 36252429_Circ_0101874 overexpression strengthens PDE4D expression by targeting miR-335-5p to promote neuronal injury in ischemic stroke. |
ENSG00000113448 |
PDE4D |
131.134423 |
0.3838814245 |
-1.381267 |
0.23206878 |
34.951362 |
0.00000000338044788700595830774649982488049698714505097996152471750974655151367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000442742753352319516388725434332535035863998018612619489431381225585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
73.5254268268761 |
11.8782112115056 |
191.531608816453 |
21.9983628733224 |
| ENSMUSG00000021728 |
13723 |
Emb |
protein_coding |
P21995 |
FUNCTION: Plays a role in targeting the monocarboxylate transporters SLC16A1 and SLC16A7 to the cell membrane (By similarity). Plays a role in the outgrowth of motoneurons and in the formation of neuromuscular junctions. Following muscle denervation, promotes nerve terminal sprouting and the formation of additional acetylcholine receptor clusters at synaptic sites without affecting terminal Schwann cell number or morphology. Delays the retraction of terminal sprouts following re-innervation of denervated endplates. {ECO:0000250, ECO:0000269|PubMed:19164284}. |
Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Phosphoprotein;Reference proteome;Repeat;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:13723; |
axon [GO:0030424]; plasma membrane [GO:0005886]; synapse [GO:0045202]; cell-cell adhesion mediator activity [GO:0098632]; axon guidance [GO:0007411]; cell adhesion [GO:0007155]; dendrite self-avoidance [GO:0070593]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156]; plasma membrane lactate transport [GO:0035879] |
19164284_embigin enhances neural cell adhesion molecule-dependent neuromuscular adhesion and thereby modulates neuromuscular junction formation and plasticity. |
ENSG00000170571 |
EMB |
92.443725 |
0.4769157501 |
-1.068194 |
0.27449178 |
15.121789 |
0.00010079321400410928290768131088128711780882440507411956787109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006001885206061738191329890668157531763426959514617919921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
57.0455948679515 |
10.9192801830727 |
119.613568759999 |
16.5546179998935 |
| ENSMUSG00000021765 |
14313 |
Fst |
protein_coding |
P47931 |
FUNCTION: Binds directly to activin and functions as an activin antagonist. Specific inhibitor of the biosynthesis and secretion of pituitary follicle stimulating hormone (FSH). |
Alternative splicing;Disulfide bond;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
The protein encoded by this gene binds to and negatively regulates activin, as well as other members of the transforming growth factor beta family, and acts to prevent uncontrolled cellular proliferation. This protein also contains a heparin-binding sequence. It is expressed in many of the tissues in which activin is synthesized and is thought to clear activin from the circulation by attachment to the cell surface. Alternative splicing results in multiple transcript variants that encode multiple protein isoforms, including FST315 and FST288, that differ at their C-terminus. Another isoform, FST303 is thought to be produced by proteolytic cleavage of FST315. These isoforms differ in their localization and in their ability to bind heparin. While FST315 is a circulating protein, FST288 is tissue-bound, and FST303 is gonad-specific. While deletion of all isoforms results in embryonic lethality, expression of just FST288 is sufficient for embryonic development, but the resultant mice have fertility defects. [provided by RefSeq, Aug 2014]. |
mmu:14313; |
cytoplasm [GO:0005737]; extracellular space [GO:0005615]; nucleus [GO:0005634]; activin binding [GO:0048185]; activin receptor antagonist activity [GO:0038102]; heparan sulfate proteoglycan binding [GO:0043395]; ameloblast differentiation [GO:0036305]; BMP signaling pathway [GO:0030509]; cell differentiation [GO:0030154]; female gonad development [GO:0008585]; gamete generation [GO:0007276]; hair follicle morphogenesis [GO:0031069]; hematopoietic progenitor cell differentiation [GO:0002244]; keratinocyte proliferation [GO:0043616]; negative regulation of activin receptor signaling pathway [GO:0032926]; negative regulation of epithelial cell differentiation [GO:0030857]; negative regulation of transcription by RNA polymerase II [GO:0000122]; odontogenesis of dentin-containing tooth [GO:0042475]; pattern specification process [GO:0007389]; positive regulation of hair follicle development [GO:0051798]; regulation of BMP signaling pathway [GO:0030510]; response to organic cyclic compound [GO:0014070]; skeletal system development [GO:0001501] |
12203361_we studied the expression of follistatin in rat and mouse liver tumors as a possible mechanism to overcome activin growth control, 40% of the tumors displayed increased levels of follistatin mRNA when compared to tumor-surrounding liver tissue. 12761853_up-regulation of the follistatin mRNA level in the small myotubes of the regenerating soleus as well as the down-regulation of the myostatin transcript level after denervation contribute to the differentiation process in denervated regenerating muscle. 15136138_Data suggest that Follistatin antagonises Myostatin by direct protein interaction, which prevents Myostatin from executing its inhibitory effect on muscle development. 15162500_germ cells in the ovarian cortex are almost completely lost in both Wnt4 and Fst null gonads before birth 15451564_review with emphasis on authors' works on paracrine and autocrine regulation in activities of Fst and Fstl3, regulation of gonadal development, and structural elements necessary for biological activities 17482756_experiments provide evidence that GnRH stimulates follistatin through multiple signaling pathways 17597062_decorin up-regulated the expression of follistatin, an antagonist of myostatin. 18768463_repression of Sp1 transcriptional activity by PPARgamma is the underlying mechanism responsible for PPARgamma-mediated regulation of follistatin expression 18804092_Antral hyperplasia in Gas-/- mice involves amplification of mucous cell lineages due to follistatin, suggesting its role in the development of antral gastric tumors. 18922734_This review summarises the activin signal transduction pathways and the interactions between activins and other TGFbeta signalling systems during hair follicle formation, hair growth cycling, skin function and wound healing.[review] 18948405_Regulation of myogenic differentiation by androgens: cross talk between androgen receptor/ beta-catenin and follistatin/transforming growth factor-beta signaling pathways. 19079322_Keratinocyte-derived follistatin regulates epidermal homeostasis and wound repair. 19106105_FoxL2 and Smad3 coordinately regulate follistatin gene transcription.( 19435857_Satellite cell proliferation and both Mstn and Act inhibition are involved in the FS-induced muscle hypertrophy. 19474151_Results suggestthat interactions between mesenchyme-derived follistatin and epithelial BMP7 play a central role in the morphogenesis, innervation and maintenance of taste buds and their stem/progenitor cells. 19715499_This study examined the expression of myostatin and follistatin in Mdx mice and B10 mice; a temporary and coincident expression of follistatin and myostatin in both mouse strains, but this expression was significantly higher in Mdx mice. 20032047_The FST288-only isoform is sufficient for development. Loss of FST303 and FST315 isoforms results in fertility defects that resemble activin hyperactivity and premature ovarian failure. 20566686_link between mTOR and miRNA biogenesis; an mTOR-miR-1-HDAC4-follistatin pathway that regulates myocyte fusion during myoblast differentiation in vitro and skeletal muscle regeneration in vivo. 20810712_Data show that follistatin mutant mice exhibit haploinsufficiency, with muscles of reduced size, a shift toward more oxidative fiber types, impairment of muscle remodeling in response to injury, and reduction in tetanic force production. 21068158_Changes in hepatically metabolised follistatin during and after exercise are reported. 21084449_These data support a model in which FOXL2 and BMP2 cooperate to ensure correct expression of Fst in the developing ovary. 21106872_Results show that FST is critical for normal regulation of germ cell nest breakdown and that loss of the FST303 and/or FST315 isoforms leads to excess primordial follicles with accelerated demise, resulting in premature cessation of ovarian function. 21416223_gradual increase in muscle regeneration, production stimulated in the presence of necrosis 21546932_These results demonstrate that body composition and glucose homeostasis are differentially regulated by FST and FSTL3 and that their combined loss is associated with increased fat mass and insulin resistance despite elevated insulin production. 21689628_follistatin stimulated myoblasts to express MyoD, Myf5, and myogenin, which are myogenic transcription factors that promote myogenic differentiation 21852401_the transition between stem cells and committed progenitors is neither sharp nor irreversible and GDF11, ACTbetaB and FST are crucial components of a circuit that controls both total cell number and the ratio of neuronal versus glial cells in this system 22138414_We conclude that testosterone stimulation of satellite cell proliferation and myogenic differentiation are associated with up regulation of Fst and inhibition of TGF-beta-signaling. 22311089_Data from transgenic mice suggest that overexpression of follistatin negatively influences bone metabolism decreasing all measured biomechanical strength parameters in developing bone. 22711699_regulation of Smad3- and mTOR-dependent events by follistatin occurred independently of overexpression or knockout of myostatin, a key repressor of muscle development that can regulate Smad3 and mTOR signaling and that is itself inhibited by follistatin 22855279_These data indicate that the rapid increase in circulating activin A during LPS-induced inflammation is regulated at the posttranscriptional level. 23143061_Although it is a promising therapeutic tool in chronic or acute inflammatory conditions not caused by virulent pathogens, FS does not seem to increase the resistance to bacterial infections. 23178301_Report role of activin A-myostatin-follistatin system on aging bone and muscle progenitor cells. 24443561_Results implicate a role of follistatin (Fst) in the induction of brown adipocyte character and regulation of energy metabolism. 24514266_Follistatin aids in maintaining proper somite size, and consequently sclerotome progenitor numbers, by preventing paraxial mesoderm from adopting an intermediate/lateral plate mesodermal fate in the Noggin-deficient state. 24618599_follistatin induces hair wave propagation and its level decreases in aging mice. 24630125_Oviducts of tghFST315 mice failed to coil, the myometrium was disorganised, endometrial gland number was reduced and oviducts and uteri contained abundant leukocytes. These studies show that follistatin is crucial to postnatal oviductal-uterine development and function. 25052838_This study assesses the regulation of activin A and follistatin in a murine myocardial ischaemia-reperfusion model. 25827793_OVOL1-regulated Fst and SFRP1 affect hair inductive potency of neonatal dermal cells. 28205525_These data suggest that activin-follistatin regulation may play a role during the development of Experimental autoimmune epididymo-orchitis. 28324027_Fst promotes brown adipocyte characteristics in both white adipose tissue and brown adipose tissue depots in vivo through distinct mechanisms. 28363632_Results showed activin-C and follistatin are differentially expressed during prostate development and suggested that the antagonistic property of follistatin is secondary to the action of activin-C. Study provides evidence to support a role of activin-C in prostate development and provides new insights in the spatiotemporal localization of activins and their antagonists during mouse prostate development. 28471505_Hypergravity affects follistatin levels in muscle through the vestibular system in mice. Follistatin may play some roles in the interactions between muscle and bone metabolism in response to gravity change. 28500669_Surface plasmon resonance analysis showed no significant association between FS288 and AMHC , suggesting that FS288 indirectly regulates AMH signaling. Activin A, a direct target of FS288, did not itself induce reporter activity in P19 cells, but did prevent the FS288-induced increase in AMH signaling. Hence, local concentrations of FS288 and Activin A may influence the response of some cell types to AMH. 28559342_Uterine Fst-cKO mice demonstrate severe fertility defects and deliver only 2% of the number of pups delivered by control females. In Fst-cKO mice, the uterine luminal epithelium does not respond properly to estrogen and progesterone signals and remains unreceptive to embryo attachment by continuing to proliferate and failing to differentiate. 28667125_Data suggest that the level of circulating follistatin is involved in testicular damage due to autoimmune orchitis; these studies were conducted using gene transfer to increase circulating levels of follistatin in experimental model of autoimmune orchitis. 29867232_Fst is a pathological hepatokine that might be targeted for diabetes therapy during hepatic insulin resistance. 30995923_this study identified Sp1 as the critical transcription factor regulating activation of the FST promoter in cav-1 KO MC through binding to a region within 123 bp of the transcription start site. 31184201_our data support a novel role for the antifibrotic protein FST as an ROS scavenger with protective effects both in vitro and in vivo in CKD 31187730_Activin A and its antagonist follistatin as key regulators of hair cell differentiation. 31242421_These results support a model in which an imbalance of activin A and BMP4 signaling underpins the degeneration of postnatal thymic epithelial cell (TEC) maintenance during aging, and its reversal enables the transient replenishment of mature TECs. 31492508_The caveolin-1 regulated protein follistatin protects against diabetic kidney disease. 33420269_miR299a-5p promotes renal fibrosis by suppressing the antifibrotic actions of follistatin. 33678425_Voluntary wheel running with and without follistatin overexpression improves NMJ transmission but not motor unit loss in late life of C57BL/6J mice. 36497076_Follistatin Is a Novel Chemoattractant for Migration and Invasion of Placental Trophoblasts of Mice. 36577057_Follistatin regulates the specification of the apical cochlea responsible for low-frequency hearing in mammals. |
ENSG00000134363 |
FST |
3134.106563 |
0.3901990037 |
-1.357718 |
0.06117129 |
483.247465 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000004198628150485662337381142756128216244339880240353104621385467600135260544110133067050476664497612270810340727333878753891874181920491756373288926148556928070 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000245975015339221550861188643751204009354544448286303330935508422215297152366392962185544793288129076938362835713034282960920450685713794335274420140483391078 |
Yes |
No |
1747.44197867415 |
74.1149815592538 |
4478.33531618614 |
137.992944903945 |
| ENSMUSG00000021806 |
18074 |
Nid2 |
protein_coding |
O88322 |
FUNCTION: Cell adhesion glycoprotein. Might be involved in osteoblast differentiation. It probably has a role in cell-extracellular matrix interactions. |
Basement membrane;Calcium;Cell adhesion;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Methylation;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:18074; |
basement membrane [GO:0005604]; cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell-matrix adhesion [GO:0007160] |
12243745_These findings revealed extensive coregulation of nidogen-1 and -2 expression and much more complementary functions of the two nidogens than previously recognized. 16024816_Nid2 is essential for late stages of lung development and for maintenance and/or integrity of cardiac tissue 17008882_nidogen-2 is equivalent to nidogen-1 in skin-organotypic coculture, and both can promote the development of a functional basement membrane zone 17023412_nidogen-1 and -2 have roles in development of the ectodermal basement membrane of the limb bud 17177854_decreased expression of the entactin-2 gene after treatment of the cells with ent-2-specific siRNA preparation led to reduced expression of the cell cycle inhibitor p21 17616934_The lack of nidogen-1, nidogen-2, or both nidogens, plays no crucial role in the occurrence and localization of laminin-111, collagen type IV, and perlecan in murine tubular renal basement membranes. 18356808_In capillaries, basement membrane formation is severely impaired in the absence of nidogen-1 and -2, showing an irregular, patchy distribution and a dramatically reduced deposition of collagen IV, perlecan, and particularly laminin-411. 18817539_Neuromuscular junctions in mutant mice lacking nidogen-2 appear normal at birth, but become topologically abnormal as they mature 21084308_Basement membrane deposition of nidogen 1 but not nidogen 2 requires the nidogen binding module of the laminin gamma1 chain. 22260998_show that loss of nidogen 2, but not of nidogen 1, promotes lung metastasis of melanoma cells. Histological and ultrastructural analysis of nidogen 1- and 2-deficient lungs did not reveal differences in morphology and ultrastructure of basement membrane 22623588_finding demonstrates that nidogens, although homologous proteins, exert their effect on BM assembly through different binding domains, which may in turn result in alterations of BM structure and functions 25450682_Nidogen-2, a protein shown to be expressed intracellularly and for secretion by pre-adipocytes, was shown to interact, through its globular G3 domain, with TRAP 5a in vitro. 36354004_Nidogen-2 is a Novel Endogenous Ligand of LGR4 to Inhibit Vascular Calcification. |
ENSG00000087303 |
NID2 |
2086.655890 |
2.1281191153 |
1.089579 |
0.08363520 |
167.511630 |
0.00000000000000000000000000000000000002586272746046167315124960832371499711098615541552572046500439854643056068150280247223912405992016141226264025476666574832051992416381835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000027939082601259022302157087272906889483606503257130433696754359504522670632779412999893031986685734691100435611588181927800178527832031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2888.93675141754 |
167.56982438859 |
1357.50707308573 |
59.9167223139914 |
| ENSMUSG00000021835 |
12159 |
Bmp4 |
protein_coding |
P21275 |
FUNCTION: Growth factor of the TGF-beta superfamily that plays essential roles in many developmental processes, including neurogenesis, vascular development, angiogenesis and osteogenesis (PubMed:14973287, PubMed:15206957, PubMed:10049358). Acts in concert with PTHLH/PTHRP to stimulate ductal outgrowth during embryonic mammary development and to inhibit hair follicle induction (PubMed:17301089). Initiates the canonical BMP signaling cascade by associating with type I receptor BMPR1A and type II receptor BMPR2. Once all three components are bound together in a complex at the cell surface, BMPR2 phosphorylates and activates BMPR1A. In turn, BMPR1A propagates signal by phosphorylating SMAD1/5/8 that travel to the nucleus and act as activators and repressors of transcription of target genes. Can also signal through non-canonical BMP pathways such as ERK/MAP kinase, PI3K/Akt or SRC cascades. For example, induces SRC phosphorylation which, in turn, activates VEGFR2, leading to an angiogenic response (By similarity). {ECO:0000250|UniProtKB:P12644, ECO:0000269|PubMed:10049358, ECO:0000269|PubMed:14973287, ECO:0000269|PubMed:15206957, ECO:0000269|PubMed:17301089}. |
Chondrogenesis;Cleavage on pair of basic residues;Cytokine;Developmental protein;Differentiation;Disulfide bond;Extracellular matrix;Glycoprotein;Growth factor;Osteogenesis;Phosphoprotein;Reference proteome;Secreted;Signal |
|
This gene encodes a secreted ligand of the TGF-beta (transforming growth factor-beta) superfamily of proteins. Ligands of this family bind various TGF-beta receptors leading to recruitment and activation of SMAD family transcription factors that regulate gene expression. The encoded preproprotein is proteolytically processed to generate each subunit of the disulfide-linked homodimer. This protein regulates heart development and adipogenesis. Homozygous knockout mice die in utero, while a conditional knockout mouse exhibits defects in heart development. Transgenic mice overexpressing this gene in a neuron-specific manner exhibit a phenotype resembling the rare hereditary connective tissue disease, fibrodysplasia ossificans progressiva. [provided by RefSeq, Jul 2016]. |
mmu:12159; |
cytoplasm [GO:0005737]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; vesicle [GO:0031982]; BMP receptor binding [GO:0070700]; chemoattractant activity [GO:0042056]; co-receptor binding [GO:0039706]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; ameloblast differentiation [GO:0036305]; anatomical structure development [GO:0048856]; anatomical structure formation involved in morphogenesis [GO:0048646]; angiogenesis [GO:0001525]; anterior/posterior axis specification [GO:0009948]; aortic valve morphogenesis [GO:0003180]; blood vessel development [GO:0001568]; blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:0002043]; BMP signaling pathway [GO:0030509]; BMP signaling pathway involved in heart development [GO:0061312]; BMP signaling pathway involved in heart induction [GO:0003130]; BMP signaling pathway involved in nephric duct formation [GO:0071893]; BMP signaling pathway involved in renal system segmentation [GO:0061151]; BMP signaling pathway involved in ureter morphogenesis [GO:0061149]; bone development [GO:0060348]; branching involved in prostate gland morphogenesis [GO:0060442]; branching involved in ureteric bud morphogenesis [GO:0001658]; branching morphogenesis of an epithelial tube [GO:0048754]; bronchus development [GO:0060433]; bud dilation involved in lung branching [GO:0060503]; bud elongation involved in lung branching [GO:0060449]; camera-type eye development [GO:0043010]; camera-type eye morphogenesis [GO:0048593]; cardiac muscle cell differentiation [GO:0055007]; cell death [GO:0008219]; cell fate commitment [GO:0045165]; cell population proliferation [GO:0008283]; cellular response to BMP stimulus [GO:0071773]; cellular response to dexamethasone stimulus [GO:0071549]; cellular response to growth factor stimulus [GO:0071363]; cellular response to oxidative stress [GO:0034599]; chondrocyte differentiation [GO:0002062]; cloacal septation [GO:0060197]; common-partner SMAD protein phosphorylation [GO:0007182]; coronary vasculature development [GO:0060976]; cranial suture morphogenesis [GO:0060363]; deltoid tuberosity development [GO:0035993]; dorsal/ventral neural tube patterning [GO:0021904]; embryonic cranial skeleton morphogenesis [GO:0048701]; embryonic digit morphogenesis [GO:0042733]; embryonic hindlimb morphogenesis [GO:0035116]; embryonic limb morphogenesis [GO:0030326]; embryonic morphogenesis [GO:0048598]; embryonic skeletal joint morphogenesis [GO:0060272]; embryonic skeletal system development [GO:0048706]; embryonic skeletal system morphogenesis [GO:0048704]; endocardial cushion development [GO:0003197]; endochondral ossification [GO:0001958]; endoderm development [GO:0007492]; epithelial cell proliferation [GO:0050673]; epithelial cell proliferation involved in lung morphogenesis [GO:0060502]; epithelial tube branching involved in lung morphogenesis [GO:0060441]; epithelial-mesenchymal cell signaling [GO:0060684]; epithelium development [GO:0060429]; erythrocyte differentiation [GO:0030218]; forebrain development [GO:0030900]; gene expression [GO:0010467]; germ cell development [GO:0007281]; glomerular capillary formation [GO:0072104]; heart development [GO:0007507]; hematopoietic progenitor cell differentiation [GO:0002244]; inner ear auditory receptor cell differentiation [GO:0042491]; intermediate mesodermal cell differentiation [GO:0048392]; kidney development [GO:0001822]; lens induction in camera-type eye [GO:0060235]; lung alveolus development [GO:0048286]; lung development [GO:0030324]; lung morphogenesis [GO:0060425]; lymphoid progenitor cell differentiation [GO:0002320]; macrophage differentiation [GO:0030225]; mammary gland formation [GO:0060592]; membranous septum morphogenesis [GO:0003149]; mesenchymal cell differentiation involved in kidney development [GO:0072161]; mesenchymal cell differentiation involved in renal system development [GO:2001012]; mesenchymal cell proliferation involved in ureter development [GO:0072198]; mesenchymal cell proliferation involved in ureteric bud development [GO:0072138]; mesenchymal to epithelial transition involved in metanephros morphogenesis [GO:0003337]; mesoderm formation [GO:0001707]; mesodermal cell differentiation [GO:0048333]; mesodermal cell fate determination [GO:0007500]; mesonephros development [GO:0001823]; metanephric collecting duct development [GO:0072205]; metanephros development [GO:0001656]; monocyte differentiation [GO:0030224]; negative regulation of apoptotic process [GO:0043066]; negative regulation of branch elongation involved in ureteric bud branching by BMP signaling pathway [GO:0072097]; negative regulation of branching involved in ureteric bud morphogenesis [GO:0090191]; negative regulation of cell cycle [GO:0045786]; negative regulation of cell death [GO:0060548]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cell proliferation involved in heart morphogenesis [GO:2000137]; negative regulation of chondrocyte differentiation [GO:0032331]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of extrinsic apoptotic signaling pathway [GO:2001237]; negative regulation of gene expression [GO:0010629]; negative regulation of glomerular mesangial cell proliferation [GO:0072125]; negative regulation of glomerulus development [GO:0090194]; negative regulation of immature T cell proliferation in thymus [GO:0033088]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of mesenchymal cell proliferation involved in ureter development [GO:0072200]; negative regulation of metanephric comma-shaped body morphogenesis [GO:2000007]; negative regulation of metanephric S-shaped body morphogenesis [GO:2000005]; negative regulation of miRNA transcription [GO:1902894]; negative regulation of mitotic nuclear division [GO:0045839]; negative regulation of myoblast differentiation [GO:0045662]; negative regulation of oligodendrocyte differentiation [GO:0048715]; negative regulation of phosphorylation [GO:0042326]; negative regulation of prostatic bud formation [GO:0060686]; negative regulation of striated muscle tissue development [GO:0045843]; negative regulation of T cell differentiation in thymus [GO:0033085]; negative regulation of thymocyte apoptotic process [GO:0070244]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural tube closure [GO:0001843]; neuron fate commitment [GO:0048663]; odontogenesis [GO:0042476]; odontogenesis of dentin-containing tooth [GO:0042475]; organ induction [GO:0001759]; osteoblast differentiation [GO:0001649]; outflow tract morphogenesis [GO:0003151]; outflow tract septum morphogenesis [GO:0003148]; ovarian follicle development [GO:0001541]; pericyte cell differentiation [GO:1904238]; pharyngeal arch artery morphogenesis [GO:0061626]; pituitary gland development [GO:0021983]; podocyte development [GO:0072015]; positive chemotaxis [GO:0050918]; positive regulation of apoptotic process [GO:0043065]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of bone mineralization [GO:0030501]; positive regulation of branching involved in lung morphogenesis [GO:0061047]; positive regulation of branching involved in ureteric bud morphogenesis [GO:0090190]; positive regulation of calcium ion transport into cytosol [GO:0010524]; positive regulation of cardiac muscle fiber development [GO:0055020]; positive regulation of cardiac neural crest cell migration involved in outflow tract morphogenesis [GO:1905312]; positive regulation of cartilage development [GO:0061036]; positive regulation of cell death [GO:0010942]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cell proliferation involved in outflow tract morphogenesis [GO:1901964]; positive regulation of collagen biosynthetic process [GO:0032967]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of endothelial cell apoptotic process [GO:2000353]; positive regulation of endothelial cell differentiation [GO:0045603]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of epidermal cell differentiation [GO:0045606]; positive regulation of epithelial cell differentiation [GO:0030858]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of epithelial to mesenchymal transition [GO:0010718]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of hepatocyte differentiation [GO:0070368]; positive regulation of kidney development [GO:0090184]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of mesenchymal stem cell proliferation [GO:1902462]; positive regulation of miRNA maturation [GO:1903800]; positive regulation of miRNA transcription [GO:1902895]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of neuron projection development [GO:0010976]; positive regulation of ossification [GO:0045778]; positive regulation of osteoblast differentiation [GO:0045669]; positive regulation of p38MAPK cascade [GO:1900745]; positive regulation of pathway-restricted SMAD protein phosphorylation [GO:0010862]; positive regulation of protein binding [GO:0032092]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of SMAD protein signal transduction [GO:0060391]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of store-operated calcium channel activity [GO:1901341]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of vascular endothelial growth factor receptor signaling pathway [GO:0030949]; post-embryonic development [GO:0009791]; prostate gland morphogenesis [GO:0060512]; prostatic bud formation [GO:0060513]; protein localization to nucleus [GO:0034504]; pulmonary artery endothelial tube morphogenesis [GO:0061155]; pulmonary valve morphogenesis [GO:0003184]; regulation of branching involved in prostate gland morphogenesis [GO:0060687]; regulation of cartilage development [GO:0061035]; regulation of cell differentiation [GO:0045595]; regulation of cell fate commitment [GO:0010453]; regulation of endothelial cell proliferation [GO:0001936]; regulation of ERK1 and ERK2 cascade [GO:0070372]; regulation of gene expression [GO:0010468]; regulation of mesodermal cell differentiation [GO:1905770]; regulation of morphogenesis of a branching structure [GO:0060688]; regulation of odontogenesis of dentin-containing tooth [GO:0042487]; regulation of pathway-restricted SMAD protein phosphorylation [GO:0060393]; regulation of protein import into nucleus [GO:0042306]; regulation of smooth muscle cell differentiation [GO:0051150]; regulation of smooth muscle cell proliferation [GO:0048660]; renal system process [GO:0003014]; secondary heart field specification [GO:0003139]; skeletal system development [GO:0001501]; SMAD protein signal transduction [GO:0060395]; smooth muscle cell differentiation [GO:0051145]; smooth muscle tissue development [GO:0048745]; specification of animal organ position [GO:0010159]; specification of segmental identity, mandibular segment [GO:0042305]; specification of ureteric bud anterior/posterior symmetry by BMP signaling pathway [GO:0072101]; steroid hormone mediated signaling pathway [GO:0043401]; telencephalon development [GO:0021537]; telencephalon regionalization [GO:0021978]; tendon cell differentiation [GO:0035990]; trachea development [GO:0060438]; trachea formation [GO:0060440]; transcription by RNA polymerase II [GO:0006366]; type B pancreatic cell development [GO:0003323]; ureter epithelial cell differentiation [GO:0072192]; ureter smooth muscle cell differentiation [GO:0072193]; ureteric bud development [GO:0001657]; vasculature development [GO:0001944] |
11722794_Haploinsufficient Bmp4 ocular phenotypes include anterior segment dysgenesis with elevated intraocular pressure. 11850175_Derivation in culture of primordial germ cells from cells of the mouse epiblast: phenotypic induction and growth control by Bmp4 signalling. 12013553_involved in cusp formation and differentiation of ameloblasts in the occlusal region of molars 12054672_Dissection of promoter control modules that direct Bmp4 expression in the epithelium-derived components of hair follicles. 12119341_BMP4 is expressed by thymic stroma and inhibits the proliferation of CD4(-)CD8(-) double-negative (DN) thymocytes and their differentiation to the CD4(+)CD8(+) double-positive (DP) stage in vitro. 12141440_BMP4 has a role in mouse metanephric development 12142028_Dlx5 is essential for vestibular morphogenesis through a BMP4-mediated pathway 12151088_BMP-4 transferred by electroporation can induce in vivo and in situ ectopic bone formation in skeletal muscle. 12163415_Msx1 thus controls a genetic hierarchy involving BMP and Shh signals that regulates the growth of the anterior region of palate during mammalian palatogenesis 12223402_Bmp-4 signaling is required for development of the ciliary body 12361961_we provide evidence in the mouse that bone morphogenetic protein 4 (Bmp4) is required independently in two different sites for node morphogenesis and for LR patterning. 12421925_The BMP4 pathway functions in thymic homeostasis by regulating T cell lineage commitment and differentiation. 12552124_BMP4 has a role pituitary prolactinoma pathogenesis through a Smad/estrogen receptor crosstalk 12618141_BMPs are likely important mediators of astrocyte development in vivo. 12619129_BMP4 is an essential mediator of chondrogenesis in the developing otic capsule in situ. 12631064_Depending on local context, BMP4 has several biological actions on the morphogenesis of different portions of the excretory system, namely, the development of the ureterovesical junction, the ureter, and the kidney. 12648489_BMP4 is not expressed in the early tongue, but is precisely coexpressed with Shh in papillary placodes, which serve as a signaling center for both gustatory and papillary development. 12665593_Twisted gastrulation (TSG) acts as both a BMP-4 agonist in skeletogenesis and a BMP-4 antagonist in T-cell development 12687003_Syndromic craniosynostoses may be the result of inappropriate downregulation of BMP antagonist noggin expression 12694205_important role of bmp4 in mediating the fusion process, possibly through apoptosis of cells in the putative site of fusion, during midfacial morphogenesis 12766166_BMP4 is a mechanosensitive, inflammatory factor playing a critical role in early steps of atherogenesis in the lesion-prone areas. 12781691_Recombinant Shh protein upregulates this mesodermal biological marker. 12781692_Lack or excess of O-sulfates prevents FGF10 activation of Bmp4. 12920023_BMP4 acts upstream of FGF7/FGF10 in the regulation of early T-cell development and BMP4 acts primarily through the thymic stroma, thereby altering the thymic microenvironment and affecting thymopoiesis. 12944425_These data identify a signaling pathway essential for digit regeneration, in which Msx1 functions to regulate BMP4 production. 12975322_identification Bmp4 as a signal from the myocardium directly mediating atrioventricular septation 14586015_Exogenous Bmp4 reduced interneuron production by embryonic telencephalon stem cell explants and inhibited induction by exogenous Shh; inhibiting BMP signaling with dominant-negative BMP receptor Ib increased interneuron number even with Shh blocked 14654219_BMP4 controls Dlx5 expression in dental mesenchyme, and functions upstream to both Dlx5 and Cbfa1 to regulate alveolar bone formation during tooth development 14681194_Tsg-/-;Bmp4+/- compound mutants die at birth and display holoprosencephaly, first branchial arch and eye defects 14702176_Bmp4 is down-regulated in tretinoin-induced cleft palate formation. 14973275_role of target-derived BMP signaling in development of sensory ganglia and the sensory innervation of the skin was examined in transgenic animals that overexpress either the BMP inhibitor noggin or BMP4 15017003_BMP-4 expression occurs exclusively in the intravillus mesenchyme; data indicate that intestinal BMP signaling represses de novo crypt formation and polyp growth 15070745_A previously uncharacterized function for Bmp4 in vascular remodeling of the bronchial arch arteries. 15075392_major effect of bone morphogenetic protein 4 on the self-renewal of embryonic stem cells is accomplished by means of the inhibition of mitogen activated protein kinase pathways 15218525_the amino-terminal basic amino-acid motif of mature BMP4 controls long-range activity for dorsoventral patterning of the vertebrate neural tube 15226637_play a crucial role in early cardiomyogenesis 15280210_the inhibitory effects of BMP4 signaling on oligodendroglial lineage commitment are mediated by induction of ID4 and ID2, and their sequestration of both OLIG proteins and E2A proteins 15388638_BMP4 produced in ECs by oscillatory shear sress stimulates ROS release from the nox1-dependent NADPH oxidase leading to inflammation, a critical early atherogenic step. 15466481_BMP-4 signaling in mouse lung is regulated by LTBP-4 and TGF-beta1 15531371_The developmental expression and function of Bmp4 was studied in spermatogenesis and in the maintenance of epididymal integrity. 15531758_Is an important protein in lung morphogenesis. 15581864_data show that bone morphogenetic protein 4 in limb mesoderm regulates apical ectodermal ridge (AER) induction and maturation and implicate signaling from the AER in regulation of digit number and identity. 15591122_BMP4/Madh5-dependent signaling, regulated by hypoxia, initiates the differentiation and expansion of erythroid progenitors in the spleen. 15623804_megalin has a role in neural tube specification, possibly by acting as BMP4 clearance receptor in the neuroepithelium 15673289_BMP-4 plays an important role in the regulation of ureteric branching morphogenesis. 15673572_Mixl1 is required for efficient haematopoiesis and BMP4-induced ventral mesoderm patterning in differentiating ES cells 15716346_distinct roles in lip and palate fusion 15936012_Bmp4 plays role in threshold-specific regulation of Fgf8 transcription in mandibular development 15987774_Gata4 lateral mesoderm enhancer activation requires BMP4, supporting a model in which GATA4 serves as a downstream effector of BMP signaling in the lateral mesoderm. 16059633_BMP4 has roles in endochondral bone formation 16122729_The reduction of Bmp4 dosage results in an extensive rescue of the axial skeleton of Noggin mutant embryos. 16154126_Bmp4 AND Bmp7 can function interchangeably to activate all the essential signaling pathways for growth and morphogenesis of the kidney. 16314487_LIF and BMP signaling generate different astrocytic cell types, and propose that these cells are, respectively, adult progenitor cells and mature astrocytes. 16373339_OAZ can alter the intensity and duration of the BMP4 stimulus through Smad6 16517216_Bmp4 expression during the early stages of thymus and parathyroid organogenesis. 16546160_Bmp-4 gene expression was lost in the absence of Wnt-4 while Wnt-4 signaling led to a rescue of Bmp-4 expression and induction of alpha-smooth muscle actin-positive cells 16547003_BMP4 stimulation promotes Id2 binding to the bHLH transcription factor NeuroD, which is required for the differentiation of pancreatic islet cells. 16651263_the partnership of Pax9 and Msx1 in a signaling pathway that involves Bmp4 determines the fate of the transition from bud to cap stage during tooth development 16712836_All Noggin-/- pups are born with lumbar spina bifida; elevated BMP4 signaling in the mesoderm between the limb buds leads to abnormal somite differentiation and axial skeletal malformation 16769906_Data show that ectodysplasin receptor (Edar)-bone morphogenetic protein (BMP)-4 and -7 signaling and transcriptional interactions are central to generation of the primary hair follicle pattern. 16773658_cell-cell interaction is required before P19-SI cells can undergo a cardiogenic response to BMP 4 16837459_Bone morphogenetic protein-4 is a novel modulator of melanogenesis 16880207_an intracellular gremlin-BMP4 interaction negatively regulates BMP4 activation and secretion 16916928_These studies provide compelling evidence for the participation of BMP-4 in adipocyte lineage determination. 16952347_These studies demonstrate several novel roles for Bone morphogenetic protein signaling and highlight new functions for sialyltransferases in the developing enteric nervous system (ENS). 16960130_Active BMP signaling functions to prevent Epithelial stem cells (EP-SC) activation and expansion. Dynamic expression of Noggin, a BMP antagonist, releases EP-SCs from BMP-mediated restriction, leading to EP-SC activation. 17028893_Tbx3 and Bmp4 strongly up- and down-regulate each other in order to control the proliferation of the palatal shelf during secondary palate development. 17071745_mutual interactions between Bmp4 and Tbx3 determine the presumptive dorsoventral boundary and formation of mammary glands in early embryogenesis 17173667_correct level of BMP4 signaling is critical for normal growth of the mammalian embryonic eye 17194222_A threshold level of bone morphogenetic protein (BMP) signaling is required for the onset of chondrogenesis, hence some chondrogenic condensations fail to form in limbs deficient in both BMP2 and BMP4. 17213321_integrate three key determinants of hematopoietic stem cell development; the Scl transcriptional network, Runx1 activity, and the Bmp4/Smad signaling pathway 17222801_We propose that PRDC might serve as a mediator to antagonize BMP-4 signaling by Wnt. 17244894_Inhibition of MAPK signaling and mineralocorticoid receptor expression by BMP-4 and BMP-7 may be involved in ameliorating renal damage due to mesangial proliferation caused by aldosterone. 17275231_Quantitative testing of the vestibulo-collic reflex, which helps maintain head stability, demonstrated that Bmp4(+/-) mice that exhibit circling behavior have a poor response in the yaw axis, consistent with semicircular canal dysfunction. 17284534_SCF and hypoxia synergize with BMP4 to promote the expansion and differentiation of stress BFU-E during the recovery from acute anemia. 17301089_PTHrP signaling sensitizes the mammary mesenchyme to the actions of BMP4, triggering outgrowth of the mammary buds and inducing MSX2 expression, which, in turn, leads to lateral inhibition of hair follicle formation within the developing nipple sheath. 17339028_results provide evidence that an autocrine, positive BMP4-BMPR1A signaling feedback loop in beta cells plays a key role in maintaining and enhancing glucose-stimulated insulin secretion 17350185_These data support a model in which TNF-alpha-induced RelA interacts with SP1 to bring about transcriptional repression of Bmp4 gene. 17472960_RGMa facilitates the use of ActRIIA by endogenous BMP2 and BMP4 ligands that otherwise prefer signaling via BMPRII and that increased utilization of ActRIIA leads to generation of an enhanced BMP signal 17500053_The presence of cathepsin H and BMP4 in lung or other developing organs suggest that cathepsin H regulates BMP4 proteolysis in morphogenesis. 17522159_initiation of metanephric kidney development requires the reduction of BMP4 activity by the antagonist gremlin 1 in the mesenchyme, which in turn enables ureteric bud outgrowth and establishment of autoregulatory GDNF/WNT11 feedback signalling 17569791_The tumors generated were serially transplantable as long as they retained BMP4 expression. 17618464_Interactions between NOGGIN and bone morphogenetic protein in periodontal ligaments show higher levels of proliferation. 17644284_In vivo BMP-4 acts as transition factor in transition of primordial to primary follicle. 17698011_Retrograde BMP4 signaling regulates trigeminal sensory neuron identities and the formation of precise face maps. 17706674_Endothelial Bmp4 expression is upregulated after carotid ligation in vivo, and furthermore, activating the BMP signaling cascade results in decreased smooth muscle cell proliferation and migration. 17722066_These results indicate that the BMP4 are differentially expressed in adult spinal cord and are up-regulated during EAE. 17881493_Data show that Bmps, in particular Bmp4, are crucial for vascular tube formation, that Bmp4 expression requires Foxf1, and that hedgehog proteins activate Foxf1 expression in the mesoderm. 17942544_Friend virus induces the bone morphogenetic protein 4 (BMP4)-dependent stress erythropoiesis pathway in the spleen, which rapidly amplifies target cells, propagating their infection and resulting in acute splenomegaly. 17960174_We have identified BMP-4 as an important factor for prenatal Kit-negative melanoblasts just prior to entering the Kit-dependent cycle of melanogenesis 17982705_BMP-4 acts synergistically with the chondrocyte-conditioned medium causing a further increase in VSMC calcification 18177426_Bmp4 was inducing posterior and extraembryonic mesoderm at the expense of anterior primitive streak cells along with FGF. 18202659_periodic expression of bone morphogenetic protein 2 (Bmp2) and Bmp4 in the dermis regulates cyclic hair regeneration 18233958_Bmp4 mRNA is reduced approximately twofold in Gata2(fGN)/(/fGN) mice (as in Bmp4 null heterozygotes), and that GATA-2 trans-activates a Bmp4 first intron element-directed reporter plasmid in co-transfection assays. 18292351_BMP4 is not required for bone formation and function in the limb 18295595_These data suggest that icariin exerts its potent osteogenic effect through induction of Runx2 expression, production of BMP-4 and activation of BMP signaling. 18310445_BMPs are important modulators of activin and/or GnRH-stimulated gonadotrophin synthesis and release 18374325_These data suggest that steady state erythropoiesis is unable to generate sufficient erythrocytes to maintain the rapid growth of the embryo leading to the induction of the BMP4 dependent stress erythropoiesis pathway. 18393306_Results suggest that reciprocal changes in sonic hedgehog and BMP4 signaling by retinoic acid may regulate prostatic bud initiation. 18398851_increased expression of BMP4 mRNA within the dentate gyrus of the hippocampus may contribute to the decrease in cell proliferation in APPswe/PS1DeltaE9 tg mice 18404215_Bmp4 is essential for the formation of all three cristae and their associated canals. 18628975_analysis of a transcriptionally-based regulation of BMP2/4 signaling by HDACs both in vivo and in vitro that is critical for neurogenesis in the ganglionic eminences 18629540_Mesenchymally expressed BMP4 regulates Hertwig's epithelial root sheath development by preventing elongation and maintaining cell proliferation. BMP4 may be useful as a root-formation regulatory agent in tissue-engineering applications. 18634775_In Tp63 mutant mice, clefting defects result from increased Bmp4 signaling acting antagonistically on Fgf8 and Shh, led to a reduction in mesenchymal cell proliferation and increased cell death in specific regions of the facial processes. 18643848_BMP-4 signalling activates AHF/THH transcription in hair follicles. 18657169_Bone morphogenetic protein 4 signaling regulates development of the anterior visceral endoderm in the mouse embryo. 18692041_We found that Bmp signaling is prominently present in the anterior foregut where the tracheal primordium originates and targeted ablation of Bmp4 (Bmp4(cko)) resulted in a loss-of-trachea phenotype. 18758906_These data indicate that cAMP-PKA/CREB/CRE signaling potentially enhances BMP4-induced transcription through the BRE in the promoter of the BMP-responsive gene through a PKA-mediated pathway. 18776146_Tshz3 is required for proximal ureteric smooth muscle cell differentiation downstream of SHH and BMP4. 18924235_BMP4 is required in the anterior heart field and its derivatives for endocardial cushion remodeling, outflow tract septation, and semilunar valve development 18927434_Hedgehog induces the expression of BMP4 19021035_Bmp4 expression and activity in glucocorticoid-arrested MC3T3-E1 osteoblasts: Smad signaling, not alkaline phosphatase activity, predicts rescue of mineralization. 19091945_In silico mutations in key signaling molecules Fgf10 and Bmp4 resulted in altered patterning of the developing pancreas that were in general agreement with in vivo data 19116164_These results identify numerous BMP4-dependent developmental processes that are also very sensitive to BMP2 dosage, thus revealing novel functions of Bmp2. 19159624_Results show that an ancient, mesoderm-specific cis-regulatory element resides nearly 50 kb 5' to mouse Bmp4. 19210962_Results suggest that apical ectodermal ridge expression of Bmp2 and Bmp4 is required for digit and dorsal-ventral patterning but surprisingly not for limb outgrowth. 19221194_Study demonstrates that as molar concentrations of Bmper exceed Bmp4, Bmper dynamically switches from an activator to an inhibitor of Bmp4 signaling. 19299468_NDST1-dependent heparan sulfate regulates BMP2/4 signaling and internalization in lung development. 19375646_the BMP-4-dependent stress erythropoiesis pathway regulates extramedullary stress erythropoiesis, which occurs primarily in the murine spleen or in the case of splenectomized mice, in the adult liver. 19391114_Results suggest that the dosage of both BMP2 and 4 is critical for functional heart formation during embryogenesis and after birth. 19393343_Data present a detailed study of the developmental expression profiles of three BMP ligands (Bmp2, Bmp4, Bmp7) and three BMP receptors (Bmpr1a, Bmpr1b, and BmprII), as well as noggin, in the early embryo during the initial steps of organogenesis. 19432819_Data show that a specific threshold of Wnt and BMP and their synergy seemed necessary for providing the guiding cues in orchestrating mesoderm induction and subsequent cardiomyogenesis. 19454350_Bmp4 signaling is required for regulation of the uroepithelial stem response to uropathogenic E. coli infection. 19463786_These results suggest that BMP4 and Noggin co-modulate neurogenesis. 19592576_BMP4 signaling through type I receptors regulates the differentiation of two major medial ganglionic eminence-derived interneuron populations and defines their relative numbers in the cortex. 19608221_Bmp-4, Shh and Wnt-5a gene expressions may act together on the epithelial-mesenchymal interactions during early mouse craniofacial development, such as odontogenesis, neuronal development, jaw ossification, palatogenesis and tongue formation. 19653318_Hoxa2 also repressed the expression of its downstream targets Bmp4 within the palate. 19661459_Heat shock protein 70 enhances vascular bone morphogenetic protein-4 signaling by binding matrix Gla protein. 19737815_Axin2 is a negative regulator in bone remodeling in adult mice and regulates psteoblast differentiation through the beta-catenin-BMP2/4 signaling pathway in osteoblasts. 19759357_BMP4 is a critical component of the hematopoietic microenvironment that regulates both hematopoietic stem cell number and function 19782673_Studies suggest a model of molecular interactions integrating FGF10 and BMP4 signaling during rugae formation within the RGZ. 19795511_PTHrP and BMP4 regulate MMP2 gene expression and MMP2 activity in mesenchymal cells. 19819988_analysis of BMP-4 induction of arrest and differentiation of osteoblast-like cells via p21 CIP1 and p27 KIP1 regulation 19841742_BMP4 signaling is a fundamental mechanism linking environmental exposure with changes in cognitive function and cellular properties in the hippocampus 19854269_Sonic hedgehog is involved in formation of the ventral optic cup by limiting Bmp4 expression to the dorsal domain 19890878_Trophoblast stem cell FRS2alpha mediates activation of the ERK pathway to enhance Cdx2 expression in response to FGF4. Cdx2 in turn binds to an FGF4-responsive enhancer element of the promoter region of Bmp4, leading to production and secretion of Bmp4. 20042595_Results demonstrate that Dies1 is required for BMP4/Smad1 signaling cascade. 20042692_Bmp2 and Bmp4 exert opposing roles in hypoxic pulmonary hypertension 20043899_results show that processes of cell specification and morphogenesis during thymus-parathyroid development are independently controlled, and suggest a specific temporal and spatial role for BMP4-mediated epithelial-mesenchymal interactions 20056821_inhibited basal and CRH-induced ACTH production and proopiomelanocortin transcription in corticotrope AtT20 cells 20059955_Study shows that tendon attachment to the skeleton regulates bone ridge formation and identify SCX regulation of Bmp4 as the underlying molecular mechanism. 20063299_Noggin is likely to regulate Bmp4 in the cochlea. 20096288_Bmp4 has a role in the pathogenesis of myocardial infarction 20097443_The pathway analysis revealed a reduction of the hedgehog target Foxf1 in yolk sac mesoderm of keratin(-/-) embryos, and subsequent reduction of BMP-4. 20206312_Data observed BMP4 mutant dysmorphogenic odontoblasts that failed to properly elongate and differentiate, resulting in failed differentiation process leads to permanent loss of dentin thickness, apparent enlarged pulp chambers in the molars. 20231279_findings suggest that the Smad signaling pathway may play a dual role in the BMP-induced conversion of myoblasts to osteoblastic cells 20233579_the inner ear and circling behavior in Bmp4 heterozygotes were analysed. 20336610_Data show dynamic changes of Bmp4-expressing cells of different stage hair follicles. 20501701_BMP4-dependent chondrogenesis in early limb mesenchyme is regulated by TGFbeta1 signal pathway. 20504958_Results suggest that once neural differentiation is initiated, BMP4 cannot inhibit it; the BMP4-sensitive window is a crucial time during which BMP4 switches its function from maintaining ESC pluripotency to promoting non-neural differentiation. 20585586_data show that hypoxia plays a key role in initiating the BMP4 dependent stress erythropoiesis pathway by regulating BMP4 expression 20705941_Gremlin-mediated BMP inhibition results in activation of epithelial cells and transient fibrosis, but also induction of epithelium-protective FGF10 20711995_Studies indicate that BMP and IL-3 signaling pathways are critical for the growth and potential of embryonic HSCs. 20724703_BMP4/reactive oxygen species/COX-2 cascade is important in the maintenance of endothelial dysfunction in hypertension 20845426_Results demonstrate that FGF8 induces Tbx22 in the early face while BMP4 represses and thus restricts its expression. This regulation is conserved between chicken and mouse. 20883686_beta-catenin, Lef1 & Tcf1 are needed for Bmp4 expression maintaining Shh expression in the dental ectoderm. The number of incisors from one placode can be varied through local changes of a mesenchymal signaling circuit involving these proteins. 20942907_perigemmal BMP4 cells appear mitotically silent in both fungiform and circumvallate taste papillae, as we do not find evidence of their active proliferation using cell cycle 21060151_BMP4-dependent stress erythropoiesis pathway was required and specific for erythroid short-term radioprotection following bone marrow transplantation. BMP4 signaling promoted the development of three populations of stress erythroid progenitors. 21068310_BMP4 upregulates markers of the future outer sulcus and downregulates marker genes of Kolliker's organ in cochlear organ cultures. Our results suggest BMP signaling is required for patterning sensory and nonsensory tissue in the mammalian cochlea. 21123999_A conditional gene inactivation approach was applied to test the hypothesis that Bmp4 expressed from the Tbx1 expression domain plays a critical role for normal development of outflow tract and pharyngeal arch arteries. 21411747_Data suggest that down-regulation of BMP4 expression by TNF in CNV and mechanisms established might be useful for defining novel targets for AMD therapy. 21471216_BMP4 may act as an upstream regulatory molecule for the process of ECM accumulation in DN and thereby reveals a new aspect of the molecular mechanisms involved in DN. 21482757_Fstl1 modulates lung development and alveolar maturation, in part, through BMP4 signaling. 21518886_Smad1-dependent BMP4 signaling is developmentally regulated and governs axonal growth in dorsal root ganglion neurons. 21532395_BMP signaling is essential to cochlear sensory formation and hair cell differentiation 21546760_The objective of these studies was to gain further insight into the molecular relationship between Pax9, Msx1, and Bmp4 in dental mesenchyme. 21599637_Data show that both Wnt3a and BMP4 inhibit oligodendrocyte (OL) differentiation in vitro, and that when BMP signalling is blocked, neither Wnt3a nor BMP4 have inhibitory effects on OL differentiation. 21673106_down-regulation of Kruppel-like factor-4 (KLF4) by microRNA-143/145 is critical for modulation of vascular smooth muscle cell phenotype by transforming growth factor-beta and bone morphogenetic protein 4. 21717271_Different doses of bone morphogenetic protein 4 promote the expression of early germ cell-specific gene in bone marrow mesenchymal stem cells. 21732075_Antigen presenting cell (APC) candidates from developing mouse cerebellum are characterized in vitro and eventually found to be glial fibrillary acidic protein (GFAP)and bone morphogenetic protein (BMP)4, surviving markers of the original cells. 21810858_BMP4 protein expression in the mouse female reproductive tract is of importance in folliculogenesis. 21896629_immature AVC expansion in wkm mutants is rescued by depleting Bmp4, indicating that Tmem2 restricts bmp4 expression to delimit the AVC primordium during cardiac development 21898398_Provide evidence that CCN3 enhances BMP-4 expression and bone nodule formation in osteoblasts, and that the integrin receptor, ILK, p38, JNK, and AP-1 signaling pathways may be involved. 21959449_Laser capture microdissection collected clean populations of primary lip and palate epithelial cells destined to fuse. Transcripts of Bmp-4/Alk-3 and Tgfbeta-2/Alk-5 were restricted to epithelial seam of fusion site, and epithelium of the prefusion site. 21964441_presence of myotubes inhibits Bmp4 expression in proliferating myoblasts through transcriptional regulation, although the expression is intrinsically increased with time of culture 21978004_Data show that Bmp4, Bmp7, Gremlin and Twisted gastrulation (Twsg1) are all expressed in the thymus and expression was clearly different for each gene investigated. 22027433_BMP4 and laminin synergise to induce visceral endoderm differentiation of XEN cells. 22064324_BMP4 causes endothelial cells (ECs) apoptosis through activation of caspase-3 in a ROS/p38MAPK/JNK-dependent signaling cascade. 22219353_Data support the hypothesis that Bmp4 signaling regulates craniofacial skeletal development by balancing self-renewal and differentiation pathways in cranial neural crest progenitors. 22253730_Rubie is the gene mutated in Ecl mice, that it is involved in regulating inner ear expression of Bmp4, and that aberrant Bmp4 expression contributes to the Ecl phenotype. 22266311_these suggest that cell stretch suppresses the BMP4 induction of MSC adipogenesis potentially via upregulating ERK but not through the downregulation of Smad or p38. 22350174_the expression of Zeb1 and Zeb2 in the dental mesenchyme was recovered by Bmp4 signaling 22392094_an anti-angiogenic role for BMP4 in laser-induced CNV, mediated by direct inhibition of MMP-9 and indirect inhibition of VEGF. 22474292_Scx and Id1 are involved in the BMP4-Smad1-SMA signal transduction pathway besides the TGFbeta1-Smad1-SMA signaling pathway and modulate phenotypic changes in MCs in diabetic nephropathy. 22493062_GATA-2 expression in the caudal urogenital mesenchyme as directed by the UG4 enhancer is crucial for proper development of the urinary tract and that its regulation of BMP4 expression is a critical aspect of this function 22556407_Heparan sulfate is required for embryonic stem cell pluripotency, in particular lineage specification into mesoderm through facilitation of FGF and BMP signaling. 22658483_CHD7 may directly regulate Bmp4 expression by binding with an enhancer element downstream of the Bmp4 locus. 22662233_Bmp2, Bmp4 and Bmp7 are co-required in the mouse AER for normal digit patterning but not limb outgrowth 22701669_Pitx homeoprotein family members bind a specific site in the Bmp4 incisor epithelium limb bud enhancer element that is necessary for its activity in vivo. 22711842_Progenitor cells of the first and second heart fields depend on cardiac-specific transcription factors for their differentiation and Wnt/beta-catenin and Bmp act downstream of Notch/RBPJ at this developmental stage. 22751012_A regulatory loop involving Dies1 and miR-125a controls BMP4 signaling in mouse embryonic stem cells. 22895561_The glycosaminoglycans chains of biglycan (BGN) promote BGN-assisted BMP-4 function. 22988836_Adding exogenous BMP4 to culture medium could differentiate pluripotent stage-specific embryonic antigen-1+ cells isolated from an adult tissue into gamete precursors, primordial germ cells. 23019195_study found BMP4 is constitutively expressed in the peribiliary stroma and endothelial cells of the liver and that expression is decreased after hepatectomy; evidence provided that BMP4 signals through Alk3 to inhibit liver regeneration 23212987_Effects of BMP4 on mouse primordial follicle growth were assayed by follicle count and TUNEL labeling. The expression of phospho-SMAD1/5/8, Sohlh2, and c-kit were measured by immunohistochemistry, RT-PCR, and Western blotting. 23243277_In HSPCs, BMP4 directly regulates Integrin-alpha4 expression through SMAD-independent p38 MAPK-mediated signaling. 23248151_we conclude that BMP4 is a mediator and novel therapeutic target for pathological cardiac hypertrophy. 23250216_Bmp4 signaling suppresses tooth developmental inhibitors in the tooth mesenchyme, including Dkk2 and Osr2, and synergizes with Msx1 to activate mesenchymal odontogenic potential for tooth morphogenesis and sequential tooth formation 23344708_Foxc1 sets a threshold for the Bmp-dependent activation of Msx2, thus controlling the differentiation of osteogenic precursor cells and the rate and pattern of calvarial bone development. 23344953_COCH is a part of the downstream target network of BMP4 signaling and serves as another important effector to fine-tune mouse embryonic stem cell fates. 23349763_Data indicate that agmatine treatment improved neurological and histological outcomes through modulating the BMP- 2/4/7 expressions following spinal cord injury. 23383068_short-term expression of constitutively active Akt in the endothelium of mice results in non-anemic stress erythropoiesis in the spleen 23388637_These findings provide a genetic and metabolic basis for BMP4's role in altering insulin sensitivity by affecting white adipose tissue development. 23396188_Ctnnb1 in UGS basal epithelial cell differentiation, prostatic bud initiation and bud spacing 23472198_BMP4 is a peripherally-derived factor that may regulate the survival of motor neurons. 23620745_BMP-4 and TGF-beta1 were expressed in the transitional epithelium, lamina propia and muscularis mucosa. 23626735_A novel mechanism of transcription is involved in pancreatic expression of the Bmp4 gene. 23675417_BMP-4 maintains epithelial polarity via intracellular signaling from basolaterally localized BMP receptors. 23720046_Msx1 and Tbx2 antagonistically regulate Bmp4 expression during the bud-to-cap stage transition in tooth development. 23843349_Bone morphogenetic protein 2 (BMP2), BMP4, and secreted frizzled related protein 2 are principal effectors of quiescence of adult mammalian retinal stem cells. 23869763_50 ng/ml BMP-4 seemed to induce mASCs to differentiate into the adipo-lineage. 23880664_BMP-4 suppresses kisspeptin-induced GnRH expression by reducing ERK signaling activity. 23911203_Endogenous Bmp4 in preadipocytes is indispensable for the onset of the adipogenic program, and may help to maintain the preadipocytic state during adipocyte differentiation. 24052814_Megf8 is involved in mediating BMP4 signaling and guidance of developing trigeminal ganglia axons. 24080013_The results support a model in which BMP4 induces MITF expression in pluripotent stem cells and EDN3 subsequently promotes differentiation of these MITF exp |
ENSG00000125378 |
BMP4 |
1873.825693 |
0.4771193462 |
-1.067578 |
0.10977419 |
92.571824 |
0.00000000000000000000064925229660290920736824089548970916343365541419146455567423630028667247415796737186610698699951171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000286649593676971409228797030726373734881591042720715530522576641736520741687854751944541931152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1256.9111483778 |
95.7302662913569 |
2634.3747290611 |
148.686315830185 |
| ENSMUSG00000021846 |
93834 |
Peli2 |
protein_coding |
E9QPQ6 |
FUNCTION: E3 ubiquitin ligase catalyzing the covalent attachment of ubiquitin moieties onto substrate proteins. {ECO:0000256|PIRNR:PIRNR038886}. |
Phosphoprotein;Proteomics identification;Reference proteome;Transferase;Ubl conjugation pathway |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000256|PIRNR:PIRNR038886}. |
|
mmu:93834; |
ubiquitin protein ligase activity [GO:0061630]; protein polyubiquitination [GO:0000209]; regulation of Toll signaling pathway [GO:0008592] |
12370331_Pellino-2 is a tissue-restricted component of a signaling pathway that couples the mouse pelle-like kinase/IL-1 receptor-associated kinase protein to IL-1- or lipopolysaccharide-dependent signaling. 22669975_Pellino 2 plays a critical role for TLR/IL-1R-mediated post-transcriptional control. 29674674_The functions of Pellino2 in the NLRP3 pathway are dependent on Pellino2 FHA and RING-like domains, with Pellino2 promoting the ubiquitination of NLRP3 during the priming phase of activation. 32738922_Dibutyl phthalate promotes juvenile Sertoli cell proliferation by decreasing the levels of the E3 ubiquitin ligase Pellino 2. 34588221_Dendritic Cell-Specific Role for Pellino2 as a Mediator of TLR9 Signaling Pathway. 35809381_Pellino2 accelerate inflammation and pyroptosis via the ubiquitination and activation of NLRP3 inflammation in model of pediatric pneumonia. |
ENSG00000139946 |
PELI2 |
88.287513 |
0.3177321432 |
-1.654117 |
0.56992812 |
7.517309 |
0.00611089096064992409046423560425864707212895154953002929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0220675891684731229069882374460576102137565612792968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
41.7853893282973 |
16.9397811905817 |
131.511369632796 |
38.6914540012424 |
| ENSMUSG00000021903 |
78754 |
Galnt15 |
protein_coding |
Q9D2N8 |
FUNCTION: Catalyzes the initial reaction in O-linked oligosaccharide biosynthesis, the transfer of an N-acetyl-D-galactosamine residue to a serine or threonine residue on the protein receptor. Although it displays a much weaker activity toward all substrates tested compared to GALNT2, it is able to transfer up to seven GalNAc residues to the Muc5AC peptide, suggesting that it can fill vicinal Thr/Ser residues in cooperation with other GALNT proteins. Prefers Muc1a as substrate (By similarity). {ECO:0000250}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lectin;Manganese;Membrane;Metal-binding;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. |
|
mmu:78754; |
Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; transport vesicle [GO:0030133]; carbohydrate binding [GO:0030246]; metal ion binding [GO:0046872]; polypeptide N-acetylgalactosaminyltransferase activity [GO:0004653]; protein O-linked glycosylation [GO:0006493] |
Human_homologues 20800603_Observational study of gene-disease association. (HuGE Navigator) |
ENSG00000131386 |
GALNT15 |
113.843884 |
0.3219777070 |
-1.634967 |
0.28240123 |
33.042463 |
0.00000000901679280789854160438336706578574264447922814724734053015708923339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001101393649157262088710381364373147583535228477558121085166931152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
53.717877804987 |
10.582317605754 |
166.837258119284 |
23.3844141428284 |
| ENSMUSG00000021965 |
219114 |
Ska3 |
protein_coding |
Q8C263 |
FUNCTION: Component of the SKA1 complex, a microtubule-binding subcomplex of the outer kinetochore that is essential for proper chromosome segregation. The SKA1 complex is a direct component of the kinetochore-microtubule interface and directly associates with microtubules as oligomeric assemblies. The complex facilitates the processive movement of microspheres along a microtubule in a depolymerization-coupled manner. In the complex, it mediates the microtubule-stimulated oligomerization. Affinity for microtubules is synergistically enhanced in the presence of the ndc-80 complex and may allow the ndc-80 complex to track depolymerizing microtubules. {ECO:0000250|UniProtKB:Q8IX90}. |
Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Cytoskeleton;Kinetochore;Microtubule;Mitosis;Phosphoprotein;Reference proteome |
|
|
mmu:219114; |
centrosome [GO:0005813]; cytosol [GO:0005829]; kinetochore [GO:0000776]; mitotic spindle [GO:0072686]; outer kinetochore [GO:0000940]; spindle microtubule [GO:0005876]; cell division [GO:0051301]; chromosome segregation [GO:0007059]; mitotic cell cycle [GO:0000278]; regulation of microtubule polymerization or depolymerization [GO:0031110] |
22336914_The Ska3 subunit was only localized on spindle microtubules from the Pro-MI to MII stages in mouse oocyte meiosis and knockdown by Morpholino injection into mouse oocytes resulted in spindle movement defects and enlarged polar bodies. 34533066_SKA3, negatively regulated by miR-128-3p, promotes the progression of non-small-cell lung cancer. |
ENSG00000165480 |
SKA3 |
321.838648 |
2.2541572797 |
1.172588 |
0.15744980 |
54.846529 |
0.00000000000013031870627050690439783450275486301650658405026916852875729091465473175048828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000028851955434772690171181456156180236021814577984656580156297422945499420166015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
443.802822022549 |
48.5310187519299 |
196.881923912485 |
16.518361700846 |
| ENSMUSG00000022025 |
16840 |
Cnmd |
protein_coding |
Q9Z1F6 |
FUNCTION: Bifunctional growth regulator that stimulates the growth of cultured chondrocytes in the presence of basic fibroblast growth factor (FGF) but inhibits the growth of cultured vascular endothelial cells. May contribute to the rapid growth of cartilage and vascular invasion prior to the replacement of cartilage by bone during endochondral bone development (By similarity). Inhibits in vitro tube formation and mobilization of endothelial cells (By similarity). Plays a role as antiangiogenic factor in cardiac valves to suppress neovascularization. {ECO:0000250, ECO:0000269|PubMed:16980969}. |
Chondrogenesis;Cleavage on pair of basic residues;Developmental protein;Differentiation;Disulfide bond;Extracellular matrix;Glycoprotein;Membrane;Reference proteome;Secreted;Transmembrane;Transmembrane helix |
|
|
mmu:16840; |
endomembrane system [GO:0012505]; extracellular region [GO:0005576]; membrane [GO:0016020]; cartilage development [GO:0051216]; endothelial cell morphogenesis [GO:0001886]; negative regulation of angiogenesis [GO:0016525]; negative regulation of endothelial cell proliferation [GO:0001937]; negative regulation of vascular endothelial growth factor receptor signaling pathway [GO:0030948] |
12192060_Role of ChM-I in cartilage and eye development 12509461_chondromodulin I is a bone remodeling factor 15022325_Chondromodulin I suppresses T cell responses and synovial cell proliferation, implying that this cartilage matrix protein has a therapeutic potential in rheumatoid arthritis. 16980969_chondromodulin-I has a pivotal role in maintaining valvular normal function by preventing angiogenesis that may lead to valvular heart diseases 18239943_The expression domains of ChM-I and TeM during vertebrate development incorporate the typical avascular regions of the mesenchymal tissues. 18793763_results suggest that Chm-I is involved in hypertrophic maturation of periosteal chondrocytes. Although a direct effect of Chm-I on bones is still unclear, bony callus formation was increased while external cartilaginous callus decreased in Chm1(-/-) mice 21391200_chondromodulin 1 stabilizes the chondrocyte phenotype by supporting chondrogenesis but inhibiting chondrocyte hypertrophy and endochondral ossification. 21849085_the inhibitory action of ChM-I on trophoblast migration and invasion, implying the potential role of the ChM-I expression in decidual cells for the regulated tissue remodeling and angiogenesis at feto-maternal interface. 24710035_intact 20-25 kDa ChM-I is stored as a component of extracellular matrix in the avascular cartilage zones, but it is inactivated by a single N-terminal proteolytic cleavage in the hypertrophic zone of growth-plate cartilage 25251892_These findings indicated that Chm-I was an indispensable factor for ectopic cartilage regeneration and the maintenance of cartilage homeostasis. 36795722_Chondromodulin is necessary for cartilage callus distraction in mice. |
ENSG00000136110 |
CNMD |
21.676828 |
0.1175031879 |
-3.089228 |
0.77090260 |
15.662802 |
0.00007569842257729927643538497816777521620679181069135665893554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004621396283356402740448432009401358300237916409969329833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.02599484330396 |
2.25513340347068 |
34.2628563018378 |
12.1348900845508 |
| ENSMUSG00000022089 |
57784 |
Bin3 |
protein_coding |
Q9JI08 |
FUNCTION: Involved in cytokinesis and septation where it has a role in the localization of F-actin. {ECO:0000250}. |
Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Reference proteome;Septation |
|
|
mmu:57784; |
actin cytoskeleton [GO:0015629]; cytoplasm [GO:0005737]; actin cortical patch localization [GO:0051666]; division septum assembly [GO:0000917]; endocytosis [GO:0006897]; myoblast migration involved in skeletal muscle regeneration [GO:0014839]; plasma membrane tubulation [GO:0097320]; protein localization [GO:0008104]; regulation of lamellipodium assembly [GO:0010591]; skeletal muscle fiber development [GO:0048741]; skeletal muscle tissue regeneration [GO:0043403]; unidimensional cell growth [GO:0009826] |
23872330_Bin3 forms a complex with Rac1 and Cdc42, Rho GTPases involved in actin polymerization, which are known to be essential for myotube formation. |
ENSG00000147439 |
BIN3 |
377.146586 |
0.3488389656 |
-1.519367 |
0.33575858 |
18.034836 |
0.00002168994243784959453165590848033872362066176719963550567626953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001505153545391002176644101862024172078236006200313568115234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
190.828793121445 |
44.8136072101935 |
547.039786153369 |
95.5880606659261 |
| ENSMUSG00000022144 |
14573 |
Gdnf |
protein_coding |
P48540 |
FUNCTION: Neurotrophic factor that enhances survival and morphological differentiation of dopaminergic neurons and increases their high-affinity dopamine uptake. |
Alternative splicing;Cleavage on pair of basic residues;Disulfide bond;Glycoprotein;Growth factor;Reference proteome;Secreted;Signal |
|
This gene encodes a secreted ligand of the TGF-beta (transforming growth factor-beta) superfamily of proteins. Ligands of this family bind various TGF-beta receptors leading to recruitment and activation of SMAD family transcription factors that regulate gene expression. The encoded preproprotein is proteolytically processed to generate each subunit of the disulfide-linked homodimer. The recombinant form of this protein, a highly conserved neurotrophic factor, was shown to promote the survival and differentiation of dopaminergic neurons in culture, and was able to prevent apoptosis of motor neurons induced by axotomy. This protein is a ligand for the product of the RET (rearranged during transfection) protooncogene. Homozygous knockout mice for this gene exhibit defects in kidney development and neonatal death. This gene encodes multiple protein isoforms that may undergo similar proteolytic processing. [provided by RefSeq, Aug 2016]. |
mmu:14573; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; receptor complex [GO:0043235]; glial cell-derived neurotrophic factor receptor binding [GO:0030116]; growth factor activity [GO:0008083]; protein homodimerization activity [GO:0042803]; receptor ligand activity [GO:0048018]; receptor tyrosine kinase binding [GO:0030971]; transforming growth factor beta receptor binding [GO:0005160]; branching involved in ureteric bud morphogenesis [GO:0001658]; cell population proliferation [GO:0008283]; cellular response to dexamethasone stimulus [GO:0071549]; commissural neuron axon guidance [GO:0071679]; dorsal spinal cord development [GO:0021516]; embryonic organ development [GO:0048568]; enteric nervous system development [GO:0048484]; mesenchymal to epithelial transition involved in metanephros morphogenesis [GO:0003337]; metanephros development [GO:0001656]; mRNA stabilization [GO:0048255]; negative regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001240]; negative regulation of neuron apoptotic process [GO:0043524]; nervous system development [GO:0007399]; neural crest cell migration [GO:0001755]; neural crest cell migration involved in autonomic nervous system development [GO:1901166]; neuron differentiation [GO:0030182]; neuron projection development [GO:0031175]; organ induction [GO:0001759]; peripheral nervous system development [GO:0007422]; peristalsis [GO:0030432]; positive regulation of branching involved in ureteric bud morphogenesis [GO:0090190]; positive regulation of cell differentiation [GO:0045597]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of mesenchymal to epithelial transition involved in metanephros morphogenesis [GO:0072108]; positive regulation of monooxygenase activity [GO:0032770]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of ureteric bud formation [GO:0072107]; postganglionic parasympathetic fiber development [GO:0021784]; postsynaptic membrane organization [GO:0001941]; regulation of dopamine uptake involved in synaptic transmission [GO:0051584]; regulation of gene expression [GO:0010468]; regulation of morphogenesis of a branching structure [GO:0060688]; regulation of semaphorin-plexin signaling pathway [GO:2001260]; regulation of ureteric bud formation [GO:0072106]; response to wounding [GO:0009611]; sympathetic nervous system development [GO:0048485]; transforming growth factor beta receptor signaling pathway [GO:0007179]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169]; ureteric bud development [GO:0001657]; ureteric bud formation [GO:0060676] |
11774071_Gdnf haploinsufficiency causes Hirschsprung-like intestinal obstruction and early-onset lethality in mice 11822887_Continuous, early developmental GDNF expression exerts physiological effects on newly differentiated, immature dopamine neurons that differ from those observed on more mature and adult DA neurons 11878293_non-neuronal role for glial cell line-derived neurotrophic factor in tooth development 12372284_GDNF acts as a peripheral signal to induce PEA3 expression in specific motor neuron pools, thereby regulating both cell body position and muscle innervation. 12399307_We provide evidence that activation of the receptor tyrosine kinase RET by glial cell line-derived neurotrophic factor (GDNF) is required for the directional migration of ENS progenitors towards and within the gut wall. 12668632_Data show that glial cell line-derived neurotrophic factor (GDNF) and neurturin are important for different aspects of the enteric nervous system development. 12746870_Expression of GFAP is implicated with the production of GDNF to a degree that confers neuroprotection after an excitotoxic or metabolic insult. 12783789_Wnt11 and Ret/Gdnf cooperate in coordinating ureteric branching by maintaining a balance of Wnt11-expressing ureteric epithelium and Gdnf-expressing mesenchyme in developing kidney 12797383_Data suggest that the locus coeruleus-noradrenergic system of glial cell line-derived neurotrophic factor (Gdnf) +/- mice is impaired and suggest that GDNF plays an important role in continued maintenance of this neuronal system throughout life. 12884298_GDNF/GFRalpha1 are involved in the survival and differentiation of double negative immature thymocytes. 14960346_development of the noradrenergic locus coeruleus-hippocampal pathway is dependent on the presence of GDNF, most likely through a target-derived neurotrophic function 15201220_epithelial Wnt11, mesenchymal Gdnf and stromal Fgf7 signalling is coordinated by sprouty proteins during kidney development, which regulates ureteric branching 15327782_Six2 activates Gdnf expression 15389609_GDNF in type-II cells may exert trophic actions on type-I, -II, and -III taste bud cells by binding to their GFR alpha 1 receptors. 15582775_these data indicate that fusimotor neurons depend upon GDNF for survival during development. 15671854_Experiments using GDNF mutant mice confirm that the increase in GDNF mRNA level, not reduction in mRNA level of inducible nitric oxide synthase, correlates with restoration activity of locomotor function following spinal cord injury. 15691764_the function of Spry1 is to modulate GDNF/RET signaling in the Wolffian duct, ensuring that kidney induction is restricted to a single site 15748846_GDNF signaling via glial cell line derived neurotrophic factor family receptor alpha 1 (GFRalpha1) in the developing brain is to promote differentiation of ventral precursors into GABAergic cells, enhancing their neuronal morphology and motility. 15890330_Testing the requirement for GDNF as a paracrine chemoattractive factor by altering its site of expression during excretory system development. 15905075_Jagged1 and GDNF/Ret/GFRalpha1 interact and have a role in regulating ureteric budding and branching 15937482_We show Sprouty2 to be a negative regulator of GDNF for the neonatal development or survival of enteric nerve cells. 15950786_Induction of GNDF was found during acute encephalitis. Neurotrophic factors play an important role in neuronal survival and recovery after acute injury to the central nervous system. 15960316_GDNF- and GFRalpha1-immunoreactive taste bud cells after denervation vanished following the disappearance of the taste buds and reappeared at the same time as the taste buds reappeared 16237148_GDNF-mediated RET signaling is critical for the fate of undifferentiated spermatogonia and that abnormalities in this pathway may contribute to male infertility and testicular germ cell tumors. 16364262_reduced GDNF levels augment reward and increased GDNF levels attenuate reward 16380265_Taken together, these data demonstrate that GDNF is a direct chemoattractant factor for neuroblast cells migrating along the RMS and support the participation of NCAM during this guidance process. 16467260_Thus, through Numb, GDNF might inhibit differentiation and allows the maintenance of the stem cell pool in the mouse seminiferous epithelium. 16540576_Enhanced expression of GDNF in the skin can change mechanical sensitivity of IB4-positive nociceptive afferents. This may occur through enhanced expression of specific types of channel proteins. 17022962_The positive (GDNF) and inhibitory (Sprouty1) signals have to be finely balanced throughout renal development to prevent hypoplasia or cystic hyperplasia. 17030186_GDNF induced a significant increase in NPY messenger RNA and protein expression in primary enteric neurons and the release of NPY into the culture medium 17103416_Gdnf haploinsufficiency did not cause any detectable change in the rate of neuronal differentiation of enteric neural crest-derived cells 17184739_In conclusion, cultured midbrain dopamine neurons survive in the absence of GDNF, and the addition of GDNF improved dopamine nerve fiber formation - possibly as an indirect effect of astrocytic stimulation. 17222400_GDNF promotes spermatogonial stem cell proliferation through activation of members of the Src kinase family, and these kinases exert their action through a PI3K/Akt-dependent pathway to up-regulate N-myc expression. 17356005_Partial reduction in the expression of the GDNF potentiates METH self-administration, enhances motivation to take METH, & increases drug-primed & cue-induced reinstatement of extinguished METH-seeking behavior. 17430172_Treatment of Down's syndrome cortical neurons with rodent ADNF-9 results in a significant increase in neuronal survival as well as reduction of degenerative morphological changes. 17537792_results thus place nephronectin and alpha8beta1 integrin in a pathway that regulates Gdnf expression and is essential for kidney development 17574550_Results identify common and unique pathways that regulate Etv5 and Gdnf mRNA in Sertoli cells. 17597063_GDNF up-regulation of Bcl6b, Erm, and Lhx1 expression through Src family kinase signaling is a key component of the intracellular mechanism for spermatogonial stem cells self-renewal. 17962702_GDNF induces CREB/ATF-1 family member phosphorylation and c-fos transcription via the Ras/ERK1/2 pathway to promote the proliferation of spermatogonial stem cells 18003772_analysis of glial cell-derived neurotrophic factor independent ureteric bud outgrowth from the Wolffian duct 18003856_GDNF was cytoprotective against oxidative stress-induced apoptotic death of mesenchymal stem cells. 18006159_Prokineticin-1 (Prok-1) works coordinately with glial cell line-derived neurotrophic factor (GDNF) to mediate proliferation and differentiation of enteric neural crest cells. 18234170_Completion of Meiosis I of preovulatory oocytes and facilitation of preimplantation embryo development by GDNF are reported. 18305247_This study demonstrated that the period during which spindle-innervating MNs require GDNF for survival is restricted to early neonatal development. 18420832_TGF-beta with neurturin and persephin are required for the induction of dopaminergic neurons, whereas GDNF is required for regulating and/or maintaining a differentiated neuronal phenotype 18445767_Several signaling pathways including p38, JNK, Akt and ERK molecules seem to play an important role in this neuroprotection by GDNF. 18536709_GDNF is indispensable for adult catecholaminergic neuron survival and also show that, under physiologic conditions, downregulation of a single trophic factor can produce massive neuronal death. 18551627_Gdnf-deficient embryos show no difference in the appearance or relative area of the ganglia occupied by brain-derived fatty acid binding protein (B-FABP) immunoreactive cells in the sympathetic chain compared with wild-type littermates. 18650792_AT1R-mediated inhibition of the Spry1 gene increases c-Ret tyrosine kinase activity leading to upregulation of its downstream target Wnt11, which induces GDNF in adjacent mesenchyme causing focal bursts of ureteric bud tip cell proliferation 18657279_GDNF enhances both sympathetic and parasympathetic neurite outgrowth, and stimulates tyrosine hydroxylase-positive cell migration. 18703596_GDNF-secreting NIH3T3-TG cells in combination with molecular imaging could be a platform for developing therapeutic tools for acute SCI. 18816441_compromised mouse neurons have increased survival response to GDNF. 18824086_These data point towards a cooperative role of TGF-beta2 and GDNF with regard to promotion of survival within the peripheral motor and sensory systems investigated. 18845535_analysis of how GDNF.GFR alpha 1 can mediate cell adhesion and how heparin might inhibit GDNF signaling through RET 19005738_Results suggest that GDNF is likely secreted both with and without processing by furin-like proteases, and that the pro-domain and C-terminal cysteines of GDNF play important roles in its processing and secretion in cultured astrocytes and C6 cells. 19302086_Endogenous GDNF negatively regulates the rewarding effect of ethanol and ethanol-drinking behaviors after a period of abstinence. 19721135_FGF2-induced Gdnf expression is mediated by the induction of Egr-1 through activation of the ERK and JNK/Elk-1 signaling pathways 19898483_Etv4 and Etv5 are required downstream of GDNF and Ret for kidney branching morphogenesis 19944698_GDNF promotes synaptic communication in cultured myenteric neurons. It also up-regulates 5-HT(3a)-receptor expression via modulation of Kv channel activity. 19954518_Neonatal gamma-motorneurons (MNs) are selectively dependent on muscle spindle-derived GDNF. Deletion of GDNF expression from muscle spindles results in the selective elimination of gamma-MNs with preservation of the spindle and its sensory innervation. 20084103_Fgf10 cooperates with Gdnf to promote Kidney morphogenesis, and can compensate for the loss of Gdnf/Ret in the absence of Spry1. 20107080_Increasing levels of the Ret ligand glial cell line-derived neurotrophic factor (GDNF) in mice causes alterations in ENS structure and function that are critically dependent on the time and location of increased GDNF availability. 20422010_GDNF-mediated c-RET kinase activation is associated with the pathogenesis of malignant melanoma 20448145_GDNF stimulates increased expression of transcription factors including Ngn3 during the embryonic stages of pancreas development. 20542089_GDNF (unlike neurotrophins) does not acutely modulate transmitter release during the developmental process of synapse elimination nor as the neuromuscular junctions matures. 20597970_These findings implicate GDNF in the control of epileptogenesis and offer a possible mechanism explaining lesion asymmetry in mesial temporal lobe epilepsy. 20682772_Protein-tyrosine phosphatase SHP2 contributes to GDNF neurotrophic activity through direct binding to phospho-Tyr687 in the RET receptor tyrosine kinase. 20739562_In cell lines and primary neurons, proteins encoded by pre-(alpha)pro-GDNF and pre-(beta)pro-GDNF complementary DNAs localize to Golgi compartment and secretory vesicles. 20816800_Protein kinase A regulates GDNF/RET-dependent but not GDNF/Ret-independent ureteric bud outgrowth from the Wolffian duct. 20943767_Lower nephron number in GDNF+/- mice is associated with marked early glomerular structural changes, in particular lower capillary supply, reduced podocyte density, and a thickened glomerular basement membrane. 20963849_Both GDNF and hepatic growth factor (HGF) have significant neurotrophic effects, but only HGF can promote neurogenesis, angiogenesis, and synaptogenesis and inhibit fibrosis in brains after transient middle cerebral artery occlusion. 21144620_Altogether, these data support an important role for GDNF in the regulation of uptake, synthesis, and metabolism of DA during aging. 21239429_Results implicate Ret/GDNF as a potent pleiotropic factor able to influence pituitary physiology during a period of high plasticity. 21262472_Results suggest that the histone modifications and DNA methylation of the Gdnf promoter have crucial roles in the control of behavioral responses to chronic stress. 21289216_In the tissue culture of Gdnf-deficient kidney primordium, exogenous GDNF and alternative bud inducers (FGF7 and follistatin) restored Vsnl1 expression. Hence, Vsnl1 characterizes the tip of the ureteric bud epithelium regardless of the inducer. 21420941_Addition of motor neurons to skeletal muscle cells in vitro reduces the secretion of GDNF by skeletal muscle. 21684216_GDNF exerts analgesic effects on neuropathic pain by acting on the central terminals of uninjured dorsal root ganglion neurons and/or on the spinal cells targeted by these neurons 21816850_ETV5 was found to be a critical downstream regulator of GDNF signaling that mediated the expression of several known spermatogonial stem cell self-renewal related genes, including Bcl6b and LIM homeobox. 22015719_Shp2 mediates not only GDNF/Ret but also signaling by other receptor tyrosine kinases in branching morphogenesis of the embryonic kidney. 22102605_we suggest that NANOS2 acts downstream of GDNF signaling to maintain undifferentiated state of spermatogonial stem cells. 22143971_GDNF, besides its crucial role in the self-renewal of stem cells also functions in the differentiation of chained undifferentiated spermatogonia. 22174794_These data provide direct evidence of regionally defined patch-like GDNF-positive signal. 22232066_These results provide the first direct proof that GDNF acutely regulates the number of spermatogonial stem cells in the normal adult testis. 22248285_A AT rich region in the GDNF 3' UTR have a suppressive role its gene expression. 22262884_In this article showed that GDNF production in mouse striatum develops in early postnatal life and is essentially confined to neurons. 22473739_Targeted overexpression of GDNF in the muscles of Myo-GDNF mice did not improve motor recovery after botulinum toxin type A chemodenervation. 22499581_demonstrate functionally antagonistic eicosanoid receptor subtype regulation of innate immunity-induced astrocyte GDNF expression 22670840_analysis of neuroprotection by GDNF in the ischemic brain [review] 22743091_Neuroprotective effects of carotid body transplantation in an MPTP model of Parkinson disease depends on the level of GDNF expression. 22863354_This tsudy demonistrated the role of GDNF for embryonic Schwann cells migration. 22998873_This study identifies a regulator of midline crossing and reveals interplays between Semaphorin and gdnf signaling during axon guidance. 23040482_spermatogonial stem cell chemotactic factor in vitro and in vivo 23290934_studies further support that long-term changes related to the dysfunction of the nigrostriatal pathway are influenced by GDNF expression 23333276_SorLA acts as sorting receptor for the GDNF/GFRalpha1 complex, directing it from the cell surface to endosomes. 23344256_injection of GL261 mouse glioma cells with GDNF knockdown by shRNA into mouse brains resulted in reduced tumor expansion and improved survival as compared to injection of control cells. 23426908_we show that GDNF is capable of promoting survival of 30% sympathetic precursors dissociated at embryonic day 13 23590664_NF-kappaB p65/p52 signaling mediated the effects of GDNF on Bcl-2 and Bcl-w expressions 23903190_GDNF is required for neural colonization of the pancreas. 23934644_A role is found for proGDNF signaling in brains from aging and Parkinson's disease animal models. 24105724_The data suggest that genetically defined cross-talk between neurotrophic factors and the brain serotonin system underlies the variability in behavioral response to GDNF. 24107967_These findings suggest that pleiotropic function of GDNF is at least in part governed by modulating levels of intracellular activation of GDNF downstream pathways 24726488_Study highlights role of GDNF on striatal organization, both on DARPP-32-positive neurons and tyrosine hydroxylase (TH)-positive innervation; and importance of GDNF expression in striatum for TH-positive neuron viability when connected to striatum 25025809_The transcriptional start site and the promoter activity of the 5'-flanking region of GDNF have been identified. 25036711_GDNF expression was upregulated upon radiation therapy in submandibular glands. 25165119_These data suggest that there are significant changes in the responses to GDNF as spermatogonial stem cells give rise to progenitor spermatogonia. 25181385_expression induced by testosterone in peritubular myoid cells 25448159_This hypothesis offers a dramatically different explanation for the etiology of sporadic Parkinson's disease as a manifestation of acquired resistance to GDNF. 25549043_GDNF signaling in the urogenital sinus increases proliferation. 25655216_Results show that in the inflamed intestine, smooth muscle proliferation supports the enteric nervous system , and thus its own re-innervation, by expression of GDNF 25822020_Parkin and the RET signaling cascade converge to control mitochondrial integrity and thereby properly maintain substantia nigra pars compacta dopaminergic neurons and their innervation in the striatum. 25944479_This study demonstrated that decrease in mRNA level of GDNF in brain in microgravity. 26564715_Mice overexpressing GDNF had significantly reduced P62 protein levels suggestive of accelerated autophagy. They also had reduced PPAR-gamma and CD36 gene expression and protein levels, and lower expression of mRNA coding for enzymes involved lipogenesis. 26592967_The data of this study suggested that short-term exposure to hyperoxic conditions can affect the regulation and expression of BDNF potentially leading to alterations in neural development. 26681446_Our results reveal the role of GDNF in nigrostriatal dopamine system postnatal development and adult function, and highlight the importance of correct spatial expression of GDNF 26706106_Our results show the existence of two subpopulations of peptidergic nociceptors characterized by the presence of CGRP, one expressing BDNF (plus SP), the other expressing GDNF (plus SST), suggesting a different role for these two neurotrophic factors in the discrimination of specific painful stimuli modalities. 26831079_The Gdnf cKO males sired up to two litters but became infertile due to collapse of spermatogenesis and loss of undifferentiated spermatogonia 27119228_Data show that NEDL2 regulates GDNF/Ret/Akt pathway depends on its Nedd8 ligase activity rather than ubiquitin ligase activity. 27148690_Six2 mediates the protective effects of GDNF on damaged DA neurons by regulating Smurf1 expression. 27255992_GDNF signals were able to induce the stratified aggregate formation of GFRalpha1-positive undifferentiated spermatogonia 27288458_In SOD1(G93A) spinal cords, we verified a strict correlation in the expression of the TNFalpha, TNFR1 and GDNF triad at different stages of disease progression. Yet, ablation of TNFR1 completely abolished GDNF rises in both SOD1(G93A) astrocytes and spinal cords, a condition that accelerated motor neuron degeneration and disease progression 27545711_identified IFNg, Neurturin (Nrtn), and glial-derived neurotrophic factor (GDNF) as ligands with unexpected roles in promoting neurogenic differentiation of Neural Precursor Cells in vivo. 27607574_Ret is essential to mediate GDNF's neuroprotective and neuroregenerative effect in a Parkinson disease mouse model. 27707798_The GDNF-GFRalpha1 complex is essential for proper hippocampal circuit development. 28408662_Our study provides, for the first time, a global analysis of phosphorylation events in spermatogonial progenitor cells (SPCs) in response to GDNF, and we have identified activation of mTORC1 signaling through ERK kinase-mediated phosphorylation of multiple sites of raptor protein as an important pathway for SPC proliferation. 28506996_Using an organ culture system for prostate development and Ret mutant mice, we demonstrate that RET-mediated GDNF signaling in UGS increases proliferation of mesenchyme cells and suppresses androgen-induced proliferation and differentiation of prostate epithelial cells, inhibiting prostate development. 28645787_These results suggest that chronic overexpression of GDNF in brain astrocytes exerts an opposing influence on nigrostriatal dopamine metabolism and neurotransmission. 29183737_Cross-platform single cell analysis of kidney development shows stromal cells express Gdnf. 29440301_GDNF promotes self-renewal by blocking differentiation and not by promoting proliferation. 29564810_lack of Kir6.2 promoted neuronal differentiation via inhibiting the downregulation of glia cell line-derived neurotrophic factor (GDNF), which negatively related to the level of microRNA-133b. 29913111_These results imply an important role of GDNF in restoring restrictive blood-nerve barrier function in vivo 30089897_A modest elevation of endogenous GDNF levels improves motor function but does not induce adverse behavioral outcomes. 30488198_GDNF displays antioxidant properties and protects neurons from oxidative stress 30617464_Study shows that MiR-33-5p, through its inverse regulation on glial cell-derived neurotrophic factor gene, modulates anesthesia-induced neural apoptosis in dorsal root ganglion cells. 30651543_Study utilizing mouse kidney cultures and time-lapse imaging provided quantitative experimental support that a ligand-receptor-based Turing mechanism, implemented via GDNF-RET signaling, specifies the areas of outgrowth during kidney branching morphogenesis. The positive feedback between GDNF and WNT11 enables the dense packing of the ureteric buds. 30923332_Development of the urogenital system is regulated via the 3'UTR of GDNF. 31119595_Cinnamon and its Metabolite Protect the Nigrostriatum in a Mouse Model of Parkinson's Disease Via Astrocytic GDNF. 31205031_GDNF attenuates inflammation-induced impairment of Impaired intestinal epithelial barrier function caused by the loss of DSG2 through p38 MAPK-dependent phosphorylation of cytokeratin. 31848242_Increased GDNF/GFRalpha1 signaling to sensory neurons from ischemia/reperfusion-affected muscle directly modulated nociceptive-like behaviors and increased exercise-mediated reflexes and group III/IV muscle afferent sensitization. Muscle GDNF signaling to neurons may play an important dual role in nociception and sympathetic reflexes and could provide a therapeutic target for treating complications from ischemic injuries. 31875542_GDNF and GFRalpha1 Are Required for Proper Integration of Adult-Born Hippocampal Neurons. 31930748_Role of Glial Cell Line-Derived Neurotrophic Factor in the Maintenance of Adult Mesencephalic Catecholaminergic Neurons. 32021964_Muscle stem cell renewal suppressed by Gas1 can be reversed by GDNF in mice. 32513820_GDNF drives rapid tubule morphogenesis in a novel 3D in vitro model for ADPKD. 32588893_Renal pathology in a mouse model of severe Spinal Muscular Atrophy is associated with downregulation of Glial Cell-Line Derived Neurotrophic Factor (GDNF). 32808420_Acute stress disrupts intestinal homeostasis via GDNF-RET. 33314792_TNF-alpha inhibits GDNF levels in Sertoli cells, through a NF-kappaB-dependent, HES1-dependent mechanism. 33672854_Intestinal Epithelial Barrier Maturation by Enteric Glial Cells Is GDNF-Dependent. 34032268_Postnatal prolongation of mammalian nephrogenesis by excess fetal GDNF. 34896518_Boosting effect of testosterone on GDNF expression in Sertoli cell line (TM4); comparison between TM3 cells-produced and exogenous testosterone. 35053395_Tumoral Neuroligin 1 Promotes Cancer-Nerve Interactions and Synergizes with the Glial Cell Line-Derived Neurotrophic Factor. 35798698_Bimodal regulation of axonal transport by the GDNF-RET signalling axis in healthy and diseased motor neurons. 36055989_Astrocytic SARM1 promotes neuroinflammation and axonal demyelination in experimental autoimmune encephalomyelitis through inhibiting GDNF signaling. 36077134_Overexpression of Brain- and Glial Cell Line-Derived Neurotrophic Factors Is Neuroprotective in an Animal Model of Acute Hypobaric Hypoxia. 36151131_Glial cell derived neurotrophic factor prevents western diet and palmitate-induced hepatocyte oxidative damage and death through SIRT3. |
ENSG00000168621 |
GDNF |
23.379227 |
0.3447711178 |
-1.536289 |
0.57908002 |
6.981145 |
0.00823729113680284411036502945080428617075085639953613281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0284152172116622324382628761441083042882382869720458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.7918338963828 |
4.86121780664004 |
34.2019191540131 |
9.87669968195878 |
| ENSMUSG00000022146 |
18414 |
Osmr |
protein_coding |
A0A0R4J268 |
Human_homologues FUNCTION: Associates with IL31RA to form the IL31 receptor. Binds IL31 to activate STAT3 and possibly STAT1 and STAT5. Capable of transducing OSM-specific signaling events. {ECO:0000269|PubMed:15184896, ECO:0000269|PubMed:8999038}. |
Membrane;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
|
membrane [GO:0016020]; cytokine receptor activity [GO:0004896] |
12814362_Expression of oncostatin M receptor beta in a specific subset of nociceptive sensory neurons. 12855584_regulates hematopoiesis in vivo by stimulating stromal cells as well as hematopoietic progenitors, in particular megakaryocytic and erythrocytic progenitors. 14999682_Mice deficient in OSM receptor showed impaired liver regeneration with persistent parenchymal necrosis after carbon tetrachloride exposure. Recovery of liver mass from partial hepatectomy was also significantly delayed in OSMR(-/-) mice. 15893881_the up-regulation of p-STAT3 and p-CREB may be induced possibly through OSMR in inflammation 16926070_These findings suggest that IL-31 and OSM may thus have redundant functions in the development of OSMRbeta-expressing neurons. 17379762_Bone marrow progenitor cells involved in injured liver regeneration express OSMRB. 18430728_murine OSMR initiates STAT5 activation directly via the receptor bound Janus kinases. Intriguingly, the murine receptor preferentially recruits JAK2, whereas the human receptor seems to have a higher affinity for JAK1. 18641356_IL-6 and Oncostatin M individually affect the profile of leukocyte trafficking 18927239_A dual role for oncostatin M signaling in the differentiation and death of mammary epithelial cells in vivo. 20051625_Bone formation can be stimulated independently of bone resorption and provide new insights into OSMR signaling. 22190112_These data indicate that the transient RANKL induction by intermittent PTH administration, which is associated with its anabolic action, is changed to a prolonged induction in OSMR-deficient osteoblasts, resulting in bone destruction. 23760275_Data indicate that OSM receptor beta subunit-deficient (OSMRbeta(-/-)) mice exhibited phenotypic changes in adipose tissue macrophages (ATMs) to M1, increased proinflammatory cytokines in the adipose tissue, and systemic insulin resistance. 24695736_defects in OSM signaling promote the deterioration of high-fat diet-induced obesity and related metabolic disorders 24767864_OSM signaling via OSMR in synovial fibroblasts has the potential to contribute significantly to joint destruction in inflammatory arthritis. 25103368_In astrocytes but not microglia, phosphorylation of STAT1 and STAT3 occurred in response to OSM, whereas both microglia and astrocytes responded to hyper-IL-6 (IL-6 linked to the soluble IL-6 receptor). 26198770_Oncostatin M and interleukin-31: Cytokines, receptors, signal transduction and physiology. 28258089_OSMR-beta deficiency in macrophages improved high-fat diet-induced atherogenesis and plaque vulnerability 28368383_Osmr expression in healthy mouse colon tissue was detected in endothelial and stromal cells. In agreement with observations of increased OSMR expression in inflamed colon tissue, the number of cells expressing Osmr was markedly increased in the lamina propria of mice with colitis. 29142070_The results suggest that BRG1 and STAT3 coordinately regulate gene clustering and up-regulate Gfap and Osmr transcription in astrocytes. 30134804_The current study suggested that the OSMR gene is highly expressed in human chronic autoimmune urticaria (CAU) skin tissues, and cause the up-regulation of the JAK/STAT3 signaling pathway-related genes. OSMR gene silencing in mice inhibits the activation of the JAK/STAT3 signaling pathway, thereby suppressing the development of CAU. 30431059_The results of the present study provide evidence that the overexpression of miR183 promotes the apoptosis of substantia nigra neurons by inhibiting the expression of OSMR. 32807793_OSMR controls glioma stem cell respiration and confers resistance of glioblastoma to ionizing radiation. 34769079_Murine Oncostatin M Has Opposing Effects on the Proliferation of OP9 Bone Marrow Stromal Cells and NIH/3T3 Fibroblasts Signaling through the OSMR. 34921158_Heterocellular OSM-OSMR signalling reprograms fibroblasts to promote pancreatic cancer growth and metastasis. 35765036_Oncostatin M receptor regulates osteoblast differentiation via extracellular signal-regulated kinase/autophagy signaling. |
ENSG00000145623 |
OSMR |
407.532247 |
2.0449324533 |
1.032053 |
0.18077089 |
32.338783 |
0.00000001295026478924614930435131105639426363218547066935570910573005676269531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001533891394010865904428330894204712819828273495659232139587402343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
552.498327591298 |
69.4097946802925 |
270.179255411076 |
25.7963280372405 |
| ENSMUSG00000022206 |
18162 |
Npr3 |
protein_coding |
P70180 |
FUNCTION: Receptor for the natriuretic peptide hormones, binding with similar affinities atrial natriuretic peptide NPPA/ANP, brain natriuretic peptide NPPB/BNP, and C-type natriuretic peptide NPPC/CNP (PubMed:10377427, PubMed:17951249). May function as a clearance receptor for NPPA, NPPB and NPPC, regulating their local concentrations and effects (PubMed:10377427, PubMed:17951249). May regulate diuresis, blood pressure and skeletal development (PubMed:10377427). Does not have guanylate cyclase activity (PubMed:10377427). {ECO:0000269|PubMed:10377427, ECO:0000269|PubMed:17951249}. |
Cell membrane;Chloride;Disease variant;Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:18162; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; chloride ion binding [GO:0031404]; G protein-coupled peptide receptor activity [GO:0008528]; hormone binding [GO:0042562]; identical protein binding [GO:0042802]; natriuretic peptide receptor activity [GO:0016941]; peptide binding [GO:0042277]; peptide hormone binding [GO:0017046]; protein homodimerization activity [GO:0042803]; signaling receptor activity [GO:0038023]; adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway [GO:0007193]; angiogenesis [GO:0001525]; blood vessel remodeling [GO:0001974]; negative regulation of adenylate cyclase activity [GO:0007194]; negative regulation of cold-induced thermogenesis [GO:0120163]; negative regulation of smooth muscle cell proliferation [GO:0048662]; osteoclast proliferation [GO:0002158]; pancreatic juice secretion [GO:0030157]; phosphatidylinositol-mediated signaling [GO:0048015]; phospholipase C-activating G protein-coupled receptor signaling pathway [GO:0007200]; positive regulation of nitric-oxide synthase activity [GO:0051000]; positive regulation of urine volume [GO:0035810]; regulation of blood pressure [GO:0008217]; regulation of osteoblast proliferation [GO:0033688]; response to ischemia [GO:0002931]; signal transduction [GO:0007165]; skeletal system development [GO:0001501]; vasodilation [GO:0042311] |
14704228_CNP has negative chronotropic effect on heart rate, and this effect is mediated by selectively activating NPR-C and reducing L-type Ca2+ current through coupling to Gi protein. 15191892_circulating ANP levels and data from NPR1(-/-) and NPR3(-/-) mice suggest that the ANP pathway may not be involved in the cardiovascular response to caloric restriction 15911073_discussion of neuronal regulation and function of natriuretic peptide receptor C [review] 17951249_Ostn is a naturally occurring ligand of the NPR-C clearance receptor and may act to locally modulate the actions of the natriuretic system in bone by blocking the clearance action of NPR-C, thus locally elevating levels of C-type natriuretic peptide. 19244276_Musclin binds to NPR3 competitively with ANP and may affect ANP concentrations in a local or systemic manner. 20403400_Upregulation of NPR-C mRNA in the kidney and heart of eNOS knockout mice. 21846177_NPR-C is required to control DNA damage-induced p53 levels to maintain embryonic stem cell self-renewal. 21915269_A stratified transcriptomics analysis of polygenic fat and lean mouse adipose tissues identifies Npr3 as novel candidate obesity genes. 22001395_Results demonstrate the opposite effects of NPR-A and NPR-C in LPS-mediated endothelial permeability and lung injury. 25860030_Npr3 is expressed in hard calluses of wild-type mice, suggesting a possible role of CNP signaling in fracture repair, especially in bone remodeling stage. 27740716_this study shows that sensory axon bifurcation is insensitive to increases of C-type natriuretic peptide induced cyclic GMP levels and the guanylate cyclase receptor Npr3 does not have an important scavenging function in this axonal system. 27959934_A model of kyphosis due to a novel NPR3 Tyr209Asn mutation is established. 29409758_It was the aim to investigate the expression of natriuretic peptide receptors NPR-A, NPR-B and NPR-C during adipocyte differentiation (AD), upon stimulation with fatty acids (FA), and in murine and human adipose tissue depots. 30239604_Absence of natriuretic peptide clearance receptor attenuates TGF-beta1-induced selective atrial fibrosis and atrial fibrillation. 30636477_NPRC plays a critical role in the progression of angiotensin II-mediated atrial fibrillation and atrial dysfunction. 31255729_Gene expression of proliferator-activated receptor delta and estrogen-related receptor alpha, which upregulate oxidative metabolism, was increased in the gastrocnemius muscle of NPR-C- mice, irrespective of diet. 33486984_Renal Natriuretic Peptide Receptor-C Deficiency Attenuates NaCl Cotransporter Activity in Angiotensin II-Induced Hypertension. 34528389_Deletion of natriuretic peptide receptor C alleviates adipose tissue inflammation in hypercholesterolemic Apolipoprotein E knockout mice. 34668435_Oxygen and steroids affect the regulatory role of natriuretic peptide receptor-C on surfactant secretion by type II cells. 35943160_Natriuretic peptide receptor-C mediates the inhibitory effect of atrial natriuretic peptide on neutrophil recruitment to the lung during acute lung injury. |
ENSG00000113389 |
NPR3 |
58.877848 |
0.2953504778 |
-1.759500 |
0.36380750 |
23.106431 |
0.00000153277169498392658457982910824579647623977507464587688446044921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000131904963039520734983467767365006295676721492782235145568847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
28.424189027696 |
7.24399809101799 |
96.2388455899055 |
16.982707273937 |
| ENSMUSG00000022221 |
56532 |
Ripk3 |
protein_coding |
Q9QZL0 |
FUNCTION: Serine/threonine-protein kinase that activates necroptosis and apoptosis, two parallel forms of cell death (PubMed:27321907, PubMed:27746097, PubMed:27917412, PubMed:28607035, PubMed:32200799, PubMed:32296175). Necroptosis, a programmed cell death process in response to death-inducing TNF-alpha family members, is triggered by RIPK3 following activation by ZBP1 (PubMed:19590578, PubMed:22423968, PubMed:24012422, PubMed:24019532, PubMed:24557836, PubMed:27746097, PubMed:27819681, PubMed:27819682, PubMed:24095729, PubMed:32200799, PubMed:27321907, PubMed:32296175). Activated RIPK3 forms a necrosis-inducing complex and mediates phosphorylation of MLKL, promoting MLKL localization to the plasma membrane and execution of programmed necrosis characterized by calcium influx and plasma membrane damage (PubMed:24813849, PubMed:24813850, PubMed:27321907). In addition to TNF-induced necroptosis, necroptosis can also take place in the nucleus in response to orthomyxoviruses infection: following ZBP1 activation, which senses double-stranded Z-RNA structures, nuclear RIPK3 catalyzes phosphorylation and activation of MLKL, promoting disruption of the nuclear envelope and leakage of cellular DNA into the cytosol (PubMed:32200799, PubMed:32296175). Also regulates apoptosis: apoptosis depends on RIPK1, FADD and CASP8, and is independent of MLKL and RIPK3 kinase activity (PubMed:27321907). Phosphorylates RIPK1: RIPK1 and RIPK3 undergo reciprocal auto- and trans-phosphorylation (By similarity). In some cell types, also able to restrict viral replication by promoting cell death-independent responses (PubMed:30635240). In response to flavivirus infection in neurons, promotes a cell death-independent pathway that restricts viral replication: together with ZBP1, promotes a death-independent transcriptional program that modifies the cellular metabolism via up-regulation expression of the enzyme ACOD1/IRG1 and production of the metabolite itaconate (PubMed:30635240). Itaconate inhibits the activity of succinate dehydrogenase, generating a metabolic state in neurons that suppresses replication of viral genomes (PubMed:30635240). RIPK3 binds to and enhances the activity of three metabolic enzymes: GLUL, GLUD1, and PYGL (By similarity). These metabolic enzymes may eventually stimulate the tricarboxylic acid cycle and oxidative phosphorylation, which could result in enhanced ROS production (By similarity). {ECO:0000250|UniProtKB:Q9Y572, ECO:0000269|PubMed:19590578, ECO:0000269|PubMed:22423968, ECO:0000269|PubMed:24012422, ECO:0000269|PubMed:24019532, ECO:0000269|PubMed:24095729, ECO:0000269|PubMed:24557836, ECO:0000269|PubMed:24813849, ECO:0000269|PubMed:24813850, ECO:0000269|PubMed:27321907, ECO:0000269|PubMed:27746097, ECO:0000269|PubMed:27819681, ECO:0000269|PubMed:27819682, ECO:0000269|PubMed:27917412, ECO:0000269|PubMed:28607035, ECO:0000269|PubMed:30635240, ECO:0000269|PubMed:32200799, ECO:0000269|PubMed:32296175}. |
3D-structure;Apoptosis;ATP-binding;Cytoplasm;Host-virus interaction;Kinase;Methylation;Necrosis;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase;Ubl conjugation |
|
|
mmu:56532; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; ATP binding [GO:0005524]; identical protein binding [GO:0042802]; NF-kappaB-inducing kinase activity [GO:0004704]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; protein-containing complex binding [GO:0044877]; activation of protein kinase activity [GO:0032147]; amyloid fibril formation [GO:1990000]; apoptotic process [GO:0006915]; cellular response to hydrogen peroxide [GO:0070301]; defense response to virus [GO:0051607]; execution phase of necroptosis [GO:0097528]; I-kappaB kinase/NF-kappaB signaling [GO:0007249]; lymph node development [GO:0048535]; necroptotic process [GO:0070266]; necroptotic signaling pathway [GO:0097527]; positive regulation of intrinsic apoptotic signaling pathway [GO:2001244]; positive regulation of ligase activity [GO:0051351]; positive regulation of necroptotic process [GO:0060545]; positive regulation of necrotic cell death [GO:0010940]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of oxidoreductase activity [GO:0051353]; positive regulation of phosphatase activity [GO:0010922]; positive regulation of reactive oxygen species metabolic process [GO:2000379]; programmed necrotic cell death [GO:0097300]; protein autophosphorylation [GO:0046777]; reactive oxygen species metabolic process [GO:0072593]; regulation of activated T cell proliferation [GO:0046006]; regulation of activation-induced cell death of T cells [GO:0070235]; regulation of adaptive immune response [GO:0002819]; regulation of apoptotic process [GO:0042981]; regulation of CD8-positive, alpha-beta cytotoxic T cell extravasation [GO:2000452]; regulation of reactive oxygen species metabolic process [GO:2000377]; regulation of T cell mediated cytotoxicity [GO:0001914]; regulation of type II interferon production [GO:0032649]; signal transduction [GO:0007165]; spleen development [GO:0048536]; T cell differentiation in thymus [GO:0033077]; T cell homeostasis [GO:0043029]; thymus development [GO:0048538] |
14749364_RIP3 is not an essential modulator of NF-kappa B signaling downstream of several receptor systems 19498109_study identified the protein kinase receptor-interacting protein 3 (RIP3) as a molecular switch between TNF-induced apoptosis and necrosis in NIH 3T3 cells and found that RIP3 was required for necrosis in other cells 19524512_Embryonic fibroblasts from RIP3 knockout mice are resistant to necrosis and RIP3 knockout animals are devoid of inflammation inflicted tissue damage in an acute pancreatitis model. 19524513_Using an RNA interference screen, the kinase RIP3 was identified as a crucial activator for programmed necrosis induced by TNF and during virus infection; RIP3 regulates necrosis-specific RIP1 phosphorylation. 20413098_Murine cytomegalovirus M45-encoded inhibitor of RIP activation (vIRA) targets RIP3 during infection and disrupts RIP3-RIP1 interactions characteristic of TNFalpha-induced necroptosis, thereby suppressing both death pathways. 21098270_Data show that receptor interacting protein (RIP) kinase1/3-mediated necrosis is a significant mode of photoreceptor cell loss in an experimental model of retinal detachment. 21368762_Casp8 may naturally hold alternative RIP3-dependent death pathways in check in addition to promoting apoptosis; RIP3 is responsible for the mid-gestational death of Casp8-deficient embryos 21368763_caspase-8 prevents RIPK3-dependent necrosis without inducing apoptosis by functioning in a proteolytically active complex with FLICE-like inhibitory protein long (FLIP(L), also known as CFLAR), and this complex is required for the protective function 21804564_mechanisms preventing RIP3-mediated epithelial cell death are critical for maintenance of intestinal homeostasis and indicate that programmed necrosis of intestinal epithelial cells might be implicated in the pathogenesis of inflammatory bowel disease 21876153_RIPK3 and FADD have opposing and complementary roles in promoting T-cell clonal expansion and homeostasis. 21949016_Regulatory T cells (Tregs) can suppress the proliferation of Ripk3-deficient conventional T cells resistant to apoptosis or regulated necrosis without utilizing conventional programmed cell death pathways. 22123964_Toll/IL-1 receptor domain-containing adapter inducing IFN-beta (TRIF/TICAM-1), which is dispensable for TNF-alpha-induced necrosis, forms a complex with RIP3 upon TLR3/TLR4 activation. 22193709_Studies indicate that pathogens specifically target both the caspase 8-dependent apoptotic and necrotic cell death pathway that is dependent on receptor-interacting protein 1 (RIP1; also known as RIPK1) and RIP3 (also known as RIPK3). 22195746_RIPK3 deficiency also protected against cecal ligation and puncture, underscoring the clinical relevance of RIPK kinase inhibition in sepsis. 22266724_The differential sensitivity of the RIP3-deficient A cells and the RIP3-proficient N cells to the examined Top1 inhibitors in all the tested events. 22327219_It was shown that inhibitor of apoptosis proteins are required during development to limit activity of receptor-interacting protein kinase 1 and 3 in the TNF receptor 1 signalling pathway. 22365665_activation of the cell death-inducing ripoptosome platform and RIP3 can generate bioactive IL-1beta 22423968_DAI interacts with RIP3 to mediate virus-induced necrosis analogous to the RIP1-RIP3 complex controlling death receptor-induced necroptosis. 22942356_RIP3 can phosphorylate a number of cell cycle regulators in different cell types. RIP3 also has cell type dependent functions because many of the potential RIP3 targets in the macrophages and mouse embryonic fibroblasts are enriched in different pathways. 23071110_procaspase-8 activity is essential for cell survival by inhibiting both apoptotic and nonapoptotic cell death dependent on receptor-interacting protein kinase 1 (RIP1) and RIP3 23144495_In contrast to previously described caspase-8-activation pathways, Fas-induced interleukin (IL)-1beta maturation via caspase-8 proceeds independently of RIP3 kinase. 23260196_Caspase-8 blocks kinase RIPK3-mediated activation of the NLRP3 inflammasome 23260419_phosphorylation of Daxx by RIP3 comprises an important part of ischemic necrosis in rat retinal ganglion cells 23333278_RIP3-dependent necrosis is not postapoptotic, and the increased primary necrosis in advanced atherosclerotic lesions most likely resulted from the increase of RIP3 expression. 23584955_The results indicate that RIP1 and RIP3 can independently mediate death signals being transduced by two different death stimuli, zVAD and TNFalpha. 23612963_the importance of the RIP3-MLKL interaction in the formation of functional necrosomes and suggest that translocation of necrosomes to mitochondria-associated membranes is essential for necroptosis signaling. 23744808_Acetaminophen overdose triggers the transcriptional activation of RIP3 and the newly expressed RIP3 is critical for cell necrosis by acting upstream of mitochondrial dysfunction and oxidant stress. 23846218_Characterization of a robust RIP3-dependent necroptosis signaling pathway in TLR-activated microglia upon caspase blockade. 23972991_RIP3 limits compensatory proliferation via a pathway involving Caspase-8 and JNK and promotes cholestasis and carcinogenesis. 24019532_Toll-like receptor 3-mediated necrosis via TRIF, RIP3, and MLKL. 24095729_RIP3-mediated MLKL phosphorylation, though important for downstream signaling, is dispensable for stable complex formation between RIP3 and MLKL. 24103001_RIPK3-mediated necroptosis promotes donor kidney inflammatory injury and reduces allograft survival. 24174679_Ripk3 contributes to photoreceptor degeneration in retina lacking interphotoreceptor retinoid-binding protein. 24434512_Knockdown of RIPK3 or MLKL blocks TNF-induced necroptosis. 24439895_cIAP2 (Birc3)-dependent antagonism of RIPK3-mediated programmed necrosis critically protects the host from influenza infection through maintenance of pulmonary tissue homeostasis. 24441827_Ripk3 deficiency substantially improved the clinical course of GD mice, with increased survival and motor coordination and salutary effects on cerebral as well as hepatic injury. 24453211_By comparing the phosphorylation sites in wild-type and RIP3-knockdown L929 cells, 174, 167, and 177 distinct phosphorylation sites were revealed to be dependent on RIP3 at the 0.5, 2, and 4 h time points after TNF treatment, respectively. 24556404_[review] Although studies have demonstrated that mixed lineage kinase domain-like (MLKL) protein is the only substrate of RIP3 kinase that is essential for necroptotic death, the molecular determinants acting downstream of MLKL remain ambiguous. 24557836_data indicate that the kinase activity of RIPK3 is essential for necroptosis but also governs whether a cell activates caspase-8 and dies by apoptosis 24577084_These results indicate that necroptosis caused by RIPK3 requires MLKL but not Drp1. 24799678_caspase-8 and the RIP kinases are key regulators of macrophage cell death, NF-kappaB and inflammasome activation, and host resistance after Y. pestis infection 24813850_These results clarify the complex roles for RIPK1 in postnatal life and provide insights into the regulation of FADD-caspase-8 and RIPK3-MLKL signaling by RIPK1. 24874734_Treatment with pan-caspase inhibitor ZVAD blocked the activation of caspase-8 and reduced the number of apoptotic nuclei, while increasing levels of RIP1, RIP3, and necrotic OHCs. 24882010_RIPK3 ubiquitylation and activation is modulated by the action of XIAP 24901049_The B-Raf(V600E) inhibitor dabrafenib selectively inhibits RIP3 and alleviates acetaminophen-induced liver injury. 24902899_dimers of RIPK1 and/or RIPK3 are the pivotal complexes from which both forms of cell death can progress. 24920296_RIP3-dependent necroptosis modulates post-ischaemic adverse remodelling in a mouse model of myocardial infarction 24984764_RIPK1/3 contributes to MVEC death and cardiac allograft survival through necroptotic death and the release of danger molecules 24996547_RIPK3 deficiency attenuates organ injury in sepsis and alters immune cell trafficking. 25049377_IFN-beta-induced necroptosis of macrophages proceeds through tonic IFN-stimulated gene factor 3 (ISGF3) signaling, which leads to persistent expression of STAT1, STAT2, and IRF9 and sustained Rip3 activation. 25246544_Data show that hematopoietic receptor-interacting serine/threonine-protein kinase 1 (RIPK1) deficiency triggers both apoptotic and necroptotic death that is partially prevented by receptor-interacting serine-threonine kinase 3 (RIPK3) deficiency. 25316792_Ectopic expression of ICP6, but not RHIM mutant ICP6, directly activated RIP3/MLKL-mediated necrosis. 25326752_our results reveal a specific role for the RIP1-RIP3-DRP1 pathway in RNA virus-induced activation of the NLRP3 inflammasome 25367573_The results reveal an unexpected function of RIPK3 in NF-kappaB activation, dendritic cell biology, innate inflammatory-cytokine expression, and injury-induced tissue repair. 25423287_although JNK activation and RIP3 expression are induced by FS, neither contributes to the liver injury. 25563840_Enhanced RIP3 signaling in aneurysmal tissues contributes to abdominal aortic aneursym progression by causing smooth muscle cell necroptosis, as well as stimulating vascular inflammation. 25567679_RIPK3 is an unexpected positive regulator of caspase 8 activity that promotes IL-1beta maturation in bone marrow-derived dendritic cells 25674205_RIP1 and RIP3-mediated necroptosis may involve in the pathogenesis of aplastic anemia induced by cyclophosphamide and busulfan in mice. 25674982_Human herpesvirus 1 ICP6 interacts with mouse RIP1/RIP3 through its RHIM domain and forms dimers/oliogmers by its C-terminal R1 domain. 25693118_RIPK3 can promote NLRP3 inflammasome and IL-1beta inflammatory responses independent of MLKL and necroptotic cell death 25788533_Cisplatin stimulates RIP1/RP3/MLKL-dependent necrotic cell death in renal tubules, which finally causes renal dysfunction 25906154_Data implicate the infiltrating macrophages as a source of damaging inflammasomes after photoreceptor detachment in a RIP3-dependent manner and suggest a novel therapeutic target for treatment of retinal diseases. 25939025_these studies link A20 and the ubiquitination of RIPK3 to necroptotic cell death and suggest additional mechanisms by which A20 might prevent inflammatory disease. 25950489_diverse modes of acute liver injury have differing requirements for RIP1 and RIP3; moreover, within a single injury model, RIP1 and RIP3 blockade can have diametrically opposite effects on tissue damage 26024392_we demonstrate that the phosphorylation of Ser345 is not required for the interaction between RIPK3 and MLKL in the necrosome, but is essential for MLKL translocation, accumulation in the plasma membrane, and consequent necroptosis. 26086143_Studies indicate that RIP kinases RIPK1 and RIPK3 directly regulate inflammatory signaling. 26238997_Results suggest that RGC-5 cell necroptosis following oxygen glucose deprivation is mediated by a RIP3-induced increase in oxidative stress 26283397_Lymphocyte infiltrations in the adipocyte tissue and in skin lesions of ApoE single-knockout mice were significantly mitigated in ApoE/RIP3 double-knockout mice 26381214_Results identify a crucial role for RIPK3-PGAM5-Drp1/NFAT signalling in NKT cell activation, and further suggest that RIPK3-PGAM5 signalling may mediate crosstalk between mitochondrial function and immune signalling. 26451737_RIPK3 is an essential molecule to maintain the temporal manner of the normal progression of wound closure 26471282_Data indicate that receptor-interacting serine-threonine kinase 3 (RIPK3) deletion prevents inflammatory phenotypes in CreLysM (lysozyme M) Casp8fl/fl (caspase 8) mice. 26726877_RIP3-induced activation of CaMKII, via phosphorylation or oxidation or both, triggers opening of the mitochondrial permeability transition pore and myocardial necroptosis. 26769846_Results suggest that impaired hepatic proteasome function by alcohol exposure may contribute to hepatic accumulation of RIP3 resulting in necroptosis and steatosis while RIP1 kinase activity is important for alcohol-induced inflammation. 26817517_deficiency of RIPK3 or MLKL prevents oxalate crystal-induced acute kidney injury. 26900751_CHIP is a bona fide negative regulator of the RIPK1-RIPK3 necrosome formation leading to desensitization of TNF-mediated necroptosis 26969469_Study shows that Rip3 mRNA expression is at the highest level in the spleen and duodenum, but lowest level in brain. Protein detection results revealed its localization in different type cells in different tissues that can be either nuclear or cytoplasmic. 26995086_RIP3 deletion suppresses inflammation response. 27195494_RIP3-mediated necroptosis is a major mechanism of enhanced inflammation and lung tissue injury in high dose LPS- induced severe ARDS in mice 27321907_Mice deficient in RIPK3 or doubly deficient in MLKL and FADD, but not MLKL alone, are more susceptible to influenza A virus than their wild-type counterparts, revealing an important role for RIPK3-mediated apoptosis in antiviral immunity. 27323669_The necroptosis-inducing kinase RIPK3 reduces adipose tissue inflammation and glucose intolerance. 27344176_our results reveal that the necroptosis adaptor RIPK3 has key anti-inflammatory and anti-tumoral functions in the intestine, and define RIPK3 as a novel colon tumor suppressor 27396959_this study shows that RIPK1 and RIPK3 account for acute inflammatory responses induced by lipopolysaccharide in vivo; notably, this regulation does not require exogenous manipulation of caspases 27411587_Data identify RIPK3 and the inflammasome as key tumor suppressors in acute myeloid leukemia (AML). 27411587_Results demonstrate that RIPK3 restricts malignant myeloproliferation by activating the inflammasome, which promotes differentiation and cell death, and that loss of RIPK3 increases leukemic burden in mice. Reduced RIPK3 expression is observed across several human acute myeloid leukemia subtypes. 27523270_RIPK3 and/or MLKL may exert functions independently of necroptosis. 27560715_robust caspase activation in NEMO-deficient cells is concomitant with RIPK3 recruitment to the apoptosis-mediating complex. 27584790_these results demonstrated that RIPK3-mediated signaling in Tie-2 expressing cells was responsible for the embryonic lethality of Fadd-/- with cardiac failure. 27640145_These data suggest that Rip3 plays a role in neurodegeneration and mitochondrial morphology in the absence of mitochondrial division. 27746097_These results identify DAI as a link between influenza A virus replication and RIPK3 activation and implicate DAI as a sensor of RNA viruses. 27756058_results reveal a pathway for MLKL-dependent programmed necrosis that is executed in the absence of RIPK3 and potentially drives the pathogenesis of severe liver diseases. 27840306_Aldehyde dehydrogenase 2 deficiency negates chronic low-to-moderate alcohol consumption-induced cardioprotecion possibly via ROS-dependent apoptosis and RIP1/RIP3/MLKL-mediated necroptosis. 27856241_The findings reported here indicate that palmitate induces RIP1/RIP3-dependent necrosis via MLKL-mediated pore formation of RAW 264.7 cells in the plasma membrane, which could provide a new mechanism to explain the link between elevated levels of free fatty acids (FFAs), palmitate in particular, and macrophage death. 28151480_An alternative function for RIPK1/RIPK3 in vascular permeability. 28176780_The major function of RIP1 kinase activity in TNF-induced necroptosis is to autophosphorylate serine 161. This specific phosphorylation then enables RIP1 to recruit RIP3 and form a functional necrosome, a central controller of necroptosis. 28242694_These data demonstrate a role for RIP3 in promoting in vivo thrombosis and hemostasis by amplifying platelet activation. RIP3 may represent a novel promising therapeutic target for thrombotic diseases. 28256024_Low RIP3 expression is associated with inflammation in dextran sodium sulfate-induced ulcerative colitis . 28273458_a key physiological function of RIPK3 is to promote reparative cytokine expression through intestinal CD11c(+) mononuclear phagocytes in a kinase- and necroptosis-independent manner. 28342910_RIPK3 promotes keratinocyte activation and facilitates neutrophil infiltration in the skin independent of necroptosis. These findings may shed new lights on the role of RIPK3 in the skin, and may provide an important clue for the treatment of neutrophil-mediated inflammatory skin diseases, including psoriasis. 28366204_Using a mouse model of West Nile virus encephalitis, study shows that RIPK3 restricts WNV pathogenesis independently of cell death. Ripk3(-/-) mice exhibited enhanced mortality compared to wild-type controls. 28387756_Knock-out mice deficient in MLKL or RIP3 had increased survival and reduced pulmonary injury during Serratia marcescens pneumonia. 28401933_in Mycobacterium tuberculosis-infected macrophages, mitochondria are an essential platform for induction of necrosis by activating RIPK3 function and preventing caspase 8-activation. 28410401_RIPK3 regulates type I IFN both transcriptionally, by interacting with MAVS and limiting RIPK1 interaction with MAVS, and post-transcriptionally. 28423682_RIP3-mediated activation of caspase-1 rather than necroptosis-dependent inflammation was responsible for aggressive inflammation in influenza A (H7N9) virus-infected mice. 28445730_Ablation of Ripk3 rescues Fadd-deficient mice through two processes: inhibiting necroptosis during embryonic development and restricting massive inflammation during postnatal development, which were segregated by the RIPK3 mutation. 28461567_study identifies a novel role for RIPK1 and RIPK3, a pair of homologous serine/threonine kinases previously implicated in the regulation of necroptosis and pathologic tissue injury, in directing IFN-beta production in macrophages 28564603_2-hydroxyglutarate bound to DNMT1 and stimulated its association with the RIP3 promoter, inducing hypermethylation that reduces RIP3 protein and consequently impaired RIP3-dependent necroptosis. 28660207_we assessed the role of RIP3 in synergy with Caspase-1 in the induction of IL-1beta production in BMDM after either LPS/ATP or Chlamydia muridarum stimulation. The possibility of pyroptosis and necroptosis interplays and the role of RIP3 in IL-1beta production during Chlamydia muridarum infection in BMDM was investigated as well. 28661484_Pull down experiments with biotinylated Sorafenib show that it binds independently RIPK1, RIPK3 and MLKL. Moreover, it inhibits RIPK1 and RIPK3 kinase activity. In vivo Sorafenib protects against TNF-induced systemic inflammatory response syndrome (SIRS) and renal ischemia-reperfusion injury (IRI). 28703808_Rip3 removal did not reduce pancreatic tissue damage, but rather enhanced this damage, though without an increase in local inflammation and even a reduction in macrophage counts. 28724891_necroptosis contributes to ischemic brain injury induced by oxygen-glucose deprivation and middle cerebral artery occlusion and implicate HIF-1alpha, RIP1, RIP3, and MLKL in necroptosis. 28732308_Ripk3 promotes mitochondrial apoptosis via inhibition of FUNDC1 mitophagy in cardiac ischemia reperfusion injury. 28807105_The authors report here that male reproductive organs of both Ripk3- and Mlkl-knockout mice retain 'youthful' morphology and function into advanced age, while those of age-matched wild-type mice deteriorate. Feeding of wild-type mice with an RIPK1 inhibitor prior to the normal onset of age-related changes in their reproductive organs blocked the appearance of signs of aging. 29055012_Impaired Nrf2-dependent redox homeostasis is an important mechanism that promotes cell death downstream of IFN-I and RIP3 signaling in S. Typhimurium-infected macrophages. 29238045_RIPK3 promotes adenovirus type 5 oncolytic activity. 29248164_Deficiency in RIPK3 attenuated serum and lung cytokines, lung injury and neutrophil infiltration and lung and gut apoptosis. These data suggest that RIPK3, in part, is responsible for the systemic inflammatory response in neonatal sepsis. 29413844_Ripk3 was involved in microvascular ischemia reperfusion injury via regulation of IP3R-mediated calcium overload, xanthine oxidase-dependent oxidative damage and filopodia-related cellular migration, ultimately leading to endothelial apoptosis and migratory inhibition. 29415885_Study revealed increased kidney expression of RIPK3 in mouse models of progressive kidney fibrosis and in human chronic kidney disease kidney and demonstrate a pathway by which RIPK3 promotes fibrogenesis through the AKT-dependent activation of ATP citrate lyase independently of mixed-lineage kinase domain-like protein-dependent necroptosis. 29434332_p55TNFR-IKK2-Ripk3 signalling orchestrates arthritogenic and death responses in synovial fibroblasts 29470982_RIP3-ablation attenuated oxidative stress, inflammation and apoptosis in astrocytes, which was dependent on AMPKalpha activation. 29502045_Our results demonstrated that Ripk3 was strongly upregulated in murine hearts subjected to IR injury and cardiomyocytes treated with LPS and H2O2..the present study helps to elucidate how necroptosis is mediated by ER stress, via the calcium overload /XO/ROS/mPTP opening axis 29527209_Receptor-Interacting Protein Kinases 1 and 3, and Mixed Lineage Kinase Domain-Like Protein Are Activated by Sublytic Complement and Participate in Complement-Dependent Cytotoxicity. 29634408_RIP3-mediated signaling is not a critical driver of acute radiation syndrome 29666472_These findings indicate that the triggering of inflammation by Casp8 deletion in mice occurs, in part, independently of necroptosis or other functions of RIPK3, and rather reflects enhanced RIPK1-dependent signaling for activation of inflammatory genes. 29686269_These data suggest that lysosomal dysfunction after spinal cord injury may contribute to both inhibition of autophagy and sensitize cells to necroptosis by promoting RIPK1 and RIPK3 accumulation. 29720570_We demonstrate that the necroptosis-related molecule RIPK3, but not RIPK1 or MLKL, may be important in the development of experimental VILI and that impaired FAO may be involved. 29770487_melatonin treatment inhibited the Ripk3-PGAM5-CypD-mPTP cascade and thus reduced cellular necroptosis, conferring a protective advantage to the endothelial system in IR stress. 29847649_Our results suggest that RIP3 enhances innate immune responses against ocular MCMV infection via activation of the inflammasome and nuclear factor-kappaB, which also leads to inflammation and death of bystander cells by multiple pathways including apoptosis and necroptosis. 29875323_Study revealed that in kidney tubular epithelial cells RIPK3 promotes oxidative stress and mitochondrial dysfunction by upregulation of NOX4 and inhibition of mitochondrial complex I and -III, and that RIPK3 and NOX4 are critical for kidney tubular injury in vivo. RIPK3 is required for increased mitochondrial translocation of NOX4 in response to proinflammatory stimuli by a mechanism involving protein-protein interaction. 29892302_the loss of RIPK3 did not alter overall disease outcomes, with deficient animals harboring similar bacterial numbers in the lungs and spleens compared to their wild-type counterparts. Mtb-infected macrophages were not rescued from dying by Ripk3 deletion, nor did this affect production of the pro-inflammatory cytokine IL-1beta, both in vitro and in vivo. 30126430_RIP3 participates in the NLRP3 inflammasome activation in infiltrating macrophages in acute lung injury induced by lipopolysaccharide 30131368_Data suggest that the receptor-interacting protein kinase 3 (RIPK3) kinase domain exhibits catalytically independent function that is important for both RIPK3-dependent necroptosis and apoptosis. 30175514_Deletion of both RIPK3 and caspase-8 augments intrinsic apoptosis in tubular epithelial cell and promote kidney ischaemia-reperfusion injury. 30185824_deletion of Ripk3 is sufficient to rescue the neonatal lethality of Ripk1(-/-)Tradd(-/-) animals and delays but does not completely prevent early mortality. Although Ripk3 deletion provides a significant survival advantage, Ripk1(-/-)Tradd(-/-)Ripk3(-/-) animals die between 22 and 49 days, are runty compared to littermate controls and present with splenomegaly 30192300_This study showed that RIP3 was highly expressed in DRG, SC and HIP of the sciatic nerve in CCI mice and may be involved in chronic neuroinflammation and neuronecrosis. 30194111_Ripk3 signaling is dispensable to the generation of immunity against herpesvirus infection 30194302_key player in the degenerative process in dystrophin-deficient muscles 30280195_miR-155 up-regulation in macrophages by Salmonella infection causes macrophage death and it may be mediated by both RIP1/3-related necroptosis and PARP-1-mediated necrosis. 30442927_Here we showed that c-Jun N-terminal kinases (JNK1 and JNK2) displayed kinase-dependent and -independent functions in regulating TNF- and TLRs-mediated necroptosis. We found that RIPK1 and RIPK3 promoted cell-death-independent JNK activation in macrophages, which contributed to pro-inflammatory cytokines production 30635240_this study shows that ZBP1 and RIPK3 induce IRG1 to promote an antiviral metabolic state in neurons 30703581_Receptor interacting protein kinase 3 (RIP3) regulates iPSCs generation through modulating cell cycle progression genes. 30815534_Silencing of Ripk3 in cultured motor neurons protected them from toxicity associated with SOD1(G93A) astrocytes. However, constitutive deletion of Ripk3 in Tg SOD1(G93A) mice failed to provide behavioral or neuropathological improvement, demonstrating no similar benefit of Ripk3 silencing in vivo. Lastly, we detected no genotype-specific myelin decompaction, proposed to be a proxy of necroptosis in ALS. 31000631_A novel role of RIPK3 in bacteria-induced nonresolving inflammation, and uncovers a previously unrecognized mechanism by which HMGB1 and bacterial components work in concert to orchestrate RIPK3-dependent immune responses under physiological conditions. 31026418_Receptor-Interacting Serine/Threonine-Protein Kinase 3 (RIPK3)-Mixed Lineage Kinase Domain-Like Protein (MLKL)-Mediated Necroptosis Contributes to Ischemia-Reperfusion Injury of Steatotic Livers. 31133359_Pathogenesis of lupus nephritis: RIP3 dependent necroptosis and NLRP3 inflammasome activation. 31227597_Here, we describe a role for RIPK1/RIPK3 activation in which necroptotic fibroblasts within the TME drive increased antigen uptake and activation of tumor APCs to potentiate tumor-specific CD8+ T cell immunity, which synergizes with alpha-PD-1 coadministration to confer durable tumor rejection. Our data suggest that RIPK1/RIPK3 activation within the TME warrants further development as a component of tumor immunotherapy. 31235857_The NuRD chromatin-remodeling complex enzyme CHD4 prevents hypoxia-induced endothelial Ripk3 transcription and murine embryonic vascular rupture. 31257491_The present study revealed that RIPK3 mediated renal tubular cell apoptosis in endotoxininduced acute kidney injury. 31285326_Mice deficient in CASP8 and RIPK3 (Casp8-/-Ripk3-/-) mount enhanced peak CD8 T cell levels against the natural mouse pathogen murine cytomegalovirus. 31295018_that RIP3 plays a crucial proinflammatory role in liver fibrosis by regulating the ROCK1-TLR4-NF-kappaB signaling pathway in macrophages 31358656_IFN-activated Jak/STAT signaling induces robust expression of ZBP1, which complexes with RIPK3 in the absence of RIPK1. 31430539_Ripk3 licenced protection against microbial infection in the absence of Caspase1-11 inflammasome. 31461645_RIPK3 Activation Leads to Cytokine Synthesis that Continues after Loss of Cell Membrane Integrity. 31506635_RIP1/RIP3/MLKL mediates dopaminergic neuron necroptosis in a mouse model of Parkinson disease. 31659279_Caspase-8, receptor-interacting protein kinase 1 (RIPK1), and RIPK3 regulate retinoic acid-induced cell differentiation and necroptosis. 31665609_Receptor for advanced glycation end product (RAGE) deficiency effectively blocked RIP3 phosphorylation induced by Nepsilon-(carboxymethyl) lysine (CML) and rescued CML-mediated MI/RI, indicating CML promoted RIP3-mediated necroptosis through RAGE. I 31690718_Ablation of RIP3 protects from dopaminergic neurodegeneration in experimental Parkinson's disease. 31801942_Casein kinase-1gamma1 and 3 stimulate tumor necrosis factor-induced necroptosis through RIPK3. 31953345_Cell-specific and athero-protective roles for RIPK3 in a murine model of atherosclerosis. 31961824_Gut epithelial TSC1/mTOR controls RIPK3-dependent necroptosis in intestinal inflammation and cancer. 31968247_Constitutive Interferon Attenuates RIPK1/3-Mediated Cytokine Translation. 31988496_RIPK3 upregulation confers robust proliferation and collateral cystine-dependence on breast cancer recurrence. 32119761_SIRT3 deficiency delays diabetic skin wound healing via oxidative stress and necroptosis enhancement. 32152555_RIPK3 collaborates with GSDMD to drive tissue injury in lethal polymicrobial sepsis. 32182211_Nrf2 deficiency aggravates PM2.5-induced cardiomyopathy by enhancing oxidative stress, fibrosis and inflammation via RIPK3-regulated mitochondrial disorder. 32247850_The Bcr-Abl inhibitor GNF-7 inhibits necroptosis and ameliorates acute kidney injury by targeting RIPK1 and RIPK3 kinases. 32321638_Inhibition of RIPK1/RIPK3 ameliorates osteoclastogenesis through regulating NLRP3-dependent NF-kappaB and MAPKs signaling pathways. 32542792_RIPK3 blockade attenuates kidney fibrosis in a folic acid model of renal injury. 32555451_Loss of RIPK3 does not impact MYC-driven lymphomagenesis or chemotherapeutic drug-induced killing of malignant lymphoma cells. 32633835_RAGE interacts with the necroptotic protein RIPK3 and mediates transfusion-induced danger signal release. 32703968_Catalytically inactive RIP1 and RIP3 deficiency protect against acute ischemic stroke by inhibiting necroptosis and neuroinflammation. 32895234_TRIM24-RIP3 axis perturbation accelerates osteoarthritis pathogenesis. 32989095_RIPK3 Promotes Mefv Expression and Pyrin Inflammasome Activation via Modulation of mTOR Signaling. 33024046_RIPK3-Dependent Recruitment of Low-Inflammatory Myeloid Cells Does Not Protect from Systemic Salmonella Infection. 33119828_Expression of Protein Kinases RIPK-1 and RIPK-3 in Mouse and Human Hair Follicle. 33221042_Enhanced RIPK3 kinase activity-dependent lytic cell death in M1 but not M2 macrophages. 33389663_RIP1-dependent linear and nonlinear recruitments of caspase-8 and RIP3 respectively to necrosome specify distinct cell death outcomes. 33449298_RIPK3 modulates growth factor receptor expression in endothelial cells to support angiogenesis. 33513913_RIPK3 Contributes to Lyso-Gb3-Induced Podocyte Death. 33712586_The amyloid structure of mouse RIPK3 (receptor interacting protein kinase 3) in cell necroptosis. 33712742_OTUB1 prevents lethal hepatocyte necroptosis through stabilization of c-IAP1 during murine liver inflammation. 33779513_TNF-induced necroptosis initiates early autophagy events via RIPK3-dependent AMPK activation, but inhibits late autophagy. 33833407_Stratifin promotes renal dysfunction in ischemic and nephrotoxic AKI mouse models via enhancing RIPK3-mediated necroptosis. 33836603_A specific RIP3(+) subpopulation of microglia promotes retinopathy through a hypoxia-triggered necroptotic mechanism. 33841405_Leaky Gut Driven by Dysbiosis Augments Activation and Accumulation of Liver Macrophages via RIP3 Signaling Pathway in Autoimmune Hepatitis. 33850121_Conformational interconversion of MLKL and disengagement from RIPK3 precede cell death by necroptosis. 33854696_RIPK3 Induces Cardiomyocyte Necroptosis via Inhibition of AMPK-Parkin-Mitophagy in Cardiac Remodelling after Myocardial Infarction. 33862457_Down-regulation of pro-necroptotic molecules blunts necroptosis during myogenesis. 33976111_Viral dosing of influenza A infection reveals involvement of RIPK3 and FADD, but not MLKL. 34029184_A phosphorylation of RIPK3 kinase initiates an intracellular apoptotic pathway that promotes prostaglandin2alpha-induced corpus luteum regression. 34046766_Modulation of vigabatrin induced cerebellar injury: the role of caspase-3 and RIPK1/RIPK3-regulated cell death pathways. 34103472_RIP3 impedes transcription factor EB to suppress autophagic degradation in septic acute kidney injury. 34112763_Necroptosis protects agai |
ENSG00000129465 |
RIPK3 |
175.278131 |
2.9842330180 |
1.577360 |
0.32380737 |
22.893815 |
0.00000171201819839026050583624758566481105503953585866838693618774414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000146256091967921766569538100100089650368317961692810058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
278.647272473974 |
63.0677339661272 |
93.3731618116615 |
16.1787686814347 |
| ENSMUSG00000022231 |
20356 |
Sema5a |
protein_coding |
Q3UPZ0 |
Human_homologues FUNCTION: Bifunctional axonal guidance cue regulated by sulfated proteoglycans; attractive effects result from interactions with heparan sulfate proteoglycans (HSPGs), while the inhibitory effects depend on interactions with chondroitin sulfate proteoglycans (CSPGs) (By similarity). Ligand for receptor PLXNB3. In glioma cells, SEMA5A stimulation of PLXNB3 results in the disassembly of F-actin stress fibers, disruption of focal adhesions and cellular collapse as well as inhibition of cell migration and invasion through ARHGDIA-mediated inactivation of RAC1. May promote angiogenesis by increasing endothelial cell proliferation and migration and inhibiting apoptosis. {ECO:0000250, ECO:0000269|PubMed:15218527, ECO:0000269|PubMed:19850054, ECO:0000269|PubMed:20696765, ECO:0000269|PubMed:21706053}. |
Differentiation;Disulfide bond;Glycoprotein;Membrane;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:20356; |
membrane [GO:0016020]; chondroitin sulfate proteoglycan binding [GO:0035373]; heparan sulfate proteoglycan binding [GO:0043395]; semaphorin receptor binding [GO:0030215]; syndecan binding [GO:0045545]; axonal fasciculation [GO:0007413]; diencephalon development [GO:0021536]; negative regulation of axon extension involved in axon guidance [GO:0048843]; positive regulation of axon extension involved in axon guidance [GO:0048842]; signal clustering [GO:1990256] |
12506007_development of some CNS nerves requires inhibitory sheaths to maintain integrity, this is accomplished using molecules such as Sema5A that exhibit conserved inhibitory responses in the presence of impinging signals from multiple guidance molecules. 15218527_Plexin-B3 is a functional receptor for semaphorin 5A 15603739_Semaphorin 5A (Sema5A) is a bifunctional guidance cue exerting both attractive and inhibitory effects on developing axons of the fasciculus retroflexus, a diencephalon fiber tract associated with limbic function. 15743826_A role of Sema5A in the remodeling of the cranial blood vessels was shown. 19850054_Data reveal that Sema5A is a novel proangiogenic molecule, inducing endothelial cell proliferation and migration and inhibiting apoptosis. 21777623_The results of this study concluded that the Sema5a mice do not meet the behavioral criteria for a mouse model of autism. 21835343_Sema5A and Sema5B inhibit retinal neurite outgrowth through PlexinA1 and PlexinA3 receptors both in vitro and in vivo. 25313870_Mouse Sema5A negatively regulates synaptogenesis in early, developmentally born, hippocampal dentate granule cells. |
ENSG00000112902 |
SEMA5A |
679.089953 |
0.0872623463 |
-3.518497 |
0.13019807 |
748.076658 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001051008150687556408248451980476948996906001144081443445875516927482063313479005973444386681690899977 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000100055975945455377622761325504050137740443417774981440694980748839332467103022097041914494666752291 |
Yes |
No |
106.363534948167 |
9.61196359233772 |
1218.8938237331 |
67.3820233230571 |
| ENSMUSG00000022296 |
118452 |
Baalc |
protein_coding |
Q8VHV1 |
FUNCTION: May play a synaptic role at the postsynaptic lipid rafts possibly through interaction with CAMK2A. {ECO:0000250|UniProtKB:Q920K5}. |
Alternative splicing;Cytoplasm;Lipoprotein;Membrane;Myristate;Palmitate;Reference proteome;Synapse;Synaptosome |
|
|
mmu:118452; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; membrane raft [GO:0045121]; neuron projection [GO:0043005]; nucleoplasm [GO:0005654]; postsynaptic density [GO:0014069]; sarcoplasm [GO:0016528] |
15749074_BAALC/Baalc is a marker of the mesodermal lineage, especially muscle. |
ENSG00000164929 |
BAALC |
22.459697 |
11.3880195444 |
3.509445 |
0.71142458 |
24.910031 |
0.00000060069064855133171610025591760639862570769764715805649757385253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000055675199626704137089686259820542346687943791039288043975830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
40.3611376608198 |
20.7194584355283 |
3.54417530664426 |
1.5406924281428 |
| ENSMUSG00000022306 |
22762 |
Zfpm2 |
protein_coding |
Q8CCH7 |
FUNCTION: Transcription regulator that plays a central role in heart morphogenesis and development of coronary vessels from epicardium, by regulating genes that are essential during cardiogenesis. Essential cofactor that acts via the formation of a heterodimer with transcription factors of the GATA family GATA4, GATA5 and GATA6. Such heterodimer can both activate or repress transcriptional activity, depending on the cell and promoter context. Also required in gonadal differentiation, possibly be regulating expression of SRY. Probably acts a corepressor of NR2F2. {ECO:0000269|PubMed:10330188, ECO:0000269|PubMed:10888889, ECO:0000269|PubMed:10892744, ECO:0000269|PubMed:12223418, ECO:0000269|PubMed:9927674, ECO:0000269|PubMed:9927675}. |
Activator;Alternative splicing;Cardiomyopathy;DNA-binding;Isopeptide bond;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:22762; |
cytoplasm [GO:0005737]; male germ cell nucleus [GO:0001673]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; metal ion binding [GO:0046872]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; transcription coactivator activity [GO:0003713]; transcription corepressor activity [GO:0003714]; atrioventricular valve morphogenesis [GO:0003181]; cardiac muscle tissue development [GO:0048738]; cardiac muscle tissue morphogenesis [GO:0055008]; cell differentiation [GO:0030154]; embryonic organ development [GO:0048568]; heart development [GO:0007507]; in utero embryonic development [GO:0001701]; lung development [GO:0030324]; mitral valve formation [GO:0003192]; negative regulation of cell death [GO:0060548]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of female gonad development [GO:2000195]; negative regulation of transcription by RNA polymerase II [GO:0000122]; outflow tract morphogenesis [GO:0003151]; outflow tract septum morphogenesis [GO:0003148]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of male gonad development [GO:2000020]; positive regulation of transcription by RNA polymerase II [GO:0045944]; right ventricular cardiac muscle tissue morphogenesis [GO:0003221]; tricuspid valve formation [GO:0003195]; vasculogenesis [GO:0001570]; vasculogenesis involved in coronary vascular morphogenesis [GO:0060979]; ventricular septum morphogenesis [GO:0060412] |
12213678_FOG-2, in addition to GATA-4, has a role in early gonadal development and sexual differentiation, and FOG-1 at later fetal stages, while GATA-1 executes its action postnatally 12223418_Gonadal differentiation, sex determination and normal Sry expression in mice require direct interaction between transcription partners GATA4 and FOG2 14551244_FOG-2 null mouse embryos had thin compact myocardium, higher EDP and E/A ratio, smaller -dP/dt, and decreased sucking pressure than wild littermates, indicating that decreased ventricular diastolic function might be the main cause of embryonic lethality 15507435_the N-terminal domain of FOG2 has a role in mediating transcriptional repression 17142311_A Fog2-Gata4 interaction is critical for the development of normal pulmonary lobar structure, and this phenotype is not influenced by the additional loss of Gata6 interaction. 17274974_FOG-2 attenuates endothelial-to-mesenchymal transformation in the endocardial cushions of the developing heart. 17310981_implicates FOG2 in the maintenance of epithelial cell differentiation in the mammary gland and in performing a protective role in breast cancer 17445768_Results suggest that FOG-2 repressive activity may be modulated by the generation of isoforms of FOG-2 lacking the FOG repression motif. 17540364_FOG2 is a limiting factor in the formation of a functional GATA4/FOG2 transcription complex that is required for Sox9 expression during gonadogenesis. 17848526_Fog2 transcription factors are haploinsufficient for testis determination in mice 18577233_the Lhx9 gene represents the first direct target of the GATA4/FOG2 repressor complex in cardiac development. 18658259_SERCA2 is an important target of FOG-2 and that increased FOG-2 expression may contribute to a decline in cardiac function in end-stage heart failure by impaired T3 signaling 18927154_Control of ovarian development by the GATA4-FOG2 complex presents a novel insight into the cross-talk between transcriptional regulation and extracellular signaling that occurs in ovarian development 19411759_FOG2 regulates adult heart function and coronary angiogenesis 19578715_Data show that cardiac expression of slow skeletal troponin T (Tnnt1) strictly depends on the physical interaction between GATA4-FOG2 in the myocardium of both atria and ventricles. 21385577_gonadal loss of Fog2 resulted in an early partial block in male pathway and sex reversal 23226341_a new dynamics in which GATA-4 may alter the activity of FOG-2 by influencing its SUMOylation status 23769843_regulation of ovarian differentiation is dependent upon GATA4 but not FOG2 23788640_data suggest a new mechanism for TGF-beta-induced Akt activation through FOG2 down-regulation by miR-200b/c, which can lead to glomerular mesangial hypertrophy in the progression of diabetic nephropathy 23836893_ATOH8, GATA4, and FOG2 associate in a single complex 25196150_Results indicate that FOG-2/NuRD interaction is required for cardiomyocyte proliferation. 26285119_MicroRNA-200c Promotes Suppressive Potential of Myeloid-Derived Suppressor Cells by Modulating PTEN and FOG2 Expression 27207553_FOG2 liver-specific knockout mice they exhibit enhanced insulin sensitivity and elevated hepatic TG accumulation, which were also reversed by Ad-PPARalpha. The results demonstrate a novel function of hepatic FOG2 in insulin sensitivity and lipid metabolism through PPARalpha. 27321927_identified key controls over corticothalamic projection neurons (CThPN) development and identify Fog2 as a critical control over CThPN diversity 27576955_Hypoxia-induced microRNA-429 promotes differentiation of osteoblastic cells by mediating ZFPM2 expression. 29262331_Fog2Rb-/Rb- mice are resistant to obesity and present abnormal WAT/BAT conversion. 31012180_The integrity of the FOG-2 LXCXE pRb-binding motif is required for small intestine homeostasis. |
ENSG00000169946 |
ZFPM2 |
14.702361 |
0.1742741174 |
-2.520570 |
0.72202028 |
12.276498 |
0.00045869834622494747976978457515428999613504856824874877929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0023189157682371058268044894390413901419378817081451416015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.09712426813188 |
2.13714687320438 |
23.5096543872581 |
7.87915025006498 |
| ENSMUSG00000022309 |
11600 |
Angpt1 |
protein_coding |
O08538 |
FUNCTION: Binds and activates TEK/TIE2 receptor by inducing its dimerization and tyrosine phosphorylation. Plays an important role in the regulation of angiogenesis, endothelial cell survival, proliferation, migration, adhesion and cell spreading, reorganization of the actin cytoskeleton, but also maintenance of vascular quiescence. Required for normal angiogenesis and heart development during embryogenesis. After birth, activates or inhibits angiogenesis, depending on the context. Inhibits angiogenesis and promotes vascular stability in quiescent vessels, where endothelial cells have tight contacts. In quiescent vessels, ANGPT1 oligomers recruit TEK to cell-cell contacts, forming complexes with TEK molecules from adjoining cells, and this leads to preferential activation of phosphatidylinositol 3-kinase and the AKT1 signaling cascades. In migrating endothelial cells that lack cell-cell adhesions, ANGT1 recruits TEK to contacts with the extracellular matrix, leading to the formation of focal adhesion complexes, activation of PTK2/FAK and of the downstream kinases MAPK1/ERK2 and MAPK3/ERK1, and ultimately to the stimulation of sprouting angiogenesis. Mediates blood vessel maturation/stability. Implicated in endothelial developmental processes later and distinct from that of VEGF. Appears to play a crucial role in mediating reciprocal interactions between the endothelium and surrounding matrix and mesenchyme (By similarity). {ECO:0000250}. |
Angiogenesis;Coiled coil;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal |
|
This gene encodes a secreted glycoprotein that belongs to the angiopoietin family of vascular growth factors. The encoded protein is a ligand in the vascular tyrosine kinase signaling pathway and regulates the formation and stabilization of blood vessels. This protein also functions in striated muscles by promoting proliferation, migration and differentiation of skeletal myoblasts and plays an essential role in the vascular response to tissue injury. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Oct 2013]. |
mmu:11600; |
collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; membrane raft [GO:0045121]; microvillus [GO:0005902]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; receptor tyrosine kinase binding [GO:0030971]; signaling receptor binding [GO:0005102]; vascular endothelial growth factor receptor binding [GO:0005172]; activation of transmembrane receptor protein tyrosine kinase activity [GO:0007171]; angiogenesis [GO:0001525]; branching involved in blood vessel morphogenesis [GO:0001569]; cardiac muscle tissue morphogenesis [GO:0055008]; cell-substrate adhesion [GO:0031589]; endocardium morphogenesis [GO:0003160]; endoderm development [GO:0007492]; glomerulus vasculature development [GO:0072012]; hemopoiesis [GO:0030097]; heparin biosynthetic process [GO:0030210]; in utero embryonic development [GO:0001701]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cell adhesion [GO:0007162]; negative regulation of cytokine production involved in immune response [GO:0002719]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of protein import into nucleus [GO:0042308]; negative regulation of protein phosphorylation [GO:0001933]; negative regulation of vascular permeability [GO:0043116]; neuron apoptotic process [GO:0051402]; ovarian follicle development [GO:0001541]; positive chemotaxis [GO:0050918]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of blood-brain barrier permeability [GO:1905605]; positive regulation of cell adhesion [GO:0045785]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein ubiquitination [GO:0031398]; positive regulation of receptor internalization [GO:0002092]; positive regulation of vascular endothelial growth factor receptor signaling pathway [GO:0030949]; protein localization to cell surface [GO:0034394]; regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043122]; regulation of macrophage migration inhibitory factor signaling pathway [GO:2000446]; regulation of protein binding [GO:0043393]; regulation of skeletal muscle satellite cell proliferation [GO:0014842]; regulation of tumor necrosis factor production [GO:0032680]; sprouting angiogenesis [GO:0002040]; Tie signaling pathway [GO:0048014]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169]; vasculogenesis [GO:0001570]; vasculogenesis involved in coronary vascular morphogenesis [GO:0060979] |
12048246_can offset VEGF-induced angiogenesis 12060561_in hypoplastic lungs, increased ANG-1 protein may be required to maintain vessel integrity and quiescence 12150788_Ang1 reduces blood-brain barrier leakage in ischemic brain and consequently decreases ischemic lesion volume. 12361603_Angiopoietin-1 can genetically rescue defective lymphatic vessels but not angiogenesis defects in knockout mice. 12427764_determination of binding sites in tie-2 receptor 12469114_Genetic engineering of precise multimers of the receptor-binding domain of angiopoietin-1 reveals that tetramers are the minimal size required for activating endothelial Tie2 receptors. 12766907_results indicated that skin tumor development occurred in a strong angiogenesis-prone scenario in which PlGF and Ang-2 acted cooperatively with VEGF, whereas the negative or stabilizing effect of Ang-1 was abrogated 12858538_Selective functions of angiopoietins and vascular endothelial growth factor on blood vessels: the concept of 'vascular domain'. 12858549_Complementary and coordinated roles of the VEGFs and angiopoietins during normal and pathologic vascular formation. 12960064_Role of ACTH in the regulation of angiogenic factors in adrenal gland. Ang/Tie2 system might represent key player for stabilization of adrenal endothelium. 14623307_angiopoietin-1 may regulate adipose tissue growth, suggesting that vascular maturation alters tissue plasticity 14991706_Angiopoietin 1 has a role in cardiovascular development, specifically the development of the epicardium and coronary vasculature 15047628_Angiopoietin-1 protein was present in the normal maturing retina and was upregulated 2.5-fold in the diabetic retina. 15501241_Ang1, in concert with VEGF, can act directly on mural cells or their precursors to facilitate their recruitment to new blood vessels 15520864_mast cell-derived Ang-1 promotes the growth of plasmacytomas by stimulating neovascularization 15637314_E2 increases Ang-1 and enhances capillary density in brain under basal conditions through estrogen receptor-alpha, priming the middle cerebral artery territory for survival after experimental focal ischemia 15648096_the timing of Ang1 expression is a critical determinate of its effects on VEGF-induced neovascularization in the retina 15690488_Ang1 overexpression increased overall vascularization in the mouse forebrain. Nonvascular events, involving alterations in the dendritic organization of layer II motor cortex neurons, dentate granule cells, and pyramidal cells of CA1, were also seen. 15705793_Ang signaling regulates LYVE-1-positive lymphatic vessel formation through Tie2 15746084_Ang1 has a role in lymphatic vessel endothelial proliferation, Tie2 expression, and VEGFR-3 upregulation 15958513_angiopoietin 1 has a potentially unique role among the vascular growth factors by acting to enlarge blood vessels without inducing sprouting. 16679392_data strongly suggest that endothelial NADPH oxidase-derived ROS play a critical role in Ang-1-induced angiogenesis 16836957_angiopoietin-1(Ang-1) expression in preimplantation mouse embryos as well as Angiopoietin-1 and -2 expression in mouse uteri is demonstrated, suggesting a possible role for angiopoietins in the embryo-maternal dialogue of the implantation 17273793_These results suggest that Shh may contribute to blood vessel growth, maturation and stabilization in a neurovascular network by reciprocally regulating the vascular morphogens Ang-1 and Ang-2 in a cell-type-specific manner. 17596403_Ang-1 confers a cytoprotective effect on islets, enhancing islet engraftment and preserving functional islet mass in transplants. 17705680_expression of VEGF, angiopoietins, and their receptors may be associated with capillary regression after hindlimb unweighting 17825261_These results indicate that angiogenesis induced by Ang1 expressed in osteoblasts is coupled with osteogenesis. 18006519_activation of VEGF/VEGFR2 and Angiopoietin/Tie2 signalling is modulated by BMP signaling-activation of endothelium 18252863_These results suggest that promotion of endothelial migration and proliferation by Ang-1 is mediated, in part, through the production of IL-8, which acts in an autocrine fashion to suppress apoptosis and facilitate cell proliferation and migration. 18310225_endotoxemia triggers functional inhibition of the Ang-1/Tie-2 receptor pathway by reducing Ang-1 and Tie-2 expression and inducing Ang-2 levels and that this response may contribute to enhanced vascular leakage in sepsis 18408125_Demonstrate that disturbed ang-1/Tie-2 signaling contributes to the hyperglycemic exacerbation of myocardial infarction and impaired angiogenesis. 18502941_role for ang1 monomers in cardiac remodeling 18505784_the proangiogenic activity of Drm is mediated by the activation of an Ang-1-dependent autocrine loop of stimulation in EC. 18556567_Disruption of Ang-1/Tie-2 signaling contributes to impaired myocardial vascular maturation and angiogenesis in type II diabetic mice subjected to myocardial ischemia. 18691637_Our data indicated that combination treatment of stroke with DETA-NONOate and bone marrow stromal cells promotes neovascularization, which is at least partially mediated by upregulation of the angiopoietin-1/Tie2 axis. 18823985_Angiogenesis and hepatic fibrosis are mutually stimulatory, such that fibrosis requires angiogenesis and angiogenesis requires angiopoietin 1 from activated HSCs. 19018775_Ang-1 alters tumor growth by stabilizing blood vessels or by promoting angiogenesis. 19217887_the precise regulation of Tie2 signaling through an Ang1/Ang2 expression switch is important to construct a mature lung vascular network system required for normal lung development. 19409199_Angiopoietin-1 significantly elevated tunica intima endothelial kinase 2 (Tie2) autophosphorylation level and prevented neural progenitor cells from oxygen/glucose-deprivation-induced apoptosis. 19733541_Ang1/Tie-2 signaling regulates myogenic precursor cell self-renewal by controlling the return to quiescence of a subset of satellite cells. 19815705_Data suggest that inhibition of Angiopoietin 2 leads to normalization of tumor blood vessels by permitting the unopposed action of Ang1, but decreases tumor vascularity primarily by blocking Ang2 actions. 20133818_Angiopoietin/Tie2 signaling transforms capillaries into venules primed for leukocyte trafficking in airway inflammation 20357108_The role of Ang-1 on SVZ neurogenesis (proliferation, differentiation, and axonogenesis)is unraveled. 20463289_increased expression of Tie2 and Ang2 in allergic airway epithelium and alveolar macrophages, but not Ang1 or VEGF correlates with the severity of airway remodeling 21113176_Ang-1 attenuated high glucose-induced endothelial cell dysfunction in mouse heart myocardial endothelial cells. 21179479_Ang-1 treatment downregulates pulmonary inflammation, VEGF and Ang-2 expression but does not protect against vascular leakage and impaired gas exchange induced by HVT-ventilation 21515377_Decreased angiopoietin1/Tie2 and increased angiopoietin2 expression may contribute to diabetes-induced vascular damage after stroke in type-2 diabetic mice. 21550017_The findings also show that the antileakage action of Ang1 requires PDGF-dependent actions of pericytes in maintaining endothelial stability. 21606590_ANG-1 is critical for regulating the number &diameter of developing vessels. It is not required for pericyte recruitment. It is dispensable in quiescent vessels but powerfully modulates the vascular response after injury. 21772310_coexpression of ANG1 with Vascular endothelial growth factor improved blood-brain barrier integrity and resulted in better neuroprotection 21809376_Unique angiogenic and vasculogenic properties of renal cell carcinoma in a xenograft model of bone metastasis are associated with high levels of vegf-a and decreased ang-1 expression. 22143796_Dopamine stabilizes tumor blood vessels by up-regulating angiopoietin 1 expression in pericytes and Kruppel-like factor-2 expression in tumor endothelial cells 22558265_Ang-1 attenuates cardiac apoptosis and promotes cardiac repair by a mechanism involving in promoting CD133(+)/c-kit(+) cells and angiogenesis in diabetic db/db mouse infarcted hearts. 22848540_LRP5-Tie2-Ang signaling axis plays a central role in control of both angiogenesis and alveolarization during postnatal lung development 22917924_Ang-1 appears important in normal lung development, and we have established its tissue-level expression and localization patterns at key time-points. 23041942_These results provide an insight into the molecular mechanisms by which Ang1 directly affects the myogenesis of striated muscle. 23149415_These data suggest that Angpt1-Tie2 signaling is crucial for the regulation of hematopoietic lineage differentiation and the function of LT-HSCs. 23171542_Vascular endothelial growth factor and Angiopoietin-1 were shown to play important roles in recovery from renal injury, particularly in the regeneration of endothelial injury. 23274064_Haptoglobin deficiency is associated with leakage-prone vessels via isolated VEGFA induction and/or suppressed Ang-1 expression, resulting in unbalanced VEGFalpha/Ang-1 transcription. 23424209_The Ang-1, Ang-2, Tie-2 system takes part in the pathologic processes in the brains that suffered acute carbon monoxide poisoning by participating in neovascularization. 23504320_ligand-independent dimerization of Tie2 is essential for a strong response upon stimulation with high dose Ang1. 24009238_Podocyte-specific inducible repletion of angiopoietin-1 in diabetic mice caused a 70% reduction of albuminuria and prevented diabetes-induced glomerular endothelial cell proliferation; hyperfiltration and renal morphology were unchanged. 24407281_findings report that Ang-1 induces arteriolar vasodilation via release of NO, suggesting that Ang-1 is an important regulator of microvascular tone. 24586553_The important role of Ang-1 in the activation of mast cells, and a potential therapeutic effect of Ang-1 on anaphylaxis diseases. 24609620_Genetic deletion of Ang1 exacerbated, while overexpression of Ang1 suppressed CNV formation and vascular leakage. 24807580_Sonic hedgehog secreted by neurons regulates ang-1 and ang-2 expression in neighboring fibroblasts. 24960457_These results suggest that PRP extract prevents endotoxin-induced pulmonary edema mainly through Ang-Tie2 signaling, and PRP extract could be a potential therapeutic strategy for sepsis-induced pulmonary edema 25072663_Myocardium-derived angiopoietin-1 is essential for coronary vein formation in the developing heart. 25608750_Ang-1 strongly enhances skeletal muscle regeneration in response to fiber injury and that this effect is mediated through induction of the myogenesis program in muscle progenitor cells and the angiogenesis program in endothelial cells. 25821987_Hematopoietic stem cells regulate the regeneration of their niche by secreting Angpt1. 26465321_COMP-Ang1 can enhance BMP2-induced cranial bone regeneration with increased pericyte recruitment. Combined delivery of the proteins might be a therapeutic strategy to repair cranial bone damage. 27454431_we demonstrate that targeted Ang-1 overexpression attenuates myofibroblast activation and interstitial collagen I accumulation, inhibits the upregulation of transforming growth factor beta1 and subsequent phosphorylation of Smad 2/3, dampens renal inflammation, and stimulates the growth of peritubular capillaries in the obstructed kidney. 27683553_These data define a role for dysregulation of the Ang-Tie-2 axis in the pathogenesis of cerebral malaria. 27894114_Hydroxysafflor yellow A promotes angiogenesis in ischemic hindlimb via the angiopoietin 1/ Tie-2 signaling pathway. 28376568_Mesenchymal stem cell microvesicles attenuate acute lung injury in mice at least partially via angiopopietin-1 mRNA. 28800750_Angiopoietin-1/Tek vascular growth factor system leads to increased metastasis without affecting primary tumor growth. 28920924_These results indicate that the Angpt-Tie2 system is essential for SC integrity. The impairment of this system underlies POAG-associated pathogenesis, supporting the possibility that Tie2 agonists could be a therapeutic option for glaucoma. 29079808_hyperoxia as well as increased TGFbeta signaling is a major contributor to the increased levels of miR34a, which mediate injury via angiopoietin-1 in neonatal lungs 29106382_By linking ANGPT1 with Primary congenital glaucoma , these results highlight the importance of ANGPT/TEK signaling in glaucoma pathogenesis and identify a candidate target for therapeutic development. 29237738_Late gestational deletion of the angiopoietin receptor endothelial tyrosine kinase 2 (Tie2) or both angiopoietin-1 and angiopoietin-2 prevents ascending vasa recta formation in mice. The AVRs are Tie2 signaling-dependent specialized hybrid vessels. 29293543_results suggest that loss of Angpt1 is central in capillary rarefaction and fibrogenesis and propose that manipulations to maintain Angpt1 levels may slow down fibrosis progression 29377123_Our study provides evidence supporting that Ang-1-modified Bone marrow mesenchymal stem cells may have preventive and therapeutic effects on hyperoxia-induced optic nerve injury in neonatal mice. 29559443_Data (including data from studies in transgenic mice) suggest that Dkk2 secreted from pericytes promotes angiogenesis and neurogenesis/neural regeneration via Angpt1/Tie2/Wnt signaling; here, overexpression of Dkk2 in diabetic mice enhances penile angiogenesis/neural regeneration and restores erectile function. (Dkk2 = dickkopf-2; Angpt1= angiopoietin-1; Tie2 = endothelial-specific receptor tyrosine kinase Tie2) 29774773_miR-711-dependent downregulation of Ang-1...may play a role in neuronal cell death after neuronal injury in vitro and after experimental traumatic brain injury. 29791855_myocardial Angpt1/endocardial Tie2 signaling in atrium promotes spatiotemporal degradation of cardiac jelly during early cardiac development. 30760202_the augmentation and downregulation of angiopoietin 1 attenuated renal damage and impaired renal recovery, respectively, by influencing the survival/regeneration of the endothelium. The manipulation of angiopoietin 1 represents a novel therapeutic approach for the treatment of ischemic kidney injury. 31581897_published rodent data suggest that angiopoietin-1 upregulation improves outcome following temporary cerebral ischaemia by reducing cerebral infarction size and improving blood-brain barrier integrity. 31678621_Study of ApoE-/- mice transfected with Ang-1 encoding lentiviral vector suggested that Ang-1 acted as promoter of atherosclerosis through reduction of macrophage cholesterol efflux via binding to Tie2. Ang-1 incubated with human THP-1 derived macrophages activated the Tie2/TFE3 pathway to reduce LXRalpha-mediated ABCA1 and ABCG1 expression and subsequent cholesterol efflux. 31685802_Inhibition of microRNA-711 limits angiopoietin-1 and Akt changes, tissue damage, and motor dysfunction after contusive spinal cord injury in mice. 32061671_accelerates Alzheimer's disease via FOXA2/PEN2/APP pathway in APP/PS1 mice 33020664_Sulfated glycans engage the Ang-Tie pathway to regulate vascular development. 33164880_Blocking exosomal miRNA-153-3p derived from bone marrow mesenchymal stem cells ameliorates hypoxia-induced myocardial and microvascular damage by targeting the ANGPT1-mediated VEGF/PI3k/Akt/eNOS pathway. 35014213_Dual Targeting of Angipoietin-1 and von Willebrand Factor by microRNA-671-5p Attenuates Liver Angiogenesis and Fibrosis. 35779860_Effect of Novel Biotherapeutic Elevating Angiopoietin 1 on Progression of Diabetic Nephropathy in Diabetic/Obese Mice. 36172864_Angiopoietin-1 Is Required for Vortex Vein and Choriocapillaris Development in Mice. |
ENSG00000154188 |
ANGPT1 |
28.780315 |
0.3348172471 |
-1.578554 |
0.48998971 |
10.289571 |
0.00133784225763393556374314385948309791274368762969970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0059759569701701188704223710601581842638552188873291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.137152064131 |
4.89431518853476 |
42.2234881425566 |
10.1955571996153 |
| ENSMUSG00000022332 |
13992 |
Khdrbs3 |
protein_coding |
Q9R226 |
FUNCTION: RNA-binding protein that plays a role in the regulation of alternative splicing and influences mRNA splice site selection and exon inclusion. Binds preferentially to the 5'-[AU]UAAA-3' motif in vitro (PubMed:19457263). Binds optimally to RNA containing 5'-[AU]UAA-3' as a bipartite motif spaced by more than 15 nucleotides (By similarity). Binds poly(A). RNA-binding abilities are down-regulated by tyrosine kinase PTK6 (PubMed:15471878). Involved in splice site selection of vascular endothelial growth factor (By similarity). In vitro regulates CD44 alternative splicing by direct binding to purine-rich exonic enhancer (By similarity). Can regulate alternative splicing of neurexins NRXN1-3 in the laminin G-like domain 6 containing the evolutionary conserved neurexin alternative spliced segment 4 (AS4) involved in neurexin selective targeting to postsynaptic partners such as neuroligins and LRRTM family members. High concentrations in forebrain structures block splicing inclusion of NRXN1-3 AS4 exons while low concentrations favor their inclusion. Targeted, cell-type specific splicing regulation of NRXN1 at AS4 is involved in neuronal glutamatergic synapse function and plasticity and is linked to behavioral aspects (PubMed:22196734, PubMed:23637638, PubMed:24469635, PubMed:27174676). Regulates expression of KHDRBS2/SLIM-1 in defined neuron populations in the hippocampus by modifying its alternative splicing resulting in a transcript predicted to undergo nonsense-mediated decay (PubMed:25505328). Can bind FABP9 mRNA (PubMed:19916944). May play a role as a negative regulator of cell growth. Inhibits cell proliferation. {ECO:0000250|UniProtKB:O75525, ECO:0000250|UniProtKB:Q9JLP1, ECO:0000269|PubMed:15471878, ECO:0000269|PubMed:19457263, ECO:0000269|PubMed:19916944, ECO:0000269|PubMed:22196734, ECO:0000269|PubMed:23637638, ECO:0000269|PubMed:24469635, ECO:0000269|PubMed:27174676}. |
Isopeptide bond;mRNA processing;Nucleus;Phosphoprotein;Reference proteome;RNA-binding;SH3-binding;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:13992; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; identical protein binding [GO:0042802]; mRNA binding [GO:0003729]; protein domain specific binding [GO:0019904]; RNA binding [GO:0003723]; SH3 domain binding [GO:0017124]; single-stranded RNA binding [GO:0003727]; mRNA processing [GO:0006397]; positive regulation of RNA splicing [GO:0033120]; regulation of alternative mRNA splicing, via spliceosome [GO:0000381]; regulation of mRNA splicing, via spliceosome [GO:0048024] |
15471878_expression of active BRK/Sik results in increased SLM-1 and SLM-2 phosphorylation which has functional relevance and leads to inhibition of their RNA-binding 19916944_10 target mRNA substrates from testis extract are bound by T-STAR. 25505328_A hierarchical, SLM2-dependent mechanism in hippocampus was identified for cell type-specific expression of neuronal splicing regulators. 26758068_Sam68 and T-STAR could regulate alternative splicing of some pre-mRNAs by bringing two distant UAA motifs into proximity and looping out regions of the pre-mRNA. 27174676_this study found that the RNA-binding protein SLM2 is essential for functional specification of glutamatergic synapses in the mouse hippocampus. 28009295_Our data reveal a pathway of SLM2 homeostatic auto-regulation controlling brain network activity and behavior 28939043_(Nrxn) is a critical element in selective trans-synaptic interactions. This study evaluated the synaptogenic receptor activity of NL1/2/3 isoforms in a neuron-fibroblast co-culture system, in which the Nrxn AS4 segments are manipulated using SLM2, a selective and dominant regulator of AS4 splicing. |
ENSG00000131773 |
KHDRBS3 |
20.049468 |
0.0871711992 |
-3.520005 |
0.75320049 |
21.960391 |
0.00000278335745570743539565867867568993432314528035931289196014404296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000228673682660925846029179031582145853462861850857734680175781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.3786614520394 |
1.8451578556174 |
38.7589190399059 |
13.1179137412009 |
| ENSMUSG00000022364 |
210544 |
Tbc1d31 |
protein_coding |
Q6NXY1 |
FUNCTION: Molecular adapter which is involved in cilium biogenesis. Part of a functional complex including OFD1 a centriolar protein involved in cilium assembly. Could regulate the cAMP-dependent phosphorylation of OFD1, and its subsequent ubiquitination by PJA2 which ultimately leads to its proteasomal degradation. {ECO:0000250|UniProtKB:Q96DN5}. |
Alternative splicing;Cell projection;Cilium biogenesis/degradation;Coiled coil;Cytoplasm;Cytoskeleton;Reference proteome;Repeat;WD repeat |
|
|
mmu:210544; |
centriolar satellite [GO:0034451]; centrosome [GO:0005813]; ciliary basal body [GO:0036064]; cytoplasm [GO:0005737]; molecular adaptor activity [GO:0060090]; cilium assembly [GO:0060271] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000156787 |
TBC1D31 |
679.799937 |
2.0632678448 |
1.044931 |
0.14108511 |
54.423927 |
0.00000000000016158344461776648166546165564412955540176709823274592281450168229639530181884765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000035413511200256390155038532306699521632134541881953282427275553345680236816406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
901.638141312827 |
88.3142777451192 |
436.995198457622 |
32.6408119688267 |
| ENSMUSG00000022367 |
15117 |
Has2 |
protein_coding |
P70312 |
FUNCTION: Catalyzes the addition of GlcNAc or GlcUA monosaccharides to the nascent hyaluronan polymer. Therefore, it is essential to hyaluronan synthesis a major component of most extracellular matrices that has a structural role in tissues architectures and regulates cell adhesion, migration and differentiation. This is one of the isozymes catalyzing that reaction and it is particularly responsible for the synthesis of high molecular mass hyaluronan (PubMed:10455188). Required for the transition of endocardial cushion cells into mesenchymal cells, a process crucial for heart development (PubMed:10930438). May also play a role in vasculogenesis. High molecular mass hyaluronan also play a role in early contact inhibition a process which stops cell growth when cells come into contact with each other or the extracellular matrix. {ECO:0000269|PubMed:10455188, ECO:0000269|PubMed:10930438}.; FUNCTION: Catalyzes the addition of GlcNAc or GlcUA monosaccharides to the nascent hyaluronan polymer. Therefore, it is essential to hyaluronan synthesis a major component of most extracellular matrices that has a structural role in tissues architectures and regulates cell adhesion, migration and differentiation (By similarity). This is one of three isoenzymes responsible for cellular hyaluronan synthesis and it is particularly responsible for the synthesis of high molecular mass hyaluronan (PubMed:10455188). {ECO:0000250|UniProtKB:Q92819, ECO:0000269|PubMed:10455188}. |
Cell membrane;Endoplasmic reticulum;Glycoprotein;Glycosyltransferase;Golgi apparatus;Isopeptide bond;Lysosome;Membrane;Phosphoprotein;Reference proteome;Transferase;Transmembrane;Transmembrane helix;Ubl conjugation |
PATHWAY: Glycan biosynthesis; hyaluronan biosynthesis. {ECO:0000269|PubMed:10455188}. |
|
mmu:15117; |
cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; endoplasmic reticulum membrane [GO:0005789]; extracellular vesicle [GO:1903561]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; lysosome [GO:0005764]; plasma membrane [GO:0005886]; plasma membrane raft [GO:0044853]; hyaluronan synthase activity [GO:0050501]; identical protein binding [GO:0042802]; atrioventricular canal development [GO:0036302]; bone morphogenesis [GO:0060349]; cellular response to fluid shear stress [GO:0071498]; cellular response to interleukin-1 [GO:0071347]; cellular response to platelet-derived growth factor stimulus [GO:0036120]; cellular response to tumor necrosis factor [GO:0071356]; endocardial cushion to mesenchymal transition [GO:0090500]; estrous cycle [GO:0044849]; extracellular matrix assembly [GO:0085029]; extracellular polysaccharide biosynthetic process [GO:0045226]; hyaluronan biosynthetic process [GO:0030213]; hyaluronan metabolic process [GO:0030212]; kidney development [GO:0001822]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of hyaluronan biosynthetic process [GO:1900127]; positive regulation of keratinocyte migration [GO:0051549]; positive regulation of keratinocyte proliferation [GO:0010838]; positive regulation of monocyte aggregation [GO:1900625]; positive regulation of smooth muscle cell migration [GO:0014911]; positive regulation of substrate adhesion-dependent cell spreading [GO:1900026]; positive regulation of urine volume [GO:0035810]; regulation of extracellular matrix assembly [GO:1901201]; renal water absorption [GO:0070295]; vasculogenesis [GO:0001570] |
14966127_Expression of the mouse HAS2 gene in Drosophila tissues by the Gal4/UAS system resulted in massive HA accumulation in the extracellular space,supporting the idea that in vivo HA biosynthesis does not require molecules other than the HAS protein 15531364_Has2 expression was impaired in oocyte-cumulus cell complexes in knockout mice. 15765504_strongly, albeit transiently, expressed in numerous embryonic tissues 15843373_natural antisense mRNAs of HAS2 may have an important and novel regulatory role in the control of HAS2, hyaluronan biosynthesis, and HA-dependent cell functions 19320776_repression of HA-accumulation by both COX-2 selective and non-selective COX inhibition implicates COX-2 in the regulation of HA synthesis via stimulation of HAS1 and HAS2 expression in vivo 19633173_Has2 deficiency illustrates the crucial role of hyaluronan in skeletal growth, patterning, chondrocyte maturation and synovial joint formation in the developing limb. 19846762_Tbx2, a central intermediary of Bmp-Smad signaling, has a central part in directing Has2 and Tgfbeta2 expression, facilitating endocardial cushions formation. 20507985_hyaluronan synthase 2 activity is regulated by dimerization and ubiquitination 21200430_PDGF-BB stimulates cultured cardiomyocytes to synthesize the extracellular matrix component hyaluronan via HAS2 21224752_Data show that Has2 knockout mice died near birth and displayed severe abnormality in skeletal development. 21246657_Data suggest that hyaluronan production by Has2 in chondrocytes is not only essential for formation of an organized growth plate and subsequent long bone growth but also for normal modeling of the diaphyseal bone. 21778427_Inhibition of Has2 expression and extracellular hyaluronic acid production requires MiR23 in the embryonic heart to restrict endocardial cushion formation. 22383528_NSMase2/Cer are the key mediators of the regulation of HA synthesis, via microdomains and the Akt/mTOR pathway 22529214_analysis of changes in cervical glycosaminoglycan composition during normal pregnancy and preterm birth: Has1 is expressed in preterm birth, while Has2 is induced at term 22785117_HAS2 was significantly upregulated at the level of gene expression during muscle hypertrophy. 23313125_This study identifies Has2 as a novel downstream target of Shh signaling required for joint patterning and chondrogenesis. 23391595_This study demonistrated that Has2 expression in adult mouse subventricular zone and rostral migratory stream and in ischemic cortex. 24472738_Stimulation with LPS caused rapid increases in versican mRNA and protein, a rapid increase in Has1 mRNA, and concomitant inhibition of hyaluronidases 1 and 2, the major hyaluronan degrading enzymes 25163516_Has2 expression and hyaluronan produced at the tips of epithelial cells play a critical role in driving tubulogenesis and branching in vitro. 25264594_mir-23a-3p causes cellular senescence by targeting hyaluronan synthase 2: possible implication for skin aging. 25266724_Hyaluronan synthase 2 has a role in protecting skin fibroblasts against apoptosis induced by environmental stress 25555205_these results highlight the role of nSMase2 in apoptosis evoked by nutrient starvation that could contribute to the delayed apoptosis of hypertrophic chondrocytes in the growth plate, and emphasize the antiapoptotic properties of HAS2 26521733_the ineffective repair of injured cartilage in Has1(-/-) joints can be at least partly explained by the markedly enhanced expression of particular genes in pathways linked to ECM turnover, IL-17/IL-6 cytokine signaling, and apoptosis. 27289075_Has2 mRNA was expressed in the surrounding mesenchyme from E12.0 to 18.0 in both molar and incisor tooth germs, but disappeared after birth. 27694932_Expression of the innate immune receptor Toll-like receptor 4 (TLR4) and the extracellular matrix glycosaminoglycan hyaluronan (HA) on type 2 alveolar epithelial cells (AEC@2) are important for AEC2 renewal, repair of lung injury and limiting the extent of fibrosis. Either deletion of TLR4 or HA synthase 2 in surfactant-protein-C-positive AEC2s leads to impaired renewal capacity, severe fibrosis and mortality. 28787175_allergen-challenged mice that overexpress HAS2 in myofibroblasts and smooth muscle cells develop increased airway fibrosis, which lessens airway hyperresponsiveness to bronchoconstrictors. 34542891_WNT5A Enhances LH-Mediated Expression of HAS2 in Granulosa Cells. 35069543_Has2 Regulates the Development of Ovalbumin-Induced Airway Remodeling and Steroid Insensitivity in Mice. 35662287_Hyaluronan synthase 2, a target of miR-200c, promotes carbon tetrachloride-induced acute and chronic liver inflammation via regulation of CCL3 and CCL4. |
ENSG00000170961 |
HAS2 |
103.228579 |
2.1126537615 |
1.079056 |
0.25190950 |
18.209045 |
0.00001979365947627982058316768732009194309284794144332408905029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001387469034250778598341830027962373605987522751092910766601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
136.300131007146 |
23.9205355946706 |
64.5160761740201 |
8.80920517832953 |
| ENSMUSG00000022382 |
22422 |
Wnt7b |
protein_coding |
P28047 |
FUNCTION: Ligand for members of the frizzled family of seven transmembrane receptors that functions in the canonical Wnt/beta-catenin signaling pathway (PubMed:15923619, PubMed:28803732). Required for normal fusion of the chorion and the allantois during placenta development (PubMed:11543617). Required for central nervous system (CNS) angiogenesis and blood-brain barrier regulation (PubMed:28803732). {ECO:0000269|PubMed:11543617, ECO:0000269|PubMed:15923619, ECO:0000269|PubMed:28803732}. |
Developmental protein;Disulfide bond;Extracellular matrix;Glycoprotein;Lipoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:22422; |
cell surface [GO:0009986]; cytoplasm [GO:0005737]; endoplasmic reticulum lumen [GO:0005788]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; chemoattractant activity involved in axon guidance [GO:1902379]; cytokine activity [GO:0005125]; frizzled binding [GO:0005109]; receptor ligand activity [GO:0048018]; anatomical structure regression [GO:0060033]; angiogenesis [GO:0001525]; apoptotic process involved in blood vessel morphogenesis [GO:1902262]; canonical Wnt signaling pathway [GO:0060070]; cell fate commitment [GO:0045165]; cell population proliferation [GO:0008283]; cell proliferation in forebrain [GO:0021846]; cellular oxygen homeostasis [GO:0032364]; central nervous system vasculogenesis [GO:0022009]; chemoattraction of dopaminergic neuron axon [GO:0036516]; chorio-allantoic fusion [GO:0060710]; developmental growth involved in morphogenesis [GO:0060560]; embryonic organ development [GO:0048568]; embryonic placenta morphogenesis [GO:0060669]; establishment or maintenance of polarity of embryonic epithelium [GO:0016332]; forebrain regionalization [GO:0021871]; homeostatic process [GO:0042592]; in utero embryonic development [GO:0001701]; inner medullary collecting duct development [GO:0072061]; lobar bronchus development [GO:0060482]; lung development [GO:0030324]; lung epithelium development [GO:0060428]; lung morphogenesis [GO:0060425]; lung-associated mesenchyme development [GO:0060484]; metanephric collecting duct development [GO:0072205]; metanephric epithelium development [GO:0072207]; metanephric loop of Henle development [GO:0072236]; metanephros morphogenesis [GO:0003338]; negative regulation of neurogenesis [GO:0050768]; negative regulation of smoothened signaling pathway [GO:0045879]; neuron differentiation [GO:0030182]; neuron projection development [GO:0031175]; neuron projection morphogenesis [GO:0048812]; odontogenesis of dentin-containing tooth [GO:0042475]; osteoblast differentiation [GO:0001649]; outer medullary collecting duct development [GO:0072060]; planar cell polarity pathway involved in axon guidance [GO:1904938]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of gene expression [GO:0010628]; positive regulation of JNK cascade [GO:0046330]; positive regulation of osteoblast differentiation [GO:0045669]; regulation of cell projection size [GO:0032536]; renal inner medulla development [GO:0072053]; renal outer medulla development [GO:0072054]; smooth muscle cell differentiation [GO:0051145]; stem cell development [GO:0048864]; synapse organization [GO:0050808]; trachea cartilage morphogenesis [GO:0060535]; vasculature development [GO:0001944]; Wnt signaling pathway [GO:0016055] |
11914369_promoter is regulated by TTF-1, GATA6, and Foxa2 in lung epithelium 12361974_Wnt7b signaling is required for proper lung mesenchymal growth and vascular development 12843296_Wnt 7a, Wnt 7b, and Sonic hedgehog (Shh) promote progenitor cell maturation in explant cultures, as reported for FGF2. Wnts 7a and 7b also stimulate proliferation of neurogenic progenitors 15923619_Data demonstrate that Wnt7b signals through Fzd1 and -10 and LRP5 and implicate these Wnt coreceptors in the regulation of lung airway and vascular development. 16163358_macrophage WNT7b is a short-range paracrine signal required for WNT-pathway responses and programmed cell death in the vascular endothelial cells of the temporary hyaloid vessels of the developing eye 17199045_Wnt3a signals through the Galpha(q/11) subunits of G proteins to activate phosphatidylinositol signaling and PKCdelta and Wnt7b, expressed by osteogenic cells in vivo, induces osteoblast differentiation in vitro via the PKCdelta-mediated pathway. 17510243_hCG was found to up-regulate these wnt ligands in mouse mammary gland, independent of the changes in ovarian steroidogenesis 17804636_The olfactory placode expresses beta catenin-activating Wnt genes, and the Frizzled7 receptor transduces the 'canonical' Wnt signal; we show that Wnt5a and Wnt7b are sufficient to rescue beta catenin activation in the absence of incoming axons 18045882_phenotypic shift in mesenchymal progenitor cell commitment was associated with reciprocal regulation of early adipogenic and osteoblastogenic transcription factors and with a reduction in Wnt7b and Wnt10b mRNA and beta-catenin protein levels 18367557_Wnt7b signals to neighboring cells to activate both autocrine and paracrine canonical Wnt signaling cascades. In contrast to results from hypomorphic models, Wnt7b modulates several important signaling pathways in the lung. 19023080_CNS uses the canonical Wnt signaling pathway to promote formation and CNS-specific differentiation of the organ's vasculature 19060336_These findings demonstrate that Wnt7b is a key regulator of the tissue architecture that establishes a functional physiologically active mammalian kidney. 19176588_alpha3beta1 integrin, a major laminin receptor, and c-Met, the receptor for hepatocyte growth factor, signal coordinately to regulate the expression of Wnt7b in the papilla of the developing kidney 19690384_Wnt7b regulates a program of mesenchymal differentiation in the lung that is essential for SMC development. 19734317_These data demonstrate that mesenchymal stem cells from mice and humans produce Wnt proteins and TGF-beta1 that respectively stimulate lung fibroblast proliferation and matrix production. 20160075_Because Wnt7b is known to stimulate epithelial responses during kidney development, these findings suggest that macrophages are able to rapidly invade an injured tissue and reestablish a developmental program that is beneficial for repair and regeneration 21106844_Both Wnt5a and Wnt7b are expressed in gradients along the anterior-posterior axis of the brainstem, consistent with their role as directional cues. 21354128_Dkk1 controls the degree of Wnt-7b signalling in the papilla to coordinate kidney organogenesis. 22677559_Hyperactivated c-Met led to increased NF-kappaB signaling, which in turn, drove de novo expression of Wnt7a and overexpression of Wnt7b in Pkd1(-/-) mouse kidneys. 22833126_Epithelial Wnt7b and Wnt9b as possible ligands of Fzd1-mediated beta-catenin (Ctnnb1)-dependent (canonical) Wnt signaling in the undifferentiated ureteric mesenchyme. 22949635_Pdgf signaling potentiates Wnt2-Wnt7b signaling to promote high levels of Wnt activity in mesenchymal progenitors that is required for proper development of endoderm-derived organs, such as the lung 23685555_Wnt7b preserves endothelial phenotype in aortic endothelial cells. 24222445_These findings demonstrate the importance of intrinsic Wnt7b expression in hfSCs regulation and normal HF cycling 24497849_Thus, WNT7B promotes bone formation in part through mTORC1 activation. 24638982_Results illustrated the critical role of myeloid WNT7B in tumor progression, acting at the levels of angiogenesis, invasion, and metastasis. 24727668_epithelial Wnt7a and Wnt7b are possible ligands of Fzd-mediated beta-catenin (Ctnnb1)-dependent (canonical) Wnt signaling in the adjacent undifferentiated periotic mesenchyme of fibrocytes in the murine inner ear 24953933_Transgenic Wnt7b overexpression in neural progenitor cells impairs neuronal differentiation and the development of forebrain structures at embryonic day 10.5. It decreased expression of Tbr1 and Tbr2, in both progenitor cells and post-mitotic neurons. 25351956_Stable ectopic expression of Wnt-7b in OSCC cells overexpressing miR329 or miR410 restored proliferation and invasion capabilities abolished by these miRNA 25576928_Wnt7b is required for epithelial progenitor growth and operates during epithelial-to-mesenchymal signaling in pancreatic development. 25823570_In a mouse model of myopia, there was downregulation of WNT7B expression in the cornea and upregulation in the retina, suggesting its possible role in the development of myopia. 26548512_the present study detected abnormal upregulation in the levels of Wnt2b and Wnt7b, and hypothesized that the alterations may be due to the ectopic opening of chromatin structure. 26939553_Celsr3 and Fzd3 enable immature neurons to respond to Wnt7, upregulate Jag1 and thereby facilitate feedback signals that tune the timing of neural progenitor cell fate decisions via Notch activation. 28392344_Ptgs2 inhibitor celecoxib reduces dsRNA-induced WIHN and Wnt7b, and exogenous prostaglandin E2 can rescue WIHN and Wnt7b. Although other Wnts and pathways likely contribute, these results highlight noncoding dsRNA as an upstream coordinator of prostaglandin and Wnt levels in regeneration. 28803732_experiments indicate that Reck and Gpr124 are part of the cell surface protein complex that transduces Wnt7a- and Wnt7b-specific signals in mammalian CNS epithelial cells to promote angiogenesis and regulate the BBB 29898920_Wnt7b and Fz7 induce the phosphorylation of Ca(2+)/calmodulin-dependent protein kinase II (CaMKII) and JNK proteins, which are required for dendritic development. 30113308_We identified Wnt7A and Wnt7B as major Wnts for nerve terminal development in rescue experiments. These observations demonstrate a necessary role of motoneuron Wnts in neuromuscular junction (NMJ) development, in particular presynaptic differentiation 30478038_experiments define the extent of CNS region-specific cooperation for several components of the Norrin and Wnt7a/Wnt7b systems, and they reveal substantial regional heterogeneity in the extent to which partially redundant ligands, receptors, and coactivators maintain the blood-brain barrier and blood-retina barrier 30498093_Long noncoding RNA OIP5-AS1 targets Wnt-7b to affect glioma progression via modulation of miR-410. 30913395_Compound transgenic mice overexpress Wnt7b transiently in the osteoblast lineage of postnatal mice, with or without concurrent deletion of Glut1. Wnt7b overexpression stimulated bone formation, but this was abolished without Glut1. Wnt7b increased Glut1 expression and glucose consumption in the primary cultured osteoblast lineage cells. Wnt7b promotes bone formation in part through stimulating glucose metabolism in them. 31181411_Wnt7b enhances dentinogenesis by increasing the proliferation and differentiation of dental mesenchymal cells partly through ERK1/2 pathway 31358627_Our results suggest that aberrant Wnt7b expression activated c-jun N-terminal kinase (JNK), which down-regulated mitochondria numbers by suppressing Ppargc1a Down-regulation of Ppargc1a probably decreased reactive oxygen species and enhanced glycolysis 32346881_Wnt7b-induced Sox11 functions enhance self-renewal and osteogenic commitment of bone marrow mesenchymal stem cells. 35552394_The WNT7A/WNT7B/GPR124/RECK signaling module plays an essential role in mammalian limb development. |
ENSG00000188064 |
WNT7B |
9.756883 |
0.0975486340 |
-3.357735 |
1.14324045 |
8.365912 |
0.00382324658641182554483228805963790364330634474754333496093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0147320242864216865291693281392326753120869398117065429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.93029825944826 |
1.69482529753416 |
19.7880603843255 |
10.3213490735135 |
| ENSMUSG00000022385 |
29870 |
Gtse1 |
protein_coding |
Q8R080 |
FUNCTION: May be involved in p53-induced cell cycle arrest in G2/M phase by interfering with microtubule rearrangements that are required to enter mitosis. Overexpression delays G2/M phase progression. |
Cytoplasm;Cytoskeleton;Microtubule;Phosphoprotein;Reference proteome |
|
|
mmu:29870; |
cytoplasmic microtubule [GO:0005881]; microtubule cytoskeleton [GO:0015630]; microtubule binding [GO:0008017] |
31932847_Clathrin's adaptor interaction sites are repurposed to stabilize microtubules during mitosis. |
ENSG00000075218 |
GTSE1 |
367.841695 |
2.2972681183 |
1.199919 |
0.14533811 |
67.352048 |
0.00000000000000022710598245611615104960296836802276570487375264781326755780810344731435179710388183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000065392784967326299395672993974557666169980171821940473364520585164427757263183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
514.394913432841 |
51.9081996391275 |
223.915924025485 |
17.5037704993108 |
| ENSMUSG00000022412 |
239555 |
Mief1 |
protein_coding |
Q8BGV8 |
FUNCTION: Mitochondrial outer membrane protein which regulates mitochondrial fission. Promotes the recruitment and association of the fission mediator dynamin-related protein 1 (DNM1L) to the mitochondrial surface independently of the mitochondrial fission FIS1 and MFF proteins. Regulates DNM1L GTPase activity and DNM1L oligomerization. Binds ADP and can also bind GDP, although with lower affinity. Does not bind CDP, UDP, ATP, AMP or GTP. Inhibits DNM1L GTPase activity in the absence of bound ADP. Requires ADP to stimulate DNM1L GTPase activity and the assembly of DNM1L into long, oligomeric tubules with a spiral pattern, as opposed to the ring-like DNM1L oligomers observed in the absence of bound ADP. Does not require ADP for its function in recruiting DNM1L. {ECO:0000269|PubMed:23283981, ECO:0000269|PubMed:24508339}. |
3D-structure;Direct protein sequencing;Membrane;Mitochondrion;Mitochondrion outer membrane;Nucleotide-binding;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:239555; |
mitochondrial large ribosomal subunit [GO:0005762]; mitochondrial matrix [GO:0005759]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; ADP binding [GO:0043531]; GDP binding [GO:0019003]; identical protein binding [GO:0042802]; cellular response to hypoxia [GO:0071456]; mitochondrial fission [GO:0000266]; positive regulation of mitochondrial fission [GO:0090141]; positive regulation of mitochondrial translation [GO:0070131]; positive regulation of protein targeting to membrane [GO:0090314] |
23921378_MiD49 and MiD51 can act independently of Mff and Fis1 in Drp1 recruitment and suggest that they provide specificity to the division of mitochondria. 24508339_ADP is an essential cofactor for MiD51 during mitochondrial fission. 26101352_This paper combines confocal live-cell imaging with correlative cryogenic fluorescence microscopy and soft x-ray tomography to link MiD49 and MiD51 to the involvement of the endoplasmic reticulum in mitochondrial fission. 27076521_MiD51 can suppress Mff-dependent enhancement of Drp1 GTPase activity. |
ENSG00000100335 |
MIEF1 |
337.718184 |
2.4650265799 |
1.301603 |
0.20747192 |
38.287950 |
0.00000000061038406474110014936687898965349784174971858874414465390145778656005859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000088210342259358983991209527840068882476032285921974107623100280761718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
508.129237274604 |
73.3254731864156 |
206.135398872334 |
22.23697099032 |
| ENSMUSG00000022449 |
223838 |
Adamts20 |
protein_coding |
P59511 |
FUNCTION: May play a role in tissue-remodeling process occurring in both normal and pathological conditions. May have a protease-independent function in the transport from the endoplasmic reticulum to the Golgi apparatus of secretory cargos, mediated by the GON domain. |
Alternative splicing;Cleavage on pair of basic residues;Disulfide bond;Extracellular matrix;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Repeat;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes a member of 'a disintegrin and metalloproteinase with thrombospondin motifs' (ADAMTS) family of multi-domain matrix-associated metalloendopeptidases that have diverse roles in tissue morphogenesis and pathophysiological remodeling, in inflammation and in vascular biology. The encoded preproprotein undergoes proteolytic processing to generate an active protease. Certain mutations in this gene cause defective development of neural crest-derived melanoblasts resulting in a 'belted' phenotype that is characterized by white spots in the lumbar region. [provided by RefSeq, Jul 2016]. |
mmu:223838; |
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; endopeptidase activity [GO:0004175]; metalloendopeptidase activity [GO:0004222]; zinc ion binding [GO:0008270]; apoptotic process [GO:0006915]; extracellular matrix organization [GO:0030198]; melanocyte differentiation [GO:0030318]; negative regulation of apoptotic process [GO:0043066]; positive regulation of melanocyte differentiation [GO:0045636]; positive regulation of signal transduction [GO:0009967]; proteolysis [GO:0006508]; regulation of developmental pigmentation [GO:0048070]; signal transduction [GO:0007165] |
12514189_ADAMTS-9 and ADAMTS-20 have an identical modular structure, are distinct in possessing 15 thrombospondin type I repeats and a unique C-terminal domain, and have a similar gene structure 12562771_This protein is identified and characterized and its relation to other disintegrins and thrombospondin 1 repeats is examined. 12925592_Results suggest that the role of ADAMTS proteases, including ADAMTS20, in the regulation of cell migration has been conserved in mammalian development. 18454205_Adamts20 mutations in belted (bt) mice cause white spotting of the dorsal and ventral torso, indicative of defective neural crest (NC)-derived melanoblast development. 19922873_show that combinatorial mouse alleles for the secreted metalloproteases Adamts5, Adamts20 (bt), and Adamts9 result in fully penetrant soft-tissue syndactyly. 21041365_These findings support a model in which cooperative versican proteolysis by ADAMTS9 in vascular endothelium and by ADAMTS20 in palate mesenchyme drives palatal shelf sculpting and extension. 30814516_ADAMTS9 or ADAMTS20 mutagenesis impaired neural and yolk sac ciliogenesis, leading to morphogenetic anomalies resulting from impaired hedgehog signaling, which is transduced by primary cilia. 31600785_Results provide compelling evidence that ADAMTS9 and ADAMTS20 were differentially sensitive to B3GLCT inactivation and suggest that the developmental defects in PTRPLS result from disruption of a subset of highly sensitive POFUT2/B3GLCT targets such as ADAMTS20. |
ENSG00000173157 |
ADAMTS20 |
38.430523 |
14.1437872780 |
3.822097 |
0.59532658 |
42.181325 |
0.00000000008319067088834800622588205596392280614936876048659541993401944637298583984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000013380784572030800031719379589167928656934236641973257064819335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.5099042999964 |
28.2307918166954 |
4.70241124194774 |
1.71832259079361 |
| ENSMUSG00000022483 |
12824 |
Col2a1 |
protein_coding |
P28481 |
FUNCTION: Type II collagen is specific for cartilaginous tissues. It is essential for the normal embryonic development of the skeleton, for linear growth and for the ability of cartilage to resist compressive forces. |
3D-structure;Alternative splicing;Calcium;Collagen;Disease variant;Disulfide bond;Extracellular matrix;Glycoprotein;Hydroxylation;Metal-binding;Reference proteome;Repeat;Secreted;Signal |
|
This gene encodes the alpha-1 subunit of the fibril-forming type II collagen, the major component of cartilage and the vitreous humor of the eye. The encoded preproprotein forms homotrimeric, triple helical procollagen that undergoes proteolytic processing during fibirl formation. Mice harboring certain mutations in this gene exhibit severe chondrodysplasia characterized by short limbs and trunch, craniofacial deformities and cleft palate. A complete lack of the encoded protein in mice results in postnatal lethality. Alternative splicing results in multiple transcript variants encoding different isoforms that may undergo similar proteolytic processing. [provided by RefSeq, Dec 2015]. |
mmu:12824; |
basement membrane [GO:0005604]; collagen trimer [GO:0005581]; collagen type II trimer [GO:0005585]; collagen type XI trimer [GO:0005592]; collagen-containing extracellular matrix [GO:0062023]; cytoplasm [GO:0005737]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; extracellular matrix structural constituent [GO:0005201]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; MHC class II protein binding [GO:0042289]; platelet-derived growth factor binding [GO:0048407]; proteoglycan binding [GO:0043394]; anterior head development [GO:0097065]; bone development [GO:0060348]; cartilage condensation [GO:0001502]; cartilage development [GO:0051216]; cartilage development involved in endochondral bone morphogenesis [GO:0060351]; cellular response to BMP stimulus [GO:0071773]; central nervous system development [GO:0007417]; chondrocyte differentiation [GO:0002062]; collagen fibril organization [GO:0030199]; embryonic skeletal joint morphogenesis [GO:0060272]; endochondral ossification [GO:0001958]; extracellular matrix organization [GO:0030198]; extrinsic apoptotic signaling pathway in absence of ligand [GO:0097192]; heart morphogenesis [GO:0003007]; inner ear development [GO:0048839]; inner ear morphogenesis [GO:0042472]; limb bud formation [GO:0060174]; limb morphogenesis [GO:0035108]; negative regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001240]; notochord development [GO:0030903]; ossification [GO:0001503]; otic vesicle development [GO:0071599]; proteoglycan metabolic process [GO:0006029]; regulation of gene expression [GO:0010468]; roof of mouth development [GO:0060021]; sensory perception of sound [GO:0007605]; skeletal system development [GO:0001501]; skeletal system morphogenesis [GO:0048705]; tissue homeostasis [GO:0001894]; visual perception [GO:0007601] |
12381733_a new long form cooperates with Sox9 to activate the type II collagen gene. (Lc-maf protein) 12917416_Snail family members, Snail (Sna) and Slug (Slugh), are involved in chondrocyte differentiation by controlling the expression of type II collagen (Col2a1) and aggrecan. 12968670_A missense mutation in the Col2a1 gene has been discovered, resulting in a phenotype of spondyloepiphyseal dysplasia congenita. Adult homozygous mutant animals have shortened noses, dysplastic vertebrae, femora, and tibias, retinoschisis and hearing loss. 14614991_Col2a1 mutant mice have a complete absence of mature type II collagen in the homozygote cartilage and an insufficiency of type II collagen in the heterozygote cartilage 15778499_MINT is involved in CRYBP1-mediated Col2a1 gene repression and may play a role in regulation of cartilage development 16076844_cis elements in the COL2A1 gene modulate the cell type-specific alternative splicing switch of exon 2 during cartilage development 17519362_IGF-I from cells expressing procollagen type IIalphaI regulates bone accretion that occurs during postnatal growth period. 17591962_this novel form of combined pituitary hormone deficiency is characterized by the perturbation of the hypothalamus-pituitary growth axis, associated with retarded chondrocyte development and enchondral ossification in the epiphyseal growth plate 17652426_Type II collagen proteolysis is an important determinant for the skeletal outgrowth in mouse embryos through modulation of chondrocyte survival and cartilagenous growth. 18579360_Data demonstrate that conditional ablation of the Kif3a or Pkd1 genes encoding primary cilium/intraflagellar transport-associated proteins using type II collagen-specific Cre transgenic strain results in a severe form of polycystic kidney disease in mice. 18678883_C-propeptide mutation could interrupt the normal assembly and secretion of Type II procollagen trimers, resulting in a buildup of proalpha1(II) chains in the RER and a reduced rate of matrix synthesis. 19419317_MT1-MMP activity in the type II collagen-expressing cells of the skeleton rescues not only diminished chondrocyte proliferation, but also results in amelioration of the severe skeletal dysplasia associated with MT1-MMP deficiency. 19441084_Data reveal that COL2 is attacked at a susceptible peptide bond at the surface of developing articular cartilage canals, and is cleaved by MMP-13. 19478656_Results show that in Smad3 gene knock-out mice the protein positive staining of type II collagen, aggrecan, and TGF-beta1 in the disc decreased, while that of type X collagen increased. 19710088_decreased production in LPS-treated chondrocytes treated with low molecular mass hyaluronan 20129696_The relative expressions of type IIB to type IIA procollagens are good markers to define the differentiation state of chondrocytes. 20403959_cartilaginous regeneration is due to the expression of chondrogenic factors, such as Sox 9 and type II and X collagens 20414703_Down-regulation of Col2a1 is associated with autoimmune type 1 diabetes mellitus. 20513134_Study identified 57 novel mutations including missense changes in both COL2A1 and COL11A1. 20730911_Data show that procollagen IIA (IIA), an isoform of the cartilage extracellular matrix protein encoded by an alternatively spliced transcript of Col2a1, is expressed in the prechordal plate and the anterior definitive endoderm. 21177286_beta2-adrenergic receptors inhibit the expression of collagen type II in growth plate chondrocytes by stimulating the AP-1 factor Jun-B. 21940677_B cell-reactive epitopes on collagen II (CII)are largely shared between human rheumatoid arthritis and rodent CII-induced arthritis. 22028304_A mouse model with a Ser1386Pro mutation of the Col2a1 C-propeptide domain that results in abnormal collagen processing and phenotypic features consistent with spondyloepiphyseal dysplasia congenita and secondary osteoarthritis has been established. 22049074_RB1CC1 protein suppresses type II collagen synthesis in chondrocytes and causes dwarfism 22072985_SOX9 regulates chondrocyte differentiation, proliferation.. It directly transactivates Col2a1 enhancing this gene's expression in immature/proliferating chondrocytes. 22815835_Lef1 mediated looping between promoter and 3' UTR under the permissive chromatin architecture upregulates Col2a1 expression in primary chondrocytes. 23133648_Type II collagen induces peripheral tolerance in BALB/c mice via the generation of CD8+ T regulatory cells 23239042_An autoimmune response to CII can disrupt articular cartilage, particularly that of the small joints, and the subsequent integrity of the cartilage depends on a balance between breakdown and repair. 24113490_characterization of the Col2a1(+ex2) mice has surprisingly shown that disruption of alternative splicing does not affect overt cartilage formation 24168833_The mutant G1170S Col2a1 gene in mice clearly alters the transgenic murine phenotype and cartilage matrix homeostasis. 24344131_C/EBPbeta represses the transcriptional activity of Col2a1 both directly and indirectly through modulation of Sox9 expression in chondrocytes. 24352615_regulates Mac-1-mediated complement-dependent phagocytosis 24475193_Endoplasmic reticulum stress-unfolding protein response-apoptosis cascade causes chondrodysplasia in a col2a1 p.Gly1170Ser mutated mouse model. 24735995_expression of the Col2a1 IIC splice form may have a regulatory function in controlling alternative splicing of exon 2 to generate defined proportions of IIA, IID and IIB procollagen isoforms during cartilage development 25211510_Results indicate the ability of type II collagen (CII) to generate CII-specific anterior chamber-associated immune deviation (ACAID)-mediated immune tolerance in vivo and in vitro in both DBA/1 mice and C57BL/6 mice. 25451152_a mouse of model of spondyloepiphyseal dysplasia is associated with the R992C substitution in collagen II 25541700_The MC13 mutation in Col2a1 causes an incompletely penetrant splicing defect. 25684979_The increased expressions of the cartilage-associated proteins Collagen2 and alpha-SMA during scleral chondrogenesis accompany myopia development. 25703139_Data indicate that tamoxifen decreased collage type II COL2A1 by activating matrix metallopeptidases MMP-2/13 expression. 26362410_Col2 expression was seen in the prehypertrophic zone of the mandibular condyle cartilage. 26676803_this study shows role of Del-1 in the regulation of platelet-monocyte interplay and the subsequent heterotypic aggregate formation in the blood-mediated inflammatory reaction in islet transplantation 26806019_Alpha-tectorin and beta-tectorin crosslink type II collagen fibrils and connect the tectorial membrane to the spiral limbus. 27012200_Rspo2 activates Wnt/beta-catenin signaling to reduce Col2a1 and Sox9 and to facilitate differentiation of proliferating chondrocytes into hypertrophic chondrocytes in growth cartilage 27881681_These findings suggest a novel mechanism of action of SOX5/6; namely, the SOX9/5/6 combination enhances Col2a1 transcription through a novel enhancer in intron 6 together with the enhancer in intron 1. 28796274_this study's findings indicate that endothelial Del-1 not influence atherosclerosis development in ApoE(-/-) mice 29207013_Kartogenin effectively increased the expression of Col II and aggrecan in hNPCs and slowed the degeneration of intervertebral discs stimulated by IL-1b and TNF-a 29230102_found bone destruction and bone loss phenotype in vertebral bone in beta-catenin(Col2CreER) KO mice 29322554_Detection of transcripts of Collagen type2 A1 (Col2a1) revealed that both e trabecular cartilageand the anterior part of the hypophyseal cartilage developing anterior to the persistent epithelial stalk of the anterior lobe of the pituitary gland were suppressed in the Foxc1(ch/ch) . 29660427_blocking the expression of the R992C collagen II mutant at the early prenatal stages leads to long-term positive effects. 29787757_Overexpression of Dlx2 enhances early chondrocyte differentiation by increasing accumulation of Col2alpha1, type II collagen and aggrecan, but interferes later stages of chondrocyte differentiation through inhibiting MMP13 expression. 30078186_Col2a1 positive cells were found to be specifically distributed in the superficial and inner zones of the anterior horn, as well as the inner zone of the posterior horn in mice, indicating that Col2a1-CreER(T2) mice can be a used for studying gene function in menisci 31381970_Data suggest that collagen II matrikines stimulate angiogenesis in skeletal growth and development, revealing novel strategies for stimulating angiogenesis in other contexts such as fracture healing and surgical applications. 31958497_Skeletal deterioration in COL2A1-related spondyloepiphyseal dysplasia occurs prior to osteoarthritis. 32354322_Conditional deletion of HDAC4 from collagen type 2alpha1-expressing cells increases angiogenesis in vivo. 33397982_TARM1 contributes to development of arthritis by activating dendritic cells through recognition of collagens. |
ENSG00000139219 |
COL2A1 |
2267.815502 |
0.2091470166 |
-2.257411 |
0.07210121 |
945.190886 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000147274955328490147404740585211647934555312985482990470723 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000028041151494544523733888327655213846889082678136352401607 |
Yes |
No |
776.139187627779 |
38.8050610316404 |
3710.97422439522 |
129.510570230172 |
| ENSMUSG00000022512 |
12737 |
Cldn1 |
protein_coding |
O88551 |
FUNCTION: Claudins function as major constituents of the tight junction complexes that regulate the permeability of epithelia. While some claudin family members play essential roles in the formation of impermeable barriers, others mediate the permeability to ions and small molecules. Often, several claudin family members are coexpressed and interact with each other, and this determines the overall permeability. CLDN1 is required to prevent the paracellular diffusion of small molecules through tight junctions in the epidermis and is required for the normal barrier function of the skin. Required for normal water homeostasis and to prevent excessive water loss through the skin, probably via an indirect effect on the expression levels of other proteins, since CLDN1 itself seems to be dispensable for water barrier formation in keratinocyte tight junctions. {ECO:0000269|PubMed:10508613, ECO:0000269|PubMed:11889141, ECO:0000269|PubMed:23407391}. |
Cell junction;Cell membrane;Disulfide bond;Membrane;Reference proteome;Tight junction;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the claudin family. Claudins are integral membrane proteins and components of tight junction strands. Tight junction strands serve as a physical barrier to prevent solutes and water from passing freely through the paracellular space between epithelial or endothelial cell sheets, and also play critical roles in maintaining cell polarity and signal transductions. The knockout mice lacking this gene die soon after birth as a consequence of dehydration from trandermal water loss, indicating that this gene is indispensable for creating and maintaining the epidermal barrier. The protein encoded by this gene also has gastric tumor suppressive activity, and is a key factor for hepatitis C virus (HCV) entry. [provided by RefSeq, Aug 2010]. |
mmu:12737; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; bicellular tight junction [GO:0005923]; cell junction [GO:0030054]; cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; lateral plasma membrane [GO:0016328]; membrane [GO:0016020]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; tight junction [GO:0070160]; identical protein binding [GO:0042802]; structural molecule activity [GO:0005198]; virus receptor activity [GO:0001618]; aging [GO:0007568]; bicellular tight junction assembly [GO:0070830]; calcium-independent cell-cell adhesion via plasma membrane cell-adhesion molecules [GO:0016338]; cell adhesion [GO:0007155]; cell junction maintenance [GO:0034331]; cell-cell junction organization [GO:0045216]; cellular response to butyrate [GO:1903545]; cellular response to lead ion [GO:0071284]; cellular response to transforming growth factor beta stimulus [GO:0071560]; cellular response to tumor necrosis factor [GO:0071356]; cellular response to type II interferon [GO:0071346]; establishment of blood-nerve barrier [GO:0008065]; establishment of endothelial intestinal barrier [GO:0090557]; establishment of skin barrier [GO:0061436]; hyperosmotic salinity response [GO:0042538]; liver regeneration [GO:0097421]; positive regulation of bicellular tight junction assembly [GO:1903348]; positive regulation of cell migration [GO:0030335]; positive regulation of epithelial cell proliferation involved in wound healing [GO:0060054]; positive regulation of wound healing [GO:0090303]; protein complex oligomerization [GO:0051259]; response to dexamethasone [GO:0071548]; response to ethanol [GO:0045471]; response to interleukin-18 [GO:0070673]; response to lipopolysaccharide [GO:0032496]; response to toxic substance [GO:0009636]; xenobiotic transport across blood-nerve barrier [GO:0061772] |
12651952_dynamic behavior of paired strands of this protein in cell membranes 14698084_Claudin-1 is expressed at the variety of epithelial tissues in inner ear including Organ of Corti, stria vascularis, Reissner's membrane, spiral limbus, vestibular sensory epithelia, dark cell area. 15051488_Thr203 of claudin-1 is required to enhance the barrier function of claudin-1-based tight junctions, probably via its phosphorylation and subsequent integration into tight junctions. 17217615_In the absence of follicle-stimulating hormone receptor signaling, claudin-1 decreases in ovarian surface epithelium and tumors in comparison to wild type. 17508021_Claudin-1 was located in murine Langerhans cells (LC) in epidermis. TGF-beta stimulated accumulation of claudin-1 mRNA and protein when added to cultured bone marrow cells. Claudin-1 may have a function in adhesion and migration of LC. 17828607_Cldn1 is localized in the epidermis and nail and in the dorsal surface of the tongue and glandular compartment of the stomach but is absent from the oral mucosa and the keratinized compartment of the stomach. 18547333_Nectin plays a novel role in the co-localization of JAM and claudin at the same cell-cell adhesion membrane domains. 18648642_Claudin-1 is a p63 target gene with a crucial role in epithelial development 18855015_The induction of tight-junction protein claudin-1 and paracellular permeability during the differentiation of osteoblast-like MC3T3-E1 cells after treatment with IGF-I is regulated via a MAP-kinase pathway, but not with respect to gap junctions. 19234713_This study investigated whether the conserved 'dFynPl' sequence is essential for claudin-1, continuous strand-forming claudin, to form TJ strands 19295125_LNX1p80 is involved in the ubiquitylation, endocytosis and lysosomal degradation of claudins, and the turnover of tight junctions is regulated by ubiquitylation. 19706291_The tight junction protein claudin-1 has gastric tumor suppressive activity and is a direct transcriptional target of RUNX3 19889758_Mouse CLDN1 (mCLDN1) supported HCV genotype 2a infection with only moderate efficiency. indicating CLDN1 also contributes to the restricted species tropism of HCV. CLDN1 also contributes to the restricted species tropism of HCV. 22268650_we identified Cldn1, Cldn2 and Cldn11 as genes that discriminate between diverse types of M2 macrophages 23433550_Cldn1(-/-) mice exhibited the abnormal stratum granulosum formation and stratum corneum barrier defects. 23523916_miR-155 may prevent tumorigenesis in human ovarian cancer through downregulation of CLDN1 23766441_regulates intestinal epithelial homeostasis via regulation of Notch-signalling 24141423_Downregulation of Sirt1 and upregulation of the tight junction protein Claudin-1 by SIRT1-mediated epigenetic regulation in podocytes contributed to albuminuria. 24845399_Occludin and Claudin-1 expressions in the large intestine are under the circadian control, which is associated with temporal regulation of colonic permeability and also susceptibility to colitis. 25277410_MIR29 targets and reduces expression of CLDN1 and NKRF to increase intestinal permeability in inflammatory bowel disease. 25340345_We further demonstrate that KRT76 interacts with CLDN1 and propose that this interaction is necessary to correctly position CLDN1 in tight junctions. 25479235_Cldn-1 is a positive regulator of osteoblast proliferation and differentiation. 26667042_Data show that the spatiotemporal expression of claudin-1 is dysregulated in homeobox (Msx) genes Msx1d/d/Msx2d/d uteri. 27550942_Results suggest that TLR4-dependent claudin-1 internalization and secondary anion secretion contribute to irinotecan-induced diarrhea. 28342862_The data suggested that miR-29a may regulate tumor growth and migration by targeting CLDN1. 28859979_Taken together, these results demonstrate that BTZ-induced claudin 1 expression may be a valuable therapeutic approach for AD. 29675597_Adherens as well as tight junction marker proteins were rapidly and consistently upregulated in both the germinal as well as the functional layer of the oral mucosa. This represents a previously unknown parameter of the epithelial radiation response to clinically relevant fractionation protocols. CONCLUSION: Fractionated irradiation significantly enhanced the expression of all proteins investigated. This study revealed a 29964182_In SAMP10 mice, in addition to preventing a decrease in the naive T cell ratio, aging-associated skin thinning was suppressed histologically and the expression of representative tight junction genes, such as Claudin-1 and Zo-1, was increased. 30504279_claudin-1 is incorporated in blood-brain barrier tight junction complex, impeding blood-brain barrier recovery and causing blood-brain barrier leakiness during poststroke recovery 31264974_eosinophilic esophagitis (EoE) patient biopsy analysis identified a repressed HIF-1alpha/claudin-1 axis, which was restored via pharmacologic HIF-1alpha stabilization ex vivo. Collectively, these studies reveal HIF-1alpha's critical role in maintaining barrier and highlight the HIF-1alpha/claudin-1 axis as a potential therapeutic target for EoE. 31445004_Holocrine Secretion Occurs outside the Tight Junction Barrier in Multicellular Glands: Lessons from Claudin-1-Deficient Mice. 32141492_Inhibition of glycogen synthase kinase 3beta improves cognitive function in aged mice by upregulating claudin presences in cerebral endothelial cells. 35844593_Claudin-1 Mediated Tight Junction Dysfunction as a Contributor to Atopic March. 36542691_A monoclonal antibody targeting nonjunctional claudin-1 inhibits fibrosis in patient-derived models by modulating cell plasticity. 36696800_Identification and characterization of a first-generation inhibitor of claudin-1 in colon cancer progression and metastasis. |
ENSG00000163347 |
CLDN1 |
44.761932 |
0.3825820258 |
-1.386159 |
0.38577290 |
12.835741 |
0.00034006069405457330133771276514664805290522053837776184082031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0017800015435873747880052286873819866741541773080825805664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.0289797401814 |
6.77272562346024 |
65.4212117951004 |
12.4527471884509 |
| ENSMUSG00000022514 |
16180 |
Il1rap |
protein_coding |
Q61730 |
FUNCTION: Coreceptor for IL1RL2 in the IL-36 signaling system. Coreceptor with IL1R1 in the IL-1 signaling system. Associates with IL1R1 bound to IL1B to form the high affinity interleukin-1 receptor complex which mediates interleukin-1-dependent activation of NF-kappa-B and other pathways. Signaling involves the recruitment of adapter molecules such as TOLLIP, MYD88, and IRAK1 or IRAK2 via the respective TIR domains of the receptor/coreceptor subunits. Recruits TOLLIP to the signaling complex. Does not bind to interleukin-1 alone; binding of IL1RN to IL1R1, prevents its association with IL1R1 to form a signaling complex. The cellular response is modulated through a non-signaling association with the membrane IL1R2 decoy receptor. Secreted forms (isoforms 2 and 3) associate with secreted ligand-bound IL1R2 and increase the affinity of secreted IL1R2 for IL1B; this complex formation may be the dominant mechanism for neutralization of IL1B by secreted/soluble receptors. Coreceptor for IL1RL1 in the IL-33 signaling system. Can bidirectionally induce pre- and postsynaptic differentiation of neurons by trans-synaptically binding to PTPRD (PubMed:25908590). May play a role in IL1B-mediated costimulation of IFNG production from T-helper 1 (Th1) cells (By similarity). {ECO:0000250|UniProtKB:Q9NPH3, ECO:0000269|PubMed:11880380, ECO:0000269|PubMed:15986350, ECO:0000269|PubMed:17675517, ECO:0000269|PubMed:18003919, ECO:0000269|PubMed:18450470, ECO:0000269|PubMed:25908590, ECO:0000303|PubMed:21965679}.; FUNCTION: [Isoform 2]: Associates with secreted ligand-bound IL1R2 and increases the affinity of secreted IL1R2 for IL1B; this complex formation may be the dominant mechanism for neutralization of IL1B by secreted/soluble receptors. Enhances the ability of secreted IL1R1 to inhibit IL-33 signaling. {ECO:0000269|PubMed:15986350, ECO:0000269|PubMed:18450470}.; FUNCTION: [Isoform 3]: Required for Src phosphorylation by IL1B. Required for IL1B-potentiated NMDA-induced calcium influx in neurons acting in cooperation with IL1R1 isoform 2 to mediate Akt kinase activation. {ECO:0000269|PubMed:22159118, ECO:0000269|PubMed:22778412}. |
3D-structure;Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Hydrolase;Immunity;Immunoglobulin domain;Inflammatory response;Innate immunity;Membrane;NAD;Receptor;Reference proteome;Repeat;Secreted;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:16180; |
cytoplasm [GO:0005737]; extracellular region [GO:0005576]; glutamatergic synapse [GO:0098978]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; synapse [GO:0045202]; interleukin-1 receptor activity [GO:0004908]; interleukin-1 receptor binding [GO:0005149]; interleukin-33 receptor activity [GO:0002114]; NAD(P)+ nucleosidase activity [GO:0050135]; NAD+ nucleotidase, cyclic ADP-ribose generating [GO:0061809]; cytokine-mediated signaling pathway [GO:0019221]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; interleukin-33-mediated signaling pathway [GO:0038172]; positive regulation of interleukin-13 production [GO:0032736]; positive regulation of interleukin-4 production [GO:0032753]; positive regulation of interleukin-5 production [GO:0032754]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of synapse assembly [GO:0051965]; regulation of postsynaptic density assembly [GO:0099151]; regulation of presynapse assembly [GO:1905606]; synaptic membrane adhesion [GO:0099560]; trans-synaptic signaling by trans-synaptic complex [GO:0099545] |
11847485_IL-1RAcP is essential for physiological activities of peripheral IL-1. 11880380_Identification of essential regions in the cytoplasmic tail of interleukin-1 receptor accessory protein critical for interleukin-1 signaling 13679373_determination of interaction sites in vitro mutagenesis and molecular modeling 17669273_data suggest that domain III of IL-1RAcP may be involved in the formation or stabilization of the IL-1RI/IL-1 complex by binding to epitopes on domain III of the IL-1RI created following IL-1 binding to the IL-1RI 17675517_IL-33 and ST2 form a complex with IL-1R accessory protein (IL-1RAcP), a signaling receptor subunit that is also a member of the IL-1R complex. 18003919_IL-1RAcP is used by more than one alpha-chain of the IL-1 receptor family and thus may resemble a common beta-chain of that family 18450470_these observations establish AcP as co-receptor for IL-33 signaling via ST2 and suggest a novel role for sAcP in modulating the biological activity of IL-33 19481478_an isoform of the IL-1 receptor accessory protein (termed AcPb) was identified that is expressed exclusively in the CNS. 22167021_In experimental biliary atresia, miR-29a/29b1 are upregulated, and reporter assays confirmed that Igf1 and Il1RAP are down-regulated by miR-29. 22357843_The results of this study suggested that IL-1RAcP represents an interesting molecular link between immune systems and synapse formation in the brain. 25449578_We conclude that neuron-specific AcPb plays a critical role in host defenses and sleep homeostasis. 25908590_These results thus reveal the decoding mechanism of splice-insert signaling codes for synaptic differentiation induced by trans-synaptic adhesion between PTPdelta and IL1RAPL1/IL-1RAcP. |
ENSG00000196083 |
IL1RAP |
220.620398 |
2.1724035878 |
1.119292 |
0.17423497 |
40.940121 |
0.00000000015696548859614413821763223328716431795304409035907156066969037055969238281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000024372052215050637164998457333871945129644132066459860652685165405273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
295.670317000061 |
35.7951127414206 |
136.102848780359 |
12.843483337223 |
| ENSMUSG00000022519 |
106393 |
Srl |
protein_coding |
Q7TQ48 |
|
Alternative splicing;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Sarcoplasmic reticulum;Signal |
|
|
mmu:106393; |
intracellular membrane-bounded organelle [GO:0043231]; sarcoplasmic reticulum lumen [GO:0033018]; sarcoplasmic reticulum membrane [GO:0033017]; GTP binding [GO:0005525]; endocytosis [GO:0006897]; endosomal transport [GO:0016197]; response to muscle activity involved in regulation of muscle adaptation [GO:0014873]; store-operated calcium entry [GO:0002115] |
15569689_SAR plays important roles in improving the Ca2+ handling functions of the SR in striated muscle. 15998745_sarcalumenin knockout mice have enhanced resistance to fatigue and altered calcium handling properties 18006473_sarcalumenin-SERCA2a interaction plays an essential role in preserving cardiac function under biomechanical stresses such as pressure overload. 19502553_SAR plays a critical role in maintaining cardiac function under physiological stresses, such as endurance exercise, by regulating Ca(2+) transport activity into the sarcoplasmic reticulum 22119571_The important role of SAR in maintaining Ca(2+) transport activity of SERCA2a and cardiac function in the senescent population, is reported. 23613228_Results indicated that sine oculis homeobox 1 (Six1) overexpression could significantly promote the expression of fast-type muscle genes Atp2a1, Srl, and Mylpf. |
ENSG00000185739 |
SRL |
218.468291 |
0.4776581613 |
-1.065950 |
0.28893451 |
13.433795 |
0.00024713164689307371086127163017920338461408391594886779785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0013391352705355030489747791833110568404663354158401489257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
132.191892173144 |
26.6519549636043 |
276.749991708369 |
41.213874519213 |
| ENSMUSG00000022528 |
15205 |
Hes1 |
protein_coding |
P35428 |
FUNCTION: Transcriptional repressor of genes that require a bHLH protein for their transcription. May act as a negative regulator of myogenesis by inhibiting the functions of MYOD1 and ASH1 (By similarity). Binds DNA on N-box motifs: 5'-CACNAG-3' with high affinity and on E-box motifs: 5'-CANNTG-3' with low affinity. May play a role in a functional FA core complex response to DNA cross-link damage, being required for the stability and nuclear localization of FA core complex proteins, as well as for FANCD2 monoubiquitination in response to DNA damage (By similarity). {ECO:0000250}. |
DNA-binding;Nucleus;Reference proteome;Repressor;Transcription;Transcription regulation |
|
|
mmu:15205; |
chromatin [GO:0000785]; cytoplasm [GO:0005737]; nuclear matrix [GO:0016363]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; chaperone binding [GO:0051087]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity [GO:0001217]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; E-box binding [GO:0070888]; histone deacetylase binding [GO:0042826]; HLH domain binding [GO:0043398]; identical protein binding [GO:0042802]; JUN kinase binding [GO:0008432]; N-box binding [GO:0071820]; protein homodimerization activity [GO:0042803]; protein-containing complex binding [GO:0044877]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription corepressor binding [GO:0001222]; adenohypophysis development [GO:0021984]; amacrine cell differentiation [GO:0035881]; anterior/posterior pattern specification [GO:0009952]; aorta morphogenesis [GO:0035909]; artery morphogenesis [GO:0048844]; ascending aorta morphogenesis [GO:0035910]; BMP signaling pathway [GO:0030509]; Cajal-Retzius cell differentiation [GO:0021870]; cardiac neural crest cell development involved in outflow tract morphogenesis [GO:0061309]; cell adhesion [GO:0007155]; cell fate commitment [GO:0045165]; cell fate determination [GO:0001709]; cell maturation [GO:0048469]; cell migration [GO:0016477]; cell morphogenesis involved in neuron differentiation [GO:0048667]; cell population proliferation [GO:0008283]; cellular response to fatty acid [GO:0071398]; cellular response to interleukin-1 [GO:0071347]; cellular response to nerve growth factor stimulus [GO:1990090]; cellular response to tumor necrosis factor [GO:0071356]; cochlea development [GO:0090102]; comma-shaped body morphogenesis [GO:0072049]; common bile duct development [GO:0061009]; embryonic heart tube morphogenesis [GO:0003143]; enteroendocrine cell differentiation [GO:0035883]; establishment of epithelial cell polarity [GO:0090162]; forebrain radial glial cell differentiation [GO:0021861]; glomerulus vasculature development [GO:0072012]; hair cell differentiation [GO:0035315]; hindbrain morphogenesis [GO:0021575]; in utero embryonic development [GO:0001701]; inhibition of neuroepithelial cell differentiation [GO:0002085]; inner ear auditory receptor cell differentiation [GO:0042491]; inner ear receptor cell stereocilium organization [GO:0060122]; labyrinthine layer blood vessel development [GO:0060716]; lateral inhibition [GO:0046331]; liver development [GO:0001889]; lung development [GO:0030324]; metanephric nephron tubule morphogenesis [GO:0072282]; midbrain development [GO:0030901]; midbrain-hindbrain boundary morphogenesis [GO:0021555]; negative regulation of amacrine cell differentiation [GO:1902870]; negative regulation of calcium ion import [GO:0090281]; negative regulation of cell differentiation [GO:0045596]; negative regulation of cell fate determination [GO:1905934]; negative regulation of DNA-binding transcription factor activity [GO:0043433]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of forebrain neuron differentiation [GO:2000978]; negative regulation of gene expression [GO:0010629]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of inner ear auditory receptor cell differentiation [GO:0045608]; negative regulation of inner ear receptor cell differentiation [GO:2000981]; negative regulation of neurogenesis [GO:0050768]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of neuron projection development [GO:0010977]; negative regulation of oligodendrocyte differentiation [GO:0048715]; negative regulation of pancreatic A cell differentiation [GO:2000227]; negative regulation of pro-B cell differentiation [GO:2000974]; negative regulation of stem cell differentiation [GO:2000737]; negative regulation of stomach neuroendocrine cell differentiation [GO:0061106]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural tube development [GO:0021915]; neuroendocrine cell differentiation [GO:0061101]; neuron differentiation [GO:0030182]; neuronal stem cell population maintenance [GO:0097150]; Notch signaling pathway [GO:0007219]; oculomotor nerve development [GO:0021557]; outflow tract morphogenesis [GO:0003151]; pancreas development [GO:0031016]; pancreatic A cell differentiation [GO:0003310]; pattern specification process [GO:0007389]; pharyngeal arch artery morphogenesis [GO:0061626]; pituitary gland development [GO:0021983]; positive regulation of astrocyte differentiation [GO:0048711]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA binding [GO:0043388]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of glial cell differentiation [GO:0045687]; positive regulation of mitotic cell cycle, embryonic [GO:0045977]; positive regulation of Notch signaling pathway [GO:0045747]; positive regulation of receptor signaling pathway via JAK-STAT [GO:0046427]; positive regulation of T cell proliferation [GO:0042102]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531]; protein-containing complex assembly [GO:0065003]; regulation of epithelial cell proliferation [GO:0050678]; regulation of fat cell differentiation [GO:0045598]; regulation of neurogenesis [GO:0050767]; regulation of neuron differentiation [GO:0045664]; regulation of protein-containing complex assembly [GO:0043254]; regulation of receptor signaling pathway via JAK-STAT [GO:0046425]; regulation of secondary heart field cardioblast proliferation [GO:0003266]; regulation of timing of cell differentiation [GO:0048505]; regulation of timing of neuron differentiation [GO:0060164]; regulation of transcription by RNA polymerase II [GO:0006357]; renal interstitial fibroblast development [GO:0072141]; response to alkaloid [GO:0043279]; response to organic cyclic compound [GO:0014070]; response to thyroid hormone [GO:0097066]; S-shaped body morphogenesis [GO:0072050]; smoothened signaling pathway [GO:0007224]; somatic stem cell population maintenance [GO:0035019]; stomach neuroendocrine cell differentiation [GO:0061102]; T cell proliferation [GO:0042098]; telencephalon development [GO:0021537]; thymus development [GO:0048538]; trochlear nerve development [GO:0021558]; ureteric bud morphogenesis [GO:0060675]; vascular associated smooth muscle cell development [GO:0097084]; ventricular septum development [GO:0003281]; ventricular septum morphogenesis [GO:0060412] |
12032823_Overexpression of the Notch target genes Hes in vivo induces lymphoid and myeloid alterations 12208538_study suggests complex specific relationships between Notch signaling, Math1 and Hes1/Hes5 in the control of hair cell differentiation in the developing organ of Corti 12399594_serum treatment of cultured cells induces cyclic expression of mRNA and Hes1 protein with 2-hour periodicity; cycling depends on negative autoregulation of Hes1 transcription and ubiquitin-proteasome-mediated degradation of Hes1 12478609_HES-1 is a regulator of intracellular signal transduction stimulated by cell adhesion molecules involved in neurite outgrowth. 12560091_Hes1 is expressed in lower small intestinal crypt cells just above the Paneth cells, including Musashi-1-positive cells; Musashi-1 and Hes1 were not expressed in Paneth cells 12972610_Hes1 transcription repression activity is inhibited by Hes6. 14702043_Hes1 determines biliary organogenesis by preventing the pancreatic differentiation program, probably by directly repressing transcription of Neurog3. 15033430_Misexpression of Hes1 in the developing retina promotes formation of undifferentiated precursor-like cells, whereas in Hes1-null retina, precursors are not properly maintained and prematurely differentiate into ganglion cells. 15060169_data suggest that Hes-1 has two roles in adipogenesis: one promotes adipogenesis and the other inhibits adipogenesis 15465493_the differential, spatial regulation of Hes1 and Hes5 gene expression by the Notch signaling pathway in developing nephrons may be a mechanism for patterning cell fate decisions during nephron morphogenesis 15496443_inactivation of Hes1 and Hes5, known Notch effectors, and additional inactivation of Hes3 extensively accelerate cell differentiation and cause a wide range of defects in brain formation 15536134_IkappaBalpha is recruited to the promoter regions of the Notch-target gene, hes1 15563463_Notch1 inhibits the development of erythroid/megakaryocytic cells by suppressing GATA-1 activity through HES1 15670790_Thus, Hes1 appears to have an inhibitory role in Paneth cell differentiation in the small intestine. 15870295_results have suggested that Hes1 directly contributes to the promotion of progenitor cell proliferation through transcriptional repression of a cyclin-dependent kinase inhibitor, p27(Kip1) 16038893_There are multiple requirements for Hes1 during early eye formation. 16287482_The expression profile of c-hairy-1 in the mouse stomach is wider than that of Msi-1 and indicates that c-hairy-1 expression is not always regulated by Msi-1. 16365048_Notch protein binding to Hes5-GFP is extinguished fast and recovered slowly, whereas Hes1-GFP is inhibited late and recovered quickly 16508307_These results indicate that Hes1 and Hes5 play an important role in the formation of the cranial and spinal nerve systems. 16543950_Treg-mediated immunosuppression involves activation of the Notch-HES1 axis by membrane-bound TGF-beta 16651378_Data demonstrated that targeted Hes1 overexpression is sufficient to protect melanoblasts from the elimination by apoptosis. 16710472_Data demonstrate that the Hes1-mediated Notch pathway is required for region-appropriate specification of pancreas in the developing foregut endoderm through regulation of Ptf1a expression. 16728479_High levels of Hes1 expression constitutively repress proneural bHLH gene expression and reduce cell proliferation 16899237_Hes1 and p57 coordinate cell cycle exit and self-renewal of pancreatic progenitors during an early stage in organogenesis to regulate the number of pancreatic progenitors.They also eliminate cells with aberrant cell cycle characteristics. 17150554_Hairy/enhancer of split protein (Hes1), a bHLH transcriptional repressor functioning in neuronal differentiation, is a target of mouse miRNA-23b (miR-23b). 17367776_Hes1 repressor is essential for melanotrope specification. 17426285_Mice deficient for Hes1 and Hes5 display severe pituitary hypoplasia caused by accelerated differentiation of progenitor cells. 17592117_Hes1 oscillations are important for efficient cell proliferation and provide evidence that expression modes of signaling molecules affect downstream cellular events. 17681138_These results indicate that Id proteins participate in NSC maintenance through sustaining Hes1 expression in early embryos. 18287091_Ikaros directly represses Hes1 in concert with the transcriptional repressor, RBP-Jkappa, allowing for cross-talk between Notch and Ikaros that impacts regulation of CD4 expression 18292208_Hes1 regulates corneal development and the homeostatic function of corneal epithelial stem/progenitor cells. 18302773_Hey2 functions in parallel with Hes1 and Hes5 in patterning the organ of Corti, and interacts genetically with Hes1 for early embryonic development and survival 18308300_Pax3 regulates Hairy and enhancer of split homolog-1 (Hes1) and Neurogenin2 (Ngn2) by directly binding to their promoters. 18400163_Hes1 oscillation regulates Ngn2 and Dll1 oscillations, which in turn leads to maintenance of neural progenitors by mutual activation of Notch signaling. 18410734_Data show that Notch signaling determines spinous cell fate and induces terminal differentiation by a mechanism independent of Hes1, but Hes1 is required for maintenance of the immature state of spinous cells. 18430630_oscillatory expression stimulated by FGF 18499474_Hes1 regulates 5-HT1A receptor transcription and is required for its correct developmental expression. 18762022_Liver-specific disruption of glucocorticoid receptor (GR) action improves the steatotic phenotype in fatty liver mouse models and leads to the induction of transcriptional repressor hairy enhancer of split 1 (Hes1) gene expression. 18777163_Results suggest that Notch signaling can modulate the FOXP3 promoter through RBP-J- and Hes1-dependent mechanisms. 18840712_GATA-2 regulates myeloid progenitor function through HES-1. 18950867_expression of the Notch signaling mediator RBP-J and the Notch target genes Hes-1 and Hes-5 was markedly reduced in MZ but not FO B cells of Sly1(d/d) mice. 18996108_These data reveal important overlapping functions of Hes1 and Prop1 in cell differentiation and movement that are critical for pituitary organogenesis. 19124651_Shh stimulation of progenitor proliferation and cell diversification requires Gli2 and Hes1 activity. 19214430_Notch1/NICD levels are significantly regulated by GSK-3beta and by GSK-3alpha. 19343206_Hes1 plays a role in orchestrating taste cell differentiation in developing taste buds. 19466597_required for neural development, but persistent Hes1 expression inhibits proliferation and differentiation of neuronal progenitors 19541765_observations taken together indicate a role for HES1 in the control of cell cycle exit and in mediating the balance between proliferation and differentiation, allowing for the properly timed emergence of hormone secreting cell types. 19546852_We also detected increases in Hes1 and activated Notch1 expression in our mouse model of BAVM induced by constitutively active Notch4, demonstrating molecular similarity between the mouse model and the human disease. 19670267_The Notch-responsive Hes-1 protein is capable of repressing osteocalcin gene transcription in osteoblastic cells through an E-box in the proximal promoter. 19684110_expression of Hes1 and its downstream genes oscillate in mouse embryonic stem cells, Those expressing low and high levels of Hes1 tended to differentiate into neural and mesodermal cells 19688092_demonstrated for the first time that Hes1 potentiates T cell lymphomagenesis, by up-regulating a subset of Notch target genes and by causing an accumulation of ISP thymocytes particularly vulnerable to oncogenic transformation 20067572_Here, we have clearly demonstrated the existence of a population of neural progenitors with Notch/CBF1-independent Hes-1 expression in vitro. 20110912_Expression of the Msi1 and Hes1 small intestinal stem cell markers in the small intestine was not homogenous and the markers were more highly expressed in jejunal tissues. 20122914_Hes1 expression is reduced in Tbx1 null cells and is required for the development of structures affected in 22q11 deletion syndrome. 20134146_Lack of altered phenotype in the conditional HES1;HES5 double mutant mice can be explained by the HES1 and HES5 genes not being involved in cartilage and endochondral bone development or by redundancy between the genes of the family of HES genes. 20335360_Hes1 was identified as an RBPjkappa-dependent Notch target gene important for in mesenchymal progenitor cell maintenance and the suppression of in vitro chondrogenesis. 20545770_The results indicate that sustained Hes1 expression delays the differentiation of ES cells and promotes the preference for the mesodermal rather than the neural fate by suppression of Notch signaling. 20652960_FRS2alpha fine-tunes the FGF2-signaling to control self-renewal and proliferation of neural stem cells through Hes1. 20833714_Methylation of Hes1 and Neurog2 promoter is associated with caudal neural tube development. 21084301_Hes1 inactivation in osteoblasts increased trabecular bone volume, number, and connectivity due to increased mineral apposition rate and suppressed bone resorption. 21089298_It regulates neural development.(review) 21093323_Hes1 is needed for T cell commitment, but dispensable for Notch-dependent thymocyte maturation thru and beyond the beta selection checkpoint. Hes1 is also essential for the development and maintenance of Notch-induced T cell acute lymphoblastic leukemia. 21167918_indicate that Mek-Erk pathway plays pivotal role in preventing early oligodendrocyte progenitor cells (OLPs) maturation to late OLPs and the effect is mediated by cell cycle progression as well as Hes1 induction 21205788_Data find that Hes1 expression in the early embryonic pancreas identifies multipotent, Notch-responsive progenitors, differentiation of which is blocked by activated Notch. 21221806_Musash1 and Hes1 high expression cells locate predominantly in impaired small-intestinal epithelium after transplantation and contribute to epithelial regeneration. 21300049_Hes1, Hes5 and Hey1 cooperatively inhibit hair cell formation, and one allele of Hes1, Hes5 or Hey1 is sufficient for supporting cell production probably by lateral inhibition in the sensory epithelium. 21354131_Hes1 null mice show continued misplacement of arginine vasopressin cell bodies in the paraventricular nucleus and supraoptic nucleus and additionally exhibit abnormal axonal projection 21364918_A dual role of Hes1 during thyroid development: first, control of the number of both thyrocyte and C-cell progenitors, via a p57-independent mechanism; second, adequate differentiation and endocrine function of thyrocytes and C-cells. 21389783_Hes1 expression and CYLD repression are essential events downstream of Notch1 in T-cell leukemia. 21414292_these results provide strong evidence that caveolin-1 can promote astroglial differentiation of neural stem/progenitor cells through modulating Notch1/NICD and Hes1 expressions. 21481105_These results suggest that Hes1 plays essential roles in contact inhibition of cell proliferation in 3T3-L1 cells by repressing E2F-1 expression. 21606479_Comparative gene-expression analysis identified Hes1, one of the basic helix-loop-helix proteins involved in Notch signaling, as a factor specifically expressed in IL-17-producing gammadelta T cells 21744064_The co-repressor recruitment mediated through WRPW domain of Hes-1 has contributed to the repressive effect. 21745596_this recapitulates some previous observations on Hes1 expression and suggests new, hitherto unrecognised expression domains including expression in the definitive endoderm at early somite stages before gut tube closure and thus preceding organogenesis. 21798254_Hes1 regulates the number and anterior-posterior patterning of mesencephalic dopaminergic neurons at the mid/hindbrain boundary (isthmus). 22037762_Notch signaling, which is known to play an important role for germ cell development in Drosophila and Caenorhabditis elegans, is cyclically activated in Sertoli cells and regulates stage-dependent gene expression of Hes1. 22056382_Tnf-alpha stimulation resulted in phosphorylation of histone H3 at the Hes1 promoter. This signal was lost with Ikk2 deletion. Hes1 suppresses expression of Pparg. 22096075_Hes1 is required to sustain Ptf1a expression, and in turn Dll1 expression in early multipotent pancreatic progenitor cells. 22167192_seven conserved DNA sequence blocks, representing the hes1 promoter and six novel cis-regulatory modules 22185794_Hes1, Atoh1, and Gfi1b form a fate-determining Notch signaling driven ternary switch in the developing intestinal epithelium. 22232434_the expression levels of Hes1 transcripts were also significantly decreased in denervated eyes. 22318232_in Apc mutation-induced intestinal tumors, inactivation of Hes1 was sufficient for reducing tumor cell proliferation and inducing differentiation of tumor cells; results indicate Hes genes cooperatively regulate intestinal development and homeostasis 22649105_These results point to HES1, via repression of PTEN, and c-Myc as critical mediators of Notch function at the beta-selection checkpoint. 22689348_The results suggest that Hes1 plays a critical role in regulating the development of neural crest derivatives in the mouse cervical region. 22778136_results describe a mechanism whereby GATA-1 utilizes Ikaros and Polycomb repressive complex 2 to promote Hes1 repression as an important step in erythroid cell differentiation 22840391_MicroRNA-9 Modulates Hes1 ultradian oscillations by forming a double-negative feedback loop. 22982865_Curcumin reduced arginase activity and up regulated TGF-beta1 and HES-1 transcripts in mice. 23134481_Hes1 expression dynamics in developing brain is regulated by MicroRNA9. 23199335_Data indicate that Notch1 and Notch2 are expressed and localized in the nuclei of the label-retaining cells (LRCs), and the expression of Notch-inducible genes, Hes1 and Hey2, was elevated in LRCs. 23207146_Hes-1 downregulation allows PDX-1 to act as a major determinant of cholangiocyte proliferation in response to cholestatic injury. 23255132_Differential expression of Hes1 between hematopoietic stem cells and hematopoietic progenitor cells resulted in the distinct responses of these cells to the leukemic condition. 23267012_Hes1 repressor is essential regulator of hematopoietic stem cell development downstream of Notch signaling. 23274689_Notch-Hes1 pathway regulates ovarian somatic cell development, which is necessary for oocyte survival and maturation. 23277114_In the postnatal retina, NOTCH2, as well as the Notch downstream genes, HES1 and SOX9, were detected in VSX2/Cyclin D1/SOX2-expressing cells in the postnatal retina, and in the mature retina NOTCH2 was most abundant in Muller glia 23325756_Spatial stochastic modelling of the Hes1 gene regulatory network: intrinsic noise can explain heterogeneity in embryonic stem cell differentiation 23489689_The results indicate that Jagged-1-Hes-1 signaling can suppress the skewing of CD4+ T cells toward Th17 cells via RORgammat, for which Hes-1 may be crucial. 23499991_Sustained vs. oscillating expressions of Ngn2, Dll1 and Hes1: a model of neural differentiation of embryonic telencephalon. 23509692_miR-1 promotes the differentiation of MSCs into cardiac lineage in part due to negative regulation of Hes-1 23686616_Our data indicate that the development of pharyngeal organs and survival of neural-crest-derived mesenchyme in pharyngeal arches are critically dependent on Hes1. 23850665_delivery of siRNA to Hes1 and Hes5 using a transfection reagent or siRNA to Hes1 encapsulated within nanoparticles increased hair cell numbers in non-toxin treated organotypic cultures of cochleae and maculae 23860410_Notch signaling can directly downregulate MUC5AC promoter activity through Hes1-dependent mechanisms. 23871867_Hes1 and Hes5 regulate vascular remodeling and arterial fate specification of endothelial cells in the development of the brain; they are critical transducers of Notch signals in brain vascular development. 24015274_The transcription and expression patterns of Notch pathway components (Notch 1-3, Delta1 and 4, Jagged1) and effectors (Hes1, Hes2, Hes5 and Nrarp) were evaluated in the mouse testis 24185616_Data indicate that T cell lymphopoiesis requires C/EBP-alpha by the Notch target Hes1. 24300895_results indicate that HES1 acts as a master repressor, the silencing of which is required for proper glucocorticoid signaling. 24486648_Hes1 upregulation contributes to the development of FIP1L1-PDGRA-positive leukemia in blast crisis. 25005473_A muscle-specific regulatory element of p57(kip2) directly activated by muscle regulatory factors in myoblasts but repressed by the Notch targets Hes1/Hey1 in progenitor cells, is identified. 25084035_Results suggest that Hes1 represents a negative regulator of adult neurogenesis post-traumatic brain injury (TBI) and that the precise space-time regulation of Hes1 expression in the dentate gyrus may promote the recovery of neural function following TBI 25234168_Hes1 suppresses acute myeloid leukemia development through FLT3 repression. 25687338_Hes1-mediated Notch activity determines the plasticity of adult pancreatic duct cells and that there may exist a dosage requirement of Sox9 for keeping the duct cell identity in the adult pancreas. 25701255_Findings suggest the strict spatio-temporal expression of Notch effector genes plays an important role during hippocampal neurogenesis and suggests that Notch1, Hes1 and Ngn2 were regulated by changing some specific CpG sites of their promoters 25846153_found that Hes1 was deubiquitinated and stabilized by Usp27x and its homologs ubiquitin-specific protease 22 (Usp22) and ubiquitin-specific protease 51 26348989_This study demonstrated that Hes1 and Hes5 modulate not only maintenance of progenitor cells but also pituicyte versus neuron fate specification during neurohypophysis development. 26453796_Insm1-dependent Hes1 repression is required for neuroendocrine development. 26655730_We found that miR-21 acts as a bidirectional switch in the formation of IPCs by regulating the expression of target and downstream genes (SOX6, RPBJ and HES1). 26804914_the glucose-regulated antagonism between CREB and Sirt-1 for Hes-1 transcription participates in the metabolic regulation of neurogenesis. 26932672_Hes/Hey signaling at the Atoh1 promoter has a role in selection of cell fate in the organ of Corti 27160681_the RBPjkappa-dependent Notch targets HES1 and HES5 suppress chondrogenesis and promote the onset of chondrocyte hypertrophy. 27322654_this study identifies Hes1 as a homeostatic suppressor of inflammatory responses that exerts its suppressive function by regulating transcription elongation 27405330_Hes-1 expression is maintained in neural progenitor territory throughout mouse neocortical development, a simple shift from Notch-independent to -dependent state makes it pleiotropic as the former maintains the neural stem cells in a non-dividing/slow-dividing state, whereas the latter is very much required for maintenance and proliferation of radial glial cells. 27537524_Notch1-Hes-1 signaling controls TLR7-induced autophagic death of macrophage via regulation of P62 in mice with lupus. 27639167_Hes1 plays a key role in acinar cell integrity and plasticity on cellular insults. 28286920_this paper shows the critical role of the Notch1-Hes1 signaling cascade in the regulation of innate immunity in acetaminophen-triggered liver inflammation 28536281_Hes1 disruption leads to tumor regression without perturbing normal stem cell homeostasis, preclinically validating Hes1 as a cancer therapeutic target. 28602625_that HIF-1alpha but not HIF-2alpha is able to interact with either GA-binding protein alpha or GA-binding protein beta1 28655839_there is a link between Dmrta2 modulation of Hes1 expression and the maintenance of neural progenitor cells during cortical development 28675662_Although Hes5-GFP and Hes1 are coexpressed in particular developmental contexts, we also noted cohorts of lens or retinal cells expressing just one factor. The dynamic Hes5-GFP expression pattern, coupled with its derepressed expression in Hes1 mutants, suggests that this transgene contains the relevant cis-regulatory elements that regulate endogenous Hes5 in the mouse lens and retina. 28974640_Notch signaling and Id2/3 regulate neurogenesis in a complementary manner and ID factors can induce neural stem cell maintenance and quiescence in the absence of Notch. 29070679_results suggest that SCF(FBXL14) promotes neuronal differentiation by targeting HES1 for ubiquitin-dependent proteolysis and that the C-terminal WRPW motif in HES1 is required for this process 29171783_These results suggest that hyperhomocysteinemia-enhanced brain damage is associated with increased autophagy and neuronal apoptosis in Apo E(-/-) mice, in which downregulation of hes1 and hes5 is involved. 29297498_this paper shows that epithelial Hes1 maintains gut homeostasis by preventing microbial dysbiosis 29385539_Study shows that Foxg1 Overexpression in Neocortical Pyramids Stimulates Dendrite Elongation Via Hes1 and pCreb1 Upregulation. 29389974_Olig2 represses expression of the Notch signaling pathway effectors Hes1 and Hes5 29491143_Depletion of HES1 increased cell death in response to endoplasmic reticulum stress in mouse and human cells, in a manner that depended on the pro-apoptotic gene growth arrest and DNA damage-inducible protein GADD34. 29496446_inhibition of PTEN by Notch1/Hes1 in response to high glucose concentration inhibits autophagy, which is associated with the progression of fibrosis in diabetic nephropathy 29869113_The authors found that Hes1 specifically recruited Tle4 (transducin-like enhancer protein 4) to repress expression of IL-6 and IL-12. 29902458_miR-374b regulates the proliferation and differentiation of neural stem cells through targeting Hes1. Overexpression of Hes1 reverses the effect of miR-374b overexpression on neural stem cells differentiation. 30090006_Hes1 might contribute to the maturation and the cellular structure organization of biliary epithelial cells, which provides new insight into understanding the pathology of biliary atresia. 30093553_the results raise the possibility that HES1 or other genes that affect HES1 expression could be involved in the etiology of Ectopic pancreas . 30551562_Findings indicate that nuclear translocation of MSX-1, alone or in combination with HES-1, produce chondrocyte-like cells, and simultaneous activation of HES-1 and MSX-1 increases the generation of smooth muscle and neuronal cells. 30670317_Study data suggest that Hes1 is downstream of alpha7nAChR and is important to fully activate the cholinergic anti-inflammatory pathway in peritoneal macrophages in kidney ischemia/reperfusion injury mouse model. 30705405_Mutant KRAS-induced Hes1 plays an essential role in PDAC initiation. 30745427_HeyL and Hey1 function redundantly in muscle stem cells, and HeyL requires Hes1 for effective DNA binding and biological activity 30862660_Ablation of the Hes1 oscillator in stem cells interfered with stable MyoD oscillations and led to prolonged periods of sustained MyoD expression, resulting in increased differentiation propensity 30862661_Ascl1 oscillations, which normally depend on Hes1 oscillations, regulate the active state, while high Hes1 expression and resultant Ascl1 suppression promote quiescence in neural stem cells. 30951556_HES1 is not an effector of Notch signaling during CD8+ T cell response. 31015298_Genome-wide assessment of Hes1 occupancy revealed that suppression of WDFY1 was secondary to direct binding and thus enhancement of expression of VEGF-C by Hes1 31053533_The authors show that the antimicrobial peptide alpha-defensin is functionally sustained during nutrient deprivation because of regulation of the defensin-processing enzyme MMP7 by microbiota- and host-derived factors. Sustained Mmp7 expression requires the microbiota and is mediated by de-repression of the transcription activator Atoh1 upon attenuation of the transcriptional repressor Hes1 in intestinal epithelial cells. 31160641_Hes1 expression in mature neurons in the adult mouse brain is required for normal behaviors. 31178417_we conclude that TLX, by repressing Hes1 expression, counteracts quiescence and NOTCH activation in NSCs, thereby relieving NOTCH-mediated lateral inhibition of proliferation in basal progenitors. 31949107_Simultaneous Requirements for Hes1 in Retinal Neurogenesis and Optic Cup-Stalk Boundary Maintenance. 32094111_robust Hes1 oscillations are required for maintenance and proliferation of NPCs and the normal timing of neurogenesis, thereby regulating brain morphogenesis 32454795_Notch-Hes1 Signaling Regulates IL-17A(+) gammadelta (+)T Cell Expression and IL-17A Secretion of Mouse Psoriasis-Like Skin Inflammation. 32800756_Foxi1 inactivation rescues loss of principal cell fate selection in Hes1-deficient kidneys but does not ensure maintenance of principal cell gene expression. 32957483_Visualizing Cell Cycle Phase Organization and Control During Neural Lineage Elaboration. 33067264_Hes1 Is Essential in Proliferating Ductal Cell-Mediated Development of Intrahepatic Cholangiocarcinoma. 33222180_FANCD2 and HES1 suppress inflammation-induced PPAR to prevent haematopoietic stem cell exhaustion. 33264418_Bone morphogenetic protein 9 (BMP9) directly induces Notch effector molecule Hes1 through the SMAD signaling pathway in osteoblasts. 33314792_TNF-alpha inhibits GDNF levels in Sertoli cells, through a NF-kappaB-dependent, HES1-dependent mechanism. 33371813_Ginsenoside Rg3 Suppresses Epithelial-Mesenchymal Transition via Downregulating Notch-Hes1 Signaling in Colon Cancer Cells. 33622250_The NOTCH-HES-1 axis is involved in promoting Th22 cell differentiation. 33675998_Hes1 oscillation frequency correlates with activation of neural stem cells. 34742737_Hairy and enhancer of split 1 is a primary effector of NOTCH2 signaling and induces osteoclast differentiation and function. 34928956_HES1 is a novel downstream modifier of the SHH-GLI3 Axis in the development of preaxial polydactyly. 35732696_Medial prefrontal cortex Notch1 signalling mediates methamphetamine-induced psychosis via Hes1-dependent suppression of GABAB1 receptor expression. 36075324_Context-Dependent Roles of Hes1 in the Adult Pancreas and Pancreatic Tumor Formation. |
ENSG00000114315 |
HES1 |
208.013168 |
0.2914170238 |
-1.778843 |
0.26023588 |
45.154977 |
0.00000000001820424468121842179832990017769220846000077784765380783937871456146240234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000003147412655894653869451104329102181927924064552826166618615388870239257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
92.7259645091746 |
16.8169254075992 |
318.189937210849 |
41.6083862000118 |
| ENSMUSG00000022548 |
11815 |
Apod |
protein_coding |
P51910 |
FUNCTION: APOD occurs in the macromolecular complex with lecithin-transport and binding of bilin. Appears to be able to transport a variety of ligands in a number of different contexts. |
Disulfide bond;Glycoprotein;Lipid-binding;Pyrrolidone carboxylic acid;Reference proteome;Secreted;Signal;Transport |
|
The protein encoded by this gene is a component of high-density lipoprotein (HDL), but is unique in that it shares greater structural similarity to lipocalin than to other members of the apolipoprotein family, and has a wider tissue expression pattern. The encoded protein is involved in lipid metabolism, and ablation of this gene results in defects in triglyceride metabolism. Elevated levels of this gene product have been observed in multiple tissues of Niemann-Pick disease mouse models, as well as in some tumors. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Aug 2014]. |
mmu:11815; |
cytoplasm [GO:0005737]; cytosolic ribosome [GO:0022626]; dendrite [GO:0030425]; endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; neuronal cell body [GO:0043025]; perinuclear region of cytoplasm [GO:0048471]; cholesterol binding [GO:0015485]; aging [GO:0007568]; brain development [GO:0007420]; glucose metabolic process [GO:0006006]; lipid metabolic process [GO:0006629]; lipid transport [GO:0006869]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; negative regulation of focal adhesion assembly [GO:0051895]; negative regulation of lipoprotein lipid oxidation [GO:0060588]; negative regulation of monocyte chemotactic protein-1 production [GO:0071638]; negative regulation of platelet-derived growth factor receptor signaling pathway [GO:0010642]; negative regulation of protein import into nucleus [GO:0042308]; negative regulation of smooth muscle cell proliferation [GO:0048662]; negative regulation of smooth muscle cell-matrix adhesion [GO:2000098]; negative regulation of T cell migration [GO:2000405]; peripheral nervous system axon regeneration [GO:0014012]; response to axon injury [GO:0048678]; response to reactive oxygen species [GO:0000302]; tissue regeneration [GO:0042246] |
11744388_Expression pattern of the lipocalin apolipoprotein D during mouse embryogenesis. 12815234_Apolipoprotein D-immunolabeled cells are observed in select grey matter nuclei, including the globus pallidus, thalamus, and substantia nigra, and in white matter tracts within the internal capsule and cerebellum of Neimann-Pick type C mouse brain. 14703507_apolipoprotein D and the lipogenic protein Spot 14 are LXR alpha and LXRB responsive genes both in vitro and in vivo 15668042_ApoD messenger RNA expression was localised to the spiral ligament and spiral limbus, particularly in the suprastrial and supralimbral regions. 18419796_These observations, together with its transcriptional up-regulation in the brain upon oxidative insult, identify ApoD as an acute response protein with a protective and therefore beneficial function mediated by the control of peroxidated lipids. 19465111_Taken together, our results describe receptor and region specific changes in SST and SSTR subtypes expression in ApoD(-/-) mice brain, which may be linked to specific neurological disorders. 19963028_This study concluded that glutamatergic pathways seem to be particularly affected in ApoD(-/-) mice. 20124557_Apolipoprotein D plays a significant role in lipid metabolism. 20607718_mRNA and protein expression profiles reveal that ApoD is functionally connected in an age-dependent manner to specific molecular programs triggered by injury. 20920539_Scanning cytometry revealed PACAP/ApoD induced low density lipoprotein receptors (LDLR) and ApoE receptor 2 (apoER2) in NF200-positive cells. 21632073_The findings underscore the importance of ApoD in the regulation of plasma insulin levels and triglyceride metabolism, suggesting that ApoD plays an important role in the pathogenesis of dyslipidemia. 21688324_The data of this study supported that ApoD contributes to the endurance of astrocytes and decreases their reactivity level in vitro and in vivo 22398376_Abeta25-35 induces neuroprotective ApoD expression in hippocampal cells in response to stress-induced growth arrest. 22581439_Changes in hypothalamus of ApoD(-/-) mice may indicate potential role of ApoD in regulation of endocrine functions of somatostatin in a receptor-dependent manner. 24082102_apoD may be part of an intrinsic cardioprotective system, possibly as a consequence of its antioxidation activity. 25784209_It regulates amyloid plaque pathology in a mouse model of Alzheimer's disease 26829325_apoD protein levels are variable across different brain regions. 29222871_Study shows that in myelin maturation ApoD ultimately controls the removal of the sialic-rich hydrophilic glycocalyx, by maintaining functional integrity of lysosomes. A detailed analysis of the mechanism reveals that the proper localization of Neu1 and plasma membrane Neu3, as well as of the membrane-bound Fyn kinase, depend on ApoD. 29885491_These data indicate apolipoprotein-D rarely exists as a free monomer under physiological conditions and provide insights into novel native structures of apolipoprotein-D and into oligomerisation behaviour in the lipocalin family. 32440710_Studies of ApoD(-/-) and ApoD(-/-)ApoE(-/-) mice uncover the APOD significance for retinal metabolism, function, and status of chorioretinal blood vessels. 32798728_Apolipoprotein D-mediated preservation of lysosomal function promotes cell survival and delays motor impairment in Niemann-Pick type A disease. |
ENSG00000189058 |
APOD |
4126.025734 |
0.1485799799 |
-2.750688 |
0.07677999 |
1172.396801 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000006085586 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001853913 |
Yes |
No |
1091.68498824815 |
58.1267259370366 |
7347.45682978406 |
279.835357875403 |
| ENSMUSG00000022587 |
17069 |
Ly6e |
protein_coding |
Q64253 |
FUNCTION: GPI-anchored cell surface protein that regulates T-lymphocytes proliferation, differentiation, and activation (PubMed:9575182, PubMed:8642345). Regulates the T-cell receptor (TCR) signaling by interacting with component CD3Z/CD247 at the plasma membrane, leading to CD3Z/CD247 phosphorylation modulation (PubMed:9575182). Restricts the entry of murine coronavirus, mouse hepatitis virus, by interfering with spike protein-mediated membrane fusion (PubMed:32704094). Also plays an essential role in placenta formation by acting as the main receptor for syncytin-A (SynA) (PubMed:28679758). Therefore, participates in the normal fusion of syncytiotrophoblast layer I (SynT-I) and in the proper morphogenesis of both fetal and maternal vasculatures within the placenta (PubMed:29500366). May also act as a modulator of nicotinic acetylcholine receptors (nAChRs) activity. In vitro inhibits alpha-3:beta-4-containing nAChRs maximum response (PubMed:26276394). {ECO:0000269|PubMed:26276394, ECO:0000269|PubMed:28679758, ECO:0000269|PubMed:29500366, ECO:0000269|PubMed:32704094, ECO:0000269|PubMed:8642345, ECO:0000269|PubMed:9575182}. |
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:17069; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; synapse [GO:0045202]; acetylcholine receptor binding [GO:0033130]; acetylcholine receptor inhibitor activity [GO:0030550]; acetylcholine receptor signaling pathway [GO:0095500]; adrenal gland development [GO:0030325]; epinephrine secretion [GO:0048242]; in utero embryonic development [GO:0001701]; negative regulation of viral entry into host cell [GO:0046597]; norepinephrine metabolic process [GO:0042415]; organ growth [GO:0035265]; ventricular cardiac muscle tissue morphogenesis [GO:0055010] |
15170814_Sca-2 -- a signal transducer situated at the nexus of surface molecules regulating death receptor-mediated apoptosis in hepatoma 28679758_Knocking down Ly6e greatly reduced SynA-induced cell fusion, thus suggesting that Ly6e is the sole receptor for SynA in vivo. 29500366_demonstrate that LY6E is essential for the normal fusion of syncytiotrophoblast layer I, and for the proper morphogenesis of both fetal and maternal vasculatures within the placenta 32704094_LY6E impairs coronavirus fusion and confers immune control of viral disease. 32708319_Computational Analysis of the Global Effects of Ly6E in the Immune Response to Coronavirus Infection Using Gene Networks. |
ENSG00000160932 |
LY6E |
1821.485128 |
2.3741530805 |
1.247413 |
0.07473870 |
274.521801 |
0.00000000000000000000000000000000000000000000000000000000000011733622613046149663436060079584983570641636367390418071808656812760020051372086161142112512060359939282969536259653667988193872301777321557946087800716663401834338742446561809629201889038085937500000000 |
0.0000000000000000000000000000000000000000000000000000000000283692920066538006246747685345422357780701803299893384682306561963575403875017947834060955793918664528075347404979037575469513789127894569941311353998558697497855973779223859310150146484375000000000000 |
Yes |
No |
2561.58875458944 |
132.762260781223 |
1078.94843664852 |
43.0606580317475 |
| ENSMUSG00000022596 |
57277 |
Slurp1 |
protein_coding |
Q9Z0K7 |
FUNCTION: Has an antitumor activity. Was found to be a marker of late differentiation of the skin. Implicated in maintaining the physiological and structural integrity of the keratinocyte layers of the skin. In vitro down-regulates keratinocyte proliferation; the function may involve the proposed role as modulator of nicotinic acetylcholine receptors (nAChRs) activity. In vitro inhibits alpha-7-dependent nAChR currents in an allosteric manner (By similarity). In T cells may be involved in regulation of intracellular Ca(2+) signaling (PubMed:17286989). Seems to have a immunomodulatory function in the cornea. The function may implicate a possible role as a scavenger receptor for PLAU thereby blocking PLAU-dependent functions of PLAUR such as in cell migration and proliferation (PubMed:23139280, PubMed:25168896). {ECO:0000250|UniProtKB:P55000, ECO:0000269|PubMed:17286989, ECO:0000269|PubMed:23139280, ECO:0000269|PubMed:25168896}. |
Cytokine;Disulfide bond;Reference proteome;Secreted;Signal |
|
|
mmu:57277; |
extracellular space [GO:0005615]; acetylcholine receptor activator activity [GO:0030549]; cytokine activity [GO:0005125]; locomotory behavior [GO:0007626]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of keratinocyte proliferation [GO:0010839]; neuromuscular process controlling posture [GO:0050884]; urokinase plasminogen activator signaling pathway [GO:0038195] |
20621062_Those findings suggest that diminished expression of SLURP-1 in asthma attenuates its negative regulation of airway inflammation, and that perhaps changes in SLURP-1 expression could serve as a marker of airway damage in asthma. 24499735_Slurp1 in mice has a role in neuromuscular and metabolic phenotypes in addition to its role in palmoplantar keratoderma 26474319_SLURP1 participates in pathophysiology of psoriasis by regulating keratinocyte proliferation and differentiation, and by suppressing the growth of S. aureus 26670825_These data describe the influence of age, sex, genetic background, and ocular surface health on mouse corneal expression of Slurp1, establish the baseline for human tear SLURP1 expression. 26967477_SLURP1 nonsense mutation is responsible for the pathogenesis of palmoplantar keratoderma. 28418591_The hypomorphic Egfr allele, which leads to reduced EGFR signalling in keratinocytes, did not ameliorate the PPK elicited by SLURP1 deficiency, suggesting that SLURP1 deficiency causes PPK independently (or downstream) from the EGFR pathway. 29017797_Both Slurp1-/- and Slurp1-/-; Slurp2-/- mice, which were created and maintained on an inbred background, developed hind-limb clasping. These studies indicate that this phenotype, typically found in the setting of neuropathy and other forms of neurological disease, is a bona fide phenotype of Slurp1 deficiency. 33815383_New Pathways for the Skin's Stress Response: The Cholinergic Neuropeptide SLURP-1 Can Activate Mast Cells and Alter Cytokine Production in Mice. 34721415_Endogenous alpha7 nAChR Agonist SLURP1 Facilitates Escherichia coli K1 Crossing the Blood-Brain Barrier. 34923162_Secreted Ly-6/uPAR-related protein-1 (SLURP1) is a pro-differentiation factor that stalls G1-S transition during corneal epithelial cell cycle progression. |
ENSG00000126233 |
SLURP1 |
18.430463 |
0.2375893025 |
-2.073458 |
0.65371846 |
9.702420 |
0.00184025513920794926356339082929025607882067561149597167968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0079250116710250177037844210303774161729961633682250976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.16645496197466 |
2.89076617578944 |
25.9542618128799 |
8.30922877008674 |
| ENSMUSG00000022598 |
72373 |
Psca |
protein_coding |
P57096 |
FUNCTION: May be involved in the regulation of cell proliferation. {ECO:0000250}.; FUNCTION: May act as a modulator of nicotinic acetylcholine receptors (nAChRs) activity. In vitro inhibits nicotine-induced signaling probably implicating alpha-3:beta-2- or alpha-7-containing nAChRs. {ECO:0000250|UniProtKB:O43653}. |
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:72373; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; acetylcholine receptor binding [GO:0033130]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; regulation of neurotransmitter receptor activity [GO:0099601] |
11752398_This transgenic system helps define the range of cellular changes associated with altered expression of PSCA, shows that transcriptional control is a major component regulating PSCA levels. 15860580_Sca-1-enriched prostate-regenerating cells possess multiple stem/progenitor cell properties 18044730_PSCA may play a role in limiting prostate tumor progression in certain contexts, and deletion of PSCA may promote tumor migration and metastasis. 26815912_Increase in prostate stem cell antigen expression in prostatic hyperplasia induced by testosterone and 17beta-estradiol in C57BL mice |
ENSG00000167653 |
PSCA |
24.578353 |
0.0137042948 |
-6.189228 |
1.17671778 |
44.294240 |
0.00000000002825464108192062019642407310885447993195618288098103221273049712181091308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000004776633662151108251215474470206204943290018150037212762981653213500976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.661857173781724 |
0.60171491873616 |
48.2956024394994 |
15.7821610374912 |
| ENSMUSG00000022602 |
11838 |
Arc |
protein_coding |
Q9WV31 |
FUNCTION: Master regulator of synaptic plasticity that self-assembles into virion-like capsids that encapsulate RNAs and mediate intercellular RNA transfer in the nervous system (By similarity). ARC protein is released from neurons in extracellular vesicles that mediate the transfer of ARC mRNA into new target cells, where ARC mRNA can undergo activity-dependent translation (By similarity). ARC capsids are endocytosed and are able to transfer ARC mRNA into the cytoplasm of neurons (By similarity). Acts as a key regulator of synaptic plasticity: required for protein synthesis-dependent forms of long-term potentiation (LTP) and depression (LTD) and for the formation of long-term memory (PubMed:29264923, PubMed:24094104, PubMed:31151856). Regulates synaptic plasticity by promoting endocytosis of AMPA receptors (AMPARs) in response to synaptic activity: this endocytic pathway maintains levels of surface AMPARs in response to chronic changes in neuronal activity through synaptic scaling, thereby contributing to neuronal homeostasis (PubMed:17088213, PubMed:20211139, PubMed:20228806). Acts as a postsynaptic mediator of activity-dependent synapse elimination in the developing cerebellum by mediating elimination of surplus climbing fiber synapses (PubMed:23791196). Accumulates at weaker synapses, probably to prevent their undesired enhancement (By similarity). This suggests that ARC-containing virion-like capsids may be required to eliminate synaptic material (By similarity). Required to transduce experience into long-lasting changes in visual cortex plasticity and for long-term memory (PubMed:17088210, PubMed:20228806). Involved in postsynaptic trafficking and processing of amyloid-beta A4 (APP) via interaction with PSEN1 (PubMed:22036569). In addition to its role in synapses, also involved in the regulation of the immune system: specifically expressed in skin-migratory dendritic cells and regulates fast dendritic cell migration, thereby regulating T-cell activation (PubMed:28783680). {ECO:0000250|UniProtKB:Q63053, ECO:0000269|PubMed:17088210, ECO:0000269|PubMed:17088213, ECO:0000269|PubMed:20211139, ECO:0000269|PubMed:20228806, ECO:0000269|PubMed:22036569, ECO:0000269|PubMed:24094104, ECO:0000269|PubMed:28783680, ECO:0000269|PubMed:29264923, ECO:0000269|PubMed:31151856}. |
Cell membrane;Cell projection;Coiled coil;Cytoplasm;Cytoplasmic vesicle;Cytoskeleton;Developmental protein;Endocytosis;Endosome;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Postsynaptic cell membrane;Reference proteome;RNA-binding;Synapse;Transport;Ubl conjugation |
|
|
mmu:11838; |
acrosomal vesicle [GO:0001669]; actin cytoskeleton [GO:0015629]; cell cortex [GO:0005938]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; early endosome membrane [GO:0031901]; extracellular vesicle [GO:1903561]; glutamatergic synapse [GO:0098978]; membrane raft [GO:0045121]; neuronal ribonucleoprotein granule [GO:0071598]; plasma membrane [GO:0005886]; postsynaptic density membrane [GO:0098839]; postsynaptic endosome [GO:0098845]; actin binding [GO:0003779]; mRNA binding [GO:0003729]; anterior/posterior pattern specification [GO:0009952]; cell migration [GO:0016477]; cytoskeleton organization [GO:0007010]; endocytosis [GO:0006897]; endoderm development [GO:0007492]; learning [GO:0007612]; long-term memory [GO:0007616]; modulation of chemical synaptic transmission [GO:0050804]; mRNA transport [GO:0051028]; positive regulation of AMPA receptor activity [GO:2000969]; protein homooligomerization [GO:0051260]; regulation of cell morphogenesis [GO:0022604]; regulation of dendritic spine morphogenesis [GO:0061001]; regulation of long-term synaptic depression [GO:1900452]; regulation of long-term synaptic potentiation [GO:1900271]; regulation of neuronal synaptic plasticity [GO:0048168]; regulation of postsynaptic neurotransmitter receptor internalization [GO:0099149]; vesicle-mediated intercellular transport [GO:0110077] |
12493697_highly expressed both at the RNA and protein levels in postmeiotic germ cells in the testis of adult mice 12617955_Arc, an activity-regulated cytoskeletal gene, is expressed in suprachiasmatic nucleus neurons and is possibly involved in the light-induced phase-shift of circadian rhythm. 12663748_arg 3.1/arc expression is induced by a learning task, strongly suggesting a role of arg 3.1/arc mRNA in the early and late cellular mechanisms underlying the stabilization of the memory trace. 12763607_rapid induction of Arc is observed in the granule cell dendrites in the accessory olfactory bulb after mating 16237173_In transgenic mice with neuronal production of human amyloid precursor protein (hAPP) and hAPP-derived amyloid-beta peptides, basal Arc expression was reduced in the dentate gyrus. 16287845_early growth response factors Egr1 and Egr3 can indirectly modulate synaptic plasticity by directly regulating Arc 17088210_Arc/Arg3.1 is essential for the consolidation of synaptic plasticity and memories. 18230676_Galphas* mice show decreased Arc mRNA expression in CA1, orbital cortex, and cortical regions surrounding the hippocampus; however, only the deficits in cortical regions surrounding the hippocampus are PKA dependent. 18782777_ARC may protect cardiomyocytes from hypoxic cell death by regulating its downstream, Drbp1 and pMe(2)GlyDH 19074532_BDNF and Arc expression are altered in glucocorticoid receptor heterozygous mice on stress exposure, but gene x environment interactions may play a critical role for the development of a depressive episode in GR-compromised individuals. 19116276_A major synaptic activity-responsive elements was found as a unique approximately 100-bp element located at >5 kb upstream of the Arc/Arg-3.1 transcription initiation 19556514_reductions in both the number of Arc-activated neurons and the levels of Arc mRNA were seen in the neocortices of three different transgenic mouse models of cerebral amyloidosis 19559013_Activity-regulated cytoskeleton-associated protein (Arc) shows a persistent increase in expression level in normothermic mice compared to hypothermic. 20211139_Disruption of Ube3A function in neurons leads to an increase in Arc expression and a concomitant deregulation in AMPA receptors at synapses; which may contribute to the cognitive dysfunction that occurs in Angelman Syndrome. 20228806_Arc protein is required for the experience-dependent processes that normally establish and modify synaptic connections in the visual cortex. 20452974_PDGF has a role in induction of the synaptic plasticity gene Arc/Arg3.1 20505084_This study demonstrated that Arc/Arg3.1 plays a selective role in regulating visual experience-dependent homeostatic plasticity of excitatory synaptic transmission in vivo. 20550627_Increased Arc response to novelty could be suggested as a molecular mechanism underlying failure to adapt to environmental changes, common to chronic stress and altered glutamate function. 20653942_these data suggest that Arc/Arg3.1 dependent long term synaptic changes in spinal pain transmission are a feature of anti-nociceptive, i.e. enkephalinergic, rather than pro-nociceptive neurons. 20694003_SRF binding to SRE 6.9 in the Arc promoter is required for the late phase of cerebellar long-term depression 20806412_Arc transcription is dramatically suppressed in acute slices and negatively regulated by the activation of GABAA receptors, and positively regulated by the activation of muscarinic receptors. 20817066_Cognitive improvement of schizophrenic histopathology may be related to a restoration of synaptophysin and/or Arc levels in the frontal cortex. 20921410_by linking spine morphology with AMPAR endocytosis, Arc balances synaptic downscaling with increased structural plasticity 21315255_Arc/Arg3.1 is a context-dependent Notch regulator of neuronal signaling. 21334422_Kinetics and/or detectability of cortical subcellular is consistent with Arc/Arg3.1 mRNA expression being altered by the initial exposure to the sound, suggesting exposure-induced modifications in the cytoplasmic Arc/Arg3.1 mRNA pool. 21371562_The subset of cells that were initially activated during conditioning in the lateral amygdala preferentially transcribed Arc during subsequent rest. This was most frequently observed in conditioned mice that formed robust associative fear memory. 21497182_Results demonstrate that Arc is induced from wheel running but not movement in cages without wheels. 21559295_dopamine D1 receptor agonists cause Erk-dependent increases in Zif268 and Arc/Arg3.1 expression in mouse dentate gyrus 22036569_Study reports that the immediate early gene Arc is required for activity-dependent generation of Abeta. Arc is a postsynaptic protein that recruits endophilin2/3 and dynamin to early/recycling endosomes that traffic 22179607_the importance of intracellular mGluR5 in the cascade of events associated with sustained synaptic transmission 22922786_the disruption in Arc patterns reveals plaque-associated interference with neural network integration. 22928932_Neuronal subpopulations activated during fear memory retrieval preferentially transcribe Arc during subsequent rest in the lateral amygdala. 23598246_Novelty exposure induced an increase in Arc and c-Fos mRNA in the medial prefrontal cortex (mPFC), parietal cortex, and hippocampal formation in both APP/PS1DeltaE9 transgenic and wild-type mice 23749147_This study demonistrated that Arc mediates the homeostatic response to increased activity by translocating to the nucleus, increasing promyelocytic leukemia protein expression and decreasing GluA1 transcription, ultimately downscaling synaptic strength. 23891993_trace-conditioned mice, compared to backward-conditioned (stimulation-control), delay-conditioned and naive mice, exhibited elevated amygdalar Arc expression in the basolateral (BLA) but not the central (CeA) or the lateral amygdala (LA). 24012642_Molecular analysis revealed a remarkable decline in Arc expression in both hippocampus and cerebral cortex of amnesic mice, which was recovered after i-Extract treatment. Arc may be involved in i-Extract mediated recovery from amnesia. 24094104_dendritic translation of Arc underlies the priming of mGluR-long-term synaptic depression 24431439_Deficits in Arg3.1 and neuronal morphology lead to a deficit in spatial memory consolidation. 24758170_Arc is required for long-term extinction of conditioned fear. 24806683_Our findings therefore reveal an Arc-dependent molecular pathway by which gene-experience interaction regulates the emergence of persistent firing patterns in specific neuronal populations 24857654_Arc is necessary, but not sufficient, for MEF2-dependent synapse elimination. 25589774_Arc expression regulates the perpetuation of fear memoires. 25783003_Arc enhances dynamin polymerization and GTPase activation. 25797218_Transcriptional activity of early genes involved in memory process, such as activity regulated cytoskeletal-associated protein (Arc) gene, is increased in the hippocampus of hypercholesterolemic LDL receptor knockout mice. 25802982_Arc-expressing neurons have enhanced intrinsic excitability and are preferentially recruited into newly encoded memory traces. 25848016_The enhanced seizure-like activity, but not the increased ultrasonic vocalizations or motor deficits, is rescued in juvenile Angelman syndrome mice by genetically reducing the expression level of the cytoskeleton-associated protein, Arc. 25864631_The present study provides the first atomic structure for Arc and reveals that Arc N- and C-lobes evolved from the capsid domain of Ty3/Gypsy retrotransposon. 26051420_This study demonstrated a task-specific Arc-dependent cellular consolidation process in M2 cortex during motor learning. 26563879_These findings suggest that 11beta-HSD1 may contribute to the decline in Npas4 and Arc mRNA levels associated with memory impairment during ageing 26880136_we report that late Arc expression is essential for the persistence of newly-acquired and reactivated memories. Persistent fear memories could be a factor in post-traumatic stress disorder. 27038743_Arc/Arg3.1 effects on memory formation are not only manifested at the level of molecular pathways regulating synaptic plasticity, but also at the systems level. 27160884_The dynamics of the c-Fos and Arc expression has a biphasic pattern: the first peak was observed in 15-30 min after learning and the second less pronounced peak in 1-3 h. 27365296_These results indicate that dopamine is required to drive activity-dependent amplification of Arc mRNA in the developing postnatal frontal cortex. 27488634_This is a promising novel therapeutic approach because it appears to be effective in a model producing severe injury by interfering with an array of proximal signals and effectors of the ischemic cascade, upstream of JNK, caspases, and BIM and BAX activation. 27524619_Genetic disruption of Arc in mice causes alterations in dopamine and neurobehavioral phenotypes related to schizophrenia. 27567310_Activity-regulated cytoskeleton-associated protein accumulates in the nucleus in response to cocaine and acts as a brake on chromatin remodeling and long-term behavioral alterations. 27880914_CTCF knockdown attenuates fear-conditioning-induced hippocampal gene expression of key learning genes and loss of long-range interactions at the BDNF and Arc loci. 27995769_missense mutations in TRIAD3 abolished the interaction of TRIAD3A with Arc, disrupting Arc ubiquitination, and consequently Arc degradation. 28589041_Study demonstrates in mice that explored novel spatial environments, the involvement of Egr3 in regulating the late protein-dependent phase of Arc expression in the dentate gyrus. 28729244_Data indicate that ARC overexpression diminishes amyloid-induced JNK pathway activation and apoptosis in the beta-cell, a strategy that may reduce beta-cell loss in type 2 diabetes 28765602_These data indicate that suppression of CHOP by endogenous levels of ARC is critical for beta-cell viability and maintenance of normal islet structure in this model of type 2 diabetes. 28790183_Data show that although activity-dependent expression of activity regulated cytoskeletal-associated protein (Arc) mRNA does not change with age, expression of Arc protein is maximal during the critical period and declines in adulthood. 28963515_Arc expression did not correlate with performance in both adult and aged animals, suggesting that exploration of the testing environment, rather than the specific acquisition of the active place avoidance task, induces Arc expression in the dentate granule cell layer. 29045836_PSD95 was essential for Arc assembly into 1.5-MDa complexes and activity-dependent recruitment to excitatory synapses. 29135807_Stress from escapable electroshock (ES) was associated with up-regulation of brain-derived neurotrophic factor in amygdala as well as medial prefrontal cortex (mPFC). Stres from inescapable shock suppressed c-fos in mPFC (0 h) but induced more Arc in amygdala (2 h) in comparison with ES. 29142056_Our data support the hypothesis that the kinetics of Arc induction to refine cortical representations of sensory categories is sensitive to the familiarity of the sensory experience. 29162813_Specific acetylation of lysine residues on Arc promote Arc protein stability. 29264923_It appears that palmitoylation regulates at least a subset of Arc functions in synaptic plasticity. 29328916_Arc extracellular vesicles mediate intercellular transfer of Arc mRNA in neurons. 29635031_Sleep deprivation disrupts Arc expression in dentate gyrus neurons. 29861284_ArcKR (with mutation causing disrupted temporal control of degradation) mice had intact spatial learning but showed specific deficits in selecting an optimal strategy during reversal learning. This cognitive inflexibility was coupled to changes in Arc mRNA and protein expression resulting in a reduced threshold to induce mGluR-LTD and enhanced mGluR-LTD amplitude. 29912851_Arc expression increased in the amygdala and prefrontal cortex following conditioned stimulus presentation. 30279480_These data support a novel interaction and model where PICK1 mediates Arc regulation of AMPARs particularly under depolarizing conditions. 30442670_These results demonstrate that Arc/Arg3.1 mediates a critical period for spatial learning, during which Arc/Arg3.1 fosters maturation of hippocampal network activity necessary for future learning and memory storage. 30536651_Perturbed expression pattern of the immediate early gene Arc in the dentate gyrus of GluA1 C-terminal palmitoylation-deficient mice. 30584064_these data reveal that multiple levels of Upf1-mediated inhibition of Arc gene expression may allow neurons to more effectively respond to changes in neuronal activity. 30761730_Global Arc knockout mice have altered AMPAR-subunit surface levels in the adult NAc, and the Arc-deficient mice show reductions in anxiety-like behavior, deficits in social novelty preference, and antidepressive-like behavior. 30786014_Arc/Arg3.1 expression is independent of ontogeny and phenotype and although it is restricted to a small fraction within each migDC subset and LCs, Arc/Arg3.1(+) migDCs are important to facilitate infectious migration. 31110186_Study demonstrates that the immediate early gene, Activity Regulated Cytoskeleton Associated Protein (Arc), is selectively and persistently expressed in mouse dentate gyrus of the hippocampus engram cells after fear conditioning. 31151856_Arc oligomerization is regulated by CaMKII phosphorylation of the GAG domain: an essential mechanism for plasticity and memory formation. 31488612_It remains to be seen whether misregulation of Arc/Arg3.1 might critically imbalance brain networks and lead to emergence of schizophrenia 31682894_The activity-regulated cytoskeleton-associated protein, Arc/Arg3.1, influences mouse cocaine self-administration. 31904548_Temporal dynamics of Arc/Arg3.1 expression in the dorsal striatum during acquisition and consolidation of a motor skill in mice. 32114002_The temporal effect of hippocampal Arc in the working memory paradigm during novelty exploration. 33451279_The mechanism of enriched environment repairing the learning and memory impairment in offspring of prenatal stress by regulating the expression of activity-regulated cytoskeletal-associated and insulin-like growth factor-2 in hippocampus. 33667344_Conversion of the death inhibitor ARC to a killer activates pancreatic beta cell death in diabetes. 34077555_Phosphorylation-dependent control of Activity-regulated cytoskeleton-associated protein (Arc) protein by TNIK. 34326146_Intercellular Arc Signaling Regulates Vasodilation. 36095210_Real-time imaging of Arc/Arg3.1 transcription ex vivo reveals input-specific immediate early gene dynamics. |
ENSG00000198576 |
ARC |
73.226832 |
2.3745358203 |
1.247646 |
0.29202771 |
18.166976 |
0.00002023574575951966121774294737889476891723461449146270751953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001415201466524350284049288406507116633292753249406814575195312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
102.283073863024 |
20.8456285023262 |
43.0749761656888 |
6.86911928438483 |
| ENSMUSG00000022659 |
14525 |
Gcsam |
protein_coding |
Q6RFH4 |
FUNCTION: Involved in the negative regulation of lymphocyte motility. It mediates the migration-inhibitory effects of IL6. Serves as a positive regulator of the RhoA signaling pathway. Enhancement of RhoA activation results in inhibition of lymphocyte and lymphoma cell motility by activation of its downstream effector ROCK. Is a regulator of B-cell receptor signaling, that acts through SYK kinase activation. {ECO:0000250}. |
Alternative splicing;Cell membrane;Cytoplasm;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:14525; |
cytoplasm [GO:0005737]; plasma membrane [GO:0005886]; actin binding [GO:0003779]; myosin II binding [GO:0045159]; protein kinase binding [GO:0019901]; negative regulation of lymphocyte migration [GO:2000402]; regulation of B cell receptor signaling pathway [GO:0050855] |
15482850_Assays identified a non-consensus binding site for Sp factors near sites of transcriptional initiation. The site binds Spl and Sp3 in nuclear extracts. |
ENSG00000174500 |
GCSAM |
40.537381 |
0.0084166523 |
-6.892538 |
1.11650966 |
77.802043 |
0.00000000000000000113902289626739790800061571860234569368046644897654205361159363008027867181226611137390136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000407267529482277069187849536018686806812050099342679271297029686138557735830545425415039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.6562199549356 |
0.560086842863855 |
77.9668601194106 |
19.82731480447 |
| ENSMUSG00000022793 |
56375 |
B4galt4 |
protein_coding |
Q9JJ04 |
FUNCTION: Galactose (Gal) transferase involved in the synthesis of terminal N-acetyllactosamine (LacNac) unit present on glycan chains of glycoproteins and glycosphingolipids. Catalyzes the transfer of Gal residue via a beta1->4 linkage from UDP-Gal to the non-reducing terminal N-acetyl glucosamine 6-O-sulfate (6-O-sulfoGlcNAc) in the linearly growing chain of both N- and O-linked keratan sulfate proteoglycans. Cooperates with B3GNT7 N-acetyl glucosamine transferase and CHST6 and CHST1 sulfotransferases to construct and elongate mono- and disulfated disaccharide units [->3Galbeta1->4(6-sulfoGlcNAcbeta)1->] and [->3(6-sulfoGalbeta)1->4(6-sulfoGlcNAcbeta)1->] within keratan sulfate polymer. Transfers Gal residue via a beta1->4 linkage to terminal 6-O-sulfoGlcNAc within the LacNac unit of core 2 O-glycans forming 6-sulfo-sialyl-Lewis X (sLex). May contribute to the generation of sLex epitope on mucin-type glycoproteins that serve as ligands for SELL/L-selectin, a major regulator of leukocyte migration. In the biosynthesis pathway of neolacto-series glycosphingolipids, transfers Gal residue via a beta1->4 linkage to terminal GlcNAc of a lactotriaosylceramide (Lc3Cer) acceptor to form a neolactotetraosylceramide. {ECO:0000250|UniProtKB:O60513}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lipid metabolism;Manganese;Membrane;Metal-binding;Reference proteome;Secreted;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000250|UniProtKB:O60513}.; PATHWAY: Glycolipid biosynthesis. {ECO:0000250|UniProtKB:O60513}. |
|
mmu:56375; |
extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; galactosyltransferase activity [GO:0008378]; metal ion binding [GO:0046872]; N-acetyllactosamine synthase activity [GO:0003945]; UDP-galactosyltransferase activity [GO:0035250]; carbohydrate metabolic process [GO:0005975]; glycosylation [GO:0070085]; keratan sulfate biosynthetic process [GO:0018146]; lactosylceramide biosynthetic process [GO:0001572]; protein glycosylation [GO:0006486] |
Human_homologues 28228616_expression of the beta4GalT4 gene is controlled by Sp1, and Sp1 plays a key role in the activation of the beta4GalT4 gene in colon cancer cells. 32827291_N-glycosylation of the human beta1,4-galactosyltransferase 4 is crucial for its activity and Golgi localization. 34270095_The critical role of B4GALT4 in promoting microtubule spindle assembly in HCC through the regulation of PLK1 and RHAMM expression. |
ENSG00000121578 |
B4GALT4 |
94.226924 |
0.4570600279 |
-1.129544 |
0.31079046 |
12.950041 |
0.00031991455858161756522009544490003918326692655682563781738281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016843894076443827710620748661085599451325833797454833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.2228081316083 |
12.8568930076371 |
129.573370041659 |
20.277771722802 |
| ENSMUSG00000022847 |
21832 |
Thpo |
protein_coding |
P40226 |
FUNCTION: Lineage-specific cytokine affecting the proliferation and maturation of megakaryocytes from their committed progenitor cells. It acts at a late stage of megakaryocyte development. It may be the major physiological regulator of circulating platelets. |
Alternative splicing;Cytokine;Disulfide bond;Glycoprotein;Hormone;Reference proteome;Secreted;Signal |
|
This gene encodes a humoral growth factor necessary for megakaryocyte proliferation and maturation, as well as for thrombopoiesis. The encoded protein is a ligand for the product of the myeloproliferative leukemia virus oncogene. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Mar 2010]. |
mmu:21832; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; hormone activity [GO:0005179]; signaling receptor binding [GO:0005102]; cell population proliferation [GO:0008283]; megakaryocyte development [GO:0035855]; megakaryocyte differentiation [GO:0030219]; myeloid cell differentiation [GO:0030099]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of hematopoietic stem cell proliferation [GO:1902035]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of megakaryocyte differentiation [GO:0045654]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein phosphorylation [GO:0001934]; receptor signaling pathway via STAT [GO:0097696]; thrombopoietin-mediated signaling pathway [GO:0038163] |
12005542_Administration of recombinant human IL11 after supralethal radiation exposure promotes survival in mice: interactive effect with thrombopoietin 12101178_glycan domain of thrombopoietin acts in trans to enhance secretion of the hormone and other cytokines 12163458_Thrombopoietin expands hematopoietic stem cells after transplantation 12855555_p38 mitogen-activated protein kinase (MAPK) was responsible for the TPO-induced Hoxb4 elevation 14726379_Mpl stimulation by TPO results in the activation of Lyn kinase, which appears to limit the proliferative response through a signaling cascade that regulates MAPK activity 15254242_Thrombopoietin induces HOXA9 nuclear transport in immature hematopoietic cells 15337790_mpl-tpo signaling pathways are negatively modulated by lnk 15642952_is strongly proapoptotic in the brain, causing death of newly generated neurons through the Ras-extracellular signal-regulated kinase 1/2 pathway 15665119_TPO(high) and GATA-1(low) alterations are linked in an upstream-downstream relationship along a pathobiologic pathway leading to development of myelofibrosis 15927672_Development of myelofibrosis and osteosclerosis depends on local TPO levels in Bone marrow cells. 16022585_Thrombopoietin synthesis is induced by IL-6 in the liver during acute inflammation. 17284614_results suggest that a balance in positive and negative signals downstream from the TPO signal plays a role in the regulation of the probability of self-renewal in HSCs 17468237_thrombopoietin has a role in mast cell differentiation [review] 17546634_Chronic TPO overexpression induces mesangioproliferative glomerulopathy. 17925492_roles for FAK in megakaryocyte growth and platelet function, setting the stage for manipulation of this component of the Tpo signaling apparatus for therapeutic benefit. 18410987_Transcriptional regulation of bone marrow thrombopoietin by platelet proteins.( 18473128_TPO induces HIF-1alpha expression in a manner very similar to that of hypoxia. 18773906_thrombopoietin potentiates vasculogenesis by enhancing motility and enlivenment of transplanted endothelial progenitor cells via activation of Akt/mTOR/p70S6kinase signaling pathway 18814950_TPO downmodulates c-Myb expression through induction of miR-150. 21149592_The implication of Tpo as a mediator of neuronal damage in experimental pneumococcal meningitis is reported. 21507308_Signal transduction pathway of ERK1/2 participates in the activation of thrombopoietin in inflammatory injury of BV2 cells. 22284746_Findings establish that Clock regulates Thpo and Mpl expression in vivo, and demonstrate an important link between the body's circadian timing mechanisms and megakaryopoiesis. 24184684_IEX-1 has a role in activation of Erk and NF-kB pathways, which affects thrombopoietin-promoted NHEJ DNA repair in hematopoietic stem cells 25137252_Furthermore, although the contribution of the TPO treated graft to long-term hematological engraftment was reduced, the TPO treated and untreated grafts both contributed significantly to long-term chimerism in vivo. 25212676_TPO treatment also promoted the peripheral induction of Foxp3(+) Tregs in conjunction with elevated circulating TGF-beta levels. 25298035_Thrombopoietin/MPL signaling confers growth and survival capacity to CD41-positive cells in a mouse model of Evi1 leukemia. 25339357_Mpl expression, but not Tpo, is fundamental in the development of JAK2V617F(+) myeloproliferative neoplasms 25451253_Megakaryocytes regulate cell cycle quiescence of hematopoietic stem cell through the production of thrombopoietin. 25564715_novel findings about aspects of TPO action on stem cells 26702099_Thrombopoietin-mediated phosphorylation of STAT5 triggers its genome-wide relocation to STAT consensus sites, resulting in loss of a uSTAT5 program that restrains megakaryocytic differentiation and activation of STAT5-driven gene expression. 27344013_TPO and its receptor Mpl are dispensable for platelet survival in adult mice 29217821_Increased TPO expression in HSCs following IL-11 exposure could be mimicked or blocked by inhibiting or overexpressing miR-204-5p, respectively... IL-11 promoted Hematopoietic stem cell engraftment in a mouse model of aplastic anemia an effect that was attenuated in cells overexpressing miR-204-5p. 29622652_this study assessed the physiological source of thrombopoietin, a key cytokine required for maintaining hematopoietic stem cell . 30005133_Authors demonstrate that thrombopoietin and its receptor MPL are critical for the human JAK2V617F-bearing MK-induced myeloproliferation in mice, both by directly affecting the quantity and quality of MKs and by altering the MK-endothelial interaction and vascular niche function. 30043083_The number of lung metastases was significantly lower in Tpo(-/-) mice compared to Tpo(+/+) mice. The same was true for the bone-tropic 4T1.2 derivative. 30428347_Thpo-regulated mitochondrial activity associated with mitochondrial translocation of STAT3 phosphorylated at serine 727. 31301081_Study of hepatocarcinogenesis induced by diethylnitrosamine in B6C3F1 mice, the BrafV637E mutation, corresponding to the human BRAFV600E mutation, suggested that the TPO-mediated macroenvironmental changes involving the liver and bone marrow may promote proliferation of the BRAFV600E-mutated hepatocytes, but cause hematological, renal, and pulmonary disorders that lead to death. 32721504_Erythropoietin is a major regulator of thrombopoiesis in thrombopoietin-dependent and -independent contexts. 33657206_Prolonged maintenance of hematopoietic stem cells that escape from thrombopoietin deprivation. 35294228_Hematopoietic stem cells temporally transition to thrombopoietin dependence in the fetal liver. 35358295_Megakaryocyte/platelet-derived TGF-beta1 inhibits megakaryopoiesis in bone marrow by regulating thrombopoietin production in liver. |
ENSG00000090534 |
THPO |
47.252033 |
0.4630887194 |
-1.110639 |
0.37000526 |
8.889069 |
0.00286883003476746945015207757023745216429233551025390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0115664423212223643755436341962195001542568206787109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
30.6314801937517 |
7.94240387860986 |
66.1460297200878 |
12.2422214948312 |
| ENSMUSG00000022865 |
13052 |
Cxadr |
protein_coding |
P97792 |
FUNCTION: Component of the epithelial apical junction complex that may function as a homophilic cell adhesion molecule and is essential for tight junction integrity. Also involved in transepithelial migration of leukocytes through adhesive interactions with JAML a transmembrane protein of the plasma membrane of leukocytes. The interaction between both receptors also mediates the activation of gamma-delta T-cells, a subpopulation of T-cells residing in epithelia and involved in tissue homeostasis and repair. Upon epithelial CXADR-binding, JAML induces downstream cell signaling events in gamma-delta T-cells through PI3-kinase and MAP kinases. It results in proliferation and production of cytokines and growth factors by T-cells that in turn stimulate epithelial tissues repair. {ECO:0000269|PubMed:10814828, ECO:0000269|PubMed:20813954, ECO:0000269|PubMed:9036860, ECO:0000269|PubMed:9420240}. |
3D-structure;Alternative splicing;Cell adhesion;Cell junction;Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Receptor;Reference proteome;Repeat;Secreted;Signal;Tight junction;Transmembrane;Transmembrane helix |
|
This gene encodes a protein that is part of the Cortical Thymocyte marker in Xenopus (CTX) subfamily within the immunoglobulin superfamily. Members of this subfamily, predominantly expressed on the surface of endothelial and epithelial cells, help establish cell polarity and provide a barrier function, regulating migration of immune cells. This protein, first identified as the receptor for adenovirus subgroup C and coxsakieviruses group B, is developmentally regulated and plays an important role in cardiac development. Alternative splicing results in multiple transcript variants that encode different protein isoforms. [provided by RefSeq, Jan 2013]. |
mmu:13052; |
acrosomal vesicle [GO:0001669]; adherens junction [GO:0005912]; apicolateral plasma membrane [GO:0016327]; basolateral plasma membrane [GO:0016323]; bicellular tight junction [GO:0005923]; cell body [GO:0044297]; cell junction [GO:0030054]; cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; extracellular space [GO:0005615]; filopodium [GO:0030175]; growth cone [GO:0030426]; intercalated disc [GO:0014704]; membrane raft [GO:0045121]; neuromuscular junction [GO:0031594]; neuron projection [GO:0043005]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; beta-catenin binding [GO:0008013]; cell adhesion molecule binding [GO:0050839]; cell adhesive protein binding involved in AV node cell-bundle of His cell communication [GO:0086082]; connexin binding [GO:0071253]; identical protein binding [GO:0042802]; integrin binding [GO:0005178]; PDZ domain binding [GO:0030165]; signaling receptor binding [GO:0005102]; actin cytoskeleton reorganization [GO:0031532]; AV node cell to bundle of His cell communication [GO:0086067]; AV node cell-bundle of His cell adhesion involved in cell communication [GO:0086072]; cardiac muscle cell development [GO:0055013]; cardiac muscle cell proliferation [GO:0060038]; cell-cell adhesion [GO:0098609]; cell-cell junction organization [GO:0045216]; defense response to virus [GO:0051607]; epithelial structure maintenance [GO:0010669]; gamma-delta T cell activation [GO:0046629]; germ cell migration [GO:0008354]; heart development [GO:0007507]; heterophilic cell-cell adhesion via plasma membrane cell adhesion molecules [GO:0007157]; homotypic cell-cell adhesion [GO:0034109]; mitochondrion organization [GO:0007005]; negative regulation of cardiac muscle cell proliferation [GO:0060044]; neutrophil chemotaxis [GO:0030593]; positive regulation of epithelial cell proliferation involved in wound healing [GO:0060054]; regulation of AV node cell action potential [GO:0098904]; transepithelial transport [GO:0070633] |
11479928_Review: Receptor for the group B coxsackieviruses and adenoviruses, CAR 12468544_Coxsackievirus and adenovirus receptor (CAR) is found in complex with the PDZ domain-containing protein ligand-of-numb protein-X (LNX) 12823902_The mCAR gene is situated on the distal portion of murine chromosome 16, and is composed of at least eight exons, with intron-exon boundaries similar to those reported for the human CAR gene 15864812_CAR provides positive survival signals to cardiomyocytes that are essential for normal heart development. 15979067_CAR is a partner in a protein complex organized at specific subcellular sites by Ligand-of-Numb Protein-X2 16543498_During a specific temporal window, CAR expression on cardiomyocytes is essential for normal cardiac development. 16806202_Attachment of mouse adenovirus type 1 (MAV-1) to Chinese hamster ovary (CHO) cells was not increased by stable transfection with mouse CAR, whereas binding efficiency of Ad2 was 20-fold higher in the mouse CAR-transfectant compared to wild type cells. 16841169_CAR is an immunoglobulin domain-containing protein that bears homology to cell-adhesion molecules suggesting the possibility that it may participate in organization of the developing olfactory system. 17148677_Overexpression of CAR may lead to physiological dysfunction by disturbing sarcolemmal integrity (through dystrophin deficiency), impairing sarcolemmal repair (through dysferlin deficiency), and interfering with normal signaling . 17546646_present work shows that stable and transient expression of CAR in tumor cells reduced lung metastasis in an experimental lung metastasis model; CAR expression likely acts as a metastatic suppressor 17690169_A function for CAR in testis morphogenesis and in migration of germ cells across the blood-testis barrier during spermatogenesis. 18329552_Activation of STAT1 protein is an early event in coxsackievirus-induced myocarditis preceding enhanced CAR expression during tissue reorganization. 18636119_CAR mediates atrioventricular-node function in the murine heart. 18636119_This manuscript demonstrates a unique role for CAR in the AV-node related to localization of connexin 45 to the cell-cell junction. Atrioventricular node block occurs in the absence CAR. 18779655_Systemic delivery of PEGPE-adenovirus complex is an effective tool of adenoviral delivery as it overcomes limitation due to CAR deficiency of target cells while reducing hepatic uptake and enhancing adenoviral gene expression in tumors. 18794341_Results indicate that CAR is not only relevant for virus uptake and cardiac remodeling but also has a previously unknown function in the propagation of excitation from the atrium to the ventricle. 19461877_it is proposed that adenovirus CAV-2 transport in motor neuron is dictated by an innate trafficking of CAR, suggesting an unsuspected function for this adhesion protein during neuronal homeostasis 19616768_These results demonstrate a critical function for coxsackievirus and adenovirus receptor in the pathogenesis of group B coxsackievirus infection in vivo and in virus tropism for the heart and pancreas. 19850873_CAR activation dramatically improves fatty liver and ameliorates hyperglycemia. 20144615_increased CAR expression has a role in inducing cardiomyopathy 20181587_Blocking antibodies were found to inhibit neurite extension in retina organ and retinal explant cultures, whereas the application of extracellular CAR domains promoted neurite extension and adhesion to extracellular matrices. 20530002_Adoptive transfer of Tregs protected mice from coxsackievirus B3-induced myocarditis through the transforming growth factor beta-coxsackie-adenovirus receptor pathway. 20813955_crystal structure of junctional adhesion molecule-like protein (JAML) and coxsackie and adenovirus receptor; data show how CAR-mediated clustering of JAML recruits phosphoinositide 3-kinase to a JAML intracellular sequence motif 21352828_Data show for the first time that CAR upregulation in the adult mouse heart induces cardiac inflammation reminiscent of early viral myocarditis. 21674029_CAR exerts important functions in the physiology of several organs in vivo 22282254_with significant difference). At dpc 8.5, CAR expression was increased slightly again. It is concluded that during implantation, the expression of CAR mRNA and protein is declined, resulting in the impairment of tight junction. 22624044_CAR plays an essential role in development of the lymphatic vasculature in the mouse embryo by promoting appropriate formation of lymphatic endothelial cell-cell junctions. 23620416_These findings indicate that CAR plays an important role in the initiation of coxsackievirus B virus infections and is closely associated with hepatotropism and age-specific susceptibility. 24015300_CAR belongs to the increasing list of cell surface molecules that undergo ectodomain shedding and that are substrates for -secretase-mediated RIP. 24291282_novel modifier of ventricular conduction and arrhythmia vulnerability in the setting of myocardial ischemia 24625359_Conclude that CXADR possesses no direct role in testicular physiology in vivo. 26076477_Podocyte-Specific Deletion of Murine CXADR Does Not Impair Podocyte Development, Function or Stress Response 26136571_the intracellular domain of CAR differentially regulates AdV entry and trafficking. Our study highlights the mechanistic differences that a receptor can have for two viruses from the same family. 27226313_In blastocysts, CAR was expressed at the cell contacts within the inner cell mass as well as in the trophectoderm (TE) where CAR was found together with ZO1 at the apical contacts, suggesting that CAR builds up apical TJs in TE and mediates cell adhesion in TE and inner cell mass. CAR was expressed in TE of implanting embryos as well as endometrial epithelium. 28381574_Coxsackievirus and Adenovirus Receptor and potentially an unidentified factor present in mouse serum might be important mediators of Human adenoviral serotype 5 transduction. 31083261_In this study, a CEACAM-1 overexpression vector was constructed, and the effects of CC1 overexpression post CVB3 infection were analyzed. The results suggested that CC1 may promote CAR expression in cardiomyocytes after CVB3 infection, and CAR may be a potential target for CC1 to regulate the CVB3-induced process of myocardial injury. CC1 might regulate CAR expression by activating the CC1-SYK- TNF-[alpha] signaling axis. 31373105_the Cxadr-Adam10 complex might moderate TJ integrity/stability and play pivotal roles during early embryonic development. 32852032_Induction of the Coxsackievirus and Adenovirus Receptor in Macrophages During the Formation of Atherosclerotic Plaques. 36674801_CXADR: From an Essential Structural Component to a Vital Signaling Mediator in Spermatogenesis. |
ENSG00000154639 |
CXADR |
20.642012 |
0.2842174940 |
-1.814933 |
0.61176674 |
8.871967 |
0.00289582986496967221407006221056690264958888292312622070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0116629509527281984698765526786701229866594076156616210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.36389480525296 |
3.65391209537263 |
29.4277973049627 |
8.48595361019826 |
| ENSMUSG00000022894 |
23794 |
Adamts5 |
protein_coding |
Q9R001 |
FUNCTION: Metalloproteinase that plays an important role in connective tissue organization, development, inflammation and cell migration. Extracellular matrix (ECM) degrading enzyme that shows proteolytic activity toward the hyalectan group of chondroitin sulfate proteoglycans (CSPGs) including ACAN, VCAN, BCAN and NCAN. Cleavage within the hyalectans occurs at Glu-Xaa recognition motifs. Plays a role in embryonic development, including limb and cardiac morphogenesis, and skeletal muscle development through its VCAN remodeling properties. Cleaves VCAN in the pericellular matrix surrounding myoblasts, facilitating myoblast contact and fusion which is required for skeletal muscle development and regeneration (PubMed:23233679). Participates in the development of brown adipose tissue and browning of white adipose tissue (PubMed:28702327). Plays an important role for T-lymphocyte migration from draining lymph nodes following viral infection (PubMed:27855162). {ECO:0000269|PubMed:15800625, ECO:0000269|PubMed:23233679, ECO:0000269|PubMed:27855162, ECO:0000269|PubMed:28702327}. |
Cleavage on pair of basic residues;Disulfide bond;Extracellular matrix;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Repeat;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes a member of 'a disintegrin and metalloproteinase with thrombospondin motifs' (ADAMTS) family of multi-domain matrix-associated metalloendopeptidases that have diverse roles in tissue morphogenesis and pathophysiological remodeling, in inflammation and in vascular biology. The encoded preproprotein undergoes proteolytic processing to generate an active, zinc-dependent aggrecanase enzyme. Mice lacking the encoded protein are protected from surgery-induced osteoarthritis and antigen-induced arthritis. [provided by RefSeq, May 2016]. |
mmu:23794; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; endopeptidase activity [GO:0004175]; extracellular matrix binding [GO:0050840]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; metalloendopeptidase activity [GO:0004222]; metallopeptidase activity [GO:0008237]; peptidase activity [GO:0008233]; zinc ion binding [GO:0008270]; aortic valve morphogenesis [GO:0003180]; defense response to bacterium [GO:0042742]; endocardial cushion morphogenesis [GO:0003203]; extracellular matrix disassembly [GO:0022617]; extracellular matrix organization [GO:0030198]; myoblast fusion [GO:0007520]; negative regulation of cold-induced thermogenesis [GO:0120163]; proteolysis [GO:0006508]; pulmonary valve morphogenesis [GO:0003184]; tooth eruption [GO:0044691] |
11831030_extracellular matrix degrading enzyme 15659705_ADAMTS1, ADAMTS4, and ADAMTS5 are expressed in patterns that relate to the expression pattern of versican in granulosa cells of small follicles, expanded cumulus cell-oocyte complexes, and endothelial cells of the ovary. 15800624_After surgically induced joint instability, there was significant reduction in the severity of cartilage destruction in the ADAMTS5 knockout mice compared with wild-type mice 15800625_ADAMTS5 is the major aggrecanase in mouse cartilage, both in vitro and in a mouse model of inflammatory arthritis 17255106_ADAMTS-5 is entirely responsible for cleavage in the interglobular domain, but cleavage in the chondroitin sulfate-rich region may be driven by ADAMTS-4 17938173_aggrecan loss with aggrecan processing in mice with single and double deletions of ADAMTS-4 and -5 activity (Deltacat) 17968948_Deletion of ADAMTS-4/5 provided significant protection against proteoglycan degradation ex vivo and decreased the severity of murine osteoarthritis 19250981_Data show that expression of Adamts5 during neuromuscular development and in smooth muscle cells coincides with the broadly distributed proteoglycan versican, an ADAMTS5 substrate. 19565481_Fibroblast growth factor 2 is an intrinsic chondroprotective agent that suppresses ADAMTS-5 and delays cartilage degradation in murine osteoarthritis. 19684582_The occurrence of less severe osteoarthritis-like cartilage destruction in both syndecan-4-deficient mice and syndecan-4-specific antibody-treated wild-type mice results from a marked decrease in ADAMTS-5 activity. 19922873_show that combinatorial mouse alleles for the secreted metalloproteases Adamts5, Adamts20 (bt), and Adamts9 result in fully penetrant soft-tissue syndactyly. 21337391_ADAMTS5 ablation did not eliminate aggrecanase activity from the articular cartilage but blocked fibrosis and resulted in the accumulation of aggrecan in the articular cartilage. 21539915_Matrilin-4 is processed by ADAMTS-5 in late Golgi vesicles present in growth plate chondrocytes of defined differentiation state 21566131_role of ADAMTS5 in dermal repair through CD44-mediated aggrecan accumulation and modulation of transforming growth factor beta1 (TGFbeta1) signaling 21749862_Altered versican cleavage in ADAMTS5 deficient mice; a novel etiology of myxomatous valve disease 21800360_Adamts5 induction in joint components other than cartilage, and its post-translational activation by PACE4 and/or furin may be important in the pathophysiology of arthritis. 21828051_a physiological function of ADAMTS5 in dermal fibroblasts is to maintain optimal versican content and PCM volume by continually trimming versican in hyaluronan-versican aggregates. 21928430_The role of ADAMTS5 in tendon is to remove pericellular and interfibrillar aggrecan to maintain the molecular architecture responsible for normal tissue function. 22493487_the first evidence implicating ADAMTS-5 in the regulation of proteoglycan turnover and lipoprotein retention in atherosclerosis. 23082219_The serine protease tissue plasminogen activator (tPA) and two matrix metalloproteinases, ADAMTS-4 and ADAMTS-5, were identified as Reelin cleaving enzymes. 23154421_The present study reveals ADAMT-5 expression by mast cells(MCs) and that MC activation regulates the expression of the protease, thus implicating the ADAMT-5 of protease in MC function. 23233679_Versican processing by ADAMTS5 and ADAMTS15 contribute to muscle fiber formation. 23436205_this study identified, for the first time, several genes that have an ADAMTS-5-independent role in osteoarthritis(OA), identifying them as possible OA initiation candidates. 23562508_Western blot analyses indicated that aggrecanase-generated proteoglycan fragments are produced after SCI. 23754494_Repair of biomechanically compromised tendons exhibiting midsubstance chondroid accumulation requires ADAMTS5. 23963448_RelA/p65 is a potent transcriptional activator of ADAMTS5 in chondrocytes during osteoarthritis development. 25101296_aggrecan and brevican proteolysis is compensated in Adamts4-/- or Adamts5-/- mice by ADAMTS proteoglycanase family members but a threshold of versican proteolysis is sensitive to the loss of a single ADAMTS proteoglycanase during spinal cord injury 25840345_TS5 protein functions to suppress glucose uptake in adipose-derived stromal cells and thereby inhibits the synthesis, and promotes the intracellular degradation of Acan and Vcan by an ADAMTS other than TS5. 26668318_Data suggest that ADAMTS-5 oligomerization is required for full aggrecanase activity in vitro and in situ (as seen in knee joint of mouse model of inflammatory arthritis); thus, blocking oligomerization inhibits ADAMTS-5 activity. 27254774_Adamts5(-/-) mice were protected from hepatic mitochondrial dysfunction, as indicated by increased mitochondrial respiratory chain complex activity, higher ATP levels and higher expression of antioxidant enzymes. Absence of ADAMTS5 preserves liver integrity in a diet-induced obesity model. 27855162_research emphasises the importance of ADAMTS5 expression in the control of influenza virus infection and highlights the potential for development of ADAMTS5-based therapeutic strategies to reduce morbidity and mortality 28570296_ADAMTS5 is the primary aggrecanase mediating smoking-induced intervertebral disc degeneration. 28702327_ADAMTS5 plays a functional role in development of brown adipose tissue and browning of white adipose tissue. 29293679_Energy expenditure and thermogenesis were not significantly different between KO and WT ADAMTS5-J mice (in contrast to somewhat enhanced levels in ADAMTS5-P mice). Insulin sensitivity was improved in the ADAMTS5-J KO mice, and they were protected against non-alcoholic steatohepatitis in the DIO model 29720262_Upper zone of growth plate and cartilage matrix associated protein (Ucma) physically interacted with a disintegrin-like and metalloprotease (reprolysin type) with thrombospondin type 1 motif, 5 (aggrecanase-2) protein (ADAMTS5) and blocked its aggrecanase activity in chondrocyte cultures. 30958594_In the absence of ADAMTS5, cleavage of versican in the cardiac extracellular matrix is impaired, but cardiac function, even upon exposure to a HFD, is not markedly affected. 31608494_Smo deletion mediates articular cartilage degeneration by ADAMTS-5 and Runx2 suppression 31676809_Degrading products of chondroitin sulfate can induce hypertrophy-like changes and MMP-13/ADAMTS5 production in chondrocytes. 32156081_Treatment of Dystrophic mdx Mice with an ADAMTS-5 Specific Monoclonal Antibody Increases the Ex Vivo Strength of Isolated Fast Twitch Hindlimb Muscles. 32648916_Cerebral cavernous malformations are driven by ADAMTS5 proteolysis of versican. 33561540_ADAMTS5 is required for normal trabeculated bone development in the mandibular condyle. 33934089_Internal modulation of proteolysis in vascular extracellular matrix remodeling: role of ADAM metallopeptidase with thrombospondin type 1 motif 5 in the development of intracranial aneurysm rupture. 34806902_Extracellular Matrix in Heart Failure: Role of ADAMTS5 in Proteoglycan Remodeling. 35913086_The adaptive response of the mandibular condyle to increased load is disrupted by ADAMTS5 deficiency. |
ENSG00000154736 |
ADAMTS5 |
345.632354 |
0.4305488983 |
-1.215751 |
0.14559756 |
69.161690 |
0.00000000000000009071307013931568390704467161873614372953828280388144378321158001199364662170410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000026987138366446417934498052859103516288859052341120303708521532826125621795654296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
206.377691384335 |
20.8631212971422 |
479.336243057179 |
34.758827419729 |
| ENSMUSG00000022895 |
23872 |
Ets2 |
protein_coding |
P15037 |
FUNCTION: Transcription factor activating transcription. Binds specifically the GGA DNA motif in gene promoters and stimulates transcription of those genes (By similarity). {ECO:0000250}. |
DNA-binding;Nucleus;Phosphoprotein;Proto-oncogene;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:23872; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; nuclear glucocorticoid receptor binding [GO:0035259]; protein domain specific binding [GO:0019904]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; cell differentiation [GO:0030154]; ectodermal cell fate commitment [GO:0001712]; mesoderm development [GO:0007498]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of transcription by RNA polymerase II [GO:0045944]; primitive streak formation [GO:0090009]; regulation of transcription by RNA polymerase II [GO:0006357] |
11887334_decrease in ETS2 protein levels after exposure to electromagnetic fields 12128217_Ets2 expression pattern in E8.5 to E13.5 embryos 12391198_Ets2 plays a role in the proliferation and survival of thymocytes probably via a Myc-dependent pathway. 12393756_Results demonstrate that Ets-2 and related transcription factors are crucial for the normal formation of the neuromuscular junction and control the expression of a distinct set of synaptic genes. 12637547_ets-2 has a role as a repressor and indicate that components of the mammalian SNF/SWI complex are required as co-repressors 14612405_The size of mammary tumors transplanted into fat pads of mice with Ets2 targeted alleles was correlated directly with Ets2 activity. 15590657_Ets-2 contributes to the up-regulation of HO-1 by the potent inflammatory stimulus LPS in macrophages 16007139_inactivation of Ets2 within tumor cells has no effect on tumor appearance or growth. By contrast, complete inactivation of Ets2 in both epithelial and stromal cells moderates the early hyperplastic phase of tumor development 16481355_Ets2 is necessary in the trophoblast for fundamental aspects of anteroposterior (AP) epiblast axis initiation, including mesoderm initiation at the primitive streak 16741970_Data show that murine cdk10 binds Ets2 transcription factors in vitro, does not show a direct involvement in the G2/M transition and, therefore, does not affect the proliferation rate of the cell lines analyzed. 16799155_ETS2 repressor factor associates with active and inactive Erks through distinct FXF motifs 17977525_Ets2 contributes to the regulation of multiple genes important for maintaining the undifferentiated state of trophoblast stem cells and as candidate signals for embryonic development. 18958307_ETS-1 and ETS-2 are upregulated in ocular tumors derived from the retinal epithelium. 19109434_SRC-1 specifically promotes metastasis without affecting primary tumor growth through mediating Ets-2-mediated HER2 expression and activating CSF-1 expression for macrophage recruitment 19168790_Evidence for synergy between alterations in GATA-1 and overexpression of ETS proteins in aberrant megakaryopoiesis. 19179380_Data suggest that Ets-2 transcription factor activation of Bcl-xL gene may protect glia from constitutive oxidative stress that is thought to be a key mechanism contributing to the pathogenesis of amyotrophic lateral sclerosis . 19411629_Studies in Ets1-/-;Ets2A72/A72 double mutants show that loss of both Ets1 & Ets2 resulted in embryonic lethality at midgestation, with striking defects in vascular branching, indicating establish essential and overlapping functions for Ets1 & Ets2. 19764029_Mesoderm-derived cranial skeletal elements are more affected in down syndrome mice, Ets2(+/-) mice compared to littermates, suggesting a differential interaction of Ets2-related processes with mesoderm-derived and neural crest-derived formative tissues. 20569982_This study uncovers new roles for Ets2 in trophoblast stem cell self-renewal and differentiation 21425406_Ets2 regulates colonic stem cells and sensitivity to tumorigenesis. 21437904_silencing of Smad2 is able to prevent the suppression of both HO-1 and Ets-2 by TGF-beta1 during exposure to lipopolysaccharide. 21562315_These data indicate the importance of phosphorylated ets-2 in the pathogenesis of pulmonary fibrosis through the expression of Type I collagen and (myo)fibroblast activation. 21700929_Provide evidence for the plaque-destabilizing role of Ets2 in atherosclerosis development by induction of an intraplaque proinflammatory phenotype in endothelial cells. 21807940_transcriptional cofactor that enhances inflammatory lymphangiogenesis 21989915_Results identify ETS2 as a functionally important transcription factor in adipogenesis. 22740135_Ets-2 play a key role in transcriptional regulation of CTRP5 in muscle cells. 23552073_propose a model that provides a genetic explanation as to how Ets2 in trophoblast mediates the progression of gastrulation within the epiblast 23977064_The data reveals a key function for Ets2 in tumor fibroblasts in signaling to endothelial cells to promote tumor angiogenesis. 24277931_Data indicate that differences between the N termini of transcription factors Ets1 and Ets2, rather than differences in the DNA binding domains, determine whether the proteins are capable of blocking antibody-secreting cells (ASCs) formation or not. 24362029_identified Ets2 as a key novel regulator in both the positive and negative control of miR-155 in the inflammatory response. 24968297_MicroRNA 17-92 cluster mediates ETS1 and ETS2-dependent RAS-oncogenic transformation 25446535_Elf5 and Ets2 have roles in maintaining the mouse extraembryonic ectoderm in a dosage dependent synergistic manner 27659014_These results suggest an unappreciated role for ETS2 in fibroblasts in establishing an immune-suppressive microenvironment in response to oncogenic Kras(G12D) signaling during the initial stages of tumor development. 28244611_The differentiation of ERF-overexpressing trophoblast stem cell lines also suggests that ERF may have an FGF2-independent effect during the commitment towards syncytiotrophoblasts. 29021240_Homozygous deletion of Ets2 in p53 mutant mice resulted in strong down-regulation of snoRNAs and reversed the prometastatic phenotype of mutant p53 but had no effect on osteosarcoma development, which remained 100% penetrant. 31754329_ETS2 is identified as a novel regulator of insulin production and a potential therapeutic target for diabetes mellitus treatment. 31785145_Ets2 suppresses inflammatory cytokines through MAPK/NF-kappaB signaling and directly binds to the IL-6 promoter in macrophages. 31810605_Protein C-ets-2 epigenetically suppresses TLRs-induced interleukin 6 production in macrophages. 32203187_Ets-2 deletion in myeloid cells attenuates IL-1alpha-mediated inflammatory disease caused by a Ptpn6 point mutation. 33821668_Cooperative Binding of ETS2 and NFAT Links Erk1/2 and Calcineurin Signaling in the Pathogenesis of Cardiac Hypertrophy. |
ENSG00000157557 |
ETS2 |
479.727102 |
2.6359147387 |
1.398304 |
0.15203398 |
81.941465 |
0.00000000000000000014017734077405521105128246171088479678450130517233855565947808763382909091887995600700378417968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000053647770217849476101437221843877345248289695730213328533908523354511999059468507766723632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
665.112754788436 |
70.2206627209758 |
252.327112489129 |
20.3334570964919 |
| ENSMUSG00000023015 |
26934 |
Racgap1 |
protein_coding |
Q9WVM1 |
FUNCTION: Component of the centralspindlin complex that serves as a microtubule-dependent and Rho-mediated signaling required for the myosin contractile ring formation during the cell cycle cytokinesis. Required for proper attachment of the midbody to the cell membrane during cytokinesis. Plays key roles in controlling cell growth and differentiation of hematopoietic cells through mechanisms other than regulating Rac GTPase activity. Has a critical role in erythropoiesis (By similarity). Also involved in the regulation of growth-related processes in adipocytes and myoblasts. May be involved in regulating spermatogenesis and in the RACGAP1 pathway in neuronal proliferation. Shows strong GAP (GTPase activation) activity towards CDC42 and RAC1 and less towards RHOA. Essential for the early stages of embryogenesis. May play a role in regulating cortical activity through RHOA during cytokinesis. May participate in the regulation of sulfate transport in male germ cells. {ECO:0000250|UniProtKB:Q9H0H5, ECO:0000269|PubMed:10235109, ECO:0000269|PubMed:10493933, ECO:0000269|PubMed:10979956, ECO:0000269|PubMed:11287179}. |
Acetylation;Cell cycle;Cell division;Cell membrane;Coiled coil;Cytoplasm;Cytoplasmic vesicle;Cytoskeleton;Developmental protein;Differentiation;GTPase activation;Ion transport;Isopeptide bond;Lipid-binding;Membrane;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Spermatogenesis;Transport;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:26934; |
acrosomal vesicle [GO:0001669]; centralspindlin complex [GO:0097149]; cleavage furrow [GO:0032154]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; Flemming body [GO:0090543]; midbody [GO:0030496]; mitotic spindle [GO:0072686]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; spindle midzone [GO:0051233]; alpha-tubulin binding [GO:0043014]; beta-tubulin binding [GO:0048487]; gamma-tubulin binding [GO:0043015]; GTPase activator activity [GO:0005096]; metal ion binding [GO:0046872]; microtubule binding [GO:0008017]; phosphatidylinositol-3,4,5-trisphosphate binding [GO:0005547]; protein kinase binding [GO:0019901]; actomyosin contractile ring assembly [GO:0000915]; erythrocyte differentiation [GO:0030218]; mitotic cytokinesis [GO:0000281]; mitotic spindle midzone assembly [GO:0051256]; neuroblast proliferation [GO:0007405]; positive regulation of cytokinesis [GO:0032467]; regulation of attachment of spindle microtubules to kinetochore [GO:0051988]; regulation of embryonic development [GO:0045995]; Rho protein signal transduction [GO:0007266]; spermatogenesis [GO:0007283]; sulfate transport [GO:0008272] |
11775025_Rac regulates vascular endothelial growth factor stimulated motility 11942621_These results indicate that MgcRacGAP regulates cytokinesis and cellular differentiation as a regulator of Rho family of GTPase and suggest that this process is controlled by some serine/threonine kinases. 16365040_alpha5 is required for proliferation and polarity of basal epithelial cells; the interaction between laminin-10/11-integrin alpha6beta4 and the PI3-kinase-Cdc42/Rac pathways may play a role in determining the size and shape of tooth germ 16873377_Gab2 via Shp-2 is critical for transmitting signals from Kit Tyr(567) to activate the Rac/JNK pathway controlling mast cell proliferation 16959247_Data demonstrate that normal cytokinesis in B lymphocytes requires the GAP and NH2 terminal domains but not GAP activity of mgcRacGAP. 18180334_Rac became activated within 2 min after peripheral membrane extensions adhered to new ECM islands, and this activation wave propagated outward in an oriented manner as the cells spread from island to island 18541143_mgcRacGAP is now shown to play a indispensable role in the development of hematopoietic cells in vivo. 19394428_This study demonstrated that loss of Rac1 attenuates synapse structure and function in vivo, which ultimately leads to significant defects in spatial learning and working/episodic-like memory. 22683270_several Rho family small GTPases activate PI3K by an indirect cooperative positive feedback that required a combination of Rac, CDC42, and RhoG small GTPase activities 23696789_APC(CDH1) targets MgcRacGAP for destruction in the late M phase. 23997213_TLR4 signaling shapes B cell dynamics via MyD88-dependent pathways and Rac GTPases. 24355749_Deletion of MgcRacGAP in the male germ cells impairs spermatogenesis and causes male sterility in the mouse. 32423815_RacGAP1 ameliorates acute kidney injury by promoting proliferation and suppressing apoptosis of renal tubular cells. |
ENSG00000161800 |
RACGAP1 |
1846.787453 |
2.1515110170 |
1.105350 |
0.06671836 |
272.000443 |
0.00000000000000000000000000000000000000000000000000000000000041584096005358830220946495719826632955873113261562387080311220403277271089537558791707242481481652059375756345233465257624243911371378735200312743412482535676488737408362794667482376098632812500000000000 |
0.0000000000000000000000000000000000000000000000000000000000974475308236347127610233137775463325073830250216564347060895997472696484268479597822628103475370964212902752179593260953429441617418842788695245230035091310583084123209118843078613281250000000000000000 |
Yes |
No |
2510.55656960537 |
116.143699326325 |
1166.88064793723 |
41.6461834384112 |
| ENSMUSG00000023031 |
109901 |
Cela1 |
protein_coding |
Q91X79 |
FUNCTION: Acts upon elastin. {ECO:0000250}. |
Calcium;Disulfide bond;Glycoprotein;Hydrolase;Metal-binding;Protease;Reference proteome;Secreted;Serine protease;Signal;Zymogen |
|
|
mmu:109901; |
collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; metal ion binding [GO:0046872]; serine-type endopeptidase activity [GO:0004252]; digestive system development [GO:0055123]; elastin catabolic process [GO:0060309]; exocrine pancreas development [GO:0031017]; inflammatory response [GO:0006954]; multicellular organism growth [GO:0035264]; negative regulation of transcription by RNA polymerase II [GO:0000122]; pancreas morphogenesis [GO:0061113]; positive regulation of angiogenesis [GO:0045766]; positive regulation of transcription by RNA polymerase II [GO:0045944]; post-embryonic development [GO:0009791]; proteolysis [GO:0006508]; regulation of cell differentiation [GO:0045595]; regulation of cell population proliferation [GO:0042127]; tissue remodeling [GO:0048771]; transcription by RNA polymerase II [GO:0006366]; Wnt signaling pathway [GO:0016055] |
17901120_Glucocorticoid receptor-null lungs included Chi3l3 (11-fold, P |
ENSG00000139610 |
CELA1 |
62.681839 |
2.7761231475 |
1.473072 |
0.34944979 |
17.733098 |
0.00002541655947869558563852535049143455125886248424649238586425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001738415060527575780636999613903981298790313303470611572265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
95.1359545884731 |
23.2698386971159 |
34.269356773949 |
6.62187865143786 |
| ENSMUSG00000023169 |
105727 |
Slc38a1 |
protein_coding |
Q8K2P7 |
FUNCTION: Symporter that cotransports short-chain neutral amino acids and sodium ions from the extraccellular to the intracellular side of the cell membrane (PubMed:11325958, PubMed:17179157, PubMed:32668809, PubMed:27655909). The transport is elctrogenic, pH dependent and driven by the Na(+) electrochemical gradient (PubMed:11325958). Participates in the astroglia-derived glutamine transport into GABAergic interneurons for neurotransmitter GABA de novo synthesis (PubMed:31050701, PubMed:32668809). May also contributes to amino acid transport in placental trophoblast (By similarity). Regulates synaptic plasticity (PubMed:31050701). {ECO:0000250|UniProtKB:Q9H2H9, ECO:0000269|PubMed:11325958, ECO:0000269|PubMed:17179157, ECO:0000269|PubMed:27655909, ECO:0000269|PubMed:31050701, ECO:0000269|PubMed:32668809}. |
Alternative splicing;Amino-acid transport;Cell membrane;Disulfide bond;Glycoprotein;Ion transport;Membrane;Phosphoprotein;Reference proteome;Sodium;Sodium transport;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:105727; |
axon [GO:0030424]; basolateral plasma membrane [GO:0016323]; external side of apical plasma membrane [GO:0098591]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; alanine:sodium symporter activity [GO:0015655]; amino acid:sodium symporter activity [GO:0005283]; glycine:sodium symporter activity [GO:0015375]; L-amino acid transmembrane transporter activity [GO:0015179]; L-glutamine transmembrane transporter activity [GO:0015186]; neutral L-amino acid:sodium symporter activity [GO:0005295]; proline:sodium symporter activity [GO:0005298]; amino acid import [GO:0043090]; amino acid transmembrane transport [GO:0003333]; female pregnancy [GO:0007565]; gamma-aminobutyric acid biosynthetic process [GO:0009449]; glutamine metabolic process [GO:0006541]; glutamine transport [GO:0006868]; L-alpha-amino acid transmembrane transport [GO:1902475]; neutral amino acid transport [GO:0015804]; positive regulation of L-glutamine import across plasma membrane [GO:1901036]; regulation of synaptic plasticity [GO:0048167]; regulation of synaptic transmission, GABAergic [GO:0032228] |
21364602_In this study, the in vivo endothelial membrane localization of the sodium-dependent glutamine transporters Snat3 (Slc38a3) and Snat1 (Slc38a1) was investigated in the mouse brain microvasculature. 22841570_Inner hair cells express glutamine transporter SLC38A1. 23110224_GlnT would promote both proliferation and neuronal differentiation through a mechanism relevant to the upregulation of particular proneural genes in undifferentiated P19 cells. 25673846_We found that MeCP2 acts as a microglia-specific transcriptional repressor of 25957749_Overexpression of X-box binding protein-1 led to a marked increase in luciferase activity in P19 cells transfected with the Slc38a1 reporter plasmid. These results suggest that theanine accelerates cellular proliferation and subsequent neuronal specification through a mechanism relevant to upregulation of Slc38a1 gene in undifferentiated neural progenitor cells 27373508_Changes in transporter expression likely reflect different amino acid requirements during development. Findings include the differential expression of SNAT1 in the inner and outer cells of the compacted morula and nuclear localisation of SNAT2 in the trophectoderm and placental lineages. 28095293_Hence, pro-degenerative MAPK signaling functions upstream of SARM1 by limiting the levels of the essential axonal survival factor NMNAT2 to promote injury-dependent SARM1 activation. 29065407_The amino acid transporter gene Slc38a1 appears to be a novel gene responsive to hypoxic stress in a manner dependent on HIF-1alpha in brown adipocytes. In addition to in vitro studies, we showed that the Slc38a1 expression was enhanced in the brown adipose tissue of obesity model mice in vivo. 30612149_interacts with voltage-gated CaV2.2 calcium channels 31050701_The Glutamine Transporter Slc38a1 Regulates GABAergic Neurotransmission and Synaptic Plasticity. 31552299_These results demonstrate that SNAT1 promoted ischemic brain damage via mTOR-autophagy system. 32668809_Slc38a1 Conveys Astroglia-Derived Glutamine into GABAergic Interneurons for Neurotransmitter GABA Synthesis. |
ENSG00000111371 |
SLC38A1 |
166.636956 |
29.1337199752 |
4.864618 |
0.43097415 |
100.870632 |
0.00000000000000000000000981916537510446964201053945434248241005639110221142045525529720630814045634338071977254003286361694335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000004952500893827526509373948551546715922011966582131606288876956439848342483855958562344312667846679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
250.533919686497 |
75.9597316679456 |
8.59944833340791 |
2.03378520042318 |
| ENSMUSG00000023243 |
16529 |
Kcnk5 |
protein_coding |
Q9JK62 |
Human_homologues FUNCTION: pH-dependent, outwardly rectifying potassium channel (PubMed:9812978). Outward rectification is lost at high external K(+) concentrations (PubMed:9812978). {ECO:0000269|PubMed:9812978}. |
3D-structure;Ion channel;Ion transport;Membrane;Potassium;Potassium channel;Potassium transport;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:16529; |
plasma membrane [GO:0005886]; outward rectifier potassium channel activity [GO:0015271]; potassium channel activity [GO:0005267]; potassium ion leak channel activity [GO:0022841]; voltage-gated potassium channel activity [GO:0005249]; potassium ion export across plasma membrane [GO:0097623]; potassium ion import across plasma membrane [GO:1990573]; potassium ion transmembrane transport [GO:0071805]; potassium ion transport [GO:0006813]; regulation of resting membrane potential [GO:0060075]; stabilization of membrane potential [GO:0030322] |
12707131_results do not support the notion that KCNK5 potassium channel mediates the capacity of inhaled anesthetics to produce immobility 12860925_The TASK2 channel is expressed in kidney proximal cells and could be the swelling-activated K+ channel responsible for the cell volume regulation process during osmolyte absorptions in the proximal tubules. 15141089_consequences of the task2 gene inactivation in mice are reminiscent of the clinical manifestations seen in human proximal renal tubular acidosis syndrome 15328068_Sensitivity to intracellular Na(+) may be an additional potential regulatory mechanism of TASK-2 channels. 16239344_pH sensing in TASK2 channels is conferred by the combined action of several charged residues in the large extracellular M1-P1 loop. 17003225_Cl(-) influx will be the driving force for HCO(3)(-) exit through the activation of the Cl(-)/HCO(3)(-) exchanger. This efflux of HCO(3)(-) then alkalinizes pH(e), which in turn activates TASK2 channels 17947235_the role of the TASK2 channel in the regulation of AVD and apoptosis phenomenon. 20351106_intracellular pH, together with pH(o), is a critical determinant of TASK-2 activity and therefore of its physiological function 21283586_a cycle of protonation/deprotonation of pH(o)-sensing arginine 224 side chain gates the TASK-2 channel by electrostatically tuning the conformational stability of its selectivity filter. 21307343_The authors report for the first time the expression of TASK-2 in B cells and surmise that the upregulation of TASK-2 by B cell receptor ligation is associated with the apoptosis of immature B cells. 21575593_Kv1.3/TASK2 channels appear to be mainly responsible for regulatory volume decrease in murine T-lymphocytes. 23812165_data are compatible with the concept that TASK-2 channels are modulated by Gbetagamma subunits of heterotrimeric G protein; propose that this modulation is a novel way in which TASK-2 can be tuned to its physiological functions 24107938_alkaline-activated TASK-2 channels contribute to pH sensitivity in retrotrapezoid nucleus neurons 24247225_The strong physiological impairment of KCNK5 in Ehrlich cells after long-term hypotonic stimulation is predominantly due to down-regulation of the KCNK5 protein synthesis. 25305321_TASK-2 is upregulated under hypoxia in a HIF-1alpha-dependent manner in B cells. 26068853_The data identify GPR4 and TASK-2 as distinct, parallel, and essential central mediators of respiratory chemosensitivity. 26549439_KCNK5 channels are indispensable for the maintenance of hearing. KCNK5 is mainly found in outer sulcus Claudius', Boettcher's and root cells. 28358046_The apparent affinity of TASK-2 for membrane PI(4,5)P2 is low and it is conceivable that changing competition by intracellular polycationic compounds might mediate the previously reported sensitivity of TASK-2 to changes in cell volume. A decrease in PI(4,5)P2 by activation of Gq-PCRs is also probably responsible for TASK-2 inhibition. 31861667_Results suggest a mechanism for K2P5.1 up-regulation via HIF-1 in the CD4(+) T cells of the mouse model for inflammatory bowel. 32999458_Structural basis for pH gating of the two-pore domain K(+) channel TASK2. 33169379_A role for TASK2 channels in the human immunological synapse. |
ENSG00000164626 |
KCNK5 |
28.457879 |
0.1297175189 |
-2.946555 |
0.68006539 |
17.714397 |
0.00002566766376057673176239865009407026263943407684564590454101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001753666147645990239407887134603925005649216473102569580078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.09019716141042 |
2.97833600143377 |
46.9496889213737 |
15.4593901583473 |
| ENSMUSG00000023367 |
66058 |
Tmem176a |
protein_coding |
Q9DCS1 |
|
Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:66058; |
membrane [GO:0016020]; negative regulation of dendritic cell differentiation [GO:2001199] |
20501748_These data suggest that Tmem176B and Tmem176A associate to form multimers and restrain dendritic cell maturation. 27009467_Study characterized Tmem176a and b expression in RORgamma t+ lymphocytes at transcriptional and protein levels and present evidence that both genes exert a redundant ion channel function related to a colocalisation in close proximity to the Golgi apparatus. 34233909_Dendritic Cells Require TMEM176A/B Ion Channels for Optimal MHC Class II Antigen Presentation to Naive CD4(+) T Cells. |
ENSG00000002933 |
TMEM176A |
374.446718 |
4.9179712074 |
2.298063 |
0.21036372 |
112.018012 |
0.00000000000000000000000003540786920449595664707278279328012272873454718786389014045232065939529386700002078214311040937900543212890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000020904366810964433136202099391889852921746815293425013286281825548688917404405174238490872085094451904296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
626.000176139092 |
91.6027754708246 |
127.288296280115 |
14.4878857959813 |
| ENSMUSG00000023467 |
56734 |
Tulp2 |
protein_coding |
P46686 |
|
Alternative splicing;Cytoplasm;Phosphoprotein;Reference proteome;Secreted |
|
|
mmu:56734; |
cilium [GO:0005929]; cytoplasm [GO:0005737]; extracellular region [GO:0005576]; phosphoric diester hydrolase activity [GO:0008081]; protein-containing complex binding [GO:0044877]; protein localization to cilium [GO:0061512] |
|
ENSG00000104804 |
TULP2 |
37.047635 |
0.2148300591 |
-2.218732 |
0.54061428 |
16.337562 |
0.00005300288326156174175968899264788092295930255204439163208007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003377991288033926572421938860912860036478377878665924072265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.377068742729 |
5.13086571823781 |
62.2681425389897 |
16.534716225786 |
| ENSMUSG00000023505 |
14793 |
Cdca3 |
protein_coding |
Q99M54 |
FUNCTION: F-box-like protein which is required for entry into mitosis. Acts by participating in E3 ligase complexes that mediate the ubiquitination and degradation of WEE1 kinase at G2/M phase (By similarity). {ECO:0000250}. |
Alternative splicing;Cell cycle;Cell division;Cytoplasm;Mitosis;Phosphoprotein;Reference proteome;Ubl conjugation;Ubl conjugation pathway |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:14793; |
adherens junction [GO:0005912]; cytosol [GO:0005829]; cell cycle [GO:0007049]; cell division [GO:0051301]; protein ubiquitination [GO:0016567] |
15733861_a repressor element (cell-cycle-dependent element/cell cycle gene homology region) in the vicinity of the transcription start site, and mutations within this element diminished the cell-cycle-dependent transcriptional regulation of Tome-1 |
ENSG00000111665 |
CDCA3 |
2325.085798 |
2.7420775007 |
1.455269 |
0.08392206 |
292.343102 |
0.00000000000000000000000000000000000000000000000000000000000000001534696882161796835652744208914098465655824317866681118330158005588996313528133941033437372311139661684430115452259707912041739716505169669922828046575721994040485823829200739965017419308423995971680 |
0.0000000000000000000000000000000000000000000000000000000000000038322135916538507037230316149998344281213307745475888779434364238816515630924988217563804123791706724808845451090519735917014106614649097458730786836134530725511693205476149159949272871017456054688 |
Yes |
No |
3458.81455180568 |
201.313894062794 |
1261.38468038539 |
56.0896702999486 |
| ENSMUSG00000023940 |
72415 |
Sgo1 |
protein_coding |
Q9CXH7 |
FUNCTION: Plays a central role in chromosome cohesion during mitosis by preventing premature dissociation of cohesin complex from centromeres after prophase, when most of cohesin complex dissociates from chromosomes arms. May act by preventing phosphorylation of the STAG2 subunit of cohesin complex at the centromere, ensuring cohesin persistence at centromere until cohesin cleavage by ESPL1/separase at anaphase. Essential for proper chromosome segregation during mitosis and this function requires interaction with PPP2R1A. Its phosphorylated form is necessary for chromosome congression and for the proper attachment of spindle microtubule to the kinetochore. Necessary for kinetochore localization of PLK1 and CENPF. May play a role in the tension sensing mechanism of the spindle-assembly checkpoint by regulating PLK1 kinetochore affinity. Involved in centromeric enrichment of AUKRB in prometaphase. {ECO:0000250|UniProtKB:Q5FBB7, ECO:0000269|PubMed:18084284, ECO:0000269|PubMed:18331714}. |
Alternative splicing;Cell cycle;Cell division;Centromere;Chromosome;Chromosome partition;Coiled coil;Cytoplasm;Cytoskeleton;Kinetochore;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:72415; |
centrosome [GO:0005813]; chromosome, centromeric region [GO:0000775]; condensed chromosome, centromeric region [GO:0000779]; cytosol [GO:0005829]; kinetochore [GO:0000776]; nucleoplasm [GO:0005654]; spindle pole [GO:0000922]; kinase binding [GO:0019900]; attachment of spindle microtubules to kinetochore [GO:0008608]; cell division [GO:0051301]; centriole-centriole cohesion [GO:0010457]; chromosome segregation [GO:0007059]; homologous chromosome segregation [GO:0045143]; meiotic chromosome segregation [GO:0045132]; meiotic sister chromatid cohesion [GO:0051177]; mitotic sister chromatid cohesion, centromeric [GO:0071962]; mitotic sister chromatid segregation [GO:0000070] |
18949044_Prevention of premature separation of sister chromatids in meiosis I requires the retention of centromeric Sgo1, while normal separation of sister chromatids in meiosis II requires loss of centromeric Sgo1. 19716788_report structure and function of the PP2A-shugoshin interaction. 20124418_It seems that Sgo1 sets up the centromeric protection mechanism in G2, but that its Bub1-dependent localisation to centromeres during mitosis is not required to maintain cohesion. 22262168_Haploinsufficiency of SGO1 results in genomic instability manifested as missegregation of chromosomes and formation of extra centrosomal foci in both murine embryonic fibroblasts and adult bone marrow cells. 25740822_Sgo1(-/+)-mediated ME-CIN strongly promoted/progressed development of hepatocellular carcinoma in the presence of an initiator carcinogen, and it had a mild initiator effect by itself. 25773652_Study established the Sgo1 haploinsufficient mouse as a colon cancer model and identified antagonizing oncogenic and tumor-suppressing pathways that are differentially expressed and may be responsible for the different dynamics in the tumor development. 26847209_Data show that compound mutant spindle assembly checkpoint components BubR1 and Sgo1 embryonic fibroblasts (MEFs) grew at a much slower rate, and a small fraction of cells exhibited morphologies of senescent cells at early passages. 27539729_MYCN-promoted SGO1 transcription and SGO1 expression tended to be higher in MYCN- or MYC-overexpressing cancers. Together, these findings indicate that SGO1 plays a role in the DNA damage response in interphase. 28465207_Study shows Sgo1 expression in heart, gut, eye and other central nervous system tissue during development and post-development. It demonstrates Sgo1 localization in cytoplasmic region in nucleated cells in select organs. 28781233_Molecular chaperone SET-assisted eviction of linker histones and Shugoshins is a fundamental step in mammalian mitotic progression. 29943428_The Sgo1(-/+) mouse may serve as a novel tool for investigating mechanisms of spontaneous progression of Spontaneous late-onset Alzheimer's disease (LOAD) pathology, for early diagnosis markers, and for drug development. 31019073_The results show that shugoshin protects the centromeric synaptonemal complex in meiotic prophase and, in anaphase, promotes the proper segregation of partner chromosomes that are not linked by a crossover. 32857910_GSK3-ARC/Arg3.1 and GSK3-Wnt signaling axes trigger amyloid-beta accumulation and neuroinflammation in middle-aged Shugoshin 1 mice. |
ENSG00000129810 |
SGO1 |
653.954906 |
2.1370638073 |
1.095630 |
0.12460448 |
75.855824 |
0.00000000000000000305151056004199169841397670889157884983474078618547863789300933490267198067158460617065429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000001049223676084867201081260065245844533185539120163920490114151107263751327991485595703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
884.274596065395 |
76.4691160175841 |
413.780156230026 |
27.3619961252949 |
| ENSMUSG00000023951 |
22339 |
Vegfa |
protein_coding |
Q00731 |
FUNCTION: Growth factor active in angiogenesis, vasculogenesis and endothelial cell growth. Induces endothelial cell proliferation, promotes cell migration, inhibits apoptosis and induces permeabilization of blood vessels. Binds to the FLT1/VEGFR1 and KDR/VEGFR2 receptors, heparan sulfate and heparin. May play a role in increasing vascular permeability during lactation, when increased transport of molecules from the blood is required for efficient milk protein synthesis (By similarity). Binding to NRP1 receptor initiates a signaling pathway needed for motor neuron axon guidance and cell body migration, including for the caudal migration of facial motor neurons from rhombomere 4 to rhombomere 6 during embryonic development (PubMed:26503042). Also binds the DEAR/FBXW7-AS1 receptor (PubMed:16293765). {ECO:0000250|UniProtKB:P15692, ECO:0000269|PubMed:16293765, ECO:0000269|PubMed:26503042}. |
Alternative initiation;Alternative promoter usage;Alternative splicing;Angiogenesis;Cell membrane;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Growth factor;Heparin-binding;Membrane;Mitogen;Reference proteome;Secreted;Signal |
|
This gene is a member of the PDGF/VEGF growth factor family. It encodes a heparin-binding protein, which exists as a disulfide-linked homodimer. This growth factor induces proliferation and migration of vascular endothelial cells, and is essential for both physiological and pathological angiogenesis. Disruption of this gene in mice resulted in abnormal embryonic blood vessel formation. This gene is upregulated in many known tumors and its expression is correlated with tumor stage and progression. Alternatively spliced transcript variants encoding different isoforms have been found for this gene. There is also evidence for alternative translation initiation from upstream non-AUG (CUG) codons resulting in additional isoforms. A recent study showed that a C-terminally extended isoform is produced by use of an alternative in-frame translation termination codon via a stop codon readthrough mechanism, and that this isoform is antiangiogenic. Expression of some isoforms derived from the AUG start codon is regulated by a small upstream open reading frame, which is located within an internal ribosome entry site.[provided by RefSeq, Nov 2015]. |
mmu:22339; |
adherens junction [GO:0005912]; basement membrane [GO:0005604]; cell surface [GO:0009986]; cytoplasm [GO:0005737]; extracellular space [GO:0005615]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; secretory granule [GO:0030141]; VEGF-A complex [GO:1990150]; vesicle [GO:0031982]; chemoattractant activity [GO:0042056]; cytokine activity [GO:0005125]; fibronectin binding [GO:0001968]; growth factor activity [GO:0008083]; growth factor binding [GO:0019838]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; neuropilin binding [GO:0038191]; platelet-derived growth factor receptor binding [GO:0005161]; receptor ligand activity [GO:0048018]; vascular endothelial growth factor receptor 1 binding [GO:0043183]; vascular endothelial growth factor receptor 2 binding [GO:0043184]; vascular endothelial growth factor receptor binding [GO:0005172]; activation of protein kinase activity [GO:0032147]; angiogenesis [GO:0001525]; angiogenesis involved in coronary vascular morphogenesis [GO:0060978]; apoptotic process [GO:0006915]; artery morphogenesis [GO:0048844]; basophil chemotaxis [GO:0002575]; blood vessel development [GO:0001568]; blood vessel morphogenesis [GO:0048514]; blood vessel remodeling [GO:0001974]; bone trabecula formation [GO:0060346]; bone trabecula morphogenesis [GO:0061430]; branching involved in blood vessel morphogenesis [GO:0001569]; camera-type eye morphogenesis [GO:0048593]; cardiac muscle cell development [GO:0055013]; cardiac vascular smooth muscle cell development [GO:0060948]; cell chemotaxis [GO:0060326]; cell migration [GO:0016477]; cell migration involved in sprouting angiogenesis [GO:0002042]; cell population proliferation [GO:0008283]; cell-cell adhesion [GO:0098609]; cellular response to hypoxia [GO:0071456]; cellular response to vascular endothelial growth factor stimulus [GO:0035924]; cellular stress response to acid chemical [GO:0097533]; commissural neuron axon guidance [GO:0071679]; coronary artery morphogenesis [GO:0060982]; coronary vein morphogenesis [GO:0003169]; dopaminergic neuron differentiation [GO:0071542]; endothelial cell chemotaxis [GO:0035767]; endothelial cell migration [GO:0043542]; endothelial cell proliferation [GO:0001935]; epithelial cell differentiation [GO:0030855]; epithelial cell maturation [GO:0002070]; epithelial cell proliferation [GO:0050673]; eye photoreceptor cell development [GO:0042462]; heart morphogenesis [GO:0003007]; homeostasis of number of cells within a tissue [GO:0048873]; in utero embryonic development [GO:0001701]; induction of positive chemotaxis [GO:0050930]; kidney development [GO:0001822]; lactation [GO:0007595]; lung alveolus development [GO:0048286]; lung development [GO:0030324]; lung vasculature development [GO:0060426]; lymph vessel morphogenesis [GO:0036303]; lymphangiogenesis [GO:0001946]; macrophage differentiation [GO:0030225]; mammary gland alveolus development [GO:0060749]; mesoderm development [GO:0007498]; monocyte differentiation [GO:0030224]; motor neuron migration [GO:0097475]; mRNA stabilization [GO:0048255]; negative regulation of adherens junction organization [GO:1903392]; negative regulation of apoptotic process [GO:0043066]; negative regulation of blood-brain barrier permeability [GO:1905604]; negative regulation of bone resorption [GO:0045779]; negative regulation of cell-cell adhesion [GO:0022408]; negative regulation of cell-cell adhesion mediated by cadherin [GO:2000048]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043154]; negative regulation of epithelial to mesenchymal transition [GO:0010719]; negative regulation of establishment of endothelial barrier [GO:1903141]; negative regulation of extrinsic apoptotic signaling pathway [GO:2001237]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of gene expression [GO:0010629]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of neuron death [GO:1901215]; negative regulation of programmed cell death [GO:0043069]; negative regulation of transcription by RNA polymerase II [GO:0000122]; nervous system development [GO:0007399]; neuroblast proliferation [GO:0007405]; neuron development [GO:0048666]; outflow tract morphogenesis [GO:0003151]; ovarian follicle development [GO:0001541]; peptidyl-tyrosine phosphorylation [GO:0018108]; positive chemotaxis [GO:0050918]; positive regulation of angiogenesis [GO:0045766]; positive regulation of axon extension involved in axon guidance [GO:0048842]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:1903589]; positive regulation of branching involved in ureteric bud morphogenesis [GO:0090190]; positive regulation of cell adhesion [GO:0045785]; positive regulation of cell division [GO:0051781]; positive regulation of cell migration [GO:0030335]; positive regulation of cell migration by vascular endothelial growth factor signaling pathway [GO:0038089]; positive regulation of cell migration involved in sprouting angiogenesis [GO:0090050]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cell proliferation by VEGF-activated platelet derived growth factor receptor signaling pathway [GO:0038091]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of CREB transcription factor activity [GO:0032793]; positive regulation of DNA biosynthetic process [GO:2000573]; positive regulation of endothelial cell chemotaxis by VEGF-activated vascular endothelial growth factor receptor signaling pathway [GO:0038033]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of epithelial tube formation [GO:1905278]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of focal adhesion assembly [GO:0051894]; positive regulation of gene expression [GO:0010628]; positive regulation of histone deacetylase activity [GO:1901727]; positive regulation of lymphangiogenesis [GO:1901492]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of mast cell chemotaxis [GO:0060754]; positive regulation of neuroblast proliferation [GO:0002052]; positive regulation of osteoblast differentiation [GO:0045669]; positive regulation of p38MAPK cascade [GO:1900745]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of peptidyl-tyrosine autophosphorylation [GO:1900086]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of phosphorylation [GO:0042327]; positive regulation of positive chemotaxis [GO:0050927]; positive regulation of protein autophosphorylation [GO:0031954]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein kinase C signaling [GO:0090037]; positive regulation of protein kinase D signaling [GO:1903572]; positive regulation of protein localization to early endosome [GO:1902966]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein-containing complex assembly [GO:0031334]; positive regulation of receptor internalization [GO:0002092]; positive regulation of retinal ganglion cell axon guidance [GO:1902336]; positive regulation of signal transduction [GO:0009967]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of sprouting angiogenesis [GO:1903672]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of transcription from RNA polymerase II promoter in response to hypoxia [GO:0061419]; positive regulation of trophoblast cell migration [GO:1901165]; positive regulation of vascular endothelial growth factor signaling pathway [GO:1900748]; positive regulation of vascular permeability [GO:0043117]; post-embryonic camera-type eye development [GO:0031077]; primitive erythrocyte differentiation [GO:0060319]; regulation of cell shape [GO:0008360]; regulation of endothelial cell differentiation [GO:0045601]; regulation of growth [GO:0040008]; regulation of hematopoietic progenitor cell differentiation [GO:1901532]; regulation of nitric oxide mediated signal transduction [GO:0010749]; regulation of retinal ganglion cell axon guidance [GO:0090259]; regulation of transcription by RNA polymerase II [GO:0006357]; response to hypoxia [GO:0001666]; sprouting angiogenesis [GO:0002040]; surfactant homeostasis [GO:0043129]; T-helper 1 type immune response [GO:0042088]; tube formation [GO:0035148]; vascular endothelial growth factor receptor signaling pathway [GO:0048010]; vascular endothelial growth factor receptor-2 signaling pathway [GO:0036324]; vascular endothelial growth factor signaling pathway [GO:0038084]; vascular wound healing [GO:0061042]; vasculature development [GO:0001944]; vasodilation [GO:0042311]; VEGF-activated neuropilin signaling pathway [GO:0038190] |
8756616_VEGF gene expression is induced in response to hypoxia via binding of the transcriptional activator HIF-1. 11472838_VEGF expression upregulation requires Cbfa1/Runx2 during endochondral bone formation. 11549290_The expression of VEGF is upregulated by oncogenic ras through the activation of transcriptional enhancer HIF-1alpha. 11728162_Prostaglandin E(2) modulates vascular endothelial growth factor production in macrophages 11775025_Rac regulates vascular endothelial growth factor stimulated motility 11804779_VEGF188 is important for normal endochondral bone development, not only to mediate bone vascularization but also to allow normal differentiation of hypertrophic chondrocytes, osteoblasts, endothelial cells 11815711_essential for developmental angiogenesis and is also required for female reproductive functions and endochondral bone formation. 11897714_VEGF-A produced by osteoblasts in response to BMPs is not involved in osteoblast differentiation, but couples angiogenesis to bone formation 11907124_VEGF is one of the major determinants of toluene diisocyanate-induced asthma, and its inhibition may be a good therapeutic strategy. 11919510_hypoxia induces an increased expression of VEGF in the adult mouse brain. 11934854_VEGFA expression in the yolk sac mesoderm sheet was not sufficient for normal vascular and haematopoietic development. However, expression in the visceral endoderm was necessary and sufficient for normal development of the yolk sac vasculature. 11934855_role of VEGF in normal chondrocyte maturation and regulation of osteoblastic activity. 11959648_inducible VEGF expression is sensitive to copper and topical copper sulfate accelerated closure of excisional murine dermal wound 12048246_involvement in angiogenesis; angiopoietin-1 can offset VEGF-induced angiogenesis 12053176_inhibition impairs fetal lung maturation, whereas treatment with VEGF prevents fatal respiratory distress in premature mice. 12057925_in murine macrophages there is a novel pathway regulating VEGF production, involving the interaction of adenosine A(2A) receptor agonists through A(2A) receptors with LPS through the Tlr4 pathway 12086877_VEGF-A has a critical, nonredundant role in angiogenic switching and pancreatic beta cell carcinogenesis. 12087404_VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism 12112468_there are unique temporal and spatial expressions of VEGF-A isoforms during lung development which related factors have distinct functions in vascular and lymphatic patterning of the lung 12151311_Data show that expression of only VEGF 120 and not VEGF 164 and 188 isoforms impairs lung microvascular development and delays airspace maturation, indicating an essential role for heparin-binding VEGF isoforms in normal lung development. 12213803_VPF signaling is regulated by atrial natriuretic peptide, which preserves endothelial cell tight junction functional morphology 12372425_VEGF is regulated by nitric oxide from eNOS and nNOS in skeletal muscles in mice 12386279_VEGF is an essential molecule for glomerular structuring. VEGF is secreted from Bowman's capsule and stimulates endothelial or endothelial progenitor cells in a paracrine fashion to form the glomerulus. Review. 12393646_BCR/ABL induces expression of VEGF and its transcriptional activator HIF1A through a pathway involving phosphoinositide 3-kinase and the mammalian target of rapamycin in BaF3 cells. 12397601_endothelin-1 induced VEGF synthesis in mouse osteoblasts requires JNK 12444056_results show that vascular endothelial growth factor is the major mediator of estrogen-induced increase in uterine vascular permeability and vascular endothelial growth factor-induced edema is absolutely essential for implantation to take place 12461084_Vascular permeability factor/vascular endothelial growth factor induces lymphangiogenesis as well as angiogenesis. 12482580_VEGF has a role in placental hyperplasia of p57Kip2 null mouse embryos 12482858_VEGF is induced by p38 after stimulation by sodium arsenite 12537522_Proteinuria and vascular changes after renal irradiation: the role of reactive oxygen species (ROS) and vascular endothelial growth factor (Vegf) 12552636_Vegf modulates early heart valve formation. 12591918_Data suggest inhibition of AV cushion morphogenesis results from decreased vascular endothelial growth factor A expression and is mediated by platelet endothelial cell adhesion molecule-1 expression and reduced matrix metalloproteinase-2 expression. 12716475_heme oxygenase activity up-regulates VEGF production and augments the capability of endothelial cells to respond to exogenous stimulation 12810642_Analysis of intestinal polyps in mice heterozygous for the multiple intestinal neoplasia gene (Min/+) at 5 months revealed an increase & redistribution of VEGF-A near cells expressing nuclear beta-catenin with a corresponding increase in vessel density. 12810677_In vitro experiments showed that mVEGF directly up-regulated Ang-2, which is mediated via VEGF receptor-2/flk-1/KDR pathway, in cultured endothelial cells through transcriptional activation rather than the enhanced mRNA stability. 12847526_reduced expression of Vegfa causes amyotrophic lateral sclerosis-like motoneuron degeneration in Vegfa(delta/delta) mice 12858545_Role of VEGF-A in angiogenesis, arteriogenesis, lymphangiogenesis, and vascular malformations. 12890722_full-level VEGF-A activity is important for the development of the yolk sac mesenchyme and angiogenic sprouting of blood vessels 12912908_Transformation of VEGF-deficient adult fibroblasts with ras or neu oncogenes gives rise to highly tumorigenic and angiogenic fibrosarcomas. 14506249_RANKL and VEGF, in addition to their role in osteoclast differentiation and activation of resorption, are important components of the processes regulating osteoclast chemotaxis 14507649_Role for VEGF in myoblast migration and survival and novel autocrine role of VEGF in skeletal muscle repair during ischemia. 14530359_VEGF acts directly on T lymphocytes: elevated VEGF levels may contribute to aberrant MMP-9 secretion by mammary tumor bearers' T cells. 14633603_May be functionally implicated in progressive attrition of peritubular capillaries in tubular atrophy and interstitial fibrosis. Down-regulation correlates with loss of hypoxia-inducible factor-1 alpha, which occurs in increased tissue hypoxia. 14651929_Temporal and spatial regulation of VEGF-A controls vascular patterning in the embryonic lung. 14662796_VEGF expression in Leydig cells is regulated by gonadotropin via a cAMP-dependent mechanism, and the transcription factors Sp1 and Sp3 appear to be involved in the activation of the promoter 14699103_transcription of the vascular endothelial growth factor gene in macrophages is regulated by liver X receptors 14969389_demonstrated that the production of VEGF and RANKL stimulated by ovariectomy results in increased osteoclast formation in op/op mice 14983237_VEGF acts as a key regulator of brain angiogenesis and provides instructive cues for the correct spatial organization of the vasculature 14998923_Data suggest that the neural tube is the first structure identified as a midline signaling center for embryonic vascular pattern formation in higher vertebrates, and VEGFA is a necessary component of the neural tube vascular patterning signal. 15001532_VEGF and Ang1 participate in blood vessel survival and plasticity in adult life, but the Ang1 effect is not VEGF-mediated. 15003169_X-linked spinal and bulbar muscular atrophy involves altered expression of VEGF, with a role for VEGF as a survival factor in motor neuron disease. 15067085_Neutrophil-derived VEGF-A is a major mediator of C-X-C chemokine ligand 1/macrophage inflammatory protein-2-induced angiogenesis. 15072995_Low-level recombinant mouse VEGF expression can exert arterioprotective effects in the presence of high blood cholesterol in rabbits. 15073147_Results demonstrate that hypoxia-inducible factor 1 alpha (Hif1a) and vascular endothelial growth factor A are components of a key pathway to support chondrocyte survival during embryonic bone development. 15081307_VEGF, VEGFR-2 and adhesion molecules are upregulated by IL-1 beta on endothelial cells 15084712_VEGF is an essential survival factor for muscle capillarity. Insufficient VEGF-dependent signaling leads to apoptosis in mouse skeletal muscle. 15100155_findings reveal a central role of VEGF-A in promoting lymphatic enlargement, vascular hyperpermeability, and leukocyte recruitment, thereby leading to persistent chronic inflammation 15132990_VEGF-A promotes lymphatic vasculature formation via activation of VEGFR-2. 15143069_data suggest role for mitogen-activated protein kinase (MAPK) kinase 3 (MKK3)-p38alpha and p38delta MAPK pathway in mediating vascular endothelial growth factor VEGF164 isoform-specific stimulation by transforming growth factor-beta1 in mesangial cells 15161688_Hypoxia and hypoxia-inducible factor-1 induce VEGF in central nervous system radiation injury 15175342_VEGF is regulated by PAIP2 in tumor cells 15185205_VEGF expression may have a pivotal role in lung morphogenesis and is altered in the pathogenesis of nitrofen-induced congenital diaphragmatic hernia. 15193536_VEGF-A stained epithelial cells in choroid plexus 15196703_SAPK/JNK plays a part in PGE(1)-induced VEGF synthesis 15208295_Thus a transient decrease in pulmonary VEGF leads to increased alveolar and bronchial cell apoptosis, air space enlargement, and changes in lung elastic recoil (processes that are characteristic of emphysema) that persist for at least 8 wk. 15262905_VEGF is an essential mediator in Ang II-induced vascular inflammation and structural changes through its proinflammatory actions. 15326102_PAF stimulates corneal neovascularization by a receptor-mediated mechanism. Induction of VEGF expression and stimulation of vascular endothelial cell migration are initial events in PAF-promoted corneal angiogenesis. 15371263_VPF/VEGF-induced phosphorylation of VEGF receptor 2, focal adhesion kinase, and MAPK in the endothelial cells is strikingly increased in both dopamine-depleted and dopamine D(2) receptor knockout mice 15383400_Expression relating to HIF-1alpha metabolism in response to adriamycin-induced nephrosis. 15477421_Endogenous VEGF has an important physiologic role in vascular repair. 15488701_Leukemia inhibitory factor and vascular endothelial growth factor have roles in wound healing by increasing angiogenesis and matrix production 15501235_Ang-2 may target chemotactic responses to bFGF and VEGF in vivo 15501241_Ang1, in concert with VEGF, can act directly on mural cells or their precursors to facilitate their recruitment to new blood vessels 15504909_VEGF-mediated Src activity disrupts the endothelial barrier and potentiates tumor cell extravasation and metastasis 15504975_VEGF may have significant implications for podocyte dysfunction in diabetic glomerulopathy 15572520_Regulation of VEGF translation by angiotensin II represents a novel pathway of renal response to injury 15582599_v-erb-b2 erythroblastic leukemia viral oncogene homolog 2 overexpression led to an increase in endogenous VEGF mRNA as well as ErbB3 protein levels in HC11 cells 15601856_the VEGF-A protein concentrated in the anterior region of early embryos plays an important role in the guidance of VEGFR-positive cells from the posterior portion to the head region by interacting with VEGFR 15629128_Transforming growth factor-beta1 stimulation of VEGF production by fibroblasts is regulated by Smad3 but not by Smad2 signaling 15670761_Taken together, these results strongly suggest that PPAR-gamma ligands up-regulate FGF-2-stimulated VEGF release resulting from amplifying activation of SAPK/JNK in osteoblasts. 15673567_neuropilin 1-mediated positive feedback is a component of peripheral nerve-derived VEGF promotion of arterial differentiation 15705785_TPO induces VEGF expression by increasing the level of its primary transcription factor, HIF-1alpha, by enhancing its protein stability 15710418_VEGF is significantly increased (P |
ENSG00000112715 |
VEGFA |
6122.576856 |
0.4691223691 |
-1.091964 |
0.07263868 |
222.449033 |
0.00000000000000000000000000000000000000000000000002643627110676979254087760524476199475472093811469321150749023935049285835751189936669226404629312623043186471700860796720547419777491704451222176430746912956237792968750000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000044250250714100825248433860856147645734065613924099410254839580221852157445605637625629272225853263018659245429080018291011916486965560579847078770399093627929687500000000000000000000000000000000000000000000000000 |
Yes |
Yes |
3851.17302172954 |
193.985781964866 |
8209.31440429116 |
306.8711937854 |
| ENSMUSG00000023959 |
224796 |
Clic5 |
protein_coding |
Q8BXK9 |
FUNCTION: Required for normal hearing (By similarity). It is necessary for the formation of stereocilia in the inner ear and normal development of the organ of Corti. Can insert into membranes and form poorly selective ion channels that may also transport chloride ions. May play a role in the regulation of transepithelial ion absorption and secretion. Is required for the development and/or maintenance of the proper glomerular endothelial cell and podocyte architecture (PubMed:17021174). Plays a role in formation of the lens suture in the eye, which is important for normal optical properties of the lens (PubMed:29425878). {ECO:0000250|UniProtKB:Q9NZA1, ECO:0000269|PubMed:17021174, ECO:0000269|PubMed:29425878}. |
Cell membrane;Chloride;Chloride channel;Cytoplasm;Cytoskeleton;Golgi apparatus;Hearing;Ion channel;Ion transport;Membrane;Reference proteome;Sensory transduction;Transmembrane;Transmembrane helix;Transport;Vision;Voltage-gated channel |
|
|
mmu:224796; |
actin cytoskeleton [GO:0015629]; apical plasma membrane [GO:0016324]; cell cortex [GO:0005938]; chloride channel complex [GO:0034707]; cytoplasm [GO:0005737]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; microtubule organizing center [GO:0005815]; plasma membrane [GO:0005886]; stereocilium [GO:0032420]; stereocilium bundle [GO:0032421]; chloride channel activity [GO:0005254]; voltage-gated ion channel activity [GO:0005244]; auditory receptor cell stereocilium organization [GO:0060088]; chloride transport [GO:0006821]; diet induced thermogenesis [GO:0002024]; neuromuscular process controlling balance [GO:0050885]; protein localization [GO:0008104]; regulation of ion transmembrane transport [GO:0034765]; response to dietary excess [GO:0002021]; sensory perception of sound [GO:0007605]; visual perception [GO:0007601] |
17021174_CLIC5 associates with radixin in hair cell stereocilia and may help form or stabilize connections between the plasma membrane and the filamentous actin core. 20055760_CLIC5 disrupts proliferation of C2C12 cells and is involved in the differentiation of myoblasts by enhancing the protein expression level of MyHC, myogenin and desmin. 20335315_CLIC5A colocalizes with ezrin and podocalyxin in podocytes and is required for the development/maintenance of the proper glomerular endothelial cell and podocyte architecture. 20357015_Clic5 mutant mice are resistant to diet-induced obesity. 20664558_Study identifies CLIC5 as a new component that is enriched in and necessary for foot process integrity and podocyte function in vivo. 22889583_miR-96 and miR-182 regulate the in vivo expression of CLIC5 in mouse inner ear hair cells 24285636_Biochemical assays showed interaction of CLIC5 with ERM proteins, TPRN, and possibly myosin VI (MYO6). In addition, CLIC5 and RDX failed to localize normally in fused stereocilia of MYO6 mutant mice 25344252_The mechanism of CLIC5A action involves clustered plasma membrane phosphatidylinositol 4,5-bisphosphate accumulation, in turn facilitating ezrin activation and actin-dependent cell surface remodeling. 26924049_Augmented hypertension-induced glomerular capillary injury in mice lacking CLIC5 results from abrogation of Rac1-dependent Pak and ezrin activation, perhaps reducing the tensile strength of the podocyte actin cytoskeleton. 27582103_The findings indicate that CLIC4/CLIC5A-mediated ERM activation is required for maintenance of the glomerular capillary architecture. 28779175_Results indicate CLICs-dependent chloride efflux as an essential and proximal upstream event for NLRP3 activation. 29425878_These results suggest that KLPH is required for lens-specific expression of Clic5 and that Clic5 has an important role in the machinery that controls lens fiber cell extension and organization. |
ENSG00000112782 |
CLIC5 |
45.604712 |
0.2174640121 |
-2.201151 |
0.43024188 |
25.894318 |
0.00000036062909623816959459288353749550903870613183244131505489349365234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000034353360812381482635455634544463521251600468531250953674316406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.0552006148775 |
4.55662302367272 |
69.2307681911602 |
14.279055976482 |
| ENSMUSG00000023960 |
83965 |
Enpp5 |
protein_coding |
Q9EQG7 |
FUNCTION: Can hydrolyze NAD but cannot hydrolyze nucleotide di- and triphosphates (PubMed:28898552). Lacks lysopholipase D activity. May play a role in neuronal cell communication (By similarity). {ECO:0000250|UniProtKB:P84039, ECO:0000269|PubMed:28898552}. |
3D-structure;Glycoprotein;Hydrolase;Membrane;Metal-binding;Reference proteome;Secreted;Signal;Transmembrane;Transmembrane helix;Zinc |
|
|
mmu:83965; |
extracellular region [GO:0005576]; membrane [GO:0016020]; plasma membrane [GO:0005886]; dinucleotide phosphatase activity [GO:0004551]; NAD+ diphosphatase activity [GO:0000210]; zinc ion binding [GO:0008270]; cell communication [GO:0007154]; nucleotide catabolic process [GO:0009166] |
Human_homologues 28898552_The ENPP5 is able to cleave nicotinamide adenine dinucleotide (NAD), suggesting a potential role of this enzyme in NAD-based neurotransmission. |
ENSG00000112796 |
ENPP5 |
58.904796 |
0.1749526124 |
-2.514964 |
0.39174772 |
40.513541 |
0.00000000019525810637138800219987945054340560541161764263051736634224653244018554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000030042136123727089220073164664483872376976592022401746362447738647460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
17.5970118879444 |
4.83720850400007 |
100.581589750071 |
18.3484763879454 |
| ENSMUSG00000023961 |
224794 |
Enpp4 |
protein_coding |
Q8BTJ4 |
FUNCTION: Hydrolyzes extracellular Ap3A into AMP and ADP, and Ap4A into AMP and ATP. Ap3A and Ap4A are diadenosine polyphosphates thought to induce proliferation of vascular smooth muscle cells. Acts as a procoagulant, mediating platelet aggregation at the site of nascent thrombus via release of ADP from Ap3A and activation of ADP receptors (By similarity). {ECO:0000250}. |
Alternative splicing;Blood coagulation;Cell membrane;Disulfide bond;Glycoprotein;Hemostasis;Hydrolase;Membrane;Metal-binding;Reference proteome;Signal;Transmembrane;Transmembrane helix;Zinc |
|
|
mmu:224794; |
plasma membrane [GO:0005886]; bis(5'-adenosyl)-triphosphatase activity [GO:0047710]; metal ion binding [GO:0046872]; blood coagulation [GO:0007596]; positive regulation of blood coagulation [GO:0030194]; purine ribonucleoside catabolic process [GO:0046130] |
26823374_These results indicate that zinc remains inside the ENPP4 protein, a Bacillus Calmette-Guerin activated tumoricidal macrophage protein, throughout the simulation. |
ENSG00000001561 |
ENPP4 |
16.043783 |
0.1658670306 |
-2.591901 |
0.78092741 |
10.827444 |
0.00100006616917029688730578218525124611915089190006256103515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0046188623070958041358369250417581497458741068840026855468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.06114942487918 |
2.30722528463147 |
24.4843680504259 |
9.14444527847789 |
| ENSMUSG00000023992 |
83433 |
Trem2 |
protein_coding |
Q99NH8 |
FUNCTION: Forms a receptor signaling complex with TYROBP which mediates signaling and cell activation following ligand binding (PubMed:11241283). Acts as a receptor for amyloid-beta protein 42, a cleavage product of the amyloid-beta precursor protein APP, and mediates its uptake and degradation by microglia (PubMed:27477018, PubMed:29518356). Binding to amyloid-beta 42 mediates microglial activation, proliferation, migration, apoptosis and expression of pro-inflammatory cytokines, such as IL6R and CCL3, and the anti-inflammatory cytokine ARG1 (PubMed:27477018, PubMed:29518356). Acts as a receptor for lipoprotein particles such as LDL, VLDL, and HDL and for apolipoproteins such as APOA1, APOA2, APOB, APOE, APOE2, APOE3, APOE4, and CLU and enhances their uptake in microglia (PubMed:27477018). Binds phospholipids (preferably anionic lipids) such as phosphatidylserine, phosphatidylethanolamine, phosphatidylglycerol and sphingomyelin (By similarity). Regulates microglial proliferation by acting as an upstream regulator of the Wnt/beta-catenin signaling cascade (PubMed:28077724). Required for microglial phagocytosis of apoptotic neurons (PubMed:24990881). Also required for microglial activation and phagocytosis of myelin debris after neuronal injury and of neuronal synapses during synapse elimination in the developing brain (PubMed:15728241, PubMed:28592261, PubMed:29752066, PubMed:25631124). Regulates microglial chemotaxis and process outgrowth, and also the microglial response to oxidative stress and lipopolysaccharide (PubMed:30232263, PubMed:29663649, PubMed:28483841, PubMed:29859094). It suppresses PI3K and NF-kappa-B signaling in response to lipopolysaccharide; thus promoting phagocytosis, suppressing pro-inflammatory cytokine and nitric oxide production, inhibiting apoptosis and increasing expression of IL10 and TGFB (PubMed:29663649). During oxidative stress, it promotes anti-apoptotic NF-kappa-B signaling and ERK signaling (PubMed:28592261). Plays a role in microglial MTOR activation and metabolism (PubMed:28802038). Regulates age-related changes in microglial numbers (PubMed:30548312, PubMed:29752066, PubMed:25631124). Triggers activation of the immune responses in macrophages and dendritic cells (By similarity). Mediates cytokine-induced formation of multinucleated giant cells which are formed by the fusion of macrophages (PubMed:18957693). In dendritic cells, it mediates up-regulation of chemokine receptor CCR7 and dendritic cell maturation and survival (By similarity). Involved in the positive regulation of osteoclast differentiation (PubMed:16418779). {ECO:0000250|UniProtKB:Q9NZC2, ECO:0000269|PubMed:11241283, ECO:0000269|PubMed:15728241, ECO:0000269|PubMed:16418779, ECO:0000269|PubMed:18957693, ECO:0000269|PubMed:24990881, ECO:0000269|PubMed:25631124, ECO:0000269|PubMed:27477018, ECO:0000269|PubMed:28077724, ECO:0000269|PubMed:28483841, ECO:0000269|PubMed:28592261, ECO:0000269|PubMed:28802038, ECO:0000269|PubMed:29518356, ECO:0000269|PubMed:29663649, ECO:0000269|PubMed:29752066, ECO:0000269|PubMed:29859094, ECO:0000269|PubMed:30232263, ECO:0000269|PubMed:30548312}. |
Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Lipid-binding;Membrane;Receptor;Reference proteome;Secreted;Signal;Transmembrane;Transmembrane helix |
|
The protein encoded by this gene is part of the immunoglobulin and lectin-like superfamily and functions as part of the innate immune system. This gene forms part of a cluster of genes on mouse chromosome 17 thought to be involved in innate immunity. This protein associates with the adaptor protein Dap-12 and recruits several factors, such as kinases and phospholipase C-gamma, to form a receptor signaling complex that activates myeloid cells, including dendritic cells and microglia. In humans homozygous loss-of-function mutations in this gene cause Nasu-Hakola disease and mutations in this gene may be risk factors to the development of Alzheimer's disease. In mouse mutations of this gene serve as a pathophysiological model for polycystic lipomembranous osteodysplasia with sclerosing leukoencephalopathy (Nasu-Hakola disease) and for inflammatory bowel disease. Alternative splicing results in multiple transcript variants that encode different protein isoforms. [provided by RefSeq, Jan 2013]. |
mmu:83433; |
extracellular region [GO:0005576]; membrane [GO:0016020]; plasma membrane [GO:0005886]; plasma membrane raft [GO:0044853]; amyloid-beta binding [GO:0001540]; apolipoprotein A-I binding [GO:0034186]; apolipoprotein binding [GO:0034185]; high-density lipoprotein particle binding [GO:0008035]; kinase activator activity [GO:0019209]; lipopolysaccharide binding [GO:0001530]; lipoprotein particle binding [GO:0071813]; lipoteichoic acid binding [GO:0070891]; low-density lipoprotein particle binding [GO:0030169]; peptidoglycan binding [GO:0042834]; phosphatidylethanolamine binding [GO:0008429]; phosphatidylserine binding [GO:0001786]; phospholipid binding [GO:0005543]; protein tyrosine kinase binding [GO:1990782]; protein-containing complex binding [GO:0044877]; scaffold protein binding [GO:0097110]; signaling receptor activity [GO:0038023]; sulfatide binding [GO:0120146]; transmembrane signaling receptor activity [GO:0004888]; very-low-density lipoprotein particle binding [GO:0034189]; amyloid-beta clearance [GO:0097242]; amyloid-beta clearance by cellular catabolic process [GO:0150094]; apoptotic cell clearance [GO:0043277]; astrocyte activation [GO:0048143]; cellular response to amyloid-beta [GO:1904646]; cellular response to glucose stimulus [GO:0071333]; cellular response to hypoxia [GO:0071456]; cellular response to lipid [GO:0071396]; cellular response to lipoteichoic acid [GO:0071223]; cellular response to oxidised low-density lipoprotein particle stimulus [GO:0140052]; cellular response to peptidoglycan [GO:0071224]; complement-mediated synapse pruning [GO:0150062]; CXCL12-activated CXCR4 signaling pathway [GO:0038160]; defense response to bacterium [GO:0042742]; defense response to Gram-negative bacterium [GO:0050829]; dendritic cell differentiation [GO:0097028]; dendritic spine maintenance [GO:0097062]; detection of lipopolysaccharide [GO:0032497]; detection of lipoteichoic acid [GO:0070392]; detection of peptidoglycan [GO:0032499]; excitatory synapse pruning [GO:1905805]; import into cell [GO:0098657]; innate immune response [GO:0045087]; lipid homeostasis [GO:0055088]; lipopolysaccharide-mediated signaling pathway [GO:0031663]; memory [GO:0007613]; microglial cell activation [GO:0001774]; microglial cell activation involved in immune response [GO:0002282]; microglial cell proliferation [GO:0061518]; negative regulation of amyloid fibril formation [GO:1905907]; negative regulation of apoptotic process [GO:0043066]; negative regulation of astrocyte activation [GO:0061889]; negative regulation of autophagic cell death [GO:1904093]; negative regulation of autophagy [GO:0010507]; negative regulation of cell activation [GO:0050866]; negative regulation of cholesterol storage [GO:0010887]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; negative regulation of fat cell proliferation [GO:0070345]; negative regulation of glial cell apoptotic process [GO:0034351]; negative regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043124]; negative regulation of inflammatory response to antigenic stimulus [GO:0002862]; negative regulation of interleukin-1 beta production [GO:0032691]; negative regulation of macrophage colony-stimulating factor signaling pathway [GO:1902227]; negative regulation of neuroinflammatory response [GO:0150079]; negative regulation of NLRP3 inflammasome complex assembly [GO:1900226]; negative regulation of p38MAPK cascade [GO:1903753]; negative regulation of phosphatidylinositol 3-kinase signaling [GO:0014067]; negative regulation of sequestering of triglyceride [GO:0010891]; negative regulation of toll-like receptor 2 signaling pathway [GO:0034136]; negative regulation of toll-like receptor 4 signaling pathway [GO:0034144]; negative regulation of tumor necrosis factor production [GO:0032720]; neuroinflammatory response [GO:0150076]; osteoclast differentiation [GO:0030316]; phagocytosis, engulfment [GO:0006911]; phagocytosis, recognition [GO:0006910]; positive regulation of amyloid-beta clearance [GO:1900223]; positive regulation of antigen processing and presentation of peptide antigen via MHC class II [GO:0002588]; positive regulation of ATP biosynthetic process [GO:2001171]; positive regulation of C-C chemokine receptor CCR7 signaling pathway [GO:1903082]; positive regulation of calcium-mediated signaling [GO:0050850]; positive regulation of CAMKK-AMPK signaling cascade [GO:1905291]; positive regulation of CD40 signaling pathway [GO:2000350]; positive regulation of chemotaxis [GO:0050921]; positive regulation of cholesterol efflux [GO:0010875]; positive regulation of complement activation, classical pathway [GO:0045960]; positive regulation of engulfment of apoptotic cell [GO:1901076]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of establishment of protein localization [GO:1904951]; positive regulation of gene expression [GO:0010628]; positive regulation of high-density lipoprotein particle clearance [GO:0010983]; positive regulation of interleukin-10 production [GO:0032733]; positive regulation of inward rectifier potassium channel activity [GO:1901980]; positive regulation of kinase activity [GO:0033674]; positive regulation of low-density lipoprotein particle clearance [GO:1905581]; positive regulation of macrophage fusion [GO:0034241]; positive regulation of microglial cell activation [GO:1903980]; positive regulation of microglial cell migration [GO:1904141]; positive regulation of mitochondrion organization [GO:0010822]; positive regulation of neuroinflammatory response [GO:0150078]; positive regulation of NIK/NF-kappaB signaling [GO:1901224]; positive regulation of osteoclast differentiation [GO:0045672]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phagocytosis [GO:0050766]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of proteasomal protein catabolic process [GO:1901800]; positive regulation of protein localization to plasma membrane [GO:1903078]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein secretion [GO:0050714]; positive regulation of synapse pruning [GO:1905808]; positive regulation of TOR signaling [GO:0032008]; pyroptosis [GO:0070269]; regulation of cytokine production involved in inflammatory response [GO:1900015]; regulation of gene expression [GO:0010468]; regulation of hippocampal neuron apoptotic process [GO:0110089]; regulation of innate immune response [GO:0045088]; regulation of interleukin-6 production [GO:0032675]; regulation of intracellular signal transduction [GO:1902531]; regulation of lipid metabolic process [GO:0019216]; regulation of macrophage inflammatory protein 1 alpha production [GO:0071640]; regulation of oxidative stress-induced neuron intrinsic apoptotic signaling pathway [GO:1903376]; regulation of peptidyl-tyrosine phosphorylation [GO:0050730]; regulation of plasma membrane bounded cell projection organization [GO:0120035]; regulation of resting membrane potential [GO:0060075]; regulation of toll-like receptor 6 signaling pathway [GO:0034151]; regulation of TOR signaling [GO:0032006]; respiratory burst after phagocytosis [GO:0045728]; response to axon injury [GO:0048678]; response to ischemia [GO:0002931]; social behavior [GO:0035176] |
12847223_TREM-2a and TREM-2b binding to both Gram-positive and Gram-negative bacteria is disrupted by anionic bacterial products or other anionic carbohydrates and suggests that TREM-2 recognizes pathogens via charged carbohydrates expressed on pathogen surface. 15128777_IL-4-mediated dendritic cell-activated natural killer cell activity is significantly blocked by soluble triggering receptor expressed on myeloid cell 2 (TREM2) Fc molecules in vitro. 16046394_OSCAR but not TREM-2 is involved in the positive feedback loop of the immunoreceptor-NFATc1 pathway during osteoclastogenesis 16418779_TREM2-DAP12 signals regulate both osteoclast formation and function. 16675145_In microglial cells Trem2 is distributed intracellularly in the Golgi complex and in a population of exocytic vesicles distinct from endosomes and lysosomes, where is continuously translocated to and recycled from the cell surface. 16887962_Interaction of TREM-2 and its ligand results in an inhibitory signal that can reduce the inflammatory response. 16951310_TREM-2 is expressed on newly differentiated and alternatively activated macrophages and functions to restrain macrophage activation. 18391000_Salmonella typhimurium induces ROS through a pathway that requires both triggering receptor expressed on myeloid cells 2 (TREM2) and DAP12. 18404378_Trem2/DAP12 expression is detected in embryonic day 14 CNS mRNA. Trem2/DAP12 positive microglia are found in very close apposition with CNP+ oligodendrocytes prior to myelination (post-natal day 1). 18957693_signaling through DAP12, TREM-2, and the downstream protein tyrosine kinase Syk is required for the cytokine-induced formation of giant cells 19109436_Trem2 signaling is an important pathway to promote healing of wounds in the colon where stem cell replacement is necessary 19120482_contributes to the immune response against Salmonella in a mouse model 19302484_TREM2 conferred phagocytic activity of neuronal cells demonstrating that TREM2 is both required and sufficient for competent uptake of apoptotic neuronal cells 19948717_LAB is a critical, LAT-independent regulator of TREM-2 signaling and macrophage development capable of controlling subsequent inflammatory responses 20421649_These findings indicate that DAP12, possibly through association with TREM2, contributes to alveolar macrophage chemotaxis and recruitment to the lung and may mediate macrophage accumulation in lung diseases such as emphysema. 20484116_demonstrate a previously uncharacterized interaction of SHIP1 with DAP12 that limits TREM2- and DAP12-dependent signaling and identify a mechanism through which SHIP1 regulates key ITAM-containing receptors by blocking the binding and activation of PI3K 21956652_TREM-2 receptor transduces inhibitory signals due to recognition of an endogenous ligand. 22312126_TREM2 regulates the rate of osteoclastogenesis 22761415_TLT-1s, alternative transcripts of triggering receptor expressed on myeloid cell-like transcript-1 (TLT-1), Inhibits the triggering receptor expressed on myeloid cell-2 (TREM-2)-mediated signaling pathway during osteoclastogenesis. 23301011_TREM2 appears to sustain a distinct inflammatory response after stroke in a mouse model 23462268_an epigenetic mechanism involving an NF-small ka, CyrillicB-mediated, miRNA-34a-regulated downregulation of TREM2 expression may shape innate immune and phagocytic responses 23721075_Trem-2 plays an important role in the host defense response to sepsis by enhancing bacterial clearance. 23778113_Results suggest that an epigenetic mechanism involving an aluminum-triggered, NF-kB-sensitive, miRNA-34a-mediated down-regulation of triggering receptor expressed in myeloid cells 2 (TREM2) expression may impair phagocytic responses. 23977213_TREM2 protein is expressed only in microglia/macrophages and is developmentally downregulated in a region-dependent manner. 24218563_TREM2 governs Kupffer cell activation and explains belr1 genetic resistance to malaria liver stage infection. 24368090_It may play a protective role against aging-related neuroinflammation and cognitive impairment. 24383713_TREM-2-mediated bacterial killing is dependent on the activation of PI3K/Akt signaling. 24893973_These data suggest that TREM2 is important for the microglial response to Amyloid-beta deposition 24916365_TREM-2 has a protective effect on inflammatory response of endotoxin-induced acute lung injury in mice. 24945405_Data (including data from studies using knockout mice) suggest that TREM2 is expressed on alveolar macrophages and lung mucosa and plays role innate immunity during pneumococcal pneumonia; knockout of TREM2 increases phagocytosis and survival. 25114293_TREM2 promotes adipogenesis and diet-induced obesity by upregulating adipogenic regulators in conjunction with inhibiting the Wnt10b/beta-catenin signaling pathway. 25477281_Upon infection with Escherichia coli, the otherwise beneficial effect of an exaggerated early immune response in TREM-2(-/-) animals was counteracted by a 50% reduction in bacterial phagocytosis. 25563793_These findings provide a novel interpretation of Brucella intracellular growth through inhibition of NO production produced by TREM-2-mediated activated macrophages. 25631124_These findings place TREM2 as a key regulator of microglia activation in vivo in response to tissue damage 25716838_TREM2 knockdown reduced microglial activation, decreased phagocytosis of injured neurons and worsened stroke damage. 25728668_TREM2 detects damage-associated lipid patterns associated with neurodegeneration, sustaining the microglial response to beta-amyloid accumulation. 25732305_important role for TREM2 in inflammatory macrophages and neuroinflammation 25816748_TREM2 is involved in prion-induced microglial activation but does not noticeably modulate the pathogenesis of experimental prion infections. 25860078_TREM-1/TREM-3 macrophage expression improved host defense against Klebsiella-derived pneumosepsis, whereas TREM-2 did not have a role. 25893602_Corpus callosum microglia normally expand with age, but aged Trem2(-/-) mice had fewer microglia with an abnormal morphology which failed to amplify transcripts indicative of activation, phagocytosis, and lipid catabolism in response to myelin damage. 25897174_viral replication increases lung macrophage levels of intracellular and cell surface TREM-2, and this action prevents macrophage apoptosis 25957402_results support a role of DAP12 in stabilizing TREM2-CTF, thereby protecting against excessive pro-inflammatory responses. 26364736_TREM2 attenuates tau kinase activity through restriction of neuroinflammation, and thus protects against tau pathology. 26802771_Study found that TREM2 was upregulated in the brain of P301S mice, an animal model of tau pathology, during disease progression and showed that TREM2 overexpression rescued spatial cognitive impairments and ameliorated neuropathologies including neuronal and synaptic loss as well as tau hyperphosphorylation. 26976047_TREM2 deficiency influences both acute and chronic responses to traumatic brain injury, with altered macrophage response early on and improved functional outcome at later time points. 27091843_TREM2 protects from Alzheimer's disease by enabling microglia to surround and alter Abeta plaque structure, thereby limiting neuritic damage. 27143430_TREM2 and TREML2 play opposite roles in microglia activation. 27196974_TREM2 deficiency may disrupt the formation of a neuroprotective microglia barrier that regulates amyloid compaction and insulation 27253382_This study showed that TREM-2 deficiency restricts the inflammatory response, thereby decreasing organ damage and mortality. 27402340_Loss of TREM2 reduces the ability of microglia to engulf amyloid beta-peptide. 27662313_Results suggest that TREM2 plays a critical role in inflammation and neuronal cell survival and in neurogenesis. Study showed that TREM2 is a soluble protein transported by macrophages through ventricle walls and choroid plexus, and then enters the brain parenchyma via radial glial cells. TREM2 protein is essential for neuroplasticity and myelination. A lack of TREM2 protein may accelerate aging. 27717139_Our study suggests that Vps35/retromer is responsible for recycling of Trem2 in the regulation of microglial function such as proinflammatory responses, whereas R47H mutation impairs Trem2 trafficking, which might contribute to Alzheimer disease. 27798193_Recent studies have revealed that activated microglia in the spinal dorsal horn exacerbate neuropathic pain, which has suggested that suppression of microglial activity should be considered as a therapeutic target. However, only a few molecules have been identified as regulators of microglial activity. In this study, we focused on a receptor complex of TREM2 and DAP12, both of which are expressed by microglia and have bee 28077724_This study demonstrate a critical role of TREM2-mediated Wnt/beta-catenin pathway in microglial viability and suggest that modulating this pathway therapeutically may help to combat the impaired microglial survival. 28100745_TREM2 deficiency has opposing effects on Alzheimer's disease-related pathologies at early and late stages of disease progression. 28189343_A central role of TREM2 is in the regulation of microglia response to acute neurotoxic insults. 28209725_sTREM2 plays a crucial role in regulating microglial cell survival and inflammatory responses. 28398927_Triggering receptor expressed on myeloid cells 2 (TREM2) is an immunoglobulin-like receptor of the TREM family and is expressed on activated macrophages, immature dendritic cells, osteoclasts, and microglia. 28490631_flow cytometry analyses indicated significantly lower surface expression of T66M TREM2 variant than wild type or other TREM2 variants 28559417_The authors demonstrate that a TREM2 loss-of-function mutation causes brain-wide metabolic alterations pointing toward a possible function of microglia in regulating brain glucose metabolism. 28592261_TREM2 protects against cerebral ischemia/reperfusion injury through the aspect of post-ischemic inflammatory response and neuronal apoptosis. 28802038_Microglia in Alzheimer's disease (AD) patients carrying TREM2 risk variants and TREM2-deficient mice with AD-like pathology have abundant autophagic vesicles, as do TREM2-deficient macrophages under growth-factor limitation or endoplasmic reticulum (ER) stress. Study concludes that TREM2 enables microglial responses during AD by sustaining cellular energetic and biosynthetic metabolism. 28894284_The present study provides evidence that TREM2 is required for preventing loss of memory and learning in Alzheimer's disease by regulating C/EBPalpha-dependent CD36 expression and the consequent Abeta phagocytosis. 28928485_analyses showed that these TREM-2(+) cells expressed high levels of CCR-7 and CD86 suggesting a potential role of TREM-2 in mediating maturation and migration of DC subsets in allergic airway inflammation. 28978423_This article suggests a potential explanation of why TREM2-deficient microglia are unable to respond to neurotoxic plaques in the Alzheimer's disease brain and highlight a further need to understand microglial biology. 29040522_Transcriptome analysis revealed that our Trem2-/- mouse line (Velocigene allele) results in exaggerated Treml1 upregulation. In contrast, aberrantly high Treml1 expression was absent in the Trem2 knockout line generated by the Colonna lab and the Jackson Labs CRISPR/Cas9 Trem2 knockout line. 29073081_These studies of the role of TREM2 in neuroinflammation and neurodegeneration suggest that impairing microglial TREM2 signaling reduces pure tauopathy. 29407460_The present study provide novel evidence that overexpression of TREM2 protects against MPTP-induced PD progression via modulation of microglial function through inhibiting TRAF6/TLR4-mediated activation of the MAPK and NF-kappaB signaling pathways. 29518356_TREM2 is a microglial Amyloid Beta receptor, transducing physiological and Alzheimer's disease pathological effects associated with Amyloid Beta. 29518357_Elevated TREM2 gene dosage led to improved memory performance in Alzheimer's disease models. 29587871_Our data establish a critical link between oAbeta1-42, a major pathological component of Alzheimer's disease and TREM2 29599291_TREM2 mutation is associated with Alzheimer's disease. 29611543_Data indicate a novel role for PS1 in regulating TREM2 intracellular trafficking and pathophysiological function. 29621548_TREM2 plays a crucial role in altering the proinflammatory M1 microglia to M2 phenotype and has beneficial effects in the immune pathogenesis of Parkinson's disease. 29663649_Authors concluded that TREM2 inhibited neuroinflammation by down-regulating PI3 K/AKT and NF-kB signaling in BV2 microglia. 29689568_Overexpression of TREM2 downregulated the levels of IL-1beta, ameliorated T396 expression, inhibited the activity of GSK-3beta, and improved sickness behavior. Increased Arg1 expression and a high level of synaptophysin were also observed in the transgenic mice following TREM2 overexpression. 29752066_Triggering receptor expressed on myeloid cells 2 (TREM2) knockout(-/-) mice displayed repetitive behavior and altered sociability. 29859094_These data suggest that the Alzheimer's disease-associated TREM2 R47H variant increases risk for Alzheimer's disease by conferring a loss of TREM2 function and enhancing neuritic dystrophy around plaques. 29906661_Study shows that loss of Trem2 in microglia leads to widespread disruption of cell coexpression networks in mouse brain. 30185230_Trem2 R47H variant activates a cryptic splice site that generates miss-spliced transcripts leading to Trem2 haploinsufficiency only in mice but not in humans 30232263_whereas complete TREM2 deficiency protected against tau-mediated microglial activation and atrophy, TREM2 haploinsufficiency elevated expression of proinflammatory markers and exacerbated atrophy at a late stage of disease. The differential effects of partial and complete loss of TREM2 on microglial function and tau pathology 30523715_Mice were subjected to experimental stroke, and neurological function and infarct volume were assessed. Mice with intact TREM2 in brain microglia showed better neurological recovery and reduced infarct volumes, compared with mice lacking microglial TREM2. 30548312_microglial TREM2 contributes to age-related microglial changes, phagocytic oxidative burst, and loss of neurons with possible detrimental effects during physiological aging. 30617257_We demonstrate that amyloid plaque seeding is increased in the absence of functional Trem2. Increased seeding is accompanied by decreased microglial clustering around newly seeded plaques and reduced plaque-associated apolipoprotein E (ApoE). 30684126_Study shows that up-regulation of trem2 inhibits hippocampal neuronal apoptosis and alleviates oxidative stress in epilepsy via the PI3K/Akt pathway in Mice. 30756214_an imbalance of TLR4/TREM2 may be a potential link between AD and systemic inflammation 30790627_findings indicate that the level of TREM2 may affect the microglia response to pathological process induced by amyloid beta. 30911003_Soluble TREM2 ameliorates pathological phenotypes by modulating microglial functions in an Alzheimer's disease model. 31028072_Downregulating the expression of TREM2 can inhibit the release of inflammatory factors from lipopolysaccharide-stimulated microglia. 31122931_Opposite microglial activation stages upon loss of PGRN or TREM2 result in reduced cerebral glucose metabolism. 31219699_TREM2 up-regulation induces synaptic impairment at early stage and prevents amyloidosis at late stage in APP/PS1 mice. 31235932_germline knockout of Trem2 or the TREM2(R47H) variant reduces microgliosis around amyloid-beta plaques and facilitates the seeding and spreading of neuritic plaque tau aggregates 31265185_Loss of TREM2 impairs normal neurodevelopment, resulting in reduced synapse number across the cortex and hippocampus in 1-month-old mice. TREM2 was required for microglia to limit synaptic engulfment by astrocytes during development. Changes were largely normalized later in adulthood, but high fat diet administration was sufficient to reinitiate TREM2-dependent modulation of synapse loss. 31283973_Efficacy and mechanism of cGAMP to suppress Alzheimer's disease by elevating TREM2. 31301936_Knocking out TREM2 in 5xFAD mice exacerbated Abeta pathology and neurodegeneration but reduced Iba1(+) cell numbers, all of which could not be rescued by additional CD33 knockout. Differential gene expression in 5xFAD;CD33(-/-) microglia depended on the presence of TREM2, suggesting TREM2 acts downstream of CD33. 31370707_TREM2 deficiency aggravates alpha-synuclein-induced neurodegeneration and neuroinflammation in Parkinson's disease models. 31477129_Trigger receptor expressed on myeloid cells 2 may function as a feedback mechanism to curb obesity-induced insulin resistance via regulating adipose tissue remodeling. 31590042_Curcumin signi fi cantly alleviates LPS-induced in fl ammation by regulating microglial (M1/M2) polarization by reducing the imbalance of TREM2 and TLR4 31902528_studies identify TREM2 as a key transcriptional regulator of cholesterol transport and metabolism under conditions of chronic myelin phagocytic activity, as TREM2 loss of function causes pathogenic lipid accumulation in microglia. 31932797_a TREM2 requirement in both mouse and human Alzheimer's disease, is reported. 31973822_Caldecrin inhibits lipopolysaccharide-induced pro-inflammatory cytokines and M1 macrophage polarization through the immunoreceptor triggering receptor expressed in myeloid cells-2. 31980586_Trem2 Deletion Reduces Late-Stage Amyloid Plaque Accumulation, Elevates the Abeta42:Abeta40 Ratio, and Exacerbates Axonal Dystrophy and Dendritic Spine Loss in the PS2APP Alzheimer's Mouse Model. 32198705_Pinitol Prevents Lipopolysaccharide (LPS)-Induced Inflammatory Responses in BV2 Microglia Mediated by TREM2. 32412103_Triggering receptor expressed on myeloid cells 2 activation downregulates toll-like receptor 4 expression and ameliorates cognitive impairment in the Abeta1-42 -induced Alzheimer's disease mouse model. 32466767_TREM2 activation attenuates neuroinflammation and neuronal apoptosis via PI3K/Akt pathway after intracerebral hemorrhage in mice. 32544086_Impact of TREM2R47H variant on tau pathology-induced gliosis and neurodegeneration. 32617097_TREM2 Overexpression Attenuates Cognitive Deficits in Experimental Models of Vascular Dementia. 32703241_Trem2 deficiency differentially affects phenotype and transcriptome of human APOE3 and APOE4 mice. 32711525_A1 reactive astrocytes and a loss of TREM2 are associated with an early stage of pathology in a mouse model of cerebral amyloid angiopathy. 32772264_TREM2 activation on microglia promotes myelin debris clearance and remyelination in a model of multiple sclerosis. 32783915_Coupled scRNA-Seq and Intracellular Protein Activity Reveal an Immunosuppressive Role of TREM2 in Cancer. 32783918_TREM2 Modulation Remodels the Tumor Myeloid Landscape Enhancing Anti-PD-1 Immunotherapy. 32790063_Higher CSF sTREM2 and microglia activation are associated with slower rates of beta-amyloid accumulation. 32826884_TMEM59 interacts with TREM2 and modulates TREM2-dependent microglial activities. 32907830_TREM-2 defends the liver against hepatocellular carcinoma through multifactorial protective mechanisms. 32952550_Microglia TREM2: A Potential Role in the Mechanism of Action of Electroacupuncture in an Alzheimer's Disease Animal Model. 32959884_Trem2 promotes anti-inflammatory responses in microglia and is suppressed under pro-inflammatory conditions. 32991925_Knockdown of astrocytic TREM2 in the hippocampus relieves cognitive decline in elderly male mice. 33065077_TREM2 overexpression rescues cognitive deficits in APP/PS1 transgenic mice by reducing neuroinflammation via the JAK/STAT/SOCS signaling pathway. 33097708_Gene expression and functional deficits underlie TREM2-knockout microglia responses in human models of Alzheimer's disease. 33115519_Trem2 Y38C mutation and loss of Trem2 impairs neuronal synapses in adult mice. 33139402_Loss of TREM2 Confers Resilience to Synaptic and Cognitive Impairment in Aged Mice. 33222683_Diesel exhaust impairs TREM2 to dysregulate neuroinflammation. 33308094_Geniposide Enhances Macrophage Autophagy through Downregulation of TREM2 in Atherosclerosis. 33404075_Differential expression of Triggering Receptor Expressed on Myeloid cells 2 (Trem2) in tissue eosinophils. 33422057_Engagement of TREM2 by a novel monoclonal antibody induces activation of microglia and improves cognitive function in Alzheimer's disease models. 33446504_Prior activation state shapes the microglia response to antihuman TREM2 in a mouse model of Alzheimer's disease. 33448372_MicroRNA-3473b regulates the expression of TREM2/ULK1 and inhibits autophagy in inflammatory pathogenesis of Parkinson disease. 33556472_Triggering receptor expressed on myeloid Cells-2 (TREM2) inhibits steroidogenesis in adrenocortical cell by macrophage-derived exosomes in lipopolysaccharide-induced septic shock. 33586673_TREM2 sustains macrophage-hepatocyte metabolic coordination in nonalcoholic fatty liver disease and sepsis. 33620643_Pinpointing Brain TREM2 Levels in Two Mouse Models of Alzheimer's Disease. 33628208_Trem-2 Promotes Emergence of Restorative Macrophages and Endothelial Cells During Recovery From Hepatic Tissue Damage. 33675684_Trem2 restrains the enhancement of tau accumulation and neurodegeneration by beta-amyloid pathology. 33823153_Wild-type sTREM2 blocks Abeta aggregation and neurotoxicity, but the Alzheimer's R47H mutant increases Abeta aggregation. 33841415_Triggering Receptor Expressed on Myeloid Cells-2 (TREM2) Interacts With Colony-Stimulating Factor 1 Receptor (CSF1R) but Is Not Necessary for CSF1/CSF1R-Mediated Microglial Survival. 33845849_High-salt diet downregulates TREM2 expression and blunts efferocytosis of macrophages after acute ischemic stroke. 33848212_TREM2 Dictates Antibacterial Defense and Viability of Bone Marrow-derived Macrophages during Bacterial Infection. 33863908_TREM2 is a receptor for non-glycosylated mycolic acids of mycobacteria that limits anti-mycobacterial macrophage activation. 33980160_TREM2 promotes natural killer cell development in CD3(-)CD122(+)NK1.1(+) pNK cells. 34019997_THY-Tau22 mouse model accumulates more tauopathy at late stage of the disease in response to microglia deactivation through TREM2 deficiency. 34064330_Established Beta Amyloid Pathology Is Unaffected by TREM2 Elevation in Reactive Microglia in an Alzheimer's Disease Mouse Model. 34120899_TREM2 Deficiency Disrupts Network Oscillations Leading to Epileptic Activity and Aggravates Amyloid-beta-Related Hippocampal Pathophysiology in Mice. 34172807_Modulation of microglial phenotypes by dexmedetomidine through TREM2 reduces neuroinflammation in heatstroke. 34187891_Acute Trem2 reduction triggers increased microglial phagocytosis, slowing amyloid deposition in mice. 34375614_TREM2 is thyroid hormone regulated making the TREM2 pathway druggable with ligands for thyroid hormone receptor. 34424266_TREM2-dependent lipid droplet biogenesis in phagocytes is required for remyelination. 34427354_Differential interaction with TREM2 modulates microglial uptake of modified Abeta species. 34503586_Effects of microglial depletion and TREM2 deficiency on Abeta plaque burden and neuritic plaque tau pathology in 5XFAD mice. 34663480_TREM2 modulates differential deposition of modified and non-modified Abeta species in extracellular plaques and intraneuronal deposits. 34672769_Identification and Validation of TREM2 in Intracranial Aneurysms. 34851693_AD-linked R47H-TREM2 mutation induces disease-enhancing microglial states via AKT hyperactivation. 34893068_Identification of the minimal active soluble TREM2 sequence for modulating microglial phenotypes and amyloid pathology. 34895041_Maprotiline ameliorates isoflurane-induced microglial activation via regulating triggering receptor expressed in myeloid cells 2 (TREM2). 34916658_TREM2 interacts with TDP-43 and mediates microglial neuroprotection against TDP-43-related neurodegeneration. 34959070_Downregulation of TREM2 expression exacerbates neuroinflammatory responses through TLR4-mediated MAPK signaling pathway in a transgenic mouse model of Alzheimer's disease. 35019161_Loss of TREM2 rescues hyperactivation of microglia, but not lysosomal deficits and neurotoxicity in models of progranulin deficiency. 35306602_TREM2 and CD163 Ameliorate Microglia-Mediated Inflammatory Environment in the Aging Brain. 35452628_Treadmill exercise improve recognition memory by TREM2 pathway to inhibit hippocampal microglial activation and neuroinflammation in Alzheimer's disease model. 35532794_TREM-2 mediates dendritic cell-induced NO to suppress Th17 activation and ameliorate chronic kidney diseases. 35575023_Triggering Receptor Expressed on Myeloid Cells 2 (TREM2) R47H Variant Causes Distinct Age- and Sex-Dependent Musculoskeletal Alterations in Mice. 35649351_Sustained Trem2 stabilization accelerates microglia heterogeneity and Abeta pathology in a mouse model of Alzheimer's disease. 35672148_Microglial mTOR Activation Upregulates Trem2 and Enhances beta-Amyloid Plaque Clearance in the 5XFAD Alzheimer's Disease Model. 35717259_LILRB2-mediated TREM2 signaling inhibition suppresses microglia functions. 35750136_TREM-2 plays a protective role in cholestasis by acting as a negative regulator of inflammation. 35750138_Soluble TREM2 levels reflect the recruitment and expansion of TREM2(+) macrophages that localize to fibrotic areas and limit NASH. 36002854_Microglial TYROBP/DAP12 in Alzheimer's disease: Transduction of physiological and pathological signals across TREM2. 36044273_Exon 2-mediated deletion of Trem2 does not worsen metabolic function in diet-induced obese mice. 36056435_Trem2 deletion enhances tau dispersion and pathology through microglia exosomes. 36183964_Microglial response in triggering receptor expressed on myeloid cells 2 (TREM2) knock-out mice after systemic stimulation with Escherichia coli. 36241997_TPM1 mediates inflammation downstream of TREM2 via the PKA/CREB signaling pathway. 36252333_Nobiletin protects enteric nerves and ameliorates disordered bowel motility in diet-induced obese mice via increasing Trem2 expression. 36283335_Soluble triggering receptor expressed on myeloid cells 2 (sTREM2) positively regulates lipopolysaccharide-induced expression of CXC chemokine ligand 10 and 11 in mouse macrophages. 36316319_Nrf2 regulates the arginase 1(+) microglia phenotype through the initiation of TREM2 transcription, ameliorating depression-like behavior in mice. 36333761_Trem2 deficiency impairs recovery and phagocytosis and dysregulates myeloid gene expression during virus-induced demyelination. 36389767_Triggering receptor expressed on myeloid cells 2 deficiency exacerbates injury-induced inflammation in a mouse model of tauopathy. 36417868_Plaque contact and unimpaired Trem2 is required for the microglial response to amyloid pathology. 36463233_TREM2 activation alleviates neural damage via Akt/CREB/BDNF signalling after traumatic brain injury in mice. 36721205_Trem2 H157Y increases soluble TREM2 production and reduces amyloid pathology. 36725189_Soluble Trem2 is a negative regulator of erythrophagocytosis after intracerebral hemorrhage in a CD36 receptor recycling manner. 36755323_Genetic models of cleavage-reduced and soluble TREM2 reveal distinct effects on myelination and microglia function in the cuprizone model. 36805764_TREM2 splice isoforms generate soluble TREM2 species that disrupt long-term potentiation. |
ENSG00000095970 |
TREM2 |
36.115604 |
2.6989526186 |
1.432400 |
0.46063718 |
9.608803 |
0.00193646780645147079981394089287505266838707029819488525390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0082808190982225724635634733772349136415868997573852539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
49.0294073215555 |
15.9219131420344 |
18.1660867196562 |
4.57267625000088 |
| ENSMUSG00000024011 |
74116 |
Pi16 |
protein_coding |
Q9ET66 |
FUNCTION: May inhibit cardiomyocyte growth. {ECO:0000269|PubMed:17909105}. |
Alternative splicing;Glycoprotein;Protease inhibitor;Reference proteome;Secreted;Signal |
|
|
mmu:74116; |
extracellular space [GO:0005615]; peptidase inhibitor activity [GO:0030414]; negative regulation of cell growth involved in cardiac muscle cell development [GO:0061052]; negative regulation of peptidase activity [GO:0010466] |
17909105_PI16, a novel protein secreted from the heart, is strongly upregulated early in heart failure and inhibits growth of cardiomyocytes both in vitro and in vivo 27539859_our data indicate that PI16 suppresses chemerin activation in the myocardium and suggest that this circuit may be part of the cardiac stress response. 32079726_these findings support a model in which PI16 promotes neuropathic pain by mediating a cross-talk between fibroblasts and the endothelial barrier leading to barrier opening, cellular influx, and increased pain 32227584_Overexpression of peptidase inhibitor 16 attenuates angiotensin II-induced cardiac fibrosis via regulating HDAC1 of cardiac fibroblasts. |
ENSG00000164530 |
PI16 |
138.891159 |
3.9693026483 |
1.988886 |
0.26656854 |
54.120265 |
0.00000000000018858627057586422973016001152559056308339983931698924379816162399947643280029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000041036372477308061882279073742917909969896150812473933910951018333435058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
215.841515304811 |
40.108617253533 |
54.3776916078029 |
7.97748359260061 |
| ENSMUSG00000024076 |
74199 |
Vit |
protein_coding |
Q8VHI5 |
FUNCTION: Promotes matrix assembly and cell adhesiveness (PubMed:18757743). Plays a role in spinal cord formation by regulating the proliferation and differentiation of neural stem cells (PubMed:25331329). {ECO:0000269|PubMed:18757743, ECO:0000269|PubMed:25331329}. |
Alternative splicing;Disulfide bond;Extracellular matrix;Glycoprotein;Neurogenesis;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:74199; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; interstitial matrix [GO:0005614]; glycosaminoglycan binding [GO:0005539]; extracellular matrix organization [GO:0030198]; positive regulation of cell-substrate adhesion [GO:0010811]; spinal cord development [GO:0021510] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000205221 |
VIT |
804.560000 |
2.4422440882 |
1.288207 |
0.09912450 |
167.087681 |
0.00000000000000000000000000000000000003200939194034191326653548769831587593866167222840215465344710344291402062436340475099515169974024892535213737687627144623547792434692382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000034095598464006153343165423782066182043123030484534998811911984507739056648846614990396744026938946614535552726010791957378387451171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1140.87376638178 |
78.4486961729381 |
467.141581746894 |
25.000767272415 |
| ENSMUSG00000024140 |
13819 |
Epas1 |
protein_coding |
P97481 |
FUNCTION: Transcription factor involved in the induction of oxygen regulated genes. Heterodimerizes with ARNT; heterodimer binds to core DNA sequence 5'-TACGTG-3' within the hypoxia response element (HRE) of target gene promoters (PubMed:26245371). Regulates the vascular endothelial growth factor (VEGF) expression and seems to be implicated in the development of blood vessels and the tubular system of lung. May also play a role in the formation of the endothelium that gives rise to the blood brain barrier. Potent activator of the Tie-2 tyrosine kinase expression. Activation requires recruitment of transcriptional coactivators such as CREBBP and probably EP300. Interaction with redox regulatory protein APEX seems to activate CTAD (By similarity). {ECO:0000250, ECO:0000269|PubMed:26245371}. |
3D-structure;Activator;Angiogenesis;Developmental protein;Differentiation;Direct protein sequencing;DNA-binding;Hydroxylation;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:13819; |
cytoplasm [GO:0005737]; nuclear speck [GO:0016607]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; cobalt ion binding [GO:0050897]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; protein heterodimerization activity [GO:0046982]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; transcription coactivator binding [GO:0001223]; angiogenesis [GO:0001525]; blood vessel remodeling [GO:0001974]; cell differentiation [GO:0030154]; cellular response to hypoxia [GO:0071456]; embryonic placenta development [GO:0001892]; epithelial cell maturation [GO:0002070]; erythrocyte differentiation [GO:0030218]; gene expression [GO:0010467]; hemopoiesis [GO:0030097]; iron ion homeostasis [GO:0055072]; lung development [GO:0030324]; mitochondrion organization [GO:0007005]; mRNA transcription by RNA polymerase II [GO:0042789]; myoblast fate commitment [GO:0048625]; negative regulation of transcription by RNA polymerase II [GO:0000122]; norepinephrine biosynthetic process [GO:0042421]; norepinephrine metabolic process [GO:0042415]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of dopamine biosynthetic process [GO:1903181]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of heart rate [GO:0002027]; regulation of protein neddylation [GO:2000434]; regulation of transcription by RNA polymerase II [GO:0006357]; regulation of transcription from RNA polymerase II promoter in response to oxidative stress [GO:0043619]; response to hypoxia [GO:0001666]; response to oxidative stress [GO:0006979]; surfactant homeostasis [GO:0043129]; transcription by RNA polymerase II [GO:0006366]; visual perception [GO:0007601] |
11823643_hypoxic induction of the COOH-terminal transactivation domain (CAD)occurs through abrogation of hydroxylation of a conserved asparagine in the CAD 11839666_Hypoxia-induced, perinecrotic expression of endothelial Per-ARNT-Sim domain protein-1/hypoxia-inducible factor-2alpha correlates with tumor progression, vascularization, and focal macrophage infiltration in bladder cancer. 12053176_loss impairs fetal lung maturation, whereas treatment with VEGF prevents fatal respiratory distress in premature mice 12464608_HIF-2alpha and Ets-1 binding is required for transcriptional activation of Flk-1 in endothelial cells 12606578_Data suggest that erythropoietin is one of the target genes of hypoxia-inducible factor-2 alpha responsible for experimental retinopathy of prematurity. 12750163_critical role for EPAS1/HIF-2alpha in maintaining a functional microenvironment in the bone marrow for effective hematopoiesis. 12750401_Hif2alpha(+/-) mice were fully protected against pulmonary hypertension and right ventricular hypertrophy after chronic hypoxia, revealing a critical role of HIF-2alpha in hypoxia-induced pulmonary vascular remodeling 12832481_HIF-2alpha is not a redundant transcription factor of HIF-1alpha for hypoxia-induced gene expression, but there is a cell type-specific modulator(s) that enables selective activation of HIF-1alpha but not HIF-2alpha in response to low-oxygen stress. 14608355_Adult HIF-2a/EPAS1(-/-) mice are a phenocopy of mice deficient in SOD2 and multiple major antioxidant enzyme genes including SOD2 are HIF-2 target genes, indicating HIF-2 functions as a rheostat for ROS and mitochondrial homeostasis. 14691554_HIF2alpha has a role in regulating pVHL-defective tumor growth 15190133_nucleotide preference of the heterodimers of HLF and Arnt 15258146_EPAS1 plays several supporting roles in maintaining specific aspects of adipogenesis and adipocyte function 15626745_HIF-2alpha is an essential regulator of murine erythropoietin production 15653678_Results describe the specific interaction of hypoxia-inducible factors (HIF)-2alpha, but not HIF-1alpha, with the NF-kappaB essential modulator (NEMO). 15689487_HIF and O(2) tension influence placental invasion and tumor migration by increasing cell surface expression of alphavbeta3 integrin 15851592_hypoxia may control endothelial functions directly via Hif-2alpha-regulated Tie-2 expression 16287860_HIF1alpha and HIF2alpha, are essential for determining murine placental cell fates 16394508_Sp1 and Sp3 transcription factors are involved in transcriptional regulation of HIF-2alpha expression during adipogenesis in 3T3-L1 cells 16571862_These results suggest that HIF-dependent repression of epithelial NKCC1 may provide a compensatory mechanism to prevent excessive fluid loss during hypoxia. 16611993_They report that HIF-2alpha is not transcriptionally active in embryonic stem (ES) cells, as well as possible inhibition by a HIF-2alpha-specific transcriptional repressor. 16899596_The HIF-2alpha was expressed in KS biopsies in all tumoral stages. 16971531_These results indicate that HIF-2alpha mediates the transcriptional activation of EPO expression in astrocytes, and this pathway may promote astrocytic paracrine-dependent neuronal survival during ischemia. 16977322_The pathological changes caused by pVHL inactivation in skin and liver are due largely to dysregulation of HIF target genes. 17220275_HIF2alpha C-terminal transactivation domain, in contrast to the HIF1alpha C-terminal transactivation domain, is relatively resistant to the inhibitory effects of FIH1 under normoxic conditions 17284606_HIF-2alpha is the critical alpha isoform regulating Epo under physiologic and stress conditions in adult mice 17322295_Hypoxia-inducible factor 2alpha regulates frataxin expression 17342756_Although HIF2alpha was widely transcribed by kidney endothelium and vascular smooth muscle, knockouts displayed no detectable deficits in vessel development or VEGF or Flk-1 expression. 17507435_HIF-alpha proteins and regulated genes are not increased in placentas from normotensive pregnant women delivering small, asymmetrically grown babies 17655754_A combined antiproliferative and a pro-angiogenic function of HIF2alpha actually seems to favor the establishment of neuroblastomas in vivo. 17712557_HIF-1alpha and HIF-2alpha appear to contribute to erythropoietin expression tissue specifically 17804822_HIF-alpha terminal transactivation domains (TAD), particularly the N-TADs, confer HIF target gene specificity, by interacting with additional transcriptional cofactors 17992257_enhanced expression of key HIF-2alpha genes promotes splenic erythropoiesis, resulting in the development of polycythemia in Vhl(R/R) mice 18356544_Abcg2 is a direct downstream target of HIF-2alpha which functions with other factors to initiate a cytoprotective program for this progenitor cell population that resides in the adult heart. 18434321_HIF-2alpha plays an essential role in vascular remodeling during tumor vascularization through activation of at least ephrin A1. 18439915_Hypoxia-inducible factor augments experimental colitis through an MIF-dependent inflammatory signaling cascade. 18451309_the hematopoietic microenvironment required for erythropoiesis is dynamically regulated by oxygen through the functions of HIF-2alpha in ECs. 18635823_Tfap2beta, Et-1, and Hif2alpha act in a transcriptional network during ductal smooth muscle development. Disruption of this pathway may contribute to patent ductus arteriosus by affecting development of smooth muscle in the ductus arteriosus. 18832080_Placental HIF-2alpha is an early marker of fetal hypoxic distress. 19252526_Type 2B R167Q mutant pVhl produces a unique profile of HIF dysregulation, thereby promoting tissue-specific effects on cell growth, development and tumor predisposition. 19302442_Report hypoxia-inducible factor-dependent production of profibrotic mediators by hypoxic hepatocytes. 19342889_HIF-1 and HIF-2 can individually arrest the cell cycle independent from hypoxia. 19352007_HIF-2alpha plays a crucial role in maintaining iron balance in the organism by directly regulating the transcription of the gene encoding divalent metal transporter 1 (DMT1), the principal intestinal iron transporter. 19439736_Immortalized HIF-2alpha-deficient endothelial cells displayed decreased adhesion to extracellular matrix proteins 19454749_Both HIF-1 and -2 are important transcriptional effectors regulating the responses of macrophages to a period of hypoxia. 19461047_adaptation to hypoxia exerts a protective role on cardiomyocytes subjected to ischemia and that, unexpectedly, this form of preconditioning absolutely depends on Hif-2alpha. 19498162_findings show that Sirt1 promotes HIF-2 signaling during hypoxia and likely other environmental stresses 19528226_Data demonstrate that hypoxia-inducible factor-2alpha functions as an important regulator of hepatic lipid metabolism and identify HIF-2 as a potential target for the treatment of fatty liver disease. 19662677_HIF2alpha cooperates with RAS to promote lung tumorigenesis in mice. 20005221_retinal HIF-1alpha and HIF-2alpha were not found to be increased, and the extent of hypoxia may even decrease after 12 weeks of hyperglycemia in rats. 20140661_In Hif1a(+/-) mice, hepatic HIF-2alpha mRNA was increased (2-fold) and the mass of the liver was increased, compared with wild-type littermates, without significant difference in cell volume. 20181618_EPAS1 (and not HIF1A) regulates the expression of genes encoding Sertoli cell tight junction proteins (TJP1, TJP2, and occludin) that establish an effective blood-testis barrier. 20194441_HIF-1alpha and HIF-2alpha, can have physiologically antagonistic functions, but that their antiphase regulation allows them to coordinately regulate NO production in a cytokine-induced and transcription-dependent fashion 20495569_causes osteoarthritic cartilage destruction by regulating crucial catabolic genes 20495570_central transactivator targets several crucial genes for endochondral ossification and may represent a therapeutic target for osteoarthritis 20644254_HIF-2alpha as an important regulator of innate immunity, suggesting it may be a useful therapeutic target for treating inflammatory disorders and cancer. 20678473_These results suggest that HIF2-alpha is required for normal mouse pancreatic development. 20713899_the pathway involving INT6/HIF2alpha acts as a hypoxia-independent master switch of functional angiogenesis 21042733_Two HREs located around -3.6 kb of the 5'-flanking promoter region of the mouse PAI-1 gene function as hypoxia enhancers, which control PAI-1 gene transcription by HIF-1 or HIF-2 under hypoxic environments in mouse hepatoma cells. 21106753_These data indicate that HIF2alpha acts downstream of PGC-1alpha as a key regulator of a muscle fiber-type program and the adaptive response to exercise. 21288809_HIF-2alpha-dependent redox regulation is required for maintenance of carotid body function and cardiorespiratory homeostasis. 21419768_HIF-2alpha directly binds to the ferroportin promoter and induces its expression, indicating a mechanism of transcriptional regulation of Fpn following changes in systemic levels of iron. 21447692_Artificial activation of HIF transcription factors in normoxic photoreceptors results in an increased basal expression of Fgf2 that may contribute to a transient protection of rods against light damage. 21498508_the essential role of HIF-2alpha in regulating iron absorption and also demonstrate that hypoxia sensing in the intestine, as well as in the kidney, is essential for regulation of erythropoiesis by HIF-2alpha. 21517900_HIF-1alpha and HIF-2alpha are upregulated in mouse lung in a mouse model of allergic airway inflammation. 21765015_Hypoxia-inducible factor-2alpha regulates GM-CSF-derived soluble vascular endothelial growth factor receptor 1 production from macrophages and inhibits tumor growth and angiogenesis. 21814490_overexpression of HIF1alpha and HIF2alpha upregulated propiomelanocortin gene expression 12 folds and 26 folds respectively 21869830_HIF-2alpha potentiates Fas-mediated chondrocyte apoptosis, which is associated with OA cartilage destruction. 22014577_Fh1-associated cyst formation is Hif independent, as is striking upregulation of antioxidant signaling pathways revealed by gene expression profiling. 22095691_findings show that C/EBPbeta and RUNX2, with MMP-13 as the target and HIF-2alpha as the inducer, control cartilage degradation 22128145_Deletion of HIF-2alphaalpha in the enterocytes decreases the severity of tissue iron loading in hepcidin knockout mice 22207682_Hepatic hypoxia-inducible factor-2 down-regulates hepcidin expression in mice through an erythropoietin-mediated increase in erythropoiesis. 22223045_Data show HIF1alpha and HIF2alpha with point mutations were significantly more effective in trans-activating the target gene VEGF and in inducing tube formation of endothelial cells than mutants lacking the complete oxygen-dependent degradation (ODD) domain. 22235342_Glucose-induced O consumption creates an intracellular hypoxia that activates HIF1 and HIF2 in rat beta-cells 22264788_Loss of HIF-1alpha in endothelial cells reduces NO synthesis, retards tumor cell migration through endothelial layers, and restricts tumor cell metastasis, and that loss of HIF-2alpha has in each case the opposite effect. 22295069_ureteral obstruction protected the kidney via activation of HIF-2alpha 22298531_conclude that Hif2alpha is a key regulator in alveolar maturation and the production of phospholipids by Type II cells 22299048_VHL appears to specifically repress HIF-2alpha in renal epithelia. Renal tubular HIF-2alpha expression requires VHL inactivation and causes fibrosis and cysts. 22426208_HIF-2alpha expression is required for angiogenic responses during hindlimb ischemia and for the growth of autochthonous skin tumors. 22490864_Global activation of Hif-2a suppresses inflammation and fibrogenesis in mice subjected to unilateral ureteral obstruction. 22684009_basal expression of Ctr1 is regulated by HIF2alpha; however, the induction by hypoxia is a HIF2alpha-independent event 22692279_AMP-activated protein kinase activation ameliorates airway inflammatory responses by reducing vascular permeability via HIF1alpha/hif2alpha/VEGFA as well as by inhibiting reactive oxygen species production 22807441_The acetylase/deacetylase couple CREB-binding protein/Sirtuin 1 controls hypoxia-inducible factor 2 signaling. 22869521_Mouse models of liver HIF overexpression demonstrated that both HIF-1alpha and HIF-2alpha contribute to hepcidin repression in vivo. 22869907_Specific activation of HIF-2alpha decreases tumor growth and angiogenesis in a murine melanoma model. 23103253_HIF-2alpha/mTORC1/SLC7A5 pathway mediates proliferation and tumor promoting properties of HIF-2alpha. 23114598_HIF-mediated suppression of the hepcidin gene (Hamp1) required EPO induction. 23250618_HIF-1alpha and HIF-2alpha regulate eosinophil migration and asthma airway resistance in opposing ways. 23264599_Conditional loss of PHD2 in mice leads to HIF-2alpha-dependent erythrocytosis, whereas HIF-1alpha protects these mice. 23384425_Inducible, Pax8-rtTA-based deletion of VHL leads to organ-specific expression of epithelial HIF and erythropoietin in liver and kidney without causing pathological changes. 23395173_Irp1 deletion enhanced HIF2alpha protein expression in kidneys of Irp1(-/-) mice. 23395174_These results uncover separable physiological roles of each IRP and identify IRP1 as a therapeutic target for manipulating HIF-2alpha action in hematologic, oncologic, and other disorders. 23447580_a constitutively active HIF2alpha is not sufficient to cause neoplastic transformation of proximal tubules, arguing against the idea that HIF2alpha activation is critical for ccRCC tumorigenesis 23520526_inhibition of HIF-1/2alpha did not reverse the effects of hypoxia on IL-6 gene expression. In addition, phosphorylation of p38, but not JNK, was responsible for the effects of glucose deprivation on IL-6 gene expression. 23555700_HIF-2alpha as an important regulator of iron transport machinery in copper deficiency 23563312_HIF1alpha and HIF2alpha activate independent transcriptional programs through SRC that promote metastasis by coordinately regulating cell invasion and ECM remodeling 23640890_Missense mutations in HIF-2alpha are a cause of erythrocytosis. 23777768_IRP1 is the principal regulator of HIF2alpha mRNA translation 23845627_Massive small bowel resection results in an immediate relative hypoxic state within the remnant bowel with early enhanced expression of HIF1alpha and decreased expression of HIF2alpha. 23852473_Significant accumulation of hypoxia-inducible factors 1alpha and 2alpha (HIF-1alpha and HIF-2alpha). 23860500_Intestine-specific overexpression of EPAS1, but not HIF-1alpha, led to spontaneous colitis 23894152_hematopoietic stem cells do not require Hif-2alpha to self-renew and recover after hematopoietic injury. 23900840_Hypoxia-inducible factor-2alpha-dependent hypoxic induction of Wnt10b expression in adipogenic cells. 23946285_HIF-2alpha target genes are upregulated in sclerosing glomeruli. 24025643_LPS preconditioning protected against renal ischemia/reperfusion injury by HIF-2alpha activation in endothelial cells that subsequently improved renal microvascular perfusion and reduced ischemic tubular damage. 24030251_In proximal tubular cells, AGE-BSA stimulation elevated mainly HIF-1alpha transcriptional activity and to a lesser extent HIF-2alpha. 24037094_results demonstrate an unsuspected intersection between Hif-2alpha-mediated hypoxic signaling and hepatic insulin action through Irs2 induction, which can be co-opted by Vegf inhibitors to modulate glucose metabolism 24037341_Manipulating or reducing the HIF-2alpha response in keratinocytes could be a useful means to accelerate wound healing and tissue repair. 24054150_HIF2a signaling appears to be a central pathway in chromaffin cell development and differentiation, with cells highly expressing HIF2a exhibiting a less differentiated and more stem cell-like phenotype. 24101470_Data show that the balance between HIF-1alpha and HIF-2alpha expression in keratinocytes is a control element of both tissue perfusion and systemic arterial pressure, with potential implications in human hypertension. 24196071_HIF-2alpha has a role in persistence of neutrophilic inflammation in humans, mice and zebrafish 24282296_Dysregulation of intestinal hypoxia and HIF2alpha signaling is critical for progressive iron overload in beta-thalassemia. 24326162_Suggest direct role for the adipocyte HIF-2 transcription factor in the development of adipose inflammation and pathological cardiac hypertrophy. 24421394_ption of Vhl led to a HIF-2alpha-dependent decrease in cholesterol hydroxylases involved in the conversion of cholesterol to bile acids. 24475033_Hypoxia inducible factor-2alpha regulates the development of retinal astrocytic network by maintaining adequate supply of astrocyte progenitors. 24503013_HIF-1alpha and HIF-2alpha are involved in hypoxia-induced lipid accumulation in hepatocytes via reducing PGC-1alpha mediated fatty acid beta-oxidation. 24551189_HIF-1alpha and HIF-2alpha promote angiogenesis of residual hepatocellular carcinoma after high-intensity focused ultrasound ablation. 24576667_both HIF2alpha and ETS1 are involved in the transcriptional regulation of Flvcr1a and that HIF2alpha is absolutely required for Flvcr1a induction upon hypoxia 24598112_HIF-2alpha/IGF-1R signal loop supports the proliferation and Oct4 maintenance of mouse germline stem cells. 24599965_results suggest that the HIF-2alpha/ARNT complex regulates hair follicle differentiation via induction of p21(Waf1/Cip1) and possibly p27(Kip1) 24607765_Hypoxia-mediated changes in cardiac mitochondrial function are a conserved target of both HIF-dependent and independent signaling pathways. 24760996_Suggest that in anemia each organ expresses a different threshold for cellular HIF/NOS hypoxia responses. 24789906_inactivation of endothelial HIF-2alpha, but not endothelial HIF-1alpha, resulted in increased expression of renal injury markers and inflammatory cell infiltration in the postischemic kidney, which was reversed by blockade of VCAM1 and VLA4 24828078_HIF2, but not HIF1, is both necessary and sufficient to prevent radiation-induced GI toxicity and death in knockout mice 24914685_pivotal role in the pathogenesis of rheumatoid arthritis by regulating fibroblast-like synoviocyte functions, independent of HIF-1alpha 24947359_these data suggest that macrophage HIF-2alpha would counteract the proinflammatory responses to relieve obesity-induced insulin resistance in adipose tissue. 24999590_Hypoxic treatment of primary keratinocytes induced filaggrin (Flg) gene expression in a HIF1alpha- and HIF2alpha-dependent manner. 25018103_Study shows that oxygen tension, mediated by Oligodendrocyte precursor cells (OPC)-encoded HIF function, is an essential regulator of postnatal myelination; constitutive HIF1/2alpha stabilization resulted in OPC maturation arrest through autocrine activation of canonical Wnt7a/7b. 25071089_Chronic activation of HIF-2 in renal juxtaglomerular cells leads to a reprogramming of the cells into fibroblast-like cells resembling native erythropoietin-producing cells located in the tubulointerstitium. 25087887_Letter: HIF2alpha is expressed in a distinct spatial and temporal pattern throughout venous thrombus resolution. 25103499_The PTEN/PI3K/AKT signaling axis controls hypoxia-induced HIF1alpha (HIF1A) and HIF2alpha (EPAS1) stability in macrophages. 25108527_The acetate-dependent acetyl CoA generator Acss2 links changes in metabolism that occur during stress with the coupled processes of Hif-2 acetylation, which is mediated by the acetylase/coactivator Cbp, and stable Hif-2/Cbp complex formation, both of which are required for maximal Hif-2 signaling. 25394489_these results suggest that PTEN is required for apoptosis-mediated cavitation during epithelial morphogenesis by regulating the expression of HIF-2alpha and Bnip3 25440060_Hif-2alpha activation augments peroxisome turnover by selective autophagy (pexophagy) and thereby changes lipid composition reminiscent of peroxisomal disorders. 25450522_713 HIF-1 and 795 HIF-2 binding sites were identified under hypoxia. Pretreatment with IL-10 altered the binding pattern, with 120 new HIF-1 and 188 new HIF-2 binding sites emerging 25557133_These results suggest that there is a compensatory pathway involving HIF-1alpha that regulates the expression of some HIF-2alpha target genes. 25565665_Results show that HIF2-a is up regulated in an in vitro model of nicotine plus lipopolysccharide-stimulated periodontal ligament cell line and primary culture. Its inhibition exhibits anti-inflammatory activity. 25574837_activation of HIF2alpha/VE-PTP signaling via PHD2 inhibition has the potential to prevent the formation of leaky vessels and edema in inflammatory diseases such as ARDS 25750431_we showed that muscles did not require macrophagic hif1a or hif2a to regenerate. These results indicate that macrophagic HIFs do not play a crucial role during skeletal muscle regeneration induced by sterile tissue damage. 25758454_eIF3e is an angiogenesis suppressor. Silencing of eIF3e promotes blood perfusion recovery after limb ischemia through stabilization of hypoxia-inducible factor 2alpha activity. 25833250_excess in pancreatic beta-cells does not play a significant role in beta-cell function and glucose homeostasis 25889500_HIF2alpha has a cardiomyogenesis-promoting effect in embryonic stem cells via enhancing the activation of the Wnt/beta-catenin signaling pathway. 25921247_High expression of HIF-2A is associated with leukemia and lymphoma. 25953833_This review emphasizes that sympathetic nervous system homeostasis is dependent on a balance between HIF-2alpha and HIF-1alpha. 26124130_details the importance of breast milk iron in neonatal development and uncovers an unexpected molecular mechanism for the regulation of nutritional status of breast milk through intestinal HIF-2alpha 26241779_Findings identify a reciprocal activation mechanism involving HIF-2alpha and the zinc-ZIP8-MTF1 axis during osteoarthritis pathogenesis that amplifies catabolic signaling and cartilage destruction. 26245371_crystal structures for each of mouse HIF-2alpha-ARNT and HIF-1alpha-ARNT heterodimers in states that include bound small molecules and their hypoxia response element 26296885_data suggest that targeting intestinal HIF-2alpha has a significant therapeutic potential in SCD pathophysiology 26348575_Activation of the HIF signaling pathway could be an important pathogenetic event in the development and progression of at least a subset of these tumors. 26422241_Loss of Hif-2alpha Rescues the Hif-1alpha Deletion Phenotype of Neonatal Respiratory Distress In Mice 26503986_demonstrate that HIF-2alpha is clearly implicated in the TSP1 pulmonary regulation and provide new insights on its contribution to pulmonary arterial hypertension-driven vascular remodelling and vasoconstriction 26572826_HIF-2alpha-deficient mice displayed brown adipose tissue dysregulation, associated with reduced levels of uncoupling protein 1 (UCP1) and a dysfunctional thermogenic response to cold exposure. 26642431_Chondrocytes co-cultured with HIF-2alpha-overexpressing fibroblast-like synoviocytes exhibited upregulation of Mmp3 and Mmp13, which is similar to the effects induced by interleukin (IL)-6 treatment of chondrocytes. 26642852_although Hif-1alpha and Hif-2alpha synergize to suppress the development of acute myeloid leukemia, they are not required for leukemic stem cells maintenance. 26649797_AQP1 deficiency delays long-term ischemia-reperfusion resolution, partly through repressing angiogenesis mediated by destabilizing HIF-2alpha. 26746864_in neurons HIF-1 and HIF-2 have redundant functions for cellular survival under ischemic conditions. By contrast, lack of anti-survival factors in Hif1a/Hif2a-deficient mice might protect from early acute neuronal cell death and neurological impairment, indicating a benefit of HIF-pathway inhibition in neurons in the very acute phase after ischemic stroke 26837714_HIF-2alpha inhibits high-grade soft tissue sarcoma cell growth. Loss of HIF-2alpha promotes sarcoma proliferation and increases calcium and mTORC1 signalling in undifferentiated pleomorphic sarcoma and dedifferentiated liposarcoma. 26853750_HIF2alpha is essential for the acute postprandial regulation of hepatic glucagon signaling. 26927670_genetic analysis suggests that tubulointerstitial cellular crosstalk modulates renal EPO production under conditions of epithelial HIF activation in the kidney. 26976644_Data identify endothelial HIF-2 as a key transcription factor in the pathogenesis of pulmonary hypertension. 27075652_The regulatory connection between HIF-2alpha and VEGFR-1, VEGFR-2 and MMP2. 27432976_Data suggest that chronic hypoxia enhances HIF-2alpha stability, which causes increased arginase expression and dysregulates normal vascular NO homeostasis. 27528422_HIF-1alpha-dependent, HIF-2alpha-independent angiogenesis and constitutive diuresis is caused by Vhl deletion in renal epithelia 27600268_A novel biological pathway has been discovered of soluble biglycan inducing HIF-2alpha protein stabilization and Epo production presumably in an oxygen-independent manner, ultimately giving rise to secondary polycythemia. 27671111_this study shows that HIF-2alpha acts in resting macrophages as a phagocytosis suppressor implying an important relationship between the levels of HIF-2alpha and susceptibility to infection 27956697_HIF-2alpha is a novel regulator of neutrophil recruitment to colon tumors. 28067314_EPAS1 links DOCK8 deficiency to atopic skin inflammation via IL-31 induction in CD4thorn T cells. 28112274_Myeloid-specific deletion of Epas1 had no impact on the number of myeloid cells migrating into the eye. 28232488_HIF1alpha and HIF2alpha are dispensable for muscle stem cell function under normoxia but are required for maintaining satellite cell self-renewal in hypoxic environments. 28527294_Chronic activity of HIF2 in stromal progenitors impairs kidney development. Finally, these data confirm the concept that normal stroma function is essential for normal tubular differentiation. 28696319_HIF2alpha is linked to tumor suppression in neuroblastoma. 28813675_The pseudo-hypoxic phenotype of stem-like glioma cells is achieved by stabilization of HIF-2a through interaction with CD44, independently of oxygen. 28847650_Defect in nephron formation in PHD2/PHD3 double mutants required intact hypoxia-inducible factor-2 signaling and was dependent on the extent of stromal hypoxia-inducible factor activation. Thus, hypoxia-inducible factor prolyl-4-hydroxylation in renal interstitial cells is critical for normal nephron formation. 28848049_findings indicate that HIF-2alpha increases cancer cell growth by up-regulating YAP1 activity 28928136_endothelial EPAS1 has a global protective role during glomerular hypertensive injuries without influencing the hypertensive effect of angiotensin II 29035368_intestine HIF-2alpha regulates ceramide metabolism mainly from the salvage pathway, by positively regulating the expression of Neu3, the gene encoding neuraminidase 3. These results suggest that intestinal HIF-2alpha could be a viable target for hepatic steatosis therapy 29036334_Data show that Tet1 modulates HIF-2alpha and HIF-1alpha through different mechanisms. 29074488_Increased HIF-2alpha expression is associated with the development of severe pulmonary hypertension. 29307582_we demonstrated that hypoxia significantly induced the GSC mesenchymal transition, increased the expression levels of the pluripotent transcription factor OCT4 and migration-associated proteins..he underlying mechanism involved significant IGF-1/IGF-1R activation of OCT4/CXCR4 expression through HIF-2alpha regulation. 29480820_the reversal of erythrocytosis/polycythemia to translational repression of Hif2alpha expression by Tempol-mediated increases in the IRE-binding activity of Irp1, as reversal of polycythemia was abrogated in VhlR200W mice in which Irp1 was genetically ablated. 29483579_The studies indicate that HIF2-alpha induces myocardial AREG expression in cardiac myocytes, which increases myocardial ischemia tolerance. 29533927_These findings suggest that HIF2A is a pivotal mediator of hypoxia signaling in satellite cells and may be therapeutically targeted to improve muscle regeneration. 29615467_Absence of Hif2a in retinal neuroprogenitor cells causes a marked reduction of proliferating endothelial cells at the angiogenic front. This results in delayed retinal vascular development, fewer major retinal vessels and reduced density of the peripheral deep retinal vascular plexus. 29911998_Mice with stromal deletion of Hif2a (Hif2a-sKO mice) showed infertility with impaired embryo invasion and those with epithelial deletion of Hif2a (Hif2a-eKO mice) showed normal fertility, suggesting the importance of stromal HIF2alpha in embryo invasion. 30092219_The p75NTR enhances HIF1A/HIF2A-dependent signaling in glioma. 30156910_Hematopoietic-specific deficiency of Hif2alpha ameliorated pathological neovascularization in the OIR model, which was accompanied by enhanced endothelial cell apoptosis 30209343_Signaling pathways important for acinar cell homeostasis are altered in HIF2alpha-overexpressing pancreata. 30352047_These findings demonstrate a molecular link between hepatic hepcidin and intestinal HIF-2alpha that controls physiological iron uptake and drives iron hyperabsorption during iron overload. 30388216_Hypoxia-inducible factor (HIF) 2alpha (HIF2A) accelerates extracellular matrix deposition by inducing a collagen-cross-linking enzyme, lysyl oxidase (LOX). 30431439_Lactate inhibits ATP6V0d2 expression in tumor-associated macrophages to promote HIF-2alpha-mediated tumor progression. 30523118_High HIF-2A expression is associated with metabolic diseases. 30586708_Endothelial cell HIF-2alpha, but not HIF-1alpha, is required to maintain airway microvascular structure and function. 30709906_hypothalamic HIF2alpha responds to insulin, and the up-regulation is involved in adaptive metabolic regulation as age increases, whereas impairment of HIF2alpha in the hypothalamus contributes to weight gain and glucose disorders in age-dependent manners. 30761250_The results indicate that hypoxia directly enhances the recruitment of HIF-2alpha to the proximal promoter of Slc7a5, resulting in its upregulated expression in differentiated neuronal cells. 31357086_Up-regulated HIF-2alpha contributes to osteoarthritis (OA) development through mediating the primary cilia loss, which might be developed as a therapeutic target for OA treatment. Manipulating AURKA and NEDD9 expressions could reverse the function of HIF-2alpha on primary cilia. In mice, knocking-down both AURKA and NEDD9 could alleviate OA development significantly. 31403471_retina-derived (paracrine) and astrocyte-derived (autocrine) VEGF have roles in controlling astrocyte proliferation and migration; HIF-1alpha in retinal neurons and HIF-2alpha in Muller glia play distinct roles in retinal vascular development and disease 31436357_We propose that Spint1 contributes to the tumor-suppressive function of HIF-2a in tumor-associated macrophages in breast tumor development. 31515405_Suppression of HIF2 signalling attenuates the initiation of hypoxia-induced pulmonary hypertension. 31668872_Adipocyte Hypoxia-Inducible Factor 2alpha Suppresses Atherosclerosis by Promoting Adipose Ceramide Catabolism. 31695081_Hif1a and Hif2a can be safely inactivated in cone photoreceptors. 31848220_Acute O2 sensing through HIF2alpha-dependent expression of atypical cytochrome oxidase subunits in arterial chemoreceptors. 31999648_Marked and rapid effects of pharmacological HIF-2alpha antagonism on hypoxic ventilatory control. 32234487_TET-Mediated Hypermethylation Primes SDH-Deficient Cells for HIF2alpha-Driven Mesenchymal Transition. 32513733_A hypoxia-induced Rab pathway regulates embryo implantation by controlled trafficking of secretory granules. 32515984_Endothelial HIF-2alpha as a Key Endogenous Mediator Preventing Emphysema. 32534258_Inhibition of prolyl hydroxylases increases hepatic insulin and decreases glucagon sensitivity by an HIF-2alpha-dependent mechanism. 32659785_NCOA4 is regulated by HIF and mediates mobilization of murine hepatic iron stores after blood loss. 32673288_Decreas |
ENSG00000116016 |
EPAS1 |
94.068467 |
0.2369726936 |
-2.077207 |
0.49797911 |
15.950878 |
0.00006500761530370960448836636924596632525208406150341033935546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004036673446009395494100957524352679683943279087543487548828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.8390590595612 |
12.2657176468742 |
147.017188069109 |
36.4758159043291 |
| ENSMUSG00000024164 |
12266 |
C3 |
protein_coding |
P01027 |
FUNCTION: C3 plays a central role in the activation of the complement system. Its processing by C3 convertase is the central reaction in both classical and alternative complement pathways. After activation C3b can bind covalently, via its reactive thioester, to cell surface carbohydrates or immune aggregates.; FUNCTION: Derived from proteolytic degradation of complement C3, C3a anaphylatoxin is a mediator of local inflammatory process. In chronic inflammation, acts as a chemoattractant for neutrophils (By similarity). It induces the contraction of smooth muscle, increases vascular permeability and causes histamine release from mast cells and basophilic leukocytes. The short isoform has B-cell stimulatory activity. {ECO:0000250}.; FUNCTION: [C3-beta-c]: Acts as a chemoattractant for neutrophils in chronic inflammation. {ECO:0000250}.; FUNCTION: [Acylation stimulating protein]: Adipogenic hormone that stimulates triglyceride (TG) synthesis and glucose transport in adipocytes, regulating fat storage and playing a role in postprandial TG clearance. Appears to stimulate TG synthesis via activation of the PLC, MAPK and AKT signaling pathways. Ligand for C5AR2. Promotes the phosphorylation, ARRB2-mediated internalization and recycling of C5AR2. |
3D-structure;Alternative initiation;Cleavage on pair of basic residues;Complement alternate pathway;Complement pathway;Direct protein sequencing;Disulfide bond;Fatty acid metabolism;Glycoprotein;Immunity;Inflammatory response;Innate immunity;Lipid metabolism;Phosphoprotein;Reference proteome;Secreted;Signal;Thioester bond |
|
This gene encodes complement protein C3 which plays a central role in the classical, alternative and lectin activation pathways of the complement system. The encoded preproprotein undergoes a multi-step processing to generate various functional peptides. Mice deficient in the encoded protein fail to clear bacteria from the blood stream upon infection, display diminished airway hyperresponsiveness and lung eosinophilia upon allergen-induced pulmonary allergy, and develop severe lung injury after deposition of IgG immune complexes. Deficiency of the homolog of the encoded protein in humans was found to be associated with increased susceptibility to infections, age-related macular degeneration, and atypical hemolytic uremic syndrome. [provided by RefSeq, Mar 2015]. |
mmu:12266; |
cell surface [GO:0009986]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; serine-type endopeptidase complex [GO:1905370]; C5L2 anaphylatoxin chemotactic receptor binding [GO:0031715]; endopeptidase inhibitor activity [GO:0004866]; lipid binding [GO:0008289]; amyloid-beta clearance [GO:0097242]; blood coagulation [GO:0007596]; cell surface receptor signaling pathway involved in cell-cell signaling [GO:1905114]; complement activation [GO:0006956]; complement activation, alternative pathway [GO:0006957]; complement activation, classical pathway [GO:0006958]; complement-dependent cytotoxicity [GO:0097278]; complement-mediated synapse pruning [GO:0150062]; fatty acid metabolic process [GO:0006631]; inflammatory response [GO:0006954]; neuron remodeling [GO:0016322]; opsonization [GO:0008228]; oviduct epithelium development [GO:0035846]; positive regulation of activation of membrane attack complex [GO:0001970]; positive regulation of angiogenesis [GO:0045766]; positive regulation of apoptotic cell clearance [GO:2000427]; positive regulation of apoptotic process in another organism [GO:0044533]; positive regulation of developmental growth [GO:0048639]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of G protein-coupled receptor signaling pathway [GO:0045745]; positive regulation of glucose transmembrane transport [GO:0010828]; positive regulation of immune response [GO:0050778]; positive regulation of lipid storage [GO:0010884]; positive regulation of opsonization [GO:1903028]; positive regulation of phagocytosis [GO:0050766]; positive regulation of phagocytosis, engulfment [GO:0060100]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of receptor-mediated endocytosis [GO:0048260]; positive regulation of type IIa hypersensitivity [GO:0001798]; positive regulation of vascular endothelial growth factor production [GO:0010575]; proteolysis [GO:0006508]; regulation of complement activation [GO:0030449]; regulation of complement activation, alternative pathway [GO:0030451]; regulation of triglyceride biosynthetic process [GO:0010866]; response to bacterium [GO:0009617]; vertebrate eye-specific patterning [GO:0150064] |
11441111_During the Arthus response in IgG immune complex-induced lung inflammation, complement component C3 contributes to the influx of polymorphonuclear neutrophils into the bronchoalveolar space by an apparently site-specific mechanism. 11466376_A high concentration of C3 rapidly and efficiently deposited onto the fungal cell surface in the presence of Candida-specific protective antibodies promotes enhanced phagocytosis and fungal killing in experimental blood-borne candidiasis. 11801636_Complement 3 is not essential to the development of a lupus-like phenotype in lpr mice. 11804945_Administration of an estrogenic steroid to immature or ovariectomized adult mice markedly stimulated the expression of Bf, C3, and their RNA messages in the endometrium 11805148_in vivo complement activation is required for aPL antibody-induced fetal loss and growth retardation 12042808_synthesis regulates acute renal graft rejection 12077276_Homozygous C3-deficient mice are highly resistant to collagen-induced arthritis and display a decreased type II collagen-specific IgG antibody response, which can be boosted by repeated immunization. 12097416_The failure of up-regulation of C3 in Il-6 deficient mice contributes to their resistance to induction of antibody-mediated experimental autoimmune myasthenia gravis. 12196286_critical role of complement C and viral evasion of complement in acute, persistent, and latent gamma-herpesvirus infection 12244109_acylation stimulating protein regulation of energy storage may influence energy expenditure and dynamic metabolic balance 12356574_C3 plays a role in airway immune response to particulate matter in mice 12496433_In vivo C3 enhances the development of Th2 cell responses during schistosome infection and limits the production of inflammatory mediators following the systemic antigen insult that occurs when schistosomes are killed. 12517942_C3 regulates the activation and expansion of antigen-specific CD8 T cells during an acute systemic infection with lymphocytic choriomeningitis virus. 12874322_role of complement component 3 in immune clearance and joint histopathology during experimental Lyme borreliosis 12975457_These data indicate that C3a and C5a, two potent inflammatory mediators of the innate immune response, contribute essentially to the early priming stages of hepatocyte regeneration. 14607939_C3 synthesis by myeloid cells, a relatively minor source of complement, provides a critical function during the induction of humoral responses to peripheral herpes simplex virus infection. 14615480_ASP-deficient mice had elevated oxygen consumption (VO2) in both the active (dark) and resting (light) phases of the diurnal cycle 14638805_central mediator of Helicobacter induced inflammation and clearance of gastric Helicobacter in IL-1 defic mice 14699127_C3b and iC3b have roles in reproduction 14703506_Acylation-stimulating protein (ASP) and insulin increased the V(max) of in situ LPL activity, TG synthetic lipoprotein hydrolysis, and nonesterified fatty acid incorporation into adipocytes by 60% and 41%, respectively 15102802_complement activation by the alternative pathway is critical for the survival of mice infected with P. aeruginosa and the protection provided by complement is at least in part due to C3-mediated opsonization and phagocytosis of P. aeruginosa 15192652_C3 is more critical than C5 in controlling S aureus bacteremia. 15547948_traffic of C3 is not dependent on cytoskeletal integrity and requires protein kinase C-dependent phosphorylation 15590640_complement C3 expression is regulated by the bile acid receptor FXR 15714502_Factor C3b was found to be fundamental to the opsonization and phagocytosis of metal particles by monocytes. 16034139_Data clearly show an important role for C3a signaling through the C3a receptor in the development of fatal renal disease in MRL/lpr lupus mice 16127466_Data describe a mouse model of ischemia/reperfusion injury to target complement inhibition to sites of complement activation by linking an iC3b/C3dg-binding fragment of complement receptor 2 (CR2) to a complement-inhibitory protein, Crry. 16210644_Binding of C3d to self-antigens can promote autoimmunity through enhanced antigen retention and presentation by follicular dendritic cells and B cells, respectively. 16299314_Essential function for C3 in host resistance against S. aureus infection was shown. 16304047_C3 deficiency modulates the potency of APCs to stimulate the T-cell response, suggesting a critical role for complement in the maintenance of APC function. 16452172_Bioactive fragments of C3a and C5a are present in drusen of patients with Age-related macular degeneration , and C3a and C5a induce VEGF expression in vitro and in vivo. 16517700_nonproduction of C3 by dendritic cells significantly reduced T cell stimulation and impaired allograft rejection 16547268_Complement component C3 was required for the larval killing process during both protective innate and adaptive immune responses in mice against larval strongyloides stercoralis. 16706788_Antagonizing complement receptor 3 (CR3) decreased phagocytosis by Nramp1 congenic macrophages lines. 16751013_Production of both T-helper cell type 1 (Th1)and Th2 cytokines in response to two independent routes of exposure are impaired in complement component 3-deficient mice. 16778128_results establish C3 activation as the key constituent in complement-related inflammatory tissue injury following stroke and suggest a C3a anaphylatoxin-mediated mechanism 16893077_These results indicate that the adjuvant properties of C3d are associated with CR2 interaction. 17082645_C3-deficiency or inhibition significantly improves neurological outcome as measured by several parameters poststroke. 17237432_Cxcl2 and Cxcl1 are expressed in tubular epithelial cells of postischemic kidneys; This innate immune system recognizes hypoxic injury and triggers a systemic inflammatory response through the generation of C3a and activation of the NF-kappaB system. 17237437_The altered recruitment of leukocytes into the CSF of C3-deficient mice was paralleled by a strong reduction of the brain expression of cytokines and chemokines. 17353050_transfer of wild-type encephalitogenic T cells to C3(-/-) mice resulted in attenuated EAE. C3(-/-) mice with EAE had fewer CD4(+) and CD8(+) T cells in the CNS and 50% fewer of these cells produced IFN-gamma compared to wild-type mice. 17877807_findings show C1q, C3, and C4 are elevated in transgenic SwDI mice in regions rich in fibrillar microvascular A beta 17900537_our data do not support a role for C3 in MPTP-induced dopaminergic neurotoxicity 18004729_Fatty acid-induced ASP resistance via C5L2 may contribute to altered adipose tissue function and obesity/insulin resistance phenotype 18039777_Mouse embryos facilitated such conversion of C3 to C3b, which was taken up by the embryos, resulting in the formation of more blastocysts of larger size. 18063050_The targeted disruption of the murine C5aR gene (C5aR-/- mice) and define the role of the C5aR in a model of Gram-negative bacteremia. 18068164_In the process of spontaneous abortion, the innate immune system through adipsin and complement C3 appears to be influential. 18160458_reduction/deficiency in acylation stimulating protein (ASP) is associated with an antiadipogenic state and ASP may provide a target for controlling fat storage 18192019_C3-fragment containing exosomes derived from OVA loaded antigen presenting cells induce a significantly elevated T cell response in the presence of suboptimal antigen stimulus. 18452991_The results showed that following partial hepatectomy, C3-null mice exhibit reduced hepatic regeneration compared to wildtype mice as assessed by quantification of hepatic cyclin D1 expression and hepatocellular DNA synthesis and mitosis. 18587006_The role of complement C3 and C5 in sepsis in wild-type and C3- or C5-deficient mice was examined. 18615583_Sex steroid hormone-induced ASP resistance via C5L2 may contribute to altered adipose tissue function and insulin resistance phenotype in humans 18624920_The unexpected increase in C3 levels in the APPQ-/- mice coincident with decreased neuropathology provides support for the hypothesis that complement can mediate protective events as well as detrimental events in this disease. 18671285_findings show sex steroids, particularly estradiol, and preimplantation embryos regulate the expression pattern of C3 in the oviduct; data support a physiological role of C3 in preimplantation embryo development 18694897_Results indicate that oligomannose-coated liposomes are opsonized with complement fragment iC3b in a mannose-dependent manner in the peritoneal cavity and then incorporated into peritoneal macrophages via complement receptor type 3. 18713972_iC3b opsonization does not significantly affect apoptotic cell ingestion by DCs; iC3b opsonized apoptotic cells engage CR3, but this interaction is dispensable in mediating the anti-inflammatory effects of apoptotic cells. 18804116_This is the first solution structural study of a large multidomain short complement regulator protein CR2 bound to its physiological ligand C3d. 18836527_Immune complex glomerulonephritis following bone marrow transplantation in C3 deficient mice is reported. 18947875_Crry deficiency in complement sufficient mice: C3 consumption occurs without associated renal injury. 19017934_members of the Staphylococcus aureus extracellular fibrinogen-binding protein family inhibit the interaction of C3d with complement receptor 2 19017980_CRIg thus identifies a subset of tissue resident macrophages capable of increased phagocytosis of complement C3-coated particles, a function critical for immune clearance 19051264_Complement-sufficient & complement-deficient conditions elucidated the important role of complement 3 in the hemolytic anemia characteristically seen in Cd59 deficient mice. 19112490_Increased hepatic inflammation and necrosis correlated with these higher tissue titers and likely contributed to the morbidity in the C3(-/-) mice with mousepox. 19201479_The expression of Pcp4 in naive resting mature B cells is dependent upon tonic stimulation from the CR1/CR2 proteins via a C3 ligand. 19205046_Study shows deficiency in C3 is associated with mild impairment in early pregnancy including longer estrous cycle and higher resorption rates after implantation. 19357704_C3a, in addition to enhancing responsiveness of hematopoietic stem progenitor cells to SDF-1 gradient in a C3aR independent manner, may also directly modulate HSPC homing by augmenting C3aR-mediated secretion of matrix metalloprotease-9 & cell adhesion. 19411110_C3 along the glomerular basement membrane in Cfh(-/-) mice is the C3 fragment iC3b and this is derived from plasma C3 activation. 19648268_C3 activity is required for full activation and expansion of CD8-positive and CD4-positive T cells during Listeria monocytogenes infection. 19684087_contribution of the complement C3a to asthmatic symptoms 19740327_CR2-induced complement activation results in membrane depolarization, as indicated by annexin V binding, with kinetics similar to those of C3-fragment deposition and different from those of membrane attack complex formation. 19752222_Complement 3 deficiency and complement inhibition protect both lean and steatotic livers from warm ischemia reperfusion injury. 19767107_Recombinant C3adesArg/acylation stimulating protein (ASP) is highly bioactive: a critical evaluation of C5L2 binding and 3T3-L1 adipocyte activation. 19815824_important role for complement in brain death-induced heart inflammation and injury 19843088_Robust generation of both C3a and C5a by either the alternative pathway or classical pathway alone were observed with both mouse and human sera, after adherent IgG-induced complement activation. 20213737_These results suggest that C1q and C3 facilitate the induction of intranasal tolerance. 20339115_In the C-reactive protein transgenic mouse model of carotid artery injury, mouse C3 is essential for the effect of human CRP. 20427530_These studies indicate that C3 participates in the induction of acute seizures during viral encephalitis. 20584999_Key role for immune cell-derived C3 in the pathogenesis of murine multiple low-dose streptozotocin-induced diabetes inh mice. 20625009_Mice deficient in the central complement component C3 displayed increased neovascularization in the model of retinopathy of prematurity (ROP) and in the in vivo Matrigel plug assay. 20702808_Complement C3 activation contributes to inflammatory pathways and liver damage in hemorrhagic shock/trauma. 20709903_C3(-/-) bone marrow cells exhibited lower RANKL/OPG expression ratios, produced smaller amounts of macrophage colony-stimulating factor and interleukin-6, and generated significantly fewer osteoclasts than wild-type bone marrow cells. 20712003_Data with immunochemical analyses showed the specific presence of C3 on the visceral yolk sac. 20800648_C3 deficiency can inhibit inflammation through suppressing ASTs activation and TNF-alpha expression, thereby reducing secondary injury and improving neural regeneration and functional recovery after spinal cord injury. 20817881_Through direct interaction, C3a and CpGA synergize to increase IFN-alpha production in a RAGE-dependent manner and stimulate an innate immune response. 20970501_results showed C3 mRNA exhibited biphasic patterns in heart allograft; 1st phase correlated with ischemic reperfusion injury over 2 days post-transplant; 2nd peak was found only in allografts on day 5, concurrent with secretion of IL2 and IFN-gamma 21060153_Genetic ablation of complement C3 attenuates muscle pathology in dysferlin-deficient mice. 21074891_the complement system and its component C3 participate in the regulation of T cell responses, and that complement function is required for normal T helper cell differentiation. 21187447_Although complement receptor (CR)2 is critical, complement 3 ligands are not required for the production of pathogenic autoreactive antibodies. 21270401_Ubiquitous surface protein (Usp)A of Moraxella catarrhalis binds C3 blocking complement activation and inhibiting C3a-mediated inflammation. 21357400_Adenovirus-mediated delivery of C3 to murine RPE induces significant functional and anatomic changes that reproduce many of the features of AMD as well as those of other retinal diseases. 21408070_C3 was required for protection from influenza infection. 21569105_role of C3 in T cell-mediated allergic contact dermatitis, which is a clinical manifestation of contact sensitivity 21704012_heightened production and activation of immune cell-derived complement bypasses the need for CD40/CD154 interactions and implicate antigen-presenting cell-produced C5a and C3a as molecular bridges linking CD4 help to CD8(+) T cells 21707943_C3 and C4 bind to collagen and elastin in the vascular wall and have a potential role in vascular stiffness and atherosclerosis 21767994_Higher number of CD4+CD25+ regulatory T cells (Tregs) with characteristics of expressing Foxp3 was observed in C3-/- mice. These C3-/- Tregs exhibited enhanced suppressive capacity to effector cell proliferation. 21801872_C3 and C5 Complement activation triggers MMP9 metalloproteinases release inducing cervical remodeling and preterm birth in mice 21813769_IgG autoantibodies were responsible for the inhibition, through the blockade of C3 recognition by macrophages 21816131_Complement dysregulation in the absence of CD55 provoked increased C3adesArg production that, in turn, caused altered lipid handling, resulting in atheroprotection and increased adiposity. 21979496_C3 is involved with ventilator-induced lung injury and inhibition of complement activation may be a potential therapeutic strategy. 22184721_IL-4 regulates liver regeneration by controlling complement activation and the subsequent induction of IL-6. 22194859_The complement system plays a pathophysiologic role in the development of pulmonary hypertension in mice. 22219359_C3-dependent microglial priming confers susceptibility to other challenges. 22438044_The results of this study demonstrated that C3 and Mac-1 are involved in phagocytosis and clearance of fAbeta by microglia. 22502635_The mechanism(s) of phagocytosis in the second phase of blood incompatibility reaction required neither C3 nor FcgammaRs. 22518005_Kidney injury accelerates cystogenesis via pathways modulated by heme oxygenase and complement. 22539791_Mechanisms leading to the immunoglobulin (Ig)E-mediated late-phase asthmatic response and airway hyperresponsiveness are closely associated with neutrophilic inflammation through the production of IL-17 induced by complement C3a. 22649195_Significantly reduced uptake of beta-glucan particles by peritoneal monocytes/ macrophages is observed in C3-deficient mice. 22721768_A non-classical pathway of C3 activation is involved in axotomy-induced adult mice synapse removal, and its inhibition promotes functional recovery. 22797180_C3a and C5a promote renal ischemia-reperfusion injury. 23033813_the expressions of C5a and CD88 can be inhibited in different degrees after SCI when the activation of complement system is blocked through C3 deficiency. 23056509_chronic ASP administration in C3(-/-) mice would affect lipid metabolism and insulin sensitivity via an adaptive adipose tissue inflammatory response 23074214_Mice lacking C3aR and/or C5aR developed early onset and progressive retinal degeneration, accompanied by cleaved caspase-3 upregulation. 23097680_Studied whether increased levels of complement in HD brains contributed to disease progression in the R6/2 mouse model of HD. Found C3 deficiency does not alter HD progression in the R6/2 mouse model. 23168409_PPARalpha positively regulates complement C3 expression but inhibits tumor necrosis factor alpha-mediated activation of C3 gene in hepatic cells. 23182714_Data indicate complement C3 deposition on peanut extract (PE) was abolished in immunoglobulin- and C4-deficient sera. 23237573_Data indicate that absence of complement C3, complement C5 and Fc receptor subunit gamma (FcRgamma) does not impact autoantibody production in K/BxN mice. 23299310_Hepatic stellate cells deficient in C3 are impaired in their ability to induce myeloid-derived suppressor cells. 23430572_in vitro results suggest various metabolic hormones and inflammatory factors can affect acylation stimulating protein (ASP) production and that ASP could constitute a new link between adipocytes and macrophage 23526211_ALI in H5N1-infected mice was caused by excessive complement activation, as demonstrated by deposition of C3, C5b-9, and MBL-C in lung tissue, and by up-regulation of MBL-associated serine protease-2 and the complement receptors C3aR and C5aR. 23549917_Chitin induces the generation of C3a in the lung, and chitin-dependent allergic sensitization to Aspergillus requires C3aR signaling, which suppresses regulatory dendritic cells and T cells and induces allergy-promoting T cells. 23619360_analysis of complement activation using monoclonal antibodies against C3d 23926185_C3 induces epithelial-to-mesenchymal transition and may be a primary factor to activate the renal renin-angiotensin systems to induce hypertension. 24038599_Complement protein C3 exacerbates prion disease in a mouse model of chronic wasting disease. 24175013_Radiation combined injury-induced alterations of corticosterone, CRP, C3, IgM, and PGE2 cause homeostatic imbalance and may contribute to reduced survival. 24200836_have identified Complement C3 as a candidate plasma biomarker for measuring disease state in neuroblastoma 24260573_Complement C3 and C5 deficiency affects bone fracture healing. 24279761_Data indicate that co-deficiency of factor H (FH) and MASP-1/MASP-3 did not ameliorate either the plasma Complement C3 (C3) activation or glomerular C3 accumulation in FH-deficient mice. 24378428_The results of this study suggested that C3 plays a role in the regulation of the number and function of glutamatergic synapses in the hippocampus and exerts negative effects on hippocampus-dependent cognitive performance. 24470120_These results suggest that osteoclast-derived C3a functions in the relay from bone resorption to formation and may be a candidate for a coupling factor. 24632830_a role for outside-in iC3b-CD11b signals in limiting intrinsic organ inflammation 24667943_Coagulation cascades, by generating thrombin and plasmin, respectively, provide C5 convertase activity, explaining why mobilization of hematopoietic stem cells in C3-deficient mice. 24763049_C3 secreted from mycoplasma-infected Mesenchymal stem cells (MSCs) has an important role in the immunomodulatory functions of MSCs. 24808362_Genetic and intervention studies implicating complement C3 as a major target for the treatment of periodontitis. 24948809_Loss of dopaminergic neurons was rescued by complement C3-deficient mice. 24981453_A previously unknown function of C3a promotes protection to both myeloid and lymphoid cells against Listeria monocytogenes-induced apoptosis. 25187659_These results provide new insights into the role of systemic C3 in regulating contraction following intracellular bacterial infection and may help to develop vaccines that are more effective. 25573852_Identify complement 5a as the major C3 activation product that was involved in macrophage polarization and DOCA-salt-induced vascular injury. 25733354_Data indicate that for all three pathways there was a strong correlation between the amount of Ccomplement C3 (C3) fragments and the reduction in functional complement activity. 25802332_Complement component 3 is up-regulated via non-canonical TGF-beta signaling in the retinal pigment epithelium 25851247_Carboxypeptidase B2 deficiency reveals that complement C3a limits infection in a murine polymicrobial sepsis model. 25888260_Therapeutic efficacy of radiotherapy depends on C3a and C5a. 26132276_our findings define the mechanism of receptor crosstalk between CR3 and Dectin-1 and demonstrate the importance of their collaboration in host defense against fungal infection. 26199322_Study shows that C3a is locally produced by -retinal pigment epithelial cells suggesting a key role in early macular degenerations pathogenesis. 26787717_The roles of C3, CfB, and C3a receptor in the severity of S. aureus induced septic arthritis are reported. 26822321_This study provideed the evidence that C3a plays a critical role in cerebral endothelial activation and leukocyte recruitment during inflammation in the brain. 26873964_Demonstrate that cardiac Sirt1 plays an essential role in caloritc restriction-induced cardioprotection against myocardial I/R injury by suppressing cardiac C3 expression. 26900812_Complement C3 activation occurs following intramuscular islet transplantation, but it does not seem to affect the graft function. 26945067_These results suggest that GAS utilizes diverse mechanisms to degrade C3b and thus to protect bacterial cells from the complement response of the host. 27029069_Data show that months after irradiation (IR) complement component 3 (C3-/-) mice made fewer errors than WT mice in a reversal learning test indicating better learning capacity in C3-/- mice after IR. 27041467_Time-lapse video microscopy established the localization of the complement anaphylatoxin C3a and its receptor on enteric neural crest cells during their migration in the embryonic gut. C3a plays a role in regulating collective and directional cell migration, and in ganglia network organization during enteric nervous system ontogenesis. It regulates cell migration in a N-cadherin-dependent process. 27235854_We induced anti-myeloperoxidase vasculitis in mice and confirmed a role for complement activation by demonstrating protection in C3-deficient mice. 27274010_Wild-type C57BL/6 mice with pristane-induced lupus developed a strong IFN signature, which was absent in immunoglobulin-deficient (muMT), C3(-/-) , and CD18(-/-) mice. In vivo phagocytosis of dead cells was impaired in C3-deficient mice. 27564415_Study show s the regulation of C3 Activation by the Alternative Complement Pathway in the Mouse Retina 27572255_These data indicated that alpha7-nAChR caused the inhibition of ASPinduced activation of p38 kinase and NFkappa B to inhibit the production of MCP1 and keratinocytederived chemokine. 27641978_C3 deficiency can prolong MHC-II molecule mismatched skin allograft survival. 27735945_nutrient sensing in the liver is coupled to release of C3 and potentially its metabolic and inflammatory functions. 28052876_Demonstrate local synthesis of complement proteins by both PDGFRbeta-positive pericytes and CD45-positive cells in kidney fibrosis. 28139294_C5 and C5aR have critical roles in the development of C3 glomerulopathy. 28279023_Data report C3 as a novel myogenic factor secreted by undifferentiated preadipocyte that enhances myogenic differentiation of fetal progenitor cells and adult cells. The results show that complement C3 molecule internalizes myogenic and adipogenic precursor cells and then promotes their differentiation. 28283064_Study found that cancer-cell-derived C3 activates the C3a receptor in the choroid plexus epithelium to disrupt the blood-cerebrospinal fluid (CSF) barrier. This effect allows plasma components, including amphiregulin, and other mitogens to enter the CSF and promote cancer cell growth. 28348233_C3 role in the retinal epithelium and photoreceptor degeneration 28446616_These findings suggest that C3 protects from early glaucomatous damage, a process that may involve EGFR signaling and other immune responses in the optic nerve head. 28462915_It has been shown that key proteins in the lectin arm of this pathway, MASP1, MASP2 and C3, are expressed in the developing cortex and that neuronal migration is impaired in knockout and knockdown mice. 28566429_complement C3 or downstream complement activation fragments may play an important role in Abeta plaque pathology. 28605809_Retinal C3 was expressed by microglia/macrophages located in the outer retina in AMD eyes. In rodent photo-oxidative damage, C3-expressing microglia/macrophages and complement activation were located in regions of lesion expansion in the outer retina over 2 months 28710441_IgG3-antigen complexes are deposited on follicular dendritic cells in the presence of C1q and C3. 28928904_These findings shed light on mechanisms of age-related retinal alterations by identifying C3 as a potential therapeutic target for retinal aging. 29018286_C3 reduces neurite outgrowth and neuronal viability in vitro and restricts axon regeneration in vivo. 29118090_Complement C3 role in the lung cancer progression. 29383821_Both carboxypeptidase B2 and carboxypeptidase N have roles in inactivating complement C3a. 29565179_MUC5B promoter variant is associated with higher C3 expression and suggest that the complement system may contribute to the pathogenesis of idiopathic pulmonary fibrosis. 29942300_These results suggest that C3 possesses a unique ability to actually suppress Ab formation following transfusion by reducing the availability of the target antigen on the red blood cell surface. 30068650_The findings in this study suggest that C3 is required for effective induction of humoral and cellular adaptive immune responses as well as protective immunity after nonneutralizing influenza M2e vaccination. 30179254_these data show that C3 plays an essential role in IgG2a-mediated host resistance to Salmonella in vivo 30217731_Our study uncovers a damaging effect of complement C3 or downstream complement activation in glaucoma, and it establishes AAV2.CR2-Crry as a viable therapeutic strategy to target pathogenic C3-mediated complement activation in the glaucomatous retina 30246940_These results demonstrate that high glucose induces C3 up-regulation via RAGE- p38MAPK-NF-kappaB signalling in vivo and in vitro, which might be associated with synaptic protein loss. 30714990_data provide in vivo modeling evidence that gain-of-function changes in complement C3 drive Atypical hemolytic uremic syndrome. They also show that long-term C5 deficiency is not accompanied by development of other renal complications (such as C3 glomerulopathy) despite sustained dysregulation of C3. 31292459_The astrocyte transcriptome in EAE optic neuritis shows complement activation and reveals a sex difference in astrocytic C3 expression. 31433986_Complement C3 Is Activated in Human AD Brain and Is Required for Neurodegeneration in Mouse Models of Amyloidosis and Tauopathy. 31461345_Complement C3 exacerbates renal interstitial fibrosis by facilitating macrophage M1 polarization, promoting proinflammatory cytokine expression, and deteriorating peritubular capillary rarefaction in the kidney. 31723045_Complement C3 mediates the capture of Trypanosoma congolense parasites by Kupffer cells, preventing the early mortality of infected mice. 31865842_Endothelial progenitor cell transplantation alleviated ischemic brain injury via inhibiting C3/C3aR pathway in mice. 31955888_Complement component C3 and the TLR co-receptor CD14 are not involved in angiotensin II induced cardiac remodelling. 32208992_C3 was important in forming and maintaining this undifferentiated state of smooth muscle cells from mesenchymal stem cells to generate renin with increases in transcription factor LXRalpha and KLF5 32244854_Complement Component C3 Participates in Early Stages of Niemann-Pick C Mouse Liver Damage. 32535539_Complement component 3 prevents imiquimod-induced psoriatic skin inflammation by inhibiting apoptosis in mice. 32582142_Fc Gamma Receptors and Complement Component 3 Facilitate Anti-fVIII Antibody Formation. 32682912_C3 Drives Inflammatory Skin Carcinogenesis Independently of C5. 33088172_Ablation of C3 modulates macrophage reactivity in the outer retina during photo-oxidative damage. 33161108_Protection of hUC-MSCs against neuronal complement C3a receptor-mediated NLRP3 activation in CUMS-induced mice. 33337564_Deficiency of complement component 3 may be linked to the development of constipation in FVB/N-C3(em1Hlee) /Korl mice. 33746963_Complement and Chlamydia psittaci: Non-Myeloid-Derived C3 Predominantly Induces Protective Adaptive Immune Responses in Mouse Lung Infection. 33767691_Complement and Chlamydia psittaci: Early Complement-Dependent Events Are Important for DC Migration and Protection During Mouse Lung Infection. 33782126_miR-218-2 regulates cognitive functions in the hippocampus through complement component 3-dependent modulation of synaptic vesicle release. 34071589_C3 Deficiency Leads to Increased Angiogenesis and Elevated Pro-Angiogenic Leukocyte Recruitment in Ischemic Muscle Tissue. 34176310_Role of Complement Component 3 in Early Erythrolysis in the Hematoma After Experimental Intracerebral Hemorrhage. 34309810_Complement C3 Aggravates Post-epileptic Neuronal Injury Via Activation of TRPV1. 34403734_Dissociable effects of complement C3 and C3aR on survival and morphology of adult born hippocampal neurons, pattern separation, and cognitive flexibility in male mice. 34415487_Complement Component C3 Loss leads to Locomotor Deficits and Altered Cerebellar Internal Granule Cell In Vitro Synaptic Protein Expression in C57BL/6 Mice. 34417332_Retinal Ganglion Cell Axon Regeneration Requires Complement and Myeloid Cell Activity within the Optic Nerve. 34417985_Proteome Profile of Trigeminal Ganglion in Murine Model of Allergic Contact Dermatitis: Complement 3 Pathway Contributes to Itch and Pain Sensation. 34543680_Complement C3 and C3aR mediate different aspects of emotional behaviours; relevance to risk for psychiatric disorder. 34707103_Lung mesenchymal stromal cells influenced by Th2 cytokines mobilize neutrophils and facilitate metastasis by producing complement C3. 34762761_Intracellular C3 prevents hepatic steatosis by promoting autophagy and very-low-density lipoprotein secretion. 34875452_Mesenchymal stem cell therapy attenuates complement C3 deposition and improves the delicate equilibrium between angiogenic and anti-angiogenic factors in abortion-prone mice. 35143312_Marginal zone B cells acquire dendritic cell functions by trogocytosis. 35250972_Reduced Neutrophil Extracellular Trap Formation During Ischemia Reperfusion Injury in C3 KO Mice: C3 Requirement for NETs Release. 35432387_Intracellular Complement Component 3 Attenuated Ischemia-Reperfusion Injury in the Isolated Buffer-Perfused Mouse Heart and Is Associated With Improved Metabolic Homeostasis. |
ENSG00000125730 |
C3 |
164.499230 |
6.1056630196 |
2.610148 |
0.25504742 |
100.956603 |
0.00000000000000000000000940209645248346263031283448762847722248426635962074163321783380319746070696851347747724503278732299804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000004757898111768374807975625520935708380502378676647860134441437446328748706037004012614488601684570312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
246.853637790469 |
43.8677897060131 |
40.4302754669331 |
5.82294483085854 |
| ENSMUSG00000024168 |
407831 |
Tmem204 |
protein_coding |
Q7TQI0 |
FUNCTION: Can influence paracellular permeability. Appears to be involved in cell-cell interactions through adherens (By similarity). {ECO:0000250}. |
Cell junction;Cell membrane;Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:407831; |
adherens junction [GO:0005912]; plasma membrane [GO:0005886]; lymph vessel development [GO:0001945]; regulation of vascular endothelial growth factor receptor signaling pathway [GO:0030947]; smooth muscle cell differentiation [GO:0051145] |
20439428_CLP24 is a novel regulator of VEGFR-2 and VEGFR-3 signaling pathways and of normal lymphatic vessel structure |
ENSG00000131634 |
TMEM204 |
64.550643 |
0.2583783093 |
-1.952443 |
0.36280802 |
28.396010 |
0.00000009886886442411901160237105252656664156063470727531239390373229980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000010407536578494683553479036031141902185481740161776542663574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.6819487881383 |
6.52676991024497 |
99.3966902929506 |
17.4516959614936 |
| ENSMUSG00000024210 |
271424 |
Ip6k3 |
protein_coding |
Q8BWD2 |
FUNCTION: Converts inositol hexakisphosphate (InsP6) to diphosphoinositol pentakisphosphate (InsP7/PP-InsP5). Converts 1,3,4,5,6-pentakisphosphate (InsP5) to PP-InsP4 (By similarity). {ECO:0000250}. |
ATP-binding;Cytoplasm;Kinase;Nucleotide-binding;Reference proteome;Transferase |
|
|
mmu:271424; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; ATP binding [GO:0005524]; inositol hexakisphosphate 1-kinase activity [GO:0052723]; inositol hexakisphosphate 3-kinase activity [GO:0052724]; inositol hexakisphosphate 5-kinase activity [GO:0000832]; inositol hexakisphosphate 6-kinase activity [GO:0000831]; inositol hexakisphosphate kinase activity [GO:0000828]; inositol-1,3,4,5,6-pentakisphosphate kinase activity [GO:0000827]; kinase activity [GO:0016301]; inositol phosphate biosynthetic process [GO:0032958]; locomotion [GO:0040011]; phosphatidylinositol metabolic process [GO:0046488]; phosphatidylinositol phosphate biosynthetic process [GO:0046854]; protein phosphorylation [GO:0006468] |
26245967_IP6K3 is highly expressed in cerebellar Purkinje cells. IP6K3-deleted mice display defects of motor learning and coordination. IP6K3-null mice manifest aberrations of Purkinje cells with a diminished number of synapses. 27577108_High levels of IP6K3 mRNA were found in myotubes and muscle tissues. Expression was elevated under diabetic, fasting, and disuse conditions in mouse skeletal muscles. Ip6k3(-/-) mice had lower blood glucose, less insulin, decreased fat, lower weight, increased plasma lactate, enhanced glucose tolerance, lower glucose during an insulin tolerance test, and reduced muscle Pdk4 expression. Ip6k3 deletion extended lifespan. |
ENSG00000161896 |
IP6K3 |
27.035170 |
0.4185475632 |
-1.256537 |
0.50074894 |
6.287021 |
0.01216253145004387963001857286826634663157165050506591796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0396871634633822537030489741027849959209561347961425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.3064903620312 |
5.77419432094037 |
38.9597068420935 |
9.72702580168441 |
| ENSMUSG00000024228 |
67993 |
Nudt12 |
protein_coding |
Q9DCN1 |
FUNCTION: mRNA decapping enzyme that specifically removes the nicotinamide adenine dinucleotide (NAD) cap from a subset of mRNAs by hydrolyzing the diphosphate linkage to produce nicotinamide mononucleotide (NMN) and 5' monophosphate mRNA (PubMed:31101919, PubMed:32432673). The NAD-cap is present at the 5'-end of some RNAs; in contrast to the canonical N7 methylguanosine (m7G) cap, the NAD cap promotes mRNA decay (PubMed:31101919). Preferentially acts on NAD-capped transcripts in response to nutrient stress (PubMed:31101919). Also acts on free nicotinamide adenine dinucleotide molecules: hydrolyzes NAD(H) into NMN(H) and AMP, and NADPH into NMNH and 2',5'-ADP (By similarity). May act to regulate the concentration of peroxisomal nicotinamide nucleotide cofactors required for oxidative metabolism in this organelle (By similarity). Regulates the levels of circadian clock components PER1, PER2, PER3 and CRY2 in the liver (PubMed:31875550). {ECO:0000250|UniProtKB:Q9BQG2, ECO:0000269|PubMed:31101919, ECO:0000269|PubMed:31875550}. |
3D-structure;Alternative splicing;ANK repeat;Cytoplasm;Hydrolase;Magnesium;Metal-binding;NAD;NADP;Peroxisome;Reference proteome;Repeat;Zinc |
|
|
mmu:67993; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; peroxisome [GO:0005777]; magnesium ion binding [GO:0000287]; NAD+ diphosphatase activity [GO:0000210]; NADH pyrophosphatase activity [GO:0035529]; phosphodiesterase decapping endonuclease activity [GO:1990174]; RNA NAD-cap (NMN-forming) hydrolase activity [GO:0110153]; zinc ion binding [GO:0008270]; circadian regulation of gene expression [GO:0032922]; methylguanosine-cap decapping [GO:0110156]; mRNA catabolic process [GO:0006402]; NAD catabolic process [GO:0019677]; NAD-cap decapping [GO:0110155]; NADH metabolic process [GO:0006734]; NADP catabolic process [GO:0006742] |
31875550_Decapping Enzyme NUDT12 Partners with BLMH for Cytoplasmic Surveillance of NAD-Capped RNAs. |
ENSG00000112874 |
NUDT12 |
66.037788 |
2.3824862051 |
1.252468 |
0.37290346 |
11.163476 |
0.00083423490763864566101964959088377327134367078542709350585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0039267818643856150159776596808569593122228980064392089843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
90.9040586759939 |
23.7591182017214 |
38.1551248787682 |
7.65385408806199 |
| ENSMUSG00000024247 |
106522 |
Pkdcc |
protein_coding |
Q5RJI4 |
FUNCTION: Secreted tyrosine-protein kinase that mediates phosphorylation of extracellular proteins and endogenous proteins in the secretory pathway, which is essential for patterning at organogenesis stages. Mediates phosphorylation of MMP1, MMP13, MMP14, MMP19 and ERP29 (PubMed:25171405). May also have serine/threonine protein kinase activity (PubMed:25171405). Required for longitudinal bone growth through regulation of chondrocyte differentiation (PubMed:19097194, PubMed:23792766). May be indirectly involved in protein transport from the Golgi apparatus to the plasma membrane (PubMed:19465597). Probably plays a role in platelets: rapidly and quantitatively secreted from platelets in response to stimulation of platelet degranulation (PubMed:25171405). {ECO:0000269|PubMed:19097194, ECO:0000269|PubMed:19465597, ECO:0000269|PubMed:23792766, ECO:0000269|PubMed:25171405}. |
Alternative splicing;ATP-binding;Developmental protein;Differentiation;Glycoprotein;Golgi apparatus;Kinase;Nucleotide-binding;Osteogenesis;Phosphoprotein;Protein transport;Reference proteome;Secreted;Signal;Transferase;Transport;Tyrosine-protein kinase |
|
|
mmu:106522; |
extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; ATP binding [GO:0005524]; non-membrane spanning protein tyrosine kinase activity [GO:0004715]; protein kinase activity [GO:0004672]; bone mineralization [GO:0030282]; cell differentiation [GO:0030154]; embryonic digestive tract development [GO:0048566]; limb morphogenesis [GO:0035108]; lung alveolus development [GO:0048286]; multicellular organism growth [GO:0035264]; negative regulation of Golgi to plasma membrane protein transport [GO:0042997]; peptidyl-tyrosine phosphorylation [GO:0018108]; positive regulation of bone mineralization [GO:0030501]; positive regulation of chondrocyte differentiation [GO:0032332]; protein transport [GO:0015031]; roof of mouth development [GO:0060021]; skeletal system development [GO:0001501] |
19097194_These data indicate that Pkdcc encodes a protein kinase that is required for the appropriate timing of flat proliferative chondrocytes differentiation. 19465597_Vlk is a novel vertebrate-specific PK that is involved in the regulation of the rate of protein export from the Golgi, thereby playing an important role in the formation of functional stroma by mesenchymal cells. 21553379_Adtk1 null mutants are smaller and present short limbs due to decreased mineralization, suggesting a potential role in chondrogenesis during limb development. 23792766_VLK plays a role in the Indian hedgehog /GLI3 interactions and that Vlk and Gli3 cooperate to regulate long bone development 25171405_Study shows that VLK, a putative protein kinase previously shown to be essential in embryonic development, is a secreted protein kinase, with preference for tyrosine, that phosphorylates a broad range of secreted and endoplasmic reticulum-resident substrate proteins. 31845979_Taken together, these results suggest that Vlk may function as a signaling regulator in extracellular space to modulate the Hedgehog pathway 34329392_The secreted tyrosine kinase VLK is essential for normal platelet activation and thrombus formation. |
ENSG00000162878 |
PKDCC |
1162.415775 |
2.8886793656 |
1.530410 |
0.10315864 |
214.430818 |
0.00000000000000000000000000000000000000000000000148331626906495668364750077872505389721084822578210496713047227831489564654999176520220343836830085496644221512429175693090557808240248505171621218323707580566406250000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000002353528480249731324056116998724769236226129640077928896592639838738262505491166616585728441520923251983787989555983689513030121531755867181345820426940917968750000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1734.4866257912 |
124.128649461578 |
600.442765117303 |
33.0898735922983 |
| ENSMUSG00000024261 |
20983 |
Syt4 |
protein_coding |
P40749 |
FUNCTION: Synaptotagmin family member which does not bind Ca(2+). Plays a role in dendrite formation by melanocytes (By similarity). {ECO:0000250|UniProtKB:P50232, ECO:0000250|UniProtKB:Q9H2B2}.; FUNCTION: Synaptotagmin family member which does not bind Ca(2+) (By similarity). Involved in neuronal dense core vesicles (DCVs) mobility through its interaction with KIF1A. Upon increased neuronal activity, phosphorylation by MAPK8/JNK1 destabilizes the interaction with KIF1A and captures DCVs to synapses (PubMed:29166604). Plays a role in dendrite formation by melanocytes (By similarity). {ECO:0000250|UniProtKB:P50232, ECO:0000250|UniProtKB:Q9H2B2, ECO:0000269|PubMed:29166604}. |
Calcium;Cytoplasmic vesicle;Differentiation;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Transmembrane;Transmembrane helix |
|
The protein encoded by this gene belongs to the synaptotagmin family. Members of this family are multi-domained, integral membrane proteins of synaptic vesicles, and are thought to serve as Ca2+ sensors in the process of vesicular trafficking and exocytosis. This gene is primarily expressed in the nervous tissues. [provided by RefSeq, Jul 2008]. |
mmu:20983; |
astrocyte projection [GO:0097449]; axon [GO:0030424]; dendrite [GO:0030425]; dense core granule [GO:0031045]; exocytic vesicle [GO:0070382]; glutamatergic synapse [GO:0098978]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; microvesicle [GO:1990742]; neuron projection [GO:0043005]; neuron projection terminus [GO:0044306]; neuronal cell body [GO:0043025]; neuronal dense core vesicle [GO:0098992]; neuronal dense core vesicle membrane [GO:0099012]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; postsynapse [GO:0098794]; presynapse [GO:0098793]; secretory granule membrane [GO:0030667]; somatodendritic compartment [GO:0036477]; synapse [GO:0045202]; vesicle [GO:0031982]; calcium ion binding [GO:0005509]; calcium-dependent phospholipid binding [GO:0005544]; clathrin binding [GO:0030276]; phosphatidylserine binding [GO:0001786]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; SNARE binding [GO:0000149]; syntaxin binding [GO:0019905]; syntaxin-1 binding [GO:0017075]; syntaxin-3 binding [GO:0030348]; brain development [GO:0007420]; calcium-ion regulated exocytosis [GO:0017156]; cell differentiation [GO:0030154]; cellular response to calcium ion [GO:0071277]; dense core granule cytoskeletal transport [GO:0099519]; memory [GO:0007613]; negative regulation of calcium ion-dependent exocytosis [GO:0045955]; negative regulation of catecholamine secretion [GO:0033604]; negative regulation of dense core granule exocytosis [GO:1905414]; negative regulation of neurotransmitter secretion [GO:0046929]; negative regulation of protein secretion [GO:0050709]; negative regulation of retrograde trans-synaptic signaling by neuropeptide [GO:1905433]; negative regulation of short-term neuronal synaptic plasticity [GO:0048174]; negative regulation of synaptic vesicle exocytosis [GO:2000301]; negative regulation of vesicle fusion [GO:0031339]; neurotransmitter secretion [GO:0007269]; positive regulation of dendrite extension [GO:1903861]; positive regulation of dense core granule exocytosis [GO:1905415]; positive regulation of glutamate secretion [GO:0014049]; regulation of calcium ion-dependent exocytosis [GO:0017158]; regulation of dopamine secretion [GO:0014059]; regulation of endocytosis [GO:0030100]; regulation of postsynaptic dense core vesicle exocytosis [GO:0150044]; regulation of trans-synaptic signaling by BDNF, modulating synaptic transmission [GO:0150035]; regulation of vesicle fusion [GO:0031338]; secretory granule maturation [GO:0061792]; synaptic vesicle transport [GO:0048489]; vesicle fusion [GO:0006906]; vesicle fusion with vesicle [GO:0061782]; vesicle-mediated transport [GO:0016192] |
15197251_propose that Syt IV is essential for the regulated release of glutamate from astrocytes and is a candidate Ca2+ sensor for gliotransmission by these nonneuronal cells 15380377_Syt IV plays a role in rodent mood-related behavior by regulating synaptic function in the neuronal networks that modulate these behaviors. 15390175_Syt IV is a presynaptic negative regulator of short-term plasticity in area CA1 of the hippocampus and is required for some, but not all, forms of hippocampus-dependent memory. 19136969_Given the neuroendocrine functions of the posterior pituitary, changes in Syt IV levels could be involved in endocrine transitions involving alterations in the release of the neuropeptides oxytocin and vasopressin 19448629_Thus, regulation of BDNF secretion by syt-IV emerges as a mechanism for maintaining synaptic strength in a useful range during LTP. 20010821_From the results of this study the author proposed that the differential expression of synaptotagmins determines the characteristic Ca2+ sensitivity of vesicle fusion at hair cell synapses. 20138128_these data suggest that syt IV plays a role both in the Golgi and in the maintenance of normal numbers of synaptic vesicles in presynaptic terminals 21576241_Somatodendritic dopamine release requires synaptotagmin 4 and 7 and the participation of voltage-gated calcium channels. 22514304_BDNF secretion mediated by syt-IV is subject to extensive temporal and spatial control to modulate the spatial pattern of synaptic efficacy in networks of neurons 29656931_Syt4 Overexpression Represses Basal Insulin Secretion and Impairs Islet Morphogenesis. 31743468_Synaptotagmin-4 promotes dendrite extension and melanogenesis in alpaca melanocytes by regulating Ca(2+) influx via TRPM1 channels. 32691536_Exocytosis in mouse vestibular Type II hair cells shows a high-order Ca(2+) dependence that is independent of synaptotagmin-4. |
ENSG00000132872 |
SYT4 |
21.219104 |
0.2341635137 |
-2.094412 |
0.74821660 |
7.494794 |
0.00618775978589243373650941038022210705094039440155029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0222694261876946013323230744163083727471530437469482421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.91741927181756 |
4.29273018198595 |
33.8114984119616 |
12.6880545372275 |
| ENSMUSG00000024268 |
108013 |
Celf4 |
protein_coding |
Q7TSY6 |
FUNCTION: RNA-binding protein implicated in the regulation of pre-mRNA alternative splicing. Mediates exon inclusion and/or exclusion in pre-mRNA that are subject to tissue-specific and developmentally regulated alternative splicing. Specifically activates exon 5 inclusion of cardiac isoforms of TNNT2 during heart remodeling at the juvenile to adult transition. Promotes exclusion of both the smooth muscle (SM) and non-muscle (NM) exons in actinin pre-mRNAs. Activates the splicing of MAPT/Tau exon 10. Binds to muscle-specific splicing enhancer (MSE) intronic sites flanking the alternative exon 5 of TNNT2 pre-mRNA (By similarity). {ECO:0000250}. |
Activator;Alternative splicing;Cytoplasm;mRNA processing;mRNA splicing;Nucleus;Reference proteome;Repeat;RNA-binding |
|
|
mmu:108013; |
cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; postsynapse [GO:0098794]; ribonucleoprotein complex [GO:1990904]; BRE binding [GO:0042835]; mRNA binding [GO:0003729]; pre-mRNA binding [GO:0036002]; RNA binding [GO:0003723]; alternative mRNA splicing, via spliceosome [GO:0000380]; excitatory postsynaptic potential [GO:0060079]; in utero embryonic development [GO:0001701]; mRNA splice site selection [GO:0006376]; negative regulation of excitatory postsynaptic potential [GO:0090394]; negative regulation of mRNA splicing, via spliceosome [GO:0048025]; positive regulation of mRNA splicing, via spliceosome [GO:0048026]; regulation of alternative mRNA splicing, via spliceosome [GO:0000381]; regulation of retina development in camera-type eye [GO:1902866] |
12438720_cloning and characterization 15988035_CELF protein activity is required for normal alternative splicing in the heart in vivo and normal CELF-mediated alternative splicing regulation is in turn required for normal cardiac function. 17677002_Brunol4 deficiency in mice results in a complex seizure phenotype, likely due to the coordinate dysregulation of several molecules, providing a unique new animal model of epilepsy 21745337_Data point strongly to the conclusion that Celf4 deletion primarily from excitatory neurons promotes convulsive seizure phenotypes. 23209433_we suggest that CELF4 deficiency leads to abnormal neuronal function by combining a specific effect on neuronal excitation with a general impairment of synaptic transmission. 23932931_Celf4 might be regulating alternative early in retinal development. 35306363_CELF4 regulates spine formation and depression-like behaviors of mice. |
ENSG00000101489 |
CELF4 |
134.380623 |
0.4637238601 |
-1.108662 |
0.36004894 |
8.917383 |
0.00282468986293627497344282950564320344710722565650939941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0114156741820762384215237617013372073415666818618774414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
78.7606460250783 |
19.8607287414809 |
169.843850617724 |
31.6089544645408 |
| ENSMUSG00000024301 |
16580 |
Kifc5b |
protein_coding |
E9PUA5 |
Human_homologues FUNCTION: Minus end-directed microtubule-dependent motor required for bipolar spindle formation (PubMed:15843429). May contribute to movement of early endocytic vesicles (By similarity). Regulates cilium formation and structure (By similarity). {ECO:0000250|UniProtKB:Q9QWT9, ECO:0000269|PubMed:15843429}. |
ATP-binding;Coiled coil;Microtubule;Motor protein;Nucleotide-binding;Proteomics identification;Reference proteome |
|
|
mmu:16580; |
kinesin complex [GO:0005871]; microtubule [GO:0005874]; microtubule organizing center [GO:0005815]; mitotic spindle [GO:0072686]; nucleus [GO:0005634]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule-based movement [GO:0007018]; mitotic metaphase plate congression [GO:0007080]; mitotic spindle assembly [GO:0090307] |
16638812_KIFC5A and the Nubp proteins in a common regulatory pathway involved in the control of centrosome duplication in mammalian cells. |
ENSG00000237649 |
KIFC1 |
367.741306 |
2.0522297990 |
1.037192 |
0.16751280 |
38.145911 |
0.00000000065647153357089289951149962580112875998317178982688346877694129943847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000094422798860735038253640910945775621687658940572873689234256744384765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
490.488637164542 |
57.0791908749294 |
239.002784869545 |
21.3189031048557 |
| ENSMUSG00000024338 |
16913 |
Psmb8 |
protein_coding |
P28063 |
FUNCTION: The proteasome is a multicatalytic proteinase complex which is characterized by its ability to cleave peptides with Arg, Phe, Tyr, Leu, and Glu adjacent to the leaving group at neutral or slightly basic pH. The proteasome has an ATP-dependent proteolytic activity. This subunit is involved in antigen processing to generate class I binding peptides. May participate in the inflammatory response pathway. Required for adipocyte differentiation (PubMed:21881205, PubMed:22341445, PubMed:8066463). May be involved in the generation of spliced peptides resulting from the ligation of two separate proteasomal cleavage products that are not contiguous in the parental protein (By similarity). {ECO:0000250|UniProtKB:P28062, ECO:0000269|PubMed:21881205, ECO:0000269|PubMed:22341445, ECO:0000269|PubMed:8066463}. |
3D-structure;Cytoplasm;Differentiation;Hydrolase;Immunity;Nucleus;Protease;Proteasome;Reference proteome;Threonine protease;Zymogen |
|
|
mmu:16913; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; proteasome complex [GO:0000502]; proteasome core complex [GO:0005839]; proteasome core complex, beta-subunit complex [GO:0019774]; spermatoproteasome complex [GO:1990111]; endopeptidase activity [GO:0004175]; threonine-type endopeptidase activity [GO:0004298]; antigen processing and presentation [GO:0019882]; fat cell differentiation [GO:0045444]; proteasomal protein catabolic process [GO:0010498]; regulation of endopeptidase activity [GO:0052548] |
12220526_The presence of immuno subunits in nonimmune tissues such as lens suggests that the immunoproteasome may also have nonimmune functions, such as that in lens differentiation. 15356141_Immunoproteasome LMP7 can at least in part determine subdominance and shape the epitope hierarchy of cytotoxic T cell responses in vivo. 16002717_Immunoproteasome LMP7 may affect CD8+ T cell responses to only a limited number of viral epitopes; its main biological function may lie elsewhere. 16547243_phenotype of hyperproliferation of T cells lacking both Mecl1 and LMP7 implicates a specific role for immunoproteasomes in T cell proliferation that is not obviously connected to MHC class I Ag processing 17079320_These results demonstrate the ability of the IFN-induced proteasome catalytic subunits to shape the HBV-specific CD8 T-cell response and thus potentially influence the progression of infection to acute or chronic disease. 17142736_two inducible subunits of the proteasome, lmp2 and lmp7, are transcriptionally up-regulated by heat shock; heat-shocked cells show enhanced presentation of immunoproteasome-dependent MHC I antigenic epitopes, but not immunoproteasome-independent epitopes 19830724_Results show that mice lacking LMP7 were highly susceptible to infection with T. gondii and showed a reduced number of functional CD8+ T cells. 21304910_A dominant role for the immunoproteasome in CD8+ T cell responses to murine cytomegalovirus. 21455681_Findings strongly support the concept that LMP7/MECL-1 proteasomes subunits actively function to regulate LPS-induced NO production by affecting the TRIF/TRAM pathway. 21804012_We show that mice deficient for the immunosubunits beta5i/low molecular mass polypeptide (LMP7) and beta2i/multicatalytic endopeptidase complex-like-1 develop early-stage multiorgan autoimmunity following irradiation and bone marrow transplantation 22398747_LMP7 and MECL1 regulate cytokine expression, suggesting this system represents a novel mechanism for the regulation of cytokines and cytokine signaling 22768135_Probeta5 predominantly promotes integration into LMP2/MECL-1-containing precursors in IFNgamma-stimulated, LMP7-deficient cells and infected LMP7-deficient mice. 22984077_Deletion or inhibition of LMP7 suppresses generation of T helper (Th)17 cells but promotes regulatory T cell (Treg) development. 23527234_Red blood cells of LMP7-deficient mice were more likely to deform in response to infection with malaria parasites, presumably resulting in higher susceptibility to phagocytosis and in the partial resistance to malaria. 23593249_we present novel evidence for the requirement of the beta5i immunosubunit to generate a strong Th2 response during OVA- but not HDM-induced acute asthma. 23763593_Overexpression of the immunoproteasome LMP7 subunit in antigen-presenting cells due to lipopolysaccharide exposure as well as LMP7 expression in peripheral cells, are required for CD8+ T-cell auto-reactivity. 24156634_PSMB8, ALDH1A1, and HSPA4 were identified to be located in ovarian tissues, to regulate 12 cellular pathways, and demonstrate age-dependent dynamic changes in expression profiling. 24164898_Data show that knockdown of i-proteasome catalytic subunit PSMB9 by short hairpin RNA (shRNA) decreased the expression of both PSMB9 and PSMB8 without affecting other catalytic subunits of the proteasome. 24374040_genetic polymorphism is associated with ovarian cancer and metastasis 26093043_these data support that LMP7 inhibition in the context of BMT modulates allogeneic responses by decreasing endogenous miHA presentation and that the consequential reduction in allogeneic stimulation and cytokine production reduces GVHD development. 26515636_LMP7 deficiency decreased inflammatory responses such as macrophage infiltration and chemokine expression while it increased serum adiponection levels. 27225296_Psmb8 directly regulates the differentiation of preadipocytes and additionally the differentiation of preadipocytes to mature adipocytes. Psmb8(-/-) mice had slower weight gain, reduced adipose tissue volume, less preadipocyte precursors and preadipocytes, and smaller size of mature adipocytes compared with controls. Loss of Psmb8 activity in 3T3-L1 cells disturbed the differentiation to mature adipocytes. 27264074_The regulation of miR-451 via the LMP7/NF-kappaB central inflammatory pathway during progression of DN. 28582644_The authors found that selective inhibition of LMP7 had neither an influence on allograft survival in an major histocompatibility complex-mismatch model nor in a multiple minor mismatch skin transplantation model. 29042581_deficiency of IP subunit beta5i/LMP7 does not alter initiation and progression of atherosclerosis in LDLR-/- mice. 29067678_Employing the lymphocytic choriomeningitis virus (LCMV) infection model, we showed that the immunoproteasome subunit LMP7 was absolutely required for the generation of LCMV GP118-125 -specific T cells although the class I mediated presentation of GP118-125 was not dependent on LMP7. 29759483_Proteostatic responses included a significant increase in the levels of Lmp7, a component of the immunoproteasome. Increased Lmp7 levels and activity were also quantified in postmortem human brains with PD and dementia with Lewy bodies. 30279279_co-inhibition of LMP2 and LMP7 appears to be synergistic and advantageous for the treatment of autoimmune diseases. 30642978_results provide, to our knowledge, novel evidence that ablation or pharmacological inhibition of LMP7 attenuates Ang II-induced abdominal aortic aneurysm (AAA) formation, and LMP7 might be a novel therapeutic target for treating AAA in humans 31086810_Knockout of beta5i(PSMB8) in cardiomyocytes and mice markedly attenuated the hypertrophic response, and this effect was aggravated by beta5i overexpression in cardiomyocytes. beta5i interacted with and promoted ATG5 degradation thereby leading to inhibition of autophagy and cardiac hypertrophy. 31629736_This study provides new evidence of the critical role of beta5i in deoxycorticosterone-acetate-salt-induced cardiac remodeling. 31636038_Ablation of Immunoproteasome beta5i Subunit Suppresses Hypertensive Retinopathy by Blocking ATRAP Degradation in Mice. 33019975_Macrophage pyroptosis is mediated by immunoproteasome subunit beta5i (LMP7) in abdominal aortic aneurysm. 34547302_Expression of immunoproteasome subunits in the brains of Toxoplasma gondii-infected mice. |
ENSG00000204264 |
PSMB8 |
521.286889 |
5.1235398986 |
2.357141 |
0.15066076 |
236.038601 |
0.00000000000000000000000000000000000000000000000000002874066676057236567012509487962683663147758162963291950952652888968267039574534770148196826330806173468538031691943860070934931171438182773147929083279450424015522003173828125000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000055414915961650415843459764974207213695587559302201614000823419679915326952560996873217799300939449075260309426059561883098116821452783220536275621270760893821716308593750000000000000000000000000000000000000000 |
Yes |
No |
885.180173765781 |
92.6075496206111 |
172.767303716628 |
14.3354466860556 |
| ENSMUSG00000024427 |
24066 |
Spry4 |
protein_coding |
Q9WTP2 |
FUNCTION: Suppresses the insulin receptor and EGFR-transduced MAPK signaling pathway, but does not inhibit MAPK activation by a constitutively active mutant Ras. Probably impairs the formation of GTP-Ras (By similarity). Inhibits Ras-independent, but not Ras-dependent, activation of RAF1 (By similarity). Represses integrin-mediated cell spreading via inhibition of TESK1-mediated phosphorylation of cofilin (By similarity). {ECO:0000250|UniProtKB:Q9C004}. |
Acetylation;Cell membrane;Cell projection;Cytoplasm;Developmental protein;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:24066; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; ruffle membrane [GO:0032587]; protein kinase inhibitor activity [GO:0004860]; animal organ development [GO:0048513]; cellular response to leukemia inhibitory factor [GO:1990830]; negative regulation of fibroblast growth factor receptor signaling pathway [GO:0040037]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of Ras protein signal transduction [GO:0046580]; negative regulation of substrate adhesion-dependent cell spreading [GO:1900025] |
12781690_Spry4, an intracellular FGF receptor antagonist, was expressed in epithelial cells of the fetal lung under control of a doxycycline-inducible system. 17156747_suggest both redundant and non-redundant functions for Sprouty2 and Sprouty4 on embryonic development and FGF signaling 18337453_Transgenic expression of murine Spry4 during beta-cell carcinogenesis does not significantly affect tumor outgrowth and progression to tumor malignancy. 19424491_These data suggest that Sproutys are physiologically important negative regulators of angiogenesis in vivo and novel therapeutic targets for treating peripheral ischemic diseases. 19440349_Periodic expression of Sprouty4 is controlled by the Notch segmentation clock and may work as a mediator that links the temporal periodicity of clock gene oscillations with the spatial periodicity of boundary formation. 19453770_Spry4 and Tpbg/5T4 were detected in Kit-immunoreactive cells only in Kit(K641E), but not in Kit(WT/WT) animals. 19493272_Sprouty4 deficiency potentiates Ras-independent angiogenic signals and tumor growth. 21240919_our results imply that Sprouty2 and -4 are downregulated in the hippocampus during postnatal brain development and that they can act as regulators of developmental axon growth. 21862563_Interestingly, changes in sprouty gene dosage led to a graded change in incisor number, with progressive decreases in sprouty dosage leading to increasing numbers of teeth. 23074222_ISG15 mRNA expression and IFN-dependent antiviral responses are enhanced in Spry1,2,4 triple knock-out mouse embryonic fibroblasts, consistent with negative feedback regulatory roles for Spry proteins in IFN-mediated signaling. 23606267_We propose that Sprouty genes(Spry2 and Spry4) were implicated during evolution in reduction of the cheek teeth in Muridae, and their deletion can reveal ancestral stages of murine dental evolution. 23955631_In vivo analysis revealed that Spry4 regulated integrin beta3 levels in murine embryos and yolk sacs. 25541251_We demonstrate that in spry4-/- mice inflammatory responses, such TNFalpha secretion and macrophage/neutrophil invasion into the lesion site are reduced. In addition, astrocytic gliosis is attenuated and neuronal survival is increased. 25822087_SPRY4 is a tumor suppressor at 5q whose disruption contributes to a lethal acute myeloid leukemia subtype. 25897685_in embryos with lower Spry2;Spry4 gene dosages, we observed a non-fusion of original R2 and M1 Shh signaling domains with consequent formation of a supernumerary tooth primordium from the isolated R2 bud 26291313_Autoregulatory loop between TGF-beta1/miR-411-5p/SPRY4 and p38 MAPK pathway in rhabdomyosarcoma modulates proliferation and differentiation. 26540287_In the present study, it is demonstrated that Spry2 and -4 participate in KA induced neurodegeneration possibly through inhibition of ERK signaling. 28732181_Irf6 and RTK signaling interact in regulating periderm differentiation and function, as well as provide a rationale to screen for epistatic interactions between variants in IRF6 and RTK signaling pathway genes in human orofacial clefting populations. 29964027_A Sprouty4 reporter to monitor FGF/ERK signaling activity in ESCs and mice. 34423857_Loss of Sprouty Produces a Ciliopathic Skeletal Phenotype in Mice Through Upregulation of Hedgehog Signaling. 36334381_Macrophage Sprouty4 deficiency diminishes sepsis-induced acute lung injury in mice. |
ENSG00000187678 |
SPRY4 |
61.491632 |
0.1210249526 |
-3.046624 |
0.43538800 |
47.382322 |
0.00000000000584070594648413317164500286195099655085771761520163636305369436740875244140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001059114678295789447325768329257627588246837291308111161924898624420166015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.0212473088918 |
4.29596729274362 |
115.85418546102 |
23.1687861037784 |
| ENSMUSG00000024451 |
106952 |
Arap3 |
protein_coding |
Q8R5G7 |
FUNCTION: Phosphatidylinositol 3,4,5-trisphosphate-dependent GTPase-activating protein that modulates actin cytoskeleton remodeling by regulating ARF and RHO family members. Is activated by phosphatidylinositol 3,4,5-trisphosphate (PtdIns(3,4,5)P3) binding. Can be activated by phosphatidylinositol 3,4-bisphosphate (PtdIns(3,4,5)P2) binding, albeit with lower efficiency. Acts preferentially on ARF5 and on RHOA. {ECO:0000269|PubMed:15546919}. |
Alternative splicing;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;GTPase activation;Membrane;Phosphoprotein;Reference proteome;Repeat |
|
This gene encodes a phosphoinositide binding protein containing ARF-GAP, RHO-GAP, RAS-associating, and pleckstrin homology domains. The ARF-GAP and RHO-GAP domains cooperate in mediating rearrangements in the cell cytoskeleton and cell shape. It is a specific PtdIns(3,4,5)P3/PtdIns(3,4)P2-stimulated Arf6-GAP protein. Two transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Sep 2015]. |
mmu:106952; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; lamellipodium [GO:0030027]; plasma membrane [GO:0005886]; ruffle [GO:0001726]; GTPase activator activity [GO:0005096]; phosphatidylinositol-3,4,5-trisphosphate binding [GO:0005547]; phosphatidylinositol-3,4-bisphosphate binding [GO:0043325]; positive regulation of GTPase activity [GO:0043547]; signal transduction [GO:0007165] |
15546919_ARAP3 has a role in integrin-mediated tyrosine kinase signalling pathways controlling Rho GTPases and cell spreading 18765673_the Nck adapters are needed for signaling to Rho GTPases and actin dynamics downstream of the PDGFbeta receptor 20978237_PI3K signaling through the dual GTPase-activating protein ARAP3 is essential for developmental angiogenesis. 21490342_We report here the first characterization of the role of ARAP3 in murine neutrophils 24163130_Arap3 is dysregulated in a mouse model of hypotrichosis-lymphedema-telangiectasia and regulates lymphatic vascular development. 25542002_Deletion of Arap3 in osteoblasts and mesenchymal stromal cells using Prx1-Cre resulted in no discernable phenotypes in hematopoietic development or hematopoietic stem cells homeostasis in adult mice. |
ENSG00000120318 |
ARAP3 |
24.718453 |
4.0022940775 |
2.000827 |
0.69884186 |
7.445452 |
0.00635969497294289805994216280282671505119651556015014648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0227785757503658516098798969551353366114199161529541015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.3268221448286 |
19.3001355302392 |
9.57621339221882 |
3.69667405656079 |
| ENSMUSG00000024459 |
240095 |
H2-M5 |
protein_coding |
H3BJA9 |
Human_homologues FUNCTION: Non-classical major histocompatibility class Ib molecule postulated to play a role in immune surveillance, immune tolerance and inflammation. Functions in two forms, as a heterotrimeric complex with B2M/beta-2 microglobulin and a peptide (peptide-bound HLA-F-B2M) and as an open conformer (OC) devoid of peptide and B2M (peptide-free OC). In complex with B2M, presents non-canonical self-peptides carrying post-translational modifications, particularly phosphorylated self-peptides. Peptide-bound HLA-F-B2M acts as a ligand for LILRB1 inhibitory receptor, a major player in maternal-fetal tolerance. Peptide-free OC acts as a ligand for KIR3DS1 and KIR3DL2 receptors (PubMed:28636952). Upon interaction with activating KIR3DS1 receptor on NK cells, triggers NK cell degranulation and anti-viral cytokine production (PubMed:27455421). Through interaction with KIR3DL2 receptor, inhibits NK and T cell effector functions (PubMed:24018270). May interact with other MHC class I OCs to cross-present exogenous viral, tumor or minor histompatibility antigens to cytotoxic CD8+ T cells, triggering effector and memory responses (PubMed:23851683). May play a role in inflammatory responses in the peripheral nervous system. Through interaction with KIR3DL2, may protect motor neurons from astrocyte-induced toxicity (PubMed:26928464). {ECO:0000269|PubMed:23851683, ECO:0000269|PubMed:24018270, ECO:0000269|PubMed:26928464, ECO:0000269|PubMed:27455421, ECO:0000269|PubMed:28636952}. |
Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
|
cell surface [GO:0009986]; cis-Golgi network membrane [GO:0033106]; early endosome [GO:0005769]; early endosome membrane [GO:0031901]; endoplasmic reticulum [GO:0005783]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; lysosomal membrane [GO:0005765]; MHC class I protein complex [GO:0042612]; MHC class Ib protein complex [GO:0032398]; plasma membrane [GO:0005886]; 14-3-3 protein binding [GO:0071889]; beta-2-microglobulin binding [GO:0030881]; CD8 receptor binding [GO:0042610]; chaperone binding [GO:0051087]; identical protein binding [GO:0042802]; MHC class I protein binding [GO:0042288]; natural killer cell lectin-like receptor binding [GO:0046703]; peptide antigen binding [GO:0042605]; protein homodimerization activity [GO:0042803]; signaling receptor binding [GO:0005102]; T cell receptor binding [GO:0042608]; TAP1 binding [GO:0046978]; TAP2 binding [GO:0046979]; antigen processing and presentation of endogenous peptide antigen via MHC class Ib [GO:0002476] |
Human_homologues 12874228_This is the first example of placental HLA-F expression--primarily in trophoblasts that have invaded the maternal decidua--in the same cells that simultaneously express the other two nonclassical class I antigens HLA-E and HLA-G. 14607927_HLA-F surface expression on B cell and monocyte cell lines correlates with the presence of a limited amount of endoglycosidase H (Endo H)-resistant HLA-F; however, clearly not all surface-expressed HLA-F is fully glycosylated. 16570139_The results of this analysis confirmed several previously reported coding sequence variants, identified several new allelic variants, and also defined extensive variation in intron and flanking sequences. 16709803_HLA-F is entirely dependent on its cytoplasmic tail for export from the endoplasmic reticulum. 17157219_strong positive directional selection is acting for maintaining the observed low polymorphism on HLA-E, -F and -G loci 17971048_Observational study of gene-disease association. (HuGE Navigator) 18658158_the role of NKG2D and 2B4 is not focussed on trophoblast recognition in normal pregnancy, but is more likely involved in cross-talk among maternal cells of the placental bed 18941505_Across HIV Gag protein, the rise of polymorphisms from independent origin during the last twenty years of epidemic was related to an association with an HLA allele accumulated in one of either B or F subtypes 19622345_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 19664746_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 19851445_Observational study of gene-disease association. (HuGE Navigator) 20237496_Observational study of gene-disease association. (HuGE Navigator) 20483783_These data suggest that HLA-F is expressed independently of peptide and that a physical interaction specific to MHC-I HC plays a role in the function of MHC-I HC expression in activated lymphocytes. 20593013_Observational study of gene-disease association. (HuGE Navigator) 20865824_surface marker on activated lymphocytes 22490650_Mifepristone can inhibit the effects of progesterone by down-regulating the expressions of HLA-G, HLA-E and HLA-F mRNA in trophoblasts during the first trimester. 22544725_Upregulated HLA-F expression (p = 0.026) and downregulated HLA I expression (p = 0.013) could be an independent unfavorable prognostic factor. 23542057_expression in gastric cancer lesion was unrelated to patient prognosis 23851683_A previously unrecognized model of Ag cross-presentation mediated by HLA-F & MHC-I open conformers on activated lymphocytes & monocytes may significantly contribute to the regulation of immune system functions & the immune defense. 24018270_HLA-F and MHC class I open conformers are ligands for NK cell Ig-like receptors. 25413105_This is the first study regarding HLA-F polymorphisms in a Euro-Brazilian population contributing to the Southern Brazilian genetic characterization. 25420801_Review of the impact of human leukocyte antigen molecules E, F, and G on the outcome of transplantation. 25461528_The effect of Japanese encephalitis virus and TNF-alpha exposure on NFkappaB-mediated induction of HLA-F is reported. 25862890_We identified that the HLA-E and HLA-F in gastric cancer independently affected clinical factors, including postoperative outcome 26332651_provides substantial evidence that the rs7903146 variant is significantly associated with the risk of diabetic retinopathy in Caucasian populations 26928464_Overexpression of a single MHCI molecule, HLA-F, protects human MNs from ALS astrocyte-mediated toxicity, whereas knockdown of its receptor, the killer cell immunoglobulin-like receptor KIR3DL2, on human astrocytes results in enhanced motor neurons death 27447835_Two eSNPs were associated with fecundability at a FDR of 5%; both were in the HLA region and were eQTLs for the TAP2 gene (P = 1.3x10-4) and the HLA-F gene (P = 4.0x10-4), respectively. 27455421_this study established HLA-F as a ligand of KIR3DS1 and have demonstrated cell-context-dependent expression of HLA-F that might explain the widespread influence of KIR3DS1 in human disease, including delayed progression of disease caused by human immunodeficiency virus type 1 27522114_Data suggest that the immunoproteasome is involved in pathologic MHC class I (HLA-A, B, C, F and G) expression and maintenance of myokine production in Idiopathic inflammatory myopathies (IIMs). 28185362_In this study, we describe and confirm the distinct expression of HLA-F, HLA-G, HLA-E, and HLA-C in placental tissue 28636952_the results of this study provide a blueprint of the molecular details of HLA-F, which will inform future exploration of its roles in human health and allow for the development of additional, targeted therapeutics 30031767_interactions between KIR3DS1 and HLA-F contribute to NK cell-mediated control of HCV. 30245028_The rs2523393 A allele creates a GATA2 binding site in a progesterone-responsive distal enhancer that loops to the HLA-F promoter. 30510890_high HLA-F expression is associated with Nasopharyngeal Carcinoma (NPC) local recurrence and distant metastasis and may be regarded as a poor prognostic factor for NPC patients. 30755240_This study showed that the HLA-F expression was positively correlated with malignant phenotype and negatively correlated with overall survival. 31717259_HLA-F variants bound to selected peptides, were structurally compared. 31863778_HLA-F-AS1 also enhanced the expressions of PFN1, which was validated as a target gene of miR-330-3p. CONCLUSION: HLA-F-AS1 promoted colorectal cancer progression via regulating miR-330-3p/PFN1 axis 32020202_Variation in the HLA-F gene locus with functional impact is associated with pregnancy success and time-to-pregnancy after fertility treatment. 34257557_Development of a Novel Prognostic Signature Based on Antigen Processing and Presentation in Patients with Breast Cancer. 35181585_HLA-E and HLA-F Are Overexpressed in Glioblastoma and HLA-E Increased After Exposure to Ionizing Radiation. 35276697_LncRNA HLA-F-AS1 attenuates the ovarian cancer development by targeting miR-21-3p/PEG3 axis. 35925520_Evolution and molecular interactions of major histocompatibility complex (MHC)-G, -E and -F genes. |
ENSG00000204642 |
HLA-F |
13.798661 |
3.5502839685 |
1.827934 |
0.69758294 |
6.858722 |
0.00882096392509618479338406871193001279607415199279785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0301325235494651483270089897814614232629537582397460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.912324546381 |
11.5155168798933 |
6.45365969296416 |
2.53894567280059 |
| ENSMUSG00000024521 |
58801 |
Pmaip1 |
protein_coding |
Q9JM54 |
FUNCTION: Promotes activation of caspases and apoptosis. Promotes mitochondrial membrane changes and efflux of apoptogenic proteins from the mitochondria. Contributes to p53/TP53-dependent apoptosis after radiation exposure. Promotes proteasomal degradation of MCL1. Competes with BIM/BCL2L11 for binding to MCL1 and can displace BIM/BCL2L11 from its binding site on MCL1 (By similarity). Competes with BAK1 for binding to MCL1 and can displace BAK1 from its binding site on MCL1. {ECO:0000250, ECO:0000269|PubMed:10807576, ECO:0000269|PubMed:15694340, ECO:0000269|PubMed:15901672, ECO:0000269|PubMed:16822983, ECO:0000269|PubMed:17389404}. |
3D-structure;Apoptosis;Mitochondrion;Reference proteome;Repeat |
|
|
mmu:58801; |
Bcl-2 family protein complex [GO:0097136]; cytosol [GO:0005829]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; apoptotic process [GO:0006915]; cellular response to DNA damage stimulus [GO:0006974]; cellular response to glucose starvation [GO:0042149]; defense response to virus [GO:0051607]; fibroblast apoptotic process [GO:0044346]; intrinsic apoptotic signaling pathway [GO:0097193]; intrinsic apoptotic signaling pathway by p53 class mediator [GO:0072332]; intrinsic apoptotic signaling pathway in response to DNA damage [GO:0008630]; intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator [GO:0042771]; intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress [GO:0070059]; negative regulation of fibroblast proliferation [GO:0048147]; negative regulation of mitochondrial membrane potential [GO:0010917]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of DNA damage response, signal transduction by p53 class mediator [GO:0043517]; positive regulation of extrinsic apoptotic signaling pathway via death domain receptors [GO:1902043]; positive regulation of fibroblast apoptotic process [GO:2000271]; positive regulation of glucose metabolic process [GO:0010907]; positive regulation of intrinsic apoptotic signaling pathway [GO:2001244]; positive regulation of neuron apoptotic process [GO:0043525]; positive regulation of protein insertion into mitochondrial membrane involved in apoptotic signaling pathway [GO:1900740]; positive regulation of release of cytochrome c from mitochondria [GO:0090200]; proteasomal protein catabolic process [GO:0010498]; reactive oxygen species metabolic process [GO:0072593]; regulation of mitochondrial membrane permeability [GO:0046902]; release of cytochrome c from mitochondria [GO:0001836]; response to dsRNA [GO:0043331]; response to UV [GO:0009411]; response to X-ray [GO:0010165]; T cell homeostasis [GO:0043029] |
12952892_Noxa(-/-) mice showed resistance to X-ray irradiation-induced gastrointestinal death, accompanied with impaired apoptosis. 14500851_Puma and Noxa are critical mediators of the apoptotic responses induced by p53 and other agents 15703398_Noxa is a major executor for axotomy-induced motor neuron death in the adult mouse, as a mediator located downstream of p53. 16407291_PUMA, p53 and NOXA are components of the ER stress-induced apoptotic pathway, and both contribute to ER stress-induced apoptosis 16822983_Noxa and Puma are important regulators of genotoxin-induced telencephalic neuron precursor cell death 16928686_functional eIF2alpha played an essential role in PS-341-induced Noxa expression 17024184_Puma and Noxa, the well-known p53-inducible proapoptotic members of the Bcl-2 family, differentially participate in dual pathways of the induction of apoptosis 17167080_The deleterious function in cerebral ischemia is specific for the NF-kappa B subunit RelA and may be mediated through Bim and Noxa. 17283183_Collectively, these results demonstrate that UVR activates the Bcl-2-regulated apoptotic pathway predominantly through activation of Noxa and, depending on cellular context, Puma. 17518541_A significant suppression of Noxa expression by the Suture may be a major reason why nerve suture induces survival and regeneration of nerve-injured motor neurons. 18192275_Interferon gamma induces XAF1 and Noxa expression and potentiates apoptosis by STAT3 activation 18259198_Results indicate that, at least in fibroblasts and thymocytes, p53-induced apoptosis proceeds principally via Noxa and Puma, with Puma having the predominant role in diverse cell types. 18411339_Bmi1 controls memory CD4(+) Th1/Th2 cell survival and functions through the direct repression of the Noxa gene. 18589438_The structure of the BH3 domains from the p53-inducible BH3-only protein Noxa in complex with Mcl-1 is reported. 19148184_Pmaip1 protein deficiency alone also increased B-lineage cells but did not accelerate lymphomagenesis. 19237507_Noxa-dependent cell death might contribute to particulate matter-induced alveolar epithelial dysfunction and the resulting inflammatory response 20195489_p73 is activated after genotoxin treatment only in the presence of NF-kappaB and p73 induces Noxa gene expression through the p53 element in the promoter. 20620942_Here we showed that upon T cell activation, the proapoptotic molecule Noxa (encoded by Pmaip1) and its antagonist Mcl-1 were induced 21163929_define the first physiologic function for Noxa and suggest that by repressing Noxa, induction of G arrest by p18INK4c bypasses a homeostatic cell-cycle checkpoint in intermediate plasma cells (iPCs) for PC differentiation 21660046_Function of Noxa was at least in part neutralization of induced myeloid leukemia cell differentiation protein (Mcl-1) in neutrophils and progenitors. 21689638_This study reveals Noxa to be a crucial regulator of osteoclast apoptosis. 21690328_Investigation of downstream effectors used by tumor protein p53 to impair T cell lineage development finds many p53 targets are induced in ribosomal protein Rpl22-deficient thymocytes, including Noxa, Bax, p21waf, miR-34a, and PUMA. 21742363_Here the authors demonstrate that Noxa null baby mouse kidney cells are deficient in normal cytopathic response to lytic viruses, and that reconstitution of the knockout cells with wild-type Noxa restored normal cytopathic responses. 21945433_in response to DNA-damage, Noxa efficiently induces apoptosis by 'release' of Puma from Mcl-1. 22144184_Data show that Noxa is induced in activated B cells, and its ablation provides a survival advantage both in vitro and in vivo. 22185759_two proapoptotic Bcl-2 family members Bok and Noxa/Pmaip are directly transcriptionally induced by activated MAL and upon activation of the actin-MAL-SRF pathway 22380599_The Bcl-2 proteins Noxa and Bcl-xL co-ordinately regulate oxidative stress-induced apoptosis. 22573037_The Noxa protein, even in combination with Bik, is not a potent suppressor of c-Myc-driven tumourigenesis or critical for chemotherapeutic drug-induced killing of Myc-driven tumours. 23000175_Data show that PUMA and NOXA are critical for DNA damage-induced, TAp63-mediated primordial follicle oocyte apoptosis. 23068609_Results suggest that compromised induction of Unfolded Protein Response (UPR) and increased NOXA expression contributes to hypersensitivity of PERK(-/-) MEFs to ER stress-induced apoptosis. 23277490_In acute viral infection, Noxa(-/-) mice had increased memory pool size & diversity but less cross-reactivity. Reduced T-cell apoptosis during chronic activation led to severe organ pathology & early death. 23593480_Oppositional regulation of Noxa by JNK1 and JNK2 during apoptosis induced by proteasomal inhibitors. 23665218_Induction of senescence was only impaired in cells from the p21-/- puma-/- noxa-/- mice but abrogated in cells from the p53-/- mice. 23733106_Noxa is targeted to the mitochondrial membrane where it neutralises Mcl-1 via its C-terminal BH3-domain. 23770082_Overall, these data reveal a Noxa-mediated signaling pathway that couples lysosomal membrane permeabilization with mitochondrial outer membrane permeabilization and ultimate apoptosis during oxidative stress. 23975731_Noxa controls expansion of erythroid precursors and RBC production in vivo under conditions of induced anemia. 24608793_the current findings indicate that Noxa is a novel regulator of early mitosis before 75% epiboly stage when it translates into a key mediator of apoptosis in subsequent embryogenesis. 25124553_by preventing the consumption of IL-15, Bim limits the role of Noxa and Puma in causing the death of effector cells with less memory potential. 25299781_Induction of noxa does not influence ischemic neuronal injury. 25733019_Our data demonstrate that ES cells are uniquely sensitive to CDK1 inhibition via a p53/NOXA/MCL1 pathway. 26181197_In addition, studies in leukemia Jurkat T cells support the existence of the Sall2/Noxa axis, and the significance of this axis on the apoptotic response to doxorubicin in cancer cells. 26497683_Fluorizoline bind to prohibitin, inducing mitochondrial apoptotic pathway through NOXA and BIM upregulation. 26866517_knockdown of pmaip1 mimicked the phenotype of ph8(-/Y) by showing the decreased apoptosis during early differentiation of embryonic stem cells and promoted mesodermal and cardiac commitment. 27479816_data suggest that in the context of CLL NOXA may function as an oncomodulator 27862899_Noxa is involved in X-ray-induced lung injury. 28079889_In vivo upregulation of noxa was reduced by pifithrin-alpha, suggesting transcription may be partly p53-dependent.The present findings indicate Noxa does not serve as a proapoptotic BH3-only protein during seizure-induced neuronal death in vivo 29931031_Findings indicate that prospero homeobox 1 (Prox1) regulated the differentiation of oligodendrocyte precursor cells via the regulation of phorbol-12-myristate-13-acetate-induced protein 1 (NOXA). 31580946_The vanillin derivative VND3207 protects intestine against radiation injury by modulating p53/NOXA signaling pathway and restoring the balance of gut microbiota. 32312973_NOXA-dependent contextual synthetic lethality of BCL-XL inhibition and ''osmotic reprogramming'' in colorectal cancer. |
ENSG00000141682 |
PMAIP1 |
9.607831 |
0.0998059451 |
-3.324730 |
1.14316859 |
8.205370 |
0.00417665804644001478762582735271280398592352867126464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0159525715555101053888531481561585678718984127044677734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.47812841289126 |
1.29771669862782 |
14.8100237019566 |
7.97446626726804 |
| ENSMUSG00000024534 |
67847 |
Sncaip |
protein_coding |
G5E848 |
Human_homologues FUNCTION: Isoform 2 inhibits the ubiquitin ligase activity of SIAH1 and inhibits proteasomal degradation of target proteins. Isoform 2 inhibits autoubiquitination and proteasomal degradation of SIAH1, and thereby increases cellular levels of SIAH. Isoform 2 modulates SNCA monoubiquitination by SIAH1. {ECO:0000269|PubMed:16595633, ECO:0000269|PubMed:19224863}. |
ANK repeat;Proteomics identification;Reference proteome;Repeat |
|
|
mmu:67847; |
cytoplasmic ribonucleoprotein granule [GO:0036464]; nucleoplasm [GO:0005654]; identical protein binding [GO:0042802]; ubiquitin protein ligase binding [GO:0031625]; cell death [GO:0008219]; dopamine metabolic process [GO:0042417]; regulation of inclusion body assembly [GO:0090083]; regulation of neurotransmitter secretion [GO:0046928] |
19760259_synphilin-1 has a role in skill learning and motor performance 19857556_expression of synphilin-1 shortens N1E-115 cell division doubling time, promotes neurite outgrowth, and protects against Rotenone-induced toxicity; synphilin-1 displays a neurotrophic effect in vitro, may play a neuroprotective role in Parkinson's disease 20185556_Synphilin-1 can diminish the severity of alpha-synucleinopathy and play a neuroprotective role against A53T alpha-synuclein toxicity in vivo. 22158267_Studies identify a novel function of synphilin-1 in controlling food intake and body weight, and may provide a unique obesity model for future studies of obesity pathogenesis and therapeutics. 24902662_Sncaip physically interacts with PPP1 and affects PPP1 sorting to subcellular compartments. 24947141_We exploited atomic force microscopy (AFM) for the selective, nanoscale confinement of alpha-Synuclein on gold to study the early stages of alpha-Synuclein aggregation and the effect of small molecules, like dopamine, on the aggregation process. 25886189_an important role of aSyn H50 in the pathogenesis of both sporadic and monogenic Parkinson's disease 25917213_This study shows that alpha-synuclein is present in cerebellar glutamatergic terminals. 26569394_SP1 results in a CCK response deficiency that may contribute to the increased meal size and overall hyperphagia in synphillin-1 transgenic mice 26697752_TEM analysis of protein:inhibitor assays demonstrated that scyllo-inositol inhibits the aggregation of alpha-synuclein, suggesting the therapeutic potential of the small molecule in PD. 26797281_These results suggest that increased levels of NUB1 play a potential role in degradation of detergent-insoluble alpha-synuclein in vivo, although it is insufficient to degrade abnormal alpha-synuclein in Lewy body disease model mice. 27466341_These results suggest that alpha-syn might participate in mechanisms of vulnerability of selected neuronal populations in AD and that reducing alpha-syn might be a potential approach to protecting these populations from the toxic effects of amyloid beta. 27741433_Mulberry fruit extract had inhibitory effects on the up-regulation of both alpha-synuclein and ubiquitin, the major components of lewy bodies, in substantia nigra and striatum in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine/probenecid model of early Parkinson's disease . 27818201_Neurons of A53T-alpha-Syn transgenic mice are more fragile to DNA damages. 28621812_2,3,7,8-tetrachlorodibenzo-p-dioxin promoted synphilin-1 degradation in an aryl hydrocarbon receptor-dependent way. 28870519_Enhanced aggregation and phosphorylation of alphaSyn causes the loss of dopaminergic neurons leading to the decline of motor coordination. 29124353_The finding of increased spine pathology in Thy1-E57K is a new pathogenic function of oligomeric a-syn and precedes overt neurodegeneration. Thus, it may constitute a readout for therapeutic approache. 29747690_This study showed that A53T-alpha-synuclein overexpression in murine locus coeruleus induces Parkinson's disease-like pathology in neurons and glia. 30966771_Intracytoplasmic Lewy bodies are primarily composed of the protein alpha-synuclein which increases in substantia nigra of Parkinson's disease. 30987677_Study suggests alpha-synucleinopathy induction, via retrograde and contralateral projections, within the olfactory system of transgenic animals. 31683259_Dopaminergic neuron injury in Parkinson's disease is mitigated by interfering lncRNA SNHG14 expression to regulate the miR-133b/ alpha-synuclein pathway. 31722091_alpha-Synuclein Promotes Maturation of Immature Juxtaglomerular Neurons in the Mouse Olfactory Bulb. 32502569_Transglutaminase 2 Depletion Attenuates alpha-Synuclein Mediated Toxicity in Mice. 33926043_Preclinical Detection of Alpha-Synuclein Seeding Activity in the Colon of a Transgenic Mouse Model of Synucleinopathy by RT-QuIC. 34595918_Accumulation of Prion and Abnormal Prion Protein Induces Hyperphosphorylation of alpha-Synuclein in the Brain Tissues from Prion Diseases and in the Cultured Cells. 35773260_p21-activated kinase 4 controls the aggregation of alpha-synuclein by reducing the monomeric and aggregated forms of alpha-synuclein: involvement of the E3 ubiquitin ligase NEDD4-1. 35830804_Treadmill exercise reduces alpha-synuclein spreading via PPARalpha. |
ENSG00000064692 |
SNCAIP |
59.616145 |
0.3443605197 |
-1.538008 |
0.43606721 |
12.056360 |
0.00051615976132698970779699720878852531313896179199218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0025743763865529495533934767337314042379148304462432861328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.960865557044 |
8.58069307426809 |
81.1964901733986 |
18.2183231237424 |
| ENSMUSG00000024593 |
70417 |
Megf10 |
protein_coding |
Q6DIB5 |
FUNCTION: Membrane receptor involved in phagocytosis by macrophages and astrocytes of apoptotic cells. Receptor for C1q, an eat-me signal, that binds phosphatidylserine expressed on the surface of apoptotic cells (PubMed:27170117). Cooperates with ABCA1 within the process of engulfment (By similarity). Promotes the formation of large intracellular vacuoles and may be responsible for the uptake of amyloid-beta peptides (PubMed:20828568). Necessary for astrocyte-dependent apoptotic neuron clearance in the developing cerebellum (PubMed:27170117). Plays role in muscle cell proliferation, adhesion and motility. Is also an essential factor in the regulation of myogenesis. Controls the balance between skeletal muscle satellite cells proliferation and differentiation through regulation of the notch signaling pathway (PubMed:28498977,Ref.10). May also function in the mosaic spacing of specific neuron subtypes in the retina through homotypic retinal neuron repulsion. Mosaics provide a mechanism to distribute each cell type evenly across the retina, ensuring that all parts of the visual field have access to a full set of processing elements (PubMed:22407321). {ECO:0000250|UniProtKB:Q96KG7, ECO:0000269|PubMed:18056409, ECO:0000269|PubMed:20828568, ECO:0000269|PubMed:22407321, ECO:0000269|PubMed:27170117, ECO:0000269|PubMed:28498977, ECO:0000269|Ref.10}. |
Cell adhesion;Cell membrane;Cell projection;Disulfide bond;EGF-like domain;Glycoprotein;Membrane;Myogenesis;Phagocytosis;Phosphoprotein;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:70417; |
cell projection [GO:0042995]; phagocytic cup [GO:0001891]; plasma membrane [GO:0005886]; complement component C1q complex binding [GO:0001849]; Notch binding [GO:0005112]; scavenger receptor activity [GO:0005044]; apoptotic cell clearance [GO:0043277]; apoptotic process involved in development [GO:1902742]; engulfment of apoptotic cell [GO:0043652]; homotypic cell-cell adhesion [GO:0034109]; muscle cell development [GO:0055001]; muscle cell proliferation [GO:0033002]; myoblast development [GO:0048627]; myoblast migration [GO:0051451]; positive regulation of cell-cell adhesion [GO:0022409]; positive regulation of myoblast proliferation [GO:2000288]; recognition of apoptotic cell [GO:0043654]; regulation of muscle cell differentiation [GO:0051147]; regulation of skeletal muscle tissue development [GO:0048641]; skeletal muscle satellite cell activation [GO:0014719]; skeletal muscle satellite cell differentiation [GO:0014816]; skeletal muscle satellite cell proliferation [GO:0014841] |
18056409_Megf10 represents a novel transmembrane protein that impinges on Notch signaling to regulate the satellite cell population balance between proliferation and differentiation. 22407321_in mouse, two related transmembrane proteins, MEGF10 and MEGF11, have critical roles in the formation of mosaics by two retinal interneuron subtypes, starburst amacrine cells and horizontal cells 22993420_These results indicated that both Jedi-1 and MEGF10 can mediate phagocytosis independently through the recruitment of Syk. 23954233_Cysteine mutations cause defective tyrosine phosphorylation in MEGF10 myopathy. 24270812_studies reveal a novel role for astrocytes in mediating synapse elimination in the developing and adult brain, identify MEGF10 and MERTK as critical proteins in the synapse remodelling underlying neural circuit refinement 27170117_Megf10 binds with high affinity to C1q, an eat-me signal for apoptotic cells. Cells expressing Megf10 with EMARDD mutations have impaired apoptotic cell clearance and impaired binding to C1q. 28526325_Results indicate that Multiple-EGF like domains 10 (MEGF10)may be responsible for phagocytic activity targeted toward unwanted substances such as amyloid in cholinergic and glutamatergic neurons. 30802937_We find that tubular muscle subtypes are particularly sensitive to Megf10/Drpr overexpression. Complementary genetic analyses show that Megf10/Drpr and Notch may interact to regulate myogenesis. Our findings provide a basis for investigating MEGF10 in muscle development using Drosophila. 33159715_Megf10 deficiency impairs skeletal muscle stem cell migration and muscle regeneration. |
ENSG00000145794 |
MEGF10 |
366.330148 |
0.1844516356 |
-2.438686 |
0.15020703 |
261.697945 |
0.00000000000000000000000000000000000000000000000000000000007318281589345409724650386798531558069593172332639572291846443415297514501092933518630642248518148328071491540339361149602623293462483753706820206134248119056451287178788334131240844726562500000000000000000 |
0.0000000000000000000000000000000000000000000000000000000154822312734596217376709917760014462993538259254121488981487593068030379804337228489201458215856169302310244877058438259646310901100848026693684866827993573679123073816299438476562500000000000000000000000 |
Yes |
No |
110.451963521117 |
11.5205579786533 |
598.812600069045 |
41.7855750765149 |
| ENSMUSG00000024660 |
16319 |
Incenp |
protein_coding |
Q9WU62 |
FUNCTION: Component of the chromosomal passenger complex (CPC), a complex that acts as a key regulator of mitosis. The CPC complex has essential functions at the centromere in ensuring correct chromosome alignment and segregation and is required for chromatin-induced microtubule stabilization and spindle assembly. Acts as a scaffold regulating CPC localization and activity. The C-terminus associates with AURKB or AURKC, the N-terminus associated with BIRC5/survivin and CDCA8/borealin tethers the CPC to the inner centromere, and the microtubule binding activity within the central SAH domain directs AURKB/C toward substrates near microtubules. The flexibility of the SAH domain is proposed to allow AURKB/C to follow substrates on dynamic microtubules while ensuring CPC docking to static chromatin (By similarity). Activates AURKB and AURKC. Controls the kinetochore localization of BUB1. {ECO:0000250|UniProtKB:P53352, ECO:0000250|UniProtKB:Q9NQS7}. |
Alternative splicing;Cell cycle;Cell division;Centromere;Chromosome;Chromosome partition;Coiled coil;Cytoplasm;Cytoskeleton;Glycoprotein;Kinetochore;Microtubule;Mitosis;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:16319; |
central element [GO:0000801]; chromocenter [GO:0010369]; chromosome passenger complex [GO:0032133]; chromosome, centromeric region [GO:0000775]; cytosol [GO:0005829]; kinetochore [GO:0000776]; lateral element [GO:0000800]; meiotic spindle midzone [GO:1990385]; microtubule [GO:0005874]; microtubule cytoskeleton [GO:0015630]; midbody [GO:0030496]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; pericentric heterochromatin [GO:0005721]; protein-containing complex [GO:0032991]; spindle [GO:0005819]; synaptonemal complex [GO:0000795]; molecular function activator activity [GO:0140677]; protein serine/threonine kinase activator activity [GO:0043539]; chromosome segregation [GO:0007059]; meiotic spindle midzone assembly [GO:0051257]; metaphase plate congression [GO:0051310]; mitotic cell cycle [GO:0000278]; mitotic cytokinesis [GO:0000281]; mitotic spindle midzone assembly [GO:0051256]; mitotic spindle organization [GO:0007052]; positive regulation of attachment of mitotic spindle microtubules to kinetochore [GO:1902425]; positive regulation of mitotic cell cycle spindle assembly checkpoint [GO:0090267]; positive regulation of mitotic cytokinesis [GO:1903490]; positive regulation of mitotic sister chromatid separation [GO:1901970]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein serine/threonine kinase activity [GO:0071902]; protein phosphorylation [GO:0006468] |
12584241_INCENP labels the spermatogonia synaptonemal complex central element from zygotene up to late pachytene when it begins to relocalize to heterochromatic chromocentres 16386730_INCENP recruits Aurora-C (or some other factor(s) recruit INCENP and Aurora-C) to meiotic chromosomes, while Aurora-C may either work alone or cooperate with Aurora-B to regulate chromosome segregation during male meiosis |
ENSG00000149503 |
INCENP |
1602.338638 |
2.0464266374 |
1.033107 |
0.13415374 |
57.859968 |
0.00000000000002814571599474099888709402679387084826687515787257964916534547228366136550903320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000006719679404888634952858890354719253958006713878248206128773745149374008178710937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2156.42143263228 |
200.811045247474 |
1053.74968894924 |
73.9696115526082 |
| ENSMUSG00000024678 |
66607 |
Ms4a4d |
protein_coding |
Q99N05 |
FUNCTION: May be involved in signal transduction as a component of a multimeric receptor complex. |
Membrane;Receptor;Reference proteome;Transmembrane;Transmembrane helix |
Human_homologues NA; + ;NA |
|
mmu:66607; |
plasma membrane [GO:0005886] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 21460840_ABCA7 and the MS4A6A/MS4A4E gene cluster are new Alzheimer's disease susceptibility loci. 26449541_We replicated the rs11136000 (CLU)- rs670139 (MS4A4E) interaction, demonstrated an association in APOE epsilon4 negative subjects, and reported evidence of a possible dominant effect for MS4A4E. 27781389_Addition of the minor allele for rs670139 (MS4A4E), rs9331896 (CLU), and rs12155159 (NME8) was nominally associated with change on the DWRT, DSST, and WFT, respectively, in whites. For rs670139 and rs9331896 the association was only significant in individuals bearing at least one APOE 4 allele in stratified analyses.; + ;21460841_Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. 23573206_Single nucleotide polymorphisms in APOE-epsilon4, CLU and MS4A4A influence both Alzheimer disease risk and cerebrospinal fluid Amyloid-beta1-42 levels. 25717186_MS4A4 in human mast cells effects ligand-induced KIT endocytosis pathways and reduced receptor recycling to the cell surface, thus promoting KIT signaling in the endosomes while reducing that in the plasma membrane. 28005267_MS4A4A expression is significantly upregulated in human masticatory mucosa during wound healing 28303902_MS4A4A is expressed at the plasma membrane in monocytes but not in granulocytes or lymphocytes. 31263276_MS4A4A is a tetraspan molecule selectively expressed in macrophages during differentiation and polarization, essential for dectin-1-dependent activation of natural killer cell-mediated resistance to metastasis. 32240745_The FcepsilonRIbeta homologue, MS4A4A, promotes FcepsilonRI signal transduction and store-operated Ca(2+) entry in human mast cells. |
ENSG00000214787+ENSG00000110079 |
MS4A4E+MS4A4A |
20.773356 |
117.4486768384 |
6.875887 |
1.32570960 |
43.406377 |
0.00000000004447364769201607687550176281141424243503745827865714090876281261444091796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000007379331172601185897495150701433808820839388431522820610553026199340820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.245806681278 |
40.3035943104596 |
0.325638463632051 |
0.324391368312382 |
| ENSMUSG00000024769 |
240505 |
Cdc42bpg |
protein_coding |
Q80UW5 |
FUNCTION: May act as a downstream effector of CDC42 in cytoskeletal reorganization. Contributes to the actomyosin contractility required for cell invasion, through the regulation of MYPT1 and thus MLC2 phosphorylation (By similarity). {ECO:0000250|UniProtKB:Q5VT25}. |
ATP-binding;Coiled coil;Cytoplasm;Kinase;Magnesium;Metal-binding;Nucleotide-binding;Phosphoprotein;Reference proteome;Repeat;Serine/threonine-protein kinase;Transferase;Zinc;Zinc-finger |
|
|
mmu:240505; |
cell leading edge [GO:0031252]; centriolar satellite [GO:0034451]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; ATP binding [GO:0005524]; magnesium ion binding [GO:0000287]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; actin cytoskeleton reorganization [GO:0031532]; actomyosin structure organization [GO:0031032]; peptidyl-threonine phosphorylation [GO:0018107]; protein phosphorylation [GO:0006468] |
19402746_MRCKgamma can modulate maturation of dendritic spines in cultured cortical neurons, and it is localized specifically to parallel fiber/Purkinje cell synapses in vivo. 33921698_MRCKalpha Is Dispensable for Breast Cancer Development in the MMTV-PyMT Model. |
ENSG00000171219 |
CDC42BPG |
50.614886 |
0.1888456516 |
-2.404721 |
0.39176308 |
37.460692 |
0.00000000093273989651099146323379607286972389768031632684142095968127250671386718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000132039908026537367137966596662874974033030639475327916443347930908203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.6863449668463 |
4.31216222158702 |
83.0643694074829 |
15.1086809679461 |
| ENSMUSG00000024846 |
73720 |
Cst6 |
protein_coding |
Q9D1B1 |
Human_homologues FUNCTION: High affinity inhibitor for cathepsin L, cathepsin L2 (cathepsin V), and legumain (PubMed:30425301). Involved in the regulation of epidermal cornification, and hair follicle morphogenesis and maintenance (PubMed:30425301). {ECO:0000269|PubMed:30425301}. |
Proteomics identification;Reference proteome;Signal |
|
|
mmu:73720; |
cornified envelope [GO:0001533]; cysteine-type endopeptidase inhibitor activity [GO:0004869]; epidermis development [GO:0008544] |
19262604_cystatin M/E has a role in skin barrier formation and a potential role as a tumor suppressor gene [review] 20495178_conclude that a tightly regulated balance between cathepsin L and cystatin M/E is essential for tissue integrity in epidermis, hair follicles, and corneal epithelium 28596234_Using quenched activity-based probes, we identified mouse cathepsin B (CtsB), which is expressed in the lower regions of the hair follicle, as an additional target of mouse Cst6 |
ENSG00000175315 |
CST6 |
97.222605 |
2.5259668347 |
1.336836 |
0.25687501 |
26.944083 |
0.00000020942724404927651051268040587050034417870847391895949840545654296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000020768201701553253738592643568017237498679605778306722640991210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
145.877310031487 |
26.1112311052587 |
57.7510789255075 |
8.05427061248935 |
| ENSMUSG00000024868 |
13380 |
Dkk1 |
protein_coding |
O54908 |
FUNCTION: Antagonizes canonical Wnt signaling by inhibiting LRP5/6 interaction with Wnt and by forming a ternary complex with the transmembrane protein KREMEN that promotes internalization of LRP5/6 (PubMed:18524778). Inhibits the pro-apoptotic function of KREMEN1 in a Wnt-independent manner, and has anti-apoptotic activity (PubMed:26206087). Plays a role in limb development; attenuates Wnt signaling in the developing limb to allow normal limb patterning (PubMed:18505822). {ECO:0000269|PubMed:18505822, ECO:0000269|PubMed:18524778, ECO:0000269|PubMed:26206087}. |
Developmental protein;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:13380; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; co-receptor binding [GO:0039706]; low-density lipoprotein particle receptor binding [GO:0050750]; receptor antagonist activity [GO:0048019]; canonical Wnt signaling pathway [GO:0060070]; cell morphogenesis involved in differentiation [GO:0000904]; embryonic limb morphogenesis [GO:0030326]; endoderm development [GO:0007492]; endoderm formation [GO:0001706]; face morphogenesis [GO:0060325]; forebrain development [GO:0030900]; hair follicle development [GO:0001942]; head morphogenesis [GO:0060323]; learning or memory [GO:0007611]; limb development [GO:0060173]; mesoderm formation [GO:0001707]; motor learning [GO:0061743]; negative regulation of apoptotic process [GO:0043066]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of canonical Wnt signaling pathway involved in cardiac muscle cell fate commitment [GO:1901296]; negative regulation of cardiac muscle cell differentiation [GO:2000726]; negative regulation of mesodermal cell fate specification [GO:0042662]; negative regulation of neuron projection development [GO:0010977]; negative regulation of ossification [GO:0030279]; negative regulation of pathway-restricted SMAD protein phosphorylation [GO:0060394]; negative regulation of peptidyl-serine phosphorylation [GO:0033137]; negative regulation of presynapse assembly [GO:1905607]; negative regulation of protein binding [GO:0032091]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of Wnt signaling pathway [GO:0030178]; negative regulation of Wnt-Frizzled-LRP5/6 complex assembly [GO:1904723]; positive regulation of gene expression [GO:0010628]; positive regulation of heart induction by negative regulation of canonical Wnt signaling pathway [GO:0090082]; positive regulation of JUN kinase activity [GO:0043507]; positive regulation of midbrain dopaminergic neuron differentiation [GO:1904958]; positive regulation of neuron death [GO:1901216]; positive regulation of tau-protein kinase activity [GO:1902949]; regulation of endodermal cell fate specification [GO:0042663]; regulation of receptor internalization [GO:0002090]; regulation of synapse organization [GO:0050807]; regulation of synaptic transmission, glutamatergic [GO:0051966]; regulation of Wnt signaling pathway [GO:0030111]; response to retinoic acid [GO:0032526]; Wnt signaling pathway [GO:0016055]; Wnt signaling pathway involved in somitogenesis [GO:0090244] |
11867524_In mouse embryo limb buds, Dkk-1 is expressed in anterior mesenchyme. In Shh knockouts, the level is decreased. 12050670_Kremen proteins are Dickkopf receptors that regulate Wnt/beta-catenin signalling. 12952897_Dkk1 and noggin functionally cooperate in the head organizer. 14695885_Adenovirus Dkk1 treatment of adult mice repressed expression of the Wnt target genes CD44 and EphB2 within 2 days in both small intestine and colon. 15115753_compensatory interactions between Dkk1 and Lrp6 demonstrate the importance of correctly balancing positive and negative regulation of the signaling during mammalian development. 15758175_The induction of Dkk-1 with the ensuing inhibition of Wnt signal pathway is required for development of ischemia and cell death. 16195809_Dkks were involved in craniofacial developmental processes, including facial outgrowth, myogenesis, osteogenesis, palatogenesis, olfactory epithelium and tooth development; and the expression persisted to postnatal stage in the muscles and bones. 16256739_The defective axis conversion due to Otx2 deficiency can be rescued by expression of Dkk1, a Wnt antagonist, or following removal of one copy of the beta-catenin gene. 16521043_Dkk1 was first seen in the perichondral mesenchyme in restricted domains and later in the migrating mesenchymal cells within the cartilage. 16763196_The involvement of Dkk1 in human but not murine adipogenesis indicates that inter-species differences exist in the molecular control of this process. 17068150_DKK1 is a key player in multiple myeloma bone disease 17068196_ET-1 increases osteoblast proliferation and new bone formation by activating the Wnt signaling pathway through suppression of the Wnt pathway inhibitor DKK1. 17212358_VDR-mediated inhibition of Dkk1 suppresses adipogenic differentiation of bone marrow stromal cells. 17613296_DKK1 is a negative regulator of normal bone homeostasis in vivo. 17700537_concludes that bone-derived prostate cell line that produces osteoblastic lesions induces new bone formation through Wnt canonical signaling, that LRP5 mediates this effect, and that DKK1 is involved in the balance between bone formation and resorption 17964517_Dkk-1/Wnt/beta-catenin cascade may mediate the proliferation and migration of hepatocellular carcinoma cells during the metastasis process. 17971207_WNT signaling attenuation by DKK-1 and -2 regulates BMP2-induced osteoblast differentiation and expression of OPG, RANKL and M-CSF 18068158_Dkk1 and Kremen1 play roles in blastocyst activation and uterine receptivity during the window of implantation. 18157634_DKK-1 mediated tumor suppressor effect is independent of beta-catenin dependent transcription and a CamKII pathway contributes to DKK-1 signaling 18174290_Modulation of Dickkopf-1 attenuates glucocorticoid induction of osteoblast apoptosis, adipocytic differentiation, and bone mass loss. 18294945_Dkk1 inhibits this process and may be a key factor regulating pre-osteoblast differentiation and myeloma bone disease 18297060_MesP1 drives vertebrate cardiovascular differentiation through Dkk-1-mediated blockade of Wnt-signalling 18403408_head development is sensitive to the level of WNT3 signalling and that DKK1 is the key antagonist that modulates WNT3 activity during anterior morphogenesis. 18502762_Kremen may not be essential for Dkk1-mediated Wnt antagonism and Kremen may only play a role when cells express a high level of LRP5/6 18505732_DKK1 inhibition of LRP6 is independent of LRP6 internalization and degradation 18613030_oxysterol-induced osteogenic differentiation is mediated through a Wnt signaling-related, Dkk-1-inhibitable mechanism. 18653765_FHL2-beta-catenin interaction potentiates beta-catenin nuclear translocation and TCF/LEF transcription, resulting in increased Runx2 and alkaline phosphatase expression, which was inhibited by the Wnt inhibitor DKK1. 18827832_Dkk-1 antagonists or drugs that rescue the Wnt pathway might be neuroprotective in stroke 18951693_Wnt pathway inhibitors, Axin (intracellular) or Dickkopf-1 (extracellular) blocked the regulation of VCAM-1 by diffusible Wnt3a. 19016584_Inhibiting Dkk1 prevents the suppression of bone formation and in doing so is effective in preventing the development of osteolytic bone disease in myeloma. 19057969_The results demonstrate that Dkk1 overexpression can initiate the differentiation program of ESCs toward neuroectoderm. 19874086_BMPRIA in osteoblasts negatively regulates endogenous bone mass and Wnt/beta-catenin signaling and that this regulation may be mediated by the activities of Sost and Dkk1 19936252_Lrp4 is a novel osteoblast expressed Dkk1 and sclerostin receptor with a physiological role in the regulation of bone growth and turnover 19943155_Thiazolidinediones increase the wingless-type MMTV integration site family (WNT) inhibitor Dickkopf-1 in adipocytes 20050968_This study indicated that DKK-1 expression is associated with events that lead to neuronal death in neurodegenerative diseases 20063381_The effects of Dkk1 Ab treatment depended on the timing of initiation in fracture healing of femur fracture 20093360_analysis of the frizzled8.Wnt3a.LRP6 signaling complex reveals multiple Wnt and Dkk1 binding sites on LRP6 20142103_The study indicates that full actions of parathyroid hormone (PTH) require intact Wnt signaling but that PTH can activate the Wnt pathway despite overexpression of Dkk1. 20144607_Regulation of Dkk1 expression was investigated during embryonic development in a transgenic mouse line. 20161711_Data show that removal of canonical Wnt inhibitor DKK1 from DKK1-involuted mice results in full recovery, suggesting that canonical Wnt signaling is required for the differentiation or proliferation of TEC populations. 20211162_In embryos with an epiblast-specific deletion of Bmpr1a (Bmpr1a(null/flox); Sox2Cre embryos), the anterior visceral endoderm cells migrate randomly from the distal end of embryos, resulting in an expansion of the anterior visceral endoderm. 20439124_Both Dkk1 and Dkk2 inhibit Wnt signaling that regulates early myocardial proliferation; each compensates for the loss of the other in that role. 20847303_DKK1 enhances angiogenic properties of endothelial stem cellss (ECFC) in vitro and is required for ECFC and mesenchymal stem cell angiogenic phenotypes in vivo. DKK1 also increases tumoral angiogenesis. 20929859_Generation and selection of novel fully human monoclonal antibodies that neutralize Dickkopf-1 (DKK1) inhibitory function in vitro and increase bone mass in vivo. 21304589_induction of Dkk-1 is causally related to stress-induced hippocampal damage and Dkk-1 expression is regulated by corticosteroids in the central nervous system 21347250_Dkk1 is required for midbrain dopaminergic differentiation and morphogenesis 21351149_miR-335-5p activates Wnt signaling and promotes osteogenic differentiation by downregulating DKK1. 21354128_Dkk1 controls the degree of Wnt-7b signalling in the papilla to coordinate kidney organogenesis. 21436291_beta-catenin causes renal dysplasia via upregulation of Tgfbeta2 and Dkk1. 21440550_Depletion of Dkk1 induces a strong proliferative response that promotes wound repair after colitis. 21539829_The retinal Dkk1 gene dose is critical during eye development. Dkk1 and Shh coordinatively control anterior head formation and eye induction. During eye development, retinal Dkk1 activation is depending on cilia mediated Gli3 regulation. 21540552_there are distinct functions of DKK1 and DKK2 in controlling angiogenesis 21607803_Quantitative real-time RT-PCR and Western blot analysis showed that the expression of DKK1 mRNA and protein in 32 ovarian serous papillary adenocarcinoma (OSC) tissues were elevated as compared with those in 10 normal ovarian tissues 21654186_Data propose that DKK1 may play a protective role that is not unmasked by loss-of-function in the absence of other stressors. 21773994_These data suggest that, whereas Dkk1 significantly regulates bone formation in younger animals, its role in older animals is limited to pathologies that lead to the induction of Dkk1 expression in bone and/or serum, such as traumatic injury. 21917600_controls post-natal mandibular molar dentin formation either directly or indirectly via the inhibition of Wnt signaling 21945937_These results suggest that the Acvr1-deficiency can increase bone mass by activating Wnt signaling in which both Sost and Dkk1 expression levels are diminished. 22022411_The negative feedback of the Wnt signaling pathway involves Dkk1 rather than Axin2. 22358062_DKK-1 is involved in anagen-to-catagen transition in the hair cycle by regulating the activity of follicular keratinocytes. 22374979_Knockdown of Dkk1 expression in BMSCs decreased their ability to promote myeloma and the associated bone disease in mice. Results show novel roles of BMSCs and BMSC-derived Dkk1 in the pathogenesis of multiple myeloma in vivo. 22406973_identify that mesenchymal stem cells from Prx1-Dkk1 mice have limited proliferative ability, but normal differentiation potential, which may explain the mechanism for the limb reduction defects observed 22407773_Dkk1 misexpression in endothelial cells resulted in defects of endochondral ossification and reduced skeletal size. 22459449_these findings support our hypothesis that Dkk1 is a direct target of Osx. 22488261_Expression of Dkk1 in chondrocytes inhibits osteoarthritic cartilage destruction. The protective effect of Dkk-1 appears to be associated with its capacity to inhibit Wnt-mediated expression of catabolic factors. 23152495_Matrix rigidity activates Wnt signaling through down-regulation of Dickkopf-1 protein 23171658_Data indicate that both Dickkopf-1 (DKK1) and sclerostin (SOST) were downregulated in proteoglycan-induced spondylitis (PGISp)-affected mouse spines. 23302695_DKK-1 acts predominantly by inhibiting PDGF-, TGF-beta-, and connective tissue growth factor-activated MAPK and JNK signaling cascades, acting via LRP-6 with associated WNT ligand. 23395445_Findings show that upregulating Wnt signaling by reducing Dkk1 expression can counteract age-related decrease in neurogenesis and its associated cognitive decline. 23636253_DKK1 and PTEN serve as competitive endogenous RNA, affecting the expression of each other via competition for miRNAs binding in the diabetic heart. 23685555_Dkk1 enhances endothelial-mesenchymal transition in aortic endothelial cells. 24091497_These findings demonstrate that DKK1 and DKK2 have differential roles in normalization and functionality of tumor blood vessels, in addition to angiogenesis. 24139798_Dkk1 Overexpression Inhibits Osteosarcoma Growth and Angiogenesis. 24447645_up-regulation of Dkk1 by HR contributes to abnormal development of HFs and failure in regeneration of HFs in Hr(Hp)/Hr(Hp) mice 24460718_When cellular Bmal1 was increased by transfection, osteogenesis inhibition by Dkk1 was rescued to a certain extent with activation of Wnt signalling. 24479159_formation of anus and urethral depends on Dkk1-mediated dynamic inhibition of the canonical Wnt/ss-catenin signal pathway. 24520934_Wnt inhibition during the first day has larger impact on the activation of Hesx1 and Six3, suggesting that in embryos Wnt inhibition caused by Dkk1, contributes greatly in the establishment of the anterior forebrain precursor. 24528599_DKK-1 is a novel target of the mevalonate pathway that is suppressed by zoledronic acid and atorvastatin in breast cancer. 24618599_extra-follicular modulators Bmp2, Dkk1, and Sfrp4 increase in early anagen. 24747916_results indicate that Wnt signaling may mediate the profibrotic effect of PSC activation, and Wnt2/Dkk-1 could be potential therapeutic targets for CP 24750467_Wnt10b/DKK1 can modulate hair follicle size during hair regeneration. 24819581_The combination of Dkk-1 overexpression by Dex and hypoxia causes the necrotic osteocytic cell death. 24848709_Overexpression of BRAF(V600E) in normal thyroid epithelial (H tori) cells also reduced the effects of Dkk-1 on cell survival. 25080367_Dkk-1-mediated inhibition of Wnt signaling in bone ameliorates osteoarthritis in mice. 25231759_OTX2 has a role in regulating Dkk1 and Lhx1 activity in the anterior mesendoderm 25603465_When Dkk1(+/-) mice were crossed with Bmp2(c/c);Prx1::cre mice, the offspring bearing both genetic alterations were unable to increase bone mass and heal fractures 25808845_These in vivo data support in vitro studies regarding the mechanism of HBM-causing mutations, and imply that HBM LRP5 receptors differ in their relative sensitivity to inhibition by SOST and DKK1. 25910078_R-spondin 1/dickkopf-1/beta-catenin machinery is involved in testicular embryonic angiogenesis 25910836_OTX2 and DKK1 exhibit asymmetric expression in the anterior ventral endoderm during embryonic anterior-posterior axis formation. 26206087_Dickkopf1 receptor Kremen1 is a bona fide dependence receptor, triggering cell death unless bound to its ligand. 26218891_thyroid hormone-induced changes in bone remodeling are associated with a divergent regulation of DKK1 and sclerostin 26351298_platelet-derived Dickkopf-1 (Dkk1) as the major Wnt antagonist contributing to the suppression of Wnt/beta-catenin signaling in AECs during acute lung inflammation. 26607269_Disturbed oscillatory flow increases DKK1 expression. DKK1 knockdown attenuates OSS-induced monocyte adhesion and endothelial impairment. Genetic silencing of DKK1 limits atherogenesis in ApoE-/- mice. 26648540_These results suggest that inhibiting Wnt signaling in embryonic stem cells using DKK1 may promote mouse ESCs to differentiate into endoderm in vitro and in vivo 26674341_that Dkk1 negatively regulates BMP9-induced osteogenic differentiation. 26872695_produced by platelets, promotes Th2 polarization and pathological leukocyte infiltration 27045006_Recombinant Dkk1 suppresses beta-catenin target genes in myeloid-derived suppressor cells (MDSCs) from mice and humans and anti-Dkk1 loses its antitumor effects in mice lacking beta-catenin in myeloid cells or after depletion of MDSCs, demonstrating that Dkk1 directly targets MDSCs. 27393149_Collectively, miR-9 promotes osteoblast differentiation of mesenchymal stem cells by suppressing DKK1 gene expression. 27460630_Diabetic conditions increase the expression of DKK1. 27918563_Dkk1 promotes hematopoietic regeneration via direct effects on hematopoietic stem cells, decreasing the levels of mitochondrial reactive oxygen species and suppressing senescence, and via indirect effects on bone marrow endothelial cells, inducing EGF secretion. 28155238_Collectively, our data strongly suggest that stress-related neurohormones, such as GCs, cause DP cells to secrete DKK1, which in turn inhibits hair growth. 28177772_A negative feedback loop involving WNT6 and DKK1 attenuates canonical WNT6-beta-catenin signaling, thereby allowing teratocarcinoma cells to differentiate. 28556921_Membrane-bound DKK-1 is a novel Foxp3 postive Treg cell derived mediator to maintain immunological tolerance in T-cell-mediated autoimmune colitis. 28892080_DKK1 prevents lung metastasis of breast cancer cells but promotes breast-to-bone metastasis. 29105022_Dynamic histomorphometry identified increased bone formation as the mechanism underlying the high bone mass phenotype in Dkk1 KO mice, with no changes in bone resorption 29704238_It is a direct target of miR- 3064- 3p. 29734465_lack of Dkk1 in cells of the osteoblastic lineage leads to high bone mass with increased bone formation, despite increased levels of sclerostin. 29845410_In this study we aimed to explore the regulatory interaction between DKK1 and sclerostin within the bone compartment 30216540_study discovers a functional TIMP/metalloproteinase-TNFalpha-Dkk1/RANKL nexus for optimal control of the bone microenvironment, which dictates coexistence of the osteoblast and osteoclast lineages. 30575941_miR-488 promotes fracture healing by targeting DKK1 expression in osteoblasts. 30689850_Loss of Dkk1 is not sufficient to fully reverse thyroid hormone-induced changes in bone mass and bone turnover. 30779862_Osteogenic Dkk1 Mediates Glucocorticoid-Induced but Not Arthritis-Induced Bone Loss. 30890376_The results indicate that silencing GSK3beta instead of DKK1 can inhibit osteogenic differentiation caused by co-exposure to fluoride and arsenic. 31081743_Dkk1 induces regenerative function in the EPCs 31611558_Inhibition of Tet1- and Tet2-mediated DNA demethylation promotes immunomodulation of periodontal ligament stem cells. 32057178_LncRNA TUG1 alleviates cardiac hypertrophy by targeting miR-34a/DKK1/Wnt-beta-catenin signalling. 32521258_Rab7-Mediated Endocytosis Establishes Patterning of Wnt Activity through Inactivation of Dkk Antagonism. 33085496_Balanced Wnt/Dickkopf-1 signaling by mesenchymal vascular progenitor cells in the microvascular niche maintains distal lung structure and function. 33479403_Role of osteogenic Dickkopf-1 in bone remodeling and bone healing in mice with type I diabetes mellitus. 35249365_Upregulation of Endothelial DKK1 (Dickkopf 1) Promotes the Development of Pulmonary Hypertension Through the Sp1 (Specificity Protein 1)/SHMT2 (Serine Hydroxymethyltransferase 2) Pathway. 35278242_Suppression of Sost/Sclerostin and Dickkopf-1 Augment Intervertebral Disc Structure in Mice. 35426038_Dickkopf1 (Dkk1) Alleviates Vascular Calcification by Regulating the Degradation of Phospholipase D1 (PLD1). 35924447_DKK1 drives immune suppressive phenotypes in intrahepatic cholangiocarcinoma and can be targeted with anti-DKK1 therapeutic DKN-01. 36206576_DKK1 Promotes Tumor Immune Evasion and Impedes Anti-PD-1 Treatment by Inducing Immunosuppressive Macrophages in Gastric Cancer. 36738214_[Dickkopf-1 inhibits the secretion of MUC5AC induced by Mycoplasma pneumoniae P1-C in mouse lung epithelial cells]. |
ENSG00000107984 |
DKK1 |
75.386952 |
0.0741165527 |
-3.754060 |
0.38869689 |
98.908154 |
0.00000000000000000000002644880806809690025879605904999341105439117708957122927308691011198671372284252356621436774730682373046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000012789468079150855906159220916577253223198660921519876173440069533349117136822314932942390441894531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.0701121180083 |
2.74607487696016 |
135.868598259923 |
21.6018926131864 |
| ENSMUSG00000024989 |
74107 |
Cep55 |
protein_coding |
Q8BT07 |
FUNCTION: Plays a role in mitotic exit and cytokinesis. Recruits PDCD6IP and TSG101 to midbody during cytokinesis. Required for successful completion of cytokinesis. Not required for microtubule nucleation. Plays a role in the development of the brain and kidney. {ECO:0000250|UniProtKB:Q53EZ4}. |
Alternative splicing;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Mitosis;Phosphoprotein;Reference proteome |
|
|
mmu:74107; |
centriole [GO:0005814]; centrosome [GO:0005813]; cleavage furrow [GO:0032154]; cytoplasm [GO:0005737]; Flemming body [GO:0090543]; intercellular bridge [GO:0045171]; midbody [GO:0030496]; identical protein binding [GO:0042802]; cranial skeletal system development [GO:1904888]; establishment of protein localization [GO:0045184]; midbody abscission [GO:0061952]; mitotic cytokinesis [GO:0000281]; regulation of phosphatidylinositol 3-kinase signaling [GO:0014066] |
20176808_TEX14 prevents the completion of cytokinesis by altering the destiny of CEP55 from a nidus for abscission to an integral component of the intercellular bridge. 20186884_The results suggest that Cep55 and pericentrin are required for the stable bridge between germ cells during spermatogenesis and spermiogenesis. 26582107_Cep55 may act as an MTOC-associated protein regulating spindle organization, and thus cell cycle progression during mouse oocyte meiotic maturation. 29683733_that Cep55 overexpression causes change in germ cell proportions and manifests as a Sertoli cell only tubule phenotype 31427428_Study found that Cep55-depleted mouse oocytes progress normally through early meiosis I, but that anaphase I fails as a result of persistent Cdk1 activity. 32269212_This work defines Cep55 as an abscission regulator. 33087841_Cep55 overexpression promotes genomic instability and tumorigenesis in mice. 33095425_MicroRNA-449a Suppresses Mouse Spermatogonia Proliferation via Inhibition of CEP55. 33475699_CEP55 promotes cilia disassembly through stabilizing Aurora A kinase. 33622776_Loss of Coiled-Coil Protein Cep55 Impairs Neural Stem Cell Abscission and Results in p53-Dependent Apoptosis in Developing Cortex. |
ENSG00000138180 |
CEP55 |
832.663890 |
2.5683342898 |
1.360833 |
0.11224922 |
144.367158 |
0.00000000000000000000000000000000295337087007922589179626653377620047443225697924883581739723453456736415846891348859545173288765340657846536487340927124023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000002646220299590986048724838733059207894158906430927924072322436673843415346721075513333754081202187080634757876396179199218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1195.74259322129 |
93.128927094614 |
465.571245129844 |
27.9891473232903 |
| ENSMUSG00000025083 |
226250 |
Afap1l2 |
protein_coding |
Q5DTU0 |
FUNCTION: May play a role in a signaling cascade by enhancing the kinase activity of SRC. Contributes to SRC-regulated transcription activation (By similarity). {ECO:0000250}. |
Alternative splicing;Coiled coil;Cytoplasm;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:226250; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; plasma membrane [GO:0005886]; protein tyrosine kinase activator activity [GO:0030296]; SH2 domain binding [GO:0042169]; SH3 domain binding [GO:0017124]; inflammatory response [GO:0006954]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of epidermal growth factor receptor signaling pathway [GO:0045742]; positive regulation of interleukin-8 production [GO:0032757]; regulation of interleukin-6 production [GO:0032675]; regulation of mitotic cell cycle [GO:0007346] |
27029000_we showed that XB130 might play protective roles in lipopolysaccharide-induced systemic septic response and acute lung injury 30820526_XB130 acts as a tumor suppressor in carcinogen-induced skin tumorigenesis that may be mediated through inhibiting inflammation. |
ENSG00000169129 |
AFAP1L2 |
734.758289 |
0.2160672729 |
-2.210448 |
0.12105461 |
317.377308 |
0.00000000000000000000000000000000000000000000000000000000000000000000005397436140240085560456114507162494585609453798918315414653977062172410483131775778384298525748428006839291805356021932851560890415482739964064449508367570531467123327139928917056677093455618888 |
0.0000000000000000000000000000000000000000000000000000000000000000000167783157730891787857728389628282922721367668172978008865873397719465711057331409614957395385899916602593988542697512681214119286402078762815593673732368911863056107412577044879498089358094148 |
Yes |
Yes |
263.402604776879 |
22.1277047143801 |
1219.07682383464 |
74.3478708345284 |
| ENSMUSG00000025089 |
14585 |
Gfra1 |
protein_coding |
P97785 |
FUNCTION: Receptor for GDNF. Mediates the GDNF-induced autophosphorylation and activation of the RET receptor (By similarity). {ECO:0000250|UniProtKB:Q62997}. |
Alternative splicing;Cell membrane;Disulfide bond;Endosome;Glycoprotein;Golgi apparatus;GPI-anchor;Lipoprotein;Membrane;Receptor;Reference proteome;Repeat;Signal |
|
This gene encodes a transmembrane protein that functions as the receptor for glial cell line derived neurotrophic factor (GDNF). The encoded protein undergoes proteolytic processing to generate a glycosylphosphatidylinositol-anchored cell surface coreceptor that forms a complex with the Ret tyrosine kinase in GDNF signaling pathway. Mice lacking the encoded protein exhibit deficits in the kidneys, the enteric nervous system, and spinal motor and sensory neurons similar mice deficient in GDNF or Ret. [provided by RefSeq, Jul 2016]. |
mmu:14585; |
axon [GO:0030424]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; extrinsic component of membrane [GO:0019898]; Golgi apparatus [GO:0005794]; multivesicular body [GO:0005771]; neuronal cell body [GO:0043025]; plasma membrane protein complex [GO:0098797]; receptor complex [GO:0043235]; integrin binding [GO:0005178]; neurotrophin receptor activity [GO:0005030]; signaling receptor activity [GO:0038023]; aging [GO:0007568]; anatomical structure morphogenesis [GO:0009653]; cell migration [GO:0016477]; kidney development [GO:0001822]; male gonad development [GO:0008584]; nervous system development [GO:0007399]; neuron differentiation [GO:0030182]; neuron projection development [GO:0031175]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
12697293_Following ischemic injury, GFR alpha-1 is differentially up-regulated in GFR alpha-1 (+/+) and GFR alpha-1 (+/-) mice. 12884298_GDNF/GFRalpha1 are involved in the survival and differentiation of double negative immature thymocytes. 15237212_localization pattern of c-kit, Notch-1 and GFRalpha-1 in postnatal, immature and adult testes 15389609_GDNF in type-II cells may exert trophic actions on type-I, -II, and -III taste bud cells by binding to their GFR alpha 1 receptors. 15708562_GFR alpha-1, the receptor for GDNF is strongly expressed by a subset of type A spermatogonia. 15748846_The role of GFRalpha1 in the developing brain is to promote the differentiation and migration of cortical GABAergic neurons. 15905075_Jagged1 and GDNF/Ret/GFRalpha1 interact and have a role in regulating ureteric budding and branching 15960316_GDNF- and GFRalpha1-immunoreactive taste bud cells after denervation vanished following the disappearance of the taste buds and reappeared at the same time as the taste buds reappeared 15979200_Data suggest that the combinatorial interactions of the spliced isoforms of GFRalpha, RET and NCAM may contribute to the pleiotropic biological responses. 16014811_Spermatogonial stem cells expresses GFRA1, but also the stem cell pool is heterogeneous with respect to the level of GFRA1 expression 16551639_Gas1 is related to the GDNF alpha receptors and regulates Ret signaling 17507417_Conditional ablation of GFRalpha1 in postmigratory enteric neurons triggers unconventional neuronal death in the colon and causes a Hirschsprung's disease phenotype. 17625109_Gfra1 is expressed dominantly in mouse spermatogonial stem cells; Gfra1 knockdown leads to their differentiation via the inactivation of RET tyrosine kinase, suggesting an essential role for Gfra1 in spermatogonial stem cell regulation. 18551627_The absence of Gfr alpha1 had no effect on either the appearance of brain-derived fatty acid binding protein (B-FABP) cells or the relative area of the ganglia occupied by B-FABP-immunoreactive cells. 18650792_AT1R-mediated inhibition of the Spry1 gene increases c-Ret tyrosine kinase activity leading to upregulation of its downstream target Wnt11, which induces GDNF in adjacent mesenchyme causing focal bursts of ureteric bud tip cell proliferation 18822276_The findings point to increased sensitivity of the DAergic system with age and neurotoxic exposure as a result of a genetic reduction of GFR alpha1. 18845535_analysis of how GDNF.GFR alpha 1 can mediate cell adhesion and how heparin might inhibit GDNF signaling through RET 18973577_The present study clearly establishes that genetic reductions in GFRalpha-1 can contribute to the degenerative changes observed in this system during the aging process. 19710321_From the data of this study suggested that GFR1 signaling guides the development of a subset of parvalbumin-expressing GABAergic interneurons populating discrete regions of the cerebral cortex. 20856864_Histone methylation and acetylation regulate Pou5f1 and Gfra1 in male germ cell. 22143971_GFRA1 expression is regulated by GDNF and in cultures of isolated seminiferous tubules, we found that GDNF expression and secretion by Sertoli cells is stage-dependent, being maximal in stages II-VI and decreasing thereafter. 22745058_Donor-derived GFRalpha1-positive cells formed several aggregates of A(al(aligned)) /morula-like structures in a single spermatogenic cell patch before and on day 14 post-transplant, indicating involvement in the formation of a stable spermatogenic colony. 23197722_GFRalpha1 is critical for the development and function of the main olfactory system, contributing to the development and allocation of all major classes of neurons and glial cells. 23300832_our data demonstrate that RET/GFRalpha signals are dispensable for thymic T cell development in vivo 23333276_SorLA acts as sorting receptor for the GDNF/GFRalpha1 complex, directing it from the cell surface to endosomes. 23542432_Gfra1 expression in the urinary tract strictly depends on cell-type and specific stage in kidney development. 24778213_These findings collectively demonstrate that GFRalpha1 released by nerves enhances perineural invasion through GDNF-RET signaling and that GFRalpha1 expression by cancer cells enhances but is not required for it. 26962690_LncRNA033862 is an antisense transcript of the GDNF receptor alpha1 (Gfra1) that lacks protein coding potential and regulates Gfra1 expression levels by interacting with Gfra1 chromatin. 27255992_GDNF signals were able to induce the stratified aggregate formation of GFRalpha1-positive undifferentiated spermatogonia 27707798_The GDNF-GFRalpha1 complex is essential for proper hippocampal circuit development. 27754745_GFRA1 regulates AMPK-dependent autophagy by promoting SRC phosphorylation independent of proto-oncogene RET kinase 28076782_the neural cell adhesion molecule NCAM is co-expressed and directly interacts with GFRalpha1 in embryonic Purkinje cells. 28442042_Western blot and immunofluorescence analyses showed two GIP-anchored proteins were clearly involved in the fertilisation process, namely growth arrest specific 1 (Gas1) and glial cell line-derived neurotrophic factor receptor alpha1 (Gfralpha1). This was demonstrated by evaluating oocyte fertilisability after treatment of oocytes with antibodies against the selected proteins, with their respective short interference RN... 28591570_Compromised Survival of Cerebellar Molecular Layer Interneurons Lacking GDNF Receptors GFRalpha1 or RET Impairs Normal Cerebellar Motor Learning. 29337115_these data provide support for a model of a stem cell pool in which the GFRa1+ and GFRa1- cells are closely related but show key cell-intrinsic differences and can interconvert between the two states based, in part, on access to niche factors. 29339164_We further find that overexpression of NANOS2 results in the repression of GFRA1 and PLZF in gonocytes, leading to a delay in GST. On the other hand, loss of NANOS2 results in the up-regulation of GFRA1 and PLZF, indicating a precocious entry of GST. 30594740_goblet cells may be a potential target for preventative treatment and that reduced expression of GFRa1 may contribute to Hirschsprung's disease susceptibility. 31041846_glial cell line-derived neurotrophic factor (GDNF) was identified as an essential factor for FGSCs self-renewal with a more complicated mechanism: GDNF-GFRA1 activates AKT3 via PI3K or Src family kinase (SFK), and SFK upregulates its target genes, Bcl6b, Etv5, and Lhx1, to promote self-renewal of Female germline stem cells. 31875542_GDNF and GFRalpha1 Are Required for Proper Integration of Adult-Born Hippocampal Neurons. 34748545_Adult medial habenula neurons require GDNF receptor GFRalpha1 for synaptic stability and function. |
ENSG00000151892 |
GFRA1 |
30.762926 |
0.3313598006 |
-1.593530 |
0.50552088 |
9.786960 |
0.00175753833712703386692177698336081448360346257686614990234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0076022070904354765302768370816011156421154737472534179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.8425023048669 |
5.66551500071836 |
47.8105741109625 |
12.0930570925172 |
| ENSMUSG00000025170 |
217371 |
Rab40b |
protein_coding |
B1ATT4 |
Human_homologues FUNCTION: May be a substrate-recognition component of a SCF-like ECS (Elongin-Cullin-SOCS-box protein) E3 ubiquitin ligase complex which mediates the ubiquitination and subsequent proteasomal degradation of target proteins. {ECO:0000250}. |
Reference proteome |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000256|ARBA:ARBA00004906}. |
|
Human_homologues hsa:10966; |
nuclear envelope [GO:0005635]; perinuclear region of cytoplasm [GO:0048471]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; intracellular signal transduction [GO:0035556]; protein ubiquitination [GO:0016567]; toxin transport [GO:1901998] |
27909076_This is the first study that identifies a new Rab40b-Tks5- and miR-204-dependent invadopodia transport pathway that regulates MMP2 and MMP9 secretion, and extracellular matrix remodeling during cancer progression. |
ENSG00000141542 |
RAB40B |
153.421558 |
0.4303487620 |
-1.216422 |
0.27390190 |
19.479003 |
0.00001017116123801710701529329994308170626027276739478111267089843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000752804314759361408414262140098571762791834771633148193359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
95.87261159712 |
18.3113741969911 |
222.77887158998 |
30.6592696661929 |
| ENSMUSG00000025185 |
67573 |
Loxl4 |
protein_coding |
Q924C6 |
FUNCTION: May modulate the formation of a collagenous extracellular matrix. |
Copper;Disulfide bond;Glycoprotein;LTQ;Metal-binding;Oxidoreductase;Reference proteome;Repeat;Secreted;Signal;TPQ |
|
|
mmu:67573; |
collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; membrane [GO:0016020]; receptor complex [GO:0043235]; copper ion binding [GO:0005507]; protein-lysine 6-oxidase activity [GO:0004720]; scavenger receptor activity [GO:0005044]; collagen fibril organization [GO:0030199]; peptidyl-lysine oxidation [GO:0018057] |
28060764_These results demonstrate that weak LOXL4 expression leads to remodeling of the extracellular matrix through induction of collagen synthesis, deposition, and structural changes. 33807332_LOXL4 Abrogation Does Not Exaggerate Angiotensin II-Induced Thoracic or Abdominal Aortic Aneurysm in Mice. |
ENSG00000138131 |
LOXL4 |
6830.138808 |
0.3306025798 |
-1.596830 |
0.08140367 |
370.223799 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000016728851059272420814404142111180213154441834315116266215805131605826935839155180153730675966178657984214407220639479883258916833628714457351098960481809770192381562711551696487083080 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000637034648337093810829897317870053148993901768328216209021990108797316288243099801872210490166481333164041173181833341235600780560533556515311737966670930794754597617378050593936298 |
Yes |
Yes |
3353.99441616546 |
189.348655247627 |
10145.0945066447 |
424.607656103413 |
| ENSMUSG00000025213 |
107250 |
Kazald1 |
protein_coding |
Q8BJ66 |
FUNCTION: Involved in the proliferation of osteoblasts during bone formation and bone regeneration. Promotes matrix assembly. {ECO:0000269|PubMed:15555553, ECO:0000269|PubMed:18757743}. |
Developmental protein;Differentiation;Disulfide bond;Extracellular matrix;Glycoprotein;Immunoglobulin domain;Osteogenesis;Reference proteome;Secreted;Signal |
|
|
mmu:107250; |
extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; interstitial matrix [GO:0005614]; insulin-like growth factor binding [GO:0005520]; cell differentiation [GO:0030154]; extracellular matrix organization [GO:0030198]; ossification [GO:0001503]; regulation of cell growth [GO:0001558]; regulation of signal transduction [GO:0009966] |
15261838_Expression of Bono1 was detected in osteoblasts and secretory odontoblasts by in situ hybridization. 15555553_These results suggest that IGFBP-rP10 is involved in the proliferation of osteoblasts during bone formation and bone regeneration. |
ENSG00000107821 |
KAZALD1 |
2267.357869 |
0.3941709727 |
-1.343107 |
0.06405194 |
433.391739 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000296949757631032611025968236684685900529082526763824397078442659655892999523174832001925154095144482870394567185722730639066506812086416068996199574399544648755868307025 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000015597030028399617030079622005443045629221047292147860580415273756980311082183417305399152925941060024059993548557164148495481841150110868607537826190479284629165441827 |
Yes |
No |
1285.39119859085 |
57.0868043042925 |
3260.99912846672 |
104.378192010132 |
| ENSMUSG00000025277 |
66082 |
Abhd6 |
protein_coding |
Q8R2Y0 |
FUNCTION: Lipase that preferentially hydrolysis medium-chain saturated monoacylglycerols including 2-arachidonoylglycerol (PubMed:18096503, PubMed:20657592). Through 2-arachidonoylglycerol degradation may regulate endocannabinoid signaling pathways (PubMed:18096503, PubMed:20657592). Also has a lysophosphatidyl lipase activity with a preference for lysophosphatidylglycerol among other lysophospholipids (PubMed:24095738). Also able to degrade bis(monoacylglycero)phosphate (BMP) and constitutes the major enzyme for BMP catabolism (PubMed:26491015). BMP, also known as lysobisphosphatidic acid, is enriched in late endosomes and lysosomes and plays a key role in the formation of intraluminal vesicles and in lipid sorting (PubMed:26491015). {ECO:0000269|PubMed:18096503, ECO:0000269|PubMed:20657592, ECO:0000269|PubMed:24095738, ECO:0000269|PubMed:26491015}. |
Alternative splicing;Endosome;Hydrolase;Lipid metabolism;Lysosome;Membrane;Mitochondrion;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:66082; |
AMPA glutamate receptor complex [GO:0032281]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; late endosome membrane [GO:0031902]; lysosomal membrane [GO:0005765]; membrane [GO:0016020]; mitochondrial membrane [GO:0031966]; mitochondrion [GO:0005739]; postsynaptic membrane [GO:0045211]; acylglycerol lipase activity [GO:0047372]; phospholipase activity [GO:0004620]; acylglycerol catabolic process [GO:0046464]; long-term synaptic depression [GO:0060292]; lysobisphosphatidic acid metabolic process [GO:2001311]; monoacylglycerol catabolic process [GO:0052651]; negative regulation of cell migration [GO:0030336]; negative regulation of cold-induced thermogenesis [GO:0120163]; phospholipid catabolic process [GO:0009395]; positive regulation of lipid biosynthetic process [GO:0046889]; regulation of endocannabinoid signaling pathway [GO:2000124]; regulation of retrograde trans-synaptic signaling by endocanabinoid [GO:0099178] |
18096503_Data reveal that 85% of brain 2-arachidonoylglycerol hydrolase activity can be ascribed to monoacylglycerol lipase(MAGL), and the remaining 15% is catalyzed by ABHD6 and ABHD12. 20657592_ABHD6 is a rate-limiting step of 2-AG signaling 21665953_Dual inhibition of alpha/beta-hydrolase domain 6 and fatty acid amide hydrolase increases endocannabinoid levels in neurons. 23806692_FAAH but not monoacyl glycerol lipase exerts important protective actions against 2-arachidonoyl glycerol-induced cellular damage. 24095738_ABHD6 can hydrolyze several lipid substrates, positioning ABHD6 at the interface of glycerophospholipid metabolism and lipid signal transduction. 24101490_Data suggest ABHD6 as a therapeutic target that could be relevant in treating inflammation-related conditions. 24814481_Whole-body and b-cell specific ABHD6-KO mice exhibit enhanced glucose-stimulated insulin secretion, and their islets show elevated monoacylglycerol production and insulin secretion in response to glucose. 26997277_alpha/beta-domain hydrolase-6 suppression averts diet-induced obesity and enhances adipose browning and brown adipose function via PPAR alpha/PPAR gamma activation. 27783937_These findings identify ABHD6 as a regulator of the counter-regulatory responses to major metabolic shifts. 33201859_Adipose ABHD6 regulates tolerance to cold and thermogenic programs. 33444462_Carnitine palmitoyltransferase 1C negatively regulates the endocannabinoid hydrolase ABHD6 in mice, depending on nutritional status. |
ENSG00000163686 |
ABHD6 |
84.345028 |
2.0946368562 |
1.066700 |
0.30397378 |
12.284507 |
0.00045673403319733017410864128748926304979249835014343261718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0023112866424125359972319149193253906560130417346954345703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
112.174788727156 |
23.8103310864033 |
53.5533347450677 |
8.75686896234155 |
| ENSMUSG00000025314 |
19271 |
Ptprj |
protein_coding |
Q64455 |
FUNCTION: Tyrosine phosphatase which dephosphorylates or contributes to the dephosphorylation of CTNND1, FLT3, PDGFRB, MET, KDR, LYN, SRC, MAPK1, MAPK3, EGFR, TJP1, OCLN, PIK3R1 and PIK3R2 (By similarity). Plays a role in cell adhesion, migration, proliferation and differentiation (PubMed:18249142). Involved in vascular development (PubMed:12588999). May be involved in the mechanism of contact inhibition of cell growth (PubMed:12771128). Regulator of macrophage adhesion and spreading (PubMed:19268662). Positively affects cell-matrix adhesion (PubMed:12833140). Positive regulator of platelet activation and thrombosis (PubMed:19246339). Negative regulator of cell proliferation (By similarity). Negative regulator of PDGF-stimulated cell migration; through dephosphorylation of PDGFR (PubMed:12833140). Positive regulator of endothelial cell survival, as well as of VEGF-induced SRC and AKT activation; through KDR dephosphorylation (By similarity). Negative regulator of EGFR signaling pathway; through EGFR dephosphorylation (By similarity). Enhances the barrier function of epithelial junctions during reassembly (By similarity). Negatively regulates T-cell receptor (TCR) signaling (By similarity). Upon T-cell TCR activation, it is up-regulated and excluded from the immunological synapses, while upon T-cell-antigen presenting cells (APC) disengagement, it is no longer excluded and can dephosphorylate PLCG1 and LAT to down-regulate prolongation of signaling (PubMed:12913111). {ECO:0000250|UniProtKB:Q12913, ECO:0000269|PubMed:12588999, ECO:0000269|PubMed:12771128, ECO:0000269|PubMed:12833140, ECO:0000269|PubMed:12913111, ECO:0000269|PubMed:18249142, ECO:0000269|PubMed:19246339, ECO:0000269|PubMed:19268662}. |
Cell junction;Cell membrane;Cell projection;Glycoprotein;Hydrolase;Membrane;Phosphoprotein;Protein phosphatase;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:19271; |
cell surface [GO:0009986]; cell-cell junction [GO:0005911]; immunological synapse [GO:0001772]; plasma membrane [GO:0005886]; ruffle membrane [GO:0032587]; beta-catenin binding [GO:0008013]; cadherin binding [GO:0045296]; delta-catenin binding [GO:0070097]; gamma-catenin binding [GO:0045295]; mitogen-activated protein kinase binding [GO:0051019]; phosphatase activity [GO:0016791]; platelet-derived growth factor receptor binding [GO:0005161]; protein kinase binding [GO:0019901]; protein tyrosine phosphatase activity [GO:0004725]; B cell differentiation [GO:0030183]; blood coagulation [GO:0007596]; calcium-mediated signaling using intracellular calcium source [GO:0035584]; glucose homeostasis [GO:0042593]; heart development [GO:0007507]; negative regulation of cell growth [GO:0030308]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of epidermal growth factor receptor signaling pathway [GO:0042059]; negative regulation of insulin receptor signaling pathway [GO:0046627]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of platelet-derived growth factor receptor signaling pathway [GO:0010642]; negative regulation of protein kinase B signaling [GO:0051898]; negative regulation of signaling receptor activity [GO:2000272]; negative regulation of T cell receptor signaling pathway [GO:0050860]; negative regulation of vascular permeability [GO:0043116]; oligodendrocyte differentiation [GO:0048709]; peptidyl-tyrosine dephosphorylation [GO:0035335]; peptidyl-tyrosine dephosphorylation involved in inactivation of protein kinase activity [GO:1990264]; platelet-derived growth factor receptor signaling pathway [GO:0048008]; positive chemotaxis [GO:0050918]; positive regulation of cell adhesion [GO:0045785]; positive regulation of cell-matrix adhesion [GO:0001954]; positive regulation of Fc-gamma receptor signaling pathway involved in phagocytosis [GO:1905451]; positive regulation of focal adhesion assembly [GO:0051894]; positive regulation of macrophage chemotaxis [GO:0010759]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of platelet activation [GO:0010572]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of tumor necrosis factor production [GO:0032760]; protein dephosphorylation [GO:0006470]; regulation of cell adhesion [GO:0030155]; vasculogenesis [GO:0001570] |
12089527_This protein is a candidate for the mouse colon cancer susceptibility locus (Scc1). 12588999_CD148 has a role in developmental vascular organization and regulates endothelial proliferation and endothelium-pericyte interactions 12771128_participates in contact inhibition of VEGF signaling 12833140_negative regulation of growth-factor stimulated cell migration and promotion of cell-matrix adhesion may be related to the function of DEP-1 as tumor suppressor 18249142_Analysis of CD148 and CD45 doubly deficient B cells and macrophages revealed hyperphosphorylation of C-terminal inhibitory tyrosine of SFKs accompanied by substantial alterations in B and myeloid lineage development and defective immunoreceptor signaling 19246339_CD148 is a global regulator of platelet activation and a novel antithrombotic drug target. 19268662_CD148 is a regulator of macrophage activity. 20345711_CD148 plays a critical role in regulating GPVI/FcR gamma-chain expression and maintains a pool of active SFKs in platelets by directly dephosphorylating the C-terminal inhibitory tyrosines of SFKs that is essential for platelet activation 21262971_DEP-1 is negatively regulating FLT3 signaling activity and that its loss may contribute to but is not sufficient for leukemogenic cell transformation. 21543337_differential effects of CD148 in T cells and other leukocyte subsets 22078799_CD45 and CD148 preferentially target different SFK members (Hck and Fgr versus Lyn, respectively) to positively and negatively regulate GPCR pathways. 22815804_These data indicate that PTPRJ may regulate differentiation of normal mammary epithelia and that dysregulation of protein localisation may be associated with tumorigenesis. 23065825_CD148 plays a dominant role in activating Src family kinases in platelets relative to PTP-1B. Both PTPs are required for optimal platelet activation and aggregate formation under high arterial shear rates. 23543053_phosphatase CD148 promotes airway hyperresponsiveness through SRC family kinases 23580664_The large ectodomains of CD45 and CD148 modulate their inhibitory effect by enabling their passive, size-based segregation from ligated TCR, supporting the kinetic-segregation model of TCR triggering. 23889985_DEP-1 acts as an endogenous antagonist of the insulin receptor, and downregulation of DEP-1 results in an improvement of insulin sensitivity. 24016860_Data indicate that CD148 mRNA is upregulated in diseased joints of with collagen-induced arthritis. 26063811_These results demonstrated Ptprj as a physiological enzyme that attenuates insulin signalling in vivo, and indicate that an inhibitor of Ptprj may be an insulin-sensitizing agent. 27364551_our results established DEP-1 as an essential driver of VEGF-dependent permeability, angiogenesis, and metastasis 27859601_This study demonstrated that the protein-tyrosine phosphatase DEP-1 promotes migration and phagocytic activity of microglial cells in part through negative regulation of fyn tyrosine kinase. 28912580_Ptprj-KO mice exhibit improved glucose and insulin tolerance through enhanced insulin signaling. We herein demonstrated that Ptprj-KO mice exhibited lean phenotypes with lower adiposity because of reduced food intake through enhanced leptin signaling. 29301754_This study establishes Csk and CD148 as critical molecular switches controlling the thrombotic and hemostatic capacity of platelets and reveals cell-intrinsic mechanisms that prevent pathological thrombosis from occurring. 29880609_Loss of DEP-1 (Ptprj) promotes myeloproliferative disease in FLT3-ITD acute myeloid leukemia. 30082414_Study shows that protein tyrosine phosphatase receptor type J (PTPRJ) regulates retinal axonal projections by inhibiting eph and abl kinases in mice. 31598898_The protein tyrosine phosphatase PTPRJ/DEP-1 contributes to the regulation of the Notch-signaling pathway and sprouting angiogenesis. 33605542_PTPRJ promotes osteoclast maturation and activity by inhibiting Cbl-mediated ubiquitination of NFATc1 in late osteoclastogenesis. 33784491_CD148 Deficiency in Fibroblasts Promotes the Development of Pulmonary Fibrosis. |
ENSG00000149177 |
PTPRJ |
218.115675 |
0.2317283163 |
-2.109494 |
0.20104225 |
107.578596 |
0.00000000000000000000000033245639168328750056244576734982512679088531048298725858582045486716398385862447639738093130290508270263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000183477382540573763504401526915397765710685093472669198924828142926307661397800075064878910779953002929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
81.3887866373797 |
11.3784225128137 |
351.225037805612 |
33.9275313303871 |
| ENSMUSG00000025348 |
16404 |
Itga7 |
protein_coding |
Q3TZS3 |
Human_homologues FUNCTION: Integrin alpha-7/beta-1 is the primary laminin receptor on skeletal myoblasts and adult myofibers. During myogenic differentiation, it may induce changes in the shape and mobility of myoblasts, and facilitate their localization at laminin-rich sites of secondary fiber formation. It is involved in the maintenance of the myofibers cytoarchitecture as well as for their anchorage, viability and functional integrity. Isoform Alpha-7X2B and isoform Alpha-7X1B promote myoblast migration on laminin 1 and laminin 2/4, but isoform Alpha-7X1B is less active on laminin 1 (In vitro). Acts as Schwann cell receptor for laminin-2. Acts as a receptor of COMP and mediates its effect on vascular smooth muscle cells (VSMCs) maturation (By similarity). Required to promote contractile phenotype acquisition in differentiated airway smooth muscle (ASM) cells. {ECO:0000250, ECO:0000269|PubMed:10694445, ECO:0000269|PubMed:17641293, ECO:0000269|PubMed:9307969}. |
Cell adhesion;Glycoprotein;Integrin;Membrane;Proteomics identification;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the integrin family of cell surface proteins that mediate cellular interactions with the extracellular matrix and other cells. The encoded transmembrane protein is the alpha subunit that forms a noncovalent heterodimer with the beta subunit to form the functional integrin receptor that binds to laminin. Mice lacking the encoded protein exhibit symptoms of progressive muscular dystrophy, impaired axonal regeneration and cerebral vascular defects. [provided by RefSeq, Aug 2016]. |
mmu:16404; |
integrin complex [GO:0008305]; cell adhesion [GO:0007155]; integrin-mediated signaling pathway [GO:0007229] |
12421948_Up-regulation of integrin alpha 7 expression mediated by TGF-beta 1 plays a role in the intraepithelial location of mucosal mast cells in vivo. 12691739_Enhanced axonal regeneration after conditioning lesions involves increased responsiveness to laminin and integrin alpha7beta1 expression. 14525975_the interaction of c-Myc with the promoter region of Itga7 is an important regulatory element controlling alpha7 integrin expression during muscle development and myotendinous junction formation 15117962_FHL2 and FHL3, respectively, are colocalized with alpha(7)beta(1) integrin receptor at the periphery of Z-discs, suggesting a role in mechanical stabilization of muscle cells 15252120_sarcoglycan and integrin alpha7beta1 have overlapping roles in mediating cytoskeletal-membrane-extracellular matrix interaction 15361073_Increased in vitro binding of integrin alpha7beta1 to laminin after ADP-ribosylation of the 37-kDa fragment of alpha7 requires the presence of Mn2+ and it is not observed in the presence of Mg2+ 15691705_These results define an essential interaction between alpha7 integrin and laminin for mediating neurite outgrowth of subpopulations of injured adult sensory neurons. 16003770_Important for the recruitment or survival of cerebral vascular smooth muscle cells, plays an important role in vascular development and integrity. 16129691_deltaEF1 has a role in suppressing integrin alpha 7 expression in myoblasts by displacing MYOD and competing for p300/CBP co-activator 16476707_An essential role is indicated for integrin alpha 7 in the maintenance of dystrophin-deficient muscles. 17704212_Alpha7beta1 integrin negatively regulates extracellular signal-regulated kinase activation. 17904217_Together our results confirm a role for the alpha7beta1 integrin in placental vascular development and demonstrate for the first time that loss of the alpha7 integrin results in placental defects. 18045857_Thus increasing integrin levels can compensate for defects in the extracellular matrix and cytoskeleton linkage caused by compromises in the dystrophin-glycoprotein complex without triggering apparent overt negative side effects. 18055553_Ilk may act as a cytoplasmic effector of alpha 7 beta1-integrin in the pathogenesis of progressive muscular dystrophy. 18590826_Results identify non-overlapping functions of integrins alpha3beta1, alpha7beta1 and alpha6beta1 on different laminin isoforms, and suggest a role for these interactions in controlling neurite outgrowth from adult DRG neurons. 18784336_exercise-induced muscle injury selectively increases transcription of the alpha7 integrin gene and promotes a rapid change in the alpha7beta integrin at the myotendinous junction 18940796_cleavage is a novel mechanism that regulates alpha7 integrin functions in skeletal muscle, and that the generation of such cleavage sites is another evolutionary mechanism for expanding and modifying protein functions. 19416897_laminin-111 (alpha(1), beta(1), gamma(1)), which is expressed during embryonic development but absent in normal or dystrophic skeletal muscle, increased alpha(7)-integrin expression in mouse and DMD patient myoblasts 19729483_These results indicate that the alpha7beta1 integrin, dystrophin, and utrophin complexes act in a concerted manner to maintain the structural and functional integrity of skeletal muscle. 21216283_Integrin alpha 7 integrin beta1 confers a protective effect in dystrophic muscle through the activation of the ILK, AKT, p70S6K and BAD signaling to promote muscle cell survival. 21652631_alpha7 integrin prevents muscle disease progression through augmentation and/or stabilization of the existing extracellular matrix in laminin-alpha2-deficient mice and might be a novel drug target for the treatment of MDC1A. 21753185_This study provides the first demonstration that the presence of the alpha(7)beta(1)-integrin in skeletal muscle increases fiber hypertrophy and new fiber synthesis in the early time course following a single bout of eccentric exercise. 22798625_loss of alpha7 integrin exacerbates a newly discovered muscle phenotype in sarcospan-null mice 25504048_Sarcospan-mediated amelioration of muscular dystrophy in mouse model is dependent on the presence of both utrophin and alpha7beta1 integrin. 26355035_Deletion of integrin alpha7 subunit does not aggravate the phenotype of laminin alpha2 chain-deficient mice. laminin-211 and integrin alpha7beta1 have intersecting roles in skeletal muscle. 28175314_Ptrh2 levels were up-regulated in dystrophin deficient mdx muscle, which correlates with the elevated levels of the a7b1 integrin observed in mdx muscle and Duchenne muscular dystrophy patients. Similar to the a7 integrin, Ptrh2 expression was decreased in laminin-a2 null gastrocnemius muscle 33655887_miRNAs regulating the expressions of NTF3, GNG2 and ITGA7 are involved in the pathogenesis of abdominal aortic aneurysm in mice. 35165120_Muscle stem cell polarity requires QKI-mediated alternative splicing of Integrin Alpha-7 (Itga7). 35568813_ATF1/miR-214-5p/ITGA7 axis promotes osteoclastogenesis to alter OVX-induced bone absorption. 35628462_Association between Decreased ITGA7 Levels and Increased Muscle alpha-Synuclein in an MPTP-Induced Mouse Model of Parkinson's Disease. 36444867_Integrin alpha7 Mutations Are Associated With Adult-Onset Cardiac Dysfunction in Humans and Mice. |
ENSG00000135424 |
ITGA7 |
30.529784 |
0.3866582760 |
-1.370869 |
0.45489020 |
9.076929 |
0.00258854373391053478245527053047680965391919016838073730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0106146376890383364877123995029251091182231903076171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.8996770820113 |
5.09674310283824 |
41.1207468426113 |
9.22508098875279 |
| ENSMUSG00000025407 |
14632 |
Gli1 |
protein_coding |
P47806 |
FUNCTION: Acts as a transcriptional activator. Binds to the DNA consensus sequence 5'-GACCACCCA-3'. Regulates the transcription of specific genes during normal development. Plays a role in craniofacial development and digital development, as well as development of the central nervous system and gastrointestinal tract. Mediates SHH signaling. Plays a role in cell proliferation and differentiation via its role in SHH signaling. {ECO:0000250|UniProtKB:P08151}. |
Acetylation;Activator;Cytoplasm;Developmental protein;Differentiation;DNA-binding;Isopeptide bond;Metal-binding;Nucleus;Phosphoprotein;Proto-oncogene;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:14632; |
axoneme [GO:0005930]; cilium [GO:0005929]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; GLI-SUFU complex [GO:1990788]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; microtubule binding [GO:0008017]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; transcription cis-regulatory region binding [GO:0000976]; cerebellar cortex morphogenesis [GO:0021696]; dorsal/ventral pattern formation [GO:0009953]; epidermal cell differentiation [GO:0009913]; liver regeneration [GO:0097421]; lung development [GO:0030324]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of hh target transcription factor activity [GO:1990787]; notochord regression [GO:0060032]; osteoblast differentiation [GO:0001649]; pituitary gland development [GO:0021983]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cell cycle G1/S phase transition [GO:1902808]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA replication [GO:0045740]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of smoothened signaling pathway [GO:0045880]; positive regulation of transcription by RNA polymerase II [GO:0045944]; prostate gland development [GO:0030850]; proximal/distal pattern formation [GO:0009954]; regulation of hepatocyte proliferation [GO:2000345]; regulation of osteoblast differentiation [GO:0045667]; regulation of transcription by RNA polymerase II [GO:0006357]; response to wounding [GO:0009611]; signal transduction [GO:0007165]; smoothened signaling pathway [GO:0007224]; smoothened signaling pathway involved in regulation of cerebellar granule cell precursor cell proliferation [GO:0021938]; spermatid development [GO:0007286]; spermatogenesis [GO:0007283]; ventral midline development [GO:0007418] |
12138125_regulation of transcriptional activity in nucleus by Dyrk1 12361967_in mammals, Gli1 is not required for Shh signaling and that Gli2 mediates inappropriate activation of the pathway due to loss of the negative regulator Ptc. 14555646_GLI-1 has a role in mediating bcl-2 transcriptional regulation by the sonic hedgehog signaling pathway 15614767_Fgf15 is directly regulated by Shh signaling through Gli proteins in the developing diencephalon and midbrain 15793308_Shh, functioning via Gli1/2, can specify mesodermal cells into the cardiac muscle lineage 15806168_Gli1 plays a central role in medulloblastoma formation in Ptc1+/- mice and that Gli2 may also contribute to oncogenesis. 15811134_Gli1 is not required to induce Pdgfra expression during embryonic bone development. 16396903_Results demonstrate that SHH-SMO signaling controls renal morphogenesis via transcriptional control of Gli1 and 2, renal patterning and cell cycle regulator genes in a manner that is opposed by GLI3. 16421275_Here we show that Gli is rapidly destroyed by the proteasome and that mouse basal cell carcinoma induction correlates with Gli protein accumulation,We identify two independent destruction signals in Gli1, D(N) and D(C) 16855389_investigation of GLI protein processing and binding of GLI activators and repressor to sonic hedgehog short digits(SHH) target genes provide insight into the molecular mechanisms by which SHH and its GLI family of effectors control renal embryogenesis 17115028_Hedgehog transcription factor Gli1 is targeted by Numb for Itch-dependent ubiquitination, which suppresses Hedgehog signals, thus arresting growth and promoting cell differentiation. 17392427_Melanomas require Shh-GLI signaling regulated by interactions between GLI1 and the RAS-MEK/AKT pathways. 17442700_Gli-activator targets have roles in sonic hedgehog-mediated neural patterning 17638910_transforming growth factor-beta has a role in Smad3-dependent activation of Gli2 and Gli1 expression 17901397_In this study, a remarkable feature of Gli1 is its regulation of sonic hedgehog-induced Notch/Hes-1 and Wnt1/beta-catenin activation in embryonic stem cells. 17928300_GLI1-expressing transgenic females were unable to lactate and milk protein gene expression was essentially absent. The inability to lactate was permanent and independent of continued GLI1 transgene expression. 18084326_GLI1 appears to be a transcriptionally deregulated target of EWS/FLI1 that mediates a portion of its tumorigenic phenotype. 18218620_Rab23 is involved in the control of Sox9 expression via Gli1 protein 18276799_In contrast to normal stem cells, tumor-derived stem cells have markedly higher basal Gli1 expression and do not undergo apoptosis with further elevation of Gli1. 18625210_Data show that the expression of Hedgehog (Hh) target genes, Gli1, in the optic nerves is downregulated in a conditional ablation of Shh mutant mice. 19136624_the expression of Gli target genes is maintained in Smo-negative ducts, implicating alternative means of regulating Gli transcription in the neoplastic ductal epithelium 19214186_The balance of GLI1 and p53 functions, thus, determines cell numbers, and prevalence of p53 restricts GLI1-driven stem cell expansion and tumourigenesis. 19458072_A role of Hh/GLI signaling in breast cancer development. 19549984_The kinesin protein Kif7 is a critical regulator of Gli transcription factors in mammalian hedgehog signaling. 20081843_Results show that Gli1 and Gli2 are acetylated proteins and that their HDAC-mediated deacetylation promotes transcriptional activation and sustains a positive autoregulatory loop through Hedgehog-induced upregulation of HDAC1. 20107231_Gli1 regulates normal and stress hematopoiesis 20935514_Data show that Gli1 is non-redundant in the regulation of T-cell development in the thymus, at multiple developmental stages. 21076395_SNF5 is a key mediator of Hedgehog signaling and that aberrant activation of GLI1 is a previously undescribed targetable mechanism contributing to the growth of malignant rhabdoid tumor cells. 21143927_Pituitary adenylyl cyclase activating polypeptide inhibits gli1 gene expression and proliferation in primary medulloblastoma derived tumorsphere cultures. 21209421_In fetal testes prior to Leydig cell differentiation, expression of Gli1 and Gli2 is abolished by Hedgehog inhibitor GANT61. At later stages, GANT61 inhibits Gli1 expression but has no effects on Gli2 expression and fetal Leydig cell appearance. 21674568_PACAP prevented the direct activation of the Sonic hedgehog target gene Gli-1. 21713771_Data demonstrate that the timing of gene expression along with the genetic lineage (Shh or Gli1) within the neural progenitors segregate mDA neurons into distinct spatial domains. 22493482_upon Hedgehog input, Gli1 functions collectively with Gli2 and Gli3 in osteogenesis. 23396189_Gli3 is required in Emx1+ progenitors for the development of the corpus callosum 23423383_Hh-Gli activators alter the function of BMP to specify perichondrial cells into osteoblasts 23446420_aPKC-iota/lambda functions downstream of smoothened to phosphorylate and activate GLI1, resulting in maximal DNA binding and transcriptional activation 23482563_these results define a novel role for GLI1 in carcinogenesis acting as a downstream effector of oncogenic KRAS in the tumor microenvironment. 23740243_SULF2 regulates tissue regeneration in part via the activation of a novel WNT-GLI1-CYCLIN D1 pathway. 23928057_Findings uncover a novel epigenetic link whereby menin directly represses Gli1 expression, independent of the canonical Hedgehog signaling pathway. 24013724_These data unveil a p53/PCAF/Gli1 circuitry centered on PCAF that limits Gli1-enhanced mitogenic and prosurvival response. 24072710_Dzip1-dependent stabilization of Spop/HIB is evolutionarily conserved and essential for proper regulation of Gli/Ci proteins in the Hh pathway. 24276242_Data indicate a specific genetic requirement for Gli proteins in Hedgehog (Hh)-induced embryonal rhabdhomyosarcoma (eRMS) formation. 24700818_A Gli-dependent cis-regulatory module (CRM), regulation of Grem1 in the mouse limb bud, is reported. 24895267_Study shows that the SHH-Gli axis drives proliferation in response to kainic acid-induced neurodegeneration 25024229_a new mechanism of Gli transcription factor activation and implicate ARHGAP36 dysregulation in the onset and/or progression of GLI-dependent cancers. 25027328_Ptc1-Gli1 signaling deregulation resulting in abnormal loss of glial precursor cells may contribute to a cognition decline in Alzheimer's disease brains. 25046848_Cortical activation of Gli1 protects mice from induction of hepatic encephalopathy. TGFbeta1 suppresses Gli1 in neurons via SMAD3 and promotes the neurologic decline. 25104358_GLI1 modulates the inflammatory response during pancreatic tissue remodeling. 25458012_validate our model by successfully predicting expression changes of Gli1 and Ptch1 in mutants at different developmental stages and in different tissues 25465115_These findings implicate perivascular Gli1(+) mesenchymal stem cells-like cells as a major cellular origin of organ fibrosis. 25639866_show that combined targeting of GLI and PI3K/AKT/mTOR signaling can have a synergistic therapeutic effect in cells from a subgroup of chronic lymphocytic leukemia patients 25645196_Beta-catenin stabilization increases its physical interaction with Gli1. 25799059_Gli1+ cells within the suture mesenchyme are an indispensable mesenchymal stem cell population supporting craniofacial bone turnover and injury healing. 25823816_Gli1, a known Ptch activator, preferentially bound the mutant Ptch locus in rhabdomyosarcoma. 26010061_the three GLI factors(GLI1, GLI2, and GLI3) in mature hepatocytes form an interactive transcriptional network that is involved in the control of target genes associated with metabolic zonation as well as with lipid and drug metabolism 26416758_characterization of the contribution to remyelination of a subset of adult neural stem cells, identified by their expression of Gli1, a transcriptional effector of the sonic hedgehog pathway 26797124_NANOG binds to GLI1 and GLI3 proteins and represses Hedgehog-mediated transcription. 27443289_High Gli1 expression is associated with leukemia. 27454381_The Function of SDF-1-CXCR4 Axis in SP Cells-Mediated Protective Role for Renal Ischemia/Reperfusion Injury by SHH/GLI1-ABCG2 Pathway 27621083_USP21 recruits and stabilises Gli1 at the centrosome. 27760825_Gli1 and Gli2 exhibited different functions in the regulation of p63 expression or proliferation of p63(+) cells in Kras-AR driven tumors. 27849569_Sufu is upregulated in active Shh responding tissues and accompanies Gli activators translocating into and Gli repressors out of the nucleus. 28235772_Shh production and Gli signaling is activated in vivo in lung, enhancing the Th2 response during a murine model of allergic asthma 28276480_these data demonstrate the preclinical efficacy of targeting the downstream HDAC1/2-Gli1 acetylation in the treatment of SHH Medulloblastoma. 28418873_FLT3-mutated in contrast to FLT3 wildtype cells or normal human hematopoietic progenitor cells are exquisitely sensitive to combined inhibition by FLT3, PI3K and GLI1/2 overcoming some of the limitations of current FLT3 directed therapy in AML. 28635133_Results indicate that GLI1 is important for maintaining the invasive and mesenchymal-like properties of melanoma cells independent of MITF. 28690072_Gli1-expressing bone marrow cells are responsible for primary myelofibrosis in a transgenic mouse model. 29230039_Zinc finger protein GLI1 (Gli1) marks mesenchymal progenitors responsible for both normal bone formation and fracture repair. 29476172_Foxm1, which proved to be directly regulated by Gli and Nanog, controls a pro-stemness microRNA network in neural stem cells. 29875413_data suggest a mechanism by which the stem cell niche is adjusted to meet the needs of the intestine via adaptive changes in the number of mesenchymal GLI1-expressing cells 30303712_A conditionally immortalized Gli1-positive kidney mesenchymal cell line models myofibroblast transition and kidney fibrosis. 30317648_depletion of endogenous cholesterol also reduced Shh-induced ALP activity and Gli1 expression. Likewise, cholesterol depletion inhibited osteogenic response to Purmo, although it did not affect Gli1 induction. Taken together, our findings have demonstrated that cholesterol plays a dual role in osteoblast differentiation likely through both Hh-dependent and -independent mechanisms 30918291_FOXS1 is regulated by GLI1 and miR-125a-5p and promotes cell proliferation and EMT in gastric cancer. 30982371_data suggest that Gli1 is not required for bleomycin-induced lung fibrosis. 31988246_The extracellular sulfatase SULF2 promotes liver tumorigenesis by stimulating assembly of a promoter-looping GLI1-STAT3 transcriptional complex. 32039485_Primary cilia mediate parathyroid hormone receptor type 1 osteogenic actions in osteocytes and osteoblasts via Gli activation. 32276104_Involvement of Shh/Gli1 signaling in the permeability of blood-spinal cord barrier and locomotion recovery after spinal cord contusion. 32329192_The Gli1-Snail axis contributes to Salmonella Typhimurium-induced disruption of intercellular junctions of intestinal epithelial cells. 32472648_Gli1(+) cells in PDL directly respond to orthodontic force. 32527063_Sonic Hedgehog-Gli1 Signaling and Cellular Retinoic Acid Binding Protein 1 Gene Regulation in Motor Neuron Differentiation and Diseases. 32610119_H1N1 Influenza Virus Cross-Activates Gli1 to Disrupt the Intercellular Junctions of Alveolar Epithelial Cells. 32652075_Gli1+ Periodontium Stem Cells Are Regulated by Osteocytes and Occlusal Force. 32772391_Acute Alcohol Intoxication Impairs Sonic Hedgehog-Gli1 Signaling and Activation of Primitive Hematopoietic Precursor Cells in the Early Stage of Host Response to Bacteremia. 32917668_GFI1 functions to repress neuronal gene expression in the developing inner ear hair cells. 32997394_TGF-beta/Smad2 signalling regulates enchondral bone formation of Gli1(+) periosteal cells during fracture healing. 33046884_Gli1(+) mesenchymal stromal cells form a pathological niche to promote airway progenitor metaplasia in the fibrotic lung. 33085791_Regulation of hepatic stellate cell contraction and cirrhotic portal hypertension by Wnt/beta-catenin signalling via interaction with Gli1. 33125874_Relative Levels of Gli1 and Gli2 Determine the Response of Ventral Neural Stem Cells to Demyelination. 33529374_Gli1 Defines a Subset of Fibro-adipogenic Progenitors that Promote Skeletal Muscle Regeneration With Less Fat Accumulation. 34772739_Gli1 Regulates the Postnatal Acquisition of Peripheral Nerve Architecture. 35027088_A subpopulation of astrocyte progenitors defined by Sonic hedgehog signaling. 35145123_GLI transcriptional repression is inert prior to Hedgehog pathway activation. 35594713_Inhibition of Gli1 suppressed hyperglycemia-induced meibomian gland dysfunction by promoting ppargamma expression. 35931079_The epigenetic state of EED-Gli3-Gli1 regulatory axis controls embryonic cortical neurogenesis. |
ENSG00000111087 |
GLI1 |
496.027663 |
0.1633241425 |
-2.614190 |
0.17672022 |
204.148111 |
0.00000000000000000000000000000000000000000000025981635444522034775593644145468351697944664395594128087251422169999474027740039544516844828713897100126933867119370658027577292159548960626125335693359375000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000384225505913553024545356471024072310598702455227691268813463342794929392632263206373370904549742810412288136128009657932480536146613303571939468383789062500000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
142.918873060777 |
17.5503914154942 |
875.062748824759 |
75.0925919856074 |
| ENSMUSG00000025420 |
71206 |
Katnal2 |
protein_coding |
Q9D3R6 |
FUNCTION: Severs microtubules in vitro in an ATP-dependent manner. This activity may promote rapid reorganization of cellular microtubule arrays. {ECO:0000255|HAMAP-Rule:MF_03025}. |
Alternative splicing;ATP-binding;Cytoplasm;Cytoskeleton;Isomerase;Microtubule;Nucleotide-binding;Reference proteome |
|
|
Human_homologues hsa:83473; |
cytoplasm [GO:0005737]; microtubule [GO:0005874]; spindle [GO:0005819]; spindle pole [GO:0000922]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; isomerase activity [GO:0016853]; microtubule binding [GO:0008017]; microtubule severing ATPase activity [GO:0008568]; microtubule severing [GO:0051013] |
26153462_KATNAL2 isoforms interact with themselves, with each other and with Nubp1 and Nubp2. 29136647_Authors present data suggesting that depending on context, KATNAL2 can partner with the regulatory protein KATNB1 or act autonomously. Data indicate KATNAL2 may regulate delta- and epsilon-tubulin rather than classical alpha-beta-tubulin microtubule polymers, suggesting the katanin family has a greater diversity of function than previously realised. 34096614_Biallelic mutations in KATNAL2 cause male infertility due to oligo-astheno-teratozoospermia. |
ENSG00000167216 |
KATNAL2 |
58.531323 |
2.1547406397 |
1.107514 |
0.39460775 |
7.836682 |
0.00511964994685196270879368185546809399966150522232055664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0189692308417536113773760320100336684845387935638427734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
73.3226687607466 |
20.3063140864495 |
34.0285356896252 |
7.28225887294907 |
| ENSMUSG00000025429 |
19201 |
Pstpip2 |
protein_coding |
Q99M15 |
FUNCTION: Binds to F-actin. May be involved in regulation of the actin cytoskeleton. |
Coiled coil;Cytoplasm;Direct protein sequencing;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:19201; |
actin filament [GO:0005884]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; membrane [GO:0016020]; plasma membrane [GO:0005886]; actin binding [GO:0003779]; actin filament binding [GO:0051015]; actin filament polymerization [GO:0030041]; cytoskeleton organization [GO:0007010] |
15788569_MAYP directly regulates F-actin bundling and enhances filopodia formation and motility in macrophages. 16122996_A missense mutation in pstpip2 is associated with the autoinflammatory disorder chronic multifocal osteomyelitis. 18721907_mutations are associated with spontaneous disease in mouse model of osteomyelitis 19608749_PSTPIP2 deficiency leads to autoinflammatory disease. 22646813_our pstpip2 mutant mice provide a new model for investigating the potential mechanisms of inflammatory pain. 22923495_PSTPIP2 deficiency in mice causes osteopenia and increased differentiation of multipotent myeloid precursors into osteoclasts. 23525018_Knockdown of PSTPIP2 inmacrophages enhances the assembly of FBP17, leading to'hyperactivation' of actin nucleation at podosomes and ECM degradation. 24395792_inflammasome-independent production of IL-1beta has a critical role in osteolytic bone disease; PSTPIP2 is a negative regulator of caspase-1-autonomous IL-1beta production 24395802_IL-1beta has a role in mediating autoinflammatory disease in Pstpip2-deficient mice 25602062_PSTPIP2 could play a role in innate immunity and development of autoinflammatory bone disorders, and may be associated with the pathogenesis of SAPHO syndrome. 26304991_SHIP1 and Csk are part of the PSTPIP2-dependent inhibitory network that prevents the development of autoinflammatory disease. 27760174_a new PSTPIP1/PTPN6/SHIP1/2-dependent negative feedback mechanism that counterbalances Src and PI(3,4,5)P3 signalling to control osteoclast cell polarity and activity during bone resorption 29993036_Knockdown of PSTPIP2 promoted M1 polarization and suppressed M2 polarization in vitro. 32024700_Dysregulated NADPH Oxidase Promotes Bone Damage in Murine Model of Autoinflammatory Osteomyelitis. 36324152_PSTPIP2 regulates synovial macrophages polarization and dynamics via ERbeta in the joint microenvironment. 36605205_Molecular interactions of adaptor protein PSTPIP2 control neutrophil-mediated responses leading to autoinflammation. |
ENSG00000152229 |
PSTPIP2 |
63.266591 |
2.3853276309 |
1.254187 |
0.31654649 |
15.613977 |
0.00007767828336738844333120290741589997196570038795471191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004725608195450908778287302247633760998724028468132019042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
85.7113109408537 |
18.9574611271415 |
35.9327204498123 |
6.2404720348841 |
| ENSMUSG00000025432 |
11567 |
Avil |
protein_coding |
O88398 |
FUNCTION: Ca(2+)-regulated actin-binding protein which plays an important role in actin bundling. May have a unique function in the morphogenesis of neuronal cells which form ganglia. Required for SREC1-mediated regulation of neurite-like outgrowth. Plays a role in regenerative sensory axon outgrowth and remodeling processes after peripheral injury in neonates. Involved in the formation of long fine actin-containing filopodia-like structures in fibroblast. Plays a role in ciliogenesis (PubMed:15247299, PubMed:18160648). In podocytes, controls lamellipodia formation through the regulation of EGF-induced diacylglycerol generation by PLCE1 and ARP2/3 complex assembly (By similarity). {ECO:0000250|UniProtKB:O75366, ECO:0000269|PubMed:15247299, ECO:0000269|PubMed:18160648}. |
Actin capping;Actin-binding;Calcium;Cell junction;Cell projection;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:11567; |
actin cytoskeleton [GO:0015629]; actin filament [GO:0005884]; axon [GO:0030424]; cell projection [GO:0042995]; cytoplasm [GO:0005737]; focal adhesion [GO:0005925]; lamellipodium [GO:0030027]; neuron projection [GO:0043005]; actin binding [GO:0003779]; actin filament binding [GO:0051015]; Arp2/3 complex binding [GO:0071933]; phosphatidylinositol-4,5-bisphosphate binding [GO:0005546]; actin filament organization [GO:0007015]; actin filament severing [GO:0051014]; actin polymerization or depolymerization [GO:0008154]; barbed-end actin filament capping [GO:0051016]; cilium assembly [GO:0060271]; nervous system development [GO:0007399]; positive regulation of lamellipodium assembly [GO:0010592]; positive regulation of neuron projection development [GO:0010976]; regulation of diacylglycerol biosynthetic process [GO:1900480] |
15247299_interaction of SREC-I and advillin are involved in the development of dorsal root ganglion neurons by inducing the described morphological changes. 18160648_This study found that the regenerative axon growth of Advillin-null sensory neurons is significantly shortened and that deletion of Advillin reduces the plasticity of whisker-related barrelettes patterns in the hindbrain. 30126982_study highlights a role for advillin in growth cone formation, axon regeneration, and neuropathic pain associated with IB4(+) DRG neurons in adulthood. 30221190_Advillin is expressed in all adult neural crest-derived neurons 32483224_Mouse intestinal tuft cells express advillin but not villin. 32617896_Advillin is a tuft cell marker in the mouse alimentary tract. 34847180_Oxytocin receptor disruption in Avil-expressing cells results in blunted sociability and increased inter-male aggression. |
ENSG00000135407 |
AVIL |
183.798105 |
6.7267859958 |
2.749917 |
0.22887784 |
143.456262 |
0.00000000000000000000000000000000467167371493685609524359708480151214779159266184764785386101616824897806311168595622173096981732953736354829743504524230957031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000004161341171106327199303559152411343607065497243973971303250526089453022489818951549361836228513311652932316064834594726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
320.135131236372 |
51.001480046922 |
47.5910979532498 |
6.28906285119884 |
| ENSMUSG00000025488 |
12869 |
Cox8b |
protein_coding |
P48772 |
FUNCTION: Component of the cytochrome c oxidase, the last enzyme in the mitochondrial electron transport chain which drives oxidative phosphorylation. The respiratory chain contains 3 multisubunit complexes succinate dehydrogenase (complex II, CII), ubiquinol-cytochrome c oxidoreductase (cytochrome b-c1 complex, complex III, CIII) and cytochrome c oxidase (complex IV, CIV), that cooperate to transfer electrons derived from NADH and succinate to molecular oxygen, creating an electrochemical gradient over the inner membrane that drives transmembrane transport and the ATP synthase. Cytochrome c oxidase is the component of the respiratory chain that catalyzes the reduction of oxygen to water. Electrons originating from reduced cytochrome c in the intermembrane space (IMS) are transferred via the dinuclear copper A center (CU(A)) of subunit 2 and heme A of subunit 1 to the active site in subunit 1, a binuclear center (BNC) formed by heme A3 and copper B (CU(B)). The BNC reduces molecular oxygen to 2 water molecules using 4 electrons from cytochrome c in the IMS and 4 protons from the mitochondrial matrix. {ECO:0000250|UniProtKB:P10175}. |
3D-structure;Membrane;Mitochondrion;Mitochondrion inner membrane;Reference proteome;Transit peptide;Transmembrane;Transmembrane helix |
PATHWAY: Energy metabolism; oxidative phosphorylation. {ECO:0000250|UniProtKB:P10175}. |
|
mmu:12869; |
mitochondrial respiratory chain complex IV [GO:0005751]; mitochondrion [GO:0005739]; respiratory chain complex IV [GO:0045277]; mitochondrial electron transport, cytochrome c to oxygen [GO:0006123] |
Human_homologues 20877624_Observational study of gene-disease association. (HuGE Navigator) |
ENSG00000187581 |
COX8C |
28.371531 |
0.3556203093 |
-1.491590 |
0.48505522 |
9.425596 |
0.00213977550238761849571433870664804999250918626785278320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0090210518827479116432677130887896055355668067932128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.2590136387272 |
5.22750703531905 |
42.9081614305203 |
10.224627907688 |
| ENSMUSG00000025489 |
73835 |
Ifitm5 |
protein_coding |
O88728 |
FUNCTION: Required for normal bone mineralization. {ECO:0000269|PubMed:18442316, ECO:0000269|PubMed:20838829, ECO:0000269|PubMed:24058703, ECO:0000269|PubMed:24715519}. |
Cell membrane;Developmental protein;Lipoprotein;Membrane;Mineral balance;Palmitate;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:73835; |
cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; plasma membrane [GO:0005886]; bone mineralization [GO:0030282]; bone morphogenesis [GO:0060349]; in utero embryonic development [GO:0001701]; regulation of bone mineralization [GO:0030500] |
18442316_Bril is a novel osteoblast protein and showed a role in mineralization, possibly identifying a new regulatory pathway in bone formation. 20838829_Ifitm5 deficiency might have a greater effect on prenatal bone development. 21600883_these results suggest that IFITM5 is involved not only in bone formation, but also in immune system activity. 22884724_Natural antisense transcripts enhance bone formation by increasing sense IFITM5 transcription. 23530031_Key roles for the Sp members and GLI2 that possibly cooperate to activate Bril when the promoter becomes demethylated. 24058703_These results indicate that the S-palmitoylation on IFITM5 promotes the interaction with FKBP11. 24715519_The purpose of the current study was to re-assess the topology, localization, and biochemical properties of BRIL and compare it to the osteogenesis imperfecta type V mutant in MC3T3 osteoblasts. 25251575_results suggest that expression of mutant IFITM5 causes abnormal skeletal development, low bone mass, and abnormal osteoblast differentiation 30247497_IFITM5 was expressed in the dental follicle and the developing alveolar bone 33273604_Effect of immunosuppressants on a mouse model of osteogenesis imperfecta type V harboring a heterozygous Ifitm5 c.-14C > T mutation. |
ENSG00000206013 |
IFITM5 |
2330.427951 |
0.4978098479 |
-1.006333 |
0.06692071 |
223.990913 |
0.00000000000000000000000000000000000000000000000001218704248506785083628115137436782864281142229321086178499936247963394362598918589222561656429702257332866826706149782359229647929477202694670268101617693901062011718750000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000021094662628699263399512524942363013882717750586975997468451363224811729508511752955759171251878894765192425296750176612033469969453847170370863750576972961425781250000000000000000000000000000000000000000000000000 |
Yes |
No |
1565.0956782465 |
72.6244119277566 |
3143.96287013023 |
106.616353155358 |
| ENSMUSG00000025551 |
14169 |
Fgf14 |
protein_coding |
A0A0R4J063 |
Human_homologues FUNCTION: Probably involved in nervous system development and function. |
Reference proteome |
|
|
mmu:14169; |
growth factor activity [GO:0008083] |
12123606_Fgf14-deficient mice developed ataxia and a paroxysmal hyperkinetic movement disorder. FGF14 may mediate neuronal signaling, axonal trafficking and synaptosomal function. 17208450_FGF14 in regulating synaptic plasticity via presynaptic mechanisms by affecting the mobilization, trafficking, or docking of synaptic vesicles to presynaptic active zones. 17236779_these results suggest a role for FGF14 in certain spatial learning functions and synaptic plasticity 17678857_Sodium channels in Fhf1-/-Fhf4-/- granule neurons inactivate at more negative membrane potential, inactivate more rapidly, and are slower to recover from the inactivated state. 18930825_results suggest that FGF14 is required for normal Nav1.6 expression in Purkinje neurons, and that the loss of FGF14 impairs spontaneous and repetitive firing in Purkinje neurons by altering the expression of Nav1.6 channels 23640885_inhibition of GSK3 reduces the assembly of the FGF14.Nav channel complex, modifies FGF14-dependent regulation of Na(+) currents, and induces dissociation and subcellular redistribution of the native FGF14.Nav channel complex in hippocampal neurons. 23831029_FGF14 regulates presynaptic Ca2+ channels and synaptic transmission in cultured rat neurons. 23891806_FGF14 is localized in a decreasing proximal to distal gradient along the axon initial segment of Purkinje neurons. 25269146_These data suggest that the FGF14b N-terminus is a potent regulator of resurgent NaV current in cerebellar Purkinje neurons. 25270074_TWEAK/Fn14 pathway instrumental in the pathogenesis of spontaneous lupus nephritis 25926453_The results of this study demonstrated that iFGF14 is required for spontaneous and evoked action potential firing in adult Purkinje neurons, thereby controlling the output of these cells and the regulation of motor coordination and balance. 26687232_These new insights into the biology of FGF14 in neurogenesis shed light into the signaling pathways associated with disrupted functions in complex brain diseases. 27163207_These results indicate that Fgf14(-/-) mice recapitulate salient molecular, cellular, functional and behavioral features associated with human cognitive impairment, and FGF14 loss of function might be associated with the biology of complex brain disorders such as schizophrenia. 31558566_Effects of FGF14 and NaVbeta4 deletion on transient and resurgent Na current in cerebellar Purkinje neurons. 32599005_JAK2 regulates Nav1.6 channel function via FGF14(Y158) phosphorylation. 33651884_Nav1.3 and FGF14 are primary determinants of the TTX-sensitive sodium current in mouse adrenal chromaffin cells. |
ENSG00000102466 |
FGF14 |
57.819416 |
0.1141273460 |
-3.131284 |
0.40357475 |
60.992043 |
0.00000000000000573059657830376080973312155982659490347777206387114290464523946866393089294433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000001459673028105733881571584409523020140701365238156128611990425270050764083862304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.132601044498 |
3.43837128512819 |
106.307571925792 |
18.7597947269748 |
| ENSMUSG00000025558 |
105445 |
Dock9 |
protein_coding |
A0A571BG77 |
Human_homologues FUNCTION: Guanine nucleotide-exchange factor (GEF) that activates CDC42 by exchanging bound GDP for free GTP. Overexpression induces filopodia formation. {ECO:0000269|PubMed:12172552, ECO:0000269|PubMed:19745154}. |
Proteomics identification;Reference proteome |
|
|
|
|
19156867_Thus, these results suggest that Zizimin1 plays an important role in dendrite growth in hippocampal neurons through activation of Cdc42. 32917852_Exosomal lncRNA DOCK9-AS2 derived from cancer stem cell-like cells activated Wnt/beta-catenin pathway to aggravate stemness, proliferation, migration, and invasion in papillary thyroid carcinoma. |
ENSG00000088387 |
DOCK9 |
685.413233 |
0.3716713819 |
-1.427900 |
0.15728890 |
80.168024 |
0.00000000000000000034388752943026224756612357364736396249258833946023329510705002221016002295073121786117553710937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000129017114489698370519790315967346713970385665203673315748478245268415776081383228302001953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
355.804559788147 |
38.8682568712875 |
957.309540278611 |
76.6272258279333 |
| ENSMUSG00000025738 |
214931 |
Fbxl16 |
protein_coding |
A2RT62 |
FUNCTION: Substrate-recognition component of the SCF (SKP1-CUL1-F-box protein)-type E3 ubiquitin ligase complex. {ECO:0000250}. |
Leucine-rich repeat;Methylation;Reference proteome;Repeat;Ubl conjugation pathway |
|
|
mmu:214931; |
cytoplasm [GO:0005737]; SCF ubiquitin ligase complex [GO:0019005]; SCF-dependent proteasomal ubiquitin-dependent protein catabolic process [GO:0031146] |
17603280_SCIRR1 ise involved in the succeeding injury and/or repair processes of the injured spinal cord. 24390425_FBXL16 negatively regulates the activity of B55alpha-PP2A to modulate the genesis of FLK1+ progenitor cells. |
ENSG00000127585 |
FBXL16 |
267.693555 |
0.1637114997 |
-2.610772 |
0.17625904 |
218.300919 |
0.00000000000000000000000000000000000000000000000021232589108589955176725235479850687402916663987641908484022844984817005392571666292292857325988948680915549562005768019132788404501610557417734526097774505615234375000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000344058295002172562680885566828432310017390134784568618229105096979703192238558801803074592133837892050640632222537117299726583397045942547265440225601196289062500000000000000000000000000000000000000000000000000000 |
Yes |
No |
73.629151286858 |
9.01792317967411 |
449.749415355664 |
36.4365163074902 |
| ENSMUSG00000025790 |
108116 |
Slco3a1 |
protein_coding |
Q8R3L5 |
FUNCTION: Mediates the Na(+)-independent transport of organic anions such as prostaglandins (PG) E1 and E2, thyroxine (T4), deltorphin II, BQ-123 and vasopressin. Also displays a low transport activity of estrone 3-sulfate. Shows a pH-sensitive substrate specificity towards T4 and estrone 3-sulfate which may be ascribed to the protonation state of the binding site and leads to a stimulation of substrate transport in an acidic microenvironment. Hydrogencarbonate/HCO3(-) acts as the probable counteranion that exchanges for organic anions. May contribute to regulate the transport of organic compounds in testes across the blood-testis-barrier. {ECO:0000250|UniProtKB:Q9UIG8}. |
Acetylation;Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Ion transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:108116; |
basal plasma membrane [GO:0009925]; plasma membrane [GO:0005886]; organic anion transmembrane transporter activity [GO:0008514]; prostaglandin transmembrane transporter activity [GO:0015132]; sodium-independent organic anion transmembrane transporter activity [GO:0015347]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of protein phosphorylation [GO:0001934]; prostaglandin transport [GO:0015732]; sodium-independent organic anion transport [GO:0043252] |
23643386_Expression of St8sia2 and Slco3a1 was significantly altered in metabolically active tissues in Roux-en-Y gastric bypass-treated compared to weight-matched, sham-operated mice. |
ENSG00000176463 |
SLCO3A1 |
59.223212 |
0.1972735454 |
-2.341731 |
0.42680640 |
28.559142 |
0.00000009087974350742645380669260243497453721772671997314319014549255371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000009637327749285662927084547893508137406115565681830048561096191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.0376627735456 |
6.0147842319983 |
101.572984514155 |
21.152479732552 |
| ENSMUSG00000025810 |
18186 |
Nrp1 |
protein_coding |
P97333 |
FUNCTION: Receptor involved in the development of the cardiovascular system, in angiogenesis, in the formation of certain neuronal circuits and in organogenesis outside the nervous system (By similarity). Mediates the chemorepulsant activity of semaphorins (PubMed:26503042). Recognizes a C-end rule (CendR) motif R/KXXR/K on its ligands which causes cellular internalization and vascular leakage (By similarity). Binds to semaphorin 3A (SEMA3A), the PLGF-2 isoform of PGF, the VEGF165 isoform of VEGFA and VEGFB (By similarity). Coexpression with KDR results in increased VEGF165 binding to KDR as well as increased chemotaxis. Regulates VEGF-induced angiogenesis (By similarity). Binding to VEGFA initiates a signaling pathway needed for motor neuron axon guidance and cell body migration, including for the caudal migration of facial motor neurons from rhombomere 4 to rhombomere 6 during embryonic development (PubMed:26503042). Regulates mitochondrial iron transport via interaction with ABCB8/MITOSUR (By similarity). {ECO:0000250|UniProtKB:O14786, ECO:0000269|PubMed:26503042}. |
3D-structure;Angiogenesis;Calcium;Cell membrane;Cytoplasm;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Heparan sulfate;Heparin-binding;Membrane;Metal-binding;Mitochondrion;Neurogenesis;Phosphoprotein;Proteoglycan;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:18186; |
axon [GO:0030424]; cell surface [GO:0009986]; cytosol [GO:0005829]; early endosome [GO:0005769]; focal adhesion [GO:0005925]; glutamatergic synapse [GO:0098978]; growth cone [GO:0030426]; mitochondrial membrane [GO:0031966]; neurofilament [GO:0005883]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; postsynaptic membrane [GO:0045211]; sorting endosome [GO:0097443]; growth factor binding [GO:0019838]; GTPase activator activity [GO:0005096]; heparin binding [GO:0008201]; metal ion binding [GO:0046872]; protein kinase binding [GO:0019901]; semaphorin receptor activity [GO:0017154]; signaling receptor activity [GO:0038023]; vascular endothelial growth factor binding [GO:0038085]; vascular endothelial growth factor receptor activity [GO:0005021]; actin cytoskeleton reorganization [GO:0031532]; angiogenesis [GO:0001525]; angiogenesis involved in coronary vascular morphogenesis [GO:0060978]; artery morphogenesis [GO:0048844]; axon extension [GO:0048675]; axon extension involved in axon guidance [GO:0048846]; axon guidance [GO:0007411]; axonal fasciculation [GO:0007413]; axonogenesis involved in innervation [GO:0060385]; basal dendrite arborization [GO:0150020]; basal dendrite development [GO:0150018]; blood vessel endothelial cell migration [GO:0043534]; branching involved in blood vessel morphogenesis [GO:0001569]; branchiomotor neuron axon guidance [GO:0021785]; cell migration involved in coronary vasculogenesis [GO:0060980]; cell migration involved in sprouting angiogenesis [GO:0002042]; cellular response to hepatocyte growth factor stimulus [GO:0035729]; cellular response to vascular endothelial growth factor stimulus [GO:0035924]; commissural neuron axon guidance [GO:0071679]; coronary artery morphogenesis [GO:0060982]; dendrite development [GO:0016358]; dichotomous subdivision of terminal units involved in salivary gland branching [GO:0060666]; dorsal root ganglion morphogenesis [GO:1904835]; endothelial cell chemotaxis [GO:0035767]; endothelial cell migration [GO:0043542]; facial nerve structural organization [GO:0021612]; facioacoustic ganglion development [GO:1903375]; gonadotrophin-releasing hormone neuronal migration to the hypothalamus [GO:0021828]; heart development [GO:0007507]; hepatocyte growth factor receptor signaling pathway [GO:0048012]; integrin-mediated signaling pathway [GO:0007229]; motor neuron axon guidance [GO:0008045]; motor neuron migration [GO:0097475]; negative regulation of axon extension [GO:0030517]; negative regulation of axon extension involved in axon guidance [GO:0048843]; negative regulation of extrinsic apoptotic signaling pathway [GO:2001237]; negative regulation of neuron apoptotic process [GO:0043524]; nerve development [GO:0021675]; neural crest cell migration [GO:0001755]; neural crest cell migration involved in autonomic nervous system development [GO:1901166]; neuron development [GO:0048666]; neuron migration [GO:0001764]; neuropilin signaling pathway [GO:0038189]; otic placode development [GO:1905040]; outflow tract septum morphogenesis [GO:0003148]; platelet-derived growth factor receptor signaling pathway [GO:0048008]; positive chemotaxis [GO:0050918]; positive regulation of actin cytoskeleton reorganization [GO:2000251]; positive regulation of angiogenesis [GO:0045766]; positive regulation of axon extension involved in axon guidance [GO:0048842]; positive regulation of cell migration involved in sprouting angiogenesis [GO:0090050]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of filopodium assembly [GO:0051491]; positive regulation of focal adhesion assembly [GO:0051894]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phosphorylation [GO:0042327]; positive regulation of retinal ganglion cell axon guidance [GO:1902336]; positive regulation of stress fiber assembly [GO:0051496]; positive regulation of substrate adhesion-dependent cell spreading [GO:1900026]; postsynapse organization [GO:0099173]; protein localization to early endosome [GO:1902946]; regulation of axon extension involved in axon guidance [GO:0048841]; regulation of Cdc42 protein signal transduction [GO:0032489]; regulation of retinal ganglion cell axon guidance [GO:0090259]; regulation of vascular endothelial growth factor receptor signaling pathway [GO:0030947]; renal artery morphogenesis [GO:0061441]; response to wounding [GO:0009611]; retina vasculature morphogenesis in camera-type eye [GO:0061299]; retinal ganglion cell axon guidance [GO:0031290]; semaphorin-plexin signaling pathway [GO:0071526]; semaphorin-plexin signaling pathway involved in axon guidance [GO:1902287]; semaphorin-plexin signaling pathway involved in neuron projection guidance [GO:1902285]; sensory neuron axon guidance [GO:0097374]; sprouting angiogenesis [GO:0002040]; substrate adhesion-dependent cell spreading [GO:0034446]; substrate-dependent cell migration, cell extension [GO:0006930]; sympathetic ganglion development [GO:0061549]; sympathetic nervous system development [GO:0048485]; sympathetic neuron projection extension [GO:0097490]; sympathetic neuron projection guidance [GO:0097491]; toxin transport [GO:1901998]; trigeminal ganglion development [GO:0061551]; trigeminal nerve morphogenesis [GO:0021636]; trigeminal nerve structural organization [GO:0021637]; vascular endothelial growth factor receptor signaling pathway [GO:0048010]; vasculogenesis [GO:0001570]; VEGF-activated neuropilin signaling pathway [GO:0038190]; VEGF-activated neuropilin signaling pathway involved in axon guidance [GO:1902378]; ventral trunk neural crest cell migration [GO:0036486]; vestibulocochlear nerve structural organization [GO:0021649]; viral entry into host cell [GO:0046718] |
11756651_In the present study, we first show that VEGF selectively up-regulates NRP1 but not NRP2 via the VEGF receptor 2-dependent pathway. 11891274_Targeting of both mouse neuropilin-1 and neuropilin-2 genes severely impairs developmental yolk sac and embryonic angiogenesis 11939589_High expression of NP-1 was observed in vessels, neurons, and astrocytes surrounding cerebral infarcts 12093729_Co-expression of recombinant mouse NP-1 negatively regulates PlexA1 activation by Fes in resting condition. 12401447_Between embryonic day 18 and birth there is a large increase in neuropilin 1 expression in the basilar pontine nuclei, the major source of pontocerebellar axons. 12406894_Both the 'a' and 'c' extracellular domains of neuropilin-1 are essential for rescue of defective vascularities in NP-1 mutants. NP-1 enhances vasculogenesis & angiogenesis exogenously. Dimerization is important for enhancing vascular development. 12577308_promoter regions of human and mouse NRP1 genes were cloned and sequenced; transfection demonstrated that two Sp1 elements are major contributors to constitutive and induced activity 12852851_Npn-1 is a receptor that binds multiple ligands from structurally distinct families, including secreted semaphorins and vascular endothelial growth factors (VEGF). 15376331_Nrpn1 is required for endothelial tip guidance in the developing central nervous system. 15673567_neuropilin 1-mediated positive feedback is a component of peripheral nerve-derived VEGF promotion of arterial differentiation 15920019_role for Npn-1 in regulating endothelial barrier dysfunction in response to VEGF. 16107693_VEGF can induce renal epithelial cell morphogenesis in a Nrp-1-dependent fashion 16328161_Soluble NRP1 appears to primarily regulate vessel permeability while its effect on physiological angiogenesis is less evident in this model. 16360650_Comparison of expression between mouse and chick embryos at early brain developmental stages. 16540575_Some NRP-1-immunopositive clusters colocalize with PSD-95 and are closely juxtaposed to synapsin I-positive clusters. This clustering could be an effective molecular mechanism for localized actin assembly at the site of Sema3A signaling on dendrites. 16638018_Taken together, our findings suggest that Nurr1 might influence the processes of axon guidance and/or angiogenesis via the regulation of Nrp1 expression. 16687132_an isl1/Shh/npn pathway required to affect morphogenesis at the anterior pole of the heart 17374393_results show that NP1 expression is transiently regulated in thymus during DN/TN & DP differentiation & is synchronised with CD25 down-regulation; absence of NP1 during early thymocyte differentiation does not compromise further T-cell differentiation 17428830_NRP1 contributes to both neuronal and vascular patterning by preferentially relaying SEMA3A signals in peripheral axons and VEGF164 signals in blood vessels 17626059_Functional blockage of neuropilin 1 (Npn1) inhibits cleft formation in the developing submandibular gland (SMG) cultured ex vivo. 18054858_Our data reveal a 'gating' function of neuropilin-1 in semaphorin3e-plexinD1 signaling during the assembly of forebrain neuronal circuits 18077678_These results demonstrate a role for CHL1 and Npn1 in establishment of proper targeting of specific thalamocortical projections. 18300426_These observations suggest that in vivo vascular NRP1 preferentially confers VEGF164 signals, whilst axonal NRP1 preferentially transmits SEMA3A signals. 18328743_Neuropilin-1 prolonged interactions with immature DCs (iDCs), resulting in higher sensitivity to limiting amounts of antigen 18356247_neuropilins play multiple roles in the sensory nervous system by directing cranial neural crest cells, positioning sensory neurons and organising their axonal projections. 18381067_NP-1 heterozygous mice had significantly higher mortality due to either acute or chronic heart failure in response to left ventricular pressure overload. 18384522_In liver regeneration, NOS2-synthesized NO may sensitize the remnant liver to growth factors through the induction of Nrp1 and EGFR post-partial hepatectomy. 18436584_Nrp1 is a receptor for TGF-beta1, activates its latent form, and is relevant to regulatory T cell activity and tumor biology. 18463314_Ex vivo colocalization of Cy5.5 dye revealed that in normal and at a higher level in inflamed bladder, accumulation of NRP1 and NRP2 occurs in both urothelial and ganglial cells. 18483621_bone marrow cells are recruited to the sites of neoangiogenesis through the neuropilin-1 (NP-1) receptor and they are essential for the maturation of the activated endothelium and the formation of arteries in mice 18550715_A method is reported which allows genetic defects caused by loss of function of Nrp1 gene important for cardiovascular development to be isolated even in the presence of hemodynamic defects. 18574596_Data report the presence of semaphorin-3A and of its receptor neuropilin-1 along the rostral migratory stream in the brains of young and adult mice. 18628209_binding of Neuropilin-1 to VEGFR-2 requires the PDZ-binding domain of neuropilin-1 18723443_Neuropilin-1 undergoes clathrin-dependent endocytosis in response to VEGF, and lipid raft-dependent endocytosis in response to semaphorin. The endocytosis is required for neuropilin-1 signaling. 18757501_This study identified a discrete, nonclonal cell population expressing neuropilin-1 and Flk1 that differentiates endothelial or neural phenotypes depending on microenvironmental. 19037249_in cultured podocytes, neuropilin-1, the entire signaling receptor complex of neuropilin-2, semaphorin-3A, and plexin-A1, were significantly reduced by AGE-BSA. AGE-BSA inhibited podocyte migration by down-regulating neuropilin-1 19144568_mutant mice display increased sensitivity to experimental autoimmune encephalomielitis 19296474_findings indicate that NRP1 plays an important role in the guidance of callosal axons originating from cortical excitatory neurons but does not support a role in their migration 19325129_NRP1 signaling coordinates pathway choice with neural crest cells fate and therefore confines neuronal differentiation to appropriate locations 19357391_These data highlight a central role for Npn1 in the development of projections from the cingulate cortex and further illustrate the importance of these pioneer axons in the formation of the CC. 19364973_The onset of NRP-1 expression identifies endothelial precursors in murine and human stem cells 19386662_SEMA3A/NRP1 signalling first directs one population of NCCs from the intersomitic path into the sclerotome, and SEMA3F/NRP2 signalling acts subsequently to restrict a second population to the anterior half of the sclerotome. 19403658_Nrp2 and Nrp1 work together to produce segmentation of sensory and motor nerves, and that dorsal peripheral nervous system metamerism is generated in a stepwise, Nrp-dependent process. 19520907_These data support a role for epithelial Nrp1 in the maintenance of normal alveolar structure and suggest that dysregulation of Nrp1 expression may promote epithelial cell death in response to CS exposure, thereby enhancing emphysema development. 19589963_analysis of the pre-target axon sorting for olfactory map formation; found that expression levels of Neuropilin-1 determined both pre-target sorting and projection sites of axons 19590177_advanced glycation end products inhibit NRP1 promoter transcriptional activity in podocytes by reducing the binding ability of the Sp1 transcription factor to attach to the NRP1 promoter 19805273_Data of affinity chromatography showed that the CendR peptides bind to neuropilin-1 (NRP-1) on the target cells. 19837659_alphav beta3 integrin may, in part, negatively regulate VEGF signaling by sequestering NRP1 and preventing it from interacting with VEGFR2 19859539_the guidance factor Semaphorin 3C, which is expressed by corpus callosum neurons, acts through the neuropilin 1 receptor to orient axons crossing through the corpus callosum. 20039409_Our findings suggest that anti-NP-1 peptide could be useful in alleviating chronic arthritis. 20072992_Inhibition of Nrp1 expression led to strong anti-endothelial effects, whereas tumor cells and tumor growth were not affected. 20133938_NRP1 has a role in signaling Sema3A-evoked neuronal death through Fer in cortical neurons 20345923_Sema3A-Npn-1 signaling plays an important role in the guidance and fasciculation of motor and sensory axons but does not contribute to the developmental elimination of these neurons. 20675371_NRP-1 functions as a key determinant of the diverse responses downstream of TGF-beta1 that are mediated by distinct Smad proteins and promotes myofibroblast phenotype. 20687910_identified tumor cell-secreted platelet-derived growth factor-B (PDGF-B) as a crucial factor controlling the differentiation and recruitment processes through an interaction with neuropilin-1 (NRP-1) in mesenchymal stem cells 20919742_Studies identify Plxdc1, Sema4D, and neuropilin-1 as novel LRP1-regulated cell-signaling proteins. 21059704_SEMA3A signals redundantly through both its classical receptors neuropilin (NRP) 1 and, unconventionally, NRP2, while the usual NRP2 ligand SEMA3F is dispensable for this process 21075075_the expression of Sema3A and its receptor components Npn1 and plexin A was examined in the mouse cornea during embryonic and postnatal development, both before and after eye opening. 21245328_results suggest that Nrp1 is essential for proper maintenance of peripheral tolerance and its absence can result in unchecked autoreactive responses, leading to diseases like EAE and potentially MS 21338642_Our results indicate a specific role of NRP1 in hepatocellular carcinoma growth and vascular remodelling. 21364975_Motor and sensory axons are mutually dependent on each other for the generation of their trajectories and interact in part through Npn-1-mediated fasciculation before and within the plexus region of the limbs. 21508241_Robo1 interacts with Nrp1 to modulate semaphorin signaling in the developing forebrain and direct the migration of interneurons through the subpallium and into the cortex. 21653940_Npn1 is an accurate marker of recent thymic emigrant iNKT cells and of continuous thymic output required for pro-inflammatory IL-17 secretion, a key function of adult iNKT cells. 21734098_these findings suggest a role for NRP1 in regulating the podocyte actin cytoskeleton, and therefore reduction of NRP1 expression could be critical for podocyte function. 21828096_VEGF signalling controls GnRH neuron survival via NRP1 independently of KDR and blood vessels. 21852397_cytoplasmic domain of neuropilin 1 is dispensable for angiogenesis, but promotes the spatial separation of retinal arteries and veins 21925156_Data suggest that initial hypoglossal nerve assembly and fasciculation, but not later guidance decisions depend on Npn-1 expression and axon-Schwann cell interactions. 22012397_deletion of Nrp1 in normal epidermis prevents skin tumour initiation 22025255_These observations confirm NRP1 as a valid anti-angiogenic target in malignancy and confirm a role for NRP1 in physiological, VEGF-mediated angiogenesis. 22082309_Modulation of B16(F10) cells migratory ability by semaphorin 3A can be preferentially mediated by Nrp1. Nrp1 silencing did not change the migratory ability of B16(F10) cells towards VEGF. 22237885_The NRP1 is expressed in salivary gland epithelial cells(SGEC)of both human healthy biopsies and in Sjogren's syndrome(SS) samples, and increased NRP1 expression in SS SGEC is significantly associated with pro-angiogenic factors release. 22396765_Endothelial neuropilin disruption in mice causes DiGeorge syndrome-like malformations via mechanisms distinct to those caused by loss of Tbx1 22615927_During development of the mouse cornea, Npn1 is strongly expressed by the trigeminal ganglion whereas Npn2 is expressed at low levels. 22723296_Data demonstrate an essential direct function of Sema3A-Nrp1-PlexinA1 signaling in lymphatic valve formation. 22723300_Sema3A-Nrp-1 signaling directs the maturation of the lymphatic vascular network likely via regulating the perivascular cell coverage of the vessels thus affecting lymphatic vessel function and lymphatic valve development. 22745317_the axonal wiring of Grueneberg neurons is dependent on neuropilin 1 expression 22790009_SEMA3A signalling through NRP1 has multiple cell-autonomous roles in sympathetic nervous system development. 22885184_Nrp1 plays a key role in mammosphere formation and this activity is linked to NF-kappaB activation. 22927827_Study shows that Nrp1sema/sema mutant mice, which are defective for the semaphorin-binding domain of the membrane coreceptor neuropilin-1, have a Kallmann syndrome -like phenotype 22966001_Neuropilin 1 is expressed on thymus-derived natural regulatory T cells, but not mucosa-generated induced Foxp3+ T reg cells. 22966003_Nrp-1 provides an excellent marker to distinguish distinct T reg subsets and will be useful in studying the role of nT reg versus iT reg cells in different disease settings. 23039190_LPA5 might be involved in both nociception and mechanisms of pain hypersensitivity, as well as in anxiety-related and motivational behaviors. These observations further support the proposed involvement of LPA signaling in psychopathology. 23045606_Data suggest that Neuropilin 1 (Nrp-1) acts as a key mediator of Foxp3(+) T reg cell infiltration into the tumor site resulting in a dampened anti-tumor immune response and enhanced tumor progression. 23063659_These findings indicate a critical role for Sema3-Nrp1 signaling in fetal pulmonary development. 23251405_NRP1 expression in keratinocytes is dispensable for normal skin development. Upon UVB challenge, NRP1 contributes to the prevention of keratinocyte apoptosis. 23315162_NRP1 promotes endothelial tip cell function during angiogenesis. 23639442_Loss of NRP1 expression is associated with impaired developmental arteriogenesis. 23713442_Conditional Nrp1 deletion beginning at postnatal day 5. 23803732_NRP1-driven VEGFR2 trafficking has emerged as a new and important regulatory mechanism controlling VEGF-induced ERK activation and the extent of arterial morphogenesis. 23913274_Treg-cell stability can be subverted in certain inflammatory sites, but is maintained by a Sema4a-Nrp1 axis-- this pathway is a potential therapeutic target that could limit Treg-cell-mediated tumour-induced tolerance without inducing autoimmunity 24033337_tuftsin-mediated anti-inflammatory M2 shift in microglia is caused specifically by tuftsin binding to the receptor neuropilin-1 24276892_Nrp1 is expressed in lymphatic valves and interacts with semaphorin 3A in lymphangiogenesis [review] 24332039_shows that TAMs' heterogeneity depends on their localization, which is tightly controlled by Sema3A/Nrp1 signaling 24401374_Despite the loss of VEGF binding and attenuated NRP1 expression, homozygous Nrp1(Y297A/Y297A) mice were born at normal Mendelian ratios, arguing against NRP1 functioning exclusively as a VEGF164 receptor in embryonic angiogenesis. 24561343_Our results show that Gal-1, mainly in its dimeric form, promotes functional recovery of spinal lesions by interfering with inhibitory signals triggered by Sema3A binding to NRP-1/PlexinA4 complex. 24863063_Data indicate that ABL1 forms a complex with neuropilin 1 (NRP1) in endothelial cells (ECs). 24918638_This study examines the hypothesis that Sema3A signaling through its motor neuron neuropilin-1 (NRP1) receptor triggers distal axonopathy 24977735_We cloned a novel mouse sNrp1 isoform from liver and provide evidence that this endogenous angiogenesis inhibitor may regulate VEGF or HGF bioavailability during normal physiological growth and development as well as during liver regeneration. 25077433_Sox2 is required for cutaneous squamous cell carcinoma growth in mouse and human, where it enhances Nrp1/Vegf signaling to promote the expansion of tumor initiating cells along the tumor-stroma interface. 25233427_Data (including data from mouse mutant strains) suggest Nrp1 in vascular endothelium contributes to angiogenesis via VEGFA-dependent and VEGFA-independent mechanisms; the latter involves Abl1 (c-abl proto-oncogene 1) and extracellular matrix. [REVIEW] 25244320_Nrp1-deficient vessels have reduced VEGFR2 surface expression in vivo demonstrating that NRP1 regulates its co-receptor, VEGFR2. 25245111_These data suggest that graft rejection occurs, at least in part, through the loss of Nrp1 expression on regulatory CD4(+) T cells, their stability or both. 25343644_Nrp1 had no detectable role in the tolerant phenotype. 25416424_The results of the present study suggest that some soluble factors from smooth muscle cells may affect the localization of Nrp-1 in cholinergic neuronal cells, which may, in turn, be involved in the autonomic innervation of blood vessels. 25512301_Data suggest that loss of semaphorin-3A (Sema3A)-neuropilin 1 (Npn1) signaling is not the only cause for the developmental deficits. 25659123_These findings indicate a new role for Nrp1 in the maintenance of the visceral SMC contractile phenotype required for normal GI motility in aged mice. 25843547_expression of Neuropilin1 (Npr1) and Slit2 mRNA was diminished in Islet mutant mice 25855168_These results indicate that NP1 negatively regulates excitatory synapse number by modulating neuronal excitability and show that NP1 restricts excitatory synaptic plasticity. 25924845_Study showed that Nrp1 expression paralleled with that of DDR2 during osteoblast differentiation. Nrp1 assisted the promoting role of DDR2 in osteoblast differentiation, via activation of DDR2-mediated downstream signaling. 26051942_NRP1 enables ECM-induced filopodia formation for tip cell function during sprouting angiogenesis. 26053665_analyzed a complementary set of ligand-specific and tissue-specific mutants to show that neural crest-derived SEMA3C activates NRP1 in the cardiac outflow tract endothelium 26081042_In vascular sprouting, neuropilin-1 suppresses the stalk-cell phenotype by limiting Smad2/3 activation through Alk1 and Alk5. Notch downregulates Nrp1, thus relieving the inhibition of Alk1 and Alk5, thereby driving stalk-cell behaviour. 26159543_suppression of beta3-integrin in mice leads to the activation of a neuropilin-1 (NRP1)-dependent cell migration pathway in endothelial cells via a mechanism that depends on NRP1's mobilisation away from mature focal adhesions following VEGF-stimulation 26495986_The results show that high vs. low expression of Nrp-1 or Helios does not unequivocally identify Treg clones of thymic or peripheral origin. 26534986_demonstrate that in mice on the C57Bl/6 background EMX1 plays an essential role in the midline crossing of an axonal subpopulation of the corpus callosum derived from the anterior cingulate cortex. In the absence of EMX1, cingulate axons display reduced expression of the axon guidance receptor NRP1 and form aberrant axonal bundles within the rostral corpus callosum. 26586223_Nrp1 in endothelial cells suppresses TGFbeta activation and signaling by forming intercellular protein complexes with beta8 integrin. 26708340_Both placental NRP1 and VEGF were expressed at lower levels in women with pre-eclampsia and homocysteine-treated mice, which may contribute to endothelial damage. 26804176_our data suggest a novel molecular mechanism by which tMUC1 may modulate NRP1-dependent VEGFR signaling in PDA cells. 26880314_that microglial expression of Neuropilin-1 (Nrp1) is required for tuftsin-driven amelioration of EAE symptoms 27075020_IL-10 abrogation efficiently induces regulatory T cell (Treg) formation but dampens tumoral neuropilin-1 (Nrp-1) Treg signaling, which simultaneously augments Th1 and Th17 immunity. 27356767_ErbB2 interacts with Nrp1 to form a Sema3d co-receptor expressed by developing coronary endothelium and required for proper connections of the forming coronary veins to the right atrium. 27466379_The aim was to elucidate whether ectopic muscles are a result of misguidance of myoblast precursors due to the loss of Sema3A-Npn-1 signaling. 27591257_These data identify a new molecular mechanism of brain microvascular endothelial inflammatory response through NRP1-IFNgamma crosstalk. 27618676_findings identify a dual function of NRP1 in both axon guidance and subcellular target recognition in the construction of GABAergic circuitry. 27815442_demonstrate that NRP-1 is necessary for VEGF to inhibit bone marrow-derived DC (BMDC) maturation; further demonstrate that NRP-1 associates with TLR4, but not VEGFR2, in a VEGF-dependent manner, and abrogates TLR signaling; results reveal a novel role for NRP-1 in DC physiology and identify NRP-1 as a target to manipulate BMDC maturation 27835904_Tuftsin prevents the negative immunoregulation of neuropilin-1highCD4+CD25+Regulatory T cells and improves survival rate in septic mice. 27844052_Conditional Nrp1 knockout study does not support the Nrp1 model - that olfactory-derived cAMP signals, rather than direct action of olfactory receptor molecules, determine the target destinations (glomeruli) of olfactory sensory neurons in the olfactory bulb - for the anterior-posterior patterning of glomerular positions in the olfactory bulb. 27858201_a novel role for Sema3A-neuropilin 1 signalling in progenitor cell dynamics and in the generation of interneurons in the ventral telencephalon 27988210_Reveal a postdevelopmental role of Nrp1 in renal pericytes as an important regulator of glomerular basement membrane integrity. 28289053_VEGF165-induced vascular leakage requires both VEGFR2 and NRP1, including the VEGF164-binding site of NRP1 and the NRP1 cytoplasmic domain (NCD), but not the known NCD interactor GIPC1. 28325756_These studies reported that myeloid cell-specific Nrp1-deficient mice exhibited enhanced susceptibility to cecal ligation and puncture- and LPS-induced sepsis, which correlated with significantly decreased survival rates and heightened levels of proinflammatory cytokines in both peritoneal lavage and serum. 28432397_the induction of NRP-1 on macrophages may be a therapeutic indicator for antibody treatments that exert antibody-dependent cellular cytotoxicity activity. 28637841_We show that PTP-3, a LAR homolog in Caenorhabditis elegans, participates in Sema2A-regulated axon guidance. PTPdelta, a member of vertebrate LAR class PTPs, is involved in Sema3A-regulated cortical dendritic growth. In Sema3A signaling, PTPdelta activates Fyn and Src kinases by dephosphorylating their C-terminal Tyr residues. 28659345_It was concluded that the HFD-instigated Nrp1 reduction in macrophages exacerbates insulin resistance by promoting Nlrp3 inflammasome priming and activation. 28667171_Data suggest that Nrp1 (neuropilin-1) regulates Hedgehog signaling specifically at the level of activation of GLI2 transcriptional activator function; Nrp1 localization to primary cilium does not correlate with Hedgehog signal promotion. These studies were conducted in various cell types. (GLI2 = GLI-Kruppel family member GLI2 protein) 28676569_Ectopic midline vascularisation in endothelial Nrp1 and Vegfa(188/188) mutants caused additional axonal exclusion zones within the chiasm. 28835419_Nrp1 mediates the migratory behavior of mesangial cells. 28887477_Deletion of Nrp1 results in delay of mammary development. Knockdown of Nrp1 inhibits MMTV-Wnt1 tumor growth in xenograft. 28887481_In summary, the authors concluded that glycosylation-dependent Gal-1/NRP-1 interactions activate TGF-beta and PDGF-like signaling to promote the migration and activation of hepatic stellate cells. 29059172_High NRP1 expression promotes esophageal squamous cancer progression. 29059194_These data suggest that both Nrp1 and Sema3a are expressed in the cochlea during time points when spiral ganglion cell begin to innervate the organ of Corti. 29097606_High Neuropilin 1 expression is associated with Glioma Progression. 29415620_Findings of indicate that neuropilin-1 (Nrp1) vascular progenitor cells (VPCs) in the act as an important step in the arterial differentiation process. 29543041_Cooperation between NRP1 and PDGF signaling is required for secondary septation, and manipulation of NRP1 could promote alveolar regeneration without producing fibrosis. 29549139_Our findings suggest a pivotal role for adipose tissue-resident NRP1(+)-expressing macrophages in driving healthy weight gain and maintaining glucose tolerance. 29779877_Post-crossing CCAs switch off their response to axon guidance cues, among which the secreted Semaphorin-3C (Sema3C), that act as attractants for pre-crossing axons on their way to the midline. This change is concomitant with an upregulation of the surface protein Ephrin-B1, which acts in CCAs to inhibit Sema3C signaling via interaction with the Neuropilin-1 (Nrp1) receptor. 29934640_Study shows that NPR2 is rather evenly distributed across mouse ventricular myocytes membrane and produces far-reaching, diffusible cGMP signals, whereas NPR1 is exclusively found in T-tubules where it creates a microdomain with restricted cGMP diffusion locally confined by PDE2. 30093598_inducible endothelial genetic deletion of Neuropilin1 (Nrp1) and Vascular endothelial growth factor receptor 1 (Vegfr1; also known as Flt1) renders mice resistant to diet-induced obesity. 30355201_Nrp1 signaling mediated accumulation of Tregs within tumor might play a critical role in exacerbating ischemic brain injury in cancer-bearing mice. 30589468_We observed a consistent increase of Neuropilin1 expression in the spinal cord and decrease of Neuropilin1 and Semaphorin3A in muscle tissue of transgenic SOD1(G93A) mice at the mRNA and protein level. 30683854_The pruning of collaterals mediated by Nrp1 is required for motor recovery after spinal cord injury. 31118303_These data highlight a multifaceted role for neuropilin-1 in memory CD8 T cell differentiation, dependent upon the stage of the T cell response and characteristics of the infectious agent. 31350404_Interaction of Nrp-1 with its ligand semaphorin-3A inhibits migration and tumour-specific lytic function of cytotoxic T lymphocytes. 31474563_Sema3a-Nrp1 signaling is a major mechanism for Tw2(+) cell fiber-type specificity; Sema3a, a chemorepellent ligand for Nrp1, is expressed by type I and IIa myofibers but not IIb myofibers 31934626_Epidermal autonomous VEGFA/Flt1/Nrp1 functions mediate psoriasis-like disease. 32500307_miR-128-3p Inhibits NRP1 Expression and Promotes Inflammatory Response to Acute Kidney Injury in Sepsis. 32661362_Neuropilin-1 is a T cell memory checkpoint limiting long-term antitumor immunity. 32761635_Neuropilin-1 expression in GnRH neurons regulates prepubertal weight gain and sexual attraction. 33009002_Reelin-Nrp1 Interaction Regulates Neocortical Dendrite Development in a Context-Specific Manner. 33174445_Neuropilin-1 directs PDGFRalpha-entry into lung fibroblasts and signaling from very early endosomes. 33859199_Microglial neuropilin-1 promotes oligodendrocyte expansion during development and remyelination by trans-activating platelet-derived growth factor receptor. 34039996_Architecture of the Sema3A/PlexinA4/Neuropilin tripartite complex. 34344941_Myeloid-resident neuropilin-1 influences brown adipose tissue in obesity. 34758202_The contribution of neuropilin-1 in the stability of CD4(+) CD25(+) regulatory T cells through the TGF-beta1/Smads signaling pathway in the presence of lipopolysaccharides. 34821196_Semaphorin 3A mitigates lipopolysaccharide-induced chondrocyte inflammation, apoptosis and extracellular matrix degradation by binding to Neuropilin-1. 34848712_Neuropilin 1 regulates bone marrow vascular regeneration and hematopoietic reconstitution. 35230240_Role of Nrp1 in controlling cortical inter-hemispheric circuits. 35380991_Neuropilin-1 deficiency in vascular smooth muscle cells is associated with hereditary hemorrhagic telangiectasia arteriovenous malformations. 35603221_Neuropilin-1 Identifies a New Subpopulation of TGF-beta-Induced Foxp3(+) Regulatory T Cells With Potent Suppressive Function and Enhanced Stability During Inflammation. 35858913_Neuropilin 1 and its inhibitory ligand mini-tryptophanyl-tRNA synthetase inversely regulate VE-cadherin turnover and vascular permeability. 36069030_Neuropilin-1 (NRP1) expression distinguishes self-reactive helper T cells in systemic autoimmune disease. 36173190_Neuropilin-1 Facilitates Pseudorabies Virus Replication and Viral Glycoprotein B Promotes Its Degradation in a Furin-Dependent Manner. 36653359_USP9X-mediated NRP1 deubiquitination promotes liver fibrosis by activating hepatic stellate cells. |
ENSG00000099250 |
NRP1 |
456.664856 |
0.3302892587 |
-1.598198 |
0.60388611 |
6.078566 |
0.01368317732496901299732439127865291084162890911102294921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0438322096769564670259988758971303468570113182067871093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
172.49052886836 |
74.3283827146023 |
522.240806628467 |
165.862062787218 |
| ENSMUSG00000025813 |
26557 |
Homer2 |
protein_coding |
Q9QWW1 |
FUNCTION: Postsynaptic density scaffolding protein. Binds and cross-links cytoplasmic regions of GRM1, GRM5, ITPR1, DNM3, RYR1, RYR2, SHANK1 and SHANK3. By physically linking GRM1 and GRM5 with ER-associated ITPR1 receptors, it aids the coupling of surface receptors to intracellular calcium release. May also couple GRM1 to PI3 kinase through its interaction with AGAP2 (By similarity). Isoforms can be differently regulated and may play an important role in maintaining the plasticity at glutamatergic synapses (By similarity) Required for normal hearing (PubMed:25816005). Negatively regulates T cell activation by inhibiting the calcineurin-NFAT pathway. Acts by competing with calcineurin/PPP3CA for NFAT protein binding, hence preventing NFAT activation by PPP3CA (By similarity). {ECO:0000250|UniProtKB:Q9NSB8, ECO:0000269|PubMed:25816005}. |
3D-structure;Alternative splicing;Cell membrane;Cell projection;Coiled coil;Cytoplasm;Hearing;Membrane;Reference proteome;Synapse |
|
|
mmu:26557; |
apical part of cell [GO:0045177]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; dendrite [GO:0030425]; glutamatergic synapse [GO:0098978]; intracellular organelle [GO:0043229]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; stereocilium tip [GO:0032426]; actin binding [GO:0003779]; G protein-coupled glutamate receptor binding [GO:0035256]; glutamate receptor binding [GO:0035254]; identical protein binding [GO:0042802]; protein domain specific binding [GO:0019904]; protein-containing complex binding [GO:0044877]; synaptic receptor adaptor activity [GO:0030160]; behavioral response to cocaine [GO:0048148]; calcium-mediated signaling using intracellular calcium source [GO:0035584]; G protein-coupled glutamate receptor signaling pathway [GO:0007216]; negative regulation of calcineurin-NFAT signaling cascade [GO:0070885]; negative regulation of interleukin-2 production [GO:0032703]; regulation of G protein-coupled receptor signaling pathway [GO:0008277]; regulation of store-operated calcium entry [GO:2001256]; sensory perception of sound [GO:0007605] |
12860966_Homer2 tunes G protein-coupled receptors stimulus intensity by regulating RGS proteins and PLCbeta GAP activities. 15116392_In the developing olfactory bulb, Cupidin/Homer 2a/b is up-regulated in the granular, external plexiform, and glomerular layers 15545022_Homer2 KO mice demonstrate enhanced locomotor stimulant and conditioned place preference responses to cocaine 16049182_necessary and active role for accumbens Homer2 expression in regulating EtOH-induced behavioral and cellular neuroplasticity 17386922_Expression of HOMER2 enhanced the ability of MYO18B to suppress anchorage-independent growth. These results indicate that HOMER2 and MYO18B cooperate together in tumor suppression. 17568396_Accumbens Homer2 overexpression facilitates alcohol-induced neuroplasticity in C57BL/6J mice. 18218901_study found that Homer2 and Homer3 are negative regulators of T cell activation; this is achieved through binding of nuclear factor of activated T cells (NFAT) and by competing with calcineurin 18387811_Homer2 and Homer3 proteins interact specifically with amyloid precursor protein (APP) by inhibiting APP processing and reducing secretion of Abeta peptides. 20807241_These data support the over-arching hypothesis that augmented accumbens Homer2-mediated glutamate signaling may be an endophenotype related to genetic variance in alcohol consumption 23076792_Homer 1b/c isoform is identical irrespective of the muscle; Homer 2a/b is instead characteristic of the slow-twitch phenotype. 23658151_This study deministrated that PFC Homer2 overexpression enhances cocaine-conditioned reward in mice. 25049230_Findings indicate an important regulation of plasma membrane Ca(2+)-ATPase (PMCA) by Homer2 protein that has a central role on PMCA-mediated Ca(2+) signaling in parotid acinar cells. 25816005_mouse mutants homozygous for the targeted deletion of Homer2 present with early-onset rapidly progressive hearing loss. 25916683_stress-alcohol locomotor cross-sensitization was regulated by Homer2. 26426435_NMDA receptor function, but not ethanol sensitivity, is reduced in hippocampal neurons lacking the Homer2 gene 28728878_detected an additional 15 novel Homer2-interacting proteins, most of which are part of the N-methyl-D-aspartate receptor signaling pathway. These results corroborate the central role Homer2 plays in glutamatergic transmission and expand the network of proteins potentially contributing to the behavioral abnormalities associated with altered Homer2 expression 29109058_The present results provide novel cause-effect evidence supporting a role for CEA Homer2 in the regulation of both basal anxiety and the time-dependent intensification of negative affective states in individuals with a history of binge-drinking during adolescence. 31319381_Data indicate decreased bone density in homer scaffolding proteins (omer2/3) double knockout (DKO) mice. 32070057_Distinct and Overlapping Expression Patterns of the Homer Family of Scaffolding Proteins and Their Encoding Genes in Developing Murine Cephalic Tissues. 34118975_The physiological role of Homer2a and its novel short isoform, Homer2e, in NMDA receptor-mediated apoptosis in cerebellar granule cells. 35008503_Reciprocal Homer1a and Homer2 Isoform Expression Is a Key Mechanism for Muscle Soleus Atrophy in Spaceflown Mice. |
ENSG00000103942 |
HOMER2 |
17.538615 |
0.0377344385 |
-4.727974 |
1.15386796 |
14.463176 |
0.00014292649617456801877483918428168863101745955646038055419921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008224618019384284488271230628697594511322677135467529296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.60966708790979 |
1.42912512289614 |
42.6577724001188 |
21.8137598141928 |
| ENSMUSG00000025877 |
212032 |
Hk3 |
protein_coding |
Q3TRM8 |
FUNCTION: Catalyzes the phosphorylation of hexose, such as D-glucose and D-fructose, to hexose 6-phosphate (D-glucose 6-phosphate and D-fructose 6-phosphate, respectively). Mediates the initial step of glycolysis by catalyzing phosphorylation of D-glucose to D-glucose 6-phosphate. {ECO:0000250|UniProtKB:P27926}. |
Allosteric enzyme;ATP-binding;Glycolysis;Kinase;Nucleotide-binding;Reference proteome;Repeat;Transferase |
PATHWAY: Carbohydrate metabolism; hexose metabolism. {ECO:0000250|UniProtKB:P27926}.; PATHWAY: Carbohydrate degradation; glycolysis; D-glyceraldehyde 3-phosphate and glycerone phosphate from D-glucose: step 1/4. {ECO:0000250|UniProtKB:P27926}. |
|
mmu:212032; |
cytosol [GO:0005829]; mitochondrion [GO:0005739]; protein-containing complex [GO:0032991]; ATP binding [GO:0005524]; enzyme binding [GO:0019899]; fructokinase activity [GO:0008865]; glucokinase activity [GO:0004340]; glucose binding [GO:0005536]; hexokinase activity [GO:0004396]; hormone binding [GO:0042562]; carbohydrate phosphorylation [GO:0046835]; cellular glucose homeostasis [GO:0001678]; fructose 6-phosphate metabolic process [GO:0006002]; glucose 6-phosphate metabolic process [GO:0051156]; glucose metabolic process [GO:0006006]; glycolytic process [GO:0006096]; negative regulation of hydrogen peroxide-mediated programmed cell death [GO:1901299] |
25904018_Hexokinase expression is highly enriched in neurons compared to astrocytes. 35943566_Chronic social stress disrupts the intracellular redistribution of brain hexokinase 3 induced by shifts in peripheral glucose levels. |
ENSG00000160883 |
HK3 |
13.840650 |
0.1451064631 |
-2.784816 |
0.89925578 |
8.588839 |
0.00338229502170270431016252388189968769438564777374267578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0132644484476250237164896006447634135838598012924194335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.32823663487297 |
2.21151400782835 |
22.9365154609675 |
9.36478230062517 |
| ENSMUSG00000025888 |
12362 |
Casp1 |
protein_coding |
P29452 |
FUNCTION: Thiol protease involved in a variety of inflammatory processes by proteolytically cleaving other proteins, such as the precursors of the inflammatory cytokines interleukin-1 beta (IL1B) and interleukin 18 (IL18) as well as the pyroptosis inducer Gasdermin-D (GSDMD), into active mature peptides (PubMed:21147462, PubMed:32109412). Plays a key role in cell immunity as an inflammatory response initiator: once activated through formation of an inflammasome complex, it initiates a pro-inflammatory response through the cleavage of the two inflammatory cytokines IL1B and IL18, releasing the mature cytokines which are involved in a variety of inflammatory processes (PubMed:21147462). Cleaves a tetrapeptide after an Asp residue at position P1 (PubMed:21147462). Also initiates pyroptosis, a programmed lytic cell death pathway, through cleavage of GSDMD (PubMed:32109412). In contrast to cleavage of interleukins IL1B and IL1B, recognition and cleavage of GSDMD is not strictly dependent on the consensus cleavage site but depends on an exosite interface on CASP1 that recognizes and binds the Gasdermin-D, C-terminal (GSDMD-CT) part (PubMed:32109412). Cleaves and activates CASP7 in response to bacterial infection, promoting plasma membrane repair (PubMed:18667412, PubMed:22464733, PubMed:35705808). Upon inflammasome activation, during DNA virus infection but not RNA virus challenge, controls antiviral immunity through the cleavage of CGAS, rendering it inactive (PubMed:28314590). In apoptotic cells, cleaves SPHK2 which is released from cells and remains enzymatically active extracellularly (By similarity). {ECO:0000250|UniProtKB:P29466, ECO:0000269|PubMed:21147462, ECO:0000269|PubMed:28314590, ECO:0000269|PubMed:32109412, ECO:0000269|PubMed:35705808}. |
Apoptosis;Cell membrane;Cytoplasm;Hydrolase;Membrane;Methylation;Phosphoprotein;Protease;Reference proteome;Thiol protease;Ubl conjugation;Zymogen |
Human_homologues NA; + ;NA |
|
mmu:12362; |
AIM2 inflammasome complex [GO:0097169]; caspase complex [GO:0008303]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; inflammasome complex [GO:0061702]; IPAF inflammasome complex [GO:0072557]; microtubule [GO:0005874]; mitochondrion [GO:0005739]; neuron projection [GO:0043005]; NLRP1 inflammasome complex [GO:0072558]; NLRP3 inflammasome complex [GO:0072559]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protease inhibitor complex [GO:0097179]; protein-containing complex [GO:0032991]; CARD domain binding [GO:0050700]; cysteine-type endopeptidase activity [GO:0004197]; cysteine-type endopeptidase activity involved in apoptotic signaling pathway [GO:0097199]; endopeptidase activity [GO:0004175]; identical protein binding [GO:0042802]; kinase binding [GO:0019900]; peptidase activity [GO:0008233]; scaffold protein binding [GO:0097110]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to organic substance [GO:0071310]; cellular response to type II interferon [GO:0071346]; cytokine precursor processing [GO:0140447]; defense response to Gram-negative bacterium [GO:0050829]; defense response to virus [GO:0051607]; membrane hyperpolarization [GO:0060081]; memory [GO:0007613]; microglial cell activation [GO:0001774]; mitochondrial depolarization [GO:0051882]; myoblast fusion [GO:0007520]; osmosensory signaling pathway [GO:0007231]; pattern recognition receptor signaling pathway [GO:0002221]; positive regulation of apoptotic process [GO:0043065]; positive regulation of circadian sleep/wake cycle, non-REM sleep [GO:0046010]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of cytokine production [GO:0001819]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of inflammatory response [GO:0050729]; positive regulation of interleukin-1 alpha production [GO:0032730]; positive regulation of interleukin-1 beta production [GO:0032731]; positive regulation of macrophage cytokine production [GO:0060907]; positive regulation of neuron apoptotic process [GO:0043525]; positive regulation of release of cytochrome c from mitochondria [GO:0090200]; positive regulation of tumor necrosis factor-mediated signaling pathway [GO:1903265]; programmed necrotic cell death [GO:0097300]; protein autoprocessing [GO:0016540]; protein processing [GO:0016485]; proteolysis [GO:0006508]; pyroptosis [GO:0070269]; regulation of autophagy [GO:0010506]; regulation of inflammatory response [GO:0050727]; response to ATP [GO:0033198]; response to bacterium [GO:0009617]; response to hypoxia [GO:0001666]; response to lipopolysaccharide [GO:0032496]; response to organic cyclic compound [GO:0014070]; response to xenobiotic stimulus [GO:0009410]; signaling receptor ligand precursor processing [GO:0140448]; toxin transport [GO:1901998] |
12193723_Expression of caspase-1 is stimulated by high levels of lipopolysaccharide-induced IL-18 and results in sustained synthesis of IL-18 until the death of the animals. 12393844_caspase-1 activity and IL-18 were significantly increased in the kidney in neutrophil-depleted mice with ischemic acute renal failure 12438367_essential for murine host defense against Escherichia coli infection and may represent a mechanism of innate immunity that upon excessive stimulation by microbial components may lead to endotoxic shock 12444148_caspase-1 modulates apoptosis of both peripheral blood and inflammatory neutrophils 12650962_Caspase-1-immunolabeling is observed in scattered photoreceptor cells after 3 h of light exposure but is markedly increased in many more cells at 6 h. 12718436_This enzyme and caspase-3 mRNA are differentially upregulated in motor neurons and glial cells in mutant SOD1 transgenic mouse spinal cord. 12788386_caspase 1 has a role in ventricular dilatation in mice 14662878_Caspase-1 plays an important role in Salmonella-induced dendritic cell death. 14663141_caspase-1 plays a key role in neuronal cell death and that caspase-1 is an apical activator of the cell death pathway in the premitochondrial collapse stage 14673996_may play important regulatory roles in maintaining normal homeostasis in articular cartilage matrix turnover. 14693703_caspase-1 processing of IL-1 beta and IL-18 is not absolutely required for mediation of spontaneous or chemically induced diabetes pathogenesis in the NOD mouse 15075209_Increased K(+) efflux via P2X(7) nucleotide receptor stimulation activates AG-126- and bromoenol lactone-sensitive signaling pathways in murine macrophages that result in stably maintained signals for caspase-1 regulation in cell-free assays. 15190255_cell death triggered by stimuli that engage caspase-1 was ablated in macrophages lacking either ASC or Ipaf, suggesting a coupling between the inflammatory and cell death pathways 15545923_Cytoplasmic activation of cathepsin B was observed from the early stages of infarction, and displayed an activation pattern parallel to the activation pattern of caspase-1 and -11 15569309_caspase-1 contributes to cisplatin-induced acute renal failure and acute tubular necrosis 15644489_caspase-1 -/- mice are functionally protected against endotoxemic acute renal failure 15695506_Data show that Shigella infection of macrophages can induce cell death independent of caspase-1 or toll-like receptor 4 activity. 16407888_results reveal a critical role for cryopyrin in host defence through bacterial RNA-mediated activation of caspase-1, and provide insights regarding the pathogenesis of autoinflammatory syndromes 16407890_cryopyrin is essential for inflammasome (ie. caspase 1 and asc) activation in response to signalling pathways triggered specifically by ATP, nigericin, maitotoxin, S. aureus or L. monocytogenes 16546100_NALP3 plays a specific role in the caspase-1 activation pathway. 16552444_Macrophages sense cytosolic flagellin via a toll-like receptor 5-independent pathway that leads to rapid caspase-1-dependent cell death and provides defense against intracellular bacterial pathogens. 16717117_In knockout mice, absence of caspase-1 or components of the inflammasome does not result in resistance to oral infection by S. typhimurium, but rather, leads to increased susceptibility to infection. 16861683_essential for host innate immune defense against S. enterica serovar Typhimurium, and that Casp-1 substrates, Il-1 beta and Il-18, are required at distinct times and anatomical sites. 16908867_Caspase-1 may be a critical determinant of the host response to sepsis in part due to its ability to regulate sepsis-induced splenic cell apoptosis. 16920334_Cop inhibition of cell death, at least to a certain extent, results from its interference with the activation of caspase-1 and caspase-4. 16974082_No direct influence of the Casp1 genotype on photoreceptor cell apoptosis in the Rs1h(-/Y) mouse could be identified although striking differences in the overall number of resident microglia were observed independent of the Rs1h genotype. 17008311_analysis of a mechanism mediated by cryopyrin and ASC that links dsRNA and viral infection to caspase-1 activation resulting in IL-1beta and IL-18 production 17013693_Retinal neuronal apoptosis was less prominent in Casp1-/- mice after excessive light exposure and ischemia-reperfusion injury. These data indicate that caspase-1 plays a role in retinal neuronal apoptosis. 17188500_caspase-1 and the two caspase-1-processed cytokines, interleukin (IL)-1beta and IL-18, are involved in trauma-induced neuronal cell death in the developing brain 17192486_The caspase-1/interleukin-1beta signaling pathway plays an important role in diabetes-induced retinal pathology, and its inhibition might represent a new strategy to inhibit capillary degeneration in diabetic retinopathy 17255124_These studies demonstrate a deleterious effect of the proinflammatory caspase, caspase-1, on proximal tubules in vitro in the absence of inflammatory cells and vascular effects. 17303764_A functional role for caspase-1-mediated myocardial apoptosis contributes to the progression of heart failure in a wild-type murine heart failure model. 17393315_Pralnacasan prevented dextran sulfate sodium (DSS)-induced colitis in C57BL/6 mice. The DSS-induced increase in IP-10 mRNA, but not TNF-alpha mRNA, was completely prevented by ICE inhibition. 17409187_During systemic inflammatory response syndrome, the expression of several genes, such as chemokines, GTPases, the metalloprotease ADAMTS1, IL-1RA, the inducible nitric oxide synthase, and cyclooxygenase-2, was differentially increased in casp1(-/-) mice. 17491021_changes in the intracellular concentration of K(+) potentiate caspase-1 activation induced by the recognition of bacterial products 17878154_caspase-1-mediated necrosis appears to be tightly controlled and differentially regulated by proteasomes depending on the source of caspase-1 induction. 17878377_caspase-activating recruiting domain and caspase-1 are critical for IFN-gamma-mediated control of Anaplasma phagocytophilum infection 17959595_characterization of caspase-1 substrates identifies the glycolysis pathway as a caspase-1 target and provides new insights into its function during pyroptosis and septic shock. 18003616_NOS2 interacts with CD74 (the MHC II-associated invariant chain), and the degradation of CD74 by caspases in immature dendritic cells was inhibited upon treatment with nitric oxide donor. 18070936_IPAF and capase-1 have key roles in innate immune responses to the pathogen P. aeruginosa 18081695_demonstrate a dual role for the Pseudomonas aeruginosa Type III secretion system in the regulation of Caspase-1 mediated IL-1beta production 18195026_This study showed that that pneumolysin plays a key role in the host response to S. pneumoniae, particularly in the induction of interleukin-1alpha and caspase-1-dependent cytokines. 18256184_Ipaf-dependent detection of cytosolic flagellin is a conserved mechanism by which macrophages detect the presence of pathogens 18299270_IL-18, ICE and IL-18 R are overexpressed in testicular tissue from sexually immature as compared to mature mice 18329368_Study shows that secretion of the leaderless proteins proIL-1alpha, caspase-1, and fibroblast growth factor (FGF)-2 depends on caspase-1 activity. 18337499_distinct activation pathways elicit the conserved cell death effector mechanism of caspase-1-mediated pyroptosis 18411310_Flightless-I, a member of the gelsolin superfamily of actin-remodeling proteins, interacts and regulates both caspase-11 and caspase-1. 18490757_Listeria monocytogenes activates caspase 1 in macrophages via multiple pathways, all of which detect the presence of bacteria within the cytosol 18497737_inhibition of caspase-1 blocked the cleavage of PPARgamma proteins in cultured adipocytes 18523249_Activation of caspase-1 in macrophage cytoplasm is a key event that results in interleukin (IL)-18 and interferon (IFN)-gamma production upon infection with Listeria monocytogenes. 18667412_These results demonstrate for the first time the existence of a nucleotide binding and oligomerization domain-like receptor/caspase-1/caspase-7 cascade and the existence of distinct activation mechanisms for caspase-3 and -7. 18671821_The authors report that heat-shocking anthrax lethal toxin-sensitive macrophages rapidly protects them against cytolysis by inhibiting caspase-1 activation 19039317_Adipose and hepatic IL-18 protein expressions are increased in obese mice. However, in contrast to liver, the adipose IL-18 protein level appears to be upregulated through a post-transcriptional mechanism probably involving caspase-1. 19130485_Zinc is an intrinsic regulator of caspase-1 activation and IL-1beta secretion. Zinc-dependent effect is upstream of the inflammasome and caspase-1 activation and acts by regulating pannexin-1 activity. 19131592_study showed p202 is an inhibitor of DNA-induced caspase activation; conversely AIM2 was required for caspase activation by cytoplasmic dsDNA; work indicates HIN-200 proteins can act as pattern recognition receptors mediating response to cytoplasmic dsDNA 19158675_observations identify AIM2 as a new receptor for cytoplasmic DNA, which forms an inflammasome with the ligand and ASC to activate caspase-1 19179418_Our results indicate that caspase-1-dependent immune mechanisms play an essential role in resistance against B. pseudomallei infection. 19237518_The data show that Legionella pneumophila-mediated caspase-1 activation involves the coordinate activities of inflammasomes differentially regulated by Ipaf and Asc. 19362023_Nlrp3(-/-) and Casp1(-/-) mice were more susceptible than wild-type mice after infection with a pathogenic influenza A virus 19487481_These findings reveal that the caspase 1 inflammasome is not critical to host defense against the extracellular pathogen Borrelia burgdorferi. 19587006_inflammasome components NLRP3 and ASC, which are important for caspase-1 activation and IL-1beta release; macrophage/monocyte necrosis and HMGB1 release are controlled independently of caspase-1 19683679_These findings implicate SopE-driven Rho GTPase-mediated caspase-1 activation in stromal cells as a mechanism eliciting mucosal inflammation during Salmonella typhimurium infection. 19933859_Caspase-1-, caspase-8-, and calpain-independent IL-33 production by macrophages and/or mast cells may contribute to the pathogenesis of T helper (Th)2-cell type allergic inflammation. 20197547_Cleavage of sphingosine kinase 2 by caspase-1 provokes its release from apoptotic cells. 20200276_During M. tuberculosis infection IL-1beta can be generated by a mechanism that does not require TLR signaling or caspase-1. 20226691_Casp1(-/-) mice exhibited defects in mucosal tissue repair and succumbed rapidly after dextran sulfate sodium administration. 20595632_Data indicate that caspase-1 activation is critical for rapid disease progression in rodents challenged with anthrax lethal toxin. 20671924_Data show that macrophages from A/J mice exhibited reduced caspase-1 activation and IL-1beta secretion against M. leprae compared to C57BL/6 macrophages. 20808838_the role of NLRP3 (NALP3), its adaptor protein PYCARD (ASC), and caspase-1 during infection with Mycobacterium tuberculosis 20814576_caspase-1 activation by SopE and the type iii secretion system-1 20823203_the activation of caspase-1 by bacteria expressing type 3 secretion systems allows for rapid recognition of Pseudomonas aeruginosa expressing conserved functions associated with virulence. 20920345_These results suggest that caspase-1 plays a critical role in the cascade of events involved in the genesis of inflammatory hypernociception by promoting IL-1beta maturation. 21037094_Multiple factors from both bacterium and host play a role in eliciting caspase-1 activation during infection with Aeromonas veronii biotype sobria. 21057511_demonstrates that activation of caspase-1 clears intracellular bacteria in vivo independently of IL-1beta and IL-18 21106820_Caspase-1 and interleukin-18 mediate central nervous system inflammation in a mouse model of multiple sclerosis. 21109192_Caspase-1 is present in adipose tissue and is a regulator of adipogenesis, whereas inhibition of caspase-1 in obese animals improves insulin sensitivity. 21118981_Data show that caspase-1-deficient mice have enhanced tumor formation, and suggest that the NLRC4 inflammasome is central to colonic inflammation-induced tumor formation through regulation of epithelial cell response to injury. 21147462_Catalytic activity of Caspase-1 is required for Asc-independent cell death 21170303_data confirm an inverse relationship between murine macrophage sensitivity to lethal toxin and mouse susceptibility to spore infection, and establish roles for Nlrp1b(S), caspase-1, and IL-1beta in countering anthrax infection. 21178113_Murine macrophages infected with B. cenocepacia K56-2 produce proinflammatory cytokine IL-1beta in a TLR4 and caspase-1-mediated manner. 21533069_A Yersinia effector with enhanced inhibitory activity on the NF-kappaB pathway activates the NLRP3/ASC/caspase-1 inflammasome in macrophages. 21666957_Data suggest that caspase-1 is hepatoprotective, in part through regulation of cell death pathways in the liver after major trauma. 21677131_Increased caspase-1 expression and activity after cecal ligation and puncture injury appears to contribute to diminished innate immune function that is not detectable in uninjured controls. 21681738_Data show that caspase-1 and ASC have strong protective antifungal capacities by controlling Th1 and Th17 responses during disseminated candidiasis. 21708993_caspase-1 activation of interleukin-1beta and IL-18, which is associated with the inflammasome pathway, does not contribute to P. berghei ANKA-induced immunopathology. 21731762_Chlamydia pneumoniae-induced IL-1beta secretion by macrophages requires TLR2/MyD88 and NLRP3/ASC/Caspase-1 signaling. 21771913_Formation of caspase-1-containing puncta correlated with caspase-1 processing, suggesting a role for the Asc/NLRC4/caspase-1 complex in caspase-1 cleavage. 21804020_studies indicate two conserved secretion pathways are initiated by caspase-1, lysosome exocytosis, and a parallel pathway resulting in cytokine release, and both enhance the antimicrobial nature of pyroptosis 21957143_Activation of caspase-1 and the subsequent maturation of IL-1beta and IL-18 in response to pneumolysin-producing Streptococcus pneumoniae are critically dependent on apoptosis-associated speckle-like protein(ASC), including AIM2 and NLRP3. 22079982_The NLRC4 inflammasome triggers the restriction of Legionella pneumophila replication by mechanisms dependent and independent of caspase-1 in mouse macrophages. 22106299_Data show that three components of the inflammasome--apoptosis speck-like protein containing a caspase-recruitment domain (ASC), cryopyrin, and caspase-1, localized to the granulation tissue and cardiomyocytes bordering the infarct. 22109549_The acute inflammatory response to biomaterials requires nucleotide-binding domain and leucine-rich repeat-containing 3 (Nlrp3), apoptosis-associated speck-like protein containing CARD (Asc), and caspase-1. 22143773_Salmonella mutants lacking YdiV are unable to fully repress flagellin at systemic sites, rendering them vulnerable to caspase-1 mediated colonization restriction 22213227_Caspase-1 was initially identified as the main IL-1beta-converting enzyme, and the upstream events leading to caspase-1 activation were identified as so-called inflammasome complexes. 22234969_Caspase-1 activation was found to occur in hypoxic hepatocellular carcinoma cells in a process that was dependent on the extracellular release of HMGB1 22235111_transient P2X(7) receptor activation and Ca(2+) overload act as a death trigger for native mouse macrophages independent of Panx1 and pro-inflammatory caspase-1 and TLR signaling. 22265992_caspase-1 promotes atherosclerosis by enhancing the inflammatory status of the lesion through a mechanism likely involving activation of lesion-associated immune cells and IFN-gamma expression. 22411067_Caspase-1-knockout (Casp-/-) mice on the methionine- and choline-deficient diet showed marked reduction in mRNA expression of genes involved in inflammation and fibrogenesis compared to wild type mice on the same diet. 22520462_YopJ subverts the Nod2/RICK/TAK1 pathway and activates caspase-1 to induce intestinal barrier dysfunction. 22634614_Bacterial RNA from both Gram-positive and Gram-negative bacteria induces activation of caspase-1 by murine dendritic cells and bonemarrow derived macrophages. 22647887_Data show that podocytes expressed 3 essential components of the NALP3 inflammasome complex, namely, NALP3, ASC, and caspase 1. 22819845_these findings suggest that caspase-1 plays a critical role in vascular inflammation and atherosclerosis, and that modulation of caspase-1 could be a potential target for prevention and treatment of atherosclerosis. 22883230_high-fat diet induces the cleavage of SIRT1 protein in adipose tissue by the inflammation-activated caspase-1, providing a link between dietary stress and predisposition to metabolic dysfunction. 22911706_Mycobacterium tuberculosis infection of dendritic cells leads to partially caspase-1/11-independent IL-1beta and IL-18 secretion but not to pyroptosis. 22914048_Sterile dead cells elicit an equivalent influx of neutrophils and monocytes in caspase-1-deficient and wild-type mice. 23024281_There is no significant difference in IL-1beta processing between C57BL/6 strain of mouse and caspase-1-deficient or NLRC4-deficient Pseudomonas-infected corneas. 23035103_Hypoxia/reoxygenation more than doubled caspase 1 activity in amygdala. Amygdala-based anterograde amnesia after hypoxia/reoxygenation is sustained by IL-1beta generated through adenosine-dependent activation of caspase 1 after reoxygenation. 23148704_Data indicate that Lyme arthritis is apoptosis-associated speckle-like protein ASC- and caspase-1-dependent, but independent of NLRP3 protein. 23160218_novel function for caspase-1 in controlling intestinal TG absorption and hepatic TG secretion 23209292_Deubiquitinases regulate the activity of caspase-1 and interleukin-1beta secretion via assembly of the inflammasome 23245324_YopM directly binds caspase-1, which both inhibits caspase-1 activity and sequesters it to block formation of the mature inflammasome. 23297192_Data show that bee venom detected by inflammasome and trigger activation of caspase-1 and secretion of proinflammatory cytokine IL-1beta is pyrin domain-containing 3 NLRP3, apoptosis-associated speck-like protein ASC, and IL-1 receptor dependent. 23303804_Caspase 1 and 3 are involved in Chlamydia-induced infertility. 23307811_Data indicate that Legionella activation of caspase-11 stimulated activation of caspase-1 through PYRIN domain-containing protein 3 (NLRP3) and apoptosis-associated speckle-like protein (ASC). 23356698_SMS suppressed the activation of NF-kappaB and receptor interacting protein-2/caspase-1. 23371061_A role for caspase-1 and IL-1beta in the IL-1 receptor-dependent Th17 response manifest in nitrogen dioxide-promoted allergic airway disease. 23409132_the absence of caspase-1 ameliorates the injurious effects of high fat diet-induced obesity on the liver 23487794_caspase-1 influences lipid metabolism through multiple mechanisms, not limited to cytokines. 23589298_Caspase 1 activation is protective against hepatocyte cell death by up-regulating beclin 1 protein and mitochondrial autophagy in the setting of redox stress. 23630667_These data suggest that sublytic caspase-1 activity could delay the formation of stable AIM2 speck, an inflammasome complex associated with cell death. 23644505_Data indicate that active caspase-1 accumulates on phagosomes and acts locally to control the pH by modulating buffering by the NADPH oxidase NOX2. 23812211_Caspase-1 deletion increases hematopoiesis and radioresistance of bone marrow cells in vitro. 23942110_LRRFIP2 enhances the interaction between Flightless-I and caspase-1, facilitating the inhibitory effect of Flightless-I on caspase-1 activation. 23975862_ASC controls IFN-gamma levels in an IL-18-dependent manner in caspase-1-deficient mice infected with Francisella novicida. 23977202_Caspase-1 is critical for controlling systemic dissemination, severe inflammation and the integrity of the mucosal barrier. 24022484_Lactic acidosis, induced by the addition of 25 mm lactic acid, also induced the release of 20-kDa IL-1beta. This 20-kDa product was produced independently of NLRP3 and caspase-1 but was inhibited by the cathepsin D inhibitor pepstatin A. 24058709_Silencing of Nsd1 followed by LLO stimulation led to increased caspase-1 activation, enhanced post-translational maturation of IL-1beta and IL-18 and elevated pyroptosis, a form of cell death associated with inflammation. 24098823_These results demonstrate a role for the NLRP3 inflammasome in the control of Trypanosoma cruzi infection and identify NLRP3-mediated, caspase-1-dependent and IL-1R-independent nitric oxide production as a novel effector mechanism. 24158569_Identify a novel innate immune signaling pathway (NLRP3-ASC-caspase-1-IL-1beta) activated by Ni(2+). 24312444_Mechanistically, KCs from caspase-1/11(-/-)-tp mice showed less cholesterol crystals, enhanced cholesterol efflux and increased autophagy. 24411736_This study demonistrated taht Neuronal Ig/Caspr recognition promotes the formation of axoaxonic synapses in mouse spinal cord. 24449582_Results indicate that caspase 1 is an essential component in the development of lupus and its associated vascular dysfunction and that it may play an important role in the cross-talk between environmental exposures and autoimmunity development. 24453977_NLRP12/NLRP3-dependent activation of caspase-1 is likely to be a key event in mediating systemic production of IL-1beta and hypersensitivity to secondary bacterial infection during malaria. 24492532_ASC-driven caspase-1 autoprocessing and speck formation are dispensable for the activation of caspase-1 and the NLRP1b inflammasome. 24516390_activation of caspase-1 by Shigella is triggered by the rod protein MxiI that interacts with Naip2 to induce activation of the Nlrc4 inflammasome independently of the Pkcdelta kinase 24528867_Caspase-1 directly cleaved TRIF to diminish TRIF-mediated signaling, resulting in inhibition of autophagy and in reduced type I interferon production in Pseudomonas aeruginosa infected macrophages. 24699513_The NOD-like receptor NLRP1a/Caspase-1 pathway is the best candidate to mediate the parasite-induced cell death. 24789624_The NLRP3/ASC/caspase-1 axis participates in the regulation of pro-imflammatory cytokine secretion in RAW264.7 macrophages. 25127135_Caspase-1 exhibits all-or-none activation at the single-cell level, with similar activation kinetics irrespective of the type of inflammasome or the intensity of the stimulus. 25162221_Non-classical proIL-1beta activation during mammary gland infection is pathogen-dependent but caspase-1 independent. 25193701_Chlamydia pneumoniae harnesses host NLRP3 inflammasome-mediated caspase-1 activation for optimal intracellular growth in murine macrophages. 25313054_Caspase-1 activation by inflammasomes results in mitochondrial damage. 25372293_caspase-1/ASC inflammasomes play a significant role in the activation of IL-1beta/ROS and NF-kappaB signaling of cytokine gene expression for T. cruzi control in human and mouse macrophages. 25404286_Data suggest that caspase-1 autoproteolysis in NLR family pyrin domain containing 1b (NLRP1b) and NLR family pyrin domain containing 3 (NLRP3) inflammasome function may have implications for pathogen recognition and response. 25452302_studies demonstrate a role of Nedd8 in regulating caspase-1 activation following inflammasome activation 25501827_GATA4, a key regulator of cardiomyocyte survival, is a substrate for caspase-1. 25524927_The signalling pathway governing the inflammasome formation in the heart requires a priming signal in order for an active NLRP3 to induce caspase-1 activation 25562666_There may be a protective role of caspase-1 in keratinocytes during UVB-induced skin cancer development through the induction of apoptosis. 25633245_The resilience of casp1(-/-) mice to SIRS lethality is associated with a lower inflammatory response, loss of hippocampal gene coexpression patterns, and increased hippocampal Adamts1 gene expression. 25684123_These findings suggest that endogenous caspase-1/11 activity, independent of the NLRP3 inflammasome, not only promotes the magnitude of the inflammatory response, but also suppresses protective immunity in the neonate 25705884_Inflammasome-induced activation of an intracellular caspase-1/calpain cysteine protease cascade degraded filamin. Caspase-1 specifically facilitated actin exposure, which released of highly procoagulant MPs from filopodia. 25705917_Early hyperlipidemia promotes endothelial cell activation before monocyte recruitment via a caspase-1-sirtuin 1-activator protein-1 pathway. 25714839_This study highlights a critical role of caspase-1 activation and NLPR3/ASC inflammasome complex in the down-modulation of IL-33 in vivo and in vitro. 25795756_data suggest that IL-1, independently of caspase-1, contributes to the pathogenesis of EBA. 25815426_In an inflammatory microenvironment, caspase-1 is regulated by NF-kappaB. 25817537_Data (including data from studies in knockout mice) suggest that caspase-1/11 are involved in development of atherosclerosis; hematopoietic deletion of caspase-1/11 results in strong reduction in atherosclerotic plaque size. 25929181_Data suggest that Casp1 is activated and expression/nucleocytoplasmic translocation of Hmgb1 (high mobility group box 1) is up-regulated in pulmonary mucosa in asthma; these changes involve PI3K (phosphatidylinositol 3-kinase) signal pathway. 26158519_These results argue that caspase-1, -4 or -5 can be recruited to inflammasomes under specific circumstances, often leading to distinctly organized and localized complexes that may impact the functions of these proteases 26216893_Data indicate that genetic ablation of caspase-1 and -11 from cpdm mice significantly reduced skin inflammation in sharpin-deficient mice. 26232428_Data show that caspase-1 but not caspase-11 was essential for the functional activities of the NLRC4 inflammasome. 26320999_Caspase-1-activated IL-18 induces IFN-gamma to prime caspase-11 and rapidly clear Burkholderia thailandensis infection. 26349656_caspase-1-/- mice exhibited resistance to indomethacin-induced small intestinal damage 26405027_Caspase-1-/- mice have intact immune responses but do not develop an expanded marginal zone B cell population in response to pristane-induced lupus. This may be one explanation for reduced IgG autoantibody production in these mice. 26466953_Internalized Cryptococcus neoformans Activates the Canonical Caspase-1 and the Noncanonical Caspase-8 Inflammasomes. 26567013_The results demonstrate the activation of the caspase 1 precursor by caspase 11 and suggest a new mechanism of protection of Theiler murine encephalomyelitis virus-infected astrocytes from apoptosis. 26676570_Adult neural stem cells (ANSCs) expressed NLRP3-containing inflammasome and alpha-syn activated both TLR4/NF-kappaB and NLRP3/caspase-1 signals in ANSCs. 26686473_Caspase-11 targets cofilin via the RhoA GTPase, whereas caspase-1 engages the Slingshot phosphatase 26742834_Prophylactic treatment with capsase-1 inhibitor VX756 ameliorated the severity and progression of collagen-induced arthritis. 26810037_study uncovered an important role for YopJ in inhibiting caspase-1 in activated macrophages and in promoting Yersinia virulence 26928596_Chronic kidney disease-mediated increase of contractile markers in vascular smooth muscle cells and increased expression of VSMC migration molecule alphavbeta3 integrin in NH formation were caspase-1 dependent. 26989816_TNF-alpha induces caspase-1 activation in an inflammasome-independent manner in 3T3-L1 cells and that the ERK-dependent expression of NLRP3 may play a role independently of its canonical role as a component of inflammasomes. 27053298_Data show that inhibiting caspase-1 activity in the NLR family, pyrin domain-containing 3 protein (NLRP3) inflammasome decreased gastric cell apoptosis. 27108091_Demonstrate the utility of intracellular caspase 1 and extracellular CTSS proteolytic activities as surrogate biomarkers of lysosomal rupture and acute inflammation. 27251848_it was found that GSK2656157 specifically inhibited ER stress induced by large amount of LPS and reduced LPS-induced IL-1beta production through inhibition of Caspase 1 activation. 27339879_caspase-1 regulates dopaminergic neuronal death in the pathogenesis of Parkinson's disease in mice via caspase-7/PARP1/AIF pathway. 27402835_variant p.C284A caspase-1 stabilizes pyroptosome formation, potentially enhancing inflammation by two IL-1beta-independent mechanisms: pyroptosomes convey an enhanced inflammatory stimulus through the recruitment of additional proteins (such as RIP2, receptor interacting protein kinase 2), which is further amplified through pyroptosome and cell division. 27419363_dimerized or endogenous caspase-8 can also directly cleave IL-1beta into its biologically active form, in the absence of canonical inflammasome components. 27450556_The most well-known function of active caspase-1 is to cleave the proforms of inflammatory cytokines IL-1beta and -18 into their active forms in response to inflammatory stimuli in immune cells. Caspase-1 has multiple functions in addition to this cytokine maturation role. It is at the center of many cell responses to stress and inflammation not just in immune cells but in other cell types, such as epithelia. Review. 27607578_Hemorrhagic shock primes for lung vascular endothelial cell pyroptosis following lipopolysaccharide exposure through TLR4, which activates Nlrp3, and subsequently induces caspase-1 activation. 27643624_caspase-1 activation is an upstream event of apoptotic caspase-7 induction in renal tubulointerstitial fibrosis 27702746_Findings demonstrate a critical role of caspase-1 in macrophage-driven inflammation in the adipose tissue and the development of obesity. 27808091_The enzymatic activities of caspase-1 and caspase-11 are required for growth inhibition in different cell types.These results reveal that these proteases have important functions beyond the direct induction of pyroptosis and proinflammatory cytokine secretion in the control of growth and elimination of cytosolic bacteria. 27894300_Report direct role of pleural cells in the pathogenesis of bleomycin-induced pulmonary fibrosis via caspase-1/IL-1beta pathway. 27998728_our studies have uncovered a specific role for caspase-1-mediated IL-1beta release in the manifestation of Familial Mediterranean Fever 28040362_Study suggests that Casp1 has a limited role in the development of burn-induced pain. 28153878_Inflammasome NLRP3-caspase-1-mediated degradation of smooth muscle cell contractile proteins may contribute to aortic biomechanical dysfunction and aortic aneurysm/dissection development. 28345580_Casp1Null mice expressed caspase-11 proteins and did not secrete IL-1beta and IL-18 or undergo pyroptosis in response to canonical NLRP3, NLRC4 or AIM2 inflammasome activators. 28407357_Nodakenin inhibited the mRNA expression and production of pro-inflammatory cytokines and caspase-1 activation 28410991_Data indicate that NLRC4 activation in Intestinal epithelial cells (IECs) leads to cell expulsion and IL-18 release, and implicate Caspase-8 in NLRC4 inflammasome responses in vivo by generation of doubly deficient in Caspase-1 and Caspase-8. 28423682_RIP3-mediated activation of caspase-1 rather than necroptosis-dependent inflammation was responsible for aggressive inflammation in influenza A (H7N9) virus-infected mice. 28660207_we assessed the role of RIP3 in synergy with Caspase-1 in the induction of IL-1beta production in BMDM after either LPS/ATP or Chlamydia muridarum stimulation. The possibility of pyroptosis and necroptosis interplays and the role of RIP3 in IL-1beta production during Chlamydia muridarum infection in BMDM was investigated as well. 28790324_The H7N9 influenza A virus infection results in lethal inflammation in the mammalian host via the NLRP3-Casp1- IL1R1-expressing inflammasomes. 28801921_The results of this study indicated that beta-Amyloid peptide-induced ca |
ENSG00000137752+ENSG00000204397 |
CASP1+CARD16 |
96.698274 |
13.9457158482 |
3.801750 |
0.46367091 |
60.458018 |
0.00000000000000751645638250312795848101696725527321832111167509982152523662080056965351104736328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000001892407663112192438247676111916068868938189256878601440803322475403547286987304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
176.847436680402 |
57.8209328453237 |
12.6811300764668 |
3.34935757992676 |
| ENSMUSG00000025911 |
76187 |
Adhfe1 |
protein_coding |
Q8R0N6 |
FUNCTION: Catalyzes the cofactor-independent reversible oxidation of gamma-hydroxybutyrate (GHB) to succinic semialdehyde (SSA) coupled to reduction of 2-ketoglutarate (2-KG) to D-2-hydroxyglutarate (D-2-HG). L-3-hydroxybutyrate (L-3-OHB) is also a substrate for HOT when using 2-KG as hydrogen acceptor, resulting in the formation of D-2-HG (By similarity). {ECO:0000250}. |
Acetylation;Alternative splicing;Lipid metabolism;Mitochondrion;Oxidoreductase;Phosphoprotein;Reference proteome;Transit peptide |
|
|
mmu:76187; |
mitochondrion [GO:0005739]; alcohol dehydrogenase (NAD+) activity [GO:0004022]; hydroxyacid-oxoacid transhydrogenase activity [GO:0047988]; metal ion binding [GO:0046872]; glutamate catabolic process via 2-oxoglutarate [GO:0006539]; lipid metabolic process [GO:0006629] |
17559793_Adhfe1 is a novel alcohol dehydrogenase with mitochondrial localization |
ENSG00000147576 |
ADHFE1 |
55.358452 |
2.5554076830 |
1.353553 |
0.35598539 |
14.339944 |
0.00015259256414608049620337815177606444194680079817771911621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008724812076100217682686488451793138665379956364631652832031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
81.5116336550336 |
20.3177387947357 |
31.8977023504726 |
6.15147878165821 |
| ENSMUSG00000025915 |
170755 |
Sgk3 |
protein_coding |
Q9ERE3 |
FUNCTION: Serine/threonine-protein kinase which is involved in the regulation of a wide variety of ion channels, membrane transporters, cell growth, proliferation, survival and migration. Up-regulates Na(+) channels: SCNN1A/ENAC and SCN5A, K(+) channels: KCNA3/KV1.3, KCNE1, KCNQ1 and KCNH2/HERG, epithelial Ca(2+) channels: TRPV5 and TRPV6, chloride channel: BSND, creatine transporter: SLC6A8, Na(+)/dicarboxylate cotransporter: SLC13A2/NADC1, Na(+)-dependent phosphate cotransporter: SLC34A2/NAPI-2B, amino acid transporters: SLC1A5/ASCT2 and SLC6A19, glutamate transporters: SLC1A3/EAAT1, SLC1A6/EAAT4 and SLC1A7/EAAT5, glutamate receptors: GRIA1/GLUR1 and GRIK2/GLUR6, Na(+)/H(+) exchanger: SLC9A3/NHE3, and the Na(+)/K(+) ATPase. Plays a role in the regulation of renal tubular phosphate transport and bone density. Phosphorylates NEDD4L and GSK3B. Positively regulates ER transcription activity through phosphorylation of FLII. Negatively regulates the function of ITCH/AIP4 via its phosphorylation and thereby prevents CXCR4 from being efficiently sorted to lysosomes. {ECO:0000269|PubMed:15774535, ECO:0000269|PubMed:15774536, ECO:0000269|PubMed:21113728, ECO:0000269|PubMed:21451460, ECO:0000269|PubMed:21865597}. |
3D-structure;ATP-binding;Cytoplasmic vesicle;Endosome;Kinase;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:170755; |
cytoplasmic vesicle [GO:0031410]; early endosome [GO:0005769]; recycling endosome [GO:0055037]; ATP binding [GO:0005524]; molecular adaptor activity [GO:0060090]; phosphatidylinositol binding [GO:0035091]; potassium channel regulator activity [GO:0015459]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; intracellular signal transduction [GO:0035556]; localization [GO:0051179]; negative regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001240]; peptidyl-serine phosphorylation [GO:0018105] |
15126499_CISK-PX dimerizes in the presence of the linker between the PX domain and kinase domain, suggesting a multivalent mechanism in membrane localization of CISK 15240817_SGK3 plays an important role in postnatal hair follicle development as illustrated by SGK3 knockout mice. The hair defect is linked to reduced proliferation and reduced nuclear accumulation of beta catenin in hair bulb keratinocytes. 15240817_role for SGK3 in normal postnatal hair follicle development, possibly involving effects on beta-catenin/Lef-1-mediated gene transcription 15728833_SGK3 is differentially regulated in hippocampus after contextual fear conditioning & reactivation of the context-shock memory. Consolidation changes are recapitulated during reconsolidation. Retrieval-induced transcription depends on memory remoteness. 15774536_SGK3, and to a lesser extent SGK2, but not SGK1, increase glutamate-induced currents by increasing the abundance of GluR1 protein in the cell membrane 15971077_SGK3 is required for adequate intestinal Na(+) coupled glucose transport and impaired glucose absorption may contribute to delayed growth and decreased plasma glucose concentrations of SGK3 deficient mice. 16103225_These results reveal a novel and important function for Sgk3 in controlling life and death in the hair follicle. 16246437_Lack of SGK3 leads to subtle behavioral defects which may result from deranged neuronal regulation of transporters and ion channels. 16537821_The sgk1(-/-)/sgk3(-/-) mice share the delayed hair growth with sgk3(-/-) mice and the modestly impaired renal salt retention with sgk1(-/-) mice. 16565488_SGK3 is a very important and characteristic molecule that plays a critical role in both hair follicle morphogenesis and hair cycling. 17914447_SGK3 mutation responsible for fuzzy hair phenotype in mice 19293151_These findings support the model that CISK phosphorylates FLII and activates nuclear receptor transcription and suggest a new cell survival signaling pathway mediated by PI 3-kinase and CISK. 19433625_SGK3 and Akt2 both appear to play important roles in postnatal hair follicle morphogenesis, likely because of their redundant regulation of beta-catenin-dependent transcriptional processes, which control hair follicle cell proliferation 20686074_Data suggest that SGK3 is critical for proper mast cell function. 21451460_SGK3 has a subtle but significant role in the regulation of renal tubular phosphate transport and bone density. 21865597_The study identifies SGK3 as a novel endosomal kinase that acutely regulates Na(+)/H(+) exchanger 3 (NHE3) in a PI3K-dependent mechanism. 21980074_SGK3 plays a previously unappreciated role in glucose homeostasis, likely through direct effects within pancreatic beta-cells, to stimulate proliferation and insulin release 23171960_SGK3 contributes to the regulation of store-operated calcium entry into and migration of dendritic cells. 26824455_SGK3 is a positive regulator of KCNA5, which is at least partially effective by abrogating the effect of Nedd4-2. 27457803_SGK1 and SGK3 are expressed in multiple microglial cell lines. SGK1 and SGK3 may play an important role in regulating microglial viability and inflammatory responses. 28935820_Taken together, these data strongly suggest that SGK3 plays a significant role in regulating podocyte function, likely by controlling the expression and activity of GSK3. 30385740_The SGK3-triggered ubiquitin-proteasome degradation of podocalyxin and ezrin in podocytes was associated with the stability of the PC/ezrin complex. 30975125_Study data from human hepatocellular carcinoma (HCC) liver samples, human HCC cells, and Sgk3 knockout mice model suggest that SGK3 plays a role in transducing helical domain mutant PIK3CA signaling during liver tumor development. 36269185_MicroRNA-92a-3p Regulates Retinal Angiogenesis by Targeting SGK3 in Vascular Endothelial Cells. |
ENSG00000104205 |
SGK3 |
146.184759 |
0.1017067966 |
-3.297512 |
0.28978285 |
125.135997 |
0.00000000000000000000000000004752351180999337517593619502767970460766834813700189198470216258902635042299811979349399848615576047450304031372070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000033984888821118266898598197783435048736169385242597011501738517682152148757096510323094662453513592481613159179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.0555075688207 |
5.47104413508483 |
266.01474500205 |
34.8561639794738 |
| ENSMUSG00000025920 |
29819 |
Stau2 |
protein_coding |
Q8CJ67 |
FUNCTION: RNA-binding protein required for the microtubule-dependent transport of neuronal RNA from the cell body to the dendrite. As protein synthesis occurs within the dendrite, the localization of specific mRNAs to dendrites may be a prerequisite for neurite outgrowth and plasticity at sites distant from the cell body (By similarity). {ECO:0000250|UniProtKB:Q68SB1}. |
3D-structure;Alternative splicing;Cytoplasm;Endoplasmic reticulum;Microtubule;Nucleus;Phosphoprotein;Reference proteome;Repeat;RNA-binding;Transport |
|
|
mmu:29819; |
axon [GO:0030424]; cytoplasm [GO:0005737]; cytoplasmic stress granule [GO:0010494]; dendrite [GO:0030425]; dendrite cytoplasm [GO:0032839]; dendritic shaft [GO:0043198]; endoplasmic reticulum [GO:0005783]; messenger ribonucleoprotein complex [GO:1990124]; microtubule [GO:0005874]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; nuclear membrane [GO:0031965]; nucleolus [GO:0005730]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; double-stranded RNA binding [GO:0003725]; Hsp70 protein binding [GO:0030544]; kinesin binding [GO:0019894]; mitogen-activated protein kinase binding [GO:0051019]; mRNA binding [GO:0003729]; ribosome binding [GO:0043022]; anterograde dendritic transport of messenger ribonucleoprotein complex [GO:0098964]; cellular response to oxidative stress [GO:0034599]; eye morphogenesis [GO:0048592]; germ cell development [GO:0007281]; intracellular mRNA localization [GO:0008298]; positive regulation of dendritic spine morphogenesis [GO:0061003]; positive regulation of long-term synaptic depression [GO:1900454]; positive regulation of synapse assembly [GO:0051965]; protein localization to synapse [GO:0035418]; regulation of actin cytoskeleton organization [GO:0032956]; regulation of filopodium assembly [GO:0051489]; regulation of gene expression [GO:0010468] |
12140260_Data show that Stau2 is localized to the neuronal soma and dendrites, but it does not colocalize with Stau1-containing particles. 16277607_zinc finger RNA binding protein is a native component of Staufen2-containing granules and likely plays its role during early steps of RNA transport and localization 22902294_in complex with RNA regulates maintenance and differentiation of neural stem cells 22902295_Stau2 is distributed asymmetrically during progenitor divisions in the developing mouse cortex, preferentially segregating into the Tbr2( ) neuroblast daughter, taking with it a subset of RNAs. 27433972_Stau2 is required for spindle formation and timely meiotic progression in mouse oocytes. 28683307_Staufen2 interacts with intron i16 containing CaMKIIalpha mRNA and enhances its distal dendritic localization. 29496644_Together, our data suggest that Stau2 contributes to novelty preference and explorative behavior that is a driver for proper spatial learning in mice. 30971701_Study shows that double-stranded RNA binding domains (dsRBDs) 1 and 2 of mStau2 bind RNA with similar affinities and kinetics as dsRBDs 3 and 4. While RNA binding by these tandem domains is transient, all four dsRBDs recognize their target RNAs with high stability. Rescue experiments in Drosophila oocytes demonstrate that mStau2 partially rescues dmStau-dependent mRNA localization. 30979012_Stau2 downregulation (Stau2(GT)) led to an increase of glutamate receptor ionotropic delta subunit 2 (GluD2) in Purkinje cells when animals performed physical activity by voluntary wheel running compared with the age-matched wildtype (WT) mice (C57Bl/6J). 34109316_An in vivo genome-wide CRISPR screen identifies the RNA-binding protein Staufen2 as a key regulator of myeloid leukemia. 34161769_Pumilio2 and Staufen2 selectively balance the synaptic proteome. 34345913_STAU2 binds a complex RNA cargo that changes temporally with production of diverse intermediate progenitor cells during mouse corticogenesis. |
ENSG00000040341 |
STAU2 |
67.026336 |
0.2618821072 |
-1.933011 |
0.51671456 |
12.973558 |
0.00031592114623580681582321050271389140107203274965286254882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016673980940623040099268781233377012540586292743682861328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.7743451735014 |
9.7958180531787 |
102.238161505822 |
26.6770446741112 |
| ENSMUSG00000025927 |
21419 |
Tfap2b |
protein_coding |
Q61313 |
FUNCTION: Sequence-specific DNA-binding protein that interacts with inducible viral and cellular enhancer elements to regulate transcription of selected genes. AP-2 factors bind to the consensus sequence 5'-GCCNNNGGC-3' and activate genes involved in a large spectrum of important biological functions including proper eye, face, body wall, limb and neural tube development. They also suppress a number of genes including MCAM/MUC18, C/EBP alpha and MYC. AP-2-beta appears to be required for normal face and limb development and for proper terminal differentiation and function of renal tubular epithelia. {ECO:0000250|UniProtKB:Q92481}. |
Activator;DNA-binding;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:21419; |
nucleus [GO:0005634]; chromatin binding [GO:0003682]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; anatomical structure development [GO:0048856]; aorta morphogenesis [GO:0035909]; collecting duct development [GO:0072044]; distal tubule development [GO:0072017]; ductus arteriosus closure [GO:0097070]; fat cell differentiation [GO:0045444]; forelimb morphogenesis [GO:0035136]; gene expression [GO:0010467]; glucose metabolic process [GO:0006006]; hindlimb morphogenesis [GO:0035137]; kidney development [GO:0001822]; metanephric nephron development [GO:0072210]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuron apoptotic process [GO:0051402]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of neuron apoptotic process [GO:0043525]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of BMP signaling pathway [GO:0030510]; regulation of cell differentiation [GO:0045595]; regulation of cell population proliferation [GO:0042127]; regulation of insulin secretion [GO:0050796]; regulation of transcription by RNA polymerase II [GO:0006357]; response to xenobiotic stimulus [GO:0009410]; retina layer formation [GO:0010842]; sensory organ development [GO:0007423]; skin development [GO:0043588]; smooth muscle tissue development [GO:0048745]; sympathetic nervous system development [GO:0048485]; transcription by RNA polymerase II [GO:0006366] |
16373417_AP-2beta is a candidate gene for producing adipocyte hypertrophy and may relate to the abnormal characteristics of adipocytes observed in obesity. 16373417_AP-2beta is a candidate gene for producing adipocyte hypertrophy and may relate to the abnormal characteristics of adipocytes observed in obesity. 16954217_AP-2beta might modulate the expression of adiponectin by directly inhibiting its transcriptional activity 17911046_Hlxb9 and Tcfap2b were identified as genes whose expression was elevated in the ZPA compared to the rest of the developing limb bud. 18635823_Tfap2beta, Et-1, and Hif2alpha act in a transcriptional network during ductal smooth muscle development. Disruption of this pathway may contribute to patent ductus arteriosus by affecting development of smooth muscle in the ductus arteriosus. 19022887_The results clearly show that AP-2beta directly enhanced MCP-1 secretion by binding to its promoter. Results propose that AP-2beta positively regulates MCP-1 expression. 20097161_AP-2beta transcriptional factor is a unique regulator of IRS-1 and a candidate gene for insulin resistance. 20875861_Data suggest that AP-2beta plays critical roles in the epinephrine phenotype and maturation of adrenal chromaffin cells. 21345437_PKD is thus a common modulator of the DNA binding activity of AP-2alpha and AP-2beta through their phosphorylation for negative regulation of the ABCA1 and adiponectin genes expression, respectively. 21424113_postprandial activation of PKCmicro plays a role in disordered postprandial adipocytokine expression through AP-2beta. 21539825_Whereas AP-2alpha/beta transcription factors are in vivo not required for the onset or maintenance of noradrenergic differentiation, their essential survival functions are demonstrated for sympathetic progenitors and noradrenergic neurons. 21829553_Tfap2b is associated with the development and remodeling of mouse ductus arteriosus and limb patterning. 22411557_critical roles for AP-2 activity in retinogenesis, delineating the overlapping expression patterns of Tcfap2a, Tcfap2b, and Tcfap2c in the neural retina, and revealing a redundant requirement for Tcfap2a and Tcfap2b in horizontal and amacrine cell development 25966682_the Tfap2a and Tfap2b transcription factors were identified as two major downstream effectors of Ptf1a. 27483349_AP-2beta is required in the periocular mesenchyme for normal development of the anterior segment of the eye. 27984181_Study systematically examined the expression profile of AP-2 family in the developing mouse and chick spinal cord and found that AP-2alpha and AP-2beta are specifically expressed in post-mitotic dorsal interneurons. Subsequent functional assessment in chick embryos demonstrated that AP-2alpha and AP-2beta have distinct functions in dorsal interneuron specification and differentiation. 28277615_AP-2 beta and beta-catenin interact both in vitro through GST pull-down assays and in vivo by co-immunoprecipitation. We further identified the interaction regions to the DNA-binding domain of AP-2 beta and the 1-9 Armadillo repeats of beta-catenin. 29229773_these findings reveal that the AP-2 genes have a major function in mammalian neural crest development, influencing patterning of the craniofacial skeleton 29804851_Using the Tfap2b c.435_438delCCGG homozygous mice, study verified the nature of the human c.435_438delCCGG mutation and established a new and useful animal model to explore the function of Tfap2b and the mechanisms of patent ductus arteriosus and renal formation. 31638440_In an ongoing expression screen, this study identified that Tfap2b, Casq2, Penk, Zic1, and Zic3 are expressed in unique cell types in and around the developing eye. 32769099_Functional Divergence of Mammalian TFAP2a and TFAP2b Transcription Factors for Bidirectional Sleep Control. 32878901_Sleep Architecture in Mice Is Shaped by the Transcription Factor AP-2beta. 33460639_Anomalous incisor morphology indicates tissue-specific roles for Tfap2a and Tfap2b in tooth development. 34445433_Conditional Deletion of AP-2beta in the Periocular Mesenchyme of Mice Alters Corneal Epithelial Cell Fate and Stratification. 34822722_Deletion of transcription factor AP-2beta from the developing murine trabecular meshwork region leads to progressive glaucomatous changes. |
ENSG00000008196 |
TFAP2B |
1112.222609 |
0.3715399597 |
-1.428411 |
0.15438134 |
82.495198 |
0.00000000000000000010592715416875114439748054699309265077736507304576266271477213987139975870377384126186370849609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000040641874365199433424656864276534828625570784432297424815239139661571243777871131896972656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
588.837104245338 |
63.1311891983333 |
1584.85538055889 |
125.136749373767 |
| ENSMUSG00000025964 |
23792 |
Adam23 |
protein_coding |
Q9R1V7 |
FUNCTION: May play a role in cell-cell and cell-matrix interactions. This is a non-catalytic metalloprotease-like protein (By similarity). {ECO:0000250}. |
Alternative splicing;Cell adhesion;Cell membrane;Cleavage on pair of basic residues;Disulfide bond;EGF-like domain;Glycoprotein;Membrane;Reference proteome;Secreted;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the disintegrin family of membrane-anchored proteins that play a role in diverse biological processes such as brain development, fertilization, tumor development and inflammation. The encoded protein undergoes proteolytic processing to generate a mature polypeptide comprised of an inactive metalloprotease and disintegrin domains. Transgenic disruption of this gene in mice results in postnatal neurological defects including tremor and ataxia resulting in death by 2 weeks of age. [provided by RefSeq, Sep 2015]. |
mmu:23792; |
extracellular region [GO:0005576]; glutamatergic synapse [GO:0098978]; presynaptic membrane [GO:0042734]; metalloendopeptidase activity [GO:0004222]; cell adhesion [GO:0007155]; cellular response to leukemia inhibitory factor [GO:1990830]; proteolysis [GO:0006508] |
17333391_These results suggest ADAM23 may play roles in both early and later stage of neuronal differentiation. 19477226_PrP(C) is a novel molecular partner for ADAM23 in the nervous systems. 19549921_Functional role of ADAM23 during lung cancer metastatic progression by negatively modulating alpha(v)beta(3) integrin activation. 19796686_Data suggest that LGI1 binding to ADAM23 is necessary to correctly pattern neuronal morphology, and altered anatomical patterning contributes to autosomal dominant partial epilepsy with auditory features. 22405860_Data suggest that leucine-rich glioma inactivated 3 (LGI3) may be a candidate adipokine that is perturbed in obesity and suppresses adipogenesis through its receptor, ADAM23. 22973984_Adam23 knockdown up-regulates P27KIP1 in P19/ADAM23 knockdown cells, one reason that P19/ADAM23 knockdown cells can differentiate into neurons without retinoic acid induction. 25707359_Interaction proteomics revealed the interactors of Caspr2, including CNTN2, KCNAs, members of the ADAM family (ADAM22, ADAM23 and ADAM11), members of LGI family and MAGUKs (DLGs and MPPs). 27317750_ADAM23 expression on DCs partially governs antigen-presentation capacities to responder CD4(+) T cells. Knockdown did not alter the maturation profile of DCs but impaired cognate T cell responses, decreased antigen-specific clonal expansion, and decreased Th cytokine production. Targeting the alpha(v)beta(3) integrin receptors resulted in similar phenotypes indicating their role in the ADAM23 response. 30371220_we identified ADAM23 as a negative regulator of cardiac hypertrophy through inhibiting focal adhesion kinase-protein kinase B signaling pathway, which could be a promising therapeutic target for this malady 30965109_ADAM23 had a strong negative effect on the surface expression of Kv1.1 channels and on Kv1.1 current amplitude |
ENSG00000114948 |
ADAM23 |
2633.040296 |
2.0370739246 |
1.026498 |
0.14572923 |
48.910494 |
0.00000000000267913485597163665074006766818575097114860095182109489542199298739433288574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000505682554227508925141779189553660348380170752591311611467972397804260253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3569.846453745 |
361.210154067669 |
1752.43834334352 |
133.136327630238 |
| ENSMUSG00000026051 |
78896 |
Ecrg4 |
protein_coding |
Q99LS0 |
FUNCTION: Probable hormone that may attenuate cell proliferation and induce senescence of oligodendrocyte and neural precursor cells in the central nervous system (PubMed:20404145). ECRG4-induced senescence is characterized by G1 arrest, RB1 dephosphorylation and accelerated CCND1 and CCND3 proteasomal degradation (PubMed:20404145). {ECO:0000269|PubMed:20404145}. |
Cell membrane;Cleavage on pair of basic residues;Cytoplasm;Membrane;Reference proteome;Secreted;Signal |
|
|
mmu:78896; |
apical plasma membrane [GO:0016324]; cytoplasm [GO:0005737]; dense core granule [GO:0031045]; extracellular space [GO:0005615]; neuropeptide hormone activity [GO:0005184]; anaphase-promoting complex-dependent catabolic process [GO:0031145]; cellular senescence [GO:0090398]; central nervous system development [GO:0007417]; G1 to G0 transition [GO:0070314]; negative regulation of cell population proliferation [GO:0008285]; neuropeptide signaling pathway [GO:0007218]; positive regulation of corticosterone secretion [GO:2000854]; positive regulation of corticotropin secretion [GO:0051461]; positive regulation of corticotropin-releasing hormone secretion [GO:0051466]; regulation of cell population proliferation [GO:0042127]; response to wounding [GO:0009611]; vasopressin secretion [GO:0030103] |
19735703_ECRG4 level in differentiated chondrocytes is decreased during hypertrophic maturation 20404145_identified esophageal cancer-related gene 4 (Ecrg4) as a senescence inducer with implications for the senescence-like state of postmitotic cells in the aging brain. 21436262_proteolytic cleavage by furin is a posttranslational requirement for proaugurin to suppress cell proliferation 22899245_a wound termination factor during wound resolution after cutaneous injury 29511297_These data suggest that multiple scavenger receptors recognize Ecrg4(71-132) and transduce its signals, together with Toll-like receptors, in microglia. 30745428_Ecrg4 has a role in aging of neural stem cells in a Foxg1-dependent manner 32195341_ECRG4 regulates neutrophil recruitment and CD44 expression during the inflammatory response to injury. |
ENSG00000119147 |
ECRG4 |
432.369515 |
4.7193093302 |
2.238576 |
0.13703657 |
263.430448 |
0.00000000000000000000000000000000000000000000000000000000003067459201284904359347387463910037203579381731789304355843238131908999469098139726034021715917031641097988700466009678600151797794115416648279081419589675583381449541775509715080261230468750000000000000000 |
0.0000000000000000000000000000000000000000000000000000000067715273266625601057572826669978838698673946196999343388958467987271665804882792729805893173030201550000719425956432909118820746881304030169272667460944603590178303420543670654296875000000000000000000000 |
Yes |
No |
737.512944338006 |
70.1591700893269 |
156.275609997701 |
11.9581850843588 |
| ENSMUSG00000026065 |
110895 |
Slc9a4 |
protein_coding |
Q8BUE1 |
FUNCTION: Involved in pH regulation to eliminate acids generated by active metabolism or to counter adverse environmental conditions. Major proton extruding system driven by the inward sodium ion chemical gradient. Plays an important role in signal transduction. May play a specialized role in the kidney in rectifying cell volume in response to extreme fluctuations of hyperosmolar-stimulated cell shrinkage. Is relatively amiloride and ethylisopropylamiloride (EIPA) insensitive. Can be activated under conditions of hyperosmolar-induced cell shrinkage in a sustained intracellular acidification-dependence manner. Activated by 4,4'-diisothiocyanostilbene-2,2'-disulfonic acid (DIDS) in a sustained intracellular acidification-dependence manner. Affects potassium/proton exchange as well as sodium/proton and lithium/proton exchange (By similarity). In basolateral cell membrane, participates in homeostatic control of intracellular pH, and may play a role in proton extrusion in order to achieve transepithelial HCO3(-) secretion. In apical cell membrane may be involved in mediating sodium absorption. Requires for normal levels of gastric acid secretion, secretory membrane development, parietal cell maturation and/or differentiation and at least secondarily for chief cell differentiation. {ECO:0000250, ECO:0000269|PubMed:12493726, ECO:0000269|PubMed:15684419}. |
Antiport;Cell membrane;Cytoplasmic vesicle;Glycoprotein;Ion transport;Membrane;Phosphoprotein;Reference proteome;Sodium;Sodium transport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:110895; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; cytoplasmic vesicle [GO:0031410]; plasma membrane [GO:0005886]; potassium:proton antiporter activity [GO:0015386]; sodium:proton antiporter activity [GO:0015385]; gastric acid secretion [GO:0001696]; glandular epithelial cell development [GO:0002068]; potassium ion transmembrane transport [GO:0071805]; regulation of intracellular pH [GO:0051453]; sodium ion import across plasma membrane [GO:0098719] |
12493726_the mRNAs of different NHE isoforms as well as their subcellular localization (apical versus basolateral) and functional activity were investigated in mouse endometrial epithelial cells 15684419_NHE4, which may normally be coupled with the AE2 Cl-/HCO3- exchanger, is important for normal levels of gastric acid secretion, gastric epithelial cell differentiation, and development of secretory canalicular and tubulovesicular membranes 18930060_Dra is the major Cl(-)/HCO(3)(-) exchanger coupled with Nhe3 for electroneutral NaCl absorption across mammalian small intestine. 22088435_Disruption of murine NHE4 leads to metabolic acidosis with inappropriate urinary ammonia excretion and decreases the ability of the thick ascending limb to absorb ammonia and to build the corticopapillary ammonia gradient. 32372285_SLC9A4 in the organum vasculosum of the lamina terminalis is a [Na(+)] sensor for the control of water intake. |
ENSG00000180251 |
SLC9A4 |
6.067449 |
9.2310253791 |
3.206491 |
1.23950564 |
6.700008 |
0.00964124699846938444658395184205801342613995075225830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0324542484598200342560403441893868148326873779296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.8546163672757 |
10.5162689905069 |
1.17588414304128 |
0.951693506122057 |
| ENSMUSG00000026170 |
104086 |
Cyp27a1 |
protein_coding |
Q9DBG1 |
FUNCTION: Cytochrome P450 monooxygenase that catalyzes regio- and stereospecific hydroxylation of cholesterol and its derivatives. Hydroxylates (with R stereochemistry) the terminal methyl group of cholesterol side-chain in a three step reaction to yield at first a C26 alcohol, then a C26 aldehyde and finally a C26 acid. Regulates cholesterol homeostasis by catalyzing the conversion of excess cholesterol to bile acids via both the 'neutral' (classic) and the 'acid' (alternative) pathways. May also regulate cholesterol homeostasis via generation of active oxysterols, which act as ligands for NR1H2 and NR1H3 nuclear receptors, modulating the transcription of genes involved in lipid metabolism (By similarity). Plays a role in cholestanol metabolism in the cerebellum (PubMed:28190002). Similarly to cholesterol, hydroxylates cholestanol and may facilitate sterol diffusion through the blood-brain barrier to the systemic circulation for further degradation. Also hydroxylates retinal 7-ketocholesterol, a noxious oxysterol with pro-inflammatory and pro-apoptotic effects, and may play a role in its elimination from the retinal pigment epithelium. May play a redundant role in vitamin D biosynthesis. Catalyzes 25-hydroxylation of vitamin D3 that is required for its conversion to a functionally active form (By similarity). {ECO:0000250|UniProtKB:Q02318, ECO:0000269|PubMed:28190002}. |
Acetylation;Cholesterol metabolism;Heme;Iron;Lipid biosynthesis;Lipid metabolism;Membrane;Metal-binding;Mitochondrion;Mitochondrion inner membrane;Monooxygenase;Oxidoreductase;Reference proteome;Steroid biosynthesis;Steroid metabolism;Sterol metabolism;Transit peptide |
PATHWAY: Hormone biosynthesis; cholecalciferol biosynthesis. {ECO:0000250|UniProtKB:Q02318}.; PATHWAY: Steroid metabolism; cholesterol degradation. {ECO:0000250|UniProtKB:Q02318}.; PATHWAY: Lipid metabolism; bile acid biosynthesis. {ECO:0000250|UniProtKB:Q02318}. |
|
mmu:104086; |
mitochondrial envelope [GO:0005740]; mitochondrial inner membrane [GO:0005743]; mitochondrion [GO:0005739]; 3-alpha,7-alpha,12-alpha-trihydroxycholestan-26-al 26-oxidoreductase activity [GO:0047103]; cholestanetetraol 26-dehydrogenase activity [GO:0047748]; cholestanetriol 26-monooxygenase activity [GO:0047749]; cholesterol 26-hydroxylase activity [GO:0031073]; cholesterol 7-alpha-monooxygenase activity [GO:0008123]; cholesterol monooxygenase (side-chain-cleaving) activity [GO:0008386]; heme binding [GO:0020037]; Hsp70 protein binding [GO:0030544]; iron ion binding [GO:0005506]; vitamin D3 25-hydroxylase activity [GO:0030343]; bile acid biosynthetic process [GO:0006699]; C21-steroid hormone biosynthetic process [GO:0006700]; calcitriol biosynthetic process from calciol [GO:0036378]; cholesterol catabolic process [GO:0006707]; cholesterol metabolic process [GO:0008203] |
12020639_mice do not have an absolute requirement for 27OHase in order to metabolise 7-ketocholesterol and must rely on alternative side-chain oxidising pathways 15520450_both cyp27A genotype and gender affected the regulation of hepatic bile acid, cholesterol, and fatty acid metabolism 15825828_Hepatic neutral cytosolic cholesteryl ester hydrolase decreased mRNAs for cholesterol 7alpha-hydroxylase, sterol 27-hydroxylase, and HMG-CoA reductase on day 7 after transfection, coinciding with peak hepatic free cholesterol concentrations 19671838_In CYP27A1 ko mice, urinary progesterone concentrations were decreased, 20alpha-DH-progesterone increased, and the progesterone-to-20alpha-DH-progesterone ratio decreased threefold 20501668_Increasing 27-hydroxycholesterol concentrations by genetic means alters bone mineral density. 20511491_Results are consistent with a concentration-dependent flux of 7alpha-hydroxy-4-cholesten-3-one across the blood-brain barrier in cyp27a1(-/-) mice and subsequent formation of cholestanol. 21334403_CYP27(-/-) mice exhibited enhanced CCl(4)-induce liver injury, and defective liver regeneration and prolonged steatosis after 70% partial hepatectomy. 22820291_Loss of CYP27A1 led to dysregulation of retinal cholesterol homeostasis, including unexpected upregulation of retinal cholesterol biosynthesis. Cyp27a1-/- mice developed retinal lesions with cholesterol deposition beneath the retinal pigment epithelium. 24096962_The expression of CYP27A1 modulates the concentrations of active glucocorticoids in both humans and mice and in vitro. 24280213_Data indicate that inhibition of CYP27A1 activity or knockdown and deletion of the Cyp27a1 gene induced adipocyte differentiation. 25281859_overexpression of Cyp27a1 in KCs reduces hepatic inflammation independently of 27HC levels in plasma and liver, further pointing towards KCs as specific target for improving the therapy of NASH. 25453744_The cholesterol diet reduced the levels of the 'memory protein' Arc (Activity Regulated Cytoskeleton associated protein) in the hippocampus of the wildtype mice but not in the hippocampus of the Cyp27-/- mice. 28190002_CYP46A1 is involved in cholestanol removal from the brain and that several factors contribute to the preferential increase in cholestanol in the cerebellum arising from CYP27A1 deficiency. 29191818_In het mice, RIF had no effect on plasma lipids composition, CYP27A1 activity, and atherosclerotic plaque development, despite a reduction in cholesterol absorption. In conclusion, the antiatherogenic effect of Cyp3a11 induction by RIF was also dependent on Cyp27a1 expression. 31048561_High expression of cytochrome P450 family 27 subfamily A member 1 (CYP27A1), the enzyme responsible for the synthesis of 27-Hydroxycholesterol (27HC), is associated with decreased progression free survival for ovarian cancer. ID8 tumor cell grafts are unable to effectively establish in CYP27A1-/- mice, indicating involvement of the host environment. Tumors from mice treated with 27HC had altered myeloid cell composition. 31655042_7KCh up-regulated IL-1betamRNA, TNF-alphamRNA, IL-6 mRNA, and IL-1beta secretion of RPE. U0126, a MEK1/2 inhibitor, down regulated the expression of these inflammation factors. Our findings may help elucidate the potential role of 7KCh in the pathogenesis of AMD. 33526687_Vertical sleeve gastrectomy confers metabolic improvements by reducing intestinal bile acids and lipid absorption in mice. |
ENSG00000135929 |
CYP27A1 |
32.810257 |
3.0394419992 |
1.603806 |
0.44367878 |
13.014107 |
0.00030915316771655928151582570784228209959110245108604431152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016356446858835119954195258529239254130516201257705688476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
48.4284790918488 |
15.1293146542142 |
15.9333453654537 |
3.92225710651528 |
| ENSMUSG00000026204 |
19275 |
Ptprn |
protein_coding |
A6MDD2 |
Human_homologues FUNCTION: Plays a role in vesicle-mediated secretory processes (PubMed:24843546). Required for normal accumulation of secretory vesicles in hippocampus, pituitary and pancreatic islets (By similarity). Required for the accumulation of normal levels of insulin-containing vesicles and preventing their degradation (PubMed:24843546). Plays a role in insulin secretion in response to glucose stimuli (PubMed:24843546). Required for normal accumulation of the neurotransmitters norepinephrine, dopamine and serotonin in the brain (By similarity). In females, but not in males, required for normal accumulation and secretion of pituitary hormones, such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH) (By similarity). Required to maintain normal levels of renin expression and renin release (By similarity). Seems to lack intrinsic enzyme activity (By similarity). May regulate catalytic active protein-tyrosine phosphatases such as PTPRA through dimerization (By similarity). {ECO:0000250|UniProtKB:Q60673, ECO:0000269|PubMed:24843546}.; FUNCTION: [ICA512-transmembrane fragment]: ICA512-TMF regulates dynamics and exocytosis of insulin secretory granules (SGs); binding of ICA512-TMF to SNTB2/beta-2-syntrophin is proposed to restrain SGs mobility and exocytosis by tethering them to the actin cytoskeleton depending on UTRN; the function is inhibited by cytoplasmic ICA512-CFF dimerizing with ICA512-TMF and displacing SNTB2. {ECO:0000269|PubMed:18824546, ECO:0000269|PubMed:20886068}.; FUNCTION: [ICA512-cleaved cytosolic fragment]: ICA512-CCF translocated to the nucleus promotes expression of insulin and other granule-related genes; the function implicates binding to and regulating activity of STAT5B probably by preventing its dephosphorylation and potentially by inducing its sumoylation by recruiting PIAS4 (PubMed:15596545, PubMed:16622421, PubMed:18178618). Enhances pancreatic beta-cell proliferation by converging with signaling by STAT5B and STAT3 (PubMed:15596545, PubMed:16622421, PubMed:18178618). ICA512-CCF located in the cytoplasm regulates dynamics and exocytosis of insulin secretory granules (SGs) by dimerizing with ICA512-TMF and displacing SNTB2 thus enhancing SGs mobility and exocytosis (PubMed:18824546, PubMed:20886068). {ECO:0000269|PubMed:15596545, ECO:0000269|PubMed:16622421, ECO:0000269|PubMed:18178618, ECO:0000269|PubMed:18824546, ECO:0000269|PubMed:20886068}. |
Cytoplasmic vesicle;Membrane;Phosphoprotein;Proteomics identification;Receptor;Reference proteome;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
Human_homologues hsa:5798; |
nucleus [GO:0005634]; secretory granule [GO:0030141]; synapse [GO:0045202]; transport vesicle membrane [GO:0030658]; spectrin binding [GO:0030507]; ubiquitin-like protein ligase binding [GO:0044389]; dense core granule maturation [GO:1990502]; insulin secretion [GO:0030073]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of type B pancreatic cell proliferation [GO:1904692]; response to reactive oxygen species [GO:0000302] |
11600216_IA-2 peptides have been identified that induce lymphoproliferative responses in DQ8 transgenic and NOD mice and stimulate production of Th1 and Th2 cytokines. 12031972_IA-2 is involved in glucose-stimulated insulin secretion 16306340_experiments show that the dense core vesicle proteins IA-2 and IA-2beta, alone or in combination, are involved in insulin secretion, but neither alone nor in combination are they required for the development of diabetes in NOD mice 16418280_IA-2beta, but not IA-2, is induced by ghrelin and inhibits glucose-stimulated insulin secretion 16622421_The cleaved cytosolic fragment of ICA512 enhances the transcription of secretory granule genes (including its own gene) by binding to tyrosine phosphorylated signal transducers and activators of transcription (STAT) 5 and preventing its dephosphorylation. 17611635_IA-2 beta undergoes at least three distinct proteolytic cleavages. 18310453_IA-2 and IA-2beta are dispensable for exocytosis of insulin granules, but are probably more important for cargo loading and/or stability of dense core vesicles 18824546_ICA512-CCF also dimerizes with intact ICA512 on granules, thereby displacing it from beta2-syntrophin. 19233214_Data show that the insulinoma associated protein-2 distribution in peripheral neurons appeared more intensely in neurites rather than in the cell bodies. 19361477_In mice IA-2 expressed exclusively in dense core secretory vesicles (DCV) of neuroendocrine cells of brain and impairment of conditioned learning. 19513530_IA-2-/- mice had higher sensitivity to STZ, suggesting a role of IA-2 not only in the secretion but also in the production of insulin. 24662847_Experiments showed that the knock-out of IA-2, but not IA-2beta, resulted in marked impairment of active avoidance learning; one copy of the gene was sufficient to maintain active avoidance learning 24692514_Ptprn is an activity dependent gene in mouse olfactory sensory neurons. [Ptprn] 25861885_The deletion of IA-2 and IA-2beta results in multiple pathophysiologic changes and represents a unique in vivo model for studying the effect of hormone and neurotransmitter reduction on known and still unrecognized targets. 26148972_Data show that miRNAs are involved in regulating the expression of the major type 1 diabetes (T1D) autoantigens IA-2, IA-2beta, and GAD65 enzyme. |
ENSG00000054356 |
PTPRN |
64.388599 |
2.6054390390 |
1.381526 |
0.31345582 |
19.312463 |
0.00001109795675367399980359300881715967079799156635999679565429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000815456233825192356020825079632174947619205340743064880371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
91.3124103576147 |
19.9960024770097 |
35.0468420067459 |
6.02285984219695 |
| ENSMUSG00000026259 |
53972 |
Ngef |
protein_coding |
Q8CHT1 |
FUNCTION: Acts as a guanine nucleotide exchange factor (GEF) which differentially activates the GTPases RHOA, RAC1 and CDC42. Plays a role in axon guidance regulating ephrin-induced growth cone collapse and dendritic spine morphogenesis. Upon activation by ephrin through EPHA4, the GEF activity switches toward RHOA resulting in its activation. Activated RHOA promotes cone retraction at the expense of RAC1- and CDC42-stimulated growth cone extension. {ECO:0000269|PubMed:11336673, ECO:0000269|PubMed:15848799, ECO:0000269|PubMed:17143272}. |
Alternative splicing;Cell projection;Cytoplasm;Developmental protein;Differentiation;Guanine-nucleotide releasing factor;Membrane;Neurogenesis;Phosphoprotein;Reference proteome;SH3 domain |
|
|
mmu:53972; |
cytosol [GO:0005829]; glutamatergic synapse [GO:0098978]; growth cone [GO:0030426]; membrane [GO:0016020]; postsynapse [GO:0098794]; ephrin receptor binding [GO:0046875]; guanyl-nucleotide exchange factor activity [GO:0005085]; activation of GTPase activity [GO:0090630]; cell differentiation [GO:0030154]; ephrin receptor signaling pathway [GO:0048013]; negative regulation of dendritic spine morphogenesis [GO:0061002]; nervous system development [GO:0007399]; regulation of GTPase activity [GO:0043087]; regulation of synapse pruning [GO:1905806] |
17702745_FGFR-mediated phosphorylation of ephexin1 enhances the guanine nucleotide exchange activity toward RhoA without affecting the activity to Rac1 or Cdc42. 20152127_An essential role is identified for rho guanine nucleotide exchange factor (RhoGEF) ephexin1 in the maturation of acetylcholine receptor clusters. 28769056_These findings suggest that increased EphA4-ephexin1 signaling in the PFC plays a role in the pathophysiology of depression. 29363583_We found ephexin1 essential for the development of limb nerves toward their muscle targets, concluding that it functions as an intermediary to relay Eph signaling in this context |
ENSG00000066248 |
NGEF |
164.374056 |
0.3469513213 |
-1.527195 |
0.36898659 |
15.421434 |
0.00008600722135419698068364152687692580911971163004636764526367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005196596571468180877223508318252243043389171361923217773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
86.8241426248965 |
22.4492120845241 |
250.248773529007 |
47.0352735162956 |
| ENSMUSG00000026360 |
19735 |
Rgs2 |
protein_coding |
O08849 |
FUNCTION: Regulates G protein-coupled receptor signaling cascades. Inhibits signal transduction by increasing the GTPase activity of G protein alpha subunits, thereby driving them into their inactive GDP-bound form (By similarity). It is involved in the negative regulation of the angiotensin-activated signaling pathway (By similarity). Plays a role in the regulation of blood pressure in response to signaling via G protein-coupled receptors and GNAQ. Plays a role in regulating the constriction and relaxation of vascular smooth muscle (PubMed:14608379). Binds EIF2B5 and blocks its activity, thereby inhibiting the translation of mRNA into protein (By similarity). {ECO:0000250|UniProtKB:P41220, ECO:0000269|PubMed:14608379}. |
Cell cycle;Cell membrane;Cytoplasm;GTPase activation;Membrane;Nucleus;Phosphoprotein;Reference proteome;Signal transduction inhibitor;Translation regulation |
|
|
mmu:19735; |
cytoplasm [GO:0005737]; cytoplasmic side of plasma membrane [GO:0009898]; cytosol [GO:0005829]; neuron projection [GO:0043005]; nucleolus [GO:0005730]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; adenylate cyclase inhibitor activity [GO:0010855]; beta-tubulin binding [GO:0048487]; G-protein alpha-subunit binding [GO:0001965]; GTPase activator activity [GO:0005096]; brain development [GO:0007420]; brown fat cell differentiation [GO:0050873]; cell cycle [GO:0007049]; G protein-coupled receptor signaling pathway [GO:0007186]; maternal process involved in female pregnancy [GO:0060135]; negative regulation of adenylate cyclase-inhibiting adrenergic receptor signaling pathway involved in heart process [GO:0140194]; negative regulation of cardiac muscle hypertrophy [GO:0010614]; negative regulation of cell growth involved in cardiac muscle cell development [GO:0061052]; negative regulation of G protein-coupled receptor signaling pathway [GO:0045744]; negative regulation of glycine import across plasma membrane [GO:1900924]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of phospholipase activity [GO:0010519]; negative regulation of translation [GO:0017148]; ovulation [GO:0030728]; positive regulation of cardiac muscle contraction [GO:0060452]; positive regulation of neuron projection development [GO:0010976]; regulation of G protein-coupled receptor signaling pathway [GO:0008277]; relaxation of cardiac muscle [GO:0055119]; relaxation of vascular associated smooth muscle [GO:0060087]; response to amphetamine [GO:0001975]; response to ethanol [GO:0045471]; spermatogenesis [GO:0007283] |
11968023_Analysis of regulator of G-protein signaling-2 (RGS-2) expression and function in osteoblastic cells. 11996904_Parathyroid hormone induces RGS-2 expression by a cyclic adenosine 3',5'-monophosphate-mediated pathway in primary neonatal murine osteoblasts. 12588882_Both rgs2+/- and rgs2-/- mice exhibited a strong hypertensive phenotype, renovascular abnormalities, persistent constriction of the resistance vasculature, and prolonged response of the vasculature to vasoconstrictors in vivo. 14608379_Rgs2-/- mice develop marked hypertension, and their blood vessels show enhanced contraction and decreased cGMP-mediated relaxation. 14976183_RGS2 binds directly to the third intracellular (i3) loop of the G(q/11)-coupled M1 muscarinic cholinergic receptor (M1 mAChR; M1i3). 15292238_cells adapted to deletion of RGS2 by reducing Ca(2+) signaling excitability, achieved by adaptation of all transporters to reduce Ca(2+) influx into the cytosol; thus, IP(3) receptor 1 was down-regulated and IP(3) receptor 3 was up-regulated 15489855_Genetic dissection of a behavioral quantitative trait locus shows that Rgs2 modulates anxiety in mice. 15563583_Nitric oxide donor sodium nitroprusside can reduce blood pressure in RGS2 deficient mice. 15661635_Rgs2 was expressed in undifferentiated pituitary cells, was downregulated at completion of hormone cell differentiation. Pituitary cell differentiation and morphology are normal in Rgs2 mutant mice. 15661972_The increase in blood pressure in RGS2-/-mice is not solely explained by peripheral vascular mechanisms; a central nervous system mechanism might be implicated by an increased sympathetic tone. 16135533_Overexpression of Rgs2 in the chondrogenic cell line ATDC5 resulted in accelerated hypertrophic differentiation, thus providing functional validation of microarray data 16269576_Our data suggest an interaction between RGS2 and the NO-cGMP pathway. The blunted l-NAME response in RGS2(-/-) during the day suggests impaired NO signaling. 16380388_Results suggest a potential pathophysiological role of loss of fine tuning due to selective RGS2 down-regulation in G(q/11)-mediated remodeling in hypertrophic cardiomyocytes. 17475820_RGS2 is involved in negative feedback regulation of V2R signaling 17507294_We found a mismatch between sympathetic nerve traffic and plasma norepinephrine concentrations. This may depend on altered coupling between electrical nerve activity and norepinephrine release or a changed norepinephrine uptake in RGS2-/- mice. 17613534_group VIA phospholipase A2 (iPLA2beta) plays a pivotal role in Ang II-induced RGS2 mRNA up-regulation in vascular smooth muscle cells 17644703_RGS2 deletion promotes Ang II-dependent hypertension primarily through an increase of myogenic tone and vasoreactivity, probably by sensitization of AT(1) receptors. 17681944_Regulation of Rgs2 and second messenger signaling in vascular smooth muscle cells by cGMP-dependent protein kinase. 18067675_Rgs2 was more highly expressed in mouse myoepithelial/basal mammary cells than luminal cells. 18625716_Affymetrix expression profiling shows that the C terminus of Runx2 regulates genes involved in G protein-coupled receptor signaling Rgs2, Rgs4, Rgs5, Rgs16, Gpr23, Gpr30, Gpr54, Gpr64, and Gna13. 18726905_RGS2 transcription is immediately induced via a cAMP-dependent pathway after initiation of 3T3-L1 differentiation and the RGS2 mRNA level remains consistently high throughout the differentiation progression. 19127022_Regulator of G protein signaling 2 mediates cardiac compensation to pressure overload. 19175184_RGS2 expression was upregulated by LH receptor and FP receptor activation and modulation of partner receptor signaling by RGS2 may require RGS2 translocation from the nucleus to the plasma membrane. 19262744_deficiency of RGS2 decreased intraocular pressure, presumably due to increased aqueous humor drainage in association with increased ciliary muscle contraction. 19294649_data show that RGS2 deficient mice in vivo differ from wild-type mice in both short-term and long-term synaptic plasticity suggesting that RGS2 serves as a negative regulator of long-term synaptic plasticity 19736320_Results identify a novel role for RGS2 in the control of protein synthesis that is independent of its established RGS domain function. 19966055_RGS2 is an important cholinergic regulator in the atrium. RGS2(-/-) mice have enhanced susceptibility to atrial fibrillation via enhanced M3 muscarinic receptor activity. 20032508_Ischemic stress increases RGS2 expression and that this condition contributes to enhanced apoptosis in C6 cells and primary astrocytes. 20352235_Ang II suppression by ANP was abolished in cardiomyocytes of mice deficient in GC-A, in cyclic GMP-dependent protein kinase I (PKG I) or in the regulator of G protein signalling (RGS) 2, a target of PKG I 21291891_These findings not only define RGS2 as a novel negative regulator of the beta(2)AR-G(i) signaling but also provide a potential novel target for the treatment of chronic heart failure. 21447383_RGS2(-/-) pre-adipocytes had reduced levels of differentiation markers (PPAR gamma; lipoprotein lipase; CCAAT/enhancer binding protein alpha) and also rgs2(-/-) white adipocytes were small relative to controls, suggesting altered adipogenesis. 21494556_Rgs2 is a critical regulator of the pro-angiogenic function of myeloid derived suppressor cells in the tumor microenvironment, through regulating MCP-1 production. 21510939_Data show that reducing RGS2 expression by antisense treatment prevented the increase in GLO1 and GSR1 mRNA and protein expression. 22040681_Our findings demonstrate a relationship between Rgs2 gene expression level and a propensity for anxious and depressive-like behaviour and reduced social interaction 22057271_Identification of a cAMP-response element in the regulator of G-protein signaling-2 (RGS2) promoter as a key cis-regulatory element for RGS2 22080612_Data show that Rgs2-deficient mice revealed enhanced bronchoconstriction to spasmogens and an absence of beta(2)-adrenoceptor agonist (LABA)-induced bronchoprotection. 22120521_RGS2 negatively regulates TLR2-mediated Nox1 expression via PKC-eta and PLD2 pathway. 22158566_These results indicate that Brucella abortus infection induces host RGS2 expression and that up-regulation of [Ca(2+)](i) levels is an essential factor for the intracellular survival of the bacteria within phagocytes. 22354966_systemic or endothelium-specific RGS2 deficiency causes endothelial dysfunction resulting in impaired EDHF-dependent vasodilatation. 22704538_RGS2 knockout caused spontaneous AHR in mice 22802950_Cardiomyocyte-specific RGS2 overexpression in transgenic mice in vivo did not lead to attenuate ventricular G(q/11)-mediated signaling and hypertrophy in response to pressure overload 22922103_This work provides greater insight into the stress proteome and the role of RGS2. 24105430_These findins indicate that urotensin II-induced calcium metabolism results in glomerular mesangial cell contraction and suggest that urotensin receptor activation recruts RGS2 to the plasma membrane. 24524188_Knockdown of endogenous Rgs2 expression led to abnormal embryonic development in vitro, with a considerable number of early embryos arrested at the 2- or 4-cell stage. Moreover, mRNA expression of three zygotic gene activation-related genes (i.e. Zscan4, Tcstv1 and MuERV-L) was decreased significantly in 2-cell arrested embryos. These results suggest that Rgs2 plays a critical role in early embryo development. 24846582_RGS2-deficient VSMC exhibit higher rates of proliferation and coordinate plasticity of Ca(2+)-handling mechanisms in response to PDGF stimulation. 24973550_Regulator of G protein signaling 2 deficiency accelerates the progression of kidney fibrosis 25289860_RGS2 and RGS4 are new interacting partners that play key roles in G protein coupling to negatively regulate kappa-OmicronR signaling. 25368964_genetic variations and increased inflammatory cytokines can lead to RGS2 repression, which exacerbates AHR and airway remodeling in asthma. 25909889_Rgs12 has a crucial role in the function of bone-resorbing OCs, particularly in preclinical models of pathological bone loss associated with inflammation-induced bone loss and postmenopausal osteoporosis. 25970626_FBXO44-mediated degradation of RGS2 protein uniquely depends on a Cul4B/DDB1 complex. 26160904_Regulator of G-protein signaling 2 (RGS2) suppresses premature calcium release in mouse eggs. 26193676_RGS2 deficiency impairs renal function and autoregulation by increasing renal vascular resistance and reducing renal blood flow. 26811058_RGS2 deficiency decreases uterine artery blood flow by increasing myogenic tone at least partly through prolonged G protein activation 27463806_RGS2 might play a critical role in mouse oocyte meiotic maturation by affecting beta-tubulin polymerization and chromosome segregation. 27528587_Results show that RGS2 negatively modulates D2R-mediated Galphai/o protein signaling in neuroblastoma N2A cells. Moreover, RGS2 critically regulates agonist-stimulated dopamine D2R internalization. RGS2 knockdown abolished quinpirole-stimulated D2R internalization and impaired beta-arrestin dissociation from the membrane. 27549302_Upregulated RGS2 contributes significantly to the anti-fibrotic effects of pirfenidone in idiopathic pulmonary fibrosis. 28107494_Modest anti-inflammatory and anti-remodelling roles for RGS2 are also suggested. If translatable to humans, therapies that maximize RGS2 expression may prove advantageous 28542139_RGS2 has critical roles in maintaining pancreatic beta-cell mass via modulating beta-cell function and apoptosis. 28641980_The present results suggest that expression of the eIF2B-interacting domain of RGS2, RGS2eb, can protect against pathological cardiac hypertrophy and dysfunction in vivo. 29649180_Results confirmed the regulator of G-Protein signaling 2 (RGS2) as an interaction partner of PKGI-alpha. 29932914_PKC activation appeared to increase the RGS2 translocation from nucleus to cytosol. PKA- and PKC-mediated RGS2 transcription in a Sp-1-dependent manner and a PKC-mediated RGS2 intracellular translocation were noted in granulosa cells. 30412338_Uterine RGS2 expression is regulated by exogenous estrogen and progesterone in ovariectomized mice, and downregulation of RGS2 expression in artificial decidualized ESCs inhibits trophoblast spreading in vitro. 30826455_The present results thus point to a novel function of RGS2 in the stress response directly related to its ability to reduce global protein synthesis. 31030617_Regulator of G Protein Signaling 2 Facilitates Uterine Artery Adaptation During Pregnancy in Mice. 31633064_Overexpression of RGS2 in explicitly serotonergic neurons augments male aggression in control mice and rescues male aggression in Rgs2 (-/-) mice, while anxiety is not affected. Findings specifically identify that RGS2 expression in serotonergic neurons is sufficient to drive male aggression in mice and as a potential therapeutic target for treating aggression. 32679115_Silencing of RGS2 enhances hippocampal neuron regeneration and rescues depression-like behavioral impairments through activation of cAMP pathway. 35661621_Dual loss of regulator of G protein signaling 2 and 5 exacerbates ventricular myocyte arrhythmias and disrupts the fine-tuning of Gi/o signaling. 36075764_Comparative Transcriptional Analyses in the Nucleus Accumbens Identifies RGS2 as a Key Mediator of Depression-Related Behavior. 36259376_Cardiometabolic Consequences of Deleting the Regulator of G protein Signaling-2 (Rgs2) From Cells Expressing Agouti-Related Peptide or the ANG (Angiotensin) II Type 1A Receptor in Mice. |
ENSG00000116741 |
RGS2 |
138.038634 |
0.4391810674 |
-1.187112 |
0.24692390 |
22.882367 |
0.00000172224548976496408501663925444402636344420898240059614181518554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000146964948459943604180174409057357820529432501643896102905273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
84.7142358939925 |
14.5701249045503 |
192.891365728557 |
23.7551063844524 |
| ENSMUSG00000026365 |
12628 |
Cfh |
protein_coding |
P06909 |
FUNCTION: Glycoprotein that plays an essential role in maintaining a well-balanced immune response by modulating complement activation. Acts as a soluble inhibitor of complement, where its binding to self markers such as glycan structures prevents complement activation and amplification on cell surfaces. Accelerates the decay of the complement alternative pathway (AP) C3 convertase C3bBb, thus preventing local formation of more C3b, the central player of the complement amplification loop. As a cofactor of the serine protease factor I, CFH also regulates proteolytic degradation of already-deposited C3b. In addition, mediates several cellular responses through interaction with specific receptors. For example, interacts with CR3/ITGAM receptor and thereby mediates the adhesion of human neutrophils to different pathogens. In turn, these pathogens are phagocytosed and destroyed. {ECO:0000250|UniProtKB:P08603}. |
3D-structure;Complement alternate pathway;Disulfide bond;Glycoprotein;Immunity;Innate immunity;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal;Sulfation;Sushi |
|
|
mmu:12628; |
axon [GO:0030424]; cytoplasm [GO:0005737]; external side of plasma membrane [GO:0009897]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; mitochondrion [GO:0005739]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; serine-type endopeptidase complex [GO:1905370]; complement component C3b binding [GO:0001851]; heparan sulfate proteoglycan binding [GO:0043395]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; activation of membrane attack complex [GO:0001905]; angiogenesis [GO:0001525]; ATP metabolic process [GO:0046034]; complement activation [GO:0006956]; complement activation, alternative pathway [GO:0006957]; determination of adult lifespan [GO:0008340]; gene expression [GO:0010467]; glomerulus development [GO:0032835]; immune response [GO:0006955]; inflammatory response [GO:0006954]; kidney development [GO:0001822]; mitochondrial DNA metabolic process [GO:0032042]; mitochondrial gene expression [GO:0140053]; mitochondrion organization [GO:0007005]; monocyte aggregation [GO:0070487]; neuromuscular process [GO:0050905]; organelle localization [GO:0051640]; photoreceptor cell differentiation [GO:0046530]; platelet aggregation [GO:0070527]; proteolysis [GO:0006508]; regulation of complement activation [GO:0030449]; regulation of complement activation, alternative pathway [GO:0030451]; regulation of complement-dependent cytotoxicity [GO:1903659]; response to cytokine [GO:0034097]; response to dietary excess [GO:0002021]; retina development in camera-type eye [GO:0060041]; retinal pigment epithelium development [GO:0003406]; retinal rod cell development [GO:0046548]; vascular associated smooth muscle cell differentiation [GO:0035886]; visual perception [GO:0007601] |
15574507_role for platelet-associated factor H to process immune complexes and limit their accumulation in glomeruli 16023208_mFH and mFH1-5 do act as cofactors for factor I-mediated cleavage of human C3b. 16597679_These data show that plasma Cfh limits complement activation in the circulation and other accessible sites such as the glomerulus, whereas platelet Cfh is responsible for immune complex processing. 16769899_important role for C5 in both spontaneous membranoproliferative glomerulonephritis (MPGN) and experimentally induced nephritis in factor H-deficient mice and provide preliminary evidence that C5 inhibition therapy might be useful in human MPGN type II 16849499_Alternative complement pathway activation during laser-induced choroidal neovascularization in complement-sufficient C57BL/6 mice is due to increased production of key component factor B as well as reduced expression of regulatory protein factor H. 16936129_A significant level of CFH expression is maintained in different ocular tissues during development and aging. 17562771_Mice intranasally infected with Streptococcus pneumoniae preincubated with Complement Factor H (FH) had increased bacteremia and lung invasion, but they had similar levels of nasopharyngeal colonization compared to mice challenged without FH. 17921253_CFH is critically required for the long-term functional health of the retina. 19411110_C3 along the glomerular basement membrane in Cfh(-/-) mice is the C3 fragment iC3b and this is derived from plasma C3 activation. 19620346_The data presented here provide significant new insight into the interaction between CspZ (BBH06)and Factor H and suggest that there is a correlation between CspZ production and dissemination. 19828624_Complement receptor 2 with factor H specifically targets and inhibits the alternative pathway of complement in vitro and is effective in collagen antibody-induced arthritis in vivo. 20538999_Chimeric Cfh proteins led to (AMD)age-related macular degeneration-like characteristics in mice. This may represent a good model for studying the role of complement and other components of the immune system in early AMD. 20675597_Complement factor H in the serum contributes to complement regulation on the apical surface of renal tubular epithelial cells. 20702729_In the absence of plasma CFH, the accumulated activated complement leads to spontaneous and chronic serum sickness-induced proliferative glomerulonephritis. 20813971_Loss of complement factor H is associated with choroidal neovascularization. 20854881_Deletion of adrenomedullin gene, but not of complement factor H, increases infarct volume after permanent focal cerebral ischemia in mice. 20962256_There is a significant reduction in the numbers of both neutrophils and macrophages in cardiac isografts isolated from mice treated with either complement receptor 2-Crry or CR2-factor H. 21148254_Complement factor H deficiency accelerates development of lupus nephritis. 21544060_though factor H doesn't fully prevent activation of the complement alternative pathway on ischemic tubules, its interaction with the tubule epithelial cell surface is critical for limiting complement activation and attenuating renal injury after ischemia. 21920606_Complement factor H (CFH) mRNA is produced in CNS-specific cells, and in astrocytes, the CFH promoter is greatly influenced by the c-Jun and c-Fos transcription factors 21979047_complement factor H (CFH) is a major malondialdehyde (MDA)-binding protein that can block both the uptake of MDA-modified proteins by macrophages and MDA-induced proinflammatory effects in vivo in mice 22174452_Although endogenous factor H (fH) limits the development of allergen-induced airway hyperresponsiveness and airway inflammation, the protection afforded by fH is incomplete. 22819137_A2E accumulation altered retinal microglial complement regulation by decreasing complement factor H (and increasing complement factor B expression), favoring increased complement activation and lipofuscin deposition in the outer retina. 22832515_The exaggerated humoral immune response in CFH(-/-) mice was normalized in CFH(-/-)C5aR(-/-) double knockout mice, highlighting the C5aR dependence. 22875704_CFH influences age-related macular degeneration risk by modulating oxidative stress, inflammation, and abnormal angiogenesis 22918646_While the connection between CFH deficiency and failure to upregulate CD59a remains unknown, these results suggest that expression of CD59 is tissue-specific and that neuroretinal regulation depends on CFH. 23029250_IL-27 regulates complement activation through up-regulation of complement factor H in the retina. 23184055_Properdin deficiency exacerbated renal injury in mice lacking complement factor H. 23204401_nonsense mutations in SCR19 caused glomerulonephritis 23436934_endogenous fH makes a significant contribution to inhibition of the AP in CAIA through binding to sites of immune complex formation and complement activation. 24279761_Data indicate that co-deficiency of factor H (FH) and MASP-1/MASP-3 did not ameliorate either the plasma Complement C3 (C3) activation or glomerular C3 accumulation in FH-deficient mice. 24664701_A spectrum of complement dysregulation was modeled on the APOE4 age related macular degeneration mouse model by crossing these mice to complement factor H knockout (cfh-/-) mice to test the impact of excess complement activation. 25565310_absence of plasma CfH conferred susceptibility to glomerulonephritis 25684976_Cfh and Cfhr2 genes are expressed in the mouse outer retina. Only Cfh mRNA was detected in the retinal pigment epithelium, but no protein. 25991857_This new understanding of the complicated interactions of CFH in AMD-like pathology provides an improved foundation for the development of targeted therapies for AMD 26030099_data suggest that altered interactions of Cfh with MDA-modified proteins may be relevant in explaining the effects of the Cfh variant. 26802141_role for serum FH levels in the host response to invasive pneumococcal infections 27165610_Factor H and Crry are critical for regulating complement activation at distinct anatomic sites within the kidney. 27397653_environmental factors can drive retinal disease in these mice when linked to complement deficits impairing immune function. Both groups of mice had similar levels of retinal amyloid beta accumulation. Consequently there is no direct link between this and inflammation in Cfh(-/-) mice. 27918307_VEGF inhibition decreases local CFH and other complement regulators in the eye and kidney through reduced VEGFR2/PKC-alpha/CREB signaling. 29070671_this study shows that complement regulatory protein Factor H is a soluble prion receptor that potentiates peripheral prion pathogenesis 29389016_The results demonstrate that factor H is important for limiting injury in the kidney after acute kidney ischemia/reperfusion injury, but it is not critical for controlling complement activation by immunoglobulin within the glomerulus in this setting. 30107932_our studies demonstrate a direct role of FH in the maintenance of bone structure and function and is highlighted as a promising therapeutic target in bone diseases. 30642982_the -1635 AP-1 motif on the CFH promoter region mediates Cd-inducible CFH gene expression 30711487_Complement Factor H Mutation W1206R Causes Retinal Thrombosis and Ischemic Retinopathy. 30808757_Hence, we demonstrate a functional consequence of the Y402H polymorphism in vivo, which promotes age-related macular degeneration (AMD)-like pathology development and affects lipoprotein levels in aged mice. These findings support targeting lipoproteins as a viable therapeutic strategy for treating AMD. 30972735_the expression profile of complement factor H (CFH) and miR-146a genes in experimental autoimmune encephalomyelitis (EAE) mouse model, were examined. 31133737_Possible role of complement factor H in podocytes in clearing glomerular subendothelial immune complex deposits. 32165615_an African trypanosome receptor for mammalian factor H, a negative regulator of the alternative pathway, is identified.[Cfh] 32369457_Complement factor H-deficient mice develop spontaneous hepatic tumors. 32849636_Circulating FH Protects Kidneys From Tubular Injury During Systemic Hemolysis. 32962822_Absence of complement factor H reduces physical performance in C57BL6 mice. 33410734_Changes in complement alternative pathway components, factor B and factor H during dengue virus infection in the AG129 mouse. 33708358_Tumour extracellular vesicle-derived Complement Factor H promotes tumorigenesis and metastasis by inhibiting complement-dependent cytotoxicity of tumour cells. 34139209_Local complement factor H protects kidney endothelial cell structure and function. 34775843_Complement factor H deficiency combined with smoking promotes retinal degeneration in a novel mouse model. |
|
|
8620.980595 |
0.4183447278 |
-1.257236 |
0.06243327 |
395.845118 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000044198943558126725243217170433471030932099891915223543364724956994915129484064620206368112402626959571126996701675109916519404922976060577472050011365608297456409952094741141635 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000002171736478314149510317079150098553354805236031174131223559787035737793833768634002124605290202635358101428190428530876548858857935715536433627696327688773786685001381583378592 |
Yes |
No |
5002.58485176333 |
216.556677879622 |
11958.0444542641 |
383.774776197683 |
| ENSMUSG00000026413 |
18772 |
Pkp1 |
protein_coding |
P97350 |
FUNCTION: Seems to play a role in junctional plaques (By similarity). May facilitate the formation of intermediate filaments (By similarity). {ECO:0000250|UniProtKB:Q13835}. |
Cell adhesion;Nucleus;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:18772; |
adherens junction [GO:0005912]; cornified envelope [GO:0001533]; cytoplasm [GO:0005737]; desmosome [GO:0030057]; messenger ribonucleoprotein complex [GO:1990124]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; cadherin binding [GO:0045296]; lamin binding [GO:0005521]; cell-cell adhesion [GO:0098609]; cell-cell junction assembly [GO:0007043]; intermediate filament bundle assembly [GO:0045110]; negative regulation of mRNA catabolic process [GO:1902373]; positive regulation of gene expression [GO:0010628] |
27033150_PKP1 has roles in desmosome and tight junction function and in growth control, a function that is crucial in wound healing and epidermal carcinogenesis 28507225_This approach identified a key signaling event, phosphorylation of a desmosome component, PKP1 (plakophilin-1) by RIPK4 (receptor-interacting serine-threonine kinase 4) during epidermal differentiation. 30841437_PKP1 was identified as a new potential tumor suppressor in human melanoma. 30949721_Pkp1 regulates desmoglein membrane availability, whereas Pkp1 but not Pkp3 is required for desmoglein 3 clustering. |
ENSG00000081277 |
PKP1 |
30.891972 |
5.9003300154 |
2.560796 |
0.55586150 |
20.690311 |
0.00000539884839066349644178021208174023115589079679921269416809082031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000419996213925364546238824803303657517972169443964958190917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
56.078836609162 |
22.1454058397328 |
9.50435593653232 |
2.97066469804792 |
| ENSMUSG00000026489 |
67426 |
Coq8a |
protein_coding |
Q60936 |
FUNCTION: Atypical kinase involved in the biosynthesis of coenzyme Q, also named ubiquinone, an essential lipid-soluble electron transporter for aerobic cellular respiration (PubMed:27499294). Its substrate specificity is unclear: does not show any protein kinase activity (PubMed:27499294). Probably acts as a small molecule kinase, possibly a lipid kinase that phosphorylates a prenyl lipid in the ubiquinone biosynthesis pathway, as suggested by its ability to bind coenzyme Q lipid intermediates (By similarity). Shows an unusual selectivity for binding ADP over ATP (By similarity). {ECO:0000250|UniProtKB:Q8NI60, ECO:0000269|PubMed:27499294}. |
Alternative splicing;ATP-binding;Kinase;Membrane;Mitochondrion;Nucleotide-binding;Reference proteome;Transferase;Transit peptide;Transmembrane;Transmembrane helix;Ubiquinone biosynthesis |
PATHWAY: Cofactor biosynthesis; ubiquinone biosynthesis. {ECO:0000250|UniProtKB:Q8NI60}. |
|
mmu:67426; |
extrinsic component of mitochondrial inner membrane [GO:0031314]; mitochondrion [GO:0005739]; ADP binding [GO:0043531]; ATP binding [GO:0005524]; kinase activity [GO:0016301]; phosphorylation [GO:0016310]; ubiquinone biosynthetic process [GO:0006744] |
27499294_Loss of COQ8a results in cerebellar ataxia and coenzyme Q deficiency. |
ENSG00000163050 |
COQ8A |
144.281605 |
0.3666176516 |
-1.447652 |
0.45807494 |
9.159295 |
0.00247457887425143151227668880665078177116811275482177734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0102120794940660543359545187058756710030138492584228515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
80.0781551025394 |
25.855259612048 |
218.424166821485 |
52.3030651111582 |
| ENSMUSG00000026532 |
20739 |
Spta1 |
protein_coding |
P08032 |
FUNCTION: Spectrin is the major constituent of the cytoskeletal network underlying the erythrocyte plasma membrane. It associates with band 4.1 and actin to form the cytoskeletal superstructure of the erythrocyte plasma membrane. |
Actin capping;Actin-binding;Calcium;Cell shape;Cytoplasm;Cytoskeleton;Metal-binding;Phosphoprotein;Reference proteome;Repeat;SH3 domain |
|
|
mmu:20739; |
axon [GO:0030424]; cell junction [GO:0030054]; cell projection [GO:0042995]; cortical actin cytoskeleton [GO:0030864]; cortical cytoskeleton [GO:0030863]; cuticular plate [GO:0032437]; cytoplasmic side of plasma membrane [GO:0009898]; membrane [GO:0016020]; plasma membrane [GO:0005886]; spectrin [GO:0008091]; spectrin-associated cytoskeleton [GO:0014731]; actin filament binding [GO:0051015]; calcium ion binding [GO:0005509]; actin cytoskeleton organization [GO:0030036]; actin filament capping [GO:0051693]; hemopoiesis [GO:0030097]; lymphocyte homeostasis [GO:0002260]; plasma membrane organization [GO:0007009]; porphyrin-containing compound biosynthetic process [GO:0006779]; positive regulation of protein binding [GO:0032092]; positive regulation of T cell proliferation [GO:0042102]; regulation of cell shape [GO:0008360] |
11920196_Murine recessive hereditary spherocytosis, sph/sph, is caused by a mutation in the erythroid alpha-spectrin gene. 20056793_Analysis of 3 new alleles reveals previously unknown consequences of red blood cell (RBC) spectrin deficiency, resulting in markedly reduced RBC membrane spectrin content, decreased band 3, and absent beta-adducin. 21566095_Results of this study provide evidence for a role for spectrin in different steps of megakaryocyte development through its participation in the formation of invaginated membranes and in the maintenance of proplatelet structure. 22163289_organization of a spectrin-like cytoskeleton is associated with keratinocyte differentiation, and cytoskeleton disruption is mediated by either PKCdelta(Thr505) phosphorylation associated with phosphorylated adducin or due to reduction of endogenous adducin 25305142_Calcium plays a role in regulating the expression and function of beta-adducin to sustain normal organization of the spectrin-based cytoskeleton and the differentiation properties in keratinocytes through the calmodulin/EGFR/cadherin signaling pathway. 25830635_a new function for spectrins in the stability of invadosomes and the coupling between actin regulation and ECM degradation 28446510_This study demonstrates that the FGF19-SHP-LSD1 axis maintains homeostasis by suppressing unnecessary autophagic breakdown of cellular components, including lipids, under nutrient-rich postprandial conditions. 29720258_These findings reveal spectrin (alpha/beta) / ankyrin B cytoskeletal and signaling proteins as key regulators of T-type calcium channels expressed in the nervous system. |
ENSG00000163554 |
SPTA1 |
9.541855 |
10.5307900227 |
3.396542 |
1.11485299 |
9.271146 |
0.00232791951919443951962707117786521848756819963455200195312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0096961635538336619216659428843740897718816995620727539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.3808687608296 |
15.6604192111866 |
1.74544062898133 |
1.22441964298847 |
| ENSMUSG00000026535 |
26388 |
Ifi202b |
protein_coding |
Q9R002 |
FUNCTION: Inhibits the transcriptional activity of several transcription factors, including NF-kappa-B p50 and p65, FOS, JUN, E2F1, E2F4, MYOD1 and myogenin. Has anti-apoptotic effects due to inhibition of the transcriptional activity of p53. Binds dsDNA in the cytosol. Is involved in innate immune response and has anti-inflammatory activity. Inhibits caspase activation in response to cytosolic DNA and inhibits the activation of the AIM2 inflammasome, probably by sequestering cytoplasmic DNA and preventing its being bound by AIM2. {ECO:0000269|PubMed:16670293, ECO:0000269|PubMed:19131592, ECO:0000269|PubMed:23567559}. |
3D-structure;Cytoplasm;DNA-binding;Immunity;Inflammatory response;Innate immunity;Nucleus;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:26388; |
cytoplasm [GO:0005737]; nucleolus [GO:0005730]; double-stranded DNA binding [GO:0003690]; activation of innate immune response [GO:0002218]; cellular response to interferon-beta [GO:0035458]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043154]; negative regulation of innate immune response [GO:0045824]; positive regulation of interleukin-1 beta production [GO:0032731]; response to bacterium [GO:0009617] |
12676938_p202a can impair NF-kappaB activity both by inhibiting the binding to DNA of the transcriptionally active p65 homodimers and p50/p65 heterodimers and by boosting the binding of the repressive p50 homodimers 12894219_Expression stimulation by Rb was dependent, in part, on a JunD/AP-1 DNA-binding site present in the 5'-regulatory region of the Ifi202 gene. transcriptional activation of Ifi202 gene by Rb/JunD may be important for cell growth and survival. 14764608_increased levels of IL-6 in lupus contribute to up-regulation of p202 16670293_Increased Ifi202 expression in B6.Nba2 mice increases their susceptibility to develop lupus, in part, by inhibiting p53-mediated apoptosis. 17702989_Ifi202 is an important NF-kappaB activator in dendritic cells and is involved in IL-12 production, which may account for a T(h)1-prone phenotype of BWF1 mice 18037609_The candidate lupus susceptibility gene Ifi202a is largely dispensable for B-cell function. 18370867_p202 plays a role in regulating the proliferative capacity of hematopoietic cells. 18374989_demonstrate that stimulation of T cells up-regulates the Ifi202 expression in part through the JNK/c-Jun pathway 18424712_increased Ifi202 expression in certain strains of mice contributes to lupus susceptibility in part by inhibiting E2F1-mediated functions. 18791844_p202 protein is a novel, important mediator of chondrogenic and osteogenic differentiation. 19131592_study showed p202 is an inhibitor of DNA-induced caspase activation; conversely AIM2 was required for caspase activation by cytoplasmic dsDNA; work indicates HIN-200 proteins can act as pattern recognition receptors mediating response to cytoplasmic dsDNA 19890043_female and male sex hormones differentially regulate the expression of Ifi202, thus providing support for the role of Ifi202 in sex bias in SLE. 20167632_Results suggest that Ifi202 expression in the kidney and immune organs significantly increased with disease progression in autoimmune glomerulonephritis. 21057088_Aim2 deficiency stimulates the expression of IFN-inducible Ifi202, a lupus susceptibility murine gene within the Nba2 autoimmune susceptibility locus. 21124930_Findings delineate a new role of the IFN-gamma signaling pathway in regulating T cell proliferation and apoptosis through upregulating survivin and ifi202 expression. 21326316_Our findings indicate a potential role for Ifi202b in the suppressive capacity of peptide-induced regulatory CD8(+) Ti cells. 21551362_expression of Aim2 protein is required to maintain the expression of the Fcgr2b gene and also predict epistatic interactions between the Ifi200 genes and the Fcgr2b gene within the Nba2 interval 21943709_Our observations demonstrate that cell type and gender-dependent factors differentially regulate the expression of the Aim2 and p202 proteins 22692684_Ifi202b modulates fat accumulation through expression of adipogenic genes such as 11beta-Hsd1. 22784990_Studies identify p202 as a regulator of murine BAFF. 23850291_Authors hypothesize that while p202 was evolutionarily selected to limit AIM2-mediated inflammation in some mouse strains, the same mechanism contributes to increased interferon production and lupus susceptibility. 24246779_p202 plays a role in mouse adipose-derived stem cell adipogenesis. 24419611_These results, together with the reported data on p202 HINb, lead to an interaction model for full-length p202 and dsDNA which provides a conceivable mechanism for the negative regulation of Aim2 inflammasome activation by p202. 29478099_Ifi202b and IFI16 have roles as obesity genes 29853536_p202 promotes osteogenesis in mouse bone marrow stromal cells. 30104205_Enforced expression of IFI16-b inhibits the activation of AIM2 inflammasome, whereas knockdown of IFI16-b augments interleukin-1b secretion triggered by dsDNA but not dsRNA Thus, cytoplasm-localized IFI16-b is functionally equivalent to mouse p202 that exerts an inhibitory effect on AIM2 inflammasome. . 30246031_the expression level of Ifi202b showed the most sex difference in correlation with known immune-related genes |
ENSG00000163563 |
MNDA |
15.937339 |
0.0057414204 |
-7.444377 |
1.44406006 |
29.286720 |
0.00000006242202608828052966074732355386545101794126821914687752723693847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000006714776139665883108201228576805696945939416764304041862487792968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.191191721453307 |
0.224967980269909 |
33.3004218860335 |
15.6695468194898 |
| ENSMUSG00000026546 |
71870 |
Cfap45 |
protein_coding |
Q9D9U9 |
FUNCTION: Microtubule inner protein (MIP) part of the dynein-decorated doublet microtubules (DMTs) in cilia axoneme, which is required for motile cilia beating (By similarity). It is an AMP-binding protein that may facilitate dynein ATPase-dependent ciliary and flagellar beating via adenine nucleotide homeostasis. May function as a donor of AMP to AK8 and hence promote ADP production (PubMed:33139725). {ECO:0000250|UniProtKB:Q32LN4, ECO:0000269|PubMed:33139725}. |
Cell projection;Cilium;Coiled coil;Cytoplasm;Cytoskeleton;Flagellum;Reference proteome |
|
|
mmu:71870; |
9+0 motile cilium [GO:0097728]; 9+2 motile cilium [GO:0097729]; axonemal microtubule [GO:0005879]; axoneme [GO:0005930]; extracellular region [GO:0005576]; nucleoplasm [GO:0005654]; AMP binding [GO:0016208]; cerebrospinal fluid circulation [GO:0090660]; epithelial cilium movement involved in determination of left/right asymmetry [GO:0060287]; establishment of left/right asymmetry [GO:0061966]; flagellated sperm motility [GO:0030317]; regulation of cilium beat frequency involved in ciliary motility [GO:0060296] |
33139725_CFAP45 deficiency causes situs abnormalities and asthenospermia by disrupting an axonemal adenine nucleotide homeostasis module. |
ENSG00000213085 |
CFAP45 |
19.434826 |
6.8915546623 |
2.784829 |
0.70294565 |
13.827181 |
0.00020041585692700251324430138488708053046138957142829895019531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011125125119213199673118275256911147153005003929138183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
33.813737991397 |
17.1352539863851 |
4.90654716511002 |
1.83372838485922 |
| ENSMUSG00000026574 |
56429 |
Dpt |
protein_coding |
Q9QZZ6 |
FUNCTION: Seems to mediate adhesion by cell surface integrin binding. May serve as a communication link between the dermal fibroblast cell surface and its extracellular matrix environment. Enhances TGFB1 activity (By similarity). Inhibits cell proliferation. Accelerates collagen fibril formation, and stabilizes collagen fibrils against low-temperature dissociation. {ECO:0000250, ECO:0000269|PubMed:12230512, ECO:0000269|PubMed:14980498}. |
Cell adhesion;Disulfide bond;Extracellular matrix;Pyrrolidone carboxylic acid;Reference proteome;Repeat;Secreted;Signal;Sulfation |
|
|
mmu:56429; |
collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; extracellular matrix structural constituent [GO:0005201]; cell adhesion [GO:0007155]; collagen fibril organization [GO:0030199]; negative regulation of cell population proliferation [GO:0008285] |
12230512_Targeted disruption of dermatopontin causes abnormal collagen fibrillogenesis 14980498_Cloning of early quiescence-1 gene: Identification of a role in cell quiescence. 17118749_Dermatopontin is an abundant protein in demineralized bone matrix, and it can inhibit the stimulatory effects of BMP-2 on ALP activity in C2C12 cells. 17202853_results suggest that, at the time of implantation, dermatopontin expression in the uterus is primarily regulated spatio-temporally by progesterone via progesterone receptors, and is modulated by the decidual response during implantation 22614334_Aging in the mdx diaphragm seems to be associated with a drastic increase in extracellular matrix proteins incliding dermatopontin. 24909373_The present study provides the first evidence that DPT may be a novel effector of cadmium chloride, highlighting the significant role of DPT in the regulation of claudin-11 expression 25092262_The DP-4 peptide activated Fn and enhanced cell adhesion activity 26861869_DPT may affect testicular function by affecting the integrity of the bloodtestis barrier. 27365423_These findings illustrate a previously uncharacterized role of Dpt in maintaining hematopoietic stem cells ex vivo. 28844660_These data suggest that DPT fosters adherence but is not required for steady-state hematopoiesis in vivo. |
ENSG00000143196 |
DPT |
36.757958 |
0.1904544454 |
-2.392482 |
0.60908201 |
14.717602 |
0.00012487501219984349773092779933847396023338660597801208496093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007310131382890146549066723302701120701385661959648132324218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.0873509203807 |
4.38635650520103 |
52.9646388703804 |
16.0012752001793 |
| ENSMUSG00000026576 |
11931 |
Atp1b1 |
protein_coding |
P14094 |
FUNCTION: This is the non-catalytic component of the active enzyme, which catalyzes the hydrolysis of ATP coupled with the exchange of Na(+) and K(+) ions across the plasma membrane. The beta subunit regulates, through assembly of alpha/beta heterodimers, the number of sodium pumps transported to the plasma membrane.; FUNCTION: Involved in cell adhesion and establishing epithelial cell polarity. {ECO:0000250}. |
Cell adhesion;Cell membrane;Direct protein sequencing;Disulfide bond;Glutathionylation;Glycoprotein;Ion transport;Membrane;Phosphoprotein;Potassium;Potassium transport;Reference proteome;Signal-anchor;Sodium;Sodium transport;Sodium/potassium transport;Transmembrane;Transmembrane helix;Transport |
|
This gene encodes an integral membrane protein that comprises a subunit of an ATP-metabolizing enzyme responsible for transporting sodium and potassium ions across the plasma membrane. This enzyme regulates the electrochemical gradient of these ions in cells, and plays a central role in osmoregulation and signal transmission in nerves and muscles, among other biological processes. The encoded protein is the non-catalytic beta subunit; it works together with a catalytic alpha subunit and a gamma subunit. [provided by RefSeq, Mar 2013]. |
mmu:11931; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; caveola [GO:0005901]; intercalated disc [GO:0014704]; lateral plasma membrane [GO:0016328]; membrane [GO:0016020]; myelin sheath [GO:0043209]; organelle membrane [GO:0031090]; plasma membrane [GO:0005886]; sarcolemma [GO:0042383]; sodium:potassium-exchanging ATPase complex [GO:0005890]; sperm flagellum [GO:0036126]; T-tubule [GO:0030315]; ATPase activator activity [GO:0001671]; ATPase binding [GO:0051117]; P-type sodium:potassium-exchanging transporter activity [GO:0005391]; protein C-terminus binding [GO:0008022]; protein heterodimerization activity [GO:0046982]; protein kinase binding [GO:0019901]; protein-macromolecule adaptor activity [GO:0030674]; ATP metabolic process [GO:0046034]; cardiac muscle contraction [GO:0060048]; cation transmembrane transport [GO:0098655]; cell adhesion [GO:0007155]; cellular calcium ion homeostasis [GO:0006874]; cellular potassium ion homeostasis [GO:0030007]; cellular sodium ion homeostasis [GO:0006883]; establishment or maintenance of transmembrane electrochemical gradient [GO:0010248]; membrane repolarization [GO:0086009]; metal ion transport [GO:0030001]; positive regulation of ATP-dependent activity [GO:0032781]; positive regulation of calcium:sodium antiporter activity [GO:1903281]; positive regulation of P-type sodium:potassium-exchanging transporter activity [GO:1903408]; positive regulation of potassium ion import across plasma membrane [GO:1903288]; positive regulation of potassium ion transmembrane transporter activity [GO:1901018]; positive regulation of sodium ion export across plasma membrane [GO:1903278]; potassium ion import across plasma membrane [GO:1990573]; potassium ion transport [GO:0006813]; protein localization to plasma membrane [GO:0072659]; protein stabilization [GO:0050821]; proton transmembrane transport [GO:1902600]; regulation of calcium ion transmembrane transport [GO:1903169]; regulation of cardiac muscle contraction by calcium ion signaling [GO:0010882]; regulation of gene expression [GO:0010468]; relaxation of cardiac muscle [GO:0055119]; response to hypoxia [GO:0001666]; sodium ion export across plasma membrane [GO:0036376]; sodium ion transmembrane transport [GO:0035725]; sodium ion transport [GO:0006814] |
12397396_Beta(1) is the sole expressed beta-subunit in the diaphragm throughout development, and in the hindlimb muscles at birth. 17317668_the beta1 subunit of the Na/K-ATPase is required for blastocyst formation and also for maintaining a normal Na/K-ATPase distribution and localization of tight junction-associated polypeptides during preimplantation development 18621045_These data suggest that CLN3 is involved in the regulation of plasma membrane fodrin cytoskeleton and consequently, the plasma membrane association of Na(+), K(+) ATPase. 19487957_Inhibition of the Atp1b1 isoenzyme of the sodium potassium exchanging ATPase by 8-methoxycoumestrol is reported. 19683723_Na,K-beta(1) plays an essential role in regulating cardiac contractility and that its loss is associated with significant pathophysiology of the heart. 23534996_Atp1b1 was highly expressed in luminal epithelium during peri-implantation and upregulated by progesterone. 25012180_Activities of AE1 and the sodium pump are coregulated in kidney. 28515087_FXYD5 mediates metastatic progression through regulation of the beta Na(+)-K(+)-ATPase subunit in the 4T1 mouse breast cancer model. 30012868_Data show that differentially expressed in chondrocytes 1 protein (DEC1) and circadian locomoter output cycles kaput protein (CLOCK) bound to E-boxes in the beta1 subunit of the Na(+)/K(+)-ATPase (ATP1B1) promoter and suppressed the expression of ATP1B1. 30029086_Functional selection and assembly of NKA subunit isoforms occurs in the cochlear stria vascularis lateral wall, which is disrupted and dysregulated with age. 30160348_the transepithelial potential difference (TEP) across the retinal pigment epithelial (RPE) depends on the expression of ATP1B1 and this regulates the secretion of PEDF by RPE cells and so may regulate the onset of retinal disease. Increasing the expression of PEDF using an applied EF to replenish a disease or age-reduced TEP may offer a new way of preventing or reversing retinal dysfunction. 34192507_Hypercapnia Induces Inositol-Requiring Enzyme 1alpha-Driven Endoplasmic Reticulum-associated Degradation of the Na,K-ATPase beta-Subunit. |
ENSG00000143153 |
ATP1B1 |
53.662377 |
0.3579517601 |
-1.482163 |
0.40422321 |
12.791386 |
0.00034821895434814566791872914031102936860406771302223205566406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0018159317634252420889384715962933114496991038322448730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.7895193620909 |
7.88851535860256 |
77.6348169280097 |
15.727951570578 |
| ENSMUSG00000026587 |
11899 |
Astn1 |
protein_coding |
Q61137 |
FUNCTION: Neuronal adhesion molecule that is required for normal migration of young postmitotic neuroblasts along glial fibers, especially in the cerebellum. Required for normal rate of migration of granule cells during brain development and for normal cerebellum development. {ECO:0000269|PubMed:11861479, ECO:0000269|PubMed:8602532}. |
Alternative splicing;Cell adhesion;Cell membrane;Cytoplasmic vesicle;Disulfide bond;EGF-like domain;Endosome;Glycoprotein;Membrane;Methylation;Phosphoprotein;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:11899; |
clathrin-coated vesicle [GO:0030136]; endosome [GO:0005768]; external side of plasma membrane [GO:0009897]; membrane [GO:0016020]; perikaryon [GO:0043204]; cell migration [GO:0016477]; cell-cell adhesion [GO:0098609]; locomotory behavior [GO:0007626]; neuron cell-cell adhesion [GO:0007158]; neuron migration [GO:0001764] |
11861479_astrotactin is required for appropriate and timely migration of cerebellar granule cells. 30696770_Differences in the degree of Astn1 and Astn2 maturation possibly contribute to the higher levels of the C-terminal domain of Astn1 detected on neuronal membranes of the central nervous system |
ENSG00000152092 |
ASTN1 |
49.376351 |
0.0967993701 |
-3.368859 |
0.54864336 |
35.276021 |
0.00000000286133384325748050001718426600758743427732611053215805441141128540039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000377676231373465711054342197096345135065575959743000566959381103515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.81905620205435 |
3.43522908792961 |
91.1065453982006 |
23.8337520742873 |
| ENSMUSG00000026605 |
108000 |
Cenpf |
protein_coding |
F6V6F0 |
Human_homologues FUNCTION: Required for kinetochore function and chromosome segregation in mitosis. Required for kinetochore localization of dynein, LIS1, NDE1 and NDEL1. Regulates recycling of the plasma membrane by acting as a link between recycling vesicles and the microtubule network though its association with STX4 and SNAP25. Acts as a potential inhibitor of pocket protein-mediated cellular processes during development by regulating the activity of RB proteins during cell division and proliferation. May play a regulatory or permissive role in the normal embryonic cardiomyocyte cell cycle and in promoting continued mitosis in transformed, abnormally dividing neonatal cardiomyocytes. Interaction with RB directs embryonic stem cells toward a cardiac lineage. Involved in the regulation of DNA synthesis and hence cell cycle progression, via its C-terminus. Has a potential role regulating skeletal myogenesis and in cell differentiation in embryogenesis. Involved in dendritic cell regulation of T-cell immunity against chlamydia. {ECO:0000269|PubMed:12974617, ECO:0000269|PubMed:17600710, ECO:0000269|PubMed:7542657, ECO:0000269|PubMed:7651420}. |
Proteomics identification;Reference proteome |
|
|
|
axoneme [GO:0005930]; centrosome [GO:0005813]; chromosome, centromeric region [GO:0000775]; ciliary basal body [GO:0036064]; ciliary transition fiber [GO:0097539]; cytoplasm [GO:0005737]; midbody [GO:0030496]; nuclear envelope [GO:0005635]; nuclear matrix [GO:0016363]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; outer kinetochore [GO:0000940]; pronucleus [GO:0045120]; spindle [GO:0005819]; spindle pole [GO:0000922]; DNA-binding transcription factor binding [GO:0140297]; dynein complex binding [GO:0070840]; microtubule binding [GO:0008017]; protein C-terminus binding [GO:0008022]; protein homodimerization activity [GO:0042803]; chromosome segregation [GO:0007059]; kidney development [GO:0001822]; metaphase plate congression [GO:0051310]; mitotic cell cycle [GO:0000278]; negative regulation of DNA-templated transcription [GO:0045892]; protein transport [GO:0015031]; regulation of G2/M transition of mitotic cell cycle [GO:0010389]; regulation of striated muscle tissue development [GO:0016202]; ventricular system development [GO:0021591] |
14555653_LEK1 specifically functions during murine development to regulate the activity of Rb proteins during cell division and proliferation. 16047383_These data suggest a role for LEK1 in regulating the normal embryonic cardiomyocyte cell cycle and in promoting continued mitosis in transformed, abnormally dividing cardiomyocytes. 17295207_Results indicate that Raf-arrested myoblasts may serve as a model system for satellite cell cycle studies and that E2F5 and LEK1 translocation to the nucleus is an important first step during entry into quiescence. 17498689_The domains of the C-terminus of mCenp-F have a conserved function in control of mitotic progression through protein-protein interaction with pocket proteins. 18425850_These data demonstrate that CMF1 regulates myocyte differentiation by interaction with Rb family members to induce expression of myogenic regulatory factors. 18768859_LEK1 plays a prominent role in regulating dendritic cell (DC) function for T cell activation, since knockdown of LEK1 expression results in the rapid maturation of Chlamydia-exposed DCs as measured by analysis of key activation markers. 18827011_CENPF and syntaxin 4 colocalize with components of plasma membrane recycling: SNAP-25 and VAMP2. Depletion of endogenous CENPF disrupts GLUT4 trafficking 19793914_Results identify Hook2, a linker protein that is essential for regulation of the microtubule network at the centrosome, as a binding partner of CENP-F. 20053638_Data show that the post-anaphase, KEN-box-dependent degradation of Cenp-F requires it to be farnesylated, a post-translational modification usually linked to membrane association. 22563055_Loss of CENP-F function in developing myocytes leads to decreased cell division, blunting of trabeculation and an initially smaller, thin-walled heart. 26387954_FOXM1 and CENPF are master regulators of prostate cancer malignancy, and can serve as drug response markers for antineoplastic drugs efficiency. 27146114_Our data show that mutation of the CENP-F gene leads to an unexpected hyperstabilization of the MT network, with a unique loss of dynamic instability. With disruption of MT dynamics, CENP-F-/- cells exhibit dramatic loss of directionally persistent migration, defects in focal adhesion disassembly and lamellipodial formation/retraction, change in cilia frequency, and loss of regulation of cell shape. 29765066_Results found that loss of function mutation in CENPF results in dilated cardiomyopathy with significant disruption in the microtubule network of cardiac myocytes in mouse. 30408335_Loss of CENP-F Function Induces Kidney Enlargement. 30856164_Due to the multifunctionality of CENP-F, the cellular phenotypes observed upon its depletion are difficult to interpret and there is a need to genetically separate its different functions by preventing binding to selected partners. Here we engineer a CENP-F point-mutant that is deficient in Miro1/2 binding and thus is unable to localize to mitochondria, but retains other localizations. 31478449_Loss of CENPF leads to developmental failure in mouse embryos. 34676150_Apoptosis-induced translocation of centromere protein F in its corresponding autoantibody production in hepatocellular carcinoma. 36513638_CENP-F-dependent DRP1 function regulates APC/C activity during oocyte meiosis I. |
ENSG00000117724 |
CENPF |
1631.202580 |
2.2472037330 |
1.168131 |
0.14458184 |
62.718720 |
0.00000000000000238438566790195585919134159162534506905685004890527167731306690257042646408081054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000622966766612051386796799625063261669392173855430705486924125580117106437683105468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2241.17913748047 |
224.979264584829 |
997.319070177595 |
75.487003960015 |
| ENSMUSG00000026622 |
18005 |
Nek2 |
protein_coding |
O35942 |
FUNCTION: Protein kinase which is involved in the control of centrosome separation and bipolar spindle formation in mitotic cells and chromatin condensation in meiotic cells. Regulates centrosome separation (essential for the formation of bipolar spindles and high-fidelity chromosome separation) by phosphorylating centrosomal proteins such as CROCC, CEP250 and NINL, resulting in their displacement from the centrosomes. Regulates kinetochore microtubule attachment stability in mitosis via phosphorylation of NDC80. Involved in regulation of mitotic checkpoint protein complex via phosphorylation of CDC20 and MAD2L1. Plays an active role in chromatin condensation during the first meiotic division through phosphorylation of HMGA2. Phosphorylates: PPP1CC; SGO1; NECAB3 and NPM1. Essential for localization of MAD2L1 to kinetochore and MAPK1 and NPM1 to the centrosome. Phosphorylates CEP68 and CNTLN directly or indirectly (By similarity). NEK2-mediated phosphorylation of CEP68 promotes CEP68 dissociation from the centrosome and its degradation at the onset of mitosis (By similarity). Phosphorylates and activates NEK11 in G1/S-arrested cells. Involved in the regulation of centrosome disjunction (By similarity). {ECO:0000250|UniProtKB:P51955, ECO:0000269|PubMed:14668482, ECO:0000269|PubMed:14697346}. |
ATP-binding;Cell cycle;Cell division;Centromere;Chromosome;Chromosome partition;Coiled coil;Cytoplasm;Cytoskeleton;Kinase;Kinetochore;Magnesium;Meiosis;Metal-binding;Microtubule;Mitosis;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:18005; |
centrosome [GO:0005813]; condensed nuclear chromosome [GO:0000794]; cytoplasm [GO:0005737]; kinetochore [GO:0000776]; microtubule [GO:0005874]; midbody [GO:0030496]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; spindle pole [GO:0000922]; ATP binding [GO:0005524]; metal ion binding [GO:0046872]; protein kinase activity [GO:0004672]; protein phosphatase binding [GO:0019903]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; blastocyst development [GO:0001824]; cell division [GO:0051301]; centrosome separation [GO:0051299]; chromosome segregation [GO:0007059]; meiotic cell cycle [GO:0051321]; mitotic sister chromatid segregation [GO:0000070]; mitotic spindle assembly [GO:0090307]; negative regulation of centriole-centriole cohesion [GO:1903126]; negative regulation of DNA binding [GO:0043392]; positive regulation of telomerase activity [GO:0051973]; positive regulation of telomere capping [GO:1904355]; positive regulation of telomere maintenance via telomerase [GO:0032212]; protein autophosphorylation [GO:0046777]; protein phosphorylation [GO:0006468]; regulation of attachment of spindle microtubules to kinetochore [GO:0051988]; regulation of mitotic centrosome separation [GO:0046602] |
11785960_Nek2 may be a mitotic regulator that is involved in diverse cell cycle events. 11911479_Tcp10 promoter-directed expression of the Nek2 gene in mouse meiotic spermatocytes. 11923207_The MAPK pathway triggers activation of Nek2 during chromosome condensation in mouse spermatocytes. 14668482_Functional interaction between HMGA2 and Nek2 plays a crucial role in the correct process of chromatin condensation in meiosis 14697346_These results propose a possible involvement of Nek2 in biological processes of the Golgi body, perhaps in relation to the inheritance of Golgi during mitosis or to cell cycle stage-specific regulation of exocytosis. 15479717_importance of Nek2 for proper chromosome segregation in embryonic mitoses. 15611654_identified mouse Telomeric Repeat Binding Factor 1 (Trf1) as a protein that interacts directly with the spindle checkpoint protein Mad1 and the mitotic kinase Nek2 19117032_The phenotypic similarities of the Nip2/centrobin- and Nek2-suppressed embryos suggest that Nip2/centrobin may be a substrate of Nek2 that is required for mitotic spindle assembly in early mouse embryos. 20569513_Nek2 and its substrate, centrobin/Nip2, are required for proper meiotic spindle formation of the mouse oocytes 20718755_Data show that NEK2 was identified as a tumor-associated antigen. 23708664_these data indicate that Nek2 has a pivotal role in breast cancer growth at primary and secondary sites, and thus may be an attractive and novel therapeutic target for this disease. 24214971_CIP2A strongly interacts with NEK2 during G2/M phase, thereby enhancing NEK2 kinase activity to facilitate centrosome separation in a PP1- and PP2A-independent manner. 25485281_Nek2 transgenic mice develop spontaneous germinal centers and exhibit an enhanced T cell dependent immune response our data demonstrates a novel role for Nek2 in regulating B cell development and the immune response 26220856_Cep85 is a bona fide Nek2A-binding partner that surrounds the proximal ends of centrioles where it cooperates with PP1gamma (also known as PPP1CC) to antagonize Nek2A 26320076_The results suggest that overexpression of NEK2 has the potential to serve as a biomarker for PCa prognosis. 31034758_the present study evaluates the effects of miR-138 on chondrocyte proliferation, differentiation, and apoptosis through the WNT/beta-catenin signaling pathway in mice with OA by binding to NIMA-related kinase 2 (NEK2) 33256298_[NEK2 gene expression in mouse cryptorchidism model and its mechanism involved in apoptosis]. |
ENSG00000117650 |
NEK2 |
343.256789 |
2.1598226204 |
1.110913 |
0.19163393 |
33.316541 |
0.00000000783136917755746436380681797665218035753298408963019028306007385253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000961220107272806487025597213871841439924992300802841782569885253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
474.975676902035 |
63.2769596693534 |
219.91420610992 |
22.4103495302487 |
| ENSMUSG00000026628 |
11910 |
Atf3 |
protein_coding |
Q60765 |
FUNCTION: This protein binds the cAMP response element (CRE) (consensus: 5'-GTGACGT[AC][AG]-3'), a sequence present in many viral and cellular promoters (PubMed:11916968). Represses transcription from promoters with ATF sites (PubMed:11916968). It may repress transcription by stabilizing the binding of inhibitory cofactors at the promoter (By similarity). {ECO:0000250|UniProtKB:P18847, ECO:0000269|PubMed:11916968}. |
DNA-binding;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Repressor;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:11910; |
CHOP-ATF3 complex [GO:1990622]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; RNA polymerase II transcription regulator complex [GO:0090575]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; identical protein binding [GO:0042802]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; cellular response to amino acid starvation [GO:0034198]; endoplasmic reticulum unfolded protein response [GO:0030968]; gluconeogenesis [GO:0006094]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of gene expression [GO:0010628]; positive regulation of TRAIL-activated apoptotic signaling pathway [GO:1903984]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of transcription from RNA polymerase II promoter in response to endoplasmic reticulum stress [GO:1990440]; regulation of transcription by RNA polymerase II [GO:0006357]; response to organic substance [GO:0010033]; skeletal muscle cell differentiation [GO:0035914] |
11916968_ATF3 bound to the ATF/cAMP-responsvie element site derived from the promoter of the gene encoding the gluconeogenic enzyme phosphoenolpyruvate carboxykinase 12225289_ATF3 binds to JDP2 and has a role in directing its phosphorylation by JNK 12815047_ATF3 and a novel isoform, ATF3b, regulate proglucagon transcription through the cAMP-response element/ATF site of the proglucagon gene promoter 15199129_ATF3 is induced in beta cells by signals relevant to beta-cell destruction; induction of ATF3 is mediated in part by two stress-induced pathways implicated in both type 1 and type 2 diabetes. 16469745_ATF3 promotes cell death and cell arrest, and suppresses Ras-mediated tumorigenesis 16644691_NFATc4 and ATF3 function as negative regulators of adiponectin gene expression in 3T3 adipocytes. 16688168_ATF3 seems to inhibit Il6 and Il12b transcription by altering chromatin structure, thereby restricting access to transcription factors 16982098_ATF3 appears to be part of a control mechanism that limits the amount of CCL4 released by macrophages, preventing excessive inflammation. 17102133_expression of ATF-3 in hypoxic cells represses Id-1 and prevents their loss 17295236_ATF3 may have oncogenic properties in epithelial cells 17652582_ATF3 contributes to nerve regeneration by increasing the intrinsic growth state of injured neurons. 17697053_conclude that ATF3, at best, plays a minor role in the unfolded protein response(UPR) signaling and its expression is more likely induced by the UPR as a secondary event in oligodendrocytes that is unrelated to cell death. 17785797_ATF3 behaves as a negative regulatory transcription factor in Toll-like receptor pathways and, accordingly, deficiency in atf3 alters responses to immunological challenges in vivo. 18057093_IRS2 is a target gene of ATF3, and its repression by ATF3 contributes, at least partly, to the apoptosis induced by ATF3; this explains how environmental stress factors can modulate IRS2 gene transcription. 18192274_Sp1 recruits ATF3, c-Jun, and STAT3 to obtain the requisite synergistic effect in neuronal injury through DINE neuronal injury-inducible gene 18199529_ATF3 as a novel repressor of the Nrf2-directed stress response pathway. 18268321_ATF3 acts within natural killer cells to regulate antiviral responses 18308734_I 12 acquired a higher affinity toward EphA2 with K(d) 18 nm and inhibited vascular endothelial growth factor-dependent angiogenic invasion in a Matrigel plug assay 18719024_Data indicate that the ATF3 gene is a bona fide target gene of Egr-1, CREB, and ATF2 in gonadotrophs. 18794337_Results show that activating transcription factor 3 ameliorates allergen-induced airway inflammation and hyperresponsiveness in a mouse model of human asthma. 18808719_ATF3 may play an oncogenic role in human breast tumorigenesis, and therefore may be useful as either a biomarker or therapeutic target. 18922905_both ATF3 and Smad were crucially and synergistically involved in down-regulation of Id-1, which regulated JNK phosphorylation in REIC/Dkk-3-induced apoptosis. 19120490_knock out mice succumb to LPS-induced shock much more rapidly than WT mice 19478204_ATF3, which is induced in obese adipose tissue, acts as a transcriptional repressor of saturated fatty acids/TLR4 signaling, thereby revealing the negative feedback mechanism that attenuates obesity-induced macrophage activation 19647793_ATF3 functions as a potent regulator of STAT1 stability, accelerating streptozotocin-induced diabetes and diabetic liver injury. 20018623_HSF1 activated by heat shock induced the expression of ATF3, necessary for heat-mediated suppression of IL-6, indicating a fever-mediated feedback loop consisting of HSF1 and ATF3. 20203264_A key role for ATF3 in regulating mast cell survival and mediator release. 20349223_ATF3 plays a role in islet graft rejection by contributing to islet cell death and inflammatory responses at the graft sites. Silencing the ATF3 gene may provide therapeutic benefits in islet transplantation. 20351107_c-Src modulation and ATF3 activity may contribute to differential regulation of IRF-3- versus IRF-5-mediated gene expression 20360311_ATF3-mediated epigenetic regulation protects against acute kidney injury. 20413626_ATF3 as an important early transcriptional regulator of cyclic-stretch-induced responses 20519332_Results support a model in which, before apoptosis becomes obvious, expression of ATF3 can be beneficial by helping beta-cells to cope with higher metabolic demand. 20592017_MDM2 mediates ubiquitination and degradation of activating transcription factor 3. 20600850_The activating transcription factor 3 increased STAT1 transcription by directly binding to a putative binding region (- 116 to - 96 bp) in the STAT1 promoter. 20651982_Cisplatin is a MAPK pathway-dependent inducer of ATF3, whose expression influences cisplatin's cytotoxic effects. 21152039_ATF3 deficiency affects Kdo(2)-Lipid A-induced TLR4 signaling pathways in MEF cells, and may upregulate IkappaBzeta expression 21191795_displays a unique expression confined to the left heart chambers following acute angiotensin II stimulation 21304988_direct transcriptional targets of ATF3 protein in ATF3-induced mammary tumors 21451036_Increased expression of Atf3 mediates neuroprotection through a gene repression signaling cascade. 21467165_MUC18 is involved in cell signaling regulating the expression of Id-1 and ATF-3, thus contributing to melanoma metastasis 21986529_these results suggest that ATF3 inhibits PDX-1-mediated transactivation through the inhibition of p300-stimulated coactivation, which may lead to beta-cell dysfunction by ER stress. 22033410_the increase in Atf3 gene expression induced by thapsigargin and low glucose concentrations slightly contributes to the stimulation of islet cell apoptosis under these culture conditions. 22179317_a model of SAIS, secondary infections caused considerably less mortality in Atf3(-/-) mice than in wild-type mice, indicating that ROS-induced ATF3 crucially determines susceptibility to secondary infections during SAIS. 22466652_Endoplasmic reticulum stress target Atf3 acts at the SORT1 promoter region as a transcriptional repressor. 22473958_found that deletion of this TF results in increased lipid body accumulation, and that ATF3 directly regulates transcription of the gene encoding cholesterol 25-hydroxylase 22665497_ATF3 is a novel repressor of androgen signaling that can inhibit AR functions, allowing prostate cells to restore homeostasis 22704967_ATF3 is likely an important protective regulator in cerebral ischemic injury. 22768301_the role of cholesterol in prostate health, and provide a novel role for ATF3, and associated proteins within a large signaling network, as a cholesterol-sensing mechanism. 22989952_the recruitment of multiple HDAC members to JDP2 and ATF3 is part of their transcription repression mechanism. 23160513_Upregulation of microRNA-494 contributes to inflammatory or adhesion molecule-induced kidney injury after ischemia/reperfusion by inhibiting expression of ATF3. 23333392_These results suggest that ATF3 may play a role in adipocyte hypoxia-mediated mitochondrial dysfunction in obesity. 23389954_Overexpression of atf3 in 3T3-L1 preadipocytes blocked the effect of RyR3 silencing on adiponectin expression, indicating that an atf3-dependent pathway mediates the effect downstream of RyR3 silencing. 23462798_ATF3 plays an important role in the control of glucose and energy metabolism by regulating Agrp 23697557_It describe a novel role for ATF3 as a component of TLR-mediated survival in macrophages. 23712976_ATF2 is an important transcriptional factor relating to inflammation through the suppression of ATF3 in M1 macrophages of White adipose tissue. 23868936_Triglyceride-rich lipoprotein lipolysis products activate ATF3-JNK transcription factor networks and induce endothelial cells inflammatory response. 23874609_Ectopic expression of ATF3 is sufficient to promote cardiac hypertrophy and exacerbates the deleterious effect of chronic pressure overload; conversely, ATF3 deletion protects the heart. 24100018_NF-kappaB1 and ATF3 are critical for the mechanisms through which NOD2 downregulates cytokines and contributes to intestinal immune homeostasis. 24140652_results indicate that ATF3 is expressed in both pancreatic alpha and beta cells; determined ATF3 expression was increased by low glucose and decreased by high glucose in both alphaTC-1.6 and betaTC3 cells 24315873_Taken together, these data indicate that ATF3 is a novel negative regulator of osteoblast differentiation by specifically suppressing ALP gene expression in preosteoblasts. 24420912_Epithelium-derived exosomal ATF3 RNA attenuates ischemia-reperfusion induced kidney injury by inhibiting MCP-1 gene transcription. 24470589_A dichotomous role for ATF3-mediated regulation of neutrophilic responses: inhibition of neutrophil chemokine production but promotion of neutrophil chemotaxis. 24474789_Maintenance of neuromuscular junction innervation during the course of the disease in ATF3/SOD1(G93A) mice is associated with a substantial delay in muscle atrophy and improved motor performance 24515113_Thus, in addition to attenuating cell death, synaptic activity and expression of ATF3 render hippocampal neurons more resistant to acute dendrotoxicity and loss of synapses. 24550138_Increased expression of ATF3, increased left ventricular contractility and rescued heart failure. 24600036_Data indicate that pharmacological or genetic inactivation of PIKfyve rapidly induces expression of the transcription repressor ATF3, which is necessary and sufficient for suppression of type I IFN expression. 24771044_ATF3 binds to carboxyl-terminal modulator protein and suppresses its transcription following brain ischemia. 24911970_resveratrol enhances the renal expression of the anti-aging Klotho gene, and the transcriptional factors ATF3 and c-Jun functionally interact and coordinately regulate the resveratrol-mediated transcriptional activation of Klotho. 24951825_ATF3 regulates innate immunity positively upon pneumococcus infection by enhancing TNF-alpha, IL-1beta, and IFN-gamma expression and modulating bacterial clearance. 24973221_These results suggest that metformin exhibits anti-inflammatory action in macrophages at least in part via pathways involving AMPK activation and ATF-3 induction. 25074928_Atf3-silencing reversed ethanol-mediated Gck down-regulation and beta-cell dysfunction, followed by the amelioration of impaired glucose tolerance and insulin resistance. 25122760_superinduced ATF3 attenuated ER stress-induced cancer chemokine expression by epigenetically interfering with induction of EGR-1, a transcriptional modulator crucial to cancer chemokine production. 25151953_Activation of ATF3 by endoplasmic reticulum stress is essential for the cardioprotective effects of late ischemic preconditioning. 25180886_Counterbalance between MIC-1 and ATF3 is critical for deciding the fate of enterocytes under the food chemical stress. 25401197_ATF3 confers protection to lung injury by preventing inflammatory cell recruitment and barrier disruption in a cell-specific manner, ATF3 deletion releases Nrf2 from ATF3-mediated transcriptional inhibition; however, absence of ATF3 results in Nrf2 proteasomal degradation. 25445599_ATF3-mediated inhibition of PPARgamma expression may contribute to inhibition of adipocyte differentiation during cellular stress including ER stress. 25446101_ATF3 interacts with PPARgamma and represses PPARgamma-mediated transactivation through suppression of p300-stimulated coactivation in 3T3-L1 cells 25518930_Results showed that PLY induces ATF3 expression during infection when bound to TLR4 and TLR2/4 regulate ATF3 induction via the JNK/p38 pathway. 25531328_results thus link ATF3 to the AKT signaling, and suggest that ATF3 is a tumor suppressor for the major subset of prostate cancers harboring dysfunctional Pten 25589515_Knockout of the ATF3 gene reduced the stimulatory effect of TGFbeta on fibroblasts by interfering with canonical Smad signalling and protected the mice from experimental fibrosis in two different models. 25619459_these results demonstrate that during acute inflammation Atf3 negatively regulates Ptgs2 25730876_Ethanol exposure blocked the recruitment of CREB and its coactivator CRTC2 to gluconeogenic promoters by up-regulating ATF3, a transcriptional repressor that also binds to cAMP-responsive elements 25752455_Involvement of ATF3 in the negative regulation of iNOS expression and NO production in activated macrophages 26201690_ATF3 is a key regulator in cardiac hypertrophy through a cross-talk between cardiomyocytes and macrophages. 26224859_our data demonstrate that a cell-autonomous program consisting of a stress-induced Atf3-gelsolin cascade affects the change in dendritic spine morphology in models of tuberous sclerosis complex 26342068_ATF3 suppresses fibrosis early but not late during infarct-induced heart failure. Although ATF3 deficiency was associated with more fibrosis, this did not occur at the expense of survival, which was higher in the ATF3-null mice. 26412238_Activating transcription factor 3 represses inflammatory responses by binding to the p65 subunit of NF-kappaB 26416280_Data show that activating transcription factor 3 (ATF3) acted as a transcriptional repressor and regulated interferon-beta (IFN-beta) via direct binding to a regulatory site distal to the Ifnb1 promoter. 26522727_Loss of activating transcription factor 3 (ATF3) in knockout mice promotes the emergence of keratins CK5+CK8+ epithelial cells. 26780683_Study shows that ATF3 regulates multiple targets in cardiomyocytes and plays critical roles in hypertrophy, apoptosis and autophagy of cardiomyocyte. 26900013_ATF3 was up-regulated in brain after reperfusion following transient middle cerebral artery occlusion. 27087439_The microvascular injury phenotypes observed in vitro and in vivo were similar. ATF3 plays an important role in mediating brain microvascular responses to acute and chronic lipotoxic injury and may be an important preventative and therapeutic target for endothelial dysfunction in vascular cognitive impairment . 27159257_These findings suggest that Atf3 is implicated in the pathogenesis of osteoarthritis through modulation of inflammatory cytokine expression in chondrocytes. 27260954_A novel role for ATF3 in the inhibition of lipopolysaccharide-induced TNF-alpha and in the impairment of monocyte and macrophage migration, is reported. 27480204_Conditional deletion of ATF3 in osteoclast precursors protects RANKL-induced osteoclast activation and bone loss. 27573240_Mechanistic studies showed that in MDSCs, ATF3 was transrepressed by the GC receptor GR through direct binding to the negative GR-response element. S100A9 is the major transcriptional target of ATF3 in Granulocytic Myeloid-derived Suppressor Cells. 27581653_Results show a pro-regenerative ATF3 function during PNS nerve regeneration involving transcriptional activation of a neuropeptide-encoding regeneration-associated gene cluster. 27659012_we have identified ATF3 as an important regulator of cisplatin cytotoxicity and that ATF3 inducers in combination with platins are a potential novel therapeutic approach for NSCLC. 27829167_ATF3 is a new co-factor of c-Fos and NFATc1 to activate osteoclast differentiation and activity. 27856557_ATF3 attenuates the expression of inflammation-related genes after exercise and thus facilitates molecular adaptation to training. 27870968_ATF3-KO mice escape from PE-dependent maladaptive cardiac remodeling by suppressing the IFNgamma-CXCL10-CXCR3 axis at multiple levels. 28004841_this study reveals a novel mechanism of naringenin through AMPK-ATF3-dependent negative regulation of the LPS/TLR4 signalling pathway, which thereby confers protection against murine endotoxaemia. 28082453_ATF3 has a protective role in dampening the high fat-induced cardiac remodeling processes. 28093456_The different anti-inflammatory mechanisms of the ApoA-I cysteine mutants might be associated with the regulation of ATF3 level. 28177121_ATF3 protects against LPS-induced acute lung injury by inhibiting TL1A expression. 28185985_cigarette smoke exposure up-regulated the expression of ATF3 in mice 28193539_This study shows, for the first time, that alpha-lactalbumin isolated in a rare 28kDa dimeric form induces cell death, while 14kDa monomeric alpha-lactalbumin is inactive. 28249877_ATF3 upregulation in cardiac fibroblasts in response to hypertensive stimuli protects the heart by suppressing Map2K3 expression and subsequent p38-transforming growth factor-beta signaling. 28440917_ATF3 inhibit the expression and release of TNF-alpha, IL-1beta, IL-6, and IL-18 induced by Mycoplasma pneumonia in vitro and in vivo, which is associated with its negative regulation of Egr-1/Fyn signaling pathway. 28522413_Microglia make contact through unknown neuronal signals regulated by ATF3 in hypoglossal nucleus. 28662411_The lung injury score and mortality were higher in ATF3 knock-out mice treated with Pseudomonas aeruginosa. Moreover, ATF3 was demonstrated to bind to lipopolysaccharide binding protein. These findings suggest ATF3 protects mice against acute lung injury induced by Pseudomonas aeruginosa partly due to the binding to lipopolysaccharide binding protein. 28684431_converges with CSL in negative control of cancer-associated fibroblast activation 28701342_Results reveal a critical role for ATF3 as a key regulator of the acinar cell transcriptional response during injury and may provide a link between chronic pancreatitis and PDAC. 28714032_ATF3 promotes macrophage migration and reverses M1polarized macrophages to the M2 phenotype by upregulation of TNC via the Wnt/betacatenin signaling pathway. 28784776_ATF3 in the host cells regulated a chemotherapeutic agent to immune modulation and cancer metastasis. 28822763_Myotube contraction increased ATF3 level, which modified chemokine expression. In skeletal muscle after downhill running, ATF3 also modified chemokine expression. 28869597_Study reports that Atf3-deficient mice developed spontaneous tumors, and died significantly earlier than wild-type mice. 29378244_ATF3 is involved in promoting particulate matter-induced pulmonary inflammation. 29463176_we identified ATF3 as a new transcription factor-mediating traumatic brain injury-associated inflammatory responses 29535089_Data (including data from studies using mutant, transgenic, and knockout mice) suggest that gene targets of leptin/leptin-receptor (Lep/Lepr) signaling in hypothalamic neurons regulate energy metabolism; Lep/Lepr signaling appears to up-regulate expression of Atf3 (activating transcription factor-3) in hypothalamic neurons. 29670287_electrophilic properties of itaconate and derivatives regulate the IkappaBzeta-ATF3 inflammatory axis; results demonstrate that targeting the DI-IkappaBzeta regulatory axis could be an important new strategy for the treatment of IL-17-IkappaBzeta-mediated autoimmune diseases 29716766_these data indicated that ATF3 might promote intestinal epithelial cell apoptosis in Crohn's disease via up-regulating the stability and transcription activity of p53 29895822_The present results therefore suggest that ATF3 deficiency is involved in the pathogenesis of metabolic syndrome 30185770_ATF3 deficiency activated mTOR/p70S6K/HIF-1alpha signaling. 30214444_these data suggest a mechanism in which macrophage ATF3 promotes IL-17A production in gammadelta T cells to rapidly induce host defenses during early S. pneumoniae infection. 30224057_The role of Atf3 in obesity. 30290178_It identified NFIA and ATF3 as drivers of astrocyte differentiation from neural precursor cells while RUNX2 promotes astrocyte maturation. 30455690_via IL-22-pSTAT3 signaling in the epithelium and IL-6-pSTAT3 signaling in Th17 cells, ATF3 mediates a cross-regulation in the barrier to maintain mucosal homeostasis and immunity. 30670778_ATF3 regulates the expression of essential tumor promoting factors, such as Sdf1 expressed by cancer associated fibroblasts within the tumor microenvironment, and thus restrain tumor growth. 30818334_Transgenic mice with cardiac expression of either JDP2 or ATF3 showed maladaptive remodeling and cardiac dysfunction. Surprisingly, JDP2 knockout (KO) did not protect the heart following transverse aortic constriction. Instead, the JDP2 KO mice performed worse than their wild type counterparts. 31144288_Proteomics analysis of HSV-1-induced alterations in mouse brain microvascular endothelial cells. 31529495_Hepatic IRF2BP2 Mitigates Nonalcoholic Fatty Liver Disease by Directly Repressing the Transcription of ATF3. 31667363_genetic deletion of activating transcription factor 3 (ATF3 (-/-) ) under a high-fat diet resulted in obesity and insulin resistance, which was abrogated by virus-mediated ATF3 restoration 31709711_Activating transcription factor 3 modulates the macrophage immune response to Mycobacterium tuberculosis infection via reciprocal regulation of inflammatory genes and lipid body formation. 31907991_Klotho regulation by albuminuria is dependent on ATF3 and endoplasmic reticulum stress. 32096565_Cardiolipin Synthase 1 Ameliorates NASH Through Activating Transcription Factor 3 Transcriptional Inactivation. 32184357_The mRNA levels of heat shock factor 1 are regulated by thermogenic signals via the cAMP-dependent transcription factor ATF3. 32571839_Critical Role of Intestinal Microbiota in ATF3-Mediated Gut Immune Homeostasis. 32692156_The ATF3 Transcription Factor Is a Short-Lived Substrate of the Arg/N-Degron Pathway. 32959354_Activating transcription factor 3 is a potential target and a new biomarker for the prognosis of atherosclerosis. 33311456_Transcriptional factor ATF3 promotes liver fibrosis via activating hepatic stellate cells. 33370278_The spliceosome inhibitors isoginkgetin and pladienolide B induce ATF3-dependent cell death. 33640762_Activating transcription factor 3 involved in Pseudomonas aeruginosa PAO1-induced macrophage senescence. 33652981_ATF3-Induced Mammary Tumors Exhibit Molecular Features of Human Basal-Like Breast Cancer. 34172832_Genetic loss-of-function of activating transcription factor 3 but not C-type lectin member 5A prevents diabetic peripheral neuropathy. 34298975_Stress-Inducible Gene Atf3 Dictates a Dichotomous Macrophage Activity in Chemotherapy-Enhanced Lung Colonization. 34309175_ATF3 induces RAB7 to govern autodegradation in paligenosis, a conserved cell plasticity program. 34468254_Circ_nuclear factor I X (circNfix) attenuates pressure overload-induced cardiac hypertrophy via regulating miR-145-5p/ATF3 axis. 34944038_Activating Transcription Factor 3 Protects against Restraint Stress-Induced Gastrointestinal Injury in Mice. 35061333_Nickel oxide nanoparticles induce apoptosis and ferroptosis in airway epithelial cells via ATF3. 35331713_NRF2/HO-1 pathway activation by ATF3 in a noise-induced hearing loss murine model. 35370996_ATF3 Positively Regulates Antibacterial Immunity by Modulating Macrophage Killing and Migration Functions. 35841815_Activating transcription factor 3 protects alveolar epithelial type II cells from Mycobacterium tuberculosis infection-induced inflammation. 35843476_A novel function of ATF3 in suppression of ferroptosis in mouse heart suffered ischemia/reperfusion. 36675183_ATF3/SPI1/SLC31A1 Signaling Promotes Cuproptosis Induced by Advanced Glycosylation End Products in Diabetic Myocardial Injury. 36690638_The transcription factor ATF3 switches cell death from apoptosis to necroptosis in hepatic steatosis in male mice. 36768628_Activating Transcription Factor 3 Diminishes Ischemic Cerebral Infarct and Behavioral Deficit by Downregulating Carboxyl-Terminal Modulator Protein. 36775433_Genetic loss-of-function of activating transcription factor 3 but not C-type lectin member 5A prevents diabetic peripheral neuropathy. |
ENSG00000162772 |
ATF3 |
49.311958 |
2.4562817853 |
1.296476 |
0.44354389 |
8.299634 |
0.00396530644410518842701840469544549705460667610168457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0152101606035281375350898258602683199569582939147949218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
70.4360647914935 |
21.9977017448813 |
28.675889392315 |
6.87044522008615 |
| ENSMUSG00000026676 |
74186 |
Ccdc3 |
protein_coding |
Q9D6Y1 |
FUNCTION: Negatively regulates TNF-alpha-induced pro-inflammatory response in endothelial cells (ECs) via inhibition of TNF-alpha-induced NF-kappaB activation in ECs (By similarity). Positively regulates lipid accumulation in adipose cells (PubMed:25605713). {ECO:0000250|UniProtKB:Q9BQI4, ECO:0000269|PubMed:25605713}. |
Coiled coil;Direct protein sequencing;Glycoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:74186; |
endoplasmic reticulum [GO:0005783]; extracellular region [GO:0005576]; lipid biosynthetic process [GO:0008610]; negative regulation of gene expression [GO:0010629]; negative regulation of lipid biosynthetic process [GO:0051055]; negative regulation of lipid metabolic process [GO:0045833]; negative regulation of tumor necrosis factor-mediated signaling pathway [GO:0010804]; positive regulation of fat cell differentiation [GO:0045600]; positive regulation of lipid biosynthetic process [GO:0046889] |
20043878_These results indicate that CCDC3 is a protein secreted by adipocytes and endothelial cells, and that its level is regulated both hormonally and nutritionally. 25605713_deficiency of Favine attenuated differentiation of primary adipocytes. Taken together, these data demonstrate that Favine has adipogenic and lipogenic effects on adipocytes 28827783_Altogether, these results demonstrate that CCDC3 modulates liver lipid metabolism by inhibiting liver de novo lipogenesis as a downstream player of the p63 network. |
ENSG00000151468 |
CCDC3 |
6.522115 |
0.0545810920 |
-4.195455 |
1.40281052 |
10.154369 |
0.00143959642841714877993519738907934879534877836704254150390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0063940748965331138317647052815573260886594653129577636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.64071389839919 |
0.725919314375216 |
11.7387519233941 |
6.51654676729877 |
| ENSMUSG00000026725 |
329278 |
Tnn |
protein_coding |
Q80Z71 |
FUNCTION: Extracellular matrix protein that seems to be a ligand for ITGA8:ITGB1, ITGAV:ITGB1 and ITGA4:ITGB1 (By similarity) (PubMed:14709716). Involved in neurite outgrowth and cell migration in hippocampal explants (PubMed:12812753). During endochondral bone formation, inhibits proliferation and differentiation of proteoblasts mediated by canonical WNT signaling (PubMed:17395156). In tumors, stimulates angiogenesis by elongation, migration and sprouting of endothelial cells (By similarity). Expressed in most mammary tumors, may facilitate tumorigenesis by supporting the migratory behavior of breast cancer cells (PubMed:15592496). {ECO:0000250|UniProtKB:Q9UQP3, ECO:0000269|PubMed:12812753, ECO:0000269|PubMed:14709716, ECO:0000269|PubMed:15592496, ECO:0000269|PubMed:17395156}. |
Alternative splicing;Direct protein sequencing;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:329278; |
CA3 pyramidal cell dendrite [GO:0097442]; cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; hippocampal mossy fiber expansion [GO:1990026]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; tenascin complex [GO:0090733]; identical protein binding [GO:0042802]; integrin binding [GO:0005178]; axonogenesis [GO:0007409]; cell-matrix adhesion [GO:0007160]; dendrite self-avoidance [GO:0070593]; negative regulation of canonical Wnt signaling pathway involved in osteoblast differentiation [GO:1905240]; negative regulation of neuron migration [GO:2001223]; negative regulation of osteoblast differentiation [GO:0045668]; negative regulation of osteoblast proliferation [GO:0033689]; neuron migration [GO:0001764]; neuron projection extension [GO:1990138]; osteoblast development [GO:0002076]; positive regulation of neuron projection development [GO:0010976]; positive regulation of sprouting angiogenesis [GO:1903672]; regulation of bone development [GO:1903010]; regulation of cell adhesion [GO:0030155]; regulation of cell migration [GO:0030334]; regulation of smooth muscle tissue development [GO:1905899] |
14709716_Tenascin-W is expressed during palate formation, osteogenesis and smooth muscle development. 17395156_Our results indicate that TN-W is a novel marker of preosteoblasts in early stage of osteogenesis, and that TN-W inhibits cell proliferation and differentiation of preosteoblasts mediated by canonical Wnt signaling. 22211116_Tenascin-W is able to inhibit cell spreading in vitro and it is upregulated in dermal fibroblasts taken from a tenascin-C knockout mouse. 24101721_the CD34-positive whisker follicle stem cell niche contains both tenascin-C and tenascin-W, and these glycoproteins might play a role in directing the migration and proliferation of these stem cells 25868708_Results show that tenascin-W acts as a niche component for breast cancer metastasis to bone by supporting cell migration and cell proliferation of the cancer cells. 26238448_TN-C may enhance inflammatory responses by accelerating macrophage migration and synthesis of proinflammatory/profibrotic cytokines via integrin alphaVbeta3/FAK-Src/NF-kappaB, resulting in increased fibrosis. 33552067_Pivotal Role of Tenascin-W (-N) in Postnatal Incisor Growth and Periodontal Ligament Remodeling. 36206596_TNN is first linked to auditory neuropathy. |
ENSG00000120332 |
TNN |
1000.340485 |
5.0779411456 |
2.344244 |
0.16027741 |
199.580565 |
0.00000000000000000000000000000000000000000000257847202169245040328675684319619849307623189624070294025696727910925652047089892519655301274714250248667351235270193965387619527973583899438381195068359375000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000003636600540224018931778637470272396443990105958810444158003601651459375557048003681415347260602922828770603444828446448866543505573645234107971191406250000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1671.07892317637 |
186.032035599915 |
329.085917944592 |
28.1837943954445 |
| ENSMUSG00000026726 |
65969 |
Cubn |
protein_coding |
Q9JLB4 |
FUNCTION: Endocytic receptor which plays a role in lipoprotein, vitamin and iron metabolism by facilitating their uptake. Acts together with LRP2 to mediate endocytosis of high-density lipoproteins, GC, hemoglobin, ALB, TF and SCGB1A1. Acts together with AMN to mediate endocytosis of the CBLIF-cobalamin complex. Binds to ALB, MB, Kappa and lambda-light chains, TF, hemoglobin, GC, SCGB1A1, APOA1, high density lipoprotein, and the CBLIF-cobalamin complex. Ligand binding requires calcium. Serves as important transporter in several absorptive epithelia, including intestine, renal proximal tubules and embryonic yolk sac. May play an important role in the development of the peri-implantation embryo through internalization of APOA1 and cholesterol. Binds to LGALS3 at the maternal-fetal interface. {ECO:0000269|PubMed:10766831, ECO:0000269|PubMed:11278724, ECO:0000269|PubMed:11856751, ECO:0000269|PubMed:15342463}. |
Calcium;Cell membrane;Cholesterol metabolism;Cleavage on pair of basic residues;Coated pit;Cobalamin;Cobalt;Direct protein sequencing;Disulfide bond;EGF-like domain;Endosome;Glycoprotein;Lipid metabolism;Lysosome;Membrane;Metal-binding;Phosphoprotein;Protein transport;Reference proteome;Repeat;Signal;Steroid metabolism;Sterol metabolism;Transport |
|
|
mmu:65969; |
apical part of cell [GO:0045177]; apical plasma membrane [GO:0016324]; brush border [GO:0005903]; brush border membrane [GO:0031526]; clathrin-coated pit [GO:0005905]; coated vesicle [GO:0030135]; cytoplasm [GO:0005737]; endocytic vesicle [GO:0030139]; endocytic vesicle membrane [GO:0030666]; endoplasmic reticulum [GO:0005783]; endosome [GO:0005768]; extracellular exosome [GO:0070062]; Golgi apparatus [GO:0005794]; Golgi-associated vesicle [GO:0005798]; lysosomal lumen [GO:0043202]; lysosomal membrane [GO:0005765]; membrane [GO:0016020]; microvillus membrane [GO:0031528]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; receptor complex [GO:0043235]; calcium ion binding [GO:0005509]; cargo receptor activity [GO:0038024]; cobalamin binding [GO:0031419]; hemoglobin binding [GO:0030492]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803]; signaling receptor activity [GO:0038023]; cholesterol metabolic process [GO:0008203]; cobalamin catabolic process [GO:0042366]; cobalamin transport [GO:0015889]; endocytic hemoglobin import into cell [GO:0020028]; establishment of localization in cell [GO:0051649]; in utero embryonic development [GO:0001701]; lipoprotein transport [GO:0042953]; receptor-mediated endocytosis [GO:0006898]; response to bacterium [GO:0009617]; response to nutrient [GO:0007584] |
12815097_Impaired protein endocytosis caused by invalidation of ClC-5 primarily reflects a trafficking defect of megalin and cubilin in PTC. 14983511_Cubilin expression is pronounced in the extraembryonic visceral endoderm of embryos. Cubilin, is also expressed by a subpopulation of cells dispersed within the embryonic endoderm having a migratory morphology. 15342463_Amn is an essential component of the Cubn receptor complex 16787536_Cubilin is required for embryonic development and is essential for the formation of somites, and definitive endoderm. 18400109_Inactivation of RFC1 impacts the expression of several ligands and interacting proteins in the cubilin-amnionless-megalin complex that are involved in the maternal-fetal transport of folate and other nutrients. 23592779_plasma membrane binding of Fgf8, and most likely of the Fgf8 family members Fgf17 and Fgf18, to Cubn improves Fgf ligand endocytosis and availability to FgfRs, thus modulating Fgf signaling activity. 23773363_cubilin and megalin mRNA expression is under epigenetic control 24357674_Cubilin deficiency reduces renal salvage and delivery back to the blood of albumin and apolipoprotein (apo)A-I, which decreases blood levels of albumin and apoA-I/HD 26148765_In adult mouse testes, strong Cubn immunoreactivity was found in the elongating spermatids. In Sertoli cells and peritubular cells, Cubn immunoreactivity was weak throughout the testis development. In the inter-stitium, Cubn immunoreactivity was found in foetal Leydig cells, was weak to negligible in the stem cells and progenitor Leydig cells and was strong in immature and adult Leydig cells. 29186386_Cubilin is involved in 25(OH) vitamin D uptake by adipocytes. 29186471_Low cubilin expression is associated with proteinuria. 32200668_In the proximal kidney tubules cubilin binding to albumin is tuned to capture normally filtered levels of the protein. |
ENSG00000107611 |
CUBN |
86.082863 |
2.1762014216 |
1.121812 |
0.29064755 |
14.862410 |
0.00011564468878930946255708528402905699294933583587408065795898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006809044838186168683430432579939406423363834619522094726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
120.246854433063 |
24.3893178322184 |
55.2553882373918 |
8.64492898958913 |
| ENSMUSG00000026768 |
241226 |
Itga8 |
protein_coding |
A2ARA8 |
FUNCTION: Integrin alpha-8/beta-1 functions in the genesis of kidney and probably of other organs by regulating the recruitment of mesenchymal cells into epithelial structures. It recognizes the sequence R-G-D in a wide array of ligands including TNC, FN1, SPP1 TGFB1, TGFB3 and VTN. NPNT is probably its functional ligand in kidney genesis. Neuronal receptor for TNC it mediates cell-cell interactions and regulates neurite outgrowth of sensory and motor neurons. {ECO:0000269|PubMed:10742111, ECO:0000269|PubMed:11470831, ECO:0000269|PubMed:11891185, ECO:0000269|PubMed:12787402, ECO:0000269|PubMed:17537792, ECO:0000269|PubMed:9054500, ECO:0000269|PubMed:9548928, ECO:0000269|PubMed:9614184}. |
Alternative splicing;Calcium;Cell adhesion;Cell membrane;Cleavage on pair of basic residues;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Integrin;Membrane;Metal-binding;Neurogenesis;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the integrin family of cell surface proteins that mediate cellular interactions with the extracellular matrix and other cells. The encoded protein undergoes proteolytic processing to generate the disulfide-linked heterodimeric alpha subunit which, in turn associates with a beta subunit to form the functional integrin receptor. Mice lacking the encoded protein mostly die after birth due to kidney defects, but some of animals that survive exhibit defects in the sensory hair cells of the inner ear. [provided by RefSeq, Aug 2016]. |
mmu:241226; |
apical part of cell [GO:0045177]; cell surface [GO:0009986]; dendritic spine membrane [GO:0032591]; endoplasmic reticulum [GO:0005783]; external side of plasma membrane [GO:0009897]; glutamatergic synapse [GO:0098978]; integrin complex [GO:0008305]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic density membrane [GO:0098839]; integrin binding [GO:0005178]; metal ion binding [GO:0046872]; brain development [GO:0007420]; cell adhesion mediated by integrin [GO:0033627]; cell projection organization [GO:0030030]; cell-cell adhesion [GO:0098609]; cell-matrix adhesion [GO:0007160]; establishment of protein localization [GO:0045184]; extracellular matrix organization [GO:0030198]; inner ear morphogenesis [GO:0042472]; integrin-mediated signaling pathway [GO:0007229]; kidney development [GO:0001822]; memory [GO:0007613]; mesodermal cell differentiation [GO:0048333]; metanephros development [GO:0001656]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of transforming growth factor beta receptor signaling pathway [GO:0030511]; smooth muscle cell differentiation [GO:0051145]; smooth muscle tissue development [GO:0048745]; substrate adhesion-dependent cell spreading [GO:0034446]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
12386295_Lack of alpha8 causes unilateral or bilateral renal agenesis, significantly reduced renal mass, and higher blood pressure. Many alpha8-deficient mice had malformations of the kidney with collapse or atrophy of tubuli and interstitial cell infiltrates. 12787402_Role in regulation of mesangial cell phenotype. Seems to promote adhesion, but inhibit migration and proliferation of mesangial cells. Alpha8 integrin could play important role in maintaining tissue integrity in glomerulus during glomerular injury. 15721307_Integrin alpha8beta1-fibronectin-mediated survival appears to be mediated by the PI3 kinase pathway 17537792_results thus place nephronectin and alpha8beta1 integrin in a pathway that regulates Gdnf expression and is essential for kidney development 17951999_Interactions between alpha(8)-integrin and the mesangial matrix may contribute to healing of glomerular injury by influencing cell proliferation and apoptosis 19008864_Lack of alpha8 integrin does not reduce fibrotic changes in the hearts of DOCA-salt hypertensive mice. 19096376_The induction of -integrin in -smooth muscle actin (-SMA) positive myofibroblasts of the liver and heart suggests that -integrin might modulate the function of activated myofibroblasts, cells contributing to fibrotic remodeling. 19769957_we propose that integrin alpha8beta1 plays a critical role in lung morphogenesis by regulating mesenchymal cell adhesion and migration 20132319_Mice mutant for alpha8-integrin exhibit a specific impairment of long-term potentiation at synapses of Schaffer collateral-CA1 field of the hippocampus. 20826576_alpha8-integrin is protective for the structure and function of the glomerulus and maintains podocyte integrity during the development of diabetic nephropathy 21194485_Lack of alpha8 integrin leads to morphological changes in renal mesangial cells, but not in vascular smooth muscle cells. 22613833_The findings indicate that QBRICK facilitates the integrin alpha8beta1-dependent interactions of cells with basement membranes by regulating the basement membrane assembly of nephronectin and explain why renal defects occur in Fraser syndrome. 23144868_Alpha 8 integrin expression is strongly induced in a mouse model of renal fibrosis. 25482639_Fibrillin-1 and alpha8 integrin interact, and thus, regulate mesangial cell adhesion and migration. 26938996_Itga8 seems to attenuate tubulointerstitial fibrosis in unilateral ureteral obstruction not via regulation of cell turnover, but via regulation of TGF-beta signalling, fibroblast activation and/or immune cell infiltration 27092791_Mechanistically, ligation of alpha8beta1 by the milk protein Mfge8 reduces antral smooth muscle contractile force by preventing RhoA activation through a PTEN-dependent mechanism. 28136130_alpha8 Integrin is expressed in functional placental compartments among its ligands, OPN and/or FN, across species. Although this may point to a regulatory role in trophoblast function, our data from alpha8 integrin-deficient mice indicated only mild placental pathology. 28572914_Study observed that in the Apoe-deficient model of atherosclerosis, the female sex is a risk factor to develop more severe atherosclerotic lesions, although serum fat levels are not higher in females than in males. In contrast, female mice are protected from renal damage induced by the concomitant deficiency of Apoe and Itga8 29544224_Itga8-/- renal mesangial cells showed reduced phagocytosis of matrix-coated beads and apoptotic cells compared to WT renal mesangial cells. 29813125_Role of integrin alpha8 in murine model of lung fibrosis |
ENSG00000077943 |
ITGA8 |
1062.768637 |
0.3339026837 |
-1.582500 |
0.13929614 |
125.327893 |
0.00000000000000000000000000004314310330209581304011462025512251811838776282346124726487845527018016816660477691369734998261264991015195846557617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000031105972654602006590286272155567321014777681077500687040826347863019105859441748584970355295808985829353332519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
524.785213689221 |
50.7482070309022 |
1571.67114636195 |
111.482771869615 |
| ENSMUSG00000026773 |
170768 |
Pfkfb3 |
protein_coding |
A0A0A6YW89 |
Human_homologues FUNCTION: Catalyzes both the synthesis and degradation of fructose 2,6-bisphosphate. {ECO:0000269|PubMed:10077634, ECO:0000269|PubMed:17499765, ECO:0000305|PubMed:16316985}. |
Hydrolase;Kinase;Multifunctional enzyme;Proteomics identification;Reference proteome;Transferase |
|
|
|
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; 6-phosphofructo-2-kinase activity [GO:0003873]; ATP binding [GO:0005524]; fructose-2,6-bisphosphate 2-phosphatase activity [GO:0004331]; fructose 2,6-bisphosphate metabolic process [GO:0006003]; fructose metabolic process [GO:0006000] |
15845370_Inducible 6-phosphofructo-2-kinase expression is essential for the survival of the growing embryo. 18710022_Possible role of PFKFB-3 splice isoform in cell adaptation to hypoxic conditions. 19645723_uPFK-2 regulation is phosphoinositide 3-kinase-Akt-mtor dependent. 19948719_Data show that PFKFB3(+/-) mice gained much less body weight than did wild-type littermates. 20453422_Interleukin 6 enhances glycolysis through expression of the glycolytic enzymes hexokinase 2 and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-3. 20498376_PFKFB3/iPFK2 is critically involved in the anti-diabetic effect of PPARgamma activation 21464136_Cooperation of adenosine with macrophage Toll-4 receptor agonists leads to increased glycolytic flux through the enhanced expression of PFKFB3 gene. 21860432_inducible PFKFB3 is required for increased growth, metabolic activity and is regulated through active JAK2 and STAT5. 22556414_a unique role for PFKFB3/iPFK2 in adipocytes with regard to diet-induced inflammatory responses in both adipose and liver tissues. 22841546_High-fat diet feeding stimulates intestine 6-phosphofructo-2-kinase expression and induces intestine inflammatory response. 23246158_PFKFB3/iPFK2 responds to re-feeding, which in turn stimulates hypothalamic glycolysis and decreases hypothalamic AMPK phosphorylation and alters neuropeptide expression in a pattern that is associated with suppression of food intake. 23911327_Study showed that endothelial cells (ECs)relied on glycolysis rather than on oxidative phosphorylation for ATP production and that loss of the glycolytic activator PFKFB3 in ECs impaired vessel formation. 24700124_PFKFB3 is a promising target for the reduction of endothelial glycolysis and its related pathological angiogenesis 25359860_Shear stress-mediated repression of endothelial cell metabolism via KLF2 and PFKFB3 controls endothelial cell phenotype. 26322680_Data show that inhibition of AMP-Activated kinase (AMPK) or 6-phosphofructo-2-kinase-fructose-2,6-biphosphatase 3 (PFKFB3) results in enhanced cell death in mitosis. 26342081_overexpression of PFKFB3 in HEK293 cells potentiated insulin-dependent phosphorylation of Akt and Akt substrates 27387960_HG significantly increased PFKFB3 promoter transcription activity compared with LG. 27566823_The PFKFB3-driven glycolysis selectively promotes the extrinsic antiviral capacity of macrophages, via metabolically supporting the engulfment and removal of virus-infected cells. 28576491_Blockage of glycolysis by targeting PFKFB3 alleviates sepsis-related acute lung injury via suppressing inflammation and apoptosis of alveolar epithelial cells. 31167111_Targeting PFKFB3-mediated EC glycolysis is an ef fi cient therapeutic strategy for ALI in sepsis. 31213542_mediation of endothelial glycolysis promotes pulmonary hypertension 31406177_Targeting PFKFB3 alleviates cerebral ischemia-reperfusion injury in mice. 31413316_In breast cancer stem cells (BCSCs), there is an inverse relationship between Pfkfb3 expression and autophagy, which reduces Pfkfb3 expression and elicits cellular dormancy. Disrupting autophagy and this event enables Pfkfb3 to drive dormant BCSCs and metastatic lesions to recur. 31880389_Inhibition of PFKFB3 suppresses osteoclastogenesis and prevents ovariectomy-induced bone loss. 31978523_Exercise-induced re-programming of age-related metabolic changes in microglia is accompanied by a reduction in senescent cells. 32169429_CPEB4 Increases Expression of PFKFB3 to Induce Glycolysis and Activate Mouse and Human Hepatic Stellate Cells, Promoting Liver Fibrosis. 32179636_Zhx2 Accelerates Sepsis by Promoting Macrophage Glycolysis via Pfkfb3. 33300142_Mice with a specific deficiency of Pfkfb3 in myeloid cells are protected from hypoxia-induced pulmonary hypertension. 33400016_YAP promotes ocular neovascularization by modifying PFKFB3-driven endothelial glycolysis. 33586458_Sirtuin 3 Alleviates Diabetic Cardiomyopathy by Regulating TIGAR and Cardiomyocyte Metabolism. 34021541_miR-485 inhibits histone deacetylase HDAC5, HIF1alpha and PFKFB3 expression to alleviate epilepsy in cellular and rodent models. 34101614_Disruption of endothelial Pfkfb3 ameliorates diet-induced murine insulin resistance. 34432198_PFKFB3 gene deletion in endothelial cells inhibits intraplaque angiogenesis and lesion formation in a murine model of venous bypass grafting. 34737380_Sex differences in metabolic pathways are regulated by Pfkfb3 and Pdk4 expression in rodent muscle. 34968772_IRF8 and BATF3 interaction enhances the cDC1 specific Pfkfb3 gene expression. 35087090_Aberrant upregulation of the glycolytic enzyme PFKFB3 in CLN7 neuronal ceroid lipofuscinosis. 35830274_Suppression of myeloid PFKFB3-driven glycolysis protects mice from choroidal neovascularization. 35834356_Gene-dosage effect of Pfkfb3 on monocyte/macrophage biology in atherosclerosis. 35916383_Differential role of neuronal glucose and PFKFB3 in memory formation during development. 36243313_PFKFB3 mediates tubular cell death in cisplatin nephrotoxicity by activating CDK4. 36326834_HIF1A-dependent induction of alveolar epithelial PFKFB3 dampens acute lung injury. 36589684_Inhibition of PFKFB Preserves Intestinal Barrier Function in Sepsis by Inhibiting NLRP3/GSDMD. |
ENSG00000170525 |
PFKFB3 |
645.686629 |
2.0799949205 |
1.056580 |
0.11959060 |
77.146331 |
0.00000000000000000158749298678886550876920537600352533279793524536319332743361520954294974217191338539123535156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000561036964611786457048278855948842114649197359427246212248974188696593046188354492187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
858.806078739712 |
71.2713307944449 |
412.888546162673 |
26.3483676304446 |
| ENSMUSG00000026826 |
18227 |
Nr4a2 |
protein_coding |
Q06219 |
FUNCTION: Transcriptional regulator which is important for the differentiation and maintenance of meso-diencephalic dopaminergic (mdDA) neurons during development. It is crucial for expression of a set of genes such as SLC6A3, SLC18A2, TH and DRD2 which are essential for development of mdDA neurons. {ECO:0000269|PubMed:19144721}. |
Alternative splicing;Cytoplasm;DNA-binding;Metal-binding;Nucleus;Receptor;Reference proteome;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:18227; |
chromatin [GO:0000785]; cytoplasm [GO:0005737]; nuclear speck [GO:0016607]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; nuclear glucocorticoid receptor binding [GO:0035259]; nuclear receptor activity [GO:0004879]; protein heterodimerization activity [GO:0046982]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; zinc ion binding [GO:0008270]; adult locomotory behavior [GO:0008344]; cellular response to corticotropin-releasing hormone stimulus [GO:0071376]; cellular response to extracellular stimulus [GO:0031668]; cellular response to oxidative stress [GO:0034599]; central nervous system neuron differentiation [GO:0021953]; central nervous system projection neuron axonogenesis [GO:0021952]; DNA-templated transcription [GO:0006351]; dopamine biosynthetic process [GO:0042416]; dopamine metabolic process [GO:0042417]; dopaminergic neuron differentiation [GO:0071542]; fat cell differentiation [GO:0045444]; gene expression [GO:0010467]; general adaptation syndrome [GO:0051866]; habenula development [GO:0021986]; learning or memory [GO:0007611]; negative regulation of apoptotic signaling pathway [GO:2001234]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of transcription by RNA polymerase II [GO:0000122]; nervous system development [GO:0007399]; neuron apoptotic process [GO:0051402]; neuron differentiation [GO:0030182]; neuron maturation [GO:0042551]; neuron migration [GO:0001764]; positive regulation of catalytic activity [GO:0043085]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; post-embryonic development [GO:0009791]; regulation of DNA-templated transcription [GO:0006355]; regulation of dopamine metabolic process [GO:0042053]; regulation of gene expression [GO:0010468]; regulation of respiratory gaseous exchange [GO:0043576]; regulation of transcription by RNA polymerase II [GO:0006357]; response to amphetamine [GO:0001975]; response to hypoxia [GO:0001666]; response to inorganic substance [GO:0010035]; response to insecticide [GO:0017085]; transcription by RNA polymerase II [GO:0006366] |
12385813_the loss of a single Nurr1 allele results in reduced dopamine levels in mesolimbic and mesocortical pathways and increased locomotor activity in response to mild stress 12401563_Nurr1 differentially regulates and modifies the separate signaling pathways of neuronal cell death in response to dopaminergic neurotoxins 6-hydroxydopamine and methyl phenylpyridinium. 12842300_Deletion of a single allele of the Nurr1 gene alters the locomotor activity of 12- to 15-month-old Nurr1(+/-) animals after acute exposure to methamphetamine. 12915123_Results provide evidence indicating an instructive role for Nurr1 in controlling dopamine synthesis and storage. 14559918_PIASgamma as a transcriptional co-regulator of Nurr1 and suggest that this interaction may have a physiological role in regulating the expression of Nurr1 target genes. 14671317_p57Kip2 functions by a direct protein-protein interaction with Nurr1. 14742729_The prominent Nurr1 expression in these areas, involved in respiratory control, along with the severe respiratory phenotype, indicates that Nurr1 plays a major role in the extrauterine adaption of respiratory control and the response to hypoxia. 14988426_Nurr1 activates the OPN promoter directly in osteoblastic cells and may have a role in the regulation of bone homeostasis 15016073_During in vitro embryonic stem cell differentiation Nurr1 is sufficient to induce and maintain a midbrain-like dopaminergic biochemical and functional cellular phenotype independent of neurogenesis . 15456745_the ligand binding domain of Nurr1 has a co-regulator interaction surface 15485875_Ocn is a Nurr1 target gene, which positions Nurr1 in the core of transcriptional factors regulating osteoblastic gene expression 15589522_Nurr1 plays an important role in the functional maintenance and survival of nigral dopaminergic neurons. The Nurr1+/- mouse is a useful animal model to study the pathogenesis of Parkinson disease and to explore disease-modifying strategies. 15663472_We applied the EphB1 ligand, ephrinB2, to explants of embryonic mouse ventral midbrain. Low concentrations of clustered ephrinB2 (0.25 microg/mL) enhanced Nurr1 mRNA and protein levels 15910281_a second ERK-dependent pathway, independent of MSK and CREB, was also required for the transcription of Nurr1 and Nor1 15910753_The in vivo induction of the NR4A family members by PTH suggests their involvement in, at least some, PTH-induced changes in bone metabolism. 15964844_NR4A nuclear receptors NR4A1, NR4A2, NR4A3 are potential transcriptional mediators of inflammatory signals in activated macrophages 16477036_Nurr1 and Pitx3 cooperatively promote terminal maturation to the midbrain dopamine neuron phenotype in murine and human ES cell cultures. 16638018_Taken together, our findings suggest that Nurr1 might influence the processes of axon guidance and/or angiogenesis via the regulation of Nrp1 expression. 16641236_These results suggest that signaling through D2R in association with Nurr1 using ERK, plays a critical role in mesencephalic dopaminergic neuronal development. 16690213_The Nurr1 +/- genotype is an important factor that renders mice susceptible to isolation-induced disruption of prepulse inhibition and has implications for understanding the regulation of dopamine function. 16741951_Results suggest that Nurr1 is important for osteoblast differentiation. 16782508_Quantitative real-time PCR revealed a significant reduction in tyrosine hydroxylase and GTP cyclohydrolase mRNA in ventral midbrain of Nurr1 +/- mice as compared to wild-type mice (+/+). 16999955_Nurr1 regulation of VIP is demonstrated in vivo as loss of Nurr1 function results in diminished VIP mRNA levels within the developing midbrain. 17038671_These results confirm the presence of both secreted and cell-intrinsic survival signals modulated by Nurr1 and suggest that Nurr1 is a key regulator of both survival and dopaminergic differentiation. 17291494_Over-expression of Nurr1 and Ngn2 in combination generates morphologically mature TH-expressing neurons that also express additional mesencephalic markers. 17394463_In the mouse, Nurr1 and GTPCH mRNA were first detected at E10.5, and GTPCH transcription paralleled that of Nurr1 17457314_Results show that Nurr1 (+/-) mice display a pattern of behaviors indicative of potential relevance for symptoms of schizophrenia combined with a gender-specific abnormal dopamine transmission in the striatum and prefrontal cortex, respectively. 18242186_Rat and mouse species-dependent differences of Nurr1 and Ngn2 actions in dopamine neuron differentiation. 18655117_Nurr1-null heterozygous genotype causes only subtle changes in dopamine neurotransmission, but when combined with social isolation, results in a substantial differential effect on amphetamine-stimulated dopamine overflow in the nucleus accumbens shell. 18809462_FGF-8b induces the expression of Nurr1 orphan nuclear receptors that are involved in mediating the growth promoting effect of FGF-8b in osteoblasts. 18945812_Inhibition of adipocyte differentiation by Nur77, Nurr1, and Nor1. 19074857_the orphan nuclear receptor Nurr1 has a role in inhibiting bladder cancer growth 19144721_Both Nurr1 and Pitx3 interact with the co-repressor PSF and occupy the promoters of Nurr1 target genes in concert. 19246938_Nurr1 is found to be expressed in cultured mouse microglia. The expression of Nurr1 is increased and the protein is translocated from the cytoplasm to the nucleus after activation by LPS, which could be modulated by the ERK, JNK and PI3K/Akt pathways. 19345186_These studies suggest that Nurr1 protects against loss of dopaminergic neurons in Parkinson's disease in part by limiting the production of neurotoxic mediators by microglia and astrocytes. 19465082_The significant induction (between 1 and 4h) of the NR4A genes in multiple tissues, was identified. 19515692_Dlk1 is required to prevent premature expression of Dat in meso-diencephalic dopamine neuronal precursors as part of the multifaceted process of neuronal differentiation driven by Nurr1 and Pitx3. 19549529_Cyclooxygenase-2 inhibitors down-regulate osteopontin and Nr4A2-new therapeutic targets for colorectal cancers. 19906978_these findings begin to define a gene regulatory pathway for habenula development in mammals, and the roles of Nurr1 and Brn3a therein. 19923853_These results together imply that Nurr-1 expression is a connecting point between neuroendocrine and immune systems in mediating cytokine-induced POMC gene expression. 20175204_results suggest that the increase in Nurr1 expression might be a compensatory mechanism to counteract the changes in forebrain dopamine transmission in coping with acute stress 20407282_Corticotropin-releasing factor Crf/CRH promoter contains multiple nuclear factor (NF)-kappaB and Nurr1 binding sites. 20421523_In smoth muscle cells, Nurr1 inhibits proliferation and inflammatory responses, which explains the inhibition of SMC-rich lesion formation in mice 20566846_Data identify NR4A nuclear orphan receptors as essential mediators of CREB-dependent neuroprotection. 20571502_Data show RNA interference that targets Ube3a in P19 cells caused downregulation of Mc1r and Nr4a2, whereas overexpression of Ube3a results in the upregulation of Mc1r and Nr4a2. 20829434_Results propose a MIF-NURR1 signaling axis as a regulator of the glucocorticoid sensitivity of MKP1. 21076412_Overexpression of Nurr1 drives early haematopoietic progenitors into quiescence. 21151937_decreased Nurr1 in heterozygous young adult mice leads to exacerbated acute and long-term toxicity after repeated methamphetamine exposure 21362399_Nurr1 inhibited beta-catenin-mediated expression of Axin2 in MC3T3-E1 cells. 21468021_Ectopic expression of Nr4a2 imparts Treg-like suppressive activity to naive CD4(+) T cells by inducing Foxp3 and by repressing cytokine production. 21531044_Heterozygous Nurr1 knockout influences the age-dependent decline in the number of dopamine (DA) neurons and influences DA signaling. 21545404_heterozygous constitutive deletion of Nurr1 results in a restricted phenotype characteristic of schizophrenia symptomatology, which primarily relates to motor activity, sensorimotor gating and responsiveness to the psychomimetic drug MK-801. 21621845_In response to purified LPS, strong upregulation of Nr4a3, but not of Nr4a1 or Nr4a2 was seen in mast cells 21820432_Prepulse inhibition deficits in Nurr1-deficient mice were paralleled by decreased tyrosine hydroxylase and dopamine transporter immunoreactivity in ventral parts of the striatum. 21979916_NR4A functions directly at DNA repair sites by a process that requires phosphorylation by DNA-PK 22238080_Consequences of prenatal viral-like immune activation are exacerbated in offspring with genetic predisposition to dopaminergic abnormalities induced by mutations in Nurr1. 22296971_Found that an axon genesis gene called Topoisomerase IIbeta (Top IIbeta) was down-regulated in Nurr1 knock-out mice and identified two functional NURR1 binding sites in the proximal Top IIbeta promoter. 22328530_The data provide the first evidence that miR-132 is an important molecule regulating ES cell differentiation into dopamine neurons by directly targeting Nurr1 gene expression. 22363463_There is a direct interaction between Nurr1 and the Pitx3 gene promoter. 22483304_Nurr1 gene activation improved rotarod performance, attenuated DA neuron loss and nigrostriatal DA reduction, increased expression of Nurr1, DAT and VMAT2, and alleviated microglial activation in the substantia nigra of lactacystin-lesioned mice. 22628460_This study demonistrated that Nurr1-positive cells are generated during the 2-day peak of subplate neurogenesis (E11.5-E12.5). survive preferentially after the first postnatal week compared with other subplate neurons. 22748824_Nurr1 is critical for the development and maintenance of midbrain dopaminergic (DA) neurons in mouse. 23161447_The results of this study found that the NR4A2 expressed in hippocampal neurons (associated with object location memory) as well as in the insular and perirhinal cortex (associated with object recognition memory. 23240065_Our results suggest that the plasticity of NPs and their ability to activate a dopaminergic differentiation program in response to Nurr1 is regulated during early stages of neurogenesis, possibly through mechanisms controlling CNS regionalization 23334790_results suggest that Nr4a receptors have key roles in determining CD4(+) T cell fates in the thymus and thus contribute to immune homeostasis 23341612_Nurr1 functions mainly in transcriptional activation to regulate a battery of genes expressed in DA neurons. 23437182_Thus, NR4A2 appears to control Th17 differentiation and so plays an essential role in the development of Th17-mediated autoimmune disease 24005216_NR4A nuclear receptors are involved in negative selection of thymocytes, Treg differentiation and the development of Ly6C monocytes. Nur77 and Nurr1 attenuate atherosclerosis in mice whereas NOR-1 aggravates vascular lesion formation. 24291696_Induction of Nurr1 expression is mediated by voltage-dependent calcium channels. 24496614_The interplay of Nurr1 and Foxa2 is the crucial determinant for dopamine phenotype acquisition during midbrain dopaminergic neuron development. 24753204_This study showed that nurr1 has a similar mRNA expression pattern and low levels of expression at rest. 25156412_Nurr1 protein is prominently expressed in brain areas with Abeta accumulation, that is, the subiculum and the frontal cortex, in the 5XFAD mouse and that Nurr1 is highly co-expressed with Abeta at early stages. 25447275_Results indicate that Nurr1 overexpression in olfactory bulb stem cells induces the formation of two populations of mature dopaminergic neurons with features of the ventral mesencephalon or of the olfactory bulb 25752609_Because the phosphorylation site mutants of NR4A2 cannot rescue the cell death-promoting activity, ASK1-p38 pathway-dependent phosphorylation and subsequent cytoplasmic translocation of NR4A2 may be required for oxidative stress-induced cell death. 25759364_The authors show herein that Nurr1 and Foxa2 interact to protect midbrain dopaminergic neurons against various toxic insults, but their expression is lost during aging and degenerative processes. 25855987_Nurr1 +/- genotype predisposes mice to T. gondii-induced alterations in behaviors that involve dopamine neurotransmission 25899083_Nuclear receptors of the NR4a family are not required for the development and function of follicular T helper cells 26304965_Nr4a2 factor plays crucial role regulatory T cells, mediating the two defining characteristics of regulatory T cells: stability of cell lineage and immunosuppressive functions. 26337347_Nurr1 defect induces early experimental autoimmune encephalomyelitis (EAE) onset and increases inflammatory infiltrates in spinal cord suggesting a Nurr1 role in the early phase of EAE 26873851_Knockdown of DJ-1 attenuates Nurr1 activity. 27184189_Overexpression and knockdown of Nr4a2 demonstrated that Nr4a2 orchestrates the expression of immunoregulatory genes, hence inducing a tolerogenic phenotype in bone marrow derived dendritic cells (DC) in mice. The findings suggest a previously unidentified role for Nr4a2 as a regulator of dendritic cell tolerogenicity and demonstrate its potential as therapeutic target in DC-associated pathophysiologies. 27208834_Overexpression of Nurr1 and Pitx3 in iPSCs could efficiently program induced pluripotent stem cells into functional dopaminergic-like neurons. This finding may have an impact on future stem cell therapy of Parkinson's disease. 27532877_The Nurr1 expression may play a significant role in regulating inflammation in the brain. 28385514_The study comprehensively evaluated the association of NR4A2 variation with PD, and the results failed to demonstrate that the NR4A2 polymorphisms significantly associated with PD except for rs35479735, suggesting that more studies are needed to elucidate if NR4A2 is a risk of PD. 29540663_Nurr1 regulated intestinal epithelial development and barrier function after intestinal ischemia/reperfusion injury. 29634982_Nurr1 suppressed the production of TNF-alpha via interaction with NF-kappaB/p65 and inhibiting its nuclear translocation. In addition, both NF-kappaB and Nurr1 appeared to be regulated by the TLR4-mediated signal pathway. Study demonstrated that TLR4 recognized alpha-synuclein and activated downstream signaling mechanisms leading to the release of pro-inflammatory mediators that are contra-balanced by Nurr1 expression. 30111648_NR4A1/Nur77 and NR4A2/Nurr1 dynamically regulated inflammatory gene expression in glia by modulating the transcriptional activity of NF-kappaB. 30267803_the pathogenesis of cerebral ischemia-reperfusion injury is associated with Nurr1 upregulation followed by augmented mitochondrial fission via an abnormal YAP-INF2 pathways 30365199_Acute stress suppresses but chronic stress conversely induces Nr4a2 expression in hippocampal dentate gyrus granule cells (GCs). The survival of newly generated GCs is impaired by chronic restraint stress, and long-term stress after middle age decreases the proportion of late-born GCs in aged mice. 30515963_Nurr1 critically regulates Alzheimer's disease-related pathophysiology. 30998946_Regulation of NR4A2 function in the medial habenula is critical in relapse-like behaviors. 31110021_Identify NURR1 agonists as possible exercise mimetics with the potential to ameliorate obesity and other metabolic abnormalities. 31127527_PPARalpha is a novel regulator of Nurr1 expression and associated protection of dopaminergic neurons. 31455763_Our study suggests that the NURR1 deficient male mouse may be a satisfactory model to study some ADHD behavioral phenotypes and to test the clinical efficacy of potential therapeutic agents. 31520492_elevated levels of nuclear receptor subfamily 4 group A member 2 (Nurr1) are associated with the pro-inflammatory profile of alpha-synuclein-deficient CD4(+) T cells 31664129_Chloroquine modulates inflammatory autoimmune responses through Nurr1 in autoimmune diseases. 32181533_Nurr1(Cd11bcre) conditional knockout mice display inflammatory injury to nigrostriatal dopaminergic neurons. 32188741_The transcription factor Nurr1 is upregulated in amyotrophic lateral sclerosis patients and SOD1-G93A mice. 32188976_The Transcription Factor NR4A2 Plays an Essential Role in Driving Prolactin Expression in Female Pituitary Lactotropes. 32612143_Nurr1 performs its anti-inflammatory function by regulating RasGRP1 expression in neuro-inflammation. 33750404_Exosomal miR-409-3p secreted from activated mast cells promotes microglial migration, activation and neuroinflammation by targeting Nr4a2 to activate the NF-kappaB pathway. 33902111_Nurr1 repression mediates cardinal features of Parkinson's disease in alpha-synuclein transgenic mice. 33991560_Nurr1 deficiency shortens free running period, enhances photoentrainment to phase advance, and disrupts circadian cycling of the dopamine neuron phenotype. 34119603_Nurr1 downregulation is caused by CREB inactivation in a Parkinson's disease mouse model. 34463540_Aberrant mTOR/autophagy/Nurr1 signaling is critical for TSC-associated tumor development. 35743300_Nurr1 Is Not an Essential Regulator of BDNF in Mouse Cortical Neurons. 35776225_Prolonged cardiac NR4A2 activation causes dilated cardiomyopathy in mice. 35805964_Lmx1a-Dependent Activation of miR-204/211 Controls the Timing of Nurr1-Mediated Dopaminergic Differentiation. |
ENSG00000153234 |
NR4A2 |
351.476538 |
0.3690048094 |
-1.438288 |
0.35981133 |
15.168526 |
0.00009832880333910622627693193242848224144836422055959701538085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005871204752886185779942040952050774649251252412796020507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
198.932957338571 |
50.1302677070503 |
539.106678991046 |
100.793104398678 |
| ENSMUSG00000026873 |
74016 |
Phf19 |
protein_coding |
Q9CXG9 |
FUNCTION: Polycomb group (PcG) protein that specifically binds histone H3 trimethylated at 'Lys-36' (H3K36me3) and recruits the PRC2 complex, thus enhancing PRC2 H3K27me3 methylation activity (PubMed:23104054, PubMed:23160351, PubMed:22438827, PubMed:23273982). Probably involved in the transition from an active state to a repressed state in embryonic stem cells: acts by binding to H3K36me3, a mark for transcriptional activation, and recruiting H3K36me3 histone demethylases RIOX1 or KDM2B, leading to demethylation of H3K36 and recruitment of the PRC2 complex that mediates H3K27me3 methylation, followed by de novo silencing (PubMed:23160351). Recruits the PRC2 complex to CpG islands and contributes to embryonic stem cell self-renewal (PubMed:22438827). Also binds histone H3 dimethylated at 'Lys-36' (H3K36me2) (By similarity). {ECO:0000250|UniProtKB:Q5T6S3, ECO:0000269|PubMed:22438827, ECO:0000269|PubMed:23104054, ECO:0000269|PubMed:23160351, ECO:0000269|PubMed:23273982}. |
Chromatin regulator;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:74016; |
ESC/E(Z) complex [GO:0035098]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; metal ion binding [GO:0046872]; methylated histone binding [GO:0035064]; chromatin organization [GO:0006325]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of histone H3-K27 methylation [GO:0061087]; regulation of DNA-templated transcription [GO:0006355]; stem cell differentiation [GO:0048863]; stem cell population maintenance [GO:0019827] |
31089138_In B16 mouse melanoma model miR-155 enhances Polycomb repressor complex 2 (PRC2) activity indirectly by promoting the expression of the PRC2-associated factor Phf19 through downregulation of the Akt inhibitor, Ship1. Phf19 orchestrates a transcriptional program extensively shared with miR-155 to restrain T cell senescence and sustain CD8+ T cell antitumor responses. 31383640_PHF19 is required for multiple myeloma tumorigenesis in vivo. 32869745_Structural basis for histone variant H3tK27me3 recognition by PHF1 and PHF19. 33611744_PHD Finger Protein 19 Promotes Cardiac Hypertrophy via Epigenetically Regulating SIRT2. |
ENSG00000119403 |
PHF19 |
66.204996 |
0.3099590430 |
-1.689850 |
0.46226314 |
12.835005 |
0.00034019456328056481286825629695158568210899829864501953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0017800905489143124555556640942199919663835316896438598632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
30.3658879690859 |
9.89707140597809 |
97.9674207123222 |
23.1127862406119 |
| ENSMUSG00000026885 |
74410 |
Ttll11 |
protein_coding |
A4Q9F4 |
FUNCTION: Polyglutamylase which modifies tubulin, generating polyglutamate side chains of variable lengths on the gamma-carboxyl group of specific glutamate residues within the C-terminal tail of tubulin (PubMed:17499049, PubMed:20530212). Preferentially mediates ATP-dependent polyglutamate long side-chain elongation over the initiation step of the polyglutamylation reaction (PubMed:17499049, PubMed:20530212). Preferentially modifies the alpha-tubulin tail over a beta-tail (PubMed:17499049). Required for CCSAP localization to both spindle and cilia microtubules (By similarity). Promotes tubulin polyglutamylation which stimulates spastin/SPAST-mediated microtubule severing, thereby regulating microtubule functions (PubMed:20530212). {ECO:0000250|UniProtKB:Q8NHH1, ECO:0000269|PubMed:17499049, ECO:0000269|PubMed:20530212}. |
Alternative splicing;ATP-binding;Cell projection;Cilium;Cytoplasm;Cytoskeleton;Ligase;Magnesium;Metal-binding;Microtubule;Nucleotide-binding;Reference proteome |
|
|
mmu:74410; |
ciliary basal body [GO:0036064]; cilium [GO:0005929]; cytoplasm [GO:0005737]; microtubule [GO:0005874]; ATP binding [GO:0005524]; metal ion binding [GO:0046872]; tubulin binding [GO:0015631]; tubulin-glutamic acid ligase activity [GO:0070740]; microtubule cytoskeleton organization [GO:0000226]; microtubule severing [GO:0051013]; protein polyglutamylation [GO:0018095] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20583170_Observational study of gene-disease association. (HuGE Navigator) 34040021_Genetic variant of TTLL11 gene and subsequent ciliary defects are associated with idiopathic scoliosis in a 5-generation UK family. 36511133_TTLL11 gene is associated with sustained attention performance and brain networks: A genome-wide association study of a healthy Chinese sample. |
ENSG00000175764 |
TTLL11 |
55.216611 |
2.1642333023 |
1.113856 |
0.42655666 |
6.767603 |
0.00928274480706144100583543377069872803986072540283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0314420211032154506280988925936981104314327239990234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
77.6171970290798 |
23.2846322410638 |
35.8635998014312 |
8.2789079264534 |
| ENSMUSG00000026923 |
18128 |
Notch1 |
protein_coding |
Q01705 |
FUNCTION: Functions as a receptor for membrane-bound ligands Jagged-1 (JAG1), Jagged-2 (JAG2) and Delta-1 (DLL1) to regulate cell-fate determination. Upon ligand activation through the released notch intracellular domain (NICD) it forms a transcriptional activator complex with RBPJ/RBPSUH and activates genes of the enhancer of split locus. Affects the implementation of differentiation, proliferation and apoptotic programs. Involved in angiogenesis; negatively regulates endothelial cell proliferation and migration and angiogenic sprouting. Involved in the maturation of both CD4(+) and CD8(+) cells in the thymus. Important for follicular differentiation and possibly cell fate selection within the follicle. During cerebellar development, functions as a receptor for neuronal DNER and is involved in the differentiation of Bergmann glia. Represses neuronal and myogenic differentiation. May play an essential role in postimplantation development, probably in some aspect of cell specification and/or differentiation. May be involved in mesoderm development, somite formation and neurogenesis. May enhance HIF1A function by sequestering HIF1AN away from HIF1A. Required for the THBS4 function in regulating protective astrogenesis from the subventricular zone (SVZ) niche after injury. Involved in determination of left/right symmetry by modulating the balance between motile and immotile (sensory) cilia at the left-right organiser (LRO). {ECO:0000269|PubMed:15965470, ECO:0000269|PubMed:18299578, ECO:0000269|PubMed:23160044, ECO:0000269|PubMed:23615612}. |
3D-structure;Activator;Alternative splicing;Angiogenesis;ANK repeat;Calcium;Cell membrane;Developmental protein;Differentiation;Direct protein sequencing;Disulfide bond;EGF-like domain;Glycoprotein;Hydroxylation;Isopeptide bond;Membrane;Metal-binding;Notch signaling pathway;Nucleus;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transcription;Transcription regulation;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:18128; |
acrosomal vesicle [GO:0001669]; adherens junction [GO:0005912]; apical plasma membrane [GO:0016324]; cell periphery [GO:0071944]; cell surface [GO:0009986]; cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; lamellipodium [GO:0030027]; MAML1-RBP-Jkappa- ICN1 complex [GO:0002193]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; ruffle [GO:0001726]; calcium ion binding [GO:0005509]; chromatin binding [GO:0003682]; chromatin DNA binding [GO:0031490]; enzyme binding [GO:0019899]; enzyme inhibitor activity [GO:0004857]; identical protein binding [GO:0042802]; Notch binding [GO:0005112]; transcription coactivator activity [GO:0003713]; transcription regulator activator activity [GO:0140537]; transmembrane signaling receptor activity [GO:0004888]; animal organ regeneration [GO:0031100]; aortic valve morphogenesis [GO:0003180]; apoptotic process [GO:0006915]; apoptotic process involved in embryonic digit morphogenesis [GO:1902263]; arterial endothelial cell differentiation [GO:0060842]; astrocyte differentiation [GO:0048708]; atrioventricular node development [GO:0003162]; atrioventricular valve morphogenesis [GO:0003181]; auditory receptor cell fate commitment [GO:0009912]; axon guidance [GO:0007411]; axonogenesis [GO:0007409]; branching morphogenesis of an epithelial tube [GO:0048754]; calcium-ion regulated exocytosis [GO:0017156]; cardiac atrium morphogenesis [GO:0003209]; cardiac chamber formation [GO:0003207]; cardiac epithelial to mesenchymal transition [GO:0060317]; cardiac left ventricle morphogenesis [GO:0003214]; cardiac muscle cell myoblast differentiation [GO:0060379]; cardiac muscle cell proliferation [GO:0060038]; cardiac muscle tissue morphogenesis [GO:0055008]; cardiac right atrium morphogenesis [GO:0003213]; cardiac right ventricle formation [GO:0003219]; cardiac septum morphogenesis [GO:0060411]; cardiac vascular smooth muscle cell development [GO:0060948]; cardiac ventricle morphogenesis [GO:0003208]; cell differentiation [GO:0030154]; cell differentiation in spinal cord [GO:0021515]; cell migration involved in endocardial cushion formation [GO:0003273]; cell population proliferation [GO:0008283]; cellular response to follicle-stimulating hormone stimulus [GO:0071372]; cellular response to hypoxia [GO:0071456]; cellular response to tumor cell [GO:0071228]; cellular response to vascular endothelial growth factor stimulus [GO:0035924]; cilium assembly [GO:0060271]; collecting duct development [GO:0072044]; compartment pattern specification [GO:0007386]; coronary artery morphogenesis [GO:0060982]; coronary sinus valve morphogenesis [GO:0003182]; coronary vein morphogenesis [GO:0003169]; determination of left/right symmetry [GO:0007368]; distal tubule development [GO:0072017]; embryonic hindlimb morphogenesis [GO:0035116]; embryonic limb morphogenesis [GO:0030326]; endocardial cell differentiation [GO:0060956]; endocardial cushion development [GO:0003197]; endocardial cushion morphogenesis [GO:0003203]; endocardium development [GO:0003157]; endocardium morphogenesis [GO:0003160]; endoderm development [GO:0007492]; epidermal cell fate specification [GO:0009957]; epidermis development [GO:0008544]; epithelial cell fate commitment [GO:0072148]; epithelial cell proliferation [GO:0050673]; epithelial to mesenchymal transition [GO:0001837]; epithelial to mesenchymal transition involved in endocardial cushion formation [GO:0003198]; forebrain development [GO:0030900]; foregut morphogenesis [GO:0007440]; gene expression [GO:0010467]; glial cell differentiation [GO:0010001]; glomerular mesangial cell development [GO:0072144]; growth involved in heart morphogenesis [GO:0003241]; hair follicle morphogenesis [GO:0031069]; heart development [GO:0007507]; heart looping [GO:0001947]; heart trabecula morphogenesis [GO:0061384]; homeostasis of number of cells within a tissue [GO:0048873]; humoral immune response [GO:0006959]; in utero embryonic development [GO:0001701]; inflammatory response to antigenic stimulus [GO:0002437]; inhibition of neuroepithelial cell differentiation [GO:0002085]; interleukin-17-mediated signaling pathway [GO:0097400]; keratinocyte differentiation [GO:0030216]; left/right axis specification [GO:0070986]; liver development [GO:0001889]; lung development [GO:0030324]; mesenchymal cell development [GO:0014031]; mitral valve formation [GO:0003192]; negative regulation of anoikis [GO:2000811]; negative regulation of biomineral tissue development [GO:0070168]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of calcium ion-dependent exocytosis [GO:0045955]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cardiac muscle hypertrophy [GO:0010614]; negative regulation of catalytic activity [GO:0043086]; negative regulation of cell adhesion molecule production [GO:0060354]; negative regulation of cell death [GO:0060548]; negative regulation of cell migration involved in sprouting angiogenesis [GO:0090051]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cell proliferation involved in heart valve morphogenesis [GO:0003252]; negative regulation of cell-cell adhesion mediated by cadherin [GO:2000048]; negative regulation of cell-substrate adhesion [GO:0010812]; negative regulation of cold-induced thermogenesis [GO:0120163]; negative regulation of collagen biosynthetic process [GO:0032966]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of endothelial cell chemotaxis [GO:2001027]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of extracellular matrix constituent secretion [GO:0003332]; negative regulation of gene expression [GO:0010629]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of inner ear auditory receptor cell differentiation [GO:0045608]; negative regulation of myoblast differentiation [GO:0045662]; negative regulation of myotube differentiation [GO:0010832]; negative regulation of neurogenesis [GO:0050768]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of oligodendrocyte differentiation [GO:0048715]; negative regulation of ossification [GO:0030279]; negative regulation of osteoblast differentiation [GO:0045668]; negative regulation of photoreceptor cell differentiation [GO:0046533]; negative regulation of pro-B cell differentiation [GO:2000974]; negative regulation of stem cell differentiation [GO:2000737]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural tube development [GO:0021915]; neuroendocrine cell differentiation [GO:0061101]; neuron differentiation [GO:0030182]; neuron fate commitment [GO:0048663]; neuronal stem cell population maintenance [GO:0097150]; Notch signaling involved in heart development [GO:0061314]; Notch signaling pathway [GO:0007219]; Notch signaling pathway involved in regulation of secondary heart field cardioblast proliferation [GO:0003270]; oligodendrocyte differentiation [GO:0048709]; osteoblast fate commitment [GO:0002051]; outflow tract morphogenesis [GO:0003151]; pericardium morphogenesis [GO:0003344]; positive regulation of aorta morphogenesis [GO:1903849]; positive regulation of apoptotic process [GO:0043065]; positive regulation of apoptotic process involved in morphogenesis [GO:1902339]; positive regulation of astrocyte differentiation [GO:0048711]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of cardiac epithelial to mesenchymal transition [GO:0062043]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of endothelial cell differentiation [GO:0045603]; positive regulation of ephrin receptor signaling pathway [GO:1901189]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of epithelial to mesenchymal transition [GO:0010718]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of glial cell differentiation [GO:0045687]; positive regulation of keratinocyte differentiation [GO:0045618]; positive regulation of neuroblast proliferation [GO:0002052]; positive regulation of Notch signaling pathway [GO:0045747]; positive regulation of Ras protein signal transduction [GO:0046579]; positive regulation of receptor signaling pathway via JAK-STAT [GO:0046427]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of transcription of Notch receptor target [GO:0007221]; positive regulation of viral genome replication [GO:0045070]; positive regulation of viral transcription [GO:0050434]; prostate gland epithelium morphogenesis [GO:0060740]; protein catabolic process [GO:0030163]; protein import into nucleus [GO:0006606]; pulmonary valve morphogenesis [GO:0003184]; regulation of cardioblast proliferation [GO:0003264]; regulation of cell adhesion involved in heart morphogenesis [GO:0061344]; regulation of cell migration [GO:0030334]; regulation of cell population proliferation [GO:0042127]; regulation of epithelial cell proliferation [GO:0050678]; regulation of epithelial cell proliferation involved in prostate gland development [GO:0060768]; regulation of extracellular matrix assembly [GO:1901201]; regulation of gene expression [GO:0010468]; regulation of inner ear auditory receptor cell differentiation [GO:0045607]; regulation of neurogenesis [GO:0050767]; regulation of Notch signaling pathway [GO:0008593]; regulation of somitogenesis [GO:0014807]; regulation of stem cell proliferation [GO:0072091]; regulation of transcription by RNA polymerase II [GO:0006357]; response to corticosteroid [GO:0031960]; response to lipopolysaccharide [GO:0032496]; response to muramyl dipeptide [GO:0032495]; retinal cone cell differentiation [GO:0042670]; secretory columnal luminar epithelial cell differentiation involved in prostate glandular acinus development [GO:0060528]; skeletal muscle cell differentiation [GO:0035914]; somatic stem cell division [GO:0048103]; spermatogenesis [GO:0007283]; sprouting angiogenesis [GO:0002040]; T-helper 17 type immune response [GO:0072538]; tissue regeneration [GO:0042246]; transcription by RNA polymerase II [GO:0006366]; tube formation [GO:0035148]; vasculogenesis involved in coronary vascular morphogenesis [GO:0060979]; venous blood vessel morphogenesis [GO:0048845]; venous endothelial cell differentiation [GO:0060843]; ventricular septum morphogenesis [GO:0060412]; ventricular trabecula myocardium morphogenesis [GO:0003222] |
11713346_Expression data suggest a role for Notch activity in establishing definitive hematopoiesis in fetal liver, as well as a selective use of Notch signaling in adult erythropoiesis and granulopoiesis. 11756438_These data reveal that a novel PS1/2-independent mechanism plays a partial role in Notch signal transduction 11777909_regulatory mechanism down-regulating Notch1 protein levels already at the cellular surface, possibly with consequences for Notch-dependent signal transduction during terminal differentiation processes. 11811553_osteoblastic cell differentiation is regulated positively by Notch1 11813235_a deficiency in Notch1 expression leads to increased abundance of products of specific myelin genes in myelinated areas of the brain during the first 2 weeks of postnatal life and up-regulation of Mash1 and down-regulation of Hes5 proteins 11823505_role of Notch1 in hemopoiesis 11895769_increases hematopoietic stem cell self-reneewal and favors lymphoid over myeloid lineage outcome. 11937492_Notch pathway molecules are essential for the maintenance, but not the generation, of mammalian neural stem cells 12032823_Overexpression of the Notch target genes Hes in vivo induces lymphoid and myeloid alterations 12050162_association with nephroblastoma overexpressed gene 12121668_Notch1 controls several functions necessary for alpha beta lineage thymocyte development 12123574_studies reveal that GSK3beta modulates Notch1 signaling, possibly through direct phosphorylation of the intracellular domain of Notch, and that the activity of GSK3beta protects the intracellular domain from proteasome degradation 12165504_Data from two different Notch1IC transgenic lines indicate that Notch activity promotes CD8 and inhibits CD4 SP development. 12175503_distribution of Notch1 and its putative ligands suggest distinct roles in specific subsets of cells in the postnatal brain including putative stem cells and differentiated neurons 12185362_Oncogenic Ras increases levels and activity of the intracellular form of wild-type Notch-1, and upregulates Notch ligand Delta-1 and also presenilin-1, a protein involved in Notch processing, through a p38-mediated pathway. 12186854_Notch1 is required for the correct spatial and temporal regulation of oligodendrocyte differentiation in various regions of the central nervous system. 12208538_study suggests complex specific relationships between Notch signaling, Math1 and Hes1/Hes5 in the control of hair cell differentiation in the developing organ of Corti 12242716_Notch1/Jagged1 have multiple potential paths of cell-cell signaling during mouse lung development and vascular morphogenesis 12374741_Following ectodomain shedding, Notch-1 undergoes presenilin (PS)-dependent constitutive intramembranous endoproteolysis at site-3. 12374742_SHARP is a novel component of the HDAC corepressor complex, recruited by RBP-Jkappa to repress transcription of target genes in the absence of activated Notch. 12391150_Data describe the role for p300 in the potentiation of Notch-1 and -3 IC function by MAML1 and PCAF. 12411305_Notch1-transfected stromal cells showed increased expression of RANKL and OPG genes, and strong inhibition of M-CSF gene expression, resulting in reduction of their ability to support osteoclast development 12459176_Results suggest that the presence of valine at the N-terminus of the Notch1 intracellular domain cleavage product is important for its stability. 12479824_Pax5 interfered with T lineage commitment and early thymocyte development by repressing the transcription of the T cell specification gene Notch1 12486116_modification of O-fucose by fringe protein at epidermal growth factor-like repeats within the ligand-binding site and the Abruptex region 12509463_Notch1 is required for transcriptional coactivator recruitment and cell growth in tumor cell lines 12538689_Mechanisms regulating Notch1 activation in lymphoid precursors at extrathymic sites and in the thymus have been analyzed, including stages representing the first wave of embryonic thymus colonization on embryonic day 12 of gestation. 12590261_Notch1 functions as a tumor-suppressor gene in mammalian skin. 12629177_Glycoprotein 130 signaling regulates Notch1 expression and activation in the self-renewal of mammalian forebrain neural stem cells 12730124_Notch signaling is essential for generation of left-right asymmetry. 12734340_The intracellular region of Notch1 can induce T cells expressing T cell receptors on the cell surface in the absence of a thymic environment but in a B cell-specific culture condition. 12759431_Notch1 and hemopoietic cytokines act together to direct hemopoietic stem cells to differentiate along the myeloid lineage; in addition, activated Notch1 directly increases PU.1 RNA levels, leading to a high concentration of PU.1 protein. 12807718_Radiation-induced deletions in the 5' end region of Notch1 lead to the formation of truncated proteins and are involved in the development of mouse thymic lymphomas 12815466_Transduced Notch1-ICT did not affect cell cycle progression, cell growth or surface IgM levels in NYC cells and resulted only in a slight induction of apoptosis. 12829602_regulation of Notch1 activity at defined developmental windows is essential to control alphabeta versus gammadelta T-cell development and to avoid deregulated expansion of alphabeta-lineage cells. 12842085_Notch mediates TGF alpha-induced changes in epithelial differentiation during pancreatic tumorigenesis 12907456_Notch1 and Notch2 signaling prevents differentiation of mature myeloid cells, resulting in accumulation of precursors readily able to differentiate into mature DCs once the Notch signal is stopped, and in the presence of signals provided by IL-4. 12928374_A recombinase-activating gene (RAG)-deficient model with anti-CD3 providing the pre-TCR signal demonstrates that enforced expression of a Notch1 transgene augments thymocyte double-negative to double-positive transition. 12960086_NotchI impairs osteoblast differentiation and enhances adipogenesis in stromal cell cultures. 12960155_Notch activation directly engages gamma-secretase and subsequently leads to diminished PS1 expression, suggesting a complex set of feedback interactions following Notch activation 12960312_An important role for Notch signaling is suggested during CD4+ T cell responses, which operates through augmenting a positive feedback loop involving IL-2 and its high affinity receptor CD25. 14592976_Expression of the intracellular domain of Notch1 (N1-IC) enhances the association of E47 with the SCF(Skp2) E3 ubiquitin ligase and ubiquitination of E47, followed by proteasome-mediated degradation. 14597207_activated Notch1 plays a role on tissue homeostasis in the nail. 14645852_Notch signaling is a key determinant of muscle regenerative potential that declines with age 14651921_FGF10 signaling serves to integrate cell growth and terminal differentiation at the level of Notch activation. 14657333_Notch1C has roles in multiple steps of pancreatic differentiation 14988227_Expression of activated Notch1 or Notch4 in endothelial cells causes transdifferentiation to a mesenchymal phenotype. Notch 1 is expressed in the ventricular outflow tract during stages of endocardial cushion formation. 15028713_Notch1 has a role in prostatic branching morphogenesis during development and re-growth following castration and androgen replacement 15068794_Notch signaling, which promotes keratinocyte differentiation, is upregulated in embryonic keratinocyte and epidermis. 15076712_Observation of Notch1 in the developing mesenchyme lateral to the olfactory placode, gradually disappearing from the olfactory epithelium, suggests a distinct role for Notch1 in the neurogenesis of the peripheral olfactory system. 15082708_NB-3/Notch1 pathway promotes neural progenitor cell differentiation into oligodendrocytes via Deltex1. 15162428_Notch1 expression on T cells is not required for CD4+ T helper differentiation. 15187027_a dominant negative mutant of MAML1 resulted in early inhibition of T-cell development and the appearance of intrathymic B cells, phenotypes consistent with Notch1 inhibition 15198674_We mapped the active state of the Notch1 signaling pathway in situ in the developing mouse brain. Activated Notch1 was mainly detected in nuclei of a subpopulation of radial glial cells and dividing cells in the ventricular zone 15226394_Notch signaling forms a network of transcription factors and has a role in cholangiocyte differentiation 15237212_localization pattern of c-kit, Notch-1 and GFRalpha-1 in postnatal, immature and adult testes 15251982_Notch1 is required, in a cell-autonomous manner, for the establishment of long-term, definitive hematopoietic stem cells (HSCs). 15280211_Notch has a role in inhibiting Ptf1 function and acinar cell differentiation 15356142_Notch1 signaling induces efficient in vitro differentiation of ex vivo sorted Pax5-deficient pro-B cells into alphabeta and gammadelta T cells. 15448134_ligand-induced reversal of controlled TMD dimerization by the Notch extracellular domain is unlikely to underlie the regulatory mechanism of intramembranous cleavage 15485896_two contiguous regions of noncovalently associated extracellular impose crucial restraints that prevent premature Notch receptor activation 15496460_NGF controls dendrite development in hippocampal neurons by binding to p75NTR and modulating the cellular targets of Notch 15509736_This study suggests that telencephalic progenitors with radial glial morphology are maintained by interactions between the Notch and FGF pathways. 15531372_Notch1 promotes survival of neural precursor cells. 15563463_Notch1 inhibits the development of erythroid/megakaryocytic cells by suppressing GATA-1 activity through HES1 15574878_Fringe glycosyltransferases differentially modulate Notch1 proteolysis induced by Delta1 and Jagged1. 15625786_These results suggest that testosterone affected the expression of Notch family members in activated macrophages. 15634680_Notch signaling represses Ssmooth musscle cell differentiation and maintenance of the contractile phenotype. 15640354_PrPSc in neurons and in ScN2a cells activates Notch-1 cleavage, resulting in atrophy of dendrites in the CNS and shrinkage of processes on the surface of cultured cells 15652348_dlk functionally interacts in vivo with Notch1, which may lead to the regulation of differentiation processes modulated by Notch1 activation and signaling 15689374_activation of Gata2 expression by Notch1/RBPjkappa is a crucial event for the onset of definitive hematopoiesis in the embryo 15705961_Inhibition of recombinant mouse Notch-1 potentiates cyclic-strain-induced changes in rat vascular smooth muscle cell apoptosis and proliferation. 15792957_show that the intracellular domain of Notch1, transcription factor Ets-1, and homeo box D10 (HOXD10).pre B-cell leukemia transcription factor 1(PBX1b).PREP1 activate the rat renin promoter cooperatively 15809373_identify the endothelium as the primary target tissue affected by Notch1 signaling 15866158_interconnection exists between Notch 1 and Calcineurin-NFAT pathways in keratinocyte growth/differentiation control 15866159_multiple roles for Notch signaling during segmentation 15875024_COUP-TFII has a critical role in repressing Notch signalling to maintain vein identity, which suggests that vein identity is under genetic control and is not derived by a default pathway 15887117_direct link between the inflammatory mediator iNOS and Notch signaling, having implications for the development and progression of cholangiocarcinoma. 15902259_developmental protein 'wavefront' is generated by suppression of Notch activity by mesoderm posterior 2 (Mesp2) through induction of the lunatic fringe gene (Lfng) 15939383_analysis of Notch1, Notch4 and Jagged1 expression in ovarian vessels during folliculogenesis and corpus luteum formation 15944289_Expression of the intracellular domain of mouse Notch1 in the transgenic rat thymus leads to sustained expression of the pre-T cell receptor (TCR) throughout thymocyte development, accompanied by a reduction of alpha beta TCR complexes. 15951812_Notch1 signals are required at sequential early stages of intrathymic T cell development. 15951813_Notch signaling controls the generation and ifferentiation of early T lineage progenitors. 15959516_Notch activity is required for the maintenance of proliferating crypt cells in the intestinal epithelium 15964998_p21 acts as a selective negative regulator of transcription and links the Notch1 and Wnt4 signaling pathways in keratinocyte growth control. 15965470_DNER mediates neuron-glia interaction and promotes morphological differentiation of Bergmann glia through Deltex-dependent Notch signaling. 15978571_Notch1 is essential for postnatal hair follicle development and homeostasis. 15991363_Notch1 can directly regulate Tbx21 through complexes formed on the Tbx21 promoter. 15994302_the most highly conserved O-fucose sites in Notch1 are important for both processing and ligand-mediated signaling in the context of a cell-based signaling assay 15998794_SKP2 serves to link Notch1 activation with the cell cycle machinery 16025100_NOTCH1 mutations cause an early developmental defect in the aortic valve and a later de-repression of calcium deposition that causes progressive aortic valve disease 16107537_regulates adult intestinal development by controlling the balance between secretory and absorptive cell types 16169548_both Notch1 and 2 are required in the ectoderm to receive the Jagged2 signal; Notch receives this signal at an early stage in the developmental process and memory of this event is retained by the mesenchyme 16175584_These data combined with data showing interaction between mN1-TM and APP(751) suggest that their transmebrane domains and short sequences around them are sufficient for the interaction and that APP(751) and mN1 interact in cis. 16221173_Notch 1 is activated in the comma-shaped and S-shaped bodies in kidney embryology 16245338_Notch signalling, involving Notch1 and Jagged2 is required to regulate the number of Fgf8-expressing cells that comprise the apical ectodermal ridge, and this is important for the freeing of the digits during normal limb formation. 16282337_Notch1 mutations are very frequent events that are acquired relatively early in the process of leukemic transformation and are important for leukemic cell growth 16319921_Notch1 (N1IC) directly interacts with NF-kappaB and competes with IkappaBalpha, leading to retention of NF-kappaB in the nucleus. 16332675_Notch, epidermal growth factor receptor, and beta1-integrin pathways are coordinated in neural stem cells 16354692_Notch activation occurs early in tumorigenesis and may precede transformation, as ectopic expression of the Notch targets Hes-1 and Deltex-1 is detected in thymocytes from young Ik(L/L) mice with no overt signs of transformation. 16360140_Notch 1 signaling is critical for normal cell proliferation and differentiation in the prostate, and deregulation of this pathway may facilitate prostatic tumorigenesis. 16365048_Notch protein binding to Hes5-GFP is extinguished fast and recovered slowly, whereas Hes1-GFP is inhibited late and recovered quickly 16407293_Overexpression of Notch 1 intracellular domain prevents bone morphogenetic protein (BMP)-2 and wingless/Wnt biological effects by suppressing Wnt but not BMP signaling. 16434391_release of Abeta- or Nbeta-like peptides is a common feature of the proteolysis during regulated intramembrane proteolysis signaling 16452096_Ablation of Notch1 in early progenitor cells led to enhanced cone photoreceptor production, and ablation of Notch1 at later points led to an almost exclusive production of rod photoreceptor cells. 16467260_We observed that an increase in Numb expression is concomitant with Notch degradation in these cells 16507912_Notch1 plays role in the development and transformation of the mammary gland. 16508302_Regardless, these findings demonstrate that PS1, and presumably Notch signaling, is required to maintain all NSCs. 16508303_Neural progenitor cells, as well as differentiating neurons, undergo apoptosis in the absence of Notch1 and Notch3, suggesting that Notch activity promotes the survival of both progenitors and newly differentiating cells in the developing nervous system. 16508306_Expression of Notch1 in the adult SVZ occurs in PSA-NCAM-positive neural precursors and in GFAP-positive SVZ astrocytes. Notch1 was also found in PSA-NCAM-positive neuroblasts located within the rostral migratory stream. 16508308_These results provide support for a role of Notch1 in neuronal subtype specification in the ventral spinal cord. 16510501_Notch1 functions to suppress cone-photoreceptor fate specification in the developing retina. 16518823_Expressed in cells retained within the lamina propria. 16539652_Conversely, activation of either Notch or p75NTR depressed Neurogenin 3 expression, which not only decreased the number of dendrites but also favoured inhibitory (GABAergic) synaptogenesis. 16543950_Integration of the TGF-beta and Notch1 pathways may be an important mechanism for the maintenance of immune homeostasis in the periphery. 16554359_Activation of Nocth1 signaling in cardiogenic mesoderm induces abnormal heart morphogenesis. 16554461_Notch1 regulates radial glia formation through two distinct transcriptional mechanisms and that the outcomes of Notch1 signaling may depend on the relative expression levels of its coregulators. 16607638_Jag2-Notch1 signaling is spatiotemporally regulated in the oral epithelia during palate development to prevent premature palatal shelf adhesion to other oral tissues and to facilitate normal adhesion between the elevated palatal shelves. 16622004_Notch up-regulation and subsequent signaling following macrophage activation modulate gene expression patterns known to affect the function of mature macrophages 16680150_Data suggest that Notch endangers neurons by modulating pathways that increase their vulnerability to apoptosis, and by activating microglial cells and stimulating the infiltration of proinflammatory leukocytes. 16699526_The ability of Lfng and Notch1 to control progenitor competition for limiting cortical niches is an important mechanism for the homeostatic regulation of thymus size 16736472_Results suggest that Notch1 activation may initially demarcate a prosensory region in the cochlear epithelium and then inhibit progenitor cells from becoming hair cells via classical 'lateral inhibition.' 16867989_a RBP-Jkappa-dependent pathway has a role in inducing jagged1-selective notch signaling induces smooth muscle differentiation 16870249_Analysis revealed Notch1 activating mutations in 12 of 17 Plstc/+ lymphomas (70%), analogous to those in human T-ALL. Mice acquired Notch1 mutations in lymph nodes as early as 7 weeks. 16880393_in T cell precursors, Notch/Delta signaling normally acts to modulate and channel PU.1 transcriptional activities during the stages from T lineage specification until commitment 16888092_Notch1 signaling may modulate both expression and transcriptional activity of Runx1 16954387_Results identify c-myc as a novel, direct, and critical Notch1 target gene in T-cell leukemia. 16966428_The requirement for Notch signaling at the beta-selection checkpoint in vivo is absolute and independent of the pre-T cell receptor. 17015687_Activated T cells isolated on the basis of Notch expression (Notch+) are enriched for Bcl-xL expression and demonstrate reduced susceptibility to apoptosis triggered by neglect or oxidative stress 17028268_These observations provide further support for a role for Notch-1 signaling in regulating pulmonary neuroendocrine cell differentiation. 17070841_These results support an emerging molecular mechanism for the displacement of co-repressors from DNA-bound CSL by intracellular domain of the Notch receptor . 17080428_Notch1 contribute to the maintenance of melanoblasts and melanocyte stem cells, and are essential for proper hair pigmentation. 17123719_This study suggested that Notch1 plays a role in the differentiation of VRNs during development and regeneration of VRNs after removal of AOB. 17158336_Phosphatidylinositol 3-kinase/Akt pathway mediates gamma-secretase and Notch1 activation by VEGF, and Notch1 is critical for VEGF-induced postnatal angiogenesis. Possible therapeutic target for improving angiogenesis and blood flow in ischemic limbs. 17161657_Study provides a detailed analysis of the distribution of active Notch1 in relation to its DSL (Delta, Serrate, Lag2) ligands, Jagged1, Delta-like1, and Delta-like4, during progressive stages of vascular morphogenesis and maturation. 17215306_novel roles for Notch1 signaling were identified in heart, vasculature, retina and in the stem cell compartments of self-renewing epithelia 17229764_establish distinct (non-redundant), instructive roles for Notch1 and Notch2 in nephron segmentation. 17242367_Mint deficiency impairs differentiation of early T progenitors into double-negative cells, suggesting that Notch/RBP-J signaling negatively regulates DN1-DN2 transition 17259973_delta-like 4 (Dll4)-Notch1 signalling regulates the formation of appropriate numbers of tip cells to control vessel sprouting and branching in the mouse retina 17296795_Based on our findings, we propose that mechanical forces generated during DSL ligand endocytosis function to physically dissociate hNotch, and that dissociation is a necessary step in Notch activation. 17306789_appropriate suppression of Notch signaling by Mesp1 and Mesp2 factors is essential for stripe pattern formation leading to segment boundary formation 17311993_the Dll4-Notch pathway has a role in vascular proliferation and proper maturation 17318174_Notch inhibits apoptosis by direct interference with XIAP ubiquitination and degradation. 17319963_Our results provide a mechanism by which a balance between neural induction and maintenance of neural progenitors is achieved in the rhombic lip throughout embryonic development. 17320900_Dlk1 may potentiate or inhibit adipogenesis depending upon the cellular context, and that Notch1 expression and activation are important factors in this context. 17336907_We suggest that during trabeculation Notch independently regulates cardiomyocyte proliferation and differentiation, two exquisitely balanced processes whose perturbation may result in congenital heart disease. 17368826_Notch1 intracellular domain plays the role of a negative regulator in AICD signaling via the disruption of the AICD-Fe65-Tip60 trimeric complex. 17440974_double transgenic IC-Notch1/pCX-Cre embryos have widespread expression of IC-Notch1 and die before 10.5 days of gestation, with growth arrest and developmental defects, including lack of neural tube closure, disorganized somites, and disrupted vasculature 17464091_Global gene expression profiling of day 8 hematopoietic progenitors in the absence and presence of activated Notch yield candidate genes required for normal hematopoietic differentiation. 17476283_Notch regulates erythroid homeostasis by inducing apoptosis; sole expression of the activated Notch1 protein is sufficient to induce apoptosis in hexametilene-bisacetamide-differentiating murine erythroleukemia cells. 17526737_These results suggest that ILK down-regulated the protein stability of Notch1-IC through the ubiquitin-proteasome pathway by means of Fbw7. 17585052_Notch1 co-opts lymphoid enhancer factor 1 during the process of transformation to maintain survival of T-cell lymphomas 17599070_PDK1 is essential for Notch-mediated trophic and proliferative responses in thymocytes. 17605079_these results further support a role for Notch signaling in the regulation of olfactory neurogenesis and cell diversity 17611229_Notch1 participates in a bi-compartmental signaling network that controls homeostasis, follicular proliferation rates and melanocyte population within the skin. 17636029_Tip60 protein acts as a negative regulator of Notch1 signaling by means of acetylation. 17664272_Notch1 ligand, Delta-like ligand-4, stimulates R-Ras-dependent alpha 5 beta 1 integrin-mediated adhesion, demonstrating the physiological relevance of this pathway. 17681135_Notch1 signaling is linked to vitamin A metabolism by regulating the expression of cellular retinol binding protein 1 (CRBP1), required to generate a pool of intracellular retinol. 17681139_Hes7 oscillation is initiated by Fgf signaling and propagated/maintained anteriorly by Notch signaling. 17685488_Notch1 transcription and activity was severely reduced in zebrafish and mouse Notch pathway mutants, suggesting that vertebrate Notch1 expression is regulated by a positive feedback loop 17698635_A functional link between SCL and NOTCH1 during SCL T leukemogenesis. 17705199_the Notch1 pathway plays an important role in the control of inflammatory reactions 17717603_Notch/forkhead box O1 cooperation may integrate environmental cues through Notch with metabolic cues through Foxo1 to regulate progenitor cell maintenance and differentiation 17728344_Foxn4 induces expression of delta-like 4 (Dll4) and Mash1 (Ascl1). Dll4 then signals through Notch1 to subdivide the p2 progenitor pool in the ventral spinal cord. 17849441_study shows that genetic interactions between Notch1 and Dll3 result in vertebral segmental defects similar to those seen in congenital scoliosis; craniofacial anomalies not previously observed in Dll3 homozygous animals were identified 17878313_Delta-like-1 engagement of Notch1 promotes differentiation of B lymphocytes to antibody-secreting cells 17915208_The Notch-RBP-J signaling plays crucial roles in the monolayer formation and morphogenesis of Bergmann glia. 17948123_Data demonstrate that Notch1 and VEGFR-3 interact genetically, that Notch directly induces VEGFR-3 in blood endothelial cells to regulate vascular development, and that Notch and VEGF signaling may function in tumor lymphangiogenesis. 17981791_study demonstrates that constitutive Notch signalling can bypass beta-selection during early T-cell development by inducing pre-TCR-like signals within a T-cell-promoting environment. 18024259_Results suggest that Notch1 signaling plays an essential role for mandibular condylar cartilage morphogenesis and development. 18048645_These results suggest that to ensure an appropriate outcome in the thymus in response to a lower threshold of induced Notch signaling, induction of the additional target Hes5 is required. 18049480_data suggest that Notch signaling triggers two distinct pathways, a p53-dependent one leading to a block in proliferation and a p53-independent one promoting differentiation. 18077357_These results provide evidence for the expansive prevalence of Notch signaling in hippocampal morphogenesis and plasticity, suggesting that Notch1 could be a target of diverse traumatic and environmental modulators of adult neurogenesis. 18156632_NOTCH1 acts as a net inhibitor of bone resorption, exerting its effect both directly in osteoclast precursors and indirectly via osteoblast lineage cells 18180377_The results suggested that deregulated constitutive Rap1 activation caused abnormal expansion of DP thymocytes, bypassing the pre-T-cell receptor and eventually leading to Notch1 mutations and Notch-dependent T-cell acute lymphoblastic leukemia. 18193087_Overexpression/enhanced kinase activity of BCR/ABL and altered expression of Notch1 induced acute leukemia in p210BCR/ABL transgenic mice 18197263_finds no evidence that adipose-derived adult stromal cells are of neural crest origin, that they can undergo robust differentiation into functional neurons, and that Notch 1 is likely to be a key regulator of cellular and molecular characteristics 18227520_The O-fucose glycan in EGF12 of mouse Notch1 is required for optimal Notch1 signaling and T cell development in mammals. 18242220_Inhibition of Notch signaling in vivo leads to impaired regeneration of the exocrine pancreas after acute pancreatitis. 18287091_Ikaros directly represses Hes1 in concert with the transcriptional repressor, RBP-Jkappa, allowing for cross-talk between Notch and Ikaros that impacts regulation of CD4 expression 18292500_Notch1 signaling facilitates transforming growth factor-beta-mediated effector function of regulatory T cells. 18297083_Notch signaling in bone marrow normally acts to maintain a pool of mesenchymal progenitors by suppressing osteoblast differentiation. 18299190_different downstream pathways of the Notch receptor participate in the gliogenic and chondrogenic specification of mouse mesencephalic neural crest cells 18299578_FIH-1-dependent hydroxylation of N1945 and N2012 in the Notch1-4 ICD; FIH-1 negatively regulates Notch ICD and accelerates myogenic Differentiation 18311147_activation of the Notch pathway, which is critical in glomerular patterning, contributes to the development of glomerular disease 18332140_These results implicate NOTCH1 signaling during inducible expression of SOCS3 following infection of macrophages with an intracellular bacillus-like M. bovis BCG. 18347015_Pofut1 and O-fucose have roles in mammalian Notch signaling 18353145_Suggest roles for Notch1/2 signaling in both development of melanocyte during embryogenesis and maintenance of melanocyte stem cells in adulthood. 18359890_Notch-dependent signaling controls myelopoiesis in vivo and in vitro and identifies a requirement for Notch fucosylation in the expression of Notch ligand binding activity and Notch signaling efficiency in myeloid progenitors. 18369158_Indicate Notch activation in cardiomyocytes is mediated through c-Met and Akt survival signaling pathways, and Notch1 signaling in turn enhances Akt activity. 18411251_Lipopolysaccharide (LPS) suppresses Notch signaling via the direct modification of Notch by the nitration of tyrosine residues in macrophages. 18420737_Notch signaling in osteoblasts causes osteopenia and impairs osteo-blastogenesis by inhibiting the Wnt/beta-catenin pathway. 18428029_TGF-alpha regulated DNA synthesis of mouse ES cells via PI3-K/Akt, p44/42 MAPKs and Notch/Wnt pathways 18445292_Notch1 signaling via canonical ligands is dispensable during oogenesis, blastogenesis, implantation and gastrulation 18458347_These results indicate that the Jagged2-Notch axis plays a crucial role in DC-mediated natural killer cell cytotoxicity. 18467661_mood stabilizing drugs expand the neural stem cell pool in the adult brain through activation of notch signaling 18471881_The finding that Gata3 regulates c-Myc expression levels, in a direct or indirect fashion, may explain the parallel phenotypes of mice with overexpression or deficiency of either of the two transcription factors. 18483410_Notch1 upregulates PDGFR-B expression. Notch1 ICD interacts with the part of the PDGFR-B promoter containing the -256/249 CSL-binding site. 18495817_Gain- and loss-of-function mutations in the Notch and Wnt pathways reveal that some aspects of otic placode development - such as Pax8 expression and the morphological thickening of the placode 18547789_altered trafficking pathways may account for the abnormal accumulation of the Notch1 receptor in the endoplasmic reticulum in Pofut1-null mouse embryos. 18550850_the Notch1 and TGFbeta1 signaling pathways cooperatively regulate Foxp3 expression and regulatory T-cell maintenance both in vitro and in vivo 18552977_The inverse correlation between Notch signaling and the proliferative status of the corneal epithelium is consistent with the idea that Notch plays a role in corneal epithelial differentiation. 18563491_Activation of notch1 signaling is associated with suppression of tumor progression in an animal model of medullary thyroid carcinoma 18588871_Study analyzed mice containing conditional deletion of the Notch effector Rbpj or overexpression of the activated Notch1 intracellular domain during lens formation. 18593982_activation of Notch signaling might play a crucial role in brain metastasis 18617694_Cardiovascular lineages are sensitive to an imbalance in Notch signaling, with aberrant activation reflecting a vascular phenotype comparable to a loss-of-function Notch1 mutation. 18628966_AIP4/Itch regulates Notch receptor degradation in the absence of ligand 18635581_Our study demonstrates a T cell intrinsic role for Adam10 in activation of Notch1 during thymocyte development. 18635610_In contrast to Rbpj, Notch1 and Notch2 are not essential for pancreatogenesis. 18641686_Fbxw7 does not function as an oncosuppressor in mouse embryo fibroblast. Instead, it promotes cell cycle progression and cell survival through degr |
ENSG00000148400 |
NOTCH1 |
456.420257 |
2.0179295188 |
1.012876 |
0.20855103 |
23.271983 |
0.00000140633735143329679712141493108656931099176290445029735565185546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000121781299244070358026633543735428588661307003349065780639648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
651.453062702374 |
94.5001357652263 |
322.832416405669 |
35.287878548248 |
| ENSMUSG00000026939 |
51875 |
Tmem141 |
protein_coding |
A2AJB2 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:51875; |
membrane [GO:0016020] |
|
ENSG00000244187 |
TMEM141 |
40.779027 |
2.4699533080 |
1.304484 |
0.43364684 |
8.937256 |
0.00279412085983955305393267032343374012270942330360412597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0113101379051491011257235541620502772275358438491821289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
60.5575824115849 |
18.4778002603231 |
24.5177033169686 |
5.69052199702551 |
| ENSMUSG00000026955 |
72080 |
Sapcd2 |
protein_coding |
Q9D818 |
FUNCTION: Plays a role in planar mitotic spindle orientation in retinal progenitor cells (RPCs) and promotes the production of symmetric terminal divisions (PubMed:26766442). Negatively regulates the mitotic apical cortex localization of GPSM2 (By similarity). Involved also in positive regulation of cell proliferation and tumor cell growth (By similarity). {ECO:0000250|UniProtKB:Q86UD0, ECO:0000269|PubMed:26766442}. |
Alternative splicing;Cell cycle;Cell division;Cell junction;Cell membrane;Coiled coil;Cytoplasm;Membrane;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Tight junction |
|
|
mmu:72080; |
apical cortex [GO:0045179]; apical junction complex [GO:0043296]; apical plasma membrane [GO:0016324]; bicellular tight junction [GO:0005923]; cytosol [GO:0005829]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; establishment of mitotic spindle orientation [GO:0000132]; negative regulation of protein localization to cell cortex [GO:1904777]; positive regulation of cell population proliferation [GO:0008284]; regulation of establishment of planar polarity [GO:0090175]; symmetric cell division [GO:0098725] |
Human_homologues 21998710_the regulatory relationship between miRNAs and the p42.3 gene 23192843_p42.3 overexpression is associated with mitotic progression in malignant transformed cells. 23228105_p42.3 protein plays an important role in the generation and development of gastric cancer. 23576022_p42.3 might play an important role in the progression of colorectal cancer. 23704824_Up-regulation of p42.3 in hepatocellular carcinoma promotes tumorigenicity and tumor growth. 24608043_Role of C9orf140 in the promotion of colorectal cancer progression and mechanisms of its upregulation via activation of STAT5, beta-catenin and EZH2. 24927751_Overexpression of the p42.3 gene is associated with gliomas. 26106439_p42.3 has a role in the pathogenesis of gastric carcinoma 26316259_p42.3 gene expression is associated with gastric mucosal inflammation that can be upregulated by TNF-alpha and H. pylori infection. 26766442_results uncover SAPCD2 as a key regulator of the ternary complex controlling spindle orientation during morphogenesis and asymmetric cell divisions 27449033_Data show that overexpressed p42.3 protein is correlated with gastric cancer cell proliferation, migration, and invasion, suggesting its use as a biological marker in gastric cancer. 27461626_p42.3 protein was related to the occurrence and development of gastric carcinoma. 29531269_Study showed C9orf140 as a novel Axin1-interacting protein and a negative regulator of Wnt/beta-catenin signaling. It functions upstream of beta-catenin, outcompetes PP2A for binding to Axin1, influences the balance between phosphorylation and de-phosphorylation of beta-catenin compromising Wnt3A-induced beta-catenin accumulation. C9orf140 seems to mediate a negative feedback loop of Wnt/beta-catenin signaling by bindi... 31173488_SAPCD2 is increased and positively associated with that of PXN-AS1-L isoform in nasopharyngeal carcinoma (NPC). Gain and loss-of-function experiments demonstrated that SAPCD2 promotes NPC cell proliferation, migration, and invasion. Furthermore, depletion of SAPCD2 significantly reverses the roles of PXN-AS1-L in promoting NPC cell proliferation, migration, and invasion in vitro, and NPC tumor growth in vivo. 32329855_SAPCD2 promotes invasiveness and migration ability of breast cancer cells via YAP/TAZ. 33683171_YY1-inudced activation of lncRNA DUXAP8 promotes proliferation and suppresses apoptosis of triple negative breast cancer cells through upregulating SAPCD2. 35197448_SAPCD2 promotes neuroblastoma progression by altering the subcellular distribution of E2F7. 35467005_MiR-486-5p specifically suppresses SAPCD2 expression, which attenuates the aggressive phenotypes of lung adenocarcinoma cells. |
ENSG00000186193 |
SAPCD2 |
426.875585 |
3.3392887725 |
1.739541 |
0.20337947 |
70.616847 |
0.00000000000000004337966639525456729873705066382689357595975806754055870140263095890986733138561248779296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000013321755615574951164754691797228651259637767197488433801311202842043712735176086425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
687.713965337857 |
97.2697802936972 |
205.946239511328 |
22.2587145550924 |
| ENSMUSG00000026981 |
16181 |
Il1rn |
protein_coding |
P25085 |
FUNCTION: Anti-inflammatory antagonist of interleukin-1 family of proinflammatory cytokines such as interleukin-1beta/IL1B and interleukin-1alpha/IL1A. Protects from immune dysregulation and uncontrolled systemic inflammation triggered by IL1 for a range of innate stimulatory agents such as pathogens. {ECO:0000269|PubMed:31611644, ECO:0000269|PubMed:35332327}. |
Alternative splicing;Cytoplasm;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:16181; |
cytoplasm [GO:0005737]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; vesicle [GO:0031982]; cytokine activity [GO:0005125]; interleukin-1 receptor antagonist activity [GO:0005152]; interleukin-1 receptor binding [GO:0005149]; interleukin-1 type I receptor antagonist activity [GO:0045352]; interleukin-1 type II receptor antagonist activity [GO:0045353]; interleukin-1, type I receptor binding [GO:0005150]; interleukin-1, type II receptor binding [GO:0005151]; acute-phase response [GO:0006953]; fever generation [GO:0001660]; inflammatory response [GO:0006954]; inflammatory response to antigenic stimulus [GO:0002437]; insulin secretion [GO:0030073]; lipid metabolic process [GO:0006629]; memory [GO:0007613]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cell migration [GO:0030336]; negative regulation of glutamate secretion [GO:0014050]; negative regulation of heterotypic cell-cell adhesion [GO:0034115]; negative regulation of interleukin-1-mediated signaling pathway [GO:2000660]; negative regulation of membrane potential [GO:0045837]; positive regulation of JUN kinase activity [GO:0043507]; response to glucocorticoid [GO:0051384]; sensory perception of pain [GO:0019233] |
11792572_Effect of MPC-11 myeloma and MPC-11 + IL-1 receptor antagonist treatment on mouse bone properties. 11810022_Balance between endogenous IL-1 and IL-1Ra during the primary immune response can help determine the antigen-specific effector function of T cells. 11933077_Regulation of transcription of the intracellular interleukin-1 receptor antagonist gene by AP-1 11983917_Genetic alterations of IL-1 receptor antagonist in mice affect plasma cholesterol level and foam cell lesion size 11988319_the capacity of Sertoli cell cultures, from immature mice, to produce IL-1alpha, IL-1beta and IL-1 receptor antagonist (IL-1ra) under in vitro cultures and in the presence of testicular physiological and pathological factors 12716739_highly expressed not only in liver and spleen, but also in white adipose tissue (WAT), where it is upregulated in obesity 12799211_Levels are higher in homogenates of testicular tissue from sexually immature than those from mature mice. 12874179_Absence of IL-1Ra promoted neointimal formation in mice after arterial injury 12878687_CB1 and CB2 receptors modulate release of endogenous IL-1ra from primary cultured glial cells; endogenous IL-1ra is essential for the neuro-protective effects of CBs against excessive activation of glutamate receptors in cultured neurons 14583241_Sleep deprivation markedly increased interleukin 1 receptor antagonist, a natural (anti-inflammatory) modulator of interleukin-1. 15059807_IL-1Ra plays a role in the suppression of lesion development during early atherogenesis & in the modulation of plaque composition. 15218058_Endogenously produced IL-10 plays a significant role in LPS-induced sIL-1Ra gene expression via the activation of STAT3. 15578092_Data suggest that IL-1 receptor antagonist deficiency in T cells disrupts homeostasis of the immune system and that TNF-alpha plays an important role in activating T cells through induction of OX40. 15749858_hepatocyte-derived IL-1ra may control optimal activation and migration of inflammatory dendritic cells within the liver and thereby determine the local immune responses in granulomatous liver disease 15939811_The arteritic phenotype of IL-1ra deficiency is mediated by the interleukin-1 receptor and involves effector Th1 cells 16306368_IL-1Ra is an important regulator of adipogenesis, food intake, and energy expenditure, as seen in knockout mice. 16385385_IL-1Ra reduces insulin sensitivity in rats through a muscle-specific decrease in glucose uptake. 16622029_absence of IL-1ra may suppress TGF-beta-mediated signaling pathway, which is crucial for collagen deposition and vascular endothelial growth factor-mediated neovascularization in wound heali 17021861_Study evaluated the role of Il1rn in asthma mouse model by comparing the genomic sequence between A/J and C3H/HeJ mice, results exclude Il1rn as the gene for allergen-induced bronchial hyper-responsiveness-1 locus. 18279308_Blocking IL-1 receptor-mediated activity resulted in abolishing the alterations in neurogenesis in response to acute and chronic neuroinflammation. 18594791_Knockout mice show increased energy expenditure, fat:carbohydrate oxidation ratio, body temperature, heart rate and catecholamine production. 19002106_NF kappaB was activated without any stimuli by the genetic disruption of IL-1ra, and suppressed cytochrome P450 enzyme expression, thereby reducing acetaminophen-induced liver injury. 19213834_High-fat diet-fed-induced obesity speeds recovery from hypoxia due to obesity-associated up-regulation of IL-1RA. 19559408_IL-1rn was transcriptionally and translationally induced by dendritic cells stimulated with LPS in the absence of TLR4, and the TLR4-independent expression was dependent on PKR pathway. 19656856_9E,11E-CLA exerts unique antiinflammatory effects by increasing an endogenous repressor of IL-1 signaling. 19922413_MSK1 regulates the transcription of IL-1ra in response to TLR activation in macrophages 20110570_IL-1Ra deficiency in inflammatory cells induced aortic valve inflammation and TNF-alpha participates in the development of aortic stenosis in IL-1Ra(-/-) mice. 20112392_myeloid cell-derived IL-1Ra plays a critical role in the control of the development and the severity of collagen arthritis by modulating Th1 and Th17 responses in lymphoid organs 20357263_TLR4-mediated interleukin-1 receptor antagonist production plays a negative regulatory role in low molecular weight hyaluronan (200 kDa)-induced mouse lung inflammation, promoting an anti-inflammatory response. 20610641_The development of psoriasis-like lesions in IL-1R antagonist (Il1rn)-deficient mice is dependent on locally produced tumor necrosis factor (TNF), but not on IL-6 or IL-17. 20639493_Findins define distinct roles for two major cellular sources of IL-1Ra in response to different types of systemic inflammatory stimuli in vivo. 20727895_these results show a protective effect of the IL-1ra in vivo in TLR9-associated liver injury. 20844479_study demonstrates that intestinal epithelia, despite not expressing IL-1beta, secrete sIL-1Ra in a TLR5-dependent manner suggesting that loss of TLR5 may promote inflammation by increasing IL-1beta activity 20920427_IL-1 beta and IL-1RA play a critical role in controlling tissue reactions and thereby sensor function. 21309947_This gene is known to be important for immune responses and may potentially regulate alcohol consumption. 21414240_Loss of IL-1ra gene is associated with spontaneous arthritis. 21525884_infection of IL-1RA(-/-) mice with Leishmania major led to significantly worsened disease outcome with larger lesions, dramatically higher parasite burdens, and decreased IFN-gamma production by antigen-specific T cells 22117073_Energy-sensing factors coactivator peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1alpha) and AMP-activated protein kinase control expression of inflammatory mediators in liver: induction of interleukin 1 receptor antagonist. 22140485_An imbalance in IL-1/IL-1Ra signaling at the IL-1R1 level modulates the severity of cardiac remodeling after acute myocardial infarction in the mouse. 22267332_IL-1Ra plays an important role in suppressing local responses in eyes injected with LPS. 22539301_The results showed that the local production of IL-1Ra by hepatocytes contributes to the resolution of hepatitis. The injured liver produced large amounts of IL-1Ra. The IL-1Ra isoforms were produced with different kinetics. 22567084_The results showed that CCR5 deficiency caused apoptotic cell death of melanoma through inhibition of NF-kappaB and upregulation of IL-1Ra. 22592923_Il-1rn knockout mice exhibit delayed resolution in acute lung inflammation after exposure to lipopolysaccharide, a process that appears to be mediated via the granulocyte colony-stimulating factor/IL-17A axis. 22942082_Balb/c mice deficient in IL-1ra (mouse gene of IL-1RN) develop spontaneous autoimmune arthritis while DBA/1 mice deficient in IL-1ra do not 22980031_Modulation of the IL-1ra pathway by classical IL-6 signalling demonstrates anti-inflammatory and protective activities of IL-6 in vivo. 23308180_In the mouse ischemic heart and in a mouse cardiomyocyte cell line exposed to long-lasting hypoxia, IL-1ra bound and inhibited mitochondria-activated caspases. 23396662_IL-1rn(-/-) mice develop spinal abnormalities that resemble characteristic features associated with human intervertebral disc degeneration. 24566623_IL-1Ra deficiency promoted the expression of inflammatory cytokines beyond IL-1 and altered the expression of genes involved in bone resorption in A. actinomycetemcomitans-infected osteoblasts. 24603878_We demonstrate that Aspergillus fumigatus galactosaminogalactan inhibits cytokines production by inducing Interleukin-1 receptor antagonist 25244011_normalization of IL-1Ra in obesity improved insulin sensitivity due to decreased inflammation in the liver and improved hepatic insulin sensitivity and these effects were independent of changes in body weight. 25479427_IL-1ra regulates baseline GABAergic transmission in the central amygdala and is critical for the ethanol effects at these synapses. 25839897_Results suggest that the IL-1R1/IL-1ra complex regulates specific ethanol behaviors and affects the sensitivity to flurazepam sedation 25909959_Leading to enhanced interleukin-1 receptor antagonist (IL-1RA) production. 26042735_These findings demonstrated novel roles for F-spondin and IL-1Ra in type B CpG oligodeoxynucleotides-mediated cell proliferation and migration of macrophages. 26108163_These findings demonstrate a pathogenic mechanism in which adaptive and innate immunity induce an autoimmune disease in a coordinated manner. 26209680_IL-1Ra may have a protective effect upon EBOV infection. 26276675_Allo-transplantation of mesenchymal stem cells reduces Con A liver injury by increasing IL10 production through an IL1Ra dependent macrophage switch. 26413013_IL-1Ra deficiencies might promote early plaque development in 16-week-old apoE(-/-) mice. 26474296_IL-1Ra is associated with MMP-13 expression and has a novel function in such regulation without interference of the IL-1 signaling cascade. 26661518_results provide evidence of the key role of IL1RA secreted by MSCs to both control the polarization of macrophages toward a M2 phenotype and inhibit B cell differentiation in vivo. 26873573_we demonstrate that the release of IL1RA in response to genotoxic stress contributes to the immunosuppressive effects of apoptotic cells. 26951334_results demonstrate that small intestinal eosinophils play a pivotal role in the maintenance of intestinal homeostasis by regulating Th17 cells via production of IL-1Ra. 26972847_While both contributing to pathogen clearance, NLRP3 more than NLRC4 contributes to deleterious inflammatory responses in cystic fibrosis and correlates with defective NLRC4-dependent IL-1Ra production. 27109611_the suppressed early innate/inflammatory responses instigated by the imbalance between IL-1beta and IL-1Ra is an underlying cause for delayed wound healing in the diabetic corneas 27476761_Following vasectomy, IL1alpha, IL1beta, IL1ra, IL10, and TNF-alpha may mediate immune reaction in whole epididymis, whereas IL6 and TGF-beta1 may mediate regionally different immune response primarily in the lower part of epididymis. 27482635_The current study is the first to demonstrate ex vivo airway hyperreactivity caused by systemic maternal inflammation and postnatal hyperoxia, and it reveals further preclinical mechanistic insights into IL-1Ra as a treatment targeting key pathophysiological features of Bronchopulmonary dysplasia. 28645307_Findings identify a critical role for IL1Ra in maintaining the natural diversity and composition of intestinal microbiota, and suggest a role for TLR4 in mucosal Th17 cell induction associated with the development of autoimmune disease in mice. 29444430_Beta cell IL-1Ra knockout mice had impaired insulin secretion, reduced beta cell proliferation, and decreased expression of islet proliferation genes, along with impaired glucose tolerance. 29891552_these data reveal that IL-1Ra and IL-36Ra have opposing effects on the outcome of Pseudomonas aeruginosa keratitis 30070147_Combinatorial Prg4 and Il-1ra Gene Therapy Protects Against Hyperalgesia and Cartilage Degeneration in Post-Traumatic Osteoarthritis. 31002437_Newly Defined ATP-Binding Cassette Subfamily B Member 5 Positive Dermal Mesenchymal Stem Cells Promote Healing of Chronic Iron-Overload Wounds via Secretion of Interleukin-1 Receptor Antagonist. 31611644_IL-1Ra is an important mediator of type I IFN-driven susceptibility to Mycobacterium tuberculosis infections in vivo. 31865314_Activation of Toll-Like Receptor 3 Induces Interleukin-1 Receptor Antagonist Expression by Activating the Interferon Regulatory Factor 3. 31932497_Intracellular IL-1 Receptor Antagonist Isoform 1 Released from Keratinocytes upon Cell Death Acts as an Inhibitor for the Alarmin IL-1alpha. 31972159_Hepatic stellate cells treated with IL1RN had significantly decreased cellular activation and its associated fibrogenic responses. Collectively, manipulation of TLR5 signaling may be a promising therapeutic strategy for the treatment of liver fibrosis. 32471111_Increase in the Number of Bone Marrow Osteoclast Precursors at Different Skeletal Sites, Particularly in Long Bone and Jaw Marrow in Mice Lacking IL-1RA. 33064141_Inhibition of interleukin-6 signaling attenuates aortitis, left ventricular hypertrophy and arthritis in interleukin-1 receptor antagonist deficient mice. 33493267_IL1RN promotes osteoblastic differentiation via interacting with ITGB3 in osteoporosis. 33924148_Bone Marrow-Derived IL-1Ra Increases TNF Levels Poststroke. 34437647_Impact of thermogenesis induced by chronic beta3-adrenergic receptor agonist treatment on inflammatory and infectious response during bacteremia in mice. 35191758_Enterotoxigenic Escherichia coli Enterotoxins Regulate Epithelial to Immune Relay of IL-33 and IL-1Ra Cytokines. 35332327_IL-1 and IL-1ra are key regulators of the inflammatory response to RNA vaccines. 35412062_Dexmedetomidine attenuates hippocampal neuroinflammation in postoperative neurocognitive disorders by inhibiting microRNA-329-3p and activating the CREB1/IL1RA axis. 36304082_Hypoxia inducible factor 1alpha promotes interleukin-1 receptor antagonist expression during hepatic ischemia-reperfusion injury. |
ENSG00000136689 |
IL1RN |
17.610808 |
11.2867221504 |
3.496555 |
0.73951901 |
23.648894 |
0.00000115612358464505903592688638992225236279409728012979030609130859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000102087388065585739008308760467080844591691857203841209411621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
31.6946115696218 |
16.9673947711971 |
2.80813252487397 |
1.28649402547269 |
| ENSMUSG00000026994 |
14425 |
Galnt3 |
protein_coding |
P70419 |
FUNCTION: Catalyzes the initial reaction in O-linked oligosaccharide biosynthesis, the transfer of an N-acetyl-D-galactosamine residue to a serine or threonine residue on the protein receptor (PubMed:8912633). Has activity toward HIV envelope glycoprotein gp120 (By similarity). Has activity towards EA2, MUC2 and MUC5 (PubMed:8912633). Probably glycosylates fibronectin in vivo (By similarity). Glycosylates FGF23 (By similarity). {ECO:0000250|UniProtKB:Q14435, ECO:0000269|PubMed:8912633}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lectin;Manganese;Membrane;Metal-binding;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. |
|
mmu:14425; |
Golgi apparatus [GO:0005794]; Golgi cisterna membrane [GO:0032580]; perinuclear region of cytoplasm [GO:0048471]; calcium ion binding [GO:0005509]; carbohydrate binding [GO:0030246]; manganese ion binding [GO:0030145]; polypeptide N-acetylgalactosaminyltransferase activity [GO:0004653]; protein O-linked glycosylation [GO:0006493]; protein O-linked glycosylation via serine [GO:0018242]; protein O-linked glycosylation via threonine [GO:0018243]; spermatogenesis [GO:0007283] |
19213845_Galnt3-deficient mice have a biochemical phenotype of tumoral calcinosis and provide in vivo evidence that Galnt3 plays an essential role in proper secretion of Fgf23 in mice. 20077002_The expression levels of murine ppGalNAc-Ts (mGalNAc-Ts), T1, T2, T3, T4, T6, and T7 were compared between mouse colon carcinoma colon 38 cells and variant SL4 cells, selected for their metastatic potentials. 22009723_dietary phosphate restriction normalizes biochemical and skeletal phenotypes of Galnt3 knockout mice and, thus, can be an effective therapy for tumoral calcinosis. 22912827_A mouse with an N-Ethyl-N-nitrosourea (ENU) Induced Trp589Arg Galnt3 mutation represents a model for hyperphosphataemic familial tumoural calcinosis. 23052838_The deficiency of Galnt3 results in a severe reduction of mucin-type O-glycans in spermatids and causes impaired acrosome formation, leading to oligoasthenoteratozoospermia, and Galnt3 may also be involved in the process of fertilization. 25051439_The stabilized Fgf23-Autosomal dominant hypophosphatemic rickets allele reversed the Galnt3-null phenotype and normalized total Fgf23, serum phosphorus, and bone Fgf23 mRNA. 25107907_Overexpression of Galnt3 in chondrocytes causes dwarfism due to the increase of mucin-type O-glycans and a reduction in chondroitin sulfates. 30100058_role of GALNT3 in regulating host responses during influenza A virus infection 31882545_Loss of the disease-associated glycosyltransferase Galnt3 alters Muc10 glycosylation and the composition of the oral microbiome. 36473594_GALNT3 protects against vascular calcification by reducing oxidative stress and apoptosis of smooth muscle cells. |
ENSG00000115339 |
GALNT3 |
115.731613 |
0.2153130662 |
-2.215492 |
0.36448882 |
35.045204 |
0.00000000322139496556707725580545075070963700891724101893487386405467987060546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000422638140529868415761558105795542283900090296810958534479141235351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.4885574408651 |
10.8490568434993 |
197.333855253248 |
36.0698770092853 |
| ENSMUSG00000027004 |
20378 |
Frzb |
protein_coding |
P97401 |
FUNCTION: Soluble frizzled-related proteins (sFRPS) function as modulators of Wnt signaling through direct interaction with Wnts. They have a role in regulating cell growth and differentiation in specific cell types. SFRP3/FRZB appears to be involved in limb skeletogenesis. Antagonist of Wnt8 signaling. Regulates chondrocyte maturation and long bone development (By similarity). {ECO:0000250, ECO:0000269|PubMed:9178261}. |
3D-structure;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:20378; |
cytoplasm [GO:0005737]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Wnt-protein binding [GO:0017147]; canonical Wnt signaling pathway [GO:0060070]; cochlea morphogenesis [GO:0090103]; convergent extension involved in organogenesis [GO:0060029]; epithelial cell development [GO:0002064]; hepatocyte differentiation [GO:0070365]; inner ear morphogenesis [GO:0042472]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cartilage development [GO:0061037]; negative regulation of cell development [GO:0010721]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of hepatocyte differentiation [GO:0070367]; negative regulation of Wnt signaling pathway [GO:0030178]; neural crest cell differentiation [GO:0014033]; positive regulation of apoptotic process [GO:0043065]; positive regulation of fat cell differentiation [GO:0045600]; somite development [GO:0061053] |
15116386_Secreted Frizzled-related proteins (sFrps) are expressed in the postnatal cerebral cortex in regional and lamina patterns in the major subdivisions of the cerebral cortex: the olfactory bulb, the hippocampal formation, and the neocortex. 15312239_sFRP3 may stimulate differentiation through a beta-catenin-independent pathway in addition to its previously known function as a decoy receptor for Wnt's 18050203_Loss of Frzb may contribute to cartilage damage by increasing the expression and activity of MMPs, in a WNT-dependent and WNT-independent manner. This may contribute to the development of osteoarthritis. 18166153_In conclusion, we found that different repertoires of sFRPs exert differential effects on osteoblastic differentiation of mouse mesenchymal cells and cellular apoptosis of mouse osteoblasts in vitro. 18991062_Our findings support Wnt5a and sfrp3 as regulators of otic capsule formation in the developing mouse inner ear. 22264237_Data indicate an important role for frizzled-related protein (Frzb) in joint homeostasis and highlight the complex biology of WNT signaling in the joint. 23207650_Secreted frizzled-related protein 3 (sFRP3) regulates antidepressant responses in mice. 23395446_Study identifies sFRP3 as an inhibitory niche factor from local mature dentate granule neurons that regulates multiple phases of adult hippocampal neurogenesis. 23827310_The spatiotemporally dynamic expression ofsFRP3 strongly suggests that sFRP3 has potential functions in the sensory neuron genesis and sensory circuitry formation. 24330105_left ventricles from post-MI mice showed an increased sFRP3 mRNA level, particularly in cardiac fibroblasts, and (iv) mechanical stretch enhanced sFRP3 expression and release in myocardial fibroblasts 24984954_Our results suggest that FrzB may have a protective role on cartilage degradation and matrix metalloproteinase induction in mouse chondrocytes 26337530_an inverse septal-to-temporal increase in the expression of sfrp3 that emerges during postnatal development, is reported. 26927515_Stable overexpression of Frzb in MC3T3-E1 cells increased calcium deposition and osteoblast markers compared with control. Western blot analysis showed that the increased osteogenesis was associated with reduced canonical, but increased non-canonical Wnt signaling. 28382864_particle-induced inhibition on cell proliferation, differentiation and FRZB expression were all reversed by miR-130b silence 30256452_The results of this study suggested that Frzb secreted from olfactory ensheathing cells in the olfactory nerve layer is important for olfactory axon targeting. 31171792_Study identifies Sfrp3 as a mediator of the stromal-epithelial communication that is required for normal mouse mammary gland development. Loss of Sfrp3 in mice leads to an increase of ductal invasion and branching mirroring an early pregnancy state and predisposes for invasive breast cancer. 32448375_Frizzled related protein deficiency impairs muscle strength, gait and calpain 3 levels. 33820867_Secreted frizzled-related protein 3 was genetically and functionally associated with developmental dysplasia of the hip. 36174667_Secreted frizzled-related protein 3 alleviated cardiac remodeling induced by angiotensin II via inhibiting oxidative stress and apoptosis in mice. |
ENSG00000162998 |
FRZB |
285.435777 |
0.0393713517 |
-4.666710 |
0.32476343 |
181.377096 |
0.00000000000000000000000000000000000000002425216325093754546093193245350202986839679674976293212773455600689977073451093512701207095379521513667546092629656584449548972770571708679199218750000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000029087318947896118903876949506122767013638905754729715162424901880575353289479448316763828754442640193988989238960130023770034313201904296875000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.826533246848 |
4.95494765956082 |
554.376019444371 |
81.7534293472622 |
| ENSMUSG00000027102 |
15437 |
Hoxd8 |
protein_coding |
P23463 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15437; |
nucleus [GO:0005634]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific double-stranded DNA binding [GO:1990837]; anterior/posterior pattern specification [GO:0009952]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal system morphogenesis [GO:0048705] |
18570930_Hoxd8 is increased in mid-gestational wounds compared with late-gestational control skin 19825936_Prox1 and HoxD8 play important roles in the maturation and maintenance of lymphatic vessels. |
ENSG00000175879 |
HOXD8 |
8.704382 |
0.0116294136 |
-6.426078 |
1.54799422 |
17.413890 |
0.00003006210409847932574266618499514436280151130631566047668457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002025236486634396653079559502685924599063582718372344970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.166568839257919 |
0.215050197071439 |
14.3230643287978 |
8.29035216841142 |
| ENSMUSG00000027115 |
228421 |
Kif18a |
protein_coding |
Q91WD7 |
FUNCTION: Microtubule-depolymerizing kinesin which plays a role in chromosome congression by reducing the amplitude of preanaphase oscillations and slowing poleward movement during anaphase, thus suppressing chromosome movements. May stabilize the CENPE-BUB1B complex at the kinetochores during early mitosis and maintains CENPE levels at kinetochores during chromosome congression (By similarity). {ECO:0000250}. |
ATP-binding;Cell projection;Coiled coil;Cytoplasm;Cytoskeleton;Glycoprotein;Isopeptide bond;Microtubule;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Protein transport;Reference proteome;Transport;Ubl conjugation |
|
|
mmu:228421; |
caveola [GO:0005901]; cytoplasm [GO:0005737]; kinesin complex [GO:0005871]; kinetochore [GO:0000776]; kinetochore microtubule [GO:0005828]; microtubule [GO:0005874]; microtubule cytoskeleton [GO:0015630]; microtubule organizing center [GO:0005815]; mitotic spindle astral microtubule [GO:0061673]; mitotic spindle midzone [GO:1990023]; nucleus [GO:0005634]; ruffle [GO:0001726]; actin binding [GO:0003779]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule plus-end binding [GO:0051010]; plus-end-directed microtubule motor activity [GO:0008574]; tubulin-dependent ATPase activity [GO:0070463]; cellular response to estradiol stimulus [GO:0071392]; male meiotic nuclear division [GO:0007140]; microtubule depolymerization [GO:0007019]; microtubule-based movement [GO:0007018]; mitotic metaphase plate congression [GO:0007080]; mitotic sister chromatid segregation [GO:0000070]; protein transport [GO:0015031]; regulation of microtubule cytoskeleton organization [GO:0070507]; seminiferous tubule development [GO:0072520] |
23872115_In conclusion, Kif18a is critical for colorectal carcinogenesis in the setting of inflammation by mechanisms of increased PI3K-AKT signaling. 25824710_Kif18a is specifically required for mitotic progression during germ line development. 31677127_phylogenetic analysis revealed that the KIF18A gene family is remarkably conserved across vertebrates. 34515734_Micronuclei in Kif18a mutant mice form stable micronuclear envelopes and do not promote tumorigenesis. |
ENSG00000121621 |
KIF18A |
307.301779 |
2.1799344931 |
1.124285 |
0.15903248 |
49.660989 |
0.00000000000182743681433347037866083139780429587606738750515944502694765105843544006347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000353242608577759142386051671537097870409716016126822069054469466209411621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
420.28153077019 |
46.4227419202622 |
192.795486331684 |
16.4744871341341 |
| ENSMUSG00000027168 |
18508 |
Pax6 |
protein_coding |
P63015 |
FUNCTION: Transcription factor with important functions in the development of the eye, nose, central nervous system and pancreas. Required for the differentiation of pancreatic islet alpha cells (PubMed:9163426). Competes with PAX4 in binding to a common element in the glucagon, insulin and somatostatin promoters. Regulates specification of the ventral neuron subtypes by establishing the correct progenitor domains (By similarity). Acts as a transcriptional repressor of NFATC1-mediated gene expression (PubMed:23990468). {ECO:0000250, ECO:0000269|PubMed:23990468, ECO:0000269|PubMed:9163426}. |
Alternative splicing;Cytoplasm;Developmental protein;Differentiation;Direct protein sequencing;Disease variant;DNA-binding;Homeobox;Nucleus;Paired box;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
This gene encodes a homeobox-containing protein that functions as a regulator of transcription. It plays a key role in the development of neural tissues, particularly the eye. Activity of this protein is also required for expression of glucagon in the pancreas. This gene is regulated by multiple enhancers located up to tens or hundreds of kilobases upstream and downstream of the transcription start sites. Mutations in this gene or deletion of these regulatory elements results in severe defects in eye development. Alternative splicing and the use of alternative promoters results in multiple transcript variants, some of which encode proteins that lack the N-terminal paired domain. [provided by RefSeq, Jul 2015]. |
mmu:18508; |
chromatin [GO:0000785]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; chromatin binding [GO:0003682]; chromatin DNA binding [GO:0031490]; co-SMAD binding [GO:0070410]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; double-stranded DNA binding [GO:0003690]; histone acetyltransferase binding [GO:0035035]; HMG box domain binding [GO:0071837]; minor groove of adenine-thymine-rich DNA binding [GO:0003680]; protein kinase binding [GO:0019901]; R-SMAD binding [GO:0070412]; RNA binding [GO:0003723]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II core promoter sequence-specific DNA binding [GO:0000979]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; transcription coregulator binding [GO:0001221]; ubiquitin protein ligase binding [GO:0031625]; anatomical structure development [GO:0048856]; anterior/posterior pattern specification [GO:0009952]; astrocyte differentiation [GO:0048708]; axon guidance [GO:0007411]; axonogenesis [GO:0007409]; blood vessel development [GO:0001568]; brain development [GO:0007420]; camera-type eye development [GO:0043010]; cell differentiation [GO:0030154]; cell fate commitment [GO:0045165]; cell fate determination [GO:0001709]; cell population proliferation [GO:0008283]; cellular response to leukemia inhibitory factor [GO:1990830]; cerebral cortex development [GO:0021987]; cerebral cortex regionalization [GO:0021796]; chromatin remodeling [GO:0006338]; commitment of neuronal cell to specific neuron type in forebrain [GO:0021902]; cornea development in camera-type eye [GO:0061303]; DNA demethylation [GO:0080111]; DNA methylation [GO:0006306]; dorsal/ventral axis specification [GO:0009950]; dorsal/ventral pattern formation [GO:0009953]; embryonic camera-type eye morphogenesis [GO:0048596]; epithelial cell development [GO:0002064]; establishment of mitotic spindle orientation [GO:0000132]; eye photoreceptor cell development [GO:0042462]; forebrain development [GO:0030900]; forebrain dorsal/ventral pattern formation [GO:0021798]; forebrain-midbrain boundary formation [GO:0021905]; gene expression [GO:0010467]; glandular epithelial cell differentiation [GO:0002067]; glucose homeostasis [GO:0042593]; habenula development [GO:0021986]; hindbrain development [GO:0030902]; histone acetylation [GO:0016573]; insulin metabolic process [GO:1901142]; interkinetic nuclear migration [GO:0022027]; iris morphogenesis [GO:0061072]; keratinocyte differentiation [GO:0030216]; lacrimal gland development [GO:0032808]; learned vocalization behavior or vocal learning [GO:0098598]; lens development in camera-type eye [GO:0002088]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of neural precursor cell proliferation [GO:2000178]; negative regulation of neuroblast proliferation [GO:0007406]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of protein phosphorylation [GO:0001933]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural crest cell migration [GO:0001755]; neural precursor cell proliferation [GO:0061351]; neuroblast proliferation [GO:0007405]; neuron differentiation [GO:0030182]; neuron migration [GO:0001764]; olfactory bulb development [GO:0021772]; olfactory bulb mitral cell layer development [GO:0061034]; oligodendrocyte cell fate specification [GO:0021778]; pallium development [GO:0021543]; pancreatic A cell development [GO:0003322]; pancreatic A cell differentiation [GO:0003310]; pituitary gland development [GO:0021983]; positive regulation of cell death [GO:0010942]; positive regulation of cell fate specification [GO:0042660]; positive regulation of core promoter binding [GO:1904798]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of epithelial cell differentiation [GO:0030858]; positive regulation of gene expression [GO:0010628]; positive regulation of glucagon secretion [GO:0070094]; positive regulation of glutamatergic neuron differentiation [GO:0120008]; positive regulation of neuroblast proliferation [GO:0002052]; positive regulation of neuron migration [GO:2001224]; positive regulation of transcription by RNA polymerase II [GO:0045944]; protein localization to organelle [GO:0033365]; protein phosphorylation [GO:0006468]; regionalization [GO:0003002]; regulation of asymmetric cell division [GO:0009786]; regulation of cell migration [GO:0030334]; regulation of DNA-templated transcription [GO:0006355]; regulation of gene expression [GO:0010468]; regulation of neurogenesis [GO:0050767]; regulation of neuron differentiation [GO:0045664]; regulation of neuron projection development [GO:0010975]; regulation of timing of cell differentiation [GO:0048505]; regulation of transcription by RNA polymerase II [GO:0006357]; response to wounding [GO:0009611]; retina development in camera-type eye [GO:0060041]; rhombomere morphogenesis [GO:0021593]; salivary gland morphogenesis [GO:0007435]; sensory neuron migration [GO:1904937]; signal transduction involved in regulation of gene expression [GO:0023019]; smoothened signaling pathway [GO:0007224]; telencephalon regionalization [GO:0021978]; transcription by RNA polymerase II [GO:0006366]; type B pancreatic cell differentiation [GO:0003309]; ventral spinal cord development [GO:0021517] |
11069920_Pax6 interacts with Chx10 via its paired domain or homeodomain (HD) and the HD of Chx10, resulting in enhanced transactivation by Pax6. This interaction was modeled on a demonstrated interaction between zebrafish Pax6 and mouse Chx10. 11069920_Pax6 interacts with Rax via its paired domain or homeodomain (HD) and the HD of Rax, resulting in enhanced transactivation by Pax6. This interaction was modeled on a demonstrated interaction between zebrafish Pax6 and mouse Rax. 11698186_role in embryonic eye development 11790784_identification of downstream genes 11830592_downregulaiton in melanoma cells by interleukin-6 receptor signaling 11850181_New 3' elements control Pax6 expression in the developing pretectum, neural retina and olfactory region 11896398_results demonstrate an important role of Pax6 as intrinsic fate determinant of the neurogenic potential of glial cells 11983873_The requirement of the 5a isoform in iris formation suggests that the evolution of this isoform contributed to advanced features of the vertebrate eye. 12072567_role of expression with six3 on haploinsufficient lens phenotype 12118260_High expression of one functional allele of either of the homeobox genes Emx2 or Pax6 in the dorsal telencephalon is necessary and sufficient to activate morphogenesis of cerebral cortex 12167158_role in regulating lens-specific delta1-crystallin enhancer 12175506_Promoter-specific transcripts are expressed within subdomains of total Pax6 expression, but differ from one another in their spatial localization and expression over time 12223410_Pax6 is required for establishing naso-temporal and dorsal characteristics of the optic vesicle. 12324464_mDia influences Pax6-induced transcriptional activity and axonal pathfinding in a way opposite from ROCK (Rho kinase) and that it may act via Pax6 to modulate early neuronal development 12351165_Pax6 activity is directly required for differentiation of the chamber angle in developing mouse eye 12351726_Lack of Pax6 or Foxg1 disrupts early events in thalamocortical tract formation 12388550_The paired domain of PAX6 is required for homeodomain function 12397112_consequences of the loss of Pax6 function on thalamocortical (TCA) and corticofugal axon (CFA) pathfinding during the period of embryonic day (E) 14.5 to E18.5 12485166_These results provide a list of genes with possible roles in lens biology and cataracts that are directly or indirectly regulated by Pax6. 12540617_Activates endogenous proglucagon gene expression in the rodent gastrointestinal epithelium. 12586435_Low dorsal to high ventral gradient correlated with expression of axonal guidance cue EphB2. Transfection into undifferentiated P19 cells up-regulated EphB2. Possibility that Pax6 is upstream of EphB2. 12618140_We suggest that the primary role of Pax6 is the continual regulation of cell surface properties responsible for both cellular identity and radial migration, defects of which cause regional cell sorting and abnormalities of migration in chimeras. 12648492_roles of Pax6 in the cornea, retina, and olfactory epithelium of the developing embryo 12692153_cellular adhesion is compromised in the SEY (+/-) corneal epithelium suggesting a role for Pax6 in the proper generation and maintenance of the adult cornea. 12710953_Pax6 protein directly and positively regulates its own gene expression 12756174_Results demonstrate that the redundant activities of Pax2 and Pax6 direct the determination of retinal pigment epithelium. 12783797_Pax6 regulates neurogenin 2 developmental expression 12816048_The levels of expression of Oct-4 involved in maintenance of the pluripotent status of embryonic stem cells proved to be indistinguishable in undifferentiated cells and embryoid bodies, while the expression of Pax-6 markedly increased in the latter 12878679_The cerebral cortical antihem is lost in mice homozygous for the Small eye (Pax6) mutation and suggest the loss of EGF signaling at least partially explains defects in cortical patterning and cell migration in Small eye mice 14586016_Pioneer axon outgrowth was rescued in vivo by selective replacement of R-cadherin by electroporation into cultured Pax6 mutant embryos 14732405_novel cis-acting elements that govern the regulation of the complex spatio-temporal expression of Pax6. 14762144_Pax6 and En1 have essential roles in establishing the recurrent inhibitory circuit between motor neurons and Renshaw cells 15031110_role in forming pancreatic glucagon cell identity 15080895_Neurogenesis becomes fully Pax6-dependent in the neurosphere culture system, independent of the region of origin, and that Pax6 overexpression is sufficient to direct almost all neurosphere-derived cells towards neurogenesis. 15180990_Pax-6 has a role in regulating islet-specific glucose-6-phosphatase catalytic subunit-related protein gene transcription 15376329_The gene expression profile of the Pax6 pallium mutant was studied at different embryonic days. 15514979_These results have identified potentially important interactions between series of pioneer axons and the Pax6 gene as a general regulator of longitudinal tract formation in the forebrain. 15523634_Neuron pairs showed uneven/asymmetric expression of Pax6 whereas other pairs were either symmetrically positive or negative. 15548580_Splicing as a pivotal factor regulating the potent neurogenic role of Pax6. 15558474_the regulation of SOX2 (and SOX1/3) and its partner factors, exemplified by Pax6, determines the spatio-temporal order of the occurrence of cell differentiation [review] 15558475_pRb antagonizes Pax6-mediated activation of the alphaA-crystallin promoter likely by inhibiting binding of Pax6 to DNA [review] 15634788_These findings delineate the transcription factor sequence Pax6 --> Tbr2 --> Tbr1 in the differentiation of radial glia --> intermediate progenitor cell --> postmitotic projection neuron. 15733660_Data suggest that Pax6 regulates the timing of retinal neurogenesis and couples it with specific neuron differentiation programs. 15758185_There is a selective requirement for Pax6, but not EMX2, in the formation of a subset of nuclei within the amygdaloid complex. 15878992_Pax6 delimits the appropriate proliferative zone for GABA interneurons and regulates their numbers and distributions by repressing the ventral fates of dorsal telencephalon progenitors and progeny. 15987699_we genetically dissected the complex mouse Small-eye phenotype, thereby pinpointing the underlying Pax6/PAX6 haploinsufficiency to autonomous dosage requirements within the developing iris and lens/cornea tissues. 16023139_tissue-specific expression of alphaA-crystallin is regulated by recruitment of Pax6 and c-Maf 16024294_The Pax-Eya-Six-Dach network is at best only partly conserved during lens and nasal placode development. 16029501_Pax6 transcripts are 6-10 times more prevalent than Pax6(5a) transcripts early in neurogenesis in the murine telencephalon, diencephalon and hindbrain and the ratio later falls significantly to about 3:1 in these regions. 16049175_Pax6 is required in a cell-autonomous manner for the production of cells in the dopaminergic lineage 16115881_Pax6 regulation of Optimedin in the eye and brain may directly affect multiple developmental processes, including cell migration and axon growth 16364898_These studies suggest that underlying the diversity and proportions of fates produced by the lower rhombic lip is a precise suborganization regulated by Pax6. 16407227_HIPK2 is an upstream protein kinase for Pax6 that may modulate Pax6-mediated transcriptional regulation 16425216_Our results identified three elements of Pax6 action: (i) expression of neuron-specific genes; (ii) establishment of a postmitotic phenotype; and (iii) involvement in the regulation of cell shape and cell migration. 16464444_Pax6 expression, analyzed using a BAC transgene, revealed the presence of a paired-less isoform in the eye and olfactory bulb. 16675956_Data demonstrate a novel mechanism of Pax6, c-Maf and CREB function, through regulation of chromatin-remodeling enzymes, and suggest a multistage model for the activation of alphaA-crystallin during lens differentiation. 16866875_Normal distribution and integration of Neural Crest Derived Cells in ocular tissues depend on a proper dosage of Pax6. 16914058_defective calcium signaling in Pax6+/- cells underlies wound-healing delay 16973151_Pax6 regulates delta-catenin expression during central nervous system development in mice. 16979618_These data show functions of Pax6 in developing cortical cells, to repress expression of transcription factors in the lateral ganglionic eminence, to prevent precocious differentiation and to induce development of cortical basal progenitor cells. 17010333_results reveal an evolutionarily conserved role for Pax6 in AP-restricted expression of vertebrate Hoxd4 orthologs 17049510_During pancreatic islet formation, Pax6 expression becomes dependent upon Pbx1 and Pbx2 function. 17064680_early Foxe3 expression is sensitive to halved Pax6 gene dosage & there is phenotypic similarity between Pax6 & Foxe3 mutants; we propose that many ocular malformations associated with Pax6 haploinsufficiency are consequences of reduced Foxe3 expression 17066077_Six3 directly activates Pax6 and probably also Sox2 in the PLE and regulates cell autonomously the earliest stages of mammalian lens induction. 17157287_Pax6 is required to regulate the proliferation rate of neuroepithelial progenitors present in the mouse optic vesicle 17158062_This analysis revealed time- and region-specific differences in Pax6-mediated transcription, explaining the specific function of Pax6 at early and later stages of neurogenesis. 17202185_the proliferation of cortical progenitors is sensitive to altered Pax6 levels 17264084_the combinatorial code of Tnc isoform expression in the neural stem/progenitor cell is complex and regulated by Pax6 17316600_Pax6 is required for the proliferation and expansion of RSCs. 17329367_Conditional activation of Pax6 in the developing cortex of transgenic mice causes progenitor apoptosis. 17399698_Pax6 has a role in maintaining neuronal precursors in a pre-differentiation state by counteracting the proneural activity of Ngn2. 17634386_loss of Pax6 results in the ectopic specification of interneurons in the cortical ventricular zone and axonal disorganization 17679940_Colocalization of Pax6 and AP-2alpha was mainly observed in the proliferating central lens epithelium, the same region in which the lens stalk phenotype was observed in the double heterozygous Pax6(+/lacZ)/AP-2alpha(+/-) eyes. 17901057_Pax-6 and c-Maf interact with G1 to activate basal expression of the glucagon gene 18062951_The ratio of Pax6+PD (containing the N-terminal paired-box DNA-binding domain and Pax6DeltaPD (lacking the domain) isoforms in the distal retina is important for both cornea and lens development. 18199433_Pax6 acts upstream of MafB, which in turn triggers the expression of insulin and regulate the expression required for beta-cell maturation. 18287938_We found a decrease in Sox2+ neural progenitor cells in the subventricular zone of E18.5 Pax6(-/-) mice. Pax6 binds to the Sox2 promoter and activates Sox2 expression. 18307776_The genetic interplay between the transcription factors, Pax6 and Er81, is responsible, in part, for the regional specification of a distinct sublineage of layer 5 projection neurons. 18371392_Report neurotrophin receptor-mediated death of misspecified neurons generated from embryonic stem cells lacking Pax6. 18386822_Data demonstrate that anterior eye development is very sensitive to Pax6 dosage. 18448636_Pax6 inhibits proliferation and promotes maturation in murine astrocytes 18467663_Pax6 is a multifunctional player regulating proliferation and differentiation through the control of expression of different downstream molecules in a highly context-dependent manner [review] 18507827_Before the onset of severe retinal dysplasia, Pax6 overexpression causes defects of retinal axons, preventing their normal growth and navigation through the optic chiasm 18562615_Dlx proteins and Pax6 physically interact and require each other to exert some of their key functions in adult neurogenesis. 18562673_Pax6 activity ranged between 100 and 0% showing that the extent of eye development is progressively reduced as Pax6 activity decreased. 18701439_The establishment of the epidermal growth factor receptor positive lateral cortical stream migratory route to the basal telencephalon is pax6 dependent. 18753373_This study findings indicate that Pax6 expression gradients in cortical progenitors do not directly impart thalamocortical or corticofugal areal identity. 18832594_Sox1 enhances neuroectodermal commitment and maintenance but blocks further differentiation. In contrast, Pax6 is involved in the progression of neuroectoderm toward radial glia. 19004853_Timed genetic removal of Pax6 appeared to unmask a cryptic divergence of retinal progenitor cells into qualitatively divergent progenitor pools. 19087642_Pax6 may play an important role in the development of upper incisor teeth in mice. 19097998_Cdc42-mTOR signaling pathway controls Hes5 and Pax6 expression in retinoic acid-dependent neural differentiation. 19116343_Disruption of metabolic homeostasis mainly caused by Pax6 haploinsufficiency was mainly mediated by down-regulation of GLP-1. 19132093_the present studies represent an integrative genome-wide approach to identify downstream networks controlled by Pax6 that control mouse lens and forebrain development. 19146846_Results show that Lhx2 and Pax6 transcription factors operate in a concerted manner during retinal development to promote transcriptional activation of the Six6 homeobox-gene in primitive and mature retinal progenitors. 19208436_the Math5 retinal enhancer also requires Pax6 for its activation, via Pax6 binding to a highly conserved binding site. 19239004_Phenethyl isothiocyanate down regulated the levels of PKC alpha, pax-6 and netrin-1 during the differentiation process of mouse embryonic stem cells. 19258013_These data provide important insights into specific functions of PAX6 and NKX2.2 during glial cell specification that have so far remained largely unexplored. 19378250_molecular properties of individual mouse Pax2 and Pax6 proteins are essential determinants of mouse eye development 19387074_PAX77(+/-) mice showed an overlapping but distinct spectrum of eye abnormalities to Pax6(+/-) heterozygotes (low Pax6 dose). 19399607_These findings provide new data in the chemical characterization of mouse Pax6-positive periglomerular cells. 19474196_recovered and identified the exact regions deleted in three new Pax6 deletions; deletion of Rcn1 directly or indirectly contributes to the eye phenotype in Pax6 contiguous gene deletions 19500363_ventral telencephalic Pax6 is important for formation of the Islet1-expressing corridor and the thalamic and cortical axons that grow through. 19505513_Pax6 is required for differentiation and/or maintenance of external plexiform layer interneurons in the adult mouse olfactory bulb. 19521500_Pax6 positively and directly regulates genes that promote neural stem cell self-renewal and neurogenesis. 19563798_dosage-sensitive roles of Pax6 in the formation of both the CB and the iris. 19570848_Pax6 is pivotal for initiation of the lens fiber differentiation program in the mammalian eye. 19571125_This series of experiments indicate that ablation of Pax6 function the developmental regulator Pax6 plays a role in establishing cortical circuits that underlie behavioral and cognitive functions. 19666824_These data support a model in which the mode of Pax6-Sox2 inter-regulation is stage-dependent and suggest an underlying mechanism in which DNA binding site availability is regulated. 19703295_Results clearly indicate that PRL does signal through PRL-RS in the decidua as well as the ovary, independently of PRL-RL, and activates/represses transcription factors Pax5, Pax6 and Runx1 in a tissue specific manner. 20027618_Lens morphogenesis is dependent on Pax6-mediated inhibition of the canonical Wnt/beta-catenin signaling in the lens surface ectoderm. 20081189_expression of Shroom3 is dependent on the crucial lens-induction transcription factor Pax6 20177825_The interaction of Pax6, secreted protein acidic rich in cysteine (SPARC) and p53 may influence Smad3-dependent auto-regulation of Pax6. 20377917_Study shows that inactivation of the Pax6 paired domain leads to a more severe phenotype with regards to the differentiation of pancreatic alpha-cells than the loss of the transactivation domain. 20413611_Pax6 levels are determined by transcriptional synergy of Prep1 bound to two phylogenetically conserved lower-affinity DNA-binding sites, while timing of enhancer activation is determined by binding site affinity 20538882_Defects in iridocorneal angle formation are caused by non-autonomous mechanisms due to Pax6 haploinsufficiency in lens or corneal. 20592023_Pax6 is critical for alpha cell function and differentiation through the transcriptional control of key genes involved in glucagon gene transcription, proglucagon processing, and alpha cell differentiation 20628604_Notch1 activates the expression of regulatory transcription factors such as Sox9, Pax6, Runx1, Myf5 and Id proteins that are critically involved in lineage decisions in the absence of protein synthesis. 20852734_Results describe the expression of Pax6 and Sox2 in normal olfactory epithelium and during epithelial regeneration after methyl bromide lesion or olfactory bulbectomy. 21051674_the percentage of virus-infected cells with Na(+) currents was increased by inhibition of the repressor function of Olig2 and by overexpression of Pax6 21059263_sonic hedgehog-GLI1 downstream target genes PTCH1, Cyclin D2, Plakoglobin, PAX6 and NKX2.2 are differently regulated in medulloblastoma and astrocytoma 21084637_sumoylation at K91 is required for p32 Pax-6 to bind to a homeodomain-specific site and regulate expression of target genes 21126516_the granule cell as an intrinsic target of the mutant gene and key events in the life of the developing granule cell that depend upon normal Pax6 expression. 21205789_Results demonstrate that precise regulation of the ratio of SOX2 to PAX6 is necessary to ensure accurate progenitor cell specification, and place SOX2 as a decisive factor of neural competence in the retina. 21309073_Pax6 regulates craniofacial form, at stages when cranial neural crest has just arrived in the frontonasal region, through its control of surface cephalic ectodermal competence to form an essential craniofacial patterning center. 21382351_Our data suggest functional Neurog1-Pax6 cross-talk in the activation of downstream targets in early cerebellar development 21418559_We conclude that Foxg1 exerts control over telencephalic progenitor proliferation by cell autonomous mechanisms that include the regulation of Pax6 21447684_Pax6 functions directly to maintain normal, corneal epithelial cells. 21471366_Pax6 plays an important role in the generation of multiple subtypes of neurons that contribute to the amygdala and olfactory bulb. 21500284_This study demonistrated that photoreceptor injury induces migration of PAX6-positive Muller cell nuclei toward the outer part of the INL and into the inner part of the outer nuclear layer. and express cell cycle markers. 21602652_results suggest that Pax6 regulate the EGF-responsive stem cells in the subventricular zone 21695148_an epigenetic mechanism involving regulations of Pax6 gene during embryonic stem cell differentiation to neural stem cells 21698109_provide evidence that Pax6 binds to the olig2 promoter and that an HEB sequence in the promoter is required for the Pax6 repression of olig2 transcription 21766338_Sox2 and Pax6 are both critical for self-renewal of differentiation-competent radial glia-like neural stem cells 21901510_Modulation in the expression of Pax6 and reduction in Pax6 positive cells show direct association of Pax6 with aging-related neuronal dystrophy. 21996446_CRALBP is a direct downstream target of Pax6. 21998302_Data establish a novel regulatory role for Pax6 during lens development, link together the Pax6/c-Maf/crystallin regulatory network, and suggest a novel type of GRN subcircuit that controls a major part of embryonic lens development. 22031545_Pax6 plays a role in regulating the orientation and mode of cell division of progenitors in the mouse cerebral cortex. 22077982_Pax6 mutant mice have reduced numbers of basal progenitor cells in the ventral part of the thalamus. 22220192_Enhancer activity of an ultra-conserved cis-regulatory region located near Pax6, termed E60, was characterised. 22220198_The effects of altered Pax6 dose on the corneal stroma and endothelium in Pax6+/- and PAX77Tg/- mice, were investigated. 22232070_Targets of RBPJ/N1ICD in cortical neural stem cell at a genome-wide level, were identified. 22240097_Results provide functional evidence for the independent and synergistic roles of the Pax6 upstream enhancers. 22275805_The data show key roles for endogenous Dhh signalling in maintenance and regeneration of the corneal epithelium, demonstrate an interaction between Pax6 and hedgehog signalling in the corneal epithelium. 22354470_Suggest that Pax6 plays pivotal roles in specifying neural progenitor cell commitments and maintaining certain mature neuronal fates. 22359602_expression of Pax6 is necessary and sufficient to render postmitotic neurons competent to respond to SFRP1 to increase axonal growth 22384097_analysis of FlpIn-3T3 cell lines stably expressing Pax6 or Pax6(5a) 22514120_the expression of Pax6 severely impairs tumor propagation by inducing a reduction of cell proliferation and the acquisition of differentiation traits in tumor-initiating cells. 22539874_Pax6 function is regulated through TGF-beta, and SPARC influences the shuttling of Pax6 via the TGF-beta/Smad signaling pathway. 22729175_miR-7a fine-tunes Pax6 expression as a mechanism for controlling neuronal stem cell diversity and, consequently, neuronal phenotype. 22766506_these results suggest that Tip60 binds with Pax6 and that this physical interaction leads to the full-transcriptional activation of Pax6 during retina development. 22792072_in the retinal pigment epithelium(RPE) , PAX6 cooperates with either one (or both) of two related RPE transcription factors, MITF and TFEC, to suppress extracellular signals that in the normal retina induce a signaling cascade promoting retina formation. 22817342_The identification of candidate genes that represent direct or indirect Pax6-downstream genes involved in cerebellar development. 23056534_proSAAS as a novel down-regulated target of Pax6 23326536_This study offers insight into the function of the Pax6 protein in eyelid development 23326594_upregulation in the number of ghrelin-expressing endocrine cells was observed. These findings demonstrate that Pax6 is essential for adult maintenance of glucose homeostasis and function of the endocrine pancreas. 23342162_Pax6 interactions with chromatin and identification of its novel direct target genes in lens and forebrain. 23353076_mitral cell fate is critically dependent upon down-regulation of Pax6 and the ensuing up-regulation of Tbr2 and Tbr1. 23376040_Dexamethasone in combination with methylisobutylxanthine (DM) induces Cyp1b1 and a 6.7 kb mouse Cyp1b1 promoter-luciferase reporter in each cell type prior to adipogenesis. An 18 base sequence (at -6.11 kb) (PaxE) which was essential for this reporter stimulation in 3T3-L1 cells bound the transcription factor Pax6. 23404109_Pax6 achieves its key roles in the developing forebrain by utilizing particular subdomains to coordinate patterning, neurogenesis and proliferation simultaneously. 23515312_Pax6 is uniquely required for eye development, but in brain development, Pax6 can be functionally substituted by related Pax family genes that share a similar paired domain binding specificity. 23516376_this study identifies a novel evolutionarily conserved mechanism by which Pax6 controls the down-regulation of multiple genes through direct up-regulation of miR-204. 23529217_Results of the present study suggest that Pax6 is expressed in bone and may play an important role in osteocyte differentiation by controlling canonical Wnt signaling. 23578930_Pax6 expression is highly dynamic in rhombomeres, suggesting an early function in the hindbrain 23592925_These findings demonstrate a novel allele of the paired box gene 6 (Pax6) that affects lens development in a semidominant manner leading to a classical small-eye phenotype. 23622063_The results of this study indicated that Pax6's modulation of cortical progenitor cell cycles is regional and direct. 23816521_PAX6, in addition to each patterning function, is expressed in a group of late born interneurons that derive from the p2 and p0 domains. 23933087_Deletion of either Pax6 in the subependymal zone (SEZ) causes the progeny of adult neural stem cells to convert to the ependymal lineage within the SEZ while migrating neuroblasts convert to different glial lineages en route to or in the olfactory bulb 23967157_Increased corneal epithelial turnover contributes to abnormal homeostasis in the Pax6(+/-) mouse model of aniridia. 23990468_Pax6 binds endogenously to the proximal region of the tartrate acid phosphatase (TRAP) gene promoter and suppresses nuclear factor of activated T cells c1 (NFATc1)-induced TRAP gene expression. 24047795_Mutant PAX6 binds the Pc2 promoter and regulates promoter activity. 24115331_Pax6 is a novel downstream target of the Wnt/ss-catenin pathway, and ss-catenin/Pax6 signaling plays critical roles in self-renewal and neurogenesis of radial glia/neural stem cells during neocortical development. 24143217_functional role of Pax6 loss in SQM pathogenesis 24284204_Meis2 cooperates with Pax6 in generic neurogenesis and dopaminergic fate specification in the adult subventricular zone-olfactory bulb system. 24355924_Manipulation of Pax6 dosage through a mutation-independent nonsense mutation suppression strategy limits progressive, postnatal damage in the eye. 24523460_when Pax6 was inactivated at the onset of contact between the optic vesicle and surface ectoderm in Pax6(Sey/flox) embryos, expression of lens-specific genes was not initiated and neither the lens nor the retina formed 24528677_Pax6 has an evolutionarily conserved function in establishing the temporospatial expression of Shh in the mid-diencephalic organizer in vertebrates. 24773074_PAX6 downregulates miR-124 expression to promote cell migration during embryonic stem cell differentiation 24875170_Somatic mutagenesis of Pax6 in the differentiating RPE revealed that PAX6 functions in a feed-forward regulatory loop with MITF during onset of melanogenesis. 24925705_Data (including data from heterozygous Pax6 mutant mouse strain) suggest that Pax6 plays role in development of diabetes type 2 (DM2T); early-onset DM2T can result from interaction of genetic/environmental factors involving Pax6 and high-fat diet. 24948600_The zinc-finger protein Sp8 plays a supplementary role to Pax6 in establishing the pMN/p3 domain boundary. 25117830_deregulation of the cell cycle machinery and TF expression in aOBSCs which are deficient in Pax6 may be at the origin of the phenotypes observed in this adult NSC population. 25176648_Pax6 blocks diencephalic Shh expression cell autonomously. Absence of Pax6 causes non-cell-autonomous diencephalic patterning defects. 25209290_The existence of spatial compartmentation in the rhombic lip and the interplay between Wls, Math1, and Pax6 in the rhombic lip provides novel views of early cerebellar development. 25352170_Pax6 directly regulates the activity of the Odf2 gene encoding for the appendage-specific protein Odf2 with a role for the assembly of mother centriole. 25615698_Study demonstrates that FGFR2 plays an essential role in controlling cell proliferation and differentiation, and maintaining Pax6 levels in corneal epithelium via ERK-independent pathways during embryonic development. 25689933_Retinal pigment epithelium (RPE) of a beta-catenin/Pax6 double mutant was differentiated into the neural retina; however, the tissue was thinner than that of the conditional beta-catenin mutant due to reduced proliferation; data show that Pax6 is required for the RPE differentiation by regulating pigmentation and accountable for hyperproliferation in the transdifferentiated RPE. 25794677_Onecut1 and Onecut2 have roles in operating downstream of Pax6 to regulate horizontal cell development 25808240_Pax6 expression is essential for horizontal basal cells to differentiate into neuronal cells during regeneration. 25827297_PAX6 expression in the inner retina of calcitonin receptor-like receptor transgenic (CLR(SMalphaA)) mice suggest a role in regulatory mechanisms involved in glaucoma-related retinal cell death. 26021864_NFIA and NFIB are endogenously expressed in gradients with high caudo-medial to low rostro-lateral expression ... this expression pattern deviates from that of PAX6, suggesting that PAX6 does not drive Nfi expression 26032914_The expression dynamics shows that Pax6 localizes at XY body during meiotic prophase I, especially in the mid-phase of pachytene spermatocytes which suggests that Pax6 may have some function in inactivation of sex chromosomes during meiosis. 26138486_Novel Pax6-dependent gene regulatory network that contribute to the function of Pax6 were identified in both forebrain and lens. 26187199_the Pax6 paired domain has roles in neural tube patterning and peripheral nervous system development 26252244_Upper-layer neuron production was increased in double-transgenic mouse embryos with sustained Pax6 expression in the neurogenic lineage. Increased basal progenitors existed in the subventricular zone and the intermediate zone of the neocortex. 26372689_RIP140 protects LSD1's catalytic domain and antagonizes its Jade-2-mediated ubiquitination and degradation. 26485579_This study demonstrated that Pax6 is expressed in astrocytes and binds to the GLT-1 promoter in vitro and in vivo. 26520399_results indicate that Pax6 is required by thalamic progenitors for the normal molecular patterning of the thalamic neurons that they generate 26658466_Pax6 inactivation in the adult pancreas reveals ghrelin as endocrine cell maturation marker. 26739814_Glucose-stimulated expression of ALX3 in alpha cells provides a regulatory mechanism for the downregulation of glucagon gene expression by interfering with PAX6-mediated transactivation on the glucagon G1 promoter element. 27171428_These data suggest that Pax6 and Sox2 have counteracting roles in regulating neurogenesis. 27240603_this Pax6 (fl) allele provides a useful addition to the existing Pax6 allelic series and this study demonstrates the utility of using compound heterozygotes with null alleles to unmask cryptic effects of floxed alleles. 27452274_These results demonstrate a novel role for Tlx3 and indicate that Pax6-Tlx3 expression and interaction is part of a region specific regulatory network in cerebellum and its deregulation during development could possibly lead to Autistic spectral disorders (ASD). 27455012_c.718C>T transitional mutation of PAX6 has been found to underlie the aniridia, which showed an autosomal dominant inheritance pattern in this northeastern Chinese family 27455013_novel mutation in the PAX6 gene has been identified in a Chinese family affected with aniridia 27581449_The present study shows how Pax6 is key to the development of glutamatergic cells in the cerebellum 27771509_Tgfbeta2, Bmp4 and Foxc1 are direct downstream targets of Pax6 in the developing iris. 27855195_paternal aging has profound effects on the onset of behavioral abnormalities in mice carrying a mutation of Pax6, a gene with neurodevelopmental regulatory functions. 27871855_HDAC1 binds PAX6 and protein-protein interaction leads to transcriptional repression of PAX6 target genes during mouse retinal development 27887593_The expression of Barhl2 in the thalamus and pretectum is related to, and regulated by, the expression of Pax6. Pax6 is known to be required for normal diencephalic development. The findings suggest that some of its actions might be mediated by its maintenance of a repressive influence over Barhl2 expression in the thalamus and pretectum. 27918583_we show that simultaneous deletion of Meis1 and Meis2 in presumptive lens ectoderm results in arrested lens development in the pre-placodal stage, and neither lens placode nor lens is formed. We found that |
ENSG00000007372 |
PAX6 |
158.935653 |
0.3050582320 |
-1.712843 |
0.22008349 |
60.251336 |
0.00000000000000834861505296545324329537451039815290529085348159021862102235900238156318664550781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000002098450569088610286262232064935344309178789218295335672337387222796678543090820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
71.3577723483458 |
10.9279163643918 |
233.915249163711 |
24.7012868610345 |
| ENSMUSG00000027224 |
213696 |
Duoxa1 |
protein_coding |
Q8VE49 |
FUNCTION: May be required for the maturation and the transport from the endoplasmic reticulum to the plasma membrane of functional DUOX1. {ECO:0000250}. |
3D-structure;Glycoprotein;Membrane;Protein transport;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:213696; |
cell leading edge [GO:0031252]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; plasma membrane [GO:0005886]; enzyme binding [GO:0019899]; hydrogen peroxide metabolic process [GO:0042743]; positive regulation of hydrogen peroxide biosynthetic process [GO:0010729]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of reactive oxygen species metabolic process [GO:2000379]; protein localization [GO:0008104]; protein transport [GO:0015031]; regulation of inflammatory response [GO:0050727]; regulation of thyroid hormone generation [GO:2000609] |
20233719_Nip1 has an important role in the guidance of neuronal differentiation through ROS generation and modulation of intermediate filaments 21362455_DUOXA1 is a novel p53-regulated neurogenic factor involved in p53 dependent neuronal differentiation. 24410844_DUOXA1 overexpression in muscle stem cells induces apoptosis and inhibits differentiation through DUOX1 and ASK1. 26914863_The proliferation and migration of granule cells was delayed after P15 in Duoxa-/- mice. However, these changes disappeared by P25. Although the cerebellar structure of Duoxa-/- mice was not significantly different from that of Wt mice at P25, motor coordination was impaired. It was also found that the amplitude of paired-pulse facilitation at parallel fiber-Purkinje cell synapses decreased in Duoxa-/- mice. 32929281_Structures of mouse DUOX1-DUOXA1 provide mechanistic insights into enzyme activation and regulation. |
ENSG00000140254 |
DUOXA1 |
24.948839 |
0.2960562081 |
-1.756057 |
0.51722938 |
11.351019 |
0.00075406591573870464413620329224841043469496071338653564453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0035826363158240641035301443650951114250347018241882324218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.6604088694331 |
4.27057614364323 |
39.3857941471506 |
9.88475628083661 |
| ENSMUSG00000027326 |
76464 |
Knl1 |
protein_coding |
Q66JQ7 |
FUNCTION: Performs two crucial functions during mitosis: it is essential for spindle-assembly checkpoint signaling and for correct chromosome alignment. Directly links spindle checkpoint proteins BUB1 and BUB1B to kinetochores. Part of the MIS12 complex, which may be fundamental for kinetochore formation and proper chromosome segregation during mitosis. Acts in coordination with CENPK to recruit the NDC80 complex to the outer kinetochore (By similarity). {ECO:0000250|UniProtKB:Q8NG31}. |
Cell cycle;Cell division;Centromere;Chromosome;Chromosome partition;Kinetochore;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Repeat |
|
|
Human_homologues hsa:57082; |
acrosomal vesicle [GO:0001669]; kinetochore [GO:0000776]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; attachment of spindle microtubules to kinetochore [GO:0008608]; cell division [GO:0051301]; protein localization to kinetochore [GO:0034501] |
31197172_Study in mouse model finds that following Knl1 deletion, segregation errors in mitotic neural progenitor cells give rise to DNA damage on the missegregated chromosomes. This triggers rapid p53 activation and robust apoptotic and microglial phagocytic responses that extensively eliminate cells with somatic genome damage, thus causing microcephaly. 35167144_Kinetochore scaffold 1 regulates SAC function during mouse oocyte meiotic maturation. |
ENSG00000137812 |
KNL1 |
718.405857 |
2.2526448368 |
1.171620 |
0.12118811 |
92.089521 |
0.00000000000000000000082842877837134110486893881439121701459501150522883328999532159819807475287234410643577575683593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000362604228510122663689734842507626214295974448920139063907638887762630020006326958537101745605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1002.68654540167 |
84.3259610128188 |
445.115239216242 |
28.6624258541518 |
| ENSMUSG00000027331 |
51944 |
Knstrn |
protein_coding |
Q9D9Z1 |
FUNCTION: Essential component of the mitotic spindle required for faithful chromosome segregation and progression into anaphase. Promotes the metaphase-to-anaphase transition and is required for chromosome alignment, normal timing of sister chromatid segregation, and maintenance of spindle pole architecture. The astrin (SPAG5)-kinastrin (SKAP) complex promotes stable microtubule-kinetochore attachments. Required for kinetochore oscillations and dynamics of microtubule plus-ends during live cell mitosis, possibly by forming a link between spindle microtubule plus-ends and mitotic chromosomes to achieve faithful cell division. {ECO:0000250|UniProtKB:Q9Y448}. |
Cell cycle;Cell division;Centromere;Chromosome;Coiled coil;Cytoplasm;Cytoskeleton;Kinetochore;Microtubule;Mitosis;Nucleus;Reference proteome |
|
|
mmu:51944; |
centriolar satellite [GO:0034451]; cytoplasm [GO:0005737]; kinetochore [GO:0000776]; microtubule organizing center [GO:0005815]; microtubule plus-end [GO:0035371]; mitotic spindle [GO:0072686]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; ruffle [GO:0001726]; spindle pole [GO:0000922]; microtubule plus-end binding [GO:0051010]; protein homodimerization activity [GO:0042803]; cell division [GO:0051301]; cell migration [GO:0016477]; cellular response to epidermal growth factor stimulus [GO:0071364]; chromosome segregation [GO:0007059]; microtubule cytoskeleton organization [GO:0000226]; mitotic sister chromatid segregation [GO:0000070]; regulation of attachment of spindle microtubules to kinetochore [GO:0051988]; spindle organization [GO:0007051] |
26667018_important role in the successful outcome of male gametogenesis |
ENSG00000128944 |
KNSTRN |
1048.645484 |
2.0659263886 |
1.046789 |
0.11656419 |
79.940452 |
0.00000000000000000038586528419904599859832941621568994282127612560992111975788354527594492537900805473327636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000143353658754143125043824604743152126288479940541221654970449606025795219466090202331542968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1396.89216891377 |
112.986331825959 |
676.157764685663 |
41.6219909752809 |
| ENSMUSG00000027333 |
228608 |
Smox |
protein_coding |
Q99K82 |
FUNCTION: Flavoenzyme which catalyzes the oxidation of spermine to spermidine. Can also use N(1)-acetylspermine and spermidine as substrates, with different affinity depending on the isoform (isozyme) and on the experimental conditions. Plays an important role in the regulation of polyamine intracellular concentration and has the potential to act as a determinant of cellular sensitivity to the antitumor polyamine analogs. May contribute to beta-alanine production via aldehyde dehydrogenase conversion of 3-amino-propanal. |
Alternative splicing;Cytoplasm;FAD;Flavoprotein;Nucleus;Oxidoreductase;Reference proteome |
PATHWAY: Amine and polyamine degradation; spermine degradation. {ECO:0000269|PubMed:14764092}. |
|
mmu:228608; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; nuclear membrane [GO:0031965]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; N1-acetylspermine:oxygen oxidoreductase (N1-acetylspermidine-forming) activity [GO:0052895]; norspermine:oxygen oxidoreductase activity [GO:0052894]; oxidoreductase activity [GO:0016491]; polyamine oxidase activity [GO:0046592]; spermine:oxygen oxidoreductase (spermidine-forming) activity [GO:0052901]; polyamine catabolic process [GO:0006598]; spermine catabolic process [GO:0046208] |
12458219_expression and characterization of mouse spermine oxidase 15955064_we identified a second domain that is necessary for nuclear localization of the spermine oxidase splice variant 20839014_Spermine oxidase binds spermine in a similar conformation as that observed in the yeast polyamine oxidase FMS1-spermine complex and demonstrate a major role for residues His82 and Lys367 in substrate binding and catalysis. 21839041_Spermine oxidase mediates the gastric cancer risk associated with Helicobacter pylori CagA. 23840306_Novel evidences of the complex and critical functions carried out by spermine oxidase and spermine in the mammalian brain. 23857253_ROS overexposure by mSMOX delivers an earlier adaptive response to radiation 24854736_Data (including data from studies using recombinant mutant proteins) suggest that Glu216-Ser218 is a hot spot responsible for Smox substrate specificity; Smox with mutation E216L/S218A exhibits specificity for N'-acetylspermine rather than spermine. 25406264_Spermine oxidase is an important regulator of muscle gene expression and fiber size. 29397558_Dach-SMOX transgenic mouse can be considered as a suitable in vivo genetic model to study the involvement of spermine oxidase in excitotoxicity. 29922289_Our studies indicate that SMOX has immunomodulatory effects in experimental colitis via polyamine flux. Thus, SMOX contributes to the immunopathogenesis of C. rodentium infection, but is protective in DSS colitis, indicating the divergent effects of spermidine. 31197571_Epileptic seizures and oxidative stress in a mouse model over-expressing spermine oxidase. 31545418_High SMOX expression is associated with neuroblastoma. 32350444_Spermine oxidase mediates Helicobacter pylori-induced gastric inflammation, DNA damage, and carcinogenic signaling. 33054763_Ablation of polyamine catabolic enzymes provokes Purkinje cell damage, neuroinflammation, and severe ataxia. 33153123_Emerging Role for Linear and Circular Spermine Oxidase RNAs in Skeletal Muscle Physiopathology. 34767785_Protective Role of Spermidine in Colitis and Colon Carcinogenesis. |
ENSG00000088826 |
SMOX |
530.713497 |
0.3574345473 |
-1.484249 |
0.15600981 |
88.220177 |
0.00000000000000000000585595183342874482190064571769558769964113766552421370763757690891537777133635245263576507568359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000002404254941422820615488026375131218570719443353025604080339117096798418060643598437309265136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
277.775747613121 |
30.0966252578948 |
777.137380062906 |
61.2660578977934 |
| ENSMUSG00000027375 |
17153 |
Mal |
protein_coding |
O09198 |
FUNCTION: Could be an important component in vesicular trafficking cycling between the Golgi complex and the apical plasma membrane. Plays a role in the maintenance of the myelin sheath and in axon-glia and glia-glia interactions. {ECO:0000269|PubMed:15337780}. |
Alternative splicing;Cell membrane;Golgi apparatus;Lipoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
This gene encodes a highly hydrophobic integral membrane protein belonging to the MAL family of proteolipids. The encoded protein has been localized to the endoplasmic reticulum of T-cells and is a candidate linker protein in T-cell signal transduction. In addition, this proteolipid is localized in compact myelin of cells in the nervous system and has been implicated in myelin biogenesis and/or function. The protein plays a role in the formation, stabilization and maintenance of glycosphingolipid-enriched membrane microdomains. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Jan 2010]. |
mmu:17153; |
apical plasma membrane [GO:0016324]; endoplasmic reticulum [GO:0005783]; Golgi membrane [GO:0000139]; hinge region between urothelial plaques of apical plasma membrane [GO:0120003]; membrane [GO:0016020]; membrane raft [GO:0045121]; plasma membrane raft [GO:0044853]; Schmidt-Lanterman incisure [GO:0043220]; structural constituent of myelin sheath [GO:0019911]; central nervous system myelination [GO:0022010]; myelination [GO:0042552]; positive regulation of extrinsic apoptotic signaling pathway via death domain receptors [GO:1902043]; protein insertion into plasma membrane [GO:0098737]; protein localization to paranode region of axon [GO:0002175] |
15193296_The specific reduction and mistargeting of MAL protein as a reaction to sulfatide overload may contribute to the pathogenic mechanisms in metachromatic leukodystrophy. 15337780_Our results demonstrate a critical role for MAL in the maintenance of central nervous system paranodes, likely by controlling the trafficking and/or sorting of NF155 and other membrane components in oligodendrocytes. 19508690_Our results suggest a functional role for MAL in peripheral myelination by influencing the expression of membrane components that mediate axon-glia interaction during ensheathment and myelin wrapping. 19889726_Phagosomal retention of Francisella tularensis results in TIRAP/Mal-independent TLR2 signaling. 22323295_the exclusion of MAL from the expanding 2D crystals of uroplakins explains the selective association of MAL with the hinge areas in the uroplakin-delivering fusiform vesicles, as well as at the apical surface 22895261_VIP17/MAL overexpression results in abnormal cilium and cyst development, in vitro and in vivo, and in VIP17/MAL-overexpressing mice that develop cysts secondary to a ciliary defect. 25290060_MAL overexpression leads to reduced expression of Mpz and p75NTR, despite functional pathways and normal expression of genes important for Schwann cell differentiation. 26081637_Expression of the developmentally regulated proteolipid MAL is required for the cytotoxic effect of Clostridium perfringens Epsilon Toxin. 31364376_siRNA-mediated knockdown of MAL in cells resulted in a decrease in PLC-beta3 protein expression and an increase in PIP2 33255528_Absence of Mal/TIRAP Results in Abrogated Imidazoquinolinones-Dependent Activation of IRF7 and Suppressed IFNbeta and IFN-I Activated Gene Production. |
ENSG00000172005 |
MAL |
8.359289 |
0.0211156086 |
-5.565546 |
1.49989392 |
15.796805 |
0.00007052147601508455224562227359186294961546082049608230590820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004346103727840542642335552869781167828477919101715087890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.377945877180718 |
0.46763995096643 |
17.8988862464001 |
9.44013828129443 |
| ENSMUSG00000027379 |
12235 |
Bub1 |
protein_coding |
A0A8I4YXJ8 |
Human_homologues FUNCTION: Serine/threonine-protein kinase that performs 2 crucial functions during mitosis: it is essential for spindle-assembly checkpoint signaling and for correct chromosome alignment. Has a key role in the assembly of checkpoint proteins at the kinetochore, being required for the subsequent localization of CENPF, BUB1B, CENPE and MAD2L1. Required for the kinetochore localization of PLK1. Required for centromeric enrichment of AUKRB in prometaphase. Plays an important role in defining SGO1 localization and thereby affects sister chromatid cohesion. Acts as a substrate for anaphase-promoting complex or cyclosome (APC/C) in complex with its activator CDH1 (APC/C-Cdh1). Necessary for ensuring proper chromosome segregation and binding to BUB3 is essential for this function. Can regulate chromosome segregation in a kinetochore-independent manner. Can phosphorylate BUB3. The BUB1-BUB3 complex plays a role in the inhibition of APC/C when spindle-assembly checkpoint is activated and inhibits the ubiquitin ligase activity of APC/C by phosphorylating its activator CDC20. This complex can also phosphorylate MAD1L1. Kinase activity is essential for inhibition of APC/CCDC20 and for chromosome alignment but does not play a major role in the spindle-assembly checkpoint activity. Mediates cell death in response to chromosome missegregation and acts to suppress spontaneous tumorigenesis. {ECO:0000269|PubMed:10198256, ECO:0000269|PubMed:15020684, ECO:0000269|PubMed:15525512, ECO:0000269|PubMed:15723797, ECO:0000269|PubMed:16760428, ECO:0000269|PubMed:17158872, ECO:0000269|PubMed:19487456, ECO:0000269|PubMed:20739936}. |
ATP-binding;Nucleotide-binding;Proteomics identification;Reference proteome |
|
|
Human_homologues hsa:699; |
kinetochore [GO:0000776]; ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; mitotic spindle assembly checkpoint signaling [GO:0007094]; protein phosphorylation [GO:0006468] |
14646599_mutations of Bub1 and BubR1 found in Brca2- mutant mice indeed are responsible for the chromosome instability in Brca2-mutated tumors 16969117_Our data suggest that Bub1 is a critical spindle checkpoint protein that regulates accurate chromosome alignment and homolog disjunction in mammalian oocyte meiosis. 17938250_Bub1 is important for mediating cell death when chromosomes missegregate. 19117986_Bub1 is essential for proper chromosome segregation, a defect that can lead to severe phenotypes, including perinatal lethality and a predisposition to cancer. 19249208_Data show that the anaphase-promoting complex is activated in an exponential fashion after germinal vesicle breakdown in oocytes and that this process is advanced by 5 hr in oocytes lacking Bub1. 19617567_Bub1 dysfunction is linked to inherited aneuploidy in female germ cells and may contribute to the maternal age-related increase in aneuploidy and pregnancy loss. 19962666_Bub1 insufficiency can drive tumor formation through tumor suppressor gene loss of heterozygosity, but only in restricted genetic and cellular contexts 20124418_It seems that Sgo1 sets up the centromeric protection mechanism in G2, but that its Bub1-dependent localisation to centromeres during mitosis is not required to maintain cohesion. 21646403_Results establish that Bub1 has oncogenic properties and suggest that Aurora B is a critical target through which overexpressed Bub1 drives aneuploidization and tumorigenesis. 23209306_It was shown that that Bub1 kinase activity integrates attachment error correction and mitotic checkpoint signaling by controlling the localization and activity of Aurora B kinase through phosphorylation of histone H2A at threonine 121. 26031201_Bub1 coordinates checkpoint signalling by distinct domains for RZZ and BubR1 recruitment and localizes antagonistic checkpoint activities. 29608549_Bub1 insufficiency attenuates TF expression and reduces TM suppression by blocking Akt and JNK phosphorylation, respectively, thus leading to the prevention of ARF and death caused by sepsis. |
ENSG00000169679 |
BUB1 |
1121.289108 |
2.3815916966 |
1.251926 |
0.17218311 |
51.703472 |
0.00000000000064548497509214131280307539965767093831806172632425955271173734217882156372070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000131728961343457437682591998448595137748140526667839367291890084743499755859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1533.51267137147 |
183.45684297505 |
643.902426084232 |
58.1259580308388 |
| ENSMUSG00000027386 |
70370 |
Fbln7 |
protein_coding |
Q501P1 |
FUNCTION: An adhesion molecule that interacts with extracellular matrix molecules in developing teeth and may play important roles in differentiation and maintenance of odontoblasts as well as in dentin formation. {ECO:0000269|PubMed:17699513}. |
Calcium;Cell adhesion;Coiled coil;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Heparin-binding;Reference proteome;Repeat;Secreted;Signal;Sushi |
|
|
mmu:70370; |
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; calcium ion binding [GO:0005509]; heparan sulfate proteoglycan binding [GO:0043395]; heparin binding [GO:0008201]; cell adhesion [GO:0007155] |
17699513_TM14 is a cell adhesion molecule that interacts with extracellular matrix molecules in teeth and suggest that TM14 plays important roles in both the differentiation and maintenance of odontoblasts as well as in dentin formation. 29730503_Results found that fibulin-7 is highly expressed in the adult kidney and binds heparin through its N-terminal domain. Fbln7-deficient mice were protected from renal tubular calcification and fibrosis after high phosphate diet. The study suggests that Fbln7 is a local mediator of calcium deposition in the kidney. 30518776_This study examined the in vivo antiangiogenic activity of recombinant full-length mouse Fbln7 (Fbln7-FL) and Fbln7-C-Terminal Fragment proteins using a rat corneal angiogenesis model. Both Fbln7-FL and Fbln7-C inhibited neovascularization. 32297473_A C-terminal fragment of adhesion protein fibulin-7 inhibits growth of murine breast tumor by regulating macrophage reprogramming. 36370444_Fibulin7 aggravates calcium oxalate-induced acute kidney injury by binding to calcium oxalate crystals. |
ENSG00000144152 |
FBLN7 |
172.090473 |
0.2716841693 |
-1.879998 |
0.22558383 |
67.288971 |
0.00000000000000023448950874997163040094646379960226498116572057921203064978499241988174617290496826171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000067264485824473973079251355434255780798945409046374344086416385835036635398864746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
71.406610359254 |
11.2108901739255 |
262.829485205167 |
28.8899092993257 |
| ENSMUSG00000027408 |
56264 |
Cpxm1 |
protein_coding |
Q9Z100 |
FUNCTION: May be involved in cell-cell interactions. No carboxypeptidase activity was found yet. |
Carboxypeptidase;Disulfide bond;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Secreted;Signal;Zinc |
|
|
mmu:56264; |
extracellular space [GO:0005615]; metallocarboxypeptidase activity [GO:0004181]; zinc ion binding [GO:0008270]; peptide metabolic process [GO:0006518]; protein processing [GO:0016485] |
15094061_transient up-regulation pattern of CPX-1 expression may be important for the successful progression from pre-osteoclasts to mature osteoclasts. 27006448_These findings establish CPX-1 as a positive regulator of adipogenesis situated downstream of FGF-1/BAMBI that may contribute to hyperplastic adipose tissue expansion via affecting extracellular matrix remodeling. |
ENSG00000088882 |
CPXM1 |
71.995075 |
8.0517134229 |
3.009296 |
0.37503130 |
63.968486 |
0.00000000000000126425466380350521250943302494065487050644463248166848856612887175288051366806030273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000339033926743925881321049094736972412134903484548509311480302130803465843200683593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
129.620617824528 |
34.075899836333 |
16.0985135729585 |
3.49019326892566 |
| ENSMUSG00000027416 |
57329 |
Otor |
protein_coding |
Q9JIE3 |
|
Disulfide bond;Reference proteome;Secreted;SH3 domain;Signal |
|
|
mmu:57329; |
extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; cartilage condensation [GO:0001502] |
Human_homologues 20800603_Observational study of gene-disease association. (HuGE Navigator) 30897334_results indicate that OTOR plays a crucial role in the progression of and prognosis for breast carcinoma (BC), which could help to identify future therapeutic targets for treating BC patients |
ENSG00000125879 |
OTOR |
741.071431 |
7.8730941152 |
2.976931 |
0.15852509 |
325.030955 |
0.00000000000000000000000000000000000000000000000000000000000000000000000116165365368063855136396609335975155859135585334382636717768305341082879245693101676273184804781770584719790554626933336798654813412349843647231973589617854948138820960983748581483304684880409 |
0.0000000000000000000000000000000000000000000000000000000000000000000003686314261013226208979889121818187856554783699660437125542790207904642382982928261203975949984136582171555444493054066547899221584010797235478795316012765299656927361343227052270510313292107 |
Yes |
No |
1317.43349169882 |
145.052680653234 |
167.333639407401 |
14.6255810385711 |
| ENSMUSG00000027447 |
13010 |
Cst3 |
protein_coding |
P21460 |
FUNCTION: As an inhibitor of cysteine proteinases, this protein is thought to serve an important physiological role as a local regulator of this enzyme activity. |
Disulfide bond;Protease inhibitor;Reference proteome;Secreted;Signal;Thiol protease inhibitor |
|
The protein encoded by this gene is a cysteine protease inhibitor involved in neurodegenerative and cardiovascular processes. The encoded protein inhibits aggregation of beta-amyloid protein, a hallmark of Alzheimer's disease, so it may be useful as a therapeutic. This protein also may be a biomarker for atherosclerosis. [provided by RefSeq, Aug 2015]. |
mmu:13010; |
axon [GO:0030424]; basement membrane [GO:0005604]; cell projection [GO:0042995]; collagen-containing extracellular matrix [GO:0062023]; contractile fiber [GO:0043292]; cytoplasm [GO:0005737]; endoplasmic reticulum [GO:0005783]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; lysosome [GO:0005764]; multivesicular body [GO:0005771]; neuronal cell body [GO:0043025]; nuclear membrane [GO:0031965]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; vesicle [GO:0031982]; amyloid-beta binding [GO:0001540]; cysteine-type endopeptidase inhibitor activity [GO:0004869]; endopeptidase inhibitor activity [GO:0004866]; identical protein binding [GO:0042802]; peptidase inhibitor activity [GO:0030414]; protease binding [GO:0002020]; defense response [GO:0006952]; negative regulation of cell death [GO:0060548]; negative regulation of cysteine-type endopeptidase activity [GO:2000117]; negative regulation of elastin catabolic process [GO:0060311]; negative regulation of peptidase activity [GO:0010466]; negative regulation of proteolysis [GO:0045861]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA replication [GO:0045740]; regulation of programmed cell death [GO:0043067]; response to oxidative stress [GO:0006979]; supramolecular fiber organization [GO:0097435] |
11803549_expressed in decidual cultures and the expression is controlled in part by TGF-beta(1) and EGF signaling 12044447_spatiotemporal up-regulation of cystatin C in the hippocampus is induced by entorhinal deafferentation (perforant path transections) 14607896_Cystatin C is neither necessary nor sufficient to control expression of major histocompatibility type II complexes and antigen presentation in dendritic cells. 14742906_In vivo overexpression of human cystatin C does not affect amyloid-beta levels in mice that do not deposit amyloid-beta. 15509901_Data indicate that cystatin C is involved in astrocyte differentiation during mouse brain development. 15823274_leukocyte-specific expression of cystatin C is actively involved in matrix remodelling associated with atherosclerosis plaque regression. 16051881_the protective role of cystatin C in atherosclerosis is dependent primarily on its expression in nonhematopoietic cell types 16166629_PKB activity is a critical downstream component of PDK-1 in mediating translation of cystatin C, RANKL, and Rab11a 16242633_These data demonstrate that cystatin C has a role in neuronal death and neurogenesis during SE-induced network reorganization. 16595632_Validity of cystatin C for generating neural stem cells from embryo stem cells in vitro. 17086443_We propose that in MS, remyelination may be impaired by increasing activity of cathepsins inadequately controlled by cystatin C. 17462596_Finally, in vivo studies using kidney-specific megalin knockout mice provide evidence that megalin mediates proximal tubular uptake of cystatin C. We conclude that megalin is an endocytic receptor of cystatin C in PTC. 17592728_These results suggest that CysC affects the BMP signaling cascades in osteoblastic cells and then promotes osteoblast differentiation, mineralization, and bone formation. 18681831_Plasminogen activator inhibitor-2, but not cystatin C, inhibits the prometastatic activity of tissue inhibitor of metalloproteinases-1 in the liver.( 18957217_CysC regulates soluble Abeta and Abeta-associated neuronal deficits through inhibiting CatB-induced Abeta degradation. 19446036_Cystatin C through this effect can act as an immunomodulatory molecule. 19773092_Cystatin C, a protein targeted to the classical secretory pathway by its signal peptide sequence, is secreted by primary neurons in at least 9 different cystatin C glycoforms associated with exosomes. 20093401_The embryo's cystatin C and F expression functions as a protective mechanism against the maternal proteinase cathepsin S. 20352108_CysC plays a protective role under conditions of neuronal challenge by inducing autophagy via mTOR inhibition 20472891_Data show that cystatin c (CystC) contributes to experimental abdominal aortic aneurysms (AAA) pathogenesis and that lack of CystC favors inflammation in AAA lesions induced in atherosclerotic mice. 20489058_Oxidants induce elevated cystatin C production from cardiac myocytes. Cystatin C plays a role in cardiac extracellular matrix remodelling. 20889561_Data show that cystatin C effectively rescues cystatin B loss-of-function mutation, facilitating the reversal of pathophysiological changes and suggesting a novel therapeutic intervention for patients with neurodegenerative disorders. 21085595_Data show that the absence of cystatin C reduced epithelial cell apoptosis but increased proliferation in skin. 21300820_Inflammation causes downregulation of cystatin C expression in dendritic cells and reduces serum CstC levels. 21443774_The lack of cystatin C enhances Collagen-induced arthritis and primarily affects in vivo priming of the immune system 23671283_APP expression stimulates NSPC proliferation; this effect is mediated via an increase in cystatin C secretion 24405769_Cystatin C is a potential pathogenic signal triggering neurodegeneration in multiple system atrophy. 24570004_an important role for macrophages, DC, and ROS in diseases associated with the protease inhibitory activity or amyloidogenic properties of cystatin C. 25356866_The neuroprotective activity of CysC against Amyotrophic lateral sclerosis-linked mutant Cu/Zn-superoxide dismutase (SOD1)-mediated toxicity. 26592151_Data (including data from studies in mutant mice) suggest that up-regulation of Cst3, as observed in plasma of mice with type 2 diabetes, down-regulates insulin signaling and promotes endoplasmic reticulum stress in hepatocytes but not in myotubes. 27079462_CysC plays an important regulatory role in combating cell death via the autophagic pathway in atherosclerosis. 29724997_In mouse and human lungs with interstitial fibrosis, CST3 and GDF15 expression levels were markedly reduced, and the restoration of these cytokines alleviated the fibrotic changes in mouse lungs. 31513306_cystatin C promoted skewing of T cells toward proinflammatory T-helper (Th)1/Th17 differentiation which was mediated by augmented extracellular signal-regulated kinase 1/2 mitogen-activated protein kinase phosphorylation. 33580179_pH-dependent and dynamic interactions of cystatin C with heparan sulfate. 33754041_Deciphering the autophagy regulatory network via single-cell transcriptome analysis reveals a requirement for autophagy homeostasis in spermatogenesis. 34914637_Hypoxic vasodilatory defect and pulmonary hypertension in mice lacking hemoglobin beta-cysteine93 S-nitrosylation. |
ENSG00000101439 |
CST3 |
11475.171098 |
0.4838292380 |
-1.047430 |
0.04488319 |
535.240839 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000020454377325327620162319953019438020284801211502654531198139306842486028708826495428796226701090255648982158838398709692170764064023243312290458575 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001483624168663763363655730839361190657996345118595013029261210981323554252597778924158476775789068355319017248567999785000973425372574393987919711 |
Yes |
No |
7605.52750143588 |
236.651122285858 |
15719.4458371274 |
361.96618256598 |
| ENSMUSG00000027460 |
11602 |
Angpt4 |
protein_coding |
Q9WVH6 |
FUNCTION: Binds to TEK/TIE2, modulating ANGPT1 signaling. Can induce tyrosine phosphorylation of TEK/TIE2. Promotes endothelial cell survival, migration and angiogenesis. {ECO:0000269|PubMed:15284220}. |
Angiogenesis;Coiled coil;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal |
|
This gene is a member of the angiopoietin family of secreted growth factors comprising Angiopoietins-1, -2, and -3, all of which bind the TEK Receptor Tyrosine Kinase. Angiopoietin/TEK Receptor Tyrosine Kinase signaling is involved in survival and migration of endothelial cells and regulates vascular remodeling and maintenance of vascular integrity. Angiopoietin/TEK Receptor Tyrosine Kinase signaling is also required for lymphangiogenesis. Family members bind TEK Receptor Tyrosine Kinase as multimeric clusters but their oligomerization properties differ from one another and this is thought to provide a mechanistic basis for their distinct physiologic roles. [provided by RefSeq, Jul 2016]. |
mmu:11602; |
collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; receptor tyrosine kinase binding [GO:0030971]; signaling receptor binding [GO:0005102]; transmembrane receptor protein tyrosine kinase activator activity [GO:0030297]; vascular endothelial growth factor receptor binding [GO:0005172]; angiogenesis [GO:0001525]; cellular response to hypoxia [GO:0071456]; endoderm development [GO:0007492]; negative regulation of angiogenesis [GO:0016525]; negative regulation of apoptotic process [GO:0043066]; negative regulation of blood vessel endothelial cell migration [GO:0043537]; Notch signaling pathway [GO:0007219]; positive regulation of angiogenesis [GO:0045766]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; signal transduction [GO:0007165]; Tie signaling pathway [GO:0048014] |
15280392_Angiopoietin-3 binds to the cell membrane via heparan sulfate proteoglycans 15284220_Recombinant Ang3 formed disulfide-linked dimers. Ang3 was more potent than Ang4 in phosphorylating Akt in primary cultured mouse lung microvascular endothelial cells. It is a Tie2 agonist & causes corneal angiogenesis. 16790085_angiopoietin-4 inhibits angiogenesis and reduces interstitial fluid pressure 22267746_Angptl4 is a glucocorticoid-responsive mediator of fasting-induced intracellular lipolysis and stimulates cAMP signaling in adipocytes 25977256_Ang-4 also promoted survival of LECs. Thus, blocking Ang-4 may prune the draining lymphatic vasculature and decrease interstitial fluid pressure (IFP) by reducing vascular permeability. 30444491_This study identifies Angpt4 as the first growth factor for venous-specific development and its importance in venous remodeling, retinal fluid clearance and neuronal function. 34146541_ANGPTL4 regulates CD163 expression and Kuppfer cell polarization induced cirrhosis via TLR4/NF-kappaB pathway. 36190459_Cooperation of Angiopoietin-2 and Angiopoietin-4 in Schlemm's Canal Maintenance. |
ENSG00000101280 |
ANGPT4 |
90.648761 |
0.3798678252 |
-1.396431 |
0.28163366 |
24.291573 |
0.00000082800533365682918309993608071972737150190368993207812309265136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000075206781408830188819371158348570105545149999670684337615966796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
50.8681707809344 |
9.99334827938156 |
133.910185098695 |
18.6109084168388 |
| ENSMUSG00000027469 |
72119 |
Tpx2 |
protein_coding |
A2APB8 |
FUNCTION: Spindle assembly factor required for normal assembly of mitotic spindles. Required for normal assembly of microtubules during apoptosis. Required for chromatin and/or kinetochore dependent microtubule nucleation. Mediates AURKA localization to spindle microtubules. Activates AURKA by promoting its autophosphorylation at 'Thr-288' and protects this residue against dephosphorylation. TPX2 is inactivated upon binding to importin-alpha. At the onset of mitosis, GOLGA2 interacts with importin-alpha, liberating TPX2 from importin-alpha, allowing TPX2 to activates AURKA kinase and stimulates local microtubule nucleation. {ECO:0000250|UniProtKB:Q9ULW0, ECO:0000269|PubMed:18663142}. |
Acetylation;Apoptosis;Cell cycle;Cell division;Cytoplasm;Cytoskeleton;Isopeptide bond;Microtubule;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:72119; |
aster [GO:0005818]; axon hillock [GO:0043203]; cytoplasm [GO:0005737]; intercellular bridge [GO:0045171]; microtubule cytoskeleton [GO:0015630]; mitotic spindle [GO:0072686]; nuclear microtubule [GO:0005880]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; spindle pole [GO:0000922]; importin-alpha family protein binding [GO:0061676]; microtubule binding [GO:0008017]; protein kinase activator activity [GO:0030295]; protein kinase binding [GO:0019901]; activation of protein kinase activity [GO:0032147]; apoptotic process [GO:0006915]; cell division [GO:0051301]; microtubule nucleation [GO:0007020]; mitotic spindle assembly [GO:0090307]; negative regulation of microtubule depolymerization [GO:0007026]; regulation of mitotic spindle organization [GO:0060236]; spindle assembly [GO:0051225] |
21441895_Tpx2 redistributes from the nucleus to the apical process, and promotes nuclear migration during G2-phase by altering microtubule organization. 22266221_The findings indicated that Tpx2 is essential for maintaining genomic stability through its role in spindle regulation. 26165940_Study demonstrates that the Golgi resident protein GM130 activates the spindle assembly factor TPX2 to nucleate microtubules around the Golgi and further captures them to couple mitotic membranes to the spindle. 27753540_Bcl2l10, Tpx2, and Aurka co-localized on the meiotic spindles, and Bcl2l10 was present in the same complex with Tpx2. 28205572_Depleting TPX2 decreases microtubule formation frequency specifically at the tip and the base of the neurite, and these correlate precisely with the regions where active GTP-bound Ran proteins are enriched. 33892301_Inhibition of interleukin-1beta plays a protective role in Alzheimer's disease by promoting microRNA-9-5p and downregulating targeting protein for xenopus kinesin-like protein 2. 35273149_Downregulation of TPX2 impairs the antitumor activity of CD8+ T cells in hepatocellular carcinoma. |
ENSG00000088325 |
TPX2 |
2387.786764 |
2.1596951647 |
1.110828 |
0.10113243 |
117.451720 |
0.00000000000000000000000000228597455372348176545595875656346222273568347924465890980471741725280315337548442400361636828165501356124877929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000001456902276247534498074174820688304952588626408983116709539723176248865108006746993396518519148230552673339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3256.61382471843 |
228.474538796931 |
1507.90439219512 |
79.8862133753397 |
| ENSMUSG00000027496 |
20878 |
Aurka |
protein_coding |
P97477 |
FUNCTION: Mitotic serine/threonine kinase that contributes to the regulation of cell cycle progression (By similarity). Associates with the centrosome and the spindle microtubules during mitosis and plays a critical role in various mitotic events including the establishment of mitotic spindle, centrosome duplication, centrosome separation as well as maturation, chromosomal alignment, spindle assembly checkpoint, and cytokinesis (PubMed:9245792, PubMed:19075002). Required for normal spindle positioning during mitosis and for the localization of NUMA1 and DCTN1 to the cell cortex during metaphase (By similarity). Required for initial activation of CDK1 at centrosomes (By similarity). Phosphorylates numerous target proteins, including ARHGEF2, BORA, BRCA1, CDC25B, DLGP5, HDAC6, KIF2A, LATS2, NDEL1, PARD3, PPP1R2, PLK1, RASSF1, TACC3, p53/TP53 and TPX2 (By similarity). Regulates KIF2A tubulin depolymerase activity (By similarity). Required for normal axon formation (By similarity). Plays a role in microtubule remodeling during neurite extension (PubMed:19668197). Important for microtubule formation and/or stabilization (By similarity). Also acts as a key regulatory component of the p53/TP53 pathway, and particularly the checkpoint-response pathways critical for oncogenic transformation of cells, by phosphorylating and destabilizing p53/TP53 (By similarity). Phosphorylates its own inhibitors, the protein phosphatase type 1 (PP1) isoforms, to inhibit their activity (By similarity). Inhibits cilia outgrowth (By similarity). Required for cilia disassembly via phosphorylation of HDAC6 and subsequent deacetylation of alpha-tubulin (PubMed:20643351). Regulates protein levels of the anti-apoptosis protein BIRC5 by suppressing the expression of the SCF(FBXL7) E3 ubiquitin-protein ligase substrate adapter FBXL7 through the phosphorylation of the transcription factor FOXP1 (By similarity). {ECO:0000250|UniProtKB:A0A8I3S724, ECO:0000250|UniProtKB:O14965, ECO:0000269|PubMed:19075002, ECO:0000269|PubMed:19668197, ECO:0000269|PubMed:20643351, ECO:0000269|PubMed:9245792}. |
3D-structure;Alternative splicing;ATP-binding;Cell cycle;Cell division;Cell membrane;Cell projection;Cilium;Cilium biogenesis/degradation;Cytoplasm;Cytoskeleton;Isopeptide bond;Kinase;Membrane;Microtubule;Mitosis;Nucleotide-binding;Phosphoprotein;Proto-oncogene;Reference proteome;Serine/threonine-protein kinase;Transferase;Ubl conjugation |
|
|
mmu:20878; |
axon hillock [GO:0043203]; basolateral plasma membrane [GO:0016323]; centriole [GO:0005814]; centrosome [GO:0005813]; chromosome passenger complex [GO:0032133]; ciliary basal body [GO:0036064]; cytosol [GO:0005829]; germinal vesicle [GO:0042585]; glutamatergic synapse [GO:0098978]; meiotic spindle [GO:0072687]; microtubule cytoskeleton [GO:0015630]; microtubule organizing center [GO:0005815]; mitotic spindle [GO:0072686]; mitotic spindle pole [GO:0097431]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; postsynaptic density [GO:0014069]; pronucleus [GO:0045120]; spindle [GO:0005819]; spindle microtubule [GO:0005876]; spindle midzone [GO:0051233]; spindle pole [GO:0000922]; spindle pole centrosome [GO:0031616]; ATP binding [GO:0005524]; histone serine kinase activity [GO:0035174]; molecular function activator activity [GO:0140677]; protein heterodimerization activity [GO:0046982]; protein kinase activity [GO:0004672]; protein kinase binding [GO:0019901]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; ubiquitin protein ligase binding [GO:0031625]; anterior/posterior axis specification [GO:0009948]; apoptotic process [GO:0006915]; cell division [GO:0051301]; centrosome cycle [GO:0007098]; centrosome localization [GO:0051642]; cilium disassembly [GO:0061523]; liver regeneration [GO:0097421]; meiotic cell cycle [GO:0051321]; meiotic spindle organization [GO:0000212]; microtubule cytoskeleton organization [GO:0000226]; mitotic cell cycle [GO:0000278]; mitotic centrosome separation [GO:0007100]; mitotic spindle organization [GO:0007052]; negative regulation of apoptotic process [GO:0043066]; negative regulation of gene expression [GO:0010629]; negative regulation of protein binding [GO:0032091]; neuron projection extension [GO:1990138]; peptidyl-serine phosphorylation [GO:0018105]; positive regulation of mitochondrial fission [GO:0090141]; positive regulation of mitotic cell cycle [GO:0045931]; positive regulation of oocyte maturation [GO:1900195]; positive regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032436]; proteasome-mediated ubiquitin-dependent protein catabolic process [GO:0043161]; protein localization to centrosome [GO:0071539]; protein phosphorylation [GO:0006468]; regulation of cytokinesis [GO:0032465]; regulation of protein stability [GO:0031647]; response to wounding [GO:0009611]; spindle assembly involved in female meiosis I [GO:0007057]; spindle organization [GO:0007051] |
12559175_Cells overexpressing Aurora-A inappropriately enter anaphase despite defective spindle formation. Mitosis is subsequently arrested by failure to complete cytokinesis, resulting in multinucleation 12881723_Linkage analysis and haplotype mapping in interspecific mouse crosses (Mus musculus x Mus spretus) identified the gene encoding Aurora2 (Stk6 in mouse) as a candidate skin tumor susceptibility gene. 14695913_Aurora-A is a critical regulator of cell-cycle progression and microtubule organization during mouse oocyte meiotic maturation, fertilization, and early embryo cleavage. 15480417_roles of Aurora-A and the genetic pathways of Aurora-A-induced carcinogenesis 15923640_Results suggest that the transition from protection to degradation by the ubiquitin proteasome system at the end of mitosis is an important step in controlling the level of Aurora A protein during the cell cycle. 16617331_Aurora-A overexpression promotes cell proliferation and inhibits apoptosis, suggesting a novel mechanism that is closely related to malignant phenotype and anti-cancer drugs resistance of ESCC cells. 16772293_Gadd45a interacts with aurora-A and inhibits its kinase activity 17060449_Aurora-A-mediated phosphorylation of NDEL1 is essential for centrosomal separation and centrosomal maturation and for mitotic entry 17292827_Downregulation of Aurora-A kinase is associated with lymphomas 18345035_an essential physiological role of Aurora A in normal mitotic spindle assembly, chromosome alignment segregation and maintenance of viability in mammalian embryos. 18353803_AURK may be an oocyte histone-H3 kinase capable of regulating chromatin remodeling throughout oocyte meiosis. 18372918_p53 and p16 are critical in preventing mammary gland tumorigenesis in the human Aurora A overexpression mouse model 18677115_AURKA is a critical MTOC-associated component involved in resumption of meiosis, MTOC multiplication, proper spindle formation and the metaphase I-metaphase II transition. 18790738_analysis of the negative feedback regulation of Aurora-A via phosphorylation of Fas-associated factor-1 18801727_balanced Aurora A level is critical for maintaining genomic stability 19075002_Conditional Aurora-A ablation in cultured embryonic fibroblasts causes impaired mitotic entry and mitotic arrest with a profound defect in bipolar spindle formation. 19435814_Data show that the transgenic mouse model provides a useful system that allows the study of the physiologic effects of Aurora-A on liver regeneration and the genetic pathways of Aurora-A-mediated tumorigenesis in liver. 19668197_These findings demonstrate a surprising role of aPKC-Aurora A-NDEL1 pathway in microtubule remodelling during neurite extension. 20003375_Wild-type-Aurora-A enhances focus formation and aggregation of the RasV12 transformants in mouse NIH3T3 fibroblast derived cell lines over-expressing RasV12 20444701_Cdk1 activity is required for mitotic activation of aurora A during G2/M transition 21274965_AurkA plays critical roles in mouse oocyte meiosis by regulating spindle and chromosome dynamics 22306998_FBXL7 specifically interacts with Aurora A during mitosis but not in interphase, suggesting a regulatory role for FBXL7 in controlling Aurora A abundance during mitosis. 22513362_Results suggest a posttranslational regulatory network in which the Pten and Fbxw7 pathways appear to converge on the regulation of Aurora-A level. 22832491_The deletion of Aurora kinase Ain the developing epidermis altered centrosome function of basal keratinocytes and markedly impaired their ability to divide and stratify. 22837479_Aurora kinase A drives MTOC biogenesis but does not trigger resumption of meiosis in mouse oocytes. 22862944_Aurora kinase A regulates pluripotency through phosphorylation-mediated inhibition of p53-directed ectodermal and mesodermal gene expression 22875938_This study demonistrated that cross talk between Aurora-A and CDK5 during neuronal migration. 22939930_Epiblast ablation of Aurora A allows establishment of the anteroposterior axis but leads to failure to progress through gastrulation. 23810554_Aurora A kinase regulates mitotic spindle orientation and cell fate in the mammary epithelium. 23993973_Aurora kinase A promotes inflammation and tumorigenesis in mice and human gastric neoplasia. 24242071_Aurora-A is required for tumor formation in vivo, and the differential response observed in various tissues might have relevant implications in current therapeutic strategies aimed at inhibiting this kinase in the treatment of human cancer. 24273164_Aurora-A kinase does not regulate TACC3-chTOG complex formation, indicating that Aurora-A solely functions as a recruitment factor for the TACC3-chTOG complex to centrosomes and proximal mitotic spindles. 24875059_Wdr62 interacts with Aurora A to control mitotic progression, and loss of these interactions leads to mitotic delay and cell death of NPCs, which could be a potential cause of human microcephaly. 25227280_A novel function was identified for Aurora-A in controlling the polarization of macrophages and inflammation. 25318984_Reduced levels of Aurora A protein immediately post-thaw may be associated with the impaired oocyte maturation manifested by the delayed progression through meiosis 25670627_Aurka is required for hematopoiesis, but is dispensable for megakaryocyte endomitosis. 26174602_Data show that aurora-A kinase (AURKA) supports effective spindle formation in zygote. 26569382_aurora kinase A (AURKA) represents a new therapeutic target in primary myelofibrosis. 26658517_The Aurora kinase A and c-Myc expression and histone H3 phosphorylation level were comparatively higher in the cranial tumor than the caudal. 26880200_Ndel1 acts as a novel upstream regulator of the trichoplein-Aurora A pathway to inhibit primary cilia assembly. 27091106_Aurora A inhibition causes delocalized clustering of Lck at the immunological synapses and decreases its phosphorylation levels thus indicating Aurora A is required for maintaining Lck active during T-cell activation. 27102712_Augmented expression of Aurora kinase-A and Polo-like kinase-1 at the lactogenic switch likely mediates the formation of binucleated cells. 27207661_Suppression of neuroendocrine and NEPC development by ICT was associated with dose-dependent inhibitory effects on abnormally elevated IL-6/STAT3 and Aurora kinase A in vitro and in vivo 27213815_AURKA stabilizes MYC to promote tp53-altered liver tumor cell survival. 27624071_Our findings demonstrate that prolonged overexpression of Aurora-A can be a driver somatic genetic event in mammary adenocarcinomas associated with deregulated tumor-relevant pathways in the Aurora-A subset of human breast cancer 27632389_observations revealed that the alteration of PKB-GSK-3beta axis, Plk-1, and Aurora kinase-A expressions in HSPC compartment due to DNA damage response was associated with the proliferative impairment and apoptosis during aplastic anemia. 27753540_Bcl2l10, Tpx2, and Aurka co-localized on the meiotic spindles, and Bcl2l10 was present in the same complex with Tpx2. 28359584_The high sequence similarity among the AURK family members has made discerning the individual kinase functions in meiosis challenging. Technical limitations in specifically targeting AURKB or AURKC using small-molecule inhibitors and compensatory abilities in single-knockout animals add to this challenge...proper regulation of AURKA expression is crucial for spindle formation in meiosis 28764929_Aurora kinase inhibitor CCT137690 induces necrosis-like death in pancreatic ductal adenocarcinoma cells, via RIPK1, RIPK3, and MLKL signaling. 28935709_CIP2A acts as a scaffold for CEP192-mediated microtubule organizing center assembly by recruiting Plk1 and aurora A during meiotic maturation in mouse oocytes 28972102_Aurora A and Plk4 are rate-limiting factors contributing to microtubule growth as the acentriolar oocyte resumes meiosis. 29393405_ex vivo cells derived from teratomas exhibited high self-renewal capacity that was linked to Aurora-A kinase activity and gave rise to lung metastasis when injected into the tail vein of immunocompromised mice. 29842884_AURKA is expressed in several cell types in the testis. Spermatogonia and spermatocytes express AURKA as expected based on the known role of this kinase in cell division. Surprisingly, we also found AURKA localized to spermatids and the flagellum of spermatozoa. 30415701_in the absence of AURKC, AURKA localizes to chromosomes in a CPC-dependent manner. These data suggest that AURKC prevents AURKA from localizing to chromosomes by competing for CPC binding. This competition is important for adequate spindle length to support meiosis I. There is also a unique requirement for AURKB to negatively regulate AURKC to prevent aneuploidy. 30713094_EGFR-Aurka Signaling Rescues Polarity and Regeneration Defects in Dystrophin-Deficient Muscle Stem Cells by Increasing Asymmetric Divisions 31895686_CEP215 and AURKA regulate spindle pole focusing and aMTOC organization in mouse oocytes. 32636306_Aurora A regulation by reversible cysteine oxidation reveals evolutionarily conserved redox control of Ser/Thr protein kinase activity. 33475699_CEP55 promotes cilia disassembly through stabilizing Aurora A kinase. 33754019_Aurka loss in CD19(+) B cells promotes megakaryocytopoiesis via IL-6/STAT3 signaling-mediated thrombopoietin production. 33766930_Arid1a regulates cell cycle exit of transit-amplifying cells by inhibiting the Aurka-Cdk1 axis in mouse incisor. 33854138_High-throughput kinase inhibitor screening reveals roles for Aurora and Nuak kinases in neurite initiation and dendritic branching. 33901174_Aurora kinase A is essential for meiosis in mouse oocytes. 34152366_Loss of acentriolar MTOCs disrupts spindle pole Aurora A and assembly of the liquid-like meiotic spindle domain in oocytes. 34251762_Nuclear Aurora kinase A triggers programmed death-ligand 1-mediated immune suppression by activating MYC transcription in triple-negative breast cancer. 34518881_Aurora A Kinase (AURKA) is required for male germline maintenance and regulates sperm motility in the mouse. 34636397_Aurora kinase B inhibits aurora kinase A to control maternal mRNA translation in mouse oocytes. 34715887_Effects of Aurora kinase A on mouse decidualization via Stat3-plk1-cdk1 pathway. 35255953_Using ZINC08918027 inhibitor to determine Aurora kinase-chromosomal passenger complex isoforms in mouse oocytes. 36543138_BCL-xL inhibition potentiates cancer therapies by redirecting the outcome of p53 activation from senescence to apoptosis. |
ENSG00000087586 |
AURKA |
648.798080 |
2.6240222037 |
1.391780 |
0.11818075 |
136.587204 |
0.00000000000000000000000000000014844897164019114272119333988838090392399654315670753352019603132142097277821390162617920527310388933983631432056427001953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000123561460984884763575937672609049787572602091350364583337771197301905309045438625253909847856448323000222444534301757812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
945.092728651272 |
77.5054458779721 |
360.169486107621 |
22.9051745358965 |
| ENSMUSG00000027510 |
56190 |
Rbm38 |
protein_coding |
Q62176 |
FUNCTION: RNA-binding protein that specifically bind the 3'-UTR of CDKN1A transcripts, leading to maintain the stability of CDKN1A transcripts, thereby acting as a mediator of the p53/TP53 family to regulate CDKN1A. CDKN1A is a cyclin-dependent kinase inhibitor transcriptionally regulated by the p53/TP53 family to induce cell cycle arrest. Has the ability to induce cell cycle arrest in G1 and maintain the stability of CDKN1A transcripts induced by p53/TP53. Also acts as a mRNA splicing factor. Specifically regulates the expression of FGFR2-IIIb, an epithelial cell-specific isoform of FGFR2 (By similarity). Plays a role in myogenic differentiation. {ECO:0000250, ECO:0000269|PubMed:19817877}. |
Cell cycle;Cytoplasm;Differentiation;mRNA processing;mRNA splicing;Nucleus;Reference proteome;RNA-binding |
|
|
mmu:56190; |
cytosol [GO:0005829]; nucleus [GO:0005634]; mRNA 3'-UTR binding [GO:0003730]; mRNA binding [GO:0003729]; 3'-UTR-mediated mRNA stabilization [GO:0070935]; cell differentiation [GO:0030154]; DNA damage response, signal transduction by p53 class mediator resulting in cell cycle arrest [GO:0006977]; DNA damage response, signal transduction by p53 class mediator resulting in transcription of p21 class mediator [GO:0006978]; mRNA processing [GO:0006397]; mRNA stabilization [GO:0048255]; negative regulation of cell population proliferation [GO:0008285]; regulation of alternative mRNA splicing, via spliceosome [GO:0000381]; regulation of myotube differentiation [GO:0010830]; regulation of RNA splicing [GO:0043484]; RNA splicing [GO:0008380] |
21764855_loss of RNPC1 in mouse embryonic fibroblasts increased the level of p53 protein, leading to enhanced premature senescence in a p53-dependent manner 22371495_a novel mechanism by which HuR is regulated by RNPC1 via mRNA stability and HuR is a mediator of RNPC1-induced growth suppression. 22508983_knockdown of p73 or p21, another target of RNPC1, attenuates the inhibitory effect of RNPC1 on cell proliferation and premature senescence, whereas combined knockdown of p73 and p21 completely abolishes it 23836903_knockdown of MIC-1 can decrease RNPC1-induced cell growth suppression. 25512531_Rbm38 deficiency markedly decreases the tumor penetrance in mice heterozygous for p53 via enhanced p53 expression. 28850611_The hearts of Rbm38 -/- mice were mildly hypertrophic, but cardiac function was not affected. Furthermore, Rbm38 deficiency did not affect cardiac remodeling (i.e. hypertrophy, LV dilation and fibrosis) or performance (i.e. fractional shortening) after pressure-overload induced by transverse aorta constriction. 29330147_Rbm38 promotes lymphomagenesis.Rbm38 was required for Pten expression via stabilization of Pten mRNA through an AU-rich element in its 3'UTR.Rbm38 controls T-cell lymphomagenesis by jointly modulating mutant p53 and Pten. 32774357_Alternative Splicing of Cdh23 Exon 68 Is Regulated by RBM24, RBM38, and PTBP1. |
ENSG00000132819 |
RBM38 |
135.741104 |
0.4857895912 |
-1.041597 |
0.23431772 |
19.712662 |
0.00000900030219571606604370226506484087281023676041513681411743164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000672022563946799670209913957208414103661198168992996215820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
87.5927736827162 |
14.2891564134099 |
180.310108052003 |
21.3643087384237 |
| ENSMUSG00000027533 |
16592 |
Fabp5 |
protein_coding |
Q05816 |
FUNCTION: Intracellular carrier for long-chain fatty acids and related active lipids, such as endocannabinoids, that regulate the metabolism and actions of the ligands they bind (PubMed:8608126, PubMed:12540600). In addition to the cytosolic transport, selectively delivers specific fatty acids from the cytosol to the nucleus, wherein they activate nuclear receptors (By similarity). Delivers retinoic acid to the nuclear receptor peroxisome proliferator-activated receptor delta; which promotes proliferation and survival (PubMed:17512406). May also serve as a synaptic carrier of endocannabinoid at central synapses and thus controls retrograde endocannabinoid signaling (PubMed:29531087). Modulates inflammation by regulating PTGES induction via NF-kappa-B activation, and prostaglandin E2 (PGE2) biosynthesis during inflammation (PubMed:29440395). May be involved in keratinocyte differentiation (By similarity). {ECO:0000250|UniProtKB:Q01469, ECO:0000269|PubMed:12540600, ECO:0000269|PubMed:17512406, ECO:0000269|PubMed:29440395, ECO:0000269|PubMed:29531087, ECO:0000269|PubMed:8608126}. |
3D-structure;Acetylation;Cytoplasm;Direct protein sequencing;Disulfide bond;Lipid transport;Lipid-binding;Nucleus;Phosphoprotein;Reference proteome;Secreted;Synapse;Transport |
|
The protein encoded by this gene is part of the fatty acid binding protein family (FABP). FABPs are a family of small, highly conserved, cytoplasmic proteins that bind long-chain fatty acids and other hydrophobic ligands and participate in fatty acid uptake, transport, and metabolism. In humans this gene has been associated with psoriasis and type 2 diabetes. In mouse deficiency of this gene in combination with a deficiency in Fabp4 confers protection against atherosclerosis, diet-induced obesity, insulin resistance and experimental autoimmune encephalomyelitis (the mouse model for multiple sclerosis). Alternative splicing results in multiple transcript variants that encode different protein isoforms. The mouse genome contains many pseudogenes similar to this locus. [provided by RefSeq, Jan 2013]. |
mmu:16592; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; extracellular space [GO:0005615]; nucleus [GO:0005634]; postsynaptic density [GO:0014069]; synapse [GO:0045202]; fatty acid binding [GO:0005504]; identical protein binding [GO:0042802]; long-chain fatty acid transporter activity [GO:0005324]; retinoic acid binding [GO:0001972]; fatty acid transport [GO:0015908]; glucose homeostasis [GO:0042593]; glucose metabolic process [GO:0006006]; lipid metabolic process [GO:0006629]; lipid transport across blood-brain barrier [GO:1990379]; negative regulation of glucose transmembrane transport [GO:0010829]; phosphatidylcholine biosynthetic process [GO:0006656]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of peroxisome proliferator activated receptor signaling pathway [GO:0035360]; regulation of prostaglandin biosynthetic process [GO:0031392]; regulation of retrograde trans-synaptic signaling by endocanabinoid [GO:0099178]; regulation of sensory perception of pain [GO:0051930] |
12386159_covalent modification by 4-hydroxynonenal in vitro and in vivo: evidence for a role in antioxidant biology. (epithelial fatty acid-binding protein} 12479572_These results demonstrate that epidermal fatty acid binding protein is responsible for the water permeability barrier of the skin, although the molecular mechanism remains to be further elucidated. 12540600_plays a role in obesity and insulin resistance in knockout and transgenic mice 13129924_A-FABP and E-FABP bind to hormone-sensitive lipase with high affinity 15164767_E-FABP and/or H-FABP are involved in the mediation of dipalmitoyl phosphatidylcholine synthesis in wild type TII cells 15212142_epidermal-type fatty acid binding protein (E-FABP) is localized in two discrete cells in the adrenal gland, while brain-type fatty acid binding protein (B-FABP) is localized in the sustentacular cells enclosing the chromaffin cells in the adrenal medulla 16303844_Results suggest that the balance between lipolysis and lipogenesis in adipocytes is remodeled in the FABP4 null and FABP5 transgenic mice and is accompanied by the reprogramming of adipokine expression in fat cells and changes in plasma adipokines. 16400526_The present detection of E-FABP-immunopositivity selectively in macrophages of the atretic follicles suggests possible involvement of E-FABP and/or its ligand fatty acids in the process of follicular atresia. 16411018_E-FABP elevation may be necessary for the activation of cell motility within regenerative epidermis during wound healing. 16684508_Collectively, it is suggested that E-FABP may be a novel negative regulator of IL-12 production in DCs, and this regulation may be exerted via its involvement in the p38MAPK-mediated transcription of IL-12p35. 16885566_FABPs may play an important role in the primary protection against the cellular toxicity of cholesterol, free fatty acids, and/or lipid oxidants 17579051_Metabolic-inflammatory pathway cross-regulation by epidermal-type FABP contributes to adaptive immune responses and subsequent autoimmune inflammation. 18551191_Deletion of FABP4 and FABP5 in adipocytes resulted in reduced expression of inflammatory cytokines in macrophages, whereas the same deletions in macrophages led to enhanced insulin signaling and glucose uptake in adipocytes. 19494286_expression of E-FABP by CD4(+) T cells contributes to the control of IL-6 stimulation of the IL-21/retinoic acid-related orphan receptor/IL-17 pathway and to the Th17/regulatory T cell counterbalance 19787366_localized in populations of dendritic cells in the subepithelial domes, follicles and interfollicular regions of Peyer's patches and presumptive macrophages in germinal centers, and all M cells in the follicle-associated epithelium of mouse intestine 19956834_FABP5 siRNA delivered by atelocollagen to the external environment surrounding a tumour mass can effectively inhibit prostate cancer cell growth in nude mice 20238174_Decreased Fabp5 expression induce differentiating preadipocytes apoptosis via caspase-3 activation. 21068754_Epidermal FABP (FABP5) regulates keratinocyte differentiation by 13(S)-HODE-mediated activation of the NF-kappaB signaling pathway. 21474828_Mal1 plays a proatherogenic role by suppressing PPARgamma activity, which increases expression of CC chemokine receptor 2 by monocytes, promoting their recruitment to atherosclerotic lesions. 22581784_The data suggest that Fabp7 and Fabp5 have differential roles for proliferation and survival of the NSCs/NPCs during postnatal dentate gyrus neurogenesis. 22585040_FABP5 modulate cytokine production in the mouse thymic epithelial cells 23624787_FABP5 deficiency enhances susceptibility to H1N1 influenza A virus-induced lung inflammation. 23722546_Our findings establish that FABP5 is critical for mammary tumor development 23968980_Capillary endothelial FABP4/5 are required for (fatty acid)FA transport into FA-consuming tissues that include the heart. 24114376_FABP7 and FABP5 are differentially expressed in oligodendrocyte lineage cells and regulate their proliferation and/or differentiation. 24603714_FABP4/5 play an indispensable role in thermogenesis in brown adipose tissue and skeletal muscle. 24644281_Data indicate that the fatty acid-binding protein 5 (FABP5)/nuclear receptor peroxisome proliferator-activated receptor beta/delta (PPARbeta/delta) pathway induces the expression of cognition-associated genes. 24879443_Data indicate that epidermal fatty acid binding protein (E-FABP)-immunoreactivity was found in the entire cytoplasm and on the mitochondrial outer membrane. 25027319_The frameshift and missense mutations in FABP3, FABP5, and FABP7 genes have been identified in schizophrenia and autism spectrum disorder in humans and in mouse behavioral studies. 25268051_Circulating levels of GIP were significantly decreased in FABP5-deficient mice. 25628425_FABP5 contributes to the development of Diet-induced obesity in a gastric inhibitory polypeptide-dependent manner. 25779666_These studies show, for the first time, a correlation between FABP5 and EGFR in enhancing TNBC metastasis through a novel mechanism 26105806_loss of FABP5 promotes a more anti-inflammatory response. 26311517_Mice lacking FABP5 and FABP7, which exhibit highest affinities for endocannabinoids, possessed elevated levels of the endocannabinoid anandamide and the related N-acylethanolamines palmitoylethanolamide and oleoylethanolamide. 26625874_the balance between FABP4 and FABP5 in endothelial cells may be important in regulation of angiogenic versus quiescent phenotypes in blood vessels. 27588422_Following infection with Listeria monocytogenes, we observed similar clonal expansion, contraction and formation of memory CD8 T cells in WT and E-FABP-/- mice. Analysis of Listeria-specific CD4 T cells revealed no defect in the expansion, contraction, and formation of memory CD4 T cells in E-FABP-/- mice. Our data demonstrate that E-FABP is dispensable for antigen-specific T cell response following bacterial infection. 27852782_Genetic deletion of fatty acid-binding protein 5 (FABP5) in mice reduces uptake of exogenous docosahexaenoic acid (DHA) into brain endothelial cells and brain capillaries and reduces brain parenchymal levels of endogenous DHA. Therefore, FABP5 in the brain endothelial cell is a crucial contributor to the brain levels of DHA. Critically, lowered brain DHA levels in FABP5(-/-) mice occurred in tandem with cognitive deficits 28500502_Lack of PPARbeta/delta-expression, TUNEL reaction, RA receptor (RAR)beta, and cellular retinoic acid-binding protein (CRABP)-II were induced in E-FABP-positive septoclasts under RA excess, suggesting the growth arrest/cell-death of septoclasts, whereas cartilage-derived retinoic acid-sensitive protein (CD-RAP) inducing the cell growth arrest or morphological changes was induced in septoclasts under VA deficiency. 28911862_Using a yeast two-hybrid screen, immunoprecipitation, microscale thermophoresis analysis and cellular fractionation, we discovered that Fabp5 interacts with the calnexin cytoplasmic C-tail domain at the endoplasmic reticulum. These observations identify Fabp5 as a previously unrecognized calnexin binding partner. 29020626_Fabp4/5 confers protection against metabolic diseases but does not extend lifespan. 29105065_A 42.1 +/- 12.6% decrease in the BBB transport of (14) C-DHA in 8-month-old AD transgenic mice relative to wild-type mice was associated with a 34.5 +/- 6.7% reduction in FABP5 expression in isolated brain capillaries. Short-term spatial and recognition memory deficits were seen in AD mice on a 6-month n-3 fatty acid-depleted diet, but not those on control diet. 29457656_This study demonstrated the role of FABPs may play an important role in drug-seeking behavior under stressful conditions. 29554241_Fabp4/5 knockout mice demonstrate that glucose is preferentially utilized for biomass synthesis rather than ATP production during pressure-overload-induced cardiac hypertrophy and that the efficient supplementation of energy substrates may restore cardiac dysfunction caused by energy insufficiency. 29559340_our data suggest an unknown function of E-FABP in prevention of skin tumor development, and offer E-FABP as a therapeutic target for improving skin innate immunity in chemical-induced skin tumor prevention. 29570114_The findings of this study suggest that genetic deletion, but not pharmacological inhibiton, of FABP5 affects sucrose intake and antidepressant-related behavior. 30866899_FABP4/5 have crucial roles in nutrient homeostasis during prolonged fasting for maintaining exercise endurance capacity. 33124722_The Fabp5/calnexin complex is a prerequisite for sensitization of mice to experimental autoimmune encephalomyelitis. 33398436_Expression and enhancement of FABP4 in septoclasts of the growth plate in FABP5-deficient mouse tibiae. 33929718_FABP5 Deficiency Impairs Mitochondrial Function and Aggravates Pathological Cardiac Remodeling and Dysfunction. 33982279_Fatty acid-binding protein 5 controls lung tumor metastasis by regulating the maturation of natural killer cells in the lung. 33994513_FABP5 Is a Sensitive Marker for Lipid-Rich Macrophages in the Luminal Side of Atherosclerotic Lesions. 34160301_Downregulation of FABP5 Suppresses the Proliferation and Induces the Apoptosis of Gastric Cancer Cells Through the Hippo Signaling Pathway. 34449874_Fatty acid-binding protein 5 limits the generation of Foxp3(+) regulatory T cells through regulating plasmacytoid dendritic cell function in the tumor microenvironment. 34624687_A novel fatty acid-binding protein 5 and 7 inhibitor ameliorates oligodendrocyte injury in multiple sclerosis mouse models. 34876574_Oxidative stress-induced FABP5 S-glutathionylation protects against acute lung injury by suppressing inflammation in macrophages. 34942197_Epidermal Fatty AcidBinding Protein Mediates Depilatory-Induced Acute Skin Inflammation. 35202717_Environmental enrichment sex-dependently rescues memory impairment in FABP5 KO mice not mediated by brain-derived neurotrophic factor. 35408851_Salivary and Lacrimal Gland Alterations of the Epidermal Fatty Acid-Binding Protein (E-FABP) in Non-Obese Diabetic Mice. 36100327_Fatty acid-binding protein 5 aggravates pulmonary artery fibrosis in pulmonary hypertension secondary to left heart disease via activating wnt/beta-catenin pathway. 36384126_FABP5 controls macrophage alternative activation and allergic asthma by selectively programming long-chain unsaturated fatty acid metabolism. 36426981_FABP5 Deficiency Impaired Macrophage Inflammation by Regulating AMPK/NF-kappaB Signaling Pathway. 36481733_Fatty acid-binding proteins 3 and 5 are involved in the initiation of mitochondrial damage in ischemic neurons. |
ENSG00000164687 |
FABP5 |
1799.808922 |
7.3203124830 |
2.871905 |
0.09577138 |
840.444686 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000008688116753749778653250402368360462838221409458364391884421375373576278026252612 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000945267102807975900751465516810285595013416204988008234556107025807173151485558 |
Yes |
No |
3226.18042008486 |
214.323008090581 |
440.716216359111 |
23.2647707914736 |
| ENSMUSG00000027559 |
12350 |
Car3 |
protein_coding |
P16015 |
FUNCTION: Reversible hydration of carbon dioxide. {ECO:0000250|UniProtKB:P07451}. |
Acetylation;Cytoplasm;Direct protein sequencing;Glutathionylation;Lyase;Metal-binding;Phosphoprotein;Reference proteome;Zinc |
|
|
mmu:12350; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; carbonate dehydratase activity [GO:0004089]; hydro-lyase activity [GO:0016836]; nickel cation binding [GO:0016151]; phosphatase activity [GO:0016791]; zinc ion binding [GO:0008270]; one-carbon metabolic process [GO:0006730]; response to bacterium [GO:0009617]; response to ethanol [GO:0045471]; response to oxidative stress [GO:0006979] |
15509796_Mice lacking carbonic anhydrase III were viable and fertile and had normal life spans. 18322545_lack of proximal tubule cLCN5 in mice and men is associated with CAIII induction, increased cell proliferation, and oxidative stress 20820888_Car2, Car3, Car7, and Car15 mRNAs were barely within the detection limits in the mouse harderian gland. 22507175_We conclude that down-regulation of CAIII in preadipocytes enhances adipogenesis and that CAIII is a regulator of adipogenic differentiation which acts at the level of PPAR-gamma2 gene expression. 23233153_During inner ear development, transcripts of four cytosolic isozymes (Car1, Car2, Car3, and Car13), two membrane-bound isozymes (Car12 and Car14), and two CA-related proteins (Car8 and Car11) were expressed at higher levels than other isozymes. 25684186_The results suggest that elevated isoaspartate and CPS-1, and reduced CA-III levels could serve as biomarkers of hepatocellular injury. 28437447_Our results indicate that Car3 is not required for de novo lipogenesis, and Car3 knockout mice fed high-fat diet do not have significant differences in responses to various diets to wild type mice. 28928248_CAIII is highly expressed in osteocytes, is regulated by parathyroid hormone both in vitro and in vivo, and protects osteocytes from oxidative stress. 28933591_This study illustrates that skeletal muscle cell CAR3 is critical for CHRN homeostasis in the neuromuscular junction, and its deficiency leads to accelerated degradation of CHRN (cholinergic receptor nicotinic) and development of myasthenia gravis. 29559661_these data clearly suggests that CAIII serves as an important antioxidant critical in protecting NP cells against oxidative stress-induced injury. 31390226_CAIII transgenic hearts showed significantly less decrease in cardiac function than that of wild-type control animals. |
ENSG00000164879 |
CA3 |
5013.832433 |
5.5202087580 |
2.464723 |
0.05381792 |
1994.817860 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
8425.86757334864 |
314.389274384401 |
1526.36756013451 |
45.1502408826167 |
| ENSMUSG00000027570 |
12841 |
Col9a3 |
protein_coding |
A2ACT7 |
|
Proteomics identification;Reference proteome;Signal |
|
|
|
basement membrane [GO:0005604]; collagen type IX trimer [GO:0005594]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; extracellular matrix structural constituent [GO:0005201]; extracellular matrix organization [GO:0030198]; female gonad development [GO:0008585]; male gonad development [GO:0008584] |
26429145_Results show that absence of Col IX induces early developmental, structural and biomechanical alterations in both vertebral body and intervertebral disc which eventually cause severe degenerative changes in the aging spine. 33526661_SOX9-COL9A3-dependent regulation of choroid plexus epithelial polarity governs blood-cerebrospinal fluid barrier integrity. |
|
|
26.529939 |
2.3387444702 |
1.225734 |
0.48293590 |
6.423560 |
0.01126161682076192056822794285153577220626175403594970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0370650275310815888185622668515861732885241508483886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.1556498065033 |
13.0123133840204 |
16.3145868617279 |
4.3359044367236 |
| ENSMUSG00000027654 |
71878 |
Fam83d |
protein_coding |
Q9D7I8 |
FUNCTION: Through the degradation of FBXW7, may act indirectly on the expression and downstream signaling of MTOR, JUN and MYC (By similarity). May play also a role in cell proliferation through activation of the ERK1/ERK2 signaling cascade (By similarity). May also be important for proper chromosome congression and alignment during mitosis through its interaction with KIF22 (By similarity). {ECO:0000250|UniProtKB:Q9H4H8}. |
Alternative splicing;Cell cycle;Cell division;Cytoplasm;Cytoskeleton;Mitosis;Phosphoprotein;Proto-oncogene;Reference proteome |
|
|
mmu:71878; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; intercellular bridge [GO:0045171]; microtubule cytoskeleton [GO:0015630]; mitotic spindle [GO:0072686]; mitotic spindle pole [GO:0097431]; spindle [GO:0005819]; kinesin binding [GO:0019894]; microtubule binding [GO:0008017]; protein kinase binding [GO:0019901]; cell division [GO:0051301]; cell migration [GO:0016477]; cell population proliferation [GO:0008283]; epithelial to mesenchymal transition [GO:0001837]; metaphase plate congression [GO:0051310]; positive regulation of cell cycle G1/S phase transition [GO:1902808]; protein localization to mitotic spindle [GO:1902480]; regulation of ERK1 and ERK2 cascade [GO:0070372]; regulation of protein catabolic process [GO:0042176]; regulation of TOR signaling [GO:0032006]; signal transduction [GO:0007165] |
Human_homologues 18485706_interaction partner of the chromokinesin Kid that is required for the generation of polar ejection forces and chromosome congression 24344117_Results demonstrate that FAM83D has prognostic value for breast cancer patients and is a novel oncogene in breast cancer development that at least in part acts through mTOR hyper-activation by inhibiting FBXW7. 25646692_Taken together, these results demonstrate that FAM83D is a novel oncogene in HCC development and may constitute a potential therapeutic target in HCC. 26125229_upregulation of FAM83D, a potential oncotarget gene, may be triggered by epigenetic events and can contribute to hepatocarcinogenesis 26652654_NMR-derived secondary chemical shifts and relaxation properties show that the Chica LC8 binding domain is essentially disordered with a dynamically restricted segment in one linker between motifs. 26678035_Higher level of FAM83D expression is positively correlated with an increase in genome instability in many cancer. 27769048_FAM83D promotes HCC recurrence by promoting CD44 expression and CD44+ CSCs malignancy. 28407575_FAM83D knockdown up-regulated the protein expression level of F-box and WD repeat domain-containing 7 (FBXW7), but diminished the Notch1 protein expression level. 30910840_Overexpressed in tumors, family with sequence similarity 83 member D protein FAM83D (FAM83D) is associated with gender, AJCC stage, tumor recurrence and survival in hepatocellular carcinoma (HCC). 31037837_FAM83D promotes ovarian cancer progression and its potential application in diagnosis of invasive ovarian cancer. 32006253_FAM83D promotes epithelial-mesenchymal transition, invasion and cisplatin resistance through regulating the AKT/mTOR pathway in non-small-cell lung cancer. 32092437_Fam83d modulates MAP kinase and AKT signaling and is induced during neurogenic skeletal muscle atrophy. 32228656_circFOXM1 promotes proliferation of non-small cell lung carcinoma cells by acting as a ceRNA to upregulate FAM83D. 34308751_Immune implication of FAM83D gene in hepatocellular carcinoma. 35489022_miRNA-142-3p functions as a potential tumor suppressor directly targeting FAM83D in the development of ovarian cancer. |
ENSG00000101447 |
FAM83D |
377.512174 |
2.3155108389 |
1.211331 |
0.14364662 |
70.510345 |
0.00000000000000004578592447587048625417668284964500362700356227521093777088623255622223950922489166259765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000014004240996314442212227533560530657178372143309430519764191558351740241050720214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
529.413634198699 |
52.7999306009131 |
228.637942570082 |
17.6742910379761 |
| ENSMUSG00000027660 |
20482 |
Skil |
protein_coding |
Q60665 |
FUNCTION: May have regulatory role in cell division or differentiation in response to extracellular signals. {ECO:0000250}. |
Alternative splicing;Coiled coil;Isopeptide bond;Phosphoprotein;Reference proteome;Ubl conjugation |
|
This gene encodes a member of a small family of proteins that play a key role in the response of cells to extracellular growth signals. The encoded protein regulates members of the transforming growth factor beta signaling pathway. It is highly expressed in certain cancer cells, where it may have both tumor-suppressing and tumor-promoting roles. Alternatively spliced transcript variants encoding multiple isoforms have been observed for this gene. [provided by RefSeq, Nov 2012]. |
mmu:20482; |
acrosomal vesicle [GO:0001669]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; PML body [GO:0016605]; protein-containing complex [GO:0032991]; transcription regulator complex [GO:0005667]; chromatin binding [GO:0003682]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; identical protein binding [GO:0042802]; protein domain specific binding [GO:0019904]; protein-containing complex binding [GO:0044877]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; SMAD binding [GO:0046332]; blastocyst formation [GO:0001825]; cell differentiation [GO:0030154]; extrinsic apoptotic signaling pathway via death domain receptors [GO:0008625]; intrinsic apoptotic signaling pathway in response to DNA damage [GO:0008630]; lens fiber cell differentiation [GO:0070306]; lymphocyte homeostasis [GO:0002260]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of cell differentiation [GO:0045596]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512]; positive regulation of axonogenesis [GO:0050772]; positive regulation of extrinsic apoptotic signaling pathway via death domain receptors [GO:1902043]; positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage [GO:1902231]; regulation of cell cycle [GO:0051726]; response to cytokine [GO:0034097]; response to growth factor [GO:0070848]; skeletal muscle tissue development [GO:0007519]; spermatogenesis [GO:0007283]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
12861029_Sno is a significant negative regulator of antiproliferative TGF-beta signaling in both T cells and other cell types in vivo 16966324_SnoN is directly regulated by sumoylation leading to the enhancement of the ability of SnoN to repress transcription in a promoter-specific manner 17074815_SnoN plays both pro-tumorigenic and antitumorigenic roles at different stages of mammalian malignant progression 17510063_Arkadia induces degradation of SnoN and c-Ski in addition to Smad7. 18212064_SnoN plays an essential role in AFP repression by positively regulating mSin3A protein levels. p53 and SnoN are both needed to recruit the mSin3A corepressor. 19267613_These data indicate that SnoN is expressed in a cell-specific manner during ovarian follicular development, atresia, and luteinization and that SnoN might play essential roles in these physiological processes. 19526521_The authors conclude that somatic and germ cells at all differentiation stages are actively transducing TGFbeta superfamily signals but that responses to these ligands may be selectively modulated by SnoN2. 19745809_SnoN functions as a tumour suppressor by inducing premature senescence. 20457602_The endogenous SnoN plays a role in regulating ADAM12 expression in response to TGFbeta1. 21659477_mRNA for positive regulators of Fshb expression, such as Fos and Jun, were up-regulated at slower pulse frequencies than a number of potential negative regulators, such as the corepressors Skil, Crem, and Tgif1. 22767605_SnoN mediates a negative feedback mechanism evoked by TGF-beta to inhibit BMP signaling and, subsequently, hypertrophic maturation of chondrocytes. 22833129_SnoN, a negative regulator of TGF-beta signaling, coordinates TGF-beta and prolactin signaling to control alveologenesis and lactogenesis. 24019535_Loss of SnoN is associated with partial embryonic lethality, mostly due to defects in angiogenesis in both the yolk sac and embryo body 30030373_SnoN plays an important in vivo role in adipocyte differentiation and white adipose tissue development in vivo by decreasing activity in the activin/Smad2 signaling pathway. 30425119_SnoN has been implicated in distinct aspects of the development of postmitotic neurons, this study identifies a novel function for SnoN in neuronal precursors in the mammalian brain 34473197_Arkadia-SKI/SnoN signaling differentially regulates TGF-beta-induced iTreg and Th17 cell differentiation. |
ENSG00000136603 |
SKIL |
317.932379 |
0.4214235682 |
-1.246657 |
0.15678433 |
62.647041 |
0.00000000000000247276200756216658357618450626700297275892530962648763193101331125944852828979492187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000644950529095666429495252070239011618488699992268742278156423708423972129821777343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
185.620414546478 |
20.2119573113767 |
440.46045011983 |
34.4625237151147 |
| ENSMUSG00000027669 |
14696 |
Gnb4 |
protein_coding |
P29387 |
FUNCTION: Guanine nucleotide-binding proteins (G proteins) are involved as a modulator or transducer in various transmembrane signaling systems. The beta and gamma chains are required for the GTPase activity, for replacement of GDP by GTP, and for G protein-effector interaction. |
Acetylation;Phosphoprotein;Reference proteome;Repeat;Transducer;WD repeat |
|
|
mmu:14696; |
cell body [GO:0044297]; cytoplasm [GO:0005737]; heterotrimeric G-protein complex [GO:0005834]; myelin sheath [GO:0043209]; GTPase activity [GO:0003924]; protein-containing complex binding [GO:0044877]; signaling receptor complex adaptor activity [GO:0030159]; G protein-coupled receptor signaling pathway [GO:0007186] |
Human_homologues 12454992_Gbeta4 subunit is coexpressed in physiologically ON-type cone bipolar cells with Ggamma13 and Gbeta3 but is not present in OFF-type bipolar cells. 12782285_Gbeta4 is widely expressed, located on chromosome 3 with a genomic structure like Gbeta1 to Gbeta3, but different from Gbeta5 18815590_Intron-1 haplotypes of GNB4 may, thus, serve as predictive markers for progression and survival of patients suffering from bladder cancer. 18815590_Observational study of gene-disease association. (HuGE Navigator) 19414374_Intron-1 haplotypes of GNB4 is associated with colorectal carcinoma. 19414374_Observational study of gene-disease association. (HuGE Navigator) 23434117_Analysis of GNB4 in an additional 88 unrelated CMT individuals uncovered another de novo mutation. 25916507_Suggest Gbeta4gamma1 as a modulator of M3 muscarinic receptor signaling. 28642160_A novel missense GNB4 variant causes Charcot-Marie-Tooth disease F in two patients, who represent the first record of the disease in the Japanese population. 30103729_GNB4 is important for growth of breast cancer cells and a potential target for treatment 32747927_Guanine nucleotide-binding protein subunit beta-4 promotes gastric cancer progression via activating Erk1/2. 34398348_High G protein subunit beta 4 protein level is correlated to poor prognosis of urothelial carcinoma. 35756485_Prognostic and Immunological Value of GNB4 in Gastric Cancer by Analyzing TCGA Database. |
ENSG00000114450 |
GNB4 |
239.692681 |
0.3463671274 |
-1.529626 |
0.23274678 |
42.175085 |
0.00000000008345651647596080409672169566156650286070650679448590381070971488952636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000013409384588205009224998477318827570792247882991432561539113521575927734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
122.798449013074 |
19.896847262652 |
354.532631150271 |
41.6858870342132 |
| ENSMUSG00000027699 |
13605 |
Ect2 |
protein_coding |
Q07139 |
FUNCTION: Guanine nucleotide exchange factor (GEF) that catalyzes the exchange of GDP for GTP. Promotes guanine nucleotide exchange on the Rho family members of small GTPases, like RHOA, RHOC, RAC1 and CDC42. Required for signal transduction pathways involved in the regulation of cytokinesis. Component of the centralspindlin complex that serves as a microtubule-dependent and Rho-mediated signaling required for the myosin contractile ring formation during the cell cycle cytokinesis. Regulates the translocation of RHOA from the central spindle to the equatorial region. Plays a role in the control of mitotic spindle assembly; regulates the activation of CDC42 in metaphase for the process of spindle fibers attachment to kinetochores before chromosome congression. Involved in the regulation of epithelial cell polarity; participates in the formation of epithelial tight junctions in a polarity complex PARD3-PARD6-protein kinase PRKCQ-dependent manner. Plays a role in the regulation of neurite outgrowth. Inhibits phenobarbital (PB)-induced NR1I3 nuclear translocation. Stimulates the activity of RAC1 through its association with the oncogenic PARD6A-PRKCI complex in cancer cells, thereby acting to coordinately drive tumor cell proliferation and invasion. Also stimulates genotoxic stress-induced RHOB activity in breast cancer cells leading to their cell death. {ECO:0000269|PubMed:17904126, ECO:0000269|PubMed:21350944}. |
3D-structure;Acetylation;Alternative splicing;Cell cycle;Cell division;Cell junction;Cytoplasm;Cytoskeleton;Differentiation;Guanine-nucleotide releasing factor;Isopeptide bond;Neurogenesis;Nucleus;Phosphoprotein;Protein transport;Proto-oncogene;Reference proteome;Repeat;Tight junction;Transport;Ubl conjugation |
|
|
mmu:13605; |
bicellular tight junction [GO:0005923]; cell cortex [GO:0005938]; cell-cell junction [GO:0005911]; centralspindlin complex [GO:0097149]; cleavage furrow [GO:0032154]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; midbody [GO:0030496]; mitotic spindle [GO:0072686]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; GTPase activator activity [GO:0005096]; guanyl-nucleotide exchange factor activity [GO:0005085]; protein homodimerization activity [GO:0042803]; small GTPase binding [GO:0031267]; activation of GTPase activity [GO:0090630]; activation of protein kinase activity [GO:0032147]; bicellular tight junction assembly [GO:0070830]; cell differentiation [GO:0030154]; cell morphogenesis [GO:0000902]; cellular response to calcium ion [GO:0071277]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to ionizing radiation [GO:0071479]; intracellular signal transduction [GO:0035556]; mitotic cytokinesis [GO:0000281]; nervous system development [GO:0007399]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cytokinesis [GO:0032467]; positive regulation of GTPase activity [GO:0043547]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of protein import into nucleus [GO:0042307]; protein homooligomerization [GO:0051260]; protein transport [GO:0015031]; regulation of attachment of spindle microtubules to kinetochore [GO:0051988]; regulation of cytokinesis, actomyosin contractile ring assembly [GO:2000431]; regulation of protein kinase activity [GO:0045859] |
14645260_ECT2 has a role in activating the Rho signaling pathways leading to malignant transformation 19996184_indispensable role of the maturation-promoting factor ECT2 pathwy in polar body extrusion in mouse oocytes 20141573_results demonstrate the functional role of Ect2 in postnatal proliferating cells in the two lobes of the pituitary, thereby suggesting roles in developmental growth of the mammalian pituitary 21350944_Ect2 may play a role in neuronal differentiation and brain development in vivo. 22387001_It was shown that the guanine exchange factors GEF-H1 and ECT2 must be downregulated for megakaryote polyploidization. The first (2N-4N) endomitotic cycle requires GEF-H1 downregulation, whereas subsequent cycles (>4N) require ECT2 downregulation. 27074761_These results suggest that Ect2 plays roles in multiple aspects of DNA damage response. 29486195_Loss of ECT2 expression is associated with myocardial Polyploidization Creating a Barrier to Heart Regeneration. 31145635_the oncogene ECT2 contributes to epithelial cell reprogramming in idiopathic pulmonary fibrosis |
ENSG00000114346 |
ECT2 |
866.141220 |
2.1345451773 |
1.093929 |
0.13442782 |
64.888282 |
0.00000000000000079267042176549194948223309372604879655575344172060980874050528655061498284339904785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000217157479574316076059303163506078690400951259481532318318386387545615434646606445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1169.84410535792 |
109.16184125624 |
548.053101813796 |
38.8352326139583 |
| ENSMUSG00000027715 |
12428 |
Ccna2 |
protein_coding |
P51943 |
FUNCTION: Cyclin which controls both the G1/S and the G2/M transition phases of the cell cycle. Functions through the formation of specific serine/threonine kinase holoenzyme complexes with the cyclin-dependent protein kinases CDK1 and CDK2. The cyclin subunit confers the substrate specificity of these complexes and differentially interacts with and activates CDK1 and CDK2 throughout the cell cycle. {ECO:0000250|UniProtKB:P20248}. |
3D-structure;Acetylation;Cell cycle;Cell division;Cyclin;Cytoplasm;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:12428; |
cyclin A2-CDK2 complex [GO:0097124]; cyclin-dependent protein kinase holoenzyme complex [GO:0000307]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; female pronucleus [GO:0001939]; male pronucleus [GO:0001940]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; cyclin-dependent protein serine/threonine kinase regulator activity [GO:0016538]; protein domain specific binding [GO:0019904]; protein kinase binding [GO:0019901]; animal organ regeneration [GO:0031100]; cell cycle G1/S phase transition [GO:0044843]; cell division [GO:0051301]; cellular response to cocaine [GO:0071314]; cellular response to estradiol stimulus [GO:0071392]; cellular response to hypoxia [GO:0071456]; cellular response to insulin-like growth factor stimulus [GO:1990314]; cellular response to leptin stimulus [GO:0044320]; cellular response to luteinizing hormone stimulus [GO:0071373]; cellular response to nitric oxide [GO:0071732]; cellular response to platelet-derived growth factor stimulus [GO:0036120]; cochlea development [GO:0090102]; DNA-templated transcription [GO:0006351]; G2/M transition of mitotic cell cycle [GO:0000086]; mitotic cell cycle phase transition [GO:0044772]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of fibroblast proliferation [GO:0048146]; post-translational protein modification [GO:0043687]; Ras protein signal transduction [GO:0007265]; regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0000079]; regulation of DNA replication [GO:0006275]; response to glucagon [GO:0033762] |
12646244_Data show that the presence of the truncated form of cyclin A2 in differentiating embryonic stem cells is not a function of cell density but is correlated with differentiation. 14699150_cyclin A levels are regulated by c-Fos in osteoblast growth control 15159393_critical role in mediating cardiomyocyte mitosis, a role that may significantly impact upon clinical treatment of damaged myocardium. 15214576_expression pattern during the first three cell cycles of mouse embryo development 15615710_B-Myb represses SMC elastin gene expression and cyclin A plays a role in the developmental regulation of elastin gene expression in the aorta 16137671_recruitment of maternal cyclin A2 mRNA following egg activation is linked to embryonic gene activation 17495221_Expression of cyclin A2 augments endogenous regenerative mechanisms via induction of enhanced cell proliferative capacity. Ability of cultured transgenic cardiomyocytes to undergo cytokinesis provides support for the regenerative capacity of cyclin A2. 17637565_Results indicate that the NFAT1 transcription factor represses cyclin A2 expression in lymphocytes, and may act as a silencer of gene transcription during the cell cycle. 18077453_PtdIns(4,5)P2 down-regulation caused a delay in phorbol ester-induced S phase entry and that this was at least in part channeled through cyclin A2 at the transcriptional level. 18621517_cyclin A may induce S phase arrest associated with apoptosis in 3-methylcholanthrene-induced fibrosarcomas. 19001847_REVIEW.Some cyclins exhibit unique patterns of expression in germ cells that suggest possible concomitant distinct functions; embryonic lethality has obviated understanding of the germline functions of cyclin A2 and cyclin B1 19221029_cyclin A2-Cdk1 is a key regulator of S-phase programs 19233286_Results identify the phosphorylated residues in mouse Cyclin-A2 and CDK2 recombinant proteins, after individual expression and after formation of the heterodimer complex. 19264965_MLL5 plays an integral role in novel chromatin regulatory mechanisms that suppress inappropriate expression of S-phase-promoting genes and maintain expression of determination genes in quiescent cells. 19592082_Study found that acute ablation of cyclin A in fibroblasts did not affect cell proliferation, in contrast, cyclin A function was essential for cell-cycle progression of hematopoietic and embryonic stem cells. 20126974_cyclin A expression is inhibited by bicalutamide both in androgen-dependent and -independent cell lines 20164416_CCN1 is a novel component of the extracellular matrix that activates proinflammatory genes in macrophages, implicating its role in regulating macrophage function during inflammation. 20802531_transcription of the gene for cyclin-A2 appears to be a direct target of JDP2 in the suppression of cell proliferation 20926513_Study suggested that OGP may increase the bone formation in OPG-deficient mice by stimulating MSC proliferation rather than differentiation, and probably by triggering CDK2/cyclin A pathway. 21135363_CCN1 is a novel, endogenous 'parent compound' for chemotaxis modulation. 22243368_Cyclin A expression confirmed a prolifer-ative response in cirrhotic livers in response to mitogens. 23122964_The requirement of cyclin A2 for entry into meiosis I and for sister chromatid segregation in meiosis II in mouse oocytes. 23796713_decreased miR-124 expression could increase CCNA2 in cell and animal model of HD and is involved in deregulation of cell cycle in STHdh(Q111)/Hdh(Q111) cells 24012615_AMPKalpha1 controlled cyclin A2 transcription mainly through the ATF/CREB element 24184637_Conditional knockout of cyclin A2 or the SHH proliferative target Nmyc in CGNP also resulted in PC neuron dyslamination 24802190_Loss of Cdk2 and cyclin A2 impairs cell proliferation and tumorigenesis. 24831015_The activity of PCTK3 is increased via interaction with cyclin A2 and phosphorylation by PKA. 24879294_Data indicate that cyclin A2-depleted epithelial cells adopt a fibroblastic morphology and undergo epithelial to mesenchymal transition (EMT). 25615569_Ccn2 is a regulator of the transition through cytokinesis during terminal erythropoiesis. 25668568_Loss of CCNA2 decreases cell proliferation and induces cerebellar cortical dyslamination. 25993989_Cyclin A2 modulates epithelial-mesenchymal transition via beta-catenin and phospholipase C pathways. 27020049_hese data suggest that mmu-miR-125b decreases NO production in activated macrophages at least partially by suppressing eEF2K and CCNA2 expression. 27425845_We demonstrate that CCNA2-null progenitors suffer abnormal DNA repair, and implicate Cyclin A2 in double-strand break repair. Cyclin A2's DNA repair functions are conserved among cell lines, neural progenitors, and hippocampal neurons. We further demonstrate that neuronal CCNA2 ablation results in learning and memory deficits in aged mice. 27657745_These data unveils the critical functions of cyclin A2 in regulating mammalian erythropoiesis. 27708105_cyclin A2 controlled Mre11 abundance through a C-terminal RNA binding domain that selectively and directly binds Mre11 transcripts to mediate polysome loading and translation. 28003426_inhibition of PDK4 activity in Hepatocellular carcinoma cells increased cyclin E1, cyclin A2, and E2F1 proteins. 28380054_Meioc-deficient spermatocytes that have initiated synapsis mis-express CYCLIN A2, which is normally expressed in mitotic spermatogonia, suggesting a failure to properly transition to a meiotic cell cycle program. 28819014_cyclin A2 mediates the fidelity ofmeiosis II by maintaining microtubule dynamics during the rapid formation of the meiosis II spindle. 30579783_cyclin A2 functions in a noncanonical/non-cell cycle regulatory role to maintain adult pyramidal neuron ribostasis. 30848072_The p53/miRNAs/Ccna2 pathway serves as a novel regulator of cellular senescence: Complement of the canonical p53/p21 pathway. 31980707_PDZRN3 protects against apoptosis in myoblasts by maintaining cyclin A2 expression. 32420805_Cyclin A2 is essential for mouse gonocyte maturation. 33332285_Cyclin A2 maintains colon homeostasis and is a prognostic factor in colorectal cancer. 34864392_Inducible expression of Oct-3/4 reveals synergy with Klf4 in targeting Cyclin A2 to enhance proliferation during early reprogramming. |
ENSG00000145386 |
CCNA2 |
2862.555544 |
2.0048252651 |
1.003477 |
0.07070051 |
198.715147 |
0.00000000000000000000000000000000000000000000398308855688155815243441277528485000210147249468736540939724151335447247169885679561473869404841771581945888717145830282273522016112110577523708343505859375000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000005515491354401808271815466446486122766497175722325662739227801013121131732692431207728965649081246999723156091734177497443170068436302244663238525390625000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3824.45762970102 |
187.495866058688 |
1907.62641324855 |
71.1097425389531 |
| ENSMUSG00000027776 |
16159 |
Il12a |
protein_coding |
P43431 |
FUNCTION: Heterodimerizes with IL12B to form the IL-12 cytokine or with EBI3/IL27B to form the IL-35 cytokine. IL-12 is primarily produced by professional antigen-presenting cells (APCs) such as B-cells and dendritic cells (DCs) as well as macrophages and granulocytes and regulates T-cell and natural killer-cell responses, induces the production of interferon-gamma (IFN-gamma), favors the differentiation of T-helper 1 (Th1) cells and is an important link between innate resistance and adaptive immunity (PubMed:8766560). Mechanistically, exerts its biological effects through a receptor composed of IL12R1 and IL12R2 subunits. Binding to the receptor results in the rapid tyrosine phosphorylation of a number of cellular substrates including the JAK family kinases TYK2 and JAK2. In turn, recruited STAT4 gets phosphorylated and translocates to the nucleus where it regulates cytokine/growth factor responsive genes (By similarity). As part of IL-35, plays essential roles in maintaining the immune homeostasis of the liver microenvironment and functions also as an immune-suppressive cytokine (PubMed:18033300, PubMed:30197594). Mediates biological events through unconventional receptors composed of IL12RB2 and gp130/IL6ST heterodimers or homodimers. Signaling requires the transcription factors STAT1 and STAT4, which form a unique heterodimer that binds to distinct DNA sites (By similarity). {ECO:0000250|UniProtKB:P29459, ECO:0000269|PubMed:18033300, ECO:0000269|PubMed:30197594, ECO:0000269|PubMed:8766560}. |
Cytokine;Disulfide bond;Glycoprotein;Growth factor;Reference proteome;Secreted;Signal |
|
|
mmu:16159; |
cell surface [GO:0009986]; cytoplasm [GO:0005737]; endoplasmic reticulum lumen [GO:0005788]; endosome lumen [GO:0031904]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi lumen [GO:0005796]; interleukin-12 complex [GO:0043514]; interleukin-12 receptor complex [GO:0042022]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; interleukin-12 beta subunit binding [GO:0042163]; interleukin-12 receptor binding [GO:0005143]; interleukin-27 binding [GO:0045513]; protein heterodimerization activity [GO:0046982]; cell migration [GO:0016477]; cell population proliferation [GO:0008283]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to virus [GO:0098586]; defense response to protozoan [GO:0042832]; extrinsic apoptotic signaling pathway [GO:0097191]; immune response [GO:0006955]; negative regulation of blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:1903588]; negative regulation of interleukin-17 production [GO:0032700]; negative regulation of protein secretion [GO:0050709]; negative regulation of smooth muscle cell proliferation [GO:0048662]; negative regulation of vascular endothelial growth factor signaling pathway [GO:1900747]; positive regulation of cell adhesion [GO:0045785]; positive regulation of dendritic cell chemotaxis [GO:2000510]; positive regulation of lymphocyte proliferation [GO:0050671]; positive regulation of mononuclear cell proliferation [GO:0032946]; positive regulation of natural killer cell activation [GO:0032816]; positive regulation of natural killer cell mediated cytotoxicity [GO:0045954]; positive regulation of natural killer cell mediated cytotoxicity directed against tumor cell target [GO:0002860]; positive regulation of natural killer cell proliferation [GO:0032819]; positive regulation of NK T cell activation [GO:0051135]; positive regulation of NK T cell proliferation [GO:0051142]; positive regulation of receptor signaling pathway via JAK-STAT [GO:0046427]; positive regulation of smooth muscle cell apoptotic process [GO:0034393]; positive regulation of T cell differentiation [GO:0045582]; positive regulation of T cell mediated cytotoxicity [GO:0001916]; positive regulation of T cell proliferation [GO:0042102]; positive regulation of type II interferon production [GO:0032729]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531]; response to lipopolysaccharide [GO:0032496]; response to UV-B [GO:0010224]; T cell proliferation [GO:0042098]; T-helper 1 cell activation [GO:0035711] |
11801672_During Mycobacterium tuberculosis infection mice lacking IL-12p35 exhibit a moderate ability to control bacterial growth, are able to generate antigen-specific IFN-gamma responses, and survive infection longer. 11920565_Autocrine IL-10 impairs dendritic cell (DC)-derived immune responses to mycobacterial infection by suppressing DC trafficking to draining lymph nodes and local IL-12 production. 12189243_The development and pathogenesis of experimental autoimmune encephalitis in IL-12p35 knockout mice 12223525_HSV-infected cells generate intermediary products that signal recruited inflammatory cells, which themselves are not HSV-infected, to generate IL-12. 12396623_Mucosal Ad-IFN-gamma gene transfer can effectively attenuate established allergen-induced airway inflammation and airway hyperreactivity, predominantly through an IL-12- and STAT4-dependent mechanism. 12429708_IL-12 is required for the migration of T cells into neoplasm masses 12471147_The IL-12p35-deficient C57BL/6 mouse is susceptible to induction of experimental autoimmune encephalomyelitis by myelin oligodendrocyte glycoprotein peptide 35-55, while IL-12p40-deficient animals are completely resistant. 12697150_IL-12 p35 mRNA accumulates in the Golgi apparatus as an endoglycosidase H-sensitive glycosylated integral membrane protein 12937153_ApoE(-/-)/IL-4(-/-) mice showed a 58% and 64% decrease in disease in aortic arch compared to apoE(-/-) (P |
ENSG00000168811 |
IL12A |
70.920840 |
0.4468292724 |
-1.162204 |
0.33910561 |
11.531603 |
0.00068422972693431077188114253218031990400049835443496704101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0032929501423897070133683939729962730780243873596191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
44.2630756348948 |
10.5001065294852 |
99.060375786983 |
16.8996290426063 |
| ENSMUSG00000027784 |
242083 |
Ppm1l |
protein_coding |
Q8BHN0 |
FUNCTION: Acts as a suppressor of the SAPK signaling pathways by associating with and dephosphorylating MAP3K7/TAK1 and MAP3K5, and by attenuating the association between MAP3K7/TAK1 and MAP2K4 or MAP2K6. {ECO:0000269|PubMed:12556533}. |
Alternative splicing;Hydrolase;Magnesium;Manganese;Membrane;Metal-binding;Protein phosphatase;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:242083; |
membrane [GO:0016020]; metal ion binding [GO:0046872]; myosin phosphatase activity [GO:0017018]; protein serine/threonine phosphatase activity [GO:0004722]; MAPK cascade [GO:0000165]; protein dephosphorylation [GO:0006470]; transmembrane receptor protein serine/threonine kinase signaling pathway [GO:0007178] |
12556533_in the absence of IL-1-induced signal, PP2Cepsilon contributes to keeping the TAK1 signaling pathway in an inactive state by associating with and dephosphorylating TAK1. 18165232_CERT is a physiological substrate of PP2Cepsilon and that dephosphorylation of CERT by PP2Cepsilon may play an important role in the regulation of ceramide trafficking from the ER to the Golgi apparatus. 25369058_This study provides the first in vivo evidence that PP2Ce is an essential regulator of normal lactation, possibly involving IRE1 signaling and endoplasmic reticulum stress regulation in mammary epithelium. 27680501_Study provided evidence that PPM1L is involved in the development of the central nervous system. |
ENSG00000163590 |
PPM1L |
23.843738 |
0.2830783583 |
-1.820727 |
0.57846389 |
9.781722 |
0.00176255165271217229330802389597465662518516182899475097656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0076205469128900965927742028327429579803720116615295410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.0214674237754 |
4.12675015232457 |
35.4017434783623 |
10.0504861459754 |
| ENSMUSG00000027797 |
13175 |
Dclk1 |
protein_coding |
Q9JLM8 |
FUNCTION: Probable kinase that may be involved in a calcium-signaling pathway controlling neuronal migration in the developing brain. May also participate in functions of the mature nervous system (By similarity). {ECO:0000250}. |
Alternative splicing;ATP-binding;Developmental protein;Differentiation;Kinase;Neurogenesis;Nucleotide-binding;Phosphoprotein;Reference proteome;Repeat;Serine/threonine-protein kinase;Transferase |
|
This gene encodes a member of the protein kinase superfamily and the doublecortin family. The protein encoded by this gene contains two N-terminal doublecortin domains, which bind microtubules and regulate microtubule polymerization, a C-terminal serine/threonine protein kinase domain, which shows substantial homology to Ca2+/calmodulin-dependent protein kinase, and a serine/proline-rich domain in between the doublecortin and the protein kinase domains, which mediates multiple protein-protein interactions. The microtubule-polymerizing activity of the encoded protein is independent of its protein kinase activity. The encoded protein is involved in several different cellular processes, including neuronal migration, retrograde transport, neuronal apoptosis and neurogenesis. This gene is up-regulated by brain-derived neurotrophic factor and associated with memory and general cognitive abilities. Multiple transcript variants generated by two alternative promoter usage and alternative splicing have been found, but the biological validity of some variants has not been determined. These variants encode different isoforms, which are differentially expressed and have different kinase activities. [provided by RefSeq, Sep 2010]. |
mmu:13175; |
axon [GO:0030424]; growth cone [GO:0030426]; postsynaptic density [GO:0014069]; ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; axon extension [GO:0048675]; axonogenesis [GO:0007409]; brain development [GO:0007420]; central nervous system projection neuron axonogenesis [GO:0021952]; dendrite morphogenesis [GO:0048813]; forebrain development [GO:0030900]; intracellular signal transduction [GO:0035556]; negative regulation of protein localization to nucleus [GO:1900181]; neuron migration [GO:0001764]; neuron projection morphogenesis [GO:0048812]; peptidyl-serine phosphorylation [GO:0018105]; protein localization to nucleus [GO:0034504]; protein phosphorylation [GO:0006468]; response to virus [GO:0009615] |
11473121_DCLK is a physiological substrate for the cysteine protease calpain. In neurons cleavage of DCLK by calpain represents a calcium responsive mechanism to regulate localization of the DCLK kinase domain 11479289_DCLK is a physiological substrate for activated caspases. Cleavage of DCLK is necessary for neuronal apoptosis to proceed. 11597766_Multiple transcripts generated by the DCAMKL gene are expressed in the rat hippocampus 11884394_genomic organization underlying the splice variants of DCLK and the expression profile of two splice variants affecting the kinase domain of DCLK and CPG16 (candidate plasticity gene 16). 14741399_Two splice variants of the DCLK gene are differentially expressed in areas of the adult hippocampus and display functional differences 16387637_DCLK controls mitotic division by regulating spindle formation and also determines the fate of neural progenitors during cortical neurogenesis. 16387638_Dcx and Dclk may directly or indirectly regulate microtubule-based vesicle transport, a process critical to both neuronal migration and axon outgrowth. 16387639_Doublecortin-like kinase (Dclk) gene functions in a partially redundant pathway with Dcx in the formation of axonal projections across the midline and migration of cortical neurons. 17313568_Doublecortin-like, a slice variant of DCLK1 and a microtubule-associated protein expressed in radial glia, is crucial for neuronal precursor division and radial process stability. 17313568_unique role of the DCLK gene in mitotic spindle integrity during early neurogenesis 17409237_In the embryonic brain, virtually all of the cells expressing doublecortin-like kinase contained no stem-like cells. 17409252_DCLK has a distinct role in cortical interneuron migration; Dclk distribution is different in mutant mice compared with wild-type controls. 17975023_The microtubule associated protein Doublecortin-like regulates the transport of the glucocorticoid receptor in neuronal progenitor cells. 18055444_Doublecortin and CaM kinase-like-1 (DCAMKL-1) was used to examine radiation-induced stem cell apoptosis and adenomatous polyposis coli (APC)/multiple intestinal neoplasia (min) mice to determine the effects of APC mutation on DCAMKL-1 expression. 18075264_Interestingly, Dclk1 was expressed in CA3 neurons, including in the heterotopic pyramidal layer of Dcx knockout animals, but is presumably not able to compensate for a lack of Dcx. 18231966_The microtubule-associated proteins doublecortin and doublecortin-like are differentially expressed, particularly during early corticogenesis, consistent with their different functional roles. 20659433_The DCLK gene product affects glutamatergic neuronal transmission in response to neurological stimuli. 20705078_A truncated, constitutively active form of DCLK-short is highly expressed in the hippocampus, cortex and amygdala of transgenic mice. These mice spend less time, move less in the open arms of the maze and show a reduction in the number of rim dips. 21688022_Gastric tuft cells express DCLK1 and are expanded in hyperplasia 22605579_Our findings define DCL as a novel marker for specific aspects of adult neurogenesis, which partly overlap with doublecortin 22857951_The neurons lacking Dcx and/or its structurally conserved paralogue, doublecortin-like kinase 1 (Dclk1), show impaired Kif1a-mediated transport of Vamp2, a cargo of Kif1a, with decreased run length. 23385585_Doublecortin-like kinases are critical regulators of dendritic development by means of their specific targeting to the distal dendrites, and their local control of dendritic growth and synapse maturation. 23918955_DCAMKL1 represses osteoblast activation by antagonizing Runx2, the master transcription factor in osteoblasts. 24040120_DCLK1 regulates pluripotency and angiogenic factors via microRNA-dependent mechanisms in pancreatic cancer. 24096005_Pancreatic neoplasms in mice contain morphologically and functionally distinct subpopulations, expressing DCLK1, that have cancer stem cell-like properties. 24123696_Dclk1 plays a functional role critical in the epithelial restorative response. 24368703_These results suggest that crypt epithelial cell Dclk1 expression can be used as one potential marker to evaluate the early survival of intestinal stem cells following severe radiation injury. 25211188_Dclk1 is critically involved in facilitating intestinal tumorigenesis by enhancing pluripotency and EMT factors in Apc mutant intestinal tumors and it also provides a potential therapeutic target for the treatment of colorectal cancer. 26073039_loss of parietal cells leads to the reversible emergence of a novel Dclk1-expressing sensory cell population in the gastric mucosa. 26285154_Dclk1 plays an important role in regulating colonic inflammatory response and colonic epithelial integrity 26526391_Dclk1 promotes axonal regeneration, neuronal survival, and growth core reformation via microtubule stabilization, prevention of F-actin destabilization and synergy with mTOR. 27058937_Dclk1 contributes functionally to the pathogenesis of pancreatic cancer; Dclk1 marks quiescent pancreatic progenitors that are candidates for the origin of pancreatic cancer 27876863_intestinal tuft cells play an important role in regulating the ATM mediated DNA damage response, for epithelial cell survival/self-renewal via a Dclk1 dependent mechanism 29577277_High alternative splice variants of DCLK1 mark cancer stem cells, promote self-renewal and drug-resistance in kidney cancer. 30478383_Dclk1 in tuft cells promotes inflammation-driven epithelial restitution and mitigates chronic colitis. 31748376_we reveal an important function of DCLK1 in the adult brain, and highlight its important role regulating alpha-Synuclein levels. 32994174_Doublecortin-Like Is Implicated in Adult Hippocampal Neurogenesis and in Motivational Aspects to Escape from an Aversive Environment in Male Mice. 33393460_Visualization of stem cell activity in pancreatic cancer expansion by direct lineage tracing with live imaging. 33485913_Spatial and temporal diversity of DCLK1 isoforms in developing mouse brain. 33627749_Notch signaling drives development of Barrett's metaplasia from Dclk1-positive epithelial tuft cells in the murine gastric mucosa. 34343377_Doublecortin-like expressing astrocytes of the suprachiasmatic nucleus are implicated in the biosynthesis of vasopressin and influences circadian rhythms. 34610960_DCLK1-Short Splice Variant Promotes Esophageal Squamous Cell Carcinoma Progression via the MAPK/ERK/MMP2 Pathway. 35910805_DCLK1 promotes colorectal cancer stemness and aggressiveness via the XRCC5/COX2 axis. 36064052_Doublecortin-Like Kinase 1 Facilitates Dendritic Spine Growth of Pyramidal Neurons in Mouse Prefrontal Cortex. |
ENSG00000133083 |
DCLK1 |
1331.650008 |
2.1976562839 |
1.135966 |
0.08494039 |
177.103397 |
0.00000000000000000000000000000000000000020791894502050764667486126021881237577962983529383979233383757549831350782580966394189528900100060588001377182010287469893228262662887573242187500000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000239925861405482765566132436518626351218540407244484544257166847630405362154862711168001561828119327229869028883513237815350294113159179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1804.78454626451 |
106.320239799459 |
821.231490789913 |
37.2211762175441 |
| ENSMUSG00000027859 |
18049 |
Ngf |
protein_coding |
P01139 |
FUNCTION: Nerve growth factor is important for the development and maintenance of the sympathetic and sensory nervous systems (PubMed:20036257). Extracellular ligand for the NTRK1 and NGFR receptors, activates cellular signaling cascades to regulate neuronal proliferation, differentiation and survival (PubMed:22649032). The immature NGF precursor (proNGF) functions as ligand for the heterodimeric receptor formed by SORCS2 and NGFR, and activates cellular signaling cascades that lead to inactivation of RAC1 and/or RAC2, reorganization of the actin cytoskeleton and neuronal growth cone collapse (PubMed:22155786). In contrast to mature NGF, the precursor form (proNGF) promotes neuronal apoptosis (in vitro) (PubMed:20036257). Inhibits metalloproteinase-dependent proteolysis of platelet glycoprotein VI (By similarity). Binds lysophosphatidylinositol and lysophosphatidylserine between the two chains of the homodimer (PubMed:22649032, PubMed:26144237). The lipid-bound form promotes histamine relase from mast cells, contrary to the lipid-free form (PubMed:22649032). {ECO:0000250|UniProtKB:P01138, ECO:0000269|PubMed:20036257, ECO:0000269|PubMed:22155786, ECO:0000269|PubMed:22649032, ECO:0000269|PubMed:26144237}. |
3D-structure;Cleavage on pair of basic residues;Direct protein sequencing;Disulfide bond;Endosome;Glycoprotein;Growth factor;Lipid-binding;Metalloenzyme inhibitor;Metalloprotease inhibitor;Protease inhibitor;Reference proteome;Secreted;Signal |
|
|
mmu:18049; |
axon [GO:0030424]; dendrite [GO:0030425]; endoplasmic reticulum lumen [GO:0005788]; endosome lumen [GO:0031904]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; synaptic vesicle [GO:0008021]; cysteine-type endopeptidase activator activity involved in apoptotic process [GO:0008656]; death receptor agonist activity [GO:0038177]; growth factor activity [GO:0008083]; lipid binding [GO:0008289]; metalloendopeptidase inhibitor activity [GO:0008191]; nerve growth factor receptor binding [GO:0005163]; transmembrane receptor protein tyrosine kinase activator activity [GO:0030297]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; adult locomotory behavior [GO:0008344]; axon extension [GO:0048675]; circadian rhythm [GO:0007623]; extrinsic apoptotic signaling pathway in absence of ligand [GO:0097192]; extrinsic apoptotic signaling pathway via death domain receptors [GO:0008625]; memory [GO:0007613]; modulation of chemical synaptic transmission [GO:0050804]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of peptidase activity [GO:0010466]; negative regulation of type B pancreatic cell apoptotic process [GO:2000675]; nerve development [GO:0021675]; nerve growth factor signaling pathway [GO:0038180]; neuron apoptotic process [GO:0051402]; neuron projection development [GO:0031175]; neuron projection morphogenesis [GO:0048812]; peripheral nervous system development [GO:0007422]; positive regulation of axon extension [GO:0045773]; positive regulation of cell growth [GO:0030307]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of collateral sprouting [GO:0048672]; positive regulation of DNA binding [GO:0043388]; positive regulation of DNA-binding transcription factor activity [GO:0051091]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of neuron maturation [GO:0014042]; positive regulation of neuron projection development [GO:0010976]; positive regulation of neurotrophin TRK receptor signaling pathway [GO:0051388]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of protein autophosphorylation [GO:0031954]; positive regulation of protein binding [GO:0032092]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein ubiquitination [GO:0031398]; positive regulation of Ras protein signal transduction [GO:0046579]; positive regulation of stem cell proliferation [GO:2000648]; protein phosphorylation [GO:0006468]; regulation of neuron differentiation [GO:0045664]; regulation of neurotransmitter secretion [GO:0046928]; regulation of release of sequestered calcium ion into cytosol [GO:0051279]; sensory perception of pain [GO:0019233]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
12485622_The presence of Nerve Growth Factor 1 in supporting cells in the normal and degenerating cochlea indicates their role in the sustenance of inner hair cells. 14651951_Our results indicated that icv infusion of beta-NGFm stimulates the expression levels of ChAT gene in the striatum of old rats. 15625712_analysis of NGF-TrkA receptor interaction by the novel antagonistic anti-TrkA monoclonal antibody MNAC13 16319926_NGF rapidly increases membrane expression of TRPV1 heat-gated ion channels 16762005_Although activated hepatic stellate cells may produce the factors that regulate liver regeneration, the de novo NGF production by regenerating hepatocytes may induce the death of HSCs via p75NTR, leading to termination of hepatic regeneration. 17093052_Results describe the construction of a mutated pro-nerve growth factor resistant to degradation and suitable for biophysical and cellular utilization. 17594942_a decrease or increase in NGF is accompanied by a decrease or increase in trGSH levels; elevated NGF and/or trGSH levels attenuate some of the responses to LPS such as anorexia and cytokine production 17653039_Autocrine modulation of differentiation and response of vernal keratoconjunctivitis(VKC) fibroblasts to NGF exposure with downregulation of type I collagen and upregulation of MMP9 expression supports a relevant role for NGF in tissue remodeling of VKC. 17881125_The observation that the brain leptin and NGF receptors were altered by stress suggests a functional role for these molecules in neuroendocrine and cardiovascular response to stress events. 18162309_Regulation of sympathetic neuron differentiation by endogenous nerve growth factor. 18329889_Results decribe the effects of ganglioside GM1 on the expression of nerve growth factor (NGF), Trk-A receptor, proinflammatory cytokines and on autoimmune diabetes onset in non-obese diabetic (NOD) mice. 18349525_Immunostaining for NGF was noted in articular chondrocytes. 18367547_Nerve growth factor induced the cyclin D1 promoter via Sp1, nuclear factor-kappaB, and cAMP-response element/activated transcription factor sites. 18635195_The present study pinpoints loop II of NGF to be an important structural determinant for NGF biological activity mediated by TrkA receptor. 18657279_NGF enhances sympathetic neurite outgrowth. 18709654_Pro-NGF, as well as mature NGF, binding to p75(NTR) plays an important role in inflammation-induced hyperalgesia. Interference in the binding may provide a therapeutic approach for the treatment of inflammatory pain. 18725298_study reports that, during early post-natal development, levels of pro-apoptotic proNGF in mdx mouse iris, but not in the submandibular gland, are higher than in the wild-type 18774569_Nerve growth factor shows a direct stimulatory effect on expression of WT1, whereas FSH attenuated basal and nerve growth factor-stimulated WT1 protein expression, which most likely depends on FSH responsiveness according to the follicle growth stage. 19170059_NGF was characterized as a potent promoter of mineralization during dentin formation 19193386_bone marrow stromal cells treatment benefits on experimental autoimmune encephalomyelitis mice may be fostered by decreasing the cellular expression of proNGF and p75, thereby reducing oligodendrocyte death. 19319189_knockdown of RGS14 inhibited both nerve growth factor- and basic fibrobast growth factor-mediated neuronal differentiation of PC12 cells 19334503_afobazole increases the NGF level in a final concentration of 10(-8) M and the BDNF level in final concentrations from 10(-8) to 10(-5) M. 19376199_data suggest that in the brain of mdx mice, an upregulation of the nerve growth factor-nerve growth factor receptors (TrkA and p75) system might be involved directly in the angiogenic response taking place in muscular dystrophy 19389705_the relative level of proNGF receptors determines whether NGF precursor exhibits neurotrophic or apoptotic activity 19444944_Unlike most other peripheral tissues, sympathetic efferents in adult mammalian arteries/arterioles do not undergo collateral growth in response to increased levels of smooth muscle-derived NGF. 19515925_study identified Wnt5a as a key downstream effector of NGF in mediating axonal branching and growth in developing sympathetic neurons 19728147_These results indicate that AMP-activated protein kinase supports the NGF-induced viability of human HeLa cells to glucose starvation. 19816194_Nerve growth factor expression is increased in the dorsal root ganglia of a db/db mouse model of type 2 diabetes and pain compared to controls. 19844589_NGF is able to induce TLR4/TLR9 overexpression in vernal keratoconjunctivitis-endothelial cells. 20018664_Data show that endogenous NGF induces DNA replication in a subpopulation of differentiating retinal ganglion cells that express both the neurotrophin receptor p75 and the E2F1 transcription factor, but that lack the retinoblastoma protein. 20032263_chronic urothelial NGF overexpression in transgenic mice leads to neuronal proliferation, focal increases in urinary bladder mast cells, increased urinary bladder reflex activity, and pelvic hypersensitivity 20036257_explored the binding characteristics of proNGF to sortilin using surface plasmon resonance and cell-based assays and determined that calcium ions promote the formation of a stable ternary complex of proNGF-sortilin-p75NTR 20045424_The levels of the Ngf are decreased in the hippocampus of the Mecp2(1lox) null mice,a mouse model of Rett syndrome. 20075049_Nerve growth factor promotes the proliferation of lung epithelium in vitro and the renewal of Clara cells following lung injury by naphthalene in vivo. 20350782_These results indicate that NGF is an important mediator of OA pain and that TrkAd5 represents a potent novel analgesic in this condition. 20360245_NGF elicits pleiotropic beneficial actions in the post-myocardial infarction heart. NGF should be considered as a candidate for therapeutic cardiac regeneration. 20449688_Data describe PACAP, vasoactive intestinal polypeptide, and PAC1, VPAC1, VPAC2 transcripts or protein expression in urothelium and detrusor smooth muscle and lumbosacral dorsal root ganglia in NGF-overexpressing and wildtype mice. 20507613_Results suggest that galectin-3/Mac-2+ microglia play an important role in the pathogenesis of diffuse axonal injury both acutely and chronically and that they mediate their effects, at least in part by releasing nerve growth factor. 20680101_These observations indicate that human corneal endothelial cells are receptive to the action of NGF and that these cells may regulate NGF activity through autocrine/paracrine mechanisms. 20736176_Cot/tpl2 has an important role in the NGF, G-CSF, and GM-CSF production and myeloperoxidase activity in the acute inflammatory response process and its implication in inflammatory hypernociception 21085186_NGF acts as a local stress mediator in perceived stress and allergy. increased NGF message contributes to worsening of cutaneous inflammation. 21145972_Our study indicates that neurotrophin activation of Trk stimulates an increase in VEGF transcription that is mediated by induction of HIF-1alpha. 21165786_NGF and NT-3 heterozygous mice exhibit higher levels of CXCR4 the cerebellum and hypothalamus only. 21385399_NGF- beta can be used as an indicator for the severity of adenomyosis. 21816277_Study reports that actin depolymerization is essential for initiation of NGF/TrkA endosome trafficking and that a Rac1-cofilin signaling module associated with TrkA early endosomes supports their maturation to retrograde transport-competent endosomes. 21816457_NGF exerts profibrotic activities in the airways by inducing type III collagen production in fibroblasts 21818348_The distinct structural properties of proNGF as compared to NGF agree with and rationalise a different functional role of the precursor. 22168511_that NGF derived from bronchial and alveolar epithelium plays an important role in airway hyperresponsiveness after chronic exposure to mite antigen 22187379_Therapeutic potential of NGF for the prevention of cardiomyopathy in diabetic subjects. 22520925_Letter: NGF-p75 and neuropsin/KLK8 pathways may cooperate in regulation of epidermal homeostasis in inflamed skin. 22700375_Overexpression of mouse NGF in urothelium & detrusor affected transcription of PAC1, VPAC1, VPAC2, PAPCAP, VIP & other peptides normally & in cyclophosphamide-induced cystitis. The changes were tissue- and disease-duration dependent. 22808101_Expression of NGF in hippocampus, cortex, and adrenal gland of wild type animal tended to decrease following spaceflight. 22903500_The HTM1 heterodimer of 2 NGF muteins binds p75 and TrkA on opposite sides of the heterodimer, but not 2 TrkA receptors, supporting the ligand passing of NGF from p75 to TrkA via a transient heteroreceptor complex of p75-NGF-TrkA. 22999926_Our results suggest that BDNF-TrkB but not NGF-TrkA signaling is involved in the brain repair after ICH, and early proper treadmill exercise might promote this repair process. 23100411_This study demonstrated that both TrkA and NGF support the survival of only a subset of basal forebrain cholinergic neuron during brain development. 23322532_elevated levels of NGF in target tissues stimulate sympathetic and sensory axonal sprouting 23426701_Data suggest that expression of NGF is down-regulated in wounded skin (epidermis) in diabetes; among the growth factors investigated, only expression of NGF was down-regulated in healing skin wounds of diabetic mice as compared to nondiabetic mice. 23466052_Data suggest that diet factors (i.e., olive pomace polyphenols) up-regulate NGF/TrkA (proto-oncogene trk) and BDNF/TrkB (brain derived neurotrophic factor/receptor) in hippocampus/olfactory bulb and down-regulate NGF/BDNF in frontal cortex/striatum. 23538417_ProNGFNGF imbalance triggers learning and memory deficits, neurodegeneration and spontaneous epileptic-like discharges in transgenic mice. 23538529_PIP5Kalpha acts as a negative regulator of nerve growth factor-induced neurite outgrowth by inhibiting PI3K/Akt signaling pathway in PC12 cells. 23623989_Independent of genotype, folate deficiency affects NGF levels in the frontal cortex, amygdala and hippocampus. 23633509_proNGF selectively promotes the growth of neurites from a subset of NGF-responsive neurons by a p75(NTR)-dependent mechanism during postnatal development when the axons of these neurons are ramifying within their targets in vivo 23825664_calcineurin/NFAT pathway mediates the upregulation of PAI-1 by NGF 23934089_beta-NGF gene transfection promotes the differentiation of bone marrow stromal stem cells into neurons through regulation of AKT and MAPKs signaling pathways. 23989259_mechanical stimulation may attenuate NGFbeta signaling through Rac1 24040063_Data suggest that proNGF may appeal a new pathway or possible mechanism underlying microglial toxicity in the neuroinflammation and a potential target for therapeutic manipulation of the neurodegenerative diseases. 24338202_The increased NGF concentrations abolish Sema3A-induced inhibitory effect on axon outgrowth, while they have no effect on Sema3A-induced collapse rate. 24438745_Proinflammatory cytokines in osteoarthritis (OA) joints and the increased mechanical loading of cartilage may mediate OA pain via the stimulation of NGF expression and release by chondrocytes 24787014_NGF induces removal of active caspase-3 in a lysosome-dependent manner. 24937592_These results indicate that NGF exerts antileishmanial effect by stimulating hydrogen peroxide production in macrophages. 25049196_Ginger extract has a synaptogenic effect via NGF-induced ERK/CREB activation, resulting in memory enhancement. 25088915_Results indicate that analgesic effect of CB1 activation may in part be due to inhibition of NGF-induced sensitization of TRPV1 and also that the effect of CB1 activation is at least partly mediated by attenuation of NGF-induced increased PI3 signaling 25211588_These findings suggest that overexpression of NGF in the ovary may suffice to cause both reproductive and metabolic alterations characteristic of polycystic ovary-like syndrome (PCOS) and support the hypothesis that sympathetic hyperactivity may contribute to the development and/or progression of PCOS. 25231981_NGF facilitates depolarization-induced NFAT activation by stimulating PI3K/Akt signaling, inactivating GSK3beta, and thereby slowing NFATc3 export from the nucleus 25397406_NGF exhibits anti-oxidative and hepatoprotective effects and is suggested to be therapeutically applicable in treating cholestatic liver diseases. 25460199_iron accumulation induces NGF expression in hepatocytes, which in turn leads to LSEC defenestration via TrkA 25496838_Despite being highly conserved, NGF and proNGF of mouse and human origins show distinct properties.Special care must be taken in performing experiments with cross-species systems. 25647301_NGF effects on parasympathetic nerves may regulate airway smooth muscle. 25650182_The NGF-induced up-regulation of TRPV1 via the increased synthesis and release of endogenous CGRP leads to improved cardiac performance in I/R-injured diabetic heart. 25734767_In vivo ozone(O3)-induced elevations in both nerve growth factor (NGF) and substance P (SP) are mediated by the inflammatory cytokine interleukin-1beta. Further, inhibition of NGF reduced O3 induced increases of SP in both the lung BALF and lung tissue. 26066836_In E-Reeler retinas, NGF was significantly increased in retinal ganglion cells and glial cells. E-Reeler retinal bipolar cells and RGCs overexpress NGF and p75(NTR) as a protective endogenous response to Reelin deprivation. 26347141_Identify nerve growth factor as a binding partner for MOG and demonstrate that this interaction is capable of sequestering NGF from TrkA-expressing neurons to modulate axon growth and survival. 26631553_NGF negatively regulates growth cone retrograde actin flow on laminin. 26683342_Nerve growth factor negatively regulates bone marrow granulopoiesis during small intestinal inflammation 27076121_NGF signalling directly controls basal APP phosphorylation, subcellular localization and BACE cleavage. 27306411_functional PAP(thorn) neurons are essential for the analgesic effect, which is mediated by NGF-trkA signaling. 27342083_NGF-OE mice exhibit age-dependent increases in Substance P and CGRP in the urothelium and hyperinnervation of the bladder. 27424144_These data support a role for islet NGF in fine-tuning insulin secretion. 27437725_NGF facilitates OVA with lowLPS-induced maturation of mouse BMDCs through LPS-up-regulated p75 NTR via activation of NF-kappaB pathways, providing another mechanism for the involvement of NGF in the Th2 response 27493098_results suggest that perivascular nerves innervate neovessels as neovasculatures mature and that NGF accelerates the innervation of perivascular nerves in neovessels. 27568565_nerve growth factor (NGF) signaling through neurotrophic tyrosine kinase receptor type 1 (TrkA) directs innervation of the developing mouse femur to promote vascularization and osteoprogenitor lineage progression. 27635406_TNF-alpha upregulated Nerve Growth Factor [NGF] expression in synovial fibroblasts and macrophages and IL-1beta upregulated NGF expression in synovial fibroblasts. IL-1beta and TNF-alpha may regulate NGF signaling in Osteoarthritic joints and be suitable therapeutic targets for treating Osteoarthritis pain 27825441_These findings reveal a non-neuronal role for neurotrophins and identify a new regulatory pathway in insulin secretion that can be targeted to ameliorate beta cell dysfunction. 28079757_Mechanical stress-induced upregulation of NGF in colon SMC underlies the visceral hypersensitivity in bowel obstruction through increased gene expression and activity of tetrodotoxin-resistant Na channels in sensory neurons. 28416686_both in vivo mechanical loading and in vitro mechanical stretch were shown to induce the profound up-regulation of NGF in osteoblasts within 1 h of loading. 28453662_we revealed an unexpected contribution of NGF/TrkA signaling to the dendritogenesis 28542147_This study may represent a common mechanism for selective follicular activation induced by a localized increase in NGF in interstitial cells and mediated via the mTOR signaling pathway. 29166838_antibodies raised against NGF, TrkA, and p75 (also known as CD271) were used to explore the expression of these antigens in the non-decalcified young mouse femur. 29241539_Mechanoinsensitive ''silent'' nociceptors are characterized by the expression of the nicotinic acetylcholine receptor subunit alpha-3 (CHRNA3); the mechanically gated ion channel PIEZO2 mediates NGF-induced mechanosensitivity in these neurons. 29253516_An inducible mouse model that can dissect the contribution of autocrine direct action of cleavage-resistant proNGF on systemic microvascular abnormalities in both retina and kidney, major targets for microvascular complication. 29381137_Here, the authors report that the majority of retrograde NGF-TrkA signaling endosomes in mouse sympathetic neurons are ultrastructurally and molecularly defined multivesicular bodies (MVBs). 29523844_NGF-induced differentiation requires the activation of autophagy mediated by Atg9b and Ambra1, as it is disrupted by their genetic knockdown and by autophagy blockers. 29584618_These results unveil a dual role of proNGF/p75 signaling in bladder function under pathological conditions with a CNS effect overriding the peripheral one. 29589323_intra-adipose sympathetic plasticity regulated by NGF-TrkA signal in cold-induced beiging 29875237_The authors described a long-distance signaling mechanism of Porcine hemagglutinating encephalomyelitis virus-driven deficits in neurons and suggested that such Ulk1 repression may result in limited NGF/TrkA retrograde signaling within activated Rab5 endosomes, explaining the progressive failure of neurite outgrowth and survival. 29975347_Data indicate that nerve growth factor (NGF) impacts pancreatic ductal adenocarcinoma (PDAC) progression and metastasis in a temporally dependent manner. 30061605_structural analysis of binding of NGF, proNGF, and proBDNF to SorCS2 30430919_Treatment with NGF was found to upregulate both gene and protein expressions as well as total activity of DNA methyltransferases in differentiating HT22 cells. 30934765_NGFbeta directly interacts with adiponectin and SPARC, whereas these interactions oppositely regulate NGFbeta functions. 30977543_TNF-alpha and TGF-beta may stimulate NGF production under inflammatory and non-inflammatory conditions following intervertebral disc injury. 31578352_Multiple myeloma increases nerve growth factor and other pain-related markers through interactions with the bone microenvironment. 31996225_Spatio-temporal expression profile of NGF and the two-receptor system, TrkA and p75NTR, in experimental autoimmune encephalomyelitis. 32579702_Sensitization of glutamate receptor-mediated pain behaviour via nerve growth factor-dependent phosphorylation of transient receptor potential V1 under inflammatory conditions. 32669337_Herpes Simplex Virus 2 Counteracts Neurite Outgrowth Repulsion during Infection in a Nerve Growth Factor-Dependent Manner. 32892421_Excess of ovarian nerve growth factor impairs embryonic development and causes reproductive and metabolic dysfunction in adult female mice. 33307981_Differentiation of the 50B11 dorsal ganglion cells into NGF and GDNF responsive nociceptor subtypes. 34487017_Expression of nerve growth factor in the callus during fracture healing in a fracture model in aged mice. 34639085_proNGF Involvement in the Adult Neurogenesis Dysfunction in Alzheimer's Disease. 35302859_NGF-p75 signaling coordinates skeletal cell migration during bone repair. 35306300_Localization of NGF expression in mouse spleen and salivary gland: Relevance to pleotropic functions. 35579787_NGF/TrkA promotes the vitality, migration and adhesion of bone marrow stromal cells in hypoxia by regulating the Nrf2 pathway. |
ENSG00000134259 |
NGF |
58.238647 |
2.2162158372 |
1.148098 |
0.33276106 |
11.896411 |
0.00056242199117076917269653080211355700157582759857177734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0027751252897677859687863843873856239952147006988525390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
79.3076315996931 |
18.4551218530319 |
35.7851569635584 |
6.47184224475503 |
| ENSMUSG00000027875 |
15360 |
Hmgcs2 |
protein_coding |
P54869 |
FUNCTION: Catalyzes the first irreversible step in ketogenesis, condensing acetyl-CoA to acetoacetyl-CoA to form HMG-CoA, which is converted by HMG-CoA reductase (HMGCR) into mevalonate. {ECO:0000269|PubMed:24315375}. |
Acetylation;Cholesterol biosynthesis;Cholesterol metabolism;Lipid biosynthesis;Lipid metabolism;Mitochondrion;Phosphoprotein;Reference proteome;Steroid biosynthesis;Steroid metabolism;Sterol biosynthesis;Sterol metabolism;Transferase;Transit peptide |
PATHWAY: Metabolic intermediate biosynthesis; (R)-mevalonate biosynthesis; (R)-mevalonate from acetyl-CoA: step 2/3. |
|
mmu:15360; |
mitochondrial inner membrane [GO:0005743]; mitochondrial matrix [GO:0005759]; mitochondrion [GO:0005739]; hydroxymethylglutaryl-CoA synthase activity [GO:0004421]; identical protein binding [GO:0042802]; acetyl-CoA metabolic process [GO:0006084]; adipose tissue development [GO:0060612]; brain development [GO:0007420]; cellular response to amino acid stimulus [GO:0071230]; cellular response to fatty acid [GO:0071398]; cellular response to glucocorticoid stimulus [GO:0071385]; cellular response to insulin stimulus [GO:0032869]; cellular response to lipopolysaccharide [GO:0071222]; cholesterol biosynthetic process [GO:0006695]; farnesyl diphosphate biosynthetic process, mevalonate pathway [GO:0010142]; ketone body biosynthetic process [GO:0046951]; kidney development [GO:0001822]; liver development [GO:0001889]; lung development [GO:0030324]; midgut development [GO:0007494]; multicellular organismal response to stress [GO:0033555]; response to cAMP [GO:0051591]; response to ethanol [GO:0045471]; response to glucagon [GO:0033762]; response to growth hormone [GO:0060416]; response to linoleic acid [GO:0070543]; response to metal ion [GO:0010038]; response to monosaccharide [GO:0034284]; response to nutrient [GO:0007584]; response to prostaglandin F [GO:0034696]; response to starvation [GO:0042594]; response to temperature stimulus [GO:0009266]; response to testosterone [GO:0033574]; response to triglyceride [GO:0034014]; response to xenobiotic stimulus [GO:0009410] |
25904331_HMGCS2 was identified as a major down-regulated protein in colonic crypts isolated from Keratin 8-/- mice. 27874312_Using in vitro ketosis model by glucose starvation, studied inhibition of ketosis by momilactone B. Found momilactone B could regulate the angiopoietin-like-3 (ANGPTL3)-lipoprotein lipase (LPL)pathway, and suppressed the expression of HMGCS2 through the increased expression of STAT5b. 28528772_It is a key enzyme for catalyzing b-hydroxybutyric acid production, and observed in early Alzheimer's disease.And a hepatic inflammatory factor, IL-6 enhances ketogenesis through HMGCS2 signaling activation by p38/NF-kB p65. 31025252_To elucidate the relationship between HMGCS2 and NF-kB, we tested inhibited and over-expressed NF-kB. We found that NF-kB acted as a positive regulator for HMGCS2 under ketosis treatment conditions. 31910233_To test if Hmgcs2 is important for gonad development in mammals, we pursued two lines of investigations. Firstly, we generated Hmgcs2-null mice using CRISPR/Cas9 and found that these mice had gonads that developed normally even on a sensitized background 36629048_LONP1 targets HMGCS2 to protect mitochondrial function and attenuate chronic kidney disease. |
ENSG00000134240 |
HMGCS2 |
70.677588 |
0.0504172493 |
-4.309939 |
0.43201560 |
111.264448 |
0.00000000000000000000000005178151718080394493012923759861758028591226521733069501679550499770353713668846751261298777535557746887207031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000029876366276439608826237925124836732891851827158503904412468853276849123368208438478177413344383239746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.99488749153709 |
2.12606680535178 |
138.739966618734 |
22.9955522931217 |
| ENSMUSG00000027896 |
229699 |
Slc16a4 |
protein_coding |
Q8R0M8 |
FUNCTION: Proton-linked monocarboxylate transporter. Catalyzes the rapid transport across the plasma membrane of many monocarboxylates such as lactate, pyruvate, branched-chain oxo acids derived from leucine, valine and isoleucine, and the ketone bodies acetoacetate, beta-hydroxybutyrate and acetate (By similarity). {ECO:0000250}. |
Alternative splicing;Cell membrane;Membrane;Reference proteome;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:229699; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; monocarboxylic acid transmembrane transporter activity [GO:0008028]; symporter activity [GO:0015293]; monocarboxylic acid transport [GO:0015718] |
17599960_Expression of cerebral MCT isoforms can be modulated by alterations of peripheral metabolism, suggesting that the adult brain is sensitive and adapts to new metabolic states. 25762662_This study showed that mouse MCT1, MCT2, and MCT4 are expressed in the PNS. While DRG neurons express MCT1, myelinating Schwann cells. |
ENSG00000168679 |
SLC16A4 |
213.759444 |
7.1119766334 |
2.830251 |
0.21502761 |
171.488177 |
0.00000000000000000000000000000000000000350059544558007875576083992221253201329199807041668572681596489598802338285305752063692263016081679220803390961691548000089824199676513671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000003979184315453414758971893694534088127227230853876355619806934494288700919312369126422950733131152223354121133525040931999683380126953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
373.733822699223 |
55.9099188626997 |
52.5499227517825 |
6.50530699360093 |
| ENSMUSG00000027977 |
83398 |
Ndst3 |
protein_coding |
Q9EQH7 |
FUNCTION: Essential bifunctional enzyme that catalyzes both the N-deacetylation and the N-sulfation of glucosamine (GlcNAc) of the glycosaminoglycan in heparan sulfate. Modifies the GlcNAc-GlcA disaccharide repeating sugar backbone to make N-sulfated heparosan, a prerequisite substrate for later modifications in heparin biosynthesis. Has high deacetylase activity but low sulfotransferase activity. {ECO:0000269|PubMed:11087757}. |
Alternative splicing;Disulfide bond;Glycoprotein;Golgi apparatus;Hydrolase;Membrane;Multifunctional enzyme;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Glycan metabolism; heparan sulfate biosynthesis.; PATHWAY: Glycan metabolism; heparin biosynthesis. |
|
mmu:83398; |
Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; [heparan sulfate]-glucosamine N-sulfotransferase activity [GO:0015016]; deacetylase activity [GO:0019213]; heparan sulfate N-deacetylase activity [GO:0102140]; N-acetylglucosamine deacetylase activity [GO:0050119]; sulfotransferase activity [GO:0008146]; heparan sulfate proteoglycan biosynthetic process [GO:0015012]; heparan sulfate proteoglycan biosynthetic process, polysaccharide chain biosynthetic process [GO:0015014]; heparin biosynthetic process [GO:0030210] |
18385133_NDST3 function is not essential for development or adult homeostasis despite contributing to HS synthesis in a region-specific manner 29688404_An essential role for Heparin sulfate proteoglycan sulfation via NDST1,2,3 in modulating signal transduction during mouse implantation. |
ENSG00000164100 |
NDST3 |
8.171865 |
0.0372792628 |
-4.745483 |
1.44123045 |
11.528863 |
0.00068523921140379619176830061633154400624334812164306640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0032967667934626103051765078078005899442359805107116699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.69306189582384 |
0.81340190763943 |
18.5910837266933 |
10.4625337989162 |
| ENSMUSG00000028031 |
56811 |
Dkk2 |
protein_coding |
Q9QYZ8 |
FUNCTION: Antagonizes canonical Wnt signaling by inhibiting LRP5/6 interaction with Wnt and by forming a ternary complex with the transmembrane protein KREMEN that promotes internalization of LRP5/6. DKKs play an important role in vertebrate development, where they locally inhibit Wnt regulated processes such as antero-posterior axial patterning, limb development, somitogenesis and eye formation. In the adult, Dkks are implicated in bone formation and bone disease, cancer and Alzheimer disease. {ECO:0000269|PubMed:18524778}. |
3D-structure;Developmental protein;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:56811; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; co-receptor binding [GO:0039706]; receptor antagonist activity [GO:0048019]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of Wnt signaling pathway [GO:0030178]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; Wnt signaling pathway [GO:0016055] |
16195809_Dkks were involved in craniofacial developmental processes, including facial outgrowth, myogenesis, osteogenesis, palatogenesis, olfactory epithelium and tooth development; and the expression persisted to postnatal stage in the muscles and bones. 16521043_Dkk2 was localized in the perichondral mesenchyme outlining the anterior cranial base in embryogenesis. 16672341_Dkk2 is a key regulator of the corneal versus epidermal fate of the ocular surface epithelium 17971207_WNT signaling attenuation by DKK-1 and -2 regulates BMP2-induced osteoblast differentiation and expression of OPG, RANKL and M-CSF 18367164_These data suggest that activation of Dkk2 by PITX2 provides a mechanism to locally suppress canonical Wnt signaling activity during eye development 18524778_analysis of the structural basis of the interaction between Dkk and low density lipoprotein receptor-related protein (LRP) 5/6 18689976_Dkk2 expression is associated with uterine receptivity changes as well as the process of decidualization and that it might play important roles through inhibition of canonical Wnt signaling in the periimplantation uterus. 19185060_Study identified novel expression domains for the Wnt antagonists Sfrp1, Sfrp3, Sfrp5 as well as Dkk2 and Dkk3. 20439124_Both Dkk1 and Dkk2 inhibit Wnt signaling that regulates early myocardial proliferation; each compensates for loss of the other in that role. 21540552_there are distinct functions of DKK1 and DKK2 in controlling angiogenesis 22733757_Dickkopf2 is a Wnt antagonist involved in regulation of glucose metabolism 23250216_Bmp4 signaling suppresses tooth developmental inhibitors in the tooth mesenchyme, including Dkk2 and Osr2, and synergizes with Msx1 to activate mesenchymal odontogenic potential for tooth morphogenesis and sequential tooth formation 24091497_These findings demonstrate that DKK1 and DKK2 have differential roles in normalization and functionality of tumor blood vessels, in addition to angiogenesis. 25826080_Dkk2 deletion results in alterations of liver morphology leading to an increased frequency of liver cancer 27053129_Spermatogenesis-associated protein 3 (Spata3) and dickkopf-related protein 2 (Dkk2) were confirmed to interact with MIC3. The tandem repeat EGF domains of MIC3 were critical in mediating the interactions with the identified host proteins. The results show that MIC3 interacts with host proteins that are involved in reproduction, growth, and development. 27713059_these data reveal a novel mechanism that the Bmp4-Msx1 pathway and Osr2 control tooth organogenesis through antagonistic regulation of expression of secreted Wnt antagonists. 29259207_Reprogramming of damaged erectile tissue toward neurovascular repair by use of a DKK2 therapeutic protein may represent viable treatment option for this condition. 29559443_Data (including data from studies in transgenic mice) suggest that Dkk2 secreted from pericytes promotes angiogenesis and neurogenesis/neural regeneration via Angpt1/Tie2/Wnt signaling; here, overexpression of Dkk2 in diabetic mice enhances penile angiogenesis/neural regeneration and restores erectile function. (Dkk2 = dickkopf-2; Angpt1= angiopoietin-1; Tie2 = endothelial-specific receptor tyrosine kinase Tie2) 30509557_In the absence of Dkk2, Wnt/beta-catenin signaling activity is initially broadly elevated in embryonic plantar skin and gradually becomes patterned, mimicking follicular development in normally haired areas. 33647798_Dickkopf (Dkk)-2 is a beige fat-enriched adipokine to regulate adipogenesis. 34030936_Dickkopf-2 knockdown protects against classic macrophage polarization and lipid loading by activation of Wnt/beta-catenin signaling. 34676493_Mesenchymal Stem Cell-Derived Extracellular Vesicles Inhibit Osteoporosis via MicroRNA-27a-Induced Inhibition of DKK2-Mediated Wnt/beta-Catenin Pathway. |
ENSG00000155011 |
DKK2 |
28.742046 |
0.0358655940 |
-4.801256 |
0.80550547 |
40.404175 |
0.00000000020649933183671634350777502813709942883213521724883321439847350120544433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000031612038417455913328209877506290090276586113304801983758807182312011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.05754394228963 |
1.20942068568001 |
57.3681824766193 |
17.3178250249346 |
| ENSMUSG00000028037 |
99899 |
Ifi44 |
protein_coding |
Q8BV66 |
FUNCTION: This protein aggregates to form microtubular structures. {ECO:0000250}. |
Cytoplasm;Reference proteome |
|
|
mmu:99899; |
cytoplasm [GO:0005737]; immune response [GO:0006955]; response to bacterium [GO:0009617] |
32611756_Interferon-Induced Protein 44 and Interferon-Induced Protein 44-Like Restrict Replication of Respiratory Syncytial Virus. |
ENSG00000137965 |
IFI44 |
169.331091 |
0.4601788595 |
-1.119733 |
0.36436143 |
9.238828 |
0.00236936544174082145200466165135821938747540116310119628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0098338349887183082453923077537183417007327079772949218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
105.927981067362 |
27.0380680255012 |
230.188716595651 |
42.817161289451 |
| ENSMUSG00000028039 |
13638 |
Efna3 |
protein_coding |
O08545 |
FUNCTION: Cell surface GPI-bound ligand for Eph receptors, a family of receptor tyrosine kinases which are crucial for migration, repulsion and adhesion during neuronal, vascular and epithelial development. Binds promiscuously Eph receptors residing on adjacent cells, leading to contact-dependent bidirectional signaling into neighboring cells. The signaling pathway downstream of the receptor is referred to as forward signaling while the signaling pathway downstream of the ephrin ligand is referred to as reverse signaling. {ECO:0000269|PubMed:9053851}. |
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:13638; |
plasma membrane [GO:0005886]; ephrin receptor binding [GO:0046875]; axon guidance [GO:0007411]; ephrin receptor signaling pathway [GO:0048013]; negative regulation of angiogenesis [GO:0016525]; positive regulation of aspartic-type endopeptidase activity involved in amyloid precursor protein catabolic process [GO:1902961]; regulation of neuron differentiation [GO:0045664] |
12496762_Ephrin-A3 is localized on astrocytic processes that envelop spines. Activation of EphA4 by ephrin-A3 caused spine retraction; inhibiting ephrin/EphA4 interactions distorted spine shape and organization in hippocampal slices. 12684176_Organ of Corti and spiral ganglion showed strong expression of ephrin-A3, ephrin-B2 and ephrin-B3. In lateral wall, ephrin-A3 and ephrin-B2 were strongly expressed. Ephrin-A3 was strongly expressed in utricular and saccular sensory epithelia. 12914696_Results demonstrate that neurons expressing different odorant receptors express different levels of ephrin-A3 and -A5 protein on their axons. 16025107_In mice deficient for ephrin-A2, A3 and A5, eye-specific inputs segregated but shape and location of eye-specific layers were profoundly disrupted. Ephrin-As and neural activity act together to control patterning of eye-specific retinogeniculate layers. 16466970_Whole-mount in situ hybridization revealed overlapping expression of the Epha1 receptor and its high-affinity ligands ephrin A1 (Efna1) and ephrin A3 (Efna3) in the primitive streak and the posterior paraxial mesoderm during early mouse development. 18640011_ephrin-A3 not only accelerates anagen development but also increases the density of hair follicles. 18715996_heparan sulfate modulates ephrin-A3/Eph signaling, suggesting a physiological role for heparan sulfate proteoglycans in the regulation of ephrin-A3-dependent biological processes 19592509_Results suggest that the interaction between neuronal EphA4 and glial ephrin-A3 bidirectionally controls synapse morphology and glial glutamate transport, ultimately regulating hippocampal function. 19734893_These results suggest that EphA4/ephrin-A3 signaling is a critical mechanism for astrocytes to regulate synaptic function and plasticity. 20473036_Downregulation of the EphA4 receptor via siRNA transfection reduced the repulsive effect of ephrin-A3, indicating that EphA4 mediates at least in part the repulsive effect of ephrin-A3. 20655977_Data show that a number of Eph receptors and ephrins were expressed in hematopoietic stem cells. 23165658_Ephrin-A3 suppresses Wnt signaling to control retinal stem cell potency. 25269910_Ephrin-A2 and -A3 are negative regulators of the proliferative and neurogenic potentials of Muller cells. 25281279_The ephrin-A2/-A3 DKO mice have utility as a novel ASD model with an emphasis on sensory abnormalities and restricted, repetitive behavioral symptoms. 26785076_Findings suggest a putative novel mechanism for desipramine to modulate long-term potentiation through the regulation of the ephrinA3/EphA4 signaling pathway 34235739_Ephrin-A3 is required for tonotopic map precision and auditory functions in the mouse auditory brainstem. |
ENSG00000143590 |
EFNA3 |
66.733109 |
0.1281380586 |
-2.964229 |
0.40418546 |
52.152441 |
0.00000000000051354966449011797054692171540859086395838795091606243659043684601783752441406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000106427054279094932798379157211094824669883607271003711502999067306518554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.2267523553809 |
3.7542730146471 |
103.222668618597 |
19.2326248485195 |
| ENSMUSG00000028068 |
404710 |
Iqgap3 |
protein_coding |
A0A0A6YXJ2 |
|
Proteomics identification;Reference proteome |
|
|
|
cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; lateral plasma membrane [GO:0016328]; actin filament binding [GO:0051015]; calmodulin binding [GO:0005516]; GTPase activator activity [GO:0005096]; myosin VI light chain binding [GO:0070856]; small GTPase binding [GO:0031267]; cellular response to organic substance [GO:0071310]; ERK1 and ERK2 cascade [GO:0070371]; G1/S transition of mitotic cell cycle [GO:0000082]; gene expression [GO:0010467]; mammary gland epithelial cell proliferation [GO:0033598]; negative regulation of gene expression [GO:0010629]; positive regulation of gene expression [GO:0010628]; positive regulation of mammary gland epithelial cell proliferation [GO:0033601]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of protein phosphorylation [GO:0001934]; protein phosphorylation [GO:0006468]; Ras protein signal transduction [GO:0007265]; regulation of actin cytoskeleton organization [GO:0032956]; regulation of cell size [GO:0008361] |
18604197_IQGAP3 is necessary and sufficient for driving cell proliferation and ERK acts downstream of IQGAP3 19452445_Report involvement of IQGAP3, a regulator of Ras/ERK-related cascade, in hepatocyte proliferation in mouse liver regeneration and development. 22493426_Phosphoinositide module is defined by a C-terminal 105-107 amino acid region of which IQGAP1 and -2, but not IQGAP3, binds preferentially to phosphatidylinositol 3,4,5-trisphosphate (PtdInsP(3)). |
ENSG00000183856 |
IQGAP3 |
967.598815 |
2.0361760989 |
1.025862 |
0.09302073 |
120.872143 |
0.00000000000000000000000000040757070848584579887657716792259948701145723285414487504659163053723444657995556017482385868788696825504302978515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000277148081770375153998044680201962755862034118007374403264094022376960550768654201192475738935172557830810546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1289.4072709525 |
83.1947989376692 |
633.249389201976 |
31.473873518426 |
| ENSMUSG00000028073 |
73182 |
Pear1 |
protein_coding |
Q8VIK5 |
FUNCTION: When overexpressed, reduces the number of both early and late non-adherent myeloid progenitor cells. {ECO:0000269|PubMed:17226770}. |
Alternative splicing;Cell membrane;Disulfide bond;EGF-like domain;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:73182; |
membrane [GO:0016020]; phagocytic cup [GO:0001891]; plasma membrane [GO:0005886]; signaling receptor activity [GO:0038023]; phosphatidylinositol 3-kinase signaling [GO:0014065]; platelet aggregation [GO:0070527]; protein kinase B signaling [GO:0043491]; recognition of apoptotic cell [GO:0043654] |
17226770_Jedi is involved in the fine regulation of the early stages of hematopoietic differentiation, presumably through the Notch signaling pathway. 19915564_a cellular and molecular mechanism by which neuronal corpses are culled during dorsal root ganglia development involves Jedi protein 22993420_These results indicated that both Jedi-1 and MEGF10 can mediate phagocytosis independently through the recruitment of Syk. 24743597_Jedi-1 signals through recruitment of GULP, which promotes phagocytosis through a noncanonical phosphoclathrin-dependent mechanism. 26156496_Platelet endothelial aggregation receptor-1: a novel modifier of neoangiogenesis. 29577896_Expression of the myogenic markers Desmin, MYOG, and MYH2 revealed that PEAR1 positively regulated C2C12 cell differentiation 30700416_Results establish PEAR1 as the major signaling receptor for natural fucose-based polysaccharides and synthetic glycopolymers in human, but not in mouse, platelets. 31678362_PEAR1 acts as a negative regulator in the proliferation of pulmonary microvascular endothelial cells in acute lung injury model via the PI3K/AKT pathway. 31992767_These results provide new insight into the expression pattern of Jedi-1 in the peripheral nervous system and indicate that loss of Jedi-1 alters DRG neuron activity indirectly through an intercellular interaction between non-neuronal cells and sensory neurons. 36792666_SVEP1 is an endogenous ligand for the orphan receptor PEAR1. |
ENSG00000187800 |
PEAR1 |
90.993324 |
4.7103753490 |
2.235842 |
0.42234384 |
26.323537 |
0.00000028874820574478356525628523245041723299664226942695677280426025390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000027960665415794936423021701393798466028783877845853567123413085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
153.497494371207 |
45.5804384525531 |
32.5871046357289 |
7.48179597897836 |
| ENSMUSG00000028076 |
12479 |
Cd1d1 |
protein_coding |
E9Q395 |
Human_homologues FUNCTION: Antigen-presenting protein that binds self and non-self glycolipids and presents them to T-cell receptors on natural killer T-cells. {ECO:0000269|PubMed:17475845}. |
Glycoprotein;Membrane;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
|
membrane [GO:0016020] |
11751982_Natural killer T cells restricted by CD1d1 molecules behave like inflammatory cells 11777966_Natural CD1d1-associated ligands play a chaperone-like role in the assembly of CD1d1 in vivo akin to the function of invariant chain in MHC class II assembly. 11870629_CD1d on antigen-transporting APC and splenic marginal zone B cells promotes NKT cell-dependent tolerance. 11920590_Modulation of CD1 expression on tumor cells by cytokines may be advantageous to drive cellular anti-tumor antigen directed immune responses against TAP-independent, non classical MHC restricting molecules 12042809_involved in macrophage-mediated clearance of Pseudomonas aeruginosa from lung 12097395_Antigen presentation by CD1d plays a key role in the generation of an optimal Th2 cytokine-polarized response and the development of subsequent liver pathology during murine schistosomiasis. 12538704_On infection with herpes simplex virus type 1, CD1d gene knockout mice show increased morbidity, enhanced spread of virus in the nervous system, and greatly diminished clearance of virus from the skin and nervous system. 12594829_CD1a and CD1b surface expression is independent from de novo synthesized glycosphingolipids. 12626572_CD1d expression is essential for pathogenicity of coxsackievirus B3 (CVB3)-induced myocarditis and is increased early after infection in vivo in CD1d+ mice; V gamma 4+ T cells appear to recognize CD1d expressed by CVB3-infected myocytes. 12648456_CD1d-restricted NKT cells from distinct genetic backgrounds differentially influence T(H)1/T(H)2 polarization, proinflammatory cytokine levels, pathogenesis, and fatality in the P. berghei ANKA/rodent model of cerebral malaria. 12695491_Data show that interleukin-18 fails to induce immunoglobulin E both in CD1d-/- mice that lack natural killer T (NKT) cells and in class II-/- mice that lack conventional CD4+ T cells. 12730881_CD1d is expressed functionally on IECs with a polarity of presentation (basal > apical) predicting a role in presentation of mucosal glycolipid antigens to local CD1d-restricted T cells. 12773507_A role for CD1d-restricted NKT cells in burn injury-associated T cell suppression. 12794761_CD1d-mediated stimulation of natural killer T cells selectively activates hepatic natural killer cells 14530332_The distinct pattern and levels of CD1d expression are critical for selection of NKT cells; however, unique structural features of CD1d preclude its active participation in selection of CD8+ T cells. 14530337_CD1d interacts with the adaptor protein AP-3 via its cytoplasmic tail tyrosine-based sorting motif that controls the localization of CD1d to lysosomes and the development of NK T cells. 14744994_Activation of CD1d-restricted natural killer T cells exacerbates atherosclerosis. 14768037_CD1d-dependent NKT cells exacerbate Th2-mediated airway inflammation and hyperresponsiveness in NOD mice. 15004145_The expression of CD1d1 molecules controls the magnitude of the cell-mediated immune response to an acute viral infection. 15100269_Direct evidence is provided for a physical interaction of natural killer (NK) cells with CD1d1, mediated by a functional, CD1d1-specific low-affinity inhibitory NK receptor. 15107843_Conditional deletion of the Mttp gene in hepatocytes is associated with a redistribution of CD1d expression. 15162443_CD1d antigen deficiency exacerbates inflammatory dermatitis in MRL-lpr/lpr mice. 15212710_Results provide evidence for a role for CD1d in mediation of pathology during hypersensitive responses to the mycobacterial glycolipid TDM. 15243159_Data show that phosphatidylinositol mannoside represents a mycobacterial antigen recognized by T cells in the context of CD1d. 15548574_The CD1d expression occurred during an acute virus infection is decreased on splenic dendritic cells (DC) and macrophages (MPhi)for at least 3 months post-LCMV infection. 15591216_CD1d-restricted NKT cells contribute to the formation of fatty streaks; however, their influence on lesion progression is transient, and they do not significantly affect the inflammatory cytokine milieu of mature lesions. 15652515_murine CD1d (mCD1d) was constitutively present in the plasma membrane lipid rafts on antigen presenting cells 15731095_CD1d antigen presentation required to activate natural killer T cells during Trypanosoma cruzi infection. 15749847_multiple steps, including interactions with the adaptors AP-2 and AP-3, are required for normal trafficking of CD1d, and these different steps are mediated by only a few cytoplasmic amino acids 15810882_CD1d and CD1d-restricted iNKT-cells play a pivotal role in contact hypersensitivity 15843569_CD1d expression is required for the production of Borrelia hermsii-specific IgM antibodies in marginal zone B cells at the earliest stages of infection with blood-borne spirochetes. 15944296_Expression of CD1d can be crucial for survival of mice infected with Toxoplasma gondii; there is a link between a CD1d-dependent mechanism of resistance to T. gondii and pathological effects of CD4+ T cells. 16002697_The crystal structure of mouse CD1d bound to phosphatidylcholine suggests a likely mode of recognition of lipid antigens by the natural killer T (NKT) cell receptor. 16007091_analysis of the rystal structure of CD1d in complex with a short-chain synthetic variant of alpha-galactosylceramide at a resolution of 2.2 A 16148102_APC expressing higher CD1d levels more efficiently activate NKT cell hybridomas and primary NKT cells independently of whether the CD1d-restricted TCR recognizes foreign or self-lipid Ags 16424169_Changes to the CD1d expression program modulate iNKT cell development and function. 16517731_These results highlight the variation in Ag recognition among CD1d-restricted Receptors, Antigen, T-Cell (TCRs) and suggest that TCR alpha-chain elements contribute to alpha-linked glycosphingolipid specificity 16730774_findings showed that CD1d expression in hemopoietic & non-hemopoietic cells induces Coxsackievirus B3 myocarditis; CD1d expression on hemopoietic cells was more effective in increasing Vgamma4+ cell numbers & activation & CD4+ IFNgamma+ cell response 16785503_Both CD1d expression by antigen-presenting cells and CD1d-restricted natural killer (NK)T cells are required for immune suppression after injury. 16817758_expression upregulated by decay accelerating factor in Coxsackievirsu B3 myocarditis 17088433_Hepatic reperfusion injury is initiated by CD1d-dependent activation of natural killer T (NKT) cells. 17237379_both in vitro inhibition of T cell differentiation by iNKT cells and mouse protection against diabetes by iNKT cells were CD1d-independent. 17403933_suggest a novel, unexpected role of microsomal triglyceride transfer protein at a late stage of CD1d trafficking in the lysosomal compartment 17476670_CD1d-restricted NKT cells have roles in the intestine and in inflammatory bowel diseases [review] 17607300_In a mouse model of fatty liver, CD1d expression is reduced in fatty hepatocytes with tunamycin-induced endoplasmic reticulum (ER) stress. 17705133_Protein kinase C delta is a critical regulator of CD1d-mediated antigen presentation and is involved in multiple steps of the process 17885216_Presentation of lipid antigens through the CD1d-natural killer (NK)T cell pathway modulates vascular repair responses. 18077787_Mechanism by which naatural killer cells enhance humoral immune responses involves interaction with CD1d-expressing B cell. 18212072_The role of CD1d-restricted natural killer cells in induction of toxic shock-like syndrome caused by gram-negative, lipopolysaccharide-lacking, monocytotropic Ehrlichia is reported. 18549881_Suggest a critical role for CD1d-restricted invariant NKT cells in stimulating intrahepatic CD8 T-cell responses to liver antigen. 18566372_identify critical roles for CXCR6/CXCL16 in NKT cell activation and the regulation of NKT cell homeostasis 18606153_The role of CD1d-restricted invariant natural killer T cells in laser-induced experimental choroidal neovascularization was investigated. 18624295_Co-inhibitory roles for glucocorticoid-induced TNF receptor in CD1d-dependent natural killer T cells. 18684818_Data suggest that CD1d-restricted natural killer cells may play a protective role in Theiler's murine encephalomyelitis virus-induced neurological disease by alteration of the cytokine profile and virus-specific immune responses. 18760855_tumor burden reduces CD1d expressions on liver dendritic cells, thus impeding alpha-GalCer-mediated NK cell activation and antitumor activity in the liver 18802058_A CD1d-restricted noninvariant T cell receptor natural killer (NK) T cell population is present in beta-2 microglobulin-deficient mice that is responsible for development of airway hyperreactivity but not for T helper type 2 (Th2) cell responses. 18815300_vesicular stomatitis virus -M protein plays an important role in permitting the virus to evade important components of the innate immune response by regulating specific MAPK pathways 19179286_CD1d polymorphisms affect the thymic selection of type I NKT cells and induce allogeneic T cell responses 19234223_Blocking the interaction between invariant natural killer T (iNKT) cells and the CD1d molecule reduces development of airway hyperreactivity in wild type BALB/c mice as well as in transcription factor T-bet-deficient mice. 19246642_The effects of beta-glycosphingolipids on the STAT signaling pathways were found to be dependent on CD1d 19265113_Anti-CD1d monoclonal antibody induces optimal tumor suppression when CD1d-restricted type II natural killer (NK)T cells regulate growth. 19342651_statins impair a critical component of the innate immune response, CD1d-mediated Ag presentation 19349688_Data support a role for Cd1d in regulating intestinal colonization through mechanisms that include the control of Paneth cell function. 19454494_infiltrating NKT cells are mainly observed in CD1d nodules 19587020_the intrinsic CD1d-encoded sorting motif mediates thymic selection and activation of V(alpha)14 iNKT cells by professional APCs, while for peripheral terminal differentiation the intrinsic CD1d sorting motif is dispensable. 19732779_Modifications of the sphingosine moiety or other substitutions that decrease alpha-GalCer flexibility would stabilize the F'-pocket of CD1d. 19734232_The preferential stimulatory activities of two alpha-galactosylceramide C-glycoside analogues against human vs mouse invariant natural killer (NK)T cells are due to structural differences between human CD1d molecule compared to murine. 19783672_These latter findings indicate the involvement of CD1d-restricted NKT cells and IL-9 in the same pathway that leads to the aerosolized Ag-induced recruitment of mast cells and suggest that CD1d-restricted NKT cells directly or indirectly provide the IL-9. 19830742_Studies indicate that during bacterial infections, NKT cells can be activated either indirectly by DC or directly by bacterial lipid antigens presented by CD1d. 19833737_NKT cells modulate plasmid DNA vaccine antigen expression and vaccine-elicited immune responses by MCP-1 secretion after interaction with a beta2-microglobulin-independent CD1d 19949077_NKT cells can be bypassed with CD1d-mediated induction of robust Th1 immunity 20080535_show that the mouse and human CD1d present glycolipids having different fatty acids, based in part upon a difference at a single amino acid position that is involved in positioning the sugar epitope. 20121405_TNF-alpha treatment of chimeric mice having wild-type (CD1d+) hematopoietic cells restored Coxsackievirus B3-elicited myocarditis susceptibility. 20194602_These results suggest that Bacillus anthracis has the ability to evade the host's innate immune system by reducing CD1d-mediated antigen presentation through targeting the ERK1/2 pathway. 20439539_Absence of CD1d-expression on B cells leads to increased autoreactive B cell activation and symptoms of autoimmune diseases such as systemic lupus erythematosus. 20525883_CD1d-dependent natural killer (NK)T cells might be suppressor cells in acute and chronic experimental arthritis, likely via inhibition of arthritogenic T helper (Th)1 type cells. 20861015_Calreticulin controls the rate of assembly of CD1d molecules in the endoplasmic reticulum. 21050191_Vgamma4+ T-cells selectively abrogate T regulatory cells through recognition of CD1d expressed on the regulatory cells and caspase-dependent apoptosis. 21089371_NKT cell recognition of CD1d/glycolipid antigen complex regulates immune mechanism.(review) 21145835_CD1d-KO mice developed minimal hepatic fibrosis after 9-weeks of administration of thioacetamide, which caused overt bridging fibrosis in WT mice. 21209282_CD1d may play a role in the differential regulation of autoreactive versus non-autoreactive B cells by invariant natural killer (NK)T cells. 21389252_Findings demonstrate that CD1d is able to bind and present CL to a subset of CL-responsive gammadelta T cells that exist in the spleen and liver. 21552205_These findings highlight the previously unexploited flexibility of CD1d in accommodating galactose-modified glycolipids and broaden the range of glycolipids that can stimulate iNKT cells. 21695190_Breast cancer cells, through downregulation of CD1d and subsequent evasion of natural killer T-mediated antitumor immunity, gain increased potential for metastatic tumor progression. 21987790_phenyl glycolipids showed greater binding avidity and stability for iNKT T-cell receptor when complexed with CD1d 22041925_Data indicate that invariant NKT (iNKT) cell-mediated cytokine secretion in staphylococcal enterotoxin B (SEB)-challenged HLA-DR4-transgenic mice was CD1d-independent. 22057113_Aberrant type II natural killer (NK) T cell responses directly contribute to intestinal inflammation in mice, indicating the importance of CD1d expression levels in the development and regulation of type II NKT cells. 22065098_CD1d-reactive T cells protect against the development of lupus in animal models;depletion of CD1d-reactive T cells in preclinical BWF1 mice resulted in disease acceleration with a significant increase in proteinuria and mortality 22291186_The (327-329)RRR arginine motif of CD1d is required for proper cellular distribution of CD1d and optimal antigen presentation to both type I and type II natural killer (NK)T cells. 22504642_CD1d-independent Ly49+ T cells are reduced severely in the spleen, bone marrow, and liver, but not thymus, in mice doubly deficient for IFN regulatory factor-2 (IRF-2) and CD1d. 22535665_The results of the present study implicate CD1d in a previously unrecognized regulatory role of normal and stressed hematopoiesis. 22854104_Intrinsic unresponsiveness of Mertk-/- B cells to chronic graft-versus-host disease is associated with unmodulated CD1d expression. 22995911_Human and mouse type I natural killer T cell antigen receptors exhibit different fine specificities for CD1d-antigen complex 23280365_CD1d protein structure determines species-selective antigenicity of isoglobotrihexosylceramide (iGb3) to invariant NKT cells. 23307809_Data indicate that microbial phosphatidylglycerol (PG) bound to CD1d molecules and plate-bound PG/CD1d complexes stimulated dNKT hybridomas, indicating direct recognition by the dNKT cell T-cell receptor (TCR) alpha-and beta-chains. 23523122_Although percentage of CD1d(hi)CD5+ subset in splenic B cells does not change significantly, percent IL-10-expressing cells among total splenic B cells or CD1d(hi)CD5+ population significantly increases 3-7 days after calreticulin is administered 23527709_CD1d-associated abnormalities of innate immune responses and TNF-alpha production in splenic tissue may contribute to NFkB activation and cardiac dysfunction in type 2 diabetes 23531237_We report a novel dichotomous role of beta2m and CD1d, whereby these molecules differently regulate autoimmunity against phospholipid versus non-phospholipid autoantigens. 23615906_The quality control process may play a role in ensuring stability of exported CD1d-beta2m complexes, in facilitating presentation of low abundance high affinity antigens, or in preventing deleterious responses to self lipids. 23867896_Our findings identify an essential role for CD1d in promoting UVB-induced cutaneous tissue injury and inflammation. 23943651_We demonstrate that CD1d-restricted natural killer T (NKT) cells are required to produce anti-A antibodies (Abs), probably through collaboration with B-1a cells. 24717441_Protective mucosal immunity mediated by epithelial CD1d and IL-10 24966328_CD1d is up-regulated in early adipogenesis, and is transcriptionally controlled by CCAAT/enhancer-binding protein (C/EBP)-beta and -delta. 24990409_these results suggest that the canonical TGF-beta/Smad pathway negatively regulates an important arm of the host's innate immune responses - CD1d-mediated lipid antigen presentation to NKT cells. 25070854_These results demonstrate that Bcl-xL regulates CD1d-mediated Ag processing and presentation to NKT cells by altering the late endosomal compartment and changing the intracellular localization of CD1d. 25618030_CD1d acts as a cell surface receptor that recognizes and binds oxysterols and initializes a pathway connecting oxysterol binding to PPARgamma activation 25654212_Our results indicate that genetically controlled physiological variation of CD1d expression levels modulates iNKT-cell development. 25929465_our results demonstrate that both Ets1 and miR-155 can directly regulate the expression of CD1d on B-cells 26121177_regulatory and protective role of CD1d-dependent natural killer T cells in contact hypersensitivity, at least in part via regulation of IL-10 producing B(regs) 26496152_The findings support a model in which low endosomal pH controls stability and lipid raft localization of CD1d-glycolipid complexes to regulate the outcome of iNKT cell mediated responses. 26564814_affects iNKT cell activation by mast cells and release of both IFN-gamma and IL-13 26604037_reduced development of atherosclerosis in hyperlipidaemic mice by the NKT antagonist, without affecting NKT cell or other lymphocyte numbers, suggests that targeting lesion inflammation via CD1d-dependent activation of NKT cells 27006394_the crystal structure of the first peptide identified to bind CD1d, p99, and show that it binds in the antigen-binding groove of CD1d in a manner compatible with its presentation to T cell receptors. 27069116_CD1d-restricted peripheral T cell lymphoma in mice and humans 27329323_interactions between NKT cells and CD1d-expressing adipocytes producing endogenous NKT cell ligands play a critical role in the induction of inflammation and functional modulation of adipose tissue that leads to obesity 27385215_CD1D has a role in disease progression and survival of chronic lymphocytic leukemia and may interact with CD161 27799287_our data expose a previously unappreciated role for ILCs in CD1d-mediated immunity, which can modulate tissue homeostasis and inflammatory responses. 28082459_Adipocyte CD1d plays a key role in the stimulation of adipose invariant natural killer T cells, leading to anti-inflammatory responses in high-fat diet (HFD)-fed mice. 28296542_These findings indicate that the autophagic machinery assists in the recruitment of AP2 to CD1D1 molecules resulting in attenuated Invariant natural killer T cell activation. 28338767_The expression of CD1d showed a significantly negative correlation with CD86 level in B cells from imiquimod (IMQ)-treated mice, B6.MRLlpr mice, and lupus erythematosus (SLE) patients. 28392574_M2 macrophage-specific reduction of CD1d is an initiating event that switches Natural killer T cell-mediated immune responses and disrupts the immune balance in visceral adipose tissues in obese mice. 28893990_microsomal triglyceride transfer protein, a protein involved in the transfer of lipids onto CD1d, regulates liver natural killer T cell homeostasis in a manner dependent on hepatocyte CD1d. 30467836_work strongly suggests JNK2 is a negative regulator of CD1d-mediated Ag presentation and contributes to IL-12-induced iNKT cell activation and loss during viral infections. 30508304_glycerophospholipids reduced iNKT-cell proliferation, expansion, and cytokine production in vitro and in vivo. Such superior ability of self-glycerophospholipids to compete with iNKT-cell ligands to occupy CD1d may help maintain homeostasis between the diverse subsets of lipid-reactive T cells. 30513408_disruption of the CD1d communication pathway between mast cells and NKT cells aggravates atherosclerosis, through a shift towards pro-inflammatory T cell responses 30514903_The authors identify a non-canonical function of CD1d reverse signaling activated by endogenous glycolipid isoglobotrihexosylceramide (iGb3) in inflammatory innate responses of antigen-presenting cells. 30864713_Study observed that Cd1d was predominantly expressed on dendritic cells (DCs) in 3LL tumorbearing mice, whilst a deficiency of Cd1d promoted tumor growth. Notably, Cd1d expression on DCs was not only required for presenting antigen to NKT cells, but also markedly promoted CD4+T and CD8+T cell activation, particularly cytotoxic T cells. 31653827_Our findings suggest that in the absence of exogenous iNKT cell ligands, iNKT cells enhanced the CTL production of IFN-g and CTL proliferation and cytotoxicity via direct interaction with CD1d expressed on T cells without interacting with APCs. 33283278_Hepatocyte CD1d protects against liver immunopathology in mice with schistosomiasis japonica. 33407995_CD1d deficiency limits tolerogenic properties of peritoneal macrophages. 34181769_The lysophospholipid-binding molecule CD1D is not required for the alloimmunization response to fresh or stored RBCs in mice despite RBC storage driving alterations in lysophospholipids. 35392825_Steroid-mediated liver steatosis is CD1d-dependent, while steroid-induced liver necrosis, inflammation, and metabolic changes are CD1d-independent. |
ENSG00000158473 |
CD1D |
54.912949 |
0.1478501653 |
-2.757792 |
0.48538961 |
28.961784 |
0.00000007382038484056191853815732115476344610272008139872923493385314941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000007901841896636958430624221583205013530459837056696414947509765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.4863948830884 |
4.96636888802427 |
97.9802413965924 |
23.0466953753432 |
| ENSMUSG00000028086 |
50754 |
Fbxw7 |
protein_coding |
Q8VBV4 |
FUNCTION: Substrate recognition component of a SCF (SKP1-CUL1-F-box protein) E3 ubiquitin-protein ligase complex which mediates the ubiquitination and subsequent proteasomal degradation of target proteins (PubMed:21953459, PubMed:22748924). Recognizes and binds phosphorylated sites/phosphodegrons within target proteins and thereafter brings them to the SCF complex for ubiquitination (PubMed:22748924). Mediates ubiquitination and subsequent degradation of CCNE1 and MYC (PubMed:22748924). Identified substrates include cyclin-E (CCNE1 or CCNE2), DISC1, JUN, MYC, NOTCH1 released notch intracellular domain (NICD), NOTCH2, MCL1, RICTOR and probably PSEN1 (By similarity). Acts as a negative regulator of JNK signaling by binding to phosphorylated JUN and promoting its ubiquitination and subsequent degradation (By similarity). SCF(FBXW7) complex mediates the ubiquitination and subsequent degradation of NFE2L1 (PubMed:21953459). Involved in bone homeostasis and negative regulation of osteoclast differentiation (PubMed:29149593). Regulates the amplitude of the cyclic expression of hepatic core clock genes and genes involved in lipid and glucose metabolism via ubiquitination and proteasomal degradation of their transcriptional repressor NR1D1; CDK1-dependent phosphorylation of NR1D1 is necessary for SCF(FBXW7)-mediated ubiquitination (PubMed:27238018). Also able to promote 'Lys-63'-linked ubiquitination in response to DNA damage (By similarity). The SCF(FBXW7) complex facilitates double-strand break repair following phosphorylation by ATM: phosphorylation promotes localization to sites of double-strand breaks and 'Lys-63'-linked ubiquitination of phosphorylated XRCC4, enhancing DNA non-homologous end joining (By similarity). {ECO:0000250|UniProtKB:Q969H0, ECO:0000269|PubMed:16141072, ECO:0000269|PubMed:21953459, ECO:0000269|PubMed:22748924, ECO:0000269|PubMed:27238018, ECO:0000269|PubMed:29149593}. |
Alternative splicing;Biological rhythms;Chromosome;DNA damage;DNA repair;Nucleus;Phosphoprotein;Reference proteome;Repeat;Ubl conjugation;Ubl conjugation pathway;WD repeat |
|
|
mmu:50754; |
chromosome [GO:0005694]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; endoplasmic reticulum [GO:0005783]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; mitochondrion [GO:0005739]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; Parkin-FBXW7-Cul1 ubiquitin ligase complex [GO:1990452]; perinuclear region of cytoplasm [GO:0048471]; protein-containing complex [GO:0032991]; SCF ubiquitin ligase complex [GO:0019005]; cyclin binding [GO:0030332]; identical protein binding [GO:0042802]; phosphothreonine residue binding [GO:0050816]; protein-macromolecule adaptor activity [GO:0030674]; ubiquitin binding [GO:0043130]; ubiquitin ligase-substrate adaptor activity [GO:1990756]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin protein ligase binding [GO:0031625]; ubiquitin-protein transferase activator activity [GO:0097027]; cellular response to DNA damage stimulus [GO:0006974]; cellular response to UV [GO:0034644]; DNA repair [GO:0006281]; lung development [GO:0030324]; negative regulation of gene expression [GO:0010629]; negative regulation of osteoclast development [GO:2001205]; negative regulation of RNA polymerase II regulatory region sequence-specific DNA binding [GO:1903026]; Notch signaling pathway [GO:0007219]; positive regulation of epidermal growth factor-activated receptor activity [GO:0045741]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of oxidative stress-induced neuron intrinsic apoptotic signaling pathway [GO:1903378]; positive regulation of proteasomal protein catabolic process [GO:1901800]; positive regulation of protein targeting to mitochondrion [GO:1903955]; positive regulation of protein ubiquitination [GO:0031398]; positive regulation of ubiquitin-dependent protein catabolic process [GO:2000060]; positive regulation of ubiquitin-protein transferase activity [GO:0051443]; proteasome-mediated ubiquitin-dependent protein catabolic process [GO:0043161]; protein destabilization [GO:0031648]; protein K63-linked ubiquitination [GO:0070534]; protein stabilization [GO:0050821]; protein ubiquitination [GO:0016567]; regulation of autophagy of mitochondrion [GO:1903146]; regulation of cell migration involved in sprouting angiogenesis [GO:0090049]; regulation of circadian rhythm [GO:0042752]; rhythmic process [GO:0048511]; SCF-dependent proteasomal ubiquitin-dependent protein catabolic process [GO:0031146]; sister chromatid cohesion [GO:0007062]; ubiquitin recycling [GO:0010992]; vasculogenesis [GO:0001570] |
14739463_antagonizes the apoptotic c-Jun-dependent effector arm of JNK signaling, allowing neurons to tolerate potentially neurotoxic JNK activity 14766969_Fbw7-null mice die around 10.5 days post coitus because of a combination of deficiencies in hematopoietic and vascular development and heart chamber maturation. data suggest essential functions for Fbw7 in controlling cyclin E and Notch signaling 15592418_p53-dependent loss of Fbxw7 leads to genetic instability by mechanisms that might involve the activation of Aurora-A, providing a rationale for the early occurrence of these mutations in human cancers 16989775_expression of Fbxw7 isoforms is differentially regulated in a cell cycle- or p53-dependent manner 17984302_Fbxw7 regulates the cell cycle in a differentiation-dependent manner, with its loss resulting in c-Myc accumulation that leads to hyperproliferation in immature T cells but to p53-dependent cell-cycle arrest and apoptosis in mature T cells. 18367647_Fbxw7 functions as a fail-safe mechanism against both premature hematopoietic stem cells loss and leukemogenesis. 18641686_Fbxw7 does not function as an oncosuppressor in mouse embryo fibroblast. Instead, it promotes cell cycle progression and cell survival through degradation of Notch1. 19562935_regulates cell cycle by decomposing c-Myc protein. (review) 20534483_inactivation of Fbxw7 is sufficient to convert preadipocytes into mature adipocytes in a manner dependent on C/EBPalpha 20638938_Fbw7 regulates intestinal biology and tumorigenesis by controlling the abundance of different substrates in a dose-dependent fashion, providing a molecular explanation for the heterozygous mutations of fbw7 observed in human colorectal carcinoma. 20826802_miR-223 expression is responsive to acute alterations in cyclin E regulation by the Fbw7 pathway. 20935640_Fbw7 controls neurogenesis by antagonizing Notch and c-Jun N-terminal kinase (JNK)/c-Jun signaling. 21076392_C/EBPdelta directly inhibits expression of the tumour suppressor F-box and WD repeat-domain containing 7 gene (FBXW7, FBW7, AGO, Cdc4), encoding an F-box protein that promotes degradation of the mammalian target of rapamycin (mTOR). 21123947_Hepatic ablation of Fbxw7 resulted in hepatomegaly and steatohepatitis, with massive deposition of triglyceride, a phenotype similar to that observed in humans with nonalcoholic steatohepatitis. 21147854_SGK1 inhibits the Notch1 signaling pathway via phosphorylation of Fbw7. 21205095_Loss of Fbxw7 is associated with oxidative stress. 21282377_FBXW7 influences murine intestinal homeostasis and cancer, targeting Notch, Jun, and DEK for degradation. 21349854_Fbxw7 as a key regulator of the maintenance and differentiation of neural stem cells in the brain. 21368833_the E3 ubiquitin ligase SCF(FBW7) governs cellular apoptosis by targeting MCL1, a pro-survival BCL2 family member, for ubiquitylation and destruction in a manner that depends on phosphorylation by glycogen synthase kinase 3 21827743_The F-box protein Fbw7 is required for cerebellar development. 21911492_role of Fbw7alpha in hepatic lipid metabolism through Kruppel-like factor 5 (KLF5)/peroxisome proliferator-activated receptor gamma2 (PPARgamma2) pathway but not SREBP-1c protein 22348067_identify Fbxw7 as a p53-dependent tumor susceptibility gene. 22513362_Results suggest a posttranslational regulatory network in which the Pten and Fbxw7 pathways appear to converge on the regulation of Aurora-A level. 22665065_Fbxw7 controls proliferation and differentiation of keratinocytes, and exerts both inhibitory and stimulatory actions in skin carcinogenesis by counteracting the proliferation-promoting effect of c-Myc and tumor-suppressive effect of Notch. 22708077_SCF(Fbw7) modulates the NFkB signaling pathway by targeting NFkB2 for ubiquitination and destruction. 22710480_Fbxw7 was decreased in NAFLD and negatively correlated with SREBP-1, indicating that the Fbxw7-SREBP-1 axis may play a key pathological role in the development of NAFLD. 22797700_Fbw7 gamma - mediated ubiquitination of KLF13 prevents RANTES expression in resting human but not murine T lymphocytes. 22848434_Fbxw7 is a potent positive regulator of angiogenesis that limits the activity of Notch in the endothelium of the growing vasculature. 22897623_Depletion of Fbxw7 resulted in promotion of induced pluripotent stem cell generation. 22912667_a number of miR-25 candidate gene targets 23454868_inhibition of mTOR signaling pathway suppresses the contribution of Fbxw7 loss toward tumor development. 23518349_findings show that Fbxw7 plays a pivotal role in maintenance of quiescence in nondividing leukemia-initiating cells of chronic myeloid leukemia by reducing the level of c-Myc 23518350_We demonstrate here the essential function of the Fbw7 E3 ligase for the initiation and the progression of chronic myelogenous leukemia. 23575666_findings reveal a role for C/EBPdelta upstream of Tlr4 signalling and uncover a function for FBXW7alpha as an attenuator of inflammatory signalling 23676439_Heterozygous Fbxw7 propellor tip (R482Q) mutations promote intestinal tumors in mice. Heterozygous null Fbxw7 mutations also promote tumours, but the effect is weaker than R482Q. Findings explain the FBXW7 mutation spectrum found in human cancers. 23776410_the downstream Notch signalling effector HES5 directly represses transcription of the E3 ligase Fbw7beta. 23820376_Fbw7 together with GSK3beta negatively regulates G-CSFR expression and its downstream signaling. 23858059_Loss of parkin function through biallelic mutation of PARK2 may lead to death of dopaminergic neurons through unregulated SCF(Fbw7beta)-mediated ubiquitylation-dependent proteolysis of Mcl-1. 23955342_Fbxw7 controls osteogenesis and chondrogenesis by targeting OASIS and BBF2H7, respectively, for degradation. 24457827_p50 upregulated c-Myc protein expression mainly through inhibiting its degradation. p50 exhibited this novel property by suppression of FBW7 expression. 24820417_Data propose that control of GATA3 levels by Fbw7 contributes to the fine-tuning of T-cell development. 24879440_FBXW7 regulates spermatogonial stem cell self-renewal in a negative manner by degradation of MYC 24884509_Fbxw7 induces apoptosis and growth arrest by regulating YAP abundance in hepatocellular carcinoma. 24958101_demonstrate the importance of Fbw7-dependent cyclin E control to the hematopoietic system and highlight chromosome instability as a characteristic feature of dysfunction and malignancy induced by deregulated cyclin E 25105579_Fbw7 is a master regulator of cell fate decisions in the pancreas 25437563_Dual regulation of Fbw7 activity by Usp28 is a safeguard mechanism for maintaining physiological levels of proto-oncogenic Fbw7 substrates, which is equivalently disrupted by loss or overexpression of Usp28. 25555218_these results suggest that FBXW7 antagonizes cancer development in not only a cell-autonomous manner, but also a non-cell-autonomous manner 25579381_Prion infection induced the expression of FBXW7 in brain. 25670854_Ubiquitin-dependent degradation of GATA 2 is promoted by Fbw7, is cyclin B-CDK1-mediated Thr176 phosphorylation-dependent, and influences hematopoietic cell differentiation. 25716680_In mice, an unusually direct antagonism between an E3 ligase and a deubiquitinase, Fbw7 and Usp28, modulate intestinal homeostasis and cancer. 25720964_FBXW7 modulates cellular stress response and metastatic potential through HSF1 post-translational modification. 26206584_These data show that cell cycle-dependent mechanisms can control ciliary length through a CDK5-FBW7-NDE1 pathway. 26575021_Gene expression profiling reveals transcriptional regulation by Fbxw7/mTOR pathway in radiation-induced mouse thymic lymphomas. 26774286_FBXW7 facilitates nonhomologous end-joining via K63-linked polyubiquitylation of XRCC4 in tumor cells. 27238018_Study identifies a REV-ERBalpha post-translational regulatory circuit in which cyclin-dependent kinase 1 (CDK1) phosphorylation of REV-ERBalpha is recognized by the F-box protein, FBXW7alpha, to direct REV-ERBalpha degradation via the proteasome. Disruption of this CDK1-FBXW7-mediated REV-ERBalpha degradation pathway in mouse liver alters circadian rhythmicity, in particular amplitude, and whole-body lipid/glucose home... 27594513_Myoblast differentiation potential and muscle regeneration can be regulated by Fbxw7beta. 27764699_Fbxw7 suppresses KrasG12D-induced pancreatic tumorigenesis via a Yap-dependent mechanism. 27773673_The findings reveal a PLK1-Fbw7-Myc signaling circuit that underlies tumorigenesis and validate PLK1 inhibitors, alone or with Bcl2 antagonists, as potential effective therapeutics for MYC-overexpressing cancers. 28036276_EglN2 might act as an FBW7 ubiquitin ligase substrate contributing to the progression of triple negative breast cancer. 28287082_FBXW7 is critical for RIG-I stabilization during antiviral responses. 28581198_Loss of Fbxw7 in the presence of BrafV600E mutation is consequential and sufficient to drive melanoma development. 28752701_Myeloid-specific deficiency of FBXW7 can inhibit lung metastasis of B16F10 melanoma in mice, and the mechanism may be associated with regulation of MAM in the metastatic tumor lesions 29074464_the regulatory crosstalk between KLF5, miR-29a, and Fbw7/CDC4 cooperatively promotes atherosclerotic development 29149593_These findings highlight the molecular basis of Hajdu-Cheney syndrome (HCS) pathogenesis and provide clinical insights into potential targeted therapeutic strategies for skeletal disorders associated with the aberrant FBW7/NOTCH2 pathway as observed in patients with HCS. 29346117_identify a tumor suppressor mechanism in the mammalian intestine that arises from the posttranslational regulation of FBW7 by USP9X independent of somatic FBW7 mutations. 29386660_A novel mouse line carrying a conditional knockin allele of a cancer-specific FBXW7 mutation was established for carcinogenesis study. 29472293_results thus suggest that Fbxw7 controls the transcription of MyRF target genes in various tissues through regulation of MyRF protein stability in a manner dependent on MyRF phosphorylation by GSK-3. 29475832_FBXW7 is markedly downregulated in the liver of obese mice. Mechanistically, FBXW7 directly binds to hepatokine fetuin-A to induce its ubiquitination and subsequent proteasomal degradation, comprising an important mechanism maintaining glucose homeostasis. 29550488_found that Fbw7 loss caused activation of NF-kappaB signaling. Thus, FBW7 plays a protective role in acute intestinal inflammation by modulating the inflammatory response of NF-kappaB pathway. 29633504_loss of FBXW7 promotes non-small-cell lung cancer progression as well as gefitinib resistance. 30275535_this study shows that FBXW7 aggravates TMPD-induced systemic lupus erythematosus by promoting cell apoptosis 30830867_Expression of the Fbxw7 gene is upregulated in dormant breast cancer cells and its disruption results in a purge of tumor cells from the quiescent state, rendering them susceptible to chemotherapy. Analysis of clinical data retrieved from The Cancer Genome Atlas also revealed that breast cancer patients with a high level of FBXW7 expression in the primary tumor had a poorer prognosis compared with those with a low level. 30854564_Here we report that DNA bound IRF1 turnover is promoted by GSK3b (Glycogen Synthase Kinase 3b) via phosphorylation of the T181 residue which generates a phosphodegron for the SCF (Skp-Cul-Fbox) ubiquitin E3-ligase receptor protein Fbxw7a (F-box/WD40 7). 31017010_TLR4 regulates SCF(Fbxw7alpha)-dependent Fn14 stabilization during septic acute kidney injury. 31150734_These data identify FBXW7 as a direct target of miR322 and suggest that miR322 could have potential therapeutic application for cardioprotection against ischemia/reperfusion-induced injury. 31246581_FBXW7 degrades EZH2 and increases Ccl2/Ccl7 in CX3CR1hi macrophages, which promotes the recruiting CX3CR1int pro-in fl ammatory mononuclear phagocytes into local colon tissues with colitis 31278268_Study finds that loss of Fbxw7 enhances the myelinating potential of Schwann cells (SCs). Fbxw7 mutant SCs make thicker myelin sheaths and sometimes appear to myelinate multiple axons in a fashion reminiscent of oligodendrocytes. Taken together, findings show that the restriction of myelinating SCs to myelinate a single axon may be mutable and that Fbxw7 is a critical player in regulating the myelinating potential of SCs. 31341888_study indicates that miR-195-5p promotes cardiac hypertrophy via targeting MFN2 and FBXW7 and may provide promising therapeutic strategies for interfering cardiac hypertrophy. 31423512_The FBXW7alpha/miR-205 axis may regulate tumor associated macrophage polarization by affecting SMAD1 expression. 31445163_Our results suggest that FBW7 is an important regulator in cerebrovascular remodeling 31883011_Loss of Fbxw7 in Sertoli cells impairs testis development and causes infertility in micedagger. 32005754_E3 Ubiquitin Ligase Fbw7 Regulates the Survival of Mature B Cells. 32152552_Sox12 enhances Fbw7-mediated ubiquitination and degradation of GATA3 in Th2 cells. 32371478_Inactivation of Fbxw7 Impairs dsRNA Sensing and Confers Resistance to PD-1 Blockade. 32380038_FBXW7 promotes pathological cardiac hypertrophy by targeting EZH2-SIX1 signaling. 32900488_FBW7 protects against spinal cord injury by mitigating inflammation-associated neuronal apoptosis in mice. 33070870_Loss of Fbxw7 triggers mammary tumorigenesis associated with E2F/c-Myc activation and Trp53 mutation. 33234509_Loss of Fbxw7 Impairs Development of and Induces Heterogeneous Tumor Formation in the Mouse Mammary Gland. 33260160_E3 ligase FBXW7 restricts M2-like tumor-associated macrophage polarization by targeting c-Myc. 33369138_The anti-angiogenesis role of FBXW7 in diabetic retinopathy by facilitating the ubiquitination degradation of c-Myc to orchestrate the HDAC2. 33537812_FBXW7 mediates high glucoseinduced SREBP1 expression in renal tubular cells of diabetic nephropathy under PI3K/Akt pathway regulation. 33821962_Inositol pyrophosphates promote MYC polyubiquitination by FBW7 to regulate cell survival. 34351636_Critical roles of the E3 ubiquitin ligase FBW7 in B-cell response and the pathogenesis of experimental autoimmune arthritis. 34518642_FBW7 couples structural integrity with functional output of primary cilia. 34998785_E3 ubiquitin ligase FBXW7 balances airway cell fates. 35058228_Mechanical overloading promotes chondrocyte senescence and osteoarthritis development through downregulating FBXW7. 35069531_Myeloid Fbxw7 Prevents Pulmonary Fibrosis by Suppressing TGF-beta Production. 35651754_E3 Ligase FBXW7 Facilitates Mycobacterium Immune Evasion by Modulating TNF-alpha Expression. 36104351_FBXW7 inactivation induces cellular senescence via accumulation of p53. |
ENSG00000109670 |
FBXW7 |
456.504088 |
2.0521442530 |
1.037132 |
0.14102297 |
53.705687 |
0.00000000000023288821023074914740970242887869106436157795592123420647112652659416198730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000050320485432782254516072909899484822153145624490377940674079582095146179199218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
600.841592169593 |
58.8256465451605 |
292.787210878213 |
22.0245139945887 |
| ENSMUSG00000028128 |
14066 |
F3 |
protein_coding |
A0A0R4J088 |
FUNCTION: Initiates blood coagulation by forming a complex with circulating factor VII or VIIa. The [TF:VIIa] complex activates factors IX or X by specific limited proteolysis. TF plays a role in normal hemostasis by initiating the cell-surface assembly and propagation of the coagulation protease cascade. {ECO:0000256|ARBA:ARBA00002201, ECO:0000256|PIRNR:PIRNR002498}. |
Blood coagulation;Disulfide bond;Glycoprotein;Hemostasis;Lipoprotein;Membrane;Palmitate;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a membrane-bound glycoprotein that forms the primary physiological initiator of the blood coagulation process following vascular damage. The encoded protein binds to coagulation factor VIIa and the ensuing complex catalyzes the proteolytic activation of coagulation factors IX and X. Mice lacking encoded protein die in utero resulting from massive hemorrhaging in both extraembryonic and embryonic vessels. A severe deficiency of the encoded protein in mice results in impaired uterine homeostasis, shorter life spans due to spontaneous fatal hemorrhages and cardiac fibrosis. [provided by RefSeq, Aug 2015]. |
mmu:14066; |
cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; membrane [GO:0016020]; serine-type peptidase complex [GO:1905286]; phospholipid binding [GO:0005543]; protease binding [GO:0002020]; serine-type endopeptidase activity [GO:0004252]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; activation of plasma proteins involved in acute inflammatory response [GO:0002541]; blood coagulation [GO:0007596]; positive regulation of angiogenesis [GO:0045766]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of interleukin-8 production [GO:0032757]; positive regulation of platelet-derived growth factor receptor signaling pathway [GO:0010641]; positive regulation of protein kinase B signaling [GO:0051897] |
11776298_TF deficiency in mice causes embryonic death. TF is needed for vascular development in the embryo and tumor angiogenesis. 12244306_The initiation of blood coagulation in vivo entailed the initial accumulation of tissue factor on the upstream and thrombus-vessel wall interface of the developing thrombus, and fibrin generation within the thrombus. 12393670_tissue distribution of age and obesity augmented, stress-induced expression of tissue factor gene 12426405_Tissue factor deficiency causes cardiac fibrosis and left ventricular dysfunction. TF expression by cardiac myocytes provides a secondary hemostatic barrier to protect the heart from hemorrhage. 12782720_P-selectin expression, tissue factor accumulation, and fibrin generation after platelet localization in the developing thrombus in arterioles 15003826_endotoxin-induced coagulation and inflammation are strongly influenced by blood cell-derived tissue factor 15073034_Identification of endothelial TF in the sickle lung, these studies implicate reperfusion injury physiology in its expression and suggest a rationale for use of statins in sickle disease. 15117736_Review. TF deficiency causes embryonic lethality and fatal hemorrhages in the adult mouse. 15280200_blood-borne TF associated with hematopoietic cell-derived microparticles contributes to thrombus propagation 15339841_In vitro analyses revealed that TF activity in the blood was very low and was markedly exceeded by that present in the vessel wall 15351840_data suggest that TF may mediate intimal hyperplasia by regulating smooth muscle cell migration 15576370_Annexin A5 down-regulates surface-expressed TF by activating the novel portal of cell entry 15598816_the correct balance between TF and TFPI in different organs is required to maintain hemostasis during embryonic development and in adult mice. 15613025_TF deficiency reduces cytokine production in vitro, but an enhanced induction of TF during murine endotoxemia eliminates this effect in vivo 15947236_Grp78 negatively regulates TF functional activity via direct binding to and functional inhibition of tissue factor 15983216_TF expression is an important link between the early inflammatory response to cutaneous wounding and reparative processes. 16020755_TF cytoplasmic domain signaling alters vascular remodeling and, thereby, may play a role in the development of restenosis, atherosclerotic disease, and neovascularization. 16236262_ability of epithelial cells to interact with the vascular system via expression of the TF gene (and other effectors) is under the control of complex alterations in cellular architecture 16385085_either a 50% reduction of TF in all cells or a selective reduction in hematopoietic cell-derived TF does not affect the development of atherosclerotic lesions in mice 16409465_Identified and characterized murine masTF, a soluble splice variant of TF; it is widely and abundantly expressed in adult tissues, in experimentally induced thrombi, and during development. 16409467_in the heart, TF plays an important role in the thrombogenesis and it counterbalances AT-dependent anticoagulation; regulation of TF-procoagulant activity take place differently between the liver and the heart 16634773_Streptococcus pneumoniae induces alternative splicing of tissue factor in mice 17234207_down-regulation of urokinase plasminogen activator in female low-tissue factor mice protects them from more severe cardiac fibrosis 17332247_role of the TF cytoplasmic domain in an autoregulatory pathway that controls LPS-induced TF expression in macrophages and procoagulant responses in endotoxemia 17536017_identification of TF as an important mediator of C5a-induced oxidative burst in neutrophils in antiphospholipid antibody-induced fetal injury provides a new target for therapy to prevent pregnancy loss 17562818_Findings implicate tissue factor in intestinal inflammation and support interaction between inflammation and coagulation in experimental colitis. 17663739_TF expression by cardiac myocytes is important to maintain heart hemostasis under normal and pathologic conditions 18055866_Perivascular TF expression is down-regulated during wound healing which may prevent thrombosis of neovessels during angiogenesis but renders hemophiliacs vulnerable to hemorrhage during healing. 18276908_In vivo analyses revealed significant increases in TF expression and decreases in TFPI expression in carotid arteries of CRP-Tg mice versus WT mice. 18495150_Viral myocarditis is a hypercoagulative state which is associated with increased myocardial TF expression and activity. Upregulation of TF contributes to a systemic activation of the coagulation cascade. 18647222_in the presence of a limited level of the anticoagulant, PC, placental homeostasis can be restored by reducing TF expression, thus facilitating proper embryonic implantation 18650785_VIP and PACAP inhibit LPS-induced TF expression in monocytes in vitro and in vivo 18763309_occlusion of tumor vasculature with rVEGF-TF is potentially an effective approach for cancer therapy 18772494_Study points to an important but context-dependent role of host TF in tumor formation, angiogenesis and therapy. 18931346_Vascular smooth muscle cell-derived tissue factor is critical in a macrovascular model of arterial thrombosis 19095643_regulation of TF by NF-kappaB transcription factor p50 is essential for the pathogenesis of deep vein thrombosis 19179621_The results indicate that alpha-naphthylisothiocyanate-induced liver injury is accompanied by tissue factor (TF)-dependent activation of the coagulation cascade and that TF contributes to the progression of injury. 19234141_TF is an important mediator in fetal death and growth restriction 19350121_Leukocyte- and platelet-derived microparticles correlate with thrombus weight and tissue factor activity in an experimental mouse model of venous thrombosis. 19447494_mTOR dependent IL-10 expression leads to inhibition of LPS induction of TF and proinflammatory cytokine TNFalpha in WT macrophages. In contrast, the decrease in LPS-induced TNFalpha and TF expression in PTEN(-/-) PMs requires an IL-10-independent pathway. 19535796_Targeting TF might be a good therapeutic intervention in patients with thrombotic microangiopathy. 19546589_Data suggest that tissue factor and factor V induction by LPS may in part accelerate mesangioproliferative glomerulonephritis through activation of factor X and downstream proinflammatory and procoagulant mechanisms. 19556010_mediates neutrophil activation and fetal death in a mouse model of antiphospholipid syndrome 19620042_Tanshinone IIA inhibits cell proliferation and TF and MMP-12 expressions in RAW264.7 cells stimulated by ox-LDL. 19662362_Smooth muscle cells may be the major source of TF in atherosclerosis without plaque rupture. 19712679_Laminin receptor activation inhibits endothelial TF expression by impairing JNK phosphorylation 19875693_asTF enhances angiogenesis in vivo, ex vivo, and in vitro independent of downstream coagulation factors or activation of PAR-2, but dependent on alphavbeta3 and alpha6beta1 integrin function. 19954621_TF gene and protein expression was significantly elevated in mice treated by allogenic hematopoietic stem cell transplantation. 20008134_TF-dependent thrombin generation and activation of PAR-1 amplify hepatic inflammation and injury during the pathogenesis of steatohepatitis. 20029945_Endothelial nitric oxide synthase and nitric oxide regulate endothelial tissue factor expression in vivo in the sickle transgenic mouse 20170942_review of wound healing in hemophilia B mice and low tissue factor mice [review] 20185792_Acadesine inhibits TF expression and thrombus formation by activating the phosphoinositide 3-kinase/Akt pathway. 20303465_Peripheral blood mononuclear cells indirectly promote endothelial TF expression via a NFkappaB(p50)-dependent mechanism. 20410508_Deletion of the mouse TF gene in either ECs or VSMCs did not reduce TAT levels at 8 hours after LPS, indicating that TF expressed by other nonhematopoietic cells contributes to activation of coagulation in endotoxemic mice. 20626618_tissue factor expression contributes to exacerbated diabetic nephropathy in mice lacking eNOS fed a high fat diet 20671239_RANKL induces TF in macrophages mainly through the cooperative action of AP-1 and Egr-1 via JNK and ERK1/2 pathways 20810696_the TWEAK-Fn14 axis can regulate activation of TF and PAI-1 expression in vascular cells 20858853_An accompanying genetic severe deficiency of tissue factor protects mice with a protein C deficiency from lethal endotoxemia. 20978007_Sirt1 inhibition induces tissue factor expression and arterial thrombus formation in vivo. 21042291_Upregulated tissue factor(TF) expression may be associated with age-related macular degeneration(AMD), and inflammatory and oxidative stress may contribute to TF expression in AMD eyes. 21149441_EPCR interacts with the ternary TF coagulation initiation complex to enable PAR1,2 signaling. 21262438_This study identified tissue factor (TF) as a crucial mediator of fetal and placental damage in mouse models of recurrent miscarriages and preeclampsia. 21389637_soluble alternatively spliced (as)TF may serve as a migration- and angiogenesis-promoting factor 21496886_Findings implicate role for cAMP in regulation of the gene expression of tissue factor and plasminogen activator inhibitor-1 in adipocytes. 21658367_RAR-a, RXR-a and TF may emerge as important regulatory molecules and play important role in liver injury. 21717035_Data show that tissue factor plays a detrimental role in experimental colitis by signal transduction-dependent keratinocyte-derived chemokine production in colon, thereby provoking granulocyte influx with subsequent inflammation and organ damage. 22019885_Data show that mice lacking PAR2 or the cytoplasmic domain of tissue factor were protected from weight gain and insulin resistance induced by a high-fat diet. 22192154_TF expression by myeloid cells does not significantly contribute to venous thrombogenesis in an electrolytic injury model. 22214850_Results suggest that the prothrombotic state associated with hypercholesterolemia is caused by oxLDL-mediated induction of tissue factor expression in monocytes via engagement of a TLR4/TLR6 complex. 22326437_alternatively spliced tissue factor (TF) and full-length TF protect cardiomyocytes against TNF-alpha-induced apoptosis via activation of specific signaling pathways, and up-regulation of anti-apoptotic members of the Bcl-2 protein family. 22407318_the microbiota-induced extravascular TF-PAR1 signalling loop is a novel pathway that may be modulated to influence vascular remodelling in the small intestine 22481268_Authors propose that murine alternatively spliced tissue factor is a functional homolog of murine alternatively spliced tissue factor, exerting some of its key effects via beta3 integrins. 22547577_Only TF-positive cell lines activated coagulation in nude mice, and this activation was abolished by an anti-human TF Ab. 22661702_Our data indicate that different cellular sources of TF contribute to activation of coagulation, vascular inflammation, and endothelial cell injury in sickle cell disease. 22744182_Findings suggest a gene therapy for hemophilia A treatment using factor VIII (FVIII)-dual vectors. 22865892_Cyclooxygenase-2-derived prostacyclin regulates arterial thrombus formation by suppressing tissue factor in a sirtuin-1-dependent-manner. 22950420_FVIIa binding to EPCR on the endothelium facilitates the transport of FVIIa from circulation to extravascular tissues where TF resides 23038262_PAK1 negatively regulates the expression of TFPI and additionally contributes to increased TF activity. 23149844_vascular Gas6 contributes to thrombus formation in vivo and can be explained by the ability of Gas6 to promote tissue factor expression and activity. 23238822_TF expression is regulated by JAK2-STAT3 signaling. 23291174_the circadian expression of TF mRNA is regulated by clock molecules through a cell-autonomous mechanism via an E-box element located in the promoter region. 23328933_endosomal signalling may represent a central coordinating point in the process of induction of TF expression 23347132_Data suggest a tissue factor (TF) and protease-activated receptor 2 (PAR-2)-dependent mechanism on dexamethasone-dendritic cells (DC) in vitro and unprimed DC in vivo that contributes to the low immunogenicity of these cells. 23494077_Data indicate that hfVII-LC and hIgG1-Fc can effectively inhibit tumor growth and metastases in SCID mice with tissue factor (TF) over-expressing colon cancer. 24086497_TF expression plays a pivotal role in the pathogenesis of MPE generation via regulating of tumor metastasis and vascular permeability in lung adenocarcinoma bearing activated Stat3. 24520174_Tissue factor expression provokes escape from tumor dormancy and leads to genomic alterations. 24869936_New insights into the spatiotemporal localization of prothrombinase in vivo. 24879059_Alternatively spliced tissue factor is not sufficient for embryonic development 25015658_Fas-initiated, caspase-3-dependent hepatocyte apoptosis increases tissue factor procoagulant activity through a mechanism involving phosphatidylserine externalization. 25116956_Alternatively spliced tissue factor promotes plaque angiogenesis through the activation of hypoxia-inducible factor-1alpha and vascular endothelial growth factor signaling. 25275013_demonstrate that TLR-4 contributes to arterial thrombosis formation in vivo and causes increased TF expression and activity in vitro 25293769_study provides a novel mechanism for the posttranscriptional regulation of TF expression, indicating that this is selectively regulated by PARP-14. 25339356_Klkb1(-/-) mice have a novel mechanism for thrombosis protection in addition to reduced contact activation. This pathway arises when bradykinin delivery to vasculature is compromised and mediated by increased receptor Mas, prostacyclin, Sirt1, and KLF4 25450387_Mice in which TF gene is disrupted in the neuroectodermal lineage exhibit delayed progression of spontaneous brain tumors driven by oncogenic N-ras and SV40 large T antigen (SV40LT) expressed under the control of sleeping beauty transposase. 25451155_TF is essential for the development of pericyte coverage of tumor microvessels and aPL-induced tumor cell expression of chemokine ligand 2, a mediator of pericyte recruitment 25596265_Heme promotes tissue factor-dependent coagulation activation and induces tissue factor expression on leukocytes in vivo. 25884207_Lung epithelial tissue factor regulates alveolar procoagulant activity and permeability in acute lung injury. 25955268_TF expressed on tumor microparticles contributes to the increased incidence of cancer-associated venous thrombosis in mice in vivo. 26065856_PGE2 increases both TF expression and activity through the regulation of the EP1/SIRT1 pathway. 26325534_Hepatocyte TF activates coagulation in a mouse model of chronic liver injury 26341257_These findings reveal a novel biological function and mechanism of the protein C pathway in which protein S and the aPC-cleaved form of fV are cofactors for anti-inflammatory cell signaling by aPC in the context of endotoxemia and infection 26471500_expression by myeloid cells affects macrophage differentiation, and regulate Mycobacterium tuberculosis growth 26817955_Thrombin-independent contribution of tissue factor to inflammation and cardiac hypertrophy in a mouse model of sickle cell disease. 26924425_There was no significant contribution of TF on myeloid cells in multiple models of experimental ALI, leading to the conclusion that TF in myeloid cells is not a major contributor to experimental ALI. 28028070_Ticagrelor, but not clopidogrel, reduces arterial thrombosis via endothelial tissue factor suppression. Ticagrelor reduced TNF-alpha-induced TF expression via proteasomal degradation. 28124089_Evening primrose oil and forskolin decreased the prothrombotic effect of celecoxib initiated by a LPS challenge in mice, and this effect was, at least partly, mediated by mitigating TF expression and activity. 28350198_We conclude that brain TF plays an important role in localization of hemorrhage and also regulates cerebrospinal fluid flow under normal conditions 28495929_Macrophage tissue factor prothrombotic activity is regulated by integrin-alpha4/arf6 trafficking. 28509332_Myeloid cell derived tissue factor dampens inflammation in acid-induced acute lung injury. 28883694_Tissue factor cytoplasmic domain is involved in TF-PAR-2 signalling initiating hepatic fibrosis and is a potential therapeutic target, as its deletion would not impact coagulation. 29427323_Androgen receptor is crucial for dampening prostate tissue factor expression via nuclear factor-kappaB and early growth response protein 1 pathway. 29444200_Sirt3 loss-of-function enhances experimental thrombosis in vivo via an increase of neutrophil extracellular traps and elevation of TF suggesting thrombo-protective effects of endogenous Sirt3. 30500376_TF overexpression enhanced plaque thrombogenicity, which played a pivotal role in atherothrombosis in ApoE-/- mice. 30602303_The expression of tissue factor was increased in the artery of Klf11 knockout mice. Vascular smooth muscle cells isolated from Klf11 knockout mouse aortas showed increased tissue factor expression, which was rescued by KLF11 overexpression. 31262782_These data indicate that lipopolysaccharide - or cytokine-induced TF procoagulant activity requires the decryption of newly synthesized TF protein by ASMase-mediated hydrolysis of sphingomyelin. 32559654_Hypoxia induced up-regulation of tissue factor is mediated through extracellular RNA activated Toll-like receptor 3-activated protein 1 signalling. 33498089_The Intrinsic Pathway does not Contribute to Activation of Coagulation in Mice Bearing Human Pancreatic Tumors Expressing Tissue Factor. 34155039_Cardiac Tissue Factor Regulates Inflammation, Hypertrophy, and Heart Failure in Mouse Model of Type 1 Diabetes. |
ENSG00000117525 |
F3 |
2767.667867 |
2.1660057879 |
1.115037 |
0.06303365 |
309.145135 |
0.00000000000000000000000000000000000000000000000000000000000000000000335314322442812302740571759986419664804239266822771558341497465357110196248729769175043666384050653173514846673426902243754586723379828639417564517808101329878670039539223166058512504150712629780 |
0.0000000000000000000000000000000000000000000000000000000000000000009286377744452576007758275140108928896327759696318839748935707885255096662002176389071181550063576253819862827511221178558087914173225765557959397100031568120733524267229608284424102748744189739 |
Yes |
No |
3774.70334692259 |
164.975296243782 |
1742.70233629779 |
58.361841245912 |
| ENSMUSG00000028175 |
76131 |
Depdc1a |
protein_coding |
Q8CIG0 |
FUNCTION: May be involved in transcriptional regulation as a transcriptional corepressor. The DEPDC1A-ZNF224 complex may play a critical role in bladder carcinogenesis by repressing the transcription of the A20 gene, leading to transport of NF-KB protein into the nucleus, resulting in suppression of apoptosis of bladder cancer cells (By similarity). {ECO:0000250}. |
Alternative splicing;Coiled coil;Direct protein sequencing;GTPase activation;Nucleus;Phosphoprotein;Reference proteome;Repressor;Transcription;Transcription regulation |
|
|
mmu:76131; |
nucleus [GO:0005634]; transcription repressor complex [GO:0017053]; GTPase activator activity [GO:0005096]; intracellular signal transduction [GO:0035556]; negative regulation of DNA-templated transcription [GO:0045892] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20587513_Coimmunoprecipitation and immunocytochemistry revealed that DEPDC1 interacted and colocalized with zinc finger transcription factor ZNF224, a known transcriptional repressor. 23646139_Inhibition of DEPDC1A, a bad prognostic marker in multiple myeloma, delays growth and induces mature plasma cell markers in malignant plasma cells. 25064737_DEPDC1 regulates vincristine-induced cell death by promoting JNK-dependent degradation of the BCL-2 family protein MCL1. 25605201_High DEPDC1 mRNA expression is associated with hepatocellular carcinoma/ 25902835_DEPDC1, plays a pivotal role in the regulation of proper mitotic progression. 26970279_Results suggest that protocadherin 10 (PCDH10)-Dishevelled, EGL-10 and Pleckstrin domain containing 1 (DEPDC1)-caspase signaling may be a novel regulatory axis in endometrial endometrioid carcinoma (EEC) development. 27984115_miR-130a is an epigenetically regulated miRNA involved in regulation of key molecular and phenotypic features of prostate carcinogenesis, acting as a tumour suppressor miRNA by targeting SEC23B and DEPDC1. 28555424_The results of this study suggest that DEPDC1 represents a target molecule for the treatment of glioma. 28602627_DEPDC1a, but not DEPDC1b, was required for the integrity of centrosome and organization of the bipolar spindle 28634077_DEPDC1 mRNA and protein expression levels were dramatically increased in prostate cancer tissues and cell lines. Overexpression of DEPDC1 promoted, but depletion of DEPDC1 inhibited cell proliferation by regulating the G1-S phase cell cycle transition. 29297565_Our results demonstrated that increased expression of SDP35/DEPDC1A and XPT1/DEPDC1B correlates with metastatic progression of metastatic soft tissue sarcoma 30417471_Differential DEP domain containing 1 (DEPDC1) expression level shows diagnostic and prognostic significance in liver hepatocellular carcinoma (LIHC) samples. 30419349_DEPDC1, negatively regulated by miR-26b, promotes cell proliferation and tumor growth via up-regulating FOXM1 expression, implying an important underlying mechanism of regulating the progression of triple negative breast cancer. 31162714_MicroRNA-374c-5p inhibits the development of breast cancer through TATA-box binding protein associated factor 7-mediated transcriptional regulation of DEP domain containing 1. 31189746_DEPDC1 promotes cell proliferation and suppresses sensitivity to chemotherapy in human hepatocellular carcinoma. 31322256_High DEPDC1 expression is associated with hepatocellular carcinoma cell proliferation, invasion and angiogenesis. 31366540_High expression of DEPDC1 in tumor tissue appears to be associated with tumor progression and poor prognosis 32343606_DEP Domain Containing 1 Promotes Proliferation, Invasion, and Epithelial-Mesenchymal Transition in Colorectal Cancer by Enhancing Expression of Suppressor of Zest 12. 33021072_DEPDC1 up-regulates RAS expression to inhibit autophagy in lung adenocarcinoma cells. 33140894_Overexpression of gene DEP domain containing 1 and its clinical prognostic significance in colorectal cancer. 33361586_DEPDC1 upregulation promotes cell proliferation and predicts poor prognosis in patients with gastric cancer. 33491970_Long noncoding RNA KTN1 antisense RNA 1exerts an oncogenic function in lung adenocarcinoma by regulating DEP domain containing 1 expression via activating epithelial-mesenchymal transition. 34134185_[DEPDC1 is Highly Expressed in Lung Adenocarcinoma and Promotes Tumor Cell Proliferation]. 34210956_ALPK2 acts as tumor promotor in development of bladder cancer through targeting DEPDC1A. 34741030_Linc-ROR facilitates progression and angiogenesis of hepatocellular carcinoma by modulating DEPDC1 expression. 35466755_Long non-coding RNA DEPDC1-AS1 promotes proliferation and migration of human gastric cancer cells HGC-27 via the human antigen R-F11R pathway. 35795985_FOXO3amodulated DEPDC1 promotes malignant progression of nephroblastoma via the Wnt/betacatenin signaling pathway. 36309731_Silencing eL31 suppresses the progression of colorectal cancer via targeting DEPDC1. |
ENSG00000024526 |
DEPDC1 |
701.967278 |
2.0735831266 |
1.052126 |
0.13034614 |
64.522369 |
0.00000000000000095442605926081083519547748991701465967425950000235790326996720978058874607086181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000260534368004671505792662427117252900597905475310600564853302785195410251617431640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
945.427481107633 |
85.5347433077269 |
455.939030838348 |
31.5026464398045 |
| ENSMUSG00000028179 |
107869 |
Cth |
protein_coding |
Q8VCN5 |
FUNCTION: Catalyzes the last step in the trans-sulfuration pathway from L-methionine to L-cysteine in a pyridoxal-5'-phosphate (PLP)-dependent manner, which consists on cleaving the L,L-cystathionine molecule into L-cysteine, ammonia and 2-oxobutanoate. Part of the L-cysteine derived from the trans-sulfuration pathway is utilized for biosynthesis of the ubiquitous antioxidant glutathione. Besides its role in the conversion of L-cystathionine into L-cysteine, it utilizes L-cysteine and L-homocysteine as substrates (at much lower rates than L,L-cystathionine) to produce hydrogen sulfide (H2S). In vitro, it converts two L-cysteine molecules into lanthionine and H2S, and two L-homocysteine molecules to homolanthionine and H2S, which can be particularly relevant under conditions of severe hyperhomocysteinemia. Lanthionine and homolanthionine are structural homologs of L,L-cystathionine that differ by the absence or presence of an extra methylene group, respectively (By similarity). Acts as a cysteine-protein sulfhydrase by mediating sulfhydration of target proteins: sulfhydration consists of converting -SH groups into -SSH on specific cysteine residues of target proteins such as GAPDH, PTPN1 and NF-kappa-B subunit RELA, thereby regulating their function (PubMed:19903941, PubMed:22244329). By generating the gasotransmitter H2S, it participates in a number of physiological processes such as vasodilation, bone protection, and inflammation (PubMed:18948540) (By similarity). Plays an essential role in myogenesis by contributing to the biogenesis of H2S in skeletal muscle tissue (PubMed:33826201). Can also accept homoserine as substrate (By similarity). Catalyzes the elimination of selenocystathionine (which can be derived from the diet) to yield selenocysteine, ammonia and 2-oxobutanoate (By similarity). {ECO:0000250|UniProtKB:P18757, ECO:0000250|UniProtKB:P32929, ECO:0000269|PubMed:18948540, ECO:0000269|PubMed:19903941, ECO:0000269|PubMed:22244329, ECO:0000269|PubMed:33826201}. |
Amino-acid biosynthesis;Calmodulin-binding;Cysteine biosynthesis;Cytoplasm;Lipid metabolism;Lyase;Pyridoxal phosphate;Reference proteome |
PATHWAY: Amino-acid biosynthesis; L-cysteine biosynthesis; L-cysteine from L-homocysteine and L-serine: step 2/2. {ECO:0000250|UniProtKB:P32929}. |
|
mmu:107869; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; calmodulin binding [GO:0005516]; carbon-sulfur lyase activity [GO:0016846]; cystathionine beta-lyase activity [GO:0004121]; cystathionine gamma-lyase activity [GO:0004123]; homocysteine desulfhydrase activity [GO:0047982]; identical protein binding [GO:0042802]; L-cysteine desulfhydrase activity [GO:0080146]; L-cystine L-cysteine-lyase (deaminating) [GO:0044540]; pyridoxal phosphate binding [GO:0030170]; selenocystathionine gamma-lyase activity [GO:0098606]; cellular response to leukemia inhibitory factor [GO:1990830]; cysteine biosynthetic process [GO:0019344]; cysteine biosynthetic process via cystathionine [GO:0019343]; glutathione metabolic process [GO:0006749]; homocysteine metabolic process [GO:0050667]; hydrogen sulfide biosynthetic process [GO:0070814]; lipid metabolic process [GO:0006629]; negative regulation of apoptotic process [GO:0043066]; negative regulation of apoptotic signaling pathway [GO:2001234]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; positive regulation of aortic smooth muscle cell differentiation [GO:1904831]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; protein homotetramerization [GO:0051289]; protein sulfhydration [GO:0044524]; protein-pyridoxal-5-phosphate linkage via peptidyl-N6-pyridoxal phosphate-L-lysine [GO:0018272]; transsulfuration [GO:0019346] |
16327076_The purpose of this study was to identify the seleno-L-methionine (L-SeMet) alpha,gamma-elimination enzyme that catalyzes L-SeMet to generate methylselenol (CH3SeH) 17488480_Caerulein increased the level of cystathionine gamma-Lyase. 17853447_Te CGL antiserum recognized the correct molecular weight protein for mouse retina. 18948540_study shows H2S is physiologically generated by cystathionine gamma-lyase (CSE) & genetic deletion CSE markedly reduces H2S levels in serum & tissue; mice lacking CSE display pronounced hypertension & diminished endothelium-dependent vasorelaxation 19374832_Increase in expression of the enzyme cystathionine gamma-lyase during ischaemia/reperfusion injury may be one of many endogenous mechanisms to limit renal ischaemia/reperfusion injury. 19418582_These results demonstrate that cystathionase is a farnesoid X receptor-regulated gene and provide a new molecular explanation for the pathophysiology of portal hypertension. 19695232_These data reveal that the CSE/H2S pathway is upregulated in the heart in a murine model of coxsakievirus B3 -induced myocarditis. 20051385_Demonstrate an essential role of cystathionine gamma-lyase/H2S system in regulating vascular smooth muscle cell proliferation. 20943958_The effects of CBS AND CSE gene transfer on the homocysteine-mediated inflmmation of mesangial cells induced by hydrogen sulfide and MCP-1 and MIP-2 induction are reported. 21310231_The CSE is critical for cysteine biosynthesis through the transsulfuration pathway and the combination of CSE deficiency and lack of dietary cysteine supply would threaten life sustainability. 21718679_the CSE/H(2)S system plays a critical role in regulating beta-cell functions 22215680_CSE is a critical factor in GSH synthesis and may act to protect the liver and kidney from a variety of conditions that cause ER stress. 22216325_overproduction of MG in CSE(-/-) mice is due to a H(2)S-mediated down-regulation of the PGC-1alpha-FBPase pathway, further suggesting the important role of H(2)S in the regulation of glucose metabolism and MG generation. 22387178_The 6 x Met-fed Cth(-/-) (not 1 x Met-fed Cth(-/-) or 6 x Met-fed wild type) mice displayed acute hepatitis, which was characterized by markedly elevated levels of serum alanine/aspartate aminotransferases and serum/hepatic lipid peroxidation 22870268_Differential effects of cystathionine-gamma-lyase-dependent vasodilatory H2S in periadventitial vasoregulation of rat and mouse aortas. 23074218_Vitamin D up-regulates glucose transporter 4 (GLUT4) translocation and glucose utilization mediated by cystathionine-gamma-lyase (CSE) activation and H2S formation in 3T3L1 adipocytes. 23080424_These data indicate that endogenous CSE/H2 S system contributes to TNF-alpha-caused insulin resistance in 3T3-L1 adipocytes. 23393010_In the setting of heart failure following transverse aortic constriction, CSE knockout mice exhibit significantly greater cardiac dilatation and dysfunction than wild-type mice. 23449534_Cystathionine gamma-lyase protects against renal ischemia/reperfusion by modulating oxidative stress. 23704251_Endogenous H2S is required for healthy placental vasculature, and a decrease in cystathionine gamma-lyase/H2S activity may contribute to the pathogenesis of preeclampsia. 23758073_Congenital deficiency or chemical inhibition of CSE increases thiosulfate levels in the liver and prevents ALF at least in part by augmentation of antioxidant and anti-apoptotic mechanisms. 23838794_Gene silencing the cystathionine gamma-lyase gene inhibited the proinflammatory cytokines and hydrogen sulfide production. 23877364_LPS increased the expressions of cystathionine gamma-lyase and biosynthesis of Hydrogen sulfide... the effects of LPS were not present in TLR4(-/-) mice, indicating the crucial role of TLR4 in LPS-induced expression of CSE and biosynthesis of H2S 23904600_This study showed that CSE was the predominant H2S-generating enzyme, as it was found in 50% of the neurons and in presumptive glial cells in mice. 24008358_These results indicate that endogenous hydrogen sulfide generated by CSE plays a key proinflammatory role via NF-kappaB activation in caerulein-induced pancreatitis. 24357058_Cystathionine gamma-lyase is a potent inducer of calcium resorption. It may act in early stages of osteoclastogenesis. Knockdown suppressed expression of osteoclast differentiation markers. 24670645_demonstration of a major depletion of cystathionine gamma-lyase (CSE), the biosynthetic enzyme for cysteine, in Huntington's disease tissues, which may mediate Huntington's disease pathophysiology 24702258_CSE is present in adrenocortical cells and accounts for H2S generation in adrenal glands. CSE is critical for maintenance of mitochondrial function and glucocorticoid production in adrenal cortex. 24914509_CTH inhibition did not affect the oviductal embryo transport, although delay of early embryo development was observed. 25277804_CSE and hydrogen sulfide modulate adipokine secretion in 3T3-L1 cells under high glucse exposure. 25934094_GPBAR1 plays a role in secondary bile acid induced vasodilation via reglation of cystathionine gamma-lyase. The GPBAR1/CSE pathway might contribute to endothelial dysfunction and hyperdynamic circulation in liver cirrhosis. 26194202_Endogenous cystathionine gamma-lyase/Hydrogen sulfide regulates ischaemic vascular remodelling mediated during hind limb ischaemia through NO-dependent monocyte recruitment 26232299_These data confirm a key role for the H2S-generating enzymes Cbs and Cth in pulmonary vascular development and homeostasis and in lung alveolarization. 26271715_The results of this study suggest that CSE is not critically involved in chronic pain signaling in mice and that sources different from CSE mediate the pain relevant effects of H2S. 26401978_H2S augmented the S-sulfhydration of the rate-limiting gluconeogenic enzymes and PGC-1alpha and increased their activities, which were lower in untreated : To investigate the regulation of hepatic glucose production by cystathionine gamma-lyase KO hepatocytes 26519030_3-Mercaptopyruvate sulphurtransferase and not cystathionine gamma-lyase is the primary regulator of coronary artery hydrogen sulfide production and function. 26692478_Therefore, our data suggest that DNA hypermethylation of CpG rich region in cse promoter might contribute to the decrease of cse transcription and H2S production in macrophages, and thus contribute to atherosclerosis development. 26946260_CSE-H2S increased PPARgamma activity by direct sulfhydration at the C139 site, thereby changing glucose into triglyceride storage in adipocytes. 26949091_lung MPO activity increased following induction of sepsis with CLP while siRNA treatment significantly reduced MPO activity. Liver and lung cytokine and chemokine levels in CLP-induced sepsis reduced following treatment with siRNA. These findings show a crucial pro-inflammatory role for H2S synthesized by CSE in macrophages in sepsis and suggest CSE gene silencing with siRNA as a potential therapeutic approach for this co 27273292_renal expression of CTH and other H2S-producing enzymes was markedly suppressed after IRI, which could be an integrated adaptive response for renal cell protection 27326921_CSE-derived H2S contributes to mouse uterus homeostasis. Sildenafil reduces uterus contractility and its effect involves CSE-derived H2S. 27518439_Cystathionine-Gamma-Lyase Gene Deletion is associated with reduced inflammation and liver damage. 27685807_Study shows maintaining CSE expression is critically important for stress adaptation during posttraumatic acute lung injury and CS-induced COPD, most likely in a gender-dependent manner. 27748832_the findings of this study indicate that a deficiency in 3MST does not significantly affect endotoxemia, while a deficiency in CBS or CSE slightly ameliorates the outcome of LPS-induced endotoxemia in vivo. 27848249_Data show that CSE plays an important role as an inhibitor of inflammation in the liver by producing H2S from L-cysteine. 27865915_this study reveals the molecular basis for the differential expression of Cth in mouse models of essential hypertension under basal and pathophysiological conditions. 27986657_The antiatherosclerotic effect of estrogen is mediated by CSE-generated H2S 28842346_CSE is critical for the progression of kidney fibrosis. 28851593_The results of this study show that deletion of the cystathionine-gamma-lyase gene has no effects on hypoxia-induced changes in TASK K(+) channel activity. 29044685_role of NF-kappaB in the upregulation of cystathionine-gamma-lyase in bladder pain accompanying cystitis 29191638_This study suggests that the CSE/H2S system is involved in the pathogenesis of obesity in mice. 29288927_ZYZ-803 stimulated the expression of cystathionine gamma-lyase (CSE) for H2S generation and the activity of endothelial NO synthase (eNOS) for NO production.Blocking CSE and/or eNOS suppressed ZYZ-803-induced H2S and NO production and cardioprotection. 29317536_Golgi stress response elicited by monensin stimulates CSE by acting via ATF4 with characteristics distinguishable from the endoplasmic reticulum stress response 29730290_Cystathionine gamma lyase-H2S was a dominant H2S generating system in osteoclasts, while cystathionine beta-synthase did not generate H2S. 29844591_Cystathionine-gamma-lyase ameliorates the histone demethylase JMJD3-mediated autoimmune response in rheumatoid arthritis. 29970364_Constitutive S-sulfhydration of human antigen R (on Cys13) by cystathionine gamma lyase -derived H2S prevents its homodimerization and activity, which attenuates the expression of target proteins such as CD62E and cathepsin S, preserving endothelial function and delaying atherogenesis. 30986815_Cystathionine gamma-lyase in mice may have a role in reducing airway reactivity and viral-induced disease in experimental exposure to side-stream tobacco smoke with respiratory syncytial virus infection 31026586_CSE-/- mice were more susceptive to dextran sulfate sodium -induced colitis 31319489_results suggest that elevated homocysteine or other unknown factors in preeclampsia-like Cth(-/-) dams interfere with oxytocin that regulates milk ejection reflex. 31366822_In adipose tissues of diabetic mice, the mRNA and protein expression of MMP14 increased while the mRNA and protein expression of CSE decreased. In CSE knockout mice, the mRNA and protein expression of MMP14 in adipose tissues increased. High glucose increased the expression of MMP14 in adipocytes and visceral adipose tissues through inhibiting the expression of CSE. 31421132_The cystathionine-gamma-lyase system regulates the expression of coproporphyrinogen oxidase and consequent heme synthesis. 31706571_Cystathionine gamma lyase is major key enzyme of hydrogen sulfide generation controlling perilipin 1 sulfhydration. 32900241_CD4(+) T-Cell Endogenous Cystathionine gamma Lyase-Hydrogen Sulfide Attenuates Hypertension by Sulfhydrating Liver Kinase B1 to Promote T Regulatory Cell Differentiation and Proliferation. 33276745_CSE/H2S system alleviates uremic accelerated atherosclerosis by regulating TGF-beta/Smad3 pathway in 5/6 nephrectomy ApoE(-/-) mice. 34688045_High protein diet-induced metabolic changes are transcriptionally regulated via KLF15-dependent and independent pathways. 34779492_The DR1CSE/H2S system inhibits renal fibrosis by downregulating the ERK1/2 signaling pathway in diabetic mice. 34859931_DR1 activation promotes vascular smooth muscle cell apoptosis via up-regulation of CSE/H2 S pathway in diabetic mice. 35586979_Hepatocellular cystathionine gamma lyase/hydrogen sulfide attenuates nonalcoholic fatty liver disease by activating farnesoid X receptor. 36691021_Cystathionine gamma-lyase (Cth) induces efferocytosis in macrophages via ERK1/2 to modulate intestinal barrier repair. |
ENSG00000116761 |
CTH |
43.735668 |
5.5554681489 |
2.473908 |
0.66534357 |
11.829977 |
0.00058284710008537721302041001436577971617225557565689086914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0028638474285485370288140138939070311607792973518371582031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
69.6892402521463 |
33.2908075867133 |
12.5442606066959 |
4.50268034983572 |
| ENSMUSG00000028184 |
99633 |
Adgrl2 |
protein_coding |
Q8JZZ7 |
FUNCTION: Calcium-independent receptor of low affinity for alpha-latrotoxin, an excitatory neurotoxin present in black widow spider venom which triggers massive exocytosis from neurons and neuroendocrine cells. Receptor probably implicated in the regulation of exocytosis. {ECO:0000250|UniProtKB:O88923, ECO:0000269|PubMed:24273166}. |
3D-structure;Alternative splicing;Direct protein sequencing;G-protein coupled receptor;Glycoprotein;Membrane;Phosphoprotein;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
|
glutamatergic synapse [GO:0098978]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; postsynaptic membrane [GO:0045211]; carbohydrate binding [GO:0030246]; G protein-coupled receptor activity [GO:0004930]; PDZ domain binding [GO:0030165]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; brain development [GO:0007420]; cell surface receptor signaling pathway [GO:0007166]; positive regulation of synapse assembly [GO:0051965]; response to bacterium [GO:0009617]; synapse organization [GO:0050808] |
28972101_Lphn2 appears to control synapse numbers in the entorhinal cortex/CA1 region circuit by acting as a domain-specific postsynaptic target-recognition molecule. 31206334_Lphn2 is a unique cell-surface marker of cardiomyogenic cells. 32202499_Latrophilin-2 and latrophilin-3 are redundantly essential for parallel-fiber synapse function in cerebellum. 33798451_Adhesion GPCR Latrophilin-2 Specifies Cardiac Lineage Commitment through CDK5, Src, and P38MAPK. 34083484_Reciprocal repulsions instruct the precise assembly of parallel hippocampal networks. 34818557_Parahippocampal latrophilin-2 (ADGRL2) expression controls topographical presubiculum to entorhinal cortex circuit connectivity. 35356977_The G Protein-Coupled Receptor Latrophilin-2, A Marker for Heart Development, Induces Myocardial Repair After Infarction. |
ENSG00000117114 |
ADGRL2 |
1946.539868 |
2.3671879409 |
1.243174 |
0.14716500 |
67.784189 |
0.00000000000000018240637371928410024565429712629756771919682472021112529247943712107371538877487182617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000053023165734582734878945098163552652967628266329758179864484191057272255420684814453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2843.63776827924 |
290.574293339732 |
1201.27249686659 |
92.0777711043403 |
| ENSMUSG00000028226 |
17389 |
Mmp16 |
protein_coding |
Q9WTR0 |
FUNCTION: Endopeptidase that degrades various components of the extracellular matrix, such as collagen type III and fibronectin. Activates progelatinase A. Involved in the matrix remodeling of blood vessels. It has no effect on type I, II, IV and V collagen. However, upon interaction with CSPG4, it may be involved in degradation and invasion of type I collagen by melanoma cells (By similarity). {ECO:0000250}. |
Calcium;Cell membrane;Cleavage on pair of basic residues;Collagen degradation;Disulfide bond;Glycoprotein;Hydrolase;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix;Zinc;Zymogen |
|
This gene encodes a member of the matrix metalloproteinase family of extracellular matrix-degrading enzymes that are involved in tissue remodeling, wound repair, progression of atherosclerosis and tumor invasion. The encoded preproprotein undergoes proteolytic processing to generate a mature, zinc-dependent endopeptidase enzyme. Mice lacking the encoded protein exhibit retarded growth of the skeleton, especially in the cranium and long bones. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Feb 2016]. |
mmu:17389; |
extracellular matrix [GO:0031012]; plasma membrane [GO:0005886]; metalloaminopeptidase activity [GO:0070006]; metalloendopeptidase activity [GO:0004222]; zinc ion binding [GO:0008270]; bone development [GO:0060348]; chondrocyte proliferation [GO:0035988]; collagen catabolic process [GO:0030574]; craniofacial suture morphogenesis [GO:0097094]; embryonic cranial skeleton morphogenesis [GO:0048701]; endochondral ossification [GO:0001958]; extracellular matrix organization [GO:0030198]; ossification [GO:0001503]; protein processing [GO:0016485]; proteolysis [GO:0006508]; skeletal system development [GO:0001501] |
18022611_Study demonstrates that MT3-MMP, is expressed in mesenchymal tissues of the skeleton and in peri-skeletal soft connective tissue; and MT3-MMP-deficient mice display growth inhibition tied to a decreased viability of mesenchymal cells in skeletal tissues. 21768085_Neuronal knockdown of MT3-MMP resulted in the accumulation of NgR1 at the cell surface and reduced the accumulation of the NgR1 cleavage fragment in medium conditioned by cortical neurons 27227700_miR-155 contributed to the up-regulation of MMP-16, and MMP-16 further degraded aggrecan and collagen type II, leading to the dehydration and degeneration of intervertebral discs |
ENSG00000156103 |
MMP16 |
294.305243 |
0.2388534292 |
-2.065803 |
0.30444222 |
41.265613 |
0.00000000013288618433720088779158868039204479186143892377458541886880993843078613281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000020802901950917204740760243304644599993125098080781754106283187866210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
123.047485567704 |
26.1590258442236 |
515.158965734788 |
79.8803154215094 |
| ENSMUSG00000028480 |
384009 |
Glipr2 |
protein_coding |
Q9CYL5 |
|
Coiled coil;Golgi apparatus;Lipoprotein;Membrane;Myristate;Reference proteome |
|
|
mmu:384009; |
extracellular space [GO:0005615]; Golgi membrane [GO:0000139]; protein homodimerization activity [GO:0042803]; positive regulation of epithelial cell migration [GO:0010634]; positive regulation of epithelial to mesenchymal transition [GO:0010718]; positive regulation of ERK1 and ERK2 cascade [GO:0070374] |
17055234_Increased GLIPR-2 expression in kidney contributes to development of fibrosis by increasing the pool of activated fibroblasts, possibly through the induction of epithelial to mesenchymal transition. 28733476_miR-30e is downregulated in diabetic nephropathy, and the overexpression of miR-30e can inhibit GLIPR-2. 33222586_GLIPR2 is a negative regulator of autophagy and the BECN1-ATG14-containing phosphatidylinositol 3-kinase complex. |
ENSG00000122694 |
GLIPR2 |
613.210870 |
2.2452587078 |
1.166882 |
0.10584406 |
120.649399 |
0.00000000000000000000000000045600051372066430328565739183541287611829230081525410522874659432071697416806455027682432046276517212390899658203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000305982371145073055911091228148263055455806912860446427189370775353758312442575828526969416998326778411865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
839.197106067219 |
61.6233688579849 |
373.764102626221 |
21.3573045729096 |
| ENSMUSG00000028518 |
108079 |
Prkaa2 |
protein_coding |
Q8BRK8 |
FUNCTION: Catalytic subunit of AMP-activated protein kinase (AMPK), an energy sensor protein kinase that plays a key role in regulating cellular energy metabolism (By similarity). In response to reduction of intracellular ATP levels, AMPK activates energy-producing pathways and inhibits energy-consuming processes: inhibits protein, carbohydrate and lipid biosynthesis, as well as cell growth and proliferation (By similarity). AMPK acts via direct phosphorylation of metabolic enzymes, and by longer-term effects via phosphorylation of transcription regulators (By similarity). Regulates lipid synthesis by phosphorylating and inactivating lipid metabolic enzymes such as ACACA, ACACB, GYS1, HMGCR and LIPE; regulates fatty acid and cholesterol synthesis by phosphorylating acetyl-CoA carboxylase (ACACA and ACACB) and hormone-sensitive lipase (LIPE) enzymes, respectively (PubMed:15331533, PubMed:15561936). Promotes lipolysis of lipid droplets by mediating phosphorylation of isoform 1 of CHKA (CHKalpha2) (PubMed:34077757). Regulates insulin-signaling and glycolysis by phosphorylating IRS1, PFKFB2 and PFKFB3 (By similarity). Involved in insulin receptor/INSR internalization (By similarity). AMPK stimulates glucose uptake in muscle by increasing the translocation of the glucose transporter SLC2A4/GLUT4 to the plasma membrane, possibly by mediating phosphorylation of TBC1D4/AS160 (PubMed:16804075, PubMed:16804077). Regulates transcription and chromatin structure by phosphorylating transcription regulators involved in energy metabolism such as CRTC2/TORC2, FOXO3, histone H2B, HDAC5, MEF2C, MLXIPL/ChREBP, EP300, HNF4A, p53/TP53, SREBF1, SREBF2 and PPARGC1A (PubMed:21454484, PubMed:20647423, PubMed:21459323, PubMed:17609368). Acts as a key regulator of glucose homeostasis in liver by phosphorylating CRTC2/TORC2, leading to CRTC2/TORC2 sequestration in the cytoplasm (PubMed:16148943, PubMed:16308421). In response to stress, phosphorylates 'Ser-36' of histone H2B (H2BS36ph), leading to promote transcription (PubMed:20647423). Acts as a key regulator of cell growth and proliferation by phosphorylating TSC2, RPTOR and ATG1/ULK1: in response to nutrient limitation, negatively regulates the mTORC1 complex by phosphorylating RPTOR component of the mTORC1 complex and by phosphorylating and activating TSC2 (PubMed:18439900, PubMed:21258367, PubMed:21205641). In response to nutrient limitation, promotes autophagy by phosphorylating and activating ATG1/ULK1 (PubMed:21258367, PubMed:21205641). In that process also activates WDR45/WIPI4 (By similarity). Phosphorylates CASP6, thereby preventing its autoprocessing and subsequent activation (By similarity). AMPK also acts as a regulator of circadian rhythm by mediating phosphorylation of CRY1, leading to destabilize it (PubMed:19833968). May regulate the Wnt signaling pathway by phosphorylating CTNNB1, leading to stabilize it (PubMed:20361929). Also acts as a regulator of cellular polarity by remodeling the actin cytoskeleton; probably by indirectly activating myosin (By similarity). Also phosphorylates CFTR, EEF2K, KLC1, NOS3 and SLC12A1 (By similarity). Plays an important role in the differential regulation of pro-autophagy (composed of PIK3C3, BECN1, PIK3R4 and UVRAG or ATG14) and non-autophagy (composed of PIK3C3, BECN1 and PIK3R4) complexes, in response to glucose starvation (PubMed:23332761). Can inhibit the non-autophagy complex by phosphorylating PIK3C3 and can activate the pro-autophagy complex by phosphorylating BECN1 (PubMed:23332761). {ECO:0000250|UniProtKB:P54646, ECO:0000250|UniProtKB:Q09137, ECO:0000269|PubMed:15331533, ECO:0000269|PubMed:15561936, ECO:0000269|PubMed:16148943, ECO:0000269|PubMed:16308421, ECO:0000269|PubMed:16804075, ECO:0000269|PubMed:16804077, ECO:0000269|PubMed:17609368, ECO:0000269|PubMed:18439900, ECO:0000269|PubMed:19833968, ECO:0000269|PubMed:20361929, ECO:0000269|PubMed:20647423, ECO:0000269|PubMed:21205641, ECO:0000269|PubMed:21258367, ECO:0000269|PubMed:21454484, ECO:0000269|PubMed:21459323, ECO:0000269|PubMed:23332761, ECO:0000269|PubMed:34077757}. |
ATP-binding;Autophagy;Biological rhythms;Cholesterol biosynthesis;Cholesterol metabolism;Chromatin regulator;Cytoplasm;Fatty acid biosynthesis;Fatty acid metabolism;Kinase;Lipid biosynthesis;Lipid metabolism;Magnesium;Metal-binding;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Steroid biosynthesis;Steroid metabolism;Sterol biosynthesis;Sterol metabolism;Transcription;Transcription regulation;Transferase;Ubl conjugation;Wnt signaling pathway |
|
|
mmu:108079; |
apical plasma membrane [GO:0016324]; axon [GO:0030424]; cytoplasm [GO:0005737]; cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; dendrite [GO:0030425]; Golgi apparatus [GO:0005794]; neuronal cell body [GO:0043025]; nuclear speck [GO:0016607]; nucleoplasm [GO:0005654]; nucleotide-activated protein kinase complex [GO:0031588]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; [acetyl-CoA carboxylase] kinase activity [GO:0050405]; [hydroxymethylglutaryl-CoA reductase (NADPH)] kinase activity [GO:0047322]; AMP-activated protein kinase activity [GO:0004679]; ATP binding [GO:0005524]; chromatin binding [GO:0003682]; histone serine kinase activity [GO:0035174]; metal ion binding [GO:0046872]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; protein serine/threonine/tyrosine kinase activity [GO:0004712]; protein-containing complex binding [GO:0044877]; protein-macromolecule adaptor activity [GO:0030674]; autophagy [GO:0006914]; cellular response to calcium ion [GO:0071277]; cellular response to glucose starvation [GO:0042149]; cellular response to glucose stimulus [GO:0071333]; cellular response to nutrient levels [GO:0031669]; cellular response to oxidative stress [GO:0034599]; cellular response to prostaglandin E stimulus [GO:0071380]; cellular response to xenobiotic stimulus [GO:0071466]; cholesterol biosynthetic process [GO:0006695]; chromatin organization [GO:0006325]; energy homeostasis [GO:0097009]; fatty acid biosynthetic process [GO:0006633]; fatty acid homeostasis [GO:0055089]; glucose homeostasis [GO:0042593]; intracellular signal transduction [GO:0035556]; lipid biosynthetic process [GO:0008610]; lipid droplet disassembly [GO:1905691]; negative regulation of apoptotic process [GO:0043066]; negative regulation of gene expression [GO:0010629]; negative regulation of TOR signaling [GO:0032007]; negative regulation of tubulin deacetylation [GO:1904428]; positive regulation of autophagy [GO:0010508]; positive regulation of glycolytic process [GO:0045821]; positive regulation of peptidyl-lysine acetylation [GO:2000758]; positive regulation of protein localization [GO:1903829]; protein localization to lipid droplet [GO:1990044]; protein phosphorylation [GO:0006468]; regulation of circadian rhythm [GO:0042752]; regulation of gene expression [GO:0010468]; regulation of lipid metabolic process [GO:0019216]; regulation of macroautophagy [GO:0016241]; regulation of microtubule cytoskeleton organization [GO:0070507]; regulation of stress granule assembly [GO:0062028]; response to activity [GO:0014823]; response to caffeine [GO:0031000]; response to muscle activity [GO:0014850]; rhythmic process [GO:0048511]; Wnt signaling pathway [GO:0016055] |
12444247_By sensing the energy status of the muscle cell, AMPK is a critical regulator involved in initiating mitochondrial biogenesis. 12511592_extramuscular AMPKalpha2 catalytic subunit has a role in whole-body insulin action in vivo, probably through modulation of sympathetic nervous activity 12766162_AMP-activated protein kinase alpha2 is necessary for maintaining myocardial energy homeostasis during ischemia 12890675_Transgenic mice overexpressing a constitutively active mutant form of Akt1 displayed decreased phosphorylation of cardiac alpha-AMPK. 14573616_during muscle contraction, the two alpha-isoforms of AMPK seem to substitute for each other in terms of activity 15331533_lack of AMPKalpha2 subunit may be a factor contributing to the development of obesity. 15855317_Activation in the liver reduces blood glucose levels and results in a switch from glucose to fatty acid utilization to supply energy needs. 16186119_Muscle contraction- and hyperosmolarity-induced glucose transport may be regulated by a redundant mechanism in which AMPK alpha2 is one of multiple signaling pathways. 16455782_regulation of hepatic glucose production by leptin and adiponectin, but not insulin, requires hepatic AMPKalpha2 activity 16608880_Data show that, in differentiated mouse myotubes, plectin binds to the regulatory gamma1 subunit of AMP-activated protein kinase (AMPK), the key regulatory enzyme of energy homeostasis. 16687413_hypothalamus and skeletal muscle AMP kinase activity is altered in mice with diet-induced obesity 16877552_AMPKalpha(2) is required for the metabolic response of the heart to no-flow ischemia. The remaining AMPKalpha(1) cannot compensate for the absence of AMPKalpha(2). 16954334_AMPKalpha2 may not be essential for metabolic adaptations of skeletal muscles to exercise training. 17044064_AMPK mediates preconditioning in cardiac cells by regulating the activity and recruitment of sarcolemmal K(ATP) channels without being a part of signaling pathway that regulates mitochondrial membrane potential 17327449_The effect of AMPKalpha2 deletion on mitochodrial function and biogenesis are reported. 17337600_alpha(2)-AMPK is required for a normal glucose uptake and glycogen content, which protects the heart from the development of the ischemic contracture during low flow ischemia reperfusion. 17420279_Leptin stimulates fatty acid oxidation and ppara gene expression in C2C12 myoblasts by changing the subcellular localization of the alpha2 form of AMP-activated protein kinase. 17513699_AMPK is not necessary for exercise training-induced increases in mitochondrial markers, but it is essential for fiber type IIb to IIa/x transformation and increases in hexokinase II protein 17765884_Although AMPK and p38 MAPK are both activated during myocardial ischemia, the activation of p38 MAPK occurs independently of AMPK. 17901380_AMPK activation is detrimental in a model of focal stroke. The AMPK catalytic isoform alpha-2 contributes to the deleterious effects of AMPK activation. AMPK inhibition leads to neuroprotection even when these agents are administered poststroke. 17971451_Hfe(-/-) mice demonstrate increased adiponectin levels and activation of AMP-dependent kinase 18088373_These results demonstrate that alpha-AMPK phosphorylation is dependent on method of killing and tissue preparation and that alpha-AMPK phosphorylation can increase in a manner that does not reflect biological alterations. 18163380_These results demonstrate that AMPK and PI3-K/Akt have an additive effect on oxidative stress-mediated GLUT4 translocation. 18164589_Elude to a role of PKD in contraction-induced GLUT4 translocation which does not involve AMPK-alpha2. 18387000_Data demonstrate that inhibition of AMPK resulting from a hypomorphic mutation that decreases LKB1 expression does not lead to tumorigenesis on its own, but markedly accelerates tumour development in PTEN(+/-) mice. 18653710_PPARgamma, even in the absence of exogenous activators, is required for normal rates of fatty acid uptake in oxidative skeletal muscle via mechanisms independent of AMPK and fatty acid transport protein 1. 18701654_Report stimulation of glucose transport in response to activation of distinct AMPK signaling pathways. 18728234_The lack of skeletal muscle AMPK alpha2 activity exacerbates the development of glucose intolerance and insulin resistance caused by high-fat feeding. 18729805_Regulation of Akt, p53, adenosine monophosphate-activated protein kinase (AMPK) phosphorylation, and Klotho may play a role, at least in part, in IGF-1 deficiency-induced 'desensitization' of cardiac aging. 18812163_This work shows that AMPKalpha2 plays a role of brake for the development of cardiac hypertrophy. 18812461_AMPK activation is essential for both AICAR and submaximal contraction-induced glucose transport in skeletal muscle. 18818284_Basal and A-769662-mediated IL-6 release provide evidence for a role of AMPK in the regulation of IL-6 release from oxidative skeletal muscle. 18838626_AMPKalpha2 exerts a cardiac protective effect against pressure-overload-induced ventricular hypertrophy and dysfunction. 18845612_AMPK is not essential for the regulation (phosphorylation of acetyl-CoA carboxylase 2) of fatty acid oxidation by AICAR or muscle contraction. 18955383_AMPK regulates basal VEGF expression and capillarization in skeletal muscle, but is not necessary for exercise-induced angiogenesis. 19131647_AMP-activated protein kinase functionally phosphorylates endothelial nitric oxide synthase Ser633. 19188248_A Ca(2+)-calmodulin-eEF2K-eEF2 signalling cascade, but not AMPK, contributes to the suppression of skeletal muscle protein synthesis during contractions. 19188503_mice with mutant AMPKalpha2 subunit subjected to MI/R endured greater cardiac injury (larger infarct size, more apoptosis, and poorer cardiac function) likely as a result of increased oxidative stress 19224872_Data suggest that anti-obesity effects of L-4F result in decreased visceral fat and increased adiponectin, pAMPK, pAKT, and improved insulin sensitivity, and that upregulation of HO-1 by L-4F produces adipose remodeling. 19486896_AMPKalpha1(-/-) mice exhibited severe EAE disease with profound infiltration of mononuclear cells compared to wild type mice however, AMPKalpha2 is not involved in enhancing the severity of the disease. 19502419_IL-6 activates AMPK in skeletal muscle by increasing the concentration of cAMP and, secondarily, the AMP:ATP ratio 19520133_The inhibitory effect of sulfated glucosamine on adipocyte differentiation might be mediated through the up-regulation of AMPK pathway. 19531644_Genetic evidence to support an important role for AMPK in regulating TBC1D1 in response to physiological stimuli. 19654283_AMPKalpha2 signaling is not essential in regulating glucose uptake in mouse skeletal muscle during treadmill exercise 20036235_These results indicate that p27(NCDK) is a sensitive marker for both cell stress and proliferation over and above p27 and is regulated by Akt/PKB and AMPK pathways. 20124121_AMPK functions as a physiological suppressor of endoplasmic reticulum stress by maintaining SERCA activity and intracellular Ca2+ homeostasis. 20126541_Results demonstrate that AMPK is a modulator of LTP. 20167927_AMPKalpha2 functions as a physiological suppressor of NAD(P)H oxidase, ROS production and 26S proteasome activity in endothelial cells. 20221584_Inhibition of AMPK activity (by ablation of alpha 1 and 2 subunit) within the beta cell is necessary, but not sufficient for stimulation of insulin secretion by glucose to occur. 20399918_These data support a role for AMPK signalling in skeletal physiology. 20422711_Data demonstrate that benzo(a)pyrene activates PRKAA1/2 and causes ID2 protein loss in trophoblast stem cells in a time- and dose-dependent manner. 20460585_Results uncover the role of AMPK in the maintenance of muscle cell size control and highlight the crosstalk between AMPK and mTOR/p70S6K signaling pathways coordinating a metabolic checkpoint on cell growth. 20465544_AMPK alpha2 activity is necessary to maintain normal pancreatic beta-cell glucose sensing, possibly by maintaining high beta-cell levels of UCP2. 20647423_AMPK activates transcription by direct association with chromatin and phosphorylation of histone H2B at ser36; results place AMPK-dependent H2B Ser36 phosphorylation in direct transcriptional and chromatin regulatory pathway leading to stress adaptation 20660302_AMPK regulates glucocorticoid actions on carbohydrate metabolism by targeting the glucocorticoid receptor (GR) and modifying transcription of glucocorticoid-responsive genes in a tissue- and promoter-specific fashion. 20830737_Data suggest AMPK is involved in maturation and the completion of meiosis. Immunolocalization of AMPK alpha2 subunit stained only diffusely throughout the oocyte. 20837646_Catalytic alpha2 AMP-activated protein kinase (AMPK) subunit is the main donor of AMPK activity regulating TBC1D1 Ser237 phosphorylation in skeletal muscle. 21233771_Exercise increases the nuclear MEF2A content and binding of MEF2A to their binding sites on the Glut4 gene by an AMPKalpha2-dependent mechanism. 21402702_both AMPKalpha isoforms suppress MMP-9 expression and that both the activity and presence of AMPKalpha contribute to its function as a regulator of MMP-9 expression by inhibiting the NF-kappaB pathway. 21446786_Hypothermia exerts its protective effect in part by inhibiting AMPK activation in experimental focal stroke. 21483791_Data found that both alpha1-/- and alpha2-/- mice are able to maintain a circadian rhythm of activity in dark-dark (DD) cycle, but alpha1-/- mice have a shorter circadian period whereas alpha2-/- mice showed a tendency toward a slightly longer circadian period. 21506122_Indicate that activation of AMP-activated protein kinase is involved in vincristine-induced cell apoptosis in B16 melanoma cells. 21551008_Suggest that AMPKalpha(2) is involved in the regulation of substrate uptake in a time-dependent manner in contracting muscle but is not necessary for regulation of fatty acid uptake and oxidation during caffeine treatment. 21597006_overexpression of UCP2 normalized angiogenic tube formation in aortic endothelial cells from either AMPKalpha1(-/-) or AMPKalpha2(-/-) mice 21673972_IKK is a direct substrate of AMPKalpha2 21799304_AMPK-alpha1, but not AMPK-alpha2, regulates muscle cell size through the control of mTORC1 signaling. 21825219_AMP activated protein kinase-alpha2 regulates expression of estrogen-related receptor-alpha, a metabolic transcription factor related to heart failure development. 21993711_Data suggest that stimulation of AMPK (Prkaa1 and Prkaa2) in embryos during diabetic pregnancy mediates effect of hyperglycemia-induced (or anoxia/antimycin A-induced) oxidative stress on expression of Pax3 and thereby may cause neural tube defects. 22129093_Suggest that GGEx18 improves obesity through skeletal muscle AMPK and AMPK-stimulated expression of PPARalpha and its target enzymes for fatty acid oxidation. 22308162_in insulin-resistant conditions low AMPKalpha2 activity impacts the regulation of skeletal muscle FA metabolism via changes in SIRT1 activity, PTP1B expression and Akt phosphorylation and the expression of adipose tissue pro-inflammatory markers. 22396207_These results suggest that AMPK may be modulated by calorie restriction (CR) in a ubiquitylation-dependent manner and acts as a chief dictator for the insulin-sensitizing effects of CR in skeletal muscle. 22574218_AMPK functions as a physiological suppressor of 26S proteasomes through OGT 22727014_p70S6 kinase forms a complex with the alpha-2 catalytic subunit of AMPK, resulting in phosphorylation on serine(491). 22763120_AMPK activation in blood-brain barrier-derived endothelial cells directs the trafficking of GLUT1 intracellular pools to the plasma membrane, thereby increasing endothelial sugar transport capacity. 22932492_Both AMPK and ULK1 regulate localization of a critical component of the phagophore, ATG9 23024365_Leptin activates liver AMPK alpha1 and alpha2 through sympathetic nervous system and alpha1-adrenergic receptor. 23224579_A functionally active AMPKalpha2 subunit is required for insulin-stimulated muscle glycogen synthesis. 23288166_Endoplasmic reticulum stress increases vascular smooth muscle contractility resulting in high blood pressure, and AMP-activated protein kinase activation mitigates high blood pressure through the suppression of ER stress in vivo. 23293026_WWP1 down-regulates AMPKalpha2 under high glucose culture conditions via the ubiquitin-proteasome pathway 23316058_Our data identify the microtubule cytoskeleton as a sensitive target of AMPK activity, and the data suggest a novel role for AMPK in limiting accumulation and densification of microtubules that occurs in response to hypertrophic stress. 23349504_Data from knockout mice suggest that LKB1 (liver kinase B1) acts independent of AMPKa2 in regulation of oxidation of free fatty acids in skeletal muscle during physical activity/exercise. Knockdown of LKB1 leads to decrease in muscle AMPKa2. 23382195_AMPKalpha suppression or knockdown produces the opposite effects. The results demonstrate an anti-infamatory pathway linking AMPK, PARP-1, and Bcl-6 in endothelial cells 23466593_AMPKalpha2 controls cardiac p70S6K under normoxia and regulates eEF-2 but not the mTOR-p70S6K pathway during ischemia. 23671593_The impact of ageing and high fat diet on insulin action is not worsened in mice lacking functional alpha2AMPK in skeletal muscle. 23688013_Data (including data from Prkaa2 knockout mice) suggest leptin is involved in regulation of cardiomyocyte contraction (or dysfunction) via Prkaa2 phosphorylation/activation and autophagy. 23863712_AMPK is the first identified physiological substrate for CHIP chaperone activity, establishing a link between cardiac proteolytic and metabolic pathways. 24043309_Myogenin expression and myogenesis were nearly abolished in the absence of both AMPKalpha1 and AMPKalpha2, while enhanced AMPK activity promoted myogenesis and myotube formation. 24398673_AMPKalpha2 significantly contributed to stabilization and activation of E2F1 by doxorubicin, forming a positive signal amplification loop. 24403081_AICAR reduced hepatic energy charge by approximately 20% in alpha1alpha2(lox/lox), which was exacerbated by approximately 2-fold in alpha1alpha2(lox/lox) + Albcre. 24449471_Dietary wolfberry elevated the xanthophyll concentrations and enhanced expression of BCO2 and heat shock protein 60, activated AMPKalpha2, potentiated mitophagy and mitochondrial biogenesis and enhanced lipid oxidation and secretion in the liver of mice. 24515110_AMPK alpha2 might represent an important therapeutic target for colon cancer metastasis-induced liver injury 24686086_AMPKalpha2 translocates into the nucleus via phosphorylation, AMPKalpha2 interacts with and phosphorylates hnRNP H in the nucleus, and such a protein-protein interaction modulates metformin-mediated glucose uptake. 24732703_activation of AMPKalpha2 seems crucial for maintaining skeletal muscle function, integrity and the ability to compensate chronic metabolic stress induced by SM mitochondrial uncoupling 25003940_results indicate that a reduction in AMPK levels causes alterations in cell morphology, growth rate, and nutrient transport, thus identifying an important role for AMPK in the regulation of placental trophoblast differentiation. 25103967_Low AMPK-alpha2 signaling disrupts, in part, the exercise training-induced adaptations. 25448287_Suggest that E2 treatment ameliorates estrogen deficiency-induced changes in cardiac contractile function possibly through an AMPK-dependent mechanism. 25637528_AMPKalpha2 ablation in muscle did not exacerbate high fat diet induce obesity in mice. On the contrary, it improved animal glucose tolerance and insulin sensitivity, with reduced triglyceride content in skeletal muscle. 25691571_provide evidence that high CO2 activates skeletal muscle atrophy via AMPKalpha2-FoxO3a-MuRF1, which is of biological and potentially clinical significance in patients with lung diseases and hypercapnia 25799226_AMPKalpha2 is an essential mediator of nicotine-induced whole-body IR in spite of reductions in adiposity 25923694_The increase in nuclear PPARalpha protein by four-week exercise training under the intermittent hypoxia was dependent on AMPKalpha2 activation. 25965390_the current data suggests that activation of AMPK signaling is sufficient but not required for exercise-induced accumulation in mitochondrial FAT/CD36. 26034200_AMPK directly relaxes vascular smooth muscle cell by a decrease of [Ca(2+)]i. This is achieved by calcium sequestration via SERCA activation, as well as activation of BKCa channels. 26108355_AMPKalpha signalling suppresses EMT and secretion of chemokines in renal tubular epithelia through interaction with CK2beta to attenuate renal injury. 26252187_In conclusion, our findings reveal a new functional role of activating AMPK in the CNS to attenuate inflammatory responses and acute lung injury in sepsis. 26268732_Data (including data from knockout mice) suggest expression levels/phosphorylation of Ampk-alpha1/Ampk-alpha2 are up-regulated in intestinal mucosa by ingestion of alpha-linolenic acid (which inhibits high-fat diet-induced hypertriglyceridemia). 26359931_AMPKalpha2 is involved in post-transcriptional and not transcriptional regulation of PDK4 in muscle. 26419588_Data (including data from studies in knockout mice) suggest that AMPKa1 and AMPKa2 are key in gene expression regulation in skeletal muscle in acute exercise studies but not in longer exercise training adaptation studies. 26596312_AMPKalpha2 deletion and high-fat feeding adverse effects are not additive on heart function and myocardial ischemic tolerance. 26718972_AMPKalpha2 isoform plays a fundamental role in anti-oxidant stress and anti-senescence 26797128_PKD1 phosphorylates AMPKalpha2 at Ser485/491, thus diminishing AMPK activity. 27010458_Deletion of the AMPK alpha-subunits improves oral but not intraperitoneal glucose tolerance. 27052588_Data show that metformin may protect against ciaplatin induced acute kidney injury (AKI) through stimulating AMPKalpha activation. 27136709_Collectively, our data support the notion that the activation of AMPKalpha2 contributes to the atrophic effects of denervation. 27306083_Findings show that decreased AMPK activity in muscle leads to decreased voluntary activity which is not due to secondary abnormalities in dopamine levels in the ventral striatum or sensitivity to cocaine. Thus, decreased voluntary activity in AMPK muscle deficient mice is most likely unrelated to regulation of brain dopamine content and metabolism. 27416746_These studies reveal a novel mechanism in which CYP2J2 and epoxyeicosatrienoic acids enhanced Akt1 nuclear translocation through interaction with AMPKalpha2beta2gamma1 and protect against cardiac hypertrophy. 27427333_The rs2746342 polymorphism is significantly associated with susceptibility to type 2 diabetes mellitus (T2DM) and seems to interact with the rs2143754 polymorphism in the modulation of fasting plasma glucose (FPG) in the Han Chinese population. 27439892_AMPKalpha2 deletion induces vascular smooth muscle cell phenotype switching and promotes features of atherosclerotic plaque. 27600021_Study indicate that the alpha 2 and alpha 1 subunits of AMPK have several functional differences, with alpha 2 conferring stronger osteogenic potential and a weaker ability to induce osteoblasts-associated osteoclastogenesis in MC3T3-E1 cells as well as conferring a lower adipogenic potential to 3T3-L1 cells. 27758765_AMPK phosphorylation of cortactin followed by SIRT1 deacetylation modulates the interaction of cortactin and cortical-actin in response to shear stress. Functionally, this AMPK/SIRT1 coregulated cortactin-F-actin dynamics is required for endothelial nitric oxide synthase subcellular translocation/activation and is atheroprotective. 27777247_The activation of AMPKalpha2 in neutrophils is a decisive event in the initiation of vascular repair after ischemia. 27879319_Loss of AMPKalpha2 is associated with impaired control of vesicular stomatitis virus infection. 27928820_AMPK-mediated HSPB1 expression enhanced insulin sensitivity in the skeletal muscle. 28085543_PRKAA deletion promoted mitochondrial fragmentation in vascular endothelial cells by inhibiting the autophagy-dependent degradation of DNM1L. 28179232_we used the Cre-loxP system to knock down AMPKalpha2 expression in renal epithelial cells. Combining this approach with the systemic deletion of AMPKalpha1 we achieved reduced renal AMPK activity, accompanied by a shift to a moderate water- and salt-wasting phenotype. Thus we confirm the physiologically relevant role of AMPK in the kidney. 28179583_High AMPKalpha2 phosphorylation is associated with abdominal aortic aneurysm. 28183804_TNF-alpha treatment of colonic rho(0) cells augmented IL-8 expression by 9-fold (P |
ENSG00000162409 |
PRKAA2 |
151.235310 |
3.5228607769 |
1.816747 |
0.23625305 |
58.394580 |
0.00000000000002144811231611664772837138932932007036989024463535802311753286630846560001373291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000005185676933318869203511894511674798301627611896158498439035611227154731750488281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
237.207249308693 |
39.0184642915019 |
67.3336996069884 |
8.74130050315748 |
| ENSMUSG00000028571 |
230459 |
Cyp2j13 |
protein_coding |
B1AWM5 |
Human_homologues FUNCTION: A cytochrome P450 monooxygenase involved in the metabolism of polyunsaturated fatty acids (PUFA) in the cardiovascular system (PubMed:8631948, PubMed:19965576). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (NADPH--hemoprotein reductase) (PubMed:8631948, PubMed:19965576). Catalyzes the epoxidation of double bonds of PUFA (PubMed:8631948, PubMed:19965576). Converts arachidonic acid to four regioisomeric epoxyeicosatrienoic acids (EpETrE), likely playing a major role in the epoxidation of endogenous cardiac arachidonic acid pools (PubMed:8631948). In endothelial cells, participates in eicosanoids metabolism by converting hydroperoxide species into hydroxy epoxy metabolites. In combination with 15-lipoxygenase metabolizes arachidonic acid and converts hydroperoxyicosatetraenoates (HpETEs) into hydroxy epoxy eicosatrienoates (HEETs), which are precursors of vasodilatory trihydroxyicosatrienoic acids (THETAs). This hydroperoxide isomerase activity is NADPH- and O2-independent (PubMed:19737933). Catalyzes the monooxygenation of a various xenobiotics, such as danazol, amiodarone, terfenadine, astemizole, thioridazine, tamoxifen, cyclosporin A and nabumetone (PubMed:19923256). Catalyzes hydroxylation of the anthelmintics albendazole and fenbendazole (PubMed:23959307). Catalyzes the sulfoxidation of fenbedazole (PubMed:19923256). {ECO:0000269|PubMed:19737933, ECO:0000269|PubMed:19923256, ECO:0000269|PubMed:19965576, ECO:0000269|PubMed:23959307, ECO:0000269|PubMed:8631948}. |
Heme;Iron;Membrane;Metal-binding;Reference proteome;Transmembrane;Transmembrane helix |
Human_homologues PATHWAY: Lipid metabolism; arachidonate metabolism. {ECO:0000269|PubMed:19737933, ECO:0000269|PubMed:19965576, ECO:0000269|PubMed:8631948}. |
|
mmu:230459; |
cytoplasm [GO:0005737]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; arachidonic acid 11,12-epoxygenase activity [GO:0008405]; arachidonic acid 14,15-epoxygenase activity [GO:0008404]; arachidonic acid epoxygenase activity [GO:0008392]; heme binding [GO:0020037]; iron ion binding [GO:0005506]; isomerase activity [GO:0016853]; linoleic acid epoxygenase activity [GO:0071614]; oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen, reduced flavin or flavoprotein as one donor, and incorporation of one atom of oxygen [GO:0016712]; steroid hydroxylase activity [GO:0008395]; epoxygenase P450 pathway [GO:0019373]; icosanoid metabolic process [GO:0006690]; linoleic acid metabolic process [GO:0043651]; organic acid metabolic process [GO:0006082]; xenobiotic metabolic process [GO:0006805] |
Human_homologues 12386130_expression levels of the immunoreactive protein with the CYP2J2 antibody in the small intestine were well correlated with the activities of the astemizole O-demethylation 12737630_role of activator protein-1 in the down-regulation of the human CYP2J2 gene in hypoxia 14575523_Observational study of genotype prevalence and gene-disease association. (HuGE Navigator) 14575523_The CYP2J2*6 allele is rare in the Caucasian population, and no association is inferred between this allelic variant and type 1 or type 2 diabetic complications. 15256482_Transgenic mice overexpressing human CYP2J2 have improved postischemic recovery of left ventricular function. 15361551_Overexpressoin of CYP2J2 in transgenic mice increases l-type calcium channles in cardiomyocytes. 15466638_Observational study of gene-disease association. (HuGE Navigator) 15466638_a functionally relevant polymorphism of the CYP2J2 gene is independently associated with an increased risk of coronary artery disease 15861034_Observational study of genotype prevalence. (HuGE Navigator) 15861034_characterization of novel CYP2J2 variants that cause loss of enzyme catalytic activity 15864120_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 15864120_there is an association between CYP2J2*7 (but not CYP2C8*3) genotype and hypertension in Caucasian males and Caucasians without a family history of hypertension 16008525_The c-Jun-responsive module between -122 and -50 in the CYP2J2 proximal promoter contains an atypical AP-1 element at -105/-95 that has a major role in c-Jun transactivation and acts in conjunction with the -56/-63 element to regulate expression. 16182271_Observational study of genotype prevalence. (HuGE Navigator) 16202848_No association was found between any of the variant alleles and hypertension. 16202848_Observational study of gene-disease association. (HuGE Navigator) 16842392_Observational study of gene-disease association and pharmacogenomic / toxicogenomic. (HuGE Navigator) 16957870_comprehensive analysis of the distribution of sEH, CYP2C8, 2C9 and 2J2 in human neoplastic tissues using tissue micro-arrays 17126841_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17126841_a CYP2J2*7 polymorphism may have a role in premature myocardial infarct 17162467_large inter-individual variation of the CYP2J2 suggests that this enzyme plays a significant role in the metabolism of xenobiotics 17286575_CYP2J2 gene is a susceptibility factor for essential hypertension, especially in females, and influences individual systolic blood pressure in the Chinese Han population 17286575_Observational study of gene-disease association. (HuGE Navigator) 17327508_Observational study of gene-disease association. (HuGE Navigator) 17429317_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 17429317_The G-50T polymorphism in CYP2J2 may be an important risk factor for the development of coronary heart disease events in African-Americans 17470359_These new selective inhibitors should be interesting tools to study the biological roles of CYP2J2. 17475630_Observational study of gene-disease association. (HuGE Navigator) 17475630_Results indicate the CYP2J2 gene might be considered as a novel candidate gene for common susceptibility to asthma and highlight the importance of the P-450 epoxygenase pathway of metabolism of arachidonic acid in the pathogenesis of the disease. 17638876_CYP2J2 has a role in cancer metastasis 17705402_The structural features that have been found to be important for recognition of substrates or inhibitors by CYP2J2 were also interpreted on the basis of CYP2J2-substrate interactions in this model. 17979511_Genes encoding this enzyme modify cardiovascular disease risk at the population level in humans, providing support that modulation of this pathway may represent a novel approach to prevention. [REVIEW] 18004755_Flexible docking approaches were employed to dock 4 ligands into the active site of P450 2J2 to probe ligand-binding modes. Active site architecture and certain key residues responsible for substrate specificity were identified on the enzyme. 18216721_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18219097_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 18219097_a CYP2J2*7 allele of the CYP2J2 gene is clearly associated with an increased risk of essential hypertension. 18303964_Clinical trial of gene-disease association and pharmacogenomic / toxicogenomic. (HuGE Navigator) 18496133_Observational study of gene-disease association. (HuGE Navigator) 18496133_Two intronic CYP2J2 tag-single nucleotide polymorphisms, rs10889160 and rs11572325 were associated with an increased risk of myocardial infarction. No evidence of an association was found between variation in CYP2J2 and stroke. 18675280_These results suggest that expression of CYP2J2 was up-regulated when human monocytes differentiated into macrophages and that human monocytic cells and macrophages have a pathway to metabolize arachidonic acid using CYP epoxygenases. 18729130_Genotype distribution of CYP2J2*7 polymorphisms was investigated using polymerase chain reaction and restriction fragment length polymorphism assay in Japanese, Mongolian and Ovambo populations and the findings compared with other populations. 18729130_Observational study of genotype prevalence. (HuGE Navigator) 18769365_Observational study of gene-disease association. (HuGE Navigator) 18769365_This study examined the association between calcineurin inhibitors-induced nephrotoxicity in liver transplant patients and CYP2C8 and CYP2J2 polymorphisms. 19105833_Observational study of gene-disease association. (HuGE Navigator) 19105833_among patients with high cardiovascular risk profile, carriers of the G-50T-promoter polymorphism have significantly more myocardial infarctions 19290787_Observational study of genotype prevalence. (HuGE Navigator) 19420105_Observational study of gene-disease association. (HuGE Navigator) 19448409_CYP epoxygenases efficiently promote the proliferation of tumor cells, which may be related with the activation of EGFR, ERK1/2 and PI3K/Akt signaling pathways. 19550113_Data show that CYP2J2 is highly and selectively expressed in human tumor tissues and cell lines and may be a novel biomarker of human tumors. 19576320_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 19578796_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 19615687_Observational study of gene-disease association. (HuGE Navigator) 19823578_CYP2J2 activates the nuclear receptor PPARalpha in vitro and in vivo 19851119_Observational study of gene-disease association. (HuGE Navigator) 19851119_data do not support a major role for the CYP2J2 -50G>T variant in determining blood pressure level and incident ischemic events 19913121_Observational study of gene-disease association. (HuGE Navigator) 19923256_eight novel substrates for CP2J were identified that vary in size and overall topology; data suggest that CYP2J2 may be an unrecognized participant in first-pass metabolism, but its contribution is minor relative to that of CYP3A4 20140850_Age of onset, family history, and obesity may modify typ3 2 diabetes polymorphism in younger onset typ32 diabetees. 20173083_Observational study of genotype prevalence. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20480811_Observational study of gene-disease association. (HuGE Navigator) 20480811_The data indicate that epoxygenase 2J2 gene polymorphism is not a risk factor of acute coronary syndrome in Ukrainian population. 20597138_Haplotypes of CYP2J2 are associated with myocardial infarction in a Chinese Han population. 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 21030485_CYP2J2 overexpression in cultured human-derived malignant hematologic cell lines markedly accelerated proliferation and attenuated apoptosis 21059750_These data demonstrate that potentiation of the CYP epoxygenase pathway by either increased endothelial EET biosynthesis or globally decreased EET hydrolysis attenuates NF-kappaB-dependent vascular inflammatory responses in vivo. 21642892_This case-control study as well as meta-analysis suggested no association between CYP2J2 G-50T and EPHX2 R287Q and the risk of developing coronary artery disease 22028915_monocytes and macrophages express the epoxygenases CYP2J2 and CYP2C8 22232929_No association has been found between the CYP2J2 (rs890293, -76G > T) or CYP2S1 (rs34971233, 13106C > T, P466L and rs338583, 13255A > G) gene polymorphisms and respiratory diseases. 22731644_This study provides evidence of potential association of CYP2J2*7 polymorphism with hypertension in the Saudi population with or without diabetes and obesity. 22761738_High CYP2J2 expression is associated with cancer. 23155181_uteroplacental CYP2J2 overexpression contributes to preeclampsia. 23474289_Established and emerging roles of CYP2J2 in the biology of vascular endothelial cells. [Review] 23647230_N404Y mutation could reposition the side-chain of Leu(378), making it no longer form a hydrogen bond with the carboxyl group of arachidonic acid. 23661295_Compared the effects of N-terminal modifications on the properties of CYP2J2 with respect to the activity of the protein in model lipid bilayers using Nanodiscs. 23684773_The aim of the present study was to assess the association between the human CYP2J2 gene polymorphism and coronary artery disease (CAD) in a Han and Uygur population of China. 23696562_Data indicate that cardiac-specific overexpression of arachidonate epoxygenase (CYP2J2) significantly protects against diabetic cardiomyopathy, which may be due to improved cardiac insulin resistance, glucose uptake, and reversal of cardiac hypertrophy. 23959307_CYP2J2 and CYP2C19 are the major liver microsomal enzymes responsible for biotransformation of albendazole and fenbendazole to produce active anthelmintics in vivo. 24021950_CYP2J2 activity in the cell line was inhibited by danazol. 24023684_CYP2J2 overexpression only moderately reduced transverse aortic constriction-induced cardiac hypertrophy and did not affect the hypertrophic response to beta-adrenergic stimulation. 24058654_CYP2J2 mediates bacterial phagocytosis in macrophages, and implicates a defect in the CYP2J2 pathway may regulate bacterial clearance in Crohn's disease. 24278032_These findings demonstrate that Cyp2j gene products regulate the pulmonary vascular response to hypoxia 25204827_findings can give a structural mechanism for the mutation-induced dysfunction in the CYP2J2-mediated arachidonic acid metabolism. 25388680_Patients with CYP2C8*2C and EPHX2 404del variants had worse long-term outcomes while those with EPHX2 287Gln, CYP2J2*7, and CYP2C9 g.816G variants had favorable outcomes. 25389363_Data suggest that up-regulation of CYP2J2 activity can attenuate type 2 diabetes and improve insulin resistance via down-regulation of NFkappaB and MAPK signaling pathways and up-regulation of PPARgamma. 25406731_The present study shows that elevated EET levels in BC tissues are associated with upregulation of CYP2C8, 2C9, and 2J2, and downregulation of sEH, and are also associated with aggressive cell behavior in BC patients. 25560582_Our results suggest that individuals with any conventional risk factor for MI along with CYP2J2*7 variant allele may be predisposed to risk of myocardial infarct in South Indian population 25601761_CYP2J2 expression showed significant upregulation (PT polymorphism is associated with increased risk of cerebral small vessel disease in Han Chinese. 29200270_demonstrate that doxorubicin (DOX) and 7-de-aDOX inhibit CYP2J2-mediated AA metabolism and 7-de-aDOX binds close to the active site to alter the ratio of cardioprotective epoxyeicosatrienoic acids. 29343610_Cell survival decreases with ROS exposure when CYP2J2 is chemically inhibited. 29580941_Increased epoxyeicosatrienoic acids may be part of a protective mechanism in human ulcerative colitis, with increased CYP2J2 and reduced soluble epoxide hydrolase expression. 29629756_these data can aid the design of other analogues that have reduced cardiotoxicity, which do not modulate CYP2J2's arachidonic acid metabolism. 29908721_In the villous trophoblast, CYP2C8 was the most abundant protein. Its expression is higher than the CYP2C9 and CYP2J2 in the cytotrophoblast in the embryonic stage of development and remains higher in syncytiotrophoblast of term placenta. 30012976_Data show the importance of the formation of a hydrogen bond between cytochrome P450 2J2 arginine 117 (R117) and the keto group of the substrates. 30019995_There were 15 genetic variants identified in CYP2J2 genes in three Chinese populations: Han, Tibetan and Uighur. 30219518_Our findings demonstrate that CYP2J2 improves cardiac function by increasing the concentration of circulating EETs, and boosting angiogenesis via the Jagged1/Notch1 signaling pathway in MI-induced heart failure. 30518987_In conclusion, the rs2280275 and rs11572325 SNPs of CYP2J2 may be considered novel genetic markers of hypertension, at least in Russian women 30861250_Studies report on the role of the surrounding lipid environment and macromolecular crowding in modulating enzymatic function of CYP2J2-cytochrome P450 reductase membrane protein system. 31000184_A functionally relevant polymorphism of the CYP2J2 gene was independently associated with an increased risk of CAD. 31622444_The CYP2J2*7 SNP in the donor was associated with elevated risk of delayed graft function. 31902555_CYP2J2 50G>T (rs890293) polymorphism was associated with an increased risk for psoriasis in the Turkish population 32210298_CYP2J2 Modulates Diverse Transcriptional Programs in Adult Human Cardiomyocytes. 32224017_Influence of the CYP2J2 Gene Polymorphisms on Chronic Obstructive Pulmonary Disease Risk in the Chinese Han Population. 33312341_6,8-Diprenylorobol Induces Apoptosis in Human Hepatocellular Carcinoma Cells via Activation of FOXO3 and Inhibition of CYP2J2. 33335233_Molecular determinant of substrate binding and specificity of cytochrome P450 2J2. 34258261_CYP2J2 Is a Diagnostic and Prognostic Biomarker Associated with Immune Infiltration in Kidney Renal Clear Cell Carcinoma. 34774051_CYP2J2 and EETs protect against pulmonary arterial hypertension with lung ischemia-reperfusion injury in vivo and in vitro. 36269280_Endothelial CYP2J2 overexpression restores the BRB via METTL3-mediated ANXA1 upregulation. 36587509_CYP2C8 and CYP2J2 gene variations increase the risk of hypertensive intracerebral hemorrhage. |
ENSG00000134716 |
CYP2J2 |
10.487055 |
28.9941363469 |
4.857689 |
1.17061650 |
19.608865 |
0.00000950272664387411160205956783819658539869124069809913635253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000707110562967711128896930450338231821660883724689483642578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.7224504745859 |
17.8177276389421 |
0.680222036573591 |
0.539568113532719 |
| ENSMUSG00000028613 |
16975 |
Lrp8 |
protein_coding |
Q924X6 |
FUNCTION: Cell surface receptor for Reelin (RELN) and apolipoprotein E (apoE)-containing ligands. LRP8 participates in transmitting the extracellular Reelin signal to intracellular signaling processes, by binding to DAB1 on its cytoplasmic tail. Reelin acts via both the VLDL receptor (VLDLR) and LRP8 to regulate DAB1 tyrosine phosphorylation and microtubule function in neurons. LRP8 has higher affinity for Reelin than VLDLR. LRP8 is thus a key component of the Reelin pathway which governs neuronal layering of the forebrain during embryonic brain development. Binds the endoplasmic reticulum resident receptor-associated protein (RAP). Binds dimers of beta 2-glycoprotein I and may be involved in the suppression of platelet aggregation in the vasculature. Highly expressed in the initial segment of the epididymis, where it affects the functional expression of clusterin and phospholipid hydroperoxide glutathione peroxidase (PHGPx), two proteins required for sperm maturation (PubMed:12695510). May also function as an endocytic receptor. Not required for endocytic uptake of SEPP1 in the kidney which is mediated by LRP2 (PubMed:18174160). Together with its ligand, apolipoprotein E (apoE), may indirectly play a role in the suppression of the innate immune response by controlling the survival of myeloid-derived suppressor cells (PubMed:29336888). {ECO:0000269|PubMed:12695510, ECO:0000269|PubMed:18174160, ECO:0000269|PubMed:29336888}. |
Alternative splicing;Calcium;Cell membrane;Disulfide bond;EGF-like domain;Endocytosis;Glycoprotein;Membrane;Metal-binding;Phosphoprotein;Receptor;Reference proteome;Repeat;Secreted;Signal;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
|
axon [GO:0030424]; caveola [GO:0005901]; cell surface [GO:0009986]; dendrite [GO:0030425]; extracellular space [GO:0005615]; membrane [GO:0016020]; microtubule associated complex [GO:0005875]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; receptor complex [GO:0043235]; amyloid-beta binding [GO:0001540]; apolipoprotein binding [GO:0034185]; calcium ion binding [GO:0005509]; calcium-dependent protein binding [GO:0048306]; high-density lipoprotein particle binding [GO:0008035]; kinesin binding [GO:0019894]; low-density lipoprotein particle receptor activity [GO:0005041]; protein N-terminus binding [GO:0047485]; reelin receptor activity [GO:0038025]; very-low-density lipoprotein particle receptor activity [GO:0030229]; ammon gyrus development [GO:0021541]; cellular response to cholesterol [GO:0071397]; cellular response to growth factor stimulus [GO:0071363]; chemical synaptic transmission [GO:0007268]; dendrite morphogenesis [GO:0048813]; endocytosis [GO:0006897]; hippocampus development [GO:0021766]; layer formation in cerebral cortex [GO:0021819]; modulation of chemical synaptic transmission [GO:0050804]; positive regulation of CREB transcription factor activity [GO:0032793]; positive regulation of dendrite development [GO:1900006]; positive regulation of dendritic spine morphogenesis [GO:0061003]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of protein kinase activity [GO:0045860]; positive regulation of protein tyrosine kinase activity [GO:0061098]; reelin-mediated signaling pathway [GO:0038026]; regulation of apoptotic process [GO:0042981]; regulation of innate immune response [GO:0045088]; response to xenobiotic stimulus [GO:0009410]; ventral spinal cord development [GO:0021517] |
12426372_One APOER2 isoform has 4 binding repeats & an adjacent short 13 AA insertion with a furin cleavage site. The resulting receptor fragment (the entire ligand-binding domain is secreted as soluble polypeptide) inhibits Reelin signaling in primary neurons. 12586425_Reelin receptor machinery, including ApoER2 and VLDLR and Dab1 protein, is located in radial glia precursors in cortical wall from the ventricular zone to the pial surface. 12695510_apoER2 is highly expressed in the initial segment of the epididymis, where it affects the functional expression of clusterin and phospholipid hydroperoxide glutathione peroxidase (PHGPx), two proteins required for sperm maturation 12871934_The apolipoprotein E receptor-2 undergoes sequential, gamma-secretase dependent proteolytic processing, resulting in intracellular domain release into the cytoplasm. 15169786_Apolipoprotein E receptors activate p42/44 MAPK and have roles in neurite outgrowth 15175346_Reelin treatment targets adaptor protein Disabled-1(Dab1) for proteolytic degradation by the ubiquitin-proteasome pathway and signaling through neuronal apolipoprotein E receptor 2 can activate the ubiquitin-proteasome machinery 15459198_binding of LDL to platelets resulted in tyrosine phosphorylation of apoER2' 15987942_Reelin binds several receptors. including LrpB ini cultured hippocampux neurons. 16102539_Thus, alternative splicing of Apoer2, a novel component of the NMDA receptor complex, controls the modulation of NMDA receptor activity, synaptic neurotransmission, and memory by Reelin. 16227578_ApoEr2 coprecipitated with APP alpha- and beta-CTF, and F-spondin reduced the levels of APP intracellular domain signaling. 16332682_ApoEr2 can form a multiprotein complex with NMDA receptor subunits and PSD95 17096706_platelets adhere to dimeric beta2GPI under both arterial and venous shear stresses. Platelets adhere via two receptors: GPIbalpha and apoER2'. These receptors are present in a complex on the platelet surface 17174920_Study demonstrates that a differentially spliced isoform of ApoE receptor 2 (Apoer2), is essential for protection against neuronal cell loss during normal aging. 17314095_Sertoli cell ApoER2 is a Sepp1 receptor and a component of the selenium delivery pathway to spermatogenic cells 17494763_These data demonstrate a cell-autonomous function of ApoER2, and most likely VLDLR and Dab1, in postnatal migration of neuroblasts in the forebrain, which is suggested to depend on ligands other than Reelin. 17553992_Apolipoprotein E receptor-2 deficient transgenic mice fed a selenium-deficient diet become neurologically dysfunctional and do not survive. 17907841_ApoER2 knockout mice also displayed altered use of specific search strategies in the maze 17913789_present evidence for divergent roles of the two reelin receptors Vldlr and ApoER2, with Vldlr mediating a stop signal for migrating neurons and ApoER2 being essential for the migration of late generated neocortical neurons 18039658_the activity of PCSK9 and its binding affinity on VLDLR and ApoER2 does not depend on the presence of LDLR. 18089558_Fyn, due in part to its effects on Dab1, regulates the phosphorylation, trafficking, and processing of APP and apoEr2. 18172410_Neurodegeneration in mice resulting from loss of functional apolipoprotein E receptor 2. 18279306_The plasticity associated with the Reelin-Apoer 2-exon 19 pathway is distinct from that which contributes to injury-induced enhancement of 'pain' processing. 18301736_animals null for either Vldlr or Apoer2 individually, exhibit specific and parasagittally-restricted Purkinje cell ectopias. 18706682_LRP8 is capable of altering thrombosis without affecting normal hemostasis through mechanisms both dependent on and independent of apoE. 18804529_we studied the distribution of Apolipoprotein E2 (ApoEr2) in the olfactory bulb 18946489_Data show that thrombospondin-1 (THBS-1) is a novel physiological ligand for ApoER2 and VLDLR, and that lack of THBS-1 results in anatomical abnormalities of the RMS and leads to a reduction of postnatal neuronal precursors entering the OB. 19007311_a novel signaling role for Apoer2 in sperm development and function that is independent of selenium uptake 19167437_This study suggest that ligand-binding type A (LA) repeats 8 of ApoER2 and Reelin repeat 8 interfere with the interaction between the Reelin central fragment and ApoER2. 19661487_apolipoprotein E receptor 2 (ApoER2, LRP8), a member of the LDL receptor family, is a platelet receptor for FXI. 19948739_Differential functions of ApoER2 and very low density lipoprotein receptor in Reelin signaling depend on differential sorting of the receptors. 20427281_Data show that the identification of VLDLR and ApoER2 as Idol(Mylip) targets suggests potential roles for this LXR-inducible E3 ligase in the central nervous system in addition to lipid metabolism. 20453200_Data show that while PCSK9 directly bound to recombinant LDLR, VLDLR, and apoER2 protein in vitro, changes in PCSK9 expression did not alter the level of these receptors in the mouse brain, nor did it regulate BACE1 levels or APP processing. 21048135_Reelin activates the Rho GTPase Cdc42, known for its role in neuronal morphogenesis and directed migration, in an ApoER2-, Disabled-1-, and phosphatidylinositol 3-kinase-dependent manner. 21119114_ApoER2 is involved in pathogenesis of antiphospholipids antibodies. 21347244_ApoEr2 plays important roles in structure and function of CNS synapses and dendritic spines 21976526_ApoER2 plays a pivotal role in the development of the rod bipolar pathway and maintenance of retinal synaptic connectivity. 22170052_activation of VLDLR and apoER2 by reelin and apoE induces ABCA1 expression and cholesterol efflux via a Dab1-PI3K-PKCzeta-Sp1 signaling cascade. 22589174_These results identify LRP8 as a novel positive factor of canonical Wnt signaling pathway. 22761431_apoER2-mediated uptake of long isoform Sepp1 is responsible for selenium distribution to tissues throughout the body. 23209795_Data show identified ApoER2 as being the main receptor responsible for Reelin mediated detachment of neuroblasts and correct migration of early generated interneurons within the OB, a prerequisite for correct OB lamination. 23506116_Dab1 mediated the association of CIN85 with ApoER2 or VLDLR in neurons. 23651543_Data indicate that uptake of Sepp1 and Gpx3 by d-13 visceral yolk sac was independent of apoER2 and megalin. 23758727_there are reelin-independent functions of very-low-density lipoprotein receptor (VLDLR) and low-density lipoprotein receptor-related protein 8 in geniculate nucleus development. 23976984_The Reelin receptors ApoER2 and VLDLr play essential roles in Reelin-mediated migration and positioning of mesencephalic dopaminergic neurons. 24251389_Study shows that the levels of hippocampal ApoE receptors Lrp1 and Apoer2 in vivo are affected isoform specifically by ApoE4 and that the type of receptor affected is context dependent 24381170_Clusterin is a ligand for apolipoprotein E receptor 2 (ApoER2) and very low density lipoprotein receptor (VLDLR) and signals via the Reelin-signaling pathway. 24532792_longer isoforms of Sepp1 with high selenium content interact with a binding site distinct from the ligand-binding domain of apoER2 for selenium delivery 24755222_data demonstrate that several multivalent ligands for ApoER2 induce clustering in transfected cells and primary neurons and that these complexes included other synaptic molecules, such as APP and PSD-95 24760755_Tandem Sepp1-apoER2 interactions supply selenium for maintenance of brain neurons both at the blood-brain barrier and within the brain. Sepp1 inside the blood-brain barrier is taken up by neurons via apoER2, concentrating brain selenium in them. 24840660_Apolipoprotein E receptor-2 deficiency enhances macrophage susceptibility to lipid accumulation and cell death to augment atherosclerotic plaque progression and necrosis. 25197062_genetic variants of apoE and ApoER2 impact cardiovascular health by differentially modulating endothelial function. 25308109_This study demonstrated that ApoER2 is expressed in distinct spatiotemporal patterns in developing mouse cerebral cortex. 25340851_total levels of ApoER2 and surface levels of ApoER2 are increased by active Fyn 25429077_Apoer2 lacking exon 16, the O-linked sugar domain, altered the proteolytic processing and abundance of Apoer2 in cells and synapse number and function in mice, hippocampal spine density, LTP, and cued fear learning. 25892301_This study demonistrated that LRP8-Reelin-Regulated Neuronal Enhancer Signature Underlying Learning and Memory Formation in mice. 26386179_reveals a novel function for Reelin/ApoER2 in peripheral nervous system, inducing cell migration of Schwann cells, a process relevant for peripheral nervous system development and regeneration 26990752_A1-A1, which is a soluble analog of ApoER2 that binds pathological beta2GPI/anti-beta2GPI complexes, has a positive impact on hemodynamics in lupus-prone mice with spontaneous anti-beta2GPI antibodies and hypertension 27810638_findings indicate that impaired processing or localization of apoER2 may contribute to the pathogenic effects of familial Alzheimer's disease mutations in PS1 27909010_Stage-specific labeling of newborn Apoer2-deficient neurons revealed that the neurons ectopically invaded the marginal zone and that neuronal migration of both early- and late-born neurons was disrupted in the intermediate zone in the Apoer2 KO mice. Rescue experiments showed that ApoER2 functions both in cell-autonomous and noncell-autonomous manners. 28978486_GRIP1 binds to apoER2 and EphrinB2 to induce activity-dependent AMPA receptor insertion at the synapse. 30066942_The data of the present study demonstrated that the PCSK9 shRNAmediated antiapoptotic effect induced by MCAO in hyperlipidemic mice is associated with ApoER2 downregulation, which may be a potential new therapy for stroke treatment in patients with hyperlipidemia. 32664652_Angiotensin II Infusion Leads to Aortic Dissection in LRP8 Deficient Mice. 34404233_Protein Phosphatase 2A Activation Via ApoER2 in Trophoblasts Drives Preeclampsia in a Mouse Model of the Antiphospholipid Syndrome. 34452924_Apolipoprotein E receptor 2 deficiency decreases endothelial adhesion of monocytes and protects against autoimmune encephalomyelitis. 34929721_VLDLR and ApoER2 are receptors for multiple alphaviruses. 35192893_Single molecule, long-read Apoer2 sequencing identifies conserved and species-specific splicing patterns. |
ENSG00000157193 |
LRP8 |
183.794769 |
0.2065012473 |
-2.275778 |
0.26111481 |
72.948074 |
0.00000000000000001331061712962665167521598123387592738576957894269846630064257908543368102982640266418457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000004313772768478151805282508421899934360449306580687078849223325960338115692138671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
63.7398623058068 |
11.5994284128596 |
308.66574960225 |
39.8297967288385 |
| ENSMUSG00000028634 |
16656 |
Hivep3 |
protein_coding |
A2A884 |
FUNCTION: Plays a role of transcription factor; binds to recognition signal sequences (Rss heptamer) for somatic recombination of immunoglobulin and T-cell receptor gene segments; Binds also to the kappa-B motif of gene such as S100A4, involved in cell progression and differentiation. Kappa-B motif is a gene regulatory element found in promoters and enhancers of genes involved in immunity, inflammation, and growth and that responds to viral antigens, mitogens, and cytokines. Involvement of HIVEP3 in cell growth is strengthened by the fact that its down-regulation promotes cell cycle progression with ultimate formation of multinucleated giant cells. Strongly inhibits TNF-alpha-induced NF-kappa-B activation; Interferes with nuclear factor NF-kappa-B by several mechanisms: as transcription factor, by competing for Kappa-B motif and by repressing transcription in the nucleus; through a non transcriptional process, by inhibiting nuclear translocation of RELA by association with TRAF2, an adapter molecule in the tumor necrosis factor signaling, which blocks the formation of IKK complex. Interaction with TRAF proteins inhibits both NF-Kappa-B-mediated and c-Jun N-terminal kinase/JNK-mediated responses that include apoptosis and pro-inflammatory cytokine gene expression. Positively regulates the expression of IL2 in T-cell. Essential regulator of adult bone formation. {ECO:0000269|PubMed:10625627, ECO:0000269|PubMed:11035930, ECO:0000269|PubMed:11804591, ECO:0000269|PubMed:12001065, ECO:0000269|PubMed:12193271, ECO:0000269|PubMed:14530385, ECO:0000269|PubMed:14707112, ECO:0000269|PubMed:8255760, ECO:0000269|PubMed:8812474}. |
Coiled coil;Cytoplasm;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:16656; |
cytosol [GO:0005829]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; positive regulation of DNA-templated transcription [GO:0045893]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal muscle cell differentiation [GO:0035914] |
11804591_KRC overexpression inhibits while antisense or dominant-negative KRC enhances NFkappaB-dependent transactivation and JNK phosphorylation and consequently, apoptosis and cytokine gene expression 12378523_KRC may be a previously unidentiified tumor-suppressor gene. 14707112_Overexpression of KRC in transformed and primary T cells leads to increased IL-2 production. 15627499_multiple promoters might be residual traces of regulatory regions of other genes lost in evolution (ZAS3 protein) 16728642_results show Schnurri-3 (Shn3) is an essential regulator of adult bone formation; Shn3 was found to control levels of Runx2, by promoting its degradation through recruitment of WWP1 to Runx2 17966382_study reveals an essential role for Shn3 as a regulator of postnatal bone mass 18257392_Gel mobility shift assays showed altered DNA binding activities of NF-kappaB and AP-1 proteins in ZAS3-deficient tissues, including the thymus. Lymphocyte analysis suggested a subtle function of ZAS3 in regulating T-cell development and activation 20404140_report that mice bearing parallel null mutations in the adapter proteins Schnurri2 (Shn2) and Schnurri3 (Shn3) exhibit defects in patterning of the axial skeleton during embryogenesis. 21390242_ZAS3 was a crucial molecule in osteoclast differentiation. 22573816_Shn3 expression in mesenchymal cells directly controls osteoblastic bone formation and indirectly regulates osteoclastic bone resorption. 23945236_Knockdown of Shn3 in adult mice results in a high-bone mass phenotype. 24563464_Thus, Alg2, as a downstream mediator of Hivep3, suppresses osteogenesis, whereas it promotes chondrogenesis. |
ENSG00000127124 |
HIVEP3 |
103.164289 |
0.2396575076 |
-2.060954 |
0.42957938 |
21.404102 |
0.00000371974427738069105518498719264997021127783227711915969848632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000299308741854530837157681577576084919201093725860118865966796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
40.9039772069618 |
12.3604173634393 |
170.676803006223 |
38.1900448547772 |
| ENSMUSG00000028661 |
13842 |
Epha8 |
protein_coding |
O09127 |
FUNCTION: Receptor tyrosine kinase which binds promiscuously GPI-anchored ephrin-A family ligands residing on adjacent cells, leading to contact-dependent bidirectional signaling into neighboring cells. The signaling pathway downstream of the receptor is referred to as forward signaling while the signaling pathway downstream of the ephrin ligand is referred to as reverse signaling. The GPI-anchored ephrin-A EFNA2, EFNA3, and EFNA5 are able to activate EPHA8 through phosphorylation. With EFNA5 may regulate integrin-mediated cell adhesion and migration on fibronectin substrate but also neurite outgrowth. During development of the nervous system also plays a role in axon guidance. Downstream effectors of the EPHA8 signaling pathway include FYN which promotes cell adhesion upon activation by EPHA8 and the MAP kinases in the stimulation of neurite outgrowth. {ECO:0000269|PubMed:10498895, ECO:0000269|PubMed:11416136, ECO:0000269|PubMed:12681484, ECO:0000269|PubMed:15782114, ECO:0000269|PubMed:17875921, ECO:0000269|PubMed:9214628}. |
ATP-binding;Cell adhesion;Cell membrane;Cell projection;Developmental protein;Endosome;Glycoprotein;Kinase;Membrane;Neurogenesis;Nucleotide-binding;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transferase;Transmembrane;Transmembrane helix;Tyrosine-protein kinase;Ubl conjugation |
|
|
mmu:13842; |
early endosome membrane [GO:0031901]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; ATP binding [GO:0005524]; GPI-linked ephrin receptor activity [GO:0005004]; growth factor binding [GO:0019838]; transmembrane receptor protein tyrosine kinase activity [GO:0004714]; transmembrane-ephrin receptor activity [GO:0005005]; axon guidance [GO:0007411]; cell adhesion [GO:0007155]; cellular response to follicle-stimulating hormone stimulus [GO:0071372]; ephrin receptor signaling pathway [GO:0048013]; neuron projection development [GO:0031175]; neuron remodeling [GO:0016322]; positive regulation of kinase activity [GO:0033674]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of phosphatidylinositol 3-kinase activity [GO:0043552]; protein autophosphorylation [GO:0046777]; regulation of cell adhesion [GO:0030155]; regulation of cell adhesion mediated by integrin [GO:0033628]; substrate-dependent cell migration [GO:0006929]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169] |
12666197_1-kb enhancer region, directs expression in the embryo to the anterior region of the developing midbrain. 17178831_Regulation of EphA8 gene expression is accomplished by TALE homeobox transcription factors during development of the mesencephalon. 17875921_These findings support a possible function for Anks family proteins as scaffolding proteins of the EphA8 signaling pathway. 20100865_Data suggest that Odin levels play a critical role in regulating the stability of EphA2 and A8 in response to ligand stimulation and by modulating the ubiquitination process. |
ENSG00000070886 |
EPHA8 |
141.951152 |
0.1588371599 |
-2.654380 |
0.24291858 |
119.607949 |
0.00000000000000000000000000077084295870635958860393879112640884126754615300541808894206567551530266029294335972110729926498606801033020019531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000510499128131098690152382379690353804372855274059658586650106916238244306183169385349174262955784797668457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.8986036213306 |
6.5806744605924 |
244.896116585092 |
27.0699643797461 |
| ENSMUSG00000028671 |
74246 |
Gale |
protein_coding |
Q8R059 |
FUNCTION: Catalyzes two distinct but analogous reactions: the reversible epimerization of UDP-glucose to UDP-galactose and the reversible epimerization of UDP-N-acetylglucosamine to UDP-N-acetylgalactosamine. The reaction with UDP-Gal plays a critical role in the Leloir pathway of galactose catabolism in which galactose is converted to the glycolytic intermediate glucose 6-phosphate. It contributes to the catabolism of dietary galactose and enables the endogenous biosynthesis of both UDP-Gal and UDP-GalNAc when exogenous sources are limited. Both UDP-sugar interconversions are important in the synthesis of glycoproteins and glycolipids. {ECO:0000250|UniProtKB:Q14376}. |
Carbohydrate metabolism;Galactose metabolism;Isomerase;NAD;Reference proteome |
PATHWAY: Carbohydrate metabolism; galactose metabolism. |
|
Human_homologues hsa:2582; |
cytosol [GO:0005829]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803]; UDP-glucose 4-epimerase activity [GO:0003978]; UDP-N-acetylglucosamine 4-epimerase activity [GO:0003974]; galactose catabolic process [GO:0019388]; galactose catabolic process via UDP-galactose [GO:0033499]; galactose metabolic process [GO:0006012]; glycolytic process from galactose [GO:0061623] |
23257357_results provide evidence that the Xbp1s/GalE pathway functions as a novel regulatory nexus connecting the UPR to the characteristic postprandial metabolic changes in hepatocytes. 28877911_These studies reveal a new link between hepatic GALE activity and whole-body glucose homeostasis via regulation of hepatic Tff3 expression. |
ENSG00000117308 |
GALE |
532.520529 |
0.4745360924 |
-1.075410 |
0.11592294 |
85.498666 |
0.00000000000000000002318579658436907073643889257832078714778569294741682031181506662420588327222503721714019775390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000009173144248652200042473993366383464818319127082816687862587556878679606597870588302612304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
350.103771699258 |
28.1617001705272 |
737.78112416339 |
42.8382861773919 |
| ENSMUSG00000028678 |
73804 |
Kif2c |
protein_coding |
Q922S8 |
FUNCTION: In complex with KIF18B, constitutes the major microtubule plus-end depolymerizing activity in mitotic cells (PubMed:14980225). Regulates the turnover of microtubules at the kinetochore and functions in chromosome segregation during mitosis. Plays a role in chromosome congression and is required for the lateral to end-on conversion of the chromosome-microtubule attachment (By similarity). {ECO:0000250|UniProtKB:Q99661, ECO:0000269|PubMed:14980225}. |
3D-structure;ATP-binding;Cell cycle;Cell division;Centromere;Chromosome;Chromosome partition;Coiled coil;Cytoplasm;Cytoskeleton;Kinetochore;Microtubule;Mitosis;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:73804; |
centrosome [GO:0005813]; chromosome, centromeric region [GO:0000775]; cytoplasm [GO:0005737]; kinesin complex [GO:0005871]; kinetochore [GO:0000776]; microtubule [GO:0005874]; microtubule cytoskeleton [GO:0015630]; microtubule plus-end [GO:0035371]; nucleus [GO:0005634]; spindle [GO:0005819]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule plus-end binding [GO:0051010]; attachment of mitotic spindle microtubules to kinetochore [GO:0051315]; cell division [GO:0051301]; establishment or maintenance of microtubule cytoskeleton polarity [GO:0030951]; metaphase plate congression [GO:0051310]; microtubule depolymerization [GO:0007019]; microtubule-based movement [GO:0007018]; mitotic metaphase plate congression [GO:0007080]; regulation of chromosome segregation [GO:0051983] |
16741559_Possible functions of MCAK at the inner centromere domain and at the perikinetochoric ring during both meiotic divisions. 19283064_Shugoshin 2 is necessary for the loading of MCAK at the inner centromere, but is dispensable for the loading of the outer kinetochore proteins BubR1 and CENP-E. 20406800_MCAK appears to possess a unique distribution and function in oocyte maturation. 20504960_MCAK contributes to chromosome alignment in meiosis I, but is not necessary for preventing chromosome segregation errors. 23055517_PAK1 phosphorylates MCAK and regulates both its localization and function. 31932847_Clathrin's adaptor interaction sites are repurposed to stabilize microtubules during mitosis. 35138249_KIF2C regulates synaptic plasticity and cognition in mice through dynamic microtubule depolymerization. |
ENSG00000142945 |
KIF2C |
619.684553 |
2.4516974651 |
1.293781 |
0.11402303 |
126.951979 |
0.00000000000000000000000000001903234055629412860498624982103573387380807278065929096941125133012834418506356234934706606054533040151000022888183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000014004860451858558424046860534483078506274121972387111028182801001563582109221695048972833319567143917083740234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
884.415554579044 |
69.9723472443166 |
360.736007258546 |
22.1562922765651 |
| ENSMUSG00000028766 |
11647 |
Alpl |
protein_coding |
P09242 |
FUNCTION: Alkaline phosphatase that metabolizes various phosphate compounds and plays a key role in skeletal mineralization and adaptive thermogenesis (PubMed:10620060, PubMed:11028439, PubMed:14982838, PubMed:23942722, PubMed:33981039). Has broad substrate specificity and can hydrolyze a considerable variety of compounds: however, only a few substrates, such as diphosphate (inorganic pyrophosphate; PPi), pyridoxal 5'-phosphate (PLP) and N-phosphocreatine are natural substrates (PubMed:19874193, PubMed:23942722, PubMed:33981039). Plays an essential role in skeletal and dental mineralization via its ability to hydrolyze extracellular diphosphate, a potent mineralization inhibitor, to phosphate: it thereby promotes hydroxyapatite crystal formation and increases inorganic phosphate concentration (PubMed:9056646, PubMed:10620060, PubMed:11004006, PubMed:11028439, PubMed:12082181, PubMed:14982838, PubMed:32035618). Acts in a non-redundant manner with PHOSPHO1 in skeletal mineralization: while PHOSPHO1 mediates the initiation of hydroxyapatite crystallization in the matrix vesicles (MVs), ALPL/TNAP catalyzes the spread of hydroxyapatite crystallization in the extracellular matrix (PubMed:20684022, PubMed:26457330). Also promotes dephosphorylation of osteopontin (SSP1), an inhibitor of hydroxyapatite crystallization in its phosphorylated state; it is however unclear whether ALPL/TNAP mediates SSP1 dephosphorylation via a direct or indirect manner (PubMed:23427088). Catalyzes dephosphorylation of PLP to pyridoxal (PL), the transportable form of vitamin B6, in order to provide a sufficient amount of PLP in the brain, an essential cofactor for enzymes catalyzing the synthesis of diverse neurotransmitters (PubMed:7550313). Additionally, also able to mediate ATP degradation in a stepwise manner to adenosine, thereby regulating the availability of ligands for purinergic receptors (PubMed:19874193, PubMed:23942722, PubMed:23825434, PubMed:32028019). Also capable of dephosphorylating microbial products, such as lipopolysaccharides (LPS) as well as other phosphorylated small-molecules, such as poly-inosine:cytosine (poly I:C) (By similarity). Acts as a key regulator of adaptive thermogenesis as part of the futile creatine cycle: localizes to the mitochondria of thermogenic fat cells and acts by mediating hydrolysis of N-phosphocreatine to initiate a futile cycle of creatine dephosphorylation and phosphorylation (PubMed:33981039). During the futile creatine cycle, creatine and N-phosphocreatine are in a futile cycle, which dissipates the high energy charge of N-phosphocreatine as heat without performing any mechanical or chemical work (PubMed:33981039). {ECO:0000250|UniProtKB:P05186, ECO:0000269|PubMed:10620060, ECO:0000269|PubMed:11004006, ECO:0000269|PubMed:11028439, ECO:0000269|PubMed:12082181, ECO:0000269|PubMed:14982838, ECO:0000269|PubMed:19874193, ECO:0000269|PubMed:20684022, ECO:0000269|PubMed:23427088, ECO:0000269|PubMed:23825434, ECO:0000269|PubMed:23942722, ECO:0000269|PubMed:26457330, ECO:0000269|PubMed:32028019, ECO:0000269|PubMed:32035618, ECO:0000269|PubMed:33981039, ECO:0000269|PubMed:7550313, ECO:0000269|PubMed:9056646}. |
Biomineralization;Calcium;Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Hydrolase;Lipoprotein;Magnesium;Membrane;Metal-binding;Mitochondrion;Phosphoprotein;Reference proteome;Signal;Zinc |
|
This gene encodes a preproprotein that is proteolytically cleaved to yield a signal peptide and a proproptein that is subsequently processed to generate the active mature peptide. The encoded protein is a membrane-bound glycosylated enzyme that catalyzes the hydrolysis of phosphate esters at alkaline pH. The mature peptide maintains the ratio of inorganic phosphate to inorganic pyrophosphate required for bone mineralization. Mice that lack this enzyme show symptoms of osteomalacia, softening of the bones. In humans, mutations in this gene are associated with hypophosphatasia, an inherited metabolic bone disease in which deficiency of this enzyme inhibits bone mineralization leading to skeletal defects. Mutations in the mouse gene mirror the symptoms of human hypophosphatasia. A pseudogene of this gene is present on chromosome X. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Aug 2015]. |
mmu:11647; |
extracellular matrix [GO:0031012]; extracellular membrane-bounded organelle [GO:0065010]; extracellular space [GO:0005615]; mitochondrial intermembrane space [GO:0005758]; mitochondrial membrane [GO:0031966]; plasma membrane [GO:0005886]; ADP phosphatase activity [GO:0043262]; alkaline phosphatase activity [GO:0004035]; ATP hydrolysis activity [GO:0016887]; calcium ion binding [GO:0005509]; inorganic diphosphate phosphatase activity [GO:0004427]; phosphoamidase activity [GO:0050187]; phosphoethanolamine phosphatase activity [GO:0052732]; pyridoxal phosphatase activity [GO:0033883]; pyrophosphatase activity [GO:0016462]; bone mineralization [GO:0030282]; calcium ion homeostasis [GO:0055074]; cellular homeostasis [GO:0019725]; cellular response to organic cyclic compound [GO:0071407]; cementum mineralization [GO:0071529]; dephosphorylation [GO:0016311]; developmental process involved in reproduction [GO:0003006]; endochondral ossification [GO:0001958]; multicellular organism aging [GO:0010259]; phosphate ion homeostasis [GO:0055062]; positive regulation of cold-induced thermogenesis [GO:0120162]; pyridoxal phosphate metabolic process [GO:0042822]; response to antibiotic [GO:0046677]; response to glucocorticoid [GO:0051384]; response to insulin [GO:0032868]; response to lipopolysaccharide [GO:0032496]; response to macrophage colony-stimulating factor [GO:0036005]; response to sodium phosphate [GO:1904383]; response to vitamin B6 [GO:0034516]; response to vitamin D [GO:0033280] |
12082181_correction of bone mineralization abnormalities in knockout mice null for both the TNAP (Akp2) and PC-1 (Enpp1) genes 16159911_The Alpl (Akp2) gene is located within the quantitative trait locus region for alkaline phosphatase activity on chromosome 4. 17539739_alkaline phosphatase 2(Hpp/Hpp) mice develop late-onset skeletal disease, notably defective endochondral ossification and bone mineralization that leads to arthropathies of knees and shoulders 17947448_The process selectively internalizes IAP and may contribute to the appearance of the enzyme in serum and surfactant-like particles. 18838539_Results show that inorganic phosphate (P(i)) levels and TNSALP activity increased in response to androgen/androgen receptor (AR), and P(i) signals increase the expression and translocation of AR. 18997814_TNAP-AID mice expressed alpha-fetoprotein and had deleterious mutations in the tumour suppressor gene Trp53, some of which corresponded to those found in human cancer. 19874193_Tissue-nonspecific alkaline phosphatase is the enzyme that hydrolyzes both ATP and inorganic pyrophosate in matrix vesicles. 20573958_Once the HT2B receptor is expressed, it constitutively controls tissue-nonspecific alkaline phosphatase activity at a post-translational level along the overall period of bone mineral deposition 20683129_These results demonstrate that calcium ions released from apatite are important in the synergistic effect of 20alpha-HC and apatite. 20849921_We show that TNAP knockdown reduces cell proliferation and differentiation into neurons or oligodendrocytes 21191615_Data show that alkaline phosphatase (AP) activity in brain vessels and parenchyma in which AP exhibits specific patterns is attributable to TNAP. 21289095_Results demonstrate that TNAP, regulating both ligand availability and protein expression of P2X7 receptor, is essential for axonal development. 22270475_a link between ATRA-induced mL/B/K-ALP gene transcription and chromatin remodeling 22696173_In the cerebral cortex, myelinated axons, while present in wild-type, were absent in the Akp2( -/- ) mice and these animals also displayed a significantly increased proportion of immature cortical synapses. 23427088_data suggest that the promineralization role of TNAP may be related not only to its accepted pyrophosphatase activity but also to its ability to modify the phosphorylation status of OPN. 23694930_mineralization abnormalities of dentin; reduced overall mineralization with decreased matrix vesicle mineralization in the Phospho1(-/-) mice; almost complete absence of matrix vesicles in mice; further reduction in mineralization. 24288199_Inhibition of rhBMP-2-induced ALP activity by intracellular delivery of SMURF1 in murine calvarial preosteoblast cells. 24315873_Taken together, these data indicate that ATF3 is a novel negative regulator of osteoblast differentiation by specifically suppressing ALP gene expression in preosteoblasts. 24990899_CD73 and TNAP play interactive roles to metabolize luminally applied 5'-AMP in the renal vasculature such that inhibition of both is required to inhibit the production of adenosine. 25014884_Findings demonstrate that Alpl(-/-) mice exhibit a craniofacial skeletal phenotype similar to that seen in infants with HPP, including true bony craniosynostosis in the context of severely diminished bone mineralization 25015959_TNAP plays a role in governing the phosphorylation status of phospholamban in the sarcoplasmic reticulum. 25182511_p107 is required for the efficient recruitment of an activating SWI/SNF chromatin-remodeling complex, an essential event in Alpl induction. 25428889_TNAP in the vasculature contributes to the pathology of medial vascular calcification and that it is a druggable target. 25972450_In cardiac fibroblasts, TNAP expression and activity is induced by sFRP2. 26467745_Prevention of lethal murine hypophosphatasia by neonatal ex vivo gene therapy using lentivirally transduced bone marrow cells expressing Akp-2. 26605996_Despite similar deficiencies in alkaline phosphatase, Alpl(-/-) mice develop craniosynostosis and a brachycephalic/acrocephalic craniofacial shape of variable penetrance. 27466191_These analyses revealed that TNAP deficient mice present an increased proliferation of neural precursors, an altered neuronal morphology, and an augmented neuronal activity. We found that these alterations were associated with a partial downregulation of the purinergic P2X7 receptor (P2X7R). 27932058_TNAP activation in vascular smooth muscle cells (VSMCs) appears sufficient to induce calcification. TNAP activation in VSMCs stimulates expression of chondrocyte markers. 27996212_The results are the first to demonstrate a role for ENC1 in the control of osteoblast differentiation. Additionally, the contrasting mineralization phenotypes and transcriptional patterns seen with coordinate knockdown of both ENC1 isoforms vs selective knockdown of 67 kDa ENC1 suggest opposing roles for the isoforms in regulation of osteoblastic differentiation, through effects on Alpl expression and phosphate cellular 28039309_Our results offer clear evidence that TNAP modulates T lymphocyte function and specifically T cell-dependent colitis. 28673966_Data, including data from studies using cells from transgenic/knockout mice, suggest that Med1 plays role in enamel formation; Med1 induces Alpl via stimulation of Notch1 signaling by forming Notch1-RBP-Jk complex on Alpl promoter. (Med1 = mediator complex subunit 1; Alpl = alkaline phosphatase, liver-bone-kidney; Notch1 = Notch gene homolog 1; RBP-Jk = kappa J region recombining binding protein suppressor of hairless) 28982733_High alkaline phosphatase expression is associated with the development of acute respiratory distress syndrome in influenza-infected mice. 29023576_TNAP overexpression in vascular endothelium in mice leads to an unusual course of coronary atherosclerosis and was accompanied by the reduction in body weight and left ventricular ejection fraction. 29234952_intestinal alkaline phosphatase knockout mice display higher intestinal Ca uptake, which over time appears to correlate with a positive effect on the biomechanical properties of trabecular bone. 30251694_Study results from cerulein induced pancreatitis model in TNAP+/- mice show that altered TNAP expression results in heightened pancreatic inflammation, which may be explained by an augmented response of neutrophils and by a higher sensitivity of acinar cells to cerulein injury. 30342227_TNAP stimulates Erk1/2 activity via both phosphate dependent and independent mechanisms to promote cell cycle progression and cytokinesis in calvarial bone progenitor cells. 31778743_Loss of tissue-nonspecific alkaline phosphatase (TNAP) enzyme activity in cerebral microvessels is coupled to persistent neuroinflammation and behavioral deficits in late sepsis. 31827139_Systemic inhibition of tissue-nonspecific alkaline phosphatase alters the brain-immune axis in experimental sepsis. 32028019_Tissue-nonspecific alkaline phosphatase is an anti-inflammatory nucleotidase. 32035618_Tissue-nonspecific alkaline phosphatase promotes the osteogenic differentiation of osteoprogenitor cells. 33160095_Dental defects in the primary dentition associated with hypophosphatasia from biallelic ALPL mutations. 33498907_Tissue Nonspecific Alkaline Phosphatase Function in Bone and Muscle Progenitor Cells: Control of Mitochondrial Respiration and ATP Production. 33971401_TNAP is a novel regulator of cardiac fibrosis after myocardial infarction by mediating TGF-beta/Smads and ERK1/2 signaling pathways. 33981039_Mitochondrial TNAP controls thermogenesis by hydrolysis of phosphocreatine. |
ENSG00000162551 |
ALPL |
4109.366339 |
0.0856118621 |
-3.546045 |
0.06756947 |
2563.428041 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
642.132441579774 |
30.085644424761 |
7500.50782613084 |
234.80359426503 |
| ENSMUSG00000028785 |
15444 |
Hpca |
protein_coding |
P84075 |
FUNCTION: Calcium-binding protein that may play a role in the regulation of voltage-dependent calcium channels. May also play a role in cyclic-nucleotide-mediated signaling through the regulation of adenylate and guanylate cyclases. {ECO:0000250|UniProtKB:P84074, ECO:0000250|UniProtKB:P84076}. |
Calcium;Cytoplasm;Lipoprotein;Membrane;Metal-binding;Myristate;Reference proteome;Repeat |
|
|
mmu:15444; |
axon [GO:0030424]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; dendrite cytoplasm [GO:0032839]; dendrite membrane [GO:0032590]; dendritic spine head [GO:0044327]; extrinsic component of membrane [GO:0019898]; glutamatergic synapse [GO:0098978]; neuronal cell body membrane [GO:0032809]; perikaryon [GO:0043204]; postsynapse [GO:0098794]; actin binding [GO:0003779]; calcium ion binding [GO:0005509]; identical protein binding [GO:0042802]; kinase binding [GO:0019900]; activation of phospholipase D activity [GO:0031584]; brain development [GO:0007420]; calcium-mediated signaling [GO:0019722]; cellular response to calcium ion [GO:0071277]; cellular response to electrical stimulus [GO:0071257]; cellular response to monosodium glutamate [GO:1904009]; inner ear development [GO:0048839]; negative regulation of guanylate cyclase activity [GO:0031283]; positive regulation of adenylate cyclase activity [GO:0045762]; positive regulation of phospholipase activity [GO:0010518]; positive regulation of protein targeting to membrane [GO:0090314]; regulation of postsynaptic neurotransmitter receptor internalization [GO:0099149]; regulation of voltage-gated calcium channel activity [GO:1901385]; response to Aroclor 1254 [GO:1904010]; response to ketamine [GO:1901986]; response to L-glutamate [GO:1902065]; retina development in camera-type eye [GO:0060041] |
15607944_An age-dependent increase in neurodegeneration occurred in the gene-deleted mice, showing that hippocalcin contributes to neuronal viability during aging. 15878804_These results suggest that hippocalcin plays a crucial role in the Ca(2+)-signaling pathway that underlies long-lasting neural plasticity and that leads to spatial and associative memory. 17257765_These results suggest that hippocalcin plays an important role in calcium extrusion from neurons and, in turn, helps to protect them against calcium-dependent excitotoxin damage in the hippocampus. 22639951_Suggest that the maintenance of parvalbumin and hippocalcin levels by melatonin in ischemic injury contributes to the neuroprotective effect of melatonin against neuronal cell damage. 23260046_Hippocalcin and KCNQ channels contribute to the kinetics of the slow afterhyperpolarization 25752619_results suggest that 7,8-Dihydroxy-4-methylcoumarin prevents glutamate-induced toxicity by scavenging free radicals and regulating hippocalcin expression. 30091036_neurogenic markers, including doublecortin and the neuronal nuclei antigen, and hippocalcin, otub1, and spectrin-alpha are potential molecular links between hippocampal neurogenesis and exercise in the elderly. 30256386_Hippocalcin hasa protective role in heatstroke-induced hypothalamic injury in mice. |
ENSG00000121905 |
HPCA |
9.382232 |
10.6771204024 |
3.416451 |
1.15946191 |
8.577130 |
0.00340411537105439043066068549592273484449833631515502929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0133191588317237274291526816227815288584679365158081054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.4905662190125 |
16.5126443860834 |
1.73179336020224 |
1.23463145388684 |
| ENSMUSG00000028864 |
15234 |
Hgf |
protein_coding |
Q08048 |
FUNCTION: Potent mitogen for mature parenchymal hepatocyte cells, seems to be a hepatotrophic factor, and acts as a growth factor for a broad spectrum of tissues and cell types. Activating ligand for the receptor tyrosine kinase MET by binding to it and promoting its dimerization. {ECO:0000269|PubMed:20624990}. |
3D-structure;Alternative splicing;Direct protein sequencing;Disulfide bond;Glycoprotein;Growth factor;Kringle;Pyrrolidone carboxylic acid;Reference proteome;Repeat;Serine protease homolog;Signal |
|
This gene encodes a protein that binds to the hepatocyte growth factor receptor to regulate cell growth, cell motility and morphogenesis in numerous cell and tissue types. The encoded preproprotein is proteolytically processed to generate multiple protein products, including the hepatocyte growth factor alpha and beta chains, which heterodimerize to form the mature active protein. Although this protein is a member of the peptidase S1 family of serine proteases, it lacks peptidase activity. Homozygous knockout mice for this gene exhibit embryonic lethality due to impaired development of the placenta and liver. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Sep 2015]. |
mmu:15234; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; chemoattractant activity [GO:0042056]; growth factor activity [GO:0008083]; identical protein binding [GO:0042802]; protein-containing complex binding [GO:0044877]; signaling receptor binding [GO:0005102]; animal organ regeneration [GO:0031100]; cell chemotaxis [GO:0060326]; cell morphogenesis [GO:0000902]; cellular response to hepatocyte growth factor stimulus [GO:0035729]; epithelial cell proliferation [GO:0050673]; hepatocyte growth factor receptor signaling pathway [GO:0048012]; liver development [GO:0001889]; myoblast proliferation [GO:0051450]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043154]; negative regulation of extrinsic apoptotic signaling pathway via death domain receptors [GO:1902042]; negative regulation of hydrogen peroxide-mediated programmed cell death [GO:1901299]; negative regulation of inflammatory response [GO:0050728]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of peptidyl-serine phosphorylation [GO:0033137]; negative regulation of release of cytochrome c from mitochondria [GO:0090201]; positive regulation of angiogenesis [GO:0045766]; positive regulation of cell migration [GO:0030335]; positive regulation of DNA biosynthetic process [GO:2000573]; positive regulation of interleukin-10 production [GO:0032733]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of myelination [GO:0031643]; positive regulation of neuron projection regeneration [GO:0070572]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of protein phosphorylation [GO:0001934]; proteolysis [GO:0006508]; regulation of branching involved in salivary gland morphogenesis by mesenchymal-epithelial signaling [GO:0060665]; regulation of p38MAPK cascade [GO:1900744]; regulation of tau-protein kinase activity [GO:1902947]; skeletal muscle cell proliferation [GO:0014856] |
11761336_Effects of thiol antioxidants on hepatocyte growth factor signaling in cardiac myocytes 11784715_Hepatocyte growth factor induces ERK-dependent paxillin phosphorylation and regulates paxillin-focal adhesion kinase association 11836782_Hepatocyte growth factor is essential for migration of myogenic cells and promotes their proliferation during the early periods of tongue morphogenesis in mouse embryos. 11896611_HGF promotes hepatocarcinogenesis through the autocrine activation of HGF-c-met signaling pathway in association with stimulation of angiogenesis by HGF or indirectly through VFGF 11948403_Mitogenic synergy through multilevel convergence of hepatocyte growth factor and interleukin-4 signaling pathways. 12115601_important role of the c-met/scatter factor system in dendritic cell/langerhans cell migration 12193555_estrogen may indirectly mediate mammary epithelial cell proliferation via the regulation of HGF in mammary stromal cells and HGF plays a crucial role in estrogen-induced proliferation in vivo 12351166_is expressed during the embryonic development of the testis and has multiple roles during the morphogenesis of the male gonad 12397180_data indicate that normal cerebellar development and, possibly, function, require HGF and Met, and that proliferation of granule cells in the cerebellum critically depends on full HGF/Met signaling 12482598_HGF has a role in protecting the liver from chemical injury, but its isoforms stimulate injury 12542542_hepatocyte growth factor is definitely involved in enhancing cutaneous wound healing processes, including re-epithelialization, neovascularization, and granulation tissue formation. 12670907_Hepatocyte growth factor/scatter factor induces feedback up-regulation of CD44v6 in melanoma cells through Egr-1. 12878170_HGF-induced up-regulation of CD44 was mediated by the tyrosine kinase signaling pathway, and enhanced adhesion of tumor cell lines to bone marrow-derived endothelial cells, and increased transendothelial migration of cancer cells 12915117_Results suggest that osteopontin activity contributes to hepatocyte growth factor-induced tumor growth and invasiveness. 14500721_HGF is modulated by stanninocalcin 1 and has roles in wound healing, tissue regeneration, and angiogenesis 14517989_Hgf is involved in branching morphogenesis of salivary glands. 14550779_In this study, we first report the promoting effect of HGF on the proliferation of neurospheres and neuronal differentiation of neural stem cells from mouse embryos 14576199_ERK1/2 signaling may play an important role in HGF-mediated SMC migration by contributing to focal adhesion redistribution and FAK and Pyk2 activation 14625309_hepatocyte growth factor inhibits Bid/Bax-induced cell death with p38 MAPK and Akt-dependent pathways but not with ERK1 or ERK2 14759494_HGF and c-Met show delayed expression in the periinfarct region at both protein and mRNA levels after induction of focal cerebral ischemia in mice 15072590_NK4, a proteolytic cleavage product of HGF, may exert antitumor activities not only by antagonizing HGF, but also by inhibiting HGF amplification via tumor-stromal interactions. 15383179_Our results confirm the mitogenic role of HGF/SF in liver regeneration. 15555922_These results indicate that HGF promotes the survival and growth of maturing sympathetic neurons by both PI-3 kinase- and MAP kinase-dependent mechanisms. 15576826_hepatocyte growth factor increases electrical resistance but, at the same time, decreases apical TNFalpha release in uterine epithelial cells 15856015_significant role for stromal TGF-beta signaling in mammary tissue homeostasis and mammary tumor progression via regulation of HGF signalin pathways 16144866_Data suggest that bone marrow consists of a functionally complex population of CD45+ progenitors that includes a subset of hepatocyte growth factor/c-met responsive cells capable of migration to skeletal muscle. 16177122_HGF expresses immunosuppressive effects on dendritic cells without up-regulation of IL-10 or TGF-beta. 16340242_The present findings suggest that the HGF gene delivery can be a novel therapeutic approach against diabetic nephropathy. 16380478_Homozygous p21(cip) loss releases islets from growth inhibition, markedly enhancing proliferation in response to HGF and placental lactogen. 16403606_HGF downregulates transcription factors, Distal-less 5 (Dlx-5), Distal-less 6 (Dlx-6) and Aristaless 4 (Alx-4), in RAW 264.7 osteoclast cells but has an inverse effect in undifferentiated RAW 264.7 cells 16424394_HGF may play a critical role in lymphangiogenesis and potentially contribute to lymphatic metastasis 16476577_findings indicate that: (1) HGF/c-Met signal on mucosa is needed to restore gastric injuries; and (2) the loss of local HGF leads to manifestation of gastric lesions 16513678_Overexpression of hepatocyte growth factor in the airways show increased susceptibility to lung cancer 16648972_Since we demonstrated that not only SDF-1, but also HGF and LIF are upregulated in damaged tissues, we postulate that CXCR4+ c-Met+ LIF-R+ TCSC could be mobilized from the BM into the PB, where they play a role in tissue repair/regeneration. 16651617_These results suggest that HGF ameliorates hepatic biliary fibrosis in part by blocking bile duct epithelial to mesenchymal transition. 16840775_Plasminogen activation can protect the lung against fibrosis by increasing the availability of active HGF. 16877364_Mutant CDK4 dramatically increased the number and the growth kinetics of carcinogen-induced primary melanomas in the skin and promoted the growth of spontaneous metastases in lymph nodes and lungs. 17068118_Upregulation of HGF production is required for protective mechanisms against cholestatic hepatitis. Enhancement of the intrinsic defense system by adding HGF attenuated hepatic inflammation, necrosis, and apoptosis under bile-congestive conditions. 17203235_HGF up-regulates the expression of the bone morphogenetic protein receptors, BMPR-IB and BMPR-II, in prostate cancer cells 17215404_HGF signaling via its receptor c-Met influences the development of gonadotropin hormone-releasing hormone-1. 17241389_The gene expression profiles of drug metabolizing enzymes with an overexpression of hepatocyte growth factor is reported. 17412827_HGF induces the proliferation of colonic epithelial cells via the phosphatidylinositol 3-kinase/Akt signaling pathway. 17561000_PAI-1 deficiency reduces hepatic fibrosis after bile duct obstruction mainly through the activation of tPA and HGF. 17583691_Hgf as a putative endothelium-derived factor that mediates the reciprocal signaling from the vasculature to the respiratory epithelium during distal lung morphogenesis 17688880_Direct contact between platelets and hepatocytes was necessary for the proliferative effect initiating signal transduction involved in growth factor activation: Hepatocyte growth factor, vascular endothelial growth factor, and insulin-like growth factor-1 17702746_CD44 is an important regulator of HGF/c-Met-mediated in vitro and in vivo barrier enhancement, a process with essential involvement of Tiam1, Rac1, dynamin 2, and cortactin. 17761834_Simultaneous HGF gene transfer can suppress GVHD while preserving the GVT effect. 17901400_When hepatocyte growth factor is silenced there is a diminished ability of adipose-derived stem cells to promote reperfusion of ischemic tissue. 17976157_Overexpression of NK2 acts as an antagonist of HGF and promotes liver fibrosis in CCl4-induced chronic liver injury. 18032545_HGF is a positive regulator for eliciting resident kidney tubular cell proliferation during the repair process of acute renal failure. 18073323_HGF has a central role in regulating angiogenesis in adipose tissue 18362143_HGF is a potent activator of glucose transport and metabolism and also a strong inhibitor of fatty acid oxidation in rodent myotubes 18395096_Over expression of HGF inhibited COL1A2 transcription in cultured HSC and suppressed activation of COL1A2 promoter in liver tissue induced by carbon tetrachloride administration. 18495663_analysis of the hepatocyte growth factor-binding site in the immunoglobulin-like region of Met 18504429_ARHGAP12 inactivates RAC1, thereby impairing cell motility, invasion and adhesion to the extracellular matrix. The gene is transcriptionally suppressed by Hepatocyte Growth Factor in human and murine cell lines. 18524894_Hepatocyte growth factor promotes migration of the rostral migratory stream neuroblasts. 18628208_the HGF/c-MET axis has a role in guiding stem cells toward brain injury 18829539_A role for HGF in medulloblastoma initiation and growth and efficacy of HGF-targeted therapy in a mouse model of endogenously arising tumors. 18922968_These studies characterize complex functional roles for HGF and TGF-beta signaling in mediating tumor-stromal interactions during mammary tumor cell scattering and invasion, with important implications in the metastatic process. 18973760_Steric hindrance of the interaction between HGF beta domain binding to Met is sufficient for inhibiting full-length HGF-dependent Met signaling and cell migration. 18988920_induction of Smad7 by HGF serves to limit acquisition of the myofibroblast phenotype in alveolar epithelial cells 19071214_Collectively, these results demonstrate that HGF/SF-Met signal cascade stimulates IL-6 production via PI3 kinase pathway, leading to STAT3 phosphorylation as a secondary effect. 19098908_The hepatocyte growth factor (HGF) receptor, MET, is a transcriptional target of MACC1. 19234748_NK4 could act on tumor cells to inhibit CT26 adhesion to endothelial cells by reducing FAK phosphorylation, which is regulated by inside-out HGF/c-Met signaling, and thereby suppress hematogenous pulmonary metastasis 19249600_plays an important role in tooth development by promoting the proliferation and differentiation of dental papilla cells 19330341_collagen/fibronectin matrix through extracellular signal regulated kinase and phosphoinositide 3-kinase 19448157_These results highlight a defective thrombin-dependent proHGF activation system at the fibrotic phase of bleomycin-induced pulmonary fibrosis. 19470725_Results show that blocking the HGF/c-Met pathway may be clinically useful for the treatment of HNSCC. 19812386_uPA promotes muscle regeneration through HGF activation and subsequent myoblast proliferation. 19853659_Matriptase/epithin participates in mammary epithelial cell growth and morphogenesis through HGF activation 20040596_Appl proteins are required for HGF-induced cell survival and migration via activation of Akt 20147731_Hepatocyte growth factor likely plays a role in cyclic endometrial remodeling, such as cell proliferation via autocrine/paracrine mechanisms in mouse uterus. 20332205_Antigen-specific response assays showed that HGF is a potent immunomodulatory factor that inhibits dendritic cell (DC) function along with differentiation of IL-10-producing T(reg) cells and down-regulation of surface markers of T-cell activation. 20375988_Results indicate that HGF/c-met signaling has an important role in protecting podocytes from injury, thereby reducing proteinuria. 20382450_The authors found that ethanol per se elevated NGF, BDNF, HGF, and VEGF measured by ELISA in brain limbic system areas. In the liver, early exposure to ethanol solution and red wine depleted BDNF and VEGF concentrations. 20442248_controls satellite cell growth 20506405_in myeloid cells, Gab1 is likely to enhance HGF mitogenicity by coupling Met to Shp-2 and GATA-2 expression, thereby potentially contributing to normal myeloid differentiation as well as oncogenic transformation 20508974_HGF and IGF-1 appear to play an important role during regeneration of the masseter muscle in mdx mice. 20596656_Even if serum HGF levels increased due to liver resection during the regeneration period, residual liver metastases of colorectal carcinoma was not promoted in its progression. 20639480_Data show that D74-HGF/c-Met axis defines a novel physiologic survival pathway in mature B cells, resulting in the control of the humoral immune response. 20649939_Results demonstrate that primary and metastatic melanomas developing spontaneously in Hgf-Cdk4(R24C) mice effectively evade cellular immune surveillance. 20852387_Smad2 loss causes HGF upregulation via loss of Smad2-mediated transcriptional repression and enhanced Smad3/4-mediated transactivation. 21094172_HGF from local smooth muscle cells plays an important role for the differentiation of homing bone marrow mesenchymal stem cells. 21148681_Activation of the RhoA/Rho kinase pathway up-regulates transcriptional HGF production in response to apoptotic cells. 21187969_the serum-induced increase in cell surface GLUT4 levels is not due to inhibition of its internalization and is not mediated by insulin, PDGF, IGF-1, or HGF 21209281_In vivo administration of hybrid interleukin (IL)-7/HGFbeta chain following bone marrow transplantation supports rapid, complete thymic regeneration, efficiently reconstituting the naive T cell pool and its normally diverse receptor repertoire. 21292498_statins inhibit GGPP biosynthesis in the mevalonate pathway, and then inhibit signal transduction in the Ras/ERK and Ras/Akt pathways, thereby inhibiting bFGF, HGF, TGF-beta expression 21554929_ganglioside GD1a negatively regulates HGF expression through caveolin-1. 21595927_These studies are the first confirm that interference with the HGF/c-met/Stat3 signaling pathway can block tumor cell invasion in an in vivo model. 21683144_These results suggest that hepatocyte growth factor influences multiple steps in postnatal forebrain neurogenesis 21703184_investigated the role of SOCS1 in regulating hepatocyte proliferation following partial hepatectomy and HGF stimulation 21784852_a novel role for Btk in HGF-induced DC inhibition. 21940662_Data show that gradients of lymphatic fluid stasis regulate the expression of lymphangiogenic cytokines (VEGF-A, VEGF-C, and hepatocyte growth factor) and that paradoxically the expression of these molecules is increased in wild-type mice. 22001117_HGF-induced migration was restored in myoblasts isolated from a transgenic mouse expressing alpha-syntrophin only in muscle cells. 22095660_Hepatocyte growth factor/c-met signaling is required for stem-cell-mediated liver regeneration in mice. 22245998_The data showed that the activation of hepatocyte growth factor/c-Met signaling is a major pathway involved type II cell proliferation induced by ultrafine carbon black. 22287599_HGF/c-Met plays a crucial role in host and donor cells of the liver for the cursiv selection of transplanted hepatocytes. Modulating HGF-dependent signalling seems a promising therapeutic option to favour expansion of transplanted hepatocytes. 22318499_HGF is a biomarker and mediator in cardioprotection and cardiovascular regeneration [review] 22427375_These studies indicate that HGF/c-Met signaling is essential for maternal beta-cell adaptation during pregnancy and that its absence/attenuation leads to gestational diabetes mellitus. 22441736_The findings indicate that in vivo exposure to apoptotic cells enhances transcriptional HGF production in bleomycin-stimulated lungs, resulting in attenuation of lung injury and fibrosis. 22535512_These results demonstrate that modulation of learning and memory performance is an important physiological function of HGF that contributes to normal CNS plasticity 22740630_Mer of the TAM-family receptors is responsible for mediating transcriptional HGF production through a RhoA-dependent pathway. 22996389_The binding ability of mouse HGF alpha-chain, or of beta-chain, to human c-Met was lower than that of human HGF. 23024263_HGF is a growth factor playing a key role in islet mass increase and hyperinsulinemia in diet-induced obesity rats; the HGF-Met axis may have a role on insulin signaling in the liver 23064365_In vivo inhibition of HGF-mediated c-Met activation blocked early, enhanced colon dysplasia in Tpl2-deficient mice, indicating that Tpl2 normally suppresses the HGF/c-Met pathway 23077169_Data suggest that Hgf acts as antiapoptotic and differentiation factors in fetal Leydig cells; Hgf increases expression of insulin-like 3, marker of Leydig cell differentiation, and decreases expression of nestin, marker of Leydig cell progenitors. 23224784_a new role of HGF in the modulation of signaling pathways implicated in the activation or repression of cardiomyogenesis 23229794_The ERK/Akt pathway particularly may be critical in the HGF-induced Epithelial-mesenchymal transition process 23233163_These data suggest that the PI3K/Akt, MAPK and STAT3 pathways, downstream of HGF/cMet signalling, are involved in the regulation of VEGF expression 23459311_we show that HGF signaling is necessary for alveolar homeostasis in the developing lung and that augmentation of HGF signaling can improve airspace morphology in murine emphysema. 23485708_These data suggest that IL-8 participates in the formation of cystic structures by Madin-Darby canine kidney cells in 3D culture and that HGF may stimulate tubulogenesis through the suppression of IL-8. 23527275_Conditional genetic elimination of hepatocyte growth factor in mice compromises liver regeneration after partial hepatectomy. 23560074_Bone marrow progenitor cells release HGF, which inhibits miR-155-mediated profibrosis signaling, thereby preventing cardiac fibrosis. 23627689_Adipose tissue derived stem cells from diabetic mice released lower amounts of HGF, a growth factor playing an important role in skin repair. 23722408_VEGF expression of CT26 cells was amplified through tumor-stromal interaction, i.e., the HGF paracrine loop. 23723997_Circulating tumor cells have increased expression of HGF and c-Met in comparison to the primary tumor cells. 23773083_Murine macrophages promote in-vitro wound repair. 23840657_HGF mediates the anti-inflammatory effects of platelet-rich plasma inn a mouse model of tendon injury. 23886856_It was concluded that Epim deletion inhibits polyposis in Apcmin/+ mice, associated with increased mucosal TGF-beta signaling and decreased myofibroblast HGF expression and secretion. 23922381_Up-regulation of COX-2/PGE2 and HGF through a positive-feedback loop may be an important mechanism whereby apoptotic cell instillation exerts the net results of anti-inflammatory, antiapoptotic, and antifibrotic action. 24089510_These results indicate that HGF/c-Met signaling is critical for beta-cell proliferation in situations of diminished beta-cell mass and suggest that activation of this pathway can enhance beta-cell regeneration. 24223113_Differences in the intensity and quality of melanin biosynthesis as a result of HGF overexpression. 24224701_It is a pleiotropic cytokine related with cell proliferation and survivaland ameliorates liver damage in viral hepatitis. 24246674_These results indicate that Hrs plays important roles during peripheral development and physiological functions of B lymphocytes. 24302003_Preadipocyte-secreted HGF is an important regulator of neovascularization in developing fat pads. 24349415_The enhanced thymic reconstitution in the rIL-7/HGFbeta-treated allogeneic BMT recipients results in increased number and functional activities of peripheral T cells. 24469707_In conclusion, HGF, MACC1 and C-met genes involve into malignant cervical tumors occurrence, development and prognosis, and might become potential molecular target therapy site of cervical cancer 24569592_In mechanism, Hgf expression is inhibited by NF-kappaB through suppression of PPARgamma function in the Hgf gene promoter. 24618323_enhanced corneal HGF levels increase signaling through the c-met receptor, decrease mTOR levels, and enhance proinflammatory cytokines, while decreasing anti-inflammatory cytokines, and that HGF signaling is central to disease outcome. 24634221_these data provide evidence in support of a significant role for HGF-induced c-Met/PI3k/Akt signaling and NADPH oxidase activation in lamellipodia formation and motility of lung endothelial cells. 24685580_Matriptase is required for the active form of hepatocyte growth factor induced Met, focal adhesion kinase and protein kinase B activation on neural stem/progenitor cell motility. 24811579_In hepatic stellate cells, CYLD removed HDAC7 from the hepatocyte growth factor promoter and induced HGF expression. 24865428_selected 68 different antibodies that compete with HGF for binding to MET. HGF-competing antibodies recognized 4 distinct hotspots localized in different MET domains 24959005_upregulation of the COX-2/PGE2 and HGF in macrophages following exposure to apoptotic cells represents a mechanism for mediating the anti-inflammatory and antifibrotic consequences of apoptotic cell recognition 25114100_by inducing GILZ in dendritic cells, HGF reproduces the mechanism of immune regulation induced by potent immunomodulatory factors and therefore HGF therapy may have potential in the treatment of autoimmune dysfunctions. 25362352_Regulation of Hgf expression by Hh signalling is indirect, mediated by Hedgehog-induced expression of the microRNAs miR-26a and miR-26b, which in turn downregulate expression of Hgf. 25503558_HGF/c-MET signaling promotes tumor formation after tissue injury in a mouse model of primary soft-tissue sarcoma 25510828_HGF signaling through c-Met may decrease inflammation in endothelial cells and protect against intracranial aneurysm rupture. 25521737_HGF pathway is suggested to contribute to the lymphomagenesis in the MALT lymphoma after H. heilmannii infection. 25571972_Apoptosis of cardiomyocytic & neuronal cells released HGF & HGF-mediated migration of mesenchymal stem cells towards apoptotic targets. HMGB1 was mainly released by necrotic cells& mediated recruitment of monocytes and immature dendritic cells 25586556_inhibition of PPARgamma activity reversed the expression of transforming growth factor-beta (TGF-beta), interleukin (IL)-10, and hepatocyte growth factor (HGF). 25905717_Here, we report a novel hybrid mathematical modeling strategy to systematically unravel hepatocyte growth factor (HGF) stimulated phosphoinositide-3-kinase (PI3K) and mitogen activated protein kinase (MAPK) signal. 25906153_Results indicate that the production of a second pool of HGF by myogenic cells, negatively regulated by NF-kappaB/p65, is crucial for inflammation resolution and the completion of repair in dystrophic skeletal muscle. 25938594_HGF entering alveoli from the bloodstream during pneumonia compensates for destruction by Dppi-activated inflammatory proteases to allow HGF to contribute to epithelial repair. 26001768_findings provide novel insights into the cellular and molecular mechanisms mediating the essential role of HGF/c-Met signaling during oval cell-mediated mouse liver regeneration. 26093299_HGF and c-met inhibition by means of a novel monoclonal neutralizing antibody inhibits tumor growth in prostate cancer cells and in mouse xenografts. 26165517_These data strongly indicate that paracrine Met signaling can control the function of luminal progenitors and modulate their fate during mammary development and tumorigenesis. 26290232_We show that a subset of adult calcitonin gene-related peptide (CGRP)-expressing myenteric neurons produce MET, the receptor for hepatocyte growth factor. 26393505_Molecular and functional studies revealed that ectopic Met expression in limb mesenchyme does not alter HGF expression patterns and levels, but impairs HGF bioavailability 26551462_These findings suggest that HGF overexpression can provide a potential therapeutic avenue as a combination therapy with disease-modifying therapies in SBMA. 26739889_Notch signaling plays an important role in regulation of interactions between TGF-beta and HGF signaling pathways in proximal tubule epithelial cells 26818605_Study used biochemical and morphological analyses to demonstrate that two discrete intracellular signaling pathways underlie distinct HGF-induced biological outcomes in developing neocortical neurons. Further, it identified a key developmental epoch, corresponding to the period of dendritic outgrowth and synaptogenesis, during which HGF stimulation elicits maximal activation of MET in the neocortex. 26875548_Exposure of macrophages to cyclooxygenase (COX-2) inhibitors RhoA and LA-4 cells to antagonists of prostaglandin E2 (PGE2) receptor , PGD2 receptors or the hepatocyte growth factor (HGF) receptor c-Met (PHA-665752), reversed EMT inhibition by the conditioned medium 27030285_The results suggest that PGRN may affect the differentiation of retinal precursor cells to photoreceptor cells through the HGF receptor signaling pathway. 27109105_Our results show that BMF-derived IL-6/HGF and cancer cell-derived TGF-b1 mediate the interactions between bone marrow-derived myofibroblasts and gastric cancer cells, which regulate cancer stemness and promote tumorigenesis. 27143995_HGF displays an antioxidant response by inducing the glutathione-related protection system. 27388243_this study demonstrates an important role for HGF in the protective effects mediated by mesenchymal stromal cells in vivo in the bleomycin model of idiopathic pulmonary fibrosis 27779646_this study revealed that HB-EGF as well as HGF inhibited BDL-induced cholestatic liver injury by predominantly exerting acute cytoprotective and chronic antifibrotic effects, respectively. 28270618_HGF supports hindlimb motor neurons through c-Met; CNTF supports subsets of axial motor neurons through CNTFRalpha; and Artemin acts as the first survival factor for parasympathetic preganglionic motor neurons through GFRalpha3/Syndecan-3 activation. 28429746_Arf6 expressed in skin keratinocytes through the HGF/c-Met signaling pathway in response to skin wounding plays an important role in skin wound healing by regulating membrane dynamics-based motogenic cellular function of keratinocytes. 28864680_prolonged treatment of single HGF/c-Met or Hh inhibitor leads to resistance to these single inhibitors, likely because the single c-Met treatment leads to enhanced expression of Shh, and vice versa. Targeting both the HGF/c-Met and Hh pathways simultaneously overcame the resistance to the single-inhibitor treatment and led to a more potent antitumor effect in combination with the chemotherapy treatment. 28969681_Mesenchymal stem cells microvesicles stabilization of endothelial barrier function is partly mediated by hepatocyte growth factor. 29040876_These results indicate that expression and production of HGF are regulated in a species-specific adipogenic differentiation-dependent manner and suggest that the decrease in HGF mRNA in mouse differentiated adipocytes is, at least in part, mediated by differentiation-dependent loss of its stability. 29343699_CBD-HGF combined with hydrogel scaffold may be promising for the treatment of serious SCI. 29436622_HGF is beneficial for bone regeneration via increased expression of BMP-2, which leads to neovascularization and bone regeneration through regulation of the NF-kappaB signaling pathway. 29771943_these findings highlight the relevance of cross-species protein interactions between murine feeder cells and human epithelial cells in 3T3-J2 co-culture and demonstrate that STAT6 phosphorylation occurs in response to MET activation in epithelial cells. However, STAT6 nuclear translocation does not occur in response to HGF, precluding the transcriptional activity of STAT6. 30066918_these results suggested that HGF may exert beneficial effects on type II diabetesinduced chronic renal failure via regulation of the NFkappaB signaling pathway. 30103949_c-Fos is necessary for HGF-mediated gene regulation and cell migration in Schwann cells. 30242894_findings highlight that the activation of the mTOR/STAT-3 pathway provides novel mechanistic insights into MSC-secreted HGF protection against LPS-induced vascular endothelial permeability dysfunction and apoptosis, which contributes to decreasing microvascular loss and lung injury. 30401749_Activation of hepatocyte growth factor/MET signaling initiates oncogenic transformation and enhances tumor aggressiveness in the murine prostate. 30627913_It can be assumed that Sema3a synthesized in the thymus stroma can act as a functional antagonist of HGF and KGF and have an inhibitory effect when these growth factors are administered into the body for the therapeutic purpose of restoring thymus functions. 30861238_Adipose-derived stem cell therapy ameliorates ionizing irradiation fibrosis via HGF-mediated TGFB downregulation and recruitment of bone marrow cells. 31115492_these findings suggested that NMDA receptor activation may downregulate HGF by inhibiting ERK signaling in BMMSCs, thus weakening their protective effects on BLMinduced lung epithelial cell damage. 31212972_Our results suggest the importance of the microenvironmental generation of tcHGF in the regulation of development, regeneration, and stem cell behavior. 31972251_Activated Hgf-Met Signaling Cooperates with Oncogenic BRAF to Drive Primary Cutaneous Melanomas and Angiotropic Lung Metastases in Mice. 32152201_Noncoding Microdeletion in Mouse Hgf Disrupts Neural Crest Migration into the Stria Vascularis, Reduces the Endocochlear Potential, and Suggests the Neuropathology for Human Nonsyndromic Deafness DFNB39. 32204446_A Signaling Crosstalk between BMP9 and HGF/c-Met Regulates Mouse Adult Liver Progenitor Cell Survival. 32258116_Upregulation of PDGF Mediates Robust Liver Regeneration after Nanosecond Pulsed Electric Field Ablation by Promoting the HGF/c-Met Pathway. 32522231_Pulp stem cells with hepatocyte growth factor overexpression exhibit dual effects in rheumatoid arthritis. 32539223_TLR3 promotes hepatocyte proliferation after partial hepatectomy by stimulating uPA expression and the release of tissue-bound HGF. 32983173_HGF-MET Signaling Shifts M1 Macrophages Toward an M2-Like Phenotype Through PI3K-Mediated Induction of Arginase-1 Expression. 33635004_Ursodesoxycholic acid alleviates liver fibrosis via proregeneration by activation of the ID1-WNT2/HGF signaling pathway. 34819402_Anti-inflammatory effect of HGF responses to oral traumatic ulcers using an HGF-Tg mouse model. 35093804_Resveratrol inhibited hepatocyte apoptosis and alleviated liver fibrosis through miR-190a-5p /HGF axis. 36293268_Upregulation of Reg IV and Hgf mRNAs by Intermittent Hypoxia via Downregulation of microRNA-499 in Cardiomyocytes. |
ENSG00000019991 |
HGF |
65.742794 |
6.3487882633 |
2.666481 |
0.40299347 |
42.353974 |
0.00000000007616141720515054592340174551039688397024995936135383090004324913024902343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000012289096471068358643519611057164965528532007965623051859438419342041015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
117.608832583628 |
33.2810179881251 |
18.5246109502637 |
4.18871617382141 |
| ENSMUSG00000029001 |
230903 |
Fbxo44 |
protein_coding |
F6UI76 |
Human_homologues FUNCTION: Substrate-recognition component of the SCF (SKP1-CUL1-F-box protein)-type E3 ubiquitin ligase complex. |
Proteomics identification;Reference proteome |
|
|
|
cytoplasm [GO:0005737] |
25970626_FBXO44-mediated degradation of RGS2 protein uniquely depends on a Cul4B/DDB1 complex. |
ENSG00000132879 |
FBXO44 |
25.249535 |
2.3990513831 |
1.262464 |
0.51063140 |
6.080859 |
0.01366543444581472949161948804430721793323755264282226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0438030087286721270256606430848478339612483978271484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.8458274105721 |
12.5925571827977 |
14.524835798302 |
4.06601570866854 |
| ENSMUSG00000029093 |
81840 |
Sorcs2 |
protein_coding |
Q9EPR5 |
FUNCTION: The heterodimer formed by NGFR and SORCS2 functions as receptor for the precursor forms of NGF (proNGF) and BDNF (proBDNF) (PubMed:22155786, PubMed:24908487, PubMed:27457814, PubMed:29909994). ProNGF and proBDNF binding both promote axon growth cone collapse (in vitro) (PubMed:24908487). Plays a role in the regulation of dendritic spine density in hippocampus neurons (PubMed:29909994). Required for normal neurite branching and extension in response to BDNF (PubMed:27457814, PubMed:29909994). Plays a role in BDNF-dependent hippocampal synaptic plasticity (PubMed:29909994, PubMed:27457814). Together with NGFR and NTRK2, is required both for BDNF-mediated synaptic long-term depression and long-term potentiation (PubMed:27457814). ProNGF binding promotes dissociation of TRIO from the heterodimer, which leads to inactivation of RAC1 and/or RAC2 and subsequent reorganization of the actin cytoskeleton (By similarity). Together with the retromer complex subunit VPS35, required for normal expression of GRIN2A at synapses and dendritic cell membranes (PubMed:28469074). Required for normal expression of the amino acid transporter SLC1A1 at the cell membrane, and thereby contributes to protect cells against oxidative stress (PubMed:30840898). {ECO:0000250|UniProtKB:Q96PQ0, ECO:0000269|PubMed:22155786, ECO:0000269|PubMed:24908487, ECO:0000269|PubMed:27457814, ECO:0000269|PubMed:28469074, ECO:0000269|PubMed:29909994, ECO:0000269|PubMed:30840898}. |
3D-structure;Cell membrane;Cell projection;Cytoplasmic vesicle;Disulfide bond;Endosome;Glycoprotein;Membrane;Postsynaptic cell membrane;Reference proteome;Repeat;Signal;Synapse;Synaptosome;Transmembrane;Transmembrane helix |
|
|
mmu:81840; |
cytosol [GO:0005829]; dendritic spine [GO:0043197]; early endosome membrane [GO:0031901]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic density membrane [GO:0098839]; recycling endosome membrane [GO:0055038]; intracellular protein transport [GO:0006886]; long-term synaptic depression [GO:0060292] |
15009648_SorCS3 and the homologous sorCS1 and sorCS2 genes were expressed in a combinatorial mostly non-overlapping pattern in the developing and mature central nervous system; activity-dependent induction of sorCS3 did not require protein synthesis 26964886_SorCS2 is expressed in mesodermally derived tissues such as striated muscle, adipose tissue, ossifying bone and connective tissue. 28346477_Results indicate that hearing loss and hair bundle defects are associated with SorCS2 (Sortilin-related VPS-10 domain containing protein) disruption. 29405286_THe sortilin and SorCS2 are extensively present in several distinct anatomical areas in the developing nervous system and are rarely co-expressed. 30061605_structural analysis of binding of NGF, proNGF, and proBDNF to SorCS2 31988435_SorCS2 is required for social memory and trafficking of the NMDA receptor. 34611268_Astrocytic microdomains from mouse cortex gain molecular control over long-term information storage and memory retention. |
ENSG00000184985 |
SORCS2 |
762.587576 |
0.2803386243 |
-1.834758 |
0.14199268 |
161.442684 |
0.00000000000000000000000000000000000054758021564257679746176076849574129548814754385361142901476258563140063264981199150030615008873276228218429650951293297111988067626953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000556049456311181944636479074980434939522209925162689394459198336025759420080012536130784875539057043880575292860157787799835205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
329.19992956251 |
32.4527935718127 |
1174.29387546957 |
83.6675497017271 |
| ENSMUSG00000029095 |
231148 |
Ablim2 |
protein_coding |
Q8BL65 |
FUNCTION: May act as scaffold protein. May stimulate ABRA activity and ABRA-dependent SRF transcriptional activity. |
Alternative splicing;Cytoplasm;LIM domain;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Zinc |
|
|
mmu:231148; |
actin cytoskeleton [GO:0015629]; myofibril [GO:0030016]; actin filament binding [GO:0051015]; metal ion binding [GO:0046872]; cytoskeleton organization [GO:0007010]; lamellipodium assembly [GO:0030032]; positive regulation of transcription by RNA polymerase II [GO:0045944]; transcription by RNA polymerase II [GO:0006366] |
16005990_The highest expression of both isoforms was observed in rat brain and eye and in mouse embryos. ABLIM2 protein was necessary for the normal functioning of neurons. 17194709_ABLIM-2 and -3 associate with STARS and directly bind F-actin |
ENSG00000163995 |
ABLIM2 |
28.442733 |
0.0598054533 |
-4.063579 |
0.68956965 |
37.320852 |
0.00000000100207411668315875605317833205555946851639248507126467302441596984863281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000141198824656039547230628182798370384709585323435021564364433288574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.60574106886925 |
1.29344242197355 |
43.5702920600385 |
11.8810624376406 |
| ENSMUSG00000029153 |
433904 |
Ociad2 |
protein_coding |
Q9D8W7 |
|
Acetylation;Endosome;Reference proteome |
|
|
mmu:433904; |
endosome [GO:0005768]; mitochondrial inner membrane [GO:0005743]; mitochondrion [GO:0005739]; response to bacterium [GO:0009617] |
Human_homologues 22726067_Like OCIAD1, OCIAD2 is a cancer-related protein and its expression level increases during the course of malignant progression and is thought to be a very useful marker for evaluating the malignancy of ovarian mucinous tumors. 24270855_OCIAD2 also increased the interaction of nicastrin with C99 and stimulated APP processing via gamma-secretase activation, but did not affect Notch processing 24949437_it well explained the mechanism of TGFbeta induced OCIAD2 expression in cancer microenvironment, therefore providing an important clue for the future functional analysis of OCIAD2 in tumor pathogenesis. 28911005_these data indicate that reduced expression of OCIAD2 by DNA hypermethylation plays an important role in hepatocellular carcinoma tumor growth and invasion. 30320419_Our results suggest that OCIAD2 could be a useful prognostic biomarker of lung adenocarcinoma 34371038_Cellular OCIAD2 protein is a proviral factor for hepatitis C virus replication. 34628698_Ovarian carcinoma immunoreactive antigen domain 2 controls mitochondrial apoptosis in lung adenocarcinoma. 35080992_Ovarian carcinoma immunoreactive antigen-like protein 2 (OCIAD2) is a novel complex III-specific assembly factor in mitochondria. 35920378_Aberrant OCIAD2 demethylation in lung adenocarcinoma is associated with outcome. |
ENSG00000145247 |
OCIAD2 |
136.343797 |
2.0398245935 |
1.028445 |
0.23611085 |
18.859407 |
0.00001407154623465675935816299874092649702106427866965532302856445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001015055591834949569448806605542756642535096034407615661621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
181.482230323341 |
29.8340698752528 |
88.9695275276968 |
11.0764248354535 |
| ENSMUSG00000029161 |
68567 |
Cgref1 |
protein_coding |
E9QPK1 |
Human_homologues FUNCTION: Mediates cell-cell adhesion in a calcium-dependent manner (By similarity). Able to inhibit growth in several cell lines. {ECO:0000250}. |
Calcium;Metal-binding;Proteomics identification;Reference proteome;Signal |
|
|
Human_homologues hsa:10669; |
calcium ion binding [GO:0005509] |
Human_homologues 19913121_Observational study of gene-disease association. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 26022276_Results clearly indicated that CGREF1 is a novel secretory protein, and plays an important role in regulation of AP-1 transcriptional activity and cell proliferation. |
ENSG00000138028 |
CGREF1 |
2495.668200 |
0.3651469405 |
-1.453451 |
0.07452467 |
371.053123 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000011038179624925989294274137486342294584873568034240871919600343813484135731456524477038400418542411985171156587421563788222265457694398384854460498195010942616821756292831232839986491 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000431111671915058096172034248058979263734670816621502898683834590536366829740734547217727487411997114386506202932819369285000764899836482765423894881830703945347415541953935273121168 |
Yes |
No |
1341.51142937638 |
69.3286977549818 |
3673.89475457035 |
138.195614292931 |
| ENSMUSG00000029162 |
16548 |
Khk |
protein_coding |
A0A0J9YTU1 |
Human_homologues FUNCTION: Catalyzes the phosphorylation of the ketose sugar fructose to fructose-1-phosphate. {ECO:0000269|PubMed:12941785}. |
Proteomics identification;Reference proteome |
Human_homologues PATHWAY: Carbohydrate metabolism; fructose metabolism. |
|
|
ketohexokinase activity [GO:0004454]; fructose metabolic process [GO:0006000] |
20841500_These studies are the first demonstration that neither Khk isoform is required for normal growth and development. 22371574_Fructose-induced metabolic syndrome is prevented in mice lacking both fructokinase B and A but is exacerbated in mice lacking fructokinase A. 23813872_High-fat and high-sucrose (western) diet induces steatohepatitis that is dependent on fructokinase. 24336030_Fructokinase-knockout mice with delayed hydration were protected from renal injury. Thus, recurrent dehydration can induce renal injury via a fructokinase-dependent mechanism, likely from the generation of endogenous fructose via the polyol pathway. 24876114_These studies identify fructokinase as a novel mediator of diabetic nephropathy and document a novel role for endogenous fructose production, or fructoneogenesis, in driving renal disease. 25187370_This study demonstrates that blocking KHK and redirecting fructose metabolism to alternative pathways is an effective way to prevent visceral obesity and insulin resistance induced by high fructose, a widespread component of Western diets. 26083752_myocardial hypoxia actuates fructose metabolism in human and mouse models of pathological cardiac hypertrophy through hypoxia-inducible factor 1alpha (HIF1alpha) activation of SF3B1 and SF3B1-mediated splice switching of KHK-A to KHK-C 26084694_Feedforward upregulation of fructolytic and gluconeogenic enzymes specifically requires GLUT5 and KHK and may proactively enhance the intestine's ability to process anticipated increases in dietary fructose concentrations. 26316589_Significant levels of blood fructose are maintained independent of dietary fructose, KHK, and GLUT5, probably by endogenous synthesis of fructose. 27465991_Fructokinase knockout mice were protected from renal injury both at baseline and after high salt intake (3 wk) compared with wild-type mice. This was associated with higher levels of active (phosphorylated serine 1177) endothelial nitric oxide synthase in their kidneys. 27852737_This studies provided the first evidence that fructokinase modulates vasopressin synthesis in the SON and secretion from the posterior pituitary into circulation. 29533924_studies suggest that it is the inhibition of the Khk C isoform, not the A isoform, that protects animals from Hereditary fructose intolerance. 29604362_These results suggest that KHK-C might play a deleterious role in diabetic kidney disease progression through endogenous fructose metabolism, and that KHK-A plays a unique protective role against the development of diabetic kidney disease. 31001414_Manual correction of the input Khk gene model provided a much more accurate estimation of relative Khk isoform expression when compared to quantitative PCR. 32502381_Deletion of Fructokinase in the Liver or in the Intestine Reveals Differential Effects on Sugar-Induced Metabolic Dysfunction. |
ENSG00000138030 |
KHK |
32.687831 |
0.2114067067 |
-2.241907 |
0.51528797 |
18.163130 |
0.00002027665874616155908326080303805127869054558686912059783935546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001417411959713322036364530065455369367555249482393264770507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
11.0759110732156 |
4.04063708297917 |
52.3914839210465 |
13.3744292542214 |
| ENSMUSG00000029206 |
70918 |
Nsun7 |
protein_coding |
Q14AW5 |
FUNCTION: May have S-adenosyl-L-methionine-dependent methyl-transferase activity. {ECO:0000305}. |
Alternative splicing;Methyltransferase;Reference proteome;RNA-binding;S-adenosyl-L-methionine;Transferase |
|
This gene encodes a member of the NOL1/NOP2/sun domain RNA methyltransferase family. Mice with a mutation in this gene exhibit male sterility due to impaired sperm motility. Alternative splicing of this gene results in multiple transcript variants. [provided by RefSeq, Nov 2014]. |
mmu:70918; |
methyltransferase activity [GO:0008168]; RNA binding [GO:0003723]; flagellated sperm motility [GO:0030317]; methylation [GO:0032259]; sperm mitochondrion organization [GO:0030382] |
17442852_Sun domain containing protein Nsun7 mutant sperm exhibited depressed progressive motility associated with a rigid flagellar midpiece (but not principal piece) segment |
ENSG00000179299 |
NSUN7 |
85.368144 |
2.4917136274 |
1.317138 |
0.31776408 |
16.585033 |
0.00004651668664002374267794209794502080512756947427988052368164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003005401157373664331427798313001176211400888860225677490234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
118.773185805097 |
26.3726950341709 |
47.6672698247751 |
8.06067943434373 |
| ENSMUSG00000029231 |
18595 |
Pdgfra |
protein_coding |
P26618 |
FUNCTION: Tyrosine-protein kinase that acts as a cell-surface receptor for PDGFA, PDGFB and PDGFC and plays an essential role in the regulation of embryonic development, cell proliferation, survival and chemotaxis. Depending on the context, promotes or inhibits cell proliferation and cell migration. Plays an important role in the differentiation of bone marrow-derived mesenchymal stem cells. Required for normal skeleton development and cephalic closure during embryonic development. Required for normal development of the mucosa lining the gastrointestinal tract, and for recruitment of mesenchymal cells and normal development of intestinal villi. Plays a role in cell migration and chemotaxis in wound healing. Plays a role in platelet activation, secretion of agonists from platelet granules, and in thrombin-induced platelet aggregation. Binding of its cognate ligands - homodimeric PDGFA, homodimeric PDGFB, heterodimers formed by PDGFA and PDGFB or homodimeric PDGFC -leads to the activation of several signaling cascades; the response depends on the nature of the bound ligand and is modulated by the formation of heterodimers between PDGFRA and PDGFRB. Phosphorylates PIK3R1, PLCG1, and PTPN11. Activation of PLCG1 leads to the production of the cellular signaling molecules diacylglycerol and inositol 1,4,5-trisphosphate, mobilization of cytosolic Ca(2+) and the activation of protein kinase C. Phosphorylates PIK3R1, the regulatory subunit of phosphatidylinositol 3-kinase, and thereby mediates activation of the AKT1 signaling pathway. Mediates activation of HRAS and of the MAP kinases MAPK1/ERK2 and/or MAPK3/ERK1. Promotes activation of STAT family members STAT1, STAT3 and STAT5A and/or STAT5B. Receptor signaling is down-regulated by protein phosphatases that dephosphorylate the receptor and its down-stream effectors, and by rapid internalization of the activated receptor. {ECO:0000269|PubMed:10903171, ECO:0000269|PubMed:19030102, ECO:0000269|PubMed:20110689, ECO:0000269|PubMed:9226440}. |
Alternative splicing;ATP-binding;Cell membrane;Cell projection;Chemotaxis;Developmental protein;Disulfide bond;Glycoprotein;Golgi apparatus;Immunoglobulin domain;Kinase;Membrane;Nucleotide-binding;Phosphoprotein;Proto-oncogene;Receptor;Reference proteome;Repeat;Signal;Transferase;Transmembrane;Transmembrane helix;Tyrosine-protein kinase;Ubl conjugation |
|
This gene encodes a member of the receptor tyrosine kinase family of proteins. Binding of platelet-derived growth factor protein ligands to this receptor triggers receptor dimerization and autophosphorylation, resulting in the activation of several downstream signaling pathways. Signaling through the encoded receptor plays a role in gastrulation and the development of nearly all organ systems. Mice lacking a functional copy of this gene reportedly exhibit defects in lung, skeleton, testis and the central nervous system, and die soon after birth. Alternative splicing and intronic polyadenylation of gene transcripts have been implicated in muscle regeneration and fibrosis in adult mice. [provided by RefSeq, Jan 2017]. |
mmu:18595; |
axon [GO:0030424]; cell junction [GO:0030054]; cell surface [GO:0009986]; cilium [GO:0005929]; cytoplasm [GO:0005737]; external side of plasma membrane [GO:0009897]; Golgi apparatus [GO:0005794]; microvillus [GO:0005902]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; receptor complex [GO:0043235]; ATP binding [GO:0005524]; BBSome binding [GO:0062063]; growth factor binding [GO:0019838]; phosphatidylinositol 3-kinase binding [GO:0043548]; platelet-derived growth factor alpha-receptor activity [GO:0005018]; platelet-derived growth factor binding [GO:0048407]; platelet-derived growth factor receptor binding [GO:0005161]; protein homodimerization activity [GO:0042803]; protein kinase activity [GO:0004672]; transmembrane receptor protein tyrosine kinase activity [GO:0004714]; vascular endothelial growth factor binding [GO:0038085]; vascular endothelial growth factor receptor activity [GO:0005021]; adrenal gland development [GO:0030325]; anatomical structure morphogenesis [GO:0009653]; animal organ morphogenesis [GO:0009887]; cardiac myofibril assembly [GO:0055003]; cell chemotaxis [GO:0060326]; cell migration [GO:0016477]; cellular response to amino acid stimulus [GO:0071230]; cellular response to reactive oxygen species [GO:0034614]; embryonic cranial skeleton morphogenesis [GO:0048701]; embryonic digestive tract morphogenesis [GO:0048557]; estrogen metabolic process [GO:0008210]; extracellular matrix organization [GO:0030198]; face morphogenesis [GO:0060325]; female gonad development [GO:0008585]; hematopoietic progenitor cell differentiation [GO:0002244]; in utero embryonic development [GO:0001701]; Leydig cell differentiation [GO:0033327]; lung development [GO:0030324]; lung growth [GO:0060437]; luteinization [GO:0001553]; male genitalia development [GO:0030539]; metanephric glomerular capillary formation [GO:0072277]; negative regulation of platelet activation [GO:0010544]; odontogenesis of dentin-containing tooth [GO:0042475]; peptidyl-tyrosine phosphorylation [GO:0018108]; phosphatidylinositol-mediated signaling [GO:0048015]; platelet aggregation [GO:0070527]; platelet-derived growth factor receptor signaling pathway [GO:0048008]; platelet-derived growth factor receptor-alpha signaling pathway [GO:0035790]; positive regulation of branching involved in lung morphogenesis [GO:0061047]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cell proliferation by VEGF-activated platelet derived growth factor receptor signaling pathway [GO:0038091]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of fibroblast proliferation [GO:0048146]; positive regulation of kinase activity [GO:0033674]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of phosphatidylinositol 3-kinase activity [GO:0043552]; positive regulation of phospholipase C activity [GO:0010863]; protein autophosphorylation [GO:0046777]; regulation of chemotaxis [GO:0050920]; regulation of mesenchymal stem cell differentiation [GO:2000739]; retina vasculature development in camera-type eye [GO:0061298]; roof of mouth development [GO:0060021]; signal transduction involved in regulation of gene expression [GO:0023019]; skeletal system morphogenesis [GO:0048705]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169]; white fat cell differentiation [GO:0050872]; wound healing [GO:0042060] |
11803579_PDGF-A/PDGF alpha-receptor signaling required for lung growth and the formation of alveoli but not for early branching of the lung epithelium 12490557_PDGFRalpha plays an essential, cell-autonomous role in the development of cardiac and cephalic neural crest cells. 12748302_Data examine the importance of signal regulation during development by using a mouse platelet-derived growth factor alpha receptor fused to the cytosolic domain of Drosophila Torso or the mouse fibroblast growth factor receptor 1. 14506245_elucidation of molecular basis for differential regulation of the cell cycle and transformation by alpha-PDGFR 14644164_internalization rate of alphaPDGFR in resting cells is regulated by c-src tyrosine kinase 14993293_although both c-Abl and Arg kinases are activated and form complexes with proteins in the PDGFR signaling pathway, only c-Abl functions downstream of PLC-gamma1 to mediate chemotaxis 15059936_The PDGFRalpha-p70S6K pathway is an essential regulator for FGF-2-mediated therapeutic neovascularization & for the host-derived vasculature but tumor angiogenesis, via controlling continuity of expression of multiple angiogenic growth factors. 15221957_Ba/F3 cells expressing mutant PDGFRA are a good model to investigate the mechanism of cell proliferation or growth inhibition by imatinib in mutant PDGFRA-driven cells. 15229650_VEGF-null cells require Pdgfra signaling-mediated stromal fibroblast recruitment for tumorigenesis. 15361870_both PDGF-A and PDGF-C signal through PDGFR-alpha to regulate the development of craniofacial structures, the neural tube and mesodermal organs 15543606_PDGFRalpha and PDGFA are critical regulators for the continued epithelial-mesenchymal interaction during tooth and palate morphogenesis 15708558_TGF-beta regulates PDGFR alpha in the mammary stroma via a c-Cbl-independent mechanism 15811134_Gli1 is not required to induce Pdgfra expression during embryonic bone development 15837572_In response to demyelination, oligodendrocyte progenitor amplification is significantly reduced and subsequent oligodendroglial repopulation of lesions is impaired in PDGFalpha receptor-deficient mice. 15932761_the complete PDGF-A/PDGFR-alpha signaling system is present in murine reproductive tissues, but the distribution of the long and short isoforms of PDFG-A differs 16243034_Ciliary PDGFR-alpha-alpha (homodimer of PDGFRa) signaling is linked to tissue homeostasis and to mitogenic signaling pathways. 16354679_The expression of PDGFRalpha by Galpha12QL is dependent on the small GTPase Rho. 16690743_FIP1L1-PDGFRalpha activation requires disruption of the juxtamembrane domain of PDGFRalpha and is FIP1L1-independent 16741576_Results suggest that autocrine platelet-derived growth factor receptor (PDGFR)-alpha and -beta signaling plays an essential role during cancer progression. 16846854_PDGFR alpha signaling occurs early in the adult stem cell lineage and may help regulate the balance between oligodendrocyte and neuron production. 16971512_Loss of TC-PTP specifically redirects the PDGF beta-receptor toward rapid recycling, which is the first evidence of differential trafficking of PDGF receptor family members. 17499702_PDGF receptor signaling is required for sufficient recruitment of cardiac NCCs into the conotruncal region and for formation of the aortico-pulmonary and ventricular septum 17568391_homozygous null mouse for the Pdgfralpha gene has posterolateral diaphragmatic defects 18038259_PDGFRalpha and JAK3 have roles in Galpha(12)-mediated cell proliferation 18070887_Phosphatidylinositol 3-kinase-dependent membrane recruitment of Rac-1 and p47phox is critical for alpha-platelet-derived growth factor receptor-induced production of reactive oxygen species. 18184726_Sox9 and Sox10 are required in a functionally redundant manner in oligodendrocyte precursors for PDGF-dependent survival and migration. 18192285_These results indicate that PDGFRalpha downstream effectors, especially PI3K, are essential for cell migration of a somite-derived dorsal mesenchyme and disruption of receptor signaling in these cells leads to spina bifida. 18323664_determined the nucleotide sequence of the mRNA and found that the expression of this transcript was regulated by E2F-1 through an E2F-1-responsive sequence located +711/+718 downstream of the transcription start site of the type I transcript. 18606782_Data demonstrate that PDGF receptors alpha and beta cooperate in the yolk sac mesothelium to direct blood vessel maturation and suggest that these effects are independent of their role in vascular smooth muscle cell development. 18725515_Pdgfra is an early-expressed protein that is also a marker of the late primitive endoderm lineage. 18833569_PDGF-Ralpha-expressing cells preferentially have characteristics of myofibroblasts and were more likely to contain alphaSMA than are alveolar cells that do not express PDGF-Ralpha 19015638_ILEI requires cooperation with oncogenic Ras to govern hepatocellular EMT through mechanisms involving PDGF-R/beta-catenin and PDGF-R/Stat3 signaling. 19047372_PDGFRalpha signaling stimulates osteogenesis of neural crest cells-derived osteoblasts by activating the PLC-gamma pathway, suggesting an involvement of this pathway in the etiology of human craniosynostosis. 19126548_a novel mechanism for activation of PDGFRalpha that appears capable of enhancing the responsiveness of cells to growth factors outside of the PDGF family. 19217431_PDGFRalpha has a role in normal connective tissue development and homeostasis and in signaling in systemic fibrosis diseases 19280210_PDGFRalpha-ip cells are basic cellular elements throughout the gastrointestinal musculature and are involved in the gastrointestinal functions. 19334281_misexpression of PDGFRalpha in smooth muscle cells (SMC)causes failure of vascular SMC recruitment to the aorta. 19633290_MMP-13 induction by mechanical strain requires the transactivation of PDGFR-alpha by PKC-delta and the cross-talk between PDGFR-alpha/PI3K/Akt and MEK/ERK pathways. 19939260_During both septal eruption (P4) and elongation (P12), alveolar PDGF-Ralpha may enhance the propensity of fibroblasts to transdifferentiate. 20071345_these data from human genetics and animal models support a role for PDGF-signaling in normal PV development, and in the pathogenesis of TAPVR. 20621681_Kit(low)Cd44(+)Cd34(+) progenitors are true stem cells for normal and hyperplastic ICC and give rise to gastrointestinal neoplasms resistant to Kit/Pdgfra inhibitors. 20658695_role for Pdgfralpha in transduction pathways that lead to repression of Nkx2.5 and WT1 during development of posterior heart field-derived cardiac structures. 20826533_PDGFRalpha signaling is essential for the establishment, and plays a role in the proliferation, of extra-embryonic endoderm cells, which are isolated from mouse blastocyst stage embryos and represent the primitive endoderm lineage. 21181412_PDGFR signaling plays a time-specific and crucial role in the postnatal development of lens in the mouse. 21393858_in glioblastomas with Ink4a/Arf deficiency, overexpressed PDGFRalpha promotes tumorigenesis through the PI3K/AKT/mTOR-mediated pathway regulated by SHP-2 activity 21464317_PDGFRalpha-positive cells in bone marrow are mobilized by high mobility group box 1 (HMGB1) to regenerate injured epithelia. 21659960_Fibroblasts expressing PDGF-receptor-alpha diminish during alveolar septal thinning in mice. 21685324_Platelet-derived growth factor receptor alpha (PDGFRalpha) and thrombomodulin identify a population of fibroblastic reticular cells in lymph nodes where chemokine secretion is controlled by junction adhesion molecule 3 protein (JAM-C). 21911838_Data suggest that Flk-1(+)/PDGFRalpha(+) primitive mesoderm is committed into Flk-1(+)/PDGFRalpha(-) vascular mesoderm through Etv2/ER71. 21993628_inactivation of the Pdgfra gene in beta-cells prevented mouse neonatal beta-cell expansion and adult beta-cell regeneration 22151424_Platelet-derived growth factor receptor-alpha(+) cells had a spindle shape or stellate morphology and often possessed multiple processes that contacted one another forming a loose network 22265737_These data support the hypothesis that Mixl1 directly regulates Pdgfralpha and Flk1 gene expression. 22293178_Heparan sulfate sulfatase SULF2 regulates PDGFRalpha signaling and growth in human and mouse malignant glioma 22322974_These findings demonstrate that transcriptional repression of PDGF-R-alpha after fibroblast injury involves paracrine activity of endogenous TNF-alpha, the formation of a c-Fos-YY1 complex, and negative regulatory activity by HDAC. 22652199_PDGFR-alpha expression changes dramatically following lung volume reduction and a subpopulation of PDGF-alpha expressing fibroblasts differentiate into myofibroblasts. 22730381_Phf14 acts as a negative regulator of PDGFRalpha expression in mesenchymal cells undergoing normal and abnormal proliferation, and is a potential target for new treatments of lung fibrosis. 22960170_These results suggest that an adaptive Src-Pdgfra-Raf-Mapk axis is relevant to PDGFRA inhibition in rhabdomyosarcoma. 23264740_PDGFRalpha signaling in the primary cilium regulates NHE1-dependent fibroblast migration via coordinated differential activity of MEK1/2-ERK1/2-p90RSK and AKT signaling pathways. 23335233_PDGFRalpha+ mesoderm is functionally significant in vascular development and hematopoiesis from phenotype analysis of genetically modified embryos. 23529017_Temporal activation of PDGFRalpha in liver development is important in hepatic morphogenesis. 23776077_the expression of PDGFRalpha and CD51 among bone marrow stromal cells characterizes a large fraction of Nestin cells, containing most fibroblastic CFUs, mesenspheres, and self-renewal capacity after transplantation 23928059_PDGFRalpha and IGF-1R are dynamically regulated and distributed in nucleus and cytoplasm in alveolar Rhabdomyosarcoma. 24000064_Canonical Wnt-, Hh-, and Fgfr1/Fgfr2-signalling are dispensable for epicardial development, but Pdgfra-signalling is crucial for the differentiation of cardiac fibroblasts from epicardium-derived cells. 24047695_The HDAC11 transcript is a PDGF target; the HDAC11 mRNA abundance correlates inversely with proliferative status. 24086166_We thus establish PDGFRalpha as a novel regulator of MNP development and elucidate the roles of its downstream signaling pathways at cellular and molecular levels. 24361597_TNF-alpha-induced MMP-9 expression is mediated through a c-Src-dependent PDGFR transactivation. 24396055_purines activate SK currents via mainly P2Y1 receptors in PDGFRalpha(+) cells 24449836_Nkx2.2 functions as a major 'switch' to turn off Pdgfra signaling in oligodendrocyte precursor cells and initiate the intrinsic program for oligodendrocyte differentiation. 24618081_SHP2 is required for cell transformation and ERK activation by mutant PDGF receptors. 24667490_Pdgfralpha expression is important in the fibrotic response to liver injury 24763086_Identification of a PEDF signaling mechanism involving PPARG induction, suppression of Wnt/beta-catenin signaling and down-regulation of the PDGFRA/B. 25055825_The responses of colonic muscle PDGFRalpha(+) cells and smooth muscle cells to purines suggest that SMCs are unlikely targets for purinergic neurotransmission in colonic muscles. 25470749_Data indicate that PDGF-AA promotes mesenchymal stem cel migration via bone morphogenetic protein 2 (BMP2)-smad proteins smad1/5/8 activation requires lysosome-mediated degradation of PDGF alpha Receptor (PDGFRalpha). 25605750_Multipotential Neural precursors deficient in arylsulfatase A show a higher ratio of long versus short fatty acid sulfatides, reduction in PDGFRa, decreased AKT phosphorylation, and increased exosomal shedding of PDGFRa. 25708924_PDGF signalling in the dermis and in dermal condensates is dispensable for hair follicle induction and formation. 25745058_PDGFR is a key contributor to pulmonary vascular remodeling in experimental pulmonary hypertension. 25865047_In KIT-mutant GIST, inhibition of PDGFRA disrupts a KIT-ERK-ETV1-KIT signaling loop by inhibiting ERK activation. 25940087_Ganglioside GD3 enhances invasiveness of gliomas by forming a complex with PDGFRalpha and Yes Kinase. 25980517_PDGFRalpha has a role in demarcating the cardiogenic clonogenic Sca1+ stem/progenitor cell in adult murine myocardium 26019175_PDGFRalpha targets progenitor cell plasticity as a profibrotic mechanism. 26049020_Primary high invasive Glioma initiating cells SU3 contribute to transformation of adjacent normal host glial cells in local tumor microenvironment possibly via PDGF/PDGFR signaling activation. 26250625_PDGFR alpha has an important role in cranial neural crest cell mitosis within the mandibular processes 26320158_The present study provides new insights into the time-dependent commitment of the PDGFRalpha(+) cell lineage to lipofibroblasts and myofibroblasts during late lung development 26414960_demonstrated that PDGFRalpha signaling promotes alveolar septation by regulating fibroblast activation and matrix fibroblast differentiation, whereas myofibroblast differentiation was largely PDGFRalpha independent 26435273_transient middle cerebral-arterial occlusion (MCAO) was introduced into the mice with conditional Pdgfrb-gene inactivation, including N-PRbeta-KO mice where the Pdgfrb-gene was mostly inactivated in the brain except that in vascular pericytes 26639755_PDGFRalpha may be a relevant target to regulate connective tissue remodeling. 27626655_OLIG2 modulates growth factor signaling in two distinct populations of glioma stem cells, characterized by expression of either the epidermal growth factor receptor (EGFR) or platelet-derived growth factor receptor alpha. 27856617_PDGFRalpha and PDGFRbeta are coexpressed in the craniofacial mesenchyme of mid-gestation mouse embryos and that ablation of Pdgfrb in the neural crest lineage results in increased nasal septum width, delayed palatal shelf development, and subepidermal blebbing. 27894125_polyadenylation of a PDGFRalpha intron in resident stem cells attenuates muscle fibrosis 28049691_By analyzing mice with juxtamembrane or kinase domain point mutations that increase PDGFRalpha activity (V561D or D842V), it was found that PDGFRalpha activation inhibits embryonic white adipose tissue organogenesis in a tissue-autonomous manner. 28089671_PDGFRalpha(+) cells represent the in vitro counterpart of in vivo PrE precursors, and their selection from cultured mESCs yields pure PrE precursors. 28129672_The results from this study indicate that PDGF signaling is required for fiber hypertrophy, extracellular matrix production, and angiogenesis that occur during muscle growth. 28215843_in addition to representing a white adipose tisseu (WAT) adipogenic niche, different PDGFRalpha(+) cell subsets modulate obesity-induced WAT fibrogenesis and are associated with loss of metabolic fitness. 28379539_FOXA3 is a marker of the Sertoli cell lineage and of the adult Leydig cell population, and is a regulator of Pdgfra transcription in Leydig cells. 28408205_the spatial location, mRNA expression and Immunophenotyping of PDGFRalpha(+) fibroblasts during sacculation and alveolarization, is reported. 28487538_PDGFR activity is evenly distributed at different membrane microdomains, while integrin-mediated signaling events have inhibitory effects on the activation of PDGFR specifically located in lipid rafts but not outside rafts. 28501760_our data establish that aberrant stromal PDGFRalpha signaling disrupts extracellular matrix homeostasis during mammary gland development, resulting in increased mammary stiffness and increased potential for tumor growth. 28714851_loss of Pdgfra in endothelial-derived mesenchyme in the outflow tract endocardial cushions leads to a secondary defect in neural crest migration during development. 28775097_Spreading of PDGFR-alpha-deficient lung fibroblasts was insensitive to increased rigidity, and their migration was not reduced by Rac1-guanine exchange factor (GEF)-inhibition. PDGFR-alpha-expressing fibroblasts migrated toward stiffer regions within two-dimensional substrates by increasing migrational persistence (durotaxis). 28848093_The diabetes-induced increase in PDGFRalpha+ cells may be mediated by FOXO3 up-regulation via the inhibition of the PI3K/Akt signaling pathway in STZ-induced diabetic mice. 28919262_PDGFRA is not essential for the derivation and maintenance of extraembryonic endoderm stem (XEN) cell lines . 28934221_Conditional knockout of Pdgfra in Pdgfra-expressing tissues in mouse embryos at different embryonic days (E9.5 and E10.5) resulted in multiple developmental anomalies of the frontonasal region, the cranium and the abdominal wall musculature. Furthermore, the day at which the Pdgfra is deleted influences the repertoire of the anomalies of the conditional knockout embryos. 28947535_Constitutive activation of PDGFRalpha leads to expansion of cartilage underlying the coronal sutures, which contribute to suture closure through endochondral ossification, in a process regulated in part by PI3K/AKT signaling. 28947806_These findings demonstrate how Ca(2+) influx through TRPV4 channels can activate SK channels in PDGFRalpha(+) cells and prevent bladder overactivity during filling. 29107533_Histone H3.3K27M and Trp53 loss and PDGFRA overexpression accelerates disease onset and increases tumor invasion. 29158445_PDGFRalpha/PDGFRbeta signaling balance determines progenitor commitment to beige (PDGFRalpha) or white (PDGFRbeta) adipogenesis. 29341790_PDGFR signaling is necessary for postnatal tendon growth and remodeling and MT1-MMP is a critical mediator of tendon fibroblast migration. 29360139_PDGFRalpha enrichment in hepatic stellate cell-derived extracellular vesicles drives liver fibrosis by a SHP2-dependent mechanism 29378823_Investigated the developmental process of mesenchymal stem cells (MSCs) in embryos using the gene Pdgfra as a marker. We traced cells expressing Pdgfra and other genes (brachyury, Sox1 and Pmx1) in various mutant embryos until the adult stage. Embryonic MSCs emerge in waves and almost all adult bone marrow MSCs and white adipose tissue MSCs originate from mesoderm and embryonic Pdgfralpha-positive cells. 30057274_downregulate growth signals provided by PDGFRalpha to initiate oligodendrocyte differentiation and myelination 30082792_STMN1 is a PDGFRalpha phospho-regulated target; its mis-regulation confers sensitivity to vinblastine cytotoxicity in glioblastoma 30121753_-derived growth factor receptor (PDGFR)alpha- and PDGFRbeta- expressing perivascular cell populations were identified as leptin producing within skeletal muscle of mice and humans. Furthermore, in response to 2 weeks of high-fat feeding, PDGFRbeta+ but not PDGFRalpha+ cells increased leptin production. 30178747_These findings revealed complexity in the behavior of the Pdgfra-lineaged cells as exemplified by their distinct contributions to myofibroblasts. 30201265_PDGFRalpha has a role in in pathogenesis, progression and prognosis of NK/T-cell lymphoma 30242047_A subpopulation of oligodendrocyte lineage in the developing cortex is independent of PDGFRalpha signaling. 30246400_miR-218 was demonstrated to exert an inhibitory function and promoted cell migration via targeting PDGFRalpha during cardiomyocyte differentiation from ESCs. 30323271_Conditional deletion of platelet derived growth factor receptor alpha (Pdgfra) in urorectal mesenchyme causes mesenchyme apoptosis and urorectal developmental anomalies in mice. 30485861_PDGFR alpha beta and PDGFR beta beta dimers were strong inducers of random and directionally-persistent migration in mouse skin fibroblast, respectively, that was sustained for up to 24 h. 30509307_PDGFRalpha in hepatocytes plays an important role in liver fibrosis by affecting PDGFRalpha expression in HSCs 30654933_PDGFRA/Pdgfra gene is regulated by at least seven first exons in human and mouse. 31125253_PDGFRalpha signaling is essential for cardiac fibroblast maintenance. 31992125_Autophagy inhibition blunts PDGFRA adipose progenitors' cell-autonomous fibrogenic response to high-fat diet. 32229310_Mosaic Mutant Analysis Identifies PDGFRalpha/PDGFRbeta as Negative Regulators of Adipogenesis. 32295715_PARP-1 regulates mouse embryonic neural stem cell proliferation by regulating PDGFRalpha expression. 32442967_Critical role of platelet-derived growth factor-alpha in angiogenesis after indirect bypass in a murine moyamoya disease model. 32531084_Functional heterogeneity of PDGFRalpha (+) cells in spontaneously active urogenital tissues. 32553115_Dynamic control of adipose tissue development and adult tissue homeostasis by platelet-derived growth factor receptor alpha. 32615075_Hepatic Stellate Cell-Specific Platelet-Derived Growth Factor Receptor-alpha Loss Reduces Fibrosis and Promotes Repair after Hepatocellular Injury. 32737331_Cleavage of proteoglycans, plasma proteins and the platelet-derived growth factor receptor in the hemorrhagic process induced by snake venom metalloproteinases. 32800757_Pdgfra regulates multipotent cell differentiation towards chondrocytes via inhibiting Wnt9a/beta-catenin pathway during chondrocranial cartilage development. 32918673_Vascular PDGFR-alpha protects against BBB dysfunction after stroke in mice. 33001865_PDGFRA in vascular adventitial MSCs promotes neointima formation in arteriovenous fistula in chronic kidney disease. 33168840_Genetic analyses in mouse fibroblast and melanoma cells demonstrate novel roles for PDGF-AB ligand and PDGF receptor alpha. 33174445_Neuropilin-1 directs PDGFRalpha-entry into lung fibroblasts and signaling from very early endosomes. 33259802_Histone H3.3G34-Mutant Interneuron Progenitors Co-opt PDGFRA for Gliomagenesis. 33503976_PDGF Receptor Alpha Signaling Is Key for Muller Cell Homeostasis Functions. 33824385_Identification of PDGFRalpha-positive interstitial cells in the distal segment of the murine vas deferens. 33859199_Microglial neuropilin-1 promotes oligodendrocyte expansion during development and remyelination by trans-activating platelet-derived growth factor receptor. 34285913_Lineage Contribution of PDGFRalpha-Expressing Cells in the Developing Mouse Eye. 34381120_Molecular and functional characterization of detrusor PDGFRalpha positive cells in spinal cord injury-induced detrusor overactivity. 34480532_Prenatal overexpression of platelet-derived growth factor receptor A results in central nervous system hypomyelination. 34586354_PDGF-AA promotes cell-to-cell communication in osteocytes through PI3K/Akt signaling pathway. 34708880_PDGFRalpha-lineage origin directs monocytes to trafficking proficiency to support peripheral immunity. 34714716_Sprouty1 regulates gonadal white adipose tissue growth through a PDGFRalpha/beta-Akt pathway. 35348002_Platelet-Derived Growth Factor Receptor Type alpha Activation Drives Pulmonary Vascular Remodeling Via Progenitor Cell Proliferation and Induces Pulmonary Hypertension. 35523143_PDGFRalpha-induced stromal maturation is required to restrain postnatal intestinal epithelial stemness and promote defense mechanisms. 35876806_Severe cerebellar malformations in mutant mice demonstrate a role for PDGF-C/PDGFRalpha signalling in cerebellar development. 35977484_Temporal control of PDGFRalpha regulates the fibroblast-to-myofibroblast transition in wound healing. |
ENSG00000134853 |
PDGFRA |
2704.798640 |
0.4802633500 |
-1.058102 |
0.32259420 |
10.494022 |
0.00119761363848281147601038121308647532714530825614929199218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0054291818277887454735619598977791611105203628540039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1736.43185286913 |
391.51897652055 |
3615.58268591849 |
607.455821818156 |
| ENSMUSG00000029304 |
20750 |
Spp1 |
protein_coding |
P10923 |
FUNCTION: Major non-collagenous bone protein that binds tightly to hydroxyapatite. Appears to form an integral part of the mineralized matrix. Probably important to cell-matrix interaction. {ECO:0000250|UniProtKB:P31096}.; FUNCTION: Acts as a cytokine involved in enhancing production of interferon-gamma and interleukin-12 and reducing production of interleukin-10 and is essential in the pathway that leads to type I immunity. {ECO:0000269|PubMed:10657301, ECO:0000269|PubMed:2351930}. |
Biomineralization;Cell adhesion;Cytokine;Direct protein sequencing;Glycoprotein;Phosphoprotein;Reference proteome;Secreted;Sialic acid;Signal |
|
|
mmu:20750; |
apical part of cell [GO:0045177]; cell projection [GO:0042995]; cytoplasm [GO:0005737]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; perinuclear region of cytoplasm [GO:0048471]; vesicle [GO:0031982]; cytokine activity [GO:0005125]; extracellular matrix binding [GO:0050840]; integrin binding [GO:0005178]; ion binding [GO:0043167]; small molecule binding [GO:0036094]; androgen catabolic process [GO:0006710]; bone mineralization [GO:0030282]; calcium ion homeostasis [GO:0055074]; cell adhesion [GO:0007155]; cellular calcium ion homeostasis [GO:0006874]; cellular chloride ion homeostasis [GO:0030644]; cellular phosphate ion homeostasis [GO:0030643]; cellular response to fluid shear stress [GO:0071498]; cellular response to leukemia inhibitory factor [GO:1990830]; cellular response to testosterone stimulus [GO:0071394]; cellular sodium ion homeostasis [GO:0006883]; collecting duct development [GO:0072044]; negative regulation of apoptotic process [GO:0043066]; neutrophil chemotaxis [GO:0030593]; osteoblast differentiation [GO:0001649]; positive regulation of bone resorption [GO:0045780]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of estradiol secretion [GO:2000866]; response to organic substance [GO:0010033]; response to steroid hormone [GO:0048545]; response to vitamin D [GO:0033280]; urate biosynthetic process [GO:0034418] |
11721059_Osteopontin-deficient mice were resistant to progressive EAE and had frequent remissions, and myelin-reactive T cells in OPN-/- mice produced more interleukin 10 and less interferon-gamma than in OPN+/+ mice 11807984_The osteopontin gene, which has a role in immunosurveillance, is a direct target of TP53. 11854223_Osteopontin is involved in the development of Lyme arthritis. 11859094_Attenuated experimental autoimmune encephalomyelitis in osteopontin knockout mice 11891192_osteopontin is dramatically induced in the desmin-null myocardium 11918223_evidence that OPN is required for unloading-induced three-dimensional bone loss 11920639_osteopontin induces neovascularization in mice 12009309_TPA-induced steady-state osteopontin mRNA expression in mouse JB6 cells involves the activation of MAPK mediated through PKC epsilon and/or PKC delta. 12114325_Expression of osteopontin induces both medial thickening without injury and neointimal formation after injury, thus suggesting that OPN plays a role in the development of atherosclerosis, vascular remodeling, and restenosis after angioplasty in vivo. 12162503_role of colocalization with CD44 in migration, cell fusion, and resorption in osteoclasts 12200434_OPN gene transcription is regulated by upstream stimulatory factor and activator protein-1 in aortic vascular smooth muscle cells, entrained to changes in cellular glucose metabolism 12210726_osteopontin upregulation by okadaic acid requires oncoprotein AP-1 induction in mouse cells 12377945_interaction between the C-terminal domain of osteopontin and the receptor CD44 induces macrophage chemotaxis; and engagement of beta(3)-integrin receptors by a nonoverlapping N-terminal osteopontin domain induces cell spreading and subsequent activation 12391016_role in vascular calcification 12466120_studies indicate that OPN is a potent inhibitor of ectopic calcification, and suggest a novel function for OPN in controlling mineral-dissolving inflammatory cell function at sites of ectopic calcification 12473670_curcumin potently suppresses osteopontin-induced cell migration, tumor growth, and NF kappa B-mediated pro-MMP-2 activation by blocking the IKK/I kappa B alpha signaling pathways 12506146_OPN-deficient mice demonstrated significant intratubular deposits of CaOx crystals. OPN plays critical renoprotective role in vivo as inhibitor of CaOx crystal formation and retention in renal tubules. 12631119_Favors macrophage recruitment to the postischemic kidney, inhibits apoptosis, and stimulates the development of renal fibrosis after an acute ischemic insult. 12714436_Endogenous OPN regulates normal vascular physiology and contributes to the vascular remodeling response by regulating vascular compliance and the inflammatory response. 12730087_Osteopontin plays a promoting effect in atherosclerosis and inhibitory effect in vascular calcification. 12788230_OPN expression is inhibited in a time-dependent manner by a deficiency in Ca(+2) or PO(4)(3-). 12837285_Opn was expressed in tendon cell clones. 12847283_OPN up-regulation by fibroblast growth factor-2 is a monocyte-mediated mechanism of amplification of growth factor-induced neovascularization during inflammation, wound healing, and tumor growth. 12893437_these results strongly imply that OPN plays an important role in tumor growth through the enhancement of angiogenesis in vivo. 12915117_Results suggest that osteopontin activity contributes to hepatocyte growth factor-induced tumor growth and invasiveness. 12920127_the ability of increased inorganic phosphate to stimulate three specific signaling pathways in the regulation of osteopontin, resulting in functionally significant changes in Opn gene expression. 12943672_OPN inhibits cytochrome c oxidase expression as the result of a novel CD44-dependent transcriptional regulatory mechanism of the mitochondrial H strand 12944477_Akt kinase activates two distinct genetic programs: the program of growth and survival, which is independent of osteopontin expression, and the program of invasiveness and anchorage independence, which is mediated by osteopontin. 12968681_Osteoblasts forming bone within cranial sutures subjected to tensile stress exhibited high levels of OPN protein expression. 14506237_expression is regulated by Runx2 gene product in mammary epithelial cells 14584055_Data show that cell surface CD44 is critical for chemotaxis by peritoneal macrophages and suggest that a novel intracellular form of osteopontin may modulate CD44 activities involved in these processes. 14711899_role of OPN in the maintenance of gestation 14722087_transcription and promoter activity are significantly up-regulated in response to nitric oxide in a system of endotoxin-stimulated murine macrophages 14746905_In demyelinating disease compared to controls intense osteopontin immunoreactivity was observed in cell bodies, and extracellular space of demyelinating lesions. Glial cells, were also immunopositive for osteopontin. 14977630_osteopontin expression is associated with important fibrogenic signals in the lung and that the epithelium may be an important source of osteopontin during lung fibrosis. 14988426_Nurr1 activates the OPN promoter directly in osteoblastic cells and may have a role in the regulation of bone homeostasis 15033472_OPN acts as a positive regulator in the osteoclastogenesis of arthritis through the RANK/RANKL/OPG system 15044174_Upregulation of OPN expression early in development of steatohepatitis. Possible important role for OPN in signaling onset of liver injury and fibrosis in experimental nonalcoholic steatohepatitis. 15051510_These data support the hypothesis that host-derived osteopontin may inhibit tumor growth and provide a mechanism for this effect. 15080898_Recombinant Osteopontin treatment stimulated both myelin basic protein (MBP) synthesis and myelin sheath formation in mixed cortical cultures from embryonic mouse brain, an in vitro primary culture model of myelination. 15084239_OPN is an essential regulator of the metastatic phenotype seen in polyomavirus middle T-induced mammary tumors. Yet OPN expression alone is not sufficient to cause metastasis. 15121011_role as is a negative regulator of proliferation and differentiation in MC3T3-E1 cells 15123578_OPN has a pivotal role in the development of Ang II-induced cardiac fibrosis & remodeling. The effect of eplerenone on the prevention of cardiac fibrosis, but not cardiac hypertrophy, might be partially mediated through the inhibition of OPN expression. 15165989_increased expression of OPN in the myocardium after reperfusion may promote fibrosis by protecting cardiac fibroblasts from cell death. 15218333_Role of OPN in the process of remodeling, inflammation and lipid metabolism. 15256362_Important role for leptin receptors and osteopontin in the pathogenesis of nonalcoholic steatohepatitis. 15347645_OPN transcriptional regulation is a key mediator of metastatic behavior in malignancy 15479859_osteopontin promoter polymorphisms affect its transcriptional activity 15516973_OPN knockdown affected the downstream expression of 160 '2nd tier' genes, and at least a subset of these genes appears to be involved in malignant transformation 15534078_Increased osteopontin expression may play an essential role in modulating compensatory cardiac hypertrophy in response to chronic pressure overload. 15592854_In mouse spinal cord injury lead the up-regulation osteopontin expression at 7 days and osteopontin expression was observed in activated macrophages/microglia at 3 and 7 days. 15731245_Rotavirus infection intestine caused marked increases in OPN mRNA levels and secreted OPN protein; OPN-deficient mice suffered prolonged disease 15735673_dioxin/aryl hydrocarbon receptor mediates downregulation of osteopontin gene expression in a mouse model of gastric tumourigenesis 15750688_investigated the expression of OPN mRNA in odontclasts during experimental tooth movement; OPN may play an important role in bone resorption triggered by mechanical stress 15845900_Opn is an important component of the hematopoietic stem cell (HSC) niche which participates in HSC location and as a physiologic-negative regulator of HSC proliferation. 15867270_Genistein had a dose-dependent, significant inhibitory effect on OPN transcript levels in prostates displaying advanced prostate cancer. 15937924_OPN is detected in Sertoli cell cytoplasm and luminal cells of developing mesonephric tubules, with small amounts associated with the plasma membrane of germ cells. 15949549_OPN has a role in foreign body giant cell formation in response to implantation 15970437_the 9.0 kb region of the bone sialoprotein promoter are required for strong, tissue- and differentiation-specific expression of bone sialoprotein. 15993098_Overall these results show that OPN is deregulated by BCR-ABL oncogene and suggest that OPN could be involved in CML stem cell biology. 16000302_Data show that RUNX2 is a direct regulator of bone sialoprotein in osteoblasts and that it functions in cooperation with DLX5 or a related factor to activate osteoblast-specific gene expression. 16105024_OPN is a major inhibitor of 2,8-dihydroxyadenine crystal deposition and inflammation in male mice and is a major modifier of the inflammatory response but not of crystal deposition in female mice 16195230_findings define novel mechanisms involving the intersection of three pathways, vitamin D receptor Runx2, 1,25-Dihydroxyvitamin D(3), and Notch signaling, that play a major role in the regulation of osteopontin in osteoblastic cells 16204248_TIS7, a negative regulator of transcriptional activity, represses expression of OPN and beta-catenin/Tcf-4 target genes 16221502_The increase in osteopontin expression observed during liver fibrogenesis may play a protective role. 16253490_fibroblast growth factor signaling directly activates osteopontin expression independent of chondrocytes differentiation 16265595_an increase of OPN mRNA and protein expression occurs in cardiovascular tissues of hypothyroid mice 16286640_Findings delineate the genetic basis of Opn expression in T cells and further clarify the role of Opn in the development of T helper type 1 (Th1) and type 1 CD8+ T cells (Tc1). 16412998_Neurons affected by scrpie at an early stage transiently express Opn but subsequently succumb to cell death at a later stage. 16474180_Integrin-linked kinase regulates osteopontin-dependent MMP-2 and uPA expression in metastasis of murine mammary epithelial cancer cells 16518820_No deficiency defect in embryonic pancreatic tissues, so it is possible that other proteins act redundantly to Opn or that this protein's function is dispensable for pancreas development. 16552072_Osteopontin is involved in clearance of malarial parasites through T helper cell type 1 (Th1) immune responses at an early stage of Plasmodium chabaudi chabaudi infection. 16670084_Ets-1 and runx2 regulate osteopontin in murine colorectal cancer cells 16709900_OPN provides a paracrine signal that augments vascular pro-MMP9 activity, mediated in part via superoxide generation and oxylipid formation 16741956_The suppressed TNF-alpha response in osteopontin-null mice was reflected in a marked increase in non-apoptotic cell death in diseased colons. 16889770_These data indicate that OPN acts to mediate mechanical stress signaling upstream to the genes encoding apoptosis-related molecules, and its action is associated with alteration of the genes. 16980342_Thus increased expression of OPN may play a deleterious role during streptozotocin-induced diabetic cardiomyopathy with effects on cardiac fibrosis, hypertrophy, and myocyte apoptosis. 17003824_Osteopontin plays a significant role in the recruitment and activation of interstitial fibroblasts to myofibroblasts in the progression of interstitial fibrosis in the developing hydronephrotic kidney. However, OPN also suppresses apoptosis. 17045020_Osteopontin can protect against hyperoxia-induced ALI by promoting the expression of TIMP and inhibiting the activation of MMP. 17055235_OPN plays a crucial role in conveying the effect of denervation-induced mechanical stress deprivation to the tendon fibroblasts to degrade the extracellular matrices by regulating MMP-13 expression in tendon fibroblasts. 17110428_Presence of cross talk between IL-1beta and OPN pathways and unique trans-regulatory mechanism in which IL-1beta-induced nitric oxide synthesis feedback regulates itself through up-regulation of OPN gene transcription. 17133361_Bcl-2 -/- ureteric bud cells exhibited increased fibronectin expression and decreased thrombospondin-1 and osteopontin expression. 17160024_Strong indications that OPN is the main antiapoptotic factor in mouse breast cancer cells. 17177137_Osteopontn is a known promoter for Th1 cell responses and appears to have regulatory control over Th2 cell responsse in streptozotocin diabetes. 17188882_OPN is involved in Parkinson disease-associated neurodegeneration 17211411_OPN is required for activation of Smad2/3 signal in an injured lens epithelium and lens cell epithelial-mesenchymal transition. 17237438_OPN increases STAT1 ubiquitination and subsequent 26s proteasome-mediated degradation to inhibit STAT1 dependent iNOS promoter activity, transcription, and protein expression. 17301681_osteopontin mRNA expression is increased 1 and 3 weeks following sciatic nerve section (axotomy) 17306792_A constitutive endogenous osteopontin production is important for proper basic functions of macrophages and our study indicates that the constitutive osteopontin production is involved in maintaining macrophages in a differentiated phenotype. 17390343_Results suggest that neutralization of OPN-induced signal might be effective in suppression of tumor metastasis. 17390367_OPN(+/+) bones show a decrease in mechanical properties between young and older age groups. By contrast, OPN(-/-) bones showed no significant change in mechanical properties with aging. 17435770_Therapeutic administration of recombinant Opn during pulmonary secondary antigenic challenge and protect mice from allergic disease 17475887_OPN plays a role in organ-specific experimental autoimmune uveoretinitis (EAU); clinical EAU onset is significantly delayed and its progress is ameliorated in OPN-deficient mice. 17537408_osteopontin promotes angiogenic progenitor cell proliferation through the phosphoinositide 3-kinase/Akt pathway 17613519_that cleavage of bone acidic glycoprotein-75 and bone sialoprotein by an AEBSF-sensitive, osteoblast-derived serine protease is associated with mineral crystal nucleation 17693663_PI3K pathway promotes the transformation of intestinal adenoma to adenocarcinoma. OPN, a downstream effector of PI3K, protects transformed intestinal epithelial cells from programmed cell death and stimulates their anchorage-independent growth. 17785423_Opn regulates hindlimb-unloading-induced lymphoid organ atrophy and weight loss by modulating corticosteroid production. 17823662_Osteopontin may play a key role in linking obesity to the development of insulin resistance by promoting inflammation and the accumulation of macrophages in adipose tissue. 17883724_OPN expression in the lungs is increased in a murine model of allergen-induced chronic airway remodelling, suggesting a role for this cytokine in airway remodelling in asthma. 17894420_These observations indicate that OPN deficiency suppresses specifically tooth root resorption in case of experimental force application. 17898865_OPN regulates Stat1-dependent functions, such as growth inhibition and p21 expression, in murine mammary epithelial tumor cell lines. 17900509_Our results showed that, within glomeruli, OPN expressed restrictedly within EPHL; the OPN mRNA and protein of glomeruli increased on day 11, correlating well with the early EPHL, and following sclerosis and macrophage infiltration. 18006856_(Osteopontin)OPN-/- mice exhibited delayed ischemic limb recovery, which may be attributable to defective response of OPN-/- monocytes/macrophages to chemoattraction 18048491_Osteopontin expression in adipose tissue macrophages was strongly up-regulated by obesity. 18079410_OPN is required for the differentiation and activity of myofibroblasts formed in response to the profibrotic cytokine transforming growth factor-beta1. 18158320_syndecan-4 is a critical intrinsic regulator of inflammatory reactions via its effects on OPN function 18174176_osteopontin (OPN) is highly expressed and secreted by erythroblasts during differentiation 18180311_OPN and PDGF may contribute to inflammation-associated fibrosis 18200503_A mast cell mediator, enhances mast cell responses to antigen, and thus may influence mast cell-related pathological conditions. 18234909_OPN is an ERRalpha target gene whose promoter is regulated by ERRalpha in a cell context-dependent manner. 18246866_Opn is useful to detect inflammatory and degenerative myocardial disorders 18337582_Experiments on the acute immediate effects of MT expression on cellular RNA levels showed that expression of osteopontin (OPN) was strongly induced by MT expression. 18412142_Results indicate that FGF-2 induces unique expression of osteopontin, which may play a role different from the other bone-related proteins during the process of periodontal tissue regeneration by FGF-2. 18417476_OPN also plays a significant role in the regulation of PTH actions. 18443355_Opn plays an important role in the development of albuminuria, possibly by modulating podocyte signaling and motility 18459000_analyses of osteopontin null otoconia show that the protein is not needed for crystal formation, and no evidence of compensatory protein deposition is found 18483788_Bone marrow-derived macrophages have a reduced capacity to differentiate into osteoclasts on a mineralized substrate compared to a polystyrene substrate, independent of OPN genotype. 18490187_These results suggest that myoblasts are an important source of osteopontin in damaged muscle and that osteopontin released by myoblasts may assist in controlling both the myogenic and inflammatory processes during the early stages of muscle regeneration. 18505365_Immunohistochemical staining of OPN showed that the wild type crystals contained OPN protein but not knockout crystals. We concluded that OPN plays a crucial role in the morphological conversion of calcium oxalate crystals to stones in mouse kidneys 18522644_The osteopontin is up-regulated in response to SE in distinct temporal sequences in the hippocampus, specifically in degenerating neurons and axons. 18535152_Osteopontin promotes the development of natural killer cells from hematopoietic stem cells 18539470_OPN is expressed at high levels during remyelination of toxin-induced demyelination. The increased expression is due to mRNA expression in macrophages and follows differences in macrophage responses to demyelination in young and old adult animals. 18547835_Stimulation with EMD significantly (P |
ENSG00000118785 |
SPP1 |
69286.219004 |
0.4435452495 |
-1.172847 |
0.05128500 |
509.231951 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000009317949133742890213827268323178538463103084672553687752498695817817250956377812355102806881695198233036364353792413428667837103039855844113787776944890 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000617091309587703053303219009795381621515466364055845312131660606469349743266009523341090104247835554045428378552116166017959243739698090846618001104409 |
Yes |
No |
42253.6134309414 |
1502.34998776488 |
95263.3659843728 |
2530.54702148076 |
| ENSMUSG00000029306 |
15891 |
Ibsp |
protein_coding |
Q61711 |
FUNCTION: Binds tightly to hydroxyapatite. Appears to form an integral part of the mineralized matrix. Probably important to cell-matrix interaction. Promotes Arg-Gly-Asp-dependent cell attachment (By similarity). {ECO:0000250}. |
Biomineralization;Cell adhesion;Glycoprotein;Phosphoprotein;Reference proteome;Secreted;Sialic acid;Signal;Sulfation |
|
|
mmu:15891; |
extracellular space [GO:0005615]; vesicle [GO:0031982]; integrin binding [GO:0005178]; small molecule binding [GO:0036094]; bone mineralization [GO:0030282]; cell adhesion [GO:0007155]; cellular response to growth factor stimulus [GO:0071363]; extracellular matrix organization [GO:0030198]; positive regulation of cell adhesion [GO:0045785] |
12963984_bone sialoprotein has a role in neoplasm metastasis 15020240_BSP gene transcription is autoregulated in part by a positive feedback on its own promoter. 15937924_Bsp mRNA is expressed in developing testis cords and thus may contribute to testis differentiation 16059638_BSP contributes to RANKL-mediated bone resorption by inducing osteoclastogenesis and osteoclast survival and decreasing osteoclast apoptosis 18547835_Stimulation with EMD significantly (P |
ENSG00000029559 |
IBSP |
64728.100096 |
0.3732572630 |
-1.421758 |
0.04704886 |
881.835311 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000008722689250721506539838078856034184497665579665881976602119519449227107 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001107200022224916467418703791071251961118266495882103424925783979315375 |
Yes |
No |
34878.0326140549 |
1137.63646211861 |
93442.3414303511 |
2274.99028874688 |
| ENSMUSG00000029307 |
13406 |
Dmp1 |
protein_coding |
O55188 |
FUNCTION: May have a dual function during osteoblast differentiation. In the nucleus of undifferentiated osteoblasts, unphosphorylated form acts as a transcriptional component for activation of osteoblast-specific genes like osteocalcin. During the osteoblast to osteocyte transition phase it is phosphorylated and exported into the extracellular matrix, where it regulates nucleation of hydroxyapatite (By similarity). {ECO:0000250}. |
Biomineralization;Cytoplasm;Extracellular matrix;Glycoprotein;Nucleus;Phosphoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:13406; |
cytoplasm [GO:0005737]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; nucleus [GO:0005634]; extracellular matrix binding [GO:0050840]; Hsp70 protein binding [GO:0030544]; biomineral tissue development [GO:0031214]; extracellular matrix organization [GO:0030198]; ossification [GO:0001503]; positive regulation of cell-substrate adhesion [GO:0010811]; regulation of enamel mineralization [GO:0070173] |
12369786_Dmp1 first appeared in hypertrophic cartilage cells, followed by osteoblasts, and later was expressed strongly in osteocytes. Dmp1 may play an important role in bone formation. 12489180_TGF beta-1 downregulates DMP-1 and DSPP in odontoblasts. 12489182_Transcriptional regulation of dentin matrix protein 1 (DMP1) by AP-1 (c-fos/c-jun) factors. 12615915_DMP1 is a unique molecule that initiates osteoblast differentiation by transcription in the nucleus and orchestrates mineralized matrix formation extracellularly, at later stages of osteoblast maturation 12733719_involved in the responses of osteocytes and osteoblasts to mechanical loading of bone 14578349_dentin matrix protein-1 is processed by Bone morphogenetic protein-1/Tolloid-like proteinases 14966118_Dmp-1 null mice postnatally develop a profound tooth phenotype characterized by a partial failure of maturation of predentin into dentin, enlarged pulp chambers, increased width of predentin zone with reduced dentin wall, and hypomineralization 15308641_histone acetyltransferase activity of p300 a critical role in regulating DMP1 gene expression 15316863_DMP1 may have functions other than the regulation of mineralization 15590631_DMP1 is essential for normal postnatal chondrogenesis and subsequent osteogenesis 15728181_8-kb region of the Dentin matrix protein 1 (DMP1) gene is a target for mechanotransduction in osteocytes, and its cis-regulatory activity may be correlated to local strain in bone 15937924_Dmp mRNA is expressed in developing testis cords, and thus may contribute to testis differentiation 16191382_Dmp1 is the key factor in the formation of growth plates and secondary ossification center, and plays an important role in the process of bone and cartilage formation and bone nodule remodeling. 16294270_Decreased mineral-to-matrix ratio and increased crystal size in bones of dmp1 KO mice suggest that DMP1 has multiple roles (both direct and indirect) in the regulation of postnatal mineralization. 16340123_DMP1 is a major regulator of mineralization 16612089_Different transcription factors regulate early or later cis-regulatory domains of the Dmp1 promoter, which gives rise to the unique spatial and temporal expression pattern of Dmp1 gene at different stages of tooth development. 17196192_DMP1 is a key regulator of odontoblast differentiation, formation of the dentin tubular system and mineralization and its expression is required in both early and late odontoblasts for normal odontogenesis to proceed. 18162525_degradation of MEPE and DMP-1 and release of ASARM peptides are chiefly responsible for the HYP mineralization defect and changes in osteoblast-osteoclast differentiation. 18559986_Data suggest that the regulation of extracellular matrix mineralization by DMP1 is coupled to renal phosphate handling and vitamin D metabolism through a DMP1-dependent regulation of FGF23 production by osteocytes. 18573980_Study suggests defective alveolar bone and cementum may account for the periodontal breakdown and increased susceptibility to bacterial infection in Dmp1 null mice. 18698129_The different distribution of DMP1 fragments indicates that these DMP1 variants must perform distinct functions. 18698130_A single substitution blocked the proteolytic processing of mouse DMP1 in HEK-293 cells, indicating that cleavage at the NH(2) terminus of Asp(197) is essential for exposing other cleavage sites for the conversion of DMP1 to its fragments. 18728349_The 57-kDa C-terminal fragment may be able to recapitulate the function of intact DMP1 in vivo. 20499360_mutant mice did not recapture phenotype observed in human patient but displayed a mild rachitic tooth phenotype in comparison with that in the Dmp1-null mice, suggesting that the DI III-like phenotype may be due to an as-yet-undetermined gene modifier 20663874_that the full-length form of DMP1 is an inactive precursor and its proteolytic processing is an activation step essential to the biological functions of this protein in osteogenesis. 20734454_the 57-kDa C-terminal fragment is the functional domain of DMP-1 that controls osteocyte maturation and phosphate metabolism, and affects FGF23 20877695_Data reported circling behavior and hyper reaction to touching in Dmp1 null mice 21079361_distribution of the osteocytic lacunar-canalicular system and osteocyte-secreting molecules--dentin matrix protein 1 and sclerostin--in the epiphyses and cortical bones in osteoprotegerin deficiency 21297011_proteolytic processing of DMP1 is essential to the formation and mineralization of dentin, cementum, and jaw bones 21507898_PHEX and DMP1 control a common pathway regulating bone mineralization and FGF23 production, the latter involving activation of the FGFR signaling in osteocytes. 21555863_These results indicate a potential role of the full-length form of DMP1 in chondrogenesis. 22813642_these findings suggest that, apart from its role as a constituent of dentin and bone matrix, DMP1 might play a regulatory role in the nucleus. 22879941_These findings suggest that DMP1 has an anti-apoptotic role in hyperphosphatemia. 22930691_These findings indicate a functional interaction between PHEX and DMP1 to regulate bone mineralization and circulating FGF23 levels. 23111467_the differential localization patterns of DMP1 fragments indicate that different forms of DMP1 may play distinct roles in the SMGs. 23258378_DMP1 has a protective role for odontoblasts and ameloblasts in a pro-apoptotic environment (a high Pi level). 23349460_Results indicate that dentin sialophosphoprotein (DSPP) is a downstream effector molecule that mediates the roles of dentin matrix protein 1 (DMP1) in dentinogenesis. 23558921_Klf4 was able to specifically bind to the Dmp1 promoter. 24334408_results support the hypothesis that 1,25D regulates DMP-1 expression through a VDR-dependent mechanism, possibly contributing to local changes in bone/tooth mineral homeostasis. 24874551_These results indicated that the downregulation of Dmp1 may not directly associate with, or significantly contribute to the bone and dentin defects in the Fam20C-cKO mice. 24917674_DMP1 continues its expression in osteoblasts during postnatal development and that the deletion of Dmp1 leads to an increase in osteoblast proliferation 24987156_DMP1 may play an important role in maintaining the chondrogenic phenotype and its possible involvement in altered cartilage matrix remodelling and degradation in disease conditions like osteoarthritis. 25105818_data suggest that the full-length DMP1 plays no apparent role in the nucleus during odontogenesis. 25158195_bone-specific Dentin matrix protein 1 overexpression changes the pattern in osteogenic gene expression pattern thereby influencing bone development 25537728_identify alternative splicing as a mechanism utilized by cancer cells to modulate the DMP1 locus through diminishing DMP1alpha tumour suppressor expression 25978185_We conclude that DMP-1 may be involved in regulating the temporal expression at embryonic stages in the mouse tongue 26276370_downregulation by PTH and vitamin D via the cAMP/PKA pathway 26303287_the hypophosphatemia induced osteomalacia phenotype in Dmp1 KO mice is contributed by at least two factors: the low Pi level and the DMP1 local function in mineralization 26310138_Klf10 is involved in tooth development and promotes odontoblastic differentiation via the up-regulation of Dmp1 and Dspp transcription. 26428891_An in situ hybridization study of perlecan, DMP1, and MEPE in developing condylar cartilage of the fetal mouse mandible and limb bud cartilage. 26456319_Data show that the phenotype of Notch activation in osteocytes was prevented in matrix protein 1 (Dmp1)-Cre;Rosa(Notch) mice hemizygous for the Dmp1-sclerostin (SOST) transgene. 26481310_It was concluded that endogenously secreted PTH and GHR signaling in bone are necessary to establish radial bone growth and optimize mineral acquisition during growth. 26595451_These results suggest that the LPV motif is essential for the efficient export of secretory DMP1 from the ER to the Golgi complex. 26634432_These findings indicate that glycosylation of DMP1 is a key posttranslational modification process during development and that DMP1-PG functions as an indispensable proteoglycan in osteogenesis. 26686820_Results show that transgenic expression of Dspp partially rescued the long bone defects of Dmp1-null mice and suggest that DSPP and DMP1 may function synergistically within the complex milieu of bone matrices. 27143110_The collective results indicate that the osteoanabolic response to loading can occur on the periosteal surface when beta-cat levels are significantly reduced in Dmp1-expressing cells. 28005411_mutation creates a lower set point for extracellular phosphate and maintains it through the regulation of Fgf23 cleavage and expression 28603080_Findings support the critical role of DMP1 in maintaining the integrity of the auditory ossicles and its bony membrane 28647374_Nestin(+) cells are one important type of progenitor cell in bone marrow and are associated with bone remodeling. These data indicate that DMP1 plays negative roles in differentiation of Nestin(+) cells and bone formation. 28822114_study not only revealed a novel element, i.e., DMP1-PG, that regulated BBB formation, but also assigned a new function to DMP1-PG. 29775615_This study provides a new understanding of DMP1's function in regulation of osteogenesis: not only an enhancer of bone formation, but also a negative regulator of mesenchymal stem cell differentiation in bone. 31065929_Upon inspection, fewer cartilaginous calluses and down-regulated expression levels of chondrogenesis genes were observed in the fracture sites of S89G-DMP1 mice. The results suggest that DMP1-PG is an indispensable proteoglycan in chondrogenesis during fracture healing. 31461788_Glycosylation of DMP1 maintains cranial sutures in mice. 31505764_Twist1 Inactivation in Dmp1-Expressing Cells Increases Bone Mass but Does Not Affect the Anabolic Response to Sclerostin Neutralization. 31740066_results suggest that the expression of Dmp1 is up-regulated in Opn KO mice both in vivo and in vitro, and DMP1 compensates for the lack of OPN in regulating odontoblastlike cell differentiation after tooth injury. 32331833_Glycosylation of DMP1 promotes bone reconstruction in long bone defects. 32529635_Pten deletion in Dmp1-expressing cells does not rescue the osteopenic effects of Wnt/beta-catenin suppression. 34326685_Osteocytes but not osteoblasts directly build mineralized bone structures. 35366382_Improved cognitive impairments by silencing DMP1 via enhancing the proliferation of neural progenitor cell in Alzheimer-like mice. 36175851_The role of proteoglycan form of DMP1 in cranial repair. |
ENSG00000152592 |
DMP1 |
397.533898 |
0.1928052712 |
-2.374784 |
0.14460964 |
266.253445 |
0.00000000000000000000000000000000000000000000000000000000000743833017184215276843363044153725626123850418224480344175996302774169087790192153632519847763090440538487031476362576924296950107573007258744689251025311582665722198726143687963485717773437500000000000000 |
0.0000000000000000000000000000000000000000000000000000000016661859584926423262503724239048809592649166269141459008276441964071644158212986655685005362492262751600951531745299002864303162512506025394638797809676589167793281376361846923828125000000000000000000000 |
Yes |
No |
125.403316668708 |
12.5909569708369 |
650.41435802516 |
43.9672512217494 |
| ENSMUSG00000029335 |
110075 |
Bmp3 |
protein_coding |
Q8BHE5 |
FUNCTION: Negatively regulates bone density. Antagonizes the ability of certain osteogenic BMPs to induce osteoprogenitor differentitation and ossification. {ECO:0000269|PubMed:11138004}. |
Chondrogenesis;Cleavage on pair of basic residues;Cytokine;Developmental protein;Differentiation;Direct protein sequencing;Disulfide bond;Glycoprotein;Growth factor;Osteogenesis;Reference proteome;Secreted;Signal |
|
This gene encodes a secreted ligand of the TGF-beta (transforming growth factor-beta) superfamily of proteins. Ligands of this family bind various TGF-beta receptors leading to recruitment and activation of SMAD family transcription factors that regulate gene expression. The encoded preproprotein is proteolytically processed to generate each subunit of the disulfide-linked homodimer. This protein suppresses osteoblast differentiation, and negatively regulates bone density, by modulating TGF-beta receptor availability to other ligands. Homozygous knockout mice for this gene exhibit increased bone density and volume, while overexpression of this gene in a transgenic mouse causes bone defects resulting in spontaneous rib fractures. This gene encodes distinct protein isoforms that may be similarly proteolytically processed. [provided by RefSeq, Jul 2016]. |
mmu:110075; |
extracellular space [GO:0005615]; vesicle [GO:0031982]; BMP receptor binding [GO:0070700]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; cartilage development [GO:0051216]; osteoblast differentiation [GO:0001649]; positive regulation of pathway-restricted SMAD protein phosphorylation [GO:0010862]; positive regulation of transcription by RNA polymerase II [GO:0045944]; SMAD protein signal transduction [GO:0060395] |
17662325_BMP-2 and -7 were detected in Hertwig's epithelial root sheath (HERS) and in DF, then later in differentiated periodontal cells. BMP-3 was detected after D13 of the periodontal development. 19653325_Data suggest that BMP3 exerts its effects in the skeleton by altering signaling through ActRIIB in chondrocytes and the periosteum, and this results in defects in bone collar formation and late hypertrophic chondrocyte maturation. 22074949_findings best fit a model in which BMP3, produced by mature bone cells, acts to reduce BMP signaling through Acvr2b in skeletal progenitor cells, limiting their differentiation to mature osteoblasts 23451274_This region may preserve common cis-regulatory elements that govern Bmp3 expression across eutherian mammals. 26489765_the Bmp3/Wisp1 signaling pathway play a key role in mesenchymal stem cell proliferation, and consequently adipogenesis. 26982203_In two murine models of rheumatoid arthritis, BMP3 mRNA and protein are highly expressed by osteoblasts lining inflammation-bone interfaces late in the course of arthritis. 35054971_BMP3 Affects Cortical and Trabecular Long Bone Development in Mice. |
ENSG00000152785 |
BMP3 |
31.088293 |
0.0307178005 |
-5.024781 |
0.80283432 |
48.898775 |
0.00000000000269519027781029009726783093809739701987235327429459630366181954741477966308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000508083394945623001275862121612356472888372138640988850966095924377441406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.93647870279754 |
1.13410179947598 |
63.0409297836247 |
16.7833729711221 |
| ENSMUSG00000029338 |
71914 |
Antxr2 |
protein_coding |
Q6DFX2 |
FUNCTION: Necessary for cellular interactions with laminin and the extracellular matrix. {ECO:0000250|UniProtKB:P58335}. |
Disulfide bond;Glycoprotein;Membrane;Metal-binding;Phosphoprotein;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:71914; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; membrane [GO:0016020]; plasma membrane [GO:0005886]; metal ion binding [GO:0046872]; transmembrane signaling receptor activity [GO:0004888]; reproductive process [GO:0022414]; toxin transport [GO:1901998] |
16141341_because protective antigen binds to CMG2 with much higher affinity than it does to TEM8, a lower pH is needed to attenuate CMG2 binding to allow pore formation; toxin can form pores at different points in the endocytic pathway 17459655_The mRNA transcripts of both receptors, ANTXR1 and ANTXR2 were detected in J774A.1 cells and mouse tissues suggesting that anthrax edema toxin and B. anthracis Sterne spore are involved in the ANTXR mRNA regulation in host cells. 19617532_Data show that the lethality of anthrax toxin for mice is mostly mediated by CMG2 and that TEM8 plays only a minor role. 19858192_Cytoplasmic delivery of lethal factor by anthrax toxin receptor 2 was mediated by cathepsin B. 19901963_ANTXR2/CMG2 functions to promote endothelial proliferation and morphogenesis during sprouting angiogenesis, consistent with the endothelial expression of ANTXR2/CMG2 in several vascular beds. 21075356_This work demonstrates that anthrax toxin uptake through CMG2 and the resulting impairment of myeloid cells are essential to anthrax infection. 21079738_Data show that the two different PA oligomers are equally stabilized by ANTXR interactions. 22529944_we have discovered that ANTXR1 and ANTXR2 function as positive regulators of MT1-MMP activity. 22575514_these results demonstrate that CMG2 is not required for normal mouse embryonic development but is indispensable for murine parturition 24739539_blood pressure measured under anesthesia in Antxr2 siRNA-injected mice rose significantly compared with that of the controls, suggesting that ANTXR2 is a causative gene in the human 4q21 GWAS-blood pressure locus. 24742682_Silencing CMG2 using targeted siRNAs provided almost complete protection against anthrax lethal toxin-induced cytotoxicity and death in murine and human macrophages. 27555325_Anthrax Toxin Protective Antigen Variants That Selectively Utilize either the CMG2 or TEM8 Receptors for Cellular Uptake and Tumor Targeting. 28604699_Loss of Antxr2 promotes accumulation of collagen VI. Antxr2 is also associated with collagen VI signaling and degradation. 35212873_Capillary morphogenesis gene 2 (CMG2) mediates growth factor-induced angiogenesis by regulating endothelial cell chemotaxis. |
ENSG00000163297 |
ANTXR2 |
2011.851468 |
0.3901817098 |
-1.357782 |
0.09213899 |
212.527023 |
0.00000000000000000000000000000000000000000000000385972542476774974528705838223804760366977204599579203012937912217734749179433613743588552369928555533768180622239044432267296436123160674469545483589172363281250000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000005938518956571956105313456547852783218910260528574172255389827135581148765555415019013576821162272609140590321878140894068520694304424978327006101608276367187500000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1097.75392167054 |
70.1566891399081 |
2813.44279917316 |
131.733795426853 |
| ENSMUSG00000029348 |
72898 |
Asphd2 |
protein_coding |
Q80VP9 |
FUNCTION: May function as 2-oxoglutarate-dependent dioxygenase. {ECO:0000250}. |
Dioxygenase;Glycoprotein;Iron;Membrane;Metal-binding;Oxidoreductase;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:72898; |
membrane [GO:0016020]; dioxygenase activity [GO:0051213]; metal ion binding [GO:0046872]; peptidyl-amino acid modification [GO:0018193] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000128203 |
ASPHD2 |
731.670408 |
2.8049029420 |
1.487951 |
0.13618137 |
115.537599 |
0.00000000000000000000000000600104248465079422339693256590653186496835617781347986033195131483491415423647818982999524450860917568206787109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000003685801577669391325233317472790080911763863587459776936276321194769882427788587619943427853286266326904296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1074.51583851016 |
101.578247713464 |
383.084855606296 |
27.7153798988527 |
| ENSMUSG00000029361 |
18125 |
Nos1 |
protein_coding |
Q9Z0J4 |
FUNCTION: Produces nitric oxide (NO) which is a messenger molecule with diverse functions throughout the body. In the brain and peripheral nervous system, NO displays many properties of a neurotransmitter. Probably has nitrosylase activity and mediates cysteine S-nitrosylation of cytoplasmic target proteins such SRR. Isoform NNOS Mu may be an effector enzyme for the dystrophin complex. {ECO:0000269|PubMed:17293453}. |
3D-structure;Alternative splicing;Calmodulin-binding;Cell membrane;Cell projection;FAD;Flavoprotein;FMN;Heme;Iron;Membrane;Metal-binding;NADP;Oxidoreductase;Phosphoprotein;Reference proteome;Synapse;Ubl conjugation |
|
|
mmu:18125; |
azurophil granule [GO:0042582]; calyx of Held [GO:0044305]; cell periphery [GO:0071944]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; membrane raft [GO:0045121]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; nuclear membrane [GO:0031965]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; photoreceptor inner segment [GO:0001917]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic density, intracellular component [GO:0099092]; postsynaptic specialization, intracellular component [GO:0099091]; protein-containing complex [GO:0032991]; sarcolemma [GO:0042383]; sarcoplasmic reticulum [GO:0016529]; sarcoplasmic reticulum membrane [GO:0033017]; secretory granule [GO:0030141]; synapse [GO:0045202]; T-tubule [GO:0030315]; vesicle membrane [GO:0012506]; Z disc [GO:0030018]; ATPase binding [GO:0051117]; cadmium ion binding [GO:0046870]; calcium-dependent protein binding [GO:0048306]; calmodulin binding [GO:0005516]; enzyme binding [GO:0019899]; flavin adenine dinucleotide binding [GO:0050660]; FMN binding [GO:0010181]; heme binding [GO:0020037]; identical protein binding [GO:0042802]; NADP binding [GO:0050661]; NADPH binding [GO:0070402]; nitric-oxide synthase activity [GO:0004517]; oxidoreductase activity [GO:0016491]; phosphoprotein binding [GO:0051219]; scaffold protein binding [GO:0097110]; sodium channel regulator activity [GO:0017080]; transmembrane transporter binding [GO:0044325]; zinc ion binding [GO:0008270]; arginine catabolic process [GO:0006527]; behavioral response to cocaine [GO:0048148]; calcium ion transport [GO:0006816]; cellular response to epinephrine stimulus [GO:0071872]; cellular response to growth factor stimulus [GO:0071363]; cellular response to mechanical stimulus [GO:0071260]; establishment of localization in cell [GO:0051649]; establishment of protein localization [GO:0045184]; multicellular organismal response to stress [GO:0033555]; muscle contraction [GO:0006936]; negative regulation of apoptotic process [GO:0043066]; negative regulation of blood pressure [GO:0045776]; negative regulation of calcium ion transport [GO:0051926]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cytosolic calcium ion concentration [GO:0051481]; negative regulation of heart contraction [GO:0045822]; negative regulation of hepatic stellate cell contraction [GO:0061875]; negative regulation of hydrolase activity [GO:0051346]; negative regulation of insulin secretion [GO:0046676]; negative regulation of iron ion transmembrane transport [GO:0034760]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of peptidyl-serine phosphorylation [GO:0033137]; negative regulation of potassium ion transport [GO:0043267]; negative regulation of serotonin uptake [GO:0051612]; negative regulation of vasoconstriction [GO:0045906]; nitric oxide biosynthetic process [GO:0006809]; nitric oxide mediated signal transduction [GO:0007263]; peptidyl-cysteine S-nitrosylation [GO:0018119]; positive regulation of adenylate cyclase-activating adrenergic receptor signaling pathway involved in heart process [GO:0140196]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of guanylate cyclase activity [GO:0031284]; positive regulation of histone acetylation [GO:0035066]; positive regulation of long-term synaptic potentiation [GO:1900273]; positive regulation of neuron death [GO:1901216]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of sodium ion transmembrane transport [GO:1902307]; positive regulation of the force of heart contraction [GO:0098735]; positive regulation of transcription by RNA polymerase II [GO:0045944]; potassium ion transport [GO:0006813]; regulation of heart contraction [GO:0008016]; regulation of neurogenesis [GO:0050767]; regulation of postsynaptic membrane potential [GO:0060078]; regulation of sensory perception of pain [GO:0051930]; regulation of sodium ion transport [GO:0002028]; response to estrogen [GO:0043627]; response to heat [GO:0009408]; response to hormone [GO:0009725]; response to lipopolysaccharide [GO:0032496]; response to peptide hormone [GO:0043434]; retrograde trans-synaptic signaling by nitric oxide [GO:0098924]; striated muscle contraction [GO:0006941]; synaptic signaling by nitric oxide [GO:0099163]; vasodilation [GO:0042311]; xenobiotic catabolic process [GO:0042178] |
11729880_STP(short-term potentiation)and LTF(long-term facilitation) were absent in mutant mice deficient in NOS-1. Nitric oxide generated by NOS-1 may contribute to stability of breathing during sleep apneas by way of promoting LTF. 11829530_Brain neurons strongly positive for neuronal nitric oxide synthase immunoreactivity showed neither phosphorylated neurofilament nor microtubule-associated protein 2 immunoreactivity. Role in changing phosphorylation levels for NF and MAP-2 in neurons. 11907582_spatial confinement regulates myocardial contractility 12011984_Nitric oxide synthase involved in muscarinic receptor activation 12020853_HD transgenic mice missing both copies of the nNOS gene showed accelerated disease progression relative to HD transgenic mice wildtype or heterozygous for the nNOS gene. 12072412_deletion of exon 6 of the neuronal nitric oxide synthase gene in mice results in hypogonadism and infertility 12115690_The expression of nNOS and its colocalization with calcium binding proteins have been determined in different populations of hippocampal GABAergic neurons. 12234657_neuronal nitric oxide synthase has a neuroprotective role against alcohol toxicity within the developing brain. 12270125_Bcl-2-linked apoptosis due to increase in this enzyme in brain aging of senescence-accelerated mice 12297264_Deficiency of nNOS in a sciatic nerve injury model leads to an enhanced cell loss of dorsal root ganglia neurons. 12351742_The nNOS gene is critically involved in the regulation of neurobehavioral effects of alcohol. 12397095_neuronal nitric oxide synthase has vasculoprotective roles in suppressing neointimal formation 12456803_findings demonstrate that mouse neuronal nitric oxide synthase gene expression is regulated by the steroidogenic factor-1 gene family in pituitary gonadotropes 12509490_NOS-1 appears to be a key protein in generating the cardiac vagal gain of function elicited by exercise training. 12562678_Inducible NOS and neuronal NOS isoforms are responsible for the induction of fever to LPS, whereas endothelial NOS (eNOS) is not involved. In contrast, none of the NOS isoforms appeared to trigger fever to turpentine. 12571071_the regulation of cardiovascular and renal function in relation to blood pressure homeostasis in the NOS1-/- mice. Review. 12600881_The sarcolemmal localization of neuronal nitric oxide synthase in skeletal muscle is important for vasomodulation by skeletal muscle-derived nitric oxide. 12613893_In neurons in pterygopalatine ganglion, but intensity varied. Found in nerve fibers of lacrimal gland around tear-collecting ducts, blood vessels and acini. 12620898_nNOS and the nAChR are colocalized in murine skeletal muscle and myotubes but differ in their expressional regulation. 12623792_neuronal nitric oxide synthase-derived nitric oxide is a critical link between glutamatergic synaptic activity and blood flow in the activated cerebellum 12623875_Cardiac neuronal nitric oxide synthase regulates myocardial contraction and calcium handling. 12677017_The present study was designed to test whether nNOS is required for preconditioning. nNOS knockout mice lack protection from rapid ischemic preconditioning, suggesting that nitric oxide plays a role in the molecular mechanisms of protection. 12709511_Hypertrophied hearts from NOS1-/- mice have unique features, including decreased myocyte-enriched calcineurin interacting protein and paradoxical downregulation of fetal isoforms (alpha-skeletal actin and brain natriuretic peptide). 12788381_cardiac neuronal and endothelial nitric oxide synthase signals may play an essential role in successful cardiac aging 12798794_results suggest that expression and activity in satellite cells can increase the extent and speed of repair, even in the absence of dystrophin in muscle fibres 12834873_Altered neuronal nitric oxide synthase expression in the cerebellum of calcium channel mutant mice. 12864970_Neuronal nitric oxide synthase appears to be involved in the neuroendocrine-immune response to stress, perhaps via glucocorticoid regulation. 14523625_nNOS may play a key role in the development of MPTP neurotoxicity. 14526232_that modulation of eNOS-derived NO by hyperbaric oxygen (HBO2) is responsible for the early vasoconstriction responses, whereas late HBO2-induced vasodilation depends upon both eNOS and nNOS. 14660484_neuronal NOS had no effect on LPS-induced pulmonary apoptosis in sepsis 14722775_nerve function nNOS mediated, aorta function may be at least partially reliant on nNOS-related mechanisms 14983060_Nitric oxide produced by neuronal NO synthase mediate nonadrenergic/noncholinergic intestinal relaxation. 15026120_Infrequent colocalization of nNOS and somatostatin in mouse hippocampus. Double-labeled cells may be particular subpopulation of hippocampal GABAergic nonprincipal neurons. 15075180_NO generated by macula densa nNOS does not play a specific mediator role in macula densa-dependent renin secretion. 15148562_nNOS-positive cells were surrounded by GAP-43 labeled regions that appeared to be presynaptic boutons. Possible role of autocrine mediator for nNOS in anterior ventral cochlear nucleus. 15155789_Genomic regulation of nitric oxide bioavailability from neuronal nitric oxide synthase (nNOS) in cardiac autonomic ganglia in response to training is dependent on both alleles of the nNOS gene. 15191553_nNOS expression is regulated in the process of cutaneous wound repair. 15197473_nnos has a role in the limbic system, hypothalamus and the medial prefrontal cortex as demonstrated in stress-coping ability during forced swimming 15203939_COX-2 and constitutive NOS are important signaling molecules in the anabolic responses of neonatal tibial bone to the micromechanical load in vitro. 15217783_bacteria in the ulcerated colonic mucosa of eNOS knockout mice, but not in wild-type, iNOS, or nNOS knockout mice. 15220315_NO synthesized by both nNOS and iNOS plays a role in virus-induced sleep changes and that nNOS may modulate cytokine expression in the brain 15246834_We examined regulation of NO production by neuroal CAD cells and the effect of NO on cell survival. NO is generated dose-dependently by treatment with agents known to increase calcium entry across cell membranes, and increased further by sepiapterin 15287858_NOS-1 mRNA and protein levels hint that not only NOS-3, but also NOS-1 may be involved in the regulation of systemic hyperdynamic circulation and portal hypertension 15297441_nNOS plays a hitherto unrecognized but important physiological role as a stimulator of bone turnover 15319210_Deficiency of iNOS does not significantly affect severe congestive heart failure in mice after myocardial infarction. 15328041_decreased expression of IGF-I receptor in nNOS-/- mice 15350652_Spinal eNOS was mainly distributed in superficial laminae of dorsal horn. Enhanced spinal eNOS was expressed in astrocytes, but not in neurons. 15380304_Prolonged NO deficiency in the cerebellum may affect calcium channel protein expressions, especially, of the Cav1.2 and Cav1.3 subunits. 15486091_Deficiency of NOS1 leads to profound increases in XOR-mediated O(2)(.-) production, which in turn depresses myocardial excitation-contraction coupling. 15534476_A significant decrease of nNOS amacrine cells in the glaucomatous mouse eye indicates a specific response of nNOS positive amacrine cells in glaucomatous retinopathy. 15543804_Authors review the current understanding of the functional roles of nitric oxide (NO) and NOS in the cerebellum and present NO/NOS activities that have been described in various cerebellar mutant mice and NOS knockout mice. 15729148_Neuronal nitric-oxide-synthase(nNOS)-derived nitric oxide may be involved in the regulation of oxytocin gene expression in murine anterior commissural nucleus. 15845873_basal internal anal sphincter tone is largely controlled by eNOS in the mucosa, whereas the rectoanal inhibitory reflex is controlled by nNOS 15917272_expression of transgene prevents cardiomyopathy in dystrophin-deficient hearts 15923083_We found that association is due to physical interaction of mtNOS-I with the C-terminal peptide of the Va subunit of CcOX. 15961532_These data demonstrate that NO from eNOS regulates baseline blood flow in the mouse renal cortex and medulla, while NO produced by nNOS mediates an increase in medullary blood flow in response to ANG II. 15983249_the favorable effect of rosuvastatin on diabetic neuropathy neuronal is mediated in part via nitric oxide synthase/nitric oxide and phosphatidylinositol 3-kinase/Akt-signaling pathways 16139176_Effects of phencyclidineon prepulse inhibition in neuronal NOS (nNOS) deficient mice in the startle reaction. 16181097_The results suggest that the deleterious action of nNOS would counteract the role of HO-2 in neuroprotection. (Ncnos enzyme) 16191394_Our experiments describe specific changes in cell division and gene activity in the CaMKII-nNOS transgenic model and demonstrate its utility for studying the action of NO in the adult brain. 16224055_Upregulated nNOS may play an important compensatory role under arteriosclerotic/inflammatory conditions associated with eNOS dysfunction to maintain vascular homeostasis. 16246868_A previously unrecognized role of endogenous NO in the regulation of lipid metabolism is reported. 16256069_These studies suggest that nNOS expression in LM-MP varies with gender during early stage of type 2 diabetes. 16260864_Results suggest that nitric oxide synthase-1 and soluble guanylyl cyclase contribute to the constitutive regulation of tone in myoepithelial cells. 16301341_a protective role for NOS1 activity in the heart after MI; NOS1 deficiency contributes to an imbalance between oxidative stress and tissue NO signaling 16344403_nNOS plays a crucial role in preventing adverse LV remodeling and maintaining myocardial ss-adrenergic reserve after MI; upregulation of myocardial nNOS in infarcted hearts may be an important adaptive mechanism 16397145_eNOS and nNOS both play roles in the gender differences observed in ischemia/reperfusion injury under adrenergic stimulation, and also demonstrate increased S-nitrosylation of the L-type Ca2+ channels in female cardiomyocytes. 16488973_alternatively spliced forms of nNOS are major mediators of penile erection and so may be targets for therapeutic intervention 16567525_nNOS activity contributes to the absence of time dependent potentiation in mouse islets putatively through depletion of intracellular arginine. 16598948_Both taurine and zinc could prolong anoxia endurance by decreasing the level of neuronal nitric oxide synthase in the cerebral cortex of acute hypoxic mice. 16733795_It is concluded that the intrarenal spatial relation of individual NOS-isoforms and their alteration in expression provide the basis for versatile nitric oxide-mediated renal actions. 16807372_biglycan is important for the maintenance of muscle cell integrity and plays a direct role in regulating the expression and sarcolemmal localization of the intracellular signaling proteins dystrobrevin-1 and -2, alpha- and beta1-syntrophin and nNOS 17134781_The present study revealed the diversity of NOS positive neurons in the mouse main olfactory bulb. 17148677_Overexpression of CAR may lead to physiological dysfunction by interfering with normal signaling (through alterations in caveolin-3 and neuronal nitric-oxide synthase levels). 17151270_A key role is shown for the neuronal NOS isoform in promoting demyelination in response to chemical insult by cuprizone. 17184520_Age-related differences are found in nNOS activity level in cerebral cortex, brainstem and hippocampus in senescence accelerated-prone 8 (SAMP8) mice. 17198882_reduced mitochondrial generation of toxic radical oxygen species in NOS1-/- mice also increased the longevity of hematopoiesis in continuous bone marrow cultures and conferred radioresistance to cells in vitro 17242280_physiological relevance of the interaction between PMCA4b and nNOS and suggests its signaling role in the heart 17265179_NOS1 produced nitric oxide does not contribute to the dopamine deficit seen in HPRT deficiency 17272813_Conditional myocardial-specific overexpression of nNOS in transgenic mice reduced myocardial contractility. nNOS may suppress the function of L-type Ca(2+)-channels & in turn reduce Ca(2+)-transients accounting for the negative inotropic effect. 17307368_Data suggest that leptin deficiency is linked to decreased cardiac expression of NOS1 and NO production, with a concomitant increase in xanthine oxidoreductase activity and oxidative stress, resulting in nitroso-redox imbalance. 17387708_Expression and activity of nNOS are crucial to maintain redox status in brain, and alterations in oxidative damage may explain the phenotypical characteristics of nNOS knockout mice and their differential susceptibility to brain insults. 17394464_Inhibition and/or genetic deficiency of neuronal nitric oxide synthase (nNOS) but not endothelial nitric oxide synthase (eNOS) attenuated MnSOD nitration after TBI. 17395508_role of nNOS in sensitization of dorsal root ganglion neurones in inflammatory pain 17419805_these findings suggest a novel role for nNOS showing protective activity against insults that trigger tissue toxicity leading to memory impairments 17459722_Neuronal nitric oxide synthase (NOS-I) knockout increases the survival rate of neural cells in the hippocampus. 17574882_the role of NOS1 in murine bronchial responsiveness involves the epithelium of the central airways 17593140_Neuronally derived nitric oxide modulates interstitial cells of Cajal numbers and network volume in the mouse gastric body 17596536_characterize the relaxation induced by the soluble guanylyl cyclase stimulator BAY 41-2272; its effect on enos, nnos, and subunits of NADPH oxidase 17638388_We suggest that NO as an effector molecule and calbindin as a molecular biomarker illuminate key aspects of sexual differentiation in the developing mouse brain. 17690195_Central nervous system plasticity, mediated via nNOS, is involved ventilatory acclimatization to hypoxia. 17786240_nNOS/NO mediates muscle atrophy via regulation of Foxo transcription factors 17853069_Neuronal nitric oxide synthase gene inactivation reduces the expression of vasopressin in the hypothalamic paraventricular nucleus and of catecholamine biosynthetic enzymes in the adrenal gland of the mouse. 17854382_These results suggest that reduced nNOS is involved in ischemia-induced hippocampal neurogenesis by up-regulating iNOS expression and cAMP response element-binding protein phosphorylation. 17854383_Neuronal nitric oxide synthase contributes to chronic stress-induced depression by suppressing hippocampal neurogenesis. 17885995_early and delayed intravenous boluses of ascorbate prevent/reverse sepsis-induced deficit in arteriolar conducted vasoconstriction in the cremaster muscle by inhibiting nNOS-derived NO production. 17916611_eNOS and nNOS are differentially involved in the basal regulation of mitochondrial biogenesis in skeletal muscle but are not critical for exercise-induced increases in mitochondrial biogenesis in skeletal muscle. 17922909_nNOS, especially in the dorsal root ganglion, may participate in the development and/or maintenance of mechanical hypersensitivity after nerve injury 17951372_Genetic deletion of nNOS results in the inhibition of adrenergic-adenylate cyclase signaling within sinoatrial node myocytes. 17959704_eNOS-derived NO is of greater importance than nNOS-derived NO in an isolated arteriolar preparation of macula densa 18006627_producing nitric oxide may set into phase the bursting activity of GnRH neurons at key stages of reproductive physiology 18007024_myocardial nNOS promotes left ventricular relaxation by regulating the protein kinase A-mediated phosphorylation of PLN and the rate of sarcoplasmic reticulum Ca2+ reuptake via a cGMP-independent effect on protein phosphatase activity. 18057022_nNOS and AQP4 both require interaction with the PDZ domain of alpha-syntrophin for sarcolemmal association, their localization is regulated differentially. 18077344_NOS1 deficiency causes RyR2 hyponitrosylation, leading to diastolic Ca(2+) leak and a proarrhythmic phenotype. 18091753_nNOS is necessary for stimulation of renin in response to sodium restriction. nNOS(-/-) mice are normotensive, and their blood pressure responds normally to an increased dietary sodium intake. 18091979_These results describe a new role for nNOS beyond its action in the brain and muscle and suggest a model where nNOS, expressed in stromal cells, produces NO which acts as a paracrine regulator of hematopoietic stem cells. 18180383_Absence of AT(1) receptor signaling resulted in enhanced nNOS protein expression in both microvascular and tubular structures. It may contribute to the reduced renal vascular tone and blood pressure observed with blockade of the renin-angiotensin system. 18197485_insulin resistance in nNOS null mice was not related to defective insulin stimulation of skeletal muscle perfusion and substrate delivery or insulin signaling in the skeletal muscle cell, but to a sympathetic alpha-adrenergic mechanism. 18217802_Lack of nNOS decreases the sensitivity of retinal ganglion cells to light. 18235988_Overexpression of nNOS may play a major role in the neurotoxic processes of MPTP, as compared to the production of ROS, the overexpression of iNOS and the modulation of eNOS. 18305236_These findings demonstrate that hypoxic preconditioning elicits subcortical mitochondrial biogenesis by a novel mechanism that requires nNOS regulation of PGC-1alpha and CREB. 18326824_nitric oxide derived from nNOS is involved in a neurogenic mechanism of norobaric hyperoxia-induced lung injury 18342297_After repeated HBO exposures, elevated NO may enhance the sensitivity to convulsions and this may lead to seizures during the subsequent oxygen exposures. Prevention of seizures is needed when HBO is used as preconditioning method. 18400048_The activation of nNOS, localized in smooth muscle caveolae, by calcium entering through L-type calcium channels triggers nitric oxide production which modulates muscle contraction by a cGMP-dependent mechanism. 18400986_Cardiomyocyte NOS1 signaling modulates phospholamban serine16 phosphorylation, in part, via peroxynitrite. 18406496_We discovered that expression of the messenger RNA for both neuronal and endothelial nitric oxide synthase increased in the early postnatal period in the cerebellum in vivo. 18413498_genetic disruption of the whole NOS system causes spontaneous myocardial infarction associated with multiple cardiovascular risk factors of metabolic origin in mice in vivo through the angiotensin II type 1 receptor pathway 18421425_Our findings suggest that nitric oxide synthesized by the nNOS may play some role for neuronal growth during postnatal development. 18508038_demonstrate a role for NF-kappaB in selective induction of NOS1 during early inflammatory activation of astrocytes stimulated by low-dose MPTP and inflammatory cytokines. 18586241_We suggest that the protective effect of A(2A) antagonism in HD is related to the increase in striatal nNOS-IR neurons. 18635601_PIN/LC8 is associated with cytosolic but not membrane-bound nNOS in the nitrergic varicosities of mice gut. 18640116_In nNOS(-/-) mice, loss of nitrergic pyloric inhibition leads to gastric stasis and bezoars 18652592_required for the development of behavioral sensitization to ethanol in adolescent male and female mice and adult males 18652891_These results indicate that the NMDAR/nNOS cascade, activated via MORs, provide the free zinc ions required for inactive PKCgamma to bind to HINT1/RGSZ complex at the C terminus of the receptor. 18812700_renal COX-2 and nNOS are differentially regulated due to the elevation of renin-dependent blood pressure in mice lacking Cx40. 18852886_aberrant nNOSmu signaling can negatively impact three important clinical features of dystrophinopathies and sarcoglycanopathies: maintenance of muscle bulk, force generation and fatigabil 18952239_Substance P ameliorated the impaired wound healing response observed in NOS null mice by enhancing wound closure kinetics and epithelialization. 18952716_Neuronal nitric oxide synthase-derived hydrogen peroxide is a major endothelium-dependent relaxing factor. 18953332_nNOS-null mice do not have muscle pathology and have no loss of muscle-specific force after exercise but do display this exaggerated fatigue response to mild exercise 18987049_Role of neuronal NO synthase in regulating vascular superoxide levels and mitogen-activated protein kinase phosphorylation. 19108575_Results indicate that nNOS is the main supplier of nitric oxide in shear stress-induced vasodilation in skeletal muscle, but that sarcolemmal localization of nNOS is not indispensable for the function. 19278978_the nNOS-PMCA4b complex regulates contractility via cAMP and phosphorylation of both PLB and cTnI. 19307451_Erythropoietin protects the heart via nNOS from spontaneous and CsCl-induced ventricular arrhythmia during myocardial ischemia and reperfusion. 19340535_Role of nNOS in cardiac ischemia-reperfusion (IR) and ischemia preconditioning (IP). nNOS exacerbated IR-induced injury by increasing oxidative/nitrative stress and contributed to IP-induced protection by inhibition of oxidative/nitrative stress. 19358992_nNOS-alpha and -gamma splice variants are expressed in atherosclerotic plaques of apoE ko mice. 19360314_nNOS gene deficiency does not protect from development of nerve conduction deficit, sensory neuropathy and intraepidermal nerve fiber loss. 19362797_Absence of the neuronal nitric oxide synthase gene impairs ethanol-induced conditioned place preference, but not lithium chloride-induced conditioned place preference. 19407124_These results may indicate different roles for nitric oxide synthase isoforms in T. cruzi-induced cardiomyopathy. 19429176_results of the present study suggest that the nNOS gene, the level of tyrosine hydroxylase (TH) expression in ventral tegmental area and the regulation of TH in the dorsal striatum are associated with a sensitizing-regimen of cocaine 19470653_This study suggested that nNOS gene contributes more to optimal contextual fear learning than to cued fear learning, and therefore, inhibition of the nNOS enzyme may ameliorate context-dependent fear response. 19478875_protective role for nNOS against anthrax lethal toxin -mediated cardiac damage 19479987_the primary source of NO in the mammalian retina is nNOS alpha and that nNOS gamma may contribute to NO production as well. 19531381_In the mouse, NO of nNOS origin is critically involved in the regulation of protein synthesis-dependent olfactory long-term memory consolidation within relevant brain structures including the olfactory bulb. 19538708_nNOS knockout upregulates dopamine D1 receptor signaling, and induces abnormal social behavior, hyperactivity and impaired remote spatial memory. 19542095_defects in glycolytic metabolism and increased fatigability in dystrophic muscle may be caused in part by the loss of positive allosteric interactions between nNOS and phosphofructokinase. 19545548_these observations illustrate the complexity of nNOS in pain perception and the existence of opposing nNOS systems likely due to splice variants of nNOS. 19559786_The number of nNOS positive neurons fluctuates during the estrous cycle, reaching its peak during proestrus and metaestrus, and these variations were statistically significant in CA1, CA2 and CA3 regions. 19580782_these findings show that high concentrations of NO correlate with iNOS expression rather than nNOS expression in SOD1 G93A amyotrophic lateral sclerosis mice. 19615412_NRF-1 functionally bound to Nos1, but not Nos2 (inducible) and Nos3 (endothelial) gene promoters. 19652084_Data suggest that VEGF, acting via VEGFR2, plays a critical role in BP control by promoting NO synthase expression and NO activity. 19652483_localization of NOS isoforms in mammary gland during reproductive cycle; nNOS localized to alveoli, myoepithelia, lactiferous ducts & blood vessel endothelia; eNOS localized in alveoli, lactiferous ducts & blood vessel endothelia 19679819_Show that palmitoylation of PSD95 anchors nNOSalpha to the gut varicosity membrane and that it is obligatory for NO production by the enzyme. 19706695_Nitric oxide synthase isoforms (iNOS, nNOS, and eNOS) play distinct roles during acute peritonitis. 19730405_On the basis of these results, we conclude that neuronal NO is critical in the expression of the acute antinociceptive effect of HBO2. 19775503_nNOS gene has a more prominent role in the acquisition of approach behaviour to cocaine-associated cue than cocaine-associated context. 19800018_these findings support the hypothesis that NOS1 redistribution in injured myocardium requires the formation of a complex with the PDZ adaptor protein CAPON. 19845829_Increased production of nNOS in motor neurons inactivates cytochrome c oxidase. 19926812_rhythms in colonic motility are regulated by both clock genes and a nNOS-mediated inhibitory process and suggest a connection between these two mechanisms 19931630_species-specific gene expression in spinal cord in response to avulsion 19936028_These results suggest that increased nNOS activity is responsible for the majority of increased nitric oxide in retinal neurons in early diabetic retinopathy. 19940861_nitric oxide generated by epidermal nNOS has a significant role in the cutaneous circulatory response to mechanical stimulation 19959818_Spontaneous colonic migrating motor complexes occurred in both wild-type (frequency: 0.3 cycles/min) and nNOS(-/-) mice (frequency: 0.4 cycles/min) 20124730_nNOS splice variants are critical regulators of skeletal muscle exercise performance. 20158465_These in vivo data suggest that NO production in the striatum after reperfusion is closely related to activities of both nNOS and eNOS, and is mainly related to nNOS following reperfusion. 20164327_The findings of this study indicated that both hippocampal nNOS and CREB activity mediate the anxiolytic effects of 5-HT(1A)R agonists and SSRIs. 20304785_Complete disruption of all NOS genes causes severe dyslipidaemia, atherosclerosis, and sudden cardiac death in response to a high-fat diet in mice. 20435320_we demonstrated that nNOS dislocated from the sarcolemma to the cytoplasm may be the upstream disease-modifying event of muscle atrophy in human ALS and transgenic mice 20483958_Utrophin cannot anchor nNOS to the sarcolemma. 20543469_NMDA receptors and NOS may play important roles in morphine tolerance in mice with bone cancer pain. 20561153_BPA has a powerful effect on specific portions of the nNOS-immunoreactive system belonging to the accessory olfactory system that are particularly important for the control of sexual behaviour. 20624383_Data suggest that nNOS and eNOS both contribute in significant ways to relaxation in mouse thoracic aorta. 20651700_NO, selectively derived from nNOS, mediates renin release during sodium restriction by inhibiting PDE3, which would increase renin release by elevating cAMP levels in the juxataglomerular cells 20655878_the current study demonstrates that both nNOS and iNOS are partially responsible for the pulmonary oxidative and nitrosative stress reaction in the mouse acute lung injury and sepsis model. 20661721_Neuronal nitric oxide synthase in hypothalamus is increased in hypobaric hypoxic exposure. 20662604_Immunohistochemical expression of Bcl-2, p53 and caspase-9 in hypothalamus magnocellular centers of nNOS knockout mice following water deprivation 20707635_The expression of nNOS was upregulated in the frontal cortex, but downregulated in the hippocampus in SAM-prone8 (senescence-accelerated mouse) 20720107_the first pleckstrin homology and PDZ domains of alpha-syntrophin work in concert to facilitate the localization of AChRs and nNOS at the neuromuscular junction 20727829_the pathway activated by dystrophin/nNOS controls several important circuitries increasing the robustness of the muscle differentiation program. 20739607_High protein-induced glomerular hyperfiltration is independent of the tubuloglomerular feedback mechanism and NOS isoforms. 20845474_Data conclude that NSCs-derived nNOS stimulates neurogenesis via activating telomerase, whereas neurons-derived nNOS represses neurogenesis by supplying exogenous NO that hinders CREB activation, in turn, reduces nNOS expression in NSCs. 20921441_conditional transgenic overexpression of nNOS resulted in myocardial protection after ischemia/reperfusion injury. 20953358_NOS3 is essential for the expression of NOS2 under inflammatory conditions, while NOS1 expression contributes to allergen-induced goblet cell metaplasia 21073925_Visual cue-dependent long-term memory (LTM) is impaired in nNOS KO mice; aberrant modulation of CREB in the absence of the nNOS gene may hinder cued and contextual LTM formation. 21082337_The present study demonstrates that the expression of nNOS was more pronounced than that of iNOS expression in the dentate gyrus of aged mice 21102461_Cerebral ischemia induces the interaction of nNOS with postsynaptic density protein-95. 21179208_the increased spinal cord expression of NOS1, regulated by NOS2, might be responsible for the maintenance of chronic peripheral neuropathic pain in mice 21331324_Coexpression of reelin and nNOS in several hippocampal regions suggests that reelin and nNOS may work synergistically to promote glutamatergic function. 21434910_These results suggest that nNOS is involved in the induction but not the maintenance of nerve growth factor-evoked facilitation of nociception in the brainstem. 21470341_Chronic alcohol consumption induces a marked increase in NO synthesis by jejunal myenteric neurons, accompanied by an up-regulation of iNOS-expressing neurons and a downregulation of nNOS neurons. 21539806_These results suggest that nNOS KO mice are useful tools in studying the role of nNOS signaling in cortical development and prefrontal cortical functioning. 21606640_Different isoforms of nitric oxide synthase are involved in angiotensin-(1-7)-mediated plasticity changes in the amygdala in a gender-dependent manner. 21613472_Hippocampal nNOS significantly downregulates local glucocorticoid receptor expression through both soluble guanylate cyclase and peroxynitrite extracellular signal-regulated kinase signal pathways. 21615722_The results indicate a new mechanism for endothelial dysfunction showing a critical role for nNOS-derived H2O2 in the impaired vasodilator response in atherosclerosis. nNOS may represent a novel target for vascular atherosclerosis. 21651902_NOS-1-derived NO acts as a signaling element and it is essential for the initiation of systemic inflammation 21756313_Mice lacking all three NOS genes (n/i/eNOS-/-mice) were used and investigated the behavioral phenotypes in a series of acute and chronic pain assays. 21777607_Behavioral testing reduces nNOS-positive neurons in the basolateral amygdala and ventral lateral septum of socially-housed but not in the social-nonsocial-enriched mice buffered by environmental complexity. 21780643_Interaction of neuronal NOS and catecholamines in regulation of expression of proteins of apoptosis by vasopressinergic hypothalamic neurons 21849632_nNOS is not essential for functional muscle regeneration after acute myotoxic damage. 21896719_Data show that physiological concentrations of H(2)O(2) activate eNOS but not nNOS. 21945318_The beta-isoform was co-immunoprecipitated with alpha-isoform antibodies in muscle extracts indicating an association of both nNOS-isoforms to direct the beta-variant to the sarcolemma. 21965681_Plasma membrane calcium pump (PMCA4)-neuronal nitric-oxide synthase complex regulates cardiac contractility through modulation of a compartmentalized cyclic nucleotide microdomain. 21976486_identify nNOS effects on the HIF1a/pVHL signaling pathway as critically important in the physiological responses to anemia 22027181_The nNOS deficiency-induced cell proliferation depletes the neurogenic reserve. 22058155_These results suggest that nNOS, not eNOS, plays the dominant role in modulating Ca(2+) cycling in the heart. 22205701_FcgammaR engagement relies on Ca(2+)-dependent ERK phosphorylation, which in turn increases nNOS and, to a lesser extent, eNOS, both of which produce low levels of NO that function to promote phagocytosis. 22262671_The distribution of nNOS gradually increases and extends laterally in embryo brain, in turn implying that nitric oxide might be involved in the development of mouse brain. 2 |
ENSG00000089250 |
NOS1 |
13.155923 |
23.9564755727 |
4.582344 |
1.10718982 |
19.844685 |
0.00000839959372143204313111708447792125298292376101016998291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000631191966279491284853159216439166812051553279161453247070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.2807938752303 |
22.2080439508561 |
1.09702254805916 |
0.80971936718562 |
| ENSMUSG00000029381 |
27428 |
Shroom3 |
protein_coding |
E9QMY5 |
Human_homologues FUNCTION: Controls cell shape changes in the neuroepithelium during neural tube closure. Induces apical constriction in epithelial cells by promoting the apical accumulation of F-actin and myosin II, and probably by bundling stress fibers (By similarity). Induces apicobasal cell elongation by redistributing gamma-tubulin and directing the assembly of robust apicobasal microtubule arrays (By similarity). {ECO:0000250|UniProtKB:Q27IV2, ECO:0000250|UniProtKB:Q9QXN0}. |
Actin-binding;Cytoplasm;Cytoskeleton;Microtubule;Proteomics identification;Reference proteome |
|
|
mmu:27428; |
cytoplasm [GO:0005737]; microtubule [GO:0005874]; actin filament binding [GO:0051015] |
16249236_Shroom likely facilitates neural tube closure by regulating cell shape changes via the apical positioning of an actomyosin network in the neurepithelium. 18339671_In the invaginating neural plate/tube, Shroom3 colocalized with ROCKs at the apical junctions; Shroom3 depletion or RII-C1 expression in the tube removed these apically localized ROCKs, and concomitantly blocked neural tube closure. 20081189_expression of Shroom3 is dependent on the crucial lens-induction transcription factor Pax6 20926658_Scaffold protein POSH, in association with Shroom3 and mixed lineage kinase LZK, relays axon outgrowth inhibition downstream of NogoA and PirB proteins. 21726547_Shroom3 and N-cadherin function cooperatively downstream of Pitx2 to directly regulate cell shape changes necessary for early gut tube morphogenesis 24282618_findings reveal that Lulu is essential for Shroom3-dependent apical constriction during vertebrate neural tube closure 25038041_Shroom3 has a role in apical constriction during lens pit morphogenesis and is recruited in a pathway involving p120-catenin 32511952_SHROOM3 is downstream of the planar cell polarity pathway and loss-of-function results in congenital heart defects. 33273487_SHROOM3, the gene associated with chronic kidney disease, affects the podocyte structure. 35368578_Shroom3, a Gene Associated with CKD, Modulates Epithelial Recovery after AKI. |
ENSG00000138771 |
SHROOM3 |
27.275263 |
0.1473370995 |
-2.762807 |
0.57760565 |
21.574055 |
0.00000340425862659195893793000718685615169079028419218957424163818359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000276032076631144873040193954416565702558727934956550598144531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.86611553298455 |
2.82298780578974 |
46.6014028664361 |
12.9925933436457 |
| ENSMUSG00000029403 |
53886 |
Cdkl2 |
protein_coding |
Q9QUK0 |
|
Alternative splicing;ATP-binding;Cytoplasm;Kinase;Nucleotide-binding;Nucleus;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:53886; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; ATP binding [GO:0005524]; cyclin-dependent protein serine/threonine kinase activity [GO:0004693]; protein serine kinase activity [GO:0106310]; protein phosphorylation [GO:0006468] |
14605869_cdkl2 functions mainly in mature neurons and that mechanisms governing regulation of this gene expression in mice are distinct from those of immediate-early genes. |
ENSG00000138769 |
CDKL2 |
56.728810 |
0.2632499469 |
-1.925495 |
0.40996519 |
21.642455 |
0.00000328499614143066730935580782579918945884855929762125015258789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000267291993730085041924404015700744707828562241047620773315429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.3801991809813 |
7.30965242237839 |
96.411032484823 |
19.6141431745885 |
| ENSMUSG00000029516 |
12704 |
Cit |
protein_coding |
D3YU89 |
FUNCTION: Plays a role in cytokinesis. Displays serine/threonine protein kinase activity. {ECO:0000256|PIRNR:PIRNR038145}. |
ATP-binding;Coiled coil;Cytoplasm;Kinase;Metal-binding;Nucleotide-binding;Proteomics identification;Reference proteome;Serine/threonine-protein kinase;Transferase;Zinc;Zinc-finger |
|
|
|
cleavage furrow [GO:0032154]; cytosol [GO:0005829]; Golgi cisterna [GO:0031985]; midbody [GO:0030496]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; ATP binding [GO:0005524]; metal ion binding [GO:0046872]; PDZ domain binding [GO:0030165]; protein kinase binding [GO:0019901]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; protein serine/threonine kinase inhibitor activity [GO:0030291]; scaffold protein binding [GO:0097110]; transcription coactivator binding [GO:0001223]; G2/M transition of mitotic cell cycle [GO:0000086]; Golgi organization [GO:0007030]; liver development [GO:0001889]; mitotic cytokinesis [GO:0000281]; negative regulation of hippo signaling [GO:0035331]; neuron apoptotic process [GO:0051402]; positive regulation of cytokinesis [GO:0032467]; protein phosphorylation [GO:0006468]; regulation of actin polymerization or depolymerization [GO:0008064] |
12411428_Citron-K has a distinct cell cycle-dependent expression pattern and cellular localization as a downstream target of Rho-GTPase and functions in the control of G(2)/M transition in the hepatocyte cell cycle 12781320_Quantitative analysis of citron kinase in cerebral cortex of knockout mice demonstrates that this molecule is involved in differentially regulating the morphology of the dendritic compartment in corticocollicular projecting pyramidal neurons. 17488780_These results suggest that the TTC3-RhoA-CIT-K pathway could be a crucial determinant of in vivo neuronal development, whose hyperactivity may result in detrimental effects on the normal differentiation program. 19148892_Lack of citron kinase gene in newborn mice profoundly alters barrelfield cortex cell morphology and number. 20369383_CITRON has a role in proliferation of hepatocellular carcinoma cells 20436275_Data report that Discs large 5 (Dlg5), a member of the MAGUK family, is an interactor of CitK required for CitK polarization. 22293177_p27 has a role in cytokinesis via the regulation of citron-K activity 23444367_Data propose that the CC-domain-mediated translocation and actions of Citron-K ensure proper stabilization of the midbody structure during the transition from constriction to abscission. 26586574_The CIT-K may promote this event by interacting with TUBB3 and by recruiting at the midbody casein kinase-2alpha (CK2alpha) that has previously been reported to phosphorylate the S444 residue. 27562601_in this study we have provided evidence that ASPM controls spindle orientation by regulating the dynamics of astral MT and that CITK is a critical downstream partner of ASPM for this activity. |
ENSG00000122966 |
CIT |
825.932033 |
2.1447352300 |
1.100800 |
0.22477313 |
23.523173 |
0.00000123418292451183074589528537029492483156900561880320310592651367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000108414500035549068372546510641285522069665603339672088623046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1124.85861056055 |
175.963819058799 |
524.474347611164 |
61.7105529531096 |
| ENSMUSG00000029570 |
16848 |
Lfng |
protein_coding |
O09010 |
FUNCTION: Glycosyltransferase that initiates the elongation of O-linked fucose residues attached to EGF-like repeats in the extracellular domain of Notch molecules. Modulates NOTCH1 activity by modifying O-fucose residues at specific EGF-like domains resulting in inhibition of NOTCH1 activation by JAG1 and enhancement of NOTCH1 activation by DLL1 via an increase in its binding to DLL1 (PubMed:28089369). Decreases the binding of JAG1 to NOTCH2 but not that of DLL1 (By similarity). Essential mediator of somite segmentation and patterning. During somite boundary formation, it restricts Notch activity in the presomitic mesoderm to a boundary-forming territory in the posterior half of the prospective somite. In this region, Notch function activates a set of genes that are involved in boundary formation and in anterior-posterior somite identity (PubMed:10330372). Ectopically expressed in the thymus, Lfgn inhibits Notch signaling which results in inhibition of T-cell commitment and promotes B-cell development in lymphoid progenitors (PubMed:11520458). May play a role in boundary formation of the enamel knot (PubMed:12167404). {ECO:0000250|UniProtKB:Q8NES3, ECO:0000269|PubMed:10330372, ECO:0000269|PubMed:11520458, ECO:0000269|PubMed:12167404, ECO:0000269|PubMed:28089369}. |
Developmental protein;Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Manganese;Membrane;Metal-binding;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
|
|
mmu:16848; |
Golgi membrane [GO:0000139]; acetylglucosaminyltransferase activity [GO:0008375]; metal ion binding [GO:0046872]; O-fucosylpeptide 3-beta-N-acetylglucosaminyltransferase activity [GO:0033829]; compartment pattern specification [GO:0007386]; marginal zone B cell differentiation [GO:0002315]; negative regulation of Notch signaling pathway involved in somitogenesis [GO:1902367]; ovarian follicle development [GO:0001541]; positive regulation of meiotic cell cycle [GO:0051446]; positive regulation of Notch signaling pathway [GO:0045747]; positive regulation of protein binding [GO:0032092]; regulation of Notch signaling pathway [GO:0008593]; regulation of somitogenesis [GO:0014807]; somitogenesis [GO:0001756]; T cell differentiation [GO:0030217] |
12110168_An evolutionarily conserved region in murine Lunatic fringe (Lfng) promoter drives periodic somite expression. Oscillatory Notch signaling underlies the segmentation clock and directly activates and indirectly represses Lfng expression. 12110169_Lunatic fringe (Lfng) expression in the presomitic mesoderm is required for somite segmentation. Independent Lfng regulatory cassettes advance a molecular framework for deciphering this segmentation. 12167404_Lunatic fringe, FGF, and BMP regulate the Notch pathway during epithelial morphogenesis of teeth. 12670869_Transcriptional oscillation of this protein is essential for somitogenesis. 12783854_periodic repression by Hes7 protein is critical for the cyclic transcription of Hes7 and Lfng, and this negative feedback represents a molecular basis for the segmentation clock 15659488_Lunatic fringe null female mice were found to be infertile. 15822856_The Jsr mutation disrupts the A-P polarity of somites during the somitogenesis without altering Lfng expression pattern in the presomitic mesoderm. 16342160_Lunatic fringe and Hes7 controls their oscillatory expression during somitogenesis by forming negative feedback loop 16699526_The ability of Lfng and Notch1 to control progenitor competition for limiting cortical niches is an important mechanism for the homeostatic regulation of thymus size. 17142733_Shutting down the expression of Lfng in CD4+CD8+DP cells may have a physiological role in promoting DP cells differentiation toward mature CD8+SP cells. 17600782_spondylocostal dysostosis (SCD) is caused by mutation in Delta-like 3 (DLL3), Mesoderm posterior 2 (MESP2), and Lunatic fringe (LFNG); three genes that are components of the Notch signaling pathway. 18234727_In the absence of oscillatory Lfng expression, mice exhibit severe segmentation phenotypes in the thoracic and lumbar skeleton 19026002_Jag1/fringe genes (Lfng, Rfng, and Mfng) may regulate postnatal bile duct growth and remodeling, and serve as candidate modifiers of the hepatic phenotype in Alagille syndrome. 19217325_Lunatic Fringe (Lfng) and Manic Fringe (Mfng) cooperatively enhanced the DL1-Notch2 interaction to promote marginal zone B cell development 19440349_The expression of lunatic fringe oscillates in 2-h cycles in the mouse presomitic mesoder. 19479951_No obvious synergistic defects in mice in the absence of two, or all three, fringe genes during development of the axial skeleton, limbs, hindbrain, and cranial nerves. 19897741_Lfng functions to enhance Notch signaling in myofibroblast precursor cells and thereby to coordinate differentiation and mobilization of myofibroblasts required for alveolar septation. 19956724_there are differential axial requirements for lunatic fringe and Hes7 transcription during mouse somitogenesis 20510365_Data suggest that lunatic fringe strongly augments Notch signaling mediated by Delta-like 1 but not Jagged 1. 20844195_data reveal that Lfng enhances CD4/CD8 double-negative 3b precursor competition for intrathymic Delta-like Notch ligands to maximize Notch-induced clonal expansion during the earliest stage of beta-selection. 21097675_Decreasing Lfng expression during the (TCR-CD4/CD8 double negative 3) DN3-DP (CD4/CD8 double positive) transition minimizes the potent leukemogenic potential of Notch1 signaling. 22081605_the presence of Gal on O-fucose glycans differentially affects DLL1-induced NOTCH signaling modulated by LFNG versus MFNG 22095884_Intriguing changes are observed in the cranio-caudal borders of multifidus muscle in mutant Dll3 and Lfng models of idiopathic scoliosis. 22326607_Lfng expression and activity is normal in mice whose Lfng is lengthened by 10 kb, and no effects on segmentation are evident. 22624713_Reduced LFNG expression facilitates JAG/NOTCH luminal progenitor signaling and cooperates with MET/CAVEOLIN basal-type signaling to promote basal-like breast cancer. 23072809_The repressive effect of Lfng against Notch activities in neighbouring cells can sufficiently explain the synchronization in vivo. 24367028_STAT5-dependent amplification of Notch-modifying Lfng augments Th2 response via Dll4 and is critical for amplifying viral exacerbation during allergic airway disease. 24560643_suggest that modulation of the Notch signaling by Lfng affects the clock period during development 26279302_a potent tumor-suppressive function for Lfng 26608918_each Fringe contributes to T and B cell development, and Fringe is required for optimal in vitro stimulation of T and B cells. 26811377_LFNG protein may have context-dependent effects on Notch activity; somitogenesis is disrupted by a novel dominant allele of Lfng 27966429_These results show that Notch signaling is finely calibrated in the cochlea via Lfng and Mfng to produce precisely tuned levels of signaling that first set the boundary of the organ of Corti and later regulate hair cell development. 28089369_Fringe modifications at EGF8 and EGF12 enhanced Notch1 binding to and activation from Delta-like 1, while modifications at EGF6 and EGF36 (added by Manic and Lunatic but not Radical) inhibited Notch1 activation from Jagged1. 28699891_Lfng-mediated Notch signaling appears to be a key factor governing NSC quiescence, division, and fate. 28710810_results suggest that mir-125a sites in the Lfng 3' untranslated region influence transcript turnover in both mouse and chicken embryos, and support the existence of position-dependent regulatory mechanisms in the presomitic mesoderm. 29193607_LFNG expression plays a functional role in regulating melanoma metastasis. 34872929_Lunatic Fringe-GFP Marks Lamina-Specific Astrocytes That Regulate Sensory Processing. |
ENSG00000106003 |
LFNG |
360.396085 |
2.2430925495 |
1.165489 |
0.19228871 |
35.736028 |
0.00000000225946644170452199239434849212422584674442305185948498547077178955078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000302692988918586457976561362483158834635332823381759226322174072265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
486.035528618048 |
64.972915516442 |
216.680996385713 |
22.01431054732 |
| ENSMUSG00000029636 |
245880 |
Wasf3 |
protein_coding |
Q8VHI6 |
FUNCTION: Downstream effector molecules involved in the transmission of signals from tyrosine kinase receptors and small GTPases to the actin cytoskeleton. Plays a role in the regulation of cell morphology and cytoskeletal organization. Required in the control of cell shape (By similarity). {ECO:0000250}. |
Actin-binding;Coiled coil;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:245880; |
cytoskeleton [GO:0005856]; glial cell projection [GO:0097386]; glutamatergic synapse [GO:0098978]; lamellipodium [GO:0030027]; postsynapse [GO:0098794]; SCAR complex [GO:0031209]; actin binding [GO:0003779]; Arp2/3 complex binding [GO:0071933]; protein kinase A regulatory subunit binding [GO:0034237]; actin cytoskeleton organization [GO:0030036]; cytoskeleton organization [GO:0007010]; lamellipodium assembly [GO:0030032]; modification of postsynaptic actin cytoskeleton [GO:0098885]; oligodendrocyte development [GO:0014003]; positive regulation of Arp2/3 complex-mediated actin nucleation [GO:2000601]; positive regulation of myelination [GO:0031643]; regulation of cell shape [GO:0008360] |
12185600_WAVE3 transcript is mainly expressed in the nervous system and, like all the members of the WASP gene family, WAVE3 is a key element in actin polymerization and cytoskeleton organization. 28794159_p63alpha protein up-regulates heat shock protein 70 expression via E2F1 transcription factor 1, promoting Wasf3/Wave3/MMP9 signaling and bladder cancer invasion 31542393_Elevated WASF3 Expression Promotes Metastasis in Breast Cancer. 32312818_our work suggests that PCARE is an actin-associated protein that interacts with WASF3 to regulate the actin-driven expansion of the ciliary membrane at the initiation of new outer segment disk formation. 33594155_Phosphorylation of the proline-rich domain of WAVE3 drives its oncogenic activity in breast cancer. |
ENSG00000132970 |
WASF3 |
15.527622 |
0.0073003340 |
-7.097822 |
1.51042072 |
22.563929 |
0.00000203265419342729614299910417818395558242627885192632675170898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000170868590917685305504303966417367632857349235564470291137695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.15914146857053 |
0.198761507004452 |
21.7992038600678 |
11.7672467075496 |
| ENSMUSG00000029659 |
70717 |
Medag |
protein_coding |
Q14BA6 |
FUNCTION: Involved in processes that promote adipocyte differentiation, lipid accumulation, and glucose uptake in mature adipocytes. {ECO:0000269|PubMed:22510272}. |
Alternative splicing;Cytoplasm;Reference proteome |
|
|
mmu:70717; |
cytoplasm [GO:0005737]; positive regulation of fat cell differentiation [GO:0045600] |
22510272_study identifies Meda-4 as a novel adipogenic gene, capable of promoting differentiation of preadipocytes into adipocytes, increasing lipid content and glucose uptake in adipocytes; it might play an important role in adipose tissue expansion in normal and aberrant hormonal conditions and pathophysiological states |
ENSG00000102802 |
MEDAG |
297.687898 |
2.6019198037 |
1.379576 |
0.19866949 |
47.495845 |
0.00000000000551206417000737110168038068723188739432561344955274762469343841075897216796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001003103481930134786257211805117852722324389702635016874410212039947509765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
435.270259269457 |
60.129468232028 |
167.28811497117 |
17.748142897397 |
| ENSMUSG00000029673 |
319974 |
Auts2 |
protein_coding |
A0A087WPF7 |
FUNCTION: Component of a Polycomb group (PcG) multiprotein PRC1-like complex, a complex class required to maintain the transcriptionally repressive state of many genes, including Hox genes, throughout development. PcG PRC1 complex acts via chromatin remodeling and modification of histones; it mediates monoubiquitination of histone H2A 'Lys-119', rendering chromatin heritably changed in its expressibility. The PRC1-like complex that contains PCGF5, RNF2, CSNK2B, RYBP and AUTS2 has decreased histone H2A ubiquitination activity, due to the phosphorylation of RNF2 by CSNK2B. As a consequence, the complex mediates transcriptional activation (By similarity). In the cytoplasm, plays a role in axon and dendrite elongation and in neuronal migration during embryonic brain development. Promotes reorganization of the actin cytoskeleton, lamellipodia formation and neurite elongation via its interaction with RAC guanine nucleotide exchange factors, which then leads to the activation of RAC1 (PubMed:25533347). {ECO:0000250|UniProtKB:Q8WXX7, ECO:0000269|PubMed:25533347}. |
Alternative splicing;Cell projection;Cytoplasm;Cytoskeleton;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
Human_homologues hsa:26053; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; growth cone [GO:0030426]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; actin cytoskeleton reorganization [GO:0031532]; axon extension [GO:0048675]; dendrite extension [GO:0097484]; innate vocalization behavior [GO:0098582]; neuron migration [GO:0001764]; positive regulation of histone H3-K4 methylation [GO:0051571]; positive regulation of histone H4-K16 acetylation [GO:2000620]; positive regulation of lamellipodium assembly [GO:0010592]; positive regulation of Rac protein signal transduction [GO:0035022]; positive regulation of transcription by RNA polymerase II [GO:0045944]; righting reflex [GO:0060013] |
19948250_Autism susceptibility candidate 2 (Auts2) encodes a nuclear protein expressed in developing brain regions implicated in autism neuropathology. 25180570_Our results implicate Auts2 as an active regulator of important neurodevelopmental genes and pathways and identify novel genomic regions that could be associated with ASD and other neurodevelopmental diseases. 25533347_AUTS2 activates Rac1 to induce lamellipodia but downregulates Cdc42 to suppress filopodia. 27423627_Study reveals AUTS2 signaling role in mediating heroin-induced behavioral and neurochemical adaptations in the nucleus accumbens, but not the caudate-putamen, potentially mediated through the control of Rac1 activity. 30953002_AUTS2 isoforms control neuronal differentiation. 31554716_Description of a mouse mutation, T(5G2;8A1)GSO, a reciprocal translocation that breaks between Auts2 and Galnt17 and dysregulates both genes and Is associated with phenotypes related to the human AUTS2 syndrome. 34013328_AUTS2 Regulates RNA Metabolism and Dentate Gyrus Development in Mice. 35235353_Auts2 deletion involves in DG hypoplasia and social recognition deficit: The developmental and neural circuit mechanisms. 36525998_Auts2 regulated autism-like behavior, glucose metabolism and oxidative stress in mice. |
ENSG00000158321 |
AUTS2 |
1418.245802 |
2.1727758329 |
1.119539 |
0.11245676 |
96.438903 |
0.00000000000000000000009204073062538401111802685242560878666074385994361666669668980370749844510669390729162842035293579101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000043005043217357342044080266791521769043416627871735768675253916426548528306739171966910362243652343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1881.18211680732 |
146.784970500119 |
865.796686574626 |
51.3113231849526 |
| ENSMUSG00000029675 |
13717 |
Eln |
protein_coding |
P54320 |
FUNCTION: Major structural protein of tissues such as aorta and nuchal ligament, which must expand rapidly and recover completely. Molecular determinant of the late arterial morphogenesis, stabilizing arterial structure by regulating proliferation and organization of vascular smooth muscle. {ECO:0000269|PubMed:9607766}. |
Disulfide bond;Extracellular matrix;Hydroxylation;Reference proteome;Repeat;Secreted;Signal |
|
This gene encodes elastin, the extracellular matrix protein that forms a major structural component of several tissues including lungs and arterial walls. Cleavage of the signal peptide from the encoded precursor generates soluble tropoelastin which undergoes lysine-derived crosslinking to form elastin polymers. Mice lacking the encoded protein exhibit defective lung development, and die of an obstructive arterial disease resulting from subendothelial cell proliferation and reorganization of smooth muscle. [provided by RefSeq, Aug 2015]. |
mmu:13717; |
collagen-containing extracellular matrix [GO:0062023]; elastic fiber [GO:0071953]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; mitochondrion [GO:0005739]; extracellular matrix binding [GO:0050840]; extracellular matrix constituent conferring elasticity [GO:0030023]; aortic valve morphogenesis [GO:0003180]; blood vessel remodeling [GO:0001974]; extracellular matrix assembly [GO:0085029]; extracellular matrix organization [GO:0030198]; outflow tract morphogenesis [GO:0003151]; regulation of actin filament polymerization [GO:0030833]; skeletal muscle tissue development [GO:0007519]; stress fiber assembly [GO:0043149] |
12466207_Data show that elastin induces actin stress fiber organization, inhibits proliferation, regulates migration and signals via a non-integrin, heterotrimeric G-protein-coupled pathway. 12626514_tropoelastin has domains that mediate elastin deposition in vitro and in vivo 12909585_Coordinately expressed and regulated with fibulin 5 in lung fibroblasts and may serve a key role during lung injury and repair. 14614988_elastin gene product, signaling through the VGVAPG domain, directly induces VSMC myofibrillogenesis 15615710_B-Myb represses SMC elastin gene expression and cyclin A plays a role in the developmental regulation of elastin gene expression in the aorta 15863465_The mechanical behavior of ELN(+/-) arteries is likely due to the reduced elastin content combined with adaptive remodeling during vascular development 17142349_Low levels of elastin is associated with pulmonary emphysema 17626896_developed humanized elastin mouse with elastin production controlled by human elastin gene in bacterial artificial chromosome; expression pattern of human transgene mirrors endogenous murine gene 18083765_Enhanced generation of elastin peptides in S100A4/Mts1 mice may promote increased viral entry in the vessel wall. 18173368_Early elastin expression and organization modify arterial aging through their impact on both vascular cell physiology and structure. 18311803_In MMP-9-deficient animals, vascular inflammation continued to develop, but the incidence of elastin breakdown was significantly reduced. Elastin breakdown in the coronary artery was virtually eliminated by ablation of MMP-9 18441095_TGIF is essential for EGF-mediated downregulation of tropoelastin expression 18495804_LOXL1-KO lower urogenital tract anatomical and functional phenotype resembles female pelvic floor dysfunction in humans. Elastin disorganization may lead to such functional abnormalities. 18506002_At atherosclerosis-susceptible vascular branch points, the absence of a luminal elastin barrier and the presence of a dense collagen/proteoglycan matrix contribute to increased retention of LDL 18772328_Elastin insufficiency predisposes to elevated pulmonary circulatory pressures through changes in elastic artery structure. 19372465_Reduced elastin in mice leads to adaptive remodeling, whereas the complete lack of elastin leads to pathological remodeling and death. 20495146_Elastin is only necessary for normal cardiovascular structure and function in mice starting in the last few days of fetal development. 20576933_Elastin insufficiency in a mouse model establishes a role for elastin dysregulation in aortic valve pathogenesis. 20847053_Oxidative and nitrosative modifications of tropoelastin prevent elastic fiber assembly. 21281809_Elastin degradation might be necessary but is not sufficient to induce arterial medial calcification. 21305018_miR-29 and miR-15 family miRNAs are involved in the down-regulation of elastin in the adult aorta. 21356372_TGF-beta suppresses elastin degradation by inhibiting plasmin-mediated matrix metalloproteinase 9 activation. 21536846_Eln(+/-) mice have decreased aortic diameter and compliance in ex vivo tests that are significant by postnatal day 7 21707943_C3 and C4 bind to collagen and elastin in the vascular wall and have a potential role in vascular stiffness and atherosclerosis 21730037_Report accelerated fatigue-induced damage to or protease-related degradation of initially competent elastic elastin fibres in fibrillin-1 deficiency that renders arteries in Marfan syndrome increasingly susceptible to dilatation, dissection, and rupture. 21856305_MMP-12 leads to elastin degradation in eosinophilic meningitis caused by Angiostrongylus cantonensis. 22049077_Genetic modifiers of cardiovascular phenotype caused by elastin haploinsufficiency act by extrinsic noncomplementation. 22223197_Macrophage-derived macrophage metalloelastase-12 regulates elastin degradation even in progressive experimental liver fibrosis. 22573328_the C-terminal region of tropoelastin in has a critical role in elastic fiber assembly and suggest tissue-specific differences in the elastin assembly pat 22685574_Data show that tropoelastin staining was relatively weak in the ligamentum flavum from E15 through P0, P7 was the first stage that staining intensity was observed to be substantially stronger, intensity remained relatively high until P35. 23261250_tested the hypothesis that adhesive strength varies with atherosclerotic plaque composition of collagen and elastin in apoE and MMP12 knock outs 23857752_Elastin haploinsufficiency impedes the progression of arterial calcification in MGP-deficient mice. 24036114_Two sides of MGP null arterial disease: chondrogenic lesions dependent on transglutaminase 2 and elastin fragmentation associated with induction of adipsin. 24282586_Fstl1 is crucial for elastin expression and deposition in mesenchyme during lung alveologenesis 24291458_The increased levels of elastin, type V collagen and tenascin C are probably the result of increased expression by fibroblastic cells; reversely, elastin influences myofibroblast differentiation. 24322348_Compared to control SMCs, the modulus of Eln-/- SMCs is reduced by 40%, but is unchanged in Fbln4-/- SMCs. The Eln-/- SMC modulus is rescued by soluble or alpha elastin treatment. 24414067_Eln insufficiency induced hypertension is due to increased sensitivity of the resistance vasculature to circulating ANG II and to impaired endothelium-dependent vasodilatation. 25086405_Data indicate significantly reduced volumetric density of elastin and collagen and thinner skin dermis were observed in Marfan mice. 25131766_Data suggest that expression of elastin in uterus, vagina, and bladder is down-regulated both in naturally aging mice and in mouse model of accelerated ovarian aging; such down-regulation may lead to pelvic floor disorders. 25428696_Lung histology revealed aberrant elastin production and impaired lung septation in oxygen-exposed lungs, while tropoelastin, integrin alphav, fibulin-1, fibulin-2 and fibulin-4 gene expression were elevated. 25539853_These results suggest that elastin haploinsufficiency adversely impacts pulmonary angiogenesis. 25923353_Eln was ubiquitously present, with enrichment in regions with cardiomyocyte differentiation, while there was an inverse correlation between ColI and cardiomyocyte differentiation. 26982166_Elevations of whole lung HMGB1 level were associated with impaired alveolar development and aberrant elastin production in 85% O2-exposed newborn lungs. 27183603_Elastin-Derived Peptides Promote Abdominal Aortic Aneurysm Formation by Modulating M1/M2 Macrophage Polarization 28254817_Deficient circumferential growth is the predominant mechanism for moderate obstructive aortic disease resulting from partial elastin deficiency in Williams syndrome. 28751568_mTOR-sensitive perturbation of smooth muscle cell mechanosensing contributes to elastin aortopathy. 28754555_Elastin insufficiency triggers structural defects and abnormal remodeling of renal vascular signaling involving AT1R-mediated vascular mechanotransduction and renal hyperfiltration with increased blood pressure sensitivity to dietary sodium contributing to systolic hypertension. 29366776_findings strongly suggested that elastin crosslinking and LOXL1 were co-associated with liver cirrhosis, while selective inhibition of LOXL1 arrested disease progression by reducing crosslinking of elastin. 29802129_Data (including data from studies in mutant mice and cells from such mice) suggest that elastin-derived peptides are involved in regulation of lipid storage in hepatocytes; thus, elastin-derived peptides may play role in development and progression of non-alcoholic fatty liver. 31282949_Tropoelastin: an in vivo imaging marker of dysfunctional matrix turnover during abdominal aortic dilation. 31590613_Document an unexpected heterogeneity in vascular cells that produce elastin and their roles in formation of the elastic laminae. 31691186_The Elastin-Derived Peptide VGVAPG Does Not Activate the Inflammatory Process in Mouse Cortical Astrocytes In Vitro. 32412588_Whole exome sequencing in patients with Williams-Beuren syndrome followed by disease modeling in mice points to four novel pathways that may modify stenosis risk. 32533648_Elastin homeostasis is altered with pelvic organ prolapse in cultures of vaginal cells from a lysyl oxidase-like 1 knockout mouse model. 33035438_Elastin haploinsufficiency in mice has divergent effects on arterial remodeling with aging depending on sex. 33108351_Neutrophilic inflammation during lung development disrupts elastin assembly and predisposes adult mice to COPD. 35964009_Upregulation of key genes Eln and Tgfb3 were associated with the severity of cardiac hypertrophy. |
ENSG00000049540 |
ELN |
129.802631 |
0.4605817219 |
-1.118471 |
0.35237440 |
9.581254 |
0.00196574064417410999602853216572384553728625178337097167968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0083824640235330479670627923383108281996101140975952148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
76.6661525359366 |
18.9122423462219 |
166.455047797635 |
30.1650916430196 |
| ENSMUSG00000029687 |
14056 |
Ezh2 |
protein_coding |
Q61188 |
FUNCTION: Polycomb group (PcG) protein. Catalytic subunit of the PRC2/EED-EZH2 complex, which methylates (H3K9me) and 'Lys-27' (H3K27me) of histone H3, leading to transcriptional repression of the affected target gene. Able to mono-, di- and trimethylate 'Lys-27' of histone H3 to form H3K27me1, H3K27me2 and H3K27me3, respectively. Displays a preference for substrates with less methylation, loses activity when progressively more methyl groups are incorporated into H3K27, H3K27me0 > H3K27me1 > H3K27me2. Compared to EZH1-containing complexes, it is more abundant in embryonic stem cells and plays a major role in forming H3K27me3, which is required for embryonic stem cell identity and proper differentiation. The PRC2/EED-EZH2 complex may also serve as a recruiting platform for DNA methyltransferases, thereby linking two epigenetic repression systems. Genes repressed by the PRC2/EED-EZH2 complex include HOXA7, HOXB6 and HOXC8. EZH2 can also methylate non-histone proteins such as the transcription factor GATA4 and the nuclear receptor RORA. Regulates the circadian clock via histone methylation at the promoter of the circadian genes. Essential for the CRY1/2-mediated repression of the transcriptional activation of PER1/2 by the CLOCK-BMAL1 heterodimer; involved in the di and trimethylation of 'Lys-27' of histone H3 on PER1/2 promoters which is necessary for the CRY1/2 proteins to inhibit transcription. {ECO:0000250|UniProtKB:Q15910, ECO:0000269|PubMed:12689588, ECO:0000269|PubMed:15516932, ECO:0000269|PubMed:15520282, ECO:0000269|PubMed:16717091, ECO:0000269|PubMed:18086877, ECO:0000269|PubMed:19026780}. |
3D-structure;Alternative splicing;Biological rhythms;Chromatin regulator;Chromosome;Glycoprotein;Isopeptide bond;Methyltransferase;Nucleus;Phosphoprotein;Reference proteome;Repressor;S-adenosyl-L-methionine;Transcription;Transcription regulation;Transferase;Ubl conjugation |
|
|
mmu:14056; |
chromatin [GO:0000785]; chromatin silencing complex [GO:0005677]; chromosome, telomeric region [GO:0000781]; cytoplasm [GO:0005737]; ESC/E(Z) complex [GO:0035098]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; pericentric heterochromatin [GO:0005721]; pronucleus [GO:0045120]; synapse [GO:0045202]; chromatin binding [GO:0003682]; chromatin DNA binding [GO:0031490]; DNA binding [GO:0003677]; histone H3K27 methyltransferase activity [GO:0046976]; histone H3K27 trimethyltransferase activity [GO:0140951]; histone lysine N-methyltransferase activity [GO:0018024]; histone methyltransferase activity [GO:0042054]; lncRNA binding [GO:0106222]; primary miRNA binding [GO:0070878]; promoter-specific chromatin binding [GO:1990841]; protein-lysine N-methyltransferase activity [GO:0016279]; ribonucleoprotein complex binding [GO:0043021]; RNA binding [GO:0003723]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II core promoter sequence-specific DNA binding [GO:0000979]; sequence-specific DNA binding [GO:0043565]; transcription cis-regulatory region binding [GO:0000976]; transcription corepressor activity [GO:0003714]; transcription corepressor binding [GO:0001222]; B cell differentiation [GO:0030183]; cardiac muscle hypertrophy in response to stress [GO:0014898]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to trichostatin A [GO:0035984]; cerebellar cortex development [GO:0021695]; DNA methylation [GO:0006306]; epidermal cell differentiation [GO:0009913]; facultative heterochromatin formation [GO:0140718]; G1 to G0 transition [GO:0070314]; G1/S transition of mitotic cell cycle [GO:0000082]; hemopoiesis [GO:0030097]; hepatocyte homeostasis [GO:0036333]; heterochromatin formation [GO:0031507]; hippocampus development [GO:0021766]; histone H3-K27 trimethylation [GO:0098532]; histone modification [GO:0016570]; immunoglobulin heavy chain V-D-J recombination [GO:0071707]; keratinocyte differentiation [GO:0030216]; liver regeneration [GO:0097421]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; negative regulation of DNA-binding transcription factor activity [GO:0043433]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of epidermal cell differentiation [GO:0045605]; negative regulation of G1/S transition of mitotic cell cycle [GO:2000134]; negative regulation of gene expression [GO:0010629]; negative regulation of gene expression, epigenetic [GO:0045814]; negative regulation of keratinocyte differentiation [GO:0045617]; negative regulation of retinoic acid receptor signaling pathway [GO:0048387]; negative regulation of stem cell differentiation [GO:2000737]; negative regulation of striated muscle cell differentiation [GO:0051154]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of transcription elongation by RNA polymerase II [GO:0034244]; positive regulation of cell cycle G1/S phase transition [GO:1902808]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of dendrite development [GO:1900006]; positive regulation of epithelial to mesenchymal transition [GO:0010718]; positive regulation of GTPase activity [GO:0043547]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of protein serine/threonine kinase activity [GO:0071902]; protein localization to chromatin [GO:0071168]; regulation of cell population proliferation [GO:0042127]; regulation of circadian rhythm [GO:0042752]; regulation of gene expression [GO:0010468]; regulation of gliogenesis [GO:0014013]; regulation of neurogenesis [GO:0050767]; regulation of protein phosphorylation [GO:0001932]; regulation of transcription by RNA polymerase II [GO:0006357]; response to estradiol [GO:0032355]; response to tetrachloromethane [GO:1904772]; rhythmic process [GO:0048511]; skeletal muscle satellite cell maintenance involved in skeletal muscle regeneration [GO:0014834]; stem cell differentiation [GO:0048863]; subtelomeric heterochromatin formation [GO:0031509]; synaptic transmission, GABAergic [GO:0051932] |
12123576_Eed and a second Polycomb group protein, Enx1, are directly localized to the inactive X chromosome in XX trophoblast stem (TS) cells 12496962_Ezh2-dependent histone H3 methylation as a novel regulatory mechanism controlling Igh rearrangement during early murine B cell development. 12900441_Data show that the loss of maternal enhancer of zeste 2 has a long-term effect causing severe growth retardation of neonates despite 'rescue' through embryonic transcription from the paternal allele. 15520282_Ezh2 expression was developmentally regulated in the myotome compartment of mouse somites and that its down-regulation coincided with activation of muscle gene expression and differentiation of satellite-cell-derived myoblasts. 15536069_showed that E2F6, DP1, EPC1, EZH2, and Sin3B co-elute, suggesting the identification of a novel E2F6 complex that exists in vivo in both normal and transformed human cell lines 15882624_Results show the existence of the cytosolic Ezh2-containing methyltransferase complex and tie the function of this complex to regulation of actin polymerization in various cell types. 16009709_Enzyme responsible for methylating lysine 27 of histone H3 at the interleukin(IL)4-IL13 locus of T helper type 1 but not Th2 cells, implicating EZH2 in the mechanism of IL4 and IL13 transcriptional silencing. 16025120_STAT5 functions at a distinct step in regulating distal V(H) recombination in relation to the transcription factor Pax5 and histone methyltransferase Ezh2 16717091_EZH2 is important for the maintenance of circadian rhythms and has a role in the core clockwork mechanism of mammals 17344414_Study shows that the ability of the oncogene BMI1 to repress the INK4A-ARF locus requires its direct association and is dependent on the continued presence of the EZH2-containing Polycomb-Repressive Complex 2 complex. 18281287_up-regulation of miR-26a acts to post-transcriptionally repress Ezh2, a known suppressor of skeletal muscle cell differentiation. 18687996_High levels of Ezh2 in differentiating NSCs appeared to be associated with an increase in oligodendrocytes and a reduction in astrocytes, whereas low levels of Ezh2 led to completely opposite effects. 18713946_MYC stimulates EZH2 expression by repression of its negative regulator miR-26a 18848501_The Polycomb group proteins Ezh2 and Rnf2 are independently required for genomic contraction and imprinted silencing. 19026781_Ezh2 maintains repressive chromatin through different mechanisms. 19079346_EZH2 is important in ER-negative breast cancer growth in vivo and in vitro, and BRCA1 is required for the proliferative effects of EZH2 19303854_Study shows that Ezh2 controls proliferative potential of basal progenitors by repressing the Ink4A-Ink4B locus and tempers the developmental rate of differentiation. 19390090_Enhancer of zeste homolog 2 represses Ink4a/Arf in islet beta cells 19575012_Ezh2 are significantly downregulated during murine skin repair. 19661437_EZH2 overexpression resulted in aberrant terminal end bud architecture and intraductal epithelial hyperplasia resembling the human counterpart 19709408_EZH2 is overexpressed in BRCA1-deficient mouse mammary tumors. EZH2 is responsible for repression of genes driving differentiation. BRCA1-deficient cells are dependent on EZH2, whereas BRCA1-proficient cells are not. 19717977_Targeting of EZH2 to a specific genomic site is sufficient for recruitment of Dnmt3a, however, additional events are required for de novo DNA methylation. 19818710_MiR-214 and Ezh2 establish a regulatory loop controlling PcG-dependent gene expression during differentiation. 19843474_Liver fibrosis is regulated by an epigenetic relay pathway that includes MeCP2, EZH2, and miR132. 20078939_Ezh2 represses Ngn1 expression, which ensures the normal differentiation of P19 cells. 20144788_identify polycomb-like 2 and Polycomb Repressive Complex 2 transcriptional targets and draft regulatory networks.[Polycomb Repressive Complex 2 and polycomb-like 2] 20368440_H3K27 methyltransferase Ezh2 directly represses Wnt genes to facilitate adipogenesis; acetylation and trimethylation on H3K27 play opposing roles in regulating Wnt expression 20395008_Ezh2 plays an essential role in the maintenance of both the proliferative and self-renewal capacity of hepatic stem/progenitor cells and the full execution of their differentiation 20708159_increase in endothelial EZH2 is a direct result of VEGF stimulation by a paracrine circuit that promotes angiogenesis by methylating and silencing vasohibin1 (vash1) 20798045_found that the histone methyltransferase of polycomb repressive complex 2 (PCR2), enhancer of Zeste homolog 2 (Ezh2), is essential for controlling the rate at which development progresses within cortical progenitor cell lineages 20814569_elevated levels of TNFalpha and NF-kappaB inhibit the regenerative potential of satellite cells via epigenetic silencing of the Notch-1 gene 20951942_Loss of the SNF5 tumor suppressor leads to elevated expression of the Polycomb gene EZH2. 20979531_Data suggest that overexpression of Ezh2 alone is insufficient for a complete dedifferentiation of astrocytes to neural stem cells. 21123648_An Ezh2 domain comprising T345 was found to be important for binding to HOTAIR and the 5' end of Xist 21317239_while Ezh1/2-null hair follicles (HFs) arrest morphogenesis and degenerate due to defective proliferation and increased apoptosis, epidermis hyperproliferates and survives engraftment 21439249_HBx up-regulates mEZH2 expression by transactivating the mEZH2 promoter through E2F1 transcription factor. 21490431_Ezh2 directly regulates the epigenetic status of the Nanog promoter affecting the balance of Nanog expression in pluripotent stem cells, and therefore the equilibrium between self-renewal and differentiation. 21498568_EZH2 controls self-renewal and proliferation, and maintains an appropriate transcriptional program in skeletal muscle stem cells. 21659531_a novel function for CDK1-mediated Ezh2 phosphorylation and provide a mechanism by which Ezh2 protein levels can be regulated in cells. 21674483_Data show that Mel-18 and Ezh2 positively regulate the expression of Il17a and Il17f. 21757686_The KDM2B-let-7-EZH2 pathway contributes to the proliferation of immortal Ink4a/Arf null fibroblasts suggesting the histone-modifying enzyme functions more broadly in the regulation of cellular proliferation. 21761357_Developmental programming of EZH2 is a novel mechanism by which in utero exposure to endocrine disruptors leads to epigenetic regulation of the mammary gland. 21795281_Ezh2 maintains the late phase of Hox gene expression and cell transposition experiments suggest that it regulates the plasticity with which cells respond to instructive positional cues. 21941025_The results implicate EZH2-driven mechanisms by which Myc may stimulate prostate tumor initiation and disease progression. 22023227_very small embryonic-like stem cells maintain their pluripotent state through an Ezh2-dependent bivalent domain-mediated epigenetic mechanism 22042701_Data show that deletion of Ezh2 in adult BM, did not significantly compromise hematopoiesis. 22158708_Inactivation of the PRC2 subunit Ezh2 by Nkx2-5(Cre) (Ezh2(NK)) caused lethal congenital heart malformations, namely, compact myocardial hypoplasia, hypertrabeculation, and ventricular septal defect. 22267199_Ezh2-mediated repression of Six1 in differentiating cardiac progenitors is essential for stable gene expression and homeostasis in the postnatal heart. 22279536_Data indicate that binding of EZH2, SUZ12 and EED, the 3 subunits of Plybomb repressive complex 2 (PRC2), is required for PRC2 full activity, and late during viral infection, a significant increase in PRC2 protein association with chromatin. 22312437_a wide spectrum of cardiovascular malformations in the Ezh2 mutant mice 22323620_In the testis, the apical region of the round spermatid nucleus could be a specialized epigenetic region where methylation of histones serves a role in the spermiogenic chromatin remodeling and that Ezh2 might be a key effector of this event. 22391448_EZH2 is required for tissue repair by promoting the regenerative proliferation of progenitor cells 22396593_The role of polycomb repressive complex 2 (PRC2) function in leukemia development and progression, is reported. 22431509_studies document that deregulation of EZH2 and associated genes leads to the development of mouse, and likely human adult T-cell acute leukemia 22465358_Data show that pEZH2 was found concentrated in the liver Mallory-Denk bodies (MDB). 22662197_EZH2 plays a role in the migration and proliferation of PASMCs, which is a major hallmark in Pulmonary Arterial Hypertension (PAH). 22805308_the synergy of androgen receptor and Kras signaling could elevate EZH2 expression and expand prostate cancer progenitor cells in vivo. 22808153_The epigenetic repressor Ezh2 in neural stem cells is crucial for proliferative activity and maintenance of neural stemness. 22983009_EZH2 is directly involved in transcriptional repression of MyoD, the main factor promoting myogenesis. 23038103_EZH2 and ANCCA are androgen regulated and strongly expressed in early prostate morphogenesis and during puberty, suggesting their important role in prostate development. 23203812_Ezh2 does not regulate the muscle differentiation process in vivo. Results emphasise the lineage and cell-type-specific functions of Ezh2 and Polycomb repressive complex 2. 23307742_Ezh2-dependent epigenetic regulation of intrinsic and extrinsic transcriptional programs controls topographic neuronal guidance and connectivity in the cortico-ponto-cerebellar pathway 23349306_The trithorax protein partner menin acts in tandem with EZH2 to suppress C/EBPalpha and differentiation in MLL-AF9 leukemia. 23375371_Ezh2 couples hormonal stimuli to epigenetic changes that underpin progenitor activity, lineage specificity, and alveolar expansion in the mammary gland. 23624931_The chromodomain helicase DNA-binding protein 4 (Chd4) is a critical interaction partner of Ezh2 required specifically for Polycomb group (PcG)-mediated suppression of the key astrogenic marker gene GFAP. 23673358_Deletion of Ezh1 and Ezh2 from skin results in a marked change in fate determination in epidermal progenitor cells, leading to an increase in the number of lineage-committed Merkel cells, specialized subtype of skin cells involved in mechanotransduction. 23680150_EZH2 represses proliferation checkpoint genes and helps establish bivalent chromatin domains at key regulatory loci to transiently suppress GC B cell differentiation. 23712803_EZH2 is required for the differentiation of luminal progenitors to alveolar cells. 23733190_Data indicate that during Mycobacterium bovis BCG infection, iNOS/NO responsive KLF4 induces EZH2 and miR-150 functions to regulate CIITA expression. 23880518_When the SET domain of Ezh2, a core subunit of the PRC2 complex, was deleted,the repression of Stag3 and Smc1beta during embryoid body differentiation was largely impaired, indicating that the event required the enzymatic activity of Ezh2 23982173_Gain-of-function mutations in EZH2 are frequently observed in B-cell lymphoma, implying disease-dependent effects of individual mutations. 24141370_as a gene silencer, Ezh2 was required to promote the expression of transcription factors Tbx21 and Stat4. 24141703_Using in vivo RNA-protein cross-linking, authors show that EZH2 directly binds the 5' region of nascent RNAs transcribed from a subset of these promoters and that these binding events correlate with decreased H3K27me3. 24153105_Ezh2 is essential for early preimplantation embryo development through the regulation of epigenetic modification and apoptosis. 24200695_Ezh2 inactivation in GC B cells caused impairment of GC responses, memory B cell formation, & humoral immunity. EZH2 protected them from activation-induced cytidine deaminase mutagenesis, facilitated cell cycle progression, & silenced BLIMP1. 24216481_transgene expression of Ezh2 was sufficient to increase beta cell replication and regeneration in young adult mice 24218139_Deletion of Ezh2 alone was enough to induce MDS/MPN-like diseases. Furthermore, concurrent depletion of Ezh2 and Tet2 established more advanced myelodysplasia and markedly accelerated the development of myelodysplastic disorders. 24238339_Ezh2 inactivation enhances Th1/2 differentiation & plasticity. Ezh2 controlled Tbx21 & Gata3 expression in these cells accompanied by trimethylation at lys-27 of histone 3. Ezh2 loss worsened an allergic asthma model & caused memory Th2 accumulation. 24325319_results demonstrate that Ezh2 is critical for efficient iPS cell generation, whereas it is dispensable for maintaining the reprogrammed iPS cell state. 24496623_Ezh2 is required for neural crest-derived cartilage and bone formation. 24553933_downregulation of Ezh2 transcription by NFIB is an important component of the process of neural progenitor cell differentiation during cortical development 24719098_Deletion of Ezh2 inhibits neurogenesis in hippocampus of adult mice and impairs spatial learning. 24737471_EZH2 suppresses hepatocellular differentiation of mouse bone marrow mesenchymal stem cells. 24760151_Ezh2 was also required to prevent proteasome-mediated degradation of T-bet protein in Th1 cells. 24802772_Data indicate a dramatic acceleration of lymphoma development in the combination model of Myc and histone methyltransferase EZH2(Y641F). 24878960_overexpression of Ezh2 mRNA with a 3'-untranslated region (3'-UTR) lacking a functional miR-124 binding site, but not with the wild-type Ezh2 3'-UTR, hampered neuronal and promoted astrocyte-specific differentiation 24953053_Ezh2 loss significantly promotes RUNX1S291fs-induced myelodysplastic syndrome but prevents the transformation of acute myeloid leukaemia. 25085702_These results suggest that miR-214 may induce pathologic cardiac hypertrophy in part by reducing EZH2 messenger RNA levels. 25153170_Ezh2 is required for the hepatic progenitor expansion in vivo, which is essential for the functional maturation of embryonic liver. 25189741_EZH2 modulates angiogenesis in vitro and in a mouse model of limb ischemia. 25266721_c-Rel regulates Ezh2 expression in lymphocytes and malignant lymphoid cells in a novel transcriptional network 25359725_Epigenetic repression of Creb3l1, Fosl1, Klf5 and Mmp9 by Ezh2 in endothelial cells maintains the integrity of the developing vasculature. 25431950_Pharmacologic downregulation suppresses bladder cancer in vivo 25477280_Loss of EZH2 is associated with increased liver injury and a blunted regenerative response. 25501223_Study showed that Ezh2 mRNA expression was inversely correlated with Oct4 and Sox2, but Oct4 and Sox2 negatively affect Ezh2; additionally, Oct4 and Sox2 were negative regulators of Ezh2 primarily on a post-translation level. 25516972_Ezh2 restricts the basal cell lineage during normal lung endoderm development to allow the proper patterning of epithelial lineages during lung formation. 25556712_Study used Ezh2 conditional knockout mice to clarify the role of histone H3K27me3 modification in the retina 25680271_These studies reveal a critical role for Ezh2 in the maintenance of Regulatory T cell identity during cellular activation. 25687282_Ezh2 is required for coupling of T cell receptor specificity to natural killer T cell generation. 25751747_regulates adhesion dynamics, leukocyte migration and immune responses 25790853_work reveals an unexpected role for Ezh2 in controlling basal cell fate determination in the embryonic lung endoderm, mediated in part by repression of Igf1 expression. 25989023_Findings reveal a conserved role for EZH2 in constraining the expression of potent developmental regulators to maintain lineage integrity and retinal progenitor proliferation, as well as regulating the timing of late differentiation. 25998860_Results show that EZH2 promotes mammary stem and luminal progenitor cell expansion, metastasis and inhibits ER-positive cellular differentiation. 26090605_The dichotomous function of EZH2 in regulating differentiation and senescence in effector and regulatory T cells helps to explain the apparent existing contradictions in literature. 26219303_These results clearly demonstrated an essential role of Ezh1 in the pathogenesis of hematopoietic malignancies induced by Ezh2 insufficiency, and highlighted the differential functions of Ezh1 and Ezh2 in hematopoiesis. 26250110_The presence of EZH2 is required to control progressive differentiation of milk secreting epithelium during pregnancy. 26307668_Study demonstrated that redundant EZH2 shifted mesenchymal stem cell lineage commitment to adipocyte, which contributed to the development of osteoporosis. 26324324_The study characterized the binding position of Ezh2 and menin at all annotated genes in embryonic stem cells and B and T lymphocytes. 26517694_Phosphorylation-mediated EZH2 inactivation promotes drug resistance in multiple myeloma. 26621269_Conditional deletion of Ezh2 in the developing midbrain resulted in decreased neural progenitor proliferation, which is associated with derepression of cell cycle inhibitors and negative regulation of Wnt/beta-catenin signaling. 26668377_Data indicate increased natural killer (NK) lineage cells in Ezh2 (enhancer of zeste homolog 2)-deficient mice. 26693692_This study identified Ezh2 as enriched in melanoma cancer stem cells (MCS cells) and showed that Ezh2 knockdown or treatment with small molecule Ezh2 inhibitors reduces MCS cell spheroid formation, survival, invasion and migration. 26766588_Modulation of Polycomb repressive complexes by either Ezh2 overexpression or Eed deletion enhances KRAS-driven adenomagenesis and inflammation 26773568_Ezh2 cooperates with let-7 microRNAs in silencing the fetal gene signature in BM HSPCs and restricts their transformation. 26904942_Genetic inactivation of Ezh2 or Eed cooperates with NRASQ61K in leukemogenesis. 26994968_in the skin epithelium, EED, Suz12, and Ezh1/2 function largely as subunits of the PRC2 complex and have roles in skin development 27068104_These findings indicate Ezh2/PRC2 plays crucial roles in regulating neurogenesis from both cerebellar germinal zones. 27081096_results suggest that loss of Ezh2 cooperates with Jak2V617F in the development of myelofibrosis in Jak2V617F 27150236_we found that the miRNA biogenesis enzyme DICER was required for the binding of the PRC2 core components EZH2 and SUZ12, and for the presence of the PRC2-mediated histone modification H3K27me3 at many bivalent genes 27151441_S6K1 phosphorylation of H2B mediates EZH2 trimethylation of H3 early in adipogenesis, contributing to the promotion of obesity. 27226494_EZH2 directly interacted with and stabilized BubR1, an effect driving EZH2 into the concert of meiosis regulation. 27239797_A long noncoding RNA, lnc-beta-Catm, elicits EZH2-dependent beta-catenin stabilization and sustains liver cancer stem cells self-renewal. 27250566_decline in HDAC9c expression over time was accompanied by increased EZH2 expression. 27295295_The findings obtained from apoE-/- mice provide epigenetic insights into how EZH2 increases the risk of atherosclerotic heart disease. One of the pathways by which EZH2 leads to lipid accumulation and foam cell formation is via epigenetic downregulation of ABCA1 expression. 27401344_JAK2-V617F-expressing mice treated with an Ezh2 inhibitor showed higher platelet counts than vehicle controls. Our data support the proposed tumor suppressor function of EZH2 27401345_an EZH2 insufficiency not only cooperated with active JAK2 to induce MF, but also conferred an oncogenic addiction to the H3K27ac modification in primary myelofibrosis -initiating cells that was capable of being restored by bromodomain inhibition. 27419533_the functional association between EZH2 expression and silencing of key tumor suppressor loci , was investigated. 27465826_The Ezh2 expression could be clearly seen in outer and inner hair cells, supporting cells, the stria vascularis, and spiral ganglion cells. Ezh2 continued to be expressed in the cochlear epithelium of adult mice. 27539752_the induction of EZH2 led to beta-catenin signaling activation by increasing H3K27me3 on the promoter of SFRP1, while the inhibition of EZH2 silenced beta-catenin signaling. Finally, intraarticular injection of EPZ005687 delayed OA development in mice. These results implicated EZH2 activity in OA development. Pharmacological inhibition of EZH2 may be an effective therapeutic approach for osteoarthritis. 27578795_Findings uncover an Ezh2-dependent mechanism to restrict the smooth muscle gene expression program in the developing mesothelium and allow appropriate cell fate decisions to occur in this multipotent mesoderm lineage. 27585866_Our data uncover a dual role of PRC2 in intestinal homeostasis highlighting the importance of this repressive layer in controlling cell plasticity and lineage choices in adult tissues 27694924_These findings indicate that Ezh2 targets are the major targets of the epigenetic switch in MDS with Ezh2 insufficiency. 27728805_N-Myc Induces an EZH2-Mediated Transcriptional Program Driving Neuroendocrine Prostate Cancer and redirects EZH2 activity and sensitizes cells to EZH2 inhibitors. 27758858_these data suggest that inhibition of Ezh2 promotes paracrine signaling in osteoblasts a 27807665_ompounding a previously described Bmi1-transgene and Pten-deficiency prostate cancer mouse model with the Ezh2 transgene did not enhance tumour progression or drive metastasis formation. In conclusion, we here report the generation of a wildtype Ezh2 overexpression mouse model that allows for intravital surveillance of tissues with activated transgene 27823969_Suggest that EZH1 and -2 are novel targets of miR-214-3p, and miR-214-3p might be one potential miRNA for the prevention of cardiac fibrosis. 27869166_our results revealed a novel mechanism in which EZH2 stability is regulated by SKP2 through the TRAF6-mediated and K63-linked ubiquitination, which contributes to elevated levels of H3K27me3 during prostate tumorigenesis and CRPC growth. 27889452_The evidence has been presented that the retinoblastoma protein utilizes a cell-cycle-independent interaction with E2F1 to recruit EZH2 to diverse repeat sequences. 27897169_The combined loss of Ezh1 and Ezh2 in chondrocytes severely impairs skeletal growth in mice. 27909059_these results suggest that the inflammatory milieu found in Crohn's disease could lead to or result from deregulation of FOXP3/EZH2-enforced T cell gene networks contributing to the underlying intestinal inflammation. 27926872_EZH2-deficient hESCs can initiate differentiation toward developmental lineages; however, they cannot fully differentiate into mature specialized tissues. Thus, EZH2 is required for stable ESC self-renewal, regulation of transcriptional programs, and for late-stage differentiation in this model of early human development. 27934660_Ezh2-/- deletion resulted in skeletal deformities, increased osteoclastogenic potential and upregulation of Rankl and M-csf expression by mesenchymal stem cells. 27936205_The levels of EZH2 and H3K27me3 were increased in the aorta of ApoE-/- mice fed a high-methionine diet for 16 weeks, whereas miR-92a expression was decreased. 27984727_demonstrate that a specific interaction between the polycomb protein EZH2 and RNA made from B2 SINE retrotransposons controls stress-responsive genes in mouse cells. In the heat-shock model, B2 RNA binds stress genes and suppresses their transcription. Upon stress, EZH2 is recruited and triggers cleavage of B2 RNA. B2 degradation in turn upregulates stress genes. 28049658_Eed and Ezh2 have distinct roles in urothelial differentiation 28067271_Praja1 promotes skeletal myogenesis through degradation of EZH2 upon p38alpha activation. 28215965_Menin binds on the promoter of Inhbb gene where it favours the recruitment of Ezh2 via an indirect mechanism involving Akt-phosphorylation. 28223321_we propose that through its control of PLZF concentration, Ezh2 maintains NKT cell numbers at appropriate levels and facilitates immune homeostasis. We believe that our study establishes a paradigm for alternative roles of Ezh2 and non-histone methylation in the regulation of cell fate determination. 28246193_These findings identify the Ezh2-Hsp90 interaction as a previously unrecognized mechanism essential for T-cell responses and an effective target for controlling graft-versus-host disease. 28249716_EZH2 is part of the protein machinery that shapes the aging epigenome. 28263309_Residual PRC2 activity, via EZH2 binding to mutant H3K27M histones, is required for the proliferation of H3K27M-expressing diffuse intrinsic pontine gliomas. 28329683_Gfi1 disruption antagonized the tumor-promoting effects of Ezh2 loss; conversely, Gfi1 overexpression collaborated with Myc to bypass effects of Trp53 inactivation in driving medulloblastoma progression in primary cerebellar neuronal progenitors. 28338656_It has been demonstrated that FAK depletion reduces hepatocellular carcinoma cell growth by affecting cancer-promoting genes including the pro-oncogene EZH2. 28410989_Data show that enhancer of zeste 2 polycomb repressive complex 2 subunit (EZH2-containing PRC2) is required for differentiation of terminal effector CD8+ T cells. 28439030_findings highlight an epigenetic mechanism by which EZH2 integrates the multifaceted effects of TNFalpha signaling to promote the inflammatory response and apoptosis in colitis. 28490575_results uncover a crucial role for EZH2 in adaptive lymphocytes to control the developmental timing of effectors of the pre-Ag receptor checkpoints 28602614_miR-203 is repressed by EZH2 in both embryonic and adult neural stem/progenitor cells (NSPCs). MiR-203 negatively regulates the proliferation of NSPCs. One of PRC1 components, Bmi1, is a downstream target of miR-203 in NSPCs. 28666321_Insight regarding how androgen-induced extranuclear kinase signaling and intranuclear transcription through Ezh2 modifications may influence the expression pattern of genes, ultimately affecting various downstream physiological processes. 28701722_EZH2 deficiency leads to growth retardation. 28720764_ABCG2 de-repression induced by EZH2 mutations have crucial roles in MDS pathogenesis. 28807939_High EZH2 expression is associated with Neuroblastoma. 28855249_Muscle-enriched, long noncoding RNA Chronos mediates skeletal muscle hypertrophy. 28963737_Long non-coding RNA LOC554202 may play an important role in the progression of chordoma by the direct upregulation of EZH2 and indirect promotion of RNF144B via miR-31 28982932_EZH2 plays a nonclassical role in the regulation of spermatogonial differentiation and apoptosis in murine spermatogenesis. 28992292_macroH2A1.1 (mH2A1.1), a variant of histone H2A, was upregulated during adipocyte differentiation in 3T3-L1 cells and in the white adipose tissue of obese mice. mH2A1.1 regulated Wnt/beta-catenin signaling pathway by cooperating with EZH2, a histone H3K27 methyltransferase, thus led to accumulation of H3K27me2 and H3K27me3 on the promoters of Wnt genes. 29026085_EZH2 mediates germinal centre (GC) formation through epigenetic silencing of CDKN1A and release of cell cycle checkpoints. 29033324_Biochemical as well as functional experiments revealed that Spt6 could compete for binding of the PRC2 methyltransferase Ezh2 to Suz12 and reduce PRC2 chromatin engagement. 29087303_PRC2-mediated repression of SMARCA2 predicts EZH2 inhibitor activity in SWI/SNF mutant tumors. 29101351_Ezh2 maintains the repression of key cell senescence inducer genes through H3K27me3, and deletion of Ezh2 in early pubertal mice results in premature cellular senescence, depleted MSPCs pool, and impaired osteogenesis as well as osteoporosis in later life. 29132376_Our data demonstrates that EZH2 participates in Glioblastoma multiforme-induced immune deficient and EZH2 suppression in Glioblastoma multiforme can remodel microglia immune functions 29212856_High EZH2 expression is associated with Tamoxifen Resistance in Breast Cancer. 29242551_Data show that the histone methyltransferase Ezh2 controls CD8(+) T memory precursor formation and antitumor activity. 29288200_the data demonstrate that EZH2 is essential in facilitating epigenetic changes that regulate antibody-secreting cell fate, function, and metabolism. 29408885_results, together with our previous report, support a cell lineage-specific mechanism of Ezh2-mediated gene repression, especially those critically involved in cellular function and homeostasis 29434212_CARM1 promotes EZH2-mediated silencing of EZH2/BAF155 target tumor suppressor genes by methylating BAF155. 29438697_Ezh2 and Runx1 mutations collaborate to initiate lympho-myeloid leukemia in early thymic progenitors 29445149_Let-7c inhibits cholangiocarcinoma growth but promotes tumor cell invasion and growth at extrahepatic sites at least in part, through regulation of EZH2 protein expression and through the DVL3/beta-catenin axis. 29466371_cardiomyocyte-specific loss of Ezh2 did not affect fibrotic scar size after MI or apical resection at P7, suggesting that it does not extend the regenerative time window. Our results demonstrate that Ezh2 is not required for innate neonatal cardiac regeneration 29530603_Our data show that chemical inhibition of the H3K27me3 demethylases Jmjd3/Utx blunts Neurogenin3-dependent gene activation in vitro. Conversely, inhibition of the H3K27me3 methyltransferase Ezh2 enhances both the transactivation ability of Neurogenin3 in cultured cells and the formation of insulin-producing cells during directed differentiation from pluripotent cells 29606589_Nsd1 is a key modulator of PRC2 function required for regulating the demarcation of genome-wide H3K27me2 and H3K27me3 domains in embryonic stem cells. 29626115_these findings establish Ezh2 as a macrophage lineage-specific mediator of autoimmune inflammation and highlight a previously unknown mechanism of Ezh2 function. 29632530_Here we show that loss of Ezh2 resulted in altered expression of several key cyclin-dependent kinase inhibitors including Cdkn2a (p16 and Arf) and Cdkn1c (p57) in naive CD8+ T cells after activation, which explained the cell cycle impairment of Ezh2-deficient naive CD8+ T cells in response to antigenic stimulation. 29729398_The data imply that Ezh2 is epistatic to Gli3 and suggest the possibility that hedghehog activation is repressed by the recruitment of polycomb repressive complex 2. 29789597_The current study was to determine the role of methyltransferase EZH2 and its catalysed H3K27 trimethylation (H3K27me3) in liver failure. 29899112_results suggest that Ezh2 serves a bifunctional role during bone formation by suppressing osteogenic lineage commitment while simultaneously facilitating proliferative expansion of osteoprogenitor cells. 29959321_Targeting EZH2 reactivates a breast cancer subtype-specific anti-metastatic transcriptional program driven by FOXC1. 30008323_Gain of EZH2 promotes loss of primary cilia in benign melanocytic lesions, enhancing pro-tumorigenic WNT/beta-catenin signaling, and is itself sufficient to drive metastatic melanoma in benign cells. 30022044_p38-mediated phosphorylation at threonine 367 induces EZH2 cytoplasmic localization to promote breast cancer metastasis. 30051352_TSPYL2 is recruited to promoters of specific EZH2 target genes in neurons. 30080177_The loss of Ezh2 in p53-null hematopoietic cells impeded the differentiation of early T cell precursors (ETPs) and eventually induced early T cell precursor acute lymphoblastic leukemia (ETP-ALL)-like disease, indicating that the polycomb repressive complex 2 functions as a tumor suppressor in ETPs. 30169530_Ezh2 is an important epigenetic modifier for the enteric neural crest cell development through repression of Zic1, Pax3, and Sox10. 30197001_lncRNA LINC00968 accelerates the proliferation and fibrosis of mesangial cells by epigenetically repressing p21 via recruiting EZH2 in diabetic nephropathy 30213970_network analysis of the central circadian pathway genes linked their expression to the up regulated expression of the histone methyltransferase gene EZH2, a gene that when mutated in both humans and mice controls overall skeletal growth 30222957_Exogenous RA signaling activation in the Ezh2 mutants leads to synergistic activation of the anti-osteogenic factors in the cranial mesenchyme in vivo. Thus, RA signaling and EZH2 can function in parallel to guide calvarial bone progenitor commitment by balancing the suppression of anti-osteogenic factors. 30295189_Findings |
ENSG00000106462 |
EZH2 |
1208.538963 |
2.0837483611 |
1.059181 |
0.10397530 |
101.693466 |
0.00000000000000000000000648138132057150265468843759760208983744318487343266643911962571851168024572587000875500962138175964355468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000003312899338085407029216419805714363738806132374083978915473014392745199074852280318737030029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1624.96752962023 |
117.213118929564 |
779.829061883989 |
42.6755480078072 |
| ENSMUSG00000029752 |
27053 |
Asns |
protein_coding |
Q61024 |
|
Acetylation;Amino-acid biosynthesis;Asparagine biosynthesis;ATP-binding;Glutamine amidotransferase;Ligase;Nucleotide-binding;Phosphoprotein;Reference proteome |
PATHWAY: Amino-acid biosynthesis; L-asparagine biosynthesis; L-asparagine from L-aspartate (L-Gln route): step 1/1. |
|
mmu:27053; |
cytosol [GO:0005829]; asparagine synthase (glutamine-hydrolyzing) activity [GO:0004066]; ATP binding [GO:0005524]; identical protein binding [GO:0042802]; asparagine biosynthetic process [GO:0006529]; cellular response to glucose starvation [GO:0042149]; cellular response to hormone stimulus [GO:0032870]; glutamine metabolic process [GO:0006541]; L-asparagine biosynthetic process [GO:0070981]; liver development [GO:0001889]; negative regulation of apoptotic process [GO:0043066]; positive regulation of mitotic cell cycle [GO:0045931]; response to amino acid [GO:0043200]; response to follicle-stimulating hormone [GO:0032354]; response to light stimulus [GO:0009416]; response to mechanical stimulus [GO:0009612]; response to organic cyclic compound [GO:0014070]; response to toxic substance [GO:0009636] |
24292060_Asns is involved in the regulation of plasma glucose levels. 31421261_Asparagine Synthetase Is Highly Expressed at Baseline in the Pancreas Through Heightened PERK Signaling. 32573712_Association of aberrant ASNS imprinting with asparaginase sensitivity and chromosomal abnormality in childhood BCP-ALL. 33822775_Coordination of asparagine uptake and asparagine synthetase expression modulates CD8+ T cell activation. 35857457_Tumoral microenvironment prevents de novo asparagine biosynthesis in B cell lymphoma, regardless of ASNS expression. |
ENSG00000070669 |
ASNS |
1408.435446 |
2.2605042819 |
1.176645 |
0.10116672 |
133.528800 |
0.00000000000000000000000000000069270495912779734202271552494357852198397963032487584824821680710117221002619387199006339983498037327080965042114257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000555330628286032005513426766424857735882877776861520757827802118667036812527885297763319272235094103962182998657226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1960.63785491546 |
137.599343382206 |
867.345339983487 |
46.3844062088535 |
| ENSMUSG00000029810 |
65963 |
Tmem176b |
protein_coding |
Q9R1Q6 |
FUNCTION: May play a role in the process of maturation of dendritic cells (By similarity). Required for the development of cerebellar granule cells. {ECO:0000250, ECO:0000269|PubMed:16814752}. |
Differentiation;Membrane;Nucleus;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:65963; |
nuclear membrane [GO:0031965]; dendritic cell differentiation [GO:0097028]; negative regulation of dendritic cell differentiation [GO:2001199] |
16814752_The Clast1 gene is ubiquitously expressed in various organs of adult mice and is required for development of cerebellar granule cells. 20501748_These data suggest that Tmem176B and Tmem176A associate to form multimers and restrain dendritic cell maturation. 20550525_data identify the downregulation of Clast1/LR8 as a potentially important mechanism by which oncogenic Ras-mediated neoplastic transformation occurs 24085390_Tmem176b induces the differentiation of myoblasts into an osteoblast lineage. 27009467_Study characterized Tmem176a and b expression in RORgamma t+ lymphocytes at transcriptional and protein levels and present evidence that both genes exert a redundant ion channel function related to a colocalisation in close proximity to the Golgi apparatus. 29466738_myogenic differentiation is achieved through the specific interaction between Cry2 and Bclaf1, which stabilizes mRNAs encoding cyclin D1, a G1/S phase transition regulator, and Tmem176b, a transmembrane regulator for myogenic cell fusion. 34233909_Dendritic Cells Require TMEM176A/B Ion Channels for Optimal MHC Class II Antigen Presentation to Naive CD4(+) T Cells. |
ENSG00000106565 |
TMEM176B |
511.916238 |
3.7129360456 |
1.892560 |
0.12345038 |
232.064199 |
0.00000000000000000000000000000000000000000000000000021143835744719902450723282800176981012765699616557964984949755329515819271229892995996107987781687955235071294610662443703054193841310570700642301744665019214153289794921875000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000392759641540943345377456263400642945356364056784341701581273127458839326728813098367869819058972973226894920027297524677442857980735890777168606291525065898895263671875000000000000000000000000000000000000000000 |
Yes |
No |
816.323356846992 |
69.9374813653614 |
219.859256075531 |
14.9816285661216 |
| ENSMUSG00000029814 |
140488 |
Igf2bp3 |
protein_coding |
Q9CPN8 |
FUNCTION: RNA-binding factor that may recruit target transcripts to cytoplasmic protein-RNA complexes (mRNPs). This transcript 'caging' into mRNPs allows mRNA transport and transient storage. It also modulates the rate and location at which target transcripts encounter the translational apparatus and shields them from endonuclease attacks or microRNA-mediated degradation. Preferentially binds to N6-methyladenosine (m6A)-containing mRNAs and increases their stability (By similarity). Binds to the 3'-UTR of CD44 mRNA and stabilizes it, hence promotes cell adhesion and invadopodia formation (By similarity). Binds to beta-actin/ACTB and MYC transcripts (By similarity). Increases MYC mRNA stability by binding to the coding region instability determinant (CRD) and binding is enhanced by m6A-modification of the CRD (By similarity). Binds to the 5'-UTR of the insulin-like growth factor 2 (IGF2) mRNAs. {ECO:0000250|UniProtKB:O00425, ECO:0000269|PubMed:15753088}. |
3D-structure;Cytoplasm;Isopeptide bond;mRNA transport;Nucleus;Phosphoprotein;Reference proteome;Repeat;RNA-binding;Translation regulation;Transport;Ubl conjugation |
|
|
mmu:140488; |
cytoplasm [GO:0005737]; cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; nucleus [GO:0005634]; P-body [GO:0000932]; mRNA 3'-UTR binding [GO:0003730]; mRNA 5'-UTR binding [GO:0048027]; mRNA binding [GO:0003729]; N6-methyladenosine-containing RNA binding [GO:1990247]; CRD-mediated mRNA stabilization [GO:0070934]; mRNA transport [GO:0051028]; nervous system development [GO:0007399]; regulation of gene expression [GO:0010468]; regulation of RNA metabolic process [GO:0051252]; regulation of translation [GO:0006417] |
26974154_IGF2BP3 regulated transcripts via targeting elements within 3' untranslated regions (3'UTR) 31221665_Lin28b and Igf2bp3 are at the center of a gene regulatory network that mediates the fetal-adult hematopoietic switch 31894303_Circular RNA_101237 mediates anoxia/reoxygenation injury by targeting let7a5p/IGF2BP3 in cardiomyocytes. 34321607_The RNA-binding protein IGF2BP3 is critical for MLL-AF4-mediated leukemogenesis. 34402854_The germinal center reaction depends on RNA methylation and divergent functions of specific methyl readers. 35315195_IMP3 promotes re-endothelialization after arterial injury via increasing stability of VEGF mRNAhv. 36463698_Deletion of Insulin-like growth factor II mRNA-binding protein 3 participates in the pathogenesis of recurrent spontaneous abortion by inhibiting IL-10 secretion and inducing M1 polarization. |
ENSG00000136231 |
IGF2BP3 |
439.419533 |
3.0895795092 |
1.627411 |
0.14082497 |
130.870146 |
0.00000000000000000000000000000264345102236728656813002989991108659644487945871190709945445173763158314183733090321770653474686696426942944526672363281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000002086271812056917251406593241048493505001896585630159679179203106790473597198509736472260556183755397796630859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
654.422506719354 |
63.9812617978984 |
211.816043176063 |
16.2278938185136 |
| ENSMUSG00000029816 |
93695 |
Gpnmb |
protein_coding |
Q99P91 |
FUNCTION: Could be a melanogenic enzyme. {ECO:0000250}. |
Cell membrane;Endosome;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:93695; |
cytoplasmic vesicle [GO:0031410]; early endosome membrane [GO:0031901]; melanosome membrane [GO:0033162]; membrane [GO:0016020]; plasma membrane [GO:0005886]; heparin binding [GO:0008201]; integrin binding [GO:0005178]; receptor ligand activity [GO:0048018]; syndecan binding [GO:0045545]; bone mineralization [GO:0030282]; cell adhesion [GO:0007155]; cell-cell signaling [GO:0007267]; negative regulation of cytokine production [GO:0001818]; negative regulation of G1/S transition of mitotic cell cycle [GO:2000134]; negative regulation of neuron death [GO:1901215]; negative regulation of T cell activation [GO:0050868]; negative regulation of T cell proliferation [GO:0042130]; negative regulation of tumor necrosis factor production [GO:0032720]; osteoblast differentiation [GO:0001649]; positive regulation of cell migration [GO:0030335]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of protein autophosphorylation [GO:0031954]; positive regulation of protein phosphorylation [GO:0001934]; regulation of tissue remodeling [GO:0034103]; signal transduction [GO:0007165] |
12638126_mRNA of this gene is linked to the developing retinal pigment epithelium and iris in the embryo 14696968_OA is expressed in late mouse embryonic development in the nervous system, basal layer of the skin, germinal cells of hair follicles, and in the forming nephrons of the kidney 16100390_osteoactivin might function as an activator for fibroblasts infiltrated into denervated skeletal muscles and play an important role in regulating degeneration/regeneration of extracellular matrix 17284525_Endogenous DC-HIL is a negative regulator of T lymphocyte activation 17475886_GPNMB acts as a negative regulator of macrophage inflammatory responses. 17608931_the role of Gpnmb in mechanisms involved in the subsequent IOP elevation and optic nerve degeneration remain unclear 17947650_Syndecan-4 is the T cell ligand through which DC-HIL mediates its negative coregulatory function 18036345_These results suggest that the extracellular fragments of osteoactivin produced by shedding act as a growth factor to induce MMP-3 expression via the ERK pathway in fibroblasts. 18049427_Both OA-D and OA-E peptides significantly induced osteoblast differentiation in vitro and that effect is RGD independent. 18313864_The inclusion of gpnmb in the MITF regulon suggests a role for GPNMB in mature osteoclast function. 18381073_These findings indicate that osteoactivin is a novel osteoclastic protein and plays a role in osteoclast differentiation and/or activity. 18402690_affects of Gpnmb on innate immunity influence susceptibility to glaucoma in DBA/2J mice 18555216_Osteoactivin acts as a positive regulator of osteoblastogenesis. 18983539_Gpnmb expression can be used as a marker for analyzing melanocyte development and disease progression. 19320736_Gpnmb is a melanosome-associated glycoprotein that contributes to the adhesion of melanocytes with keratinocytes. 19794069_Our findings support a dual role for DC-HIL: inhibition of adaptive immunity following ligation of syndecan-4 on activated T cells and induction of innate immunity against dermatophytic fungi. 20506259_OA plays an important role in bone formation and serves as a positive regulator of fracture healing 20570888_DC-HIL/Gpnmb confers upon melanoma the ability to downregulate the activation of melanoma-reactive T cells, thereby allowing melanoma to evade immunologic recognition and destruction. 20709912_Gpnmb is a novel prorepair gene that is necessary for crosstalk between the macroautophagic degradation pathway and phagocytosis. 20711474_ADAM10 as a sheddase capable of releasing the GPNMB/OA ectodomain from the surface of breast cancer cells 21029394_GPNMB influences the chemical composition of melanosomes. 21398278_R150X mutation in Gpnmb dramatically modified its list of genetic covariates, which may explain the associated ocular pathology. 22536365_Osteoclast-derived osteoactivin is a novel stimulator of osteoclast activity and bone resorption. 22891158_GPNMB inhibits motor neuron death and plays a critical role in motor neuron survival. 23113638_SD-4, as the T-cell ligand of DC-HIL, is a potent inhibitor of allo-reactive T cells responsible for GVHD and a potentially useful target for treating this disease. 24265122_our results suggest that OA is able to induce transdifferentiation of myoblasts into osteoblasts through increasing levels of phosphorylated FAK. 24415158_Osteoactivin is a matricellular protein that stimulates osteoblast adhesion through binding to alphav beta1 integrin and cell surface heparan sulfated proteoglycans, resulting in increased cell spreading, actin reorganization, and osteoblast differentiation. 24516197_Results indicate that the DC-HIL/syndecan-4 (SD-4) pathway regulates autoimmune responses by mediating the T cell suppressor function of myeloid-derived suppressor cells (MDSCs). 24936834_DC-HIL is the critical mediator of melanoma cells' suppressor function 25010402_GPNMB has protective effect against ischemia-reperfusion injury via phosphorylation of ERK1/2 and Akt 25054912_Glycoprotein nonmetastatic melanoma protein B plays an important role in angiogenesis during hyperoxia injury 25545823_The present study, using mice, is the first to demonstrate that GPNMB improves both spatial and non-spatial memory 25899717_that osteoactivin overexpression has an osteoinductive effect on bone mass in vivo and stimulates osteoprogenitor differentiation ex vivo 26442636_M2 macrophages express significantly greater levels of OA/GPNMB than pro-inflammatory M1 macrophages. 26458492_macrophages infiltrating injured mucosa express Gpnmb, Gpnmbpositive macrophages may ameliorate inflammation in the intestinal mucosa. 26581806_The GPNMB is a promising biomarker and therapeutic target for the development and progression of Nonalcoholic fatty liver disease in obesity. 26599547_Gpnmb-positive macrophages infiltrate the liver during the recovery phase of CCl4-induced acute liver injury. 26771826_Gpnmb Is a Potential Marker for the Visceral Pathology in Niemann-Pick Type C Disease 26880751_Finally, we identify glycoprotein NMB as a melanocytic marker up-regulated in Tsc2-null mouse uteri and human lymphangioleiomyomatosis samples 26988030_Study identified the Na+/K+-ATPase alpha 1 and 3 subunits as receptors for the extracellular fragment of GPNMB that mediates activation of cellular signaling pathways and subsequent neuroprotective effects. 27011731_Genetic modulators of IOP thus may be independently identified using the full array of BXD mice without concern for the presence of transillumination defect or mutations in Typr1 and/or Gpnmb. 27585719_Osteoactivin is a negative regulator of osteoclastogenesis in vitro and in vivo and this process is regulated through CD44 and ERK activation. 27836549_findings indicate that GPNMB promotes glioma growth via Na(+)/K(+)-ATPase alpha subunits. Thus, the interaction between GPNMB and Na(+), K(+)-ATPase alpha subunits represents a novel therapeutic target for the treatment of brain glioblastomas. 28148779_This is the first experimental study to show that GPNMB adversely influences myocardial remodeling. 28899684_Study using an in vivo model of cutaneous wound healing in mice provide evidence that Gpnmb is functionally important in promoting the recruitment of mesenchymal stem cells to the site of skin injury, which in turn modulates inflammatory responses by directing the M2 polarization of macrophages in acute wound healing. 28939899_Under the endoplasmic-reticulum stress conditions, GPNMB relocated to the nucleus and specifically up-regulated expression of BiP at the mRNA level by promoting the BiP pre-mRNA splicing, not through the pathways initiated by the three major transducers of the unfolded protein response: IRE1, PERK, and ATF6. 29857071_this study shows that DC-HIL promotes metastasis by excluding T-Lymphocytes from the pre-metastatic niches 31057322_The results suggest that Stc1 may be a downstream candidate of Gpnmb, and that both genes interact with other genes in a network to develop glaucoma through mechanisms such as apoptosis and oxidative stress. 32436108_Transgenic Overexpression of GPNMB Protects Against MPTP-Induced Neurodegeneration. 32694855_Gpnmb secreted from liver promotes lipogenesis in white adipose tissue and aggravates obesity and insulin resistance. 33986446_Quantitative trait locus mapping identifies the Gpnmb gene as a modifier of mouse macrophage lysosome function. 34464706_Genetic ablation of Gpnmb does not alter synuclein-related pathology. 34582891_GPNMB plays a protective role against obesity-related metabolic disorders by reducing macrophage inflammatory capacity. 34608215_Anti-inflammatory role of Gpnmb in adipose tissue of mice. 34695452_GPNMB mitigates Alzheimer's disease and enhances autophagy via suppressing the mTOR signal. 35289033_Tumor endothelial cell-induced CD8(+) T-cell exhaustion via GPNMB in hepatocellular carcinoma. 35444208_Glycoprotein nonmetastatic melanoma protein B regulates lysosomal integrity and lifespan of senescent cells. 35809343_Role of Gpnmb in atherosclerosis of female mice. |
ENSG00000136235 |
GPNMB |
2270.421299 |
2.0581004640 |
1.041313 |
0.16954665 |
36.670421 |
0.00000000139885957500215721994319653160741487307738850631722016260027885437011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000193527965907655402660907844117343934442487807245925068855285644531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2994.80140967043 |
352.762097694422 |
1455.12887346626 |
128.608203576203 |
| ENSMUSG00000029838 |
19242 |
Ptn |
protein_coding |
P63089 |
FUNCTION: Secreted growth factor that mediates its signal through cell-surface proteoglycan and non-proteoglycan receptors (By similarity). Binds cell-surface proteoglycan receptor via their chondroitin sulfate (CS) groups (By similarity). Thereby regulates many processes like cell proliferation, cell survival, cell growth, cell differentiation and cell migration in several tissues namely neuron and bone (PubMed:15121180, PubMed:30497772, PubMed:27445335, PubMed:19442624). Also plays a role in synaptic plasticity and learning-related behavior by inhibiting long-term synaptic potentiation (PubMed:11414790, PubMed:25000129). Binds PTPRZ1, leading to neutralization of the negative charges of the CS chains of PTPRZ1, inducing PTPRZ1 clustering, thereby causing the dimerization and inactivation of its phosphatase activity leading to increased tyrosine phosphorylation of each of the PTPRZ1 substrates like ALK or AFAP1L2 in order to activate the PI3K-AKT pathway (PubMed:27445335). Through PTPRZ1 binding controls oligodendrocyte precursor cell differentiation by enhancing the phosphorylation of AFAP1L2 in order to activate the PI3K-AKT pathway (PubMed:27445335). Forms a complex with PTPRZ1 and integrin alpha-V/beta-3 (ITGAV:ITGB3) that stimulates endothelial cell migration through SRC dephosphorylation and activation that consequently leads to ITGB3 'Tyr-773' phosphorylation (By similarity). In adult hippocampus promotes dendritic arborization, spine development, and functional integration and connectivity of newborn granule neurons through ALK by activating AKT signaling pathway (PubMed:30497772). Binds GPC2 and chondroitin sulfate proteoglycans (CSPGs) at the neuron surface, leading to abrogation of binding between PTPRS and CSPGs and neurite outgrowth promotion (By similarity). Binds SDC3 and mediates bone formation by recruiting and attaching osteoblasts/osteoblast precursors to the sites for new bone deposition (By similarity). Binds ALK and promotes cell survival and cell proliferation through MAPK pathway activation (By similarity). Inhibits proliferation and enhances differentiation of neural stem cells by inhibiting FGF2-induced fibroblast growth factor receptor signaling pathway (PubMed:15121180). Mediates regulatory mechanisms in normal hemostasis and in hematopoietic regeneration and in maintaining the balance of myeloid and lymphoid regeneration (PubMed:21791434). In addition may play a role in the female reproductive system, auditory response and the progesterone-induced decidualization pathway (PubMed:17121547, PubMed:28657144, PubMed:16619002). {ECO:0000250|UniProtKB:P21246, ECO:0000250|UniProtKB:P63090, ECO:0000269|PubMed:11414790, ECO:0000269|PubMed:15121180, ECO:0000269|PubMed:16619002, ECO:0000269|PubMed:17121547, ECO:0000269|PubMed:19442624, ECO:0000269|PubMed:21791434, ECO:0000269|PubMed:25000129, ECO:0000269|PubMed:27445335, ECO:0000269|PubMed:28657144, ECO:0000269|PubMed:30497772}. |
Disulfide bond;Growth factor;Heparin-binding;Mitogen;Reference proteome;Secreted;Signal |
|
|
mmu:19242; |
basement membrane [GO:0005604]; cell surface [GO:0009986]; cytoplasm [GO:0005737]; endoplasmic reticulum [GO:0005783]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; membrane [GO:0016020]; neuromuscular junction [GO:0031594]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; postsynapse [GO:0098794]; presynapse [GO:0098793]; protein-containing complex [GO:0032991]; Schaffer collateral - CA1 synapse [GO:0098685]; carbohydrate binding [GO:0030246]; chondroitin sulfate binding [GO:0035374]; chondroitin sulfate proteoglycan binding [GO:0035373]; glycosaminoglycan binding [GO:0005539]; growth factor activity [GO:0008083]; heparan sulfate binding [GO:1904399]; heparin binding [GO:0008201]; integrin binding [GO:0005178]; molecular function activator activity [GO:0140677]; protein kinase binding [GO:0019901]; protein phosphatase inhibitor activity [GO:0004864]; proteoglycan binding [GO:0043394]; syndecan binding [GO:0045545]; vascular endothelial growth factor binding [GO:0038085]; bone mineralization [GO:0030282]; decidualization [GO:0046697]; dendrite arborization [GO:0140059]; dendrite regeneration [GO:0031104]; estrous cycle [GO:0044849]; integrin-mediated signaling pathway [GO:0007229]; learning [GO:0007612]; leukocyte chemotaxis involved in inflammatory response [GO:0002232]; memory [GO:0007613]; modulation of chemical synaptic transmission [GO:0050804]; negative regulation of angiogenesis [GO:0016525]; negative regulation of cell migration [GO:0030336]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of long-term synaptic potentiation [GO:1900272]; negative regulation of membrane potential [GO:0045837]; negative regulation of mesenchymal cell proliferation [GO:0072201]; negative regulation of neuroblast proliferation [GO:0007406]; negative regulation of neuromuscular junction development [GO:1904397]; oogenesis [GO:0048477]; ossification [GO:0001503]; ossification involved in bone remodeling [GO:0043932]; positive regulation of apoptotic process [GO:0043065]; positive regulation of axon regeneration [GO:0048680]; positive regulation of bone mineralization [GO:0030501]; positive regulation of cell differentiation [GO:0045597]; positive regulation of cell division [GO:0051781]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of dendrite development [GO:1900006]; positive regulation of hepatocyte proliferation [GO:2000347]; positive regulation of leukocyte chemotaxis [GO:0002690]; positive regulation of neuron projection development [GO:0010976]; positive regulation of oligodendrocyte differentiation [GO:0048714]; positive regulation of ossification [GO:0045778]; positive regulation of skeletal muscle acetylcholine-gated channel clustering [GO:1904395]; positive regulation of stem cell differentiation [GO:2000738]; receptor clustering [GO:0043113]; regulation of cell shape [GO:0008360]; regulation of endothelial cell migration [GO:0010594]; regulation of hemopoiesis [GO:1903706]; regulation of myelination [GO:0031641]; regulation of stem cell population maintenance [GO:2000036]; regulation of synaptic plasticity [GO:0048167]; response to auditory stimulus [GO:0010996]; retinal rod cell differentiation [GO:0060221]; tissue regeneration [GO:0042246] |
12093164_Mice overexpressing HB-GAM learned faster in mazes and showed decreased anxiety, while HB-GAM knockouts showed opposite behavior. HB-GAM may suppress LTP in hippocampus and influence learning-related behavior. 12107166_role of anaplastic lymphoma kinase and pleiotrophin signaling in apoptosis 12112370_Pleiotrophin mRNA is strongly induced in the brain by day 7 after injury, which correlates with the expression pattern of syndecan-3 mRNA. 12412809_we propose that PTN is an accessory signaling molecule, which is involved in a variety of processes in bone formation 12413943_roles during in vivo bone formation and remodeling, probably acting as a co-factor or accessory protein that modulates the effects of primary signaling molecules 14708942_Pleiotrophin and Midkine are differently expressed and distributed during retinoic acid-induced neural differentiation 15121180_HB-GAM functions as an endogenous inhibitor of FGF-2 in stem cell proliferation in the developing cortex. 15180956_PTN mRNA is highly expressed in neural stem (progenitor) cells of mouse ventral mesencephalon. PTN promotes the production of DAergic neurons from embryonic-stem-cell-derived nestin-positive cells. 15351507_The mRNA transcripts of PTN and its receptors were detected in neurosphere, suggesting that pleiotrophin signaling systems are present in the neural stem cells and are involved in the modulation of fate of neural stem cells. 15369781_These findings suggest that Ptn gene expression has a critical role in determining the levels of expression of the enzymes of catecholamine biosynthesis in aorta. 15485659_Ptn has the potential to critically regulate the downstream activities of angiotensin II, through the regulation of its synthesis by ACE and its receptor mediated functions through regulation of both the AT1 and AT2 receptors 15589206_Ptn-deficient mice do not display detectable defects in endochondral and intramembranous ossification and intramembranous ossification or bone remodeling. 15840658_JUN-dependent expression of PTN and SDF-1 in fibroblasts has a role in keratinocyte proliferation 15985215_midkine regulates pleiotrophin gene expression with a high degree of organ specificity, suggesting that pleiotrophin gene expression follows midkine gene expression in development 16507572_PTN participates in tumorigenesis caused by PTEN loss 16914133_Together, these results demonstrate that influence of CNTF family of cytokines on the differentiation of late retinal progenitor cell population is partially mediated by the release of Ptn. 17121547_Female infertility in mice may result from deficiency in Ptn. 17239862_Knocking down the expression of PTN using siRNA resulted in an increase in phospho-GSK-3beta expression, and the accumulation of nuclear beta-catenin, which are critical downstream signaling proteins for both the PTN and Wnt signaling pathways. 17360581_Pleiotrophin is a neurotrophic factor for spinal motor neurons 17607711_Inhibition of the mitogenic, angiogenic, and tumorigenic activities of Ptn by a synthetic peptide corresponding to its C-thrombospondin repeat-I domain. 17823238_Pleiotrophin is secreted from fibroblasts and has an antiapoptotic effect in these cells and in embryonic stem cells. 17875810_IL-4 and pleiotrophin act as negative and positive angiogenic regulators, respectively. 18582856_These results demonstrate that MK and HB-GAM are involved in stem cells maintenance, cytodifferentiation and mineralization processes during mouse incisor development. 18766019_Data show that N-syndecan, the receptor for the extracellular matrix component heparin-binding growth-associated molecule, is required for survival of primary sensory neurons. 19442624_The results suggest that HB-GAM is an osteocyte-derived factor that could participate in mediating the osteogenic effects of mechanical loading on bone. 19723774_Data show that that lack of periostin in the embryonic OFT leads to ectopic expression of the proosteogenic growth factor pleiotrophin (Ptn) and overexpression of delta-like 1 homolog (Dlk1), a negative regulator of Notch1. 20192945_The data suggest an important role of PTN to limit amphetamine-induced neurotoxic and rewarding effects. 20305662_regulates expansion and regeneration of hematopoietic stem cells 20620199_Gene inactivation of Pleiotrophin results in enhanced amphetamine neurotoxicity in the striatum. 21881556_this study shows that PTN expression is upregulated in a mouse model of peritoneal fibrosis and is present in human peritoneal tissues and in peritoneal dialysate effluent. 21945314_MiR-143 down regulates PTN expression through interaction with a target site of miR-143 in the coding region of mouse PTN. 22342918_identify significant differences between PTN and MK in the control of pain processing at the spinal level, and support the hypothesis that inhibitors of the PTN/MK signaling pathway could potentiate opioid analgesia 22382872_PTN is widely expressed during tooth development and also in the adult mouse molar and incisor. 22438896_alterations of the bone microarchitecture that occurred in wild type and pleiotrophin-transgenic mice exposed to a near-zero gravity on the International Space Station 22672269_Data suggest a physiological role for HARP in modulating (i.e., antagonizing) endogenous anticoagulant activity of heparin-like material in the vasculature. 23077670_We conclude that PTN delays the maturation of the mammary gland by maintaining mammary epithelial cells in a progenitor phenotype and by inhibiting their differentiation during mammary gland development. 23084748_PTN is a secreted component of the bone marrow vascular niche that regulates hematopoietic stem cell self-renewal and retention in vivo. 23178526_the data demonstrate that maintenance of amphetamine-induced conditioning effects depend on the endogenous expression of PTN. 23459167_Pleiotrophin exerts a neuroprotective role against amphetaine-induced neurotoxicity in striatum. 23874826_Vivarium-housed Wt controls had higher levels of active TGF-beta1 and IL-2 in inguinal lymph nodes relative to PTN-Tg mice. 23891830_data suggest that the spinal antinociceptive effects caused by stimulation of alpha2-adrenoceptors are differentially regulated by endogenous expression of PTN 24015201_PTN overexpression exerts various effects on sarcolemma ion channel expression/function and resting cytosolic calcium concentrations in soleus and extensor digitorum longus muscles. 24695857_antagonizes Brd2 promoting cell differentiation associated with the neural tube: spinal cord neurogenesis and neural crest migration 25000129_the absence of PTN in vivo is associated with disruption of specific cognitive and affective processes 25073406_The results of this study indicated that PTN may be a novel genetic factor of importance in alcohol use disorders. 25250571_PTN-mediated hematopoietic regeneration occurs in a RAS-dependent manner. 26645582_findings link pleiotrophin abundance in gliomas with survival in humans and mice, and show that pleiotrophin promotes glioma progression through increased VEGF deposition and vascular abnormalization. 27445335_these data indicate that PTN-PTPRZ-A signaling controls the timing of oligodendrocyte precursor cell differentiation in vivo, in which the CS moiety of PTPRZ receptors maintains them in a monomeric active state until its ligand binding. 27642078_The data demonstrate that PTN overexpression in the brain blocks the conditioning effects of amphetamine and enhances the characteristic striatal dopaminergic denervation caused by this drug 27806344_PTN expression in glioma cells is associated with chromosome 7 gain. PTN enhances PDGFB-induced gliomagenesis by stimulating proliferation of neural progenitor cells. 28259175_The results of this study suggested that regulation of the PTN signaling pathways may constitute new therapeutic opportunities particularly in those neurological disorders characterized by increased PTN cerebral levels and neuroinflammation. 28569747_Molecular crosstalk between tumour-associated macrophages and glioma stem cells through PTN-PTPRZ1 paracrine signalling supports glioblastoma malignant growth. 28657144_Ptn may play a vital role in the progesterone-induced decidualization pathway via C/EBPB-cyclic AMP-Hand2 signaling. 29061398_Study demonstrates that regulation of astroglial responses to lipopolysaccharide administration are highly dependent on the levels of expression of pleiotrophin and midkine. 29262349_NK cells promote fetal development through the secretion of growth-promoting factors, pleiotrophin and osteoglycin. 30081728_Hmgb3 may play an important role during embryo implantation and decidualization. Pleiotropin is a downstream target of Hmgb3 in the differentiation of uterine stromal cells. 30100167_The Pleiotrophin (PTN) expression is increased in bone marrow endothelial cells (BMECs), and PTN(+) ECs are more frequent in the niche. 30327824_Results reveal for the first time that pleiotrophin is a key player in preserving insulin sensitivity, driving the dynamics of adipose tissue lipid turnover and plasticity, and regulating energy metabolism and thermogenesis. 30497772_Adult hippocampal neural stem cells continuously supply pleiotrophin factor to newborn neurons. 30643194_A new oncofetal lncRNA Ptn-dt is identified. 31613796_Mechanistically, PTN promoted chronic myelogenous leukemia stem cell survival and TKI resistance via induction of Jun and the unfolded protein response. 34502170_Pleiotrophin Deficiency Induces Browning of Periovarian Adipose Tissue and Protects against High-Fat Diet-Induced Hepatic Steatosis. 34551152_Deletion of pleiotrophin impairs glucose tolerance and liver metabolism in pregnant mice: Moonlighting role of glycerol kinase. 35250852_Pleiotrophin Expression and Actions in Pancreatic beta-Cells. 36279670_Clematichinenoside AR inhibits the pathology of rheumatoid arthritis by blocking the circPTN/miR-145-5p/FZD4 signal axis. |
ENSG00000105894 |
PTN |
11502.089666 |
3.0785732978 |
1.622262 |
0.05174725 |
941.123905 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001127708782699338933252945233050625079322716907124480300874 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000190858446423070317134267345663618358711227295597565309457 |
Yes |
No |
17539.5994842093 |
629.253298723337 |
5697.31423863767 |
154.991516077906 |
| ENSMUSG00000029847 |
243753 |
Slc23a4 |
protein_coding |
D3Z3R5 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
|
plasma membrane region [GO:0098590]; L-ascorbic acid transmembrane transporter activity [GO:0015229] |
|
|
|
29.192668 |
2.5039830175 |
1.324225 |
0.48200014 |
7.407618 |
0.00649482514331248435290877552006350015290081501007080078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0232064688207684159670929346930279280059039592742919921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.8423235024702 |
13.5602225784993 |
15.9115789623151 |
4.21948882657952 |
| ENSMUSG00000029851 |
232748 |
Tcaf2 |
protein_coding |
Q921K8 |
FUNCTION: Negatively regulates the plasma membrane cation channel TRPM8 activity. Involved in the recruitment of TRPM8 to the cell surface. Promotes prostate cancer cell migration stimulation in a TRPM8-dependent manner. {ECO:0000250|UniProtKB:A6NFQ2}. |
Cell membrane;Membrane;Reference proteome;Transport |
Human_homologues NA; + ;NA |
|
mmu:232748; |
cell junction [GO:0030054]; plasma membrane [GO:0005886]; transmembrane transporter binding [GO:0044325]; hematopoietic progenitor cell differentiation [GO:0002244]; negative regulation of anion channel activity [GO:0010360]; positive regulation of protein targeting to membrane [GO:0090314]; regulation of anion channel activity [GO:0010359] |
25559186_Report novel TRP channel-associated factors that modulate TRPM8 activity. |
ENSG00000170379+ENSG00000283528 |
TCAF2+TCAF2C |
95.178025 |
4.5012426267 |
2.170323 |
0.28081157 |
59.310761 |
0.00000000000001346382361884943342717056036705057072208743459973412370800360804423689842224121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000003318462157966255187192672341758112618042772523629935221833875402808189392089843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
156.053607855312 |
30.5670206648643 |
34.6690060496866 |
5.46086066677982 |
| ENSMUSG00000029862 |
12723 |
Clcn1 |
protein_coding |
Q64347 |
FUNCTION: Voltage-gated chloride channel (By similarity). Plays an important role in membrane repolarization in skeletal muscle cells after muscle contraction (Probable) (PubMed:8119941). {ECO:0000250|UniProtKB:P35523, ECO:0000269|PubMed:8119941, ECO:0000305|PubMed:1659665}. |
CBS domain;Cell membrane;Chloride;Chloride channel;Ion channel;Ion transport;Membrane;Phosphoprotein;Reference proteome;Repeat;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
|
mmu:12723; |
chloride channel complex [GO:0034707]; plasma membrane [GO:0005886]; sarcolemma [GO:0042383]; protein homodimerization activity [GO:0042803]; voltage-gated chloride channel activity [GO:0005247]; chloride transmembrane transport [GO:1902476]; chloride transport [GO:0006821]; muscle contraction [GO:0006936]; neuronal action potential propagation [GO:0019227]; regulation of ion transmembrane transport [GO:0034765] |
12150905_Expanded CUG repeats trigger aberrant splicing of ClC-1 chloride channel pre-mRNA and hyperexcitability of skeletal muscle in myotonic dystrophy 12196568_The A331T mutation causes an unprecedented alteration of ClC-1 gating and reveals novel processes defining transitions between open and closed states in ClC chloride channels 12370472_A distinct autosomal recessive myotonic mouse in the C57BL/6 background (line B6MT)is reported in which the Clc-1 gene shows polymorphism with no functional consequences. 16959550_CLC-1 deficiency not only affects muscle relaxation (myotonia) but also modulates diaphragm performance during the contractile phase of the contraction-relaxation cycle 17135300_alternative splicing is a posttranscriptional mechanism regulating chloride conductance during muscle development, and the chloride channelopathy in a transgenic mouse model of DM1 results from a failure to execute a splicing transition for CLCN1. 17158949_These results support a molecular mechanism for myotonia in myotonic dystrophy type 1 in which a reduction in both the number of functional sarcolemmal ClC-1 and maximal channel open probability. 17335846_Clc1 expression was dramatically decreased in Znf9+/- mice. 17483199_genetic CLC-1 chloride channel deficiency in mice not only produces myotonia but also substantially worsens the isotonic contractile performance of diaphragm muscle. 18008009_These observations indicate that the myotonia and chloride channelopathy observed in DM both result from abnormal alternative splicing of ClC-1 21078869_the majority of functional ClC-1 channels localize to the sarcolemma and provide essential insight into the basis of myofiber excitability in normal and diseased skeletal muscle. 23247112_Myotonia in adult human skeletal actin transgenic mice may be explained on the basis of a mosaic expression of ClC-1 channels in different fibres and/or on alterations of other conductances. 23625574_Sex hormones at high concentration can rapidly modulate ClC-1 in mouse skeletal muscle fibers in vitro. 23671115_The expression of the muscle chloride channel, ClC-1, in Huntington disease muscle was compromised by improper splicing and a corresponding reduction in total Clcn1 (gene for ClC-1) mRNA. 23828222_Myotonia (delayed muscle relaxation) is the most commonly observed symptom in DM1 patients and is caused by aberrant splicing of the skeletal muscle chloride channel (CLCN1) gene 26501102_Data show that Muscleblind-like 1 (Mbnl1) and Muscleblind-Like 3 (Mbnl3) bind skeletal muscle chloride channel CIC-1 (Clc-1) mRNA. 32242217_Systemic therapy in an RNA toxicity mouse model with an antisense oligonucleotide therapy targeting a non-CUG sequence within the DMPK 3'UTR RNA. |
ENSG00000188037 |
CLCN1 |
9.334520 |
4.4392126623 |
2.150304 |
0.85904425 |
6.155925 |
0.01309725871552246991014190058422173024155199527740478515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0422753644320487953489617893865215592086315155029296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.7768217601885 |
9.32800626782457 |
3.3287032823858 |
1.63565628467185 |
| ENSMUSG00000029910 |
56150 |
Mad2l1 |
protein_coding |
Q9Z1B5 |
FUNCTION: Component of the spindle-assembly checkpoint that prevents the onset of anaphase until all chromosomes are properly aligned at the metaphase plate (By similarity). In the closed conformation (C-MAD2) forms a heterotetrameric complex with MAD1L1 at unattached kinetochores during prometaphase, and recruits an open conformation of MAD2L1 (O-MAD2) which then promotes the conversion of O-MAD2 to C-MAD2 (By similarity). Required for the execution of the mitotic checkpoint which monitors the process of kinetochore-spindle attachment and inhibits the activity of the anaphase promoting complex by sequestering CDC20 until all chromosomes are aligned at the metaphase plate (By similarity). {ECO:0000250|UniProtKB:Q13257}. |
Acetylation;Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Cytoskeleton;Kinetochore;Mitosis;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:56150; |
chromosome [GO:0005694]; chromosome, centromeric region [GO:0000775]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; kinetochore [GO:0000776]; mitotic checkpoint complex [GO:0033597]; mitotic spindle [GO:0072686]; mitotic spindle assembly checkpoint MAD1-MAD2 complex [GO:1990728]; nuclear pore nuclear basket [GO:0044615]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; spindle pole [GO:0000922]; identical protein binding [GO:0042802]; protein C-terminus binding [GO:0008022]; protein homodimerization activity [GO:0042803]; cell division [GO:0051301]; establishment of centrosome localization [GO:0051660]; establishment of mitotic spindle orientation [GO:0000132]; mitotic sister chromatid segregation [GO:0000070]; mitotic spindle assembly checkpoint signaling [GO:0007094]; negative regulation of apoptotic process [GO:0043066]; negative regulation of mitotic cell cycle [GO:0045930]; negative regulation of mitotic metaphase/anaphase transition [GO:0045841]; negative regulation of protein catabolic process [GO:0042177]; negative regulation of ubiquitin protein ligase activity [GO:1904667]; negative regulation of ubiquitin-protein transferase activity [GO:0051444]; positive regulation of mitotic cell cycle spindle assembly checkpoint [GO:0090267] |
13678590_a functional Mad2-dependent spindle checkpoint exists during the first meiotic division in mammalian oocytes 15549134_Data show that the loss of Trrap leads to chromosome missegregation, mitotic exit failure and compromised mitotic checkpoints, which are caused by defective Trrap-mediated transcription of the mitotic checkpoint proteins Mad1 and Mad2. 15563594_that Brca1(Delta11/Delta11) cells displayed decreased expression of a number of genes that are involved in the spindle checkpoint, including Mad2 16600213_Data suggest that Mad2 and BubR1 must cooperate to inhibit Cdc20 activity. 16924662_Our results provide further evidence for the role of MAD2 as a spindle checkpoint protein in mouse oocytes. 17038523_Mad2 is involved in synergistic growth of immature hematopoietic progenitor cells in response to stem cell factor plus Granulocyte-Macrophage Colony-Stimulating Factor 20173739_tumours that experience transient Mad2 overexpression and consequent chromosome instability recur at markedly elevated rates 20870947_Pcid2 is essential for B cell survival through the regulation of MAD2 expression during B cell differentiation 21216274_Data show that Mad2 haploinsufficiency is protective in the presence of a cycle-specific DNA synthesis agent in vivo, and Ape1/Ref-1 inhibitor in vitro. 21665145_Mad2 is also repressed by p53 and its upregulation is required for chromosome instability in a p53 mutant tumor model 24133140_Exacerbating chromosome missegregation in CENP-E+/- mice by reducing levels of another mitotic checkpoint component, Mad2, results in elevated cell death and decreased tumor formation compared with reduction of either protein alone. 25332286_study shows that Mad2 is a novel substrate for CCP6 in megakaryocytes; Mad2 polyglutamylation plays a crucial role in the regulation of megakaryopoiesis 26544187_p31comet-induced cell death is mediated by interactions with Mad2 27292643_Mad2-positive tumors have a higher frequency of developing persistent subclones that avoid remission and continue to grow. 28506992_reduced MAD2 levels attenuate the apoptotic response to mis-segregating sex chromosomes and allow the formation of aneuploid sperm. 29147457_immunofluorescence staining for mitotic arrest deficient 2-like 1 and the protein kinase TTK, components of the spindle assembly checkpoint (SAC), suggested that this delay possibly involved SAC activation. 33420244_Acute systemic loss of Mad2 leads to intestinal atrophy in adult mice. 36247080_Inhibition of MAD2L1 Mediates Pulmonary Fibrosis through Impairment of Mitochondrial Function and Induction of Cell Senescence. |
ENSG00000164109 |
MAD2L1 |
2484.321118 |
2.3715929286 |
1.245856 |
0.16099237 |
58.571132 |
0.00000000000001960723876331523815162127803999926442177475366390737576693936716765165328979492187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000004755692051637225375314150048491812960895636086533500019868370145559310913085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3496.09094877703 |
390.943905996179 |
1474.1530499062 |
123.878401867166 |
| ENSMUSG00000029925 |
21391 |
Tbxas1 |
protein_coding |
P36423 |
FUNCTION: Catalyzes the conversion of prostaglandin H2 (PGH2) to thromboxane A2 (TXA2), a potent inducer of blood vessel constriction and platelet aggregation. Cleaves also PGH2 to 12-hydroxy-heptadecatrienoicacid (12-HHT) and malondialdehyde, which is known to act as a mediator of DNA damage. 12-HHT and malondialdehyde are formed stoichiometrically in the same amounts as TXA2. Additionally, displays dehydratase activity, toward (15S)-hydroperoxy-(5Z,8Z,11Z,13E)-eicosatetraenoate (15(S)-HPETE) producing 15-KETE and 15-HETE. {ECO:0000250|UniProtKB:P24557}. |
Endoplasmic reticulum;Fatty acid biosynthesis;Fatty acid metabolism;Heme;Iron;Isomerase;Lipid biosynthesis;Lipid metabolism;Lyase;Membrane;Metal-binding;Monooxygenase;Oxidoreductase;Prostaglandin biosynthesis;Prostaglandin metabolism;Reference proteome;Transmembrane;Transmembrane helix |
Human_homologues PATHWAY: Lipid metabolism; fatty acid metabolism. {ECO:0000305}. |
|
mmu:21391; |
cytosol [GO:0005829]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; 12-hydroxyheptadecatrienoic acid synthase activity [GO:0036134]; heme binding [GO:0020037]; hydroperoxy icosatetraenoate dehydratase activity [GO:0106256]; iron ion binding [GO:0005506]; monooxygenase activity [GO:0004497]; oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen [GO:0016705]; thromboxane-A synthase activity [GO:0004796]; cellular chloride ion homeostasis [GO:0030644]; icosanoid metabolic process [GO:0006690]; positive regulation of vasoconstriction [GO:0045907]; prostaglandin biosynthetic process [GO:0001516]; response to ethanol [GO:0045471]; response to fatty acid [GO:0070542] |
18006634_minimal level of protein kinase A activity is required for the steroidogenic effect of the TBXAS inhibitor and that inhibition of TBXAS activity or its expression increase the steroidogenic sensitivity of MA-10 mouse Leydig cells to cAMP stimulation 19735918_thromboxane synthase, prostacyclin synthase and thromboxane receptor have roles in atherosclerotic lesions and correlate with plaque composition 20383787_TXAS mRNA expression was increased within the vascular wall in mouse models of atherosclerosis with advanced lesions. 24009185_12-HHT is produced by both TxAS-dependent and TxAS-independent pathways in vitro and in vi 26974824_the administration of salted drinking water (2.7% NaCl) to wild-type mice resulted in elevated placental TXA2 synthase (TXAS) and plasma thromboxane A2, but not prostacyclin, levels, which was also found in clinical preeclampsia placenta samples. 30300909_this study shows that genetic depletion of Thromboxane A2/Thromboxane-Prostanoid Receptor signaling prevents microvascular dysfunction in ischemia reperfusion injury 31785598_Thromboxane A2 Synthase and Thromboxane Receptor Deletion Reduces Ischaemia/Reperfusion-Evoked Inflammation, Apoptosis, Autophagy and Pyroptosis. |
ENSG00000059377 |
TBXAS1 |
65.219562 |
0.2572386479 |
-1.958821 |
0.46311242 |
17.050942 |
0.00003639031351679080506086524926523395606636768206954002380371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002414186652821243646207388655611225658503826707601547241210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.4332222677007 |
7.97858314730075 |
94.9827036849345 |
22.0609066995353 |
| ENSMUSG00000030030 |
71837 |
1700003E16Rik |
protein_coding |
Q9DAQ4 |
|
Alternative splicing;Phosphoprotein;Reference proteome |
|
|
mmu:71837; |
Human_homologues |
17971504_1700003E16Rik is most abundantly expressed in tissues rich in highly ciliated cells, such as olfactory sensory neurons, and is predicted to be important to cilia. |
ENSG00000284308 |
C2orf81 |
45.816244 |
2.7793664340 |
1.474756 |
0.42293792 |
12.055460 |
0.00051640912778819214601355724880704656243324279785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0025747770325596540733070760609280114294961094856262207031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
69.86530095521 |
20.7762212982302 |
25.1371320100696 |
5.79136735022341 |
| ENSMUSG00000030032 |
75659 |
Wdr54 |
protein_coding |
Q9R0D8 |
FUNCTION: When cross-linked to form dimers and trimers, it has a regulatory effect on ERK signaling pathway activity in response to EGF stimulation. Colocalizes with the EGF receptor in WDR54-specific vesicle where it sustains the internalization and controls the degradation of the EGF receptor after EGF stimulation. {ECO:0000250|UniProtKB:Q9H977}. |
Reference proteome;Repeat;Ubl conjugation;WD repeat |
|
|
mmu:75659; |
vesicle [GO:0031982]; protein homodimerization activity [GO:0042803]; negative regulation of receptor internalization [GO:0002091]; regulation of epidermal growth factor receptor signaling pathway [GO:0042058]; regulation of MAPK cascade [GO:0043408] |
Human_homologues 29987896_WDR54 was found to be elevated and associated with prognosis in patients with colorectal cancer (CRC). Functional assays showed that WDR54 facilitates the aggressiveness of CRC cells and promotes tumor growth in mice. Knockdown of WDR54 sensitizes CRC cells to the proliferation-inhibitory effect of SHP2 inhibitor, an effect involving their common function in regulating AKT and ERK signaling. |
ENSG00000005448 |
WDR54 |
390.498574 |
2.1645600572 |
1.114074 |
0.16881815 |
42.890886 |
0.00000000005787967992434735446190413622702083170507014742156570719089359045028686523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000009459477302657285105361745117073801747409333984251134097576141357421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
529.131713316673 |
62.0581806255787 |
244.45231333015 |
21.8012920048472 |
| ENSMUSG00000030096 |
21366 |
Slc6a6 |
protein_coding |
O35316 |
FUNCTION: Mediates sodium- and chloride-dependent transport of taurine (PubMed:1465453, PubMed:9375654, PubMed:30270321). Can also mediate transport of hypotaurine, beta-alanine and gamma-aminobutyric acid (GABA) (PubMed:1465453, PubMed:9375654, PubMed:30270321). {ECO:0000269|PubMed:1465453, ECO:0000269|PubMed:30270321, ECO:0000269|PubMed:9375654}. |
Cell membrane;Glycoprotein;Membrane;Neurotransmitter transport;Phosphoprotein;Reference proteome;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:21366; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; dendrite [GO:0030425]; GABA-ergic synapse [GO:0098982]; microvillus membrane [GO:0031528]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; plasma membrane protein complex [GO:0098797]; postsynaptic membrane [GO:0045211]; alanine transmembrane transporter activity [GO:0022858]; amino acid transmembrane transporter activity [GO:0015171]; amino acid:sodium symporter activity [GO:0005283]; gamma-aminobutyric acid transmembrane transporter activity [GO:0015185]; gamma-aminobutyric acid:sodium:chloride symporter activity [GO:0005332]; taurine binding [GO:0030977]; taurine transmembrane transporter activity [GO:0005368]; taurine:sodium symporter activity [GO:0005369]; alanine transport [GO:0032328]; amino acid import across plasma membrane [GO:0089718]; gamma-aminobutyric acid import [GO:0051939]; import across plasma membrane [GO:0098739]; modulation of chemical synaptic transmission [GO:0050804]; neurotransmitter transport [GO:0006836]; nitrogen compound transport [GO:0071705]; positive regulation of cell differentiation [GO:0045597]; positive regulation of necrotic cell death [GO:0010940]; sodium ion transmembrane transport [GO:0035725]; taurine transport [GO:0015734] |
11772953_The retinal involvement identifies the taurine transporter as an important factor for the development and maintenance of normal retinal functions and morphology. 14631140_decreased sensitivity of mature erythrocytes from taut-/- mice to osmotic shock and oxidative stress, rendering them more resistant to apoptosis 14734644_Knockout of the taurine transporter identifies taurine transport as a crucial factor for the maintenance of skeletal muscle function and total exercise capacity, while cardiac muscle apparently can compensate for the loss of taurine. 15606752_in NIH3T3 cells active taurine uptake reflects TauT expression TauT activity is modulated by multiple stimuli; the subcellular localization of TauT is regulated; and volume-sensitive taurine release is not mediated by TauT 15630240_Murine TAUT is regulated in response to hypertonicity and taurine-rich conditions. 15914036_TauT localizes to the primary cilium of growth-arrested NIH3T3 fibroblasts 16249932_Taut-/- mice suffer from renal taurine loss and impaired ability to lower urine osmolality and to increase urinary water excretion. 16421246_Apoptotic liver damage develops as a consequence of taurine deficiency due to taurine transporter knockout mutation. 16550472_TAUT is responsible for taurine uptake in mouse cerebrocortical neurons, and that the expression of TAUT is increased with developing cerebrocortical neurons 17153590_Results describe the gene expressions of taurine transporter and taurine biosynthetic enzyme during mouse and chicken embryonic development. 17153611_Data show that myogenic induction of the taurine transporter prevents dexamethasone-induced muscle atrophy. 17459327_Furthermore, taut-/- and taut+/- mice develop moderate unspecific hepatitis and liver fibrosis beyond 1 year of age where hepatocyte apoptosis and activation of the CD95 system are pronounced. 17785795_In a TAUT-deficient model, lack of taurine in the skin was found to cause a significantly higher sensitivity to ultraviolet B-induced immunosuppression. 17962336_Lack of taurine leads to reduced sensitivity of synaptic and extrasynaptic GABAA receptors and consequently to disinhibition in Taut knockout mice 18407290_Multiple actions of taurine, including osmoregulation, regulation of mitochondrial protein expression and inhibition of apoptosis, collectively ensure proper maintenance of cardiac and skeletal muscular structure and function. 19423693_critical role in protecting against cisplatin-induced nephrotoxicity, possibly by attenuating a p53-dependent pathway 20100858_All taut-/- mice succumb to infections during crisis, while about 90% of the control taut(+/+) mice survive. 20804603_data suggest TauT plays a critical role in renal function; expression of TauT is negatively regulated by p53 and positively regulated by c-Jun; outcome level of TauT may determine the fate of renal cells during stress-induced acute kidney injury 21160187_study represents the first characterization of TauT in neural precursor cells 22383044_Acute exposure to ROS reduces taurine uptake as a result of modulated TauT transport kinetics. 22531910_Effective T-cell recall responses require the taurine transporter Taut. 23381576_Studied protective effect of taurine associated with the upregulation of TAUT expression in acute myocardial ischemia. Data showed that taurine reversed the apoptosis induced by hypoxia and AMI, thereby, effectively protecting the myocardium. 24321512_This study demonistrated that intracellular taurine deficits lead in particular to a hyposensitivity of nociceptive sensory neurons sensitive to noxious chemical stimulation. 25629817_both homozygous and heterozygous TauT gene deletion predispose C57BL/6 mice to develop end-stage diabetic kidney disease 27444300_These findings suggest that TauT protein expression is relatively insensitive to changes in muscle taurine content in WT and mdx mice, and that taurine and beta-alanine supplementation may be viable therapeutic strategies to improve fatigue resistance of dystrophic skeletal muscle. 28572444_This study evaluated the role of taurine and its transporter on pancreatic islet cells and insulin secretion. 29491220_Circulating levels of GDF15 were elevated in old slc6a6 knockout mice 32705197_Significance of taurine transporter (TauT) in homeostasis and its layers of regulation (Review). 32968166_Evaluation of the neuroprotective effect of taurine in Alzheimer's disease using functional molecular imaging. 35882796_Characterization of Bone Tissue and Bone Morphology in Taurine Transporter Knockout Mice. |
ENSG00000131389 |
SLC6A6 |
5488.610521 |
0.4097523700 |
-1.287176 |
0.05776570 |
485.550303 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001324415733713911530714307791613883092169718619044792716956003947380006404203169170958651742451680113523325887529688260655136649171687101196416201463182821152 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000080694001823721196863895532417309645288998018080357802720219226524492539680709385154204134761011650229769584486370454241043662201213922602087992460018441530 |
Yes |
No |
3137.93501981954 |
125.676905415721 |
7658.12536925578 |
225.481700594712 |
| ENSMUSG00000030206 |
14840 |
Gsg1 |
protein_coding |
Q8R1W2 |
FUNCTION: May cause the redistribution of PAPOLB from the cytosol to the endoplasmic reticulum. {ECO:0000269|PubMed:18325338}. |
Alternative splicing;Endoplasmic reticulum;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:14840; |
endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; plasma membrane [GO:0005886]; RNA polymerase binding [GO:0070063] |
18325338_expression of GSG1 stimulates TPAP targeting to the ER, suggesting that interactions between the two proteins lead to the redistribution of TPAP from the cytosol to the ER |
ENSG00000111305 |
GSG1 |
34.687129 |
0.2491633176 |
-2.004836 |
0.45607475 |
19.108985 |
0.00001234622630702887395787777785605143776592740323394536972045898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000900334838672813627009905435016889896360225975513458251953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.937129041061 |
4.47965423129925 |
55.9357178906879 |
12.1936568502665 |
| ENSMUSG00000030218 |
17313 |
Mgp |
protein_coding |
P19788 |
FUNCTION: Associates with the organic matrix of bone and cartilage. Thought to act as an inhibitor of bone formation. |
Chondrogenesis;Developmental protein;Differentiation;Disulfide bond;Gamma-carboxyglutamic acid;Osteogenesis;Phosphoprotein;Reference proteome;Secreted;Signal |
|
This gene encodes a member of the osteocalcin/matrix Gla family of proteins. The encoded vitamin K-dependent protein is secreted by chondrocytes and vascular smooth muscle cells, and functions as a physiological inhibitor of ectopic tissue calcification. This protein also inhibits angiogenesis. Mice lacking a functional copy of this gene exhibit impaired differentiation of endothelial cells, reduced stature, and calcification and rupture of the vasculature leading to premature death. [provided by RefSeq, Sep 2016]. |
mmu:17313; |
collagen-containing extracellular matrix [GO:0062023]; endoplasmic reticulum [GO:0005783]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; protein-containing complex [GO:0032991]; calcium ion binding [GO:0005509]; calcium-dependent protein binding [GO:0048306]; branching morphogenesis of an epithelial tube [GO:0048754]; cartilage development [GO:0051216]; cell differentiation [GO:0030154]; lung development [GO:0030324]; ossification [GO:0001503]; protein-containing complex assembly [GO:0065003]; regulation of bone mineralization [GO:0030500] |
12391016_role in vascular calcification in vivo 16612082_MGP is a known inhibitor of mineralization, and mice deficient in Mgp show severe vascular calcification and premature bone mineralization. 17068135_Inorganic phosphate(Pi) regulates MGP expression in growth plate chondrocytes, thereby suggesting a key role for Pi and ERK1/2 in the regulation of bone formation. 17407158_PTH regulates MGP via both PKA- and ERK-dependent pathways. 17670744_Matrix GLA protein is an inhibitory morphogen in pulmonary vascular development 17942075_MGP in Abcc6-/- mice is largely in inactive form and is unable to prevent the unwanted mineralization of connective tissues in PXE. 18719210_Extensive hypomineralization was observed in dentin and alveolar bone from transgenic mice expressing matrix Gla protein when compared with that in wild-type tissues. Mineralization was virtually absent in tooth root dentin and cellular cementum. 19350115_Oral anticoagulant treatment decreased local expression of MGP. 19419315_Inorganic phosphate regulates MGP expression in osteoblasts through the ERK1/2-Fra-1 pathway. 19661459_Heat shock protein 70 enhances vascular bone morphogenetic protein-4 signaling by binding matrix Gla protein. 21765215_Mgp gene deletion may have a role in arteriovenous malformations 23110920_MGP is a multi-functional inhibitor of normal and abnormal angiogenesis that may function by coordinating with both Notch and BMP signaling pathways 23857752_Elastin haploinsufficiency impedes the progression of arterial calcification in MGP-deficient mice. 23994172_The arterial calcification, not MGP deficiency itself, causes the low bone mass phenotype in Mgp-/- mice. 24036114_Two sides of MGP null arterial disease: chondrogenic lesions dependent on transglutaminase 2 and elastin fragmentation associated with induction of adipsin. 24956112_expression of MGP and OCN increased gradually in the murine developing tibial epiphysis, and the two mineral-associated proteins may occur at the same location during a particular period, but at different levels. 25614167_Matrix Gla protein limits pulmonary arteriovenous malformations in ALK1 deficiency. 25711639_Matrix Gla's restricted expression to glaucoma-associated tissues from anterior and posterior segments suggests its involvement in the development of the disease. 26364300_loss of MGP causes dysregulation of early endothelial differentiation. 27593913_We propose MGP as a strong candidate for the regulation of stiffness in glaucoma. MGP further illustrates the presence of a common function affecting key glaucomatous parameters in the front and back of the eye, and thus offers the possibility for a sole therapeutic target for the disease. 28487368_the nasal septum to be abnormally mineralized and shortened in Mgp(-/-) mice. Transgenic restoration of Mgp expression in chondrocytes fully corrected the craniofacial anomalies caused by MGP deficiency, suggesting a local role for MGP in the developing nasal septum. 30988158_Observations establish, for the first time, that MGP plays an essential role in osteoclast differentiation and function. The molecular evidence indicates that MGP controls osteoclastogenesis via modulation of the intracellular calcium flux and Src/Rac1 signaling. 32000575_Matrix Gla protein regulates adipogenesis and is serum marker of visceral adiposity. 34809448_Matrix Gla Protein Levels Are Associated With Arterial Stiffness and Incident Heart Failure With Preserved Ejection Fraction. 35414780_MGP promotes CD8(+) T cell exhaustion by activating the NF-kappaB pathway leading to liver metastasis of colorectal cancer. 35418245_Prevention of Arterial Elastocalcinosis: Differential Roles of the Conserved Glutamic Acid and Serine Residues of Matrix Gla Protein. 35778813_A preliminary study exploring the mechanical properties of normal and Mgp-deficient mouse femurs during early growth. |
ENSG00000111341 |
MGP |
20955.791382 |
2.3819306810 |
1.252131 |
0.06974088 |
311.290231 |
0.00000000000000000000000000000000000000000000000000000000000000000000114329519878893728483643813701676103099524941478296874200911856545880085876179206269724535038450565001388321291415363275456912091626380907330680317459879178497738891055637670823852403145792777650 |
0.0000000000000000000000000000000000000000000000000000000000000000003285787258104357546375482492852995146090739413409816682215720200429791540788163959990198852341736252829935538169254037898719781031618253305112232899417500710806194037447158784459588787285611033 |
Yes |
No |
29735.8511380974 |
1438.01170274787 |
12483.9280067064 |
453.023196668503 |
| ENSMUSG00000030222 |
232441 |
Rerg |
protein_coding |
Q8R367 |
FUNCTION: Binds GDP/GTP and possesses intrinsic GTPase activity. Has higher affinity for GDP than for GTP (By similarity). {ECO:0000250}. |
Cytoplasm;GTP-binding;Hydrolase;Nucleotide-binding;Proto-oncogene;Reference proteome |
|
|
mmu:232441; |
cytosol [GO:0005829]; fibrillar center [GO:0001650]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; G protein activity [GO:0003925]; GDP binding [GO:0019003]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; Ras protein signal transduction [GO:0007265]; response to hormone [GO:0009725] |
Human_homologues 18676680_Observational study of gene-disease association. (HuGE Navigator) 19170196_Observational study of gene-disease association and gene-environment interaction. (HuGE Navigator) 19625176_Observational study of gene-disease association. (HuGE Navigator) 19692168_Observational study of gene-disease association. (HuGE Navigator) 20697807_Loss of RERG expression is associated with breast cancer and distant metastasis. 21270509_Results describe the mRNA expression of CRABP1, RERG, and GRP in pituitary adenomas. 24992181_Estradiol estrogen receptor-beta and PGE2 integrate at RERG, leading to increased endometriotic cell proliferation. 27705918_this study shows that miR-382-5p expression is negatively correlated with RERG expression, and functions as an independent oncomiR for the higher incidence and poorer prognosis of breast cancer 28716111_In particular, methylation levels of ITGA4, RERG, and ZNF671 could distinguish Nasopharyngeal carcinoma (NPC) patients from non-cancer nasopharyngeal epithelium (NNE) subjects. We identified the DNA methylation rates of previously unidentified NPC candidate genes. 30431143_Study revealed that ERK5 can inhibit Raslike oestrogenregulated growth inhibitor (RERG) protein expression and that the inhibition of RERG expression promotes prostatic carcinoma cell proliferation and migration. 32986146_NF-kappaB-mediated miR-650 plays oncogenic roles and activates AKT/ERK/NF-kappaB pathways by targeting RERG in glioma cells. |
ENSG00000134533 |
RERG |
1257.079183 |
0.2998996048 |
-1.737448 |
0.12445125 |
187.740180 |
0.00000000000000000000000000000000000000000098995640900490103329540025395540403601001360547680516968726281914180340629273365292815689619586722174103576073894705800171323062386363744735717773437500000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000001267144203526273322618112325062917166092817415010310617199696408501508360054699075748040827130710043828525773745852234242192935198545455932617187500000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
570.198632852911 |
49.2480872650202 |
1901.29838042225 |
119.747838295708 |
| ENSMUSG00000030223 |
19277 |
Ptpro |
protein_coding |
E9Q612 |
FUNCTION: Possesses tyrosine phosphatase activity. Plays a role in regulating the glomerular pressure/filtration rate relationship through an effect on podocyte structure and function. {ECO:0000269|PubMed:11086029}. |
Glycoprotein;Hydrolase;Membrane;Phosphoprotein;Protein phosphatase;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:19277; |
apical plasma membrane [GO:0016324]; dendritic spine [GO:0043197]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; lateral plasma membrane [GO:0016328]; plasma membrane [GO:0005886]; postsynaptic density membrane [GO:0098839]; cadherin binding [GO:0045296]; protein homodimerization activity [GO:0042803]; protein tyrosine phosphatase activity [GO:0004725]; Wnt-protein binding [GO:0017147]; axon guidance [GO:0007411]; cell morphogenesis [GO:0000902]; glomerulus development [GO:0032835]; lamellipodium assembly [GO:0030032]; monocyte chemotaxis [GO:0002548]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of glomerular filtration [GO:0003105]; peptidyl-tyrosine dephosphorylation [GO:0035335]; peptidyl-tyrosine dephosphorylation involved in inactivation of protein kinase activity [GO:1990264]; podocyte differentiation [GO:0072112]; regulation of glomerular filtration [GO:0003093]; regulation of synapse organization [GO:0050807]; slit diaphragm assembly [GO:0036060] |
12532410_PTPRO is involved in the differentiation and axonogenesis of central and peripheral nervous system neurons, where it is in a position to modulate intracellular responses to neurotrophin-3 and/or nerve growth factor. 15673668_We find that at least one NPCD isoform is tyrosine phosphorylated in vivo and can serve as a substrate for PTPRO in vitro. 16680165_Ptpro moderates the amount of maximal activation of Eph receptors. In the chick retinotectal projection system, Ptpro controls the sensitivity of retinal axons to ephrins. 19244239_PTP-oc is a positive regulator of osteoclasts 19800005_Data indicate that PTPRO is required for peptidergic differentiation and process outgrowth of sensory neurons, as well as mature sensory function, and provide the first evidence that RPTPs regulate DRG development. 19924828_PTPRO might participate in regulation of dendritic morphology or synapse formation of interneurons in the adult mouse olfactory bulb 20804755_These results suggest that ptpro is a target gene of Wnt/beta-catenin signaling and that PTPRO may function as a novel receptor for Wnt. 23516305_PTPRO is expressed mainly in TrkB-expressing (TrkB(+)) and Ret(+) mechanoreceptors within the trigeminal ganglia during embryogenesis. 24128416_PTPRO deficiency resulted in reduction of NF-kappaB activation in both hepatocytes and macrophages and was correlated to c-Src phosphorylation. 25339662_Survival and inflammation promotion effect of PTPRO in fulminant hepatitis is associated with NF-kappaB activation. 25646811_The loss of PTPRO in the tumor niche was correlated with larger tumor volume, more metastases, increased number of circulating tumor cells, less apoptosis and reduced necrosis rates in the orthotopic mouse model of breast cancer. 25676701_Study show that EphA4 is a substrate for PTP-oc in osteoclasts and that the molecular mechanism contributing to the PTP-oc-induced up-regulation of the osteoclast activity in part involves its dephosphorylation and inactivation of the EphA4 signaling. 25826083_PTPRO regulates insulin and lipid metabolism via the PI3K/Akt/MDM4/MDM2/P53 axis by affecting autophagy. 26117839_PTPRO truncated serves as an important tumor suppressor in hepatocellular carcinoma microenvironment. 26303994_PTPRO plays an important role in FH by interacting with TLR4. 28166196_PTPROt thus functions as an obligate haploinsufficient TS in CLL, where its expression levels determine its role as a promoter or inhibitor of the tumorigenic process in mice. Partial loss of PTPROt generates the strongest disease phenotype, suggesting that its intermediate expression levels in CLL are selected for. 28578349_TLR4 and NF-kappaB/p65 phosphorylation was significantly enhanced in PTPRO over-expressing cells, while significantly down-regulated in PTPRO knockout cells. PTPRO plays ital roles in atherosclerosis (AS) via promoting ox-LDL induced oxidative stress and cell apoptosis through TLR4/NF-kappaB pathway. 28871037_Using loss-of-function and gain-of-function approaches, we show that PTPRO promotes the formation of excitatory synapses 30622194_Data suggest that tyrosine (Tyr399) in PTPRQ Phosphatase (PTPROt) might participate in regulating the activity of this phosphatase toward src Kinases (Src). 35904817_PTPRO represses colorectal cancer tumorigenesis and progression by reprogramming fatty acid metabolism. |
ENSG00000151490 |
PTPRO |
136.132113 |
559.8616222238 |
9.128926 |
2.06733272 |
10.721397 |
0.00105903672834807300456860801318725862074643373489379882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0048588094717463402999468868870280857663601636886596679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
263.600723833359 |
520.942348334298 |
0.470831922335269 |
0.676979858193769 |
| ENSMUSG00000030246 |
16832 |
Ldhb |
protein_coding |
P16125 |
FUNCTION: Interconverts simultaneously and stereospecifically pyruvate and lactate with concomitant interconversion of NADH and NAD(+). {ECO:0000250|UniProtKB:P07195}. |
Acetylation;Cytoplasm;Direct protein sequencing;Membrane;Mitochondrion;Mitochondrion inner membrane;NAD;Oxidoreductase;Phosphoprotein;Reference proteome |
PATHWAY: Fermentation; pyruvate fermentation to lactate; (S)-lactate from pyruvate: step 1/1. {ECO:0000250|UniProtKB:P07195}. |
This gene encodes the B subunit of lactate dehydrogenase enzyme, which catalyzes the interconversion of pyruvate and lactate with concomitant interconversion of NADH and NAD+ in a post-glycolysis process. Alternatively spliced transcript variants have also been found for this gene. Recent studies have shown that a C-terminally extended isoform is produced by use of an alternative in-frame translation termination codon via a stop codon readthrough mechanism, and that this isoform is localized in the peroxisomes. Pseudogenes have been identified on chromosomes 1 and 19. [provided by RefSeq, Feb 2016]. |
mmu:16832; |
cytosol [GO:0005829]; membrane raft [GO:0045121]; mitochondrial inner membrane [GO:0005743]; mitochondrion [GO:0005739]; myelin sheath [GO:0043209]; oxidoreductase complex [GO:1990204]; identical protein binding [GO:0042802]; kinase binding [GO:0019900]; L-lactate dehydrogenase activity [GO:0004459]; lactate dehydrogenase activity [GO:0004457]; NAD binding [GO:0051287]; lactate biosynthetic process from pyruvate [GO:0019244]; lactate metabolic process [GO:0006089]; NAD metabolic process [GO:0019674]; pyruvate metabolic process [GO:0006090] |
17853776_beta-agonists increased serum LDH levels, but it was not due to release from the heart. 18790003_Studies suggest that the expression of the ldh2 gene in spermatogonia and early spermatocytes are regulated by SP-mediated transcriptional mechanisms. 21041631_The increased LDH-A/LDH-B ratio causes high brain lactate levels, which, in turn, are predictive of aging phenotypes. 21199794_findings offer proof of concept for targeting LDHB as a therapeutic strategy in cancers driven by aberrant activation of the RTK-PI3K-AKT-mTOR signaling cascade 22967998_the Ldhb gene, which encodes the H isoform of lactate dehydrogenase, as a robust PPARgamma target in trophoblasts. 23650363_PGC-1alpha in skeletal muscle drives the expression of lactate dehydrogenase (LDH) B in an estrogen-related receptor-alpha-dependent manner. 25576887_Data show that PI3K/Akt signalling pathway and lactate dehydrogenase (LDH) play a crucial role in increase of cardiac ATP-binding cassette transporter sub-family C member 9 (SUR2A) induced by in vivo exposure to 18% oxygen. 25620379_drs regulates glucose metabolism via LDH-B. Downregulating drs may contribute to the Warburg effect, which is closely associated with malignant progression of cancer cells. 25907793_Exercise-induced changes in tumour LDH-B and MCT1 expression are modulated by oestrogen-related receptor alpha in breast cancer-bearing BALB/c mice 27077737_Findings suggest that RANKL protein-induced lactate dehydrogenase (LDH) activation stimulates glycolytic and mitochondrial respiratory metabolism via transcription factor NFATc1 signaling. 27738103_these results identified a previously unrecognized Ldhb-driven alteration in muscle mitochondrial function and suggested a mechanism for the adaptive metabolic response induced by exercise training. 33719570_Expression of lactate dehydrogenase A and B isoforms in the mouse kidney. 34158867_Lactate dehydrogenase B regulates macrophage metabolism in the tumor microenvironment. |
ENSG00000111716 |
LDHB |
404.378703 |
4.7277758012 |
2.241162 |
0.15809600 |
195.511645 |
0.00000000000000000000000000000000000000000001992259263944453993075043082641240699332733591756433950937230473592406198586395576126043888072335721055492384031545410882912960914836730808019638061523437500000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000027094725989644574504957074815477408139535226568851477431634403839934072560010711508584559566916035924240930762851853330630547134205698966979980468750000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
666.136913681128 |
73.1440013142219 |
140.898583539895 |
12.3376330566505 |
| ENSMUSG00000030254 |
58186 |
Rad18 |
protein_coding |
Q9QXK2 |
FUNCTION: E3 ubiquitin-protein ligase involved in postreplication repair of UV-damaged DNA. Postreplication repair functions in gap-filling of a daughter strand on replication of damaged DNA. Associates to the E2 ubiquitin conjugating enzyme UBE2B to form the UBE2B-RAD18 ubiquitin ligase complex involved in mono-ubiquitination of DNA-associated PCNA on 'Lys-164'. Has ssDNA binding activity. |
Acetylation;Cytoplasm;Cytoskeleton;DNA damage;DNA repair;DNA-binding;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Transferase;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:58186; |
centrosome [GO:0005813]; chromatin [GO:0000785]; cytoplasm [GO:0005737]; male germ cell nucleus [GO:0001673]; nuclear body [GO:0016604]; nuclear inclusion body [GO:0042405]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; Rad6-Rad18 complex [GO:0097505]; replication fork [GO:0005657]; site of double-strand break [GO:0035861]; XY body [GO:0001741]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; molecular function inhibitor activity [GO:0140678]; polyubiquitin modification-dependent protein binding [GO:0031593]; protein-containing complex binding [GO:0044877]; single-stranded DNA binding [GO:0003697]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin protein ligase binding [GO:0031625]; Y-form DNA binding [GO:0000403]; cellular response to DNA damage stimulus [GO:0006974]; negative regulation of cell death [GO:0060548]; negative regulation of DNA recombination [GO:0045910]; positive regulation of chromosome segregation [GO:0051984]; postreplication repair [GO:0006301]; protein autoubiquitination [GO:0051865]; protein monoubiquitination [GO:0006513]; response to UV [GO:0009411]; spermatogenesis [GO:0007283] |
12509447_RAD18 contributes to maintenance of genomic stability through postreplication repair 15383616_Rad18Sc localizes to the XY body and to other chromosomal regions that are unpaired and transcriptionally silenced during male meiotic prophase 16710452_Rad18 suppresses viral infection in a non-specific fashion, probably by targeting incoming DNA. 19068231_RAD18 is intrinsically required for the long-term maintenance of spermatogenesis. 21807948_repairs meiotic double-strand DNA breaks and and maintains meiotic sex chromosome inactivation 25675240_RAD18 can function as a mediator for DNA damage response signals to activate the G2/M checkpoint in order to maintain genome integrity and cell survival after IR exposure. 26795561_REV1 promote PCNA monoubiquitylation after UV radiation through interacting with ubiquitylated RAD18. 26883629_Rad18 confers DNA damage tolerance and tumor-suppression in a physiological setting. 28082021_Somatic hypermutation of immunoglobulin genes (SHM) in Rad18 KO mice was similar to wild-type mice, suggesting that Rad18 is dispensable for SHM. 29859927_these results demonstrate that the tumor-suppressive role of DNA polymerase Eta in UV-treated skin is Rad18 independent 35459258_Genome-wide CRISPR screen identified Rad18 as a determinant of doxorubicin sensitivity in osteosarcoma. |
ENSG00000070950 |
RAD18 |
632.882507 |
2.1180535093 |
1.082739 |
0.12388585 |
75.561162 |
0.00000000000000000354261701675518540664630130996294242515346613897901824341563070674965274520218372344970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000001207184393718456186448755062382501871901776586580232186740602173813385888934135437011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
868.024800060143 |
74.6299093125274 |
409.821940880252 |
27.0900724979028 |
| ENSMUSG00000030268 |
12035 |
Bcat1 |
protein_coding |
P24288 |
FUNCTION: Catalyzes the first reaction in the catabolism of the essential branched chain amino acids leucine, isoleucine, and valine. |
Acetylation;Amino-acid biosynthesis;Aminotransferase;Branched-chain amino acid biosynthesis;Cytoplasm;Lipid metabolism;Pyridoxal phosphate;Reference proteome;Transferase |
|
|
mmu:12035; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; mitochondrion [GO:0005739]; branched-chain-amino-acid transaminase activity [GO:0004084]; identical protein binding [GO:0042802]; L-isoleucine transaminase activity [GO:0052656]; L-leucine transaminase activity [GO:0052654]; L-leucine:2-oxoglutarate aminotransferase activity [GO:0050048]; L-valine transaminase activity [GO:0052655]; branched-chain amino acid catabolic process [GO:0009083]; leucine biosynthetic process [GO:0009098]; lipid metabolic process [GO:0006629]; valine biosynthetic process [GO:0009099] |
14563018_Bcat1 is a candidate for the type I diabetes susceptibility locus Idd6 15064703_Bcat1 is part of the complex multigenic Pas1 locus, with a functional role for its intragenic polymorphisms in lung tumor susceptibility. 16828066_These results demonstrate that the expression of the BCATc gene in the brain is specifically regulated by BDNF in a time- and region-dependent fashion. 17150414_BCATc mRNA gradually appears in different brain regions starting from early stages of neural development, and is maintained until adulthood. 17767905_BCATm(-/-) mice had elevated plasma branched-chain amino acids & decreased adiposity & body weight, despite eating more food, along with increased energy expenditure, improvements in glucose & insulin tolerance & protection from diet-induced obesity 20736162_analysis of the biochemical mechanism of BCATm (branched-chain aminotransferase) catalysis of reversible transamination of leucine and alpha-ketoglutarate to KIC and glutamate 24349566_leucine supplementation increased the expression of enzymes (BCAT1, BCAT2 and BCKDK) that metabolize branched-chain amino acids. 24847056_BCATc as a novel regulator of T cell activation and metabolism 26351290_results suggest transcriptional adaptations occur in BCATm KO mice that along with altered nutrient signaling may contribute to their previously reported protein turnover, metabolic and exercise phenotypes 28699638_regulatory role in macrophage function 30318512_Global deletion of BCATm caused a dramatic build-up of branched-chain amino acids, which could not be utilised for energy or amino acid synthesis, ultimately delaying the growth of lymphoma tumours. 32938905_BCAT1 binds the RNA-binding protein ZNF423 to activate autophagy via the IRE1-XBP-1-RIDD axis in hypoxic PASMCs. 33271468_Branched-chain amino acid aminotransferase-1 regulates self-renewal and pluripotency of mouse embryonic stem cells through Ras signaling. 35562710_Impaired expression of BCAT1 relates to muscle atrophy of mouse model of sarcopenia. 35760874_BCAT1 promotes osteoclast maturation by regulating branched-chain amino acid metabolism. 36182869_The proatherosclerotic function of BCAT1 in atherosclerosis development of aged-apolipoprotein E-deficient mice. 36260995_BCAT1 redox function maintains mitotic fidelity. |
ENSG00000060982 |
BCAT1 |
552.248032 |
2.3228999809 |
1.215927 |
0.14720823 |
67.522770 |
0.00000000000000020826791495144059287288344948377750407935804326242656969725430826656520366668701171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000060310587082516026739736144740747920680391845962842189976527151884511113166809082031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
792.417658521439 |
80.9962781042473 |
341.132922224369 |
26.7864255644876 |
| ENSMUSG00000030306 |
387314 |
Tmtc1 |
protein_coding |
Q3UV71 |
FUNCTION: Transfers mannosyl residues to the hydroxyl group of serine or threonine residues. The 4 members of the TMTC family are O-mannosyl-transferases dedicated primarily to the cadherin superfamily, each member seems to have a distinct role in decorating the cadherin domains with O-linked mannose glycans at specific regions. Also acts as O-mannosyl-transferase on other proteins such as PDIA3. {ECO:0000250|UniProtKB:Q8IUR5}. |
Alternative splicing;Endoplasmic reticulum;Membrane;Reference proteome;Repeat;TPR repeat;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000250|UniProtKB:Q8IUR5}. |
|
mmu:387314; |
endoplasmic reticulum [GO:0005783]; membrane [GO:0016020]; mitochondrion [GO:0005739]; dolichyl-phosphate-mannose-protein mannosyltransferase activity [GO:0004169]; mannosyltransferase activity [GO:0000030]; protein O-linked mannosylation [GO:0035269]; RNA processing [GO:0006396] |
Human_homologues 19913121_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20877624_Observational study of gene-disease association. (HuGE Navigator) 24764305_findings provide an important link between aneuploidy and the stress pathways activated by Aurora B inhibition and also support the use of Aurora B inhibitors in combination therapy for treatment of cancer. 28973932_O-mannosylation pathway dedicated to cadherins/ protocadherins orchestrated by the four TMTC1-4 genes 33436046_Conserved sequence motifs in human TMTC1, TMTC2, TMTC3, and TMTC4, new O-mannosyltransferases from the GT-C/PMT clan, are rationalized as ligand binding sites. |
ENSG00000133687 |
TMTC1 |
65.504723 |
0.1863443126 |
-2.423957 |
0.35412950 |
46.248188 |
0.00000000001041828750252030280163888101675081468595884803818307773326523602008819580078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001845248316725456367693866163213578350521526516558878938667476177215576171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.1533337917127 |
4.99674246875646 |
108.151053890952 |
17.8334508528479 |
| ENSMUSG00000030313 |
320560 |
Dennd5b |
protein_coding |
A2RSQ0 |
FUNCTION: Guanine nucleotide exchange factor (GEF) which may activate RAB39A and/or RAB39B. Promotes the exchange of GDP to GTP, converting inactive GDP-bound Rab proteins into their active GTP-bound form (By similarity). {ECO:0000250}. |
Acetylation;Guanine-nucleotide releasing factor;Membrane;Phosphoprotein;Reference proteome;Repeat;Transmembrane;Transmembrane helix |
|
|
mmu:320560; |
membrane [GO:0016020]; guanyl-nucleotide exchange factor activity [GO:0005085]; small GTPase binding [GO:0031267]; positive regulation of triglyceride transport [GO:1905885] |
30837651_DENND5B Regulates Intestinal Triglyceride Absorption and Body Mass. 36243100_Dennd5b-Deficient Mice are Resistant to PCSK9-Induced Hypercholesterolemia and Diet-Induced Hepatic Steatosis. |
ENSG00000170456 |
DENND5B |
78.594473 |
0.1489044346 |
-2.747541 |
0.33365565 |
68.042945 |
0.00000000000000015997320437439068289805016564908916515606627042379861425303033684031106531620025634765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000046591048738637076203235443024626614911724116138702633804768993286415934562683105468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.4004703322273 |
4.52689939098879 |
130.288062875255 |
19.720453477409 |
| ENSMUSG00000030316 |
68971 |
Tamm41 |
protein_coding |
G5E881 |
FUNCTION: Catalyzes the conversion of phosphatidic acid (PA) to CDP-diacylglycerol (CDP-DAG), an essential intermediate in the synthesis of phosphatidylglycerol, cardiolipin and phosphatidylinositol. {ECO:0000256|ARBA:ARBA00003203, ECO:0000256|PIRNR:PIRNR028840}. |
Lipid biosynthesis;Lipid metabolism;Magnesium;Membrane;Mitochondrion;Mitochondrion inner membrane;Nucleotidyltransferase;Phospholipid biosynthesis;Phospholipid metabolism;Proteomics identification;Reference proteome;Transferase |
PATHWAY: Lipid metabolism. {ECO:0000256|ARBA:ARBA00005189}.; PATHWAY: Phospholipid metabolism; CDP-diacylglycerol biosynthesis; CDP-diacylglycerol from sn-glycerol 3-phosphate: step 3/3. {ECO:0000256|ARBA:ARBA00005119, ECO:0000256|PIRNR:PIRNR028840}. |
|
mmu:68971; |
extrinsic component of membrane [GO:0019898]; mitochondrial inner membrane [GO:0005743]; phosphatidate cytidylyltransferase activity [GO:0004605]; cardiolipin biosynthetic process [GO:0032049]; CDP-diacylglycerol biosynthetic process [GO:0016024] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20877624_Observational study of gene-disease association. (HuGE Navigator) 34152079_Association of mitochondrial variants and haplogroups identified by whole exome sequencing with Alzheimer's disease. |
ENSG00000144559 |
TAMM41 |
260.670811 |
2.0386517682 |
1.027615 |
0.16383341 |
39.004575 |
0.00000000042281357629650662051606936247584184879189095340734638739377260208129882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000062105076124863926259077315564748056342381232752813957631587982177734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
355.805004327253 |
40.4923542687674 |
174.529564041295 |
15.2554240807977 |
| ENSMUSG00000030322 |
17193 |
Mbd4 |
protein_coding |
A0A0R4J0A8 |
FUNCTION: Mismatch-specific DNA N-glycosylase involved in DNA repair. Has thymine glycosylase activity and is specific for G:T mismatches within methylated and unmethylated CpG sites. Can also remove uracil or 5-fluorouracil in G:U mismatches. Has no lyase activity. Was first identified as methyl-CpG-binding protein. {ECO:0000256|PIRNR:PIRNR038005}. |
DNA damage;DNA repair;DNA-binding;Hydrolase;Nucleus;Phosphoprotein;Proteomics identification;Reference proteome |
|
|
mmu:17193; |
nuclear speck [GO:0016607]; DNA binding [GO:0003677]; pyrimidine-specific mismatch base pair DNA N-glycosylase activity [GO:0008263]; DNA repair [GO:0006281] |
10499592_G/T mismatch glycosylse. Acts to maintain levels of 5-methylcytosine in the genome by initiating repair of the results of deamination of 5-methylcytosine 12130785_suppresses CpG mutability and tumorigenesis in vivo 12417741_studies indicate that, although inactivation of Mbd4 does not by itself cause cancer predisposition in mice, it can alter the mutation spectrum in cancer cells and modify the cancer predisposition phenotype 12456671_structure and activity of the mismatch-specific thymine glycosylase domain of MBD4 12574322_Mbd4 glycosylase does not significantly contribute to mechanisms of antibody diversification, as analyzed by an in vitro switch assay and by in vivo immunizations in Mbd4-/- knockout mice. 14562041_results establish a novel functional role for MBD4 in the cellular response to DNA damage and may have implications for its role in suppressing neoplasia 14614141_MED1 has a role in cell cycle arrest and apoptosis induced by DNA damage 15184874_in the context of MMR deficiency, additional loss of Mbd4 does not alter spontaneous mutation frequency at the endogenous Dlb-1b locus, nor does it modify tumour onset, tumour spectrum or MSI 16723356_Med1 depleted cells exhibited an exacerbated response to retinoids, both in terms transcriptional responses and of cellular differentiation. 17132730_Acts as a pivotal coactivator for GATA-1 in erythroid development. 18452947_Data report the characterization of interactions between Dnmt3b and both thymine-DNA glycosylase (Tdg) and methyl-CpG binding domain protein 4. 19000668_Studies on a complex A mutant mouse, defective for the Ift122 gene, is reported. 23316048_the crystal structure of MBD4 bound to 5-hydroxymethylcytosine further demonstrates that MBDMBD4 is able to recognize a wide range of 5-methylcytosine modifications 24205214_A variant Mbd4 transcript spanning exons 1,6-8 is expressed in Mbd4 deficient B cells. 27086921_Mbd4-/- mice displayed more severe symptoms, decreased survival, and a greater tumor burden than wild-type (WT) controls. 27777312_Mbd4 is a component of mismatch repair-directed DNA end processing. 34592200_Expression of mBD4, mBD3 and CRAMP during type II collagen-induced arthritis/CIA and their association with inflammation and bone-remodeling markers. |
ENSG00000129071 |
MBD4 |
657.735359 |
2.0791396403 |
1.055987 |
0.10980611 |
91.747700 |
0.00000000000000000000098462645883882193857671619787993180910583765788436237916870542863367887775893905200064182281494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000429737255616989564599189902102229041865321078176561460804788739054060897615272551774978637695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
885.603825262829 |
67.4699957799419 |
425.947256313358 |
25.0679863759622 |
| ENSMUSG00000030346 |
19362 |
Rad51ap1 |
protein_coding |
A0A0R4J1G4 |
Human_homologues FUNCTION: Structure-specific DNA-binding protein involved in DNA repair by promoting RAD51-mediated homologous recombination (PubMed:17996710, PubMed:17996711, PubMed:20871616, PubMed:25288561, PubMed:26323318). Acts by stimulating D-Loop formation by RAD51: specifically enhances joint molecule formation through its structure-specific DNA interaction and its interaction with RAD51 (PubMed:17996710, PubMed:17996711). Binds single-stranded DNA (ssDNA), double-stranded DNA (dsDNA) and secondary DNA structures, such as D-loop structures: has a strong preference for branched-DNA structures that are obligatory intermediates during joint molecule formation (PubMed:9396801, PubMed:17996711, PubMed:22375013, PubMed:17996710). Cooperates with WDR48/UAF1 to stimulate RAD51-mediated homologous recombination: both WDR48/UAF1 and RAD51AP1 have coordinated role in DNA-binding during homologous recombination and DNA repair (PubMed:27463890, PubMed:27239033, PubMed:32350107). WDR48/UAF1 and RAD51AP1 also have a coordinated role in DNA-binding to promote USP1-mediated deubiquitination of FANCD2 (PubMed:31253762). Also involved in meiosis by promoting DMC1-mediated homologous meiotic recombination (PubMed:21307306). Key mediator of alternative lengthening of telomeres (ALT) pathway, a homology-directed repair mechanism of telomere elongation that controls proliferation in aggressive cancers, by stimulating homologous recombination (PubMed:31400850). May also bind RNA; additional evidences are however required to confirm RNA-binding in vivo (PubMed:9396801). {ECO:0000269|PubMed:17996710, ECO:0000269|PubMed:17996711, ECO:0000269|PubMed:20871616, ECO:0000269|PubMed:21307306, ECO:0000269|PubMed:22375013, ECO:0000269|PubMed:25288561, ECO:0000269|PubMed:26323318, ECO:0000269|PubMed:27239033, ECO:0000269|PubMed:27463890, ECO:0000269|PubMed:31253762, ECO:0000269|PubMed:31400850, ECO:0000269|PubMed:32350107, ECO:0000269|PubMed:9396801}. |
Proteomics identification;Reference proteome |
|
|
mmu:19362; |
chromatin [GO:0000785]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; D-loop DNA binding [GO:0062037]; double-stranded DNA binding [GO:0003690]; RNA binding [GO:0003723]; single-stranded DNA binding [GO:0003697]; cellular response to ionizing radiation [GO:0071479]; double-strand break repair via homologous recombination [GO:0000724]; interstrand cross-link repair [GO:0036297]; positive regulation of double-strand break repair via homologous recombination [GO:1905168]; positive regulation of reciprocal meiotic recombination [GO:0010845] |
21307306_RAD51AP1 foci colocalize with a subset of DMC1 foci in spermatocytes. 23271971_VipD of Legionella pneumophila targets activated Rab5 and Rab22 to interfere with endosomal trafficking in macrophages. 32665355_RAD51AP1 Deficiency Reduces Tumor Growth by Targeting Stem Cell Self-Renewal. 34099522_RAD51AP1 Loss Attenuates Colorectal Cancer Stem Cell Renewal and Sensitizes to Chemotherapy. |
ENSG00000111247 |
RAD51AP1 |
1346.316531 |
3.0723369932 |
1.619336 |
0.12880546 |
152.465864 |
0.00000000000000000000000000000000005011973919511241508156429100034115287230455966086526709998800375151695989602741800229704707825284515365638071671128273010253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000046550235818289775288604906702464723505772626819734882938250127367427800809473268715105685744415353610747843049466609954833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2033.93873129429 |
181.833716915954 |
662.016808635125 |
45.0133958499441 |
| ENSMUSG00000030443 |
213011 |
Zfp583 |
protein_coding |
Q3V080 |
FUNCTION: May be involved in transcriptional regulation. |
DNA-binding;Metal-binding;Nucleus;Reference proteome;Repeat;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:213011; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
ENSG00000198440 |
ZNF583 |
21.851996 |
3.5160764120 |
1.813966 |
0.58194809 |
9.648275 |
0.00189529462497756455083286475371551205171272158622741699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0081344400472409868901868534862842352595180273056030273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.7461818206444 |
13.984553627985 |
9.59768158212246 |
3.12902347965051 |
| ENSMUSG00000030494 |
52428 |
Rhpn2 |
protein_coding |
Q8BWR8 |
FUNCTION: Binds specifically to GTP-Rho. May function in a Rho pathway to limit stress fiber formation and/or increase the turnover of F-actin structures in the absence of high levels of RhoA activity (By similarity). {ECO:0000250}. |
3D-structure;Coiled coil;Cytoplasm;Phosphoprotein;Reference proteome |
|
|
mmu:52428; |
perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; glomerular filtration [GO:0003094]; negative regulation of stress fiber assembly [GO:0051497]; signal transduction [GO:0007165] |
15767687_Mice lacking rhophilin 2 develop normally, have normal life spans, and are fertile; rhophilin 2 does not play an essential role in thyroid physiology 36210710_[RHPN2 is highly expressed in osteosarcoma cells to promote cell proliferation and migration and inhibit apoptosis]. |
ENSG00000131941 |
RHPN2 |
156.925788 |
0.4656519600 |
-1.102676 |
0.23517999 |
21.856321 |
0.00000293846962681219156456220720219363329306361265480518341064453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000240380071727192794596526109751621902432816568762063980102539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.141166915012 |
16.3968431564717 |
215.055826055378 |
25.4907585016657 |
| ENSMUSG00000030513 |
18553 |
Pcsk6 |
protein_coding |
H3BLN2 |
Human_homologues FUNCTION: Serine endoprotease that processes various proproteins by cleavage at paired basic amino acids, recognizing the RXXX[KR]R consensus motif. Likely functions in the constitutive secretory pathway, with unique restricted distribution in both neuroendocrine and non-neuroendocrine tissues. |
Hydrolase;Protease;Proteomics identification;Reference proteome;Serine protease |
|
|
|
cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; membrane [GO:0016020]; endopeptidase activity [GO:0004175]; heparin binding [GO:0008201]; nerve growth factor binding [GO:0048406]; serine-type endopeptidase activity [GO:0004252]; determination of left/right symmetry [GO:0007368]; glycoprotein metabolic process [GO:0009100]; nerve growth factor production [GO:0032902]; peptide hormone processing [GO:0016486]; protein processing [GO:0016485]; secretion by cell [GO:0032940]; zygotic determination of anterior/posterior axis, embryo [GO:0007354] |
11960907_Malignant conversion of non-tumorigenic murine skin keratinocytes overexpressing PACE4 12447384_Spc4 regulates Nodal signalling during gastrulation 12533406_plays an important role in the processes of stromal cell decidualization and embryo implantation 16135528_the CRD of PC5A and PACE4 functions as a cell surface anchor favoring the processing of their cognate surface-anchored substrates, including endothelial lipase. 17914070_The effects of FSH and oocytes on the expression of Pcsk6 mRNA during the the preantral to antral follicle transition are reported. 19520771_PACE4 mRNA levels increased markedly during differentiation of skeletal myoblast cell line; results suggest PACE4 plays an important role in myogenic differentiation through association with the IGF-II pathway 20651981_PACE4, together with other proprotein convertases, are suitable targets to slow down or block tumor progression. 21183657_PCSK6 activity plays a role in maintaining normal cellular and tissue homeostasis in the ovary. 21896659_Furin and Pace4 are released by the extraembryonic microenvironment, and that they cleave a membrane-bound reporter substrate in adjacent epiblast cells and activate Nodal to maintain pluripotency 22440827_A variant in PCSK6 is strongly associated with protection against pain in knee osteoarthritis. 23441131_we observed an increased susceptibility to UV in single furin transgenic mice that was not substantially enhanced in the double furin/PACE4 transgenic mice. 24557631_PACE4 participates in bone formation at least in osteoblast differentiation, and estrogen receptor-mediated stimuli induce osteoblast differentiation through the upregulation of PACE4 expression 24845697_Mice overexpressing PACE4 exhibited tumors of increased growth rate. 26416966_PC7 and the related proteases Furin and Pace4 regulate E-cadherin function during blastocyst formation. 32325687_Lack of PCSK6 Increases Flow-Mediated Outward Arterial Remodeling in Mice. 36362216_Proprotein Convertase Subtilisin/Kexin 6 in Cardiovascular Biology and Disease. |
ENSG00000140479 |
PCSK6 |
1483.047420 |
0.4576718116 |
-1.127615 |
0.13688547 |
66.839435 |
0.00000000000000029454654381684945843259805339775975171560850533573894249883551310631446540355682373046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000083703973048847965147624474329298380750945208703117650372860225616022944450378417968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
929.53192261019 |
88.3280303904569 |
2031.00103387796 |
142.958484586173 |
| ENSMUSG00000030522 |
233315 |
Mtmr10 |
protein_coding |
Q7TPM9 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:233315; |
cytoplasm [GO:0005737]; phosphatidylinositol-3-phosphate phosphatase activity [GO:0004438]; phosphatidylinositol dephosphorylation [GO:0046856] |
|
ENSG00000166912 |
MTMR10 |
379.533995 |
0.4535236645 |
-1.140750 |
0.14398710 |
62.350323 |
0.00000000000000287484221338479821035996037047212410791007268159868992540850740624591708183288574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000747262740516676528982901666126916861136340528015686857088439865037798881530761718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
232.784376798665 |
23.2714603120846 |
513.279449409139 |
37.0448108137124 |
| ENSMUSG00000030562 |
50490 |
Nox4 |
protein_coding |
Q9JHI8 |
FUNCTION: Constitutive NADPH oxidase which generates superoxide intracellularly upon formation of a complex with CYBA/p22phox. Regulates signaling cascades probably through phosphatases inhibition. May function as an oxygen sensor regulating the KCNK3/TASK-1 potassium channel and HIF1A activity. May regulate insulin signaling cascade. May play a role in apoptosis, bone resorption and lipolysaccharide-mediated activation of NFKB. {ECO:0000269|PubMed:10869423, ECO:0000269|PubMed:11098048}. |
Alternative splicing;Cell junction;Cell membrane;Disulfide bond;Endoplasmic reticulum;Glycoprotein;Membrane;NADP;Oxidoreductase;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:50490; |
apical plasma membrane [GO:0016324]; cell periphery [GO:0071944]; endoplasmic reticulum membrane [GO:0005789]; focal adhesion [GO:0005925]; mitochondrion [GO:0005739]; NADPH oxidase complex [GO:0043020]; nucleus [GO:0005634]; perinuclear endoplasmic reticulum [GO:0097038]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; stress fiber [GO:0001725]; modified amino acid binding [GO:0072341]; NAD(P)H oxidase H2O2-forming activity [GO:0016174]; oxidoreductase activity, acting on NAD(P)H, oxygen as acceptor [GO:0050664]; protein tyrosine kinase binding [GO:1990782]; superoxide-generating NAD(P)H oxidase activity [GO:0016175]; aging [GO:0007568]; bone resorption [GO:0045453]; cardiac muscle cell differentiation [GO:0055007]; cell morphogenesis [GO:0000902]; cellular response to cAMP [GO:0071320]; cellular response to gamma radiation [GO:0071480]; cellular response to glucose stimulus [GO:0071333]; cellular response to transforming growth factor beta stimulus [GO:0071560]; defense response [GO:0006952]; gene expression [GO:0010467]; heart process [GO:0003015]; homocysteine metabolic process [GO:0050667]; negative regulation of cell population proliferation [GO:0008285]; positive regulation of apoptotic process [GO:0043065]; positive regulation of DNA biosynthetic process [GO:2000573]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of hydrogen peroxide biosynthetic process [GO:0010729]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein tyrosine kinase activity [GO:0061098]; positive regulation of reactive oxygen species metabolic process [GO:2000379]; positive regulation of smooth muscle cell migration [GO:0014911]; positive regulation of stress fiber assembly [GO:0051496]; reactive oxygen species metabolic process [GO:0072593]; response to hypoxia [GO:0001666]; superoxide anion generation [GO:0042554]; superoxide metabolic process [GO:0006801] |
12855428_neutrophil subunit gp91phox contributes to NADPH oxidase function in vascular fibroblasts, but not vascular smooth muscle cells. 14551238_N-acetyl-cysteine, inhibited pressure-overload-induced LVH in both gp91phox-/- and wild-type mice. These data suggest a differential response of the cardiac Nox isoforms, gp91phox and Nox4, to Ang II versus pressure overload 15108351_JNK was activated and that NF-kappa B was inhibited in Nox4/P22 cotransfected osteoclasts 15802177_Presence of the ROS producing NADPH oxidase NOX4 in neurons and demonstrates an upregulation of its expression under ischemic conditions. 16775014_NOX4 regulates vertebrate developmental processes through Reactive Oxygen Species-dependent p38 mitogen-activated protein kinase activation and nuclear translocation of the cardiac transcription factor myocyte enhancer factor 2C (MEF2C). 17443690_Nox4 expression was not altered during glucose-induced oxidative stress in microvascular endothelial cells. 17553579_Data suggest that decreased NOX4 mRNA content is a hallmark of adipocyte differentiation and that NOX4 expression measured in whole adipose tissue is not an unequivocal indicator of intact or impaired insulin action. 17568784_Pitavastatin ameliorates diabetic nephropathy in db/db mice by minimizing oxidative stress by downregulating NOX4 expression. 17585072_NOX4 was found to be upregulated in the pulmonary vasculature in chronic hypoxic pulmonary hypertension suggesting a contribution of NOX4 to the development of this disease 18417717_Suggest that a NOX4 isotype plus p22 phox account for the swelling-induced increase in the reactive oxygen species production in NIH3T3 cells. 18440230_Data demonstrated that increased Nox4 is unique to cardiac tissue of dystrophic mice showing echocardiographic evidence of cardiomyopathy. 18467643_Nox2 and Nox4 exhibit distinctive patterns of acute activation by angiotensin II, TNFalpha, and insulin and regulate the activation of distinct protein kinases. 18554521_E2F factor(s) are positive regulators of NOX4 transcription in vascular smooth muscle cells 18952568_Chronic intermittent hypoxia-induced pulmonary hypertension was associated with increased lung levels of the NADPH oxidase subunits, Nox4 and p22phox. 19036941_Embryonic stem cell differentiation into smooth muscle cells is mediated by Nox4-produced H2O2. 19057021_Nox4 acts as a switch between differentiation and proliferation in preadipocytes. 19204183_Macula densa cells express the NOX2 and NOX4 isoforms, and NOX2 is primarily responsible for NaCl-induced O2- generation. 19471020_MKs isolated from thrombopoietin (TPO)-treated bone marrow (BM) cultures indicated high expression of Nox1, a weak expression of Nox4, and no significant expression of Nox2. 19542021_Results suggest that NADPH oxidase-dependent superoxide production links palmitate-stimulated TLR4 activation to NF-kappaB signaling in endothelial cells. 19701206_NOX-4 is upregulated in noninfectious lung injury and has a role in tissue repair functions of myofibroblasts and fibrogenesis. 19817625_Results connect Nox-4 to MCP-1 to form a major axis of control that regulates the fate of hemangioendothelioma development in vivo. 19925862_increased protein levels in irradiated hematopoietic stem cells 20185797_Upregulation of Nox4 by hypertrophic stimuli and aging induces oxidative stress, apoptosis and left ventricular dysfunction, via mitochondrial insufficiency caused by increased O(2)(-) production and cysteine oxidation in mitochondrial proteins. 20224597_Besides the activation of p38, the gp96/TLR4 interaction is required for activation of ASK1/JNK signaling in posthypoxic renal tubule epithelial cells. The 28-kDa NOX4 has a key role in TLR4-mediated apoptosis during renal ischemia/reperfusion injury. 20363222_Nox4 siRNA reduced NMDA-induced H2O2 production by 54% and cytotoxicity in parallel, suggesting that Nox4-containing NADPH oxidase functions NMDA receptor-mediated H2O2 production resulting in neurotoxicity. 20413786_Nox4-mediated oxidative stress in the paraventricular nucleus plays a causal role in cardiac dysfunction following myocardial infaction. Nox4 silencing may be cardioprotective. 20427574_These data suggest an important role of the signaling axis between NOX4 and HIF-1alpha in various cardiovascular disorders under hypoxic and also nonhypoxic conditions. 20493939_increased superoxide formation by upregulated NADPH oxidase contributes significantly to age-dependent alterations in calcium handling and contractility of murine ventricular myocytes 20606471_IL-17A causes the NAD(P)H-oxidase dependent generation of reactive oxygen species leading to a pro-inflammatory activation of vascular endothelium 20630933_in diabetic conditions: 1) renal Nox4 is upregulated in a cortex-specific manner, 2) proximal tubule cells possess functionally active Nox4-based NADPH, 3) Nox4 is a major source of renal ROS. 20686447_Bilirubin and biliverdin protect rodents against diabetic nephropathy by downregulating NAD(P)H oxidase. 20713697_Results suggest that Nox4 in cardiac myocytes is a major source of mitochondrial oxidative stress, thereby mediating mitochondrial and cardiac dysfunction during PO. 20861022_AMPK negatively regulates Nox4-dependent activation of p53 and podocytes apoptosis in diabetes. 20877473_details indicate both the strong specificity and universality of NOX4 deficiency in protecting against stroke-mediated brain damage 20877715_Upon ischemia, NOX4 was induced in human and mouse brain. Mice deficient in NOX4 (Nox4(-/-)) of either sex, but not those deficient for NOX1 or NOX2, were largely protected from oxidative stress, blood-brain-barrier leakage, and neuronal apoptosis 20921387_data indicate that cardiomyocyte Nox4 is a unique inducible regulator of myocardial angiogenesis, a key determinant of cardiac adaptation to overload stress 20952384_ROS generated by Nox2 or Nox4 are essential for survival and differentiation in the early stage of chondrogenesis. 21029048_results indicate that Nox4-derived reactive oxygen species regulate BMP-2 (bone morphogenetic protein-2) gene transcription induced by BMP-2 21212270_Control of hepatic nuclear superoxide production by glucose 6-phosphate dehydrogenase and NADPH oxidase-4 21217672_Question of whether NOX-4 is expressed in fibrotic foci of subjects with idiopathic pulmonary fibrosis remains open. 21343298_H(2)O(2) formation is an intrinsic property of Nox4 that involves its E-loop. 21391892_ROS generation by NOX4 is a key player in epithelial cell death leading to pulmonary fibrosis. 21415386_Endothelial Nox4 exerts potentially beneficial effects on vasodilator function and blood pressure that are attributable to H(2)O(2) production. 21513813_NOX4 is increased in pulmonary fibroblasts from IPF patients and deletion of Nox4 in mice prevents bleomycin-induced pulmonary fibrosis. 21554947_demonstrate that Nox4 can activate the Nrf2-regulated pathway, and suggest a potential role for Nox4 in the regulation of GSH redox in cardiomyocytes 21788590_In wild-type C57/Bl6 mice with hindlimb ischemia there was increased Nox4 expression in ischemic gastrocnemius & thigh adductor muscles. 21896730_Data show that O(2)-coupled hydrogen peroxide production by Nox4 governs the redox state of regulatory RyR1 thiols and thereby governs muscle performance. 21935366_NADPH oxidase subunit 4-mediated reactive oxygen species contribute to cycling hypoxia-promoted tumor progression in glioblastoma multiforme 22031853_Insulin treatment rapidly increased the generation of H(2)O(2) in podocytes, and it increased the surface expression of the NADPH oxidase NOX4 in cultured podocytes. 22095986_NADPH oxidase 4 mediates monocyte priming and accelerated chemotaxis induced by metabolic stress. 22401855_found that alpha-MSH activated its downstream signal transducer microphthalmia-associated transcription factor (MITF) to stimulate Nox4 gene expression 22456182_Endogenous Nox4 protects the vasculature during ischemic or inflammatory stress. 22749956_The first in vivo data of the kidney do not support the view that Nox4 is a main driver of renal disease. It rather appears that under specific conditions Nox4 may even slightly limit injury and disease progression. 22824863_NADPH oxidase-mediated upregulation of Cx43 contributes to podocyte injury. 22836249_Our results suggest that Nox4 essentially contributes to nociceptive processing in neuropathic pain states. 22844079_The NADPH subunit Nox4 plays a crucial role in inhibiting DNA damage by Ang II. 22896039_We conclude that Nox4 mediates flow-induced superoxide production in thick ascending limb of loop of Henle. 22920224_Data indicate that renal mRNA expression of Nox4 increased, and Nox1 was barely detectable in db/db (diabetic) mice. 23049784_NADPH oxidase NOX4 mediates stellate cell activation and hepatocyte cell death during liver fibrosis development. 23100220_The absence of NOX4 promotes kidney fibrosis, independent of NOX2, through enhanced tubular cell apoptosis, decreased microvascularization, and enhanced oxidative stress. 23152062_MiR23b effectively reduces neuropathic pain by reducing the expression of the NOX4. 23216904_Ablation of Nox4 by siRNAs blocks the Ras-induced senescence in fibroblasts and overexpression of Nox4 induces senescence in fibroblasts 23261430_Nox4 acts as an intermediary in the signaling of TGFbeta(1) to facilitate collagen synthesis. 23271793_Knockdown of Nox4, but not Nox2, attenuated O(2)(-) production in the nucleus and prevented phenylephrine-induced oxidation and nuclear exit of HDAC4. 23329835_Data indicate that thioredoxin-interacting protein (TxNIP) is a critical component of the high glucos-reactive oxygen species signaling pathway, required for the induction of mitochondrial and total cell ROS and the NADPH oxidase isoform, Nox4. 23393389_Nox4D is a nuclear-localized and functionally active splice variant of Nox4 that may have important pathophysiologic effects through modulation of nuclear signaling and DNA damage in vascular cells. 23413427_Toll-like receptor 4 plays a key role in obesity and diabetes-associated endothelial dysfunction by increasing oxidative stress via NOX1/4. 23503725_Cellular differentiation may be affected by Nox4-mediated endogenous ROS generation. 23589292_NADPH oxidase 4 regulates cardiomyocyte differentiation via redox activation of c-Jun protein and the cis-regulation of GATA-4 gene transcription. 23612968_Disruption of Nox4 recruitment to SHPS-1 in aorta from diabetic mice inhibited IGF-I-stimulated Src oxidation and activation as well as cell proliferation. 23678045_our study is the first to provide evidence that Nox4 is downstream of ADAM17 23751520_angiotensin II/Nox4 axis is an important regulator of neural stem cell self-renewal and as such may fine-tune normal, stress- or disease-modifying neurogenesis 23832701_In conclusion, we report that c-kit(+) CPCs demonstrate reduced Nox2 expression and ROS levels and that increases in Nox2 and Nox4 influence their differentiation into mature cells. 23839498_An increase in NADPH oxidase activity might be an early hallmark to predict future oxidative stress in renal tissue during the aging process. 23884197_Atrial fibroblasts show greater fibrotic and oxidative responses to TGF-beta1 than ventricular fibroblasts and suggest role for Nox4. 23933064_SphK1 deficiency attenuated hyperoxia-induced accumulation of IL-6 in bronchoalveolar lavage fluids and NADPH oxidase (NOX) 2 and NOX4 protein expression in lung tissue. 24051369_Following 30min of ischemia and 24h of reperfusion, a significant decrease in the size of myocardial infarct was observed in Nox1-, Nox2- and Nox1/Nox2-, but not in Nox4-deficient mice. 24081881_Nox4 critically mediates autophagy in response to energy stress in cardiomyocytes by eliciting reactive oxygen species in the endoplasmic reticulum and stimulating the protein kinase RNA-activated-like ER kinase signaling pathway. 24205282_circadian clock plays an important role in the control of Nox4 24213612_Nox4 mRNA and protein levels increase in early pulmonary hypertension induced by pulmonary artery banding. 24216508_Nox4(-/-) mice displayed higher bone density and reduced numbers and markers of osteoclasts. Ex vivo, differentiation of monocytes into osteoclasts with RANKL and M-CSF induced Nox4 expression. 24371139_CnAbeta/NFAT pathway modulates Nox. 24432311_Metformin, which increases extracellular ATP concentration leads to activation of P2 receptors and consequent modulation of the podocytes' metabolism through AMPK and NAD(P)H oxidase which, in turn, may affect podocyte functioning 24466344_NADPH oxidase 4 deficiency reduces aquaporin-2 mRNA expression in cultured renal collecting duct principal cells via increased PDE3 and PDE4 activity. 24482376_Identify iPLA2beta and lysophosphatidic acid derived from its action as critical in regulating macrophage Nox4 activity and migration in the diabetic state in vivo and under similar conditions ex vivo. 24497637_significance of dicer and microRNA in promoting endothelial cell tumor growth in vivo 24511132_These results identify Nox4 as a key source of ROS responsible for kidney injury in diabetes and provide proof of principle for an innovative small molecule approach to treat and/or prevent chronic kidney failure. 24657416_Schistosoma japonicum infection causes upregulation of cannabinoid receptor-1 and NADPH oxidase leading to fibrotic activation of mouse hepatic stellate cells. 24718857_Restoration of Nox4-Nrf2 redox balance in myofibroblasts may be a therapeutic strategy in age-associated fibrotic disorders. 24936437_Increased expression of Nox4 in AS may drive SMC phenotypes that lead to the plaque instability and rupture responsible for myocardial infarction and stroke. 24947524_Nox4 is prominently expressed in the adventitia and contributes to altered fibroblast behavior, hypertensive vascular remodeling, and development of pulmonary hypertension. 25152242_Weakly positive reactions with Nox1, Noxa1, and Noxo1 were observed in the zones of proliferative and prehypertrophic chondrocytes at 3 weeks of age 25229402_Smad3-Nox4 axis-mediated mitochondrial dysfunction is involved in puromycin aminonucleoside-induced podocyte damage. 25410908_Results suggest that Nox4-mediated reactive oxygen species generation likely plays important role in fate determination and differentiation of neural crest stem cells. 25534702_Nicotinamide adenine dinucleotide phosphate oxidase-4-dependent upregulation of nuclear factor erythroid-derived 2-like 2 protects the heart during chronic pressure overload. 25652847_Nox4 maintains VEGF expression and prevents an increase in angiopoietin 1 expression, thereby contributing to angiogenesis in exercise. Nox4 does not influence developmental angiogenesis. 25866826_Nox4 promotes retinal neovascularization through H2O2/VEGFR2/ERK signaling pathway 25982278_IFNgamma induces oxidative stress, DNA damage and tumor cell senescence via TGFbeta/SMAD signaling-dependent induction of Nox4 and suppression of ANT2 26036345_The present study provides evidence that augmented EGFRtk impairs vascular function by NADPH oxidase-dependent mechanism. Therefore, EGFRtk and oxidative stress should be potential targets to treat vascular dysfunction in TD2. 26054376_Data suggest that NOX4 and mitochondrial oxidative stress are mediators of cardiovascular disease in aging under hyperlipidemic conditions. 26196532_Intradermal administration of siHSP47-nanoparticles effectively reduced HSP47 protein expression in skin to normal level. 26222337_NOX1 and NOX4 signaling mediates the pathogenesis of liver fibrosis, including the direct activation of HSC. 26231202_activated PKCzeta phosphorylated p65 rel, which led to increased Nox4 synthesis 26237157_NOX4 may thus associate with mitochondrial complex I proteins, but in cardiac and renal mitochondria under basal conditions, expression is beyond our detection limits. 26297224_Nox4 mRNA expression was increased in diet-induced obese (DIO) mice. 26408539_Fluid shear stress regulates PLGF expression via NADPH oxidase 4 and reactive oxygen species signaling. 26412187_eNOS plays a key role in the regulation of expression of NOX2 and NOX4 in the lungs exposed to hypoxia. 26445208_These results indicate that Ang-II induces cardiac fibroblast proliferation and migration in part via Nox4/ROS-dependent IL-18 induction and MMP9 activation, and may involve AT1/Nox4 physical association. 26508318_Deleterious effect of Nox4 expression in podocytes by promoting podocytopathy in association with albuminuria and extracellular matrix accumulation in experimental diabetes, emphasizes the role of NOX4 as a target for new renoprotective agents. 26513738_Nox4 promotes tumour angiogenesis and may represent a novel target for anti-angiogenic tumour therapy. 26565025_ERK1/2 plays an important role in TGF-beta1-induced mTOR activation and translational regulation of Nox4 protein. 26572549_TRAIL can promote angiogenesis following hindlimb ischemia in vivo via NOX4/eNOS/nitric oxide signaling. 26582463_Downregulation of smooth muscle Nox4 inhibits neointimal hyperplasia by suppressing TSP1. 26661159_Nox4 is upregulated in pericytes in peri-infarct areas after acute brain ischemia and may enhance blood-brain barrier breakdown through activation of NFkappaB and matrix metalloproteinase 9, thereby causing enlargement of infarct volume. 26715682_H2O2-forming NOX4, unlike the superoxide-forming NOX1, can act as a negative modulator of inflammation and remodeling and convey atheroprotection. 26731985_current data suggested that miR-31 contributed to macrophage apoptosis by regulating the expression of NOX4 26812119_of smooth muscle Nox4 inhibits atherosclerosis by suppressing sEH, which, at least in part, accounts for inhibition of SMC proliferation, migration and inflammation 26908870_Data (including data from studies using transgenic mice) suggest that Glut1 (glucose transporter type 1) is a critical downstream target of Hif1a (hypoxia-inducible factor 1, alpha subunit) mediating hyperglycemia-induced extracellular matrix accumulation in kidney via regulation of Nox4 (NADPH oxidase type 4) expression in nephropathy due to diabetes type 1. 26945079_Nox4 mitigates cerebral artery structural changes in a murine model of Marfan syndrome. 26952102_the reduction of ROS generation and NOX4 expression by EA preconditioning might be involved in BBB recovery. Therefore, EA may serve as a potential preventive strategy for patients at high risk of ischemic stroke. 27140231_Strong dityrosine formation was observed, but was significantly weaker in NOX4-/- mice (p |
ENSG00000086991 |
NOX4 |
91.234101 |
2.4996715431 |
1.321739 |
0.44010545 |
8.428585 |
0.00369367750432136529581406136912846704944968223571777343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0143014986644186669478839846192386175971478223800659179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
133.607933906463 |
41.3932576982511 |
53.4501959976843 |
12.3976341753874 |
| ENSMUSG00000030587 |
72275 |
2200002D01Rik |
protein_coding |
Q9D809 |
|
Proteomics identification;Reference proteome |
|
|
mmu:72275; |
cytosol [GO:0005829]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886] |
Human_homologues 11606055_H2RSP and H2RSP/HAI-2 chimeric peptides might function as a transcriptional regulatory peptide at the nucleus. 14981917_results showed the up-regulation of IMUP-1 and IMUP-2 in human ovarian epithelial tumors and suggested that the altered mRNA level of these molecules is possibly associated with ovarian tumorigenesis 16189703_Nuclear translocation of H2RSP may be related to a signaling involved in the transition from cellular proliferation to differentiation in experimental murine colitis. 17309599_Our findings suggest that the balance between hepsin and its inhibitor, HAI-2, may have prognostic value in RCC. 18507030_These findings demonstrate the up-regulation of imup-1 and imup-2 in human endometrial carcinomas and indicate that these molecules play a role in endometrial carcinogenesis in both Korean and Japanese patients. 20432246_results suggest that IMUP-2 expression is specifically elevated in preterm pre-eclampsia and under hypoxic conditions, and that IMUP-2 induces apoptosis of the trophoblast 22886722_Hypoxia-induced down-regulation of XIAP mediates apoptosis in trophoblasts through interaction with increased IMUP-2; this mechanism underlies the pathogenesis of pre-eclampsia. |
ENSG00000167644 |
C19orf33 |
35.911995 |
2.7602535253 |
1.464801 |
0.48254188 |
8.664012 |
0.00324555943364214808874446127617829915834590792655944824218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0128139868567229645718308361779236292932182550430297851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.324654102895 |
17.8293048374224 |
18.9564667245581 |
4.94451464549102 |
| ENSMUSG00000030641 |
74041 |
Ddias |
protein_coding |
A0A140LHT2 |
Human_homologues FUNCTION: May be an anti-apoptotic protein involved in DNA repair or cell survival. {ECO:0000269|PubMed:24214091}. |
Reference proteome |
|
|
|
negative regulation of intrinsic apoptotic signaling pathway in response to DNA damage [GO:1902230] |
17515607_Noxin may be a component of the cell defense system: it is activated by various stress stimuli, helps cells to withdraw from cycling, and opposes apoptosis. |
ENSG00000165490 |
DDIAS |
167.486912 |
2.0178044522 |
1.012786 |
0.22973010 |
18.987961 |
0.00001315458190107452080030211311045817978992999996989965438842773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000953691535065050481016074579443397851719055324792861938476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
219.877650007543 |
35.1607597428129 |
108.968760462521 |
13.3132541075684 |
| ENSMUSG00000030653 |
207728 |
Gm45837 |
protein_coding |
F7D3W5 |
|
cGMP;Hydrolase;Metal-binding;Reference proteome |
|
|
|
cytosol [GO:0005829]; mitochondrial inner membrane [GO:0005743]; mitochondrial matrix [GO:0005759]; mitochondrial outer membrane [GO:0005741]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; synaptic membrane [GO:0097060]; 3',5'-cyclic-AMP phosphodiesterase activity [GO:0004115]; 3',5'-cyclic-GMP phosphodiesterase activity [GO:0047555]; 3',5'-cyclic-nucleotide phosphodiesterase activity [GO:0004114]; cGMP-stimulated cyclic-nucleotide phosphodiesterase activity [GO:0004118]; metal ion binding [GO:0046872]; protein homodimerization activity [GO:0042803]; cAMP-mediated signaling [GO:0019933]; cGMP catabolic process [GO:0046069]; cGMP-mediated signaling [GO:0019934]; negative regulation of cAMP-mediated signaling [GO:0043951]; negative regulation of cGMP-mediated signaling [GO:0010754]; positive regulation of gene expression [GO:0010628]; regulation of mitochondrion organization [GO:0010821]; signal transduction [GO:0007165] |
15210692_cGMP binding to PDE2 GAF-B activates the enzyme rapidly, stoichiometrically, and in an all or none fashion, rather than variably over a large range of cyclic nucleotide concentrations 18849438_HV(T)-induced endothelial barrier dysfunction resulted from a simultaneous increase in NO/sGC-derived cGMP and PDE2A expression causing decreased cAMP. 19390057_Phosphodiesterase 2 mediates redox-sensitive endothelial cell proliferation and angiogenesis by thrombin via Rac1 and NADPH oxidase 2. 19632989_dual acylation as mechanism targeting neuronal PDE2A3 to synapses thereby ensuring local control of cyclic nucleotides. 23045693_results demonstrate strong presynaptic inhibition by natriuretic peptides in the brain and suggest important physiological and behavioral roles of PDE2A in modulating neurotransmitter release 25063878_We conclude that increased AM PDE2A is an important negative regulator of macrophage iNOS expression. 25442113_Effect of PDE2 inhibition on stress-induced memory impairment is potentially mediated via modulation of neuroplasticity-related NMDAR-CaMKII-cGMP/cAMP signaling. 26243800_Cardiac hypertrophy is inhibited by a local pool of cAMP regulated by PDE2A. 29409032_Pde2A is indispensable for heart development because Pde2A knockout embryos show severe atria and ventricular defects. 31100470_PDE2 regulates membrane potential, respiration and permeability transition of rodent subsarcolemmal cardiac mitochondria. 34062838_Cellular Mechanisms of the Anti-Arrhythmic Effect of Cardiac PDE2 Overexpression. |
|
|
50.490857 |
6.7732074136 |
2.759839 |
0.66192389 |
16.390410 |
0.00005154535376879542865743641377740402731433277949690818786621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003296132781722468235718892515251354780048131942749023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.693514933545 |
47.8372858725977 |
14.8664449181912 |
5.37507001437934 |
| ENSMUSG00000030657 |
233781 |
Xylt1 |
protein_coding |
F8VPK6 |
Human_homologues FUNCTION: Catalyzes the first step in the biosynthesis of chondroitin sulfate and dermatan sulfate proteoglycans, such as DCN. Transfers D-xylose from UDP-D-xylose to specific serine residues of the core protein (PubMed:15461586, PubMed:17189265, PubMed:24581741, PubMed:23982343). Required for normal embryonic and postnatal skeleton development, especially of the long bones (PubMed:24581741, PubMed:23982343). Required for normal maturation of chondrocytes during bone development, and normal onset of ossification (By similarity). {ECO:0000250|UniProtKB:Q811B1, ECO:0000269|PubMed:15461586, ECO:0000269|PubMed:17189265, ECO:0000269|PubMed:23982343, ECO:0000269|PubMed:24581741}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Membrane;Metal-binding;Proteomics identification;Reference proteome;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Glycan metabolism; chondroitin sulfate biosynthesis. {ECO:0000256|ARBA:ARBA00004840}.; PATHWAY: Glycan metabolism; heparan sulfate biosynthesis. {ECO:0000256|ARBA:ARBA00005093}. |
|
mmu:233781; |
extracellular space [GO:0005615]; Golgi membrane [GO:0000139]; metal ion binding [GO:0046872]; protein xylosyltransferase activity [GO:0030158]; chondroitin sulfate biosynthetic process [GO:0030206]; heparan sulfate proteoglycan biosynthetic process [GO:0015012] |
24161523_We show that the pug mutation disrupts Xylt1 activity and subcellular localization, leading to a reduction in GAG chains in pug mutants. The pug mutant serves as a novel model for mammalian dwarfism. 26414444_Notch-modifying xylosyltransferase structures support an SNi-like retaining mechanism. 32965647_Xylosyltransferase 2 deficiency and organ homeostasis. |
ENSG00000103489 |
XYLT1 |
25.029023 |
0.0587470086 |
-4.089341 |
0.76954202 |
28.451797 |
0.00000009606057458474129943384010876794576461179531179368495941162109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000010132927091930606425512068069494375777139794081449508666992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.7198683900762 |
1.52057624201325 |
46.2979895453083 |
15.2508481060112 |
| ENSMUSG00000031004 |
17345 |
Mki67 |
protein_coding |
E9PVX6 |
FUNCTION: Required to maintain individual mitotic chromosomes dispersed in the cytoplasm following nuclear envelope disassembly (PubMed:27362226). Associates with the surface of the mitotic chromosome, the perichromosomal layer, and covers a substantial fraction of the chromosome surface (PubMed:27362226). Prevents chromosomes from collapsing into a single chromatin mass by forming a steric and electrostatic charge barrier: the protein has a high net electrical charge and acts as a surfactant, dispersing chromosomes and enabling independent chromosome motility (PubMed:27362226). Binds DNA, with a preference for supercoiled DNA and AT-rich DNA (By similarity). Does not contribute to the internal structure of mitotic chromosomes (PubMed:26949251). May play a role in chromatin organization (PubMed:26949251). It is however unclear whether it plays a direct role in chromatin organization or whether it is an indirect consequence of its function in maintaining mitotic chromosomes dispersed. {ECO:0000250|UniProtKB:P46013, ECO:0000269|PubMed:26949251, ECO:0000269|PubMed:27362226, ECO:0000305}. |
Acetylation;Alternative splicing;ATP-binding;Cell cycle;Chromosome;DNA-binding;Isopeptide bond;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Ubl conjugation |
|
|
mmu:17345; |
chromosome [GO:0005694]; chromosome, centromeric region [GO:0000775]; condensed chromosome [GO:0000793]; cytoplasm [GO:0005737]; nuclear body [GO:0016604]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; ATP binding [GO:0005524]; DNA binding [GO:0003677]; protein C-terminus binding [GO:0008022]; cell population proliferation [GO:0008283]; cholangiocyte proliferation [GO:1990705]; epithelial cell proliferation [GO:0050673]; hepatocyte proliferation [GO:0072574]; meiotic cell cycle [GO:0051321]; regulation of chromatin organization [GO:1902275]; regulation of chromosome segregation [GO:0051983]; regulation of mitotic nuclear division [GO:0007088]; stem cell proliferation [GO:0072089] |
12168898_Vascular endothelial growth factor (VEGF) expression correlates with p53 and ki-67 expressions in tongue squamous cell carcinoma. 12175484_Age-related changes in proliferative markers in labial salivary glands: a study of argyrophilic nucleolar organizer regions (AgNORs) and Ki-67 15237214_the re-establishment of the cell cycle-dependent distribution of pKi-67 during early mouse development 16410075_p63 protein is essential for the embryonic development of vibrissae and teeth; while it localizes with K5 in vibrissae, it is not fully colocalized with nuclear Ki67 expression 18649926_Data show that C57BL/6 mice showed significantly higher levels of Ki-67-immunopositive cells in the subgranular zone (SGZ) of the DG compared to ICR and BALB/c mice. 19385968_Immunohistochemical staining studies show that greast tumor cells positive for ALDH1 are more likely to be Ki67 positive. 19644632_Ki-67 antigen was found to be increased by urethane, but K-ras exon 1 mutations do not play any role in the interfering effects of urethane followed by sodium nitrite and sodium chloride 19863319_The majority of tumours showed strong p16, p21, p27, pRb and cyclin D1 staining and little or no p53 expression. Tumours harbouring dysplasia were significantly more likely to be p53-positive and exhibit up-regulated p21 and p27. 20336306_no difference in antigen expression between acute generalized exanthematous pustulosis and pustular psoriasis 22789860_Late stage cathepsin C, CXCL13 and Ki-67 overexpression correlate with regional neuropathology in a bovine spongiform encephalopathy transgenic murine model. 23417197_Intratumoral FLT uptake level markedly decreased at 6 h and then gradually increased with time. 23744762_Both HDAC1 and HDAC2 play crucial roles in the regulation of liver regeneration. The loss of HDAC1/2 inhibits Ki67 expression and results in defective hepatocyte mitosis and impaired liver regeneration. 24174624_Whey proteins reduced Ki-67 and 8-OHdG expression in the skin of chronically UVB-irradiated mice. 26042621_Ki67-positive cells are localized near inner enamel epithelium and supra-IEE, the stellate reticulum next to these is Ki67-negative. The IEE and supra-IEE contain intense Ki67-immunoreactivity, outer enamel epithelium lacks proliferative cells in mice. 27846311_injection of a b3-adrenergic receptor (b3-AR) agonist for continuous 5 days increased the number of Ki67-positive brown adipocytes even at Day 1 but not that of SV cells. In addition, the b3-AR antagonist, but not b1-AR antagonist, attenuated the cold exposure-induced increase in the number of Ki67-positive brown adipocytes 30472241_This study demonstrated that thr Ki-67 expression was mainly observed in the olfactory bulb, rostral migratory stream, sub ventricular zone of lateral ventricle and the sub granular zone of the dentate gyrus and fewer Ki-67 positive cells were also observed in the neocortex, cerebellum and tectum. 33094683_Deletion of NKX3.1 via CRISPR/Cas9 Induces Prostatic Intraepithelial Neoplasia in C57BL/6 Mice. 33247410_Photobiomodulation invigorating collagen deposition, proliferating cell nuclear antigen and Ki67 expression during dermal wound repair in mice. 33301235_Somatostatin Analogs for Pancreatic Neuroendocrine Tumors: Any Benefit When Ki-67 Is >/=10%? 33658388_Ki-67 regulates global gene expression and promotes sequential stages of carcinogenesis. 33897702_To Ki or Not to Ki: Re-Evaluating the Use and Potentials of Ki-67 for T Cell Analysis. 34073605_S1P Lyase Regulates Intestinal Stem Cell Quiescence via Ki-67 and FOXO3. 35187889_Upregulation of h-TERT and Ki-67 in ectopic endometrium is associated with recurrence of endometriosis. 35248490_Changes in Ki-67 in Residual Tumor and Outcome of Primary Inflammatory Breast Cancer Treated With Trimodality Therapy. 36161862_Expression of Ki67 as detected by MIB-1 and its association with histopathological high-risk factors among patients with retinoblastoma tumour: a cross-sectional study. 36510965_Immunohistochemical expression of Ki-67 and Glypican-3 to distinguish aggressive from nonaggressive benign odontogenic tumors. |
ENSG00000148773 |
MKI67 |
5888.240052 |
2.1979007377 |
1.136126 |
0.04987969 |
510.714460 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000004433726017187307423220370749336223402696600621546403008806615975679310556590234784486434605126404855334158633785393564354907850660345838007866437931749 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000306975066789986656611717359024148943126051080309794479756704916945383499879252205002656937444621707607762918885682409228396151550907285546850949254366 |
Yes |
No |
8009.93691118403 |
276.990441305802 |
3644.35789746491 |
96.1002303091138 |
| ENSMUSG00000031098 |
55925 |
Syt8 |
protein_coding |
Q9R0N6 |
FUNCTION: Involved in the trafficking and exocytosis of secretory vesicles in non-neuronal tissues. Mediates Ca(2+)-regulation of exocytosis acrosomal reaction in sperm. May mediate Ca(2+)-regulation of exocytosis in insulin secreted cells. {ECO:0000269|PubMed:11751263, ECO:0000269|PubMed:15774481, ECO:0000269|PubMed:7791877}. |
Alternative splicing;Cell membrane;Cytoplasm;Cytoplasmic vesicle;Membrane;Reference proteome;Repeat;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:55925; |
acrosomal vesicle [GO:0001669]; axon [GO:0030424]; cytoplasm [GO:0005737]; dense core granule [GO:0031045]; exocytic vesicle [GO:0070382]; plasma membrane [GO:0005886]; synaptic vesicle membrane [GO:0030672]; vesicle [GO:0031982]; calcium ion binding [GO:0005509]; calcium-dependent phospholipid binding [GO:0005544]; calcium-dependent protein binding [GO:0048306]; clathrin binding [GO:0030276]; phosphatidylserine binding [GO:0001786]; SNARE binding [GO:0000149]; syntaxin binding [GO:0019905]; acrosome reaction [GO:0007340]; calcium ion-regulated exocytosis of neurotransmitter [GO:0048791]; calcium-ion regulated exocytosis [GO:0017156]; cellular response to calcium ion [GO:0071277]; regulation of calcium ion-dependent exocytosis [GO:0017158]; regulation of dopamine secretion [GO:0014059]; synaptic vesicle endocytosis [GO:0048488]; vesicle-mediated transport [GO:0016192] |
11751263_Syt VIII is a candidate for the Ca(2+) sensor that regulates acrosomal exocytosis in mammalian sperm |
ENSG00000149043 |
SYT8 |
52.379886 |
0.1305781310 |
-2.937015 |
0.41036567 |
46.742916 |
0.00000000000809369906627689900214554213206253458455508020819024750380776822566986083984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001452099224705886101542135407371238685347858421437194920144975185394287109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
11.5679858367772 |
3.33499240076594 |
88.590530049781 |
18.8093023909746 |
| ENSMUSG00000031099 |
93761 |
Smarca1 |
protein_coding |
Q6PGB8 |
FUNCTION: Helicase that possesses intrinsic ATP-dependent chromatin-remodeling activity (By similarity). ATPase activity is substrate-dependent, and is increased when nucleosomes are the substrate, but is also catalytically active when DNA alone is the substrate (By similarity). Catalytic subunit of ISWI chromatin-remodeling complexes, which form ordered nucleosome arrays on chromatin and facilitate access to DNA during DNA-templated processes such as DNA replication, transcription, and repair (By similarity). Within the ISWI chromatin-remodeling complexes, slides edge- and center-positioned histone octamers away from their original location on the DNA template (By similarity). Catalytic activity and histone octamer sliding propensity is regulated and determined by components of the ISWI chromatin-remodeling complexes (By similarity). The BAZ1A-, BAZ1B-, BAZ2A- and BAZ2B-containing ISWI chromatin-remodeling complexes regulate the spacing of nucleosomes along the chromatin and have the ability to slide mononucleosomes to the center of a DNA template (By similarity). The CECR2- and RSF1-containing ISWI chromatin-remodeling complexes do not have the ability to slide mononucleosomes to the center of a DNA template (By similarity). Within the NURF-1 and CERF-1 ISWI chromatin remodeling complexes, nucleosomes are the preferred substrate for its ATPase activity (By similarity). Within the NURF-1 ISWI chromatin-remodeling complex, binds to the promoters of En1 and En2 to positively regulate their expression and promote brain development (By similarity). May promote neurite outgrowth (By similarity). May be involved in the development of luteal cells (By similarity). {ECO:0000250|UniProtKB:P28370}. |
Activator;Alternative splicing;ATP-binding;Chromatin regulator;Coiled coil;DNA-binding;Helicase;Hydrolase;Isopeptide bond;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:93761; |
ATPase complex [GO:1904949]; CERF complex [GO:0090537]; chromatin [GO:0000785]; intracellular membrane-bounded organelle [GO:0043231]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; NURF complex [GO:0016589]; ATP binding [GO:0005524]; ATP-dependent activity, acting on DNA [GO:0008094]; ATP-dependent chromatin remodeler activity [GO:0140658]; ATP-dependent DNA/DNA annealing activity [GO:0036310]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; helicase activity [GO:0004386]; hydrolase activity [GO:0016787]; nucleosome binding [GO:0031491]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; brain development [GO:0007420]; chromatin remodeling [GO:0006338]; neuron differentiation [GO:0030182]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of neural precursor cell proliferation [GO:2000177] |
16740656_The imitation switch protein SNF2L regulates steroidogenic acute regulatory protein expression during terminal differentiation of ovarian granulosa cells. 19996304_Cancers are sensitive to SNF2L knockdown because, unlike their normal counterparts, they lack sufficient compensation from other family members. 23616592_These results begin to establish a role for SNF2L in the precise coordination of gene expression in granulosa cells during folliculogenesis and its broader implications in fertility. |
ENSG00000102038 |
SMARCA1 |
143.064832 |
6.6118795109 |
2.725060 |
0.28761383 |
85.278484 |
0.00000000000000000002591679942650657516077270501845119815838333053046717920249453204206702139344997704029083251953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000010200637955156281819999436256037844746755206055383144621295787857206960325129330158233642578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
245.570673044887 |
49.2815869289145 |
37.1408269979272 |
6.02868717663712 |
| ENSMUSG00000031125 |
70691 |
3830403N18Rik |
protein_coding |
Q9D6C3 |
Human_homologues FUNCTION: Component of the synaptonemal complexes (SCS), formed between homologous chromosomes during meiotic prophase. Required for centromere pairing during meiosis in male germ cells (By similarity). Required for normal meiosis during spermatogenesis and male fertility (PubMed:14643120). Plays a lesser role in female fertility. Required for efficient phosphorylation of HORMAD1 and HORMAD2 (By similarity). {ECO:0000250|UniProtKB:P70281, ECO:0000269|PubMed:14643120}. |
Coiled coil;Reference proteome |
|
|
mmu:70691; |
synaptonemal complex [GO:0000795]; hematopoietic progenitor cell differentiation [GO:0002244]; meiotic cell cycle [GO:0051321]; spermatid development [GO:0007286] |
Human_homologues 14643120_Observational study of gene-disease association. (HuGE Navigator) 14643120_We suggest that SYCP3 has an essential meiotic function in human spermatogenesis that is compromised by the mutant protein via dominant negative interference. 16213863_In contrast to previously reported high frequency of SYCP3 mutations in patients with azoospermia, only polymorphisms are found in the present study 16824523_SYCP3 is expressed in human testis and is restricted to germ cells. Lack of SYCP3 expression in human testis may have a negative effect on spermatogenesis and male fertility. 17434513_No functional SYCP3 exonic SNP was found in infertile Spanish patients with meiosis arrest, suggesting that SYCP3 mutations very likely are not relevant in Spain in genetic susceptibility to meiosis arrest. 18256545_These observations suggest that SCP3 is expressed in adult human and monkey ovaries. 19110213_Observational study of gene-disease association. (HuGE Navigator) 19110213_mutations in SYPR3 are likely to generate an aberrant synaptonemal complex in a dominant-negative manner and contribute to abnormal chromosomal behavior that might lead to recurrent miscarriage. 20395201_X-linked lymphocyte-regulated protein pM1 (XMR), XLR, and SCP3 render tumor cells resistant to antitumor immunity 21126912_mutation analysis of SYCP3, DNMT3L and MSH4 in patients with maturation arrest of spermatogenesis and couples with recurrent miscarriages; heterozygous change, detected in a conserved functional domain of the SYCP3 gene, was absent in >200 controls 21357605_The 657T>C mutation of SYCP3 may not be associated with recurrent miscarriage caused by aneuploidy. 22116401_Expression of SYCP3 inhibits the homologous recombination (HR) pathway mediated by RAD51. 22197129_mutations in SYCP3 do not contribute significantly to risk for recurrent miscarriage through maternal meiotic nondisjunction. 22670862_SYCP3 mutations are not associated with the genetic susceptibility for meiotic arrest in infertile male patients with nonobstructive azoospermia in the Turkish population 23069255_Positive synaptonemal complex protein 3 expression is a portent of poor outcome and may be a potential biomarker in the early stages of the non-small cell lung cancer for survival. 23100464_screening for genetic variants in AURKB and SYCP3 among these patients with reproductive problems using Sanger sequencing. Only one apparently non-pathogenic deletion was found in SYCP3. 24905095_SCP3 plays an important role in the progression of cervical cancer 24950965_SYCP3 binding and assembly on meiotic chromosomes leads to their organisation into compact structures compatible with recombination and crossover formation 25059562_The T657C polymorphism of the SYCP3 gene is possibly associated with recurrent pregnancy loss of unknown cause in human. 28287952_In the model, the reported compaction of chromosomal DNA caused by SYCP3 would result from its ability to bridge distant sites on a DNA molecule with the DNA-binding domains located at each end of its strut-like structure. 28623914_SCP3 is associated with lymphangiogenesis and provides insight into the SCP3-VEGF-C/VEGF-D axis based cancer therapy strategy. 28745000_SYCP3 binds to RAD51 and attenuates RAD51 activity during meiosis. |
ENSG00000139351 |
SYCP3 |
39.375094 |
0.4470797632 |
-1.161396 |
0.40697096 |
8.062713 |
0.00451854737487139990897988539586549450177699327468872070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0170616047630245820654337762789509724825620651245117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.5213302688582 |
6.43765309886638 |
50.3743003396143 |
10.2472526419464 |
| ENSMUSG00000031209 |
15203 |
Heph |
protein_coding |
Q9Z0Z4 |
FUNCTION: May function as a ferroxidase for ferrous (II) to ferric ion (III) conversion and may be involved in copper transport and homeostasis. Implicated in iron homeostasis and may mediate iron efflux associated to ferroportin 1. |
Alternative splicing;Copper;Copper transport;Disulfide bond;Glycoprotein;Ion transport;Iron;Iron transport;Membrane;Metal-binding;Oxidoreductase;Phosphoprotein;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:15203; |
basolateral plasma membrane [GO:0016323]; extracellular region [GO:0005576]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; copper ion binding [GO:0005507]; ferrous iron binding [GO:0008198]; ferroxidase activity [GO:0004322]; oxidoreductase activity [GO:0016491]; copper ion transport [GO:0006825]; iron ion homeostasis [GO:0055072]; iron ion transport [GO:0006826] |
12730111_Hephaestin and Ireg1 expression respond to systemic rather than local signals of iron status. 14751926_Hephaestin has both amine oxidase and ferroxidase activity primary intestinal enterocytes and may facilitate iron export from intestinal enterocytes; also activity and structural studies of truncated Heph in sla mice 15365174_Ceruloplasmin ane hephaestin are critical for CNS iron homeostasis and that loss of Cp and Heph in the mouse leads to age-dependent retinal neurodegeneration. 15825077_CDX2 expression was modulated in response to changes in intracellular iron levels, implying a regulatory pathway in which increased iron levels lead to increased expression of CDX2 and HEPH and enhanced iron export. 16614410_Copper is required for the proper processing and/or stability of hephaestin. 19747625_Expression of hephaestin (Heph) increased significantly in cerebral cortex and caudate putamen of old (80-week-old), ceruloplasmin-deficient (CP-/-) mice; expression of Heph was unaltered in substantia nigra and cerebellum. 21917813_results show that Heph is expressed by oligodendrocytes and plays an important role in iron efflux from these cells. 22342521_Cp and Heph are necessary for iron export from the retina but are not essential for iron import into the retina. 24896847_The multicopper ferroxidase hephaestin enhances intestinal iron absorption in mice. 25788583_Data (including data from studies in knockout mice) suggest that hephaestin and ceruloplasmin play distinct roles in regulation of gene expression in various regions of the brain and are involved in iron homeostasis. 26303407_In mice with mutation of Cp and Heph, iron accumulates in glia, while neurons have low iron levels. Both neurons and glia degenerate and mice become ataxic unless given an iron chelator. 27991585_Hephaestin and ceruloplasmin have roles in facilitating iron metabolism in the mouse kidney 29355933_Results show that both HEPH and CP are expressed in subcutaneous adipose tissue. Ablation of either MCF leads to a compensatory increase in the other, which contributes to the balance of iron status. However, ablation of both induces severe iron deposition in adipocytes and displays disordered carbohydrate metabolism characterized as type 2 diabetes. 29883959_We thus used hephaestin (Heph) and ceruloplasmin (Cp) single-knockout mice and Heph/Cp double-knockout mice to investigate the roles of multicopper ferroxidases(MCF) in pancreatic iron homeostasis. We found that both HEPH and CP were expressed in the mouse pancreas, and that ablation of either MCF had limited effect on the pancreatic iron levels. 30060949_results suggest that ablation of hephaestin and ceruloplasmin could lead to severe systemic iron deficiency and local tissue iron overload, which disrupt the whole body iron homeostasis and impact on tissue functions 30182051_HEPH and CP are not essential for intestinal iron absorption but are required for proper systemic iron distribution. |
ENSG00000089472 |
HEPH |
247.085728 |
26.7597865033 |
4.741995 |
0.26196182 |
384.151709 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000015526791351455625795264981193357554141670701512954641554339212456720606084996137015294037421817010935872652876588878183429318327566052977751800078488913479391464313879948637457133 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000675725959615348841925051285741480642426523638997968140937066492982392208595837279562445378400600768252045193094248968297615481477268406277969804134454594630323197617879628650714 |
Yes |
No |
470.947115062557 |
85.9843610683353 |
17.5990610016437 |
2.89959979071284 |
| ENSMUSG00000031214 |
94190 |
Ophn1 |
protein_coding |
Q99J31 |
FUNCTION: Stimulates GTP hydrolysis of members of the Rho family. Its action on RHOA activity and signaling is implicated in growth and stabilization of dendritic spines, and therefore in synaptic function. Critical for the stabilization of AMPA receptors at postsynaptic sites. Critical for the regulation of synaptic vesicle endocytosis at presynaptic terminals (By similarity). Required for the localization of NR1D1 to dendrites, can suppress its repressor activity and protect it from proteasomal degradation. {ECO:0000250, ECO:0000269|PubMed:21874017}. |
Cell projection;Cytoplasm;Endocytosis;GTPase activation;Neurogenesis;Reference proteome;Synapse |
|
|
mmu:94190; |
actin cytoskeleton [GO:0015629]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; glutamatergic synapse [GO:0098978]; presynapse [GO:0098793]; terminal bouton [GO:0043195]; actin binding [GO:0003779]; GTPase activator activity [GO:0005096]; ionotropic glutamate receptor binding [GO:0035255]; phospholipid binding [GO:0005543]; actin cytoskeleton organization [GO:0030036]; cell junction assembly [GO:0034329]; cell morphogenesis involved in neuron differentiation [GO:0048667]; cerebellar granule cell differentiation [GO:0021707]; cerebral cortex neuron differentiation [GO:0021895]; establishment of epithelial cell apical/basal polarity [GO:0045198]; maintenance of postsynaptic specialization structure [GO:0098880]; negative regulation of proteasomal protein catabolic process [GO:1901799]; neuron differentiation [GO:0030182]; neuron projection development [GO:0031175]; regulation of endocytosis [GO:0030100]; regulation of postsynaptic neurotransmitter receptor internalization [GO:0099149]; regulation of Rho protein signal transduction [GO:0035023]; regulation of synaptic transmission, glutamatergic [GO:0051966]; regulation of synaptic vesicle endocytosis [GO:1900242]; signal transduction [GO:0007165]; synaptic vesicle endocytosis [GO:0048488] |
12932438_Oligophrenin-1 is expressed in developing CNS areas that evince synaptic plasticity. OPHN1 is expressed in both glial and neuronal cells, where it colocalizes with actin at the tip of growing neurites. 17728457_This study indicates that cognitive impairment related to OPHN1 loss of function is associated with both presynaptic and postsynaptic alterations. 19401298_Loss of OPHN1 leads to the activation of the RhoA/ROCK signaling pathway. 19481455_Results sugest that reduced or defective OPHN1 signaling impairs synaptic vesicle cycling at hippocampal synapses. 21874017_The results of this study indicated the presence of a circadian oscillator in the hippocampus, involving the clock gene Bmal1 (also known as Arntl), that is modulated by Rev-erbalpha and requires oligophrenin-1 for normal oscillation. 22017989_our data establish a role for rapid OPHN1 synthesis in mGluR long-term depression. 22119667_Ophn1 is involved in processes of normal retinal vessel function during adulthood. 22298643_OPHN1 is a powerful regulator of Rho GTPase activity in platelets that is critical for the reorganization of the cytoskeleton, which is a major process required for stable platelet adhesion and thrombus formation to occur. 25556321_Identify OPHN1 as an important regulator of platelet cytoskeletal reorganization and thrombus formation. 26245966_OPHN1 is a bifunctional protein that is able, through distinct mechanisms, to regulate and most likely link exocytosis to compensatory endocytosis in chromaffin cells. 27117132_OPHN1 mediated regulation of RhoA, Rac1 and Cdc42 is crucial for the preservation of cardiac function after myocardial injury. 27146843_Here, we report that chronic treatment in adult mouse with Fasudil, is able to counteract vertical and horizontal hyperactivities, restores recognition memory and limits the brain ventricular dilatation observed in Ophn1(-)(/y) However, deficits in working and spatial memories are partially or not rescued by the treatment 27742778_Furthermore, we found that olfactory behaviour was perturbed in OPHN1 ko mice. Chronic treatment with a Rho kinase inhibitor rescued most of the defects of the newly generated neurons. Altogether, our data indicated that OPHN1 plays a key role in regulating the number, morphology and function of adult-born inhibitory interneurons and contributed to identify potential therapeutic targets. 29030432_Ophn1 deficiency generates severe impairments in performance at spatial working memory tests. 33376209_Altered Cl(-) homeostasis hinders forebrain GABAergic interneuron migration in a mouse model of intellectual disability. 33831348_Oligophrenin-1 moderates behavioral responses to stress by regulating parvalbumin interneuron activity in the medial prefrontal cortex. 34738630_Androgen deprivationinduced OPHN1 amplification promotes castrationresistant prostate cancer. 35563851_Hippocampal Excitatory Synaptic Transmission and Plasticity Are Differentially Altered during Postnatal Development by Loss of the X-Linked Intellectual Disability Protein Oligophrenin-1. |
ENSG00000079482 |
OPHN1 |
596.395731 |
0.4879270247 |
-1.035263 |
0.14459295 |
50.809111 |
0.00000000000101800072510948825787251756300375412278222397866045412229141220450401306152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000202960563414499026975157989074809942314780997207890322897583246231079101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
372.568889227294 |
37.4029624091555 |
763.575023232443 |
56.1659400347001 |
| ENSMUSG00000031216 |
236920 |
Stard8 |
protein_coding |
Q8K031 |
FUNCTION: Accelerates GTPase activity of RHOA and CDC42, but not RAC1. Stimulates the hydrolysis of phosphatidylinositol 4,5-bisphosphate by PLCD1 (By similarity). {ECO:0000250}. |
Cell junction;GTPase activation;Methylation;Phosphoprotein;Reference proteome |
|
|
mmu:236920; |
focal adhesion [GO:0005925]; GTPase activator activity [GO:0005096]; lipid binding [GO:0008289]; actin cytoskeleton organization [GO:0030036]; regulation of Rho protein signal transduction [GO:0035023]; signal transduction [GO:0007165] |
Human_homologues 19913121_Observational study of gene-disease association. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 25673874_DLC3 is recruited to Rab8-positive membrane tubules and is required for the integrity of the Rab8 and Golgi compartments. 27505894_a new function for Scribble in Rho regulation that entails positioning of DLC3 GAP activity at cell junctions in polarized epithelial cells, is reported. 31076513_The authors propose that DLC3 function is required to limit endosomal actin polymerization, Rab4-dependent recycling of MT1-MMP and, consequently, matrix degradation mediated by invadopodial activity. 36326091_A conserved function of Human DLC3 and Drosophila Cv-c in testis development. |
ENSG00000130052 |
STARD8 |
137.045260 |
5.3145038896 |
2.409935 |
0.28299376 |
70.013526 |
0.00000000000000005889920620928953928095245116918347602380353142262695009812034641072386875748634338378906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000017907239700197571318757633497543148173606285776526814146336619160138070583343505859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
243.207936854173 |
48.0133020688453 |
45.7630555749177 |
7.18704923088032 |
| ENSMUSG00000031239 |
16431 |
Itm2a |
protein_coding |
Q61500 |
|
Disulfide bond;Glycoprotein;Membrane;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:16431; |
Golgi apparatus [GO:0005794]; plasma membrane [GO:0005886]; amyloid-beta binding [GO:0001540]; negative regulation of amyloid precursor protein biosynthetic process [GO:0042985]; plasma cell differentiation [GO:0002317] |
15325075_Results support the involvement of Itm2a in the early stages of chondrogenesis in vitro. 17873332_Both Itm2a and Itm2c were not detected in the epididymis and vas deferens during sexual maturation. 23650549_Itm2a is not only expressed in adult muscle fibres but also in the satellite cells responsible for regeneration. 24831988_Itm2a is a target gene of GATA-3 that plays a minimal role in regulating the development and function of T cells 25079563_Itm2a possesses diverse functional properties as evidenced by differential expression during odontogenesis. 28872940_Itm2a knockdown is sufficient to rescue the inhibitory effects of lamin A WT and R482W mutant overexpression on 3T3-L1 differentiation. 34440772_Integral Membrane Protein 2A Is a Negative Regulator of Canonical and Non-Canonical Hedgehog Signalling. |
ENSG00000078596 |
ITM2A |
105.175708 |
0.2077062146 |
-2.267384 |
0.28129367 |
64.101353 |
0.00000000000000118180348557520025454999534259619779525872995066398951635733283183071762323379516601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000318042945093311839233227672949092586663494275356356411066371947526931762695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.3372653260053 |
7.32615617933197 |
179.759981635154 |
23.8377005432703 |
| ENSMUSG00000031250 |
64103 |
Tnmd |
protein_coding |
Q9EP64 |
FUNCTION: May be an angiogenesis inhibitor. |
Disulfide bond;Glycoprotein;Membrane;Nucleus;Phosphoprotein;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:64103; |
membrane [GO:0016020]; nuclear envelope [GO:0005635]; endothelial cell morphogenesis [GO:0001886]; negative regulation of angiogenesis [GO:0016525]; negative regulation of endothelial cell proliferation [GO:0001937] |
15632070_Results suggest that tenomodulin (Tnmd) is a regulator of tenocyte proliferation and is involved in collagen fibril maturation but do not confirm an in vivo involvement of Tnmd in angiogenesis. 18239943_The expression domains of ChM-I and TeM during vertebrate development incorporate the typical avascular regions of the mesenchymal tissues. 18838562_Tenomodulin is expressed abundantly in the elastin-rich subendothelial outer layer of the normal chordae tendineae cordis. 25107480_In our study of MM development, the laminar structure regulating TeM also prevents vascular invasion during the formation of compartment of the MM. 25351164_Loss of Tnmd affects significantly the self-renewal and senescence properties, but not the multipotential of tendon stem cells. 26880110_Upon high-fat diet feeding, transgenic mice overexpressing Tnmd develop increased epididymal white adipose tissue (eWAT) mass, and preadipocytes derived from Tnmd transgenic mice display greater proliferation, consistent with elevated adipogenesis. 27098985_The results of enhanced tenogenic differentiation and neotendon formation indicated that Tnmd may serve not only as a tenogenic marker but also as a positive regulator of MSCs tenogenic differentiation, which might be applied to MSCs-mediated tendon regeneration. 27777108_In vitro comparison of tenogenic differentiation potentials among bone marrow derived stem cells (BMSCs), adipose derived stem cells (ASCs), dermal fibroblasts (DFs) and tenocytes (TCs) of the same Tnmd transgenic mice. BMSCs exhibited the best tenogenic potential than other three cell types. 28566251_our study demonstrates that Tnmd is required for proper tendon tissue adaptation to endurance running and aids in better understanding of the structural-functional relationships of tendon tissues. 29022912_Tnmd is required for prevention of adipocyte accumulation and fibrovascular scar formation during early tendon healing. 29453333_Scleraxis is a transcriptional activator that regulates the expression of Tenomodulin, a marker of mature tenocytes and ligamentocytes 30922565_Tnmd-deficient mouse tendon stem/progenitor cells exhibit significantly reduced adhesion to collagen type I. 32083813_Loss of tenomodulin expression is a risk factor for age-related intervertebral disc degeneration. 34486420_Tenomodulin and Chondromodulin-1 Are Both Required to Maintain Biomechanical Function and Prevent Intervertebral Disc Degeneration. 34741033_Tenomodulin knockout mice exhibit worse late healing outcomes with augmented trauma-induced heterotopic ossification of Achilles tendon. |
ENSG00000000005 |
TNMD |
359.574326 |
0.2099417881 |
-2.251939 |
0.16035519 |
193.438585 |
0.00000000000000000000000000000000000000000005646724559060008445813785222832134397598411657494166665869596559930345961492340111103555213075827161753376487408548345392489409277914091944694519042968750000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000075448165336493015402382346519571248265974945529541210298629816368624805799303320179504875854987894003440265441873258112082112347707152366638183593750000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
123.86713378011 |
13.7961664021408 |
590.007043832698 |
45.154219590244 |
| ENSMUSG00000031286 |
320302 |
Glt28d2 |
protein_coding |
Q8BML3 |
|
Proteomics identification;Reference proteome;Transferase |
|
|
mmu:320302; |
N-acetylglucosaminyldiphosphodolichol N-acetylglucosaminyltransferase activity [GO:0004577]; dolichol-linked oligosaccharide biosynthetic process [GO:0006488] |
|
|
|
58.789475 |
0.2824068090 |
-1.824153 |
0.34559158 |
27.552201 |
0.00000015291123536522238640696243726357916870028930134139955043792724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000015642336716474595447015681973224054956972395302727818489074707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.847846984823 |
6.00928666131387 |
87.9860052573097 |
14.6714729654995 |
| ENSMUSG00000031290 |
210297 |
Lrch2 |
protein_coding |
Q3UMG5 |
FUNCTION: May play a role in the organization of the cytoskeleton. {ECO:0000250|UniProtKB:Q960C5, ECO:0000250|UniProtKB:Q96II8}. |
Alternative splicing;Leucine-rich repeat;Reference proteome;Repeat |
|
|
mmu:210297; |
|
|
ENSG00000130224 |
LRCH2 |
22.697767 |
0.0040149437 |
-7.960405 |
1.32698172 |
50.431719 |
0.00000000000123383786179874057035724310484550199232614553501719001360470429062843322753906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000243128309326240813387826846781559564716901356007383583346381783485412597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.194927369220744 |
0.205665072725799 |
48.5504610555163 |
16.6363899361965 |
| ENSMUSG00000031298 |
237175 |
Adgrg2 |
protein_coding |
Q8CJ12 |
FUNCTION: Orphan receptor. Could be involved in a signal transduction pathway controlling epididymal function and male fertility. May regulate fluid exchange within epididymis. {ECO:0000269|PubMed:15367682}. |
3D-structure;Alternative splicing;Cell membrane;G-protein coupled receptor;Glycoprotein;Membrane;Phosphoprotein;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:237175; |
apical plasma membrane [GO:0016324]; cytosol [GO:0005829]; plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; cell surface receptor signaling pathway [GO:0007166] |
17034053_The knockout of the HE6/Gpr64 receptor was mainly associated with the downregulation of genes specific to the initial segment. 18625716_Affymetrix expression profiling shows that the C terminus of Runx2 regulates genes involved in G protein-coupled receptor signaling Rgs2, Rgs4, Rgs5, Rgs16, Gpr23, Gpr30, Gpr54, Gpr64, and Gna13. 23338946_GPR64 expression in ES maintains an immature phenotype that is less sensitive to TRAIL-induced apoptosis and via its up-regulation of PGF and MMP1 orchestrates and promotes invasiveness and metastatic spread in Ewing sarcoma. 26188515_examined the G protein-coupling abilities of GPR64 and showed that it is activated through a tethered agonist sequence, which we have previously identified as the Stachel sequence 28694502_Study identified Gpr64 as a P4-PGR target gene in the mouse uterus. P4 induced GPR64 expression in the epithelial and stromal cells of the uterus in ovariectomized wild-type mice, but not in PRKO mice. ChIP analysis confirmed that PGR proteins were recruited on progesterone response element of Gpr64 gene in the uteri of wild-type mice treated with P4. 29393851_The authors demonstrated that the ubiquitous G protein-coupled receptor signaling proteins Gq and beta-arrestin-1 are essential for fluid reabsorption because they mediate coupling between an orphan receptor ADGRG2 (GPR64) and the ion channel CFTR. 33303626_Optimization of a peptide ligand for the adhesion GPCR ADGRG2 provides a potent tool to explore receptor biology. 33831593_Adhesion receptor ADGRG2/GPR64 is in the GI-tract selectively expressed in mature intestinal tuft cells. |
ENSG00000173698 |
ADGRG2 |
22.903694 |
0.2324689444 |
-2.104890 |
0.72247005 |
8.087857 |
0.00445628229698194142094358483063842868432402610778808593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0168532450558101926096910005981044378131628036499023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.49312177536292 |
3.38923322089546 |
27.9311363209578 |
10.2133714218974 |
| ENSMUSG00000031304 |
16186 |
Il2rg |
protein_coding |
P34902 |
FUNCTION: Common subunit for the receptors for a variety of interleukins (PubMed:7718508). Probably in association with IL15RA, involved in the stimulation of neutrophil phagocytosis by IL15 (By similarity). {ECO:0000250|UniProtKB:P31785, ECO:0000269|PubMed:7718508}. |
3D-structure;Cell membrane;Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a transmembrane protein that is a common subunit of several interleukin receptor complexes. These receptors are comprised of alpha and beta subunits in addition to this gamma subunit. Signalling through this pathway in important in immune cell differentiation and function. Alternative splicing results in multiple transcript variants. [provided by RefSeq, May 2015]. |
mmu:16186; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; cytokine binding [GO:0019955]; cytokine receptor activity [GO:0004896]; interleukin-15 receptor activity [GO:0042010]; interleukin-2 binding [GO:0019976]; B cell differentiation [GO:0030183]; CD4-positive, CD25-positive, alpha-beta regulatory T cell differentiation [GO:0002361]; cytokine-mediated signaling pathway [GO:0019221]; gene expression [GO:0010467]; interleukin-15-mediated signaling pathway [GO:0035723]; lymphocyte differentiation [GO:0030098]; mature B cell differentiation [GO:0002335]; positive regulation of B cell differentiation [GO:0045579]; positive regulation of CD4-positive, CD25-positive, alpha-beta regulatory T cell differentiation [GO:0032831]; positive regulation of gene expression [GO:0010628]; positive regulation of phagocytosis [GO:0050766]; positive regulation of T cell differentiation in thymus [GO:0033089]; regulation of gene expression [GO:0010468]; T cell differentiation in thymus [GO:0033077] |
11788580_These data suggest that IRS-dependent signaling pathways work by recruiting different signaling molecules to determine specificity of IL-2R gamma superfamily cytokines. 11815609_Structural basis for binding multiple ligands by the common cytokine receptor gamma-chain 12069389_The estrous cycles in gamma(c) knockout (gamma(c) KO) mice were irregular compared with wild-type mice, although, the mutants could become pregnant. 14500690_Effector mechanisms in nonobese diabetic (NOD) hosts are inherently complex, and islet allograft rejection in this model involves common gamma-chain-dependent and -independent mechanisms. 15661875_Although IL-2 has been reported to influence natural killer (NK) cell differentiation, mice deficient in IL-2 common gamma chain receptor subunit have normal NK cell numbers, phenotype, and effector functions. 15825075_Colonic IL-6-producing thymus-derived CD4 + T cells are responsible for the development of colitis in CR gamma -/Y mice. 16418296_Stat5 has a role in severe combined immunodeficiency, similar in many respects to the roles of IL-7R, gammac, and Jak3 18304352_Different gamma chain cytokines regulate regulatory T cell homeostasis in the periphery by differentially regulating survival and proliferation. 18390713_naive T cells can be induced to undergo homeostatic proliferation of variable speed with a few members of the common gamma-chain (CD132) family of cytokines, the speed of proliferation depending on the levels of the particular cytokine involved. 19155493_Evidence supports complementary and essential roles for IL2Rg and FMS-like tyrosine kinase 3 ligand Flt3l in regulation of developing thymic and bone marrow natural killer (NK) cell compartments. 19802382_dengue virus infection and virus-specific HLA-A2 restricted immune response does not require IL2rgamma 19843944_High expression of the cytokine receptor common gamma-chain confers longevity of intestinal eosinophils and suggests that common gamma-chain-dependent tissue- intrinsic signals contribute to eosinophil homeostasis. 20438766_IL2Rgamma KO mice showed more depressive-like behavior than controls in both genders, and male mice showed improved contextual fear memory. 20439728_gamma c deficiency precludes CD8+ T cell memory despite formation of potent T cell effectors. 21155807_IL2Rgamma and its co-receptor IL15Ralpha are localized along with interleukin-15 in cerebral endothelia during robust upregulation of neuroinflammation. 21512274_Results reflect the suppression of the leaky phenomenon in NOG mice through the inactivation of the IL-2Rgamma gene, which is commonly expressed in T and B cell growth factor receptors to IL-2, IL-4 and IL-7. 21559522_CD34+ CD133+ hematopoietic stem cells cultured with growth factors including Angptl5 efficiently engraft adult NOD-SCID Il2rgamma-/- (NSG) mice 21585397_IL-2 receptor complex has an essential protective role in immunity to blood-stage malaria. Protection is achieved by gammac cytokine family members signalling through the IL-2Rgammac. 21611197_Hematopoietic stem cells -infused HLA-DR4.NOD.Rag1knockout (KO) .IL-2RgammacKO mice developed a very high reconstitution rate (>90%) with long-lived and functional human T and B cells. 21926555_Letter: Hyperglycemia-induced proliferation of adult human beta cells engrafted into spontaneously diabetic immunodeficient NOD-Rag1null IL2rgammanull Ins2Akita mice. 22306295_physiologic impact of CB hematopoietic progenitor cell hypofucosylation was investigated in vivo in NOD-SCID interleukin (IL)-2Rgamma(null) (NSG) mice 22569301_critical for intestinal T-cell reconstitution in humanized mice 22645657_Interleukin-2 signalling is modulated by a labile disulfide bond in the CD132 chain of its receptor. 22737248_analysis of multiorgan metastasis of human HER-2+ breast cancer in Rag2-/-;Il2rg-/- mice and treatment with PI3K inhibitor 22829596_Results indicated that the extracellular and transmembrane domains of gammaC and IL-13Ralpha1 controlled the magnitude of the intracellular signaling response to IL-4 and IL-13. 23109710_gammac expression was required to signal the differentiation of MHC class I-specific thymocytes into CD8+ cytotoxic lineage T cells and into invariant natural killer T cells. 23940740_The potential for dysregulated T cell and dendritic cell (DC) activation in the gamma(c)-deficient environment leading to allergic lung inflammation. 24368085_findings demonstrate that NOD SCID IL2R common gamma chain -/- mice are capable of engraftment of canine PBLs yet develop graft versus host disease similar to Hu-PBL-SCID mice 24798995_Human colon cancer xenografts propagated in NSG and NRG mice maintain structural fidelity while replacing human stromal cells with murine stromal cells. 25109334_Results demonstrate that IL2Rgamma has an onogenic role in JAK3-mutation-positive leukemias. 26113085_We have confirmed the activation of human peripheral blood lymphocytes after being xenografted in immunodeficient Rag2-/-IL2Rgnull mice. 28089909_degree to which immune rejection contributes to these disappointing outcomes using an immunodeficient IL2 receptor gamma (IL2rgamma)-null mouse model, was examined. 28111009_Renal lesions are indeed modulated by intrinsic glomerular cells through the gamma-C/IL-2-beta receptor response, to date classically described only in immune cells. 28331303_Suggest that soluble common gamma chain may be one of the target molecules for the control of immunopathogenic progresses in COPD. 28678791_Results indicate that conjugated-interleukin-2 (IL2) protein derived from NKTR-214 occupy interleukin-2 (IL2) receptor complex (IL2Ralphabetagamma) to a greater extent compared to free-IL2 protein. 28963568_IL2RG mutations cause X-linked severe combined immunodeficiency in humans and mice that can be corrected by gene editing. 30154103_Data indicate a role for ROR-gamma (RORgammat) and Bcl-XL protein (Bcl-xL) in limiting interleukin 2 receptor gamma chain (gammac) abundance at the cell surface. 30800133_Homodimeric sgammac has pathogenic roles to exacerbate rheumatoid arthritis progression with differentiation of local Th17, NKT1, and NKT17 cells. Therefore, sgammac is suggested as target of a therapeutic strategy for rheumatoid arthritis. 33253759_Improved engraftment of human peripheral blood mononuclear cells in NOG MHC double knockout mice generated using CRISPR/Cas9. 34971407_Protein abundance of the cytokine receptor gammac controls the thymic generation of innate-like T cells. |
ENSG00000147168 |
IL2RG |
73.428547 |
0.4437048832 |
-1.172328 |
0.32805867 |
12.698626 |
0.00036592429710337754647161978560632178414380177855491638183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0018958363583260703401356384034670554683543741703033447265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.0077425829028 |
9.6347981032739 |
94.6749611605973 |
15.6733644117939 |
| ENSMUSG00000031340 |
14404 |
Gabre |
protein_coding |
A2AMW3 |
Human_homologues FUNCTION: GABA, the major inhibitory neurotransmitter in the vertebrate brain, mediates neuronal inhibition by binding to the GABA/benzodiazepine receptor and opening an integral chloride channel. |
Cell membrane;Chloride;Chloride channel;Disulfide bond;Glycoprotein;Ion channel;Ion transport;Membrane;Postsynaptic cell membrane;Reference proteome;Signal;Synapse;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:14404; |
chloride channel complex [GO:0034707]; dendrite membrane [GO:0032590]; GABA-A receptor complex [GO:1902711]; GABA-ergic synapse [GO:0098982]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; postsynapse [GO:0098794]; postsynaptic membrane [GO:0045211]; synapse [GO:0045202]; excitatory extracellular ligand-gated ion channel activity [GO:0005231]; GABA-A receptor activity [GO:0004890]; GABA-gated chloride ion channel activity [GO:0022851]; inhibitory extracellular ligand-gated ion channel activity [GO:0005237]; neurotransmitter receptor activity [GO:0030594]; transmitter-gated ion channel activity involved in regulation of postsynaptic membrane potential [GO:1904315]; chemical synaptic transmission [GO:0007268]; chloride transmembrane transport [GO:1902476]; chloride transport [GO:0006821]; gamma-aminobutyric acid signaling pathway [GO:0007214]; ion transmembrane transport [GO:0034220]; negative regulation of chloride transport [GO:2001226]; nervous system process [GO:0050877]; regulation of membrane potential [GO:0042391]; regulation of postsynaptic membrane potential [GO:0060078]; signal transduction [GO:0007165]; synaptic transmission, GABAergic [GO:0051932] |
Human_homologues 15755675_Analysis of epilepsy inter-patient variability of eleven highly expressed GABAAR subunit mRNAs found seven of the subunits varied between patients, as did whole cell GABAAR currents. 15946973_We report here that the GABA-activated current can be potentiated at pH 8.4 for both alphabeta and alphabeta gamma subunit-containing receptors, but only at GABA concentrations below the EC40. 17293408_We infer that activation by anesthetics and GABA induces a similar conformational change at the M2 segment 6' position that is related to channel opening. 17615250_Increased intracortical excitability is noted in subjects affected by the GABA A receptor gamma 2 subunit (GABRG2 Arg43Glu) mutation; findings are likely to represent an important clue to mechanisms linking this gene defect and the epilepsy phenotype. 17714454_Increased expression of epsilon, in conjunction with alpha2 and beta3 subunits, results in expression of GABAA receptors with correspondingly altered rectification, deactivation and levels of spontaneous openings, but not increased total current density. 19078961_Observational study of gene-disease association. (HuGE Navigator) 19086053_Observational study of gene-disease association. (HuGE Navigator) 19087248_Although GABRE genes did not show positive association, further studies are necessary to consider the role of other GABA receptor genes in migraine susceptibility 19087248_Observational study of gene-disease association. (HuGE Navigator) 19874574_Observational study of gene-disease association. (HuGE Navigator) 20468064_Observational study of gene-disease association. (HuGE Navigator) 20609421_Using published alpha subunit data we identified two principal components labeled 'Decreasing' (alpha2, alpha5, beta1, gamma1 and gamma3) and 'Dynamic' (alpha1, alpha4, beta2 and gamma2) responsible for 84% of variation in GABA(A)R subunit development. 21422964_single nucleotide polymorphisms studied in the GABRA4, GABRE, and GABRQ genes are not related to the risk for familial ET. 24022508_GABRE protein levels were significantly increased in schizophrenia, major depressive disorder, and bipolar disorder patients' lateral cerebellum compared to controls. 24737792_Data indicate that GABA(A) receptor subunit epsilon (GABRE) approximately miR-452 approximately miR-224 locus is downregulated and hypermethylated in prostate cancer and is a promising epigenetic candidate biomarker for prostate cancer diagnosis and prognosis. 25660468_This study showed that in male groups, the expression of GABRE was generally lower in schizophrenia cases compared to the control. 29299688_data from the present study suggest a marginal role of GABRE rs1139916 in the risk for migraine in women. 31719049_Comprehensive Transcriptome Profiling of Cryptic CBFA2T3-GLIS2 Fusion-Positive AML Defines Novel Therapeutic Options: A COG and TARGET Pediatric AML Study. 32588540_Rare variants in the GABAA receptor subunit epsilon identified in patients with a wide spectrum of epileptic phenotypes. |
ENSG00000102287 |
GABRE |
53.301275 |
28.3092785480 |
4.823203 |
0.60324942 |
71.561975 |
0.00000000000000002686849248702653200283837517053817100236290213346578709119327754706318955868482589721679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000008526268282549752781147538907189748066222298168541360308125831579673103988170623779296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.252569179079 |
43.1519468801833 |
3.54133253551609 |
1.36012447616723 |
| ENSMUSG00000031351 |
22673 |
Zfp185 |
protein_coding |
B1AXE7 |
Human_homologues FUNCTION: May be involved in the regulation of cellular proliferation and/or differentiation. |
Proteomics identification;Reference proteome |
|
|
|
|
18239999_These data offered evidence for the association of mouse Zfp185 with F-actin, which supports the potential role of Zfp185 in cell fundamental activity. 20937232_ZNF185 is the most abundant in the testes and is located in Leydig cells and sperm. Leydig cells has weak expression and in round spermatids and mature sperm it is highly expressed. 28627666_ZNF185 was highly expressed in Leydig cells of the testis and involved in the secretion of testosterone and the knockdown of ZNF185 expression did not significantly affect cell cycle progression or apoptosis. 36631535_ZNF185 prevents stress fiber formation through the inhibition of RhoA in endothelial cells. |
ENSG00000147394 |
ZNF185 |
116.612682 |
2.5261416925 |
1.336936 |
0.25556249 |
27.143435 |
0.00000018890560387541339506513001552517883041559798584785312414169311523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000018942792351746522348213304376418619767719064839184284210205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
164.310239867992 |
29.2587762679602 |
65.0439523460981 |
9.017667511703 |
| ENSMUSG00000031355 |
11856 |
Arhgap6 |
protein_coding |
O54834 |
FUNCTION: GTPase activator for the Rho-type GTPases by converting them to an inactive GDP-bound state. Could regulate the interactions of signaling molecules with the actin cytoskeleton. Promotes continuous elongation of cytoplasmic processes during cell motility and simultaneous retraction of the cell body changing the cell morphology (By similarity). {ECO:0000250}. |
Alternative splicing;Cytoplasm;GTPase activation;Phosphoprotein;Reference proteome;SH3-binding |
|
|
mmu:11856; |
actin cytoskeleton [GO:0015629]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; GTPase activator activity [GO:0005096]; phospholipase activator activity [GO:0016004]; phospholipase binding [GO:0043274]; SH3 domain binding [GO:0017124]; actin filament organization [GO:0007015]; activation of phospholipase C activity [GO:0007202]; focal adhesion assembly [GO:0048041]; negative regulation of focal adhesion assembly [GO:0051895]; negative regulation of stress fiber assembly [GO:0051497]; positive regulation of GTPase activity [GO:0043547]; positive regulation of phospholipase activity [GO:0010518]; regulation of GTPase activity [GO:0043087]; signal transduction [GO:0007165] |
11668396_Analysis of Mid1, Hccs, Arhgap6, and Msl3l1 in X-linked polydactyly (Xpl) and Patchy-fur (Paf) mutant mice |
ENSG00000047648 |
ARHGAP6 |
182.179730 |
2.6559777358 |
1.409243 |
0.48682234 |
7.623059 |
0.00576266523447900060050930193256135680712759494781494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0210042873538129060140278170365490950644016265869140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
186.85410858212 |
64.2553667110399 |
70.3522872448178 |
18.1371613998404 |
| ENSMUSG00000031442 |
17207 |
Mcf2l |
protein_coding |
D3Z3S7 |
Human_homologues FUNCTION: Guanine nucleotide exchange factor that catalyzes guanine nucleotide exchange on RHOA and CDC42, and thereby contributes to the regulation of RHOA and CDC42 signaling pathways (By similarity). Seems to lack activity with RAC1. Becomes activated and highly tumorigenic by truncation of the N-terminus (By similarity). Isoform 5 activates CDC42 (PubMed:15157669). {ECO:0000250|UniProtKB:Q63406, ECO:0000269|PubMed:15157669}.; FUNCTION: [Isoform 3]: Does not catalyze guanine nucleotide exchange on CDC42 (PubMed:15157669). {ECO:0000269|PubMed:15157669}. |
Proteomics identification;Reference proteome |
|
|
|
|
12637522_the PH domain of Dbs has a role in regulating Rho GTPase activation 12637530_the PH domain of Dbs has two roles in the regulation of DH domain function, one for GTPase association and activation in vitro and one for phosphoinositide binding and GTPase interaction in vivo, that together promote Dbs association with membranes. 14701795_Dbs is activated by Rac1 at the pleckstrin homology domain 14991610_Dbs has the potential to promote proliferation of immature thymocytes, but also sensitizes immature thymocytes to deletion. 15274927_Crystal structure of the DH/PH fragment of Dbs without bound GTPase 15531584_the Sec14 domain regulates Dbs transformation through at least two distinct mechanisms, neither of which appears to directly influence the in vivo exchange activity of the protein 16613852_Dbs transformation is associated with increased phosphorylation of myosin light chain and stress fiber formation, both of which occur in a ROCK-dependent manner 31732956_miR-140-3p exhibits repressive functions on preosteoblast viability and differentiation by downregulating MCF2L in osteoporosis. |
ENSG00000126217 |
MCF2L |
147.253426 |
2.3281011881 |
1.219154 |
0.27390593 |
19.561838 |
0.00000973953422969905720110814911771868196410650853067636489868164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000724024330828580031009858442025972635747166350483894348144531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
194.353424462146 |
37.1214594208007 |
83.4815193835159 |
12.0985124516849 |
| ENSMUSG00000031461 |
17930 |
Myom2 |
protein_coding |
Q14BI5 |
Human_homologues FUNCTION: Major component of the vertebrate myofibrillar M band. Binds myosin, titin, and light meromyosin. This binding is dose dependent. |
Coiled coil;Immunoglobulin domain;Proteomics identification;Reference proteome;Repeat |
|
|
mmu:17930; |
cytoskeleton [GO:0005856]; M band [GO:0031430]; actin filament binding [GO:0051015]; kinase binding [GO:0019900]; structural constituent of cytoskeleton [GO:0005200]; muscle contraction [GO:0006936]; sarcomere organization [GO:0045214] |
Human_homologues 12972258_Results identify muscle-type creatine kinase as a binding partner of a central portion of myomesin and the closely related M-protein. 23792176_a direct interaction of dysferlin with Trim72/MG53, AHNAK, cytoplasmic dynein, myomesin-2 and calsequestrin-1, but not with caveolin-3 or dystrophin, is reported. 33033063_Identification of MYOM2 as a candidate gene in hypertrophic cardiomyopathy and Tetralogy of Fallot, and its functional evaluation in the Drosophila heart. |
ENSG00000036448 |
MYOM2 |
37.368883 |
24.1520140253 |
4.594072 |
0.72757893 |
37.048797 |
0.00000000115209613770899709909971425506394759885608891636366024613380432128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000161293459279259593873959995708952663839852448290912434458732604980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.7838664031003 |
27.7627682355516 |
2.18548508409303 |
0.971636476942128 |
| ENSMUSG00000031558 |
20563 |
Slit2 |
protein_coding |
Q9R1B9 |
FUNCTION: Thought to act as molecular guidance cue in cellular migration, and function appears to be mediated by interaction with roundabout homolog receptors. During neural development involved in axonal navigation at the ventral midline of the neural tube and projection of axons to different regions. SLIT1 and SLIT2 seem to be essential for midline guidance in the forebrain by acting as repulsive signal preventing inappropriate midline crossing by axons projecting from the olfactory bulb. In spinal cord development, may play a role in guiding commissural axons once they reached the floor plate by modulating the response to netrin. In vitro, silences the attractive effect of NTN1 but not its growth-stimulatory effect and silencing requires the formation of a ROBO1-DCC complex. May be implicated in spinal cord midline post-crossing axon repulsion. In vitro, only commissural axons that crossed the midline responded to SLIT2. In the developing visual system, appears to function as repellent for retinal ganglion axons by providing a repulsion that directs these axons along their appropriate paths prior to, and after passage through, the optic chiasm. In vitro, collapses and repels retinal ganglion cell growth cones. Seems to play a role in branching and arborization of CNS sensory axons, and in neuronal cell migration. In vitro, Slit homolog 2 protein N-product, but not Slit homolog 2 protein C-product, repels olfactory bulb (OB) but not dorsal root ganglia (DRG) axons, induces OB growth cones collapse and induces branching of DRG axons. Seems to be involved in regulating leukocyte migration (By similarity). {ECO:0000250, ECO:0000269|PubMed:11804571, ECO:0000269|PubMed:12097499}. |
Chemotaxis;Developmental protein;Differentiation;Disulfide bond;EGF-like domain;Glycoprotein;Heparin-binding;Leucine-rich repeat;Neurogenesis;Reference proteome;Repeat;Secreted;Signal |
|
The protein encoded by this gene is a member of the Slit family of secreted glycoproteins, which function as ligands for the Robo family of immunoglobulin receptors. Slit proteins play highly conserved roles in axon guidance and neuronal migration and may also have functions during other cell migration processes including leukocyte migration. In mammals, members of the slit family are characterized by an N-terminal signal peptide, four leucine-rich repeats, nine epidermal growth factor repeats, and a C-terminal cysteine knot. Mice deficient for this gene exhibit abnormal axonal projections in the embryonic forebrain and develop supernumerary uretic buds that maintain improper connections to the nephric duct. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Sep 2015]. |
mmu:20563; |
cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; cytoplasm [GO:0005737]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; chemorepellent activity [GO:0045499]; GTPase inhibitor activity [GO:0005095]; heparan sulfate proteoglycan binding [GO:0043395]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; laminin-1 binding [GO:0043237]; protein homodimerization activity [GO:0042803]; proteoglycan binding [GO:0043394]; Roundabout binding [GO:0048495]; signaling receptor binding [GO:0005102]; aortic valve morphogenesis [GO:0003180]; axon extension involved in axon guidance [GO:0048846]; axon guidance [GO:0007411]; axonogenesis [GO:0007409]; branching morphogenesis of an epithelial tube [GO:0048754]; cell migration involved in sprouting angiogenesis [GO:0002042]; cell-cell adhesion [GO:0098609]; cellular response to heparin [GO:0071504]; chemorepulsion involved in postnatal olfactory bulb interneuron migration [GO:0021836]; corticospinal neuron axon guidance through spinal cord [GO:0021972]; dorsal/ventral axon guidance [GO:0033563]; in utero embryonic development [GO:0001701]; induction of negative chemotaxis [GO:0050929]; kidney development [GO:0001822]; mammary duct terminal end bud growth [GO:0060763]; mammary gland duct morphogenesis [GO:0060603]; metanephros development [GO:0001656]; motor neuron axon guidance [GO:0008045]; negative chemotaxis [GO:0050919]; negative regulation of actin filament polymerization [GO:0030837]; negative regulation of axon extension [GO:0030517]; negative regulation of cell growth [GO:0030308]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cellular response to growth factor stimulus [GO:0090288]; negative regulation of chemokine-mediated signaling pathway [GO:0070100]; negative regulation of endothelial cell migration [GO:0010596]; negative regulation of gene expression [GO:0010629]; negative regulation of inflammatory response [GO:0050728]; negative regulation of lamellipodium assembly [GO:0010593]; negative regulation of leukocyte chemotaxis [GO:0002689]; negative regulation of monocyte chemotaxis [GO:0090027]; negative regulation of mononuclear cell migration [GO:0071676]; negative regulation of neutrophil chemotaxis [GO:0090024]; negative regulation of protein phosphorylation [GO:0001933]; negative regulation of retinal ganglion cell axon guidance [GO:0090260]; negative regulation of small GTPase mediated signal transduction [GO:0051058]; negative regulation of smooth muscle cell chemotaxis [GO:0071672]; negative regulation of smooth muscle cell migration [GO:0014912]; negative regulation of vascular permeability [GO:0043116]; neuron projection morphogenesis [GO:0048812]; olfactory bulb development [GO:0021772]; positive regulation of apoptotic process [GO:0043065]; pulmonary valve morphogenesis [GO:0003184]; retinal ganglion cell axon guidance [GO:0031290]; Roundabout signaling pathway [GO:0035385]; telencephalon cell migration [GO:0022029]; ureteric bud development [GO:0001657]; ventricular septum morphogenesis [GO:0060412] |
12051827_role in antagonizing netrin-1 attractive effects during migration of inferior olivary neurons 12097499_Slit proteins control the development of the lateral olfactory tract 12141424_Analysis of alternative splicing and conserved domains in human and mouse slit genes 12488830_N-Slit2 promoted neuronal survival and neurite extension in chick dorsal root and autonomic ganglia. Slit2 also promoted neuronal survival in mouse dorsal root ganglia. 12655597_Slit2 mRNA was intensely expressed in GFAP-positive astrocytes surrounding necrotic brain tissue, suggesting that slit2 might prevent regenerating axons from entering into the lesion. 12954717_Data suggest that the Slit family of axon guidance molecules (Slit 1-3) and their Robo 1 and 2 receptors contribute to the topographic targeting of basal vomeronasal axons. 12954881_The principle function of Slit2 at the cortical midline may be to channel the axons along the correct path and possibly repel them away from the midline. We find no evidence that Slit2 prevents axons from recrossing the midline in the brain. 14662852_Slit2 inhibits the migration of Langerhans cells out of the skin, suppressing the development of contact hypersensitivity responses after hapten sensitization. 14960623_The Slit receptors Robo2 and Robo3/Rig-1 are expressed in the subventricular zone and the rostral migratory stream and Slit1 and Slit2 are present in the adult septum and are responsible for both the septum and the CP repulsive activity in vitro 15091338_Experiments with triple mutant mice demonstrate a key role for Slit2 signaling in vertebrate midline commissural axon guidance. 15130495_Mutants lacking SLIT2 develop supernumerary ureteric buds that remain inappropriately connected to the nephric duct; the SLIT2 signal is transduced in the nephrogenic mesenchyme. 15162513_Slit-2 and Robo-1 expression is present throughout mesenchyme at midgestation and is not detectable by newborn day 1 16439476_SLIT2 acts in parallel with NTN1 to generate cell boundaries along ducts during bi-layered tube formation. 16828733_Slit2 helps prevent retinal ganglion cell axons from crossing the diencephalic/telencephalic boundary in the developing optic tract. 17496152_Slit2 mRNA level decreased during development of vascular decline in pulmonary fibrosis. 18054781_The role of Slit2-Robo signaling in gthe generation, migration and morphological differentiation of cortical interneurons is reported. 18417704_Slit1 and Slit2 cooperate with Robo1 and Robo2 to control the convergence ofolfactory receptor neuron (ORN) axons to the olfactory bulb and the precise targeting of ORN axons to specific glomeruli. 18842816_Slit1;Slit2 double mutants had severe longitudinal errors, particularly for ventral axons, including midline crossing and wandering longitudinal trajectories 19033678_Slit2 plays a role in regulating in vitro osteoblast differentiation. 19048120_Slit2 inhibits tumor growth and metastasis of fibrosarcoma and squamous cell carcinoma and its effect on cell cycle and apoptosis signal pathways is an important mechanism for Slit2-mediated tumor suppression 19115401_These findings suggest that the combination of Netrin and Slit may be involved in proper axonal projection from the mammillary bodies and that their misexpression in the diencephalon may cause the misrouting of these axons in Pax6 mutant mice. 19759280_Slit2 potently inhibits chemotaxis but not random motion of circulating neutrophils 19782674_results suggest strongly that Robo2 redundancy with other guidance signals rather than different receptor utilisation is the most likely explanation for the regional specificity of Slit function during intraretinal axon pathfinding 20375003_Activating with the soluble ligand Slit an endothelium-specific, Robo4-dependent signaling pathway that strengthens the vascular barrier, diminishing deleterious aspects of the host's response to the pathogen-induced cytokine storm. 20944010_the newly identified Slit2 gradient at the bronchus-alveoli axis induces attractive PI3K signaling in eosinophils and repulsive srGAP1 signaling in neutrophils through differential srGAP1 expression during lung inflammation. 20947829_Slit2 expression (and that of Slit3) are linked to posttranscriptional regulation of Robo receptors during blood vessel development. 21118670_These results indicate that Slit1/2 - Robo1/2 signaling is critical during the initial establishment of dopaminergic pathways, with roles in the dorsoventral positioning and precise pathfinding of these ascending longitudinal axons. 21135133_Cooperation between Slit2 and ephrin-A1 regulates a balance between the pro- and antiangiogenic functions of Slit2. 21435555_the midline repellent Slit2 orients migration of corridor neurons and thereby switches thalamic axons from an external to a mammalian-specific internal path. 21986575_Over-expression of human Slit2 in transgenic mouse significantly increased brain vessel density and the permeability of brain vessels to large molecules. 22306607_The results of this study supported the idea that Slit2 promotes axon fasciculation via an autocrine and/or juxtaparacrine mechanism. 22313734_Data found that expression of Slit2 and SDF1 were dynamically regulated in mesenchymal stem cells and OBC-like populations following radiation, while Robo4 expression was restricted to endothelial cells. 22399681_Nkx2.9 controls SACMN axon exit from the mammalian spinal cord by regulating Robo-Slit signaling 22865890_The cell motility modulator Slit2 is a potent inhibitor of platelet function. 22912413_Data show that the gooseberry/gooseberry-neuro (gsb/gsb-n) transcription factor genes act to specify MP3 cell fate. 23083737_This study report that central nervous system progenitors express Robo1 and Robo2, receptors for Slit proteins that regulate axon guidance, and that absence of these receptors or their ligands leads to loss of ventricular mitoses. 23320558_This study demonistrated that Slit2 can act alone to control aspects of retinal axon routing across the ventral diencephalic midline. 23438940_Slit/Robo1 signaling is involved in regulating neural tube development by tightly coordinating cell proliferation and differentiation during neurulation. 23811810_SDF-1-CXCR4 differentially regulates autoimmune diabetogenic T cell adhesion through ROBO1-SLIT2 interactions in mice. 23814012_Prostaglandin F2alpha upregulates Slit2 and Robo1 expression in mouse corpus luteum during luteolysis. 23884932_Slit/Robo signaling imposes a restriction force on spiral ganglia neurons to ensure their precise positioning for correct spiral ganglia-cochlear hair cells innervations. 24065825_Slit production is required for beta-cell survival and optimal function via a mechanism involving endoplasmic reticulum Ca2+ homeostasis and actin remodeling. 24840330_Results suggest that Slit2 might be involved in skin tumorigenesis. 25366972_Robo1-Slit2 interaction required for pathfinding mechanism essential to establish the functionally important habenulo-interpeduncular connection. 25490006_Results provide novel evidence that low expression of SLIT2 correlates with poor prognosis and promotes metastasis in ESCC, which may be regulated by the Cdc42-mediated pathways. 25490618_These observations from mutant Slit and Robo mice give great support for Slit/Robo role in neural crest cell migration during Enteric Nervous System development. 25504371_Data shows that modulation of angiostatic factor Slit2 by EphA2 receptor regulates endothelial responses to VEGF-mediated angiogenesis and tumor neovascularization. 25605242_Slit2/Robo1 signaling promotes intestinal tumorigenesis through Src-mediated activation of the Wnt/beta-catenin pathway. 25691540_Cardiac defects in mutants for Robo or Slit range from membranous ventricular septum defects to bicuspid aortic valves. 25894826_Slit2 signaling through Robo1 and Robo2 has a role in retinal neovascularization 25904499_Slit2 acts as a repellant cue to mediate axon guidance in the formation of the anterior commissure. 26026792_Mutations of the SLIT2-ROBO2 pathway genes SLIT2 and SRGAP1 confer risk for congenital anomalies of the kidney and urinary tract 26047956_the two genes neuropeptide Y (Npy) and Slit homolog 2 (Drosophila) (Slit2) gradually increase during aging, and upon suppression of these two genes 26159615_Together, these observations suggest that Slit2 serves as a factor utilized by muscle Ctnnb1 to direct presynaptic differentiation. 26244297_Slit2 may promote angiogenesis by upregulating Robo1 and activating the VEGFR2-ERK1/2 pathway. 26282852_overexpression promoted vascular remodeling by increasing the diameter of the maternal blood sinusoids and fetal capillaries 26297762_administration of Slit2 to atherosclerosis-prone LDL receptor-deficient mice inhibited monocyte recruitment to nascent atherosclerotic lesions. 26876562_Slit2 induces a robust activation of PKA signaling, which is required for its prothermogenic activity. 27503415_Study provides evidence that Slit2 is a novel quantitative trait gene and a positive regulator of the number and function of murine hematopoietic stem cells. 27521753_Transgenic over-expression of slit2 enhances disruption of blood-brain barrier and increases cell death after traumatic brain injury in mice. This suggest that over expression of slit2 plays a detrimental role in the pathophysiology of mild TBI. 27770832_Contralateral migration of oculomotor neurons is regulated by Slit/Robo signaling. Results demonstrate that a migratory subset of motor neurons respond to floor plate-derived Slit repulsion to properly control the timing of contralateral migration. 27819291_Both ISL1-LHX3 and ISL1-LHX4 bound to the Slit2 enhancer. 28234971_While Slit1 and Robo2 are only expressed in peripheral axons and their cell bodies, Slit2, Slit3 and Robo1 are also expressed in satellite cells of the dorsal root ganglion, Schwann cells and fibroblasts of peripheral nerves. 29660433_High Slit-2 expression is associated with hepatic fibrosis. 30177526_Trio induces RhoA activation downstream of Slit2, and support a functional role in ensuring the proper positioning of both guidepost cells and a major axonal tract. 31008488_Compared with proestrus ovaries, the expression levels of Slit2 and Robo1 mRNA in the ovaries during diestrus were significantly up-regulated. The mRNA expression levels of Slit2 and Robo1 in late-phase corpus luteum were significantly increased when compared with pregnant corpus luteum. 31084928_The results indicate that SLIT2 inhibits osteoclastogenesis and the resultant bone resorption by decreasing Cdc42 activity, suggesting that this is a potential therapeutic target in metabolic bone diseases related to high bone turnover states. 31356769_investigation of how Slit/Robo signals are involved in preventing emigrant motor neurons in the spinal cord 31545977_Slit2 is involved in the activation of HSCs and liver fibrogenesis. 32398956_Slit2/Robo1 Mitigates DSS-induced Ulcerative Colitis by Activating Autophagy in Intestinal Stem Cell. 32807784_SLIT2/ROBO1-signaling inhibits macropinocytosis by opposing cortical cytoskeletal remodeling. 32999457_Tumoural activation of TLR3-SLIT2 axis in endothelium drives metastasis. 33574432_The neurorepellent, Slit2, prevents macrophage lipid loading by inhibiting CD36-dependent binding and internalization of oxidized low-density lipoprotein. 34341180_Slit2-Robo Signaling Promotes Glomerular Vascularization and Nephron Development. 34490644_Ductular reaction promotes intrahepatic angiogenesis through Slit2-Roundabout 1 signaling. 36792623_Coadaptation fostered by the SLIT2-ROBO1 axis facilitates liver metastasis of pancreatic ductal adenocarcinoma. |
ENSG00000145147 |
SLIT2 |
1233.144397 |
0.2920307326 |
-1.775808 |
0.10394203 |
283.609338 |
0.00000000000000000000000000000000000000000000000000000000000000122765565841907676528545633338983776882034896987867449531491686732986986447146400510367420387868229572648084237349258733751508009608355530655249787912621352390513118635340106266085058450698852539062500 |
0.0000000000000000000000000000000000000000000000000000000000003016072740167641782355880017654953624756021858140884261338852914220964567816910909698692470744388476563433370867715694947851236296463885196194811749768969338925117540384235326200723648071289062500000 |
Yes |
No |
559.726852294905 |
40.3616089817489 |
1916.67105485271 |
99.4909752800482 |
| ENSMUSG00000031565 |
14182 |
Fgfr1 |
protein_coding |
P16092 |
FUNCTION: Tyrosine-protein kinase that acts as cell-surface receptor for fibroblast growth factors and plays an essential role in the regulation of embryonic development, cell proliferation, differentiation and migration. Required for normal mesoderm patterning and correct axial organization during embryonic development, normal skeletogenesis and normal development of the gonadotropin-releasing hormone (GnRH) neuronal system. Phosphorylates PLCG1, FRS2, GAB1 and SHB. Ligand binding leads to the activation of several signaling cascades. Activation of PLCG1 leads to the production of the cellular signaling molecules diacylglycerol and inositol 1,4,5-trisphosphate. Phosphorylation of FRS2 triggers recruitment of GRB2, GAB1, PIK3R1 and SOS1, and mediates activation of RAS, MAPK1/ERK2, MAPK3/ERK1 and the MAP kinase signaling pathway, as well as of the AKT1 signaling pathway. Promotes phosphorylation of SHC1, STAT1 and PTPN11/SHP2. In the nucleus, enhances RPS6KA1 and CREB1 activity and contributes to the regulation of transcription. FGFR1 signaling is down-regulated by IL17RD/SEF, and by FGFR1 ubiquitination, internalization and degradation (By similarity). {ECO:0000250|UniProtKB:P11362, ECO:0000269|PubMed:10821861, ECO:0000269|PubMed:10896947, ECO:0000269|PubMed:1309590, ECO:0000269|PubMed:17086194, ECO:0000269|PubMed:8001822, ECO:0000269|PubMed:8001823}. |
3D-structure;Alternative splicing;ATP-binding;Cell membrane;Cytoplasm;Cytoplasmic vesicle;Disulfide bond;Glycoprotein;Heparin-binding;Immunoglobulin domain;Kinase;Membrane;Nucleotide-binding;Nucleus;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transferase;Transmembrane;Transmembrane helix;Tyrosine-protein kinase;Ubl conjugation |
|
|
mmu:14182; |
cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; cytosol [GO:0005829]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; ATP binding [GO:0005524]; cell adhesion molecule binding [GO:0050839]; fibroblast growth factor binding [GO:0017134]; fibroblast growth factor receptor activity [GO:0005007]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803]; protein tyrosine kinase activity [GO:0004713]; protein-containing complex binding [GO:0044877]; receptor-receptor interaction [GO:0090722]; SH2 domain binding [GO:0042169]; signaling receptor binding [GO:0005102]; transmembrane receptor protein tyrosine kinase activity [GO:0004714]; angiogenesis [GO:0001525]; auditory receptor cell development [GO:0060117]; blood vessel morphogenesis [GO:0048514]; brain development [GO:0007420]; branching involved in salivary gland morphogenesis [GO:0060445]; calcium ion homeostasis [GO:0055074]; cardiac muscle cell proliferation [GO:0060038]; cell maturation [GO:0048469]; cell population proliferation [GO:0008283]; cementum mineralization [GO:0071529]; central nervous system neuron development [GO:0021954]; chondrocyte differentiation [GO:0002062]; diphosphate metabolic process [GO:0071344]; ear development [GO:0043583]; embryonic limb morphogenesis [GO:0030326]; epithelial to mesenchymal transition [GO:0001837]; fibroblast growth factor receptor signaling pathway [GO:0008543]; fibroblast growth factor receptor signaling pathway involved in orbitofrontal cortex development [GO:0035607]; gene expression [GO:0010467]; generation of neurons [GO:0048699]; in utero embryonic development [GO:0001701]; inner ear morphogenesis [GO:0042472]; lung development [GO:0030324]; lung-associated mesenchyme development [GO:0060484]; mesenchymal cell differentiation [GO:0048762]; mesenchymal cell proliferation [GO:0010463]; midbrain development [GO:0030901]; middle ear morphogenesis [GO:0042474]; motogenic signaling involved in postnatal olfactory bulb interneuron migration [GO:0021837]; negative regulation of fibroblast growth factor production [GO:0090272]; negative regulation of gene expression [GO:0010629]; negative regulation of osteoblast differentiation [GO:0045668]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuron projection development [GO:0031175]; orbitofrontal cortex development [GO:0021769]; organ induction [GO:0001759]; outer ear morphogenesis [GO:0042473]; paraxial mesoderm development [GO:0048339]; peptidyl-tyrosine phosphorylation [GO:0018108]; positive regulation of blood vessel endothelial cell migration [GO:0043536]; positive regulation of cardiac muscle cell proliferation [GO:0060045]; positive regulation of cell cycle [GO:0045787]; positive regulation of cell differentiation [GO:0045597]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of endothelial cell chemotaxis to fibroblast growth factor [GO:2000546]; positive regulation of fibroblast migration [GO:0010763]; positive regulation of hepatic stellate cell activation [GO:2000491]; positive regulation of inflammatory response [GO:0050729]; positive regulation of kinase activity [GO:0033674]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of MAPKKK cascade by fibroblast growth factor receptor signaling pathway [GO:0090080]; positive regulation of mesenchymal cell proliferation [GO:0002053]; positive regulation of mitotic cell cycle DNA replication [GO:1903465]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of neuron projection development [GO:0010976]; positive regulation of parathyroid hormone secretion [GO:2000830]; positive regulation of phospholipase C activity [GO:0010863]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of stem cell proliferation [GO:2000648]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of vascular endothelial cell proliferation [GO:1905564]; protein autophosphorylation [GO:0046777]; regulation of branching involved in salivary gland morphogenesis by mesenchymal-epithelial signaling [GO:0060665]; regulation of cell population proliferation [GO:0042127]; regulation of epithelial cell proliferation [GO:0050678]; regulation of extrinsic apoptotic signaling pathway in absence of ligand [GO:2001239]; regulation of gene expression [GO:0010468]; regulation of lateral mesodermal cell fate specification [GO:0048378]; regulation of phosphate transport [GO:0010966]; regulation of phosphorus metabolic process [GO:0051174]; regulation of sensory perception of pain [GO:0051930]; regulation of stem cell proliferation [GO:0072091]; response to sodium phosphate [GO:1904383]; salivary gland morphogenesis [GO:0007435]; sensory perception of sound [GO:0007605]; stem cell differentiation [GO:0048863]; stem cell population maintenance [GO:0019827]; stem cell proliferation [GO:0072089]; transmembrane receptor protein tyrosine kinase signaling pathway [GO:0007169]; ureteric bud development [GO:0001657]; vasculogenesis involved in coronary vascular morphogenesis [GO:0060979]; ventricular zone neuroblast division [GO:0021847]; vitamin D3 metabolic process [GO:0070640] |
12011115_FGFR1 signaling results in increased lateral budding of the mammary ductal epithelium, and sustained activation induces alveolar hyperplasia and invasive lesions. 12112370_FGF receptor 1 mRNA level is upregulated in reactive astrocytes in the area of the brain surrounding the necrotic tissue at day 7 after cryo-injury. 12121735_FGF-1, FGF-2 and FGF receptor-1 levels in the cochlear nucleus following acoustic overstimulation 12242295_crystal structure of the dimeric FGF2-FGFR1-SOS complex at 2.6-A resolution reveals a symmetric assemblage of two 1:1:1 FGF2-FGFR1-SOS ternary complexes 12421715_FGFR1 signaling plays a central role in mouse development by regulating both FGF and BMP expression 12514106_Fgfr1 patterns the pharyngeal region to create a permissive environment for neural crest cell migration. 12604616_FGFR1 tyrosine phosphorylation is inhibited by sef protein 12682014_Data suggest that after establishment of the isthmic organizer, fibroblast growth factor receptor 1 (FGFR1) is needed for continued response to the isthmic signals in the developing mid- and hindbrain. 12791257_Results describe a direct interaction between neural cell adhesion molecule (fibronectin type III [F3] modules 1 and 2) and fibroblast growth factor receptor R1 (Ig modules 2 and 3) by surface plasmon resonance analysis. 12893744_Data point to a nonredundant role for FGFR1-mediated signaling in cardiomyocyte development. 14559809_Activation of iFGFR1, but not iFGFR2, led to strong up-regulation of osteopontin in prostate adenocarcinoma. 14617521_FGF signaling is required for maintenance of surfactant homeostasis and lung function during hyperoxia in vivo, mediated, at least in part, by its role in the maintenance of SP-B expression. 14660670_cell transformation mediated by ZNF198-FGFR1 requires STAT5 activation 14668415_Fgfr1-mediated developmental pathways exhibit differing signalling dynamics, where development of the cranial sutures and sternum follows a morphogen mode, whereas development of the vertebral column and the hind limbs has threshold signalling properties. 14673947_Chronic activity of ectopic type fgfr-1 tyrosine kinase in prostate epithelium results in hyperplasia accompanied by intraepithelial neoplasia. 14678983_By timed removal of FGFR1 signaling, it is shown that induced prostatic hyperplasia is reversible until extensive intraductal vascularization occurs, but continued progression requires prolonged FGFR1 signaling. 15041185_mouse leukemia virus infection reduces bFGF transcript expression but not its surface-receptors bek and flg in infected stromal cells 15085950_FGFR1-IIIb is dispensable for skin development and wound repair 15513912_FGFR kinase regulates the cell cycle through phosphorylation-dependent release of Cks1 from FGFR substrate 2 15564375_FRS2-dependent SRC activation is required for FGFR-induced phosphorylation of Sprouty and suppression of ERK activity 15576401_FGFR1 and FGFR4 are involved in patterning the foregut 15581874_Data suggest that fibroblast growth factor receptor (FGFR) 2, but not FGFR1, appears crucial for ureteric bud branching morphogenesis and stromal mesenchyme patterning. 15680361_Fgfr1 is required for establishment of a straight border between developing mid- and hindbrain. 15774903_FGFR-1 is expressed in early hematopoietic/endothelial precursor cells, as well as in a subpool of endothelial cells in tumor vessels, and that it is critical for hematopoietic but not for vascular development 15830353_developmental expression analysis in the mid and hindbrains 16120640_During autopod patterning, FGFR1 influences digit number and identity. 16172381_Npas3-/- mice are deficient in expression of hippocampal FGF receptor subtype 1 mRNA. 16207751_FGF/FGFR1 signals play indispensable roles in the early stages of limb initiation, eliciting a profound effect on the later stages of limb development, including cell survival, autopod formation and digit patterning. 16280414_findings describe a direct role of FGFR signaling in the elongation and guidance of GNRH1 axons to the median eminence 16291796_FGF-mediated activation of the MAP kinase pathway occurs in germ cells during their migration, FGF2, a ligand for FGFR1-IIIc, affects motility, whereas FGF7, a ligand for FGFR2-IIIb, affects germ cell numbers. 16301332_FGFR1 has roles in cell proliferation, survival, and migration in mouse mammary epithelium 16309667_Fgfr1 is found to be essential for establishing all three commissural tracts. 16352663_Data show that fibroblast growth factor receptor-1 is required for normal spermatogenesis and has a key role in the regulation of the global tyrosine phosphorylation events associated with sperm capacitation. 16421190_Fgfr1 uses distinct signal transduction mechanisms in different developmental contexts, and that some essential functions of this receptor are mediated by Frs-independent signaling. 16446698_Our results demonstrate that Fgfr1 is not cell autonomously essential for lens development and suggests functional redundancy among different FGF receptor genes with respect to lens fiber differentiation. 16524369_Thus, this study establishes a new developmental model for a schizophrenia-like disorder in which the inhibition of FGF signaling leads to alterations in DA neurons and DA-mediated behavior. 16715082_FGFR1 has an important role in the transition from radial glia to astrocytes by stimulating somal translocation of radial glial cells. 16731982_the NMR structure of the Ig1 module of mouse FGFR1. The three-dimensional fold of the module belongs to the intermediate Ig subgroup and can be described as a beta-barrel consisting of two beta-sheets. 16782087_FGFR1 regulates gene expression in the midbrain-hindbrain boundary in embryo 16815385_data demonstrate that signaling through FGFR1 in osteoblasts is necessary to maintain the balance between bone formation and remodeling through a direct effect on osteoblast maturation 17005551_Co-immunoprecipitation and co-clustering paradigms were used to show that both NCAM and N-cadherin can interact with the 3Ig IIIC isoform of the FGFR1 in a number of cell types. 17008716_identify binding sites involved in the Ig1-Ig2 interaction; results suggest that the function of the Ig1 module is not only regulation of the FGFR-ligand binding affinity but also prevention of spontaneous FGFR dimerization in the absence of FGF. 17077327_identified FGFR1 IIIc as the receptor mediating FGF2 inhibition of SDF-1 expression in bone marrow stromal cells 17239352_A critical role of FGFR1 in adult neurogenesis in vivo, provides a potential link between proliferative neurogenesis and dentate LTP, and raises the possibility that adult neurogenesis might contribute to memory consolidation. 17284663_FGFR1 and FGFR2 markedly differ in their ability to induce phenotypic changes characteristic in the early stages of mammary carcinogenesis. 17559077_Anti-apoptotic Bcl-2 enhancing requires Fgf2/Fgfr1 binding in mouse osteoblasts. 17875810_FGFR-1 negatively regulates endothelial cell function by altering the balance of modulatory cytokines 17901128_Results suggest that although the tyrosine autophosphorylation sites of FGFR1 did not constitute the binding sites for FRS2alpha, phosphorylation of these residues was essential for optimal interaction with FRS2alpha. 17965051_Role of FGF signaling in segmentation, and a signaling hierarchy controlling clock oscillations downstream of FGF signaling in the mouse. 17975556_Data show that internalization of activated FGFR1 is associated with release from complexes containing activated Src and FGFR1-mediated signalling requires for Src family kinases. 17988653_Fgfr1(f/f;hGfapCre) mice have spontaneous locomotor hyperactivity characterized by longer bouts of locomotion and fewer resting points and parvalbumin+, somatostatin+, and calbindin+ inhibitory interneurons were reduced in number in the cerebral cortex. 18042549_the involvement of FGFR-1 through FGF2 in eliciting PGE(2) angiogenic responses 18068632_Activation of iFGFR1 with chemical inducers of dimerization (CID) led to highly synchronous, step-wise progression to adenocarcinoma that is linked to an epithelial-to-mesenchymal transition. 18093225_Expression of R522K-Shb results in up-regulation of FGFR1 in embryoid bodies. Interplay between the FGF system and activin A participates in endoderm differentiation. 18171671_Basic fibroblast growth factor-induced neuronal differentiation of mouse bone marrow stromal cells requires FGFR-1, MAPK/ERK, and transcription factor AP-1 18187602_Data show FGF21 alone does not activate FGFRs and that betaKlotho is required for FGF21 to activate two specific FGFR subtypes: FGFR1c and FGFR3c. 18191119_Negative feedback of fibroblast factors triggers excessive cell proliferation then inhibits the expression of Fgfr1b and activates the expression of Wnt11 to fuse each palate. 18296607_Enamel-secreting ameloblasts failed to adhere to each other, although ameloblast differentiation was unaffected at early stages. Prior to eruption, enamel structure was compromised; it displayed severe enamel defects like amelogenesis imperfecta. 18411303_p38alpha MAPK has a critical role in the regulated translocation of exogenous FGF1 into the cytosol/nucleus 18440247_data reveal that the location of cytosolic FGFR1 depends on the motile characteristics of the urothelial cell 18455718_Deletion of 3 FGF receptors (Fgfr1-3) early in lens development showed that expression of only a single allele of Fgfr2 or Fgfr3 was sufficient for normal lens development, while mice with only a single Fgfr1 allele developed cataracts and microphthalmia. 18566132_the early emergence of GnRH neurons from the embryonic olfactory placode requires FGF8 signaling, which is mediated through FGFR1, not FGFR3 18624916_The interaction between NCAM and FGFR probably involves multiple contact sites at an interface formed by the two NCAM FN3 modules and FGFR; encamins are pharmacological tools for the study of NCAM, including neuroprotection and modulation of plasticity. 18753255_neither FGFR3 nor FGFR4 is the principal mediator of FGF23 effects in the proximal tubule, and co-localization of FGFR1 and Klotho suggests that the distal tubule may be an effector site of FGF23 18937030_bFGFR-1 mediates AAV2 transduction of Purkinje cells. 18973581_The results also suggest a role of the deficit in NCAM signalling through the FGF receptor in depression. 19176828_Fgfr1 is required for cortical regeneration and repair after perinatal hypoxia. 19238727_Signaling pathways activated by epidermal growth factor receptor or fibroblast growth factor receptor differentially regulate branching morphogenesis in fetal mouse submandibular glands. 19261810_Fluorescence recovery after photobleaching analysis revealed three nuclear FGFR1 populations: i) a fast mobile, ii) a slower mobile population reflecting chromatin-bound FGFR1, and iii) an immobile FGFR1 population associated with the nuclear matrix. 19408015_A detailed double-label immunohistochemical investigation of the localization patterns of FGF1 and its receptors FGFR1 and FGFR2 in adult and early postnatal mouse retinas, is reported. 19502291_FGF signaling is required for the induction of alphaSMA in the PDGFRalpha-positive myofibroblast progenitor and the progression of alveolar regeneration 19506298_An important event in the process of ZNF198-FGFR1-induced T-cell leukemia. 19515808_FGF23 decreases renal NaPi-2a and NaPi-2c expression and induces hypophosphatemia in vivo predominantly via FGF receptor 1. 19549074_Aim of this study was the characterization of FGFR1 signalling requirements necessary for cardiomyocyte differentiation in murine embryonic stem cells. 19574212_that the main role of heparin in FGF-induced signaling is to protect this naturally unstable protein against heat and/or proteolytic degradation and heparin is not essential for a direct FGF1-FGFR interaction and receptor activation 19598249_Dysfunction of glial cells and FGF receptor signaling may therefore be implicated in neurodegenerative hearing loss associated with normal aging. 19663702_monoclonal antibody analysis of multiple FGFR1 isoforms, generated by alternative splicing and post-translational modifications through glycosylation 19666467_a neurofascin intracellular domain activates FGFR1 for neurite outgrowth, whereas the extracellular domain functions as an additional, regulatory FGFR1 interaction domain in the course of development 19715689_Results show that Fgfrl1-/- embryos fail to develop the metanephric kidney, and suggest that Fgfrl1 is essential for mesenchymal differentiation in the early steps of nephrogenesis. 19728793_The aim of this study was to investigate and compare FGFR expression in in vivo embryonic limb development and in vitro chondrogenesis of mesenchymal stem cells. 19739251_Activation of MAPK signalling pathways following interaction with the FGFR is involved in the survival effect of NCAM. 19800312_FGFR1 is indispensable for complete differentiation and activation of osteoclasts in mice. 19909731_We conclude that in NCAM deficient mice FGFR1-dependent signaling is disrupted and it can be restored by FGL peptide. 20103604_brivanib is a dual inhibitor of vascular endothelial growth factor receptor-2 and fibroblast growth factor receptor-1 kinases 20131355_analysis of FGF receptor expression in the developing mammalian cochlea 20308431_The role for Fibroblast growth factor receptor 1 in the regulation of the epidermal barrier and in the prevention of cutaneous inflammation, and highlight the importance of stromal-epithelial interactions in skin homeostasis and disease. 20405041_autoregulation of Fgfr1 and Fgfr3 expression by Fgf8 represents a mechanism by which FGF8 could fine-tune its regulatory actions 20463222_results indicate that MAPK/ERK activation downstream of FGFR1 is necessary for motor axon guidance and that embryonic stem cell-derived neurons provide an important tool for dissecting intracellular pathways required for axon guidance 20501844_Fibroblast growth factor receptor signaling dramatically accelerates tumorigenesis and enhances oncoprotein translation in the mouse mammary tumor virus-Wnt-1 mouse model of breast cancer. 20501936_Data show that knockout of gene Axin2, a negative regulator of the WNT-beta-catenin pathway, and gene FGFR1 induced ectopic chondrogenesis, leading to abnormal suture morphogenesis and fusion. 20553372_Results support the hypothesis that patients with digenic mutations in Fgfr1/Fgf8, but not Fgfr3, may have a further reduction in the GnRH neuronal population compared to patients harbouring monogenic haploid mutations in Fgfr1 or Fgf8. 21124973_Data show that FGF signaling plays a key role in determining biological differences between the two types of osteoblasts. 21128305_Fgfr2IIIc and Fgfr1 in kidney mesenchyme (together) are critical for normal early renal development. 21223966_The results demonstrate that the FGF1/FGFR1 complex constitutes a signalling module that independently of the receptor tyrosine kinase can convey a signal that initiates a strictly timed and periodic release of endocytosed FGF1 into the cytosol/nucleus. 21451043_Fgfr1 and Fgfr2 (but not Fgfr3) are required for the generation of oligodendrocyte progenitor cells in the forebrain, with Fgfr1 being a stronger inducer than Fgfr2. 21479780_A missense mutation (W691R) in the kinase domain of Fgfr1 causes ear and skull defects in hush puppy mice. 21527526_Fgfr1/2 are expressed by bone-derived mesenchymal stem cells in vivo and in vitro and are developmentally regulated during their differentiation. 21527531_The study demonstrates frequent, constitutive activation of Notch1 and its downstream target genes in T-cell lymphomas that arose in a murine model of Zmym2-Fgfr1 stem cell leukemia-lymphoma syndrome. 21937681_Src was constitutively active in BaF3 cells expressing exogenous FGFR1 chimeric kinases cultured in vitro as well as in T-cell or B-cell lymphomas they induced in vivo. 22247553_Fibronectin induces endothelial cell migration through beta1 integrin and Src-dependent phosphorylation of fibroblast growth factor receptor-1 at tyrosines 653/654 and 766. 22276098_S100B is a danger signal released from injured muscles participates in skeletal muscle regeneration by activating the promyogenic RAGE or the mitogenic bFGF/FGFR1 depending on its own concentration, the absence or presence of bFGF, and myoblast density. 22278983_study examined if female mice hypomorphic for Fgf8, Fgfr1, or both (compound hypomorphs) exhibited altered parameters of pubertal onset, estrous cyclicity, and fertility 22282599_Conditional deletion of fibroblast growth factor receptors (Fgfrs) 1 and 2 in the metanephric mesenchyme (MM) of mice leads to a virtual absence of MM and unbranched ureteric buds that are occasionally duplex. 22370560_In contrast to wild-type FGF23, FGF23-21c (a variant of FGF23 where the C-terminal domain of FGF23 was replaced with the corresponding regions from FGF21), FGF23-21c gained the ability to activate FGFR1c and FGFR2c in the presence of betaKlotho. 22442730_Differential specificity of endocrine FGF19 and FGF21 to FGFR1 and FGFR4 in complex with KLB. 22457778_generated a variant of FGF19, FGF19-7, that has altered receptor specificity with a strong bias toward FGFR1c 22523080_KLB and FGFR1 form a 1:1 heterocomplex independent of the galectin lattice that transitions to a 1:2 complex upon the addition of FGF21. 22573685_Fgfr1 and Fgfr2 activation in oligodendrocytes is an important late-stage regulatory mechanism in the control of myelin growth and myelin sheath thickness in the central nervous system. 22578469_Disrupted cerebellar size and laminar architecture resulting from loss of FGFR1 signaling impair motor learning and coordination in FGFR double knockout mice. 22735182_Ascorbic acid rescues cardiomyocyte development in Fgfr1(-/-) murine embryonic stem cells. 22751148_NOSTRIN participates in FGF signal transduction and vascular development. 22761819_These data confirm that both FGF and VEGF signaling are necessary for the maintenance of vascular morphogenesis 22802336_Data indicate that FGF receptor 1 (FGFR1) conditional knockout mice showed defects in both proliferation and subsequent mobilization of hematopoietic stem and progenitor cell (HSPC). 22833219_Disruption of FGFR-1 in adult mouse articular chondrocytes inhibits the progression of cartilage degeneration. 22836867_Phosphate-independent effects of high-molecular weight (HMW) isoforms in vitro may be directly mediated in part via FGF23 and HMW isoforms signal via FGF23/FGFR/MAPK to inhibit bone formation in vitro. 22893608_These results indicate that macrophage-derived Cxcr2 ligands may be important for promoting mammary tumor formation regulated by FGFR signaling. 22950531_transgenic mice with reduced FGF signaling have not only suboptimal reproductive physiology, but also suboptimal maternal behavior. 22992463_FGF receptors 1 and 2 are key regulators of keratinocyte migration in vitro and in wounded skin. 23097355_a significant reduction in outgrowth potential was observed upon the deletion of both FGFR1 and FGFR2, documenting the requirement for functional FGFR signaling in mammary stem cells during development. 23137310_fibroblast growth factor receptor (FGFR)1 and FGFR2 are found in a variety of embryonic olfactory cells, including olfactory ensheathing cells and their precursors, and neuronal nestin+ and Mash1+ progenitors 23185502_our results demonstrate that FGFR1 is crucial for S115 breast cancer cell proliferation and tumor growth and angiogenesis, whereas FGFR2 and FGFR3 are less critical for the growth of these cells 23451204_work reveals the central role of FGFR1 in the regulation of FGF23 production and signal transduction, and has implications in the pathogenesis of FGF23-related hypophosphatemic disorders 23564461_Phosphorylation of serine 779 in fibroblast growth factor receptor 1 and 2 by protein kinase C(epsilon) regulates Ras/mitogen-activated protein kinase signaling and neuronal differentiation. 23576558_Data indicate that mice carrying the fgfr1 null allele survived significantly longer than those without fgfr1 deletion. 23684572_Therefore, constitutive deletion of Fgf2 or Fgfr1 knockdown in oligodendrocyte lineage cells is sufficient to overcome impairment of sensorimotor coordination after cuprizone demyelination. 23715864_Data indicate that FGF-2 isoforms differentially modulate bone morphogenetic protein 2 (BMP-2) effect on FGF receptor 1 and FGF receptor 2 levels in osteoblast. 23754280_Data indicatethat deleting Fgfr1 in neural crest cells caused defects in both palate shelf epithelium and mesenchyme and led to cleft palate. 23777766_CNTRL and FGFR1 have roles in myeloid and lymphoid malignancies in both human and mouse models 24000064_Canonical Wnt-, Hh-, and Fgfr1/Fgfr2-signalling are dispensable for epicardial development, but Pdgfra-signalling is crucial for the differentiation of cardiac fibroblasts from epicardium-derived cells. 24026051_Evidence that the novel receptor FGFRL1 signals indirectly via FGFR1. 24349409_FGFR overexpression results in HCM with a dynamic outflow tract obstruction, and may serve as a unique model of HCM. 24465223_Analysis of a mutant Fgfr1 allele, unable to bind to the adaptor protein, Frs2/3, indicates that Sox2 maintenance can be regulated by MAP kinase. 24512770_This study demonistrated that Fgf8- and Fgfr1/Fgf8-deficient mice diplay increased anxiety-like behavior and reductions in specific populations of serotonergic neurons in the brain. 24618085_Data suggest that fibroblast growth factor receptor type 1 (FGFR1) isoform expression can be used as a predictive biomarker for therapeutic application of its kinase inhibitors. 24752320_This data shows that primitive endoderm cells of the outer layer of embryoid bodies gradually polarise, and formation of a polarised primitive endoderm layer requires the Fgf receptor/Erk signalling pathway. 25089825_ctivation of autocrine/paracrine FGF pathways is involved in the pathogenesis of Hyp through FGFR1-dependent regulation of FGF23 by both transcriptional and post-transcriptional mechanisms. 25116473_Fgfr1 inactivation in the mouse telencephalon results in impaired maturation of interneurons expressing parvalbumin. 25249657_Endothelial cell-specific deletion of Fgfr1 enhanced endothelial-to-mesenchymal transition in knockout mice. FGFR1 is the key regulator of TGFbeta signaling and EndMT development. 25285038_This study suggests that targeted deletion of Fgfr1 in osteoblasts enhances mobilization of endothelial progenitor cells into peripheral blood through up-regulating SDF-1alpha secretion from osteoblasts. 25300351_FGF receptor 1-mediated anosmin-1 activity plays a crucial role in the continuous remodelling of the adult olfactory bulb. 25400756_miR-34a negatively modulated anesthesia-induced hippocampal neurotoxicity via FGFR1. 25616962_FGFR1 signaling in hypertrophic chondrocytes is attenuated by the Ras-GAP neurofibromin during endochondral bone formation 25641696_Fgfr1 and Fgfr2 have synergistic roles in maintaining nephron progenitors. 25792827_Studied the protein expression of BCL2, FGF-R1, and HSP70 after short-time magnetic thermoablative tumor treatment using immunohistochemistry in a human BT474 breast cancer mouse xenograft model. 25820239_Conditional knockout of Fgfr1 revealed stage- and tissue-specficic roles of FGF signaling in multiple processes of external genitalia development among the 3 tissue layers at each developmental stage. 25913734_The results of this study concluded that since the OPC response was not altered in Fgfr1/Fgfr2 dKO mice either after initial demyelination or after chronic demyelination. 25923916_Nuclear FGFR1 targets the consensus sequences of transcription factors known to engage CREB-binding protein, a common coregulator of transcription and established binding partner of nuclear FGFR1. 26024354_These findings suggest that the ternary complex of EphA, FGFR and FRS2alpha formed by ligand stimulation regulates self-renewal and differentiation of mouse embryonic neural stem/progenitor cells 26250517_Fgfr1 and Fgfr2 in the palatal and mandibular mesenchyme have roles in regulating shelf medial wall protrusion and growth of the mandible to coordinate the craniofacial tissue movements that are required for palatal shelf elevation 26311115_Activation of FGFR1 is essential for the high levels of FGF23 in acute and chronic experimental uremia. 26341559_Frs2 binding to Fgfr1 has the most pleiotropic functions in development but also that the receptor uses multiple proteins additively in vivo 26476925_demonstrated that Id1 and E2-2 are critical regulators of EPCs function in vitro. Id1 interacts with E2-2 and relieves the E2-2-mediated repression of FGFR1 and VEGFR2 expression to modulate EPCs functions 26581390_a transplantable Wnt1/inducible fibroblast Arowth factor receptor (FGFR) 1 mouse mammary tumor model. 26589915_FGFR1OP2-FGFR1 fusion in hematopoietic stem cells induced myeloid leukemia and T-cell lymphoma in a mouse model. 26747503_Data demonstrate an essential role for FGFR1 and FGFR2 in endothelial cells for cardiac functional recovery and vascular remodeling following in vivo cardiac ischemia-reperfusion injury, without affecting the cardiac hypertrophic response. 26762170_Data suggest that signal transduction via Fgf23/Fgfr1 and calcitriol/calcitriol receptor have opposite roles in innate immunity; Fgf23 suppresses arginase-1 expression in macrophages; calcitriol stimulates arginase-1 expression in macrophages. 26839958_FGFR1 has dual functions to directly regulate proximal and distal tubule phosphate and calcium reabsorption, indicating a physiological role of FGFR1 signaling in both phosphate and calcium homeostasis. 26847131_Data suggest that Fgf1-mediated signaling represents an important signaling cascade related to adipogenesis and visceral adiposity; expression of Fgf1 (fibroblast growth factor 1) and Fgfr1 (fibroblast growth factor receptor 1) is up-regulated in adipose tissue of obese mice (both obese mice due to high-fat diet and obese mice due to genetic deletion of leptin). 26872365_MiR-214 was up-regulated in mesenchymal stem cells of osteoporotic mice and down-regulated during osteoblast differentiation of mesenchymal stem cells. FGFR1 is a direct target of miR-214. 26881702_The study supports a pro-adipogenic role for betaKlotho in skeletal muscle fibro/adipogenesis and calls for further research on involvement of the FGF-FGFR-betaKlotho axis in the fibro/adipogenic infiltration associated with functional deterioration of skeletal muscle in aging and muscular dystrophy. 27078042_the localization of FGF9 and its receptors at different embryonic and postnatal stages in mice testes, was examined. 27133954_data suggest that FGF2 levels are critically related to anxiety behavior and hypothalamic pituitary- adrenal axis activity, likely through modulation of hippocampal glucocorticoid receptor expression, an effect that is likely receptor mediated, albeit not by FGFR1, FGFR2, and FGFR3. 27391347_FGFR1 is a driver oncogene in de novo, FGFR1-overexpressing acute myeloid leukemia 27690692_These new findings reveal that the FGF21-betaKlotho-FGFR1 signaling axis plays roles in maintaining phospholipid homeostasis and the dynamic functions of the lipid droplet, whereas protecting against ER stress, and suggest a potential link of phospholipid biosynthesis, lipid droplet dynamics, ER stress, and energy homeostasis in adipose tissue coordinated by this signaling axis. 28031461_clearly demonstrate the different specificity of FGF12-FGFR1c2 and FGF22-FGFR1c2 for well defined HS structures and suggest that it is now possible to chemoenzymatically synthesize precise HS polysaccharides that can selectively mediate growth factor signaling 28117910_Oligodendroglial FGFR1 deficient mice (-/-) showed a significantly ameliorated disease course in MOG35-55 -induced experimental autoimmune encephalomyelitis. Less myelin and axonal loss, and reduced lymphocyte and macrophage/microglia infiltration were found in Fgfr1(-/-) mice. Reduction in disease severity in Fgfr1(ind-/-) mice was accompanied by ERK/AKT phosphorylation, and increased expression of BDNF and TrkB. 28193689_It is well accepted that myelin is a biologically active membrane in active communication with the axons. However, the axonal signals, the receptors on myelin, and the integration of intracellular signaling pathways emanating downstream from these receptors that drive the growth of the myelin sheath remain poorly understood in the CNS. This study brings up the intriguing possibility that FGF receptor 2, in the oligodendr 28465335_CDC42 is involved in the trafficking of FGF receptors to the cell membrane to regulate epicardium formation. 28537241_MAPK cascades participate in osteogenesis, but only the ERK signaling pathway responds to FGFR1. 28771231_the close proximity between AcSDKP and FGFR1 was essential for the suppression of TGFbeta/smad signaling and EndMT associated with MAP4K4 phosphorylation (P-MAP4K4) in endothelial cells. 28783178_Visceral adipose tissue-derived factors stimulate cell transformation through FGFR-1. 28813681_Suboptimal FGFR activation by a weak FGF1-FGFR dimer is sufficient to evoke a metabolic response, whereas full FGFR activation by stable and sustained dimerization is required to elicit a mitogenic response. 28923346_FGFR1 and FGFR2 regulate FGFRL1 expression. 28993502_In summary, genetic deletion of FGFR1 in the kidney, likely due to loss of FGFR1 in theDT, leads to hypertension and LVH, whereas an FGFR1-activating antibody, R1MAb1, lowers BP in a hypertensive mouse model, and reverses hypertension observed in the Hypmousemodel of FGF-23 excess. Our findings suggest that FGFR1 in renal tubules plays a physiologic role in regulating systemic hemodynamics. 29691281_Deletion of Fgfr1 in Temporomandibular joint chondrocytes delayed Temporomandibular joint Temporomandibular joint osteoarthritis progression in the age-associated spontaneous Temporomandibular joint osteoarthritis model and the abnormal dental occlusion Temporomandibular joint osteoarthritis model. 29844125_FGFR1-Activated Translation of WNT Pathway. 30061390_a modified ciliary transport pathway used for Pcdh15 transport into the cilium of the inner ear hair cell and coordinated by FGFR1 activity. 30217817_FGFR5 as a co-receptor that is up-regulated by inflammation and promotes FGFR1-induced survival. 30283071_These studies reveal previously unrecognized Vegfr2 gene dosage effects in tumor angiogenesis and a lack of synergy between VEGFR2 and endothelial FGFR1/2 signaling during tumor growth. 30439482_These studies provide insights into the molecular etiology of development and progression of FGFR1 driven AML. 30499042_Loss of Fgfr1 in neural crest cells leads to heterotopic chondrogenesis and osteogenesis. 30552867_Results suggest that while the trophectoderm (TE) development is specified in fibroblast growth factor receptor 1 (Fgfr1) mutants, its differentiation abilities are compromised leading to defects at implantation. 30763736_Aberrant Notch2 expression enhances fibroblast growth factor receptor-1 (FGFR1) activity to specifically target the AKT-GSK3 signalling pathway to block apoptosis. These results have implications for understanding molecular changes involved in both tumorigenesis and therapy resistance. 31206783_Osteocyte Death and Bone Overgrowth in Mice Lacking Fibroblast Growth Factor Receptors 1 and 2 in Mature Osteoblasts and Osteocytes. 31208421_regulation of fibroblast growth factor receptor 1 trafficking and function by extracellular galectins 31276493_Results find that FGFR1 regulates Sox2 expression and provide evidence that Fgfr1 is involved in prosensory specification and differentiation. 31525277_Authors have deleted the STK domain and mutated the critical Y177 residue and demonstrate that the transforming activity of these mutated genes is reduced compared to the BCR-FGFR1 parental kinase. In addition, we demonstrate that deletion of the FGFR1 tyrosine kinase domain abrogates transforming ability, which is not compensated for by BCR STK activity. 31830366_Mouse genetics identifies unique and overlapping functions of fibroblast growth factor receptors in keratinocytes. 32187553_FGFR1 Oncogenic Activation Reveals an Alternative Cell of Origin of SCLC in Rb1/p53 Mice. 32422143_DC-SCRIPT affects mammary organoids branching morphogenesis by modulating the FGFR1-pERK signaling axis. 32735383_FGF20-FGFR1 signaling throug |
ENSG00000077782 |
FGFR1 |
5867.978940 |
0.4131338335 |
-1.275319 |
0.07098809 |
314.479813 |
0.00000000000000000000000000000000000000000000000000000000000000000000023086060229785573296316397328960995650583365600333519207737124296118963556192603640844715782722659090071928366864246792859273227219927233845927412577124351130582655101074196490884560262202285230 |
0.0000000000000000000000000000000000000000000000000000000000000000000689503665529595816570290210721122633505168111112302272915773212228741456539126973292354602444399022975500787769435579898730510986193691484590703654808567544328296174778111793379764549172250554 |
Yes |
No |
3467.2773625042 |
170.676913966245 |
8392.62505618449 |
305.545124129713 |
| ENSMUSG00000031586 |
19663 |
Rbpms |
protein_coding |
Q9WVB0 |
FUNCTION: Acts as a coactivator of transcriptional activity. Required to increase TGFB1/Smad-mediated transactivation. Acts through SMAD2, SMAD3 and SMAD4 to increase transcriptional activity. Increases phosphorylation of SMAD2 and SMAD3 on their C-terminal SSXS motif, possibly through recruitment of TGFBR1. Promotes the nuclear accumulation of SMAD2, SMAD3 and SMAD4 proteins. Binds to poly(A) RNA. {ECO:0000250|UniProtKB:Q93062}. |
Acetylation;Activator;Alternative splicing;Cytoplasm;Nucleus;Phosphoprotein;Reference proteome;RNA-binding;Transcription;Transcription regulation |
|
|
mmu:19663; |
cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; P-body [GO:0000932]; mRNA binding [GO:0003729]; poly(A) binding [GO:0008143]; protein homodimerization activity [GO:0042803]; RNA binding [GO:0003723]; transcription coactivator activity [GO:0003713]; positive regulation of pathway-restricted SMAD protein phosphorylation [GO:0010862]; positive regulation of SMAD protein signal transduction [GO:0060391]; response to oxidative stress [GO:0006979] |
25651939_Cytoplasmic granule containing HERMES, NonO, PSF, and G3BP1 is a neuronal RNA-protein granule that is transported in neurites during retinal differentiation. 35472321_RBPMS is an RNA-binding protein that mediates cardiomyocyte binucleation and cardiovascular development. |
ENSG00000157110 |
RBPMS |
21.741742 |
0.0898964111 |
-3.475593 |
0.79139897 |
17.464303 |
0.00002927532919284707774360866638918565740823396481573581695556640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001975728020671008850189104277816909416287671774625778198242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.35498475066566 |
1.93409569627924 |
37.3205638417794 |
13.8136051786417 |
| ENSMUSG00000031595 |
68797 |
Pdgfrl |
protein_coding |
A0A0R4J0C9 |
|
Disulfide bond;Glycoprotein;Reference proteome;Repeat;Signal |
|
|
mmu:68797; |
extracellular region [GO:0005576] |
28407304_These findings suggest that PDGFRL plays the different roles, depending upon cell types. Particularly, in chondrocytes, PDGFRL may play a new and important role which is distinct from the function previously reported. |
ENSG00000104213 |
PDGFRL |
291.535752 |
0.4075752614 |
-1.294862 |
0.17627997 |
53.484402 |
0.00000000000026065420900453078622871266835330301194504146189423465784784639254212379455566406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000055840856702630280596006126151774979961966982600074516085442155599594116210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
165.581859608053 |
20.2824914901912 |
406.2608192469 |
35.9024224409897 |
| ENSMUSG00000031626 |
234214 |
Sorbs2 |
protein_coding |
Q3UTJ2 |
FUNCTION: Adapter protein that plays a role in the assembling of signaling complexes, being a link between ABL kinases and actin cytoskeleton. Can form complex with ABL1 and CBL, thus promoting ubiquitination and degradation of ABL1 (By similarity). May play a role in the regulation of pancreatic cell adhesion, possibly by acting on WASF1 phosphorylation, enhancing phosphorylation by ABL1, as well as dephosphorylation by PTPN12 (By similarity). {ECO:0000250, ECO:0000250|UniProtKB:O94875}. |
Alternative splicing;Cell junction;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;Membrane;Phosphoprotein;Reference proteome;Repeat;SH3 domain;Ubl conjugation |
|
|
mmu:234214; |
apical plasma membrane [GO:0016324]; cytoskeleton [GO:0005856]; dendrite [GO:0030425]; focal adhesion [GO:0005925]; lamellipodium [GO:0030027]; neuronal cell body [GO:0043025]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; synapse [GO:0045202]; Z disc [GO:0030018]; identical protein binding [GO:0042802]; protein domain specific binding [GO:0019904]; acetylcholine receptor signaling pathway [GO:0095500]; cell growth involved in cardiac muscle cell development [GO:0061049]; Notch signaling pathway [GO:0007219]; regulation of skeletal muscle acetylcholine-gated channel clustering [GO:1904393] |
26527617_The study screened CrkL binding proteins using RNA interference (RNAi) and identified Sorbs1 and Sorbs2 as two proteins that are enriched at AChR clusters and are required for the formation of AChR aggregation in vitro. 27033308_the present study provides strong evidence that miR-21-3p controls sepsis-associated cardiac dysfunction via regulating SORBS2. Inhibition of miR-21-3p might be a protective strategy to treat sepsis-induced cardiac dysfunction. 27530683_The nArgBP2 binds to the two most prominent postsynaptic proteins that are associated with various mood disorders, we propose that nArgBP2could function as a hub able to link together etiological factors of different disorders. 32808564_Knockout of SORBS2 Protein Disrupts the Structural Integrity of Intercalated Disc and Manifests Features of Arrhythmogenic Cardiomyopathy. 34487812_Changes in ion channel expression and function associated with cardiac arrhythmogenic remodeling by Sorbs2. 35050860_The interaction protein of SORBS2 in myocardial tissue to find out the pathogenic mechanism of LVNC disease. 35730644_Knockout of Sorbin And SH3 Domain Containing 2 (Sorbs2) in Cardiomyocytes Leads to Dilated Cardiomyopathy in Mice. |
ENSG00000154556 |
SORBS2 |
3504.327935 |
0.4047616469 |
-1.304856 |
0.06127336 |
444.720974 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001016231637980444383968403446062214991190686719262144139326917090072863414887185518777001318099993738952614299363089953202847957375957641636003128649260645966605879917 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000055283001106136174487881147465784495520773357527860641179384289699963769769862892221468871704639659399022217885352093454234928881252095704998570198519779140583359867 |
Yes |
No |
2000.109309919 |
84.9730926382663 |
4941.4496783572 |
153.109410177525 |
| ENSMUSG00000031659 |
11513 |
Adcy7 |
protein_coding |
P51829 |
FUNCTION: Catalyzes the formation of cAMP in response to activation of G protein-coupled receptors (Probable). Functions in signaling cascades activated namely by thrombin and sphingosine 1-phosphate and mediates regulation of cAMP synthesis through synergistic action of the stimulatory G alpha protein with GNA13 (PubMed:18541530). Also, during inflammation, mediates zymosan-induced increase intracellular cAMP, leading to protein kinase A pathway activation in order to modulate innate immune responses through heterotrimeric G proteins G(12/13) (PubMed:23178822). Functions in signaling cascades activated namely by dopamine and C5 alpha chain and mediates regulation of cAMP synthesis through synergistic action of the stimulatory G protein with G beta:gamma complex (By similarity). Functions, through cAMP response regulation, to keep inflammation under control during bacterial infection by sensing the presence of serum factors, such as the bioactive lysophospholipid (LPA) that regulate LPS-induced TNF-alpha production. However, it is also required for the optimal functions of B and T cells during adaptive immune responses by regulating cAMP synthesis in both B and T cells (PubMed:20505140). {ECO:0000250|UniProtKB:P51828, ECO:0000269|PubMed:18541530, ECO:0000269|PubMed:20505140, ECO:0000269|PubMed:23178822, ECO:0000305|PubMed:18541530, ECO:0000305|PubMed:23178822, ECO:0000305|PubMed:23229509}. |
ATP-binding;cAMP biosynthesis;Glycoprotein;Lyase;Magnesium;Manganese;Membrane;Metal-binding;Nucleotide-binding;Reference proteome;Repeat;Transmembrane;Transmembrane helix |
|
|
mmu:11513; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; adenylate cyclase activity [GO:0004016]; ATP binding [GO:0005524]; metal ion binding [GO:0046872]; activation of adenylate cyclase activity [GO:0007190]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; cAMP biosynthetic process [GO:0006171]; cellular response to ethanol [GO:0071361]; cellular response to lithium ion [GO:0071285]; intracellular signal transduction [GO:0035556]; maternal process involved in female pregnancy [GO:0060135]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; regulation of adaptive immune response [GO:0002819] |
18541530_AC7 is a specific downstream effector of the G(12/13) pathway 20505140_Adenylyl cyclase 7 controls the extent of immune responses toward bacterial infection and is required for optimal functions of B and T cells during adaptive immune responses. It regulates cyclic adenosine monophosphate (cAMP) synthesis in B and T cells. 36613790_Role of Adenylyl Cyclase Type 7 in Functions of BV-2 Microglia. |
ENSG00000121281 |
ADCY7 |
379.219868 |
0.0920390798 |
-3.441610 |
0.78600999 |
15.584829 |
0.00007888497296432754005580251233098465490911621600389480590820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004792883558805891811190058593439289325033314526081085205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
62.755317872852 |
35.9071250991642 |
681.8333909672 |
285.285497336434 |
| ENSMUSG00000031698 |
213435 |
Mylk3 |
protein_coding |
Q3UIZ8 |
FUNCTION: Kinase that phosphorylates MYL2 in vitro (PubMed:18202317, PubMed:17885681). Has been proposed to be calmodulin-dependent (PubMed:17885681), although MYL2 phosphorylation has also been observed in the presence or absence of calmodulin (PubMed:18202317). Promotes sarcomere formation in cardiomyocytes and increases cardiomyocyte contractility (By similarity). {ECO:0000250|UniProtKB:E9PT87, ECO:0000269|PubMed:17885681, ECO:0000269|PubMed:18202317}. |
Alternative splicing;ATP-binding;Cytoplasm;Kinase;Magnesium;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:213435; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; ATP binding [GO:0005524]; calmodulin-dependent protein kinase activity [GO:0004683]; myosin light chain kinase activity [GO:0004687]; cardiac myofibril assembly [GO:0055003]; cellular response to interleukin-1 [GO:0071347]; positive regulation of sarcomere organization [GO:0060298]; protein phosphorylation [GO:0006468]; regulation of vascular permeability involved in acute inflammatory response [GO:0002528]; sarcomere organization [GO:0045214]; sarcomerogenesis [GO:0048769] |
15467390_Ubiquitously expressed smooth muscle MLCK activity in MLCK null mice may be sufficient, but is not necessary for cytokinesis and early morphogenesis; other Ca2+-dependent kinases can phosphorylate myosin light-chain (20) and induce contraction in embryos. 15680356_MLCK is involved in egg polarity because inhibitors prevent sperm chromatin-induced cortical reorganization. 15988034_Inhibiting MLCK increases the number of apoptotic cells and retards the growth of mammary cancer cells in mice. 16299103_Myosin light chain phosphorylation by the dedicated skeletal muscle Ca(2+)/calmodulin-dependent MLCK is a primary biochemical mechanism for tension potentiation due to repetitive stimulation in fast-twitch skeletal muscle. 16672772_MLCK may be a regulatory target of Ca(2+)/calmodulin and may play an important role in insulin-stimulated glucose transport in adipocytes. 18066127_MLCK is involved in the activation process of nonselective cationic currents by muscarinic stimulation in murine ileal myocytes 18202317_characterization of cardiac MKLCK; both overexpression and knockdown of cardiac MLCK in cultured cardiomyocytes revealed that cardiac MLCK is likely a new regulator of MLC2 phosphorylation, sarcomere organization, and cardiomyocyte contraction 20943660_cMLCK appears to be the predominant protein kinase that maintains basal RLC phosphorylation that is required for normal physiological cardiac performance in vivo 21305056_guanylyl cyclase C signaling plays a protective role in the integrity of the intestinal mucosal barrier by regulating myosin light chain kinase activation and tight junction disassembly. 21670231_Nonmuscle myosin light-chain kinase contributes to atherosclerosis by regulating endothelial barrier function and monocyte migration via mechanisms involving not only kinase-mediated MLC phosphorylation but also Src activation. 22529961_MLCK-dependent MLC phosphorylation mediates intestinal epithelial barrier dysfunction after severe burn injury. 23362260_analysis of differential contributions of MLCK to signaling in smooth muscles 23555849_nmMLCK has a critical role in thrombin-induced endothelial cell inflammation and lung PMN infiltration 23656735_these studies show that the IL-1beta-induced increase in intestinal tight junction permeability was regulated by p38 kinase activation of ATF-2 and by ATF-2 regulation of MLCK gene activity 24116218_Calcium regulation of MLCK facilitates GLUT4-mediated glucose uptake in 3T3-L1 adipocytes. 24481113_MLCK may regulate the structure and the motility of stereocilia through F-actin polymerization. 24520376_Silencing TNFR2, but not TNFR1, resulted in restoration of epithelial tight junction (TJ) associated with decreased MLCK expression. 24911373_epithelial MLCK-activated brush border fanning by IFN-gamma promotes adherence and internalization of normally noninvasive enteric bacteria 26146960_Data suggest that galectin-3 in hepatocellular carcinoma cells promotes cell migration by inducing RhoA GTPase activity, activating Mlck, up-regulating phosphorylation of MLC2 (myosin light chain 2), and inducing actin rearrangement. 26802366_This study demonstrated that catchlike property during dynamic performance in mouse hindlimb muscles with (wild-type) and without MLCK(-/-) ) the primary mechanism for PTP (myosin phosphorylation). 27025239_Ablation of cardiac Mylk3 suggests that Mylk3 plays a pivotal role in the transition from compensated to decompensated hypertrophy via cardiac myocyte atrophy and sarcomeric disorganization. 28114272_that MLCK is competing with centrally active ROCK for a limiting pool of monomer with which to drive lamellar filament assembly 28405145_Acanthopanax senticosus polysaccharide pretreatment may be associated with inhibition of the NF-kappaB/MLCK pathway and concomitant amelioration of LPS-induced tight junction dysfunction of intestinal epithelium in endotoxemia 28912435_Pharmacological study revealed that lamellipodia formation mediated by arp2/3 and contractility regulated by myosin light chain kinase (MLCK) were responsible for the intriguing turning behavior of T cells climbing the ramp-like structures. 32213617_Mylk3 null C57BL/6N mice develop cardiomyopathy, whereas Nnt null C57BL/6J mice do not. 34106787_A temporal Ca(2+) desensitization of myosin light chain kinase in phasic smooth muscles induced by CaMKKbeta/PP2A pathways. |
ENSG00000140795 |
MYLK3 |
23.607794 |
2.7391932702 |
1.453751 |
0.52118620 |
7.714890 |
0.00547672102351149346632919190369648276828229427337646484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0200895655613315890497538873660232638940215110778808593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
35.7188553941289 |
13.1862595817873 |
13.0399179138958 |
3.75506705378311 |
| ENSMUSG00000031737 |
54352 |
Irx5 |
protein_coding |
Q9JKQ4 |
FUNCTION: Establishes the cardiac repolarization gradient by its repressive actions on the KCND2 potassium-channel gene. Required for retinal cone bipolar cell differentiation. May regulate contrast adaptation in the retina and control specific aspects of visual function in circuits of the mammalian retina. Involved in craniofacial and gonadal development (By similarity). Modulates the migration of progenitor cell populations in branchial arches and gonads by repressing CXCL12. {ECO:0000250}. |
DNA-binding;Homeobox;Nucleus;Phosphoprotein;Reference proteome;Sensory transduction;Transcription;Vision |
|
|
mmu:54352; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; cell development [GO:0048468]; embryonic cranial skeleton morphogenesis [GO:0048701]; gonad development [GO:0008406]; neuron differentiation [GO:0030182]; neuron maturation [GO:0042551]; regulation of gene expression [GO:0010468]; regulation of heart rate [GO:0002027]; regulation of transcription by RNA polymerase II [GO:0006357]; response to stimulus [GO:0050896]; retinal bipolar neuron differentiation [GO:0060040]; visual perception [GO:0007601] |
16182275_Regulates the development of type 2 and type 3 cone bipolar cells independently of visual system homeobox 1 (Vsx-1). 16239150_Irx5 establishes the cardiac ventricular repolarization gradient. 18322081_Iroquois related homeobox 5 (Irx5) transcription factor controls specific aspects of visual function in circuits of the mammalian retina. 22992950_postnatal Irx3 activity can be repressed by Irx5 24726282_Irx3 and Irx5 (Irx3/5) are essential in the initiating limb bud to specify progenitors of the femur, tibia, and digit 1. 30071018_Irx3 and Irx5 first work together to define the pregranulosa cell population of germline nests. During primordial follicle formation, they transition to oocyte- and granulosa cell-specific expression patterns where they cooperate in neighboring cells to build the foundation for follicle integrity. 30246301_Promotion of IRX3 signalling or inhibition of IRX5 signalling could be a route towards differentiation therapy for Wilms tumour, in which WNT5A is a candidate molecule for enforced tubular maturation. 30538277_Studied role of Iroquois homeobox 5 (Irx5) in adipocytes, amyloid precursor protein, and mitochondrial respiration in obesity mouse models and patients. 32108023_Canonical Wnt/beta-catenin activity and differential epigenetic marks direct sexually dimorphic regulation of Irx3 and Irx5 in developing mouse gonads. 32507881_IRX3 and IRX5 collaborate during ovary development and follicle formation to establish responsive granulosa cells in the adult mousedagger. 32662900_IRX3 and IRX5 Inhibit Adipogenic Differentiation of Hypertrophic Chondrocytes and Promote Osteogenesis. 32907847_IRX3/5 regulate mitotic chromatid segregation and limb bud shape. 33859429_Irx3 and Irx5 in Ins2-Cre(+) cells regulate hypothalamic postnatal neurogenesis and leptin response. 34083488_Extensive pleiotropism and allelic heterogeneity mediate metabolic effects of IRX3 and IRX5. 35245131_Irx5 and transient outward K(+) currents contribute to transmural contractile heterogeneities in the mouse ventricle. 36115924_Deficiency of Irx5 protects mice from obesity and associated metabolic abnormalities. |
ENSG00000176842 |
IRX5 |
1785.911860 |
0.3683910938 |
-1.440690 |
0.07360749 |
376.030684 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000910239040273915473366711245159620259346863250337273991091360760026084867125539424103634591036892856648286354340454435590218468365415552993390783501338732270587399159674045200782169 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000037472327193114271911793660955611605322345017231362896432916731261005155687797575282313902723037684404412365372420048105369060745750027551823545539973688783948362148230657512636710 |
Yes |
No |
960.953465156126 |
49.0499107205738 |
2608.51437832937 |
95.7720751615311 |
| ENSMUSG00000031750 |
76527 |
Il34 |
protein_coding |
Q8R1R4 |
FUNCTION: Cytokine that promotes the proliferation, survival and differentiation of monocytes and macrophages. Promotes the release of pro-inflammatory chemokines, and thereby plays an important role in innate immunity and in inflammatory processes. Plays an important role in the regulation of osteoclast proliferation and differentiation, and in the regulation of bone resorption. Signaling via CSF1R and its downstream effectors stimulates phosphorylation of MAPK1/ERK2 AND MAPK3/ERK1 (By similarity). {ECO:0000250}. |
3D-structure;Alternative splicing;Cytokine;Glycoprotein;Growth factor;Immunity;Inflammatory response;Innate immunity;Reference proteome;Secreted;Signal |
|
|
mmu:76527; |
extracellular space [GO:0005615]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; identical protein binding [GO:0042802]; macrophage colony-stimulating factor receptor binding [GO:0005157]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; interleukin-34-mediated signaling pathway [GO:0061514]; microglial cell proliferation [GO:0061518]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of gene expression [GO:0010628]; positive regulation of macrophage chemotaxis [GO:0010759]; positive regulation of macrophage differentiation [GO:0045651]; positive regulation of macrophage proliferation [GO:0120041]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of monocyte differentiation [GO:0045657]; positive regulation of oligodendrocyte differentiation [GO:0048714]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of protein tyrosine kinase activity [GO:0061098] |
20504948_The different spatiotemporal expression of IL-34 and CSF-1 allows for complementary activation of the CSF-1R in developing and adult tissues. 21494622_This study was to explore the biological function, specifically osteoclastogenesis and bone metabolism, of il-34. 21872563_The function of Interleukin-34 (IL-34), a newly discovered cytokine, on microglia was examined. 22542597_Postnatal neocortical expression showed that CSF-1 was expressed in layer VI, whereas IL-34 was expressed in the meninges and layers II-V. The broader expression of IL-34 is consistent with its previously implicated role in microglial development. 22579672_Differentiated signaling between IL-34 and CSF-1 is likely achieved by the relative thermodynamic independence of IL-34 versus negative cooperativity of CSF-1 at the CSF-1 receptor recognition sites. 22729249_IL-34 specifically directs the differentiation of myeloid cells in the skin epidermis and central nervous system. 22985514_These findings suggest that TGF-beta produced by IL-34-treated microglia is a negative regulator of microglial proliferation and enhances the neuroprotective property of microglia. 23177320_IL-34 as a nonredundant cytokine for the development of Langerhans cells during embryogenesis as well as for their homeostasis in the adult skin. 24970360_Tumor necrosis factor-alpha induces IL-34 expression via NF-kappaB in MC3T3-E1 osteoblastic cells. 25471534_In vitro and in vivo experiments indicate that IL-34 expression is regulated by TNF-a and IL-1b and that its overexpression is associated with an increase in osteosarcoma growth and metastasis. 25535736_IL-34 protected blood-brain barrier integrity by restored expression levels of tight junction proteins, which were downregulated by pro-inflammatory cytokines. 25896238_the expression pattern of IL-34 in ileum and colon and suggest IL-34 as a new modulator of inflammation in inflammatory bowel disease 26121749_IL-34-dependent, Mo-mediated, CSF-1 nonredundant mechanisms promote persistent ischemia-incited AKI that worsens subsequent CKD. 26146640_constitutive IL-34 expressed by skin keratinocytes might suppress resident macrophage responses to C. albicans colonisation by maintaining low levels TLR2 and Dectin-1 expression by macrophages. 26634935_study concludes that Langerhans cells require IL-34 when residing in fully differentiated and anatomically intact skin epidermis, but rely on neutrophil-derived CSF1 during inflammation 27865758_The current study aimed to assess the IL-34 expression in response to two members of the transforming growth factor (TGF)-beta family, TGF-beta1 and bone morphogenetic protein (BMP)-2, in synovial fibroblasts from rheumatoid arthritis patients. 29664689_The IL-34 acts as a proinflammatory factor, aggravating the severity of arthritis in CIA mice by inducing the production of IL-17. 30573681_These results indicated a novel GRP78-dependent localization and specific function of IL-34 in FL-Y cells related to monocytic cell differentiation. 30782613_This study describes for the first time the expression of IL-34 in multiple myeloma cells, indicating that it may enhance osteolysis. Targeting Il34 by specific small interfering RNA impaired osteoclast formation in vitro and attenuated osteolytic disease in vivo. 31201067_found that the loss of interleukin-34 resulted in higher sensitivity to Con A-induced hepatitis 31332295_Fate decision of satellite cell differentiation and self-renewal by miR-31-IL34 axis. 31552020_Function of CSF1 and IL34 in Macrophage Homeostasis, Inflammation, and Cancer. 31616414_CSF1R Ligands IL-34 and CSF1 Are Differentially Required for Microglia Development and Maintenance in White and Gray Matter Brain Regions. 32578004_Interleukin-34 contributes to poor prognosis in triple-negative breast cancer. 32765504_Effects of IL-34 on Macrophage Immunological Profile in Response to Alzheimer's-Related Abeta42 Assemblies. 34138470_Interleukin-34 accelerates intrauterine adhesions progress related to CX3CR1(+) monocytes/macrophages. 34582356_Bone morphogenetic protein 2 controls steroid-induced osteonecrosis of the femoral head via directly inhibiting interleukin-34 expression. 35200127_Il-34 regulates MAPKs, PI3K/Akt, JAK and NF-kappaB pathways and induces the expression of inflammatory factors in RA-FLS. 35288653_IL-34 and protein-tyrosine phosphatase receptor type-zeta-dependent mechanisms limit arthritis in mice. 36030499_IL-34 deficiency impairs FOXP3(+) Treg function in a model of autoimmune colitis and decreases immune tolerance homeostasis. |
ENSG00000157368 |
IL34 |
59.196524 |
0.4837484613 |
-1.047671 |
0.40122950 |
6.772466 |
0.00925748433980543190824352706158606451936066150665283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0313773924040757282161173691292788134887814521789550781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
35.3981466568708 |
9.97201569423612 |
73.1746961291087 |
15.0625503490804 |
| ENSMUSG00000031762 |
17750 |
Mt2 |
protein_coding |
P02798 |
FUNCTION: Metallothioneins have a high content of cysteine residues that bind various heavy metals; these proteins are transcriptionally regulated by both heavy metals and glucocorticoids. |
Acetylation;Direct protein sequencing;Metal-binding;Metal-thiolate cluster;Phosphoprotein;Reference proteome |
|
|
mmu:17750; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleus [GO:0005634]; cadmium ion binding [GO:0046870]; metal ion binding [GO:0046872]; zinc ion binding [GO:0008270]; cellular response to cadmium ion [GO:0071276]; cellular response to copper ion [GO:0071280]; cellular response to zinc ion [GO:0071294]; cellular zinc ion homeostasis [GO:0006882]; detoxification of copper ion [GO:0010273]; negative regulation of growth [GO:0045926]; nitric oxide mediated signal transduction [GO:0007263]; response to bacterium [GO:0009617] |
11771944_Reduces inflammatory response, oxidative stress and apoptosis and increases angiogenesis following focal brain cryolesion 11850030_Zn-MTs(I + II) are crucial in zinc homeostasis for endocrine-immune efficiency during the entire life assuming a role of potential and novel 'biological clock of ageing' 11891201_MT-null phenotype precludes lead-induced inclusion body formation and increases lead toxicity at the organ and cellular level despite reducing lead accumulation 11950277_MT-2 deficiency increases brain pathology in transgenic mice with astrocyte-targeted expression of interleukin 6. 14499492_High metallothionein (MT) has harmful role in ageing, Lowering zinc ion bioavailability levels required for immune efficiency and increasing c-myc expression. Immune depression and enhanced c-myc, via high MT, may cause age-related degenerative diseases. 14536000_Mice lacking MT are more susceptible to H. pylori colonization and gastric inflammation, indicating that MT may be protective against H. pylori-induced gastritis. 14617770_endogenously via iNOS, synthesized NO increases the constitutive mRNA expression of both MT-1 and MT-2 genes 15306638_MDMA-induced toxicity to dopamine neurons is associated with increased Mt1 and Mt2 gene transcription and translation 15592854_In mouse spinal cord injury lead the up-regulation MT-2 expression at 1 day and MT-2 expression was observed in activated macrophages/microglia at 1 day. 16133945_MTs directly or indirectly influence the radial equilibrium of large myelinated motor axons 16166738_Data show that metallothioneins 1 and 2 ultimately improve survival in the progression of acute lung injury in mice. 16243847_MT2a has a role in oxidative stress, and the formation of MT disulfide bonds is involved in the regulation of zinc homeostasis 16387980_results suggest that MT protects against coagulatory and fibrinolytic disturbance and multiple organ damages induced by LPS, at least partly, via the inhibition of the expression of proinflammatory proteins 16563532_These results suggest that Zn alone does not completely exert an anti-oxidant effect against Cu and Fe toxicity, but that induction of MT is necessary. 17031522_neurodegenerative diseases as well as metabolic stress enhance the metallothionein I/II expression in perivascular regions of brain cerebral cortex, predominantly in astrocytes 17409522_Mt-2 synthesis was induced by cilostazol. 17624681_These findings indicate that MTs are induced, and may be neuroprotective against neuronal damage, after MCAO. Furthermore, EPO is neuroprotective in vivo during permanent MCAO, and this may be at least partly mediated by MTs. 18083145_Metallothionein II deficient mice exhibit impaired rate of maze learning but retained the information as well as the wild-type mice. The absence of functional MT-II does not result in significantly worse injury following 45 min of hypoxia-ischemia. 18226494_During the early phase of learning metallothionein-2 null mice were more susceptible to mercury-induced impairment compared to wildtype mice 18239062_Suggest that MT1/2 in the gastric mucosa might play an important role in the protection against H. pylori-induced gastric ulceration. 18449439_Results suggest MT-1/MT-2 protect against coagulatory disturbances associated with acute liver injury. 18579766_MT-1 and -2 alone are not sufficient for protecting photoreceptors against hyperbaric oxygen-induced cell death. 19828842_Metallothionein deficiency leads to soleus muscle contractile dysfunction following acute spinal cord injury in mice. 20144643_MT-l/II null cardiomyocytes were more sensitive to butenolide-induced cytotoxicity than wild-type cardiomyocytes 20219986_preventive role against high-fat diet-induced obesity by regulating adipocyte enlargement and leptin signaling 20357188_Induction of metallothionein-I and -II occurs in the retinitis pigmentosa models studied and correlates with glial activation 20798270_Knockdown of metallothionein 2 does not affect atrophy or oxidant activity in skeletal myotubes subject to serum reduction. 21913972_the absence of MT1 and MT2 has no unfavorable effect on ischemic brain injury 22152221_Data suggest that metallothionein (MT)-I/II(-/-) mice have altered immune function and provide a new hypothesis that this alteration is partly responsible for the differences observed in MT-I/II(-/-) mice after brain injury relative to wild type mice. 22363575_MT-I/II is up-regulated in the liver after brain injury and modulates the amount of zinc that is sequestered to the liver. 22395140_Down-regulation of MT1 and MT2 after electromagnetic fields in mouse testis might be followed by some consequences that result in infertility. 22766972_MT1 + 2 effectively alters the amyloid cascade, survival, body weight, and behavior in a complex manner 22917840_Zn deficiency in utero induces fetal epigenetic alterations of Mt2 gene and these changes are being stored as an epigenetic memory until adulthood. 23467591_These data indicate that metallothioneins play an important role in the prevention of colonic mucosal inflammation in a mouse model of dextran sulfate sodium-induced colitis. 23630250_Metallothioneins act as negative regulators for IL-27-induced Tr1 cells. 23880315_these results demonstrate that MT-I/II knockout mice are more vulnerable to cadmium-induced fetal growth retardation. 23925449_the metal binding abilities of mouse MT2 (mMT2) with divalent (Zn(II) and Cd(II)) and monovalent (Cu(I)) ions were analyzed and compared to those of the mouse MT1 (mMT1) isoform. 24154695_The results of this study suggest that metallothioneins may play a key role in protecting the kidney against high glucose-induced ROS and subsequent inflammation in diabetic nephropathy. 25485709_Metallothionein blocks oxidative DNA damage induced by acute inorganic arsenic exposure 25501601_The MT2 receptor activates Akt/GSK-3beta/CRMP-2 signaling and is necessary and sufficient to mediate functional axonogenesis and synaptic formation in central neurons. 25801306_Zinc-binding MT1 and MT2 are key modulators of Fc epsilonRI-induced basophil production of IL-4. 25871729_Enhanced senescence due to absence of metallothionein genes may shorten lifespan. 26148447_MT1/2 deficiency aggravated depleted uruanium-induced nephrotoxicity, and the molecular mechanisms appeared to be related to the increased oxidative stress and apoptosis, and decreased SGLT expression. 26833479_Metallothioneins may have a protective role against post-burn inflammation and inflammatory organ damage, at least partly through inhibiting the p38 MAPK signaling. 27251638_these results demonstrate that MT2 plays a role in CD4+ T cell activation by transducing reactive oxygen species signals into an increased [Zn2+]i that subsequently affects downstream effector function 27403038_Cardioprotective effects of MT1/2 seem to be exerted via modulation of contractile elements, antioxidative enzymes, inflammatory response, and myocardial remodeling. 27956698_blockade of metallothioneins 1 and 2 constitutes a promising approach for the treatment of conditions which result in muscle atrophy. 30025861_Cuprizone-induced demyelination is followed by high upregulation of MT-I/II and megalin immunoreactivity in astrocytes and epithelial and endothelial cells located in cerebral and cerebellar white and gray matter, as well as in choroid plexus and in numerous constituents of the neurogenic niches in subgranular zone (SGZ) of DG and in SVZ of lateral ventricles. 31104354_Cardiac-specific overexpression of metallothionein attenuates L-NAME-induced myocardial contractile anomalies and apoptosis. 32398691_Metallothioneins regulate ATP7A trafficking and control cell viability during copper deficiency and excess. |
|
|
1246.366983 |
2.0726926068 |
1.051506 |
0.10182915 |
105.690227 |
0.00000000000000000000000086211532844645524943922299195302483200791261870645112091220652278476116459060563101957086473703384399414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000464019105402699880289382686933579097329430406746271526669534433252872762665219852351583540439605712890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1698.33604210365 |
119.972510122202 |
819.386355943819 |
44.0999847762142 |
| ENSMUSG00000031785 |
14766 |
Adgrg1 |
protein_coding |
Q8K209 |
FUNCTION: Receptor involved in cell adhesion and probably in cell-cell interactions. Mediates cell matrix adhesion in developing neurons and hematopoietic stem cells. Receptor for collagen III/COL3A1 in the developing brain and involved in regulation of cortical development, specifically in maintenance of the pial basement membrane integrity and in cortical lamination (PubMed:21768377). Binding to the COL3A1 ligand inhibits neuronal migration and activates the RhoA pathway by coupling to GNA13 and possibly GNA12 (By similarity). Plays a role in the maintenance of hematopoietic stem cells and/or leukemia stem cells in bone marrow niche (PubMed:23478665). Plays a critical role in tumourigenesis (By similarity). Plays essential role in testis development (PubMed:20981830). {ECO:0000250|UniProtKB:Q9Y653, ECO:0000269|PubMed:18378689, ECO:0000269|PubMed:18509043, ECO:0000269|PubMed:19515912, ECO:0000269|PubMed:20981830, ECO:0000269|PubMed:21768377, ECO:0000269|PubMed:23478665, ECO:0000269|PubMed:24531968}. |
3D-structure;Cell adhesion;Cell membrane;Developmental protein;Differentiation;G-protein coupled receptor;Glycoprotein;Heparin-binding;Membrane;Neurogenesis;Receptor;Reference proteome;Secreted;Signal;Transducer;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:14766; |
extracellular region [GO:0005576]; glial limiting end-foot [GO:0097451]; membrane raft [GO:0045121]; plasma membrane [GO:0005886]; collagen binding [GO:0005518]; extracellular matrix binding [GO:0050840]; G protein-coupled receptor activity [GO:0004930]; heparin binding [GO:0008201]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; angiogenesis [GO:0001525]; brain development [GO:0007420]; cell adhesion [GO:0007155]; cell migration [GO:0016477]; cell surface receptor signaling pathway [GO:0007166]; cerebral cortex radial glia-guided migration [GO:0021801]; cerebral cortex regionalization [GO:0021796]; hematopoietic stem cell homeostasis [GO:0061484]; layer formation in cerebral cortex [GO:0021819]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of neuron migration [GO:2001223]; neural precursor cell proliferation [GO:0061351]; positive regulation of cell adhesion [GO:0045785]; positive regulation of neural precursor cell proliferation [GO:2000179]; positive regulation of Rho protein signal transduction [GO:0035025]; protein kinase C signaling [GO:0070528]; Rho protein signal transduction [GO:0007266]; seminiferous tubule development [GO:0072520]; vascular endothelial growth factor production [GO:0010573] |
15044805_preferential expression in neuronal progenitor cells of the cerebral cortical ventricular and subventricular zones duing periods of neurogenesis 16888225_potential pleiotrophic role of secretin during embryonic development 17008357_These findings suggest that the secretin receptor system has an important role in the central nervous system relating to social behavior 17283064_Mutation of the SCTR gene could lead to mild polydipsia and polyuria 17576745_mutations in GPR56 impair trafficking of the mutant protein to the plasma membrane, thus providing insights into how BFPP-associated mutations affect GPR56 function 17932623_These results define the biochemical properties of GPR56 protein, and suggest that the expression of GPR56 protein is suppressed in human pancreatic cancer cells. 18378689_results indicate that GPR56 participates in the regulation of neural progenitor cells movement through the Galpha(12/13) and Rho signaling pathway, suggesting its important role in the development of the central nervous system 18509043_GPR56 regulates pial basement membrane integrity and cortical lamination. 19515912_These studies establish a novel role for GPR56 in the adhesion of developing neurons to basal lamina molecules and suggest that this adhesion is critical for maintenance of the pia and proper cerebellar morphogenesis. 20981830_Data suggest that GPR56 might act to establish a spatial and/or temporal cue for asymmetric cord remodeling during male gonad development. 21349848_GPR56 mutations cause bilateral frontoparietal polymicrogyria via multiple mechanisms 21768377_Data show that that Col3a1 null mutant mice exhibit overmigration of neurons beyond the pial basement membrane and a cobblestone-like cortical malformation similar to the phenotype seen in Gpr56 null mutant mice. 22351047_We revealed that GPR56 is expressed in multiple cell types in the preplate, marginal zone, subventricular zone, and ventricular zone in the developing cerebral cortex 23874761_GPR56 functions together with alpha3beta1 integrin in regulating cerebral cortical development. 24356421_Role for TG2 in GPR56-mediated melanoma inhibition. The uncovered antagonistic relationship between GPR56 and TG2 proposes a mechanism by which extracellular matrix accumulation/crosslinking in tumors may be reversed. 24949629_adhesion G protein-coupled receptor GPR56-mediated RhoA activation induced by collagen III stimulation 25336758_These data illustrate a signaling pathway through GPR56 which regulates muscle hypertrophy associated with resistance/loading-type exercise. 25547674_Data show that Gpr56, a G-coupled protein receptor, is required for hematopoietic cluster formation during transdifferentiation process in endothelial to hematopoietic cell transition (EHT). 25607655_GPR56 is a cell-autonomous regulator of oligodendrocyte development 25607772_Gpr56 regulates oligodendrocyte development via interactions with Galpha12/13 and RhoA 25840412_Although GPR56 is abundantly and selectively expressed by primitive HSPCs, its high level expression is largely dispensable for steady-state and regenerative hematopoiesis. 26116532_we knocked down GPR56 in cardiomyocytes and found that GPR56 promoted Ang II-induced cardiomyocyte hypertrophy and it contributed to PCBP2 effects on cardiomyocyte hypertrophy 27063597_knockdown of Gpr56 delayed onset of HOXA9/MEIS1-induced AML 27657451_determine the crystal structure of the GPR56 extracellular domain. 28380450_Deletion of G-protein coupled receptor 56 (GPR56) in the mouse germline abrogated progastrin-dependent colonic mucosal proliferation and increased apoptosis. 29225194_all hematopoietic stem cells (HSCs) in mouse bone marrow mononuclear cells express GPR56 protein on their surface and that GPR56 is a positive marker for HSCs. 29367382_Gpr56 as a new regulator in the development and maintenance of peripheral myelin. 29809138_Microglial transglutaminase-2 drives myelination and myelin repair via ADGRG1 in oligodendrocyte precursor cells. 29855662_Collagen III protected beta-cells from cytokine-induced apoptosis, triggered increases in [Ca(2+)]i and potentiated glucose-induced insulin secretion from WT islets but not from Gpr56(-/-) islets. Deletion of GPR56 did not affect glucose-induced insulin secretion in vitro and it did not impair glucose tolerance in adult mice 31304602_Adhesion G-protein coupled receptor 56 is required for 3T3-L1 adipogenesis. 32242018_Gpr56 knockdown in mouse prefrontal cortex is associated with depressive-like behaviors. 32452062_A splicing isoform of GPR56 mediates microglial synaptic refinement via phosphatidylserine binding. 33097663_GPR56/ADGRG1 is a platelet collagen-responsive GPCR and hemostatic sensor of shear force. 33560396_Unexpected redundancy of Gpr56 and Gpr97 during hematopoietic cell development and differentiation. 34585770_Phospholipid-flippase chaperone CDC50A is required for synapse maintenance by regulating phosphatidylserine exposure. 34632664_Crucial contribution of GPR56/ADGRG1, expressed by breast cancer cells, to bone metastasis formation. 34685654_The Inhibitory Receptor GPR56 (Adgrg1) Is Specifically Expressed by Tissue-Resident Memory T Cells in Mice But Dispensable for Their Differentiation and Function In Vivo. 36228547_Adgrg1 is a new transcriptional target of Hand1 during trophoblast giant cell differentiation. 36336959_GPR56 S4 variant is required for microglia-mediated synaptic pruning. 36453010_GPR56 C-terminal fragment mediates signal received by N-terminal fragment of another adhesion GPCR Latrophilin1 in neurons. |
ENSG00000205336 |
ADGRG1 |
39.204316 |
2.4366955672 |
1.284926 |
0.43165563 |
8.736345 |
0.00311929196399206472289922231766468030400574207305908203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0123796391859111853256392166144905786495655775070190429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
55.9005612386602 |
16.9761642294003 |
22.94113470361 |
5.35174043883452 |
| ENSMUSG00000031841 |
12554 |
Cdh13 |
protein_coding |
Q9WTR5 |
FUNCTION: Cadherins are calcium-dependent cell adhesion proteins. They preferentially interact with themselves in a homophilic manner in connecting cells; cadherins may thus contribute to the sorting of heterogeneous cell types. May act as a negative regulator of neural cell growth. |
3D-structure;Calcium;Cell adhesion;Cell membrane;Cleavage on pair of basic residues;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Metal-binding;Reference proteome;Repeat;Signal |
|
This gene encodes a member of the cadherin family of calcium-dependent glycoproteins that mediate cell adhesion and regulate many morphogenetic events during development. The encoded preproprotein is further processed to generate a mature protein. This gene is highly expressed in the vasculature including endothelial cells, smooth muscle cells and pericytes, where the encoded protein binds to adiponectin and has been implicated in the modulation of angiogenesis. Multiple distinct genes of the cadherin family, including this gene, are found on chromosome 8. [provided by RefSeq, Nov 2015]. |
mmu:12554; |
catenin complex [GO:0016342]; caveola [GO:0005901]; cytoplasm [GO:0005737]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; GABA-ergic synapse [GO:0098982]; neuron projection [GO:0043005]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; synapse [GO:0045202]; adiponectin binding [GO:0055100]; cadherin binding [GO:0045296]; calcium ion binding [GO:0005509]; identical protein binding [GO:0042802]; lipoprotein particle binding [GO:0071813]; low-density lipoprotein particle binding [GO:0030169]; calcium-dependent cell-cell adhesion via plasma membrane cell adhesion molecules [GO:0016339]; cell-cell adhesion via plasma-membrane adhesion molecules [GO:0098742]; endothelial cell migration [GO:0043542]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156]; keratinocyte proliferation [GO:0043616]; lamellipodium assembly [GO:0030032]; localization within membrane [GO:0051668]; low-density lipoprotein particle mediated signaling [GO:0055096]; negative regulation of cell adhesion [GO:0007162]; negative regulation of cell population proliferation [GO:0008285]; positive regulation of calcium-mediated signaling [GO:0050850]; positive regulation of cell migration [GO:0030335]; positive regulation of cell-matrix adhesion [GO:0001954]; positive regulation of endothelial cell proliferation [GO:0001938]; positive regulation of positive chemotaxis [GO:0050927]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of transcription by RNA polymerase II [GO:0045944]; Rac protein signal transduction [GO:0016601]; regulation of endocytosis [GO:0030100]; regulation of epidermal growth factor receptor signaling pathway [GO:0042058]; Rho protein signal transduction [GO:0007266]; sprouting angiogenesis [GO:0002040] |
12060406_Expression of T-cadherin in Basal keratinocytes of skin. 12509455_T-cadherin has a role in growth regulation involving p21(CIP1/WAF1) expression and G2 arrest 15210937_Because T-cadherin is a glycosylphosphatidylinositol-anchored extracellular protein, it may act as a coreceptor for an as-yet-unidentified signaling receptor through which adiponectin transmits metabolic signals. 16873731_T-cadherin is novel modulator of angiogenesis. Molecule can be exploited as target for modulation of therapeutic angiogenesis, as well as for prevention of pathological conditions associated with abnormal neovascularization. 17486418_T-cadherin suppresses angiogensis in vivo by inhibiting migration of endothelial cells. 18316604_the vascular T-cadherin-adiponectin association may contribute to the molecular cross-talk between tumor cells and the stromal compartment in breast cancer 18635739_both cellular and acellular presentation of T-cadherin can be used to modulate the hepatocyte phenotype in vitro for tissue engineering applications. 19463088_T-cadherin transcriptionally activated Rac1 and Cdc42. 20873143_hyperexpression of T-cadherin in the B16F10 cells suppresses the proliferation of these cells in vitro and the growth of the tumor masses formed by melanoma cells on the chorioallantoic membrane and their neovascularization 21041950_T-cadherin protects from stress-induced pathological cardiac remodeling by binding APN and activating its cardioprotective functions. 21766307_T-cad can exert pleiotropic effects on squamous cell carcinoma progression; up- or down-regulation of T-cad can promote tumour expansion in vivo. 21975561_T-cadherin is a component of insulin granules, suggesting that it contributes to the regulation of insulin secretion independently of direct interactions with adiponectin. 22815927_T-cad does not mediate the protective effects of adiponectin in allergic airways responses in mice 23824191_These data highlight a previously unrecognized role for T-cadherin in limb revascularization and show that it is essential for mediating the vascular actions of adiponectin. 25514086_These data show that both circulating and tissue-bound Adipo levels are dependent on Tcad and, in reverse, regulate tissue Tcad levels through a positive feedback loop. 26460479_CDH13 is a negative regulator of inhibitory synapses in the hippocampus, and provide insights into how CDH13 dysfunction may contribute to the excitatory/inhibitory imbalance observed in Attention Deficit Disorder with Hyperactivity. 28062540_T-cadherin was essential for accumulation of adiponectin in the neointima and atherosclerotic plaque lesions, and the adiponectin-T-cadherin association protected against vascular injury. 28103886_T-cadherin deficiency causes endothelial dysfunction in Type II Diabetes Mellitus. 28325833_a unique key feature of the T-cad prodomain is its involvement in binding of the T-cad repeats 1 and 2 to adiponectin; adiponectin positively regulates T-cad abundance 28386779_Studies findings identified a novel protective role for cadherin-13 in cortical neuron development. 28392425_using rat and murine aortic smooth muscle cells as experimental models, we surveyed the ability of T-cadherin to regulate autophagy in SMCs during serum-starvation stress. Overall our findings have identified T-cadherin as a novel positive regulator of autophagy and survival in smooth muscle cells 29446202_Findings show that Cdh13 is critical for inhibitory function of Golgi cells, and that GlyT2::Cre-mediated deletion of Cdh13 in non-executive centers of the brain, such as the cerebellum, may contribute to cognitive and social behavioral deficits linked to neurological disorders. 29467502_Studied role of cadherin 13 (CDH13) levels in adipose tissue and obesity; found CDH13 levels are affected by obesity in mouse models and humans and are restored by weight loss in humans. 30165120_Neonatal maternal separation (MS) lead to increased anxiety-like behavior in Cdh13(-/-) mice compared to the other two MS groups. Cdh13(-/-) mice showed a context-dependent effect on stress- & anxiety-related behavior, impaired extinction learning following contextual fear conditioning & decreased impulsivity, & a mild decrease in errors in the Barnes maze & reduced risk-taking in the light-dark transition test after MS. 32113967_Serotonin (5-HT) neuron-specific inactivation of Cadherin-13 impacts 5-HT system formation and cognitive function. 32396872_Plasma proteomic profiling of young and old mice reveals cadherin-13 prevents age-related bone loss. 33284092_Increased vascular permeability and severe renal tubular damage after ischemia-reperfusion injury in mice lacking adiponectin or T-cadherin. |
ENSG00000140945 |
CDH13 |
30.700556 |
0.1520820738 |
-2.717078 |
0.61147874 |
21.022007 |
0.00000454038035821594075788169148077955128428584430366754531860351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000358151598220327326308962123668777621787739917635917663574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
8.25084496885556 |
3.60272702513482 |
54.2525806156485 |
13.6254065213606 |
| ENSMUSG00000031849 |
12845 |
Comp |
protein_coding |
Q9R0G6 |
FUNCTION: Plays a role in the structural integrity of cartilage via its interaction with other extracellular matrix proteins such as the collagens and fibronectin (PubMed:32686688). Can mediate the interaction of chondrocytes with the cartilage extracellular matrix through interaction with cell surface integrin receptors. Could play a role in the pathogenesis of osteoarthritis. Potent suppressor of apoptosis in both primary chondrocytes and transformed cells. Suppresses apoptosis by blocking the activation of caspase-3 and by inducing the IAP family of survival proteins (BIRC3, BIRC2, BIRC5 and XIAP) (By similarity). Essential for maintaining a vascular smooth muscle cells (VSMCs) contractile/differentiated phenotype under physiological and pathological stimuli. Maintains this phenotype of VSMCs by interacting with ITGA7 (By similarity). {ECO:0000250|UniProtKB:P35444, ECO:0000250|UniProtKB:P49747, ECO:0000269|PubMed:32686688}. |
3D-structure;Apoptosis;Calcium;Cell adhesion;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Heparin-binding;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:12845; |
collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; protein-containing complex [GO:0032991]; BMP binding [GO:0036122]; calcium ion binding [GO:0005509]; collagen binding [GO:0005518]; extracellular matrix structural constituent [GO:0005201]; fibronectin binding [GO:0001968]; heparan sulfate proteoglycan binding [GO:0043395]; heparin binding [GO:0008201]; integrin binding [GO:0005178]; protease binding [GO:0002020]; proteoglycan binding [GO:0043394]; vitamin D binding [GO:0005499]; apoptotic process [GO:0006915]; artery morphogenesis [GO:0048844]; blood coagulation [GO:0007596]; BMP signaling pathway [GO:0030509]; bone growth [GO:0098868]; bone mineralization [GO:0030282]; bone morphogenesis [GO:0060349]; cartilage development [GO:0051216]; cartilage homeostasis [GO:1990079]; chondrocyte development [GO:0002063]; chondrocyte proliferation [GO:0035988]; collagen fibril organization [GO:0030199]; endochondral bone growth [GO:0003416]; growth plate cartilage development [GO:0003417]; limb development [GO:0060173]; multicellular organism aging [GO:0010259]; multicellular organism growth [GO:0035264]; muscle cell development [GO:0055001]; musculoskeletal movement [GO:0050881]; negative regulation of apoptotic process [GO:0043066]; negative regulation of hemostasis [GO:1900047]; neuromuscular process [GO:0050905]; ossification [GO:0001503]; platelet aggregation [GO:0070527]; positive regulation of chondrocyte proliferation [GO:1902732]; protein homooligomerization [GO:0051260]; protein processing [GO:0016485]; protein secretion [GO:0009306]; regulation of bone mineralization [GO:0030500]; regulation of gene expression [GO:0010468]; response to unfolded protein [GO:0006986]; skeletal system development [GO:0001501]; skin development [GO:0043588]; tendon development [GO:0035989]; vascular associated smooth muscle cell development [GO:0097084]; vascular associated smooth muscle contraction [GO:0014829] |
12024046_Cartilage oligomeric matrix protein-deficient mice have normal skeletal development. 12837285_Comp was expressed in tendon clone cells. 15183430_Negative regulation of transcription is an important mechanism for chondrocyte-specific expression of the COMP gene 16585064_ADAMTS-7 is the first metalloproteinase found to bind directly to and degrade COMP 17307734_COMP appears to be required for granulin PC cell-derived growth factor-mediated chondrocyte proliferation 17588960_a mutation in the C-terminal domain of COMP exerts a dominant-negative effect on both intra- and extracellular processes. 18191556_contribution of COMP to the phenotype of mice deficient in both collagen IX and COMP appears minor, even though clear differences in the deposition of matrilin-3 were detected 19014566_COMP-deficient mice develop an early onset collagen-induced arthritis and more severe arthritis during the chronic phase. 19950300_Loss of type IX collagen and COMP leads to matrix aberrations that may make cartilage more susceptible to degeneration. 20033473_Data show that cartilage oligomeric matrix protein (COMP) promotes cell attachment via independent mechanisms involving cell surface CD47 and alphaVbeta3 integrin and that cell attachment to COMP induces formation of fascin-stabilized actin microspikes. 20421976_reducing steady state levels of COMP mRNA alleviates intracellular retention of other extracellular matrix proteins associated with the pseudoachondroplasia cellular pathology 22006726_A novel form of chondrocyte stress triggered by the expression of a human-like mutation in COMP is central to the pathogenesis of pseudoachondroplasia. 22378539_Lack of COMP and matrilin 3 leads to increased deposition of TIMP-3, which causes partial inactivation of matrix metalloproteinases in bone, including MMP-13. 23098652_results imply that COMP is not a key upstream mediator of the anabolic effects of ML on the skeleton. 23754310_The lack of arthritis, together with high levels of COMP-specific antibodies, in COMP-deficient mice indicates that susceptibility to arthritis is COMP specific and that endogenous expression of COMP in wild-type mice tolerizes B cells in vivo. 24312420_study will facilitate better awareness of the differential diagnoses that might be associated with the PSACH/MED spectrum and subsequent care of PSACH/MED patients 25133350_COMP immunoreactivity was observed in about half of the investigated plaques from the ApoE null mice, mainly located along the intima-medial border. Plaques in the brachiocephalic artery from ApoE mice lacking COMP were increased in size with 54%. 25995382_The accumulation and thereby the functionality of thrombospondin in extracellular matrix is controlled by concentration-dependent, intermolecular 'matrix trapping' mechanism. 26045608_COMP deficiency shortened tail-bleeding and clotting time and accelerated ferric-chloride-induced thrombosis. COMP specifically inhibited thrombin-induced platelet aggregation, activation, and retraction and the thrombin-mediated cleavage of fibrinogen. 26465321_COMP-Ang1 can enhance BMP2-induced cranial bone regeneration with increased pericyte recruitment. Combined delivery of the proteins might be a therapeutic strategy to repair cranial bone damage. 26746240_COMP forms a complex with collagens intracellularly that is a prerequisite for collagen secretion. 27151399_COMP deficiency drove macrophages toward the atherogenic phenotype and thereby aggravated atherosclerotic calcification. 27498005_these findings revealed the essential role of COMP in retarding the development of vascular aging and vascular smooth muscle cell senescence. 28860005_COMP could normally have a protective role against PASMC phenotype switching 29309831_Study describes a novel mechanism by which the deleterious consequences of mutant COMP retention results in upregulation of miR-223 disturbing the adipogenesis - osteogenesis balance. This results in reduction in bone mineral density, bone quality, mechanical strength and subchondral bone thickness which align with the pseudoachondroplasia phenotype. 29867124_COMP-prohibitin 2 interaction in mitochondria plays an important role in maintaining the contractile phenotype of Vascular smooth muscle cells by regulating mitochondrial oxidative phosphorylation. 29981303_COMP N-terminus directly interacted with the EGF11-12 domain of Notch1 and activated Notch1 signaling, as evidenced by co-immunoprecipitation and mammalian two-hybrid assay. In conclusion, COMP served as a potential ligand of Notch1, thereby driving embryonic stem cell (ESC)-vascular smooth muscle cells (VSMCs) differentiation. 30188931_Of the six biomarkers, only COMP and DPP4 showed high and SPP1 moderate correlation with the spinal muscular atrophy phenotype. PLS3 overexpression neither influenced the SMN level nor the six biomarkers, supporting the hypothesis that PLS3 acts as an independent protective modifier. 30553437_While identifying the cellular pathologic mechanisms underlying the murine mutant (MT)-COMP model of pseudoachondroplasia, increased midline-1 (MID1) expression and mammalian target of rapamycin complex 1 (mTORC1) signaling was found. 30720083_presents the expression levels of ADAMTS-7 and COMP in the decidual tissues of mice and humans suffering from SA. 31389735_COMP regulation of BMPR2 appears to have an important role in controlling hypoxia-elicited changes in pulmonary artery superoxide and its potential regulation of contractile phenotype proteins. 32347019_Mutant COMP shapes growth and development of skull and facial structures in mice and humans. 33068727_Cartilage oligomeric matrix protein fine-tunes disturbed flow-induced endothelial activation and atherogenesis. 34983194_COMP (Cartilage Oligomeric Matrix Protein), a Novel PIEZO1 Regulator That Controls Blood Pressure. 35468843_Duchenne muscular dystrophy trajectory in R-DMDdel52 preclinical rat model identifies COMP as biomarker of fibrosis. |
ENSG00000105664 |
COMP |
119.860826 |
0.4857907644 |
-1.041593 |
0.32080934 |
10.408060 |
0.00125466487353390141606945817898122186306864023208618164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0056524860555067692885478081166183983441442251205444335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
76.9533407744739 |
17.2533406636285 |
158.408406278411 |
26.0279597070433 |
| ENSMUSG00000031906 |
58994 |
Smpd3 |
protein_coding |
Q9JJY3 |
FUNCTION: Catalyzes the hydrolysis of sphingomyelin to form ceramide and phosphocholine. Ceramide mediates numerous cellular functions, such as apoptosis and growth arrest, and is capable of regulating these 2 cellular events independently. Also hydrolyzes sphingosylphosphocholine. Regulates the cell cycle by acting as a growth suppressor in confluent cells. Probably acts as a regulator of postnatal development and participates in bone and dentin mineralization (PubMed:15051724, PubMed:15764706, PubMed:15929065, PubMed:16025116). Binds to anionic phospholipids (APLs) such as phosphatidylserine (PS) and phosphatidic acid (PA) that modulate enzymatic activity and subcellular location (PubMed:21550973). May be involved in IL-1-beta-induced JNK activation in hepatocytes (By similarity). May act as a mediator in transcriptional regulation of NOS2/iNOS via the NF-kappa-B activation under inflammatory conditions (By similarity). {ECO:0000250|UniProtKB:O35049, ECO:0000269|PubMed:15051724, ECO:0000269|PubMed:15764706, ECO:0000269|PubMed:15929065, ECO:0000269|PubMed:16025116, ECO:0000269|PubMed:21550973}. |
Cell cycle;Cell membrane;Developmental protein;Golgi apparatus;Hydrolase;Lipid metabolism;Lipoprotein;Magnesium;Membrane;Metal-binding;Palmitate;Phosphoprotein;Reference proteome;Sphingolipid metabolism |
PATHWAY: Lipid metabolism; sphingolipid metabolism. {ECO:0000269|PubMed:15051724}. |
|
mmu:58994; |
cytoplasm [GO:0005737]; extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; Golgi cis cisterna [GO:0000137]; Golgi membrane [GO:0000139]; membrane [GO:0016020]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; neutral sphingomyelin phosphodiesterase activity [GO:0061751]; phosphatidic acid binding [GO:0070300]; phosphatidylserine binding [GO:0001786]; phospholipase activity [GO:0004620]; phosphoric diester hydrolase activity [GO:0008081]; sphingomyelin phosphodiesterase activity [GO:0004767]; artery smooth muscle contraction [GO:0014824]; BMP signaling pathway [GO:0030509]; bone development [GO:0060348]; bone growth [GO:0098868]; bone mineralization [GO:0030282]; cartilage development [GO:0051216]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to magnesium ion [GO:0071286]; cellular response to oxidised low-density lipoprotein particle stimulus [GO:0140052]; cellular response to peptide [GO:1901653]; cellular response to reactive oxygen species [GO:0034614]; cellular response to redox state [GO:0071461]; cellular response to tumor necrosis factor [GO:0071356]; ceramide metabolic process [GO:0006672]; chondrocyte development [GO:0002063]; chondrocyte development involved in endochondral bone morphogenesis [GO:0003433]; collagen metabolic process [GO:0032963]; dentinogenesis [GO:0097187]; DNA biosynthetic process [GO:0071897]; dopamine uptake [GO:0090494]; endochondral ossification [GO:0001958]; extracellular matrix assembly [GO:0085029]; G1 to G0 transition [GO:0070314]; hematopoietic progenitor cell differentiation [GO:0002244]; lung alveolus development [GO:0048286]; lung development [GO:0030324]; mitotic nuclear division [GO:0140014]; multicellular organism growth [GO:0035264]; negative regulation of cytosolic calcium ion concentration [GO:0051481]; negative regulation of hyaluronan biosynthetic process [GO:1900126]; ossification [GO:0001503]; peptide hormone secretion [GO:0030072]; platelet-derived growth factor receptor signaling pathway [GO:0048008]; polysaccharide transport [GO:0015774]; positive regulation of ceramide biosynthetic process [GO:2000304]; positive regulation of exosomal secretion [GO:1903543]; positive regulation of mitotic nuclear division [GO:0045840]; positive regulation of NIK/NF-kappaB signaling [GO:1901224]; positive regulation of nitric oxide biosynthetic process [GO:0045429]; positive regulation of smooth muscle cell proliferation [GO:0048661]; protein kinase B signaling [GO:0043491]; regulation of cartilage development [GO:0061035]; regulation of hyaluronan biosynthetic process [GO:1900125]; regulation of leukocyte migration [GO:0002685]; regulation of protein phosphorylation [GO:0001932]; respiratory system development [GO:0060541]; signal transduction [GO:0007165]; skeletal system development [GO:0001501]; sphingolipid mediated signaling pathway [GO:0090520]; sphingolipid metabolic process [GO:0006665]; sphingomyelin catabolic process [GO:0006685]; sphingomyelin metabolic process [GO:0006684] |
12566438_SMase2 is a structural gene for nSMase, acts as a bona fide N-SMase in cells, and has a role in the regulation of cell growth and cell signaling 15764706_the smpd3-null mouse develops a novel form of dwarfism and delayed puberty as part of a hypothalamus-induced combined pituitary hormone deficiency 16025116_role of deletion in gene leads to severe osteogenesis and dentinogenesis imperfecta 17591962_this novel form of combined pituitary hormone deficiency is characterized by the perturbation of the hypothalamus-pituitary growth axis, associated with retarded chondrocyte development and enchondral ossification in the epiphyseal growth plate. 18299447_in a mouse osteosarcoma model, a somatic homozygous deletion specifically targeting Smpd3 was identified 19250608_Results suggest that the smpd3 promoter is activated by BMP2 and is directly regulated by the Runx2 transcription factor. 20352118_nSMase2 activation is not involved in apoptosis mediated by TNFalpha and oxidized LDLs in murine fibroblasts, and in the hepatotoxicity of TNFalpha in mice, but is required for the mitogenic response to stress-inducing agents 20629193_This study identified nSMase2 as an important component for efficient memory formation and underscores the importance of ceramide in regulating synaptic events related to learning and memory. 21550973_interactions between nSMase2 and anionic phospholipids 21788370_The Col1a1-Smpd3 mice, in which osteoblast-specific expression of Smpd3 corrected the bone abnormalities observed in fro/fro embryos without affecting the cartilage phenotype. 22074919_initial structure/function insights regarding nSMase2 phosphorylation 22383528_NSMase2/Cer are the key mediators of the regulation of HA synthesis, via microdomains and the Akt/mTOR pathway 22945695_Neutral sphingomyelinase 2 deficiency is associated with lung anomalies similar to emphysema. 23046545_Data show that although sphingomyelin (SM) accumulated in both ASMase(-/-) and fro/fro (NSMase2(-/-)) fibroblasts, the reduction of ceramides was more dramatic in fro/fro cells. 23428435_Smpd3 expression in odontoblasts is required for tooth mineralization. 23439645_a requirement for nSMase2-mediated cancer cell exosomal miRNAs in the regulation of metastasis through the induction of angiogenesis in inoculated tumors. 23651497_The H2O2-induced src/PDGFRbeta/SK1 signaling cascade was impaired in nSMase2-deficient fro/fro cells and was rescued by exogenous C2Cer that activated src/PDGFRbeta/SK1. 24505141_Smpd3/nSMase2-ceramide-Akt signaling axis negatively regulates BMP-induced chondrocyte maturation. 24585429_Data confirms a crucial pro-survival role for SMPD3 during embryonic development. 25047167_These results suggest that OTC is a potent stimulant of nSMase-2 expression and that there may be unanticipated complications of OTC supplementation. 25287744_Smpd3 was identified as a redox-sensitive enzyme, whose basal activity mediated changes in its oligomeric state. 25347576_Src and p38 mitogen-activated protein kinase activities are critical for regulating nSMase2 phosphorylation. 25555205_these results highlight the role of nSMase2 in apoptosis evoked by nutrient starvation that could contribute to the delayed apoptosis of hypertrophic chondrocytes in the growth plate, and emphasize the antiapoptotic properties of HAS2 26138644_nSMase2 activation is required for the development of infection-induced diaphragm calpain activation and muscle weakness. 27325675_Study provides evidence that Smpd3 expression in both chondrocytes and osteoblasts is required for normal endochondral bone development. 27882938_SMPD3 in the regulation of the protein vesicular secretory pathway may become a diagnostic target in the etiology of unknown forms of juvenile growth and developmental inhibition. 28377412_Data suggest that IL-1beta induction of ceramide, cytokine expression, and leukocyte influx are regulated by brain neutral sphingomyelinase II protein (nSMase). 28889482_The enzymes involved in sphingolipid metabolism are expressed abnormally in B cells from lupus-prone mice. TLR signaling induced the abnormal expression of sphingomyelin phosphodiesterase 3 (SMPD3). TLR signaling also induced the transport of SMPD3 from the Golgi apparatus. Furthermore, the dysfunction of SMPD3 enhanced TLR-induced inflammatory response of B cells and macrophages in turn. 28961856_The authors report here that host delivery of Cryptosporidium parvum Cdg7_FLc_1000 RNA results in inhibition of epithelial cell migration through suppression of the gene encoding sphingomyelinase 3 (SMPD3). 29141446_hypoxic culture augments miR-210 and nSMase2 activities in MSCs and their secreted exosomes, and this is responsible at least in part for the enhanced cardioprotective actions of exosomes derived from hypoxia-treated cells. 29725009_SMPD3 deficiency perturbs neuronal proteostasis and causes progressive cognitive impairment in a mouse model. 30530524_Findings show that SMPD3 plays a significant role in ECM mineralization and chondrocyte apoptosis during fracture healing. 30866785_Results suggest that ZIKV modulates SMPD3 activity in cortical neurons for its infection and transmission through exosomes perhaps leading to severe neuronal death that may result in neurological manifestations such as microcephaly in the developing embryonic brains. 31199918_Loss of HAMP expression is associated with Chondrodysplasia with Unimpaired Skeletal Mineralization. 33727246_Neutral Sphingomyelinase 2 Heightens Anti-Melanoma Immune Responses and Anti-PD-1 Therapy Efficacy. 33875010_Pharmacological inhibition of nSMase2 reduces brain exosome release and alpha-synuclein pathology in a Parkinson's disease model. 33938457_Suppressing the intestinal farnesoid X receptor/sphingomyelin phosphodiesterase 3 axis decreases atherosclerosis. 34153465_Neonatal stroke enhances interaction of microglia-derived extracellular vesicles with microglial cells. 34272885_Regulatory T cells reduce endothelial neutral sphingomyelinase 2 to prevent T-cell migration into tumors. 34584229_Neutral sphingomyelinase mediates the co-morbidity trias of alcohol abuse, major depression and bone defects. 34951654_Activation of neutral sphingomyelinase 2 through hyperglycemia contributes to endothelial apoptosis via vesicle-bound intercellular transfer of ceramides. 36603748_Neuronal deletion of nSMase2 reduces the production of Abeta and directly protects neurons. |
ENSG00000103056 |
SMPD3 |
6997.808346 |
0.4092690253 |
-1.288879 |
0.09774656 |
169.122940 |
0.00000000000000000000000000000000000001150079723698151304914025772566817992652627513365097046122665586597890363225491744482962530063635629012930294834404776338487863540649414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000012602888022568517751196887973499571483009818058828383921468785737889067825125273879003593444428354963804395083570852875709533691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4045.27132263721 |
274.287992744325 |
9884.13750576293 |
499.012598282302 |
| ENSMUSG00000031963 |
73230 |
Bmper |
protein_coding |
Q8CJ69 |
FUNCTION: Inhibitor of bone morphogenetic protein (BMP) function, it may regulate BMP responsiveness of osteoblasts and chondrocytes. {ECO:0000250}. |
Disulfide bond;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
This gene encodes a secreted protein that contains five Von Willebrand factor type C domains and a Von Willebrand factor type D domain and a trypsin inhibitory-like domain. The encoded protein binds to bone morphogenetic proteins (BMP) and regulates their activity. Mutation of the related gene in humans causes diaphanospondylodysostosis. [provided by RefSeq, Mar 2013]. |
mmu:73230; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; blood vessel development [GO:0001568]; blood vessel endothelial cell proliferation involved in sprouting angiogenesis [GO:0002043]; BMP signaling pathway [GO:0030509]; common-partner SMAD protein phosphorylation [GO:0007182]; endothelial cell activation [GO:0042118]; inner ear development [GO:0048839]; negative regulation of BMP signaling pathway [GO:0030514]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of sprouting angiogenesis [GO:1903672]; regulation of angiogenesis [GO:0045765]; regulation of endothelial cell migration [GO:0010594]; regulation of pathway-restricted SMAD protein phosphorylation [GO:0060393]; regulation of protein localization [GO:0032880]; ureteric bud development [GO:0001657] |
15188429_Cv2 is a conserved, positive regulator of bone morphogenetic protein signaling. Its cysteine-rich domains acts as both positive and negative modulators of BMP signaling. 17035289_crossveinless 2 has essential pro-Bmp functions for locally restricted signal enhancement in multiple aspects of mammalian organogenesis. 18644438_The context-dependent functional relationship between Tsg and Cv2 during mouse development, is presented. 18662983_Cv2 may specify cardiac mesodermal lineage through inhibition of BMP signaling at early stage of cardiogenesis. 18789316_Development of the vertebral morphogenetic field is reported in the interactions of CV-2 and Twg. 19221194_Study demonstrates that as molar concentrations of Bmper exceed Bmp4, Bmper dynamically switches from an activator to an inhibitor of Bmp4 signaling. 19914233_Cv2 enhances pro-BMP activity of Tsg. 20807528_The data indicate a role for CV2 and Chd in the establishment of the vertebral morphogenetic field through the long-range relocalization of Chd/BMP complexes. 21737784_BMPER is important in the regulation of BMP signaling and revascularization in the hypoxic retina. 21900199_BMP modulator BMPER is a new protective regulator of vascular inflammation that modulates leukocyte adhesion and migration in vitro and in vivo. 22020334_The data unequivocally demonstrated that BMPER is highly expressed in malignant tumors and that the growth of lung, colon, and uterine carcinomas is dependent on the presence of BMPER. 22144676_Bmper may play an important role in suppressing hepcidin production in hypotransferrinemic mice. 22772758_Bmper is a critical regulator of Bmp-mediated vascular inflammation and that the fine-tuning of Bmp and Bmper levels is essential in the maintenance of normal vascular homeostasis. 23200275_BMPER appears to play a role in regulating both vessel density and cardiac development 23716395_BMPER expression is decreased following lung injury, which in turn impairs epithelial integrity, characterized by reduction of E-cadherin and epithelial leakage in vitro and in vivo. 24373957_BMPER-induced BMP signaling promotes coronary artery remodeling. 25503991_The proangiogenic BMPER effect in endothelial cells is mediated by inhibition of antiangiogenic thrombospondin-1 and enhanced expression and activation of the FGF signaling pathway that is crucial in the promotion of angiogenesis. 28464239_CV2 binds directly to BMP10, as determined by co-immunoprecipitation, and inhibits BMP10 from initiating SMAD signaling, as determined by luciferase reporter gene assays. BMP10 and CV2 have important roles in coordinating cardiomyogenesis in progenitor cells. 28596374_BMPER/low-density lipoprotein receptor-related protein 1 axis plays a pivotal role in pulmonary inflammatory response. 29093060_BMPER contributes to the precise control of BMP activity within the aorta-gonad-mesonephros region, enabling the maturation of hematopoietic stem cells within a BMP-negative environment. 32468615_BMPER is upregulated in obesity and seems to have a role in pericardial adipose stem cells. 33772019_Loss of bone morphogenetic protein-binding endothelial regulator causes insulin resistance. 34904361_BMPER alleviates ischemic brain injury by protecting neurons and inhibiting neuroinflammation via Smad3-Akt-Nrf2 pathway. |
ENSG00000164619 |
BMPER |
258.948426 |
0.1937591575 |
-2.367664 |
0.22273198 |
108.270498 |
0.00000000000000000000000023448907349665378474252569837114198687076166840573974644880282007866623139591411018045619130134582519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000133772942603034833095603011382024138382072524025523182414552339665470004348435395513661205768585205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
84.2340393726617 |
13.0562825594485 |
434.73578472416 |
47.5658250398935 |
| ENSMUSG00000032062 |
66952 |
2310030G06Rik |
protein_coding |
Q9D8L0 |
|
Alternative splicing;Phosphoprotein;Reference proteome |
|
|
mmu:66952; |
Human_homologues extracellular exosome [GO:0070062] |
|
ENSG00000149300 |
C11orf52 |
41.829700 |
0.2781085543 |
-1.846280 |
0.40616389 |
20.511697 |
0.00000592679610482441208697520010351134089887636946514248847961425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000457793906027816695138848990254132331756409257650375366210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.1087806454278 |
5.16580391251973 |
65.114072790745 |
12.7212246932203 |
| ENSMUSG00000032066 |
170752 |
Bco2 |
protein_coding |
Q99NF1 |
FUNCTION: Broad specificity mitochondrial dioxygenase that mediates the asymmetric oxidative cleavage of carotenoids (PubMed:11278918, PubMed:21106934). Cleaves carotenes (pure hydrocarbon carotenoids) such as all-trans-beta-carotene and lycopene as well as xanthophylls (oxygenated carotenoids) such as zeaxanthin, lutein and beta-cryptoxanthin at both the 9,10 and the 9',10' carbon-carbon double bond (PubMed:11278918, PubMed:21106934). Through its function in carotenoids metabolism regulates oxidative stress and the production of important signaling molecules (PubMed:21106934). {ECO:0000269|PubMed:11278918, ECO:0000269|PubMed:21106934}. |
Dioxygenase;Iron;Lipid metabolism;Metal-binding;Mitochondrion;Oxidoreductase;Reference proteome |
|
|
mmu:170752; |
mitochondrion [GO:0005739]; 9,10 (9', 10')-carotenoid-cleaving dioxygenase activity [GO:0010437]; beta,beta-carotene-9',10'-cleaving oxygenase activity [GO:0102076]; beta-carotene 15,15'-dioxygenase activity [GO:0003834]; carotenoid dioxygenase activity [GO:0010436]; metal ion binding [GO:0046872]; oxidoreductase activity, acting on single donors with incorporation of molecular oxygen, incorporation of two atoms of oxygen [GO:0016702]; carotene catabolic process [GO:0016121]; carotene metabolic process [GO:0016119]; carotenoid metabolic process [GO:0016116]; lutein catabolic process [GO:0062172]; lycopene catabolic process [GO:1901176]; regulation of mitochondrial membrane potential [GO:0051881]; regulation of reactive oxygen species metabolic process [GO:2000377]; retinal metabolic process [GO:0042574]; retinoic acid metabolic process [GO:0042573]; xanthophyll catabolic process [GO:0016124]; xanthophyll metabolic process [GO:0016122]; zeaxanthin catabolic process [GO:1901826] |
21106934_we found that the third family member, beta,beta-carotene-9',10'-oxygenase (BCDO2), is a mitochondrial carotenoid-oxygenase with broad substrate specificity 24034573_Pleiotropic actions of carotene-9',10'-monooxygenase supporting a strong role of these protein in regulating tissue carotenoid accumulation as well as tomato carotenoid-independent regulation of lipid metabolism. 24106281_a stepwise cleavage by BCO2 and BCO1 with APO10ol as an intermediate could provide a mechanism to tailor asymmetric carotenoids such as beta-cryptoxanthin for vitamin A production. 24553694_Data from knockout mice suggest that regulation of hepatic gene expression by dietary factors (here, supplementation with lycopene or tomato powder nutraceuticals) is dependent on Bco2. 25644347_Both lycopene and APO10LA supplementation were effective in reducing high saturated fat-induced liver steatosis in mice where BCO2 expression was absent. 26307071_Role of beta-Carotene 9,10-Dioxygenase in Macular Pigment Metabolism 27991717_Loss of BCO2 induces mitochondrial hyperactivation, mitochondrial stress, and changes of the mitochondrial proteome, leading to mitochondrial energy insufficiency. BCO2 appears to be critical for proper hepatic mitochondrial function. 28482236_BCO2 regulates hypothalamic mitochondrial function, nutrient metabolism, and local oxidative stress and inflammation. 30006135_provided novel experimental evidence that BCO1 and BCO2 could play a significant role in maintaining normal hepatic lipid and cholesterol homeostasis, potentially through the regulation of the FXR/miR-34a/SIRT1 pathway 33453359_beta-carotene oxygenase 2 deficiency-triggered mitochondrial oxidative stress promotes low-grade inflammation and metabolic dysfunction. 34161819_Lutein and zeaxanthin reduce A2E and iso-A2E levels and improve visual performance in Abca4(-/-)/Bco2(-/-) double knockout mice. |
ENSG00000197580 |
BCO2 |
30.436234 |
3.8887139843 |
1.959293 |
0.55040231 |
12.272497 |
0.00045968281921373888585305444998141410906100645661354064941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0023231216663117683872419316770674413419328629970550537109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
46.6326702359914 |
18.2255477060868 |
11.9917973973472 |
3.69961627584133 |
| ENSMUSG00000032076 |
54725 |
Cadm1 |
protein_coding |
Q8R5M8 |
FUNCTION: Mediates homophilic cell-cell adhesion in a Ca(2+)-independent manner (PubMed:12202822, PubMed:12799182, PubMed:12826663). Also mediates heterophilic cell-cell adhesion with CADM3 and NECTIN3 in a Ca(2+)-independent manner (PubMed:12826663). Interaction with CRTAM promotes natural killer (NK) cell cytotoxicity and interferon-gamma (IFN-gamma) secretion by CD8+ T-cells in vitro as well as NK cell-mediated rejection of tumors expressing CADM1 in vivo (PubMed:15811952). In mast cells, may mediate attachment to and promote communication with nerves (By similarity). CADM1, together with MITF, is essential for development and survival of mast cells in vivo (PubMed:15158462, PubMed:16605125). By interacting with CRTAM and thus promoting the adhesion between CD8+ T-cells and CD8+ dendritic cells, regulates the retention of activated CD8+ T-cell within the draining lymph node (PubMed:19752223). Required for the intestinal retention of intraepithelial CD4+ CD8+ T-cells and, to a lesser extent, intraepithelial and lamina propria CD8+ T-cells and CD4+ T-cells (PubMed:24687959). Interaction with CRTAM promotes the adhesion to gut-associated CD103+ dendritic cells, which may facilitate the expression of gut-homing and adhesion molecules on T-cells and the conversion of CD4+ T-cells into CD4+ CD8+ T-cells (PubMed:24687959). Acts as a synaptic cell adhesion molecule and plays a role in the formation of dendritic spines and in synapse assembly (PubMed:12202822, PubMed:23209303). May be involved in neuronal migration, axon growth, pathfinding, and fasciculation on the axons of differentiating neurons (PubMed:15707673). May play diverse roles in the spermatogenesis including in the adhesion of spermatocytes and spermatids to Sertoli cells and for their normal differentiation into mature spermatozoa (PubMed:12606335, PubMed:16612000). {ECO:0000250|UniProtKB:Q9BY67, ECO:0000269|PubMed:12202822, ECO:0000269|PubMed:12606335, ECO:0000269|PubMed:12799182, ECO:0000269|PubMed:12826663, ECO:0000269|PubMed:15158462, ECO:0000269|PubMed:15707673, ECO:0000269|PubMed:15811952, ECO:0000269|PubMed:16605125, ECO:0000269|PubMed:16612000, ECO:0000269|PubMed:19752223, ECO:0000269|PubMed:23209303, ECO:0000269|PubMed:24687959}. |
Alternative splicing;Apoptosis;Cell adhesion;Cell membrane;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Immunity;Immunoglobulin domain;Membrane;Phosphoprotein;Reference proteome;Repeat;Signal;Spermatogenesis;Synapse;Transmembrane;Transmembrane helix;Tumor suppressor |
|
|
mmu:54725; |
axon [GO:0030424]; basolateral plasma membrane [GO:0016323]; cell-cell junction [GO:0005911]; dendrite [GO:0030425]; glutamatergic synapse [GO:0098978]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic membrane [GO:0045211]; presynaptic membrane [GO:0042734]; Schaffer collateral - CA1 synapse [GO:0098685]; synapse [GO:0045202]; synaptic vesicle [GO:0008021]; cell adhesion molecule binding [GO:0050839]; PDZ domain binding [GO:0030165]; protein homodimerization activity [GO:0042803]; signaling receptor binding [GO:0005102]; apoptotic process [GO:0006915]; bone development [GO:0060348]; calcium-independent cell-cell adhesion via plasma membrane cell-adhesion molecules [GO:0016338]; cell adhesion [GO:0007155]; cell differentiation [GO:0030154]; cell recognition [GO:0008037]; detection of stimulus [GO:0051606]; heterophilic cell-cell adhesion via plasma membrane cell adhesion molecules [GO:0007157]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156]; liver development [GO:0001889]; lymphocyte migration into lymphoid organs [GO:0097021]; maintenance of postsynaptic specialization structure [GO:0098880]; negative regulation of ERBB4 signaling pathway [GO:0120154]; negative regulation of protein phosphorylation [GO:0001933]; positive regulation of cytokine production [GO:0001819]; positive regulation of natural killer cell mediated cytotoxicity [GO:0045954]; presynapse assembly [GO:0099054]; retrograde trans-synaptic signaling by trans-synaptic protein complex [GO:0098942]; spermatogenesis [GO:0007283]; susceptibility to natural killer cell mediated cytotoxicity [GO:0042271]; synapse assembly [GO:0007416]; synaptic membrane adhesion [GO:0099560]; unidimensional cell growth [GO:0009826] |
12202822_SynCAM1 is expressed in brain and localized to pre- and postsynaptic sites of neurons. SynCAM1 is a homophilic cell adhesion molecule that induces formation of presynaptic terminals by neurons. 12242005_The Tslc1 gene is mapped on mouse chromosome 9, a synteny of human chromosome 11q, and is composed of ten exons, the exon-intron junctions being highly conserved between human and mouse. 12456501_results showing that SgIGSF mediates the adhesion of mast cells to fibroblasts and that the transcription of SgIGSF is critically regulated by MITF. 12606335_SgIGSF on the surface of spermatogenic cells binds to some membrane molecules on Sertoli cells in a heterophilic manner and thereby may play diverse roles in the spermatogenesis 12826663_Possibly involved in localization of a transmembrane protein(s) through Pals2. 15158462_SgIGSF has a role in survival of cultured mast cells 15474495_SgIGSF and KIT independently play significant roles in cell migration 15707673_the tscl1 protein was localized in the brain neuroepithelium at embryonic day 9.5, neural crest at E10.5, motor neurons at E10.5, and olfactory epithelium at E16.5. 15781451_Necl2/CRTAM molecular pair could regulate a large panel of cell/cell interactions both within and outside of the immune system 15811952_NK cells and T8 cells recognize Necl-2 through CRTAM, expressed only on activated cells. CRTAM-Necl-2 interactions promote cytotoxicity of NK cells and IFN-gamma secretion of T8 cells as well as NK cell-mediated rejection of tumors expressing Necl-2 15862624_The spatiotemporal patterns of the expression suggest that IGSF4 plays crucial roles in the development of both telencephalon and cerebellum. 16382161_RA175-based cell junction is necessary for retaining elongating spermatids in the invagination of Sertoli cells for their maturation and translocation to the adluminal surface for timely release. 16605125_Results suggest that MITF and SgIGSF appear essential for the development and survival of mast cells in tissues of adult WBB6F1-KitW/KitW-v mice. 16611999_Tslc1 is essential for normal spermatogenesis in mice. 16612000_TSLC1/IGSF4 expression is indispensable for the adhesion of spermatocytes and spermatids to Sertoli cells and for their normal differentiation into mature spermatozoa. 16837733_results establish an essential role for Tslc1 in spermatozoal maturation & motility, distinct from other members of the nectin family 17300670_These results indicate that TSLC1 is a novel interameloblast adhesion molecule that may be downregulated during ameloblastic tumorigenesis. 17314315_Heterophilic binding of PVR to IGSF4A is responsible, at least in part, for the interaction between Sertoli and spermatogenic cells during mouse spermatogenesis. 17326163_We propose that SgIGSF is a novel and functional biliary epithelial cell adhesion molecule that is expressed for a limited time during active bile duct/ductule formation. 17492651_The retrograde response to axotomy of spinal motoneurons involves a rapid down-regulation of SynCAM1 mRNA, indicating that down-regulation of synaptic adhesion molecules plays a role in postinjury synapse elimination 18055550_In the Ra175(-/-) elongating spermatid, Par-3 was absent, and Jam-C was absent or abnormally localized. 18471525_The strong correlation between CADM1 expression and hormonally functional phenotypes suggests that CADM1 is involved in hormone secretion from ICTs. 18794147_Cadm1 promoter hypermethylation was significantly induced in transgenic transformed cells. 18922876_TSLC1 expression in Adult T-cell leukemia (ATL) cells plays an important role in the growth and organ infiltration of ATL cells. 19321127_the effect of 4.1B deficiency on tumor suppressor in lung cancer-1 distribution were also investigated using rodent pheochromocytoma and 4.1B-knockout mice. 19337787_Cell adhesion molecules between spermatogenic and Sertoli cells play essential roles in spermatogenesis. 19561085_The extracellular region of ErbB3, but not ErbB2, interacted in cis with that of Necl-2. 19783739_Necl2 overexpression resulted in reduced proliferation and increased levels of calcium/calmodulin-associated Ser/Thr kinase and E-cadherin at the leading edge of healing wounds 20368431_SynCAM 1 acts in developing neurons to shape migrating growth cones and contributes to the adhesive differentiation of their axo-dendritic contacts 20450890_this study demonstrates that CADM1 plays roles in regulating emotional behaviors, social behaviors and motor performances, and that CADM1 has important implications for psychiatric disorders with disruptions in social behavior, such as autism. 20479255_Analysis of serial sections of perinatal Ncam(-/-) brain revealed that polySia-SynCAM 1 is expressed exclusively by NG2 cells, a multifunctional glia population that can receive glutamatergic input via unique neuron-NG2 cell synapses. 21145003_Overexpression of SynCAM 1 in transgenic mice promotes excitatory synapse number, while loss of SynCAM 1 results in fewer excitatory synapses. SynCAM 1 also functions at mature synapses to alter their plasticity by regulating long-term depression. 21364653_The mutated CADM1 showed morphological abnormalities, including shorter dendrites, and impaired synaptogenesis in neurons. 21482734_results indicate that CADM1d is a specific neuronal isoform that enhances nerve-mast cell interaction, and they suggest that nerve-mast cell interaction may be reinforced as the brain grows mature because CADM1d becomes predominant 21486398_From pachytene spermatocytes at stage VI to step 4 spermatids, RA175 was not detected by the enzyme-labelled antibody method and was faintly detected by the indirect immunofluorescence method. 21486931_SynCAM1 mediates astrocyte-to-astrocyte and astrocyte-to-GnRH neuron adhesiveness in the hypothalamus. 21486934_SynCAM1 is expressed in astrocytes and contributes to erbB4 receptor-mediated control of female sexual development. 21926970_Synaptic cell adhesion molecule SynCAM 1 self-assembles laterally via its extracellular, membrane-proximal immunoglobulin domains 2 and 3, generating SynCAM oligomers and instructing interactions of SynCAM 1 across the nascent and mature synaptic cleft. 22084409_IGSF4 plays a central role in T cell functioning by dual independent mechanisms, control of T cell receptor signaling and control of T cell-antigen-presenting cell interaction. 22172944_These results clarify the detailed mechanism of membrane-proximal cleavage of CADM1, suggesting the possibility of RIP-mediated CADM1 signaling. 22210910_Cadm1 expression in the hair follicle plays a role in autoimmune alopecia. 22272290_Results suggest that although the Foxp2 transcription factor does not target Cadm1, Cadm1 at the synapses of Purkinje cells and parallel fibers is necessary for ultrasonic vocalization (USV) function. 22513384_CADM1 appeared to sustain gap junctional intercellular communication among alpha-cells via homophilic binding and, based on this action, CADM1 appears to prevent excessive secretion of glucagon from alpha-cells. 22553910_Cadm1 is indeed a bona fide tumor suppressor gene and provide new insights into genetic partners that co-operate in tumorigenesis when Cadm1-expression is lost. 22558465_disruption of SynCAM1-dependent astroglial function results in behavioral abnormalities similar to those described in animals model of attention-deficit hyperactive disorder 22701659_Epigenetic silencing complexity in the promoter region of Cadm1 is not only defined by DNA hypermethylation, but high nucleosome occupancy, altered nucleosome positioning and 'bivalent' histone modifications. 22908220_Polysialylation of the synaptic cell adhesion molecule 1 (SynCAM 1) depends exclusively on the polysialyltransferase ST8SiaII in vivo. 22994563_Cadm1 specifically interacts with Mupp1, and may form a ternary complex with Mupp1-GABBR2 in the cerebellum. 23142238_These results indicated that CADM1 was a novel osteoblastic adhesion molecule that is expressed transiently during osteoblastic maturation, and a useful diagnostic marker for osteosarcoma cells. 23169347_Loss of SynCAM1 modulates cocaine effects on spine structures in the nucleus accumbens. 23541110_The bone volume fraction did not differ between the CADM1 knockout mice and the wild type mice for both 8-week old and 11-week old mice. 23741316_TGF-beta1 is a potent cytokine that provides an effective mechanism in controlling Necl-2 expression in the testis via Smad-dependent gene repression and clathrin-mediated endocytosis. 23769722_Necl-2 co-localizes with ErbB4 in hippocampus and and regulates ErbB4 signaling in cultured neurons. 23982969_This study demonistrated that SynCAM 1 in the synaptic organization of the rod visual pathway and provide evidence for novel roles of synaptic adhesion in the structural and functional integrity of ribbon synapses. 25752299_Study demonstrates that polysialic acid-SynCAM 1 defines a subpopulation of NG2 cells and identifies the presence of polysialic acid-NRP2 in microglia 25774694_Our study reveals a novel mechanism of chronic NF-kappaB activation by CADM1 in HTLV-1 infected cells 26332750_This experiments show that the survival rate of adult-generated neurons is increased by overexpressing SynCAM 1 26867147_NF-kB mediated upregulation of CADM1 was identified as a mechanism of TNFalpha induced migration. 27053173_Data show that cell adhesion molecule 1 (SynCAM 1) generally prolongs the lifetime of spines. 27413156_The results of this study demonstrate that the synapse-organizing adhesion molecule SynCAM 1 has pronounced effects on neuronal connectivity in the hippocampal CA3 area. 27473321_SynCAM1, Neuroligin-1B and Neuroligin-2A were overexpressed in newborn neurons in the dentate gyrus of 7- to 9-week-old mice. SynCAM1 increased the morphological maturation of dendritic spines and mossy fiber terminals while Neuroligin-1B increased spine density. In contrast, Neuroligin-2A increased both spine density and size as well as GABAergic innervation and resulted in a drastic increase of neuronal survival. 28393893_alternative splicing can give rise to both shedding-susceptible and shedding-resistant CADM1 (cell adhesion molecule 1) variant proteins. 28414795_Cadm1 is a direct target of NFATc1, which is induced by RANKL through epigenetic modification, and regulates osteoclastic bone resorption in a negative feedback manner. 31635798_Comparative expression of cell adhesion molecule1 (CADM1) in the testes of experimental mice and some farm animals. 32535046_Nectin-like molecule 2, a necessary sexual maturation regulator, participates in congenital hypogonadotropic hypogonadism. 32616310_CADM1 suppresses c-Src activation by binding with Cbp on membrane lipid rafts and intervenes colon carcinogenesis. 33397712_Differential Contribution of Cadm1-Cadm3 Cell Adhesion Molecules to Peripheral Myelinated Axons. 33597944_Potentiating the Antitumor Activity of Cytotoxic T Cells via the Transmembrane Domain of IGSF4 That Increases TCR Avidity. 34332980_Expression of cell adhesion molecule 1 in human and murine endometrial glandular cells and its increase during the proliferative phase by estrogen and cell density. 34638130_The CADM1 tumor suppressor gene is a major candidate gene in MDS with deletion of the long arm of chromosome 11. 35133983_Evidence of islet CADM1-mediated immune cell interactions during human type 1 diabetes. 35213073_Trans-homophilic interaction of CADM1 promotes organ infiltration of T-cell lymphoma by adhesion to vascular endothelium. 36270981_Epstein Barr virus-mediated transformation of B cells from XIAP-deficient patients leads to increased expression of the tumor suppressor CADM1. 36709330_Concerted roles of LRRTM1 and SynCAM 1 in organizing prefrontal cortex synapses and cognitive functions. |
ENSG00000182985 |
CADM1 |
94.807046 |
0.0095516333 |
-6.710037 |
0.65637488 |
197.825097 |
0.00000000000000000000000000000000000000000000622955570484008508015599915059080041593010485907206930759618744695098347280458097714656013623328888503799663701370262214318529458978446200489997863769531250000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000008548521846497673666066421739414106006055477005213400526443255667012436480994037492673477086438687687774560988035466024825836939271539449691772460937500000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.93266591675718 |
0.90994425642232 |
202.338789612382 |
31.9514729355682 |
| ENSMUSG00000032118 |
235180 |
Fez1 |
protein_coding |
Q8K0X8 |
FUNCTION: May be involved in axonal outgrowth as component of the network of molecules that regulate cellular morphology and axon guidance machinery. May participate in the transport of mitochondria and other cargos along microtubules (By similarity). {ECO:0000250}. |
Cell membrane;Coiled coil;Cytoplasm;Cytoskeleton;Membrane;Microtubule;Phosphoprotein;Reference proteome;Transport;Ubl conjugation |
|
|
mmu:235180; |
axon [GO:0030424]; cell projection [GO:0042995]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; Golgi apparatus [GO:0005794]; growth cone [GO:0030426]; microtubule [GO:0005874]; mitochondrion [GO:0005739]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; gamma-tubulin binding [GO:0043015]; protein kinase C binding [GO:0005080]; protein N-terminus binding [GO:0047485]; cellular response to growth factor stimulus [GO:0071363]; establishment of cell polarity [GO:0030010]; establishment of mitochondrion localization [GO:0051654]; hippocampus development [GO:0021766]; mitochondrion morphogenesis [GO:0070584]; negative regulation of autophagosome assembly [GO:1902902]; positive regulation of anterograde axonal transport of mitochondrion [GO:0061881]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of neuron projection development [GO:0010976] |
15579145_Fez1 is mainly expressed in cortical layers V and VI, not in gamma-aminobutyric acid neurons but in pyramidal neurons, the projection neurons of the cerebral cortex. Immunohistochemistry also shows that Fez1 is expressed in deep layers of the neocortex. 18647754_investigated the role of FEZ1 in brain development and the pathogenesis of schizophrenia by generating mice that lack Fez1 22099459_Fezzeta-1 interacts with disrupted in schizophrenia (DISC)1 to synergistically regulate dendritic growth of newborn neurons in the adult mouse hippocampus. 24726361_Dendrite growth depends on degradation of FEZ1 regulated by Cdc20/APC. 27247180_Phosphorylation of FEZ1 by MARK regulates its function in presynaptic protein trafficking. 28928414_Loss of HDAC11 had no obvious impact on brain morphology and neural stem/precursor cells isolated from Hdac11 (KO/KO) mice had comparable proliferation and differentiation characteristics. However, in differentiating neural cells this study observed decreased expression of schizophrenia-associated gene Fez1. |
ENSG00000149557 |
FEZ1 |
28.420877 |
0.1680879569 |
-2.572712 |
0.55917601 |
21.570163 |
0.00000341117558319798007599767120101574846557923592627048492431640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000276377800442934197641328686323447527684038504958152770996093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.08975754702882 |
3.21461784531318 |
48.1281211021555 |
11.6831289882103 |
| ENSMUSG00000032172 |
244723 |
Olfm2 |
protein_coding |
Q8BM13 |
FUNCTION: Involved in transforming growth factor beta (TGF-beta)-induced smooth muscle differentiation (By similarity). TGF-beta induces expression and nuclear translocation of OLFM2 where it binds to SRF, causing its dissociation from the transcriptional repressor HEY2/HERP1 and facilitating binding of SRF to target genes (By similarity). Plays a role in AMPAR complex organization (PubMed:25218043). Is a regulator of vascular smooth-muscle cell (SMC) phenotypic switching, that acts by promoting RUNX2 and inhibiting MYOCD binding to SRF. SMC phenotypic switching is the process through which vascular SMCs undergo transition between a quiescent contractile phenotype and a proliferative synthetic phenotype in response to pathological stimuli. SMC phenotypic plasticity is essential for vascular development and remodeling (By similarity). {ECO:0000250|UniProtKB:O95897, ECO:0000250|UniProtKB:Q568Y7, ECO:0000269|PubMed:25218043}. |
Coiled coil;Cytoplasm;Disulfide bond;Glycoprotein;Membrane;Nucleus;Reference proteome;Secreted;Signal;Synapse |
|
|
mmu:244723; |
AMPA glutamate receptor complex [GO:0032281]; cytoplasm [GO:0005737]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; extrinsic component of synaptic membrane [GO:0099243]; glutamatergic synapse [GO:0098978]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; synaptic membrane [GO:0097060]; locomotory behavior [GO:0007626]; positive regulation of smooth muscle cell differentiation [GO:0051152]; protein secretion [GO:0009306]; regulation of vascular associated smooth muscle cell dedifferentiation [GO:1905174]; signal transduction [GO:0007165]; visual perception [GO:0007601] |
21228389_Highly conserved OLFM2 protein may play an important role in the course of retinal and eye development. 25218043_These results suggest that heterodimers of Olfm1 and Olfm2 interact with AMPAR more efficiently than Olfm2 homodimers and that Olfm2 plays a role in the organization of the AMPA receptor complexes. |
ENSG00000105088 |
OLFM2 |
79.921681 |
0.1137563975 |
-3.135980 |
0.43953888 |
48.179850 |
0.00000000000388863822699845347985867861575398683835619317861187482776585966348648071289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000720580747854506504163780759629291932516492735771862498950213193893432617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.1683608658855 |
5.00249525718919 |
142.131442454723 |
29.308579954251 |
| ENSMUSG00000032184 |
70082 |
Lysmd2 |
protein_coding |
A0A1L1SQI7 |
|
Proteomics identification;Reference proteome |
|
|
|
|
|
ENSG00000140280 |
LYSMD2 |
130.714866 |
0.2841440413 |
-1.815306 |
0.25139109 |
50.778157 |
0.00000000000103418276724368989214675427081486741120834593754551633537630550563335418701171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000205648458363653836403298824058035691036971925882426148746162652969360351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.9669762403257 |
10.5022669729169 |
211.044285693698 |
25.7787030637662 |
| ENSMUSG00000032186 |
50876 |
Tmod2 |
protein_coding |
Q9JKK7 |
FUNCTION: Blocks the elongation and depolymerization of the actin filaments at the pointed end. The Tmod/TM complex contributes to the formation of the short actin protofilament, which in turn defines the geometry of the membrane skeleton (By similarity). {ECO:0000250}. |
Actin-binding;Alternative splicing;Cytoplasm;Cytoskeleton;Direct protein sequencing;Phosphoprotein;Reference proteome |
|
|
mmu:50876; |
cytoskeleton [GO:0005856]; growth cone [GO:0030426]; myofibril [GO:0030016]; neuron projection [GO:0043005]; striated muscle thin filament [GO:0005865]; synapse [GO:0045202]; actin binding [GO:0003779]; tropomyosin binding [GO:0005523]; actin filament organization [GO:0007015]; learning or memory [GO:0007611]; muscle contraction [GO:0006936]; myofibril assembly [GO:0030239]; neuron-neuron synaptic transmission [GO:0007270]; pointed-end actin filament capping [GO:0051694]; positive regulation of G protein-coupled receptor signaling pathway [GO:0045745] |
12799133_Tmod2 plays a role in behavior, learning, memory, and synaptic plasticity. 24746171_Role of Tmod2 protein's leucine rich repeat domain in the formation of neurite-like processes |
ENSG00000128872 |
TMOD2 |
31.666638 |
0.0445435550 |
-4.488639 |
0.71168948 |
42.873620 |
0.00000000005839276935722219372267574992056487701019840130811644485220313072204589843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000009533104639326993944483563134337927857941963338817004114389419555664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.69438133896586 |
1.38372135716146 |
60.4886911340462 |
16.2773729684381 |
| ENSMUSG00000032207 |
15450 |
Lipc |
protein_coding |
P27656 |
FUNCTION: Catalyzes the hydrolysis of triglycerides and phospholipids present in circulating plasma lipoproteins, including chylomicrons, intermediate density lipoproteins (IDL), low density lipoproteins (LDL) of large size and high density lipoproteins (HDL), releasing free fatty acids (FFA) and smaller lipoprotein particles (By similarity). Also exhibits lysophospholipase activity (By similarity). Can hydrolyze both neutral lipid and phospholipid substrates but shows a greater binding affinity for neutral lipid substrates than phospholipid substrates (By similarity). In native LDL, preferentially hydrolyzes the phosphatidylcholine species containing polyunsaturated fatty acids at sn-2 position (By similarity). {ECO:0000250|UniProtKB:P07867, ECO:0000250|UniProtKB:P11150}. |
Glycoprotein;HDL;Heparin-binding;Hydrolase;Lipid degradation;Lipid metabolism;Reference proteome;Secreted;Signal |
|
|
mmu:15450; |
cell surface [GO:0009986]; early endosome [GO:0005769]; extracellular space [GO:0005615]; high-density lipoprotein particle [GO:0034364]; late endosome [GO:0005770]; microvillus [GO:0005902]; 1-acyl-2-lysophosphatidylserine acylhydrolase activity [GO:0052740]; acylglycerol lipase activity [GO:0047372]; acyltransferase activity [GO:0016746]; apolipoprotein binding [GO:0034185]; chylomicron binding [GO:0035478]; heparan sulfate proteoglycan binding [GO:0043395]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; lipase activity [GO:0016298]; lipid binding [GO:0008289]; lipoprotein lipase activity [GO:0004465]; low-density lipoprotein particle binding [GO:0030169]; lysophospholipase activity [GO:0004622]; phosphatidyl phospholipase B activity [GO:0102545]; phosphatidylserine 1-acylhydrolase activity [GO:0052739]; phospholipase A1 activity [GO:0008970]; phospholipase activity [GO:0004620]; triglyceride lipase activity [GO:0004806]; cholesterol homeostasis [GO:0042632]; cholesterol metabolic process [GO:0008203]; cholesterol transport [GO:0030301]; chylomicron remnant clearance [GO:0034382]; chylomicron remodeling [GO:0034371]; fatty acid biosynthetic process [GO:0006633]; fatty acid metabolic process [GO:0006631]; glycerophospholipid catabolic process [GO:0046475]; heparan sulfate proteoglycan biosynthetic process [GO:0015012]; high-density lipoprotein particle remodeling [GO:0034375]; lipid catabolic process [GO:0016042]; low-density lipoprotein particle clearance [GO:0034383]; low-density lipoprotein particle remodeling [GO:0034374]; neutral lipid catabolic process [GO:0046461]; phosphatidic acid metabolic process [GO:0046473]; phosphatidylcholine metabolic process [GO:0046470]; phosphatidylethanolamine metabolic process [GO:0046337]; phosphatidylserine metabolic process [GO:0006658]; regulation of plasma lipoprotein particle levels [GO:0097006]; triglyceride catabolic process [GO:0019433]; triglyceride homeostasis [GO:0070328]; triglyceride metabolic process [GO:0006641]; very-low-density lipoprotein particle remodeling [GO:0034372] |
11906928_findings indicate that hepatic lipase is required for optimal reproductive performance 11971936_synthesized in peritoneal macrophages 12117557_indicate a role for hepatic lipase in clearing phospholipase/free cholesterol from the vesicular lipoproteins of phospholipid transfer protein knock out mice. 12700335_the determinants of cell surface binding exist within the carboxyl terminal 70 amino acids of hepatic lipase, of which the last five residues play an important role 12897204_Hepatic lipase expression in macrophages contributes to atherosclerosis in apoE-deficient and LCAT-transgenic mice. 12939599_Not essential for diet-induced gallstone formation in mice. 15314098_hepatic lipase activity is influenced by epistatic interaction between two nonstructural loci on chromosomes 7 and 3 16368783_The results demonstrate that hepatic lipase deficiency, like LDLR deficiency, blunts the adrenal response to chronic ACTH stimulation and suggest a novel role for hepatic lipase in adrenal physiology. 16397139_HL-deficiency delays the onset and/or progression of atherosclerosis via a SR-BI-independent mechanism 17071916_Establishment of an antiatherogenic role of the ligand-binding function of hepataic lipase in LDL receptor-deficient mice. 17234195_ciHL expression reduces atherosclerosis 19524061_Elevated PLTP activity lowers plasma HDL-cholesterol by stimulating the plasma turnover and hepatic uptake of HDL cholesteryl esters. 19965617_highly divergent effects of naturally occurring HL coding sequence variants on lipid and lipoprotein metabolism 20056822_HL deficiency protects against diet-induced obesity and its hepatic sequelae. 20373033_These results demonstrated that fatty acid synthase and hepatic lipase might be FXR-regulated genes in liver cells. 20558822_The additive effect of hepatic lipase and endothelial lipase on HDL metabolism but not macrophage reverse cholesterol transport in mice. 21750705_Data show a novel pathway involving HL hydrolysis of VLDL that activates PPARdelta through generation of specific monounsaturated fatty acids. 23328279_Hepatic lipase- and endothelial lipase-deficiency in mice promotes macrophage-to-feces RCT and HDL antioxidant properties. 23355893_interaction of LIGHT with LTbetaR on hepatocytes, but not Kupffer cells, is sufficient to down regulate hepatic lipase expression and that this effect can be independent of LIGHT's costimulatory function. 24440515_HL expressed by osteoblasts has an impact on osteoblast OPG expression and that lack of HL leads to increased bone mass. 24777581_Hepatic lipase and endothelial lipase influence molecular species of several classes of plasma lipids. 24818611_Bone marrow (BM)-derived hepatic lipase mitigates the HDL-lowering, HDL-modulating, and cholesterol-raising effects of BM-derived cholesteryl ester transfer protein. 26423094_hepatic lipase KO mice had dyslipidemia including hypercholesterolemia, hypertriglyceridemia and increased non-esterified fatty acid levels. Also glucose intolerance, pancreatic and hepatic inflammation and steatosis. 27172975_The LIGHT (Tumour necrosis factor ligand superfamily member 14, TNFSF14)/Lymphotoxin beta-Receptor(LTbeta-R) pathway, which is involved in T-cell and macrophage activation, was diminished in plasma and in apoE-/-Irs2+/-HL-/- atheromas. 34197964_Potential role of hepatic lipase in the accretion of docosahexaenoic acid (DHA) by the brain. |
ENSG00000166035 |
LIPC |
234.781546 |
0.0640973520 |
-3.963591 |
0.24767756 |
262.739911 |
0.00000000000000000000000000000000000000000000000000000000004338032954484593340566152414465151967748080325542444797875592496623954902002159251165605582765780033810245737240898210822616024008656827039379652935502384991650615120306611061096191406250000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000094395597089584753354639246245439798695478050605751543972405692261321806063220707789238907129194652220593810232601469930988234506647829970347584466194845731479290407150983810424804687500000000000000000000 |
Yes |
Yes |
27.5138617990197 |
4.74673407486559 |
429.25114572131 |
44.4454557992554 |
| ENSMUSG00000032218 |
12442 |
Ccnb2 |
protein_coding |
P30276 |
FUNCTION: Essential for the control of the cell cycle at the G2/M (mitosis) transition. |
Cell cycle;Cell division;Cyclin;Mitosis;Phosphoprotein;Reference proteome |
|
|
mmu:12442; |
centrosome [GO:0005813]; cyclin-dependent protein kinase holoenzyme complex [GO:0000307]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; membrane [GO:0016020]; microtubule cytoskeleton [GO:0015630]; nucleus [GO:0005634]; cyclin-dependent protein serine/threonine kinase regulator activity [GO:0016538]; cell division [GO:0051301]; G2/MI transition of meiotic cell cycle [GO:0008315]; in utero embryonic development [GO:0001701]; mitotic cell cycle phase transition [GO:0044772]; regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0000079]; regulation of growth [GO:0040008]; spindle assembly involved in female meiosis I [GO:0007057]; T cell homeostasis [GO:0043029]; thymus development [GO:0048538] |
20404349_menin-mediated repression of cyclin B2 is crucial for inhibiting G(2)/M transition and cell proliferation through a previously unrecognized molecular mechanism for menin-induced suppression of MEN1 tumorigenesis 21819345_this study identifies a correlation between the presence of AIRE and proteasomal breakdown of cyclin B2, which leads us to speculate that cyclin B2 could be a target of AIRE's E3-ubiquitin ligase activity 22193451_Menin may inhibit MEN1 insulinoma by suppressing cyclin B2 expression via histone modification. 23541922_Hec1-dependent cyclin B2 stabilization during early metaphase is important for early-stage spindle assembly. 24776885_Report roles for cyclin B2 and p53 in centrosome separation that may be perturbed in many human cancers. 29074977_Rate of translation of Cyclin B1 and B2 proteins, together with their degradation, is a major determinant of their divergent temporal accumulation during oocyte maturation. The Cdk1-independent translation of Ccnb2 mRNA strongly suggest that this Cyclin is present in prophase and may play a critical role in setting the stockpile of Cyclin B protein for the assembly of pre-maturation promoting factor. 30097513_These results have revealed a hidden compensatory mechanism between Cyclin B1 and Cyclin B2 in regulating MPF and oocyte meiotic resumption. 31704793_results indicate that cyclin B2/CDK1 is also responsible for separase inhibition via inhibitory phosphorylation to regulate chromosome separation in oocyte meiosis, which may not occur in other cell types. 32439164_5'-UTR and ORF elements, as well as the 3'-UTR regulate the translation of Cyclin. 32518258_CYCLIN-B1/2 and -D1 act in opposition to coordinate cortical progenitor self-renewal and lineage commitment. 35349480_Cyclin B2 overexpression promotes tumour growth by regulating jagged 1 in hepatocellular carcinoma. |
ENSG00000157456 |
CCNB2 |
654.788059 |
2.0407120706 |
1.029073 |
0.10833452 |
89.451593 |
0.00000000000000000000314234000339634545033307507819048207185160131243735126744566488188326047747978009283542633056640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001311345833746113119011158782863104346479492928920863653238781942889090714743360877037048339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
883.141351215143 |
66.3789891018079 |
432.761369894617 |
25.0695029990269 |
| ENSMUSG00000032254 |
71819 |
Kif23 |
protein_coding |
E9Q5G3 |
FUNCTION: Component of the centralspindlin complex that serves as a microtubule-dependent and Rho-mediated signaling required for the myosin contractile ring formation during the cell cycle cytokinesis. Essential for cytokinesis in Rho-mediated signaling. Required for the localization of ECT2 to the central spindle. Plus-end-directed motor enzyme that moves antiparallel microtubules in vitro (By similarity). {ECO:0000250}. |
ATP-binding;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Isopeptide bond;Microtubule;Mitosis;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:71819; |
centralspindlin complex [GO:0097149]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; Flemming body [GO:0090543]; intercellular bridge [GO:0045171]; kinesin complex [GO:0005871]; microtubule [GO:0005874]; midbody [GO:0030496]; mitotic spindle [GO:0072686]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle [GO:0005819]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule-based movement [GO:0007018]; mitotic cytokinesis [GO:0000281]; mitotic spindle midzone assembly [GO:0051256]; positive regulation of cytokinesis [GO:0032467] |
27212033_depletion of KIF23 inhibited lung tumor formation in vivo, similar to what is seen with deletion of MMB. In vitro, the depletion of KIF23 in lung cancer cell lines showed strong anti-proliferative activity, which was accompanied by apoptosis in a subset of cell lines. 31007778_Kif23 up-regulation promotes cell proliferation and gastric cancer progression. 32818800_Differential tissue specific expression of Kif23 alternative transcripts in mice with the human mutation causing congenital dyserythropoietic anemia type III. |
ENSG00000137807 |
KIF23 |
1173.583710 |
2.4408078853 |
1.287359 |
0.15172990 |
70.246520 |
0.00000000000000005233751570169324293940163375282204909735456289173079202914351526487735100090503692626953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000015976052889142112590670820145758813682082625498223626969718225154792889952659606933593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1667.5761811779 |
175.704406705862 |
683.206651060976 |
54.4403963309027 |
| ENSMUSG00000032289 |
207596 |
Thsd4 |
protein_coding |
Q3UTY6 |
FUNCTION: Promotes FBN1 matrix assembly. Attenuates TGFB signaling, possibly by accelerating the sequestration of large latent complexes of TGFB or active TGFB by FBN1 microfibril assembly, thereby negatively regulating the expression of TGFB regulatory targets, such as POSTN. {ECO:0000269|PubMed:18757743, ECO:0000269|PubMed:19940141, ECO:0000269|PubMed:21880733}. |
Alternative promoter usage;Extracellular matrix;Hydrolase;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:207596; |
extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; microfibril [GO:0001527]; hydrolase activity [GO:0016787]; elastic fiber assembly [GO:0048251]; microfibril assembly [GO:0160054] |
19940141_Results indicated that ADAMTSL-6 is a novel microfibril-associated protein that binds directly to fibrillin-1 and promotes fibrillin-1 matrix assembly. 21880733_ADAMTSL6beta has a role in fibrillin-1 microfibril formation 32855533_Pathogenic variants in THSD4, encoding the ADAMTS-like 6 protein, predispose to inherited thoracic aortic aneurysm. |
ENSG00000187720 |
THSD4 |
392.098413 |
2.5999985334 |
1.378511 |
0.19557615 |
48.376471 |
0.00000000000351763643539391769125266037303539061074572624754353000753326341509819030761718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000655821764797064233639659359343851950663317751377690001390874385833740234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
545.056685092861 |
74.1160759079854 |
209.637304825613 |
21.8535499637289 |
| ENSMUSG00000032291 |
12903 |
Crabp1 |
protein_coding |
P62965 |
FUNCTION: Cytosolic CRABPs may regulate the access of retinoic acid to the nuclear retinoic acid receptors. |
3D-structure;Cytoplasm;Reference proteome;Retinol-binding;Transport;Vitamin A |
|
|
mmu:12903; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleus [GO:0005634]; fatty acid binding [GO:0005504]; retinal binding [GO:0016918]; retinoic acid binding [GO:0001972]; retinoid binding [GO:0005501]; retinol binding [GO:0019841]; fatty acid transport [GO:0015908] |
12623064_These data suggest that CRABPI functions to regulate the intracellular concentrations of retinoic acid and to maintain high levels of oxidized retinoic acid metabolites such as 4-oxoretinoic acid within cells. 15072263_The expression of CRABP I in the embryonic cerebellum coincides with earliest cerebellar differentiation; its involvement in a complex network of regulatory genes expressed in the mes/methencephalon is discussed in this review. 15130509_This study presents the first evidence that over-expressed receptor interacting protein 140 acts as a thyroid hormone (T3)-dependent negative co-regulator for T3 induction of the endogenous cellular retinoic acid binding I protein gene in P19 cells. 15152096_proline isomerization may not be responsible for the slowest folding phase of CRABP I so the loss of a slow refolding phase upon mutation may not be diagnostic for proline isomerization effects on protein folding 18006143_Increases in CRABPI and II transcripts in the absence of leukemia inhibitory factor may regulate aspects of embryonic stem cell differentiation in response to retinol. 22982089_CRABPI binds to all-trans retinoic acid in the cytoplasm, resulting in rapid ERK1/2 activation that is independent of membrane signal and nuclear RA receptors. 24236614_Only a small fraction of CRABP1 populates this functionally important but vulnerable conformation of CRABP1, enabling efficient ligand binding. 26935534_This study reveals a physiological relevance of the non-genomic action of atRA, mediated by Crabp1, in modulating cell cycle progression and apoptosis induction 28911165_The physiological role of Crabp1 in modulating stem cell proliferation and hippocampus-dependent brain activities such as learning and memory. 29371236_This is the first report of a physiological role of cellular retinoic acid binding protein I( CRABP)1 as an endogenous regulator of calcium-calmodulin protein kinase II, which protects the heart from isoproterenol-induced damage. 31164723_Cellular retinoic acid binding protein 1 protects mice from high-fat diet-induced obesity by decreasing adipocyte hypertrophy. 31344789_These are supported by the fact that CRABP1 gene knockout (KO) mice exhibit multiple phenotypes including hippocampal NSC expansion and spontaneous cardiac hypertrophy. This indicates that more potential processes/signaling pathways involving atRA-CRABP1 may exist, which remain to be identified. 31358819_A new regulatory mechanism for Raf kinase activation, retinoic acid-bound Crabp1. 32527063_Sonic Hedgehog-Gli1 Signaling and Cellular Retinoic Acid Binding Protein 1 Gene Regulation in Motor Neuron Differentiation and Diseases. 34193153_Regulation of exosome secretion by cellular retinoic acid binding protein 1 contributes to systemic anti-inflammation. 34830120_Crabp1 Modulates HPA Axis Homeostasis and Anxiety-like Behaviors by Altering FKBP5 Expression. 35794192_Modulation of adipose inflammation by cellular retinoic acid-binding protein 1. |
ENSG00000166426 |
CRABP1 |
14.203567 |
0.0064014273 |
-7.287391 |
1.41180182 |
30.855421 |
0.00000002779845274590161096615462457942030694013624270155560225248336791992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003143474626767433895329956119318870477741256763692945241928100585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.189358769872797 |
0.216328246749243 |
29.5807108819459 |
12.9401957588695 |
| ENSMUSG00000032355 |
69642 |
Mlip |
protein_coding |
Q5FW52 |
FUNCTION: Required for precocious cardiac adaptation to stress through integrated regulation of the AKT/mTOR pathways and FOXO1. Regulates cardiac homeostasis and plays an important role in protection against cardiac hypertrophy (PubMed:26359501, PubMed:22343712, PubMed:26436652). Acts as a transcriptional cofactor, represses transactivator activity of ISL1 and MYOCD (PubMed:22343712). {ECO:0000269|PubMed:22343712, ECO:0000269|PubMed:26359501, ECO:0000269|PubMed:26436652}. |
Alternative splicing;Cell membrane;Membrane;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:69642; |
nuclear envelope [GO:0005635]; nuclear lumen [GO:0031981]; nucleus [GO:0005634]; PML body [GO:0016605]; sarcolemma [GO:0042383]; lamin binding [GO:0005521]; transcription corepressor activity [GO:0003714]; negative regulation of cardiac muscle hypertrophy [GO:0010614]; negative regulation of cardiac muscle hypertrophy in response to stress [GO:1903243]; negative regulation of transcription by RNA polymerase II [GO:0000122]; transcription by RNA polymerase II [GO:0006366] |
21498514_MLIP is expressed most highly in striated and smooth muscle, it is likely to contribute to the mesenchymal phenotypes of laminopathies. 22343712_studies identify cardiac Isl1-interacting protein (CIP) as a novel regulator of cardiac hypertrophy. 26359501_Mlip-deficient hearts showed an impaired capacity to adapt to stress (isoproterenol-induced hypertrophy), likely because of deregulated Akt/mTOR activity. 26436652_Deletion of the CIP-encoding gene accelerated progress from hypertrophy to heart failure in several cardiomyopathy models. 30389785_Mlip tissue-specific expression and alternative splicing play a critical role in determining MLIP's functions in mice 32719146_Regulation of myonuclear positioning and muscle function by the skeletal muscle-specific CIP protein. 34400329_Cardiac CIP protein regulates dystrophic cardiomyopathy. |
ENSG00000146147 |
MLIP |
359.741579 |
0.4910444453 |
-1.026074 |
0.14093264 |
52.603508 |
0.00000000000040815949701635474813072461619782260147833163888009266884182579815387725830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000085871346112612092063067134444619827021022429036634093790780752897262573242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
237.124605255055 |
23.2008649800511 |
482.898457606126 |
34.0457067380247 |
| ENSMUSG00000032356 |
19417 |
Rasgrf1 |
protein_coding |
P27671 |
FUNCTION: Promotes the exchange of Ras-bound GDP by GTP. {ECO:0000269|PubMed:10373510}. |
3D-structure;Guanine-nucleotide releasing factor;Phosphoprotein;Reference proteome;Repeat;Ubl conjugation |
|
|
mmu:19417; |
apical dendrite [GO:0097440]; apicolateral plasma membrane [GO:0016327]; basolateral plasma membrane [GO:0016323]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; growth cone [GO:0030426]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; glutamate receptor binding [GO:0035254]; guanyl-nucleotide exchange factor activity [GO:0005085]; protein C-terminus binding [GO:0008022]; protein kinase binding [GO:0019901]; receptor tyrosine kinase binding [GO:0030971]; small GTPase binding [GO:0031267]; activation of GTPase activity [GO:0090630]; neuron projection development [GO:0031175]; positive regulation of fibroblast proliferation [GO:0048146]; positive regulation of GTPase activity [GO:0043547]; positive regulation of Ras protein signal transduction [GO:0046579]; Ras protein signal transduction [GO:0007265]; regulation of neuronal synaptic plasticity [GO:0048168]; regulation of NMDA receptor activity [GO:2000310]; regulation of Rac protein signal transduction [GO:0035020]; regulation of Ras protein signal transduction [GO:0046578]; regulation of synaptic plasticity [GO:0048167]; response to endoplasmic reticulum stress [GO:0034976]; type B pancreatic cell proliferation [GO:0044342] |
12496377_First report of the role of RhoA-specific p190RhoGEF in immune cell activation following CD40 stimulation of WEHI 231 B cells. 12740578_At Rasgrf1, a repeated DNA element is needed to establish methylation and expression of the active paternal allele. 12805218_Results demonstrate a novel and specific role for Ras-GRF1 pathways in the development and maintenance of normal beta-cell number and function. 12925882_RASGRF1 is an imprinted gene with a role in postnatal growth control 14676298_phospholipase C-gamma2 regulates RasGRP3 localization and thereby Ras activation. 15029245_Data show that Ras-GRF1 and Ras-GRF2 couple NMDA glutamate receptors to the activation of the Ras/Erk signaling cascade and to the maintenance of CREB transcription factor activity in cortical neurons of adult mice. 15513915_RasGrf1 as a novel target of neurotrophin activation; an additional pathway whereby neurotrophin-stimulated neurite outgrowth may be regulated 16236762_p190RhoGEF is involved in aggregation of NF-L protein and support a working hypothesis that aggregation of p190RhoGEF and NF-L is an upstream event triggering neurotoxicity in motor neuron disease. 16314537_The differentially methylated domain (DMD)is an enhancer blocker that binds CTCF in a methylation-sensitive manner and thus regulates Rasgrf1. 16325815_The isolated catalytic hairpin of Rasgrf1 retains dissociation activity but has impaired nucleotide exchange activity. 16407208_Ras-GRF proteins have roles in coupling calcium-permeable AMPA glutamate receptors to Ras/Erk signaling in cortical neurons 16467520_Mediates forms of synaptic plasticity by coupling different classes of NMDA receptors to distinct MAP kinase pathways. 16513268_results suggest that Ptprz plays a critical role in memory formation by modulating Rho GTPase activity through dephosphorylation at Y1105 on p190 RhoGAP. 16921254_These data show that phosphorylation of RasGRF1 by Cdk5 tightly regulates its levels, which is essential for proper cellular organization. 17028431_We conclude that p190 RhoGAPs control pivotal aspects of neural development, including neuronal differentiation and process outgrowth, and that these effects are mediated by signaling systems that include, but are not limited to, RhoA. 17030618_Rasgrf1 repeats serve at least two functions: first, to establish Rasgrf1 DNA methylation in the male germ line, and second, to resist global demethylation in the preimplantation embryo. 17075039_RasGRF1 lacks the allosteric activation switch that is crucial for Sos1 activity 17135267_a physical and functional interaction between RasGRF1 and SCLIP; this novel interplay may have possible implications in mechanisms that regulate neuronal morphology and structural plasticity 17993462_Delta-catenin T454A, a defective form in p190RhoGEF binding, did not decrease the binding between p190RhoGEF and RhoA. 18195107_p190RhoGEF overexpression enhances RhoA activation and focal adhesion (FA) formation in Mouse embryonic fibroblasts dependent on FAK binding and is associated with p190RhoGEF FA recruitment and tyrosine phosphorylation. 18502760_p190A is a novel GSK-3beta substrate; there is a mechanism by which GSK-3beta contributes to cellular polarization in directionally migrating cells via effects on Rho GTPase activity 18584336_chronic tetrahydrocannabinol modulates the expression and subcellular localization of proteins implicated in Ras signaling, calcium-buffering potential, and trafficking 18670629_The repressive H3K27 methylation and DNA methylation modifications are mutually antagonistic in the Rasgrf1 region. 19513790_Imprinted Rasgrf1 expression levels are critical for development and affects IGF-1 function. 21072176_Data show that noncoding RNA, AK029869, within the Rasgrf1 locus is imprinted. 21115823_These data reveal the central role of Ras-GRF1 in governing striatal adaptations to dopamine replacement therapy and validate a viable treatment for LID based on intracellular signaling modulation. 21182853_the evidence points towards the involvement of Rasgrf1 in the regulation of longevity, hypothetically through a mechanism similar to that observed in other long-lived mice of low GH/IGF-1 signaling causing a low body weight and life-extension 21251221_these results indicate that Rasgrf1 expression from the wild-type paternal allele contributes to learning and memory in neonatal mice. 21422498_role of RASGRF1 in aging, lifespan and metabolic parameters were analyzed in aged RasGrf1(-/-) mice. We observed that mice deficient for RasGrf1(-/-) display an increase in average and most importantly, in maximal lifespan (20% higher than controls). 21566194_study found components of the piRNA pathway are required for de novo methylation of the differentially methylated region of imprinted Rasgrf1 locus; propose a model in which piRNAs and a target RNA direct sequence-specific methylation of Rasgrf 21620943_Present RasGRF1-derived peptides displaying both in vitro and in vivo Ras inhibitory properties. 21685891_Demonstrate a role for RasGRF1/2 as negative regulators of Cdc42 activation, suppressing tumor cell movement, cytoskeletal dynamics and cell transformation. 21945077_p190A is required in both the epithelial and stromal compartments for ductal outgrowth and it may play a role in mammary epithelial cell differentiation 22547676_identified the guanine nucleotide exchange factor Rasgrf1 as a direct Zac1/Plagl1 target gene in beta cells 23145787_Ras-GRF1 and -GRF2 may act as adaptors that bind PLCgamma1 and restrict Ca2+ signalling to the vicinity of focal adhesions, indicating a new role for these GRFs that is required for IL-1 induction of the Ras-->ERK pathway and MMP-3 expression 23766509_Data indicate that contextual discrimination involves term potentiation (LTP) promoted by calcium-permeable AMPA-type glutamate receptors, RAS-GRF1 and p38 MAP kinase. 24174283_GRF1 is expressed in new neurons when GRF1 loss begins to effect neuronal function, promoting late stages of adult neurogenesis. GRF1 is an age-dependent regulator of adult hippocampal neurogenesis and the ability to distinguish closely related contexts. 24844602_Study suggests a complex role of ERK-dependent and Ras-GRF1-dependent signaling in corticostriatal plasticity, highlights differences between synaptic mechanisms in naive slices and dopamine-depleted preparations from L-DOPA-treated dyskinetic animals 25267792_CARD9 regulates H-Ras activation by linking Ras-GRF1 to H-Ras, which mediates Dectin-1-induced extracellular signal-regulated protein kinase (ERK) activation and proinflammatory responses when stimulated by their ligands. 25421944_RasGrf1 is an important upstream component of signal transduction pathways regulating Pttg1 expression and controlling beta cell development and physiological responses. 25644714_VLDLR requires RasGRF1/CaMKII to alter dendritic spine formation. 26246084_Data (including data from studies in knockout mice) suggest mechanisms involving RasGRF1 exist to regulate hypothalamic-pituitary-adrenal axis in early-adolescent females; such mechanisms appear absent in younger/older female or adolescent male mice. 26890742_Novel mercaptoacetamide-based class II histone deacetylase inhibitor HDACIW2 treatment regulated the levels of RasGRF1 and p-ERK, and that W2 requires RasGRF1 and ERK signaling to alter dendritic spine formation, suggesting that W2 regulates dendritic spine formation via a RasGRF1/ERK-dependent signaling pathway 28611277_These findings demonstrate that RASgrf1 is closely associated with epilepsy via the aberrant methylation of RASgrf1, and regulating the methylation status of relevant genes might be an intriguing topic in future research on epilepsy. 30242550_hypermethylation of RASgrf1 after kainate induced status epilepticus could be reversed with corresponding changes of RASgrf1 expression 30308936_The deletion of RasGRF1 attenuated myocardial fibrosis and improved cardiac function in diabetic mice through inhibiting inflammation and oxidative stress. 31182637_RasGRF1 has no effect on self administration for cocaine. 31361349_p190RhoGEF may play a role in the stage of plasma cell differentiation during T-cell-dependent humoral immune responses. 35422804_Expression of a RhoA-Specific Guanine Nucleotide Exchange Factor, p190RhoGEF, in Mouse Macrophages Negatively Affects M1 Polarization and Inflammatory Responses. 36098964_Loss of MLL Induces Epigenetic Dysregulation of Rasgrf1 to Attenuate Kras-Driven Lung Tumorigenesis. |
ENSG00000058335 |
RASGRF1 |
32.935794 |
0.0862515438 |
-3.535306 |
0.61199333 |
32.835927 |
0.00000001002743462127898199376876969693764274360603394598001614212989807128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001209326081958206195193664209824868649434392864350229501724243164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.94230361650455 |
2.15997792806235 |
57.3010453048333 |
15.3302340995376 |
| ENSMUSG00000032373 |
76459 |
Car12 |
protein_coding |
A0A0R4J0W4 |
FUNCTION: Reversible hydration of carbon dioxide. {ECO:0000256|RuleBase:RU367011}. |
Lyase;Membrane;Metal-binding;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix;Zinc |
|
This gene encodes a membrane-bound member of the alpha carbonic anhydrase family of enzymes that catalyze the reversible hydration of carbon dioxide to bicarbonate. These proteins participate in a variety of biological processes, including respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor, cerebrospinal fluid, saliva, and gastric acid. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Apr 2015]. |
mmu:76459; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; carbonate dehydratase activity [GO:0004089]; zinc ion binding [GO:0008270]; chloride ion homeostasis [GO:0055064]; estrous cycle [GO:0044849] |
14983512_High expression in the kidney and colon suggests a role for carbonic anhydrase XII in the maintenance of body ion and pH homeostasis in the mouse. 16719910_CA12 may regulate ion and pH homeostasis within the developing embryo. 20820888_Car4, Car6, and Car12 showed moderate expression levels in the mouse harderian gland. 23233153_During inner ear development, transcripts of four cytosolic isozymes (Car1, Car2, Car3, and Car13), two membrane-bound isozymes (Car12 and Car14), and two CA-related proteins (Car8 and Car11) were expressed at higher levels than other isozymes. 25771146_P2Y2R-mediated expression of CAXII by VICs acidifies the extracellular space and promotes the regression of CAVS. |
ENSG00000074410 |
CA12 |
44.518517 |
16.0860379537 |
4.007737 |
0.56613472 |
51.660174 |
0.00000000000065987796793100117518048497798204980778827288823151775432052090764045715332031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000134016816100333470335538163302843304644507149703258619410917162895202636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
84.4487222320565 |
33.9959974856193 |
5.24981493113821 |
1.80342977356753 |
| ENSMUSG00000032380 |
13143 |
Dapk2 |
protein_coding |
Q8VDF3 |
FUNCTION: Calcium/calmodulin-dependent serine/threonine kinase involved in multiple cellular signaling pathways that trigger cell survival, apoptosis, and autophagy. Capable of regulating both type I apoptotic and type II autophagic cell death signals. The former involves caspase activation, chromatin and mitochondrial condensation while the latter involves caspase-independent cell death in conjunction with accumulation of mature autophagic vesicles, plasma membrane blebs, and nuclear condensation without DNA degradation. Mediator of anoikis and a suppressor of beta-catenin-dependent anchorage-independent growth of malignant epithelial cells. May play a role in granulocytic maturation (By similarity). Regulates granulocytes motility by controlling cell spreading and polarization (PubMed:24163421). {ECO:0000250|UniProtKB:Q9UIK4, ECO:0000269|PubMed:24163421}. |
3D-structure;Alternative splicing;Apoptosis;ATP-binding;Calmodulin-binding;Cytoplasm;Cytoplasmic vesicle;Kinase;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:13143; |
autophagosome lumen [GO:0034423]; cytoplasm [GO:0005737]; cytoplasmic vesicle [GO:0031410]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; nucleus [GO:0005634]; ATP binding [GO:0005524]; calmodulin binding [GO:0005516]; identical protein binding [GO:0042802]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; anoikis [GO:0043276]; intracellular signal transduction [GO:0035556]; neutrophil migration [GO:1990266]; positive regulation of apoptotic process [GO:0043065]; positive regulation of eosinophil chemotaxis [GO:2000424]; positive regulation of neutrophil chemotaxis [GO:0090023]; protein phosphorylation [GO:0006468]; regulation of intrinsic apoptotic signaling pathway [GO:2001242] |
18535204_DAPK2 was substantially up-modulated during late erythropoiesis 21497605_The crystal and solution structures of murine DAPK2 were determined in the presence of the autoinhibitory domain, with and without bound nucleotides in the active site. Dimers of DAPK2 had a conformation that did not permit protein substrate binding. 22580283_These results suggest that DAPK2 is one of the targets of cGK-I in apoptosis induction. 24163421_The defect in chemotaxis in DAPK2-inactive granulocytes is likely a result of reduced polarization of the cells, mediated by a lack of MLC phosphorylation, resulting in radial F-actin and pseudopod formation. 24906443_DAPK2 is strongly and specifically expressed in interstitial cells of the cortex, providing a useful marker for this important cell population 26038578_This study links adipocyte expression of an autophagy-regulating kinase, lysosome-mediated clearance and fat cell lipid accumulation; it demonstrates obesity-related attenuated autophagy in adipocytes, and identifies DAPK2 dependence in this regulation. 27653365_Thyroid hormone promotes selective autophagy via induction of DAPK2-SQSTM1 cascade, which in turn protects hepatocytes from diethylnitrosamine-induced hepatotoxicity or carcinogenesis. |
ENSG00000035664 |
DAPK2 |
203.099889 |
0.1127256975 |
-3.149112 |
0.21466971 |
219.443768 |
0.00000000000000000000000000000000000000000000000011959488499928250674385812205477099423221262912624743314606301536361620589343109142151329702973948557569706278822645350280403676868079543282874510623514652252197265625000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000195878418097749569817353944428201236807442099719053768207243430002468094352574774521629056419114419166966593001595088596733518215842195786535739898681640625000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
40.6086091746593 |
6.06479003901288 |
360.242695845774 |
33.6862565370237 |
| ENSMUSG00000032492 |
19228 |
Pth1r |
protein_coding |
P41593 |
FUNCTION: Receptor for parathyroid hormone and for parathyroid hormone-related peptide (PubMed:8197183). The activity of this receptor is mediated by G proteins which activate adenylyl cyclase and also a phosphatidylinositol-calcium second messenger system. {ECO:0000250|UniProtKB:Q03431, ECO:0000269|PubMed:8197183}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:19228; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; brush border membrane [GO:0031526]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; G protein-coupled peptide receptor activity [GO:0008528]; parathyroid hormone receptor activity [GO:0004991]; peptide hormone binding [GO:0017046]; protein homodimerization activity [GO:0042803]; protein self-association [GO:0043621]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; adenylate cyclase-modulating G protein-coupled receptor signaling pathway [GO:0007188]; bone mineralization [GO:0030282]; bone resorption [GO:0045453]; cell maturation [GO:0048469]; cell population proliferation [GO:0008283]; cell surface receptor signaling pathway [GO:0007166]; cellular calcium ion homeostasis [GO:0006874]; chondrocyte differentiation [GO:0002062]; G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger [GO:0007187]; in utero embryonic development [GO:0001701]; negative regulation of cell population proliferation [GO:0008285]; ossification [GO:0001503]; osteoblast development [GO:0002076]; phospholipase C-activating G protein-coupled receptor signaling pathway [GO:0007200]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of inositol phosphate biosynthetic process [GO:0060732]; skeletal system development [GO:0001501] |
12413769_The variety of the PTH/PTHrP receptor mRNA expression during hair cycling suggest that PTH, PTHrP and their receptors might participate in the regulation of hair cycle in mice skin. 12586782_findings indicate that the parathyroid hormone type 1 receptor is required for the development of normal cardiomyocyte function 12705476_Pthr immunoreactivity was demonstrated in epithelial cells of striated, interlobular and main excretory ducts of the sublingual gland. It was absent in the epithelial cells of the intercalated ducts. 12960101_Smooth-muscle-specific overexpression of PTH/PTHrP type 1 receptor resulted in more vasodilation to saline volume expansion and less vasoconstriction to ANG II. Can act as endogenous vasorelaxant factor to modulate renal responses to vasoactive stimuli. 15459318_Gsalpha is the primary mediator of the actions of the Pthrp receptor in growth plate chondrocytes. 15611335_The PTH/PTHrP signaling is a major regulator of the ontogeny of the bone marrow and its stromal tissue, and of the skeletal stem cell compartment. 15771625_Upregulation of PTH/PTHrP receptor mRNA expression in the developing growth plate. 15880431_Reduced smooth muscle appears to underlie the majority of nipple deficiencies in mice with lower levels of the Ppr expression 16476422_An unexpected interaction is found between parathyroid hormone-related protein (PTHrP) and Runx2 in controlling the rate of chondrocyte proliferation. 16574786_PTH1R forms a complex with the transport regulatory proteins, importin alpha(1) and importin beta, and nuclear PTH1R is associated with the nucleoplasm 16672315_parathyroid hormone-related peptide, by interaction with the PTH1 receptor, can increase tubulointerstitial cell survival and seems to act as a proinflammatory and profibrogenic factor in the FA-damaged kidney 16675960_fetal skins of the PTHrP and PPR knockouts (KOs) had reciprocal increases in the length, diameter, and density of capillaries 16783882_Collectively, these results indicate that the renal PTHrP/PTH1R system is upregulated in streptozotozin-induced diabetes in mice, and appears to adversely affect the outcome of diabetic renal disease. 17185824_PTHR1 signaling in osteocytes is sufficient for inhibition of Sost expression and leads to a concomitant increase in bone mass. 17317770_PTH1R shuttles from the nucleus to the cytoplasm under normal physiological conditions and that this nuclear-cytoplasmic transport is dependent upon importin-alpha/beta and CRM1 17500070_constitutive activation of PTH/PTHrP receptor signaling in osteoblastic cells suppresses unloading-induced bone loss specifically through the regulation of osteoclastic activity. 17525124_betaAr-dependent late phase of ERK activation by PTH requires the participation of betaAr2 and PTH1R internalization 17526947_PTH(1-34) and PTHrP(7-34) induce a conformational change in the PTH1R that promotes arrestin binding and dissociates NHERF1 from PTH1R internalization. 18946036_As compared with PTH(1-34), M-PTH(1-34) bound to R(0) PTHR conformation with higher affinity and induced larger increases in trabecular bone volume and greater increases in cortical bone turnover 19574446_The regulation of PTH1R expression by endogenous PTHrP and the associated effects on vascular smooth muscle cell proliferation, was investigated. 19761883_Ihh has both PTHrP-dependent and -independent functions during postnatal endochondral bone development. 20139972_TGFBR2 forms an endocytic complex with PTH1R in response to PTH and regulates signalling by PTH and TGF-beta. TGFBR2 directly phosphorylates the PTH1R cytoplasmic domain, which modulates PTH-induced endocytosis of the PTH1R-TGFBR2 complex. 20501677_PLC signaling through the PTHR is required for skeletal homeostasis. 20808842_PTH receptor 1 signaling in T cells plays an essential role in parathyroid hormone induced bone loss 20929987_In primary calvarial osteoblasts from 2-week-old phosphorylation-deficient or wild-type mice, PTH and PTH-related peptide receptor decreased the levels of phosphorylated extracellular signal-regulated kinases 1/2 and cyclin D1. 20974122_Adipocyte-PTHR, an all-trans retinoic acid-responsive gene, was found to be an up-stream regulator of pleiotrophin. 21140374_PTH receptor signaling influences cortical bone through actions on osteocytes and defines the role of Wnt signaling in PTH receptor action. 21173257_Inactivation of PPR in chondrocytes postnatally leads to accelerated differentiation of chondrocytes, followed by disappearance of the growth plate. 21726676_PTH receptor signaling has the potential to modulate the endocrine and auto/paracrine functions of osteocytes by regulating FGF23 through cAMP- and Wnt-dependent mechanisms. 21866553_These results indicate that constitutive activation of PTH/PTHrP receptor in differentiated osteoblasts enhances bone marrow ablation-induced recruitment, proliferation, and differentiation of osteoprogenitors. 22338074_PTHR1 phosphorylation does not play a major role in the anabolic action of PTH. 22393015_direct PPR signaling in T cells plays an important role in PTH-induced bone anabolism by promoting T-cell production of Wnt10b 22538810_Data implicate a functional interaction between parathyroid hormone type 1 receptor (PTHR) and beta(2)-adrenergic receptor (beta(2)AR) controls bone formation and parathyroid hormone (PTH) anabolism. 23235147_The IRE1alpha-XBP1 pathway positively regulates parathyroid hormone (PTH)/PTH-related peptide receptor expression and is involved in PTH-induced osteoclastogenesis. 23300710_LCPUFAs, EPA and DHA, can activate PTH1R receptor at nanomolar concentrations and consequently provide a putative molecular mechanism for the action of fatty acids in bone 23533279_PTH-induced phosphorylation of the PTHR1 in is important in regulating acute ligand responses 23729679_Data indicate that PTH/PTH-related peptide (PTHrP) type 1 receptor (PPR) signaling in osteocytes is required for bone remodeling. 23963454_Data suggest that parathyroid hormone (PTH) receptor signaling could enhance the therapeutic potential of the pathway. 24821091_Cbfbeta functions in upregulating Ihh expression to promoter chondrocyte proliferation and osteoblast differentiation, and inhibiting PPR expression to enhance chondrocyte differentiation. 24877630_PTH receptor signaling enhances bone resorption by directly regulating Rankl gene expression in osteocytes. 25196529_These studies reveal that PTH1R signaling in osteoblasts regulates cartilaginous growth plate for postnatal growth of bone. 25228504_Osteocytic gp130 is required to maintain PTH1R expression in the osteoblast lineage, and for the stimulation of osteoblast differentiation that occurs in response to PTH. 25529820_PTH1R is an important component of mechanical signal transduction in osteocytic MLO-Y4 cells. 26428657_PTH1R regulates systemic mineral ion homeostasis and induction of FGF23. 26620087_Ihh and PTH1R signaling in limb mesenchyme are both essential to regulate proper development of digit structures, although they appear to use different mechanisms. 26647715_we propose that PTH has a direct effect on osteoblasts and osteoclasts, and that this effect is mediated through PTH1R, thus contributing to bone remodeling 26669699_PTHrP and PTH mediate wasting through a common mechanism involving PTHR. 26912788_A critical role for SNX27-retromer mediated transport of PTHR in normal bone development. 27068606_cells expressing osterix are mesenchymal progenitors contributing to all relevant cell types during morphogenesis. 27070462_Activation of PTHrP-cyclic AMP-CREB1 signaling following p53 loss is essential for osteosarcoma initiation and maintenance. 27704638_Pth1r in DMP1-8kb-expressing cells is required to maintain basal levels of bone resorption but is dispensable for the catabolic action of chronic PTH elevation. 28727763_Results provide in vivo evidence of a role for RAMP2 in placental development distinct from the RAMP2-calcitonin receptor-like receptor/adrenomedullin signaling paradigm; decreases Pthr1 expression and causes a blunted response to systemic parathyroid hormone; and identify additional pathways underlying the endocrine and fertility defects of the previously characterized Ramp2 heterozygous adult females. 28934958_ZFPM2, LEF1, NR4A2, HAS2, and RHOC might be potential targets of PTHR1 in osteosarcoma. 29121993_Knock-down of PTHR1 reveals its pro-malignant effects in osteosarcoma cells. 30053608_acute calcemic response to PTH in vivo in mice lacking PTH receptors in osteocytes vs littermate controls, is resported. 30682096_data demonstrate that PPR expression along the osteoblast lineage is essential for exercise to improve the mechanical properties of cortical bone. Furthermore, the influence of PPR activation on material properties is unique to exercise and not during normal growth and development. 31382618_the relieving effect of Smo knockout mice was attenuated when Pth1r knockout was also applied. Our data suggest a chondrocyte protective effect of suppressing Ihh signaling in temporomandibular joint osteoarthritis (OA) cartilage which is dependent on PTH1R signaling 31430259_SIK inhibition is central to PTH1R action in bone development and remodeling 31536386_PTHR1 may play important roles in progression of Osteosarcoma by activating miR-124-3p-AR-Tgfb1i1, miR-27a-3p-PPARG-Abca1, and miR-103/590-3p-AXIN2 axes. 31987988_Osteocytes' expression of the PTH/PTHrP receptor has differing effects on endocortical and periosteal bone formation during adenine-induced CKD. 32039485_Primary cilia mediate parathyroid hormone receptor type 1 osteogenic actions in osteocytes and osteoblasts via Gli activation. 32160132_PTH/PTHrP Receptor Signaling Restricts Arterial Fibrosis in Diabetic LDLR(-/-) Mice by Inhibiting Myocardin-Related Transcription Factor Relays. 32585127_Role of PTH1R Signaling in Prx1(+) Mesenchymal Progenitors during Eruption. 33361757_Kindlin-2 regulates skeletal homeostasis by modulating PTH1R in mice. 33434347_Pleckstrin homology (PH) domain and Leucine Rich Repeat Phosphatase 1 (Phlpp1) Suppresses Parathyroid Hormone Receptor 1 (Pth1r) Expression and Signaling During Bone Growth. 33511766_Parathyroid hormone type 1 receptor regulates osteosarcoma K7M2 Cell growth by interacting with angiotensinogen. 34740971_Activation of PTH1R alleviates epididymitis and orchitis through Gq and beta-arrestin-1 pathways. 35490308_Loss of Parathyroid Hormone Receptor Signaling in Osteoprogenitors Is Associated With Accumulation of Multiple Hematopoietic Lineages in the Bone Marrow. |
ENSG00000160801 |
PTH1R |
7589.391682 |
0.4903522268 |
-1.028110 |
0.06819388 |
223.880351 |
0.00000000000000000000000000000000000000000000000001288287898102140016950720222744439562652761367399730482230613225910474705459392587212187733854135239575207882708326921116431329830676055792082479456439614295959472656250000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000022048540745945837870871460330536179430604609235833434074264739280020604923952140321595447267315431310568794036722961766656264659403063888021279126405715942382812500000000000000000000000000000000000000000000000000 |
Yes |
No |
4982.88971908336 |
235.620925325674 |
10161.8580409969 |
357.142898118316 |
| ENSMUSG00000032500 |
245038 |
Dclk3 |
protein_coding |
Q8BWQ5 |
|
ATP-binding;Cytoplasm;Kinase;Nucleotide-binding;Nucleus;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
This gene encodes a member of the protein kinase superfamily and the doublecortin family. Differently from the other two closely related family members (DCLK1 and DCLK2), the protein encoded by this gene contains only one N-terminal doublecortin domain and is unable to bind microtubules and to regulate microtubule polymerization. The protein contains a C-terminal serine/threonine protein kinase domain, which shows substantial homology to Ca2+/calmoduline-dependent protein kinase, and a serine/proline-rich domain in between the doublecortin and the protein kinase domains, which mediates multiple protein-protein interactions. [provided by RefSeq, Sep 2010]. |
mmu:245038; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; intracellular signal transduction [GO:0035556]; negative regulation of protein localization to nucleus [GO:1900181]; peptidyl-serine phosphorylation [GO:0018105]; protein localization to nucleus [GO:0034504] |
29534157_This study suggest that the presence of DCLK3 in striatal neurons may play a key role in transcription regulation and chromatin remodelling in these brain cells, and show that reduced expression of the kinase in Huntington's disease could render the striatum highly vulnerable to neurodegeneration. |
ENSG00000163673 |
DCLK3 |
17.851948 |
0.2567769646 |
-1.961412 |
0.73141196 |
6.909715 |
0.00857286458878812190897988898541370872408151626586914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0294541044920322882261221053568078787066042423248291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.26761991650099 |
3.84438577108625 |
28.3032394534923 |
10.2917469412873 |
| ENSMUSG00000032528 |
22354 |
Vipr1 |
protein_coding |
P97751 |
FUNCTION: This is a receptor for VIP. The activity of this receptor is mediated by G proteins which activate adenylyl cyclase. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:22354; |
plasma membrane [GO:0005886]; receptor complex [GO:0043235]; G protein-coupled peptide receptor activity [GO:0008528]; peptide hormone binding [GO:0017046]; vasoactive intestinal polypeptide receptor activity [GO:0004999]; adenylate cyclase-modulating G protein-coupled receptor signaling pathway [GO:0007188]; cell surface receptor signaling pathway [GO:0007166]; G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger [GO:0007187] |
11859927_VPAC1 suggested as mediator of the neuroprotective effects of VIP 12409228_VIP modulation of neural crest differentiation is mediated via VPAC1 and high expression of VPAC1 induces differentiation in and decreases tumorigenicity of neuroblastoma cells. 15632701_VIP attenuated the experimental acute pancreatitis enzymatically and morphologically by inhibiting proinflammatory cytokine production from monocytes mainly through the VPAC1-R. 17401158_In neonatal mice, a defect occurs in the signal transduction mechanism of the VPAC-1 VIP receptor and cortical astrocytes from neonatal brains are dysfunctional. 18442091_VIP inhibits beta-amyloid protein (Abeta)-induced neurodegeneration by indirectly inhibiting production of a wide panel of inflammatory and neurotoxic agents by activated microglia cells. 18616823_Data show that VIP and its receptors, VPAC1R and VPAC2R, are expressed in hippocampus, cortex, amygdala and hypothalamus which are involved in learned behaviors. 19650041_In contrast, selective VPAC(1) receptor activation shifts NSPC fate toward granule cell neurogenesis without any trophism. 20150284_VIP and its receptors (VPAC1, VPAC2) were identified in type II taste cells of the taste bud, and VIP knockout mice exhibit enhanced taste preference to sweet tastants. 20449688_Data describe PACAP, vasoactive intestinal polypeptide, and PAC1, VPAC1, VPAC2 transcripts or protein expression in urothelium and detrusor smooth muscle and lumbosacral dorsal root ganglia in NGF-overexpressing and wildtype mice. 20951679_VPAC(1)-R activation aggravates atherosclerotic lesion formation in apolipoprotein E-deficient mice through enhanced inflammatory activity in the vessel wall. 21295288_VIP enhancement of the severity of dextran sodium sulfate-induced colitis is mediated solely by VPAC1 receptors in mice. 21697765_Homozygous deletion of VPAC1 resulted in fetal, neonatal, and postweaning death owing to failure to thrive, intestinal obstruction, and hypoglycemia. 21878358_Data support the notion that both VPAC1 and VPAC2 receptors are dynamically regulated by Ikaros, a master transcriptional regulator for thymocyte differentiation, during early thymic development. 22059987_results support that decline in VIP/VPAC local levels may influence survival/apoptosis intracellular set point in NOD acinar cells and their clearance, contributing to gland homeostasis loss in a model of Sjogren's syndrome 22669509_VPAC1R mRNA expression was significantly decreased 3 days after ischemia induced by bilateral common carotid artery occlusion 22700375_Cyclophosphamide-induced cystitis decreased VPAC1 receptor transcript expression in the urothelium of WT (4 h, 48 h, & chronic) & NGF-OE mice. 27357191_This study demonstrated that VPAC1 receptor (Vipr1)-deficient mice exhibit ameliorated experimental autoimmune encephalomyelitis, with specific deficits in the effector stage. 30407703_follicle-associated epithelium-adjacent villus epithelium in ileum of Crohn's disease possessed more mucosal mast cells and mucosal mast cells expressing VPAC1 but not VPAC2, compared to controls 36328148_VPAC1 and VPAC2 receptor deficiencies negatively influence pregnancy outcome through distinct and overlapping modulations of immune, trophoblast and vascular functions. |
ENSG00000114812 |
VIPR1 |
10.070637 |
0.1405609798 |
-2.830732 |
0.95555462 |
8.834201 |
0.00295637085342424126357951941201918089063838124275207519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0118503791682521182615506205593192134983837604522705078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.36158696602544 |
1.68107408052879 |
16.8011561144947 |
7.35167472454717 |
| ENSMUSG00000032586 |
22036 |
Traip |
protein_coding |
Q8VIG6 |
FUNCTION: E3 ubiquitin ligase required to protect genome stability in response to replication stress (PubMed:33590678). Acts as a key regulator of interstrand cross-link repair, which takes place when both strands of duplex DNA are covalently tethered together, thereby blocking replication and transcription (By similarity). During mitosis, controls the choice between the two pathways of replication-coupled interstrand-cross-link repair by mediating ubiquitination of MCM7 subunit of the CMG helicase complex (PubMed:33590678). Short ubiquitin chains on MCM7 promote recruitment of DNA glycosylase NEIL3 (By similarity). If the interstrand cross-link cannot be cleaved by NEIL3, the ubiquitin chains continue to grow on MCM7, promoting the unloading of the CMG helicase complex by the VCP/p97 ATPase, enabling the Fanconi anemia DNA repair pathway (By similarity). Only catalyzes ubiquitination of MCM7 when forks converge (By similarity). Also involved in the repair of covalent DNA-protein cross-links (DPCs) during DNA synthesis: promotes ubiquitination of DPCs, leading to their degradation by the proteasome (By similarity). Has also been proposed to play a role in promoting translesion synthesis by mediating the assembly of 'Lys-63'-linked poly-ubiquitin chains on the Y-family polymerase POLN in order to facilitate bypass of DNA lesions and preserve genomic integrity (By similarity). The function in translesion synthesis is however controversial (By similarity). Acts as a regulator of the spindle assembly checkpoint (By similarity). Also acts as a negative regulator of innate immune signaling by inhibiting activation of NF-kappa-B mediated by TNF (PubMed:17544371, PubMed:22945920). Negatively regulates TLR3/4- and RIG-I-mediated IRF3 activation and subsequent IFNB1 production and cellular antiviral response by promoting 'Lys-48'-linked polyubiquitination of TNK1 leading to its proteasomal degradation (By similarity). {ECO:0000250|UniProtKB:Q6NRV0, ECO:0000250|UniProtKB:Q9BWF2, ECO:0000269|PubMed:17544371, ECO:0000269|PubMed:22945920, ECO:0000269|PubMed:33590678}. |
Alternative splicing;Chromosome;Coiled coil;Cytoplasm;DNA damage;DNA repair;Metal-binding;Nucleus;Reference proteome;Repeat;Transferase;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000269|PubMed:17544371, ECO:0000269|PubMed:22945920}. |
|
mmu:22036; |
nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; site of DNA damage [GO:0090734]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin-protein transferase activity [GO:0004842]; cellular response to DNA damage stimulus [GO:0006974]; negative regulation of interferon-beta production [GO:0032688]; negative regulation of NF-kappaB transcription factor activity [GO:0032088]; negative regulation of tumor necrosis factor-mediated signaling pathway [GO:0010804]; protein ubiquitination [GO:0016567]; protein-DNA covalent cross-linking repair [GO:0106300]; replication fork processing [GO:0031297]; signal transduction [GO:0007165] |
17927961_TRIP is an essential factor during early mouse embryonic development in vivo 22945920_TRIP as a negative regulator in TLR3/4- and RIG-I-triggered antiviral responses and suggested TRIP as a potential target for the intervention of diseases with uncontrolled IFN-beta production. 27405720_Taken together, these data indicate that TRAIP plays important roles in oocyte meiosis regulation. |
ENSG00000183763 |
TRAIP |
302.479030 |
2.3873838606 |
1.255431 |
0.17044136 |
53.508079 |
0.00000000000025753160728896639768241752918489621972759559542787144437170354649424552917480468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000055327523867779064563502137295315108254586955460752051294548436999320983886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
458.392610298766 |
54.2810022830567 |
192.006244934958 |
17.3421473766478 |
| ENSMUSG00000032589 |
12217 |
Bsn |
protein_coding |
O88737 |
FUNCTION: Scaffold protein of the presynaptic cytomatrix at the active zone (CAZ) which is the place in the synapse where neurotransmitter is released (PubMed:12628168, PubMed:12628169, PubMed:19812333). After synthesis, participates in the formation of Golgi-derived membranous organelles termed Piccolo-Bassoon transport vesicles (PTVs) that are transported along axons to sites of nascent synaptic contacts (By similarity). At the presynaptic active zone, regulates the spatial organization of synaptic vesicle cluster, the protein complexes that execute membrane fusion and compensatory endocytosis (By similarity). Functions also in processes other than assembly such as the regulation of specific presynaptic protein ubiquitination by interacting with SIAH1 or the regulation of presynaptic autophagy by associating with ATG5 (By similarity) (PubMed:28231469). Mediates also synapse to nucleus communication leading to reconfiguration of gene expression by associating with the transcriptional corepressor CTBP1 and by subsequently reducing the size of its pool available for nuclear import (By similarity). {ECO:0000250|UniProtKB:O88778, ECO:0000250|UniProtKB:Q9UPA5, ECO:0000269|PubMed:12628168, ECO:0000269|PubMed:12628169, ECO:0000269|PubMed:19812333, ECO:0000269|PubMed:28231469}. |
Alternative splicing;Cell projection;Coiled coil;Cytoplasm;Cytoplasmic vesicle;Cytoskeleton;Glycoprotein;Lipoprotein;Membrane;Metal-binding;Methylation;Myristate;Phosphoprotein;Reference proteome;Repeat;Synapse;Zinc;Zinc-finger |
|
|
mmu:12217; |
axon [GO:0030424]; axon cytoplasm [GO:1904115]; cell surface [GO:0009986]; cochlear hair cell ribbon synapse [GO:0098683]; cytoskeleton of presynaptic active zone [GO:0048788]; cytosol [GO:0005829]; dendrite [GO:0030425]; excitatory synapse [GO:0060076]; extrinsic component of membrane [GO:0019898]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; Golgi-associated vesicle [GO:0005798]; hippocampal mossy fiber to CA3 synapse [GO:0098686]; inhibitory synapse [GO:0060077]; neuron projection terminus [GO:0044306]; neuronal cell body [GO:0043025]; piccolo-bassoon transport vesicle [GO:1990257]; postsynaptic density [GO:0014069]; presynapse [GO:0098793]; presynaptic active zone [GO:0048786]; presynaptic active zone cytoplasmic component [GO:0098831]; ribbon synapse [GO:0097470]; Schaffer collateral - CA1 synapse [GO:0098685]; synapse [GO:0045202]; synaptic membrane [GO:0097060]; synaptic vesicle [GO:0008021]; trans-Golgi network [GO:0005802]; dynein light chain binding [GO:0045503]; metal ion binding [GO:0046872]; structural constituent of presynaptic active zone [GO:0098882]; transcription corepressor binding [GO:0001222]; axo-dendritic transport [GO:0008088]; presynapse to nucleus signaling pathway [GO:0099526]; presynaptic active zone assembly [GO:1904071]; protein localization to synapse [GO:0035418]; regulation of synaptic vesicle cycle [GO:0098693]; regulation of ubiquitin protein ligase activity [GO:1904666]; retrograde axonal transport [GO:0008090]; synapse organization [GO:0050808]; synaptic vesicle clustering [GO:0097091] |
12628168_Deficiency of Bassoon, a major component of the photoreceptor ribbon, results in impaired photoreceptor synaptic transmission, abnormal dendritic branching of neurons postsynaptic to photoreceptors, and the formation of ectopic synapses. 12628169_Loss of Bassoon in Bsn mutant mice causes a reduction in normal synaptic transmission, which can be attributed to the inactivation of a significant fraction of glutamatergic synapses. 16452644_the absence of the functional presynaptic protein Bassoon causes disturbance in the formation of normal basal cortical activation patterns and thereby in the functional cortical architecture 17652465_altered neuronal activation pattern in the cortex is reflected not only in defined cytoarchitectural anomalies but also in metabolic disturbances in the glutamine-glutamate and N-acetyl aspartate metabolism in Bsn mutants 18095322_true cone-selective type 1 and 2 OFF cone bipolar cells did not show sprouting in the Bassoon mouse mutant 20188438_Our findings reveal a novel function of Bassoon in the early formation and delivery of precursor spheres to nascent ribbon synaptic sites 20332206_piccolo and bassoon play a redundant role in synaptic vesicle clustering in nerve terminals without directly participating in neurotransmitter release. 20421286_Results support the notion that Bassoon is important for organizing the presynaptic active zone during the postnatal maturation of glutamatergic synapses. 21935677_hippocampal overgrowth in Bassoon-mutant mice arises from a dysregulation of neurogenesis and apoptosis that might be associated with unbalanced BDNF levels. 23295987_proper assembly of ribbon synapses depend on Bassoon 23403927_a novel role for Bassoon and Piccolo as critical regulators of presynaptic ubiquitination and proteostasis. 23467361_This studyy demonistrated that reduced bassoon expression or complete lack of full-length bassoon impaired sound encoding to a similar extent, which is consistent with the comparable reduction of the readily releasable vesicle pool 23516560_phosphorylation-induced 14-3-3 binding to Bassoon modulates its anchoring to the presynaptic cytomatrix 24442636_Authors conclude that bassoon promotes vesicular replenishment and, consequently, a large standing pool of readily releasable synaptic vesicles at the endbulb synapse. 24698275_These data establish Bassoon as a major regulator of the molecular composition of the presynaptic neurotransmitter release sites. 27321892_Piccolo puncta sandwiched Bassoon puncta and aligned in a Piccolo-Bassoon-Piccolo structure in adult neuromuscular junctions (NMJs). P/Q-type voltage-gated calcium channel (VGCC) puncta colocalized with Bassoon puncta. The P/Q-type VGCC and Bassoon protein levels decreased significantly in NMJs from aged mouse. In contrast, the Piccolo levels in NMJs from aged mice were comparable to levels in adult mice. 29915867_The expression of Bassoon in excitatory forebrain neurons is required for the normal maturation of the dentate gyrus and important for spatial and contextual memory. 32364493_Parkin contributes to synaptic vesicle autophagy in Bassoon-deficient mice. 32651614_Bassoon inhibits proteasome activity via interaction with PSMB4. 32718208_BSN (bassoon) and PRKN/parkin in concert control presynaptic vesicle autophagy. 33811381_Genetic disruption of bassoon in two mutant mouse lines causes divergent retinal phenotypes. 34320240_Dopaminergic neurons establish a distinctive axonal arbor with a majority of non-synaptic terminals. 34360710_The Presynaptic Scaffold Protein Bassoon in Forebrain Excitatory Neurons Mediates Hippocampal Circuit Maturation: Potential Involvement of TrkB Signalling. |
ENSG00000164061 |
BSN |
580.032974 |
8.4604003668 |
3.080726 |
1.03652056 |
7.577388 |
0.00591050238272063446776094508550158934667706489562988281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0214456341814198911399635960606246953830122947692871093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1002.72731581807 |
784.018800660729 |
118.520078523641 |
66.8410188007153 |
| ENSMUSG00000032641 |
14760 |
Gpr19 |
protein_coding |
Q61121 |
FUNCTION: Orphan receptor. |
Alternative splicing;Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:14760; |
cilium [GO:0005929]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; G protein-coupled receptor signaling pathway [GO:0007186] |
34789778_Gpr19 is a circadian clock-controlled orphan GPCR with a role in modulating free-running period and light resetting capacity of the circadian clock. |
ENSG00000183150 |
GPR19 |
219.346996 |
2.2117946079 |
1.145217 |
0.19564476 |
34.006539 |
0.00000000549271634669641321055685176063566876880628342405543662607669830322265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000693165330512674082421976473437907895203125008265487849712371826171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
295.806691547026 |
40.2375927465839 |
133.740579024973 |
14.0289076853786 |
| ENSMUSG00000032648 |
19309 |
Pygm |
protein_coding |
Q9WUB3 |
FUNCTION: Allosteric enzyme that catalyzes the rate-limiting step in glycogen catabolism, the phosphorolytic cleavage of glycogen to produce glucose-1-phosphate, and plays a central role in maintaining cellular and organismal glucose homeostasis. {ECO:0000250|UniProtKB:P11217}. |
Acetylation;Allosteric enzyme;Carbohydrate metabolism;Glycogen metabolism;Glycosyltransferase;Nucleotide-binding;Phosphoprotein;Pyridoxal phosphate;Reference proteome;Transferase |
|
This gene encodes a glycolysis enzyme found in muscle. Highly similar enzymes encoded by different genes are found in liver and brain. The encoded protein is involved in regulating the breakdown of glycogen to glucose-1-phosphate, which is necessary for ATP generation. [provided by RefSeq, Dec 2015]. |
mmu:19309; |
cytoplasm [GO:0005737]; sarcoplasmic reticulum [GO:0016529]; Z disc [GO:0030018]; AMP binding [GO:0016208]; carbohydrate binding [GO:0030246]; glycogen phosphorylase activity [GO:0008184]; linear malto-oligosaccharide phosphorylase activity [GO:0102250]; organic cyclic compound binding [GO:0097159]; pyridoxal phosphate binding [GO:0030170]; SHG alpha-glucan phosphorylase activity [GO:0102499]; cellular calcium ion homeostasis [GO:0006874]; glycogen catabolic process [GO:0005980]; glycogen metabolic process [GO:0005977]; response to cAMP [GO:0051591]; response to hypoxia [GO:0001666]; response to organic substance [GO:0010033] |
17689562_Data show that glycogen phosphorylase is irreversibly impaired by exposure to peroxynitrite, and suggest that the peroxynitrite-dependent inactivation of the enzyme could be due to the nitration of Tyr613 at the allosteric inhibitor site of the enzyme. 22730558_this is the first successful attempt to develop a genetically modified animal model of McArdle disease. 24269889_Data suggest that the irreversible inhibition of glycogen phosphorylase (GP) could represent one of the mechanisms that contribute to mercury-dependent muscle toxicity. 27156240_Results show that PYGM and RAC1 are altered in the dorsolateral prefrontal cortex in chronic schizophrenia and are controlled by NMDA signaling in the rodent cortex and cortical astrocytes suggesting an altered NMDA-dependent glycogenolysis in astrocytes in schizophrenia. |
ENSG00000068976 |
PYGM |
341.468625 |
0.4031357230 |
-1.310662 |
0.17080445 |
58.087633 |
0.00000000000002506973573979895818013668262708663223446085670242933218787584337405860424041748046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000006032578432679584417683878017514446728212368364907547402253840118646621704101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
191.327069867088 |
22.7046685366982 |
474.59716154273 |
40.6812086507746 |
| ENSMUSG00000032702 |
107351 |
Kank1 |
protein_coding |
E9Q238 |
Human_homologues FUNCTION: Involved in the control of cytoskeleton formation by regulating actin polymerization. Inhibits actin fiber formation and cell migration (PubMed:25961457). Inhibits RhoA activity; the function involves phosphorylation through PI3K/Akt signaling and may depend on the competitive interaction with 14-3-3 adapter proteins to sequester them from active complexes (PubMed:25961457). Inhibits the formation of lamellipodia but not of filopodia; the function may depend on the competitive interaction with BAIAP2 to block its association with activated RAC1 (PubMed:25961457). Inhibits fibronectin-mediated cell spreading; the function is partially mediated by BAIAP2. Inhibits neurite outgrowth. Involved in the establishment and persistence of cell polarity during directed cell movement in wound healing. In the nucleus, is involved in beta-catenin-dependent activation of transcription. Potential tumor suppressor for renal cell carcinoma. Regulates Rac signaling pathways (PubMed:25961457). {ECO:0000269|PubMed:16968744, ECO:0000269|PubMed:18458160, ECO:0000269|PubMed:19171758, ECO:0000269|PubMed:22084092, ECO:0000269|PubMed:25961457}. |
3D-structure;ANK repeat;Coiled coil;Proteomics identification;Reference proteome;Repeat |
|
|
Human_homologues hsa:23189; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; ruffle membrane [GO:0032587]; beta-catenin binding [GO:0008013]; actin cytoskeleton organization [GO:0030036]; cell population proliferation [GO:0008283]; negative regulation of actin filament polymerization [GO:0030837]; negative regulation of cell migration [GO:0030336]; negative regulation of insulin receptor signaling pathway [GO:0046627]; negative regulation of lamellipodium morphogenesis [GO:2000393]; negative regulation of neuron projection development [GO:0010977]; negative regulation of Rho protein signal transduction [GO:0035024]; negative regulation of ruffle assembly [GO:1900028]; negative regulation of substrate adhesion-dependent cell spreading [GO:1900025]; podocyte cell migration [GO:0090521]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; positive regulation of Wnt signaling pathway [GO:0030177]; positive regulation of wound healing [GO:0090303]; regulation of establishment of cell polarity [GO:2000114]; regulation of Rho protein signal transduction [GO:0035023] |
18458160_Data suggest that Kank negatively regulates the formation of actin stress fibers and cell migration through the inhibition of RhoA activity, which is controlled by binding of Kank to 14-3-3 in PI3K-Akt signaling. 29158259_Several key residues (i.e. Thr-1147, Leu-1152, Leu-1153, and Tyr-1154) at the C-terminal half of the KIF21A KBD peptide contact with the hydrophobic patch formed by Tyr-1176, Met-1209, Leu-1210, Leu-1213, and Leu-1248 from KANK1. 29217769_KANK1 binds to KIF21A through ankyrin repeat. 33253712_Tissue distribution and subcellular localization of the family of Kidney Ankyrin Repeat Domain (KANK) proteins. |
ENSG00000107104 |
KANK1 |
1290.720721 |
0.3207770224 |
-1.640357 |
0.14978080 |
115.097488 |
0.00000000000000000000000000749222041123075650811777437576779119182044058846714178094609398695855051464320695231435820460319519042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000004546673358719796063868705975054962257872532655907756511399311734268786910906356979467091150581836700439453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
617.276653903288 |
64.2009342617453 |
1924.31692658597 |
147.434390601469 |
| ENSMUSG00000032726 |
12163 |
Bmp8a |
protein_coding |
P34821 |
FUNCTION: Growth factor of the TGF-beta superfamily that plays important role in various biological processes, including spermatogenesis, osteogenesis, steroidogenesis as well as regulation of energy balance (PubMed:9463357, PubMed:12925636, PubMed:28465413). Initiates the canonical BMP signaling cascade by associating with type I receptor BMPR1A and type II receptor BMPR2. Once all three components are bound together in a complex at the cell surface, BMPR2 phosphorylates and activates BMPR1A (By similarity). In turn, BMPR1A propagates signal by phosphorylating SMAD1/5/8 that travel to the nucleus and act as activators and repressors of transcription of target genes. In addition, activates the SMAD2/3 pathway (PubMed:28465413). {ECO:0000250|UniProtKB:Q7Z5Y6, ECO:0000269|PubMed:12925636, ECO:0000269|PubMed:28465413, ECO:0000269|PubMed:9463357}. |
Alternative splicing;Chondrogenesis;Cytokine;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Growth factor;Osteogenesis;Reference proteome;Secreted;Signal |
|
This gene encodes a secreted ligand of the TGF-beta (transforming growth factor-beta) superfamily of proteins. Ligands of this family bind various TGF-beta receptors leading to recruitment and activation of SMAD family transcription factors that regulate gene expression. The encoded preproprotein is proteolytically processed to generate each subunit of the disulfide-linked homodimer. This protein may play a role in development of the reproductive system. Mice lacking a functional copy of this gene exhibit degeneration of germ cells and the epididymal epithelium. This gene may have arose from a gene duplication event and its gene duplicate is also present on chromosome 4. [provided by RefSeq, Jul 2016]. |
mmu:12163; |
extracellular space [GO:0005615]; BMP receptor binding [GO:0070700]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; BMP signaling pathway [GO:0030509]; cartilage development [GO:0051216]; germ cell development [GO:0007281]; osteoblast differentiation [GO:0001649]; positive regulation of pathway-restricted SMAD protein phosphorylation [GO:0010862]; SMAD protein signal transduction [GO:0060395]; spermatogenesis [GO:0007283]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
21277400_BMP-8 might be an essential player in bone metabolism, especially in response to glucocorticoids 25451979_Stromal Fgf10 and Bmp8a serve as potential paracrine factors for estrogen-dependent regulation of epithelial proliferation in the uterus. |
|
|
379.183269 |
0.1165807243 |
-3.100599 |
0.17405790 |
310.935128 |
0.00000000000000000000000000000000000000000000000000000000000000000000136620021564844020502888100510408739426547602585210142097470450952424877039364205149611762570360080129647748697584608612588367200930487648999874246482889044212078578940483368497904592686609248631 |
0.0000000000000000000000000000000000000000000000000000000000000000003853696608288340762191806032240324384908339220103425680406839172652040031271600697057682160826072121208886111852614923468565090424076672948171157388346462412619629345211791360270581208169460297 |
Yes |
No |
75.0915698217214 |
9.08162302801697 |
644.116514678555 |
51.0356243621032 |
| ENSMUSG00000032766 |
66066 |
Gng11 |
protein_coding |
P61953 |
FUNCTION: Guanine nucleotide-binding proteins (G proteins) are involved as a modulator or transducer in various transmembrane signaling systems. The beta and gamma chains are required for the GTPase activity, for replacement of GDP by GTP, and for G protein-effector interaction (By similarity). {ECO:0000250}. |
Cell membrane;Direct protein sequencing;Lipoprotein;Membrane;Methylation;Prenylation;Reference proteome;Transducer |
|
|
mmu:66066; |
heterotrimeric G-protein complex [GO:0005834]; G-protein beta-subunit binding [GO:0031681]; G protein-coupled receptor signaling pathway [GO:0007186] |
Human_homologues 17092487_Collectively, these results suggest a novel senescence pathway mediated by GNG11 in response to environmental cues. 28380310_expression of GNG11 induced the generation of reactive oxygen species (ROS) and abnormal nuclear morphology in SUSM-1 cells but not in HeLa cells |
ENSG00000127920 |
GNG11 |
157.114845 |
4.8659489062 |
2.282721 |
0.24720292 |
83.099882 |
0.00000000000000000007801035549698713780064949451148331851100370699640226455039149211501126046641729772090911865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000030082373036205268479369480162364623605006506593351157594073974621551315067335963249206542968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
266.468034484914 |
45.8825194856947 |
54.761782258946 |
7.5244652176212 |
| ENSMUSG00000032783 |
78733 |
Troap |
protein_coding |
B7ZNG4 |
FUNCTION: Could be involved with bystin and trophinin in a cell adhesion molecule complex that mediates an initial attachment of the blastocyst to uterine epithelial cells at the time of the embryo implantation. {ECO:0000250|UniProtKB:Q12815}. |
Cell adhesion;Cytoplasm;Phosphoprotein;Reference proteome |
|
|
mmu:78733; |
cytoplasm [GO:0005737]; cell adhesion [GO:0007155] |
Human_homologues 12049630_plays an important role in mammalian cells by associating with the microtubular cytoskeleton 18218922_the major function of tastin during mitosis is to maintain the structural and dynamic features of centrosomes, thereby contributing to spindle bipolarity. 29117881_these results indicate that TROAP suppresses cellular growth and migration in hepatocellular carcinoma 29956806_the present study provided evidence that TROAP serves a role in GC cells by promoting cell proliferation, cell cycle progression and invasion in vitro. Combined analysis of cell lines and datasets from a public database, demonstrated that TROAP overexpression may be used as a predictor of poor survival in patients with GC. 30284652_Elevated TROAP expression is an independent predictor of poor survival in liver cancer. 30431120_Study found that TROAP expression correlates with patient survival and speculated that it may be involved in prostate cancer progression. 31198787_The present study suggests that TROAP plays an important role in promoting the proliferation, invasion, and metastasis of breast cancer. 33500384_TROAP switches DYRK1 activity to drive hepatocellular carcinoma progression. 34287099_MiR-532-3p suppresses cell viability, migration and invasion of clear cell renal cell carcinoma through targeting TROAP. |
ENSG00000135451 |
TROAP |
504.336054 |
2.4615610496 |
1.299574 |
0.14463102 |
79.877263 |
0.00000000000000000039840483175828606266402815425631771146858276740831625046396213818411524698603898286819458007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000147652126456014900556374273721527872581890203254433024948610864157672040164470672607421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
721.488511757063 |
72.4508588329117 |
293.102018284513 |
22.7720141446623 |
| ENSMUSG00000032796 |
16772 |
Lama1 |
protein_coding |
P19137 |
FUNCTION: Binding to cells via a high affinity receptor, laminin is thought to mediate the attachment, migration and organization of cells into tissues during embryonic development by interacting with other extracellular matrix components. |
3D-structure;Basement membrane;Cell adhesion;Coiled coil;Direct protein sequencing;Disulfide bond;Extracellular matrix;Glycoprotein;Laminin EGF-like domain;Phosphoprotein;Pyrrolidone carboxylic acid;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:16772; |
basement membrane [GO:0005604]; cell-cell junction [GO:0005911]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; laminin complex [GO:0043256]; laminin-1 complex [GO:0005606]; laminin-3 complex [GO:0005608]; membrane [GO:0016020]; protein complex involved in cell-matrix adhesion [GO:0098637]; extracellular matrix structural constituent [GO:0005201]; glycosphingolipid binding [GO:0043208]; protein C-terminus binding [GO:0008022]; signaling receptor binding [GO:0005102]; animal organ morphogenesis [GO:0009887]; axon guidance [GO:0007411]; blood vessel morphogenesis [GO:0048514]; branching involved in salivary gland morphogenesis [GO:0060445]; camera-type eye development [GO:0043010]; cell adhesion [GO:0007155]; cell surface receptor signaling pathway [GO:0007166]; epithelial tube branching involved in lung morphogenesis [GO:0060441]; establishment of epithelial cell apical/basal polarity [GO:0045198]; morphogenesis of an epithelial sheet [GO:0002011]; neuron projection development [GO:0031175]; positive regulation of cell adhesion [GO:0045785]; positive regulation of integrin-mediated signaling pathway [GO:2001046]; positive regulation of muscle cell differentiation [GO:0051149]; protein phosphorylation [GO:0006468]; regulation of basement membrane organization [GO:0110011]; regulation of cell migration [GO:0030334]; regulation of embryonic development [GO:0045995]; retina development in camera-type eye [GO:0060041]; retinal blood vessel morphogenesis [GO:0061304]; tissue development [GO:0009888] |
8132708_Adhesion site formed by 'sinnnrwhsiyitrfgnmgs' is involved in formation of lung 'alveoli' in vitro. 9124607_Residues 'sinnnrwhsiyitrfgnmgs' promote adhesion in a heparin-sensitive but integrin-independent manner and binds approx. 155 kDa and 180 kDa cell surface proteins. 11827968_Exposure of cryptic domains in the alpha 1-chain of laminin-1 by elastase stimulates macrophages urokinase and matrix metalloproteinase-9 expression. 11969289_Expression and distribution of laminin alpha1 in embryonic and adult mouse tissues 12519763_mouse laminin alpha 1 gene has an upstream enhancer that defines its expression in parietal endoderm cells 12554653_Data suggest that surface-exposed, conserved residues of the laminin gamma1 chain interact with the N-terminal domain of agrin via hydrophobic and ionic interactions. 12631063_The absence of a alpha3(IV), alpha4(IV), alpha5(IV) network may stimulate reexpression of laminin-1 by Alport mouse endothelial cells and podocytes 14634001_the first characterization of the Lama1 promoter; identification of a regulatory elements that may account for the expression pattern of the endogenous protein in the mouse intestine 15452149_Mouse embryonic stem cells seeded on laminin formed hemispheric epithelial structures whose outermost layer terminally differentiated to an epithelium that resembled the visceral endoderm. 15668394_Deletion of binding domains impair fetal development by preventing conversion of stem cells to polarized epithelium. 15777791_The transient expression of laminin alpha1 may promote formation of sinusoids after partial hepatectomy. 16475793_Grb2-binding to syntrophin is increased by addition of either laminin-1 or isolated laminin alpha1 globular domain modules LG4-5. LG4-5 sequences are the region of laminin responsible for signaling. Alpha-dystroglycan is the laminin-signaling receptor. 16504180_suggest that LNalpha1 chain in part ameliorates the development of LNalpha2 chain deficient muscular dystrophy by retaining the binding sites for integrin alpha7Bbeta1D and alpha-dystroglycan, respectively 17307732_analysis of structure and activities reveal differences in laminin G-like domain interactions that should enable dissection of biological roles of different laminin ligands 17699809_Upregulation of Lama5 transcription and concentration of laminin alpha1 and alpha5 within Alport glomerular basement membrane thickenings contribute to abnormal permeabilities. 18215127_define sites for the interaction of acetylcholinesterase and laminin and suggest that the interaction plays a role in cell adhesion 18263654_The nonsulfated HNK-1 epitope on laminin-1 may regulate its binding and play a role in maintenance of the proper structure in the kidney basal lamina. 18570250_Data examined expression of laminin-1, perlecan, tenascin-C, and CSPGs and found a coordinated pattern of expression of these cues in the olfactory pathway. These appear to restrict axons to the pathway while promoting axon outgrowth within. 19200854_Our data suggest that doxycycline may provide a neuroprotection against global cerebral ischemia since it reduces perineuronal laminin degradation by inhibiting MMP-9 activity. 19416897_laminin-111 (alpha(1), beta(1), gamma(1)), which is expressed during embryonic development but absent in normal or dystrophic skeletal muscle, increased alpha(7)-integrin expression in mouse and DMD patient myoblasts 19657675_Data suggest that laminin alpha1 domains LG4-5 provide an autocrine signal necessary for the complete differentiation of a functional visceral endoderm and vital signals for the polarization of the epiblast. 19783738_Recovery of Lama1 expression or addition of exogenous laminin-111 to Shh(-/-);Gli3(-/-) embryos restores the myotomal basement membrane, demonstrating that laminin-111 is necessary and sufficient to initiate assembly of the myotomal basement membrane. 20048158_This first report of viable Lama1 mouse mutants emphasizes the importance of this gene in retinal development, and data suggest that hypomorphic mutations in human LAMA1 could lead to retinal disease. 20544910_we analyze the lifespan of laminin alpha2 chain-deficient mice with transgenic overexpression of laminin alpha1 chain 20657839_The results show that laminin alpha1LG domains and consequently their receptors have disparate functions in the neuromuscular system 21435454_Laminin alpha1 chain is not responsible for muscle disease in the mdx mouse model for Duchenne muscular dystrophy 21983115_Laminin alpha1 is essential for mouse cerebellar development. 21999428_Mutations in Lama1 allow developing retinal vessels to enter the vitreous where they anastomose with vessels of the hyaloid system which persist and expand 22027433_Laminin promotes epithelial formation of XEN cells, and up-regulation of visceral endoderm markers. 22270369_-DG prepared from lung- or testis-derived cells lacks the post-phosphoryl moiety and shows little laminin a1 and a2 binding activity. 22274713_Our data show that the absence of Lama1 has an impact on the Bergmann glia scaffold that aberrantly develops. 22761447_alpha1- and alpha5-containing laminins differentially regulate the sequential events to form epithelial tissues via beta1 integrin signals. 24720953_These findings suggest that LAMA1 plays a critical role in kidney function and kidney aging by regulating the mesangial cell population and mesangial matrix deposition through TGF-beta/Smad signaling. 25693840_interaction of alpha6beta1 in embryonic stem cells (ECCs) with laminin-1 activates alpha6beta1/CD151 signaling which programs ESCs toward the endothelial cells lineage fate 25812799_while MMP-9 is not essential for hypoxic-induced cerebral angiogenesis, it plays an important role in post-hypoxic vascular pruning by degrading laminin and claudin-5 27138650_Laminin regulates PDGFRbeta cell differentiation, fate determination, cell stemness and muscle development via gpihbp1. 27964993_Laminin is instructive and CaMKII is non-permissive for the formation of complex aggregates of acetylcholine receptors on myotubes in culture. 28173861_Laminin differentially regulates the stemness of type I and type II pericytes. 28983681_Altered LAMA1 expression in the aganglionic region may contribute to impaired ENCC migration, resulting in HD. These data could help in understanding the pathophysiologic interactions between LAMA1 and ENCC migration 29018955_laminin-1 does not have a positive effect on ENCC migration in HD mice on E12.5, in contrast to the phenomenon seen in normal mice gut specimens, where laminin-1 promotes ENCC migration during the same period. 29544677_Laminin alpha1 reduces muscular dystrophy in dy(2J) mice. 30232270_Lama1 as the genetic modifier of TGF-beta1 effector responses that significantly affects the development of pulmonary fibrosis. 31753553_Effect of laminin environments and tumor factors on the biology of myeloid dendritic cells. 31950499_Sleep deprivation regulates availability of PrP(C) and Abeta peptides which can impair interaction between PrP(C) and laminin and neuronal plasticity. 36476399_Effects of experimental glaucoma in Lama1[nmf223] mutant mice. |
ENSG00000101680 |
LAMA1 |
6.653591 |
70.5265780235 |
6.140095 |
1.64512607 |
13.498729 |
0.00023872513545239858081918604693072438749368302524089813232421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0012972747995757885619561022849666187539696693420410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.6553353522882 |
19.172318970693 |
0.193619706711793 |
0.232144350343536 |
| ENSMUSG00000032839 |
22063 |
Trpc1 |
protein_coding |
B2RPS7 |
FUNCTION: Thought to form a receptor-activated non-selective calcium permeant cation channel. Probably is operated by a phosphatidylinositol second messenger system activated by receptor tyrosine kinases or G-protein coupled receptors. Seems to be also activated by intracellular calcium store depletion. {ECO:0000256|ARBA:ARBA00003698}. |
ANK repeat;Ion transport;Membrane;Phosphoprotein;Proteomics identification;Receptor;Reference proteome;Repeat;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:22063; |
plasma membrane [GO:0005886]; receptor complex [GO:0043235]; ATPase binding [GO:0051117]; calcium channel activity [GO:0005262]; inositol 1,4,5 trisphosphate binding [GO:0070679]; signaling receptor binding [GO:0005102]; melanin biosynthetic process [GO:0042438]; positive regulation of release of sequestered calcium ion into cytosol [GO:0051281]; regulation of cytosolic calcium ion concentration [GO:0051480]; response to calcium ion [GO:0051592] |
14505576_Homer facilitates a physical association between TRPC1 and the IP(3)R that is required for the TRP channel to respond to signals 14530267_TRPC1 store dependence is suppressed by I-mfa1 15665854_TRPC1 is a component of the vertebrate MscCa, which is gated by tension developed in the lipid bilayer, as is the case in various prokaryotic mechanosensitive (Ms) channels. 16044320_The expression of TRPC1 and TRPC5 channels in mammalian temporal lobe structures suggests their involvement in neuronal plasticity, learning and memory. 16899508_We suggest that the anti-apoptotic effect of TRPC1 could potentially contribute to abnormal keratosis in DD. 17224452_dynamic assembly of TRPC1-STIM1-Orai1 ternary complex is involved in activation of SOC channel in response to internal Ca2+ store depletion 17478725_These results indicate that mouse sinoatrial node exhibits store-operated Ca(2+) activity (SOCC) which may be attributable to TRPC expression, and suggest that SOCCs may be involved in regulating pacemaker firing rate. 17647013_TRPC1 is not an obligatory component of store-operated and stretch-activated ion channel complexes in vascular smooth muscle cells. 17956991_TRPC1 is a critical component of the store-operated Ca(2+) channel in salivary gland acinar cells and is essential for neurotransmitter-regulation of fluid secretion 18268005_Homer 1 functions as an important scaffold for TRP channels and regulates mechanotransduction in skeletal muscle 18544631_TRPC1/caveolin-3 pathway contributes to the pathogenesis of Duchenne muscular dystrophy 18546016_Store-operated Ca(2+) entry in platelets occurs independently of transient receptor potential (TRP) C1. 18802326_expression in kidney reduced in diabetic db/db mice 18822346_Thus, TRPC1 may represent a promising target for pharmacological interventions to prevent or reduce glutamate-induced neuronal damage. 19332490_TRPC1 and STIM1 mediate capacitative Ca2+ entry in mouse pulmonary arterial smooth muscle cells. 19351713_Data suggest that TRPC channel 1 (TRPC1), besides acting as a store-operated channel, represents an essential component of stretch-activated channels in C2C12 skeletal myoblasts, as assayed by whole-cell patch-clamp and atomic force microscopic pulling. 19439599_study concludes TRPC1 and TRPC6 channels cooperate with TRPV4 channels to mediate mechanical hyperalgesia and primary afferent nociceptor sensitization, although they may have distinctive roles 19648473_canonical transient receptor potential 1 and canonical transient receptor potential 3 have roles in normal and asthmatic airway smooth muscle cells 19695701_MPTP induces neuronal loss by decreasing TRPC1 levels followed by the initiation of apoptosis mainly by disrupting mitochondrial membrane potential. 19797170_TRPC1 channels are critical for the adaptation to biomechanical stress and TRPC dysregulation leads to maladaptive cardiac hypertrophy and failure. 19812031_TRPC1 and TRPC4 channels may form sarcolemmal channels anchored to the DAPC, and alpha1-syntrophin is necessary to maintain the normal regulation of TRPC-supported cation entry in skeletal muscle. 19846750_TRPC1 ion channels modulate the entry of Ca++ during repeated contractions and help muscles to maintain their force during sustained repeated contractions. 20097773_TRPC1 may be expressed in the plasma membrane [letter] 20571036_TRPC1 participates in calcium influx and other key events required for mast cell degranulation. 20614160_Data show that gene silencing and pharmacological inhibition of TRPC1 significantly reduced connexin 43 up-regulation and Cx43/cytoskeletal interaction elicited by sphingosine 1-phosphate. 20718731_data demonstrate that TRPC1 acts as a negative regulator of endothelial KCa channel-dependent EDHF-type vasodilatations and thereby contributes to blood pressure regulation 20966350_Klotho protein is associated with VEGFR-2/TRPC-1 in causing cointernalization, thus regulating TRPC-1-mediated Ca(2+) entry to maintain endothelial integrity. 21524792_the enhanced expression of TRPC1 increases SOCE and has a negative effect on the STIM1-Orai1 system, indicating an interaction between these proteins. 21900691_It is suggested that different levels of the stretch-activated calcium channel protein TRPC1 may contribute to the different degrees of the dystrophic phenotype seen in mdx mice 21930300_An enhanced vesicular trafficking of heteromeric TRPV4-C1 channels contributes to store-operated Ca(2+) entry in vascular endothelial cells. 21968068_Agents that deplete intracellular Ca(2+) stores activate native heteromeric TRPC1/C5 channels in vascular smooth muscle cells. 22072513_The results suggested that TRPC1 may be important in the detection of innocuous mechanical force. 22083217_Data suggest that UCN regulates osteoclast resorption by suppressing osteoclast maturation/function via suppression of constitutively active cation channel (CRFR2beta) with properties of canonical transient receptor potential 1 (TRPC1) channel. 22144671_report that the large depolarizing plateau potential that underlies the epileptiform burst firing induced by metabotropic glutamate receptor agonists in lateral septal neurons was completely abolished in TRPC1/4 double-knockout mice 22157757_a novel regulatory mechanism relying on the expression of TRPC1 and the subsequent formation of heteromeric TRPC channel complexes with reduced calcium permeability, thereby fine-tuning neuronal migration. 22192412_Study concludes that mechanical injury of brain endothelial cells induces a rapid influx of calcium, mediated by TRPC1 and TRPP2 channels, which leads to NO synthesis and actin cytoskeletal rearrangement. 22210847_TRPC1 and TRPC4 can interact with STIM1 to form functional store-operated Ca(2+)-entry channels, which are essential for mediating Ca(2+) entry-dependent disruption of the endothelial barrier 22399301_the involvement of Trpc1 channels in skeletal muscle development in vitro and in vivo, and identify a Ca(2+)-dependent activation of the PI3K/Akt/mTOR/p70S6K pathway during myoblast differentiation and muscle regeneration. 22446186_TRPC1 is involved in regulating Ca2+ homeostasis and inhibiting the unfoled protein rsponse and thus contributes to neuronal survival after neurotoxin exposure. 22539836_This study concluded that TRPC1 can be activated downstream of NMDA receptor activation and contribute to slow synaptic transmission in the olfactory bulb, including the calcium dynamics required for asynchronous release from the granule cell spine. 22579301_The suppression of TRPC1 expression arrested the adult neural progenitor cell cycle at G0/G1 phase. 22612431_mGluR5 activation of cation current-carrying TRPC channels plays an important role in governing the cycle-by-cycle variability of the respiratory rhythm. 22674725_Mammalian rods express two new calcium signalling mechanisms associated with SOCE and TRPC1 signalling which modulate calcium homeostasis and may protect against prolonged [Ca2+]i decreases in saturating light 23034388_The data provided direct evidence that TRPC1 and Orai1 channels mediate capacitative calcium entry through activation of STIM1 in acute hypoxic mouse pulmonary arterial smooth muscle cells. 23095462_TRPC1 attenuates receptor-mediated aortic contraction through activation and/or expression of smooth muscle BK(Ca) channels and participates in endothelial cell Ca(2+) influx and vasorelaxation of aorta. 23188715_Is unaltered in TRPC5 KO mice, but is abolished in TRPC1 KO and TRPC1/4 DKO mice. 23489970_promotes recovery from the anaphylactic response by repressing antigen-mediated TNF-alpha release from mast-cells 23770672_propose that the positive and negative modulation of the CRAC channel by TRPC1epsilon and I-mfa, respectively, fine-tunes the dynamic range of the CRAC channel regulating osteoclastogenesis 23852583_Soleus muscle TRPC1 mRNA was unaltered during the hindlimb unloading-reloading period. However, protein expression remained depressed through the reloading process. Decreased localization of TRPC1 to the sarcolemma was observed. 23922735_Data suggest that TRPC1 and TRPC4 are involved in the sprouting of sensory neurons following bladder cystitis, which leads to overactive bladder disease. 24144647_TRPC1 participates in the pathophysiological development of hypoxic pulmonary hypertension in the mouse model. 24251695_Results indicate an important role of TRPC1 in pulmonary vascular remodeling underlying the development of hypoxia-induced pulmonary hypertension. 24613381_These results suggest that the AR region of TRPC3 could mediate the heteromeric TRPC3/1 formation, and the heteromeric TRPC3/1 could participate in regulating the resting cytosolic Ca(2+) levels in skeletal muscle. 25047165_Transient receptor potential channels contribute to pathological structural and functional remodeling after myocardial infarction. 25595528_TRPC1 controls the activity of further Ca(2) influx channels and thus regulates the maintenance of intracellular Ca(2) gradients which are critical for cell migration. 25899321_Together the data suggests that Ca(2+) entry via the TRPC1 channels is essential for the activation of CaCC 26031335_TRPC1(-/-) mice exhibited decreased survival, severe lung injury, and systemic bacterial dissemination upon infection. 26316271_transient receptor potential channel 1 suppression of basal sphingosine kinase 1 activity enables endothelial cell-barrier destabilization by edemagenic agonists 26752511_Down-regulation of TRPC1 in weight-bearing soleus muscles resulted in reduced muscle mass and reduced myofibre cross-sectional area. 26910815_TRPC1 is indispensable for the enriched environment-induced hippocampal neurogenesis and cognitive enhancement. 26985029_The results of this study showed that Loss of TRPC1 facilitated the gliotic response induced by increased intraocular pressure (IOP), suggesting that the channel might contribute to the glial mechanosusceptibility. 27034165_TRPC1 regulated directly or indirectly the expression of multiple proteins, which may be crucial for the maintenance of memory ability. 27535226_these data indicate for the first time a functional interaction between Orai1, TRPC1, and CaV1.2 channels in Vascular Smooth Muscle Cells, confirming that upon agonist stimulation, vessel contraction involves Ca(2+) entry due to co-activation of Orai1- and TRPC1-dependent store-operated Ca(2+) channels and L-type Ca(2+) channels. 27670661_mechanosensitive TRPC1 channels in murine PSCs exposed to elevated ambient pressure 27738307_Data show that transient receptor potential channel 1 (TRPC1) deficiency caused neuronal apoptosis in basal ganglia. 28258168_Ca(2+) entry serves critical cellular functions in virtually every cell type, and appropriate regulation of Ca(2+) in neurons is essential for proper function. 28636132_TRPC3-induced Ca2+ entry promotes astrocyte proliferation and migration i.e. astrocyte activity in vitro which is attenuated by the presence of TRPC1. Following brain injury, the absence of TRPC3 results in a significant reduction of astrogliosis and cortical edema in vivo, suggesting that a targeted therapy to reduce TRPC3 channel activity might be beneficial in traumatic brain injury. 28656279_Silencing of beta 1 integrin gene regulates the gene expression of storeoperated Ca2+ entry (SOCE)-associated genes (STIM1, ORAI1 and TRPC1). 28779972_we confirmed that the activation of OTX2, a determinant of DA neuron development and the expression of which is induced by thyroid hormone, is dependent on TRPC1-mediated calcium signaling. 28790178_These results indicate the contribution of heteromultimeric channels from TRPC1, TRPC4, and TRPC5 subunits to the regulation of mechanisms underlying spatial working memory and flexible relearning by facilitating proper synaptic transmission in hippocampal neurons. 28949388_the role of TRPC1 in the development of podocyte injury and disorders of the podocyte cytoskeleton, which may contribute to the development of novel therapeutics for podocyte injury-associated kidney diseases. 29074621_Data (including data from studies using knockout mice) suggest that TRPC1 inhibits positive effects of exercise on insulin resistance and type II diabetes in a high-fat diet-induced obesity environment. 29150520_these results suggest that dopaminergic neurotoxins initially decreased Ca(2+) entry, which inhibited the binding of NF-kappaB to the TRPC1 promoter, thereby inhibiting TRPC1 expression and resulting in cell death by preventing autophagy. 29480205_that inhibition of TRPC1 may serve as a neuroprotective strategy for HD treatment 29524421_By use of electrophysiology and intracellular Ca2+ imaging, this study characterises a Ca2+ permeable channel in white adipocytes. The current shows functional characteristics resembling the Ca2+ -permeable transient receptor potential channel 1 (TRPC1). 29660945_TRPC1 null exacerbates Abeta-induced learning and memory deficits with an increased cell apoptosis, although it does not affect cognitive functions or cell viability in physiological conditions. Overexpression of TRPC1 attenuates Abeta-induced cell death with the mechanism involving direct binding to AbetaPP through its TM2 domain and thus to reduce Abeta production. 29989850_We show that TRPC1 is highly expressed in all adipocyte depots including BAT and that Trpc1-deficient mice are prone to weight gain and manifest reduced metabolic control. knockdown of TRPC1 in cultured brown adipocytes leads to a downregulation of several metabolic genes, including UCP1 and PPARgamma, as well as upregulation of a BAT-specific thermosensitive channel TRPV2, resulting in impaired respiratory function. 30067380_findings identify TRPC1 as a modulator of HDM-induced airway remodeling via STAT3-mediated increase in mucus production, which provide new insight in our understanding of the molecular basis of airway remodeling 30504729_TRPC1 deficiency potentiates Reactive Oxygen Species generation via Nox4-containing NADPH oxidase, which exacerbates cerebral Ischemia/Reperfusion injury. 31182642_These results suggest an unequivocal role of TRPC1 in adipocyte differentiation and adiponectin secretion, and that loss of TRPC1 disturbs metabolic homeostasis. 31518158_Magnetism thus deploys an authentic myogenic directive that relies on an interplay between mitochondria and TRPC1 to reach fruition. 31603369_Stimulation of calcium-sensing receptors activates heteromeric TRPV4/TRPC1 channels and NO production in endothelial cells, which are responsible for endothelium-dependent vasorelaxations. 31722977_Loss of Ca(2+) entry via Orai-TRPC1 induces ER stress, initiating immune activation in macrophages. 32085504_TRPC1 Regulates the Activity of a Voltage-Dependent Nonselective Cation Current in Hippocampal CA1 Neurons. 32321939_Store-operated Ca(2+) entry in primary murine lung fibroblasts is independent of classical transient receptor potential (TRPC) channels and contributes to cell migration. 32875708_Cell-Derived Vesicles as TRPC1 Channel Delivery Systems for the Recovery of Cellular Respiratory and Proliferative Capacities. 35398674_TRPV4 and TRPC1 channels mediate the response to tensile strain in mouse Muller cells. |
ENSG00000144935 |
TRPC1 |
18.725604 |
0.3045943824 |
-1.715039 |
0.66002459 |
6.662596 |
0.00984573748485126527818511732448314432986080646514892578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0330476583008493807191996438632486388087272644042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.56874914142167 |
4.05833973952682 |
28.1316716191164 |
9.26063665310877 |
| ENSMUSG00000032860 |
18442 |
P2ry2 |
protein_coding |
A0A0R4J289 |
Human_homologues FUNCTION: Receptor for ATP and UTP coupled to G-proteins that activate a phosphatidylinositol-calcium second messenger system. The affinity range is UTP = ATP > ATP-gamma-S >> 2-methylthio-ATP = ADP. |
Cell membrane;G-protein coupled receptor;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:18442; |
plasma membrane [GO:0005886]; G protein-coupled purinergic nucleotide receptor activity [GO:0045028]; blood vessel diameter maintenance [GO:0097746]; phospholipase C-activating G protein-coupled receptor signaling pathway [GO:0007200]; positive regulation of mucus secretion [GO:0070257] |
12193392_study shows that P2Y2 receptors are expressed in cumulus cell-enclosed oocytes, and that their stimulation opens at least two different types of ion channels 12644577_Knock-out mice reveal tissue-specific roles of P2Y receptor subtypes in different epithelia. 12814360_Purinergic receptors on microglial cells: functional expression in acute brain slices and modulation of microglial activation in vitro. 15718265_Both the P2Y2 and P2Y4 receptors are present in the luminal membrane of mouse distal colonic mucosa, and stimulation of these receptors leads to potassium ion secretion. 16133936_functional interaction between endogenous P2Y2 receptor and TRPV1 channels could explain the ATP-induced pain 16365320_P2Y(2) has a role as a morphogen receptor that potentiates neurotrophin signaling in neuronal development and regeneration 16842754_identify Src family kinases as mediating nucleotide-enhanced neurotrophin-dependent neuronal differentiation and thus, as a key convergence point for interaction between TrkA receptor tyrosine kinases and P2Y(2) G-protein-coupled receptors 16980298_The role of P2Y2 and P2Y4 receptors in peritoneal macrophage calcium signaling is reported. 17291397_I(Cl.UTP) is due to the activation of CFTR Cl- channels through Gq/11-coupled P2Y2 receptor-PLC-PKC signaling and ATP hydrolysis in mouse heart 17382903_oncogenic potential of P2RY2 was confirmed in vitro with the focus formation assay as well as soft agar-growth assay, and also in vivo with a tumorigenicity assay in nude mice. 17728398_Novel protein kinase C epsilon (nPKCepsilon) is the effector isoform downstream of P2Y(2)-R activation in the tracheal goblet cell secretory response. 18091570_Involved in the influx of neutrophils into the lungs after sepsis. 18202316_This study demonstrates that P2Y(2)R/FLNa interaction selectively regulates spreading and migration of vascular SMCs. 18343036_P2Y2 is the principal uridine triphosphate (UTP) receptor in somatosensory neurons: P2Y2 is highly expressed in dorsal root ganglia and potentiates transient receptor potential cation channel V1 (TRPV1) activity in isolated sensory neurons. 18829742_genetic deletion of P2Y2-R results in increased abundances of key proteins involved in urinary concentration in the inner medulla. 18981175_P2Y2 receptor activation by nucleotides provides physiologically important regulation of ENaC and electrolyte handling in mammalian kidney 19074643_P2Y receptors mediate Ca2+ signaling in duodenocytes and contribute to duodenal mucosal bicarbonate secretion. 19423692_AQP2-mediated water transport is downregulated not only by basolateral nucleotides, mediated by P2Y(2) receptors, but also by luminal nucleotides, mediated by P2X(2) and/or P2Y(4) receptors. 20147562_These results support involvement of P2Y(2) receptors in bladder sensation, suggesting an important contribution to bladder neuron excitability and hypersensitivity. 20519655_Neutralizing intrapulmonary ATP levels or blocking airway P2Y2 receptors on lung neutrophils and macrophages inhibits smoke-induced lung inflammation and confers protection from the development of emphysema. 20664064_Data show that autocrine purinergic receptor P2Y(2), or P2Y(12) signaling amplifies and translates chemotactic cues into directional motility. 20720203_Eosinophil accumulation, a distinctive feature of lung allergic inflammation, is defective in ovalbumin (OVA)-treated P2Y2 receptor-deficient mice compared with OVA-treated wild type animals. 20813869_Control of ENaC by purinergic signaling (P2Y2) is necessary for aldosterone escape. 20880147_P2Y(2) R appears to be involved in asthmatic airway inflammation 21236346_Data show that tlt and mlh mutations uncouple Otopetrin 1 from inhibition of P2Y receptor function. 21464294_Heterologous down-regulation of angiotensin type 1 receptors by purinergic P2Y2 receptor stimulation through S-nitrosylation of NF-kappaB. 21520257_role in chondrocyte mechanotransduction 21613580_Systemic activation of P2Y(2) receptors lowers blood pressure and inhibits renal sodium reabsorption. 21653634_Activation of P2Y1 and P2Y2 receptors induces chloride secretion via calcium-activated chloride channels in kidney inner medullary collecting duct cells. 21839729_These results indicate that the P2Y(2) receptor mediates uridine-5'-triphosphate cardioprotection, in vivo. 21975874_genetic deletion of P2Y2-R offers significant resistance to the development of Li-induced polyuria, without altering blood Li levels and renal accumulation of Li, and suppression of PGE2 production 22057273_P2Y2 and P2Y12 receptor agonists are not chemoattractants and macrophage chemotaxis to complement C5a is phosphatidylinositol 3-kinase (PI3K)- and p38 mitogen-activated protein kinase (MAPK)-independent. 22199395_Real-time imaging reveals that P2Y2 and P2Y12 receptor agonists are not chemoattractants and macrophage chemotaxis to complement C5a is phosphatidylinositol 3-kinase (PI3K)- and p38 mitogen-activated protein kinase (MAPK)-independent. 22742194_Although C/EBPbeta was sufficient to induce P2Y(2) transcription, the effect of C/EBPbeta and NF-kappaB p65 on receptor transcription was synergistic. 22974709_Purinergic P2Y receptors promote neutrophil infiltration and hepatocyte death in mice with acute liver injury. 23046422_Stimulation of astrocytic P2Y1R receptors significantly reduces brain injury after acute trauma and is mediated by the inositol 1,4,5-trisphosphate (IP3) receptor-signaling pathway. 23550835_up-regulation of P2Y2Rs in primary cortical neurons under pro-inflammatory conditions can promote cofilin-dependent neurite outgrowth, a neuroprotective response that may be a novel pharmacological target in the treatment of neurodegenerative diseases. 23810565_Mice deficient in P2Y2 or lacking ATP secretion from platelets show strongly reduced tumor cell metastasis 23828651_Thus, P2Y2R activation protects cardiomyocytes from hypoxia in vitro and reduces post-ischemic myocardial damage in vivo. 24463702_P2Y2-mediated increase of cytoplasmic Ca(2+) concentration down-regulates the expression of NCC. 24631773_P2Y2 receptor fails to repress IL-6 expression by valve interstitial cells through Akt in calcific aortic valve disease. 24696143_Osteoblasts can regulate their mechanosensitivity through P2Y2R activation leading to increased cell stiffness. 24760984_UTP-induced migratory responses required for salivary gland acinar cell self-organization are mediated by the P2Y2R. 24816122_P2Y2R has a role in the skin wound-healing process in mouse models 25088742_P2Y2R deficiency does not alter baseline collateral vessel formation but does significantly impair collateral maturation, with resultant persistent limb ischemia despite enhanced angiogenesis. 25268784_P2Y2 receptors play important roles in bone marrow cell differentiation and mineralization as well as in bone cell mechanotransduction, leading to an osteopenic phenotype in P2Y2 knockout mice. 25545736_The data suggest that the P2Y2/4 receptor activation elicits blood pressure responses via distinct mechanisms involving KCa3.1 and Cx37. 25575683_Calcium-mediated purinergic receptors regulate the migration and phagocytic ability of microglia during post-natal brain development. 25692550_P2Y2-/- mice are less susceptible to mount an autoimmune response against IRBP 26036470_In P2Y1R (-/-) mice, the expression of P2Y2 receptor in muscle was reduced by over 50 %, as compared to P2Y1R (+/+) mice. 26062804_expression of P2Y(2) receptor in peripheral sensory neurons that innervate the injured tissue and the activation of P2Y receptors contributes to mechanical allodynia following inflammation 26168216_P2Y2 and Gq/G11 are required for basal endothelial NO formation, vascular tone, and blood pressure. 26538394_gene deficiency restricted to hematopoietic tissues results in longer survivak after GvHD 27220808_This study suggests that P2Y2 may participate in cardiomyopathy in mdx mice. 27339459_Extracellular ATP induces vascular inflammation and atherosclerosis via activation of P2Y2. 27393251_Results demonstrate that P2Y2 contributes to response properties of cutaneous afferents, as P2Y2 deletion reduces responsiveness of conventional unmyelinated polymodal afferents to heat and appears to result in the acquisition of mechanical responsiveness in a subset of TRPV1-expressing afferents. 27522265_both LRP1 and LDLR expression and agLDL uptake are regulated by P2Y2R in vascular smooth muscle cells, and agLDL uptake due to P2Y2R activation is dependent upon cytoskeletal reorganization mediated by P2Y2R binding to FLN-A 27565965_knockdown of P2Y2R retarded cyst expansion in vitro and prevented ATP- and HIF-1alpha-dependent cyst growth. In conclusion, P2Y2R mediates ATP-dependent cyst growth and is transcriptionally regulated by HIF-1alpha. 27768902_The P2Y2R enhances Ubiquitin-Proteasome System activity by increasing the subunits expression beta 5 and beta 1. 27856454_Endothelial cell-specific P2Y2R deficiency reduces atherosclerotic burden and promotes plaque stability in ApoE(-/-) mice through impaired macrophage infiltration acting together with reduced matrix metalloproteinase-2 activity and increased smooth muscle cell migration. 28038380_P2Y2R is an inhibitor of arterial intimal calcification, regulating the osteoblastic trans-differentiation of smooth muscle cells through P2Y2R-mediated Runx2 antagonism. 28415591_Our results clearly demonstrate the involvement of P2Y2R subtypes in the pathogenesis of fibrotic lung diseases in humans and mice and hence support the development of selective P2Y2R antagonists for the treatment of IPF. 28420708_These findings are consistent with the notion that the primary action of P2Y2 receptor signalling in bone is to regulate extracellular ATP levels. 29086245_We conclude that P2Y2 receptors on myeloid cells are important in mediating acute inflammation but are dispensable for the development of whole body insulin resistance in diet-induced obese mice 29297959_The results of this study suggest that P2Y2Rs contribute to the development of salivary gland inflammation in IL-14alphaTG mice and may also contribute to autoimmune sialadenitis in humans. 29401585_results suggest that LPS induces unbalanced proteostasis in astrocytes by blocking P2Y2R. Finally, our findings point to the design of selective P2Y2R agonist drugs as a new therapeutic approach to treat the neuroinflammatory status. 30236554_P2Y2 receptor expression was mechanistically upregulated by TNF-alpha mainly through extracellular regulated protein kinase (ERK) and c-Jun N-terminal kinase (JNK) signaling pathways. 30338362_interfering with somatic P2Y2 signalling prevents excessive immune cell deposition in diet-induced obesity (DIO), both attenuating adipose tissue inflammation and ameliorating the metabolic phenotype. 30684640_Data indicate that elevated purinergic responses to the increased expression of P2Y2 receptors as shown by reduced motility of mdx myoblasts. 31639617_Blockade of the extracellular ATP-P2Y2R signalling represents a potential therapeutic approach against alcoholic liver disease, and CD39 is a potential therapeutic target. 31822494_Extracellular ATP and Purinergic P2Y2 Receptor Signaling Promote Liver Tumorigenesis in Mice by Exacerbating DNA Damage. 31829837_P2Y2 Nucleotide Receptor Is a Regulator of the Formation of Cardiac Adipose Tissue and Its Fat-Associated Lymphoid Clusters. 32582029_P2Y2 Receptor Promotes High-Fat Diet-Induced Obesity. 32630144_Signaling Through Purinergic Receptor P2Y2 Enhances Macrophage IL-1beta Production. 32843542_Blastocyst-induced ATP release from luminal epithelial cells initiates decidualization through the P2Y2 receptor in mice. 32987187_P2Y2R contributes to the development of diabetic nephropathy by inhibiting autophagy response. 33575337_Purinergic Signaling Mediates PTH and Fluid Flow-Induced Osteoblast Proliferation. 33754385_Astrocyte-mediated purinergic signaling is upregulated in a mouse model of Fragile X syndrome. 33817825_P2Y2 deficiency impacts adult neurogenesis and related forebrain functions. 34073834_P2Y2R Deficiency Ameliorates Hepatic Steatosis by Reducing Lipogenesis and Enhancing Fatty Acid beta-Oxidation through AMPK and PGC-1alpha Induction in High-Fat Diet-Fed Mice. 34380439_P2Y2 promotes fibroblasts activation and skeletal muscle fibrosis through AKT, ERK, and PKC. 36520031_Deletion of the P2Y2 receptor aggravates internal elastic lamina calcification in chronic kidney disease mice through upregulation of alkaline phosphatase and lipocalin-2. |
ENSG00000175591 |
P2RY2 |
72.674894 |
2.6954739156 |
1.430539 |
0.32315637 |
19.295290 |
0.00001119822308504342778625369886347229453349427785724401473999023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000821238969818880571284805403031725745677249506115913391113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
101.133844592546 |
22.843378393394 |
37.519875078941 |
6.56122875762716 |
| ENSMUSG00000032875 |
207212 |
Arhgef17 |
protein_coding |
Q80U35 |
FUNCTION: Acts as guanine nucleotide exchange factor (GEF) for RhoA GTPases. {ECO:0000250}. |
Alternative splicing;Guanine-nucleotide releasing factor;Phosphoprotein;Reference proteome |
|
|
mmu:207212; |
guanyl-nucleotide exchange factor activity [GO:0005085]; actin cytoskeleton organization [GO:0030036] |
34808208_Gbetagamma mediates activation of Rho guanine nucleotide exchange factor ARHGEF17 that promotes metastatic lung cancer progression. |
ENSG00000110237 |
ARHGEF17 |
2593.598201 |
2.2084308213 |
1.143022 |
0.39250016 |
8.312711 |
0.00393685848256932098554461418871142086572945117950439453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0151195194114153216163876081168382370378822088241577148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
3785.30311386073 |
1042.58224687077 |
1714.0238568563 |
351.405235257765 |
| ENSMUSG00000032892 |
57785 |
Rangrf |
protein_coding |
Q9JIB0 |
FUNCTION: May regulate the intracellular trafficking of RAN (PubMed:10811801, PubMed:11733047). Promotes guanine nucleotide release from RAN and inhibits binding of new GTP by preventing the binding of the RAN guanine nucleotide exchange factor RCC1 (PubMed:10811801, PubMed:11733047). Regulates the levels of GTP-bound RAN in the nucleus, and thereby plays a role in the regulation of RAN-dependent mitotic spindle dynamics (By similarity). Enhances the expression of SCN5A at the cell membrane in cardiomyocytes (PubMed:18184654, PubMed:23420830). {ECO:0000250|UniProtKB:Q9HD47, ECO:0000269|PubMed:10811801, ECO:0000269|PubMed:11733047, ECO:0000269|PubMed:18184654, ECO:0000269|PubMed:23420830}. |
Cell membrane;Cytoplasm;Guanine-nucleotide releasing factor;Membrane;Nucleus;Protein transport;Reference proteome;Transport |
|
|
mmu:57785; |
caveola [GO:0005901]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; intercalated disc [GO:0014704]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; rough endoplasmic reticulum [GO:0005791]; guanyl-nucleotide exchange factor activity [GO:0005085]; small GTPase binding [GO:0031267]; sodium channel regulator activity [GO:0017080]; transmembrane transporter binding [GO:0044325]; endoplasmic reticulum to Golgi vesicle-mediated transport [GO:0006888]; heart contraction [GO:0060047]; nucleocytoplasmic transport [GO:0006913]; positive regulation of protein localization to cell surface [GO:2000010]; positive regulation of protein localization to plasma membrane [GO:1903078]; protein exit from endoplasmic reticulum [GO:0032527]; protein import into nucleus [GO:0006606]; regulation of bundle of His cell action potential [GO:0098905]; regulation of cardiac muscle cell action potential involved in regulation of contraction [GO:0098909]; regulation of membrane depolarization [GO:0003254]; regulation of membrane depolarization during cardiac muscle cell action potential [GO:1900825]; regulation of membrane potential [GO:0042391]; regulation of microtubule nucleation by Ran protein signal transduction [GO:0090226]; regulation of sodium ion transmembrane transport [GO:1902305]; regulation of sodium ion transmembrane transporter activity [GO:2000649] |
18184654_in cardiomyocytes, MOG1 is mostly localized in the cell membrane and co-localized with Nav1.5, indicating that MOG1 is a critical regulator of sodium channel function in the heart 35533905_Mog1 deficiency promotes cardiac contractile dysfunction and isoproterenol-induced arrhythmias associated with cardiac fibrosis and Cx43 remodeling. |
ENSG00000108961 |
RANGRF |
314.902131 |
2.2666246536 |
1.180546 |
0.15243424 |
59.615839 |
0.00000000000001153034207789789467895105345991100480640997698322802733628122950904071331024169921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000002855775130577897972153396922452613256558640042470642583793960511684417724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
450.850982791399 |
47.7253111436053 |
198.9085321559 |
16.2629618583265 |
| ENSMUSG00000032925 |
223272 |
Itgbl1 |
protein_coding |
Q8VDV0 |
|
Alternative splicing;EGF-like domain;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:223272; |
extracellular region [GO:0005576]; focal adhesion [GO:0005925]; membrane [GO:0016020]; integrin binding [GO:0005178]; cell adhesion mediated by integrin [GO:0033627]; cell migration [GO:0016477]; cell-matrix adhesion [GO:0007160]; integrin-mediated signaling pathway [GO:0007229] |
30305454_These results reveal ITGBL1 signaling as an underlying mechanism of protection against destructive cartilage disorders 33413419_ITGBL1 is a new immunomodulator that favors development of melanoma tumors by inhibiting natural killer cells cytotoxicity. |
ENSG00000198542 |
ITGBL1 |
1012.840516 |
0.1375345045 |
-2.862134 |
0.11549589 |
585.193579 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000278214779964256214958682238855437266598818728745112076695232038359358988938807519597718455817864759514864817355038933482876076958492615 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000022304039623239742852008024308973372945899511925199975214487594818607564954714145102709550207378074692904953109269124868508532201518100 |
Yes |
No |
242.171269785729 |
19.4078941671794 |
1760.80373904795 |
96.3711536802134 |
| ENSMUSG00000032946 |
19395 |
Rasgrp2 |
protein_coding |
Q9QUG9 |
FUNCTION: Functions as a calcium- and DAG-regulated nucleotide exchange factor specifically activating Rap through the exchange of bound GDP for GTP. May also activate other GTPases such as RRAS, RRAS2, NRAS, KRAS but not HRAS. Functions in aggregation of platelets and adhesion of T-lymphocytes and neutrophils probably through inside-out integrin activation. May function in the muscarinic acetylcholine receptor M1/CHRM1 signaling pathway. {ECO:0000269|PubMed:10777492, ECO:0000269|PubMed:10913189, ECO:0000269|PubMed:11278453, ECO:0000269|PubMed:11432821, ECO:0000269|PubMed:12239348, ECO:0000269|PubMed:15334074, ECO:0000269|PubMed:16357324, ECO:0000269|PubMed:17492052, ECO:0000269|PubMed:9789079}. |
Alternative splicing;Calcium;Cell membrane;Cell projection;Cytoplasm;Guanine-nucleotide releasing factor;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Synapse;Synaptosome;Zinc;Zinc-finger |
|
|
mmu:19395; |
cytosol [GO:0005829]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; ruffle membrane [GO:0032587]; synapse [GO:0045202]; calcium ion binding [GO:0005509]; guanyl-nucleotide exchange factor activity [GO:0005085]; cellular response to calcium ion [GO:0071277]; positive regulation of GTPase activity [GO:0043547]; Ras protein signal transduction [GO:0007265] |
12239348_CalDAG-GEFI plays a role in inside-out signaling to alphaIIbbeta3 17492052_CalDAG-GEFI regulated the activation of beta(1) and beta(3) integrins in platelets, and CalDAG-GEFI deficiency caused complete inhibition of arterial thrombus formation. 18544684_CalDAG-GEFI and PKC represent separate, but synergizing, pathways important for alphaIIbbeta3 activation in platelets activated through the PAR4 receptor. 20147317_In a brain-slice explant model of Huntington's disease, knock-down of CalDAG-GEFI expression rescues striatal neurons from pathology induced by transfection of polyglutamine-expanded Htt exon 1. 20413728_CalDAG-GEFI helps regulate neutrophil chemotaxis, independent of its established role in integrin activation, through a mechanism that involves actin cytoskeleton and cellular polarization. 20971951_Studies indicate that CalDAG-GEFI mediates the rapid but reversible activation of integrin alphaIIbbeta3, and the adenosine diphosphate receptor P2Y12 facilitates delayed but sustained integrin activation. 21652673_we evaluated the contribution of CalDAG-GEFI to platelet activation in a model of immune-mediated thrombocytopenia and thrombosis syndromes 21848641_(i) STIM1/SOCE is critical for procoagulant activity but not the proadhesive function of platelets; and (ii) at the site of vascular injury, STIM1 and CalDAG-GEFI are critical for the first wave of thrombin generation mediated by procoagulant platelets 26988592_CalDAG-GEFI is critical for atherosclerotic plaque development in hypercholesterolemic Ldlr(-/-) mice because of its contribution to platelet-leukocyte aggregate formation and leukocyte recruitment to the lesion area. 34371144_CalDAG-GEFI mediates striatal cholinergic modulation of dendritic excitability, synaptic plasticity and psychomotor behaviors. |
ENSG00000068831 |
RASGRP2 |
231.332132 |
0.3598811442 |
-1.474408 |
0.23984560 |
36.927049 |
0.00000000122632780669453478869914279437459969646084800842800177633762359619140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000171370872950194094406999560523466308303852656536037102341651916503906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
122.320141105739 |
20.4293194791308 |
339.890386219025 |
41.5537213279144 |
| ENSMUSG00000033022 |
12583 |
Cdo1 |
protein_coding |
P60334 |
FUNCTION: Catalyzes the oxidation of cysteine to cysteine sulfinic acid with addition of molecular dioxygen. {ECO:0000269|PubMed:16492780}. |
3D-structure;Dioxygenase;Iron;Metal-binding;Oxidoreductase;Reference proteome;Thioether bond |
PATHWAY: Organosulfur biosynthesis; taurine biosynthesis; hypotaurine from L-cysteine: step 1/2. |
|
mmu:12583; |
cytosol [GO:0005829]; plasma membrane [GO:0005886]; cysteine dioxygenase activity [GO:0017172]; ferrous iron binding [GO:0008198]; nickel cation binding [GO:0016151]; oxidoreductase activity, acting on single donors with incorporation of molecular oxygen, incorporation of two atoms of oxygen [GO:0016702]; zinc ion binding [GO:0008270]; L-cysteine catabolic process [GO:0019448]; L-cysteine catabolic process to taurine [GO:0019452]; lactation [GO:0007595]; response to amino acid [GO:0043200]; response to cAMP [GO:0051591]; response to ethanol [GO:0045471]; response to glucagon [GO:0033762]; response to glucocorticoid [GO:0051384]; response to organonitrogen compound [GO:0010243]; taurine biosynthetic process [GO:0042412]; taurine metabolic process [GO:0019530] |
11602353_The CDO gene displays tissue-specific expression, with the highest mRNA level present in liver and with detectable levels found in kidney, lung, brain and small intestine. 16492780_The x-ray crystal structure of CDO from Mus musculus was solved to a nominal resolution of 1.75 Angstroms 17602574_Characterization of the nitrosyl adduct of substrate-bound mouse cysteine dioxygenase by electron paramagnetic resonance 20397631_resting and substrate-bound forms of CDO in the Fe(II) and Fe(III) states, both of which are proposed to have important roles in this enzyme's catalytic mechanism, were characterized. 21693692_Control of cysteine levels by regulation of CDO may be necessary to maintain low H(2)S/sulfane sulfur levels and facilitate the use of H(2)S as a signaling molecule. 21992268_The catalytic cycle of CDO is primed by one electron through chemical oxidation to produce CDO with ferric iron in the active site. 22414809_The hepatic CDO-knockout mice were able to maintain normal levels of glutathione, taurine, and sulfate. 23350603_A critical function of CDO appears to be to remove cysteine by a pathway in which the sulfur atom is oxidized in the first step. 23569214_Cdo(-/-) mice displayed megaesophagus and achalasia, and their lower esophageal sphincter was resistant to nitric oxide-induced relaxation. 24279989_Data from kinetic, spectroscopic, and computational studies suggest that in cysteine dioxygenase (CDO) a covalently cross-linked cysteine-tyrosine pair (C93-Y157) plays a vital role in CDO-mediated catalysis. 24609271_Cdo1 knockout mice show increased cysteine concentrations and higher rates of metabolism of cysteine to hydrogen sulfide and thiosulfate. 25387045_the timing of chemical steps in the CDO kinetic mechanism is investigated by pH/pD-dependent steady-state kinetics and solvent isotope effects on kcat, kcat/KM, and (O2/CSA) coupling 25444857_In light of these results, the minimal substrate requirements for CDO catalysis and O-activation are discussed. 25637537_Cdo1 is required for adipogenesis.Cdo1 interacts with Ppar-gamma during adipogenesis. 26119969_We investigated the ontogeny of Cdo1 mRNA expression in mouse fetal and placental tissues, which showed increasing levels from embryonic day 10.5 and was localised to the decidua and several fetal tissues including nasal cavities and brain. 26763277_our findings indicate Cdo1 suppresses osteogenic differentiation of BMSCs, through a potential mechanism which involves in Wnt signaling reduction concomitantly 26906632_Cdo is required for efficient cardiomyogenesis of pluripotent stem cells and an excellent target to improve the differentiation potential of stem cells for generation of transplantable cells to treat cardiomyopathies. 28826737_Presented here are the results of O2-dependent 2-mercaptoaniline reaction using two different thiol dioxygenase enzymes mouse CDO and 3-mercaptopropionic acid dioxygenase isolated from Azotobacter vinelandii. Benzothiazoles are produced by the condensation of 2-mercaptoaniline with aldehydes formed by an off-pathway oxidation of primary alcohols added to aqueous reactions to solubilize the substrate. 29604178_findings suggest that epididymal CDO plays a key role in post-testicular sperm maturation, enabling sperm to osmoregulate as they transition from the male to the female reproductive tract 36253617_Cdo1 promotes PPARgamma-mediated adipose tissue lipolysis in male mice. |
ENSG00000129596 |
CDO1 |
548.855342 |
0.1319369403 |
-2.922080 |
0.15080580 |
362.973371 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000634086189957545802500927216482161300744342614439377891905458260421326465573583413926963668855878921623335081752947223797385386789727247662412391106514422366801178583566477782927993346 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000022996192489126997833142876321160903137370933669161510258134881316661071934685381011815885738047621318512276380053500107080192832230762426838667459374553151581422027552405242710675900 |
Yes |
No |
126.211803392787 |
13.2170362186484 |
956.607020873778 |
66.9325821437034 |
| ENSMUSG00000033031 |
224171 |
Cip2a |
protein_coding |
Q8BWY9 |
FUNCTION: Promotes anchorage-independent cell growth and tumor formation. {ECO:0000250|UniProtKB:Q8TCG1}. |
Acetylation;Coiled coil;Cytoplasm;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:224171; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; plasma membrane [GO:0005886]; protein homodimerization activity [GO:0042803]; neural precursor cell proliferation [GO:0061351]; neuroblast proliferation [GO:0007405]; positive regulation of neuroblast proliferation [GO:0002052]; spermatogenesis [GO:0007283] |
20447748_Loss of CIP2A in Myc-overexpressing neural progenitor cells significantly reduces the ability of Myc to increase self-renewal and proliferation. 22461891_Data show that CIP2A expression can be systematically inhibited without severe consequences to normal mouse development and viability may have clinical relevance regarding targeting of oncogenic CIP2A for future cancer therapies. 24214971_CIP2A strongly interacts with NEK2 during G2/M phase, thereby enhancing NEK2 kinase activity to facilitate centrosome separation in a PP1- and PP2A-independent manner. 24884612_Downregulation of CIP2A suppresses cell proliferation and growth of nasopharyngeal carcinoma. 26813457_Knockdown of CIP2A by stable CIP2A siRNA transfection inhibited MDA-MB-231 cell proliferation, invasion, colony growth in vitro, and xenograft growth and metastasis in vivo of breast cancer in mice. 27100879_CIP2A as a hitherto unrecognized mediator of T-cell activation. 28935709_CIP2A acts as a scaffold for CEP192-mediated microtubule organizing center assembly by recruiting Plk1 and aurora A during meiotic maturation in mouse oocytes 30021167_CIP2A-mediated PP2A inhibition drives tau/APP hyperphosphorylation and increases APP beta-cleavage and Abeta production. 30594047_that CIP2A-promoted astrogliosis induces synaptic degeneration and cognitive deficits in Alzheimer's disease 33070372_Polyphyllin I attenuates cognitive impairments and reduces AD-like pathology through CIP2A-PP2A signaling pathway in 3XTg-AD mice. 35195302_ChK1 activation induces reactive astrogliosis through CIP2A/PP2A/STAT3 pathway in Alzheimer's disease. 35286657_Chk1 Inhibition Ameliorates Alzheimer's Disease Pathogenesis and Cognitive Dysfunction Through CIP2A/PP2A Signaling. 36305233_CIP2A deficiency promotes depression-like behaviors in mice through inhibition of dendritic arborization. 36690227_Oct4 reduction contributes to testicular injury of unilateral testicular torsion in mice model and apoptotic death of Sertoli cells through mediating CIP2A expression. |
ENSG00000163507 |
CIP2A |
1060.584483 |
2.4333790264 |
1.282961 |
0.14312835 |
78.091929 |
0.00000000000000000098354978612915036075213357143473004140289868587715112549507701089623878942802548408508300781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000354170930078468560737998626091811063954403309686144729617751636396860703825950622558593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
1494.72807485317 |
148.533872854308 |
614.260277021657 |
46.2138958479316 |
| ENSMUSG00000033102 |
218294 |
Cdc14b |
protein_coding |
Q6PFY9 |
FUNCTION: Dual-specificity phosphatase involved in DNA damage response. Essential regulator of the G2 DNA damage checkpoint: following DNA damage, translocates to the nucleus and dephosphorylates FZR1/CDH1, a key activator of the anaphase promoting complex/cyclosome (APC/C). Dephosphorylates SIRT2 around early anaphase. Dephosphorylation of FZR1/CDH1 activates the APC/C, leading to the ubiquitination of PLK1, preventing entry into mitosis. Preferentially dephosphorylates proteins modified by proline-directed kinases (By similarity). {ECO:0000250}. |
Alternative splicing;DNA damage;DNA repair;Hydrolase;Nucleus;Protein phosphatase;Reference proteome |
Human_homologues NA; + ;NA |
|
mmu:218294; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; mitotic spindle [GO:0072686]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; spindle pole [GO:0000922]; myosin phosphatase activity [GO:0017018]; protein serine/threonine phosphatase activity [GO:0004722]; protein tyrosine phosphatase activity [GO:0004725]; protein tyrosine/serine/threonine phosphatase activity [GO:0008138]; cilium assembly [GO:0060271]; DNA repair [GO:0006281]; microtubule cytoskeleton organization [GO:0000226]; mitotic G2 DNA damage checkpoint signaling [GO:0007095]; positive regulation of cytokinesis [GO:0032467]; positive regulation of ubiquitin protein ligase activity [GO:1904668]; protein dephosphorylation [GO:0006470]; regulation of exit from mitosis [GO:0007096] |
19129509_CDC14B is a negative regulator of meiotic resumption and may regulate meiosis I in mouse oocytes. 19923902_CDC14B is a negative regulator of the 1-to-2-cell transition and of zygotic genome activation in mouse embryogenesis. 21262768_Cdc14b is required for efficient DNA damage repair. 21502810_Cdc14B has oncogenic activity in mammals and point to the Ras-MAP kinase pathway as a major effector pathway during oncogenic transformation. 22355704_Cdc14b regulates mammalian RNA polymerase II and represses cell cycle transcription 26283732_Cdc14B knockout (Cdc14B(-/-)) mouse embryonic fibroblasts showed defects in repairing ionizing radiation-induced DNA double-strand breaks, which occurred only at late passages when Cdc14A levels were low. 32341029_Mis12 controls cyclin B1 stabilization via Cdc14B-mediated APC/C(Cdh1) regulation during meiotic G2/M transition in mouse oocytes. |
ENSG00000081377+ENSG00000218305 |
CDC14B+CDC14C |
258.246669 |
0.2994485358 |
-1.739620 |
0.25893877 |
42.464185 |
0.00000000007198841337452664684482815616464539652807896885633454076014459133148193359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000011640419453511569098231384565794548080486237040531705133616924285888671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
120.018240358721 |
21.6570363829141 |
400.797552814738 |
52.7942828683728 |
| ENSMUSG00000033152 |
319655 |
Podxl2 |
protein_coding |
D3YW47 |
Human_homologues FUNCTION: Acts as a ligand for vascular selectins. Mediates rapid rolling of leukocytes over vascular surfaces through high affinity divalent cation-dependent interactions with E-, P- and L-selectins. {ECO:0000269|PubMed:18606703}. |
Glycoprotein;Membrane;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
|
membrane [GO:0016020]; leukocyte tethering or rolling [GO:0050901] |
Human_homologues 12889478_endoglycan functions as an L-selectin ligand through modification with tyrosine sulfation and sialyl Lewis x 18606703_findings indicate that endoglycan can function as a ligand for the vascular selectins. 32669966_PODXL2 maintains cellular stemness and promotes breast cancer development through the Rac1/Akt pathway. 34208313_ITGB6-Knockout Suppresses Cholangiocarcinoma Cell Migration and Invasion with Declining PODXL2 Expression. 34390512_Endoglycan (PODXL2) is proteolytically processed by ADAM10 (a disintegrin and metalloprotease 10) and controls neurite branching in primary neurons. |
ENSG00000114631 |
PODXL2 |
12.328435 |
0.1626310610 |
-2.620325 |
1.01235988 |
6.507931 |
0.01073943978115890311719748950736175174824893474578857421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0355770219109639920795906675721198553219437599182128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.20718596262649 |
2.43982397171839 |
19.7206237458323 |
9.10777413608176 |
| ENSMUSG00000033208 |
20203 |
S100b |
protein_coding |
P50114 |
FUNCTION: Weakly binds calcium but binds zinc very tightly-distinct binding sites with different affinities exist for both ions on each monomer. Physiological concentrations of potassium ion antagonize the binding of both divalent cations, especially affecting high-affinity calcium-binding sites. Binds to and initiates the activation of STK38 by releasing autoinhibitory intramolecular interactions within the kinase. Interaction with AGER after myocardial infarction may play a role in myocyte apoptosis by activating ERK1/2 and p53/TP53 signaling. Could assist ATAD3A cytoplasmic processing, preventing aggregation and favoring mitochondrial localization. May mediate calcium-dependent regulation on many physiological processes by interacting with other proteins, such as TPR-containing proteins, and modulating their activity (By similarity). {ECO:0000250}. |
Acetylation;Calcium;Cytoplasm;Metal-binding;Nucleus;Reference proteome;Repeat;Zinc |
|
|
mmu:20203; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; intracellular membrane-bounded organelle [GO:0043231]; neuronal cell body [GO:0043025]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; ruffle [GO:0001726]; calcium ion binding [GO:0005509]; calcium-dependent protein binding [GO:0048306]; identical protein binding [GO:0042802]; ion binding [GO:0043167]; protein homodimerization activity [GO:0042803]; RAGE receptor binding [GO:0050786]; S100 protein binding [GO:0044548]; signaling receptor binding [GO:0005102]; tau protein binding [GO:0048156]; zinc ion binding [GO:0008270]; cellular calcium ion homeostasis [GO:0006874]; energy reserve metabolic process [GO:0006112]; learning or memory [GO:0007611]; memory [GO:0007613]; negative regulation of skeletal muscle cell differentiation [GO:2001015]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of myelination [GO:0031643]; positive regulation of synaptic transmission [GO:0050806]; regulation of cell shape [GO:0008360]; regulation of cytokine production [GO:0001817]; regulation of long-term neuronal synaptic plasticity [GO:0048169]; regulation of neuronal synaptic plasticity [GO:0048168] |
11835317_constitutive overexpression of S100beta in transgenic mice does not modify serotonin levels during development, nor does it protect the serotonergic neurons from selective neurotoxicity or modify the serotonergic sprouting induced by partial lesion. 11872254_The distribution of serotonergic fibers in the brains of S100B-knockout mice was normal. S100B may not influence neurite extension of serotonergic neurons 11891290_Glial protein S100B modulates long-term neuronal synaptic plasticity 12377780_the major cytoplasmic S100B target protein in different glial cell lines in the presence of Zn(2+) and Ca(2+) is IQGAP1 12393261_These results implicate normal levels of S100B in the attenuation of epileptogenesis. 12561079_the -1,669/+3,106 sequence of the S100B gene is a useful reagent for driving expression of transgenes in most S100B-expressing cells of mouse brain. 12673835_S-100b-containing cells and GFAP-containing cells increased in the striatum and substantia nigra at 3 days after MPTP treatment. S-100 immunoreactivity was observed only in GFAP-positive astrocytes. 12742260_S-100 beta overexpressing animals were more active than the CD-1 control animals and showed significantly less social sniffing. 15126113_Our findings show increased clusterin expression in the aged S100B mice compared to their CD-1 controls, a finding we have interpreted as further evidence of pathological brain aging. 15236402_results support the idea that elevated levels of S100B in the brain are associated with increased vulnerability to neurological injury 15555923_Mice lacking S100B show an apparent delay in OPC maturation in response to demyelinating insult. Nuclear S100B participates in the regulation of oligodendroglial cell maturation. 15621007_5-HT may modulate glial morphology by inducing a release of intracellular S-100B, and this pathway is inoperable in the S-100B knockout (-/-). 15782413_These results argue against the current view that S100B expression is restricted to the astrocytic lineage in the CNS, and indicate that the use of S100B in combination with other molecular markers will help discriminate oligodendrocytes from astrocytes. 15810011_Our data are consistent with a model in which S-100B overexpression in AD enhances glial activation and leads to an augmented neuroinflammatory process that increases the severity of neuropathologic sequelae. 17078026_Altogether, our results indicate that the S100B expression defines a late developmental stage after which GFAP-expressing cells lose their NSC potential and suggest that S100B expression is repressed by adult SVZ microenvironment. 17729158_possible interaction between FGF-2 and S100Beta in activated satellite cells of the dorsal root ganglia, which might trigger paracrine actions in the axotomized sensory neurons 18451356_S100B has a role in exacerbating brain damage and periinfarct reactive gliosis (astrocytosis and microgliosis) during the subacute phase of middle cerebral artery occlusion 18599158_Given the crucial roles of COX-2, IL-1beta and TNF-alpha in the inflammatory response, we propose that, by engaging RAGE, S100B might play an important role in microglia activation in the course of brain damage. 18638525_There was a significant increase of hippocampal BDNF (+53%) and a decrease of hippocampal (-12%) and residual neocortical (-15%) NA in 10-month-old S100B KO mice compared to wildtype mice. 18649404_Our findings suggest further investigation into the potential role of vitamin E in reducing the oxidation state of the S100B protein and its influence on neuroinflammatory processes marked by microglial activation in vivo. 18812013_The data of this study indicated that genetic differences in S100B gene expression may predispose individual differences in the responsivity to repeated intake of MDMA. 19713945_attenuates the hemodynamic response to catecholamines 20069545_Intracellular S100B might modulate myoblast differentiation by interfering with MyoD expression in an NF-kappaB-dependent manner. 20099023_These results suggest that S100beta is important for brain development and establishment of proper brain functions. 21080947_Data support S100B inhibition as a novel strategy for reducing cortical plaque load, gliosis and neuronal dysfunction in Alzheimer disease (AD) and suggest that both extracellular as well as intracellular S100B contribute to AD histopathology. 21146588_Unlike in the hippocampus-dependent tasks, S100B-KO mice were indistinguishable from wild-type mice in both cerebellum-dependent motor coordination and delay eyeblink conditioning. 21264954_The RAGE pathway may play an important role in STAT3 induction in glioma-associated macrophages and microglia, a process that may be mediated through S100B. 21779383_S100B and APP levels are simultaneously increased within Down syndrome neural progenitors, their secretions are synergistically enhanced in a paracrine fashion, and their overexpressions disrupt mitochondrial membrane potentials and redox states. 22508049_S100B is expressed in mouse photoreceptor cone outer segments but not in rods. 23000886_S100B expression may serve to modulate cardiac metabolism and adverse consequences of AGE in diabetic post-MI remodeling and function 23719262_Gioma production of S100B enhancestumor growth through CCL2 upregulation and tumor-associated macrophages chemoattraction. 23729438_S100B is a proinflammatory cytokine bridging RAGE and CD166/ALCAM downstream effector mechanisms, both being compensatory upregulated after genetic deletion of its counterpart. 23804363_Data suggest up-regulation of S100b/RAGE (advanced glycosylation end-product receptor) signaling plays role in inflammatory interaction between adipocytes/macrophages; adipocyte secretion of S100b is up-regulated by Tnf (tumor necrosis factor-alpha). 23899512_HMGB1, S100B, and RAGE signaling modulate the hippocampal inflammatory response and might play key roles in surgery-induced cognitive decline. 25152033_S100A1 and S100B are dispensable for endochondral ossification during skeletal development. 25482070_high glucoseinduced profibrotic genes (TGFbeta, type IV collagen and fibronectin) and cell hypertrophyrelated p21WAF1 are dependent on S100B. 25817898_Data show that high glucose increased protein-protein interaction between Steap4 and S100B in mesangial (MES13) cells. 26204512_Data show that S100B has direct effects on macrophages, enhancing particularly CCL22 and IL-1beta expression and modulates the inflammatory response in uveoretinitis and this is likely to be, at least in part, via a direct effect on macrophages. 26499172_The results of this study showed that S100B affects behavioral despair in female mice through functional interaction with the 5-HT7 receptor. 27601207_S100B inhibits C3H/10T1/2 murine embryonic mesenchyma.l cells into osteoblasts. S100B stimulates C3H/10T1/2 cell differentiation into adipocytes. 28174179_Data (including data from studies in knockout mice) suggest that S100b acting as a humoral factor impairs glycolysis in muscle (myoblasts, myotubes, and skeletal muscles) independent of insulin action; this effect appears to be due to inhibition of Gapdh activity from enhanced poly(ADP-ribosyl)ation of Gapdh. (S100B = S100 protein, beta polypeptide, neural; Gapdh = glyceraldehyde-3-phosphate dehydrogenase) 28970581_levels of S100B differentially affect skeletal muscle repair upon acute injury and in the context of muscular dystrophy, and S100B might be regarded as a potential molecular target in Duchenne muscular dystrophy . 29499206_S100B may be a promising target for pharmacological interventions aimed at repressing diabetes. 30076898_Findings indicate the role of S100B in gliomagenesis and suggest the development of more potent S100B inhibitors for glioma therapy. 30229393_Our findings demonstrate that S100B promotes microglia M1 polarization to aggravate cerebral ischemia, and provide a better understanding on the therapeutic effects of S100B and/or its antagonist/neutralization antibody in stroke. 31043739_A deficiency of S100b phenocopies deficiency of calsyntenin 3beta, and forced expression of S100b in brown adipocytes rescues the defective sympathetic innervation that is caused by ablation of calsyntenin 3beta; data reveal a mammal-specific mechanism of communication between thermogenic adipocytes and sympathetic neurons 32240725_Implications of increased S100beta and Tau5 proteins in dystrophic nerves of two mdx mouse models for Duchenne muscular dystrophy. 33460952_Characterization of cell type-specific S100B expression in the mouse olfactory bulb. 34615523_S100B is selectively expressed by gray matter protoplasmic astrocytes and myelinating oligodendrocytes in the developing CNS. 34741005_S100B dysregulation during brain development affects synaptic SHANK protein networks via alteration of zinc homeostasis. |
ENSG00000160307 |
S100B |
793.011496 |
0.1773757536 |
-2.495119 |
0.12322144 |
394.480086 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000087613521112319145270022155091593803077262237698066565756007390192294531938105918140196175719091079763294787047477729595301369124516503313809288639641899164990512121308369675819 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000004170403604946391252345931621610451936552019716479634809959623931281417935051647151962178188078990320731768167981782057296646885376843388787761283497052593551342939053416766149 |
Yes |
No |
236.260820374328 |
20.2037205201862 |
1331.97923411927 |
78.8816265023569 |
| ENSMUSG00000033453 |
235130 |
Adamts15 |
protein_coding |
P59384 |
FUNCTION: Metalloprotease which has proteolytic activity against the proteoglycan VCAN, cleaving it at the 'Glu-1401-|-1402-Ala' site (PubMed:24220035). Cleaves VCAN in the pericellular matrix surrounding myoblasts, facilitating myoblast contact and fusion which is required for skeletal muscle development and regeneration (PubMed:23233679). {ECO:0000269|PubMed:23233679, ECO:0000269|PubMed:24220035}. |
Cleavage on pair of basic residues;Disulfide bond;Extracellular matrix;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Repeat;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes a member of 'a disintegrin and metalloproteinase with thrombospondin motifs' (ADAMTS) family of multi-domain matrix-associated metalloendopeptidases that have diverse roles in tissue morphogenesis and pathophysiological remodeling, in inflammation and in vascular biology. The encoded preproprotein undergoes proteolytic processing to generate an active versicanase enzyme. This gene is located adjacent to a related ADAMTS gene (Adamts8) on chromosome 9. [provided by RefSeq, Jul 2016]. |
mmu:235130; |
cell surface [GO:0009986]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; endopeptidase activity [GO:0004175]; extracellular matrix binding [GO:0050840]; heparin binding [GO:0008201]; metalloendopeptidase activity [GO:0004222]; zinc ion binding [GO:0008270]; extracellular matrix disassembly [GO:0022617]; extracellular matrix organization [GO:0030198]; myoblast fusion [GO:0007520]; proteolysis [GO:0006508] |
24220035_these data suggest a role for ADAMTS15 in a wide range of biological processes that are potentially mediated through the processing of versican. 25349050_Results show that ADAMTS15 and ADAMTS4 not only exhibit unique cellular expression patterns in the brain but their developmental upregulation by these cell types coincides with critical aspects of neural development 25510509_spiking parvalbumin interneurons enwrapped in the perineuronal net express the metallopeptidases Adamts15 in mice. 35962790_Biallelic variants in ADAMTS15 cause a novel form of distal arthrogryposis. |
ENSG00000166106 |
ADAMTS15 |
11.146213 |
0.0442905341 |
-4.496858 |
1.15921656 |
17.468371 |
0.00002921275216265250925158115935520442008055397309362888336181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001972378727577672968915983853221973731706384569406509399414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.820016074913332 |
0.732112616468801 |
18.5144769878119 |
8.2258625549802 |
| ENSMUSG00000033585 |
17984 |
Ndn |
protein_coding |
P25233 |
FUNCTION: Growth suppressor that facilitates the entry of the cell into cell cycle arrest. Functionally similar to the retinoblastoma protein it binds to and represses the activity of cell-cycle-promoting proteins such as SV40 large T antigen, adenovirus E1A, and the transcription factor E2F. Necdin also interacts with p53 and works in an additive manner to inhibit cell growth. Also functions as a transcription factor and directly binds to specific guanosine-rich DNA sequences. |
Cytoplasm;DNA-binding;Growth regulation;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:17984; |
cell projection [GO:0042995]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; nuclear matrix [GO:0016363]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; gamma-tubulin binding [GO:0043015]; promoter-specific chromatin binding [GO:1990841]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; axon extension [GO:0048675]; axonal fasciculation [GO:0007413]; axonogenesis [GO:0007409]; central nervous system development [GO:0007417]; genomic imprinting [GO:0071514]; glial cell migration [GO:0008347]; multicellular organismal homeostasis [GO:0048871]; negative regulation of DNA-binding transcription factor activity [GO:0043433]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuron development [GO:0048666]; neuron differentiation [GO:0030182]; neuron migration [GO:0001764]; neurotrophin TRK receptor signaling pathway [GO:0048011]; positive regulation of protein deacetylation [GO:0090312]; positive regulation of transcription by RNA polymerase II [GO:0045944]; post-embryonic development [GO:0009791]; regulation of DNA-templated transcription [GO:0006355]; respiratory gaseous exchange by respiratory system [GO:0007585]; respiratory system process [GO:0003016]; sensory perception of pain [GO:0019233] |
9302265_The necdin gene is imprinted, with preferential expression from the paternal allele in human and mouse. 12083804_necdin mediates the terminal differentiation and survival of NGF-dependent dorsal root ganglion neuron and that necdin-deficient nascent neurons are destined to caspase-3-dependent apoptosis. 12198120_Necdin promotes differentiation and survival of neurons through its antagonistic interactions with E2F1. 12414813_Necdin and MAGE-H1 are interactors for the intracellular domain of p75 12629158_Absence of Ndn causes deficiency of central respiratory drive in newborn mice; most die in the neonatal period of apparent respiratory insufficiency 14643685_The gene expression pattern of Necdin during mouse nervous system development. 15155529_Necdin acts as a master gene regulating smooth muscle (SM) differentiation in at least a subset of SMCs. 15272023_necdin and MAGE-D1 cooperate to modulate the function of Dlx/Msx homeodomain proteins in cellular differentiation 15470499_Data show that NSCL-1 and -2, together with additional cofactors, directly control transcription of the necdin gene. 15634360_Snurf-Snrpn and C/D-box small nucleolar RNAs MBII-85 and MBII-52 and Ndn and Magel2 genes have highly similar expression patterns in the central nervous system, suggesting that they share a common central nervous system-specific regulatory element 15649943_loss of necdin impinges on axonal outgrowth, contributes to the neurological phenotype of Prader-Willi syndrome 15895078_Insulin receptor substrate proteins regulate a necdin-E2F4 interaction that represses peroxisome-proliferator-activated receptor gamma (PPARgamma) transcription via a cyclic AMP response element binding protein (CREB)-dependent pathway. 16049186_paternally expressed necdin facilitates TrkA signaling to promote the survival of NGF-dependent nociceptive neurons 16707790_These results suggest that paternally expressed necdin facilitates the differentiation and specification of GABAergic neurons in cooperation with Dlx homeodomain proteins. 16720720_Results demonstrate that thyroid hormone regulates the transcription of the Necdin gene. 17108174_These results suggest that endogenous necdin attenuates neuronal apoptosis by suppressing the E2F1-Cdc2 system. 17116257_in early development of the nervous system, Necdin is an anti-apoptotic or survival factor 17954612_These data clarify the function and mechanism of necdin in skeletal muscle and show the importance of necdin in muscle regeneration. 18085261_Knockout mice show variable defects resembling the Prader-Willi syndrome phenotype. 18085265_Ndn-deficiency in mice induces respiratory and serotonin alterations reminiscent of Prader Willi syndrome. 18272695_Lack of Necdin expression induces perinatal serotonergic alterations that affect the maturation and function of the respiratory network, inducing breathing deficits in mice and probably in Prader-Willi patients. 18570257_Results demonstrate a novel role for necdin in cellular migration, in addition to its roles in survival and axon outgrowth. 18753379_These results suggest that necdin downregulates p53 acetylation levels by forming a stable complex with p53 and Sirt1 to protect neurons from DNA damage-induced apoptosis. 18930956_Necdin regulates both the expression of the GnRH gene and GnRH neurons during their development. 19339547_Necdin is expressed in cachectic skeletal muscle to protect fibers from tumor-induced wasting. 19770359_we provide evidence that necdin plays an important role in restricting excessive Hematopoietic stem cell proliferation during hematopoietic regeneration 21502952_Necdin is a binding partner of dysbindin-1. Dysbindin-1 recruits necdin to the cytoplasm, thereby attenuating the repressive effects of necdin on p53 transcriptional activity. 21862615_demonstrated that CREB and FoxO1 are recruited to the necdin promoter, likely interacting with specific consensus sequences in the proximal region 21912643_Necdin is implicated through the TNF-receptor 1 pathway in the developmental death of motoneuron 22292082_Necdin prevents excessive preadipocyte proliferation induced by adipogenic stimulation to control white adipocyte number during adipose tissue development. 22442722_necdin has multiple roles within protein complexes in different subcellular compartments, and indicate that it can utilize multiple karyopherin-dependent pathways to modulate its localization. 22514318_These results suggest that necdin regulates Foxo1 acetylation and neuropeptide gene expression in the arcuate neurons to modulate the hypothalamic-pituitary-thyroid axis during development. 22776820_Necdin-null adult hematopoietic stem cells are less quiescent and more proliferative than normal hematopoietic stem cells, demonstrating the similar role of necdin and p53 in promoting hematopoietic stem cells quiescence during steady-state conditions. 22905258_necdin exerts its pro-survival activity by counteracting the action of the pro-apoptotic protein Cell Cycle Apoptosis Regulatory Protein (CCAR1/CARP1) 23785149_oxygen tension regulates the necdin protein level in NSCs through HIF-2alpha-mediated proteasomal 24039599_using several mouse models, we reveal a competition between non-imprinted Ndn promoters which results in monoallelic (paternal or maternal) Ndn expression, suggesting that Ndn allelic exclusion occurs in the absence of imprinting regulation. 24349431_Myc overexpression in the cpk kidney results from the dysregulation of the cystin-necdin regulatory complex and c-Myc, in turn, contributes to cystogenesis in the cpk mouse. 24392139_Antagonistic interplay between necdin and Bmi1 controls proliferation of neural precursor cells in the embryonic mouse neocortex. 24911587_Our data suggest that necdin suppresses PIAS1 both by inhibiting SUMO E3 ligase activity and by promoting ubiquitin-dependent degradation. 25012566_Necdin is a candidate downstream effector for Galphao. 26971449_Data reveal that necdin promotes mitochondrial biogenesis through stabilization of endogenous PGC-1alpha to exert neuroprotection against mitochondrial insults. 28528976_The Necdin expression was further associated with suppression of both cell proliferation and death in osteoblasts. 29087295_Overall, these results demonstrate that an increase of SERT activity is sufficient to cause the apneas in Necdin-knockout pups. 31433978_SENP2 Suppresses Necdin Expression to Promote Brown Adipocyte Differentiation. 32529326_The necdin interactome: evaluating the effects of amino acid substitutions and cell stress using proximity-dependent biotinylation (BioID) and mass spectrometry. 32667666_Necdin regulates BMAL1 stability and circadian clock through SGT1-HSP90 chaperone machinery. 34210967_Genetic dissection identifies Necdin as a driver gene in a mouse model of paternal 15q duplications. 35333696_Necdin, one of the important pathway proteins in the regulation of osteosarcoma progression by microRNA-200c. 36586641_Identification and characterization of Necdin as a target for the Cockayne syndrome B protein in promoting neuronal differentiation and maintenance. |
ENSG00000182636 |
NDN |
66.736173 |
0.4826121913 |
-1.051064 |
0.30983251 |
11.503517 |
0.00069464612847366003119120581033030248363502323627471923828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0033346516952129810415494937103630945784971117973327636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
44.1905289260487 |
9.56345744132213 |
91.5652976040554 |
14.1685585215794 |
| ENSMUSG00000033684 |
104009 |
Qsox1 |
protein_coding |
Q8BND5 |
FUNCTION: Catalyzes the oxidation of sulfhydryl groups in peptide and protein thiols to disulfides with the reduction of oxygen to hydrogen peroxide (PubMed:26819240). Plays a role in disulfide bond formation in a variety of extracellular proteins (PubMed:26819240). In fibroblasts, required for normal incorporation of laminin into the extracellular matrix, and thereby for normal cell-cell adhesion and cell migration (PubMed:26819240). {ECO:0000269|PubMed:26819240}. |
3D-structure;Alternative splicing;Disulfide bond;FAD;Flavoprotein;Glycoprotein;Golgi apparatus;Membrane;Oxidoreductase;Reference proteome;Secreted;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:104009; |
endoplasmic reticulum [GO:0005783]; extracellular exosome [GO:0070062]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; intercellular bridge [GO:0045171]; intracellular membrane-bounded organelle [GO:0043231]; FAD binding [GO:0071949]; flavin-linked sulfhydryl oxidase activity [GO:0016971]; protein disulfide isomerase activity [GO:0003756]; extracellular matrix assembly [GO:0085029]; negative regulation of macroautophagy [GO:0016242]; protein folding [GO:0006457] |
12354420_cloned skin SOx cDNA and characterized it as one of the disulfide (S-S) cross-linking enzymes 16160860_In seminiferous tubules, where a high level of expression was noticed, QSOX might play an important physiological role in sperm function and serve as a marker for the diagnosis of male infertility. 22801504_crystal structure 24475161_QSOX1 inhibits autophagy in breast cancer cells. 27581528_Serum thioredoxin reductase is highly increased in mice with hepatocellular carcinoma and its activity is restrained by several mechanisms, in particular, by the serum QSOX1. 29723491_the data indicated that QSOX1 is required 1) for a proper protein folding in the endo/sarcoplasmic reticulum (ER/SR) and 2) for resolution and protective response during acute stress. 29800099_our study provides the first comprehensive comparisons between QSOX1 and QSOX2 in the mouse epididymis, revealing their distinct epididymal distribution, cellular localization, mechanisms of secretion and sperm membrane association 31812669_Quiescin/sulfhydryl oxidase 1b (QSOX1b) induces migration and proliferation of vascular smooth muscle cells by distinct redox pathways 35228412_Polarized epithelium-sperm co-culture system reveals stimulatory factors for the secretion of mouse epididymal quiescin sulfhydryl oxidase 1. 35809071_Functional significance of mouse seminal vesicle sulfhydryl oxidase on sperm capacitation in vitro. 36245281_The disulfide catalyst QSOX1 maintains the colon mucosal barrier by regulating Golgi glycosyltransferases. |
ENSG00000116260 |
QSOX1 |
7449.214461 |
2.1127848134 |
1.079146 |
0.07213545 |
219.583121 |
0.00000000000000000000000000000000000000000000000011151052135701178084083366205317916746495329977786904926414404621938877568070286670424757102912299533300932583110350692067467073820274947593134129419922828674316406250000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000184622637098913420660459974949903246995398790466859964477947244139216189657032917626234790474599786090998099608063851484116013601521899545332416892051696777343750000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
10101.7244230035 |
505.301598840883 |
4781.23676351571 |
180.098142080225 |
| ENSMUSG00000033706 |
232187 |
Smyd5 |
protein_coding |
Q3TYX3 |
FUNCTION: Histone methyltransferase that specifically trimethylates 'Lys-20' of histone H4 to form trimethylated histone H4 lysine 20 (H4K20me3) which represents a specific tag for epigenetic transcriptional repression (PubMed:22921934, PubMed:28250819). In association with the NCoR corepressor complex, is involved in the repression of toll-like receptor 4 (TLR4)-target inflammatory genes in macrophages by catalyzing the formation of H4K20me3 at the gene promoters (PubMed:22921934). Plays an important role in embryonic stem (ES) cell self-renewal and differentiation (PubMed:28951459). Promotes ES cell maintenance by silencing differentiation genes through deposition of H4K20me3 marks (PubMed:28951459). Maintains genome stability of ES cells during differentiation through regulation of heterochromatin formation and repression of endogenous repetitive DNA elements by depositing H4K20me3 marks (PubMed:28250819). {ECO:0000269|PubMed:22921934, ECO:0000269|PubMed:28250819, ECO:0000269|PubMed:28951459}. |
Metal-binding;Methyltransferase;Reference proteome;S-adenosyl-L-methionine;Transferase;Zinc;Zinc-finger |
|
|
mmu:232187; |
histone H4K20 methyltransferase activity [GO:0042799]; histone H4K20 trimethyltransferase activity [GO:0140943]; histone lysine N-methyltransferase activity [GO:0018024]; metal ion binding [GO:0046872]; histone lysine methylation [GO:0034968]; negative regulation of gene expression, epigenetic [GO:0045814]; negative regulation of transposition [GO:0010529]; regulation of stem cell differentiation [GO:2000736]; regulation of stem cell division [GO:2000035] |
28250819_Depletion of SMYD5 leads to compromised self-renewal, including dysregulated expression of OCT4 targets, and perturbed differentiation. 35182940_SMYD5 is a histone H3-specific methyltransferase mediating mono-methylation of histone H3 lysine 36 and 37. |
ENSG00000135632 |
SMYD5 |
920.989927 |
2.0318158082 |
1.022770 |
0.10775698 |
88.936266 |
0.00000000000000000000407739533600706753145392555334242646987963712941844399216366479077322537705185823142528533935546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001687687112990751414709978551965790328054707879886509329543653468164166042697615921497344970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1237.53477863548 |
92.5192332963283 |
609.078231228492 |
34.7462852719893 |
| ENSMUSG00000033849 |
26878 |
B3galt2 |
protein_coding |
O54905 |
FUNCTION: Beta-1,3-galactosyltransferase that transfers galactose from UDP-galactose to substrates with a terminal beta-N-acetylglucosamine (beta-GlcNAc) residue. Can also utilize substrates with a terminal galactose residue, albeit with lower efficiency. Involved in the biosynthesis of the carbohydrate moieties of glycolipids and glycoproteins. {ECO:0000269|PubMed:9417047}. |
Glycoprotein;Glycosyltransferase;Golgi apparatus;Lipid metabolism;Manganese;Membrane;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. |
|
mmu:26878; |
Golgi membrane [GO:0000139]; glucosaminylgalactosylglucosylceramide beta-galactosyltransferase activity [GO:0047275]; glycosyltransferase activity [GO:0016757]; UDP-galactose:beta-N-acetylglucosamine beta-1,3-galactosyltransferase activity [GO:0008499]; UDP-glycosyltransferase activity [GO:0008194]; galactosylceramide biosynthetic process [GO:0006682]; oligosaccharide biosynthetic process [GO:0009312]; protein glycosylation [GO:0006486] |
33482286_beta-1, 3-galactosyltransferase 2 deficiency exacerbates brain injury after transient focal cerebral ischemia in mice. 33524473_beta-1, 3-galactosyltransferase 2 ameliorates focal ischemic cerebral injury by maintaining blood-brain barrier integrity. 36504040_Effects of the genetic knockout of the beta-1,3-galactosyltransferase 2 on spatial learning and neurons in the adult mouse hippocampus and somatosensory cortex. |
ENSG00000162630 |
B3GALT2 |
28.234760 |
0.0969186990 |
-3.367081 |
0.59571947 |
32.901709 |
0.00000000969379879088602743241601176864274957978295788052491843700408935546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001173735637382956938793020945123457554615242770523764193058013916015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.96023977877346 |
2.10689077146717 |
51.1793888319522 |
13.0897769128665 |
| ENSMUSG00000033952 |
12316 |
Aspm |
protein_coding |
Q8CJ27 |
FUNCTION: Involved in mitotic spindle regulation and coordination of mitotic processes. The function in regulating microtubule dynamics at spindle poles including spindle orientation, astral microtubule density and poleward microtubule flux seem to depend on its association with the katanin complex formed by KATNA1 and KATNB1. Enhances the microtubule lattice severing activity of KATNA1 by recruiting the katanin complex to microtubules. Can block microtubule minus-end growth and reversely this function can be enhanced by the katanin complex (PubMed:28436967). May have a preferential role in regulating neurogenesis. {ECO:0000269|PubMed:12355089, ECO:0000269|PubMed:28436967, ECO:0000269|PubMed:9819352}. |
3D-structure;Alternative splicing;Calmodulin-binding;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Microtubule;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:12316; |
apical plasma membrane [GO:0016324]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; meiotic spindle [GO:0072687]; microtubule [GO:0005874]; microtubule minus-end [GO:0036449]; midbody [GO:0030496]; mitotic spindle pole [GO:0097431]; nucleus [GO:0005634]; spindle pole [GO:0000922]; calmodulin binding [GO:0005516]; asymmetric cell division [GO:0008356]; brain development [GO:0007420]; cerebral cortex development [GO:0021987]; developmental growth [GO:0048589]; forebrain neuroblast division [GO:0021873]; maintenance of centrosome location [GO:0051661]; male gonad development [GO:0008584]; meiotic spindle assembly [GO:0090306]; negative regulation of asymmetric cell division [GO:0045769]; negative regulation of neuron differentiation [GO:0045665]; neuroblast proliferation [GO:0007405]; neuron migration [GO:0001764]; neuronal stem cell population maintenance [GO:0097150]; oogenesis [GO:0048477]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; positive regulation of neuroblast proliferation [GO:0002052]; regulation of meiotic cell cycle [GO:0051445]; spermatogenesis [GO:0007283]; spindle localization [GO:0051653]; spindle organization [GO:0007051] |
12351193_tissue distribution of Calmbp1 in fetal and adult mice 15045028_evolutionary selection of specific segments of the ASPM sequence strongly relates to differences in cerebral cortical size 16798874_Aspm is crucial for maintaining a cleavage plane orientation that allows symmetric, proliferative divisions of neuroepithelial cells during brain develo 18728014_NS5A protein down-regulates the expression of spindle gene Aspm through PKR-p38 signaling pathway 20823249_truncated Aspm proteins cause a massive loss of germ cells, resulting in a severe reduction in testis and ovary size accompanied by reduced fertility 21559369_AspM is expressed by proliferating cells of the adult mouse SVZ that can generate neuroblasts fated to become olfactory bulb neurons 21937711_knockdown of Aspm results in decreased Wnt-mediated transcription 23152892_ASPM plays a critical role in meiotic spindle assembly and meiotic progression in mouse oocytes. 24220505_Aspm is essential to the proliferation and differentiation of neural stem/progenitor cells. The Aspm gene loss model provided a novel pathogenetic insight into acquired microcephaly, which can be caused by in utero exposure teratogens. 26450969_Aspm, a gene that is mutated in familial microcephaly, regulates postnatal neurogenesis in the cerebellum and supports the growth of medulloblastoma, the most common malignant pediatric brain tumor. 26581405_Interaction between ASPM and the Cdk2/Cyclin E complex regulated the Cyclin activity by modulating its ubiquitination and localization into the nucleus. 27562601_in this study we have provided evidence that ASPM controls spindle orientation by regulating the dynamics of astral MT and that CITK is a critical downstream partner of ASPM for this activity. 27974163_Aspm and Wdr62 interact genetically to control brain size, with mice lacking Wdr62, Aspm, or both showing gene dose-related centriole duplication defects that parallel the severity of the microcephaly and increased ectopic basal progenitors, suggesting premature delamination from the ventricular zone. 32066665_A truncating Aspm allele leads to a complex cognitive phenotype and region-specific reductions in parvalbuminergic neurons. 35901620_Loss of abnormal spindle-like, microcephaly-associated (Aspm) disrupts female folliculogenesis in mice during maturation and aging. |
ENSG00000066279 |
ASPM |
1128.359286 |
2.5142630784 |
1.330136 |
0.09661717 |
186.909209 |
0.00000000000000000000000000000000000000000150318468097223334223615213907723953833785514446371238043640621269278756692609018369076070745002494928957165881699964415929571259766817092895507812500000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000001908042421714088121106500906884078900764620491950235209680150597759643479132387395355188460099416802059728304197250281504238955676555633544921875000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1624.98935845184 |
108.906773406947 |
646.308404393885 |
33.3759792413049 |
| ENSMUSG00000034023 |
211651 |
Fancd2 |
protein_coding |
Q80V62 |
FUNCTION: Required for maintenance of chromosomal stability. Promotes accurate and efficient pairing of homologs during meiosis. Involved in the repair of DNA double-strand breaks, both by homologous recombination and single-strand annealing. May participate in S phase and G2 phase checkpoint activation upon DNA damage. Plays a role in preventing breakage and loss of missegregating chromatin at the end of cell division, particularly after replication stress (By similarity). Promotes BRCA2/FANCD1 loading onto damaged chromatin. May also be involved in B-cell immunoglobulin isotype switching. {ECO:0000250, ECO:0000269|PubMed:12893777}. |
3D-structure;Alternative splicing;Cell cycle;Developmental protein;DNA damage;DNA repair;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:211651; |
condensed chromosome [GO:0000793]; cytosol [GO:0005829]; DNA repair complex [GO:1990391]; nuclear body [GO:0016604]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA polymerase binding [GO:0070182]; brain morphogenesis [GO:0048854]; cellular response to DNA damage stimulus [GO:0006974]; cellular response to oxidative stress [GO:0034599]; double-strand break repair involved in meiotic recombination [GO:1990918]; gamete generation [GO:0007276]; homologous chromosome pairing at meiosis [GO:0007129]; interstrand cross-link repair [GO:0036297]; mitotic intra-S DNA damage checkpoint signaling [GO:0031573]; neuronal stem cell population maintenance [GO:0097150]; regulation of CD40 signaling pathway [GO:2000348]; regulation of DNA-binding transcription factor activity [GO:0051090]; regulation of inflammatory response [GO:0050727]; regulation of regulatory T cell differentiation [GO:0045589]; response to gamma radiation [GO:0010332] |
12893777_Fanconi anemia complementation group D2 knockout mice develop epithelial cancer. 15199141_results demonstrate that monoubiquitination of FANCD2, which is regulated by the FA pathway, promotes BRCA2 loading into chromatin complexes. These complexes appear to be required for normal homology-directed DNA repair. 15377654_FANCD2, which is activated by the Fanconi anemia complex, is required to maintain the G2 checkpoint 15665282_Fancd2 deficient and Trp53 deficient cells showed an increase in aneuploidy and had multiple gross chromosomal rearrangements. 16127665_Homozygous germ-line mutation in exon 27 of Brca2 disrupts the Fancd2-Brca2 pathway in the homologous recombination-mediated DNA interstrand cross-links' repair, but does not affect meiosis. 16135554_The role of Fancd2 in DSB repair may account for the moderate sensitivity of FA cells to irradiation and FA cells sensitivity to ICLs that are repaired via a DSB intermediate. 18482162_Chronic exposure to ethanol induced FANCD2 in the midbrain; however, there was no concomitant increase in the amount of ubiquitinated FANCD2. 19217432_mouse Usp1 functions downstream in the Fanconi anemia pathway, and its deubiquitination is required for Fancd2 nuclear foci assembly 20506303_Bone marrow from Fancd2-/- mice and Usp1-/- mice exhibited marked hematopoietic defects. 20805509_found that C/EBPdelta promotes monoubiquitination of the Fanconi anemia complementation group D2 protein (FANCD2), which is necessary for its function in replication-associated DNA repair 20935219_Data sshow that K14E7/FancD2(-/-) mice had a significantly higher incidence of HNSCC compared with K14E7/FancD2(+/+) mice. 21118969_Data show that Fancd2(-/-) mice displayed a higher magnitude of chromosomal breakage and micronucleus formation than the wild-type or Fancg(-/-) mice. 21734703_the Fanconi anaemia DNA repair pathway counteracts acetaldehyde-induced genotoxicity in mice; Aldh2(-/-)Fancd2(-/-) mice spontaneously develop acute leukaemia 21764741_crystal structure of FANCI-FANCD2(ID) complex; crystallographic electron-density map of FANCI protein bound to splayed Y DNA; data suggest ID complex recognizes DNA structures resulting from encounter of replication forks with an interstrand cross-link 23417416_Studies indicate that in Fancd2-Knockout mice, the formation of sex-cords and intraovarian tubules lead to the formation of tumours with multiple phenotypes including luteomas, papillary cysts and malignant carcinomas. 23806336_Fancd2-Ub activates the transcription of the tumor suppressor TAp63, thereby promoting cellular senescence and blocking skin tumorigenesis. 24086435_E7 induces human papillomavirus-associated head and neck cancers by promoting DNA damage through the inactivation of pocket proteins. 24483844_Combined deficiency of Foxo3a and Fancc or Fancd2 not only impairs the self-renewal capacity but also markedly increases the apoptosis of neural stem and progenitor cells (NSPCs), leading to defective neurogenesis. 24501220_CD25(+)Foxp3(+) Tregs of Fanca(-/-) or Fancd2(-/-) mice were less efficient in suppressing the production of GVHD-associated inflammatory cytokines. 25505262_Data demonstrated that Fancd2 was required for nuclear retention of CA-FOXO3a and for maintaining hematopoietic repopulation of the HSCs. 26412304_Results show that FANCD2 and ADH5 protect hematopoietic stem cells, hepatocytes, and nephrons from endogenous DNA damage resulting from accumulation of endogenous formaldehyde. 26783108_Data that suggest Usp1 (ubiquitin specific peptidase 1) down-regulation by autocleavage is critical for Usp1 to exert role in DNA interstrand crosslink repair; Usp1 role is de-ubiquitination of Fancd2 and Pcna (proliferating cell nuclear antigen). 26797144_recruitment of Fan1 by ubiquitinated-Fancd2 is dispensable for DNA interstrand cross-links repair 27637088_Data show that stromal cell lines derived from both K14E7 Fancd2-/- and Fancd2-/- cultures were radiosensitive. 27720904_Fancd2-/- mice exhibit a pervasive developmental HSPC defect that echoes the constitutional defects evident at birth in a subset of FA patients. 27915139_loss of Fancd2 yields significant defects to fetal liver hematopoiesis, particularly the HSC population, which mimics key phenotypes from adult Fancd2 KO bone marrow independently of aging-accrued DNA damage. 28378742_Fancd2 localizes in the mitochondrion and associates with the nucleoid complex components Atad3 and Tufm. 30254368_Data indicate that linker of T-cell receptor pathways protein(Lnk) deficiency restores phenotypic hematopoietic stem cells (HSCs) in Fanconi anemia, complementation group D2 protein knockout (Fancd2-/-) mice. 31078270_The anemia observed in the Fancd2(-/-) mice has not been described in other Fanconia anemia models. 31219578_Studies indicate common and distinct functions of Fanconi anemia complementation group I (FANCI) and Fanconi Anemia Group D2 Protein (FANCD2) during mouse development, meiotic recombination and hematopoiesis. 31434739_Upon Paigen diet challenge, male Fancd2 (-/-) mice had altered expression of genes encoding hepatic bile acid transporters and cholesterol and fatty acid metabolism proteins. 31472450_Deletion of Fancd2 in mouse hematopoietic stem and progenitor cells leads to increase in mitochondrial number, and enzyme activity of mitochondrion-encoded respiratory complexes. 32163912_FANCD2 is required for the repression of germline transposable elements. 32717583_Novel TALEN-generated mCitrine-FANCD2 fusion reporter mouse model for in vivo research of DNA damage response. 33222180_FANCD2 and HES1 suppress inflammation-induced PPAR to prevent haematopoietic stem cell exhaustion. |
ENSG00000144554 |
FANCD2 |
728.541057 |
2.7239986451 |
1.445726 |
0.10743773 |
178.325503 |
0.00000000000000000000000000000000000000011247088615321369486024225103685415392395226737095938854242256126805974873522927246343991884114579471389168408013148336976883001625537872314453125000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000130775308235553493128244698125775564093955550836145294387430210404360086657800818082236751343575540708352722418794655823148787021636962890625000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1055.07091919716 |
78.6439444277752 |
387.324318642258 |
22.5496394988362 |
| ENSMUSG00000034127 |
216350 |
Tspan8 |
protein_coding |
Q8R3G9 |
|
Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:216350; |
cell surface [GO:0009986]; plasma membrane [GO:0005886]; integrin binding [GO:0005178]; negative regulation of blood coagulation [GO:0030195]; regulation of gene expression [GO:0010468]; spermatogenesis [GO:0007283] |
20531244_Results suggest that alterations in gene expression of Gata6, Tspan8, S100a8, and Lmo2 may act via novel pathways that play functionally important roles in Men1-associated tumor progression. 20733586_results argue for a role for Tspan8 in body-weight regulation in males, but do not show differences in T2D-associated traits that were anticipated from previous human genome-wide association studies. 27733379_Transplantation studies provided definitive evidence that a TSPAN8-high subpopulation is enriched for spermatogonial stem cells. 28777493_Single nucleotide polymorphism in TSPAN8 gene is associated with pathogenesis of bipolar disorder. 32195353_Extracellular vesicle tetraspanin-8 level predicts distant metastasis in non-small cell lung cancer after concurrent chemoradiation. |
ENSG00000127324 |
TSPAN8 |
41.596767 |
0.0804838792 |
-3.635156 |
0.52230684 |
51.682481 |
0.00000000000065242304309402204625785176525440943202244215015639383636880666017532348632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000132840506151442493118006775132310874447394422048773776623420417308807373046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.86019720284494 |
2.16825041682882 |
72.8120619882024 |
15.2106007926835 |
| ENSMUSG00000034156 |
207777 |
Tspoap1 |
protein_coding |
Q7TNF8 |
|
Alternative splicing;Cytoplasm;Mitochondrion;Reference proteome;Repeat;SH3 domain |
|
|
mmu:207777; |
calyx of Held [GO:0044305]; cytoplasm [GO:0005737]; glutamatergic synapse [GO:0098978]; mitochondrion [GO:0005739]; benzodiazepine receptor binding [GO:0030156]; voltage-gated calcium channel activity involved in regulation of presynaptic cytosolic calcium levels [GO:0099626] |
33539324_Biallelic variants in TSPOAP1, encoding the active-zone protein RIMBP1, cause autosomal recessive dystonia. |
ENSG00000005379 |
TSPOAP1 |
59.064471 |
3.1530196364 |
1.656734 |
0.40228970 |
16.097725 |
0.00006015624758817488811979759533166145502036670222878456115722656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003764584894260804802390563139624646282754838466644287109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
83.8069948979464 |
23.6732685970233 |
26.5799153069803 |
5.77132224437456 |
| ENSMUSG00000034206 |
77782 |
Polq |
protein_coding |
Q8CGS6 |
FUNCTION: DNA polymerase that promotes microhomology-mediated end-joining (MMEJ), an alternative non-homologous end-joining (NHEJ) machinery triggered in response to double-strand breaks in DNA. MMEJ is an error-prone repair pathway that produces deletions of sequences from the strand being repaired and promotes genomic rearrangements, such as telomere fusions, some of them leading to cellular transformation (PubMed:25275444, PubMed:25642963). POLQ acts as an inhibitor of homology-recombination repair (HR) pathway by limiting RAD51 accumulation at resected ends (PubMed:25642963). POLQ-mediated MMEJ may be required to promote the survival of cells with a compromised HR repair pathway, thereby preventing genomic havoc by resolving unrepaired lesions (PubMed:25642963). The polymerase acts by binding directly the 2 ends of resected double-strand breaks, allowing microhomologous sequences in the overhangs to form base pairs. It then extends each strand from the base-paired region using the opposing overhang as a template. Requires partially resected DNA containing 2 to 6 base pairs of microhomology to perform MMEJ. The polymerase activity is highly promiscuous: unlike most polymerases, promotes extension of ssDNA and partial ssDNA (pssDNA) substrates. Also exhibits low-fidelity DNA synthesis, translesion synthesis and lyase activity, and it is implicated in interstrand-cross-link repair, base excision repair and DNA end-joining (By similarity). Involved in somatic hypermutation of immunoglobulin genes, a process that requires the activity of DNA polymerases to ultimately introduce mutations at both A/T and C/G base pairs (PubMed:16222339, PubMed:16172387, PubMed:16890500, PubMed:17449470). However, POLQ does not play a major role in somatic hypermutation (PubMed:18485835). {ECO:0000250|UniProtKB:O75417, ECO:0000269|PubMed:16172387, ECO:0000269|PubMed:16222339, ECO:0000269|PubMed:16890500, ECO:0000269|PubMed:17449470, ECO:0000269|PubMed:18485835, ECO:0000269|PubMed:21883722, ECO:0000269|PubMed:25275444, ECO:0000269|PubMed:25642963}. |
Acetylation;Alternative splicing;ATP-binding;Chromosome;Coiled coil;DNA damage;DNA repair;DNA-directed DNA polymerase;Nucleotide-binding;Nucleotidyltransferase;Nucleus;Reference proteome;Transferase |
|
|
mmu:77782; |
chromosome [GO:0005694]; cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; nucleoplasm [GO:0005654]; 5'-deoxyribose-5-phosphate lyase activity [GO:0051575]; ATP binding [GO:0005524]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-directed DNA polymerase activity [GO:0003887]; identical protein binding [GO:0042802]; single-stranded DNA helicase activity [GO:0017116]; base-excision repair [GO:0006284]; cellular response to DNA damage stimulus [GO:0006974]; DNA repair [GO:0006281]; DNA-templated DNA replication [GO:0006261]; double-strand break repair [GO:0006302]; double-strand break repair via alternative nonhomologous end joining [GO:0097681]; negative regulation of double-strand break repair via homologous recombination [GO:2000042]; protein homooligomerization [GO:0051260]; somatic hypermutation of immunoglobulin genes [GO:0016446] |
14735462_DNA Pol theta has a specialized function in lymphocytes and in tumor progression 15542845_Polq has a role in maintaining genomic integrity, which is probably distinctive from the major homologous recombination pathway regulated by ATM 16172387_Polq-inactive mice exhibited a reduced level of serum IgM and IgG1. 16222339_dominant role in Ig somatic hypermutation possibly by introducing mismatches while bypassing abasic sites generated by UDG-mediated deglycosylation of AID-effected dU, by extending DNA past such abasic sites & by synthesizing DNA during mismatch repair 16436048_These results reveal an important role for Poltheta in preventing spontaneous cell death and in tolerance of not only DNA interstrand cross-links and double strand breaks but also UV adducts and alkylation damage in mammalian lymphocytes. 16890500_POLQ has a role in somatic hypermutation. 17449470_Results reveal genetic and biochemical interactions between DNA polymerases eta (POLH) and theta (POLQ) and suggest that POLQ might cooperate with POLH to generate some of the A/T mutations during the somatic hypermutation of Ig genes. 19630521_The sensitivity of POLQ-defective bone marrow stromal cells to ionizing radiation and bleomycin and the increase in micronuclei in red blood cells support a role for this DNA polymerase in cellular tolerance of DNA damage. 21883722_the polymerase and other functional domains of POLQ both play important roles in tolerance to etoposide and gamma-irradiation. 25275444_A role for POLQ in an alternative end joining pathway that suppresses the formation of chromosomal translocations. 25642960_loss of Polq in mice results in increased rates of homology-directed repair, evident by recombination of dysfunctional telomeres and accumulation of RAD51 at double-stranded breaks 25642963_genetic inactivation of an homologous-recombination gene (Fancd2) and Polq in mice results in embryonic lethality 27311885_Here, the authors discover that mammalian Poltheta; transfers nucleotides to the 3' terminus of DNA during alt-EJ in vitro and in vivo by oscillating between three different modes of terminal transferase activity: non-templated extension, templated extension in cis, and templated extension in trans. 27453047_DNA polymerase theta mediates end joining repair of chromosome breaks and helps to sustain cell viability and genome stability by rescuing chromosome break repair when resection is misregulated or NHEJ is compromised. 29058711_Poltheta;-helicase promotes chromosomal translocations by alternative nonhomologous end joining in mouse embryonic stem cells and also suppresses CRISPR-Cas9- mediated gene targeting by homologous recombination. Poltheta;-helicase activity facilitates the removal of RPA from resected double-strand breaks to allow their annealing and subsequent joining by alternative nonhomologous end joining. 29079701_Classical non-homologous end-joining (cNHEJ) and polymerase theta-mediated end-joining (TMEJ) act both parallel and redundant in mouse embryonic stem cells and account for virtually all end-joining activity. 29215638_DNA polymerase theta, the versatile mediator of microhomology-based repair of DNA double-strand breaks and stalled replication forks, is now revealed to be a member of an elite group of proteins known as annealing helicases. |
ENSG00000051341 |
POLQ |
369.668898 |
2.1840798842 |
1.127026 |
0.15920424 |
49.561218 |
0.00000000000192277226836362647244983052199141974154469880708973050786880776286125183105468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000370729964452085535001684583649090350826460493749436864163726568222045898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
473.27782699221 |
52.3332033547672 |
216.694375704284 |
18.3851258223222 |
| ENSMUSG00000034258 |
217721 |
Flvcr2 |
protein_coding |
A0A0R4J0E9 |
Human_homologues FUNCTION: Acts as an importer of heme. Also acts as a transporter for a calcium-chelator complex, important for growth and calcium metabolism. {ECO:0000269|PubMed:20823265}. |
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:217721; |
membrane [GO:0016020]; heme binding [GO:0020037]; heme transmembrane transporter activity [GO:0015232] |
32369449_Deficiency of MFSD7c results in microcephaly-associated vasculopathy in Fowler syndrome. 32369453_Lack of Flvcr2 impairs brain angiogenesis without affecting the blood-brain barrier. 32973183_MFSD7C switches mitochondrial ATP synthesis to thermogenesis in response to heme. |
ENSG00000119686 |
FLVCR2 |
66.472492 |
0.2351636463 |
-2.088263 |
0.39270961 |
27.310350 |
0.00000017328136772185350436721856109967276893257803749293088912963867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000017526041123102739365549824004930634657739574322476983070373535156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.1784774662013 |
7.21425392487082 |
111.320256694622 |
21.3976104271261 |
| ENSMUSG00000034265 |
224454 |
Zdhhc14 |
protein_coding |
Q8BQQ1 |
FUNCTION: Palmitoyltransferase that could catalyze the addition of palmitate onto various protein substrates. May have a palmitoyltransferase activity toward the beta-2 adrenergic receptor/ADRB2 and thereby regulate G protein-coupled receptor signaling. May play a role in cell differentiation and apoptosis. {ECO:0000250|UniProtKB:Q8IZN3}. |
Acyltransferase;Alternative splicing;Endoplasmic reticulum;Golgi apparatus;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Reference proteome;Transferase;Transmembrane;Transmembrane helix |
|
|
mmu:224454; |
endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; palmitoyltransferase activity [GO:0016409]; protein-cysteine S-palmitoyltransferase activity [GO:0019706]; peptidyl-L-cysteine S-palmitoylation [GO:0018230]; protein palmitoylation [GO:0018345]; protein targeting to membrane [GO:0006612] |
33185190_The palmitoyl acyltransferase ZDHHC14 controls Kv1-family potassium channel clustering at the axon initial segment. |
ENSG00000175048 |
ZDHHC14 |
20.142242 |
0.0443191254 |
-4.495927 |
0.83442418 |
30.608028 |
0.00000003157844440706933960815515718446866433311015498475171625614166259765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003549836643605020097101015487395248015900506288744509220123291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.72062650934755 |
1.05159389172301 |
38.8235664267542 |
12.8257946581175 |
| ENSMUSG00000034311 |
16571 |
Kif4 |
protein_coding |
P33174 |
FUNCTION: Iron-sulfur (Fe-S) cluster binding motor protein that has a role in chromosome segregation during mitosis (By similarity). Required for mitotic chromosomal positioning and bipolar spindle stabilization. {ECO:0000250|UniProtKB:O95239, ECO:0000269|PubMed:7929562}. |
3D-structure;ATP-binding;Chromosome;Coiled coil;Cytoplasm;Cytoskeleton;DNA-binding;Iron;Iron-sulfur;Metal-binding;Microtubule;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:16571; |
chromosome [GO:0005694]; cytosol [GO:0005829]; microtubule [GO:0005874]; microtubule associated complex [GO:0005875]; midbody [GO:0030496]; nucleoplasm [GO:0005654]; ATP binding [GO:0005524]; DNA binding [GO:0003677]; iron-sulfur cluster binding [GO:0051536]; metal ion binding [GO:0046872]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule-based movement [GO:0007018]; mitotic cytokinesis [GO:0000281]; mitotic spindle midzone assembly [GO:0051256]; mitotic spindle organization [GO:0007052]; spindle elongation [GO:0051231] |
16630823_Results suggest that KIF4 controls the activity-dependent survival of postmitotic neurons by regulating PARP-1 activity in brain development. 16890532_Embryonic stem cells genetically depleted of KIF4 support anchorage-independent growth and form tumors in nude mice. 19158085_KIF4 is involved in the anterograde trafficking of ribosomal constituents to axons 23500491_Closure of the backdoor by ATP binding and hydrolysis stabilizes two mobile regions, switch I and switch II, to generate the phosphate tube from which hydrolyzed phosphate is released. 24658398_Kif4 is necessary for efficient migration of cells. 28125646_Further investigation of the phosphoregulation of meiotic Kif4 revealed that Aurora Kinase and Cdk activity is critical for Kif4 kinetochore localization and interaction with Ndc80 and CENP-C. Finally, Kif4 protein but not gene expression was found to be upregulated with age, suggesting a role for this protein in the decline of oocyte quality with age 28350061_the silencing of Kif4 regulated VRGFR1 mRNA transcription and sVEGFR1 production in monocytes via the PI3K/Akt signaling pathway, which may be one of the mechanisms responsible for these processes. 30081192_KIF4 was first found in neuron, then in chromatin and later in cytoplasm. It is expressed in various types of cells including cancer cells and germ cells. KIF4 also showed dynamic changes throughout the cell cycle. It is abnormally expressed in many cancers. [review] 31067151_This work reveals essential roles for Kif4 during the meiotic divisions. 31486502_Whole genome expression microarray reveals novel roles for Kif4 in monocyte/macrophage cells. 35579738_CircKIF4A enhances osteosarcoma proliferation and metastasis by sponging MiR-515-5p and upregulating SLC7A11. 36482480_KIF4 regulates neuronal morphology and seizure susceptibility via the PARP1 signaling pathway. |
ENSG00000090889 |
KIF4A |
772.106139 |
2.3713882275 |
1.245732 |
0.14700449 |
70.696969 |
0.00000000000000004165318649876378794752795619564600449974210689225648240885391260235337540507316589355468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000012817400742407473952062928766534769633067043391114192552038275607628747820854187011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1098.26253717092 |
112.102038631361 |
463.130635649936 |
35.9290012810796 |
| ENSMUSG00000034394 |
16878 |
Lif |
protein_coding |
P09056 |
FUNCTION: LIF has the capacity to induce terminal differentiation in leukemic cells. Its activities include the induction of hematopoietic differentiation in normal and myeloid leukemia cells, the induction of neuronal cell differentiation, and the stimulation of acute-phase protein synthesis in hepatocytes. |
3D-structure;Cytokine;Direct protein sequencing;Disulfide bond;Glycoprotein;Growth factor;Reference proteome;Secreted;Signal |
|
|
mmu:16878; |
cytosol [GO:0005829]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; leukemia inhibitory factor receptor binding [GO:0005146]; signaling receptor binding [GO:0005102]; animal organ regeneration [GO:0031100]; astrocyte differentiation [GO:0048708]; blood vessel remodeling [GO:0001974]; cell population proliferation [GO:0008283]; decidualization [GO:0046697]; embryo implantation [GO:0007566]; fibroblast proliferation [GO:0048144]; immune response [GO:0006955]; leukemia inhibitory factor signaling pathway [GO:0048861]; lung alveolus development [GO:0048286]; lung development [GO:0030324]; lung lobe morphogenesis [GO:0060463]; lung vasculature development [GO:0060426]; maternal process involved in female pregnancy [GO:0060135]; meiotic nuclear division [GO:0140013]; muscle organ morphogenesis [GO:0048644]; negative regulation of angiogenesis [GO:0016525]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; negative regulation of hormone secretion [GO:0046888]; negative regulation of meiotic nuclear division [GO:0045835]; neuron development [GO:0048666]; positive regulation of astrocyte differentiation [GO:0048711]; positive regulation of cell adhesion mediated by integrin [GO:0033630]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of corticotropin secretion [GO:0051461]; positive regulation of fibroblast proliferation [GO:0048146]; positive regulation of gene expression [GO:0010628]; positive regulation of histone H3-K27 acetylation [GO:1901676]; positive regulation of macrophage differentiation [GO:0045651]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of mesenchymal to epithelial transition involved in metanephros morphogenesis [GO:0072108]; positive regulation of neuron projection development [GO:0010976]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of peptidyl-serine phosphorylation of STAT protein [GO:0033141]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of protein localization to nucleus [GO:1900182]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531]; regulation of cell differentiation [GO:0045595]; regulation of metanephric nephron tubule epithelial cell differentiation [GO:0072307]; regulation of RNA polymerase II regulatory region sequence-specific DNA binding [GO:1903025]; retina development in camera-type eye [GO:0060041]; somatic stem cell population maintenance [GO:0035019]; spongiotrophoblast differentiation [GO:0060708]; stem cell differentiation [GO:0048863]; transdifferentiation [GO:0060290]; trophoblast giant cell differentiation [GO:0060707]; tyrosine phosphorylation of STAT protein [GO:0007260] |
11683174_increases myoblast replication and survival and affects extracellular matrix production 11855863_LIF induces both ERK-1 and STAT3 activation in mouse P19 EC cells. LIF enhances the cell proliferation rate, which depends on ERK activity but does not require activation of STAT3. 11897091_supportes the expression of calcitonin gene related peptide in the primary sensory neurons of adult mouse lumbar dorsal root ganglia and in their outgrowing axons 11934677_although LIF induces ACTH, SOCS-3 acts to counterregulate the HPA axis response to inflammation. 11950938_CD9 may play a role in LIF-mediated maintenance of undifferentiated ES cells. 12007288_controls utrine receptivity and nidation (REVIEW) 12084939_role in inhibiting neuronal terminal differentiation through STAT3 activation 12124777_LIF is one of the keratinocyte-derived factors involved in regulating the proliferation and differentiation of neonatal mouse epidermal melanocytes 12392991_LIF stimulation down-regulates several key components of the remodeling process, including collagen content and matrix metalloproteinase (MMP) activation 12490192_Leukemia inhibitory factor (LIF) in the mouse mammary gland may play a role during the first stage of mammary gland involution. LIF-induced mammary epithelium apoptosis could be mediated by Stat3 activation. 12591604_LIF and IGF-I, thus, have cooperative and distinct tissue functions during development 12629183_Leukemia inhibitory factor is a key signal for neurogenesis in injured adult mouse olfactory epithelium 12764151_LIF has a role in src family kinase-independent signal transduction and gene induction 12782633_LIF may be an important regulator of B cell-mediated responses and oxidant injury in the lung. 12860906_In a mouse myocardial infarction model left ventricular remodeling, infarct extent and myocardial fibrosis were markedly attenuated and neovascularization induced in LIF cDNA-injected mice 14695905_LIF is a critical factor for the normal development of embryos at the preimplantation stages. 14741342_Haploinsufficiency of the Igf-I gene on a Lif null background caused a marked reduction in body mass index and white adipose tissue in female mice. 14970834_The LIF knockout mice show reduced immobility in the forced swim test, suggesting that LIF might have a potential role in the etiology of some forms of depression. 14976223_Msx-1 and Wnt4 signaling during implantation becomes abberant in mice with deleted leukemia inhibitory factor. 15246839_LIF is a potent pro-inflammatory factor in the adult spinal cord and represents a potential target for the manipulation of inflammatory reactions after spinal cord injury 15488701_Leukemia inhibitory factor and vascular endothelial growth factor have roles in wound healing by increasing angiogenesis and matrix production 15519241_LIF enhances neural stem cell self-renewal in the context of ongoing proliferation induced by epidermal growth factor (EGF) and fibroblast growth factor-2 (FGF-2). 15576827_alterations of the uterine leukocyte subpopulations in Lif knockout mice may disrupt pregnancy and contribute to failure of implantation 15597051_IGF-I and LIF regulate cooperatively motoneuron numbers in specific brain stem nuclei. 15618884_LIF may be useful when developing stem cell therapy to replace damaged olfactory sensory neurons. 15634701_demonstrate a novel region-specific role for ciliary neurotrophic factor/LIF/gp130 signaling in the development of the germinal layers of the embryonic telencephalon 15647843_signaling other than the activation of STAT3 and MEK1 may be necessary for M1 cell-growth arrest and differentiation, while a set of early genes of LIF are induced by only STAT3 activation 15673569_LIF and STAT3 regulate embryonic stem cell self-renewal and pluripotency 15716414_LIF deficiency leads to pronounced loss of distal axons and motor endplate alterations. LIF plays a more important role for postnatal maintenance of distal axons and motor endplates than CNTF or CT-1. 15785381_role for LIF in the regulation of transplantation tolerance in vivo 15848385_Data point to a profound disturbance of normal uterine luminal epithelial and stromal differentiation during early pregnancy in leukemia inhibitory factor-null mice. 16099433_removal of LIF from culture media results in downregulation of chromatin-related proteins in pluripotent mouse embryonic stem cells 16206277_LIF transgene reduced the numbers of cells showing markers characteristic of mature retinal neurons and caused synaptic ectopia. The net effect was disrupted morphological development and disturbed synaptic organization. 16291739_induces DNA replication without increasing cyclin D1 expression; induces DNA synthesis without promoting full phosphorylation of retinoblastoma protein. 16314487_LIF and BMP signaling generate different astrocytic cell types, and propose that these cells are, respectively, adult progenitor cells and mature astrocytes. 16369772_expression and function of LIF and its receptor in the pancreas 16407199_LIF regulates the commitment of cardiac stem cells into the endothelial cell lineage, contributing to neovascularization in the process of tissue remodeling and/or regeneration. 16458863_LIF is required for normal glial responses to brain damage caused by pilocaarpine, and, as in the periphery, LIF regulates neuropeptide Y expression in the central nervous system. 16467531_In embryonic deficient mice, preganglionic sympathetic neurons were reduced. (Leukemia inhibitory factor receptor beta) 16498619_We conclude that endogenous LIF is induced in response to autoimmune demyelination in the spinal cord and protects mature oligodendrocytes from demyelinating injury and cell death, thereby resulting in attenuation of clinical disease severity. 16636663_DSP2 acts as a negative regulator of the IL-6/LIF/STAT3-mediated signaling pathway. 16648972_Since we demonstrated that not only SDF-1, but also HGF and LIF are upregulated in damaged tissues, we postulate that CXCR4+ c-Met+ LIF-R+ TCSC could be mobilized from the BM into the PB, where they play a role in tissue repair/regeneration. 16691571_LIF collaborates with insulin-like growth factor 1 in lung alveolar epithelium and vascular maturation 16781162_LIF expression in loaded skeletal muscle is critical for the development of skeletal muscle hypertrophy in the functional overload model 16790614_Ovarian stimulation may inhibit the expression of endometrial LIF and impair endometrial receptivity in mice. 16987252_Activation of mGlu5 receptors sustains the increase in c-Myc induced by leukemia inhibitory factor in embryonic stem cells by inhibiting both glycogen synthase kinase-3beta and phosphatidylinositol 3-kinase. 17021343_These results demonstrate that LIF is useful in the initiation of germline stem cell culture and suggest that LIF or a related cytokine is involved in the maturation of gonocytes into spermatogonia. 17108182_Our results have implications for the development of therapeutic strategies for brain repair and suggest that LIF may be useful, in combination with other factors, in promoting regeneration in the adult brain. 17150369_Induction of interleukin-6 mRNA after infarct was significantly abrogated in cardiotrophin-1 null mice compared to wild-type mice 17249568_leukemia inhibitory factor regulates both rod and cone gene expression, while Gp130 regulates cone gene expression 17313947_Lif null and Hoxa10 null mice, in which implantation is impaired, have a similar number of endometrial pinopodes compared to fertile ICR mice, therefore pinopodes are not a good indicator of receptivity in mice. 17332343_LIF-LIF-R axis may direct rhabdomyosarcoma metastasis. 17356004_Switch-like LIF signaling controls a positive feedback loop for self-renewal in embryonic stem cells. High exogenous LIF maintains the 'on' state: transcriptional expression of LIF signaling pathway components. Low LIF signals the 'off' state. 17360716_HIF-1alpha mediated suppression of LIF-STAT3 signaling. 17451373_LIF inhibits osteoblast development at least in part by stimulating high molecular weight hyaluronan synthesis through hyaluronan synthase 2 17680539_Lif mRNA expression is related to the implantation of ovarian stimulated embryos, but not parallel to the number of implantation . 17935626_LIF is a powerful tool to control neural differentiation and maintenance of stem cell-derived murine spiral ganglion neuron precursors 18046411_results demonstrate a function for p53 in maternal reproduction through the regulation of LIF 18088375_Our data suggest that photoreceptor protection in response to LIF can be directly mediated by activation of STAT3 in photoreceptors. 18239353_LIF, which has a crucial role in E2 action for initiation of implantation, causes transient induction of Pgr mRNA and subsequent upregulation of Ihh mRNA, which mediates progesterone-Pgr actions for successful implantation 18278460_endometrial LIF is increased in mice with embryonic implantation dysfunction after administration of the Bushenantai recipe of traditional Chinese medicine 18293407_Thus endogenous LIF receptor signaling is not only protective of oligodendrocytes but can also enhance remyelination, and exogenous LIF has therapeutic potential in limiting the consequences of oligodendrocyte damage. 18337516_In Lif-deficient mice, immunostaining indicated a reduction of endometrial IL1A at the time of implantation and of IL1B in stroma. 18385284_Neuroinflammation facilitates LIF entry into brain by a TNF-dependent pathway. 18451094_Leukemia inhibitory factor regulates trophoblast giant cell differentiation via Janus kinase 1-signal transducer and activator of transcription 3-suppressor of cytokine signaling 3 pathway 18521186_Tissues and advancing vasculature communicate to ensure adequate vascularization using LIF as well as oxygen, which suggests a new strategy for antiangiogenic therapy in human diseases such as diabetic retinopathy and cancer. 18546535_Superovulation induced by GnRHa down regulates the expressions of ER, PR and the LIF mRNA on the mice of secretive phase endometrium. 18548006_The bones of Fra-2-deficient newborn mice have giant osteoclasts, and signalling through leukaemia inhibitory factor (LIF) and its receptor is impaired 18552402_TNFalpha promotes myoblast proliferation through JNK1 and prevents myoblast differentiation through JNK1-mediated secretion of LIF 18829014_Leukemia inhibitory factor (LIF) is not necessary in establishing and self-renewing embryonic stem cells (ESCs). 18841027_results suggest a role for LIF as an important factor controlling the proliferative potential of hematopoietic stem cells and their respective progenitor cells 19021297_LIF plays a central role in maintaining the size and integrity of the population of immature neurons within the olfactory epithelium; this population is critical to the rapid recovery of olfactory function after injury. 19091967_Our data establish LIF as a Muller cell derived neuronal survival factor which controls an intrinsic protective mechanism that includes Edn2 signaling to support photoreceptor cell survival and to preserve vision in the injured retina. 19105399_LIF was also involved into the inhibition of spontaneous cell differentiation and apoptotic cell death; it also decreased the rations of S/G2+M cell cycle and doubling-time of cell population. 19267953_The results show that endogenous LIF is required for normal development of hippocampal astrocytes, and this process is regulated by spontaneous neural impulse activity through the release of ATP. 19342884_LIF and IL-6 behave as polar opposites in promoting commitment to the Treg and Th17 lineages. 19541590_The role of LIF in murine embryonic stem cells was explored using transcriptome analysis of different categories of LIF targets. Review. 19571885_two LIF signalling pathways are each connected to the core circuitry via different transcription factors 19598242_During normal development of mouse optic nerve, there is a developmental time window when LIF is required for correct myelination. Myelination recovers by postnatal day 14, so LIF isn't needed for completion of myelination during postnatal development. 19635738_Blockade of LIF signalling results in a 50% decrease in embryo implantation. 19646521_Data show that leukemia inhibitory factor and bone morphogenetic protein 2 synergistically promote astrocyte differentiation of retinal precursor cells isolated from P0 mouse retina. 19693290_A lack of LIF reduced survival of photoreceptor cells after light exposure. 19795791_the recombinant protein LIF induces jumps of current in bilayer lipid membranes, which indicates the formation of ionic channels. 19906980_These data confirm a key role for CNTF in directly mediating the neuroprotective and axon regenerative effects of inflammatory stimulation in the eye and identify LIF as an additional contributing factor. 20091786_Our results demonstrate that LIF signaling via the gp130/JAK/STAT3 pathway is required for the initiation of the astrogliosis-like reaction of retinal Muller cells after optic nerve injury. 20345762_despite its dependence on LIF, Janus kinase 3 is not essential for LIF-mediated photoreceptor protection or gene expression 20564184_data suggest that TNFalpha may contribute to mammary gland involution by, among other activities, eliciting LIF expression through ERK1/2 and AP1 activation 20598020_OSM and LIF use different mechanisms to achieve neuroprotection and neuromodulation. 20864644_These data demonstrate that epithelial Fut2 mRNA expression is regulated by factors including LIF and IL1B secreted from macrophages recruited during the inflammatory response to insemination. 20962578_Data demonstrate that T cells retain responsiveness to a LIF/Nanog axis and that this axis is linked to MARCH-7, a RINGv E3 ligase that regulates the LIF-receptor. 20974049_These results suggest that leukaemia inhibitory factor- activated STAT3 is responsible for the inhibition of cardiomyogenesis in embryonic stem cells. 21035850_low dose aspirin alters the expression of endometrial LIF and integrin beta3 21254450_These preliminary studies reveal altered social and repetitive behavior in these mice, which suggests that the integrin beta3 subunit may be involved in brain systems relevant to ASD. 21385842_YAP, TEAD2 and Yes are highly expressed in self-renewing embryonic stem cells, and are activated by LIF. 21402718_Data show that p53 and estrogen receptor alpha were activated in endometrial tissues during implantation to coordinately regulate LIF production. 21425410_Pramel7 mediates LIF/STAT3-dependent self-renewal in embryonic stem cells. 21613225_Extensive mannose phosphorylation on leukemia inhibitory factor (LIF) controls its extracellular levels by multiple mechanisms. 21681859_Ccl2 cooperatively activates the Stat3-pathway with leukemia inhibitory factor in feeder-free conditions to maintain stem cell pluripotency 21785422_Brg1 is required to establish chromatin accessibility at STAT3 binding targets, preparing these sites to respond to LIF signalling. 21835648_inhibits T helper 17 cell differentiation and confers treatment effects of neural progenitor cell therapy in autoimmune disease 21930391_Liver sinusoidal endothelial cells constitutively express LIF at the mRNA and protein level. 21966484_Study demonstrated that LIF was an important regulator of decidualization in humans and mice. 22074622_LIF removal results in the rapid phosphorylation of multiple proteins regulating murine embryonic stem cell self-renewal on Tyr, Ser, & Thr residues. These posttranslational modifications help drive the exit of mESCs from the pluripotent state. 22143976_LIF controls vascular size and osteoclast differentiation during the transition of cartilage to bone, whereas an anatomically separate LIF-dependent pathway regulates osteoblast and adipocyte commitment in bone remodeling 22210859_Aute short-term depletion of tet1 results in impairment of LIF-dependent Stat3-mediated gene activation. 22285730_LIF expression might be regulated by microRNA-199a 22581855_The LIF-STAT3 axis is identified in this study as a critical determinant of lung injury with clinical implications for pneumonia patients. 22774981_These results provide further evidence for the role of LIF in inflammation and cartilage bone resorption and provide impetus to test the effects of LIF blockade as a therapeutic strategy in rheumatoid arthritis. 22894638_The rapid release of leukemia inhibitory factor from cortical neurons protects them against excitotoxicity. 22905257_Acyloxy nitroso compounds inhibit LIF signaling in endothelial cells and cardiac myocytes, showing that STAT3 signaling is redox-sensitive 23077604_These studies demonstrate that endogenous CNTF and LIF act centrally to protect axons from acute inflammatory destruction via an oligodendrocyte-independent mechanism. 23258129_we show that LIF increases the expression of the core transcription factors 23409146_LIF was able to modulate the levels of nitric oxide induced by LPS in vitro, suggesting that it could be a potential mediator of the inflammatory action of progesterone 23628590_A pivotal role of PPARgamma on mouse embryonic stem cell self-renewal depends on the presence and absence of LIF. 23942233_Tfcp2l1 is the principal bridge between LIF/Stat3 input and the transcription factor core of naive pluripotency. 23963683_Results show that endogenous levels of Pkig reciprocally regulate osteoblast and adipocyte differentiation and that this reciprocal regulation is mediated in part by LIF. 24008729_Expression of Lif in the injured retina and activation of the endogenous survival pathway involve signaling through p38 MAPK. 24169527_Induced pluripotent stem cells-derived neural precursors exert a neuroprotective role in immune-mediated demyelination via the secretion of LIF. 24310780_Addition of leukemia inhibitory factor (LIF) neutralizing antibodies inhibited oligodendrocyte differentiation, indicating a crucial role of TNFR2-induced astrocyte derived LIF for oligodendrocyte maturation 24464633_LIF suppresses osteoblast differentiation through the LIF/STAT3/SOCS3 signaling pathway. 24664722_This mini review will summarize the findings that are related to LIF signaling and discuss the neuroprotective effects of LIF in different animal models. 24698551_Data show that the contribution of leukemia inhibitory factor (LIF) for embryo implantation was dependent on the strains of mice. 24877624_Uterine kisspeptin signaling regulates glandular Lif levels, regulating embryo implantation 25012664_study presents for the first time that estrogen regulates Egr1 expression through LIF-STAT3 signaling pathway in mouse uterus, and Egr1 functions as a critical mediator of stromal cell decidualization by regulating Wnt4 25031358_Data suggest LIF induces dynamic/complex network of changes in endometrium, changes that appear essential to reproduction; changes involve gene expression regulation for cytoskeletal proteins and proteins involved in chromatin assembly/disassembly. 25147827_LIF and miR-302 family positively affect the expression of pluripotency markers. 25277705_Expression of LIF protects the lung from lung injury and enhanced pathology during respiratory syncytial virus infection. 25341493_prenatal immunological stress delays the GnRH neuron migration in the nasal compartment of mouse fetuses, which may be mediated by the regulation of IL-6, MCP-1 and LIF secretion in the maternal-fetal system. 25514345_In a preclinical animal model of MS shifting the LIF/IL-6 balance in favor of LIF by CNS-targeted overexpression increased the number of Tregs in the CNS during active autoimmune responses and reduced disease symptoms. 25564647_The difference in the balance between the JAK-Stat3 pathway and the MAP kinase pathway underlies the differential response to LIF amongst mice strains. 25635127_these results reveal a novel function of miR-181 in embryo implantation through the regulation of LIF, and also indicate a potential link between miR-181 dysregulation and human embryo implantation defects. 25639936_Study shows that LIF-treated postmitotic oligodendrocytes secrete a number of neuropeptides at the mRNA and peptide level, and confirm LIF-induced galanin synthesis and secretion in primary rat oligodendrocytes but not in astrocytes 25887270_The role of LIF in acetylcholine synthesis via the STAT3, PI3 kinases, or cAMP-protein kinase A pathways, are reported. 26043040_These findings suggest that maternal IL-6 interferes the maternal-fetal LIF signal relay by inducing SOCS3 in the placenta and leads to decreased neurogenesis. 26092726_The data support an important role of LIF-JAK2-STAT3 regulation in the development of colonic adenocarcinoma-related cachexia. 26187859_Despite common signal transduction mechanisms (JAK/STAT, MAPK and PI3K) LIF can have paradoxically opposite effects in different cell types including stimulating or inhibiting each of cell proliferation, differentiation and survival. 26272398_Our data suggest that LIF plays an important role in placentation in vivo and the maintenance of healthy pregnancy. 26303070_These results establish that Bcl3 positively regulates pluripotency genes and thus shed light on the mechanism of Bcl3 as a downstream molecule of LIF/STAT3 signaling in pluripotency maintenance. 26321142_Mechanistically, LIF activates STAT, which binds to a Stat consensus sequence in the Six2 proximal promoter and sustains SIX2 levels. 26395492_LIF has a role in primitive endoderm expansion during pre-implantation development 26427868_LIF gene breakage was captured at the 18th hour after carbon disulfide exposure by Comet-FISH and the protein and mRNA of LIF in uterine tissue were down-regulated after carbon disulfide exposure through the peri-implantation period 26865369_mouse embryonic stem cells inside microchambers formed colonies and expressed markers of pluripotency in the absence of feeders or pluripotency-inducing signals such as leukemia inhibitory factor. 26995887_Studied the expression of heme oxygenase-1 (HO-1) and leukemia inhibitory factor (LIF) in maternal plasma and placental tissue in intrauterine infection-induced preterm birth. 27096934_Either LIF or EGF is needed during development of pre-implantation embryo. 27227407_LIF stimulates, in part, stem cell-derived cardiomyocyte regeneration by activating cardiac stem or precursor cells. 27676194_LIF/STAT3 and MEK/ERK signaling in naive embryonic stem cell pluripotency, is reported. 28122728_Our data show that absence of SOCS2 turns cardiomyocytes unresponsive to LIF-induced [Ca(2+)] raise, indicating that endogenous levels of SOCS2 are crucial for full activation of LIF signaling in the heart. 28522567_lung epithelium, specifically alveolar type II cells, is the predominant site of LIF transcript induction in pneumonic mouse lungs. 28865476_Inhibition of Lif protein in the hypothalamus transforms obesity-resistant mice into obesity-prone mice. 29222114_blood-derived vitronectin (VTN) rapidly and potently activates leukemia inhibitory factor (LIF) and pro-inflammatory interleukin 6 (IL-6) in vitro and after vascular injury in the brain. 29391217_The amplitude of inward currents in Muller cells from the postischemic retina was reduced to 51% in wild type and to 70% in LIF(-/-) mice. This demonstrates that decrease of inward currents takes place in reactive Muller cells even in the absence of LIF. 29721979_An existing single Muller cell microarray dataset was used to determine which processes are upregulated in Muller cells that express LIF, by correlating LIF expression to the expression of other genes using a robust correlation method. Some enriched processes include divalent inorganic cation homeostasis, negative regulation of stem cell proliferation, and gamma-glutamyl transferase activity. 30270531_The results provide direct evidence for LIF as a major cachexia initiating factor for the C26 tumour in vivo. Tumour-derived LIF was also a regulator of multiple cytokines in C26 tumour cells and in C26 tumour-bearing mice. 30996350_findings reveal a function of LIF in pancreatic ductal adenocarcinoma tumorigenesis, and suggest its translational potential as an attractive therapeutic target and circulating marker; studies underscore how a better understanding of cell-cell communication within the tumour microenvironment can suggest novel strategies for cancer therapy 31142168_results can reflect a role for LIF in the generation and particularly maturation of DCs. It can be assumed that LIF rather modulates the maturation level, leading to the development of semi-mature and tolerogenic DCs. 31740000_Leukemia inhibitory factor induces corticotropin-releasing hormone in mouse trophoblast stem cells. 32169845_Leukemia Inhibitory Factor Inhibits Plasmacytoid Dendritic Cell Function and Development. 32651426_Immunization against leukemia inhibitory factor and its receptor suppresses tumor formation of breast cancer initiating cells in BALB/c mouse. 32719388_LIF is essential for ISC function and protects against radiation-induced gastrointestinal syndrome. 33846773_Changes in the phosphorylation of nucleotide metabolismassociated proteins by leukemia inhibitory factor in mouse embryonic stem cells. 34392367_Myeloid cell-mediated targeting of LIF to dystrophic muscle causes transient increases in muscle fiber lesions by disrupting the recruitment and dispersion of macrophages in muscle. 35078447_Osteoclasts secrete leukemia inhibitory factor to promote abnormal bone remodeling of subchondral bone in osteoarthritis. 35981499_Leukemia inhibitory factor protects against graft-versus-host disease while preserving graft-versus-leukemia activity. |
ENSG00000128342 |
LIF |
87.074298 |
0.1564376038 |
-2.676341 |
0.51715746 |
23.471608 |
0.00000126770838221985022826955764874146126430787262506783008575439453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000110975483206740002054685456656102360284421592950820922851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
23.1761809909512 |
8.48694942626611 |
148.149680304576 |
39.6632225208945 |
| ENSMUSG00000034706 |
432611 |
Dnai2 |
protein_coding |
A2AC93 |
FUNCTION: Part of the dynein complex of respiratory cilia. {ECO:0000250}. |
Alternative splicing;Cell projection;Cilium;Cilium biogenesis/degradation;Cytoplasm;Cytoskeleton;Dynein;Microtubule;Motor protein;Reference proteome;Repeat;WD repeat |
|
|
mmu:432611; |
9+2 motile cilium [GO:0097729]; axonemal dynein complex [GO:0005858]; axoneme [GO:0005930]; cilium [GO:0005929]; cytoplasm [GO:0005737]; dynein axonemal particle [GO:0120293]; external side of plasma membrane [GO:0009897]; microtubule [GO:0005874]; outer dynein arm [GO:0036157]; sperm flagellum [GO:0036126]; dynein heavy chain binding [GO:0045504]; dynein light chain binding [GO:0045503]; microtubule motor activity [GO:0003777]; cilium assembly [GO:0060271]; cilium movement [GO:0003341]; determination of left/right symmetry [GO:0007368]; microtubule-based movement [GO:0007018]; outer dynein arm assembly [GO:0036158] |
18547164_These data suggest that Dnaic2 plays a role in ovarian follicular development. 27823762_Results obtained with Western blot analysis revealed that the expression of DNAI2 protein was significantly less in oviducts of dNOD mice as compared to that of cNOD mice corroborating the results obtained with immunohistochemistry 30387321_Dnaic2 plays a role in oogenesis and spermatogenesis by activation of Stat3. |
ENSG00000171595 |
DNAI2 |
134.162727 |
2.4261971513 |
1.278697 |
0.26676113 |
22.104595 |
0.00000258190936818953579803023572203368729560679639689624309539794921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000213505122129549464399331931696579545132408384233713150024414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
189.324586812409 |
35.2068326572916 |
78.0334717284296 |
11.1834466394058 |
| ENSMUSG00000034773 |
217216 |
Hrob |
protein_coding |
Q32P12 |
FUNCTION: DNA-binding protein involved in homologous recombination that acts by recruiting the MCM8-MCM9 helicase complex to sites of DNA damage to promote DNA repair synthesis. {ECO:0000250|UniProtKB:Q8N3J3}. |
Alternative splicing;Chromosome;DNA damage;DNA recombination;DNA repair;DNA synthesis;DNA-binding;Methylation;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:217216; |
nucleus [GO:0005634]; site of DNA damage [GO:0090734]; single-stranded DNA binding [GO:0003697]; cellular response to DNA damage stimulus [GO:0006974]; DNA synthesis involved in DNA repair [GO:0000731]; female gamete generation [GO:0007292]; interstrand cross-link repair [GO:0036297]; male gamete generation [GO:0048232]; recombinational repair [GO:0000725] |
34707299_Meiotic genes in premature ovarian insufficiency: variants in HROB and REC8 as likely genetic causes. |
ENSG00000125319 |
HROB |
222.044200 |
2.0667237446 |
1.047346 |
0.17576135 |
35.260050 |
0.00000000288489502900681894461846149031389319183915631583658978343009948730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000380403416435047750519684130795367016375507773773279041051864624023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
287.570572393813 |
35.121015705733 |
139.143208252245 |
13.101493662099 |
| ENSMUSG00000034845 |
84094 |
Plvap |
protein_coding |
G3X924 |
Human_homologues FUNCTION: Endothelial cell-specific membrane protein involved in the formation of the diaphragms that bridge endothelial fenestrae. It is also required for the formation of stomata of caveolae and transendothelial channels. Functions in microvascular permeability, endothelial fenestrae contributing to the passage of water and solutes and regulating transcellular versus paracellular flow in different organs. Plays a specific role in embryonic development. {ECO:0000250|UniProtKB:Q91VC4}. |
Coiled coil;Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:84094; |
caveola [GO:0005901]; cell surface [GO:0009986]; identical protein binding [GO:0042802]; MAPK cascade [GO:0000165]; positive regulation of cellular extravasation [GO:0002693]; tumor necrosis factor-mediated signaling pathway [GO:0033209] |
16969073_The PV-1 protein is negatively regulated in pulmonary endothelial cells by VEGF-R2 signaling. 16969078_Compelling evidence for a functional role of PV-1 in the formation of large transendothelial channels and modulation of nuclear shape. 17075074_PV-1 is necessary for fenestral pore architecture and the ordered arrangement of fenestrae in sieve plates. 20590523_upregulation of microvascular expression of PV-1 post-spinal cord injury may promote major components of secondary injury 22403691_formation of diaphragms is the only role of PV1 22568538_PV1 is involved in the tumor angiogenesis 25541982_Results indicate that PLVAP is not only required for the formation of endothelial diaphragms, but also for the formation of endothelial fenestrae, even of those without diaphragms. 25665101_In the lymphatic sinus, PLVAP forms a physical sieve that regulates the parenchymal entry of lymphocytes and soluble antigens. 27732581_an endothelium-specific molecule, plasmalemma vesicle-associated protein (PLVAP), regulates the seeding of fetal monocyte-derived macrophages to tissues in mice 27742829_These results show that Plvap expression in endothelial cells is important in the maintenance of IgM(+) B cells in the spleen and peritoneal cavity. 31666588_Fenestral diaphragms and PLVAP associations in liver sinusoidal endothelial cells are developmentally regulated. 31759628_Retinal VEGFA maintains the ultrastructure and function of choriocapillaris by preserving the endothelial PLVAP. 32663411_PV1 in Caveolae Controls Lung Endothelial Permeability. 34769404_A Potential New Role for Zinc in Age-Related Macular Degeneration through Regulation of Endothelial Fenestration. |
ENSG00000130300 |
PLVAP |
85.151562 |
0.2986808902 |
-1.743323 |
0.30241712 |
32.364879 |
0.00000001277750980140106652479790215947916265371020472230156883597373962402343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001514607231867245584134082500224471878880194708472117781639099121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.248770407445 |
8.28769500741919 |
131.407035730289 |
19.3691840152943 |
| ENSMUSG00000034903 |
319876 |
Cobll1 |
protein_coding |
Q3UMF0 |
|
Alternative splicing;Phosphoprotein;Reference proteome |
|
|
mmu:319876; |
actin monomer binding [GO:0003785] |
Human_homologues 19401544_The gene ratio test with the COBLL1/ARHGDIA genes for survival of patients with malignant pleural mesothelioma has robust predictive value. 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20468071_Observational study of gene-disease association. (HuGE Navigator) 21569526_We provide evidence that ARHGDIA, COBLL1, and TM4SF1 are negative regulators of apoptosis in cultured tumor cells. 23463496_genetic association study in children/adolescents in Sardinia: Data suggest that an SNP in COBLL1 (rs7607980; TC or CC) is associated with lower insulin resistance and lower serum insulin levels in overweight/obese children/adolescents. 26757982_Aggregation of rare variants in COBLL1were nominally associated with higher waist-to-hip ratio in European-Americans. 27185377_The expression of COBLL1, LPL, and ZAP70 corresponded to patient prognosis and to IGHV mutational status, although not absolutely. When we combined all three markers together and performed the ROC analysis, AUC increased compared to the AUC of individual gene expression. 28232743_High-level expression of Cobll1 promotes the transformation of CML and generates drug resistance by increasing IKKgamma stability, which results in activation of the canonical NF-kappaB pathway. 28258026_polymorphism, rs7607980 of COBLL1 didnot show any significant association with T2D 29122990_identified COBLL1 as a novel interaction partner of ROR1 29686105_nuclear COBLL1 interacts with AR to enhance complex formation with CDK1 and facilitates AR phosphorylation for genomic binding in castration-resistant prostate cancer model cells. 35352878_Reciprocal interactions among Cobll1, PACSIN2, and SH3BP1 regulate drug resistance in chronic myeloid leukemia. 35955692_Metabolic Effects of the Waist-To-Hip Ratio Associated Locus GRB14/COBLL1 Are Related to GRB14 Expression in Adipose Tissue. 36493769_Genome-wide analysis of copy-number variation in humans with cleft lip and/or cleft palate identifies COBLL1, RIC1, and ARHGEF38 as clefting genes. |
ENSG00000082438 |
COBLL1 |
91.736745 |
0.4069262588 |
-1.297161 |
0.37234093 |
11.740171 |
0.00061165402154205524443836283765563166525680571794509887695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0029880190490395417031443958677527916734106838703155517578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
54.3191847746001 |
14.1752306624192 |
133.486555858688 |
25.0989128469394 |
| ENSMUSG00000034959 |
271221 |
Rubcnl |
protein_coding |
F8WGX2 |
Human_homologues FUNCTION: Regulator of autophagy that promotes autophagosome maturation by facilitating the biogenesis of phosphatidylinositol 3-phosphate (PtdIns(3)P) in late steps of autophagy (PubMed:28306502, PubMed:30704899). Acts by antagonizing RUBCN, thereby stimulating phosphatidylinositol 3-kinase activity of the PI3K/PI3KC3 complex (PubMed:28306502). Following anchorage to the autophagosomal SNARE STX17, promotes the recruitment of PI3K/PI3KC3 and HOPS complexes to the autophagosome to regulate the fusion specificity of autophagosomes with late endosomes/lysosomes (PubMed:28306502). Binds phosphoinositides phosphatidylinositol 3-phosphate (PtdIns(3)P), 4-phosphate (PtdIns(4)P) and 5-phosphate (PtdIns(5)P) (PubMed:28306502). In addition to its role in autophagy, acts as a regulator of lipid and glycogen homeostasis (By similarity). May act as a tumor suppressor (Probable). {ECO:0000250|UniProtKB:Q3TD16, ECO:0000269|PubMed:28306502, ECO:0000269|PubMed:30704899, ECO:0000305|PubMed:23522960}. |
Proteomics identification;Reference proteome |
|
|
Human_homologues hsa:80183; |
autophagosome membrane [GO:0000421]; phosphatidylinositol-3-phosphate binding [GO:0032266]; phosphatidylinositol-4-phosphate binding [GO:0070273]; phosphatidylinositol-5-phosphate binding [GO:0010314]; autophagosome maturation [GO:0097352]; autophagosome-endosome fusion [GO:0061910]; autophagosome-lysosome fusion [GO:0061909] |
30704899_mTORC1 and GSK3-TIP60 signaling converge to modulate autophagosome maturation and lipid metabolism through Pacer. |
ENSG00000102445 |
RUBCNL |
97.401481 |
3.4947886875 |
1.805205 |
0.51376489 |
11.547169 |
0.00067852512934842826222009426473391613399144262075424194335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0032675607873016948405620052398035113583318889141082763671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
149.214048420089 |
54.2675815444345 |
42.6961575546888 |
11.7506781390684 |
| ENSMUSG00000035095 |
219148 |
Fam167a |
protein_coding |
Q6P1G6 |
|
Coiled coil;Reference proteome |
|
|
mmu:219148; |
|
35241148_FAM167A is a key molecule to induce BCR-ABL-independent TKI resistance in CML via noncanonical NF-kappaB signaling activation. |
ENSG00000154319 |
FAM167A |
237.758355 |
3.1496996958 |
1.655214 |
0.20190000 |
65.368805 |
0.00000000000000062114385502909887765868117112840718379139655878906634001168640679679811000823974609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000171089750448521421165196188332209228881290209722809692038936191238462924957275390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
357.889057396108 |
50.2489386593111 |
113.626406313511 |
12.4619601690585 |
| ENSMUSG00000035104 |
232146 |
Eva1a |
protein_coding |
Q91WM6 |
FUNCTION: Acts as a regulator of programmed cell death, mediating both autophagy and apoptosis. {ECO:0000250}. |
Apoptosis;Autophagy;Endoplasmic reticulum;Lysosome;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:232146; |
endoplasmic reticulum membrane [GO:0005789]; intracellular membrane-bounded organelle [GO:0043231]; lysosomal membrane [GO:0005765]; plasma membrane [GO:0005886]; apoptotic process [GO:0006915]; autophagy [GO:0006914] |
26905199_EVA1A deletion results in defective self-renewal and differentiation of NSCs. EVA1A modulates autophagy through the PIK3CA/AKT-mTOR pathway. 28044434_ATP synthesis, oxidative phosphorylation, and the TCA cycle, are involved in the EVA1A regulatory network.EVA1A plays an important role in neuronal differentiation. 28151473_Eva1a improves cardiac function. 31977009_Eva1a ameliorates atherosclerosis by promoting re-endothelialization of injured arteries via Rac1/Cdc42/Arpc1b. |
ENSG00000115363 |
EVA1A |
14.279075 |
26.2663895992 |
4.715146 |
1.09160912 |
22.625979 |
0.00000196805389127755573315851043980639900610185577534139156341552734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000165987801062789210114045007626870642525318544358015060424804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
29.2890278392274 |
24.3373995215655 |
1.11507627375217 |
0.818206684219466 |
| ENSMUSG00000035177 |
232827 |
Nlrp2 |
protein_coding |
A0A140LHK4 |
Human_homologues FUNCTION: Suppresses TNF- and CD40-induced NFKB1 activity at the level of the IKK complex, by inhibiting NFKBIA degradation induced by TNF. When associated with PYCARD, activates CASP1, leading to the secretion of mature pro-inflammatory cytokine IL1B. May be a component of the inflammasome, a protein complex which also includes PYCARD, CARD8 and CASP1 and whose function would be the activation of pro-inflammatory caspases. {ECO:0000269|PubMed:15456791}. |
Reference proteome |
|
|
|
chromosome, telomeric region [GO:0000781]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; aspartic-type endopeptidase inhibitor activity [GO:0019828]; caspase binding [GO:0089720]; identical protein binding [GO:0042802]; interleukin-1 binding [GO:0019966]; Pyrin domain binding [GO:0032090]; regulation of inflammatory response [GO:0050727] |
21976665_POP2 acts as a regulator of inflammatory signals and exerts its two known functions through distinct modalities employed by its first alpha-helix 22295082_Nlrp2 is a member of the mammalian maternal effect genes and required for early embryonic development in the mouse. 27693696_high expression of NLRP2, especially in astrocytes, may play important roles in the pathophysiological process of ischemic stroke and has potential clinical value for the treatment of ischemic stroke. 27881734_This work identifies Nlrp2 as a critical regulator of oocyte quality 28317850_female mice lacking NLRP2 are subfertile because of early embryonic loss and the production of fewer offspring that have a wide array of developmental phenotypes and abnormal DNA methylation at imprinted loci. 28630100_NLRP2 interacts with FAF1 under normal physiological conditions 30172704_Study shownd that apoptosis signal-regulating kinase 1 (ASK1) silencing and ASK1 inhibition reduced ischemic injury-induced nucleotide-binding oligomerization domain-like receptor pyrin domain-containing 2 inflammasomes in the brain and astrocyte cell line, respectively. 30454891_NLRP2 might be a potential target for developing effective therapeutic strategy to prevent Nonalcoholic fatty liver disease progression. 31162334_Activation of the NLRP2 inflammasome leads to activation of dorsal root ganglion neurons and subsequent development of pain hypersensitivity. 33090377_Reproductive Outcomes from Maternal Loss of Nlrp2 Are Not Improved by IVF or Embryo Transfer Consistent with Oocyte-Specific Defect. |
ENSG00000022556 |
NLRP2 |
73.203033 |
0.0040814382 |
-7.936707 |
0.95192467 |
123.602394 |
0.00000000000000000000000000010293489815028948706478262307627564456100887139902801208104207737388647951864290019940995080105494707822799682617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000071922218744275657830135857681647464352026005492159277894104074136809616121790433851401758147403597831726074218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.56586343845777 |
0.401058340181916 |
138.643145938117 |
33.2721768694072 |
| ENSMUSG00000035208 |
276950 |
Slfn8 |
protein_coding |
B1ARD8 |
FUNCTION: Endoribonuclease that cleaves tRNAs and rRNAs (PubMed:29563550). Cleaves tRNAs 11 nucleotides from the 3'-terminus at the acceptor stem (PubMed:29563550). May be involved in immune system via regulation of inflammation (PubMed:29528433). {ECO:0000269|PubMed:29528433, ECO:0000269|PubMed:29563550}. |
ATP-binding;Cytoplasm;Endonuclease;Hydrolase;Immunity;Magnesium;Metal-binding;Nuclease;Nucleotide-binding;Reference proteome;Zinc |
|
|
mmu:276950; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; site of DNA damage [GO:0090734]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; endoribonuclease activity [GO:0004521]; tRNA binding [GO:0000049]; zinc ion binding [GO:0008270]; defense response to virus [GO:0051607]; immune system process [GO:0002376]; rRNA catabolic process [GO:0016075]; tRNA catabolic process [GO:0016078] |
15351786_functional participation of slfn8 in the regulatory networks governing T cell development and growth appears to be cell type specific 24089532_several mouse Slfn genes are up-regulated in response to IFN treatment of mouse melanoma and renal cell carcinoma cells, including Slfn1, Slfn2, Slfn4, Slfn5, and Slfn8. 29528433_We demonstrated that Slfn8-/- mice were resistant to the development of EAE. |
ENSG00000154760 |
SLFN13 |
30.668700 |
2.2293907021 |
1.156649 |
0.46801033 |
6.077511 |
0.01369135584153265486340167456091876374557614326477050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0438491867489960926440595301301073050126433372497558593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.3577382744719 |
12.9927679306448 |
17.6540335605107 |
4.48385036532256 |
| ENSMUSG00000035237 |
16816 |
Lcat |
protein_coding |
P16301 |
FUNCTION: Central enzyme in the extracellular metabolism of plasma lipoproteins. Synthesized mainly in the liver and secreted into plasma where it converts cholesterol and phosphatidylcholines (lecithins) to cholesteryl esters and lysophosphatidylcholines on the surface of high and low density lipoproteins (HDLs and LDLs) (PubMed:19065001). The cholesterol ester is then transported back to the liver. Also produced in the brain by primary astrocytes, and esterifies free cholesterol on nascent APOE-containing lipoproteins secreted from glia and influences cerebral spinal fluid (CSF) APOE- and APOA1 levels (PubMed:19065001). Together with APOE and the cholesterol transporter ABCA1, plays a key role in the maturation of glial-derived, nascent lipoproteins (PubMed:19065001). Required for remodeling high-density lipoprotein particles into their spherical forms (PubMed:19065001). Has a preference for plasma 16:0-18:2 or 18:O-18:2 phosphatidylcholines (PubMed:8820107). Catalyzes the hydrolysis of 1-O-alkyl-2-acetyl-sn-glycero-3-phosphocholine (platelet-activating factor or PAF) to 1-O-alkyl-sn-glycero-3-phosphocholine (lyso-PAF) (PubMed:10393212). Also catalyzes the transfer of the acetate group from PAF to 1-hexadecanoyl-sn-glycero-3-phosphocholine forming lyso-PAF (By similarity). Catalyzes the esterification of (24S)-hydroxycholesterol (24(S)OH-C), also known as cerebrosterol to produce 24(S)OH-C monoesters (By similarity). {ECO:0000250|UniProtKB:P04180, ECO:0000269|PubMed:11809774, ECO:0000269|PubMed:11893779, ECO:0000269|PubMed:15654758, ECO:0000269|PubMed:19065001, ECO:0000269|PubMed:8820107}. |
Acyltransferase;Cholesterol metabolism;Disulfide bond;Glycoprotein;Hydrolase;Lipid metabolism;Reference proteome;Secreted;Signal;Steroid metabolism;Sterol metabolism;Transferase |
|
|
mmu:16816; |
extracellular space [GO:0005615]; high-density lipoprotein particle [GO:0034364]; 1-alkyl-2-acetylglycerophosphocholine esterase activity [GO:0003847]; apolipoprotein A-I binding [GO:0034186]; phosphatidylcholine-sterol O-acyltransferase activity [GO:0004607]; phospholipase A2 activity [GO:0004623]; platelet-activating factor acetyltransferase activity [GO:0047179]; sterol esterase activity [GO:0004771]; cholesterol homeostasis [GO:0042632]; cholesterol metabolic process [GO:0008203]; cholesterol transport [GO:0030301]; high-density lipoprotein particle remodeling [GO:0034375]; lipid metabolic process [GO:0006629]; lipoprotein biosynthetic process [GO:0042158]; lipoprotein metabolic process [GO:0042157]; phosphatidylcholine biosynthetic process [GO:0006656]; phosphatidylcholine metabolic process [GO:0046470]; phospholipid metabolic process [GO:0006644]; regulation of high-density lipoprotein particle assembly [GO:0090107]; response to copper ion [GO:0046688]; response to glucocorticoid [GO:0051384]; reverse cholesterol transport [GO:0043691]; very-low-density lipoprotein particle remodeling [GO:0034372] |
11809774_Oxidative stress is markedly elevated in lecithin:cholesterol acyltransferase-deficient mice and is paradoxically reversed in the apolipoprotein E knockout background in association with a reduction in atherosclerosis 11893779_LCAT contributes significantly to the cholesteryl ester (CE) fatty acid pool of apoB lipoprotein and is the only source of plasma long chain polyunsaturated CE in LDL receptor and apoE knockout mice. 14668345_hypertriglyceridemia and the improved fasting glucose phenotype seen in the lecithin-cholesterol acyltransferase and ldl receptor double knockout mice are in part attributable to up-regulation of hepatic sterol regulatory element binding factor-1c gene 15251433_LCAT has a role in regulating plasma HDL cholesterol levels and biliary cholesterol excretion 15654758_apoE is a more significant activator of LCAT than apoA-I on mouse apoB lipoproteins 15805543_ACAT2 provides core CE of newly secreted VLDL, whereas LCAT adds CE during LDL particle formation 16105858_In LCAT deficient mice, alterations in hepatic expression of the gene for fads2, soat2, and dgat2, results in changes in total hepatic and cholesterol ester-associated polyunsaturated fatty acids. 17551001_LCAT deficiency induces complex alterations in hepatic signal transduction cascades. 19065001_These data show that brain LCAT esterifies cholesterol on glial-derived apoE-lipoproteins, and influences CSF apoE and apoA-I levels. 19564558_LCAT overexpression does not promote an increased rate of macrophage reverse cholesterol transport. 19729076_Cholesterol homeostasis was examined in mice with the two major cholesterol transport pathways for catabolism interrupted by disrupting abca1, lcat, or both. 21454561_LCAT deficiency confers gender-specific protection against diet-induced obesity and insulin resistance at least in part through regulation in UPR, white adipose tissue adipogenesis, and brown adipocyte partitioning 22326749_Studies suggest that absence of lecithin cholesterol acyltransferase (LCAT) may protect against insulin resistance, diabetes and obesity. 22370449_Data show that LCAT activity was significantly higher in long chain base biosynthesis protein 2 (Sptlc2)+/- and sphingomyelin synthase 2 (Sms2)-/- mice, but markedly lower in ApoE-/- and Ldlr-/- mice. 22500017_Lecithin:cholesterol acyltransferase deficiency protects against cholesterol-induced hepatic endoplasmic reticulum stress in mice. 23132909_a novel function of apoA-IV in the biogenesis of discrete HDL-A-IV particles with the participation of ABCA1 and LCAT 23178225_adrenal glucocorticoid function in LCAT knockout (KO) mice 23920041_DYRK1A overexpression decreases plasma lecithin:cholesterol acyltransferase activity and apolipoprotein A-I levels. 24950691_Taken together, these results suggest that apoE-containing discoidal HDLs do not require LCAT-dependent maturation to mediate efficient Abeta clearance 25948084_Gene transfer of WT LCAT in LCAT(-/-) mice increased 11.8-fold the plasma cholesterol, whereas the LCAT[T147I] and LCAT[P274S] mutants caused a 5.2- and 2.9-fold increase, respectively. 25964513_increased cholesterol esterification by LCAT is atheroprotective 29369831_metabolic phenotypes in lecithin cholesterol acyltyransferase-deficient mice 30742993_It has been reported that universal deletion of Apoa1 or Lcat expression in mice fed western-type diet results in increased sensitivity to body-weight gain compared to control C57BL/6 group. 32777483_Esterification of side-chain oxysterols by lysosomal phospholipase A2. 33020907_Apolipoprotein A-I in mouse cerebrospinal fluid derives from the liver and intestine via plasma high-density lipoproteins assembled by ABCA1 and LCAT. 34321507_High density lipoproteins mediate in vivo protection against staphylococcal phenol-soluble modulins. |
ENSG00000213398 |
LCAT |
71.196522 |
0.4854555659 |
-1.042589 |
0.29554771 |
12.421233 |
0.00042447984636200295589444908550547097547678276896476745605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0021682350837645973824774348059918338549323379993438720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.103866483728 |
9.71724239374822 |
97.0302326200214 |
14.3408652428421 |
| ENSMUSG00000035258 |
320712 |
Abi3bp |
protein_coding |
A0A338P6S8 |
|
Proteomics identification;Reference proteome;Signal |
|
|
|
collagen-containing extracellular matrix [GO:0062023]; dendritic shaft [GO:0043198]; dendritic spine [GO:0043197]; extracellular matrix [GO:0031012]; interstitial matrix [GO:0005614]; lamellipodium [GO:0030027]; postsynaptic density [GO:0014069]; actin filament binding [GO:0051015]; collagen binding [GO:0005518]; glycosaminoglycan binding [GO:0005539]; heparin binding [GO:0008201]; extracellular matrix organization [GO:0030198]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of connective tissue replacement involved in inflammatory response wound healing [GO:1904597]; positive regulation of cardiocyte differentiation [GO:1905209]; positive regulation of cell-substrate adhesion [GO:0010811]; regulation of actin cytoskeleton reorganization [GO:2000249]; regulation of dendritic spine morphogenesis [GO:0061001]; regulation of postsynaptic density assembly [GO:0099151] |
17101133_NESH (Abi-3), like Abi-1 and Abi-2, is a component of the Abi/WAVE complex, but likely plays a different role in the regulation of c-Abl. 19994723_a p53-dependent apoptosis function of the mouse TARSH gene. 23666637_Abi3bp knockout increased MSC number and proliferation in bone marrow, lung, and liver. 25296984_Identified Abi3bp as a protein important for cardiac progenitor cell differentiation and proliferation. 27521794_Constructed an Abi3bp-deficient mouse model and observed phenotypic changes. The generated Abi3bp-knockout mice are viable and fertile, develop normally and exhibit no significant differences in anxiety or depression-like behaviors, olfactory function and tumor incidence. 28441029_This is the first report of susceptibility to cigarette smoke-induced emphysema in 34 inbred strains of mice, and Abi3bp is identified as a potential contributor to this phenotype. |
ENSG00000154175 |
ABI3BP |
1277.772376 |
0.1668402471 |
-2.583461 |
0.48397961 |
25.392632 |
0.00000046770107557068958084085406531893447379388817353174090385437011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000043867135363871574186785985693948219932281062938272953033447265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
364.053882128287 |
124.432472226383 |
2182.05072514616 |
552.105055329585 |
| ENSMUSG00000035273 |
15442 |
Hpse |
protein_coding |
Q6YGZ1 |
FUNCTION: Endoglycosidase that cleaves heparan sulfate proteoglycans (HSPGs) into heparan sulfate side chains and core proteoglycans. Participates in extracellular matrix (ECM) degradation and remodeling. Selectively cleaves the linkage between a glucuronic acid unit and an N-sulfo glucosamine unit carrying either a 3-O-sulfo or a 6-O-sulfo group. Can also cleave the linkage between a glucuronic acid unit and an N-sulfo glucosamine unit carrying a 2-O-sulfo group, but not linkages between a glucuronic acid unit and a 2-O-sulfated iduronic acid moiety. It is essentially inactive at neutral pH but becomes active under acidic conditions such as during tumor invasion and in inflammatory processes. Facilitates cell migration associated with metastasis, wound healing and inflammation. Enhances shedding of syndecans, and increases endothelial invasion and angiogenesis in myelomas. Acts as procoagulant by increasing the generation of activation factor X in the presence of tissue factor and activation factor VII. Increases cell adhesion to the extracellular matrix (ECM), independent of its enzymatic activity. Induces AKT1/PKB phosphorylation via lipid rafts increasing cell mobility and invasion. Heparin increases this AKT1/PKB activation. Regulates osteogenesis. Enhances angiogenesis through up-regulation of SRC-mediated activation of VEGF. Implicated in hair follicle inner root sheath differentiation and hair homeostasis. {ECO:0000269|PubMed:15677344, ECO:0000269|PubMed:15793281, ECO:0000269|PubMed:17689495, ECO:0000269|PubMed:18589009}. |
Calcium;Cell adhesion;Direct protein sequencing;Disulfide bond;Glycoprotein;Hydrolase;Lysosome;Magnesium;Membrane;Nucleus;Reference proteome;Secreted;Signal |
|
This gene encodes an endoglucuronidase enzyme that plays an important role in tumor invasion and metastasis. The encoded preproprotein undergoes proteolytic processing to generate an active heterodimeric enzyme that cleaves the heparan sulfate proteoglycans associated with the cell surface and extracellular matrix. Mice lacking the encoded protein do not show any prominent pathological alterations under normal conditions but fail to develop albuminuria and renal damage in response to drug-induced diabetes. [provided by RefSeq, Aug 2016]. |
mmu:15442; |
autophagosome [GO:0005776]; endosome [GO:0005768]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; heparanase complex [GO:1904974]; intracellular membrane-bounded organelle [GO:0043231]; lysosomal lumen [GO:0043202]; lysosomal membrane [GO:0005765]; lysosome [GO:0005764]; membrane raft [GO:0045121]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; cargo receptor ligand activity [GO:0140355]; heparanase activity [GO:0030305]; molecular function activator activity [GO:0140677]; syndecan binding [GO:0045545]; angiogenesis involved in wound healing [GO:0060055]; cell-matrix adhesion [GO:0007160]; establishment of endothelial barrier [GO:0061028]; Factor XII activation [GO:0002542]; heparan sulfate proteoglycan catabolic process [GO:0030200]; heparin metabolic process [GO:0030202]; inflammatory response [GO:0006954]; positive regulation of angiogenesis [GO:0045766]; positive regulation of autophagy [GO:0010508]; positive regulation of blood coagulation [GO:0030194]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cell-matrix adhesion [GO:0001954]; positive regulation of extracellular matrix disassembly [GO:0090091]; positive regulation of hair follicle development [GO:0051798]; positive regulation of osteoblast proliferation [GO:0033690]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of substrate adhesion-dependent cell spreading [GO:1900026]; positive regulation of vascular endothelial growth factor production [GO:0010575]; protein transmembrane transport [GO:0071806]; receptor-mediated endocytosis [GO:0006898]; regulation of hair follicle development [GO:0051797]; response to organic substance [GO:0010033]; vascular wound healing [GO:0061042]; wound healing [GO:0042060] |
12460766_A full-length heparanase gene was cloned from a mouse embryo cDNA library and determined to encode a protein of 535 amino acids. The full-length mouse gene was stably expressed in NS0 myeloma cells. 14618334_expression of heparanase mRNA and VEGF, and vessel formation in mouse fractured bone 15645118_HPSE-1 likely plays important roles in regulating the in vivo growth and progression of melanoma 15749484_heparanase is actively involved in embryo implantation. 15799822_heparanase may have a role in lymph node metastatic dissemination 16093249_EGR1 regulates heparanase transcription in tumor cells and importantly, can have a repressive or activating role depending on the tumor type 16384929_By monitoring in vivo activation of luciferase gene driven by the heparanase promoter, we demonstrate activation of heparanase transcription at an early stage of DTH. 17051139_HPSE is involved in the pathogenesis of proteinuria in overt diabetic nephropathy by degradation of heparan sulfates 17213834_study demonstrates that mouse Hpse is expressed and regulated in a similar manner to human HPSE and also provides some novel insights into mechanisms of Hpse gene regulation that are likely to be relevant to control of the human gene 17507691_Heparanase plays a role in blastocyst implantation. 17959718_Heparanase releases FGF10 from perlecan in the basement membrane, which results in increased branching morphogenesis. 18316618_the mHpa398 and mHpa519 peptides are novel H-2K(b)-restricted CTL epitopes capable of inducing heparanase-specific CTLs in vitro and in vivo 18334674_heparanase modification of the BM microenvironment regulates the retention and proliferation of hematopoietic progenitor cells. 18589009_Data suggest that Heparanase facilitates cartilage replacement by bone at the chondro-osseous junction by removing the HS component of proteoglycans, that otherwise prevent osteogenic cells from remodeling hypertrophic cartilage. 19096032_role for heparanase in the regulation of arterial structure, mechanics, and repair 19360105_Newly generated Hpse knock-out mice unravel co-regulation of Hpse and matrix metalloproteinases. 20100191_Chronic skin barrier disruption activates heparanase and induces gene expression changes, leading to increased growth factor interaction between epidermis and dermis, and facilitating various cutaneous changes, including wrinkle formation. 20226534_findings implicate heparanase in normal processing of heparan sulfate in macrophages, and indicate that heparanase regulates intracellular Ym1 accumulation and crystal formation in airways. 20399501_heparanase induces upregulation of Th2 cytokines, resulting in inhibition of the inflammatory lesion of experimental autoimmune encephalitis 20800024_Downregulating heparanase-1 with siRNA inhibits the invasiveness of mouse hepatoma cells. 21257720_SST0001, a chemically modified heparin, inhibits myeloma growth and angiogenesis via disruption of the heparanase/syndecan-1 axis 21490396_heparanase generates a vicious cycle that powers colitis and the associated tumorigenesis: heparanase, acting synergistically with the intestinal flora, stimulates macrophage activation 21575986_These findings reveal a novel function of heparanase in maintaining mast cell homeostasis 21757697_Heparanase-mediated loss of nuclear syndecan-1 enhances histone acetyltransferase (HAT) activity to promote expression of genes that drive an aggressive tumor phenotype 22106160_Crucial role of heparanase in the pathogenesis of diabetic nephropathy(DN) and its potential as a highly relevant target for therapeutic interventions in patients with DN. 22169133_Cerebral ischemia markedly increased heparanase levels in endothelial cells and then in astrocytes. 22182841_autoimmune destruction of islets in NOD mice was associated with production of catalytically active heparanase 22479599_heparanase acts as a negative modulator of AgRP signaling at MC4R, through cleavage of heparan sulfate chains presumably linked to syndecan-3 22590508_Together with CCR7 and its ligands, and probably MMP-14, heparanase controls DC trafficking. 23054193_Levels of Ext1 and HPSE influence the motility of FBJ osteosarcoma cells. 23162016_Heparanase activates macrophages, resulting in TLR2/3 mediated induction of cytokine expression associated with plaque progression toward vulnerability. 23442498_AGE-induced macrophage migration is dependent on HPA involving RAGE-HPA-PI3K/AKT pathway. Nonenzymatic activity of HPA may play a key role in AGE-induced macrophage migration associated with inflammation in diabetic vascular complication. 23504323_Characterization of heparanase-induced phosphatidylinositol 3-kinase-AKT activation and its integrin dependence. 23509775_heparanase is important for mouse palate formation because it mediates degradation of the ECM of palatal shelves 23644656_Heparanase has roles in lymphangiogenesis and tumor invasion in pancreatic neuroendocrine tumors 24169805_Involvement of heparanase in the pathogenesis of psoriasis and a role for the enzyme in facilitating abnormal interactions between immune and epithelial cell subsets of the affected skin. 24344642_Cleavage of macromolecular heparin by heparanase accelerates the release of heparin and chymase from extracellular matrices. 24532362_Findings indicate an important role for heparanase in immunity and identify the enzyme as a potential target for regulation of an immune response. 24624965_Our findings indicated cell-autonomous HPSE1 modulates clonogenicity, proliferative potential and migration of BM-MSCs and suggested the HS-GAGs may contribute to the niche microenvironment of BM-MSCs 24699306_We found that heparanase induction is associated with decreased levels of CXCL10, suggesting that this chemokine exerts tumor-suppressor properties in myeloma. 24753252_Because HS with unsubstituted glucosamine residues accumulates in heparanase-expressing breast cancer cells, we assumed that these HS structures are resistant to heparanase and can therefore be utilized as a heparanase inhibitor. 25008182_Under diabetic conditions, latent heparanase, overexpressed by glomerular cells and posttranslationally activated by cathepsin L of tubular origin creates chronic inflammatory conditions and fosters macrophage-mediated renal injury. 25189888_Heparanase is suggested to be a prognostic and diagnostic marker for oral premalignant lesions which could have a major impact on future prognosis and diagnosis of SCC of the oral cavity. 25326645_Novel function of heparanase guides cancer-promoting action of TAM and heparanase expression status may be predictive of treatment potential, targeting TAM/IL-6/STAT3. 25702768_Data suggest a potential mechanism of diabetic enteropathy, which is depending remarkably on syndecan-1 (Sdc1) and -beta-D-glucuronidase heparanase (HPSE). 25735873_Results show a critical role for heparanase in regulating the branching and invasive behavior of normal mammary epithelia which could be the result of its mutually reciprocal feedback with MMP-14. 25745999_Findings identify a dual function for HPSE in malignant melanoma with a protumorigenic extracellular activity and a tumor-suppressive nuclear action. 25912399_upregulated through NF-kB and translocated to the cell surface upon herpes simplex virus-1 infection for the removal of heparan sulfate to facilitate viral release 26039697_heparanase contributes to allergen-induced eosinophil recruitment to the lung. 26191183_Unfractionated heparin inhibits heparanase, and reverses the activation of NF-kappaB and MAPK P38 signaling pathways to attenuate inflammatory responses induced by sepsis. These results suggest that UFH 26249176_Studies in heparanase-deficient mice established its contributions to autophagy. 27129145_Heparanase has a role in upregulating platelet adhesion activity and thrombogenicity 27505185_eNOS prevents heparanase induction and the development of proteinuria 27849593_heparanase as a key mediator of macrophage activation and function in tumorigenesis and cross-talk with the tumor microenvironment. 27890389_Study reveals that HPSE stimulates the proliferation of ES cells as well as the expansion of emerging progenitors during neural differentiation. 28081450_Heparanase increases the inflammation in AGEs-stimulated macrophages through activating the RAGE-NF-kappaB pathway. Heparanase driven inflammation from AGEs-stimulated macrophages increases the adherence of glomerular endothelial cells (GEnCs) and augments the permeability of GEnCs. 28170292_the activation of intestinal heparanase contributes to intestinal injury during early sepsis by facilitating the destruction of mucosal epithelial glycocalyx and promoting inflammatory responses. 28209511_Data show that heparanase-1 (HPA-1) induced shedding of heparan sulfate chain from syndecan-1 (SDC-1) facilitated the release of vascular endothelial growth factor C (VEGF-C) from SDC-1/VEGF-C complex into the medium of hepatocarcinoma cell. 28388547_Results demonstrate that acute ischemic injury up-regulates renal heparanase expression and suggest that heparanase plays a deleterious role in the development of renal injury and kidney dysfunction. 28401953_elevated expression of heparanase augmented both the innate and adaptive immune system and propagated inflammatory reactions in the murine rheumatoid arthritis model. 28478303_AGEs and RAGE interaction increases podocyte heparanase expression by activating NF-kappaB signal pathway. 28970256_These results show for the first time that HPSE regulates macrophage polarization as well as renal damage and repair after I/R. HPSE inhibitors could therefore provide a new pharmacologic approach to minimize acute kidney injury and to prevent the chronic profibrotic damages induced by I/R. 29097791_HPSE expression increased in mouse livers after carbon tetrachloride (CCl4)-mediated chronic induction of fibrosis, but with a trend to decline during progression of the disease. 30149916_rHpse was taken up into mast cells and the uptake was not a mere internalization but a process by which proheparanase was properly processed and sorted into secretory granules. Hpse was incorporated at least partly into secretory granules, which was supported by release of Hpse as a result of degranulation. 31128083_metastasis has a predilection to organs with low levels of heparanase, TF, TFPI, and TFPI-2 in the microcirculation, which enables tumor cell retention. Heparanase has a role in regulating the microcirculation milieu. 31481501_Heparanase is preferentially expressed in experimental obesity-associated breast tumors. Heparanase deficiency abolished obesity-accelerated tumor progression in vivo. Heparanase was required for adipose tissue macrophages to produce inflammatory mediators responsible for local induction of aromatase, a rate-limiting enzyme in estrogen biosynthesis. Estrogen upregulated heparanase in hormone-responsive breast tumors. 31690669_Cytokine induction by chemotherapy was not observed in macrophages isolated from heparanase-knockout mice. Mechanistically, paclitaxel and cisplatin activated methylation of histone H3 on lysine 4 (H3K4) in wild-type but not in heparanase-knockout macrophages. This epigenetic, heparanase-dependent host-response mechanism adds a new perspective to the tumor-promoting functions of chemotherapy. 32445051_Upregulation of Fecal Epithelial Heparanase mRNA Is Associated with Increased Ulcerative Colitis Activity and Cancerization Risk. 33051777_Dichotomic role of heparanase in a murine model of metabolic syndrome. 33621216_Disruption of innate defense responses by endoglycosidase HPSE promotes cell survival. 33971986_Heparanase overexpression impedes perivascular clearance of amyloid-beta from murine brain: relevance to Alzheimer's disease. 34592492_Osteoblasts-derived exosomes regulate osteoclast differentiation through miR-503-3p/Hpse axis. 35073921_Histone H4 induces heparan sulfate degradation by activating heparanase in chlorine gas-induced acute respiratory distress syndrome. 35563215_Implications of Heparanase on Heparin Synthesis and Metabolism in Mast Cells. |
ENSG00000173083 |
HPSE |
156.842245 |
2.9031430317 |
1.537616 |
0.21099239 |
52.478393 |
0.00000000000043500809648530770426246640948754138418204731597604961734759854152798652648925781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000091142274080663104530161366459462565867838401700851136411074548959732055664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
231.718868954314 |
34.0095373878914 |
79.8165527580468 |
9.24185838309645 |
| ENSMUSG00000035365 |
75317 |
Parpbp |
protein_coding |
E9QA71 |
Human_homologues FUNCTION: Required to suppress inappropriate homologous recombination, thereby playing a central role DNA repair and in the maintenance of genomic stability. Antagonizes homologous recombination by interfering with the formation of the RAD51-DNA homologous recombination structure. Binds single-strand DNA and poly(A) homopolymers. Positively regulate the poly(ADP-ribosyl)ation activity of PARP1; however such function may be indirect. {ECO:0000269|PubMed:20931645, ECO:0000269|PubMed:22153967}. |
Cytoplasm;Nucleus;Reference proteome |
|
|
|
cytoplasm [GO:0005737]; nucleus [GO:0005634]; negative regulation of double-strand break repair via homologous recombination [GO:2000042] |
23039964_Parpbp(4930547N16Rik) is strongly co-expressed with known cancer-associated genes. 28894029_PARI is a latent modulator of stalled fork processing, which is required for stable genome inheritance under both endogenous and exogenous replication stress |
ENSG00000185480 |
PARPBP |
381.779925 |
2.0098185610 |
1.007065 |
0.18040099 |
30.405607 |
0.00000003505136076115130688845444471499201988251570583088323473930358886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003919987717429197385996225266174830537124762486200779676437377929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
505.047716623783 |
63.3181157993331 |
251.290204218358 |
23.9812554614124 |
| ENSMUSG00000035378 |
72171 |
Shq1 |
protein_coding |
Q7TMX5 |
FUNCTION: Required for the quantitative accumulation of H/ACA ribonucleoproteins (RNPs), including telomerase, probably through the stabilization of DKC1, from the time of its synthesis until its association with NOP10, NHP2, and NAF1 at the nascent H/ACA RNA. {ECO:0000250}. |
Alternative splicing;Cytoplasm;Nucleus;Reference proteome |
|
|
mmu:72171; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; unfolded protein binding [GO:0051082]; box H/ACA snoRNP assembly [GO:0000493]; negative regulation of rRNA processing [GO:2000233]; positive regulation of apoptotic process [GO:0043065]; ribonucleoprotein complex assembly [GO:0022618] |
Human_homologues 21931644_study suggests that GRIM-1 (SHQ1) might act as a co-tumor suppressor in the prostate 25081541_The expression of GRIM-1 and GRP78 was negatively correlated in human non-small cell lung cancer (NSCLC) tissues, and the down-regulation of GRP78 by GRIM-1 provides a possible mechanism for their interaction. 25553844_these results provide insight into the Shq1/dyskerin (Cbf5) interaction that plays a critical role in H/ACA RNP biogenesis and assembly in eukaryotes 26288249_Results from a study on gene expression variability markers in early-stage human embryos shows that SHQ1 is a putative expression variability marker for the 3-day, 8-cell embryo stage. 30323192_a mechanism of NOTCH1-SHQ1-MYC axis in T-cell leukemogenesis, is reported. 32522979_SHQ1 is an ER stress response gene that facilitates chemotherapeutics-induced apoptosis via sensitizing ER-stress response. 34542157_Compound heterozygous variants in SHQ1 are associated with a spectrum of neurological features, including early-onset dystonia. |
ENSG00000144736 |
SHQ1 |
214.915444 |
2.1756035053 |
1.121416 |
0.17785403 |
39.421935 |
0.00000000034143992498956660399683633450300824369039176531259727198630571365356445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000050938422501871489450619650911876445942283453405252657830715179443359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
295.014317078621 |
36.4612666057222 |
135.601140725345 |
12.9187208145654 |
| ENSMUSG00000035407 |
242553 |
Kank4 |
protein_coding |
Q6P9J5 |
FUNCTION: May be involved in the control of cytoskeleton formation by regulating actin polymerization. {ECO:0000250|UniProtKB:Q5T7N3}. |
ANK repeat;Coiled coil;Cytoplasm;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:242553; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; microtubule cytoskeleton [GO:0015630]; negative regulation of actin filament polymerization [GO:0030837]; negative regulation of stress fiber assembly [GO:0051497] |
33253712_Tissue distribution and subcellular localization of the family of Kidney Ankyrin Repeat Domain (KANK) proteins. 35477278_KANK4 Promotes Arteriogenesis by Potentiating VEGFR2 Signaling in a TALIN-1-Dependent Manner. |
ENSG00000132854 |
KANK4 |
359.458406 |
2.1008440313 |
1.070969 |
0.19969580 |
28.253721 |
0.00000010640987970539440969296559246978772783620570407947525382041931152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000011139761427302871448982712898612135177245363593101501464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
474.846015725908 |
65.9375980493672 |
226.02630593112 |
23.934950814481 |
| ENSMUSG00000035459 |
192188 |
Stab2 |
protein_coding |
Q8R4U0 |
FUNCTION: Phosphatidylserine receptor that enhances the engulfment of apoptotic cells. Hyaluronan receptor that binds to and mediates endocytosis of hyaluronic acid (HA). Acts also, in different species, as a primary systemic scavenger receptor for heparin (Hep), chondroitin sulfate (CS), dermatan sulfate (DS), nonglycosaminoglycan (GAG), acetylated low-density lipoprotein (AcLDL), pro-collagen propeptides and advanced glycation end products (AGE). May serve to maintain tissue integrity by supporting extracellular matrix turnover or it may contribute to maintaining fluidity of bodily liquids by resorption of hyaluronan. Counter receptor which plays an important role in lymphocyte recruitment in the hepatic vasculature. Binds to both Gram-positive and Gram-negative bacteria and may play a role in defense against bacterial infection. The proteolytically processed short form also functions as an endocytosis receptor for heparin internalization as well as HA and CS. {ECO:0000269|PubMed:15572036, ECO:0000269|PubMed:18230608}. |
Cell membrane;Cytoplasm;Disulfide bond;EGF-like domain;Endocytosis;Glycoprotein;Hyaluronic acid;Laminin EGF-like domain;Membrane;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:192188; |
cytosol [GO:0005829]; external side of plasma membrane [GO:0009897]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cargo receptor activity [GO:0038024]; hyaluronic acid binding [GO:0005540]; low-density lipoprotein particle binding [GO:0030169]; low-density lipoprotein particle receptor activity [GO:0005041]; scavenger receptor activity [GO:0005044]; cell adhesion [GO:0007155]; defense response to bacterium [GO:0042742]; defense response to Gram-positive bacterium [GO:0050830]; endocytosis [GO:0006897]; hyaluronan catabolic process [GO:0030214]; receptor-mediated endocytosis [GO:0006898] |
12473645_FEEL-1 and this protein are endocytic receptors for advanced glycation end products. 14598175_mStab-2 might be involved in the clearance of hyaluronan from the lymph or the blood circulation via the network of endothelial sinuses 15907457_analysis of mouse fasciclin-containing genes (betaigH3, periostin, stabilin-1, and stabilin-2) expression patterns during valvuloseptal morphogenesis 17626278_describe the development of hepatic sinusoids in mice based on the expression of hyaluronan receptors Stab2 and Lyve-1 19328203_Stabilin-2-mediated aggregation is mediated by homophilic interactions and enhanced in the presence of Ca(2+) and Mg(2+). Interestingly, exogenous addition of FAS1 domains but not EGF-like domains enhances stabilin-2-mediated cell aggregation 19850011_stabilin-2 contributes to adhesion and homing of circulating stem and progenitor cells to bone marrow. 21293057_Data suggest that stabilin-1 and -2 together guarantee proper hepatic clearance of potentially noxious agents in the blood and maintain tissue homeostasis not only in the liver but also distant organs. 21427291_we show that stabilin-1 and stabilin-2 in hepatic sinusoidal endothelial cells (HSECs) are critical in effectively clearing damaged RBCs in mouse liver 21782867_Adrenomedullin treatment from day 14 to day 20 promoted the differentiation of stabilin-2-positive endothelial cells. 22371575_A dramatic elevation in circulating hyaluronic acid levels in Stab2-deficient mice without any overt phenotype, is reported. 26972991_Results show that Stab2 contributes to phosphatidylserine-dependent myoblast fusion and provide new insights into the molecular mechanism by which phosphatidylserine mediates myoblast fusion during muscle growth and regeneration. 27174501_Stab2 recognizes phospatidylserine exposed on apoptotic cells for sensing as an 'eat-me' signal. 27977633_Data show that stabilin-1 and stabilin-2 differentially expressed in isolated pancreatic Islets. 30367035_Stabilin-2 interacts with G protein-coupled receptor BAI3 (BAI3) and stimulates its fusion promoting activity. Stabilin-2 activates the GPCR activity BAI3. The resulting activated heterotrimeric G-proteins contribute to the initial recruitment of Elmo proteins to the membrane, which are then stabilized on BAI3 through a direct interaction. 31912264_Ectopic expression of the Stabilin2 gene triggered by an intracisternal A particle (IAP) element in DBA/2J strain of mice. 33008099_Identifying Stabilin-1 and Stabilin-2 Double Knockouts in Reproduction and Placentation: A Descriptive Study. 33130055_Exploring the transcriptomic network of multi-ligand scavenger receptor Stabilin-1- and Stabilin-2-deficient liver sinusoidal endothelial cells. 34152080_Stabilin-2 deficiency increases thrombotic burden and alters the composition of venous thrombi in a mouse model. 34845225_Bone marrow sinusoidal endothelium controls terminal erythroid differentiation and reticulocyte maturation. |
ENSG00000136011 |
STAB2 |
139.745643 |
10.9311200739 |
3.450369 |
1.02032794 |
7.962476 |
0.00477570294438548620385986964720359537750482559204101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0179038905362736214255114930438139708712697029113769531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
133.830397201532 |
102.739970412176 |
12.2430635009801 |
6.89599420855786 |
| ENSMUSG00000035493 |
21810 |
Tgfbi |
protein_coding |
P82198 |
FUNCTION: Plays a role in cell adhesion (PubMed:8024701). May play a role in cell-collagen interactions (By similarity). {ECO:0000250|UniProtKB:O11780, ECO:0000269|PubMed:8024701}. |
Cell adhesion;Disulfide bond;Extracellular matrix;Gamma-carboxyglutamic acid;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:21810; |
basement membrane [GO:0005604]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; trans-Golgi network [GO:0005802]; cell adhesion molecule binding [GO:0050839]; collagen binding [GO:0005518]; extracellular matrix binding [GO:0050840]; identical protein binding [GO:0042802]; angiogenesis [GO:0001525]; cell adhesion [GO:0007155]; chondrocyte differentiation [GO:0002062]; extracellular matrix organization [GO:0030198]; localization [GO:0051179] |
12704192_betaig-h3 bears alphavbeta3 integrin-interacting motifs that mediate endothelial cell adhesion and migration and angiogenesis 12963107_The spatiotemporal distribution of Beta-ig during various stages of murine development was examined and its ability to support adhesion of various cell types assessed 14967934_During pregnancy, high levels of betaig-h3 mRNA were detected in the uterus on Day 4 of gestation, when the trophoblastic invasion of the implanted blastocysts was very active. 15780949_Recombinant beta ig-h3 and its FAS1 domain significantly inhibit in vitro osteoblast differentiation as evaluated by matrix mineralization/bone nodule formation. 15907457_analysis of mouse fasciclin-containing genes (betaigH3, periostin, stabilin-1, and stabilin-2) expression patterns during valvuloseptal morphogenesis 18249103_keratoepithelin may help to build up a bridge between collagen VI and yet unidentified muscle receptor 18779658_Betaig-h3 may play a critical role as a regulator of chondrogenic differentiation in endochondral ossification. 19415673_Blocking of betaig-h3 expression in E12.5 embryos with antisense oligodeoxynucleotides resulted in cleft of the secondary palate 22644324_the observed effect of TGFBI on bone mass and bone size was largely caused by the effect of TGFBI on periosteal bone formation 22924741_Collectively, our findings indicate that betaig-h3 promotes the fibrogenic influence of TGFbeta signaling, neutralizing the prochondrogenic influence of the hypoxic-inducible factor 1 activated by the microenvironment of limb mesenchymal aggregates. 24506343_High blood TGFBIp levels were observed in cecal ligation and puncture (CLP)-induced septic mice. The absence of the TGFBIp gene in mice attenuated CLP-induced sepsis. 24728038_TGFBI is a potential risk gene for human diabetes. 25378215_The results support a novel inflammatory pathway for diabetic retinopathy. This pathway is initiated by TGFbeta released from macrophages, which promotes synthesis and release of BIGH3 protein by REC and REC apoptosis. 25450762_Tgfbi deficiency is associated with the degradation of cartilage matrix 25740786_Studies reveal that growth hormone (GH)increases TGFBI expression in a mouse and rat models of diabetic nephropathy (DN) which correlate with clinical results, and suggest that this mechanism can explain the podocytes depletion in DN. 26084452_Vicenin-2 and scolymoside could be a potential therapeutic agent for treatment of various severe vascular inflammatory diseases via inhibition of the TGFBIp signaling pathway. 26177052_the data suggest a model by which cytokine-induced Myt3 suppression leads to Tgfbi de-repression and subsequently to impaired islet-cell migration, revealing a novel role for Myt3 in regulating islet-cell migration. 26857144_findings demonstrate a novel role of lithium in the inhibition of colon cancer metastasis by blocking TGFBIp expression, and thereby TGFBIp-induced lymphangiogenesis, in primary tumors. 26878215_TGF-beta-induced matricellular protein-null lungs displayed diminished elastic recoil and gas exchange efficiency 28099942_Data suggest that big-h3 protein (BIGH3) acts as a tumor suppressor in the retina. 32558206_Protein Analysis of the TGFBI(R124H) Mouse Model Gives Insight into Phenotype Development of Granular Corneal Dystrophy. 33264084_Transforming Growth Factor-induced Protein Promotes NF-kappaB-mediated Angiogenesis during Postnatal Lung Development. 33958649_Endothelial angiogenic activity and adipose angiogenesis is controlled by extracellular matrix protein TGFBI. |
ENSG00000120708 |
TGFBI |
1056.153569 |
2.7533964389 |
1.461212 |
0.12683504 |
129.434425 |
0.00000000000000000000000000000544871549436847115789569219802688139623200828142644208173891359036097473966266634126665024950852966867387294769287109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000004129096239314454768104366017128897625061613458182889986736032180874284946807406537772067167679779231548309326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1541.69732342051 |
135.713523098375 |
559.925661870181 |
37.6699118968 |
| ENSMUSG00000035513 |
171171 |
Ntng2 |
protein_coding |
Q8R4F1 |
FUNCTION: Involved in controlling patterning and neuronal circuit formation at the laminar, cellular, subcellular and synaptic levels. Promotes neurite outgrowth of both axons and dendrites. {ECO:0000269|PubMed:11804778, ECO:0000269|PubMed:31668703, ECO:0000269|PubMed:31692205}. |
Alternative splicing;Cell membrane;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;GPI-anchor;Laminin EGF-like domain;Lipoprotein;Membrane;Neurogenesis;Reference proteome;Repeat;Signal |
|
The protein encoded by this gene belongs to a subclass of the netrin family called netrin-G proteins. Unlike classic netrins, which act as diffusible chemoattractants, netrin-Gs are glycosylphosphatidylinositol-anchored membrane proteins that interact with specific transmembrane proteins. In mouse, this gene is preferentially expressed in the cerebral cortex, habenular nucleus and superior colliculus. Knockout mutant mice display a lack of behavioral startle in response to acoustic stimuli. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Apr 2015]. |
mmu:171171; |
axon [GO:0030424]; glutamatergic synapse [GO:0098978]; laminin complex [GO:0043256]; plasma membrane [GO:0005886]; presynaptic active zone membrane [GO:0048787]; Schaffer collateral - CA1 synapse [GO:0098685]; animal organ morphogenesis [GO:0009887]; axonogenesis [GO:0007409]; basement membrane assembly [GO:0070831]; cell migration [GO:0016477]; modulation of chemical synaptic transmission [GO:0050804]; postsynaptic specialization assembly [GO:0098698]; regulation of neuron migration [GO:2001222]; regulation of neuron projection arborization [GO:0150011]; regulation of neuron projection development [GO:0010975]; regulation of presynapse assembly [GO:1905606]; substrate adhesion-dependent cell spreading [GO:0034446]; synaptic membrane adhesion [GO:0099560]; tissue development [GO:0009888] |
11906208_selectively expressed in distinct neuronal subsets that include populations in forebrain, midbrain, hindbrain, spinal cord, and spinal ganglia 17785411_Axonal Ntng2 transneuronally determines lamina-specific subdendritic segments. 17973922_Mice in which netrin-G2 has been genetically inhibited do not startle to an acoustic stimulus, but otherwise perform normally through a behavioral test battery. There were no inner ear structural abnormalities. 24642214_The complementary expression patterns of Ntng1 and Ntng2 are determined by transcriptional cis-regulatory elements widely scattered in these loci. 25411505_netrin-G/NGL interactions differentially control synaptic plasticity in distinct circuits via retrograde signaling mechanisms 26746425_Pre-synaptic netrin-G1 and netrin-G2 diversify the complexity of vertebrate behaviors and differentially regulate post-synaptic properties. |
ENSG00000196358 |
NTNG2 |
436.719118 |
0.3666059457 |
-1.447698 |
0.17489918 |
66.436376 |
0.00000000000000036137145172780578091010891579585811930978297501840246219728669530013576149940490722656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000101557379201437967313139818339946643245514735012791085466687945881858468055725097656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
238.557541871446 |
28.9914316278812 |
650.719238740704 |
57.5043312336941 |
| ENSMUSG00000035818 |
226245 |
Plekhs1 |
protein_coding |
Q8BW88 |
|
Alternative splicing;Reference proteome |
|
|
mmu:226245; |
|
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 27523322_this gene is involved in the mild elevation of blood glucose levels and insulin resistance in obesity. 32326836_Risk Stratification Using a Novel Genetic Classifier Including PLEKHS1 Promoter Mutations for Differentiated Thyroid Cancer with Distant Metastasis. |
ENSG00000148735 |
PLEKHS1 |
20.956318 |
3.5324734123 |
1.820679 |
0.65166072 |
7.572281 |
0.00592727333580437241999927522329016937874257564544677734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0214968201944023898364299896002194145694375038146972656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.9471687916263 |
15.8605991371844 |
9.61002811056393 |
3.47576141239428 |
| ENSMUSG00000035829 |
241289 |
Ppp1r26 |
protein_coding |
Q6A025 |
FUNCTION: Inhibits phosphatase activity of protein phosphatase 1 (PP1) complexes. May positively regulate cell proliferation. {ECO:0000269|PubMed:16053918}. |
Nucleus;Phosphoprotein;Protein phosphatase inhibitor;Reference proteome |
|
|
mmu:241289; |
nucleolus [GO:0005730]; protein phosphatase inhibitor activity [GO:0004864]; negative regulation of phosphatase activity [GO:0010923] |
Human_homologues 16053918_we showed that KIAA0649 mRNA is widely expressed in human multiple tissues and cell lines. We have also demonstrated that KIAA0649 has oncogenic characteristics: it enhances colony formation, allows anchorage-independent growth. 26442585_identified NRBE3 as a novel ubiquitin E3 ligase for RB that might play a role as a potential oncoprotein in human cancers 30262434_Results show that up-regulation of NRBE3 is correlated with lymphatic metastasis in human breast cancer (BC) tissues. Ectopic expression of NRBE3 promotes migration and invasion in BC cells. Accordingly, its knockdown inhibits migration and invasion in BC cells and lung metastasis in vivo. |
ENSG00000196422 |
PPP1R26 |
17.900832 |
14.1002754401 |
3.817651 |
0.85935111 |
19.347265 |
0.00001089752263612475758681498450020086465883650816977024078369140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000802664723372593322823012607081238911632681265473365783691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.0086972967305 |
20.2138398133186 |
2.27007603026291 |
1.20346115954973 |
| ENSMUSG00000035864 |
20979 |
Syt1 |
protein_coding |
P46096 |
FUNCTION: Calcium sensor that participates in triggering neurotransmitter release at the synapse (PubMed:11242035). May have a regulatory role in the membrane interactions during trafficking of synaptic vesicles at the active zone of the synapse (PubMed:7961887). It binds acidic phospholipids with a specificity that requires the presence of both an acidic head group and a diacyl backbone. A Ca(2+)-dependent interaction between synaptotagmin and putative receptors for activated protein kinase C has also been reported. It can bind to at least three additional proteins in a Ca(2+)-independent manner; these are neurexins, syntaxin and AP2. Plays a role in dendrite formation by melanocytes (By similarity). {ECO:0000250|UniProtKB:P21579, ECO:0000269|PubMed:11242035, ECO:0000269|PubMed:7961887}. |
3D-structure;Calcium;Cytoplasm;Cytoplasmic vesicle;Differentiation;Direct protein sequencing;Glycoprotein;Lipoprotein;Membrane;Metal-binding;Palmitate;Phosphoprotein;Reference proteome;Repeat;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:20979; |
axon [GO:0030424]; chromaffin granule membrane [GO:0042584]; cytoplasm [GO:0005737]; dense core granule [GO:0031045]; excitatory synapse [GO:0060076]; exocytic vesicle [GO:0070382]; glutamatergic synapse [GO:0098978]; Golgi apparatus [GO:0005794]; hippocampal mossy fiber to CA3 synapse [GO:0098686]; intracellular organelle [GO:0043229]; neuron projection [GO:0043005]; neuron projection terminus [GO:0044306]; plasma membrane [GO:0005886]; presynaptic membrane [GO:0042734]; secretory granule [GO:0030141]; synaptic vesicle [GO:0008021]; synaptic vesicle membrane [GO:0030672]; calcium ion binding [GO:0005509]; calcium ion sensor activity [GO:0061891]; calcium-dependent phospholipid binding [GO:0005544]; calcium-dependent protein binding [GO:0048306]; calmodulin binding [GO:0005516]; clathrin binding [GO:0030276]; identical protein binding [GO:0042802]; low-density lipoprotein particle receptor binding [GO:0050750]; phosphatidylinositol-4,5-bisphosphate binding [GO:0005546]; phosphatidylserine binding [GO:0001786]; phospholipid binding [GO:0005543]; protein C-terminus binding [GO:0008022]; protein heterodimerization activity [GO:0046982]; SNARE binding [GO:0000149]; syntaxin binding [GO:0019905]; syntaxin-1 binding [GO:0017075]; syntaxin-3 binding [GO:0030348]; brain development [GO:0007420]; calcium ion-regulated exocytosis of neurotransmitter [GO:0048791]; calcium-dependent activation of synaptic vesicle fusion [GO:0099502]; calcium-ion regulated exocytosis [GO:0017156]; cell differentiation [GO:0030154]; cellular response to calcium ion [GO:0071277]; detection of calcium ion [GO:0005513]; fast, calcium ion-dependent exocytosis of neurotransmitter [GO:0098746]; neurotransmitter secretion [GO:0007269]; positive regulation of calcium ion-dependent exocytosis [GO:0045956]; positive regulation of calcium ion-dependent exocytosis of neurotransmitter [GO:1903235]; positive regulation of dendrite extension [GO:1903861]; positive regulation of dopamine secretion [GO:0033603]; positive regulation of synaptic transmission [GO:0050806]; positive regulation of vesicle fusion [GO:0031340]; protein heterooligomerization [GO:0051291]; regulation of calcium ion-dependent exocytosis [GO:0017158]; regulation of dopamine secretion [GO:0014059]; regulation of regulated secretory pathway [GO:1903305]; regulation of synaptic transmission, glutamatergic [GO:0051966]; regulation of synaptic vesicle exocytosis [GO:2000300]; response to calcium ion [GO:0051592]; spontaneous neurotransmitter secretion [GO:0061669]; synaptic vesicle endocytosis [GO:0048488]; synaptic vesicle exocytosis [GO:0016079]; synchronous neurotransmitter secretion [GO:0071911]; vesicle docking [GO:0048278]; vesicle-mediated transport [GO:0016192] |
12047557_Ca2+ modulates dynamic docking of granules to the plasma membrane and this process is due to a Ca2+-dependent interaction between SNAP-25 and synaptotagmin 12089155_synaptotagmin-I expressing neuroblastoma cells require gangliosides for Botulinum neurotoxin A activity 12526776_In synaptotagmin 1 knockout mice, the fast component of both Ca(2+)- and Sr(2+)-induced neurotransmitter release is impaired, suggesting that both cations partly act by binding to synaptotagmin 1. 12939392_Ca2+-dependent phospholipid binding to synaptotagmin 1 mirrors the intracellular Ca2+ dependence of exocytosis. 14504267_syts I and II can function as protein receptors for Botulinum neurotoxin b and mediate its entry 14559220_alternative translation of synaptotagmin 1 mediates the non-classical release of fibroblast growth factor 1 15123599_Synaptotagmins I and II are nerve cell receptors for botulinum neurotoxin G 15240804_Synaptotagmin I synchronizes the rapid release of neurotransmitters after Ca2+ entry into presynaptic terminals and also appears to suppress the slower, asynchronous form of transmitter release. 15456828_We have used point mutagenesis to determine the functional contributions to triggering synchronous neurotransmitter release of the five negatively charged aspartate residues that constitute the Ca2+-binding sites in the C2B domain of synaptotagmin I. 15466855_SV2B-synaptotagmin 1 complex is involved in the regulation of synaptotagmin 1 stability and/or trafficking 15492212_endocytosis is significantly impaired in the absence of SytI with the relative rates of endocytosis compared with exocytosis reduced approximately 3-fold with respect to wild type 15561725_synaptotagmin 1 function requires a free C terminus, possibly because of spatial constraints at the release sites 15886015_We conclude that synaptotagmin self-oligomerization is unlikely to play any significant role in triggering synaptic vesicle exocytosis. 16301172_Synaptotagmin 1 occupies release 'slots' at the active zone, possibly in a Ca(2+)-independent complex with SNARE proteins that are freed when action potential-induced Ca(2+) influx activates synaptotagmin 1. 16352718_apparent Ca2+ sensitivity of release is dictated by the apparent Ca2+ affinity of synaptotagmin 1 in both directions, and that Ca2+ binding to both C2 domains contributes to Ca2+ triggering of release 16407561_Overexpression of Syt1 rescued the readily releasable pool & increased its size above wild-type values, whereas the size of the slowly releasable pool decreased. Availability of Syt1 regulates the relative size of the two releasable pools. 16595652_Ca2+-dependent phospholipid binding modes of synaptotagmin 1 C2 domains may underlie their functional asymmetry and suggest that phosphatidylinositol polyphosphates may serve as physiological modulators of Ca2+ affinity of synaptotagmin 1 in vivo 16930531_FGF1 and p40 synaptotagmin 1 release correlates with membrane destabilizing ability 16990140_Here we propose that complexin binding activates SNARE complexes into a metastable state and that Ca(2+) binding to synaptotagmin 1 triggers fast exocytosis by displacing complexin from metastable SNARE complexes. 17135417_Fast vesicle exocytosis is driven by a dual Ca2+-dependent activity of synaptotagmin-1, namely calcium-dependent binding both to soluble N-ethylmaleimide-sensitive factor attachment protein receptor complex (SNARE) complexes and to phospholipids. 17492637_The cell-, temporal-, and species-specific expression of synaptotagmin isoforms suggests that each may have distinct functions in neurotransmitter release 18158333_BoNT/B and G fail to bind and enter hippocampal neurons cultured from Syt I knockout mice. Syts I/II are protein receptors for BoNT/B and G. 18308932_These data establish synaptotagmin-7 as a major Ca(2+) sensor for exocytosis in chromaffin cells, which, together with synaptotagmin-1, mediates almost all of the Ca(2+) triggering of exocytosis in these cells. 18374609_upregulation of TrkB and synaptotagmin in the hippocampus contributes to the exercise-facilitated aversive memory 18622390_Ca(2+)-synaptotagmin must insert into the target membrane to accelerate SNARE-catalyzed fusion 18832148_Quantitative analysis revealed that elimination of synaptotagmin 1 reduced release probability and depression but maintained the synchrony of transmitter release at BC-GC (basket cells (BCs) and granule cells (GCs)) synapses 18953334_Data show that synaptotagmin-1 function depends on the bottom face of the C(2)B domain and support the notion that synaptotagmin-1 triggers membrane fusion and neurotransmitter release by bringing the vesicle and plasma membranes together. 19412166_These data suggest that Syt1 controls both evoked and spontaneous release at a synapse as a simultaneous Ca2+-dependent activator and clamp of exocytosis. 19451201_both treadmill and wheel running exercise protocols upregulated the BDNF-TrkB pathway and Syt I in the hippocampus, whereas only treadmill exercise upregulated their expression levels in the amygdala 19716167_Study identifies the vesicular docking partner as synaptotagmin-1 and SNAP-25 as an essential plasma membrane docking factor, which, together with the previously known docking factors Munc18-1 and syntaxin, form the minimal docking machinery. 19805322_Data show that the unique requirement for Ca2+ binding to the C2B domain for synaptotagmin-1 in Ca2+-triggered neurotransmitter release may be accounted for by the unusual phospholipid-binding properties of its double C2A/B-domain fragment. 19926819_intestinal epithelial Syt 1 plays an important role in cAMP-stimulated endocytosis of apical NHE3 through cAMP-dependent phosphorylation of S605 that is required for NHE3 and Syt 1 association 20219474_1.9-A X-ray structure of the BoNT serotype G receptor binding domain on the protein receptor synaptotagmin (Syt) I/II. 20808911_Expression of Syt1 in the mouse prefrontal cortex is modulated by a sequence variant (B2 SINE indel) in the 3' UTR of Comt (catechol-O-methyltransferase). 20824061_The Syt7 C2B domain structure is very similar to that of the Syt1 C2B domain and contains three Ca2+-binding sites. 20926654_Transient expression of Syt1 is no longer detected in cochlear inner hair cells (IHCs) after the onset of hearing, indicating that the most common calcium-ion sensors in the central nervous system are not involved in the mature IHC ribbon synapse. 21129439_new-synthesized synaptotagmin1 following synaptic plasticity may contribute to long-lasting synaptic plasticity via positive, feedfoward mechanisms 21451027_A test of Syt1 exocytosis in otoferlin-deficient hippocampal neurons and Syt1-deficient inner hair cells finds no deficits in vesicle fusion. 21816209_this study demonistrated that Alcohol induces synaptotagmin 1 expression in neurons via activation of heat shock factor 1 22184197_This system recapitulates some of the effects of syt mutations that alter synaptic transmission in neurons. It demonstrate that the cytoplasmic domain of syt exhibited mixed agonist/antagonist activity during regulated membrane fusion in cells. 22197832_This study demonistrated that syt1 functions as a dual Ca(2+) sensor for both endo- and exocytosis, potentially coupling these two components of the vesicle cycle. 22313732_Data suggest that Syt1 may act as a MTOC-associated protein to play important roles in mouse oocyte spindle organization/stability, and that it is indispensable for the metaphase-anaphase transition to promote mouse oocyte meiotic maturation. 22423098_The dynamics of vesicle fission during clathrin-mediated endocytosis is Ca(2)+ dependent but becomes Ca(2)+ independent in synaptotagmin 1 knock-out cells. 22731840_APP interacts with the calcium sensor synaptotagmin-1 of synaptic vesicles and might thus play a role in the regulation of synaptic vesicle exocytosis. 22810233_SNAREpin assembly by Munc18-1 requires previous vesicle docking by synaptotagmin 1. 22908222_Glycosylation is dispensable for sorting of synaptotagmin 1 but is critical for targeting of SV2 and synaptophysin to recycling synaptic vesicles. 23648184_Synaptotagmins 1 and 2 as mediators of rapid exocytosis at nerve terminals: the dyad hypothesis 24005294_Syt-1 SNARE-interacting domains are essential for vesicle docking, priming, and fast fusion-triggering in chromaffin cells. 24267651_Ca(2+)-triggered release at a synapse is due to synaptotagmins, with synaptotagmin-7 mediating a slower form of Ca(2+)-triggered release that is normally occluded by faster synaptotagmin-1-induced release but becomes manifest upon synaptotagmin-1 deletion 24501351_In mixed glutamatergic/GABAergic neurons, excitatory postsynaptic potential (EPSP) frequencies were increased in synaptotagmin-1-null neurons. 24657966_The syt function is markedly affected by changes in the physical nature of the linker that connects its tandem C2 domains. Moreover, the linker mutations uncoupled syt-mediated regulation of evoked and spontaneous release. 24973144_Increased expression of Syt1 in neural stem cells is essential for neurogenesis progression. 25242442_the Ca(2+) dependence of the interaction between PRIP-C2 and Syt1-C2A was attributed to Ca(2+) binding with Syt1-C2A, but not PRIP-C2, using a series of mutants prepared from both C2 domains. 25673844_This study reveals fundamental details of how phosphorylation of Thr84 on SV2A controls its interaction with synaptotagmin-1 and implicates SV2A as a phospho-dependent chaperone 25735570_Tissue-specific splicing, posttranslational modification and the variation in expression might suggest a different requirement of SYT1 for the specific function in each organ. 26202512_Syt-1 regulates Abeta levels in mouse neurons. Knockdown of endogenous Syt-1 in mouse primary neurons led to a significant reduction in both Abeta40 and Abeta42 generation. 26280336_atomic-resolution crystal structures of Ca(2+)- and Mg(2+)-bound complexes between synaptotagmin-1 and the neuronal SNARE complex 26400647_The interaction of Dvl1 with Syt-1, which is regulated by Wnts, modulates neurotransmitter release. 26437117_Data (including data from studies in knockout mice) suggest that loss of both Syt1/Syt7 decreases capacity of readily-releasable pool (RRP) of Ca2+ in synaptic vesicles without altering rate of priming into RRP; Syt1/Syt7 functions appear redundant. 26490858_This study provides new insights in how the two opposite sides of the C2B domain of Synaptotagmin-1 participate in secretory vesicle fusion, and in more upstream steps, especially vesicle docking. 26709651_data show that hepatic Syt1 expression is influenced by diet and hormonal milieu 27001899_different structural states of syt underlie the control of distinct forms of synaptic transmission. 27091977_We conclude that synaptotagmin-1 phosphorylation is an essential step in PKC-dependent potentiation of synaptic transmission, acting downstream of the two other essential DAG/PKC substrates, Munc13-1 and Munc18-1. 27210552_demonstrates a developmental Syt1-Syt2 isoform switch at an identified synapse, a mechanism that could fine-tune the speed, reliability, and plasticity of transmitter release at fast releasing CNS synapses. 27399837_the combined inactivation of all 3 E-Syt genes has no effect on mouse viability or fertility. 27881774_The function of synaptotagmin-1 (syt-1):soluble NSF attachment protein receptor (SNARE) interactions during neurotransmission remains unclear. 28173138_Authors propose that the strong reduction of Syt2 and SV2B are key factors of the functional synaptic alteration and that the physiological downregulation of Syt1 plays a determinant role in muscle vulnerability in SMA. 28193235_that reduction in the synaptotagmin 1 level and presenilin 1-synaptotagmin 1 interactions in AD brain may present molecular underpinning of the pathogenic presenilin 1 conformation 28355182_results suggest that postsynaptic Syt1 and Syt7 act as redundant Ca(2+)-sensors for Ca(2+)-dependent exocytosis of AMPA receptors during long-term potentiation, and thereby delineate a simple mechanism for the recruitment of AMPA receptors that mediates LTP 28363983_we identify Syt2 as a functionally important Ca(2+) sensor at fast-releasing inhibitory synapses, and show that Syt1 and Syt2 can redundantly control transmitter release at specific brain synapses 28686803_Study showed postsynaptic localization of synaptotagmin 1, at concentrations moderately lower than, but comparable to presynaptic concentrations. It is present in significant concentrations at the postsynaptic density, pointing to the likelihood of insertion of glutamate receptors directly into the synaptic plasma membrane. Synaptotagmin 1 is reduced in postsynaptic spines after eight weeks of kainite-induced epilepsy. 29230057_The data of this study suggested that Syt1-mediated docking activity works in concert with docking induced by SNARE complex assembly. 29274147_Mutating Ca(2+)-coordinating aspartates in the C2A-domain localizes Doc2B permanently at the plasma membrane, and renders an upstream priming step Ca(2+)-independent, whereas a separate function in downstream priming depends on SNARE-binding, Ca(2+)-binding to the C2B-domain of Doc2B, interaction with ubMunc13-2 and the presence of synaptotagmin-1. 29754754_Doc2alpha promoted glutamatergic spontaneous neurotransmitter release, while Doc2beta and syt1 both regulated GABAergic spontaneous neurotransmitter release. Ca(2+) entry via voltage-gated Ca(2+) channels triggered miniature GABA release by activating syt1. 31172949_Photoreceptors lacking Syt1 exhibited marked reductions in exocytosis as measured by electroretinography and single-cell recordings. Syt1 mediated all evoked release in cones, whereas rods appeared capable of some slow Syt1-independent release. 31383906_Synaptotagmin-1 enables frequency coding by suppressing asynchronous release in a temperature dependent manner. 31501440_the C2B domain of syt1, independent of complexin, is the molecular clamp that arrests synaptic vesicles prior to Ca(2+)-triggered fusion. 31787133_Synaptotagmin 1 regulates cortical granule exocytosis during mouse oocyte activation. 32015138_Syt1(F349A) is more efficient than wild-type Syt1 (Syt1(WT)) in triggering synchronous transmitter release. 32098902_Doc2 Proteins Are Not Required for the Increased Spontaneous Release Rate in Synaptotagmin-1-Deficient Neurons. 32341095_An Epilepsy-Associated SV2A Mutation Disrupts Synaptotagmin-1 Expression and Activity-Dependent Trafficking. 32490813_Synaptotagmin-1 is the Ca(2+) sensor for fast striatal dopamine release. 32515733_Acute disruption of the synaptic vesicle membrane protein synaptotagmin 1 using knockoff in mouse hippocampal neurons. 32771460_Hepatic Synaptotagmin 1 is involved in the remodelling of liver plasma- membrane lipid composition and gene expression in male Apoe-deficient mice consuming a Western diet. 32831174_Post-tetanic potentiation lowers the energy barrier for synaptic vesicle fusion independently of Synaptotagmin-1. 33571908_miR-128 regulates epilepsy sensitivity in mice by suppressing SNAP-25 and SYT1 expression in the hippocampus. 33730593_Synaptotagmin-1 interacts with PI(4,5)P2 to initiate synaptic vesicle docking in hippocampal neurons. 33753311_Ca(2+) sensor proteins in spontaneous release and synaptic plasticity: Limited contribution of Doc2c, rabphilin-3a and synaptotagmin 7 in hippocampal glutamatergic neurons. 33903230_Neuromodulator release in neurons requires two functionally redundant calcium sensors. 34151409_miR-128 as a Regulator of Synaptic Properties in 5xFAD Mice Hippocampal Neurons. 35193927_Deconstructing Synaptotagmin-1's Distinct Roles in Synaptic Vesicle Priming and Neurotransmitter Release. 35361702_Synaptotagmins 1 and 7 Play Complementary Roles in Somatodendritic Dopamine Release. 35537054_Synaptotagmin-1 is a bidirectional Ca(2+) sensor for neuronal endocytosis. 35701163_Polybasic Patches in Both C2 Domains of Synaptotagmin-1 Are Required for Evoked Neurotransmitter Release. 36640316_Synaptotagmin-1 is a Ca[2+] sensor for somatodendritic dopamine release. |
ENSG00000067715 |
SYT1 |
9.524503 |
0.0520241618 |
-4.264674 |
1.19392342 |
14.013503 |
0.00018150249735250854042113710651307201260351575911045074462890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010201645902853911495461458613931426953058689832687377929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.02151485962822 |
0.945221996975581 |
19.6353929319006 |
9.2640193099076 |
| ENSMUSG00000035954 |
238130 |
Dock4 |
protein_coding |
P59764 |
FUNCTION: Functions as a guanine nucleotide exchange factor (GEF) that promotes the exchange of GDP to GTP, converting inactive GDP-bound small GTPases into their active GTP-bound form. Involved in regulation of adherens junction between cells. Plays a role in cell migration. {ECO:0000250|UniProtKB:Q8N1I0}.; FUNCTION: [Isoform 2]: Has a higher guanine nucleotide exchange factor activity compared to other isoforms. {ECO:0000250|UniProtKB:Q8N1I0}. |
Alternative splicing;Cell membrane;Cytoplasm;Guanine-nucleotide releasing factor;Membrane;Phosphoprotein;Reference proteome;SH3 domain;SH3-binding |
|
|
mmu:238130; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; nucleolus [GO:0005730]; plasma membrane [GO:0005886]; stereocilium [GO:0032420]; stereocilium bundle [GO:0032421]; GTPase activator activity [GO:0005096]; guanyl-nucleotide exchange factor activity [GO:0005085]; PDZ domain binding [GO:0030165]; receptor tyrosine kinase binding [GO:0030971]; SH3 domain binding [GO:0017124]; small GTPase binding [GO:0031267]; cell chemotaxis [GO:0060326]; negative regulation of vascular associated smooth muscle contraction [GO:1904694]; positive regulation of vascular associated smooth muscle cell migration [GO:1904754]; small GTPase mediated signal transduction [GO:0007264] |
16464467_Rac-DOCK4-actin-binding protein harmonin-activated signaling pathway is possibly involved in regulating actin cytoskeleton organization in stereocilia of the inner ear. 23720743_The Atypical guanine nucleotide exchange factor Dock4 regulates neurite differentiation through modulation of Rac1 GTPase and actin dynamics. 24508479_Dock4 binds to SH3YL1 through the C-terminal proline-rich region. 31019307_SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis 31388105_Autism-like social deficit generated by Dock4 deficiency is rescued by restoration of Rac1 activity and NMDA receptor function. 33559155_DOCK4 stimulates MUC2 production through its effect on goblet cell differentiation. 33705839_Altered postnatal developmental patterns of ultrasonic vocalizations in Dock4 knockout mice. 35302184_DOCK4 regulates ghrelin production in gastric X/A-like cells. 35477279_DOCK4 Regulation of Rho GTPases Mediates Pulmonary Vascular Barrier Function. |
ENSG00000128512 |
DOCK4 |
280.849228 |
0.4012406412 |
-1.317460 |
0.17041044 |
59.250680 |
0.00000000000001388123805331009297598232192464612704201445177870644087647633568849414587020874023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000003415816123231330092512972053315374384584342248860622248685103841125965118408203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
162.602380062287 |
19.2512072964989 |
405.249028503397 |
34.4351245670813 |
| ENSMUSG00000036036 |
22715 |
Zfp57 |
protein_coding |
Q8C6P8 |
FUNCTION: Transcription regulator required to maintain maternal and paternal gene imprinting, a process by which gene expression is restricted in a parent of origin-specific manner by epigenetic modification of genomic DNA and chromatin, including DNA methylation. Acts by controlling DNA methylation during the earliest multicellular stages of development at multiple imprinting control regions (ICRs) (PubMed:15070898, PubMed:18854139, PubMed:23059534, PubMed:30602440). Acts together with ZNF445, but ZFP57 plays the predominant role in imprinting maintenance. In contrast, in humans, ZNF445 seems to be the major factor early embryonic imprinting maintenance (PubMed:30602440). Required for the establishment of maternal methylation imprints at SNRPN locus. Acts as a transcriptional repressor in Schwann cells. Binds to a 5'-TGCCGC-3' consensus sequence and recognizes the methylated CpG within this element (PubMed:15070898, PubMed:18854139, PubMed:23059534). {ECO:0000269|PubMed:15070898, ECO:0000269|PubMed:18854139, ECO:0000269|PubMed:23059534, ECO:0000269|PubMed:30602440}. |
3D-structure;Alternative splicing;Developmental protein;DNA-binding;Metal-binding;Nucleus;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:22715; |
heterochromatin [GO:0000792]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; double-stranded methylated DNA binding [GO:0010385]; metal ion binding [GO:0046872]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; DNA methylation involved in embryo development [GO:0043045]; genomic imprinting [GO:0071514]; maintenance of DNA methylation [GO:0010216]; negative regulation of transcription by RNA polymerase II [GO:0000122]; regulation of gene expression by genomic imprinting [GO:0006349]; regulation of genetic imprinting [GO:2000653]; regulation of transcription by RNA polymerase II [GO:0006357] |
18854139_Zpf57 maintains both maternal and paternal genomic imprints. 22055183_In embryonic stem cells, ZFP57/KAP1 recognize a methylated hexanucleotide to affect chromatin and DNA methylation of imprinting control regions. 22144682_ZFP57 may recruit DNA methyltransferases to its target regions to maintain DNA methylation imprint, and this interaction is likely facilitated by KAP1/TRIM28/TIF1beta. 23059534_the structure of the DNA-binding domain of Zfp57, consisting of two adjacent zinc fingers, in complex with fully methylated DNA 24135613_Mouse and human ZFP57 are orthologs despite relatively low sequence identity. 24236546_Data suggest that Glu182 in the DNA binding site in zinc finger motif of Zfp57 plays important role in binding specificity and affinity for 5-carboxylcytosine and other cytosine analogs. 25445595_Taken together, our results suggest that Nanog positively regulates Zfp57 expression in multiple types of cells. 25848000_ZFP57 may modulate NOTCH signaling during cardiac development. 26025256_Within established mouse imprinted clusters, ZFP57 specifically binds known or predicted imprinting control region elements, but does not interact with secondary somatically differentially methylated regions (DMRs) or germline DMRs that are not imprinting controlling. 26481358_Data show that ZFP57 binding is affected by single nucleotide polymorphism (SNPs) and closely associated TGCCGC motif sequences that are present at the target sites. 27257070_another function of ZFP57 in early embryogenesis is to repress cis-acting regulatory elements whose activity is not yet required. 28476430_A group of functionally significant composite DNA elements (ZFP57 binding site (ZFBS)-Morph overlaps) that may play dual roles in the regulation of allele-specific gene expression. 34226608_Zfp57 inactivation illustrates the role of ICR methylation in imprinted gene expression during neural differentiation of mouse ESCs. |
ENSG00000204644 |
ZFP57 |
185.904183 |
3.0099668607 |
1.589748 |
0.23414610 |
45.558718 |
0.00000000001481309141196618481838055645679843490628013746857050136895850300788879394531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000002596467300196420133805332652274797458624711055108491564169526100158691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
284.591650493822 |
46.3916112361796 |
94.5497620626995 |
12.1917410018403 |
| ENSMUSG00000036086 |
226409 |
Zranb3 |
protein_coding |
Q6NZP1 |
FUNCTION: DNA annealing helicase and endonuclease required to maintain genome stability at stalled or collapsed replication forks by facilitating fork restart and limiting inappropriate recombination that could occur during template switching events. Recruited to the sites of stalled DNA replication by polyubiquitinated PCNA and acts as a structure-specific endonuclease that cleaves the replication fork D-loop intermediate, generating an accessible 3'-OH group in the template of the leading strand, which is amenable to extension by DNA polymerase. In addition to endonuclease activity, also catalyzes the fork regression via annealing helicase activity in order to prevent disintegration of the replication fork and the formation of double-strand breaks. {ECO:0000250|UniProtKB:Q5FWF4}. |
ATP-binding;Chromosome;Direct protein sequencing;DNA damage;DNA repair;Endonuclease;Helicase;Hydrolase;Metal-binding;Multifunctional enzyme;Nuclease;Nucleotide-binding;Nucleus;Reference proteome;Zinc;Zinc-finger |
|
|
mmu:226409; |
nuclear replication fork [GO:0043596]; nucleoplasm [GO:0005654]; ATP binding [GO:0005524]; ATP-dependent chromatin remodeler activity [GO:0140658]; ATP-dependent DNA/DNA annealing activity [GO:0036310]; endodeoxyribonuclease activity [GO:0004520]; helicase activity [GO:0004386]; K63-linked polyubiquitin modification-dependent protein binding [GO:0070530]; metal ion binding [GO:0046872]; cellular response to DNA damage stimulus [GO:0006974]; DNA repair [GO:0006281]; DNA rewinding [GO:0036292]; negative regulation of DNA recombination [GO:0045910]; replication fork processing [GO:0031297]; replication fork protection [GO:0048478]; response to UV [GO:0009411] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 21078962_structural and functional differences between AH2 and HARP suggest that different annealing helicases have distinct functions in the cell. 22704558_ZRANB3 translocase, a SNF2 family member related to the SIOD disorder SMARCAL1 protein, is recruited by polyubiquitinated PCNA to promote fork restart following replication arrest. 22705370_AH2 is recruited to stalled replication forks and that cells depleted of AH2 are hypersensitive to replication stresses. 22759634_a role for ZRANB3 in the replication stress response and suggest new insights into how DNA repair is coordinated with DNA replication to maintain genome stability. 26884333_Here is described a substrate recognition domain within ZRANB3 that is needed for it to recognize forked DNA structures, hydrolyze ATP, catalyze fork remodeling, and act as a structure-specific endonuclease. 28621305_PCNA and ATP-dependency serve as a multi-layered regulatory mechanism that modulates ZRANB3 activity at replication forks. 28886337_Damage-induced fork reversal in mammalian cells requires PCNA ubiquitination, UBC13, and K63-linked polyubiquitin chains, previously involved in error-free damage tolerance. Fork reversal in vivo also requires ZRANB3 translocase activity and its interaction with polyubiquitinated PCNA, pinpointing ZRANB3 as a key effector of error-free DNA damage tolerance. 28954549_A large number of SNF2 family, DNA and ATP-dependent motor proteins are needed during transcription, DNA replication, and DNA repair to manipulate protein-DNA interactions and change DNA structure. SMARCAL1, ZRANB3, and HLTF are three related members of this family with specialized functions that maintain genome stability during DNA replication. [review] 29053959_depletion of SMARCAL1, a SNF2-family DNA translocase that remodels stalled forks, restores replication fork stability and reduces the formation of replication stress-induced DNA breaks and chromosomal aberrations in BRCA1/2-deficient cells. In addition to SMARCAL1, other SNF2-family fork remodelers, including ZRANB3 and HLTF, cause nascent DNA degradation and genomic instability 30150681_Nuclear RNR-alpha antagonizes cell proliferation by directly inhibiting ZRANB3 31324766_ZRANB3 is an African-specific type 2 diabetes locus associated with beta-cell mass and insulin response. 32971328_Time for remodeling: SNF2-family DNA translocases in replication fork metabolism and human disease. |
ENSG00000121988 |
ZRANB3 |
387.366419 |
2.0798137678 |
1.056454 |
0.14750790 |
50.392708 |
0.00000000000125861044890131601446783182496603518897744511395586641810950823128223419189453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000247689332786367490711596398168854878639455474598207729286514222621917724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
528.622429932553 |
54.143055391727 |
254.168155878728 |
19.9794757788149 |
| ENSMUSG00000036098 |
225908 |
Myrf |
protein_coding |
Q3UR85 |
FUNCTION: [Myelin regulatory factor]: Constitutes a precursor of the transcription factor. Mediates the autocatalytic cleavage that releases the Myelin regulatory factor, N-terminal component that specifically activates transcription of central nervous system (CNS) myelin genes. {ECO:0000269|PubMed:23966833, ECO:0000269|PubMed:28623291}.; FUNCTION: [Myelin regulatory factor, C-terminal]: Membrane-bound part that has no transcription factor activity and remains attached to the endoplasmic reticulum membrane following cleavage. {ECO:0000269|PubMed:23966833}.; FUNCTION: [Myelin regulatory factor, N-terminal]: Transcription factor that specifically activates expression of myelin genes such as MBP, MOG, MAG, DUSP15 and PLP1 during oligodendrocyte (OL) maturation, thereby playing a central role in oligodendrocyte maturation and CNS myelination (PubMed:19596243, PubMed:22956843, PubMed:23966833, PubMed:24204311, PubMed:27532821). Specifically recognizes and binds DNA sequence 5'-CTGGYAC-3' in the regulatory regions of myelin-specific genes and directly activates their expression. Not only required during oligodendrocyte differentiation but is also required on an ongoing basis for the maintenance of expression of myelin genes and for the maintenance of a mature, viable oligodendrocyte phenotype (PubMed:19596243, PubMed:22956843, PubMed:23966833). {ECO:0000269|PubMed:19596243, ECO:0000269|PubMed:22956843, ECO:0000269|PubMed:23966833, ECO:0000269|PubMed:24204311, ECO:0000269|PubMed:27532821}. |
3D-structure;Acetylation;Activator;Alternative splicing;Autocatalytic cleavage;Coiled coil;Cytoplasm;Differentiation;DNA-binding;Endoplasmic reticulum;Glycoprotein;Hydrolase;Membrane;Nucleus;Protease;Reference proteome;Transcription;Transcription regulation;Transmembrane;Transmembrane helix |
|
|
mmu:225908; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; endoplasmic reticulum membrane [GO:0005789]; Golgi apparatus [GO:0005794]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; identical protein binding [GO:0042802]; peptidase activity [GO:0008233]; sequence-specific DNA binding [GO:0043565]; central nervous system myelin maintenance [GO:0032286]; central nervous system myelination [GO:0022010]; oligodendrocyte development [GO:0014003]; oligodendrocyte differentiation [GO:0048709]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of myelination [GO:0031643]; protein autoprocessing [GO:0016540]; regulation of transcription by RNA polymerase II [GO:0006357] |
19596243_Study identifies gene model 98, which we have named myelin gene regulatory factor (MRF), as a transcriptional regulator required for CNS myelination. 21938520_NPC1 protein was expressed in oligodendrocytes and the amount of myelin protein was generally decreased in all parts of the brain and spinal cord in NPC1-mutant mice 22956843_Expression of MRF within the adult central nervous system (CNS) is critical to maintain oligodendrocyte identity and integrity of CNS myelin. 24204311_Here we identify Myrf as an oligodendrocyte-specific target of Sox10 and map a Sox10 responsive enhancer to an evolutionarily conserved element in intron 1 of the Myrf gene 25789755_mutant huntingtin binds abnormally to MYRF and affects its transcription activity 28160598_molecular mechanisms underlying the homo-trimerization of Myrf N-terminal fragments and the homo-trimeric DNA binding of Myrf N-terminal fragments 28695847_Purification, crystallization and X-ray analysis of DNA-binding domain of MRF has been reported. 29472293_results thus suggest that Fbxw7 controls the transcription of MyRF target genes in various tissues through regulation of MyRF protein stability in a manner dependent on MyRF phosphorylation by GSK-3. 30166609_this study identifies the N-terminal-most domain as the transactivation domain of Myrf. 30249802_TMEM98 membrane protein physically interacts with MYRF in the endoplasmic reticulum and subsequently blocks its self-cleavage. 32236127_findings highlight the importance of the interplay between TMEM98 and MYRF in determining the size of the eye 33646289_Nanophthalmos-Associated MYRF Gene Mutation Causes Ciliary Zonule Defects in Mice. 36730342_DUSP15 expression is reduced in the hippocampus of Myrf knock-out mice but attention and object recognition memory remain intact. |
ENSG00000124920 |
MYRF |
40.093250 |
12.0600564333 |
3.592165 |
0.58567775 |
36.682296 |
0.00000000139036368281611861007767058098175821934106011212861631065607070922851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000192702635274386888015995253960882527266562647128012031316757202148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
69.769774199332 |
29.108215371345 |
5.78519467013446 |
2.0198265653306 |
| ENSMUSG00000036139 |
15427 |
Hoxc9 |
protein_coding |
P09633 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15427; |
aggresome [GO:0016235]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription repressor complex [GO:0017053]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; anterior/posterior pattern specification [GO:0009952]; embryonic skeletal system development [GO:0048706]; embryonic skeletal system morphogenesis [GO:0048704]; proximal/distal pattern formation [GO:0009954]; regulation of transcription by RNA polymerase II [GO:0006357] |
20826310_Hoxc9, has an essential role in organizing the motor system through global repressive activities. Hoxc9 is required for the generation of thoracic motor columns, and in its absence, neurons acquire the fates of limb-innervating populations 33028607_Differential abilities to engage inaccessible chromatin diversify vertebrate Hox binding patterns. |
ENSG00000180806 |
HOXC9 |
99.258658 |
0.4587090442 |
-1.124349 |
0.26013877 |
18.479042 |
0.00001717829652259656044409330544198155621415935456752777099609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001219859266350539853682524715772217405174160376191139221191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
63.5152145394939 |
11.514873136994 |
138.465145492522 |
17.9463272108038 |
| ENSMUSG00000036168 |
237465 |
Ccdc38 |
protein_coding |
Q8CDN8 |
|
Alternative splicing;Coiled coil;Cytoplasm;Cytoskeleton;Reference proteome |
|
|
mmu:237465; |
centrosome [GO:0005813]; cytoplasm [GO:0005737] |
27278724_these results suggest that Ccdc38 is a testes-specific gene, which may be important for mouse spermatogenesis. 35587122_CCDC38 is required for sperm flagellum biogenesis and male fertility in mice. |
ENSG00000165972 |
CCDC38 |
85.785941 |
2.7161536248 |
1.441565 |
0.36256243 |
14.890162 |
0.00011395574929336459407257925446899093913089018315076828002929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006717391537293070351982704480064967356156557798385620117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
123.68675849124 |
31.4118332821082 |
45.5374678965882 |
8.88536623734376 |
| ENSMUSG00000036169 |
66042 |
Sostdc1 |
protein_coding |
Q9CQN4 |
FUNCTION: May be involved in the onset of endometrial receptivity for implantation/sensitization for the decidual cell reaction. Enhances Wnt signaling and inhibits TGF-beta signaling (By similarity). Directly antagonizes activity of BMP2, BMP4, BMP6 and BMP7 in a dose-dependent manner. {ECO:0000250, ECO:0000269|PubMed:14623234}. |
Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal;Wnt signaling pathway |
|
|
mmu:66042; |
extracellular space [GO:0005615]; BMP binding [GO:0036122]; BMP receptor activity [GO:0098821]; BMP signaling pathway [GO:0030509]; canonical Wnt signaling pathway [GO:0060070]; cell fate commitment [GO:0045165]; epithelial cell fate commitment [GO:0072148]; hair follicle morphogenesis [GO:0031069]; mammary gland bud morphogenesis [GO:0060648]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cell fate commitment [GO:0010454]; negative regulation of determination of dorsal identity [GO:2000016]; negative regulation of myoblast differentiation [GO:0045662]; negative regulation of Wnt signaling pathway [GO:0030178]; odontogenesis of dentin-containing tooth [GO:0042475]; pattern specification process [GO:0007389] |
14623234_Data suggest that ectodin is a novel bone morphogenetic protein (BMP) inhibitor which integrates BMP signaling with the SHH and FGF signal pathways 16179481_reported that ectodin, a secreted bone morphogenetic protein (BMP) inhibitor, is expressed as a 'negative' image of enamel knots; proposed that ectodin is critical for robust spatial delineation of enamel knots and cusps 16341262_Uterine sensitization-associated gene-1 (USAG-1) plays a critical role in the modulation of renoprotective action of bone morphogenetic protein. 17389776_Expression of USAG-1 mRNA appears to be associated with blastocyst implantation to the luminal epithelium, suggesting that physiological or biochemical contact of the blastocyst to the uterus is required for USAG-1 expression 17555714_USAG-1 controls the number of teeth in the maxillary incisor region by regulating apoptosis 18329379_Enhanced BMP signaling results in supernumerary teeth and BMP signaling was modulated by Wnt signaling in the USAG-1 deficient mouse model. 19141669_minimizing the amount of dental mesenchyme in cultured Sostdc1-deficient incisors causes the formation of additional de novo incisors that resemble the successional incisor development that results from activated Wnt signaling. 20197625_the pathogenetic role of USAG-1 in Col4a3-/- mice might involve crosstalk between kidney tubules and the glomerulus and that inhibition of USAG-1 may be a promising therapeutic approach for the treatment of Alport syndrome. 20724449_analyses demonstrate that the Fgf and Shh pathways are major downstream targets of Wise-regulated Wnt signaling. 21447550_We propose a new reaction-diffusion model in which Wnt, Shh and Sostdc1 act as the activator, mediator and inhibitor, respectively, and confirm that such interactions can generate the tooth pattern of a wild-type mouse. 22509524_The data suggested that functions of Sostdc1 can be largely attributed to its ability to attenuate Wnt/beta-catenin signaling. 22525429_The data demonstrate that simvastatin contributes to prevent the progression of renal fibrosis by upregulating BMP-7-mediated anti-fibrotic signaling and that one aspect of crucial efficacies is achieved by regulating HOXA13 and USAG-1. 22829579_Data suggest that Sostdc1 primarily regulates bone morphogenetic protein pathway in pancreatic islets; knockout/mutation of Sostdc1 enhances down-regulation of Ctgf (connective tissue growth factor) and gremlin in islets after high-fat diet. 23293290_Wise controls the number and distribution of the mammary epithelial cells via inhibition of Wnt/beta-catenin signaling. 23994639_Sost and its paralog Sostdc1 coordinate digit number in a Gli3-dependent manner. 24789067_Findings strongly suggest that Wise and Sost are key modulators of bone development through the ability of their encoded proteins to interact with Lrp5 and control the balance or levels of Wnt signaling. 24816837_Interactions between BMP-7 and USAG-1 (uterine sensitization-associated gene-1) regulate supernumerary organ formations 27102547_Sostdc1 may promote and maintain mesenchymal stem cell quiescence in the periosteum. 27178071_the in vivo inter-relationships between Bmp7 and Usag-1, was examined. 27518316_suggest that RUNX2 and USAG-1 act in an antagonistic manner 30235284_Wise and Lrp4 are thus likely to control palatal rugae development by regulating reaction-diffusion mechanisms through Shh and Fgf signaling. 30776499_sclerostin domain containing 1 (Sostdc1) is expressed at low levels in multiple myeloma and osteoblast lineage cells when these cells are grown separately in cell culture but its expression is significantly induced in both cell types when these cells are in contact. 30814306_these data support a role for Sostdc1 in the regulation of NK cell maturation and cytotoxicity, and identify potential NK cell niches. 32820125_SOSTDC1-producing follicular helper T cells promote regulatory follicular T cell differentiation. 34211084_Local application of Usag-1 siRNA can promote tooth regeneration in Runx2-deficient mice. |
ENSG00000171243 |
SOSTDC1 |
40.628519 |
0.0295385237 |
-5.081258 |
0.68387599 |
71.317651 |
0.00000000000000003041031466271950884786478915812611666614345356878489068375159831703058443963527679443359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000009550719854485433465458154787748305446968649602343393034686869214056059718132019042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.30337825437915 |
1.13321520767558 |
77.9787871861883 |
17.8073326072959 |
| ENSMUSG00000036196 |
224661 |
Slc26a8 |
protein_coding |
Q8R0C3 |
FUNCTION: Acts as a DIDS-sensitive anion exchanger mediating chloride, sulfate and oxalate transport. May fulfill critical anion exchange functions in male germ line during meiosis and hence may play a role in spermatogenesis. May be involved in a new regulatory pathway linking sulfate transport to RhoGTPase signaling in male germ cells. A critical component of the sperm annulus that is essential for correct sperm tail differentiation and motility and hence male fertility. May form a molecular complex involved in the regulation of chloride and bicarbonate ions fluxes during sperm capacitation (By similarity). {ECO:0000250, ECO:0000269|PubMed:17517695}. |
Alternative splicing;Anion exchange;Developmental protein;Differentiation;Glycoprotein;Ion transport;Meiosis;Membrane;Reference proteome;Spermatogenesis;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:224661; |
plasma membrane [GO:0005886]; bicarbonate transmembrane transporter activity [GO:0015106]; chloride channel activity [GO:0005254]; chloride transmembrane transporter activity [GO:0015108]; oxalate transmembrane transporter activity [GO:0019531]; sulfate transmembrane transporter activity [GO:0015116]; anion transport [GO:0006820]; cell differentiation [GO:0030154]; chloride transport [GO:0006821]; meiotic cell cycle [GO:0051321]; oxalate transport [GO:0019532]; spermatogenesis [GO:0007283]; sulfate transport [GO:0008272] |
17517695_Tat1 (Slc26A8) is a critical component of the sperm annulus that is essential for proper sperm tail differentiation and motility 35181959_The heterozygous mutations of SLC26A8 are not the main actors for male infertility. |
ENSG00000112053 |
SLC26A8 |
28.893077 |
3.5696320825 |
1.835775 |
0.55798604 |
10.557975 |
0.00115687849965557328253684676866441805032081902027130126953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0052586014045818236725682304211204609600827097892761230468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
45.0861198816693 |
17.8758102940631 |
12.6304669051151 |
3.83153325437223 |
| ENSMUSG00000036206 |
98402 |
Sh3bp4 |
protein_coding |
Q921I6 |
FUNCTION: May function in transferrin receptor internalization at the plasma membrane through a cargo-specific control of clathrin-mediated endocytosis. Alternatively, may act as a negative regulator of the amino acid-induced TOR signaling by inhibiting the formation of active Rag GTPase complexes. Preferentially binds inactive Rag GTPase complexes and prevents their interaction with the mTORC1 complex inhibiting its relocalization to lysosomes and its activation. Thereby, may indirectly regulate cell growth, proliferation and autophagy (By similarity). {ECO:0000250}. |
Coated pit;Cytoplasmic vesicle;Endocytosis;Membrane;Nucleus;Phosphoprotein;Reference proteome;Repeat;SH3 domain |
|
|
mmu:98402; |
clathrin-coated pit [GO:0005905]; clathrin-coated vesicle [GO:0030136]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; GDP-dissociation inhibitor activity [GO:0005092]; identical protein binding [GO:0042802]; small GTPase binding [GO:0031267]; cellular response to amino acid stimulus [GO:0071230]; endocytosis [GO:0006897]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of GTPase activity [GO:0034260]; negative regulation of TOR signaling [GO:0032007]; positive regulation of autophagy [GO:0010508]; protein localization to lysosome [GO:0061462]; regulation of catalytic activity [GO:0050790] |
19122209_TTP Ser(245) is the principal target for the modulation of this protein via the AKT signalling cascade. 22575674_SH3BP4 is a negative regulator of the Rag GTPase complex and amino acid-dependent mTORC1 signaling |
ENSG00000130147 |
SH3BP4 |
702.849849 |
0.4424174162 |
-1.176520 |
0.11556519 |
102.237312 |
0.00000000000000000000000492534179346303694942631551018986487784313913031091651686708363664171650420087189559126272797584533691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000002543145972814541954351835797790307855735778140480792460382522199302535170772898709401488304138183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
425.912312502885 |
34.1536429111532 |
962.693368040354 |
56.0054138446459 |
| ENSMUSG00000036298 |
239606 |
Slc2a13 |
protein_coding |
Q3UHK1 |
FUNCTION: H(+)-myo-inositol cotransporter. Can also transport related stereoisomers. {ECO:0000250|UniProtKB:Q96QE2}. |
Cell membrane;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:239606; |
apical plasma membrane [GO:0016324]; astrocyte end-foot [GO:0097450]; cell body [GO:0044297]; cell periphery [GO:0071944]; cell projection [GO:0042995]; cytoplasm [GO:0005737]; growth cone [GO:0030426]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; organelle membrane [GO:0031090]; plasma membrane [GO:0005886]; ATPase binding [GO:0051117]; myo-inositol transmembrane transporter activity [GO:0005365]; myo-inositol:proton symporter activity [GO:0005366]; protease binding [GO:0002020]; transmembrane transporter activity [GO:0022857]; myo-inositol transport [GO:0015798]; positive regulation of amyloid-beta formation [GO:1902004]; transmembrane transport [GO:0055085] |
19607714_Inositol transport and homeostasis were unaffected in H+/myo-inositol transporter targeted null-mutant mice. 22564531_astrocytes obtained directly from the brain express HMIT and is expressed in neurons 26094765_SLC2A13 is a novel gamma-secretase associated protein that regulates amyloid beta production without affecting Notch cleavage. |
ENSG00000151229 |
SLC2A13 |
41.073494 |
4.7221225446 |
2.239435 |
0.43435379 |
26.415103 |
0.00000027537890561210795557683440361318449873806457617320120334625244140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000026751093688033342138452973929529932206605735700577497482299804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
67.5326598078133 |
20.6406912591115 |
14.3013357171934 |
3.53324775586683 |
| ENSMUSG00000036381 |
140795 |
P2ry14 |
protein_coding |
Q9ESG6 |
FUNCTION: Receptor for UDP-glucose coupled to G-proteins. {ECO:0000250}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:140795; |
plasma membrane [GO:0005886]; G protein-coupled purinergic nucleotide receptor activity [GO:0045028]; G protein-coupled receptor signaling pathway [GO:0007186]; hematopoietic stem cell homeostasis [GO:0061484]; immune response [GO:0006955] |
19164486_These results demonstrate a novel function of the P2Y14 receptor in the rodent stomach and an ability of UDP-glucose to delay gastric emptying without involving the P2Y14 receptor. 19454705_UDP-glucose and its receptor P2RY14 are key front line players able to trigger innate mucosal immune responses in the FRT bypassing the recognition of pathogen-associated molecular patterns 20398327_Data detected mRNA and protein for ADP receptors P2Y12 and P2Y13 in mouse dorsal root ganglia (DRG). P2Y14, a homologous Gi-coupled nucleotide receptor, is also expressed in DRG. 22778393_These studies indicate that GPR105 ablation mitigates high fat diet-induced insulin resistance by inhibiting macrophage recruitment and tissue inflammation 23828566_P2Y14 is a receptor governing in utero tolerance to genotoxic stress. 24048691_Results suggest that P2Y14 may act as the receptor for UDP-sugars in osteoclast precusors and may regulate RANKL-induced osteoclastogenesis. 24911208_the physiological importance of P2Y14 receptors in the vasculature 24937426_P2Y14 on stem/progenitor cells of the hematopoietic system inhibits cell senescence by monitoring and responding to the extracellular manifestations of tissue stress 24993824_Insulin secretion tests revealed a reduced insulin release from P2Y14-deficient islets, highlighting P2Y14 as a new modulator of proper insulin secretion 28804125_Present study suggests that P2Y14 expression could be used as a phenotypic marker to further dissect placental HSPCs. 29189936_P2Y14 receptor activation decreases interleukin-6 production and glioma GL261 cell proliferation in microglial transwell cultures. These data suggest that P2Y14 receptors may be a key a receptor involved in glioma cell-microglial/macrophage communication in the tumor environment. 32287042_Proinflammatory P2Y14 receptor inhibition protects against ischemic acute kidney injury in mice. 32326617_Role of UDP-Sugar Receptor P2Y14 in Murine Osteoblasts. 33792561_UDP-glucose and P2Y14 receptor amplify allergen-induced airway eosinophilia. 35385339_The purinergic P2Y14 receptor links hepatocyte death to hepatic stellate cell activation and fibrogenesis in the liver. 35432308_GPR105-Targeted Therapy Promotes Gout Resolution as a Switch Between NETosis and Apoptosis of Neutrophils. |
ENSG00000174944 |
P2RY14 |
10.192458 |
0.1489534681 |
-2.747066 |
0.90773128 |
9.249216 |
0.00235596221857457474579966572036937577649950981140136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0097970290888849051458286965043953387066721916198730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.6136852500885 |
1.75518147853298 |
17.5469915756425 |
7.1424260485542 |
| ENSMUSG00000036523 |
268527 |
Greb1 |
protein_coding |
Q3UHK3 |
FUNCTION: May play a role in estrogen-stimulated cell proliferation. {ECO:0000250}. |
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:268527; |
membrane [GO:0016020] |
28911214_GREB1 is a novel progesterone-responsive gene required for endometrial stromal decidualization. 29100091_A significant association between GREB1L variants and BKA. 29955957_Craniofacial Greb1l RNA expression peaks in mice during this time frame (E8.5). It also peaks in the developing inner ear during E13-E16, after which it decreases in adulthood. The crucial function of Greb1l in craniofacial development is also evidenced in knockout mice, which develop severe craniofacial abnormalities 33731348_GREB1: An evolutionarily conserved protein with a glycosyltransferase domain links ERalpha glycosylation and stability to cancer. |
ENSG00000196208 |
GREB1 |
60.011298 |
0.2053840294 |
-2.283604 |
0.67585055 |
9.916844 |
0.00163772955925828734430327315862996329087764024734497070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0071580765126606129417075052856489492114633321762084960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.5378509818109 |
9.9770340165582 |
99.9973125427826 |
34.9747915042162 |
| ENSMUSG00000036560 |
243914 |
Lgi4 |
protein_coding |
Q8K1S1 |
FUNCTION: Component of Schwann cell signaling pathway(s) that controls axon segregation and myelin formation. {ECO:0000269|PubMed:16341215}. |
Glycoprotein;Leucine-rich repeat;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:243914; |
extracellular space [GO:0005615]; adult locomotory behavior [GO:0008344]; glial cell development [GO:0021782]; glial cell proliferation [GO:0014009]; gliogenesis [GO:0042063]; myelination [GO:0042552]; myelination in peripheral nervous system [GO:0022011]; neuron maturation [GO:0042551]; regulation of myelination [GO:0031641]; Schwann cell development [GO:0014044] |
14694902_Fxyd3 and Lgi4 transcripts potentially form double-stranded RNA molecules in many cell types in vivo, which may impact on their respective expression 16341215_the abnormalities observed in claw paw mice are attributable to the loss of Lgi4 function 19833108_lGI4 distribution in the vertical limb of the diagonal band and in putative hippocampal interneurons suggests that the function of these genes might be related to the generation of hippocampal theta rhythm. 21068328_Our results identify a new mechanism regulating enteric gliogenesis as well as novel functions for Lgi4 regulating the proliferation and maturation of glial lineage cells throughout the peripheral nervous system |
ENSG00000153902 |
LGI4 |
130.757925 |
0.4316612251 |
-1.212029 |
0.23022444 |
27.455339 |
0.00000016076444737421913262015395109510995652613019046839326620101928710937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000016346889602163588645143514621604907688379171304404735565185546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
80.7876363198899 |
12.9468313255261 |
187.155184711808 |
21.6413783682025 |
| ENSMUSG00000036585 |
14164 |
Fgf1 |
protein_coding |
P61148 |
FUNCTION: Plays an important role in the regulation of cell survival, cell division, angiogenesis, cell differentiation and cell migration. Functions as potent mitogen in vitro. Acts as a ligand for FGFR1 and integrins. Binds to FGFR1 in the presence of heparin leading to FGFR1 dimerization and activation via sequential autophosphorylation on tyrosine residues which act as docking sites for interacting proteins, leading to the activation of several signaling cascades. Binds to integrin ITGAV:ITGB3. Its binding to integrin, subsequent ternary complex formation with integrin and FGFR1, and the recruitment of PTPN11 to the complex are essential for FGF1 signaling. Induces the phosphorylation and activation of FGFR1, FRS2, MAPK3/ERK1, MAPK1/ERK2 and AKT1. Can induce angiogenesis. {ECO:0000250|UniProtKB:P05230}. |
Acetylation;Angiogenesis;Cytoplasm;Developmental protein;Differentiation;Growth factor;Heparin-binding;Mitogen;Nucleus;Phosphoprotein;Reference proteome;Secreted |
|
|
mmu:14164; |
cell cortex [GO:0005938]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; fibroblast growth factor receptor binding [GO:0005104]; growth factor activity [GO:0008083]; heparin binding [GO:0008201]; Hsp70 protein binding [GO:0030544]; integrin binding [GO:0005178]; S100 protein binding [GO:0044548]; activation of protein kinase B activity [GO:0032148]; angiogenesis [GO:0001525]; animal organ morphogenesis [GO:0009887]; branch elongation involved in ureteric bud branching [GO:0060681]; cardiac muscle cell proliferation [GO:0060038]; cell differentiation [GO:0030154]; cellular response to heat [GO:0034605]; epithelial cell proliferation [GO:0050673]; fibroblast growth factor receptor signaling pathway [GO:0008543]; lung development [GO:0030324]; mesonephric epithelium development [GO:0072163]; organ induction [GO:0001759]; positive regulation of angiogenesis [GO:0045766]; positive regulation of cell division [GO:0051781]; positive regulation of cell migration [GO:0030335]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cholesterol biosynthetic process [GO:0045542]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of endothelial cell migration [GO:0010595]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of hepatocyte proliferation [GO:2000347]; positive regulation of intracellular signal transduction [GO:1902533]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of sprouting angiogenesis [GO:1903672]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of cell migration [GO:0030334]; regulation of endothelial cell chemotaxis to fibroblast growth factor [GO:2000544]; regulation of endothelial tube morphogenesis [GO:1901509]; vasculogenesis involved in coronary vascular morphogenesis [GO:0060979]; wound healing [GO:0042060] |
12095987_the neuroprotective effects of FGF1 involve inactivation of GSK3beta 12121735_FGF-1, FGF-2 and FGF receptor-1 levels in the cochlear nucleus following acoustic overstimulation 12128203_the depletion of FGF1 may be a key regulatory component in initial phase of branching morphogenesis of the lung bud epithelium in vitro 14507672_Agonist role for FGF1 and FGF2 in specifically insult-induced liver matrix deposition and hepatic fibrogenesis and a potential target for the prevention of hepatic fibrosis. 14559220_non-classical release is mediated by synaptotagmin 1 14592461_High steady-state levels of ONOO(-) may induce cysteine oxidation, tyrosine nitration, and non-nreversible inactivation of FGF-1, a inhibitory feedback mechanism restoring cellular homeostatis during resolution of inflammation and repair. 15199124_Gab1 is essential for FGF1 stimulation of both PI 3-kinase and the antiapoptotic protein kinase Akt, while FGF1-induced MAPK stimulation is not affected by Gab1 deficiency. 15314170_Data show that mouse and human fibroblast growth factor 1 (FGF-1) internal ribosome entry sites show similar activity profiles, and have a conserved structural domain at both the nucleotide sequence and RNA structure levels. 15574884_Phosphorylation of Fgf1 occurs in the nucleus by pkcdelta. It is then exported to the cytosol. 15607950_These results thus suggest that in addition to vesicular trafficking, DCX may play a role in the regulation of cell adhesion via its interaction with DFFRX in migrating and differentiating neurons. 16439359_certain CpG sites within the Fgf-1 gene locus are preferentially methylated by Dnmt3a but not by Dnmt3b 16495214_Hsp90 is required for translocation of FGF-1 and FGF-2 across the endosomal membrane 16728399_FGF7- and FGF1-induced mitogenesis and downstream signaling is controlled by distinct heparin octasaccharide motifs 16930531_FGF1 and p40 synaptotagmin 1 release correlates with membrane destabilizing ability 17027650_These results identify a novel aspect of the crosstalk between FGF and thrombin signaling pathways which both play important roles in tissue repair and angiogenesis. 17481602_Shisa is antagonistic to Wnt and Fgf signalings 17548887_In apoE-deficient mouse astrocytes, FGF-1 stimulated cholesterol biosynthesis without enhancing its release, indicating a signaling pathway independent of apoE biosynthesis upregulation. 17616529_Formation of the FGF-1-exportin-1-Ran-GTP complex in vitro as well as nuclear export of FGF-1 in vivo was dependent on phosphorylation of FGF-1, and it was abolished by leptomycin B. 17643421_sphingosine kinase 1 is a component of the copper-dependent FGF1 release pathway. 17660197_A conserved, specific and stage-dependent requirement for Erk1/2 signalling downstream of FGF-induced neural specification in higher vertebrates. 17852407_FGF and PDGF have roles in cell proliferation and migration in osteoblastic cells 17963525_analysis of expression of bicistronic vector driven by the FGF-1 IRES in mouse muscle 18281280_6-O-sulfate in HS may regulate the signalings of some of HB-GFs, including FGF-1, FGF-2 and FGF-4, by inducing different interactions between ligands and their receptors 18694942_and fibroblast growth factor signaling interact to control the proximal-distal pattern of forming airways in the mammalian lung 18784255_These data demonstrate the existence of novel cross-talk between thrombin, FGF, and Notch signaling pathways, which play important roles in vascular formation and remodeling. 19168680_An interplay between retinoic acid, Fgf and Shh signalling is likely to be an important mechanism underpinning the tight regulation of caudal embryonic development. 19408015_A detailed double-label immunohistochemical investigation of the localization patterns of FGF1 and its receptors FGFR1 and FGFR2 in adult and early postnatal mouse retinas, is reported. 19561198_Results revealed a mechanism of molecular coupling of mRNA transcription and translation, involving a unique process of IRES activation by a FGF1 promoter element. 19574212_that the main role of heparin in FGF-induced signaling is to protect this naturally unstable protein against heat and/or proteolytic degradation and heparin is not essential for a direct FGF1-FGFR interaction and receptor activation 20189986_RFX1 may negatively regulate the self-renewal of GBM-SCs through modulating FGF-1B and FGF1 expression levels by binding the 18-bp cis-elements of the F1B promoter. 21223966_The results demonstrate that the FGF1/FGFR1 complex constitutes a signalling module that independently of the receptor tyrosine kinase can convey a signal that initiates a strictly timed and periodic release of endocytosed FGF1 into the cytosol/nucleus. 22396766_RA via RALDH2 has separable functions in the developing spinal cord to (i) maintain high levels of FGF and Notch signaling and (ii) drive stem cell differentiation, thus restricting both the numbers and the pluripotent character of neural stem cells 22522926_discovery of a phenotype for the FGF1 knockout mouse establishes the PPARgamma-FGF1 axis as critical for maintaining metabolic homeostasis and insulin sensitization 22606265_Although FGF1 transgenic mice had a normal phenotype with unperturbed kidney structure, they showed a severely inhibited kidney repair after unilateral ischemia/reperfusion. This was manifested by a strong decrease of postischemic kidney size and weight. 24595027_Nucleolin-FGF1 interaction is critical for the intranuclear phosphorylation of FGF1 by PKCdelta and thereby the regulation of nuclear export of FGF1. 25043058_parenteral delivery of a single dose of recombinant FGF1 (rFGF1) results in potent, insulin-dependent lowering of glucose levels in diabetic mice that is dose-dependent but does not lead to hypoglycaemia 25589163_Study presents a transcript profiling of remyelinated multiple sclerosis lesions and identified FGF1 as a promoter of remyelination 25685806_These results suggest that FGFs promote hair growth by inducing the anagen phase in resting hair follicles and might be a potential hair growth-promoting agent. 26847131_Data suggest that Fgf1-mediated signaling represents an important signaling cascade related to adipogenesis and visceral adiposity; expression of Fgf1 (fibroblast growth factor 1) and Fgfr1 (fibroblast growth factor receptor 1) is up-regulated in adipose tissue of obese mice (both obese mice due to high-fat diet and obese mice due to genetic deletion of leptin). 26881702_The study supports a pro-adipogenic role for betaKlotho in skeletal muscle fibro/adipogenesis and calls for further research on involvement of the FGF-FGFR-betaKlotho axis in the fibro/adipogenic infiltration associated with functional deterioration of skeletal muscle in aging and muscular dystrophy. 27443835_Fibroblast growth factor (FGF1 and FGF2), but not vascular endothelial growth factor (VEGF) rescued Psen1-/- cells from serum starvation induced apoptosis.In the absence of serum, FGF2 immunoreactivity was distributed diffusely in cytoplasmic and nuclear vesicles of wt and Psen1-/- cells, as levels of FGF2 in nuclear and cytosolic fractions were not significantly different. 27638903_fibroblast growth factor 1 (FGF1) to be synergistically induced by heat shock and wounding. 28031461_clearly demonstrate the different specificity of FGF12-FGFR1c2 and FGF22-FGFR1c2 for well defined HS structures and suggest that it is now possible to chemoenzymatically synthesize precise HS polysaccharides that can selectively mediate growth factor signaling 28076781_the HDAC3-N-CoR corepressor complex leaves the Fgf1b promoter and a complex involving the translocated CRTC1, phosphorylated CREB, and histone acetyltransferase CBP induces transient transcription. 28502041_glucose administration increased phosphorylated Akt, phosphorylated CREB, exon 1- and exon 4-specific BDNF transcripts, and FGF1 transcripts that are associated with the epigenetic changes. 28655615_lncRNA-Map2k4 is the target gene of miR-199a, and its down-regulation promotes miR-199a expression in neurons. miR-199a targeted regulation of FGF1 expression in neurons. 28813681_Suboptimal FGFR activation by a weak FGF1-FGFR dimer is sufficient to evoke a metabolic response, whereas full FGFR activation by stable and sustained dimerization is required to elicit a mitogenic response. 29048426_FGF1 protects neuroblastoma cells from p53-dependent apoptosis through an intracrine pathway regulated by FGF1 phosphorylation. 29268138_TUG1 knockdown ameliorates atherosclerosis by modulating FGF1 via miR-133a in ApoE knockout mice. 29360039_Here, we show that the Crk family of adaptor proteins, Crk and Crkl, are required for mouse lens morphogenesis but not differentiation upon Fibroblast Growth Factor stimulation. 29845277_The present results provided evidence that aFGF accelerates the progression of atherosclerosis and suggested that aFGF may be a potential therapeutic target for the prevention of atherosclerosis development. 30063763_miR-18a participates in the regulation of proliferation by partly decreasing the expression of Fgf1. 30320493_We conclude that FGF1-associated glucose decreases and subsequent reduction of cellular stress is the another potential molecule mechanism underlying FGF1 treatment for Diabetic nephropathy (DN) . 30972920_FGF1-Tek mice display increased locomotor activity that likely promotes the utilization of dietary fatty acids before they can accumulate in adipose and liver 32817243_Persistence of Fear Memory Depends on a Delayed Elevation of BAF53b and FGF1 Expression in the Lateral Amygdala. 33020467_miR-188-3p Inhibits Vascular Smooth Muscle Cell Proliferation and Migration by Targeting Fibroblast Growth Factor 1 (FGF1). 33762571_FGF1(DeltaHBS) prevents diabetic cardiomyopathy by maintaining mitochondrial homeostasis and reducing oxidative stress via AMPK/Nur77 suppression. 33994863_MicroRNA-370 carried by M2 macrophage-derived exosomes alleviates asthma progression through inhibiting the FGF1/MAPK/STAT1 axis. 34358309_MicroRNA-27b-3p down-regulates FGF1 and aggravates pathological cardiac remodelling. 34831221_Regulation of Transplanted Cell Homing by FGF1 and PDGFB after Doxorubicin Myocardial Injury. 36161959_Autocrine FGF1 signaling promotes glucose uptake in adipocytes. 36225175_nFGF1 Protects beta-Cells against High Glucose-Induced Apoptosis via the AMPK/SIRT1/PGC-1alpha Axis. 36625595_Novel Muscle-Homing Peptide FGF1 Conjugate Based on AlphaFold for Type 2 Diabetes Mellitus. |
ENSG00000113578 |
FGF1 |
229.425477 |
0.0187748219 |
-5.735057 |
0.35632973 |
306.732897 |
0.00000000000000000000000000000000000000000000000000000000000000000001124483464797368769637452195558494428470006966226702547046732379289026463078747150250452824045717621614529950660884594494290716458416455833849335361023454286636237561629834968979224640861502848566 |
0.0000000000000000000000000000000000000000000000000000000000000000030049354624199157649841076338878084690346833483048947001219938900054059563997054635803409170030910040374410843231666541078572614083295643809632936253470578399834287411374056375734653556719422340 |
Yes |
No |
8.47599359388311 |
2.11482665757567 |
451.455338855591 |
56.2668107812536 |
| ENSMUSG00000036661 |
105841 |
Dennd3 |
protein_coding |
A2RT67 |
FUNCTION: Guanine nucleotide exchange factor (GEF) activating Rab12. Promotes the exchange of GDP to GTP, converting inactive GDP-bound Rab12 into its active GTP-bound form. Regulates autophagy in response to starvation through Rab12 activation (PubMed:24719330, PubMed:25925668, PubMed:28249939). Starvation leads to ULK1/2-dependent phosphorylation of Ser-554 and Ser-572, which in turn allows recruitment of 14-3-3 adapter proteins and leads to up-regulation of GEF activity towards Rab12 (PubMed:25925668). Also plays a role in protein transport from recycling endosomes to lysosomes, regulating, for instance, the degradation of the transferrin receptor and of the amino acid transporter PAT4 (PubMed:21718402, PubMed:24719330). Starvation also induces phosphorylation at Tyr-940, which leads to up-regulated GEF activity and initiates autophagy (PubMed:28249939). {ECO:0000269|PubMed:21718402, ECO:0000269|PubMed:24719330, ECO:0000269|PubMed:25925668, ECO:0000269|PubMed:28249939}. |
3D-structure;Cytoplasm;Guanine-nucleotide releasing factor;Phosphoprotein;Reference proteome;Repeat;WD repeat |
|
|
mmu:105841; |
cytoplasmic vesicle [GO:0031410]; guanyl-nucleotide exchange factor activity [GO:0005085]; endosome to lysosome transport [GO:0008333]; protein catabolic process [GO:0030163]; regulation of Rab protein signal transduction [GO:0032483] |
24719330_Dennd3 not only functions as a Rab12-GEF but also modulates Akt signaling in mouse embryonic fibroblast cells |
ENSG00000105339 |
DENND3 |
39.654497 |
0.2309067592 |
-2.114618 |
0.47306922 |
18.834770 |
0.00001425447134882283437690413813170309254019230138510465621948242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001027077140895314206646068977413222000905079767107963562011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.6449161700912 |
4.88868538009925 |
63.4235057622612 |
14.9503550234958 |
| ENSMUSG00000036768 |
209737 |
Kif15 |
protein_coding |
Q6P9L6 |
FUNCTION: Plus-end directed kinesin-like motor enzyme involved in mitotic spindle assembly. {ECO:0000250}. |
Acetylation;ATP-binding;Coiled coil;Cytoplasm;Cytoskeleton;Microtubule;Motor protein;Nucleotide-binding;Phosphoprotein;Reference proteome |
|
|
mmu:209737; |
cytoplasm [GO:0005737]; kinesin complex [GO:0005871]; microtubule [GO:0005874]; spindle [GO:0005819]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule-based movement [GO:0007018]; regulation of mitotic spindle assembly [GO:1901673] |
14618103_Kif15 opposes the capacity of other motors to generate independent microtubule movements within key regions of developing neurons. 35801464_Kinesin family member 15 can promote the proliferation of glioblastoma. 35835966_Kinesin KIF15 regulates tubulin acetylation and spindle assembly checkpoint in mouse oocyte meiosis. |
ENSG00000163808 |
KIF15 |
807.245522 |
2.2218917505 |
1.151789 |
0.13548526 |
71.384819 |
0.00000000000000002939248804314457271026741042754924686702437010960529961689502442823140881955623626708984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000009250131774239218962543579218954995513713113414536182155245569447288289666175842285156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1113.54950104262 |
104.728577945051 |
501.171805860502 |
35.8357087355842 |
| ENSMUSG00000036826 |
101883 |
Igflr1 |
protein_coding |
Q3U4N7 |
FUNCTION: Probable cell membrane receptor for the IGF-like family protein IGFL. {ECO:0000269|PubMed:21454693}. |
Alternative splicing;Cell membrane;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:101883; |
nucleoplasm [GO:0005654]; plasma membrane [GO:0005886] |
21454693_Murine insulin growth factor-like (IGFL) and human IGFL1 proteins are induced in inflammatory skin conditions and bind to a novel tumor necrosis factor receptor family member, IGFLR1. |
ENSG00000126246 |
IGFLR1 |
14.715319 |
20.5084934987 |
4.358150 |
1.28751419 |
8.938658 |
0.00279197755310432249933882964398890180746093392372131347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0113044662649880479909914043901153490878641605377197265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.7448455033892 |
25.1335202900805 |
1.20656573360513 |
1.02497838730504 |
| ENSMUSG00000036854 |
243912 |
Hspb6 |
protein_coding |
Q5EBG6 |
FUNCTION: Small heat shock protein which functions as a molecular chaperone probably maintaining denatured proteins in a folding-competent state. Seems to have versatile functions in various biological processes. Plays a role in regulating muscle function such as smooth muscle vasorelaxation and cardiac myocyte contractility. May regulate myocardial angiogenesis implicating KDR. Overexpression mediates cardioprotection and angiogenesis after induced damage. Stabilizes monomeric YWHAZ thereby supporting YWHAZ chaperone-like activity. {ECO:0000250|UniProtKB:O14558}. |
Chaperone;Cytoplasm;Nucleus;Phosphoprotein;Reference proteome;Secreted;Stress response |
|
|
mmu:243912; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; nucleolus [GO:0005730]; nucleus [GO:0005634]; chaperone binding [GO:0051087]; protein folding chaperone [GO:0044183]; protein homodimerization activity [GO:0042803]; structural constituent of eye lens [GO:0005212]; unfolded protein binding [GO:0051082]; chaperone-mediated protein folding [GO:0061077]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cardiac muscle cell apoptotic process [GO:0010667]; positive regulation of angiogenesis [GO:0045766]; protein refolding [GO:0042026]; response to heat [GO:0009408] |
14615292_identification and characterization of a cardiac isoform of p20 associated with beta-adrenergic signaling in adult cardiomyocytes. 15105294_Recombinant mouse Hsp20 translocates to and interacts with actin cytoskeleton in response to isoproterenol stimulation and prevents ss-agonist-induced apoptosis in adult rat cardiomyocytes. 19380620_miR-320 is involved in the regulation of I/R-induced cardiac injury and dysfunction via antithetical regulation of Hsp20 19501592_Hsp20 expression prevents endotoxin-induced myocardial dysfunction and apoptosis via inhibition of NF-kappaB activation 19850943_Blockade of Hsp20 phosphorylation exacerbates cardiac ischemia/reperfusion injury by suppressed autophagy and increased cell death. 21493896_the activity of type 1 protein phosphatase (PP1), a known regulator of PLN signaling, was significantly reduced by Hsp20 overexpression, suggesting that the Hsp20 stimulatory effects are partially mediated through the PP1-PLN axis. 21722304_potential protein targets for the loss of PostC may include F(1)-ATPase gamma, Echs1 and Hsp20 that could regulate cellular ATP consumption/production and defense response to ischaemic stress 23713735_Increased expression of Hsp20 in neuroblastoma cells protected against ischemia-reperfusion injury. 29925002_These findings suggest a role of Hsp20 in mediating adipocyte function by linking beta-adrenergic signaling to PPARgamma activity. 30171788_Our study was designed to assess the efficacy of small heat shock proteins B1 (Hsp27) and B6 (Hsp20) as an adjuvant accompanied by HPV16 E7 and hPP10-E7 antigens in tumor mouse model. A major key for successful DNA and protein transfer into cells is the development of delivery systems with high efficiency and low cytotoxicity. 31210321_HSP20-mediated cardiomyocyte exosomes improve cardiac function in mice with myocardial infarction by activating Akt signaling pathway. 32218573_Hsp20-S10F mice treated with probenecid had decreased mortality, hypertrophy, TRPV2 expression and molecular parameters of heart failure. 33339131_The Cardioprotective PKA-Mediated Hsp20 Phosphorylation Modulates Protein Associations Regulating Cytoskeletal Dynamics. |
ENSG00000004776 |
HSPB6 |
323.928203 |
2.0527143915 |
1.037533 |
0.34363548 |
8.389201 |
0.00377456809940626865362833797234998201020061969757080078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0145702537481389483853266852975139045156538486480712890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
398.23400673419 |
95.754819872895 |
194.003612185059 |
34.8367464150306 |
| ENSMUSG00000036863 |
214804 |
Syde2 |
protein_coding |
E9PUP1 |
Human_homologues FUNCTION: GTPase activator for the Rho-type GTPases by converting them to an inactive GDP-bound state. {ECO:0000250}. |
Proteomics identification;Reference proteome |
|
|
mmu:214804; |
synaptic membrane [GO:0097060]; GTPase activator activity [GO:0005096]; cell migration [GO:0016477]; regulation of Ras protein signal transduction [GO:0046578]; signal transduction [GO:0007165] |
23791195_In synapse-defective-1A knockout mice synapses assemble in normal numbers, but there is a significant reduction in synaptic vescicle docking. |
ENSG00000097096 |
SYDE2 |
100.770261 |
0.4982313075 |
-1.005112 |
0.24968826 |
16.169496 |
0.00005791934020126402273866139669422636870876885950565338134765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003642557349073714638432175405569068971090018749237060546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.5886400618585 |
11.582162913775 |
133.650051824036 |
16.6528803240013 |
| ENSMUSG00000036904 |
14370 |
Fzd8 |
protein_coding |
Q61091 |
FUNCTION: Receptor for Wnt proteins. Component of the Wnt-Fzd-LRP5-LRP6 complex that triggers beta-catenin signaling through inducing aggregation of receptor-ligand complexes into ribosome-sized signalsomes (By similarity). The beta-catenin canonical signaling pathway leads to the activation of disheveled proteins, inhibition of GSK-3 kinase, nuclear accumulation of beta-catenin and activation of Wnt target genes. A second signaling pathway involving PKC and calcium fluxes has been seen for some family members, but it is not yet clear if it represents a distinct pathway or if it can be integrated in the canonical pathway, as PKC seems to be required for Wnt-mediated inactivation of GSK-3 kinase. Both pathways seem to involve interactions with G-proteins. May be involved in transduction and intercellular transmission of polarity information during tissue morphogenesis and/or in differentiated tissues. Coreceptor along with RYK of Wnt proteins, such as WNT1. {ECO:0000250, ECO:0000269|PubMed:10097073, ECO:0000269|PubMed:10395542, ECO:0000269|PubMed:15454084, ECO:0000269|PubMed:16543246}. |
3D-structure;Cell membrane;Developmental protein;Disulfide bond;G-protein coupled receptor;Glycoprotein;Golgi apparatus;Membrane;Receptor;Reference proteome;Signal;Transducer;Transmembrane;Transmembrane helix;Ubl conjugation;Wnt signaling pathway |
|
|
mmu:14370; |
extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; neuronal dense core vesicle [GO:0098992]; plasma membrane [GO:0005886]; Wnt-Frizzled-LRP5/6 complex [GO:1990851]; G protein-coupled receptor activity [GO:0004930]; PDZ domain binding [GO:0030165]; signaling receptor binding [GO:0005102]; ubiquitin protein ligase binding [GO:0031625]; Wnt receptor activity [GO:0042813]; Wnt-protein binding [GO:0017147]; angiogenesis [GO:0001525]; canonical Wnt signaling pathway [GO:0060070]; positive regulation of JUN kinase activity [GO:0043507]; positive regulation of protein phosphorylation [GO:0001934]; T cell differentiation in thymus [GO:0033077]; Wnt signaling pathway [GO:0016055] |
16543246_Several lines of evidence that the mouse Cristin/R-spondin family proteins function as Fzd8 and LRP6 receptor ligands and induce the canonical Wnt/beta-catenin signaling pathway, leading to TCF-dependent gene activation, are presented. 17506343_Molecular modeling of the complex between the xWNT8 protein and the CRD domain of mFZD8 18528331_Insulin-like growth-factor-binding protein-4 physically interacted with a Wnt receptor, Frizzled 8 (Frz8), and a Wnt co-receptor, low-density lipoprotein receptor-related protein 6 (LRP6), and inhibited the binding of Wnt3A to Frz8 and LRP6 20093360_analysis of the frizzled8.Wnt3a.LRP6 signaling complex reveals multiple Wnt and Dkk1 binding sites on LRP6 21343368_A major role for frizzled 4 and frizzled 8 in controlling ureteric growth in the developing kidney. 22228100_studies suggest a dose-dependent regulation of signaling by Fz5 and Fz8 in optic fissure/disc formation and progenitor expansion 22653731_study presents the structure of Xenopus Wnt8 (XWnt8) in complex with the mouse Fz8-cysteine-rich domain to a resolution of 3.25 A 22817897_Study shows Flamingo (Fmi) and Frizzled (Fz) 8, members of noncanonical Wnt signaling, both express in and functionally maintain quiescent long-term hematopoietic stem cells. 23401003_Fzd8 and beta-catenin negatively regulate osteoclast differentiation independent of osteoblasts. 24885675_Increasing amounts of soluble Frizzled8-cysteine-rich-domain protein modulated Wnt3a signaling in a biphasic manner. |
ENSG00000177283 |
FZD8 |
591.019133 |
0.3936601075 |
-1.344978 |
0.11984065 |
124.106867 |
0.00000000000000000000000000007982648191848854383680615684382787772245411047776252210924847635096413174266710699455984467931557446718215942382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000056292452434371182668517567594481111735981110927835672525911856629053447390736986122306007018778473138809204101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
338.071023745927 |
28.1149232830429 |
858.789136347463 |
51.2233282318994 |
| ENSMUSG00000036957 |
233067 |
Lrfn3 |
protein_coding |
Q8BLY3 |
FUNCTION: Cell adhesion molecule that mediates homophilic cell-cell adhesion in a Ca(2+)-independent manner. Promotes neurite outgrowth in hippocampal neurons. {ECO:0000269|PubMed:18227064, ECO:0000269|PubMed:18585462}. |
Cell adhesion;Cell membrane;Cell projection;Disulfide bond;Glycoprotein;Immunoglobulin domain;Leucine-rich repeat;Membrane;Postsynaptic cell membrane;Reference proteome;Repeat;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:233067; |
axon [GO:0030424]; cell surface [GO:0009986]; dendrite [GO:0030425]; glutamatergic synapse [GO:0098978]; postsynaptic density membrane [GO:0098839]; postsynaptic specialization membrane [GO:0099634]; presynaptic active zone membrane [GO:0048787]; regulation of presynapse assembly [GO:1905606]; regulation of synaptic membrane adhesion [GO:0099179]; synaptic membrane adhesion [GO:0099560] |
18585462_Data show that the C-terminal PDZ binding domains of synaptic adhesion-like molecules (SALMs)1-3 are required for neurite outgrowth, and that the N-termini of SALMs 2 and 4 are alsoinvolved in neurite outgrowth. 27480238_SALM4 cis-interacts with SALM3, inhibits trans-synaptic SALM3 interaction with presynaptic LAR family receptor tyrosine phosphatases and suppresses SALM3-dependent presynaptic differentiation. 34588597_SALM4 negatively regulates NMDA receptor function and fear memory consolidation. |
ENSG00000126243 |
LRFN3 |
35.573886 |
0.1568498153 |
-2.672544 |
0.51334783 |
26.798945 |
0.00000022576052540195263262134462746433261060019503929652273654937744140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000022200027907827906691897411339819967679432011209428310394287109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
9.70618319633208 |
3.52705129620016 |
61.8820186441586 |
14.762857139504 |
| ENSMUSG00000037031 |
70423 |
Tspan15 |
protein_coding |
F7BWT7 |
FUNCTION: Regulates maturation and trafficking of the transmembrane metalloprotease ADAM10 (PubMed:23035126, PubMed:26668317). Promotes ADAM10-mediated cleavage of (CDH2) (PubMed:26668317). Negatively regulates ligand-induced Notch activity probably by regulating ADAM10 activity (By similarity). {ECO:0000250|UniProtKB:O95858, ECO:0000269|PubMed:23035126, ECO:0000269|PubMed:26668317}. |
Alternative splicing;Cell membrane;Endosome;Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:70423; |
cell junction [GO:0030054]; cell surface [GO:0009986]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; late endosome membrane [GO:0031902]; nuclear body [GO:0016604]; plasma membrane [GO:0005886]; tetraspanin-enriched microdomain [GO:0097197]; enzyme binding [GO:0019899]; negative regulation of Notch signaling pathway [GO:0045746]; protein localization to plasma membrane [GO:0072659]; protein maturation [GO:0051604] |
29520422_Data show that tetraspanin15 (Tspan15) is abundantly expressed in brain, where it specifically interacts with endogenous a disintegrin and metalloproteinase 10 (ADAM10). 32111735_The tetraspanin Tspan15 is an essential subunit of an ADAM10 scissor complex. |
ENSG00000099282 |
TSPAN15 |
18.699592 |
0.2539337233 |
-1.977476 |
0.76783754 |
6.311297 |
0.01199710209157360714382623001483807456679642200469970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0391893328455606218541085183915129164233803749084472656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.9699040588489 |
3.32947083675527 |
23.5096937179393 |
9.25420508458783 |
| ENSMUSG00000037035 |
16324 |
Inhbb |
protein_coding |
Q04999 |
FUNCTION: Inhibins and activins inhibit and activate, respectively, the secretion of follitropin by the pituitary gland. Inhibins/activins are involved in regulating a number of diverse functions such as hypothalamic and pituitary hormone secretion, gonadal hormone secretion, germ cell development and maturation, erythroid differentiation, insulin secretion, nerve cell survival, embryonic axial development or bone growth, depending on their subunit composition. Inhibins appear to oppose the functions of activins. |
Cleavage on pair of basic residues;Disulfide bond;Glycoprotein;Growth factor;Hormone;Reference proteome;Secreted;Signal |
|
This gene encodes a member of the TGF-beta (transforming growth factor-beta) superfamily of proteins. The encoded preproprotein is proteolytically processed to generate a subunit of the dimeric activin and inhibin protein complexes. These complexes activate and inhibit, respectively, follicle stimulating hormone secretion from the pituitary gland. Homozygous knockout mice for this gene exhibit eyelid defects. [provided by RefSeq, Aug 2016]. |
mmu:16324; |
cell periphery [GO:0071944]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; perinuclear region of cytoplasm [GO:0048471]; cytokine activity [GO:0005125]; growth factor activity [GO:0008083]; hormone activity [GO:0005179]; protein homodimerization activity [GO:0042803]; activin receptor signaling pathway [GO:0032924]; apoptotic signaling pathway [GO:0097190]; cellular response to calcium ion [GO:0071277]; cellular response to cAMP [GO:0071320]; cellular response to cholesterol [GO:0071397]; cellular response to insulin stimulus [GO:0032869]; cellular response to interleukin-1 [GO:0071347]; cellular response to leptin stimulus [GO:0044320]; cellular response to starvation [GO:0009267]; cellular response to Thyroglobulin triiodothyronine [GO:1904017]; cellular response to thyroid hormone stimulus [GO:0097067]; eye development [GO:0001654]; fat cell differentiation [GO:0045444]; female gonad development [GO:0008585]; multicellular organism aging [GO:0010259]; negative regulation of follicle-stimulating hormone secretion [GO:0046882]; negative regulation of hepatocyte growth factor production [GO:0032686]; negative regulation of insulin secretion [GO:0046676]; oocyte development [GO:0048599]; pituitary gland development [GO:0021983]; positive regulation of apoptotic signaling pathway [GO:2001235]; positive regulation of follicle-stimulating hormone secretion [GO:0046881]; positive regulation of ovulation [GO:0060279]; positive regulation of pathway-restricted SMAD protein phosphorylation [GO:0010862]; response to gonadotropin [GO:0034698]; response to insecticide [GO:0017085]; response to wounding [GO:0009611]; seminiferous tubule development [GO:0072520]; SMAD protein signal transduction [GO:0060395]; spermatogenesis [GO:0007283] |
14551263_Inhbb-deficient mice have failure of eyelid fusion at birth and demonstrate more severe effects on somatic growth and survival. 16127153_Activin affects both stromal cells and keratinocytes in normal and wounded skin, and the effect on keratinocytes is dose-dependent in vivo. 16601134_inhibins A and B differentially antagonize the actions of activins and BMPs to control adrenocortical C(19) steroid production 18480258_ALK7 plays an important role in regulating the functional plasticity of pancreatic islets, negatively affecting beta-cell function by mediating the effects of activin B on Ca(2+) signaling 20454446_Maintenance of female germ cells hinge upon a delicate balance between positive (WNT4 and beta-catenin) and negative (activin betaB) regulators derived from the somatic cells in the fetal ovary. 21408162_These data support the role of proconvertase PCSK5 in the processing of ovarian inhibin subunits during folliculogenesis. 21852401_the transition between stem cells and committed progenitors is neither sharp nor irreversible and GDF11, ACTbetaB and FST are crucial components of a circuit that controls both total cell number and the ratio of neuronal versus glial cells in this system 22611157_There is a dramatic induction of Inhbb mRNA, encoding the activin beta(B)-subunit, in liver challenged with lipopolysaccharide, slightly preceding an increase in Smad1/5/8 phosphorylation and Hamp mRNA. 22739106_No differences were observed in fasting glucose/insulin levels, glucose tolerance, insulin sensitivity, or insulin secretion in comparing young or older males on in comparing insulin secretion from islets of wild type or activin B knockout males. 24917498_in mammalian spinal cord, Tgfbeta ligands and ActB together support oligodendrocyte development and myelin formation 26063610_Data suggest Inhbb is physiologically essential for early folliculogenesis; Inhbb appears to participate in regulation of cell cycle progression, apoptosis, and steroidogenesis (estradiol/progesterone synthesis) in ovarian granulosa cells in culture. 26711255_Activin B, a transforming growth factor beta - related ligand, is upregulated during tumorigenesis and drives the loss of insulin expression and beta-cell maturity in a mouse insulinoma model. 27465829_This study tested the hypothesis that inhibins act in an autocrine manner on Leydig cells using a pre-pubertal Leydig cell line, TM3, as a model of immature Leydig cells. 27903526_Inflammatory stimuli strongly induce Smad2 and Smad5 phosphorylation in wild-type mice. None of these Smad effectors are activated in mice deficient in activin B. Activin B, whose messenger RNA expression is strongly induced after LPS administration or E coli infection, is required for activation of Smad1/5/8 signaling. Hepcidin expression did not depend on activin B expression. 28205525_These data suggest that activin-follistatin regulation may play a role during the development of Experimental autoimmune epididymo-orchitis. 28214591_These results suggest that prolonged exposure to hyperglycemia in the Ins2(Akita+/-) mice leads to progressive testicular disruption mediated by testicular activin activity, rather than hormonal dysregulation. 28215965_Menin binds on the promoter of Inhbb gene where it favours the recruitment of Ezh2 via an indirect mechanism involving Akt-phosphorylation. 34543458_Tubule-derived INHBB promotes interstitial fibroblast activation and renal fibrosis. 35022746_Human INHBB Gene Variant (c.1079T>C:p.Met360Thr) Alters Testis Germ Cell Content, but Does Not Impact Fertility in Mice. |
ENSG00000163083 |
INHBB |
59.587598 |
4.2804801185 |
2.097773 |
0.38497987 |
28.798044 |
0.00000008033220214953004729441108028656359785202312195906415581703186035156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000008568768229283204691806787207941642492414757725782692432403564453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
97.5887561673383 |
26.3514954672597 |
22.7985537758555 |
4.89082182804165 |
| ENSMUSG00000037139 |
242702 |
Myom3 |
protein_coding |
A2ABU4 |
FUNCTION: May link the intermediate filament cytoskeleton to the M-disk of the myofibrils in striated muscle. {ECO:0000250}. |
Coiled coil;Cytoplasm;Immunoglobulin domain;Reference proteome;Repeat |
|
|
mmu:242702; |
M band [GO:0031430]; actin filament binding [GO:0051015]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803] |
18177667_Myomesin 3, a novel structural component of the M-band in striated muscle. 26060189_MYOM3 fragments hold promise for minimally invasive assessment of experimental therapies for DMD and other neuromuscular disorders. |
ENSG00000142661 |
MYOM3 |
11.732438 |
0.1733905349 |
-2.527903 |
0.91899900 |
7.596417 |
0.00584844079463455162465335135379973507951945066452026367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0212508230400461559095326435908646089956164360046386718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.56881292013912 |
2.43023145919882 |
20.5825128991248 |
8.25271619592754 |
| ENSMUSG00000037188 |
230824 |
Grhl3 |
protein_coding |
Q5FWH3 |
FUNCTION: Transcription factor playing important roles in primary neurulation and in the differentiation of stratified epithelia of both ectodermal and endodermal origin. Binds directly to the consensus DNA sequence 5'-AACCGGTT-3' acting as an activator and repressor on distinct target genes. Essential for epidermal differentiation and barrier formation at the end of embryogenesis with TGM3 as critical direct target (PubMed:21081122, PubMed:20654612, PubMed:25347468). Exhibits functional redundancy with GRHL2 in epidermal morphogenetic events such as eyelid fusion and epidermal wound repair (PubMed:21081122). Despite being dispensable during normal epidermal homeostasis in the adulthood, is again required for barrier repair after immune-mediated epidermal damage, regulates distinct gene batteries in embryonic epidermal differentiation and adult epidermal barrier reformation after injury (PubMed:25347468). Plays unique and cooperative roles with GRHL2 in establishing distinct zones of primary neurulation. Essential for spinal closure, functions cooperatively with GRHL2 in closure 2 (forebrain/midbrain boundary) and posterior neuropore closure (PubMed:14608380, PubMed:20654612). Also required for proper development of the oral periderm (PubMed:24360809). No genetic interaction with GRHL1, no functional cooperativity due to diverse target gene selectivity (PubMed:21081122). {ECO:0000269|PubMed:14608380, ECO:0000269|PubMed:16949565, ECO:0000269|PubMed:20654612, ECO:0000269|PubMed:21081122, ECO:0000269|PubMed:24360809, ECO:0000269|PubMed:25347468}. |
DNA-binding;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:230824; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin DNA binding [GO:0031490]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; central nervous system development [GO:0007417]; cochlea morphogenesis [GO:0090103]; ectoderm development [GO:0007398]; epidermis development [GO:0008544]; establishment of planar polarity [GO:0001736]; establishment of skin barrier [GO:0061436]; eyelid development in camera-type eye [GO:0061029]; gene expression [GO:0010467]; neural tube closure [GO:0001843]; pattern specification process [GO:0007389]; planar cell polarity pathway involved in neural tube closure [GO:0090179]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of GTPase activity [GO:0043547]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of actin cytoskeleton organization [GO:0032956]; regulation of transcription by RNA polymerase II [GO:0006357]; transcription by RNA polymerase II [GO:0006366]; wound healing [GO:0042060] |
15678492_maps to Chromosome 4, 135 Mb (66 cM) from the centromere, probably not Idb3, Wnt4, Cdc42, perlecan or Grhl-3 15831758_formation and maintenance of the epidermal barrier in mice are dependent on a mammalian homolog of grainy head, Grainy head-like 3[Grhl3] 16831572_Unique role of Grhl3 in neurulation and epidermal morphogenesis. 16949565_These findings indicate that the Get-1 and LMO4 genes interact functionally to regulate epidermal terminal differentiation. 17720888_This study provides evidence for a critical role of diminished Grhl3 expression in causing spinal NTDs in the curly tail mouse model. 18485343_The failure of eyelid closure is due to critical functions of Get1 in promoting F-actin polymerization, filopodia formation, and the cell shape changes that are required for migration of the keratinocytes at the leading edge during eyelid closure. 18619436_Study demonstrates that the LIM-only domain protein, LMO4 serves as a functional partner of GRHL3 in its established roles, and define a new cooperative role for these factors in another developmental epidermal migration event, eyelid fusion. 19824061_folate deficiency caused a similar-sized, statistically significant increase in the frequency of exencephaly among both curly tail (Grhl3 mutant) embryos and background-matched embryos that are wild type for Grhl3. 20654612_Loss of Grhl3 alone defines a distinct lower spinal closure defect, also with defective dorso-lateral hinge points formation 21081122_Both GRHL2 and GRHL3 bind to and regulate expression of the wound repair gene Rho GEF 19, but regulation of the barrier forming gene, Transglutaminase 1 (TGase1), is unique to GRHL3. 22094257_The developmental transcription factor Grhl3 is a potent tumor suppressor of squamous cell carcinoma in mice. 22614019_decreased Grhl3 expression contributes to tumor progression and upregulation of the oncomir miR-21 in squamous cell carcinoma of the skin. 25347468_This study identifies a GRHL3-regulated epidermal barrier repair pathway that suppresses disease initiation and helps resolve existing lesions in immune-mediated epidermal hyperplasia. 25658689_Upregulation of miR21 and repression of Grhl3 by leptin mediates sinusoidal endothelial injury in experimental nonalcoholic steatohepatitis. 27586853_Based on the results, it proposes that developmental stage-specific Grhl3 plays a significant role in circumvallate papilla (CVP) morphogenesis not by just disruption of epithelial integrity but by regulating epithelial cell proliferation, apoptosis, and migration via Shh, Wnt, and apoptosis signaling during mouse embryogenesis. 27756203_Findings suggest that transcription factor Grainyhead-like 3 (Grhl3) may be involved in the developmental pathogenesis of craniosynostosis. 27907249_This study demonstrated that Grainyhead-like 3 (Grhl3) deficiency in brain leads to altered locomotor activity and decreased anxiety-like behaviors in aged mice. 29397878_Diminished Grhl3 expression in the hindgut is the cause of spinal NTDs in the curly tail, carrying a hypomorphic Grhl3 allele. Complete loss of Grhl3 produces a more severe phenotype in which closure fails earlier in neurulation, before onset of expression in the hindgut of wild-type embryos. Abnormal surface ectoderm function, where Grhl3 mRNA is first detected, is responsible for early spinal neurulation failure. 30189017_we observed a striking additive genetic interaction between Grhl2 and Grhl3 gain-of-function alleles. moderate Grhl3 overexpression also interacted with the Vangl2Lp allele to cause SB, demonstrating genetic interaction with the planar cell polarity signalling pathway that is implicated in mouse and human neural tube defects. 30283008_cytoplasmic localization of GRHL3 upon epidermal differentiation directly triggers epithelial morphogenesis. 30341279_Deletion of the transcription factor GRHL3 in the skin initiates a chemokine responsive signal leading to keratinocytes hyperproliferation and a pro-tumorigenic state. 30689861_these data suggest that the Tfap2a-Irf6-Grhl3 genetic pathway is shared by two embryologically distinct morphogenetic events that previously were considered independent during mammalian development 32109382_Epithelial Migration and Non-adhesive Periderm Are Required for Digit Separation during Mammalian Development. 33093151_Epithelial dynamics shed light on the mechanisms underlying ear canal defects. 33116143_Murine interfollicular epidermal differentiation is gradualistic with GRHL3 controlling progression from stem to transition cell states. 33378081_Loss of Grhl3 is correlated with altered cellular protrusions in the non-neural ectoderm during neural tube closure. 34494554_GRHL3 activates FSCN1 to relax cell-cell adhesions between migrating keratinocytes during wound reepithelialization. 36283337_Inhibition of retinoic acid signaling impairs cranial and spinal neural tube closure in mice lacking the Grainyhead-like 3 transcription factor. |
ENSG00000158055 |
GRHL3 |
123.191245 |
2.8119555717 |
1.491574 |
0.23414244 |
40.321447 |
0.00000000021543098727001603155279566305098197520617731015590834431350231170654296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000032913187543599641412843181476413634278799236199120059609413146972656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
180.823571450806 |
29.4757936768898 |
64.3052732652438 |
8.25984955516508 |
| ENSMUSG00000037211 |
24063 |
Spry1 |
protein_coding |
Q9QXV9 |
FUNCTION: Inhibits fibroblast growth factor (FGF)-induced retinal lens fiber differentiation, probably by inhibiting FGF-mediated phosphorylation of ERK1/2 (PubMed:29501879). Inhibits TGFB-induced epithelial-to-mesenchymal transition in lens epithelial cells (PubMed:25576668). {ECO:0000269|PubMed:25576668, ECO:0000269|PubMed:29501879}. |
Acetylation;Cytoplasm;Developmental protein;Membrane;Reference proteome |
|
|
mmu:24063; |
cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; membrane [GO:0016020]; nucleoplasm [GO:0005654]; animal organ development [GO:0048513]; bud elongation involved in lung branching [GO:0060449]; epithelial to mesenchymal transition involved in cardiac fibroblast development [GO:0060940]; ERK1 and ERK2 cascade [GO:0070371]; establishment of mitotic spindle orientation [GO:0000132]; metanephros development [GO:0001656]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of epithelial to mesenchymal transition [GO:0010719]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; negative regulation of fibroblast growth factor receptor signaling pathway [GO:0040037]; negative regulation of GTPase activity [GO:0034260]; negative regulation of lens fiber cell differentiation [GO:1902747]; negative regulation of MAP kinase activity [GO:0043407]; negative regulation of neurotrophin TRK receptor signaling pathway [GO:0051387]; negative regulation of Ras protein signal transduction [GO:0046580]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512]; organ induction [GO:0001759]; positive regulation of apoptotic process [GO:0043065]; ureteric bud development [GO:0001657] |
12882970_sprouty1 represents a physiologically relevant target gene of WT1 during kidney development 15691764_Spry1(-/-) embryos have supernumerary ureteric buds, resulting in the development of multiple ureters and multiplex kidneys. 16670312_Spry1 uses a novel mechanism to bring about differential effects on T cell receptor signaling through the same receptor, depending on the differentiation state of the T cell. 17022962_Epithelial cysts develop in Spry1-deficient kidneys that share several molecular characteristics with those observed in human disease, suggesting that Spry1 null mice may be useful animal models for cystic hyperplasia. 18582454_Results suggest that constitutive expression of Spry1 in chondrocytes results in attenuated FGFR2 degradation, sustained ERK activation, and up-regulation of p21Cip and STAT1 causing dysregulated chondrocyte proliferation and terminal differentiation. 18650792_AT1R-mediated inhibition of the Spry1 gene increases c-Ret tyrosine kinase activity leading to upregulation of its downstream target Wnt11, which induces GDNF in adjacent mesenchyme causing focal bursts of ureteric bud tip cell proliferation 19056869_Sprouty1 can modulate ERK signaling downstream of Ret, independent of Grb2/Sos/Ras, during renal morphogenesis 19384958_Fgf4 positively regulated Pea3, Sprouty1, and 2 expression in chick limb mesenchyme. 19915061_Sprouty1 induced by TCR signal negatively regulates further TCR signaling by interacting with proximal signaling molecules in immune synapse, providing a novel regulatory mechanism of T cells. 20084103_Fgf10 cooperates with Gdnf to promote Kidney morphogenesis, and can compensate for the loss of Gdnf/Ret in the absence of Spry1. 20144785_demonstrate that Sprouty1 (Spry1) is expressed in quiescent Pax7(+) satellite cells in uninjured muscle, downregulated in proliferating myogenic cells after injury, and reinduced as Pax7(+) cells re-enter quiescence. 20410440_Spry1 is a critical regulator of adipocyte differentiation and mesenchymal stem cell (MSC) lineage allocation, potentially acting through regulation of CEBP-beta and TAZ. 20459789_These data suggest that Spry1 is an important regulator of craniofacial and cardiac morphogenesis and perturbations in Spry1 levels may contribute to congenital disorders involving tissues of neural crest origin. 20719962_Phosphatidylinositol-specific phospholipase C (PLC)-gamma was identified as a partner of the Spry1 and Spry2 proteins. 20813052_Results suggest that SPRY1 is an endogenous angiogenesis inhibitor. 21310958_the role of miR-29c in a previously unrecognized signaling cascade involving Spry1 and Rho kinase activation. 21362415_report that in Spry1, Spry2 compound mutant embryos, the otic placode is increased in size 21483770_Spry1 over expression inhibits primitive hematopoietic progenitor and erythroblastic cell development and expansion while having no obvious effect on endothelial cell development 21518456_Using deletion mutants we identified the N-terminus and the CR conserved region (CR) 3 of E1A- and the C-terminal half of Spry1, which contains the highly conserved Spry domain, as the essential sites for direct interaction between Spry and E1A. 21595564_SHP2, a major determinant of balance between mESC self-renewal and differentiation, directly regulates Spry1 activity to modulate ERK1/2 signaling and lineage-specific differentiation in mESCs 21693512_Sprouty genes prevent excessive FGF signalling in multiple cell types throughout development of the cerebellum. 21743007_Corneal epithelial precursors in Spry1 and -2 double mutants showed increased proliferation with elevated expression of Erm and DUSP6 and decreased expression of the corneal differentiation marker K12. 21806979_Sprouty genes are essential for the normal development of epibranchial ganglia in the mouse embryo. 22142492_investigation of role of Spry1: Overexpression of Spry1 in transgenic mice leads to lower body fat, reduced bone loss, and normal metabolic function (i.e., prevents liver steatosis/glucose intolerance) compared with Spry1-negative transgenic mice. 22158037_findings identify Spry1 as a candidate tumor-suppressor gene in medullary thyroid carcinoma 22328578_Spry1/Spry2 highly expressed in lateral pterygoid and temporal muscles. combined inactivation of Spry1 and Spry2 results in muscle overgrowth disrupting normal glenoid fossa development. TMJ condyle and disc develop independently of the mandibular fossa. 22508938_Studies implicate Spry1 as a novel regulator of erythropoiesis during anemia, transducer of EPO/EPOR signals, and candidate suppressor of Jak2 activity. 23064016_To determine whether modulating RTK signalling may overcome the abnormal nephrogenesis of Fraser syndrome, we introduced a single null Sprouty1 allele into Fras1(bl/bl) mice, reducing the ureteric bud's expression of this anti-branching molecule. 23074222_ISG15 mRNA expression and IFN-dependent antiviral responses are enhanced in Spry1,2,4 triple knock-out mouse embryonic fibroblasts, consistent with negative feedback regulatory roles for Spry proteins in IFN-mediated signaling. 23150596_When Spry1 and Spry2 loss-of-function occurs in the context of heterozygosity for a null allele of the tumor suppressor gene Pten, there is a striking increase in prostatic intraepithelial neoplasia and evidence of neoplastic invasion. 23355454_Findings demonstrate that Pokemon suppresses Sprouty1 expression through a miR-21-mediated mechanism, affecting the growth and proliferation of liver cancer cells. 24055172_conjunctival epithelial Spry1 and Spry2 redundantly promote eyelid closure. 24270409_In vivo analysis of thyroid glands from Spry1 knockout mice reveals that Spry1 induces a senescence-associated secretory phenotype via activation of the NFkappaB pathway. 24361260_Spry1 and Spry2 coordination is required for normal development of the external genitalia in mice 24510305_showed that hSpry1 overexpression prevents VEGF secretion 26075267_Cosuppression of Sprouty and Sprouty-related negative regulators of FGF signalling in prostate cancer 26375880_Sprouty gain of function disrupts lens cellular processes and growth by restricting receptor tyrosine kinase signaling. 26830459_Here, we present a novel mouse model of pheochromocytoma consisting of double-heterozygous mice for Pten and Sprouty1 (Spry1), which leads to pheochromocytomas that appear at earlier onset and grow at a higher rate than those from Pten+/- mice. 27621461_modulation of stromal paracrine signaling and extracellular matrix remodeling by SPRY1 regulates mammary epithelial morphogenesis during postnatal development. 28186499_The results show that lnc-Spry1 could act as an early mediator of TGF-beta signaling and reveal different roles for a long noncoding RNA in modulating transcriptional and posttranscriptional gene expression. 29105817_Study demonstrated that Spry1 has a role in supporting mitogen-induced vascular hyperplasia, at least in part by promoting Akt/Rb signaling and increasing mitogen-induced cyclinD1 expression and decreasing p27Kip1 expression. 29398368_Inhibition of miR-29b could attenuate atherosclerosis by inhibiting the SPRY1/MAPK signaling pathway and inflammation in aorta. 30126987_Loss of Spry1/2 enhances the survival of effector CD8+ T cells and results in the formation of more protective memory cells. Deleting Spry1/2 in antigen-specific CD8+ T cells may have therapeutic potential for enhancing the survival and functionality of effector and memory CD8+ T cells in vivo. 30569452_that SPRY1 contributes to the regulation of CNS functions by influencing both neuronal differentiation under normal physiological processes and neuronal survival under pathological conditions 30635790_Our findings indicate that Spry1 is an important regulator of cardiomyocyte viability during ischemia-reperfusion injury 31300484_Tyrosine 53 is absolutely necessary for Sprouty1 function during genitourinary development in mice 32357909_MiR-21-5p regulates extracellular matrix degradation and angiogenesis in TMJOA by targeting Spry1. 32753399_Nephron Progenitor Maintenance Is Controlled through Fibroblast Growth Factors and Sprouty1 Interaction. 34164910_METTL3/N6-methyladenosine/ miR-21-5p promotes obstructive renal fibrosis by regulating inflammation through SPRY1/ERK/NF-kappaB pathway activation. 34714716_Sprouty1 regulates gonadal white adipose tissue growth through a PDGFRalpha/beta-Akt pathway. |
ENSG00000164056 |
SPRY1 |
584.195881 |
0.3261953952 |
-1.616192 |
0.16379296 |
94.621800 |
0.00000000000000000000023046620598721692906619456140898903432186965047533438963751331249318887728350091492757201194763183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000104789888047680242810085296145203346077379119361656394288952187654473391376086510717868804931640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
286.489064213824 |
32.5957763680332 |
878.274397616795 |
72.625025167365 |
| ENSMUSG00000037259 |
241688 |
Dzank1 |
protein_coding |
Q8C008 |
|
ANK repeat;Direct protein sequencing;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Zinc;Zinc-finger |
|
|
mmu:241688; |
metal ion binding [GO:0046872]; eye photoreceptor cell development [GO:0042462] |
27310661_Association of a chromosomal rearrangement event with mouse posterior polymorphous corneal dystrophy and alterations in Csrp2bp, Dzank1, and Ovol2 gene expression have been reported. |
ENSG00000089091 |
DZANK1 |
66.609742 |
0.2050934700 |
-2.285647 |
0.76101242 |
8.067802 |
0.00450587520078059907552292528976067842449992895126342773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0170264180248797039607389081083965720608830451965332031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.2094050527395 |
10.0570304477867 |
88.7858840821257 |
35.9062328980837 |
| ENSMUSG00000037370 |
18605 |
Enpp1 |
protein_coding |
A0A0R4J1Q7 |
Human_homologues FUNCTION: Nucleotide pyrophosphatase that generates diphosphate (PPi) and functions in bone mineralization and soft tissue calcification by regulating pyrophosphate levels (By similarity). PPi inhibits bone mineralization and soft tissue calcification by binding to nascent hydroxyapatite crystals, thereby preventing further growth of these crystals (PubMed:11004006). Preferentially hydrolyzes ATP, but can also hydrolyze other nucleoside 5' triphosphates such as GTP, CTP and UTP to their corresponding monophosphates with release of pyrophosphate, as well as diadenosine polyphosphates, and also 3',5'-cAMP to AMP (PubMed:27467858, PubMed:8001561, PubMed:25344812, PubMed:28011303). May also be involved in the regulation of the availability of nucleotide sugars in the endoplasmic reticulum and Golgi, and the regulation of purinergic signaling (PubMed:27467858, PubMed:8001561). Inhibits ectopic joint calcification and maintains articular chondrocytes by repressing hedgehog signaling; it is however unclear whether hedgehog inhibition is direct or indirect (By similarity). Appears to modulate insulin sensitivity and function (PubMed:10615944). Also involved in melanogenesis (PubMed:28964717). Also able to hydrolyze 2',3'-cGAMP (cyclic GMP-AMP), a second messenger that activates TMEM173/STING and triggers type-I interferon production (PubMed:25344812). 2',3'-cGAMP degradation takes place in the lumen or extracellular space, and not in the cytosol where it is produced; the role of 2',3'-cGAMP hydrolysis is therefore unclear (PubMed:25344812). Not able to hydrolyze the 2',3'-cGAMP linkage isomer 3'-3'-cGAMP (PubMed:25344812). {ECO:0000250|UniProtKB:P06802, ECO:0000269|PubMed:10615944, ECO:0000269|PubMed:25344812, ECO:0000269|PubMed:27467858, ECO:0000269|PubMed:28011303, ECO:0000269|PubMed:28964717, ECO:0000269|PubMed:8001561, ECO:0000305|PubMed:11004006}. |
Calcium;Disulfide bond;Glycoprotein;Hydrolase;Membrane;Metal-binding;Proteomics identification;Reference proteome;Repeat;Secreted;Transmembrane;Transmembrane helix;Zinc |
|
This gene encodes a member of the nucleoside pyrophosphatase/phosphodiesterase family of enzymes that catalyzes the hydrolysis of pyrophosphate and phosphodiester bonds in nucleotide triphosphates and oligonucleotides, respectively, to generate nucleoside 5'-monophosphates. The encoded protein is a type II transmembrane glycoprotein that negatively regulates bone mineralization. Mice harboring a nonsense mutation in this gene, termed tiptoe walking (ttw), exhibit ectopic ossification of the spinal ligaments. The encoded protein binds to the insulin receptor, inhibits downstream signaling events and induces insulin resistance and glucose tolerance. This gene is located adjacent to a paralog on chromosome 10. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Apr 2015]. |
mmu:18605; |
cell surface [GO:0009986]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; ATP binding [GO:0005524]; cyclic-GMP-AMP hydrolase activity [GO:0106177]; dinucleotide phosphatase activity [GO:0004551]; insulin receptor binding [GO:0005158]; metal ion binding [GO:0046872]; nucleic acid binding [GO:0003676]; nucleoside triphosphate diphosphatase activity [GO:0047429]; phosphatase activity [GO:0016791]; phosphodiesterase I activity [GO:0004528]; polysaccharide binding [GO:0030247]; protein homodimerization activity [GO:0042803]; scavenger receptor activity [GO:0005044]; 3'-phosphoadenosine 5'-phosphosulfate metabolic process [GO:0050427]; ATP metabolic process [GO:0046034]; bone mineralization [GO:0030282]; cellular phosphate ion homeostasis [GO:0030643]; cellular response to insulin stimulus [GO:0032869]; gene expression [GO:0010467]; generation of precursor metabolites and energy [GO:0006091]; immune response [GO:0006955]; inorganic diphosphate transport [GO:0030505]; melanocyte differentiation [GO:0030318]; negative regulation of cell growth [GO:0030308]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of glucose import [GO:0046325]; negative regulation of glycogen biosynthetic process [GO:0045719]; negative regulation of insulin receptor signaling pathway [GO:0046627]; negative regulation of protein autophosphorylation [GO:0031953]; nucleoside triphosphate catabolic process [GO:0009143]; response to ATP [GO:0033198]; response to inorganic substance [GO:0010035]; sequestering of triglyceride [GO:0030730] |
12082181_correction of bone mineralization abnormalities in knockout mice null for both the TNAP (Akp2) and PC-1 (Enpp1) genes. 12533192_functional characterization of the non-catalytic ectodomains of this enzyme 12817751_Linked regulatory effects on extracellular PP(i) and OPN expression mediate the ability of PC-1 and ANK to regulate calcification. 15075217_Specific targeting motif in the NPP1 cytosolic tail that delivers PP(i)-generating NPP activity to osteoblast matrix vesicles for control of calcification. 15625282_NPP1 and PP(i) deficiencies modulate phenotype plasticity in artery smooth muscle cells and chondrogenesis in mesenchymal precursors 17311850_Lubrol resistance is an intrinsic property of NPP3 but not NPP1, which is acquired early during the biosynthetic process irrespective of its final destination, and depends on positively charged residues in its cytoplasmic tail. 17552941_expression of PC-1 downstream of fibroblast growth factor signaling is an integral aspect of osteoblastic function and pyrophosphate elaboration may play a significant role in the pathology of craniosynostosis 17849011_ENPP1 expression was found down-regulated during 3T3-L1 maturation, and over-expression of human ENPP1 in 3T3-L1 (pQCXIP-ENPP1 vector) resulted in adipocyte insulin resistance and in defective adipocyte maturation 18698132_These results indicate that the mechanism of PC-1 induction by FGF2 in preosteoblastic cells is distinct from the mechanism of induction that occurs with osteoblast differentiation. 19049325_FGF2 is a specific inducer of PC-1 in pre-osteoblast cells and that FGF2 induces PC-1 expression through a mechanism involving Runx2. 19419305_results support a role for the Enpp1 gene in the pathogenesis associated with mineralization of articular cartilage and vascular calcification 19577557_Suppression of PC-1 expression improves insulin sensitivity in vitro and in an animal model of diabetes 19874193_NPP1 does not have a major role in PP(i) generation from ATP at the level of MVs, in contrast to its accepted role on the surface of the osteoblasts and chondrocytes, but rather acts as a phosphatase in the absence of TNAP. 20048161_Data show that reconstitution of TNAP and NPP1 into proteoliposome membranes generates a phospholipid microenvironment that allows the kinetic study of phosphosubstrate catabolism in a manner that recapitulates the native MV microenvironment. 20803545_Msx2 and Runx2 likely function together to induce PC-1 gene expression in osteoblastic cells and FGF signaling stimulates Msx2 transcriptional activity through the Frs2 mediated MAPK signaling pathway 21099228_RAGE promotes ectopic chondrogenic differentiation and arterial calcification in Enpp1-/- mice. 21477221_Npp1 promotes atherosclerosis, potentially mediated by Opn expression in ApoE knockout mice 21930712_ENPP1 plays multiple and distinct roles in the development of mineralized tissues and that the influence of ENPP1 on osteoblast differentiation and gene expression may include a mechanism that is independent of its catalytic activity. 22359666_NPP1 is essential for normal bone development and control of physiological bone mineralization 22510396_Decreased levels of nucleotide pyro- phosphatase phosphodiesterase 1 trigger osteoarthritic changes in mice. 22750863_crystal of Enpp1 belonged to space group P3(1), with unit-cell parameters a = b = 105.3, c = 173.7 A. 23169635_Expression of plasma cell alloantigen 1 defines layered development of B-1a B-cell subsets with distinct innate-like functions. 23798568_The mutant ENPP1 protein is largely absent in the liver of 'ages with stiffened joints' mice, and the lack of enzymatic activity results in reduced inorganic pyrophosphate (PPi) levels in the plasma, accompanied by extensive mineralization. 23861746_these data suggest a potential role for Enpp1 in the development of breast cancer bone metastasis. 24530784_Both mono-allelic and bi-allelic ENPP1 deficiency promote dysregulated VSMC function, with robust C/EBP homologous protein expression and enhanced neointimal hyperplasia after injury in vivo, but marked post-injury calcification limited to Enpp1-/- mice. 24732453_Studied the role of genetic modulation and the role of diet in nephrocalcinosis using two established mouse models of ectopic mineralization, Abcc6(tm1Jfk) and Enpp1(asj) mice. 24770645_Its mutaion causes osteochondral diseases and malnutrition. 25260930_these data highlight the key role of NPP1 in regulating calcification of both soft and skeletal tissues. 25344812_ENPP1 2'3'-cGAMP-hydrolyzing activity is repsonsible for tumor progression in humans and mice. 25368121_NPP1 has a role in obesity and diabetes in a mouse model 25479107_we have characterized the phenotypic and histopathologic features of this spontaneous mutant mouse, designated as asj-2J, and we have identified a large deletion/insertion mutation in the Enpp1 gene 25644539_Expression of NPP1 and 5'-nucleotidase by valve interstitial cells promotes the mineralization of the aortic valve through A2aR and a cAMP/PKA/CREB pathway. 26033523_Increased NPP1 expression and activity might contribute to the decreased mineralisation observed when osteoblasts are exposed to acid conditions. 26054750_Vitamin D3 regulates Enpp1 expression, which presumably, in the context of adequate tissue non-specific alkaline phosphatase activity, provides phosphate to stimulate mineralisation. 26624227_ENPP1-Fc fusion protein prevents the mortality, vascular calcifications and sequela of disease in mouse models of generalized arterial calcification of infancy. 27406238_identified ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1) as the first known mammalian enzyme lacking a NUDIX domain to generate pR from ADP-ribose on modified proteins in vitro 27863377_Report the temporal ectopic mineralization phenotype of connective tissues in Enpp1asj-2J mouse model for generalized arterial calcification of infancy. 27959908_Enpp1asj mutant mouse provides a new animal model for studying tympanosclerotic otitis and otitis media with effusion 28080219_Evidence of a causative link between ENPP1 and alterations in insulin signaling, glucose uptake, and lipid metabolism in subcutaneous abdominal Adipose tissue of gestational diabetes, which may mediate insulin resistance and hyperglycemia in Gestational diabetes. 28704395_Results show that urine pyrophosphate (PPI) levels are increased in Npt2a-/- mice when compared to WT, possibly to protect from renal mineralization in the setting of hyperphosphaturia. However, the presence of two hypomorphic Enpp1asj/asj alleles decreases urine PPi and worsens renal calcium phosphate deposit formation in Npt2a-/- mice. 28798354_Enpp1 plays a crucial role in regulating aging via Klotho expression under phosphate overload conditions. 29259245_ENPP1 deficiency confers an energetic disadvantage to PCs for long-term survival and antibody production. 29516286_found that, like NTPDase1 (ectonucleoside triphosphate diphosphohydrolase 1, CD39), NPP1 (ectonucleotide pyrophosphatase/phosphodiesterase 1, PC-1) is also highly expressed in primary cultured murine microglia 30047979_Runt-related transcription factor 2 (Runx2) was confirmed to have synergistic effects with Osx in activating ENPP1 promoter. 30111653_Enpp1 inhibits hedgehog signaling and chondrocyte hypertrophy by activating Galphas-PKA signaling. 31805212_Human Heterozygous ENPP1 Deficiency Is Associated With Early Onset Osteoporosis, a Phenotype Recapitulated in a Mouse Model of Enpp1 Deficiency. 32980560_Genetic pathways disrupted by ENPP1 deficiency provide insight into mechanisms of osteoporosis, osteomalacia, and paradoxical mineralization. 33305372_Osteoblast-specific deficiency of ectonucleotide pyrophosphatase or phosphodiesterase-1 engenders insulin resistance in high-fat diet fed mice. 33465815_Response of the ENPP1-Deficient Skeletal Phenotype to Oral Phosphate Supplementation and/or Enzyme Replacement Therapy: Comparative Studies in Humans and Mice. 35588451_ENPP1's regulation of extracellular cGAMP is a ubiquitous mechanism of attenuating STING signaling. 35773783_Catalysis-Independent ENPP1 Protein Signaling Regulates Mammalian Bone Mass. 36243801_ENPP1 deletion causes mouse osteoporosis via the MKK3/p38 MAPK/PCNA signaling pathway. |
ENSG00000197594 |
ENPP1 |
49.964470 |
0.3438609645 |
-1.540103 |
0.36202328 |
18.073205 |
0.00002125720035222816183310100279779675247482373379170894622802734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001477816867937651018182626527774914393376093357801437377929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
25.1501796817253 |
6.37751287424547 |
73.1405488870479 |
12.8185428059275 |
| ENSMUSG00000037447 |
214855 |
Arid5a |
protein_coding |
Q3U108 |
FUNCTION: DNA-binding protein that may regulate transcription and act as a repressor by binding to AT-rich stretches in the promoter region of target genes (By similarity). May positively regulate chondrocyte-specific transcription such as of COL2A1 in collaboration with SOX9 and positively regulate histone H3 acetylation at chondrocyte-specific genes. May stimulate early-stage chondrocyte differentiation and inhibit later stage differention (PubMed:21346191). Can repress ESR1-mediated transcriptional activation; proposed to act as corepressor for selective nuclear hormone receptors (By similarity). As RNA-binding protein involved in the regulation of inflammatory response by stabilizing selective inflammation-related mRNAs, such as IL6, STAT3 and TBX21. Binds to stem loop structures located in the 3'UTRs of IL6, STAT3 and TBX21 mRNAs; at least for STAT3 prevents binding of ZC3H12A to the mRNA stem loop structure thus inhibiting its degradation activity. Contributes to elevated IL6 levels possibly implicated in autoimmunity processes. IL6-dependent stabilization of STAT3 mRNA may promote differentiation of naive CD4+ T-cells into T-helper Th17 cells (PubMed:23676272, PubMed:27022145). In CD4+ T-cells may also inhibit RORC-induced Th17 cell differentiation independently of IL6 signaling (PubMed:24782182). Stabilization of TBX21 mRNA contributes to elevated interferon-gamma secretion in Th1 cells possibly implicated in the establishment of septic shock (PubMed:27671645). Stabilizes TNFRSF4/OX40 mRNA by binding to the conserved stem loop structure in its 3'UTR; thereby competing with the mRNA-destabilizing functions of RC3H1 and endoribonuclease ZC3H12A (PubMed:29244194). {ECO:0000250|UniProtKB:Q03989, ECO:0000269|PubMed:21346191, ECO:0000269|PubMed:23676272, ECO:0000269|PubMed:24782182, ECO:0000269|PubMed:27022145, ECO:0000269|PubMed:27671645, ECO:0000269|PubMed:29244194}. |
Activator;Alternative splicing;DNA-binding;Immunity;Innate immunity;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Repressor;RNA-binding;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:214855; |
nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; chromatin binding [GO:0003682]; identical protein binding [GO:0042802]; nuclear androgen receptor binding [GO:0050681]; nuclear estrogen receptor binding [GO:0030331]; nuclear retinoid X receptor binding [GO:0046965]; nuclear thyroid hormone receptor binding [GO:0046966]; RNA stem-loop binding [GO:0035613]; sequence-specific DNA binding [GO:0043565]; transcription cis-regulatory region binding [GO:0000976]; transcription corepressor activity [GO:0003714]; cellular response to estrogen stimulus [GO:0071391]; cellular response to lipopolysaccharide [GO:0071222]; chondrocyte differentiation [GO:0002062]; innate immune response [GO:0045087]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of histone acetylation [GO:0035066]; positive regulation of interleukin-17 production [GO:0032740]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of T-helper 1 cell cytokine production [GO:2000556]; positive regulation of T-helper 17 type immune response [GO:2000318]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of tumor necrosis factor production [GO:0032760]; positive regulation of type II interferon production [GO:0032729]; regulation of transcription by RNA polymerase II [GO:0006357] |
21346191_AT-rich interactive domain-containing protein 5a (Arid5a) cooperates with Sox9 to stimulate chondrocyte-specific transcription. 23676272_Arid5a controls IL-6 mRNA stability, which contributes to elevation of IL-6 level in vivo. 27022145_T cell-intrinsic role of Arid5a on fate decisions of naive CD4(+)T cells through selective stabilization of Stat3 mRNA. 27671645_Arid5a deficiency resulted in decreased levels of IFN-gamma under Th1 cell conditions, in which T-box expressed in T cells (T-bet) mRNA expression was inhibited. 28168301_data showed that regulation of Arid5a by NF-kappaB and MAPK signaling is required to maintain the balance of IL-6 mRNA expression 28379390_this study shows that Arid5a-deficient mice were highly refractory to bleomycin-induced lung injury 29244194_Arid5a regulates the augmentation of OX40 in CD4+ T cells, and impairs destabilizing functions of Regnase-1 and Roquin-1 on OX40 alternative decay element-like stem-loop in the 3' untranslated region 29358370_High Arid5a expression is associated with inflammation. 30301788_Arid5a orchestrates a feed-forward amplification loop, which promoted IL-17 signaling by controlling mRNA stability and translation 31289228_Feedback regulation of Arid5a and Ppar-gamma2 maintains adipose tissue homeostasis. 34389433_The novel long noncoding RNA AU021063, induced by IL-6/Arid5a signaling, exacerbates breast cancer invasion and metastasis by stabilizing Trib3 and activating the Mek/Erk pathway. 35940634_Arid5a Mediates an IL-17-Dependent Pathway That Drives Autoimmunity but Not Antifungal Host Defense. |
ENSG00000196843 |
ARID5A |
232.297299 |
0.4604181290 |
-1.118983 |
0.26942298 |
16.584681 |
0.00004652531466049829268413762228817631694255396723747253417968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003005401157373664331427798313001176211400888860225677490234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
139.469089443621 |
26.1974797449043 |
302.918327200215 |
41.986840941994 |
| ENSMUSG00000037544 |
218977 |
Dlgap5 |
protein_coding |
A0A0R4J0G7 |
Human_homologues FUNCTION: Potential cell cycle regulator that may play a role in carcinogenesis of cancer cells. Mitotic phosphoprotein regulated by the ubiquitin-proteasome pathway. Key regulator of adherens junction integrity and differentiation that may be involved in CDH1-mediated adhesion and signaling in epithelial cells. {ECO:0000269|PubMed:12527899, ECO:0000269|PubMed:14699157, ECO:0000269|PubMed:15145941}. |
Proteomics identification;Reference proteome |
|
|
mmu:218977; |
centriolar satellite [GO:0034451]; cytosol [GO:0005829]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; spindle pole centrosome [GO:0031616]; signaling [GO:0023052] |
12527899_endogenous levels of HURP mRNA were found to be tightly regulated during cell cycle progression and in regenerating mouse liver after partial hepatectomy [HURP] 16364248_These results indicate that the HURP gene might be a potential oncogenic gene and capable of enhancing the chemosensitivity of deoxycytosine analogs in NIH3T3 cells. 18676373_HURP has an essential function in uterine biology 21173113_The process of bipolar spindle assembly in the presence of multiple MTOCs in female meiosis resembles the clustering of extra centrosomes during cancer cell division. HURP plays an essential role in both processes. |
ENSG00000126787 |
DLGAP5 |
647.365484 |
2.4945629008 |
1.318787 |
0.18606673 |
49.207478 |
0.00000000000230271152495724395258397500587966975254053392241360143088968470692634582519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000438985005608870284243829202891083642729119773662205261643975973129272460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
921.855061401814 |
119.22302859312 |
369.545727267678 |
36.2628336201564 |
| ENSMUSG00000037664 |
12577 |
Cdkn1c |
protein_coding |
P49919 |
FUNCTION: Potent tight-binding inhibitor of several G1 cyclin/CDK complexes (cyclin E-CDK2, cyclin D2-CDK4, and cyclin A-CDK2) and, to lesser extent, of the mitotic cyclin B-CDC2. Negative regulator of cell proliferation. May play a role in maintenance of the non-proliferative state throughout life. |
Alternative splicing;Cell cycle;Methylation;Nucleus;Protein kinase inhibitor;Reference proteome |
|
|
mmu:12577; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; cyclin-dependent protein serine/threonine kinase inhibitor activity [GO:0004861]; molecular function inhibitor activity [GO:0140678]; protein kinase inhibitor activity [GO:0004860]; protein-containing complex binding [GO:0044877]; adrenal gland development [GO:0030325]; aging [GO:0007568]; camera-type eye development [GO:0043010]; cell cycle [GO:0007049]; digestive system development [GO:0055123]; embryonic placenta morphogenesis [GO:0060669]; genomic imprinting [GO:0071514]; kidney development [GO:0001822]; multicellular organism growth [GO:0035264]; myeloid cell differentiation [GO:0030099]; negative regulation of cyclin-dependent protein kinase activity [GO:1904030]; negative regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0045736]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of kinase activity [GO:0033673]; negative regulation of mitotic cell cycle [GO:0045930]; negative regulation of phosphorylation [GO:0042326]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuron maturation [GO:0042551]; placenta development [GO:0001890]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transforming growth factor beta receptor signaling pathway [GO:0030511]; regulation of exit from mitosis [GO:0007096]; regulation of lens fiber cell differentiation [GO:1902746]; skeletal system development [GO:0001501]; uterus development [GO:0060065] |
11891335_Induction of p57(KIP2) expression by p73beta 12478365_The cell cycle inhibitors p27 and p57 synergistically regulate podocyte numbers and glomerular size in mice. 12482580_VEGF has a role in placental hyperplasia of p57Kip2 null mouse embryos 12925736_SCFSkp2 complex plays an important role in cell-cycle progression by determining the abundance of p57Kip2 and that of the related CDK inhibitor p27Kip1. 12963725_p57KIP2 modulates stress-activated signaling by functioning as an endogenous inhibitor of JNK/SAPK 14671317_p57Kip2 influences postmitotic differentiation of dopamine neurons. 15124025_p57kip2 mediates proliferative actions of PTHrP in chondrocytes. 15294951_p57(Kip2) is involved in the regulation of several aspects of the T cell cycle 15647320_Methylation of the 5' DMR plays an important role in the imprinting of the Cdkn1c gene. 15754413_There is a marked loss or absence of p57(kip2) expression and high expression of PCNA in hepatocellular carcinoma 15890200_Cdkn1c gene expression was analyzed at three different stages in differentiating MyoD(-)C2C12 myoblasts 16176803_Maintenance of bone marrow side population cells in G0/G1 may be controlled by gene expression of p57(Kip2). 16575194_Recent advances in epigenetic control of the CDKN1C/KCNQ1OT1 imprinted domain in both humans and mice, causing Beckwith-Wiedemann syndrome and cancer. 16899237_Hes1 and p57 coordinate cell cycle exit and self-renewal of pancreatic progenitors during an early stage in organogenesis to regulate the number of pancreatic progenitors.They also eliminate cells with aberrant cell cycle characteristics. 17092932_both p57 and p27 play essential roles in neuronal migration and may, in concert, coordinate the timing of neuronal differentiation, migration, and possibly cell cycle arrest in neocortical development 17517131_Cdkn1c, an imprinted gene with expression only from the maternal allele, is the major regulator of embryonic growth in the IC2 imprinted domain on mouse distal chromosome 7. 17709399_Data show that the Notch effector Herp2 is expressed in lens epithelium and directly suppresses p57Kip2 expression, providing a molecular link between Notch signaling and the cell cycle control machinery during lens development. 18312674_These data suggest that forced cardiac expression of p57Kip2 does not affect myocardial growth, differentiation and baseline function but attenuates injury from ischemia-reperfusion in the adult mouse heart. 18430730_cyclin-dependent kinase inhibitor p57(Kip2) and vascular endothelial growth factor mRNAs are selectively translated by an IRES-independent mechanism under hypoxic stress 18483241_p57(Kip2) is an important gene in prostate cancer tumorigenesis 18513743_Data show that the minimal promoter element able to confer MyoD responsiveness contains multiple Sp1 and Egr1 recognition sites and that both transcription factors are necessary for the increase in p57 RNA. 18625706_Hdac3 overexpression inhibits several cyclin-dependent kinase inhibitors, including Cdkn1a, Cdkn1b, Cdkn1c, Cdkn2b, and Cdkn2c. Hdac3-Tg mice did not develop cardiac hypertrophy at 3 months of age, unlike previously reported Hdac2-Tg mice 19139274_During normal development, progenitor cell cycle exit is controlled by p57(Kip2) followed by p27(Kip1) in differentiated cells. 19229324_C/EBPbeta transactivates p57(Kip2) to promote transition from proliferation to hypertrophic differentiation of chondrocytes during endochondral ossification 19276117_p57 and p27 share many characteristics in vivo, but p57 also performs specific functions not amenable to substitution with p27. 20140255_Data provide evidence that sik1 via p57Kip2 plays a central role in the time clock mechanism that regulates cardiac cell proliferation and differentiation. 20372090_In the absence of maintained DNA methylation (Dnmt1(-/-)), Cdkn1c escapes silencing demonstrating the requirement for DNA methylation in long term silencing in vivo. 21245411_p57(Kip2) coordinates multiple stages of corticogenesis and exhibits distinct and common activities compared with related family member p27(Kip1). 21729874_Cdkn1c mutants attained a 20% increase in weight during gestation but experienced a rapid reversal of this positive growth trajectory very late in gestation. 21816904_We particularly focus our attention on p57(Kip2) changes in cancers and pharmacological approaches for modulating p57(Kip2) levels. 21844226_Observations indicate that p57Kip2 regulates the activities of E2f1 and p53 to ensure the proper proliferation and survival of cells in the subcommissural organ (SCO) as well as of Pax2-positive cells in the cerebellar primordium. 21885020_cooperates with p27(Kip1)to maintain hematopoietic stem cell quiescence through interactions with Hsc70 22209285_Hesperetin prevented the xenograft growth by down-regulating the expression of cyclin D1, CDK4 and Bcl-xL and up-regulating p57Kip2 expression in MCF-7 cells. 22223678_The data suggested that p57KIP2 and p27KIP1 control neuronal output for distinct cortical layers by regulating different stages of precursor proliferation. 22569127_A novel function was uncovered for p57 that integrates external signals transduced by p38 stress-activated protein kinase to control the cell-cycle, establishing a checkpoint in G1 which is critical to permit cellular adaptation to stress. 23481253_The regulation of NSC quiescence by p57 might thus have important implications for the short-term (extrinsic stimuli-dependent) and long-term (age-related) modulation of neurogenesis. 23968833_Results support a model where ARX regulates the expansion of cortical progenitor cells through repression of Cdkn1c 24848107_p57 and p21 serve nonredundant functions in trophoblast giant cells; the role of p21 in suppressing apoptosis is restricted to terminally differentiated cells. 25005473_A muscle-specific regulatory element of p57(kip2) directly activated by muscle regulatory factors in myoblasts but repressed by the Notch targets Hes1/Hey1 in progenitor cells, is identified. 25086570_Data indicate that cyclin-dependent kinase inhibitor 1C (P57 was post-transcriptionally regulated by microRNA miR-221 in embryonic stem (ES) cells. 26071365_Lhx6 and Lhx8 promote palate development through negative regulation of a cell cycle inhibitor gene, p57Kip2 26100882_Data show that disruption of potassium voltage-gated channel, KQT-like subfamily Q, member1 (KCNQ1) results in increased expression of cyclin-dependent kinase inhibitor 1C (Cdkn1c) only when the mutation is on the paternal allele. 26165599_hepatoblasts in p57(Kip2)-/- mice were highly proliferative and had deficient maturation compared with those in wild-type (WT) mice. 26872540_indicate that the effects of insulin-like growth factor 2 (IGF2) are mediated by direct upregulation of the cyclin-dependent kinase inhibitor p57 (p57). 26876150_Data show that transforming growth factor beta (TGFbeta)-induced changes in Gata2 transcription factor and cyclin-dependent kinase inhibitor 1C (P57) expression in hematopoietic progenitors are conveyed through Smad signaling via Smad4 protein. 26963625_This study reveals a key requirement for Cdkn1c in the early development of the brown adipose lineages. 26988311_Immunohistochemistry revealed robust p57(kip2) staining in trophoblast giant cells and in the ectoplacental cone at E8.5. p57(kip2) protein was seen in giant cells and throughout the labyrinth, although its abundance was reduced in the junctional zone at E9.5, and became more diffuse by E12.5. 27415617_Dnmt3a-cKO muscles exhibit fewer Pax7+ SCs, which show increased expression of p57Kip2 protein 27798108_This is the first report linking elevated Cdkn1c to altered behaviour in mice. Importantly, the findings from our study may have relevance for Silver Russell Syndrome and highlight a potentially underreported aspect of this disorder. 28147266_Cdkn1c-luciferase mice offer non-invasive tools to identify factors that disrupt epigenetic processes. 28560351_progenitors are dependent on p57(KIP2)-mediated slowing of replication forks for self-renewal, a novel function for cyclin-dependent kinase inhibitors. The switch to differentiation entails rapid down-regulation of p57(KIP2) with a consequent global increase in replication fork speed and an abruptly shorter S phase. 28857482_the consequence of elevated Cdkn1c expression on dopamine-related behaviours highlighting the importance of correct dosage of this imprinted gene in the brain 28926866_Our data indicate that mouse embryonic stem cells are induced into islet-like cells in vitro. The gene imprinting status of Kcnq1 and Cdkn1c may be changed in differentiated cells during the induction in vitro. 28939666_this study identifies E2A target genes in embryonic neural stem cells and demonstrates that E47 regulates neuronal differentiation via p57(KIP2). 29284139_The results reveal a novel Shh-Foxd1-Cdkn1c regulatory circuit that drives the mitogenic action of Shh signaling. Shh pathway activation of Foxd1 is followed by downregulation of Cdkn1c, which encodes a cyclin-dependent kinase inhibitor. 29632530_Here we show that loss of Ezh2 resulted in altered expression of several key cyclin-dependent kinase inhibitors including Cdkn2a (p16 and Arf) and Cdkn1c (p57) in naive CD8+ T cells after activation, which explained the cell cycle impairment of Ezh2-deficient naive CD8+ T cells in response to antigenic stimulation. 30014245_Cdkn1c expression and methylation may associate with cell cycle exit and differentiation of odontoblasts. 30284969_We propose that CDKN1c activity is restricted to differentiating myoblasts by regulated cyto-nuclear relocalization, coordinating the balance between proliferation and growth arrest. 31924768_Cdkn1c locus regulates cortical development through distinct cell-autonomous and non-cell-autonomous mechanisms. 32024956_Role of the imprinted allele of the Cdkn1c gene in mouse neocortical development. 35523142_p57(Kip2) imposes the reserve stem cell state of gastric chief cells. 36633189_IGF2 interacts with the imprinted gene Cdkn1c to promote terminal differentiation of neural stem cells. 36791055_p57Kip2 is an essential regulator of vitamin D receptor-dependent mechanisms. |
ENSG00000129757 |
CDKN1C |
14.809498 |
10.7037378627 |
3.420043 |
0.98179990 |
11.656541 |
0.00063977174347898207139306858692862078896723687648773193359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0031025161402966744926545938909612232237122952938079833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.7258151946196 |
20.3587909124922 |
2.59029280708195 |
1.52355353066437 |
| ENSMUSG00000037686 |
104816 |
Aspg |
protein_coding |
A0JNU3 |
FUNCTION: Exhibits lysophospholipase, transacylase, PAF acetylhydrolase and asparaginase activities (By similarity). Can catalyze three types of transacylation reactions: (1) acyl transfer from 1-acyl-sn-glycero-3-phosphocholine (1-acyl-GPC) to the sn-1(3) positions of glycerol and 2-acylglycerol (sn-1 to -1(3) transfer), (2) acyl transfer from 1-acyl-GPC to the sn-2 positions of 1-acyl-GPC, 1-acyl-sn-glycero-3-phosphoethanolamine (1-acyl-GPE), and other lysophospholipids (sn-1 to -2 transfer) and (3) acyl transfer from 2-acyl-GPC to the sn-1 position of 2-acyl-GPC and 2-acyl-GPE (sn-2 to -1 transfer) (By similarity). Mediates the synthesis of 1-arachidonoyl species of phospholipids by transferring the arachidonoyl residue from 2-arachidonoyl lysophospholipid to the sn-1 position of 2-acyl lysophospholipid (By similarity). {ECO:0000250|UniProtKB:O88202}. |
Acyltransferase;ANK repeat;Hydrolase;Lipid degradation;Lipid metabolism;Phosphoprotein;Reference proteome;Repeat;Transferase |
|
|
mmu:104816; |
1-alkyl-2-acetylglycerophosphocholine esterase activity [GO:0003847]; acyltransferase activity, transferring groups other than amino-acyl groups [GO:0016747]; asparaginase activity [GO:0004067]; lysophospholipase activity [GO:0004622]; phosphatidyl phospholipase B activity [GO:0102545]; asparagine metabolic process [GO:0006528]; lipid catabolic process [GO:0016042]; phospholipid metabolic process [GO:0006644] |
Human_homologues 21063096_The 60kDa lysophospholipase downregulates basal and Sgk1-dependent ENaC activity. 22861376_In the cleaved state, Thr168 of L-asparaginase directly activates its hydroxyl group (distance of 2.9 A). 24657844_Human 60-kDa lysophospholipase contains an N-terminal L-asparaginase domain that is allosterically regulated by L-asparagine. 24768817_To determine the individual contribution of each of the three conserved active-site threonines (threonine triad Thr168, Thr186, Thr219) for the enzyme-activating autocleavage and asparaginase reactions, we prepared the T168S, T186V and T219A/V mutants. 29554021_Incidence of symptomatic L-asparaginase associated Cerebral Sinovenous Thrombosis during ALL treatment was 1.5% with high case fatality rate (28%). 34461521_Molecular dynamics simulations of human L-asparaginase1: Insights into structural determinants of enzymatic activity. 35979719_In vivo stabilization of a less toxic asparaginase variant leads to a durable antitumor response in acute leukemia. |
ENSG00000166183 |
ASPG |
14.278085 |
5.6388243776 |
2.495394 |
0.90792630 |
6.600834 |
0.01019309845102025699481451681549515342339873313903808593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0340280451415031146011891394209669670090079307556152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
23.1075502545262 |
15.5212946120823 |
4.09793756768333 |
2.15122593992146 |
| ENSMUSG00000037709 |
58909 |
Fam13a |
protein_coding |
Q8BGI4 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:58909; |
Human_homologues cytosol [GO:0005829]; GTPase activator activity [GO:0005096]; regulation of small GTPase mediated signal transduction [GO:0051056]; signal transduction [GO:0007165] |
26862784_A genetic locus within the FAM13A gene has been consistently associated with chronic obstructive pulmonary disease (COPD) in genome-wide association studies. 29386390_Genetic loss of Fam13a exacerbated obesity-related metabolic disorders. 30301961_Fam13a(-/-) mice only exhibited a tendency of higher adiposity and were not protected from DIO and insulin resistance. While Fam13a(-/-) SVCs maintained normal adipogenesis, overexpression of FAM13A in 3T3-L1 preadipocytes downregulated beta-catenin signaling and rendered preadipocytes more susceptible to apoptosis 32029695_Loss of Family with Sequence Similarity 13, Member A Exacerbates Pulmonary Fibrosis Potentially by Promoting Epithelial to Mesenchymal Transition. 32053709_Fam13a expression was reduced in the lungs of mice with hypoxia-induced pulmonary hypertension. Genetic loss of Fam13a exacerbated pulmonary hypertension in mice exposed to chronic hypoxia in association with deteriorated pulmonary artery remodeling. Study data revealed a protective role of Fam13a in the development of pulmonary hypertension. 32193374_GWAS-associated variants within the FAM13A locus alter adipose FAM13A expression, which in turn, regulates adipocyte differentiation and contribute to changes in body fat distribution. 34224973_Tempo-spatial regulation of the Wnt pathway by FAM13A modulates the stemness of alveolar epithelial progenitors. |
ENSG00000138640 |
FAM13A |
158.205505 |
2.0722788557 |
1.051218 |
0.26755790 |
15.325553 |
0.00009048424144792535661358662268938246597826946526765823364257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005434763271824916966087903880122667032992467284202575683593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
213.054739639794 |
39.7393844157129 |
102.811809834387 |
14.568879247342 |
| ENSMUSG00000037762 |
66859 |
Slc16a9 |
protein_coding |
Q7TM99 |
FUNCTION: Proton-linked monocarboxylate transporter. May catalyze the transport of monocarboxylates across the plasma membrane. {ECO:0000250}. |
Cell membrane;Membrane;Reference proteome;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:66859; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; carnitine transmembrane transporter activity [GO:0015226]; creatine transmembrane transporter activity [GO:0005308]; monocarboxylic acid transmembrane transporter activity [GO:0008028]; symporter activity [GO:0015293]; carnitine transmembrane transport [GO:1902603]; creatine transmembrane transport [GO:0015881]; monocarboxylic acid transport [GO:0015718]; urate metabolic process [GO:0046415] |
20022372_In situ hybridization survey of MCT subtypes in placenta detected intense mRNA expression of MCT1, MCT4, & MCT9 (gestational day 11.5 through day 18.5). |
ENSG00000165449 |
SLC16A9 |
97.516004 |
0.4512577499 |
-1.147976 |
0.27595502 |
17.239555 |
0.00003295039768240918756506477316214898110047215595841407775878906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002204218082997174918153532319209375600621569901704788208007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
60.7566684332437 |
11.6923754556413 |
134.638504144525 |
18.6162758694919 |
| ENSMUSG00000037852 |
12876 |
Cpe |
protein_coding |
Q00493 |
FUNCTION: Sorting receptor that directs prohormones to the regulated secretory pathway. Acts also as a prohormone processing enzyme in neuro/endocrine cells, removing dibasic residues from the C-terminal end of peptide hormone precursors after initial endoprotease cleavage. {ECO:0000269|PubMed:10896946, ECO:0000269|PubMed:16219686}. |
Carboxypeptidase;Cleavage on pair of basic residues;Cytoplasmic vesicle;Direct protein sequencing;Disease variant;Glycoprotein;Hydrolase;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes carboxypeptidase E, a prohormone-processing exopeptidase found in secretory granules of endocrine and neuroendocrine cells. The encoded preproprotein undergoes proteolytic processing to generate a mature, functional enzyme that cleaves the C-terminal basic residues of protein substrates. A missense mutation in this gene is responsible for the obesity phenotype in a mouse model known as the 'fat mouse.' Mice lacking the functional product of this gene exhibit impaired processing of multiple peptide hormones such as proinsulin, prodynorphin, proneurotensin, promelanin-concentrating hormone and pro-opiomelanocortin. [provided by RefSeq, Jan 2016]. |
mmu:12876; |
dendrite [GO:0030425]; dense core granule [GO:0031045]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; membrane raft [GO:0045121]; neuronal cell body [GO:0043025]; perikaryon [GO:0043204]; secretory granule [GO:0030141]; secretory granule membrane [GO:0030667]; synaptic membrane [GO:0097060]; transport vesicle membrane [GO:0030658]; carboxypeptidase activity [GO:0004180]; cell adhesion molecule binding [GO:0050839]; cobalt ion binding [GO:0050897]; metallocarboxypeptidase activity [GO:0004181]; molecular function activator activity [GO:0140677]; neurexin family protein binding [GO:0042043]; peptidase activity [GO:0008233]; protein domain specific binding [GO:0019904]; zinc ion binding [GO:0008270]; cardiac left ventricle morphogenesis [GO:0003214]; enkephalin processing [GO:0034230]; insulin processing [GO:0030070]; negative regulation of branching morphogenesis of a nerve [GO:2000173]; peptide catabolic process [GO:0043171]; peptide hormone secretion [GO:0030072]; peptide metabolic process [GO:0006518]; protein localization to membrane [GO:0072657]; protein localization to secretory granule [GO:0033366]; protein processing [GO:0016485]; Wnt signaling pathway [GO:0016055] |
15181368_Data suggest that ischemia has an adverse impact on the neuropeptide-processing system in the brain and that the lack of an active carboxypeptidase E neuropeptide-processing enzyme exacerbates ischemic brain injury. 15358678_CPE KO mice display a wide range of neural and endocrine abnormalities and suggest that CPE may have additional physiological roles beyond those ascribed to peptide processing and sorting of prohormones in cells. 15572367_modifying hypothalamic peptides in Cpe fat/fat mice can cause a decrease in body mass 15618358_Role of carboxypeptidase E in processing of pro-islet amyloid polypeptide in {beta}-cells. 15664176_a specific motif I(16)E(18)I(105)D(106) interacts with CPE to sort proBDNF into regulated pathway vesicles for activity-dependent secretion. 15870393_identification of Cpe(fat)-interacting genetic determinants for obesity and Type II diabetes 16219026_Carboxypeptidase E is required for normal-sized photoreceptor synaptic terminal and normal signal transmission to the inner retina. 16219686_SgIII and CPE form the separate functional sorting complex by anchoring to cholesterol-rich SG membranes, and POMC-derived peptides are transferred from CPE to SgIII, and subsequently to CgA. 17177860_Role of Cpe in murine placentation by determining its expression at various stages of gestation, and by phenotypic analysis of Cpe mutant placentas. 18288386_Six putative caspase substrates, including five novel proteins (ABCF1, AKAP1, CPE, DOPEY1 and GOPC1) that may be targeted specifically by the initiator caspases 8 and 10 during the early stages of apoptosis, were identified.[caspase 10] 18570185_carboxypeptidase E is essential for the survival of adult hippocampal CA3 neurons to maintain normal cognitive function. 18573344_Data show that the carboxypeptidase E cytoplasmic tail binds dynactin that recruits kinesins or dynein for driving bi-directional transport of BDNF vesicles to maintain vesicle homeostasis and secretion in hippocampal neurons. 19428990_CPE expression levels were compared among brain cortex tissue, NS20Y tumor cells and its differentiated cells; more CPE and preproNPY protein expression in the tumor cells were observed 19598241_Our findings suggest that CPE is an important, novel player in mediating appropriate dendritic patterning and spine formation in CNS neurons. 19767734_The obesity susceptibility gene Cpe links FO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. 20299617_These results demonstrate that a 25% increase in body weight is sufficient to augment pulmonary responses to O3, but innate AHR is not manifest until the mice become much heavier. 20460579_patients, bearing CPE and cocaine- and amphetamine-regulated transcript mutations leading to inactive forms of these molecules, may be at a higher risk of developing osteoporosis 20492353_CPE cytoplasmic tail mediates the localization of synaptic vesicles in the actin-rich pre-active zone in hypothalamic neurons and PC12 pheochromocytoma cells. 20655338_The results, whereby RNAi was used to acutely suppress CPE, do not support a role for this protein as necessary for or central to sorting of POMC/ACTH to the regulated secretory pathway in AtT-20 cells. 23418362_There are multiple mechanisms for sorting proglucagon to the regulated secretory pathway, including a role for carboxypeptidase E in pancreatic alpha cells, initial cleavage at K70R71 and multiple sorting signals. 23825125_Data suggests that CPE plays critical role in neuroprotection/allostasis in hippocampal neurons during mild chronic stress; up-regulation of CPE and BCL2 (B cell leukemia/lymphoma 2) expression is possibly mediated by glucocorticoids. 23977080_This study has identified CPE as a new neurotrophic factor that can protect neurons against degeneration. 24843127_Disruption of insulin receptor (IR) expression in beta cells has a direct impact on the expression of the convertase enzyme carboxypeptidase E (CPE) by inhibition of the eukaryotic translation initiation factor Eif4g1. 25009214_Report the effects of ovalbumin sensitization and challenge on oscillatory mechanics of the lung and pulmonary inflammation in obese carboxypeptidase E-deficient mice. 25051500_An anti-inflammatory role of CPE in the intestine by influencing local cytokine levels and thus regulating the migration of myeloid immune cells into the mucosa. 25220258_Carboxypeptidase E is an important modulator of RANKL-induced osteoclast differentiation. 25330741_This study shown that NF-alpha1-mediated upregulation of hippocampal FGF2 and neurogenesis prevents an onset of chronic stress-induced depressive-like behavior 25426952_The neuroprotective effect of CPE-DeltaN against neuronal glutamate neurotoxicity was mediated by secreted FGF2. 26646096_we show by RNA silencing that CPE and SgIII play a synergistic role in the trafficking of POMC to granules of the regulated secretory pathway in AtT20 cells 27709799_NF-alpha1 is critical for regulating antiproliferation and cell fate determination, through differentiating embryonic stem cells to GFAP-positive astrocytes for normal neurodevelopment. 28386642_Cpe knockout leads to dysfunction of BDNF-TRKB signaling in hippocampus and lack of long-term potentiation formation. 30063439_HT22 cells and embryonic cortical neurons overexpressing 40 kDa (CPE/NF-alpha1)-DeltaN exhibited enhanced proliferation, which was inhibited by IGFBP2 short interfering RNA treatment 31300553_It shows that although carboxypeptidase E (CPE) is not directly OGlcNAc modified in islets, overexpression of the suspected OGT target eIF4G1, previously shown to regulate CPE translation in beta-cells, increases islet CPE levels, and fully reverses betaOGTKO islet-induced hyperproinsulinemia. 34266900_Carboxypeptidase E Regulates Activity-Dependent TrkB Neuronal Surface Insertion and Hippocampal Memory. |
ENSG00000109472 |
CPE |
5498.207663 |
0.2685559014 |
-1.896706 |
0.06684167 |
764.081448 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000348037000302373736101338557985656175847330059546282206702629859920148701946148408523857037285809 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000035341997257371713824231804694179138436718574354651179937867512567599541500894863167159403520485 |
Yes |
No |
2321.54128154852 |
107.598083857758 |
8644.53646164101 |
293.027286066935 |
| ENSMUSG00000037860 |
383619 |
Aim2 |
protein_coding |
Q91VJ1 |
FUNCTION: Involved in innate immune response by recognizing cytosolic double-stranded DNA and inducing caspase-1-activating inflammasome formation in macrophages (PubMed:19131592, PubMed:19158675, PubMed:19158676, PubMed:20351692, PubMed:20351693, PubMed:20417169, PubMed:20457908, PubMed:21902795, PubMed:23567559). Upon binding to DNA is thought to undergo oligomerization and to associate with PYCARD initiating the recruitment of caspase-1 precursor and processing of interleukin-1 beta and interleukin-18 (PubMed:19131592, PubMed:19158675, PubMed:19158676, PubMed:20351692, PubMed:20351693, PubMed:20417169, PubMed:20457908, PubMed:21902795, PubMed:23567559). Detects cytosolic dsDNA of viral and bacterial origin in a non-sequence-specific manner (PubMed:19131592, PubMed:19158675, PubMed:20351692, PubMed:23567559). Also acts as a mediator of pyroptosis, necroptosis and apoptosis (PANoptosis), an integral part of host defense against pathogens, in response to bacterial infection (PubMed:34471287). Can also trigger PYCARD-dependent, caspase-1-independent cell death that involves caspase-8 (PubMed:22555457). {ECO:0000269|PubMed:19131592, ECO:0000269|PubMed:19158675, ECO:0000269|PubMed:19158676, ECO:0000269|PubMed:20351692, ECO:0000269|PubMed:20351693, ECO:0000269|PubMed:20417169, ECO:0000269|PubMed:20457908, ECO:0000269|PubMed:21902795, ECO:0000269|PubMed:22555457, ECO:0000269|PubMed:23567559, ECO:0000269|PubMed:34471287}. |
3D-structure;Apoptosis;Cytoplasm;DNA-binding;Immunity;Inflammatory response;Innate immunity;Nucleus;Reference proteome |
|
|
mmu:383619; |
AIM2 inflammasome complex [GO:0097169]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; mitochondrion [GO:0005739]; nucleoplasm [GO:0005654]; double-stranded DNA binding [GO:0003690]; identical protein binding [GO:0042802]; activation of innate immune response [GO:0002218]; apoptotic process [GO:0006915]; cellular response to interferon-beta [GO:0035458]; cellular response to xenobiotic stimulus [GO:0071466]; defense response to virus [GO:0051607]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; negative regulation of NF-kappaB transcription factor activity [GO:0032088]; NLRP3 inflammasome complex assembly [GO:0044546]; pattern recognition receptor signaling pathway [GO:0002221]; positive regulation of cysteine-type endopeptidase activity [GO:2001056]; positive regulation of defense response to virus by host [GO:0002230]; positive regulation of inflammatory response [GO:0050729]; positive regulation of interleukin-1 beta production [GO:0032731]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; pyroptosis [GO:0070269]; pyroptosome complex assembly [GO:1904270]; tumor necrosis factor-mediated signaling pathway [GO:0033209] |
19131592_study showed p202 is an inhibitor of DNA-induced caspase activation; conversely AIM2 was required for caspase activation by cytoplasmic dsDNA; work indicates HIN-200 proteins can act as pattern recognition receptors mediating response to cytoplasmic dsDNA 19158675_observations identify AIM2 as a new receptor for cytoplasmic DNA, which forms an inflammasome with the ligand and ASC to activate caspase-1 19158676_identification of AIM2 as an important inflammasome component that senses potentially dangerous cytoplasmic DNA, leading to activation of the ASC pyroptosome and caspase-1 19158679_AIM2 is a candidate cytoplasmic DNA sensor which shows specificity for double-stranded DNA. 20333626_these data establish a role for DNA sensing through the AIM2 inflammasome in the detection of intracellularly replicating bacteria. 20351692_essential for host defense against cytosolic bacteria and DNA viruses 20351693_critical for innate immunity to Francisella tularensis 20417169_Wild-type Listeria monocytogenes were also found to lyse, albeit at low levels, and trigger AIM2-dependent pyroptosis. 20457908_show that macrophages from mice lacking absent in melanoma 2 (AIM2) cannot sense cytosolic double-stranded DNA and fail to trigger inflammasome assembly. 20490635_as a component of inflammasome,is involved in Caspase-1 activation by Listeria monocytogenes 20562263_Data demonstrate that the third detector senses bacterial DNA and identify it as Aim2. 20566831_Data show that Knockdown of AIM2 significantly decreased the secretion of IL-1beta and IL-18 from L. monocytogenes-infected macrophages 21057088_Aim2 deficiency stimulates the expression of IFN-inducible Ifi202, a lupus susceptibility murine gene within the Nba2 autoimmune susceptibility locus. 21551362_expression of Aim2 protein is required to maintain the expression of the Fcgr2b gene and also predict epistatic interactions between the Ifi200 genes and the Fcgr2b gene within the Nba2 interval 21943709_Our observations demonstrate that cell type and gender-dependent factors differentially regulate the expression of the Aim2 and p202 proteins 22140256_a pathway activating DNA damage signaling plays an important independent role in detecting intracellular foreign DNA, thereby complementing the induction of IFN and activation of the AIM2 inflammasome 22474394_Data show that SdhA is required for preventing AIM2 inflammasome activation. 22555457_The AIM2/ASC complex acts as a novel caspase-8 activation platform and triggers apoptosis of infected Casp1KO macrophages. 22695634_These findings demonstrate that AIM2 plays an important role in M. tuberculosis infection through the recognition of Mtb DNA. 22949200_AIM2 HIN-200 domain in complex with the 15 bp DNA crystallized in the cubic space group I23 or I2(1)3, with unit-cell parameter a = 235.60 A 23045604_Data indicate tha AIM2 is abundantly expressed and inducible. 23479181_AIM2 might act as an important DNA sensor and a potential biomarker for apopDNA-induced macrophage functional maturation and SLE disease 23630667_These data suggest that sublytic caspase-1 activity could delay the formation of stable AIM2 speck, an inflammasome complex associated with cell death. 23645208_Here we show that AIM2 and NLRP3 inflammasomes activate caspase-8 and -1, leading to both apoptotic and pyroptotic cell death. 23850291_Authors hypothesize that while p202 was evolutionarily selected to limit AIM2-mediated inflammation in some mouse strains, the same mechanism contributes to increased interferon production and lupus susceptibility. 23901081_The AIM2 inflammasome is involved in macrophage activation during infection with virulent Mycobacterium bovis strain. 23997220_we report a novel immune evasion mechanism of M. tuberculosis that involves the ESX-1-dependent, direct or indirect, suppression of the host cell AIM2 inflammasome activation during infection. 24484406_The AIM2 inflammasome is a critical molecular platform for regulating IL-1beta release and survival during acute CNS S. aureus infection. 24650018_AIM2 inflammasome expression and activation are further augmented by TLR9 ligands in dietary steatohepatitis. 25327779_Our findings suggest that reactive-oxygen-species-mediated activation of NLRP3 and AIM2 inflammasomes leads to I/R-induced inflammatory responses in which Kupffer cells play a crucial role. 25488991_The Aim2 inflammasome directs maturation of the proinflammatory cytokines IL-1beta and IL-18 and an inflammatory form of cell death called pyroptosis. 25548216_AIM2 responds to endogenous ligands in this setting and contributes to the arthritic phenotype through inflammasome activation. 25562161_For the first time, this paper demonstrated that miR-223 acted to inhibit IL-1beta production via AIM2 pathway, suppressing Kupffer cells pro-inflammatory activation at the early stage of ALF. Thus, it played an important role in the pathogenesis of ALF. 25704009_Mice lacking both AIM2 and NLRP3 fail to confine Aspergillus hyphae to inflammatory foci, leading to widespread hyphal dissemination to lung blood vessels. 25774715_these results reveal a specific requirement for IRF1 and GBPs in the liberation of DNA for sensing by AIM2 depending on the pathogen encountered by the cell. 25774716_this study identified guanylate-binding proteins GBP2 and GBP5 as key activators of AIM2 during infection with F. novicida. 25775556_AIM2 and NLRC4 inflammasomes contribute with ASC to acute brain injury independently of NLRP3 25888795_The comparison shows mouse AIM2 PYD domain adopts a unique alpha2-alpha3 helix conformation distinct from its human homologues, but similar to the pyrin domain of human NLRP10/PYNOD, which belongs to another family. 26095253_Study found that Aim2-deficient mice were hypersusceptible to colonic tumor development. Production of inflammasome-associated cytokines and other inflammatory mediators was largely intact in Aim2-deficient mice; however, intestinal stem cells were prone to uncontrolled proliferation. 26107252_AIM2 reduced Akt activation and tumor burden in colorectal cancer models, while an Akt inhibitor reduced tumor load in Aim2(-/-) mice 26107502_AIM2 inflammsome is functionally redundant in the inflammasomemediated caspase-1 activation and IL-1beta production during P. aeruginosa infection. 26114879_A tissue-specific role for the AIM2 inflammasome as a sensor for endogenous DNA species in the course of a ligand-dependent autoinflammatory condition. 26838050_AIM2 contributes to the maintenance of intestinal integrity via Akt and protects against Salmonella mucosal infection. 26927800_in vitro activation of the AIM2 inflammasome in murine macrophages and dendritic cells leads to reduced activation of the STING pathway, in part through promoting caspase-1-dependent cell death. 27037024_Molecular docking analysis reveals that the conformation switching may be important for the autoinhibition of the AIM2 protein. 27148820_These results show for the first time that AIM2 plays an important anti-inflammatory, yet inflammasome-independent function in cardiomyocytes 27188492_These data provide evidence that the inflammasome components ASC, NLRP3 and AIM2 play a role in regulating macrophage adhesion and activation in response to surface nanotopography and chemistry. 27409673_AIM2 cytosolic DNA sensor may conjugate competitively with cytosolic Mycobacterium bovis DNA to restrict Mycobacterium bovis induced STING-TBK1-dependent autophagy activation and IFN-beta secretion. 27496731_this study shows that aim2-like receptors are dispensable for the type I interferon response to transfected DNA ligands, DNA virus infection, and lentivirus infection 27498865_TRIM11 suppresses AIM2 inflammasome by degrading AIM2 via p62-dependent selective autophagy. 27524110_These results demonstrate a central role for the AIM2 inflammasome in preventing dysbiosis and intestinal inflammation through regulation of the IL-18/IL-22BP/IL-22 and STAT3 pathway 27561456_Studies suggest that absent in melanoma 2 (Aim2) inflammasomes regulate neuronal morphology and influence behaviors. 27846608_AIM2 senses radiation-induced DNA damage in the nucleus to mediate inflammasome activation and cell death. 28424239_results suggest that the function of AIM2 is focused on the innate immune response, and AIM2 is a detrimental host factor for influenza-induced lung injury and mortality. 28465544_lentinan is suggested as an anti-AIM2 and anti-non-canonical inflammasome candidate despite its up-regulation of cytokine expression. 28618410_This study found that Aim2 knockout mitigates Abeta deposition in the cerebral cortex and hippocampus of 5XFAD mice. 28746866_The AIM2 inflammasome amplifies signals of infection, triggering noncanonical activation of NLRP3. During infection, AIM2 and caspase-11 induced membrane damage, which was sufficient and essential for activating the NLRP3 inflammasome. 28947539_study identifies the AIM2 inflammasome and cGAS/IFI16-STING-type I IFN pathway as a novel mechanism for host innate immunity to the ALVAC vaccine vector. 29100888_AIM2 inflammasome activates caspase-1 independent of caspase-8. Caspase-1 and caspase-8 are both upstream of caspase-3 cleavage initiated by AIM2. 29439156_In a mouse model of unilateral ureteral obstruction, Aim2 deficiency attenuated the renal injury, fibrosis, and inflammation observed in wild-type littermates. 29448104_Thus, we demonstrated that AIM2 could promote the progression of atherosclerotic plaque by increasing migration in VSMCs. 29510138_AIM2 overexpression and inhibition were studied in ApoE-/- mice that were fed a high-fat diet. The results showed that high fat diet increases the expression of AIM2. 29886252_NLRs and AIM2 inflammasome-mediated pyroptosis could aggravate BBB damage after TBI. 30012706_neutralization of Aim2 promotes atherosclerotic plaque stabilization 30031129_that AIM2 might serve as a novel prognostic indicator as well as a potential therapeutic target for renal carcinoma treatment 30133699_Recognizing AIM2 as a potential therapeutic target in this disease. 30248400_The increased bacterial burden observed in vivo in AIM2 Knockout mice confirmed that AIM2 is essential for an effective innate immune response against Brucella infection. 30587341_The in vivo short-term metastasis assay using orthotopic implantation into immunodeficient mice demonstrated that oral squamous cell carcinoma cells with high levels of AIM2 expression exhibited enhanced tumor growth in the tongue, resulting in decreased survival of the mice. 30904426_Interferon (IFN)-inducible Absent in Melanoma 2 proteins in the negative regulation of the type I IFN response: Implications for lupus nephritis. 31266383_results indicate that the AIM2 inflammasome is essential for caspase-1-dependent cytokine IL-1beta production and eventual protection from pneumococcal infection in mice. 31301379_AIM2 deletion promotes neuroplasticity and spatial memory of mice. 31332247_AIM2 inflammasome-derived IL-1beta induces postoperative ileus in mice. 31333677_Tyrosine Dephosphorylation of ASC Modulates the Activation of the NLRP3 and AIM2 Inflammasomes. 31412958_S. mansoni infection promotes both NLRP3 and AIM2 inflammasome activation. 31511929_These results demonstrate a fundamental role for AIM2 in energy metabolism, inflammation and insulin resistance. 31845220_Results provide evidence that the activation of AIM2 inflammasome in BALB/c mice was short-lived, which is quite possibly related to the chronicity of MCMV infection. 31862870_Inhibition of AIM2 inflammasome-mediated pyroptosis by Andrographolide contributes to amelioration of radiation-induced lung inflammation and fibrosis. 32053518_Aim2-mediated/IFN-beta-independent regulation of gastric metaplastic lesions via CD8+ T cells. 32171704_Chronic cerebral hypoperfusion activates AIM2 and NLRP3 inflammasome. 32201254_AIM2 inflammasome contributes to brain injury and chronic post-stroke cognitive impairment in mice. 32350463_AIM2 inflammasome surveillance of DNA damage shapes neurodevelopment. 32760056_Radiation causes tissue damage by dysregulating inflammasome-gasdermin D signaling in both host and transplanted cells. 32933486_Deficiency in Aim2 affects viability and calcification of vascular smooth muscle cells from murine aortas and angiotensin-II induced aortic aneurysms. 33505023_AIM2 in regulatory T cells restrains autoimmune diseases. 33707120_AIM2 inflammasome activation may mediate high mobility group box 1 release in murine allergic rhinitis. 33710283_AIM2 controls microglial inflammation to prevent experimental autoimmune encephalomyelitis. 33731931_The AIM2 inflammasome exacerbates atherosclerosis in clonal haematopoiesis. 33866944_Latent Upregulation of Nlrp3, Nlrc4 and Aim2 Differentiates between Asymptomatic and Symptomatic Trichomonas vaginalis Infection. 33963060_The AIM2 and NLRP3 inflammasomes trigger IL-1-mediated antitumor effects during radiation. 34006824_Perfluoroalkyl substance pollutants activate the innate immune system through the AIM2 inflammasome. 34047695_Inflammation drives alternative first exon usage to regulate immune genes including a novel iron-regulated isoform of Aim2. 34085308_Diabetes induces macrophage dysfunction through cytoplasmic dsDNA/AIM2 associated pyroptosis. 34156153_AIM2 deletion enhances blood-brain barrier integrity in experimental ischemic stroke. 34325468_AIM2 regulates anti-tumor immunity and is a viable therapeutic target for melanoma. 34471287_AIM2 forms a complex with pyrin and ZBP1 to drive PANoptosis and host defence. 34474007_AIM2 inflammasome mediates apoptotic and pyroptotic death in the cerebellum following chronic hypoperfusion. 34658166_Nuclear AIM2-Like Receptors Drive Genotoxic Tissue Injury by Inhibiting DNA Repair. 34963084_AIM2 Inflammasome Activation Contributes to Aortic Dissection in a Sporadic Aortic Disease Mouse Model. 35343082_The IL-21-TET2-AIM2-c-MAF pathway drives the T follicular helper cell response in lupus-like disease. 35432843_AIM2 Promotes Gastric Cancer Cell Proliferation via the MAPK Signaling Pathway. 35451371_The AIM2 inflammasome is activated in astrocytes during the late phase of EAE. |
ENSG00000163568 |
AIM2 |
60.170933 |
3.0309421121 |
1.599766 |
0.55829688 |
7.127770 |
0.00758990830787247862387889796309536905027925968170166015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0265220195791497116710733195077409618534147739410400390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
82.0167294115267 |
32.5371221798168 |
27.0598138725184 |
8.04869428329435 |
| ENSMUSG00000037868 |
13654 |
Egr2 |
protein_coding |
P08152 |
FUNCTION: Sequence-specific DNA-binding transcription factor (PubMed:1969796, PubMed:1674431, PubMed:11823429, PubMed:31852952). Plays a role in hindbrain segmentation by regulating the expression of a subset of homeobox containing genes and in Schwann cell myelination by regulating the expression of genes involved in the formation and maintenance of myelin (PubMed:1969796, PubMed:1674431, PubMed:11823429, PubMed:31852952, PubMed:8093858). Binds to two EGR2-consensus sites EGR2A (5'-CTGTAGGAG-3') and EGR2B (5'-ATGTAGGTG-3') in the HOXB3 enhancer and promotes HOXB3 transcriptional activation (PubMed:11823429). Binds to specific DNA sites located in the promoter region of HOXA4, HOXB2 and ERBB2 (PubMed:1969796, PubMed:8093858, PubMed:17938205). Regulates hindbrain segmentation by controlling the expression of Hox genes, such as HOXA4, HOXB3 and HOXB2, and thereby specifying odd and even rhombomeres (PubMed:11823429, PubMed:1674431). Promotes the expression of HOXB3 in the rhombomere r5 and of HOXB3 in r3 and r5 in the hindbrain (PubMed:11823429, PubMed:8093858). Regulates myelination in the peripheral nervous system after birth, possibly by regulating the expression of myelin proteins, such as MPZ, and by promoting the differentiation of Schwann cells (PubMed:7935840, PubMed:10068633). Involved in the development of the jaw openener musculature, probably by playing a role in its innervation through trigeminal motor neurons (PubMed:11509834). May play a role in adipogenesis, possibly by regulating the expression of CEBPB (PubMed:16054051). {ECO:0000269|PubMed:10068633, ECO:0000269|PubMed:11509834, ECO:0000269|PubMed:11823429, ECO:0000269|PubMed:16054051, ECO:0000269|PubMed:1674431, ECO:0000269|PubMed:17938205, ECO:0000269|PubMed:1969796, ECO:0000269|PubMed:31852952, ECO:0000269|PubMed:7935840, ECO:0000269|PubMed:8093858}.; FUNCTION: E3 SUMO-protein ligase helping SUMO1 conjugation to its coregulators NAB1 and NAB2, whose sumoylation down-regulates EGR2 transcriptional activity. {ECO:0000250|UniProtKB:P11161}. |
Acetylation;Activator;Alternative splicing;DNA-binding;Metal-binding;Nucleus;Reference proteome;Repeat;Transcription;Transcription regulation;Transferase;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein sumoylation. |
|
mmu:13654; |
cytoplasm [GO:0005737]; intracellular membrane-bounded organelle [GO:0043231]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; HMG box domain binding [GO:0071837]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; SUMO ligase activity [GO:0061665]; transcription cis-regulatory region binding [GO:0000976]; ubiquitin protein ligase binding [GO:0031625]; aorta development [GO:0035904]; brain segmentation [GO:0035284]; cellular response to organic substance [GO:0071310]; facial nerve structural organization [GO:0021612]; fat cell differentiation [GO:0045444]; gene expression [GO:0010467]; learning or memory [GO:0007611]; motor neuron axon guidance [GO:0008045]; myelination [GO:0042552]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of myelination [GO:0031643]; positive regulation of Schwann cell differentiation [GO:0014040]; positive regulation of transcription by RNA polymerase II [GO:0045944]; protein export from nucleus [GO:0006611]; protein sumoylation [GO:0016925]; regulation of DNA-templated transcription [GO:0006355]; regulation of neuronal synaptic plasticity [GO:0048168]; regulation of ossification [GO:0030278]; regulation of transcription by RNA polymerase II [GO:0006357]; response to insulin [GO:0032868]; rhombomere 3 development [GO:0021569]; rhombomere 3 formation [GO:0021660]; rhombomere 3 structural organization [GO:0021659]; rhombomere 5 formation [GO:0021666]; rhombomere 5 structural organization [GO:0021665]; rhythmic behavior [GO:0007622]; Schwann cell differentiation [GO:0014037]; skeletal muscle cell differentiation [GO:0035914] |
12538520_These results suggest that the dependency of the enhancer on both crest- (Sox10) and r5- (Krox20) specific factors limits its activity to the r5-derived neural crest. 12706208_Krox-20 protein has a role in the maturation of particular brainstem and cerebellar neurons and fluctuations in Krox-20 protein expression coincide with the development of circuitry underlying brainstem-mediated behaviors 12799134_that the role of Krox-20 is to amplify an earlier Krox-20-independent activation of the periaxin gene in Schwann cells 14560009_These results support a role for Egr-1, -2, and -3 in nonlymphoid expression of Fas ligand, TRAIL, and tumor necrosis factor. 14699164_Ddx20 repressed Egr2-mediated transcriptional activation with significant promoter specificity 14757751_Data suggest that Krox-20 can control suppression of mitogenic and death responses, up-regulate the scaffold protein c-Jun NH2-terminal protein kinase (JNK)-interacting protein 1, and regulate JNK activity during Schwann cell development. 15173177_Egr-2 and Egr-3 transcription is enhanced by Hepatitis B virus X protein, which induces fasL gene expression 15585857_Sustained Egr-2 expression is necessary to induce a full anergic state through the actions of genes regulated by transcription factor Egr-2. 15751078_Because glucocorticoids inhibit Egr2/Krox20 expression in osteoblasts and trabecular bone formation is arrested in Egr2/Krox20-knockout mice, inhibition of Egr2/Krox20 activity likely contributes to glucocorticoid-induced osteoporosis. 15834410_Egr-2 and Egr-3 are involved in promoting a T cell receptor-induced negative regulatory genetic program 15836632_Direct induction of cholesterol/lipid biosynthetic genes by Egr2/Krox20 is a part of the myelination program regulated by the sterol regulatory element binding protein (SREBP) pathway. 15886484_Krox-20 regulates the period and extent of Me5 apoptosis, impacting the final number of Me5 neurons. The loss of Me5 in Krox-20-/- mice may highlight species-specific differences in the origin of these cells. 16046619_analysis of an Egr-2-directed transcriptional regulatory network controlled by striatal A2ARs 16054051_Krox20 is necessary for adipogenesis and can stimulate adipogenesis via C/EBPbeta-dependent and -independent mechanisms 16373334_Results suggest that cooperative interactions between Egr2 and Sox10 mediate a large increase in myelin protein zero expression to the high levels found in myelinating Schwann cells. 16477142_This review summarizes studies of Krox-20's impact on the development of motor and sensory systems critical for production of consummatory behaviors (such as suckling/chewing), and its role in the regulation of neurons and muscle in the developing brain. 16495311_Control of Krox20 transcription relies on three very long-range enhancer elements (A, B and C) that are conserved between chick, mouse and human genomes. 16875803_Krox20 as a nodal point of FGF and IGF signalling pathways controlling Igfbp5 expression 16988048_Krox20 is required for the onset of myelination, and it is also crucial for the maintenance of the myelinating state. 17299135_Immediate early gene mRNAs encoding early growth response (Egr)1 and Egr2, which activate the gonadotropin LH beta-subunit gene promoter. 17325040_neuropathy-associated Egr2 mutants antagonize binding of Sox10 at specific sites, thereby disrupting genetic control of the myelination program 17897445_While Krox20 is required for the development of the rhythm-promoting parafacial respiratory group (pFRG) modulating respiratory frequency, Hoxa2 inactivation did not affect neonatal breathing frequency. 17938205_These observations suggest that, in addition to an increased gene copy number and upregulation of EGR2 and CITED1, an elevated erbB2 transcript level involves the loss of 14-3-3sigma, which sequesters a key transcriptional regulator of the erbB2 promoter. 17973254_Krox20 may contribute to the opposing effects of GCs and Wnt signaling on bone formation. 18198176_M-CSF, therefore, promotes osteoclast survival through MEK/ERK-dependent induction of Egr2 to control the Mcl1/Bim ratio, documenting a novel function of Egr2 in promoting survival. 18203138_Helps control the fate of antigen recognition in T cells. 18280047_The results of this study suggestes specific role of egr-2-mediated transcriptional activation in cognitive functions associated with attention. 18456662_Egr2 directly represses genes during myelination in conjunction with NAB corepressors 18524893_When a mutation is introduced into Krox20 in the mouse germ line, the mice develop a severe hypomyelination phenotype that mimics human peripheral neuropathy. 18779345_These results demonstrate that Egr-2 is an intrinsic regulator of effector T cells and controls the expansion of self-reactive T cells and development of autoimmune disease. 18787068_Rostral hindbrain patterning involves the direct activation of a Krox20 transcriptional enhancer by Hox/Pbx and Meis factors. 19000991_MeCP2 and EGR2 show coordinately increased levels during postnatal development of both mouse and human cortex. 19017967_Egr2 plays a central role in the up-regulation of the survival molecule Bcl-2 during positive selection. 19169262_Egr2 is required for the productive selection and terminal maturation of NKT cells via the calcineurin-NFAT pathway. 19218566_Data show diversity in the modes of action of a Krox20 in a single tissue, since regulation of each of the five tested target genes requires different parts of the protein and/or presumably different co-factors. 19229250_Egr1 and Egr2 exert opposing influences on adipocyte differentiation and that the careful regulation of both is required for maintaining appropriate levels of adipogenesis. 19244508_Congenital hypomyelinating neuropathy with lethal conduction failure in mice carrying the Egr2 I268N mutation. 19401217_findings indicate that egr-2 expression is a sensitive indicator of reversal learning and that normal function of D2 receptors in subregions of the frontal cortex is required for cognitive flexibility in tests involving rule reversals 19487693_Early growth response 2 binding to both the intron-associated and upstream enhancers is required for high level induction of myelin protein zero expression in transgenic assays. 19651900_AIP2 regulates activation-induced T-cell death by suppressing EGR2-mediated FasL expression via the ubiquitin pathway 19666526_Egr-2 controls CD4+CD25-LAG3+ regulatory T cells 19706684_Egr transcription factors are essential for conversion of the mitogenic signal of epidermal growth factor into a proliferative response. 19877014_survival function of Egr2 at least partly depends upon its ability to activate the cytokine-mediated survival pathway. 20427655_data suggest that maintenance of the CNS/PNS boundary requires a Krox20 function separable from myelination control 20716776_the antimitogenic role of Krox20 in preosteoclasts is the predominant mechanism underlying the low bone mass phenotype of Krox20-deficient mice 20886122_EGR-2 is not required for CD4 T cell mediated immune responses. 21173153_EGR1 and EGR2 involvement in vertebrate tendon differentiation. 21193208_Inactivation of Krox20/Egr2, a master regulatory gene for myelination in Schwann cells, results in transgression of the CNS/PNS boundary by astrocytes and oligodendrocytes and in myelination of nerve root axons by oligodendrocytes. Review. 21411665_Egr2 and Sox10 activity are directly involved in mediating the developmental induction of Pmp22 expression through an intronic enhancer. 21514423_these findings suggest that Egr-2 plays an important nonredundant role in the pathogenesis of fibrosis. 21533228_results demonstrate that Egr-2 expressed in restricted stages of lymphocyte development plays a dynamic, but similar role for the development of T, NKT and B cells 22049075_Loading-related regulation of transcription factor EGR2/Krox-20 in bone cells is ERK1/2 protein-mediated and prostaglandin, Wnt signaling pathway-, and insulin-like growth factor-I axis-dependent. 22306690_found that TCR-induced transcription factors Egr2 and Egr1 had higher and more-prolonged expression in precursors of the natural killer T (NKT) than in cells of conventional lineages 22451728_Expression of ALDH1A2, BEX2, EGR2, CCL3 and PLAU are upregulated in Toxoplasma gondiisusceptible C57BL/6 mice. 22842221_results identify Egr2 as an important modulator of RANKL-induced osteoclast differentiation and provide the link between RANKL, Egr2 and Id proteins in osteoclast-lineage cells 23021953_discovered that Egr2 and/or Egr3 directly induced expression of suppressor of cytokine signaling-1 (SOCS1) and SOCS3, inhibitors of STAT1 and STAT3, and also blocked the function of Batf, an AP-1 inhibitor, in B and T cells 23129747_Transcriptional regulator early growth response gene 2 (Egr2) is required for T cell anergy in vitro and in vivo. 23203924_Egr-2 induced during T helper (Th)17 cell differentiation serves as a negative feedback inhibitor to control Th17-mediated inflammation. 23261662_These findings support a model in which Egr2-neurons are a critical component of the respiratory chemoreflex into adulthood. 23536637_Mast cell Egr2 was essential for migration of CD4(+) T cells to the inflamed lung. 23548837_ChIP-Seq analysis identified genes that bind Egr2 in anergic T cells, revealing 49 targets that are directly regulated by Egr2. 23716134_Egr2 acts as an intrinsic negative regulator of dendritic cell immunogenicity and can be an attractive molecular target for dendritic cell vaccine development. 23720781_Data indicate that epidermal growth factor receptor (EGFR) signaling stimulates the expression of transcription factor early growth response 2 (EGR2) to promote osteoprogenitor proliferation and survival. 23751188_The results indicated a novel role for Egr2 in repressing adipocyte lineage commitment and promoting early smooth muscle-like cell differentiation. 24061538_A direct, positive autoregulatory loop amplifies and maintains the expression of Krox20, a transcription factor governing vertebrate hindbrain segmentation. 25344368_During heart valve development, Krox20-mediated activation of fibrillar Col1a1 and Col3a1 genes is crucial to avoid postnatal degeneration of the aortic valve leaflets. 25368162_EGR2 promotes peripheral naive T-cell differentiation, with delayed T-cell receptor-induced proliferation in naive T cells from Egr2 conditional knockout mice, and decreased production of cytokines in cells subjected to T-helper differentiation. 25553923_Egr-2 was upregulated by NF-kappaB activation in p50+/+ hippocampal slices. 25864617_Mir106b regulates pro-allergic properties of dendritic cells and Th2 polarisation by targeting Egr-2 in vitro 25979336_demonstrate a novel function of EGR2/3 that is important for Tfh cell development and Tfh cell-mediated B cell immune responses 26466203_let-7 miRNAs promote expression of the myelination-driving master transcription factor Krox20 26519537_The data suggest that Pak2 controls thymic Natural Killer T-cell development by providing a signal that links Egr2 to induce PLZF, in part by regulating signaling lymphocyte activation molecule 6 expression. 26711547_Decreased expression of Krox20 in mice causes degeneration of the aortic leaflets and disorganization of the extracellular matrix, causing valvular dysfunction. 26740329_these results suggested that the basal promoter activity of the mIRF-3 gene is regulated by transcription factors Egr2 and YY1 in NIH3T3 cells 27288457_We discovered that Tead1 and co-activators Yap and Taz are required for Pmp22 expression, as well as for the expression of Egr2 Tead1 directly binds Pmp22 and Egr2 enhancers early in development and Tead1 binding is induced during myelination, correlating with Pmp22 expression. The data identify Tead1 as a novel regulator of Pmp22 expression during development in concert with Sox10 and Egr2 27777310_endogenous KLF4 and Krox20 are dispensable for adipogenesis in culture and for brown adipose tissue development in mice. In contrast, the master adipogenic transcription factor peroxisome proliferator-activated receptor gamma (PPARgamma) is essential. 27911796_Egr2 and Egr3 expressed in T cells cooperatively prevent humoral immune responses by supporting TGF-beta3 secretion. 28169300_Study shows the conversion of mouse skin fibroblasts into induced Schwann cells by driving the expression of two transcription factors, Sox10 and Egr2. 28455436_Egr2 and 3 are antagonists of T-bet function in effector T cells and are important for the control of inflammatory responses of T cells. 28487311_Egr2 and 3 are upstream regulators of effector CD4 and CD8 T cells that are essential for optimal responses with limited immunopathology. 28611210_Prss56 expression was regulated by Egr2 during mouse decidualization. 28723564_study revealed GCN5-mediated EGR2 acetylation as a molecular mechanism that regulates iNKT development. 29158447_findings demonstrate a crucial role for Krox20 in arterial valve development and reveal that an excess of neural crest cells may be associated with bicuspid aortic valve. 29314730_Study findings demonstrated that despite impaired proliferation and IL2 production, tolerant T cells can display inflammatory responses in response to antigen stimulation and this is controlled at least partly by Egr2 and Egr3. 29491350_miR-138 is an Egr2-dependent microRNA but is dispensable for Schwann cell myelination 29781132_Egr2 may play an important role during embryo implantation and decidualization in the mouse. 30012846_data reveal a regulatory circuit involving Egr2-Id3-E2A, which normally restricts the population size of gammadelta NKT cells by adjusting Egr2 dosage and c-Myc expression. 30443252_Egr2 was critical for the expression of transcription factors CEBPbeta and PPARgamma in M2 macrophages, and CEBPbeta was highly expressed in M1-polarized macrophages. 30679703_our data indicate that Gfi1 inhibits the expression of c-Fos, Egr-1 and Egr-2 through direct transcriptional repression and indirect inhibition of Erk1/2 signaling, and that Gfi1-mediated downregulation of c-Fos, Egr-1 and Egr-2 may contribute to the role of Gfi1 in granulopoiesis. 31398266_Hormone-Independent Sexual Dimorphism in the Regulation of Bone Resorption by Krox20. 32646370_EGR2 is elevated and positively regulates inflammatory IFNgamma production in lupus CD4(+) T cells. 32672815_Egr2-guided histone H2B monoubiquitination is required for peripheral nervous system myelination. 32709717_Egr2 and 3 control inflammation, but maintain homeostasis, of PD-1(high) memory phenotype CD4 T cells. 33011290_Early growth response genes 2 and 3 induced by AP-1 and NF-kappaB modulate TGF-beta1 transcription in NK1.1(-) CD4(+) NKG2D(+) T cells. 33060136_The transcription factor EGR2 is the molecular linchpin connecting STAT6 activation to the late, stable epigenomic program of alternative macrophage polarization. 33724178_Egr2 induction in spiny projection neurons of the ventrolateral striatum contributes to cocaine place preference in mice. 33819189_Silencing long non-coding RNA MIAT ameliorates myocardial dysfunction induced by myocardial infarction via MIAT/miR-10a-5p/EGR2 axis. 33986293_Antigen-driven EGR2 expression is required for exhausted CD8(+) T cell stability and maintenance. 34797692_The transcription factor EGR2 is indispensable for tissue-specific imprinting of alveolar macrophages in health and tissue repair. 35168080_Downregulation of mmu_circ_0000943 ameliorates renal ischemia reperfusion-triggered inflammation and oxidative stress via regulating mmu-miR-377-3p/Egr2 axis. 35513123_Susceptibility to kidney fibrosis in mice is associated with early growth response-2 protein and tissue inhibitor of metalloproteinase-1 expression. 35577909_Egr2 contributes to age-dependent vulnerability to sevoflurane-induced cognitive deficits in mice. 35784356_EGR2 Deletion Suppresses Anti-DsDNA Autoantibody and IL-17 Production in Autoimmune-Prone B6/lpr Mice: A Differential Immune Regulatory Role of EGR2 in B6/lpr Versus Normal B6 Mice. 35971091_EGR2 is a hub-gene in myocardial infarction and aggravates inflammation and apoptosis in hypoxia-induced cardiomyocytes. 36208355_BMSC-Derived Exosomal Egr2 Ameliorates Ischemic Stroke by Directly Upregulating SIRT6 to Suppress Notch Signaling. 36646797_Early growth response 2 in the mPFC regulates mouse social and cooperative behaviors. |
ENSG00000122877 |
EGR2 |
187.745759 |
2.6799241320 |
1.422192 |
0.23198653 |
36.897175 |
0.00000000124526228616003772251697607478197055153046335362887475639581680297851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000173380577173580407204225503625671933605190133675932884216308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
269.229039509895 |
43.4790563357977 |
100.461440789337 |
12.5474551099743 |
| ENSMUSG00000037902 |
19261 |
Sirpa |
protein_coding |
P97797 |
FUNCTION: Immunoglobulin-like cell surface receptor for CD47 (PubMed:18045614). Acts as docking protein and induces translocation of PTPN6, PTPN11 and other binding partners from the cytosol to the plasma membrane. Supports adhesion of cerebellar neurons, neurite outgrowth and glial cell attachment. May play a key role in intracellular signaling during synaptogenesis and in synaptic function. Involved in the negative regulation of receptor tyrosine kinase-coupled cellular responses induced by cell adhesion, growth factors or insulin. Mediates negative regulation of phagocytosis, mast cell activation and dendritic cell activation. CD47 binding prevents maturation of immature dendritic cells and inhibits cytokine production by mature dendritic cells (By similarity). Plays a role in antiviral immunity and limits new world arenavirus infection by decreasing virus internalization (PubMed:30726215). Receptor for THBS1 (By similarity). Interaction with THBS1 stimulates phosphorylation of SIRPA (By similarity). In response to THBS1, involved in ROS signaling in non-phagocytic cells, stimulating NADPH oxidase-derived ROS production (By similarity). {ECO:0000250|UniProtKB:P78324, ECO:0000250|UniProtKB:P97710, ECO:0000269|PubMed:18045614, ECO:0000269|PubMed:30726215}. |
3D-structure;Alternative splicing;Direct protein sequencing;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Phosphoprotein;Reference proteome;Repeat;SH3-binding;Signal;Transmembrane;Transmembrane helix |
Human_homologues NA; + ;NA; + ;NA |
|
mmu:19261; |
cell surface [GO:0009986]; plasma membrane [GO:0005886]; cell-cell adhesion mediator activity [GO:0098632]; GTPase regulator activity [GO:0030695]; protein antigen binding [GO:1990405]; protein binding involved in heterotypic cell-cell adhesion [GO:0086080]; protein phosphatase binding [GO:0019903]; protein phosphorylated amino acid binding [GO:0045309]; protein tyrosine kinase binding [GO:1990782]; SH3 domain binding [GO:0017124]; actin filament organization [GO:0007015]; cell migration [GO:0016477]; cell-matrix adhesion [GO:0007160]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to interleukin-1 [GO:0071347]; cellular response to interleukin-12 [GO:0071349]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to type II interferon [GO:0071346]; cytoskeleton organization [GO:0007010]; granulocyte migration [GO:0097530]; hematopoietic progenitor cell differentiation [GO:0002244]; monocyte extravasation [GO:0035696]; negative regulation of chemokine (C-C motif) ligand 5 production [GO:0071650]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; negative regulation of ERK1 and ERK2 cascade [GO:0070373]; negative regulation of I-kappaB phosphorylation [GO:1903720]; negative regulation of inflammatory response [GO:0050728]; negative regulation of interferon-beta production [GO:0032688]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of JNK cascade [GO:0046329]; negative regulation of macrophage inflammatory protein 1 alpha production [GO:0071641]; negative regulation of nitric oxide biosynthetic process [GO:0045019]; negative regulation of phagocytosis [GO:0050765]; negative regulation of protein phosphorylation [GO:0001933]; negative regulation of tumor necrosis factor production [GO:0032720]; phagocytosis, engulfment [GO:0006911]; phagocytosis, recognition [GO:0006910]; positive regulation of phagocytosis [GO:0050766]; positive regulation of T cell activation [GO:0050870]; regulation of gene expression [GO:0010468]; regulation of interleukin-1 beta production [GO:0032651]; regulation of interleukin-6 production [GO:0032675]; regulation of nitric oxide biosynthetic process [GO:0045428]; regulation of tumor necrosis factor production [GO:0032680]; regulation of type II interferon production [GO:0032649] |
12134038_SHP-1 is a critical factor in controlling virus replication in the CNS glia and virus-induced demyelination. 12167615_SHPS-1 contributes to the survival of circulating platelets and down-regulates the macrophage phagocytic response 12446684_role in inhibiting tumor necrosis factor-mediated apoptosis by activation of NF-kappa B 12483539_role in IL-1beta- and TNFalpha-dependent signaling that leads to the activation of Erk 1/2 and Akt 12571228_SHP-1 may be involved in the regulation of beta-catenin transcriptional function and in the negative control of intestinal epithelial cell proliferation 12742627_substrate 1 protein plays an important role in the regulation of glial activation and proliferation in the ischemic CNS. 14500659_SHP-1 may serve as a negative regulator of receptor activator of NF-kappa B ligand (RANKL)-induced intracellular signals and osteoclastogenesis in motheaten viable Mev/Mev mice. 14531999_Data show that surfactant proteins-A and -D bind SIRPalpha through globular heads to block proinflammatory mediator production, while binding to their collagenous tails stimulates proinflammatory mediator production through binding to calreticulin. 14612947_SHPS-1-PLC-gamma complex activates the PLC-PKC cascade, which is required for the activation of IL-1beta-dependent Erk and Akt signalings and cell proliferation 14634079_The interaction between SHPS-1 expressed in phagocytes and CD47 expressed in apoptotic cells works as a tethering step in phagocytosis. 14635051_CD22 is an inhibitory coreceptor for B cell receptor (BCR) signaling. The inhibition is most likely mediated by activation of SHP-1. 14739297_CD47 binds to and thereby down-regulates SHPS-1 on adjacent cells, resulting in inhibition of cell motility 14987753_Lack of SHP-1 results in robust microglial but not astrocyte activation within ablated P14 me/me mouse anteroventral cochlear nucleus. Neuronal death (28%)at P14, a time when normal wild-type littermate controls show little cell death. 15070900_SHP-1 can dephosphorylate alpha-actinin in vitro and in vivo. 15123722_shedding of the ectodomain of SHPS-1 plays an important role in regulation of cell migration and spreading by this protein 15842360_SHPS-1 negatively regulates platelet function via CD47, especially alpha(IIb)beta(3)-mediated outside-in signaling 16141346_SHPS-1 negatively regulates the phagocytosis of RBCs by splenic macrophages, thereby determining both the lifespan of individual RBCs and the number of circulating erythrocytes 16564725_The CD47-SIRPalpha interaction seems to be important in limiting destruction of host cells in experimental models of autoimmune diseases like autoimmune hemolytic anemia (AIHA) or immune thrombocytopenia. 16580635_Phagocytosis of CD47(-/-) RBCs by splenic macrophages from SHPS-1 mutant mice was markedly reduced compared with wild-type macrophages. 16697668_highlight major species differences in CD47-SIRPalpha interactions and CD47 integration, suggesting that signaling by CD47 in man may be qualitatively different from mouse. 16785533_Sirp alpha gene shows a similar expression profile on conventional dendritic cell (DC) subtypes but was also expressed by plasmacytoid pre-DC. 16920950_inhibition by SHPS-1 of phagocytosis in macrophages is mediated, at least in part, in a manner independent of the transinteraction between CD47 and SHPS-1 17109464_Plays an important role in the maturation of Langerhans cells ex vivo and in vivo; SHPS-1-CD47 interaction may negatively regulate contact hypersensitivity. 17475843_SHPS-1 on dendritic cells appears to be essential for prevention of experimental lung cancer metastasis and T helper (Th) type-1 and -2 responses of NKT cells. 17584740_the importance of CD47 varies with cell type, since ligation of CD47 is not necessary for SIRPalpha phosphorylation in myeloid cells, whereas it is required in endothelial cells. 17825046_SHPS-1 is a receptor for Con A that mediates Con A-dependent MMP-9 secretion through SHP-2-promoted activation of both Akt and Erk pathways 18378677_SIRPalpha and PZR serve as scaffolds that facilitate plasma membrane recruitment and signaling of NS-associated SHP-2 mutants. 18450421_SIRP1alpha contributes to IL-6 signalling by counteracting SHP2 phosphorylation which consequently affects ERK-activation and STAT3-dependent transactivation as well as target gene expression. 18455510_SHPS-1 is essential for development of collagen-induced arthritis. 18471880_SIRPalpha may accomplish its inhibitory function in type I interferon induction, in part, through its association and sequestration of the signal transducer PI3K. 18635660_SHPS-1 promotes insulin secretion from beta-cells and thereby protects against diabetes. 18728125_Data show that signal regulatory protein alpha1 is involved in the glucocorticoid receptor effects on the proliferation of murine macrophages. 18809435_SHPS-1 is essential for development of contact hypersensitivity of skin, likely as a result of its positive regulation of the priming by DCs of CD4(+) T cells. 18819922_the presynaptic organizing activity of SIRP-alpha is distinct from FGF22 and is mediated in part by CD47 and its homologues, SIRP-beta and -gamma also have synaptic vesicle clustering activity 19223164_role in xenotransplant tolerance and synaptic patterns organization 19667119_This identifies SHP-1-recruiting inhibitory immunoreceptor signal regulatory protein 1 (SIRPalpha) as a bona fide inhibitory receptor that regulates the bone-resorption activity. 19675159_Sirp alpha-positive conventional dendritic cells (cDCs) have a greater capacity to capture blood-borne antigens than do Sirp alpha-negative cDCs, and may function as specialized antigen presenting cells. 19771158_platelet CD47 down-regulation may be a novel mechanism underneath STEC-induced platelet depletion, and the interactions between CD47 and its receptor, signal regulatory protein alpha (SIRPalpha), play an essential role in modulating platelet homeostasis 19877019_show regulated expression of SIRPa on Kupffer cell following L. donovani infection and propose that SIRPa-CD47 interactions regulate the activation threshold for invariant NKT cytokine production. 19881493_hyperglycemia persistently activates protein kinase C-delta and p38alpha mitogen-activated protein kinase to increase the expression of Src homology-2 domain-containing phosphatase-1, a protein tyrosine phosphatase 20207740_Insulin-like growth factor-I-stimulated insulin receptor substrate-1 negatively regulates Src homology 2 domain-containing protein-tyrosine phosphatase substrate-1 function in vascular smooth muscle cells. 20299253_Inhibition of engulfment correlates with affinity of CD47 for Sirpa - but only at low levels of CD47. 20682853_We now show that the number of CD11c(high) dendritic cells (cDCs), in particular, that of CD8-CD4+ (CD4+) cDCs, is selectively reduced in secondary lymphoid tissues of mice expressing a mutant form of SIRPa that lacks the cytoplasmic region. 20854790_These results suggest that SIRPalpha is indispensable for protective immunity against L. major by the induction of Th1 response. 20955735_SIRPalpha is important for homeostasis of LCs in the skin, as well as of migratory DCs in the LNs, but unlikely for migration of these cells from the skin to draining LNs. 21040253_The data thus suggest that SIRPalpha expressed on CD11c(+) LP cells is important for the production of IL-17 or IFN-gamma in the LP as well as for the development of colitis induced by IL-10 deficiency. 21108471_protein targeting (CD47-Fc fusion protein) protects from Th2-driven allergic airway inflammation 21178002_Thymic signal-regulatory protein (Sirp)alpha-positive conventional dendritic cells (cDCs) and CD8alpha-positive cDCs are equally competent in mediating presentation events to blood-borne hen egg white lysozyme by major histocompatibility class II molecules. 21401967_Data show that myelin down-regulates its own phagocytosis through CD47-SIRPalpha interactions. 21632712_The size of the T cell zone and number of resident CD4-positive T cells in the spleen are markedly reduced in the spleen of SIRPalpha mutant mice. 21638321_Disrupting the CD47-SHPS-1 association leads to imapired osteoclastogenesis. 21775684_our results indicated that SIRPalpha/CD172a regulates eosinophil homeostasis 22042861_provides a rational basis for targeting CD47-SIRPalpha interactions, using for instance the antagonistic antibodies against human SIRPalpha described herein, to potentiate the clinical effects of cancer therapeutic antibodies 22391241_SIRPalpha and Rho kinases interact in the cell migration of dendritic cells in skin. 22468987_Our data indicate that tyrosine phosphorylation of SIRPalpha is an immediate response of neurons in the brain to cold stress. 22671569_These results provide the first demonstration that SHPS-1 plays an important role in the oxidative stress and brain injury induced by acute cerebral ischemia 22815949_thymic Sirpalpha(+) cDCs crucially contribute to a process of intrathymic tumor immune tolerance that involves CCR2 and CCL2 22851710_SIRPalpha demonstrates an indispensable role for protective immunity in Salmonella infection. 22976304_Sirpalpha is not exclusively immunoinhibitory, and illuminate previously unexplained observations implicating Skap2 and Sirpalpha in mouse models of inflammatory disease. 22988019_Our findings demonstrate that B. pseudomallei evades macrophage intracellular killing by preventing the downregulation of SIRPalpha that results in the inhibition of gene expression downstream of the MyD88-independent pathway. 23022485_CD47-SIRPalpha interactions and ITIM-dependent downstream signaling via SIRPalpha control the magnitude of the phagocyte respiratory burst by regulating the expression levels of gp91phox. 23293079_Our data show that efficient human cell engraftment found in NOD-based models is mounted solely by their polymorphic Sirpa. 23472187_genetic induction of the expression of recipient CD47 on xenogeneic donor cells could provide inhibitory signals to recipient macrophages via SIPRalpha 23504624_regulator of FcepsilonRI signaling in mast cells 23504854_Signal regulatory protein alpha is associated with tumor-polarized macrophages phenotype switch and plays a pivotal role in tumor progression. 23515050_SIRP-alpha is part of a novel mechanism for inflammation-mediated insulin resistance in muscle and muscle wasting in chronic kidney disease. 23559001_Downregulation of SHP-1 activity promotes corticospinal tract sprouting and functional recovery after brain injury. 23842779_SIRPalpha is involved in the regulation of podocyte structure and function as a filtration barrier under both physiological and pathological conditions. 23990469_Data indicate that lack of CD47 strongly impairs SIRPalpha-dependent osteoblast differentiation, deteriorate bone formation, and cause reduced formation of osteoclasts. 24036914_Ectodomain shedding of SIRPalpha is an activity-dependent trans-synaptic mechanism for the maturation of functional synapses. 24101669_SIRPA plays a protective role in cardiac hypertrophy through negative regulation of the Toll-like receptor 4/nuclear factor-kappaB pathway. 24143245_'clustering' SIRPalpha into plasma membrane microdomains is essential for activated monocytes and macrophages to effectively interact with CD47 and initiate intracellular signaling 24145514_Using formal genetic, genomic, and immunological analyses, we found that the regulation of phagocytosis previously ascribed to PrP(C) is instead controlled by a linked locus encoding the signal regulatory protein alpha (Sirpa). 24269920_Studies suggest that the mouse strain effect on xenogeneic engraftment might be ascribed mainly to the binding affinity of strain-specific polymorphic SIRPA with human CD47. 24511121_These results suggest that thrombospondin-1 binding to SIRP-alpha on nonphagocytic cells activates NADPH oxidase, limits vasodilation, and promotes renal ischemia reperfusion injury. 24684700_sought to investigate the effect of Hsp70-peptide complex on the expression of CD172alpha and CD47 receptors in normal peritoneal macrophages (NMO) 25236797_These data show a previously unappreciated and crucial role for SIRPalpha in the homeostasis of CD103(+) CD11b(+) DCs in the intestine 25305319_Polymorphism in the innate immune receptor SIRPalpha controls CD47 binding and autoimmunity in the nonobese diabetic mouse. 25817792_data identified SIRPalpha signaling in non-hematopoietic cells to play an important role in regulating the numbers and positioning MZB cell in the spleen 25818708_SIRPalpha intrinsic to CD11c(+) dendritic cells is essential for homeostasis of conventional dendritic cells in the secondary lymphoid organs and skin. 25837251_Velcro' engineering of high affinity CD47 ectodomain as signal regulatory protein alpha (SIRPalpha) antagonists that enhance antibody-dependent cellular phagocytosis 26057870_These findings demonstrate, for the first time, that SIRPalpha signaling can directly control phagocyte migration. 26085683_Loss of cell surface CD47 clustering formation and binding avidity to SIRPalpha facilitate apoptotic cell clearance by macrophages. 26222253_a novel and opposite contribution of SIRPalpha signaling within non-hematopoietic and hematopoietic cells 26907705_TLR5 mediates CD172a(+) intestinal lamina propria dendritic cell induction of Th17 cells. 27091932_These results indicate an important role for CD47-SIRPalpha interactions in innate control of malaria and suggest novel targets for intervention. 27422603_In conclusion, our data suggest that SIRPalpha signaling through SHP-2-PI3K-Akt2 strongly influences osteoblast differentiation from bone marrow stromal cells. 27578867_Mechanistic studies suggest a PKC-Syk-mediated signaling pathway, to which IL-10 conversely inhibits, is required for activating macrophage self-targeting, followed by phagocytosis independent of calreticulin Moreover, we identified spleen red pulp to be one specific tissue that provides stimuli constantly activating macrophage phagocytosis albeit lacking in Cd47(-/-) or Sirpalpha(-/-) mice. 28268219_Treg cells protect dopaminergic neurons against MPP+ neurotoxicity by a cell-to-cell contact mechanism underlying CD47-SIRPA interaction and Rac1/Akt activation. 28545141_Data provide evidence that SIRPalpha-mediated phagocytosis is not a major determinant in prion disease pathogenesis. 28669759_Accumulation of SIRPalpha-inhibited macrophages in tumors favored tumor regression for 1-2 weeks, but donor macrophages quickly differentiated toward non-phagocytic, high-SIRPalpha TAMs. 28801234_these findings have demonstrated how tumor cells inhibit innate sensing in dendritic cells and suggested that the CD47-SIRPalpha axis is critical for dendritic cell-driven antitumor immunity 28978470_CCR7 deficiency results in apoptosis of Sirpa- dendritic cells, which is counterbalanced by expansion of immature Sirpa+ dendritic cells that efficiently induce Treg generation. 29109283_SIRPalpha-expressing dendritic cells regulate homeostasis of fibroblastic reticular cells via TNF receptor ligands in the adult mouse spleen. 29158380_Anti-SIRPA antibody immunotherapy enhances neutrophil and macrophage antitumor activity. 29914984_Tyrosine phosphorylation of SIRPalpha is necessary for SIRPalpha cleavage and presynaptic maturation. 30308165_these results demonstrate that CD47-SIRPalpha signaling prevents excess microglial phagocytosis and show that molecular brakes can be regulated by activity to protect specific inputs 30602457_Microglia are effector cells of CD47-SIRPalpha antiphagocytic axis disruption against glioblastoma. 30770827_Data indicate that only signal regulatory protein alpha (SIRPalpha+) CD8(+) T cells actively proliferate and show cytolytic activity. 30888336_Signal regulatory protein alpha protects podocytes through promoting autophagic activity. 30894504_Serum deprivation and lipopolysaccharide/INF-Gamma induction resulted in increased SIRP-alpha expression and erythrophagocytosis. 30910011_Thus, microglial SIRPalpha suppresses the induction of CD11c(+) microglia that have the potential to accelerate the repair of damaged white matter. 30910815_these results identify SIRPalpha as a counter-receptor for Mac-1 and suggest that the Mac-1-SIRPalpha interaction may be involved in macrophage fusion. 30929259_this study shows that miR-378a modulates macrophage phagocytosis and differentiation through targeting CD47-SIRPalpha axis in atherosclerosis 31099900_SIRPalpha(+) DCs are important for the proliferation and differentiation of Pdpn(+) FRCs in peripheral LNs. 31322787_Dysregulation of SIRPalpha signaling in microglia during aging plays a critical role in the pathogenesis of age-related neurological disorders such as Parkinson's disease. 31370677_Renal tubular epithelial cells from SIRP-alpha(mut) mice produced fewer ROS. 31883525_SIRPalpha inhibits myelin debris phagocytosis in macrophages present in Wallerian degeneration that follows nerve crush injury 32141563_HSF1 promotes the viability of islet beta-cells via upregulating SIRPalpha expression. 32296015_The Ap-2alpha/Elk-1 axis regulates Sirpalpha-dependent tumor phagocytosis by tumor-associated macrophages in colorectal cancer. 32438469_SIRPalpha on CD11c(+) cells induces Th17 cell differentiation and subsequent inflammation in the CNS in experimental autoimmune encephalomyelitis. 32768386_CD47 Ligation Repositions the Inhibitory Receptor SIRPA to Suppress Integrin Activation and Phagocytosis. 33028619_Inflammation Unrestrained by SIRPalpha Induces Secondary Hemophagocytic Lymphohistiocytosis Independent of IFN-gamma. 33074246_Selective SIRPalpha blockade reverses tumor T cell exclusion and overcomes cancer immunotherapy resistance. 33162986_SIRPalpha on Mouse B1 Cells Restricts Lymphoid Tissue Migration and Natural Antibody Production. 33416832_The SIRPalpha-CD47 immune checkpoint in NK cells. 33795678_Loss of microglial SIRPalpha promotes synaptic pruning in preclinical models of neurodegeneration. 34050181_Intratumoral SIRPalpha-deficient macrophages activate tumor antigen-specific cytotoxic T cells under radiotherapy. 34097709_Signal-regulatory protein alpha is an anti-viral entry factor targeting viruses using endocytic pathways. 34197341_Augmentation of CD47/SIRPalpha signaling protects cones in genetic models of retinal degeneration. 34237430_Protein tyrosine phosphatase Shp2 positively regulates cold stress-induced tyrosine phosphorylation of SIRPalpha in neurons. 34917090_Imaging of Glioblastoma Tumor-Associated Myeloid Cells Using Nanobodies Targeting Signal Regulatory Protein Alpha. 35413056_SIRPalpha - CD47 axis regulates dendritic cell-T cell interactions and TCR activation during T cell priming in spleen. 35511221_Endothelial SIRPalpha signaling controls VE-cadherin endocytosis for thymic homing of progenitor cells. 35722884_SIRPalpha Mediates IGF1 Receptor in Cardiomyopathy Induced by Chronic Kidney Disease. 36202053_SIRPalpha maintains macrophage homeostasis by interacting with PTK2B kinase in Mycobacterium tuberculosis infection and through autophagy and necroptosis. 36379210_Neuronal signal-regulatory protein alpha drives microglial phagocytosis by limiting microglial interaction with CD47 in the retina. |
ENSG00000101307+ENSG00000198053+ENSG00000089012 |
SIRPB1+SIRPA+SIRPG |
434.398998 |
0.4797147152 |
-1.059751 |
0.14508301 |
52.389398 |
0.00000000000045517463510700154557813388490734706891705646514267868951719719916582107543945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000095105899066527399377431437001245110186148368214276160870213061571121215820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
276.923630411614 |
27.8954780194184 |
577.267324989198 |
42.3523818682632 |
| ENSMUSG00000037962 |
73121 |
Rflna |
protein_coding |
Q7TS73 |
FUNCTION: Involved in the regulation of the perinuclear actin network and nuclear shape through interaction with filamins. Plays an essential role in the formation of cartilaginous skeletal elements. {ECO:0000269|PubMed:21709252, ECO:0000269|PubMed:24436304}. |
Cytoplasm;Cytoskeleton;Methylation;Reference proteome |
|
|
mmu:73121; |
actin filament bundle [GO:0032432]; cytoplasm [GO:0005737]; filamin binding [GO:0031005]; actin filament bundle organization [GO:0061572]; negative regulation of bone mineralization involved in bone maturation [GO:1900158]; negative regulation of chondrocyte development [GO:0061182]; skeletal system morphogenesis [GO:0048705] |
15661651_cfm does not have any known functional domains, but is conserved in human, chick, Xenopus and zebrafish. A cfm null mutant was live-born without any apparent defects. 24436304_Filamin a,b-interacting proteins, Cfm1 and Cfm2, are essential for the formation of cartilaginous skeleton. |
ENSG00000178882 |
RFLNA |
35.599771 |
0.2899380501 |
-1.786183 |
0.43567670 |
16.659995 |
0.00004471407299472572116974533606992281420389190316200256347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002908133048060043386942585819099349464522674679756164550781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.6462996043282 |
5.10374064655052 |
57.4132977687326 |
12.0308925627922 |
| ENSMUSG00000038028 |
319801 |
Tigar |
protein_coding |
Q8BZA9 |
FUNCTION: Fructose-bisphosphatase hydrolyzing fructose-2,6-bisphosphate as well as fructose-1,6-bisphosphate (By similarity). Acts as a negative regulator of glycolysis by lowering intracellular levels of fructose-2,6-bisphosphate in a p53/TP53-dependent manner, resulting in the pentose phosphate pathway (PPP) activation and NADPH production (PubMed:23726973). Contributes to the generation of reduced glutathione to cause a decrease in intracellular reactive oxygen species (ROS) content, correlating with its ability to protect cells from oxidative or metabolic stress-induced cell death (PubMed:23726973). Plays a role in promoting protection against cell death during hypoxia by decreasing mitochondria ROS levels in a HK2-dependent manner through a mechanism that is independent of its fructose-bisphosphatase activity (By similarity). In response to cardiac damage stress, mediates p53-induced inhibition of myocyte mitophagy through ROS levels reduction and the subsequent inactivation of BNIP3 (PubMed:22044588). Reduced mitophagy results in an enhanced apoptotic myocyte cell death, and exacerbates cardiac damage (PubMed:22044588). Plays a role in adult intestinal regeneration; contributes to the growth, proliferation and survival of intestinal crypts following tissue ablation (PubMed:23726973). Plays a neuroprotective role against ischemic brain damage by enhancing PPP flux and preserving mitochondria functions (PubMed:24872551). Protects glioma cells from hypoxia- and ROS-induced cell death by inhibiting glycolysis and activating mitochondrial energy metabolism and oxygen consumption in a TKTL1-dependent and p53/TP53-independent manner. Plays a role in cancer cell survival by promoting DNA repair through activating PPP flux in a CDK5-ATM-dependent signaling pathway during hypoxia and/or genome stress-induced DNA damage responses (By similarity). Involved in intestinal tumor progression (PubMed:23726973). {ECO:0000250|UniProtKB:Q9NQ88, ECO:0000269|PubMed:22044588, ECO:0000269|PubMed:23726973, ECO:0000269|PubMed:24872551}. |
Apoptosis;Autophagy;Cytoplasm;Hydrolase;Mitochondrion;Nucleus;Reference proteome |
|
|
mmu:319801; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; bisphosphoglycerate 2-phosphatase activity [GO:0004083]; fructose-2,6-bisphosphate 2-phosphatase activity [GO:0004331]; cardiac muscle cell apoptotic process [GO:0010659]; cellular response to cobalt ion [GO:0071279]; cellular response to DNA damage stimulus [GO:0006974]; cellular response to hypoxia [GO:0071456]; fructose 1,6-bisphosphate metabolic process [GO:0030388]; fructose 2,6-bisphosphate metabolic process [GO:0006003]; glucose catabolic process to lactate via pyruvate [GO:0019661]; glycolytic process [GO:0006096]; intestinal epithelial cell development [GO:0060576]; mitophagy [GO:0000423]; negative regulation of glucose catabolic process to lactate via pyruvate [GO:1904024]; negative regulation of glycolytic process [GO:0045820]; negative regulation of kinase activity [GO:0033673]; negative regulation of mitophagy [GO:1901525]; negative regulation of neuron death [GO:1901215]; negative regulation of programmed cell death [GO:0043069]; negative regulation of reactive oxygen species metabolic process [GO:2000378]; positive regulation of cardiac muscle cell apoptotic process [GO:0010666]; positive regulation of DNA repair [GO:0045739]; positive regulation of hexokinase activity [GO:1903301]; reactive oxygen species metabolic process [GO:0072593]; regulation of pentose-phosphate shunt [GO:0043456]; regulation of response to DNA damage checkpoint signaling [GO:1902153]; response to gamma radiation [GO:0010332]; response to ischemia [GO:0002931]; response to xenobiotic stimulus [GO:0009410] |
20935145_p53 and TIGAR inhibit glycolysis in hypoxic myocytes and that inhibition of glycolysis is closely involved in apoptosis, suggesting that p53 and TIGAR are significant mediators of cellular energy homeostasis and cell death under ischemic stress. 22044588_p53/TIGAR-mediated inhibition of myocyte mitophagy is responsible for impairment of mitochondrial integrity and subsequent apoptosis. 23726973_TIGAR has roles in efficient intestinal regeneration and tumorigenesis 24509157_Therefore, we conclude that TIGAR knockdown-induced radiosensitization of glioma cells may be dependent on the inhibition of TRX1 nuclear translocation. 24872551_TIGAR protects ischemic brain injury and preserves mitochodrial function. 25445985_TIGAR protein expression in brain is increased following ischemia reperfusion injury. 26219221_Results suggest that TIGAR expression changes during development and its expression level may be correlated with the vulnerability of neurons to ischemic injury. 26247727_Although mouse TIGAR expression is clearly induced in the intestines of mice following DNA-damaging stress of ionizing radiation, that was not dependent on p53 or TAp73. 27256465_TIGAR contributes to ischemic tolerance induced by cerebral preconditioning through scavenging of reactive oxygen species and inhibition of apoptosis. 29331305_Study demonstrated that TIGAR played a positive role in survival of astrocytes and a negative role in inflammatory response after stroke attack. TIGAR exerted these functions through increasing flux of pentose phosphate pathway and inhibiting NF-kappaB activation. 29587485_The study is the first to indicate that Wnt signaling activates TIGAR and protects SGNs against cisplatin-induced damage through the inhibition of oxidative stress and apoptosis in SGNs, and this might offer novel therapeutic targets for the prevention of SGN injury. 29776966_These data indicate a role for TIGAR in modulating redox homeostasis during oocyte maturation. 30726106_TIGAR regulates mitochondrial function through SIRT1-PGC1alpha and translocation of TIGAR into mitochondria, which contribute to exercise endurance of mice. 30977208_The results suggest that p53-TIGAR metabolic axis-mediated glycolytic suppression may play a compensatory role in attenuating DNA damage and proliferative exhaustion in Fanconi Anemia hematopoietic stem cells. 30978385_this study shows that TIGAR alleviates ischemia/reperfusion-induced autophagy and ischemic brain injury 32632140_TIGAR/AP-1 axis accelerates the division of Lgr5(-) reserve intestinal stem cells to reestablish intestinal architecture after lethal radiation. 32946259_TIGAR reduces smooth muscle cell autophagy to prevent pulmonary hypertension. 33512431_Impaired ribosome biogenesis checkpoint activation induces p53-dependent MCL-1 degradation and MYC-driven lymphoma death. 33586458_Sirtuin 3 Alleviates Diabetic Cardiomyopathy by Regulating TIGAR and Cardiomyocyte Metabolism. 33928637_Regulatory role of TIGAR on endothelial metabolism and angiogenesis. 33994201_TIGAR mitigates atherosclerosis by promoting cholesterol efflux from macrophages. 34826530_TIGAR plays neuroprotective roles in KA-induced excitotoxicity through reducing neuroinflammation and improving mitochondrial function. 35254259_TIGAR deficiency enhances skeletal muscle thermogenesis by increasing neuromuscular junction cholinergic signaling. 35421414_The inhibition of TRIM35-mediated TIGAR ubiquitination enhances mitochondrial fusion and alleviates renal ischemia-reperfusion injury. 35441828_TIGAR deficiency sensitizes angiotensin-II-induced renal fibrosis and glomerular injury. 35576689_TIGAR alleviates oxidative stress in brain with extended ischemia via a pentose phosphate pathway-independent manner. 35621078_Knockout of TIGAR enhances myocardial phosphofructokinase activity and preserves diastolic function in heart failure. |
ENSG00000078237 |
TIGAR |
61.542273 |
2.6901338217 |
1.427678 |
0.36503101 |
14.930289 |
0.00011155746604542667249973519894012952136108651757240295410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006596441470512185616462130788306694739731028676033020019531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
89.9936079092957 |
23.0139360173653 |
33.4532086042575 |
6.61548228274434 |
| ENSMUSG00000038068 |
218215 |
Rnf144b |
protein_coding |
Q8BKD6 |
FUNCTION: E3 ubiquitin-protein ligase which accepts ubiquitin from E2 ubiquitin-conjugating enzymes UBE2L3 and UBE2L6 in the form of a thioester and then directly transfers the ubiquitin to targeted substrates such as LCMT2, thereby promoting their degradation. Induces apoptosis via a p53/TP53-dependent but caspase-independent mechanism. However, its overexpression also produces a decrease of the ubiquitin-dependent stability of BAX, a pro-apoptotic protein, ultimately leading to protection of cell death; But, it is not an anti-apoptotic protein per se (By similarity). {ECO:0000250}. |
Apoptosis;Cytoplasm;Membrane;Metal-binding;Mitochondrion;Reference proteome;Repeat;Transferase;Transmembrane;Transmembrane helix;Ubl conjugation;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:218215; |
cytoplasm [GO:0005737]; mitochondrial membrane [GO:0031966]; ubiquitin ligase complex [GO:0000151]; metal ion binding [GO:0046872]; ubiquitin conjugating enzyme binding [GO:0031624]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin-protein transferase activity [GO:0004842]; apoptotic process [GO:0006915]; negative regulation of apoptotic process [GO:0043066]; positive regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032436]; protein polyubiquitination [GO:0000209]; ubiquitin-dependent protein catabolic process [GO:0006511] |
20615966_PIR2, by being induced by TAp73 and degrading DeltaNp73, differentially regulates TAp73/DeltaNp73 stability.[PIR2] |
ENSG00000137393 |
RNF144B |
304.637372 |
0.3773820528 |
-1.405902 |
0.17526520 |
63.361679 |
0.00000000000000172032409577140358016770191669048627940487598744073727274894736183341592550277709960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000457311983015532607975016531657243910299675757058501801566308131441473960876464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
167.5124793077 |
20.4002764688316 |
443.880354336505 |
38.8350446447404 |
| ENSMUSG00000038086 |
69253 |
Hspb2 |
protein_coding |
Q99PR8 |
FUNCTION: May regulate the kinase DMPK. {ECO:0000250}. |
Alternative splicing;Cytoplasm;Nucleus;Reference proteome;Stress response |
|
|
mmu:69253; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; structural constituent of eye lens [GO:0005212]; unfolded protein binding [GO:0051082]; protein refolding [GO:0042026]; response to heat [GO:0009408]; somatic muscle development [GO:0007525] |
12112005_results strongly suggest that glucocorticoid stimulates the induction of neither HSP70 nor HSP90, but HSP27 in osteoblasts, and that p38 MAP kinase is involved in the induction of HSP27 12403771_intergenic enhancer elements have orientation-dependent influence on MKBP 12430713_AlphaB-crystallin and HSP27 were upregulated in the spinal cord of mutant SOD1 mice compared to mice overexpressing wild-type SOD1. HSP70 levels were normal. 12639970_neuroprotective effects of overexpression in transgenic animals against kainate-induced seizures and hippocampal cell death 14592939_whereas neither alphaBC nor HSPB2 is essential for myocardial development and function under nonstressful conditions, one or both are required for maximal functional recovery and protection from I/R-induced necrosis and apoptosis. 15309411_mice over expressing Heat shock protein 27 (verified by Western blotting analysis) were found to be protected from lethal ischaemia/reperfusion injury compared to their negative littermates 15509539_stathmin expression is disregulated, and Hsp25, Hsp27, and peroxiredoxin 6 are upregulated in a mouse model of familial amyotrophic lateral sclerosis 16217658_This study suggests that, during ischemic insults, alphaB-crystallin/HSPB2 may not be important for the contraction process itself, but rather serve to maintain muscular elasticity. 16514645_Phosphatidylinositol 3-kinase/Protein Kinase B plays a part in the sphingosine 1-phosphate-stimulated induction of HSP27 16678848_These findings indicate an important role of CRYAB and/or HSPB2 in mitochondrial function. 16817323_The small heat shock protein sHSP27 exists as both dimers and large multimers and is distributed homogeneously throughout the cytoplasm. 17015449_MK2 mediates the incorporation of p38 into the pre-existing complex of hsp27 with Akt 17360721_Study is the first to suggest a differential modulation of Hsp27 activation in vivo and illustrates the diverse effect of Hsp27 on acute versus chronic models of disease. 17439945_first demonstration of a specific interaction between HspB1 and a stratum corneum protein and indicates that HspB1 has chaperone activity during stratum corneum formation 17481944_Findings indicate that Hsp27 may play a key role in resistance to doxorubicin-incuded cardiac dysfunction. 17510974_heat shock protein 27 17846079_CryAB and HSPB2 proteins play nonredundant roles in the heart, CryAB in structural remodeling and HSPB2 in maintaining energetic balance 17873008_We discuss the implications of these disparate results in the context of phenotypic responses reported for CRYAB/HSPB2-deficient mice to different ischemic challenges. 17939115_mouse alphaB-crystallin and MKBP/HspB2 promoter activities are regulated by shared and gene specific intergenic elements 18255302_This study provides further support for the therapeutic potential of HSPs in ALS and other motor neuron disorders. 18263706_HSP27 binding to p53 attenuated the doxorubicin toxicity by upregulating p21 and prevented cell death. 18368538_Data suggest that Brn-3a and b POU factors may play an important role in the development and maintenance of critical cell types and structures within the heart, in part via developmental regulation of myocardial Hsp27 expression. 18392951_Results show that bicyclol induces hepatic HSP27/70 expression via activation of HSF1 and that the cytoprotective action of bicyclol against liver injury is mediated by its induction of HSP27/70. 18566345_Extracellular release of the atheroprotective heat shock protein 27 is mediated by estrogen and competitively inhibits acLDL binding to scavenger receptor-A. 19052195_we conclude that overexpression of Hsp27 confers long-lasting neuroprotection against ischemic brain injury via a previously unexplored association and inhibition of ASK1 kinase signaling. 19109201_HSP27 augments cytokine production and the induction of allergic contact hypersensitivity by dendritic cells; HSP27 is a key participant in the induction of immune responses to topically applied haptens. 19513538_Rho-kinase regulates sphingosine 1-phosphate-stimulated induction of HSP27 at a point upstream of p38 MAP kinase in osteoblasts. 19997117_Results provide support for the powerful neuroprotective effects of HSP27 in cerebral ischemia. 20027617_Hspb2 promoter drives Cre expression in spermatocytes of transgenic mice. 20382860_Therapeutic hypothermia cardioprotection is mediated by enhanced Akt/HSP27 phosphorylation and enhanced NO generation, resulting in the attenuation of reactive oxygen species generation and the maintenance of DeltaPsim following ischemia-reperfusion. 20465849_Downregulation of both gene expression and activity of Hsp27 improved oocyte maturation in vitro. 20566634_p27 suppresses Hsp27 and Hsp70 expression at the transcriptional level specifically through JNK2/c-Jun- and HSF-1-dependent pathways upon arsenite exposure, providing additional important molecular mechanisms for the tumor-suppressive function of p27. 20693246_Dexamethasone inhibits ACh-induced ASM contraction through phosphorylation of p38 MAPK and HSP27 20871982_These results imply that HspB2 and HspB7 might be up-regulated by ethanol during the short-term differentiation of neural stem cells. 21225223_HSP27 induction is critical for TGF-beta-induced VEGF release in osteoblasts. 21544852_This suggests that the increased tau phosphorylation at S396/404 and S262 sites may be induced by Hsp27 through enhancement of GSK3beta activity in the mouse cerebellum. 21837772_these results demonstrate that disruption of the primary cilia structure or function compromises the endothelium through the suppression of hsp27 dependent actin organization and focal adhesion formation 22023707_Hsp27 participates in the maintenance of breast cancer stem cells through regulation of epithelial-mesenchymal transition 22027643_HSP27 may serve as an indicator of stimulus strong enough to show its expression. 22357851_This study demonstrated that HSP27 requires PKD-mediated phosphorylation for its suppression of ASK1 cell death signaling and neuroprotection against ischemic injury. 22870288_HSPB2 can be replaced by other members of the multigene small HSP family under basal conditions while HSPB2 is implicated in the regulation of metabolic/mitochondrial function under cardiac stress such pressure overload. 22971995_Tubular expression of heat-shock protein 27 inhibits fibrogenesis in obstructive nephropathy. 23117011_HSP27 is increased in hyperlipidemic animals treated with reconstituted high-density lipoprotein. 23185379_activation of PP2A or inactivation of the p38MAPK-MAPKAPK2-Hsp27 has a role in survival of cancer stem cells under hypoxia and serum depletion via decrease in PP2A activity 23409070_Chronic over-expression of HSP27 is atheroprotective in both sexes and coincides with reductions in lesion cholesterol accumulation as well as favorable plaque remodeling. 23469858_findings reveal a novel role for HSP27 in the maintenance of BBB integrity. 23605646_Spatial learning was impaired in AD model mice rescued by Hsp27 overexpression. 23939398_Heat shock protein-27 attenuates foam cell formation and atherogenesis by down-regulating scavenger receptor-A expression via NF-kappaB signaling. 24025642_protective role for endothelial S1P1R against ischemic acute kidney injury most likely by regulating endothelial barrier integrity and endothelial HSP27 expression 24219791_It plays a protective role against AP-induced liver injury. 24918749_The myocardium releases HSP27 after global ischemia: the proinflammatory effect of extracellular HSP27 through TLR2 and TLR4. 25187648_Hsp27 binding to the 3'UTR of Bim mRNA prevents neuronal death during oxidative stress-induced injury. 25840912_These findings indicate that in mice HSP27 and HSP70 play a key role in the induction of cell-mediated immunity to carcinogenic polyaromatic hydrocarbons 26329039_The phosphorylation of S82 of the heat shock protein 27 kDa (HSP27), was especially abundant and consistently upregulated in tissues with delays in freezing as short as 2 minutes. 26465331_HspB2 has roles in maintaining ATP levels and as well as chaperone-dependent functions for metabolic homeostasis 26666831_Heat shock proteins 27 and 70 were involved in the protective effect. 27185187_phosphorylated Hsp27 stimulates autophagy and lipid droplet clearance and interacts with STAT3. 27909724_HSP27 expression is upregulated in lung fibroblasts during pulmonary fibrosis, and subsequently, HSP27 modulates lung fibroblast differentiation through the Smad3 and ERK pathways. 28595321_We found significantly slower retrograde transport of mitochondria in Ser135Phe, Pro39Leu and Arg140Gly mutant Hsp27 expressing motor neurons than in wild type Hsp27 neurons, although anterograde movement velocities remained normal. 29232010_HSP27 exerts its protective role by suppressing reactive oxygen species and atherosclerosis progression by inhibiting mitochondria apoptosis pathway in coronary heart disease. 29650953_HSP27 is a partner of JAK2-STAT5 and a potential therapeutic target in myelofibrosis. |
ENSG00000170276 |
HSPB2 |
175.588161 |
0.3648454445 |
-1.454643 |
0.19783115 |
53.407519 |
0.00000000000027105740734080447258208224671481617373868314158080750075896503403782844543457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000057744705295316561070413561077353158329864002418219115497777238488197326660156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
93.0440293380592 |
12.798788070617 |
255.023135818727 |
24.8262738828069 |
| ENSMUSG00000038141 |
77106 |
Tmem181a |
protein_coding |
A0A338P7C9 |
Human_homologues FUNCTION: Mediates action of cytolethal distending toxins (CDT), which are secreted by many pathogenic bacteria. Expression level of TMEM181 is rate-limiting for intoxication. {ECO:0000269|PubMed:19965467}. |
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
|
membrane [GO:0016020]; toxic substance binding [GO:0015643] |
|
ENSG00000146433 |
TMEM181 |
495.741262 |
0.4617284592 |
-1.114883 |
0.12290459 |
81.874622 |
0.00000000000000000014499921837412272372393550345856844041831325810476869360085561133644205256132408976554870605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000055354087575805440323759425138757289065872482807960028305949151672393782064318656921386718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
316.630245412832 |
27.0066758505292 |
685.749901544285 |
42.1292457993825 |
| ENSMUSG00000038146 |
18131 |
Notch3 |
protein_coding |
Q61982 |
FUNCTION: Functions as a receptor for membrane-bound ligands Jagged1, Jagged2 and Delta1 to regulate cell-fate determination. Upon ligand activation through the released notch intracellular domain (NICD) it forms a transcriptional activator complex with RBPJ/RBPSUH and activates genes of the enhancer of split locus. Affects the implementation of differentiation, proliferation and apoptotic programs (By similarity). May play a role during CNS development. {ECO:0000250|UniProtKB:Q9R172, ECO:0000250|UniProtKB:Q9UM47}. |
Activator;ANK repeat;Cell membrane;Developmental protein;Differentiation;Disulfide bond;EGF-like domain;Glycoprotein;Membrane;Methylation;Notch signaling pathway;Nucleus;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transcription;Transcription regulation;Transmembrane;Transmembrane helix |
|
|
mmu:18131; |
actin cytoskeleton [GO:0015629]; cell surface [GO:0009986]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; calcium ion binding [GO:0005509]; enzyme binding [GO:0019899]; identical protein binding [GO:0042802]; signaling receptor activity [GO:0038023]; artery morphogenesis [GO:0048844]; axon guidance [GO:0007411]; forebrain development [GO:0030900]; glomerular capillary formation [GO:0072104]; negative regulation of cell differentiation [GO:0045596]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neuroblast differentiation [GO:0014016]; neuron differentiation [GO:0030182]; neuron fate commitment [GO:0048663]; Notch signaling pathway [GO:0007219]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355] |
11700858_component in spermatogenesis and Sertoli cells in neonatal mice 12126955_expression pattern analyzed in the pre- and postnatal mouse brain; data show expression is conserved between mouse and human and suggest that the mouse is a valid system for analysis of CADASIL 12391150_Data describe the role for p300 in the potentiation of Notch-1 and -3 IC function by MAML1 and PCAF. 12673204_Ectopic expression of Notch3 in airway epithelium potentially contributes to the multistep evolution of lung cancer through the inhibition of terminal differentiation. 15076712_Notch3 is observed in the lower compartment of developing olfactory neuroepithelium until it gradually disappears, which suggests a distinct role for Notch3 in the neurogenesis of the peripheral olfactory system. 15857853_Heterodimerization of mutant Notch3 with Fringe is associated with pathophysiology of cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy 15882997_results support the idea that other Notch genes functionally compensate for Notch3 during embryonic development 16186803_activated Notch3 can function as an oncogene in the developing brain, and link the Notch pathway to human choroid plexus tumor pathogenesis 16507912_Notch3 plays role in the development and transformation of the mammary gland. 16508304_Infection of embryonic day 9.5 telencephalic progenitors in vivo with Notch 3 promoted radial glial/progenitor character embryonically and astrocyte fate postnatally. 16518823_Expressed in cells retained within the lamina propria and in olfactory ensheathing cells. 17332745_Notch3 and the Notch3-upregulated RNA-binding protein HuD regulate Ikaros alternative splicing. 17981798_These results offer a novel mechanism by which TGF-beta1 promotes the expression of smooth muscle differentiation genes through the inhibition of Notch3 and activation of Hes1. 18299578_FIH-1-dependent hydroxylation of N1945 and N2012 in the Notch1-4 ICD; FIH-1 negatively regulates Notch ICD and accelerates myogenic Differentiation 18347334_Cellular and molecular analyses of vascular smooth muscle cells derived from these animals associate Notch 3 activity to the expression of specific gene targets. 18483410_PDGF receptor (PDGFR)-beta is a novel immediate Notch target gene. PDGFR-beta expression was upregulated by Notch ligand induction or by activated forms of the Notch receptor; dysregulated Notch signaling perturbs VSMC differentiation and function. 18758477_Mutation, amplification & deregulated expression of Notch3 have been correlated with the disruption of cell differentiation in transgenic mice & to development of leukemia & other diseases in mice. Review. 18818417_Notch3 plays an important role in the control of vascular mechano-transduction, by modulating the RhoA/Rho kinase pathway, with opposite effects on myogenic tone and flow-mediated dilation in the resistance circulation. 19147503_TSP2 binds directly to Notch3 and Jagged1. TSP1 also binds to Notch3 and Jagged1; however, only TSP2 augments the interaction between Notch3 and Jagged1. 19446551_FRalpha regulates pituitary tumor cell proliferation and mechanistically may involve the NOTCH pathway 19640841_Data demonstrate that extracellular YB-1 associates with outer cell membrane components and interacts with extracellular Notch-3 receptor domains. 19735738_critical role for lysosomes in the degradation 19897741_Lfng functions to enhance Notch signaling in myofibroblast precursor cells and thereby to coordinate differentiation and mobilization of myofibroblasts required for alveolar septation. 19966856_Data show that that the increased Notch3-IC degradation correlates with higher levels of c-Cbl tyrosine phosphorylation in Notch3-IC/pTalpha(-/-) double-mutant thymocytes. 20472562_LRP1 and TSP2 stimulate Notch activity by driving trans-endocytosis of the Notch ectodomain into the signal-sending cell 20689064_Notch3 is important for the investment of mural cells and is a critical regulator of developmental and pathological blood vessel formation 20960513_Notch3 pathway might be distinct from Notch1 in muscle regeneration. 20967782_Dominant mutations in NOTCH3 cause cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL). 21235539_Data suggest that selective inhibition of Notch signalling mediated by Notch3 or Delta-like1 may offer a new strategy for the treatment of RA. 21372592_Notch-3 expression is significantly upregulated in renal cells from mice with a targeted disruption of one Y box (YB)-1 allele. 21508258_Data indicate that the cooperation among Notch3, protein kinase C, and p65/NF-kappaB subunit modulates Foxp3 expression. 21518968_Data show that Notch3 is necessary for the adaptive response of the renal vasculature to vasoactive systems. 21536678_mechanism of Notch activation 21555590_cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy is associated with hypomorphic Notch 3 function in vascular smooth muscle cells. 21768299_Notch3 is differentially expressed by progenitor thymocytes, peaking at the DN3 progenitor stage. Notch1 & Notch3 carry out nonoverlapping functions during thymocyte differentiation. Notch3 is not essential at later stages. 21912215_Suppression of Notch3 disrupts quiescence and promotes expansion of self-renewing populations. 22615991_In vitro analysis show that both Notch2 and Notch3 robustly activate smooth muscle differentiation genes, and Notch3, but not Notch2 is a target of Notch signaling 22806125_Notch-3 receptor activation drives inflammation and fibrosis following tubulointerstitial kidney injury. 23055528_Mef2c induces microRNAs miR-1 and miR-206, which directly down-regulate Notch3 and allow differentiation to proceed. 23117660_Identify Grip2 and Notch3 target regulating myogenic response of cerebral vessels. 23492191_Ccr2 deficient mice display increased Notch3 signaling and dysregulated Notch ligand expression. 23675950_Data indicate that expression of Dll4 is specific to theca layer endothelial cells (ECs); Notch1/Notch4 in theca layer ECs and vascular smooth muscle cells (VSMCs), Notch3 is restricted to VSMCs; Notch2 in granulosa cells (GCs) of small follicles. 23928992_High notch3 expression is associated with inflammatory breast cancer that frequently metastasized. 24015274_The transcription and expression patterns of Notch pathway components (Notch 1-3, Delta1 and 4, Jagged1) and effectors (Hes1, Hes2, Hes5 and Nrarp) were evaluated in the mouse testis 24100291_Notch3 is expressed in a highly clonogenic and transiently quiescent luminal progenitor population that gives rise to a ductal lineage. 24425116_cerebral autosomal dominant arteriopathy with subcortical infarcts and leucoencephalopathy can develop without impairment of NOTCH3 signaling 24723319_Stroke outcome and memory impairments are worse in notch3 null mice; lack of the notch3 receptor affects white matter stroke susceptibility. 24825564_These data indicate that both mTORC1 and Notch3 signaling are involved in HPH and the downstream effects of mTORC1 activation in HPH are partially dependent on the activation of Notch3 signaling. 24943269_study demonstrates activity-dependent reduction of DLL4 expression and proteolytic cleavage of Notch3 in the hypothalamic-neurohypophysial system. 25100656_Notch signaling controls the overall tempo of retinogenesis through Atoh7 and Neurog2, by integrating cell fate specification, the wave of neurogenesis and the developmental status of cells ahead of this wave 25164209_Loss of Notch3 is associated with impaired neuronal maturation. 25381438_Notch3 activation is sufficient but not required for inducing human T-lineage specification. 25421557_Notch3 protein activation in glomeruli promotes the development of progressive renal disease. 25477343_Notch3 is important for survival of vascular smooth muscle cells, and reveal a critical role for Notch3 and VSMC in blood vessel integrity and blood-brain barrier function in the mammalian vasculature. 25483078_A viral cyclin-CDK6 complex was found to be a regulator of Notch3 in lymphoma cells. 25499214_Dysregulated expression and processing of Jagged1 protein, sustained by Notch3, takes part in a multistep oncogenic process, playing a role in controlling cell growth, apoptosis, and migration 25555543_Study showed Notch3 expressed in proliferating hippocampal precursor cells and down-regulates their activation and proliferation; adult neurogenesis in CADASIL transgenic mice was altered prior to vascular abnormalities due to deficits in Notch3 signaling 25564622_Endogenous activation of Notch3 signaling selectively controls the pool of undifferentiated progenitors of upper airways available for differentiation. 25658374_Relative to air-exposed controls, ozone increased bronchoalveolar lavage fluid protein, a marker of lung permeability, in all genotypes, but significantly greater concentrations were found in Notch4-/- compared with wild-type and Notch3-/- mice. 25957400_NOTCH3, but not NOTCH2 protects vascular smooth muscle cell from apoptosis. 26093524_The mechanism of Oncostatin M on cardiac ischemia/reperfusion injury is partly mediated by the Notch3/Akt pathway. 26122050_miR-7a regulates the differentiation of Muller glia through the suppression of Notch3 expression. 26312599_Mutant Notch3 accumulates in pericytes and causes progressive pericyte loss and BBB leakage in the cerebral cortex in CADASIL mouse model. 26453897_In this study, authors use a smooth muscle-specific deletion of Notch2 together with a global Notch3 deletion to produce mice with combinations of mutant and wild-type Notch2/3 alleles in vascular smooth muscle cells 26487518_Notch3 is an important protective factor for cardiac fibrosis in a myocardial infarction model, and the protective effect of Notch3 is attributable to its action on TGF-beta1/Smad3 signaling. 26563570_Data indicate that Notch receptors Notch1 and Notch3 deficiency compromises pericyte function and contributes to vascular pathologies. 26648042_Elevated levels of TIMP3 and vitronectin, acting downstream of Notch3(ECD) deposition, play a role in CADASIL, producing divergent influences on early CBF deficits and later white matter lesions. 27791012_We find that across species, the atypical receptor NOTCH3 is differentially overexpressed; it is progressively up-regulated with disease development and promotes tumor cell survival via activation of PI3k-Akt 27821617_Knock-in mice with the R169C mutation (Notch3(R170C/R170C)) exhibited similar reductions in arterial lumen, and both TgNotch3(R169C) and Notch3(R170C/R170C) mice showed increased cerebral artery expression of Notch3 target genes. 28131704_Notch3 mutation impairs recovery of cardiac function post-myocardial ischemia. 28669409_this study shows that Notch signaling regulates basophils biological function, at least partially via the modulation of MAPK 28719575_endothelial expression of Notch3 limits tumour angiogenesis; tumour growth and angiogenesis are increased when Notch3 is silenced in the stroma 28747678_Lnc-LFAR1 binds directly to Smad2/3 and promotes transcription of TGFbeta, Smad2, Smad3, Notch2 and Notch3 which, in turn, results in TGFbeta and Notch pathway activation. 29101245_Notch3 plays an important role in the maintenance of quiescent neural stem cells in the subependymal zone. 29128180_overexpression demonstrates an antiproliferative effect on established metastatic medullary thyroid cancer liver tumors 29161703_Double immunostaining with differentiation markers revealed that NOTCH3 was expressed in some undifferentiated and differentiated spermatogonia in mouse testes. 29712618_High NOTCH3 expression is associated with melanoma metastasis and invasion. 29751972_Study revealed a strong de novo production of Notch3 by injured tubular epithelium and a subset of macrophages after ischemia/reperfusion (I/R). Mice lacking Notch3 showed preserved renal function and structure. Inversely, in a mouse strain conditionally expressing Notch3 intracellular domain in renal tubules, I/R-caused damage was exacerbated. 29991510_NOTCH3 contributes to rhinovirus-induced goblet cell hyperplasia in COPD airway epithelial cells. 30042232_The Notch3(tm1.1Ecan) mutation was introduced into mice, and it was found that this mutation causes osteopenia despite an increase in osteoblast proliferation and function and is associated with enhanced Tnfsf11 expression in osteoblasts and osteocytes. 30347392_Notch signaling is blunted in diabetic retinas with pericyte loss. 30763672_our results provide direct evidences that miR-206 regulates skeletal muscle cells proliferation and cell cycle arrest partly by targeting the Notch3 gene, which strengthens our understanding of regulatory mechanisms on miR-206 in skeletal muscle growth and development. 31188489_An antibody to Notch3 reverses the skeletal phenotype of lateral meningocele syndrome in male mice. 31400166_APOBEC3 regulates keratinocyte differentiation and expression of Notch3. 31647781_ER stress and Rho kinase activation underlie the vasculopathy of CADASIL. 31882173_The YB-1:Notch-3 axis modulates immune cell responses and organ damage in systemic lupus erythematosus. 32029735_Beta secretase 1-dependent amyloid precursor protein processing promotes excessive vascular sprouting through NOTCH3 signalling. 32495894_MiR-201-5p alleviates lipopolysaccharide-induced renal cell dysfunction by targeting NOTCH3. 32499639_results indicate that synovial fibroblasts exhibit a positional identity that is regulated by endothelium-derived Notch signalling, and that this stromal crosstalk pathway underlies inflammation and pathology in inflammatory arthritis 32525899_Notch3 signaling promotes tumor cell adhesion and progression in a murine epithelial ovarian cancer model. 32616814_Enhanced Notch3 signaling contributes to pulmonary emphysema in a Murine Model of Marfan syndrome. 32749453_AKT-dependent NOTCH3 activation drives tumor progression in a model of mesenchymal colorectal cancer. 32859670_Fibrosis and Immune Cell Infiltration Are Separate Events Regulated by Cell-Specific Receptor Notch3 Expression. 32908186_NOTCH3 signaling is essential for NF-kappaB activation in TLR-activated macrophages. 33493092_Notch3 Deficiency Attenuates Pulmonary Fibrosis and Impedes Lung-Function Decline. 33774049_Activation of Notch3 in osteoblasts/osteocytes causes compartment-specific changes in bone remodeling. 34374252_[Effects of Notch3 on gene expression and signal pathway of pancreatic stellate cell activation]. 34854382_KCTD10 regulates brown adipose tissue thermogenesis and metabolic function via Notch signaling. 35055068_Activation of Notch3 in Renal Tubular Cells Leads to Progressive Cystic Kidney Disease. 35211631_Impact of Notch3 Activation on Aortic Aneurysm Development in Marfan Syndrome. 35536858_Use of antisense oligonucleotides to target Notch3 in skeletal cells. 35760307_Induction of a NOTCH3 Lehman syndrome mutation in osteocytes causes osteopenia in male C57BL/6J mice. 36555708_Deletion of Notch3 Impairs Contractility of Renal Resistance Vessels Due to Deficient Ca[2+] Entry. |
ENSG00000074181 |
NOTCH3 |
538.501270 |
2.7185764075 |
1.442851 |
0.16650978 |
73.148406 |
0.00000000000000001202586735695175498774596163140177189021499778222865431609456265960034215822815895080566406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000003905714532645823687376685489700080913195332713971213989267994293186347931623458862304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
781.60147576865 |
90.4095571400211 |
287.503957445684 |
25.4672331839048 |
| ENSMUSG00000038173 |
320981 |
Enpp6 |
protein_coding |
Q8BGN3 |
FUNCTION: Choline-specific glycerophosphodiesterase that hydrolyzes glycerophosphocholine (GPC) and lysophosphatidylcholine (LPC) and contributes to supplying choline to the cells (PubMed:15788404, PubMed:26888014). Has a preference for LPC with short (12:0 and 14:0) or polyunsaturated (18:2 and 20:4) fatty acids (PubMed:15788404). In vitro, hydrolyzes only choline-containing lysophospholipids, such as sphingosylphosphorylcholine (SPC), platelet-activating factor (PAF) and lysoPAF, but not other lysophospholipids (PubMed:15788404). {ECO:0000269|PubMed:15788404, ECO:0000269|PubMed:26888014}. |
3D-structure;Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Hydrolase;Lipid degradation;Lipid metabolism;Lipoprotein;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Signal;Zinc |
|
This gene encodes a member of the nucleotide pyrophosphatase/phosphodiesterase family of enzymes that play an important role in regulating extracellular nucleotide metabolism. The encoded preproprotein undergoes proteolytic processing to generate a glycosylphosphatidylinositol (GPI)-anchored membrane protein that hydrolyzes choline-containing lysophospholipids such as glycerophosphocholine. Mice lacking the encoded protein develop fatty liver and myelin sheath abnormalities. [provided by RefSeq, Jul 2016]. |
mmu:320981; |
extracellular region [GO:0005576]; plasma membrane [GO:0005886]; glycerophosphocholine cholinephosphodiesterase activity [GO:0047390]; glycerophosphodiester phosphodiesterase activity [GO:0008889]; metal ion binding [GO:0046872]; phosphoric diester hydrolase activity [GO:0008081]; choline metabolic process [GO:0019695]; lipid catabolic process [GO:0016042]; lipid metabolic process [GO:0006629] |
15788404_NPP6 has a specific role through the hydrolysis of polyunsaturated LPC, glycerophosphorylcholine, or sphingosylphosphorylcholine in kidney, brain and heart 26888014_The crystal structure of ENPP6 in complex with phosphocholine revealed that the choline moiety of the phosphocholine is recognized by a choline-binding pocket formed by conserved aromatic and acidic residues. The present study provides the molecular basis for ENPP6-mediated choline metabolism at atomic, cellular and tissue levels. |
ENSG00000164303 |
ENPP6 |
263.776495 |
0.4750173845 |
-1.073948 |
0.18282806 |
34.227727 |
0.00000000490254369361522472011353962245867876834637399952043779194355010986328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000624899962687423496626891645557011489842125229188241064548492431640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
166.804198424701 |
21.1951793469341 |
351.153881671311 |
31.8982118903076 |
| ENSMUSG00000038178 |
215113 |
Slc43a2 |
protein_coding |
Q8CGA3 |
FUNCTION: Uniporter that mediates the transport of the stereospecific L-phenylalanine, L-methionine and L-branched-chain amino acids, between the extracellular space and the cytoplasm and may control the transepithelial (re)absorption of neutral amino acid in kidney and small intestine (PubMed:25480797, PubMed:15659399). Transport activity is mediated through facilitated diffusion and is sodium ions-, chloride ions- and pH-independent (PubMed:25480797). {ECO:0000269|PubMed:15659399, ECO:0000269|PubMed:25480797}. |
Cell membrane;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:215113; |
basolateral plasma membrane [GO:0016323]; plasma membrane [GO:0005886]; L-amino acid transmembrane transporter activity [GO:0015179]; L-isoleucine transmembrane transporter activity [GO:0015188]; L-leucine transmembrane transporter activity [GO:0015190]; L-methionine transmembrane transporter activity [GO:0015191]; L-phenylalanine transmembrane transporter activity [GO:0015192]; neutral L-amino acid transmembrane transporter activity [GO:0015175]; isoleucine transport [GO:0015818]; L-amino acid transport [GO:0015807]; leucine transport [GO:0015820]; methionine transport [GO:0015821]; negative regulation of amino acid transport [GO:0051956]; negative regulation of leucine import [GO:0060358]; neutral amino acid transport [GO:0015804]; phenylalanine transport [GO:0015823] |
15659399_In situ hybridization experiments show that LAT4 mRNA is restricted to the epithelial cells of the distal tubule and the collecting duct in the kidney. In the intestine, LAT4 is mainly present in the cells of the crypt. 25480797_Mice lacking Lat4 display a slight intrauterine growth restriction, postnatal malnutrition and early death. 30379325_LAT4 is subject to a circadian regulation by phosphorylation in the mouse small intestine, and this novel regulatory mechanism is suggested to play an important role in the diurnal regulation of amino acid absorption. 32470053_Phosphorylation of mouse intestinal basolateral amino acid uniporter LAT4 is controlled by food-entrained diurnal rhythm and dietary proteins. 32879489_Cancer SLC43A2 alters T cell methionine metabolism and histone methylation. |
ENSG00000167703 |
SLC43A2 |
238.206233 |
0.4617971152 |
-1.114669 |
0.30564165 |
13.044953 |
0.00030410279205141598716372053345935455581638962030410766601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016140486109750649002664291131736717943567782640457153320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
150.558445691393 |
32.1356411986387 |
326.027254697657 |
51.6477052611689 |
| ENSMUSG00000038236 |
15404 |
Hoxa7 |
protein_coding |
P02830 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15404; |
nuclear membrane [GO:0031965]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; angiogenesis [GO:0001525]; anterior/posterior pattern specification [GO:0009952]; embryonic skeletal system morphogenesis [GO:0048704]; negative regulation of cell-matrix adhesion [GO:0001953]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of keratinocyte differentiation [GO:0045617]; negative regulation of leukocyte migration [GO:0002686]; negative regulation of monocyte differentiation [GO:0045656]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; stem cell differentiation [GO:0048863] |
12068955_The retinoic acid response element in the regulatory region affects the rate, not the formation of anterior boundary expression 12082612_Nup98-HoxA9 immortalizes myeloid progenitors, enforces expression of Hoxa9, Hoxa7 and Meis1, and alters cytokine-specific responses in a manner similar to that induced by retroviral co-expression of Hoxa9 and Meis1. 12549822_novel dna binding protein of Hoxa7 promoter; sequence analysis revealed that protein C171 contained one hydrophobic transmembrane domain in the N-terminal region and several putative phosphorylation and glycosylation sites 15070702_impact of Hoxa7 or Hoxa9 nullizygosity on hematopoietic progenitor compartments and their susceptibility to MLL-induced leukemias 15470633_during development, Hox expression boundaries become set along instructional cdx gradients, formed by cdx decay in cells moving away from the primitive streak region 27554164_the cohesin complex regulates PRC2 targeting to silence Hoxa7 and Hoxa9 and negatively regulate self-renewal. Our studies identify a novel epigenetic mechanism underlying leukemogenesis in AML patients with cohesin mutations. |
ENSG00000122592 |
HOXA7 |
53.956300 |
0.4866782405 |
-1.038960 |
0.37950602 |
7.305057 |
0.00687608124515631802253778914746362715959548950195312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0243743238366816473983167412598049850203096866607666015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.8968633873335 |
9.0198806983548 |
69.6494327588363 |
13.3578115030369 |
| ENSMUSG00000038239 |
15464 |
Hrc |
protein_coding |
G5E8J6 |
Human_homologues FUNCTION: May play a role in the regulation of calcium sequestration or release in the SR of skeletal and cardiac muscle. |
Proteomics identification;Reference proteome;Signal |
|
|
mmu:15464; |
sarcoplasmic reticulum membrane [GO:0033017]; Z disc [GO:0030018]; ATPase binding [GO:0051117]; calcium ion binding [GO:0005509]; transmembrane transporter binding [GO:0044325]; calcium ion homeostasis [GO:0055074]; negative regulation of calcium ion import into sarcoplasmic reticulum [GO:1902081]; negative regulation of cytosolic calcium ion concentration [GO:0051481]; positive regulation of heart contraction [GO:0045823]; positive regulation of heart rate [GO:0010460]; positive regulation of relaxation of cardiac muscle [GO:1901899]; regulation of calcium ion transmembrane transport [GO:1903169]; regulation of cell communication by electrical coupling involved in cardiac conduction [GO:1901844]; regulation of cytosolic calcium ion concentration [GO:0051480]; regulation of heart contraction [GO:0008016]; regulation of heart rate [GO:0002027]; regulation of peptidyl-serine phosphorylation [GO:0033135]; regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum [GO:0010880]; regulation of ryanodine-sensitive calcium-release channel activity [GO:0060314] |
15082771_Results identify the HRC enhancer as the first MEF2-dependent, CArG-motif-independent transcriptional target in smooth muscle and represent the first analysis of the transcriptional regulation of a sarcoplasmic reticulum gene in vivo. 15191886_alterations in expression levels of HRC are associated with impaired cardiac SR Ca homeostasis and contractile function. 16600288_HRC may be an integral regulatory protein involved in cardiac muscle SR Ca uptake and Ca homeostasis 17030629_The exaggerated response of HRC knockout mice to the induction of cardiac hypertrophy is consistent with a regulatory role for HRC in calcium handling in vivo. 17526652_Histidine-rich Ca-binding protein may play a key role in the regulation of SR Ca cycling through its direct interactions with SERCA2 and triadin, mediating a fine cross talk between SR Ca uptake and release in the heart. 23553082_Ablation of HRC is associated with poorly regulated SR Ca(2+)-cycling, and severe pathology under pressure-overload stress, suggesting an essential role of HRC in maintaining the integrity of cardiac function. 26410369_While CASQ2 stabilizes RyR2 rendering it refractory in the diastolic phase, HRC enhances RyR2 activity facilitating RyR2 recovery from refractoriness. 26674963_The minimal KEKE motif of TRN involved in the interactions with CSQ2, HRC and RyR2 within the Ca2+ release units of cardiac sarcoplasmic reticulum has been defined. |
ENSG00000130528 |
HRC |
78.732073 |
0.0966999166 |
-3.370342 |
0.35913061 |
90.407528 |
0.00000000000000000000193827235992872370904377749613903021392817345671960344873465190151939907536871032789349555969238281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000829319229956020127836050702836352134319891210284147640367097276126173710508737713098526000976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.7594553925788 |
3.46063008511774 |
142.290250822976 |
21.92185922152 |
| ENSMUSG00000038252 |
68298 |
Ncapd2 |
protein_coding |
A0A0R4J0H7 |
FUNCTION: Regulatory subunit of the condensin complex, a complex required for conversion of interphase chromatin into mitotic-like condense chromosomes. The condensin complex probably introduces positive supercoils into relaxed DNA in the presence of type I topoisomerases and converts nicked DNA into positive knotted forms in the presence of type II topoisomerases. {ECO:0000256|PIRNR:PIRNR017127}. |
Cell cycle;Cell division;Chromosome;DNA condensation;Mitosis;Nucleus;Proteomics identification;Reference proteome |
|
|
mmu:68298; |
condensed chromosome [GO:0000793]; condensin complex [GO:0000796]; cytosol [GO:0005829]; nuclear chromosome [GO:0000228]; nucleoplasm [GO:0005654]; histone binding [GO:0042393]; cell division [GO:0051301]; mitotic chromosome condensation [GO:0007076]; positive regulation of chromosome condensation [GO:1905821] |
16144839_The characterization of two novel E2F target genes, chromosome condensation-related SMC-associated protein 1 (Cnap1) and maternal embryonic leucine zipper kinase (Melk) is reported. 21858164_Data show that that DNA repear process is dictated by PARP1 through its interaction with the chromosome-targeting domain of the hCAP-D2 subunit of condensin I. 23531880_Knock-down of a condensin subunit protein or YY1 reduced rearrangement of Igkappa Vkappa genes suggesting a direct role for YY1-condensin complexes in Igkappa locus structure and rearrangement. 25474630_Condensins I and II have overlapping and non-overlapping functions in Neural stem cells. 34229059_NCAPD2 inhibits autophagy by regulating Ca(2+)/CAMKK2/AMPK/mTORC1 pathway and PARP-1/SIRT1 axis to promote colorectal cancer. |
ENSG00000010292 |
NCAPD2 |
3490.164015 |
2.4673231035 |
1.302947 |
0.10283532 |
156.672228 |
0.00000000000000000000000000000000000603633020595414860215016122155932528204375153109543166827566640977080380527511781536480721518586611828993682138388976454734802246093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000005782728408622238029287647666086213118007362730430943736117669036773850111911359410881523432368389592284074751660227775573730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4946.46023368116 |
352.882377551739 |
2004.78819600642 |
107.991661554139 |
| ENSMUSG00000038264 |
20361 |
Sema7a |
protein_coding |
Q9QUR8 |
FUNCTION: Plays an important role in integrin-mediated signaling and functions both in regulating cell migration and immune responses. Promotes formation of focal adhesion complexes, activation of the protein kinase PTK2/FAK1 and subsequent phosphorylation of MAPK1 and MAPK3. Promotes production of pro-inflammatory cytokines by monocytes and macrophages. Plays an important role in modulating inflammation and T-cell-mediated immune responses. Promotes axon growth in the embryonic olfactory bulb. Promotes attachment, spreading and dendrite outgrowth in melanocytes. {ECO:0000269|PubMed:12879062, ECO:0000269|PubMed:16713976, ECO:0000269|PubMed:17377534}. |
Cell membrane;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;GPI-anchor;Immunoglobulin domain;Inflammatory response;Lipoprotein;Membrane;Methylation;Neurogenesis;Reference proteome;Signal |
|
|
mmu:20361; |
external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; chemorepellent activity [GO:0045499]; integrin binding [GO:0005178]; semaphorin receptor binding [GO:0030215]; axon extension [GO:0048675]; axon guidance [GO:0007411]; inflammatory response [GO:0006954]; integrin-mediated signaling pathway [GO:0007229]; negative chemotaxis [GO:0050919]; negative regulation of axon extension involved in axon guidance [GO:0048843]; neural crest cell migration [GO:0001755]; neuron projection development [GO:0031175]; olfactory lobe development [GO:0021988]; positive regulation of axon extension [GO:0045773]; positive regulation of cell migration [GO:0030335]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of macrophage cytokine production [GO:0060907]; positive regulation of protein phosphorylation [GO:0001934]; protein phosphorylation [GO:0006468]; regulation of inflammatory response [GO:0050727]; semaphorin-plexin signaling pathway [GO:0071526] |
12879062_Sema7A enhancement of axon outgrowth requires integrin receptors and activation of MAPK signalling pathways 15859945_we have investigated its regulation and function in bone cells.Its expression is also regulated during primary osteoclast differentiation in vitro 17377534_findings not only identify a function of Sema7A as an effector molecule in T-cell-mediated inflammation, but also reveal a mechanism of integrin-mediated immune regulation 17485510_Semaphorin 7A plays a critical role in TGF-beta1-induced pulmonary fibrosis. 19741144_Semaphorin 7A is a novel substrate for caspase-9 that can be used to monitor caspase-9 activity in mice. 21484765_interventions that reduce semaphorin 7a expression or prevent the semaphorin 7a-beta1 integrin interaction may ameliorate TGFbeta1-driven or fibrocyte-associated autoimmune fibroses. 21903667_GnRH-1 cells differentially express the Sema7 receptors beta1-integrin and Plexin C1 as a function of their migratory stage. 21999225_Sema7A was expressed in vascularized corneas and showed pro-angiogenic properties in our corneal model. 22198947_Sema7A plays crucial roles in suppressing intestinal inflammation through alphavbeta1 integrin, but also provides a novel mode of IL-10 induction via interactions between IECs and Mvarphis 22700709_Sema7a is expressed constitutively in the cornea, and potently stimulates nerve regeneration and inflammatory cell influx. 22875994_Oncogenic Ras may play a role in epithelial mesenchymal transition via the ERF and regulation of Semaphorin-7a. 22891341_We report here the expression and induction of semaphorin 7A (SEMA7A) on endothelium through hypoxia-inducible factor 1alpha during hypoxia. 22896629_Sema7A mediates signaling through smad6 during West Nile virus infection in an analysis of smad6 expression in cortical neurons from sema7A-deficient mice. 23850082_SEMA7A was expressed in the liver and was increased in the course of liver fibrosis, both in mice and in humans. 24831527_these results suggest that Sema3A and Sema7A have opposite effects and are involved in different stages of synapse elimination. 25201975_These data identify Sema7A as a regulator of thalamocortical and local circuit development in layer 4 and provide a molecular handle that can be used to explore the coordinated generation of excitatory and inhibitory cortical circuits. 25511377_Sem7a and its receptors regulate Chi3l1 in pulmonary melanoma metastasis 26144372_SEMA7A plays a critical role in IgE-mediated neutrophilic airway inflammation 26597008_This study demonstrates that Sema7A controls the assembly of actin-based protrusions that drive Dendritic cell migration in response to CCL21. 26686462_Data suggest that sema7A is involved in the pathogenesis of experimental autoimmune encephalomyelitis, in the modulation of the immune response and in the neurodegeneration that take place in the CNS. 26752048_SEMA7A has a role in the development of lung injury 27065336_Our results suggest that semaphorin 7a may be novel target for blocking breast tumor progression. 27714632_sema7A is involved in peripheral immunity and central nervous system inflammation in multiple sclerosis pathogenesis. 28109308_These findings indicate that Sema7A as a potent activator of T cells and monocytes in the immune response contributes to the inflammation and progression of rheumatoid arthritis 28281529_During differentiation of adult-born dentate gyrus granule cells, Sema7A promotes dendrite growth, complexity and spine development through beta1-subunit-containing integrin receptors. 29048670_Genetic ablation of host-derived SEMA7A further enhanced the antitumor effects of SEMA7A shRNA gene silencing in 4T1 cells. Our preclinical findings demonstrate a critical role for SEMA7A in mediating mammary tumor progression. 29108785_Sema7A, as an inflammation regulator in CVB3-induced Viral myocarditis, might interact with alpha1beta1-integrin in macrophages to enhance the inflammatory response and aggravate disease severity. 29269512_Sema7A has a significant role in atherosclerosis by mediating endothelial dysfunction in a beta1 integrin-dependent manner. 29743476_Sema7A-PlxnC1 interaction is essential to form the post-synaptic assembly. 32161256_Study reports that soluble SEMA7A is elevated in plasma of patients with acute Myocardial infarction (MI), and that Semaphorin 7A holds significant impact on the extent of MI reperfusion injury. In mouse model Sema7a promotes myocardial thrombo-inflammation and tissue damage by reinforcing platelet thrombotic activity and platelet-neutrophil complexes formation through a platelet GPIb-dependent mechanism. 33128105_Semaphorin 7A restricts serotonergic innervation and ensures recovery after spinal cord injury. 33637648_Sema7A is crucial for resolution of severe inflammation. 33780330_The olfactory critical period is determined by activity-dependent Sema7A/PlxnC1 signaling within glomeruli. 34561423_Anoikis resistance in mammary epithelial cells is mediated by semaphorin 7a. 34585848_A homozygous R148W mutation in Semaphorin 7A causes progressive familial intrahepatic cholestasis. 35218761_Semaphorin 7A Accelerates the Inflammatory Osteolysis of Periapical Lesions. 35938531_SEMA7AR148W mutation promotes lipid accumulation and NAFLD progression via increased localization on the hepatocyte surface. 36705403_Knockdown of SEMA7A alleviates MPP[+] -induced apoptosis and inflammation in BV2 microglia via PPAR-gamma activation and MAPK inactivation. 36717921_Semaphorin 7A interacts with nuclear factor NF-kappa-B p105 via integrin beta1 and mediates inflammation. |
ENSG00000138623 |
SEMA7A |
186.505578 |
0.0533816271 |
-4.227513 |
0.29669101 |
205.785358 |
0.00000000000000000000000000000000000000000000011413654134915330124362668753368847189857722686050408289738735147405912509352515131504258727857452958604180069137717630847750349687430571066215634346008300781250000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000172131465131713157956758144310340089780156543032021908589043386983107697737686140121176647005107982441848127238550811290984654533531283959746360778808593750000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.0462838948909 |
3.94454581902678 |
356.794742749639 |
43.9618191166736 |
| ENSMUSG00000038267 |
73102 |
Slc22a23 |
protein_coding |
Q3UHH2 |
|
Alternative splicing;Glycoprotein;Ion transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:73102; |
membrane [GO:0016020]; transmembrane transporter activity [GO:0022857]; ion transport [GO:0006811] |
Human_homologues 19068216_Observational study of gene-disease association. (HuGE Navigator) 19174780_Observational study of gene-disease association. (HuGE Navigator) 19422935_Observational study of gene-disease association. (HuGE Navigator) 19760754_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20886065_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 20921969_Observational study and genome-wide association study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 21072187_Observational study of gene-disease association. (HuGE Navigator) 24740203_Genetic variations in the SLC22A23 locus contribute to the susceptibility to inflammatory bowel disease in a small fraction of white patients. |
ENSG00000137266 |
SLC22A23 |
129.341396 |
0.1779238090 |
-2.490669 |
0.37356466 |
41.570184 |
0.00000000011371439545563882885678445093424380162028253238304387195967137813568115234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000018023909173572224434118259974195105921346282684680772945284843444824218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.3151994621943 |
9.77055586672923 |
209.725722863457 |
38.7142565135393 |
| ENSMUSG00000038296 |
233733 |
Galnt18 |
protein_coding |
Q8K1B9 |
FUNCTION: Catalyzes the initial reaction in O-linked oligosaccharide biosynthesis, the transfer of an N-acetyl-D-galactosamine (GalNAc) residue from UDP-GalNAc to a serine or threonine residue on the protein receptor. {ECO:0000250|UniProtKB:Q6P9A2}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lectin;Manganese;Membrane;Metal-binding;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000250|UniProtKB:Q6P9A2}. |
|
mmu:233733; |
Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; carbohydrate binding [GO:0030246]; metal ion binding [GO:0046872]; polypeptide N-acetylgalactosaminyltransferase activity [GO:0004653]; protein O-linked glycosylation [GO:0006493] |
Human_homologues 19240061_Observational study of gene-disease association. (HuGE Navigator) 20080650_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 33909026_Polypeptide N-acetylgalactosaminyltransferase 18 retains in endoplasmic reticulum depending on its luminal regions interacting with ER resident UGGT1, PLOD3 and LPCAT1. |
ENSG00000110328 |
GALNT18 |
19.042204 |
0.2796065771 |
-1.838530 |
0.60051465 |
9.272519 |
0.00232617575530877449679900692558476293925195932388305664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0096961635538336619216659428843740897718816995620727539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.28527129940419 |
3.54915636604112 |
29.6318898743871 |
8.62104681421176 |
| ENSMUSG00000038354 |
213121 |
Ankrd35 |
protein_coding |
D6RGN5 |
|
Reference proteome |
|
|
|
|
Human_homologues 33203765_Identifying chromosomal subpopulations based on their recombination histories advances the study of the genetic basis of phenotypic traits. |
ENSG00000198483 |
ANKRD35 |
62.313234 |
0.4760324911 |
-1.070868 |
0.33219110 |
10.385050 |
0.00127039751056804133683142410404798283707350492477416992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0057142207501718750295438553621352184563875198364257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
41.3934367302529 |
9.61558652037052 |
86.9550660943279 |
14.462754436955 |
| ENSMUSG00000038370 |
66425 |
Pcp4l1 |
protein_coding |
Q6W8Q3 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:66425; |
|
15053978_Pcp4l1 encodes a polypeptide with significant sequence similarity to the Purkinje cell protein 4 gene (Pcp40;it is expressed during mouse development. 22458599_We speculate Pcp4l1 may be a latent calmodulin inhibitor regulated by post-translational modification and/or co-factor interactions 24692514_Pcp4l1 is an activity-dependent gene in mouse olfactory sensory neurons. [Pcp4l1] |
ENSG00000248485 |
PCP4L1 |
138.847494 |
0.1851993374 |
-2.432849 |
0.26812282 |
80.272634 |
0.00000000000000000032615495197432918433919241846810381975965843658662448797946886713816638803109526634216308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000122666474777110677325368354962607022517958747830427168601374887657584622502326965332031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.4450520971613 |
7.93384131360927 |
229.185766476039 |
29.2222955877405 |
| ENSMUSG00000038375 |
68728 |
Trp53inp2 |
protein_coding |
Q8CFU8 |
FUNCTION: Dual regulator of transcription and autophagy. Positively regulates autophagy and is required for autophagosome formation and processing. May act as a scaffold protein that recruits MAP1LC3A, GABARAP and GABARAPL2 and brings them to the autophagosome membrane by interacting with VMP1 where, in cooperation with the BECN1-PI3-kinase class III complex, they trigger autophagosome development. Acts as a transcriptional activator of THRA. {ECO:0000269|PubMed:18030323}. |
Activator;Alternative splicing;Autophagy;Cytoplasm;Cytoplasmic vesicle;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:68728; |
autophagosome [GO:0005776]; cytoplasmic vesicle [GO:0031410]; cytosol [GO:0005829]; nucleus [GO:0005634]; PML body [GO:0016605]; ubiquitin binding [GO:0043130]; autophagosome assembly [GO:0000045]; macroautophagy [GO:0016236]; negative regulation of protein localization [GO:1903828]; osteoblast differentiation [GO:0001649]; positive regulation of DNA-templated transcription [GO:0045893]; protein localization [GO:0008104]; tissue homeostasis [GO:0001894]; ubiquitin-dependent protein catabolic process [GO:0006511] |
24713655_TP53INP2 repression is part of an adaptive mechanism aimed at preserving muscle mass under conditions in which insulin action is deficient. 29593329_TP53INP2 modulates adipogenesis through autophagy-dependent sequestration of GSK3beta into late endosomes and beta catenin activation, regulating adiposity. 30853298_Tp53inp2 is an atypical mRNA that regulates axon growth by enhancing NGF-TrkA signaling in a translation-independent manner. |
ENSG00000078804 |
TP53INP2 |
776.506938 |
0.4450217103 |
-1.168052 |
0.11824257 |
96.002003 |
0.00000000000000000000011476730354308905862959955394296931329482464104112612604955876403900472126906606717966496944427490234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000053134819682928041721682326922869203474358876987002947469684172032700075760658364742994308471679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
471.284096327569 |
38.6694686050135 |
1059.01371877904 |
63.4235634508009 |
| ENSMUSG00000038388 |
56524 |
Pals2 |
protein_coding |
Q9JLB0 |
|
Alternative splicing;Membrane;Phosphoprotein;Reference proteome;Repeat;SH3 domain |
|
|
mmu:56524; |
cell-cell junction [GO:0005911]; membrane [GO:0016020]; plasma membrane [GO:0005886]; guanylate kinase activity [GO:0004385] |
22025680_In the mouse sciatic nerve, protein 4.1G colocalized at Schmidt-Lanterman incisures (SLI) and the paranodes with a member of the membrane-associated guanylate kinase (MAGUK) family, membrane protein palmitoylated 6. 23306908_The findings indicate that the Src-MPP6-4.1G protein complex in Schmidt-Lanterman incisures(SLIs) has a role in signal transduction in the peripheral nervous system(PNS). 26496923_This paper herein showed the immunolocalization and interaction proteins of MPP6 in the mouse small intestine, and also that 4.1B in epithelial cells was not essential for the sorting of MPP6. 30357511_The results indicate that the MPP6-Lin7 complex regulates myelin formation. |
ENSG00000105926 |
PALS2 |
353.017695 |
2.4103590146 |
1.269248 |
0.18978969 |
44.138055 |
0.00000000003060135291828661621622713194889697611855705616790146450512111186981201171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000005133478057834159688784947027861364082790274210310599301010370254516601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
494.598672640797 |
65.2533698029953 |
205.197097051084 |
20.6979200831686 |
| ENSMUSG00000038400 |
65112 |
Pmepa1 |
protein_coding |
A2APF5 |
Human_homologues FUNCTION: Functions as a negative regulator of TGF-beta signaling and thereby probably plays a role in cell proliferation, differentiation, apoptosis, motility, extracellular matrix production and immunosuppression. In the canonical TGF-beta pathway, ZFYVE9/SARA recruits the intracellular signal transducer and transcriptional modulators SMAD2 and SMAD3 to the TGF-beta receptor. Phosphorylated by the receptor, SMAD2 and SMAD3 then form a heteromeric complex with SMAD4 that translocates to the nucleus to regulate transcription. Through interaction with SMAD2 and SMAD3, LDLRAD4 may compete with ZFYVE9 and SMAD4 and prevent propagation of the intracellular signal (PubMed:20129061, PubMed:24627487). Also involved in down-regulation of the androgen receptor (AR), enhancing ubiquitination and proteasome-mediated degradation of AR, probably by recruiting NEDD4 (PubMed:18703514). {ECO:0000269|PubMed:18703514, ECO:0000269|PubMed:20129061, ECO:0000269|PubMed:24627487}. |
Endosome;Membrane;Reference proteome;Signal transduction inhibitor;Transmembrane;Transmembrane helix |
|
|
|
early endosome membrane [GO:0031901]; R-SMAD binding [GO:0070412]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512] |
20889500_Wnt/beta-catenin/TCF7L2 pathway is preferentially able to alter the transcriptional regulation of the TGF-beta-target gene, TMEPAI. 26215835_binding of SMAD2/3, the intracellular effectors of activin signaling, was significantly enriched at the Pmepa1 gene, which encodes a negative feedback regulator of TGF-beta signaling in cancer cells, and at the Kdm6b gene, which encodes an epigenetic regulator promoting transcriptional plasticity. 28802000_Pmepa1 is induced by RANK-p38 MAPK pathway signaling. 30557043_Pmepa1 expression was barely detected in osteoclasts formed on plastic surfaces in vitro, but was markedly increased in activated osteoclasts formed on calcified matrix. Inhibitors of bone resorption suppressed its expression. Pmepa1 protein localized to lysosomes in osteoclasts. 33311522_Hdac3 deletion in myeloid progenitor cells enhances bone healing in females and limits osteoclast fusion via Pmepa1. 33484199_PMEPA1 and NEDD4 control the proton production of osteoclasts by regulating vesicular trafficking. |
ENSG00000124225 |
PMEPA1 |
509.484766 |
0.4501183418 |
-1.151624 |
0.12098424 |
89.863766 |
0.00000000000000000000255137126746125196866543487137085358314696985455834134828541695516612719529803143814206123352050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001085544333686307097187514966022263394481781363620201321523728310580736433621495962142944335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
318.948925597197 |
26.7783813479417 |
708.589044141629 |
42.8340950438599 |
| ENSMUSG00000038402 |
14238 |
Foxf2 |
protein_coding |
O54743 |
FUNCTION: Probable transcription activator for a number of lung-specific genes (PubMed:9676429). Mediates up-regulation of the E3 ligase IRF2BPL and drives ubiquitination and degradation of CTNNB1 (By similarity). {ECO:0000250|UniProtKB:Q12947, ECO:0000269|PubMed:9676429}. |
Activator;DNA-binding;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:14238; |
nucleus [GO:0005634]; transcription regulator complex [GO:0005667]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; general transcription initiation factor binding [GO:0140296]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; TFIIB-class transcription factor binding [GO:0001093]; animal organ morphogenesis [GO:0009887]; embryonic camera-type eye morphogenesis [GO:0048596]; embryonic digestive tract development [GO:0048566]; establishment of planar polarity of embryonic epithelium [GO:0042249]; extracellular matrix organization [GO:0030198]; genitalia development [GO:0048806]; negative regulation of DNA-templated transcription [GO:0045892]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of proteasomal ubiquitin-dependent protein catabolic process [GO:0032434]; regulation of protein polyubiquitination [GO:1902914]; regulation of transcription by RNA polymerase II [GO:0006357]; roof of mouth development [GO:0060021] |
12812790_Foxf2 gene plays key roles in palatogenesis by reshaping the growing tongue. 16439479_Foxf proteins are mesenchymal factors that control epithelial proliferation and survival, and link hedgehog to Bmp and Wnt signaling. 20009030_findings argue that Foxf2 is a previously unrecognized regulator of cellular and systemic whole body glucose tolerance, at least in part, due to lower levels of IRS1. 22022403_These data provide evidence that the Foxf2 gene, separated from Foxc1 by less than 70 kb of genomic sequence (250 kb in human DNA), may explain human abnormalities in some cases of ASD where FOXC1 has been excluded genetically. 23376422_Foxf2 is a fibroblast factor that inhibits paracrine Wnt signaling and restricts the crypt stem cell niche in intestines of mice. Loss of Foxf2 promotes adenoma formation and growth. 25356765_Our findings implicate Foxf genes(Foxf1a and Foxf2 ) in atrioventricular septation, describe the molecular underpinnings of the genetic interaction between Hedgehog signaling and Tbx5 25631042_results indicate that Foxf2 signaling in smooth muscle cells is essential for intestinal development and serum response factor signaling 26120030_Foxf2 inactivation in adults resulted in blood-brain barrier breakdown, endothelial thickening, and increased trans-endothelial vesicular transport 26745863_novel Shh-Foxf-Fgf18-Shh circuit in the palate development molecular network, in which Foxf1 and Foxf2 regulate palatal shelf growth downstream of Shh signaling, at least in part, by repressing Fgf18 expression 27180663_Foxf2(-/-) maxillary explants cultured in vitro, in the absence of tongue and mandible, failed to close the secondary palate. Proliferation and collagen content were decreased in Foxf2(-/-) palatal shelf mesenchyme. Phosphorylation of Smad2/3 was reduced in mutant palatal shelf, diagnostic of attenuated canonical Tgfbeta signaling, whereas phosphorylation of p38 was increased. 28506991_direct targeting of Foxf2 by Shh signaling drives cranial neural crest cell mesenchyme proliferation during upper lip morphogenesis, and disruption of this sequence results in cleft lip. 30285803_Expression of the transcription factor forkhead box protein F2 (Foxf2) is upregulated during the epithelial to mesenchymal transition (EMT) process. 30565344_Knockout of Foxf1 and Foxf2 together, but not alone, from mouse smooth muscle resulted in delayed liquid gastric emptying 35668101_Foxf2 represses bone formation via Wnt2b/beta-catenin signaling. 36227576_The transcription factors Foxf1 and Foxf2 integrate the SHH, HGF and TGFbeta signaling pathways to drive tongue organogenesis. 36369237_Stromal FOXF2 suppresses prostate cancer progression and metastasis by enhancing antitumor immunity. |
ENSG00000137273 |
FOXF2 |
152.015830 |
0.1984616972 |
-2.333067 |
0.25889788 |
78.748736 |
0.00000000000000000070533861838433562833447090163116424160321365929466621257082348250833092606626451015472412109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000255195197986465572274975678231935284733432110586531099682972012487880419939756393432617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
49.9363300555685 |
9.00946638247573 |
251.616965735037 |
31.2401423016683 |
| ENSMUSG00000038408 |
71307 |
1700018A04Rik |
lncRNA |
|
|
|
|
|
|
|
33816629_A Novel Long Noncoding RNA, Lnc-OAD, Is Required for Bone Morphogenetic Protein 2- (BMP-2-) Induced Osteoblast Differentiation. |
|
|
99.090007 |
0.1864295387 |
-2.423298 |
0.29972939 |
64.265561 |
0.00000000000000108729778475428436458503506597276464420925932551770243250643943611066788434982299804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000294692524152620305301078005085744477518092457324350164071802282705903053283691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
30.7030911053401 |
6.42475586561213 |
164.690055665277 |
23.350943554789 |
| ENSMUSG00000038415 |
15220 |
Foxq1 |
protein_coding |
O70220 |
FUNCTION: Plays a role in hair follicle differentiation. {ECO:0000269|PubMed:16835220}. |
DNA-binding;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15220; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; anatomical structure morphogenesis [GO:0009653]; cell differentiation [GO:0030154]; hair follicle morphogenesis [GO:0031069]; negative regulation of neuron apoptotic process [GO:0043524]; negative regulation of transcription by RNA polymerase II [GO:0000122]; regulation of transcription by RNA polymerase II [GO:0006357] |
16835220_satin hair mutant gene Foxq1 is among multiple and functionally diverse regulatory targets for Hoxc13 during hair follicle differentiation 18544931_Ultrastructural analysis suggests that the gastric acid secretion defect in Foxq1-deficient mice might be due to impairment in the fusion of cytoplasmic tubulovesicles to the apical membrane of secretory canaliculi. 18558092_Transcription factor foxq1 controls MUC5AC gene expression and granule content in mouse stomach surface mucous cells. 20717954_This study has elucidated the functional impact of FoxQ1 on epithelial differentiation. 23489430_R164C mutation in FOXQ1 H3 domain affects formation of the hair medulla in mice. 27421728_FOXQ1 is a novel factor involved in regulating hepatic gluconeogenesis, and the decreased FOXQ1 expression in liver may contribute to the development of type 2 diabetes. 29463842_data reveal that FOXQ1 acts as a critical mediator of BRAF(V600E)-dependent regulation of MITF levels, thus providing a novel link between two major signal transduction pathways controlling MITF and differentiation in melanocytic cells. 29945296_the Fox transcription factor binding motif was frequently observed within EWS-FLI1 peaks and Foxq1 was identified as the cooperative partner that interacts with the EWS portion of EWS-FLI1. 32943107_FOXQ1 promotes the osteogenic differentiation of bone mesenchymal stem cells via Wnt/beta-catenin signaling by binding with ANXA2. 34091745_Identification of testicular Foxq1 as a critical modulator of lactate metabolism in mouse Sertoli cells. |
ENSG00000164379 |
FOXQ1 |
57.107104 |
0.2860651243 |
-1.805584 |
0.42086565 |
17.192597 |
0.00003377500615657379374762864232550896304019261151552200317382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002253442373092124446833789397359737449733074754476547241210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.6023530382743 |
7.87102836100035 |
92.9940449792138 |
19.369392853719 |
| ENSMUSG00000038486 |
64051 |
Sv2a |
protein_coding |
Q9JIS5 |
FUNCTION: Plays a role in the control of regulated secretion in neural and endocrine cells, enhancing selectively low-frequency neurotransmission. Positively regulates vesicle fusion by maintaining the readily releasable pool of secretory vesicles.; FUNCTION: (Microbial infection) Receptor for C.botulinum neurotoxin type A (BoNT/A, botA); the toxin probably binds via extracellular loop 4 (PubMed:16543415). {ECO:0000269|PubMed:16543415}.; FUNCTION: (Microbial infection) Possible receptor for C.botulinum neurotoxin type D (BoNT/D, botD); BoNT/D does not bind to extracellular loop 4 as do BoNT/A and BoNT/E (PubMed:21483489). {ECO:0000269|PubMed:21483489}.; FUNCTION: (Microbial infection) Receptor for C.botulinum neurotoxin type E (BoNT/E); the toxin probably binds via extracellular loop 4 (PubMed:18815274). It probably requires glycosylation of Asn-573 (PubMed:18815274). {ECO:0000269|PubMed:18815274, ECO:0000305|PubMed:18815274}. |
Cell projection;Cytoplasmic vesicle;Glycoprotein;Membrane;Neurotransmitter transport;Phosphoprotein;Receptor;Reference proteome;Synapse;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:64051; |
cell-cell junction [GO:0005911]; dendrite [GO:0030425]; endoplasmic reticulum [GO:0005783]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; neuromuscular junction [GO:0031594]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; presynaptic active zone [GO:0048786]; synapse [GO:0045202]; synaptic vesicle [GO:0008021]; synaptic vesicle membrane [GO:0030672]; protein kinase binding [GO:0019901]; transmembrane transporter activity [GO:0022857]; cellular calcium ion homeostasis [GO:0006874]; chemical synaptic transmission [GO:0007268]; regulation of gamma-aminobutyric acid secretion [GO:0014052]; synaptic vesicle priming [GO:0016082] |
16436618_SV2 action modulates synaptic networks by ensuring that low-frequency neurotransmission is faithfully conveyed. 16543415_findings show that botulinum neurotoxin (BoNT/A) enters neurons by binding to the synaptic vesicle protein SV2 (isoforms A, B, and C);SV2 acts as the protein receptor for BoNT/A 18692481_This study provides the first direct structural data on SV2A, and supports a transporter function suggested by sequence homology to MFS class of transporter proteins. 18815274_Data demonstrate that glycosylated SV2A and SV2B act in conjunction with gangliosides to mediate the entry of BoNT/E into neurons. 19041904_SV2A (+/-) mice display significant increased avoidance of open elevated arms, and increased avoidance of the lit area during the first sessions of passive avoidance procedure without foot shock. 20702688_These results indicate that SV2 performs at least two actions at the synapse that contribute to neurotransmitter release. 20843861_SV2A is selectively localized in motor nerve terminals on slow (type I and small type IIA) muscle fibers. 21124874_Loss of synaptic vesicle glycoprotein 2a is associated with inhibition of tetanus neurotoxin into central neurons. 22266237_study suggests that SV2A specifically regulates hilar GABAergic neurotransmission in the kindled hippocampus probably as a compensatory or prophylactic mechanism against kindling epileptogenesis 22847229_Action potential- and Ca(2+) -dependent glutamatergic & GABAergic synaptic transmission are differentially altered in the hippocampus of SV2A-deficient mice, but the mechanism of exocytosis itself is not changed. 22908222_Glycosylation is dispensable for sorting of synaptotagmin 1 but is critical for targeting of SV2 and synaptophysin to recycling synaptic vesicles. 23937191_observed alterations of SV2A expression in hippocampus are consistent with the hypothesis that SV2A absence favors epileptic seizures around postnatal day 7 25673844_This study reveals fundamental details of how phosphorylation of Thr84 on SV2A controls its interaction with synaptotagmin-1 and implicates SV2A as a phospho-dependent chaperone 32341095_An Epilepsy-Associated SV2A Mutation Disrupts Synaptotagmin-1 Expression and Activity-Dependent Trafficking. |
ENSG00000159164 |
SV2A |
70.512698 |
2.3320703890 |
1.221611 |
0.37534420 |
10.475120 |
0.00120992942807890001451209815996890029055066406726837158203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0054768633130751276719139220006127288797870278358459472656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
86.3668889765404 |
22.7243170803203 |
37.0344263116291 |
7.47755420851216 |
| ENSMUSG00000038517 |
73174 |
Tbkbp1 |
protein_coding |
A2A9T0 |
FUNCTION: Adapter protein which constitutively binds TBK1 and IKBKE playing a role in antiviral innate immunity. Essential for the efficient induction of IRF-dependent transcription following infection with Sendai virus. {ECO:0000269|PubMed:17568778}. |
Alternative splicing;Coiled coil;Immunity;Innate immunity;Metal-binding;Phosphoprotein;Reference proteome;Zinc;Zinc-finger |
|
|
mmu:73174; |
cytoplasm [GO:0005737]; metal ion binding [GO:0046872]; I-kappaB kinase/NF-kappaB signaling [GO:0007249]; innate immune response [GO:0045087] |
17568778_results suggest that efficient signal transduction upon viral infection requires SINTBAD, TANK and NAP1 because they link TBK1 and IKKi to virus-activated signalling cascades 30022064_SINTBAD regulates IL-15-induced autophagy and NKT cell survival. |
ENSG00000198933 |
TBKBP1 |
228.390791 |
3.9912043539 |
1.996824 |
0.19023293 |
107.890654 |
0.00000000000000000000000028402352248885847308836290075288068801082940730083141321266210086110906574141665714705595746636390686035156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000157892200531032570315735504357231012970951413982505802214176900306885631408704284694977104663848876953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
359.99759566782 |
47.6067495609752 |
90.1977357582082 |
9.44103278484706 |
| ENSMUSG00000038523 |
75480 |
1700003F12Rik |
protein_coding |
A2AKE5 |
|
Reference proteome |
|
|
|
|
|
ENSG00000149609 |
C20orf144 |
76.468618 |
2.1599764513 |
1.111016 |
0.32193483 |
11.866629 |
0.00057148755717349925854764425281473450013436377048492431640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0028152970474989457740078169223352233530022203922271728515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
105.334293101784 |
23.7007202994886 |
48.7664080970949 |
8.44731746493304 |
| ENSMUSG00000038530 |
19736 |
Rgs4 |
protein_coding |
Q5D078 |
FUNCTION: Inhibits signal transduction by increasing the GTPase activity of G protein alpha subunits thereby driving them into their inactive GDP-bound form. Activity on G(z)-alpha is inhibited by phosphorylation of the G-protein. Activity on G(z)-alpha and G(i)-alpha-1 is inhibited by palmitoylation of the G-protein. {ECO:0000256|ARBA:ARBA00025480}. |
Lipoprotein;Palmitate;Phosphoprotein;Reference proteome |
|
|
mmu:19736; |
cytosol [GO:0005829]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; G-protein alpha-subunit binding [GO:0001965]; GTPase activator activity [GO:0005096]; protein kinase binding [GO:0019901]; brain development [GO:0007420]; dorsal root ganglion development [GO:1990791]; negative regulation of cell growth involved in cardiac muscle cell development [GO:0061052]; negative regulation of dopamine receptor signaling pathway [GO:0060160]; negative regulation of G protein-coupled receptor signaling pathway [GO:0045744]; negative regulation of glycine import across plasma membrane [GO:1900924]; negative regulation of potassium ion transmembrane transport [GO:1901380]; positive regulation of excitatory postsynaptic potential [GO:2000463]; positive regulation of GTPase activity [GO:0043547]; positive regulation of heart rate [GO:0010460]; regulation of actin filament organization [GO:0110053]; regulation of calcium ion transport [GO:0051924]; response to amphetamine [GO:0001975]; response to cocaine [GO:0042220]; response to ethanol [GO:0045471]; response to morphine [GO:0043278] |
14627646_In embryonic development transcription of RGS4 occurs in a highly dynamic manner in a small set of peripheral and central neuronal precursors. This expression pattern overlaps extensively with that of the neuronal identity determinant Phox2b 15548600_Rgs4 regulates vascular endothelial growth factor signaling. 15561937_RGS4 transgenic mice were resistant to streptozotocin induced cardiac fetal gene induction. 15870291_Behavioral testing on mutant adults revealed subtle sensorimotor deficits but, so far, supported neither the proposed status of Rgs4 as a schizophrenia susceptibility gene nor a role of Rgs4 in the acute or chronic response to opioids. 16217033_These results establish RGS4 and RGS5 as in vivo substrates of the mammalian N-end rule pathway and also suggest that the O2-ATE1-UBR1/UBR2 proteolytic circuit plays a role in RGS-regulated G protein signaling in the cardiovascular system. 16996696_These data suggest that altering RGS4 function will produce a complex neuronal phenotype with cell- and subdomain-specificity in the cerebral cortex. 17707117_Full length cloning and expression analysis of splice variants of Rgs4 were performed. 17707117_Multiple splice variant forms of RGS4 and Rgs4 are expressed in human and mouse brain and show differential expression across brain compartments. 18443239_RGS4 plays important roles in guanylyl cyclase-A -mediated inhibition of cardiac hypertrophy. 18493969_dramatic changes in expression of RGS4 mRNA in selective regions of the PLC-beta1 knockout mouse brain, particularly the CA1 region of the hippocampus 18625716_Affymetrix expression profiling shows that the C terminus of Runx2 regulates genes involved in G protein-coupled receptor signaling Rgs2, Rgs4, Rgs5, Rgs16, Gpr23, Gpr30, Gpr54, Gpr64, and Gna13. 18635652_RGS4 controls the balance between adipose tissue lipolysis and lipogenesis. 18658048_RGS4 plays an important role in regulating sinus rhythm by inhibiting parasympathetic signaling and I(KACh) activity 19059885_Regulator of G-protein signaling 4 suppresses LPS-induced MUC5AC overproduction in the airway. 19201815_Prostaglandin E(2) can induce MUC5AC overproduction via the EP(4) receptor and that RGS4 may have suppressive effects in controlling MUC5AC overexpression in the airway. 19914603_These findings provide fundamentally novel information concerning the role of RGS4 in the cellular mechanisms underlying the diverse actions of opiate drugs in the nervous system 20385802_RGS4 is a potent negative regulator of M3R function in pancreatic beta-cells, and may represent a potential target to promote insulin release for therapeutic purposes. 20430014_The RGS protein family may play an important role in the functioning of neural systems that are affected by chronic alcohol abuse; cirrhotic alcoholics have lower expression levels of RGS4 mRNA than controls and non-cirrhotic alcoholics. 21215802_Data show that absence of the regulatory protein RGS4 rescues several fragile-X related phenotypes, including increased body weight, abnormal synaptic protein expression and inhibition of social behaviors, in FMR1 knockout mice. 21412219_RGS4 antagonizes the development of renal dysfunction in response to ischemia reperfusion injury. 21911471_Rgs4 is expressed in endocrine progenitors of both zebrafish and mouse, and its expression in the mouse pancreatic epithelium is strictly dependent upon Ngn3, and loss of function of Rgs4 results in islet fragmentation in both organisms. 22730439_Findings support the concept that the inhibitory effects of SPL on M3R activity are mediated by RGS4. 23106268_RGS4 expression in mouse embryo is regulated by Lmx1a transcription factor. 23454748_A novel nitric oxide-RGS4 signal pathway mechanism that couples cardiac vessel growth with myocyte growth and heart size. 23630294_dramatic up-regulation of RGS4 expression in the nucleus accumbens of mice treated with monoamine-directed antidepressants 24318880_RGS6-Gbeta5, but not RGS4, is the primary RGS modulator of parasympathetic HR regulation and SAN M2R-IKACh signaling in mice. 25469849_Thus, RGS4 expression, specific to renal vascular smooth muscle cells, inhibits angiotensin II-mediated cytokine signaling and macrophage recruitment during reperfusion, distinct from vasomotor regulation. 25695583_Spinal RGS4 inhibited the endogenous or exogenous OR-mediated antinociceptive effect in the formalin pain test. 26926565_RGS4 in the paraventricular nucleus regulates blood corticosteroid-related GABAB receptor signaling during the acute stress response. 28219718_Study investigated whether RGS4 could participate in signalling pathways to regulate neurotropic events. These observations suggest that RGS4 is implicated in opioid dependent neuronal differentiation and neurite outgrowth via a 'non-canonical' signaling pathway regulating STAT5B-directed responses. 28853915_These results provide support for the hypothesis that increasing RGS4 expression and/or function could be a viable therapeutic strategy for asthma. 29754475_The nucleus-accumbens-specific knockdown of RGS4 significantly increased the behaviors associated with morphine and did so by phosphorylation of the GluR1 (Ser831) and NR2A (Tyr1325) glutamate receptors in the NAc. 30279737_Genetic and pharmacological inhibition of RGS4 resulted in a significant decrease in SLC7A11 (xCT) expression and hypofunction of system xc(-) and reduced glutamatergic function in organotypic brain slice cultures. RGS4 deficit induces dysregulation and dysfunction of system xc(-), which further results in functional deficits of the glutamatergic system and subsequently to schizophrenia-related behavioral phenotypes. 31030617_Regulator of G Protein Signaling 2 Facilitates Uterine Artery Adaptation During Pregnancy in Mice. 31308097_RGS4 plays a prominent role in the maintenance of chronic pain symptoms in male and female mice. Using genetically modified mice, we show a dynamic role of RGS4 in recovery from symptoms of sensory hypersensitivity deriving from hindpaw inflammation or hindlimb nerve injury. We also demonstrate an important role of RGS4 actions in gene expression patterns induced by chronic pain states in the mouse thalamus. 32199913_RGS4 promotes allergen- and aspirin-associated airway hyperresponsiveness by inhibiting PGE2 biosynthesis. 33892733_The association between RGS4 and choline in cardiac fibrosis. |
ENSG00000117152 |
RGS4 |
122.781262 |
3.0533259997 |
1.610382 |
0.23785596 |
45.542429 |
0.00000000001493678971274551597701920189583866226123998188768382533453404903411865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000002615140010397008303673747795322539547169249374292121501639485359191894531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
188.383428199714 |
31.1995187723495 |
61.6977775125141 |
8.05361394571193 |
| ENSMUSG00000038600 |
140494 |
Atp6v0a4 |
protein_coding |
Q920R6 |
FUNCTION: Subunit of the V0 complex of vacuolar(H+)-ATPase (V-ATPase), a multisubunit enzyme composed of a peripheral complex (V1) that hydrolyzes ATP and a membrane integral complex (V0) that translocates protons (By similarity). V-ATPase is responsible for acidifying and maintaining the pH of intracellular compartments and in some cell types, is targeted to the plasma membrane, where it is responsible for acidifying the extracellular environment (By similarity). Involved in normal vectorial acid transport into the urine by the kidney (By similarity). {ECO:0000250|UniProtKB:Q29466, ECO:0000250|UniProtKB:Q93050, ECO:0000250|UniProtKB:Q9HBG4}. |
Cell membrane;Hydrogen ion transport;Ion transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:140494; |
apical part of cell [GO:0045177]; apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; brush border [GO:0005903]; brush border membrane [GO:0031526]; endosome [GO:0005768]; plasma membrane [GO:0005886]; synaptic vesicle membrane [GO:0030672]; vacuolar proton-transporting V-type ATPase complex [GO:0016471]; vacuolar proton-transporting V-type ATPase, V0 domain [GO:0000220]; ATPase binding [GO:0051117]; P-type proton-exporting transporter activity [GO:0008553]; proton-transporting ATPase activity, rotational mechanism [GO:0046961]; ossification [GO:0001503]; proton transmembrane transport [GO:1902600]; regulation of pH [GO:0006885]; renal tubular secretion [GO:0097254]; sensory perception of sound [GO:0007605]; synaptic vesicle lumen acidification [GO:0097401]; vacuolar acidification [GO:0007035] |
17595521_Immunolocalization demonstrated expression of all 'a' subunits in the proximal tubule and in the intercalated cells of the collecting system. PCR detected mRNAs encoding all four 'a' isoforms with a relative abundance in the following order: a4>a2=a1>a3. 19214237_Data suggestthat Foxi1 is necessary for expression of at least four subunits in three different epithelia and most likely is a major determinant for proper assembly of a functional vacuolar H(+)-ATPase complex at these locations. 22872862_Atp6v0a4 knockout mouse is a model of distal renal tubular acidosis with hearing loss, with additional extrarenal phenotype 22933323_V-ATPase a4 knockout mice suffer not only from severe acidosis but also from proximal tubule dysfunction with defective endocytic trafficking, proteinuria, phosphaturia and accumulation of lysosomal material. 23065636_The first detailed analysis of the structure and function of the auditory system in Atp6v0a4(-/-) knockout mice, is reported. 27368196_The identification of novel locations for the a4 subunit and other tissue-restricted subunits increases the range of unique subunit combinations making up the proton pump. 35988642_Isoform a4 of the vacuolar ATPase a subunit promotes 4T1-12B breast cancer cell-dependent tumor growth and metastasis in vivo. |
ENSG00000105929 |
ATP6V0A4 |
79.163831 |
2.9340463902 |
1.552892 |
0.30208079 |
26.140567 |
0.00000031744407591799533036787416863389044152654605568386614322662353515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000030642003576571007910939875573674839870363939553499221801757812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
117.565916352142 |
24.7969811270499 |
40.069549255704 |
6.65233299252091 |
| ENSMUSG00000038677 |
268935 |
Scube3 |
protein_coding |
Q66PY1 |
FUNCTION: Is a positive regulator of the BMP signaling pathway, required for proper chondrogenesis, osteogenesis and skeletal development (PubMed:33308444). It acts as coreceptor for BMP ligands, particularly BMP2 and BMP4, facilitating their interactions with BMP type I receptors (By similarity). It is required for ligand-induced recruitment of BMP receptors to lipid rafts (PubMed:33308444). Binds to TGFBR2 and activates TGFB signaling (By similarity). {ECO:0000250|UniProtKB:Q8IX30, ECO:0000269|PubMed:33308444}. |
Alternative splicing;Calcium;Disulfide bond;EGF-like domain;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:268935; |
cell surface [GO:0009986]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; BMP binding [GO:0036122]; BMP receptor binding [GO:0070700]; calcium ion binding [GO:0005509]; identical protein binding [GO:0042802]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of osteoblast differentiation [GO:0045669]; positive regulation of smoothened signaling pathway [GO:0045880]; signal transduction [GO:0007165] |
23383134_Scube3 is expressed in multiple tissues during development but is dispensable for embryonic survival in the mouse. 24849601_Scube3 may be a critical upstream regulator of fast fiber myogenesis by modulating fgf8 signaling during zebrafish embryogenesis 27815347_These findings correlate with characteristics of the rare metabolic bone disorder Paget disease of bone (PDB), associated with the chromosomal region of human SCUBE3 In addition, alterations in energy metabolism, behavior, and neurological functions were detected in Scube3(N294K/N294K) mice. The Scube3(N294K/N294K) mutant mouse line may serve as a new model for further studying the effect of impaired SCUBE3 gene function. |
ENSG00000146197 |
SCUBE3 |
489.414532 |
0.3060449998 |
-1.708184 |
0.23894504 |
48.980367 |
0.00000000000258537432456195494662352679913249874771308256526936020236462354660034179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000489806240195618134074802331902055404014295625358954566763713955879211425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
201.840472078347 |
33.5826895701264 |
659.512399133731 |
81.0006845017582 |
| ENSMUSG00000038721 |
15415 |
Hoxb7 |
protein_coding |
P09024 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15415; |
cytosol [GO:0005829]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; anterior/posterior pattern specification [GO:0009952]; embryonic skeletal system morphogenesis [GO:0048704]; myeloid cell differentiation [GO:0030099]; positive regulation of branching involved in ureteric bud morphogenesis [GO:0090190]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
15716052_three short cis-acting sequences within this promoter segment that cooperate to induce high-level expression specifically in kidney collecting duct cells. 18463397_Hoxb7 inhibits transgenic HER-2/neu-induced mouse mammary tumor onset but promotes progression and lung metastasis 25428821_Results demonstrate HOXB7 as a master factor driving progenitors behavior lifetime, providing a better understanding of bone senescence and leading to an optimization of MSC performance. 26902420_Results point out the complex interplay between the DSB DNA repair activity and the homeoproteins HoxB7 and Cdx2 in colon cancer cells. 30664713_expression resulted in significantly greater numbers of macroscopic lung metastatic cancer nodules |
ENSG00000260027 |
HOXB7 |
11.703507 |
0.1280629572 |
-2.965075 |
0.91426383 |
10.505799 |
0.00119000493793639211363710739277621541987173259258270263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0053962950921843180762671465799940051510930061340332031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
2.98926872571905 |
2.02372385862026 |
23.3421809942016 |
9.48813485901583 |
| ENSMUSG00000038738 |
243961 |
Shank1 |
protein_coding |
D3YZU1 |
FUNCTION: Seems to be an adapter protein in the postsynaptic density (PSD) of excitatory synapses that interconnects receptors of the postsynaptic membrane including NMDA-type and metabotropic glutamate receptors, and the actin-based cytoskeleton. Plays a role in the structural and functional organization of the dendritic spine and synaptic junction. Overexpression promotes maturation of dendritic spines and the enlargement of spine heads via its ability to recruit Homer to postsynaptic sites, and enhances presynaptic function (By similarity). {ECO:0000250}. |
ANK repeat;Cytoplasm;Differentiation;Methylation;Neurogenesis;Phosphoprotein;Reference proteome;Repeat;SH3 domain;Synapse |
|
|
mmu:243961; |
cytoplasm [GO:0005737]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; excitatory synapse [GO:0060076]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic membrane [GO:0045211]; Schaffer collateral - CA1 synapse [GO:0098685]; synapse [GO:0045202]; ankyrin repeat binding [GO:0071532]; identical protein binding [GO:0042802]; ionotropic glutamate receptor binding [GO:0035255]; protein C-terminus binding [GO:0008022]; protein-containing complex binding [GO:0044877]; scaffold protein binding [GO:0097110]; SH3 domain binding [GO:0017124]; signaling receptor complex adaptor activity [GO:0030159]; somatostatin receptor binding [GO:0031877]; structural constituent of postsynaptic density [GO:0098919]; synaptic receptor adaptor activity [GO:0030160]; adult behavior [GO:0030534]; associative learning [GO:0008306]; dendritic spine morphogenesis [GO:0060997]; determination of affect [GO:0050894]; habituation [GO:0046959]; long-term memory [GO:0007616]; modulation of chemical synaptic transmission [GO:0050804]; negative regulation of actin filament bundle assembly [GO:0032232]; neuromuscular process controlling balance [GO:0050885]; olfactory behavior [GO:0042048]; positive regulation of dendritic spine development [GO:0060999]; positive regulation of excitatory postsynaptic potential [GO:2000463]; protein localization to synapse [GO:0035418]; protein-containing complex assembly [GO:0065003]; regulation of AMPA receptor activity [GO:2000311]; righting reflex [GO:0060013]; social behavior [GO:0035176]; synapse maturation [GO:0060074]; vocalization behavior [GO:0071625] |
18272690_These results affirm the importance of Shank1 for synapse structure and function in vivo, and they highlight a differential role for Shank1 in specific cognitive processes. 19640847_Semi-quantitative analysis of RNA levels in FMRP immunoprecipitates showed that in the mouse brain mRNAs encoding PSD components, such as Shank1, SAPAP1-3, PSD-95, and the glutamate receptor subunits NR1 and NR2B, are associated with FMRP. 20868654_Shank1 null mice do not demonstrate autism-relevant social interaction deficits, but confirm and extend a role for Shank1 in motor functions. 21695253_The reduced levels of ultrasonic vocalizations and scent marking behavior in Shank1(-/-) mice are consistent with a phenotype relevant to social communication deficits in autism. 22984429_These findings provide convincing evidence for Shank 1A expression in both the inner and outer plexiform layers, and indicate a potential role for PSD-95/Shank 1 complexes at cone synapses in the outer retina. 24820912_This study demonistrated that Shank1+/- heterozygous and to a lesser extent Shank1-/- null mutant mice displayed slightly elevated levels of self-grooming behavior as adults, but not as juveniles. 25637745_Deletion of Shank1 has minimal effects on the molecular composition and function of glutamatergic afferent postsynapses in the mouse inner ear. 25816842_Results suggest that Shank1 scaffold in PV+ interneurons regulates excitatory synaptic strength and participates in the maintenance of excitatory and inhibitory balance in excitatory neurons. 25917711_Since mutations in all three human SHANK genes are linked to neuropsychiatric disorders such as autism and schizophrenia, Shank mutant mice serve as corresponding in vivo model systems--{REVIEW} 26419918_Expression of SHANK genes is a key component of autism spectrum disorder-associated pathways as the protein products are enriched in the postsynaptic density (PSD) of glutamatergic neurons. 26819149_reduction of PV(+) neurons was in all cases, i.e., in PV+/-, Shank1-/- and Shank3B-/- mice, was due to a reduction in Pvalb mRNA and PV protein 28500650_The findings of this study indicated that Shank1 deletions lead to an aberrant cognitive phenotype characterized by severe impairments in object recognition memory and increased hippocampal BDNF levels, possibly due to epigenetic modifications. 28963042_A review: Shank1 knockout mouse model for autism spectrum disorder (ASD) displays moderate impairments in (I) social behavior, (II) communication, and (III) repetitive and stereotyped patterns of behavior, together with (IV) aberrant cognitive phenotypes, consistent with the symptoms present in humans diagnosed with ASD and carrying SHANK1 deletions. 29218848_Study demonstrates that SHANK1 is developmentally expressed in hippocampal neurons and glial cells. Although the specific role for this developmental expression pattern in these different cell types is not fully understood, the current study provides the first developmental cell-type-specific expression profile. 30053575_SHANK1 plays a role in neocortical learning-induced dendritic spine plasticity underlying learning and normal cognition; and in mechanisms mediating abnormalities of impaired cognition. 33402706_Developmental impaired Akt signaling in the Shank1 and Shank3 double knock-out mice. 35181969_Suv39h1 regulates memory stability by inhibiting the expression of Shank1 in hippocampal newborn neurons. 35388181_A recurrent SHANK1 mutation implicated in autism spectrum disorder causes autistic-like core behaviors in mice via downregulation of mGluR1-IP3R1-calcium signaling. |
ENSG00000161681 |
SHANK1 |
3782.109689 |
2.0603470460 |
1.042887 |
0.09494564 |
118.081288 |
0.00000000000000000000000000166425737834022431134998844540854932754768278268463716950295981500051946573581762578442067024298012256622314453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000001073537820671256131404098629808763246686126267512473737189066791804454084613418274329887935891747474670410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5071.37526101255 |
333.994790271619 |
2461.41797849179 |
122.198958201921 |
| ENSMUSG00000038765 |
16917 |
Lmx1b |
protein_coding |
H3BJD7 |
Human_homologues FUNCTION: Transcription factor involved in the regulation of podocyte-expressed genes (PubMed:24042019, PubMed:28059119). Essential for the specification of dorsal limb fate at both the zeugopodal and autopodal levels. {ECO:0000269|PubMed:24042019, ECO:0000269|PubMed:28059119}. |
DNA-binding;Homeobox;LIM domain;Metal-binding;Nucleus;Reference proteome;Repeat;Zinc |
|
|
|
nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; system development [GO:0048731] |
11956244_Transcriptional induction of slit diaphragm genes by Lmx1b is required in podocyte differentiation. 11956245_The LIM-homeodomain transcription factor Lmx1b plays a crucial role in podocytes 11978876_Review. Studies of Lmx1b knockout mice show it is a transactivator required for coordinate expression of Col4a3 and Col4a4, and other podocyte function genes. 12897786_During development of 5-HT neurons, Lmx1b is a critical intermediate factor that couples early specification with nerve terminal differentiation. 14602809_Ectopic expression of Lmx1b plus Pet-1 is able to induce formation of 5-HT cells in the most ventral spinal cord, where Nkx2.2 is normally expressed. Combined expression of Lmx1b, Pet-1, and Nkx2.2, drives 5-HT differentiation in the dorsal spinal cord 15007643_Myogenic precursors in limb bud were devoid of Lmx1b expression. Lmx1b is not only expressed at early stages of limb development but maintains its pattern, at least until after birth. New details of Lmx1b expression on cellular basis within limb. 15183306_Lmx1b expression localized to dorsal joint-forming regions, to developing tendons and ligaments 15215291_data identify Lmx1b as a key regulator of spinal cord roof plate induction and function 15229182_Results indicate that Lmx1b has a pivotal role in genetic cascades that control the assembly of circuitry in the superficial dorsal horn. 15562281_Lmx1b is required for the differentiation and migration of neurons within the dorsal spinal cord. The inability of afferent sensory neurons to migrate into the dorsal horn is entirely consistent with diminished pain responses in nail patella syndrome 17151281_A critical role is revealed for Lmx1b in maintaining the differentiated status of serotonergic (5-HT) neurons in conditional knock-out Lmx1bf/f/p mice compared with wild-type mice. 17166916_Lmx1b plays an essential role in the development of the tectum and cerebellum by regulating expression of Fgf8, Wnt1 and several isthmus-related transcription factors in the midbrain-hindbrain boundary 17316599_The podocyte-specific inactivation of Lmx1b, Ldb1 and E2a yields new insight into a transcriptional network in podocytes 17657578_First report of a phenotype in heterozygous Lmx1b (Lmx1b+/-) knockout animals. 18351676_Results suggest that LMX1B controls different targets along the proximal-distal axis of the limb, and suggest the existence of a dorsal proximal limb region that is rich in mRNAs requiring Lmx1b for their expression. 18512225_Lmx1b is involved in the postnatal maturation of certain types of neurons and maintenance of their normal functions in the adult brain. 18522499_expression of shRNA against GPM6A markedly reduced the expression of neuroectodermal-associated genes (OTX1, Lmx1b, En1, Pax2, Pax5, Sox1, Sox2, and Wnt1), and also the number of neural stem cells (NSC) derived from D3mshM6A cells 18942141_The second wave of expression at E12.5 includes Lhx3, 5, 9, Isl2, and Lmx1b in the differentiating sensory epithelia with cellular specificities. 18996370_These data demonstrate for the first time that LMX1B directly regulates transcription of a subset of NF-kappaB target genes in cooperation with nuclear p50/p65 NF-kappaB. 19607821_Foxa1 and Foxa2 function both upstream of and cooperatively with Lmx1a and Lmx1b in a feedforward loop promoting mesodiencephalic dopaminergic neuron development 19741143_When Lmx1b expression is deleted at the dorsal midline, Lmx1b/Lmx1a-null mutants fail to generate a hindbrain roof plate. 20035737_obtained evidence that Lmx1a and Lmx1b cooperate with Foxa2 to specify mesDA neuron identity by gain-of-function approaches using transgenic mice 20215352_Results indicate that whereas Lmx1b activity is required autonomously in skeletal progenitors to direct dorsal pattern, loss or gain of Lmx1b activity in skeletal progenitors has no effect on muscle or connective tissue patterning. 20568247_link a specific transcription factor Lmx1b to trabecular meshwork formation and demonstrate specific requirements for Lmx1b in maintaining the integrity of adult anterior segment. 20589901_report 14 genes that require Lmx1b for their normal expression in the dorsal limb or the restriction of their expression to the ventral limb. 20621716_In Lmx1b knockout ((-)(/)(-)) mice, V primary afferent projections to PrV are normal, albeit reduced in number, whereas the PrV-thalamic lemniscal pathway is sparse and develops late. 21752929_data establish that Lmx1b influences the differentiation of multiple neuronal subtypes in the ventral midbrain, whereas Lmx1a appears to be exclusively devoted to the differentiation of the dopamine neuron lineage 21880902_This study demonstrated that Lmx1a and Lmx1b function cooperatively to regulate proliferation, specification, and differentiation of mDA progenitors, including their floor plate-like properties. 22397680_This study demonistrated that Lmx1b promotes a neuronal glutamate phenotype and suppresses a GABA one in the embryonic trigeminal brainstem complex 22417325_Lmx1b genes are associated with skeletal development. 23308148_A protein complex was identified containing PSPC1, PSF and LMX1B, suggesting the existence of this complex in mesodiencephalic dopaminergic neurons. 23532063_Lmx1b, a key transcription factor for the specification of 5-HT and dopaminergic transmitter phenotypes during embryogenesis, determines some peptide phenotypes in these neurons as well. 23716677_Data indicate that tryptophan hydroxylase 2 Tph2-/- and LIM homeobox transcription factor 1 beta Lmx1b-/- females displayed a change in sexual preference. 23990680_Cell biological and biophysical experiments with primary podocytes isolated after 1 week of Lmx1b inactivation indicated dysregulation of actin cytoskeleton organization 24348261_We posit that microRNA modulation of the Lmx1b/Wnt axis in the early midbrain/isthmus could determine midbrain size and allocation of dopamine progenitors. 24809698_Co-immunoprecipitation experiments show that both wild-type and Icst LMX1B are found in complexes with LIM domain binding protein 1 (LDB1), resulting in lower levels of functional LMX1B in Icst heterozygotes than null heterozygotes 25834049_results demonstrate an important role for the intersection of Lmx1b and Hoxb8::cre expression in the development of nociceptive dorsal horn circuits critical for mechanical and thermal pain processing. 26444681_Otx2 critically depends on Lmx1b for the formation of mdDA neurons. 27407143_Data show that sustained expression of Lmx1a and Lmx1b is required for the survival of adult midbrain dopaminergic neurons. 28455377_This is the first report to describe genome-wide Lmx1b binding during limb development. 29038581_Lmx1b is required for the topographical organization of dopaminergic innervation in the striatum.Lmx1b regulates Plxnc1 expression. 29852132_Authors conclude that Lmx1b plays a key role as an anti-osteogenic factor in patterning the head mesenchyme into areas with different osteogenic competence. In turn, this patterning event is crucial to generating the proper organization of the bones and soft tissue joints of the calvaria. 31053111_identified a novel NPS-causing LMX1B mutation and expanded the spectrum of mutations in the LMX1B gene. The c.712_714delTTC mutation may affect the quaternary structure of LMX1B, which is essential for the specification of dorsal limb fate at both zeugopodal and autopodal levels, leading to typical NPS 31355748_The authors report that LIM homeodomain factor 1b (Lmx1b)-deficient serotonin (5-HT) neurons fail to generate axonal projections to the forebrain and spinal cord. Stage-specific targeting demonstrates that Lmx1b is required at successive stages to control 5-HT axon primary outgrowth, selective routing, and terminal arborization. 34545091_Identification of limb-specific Lmx1b auto-regulatory modules with Nail-patella syndrome pathogenicity. 36641754_Failure of digit tip regeneration in the absence of Lmx1b suggests Lmx1b functions disparate from dorsoventral polarity. |
ENSG00000136944 |
LMX1B |
55.767615 |
4.1969710471 |
2.069349 |
0.41976276 |
23.156641 |
0.00000149326637670169010036781958444818130260500765871256589889526367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000128723449065761980103245881723594834511459339410066604614257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
90.3871875299137 |
26.6714522430165 |
21.5362904616062 |
4.92774643710123 |
| ENSMUSG00000038807 |
380711 |
Rap1gap2 |
protein_coding |
Q5SVL6 |
FUNCTION: GTPase activator for the nuclear Ras-related regulatory protein RAP-1A (KREV-1), converting it to the putatively inactive GDP-bound state. {ECO:0000250}. |
Alternative splicing;Cytoplasm;GTPase activation;Phosphoprotein;Reference proteome |
|
|
mmu:380711; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; neuron projection [GO:0043005]; nuclear membrane [GO:0031965]; plasma membrane [GO:0005886]; GTPase activator activity [GO:0005096]; activation of GTPase activity [GO:0090630]; negative regulation of neuron projection development [GO:0010977]; regulation of cell size [GO:0008361]; regulation of small GTPase mediated signal transduction [GO:0051056] |
16169686_Some mKIAA genes are apparently relevant to retinoic acid-induced neurite outgrowth. (mKIAA gene) 22732430_Rap1gap2 overexpression limits neurite outgrowth and branching in Neuro-2a cells, and counteracts Rap1-induced augmentation of neurite outgrowth |
ENSG00000132359 |
RAP1GAP2 |
28.755070 |
0.1209815000 |
-3.047142 |
0.55968475 |
29.931922 |
0.00000004474846508668431692615135371500489913643150430289097130298614501953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000004921361878703071863222647495950123897046069032512605190277099609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.26090687389046 |
2.49026541301033 |
51.750944367891 |
13.0344116806297 |
| ENSMUSG00000039055 |
268465 |
Eme1 |
protein_coding |
Q8BJW7 |
FUNCTION: Interacts with MUS81 to form a DNA structure-specific endonuclease with substrate preference for branched DNA structures with a 5'-end at the branch nick. Typical substrates include 3'-flap structures, replication forks and nicked Holliday junctions. May be required in mitosis for the processing of stalled or collapsed replication forks. {ECO:0000269|PubMed:14609959}. |
DNA damage;DNA recombination;DNA repair;Endonuclease;Hydrolase;Isopeptide bond;Magnesium;Metal-binding;Nuclease;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:268465; |
chromatin [GO:0000785]; endodeoxyribonuclease complex [GO:1905347]; heterochromatin [GO:0000792]; Holliday junction resolvase complex [GO:0048476]; nuclear replication fork [GO:0043596]; nucleolus [GO:0005730]; DNA binding [GO:0003677]; endonuclease activity [GO:0004519]; metal ion binding [GO:0046872]; double-strand break repair [GO:0006302]; mitotic intra-S DNA damage checkpoint signaling [GO:0031573]; replication fork processing [GO:0031297]; resolution of meiotic recombination intermediates [GO:0000712]; response to intra-S DNA damage checkpoint signaling [GO:0072429] |
17036055_Mus81-Eme1- and Rad54-mediated homologous recombination are involved in the same DNA replication-dependent interstrand crosslinks repair pathway 18692478_These findings link Eme1 with the replication-associated chromatin modifier functions of Np95 in the cellular response to DNA damage. 24076219_indicate that SLX1 and MUS81-EME1 nucleases act together to resolve Holliday junctions (HJs) in a manner that requires tethering to SLX4. 27383418_Study found that a Gen1 mutation alone did not affect DNA repair or meiotic recombination in mice; it caused synthetic lethality when combined with Eme1 mutations at an early embryonic stage. Also, combination of Gen1 and Eme1 mutations makes embryonic fibroblasts more sensitive to DNA damage & mice less capable of meiotic recombination. These results indicate that Gen1 and Eme1 are functionally redundant in mice. 31170160_Double mutant mice homozygous for the Mlh3DN/DN and Mus81-/- mutations show losses in chiasmata close to those observed in Mlh3-/- males, indicating that the MUS81-EME1-regulated crossover pathway can only partially account for the increased residual chiasmata in Mlh3DN/DN spermatocytes |
ENSG00000154920 |
EME1 |
236.827575 |
2.0423114486 |
1.030203 |
0.17505118 |
34.319239 |
0.00000000467735149524529536966294687933345941566187775606522336602210998535156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000598198303741195050331632375451018646117518073879182338714599609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
316.03474008572 |
38.4406261143619 |
154.743655921818 |
14.5349366258281 |
| ENSMUSG00000039109 |
74145 |
F13a1 |
protein_coding |
Q8BH61 |
FUNCTION: Factor XIII is activated by thrombin and calcium ion to a transglutaminase that catalyzes the formation of gamma-glutamyl-epsilon-lysine cross-links between fibrin chains, thus stabilizing the fibrin clot. Also cross-link alpha-2-plasmin inhibitor, or fibronectin, to the alpha chains of fibrin. {ECO:0000250|UniProtKB:P00488}. |
Acetylation;Acyltransferase;Blood coagulation;Calcium;Cytoplasm;Glycoprotein;Hemostasis;Metal-binding;Reference proteome;Secreted;Transferase;Zymogen |
|
This gene encodes subunit A of the coagulation factor XIII that catalyzes the final step of the blood coagulation pathway. The encoded protein associates with subunit B to form a heterotetrameric proenzyme that undergoes thrombin-mediated proteolysis to generate active factor XIIIa. The transglutaminase activity of factor XIIIa is required for the calcium-dependent crosslinking of fibrin, leading to the formation of a clot. Mice lacking the encoded protein display impaired reproduction and reduced survival due to bleeding episodes, hematothorax, hematoperitoneum and subcutaneous hemorrhage. Additionally, mice lacking the encoded protein exhibit impaired wound healing and inadequate healing of myocardial infarction. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Sep 2015]. |
mmu:74145; |
collagen-containing extracellular matrix [GO:0062023]; cytoplasm [GO:0005737]; extracellular region [GO:0005576]; metal ion binding [GO:0046872]; protein-glutamine gamma-glutamyltransferase activity [GO:0003810]; blood coagulation [GO:0007596]; blood coagulation, fibrin clot formation [GO:0072378]; peptide cross-linking [GO:0018149] |
12529747_impaired clot stabilization in FXIII-A mutant mice 12933578_Maternal FXIII plays a critical role in uterine hemostasis and maintenance of the placenta during gestation. 15131115_platelet FXIII and calpain have roles in regulating integrin alpha(IIb)beta3 adhesive function 16113836_FXIII has an essential role in wound repair and remodeling 16368540_analysis of the expression, localization and activity of TG2 and FXIIIA in mineralizing tissues [review] 16469487_Results demonstrate that MC3T3-E1/C14 osteoblasts express two transglutaminase isoforms--TG2 and FXIIIA. 17183677_All FXIII2/2 and FXIII2/+ mice died within 5 days after MI from left ventricular (LV) rupture and had locally reduced levels of FXIII. 17341477_the entire FXIIIA gene is expressed, thus indicating that the 37-kDa FXIIIA is not a splice variant but a product of posttranslational proteolytic processing. 18278192_Carditis and fibrosis in FXIII-deficient mice might be caused by a faulty or delayed reparative process that was initiated by abnormal haemorrhagic events within heart tissue. 18315549_FXIIIa supports the early survival of micrometastases by a mechanism linked to NK cell function 19996413_Factor XIII A subunit deficieny is associated with impaired clot retraction 20086247_Knockout mouse studies suggest that plasma FXIII-A does not derive primarily (if at all) from circulating platelets and indeed may not derive from the megakaryocyte lineage. 20810110_Inhibition of TG2 and FXIIIA during early development of lesions reduced the macrophage content in the media of atherosclerotic vessels, while not affecting lesion size or geometric remodeling 21283799_factor XIIIA transglutaminase activity regulates osteoblast matrix secretion and deposition by affecting microtubule dynamics 21959563_FXIIIA mRNA was regulated by the MAP kinase pathway. 22045386_Physiological significance of mouse TG2 and Factor XIII through identification of substrate proteins in MC3T3-E1 cells. 22272319_Data indicate that FXIIIa-targeted probe signals showed good overlap with immune-fluorescent fibrin staining images. 23075332_Studies indicate that in FXIII-A-knockout mice, tail-tip bleeding times are doubled and clot stability, assessed by thromboelastography, was impaired compared with wild-type mice. 24934257_FXIII-A serves as a preadipocyte-bound proliferation/differentiation switch that mediates effects of hepatocyte-produced circulating pFN. 24983320_Fibrinogen from mice harboring the Fibgamma390-396A mutation exhibited reduced binding to FXIII, and plasma from these mice exhibited delayed FXIII activation and fibrin crosslinking, indicating these residues mediate FXIII binding and activation. 25336631_High Transglutaminase factor XIII promotes arthritis through mechanisms linked to inflammation and bone erosion. 25460579_5-HT inhibits Factor XIII-A-mediated plasma fibronectin matrix assembly and crosslinking in osteoblast cultures via direct competition with transamidation 26018251_related to islet dysfunction during the development of type 2 diabetes 26098308_FXIII is a significant determinant of mucosal healing and clinical outcome following inflammatory colitis induced mucosal injury 26324704_These findings expose a newly recognized, essential role for fibrin crosslinking during whole blood clot formation and consolidation and establish FXIIIa activity as a key determinant of thrombus composition and size. 27302551_Mast cells have the ability to produce and degrade FXIIIA depending on their chymase expression profile: mast cells expressing chymase degrade FXIIIA, whereas mast cells that do not express chymase mainly produce FXIIIA 27338836_A lack of factor XIII leads to prolonged clotting times, increased signs of inflammation, and elevated bacterial dissemination in streptococcal skin diseases. 27561317_These findings provide insight into assembly of the fibrinogen/FXIII-A2B2 complex in both physiologic and therapeutic situations. 28387755_Factor XIII-A and TG2 regulate resorption, adipogenesis and plasma fibronectin homeostasis in bone and bone marrow. 28596376_Suggest that resident macrophages maintain plasma FXIII-A and exclude the platelet lineage as a major contributor. 31786225_Assessment of expression and specific activities of transglutaminases TG1, TG2, and FXIII-A during osteoclastogenesis. 32678423_Sustained depletion of FXIII-A by inducing acquired FXIII-B deficiency. 34183396_Elimination of fibrin gamma-chain cross-linking by FXIIIa increases pulmonary embolism arising from murine inferior vena cava thrombi. 36006736_Mast cell-derived factor XIIIA contributes to sexual dimorphic defense against group B streptococcal infections. |
ENSG00000124491 |
F13A1 |
4987.037243 |
0.2842088460 |
-1.814977 |
0.09132257 |
374.168953 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000002314714521400651518643685389023578762218894815377957692780225229470777522569722779390378992353495249836209609068884224261546183798445201669996633004126290562284084633961812439136107 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000092783504184144008868893298111956190651095463591568061017435763263149698061377270822179630554338109755549823843751285502066885778546091987200916377708052403397997023914744357426399 |
Yes |
No |
2181.70272870976 |
138.193989700564 |
7676.40683642699 |
359.365805002607 |
| ENSMUSG00000039167 |
170757 |
Adgrl4 |
protein_coding |
Q923X1 |
FUNCTION: Endothelial orphan receptor that acts as a key regulator of angiogenesis. {ECO:0000250|UniProtKB:Q9HBW9, ECO:0000269|PubMed:23871637}. |
Calcium;Cell membrane;Disulfide bond;EGF-like domain;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Repeat;Signal;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:170757; |
cytoplasmic vesicle [GO:0031410]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; G protein-coupled receptor activity [GO:0004930]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; cell surface receptor signaling pathway [GO:0007166]; protein-containing complex assembly [GO:0065003] |
22606234_ELTD1 deficiency exacerbates cardiac hypertrophy and cardiac function induced by aortic banding-induced pressure overload by promoting both cardiomyocyte hypertrophy and cardiac fibrosis. 28806758_loss of GPR116 and ELTD1 expressed by other cells accounts for the observed cardiovascular and renal defects. 34347079_ELTD1 deletion reduces vascular abnormality and improves T-cell recruitment after PD-1 blockade in glioma. |
ENSG00000162618 |
ADGRL4 |
36.435318 |
0.0073699525 |
-7.084129 |
1.34592569 |
39.005891 |
0.00000000042252860722110249955207861566732017305492163927738147322088479995727539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000062105076124863926259077315564748056342381232752813957631587982177734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.519179811500407 |
0.557736949396037 |
70.4454758651894 |
26.2564982816855 |
| ENSMUSG00000039208 |
210029 |
Metrnl |
protein_coding |
Q8VE43 |
FUNCTION: Hormone induced following exercise or cold exposure that promotes energy expenditure. Induced either in the skeletal muscle after exercise or in adipose tissue following cold exposure and is present in the circulation. Able to stimulate energy expenditure associated with the browning of the white fat depots and improves glucose tolerance. Does not promote an increase in a thermogenic gene program via direct action on adipocytes, but acts by stimulating several immune cell subtypes to enter the adipose tissue and activate their prothermogenic actions. Stimulates an eosinophil-dependent increase in IL4 expression and promotes alternative activation of adipose tissue macrophages, which are required for the increased expression of the thermogenic and anti-inflammatory gene programs in fat. Required for some cold-induced thermogenic responses, suggesting a role in metabolic adaptations to cold temperatures. {ECO:0000269|PubMed:24906147}. |
Alternative splicing;Disulfide bond;Glycoprotein;Hormone;Reference proteome;Secreted;Signal |
|
|
mmu:210029; |
extracellular space [GO:0005615]; hormone activity [GO:0005179]; brown fat cell differentiation [GO:0050873]; energy homeostasis [GO:0097009]; fat cell differentiation [GO:0045444]; negative regulation of inflammatory response [GO:0050728]; positive regulation of brown fat cell differentiation [GO:0090336]; response to cold [GO:0009409]; response to muscle activity [GO:0014850] |
21985865_Cometin is a novel neurotrophic factor(meteorin protein family) that promotes neurite outgrowth and neuroblast migration in vitro and supports survival of spiral ganglion neurons in vivo. 24393292_Results show that Subfatin is a novel adipokine regulated by adipogenesis and obesity, with tissue distribution different from its homologue Meteorin 24906147_Study reports the identification of meteorin-like (Metrnl), a circulating factor that is induced in muscle after exercise and in adipose tissue upon cold exposure; increasing circulating levels of Metrnl stimulates energy expenditure and improves glucose tolerance and the expression of genes associated with beige fat thermogenesis and anti-inflammatory cytokines. 25486603_Metrnl represents a novel cytokine, which is likely involved in both innate and acquired immune responses. 26307585_Data (including data from studies in transgenic/knockout mice and 3T3-L1 cells) suggest expression of Metrnl in white adipocytes controls insulin sensitivity via activation of PPARg (peroxisome proliferator activated receptor gamma) signaling pathway. 29703203_The results of this study suggest that regular exercise is indispensable to reduce body weight and fat mass through upregulation of the muscle energy-sensing network and Metrnl protein levels, and retraining with dietary change is necessary to obtain the retraining effects more quickly. 29854769_Exercise-induced muscle Metrnl effectively reduces fat accumulation. 30213948_METRNL alleviates inflammation and insulin resistance and induces fatty acid oxidation through AMPK or PPARdelta-dependent signaling in skeletal muscle. 30464051_these observations indicate that Metrnl encodes a novel immunoregulatory cytokine associated with inflammatory responses 31949292_Aggravated ulcerative colitis caused by intestinal Metrnl deficiency is associated with reduced autophagy in epithelial cells. 32196931_The myokine meteorin-like (metrnl) improves glucose tolerance in both skeletal muscle cells and mice by targeting AMPKalpha2. 32694780_Meteorin-like facilitates skeletal muscle repair through a Stat3/IGF-1 mechanism. 33635944_Meteorin-like/Meteorin-beta protects heart against cardiac dysfunction. 34943988_Exercise Inhibits NLRP3 Inflammasome Activation in Obese Mice via the Anti-Inflammatory Effect of Meteorin-like. 35076116_The role of Meteorin-like in skeletal development and bone fracture healing. 35709278_Meteorin-like promotes heart repair through endothelial KIT receptor tyrosine kinase. 36130501_Meteorin links the bone marrow hypoxic state to hematopoietic stem/progenitor cell mobilization. |
ENSG00000176845 |
METRNL |
906.913958 |
0.4562052811 |
-1.132245 |
0.10658240 |
111.532243 |
0.00000000000000000000000004523875757600952704880301953065083758146686856278198880700784479091383291637917807292978977784514427185058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000026300639518999125723925515861564228382713483550808732611006271083151086820350883499486371874809265136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
574.631891930955 |
42.4908753915822 |
1259.59061798201 |
67.7928645350686 |
| ENSMUSG00000039209 |
68172 |
Rpl39l |
protein_coding |
Q9CQD0 |
|
3D-structure;Proteomics identification;Reference proteome;Ribonucleoprotein;Ribosomal protein |
|
|
mmu:68172; |
cytosolic large ribosomal subunit [GO:0022625]; structural constituent of ribosome [GO:0003735]; translation [GO:0006412] |
17660561_Transcripts highly enriched in ovary; gene identified as candidate female fertility factor. 24452241_Results show the RP paralog Rpl39l is highly expressed in Embryonic Stem Cells and its expression strongly correlates with hepatocellular carcinoma tumor (HCC) samples with high tumor grading and alpha-fetoprotein level giving it diagnostic potential. |
|
|
8.894079 |
0.0102481320 |
-6.608495 |
1.54669103 |
18.456079 |
0.00001738653402299020550776133264836431635558255948126316070556640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001232348470163735769822982435428571079683024436235427856445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.187328683420823 |
0.241576145259351 |
18.2793004803336 |
10.4413598205648 |
| ENSMUSG00000039217 |
16173 |
Il18 |
protein_coding |
P70380 |
FUNCTION: Pro-inflammatory cytokine primarily involved in epithelial barrier repair, polarized T-helper 1 (Th1) cell and natural killer (NK) cell immune responses (PubMed:26638072, PubMed:26638073). Upon binding to IL18R1 and IL18RAP, forms a signaling ternary complex which activates NF-kappa-B, triggering synthesis of inflammatory mediators (By similarity). Synergizes with IL12/interleukin-12 to induce IFNG synthesis from T-helper 1 (Th1) cells and natural killer (NK) cells (By similarity). Involved in transduction of inflammation downstream of pyroptosis: its mature form is specifically released in the extracellular milieu by passing through the gasdermin-D (GSDMD) pore (PubMed:30392956). {ECO:0000250|UniProtKB:Q14116, ECO:0000269|PubMed:26638072, ECO:0000269|PubMed:26638073, ECO:0000269|PubMed:30392956}. |
Cytokine;Cytoplasm;Direct protein sequencing;Inflammatory response;Reference proteome;Secreted |
|
|
mmu:16173; |
apical plasma membrane [GO:0016324]; cytoplasm [GO:0005737]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; interleukin-18 receptor binding [GO:0045515]; activation of protein kinase B activity [GO:0032148]; angiogenesis [GO:0001525]; cell population proliferation [GO:0008283]; cellular response to organic cyclic compound [GO:0071407]; cellular response to peptide hormone stimulus [GO:0071375]; cholesterol homeostasis [GO:0042632]; defense response to Gram-positive bacterium [GO:0050830]; detection of mechanical stimulus involved in sensory perception of pain [GO:0050966]; establishment of skin barrier [GO:0061436]; extrinsic apoptotic signaling pathway via death domain receptors [GO:0008625]; immune response [GO:0006955]; inflammatory response [GO:0006954]; interleukin-18-mediated signaling pathway [GO:0035655]; lipopolysaccharide-mediated signaling pathway [GO:0031663]; MAPK cascade [GO:0000165]; natural killer cell activation [GO:0030101]; natural killer cell mediated cytotoxicity [GO:0042267]; negative regulation of myoblast differentiation [GO:0045662]; negative regulation of neutrophil apoptotic process [GO:0033030]; negative regulation of peptidyl-tyrosine phosphorylation [GO:0050732]; neutrophil activation [GO:0042119]; NIK/NF-kappaB signaling [GO:0038061]; positive regulation of activated T cell proliferation [GO:0042104]; positive regulation of apoptotic process [GO:0043065]; positive regulation of blood vessel remodeling [GO:2000504]; positive regulation of chemokine production [GO:0032722]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of collagen biosynthetic process [GO:0032967]; positive regulation of gene expression [GO:0010628]; positive regulation of granulocyte macrophage colony-stimulating factor production [GO:0032725]; positive regulation of hepatocyte proliferation [GO:2000347]; positive regulation of inflammatory response [GO:0050729]; positive regulation of interleukin-13 production [GO:0032736]; positive regulation of interleukin-17 production [GO:0032740]; positive regulation of interleukin-8 production [GO:0032757]; positive regulation of macrophage derived foam cell differentiation [GO:0010744]; positive regulation of male germ cell proliferation [GO:2000256]; positive regulation of natural killer cell proliferation [GO:0032819]; positive regulation of neuron apoptotic process [GO:0043525]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of NIK/NF-kappaB signaling [GO:1901224]; positive regulation of NK T cell proliferation [GO:0051142]; positive regulation of phosphatidylinositol 3-kinase signaling [GO:0014068]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of smooth muscle cell migration [GO:0014911]; positive regulation of smooth muscle cell proliferation [GO:0048661]; positive regulation of superoxide anion generation [GO:0032930]; positive regulation of T-helper 1 cell cytokine production [GO:2000556]; positive regulation of T-helper 2 cell differentiation [GO:0045630]; positive regulation of transcription by RNA polymerase II [GO:0045944]; positive regulation of tumor necrosis factor production [GO:0032760]; positive regulation of type II interferon production [GO:0032729]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531]; positive regulation of vascular permeability [GO:0043117]; regulation of cell adhesion [GO:0030155]; response to hypoxia [GO:0001666]; sleep [GO:0030431]; T-helper 1 type immune response [GO:0042088]; triglyceride homeostasis [GO:0070328] |
11710997_synergistic effects of interleukin-18 and interleukin-12 in augmenting cytotoxic T lymphocyte responses and IFN-gamma production in aging 11751983_the role of IL-18 in the antibacterial host response during pneumococcal pneumonia 11854182_plays an essential role in ifn-gamma induction by spleen cells stimulated with listeriolysin O. 11877284_NK cell activity and IFN-gamma production induced by IL-18 were reduced by the absence of tyk2. The synergistic effect of IL-12 and IL-18 for the production of IFN-gamma was also abrogated by the absence of ty 11905033_critically involved in regulation of IgE production in vivo, providing a potential therapeutic target for allergic disorders. 11907086_Endogenous, caspase-1-dependent IL-18 significantly contributes to the initiation of contact hypersensitivity. 11971017_The role of IL-18 in blood-stage immunity against malaria Plasmodium yoelii and Plasmodium berghei 11978780_Lack of evidence for a role for IL-18 in cytotoxic T lymphocyte activation and IFN-gamma production during experimental acute graft-versus-host disease 12006705_produced by primary pancreatic mouse islets and NIT-1 beta cells and participates in the progression towards destructive insulitis. 12052461_The combination of IL-18 and IL-12 synergistically inhibits osteoclastic bone-resorbing activity in vitro. 12077261_IL-18 plays an important role in Th1 response and host defense against Mycobacterium tuberculosis infection, although less pronounced in comparison to that of IL-12p40. 12093672_IL18 induced acute biphasic reduction in the levels of circulating leukocytes is mediated by NO and IFN-gamma, with NO being involved in the first phase of reduction and IFN-gamma being involved in both phases 12151598_contributes to the spontaneous development of atopic dermatitis-like inflammatory skin lesion independently of IgE/stat6 under specific pathogen-free conditions 12165484_IFN-alphabeta-dependent pathway of IFN-gamma induction in mice, based on a synergism of IFN-alphabeta and IL-18 12172382_Results show increased levels of interleukin-18 in mouse brain homogenates following traumatic brain injury, and that administration of the IL-18 inhibitor, IL-18-binding protein, enhances neurological recovery. 12176045_Heat shock response inhibits IL-18 expression through the JNK pathway in murine peritoneal macrophages 12193723_Regulation of serum IL-18 expression is critical to the outcome of innate immune responses to lipopolysaccharide. 12193725_IL-18 has a direct effect on mucosal mast cell and Th2 responses in the small intestine and plays a significant role in the development of intestinal immunity against Trichinella spiralis infection. 12207348_Butyrate and acetate increased intracellular IL-18 content in a time- and dose-dependent fashion in mice. 12370378_IL-18 treatment restores LPS-induced IFN-gamma production following burn injury, inhibits IL-10 production in burned mice, and increases survival of mice with postburn bacterial peritonitis. 12388820_vaccinia virus C12L protein inhibits mouse IL-18 12393844_caspase-1 activity and IL-18 were significantly increased in the kidney in neutrophil-depleted mice with ischemic acute renal failure 12420106_IL-18 is synthesized in keratinocytes mainly in inactive precursor form, and its expression is upregulated as basal keratinocytes differentiate in epidermis. 12433285_Bacterial infection of osteoblasts induces genetic transcription of this protein but not protein synthesis. 12442336_endogenous IFN-gamma and IL-18 are potent, independent, negative regulators of the development of type 2 immunity to ubiquitous environmental antigens 12559970_Results suggest that the destabilization of interferon-gamma (IFN-gamma) mRNA induced by transforming growth factor beta leads to the inhibition of antiviral activity and IFN-gamma production in interleukin-18-stimulated LNK5E6 cells. 12574340_Costimulation of the activating Ly49D murine NK cell receptor with IL-18 is capable of over-riding the inhibitory Ly49G2 receptor blockade for cytokine (e.g., IFN-gamma)production both in vitro and in vivo. 12654774_Endogenous IL-18 activity facilitates inflammatory responses in the lung during Pseudomonas aeruginosa pneumonia, concurrently impairing bacterial clearance 12670447_IL-18 is not inhibited by IL-10 12707353_Increased IL-18 levels in the heart during acute coxsackie B3 virus-induced myocarditis is directly associated with increased inflammation. 12707362_This report demonstrates selective induction of TNF-alpha, CCR4, and CCR7 by IL-18 in T cell lines specific for mouse myelin oligodendrocyte glycoprotein (MOG35-55 peptide). 12734344_Using the NF-kappa B pathway of cell activation, IL-18 induces transcription of the T cell neurokinin 1 receptor. 12742650_IL-18 is constitutively expressed in murine brain tissue and upregulated early in the innate immune response during pneumococcal meningitis; moreover, its elimination suppresses the local inflammatory response and improves the outcome in this model. 12771567_IL-18 does not contribute to acute ischemic brain damage. 12787061_IL-18 induced intracellular expression of IL-1 alpha and proIL-1 beta, and release of IL-6 from mixed glia; deletion of the IL-18 gene did not affect IL-1 beta expression; IL-18 may contribute to inflammatory responses in the brain 12788303_This study compared changes in IL-1beta and IL-18 mRNA expression in the liver of rats in response to peripheral injection of LPS. Unlike IL-18, IL-18BP mRNA was up-regulated dramatically. 12847274_Direct evidence reveals that IL-18 activates neutrophils via TNF-alpha induction, which drives the production of leukotriene B4, which in turn leads to neutrophil accumulation and subsequent local inflammation. 12850812_Gm-Csf induced high levels of IL-18. 12874202_IL-18 significantly contributes to the eliciation of murine contact hypersensitivity by augmenting the recruitment of interferon-gamma producing alpha beta T cells to the inflammatory focus. 12919851_the requirement for endogenous pro-inflammatory cytokines for a protective immune response against Trypanosoma cruzi is satisfied by the expression of IL-12, while IL-18 is dispensable 12925208_overexpression of interleukin 18 in the skin aggravates allergic and nonallergic cutaneous inflammation, which is accompanied by high expression of T helper 1 and T helper 2 cytokines and chemokines in the skin. 12940437_results suggest that IL-18 might function differently from IL-12 at some pathological conditions in the development of colitis 14500466_endogenous IL-18 plays an important role in the early antibacterial host response during E. coli-induced peritonitis 14599848_it was demonstrated that IL-18 is essential for the development of streptozotocin-induced diabetes in mice, and could be an important target in prevention and treatment of autoimmune diabetes. 14634096_IL-18 plays a promoting role as an enhancer of Th1-type immune responses in diabetes development early in the spontaneous disease process, which may contribute to elucidating the pathogenesis of type 1 diabetes. 14646616_IL-18 production is regulated by vitamin C through the MAPK signalling pathway 14688108_IL-18 responses have an important role in host defenses against Group B Streptococcus and rIL-18 may be useful in alternative strategies to treat neonatal GBS disease 14970180_IL-18 stimulates memory Th1 cells to induce severe airway inflammation and AHR in the naive host. 14999688_IL-18 is required for facilitating neutrophil-dependent hepatic ischemia/reperfusion injury through suppressing anti-inflammatory cytokine expression. 15122309_Development of the retinal vasculature was compared in IL-18 knockout (KO) and wild-type (WT) mice at several different time points. IL-18 KO mice showed angiectasis and vascular leakage at P7, especially in the mid-peripheral retina. 15178407_induction of expression is induced by a beta-2-adrenergic receptor and NF-kappa B dependent mechanism 15188378_some involvement of IL-18 in the pathogenesis of group B streptococcal-induced arthritis. 15280194_Administering IL-18 significantly enhanced allospecific cytotoxic function and expansion of CD8(+) cells. 15350655_IL-18 may participate in microglial activation and dopaminergic neurodegeneration. 15356153_In respiratory syncytial virus (RSV) infection, IL-18 functions to limit lung inflammation and Th2 effector molecules, thus reducing airway mucus production, a hallmark of RSV bronchiolitis. 15385528_limited role for IL-18 in the host defense against P. aeruginosa 15476451_IL-18 is involved in the regulation of the central part of the hypothalamus pituitary thyroid axis during illness. 15543578_transcription of IL-18 is regulated by p300 15574430_IL-18 induces cardiomyocyte hypertrophy via PI3K-dependent signaling, mediates ANF gene transcription, and has a role in inflammatory heart diseases including heart failure 15604417_the proatherogenic effect of IL-18 can occur in the absence of T cells and IFN-gamma secreted by macrophages, NK cells, and vascular cells is sufficient for the disease progression 15681795_A study using gene-deficient mice revealed that IL-18 is essential for inducing antigen-specific regulatory T cells and oral tolerance. 15770015_Influence of IL-18 in pneumococcal pneumonia differs with the mouse strain genetic background. 15778395_functional polarization of Ag-specific CD4(+) Th2 cells entering the CNS during the accelerating phase of experimental autoimmune encephalomyelitis is flexible and dependent on IL-18, which is overturned by IL-18 binding protein 15821032_IL-18 causes blunted beta-adrenergic responsiveness to isoproterenol. IL-18 reduces responsiveness of myofilaments to Ca2+. Induction of myocardial hypertrophy by IL-18 indicates role in myocardial remodeling. 15860230_corticotropin releasing hormone is an important mediator that regulates IL-18 expression in the brain during stress 15860756_IL-18 activates key signaling pathways in vascular smooth muscle cells (VSMCs) including transcription factors NF-kB and AP-1. 15888563_These studies demonstrate that in severe experimental crescentic glomerulonephritis, IL-12p40 is the key Th1-defining cytokine chain, whereas IL-18 has local proinflammatory roles. 16052682_Gut ischemia/reperfusion induces the increase of IL-18 expression, which may make IL-18 act as an important proinflammatory cytokine and contribute to gut ischemia/reperfusion-induced lung inflammation. 16129701_IL-18 with IL-2 protects against Strongyloides venezuelensis infection by activating mucosal mast cell-dependent type 2 innate immunity. 16210597_Preventing induction of type 2 T helper (Th2) cellular immune responses is critically dependent on altered T cell responsiveness to IL-18 and IL-12. 16212914_IFN-gamma productions under the normal condition were independent upon IL-12 and IL-18, its expressions in various tissues were different, and optimal IFN-gamma is necessary for the normal growth and development of mammals 16293799_increased expression of Mif and IL-18 cytokines is associated with intima-media thickening in SJL mice, likely by stimulating inflammation and proliferation 16331327_Weidentified a novel short form of IL-18 in mouse, named IL-18s. Its cDNA contains a 57 bp in-frame deletion. IL-18s could enhance the ability of IL-18 to increase IFN-lambda production by approximately 40% in mouse splenocytes. 16339568_IL-18 activated the PI3K/Akt and MEK/ERK1/2 pathways. Inhibition of either of these pathways attenuated MCP-1 production in macrophages, and inhibition of both signaling pathways resulted in the complete inhibition of MCP-1 production. 16493047_Through its capacity to affect DNA repair, IL-18 can restore an immune response that is suppressed by ultraviolet radiation. 16517748_vitamin D modulates cutaneous inflammatory reactions, at least in part, by increasing the IL-1Ra to IL-1alpha ratio and suppressing IL-18 synthesis in keratinocytes 16568498_IL-18 is specifically required for the in vivo up-regulation of IL-13 production and associated host protection. 16732281_findings indicate a new role of IL-18 in the homeostasis of energy intake and insulin sensitivity 16785519_IL-18 profoundly improves antigen-specific T cell immunity throughout lymphoid and nonlymphoid tissue by bridging the innate and adaptive arms of the immune system through interferon-gamma and the CD134 costimulatory pathway. 16861683_Important role in innate immunity against Salmonella enterica serovar Typhimurium infection. 16875741_IL-18 was found to induce apoptosis of adherent cells in TNF-alpha-mediated osteoclastogenesis mouse bone marrow cells 16959946_IL-18 is involved in the induction of gastric lesions by stress restrasint through augmentation of histidine decarboylase activity and production of histamine in the stomach. 17008311_analysis of a mechanism mediated by cryopyrin and ASC that links dsRNA and viral infection to caspase-1 activation resulting in IL-1beta and IL-18 production 17014914_IL-18 enhances the ability of melanoma cells to migrate via the generation of ROI and the MAPK pathway. 17075045_role for islet-derived IL-18 activity during inflammation-mediated islet injury 17188500_caspase-1 and the two caspase-1-processed cytokines, interleukin (IL)-1beta and IL-18, are involved in trauma-induced neuronal cell death in the developing brain 17194566_results suggest that IL-18 plays a significant role in antigen-induced inflammatory hypernociception via the production of IFN-gamma, ET-1 and PGE2 17234681_We therefore concluded that IL-18 regulates pathogenic retinal NV by promoting its regression rather than inhibiting its development. This suggests some useful, new approaches to treating retinopathy in humans. 17237446_IL-18 and the IL-18 pathway are activated in cigarette smoke-exposed mice and man; IL-18R signaling plays a critical role in the pathogenesis of CS-induced inflammation and emphysema 17250807_Plays a key role in compensatory cardiac growth in response to pressure overload. 17292338_We conclude that CpG promotes high-level IL-18 synthesis by an IFNgamma-dependent and IFNgamma-sufficient mechanism in vivo that is optimally triggered by LPS 17316614_Role of IL-18 in excitotoxic injury in IL-18 deficient mice may be overcompensated by increased IL-12 secretion. 17330820_IL-18 modulates interleukin-4 and interferon-gamma production in ligand-activated natural killer T (NKT) cells time-dependently by regulating/inducing suppressor of cytokine signaling 3 (SOCS3) expression. 17393315_IL-18-mediated cytokine production is a key element in the pathogenesis of dextran sulfate sodium-induced colitis. 17400610_IL-18 acts directly on activated CD8+ T cells through IL-18Rs and promotes their survival to expand the population 17400729_Overproduction of IL-18 in lungs induces emphysema in mice 17475863_CD4 T cells in Salmonella-infected mice acquire the ability to respond to innate stimuli, by a mechanism that is partially dependent on IL-18. 17517876_The data indicate that the enhanced production of IL-18 in melioidosis is an essential part of a protective immune response to this severe infection. 17522234_Serum Il-18 was induced with Proprionibacterium acnes and lipopolysaccharides in phagocytic macrophage-inactivated mice. 17578927_endogenous IL-18 signaling modulates food intake, metabolism, and adiposity 17664259_These data identify IL-18 as part of an adequate innate immune response during Nontypeable Haemophilus influenzae pneumonia. 17709537_IL-18, but not IL-12, plays a key role in the rapid activation of NK cells and therefore in control of early HSV-1 replication in the lung. 17766435_Coadministration of lipopolysaccharides and ovalbumin into Th1 mice induced bronchial hyperreactivity and airway inflammation via endogenous Il-18. 17850464_LPS strongly induced the expression of interleukin-18 in astrocytes from midbrain; estrogen and progesterone together diminished LPS-induced IL-18 expression in the midbrain completely. 17878377_caspase-activating recruiting domain and caspase-1 are critical for IFN-gamma-mediated control of Anaplasma phagocytophilum infection 17947451_IL-18 plays a crucial role in experimental necrotizing enterocolitis pathogenesis. 17979156_IL-15 induced IL-18 production. Neutrophil migration depended on a signal cascade involving IL-18. 17984289_IL-18 is a key proximal mediator of Langerhans-cell migration & contact hypersensitivity, acting upstream of IL-1 beta & TNF-alpha. It may play a central role in regulation of cutaneous immune responses. 18003981_These findings demonstrate a feed-forward pathway in HIF-1alpha-mediated tumor progression, in which the induction of IL-18 by hypoxia or inflammatory cells augments the expression of both HIF-1alpha and tumor cell metastasis. 18022572_host-derived IL-18 is a major factor for IFN-gamma production that may have a protective effect on CD4(+)-mediated aGVHD after bone marrow transplants 18086672_IL-18 selectively suppresses adiponectin expression via ERK1/2-dependent NFATc4 activation and suggest that the inverse relationship observed between IL-18 and adiponectin in various chronic pathological conditions is causally related 18165223_IL-18 is regulated by parathyroid hormone and is required for its bone anabolic actions 18247160_IL-18 upregulated phosphorylated Akt and downregulated phosphorylated P38 MAPK; the elevated serum IL-18 levels in obesity and diabetes might be a compensatory response to insulin resistance. 18250441_Has a positive regulatory role in recovery from kainate-induced ataxia. 18299270_IL-18, ICE and IL-18 R are overexpressed in testicular tissue from sexually immature as compared to mature mice 18299271_IL-18 and IL-18BP may have roles in testicular development, and also in the regulation of testicular functions under physiological conditions 18374640_These results suggest that high levels of serum IL-18 promote the overexpression of endogenous IL-18 in articular chondrocytes, resulting in cartilage loss through suppression of aggrecan synthesis. 18386818_study found that IL18 was downregulated in the UVR-induced melanomas relative to the spontaneous melanomas 18387310_IL-18 did not suppress or enhance the pulmonary allergic immune response in a murine experimental model of asthma 18511039_a role for IL-18 in writhing response in mice. 18514661_IL-18 appears one of the stimuli that enhance sLOX-1 release in acute coronary syndrome, and ADAM10 may be involved in this process 18515422_Short-term leptin reconstitution in the absence of major weight loss did not protect ob/ob mice, whereas leptin deficiency in the absence of obesity resulted in a significant reduction in the severity of the pancreatitis. 18545704_results indicate that a positive regulatory loop involving IFN-gamma and IL-18 signaling contributes to the accelerated memory CD8(+) T cell proliferation during a recall response to antigen presented by DCs 18573894_The authors found that mice deficient in IL-18 were partially resistant to primary infection with Listeria monocytogenes. 18575774_These results suggest that IL-18 suppresses Fas-mediated apoptosis of hepatocytes by up-regulation of NFkappaB and XIAP, following inhibition of caspase-3 activity. 18599478_The diastolic dysfunction observed in alveolar hypoxia might be caused by increased circulating IL-18, thereby inducing an increase in PP2A and a reduction in phosphorylation of phospholamban at Ser16. 18641298_priming by IL-18 leads to an improved translation of IFN-gamma mRNA 18692153_IL-18/MyD88-dependent signalling is involved in blood stage Plasmodium yoelii control and induction of pro-inflammatory cytokines. 18705842_results demonstrate that lipopolysaccharide increased the capacity of Leydig cells to produce the IL-18 family molecules; IL-18, in the testis, might be involved in the regulation of physiological and infection/inflammatory processes 18753296_Protection against ischemic acute kidney injury in IL-18 binding protein Tg mice (functional neutralization of IL-18) associated with less macrophage infiltration and less production of CXCL1 in the kidney. 18786924_identifies proIL-18 as a biologically important substrate for meprin beta and implicates meprins in the modulation of inflammation 18815244_IL-18, derived primarily from cells of bone marrow origin, contributes to the renal damage observed during ischemia-reperfusion injury 18818761_IL-18 has an effect on the engraftment and function of CD8 T cells and regulatory T cells 18940934_induces a marked gene expression profile change and increased Ccl1 (I-309) production in mouse mucosal mast cell homologs 19039317_Adipose and hepatic IL-18 protein expressions are increased in obese mice. However, in contrast to liver, the adipose IL-18 protein level appears to be upregulated through a post-transcriptional mechanism probably involving caspase-1. 19046143_LPS increases the expression levels of the IL-18 family in mouse testis and spleen, but the time of expression differs between the two organs. 19054060_maturation of DC with anti-CD40 plus IL-12 and IL-18 potently stimulates the generation of an anti-tumor immune response 19129255_IL-18 from adoptive transfer of macrophages is not sufficient to cause ischemic acute kidney injury. 19141531_Data suggest that IL-18 may be a crucial determinant for the vulnerability of the rectum to psychosocial stress. 19164288_IL-18 plays a critical role in I/R injury and thus represents a promising therapeutic target. 19201478_in IL-18 deficiency, cholesterol in HCD synergize mechanistically with homocysteine to accelerate atherosclerosis via the alternative IL-23/Th17 pathway, demonstrating a new role for Th17 in atherosclerosis 19265174_the levels of IL-1beta and IL-18 mRNA and protein, pulmonary inflammation, and lung fibrosis were examined in models of bleomycin-induced lung injury 19394645_IL-18 plays an important role in the development of pulmonary injury after hepatic ischemia/reperfusion by up-regulating proinflammatory mediators. 19451398_IL-4 and IL-13 are produced in response to IL-18 or IL-33 stimulation. IL-18- or IL-33-mediated Th2 cytokine production is dependent on MyD88 and p38alpha signaling proteins 19535628_Coinjection of IL-12 plus IL-10 cDNAs results in increased survival of tumor- bearing mice that correlates with diminished serum levels of inflammatory cytokines tumor necrosis factor (TNF)-alpha and interferon (IFN)-gamma. 19536084_IL-18 is a significant mediator of obstruction-induced renal fibrosis and epithelial- mesenchymal transition independent of downstream TGF-beta1 or TNF-alpha production. 19638429_endogenous IL-18 might facilitate stomach cancer cell immune escape by suppressing CD70 and increasing metastatic ability by upregulating CD44 and VEGF 19657092_A deficiency in both IL-12p40 and IL-18 in C57BL mice results in increased susceptibility to infectious ectromelia. 19727119_IL-23, similar to IL-12 and IL-18, can reduce UVR-induced DNA damage and thereby prevent immunosuppression 19758166_Studies show that IL-18 has a multifaceted role in autoimmune lupus. 19802695_Combined treatment with IL-12/TNF-alpha was superior to all the triple combination of IL-12/IL-18/TNF-alpha in ablation of the primary breast tumor, eradicating distant disease and enhancing survival. 19853308_promotes expansion of gammadelta T cells with potent anti-tumor cytotoxicity 19950174_Data show that IL-18 plays a decisive role in protective immunity against tuberculosis. 19950176_Data show that dihydrotestosterone significantly inhibited the T-cell stimulatory capacity of DC of male IL-4KO mice, and this finding was associated with dramatically reduced IL-18 mRNA expression. 20056085_N-acetylcysteine antagonizes the expression of TNF-alpha and IL-6 induced by IL-18 in vascular smooth muscle cells. 20089076_TGF-beta signalling may play an important role in the pathogenesis of IL-18 plus IL-2-induced ILD in mice. 20111957_The present results suggest that IL-18 may inhibit TNF-alpha-mediated osteoclastogenesis in vivo via a T cell-independent mechanism. 20130211_Data show that SIGN-R1(-/-) mice have reduced susceptibility to experimental colitis, with a reduction in the disease severity, and levels of IL-1beta, TNF-alpha, IL-6 and IL-18. 20213736_IL-15 is not functioning as a universal NK-cell priming signal and that IL-18 contributes to the NK-cell response in visceral leishmaniasis 20351186_IL-18 acts on natural killer (NK) cells because myeloid differentiation factor MyD88 expression is required in responding NK cells, but not in infected bone marrow dendritic cells. 20497957_The results suggest the importance of IL-18 for the development of asthmatic inflammation associated with airway exposure to bacterial proteins. 20536664_expression increased in restraint stress 20538003_Data show for the first time that EMMPRIN stimulates the activation of NF-kappaB, AP-1, CREB, and ATF-2 in cardiomyocytes, and induces IL-18 expression via Rac1-dependent PI3K/Akt/IKK/NF-kappaB and MKK7/JNK/AP-1 signaling. 20565717_IL-18 may serve a hierarchic position for initiating joint inflammatory responses. 20580925_We identified low activity and high anxiety-like behaviors in Il18r1(-/-) mice, whereas Il18(-/-) mice displayed little anxiety-like behavior. 20624890_MyD88 signaling has a protective role in the development of azoxymethane /dextran sodium sulfate colitis-associated cancer (CAC). The susceptibility of MyD88 -/- mice to CAC is in part a result of their inability to signal through the IL-18 receptor. 20805006_The results of this study suggested that IL-18-dependent vasopressinergic and oxytocinergic circuitry in the amygdala may regulate depressive-like behaviors in mice. 20855874_IL-18 production downstream of the Nlrp3 inflammasome is critically involved in protection against colorectal tumorigenesis 20980973_Circulating IL-18 levels were elevated in Stat4-deficient compared to Stat4-intact mice, suggesting that this interleukin might contribute to the progression of lupus nephritis independent of Stat4. 21042583_epididymal white adipose tissue and aortic expression of IL-18 and IL-18 plasma levels, a pro-atherosclerotic cytokine, was markedly reduced in ApoE(-/-) PGC-1alpha(-/-) mice 21106643_IL-18 modulates liver inflammation by the recruitment of inflammatory cells. 21106820_Interleukin-18 and caspase-1 mediate mouse central nervous system inflammation in a mouse model of multiple sclerosis. 21248035_The authors suggest that MyD88 functions to support T-cell-specific IL-18 receptor signaling, which in turn is essential for the formation of adaptive immunity to fowlpox virus -encoded chicken ovalbumin. 21257923_NK1.1(+) gammadeltaT cells function as inflammatory mediators in the early phase of IL-18/IL-2-induced interstitial lung disease 21263463_These results reveal novel requirements for IL-18 in innate immune cell homeostasis and activation. 21414755_IL-18 is required for self-reactive T cell expansion in NOD mice. 21471445_findings demonstrate that caspase-1-processed cytokines IL-1beta and IL-18 not only promote autoimmunity by stimulating innate IL-17 production by T cells but also reveal redundancy in the functions of IL-1beta and IL-18 21557898_MiR-146a mimics suppressed IL-18 expression (P |
ENSG00000150782 |
IL18 |
413.552748 |
2.0343140767 |
1.024542 |
0.13635248 |
56.122828 |
0.00000000000006808203405103928646888435636765361546370988604648388786699797492474317550659179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000015617854558214313377452895307331860177361276820207081073021981865167617797851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
558.184668301077 |
52.8339131787581 |
274.384705243602 |
19.9501755290924 |
| ENSMUSG00000039232 |
74732 |
Stx11 |
protein_coding |
A0A1W2P6E3 |
Human_homologues FUNCTION: SNARE that acts to regulate protein transport between late endosomes and the trans-Golgi network. |
Proteomics identification;Reference proteome |
|
|
|
|
19169743_Stx11 were highly expressed at the end of fetal development but downregulated at 4 and 11 weeks in brain, pancreas, and heart. 21388490_Results suggest that by regulating the availability of Vti1b, Stx11 regulates trafficking steps between late endosomes, lysosomes and the cell surface in macrophages. 23042080_required for NK and CD8+ T cell cytotoxicity and neutrophil degranulation 23160464_Distinct severity of HLH in both human and murine mutants with complete loss of cytotoxic effector PRF1, RAB27A, and STX11. 23190531_Blockade of inhibitory receptors on T cells in Stx11-deficient mice converted nonfatal disease course into fatal HLH, identifying T-cell exhaustion as an important factor for determination of disease severity in hemophagocytic lymphohistiocytosis. 30811271_Findings suggest that stx11 regulates the stimulus-dependent transport of TLR4 to the plasma membrane by cooperating with SNAP-23 in macrophages. Our results clarify the regulatory mechanisms underlying intracellular transport of TLR4 and have implications for microbial pathogenesis and immune responses. |
ENSG00000135604 |
STX11 |
292.193341 |
2.2818028965 |
1.190174 |
0.27504777 |
17.929678 |
0.00002292187419678763462302199804820901363200391642749309539794921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001581993601112230483744791609268531829002313315868377685546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
389.014751028091 |
74.615206825825 |
170.48569428724 |
24.5938627367979 |
| ENSMUSG00000039270 |
230316 |
Megf9 |
protein_coding |
Q8BH27 |
|
Disulfide bond;Glycoprotein;Laminin EGF-like domain;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:230316; |
basement membrane [GO:0005604]; membrane [GO:0016020]; animal organ morphogenesis [GO:0009887]; cell migration [GO:0016477]; substrate adhesion-dependent cell spreading [GO:0034446]; tissue development [GO:0009888] |
16981854_MEGF9 represents a novel putative receptor, expressed in neuronal and non-neuronal tissues, that is regulated during development and could function as a guidance or signalling molecule. |
ENSG00000106780 |
MEGF9 |
704.274485 |
0.4953952397 |
-1.013348 |
0.16935217 |
34.862603 |
0.00000000353811939348806818490863306794682457967482491767441388219594955444335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000462200982861151409783619743373850319301254785386845469474792480468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
467.148802361257 |
54.9627419741936 |
942.982017002342 |
81.9383564213221 |
| ENSMUSG00000039304 |
22035 |
Tnfsf10 |
protein_coding |
P50592 |
FUNCTION: Cytokine that binds to TNFRSF10A/TRAILR1, TNFRSF10B/TRAILR2, TNFRSF10C/TRAILR3, TNFRSF10D/TRAILR4 and possibly also to TNFRSF11B/OPG. Induces apoptosis. Its activity may be modulated by binding to the decoy receptors TNFRSF10C/TRAILR3, TNFRSF10D/TRAILR4 and TNFRSF11B/OPG that cannot induce apoptosis. {ECO:0000250|UniProtKB:P50591}. |
Apoptosis;Cell membrane;Cytokine;Glycoprotein;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Secreted;Signal-anchor;Transmembrane;Transmembrane helix;Zinc |
|
|
mmu:22035; |
extracellular space [GO:0005615]; plasma membrane [GO:0005886]; cytokine activity [GO:0005125]; identical protein binding [GO:0042802]; TRAIL binding [GO:0045569]; tumor necrosis factor receptor binding [GO:0005164]; tumor necrosis factor receptor superfamily binding [GO:0032813]; zinc ion binding [GO:0008270]; apoptotic process [GO:0006915]; immune response [GO:0006955]; male gonad development [GO:0008584]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of extrinsic apoptotic signaling pathway [GO:2001238]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of release of cytochrome c from mitochondria [GO:0090200]; response to insulin [GO:0032868] |
11801676_The use of TRAIL gene-targeted mice, devoid of TRAIL expression and TRAIL-mediated cytotoxicity, demonstrates the key role of TRAIL in suppressing tumor initiation and metastasis. 11805143_TRAIL plays a critical role in the NK cell-mediated and IFN-gamma-dependent tumor surveillance 12088418_TRAIL acts as the effector molecule that eliminates subcutaneously developing TRAIL-sensitive tumors. 12209599_role of TRAIL in mouse mastocytoma 12209637_TRAIL/Apo2L is important in controlling tumors in vivo, but is not an essential regulator of osteoprotegerin-mediated biology, under normal physiological conditions. 12391182_Ocular tissue displays functional TRAIL as determined by in vitro killing of TRAIL-sensitive tumor cell lines. 12517799_Mediates apoptosis is an important endogenous mechanism for resistance to liver metastases in murine renal cancer. 12842625_GDVII virus induces apoptosis in murine astrocytes through tumor necrosis factor (TNF)-receptor and TNF-related apoptosis-inducing ligand 12882912_TRAIL is an important immune regulator of the development of type 1 diabetes. 12900523_Results suggest that TRAIL is not a critical factor for thymocyte negative selection. 12941766_TRAIL deficiency accelerates autoimmune diabetes and enhances autoimmune responses. 14560009_These results support a role for Egr-1, -2, and -3 in nonlymphoid expression of Fas ligand, TRAIL, and tumor necrosis factor. 14645705_sensitization to TRAIL requires p53 14688317_Immature dendritic cells (DC) are rapidly eliminated by natural killer cells in vivo via a pathway dependent on TRAIL, highlighting the potential importance of innate effector mechanisms that limit DC-based vaccination. 14702109_plays a crucial role in hepatic cell death and hepatic inflammation 14702523_TRAIL appears to play an important role in corneal-allograft rejection and may be used to prolong the survival of allografts. 15128592_presence and function of TRAIL and MK, a death-inducing ligand and its receptor, in mammalian preimplantation embryos. 15163747_matrix protein of lyssaviruses plays a major role in the early induction of TRAIL-mediated apoptosis 15213096_TRAIL blocks osteoclastic differentiation induced by RANKL plus M-CSF 15494516_Trail plays a crucial role in Listeria-induced apoptosis and listeriosis. 15536146_we demonstrate that TRAIL is the dominant cytotoxic effector molecule expressed by NK cells in fetal mice; a phenotypically immature TRAIL+ NK cell subpopulation is retained in the liver of adult mice, and its retention is dependent on IFN-gamma 15744305_Regulation of Trail expression can therefore account for the role of CD4+ T cells in the generation of CD8+ T cell memory and represents a novel mechanism for controlling adaptive immune responses 15919918_plays an important role in the immune response to influenza virus infection 16174101_suppresses autoimmune damage in relapsing-remitting, and non-remitting models of experimental autoimmune encephalomyelitis 16636669_signaling of TRAIL-DR5 complexes requires ceramide-enriched membrane platforms 16772303_neurotrophic factors have effects on tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis of murine adrenal chromaffin cells 16818756_TRAIL deficiency might have an initial protective role but is not sufficient for the in vivo maintenance of long-term memory CD8 T cells in the absence of CD4 T help. 17202353_prevention of experimental autoimmune encephalomyelitis by treatment with embryonic stem cell-derived dendritic cells(ES-DC)-TRAIL/myelin oligodendrocyte glycoprotein (MOG) is mediated by MOG-reactive CD4(+)CD25(+) Treg propagated by ES-DC-TRAIL/MOG 17237443_led to decreased c-FLIP expression and allowed all MCL samples to undergo TRAIL-mediated apoptosis 17571163_ability of TRAIL to modify inflammatory responses and to reduce neuronal cell death in meningitis suggests that it may be used as a novel antiinflammatory agent in invasive infections 17620297_Tnfsf10 inhibits osteoclastic differentiation by counteracting RANKL-dependent p27Kip1 accumulation in pre-osteoclast precursors. 17703232_Variants of human TRAIL (hTRAIL) and human CD95L (hCD95L), encompassing the TNF homology domain (THD), interact with the corresponding receptors and stimulate CD95 and TRAILR2 signaling after cross-linking. 17762882_These findings suggest that the TRAIL/DR5 pathway is one of the mechanisms used by T regulatory cells to regulate immune responses both in vitro and in vivo. 17947462_nuclear factor kappaB pathway inhibition blocks myeloma cell growth and induces apoptosis in strong synergy with TRAIL 18070069_Results from this study suggest that osteoblasts containing intracellular S. aureus induce TRAIL expression in uninfected osteoblasts present in infected cultures. 18079967_adherent TRAIL-R-expressing skin carcinoma cells were TRAIL resistant in vitro but were sensitized to TRAIL upon detachment by inactivation of the ERK signaling pathway 18089829_Local treatment with mTRAIL and CpG oligodeoxyribonucleotide can augment tumor antigen cross-presentation resulting in T-cell proliferation, enhanced cytotoxic T-lymphocyte activity, and increased animal survival in kidney cancer. 18220275_pivotal role in cholestatic liver injury mediated by NK 1.1-positive NK/NKT cells 18354179_CD4(+) T cell help for the CD8(+) T cell response is not contingent on the silencing of TRAIL expression and prevention of TRAIL-mediated apoptosis 18362335_IL-15 codelivered with vaccines can overcome CD4+ helper T cell deficiency for induction of functionally efficient CD8+ T cells and maintenance of CD8+ cytotoxic T lymphocytes (CTLs) 18362888_rpS6, especially in its unphosphorylated form, is a selective mediator of TRAIL-induced apoptosis 18802095_Important component of immunity to influenza infections; TRAIL-mediated apoptosis is a mechanism used by influenza-specific CD8-positive T lymphocytes to eliminate influenza-infected cells and drive recovery from influenza. 18830416_CD4+cd25+ Tregs control the TRAIL-dependent cytotoxicity of tumor-infiltrating DCs in rodent models of colon cancer. 18988676_In situ infection of adenovirus expressing TRAIL or indoleamine 2, 3-dioxygenase, but not green fluorescent protein, significantly suppressed thyroiditis scores. 19064696_Data demonstrate a key role for exudate macrophages in the induction of alveolar leakage and mortality in IV pneumonia, and suggest that epithelial cell apoptosis induced by TRAIL-expressing macrophages is a major underlying mechanism. 19361506_Deficiency of GMDS leads to escape from natural killer cell-mediated tumor surveillance through modulation of TRAIL signaling. 19481529_Results identify PACS-2 as an essential TRAIL effector, and show that Akt cooperates with 14-3-3 to regulate the homeostatic and apoptotic properties of PACS-2 that mediate TRAIL action. 19633419_Data show that G-CSF treatment increased the amount of Fas-L, TRAIL and Bax proteins, whereas decreased the expression of procaspase 3 and Bcl-2. 19635861_essential mechanism of NEMO-mediated protection of the liver by preventing NK cell tissue damage via TRAIL/DR5 signaling 19893042_TRAIL plays a role in the control of retinal neovascularization 19901518_Data show that alpha-GalCer/AdmIL-2 treatment expanded the absolute numbers of lung and liver NK, NKT and T-cells as well as the TNF-related apoptosis-inducing ligand (TRAIL) expression of these cells. 19955659_The cytolytic molecules Fas ligand and TRAIL are required for murine thymic graft-versus-host disease. 20062539_results indicate that cells that survive TRAIL treatment may do so by activation of a TAK1-NF-kappaB pathway that drives expression of cFlipL, and suggest that TAK1 may be a good target for overcoming TRAIL resistance 20107429_TRAIL- and GITRL-activated pathways synergise in the development of SCI-related inflammatory damage. 20150555_TRAIL induction involves FGF-2, Sp1-phosphorylation and NFkappaB and that TRAIL promotes VSMC proliferation and neointima formation after arterial injury. 20185587_mice defective in both tumor necrosis factor-related apoptosis-inducing ligand and Fas ligand reveal new roles for TRAIL in lymphocyte homeostasis and autoimmune lymphoproliferative syndromes 20190248_sTRAIL is inversely associated with the mortality risk in chronic kidney disease patients. 20213318_Data show that in Abelson cells, the inhibition of both NF-kappaB and FoxO3a activity is required for suppression of TRAIL transcription and maintenance of the transformed state. 20483771_apoptotic cells generated during sepsis induced a CD8+ regulatory T cell that suppressed delayed-type hypersensitivity by TRAIL production 20711434_extrinsic and intrinsic pathways, via Trail/Dr5 and Puma respectively, could be engaged in distinct subpopulations of spermatogonia 20921531_TRAIL suppresses autoimmunity by two mechanisms: the inhibition of Th1 cells and the promotion of Tregs 21169546_CD8-positive regulatory T cells generated after anterior chamber injection of antigen via the retro-orbital plexus enforce systemic tolerance in a TRAIL-dependent manner to inhibit inflammation in the eye. 21209944_resveratrol can enhance the apoptosis-inducing potential of TRAIL by activating FKHRL1 and its target genes 21225479_Results suggest that excessive iodine could induce TRAIL and DR5 abnormal expression in thyroid. TRAIL band with DR5 to promote follicular cells apoptosis thus mediate thyroid destruction in EAT. 21324463_study concludes that TRAIL attenuates plaque size at early stages of atherosclerosis 21426505_TRAIL and osteoprotegerin are expressed in atherosclerotic plaques in a calcification-prone mouse model 21463519_Data show that the number of tumors observed in TRAIL transgenic animals was significantly lower than in the control animals, and the lesions observed were mostly benign. 21470210_is not required for the development of peripheral lymphopenia or granuloma necrosis during infection with Mycobacterium avium 21562052_TRAIL-deficient mice had an enhanced inflammatory response with increased neutrophil numbers and reduced neutrophil apoptosis 21627969_Data suggest that deficiency of TRADD sensitizes cells to TRAIL-induced apoptosis, and that enhanced cell death in TRADD(-/-) MEFs is associated with defective NF-kappaB activation. 21654829_TRAIL strongly synergizes with APAP in inducing cell death in hepatocyte-like cells lines and primary hepatocyte. 21832159_NK cells inhibit dendritic cell cross-priming, but not direct priming, in a TRAIL/DR5-dependent manner. 21846721_a novel mechanism for the tumoricidal activity of PEDF, which involves tumor cell killing via PPARgamma-mediated TRAIL induction in macrophages. 21940678_TRAIL regulates the magnitude of the influenza A virus (IAV)-specific CD8 T cell response during a clinically significant IAV infection to decrease the chance for infection-induced immunopathology. 21965021_It was shown for the first time that TRAIL deficiency promotes numerous features of diabetes typical of human disease, and are associated with reduced insulin and pancreatic inflammation/apoptosis. 22116828_the tumoricidal activity of CnB-activated peritoneal macrophages is partially dependent on TRAIL. 22131069_Epirubicin enhances the antiarthritic effect of rAAV2/5-TRAIL and that combination treatment might be an important therapeutic alternative, with practical significance for rheumatoid arthritis. 22616837_findings shed light on the possible anti-adipogenic and anti-inflammatory effects of TRAIL and open new therapeutic possibilities against obesity, systemic inflammation and T2DM 22719861_data demonstrate that TRAIL induces osteoclastic differentiation via a TRAF-6 dependent signaling pathway 22740407_CD8+ T cells use TRAIL to restrict West Nile virus pathogenesis by controlling infection in neurons. 23071253_Data indicate that TNF-related apoptosis-inducing ligand knockout mice (TRAIL -/-) showed significantly decreased lung bacterial clearance and survival in response to Streptococcus pneumoniae. 23071256_Data indicate the importance of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in pulmonary arterial hypertension (PAH) pathogenesis and suggest its potential as a therapeutic target to direct future translational therapies 23108456_ASMase is essential for TRAIL-induced lysosomal trafficking, membrane fusion and formation of membrane raft redox signaling. 23347256_TRAIL-DR5 interaction promoted malignant behaviors of B16F10 cells. 23408429_oncogene metadherin modulates the apoptotic pathway based on the tumor necrosis factor superfamily member TRAIL (Tumor Necrosis Factor-related Apoptosis-inducing Ligand) in breast cancer 23430714_TRAIL expression by osteoclast-like cells is increased in the presence of RANKL and after scraping; DcR2 expression peaks at 24 hours, and and decreases at 5 days; DR5 expression peaks at 5 days 23497038_The extracellular domain of Fn14 fused to the extracellular domain of the TRAIL ligand is designed to modulate the immune system as an anti-inflammatory agent. 24040204_TRAIL-deficiency accelerates vascular calcification in atherosclerosis via modulation of RANKL. 24045576_Tumor necrosis factor-related apoptosis-inducing ligand translates neonatal respiratory infection into chronic lung disease. 24097299_These results suggest that TRAIL inhibits VEGF-induced angiogenesis by increasing caspase-8 activity and subsequently decreasing non-apoptotic signaling functions of procaspase-8, without inducing caspase-3 activation and endothelial cell cytotoxicity. 24509861_TRAIL signaling and cell death in the immature central nervous system after hypoxia-ischemia and inflammation 24667560_TRAIL-deficiency in ApoE(-/-) mice exacerbates nephropathy and insulin resistance 24804996_expression of TRAIL on natural killer cells is protective in a murine model of hepatic ischemia-reperfusion injury through modulation of natural killer cell cytotoxicity and NK cell differentiation 24887152_TRAIL induces apoptosis in tissue-infiltrating neutrophils thereby protecting organs from sepsis-induced injury. 25031778_These results demonstrated that TRAIL plays a deleterious role in acute kidney injury pathogenesis induced by scald burns 25356750_These findings indicate that acute fasting enhances TRAIL-mediated liver natural killer cell activity against neoplastic cells through upregulation of heat shock protein 70. 25607248_PARP1 acts as a prominent upstream regulator of high mobility group box 1-mediated autophagy and maintains a homeostatic balance between apoptosis and autophagy, which provides new insight into the mechanism of TNFSF10 resistance 25827060_Data suggest that Socs3 (suppressor of cytokine signaling 3) plays critical negative role in regulation of Trail expression in endoplasmic reticulum stress in macrophages via Jun N-terminal kinase/AP-1 transcription factor signaling. 26555706_this study shows pathogenic role for TRAIL in chronic obstructive pulmonary disease 26572549_TRAIL can promote angiogenesis following hindlimb ischemia in vivo via NOX4/eNOS/nitric oxide signaling. 26783750_Study reports that loss of the Opg gene results in deterioration of abdominal aortic aneurysms (AAA), possibly through involvement of TRAIL in smooth muscle actin (SMA)-positive cells and myofibroblasts. 26935795_Membrane-proximal TRAIL species are incapable of inducing short circuit apoptosis signaling: Implications for drug development and basic cytokine biology.( 27142480_In this study, the authors identified by gene expression profiling that microgravity induces high levels of TRAIL expression in murine preosteoclast cells in the absence of RANKL stimulation compared to ground based cultures. 27617958_pCLE results indicated that endomicroscopy could effectively quantify injected MSCs that homed to subcutaneous xenograft tumor sites in vivo and correlated well with the therapeutic effects of the TRAIL gene 27742702_Tnfsf10 expression is increased in eosinophilic esophagitis. Tnfsf10(-/-) mice were largely protected from esophageal fibrosis and eosinophilic inflammation. 27836899_the aim of this study is to elucidate the role of TRAIL during rhinovirus (RV) infection in vivo. 28055006_downregulation of cIAPs in PSC cholangiocytes may contribute to the development of the disease. Our results also indicate that inhibition of TRAIL signaling pathways may be beneficial in the treatment of PSC 28070143_Diabetes significantly increased OPG and the OPG/TRAIL ratio expression in the aorta, while dyslipidemia was the major determinant of the changes observed in the heart, where it significantly increased OPG and reduced TRAIL expression, thus increasing cardiac OPG/TRAIL ratio. 28392572_results provide a novel apoptosis-independent, immune regulatory role for TRAIL in suppressing inflammatory arthritis and shed light on the development of effective new therapies for autoimmune inflammatory diseases. 28461078_Isolated highly-pure population of DcR2-positive renal tubular epithelial cells was isolated by MACS, which was confirmed to comprise of active senescent RTECs based on the cell cycle phase. 28507343_TRAIL protects against insulin resistance, NAFLD and vascular inflammation. Increasing TRAIL levels may be an attractive therapeutic strategy, to reduce features of diabetes, as well as liver and vascular injury, so commonly observed in individuals with NAFLD. 29088306_Data suggest that the CXCL9-CXCR3 axis plays a pivotal role in the liver-specific distribution of TRAIL+ NK cells in mice. 29403497_TRAIL/TRAIL-R interaction regulates CD4(+) T cell activation in autoimmune inflammation and directly suppresses T cell activation via inhibiting TCR signaling, suggesting that TRAIL-R serves as a novel immune checkpoint in T cell responses. 29661825_In the absence of NKp46, ILC1s fail to express normal levels of TRAIL on the surface. 30165101_These findings demonstrate for the first time that TRAIL protects against several forms of endothelial dysfunction involving its ability to control endothelial cell ROS generation 30732588_MID1 levels were inversely associated while PP2A activity levels correlated with DLco. Tnfsf10(-/-) and mice treated with the PP2A activator AAL(s) were largely protected against bleomycin-induced reductions in lung function and fibrotic changes. TRAIL signalling through MID1 deactivates PP2A and promotes fibrosis with corresponding lung function decline. 31076664_Our results demonstrate that TRAIL effectively inhibited colonic T-cell activation and suppressed autoimmune colitis, suggesting a potential therapeutic application of TRAIL in human inflammatory bowel disease. 31355333_Our study indicates that TRAIL liposomes, alone or in combination with existing clinically approved therapies, may neutralize distant metastasis of a broad range of tumor types systemically. 31742873_Non-apoptotic TRAIL function modulates NK cell activity during viral infection. 31894448_PDGFRbeta-targeted TRAIL specifically induces apoptosis of activated hepatic stellate cells and ameliorates liver fibrosis. 31995154_Our findings offer novel insight into the regulatory function of acinar cell-derived TRAIL in limiting inflammatory damage and could be implicated in the development of potential therapeutic strategies for dry eye disease. 32410328_Mesenchymal stem cells engineered by modified polyethylenimine polymer for targeted cancer gene therapy, in vitro and in vivo. 32485960_M1 Macrophages Promote TRAIL Expression in Adipose Tissue-Derived Stem Cells, Which Suppresses Colitis-Associated Colon Cancer by Increasing Apoptosis of CD133(+) Cancer Stem Cells and Decreasing M2 Macrophage Population. 32645396_Muscle-derived TRAIL negatively regulates myogenic differentiation. 33408417_Gut-licensed IFNgamma(+) NK cells drive LAMP1(+)TRAIL(+) anti-inflammatory astrocytes. 33483333_Regulatory T Cell-Derived TRAIL Is Not Required for Peripheral Tolerance. 33875613_TRAIL induces proliferation in rodent pancreatic beta cells via AKT activation. 33932348_Influence of TRAIL Deficiency on Th17 Cells and Colonic Microbiota in Experimental Colitis Mouse Model. 34843580_Virus-infection in cochlear supporting cells induces audiosensory receptor hair cell death by TRAIL-induced necroptosis. 34933177_Cardioprotection of M2 macrophages-derived exosomal microRNA-24-3p/Tnfsf10 axis against myocardial injury after sepsis. 35726345_Acute portal hypertension using portal vein ligation abrogates TRAIL expression of liver-resident NK cells. 35840556_TRAIL protects the immature lung from hyperoxic injury. |
ENSG00000121858 |
TNFSF10 |
38.564224 |
10.1262251294 |
3.340025 |
0.52031757 |
41.487828 |
0.00000000011860714301188278871257119835341733299349797903232683893293142318725585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000018702111825641806764442311714676664413659068486595060676336288452148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
65.3630087347271 |
24.0879874631114 |
6.45482476437304 |
2.03419826733209 |
| ENSMUSG00000039323 |
16008 |
Igfbp2 |
protein_coding |
P47877 |
FUNCTION: Inhibits IGF-mediated growth and developmental rates (By similarity). IGF-binding proteins prolong the half-life of the IGFs and have been shown to either inhibit or stimulate the growth promoting effects of the IGFs on cell culture. They alter the interaction of IGFs with their cell surface receptors. {ECO:0000250}. |
Disulfide bond;Glycoprotein;Growth factor binding;Growth regulation;Reference proteome;Secreted;Signal |
|
The protein encoded by this gene is one of several similar proteins that bind insulin-like growth factors I and II (Igf-I and Igf-II). The encoded protein can be secreted into the bloodstream, where it binds Igf-I and Igf-II with high affinity, or it can remain intracellular, interacting with many different ligands. Two transcript variants, one encoding a secreted isoform and the other encoding a nonsecreted isoform, have been found for this gene. [provided by RefSeq, Sep 2015]. |
mmu:16008; |
apical plasma membrane [GO:0016324]; cytoplasmic vesicle [GO:0031410]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; insulin-like growth factor binding [GO:0005520]; insulin-like growth factor I binding [GO:0031994]; insulin-like growth factor II binding [GO:0031995]; aging [GO:0007568]; cellular response to hormone stimulus [GO:0032870]; female pregnancy [GO:0007565]; positive regulation of activated T cell proliferation [GO:0042104]; regulation of insulin-like growth factor receptor signaling pathway [GO:0043567]; response to estradiol [GO:0032355]; response to estrogen [GO:0043627]; response to glucocorticoid [GO:0051384]; response to lithium ion [GO:0010226]; response to mechanical stimulus [GO:0009612]; response to nutrient [GO:0007584]; response to retinoic acid [GO:0032526]; response to xenobiotic stimulus [GO:0009410]; signal transduction [GO:0007165] |
12409314_Data show that IGFBP-2 potently separates hypertrophic and hyperplastic effects of GH/IGF-I excess on adrenocortical cells. 12914536_During cell differentiation, secretion is controlled by hormones in mouse mammary epithelial cells. 14710363_effect of IGFBP-2 overexpression on the activity of various antioxidative enzymes in two malignant cell lines 15044367_menin regulates the expression of the endogenous IGFBP-2 gene at least in part through the promoter of IGFBP-2. 15474485_intact insulin-like growth factor binding protein-2 and a distinct carboxyl-terminal IGFBP-2 fragment localize to the cell nucleus 16556765_IGFBP2 expression is responsible for increased insulin receptor substrate-1 phosphorylation in mesangial cells from mice susceptible to glomerulosclerosis 18276763_IGFBP-2 may regulate bone turnover in both an IGF-I-dependent and -independent manner. 18392786_IGF axis as key mechanisms underlying nephropathic responses of mesangial cells to Ang II and high glucose. 19095771_Results suggest that the growth-promoting role of IGFBP-2 in prostate cancer is inhibited by its intracellular interaction with PAPA-1. 19142966_IGFBP-2 overexpression reduces the appearance of dysplastic aberrant crypt foci and inhibits growth of adenomas in chemically induced colorectal carcinogenesis 20074524_Overexpression of IGFBP2 by an adenovirus reversed diabetes in insulin-resistant and diet-induced obese mice, as well as insulin-deficient streptozotocin-treated mice. 20501873_IGFBP-2 significantly inhibits postnatal skeletal myofiber growth by decreasing myogenic proliferation and protein accretion and enhances glycolytic muscle metabolism 20730703_The present study was aimed at determining IGFBP2 mRNA levels in adipose tissue in conditions of insulin resistance such as aging and obesity. 20858761_We propose that Igfbp2 mediates epidermal progenitor cell expansion and therefore represents an epidermal progenitor cell-specific target of Hedgehog signaling that promotes basal cell carcinoma development 21372140_the HBD peptide of IGFBP-2 has anabolic activity by activating IGF-I/Akt and beta-catenin signaling pathways 21821709_IGF binding protein 2 supports the survival and cycling of hematopoietic stem cells. 22006816_IGFBP-2 is an important regulator of osteoclastogenesis. 22202803_Data from transgenic mice suggest that hepatic leptin signaling (i.e., direct action of leptin on hepatocytes) is not required for leptin up-regulation of plasma levels of IGFBP-2; intact vagus nerve is also not required. 22410287_findings suggest that insulin upregulates IGFBP-2 expression through a PI3K/mTOR/C/EBP-alpha pathway in white adipocytes 22869525_IGFBP-2 bound receptor protein tyrosine phosphatase beta, which led to its dimerization and inactivation. Analysis of aortas obtained from IGFBP-2(-/-) mice showed that receptor protein tyrosine phosphatase beta was activated. 22973541_we have identified a single polymorphic locus that affects skin and lung tumorigenesis and identify Igfbp5 and Igfbp2 as candidate modifier genes of lung tumorigenesis. 23813480_Igfbp2-5 are expressed in distinct and complementary patterns during cochlear development. 23981772_These findings demonstrate that the HBD2 domain of IGFBP-2 is the primary region that accounts for its ability to inhibit adipogenesis and that a peptide encompassing this region has activity that is comparable with native IGFBP-2. 24191913_IGFBP2 is a critical cell-autonomous factor that promotes the survival and migration of acute leukemia cells. 24359611_IGFBP-2 plays an IGF-I-dependent and -independent role in the brain's acute (neuroprotection) and chronic (tissue remodeling) response to hypoxic-ischemic injury. 24424049_physiological levels of IGFBP2 are neither sufficient to mimic nor required for the physiological action of leptin 24839202_The results suggest that stimulation of differentiation is an important mechanism by which IGFBP-2 regulates the acquisition of normal bone mass in mice. 25695641_PPARalpha controls IGF-1 signalling through the up-regulation of hepatic Igfbp-2 transcription during fasting. 25787077_protein(s) that associated with RPTPbeta in response to IGF-I and IGFBP-2 in vascular smooth muscle cells 26105006_Impaired glucose clearance in female IGFBP-2 transgenic mice is dependent on the presence of the RGD motif and that translocation of GLUT4 in the muscle may be regulated by IGFBP-2 via RGD-dependent mechanisms. 26230658_estrogen deficiency has a profound effect on body and bone composition in the absence of IGFBP-2 and may be related to changes in fibroblast growth factor 21. 26507795_that the lacking the Arg-Gly-Asp-domain has a major influence on the pleiotropic effects of IGFBP-2 and suggest that somatic growth and time of sexual maturity or somatic growth and life expectancy are less closely related than thought previously 26556533_Data (including data from studies in knockout mice) suggest, as osteoblasts differentiate, early induction of AMPK in response to IGF-I/IGFBP-2 is followed by suppression of AMPK as required for osteogenesis; activation of AMPK stimulates autophagy. 26773517_Peptides that disrupted p62/PKCzeta or p62/IRS-1 inhibited IGF-I/IGFBP-2 stimulated PKCzeta activation, vimentin phosphorylation, PTEN tyrosine phosphorylation, AKT activation, and osteoblast differentiation. 27125842_IGFBP2 not only is a driver of glioma progression and a prognostic factor but is also required for tumour maintenance and thus represents a viable therapeutic target in the treatment of glioma. 27126637_In human whole blood cells, methylation of IGFBP2 at the homologous CpG site was increased in obese men with impaired glucose tolerance. In conclusion, our data show that increased methylation of hepatic Igfbp2 during infancy predicts the development of fatty liver later in life and is linked to deterioration of glucose metabolism. 27544574_AAV6-mediated IGFBP-2 overexpression in the tibialis anterior (TA) muscles of 4-week-old C57BL/10 and mdx mice reduced the mass of injected muscle after 8weeks, inducing a slower muscle phenotype in C57BL/10 but not mdx mice. Analysis of inflammatory and fibrotic gene expression revealed no changes between control and IGFBP-2 injected muscles in dystrophic (mdx) mice. 30677223_An integrative systems approach identifies IGFBP-2, Mfap4, and Spp1 as novel candidates in Marfan syndrome-related pathophysiology. 31560714_Study in syngeneic mouse glioblastoma (GBM) model, human GBM samples, and cancer-immune cell co-culture revealed that IGFBP2-promoted immunosuppression associated with its induction of the mesenchymal feature of GBM cells and the inhibitory phosphorylated FcgammaRIIB of GBM exposed immune cells. Inhibition of IGFBP2 relieved immunosuppression by increasing CD8+ T and CD19+ B cells and decreasing CD163+ M2 macrophages. 32228319_Downregulated microRNA-130b-5p prevents lipid accumulation and insulin resistance in a murine model of nonalcoholic fatty liver disease. 32532003_Physiological Disturbance in Fatty Liver Energy Metabolism Converges on IGFBP2 Abundance and Regulation in Mice and Men. 32993096_Sex-Specific Control of Muscle Mass: Elevated IGFBP Proteolysis and Reductions of IGF-1 Levels Are Associated with Substantial Loss of Carcass Weight in Male DU6PxIGFBP-2 Transgenic Mice. 33763996_Serum insulin-like growth factor binding protein 2 levels as biomarker for pancreatic ductal adenocarcinoma-associated malnutrition and muscle wasting. 35226161_Insulin-like growth factor binding protein 2 null mice (Igfbp2-/-) are protected against trabecular bone loss after vertical sleeve gastrectomy. |
ENSG00000115457 |
IGFBP2 |
2204.978453 |
2.1562014180 |
1.108492 |
0.07079437 |
242.453586 |
0.00000000000000000000000000000000000000000000000000000114742786749986770366612939765884543958177851038891280467336894020036337641055848816356676135628024515757727004027556341595778051399497380821723702126746502472087740898132324218750000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000002330349503701064818922480662973321575087775400557273593679280582475977439437880295769734832262798606441718068901385110931839468627942840717182093612791504710912704467773437500000000000000000000000000000000000 |
Yes |
No |
3040.60289924263 |
149.265071776134 |
1410.16645003632 |
52.9587281755065 |
| ENSMUSG00000039457 |
19041 |
Ppl |
protein_coding |
G5E898 |
Human_homologues FUNCTION: Component of the cornified envelope of keratinocytes. May link the cornified envelope to desmosomes and intermediate filaments. May act as a localization signal in PKB/AKT-mediated signaling. {ECO:0000269|PubMed:9412476}. |
Coiled coil;Phosphoprotein;Proteomics identification;Reference proteome;Repeat;SH3 domain |
|
|
mmu:19041; |
cytoskeleton [GO:0005856]; intermediate filament cytoskeleton organization [GO:0045104]; response to mechanical stimulus [GO:0009612] |
15226441_Data suggest that the primary role of periplakin may not relate to the physiology of the cornified cell envelope in epidermal keratinocytes but may reside in the challenges, which normal laboratory mice do not encounter. 15590649_Periplakin interferes with G protein activation by the melanin-concentrating hormone receptor-1 by binding to the proximal segment of the receptor C-terminal tail 18166659_Thus, combined loss of the cornified envelope proteins not only impairs the epidermal barrier, but also changes the composition of T cell subpopulations in the skin. 29515024_Study in a mouse model of lung fibrosis induced by bleomycin found that Ppl expression was downregulated in the whole lung and in alveolar epithelial cells (AECs) after bleomycin-induced injury. Deletion of Ppl reduced lung fibrosis after bleomycin injury by promoting antiinflammatory cytokine expression and profoundly altering cellular signaling in AECs, including STAT3 activation, and TGF-beta/smad pathway activation. |
ENSG00000118898 |
PPL |
16.242360 |
0.1942112441 |
-2.364301 |
0.71601422 |
11.087962 |
0.00086889922441726625407243389531686261761933565139770507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0040735835599642351151361552297203161288052797317504882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.1404610835406 |
2.65726005064001 |
26.4684009771482 |
8.52741448839195 |
| ENSMUSG00000039462 |
12813 |
Col10a1 |
protein_coding |
Q05306 |
FUNCTION: Type X collagen is a product of hypertrophic chondrocytes and has been localized to presumptive mineralization zones of hyaline cartilage. |
Calcium;Collagen;Extracellular matrix;Hydroxylation;Metal-binding;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:12813; |
cell cortex [GO:0005938]; collagen trimer [GO:0005581]; collagen type X trimer [GO:0005599]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; receptor complex [GO:0043235]; extracellular matrix structural constituent [GO:0005201]; metal ion binding [GO:0046872]; cartilage development [GO:0051216]; endochondral ossification [GO:0001958]; extracellular matrix organization [GO:0030198] |
12057907_demonstration of sleketal defects and altered marrow and hematopoiesis in all Tg mice with dominant interference mutations for collagen X 12952936_These data suggest that alpha1(X) collagen (Col10a1) is a direct transcriptional target of Runx2 during chondrogenesis. 15464363_The 4.6 kb promoter is able to drive specific expression of Col10a1 in hypertrophic cartilage. 16806867_the triple-helical region of collagen X contains a specific DDR2 binding site that is capable of receptor activation 16905550_TSG is an important modulator of BMP-regulated induced collagen II and X expression and cartilage development and chondrocyte differentiation 17403716_nonsense/frame-shift mutations can elicit a gain-of-function effect, affecting chondrocyte differentiation in the growth plate 18059015_Indian hedgehog, expressed in pre-hypertrophic cells and collagen type X, expressed in hypertrophic chondrocytes, were both significantly inhibited after incubation with isoproterenol 18304492_These data suggest that the 3' UTR participates in collagen X last-exon mRNA decay and that overall 3' UTR configuration, rather than specific linear-sequence motifs, may be important in specifying decay of Col10a1 mRNA containing nonsense mutations 19478656_Results show that in Smad3 gene knock-out mice the protein positive staining of type II collagen, aggrecan, and TGF-beta1 in the disc decreased, while that of type X collagen increased. 19834559_Mice expressing the collagen X mutation had shortened limbs and an expanded hypertrophic zone. 20048163_we have uncovered a novel molecular mechanism linking GADD45beta via the MTK1/MKK3/6/p38 axis to C/EBPbeta-TAD4 activation of Col10a1 transcription in terminally differentiating chondrocytes 20209091_endochondral ossification-specific collagen X contributes to the establishment of a hematopoietic niche at the chondro-osseous junction 20403959_cartilaginous regeneration is due to the expression of chondrogenic factors, such as Sox 9 and type II and X collagens 21328521_Studies indicate that Col10a1 is highly expressed in hypertrophic chondrocytes and commonly used as a gene marker for this cell population. 21887706_Runx2 directly interacts with murine Col10a1 cis-enhancer. 22072985_SOX9 directly represses Col10a1 expression in immature/proliferating chondrocytes of the growth plate 22692895_Annexins and collagen X may rather fulfill functions in growth plate cartilage not directly linked to the mineralization process. 23441228_Downregulation of carbonic anhydrase IX promotes Col10a1 expression in chondrocytes. 24269824_these results show that increased Col10a1 expression in Phex-deficient osteoblasts is not a major cause of the XLHR phenotype, which was an important issue to address based on the previous findings 25028519_Data indicate Indian hedgehog (Ihh) induces collagen type X COL10A1 expression and promotes its transcription through Runx2/Smad1 interactions on a specific promoter region. 25321476_These data support a Cox-2-containing, Runx2-centered Col10a1 regulatory mechanism, during chondrocyte hypertrophic differentiation. 26362410_Col10 expression was seen in the hypertrophic zone at and below the tidemark of the mandibular condyle cartilage. 26901593_In a model of instability-induced osteoarthritis, Col X was overexpressed in articular chondrocytes and osteophyte-forming cells in LOX-1(+)/(+) mice and was significantly downregulated in articular chondrocytes and osteophyte-forming cells in LOX-1(-)/(-) mice. 27121205_Results indicate that Cox-2 promotes Col10a1 expression and chondrocyte hypertrophy in vitro. 29522813_Metaphyseal chondrodysplasia Schmid type disease severity in mice is increased by ATF6alpha ablation and decreased by ATF6beta ablation triggered by mutations in collagen X. 30010889_treatment of the mice with carbamazepine (CBZ), a drug which stimulates intracellular proteolysis and alleviates ER stress, effectively reduced the disease severity in this model of metaphyseal chondrodysplasia type Schmid caused by a premature stop codon in the Col10a1 gene 30154451_The mechanistic link between COL10A1 and the TGF-beta1-SOX9 axis. 32533783_A quantitative serum biomarker of circulating collagen X effectively correlates with endochondral fracture healing. 34520774_COL10A1 is a novel factor in the development of choroidal neovascularization. 36246404_Cancer-Associated Fibroblasts Hinder Lung Squamous Cell Carcinoma Oxidative Stress-Induced Apoptosis via METTL3 Mediated m(6)A Methylation of COL10A1. |
|
|
1713.269532 |
0.0005810692 |
-10.749002 |
0.59755765 |
2357.617668 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.00251373623359 |
0.853352170461426 |
3446.25691970215 |
190.529054446272 |
| ENSMUSG00000039518 |
386463 |
Cdsn |
protein_coding |
Q7TPC1 |
FUNCTION: Important for the epidermal barrier integrity. {ECO:0000250}. |
Reference proteome;Secreted;Signal |
|
|
mmu:386463; |
desmosome [GO:0030057]; extracellular region [GO:0005576]; protein homodimerization activity [GO:0042803]; amyloid fibril formation [GO:1990000]; cell-cell adhesion [GO:0098609]; corneocyte desquamation [GO:0003336]; negative regulation of cornification [GO:1905716]; skin morphogenesis [GO:0043589] |
18436651_These results underscore the essential roles of CDSN in hair physiology and suggest functional relevance of CDSN gene polymorphisms to psoriasis susceptibility. |
ENSG00000204539 |
CDSN |
1828.915885 |
3.2906857317 |
1.718388 |
0.14152521 |
140.406034 |
0.00000000000000000000000000000002169823030830356043667347039114483153700110498478812002676424533856359266556870597970375913465801431811996735632419586181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000018567833935734820696573696145776606771756489852653897733885083574556380936658125414828734989214353845454752445220947265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2776.32962045149 |
272.78886063884 |
843.693335325468 |
62.7977508525724 |
| ENSMUSG00000039519 |
13123 |
Cyp7b1 |
protein_coding |
Q60991 |
FUNCTION: A cytochrome P450 monooxygenase involved in the metabolism of endogenous oxysterols and steroid hormones, including neurosteroids (PubMed:9295351, PubMed:9144166, PubMed:10748047, PubMed:11067870). Mechanistically, uses molecular oxygen inserting one oxygen atom into a substrate, and reducing the second into a water molecule, with two electrons provided by NADPH via cytochrome P450 reductase (CPR; NADPH-ferrihemoprotein reductase). Catalyzes the hydroxylation of carbon hydrogen bonds of steroids with a preference for 7-alpha position. Usually metabolizes steroids carrying a hydroxy group at position 3, functioning as a 3-hydroxy steroid 7-alpha hydroxylase (PubMed:9295351, PubMed:9144166, PubMed:10748047, PubMed:11067870). Hydroxylates oxysterols, including 25-hydroxycholesterol and (25R)-cholest-5-ene-3beta,26-diol toward 7-alpha hydroxy derivatives, which may be transported to the liver and converted to bile acids (PubMed:9295351, PubMed:9144166, PubMed:10748047, PubMed:11067870). Via its product 7-alpha,25-dihydroxycholesterol, a ligand for the chemotactic G protein-coupled receptor GPR183/EBI2, regulates B cell migration in germinal centers of lymphoid organs, thus guiding efficient maturation of plasma B cells and overall antigen-specific humoral immune response (PubMed:22999953). 7-alpha hydroxylates neurosteroids, including 3beta-hydroxyandrost-5-en-17-one (dehydroepiandrosterone) and pregnenolone, both involved in hippocampus-associated memory and learning (PubMed:9144166). Metabolizes androstanoids toward 6- or 7-alpha hydroxy derivatives (By similarity). {ECO:0000250|UniProtKB:O75881, ECO:0000269|PubMed:10748047, ECO:0000269|PubMed:11067870, ECO:0000269|PubMed:22999953, ECO:0000269|PubMed:9144166, ECO:0000269|PubMed:9295351}. |
Cholesterol metabolism;Endoplasmic reticulum;Heme;Iron;Lipid biosynthesis;Lipid metabolism;Membrane;Metal-binding;Microsome;Monooxygenase;Oxidoreductase;Reference proteome;Steroid biosynthesis;Steroid metabolism;Sterol metabolism;Transmembrane;Transmembrane helix |
PATHWAY: Lipid metabolism; bile acid biosynthesis. {ECO:0000269|PubMed:10748047, ECO:0000269|PubMed:11067870, ECO:0000269|PubMed:9144166, ECO:0000269|PubMed:9295351}.; PATHWAY: Steroid hormone biosynthesis. {ECO:0000269|PubMed:9144166}. |
|
mmu:13123; |
endoplasmic reticulum membrane [GO:0005789]; intracellular membrane-bounded organelle [GO:0043231]; 24-hydroxycholesterol 7alpha-hydroxylase activity [GO:0033782]; 25-hydroxycholesterol 7alpha-hydroxylase activity [GO:0033783]; 27-hydroxycholesterol 7-alpha-monooxygenase activity [GO:0047092]; heme binding [GO:0020037]; iron ion binding [GO:0005506]; oxysterol 7-alpha-hydroxylase activity [GO:0008396]; steroid hydroxylase activity [GO:0008395]; B cell chemotaxis [GO:0035754]; bile acid biosynthetic process [GO:0006699]; cholesterol homeostasis [GO:0042632]; cholesterol metabolic process [GO:0008203]; circadian rhythm [GO:0007623]; digestion [GO:0007586]; epithelial cell proliferation [GO:0050673]; intracellular estrogen receptor signaling pathway [GO:0030520]; memory [GO:0007613]; negative regulation of intracellular estrogen receptor signaling pathway [GO:0033147]; positive regulation of epithelial cell proliferation [GO:0050679]; prostate gland epithelium morphogenesis [GO:0060740] |
12370428_Because CYP7B1 represents the major pathway for inactivating 3betaAdiol in the prostate, we suggest that ERbeta, 3betaAdiol, and CYP7B1 are the components of a pathway that regulates growth of the rodent ventral prostate. 18055760_Identification of Cyp7b1 as a novel RORalpha (NR1F1) target gene and a functional cross-talk between RORalpha and liver X receptor (NR1H3). 19258494_Data show that ten genes (Fsp27, Aqp4, Cd36, Ly6d, Scd1, Hsd3b5, Syt1, Cyp7b1, and Tff3) showed significant associations among their expressions and the degree of hepatic steatosis. 19732851_CYP7B1-mediated catalysis may play a role for control of the cellular levels of androgens, not only of estrogens. 20501668_Increasing 27-hydroxycholesterol concentrations by genetic means leads to decrease bone mineral density. 22999953_Findings indicate that CYP7B1 and HSD3B7, as well as CH25H, have essential roles in controlling oxysterol production in lymphoid tissues. 30710743_This study provides evidence for CYP7B1 as a key regulator of three vital intracellular regulatory oxysterol levels. 30728500_results indicate that osteoarthritis is a disease associated with metabolic disorders and suggest that targeting the CH25H-CYP7B1-RORalpha axis of cholesterol metabolism may provide a therapeutic avenue for treating osteoarthritis. 34685636_Oxysterol 7-alpha Hydroxylase (CYP7B1) Attenuates Metabolic-Associated Fatty Liver Disease in Mice at Thermoneutrality. |
ENSG00000172817 |
CYP7B1 |
69.695895 |
2.2166139176 |
1.148358 |
0.34446126 |
10.838682 |
0.00099401460959210236924266101965486086555756628513336181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0045956586635728553791313544252261635847389698028564453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
95.4780508059073 |
23.0137775258984 |
43.0738298845564 |
7.99775154332493 |
| ENSMUSG00000039601 |
53901 |
Rcan2 |
protein_coding |
Q9JHG2 |
FUNCTION: Inhibits calcineurin-dependent transcriptional responses by binding to the catalytic domain of calcineurin A. Could play a role during central nervous system development. |
Phosphoprotein;Reference proteome |
|
|
mmu:53901; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; calcium-dependent protein serine/threonine phosphatase regulator activity [GO:0008597]; nucleic acid binding [GO:0003676]; calcineurin-NFAT signaling cascade [GO:0033173]; calcium-mediated signaling [GO:0019722]; locomotion involved in locomotory behavior [GO:0031987]; Notch signaling pathway [GO:0007219]; response to oxidative stress [GO:0006979]; short-term memory [GO:0007614] |
19717730_Genetic inhibition of calcineurin in Zaki-4 transgenic mice induces diastolic dysfunction in mice with chronic pressure overload. 21298050_an Rcan2-dependent mechanism which regulates food intake and promotes weight gain through a leptin-independent pathway 22593270_Rcan2 regulates osteoblast function and its expression in bone is regulated by thyroid status in vivo 27604858_Rcan2 and Rps6kb1 mutations both affect growth and body weight of mice, though likely through different mechanisms 27917924_RCANs play critical roles in bone homeostasis by regulating both osteoclastogenesis and osteoblastogenesis, and they serve as inhibitors for calcineurin-NFATc1 signaling both in vivo and in vitro. 28624805_our results indicate that Rcan2 and estradiol might regulate body weight through different mechanisms in female mice. Rcan2 increases food intake and promotes weight gain, while estradiol increases energy expenditure possibly by regulating physical activity. 34146582_A novel transcriptional complex on the VE-cadherin promoter regulated the downregulation of VE-cadherin in the Down Syndrome Candidate Region 1 isoform 1L-mediated angiogenesis. 34348766_NUAK2 and RCan2 participate in the p53 mutant pro-tumorigenic network. 36267816_The Overexpression of miR-377 Aggravates Sepsis-Induced Myocardial Hypertrophy by Binding to Rcan2 and Mediating CaN Activity. |
ENSG00000172348 |
RCAN2 |
49.477081 |
0.2530921255 |
-1.982265 |
0.47955509 |
16.598458 |
0.00004618856196660050454725482405038405886443797498941421508789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002988717824448848493432140394077123346505686640739440917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.5944096887197 |
6.63383278124439 |
77.420068488309 |
18.3723364663099 |
| ENSMUSG00000039621 |
277360 |
Prex1 |
protein_coding |
Q69ZK0 |
FUNCTION: Functions as a RAC guanine nucleotide exchange factor (GEF), which activates the Rac proteins by exchanging bound GDP for free GTP. Its activity is synergistically activated by phosphatidylinositol 3,4,5-trisphosphate and the beta gamma subunits of heterotrimeric G protein. May function downstream of heterotrimeric G proteins in neutrophils (By similarity). {ECO:0000250, ECO:0000269|PubMed:16243036}. |
Alternative splicing;Cell membrane;Cytoplasm;Guanine-nucleotide releasing factor;Membrane;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:277360; |
cytosol [GO:0005829]; dendritic shaft [GO:0043198]; growth cone [GO:0030426]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; guanyl-nucleotide exchange factor activity [GO:0005085]; phospholipid binding [GO:0005543]; G protein-coupled receptor signaling pathway [GO:0007186]; intracellular signal transduction [GO:0035556]; neutrophil chemotaxis [GO:0030593]; positive regulation of cell adhesion [GO:0045785]; positive regulation of cell migration [GO:0030335]; positive regulation of GTPase activity [GO:0043547]; positive regulation of substrate adhesion-dependent cell spreading [GO:1900026]; reactive oxygen species metabolic process [GO:0072593]; regulation of actin filament polymerization [GO:0030833]; regulation of dendrite development [GO:0050773]; T cell differentiation [GO:0030217] |
16243035_P-Rex1 is an important regulator of neutrophil function by mediating a subset of Rac-dependent neutrophil responses. 16243036_P-Rex1 is a primary Rac2 guanine nucleotide exchange factor in mouse neutrophils. 18502673_P-Rex1 is one of the major GEFs in macrophage regulating Rac1 activation and chemotaxis. 20694145_PI3K/P-Rex/Rac pathway is required for late phase long-term potentiation in the mouse cerebellum 21178006_Either P-Rex1 or Vav1 alone can produce sufficient active Rac for G protein-coupled receptor-dependent cell responses but under normal circumstances both contribute, presumably to ensure robust cell responses of primary neutrophils. 22002247_Studies identify a novel RacGEF in adipocytes as P-Rex1 that, at physiological insulin concentrations, functions as an insulin-dependent regulator of the actin cytoskeleton that contributes to GLUT4 trafficking to the plasma membrane. 22109529_Data conclude that P-Rex1 has an important role in melanoblast migration and cancer progression to metastasis in mice and humans. 22207728_Demonstrate presence of P-Rex1 in platelets as well as its role in platelet secretion and aggregation induced by low-dose agonists for g-protein coupled receptors and by collagen. 22965143_P-rex1 expressed in endothelial cells is a critical mediator of vascular barrier disruption by an upstream pathway. 23343834_In vivo, in an ischemia-reperfusion-induced model of acute kidney injury, abolished selectin-mediated integrin activation contributed to decreased neutrophil recruitment and reduced kidney damage in P-Rex1-deficient mice. 24327733_P-Rex1 may serve as a biomarker to predict response to single-agent PI3K inhibitors within a subset of breast cancers. 24659802_regulates GPCR-driven Rac signalling and actin polarity in neutrophils 25075639_We have elucidated a proliferative role of P-Rex1 when Rac1 is deleted in melanoblasts. 25538043_Platelet P-Rex and Vav family Rac-GEFs play important proinflammatory roles in leukocyte recruitment. 26621702_findings suggest a vital role of P-Rex1 signaling in CA1 LTD that is critical for social behavior and cognitive function and offer new insight into the etiology of ASDs 26792863_A novel mechanism of P-Rex1 regulation through the GPCR-adaptor protein Norbin. 26887924_Tiam1 and P-Rex1 promote Rac1 anti- and pro-migratory signalling cascades. 27173636_These findings identify P-Rex1 as a novel player of pulmonary fibrosis. 33097662_PtdIns(3,4,5)P3-dependent Rac exchanger 1 (P-Rex1) promotes mammary tumor initiation and metastasis. |
ENSG00000124126 |
PREX1 |
1033.658360 |
0.4312174448 |
-1.213513 |
0.10753498 |
125.033796 |
0.00000000000000000000000000005003523568085281932270368490050228058275003119076254533445642027832111794051570680119667144936101976782083511352539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000035613864948165894877598404299682607507149270789608814181022767375998689005911490568223598529584705829620361328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
611.689482218709 |
45.636078254144 |
1418.51747772887 |
77.0861190459318 |
| ENSMUSG00000039865 |
213603 |
Slc44a3 |
protein_coding |
Q921V7 |
Human_homologues |
Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:213603; |
membrane [GO:0016020]; organic cation transmembrane transporter activity [GO:0015101]; transmembrane transporter activity [GO:0022857]; nitrogen compound transport [GO:0071705]; transmembrane transport [GO:0055085] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000143036 |
SLC44A3 |
74.257060 |
8.1754977564 |
3.031307 |
0.73642471 |
14.823025 |
0.00011808496121991705993681948116957869387988466769456863403320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006939313770454385525005003287901672592852264642715454101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
130.934793893557 |
69.7762605426123 |
16.0155134030185 |
6.34049313900301 |
| ENSMUSG00000039960 |
69581 |
Rhou |
protein_coding |
Q9EQT3 |
FUNCTION: Acts upstream of PAK1 to regulate the actin cytoskeleton, adhesion turnover and increase cell migration. Stimulates quiescent cells to reenter the cell cycle. Has no detectable GTPase activity but its high intrinsic guanine nucleotide exchange activity suggests it is constitutively GTP-bound. Plays a role in the regulation of cell morphology and cytoskeletal organization. Required in the control of cell shape (By similarity). {ECO:0000250}. |
Cell junction;Cell membrane;Cell projection;Golgi apparatus;GTP-binding;Lipoprotein;Magnesium;Membrane;Metal-binding;Nucleotide-binding;Palmitate;Phosphoprotein;Reference proteome |
|
|
mmu:69581; |
cell projection [GO:0042995]; focal adhesion [GO:0005925]; Golgi membrane [GO:0000139]; plasma membrane [GO:0005886]; podosome [GO:0002102]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; metal ion binding [GO:0046872]; actin cytoskeleton organization [GO:0030036]; Cdc42 protein signal transduction [GO:0032488]; cytoskeleton organization [GO:0007010]; endocytosis [GO:0006897]; establishment or maintenance of cell polarity [GO:0007163]; G1/S transition of mitotic cell cycle [GO:0000082]; Rac protein signal transduction [GO:0016601]; regulation of cell shape [GO:0008360] |
14731133_G28K was differentially expressed in the small intestines; interstitial cells of Cajal within the small intestine expressed mRNA for G28K; downregulated in the jejunum of W/W V mice. 16046391_Wrch-1 membrane association, subcellular localization, and biological activity are mediated by a novel membrane-targeting mechanism distinct from that of Cdc42 and other isoprenylated Rho family GTPases 18086875_Wrch1 collaborates with FAK2 and Src in regulating cytoskeletal dynamics. 21903671_Rhou activity maintains the epithelial architecture of the endoderm progenitors, and its downregulation accompanies the transition of the columnar epithelium in the embryonic foregut to a multilayered cell sheet during organ formation. 30834544_Loss of RhoU function in the mouse gut epithelium increases RhoA activity and the level of phosphorylated myosin light chain-2. Rhou knockdown in the mouse gut causes cell hyperplasia. 31556874_Coupling our top hit with gain/loss-of-function genetics, interactome proteomics and tissue imaging, the authors show that RHOU, an atypical Rho, governs the cytoskeletal-junction dynamics that establish columnar shape and planar cell polarity in epidermal progenitors. |
ENSG00000116574 |
RHOU |
64.258529 |
0.4936168300 |
-1.018537 |
0.34172021 |
8.861101 |
0.00291311759930291134038093936453606147551909089088439941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0117232780112501832475713214876122947316616773605346679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
41.8177649744487 |
9.99793492075037 |
84.7170566989943 |
14.6540512574179 |
| ENSMUSG00000040084 |
12236 |
Bub1b |
protein_coding |
Q9Z1S0 |
FUNCTION: Essential component of the mitotic checkpoint. Required for normal mitosis progression and tumor suppression. The mitotic checkpoint delays anaphase until all chromosomes are properly attached to the mitotic spindle. One of its checkpoint functions is to inhibit the activity of the anaphase-promoting complex/cyclosome (APC/C) by blocking the binding of CDC20 to APC/C, independently of its kinase activity. The other is to monitor kinetochore activities that depend on the kinetochore motor CENPE. Required for kinetochore localization of CENPE. Negatively regulates PLK1 activity in interphase cells and suppresses centrosome amplification. Also implicated in triggering apoptosis in polyploid cells that exit aberrantly from mitotic arrest. Essential for tumor suppression. May play a role in regulating aging and fertility (By similarity). {ECO:0000250, ECO:0000269|PubMed:14744753, ECO:0000269|PubMed:15208629}. |
Acetylation;Apoptosis;ATP-binding;Cell cycle;Cell division;Centromere;Chromosome;Cytoplasm;Kinase;Kinetochore;Mitosis;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase;Tumor suppressor;Ubl conjugation |
|
|
mmu:12236; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; kinetochore [GO:0000776]; mitotic checkpoint complex [GO:0033597]; outer kinetochore [GO:0000940]; perinuclear region of cytoplasm [GO:0048471]; spindle [GO:0005819]; ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; apoptotic process [GO:0006915]; cell division [GO:0051301]; meiotic sister chromatid cohesion, centromeric [GO:0051754]; metaphase/anaphase transition of mitotic cell cycle [GO:0007091]; mitotic spindle assembly checkpoint signaling [GO:0007094]; negative regulation of ubiquitin-protein transferase activity [GO:0051444]; protein localization to chromosome, centromeric region [GO:0071459]; protein phosphorylation [GO:0006468] |
14576056_BUBR1 is essential for early embryonic development and normal hematopoiesis 14646599_mutations of Bub1 and BubR1 found in Brca2- mutant mice indeed are responsible for the chromosome instability in Brca2-mutated tumors. 14744753_BubR1 is essential for spindle checkpoint activation and tumor suppression. 15208629_role for BubR1 in regulating aging and infertility 15767571_BubR1 and Apc functionally interact in regulating metaphase-anaphase transition, deregulation of which may play a key role in genomic instability and development and progression of colorectal cancer. 15870277_BubR1 transcription is largely controlled by p53; BubR1 spindle checkpoint protein plays a critical role in suppression of centrosome amplification and stabilization of chromosomes in late-passage p53(-/-) cells 16600213_Data suggest that Mad2 and BubR1 must cooperate to inhibit Cdc20 activity. 17272762_BubR1 may have a role in suppressing the vascular aging process. 17531083_there exists a cross-talk between spindle checkpoint components and ERKs and MEK and that BubR1 may play an important role in mediating the cross-talk 18516091_Data show that skeletal muscle and fat, two tissues that develop early ageing-associated phenotypes in response to BubR1 insufficiency, have high levels of p16(Ink4a) and p19(Arf). 19139399_TAp73 regulates the spindle assembly checkpoint by modulating BubR1 activity. 19154723_Data show that the BubR1 N terminus binds to Cdc20 to inhibit APC/C activity in interphase, thereby allowing accumulation of cyclin B in G(2) phase prior to mitosis onset. 19283064_Shugoshin 2 is necessary for the loading of MCAK at the inner centromere, but is dispensable for the loading of the outer kinetochore proteins BubR1 and CENP-E. 19965510_study found that the spindle assembly checkpoint protein BubR1 regulates prophase I arrest, which is important for sustaining the oocyte pool, and the progression through meiosis I in oocytes 20237433_BubR1 is a spindle assembly checkpoint protein regulating meiotic progression of oocytes. 23242215_Hhigh-level expression of BubR1 extends lifespan and delays age-related deterioration and aneuploidy in several tissues. 23300461_these data demonstrate that the BUBR1 GTTA mutation compromises longevity and healthspan, raising the interesting possibility that mono-allelic changes in BUBR1 might contribute to differences in aging rates in the general population. 23602569_BubR1 insufficiency elicits an aging response that is counteracted by p53 and involves single or multiple p53 targets, depending on the tissue type. 23878276_BubR1 acetylation is essential for embryonic development. 24825348_the loss of BubR1 levels with age is due to a decline in NAD(+) and the ability of SIRT2 to maintain lysine-668 of BubR1 in a deacetylated state, which is counteracted by the acetyltransferase CBP. 24981203_BubR1 (-/-) embryos were aneuploid and had an increased level of premature sister chromatid separation. 25524773_Low BubR1 expression almost completely inhibited intimal hyperplasia after carotid ligation by suppressing the proliferation of VSMCs, which was caused, in part, by delayed cell cycle progression. 25897860_These data reveal that BubR1 plays a multifaceted role in chromosome segregation during the first meiotic division and suggest that age-related decline of BubR1 is a key determinant of the formation of aneuploid oocytes during aging. 26681803_we quantified the frequency of aneuploidy of three autosomes in the cerebral cortex and cerebellum of adult and developing brain of Bub1b(H/H) mice, which have a faulty mitotic checkpoint, and Ercc1(-/Delta7) mice, defective in nucleotide excision repair and inter-strand cross-link repair.we found that Bub1b(H/H), but not Ercc1(-/Delta7) mice, have a significantly higher frequency of aneuploid nuclei relative to wild-t... 26847209_Data show that compound mutant spindle assembly checkpoint components BubR1 and Sgo1 embryonic fibroblasts (MEFs) grew at a much slower rate, and a small fraction of cells exhibited morphologies of senescent cells at early passages. 27226494_EZH2 directly interacted with and stabilized BubR1, an effect driving EZH2 into the concert of meiosis regulation. 27561386_Up-regulation of desmocollin-1 (DSC1) was observed in wild-type, but not Low-budding uninhibited by benzimidazole-related 1(BubR1)-expressing mutant (BubR1(L/L)) mice after partial hepatectomy (PHx). 27815460_The combination of budding uninhibited by benzimidazole-related 1 (BUBR1) insufficiency and administration of KBrO3 worsens the proliferative capacity compared with the impaired proliferation induced by BUBR1 insufficiency alone. 27922816_Consistent with defective myelination, BubR1(H/H) mice exhibited various motor deficits, including impaired motor strength, coordination, and balance, irregular gait patterns and reduced locomotor activity. 28383136_Age-related decline in BubR1 impairs adult hippocampal neurogenesis. 28974341_BubR1 insufficiency impairs angiogenesis and results in limb loss in ischemic hind limbs. 29067790_that Sirt2-dependent BubR1 deacetylation involves in the regulation of meiotic apparatus in normal oocytes and mediates the effects of advanced maternal age on oocyte quality 30078214_the BubR1 signal is markedly elevated in metaphase kinetochores of oocytes with SETD2 depletion, indicative of the activation of spindle assembly checkpoint. In addition, we note that loss of SETD2 results in a drastic decrease in the trimethylation level of H3K36 in oocytes. Collectively, our data demonstrate that SETD2 is required for oocyte maturation and indicate a novel mechanism controlling the meiotic apparatus. 30668728_the ependymal cell layer does not form in the conditional knockout, revealing an unrecognized role of BubR1 in assuring the integrity of the ventricular system, which may account for the presence of hydrocephalus in some patients. 31738183_BUBR1 allelic effects beyond protein level and aneuploidy contribute to disease heterogeneity in both Mosaic-variegated aneuploidy (MVA) patients and heterozygous carriers of MVA mutations. 32716490_Rare deleterious BUB1B variants induce premature ovarian insufficiency and early menopause. 33130993_Spindle assembly checkpoint gene BUB1B is essential in breast cancer cell survival. 33955658_Loss of BubR1 acetylation provokes replication stress and leads to complex chromosomal rearrangements. |
ENSG00000156970 |
BUB1B |
1192.722231 |
2.3171007923 |
1.212321 |
0.11210941 |
114.840428 |
0.00000000000000000000000000852919045109872880258185273787528156705704151895859648330556430243176930031112625840705732116475701332092285156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000005135044622574539009377699153649559909458409979280689900179599296798499397098680674389470368623733520507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1656.92334471334 |
128.88635266602 |
715.084708548631 |
42.4269102618703 |
| ENSMUSG00000040118 |
12293 |
Cacna2d1 |
protein_coding |
O08532 |
FUNCTION: The alpha-2/delta subunit of voltage-dependent calcium channels regulates calcium current density and activation/inactivation kinetics of the calcium channel. Plays an important role in excitation-contraction coupling (By similarity). {ECO:0000250}. |
Alternative splicing;Calcium;Calcium channel;Calcium transport;Disulfide bond;Glycoprotein;Ion channel;Ion transport;Membrane;Metal-binding;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
This gene encodes a regulatory component of the voltage-dependent calcium channel complex. The product of this gene is a proprotein that is proteolytically processed into alpha-2 and delta subunits, which are linked by a disulfide bond. Alternatively spliced transcript variants encoding multiple isoforms have been observed for this gene. [provided by RefSeq, Jan 2013]. |
mmu:12293; |
glutamatergic synapse [GO:0098978]; L-type voltage-gated calcium channel complex [GO:1990454]; neuronal dense core vesicle [GO:0098992]; postsynaptic membrane [GO:0045211]; presynaptic active zone membrane [GO:0048787]; sarcoplasmic reticulum [GO:0016529]; T-tubule [GO:0030315]; voltage-gated calcium channel complex [GO:0005891]; metal ion binding [GO:0046872]; voltage-gated calcium channel activity [GO:0005245]; voltage-gated calcium channel activity involved in bundle of His cell action potential [GO:0086057]; calcium ion transmembrane transport [GO:0070588]; calcium ion transmembrane transport via high voltage-gated calcium channel [GO:0061577]; calcium ion transport [GO:0006816]; calcium ion transport into cytosol [GO:0060402]; cardiac muscle cell action potential involved in contraction [GO:0086002]; cellular response to amyloid-beta [GO:1904646]; membrane depolarization during bundle of His cell action potential [GO:0086048]; positive regulation of high voltage-gated calcium channel activity [GO:1901843]; positive regulation of muscle contraction [GO:0045933]; positive regulation of voltage-gated calcium channel activity [GO:1901387]; regulation of calcium ion transmembrane transport via high voltage-gated calcium channel [GO:1902514]; regulation of calcium ion transport [GO:0051924]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of membrane repolarization during action potential [GO:0098903]; regulation of ventricular cardiac muscle cell membrane repolarization [GO:0060307] |
1847144_This study of the rabbit ortholog of dihydropyridine-sensitive calcium channel alpha 2 provides N-terminal sequencing evidence of the cleavage junction of the alpha2 and delta subunits from the preprotein 16460711_alpha(2)-delta Type 1 subunit of voltage-gated calcium channels is the major binding protein for pregabalin in the brain and spinal cord. 17949410_These results indicate that BZDs examined here have the potential to increase L-type HVCC functions mediated via the enhanced expression of alpha2/delta1 subunit after their sustained exposure. 18063658_The calcium channel alpha2/delta1 subunit in skeletal muscle plays a role in extracellular signalling. The alpha2/delta1 subunit may be crucial for muscle development, muscle repair, and myoblast attachment and migration. 19216737_mechanism of Cavalpha2delta1-mediated spinal sensitization in which elevated Cavalpha2delta1 causes increased pre-synaptic glutamate release that leads to reduced excitation thresholds of post-synaptic dorsal horn neurons to innocuous stimuli. 19596051_Upregulation of alpha(2)/delta subunit of VGCC in the frontal cortex and the limbic forebrain plays a critical role in development of morphine-induced rewarding effect and behavioral sensitization following neuronal plasticity. 21490313_association of calcium channel alpha2/delta subunit 1 and ATP5b occurs in intracellular membranes and at the plasma membrane of developing muscle cells, where they form a signaling complex capable of accelerating the rate of decline of calcium transients 21638318_The alpha(2) delta(1) subunit, previously described as an L-type auxiliary subunit, complexes with the T-type Ca(v)3.2 (alpha(1H)) subunit in MLO-Y4 cells. 22534482_This study demonistrated that Cacna2d1 gene expression in mouse dorsal raphe nucleus 22849384_Expression of Cacna2d1 is enhanced in the amygdala in a model of innate anxiety. 23296739_These findings suggest that the alpha(2)delta(4) subunit participates in the modulation of L-type Ca(2+) current regulating neurotransmitter release from photoreceptor terminals 24133248_alpha2delta-1 is essential for rapid development of mechanical hypersensitivity in a nerve injury model of neuropathic pain. 24151064_Our findings support a role of peripheral nerve injury-induced alpha2delta1 in enhancing pre-synaptic excitatory 24403154_These results suggest calcium channel-independent effects of alpha2delta-1 on feeding and implicate alpha2delta-1-thrombospondin interactions known to facilitate excitatory synapse assembly 24889613_CaV2.2 and alpha2delta-1 are intimately associated at the plasma membrane 27129212_a TSP4/Cavalpha2delta1-dependent pathway activated by TSP4 or peripheral nerve injury promotes exaggerated presynaptic excitatory input and evoked sensory neuron hyperexcitability and excitatory synaptogenesis. 27231153_Linkage mapping, transcript covariance and pharmacological testing suggest that genetic variation affecting Cacna2d1 may contribute to individual differences in von Frey filament response. 28115397_Loss of the Ca(2+) channel alpha2delta-1 subunit function increases the susceptibility for developing diabetes in a sex-dependent manner. 28193459_Results show that overexpression of alpha2delta-1 alone in TG mice can enhance excitatory connectivity in neocortex and lead to neural circuit hyperexcitability and episodes of electrographic epileptiform activity, associated with behavioral arrests in transgenic mice. 28646556_REVIEW: recent findings related to maladaptive changes in the dysregulated VGCC alpha2 delta1 subunit (Cav alpha2 delta1 ) with a focus on exploring the mechanisms underlying the contribution of Cav alpha2 delta1 to pain signal transduction 29109971_Study identified a critical role for alpha2delta-1 in freeze-lesion-induced developmental cortical malformation pathologies and in mediating the neuroprotective effects of gabapentin (GBP). Genetic deletion of alpha2delta-1 did not eliminate GBP's effects on synaptogenesis, suggesting that GBP can have alpha2delta-1-independent effects. 29176626_Cacna2d1 regulated the intraocular pressure and glaucoma susceptibility. 29466724_large conductance calcium-activated potassium (BK) channels interact with the Cavalpha2delta subunit. 29490268_Cacna2d1 overexpression potentiates presynaptic and postsynaptic NMDAR activity of spinal dorsal horn neurons to cause pain hypersensitivity. 29921713_alpha2delta-1-NMDAR complexes in the hypothalamus serve as an important molecular substrate for the interaction between the sympathetic nervous system and the renin-angiotensin system 30054448_TSP-alpha2delta-1 interactions control synaptogenesis postsynaptically via Rac1, suggesting potential molecular mechanisms that underlie both synaptic development and pathology. 30355732_the interaction between alpha2delta-1 and NMDARs is of high physiological relevance 30683685_Presynaptic expression of a single alpha2delta calcium channel variant can modulate synaptic connectivity and the localization of inhibitory postsynaptic receptors. 32337532_Sex-specific Effects of alpha2delta-1 in the Ventromedial Hypothalamus of Female Mice Controlling Glucose and Lipid Balance. 33782113_Presynaptic alpha2delta subunits are key organizers of glutamatergic synapses. 34032393_The alpha2delta-1/NMDA receptor complex is involved in brain injury after intracerebral hemorrhage in mice. 36222452_alpha2delta-1 protein drives opioid-induced conditioned reward and synaptic NMDA receptor hyperactivity in the nucleus accumbens. |
ENSG00000153956 |
CACNA2D1 |
209.244037 |
0.2874476626 |
-1.798629 |
0.18835715 |
89.831215 |
0.00000000000000000000259369645410246380169013479159085217446152687013473919352014168682174499735992867499589920043945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001097421788580242518043627076447486278615967138890723107241820244439622911158949136734008789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
92.2339439139367 |
12.0762322042252 |
320.872130525034 |
29.4930647893344 |
| ENSMUSG00000040164 |
16538 |
Kcns1 |
protein_coding |
O35173 |
FUNCTION: Potassium channel subunit that does not form functional channels by itself. Can form functional heterotetrameric channels with KCNB1 and KCNB2; modulates the delayed rectifier voltage-gated potassium channel activation and deactivation rates of KCNB1 and KCNB2 (PubMed:9305895). {ECO:0000269|PubMed:9305895}. |
Cell membrane;Ion channel;Ion transport;Membrane;Potassium;Potassium channel;Potassium transport;Reference proteome;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
|
mmu:16538; |
membrane [GO:0016020]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; voltage-gated potassium channel complex [GO:0008076]; delayed rectifier potassium channel activity [GO:0005251]; potassium channel regulator activity [GO:0015459]; voltage-gated potassium channel activity [GO:0005249]; potassium ion transmembrane transport [GO:0071805]; potassium ion transport [GO:0006813]; protein homooligomerization [GO:0051260]; regulation of delayed rectifier potassium channel activity [GO:1902259] |
29697531_After neuropathic injury, Kcns1 KO mice exhibited exaggerated mechanical pain responses and hypersensitivity to both noxious and innocuous cold, consistent with increased A-fiber activity. Interestingly, Kcns1 deletion also improved locomotor performance in the rotarod test, indicative of augmented proprioceptive signalling. |
ENSG00000124134 |
KCNS1 |
67.710496 |
6.2240049778 |
2.637843 |
0.35678967 |
54.613619 |
0.00000000000014671534469076839682073547441079101140209636766620349135337164625525474548339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000032247736368395155470298917000796372106692644354808408024837262928485870361328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
117.74728004264 |
29.4175578695993 |
18.9182496581491 |
3.87716247419893 |
| ENSMUSG00000040219 |
235330 |
Ttc12 |
protein_coding |
Q8BW49 |
FUNCTION: Cytoplasmic protein that plays a role in the proper assembly of dynein arm complexes in motile cilia in both respiratory cells and sperm flagella. {ECO:0000250|UniProtKB:Q9H892}. |
Cilium biogenesis/degradation;Cytoplasm;Phosphoprotein;Reference proteome;Repeat;TPR repeat |
|
|
mmu:235330; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; axonemal dynein complex assembly [GO:0070286]; sperm axoneme assembly [GO:0007288] |
Human_homologues 17085484_Observational study of gene-disease association. (HuGE Navigator) 17657212_the TTC12 gene is hypermethylated in acute lymphoblastic leukemia 17761687_Observational study of gene-disease association. (HuGE Navigator) 17761687_association studies of alcohol dependence and 43 SNPs mapped to the gene cluster of NCAM1, TTC12, ANKK1 and DRD2 18828801_Genetic linkage tests conclude that variants in TTC12 exon 3 are involved in the regulation of risk for comorbid alcohol and drug dependence. 18828801_Observational study of gene-disease association. (HuGE Navigator) 20133381_Observational study of gene-disease association. (HuGE Navigator) 20133381_This study evaluated the interaction of ANKK1, TTC12, sex, and continental ancestry in tobacco smokers. 21168125_TTC12-ANKK1-DRD2s seemed to influence smoking behavior mainly in adolescence, and its effect is partially mediated by personality characteristics promoting drug-seeking behavior. 23303482_TTC12 SNP rs7130431 was associated with heroin dependence. 30104163_Meta-analysis found that the minor G-allele of rs2236709, mapping TTC12, was associated with self-reported smoking and higher plasma cotinine levels. This risk allele was linked to an increased ventral-striatal blood-oxygen level-dependent response during reward anticipation and with higher DRD2 gene expression in the striatum, but not with TTC12 or ANKK gene expression. 31978331_TTC12 Loss-of-Function Mutations Cause Primary Ciliary Dyskinesia and Unveil Distinct Dynein Assembly Mechanisms in Motile Cilia Versus Flagella. 34285142_An association study in the Taiwan Biobank elicits three novel candidates for cognitive aging in old adults: NCAM1, TTC12 and ZBTB20. 36273201_Biallelic mutations of TTC12 and TTC21B were identified in Chinese patients with multisystem ciliopathy syndromes. |
ENSG00000149292 |
TTC12 |
266.706444 |
2.0667959778 |
1.047396 |
0.16037075 |
42.524360 |
0.00000000006980737395603854156496812049224626878229926774110936094075441360473632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000011311765107429564416930075113846415613139129163755569607019424438476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
354.915556117116 |
39.5339042121707 |
171.722588940326 |
14.8615230372346 |
| ENSMUSG00000040253 |
229900 |
Gbp7 |
protein_coding |
Q91Z40 |
FUNCTION: Hydrolyzes GTP to GMP in two consecutive cleavage reactions and predominantly uses GTP and not GDP or GMP as the substrate (PubMed:21551061, PubMed:31689351). Confers protection to several pathogens, including the bacterial pathogens Listeria monocytogenes and Mycobacterium bovis BCG as well as the protozoan pathogen Toxoplasma gondii (PubMed:21551061, PubMed:31964735, PubMed:18025219). Promotes IFN-gamma-mediated host defense against bacterial infections by regulating oxidative responses and bacteriolytic peptide generation (PubMed:21551061). May help to assemble NADPH oxidase on phagosomal membranes by acting as a bridging protein between NADPH oxidase cytosolic subunits NCF2-NCF4 and the membrane subunits CYBA-CYBB (PubMed:21551061). Participates along with GBP1 in trafficking monoubiquinated protein cargo to autolysosomes for generating ubiquitin-derived antimicrobial peptides (PubMed:21551061). Required for disruption of the parasitophorous vacuole formed following T.gondii infection and subsequent killing of the parasite (PubMed:31964735). Facilitates influenza A virus replication by inhibiting the activation of NF-kappaB and JAK-STAT signaling pathways and the expression of type I, type III interferons and pro-inflammatory cytokines (By similarity). {ECO:0000250|UniProtKB:Q8N8V2, ECO:0000269|PubMed:21551061, ECO:0000269|PubMed:31689351, ECO:0000269|PubMed:31964735, ECO:0000303|PubMed:18025219}. |
Antimicrobial;Antiviral defense;Coiled coil;Cytoplasmic vesicle;GTP-binding;Hydrolase;Immunity;Membrane;Nucleotide-binding;Reference proteome |
|
|
mmu:229900; |
cytoplasmic vesicle [GO:0031410]; membrane [GO:0016020]; symbiont-containing vacuole membrane [GO:0020005]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; adhesion of symbiont to host [GO:0044406]; cellular response to interferon-beta [GO:0035458]; cellular response to type II interferon [GO:0071346]; defense response to Gram-positive bacterium [GO:0050830]; defense response to protozoan [GO:0042832]; defense response to virus [GO:0051607]; negative regulation of cytokine production [GO:0001818]; negative regulation of receptor signaling pathway via JAK-STAT [GO:0046426]; negative regulation of type I interferon production [GO:0032480]; negative regulation of type III interferon production [GO:0034345]; positive regulation of viral genome replication [GO:0045070]; regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043122] |
21551061_findings show that at least 4 IFN-gamma-inducible Gbps--Gbp1, Gbp6, Gbp7, and Gbp10--conferred cell-autonomous immunity to listerial or mycobacterial infection within macrophages and gene-deficient animals 31689351_GBP7 exhibits a concentration-dependent GTPase activity and an apparent GTP turnover number of 20 min-1. 33408175_Inducible Guanylate-Binding Protein 7 Facilitates Influenza A Virus Replication by Suppressing Innate Immunity via NF-kappaB and JAK-STAT Signaling Pathways. |
ENSG00000162654 |
GBP4 |
43.623238 |
2.6316148547 |
1.395948 |
0.45442792 |
9.327251 |
0.00225770785972971739508086130854280781932175159454345703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0094554319822389484156976990902876423206180334091186523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
63.9794270679101 |
20.4874872599409 |
24.3118505555917 |
5.9936162503557 |
| ENSMUSG00000040258 |
104080 |
Nxph4 |
protein_coding |
G3X9N5 |
FUNCTION: May be signaling molecules that resemble neuropeptides. {ECO:0000256|PIRNR:PIRNR038019}. |
Proteomics identification;Reference proteome;Signal |
|
|
mmu:104080; |
extracellular region [GO:0005576]; signaling receptor binding [GO:0005102] |
31524598_data illustrate how selectively expressed Neurexophilin4, an alpha-neurexin ligand, regulates specific synapse function and modulates cerebellar motor control. |
ENSG00000182379 |
NXPH4 |
67.772920 |
4.0552693047 |
2.019798 |
0.36543316 |
29.613921 |
0.00000005272490896485402030815053107427881062818642021738924086093902587890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000005740570502878173345947350122986385656531638232991099357604980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
104.491240473909 |
26.7514602101057 |
25.7667820859746 |
5.25455954444698 |
| ENSMUSG00000040280 |
407790 |
Ndufa4l2 |
protein_coding |
Q4FZG9 |
|
Reference proteome |
|
|
mmu:407790; |
mitochondrial respiratory chain complex IV [GO:0005751] |
22100406_Induction of the mitochondrial NDUFA4L2 protein by HIF-1alpha decreases oxygen consumption by inhibiting Complex I activity. 31740659_The results demonstrated that the upregulation of NDUFA4L2 ameliorated the apoptosis of NP cells by repressing excessive mitophagy, which ultimately alleviated IVDD. These findings show for the first time that NDUFA4L2 and mitophagy may be potential therapeutic targets for IVDD. 34724256_Mitochondrial NDUFA4L2 is a novel regulator of skeletal muscle mass and force. |
ENSG00000185633 |
NDUFA4L2 |
834.293680 |
3.7651333719 |
1.912701 |
0.13761271 |
185.722642 |
0.00000000000000000000000000000000000000000272923255727050157011182128795555773208622648702287211710405734422805152704976274627354757328664896342052047055470875847049683216027915477752685546875000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000003379810594499534995619857714066599444743804733937144470605577027267090476862699037248267500800649835177372204420009893510723486542701721191406250000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1306.09167363354 |
124.771684535654 |
346.891210645642 |
25.6398849821065 |
| ENSMUSG00000040287 |
237611 |
Stac3 |
protein_coding |
Q8BZ71 |
FUNCTION: Required for normal excitation-contraction coupling in skeletal muscle and for normal muscle contraction in response to membrane depolarization (PubMed:23818578, PubMed:27621462, PubMed:29467163). Required for normal Ca(2+) release from the sarcplasmic reticulum, which ultimately leads to muscle contraction (PubMed:23818578). Probably functions via its effects on muscle calcium channels. Increases CACNA1S channel activity, in addition to its role in enhancing the expression of CACNA1S at the cell membrane (PubMed:27621462). Has a redundant role in promoting the expression of the calcium channel CACNA1S at the cell membrane (PubMed:25548159, PubMed:27621462, PubMed:29467163). Slows down the inactivation rate of the calcium channel CACNA1C (PubMed:25548159, PubMed:29363593). {ECO:0000269|PubMed:23818578, ECO:0000269|PubMed:25548159, ECO:0000269|PubMed:27621462, ECO:0000269|PubMed:29363593, ECO:0000269|PubMed:29467163}. |
Cell membrane;Cytoplasm;Membrane;Metal-binding;Reference proteome;Repeat;SH3 domain;Zinc;Zinc-finger |
|
|
mmu:237611; |
cytosol [GO:0005829]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; nucleoplasm [GO:0005654]; synapse [GO:0045202]; T-tubule [GO:0030315]; voltage-gated calcium channel complex [GO:0005891]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; neuromuscular synaptic transmission [GO:0007274]; positive regulation of protein localization to plasma membrane [GO:1903078]; positive regulation of voltage-gated calcium channel activity [GO:1901387]; skeletal muscle contraction [GO:0003009]; skeletal muscle fiber development [GO:0048741] |
23626854_Stac3 gene plays a critical role in skeletal muscle development and function in mice. 23818578_STAC3 mediates voltage-induced Ca2+ release and contractility in skeletal muscles. 24788338_Stac3 may play a role in preventing precocious myoblast differentiation during skeletal muscle development 27073615_STAC3 is important to myofiber hypertrophy, myofiber-type composition, contraction, and excitation-induced calcium release from the sarcoplasmic reticulum in the postnatal skeletal muscle. 27184118_These data together confirm a critical role of STAC3 in excitation-contraction coupling but also suggest that STAC3 may have additional functions in skeletal muscle, at least in the hindlimb muscle. 27621462_Stac3 is directly involved in conformational coupling between CaV1.1 and RyR1. 28112192_Thus, the C1 domain of STAC3 is responsible for its stable incorporation into the voltage-sensor (CaV1.1) of excitation-contraction-coupling complex, whereas the SH3 domain containing the Native American myopathy mutation site may be involved in low-affinity functional interactions in excitation-contraction-coupling. 29467163_RyR1 Ca2+ release by CaV1.1 depends on Stac3 being bound to critical domain residues in the II-III loop. 30071129_we provide evidence that endogenous STAC3 incorporates into triads in the absence of the DHPR in myotubes and muscle fibers of dysgenic mice. This finding demonstrates that STAC3 interacts with additional triad proteins and is consistent with its proposed role in directly or indirectly linking the DHPR with the RyR1 |
ENSG00000185482 |
STAC3 |
34.182162 |
0.3998202950 |
-1.322576 |
0.48486577 |
7.330294 |
0.00678019659046665695845135246599966194480657577514648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0240961163009771640874934206522084423340857028961181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.8808574136395 |
6.46567509914087 |
47.2233592187614 |
11.5639433472806 |
| ENSMUSG00000040289 |
15213 |
Hey1 |
protein_coding |
Q9QUM5 |
Human_homologues FUNCTION: Transcriptional repressor which binds preferentially to the canonical E box sequence 5'-CACGTG-3' (PubMed:11095750). Downstream effector of Notch signaling required for cardiovascular development. Specifically required for the Notch-induced endocardial epithelial to mesenchymal transition, which is itself criticial for cardiac valve and septum development. May be required in conjunction with HEY2 to specify arterial cell fate or identity. Promotes maintenance of neuronal precursor cells and glial versus neuronal fate specification. Represses transcription by the cardiac transcriptional activators GATA4 and GATA6 and by the neuronal bHLH factors ASCL1/MASH1 and NEUROD4/MATH3 (PubMed:15485867). Involved in the regulation of liver cancer cells self-renewal (PubMed:25985737). {ECO:0000250|UniProtKB:Q9WV93, ECO:0000269|PubMed:11095750, ECO:0000269|PubMed:15485867, ECO:0000269|PubMed:25985737}. |
Reference proteome;Repressor |
|
|
mmu:15213; |
DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; protein dimerization activity [GO:0046983]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific double-stranded DNA binding [GO:1990837]; angiogenesis [GO:0001525]; negative regulation of Notch signaling pathway [GO:0045746]; negative regulation of transcription from RNA polymerase II promoter involved in smooth muscle cell differentiation [GO:2000820]; negative regulation of transcription regulatory region DNA binding [GO:2000678]; Notch signaling pathway [GO:0007219] |
12947105_results indicate that hairy enhancer of split related with YRPW motif 1 and 2 negatively regulate neuronal basic helix- loop-helix genes, promote maintenance of neural precursor cells, and increase late-born cell types in the developing brain 16199874_Loss of Hey1/2 leads to elevated GATA4/6 and ANF mRNA levels in embryoid bodies, while forced expression of Hey factors strongly represses expression of the GATA4 and GATA6 promoter. 17259303_Hesr1 and Hesr2 may prevent cells from expressing the AV canal-specific genes that lead to the precise formation of the AV boundary 17303760_We report that combined inactivation of Hey1 and HeyL, two primary target genes of Notch, causes severe heart malformations, including membranous ventricular septal defects and dysplastic atrioventricular and pulmonary valves. 18291358_Hesr1 and Hesr2 are the downstream mediators of the prosensory function of Notch in early cochlear development. 18986983_Hey1 and Runx2 were shown to act synergistically in BMP9-induced osteogenic differentiation, and Runx2 expression significantly decreased in the absence of Hey1, suggesting that Runx2 may function downstream of Hey1 19433762_Notch1, rather than Notch3, mediates SMC proliferation and neointimal formation after vascular injury through hey1 19917614_Hey1 inhibits myogenesis by associating with and repressing expression of key myogenic targets. 21300049_Hes1, Hes5 and Hey1 cooperatively inhibit hair cell formation, and one allele of Hes1, Hes5 or Hey1 is sufficient for supporting cell production probably by lateral inhibition in the sensory epithelium. 21989910_These results indicate that Hesr1 and Hesr3 are essential for the generation of adult satellite cells and for the maintenance of skeletal muscle homeostasis. 22457635_Notch-RBPjk signaling functions in part through Hey1-mediated inhibition of NFATc1 to suppress osteoblastogenesis, contributing to bone homeostasis in vivo 24431082_This study demonistrated that demonstrates that the lack of Hesr1 leads to an alteration in sensitivity to dopamine accompanied by enhanced prepulse inhibition. 25005473_A muscle-specific regulatory element of p57(kip2) directly activated by muscle regulatory factors in myoblasts but repressed by the Notch targets Hes1/Hey1 in progenitor cells, is identified. 25232121_findings indicate that Hey1 and Hey2 control the spatial and temporal pattern of auditory HC differentiation. 25264302_Hey1 and Hey2 in endothelial cells play important roles in vascular development. 25446183_Hey proteins mechanistically repress target genes via histone deacetylase recruitment and histone deacetylation. 26647760_Cyclic stretch enhanced the BMP-2induced osteoblastic differentiation through the inhibition of Hey1. 26719988_These in vitro and in vivo data support an anti-adipogenic role of COUP-TFII via downregulating the Notch signaling target gene Hey1. 26932672_Hes/Hey signaling at the Atoh1 promoter has a role in selection of cell fate in the organ of Corti 28139517_A Jagged1-Hey1 signal might mediate the impairment of angiogenesis induced by Ang II during cardiac hypertrophy. 29138798_stable Hey1overexpressing cells expressed higher levels of dentin sialophosphoprotein (DSPP) and exhibited higher mineralization capabilities following stimulation by differentiation medium. Furthermore, RNA interferencemediated knockdown of Hey1 downregulated the expression levels of DSPP in OLCs stimulated by differentiation medium. 29155305_Data implied that Hey2 function is restricted to transient amplifying cells of the ameloblast cell lineage and that Hey1 plays a role in the composition of the subodontoblastic layer, in addition to ameloblast differentiation. 30538222_our findings suggested that CCN3 significantly inhibited the osteogenic differentiation of mouse embryonic fibroblasts. The inhibitory effect of CCN3 was mainly through the inhibition of BMP signaling and the mutual inhibition with DLL1, so as to inhibit the expression of Hey1, the target gene shared by BMP and Notch signaling pathways. 30745427_HeyL and Hey1 function redundantly in muscle stem cells, and HeyL requires Hes1 for effective DNA binding and biological activity 31550547_BMP9 prevents induction of osteopontin in JNK-inactivated osteoblasts via Hey1-Id4 interaction. 33454003_Importance of endothelial Hey1 expression for thoracic great vessel development and its distal enhancer for Notch-dependent endothelial transcription. 35864959_Babam2 negatively regulates osteoclastogenesis by interacting with Hey1 to inhibit Nfatc1 transcription. |
ENSG00000164683 |
HEY1 |
13.083137 |
0.0948748273 |
-3.397831 |
0.84931119 |
16.668257 |
0.00004451974279765492600348386620368046351359225809574127197265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002900447914002907763737770796552695173886604607105255126953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.29364539483571 |
1.42961869154855 |
24.1754895431063 |
8.75754428914748 |
| ENSMUSG00000040447 |
216892 |
Spns2 |
protein_coding |
Q91VM4 |
FUNCTION: Lipid transporter that specifically mediates export of sphingosine-1-phosphate (sphing-4-enine 1-phosphate, S1P) and sphinganine-1-phosphate in the lymph, thereby playing a role in lymphocyte trafficking (PubMed:22664872, PubMed:23103166, PubMed:22406534, PubMed:23180825, PubMed:34260944). S1P is a bioactive signaling molecule that regulates many physiological processes important for the development and for the immune system (PubMed:22664872, PubMed:23103166, PubMed:22406534, PubMed:23180825, PubMed:34260944). Regulates levels of S1P and the S1P gradient that exists between the high circulating concentrations of S1P and low tissue levels that control lymphocyte trafficking (PubMed:22664872, PubMed:22406534, PubMed:23180825, PubMed:34260944). Required for the egress of T-cells from lymph nodes during an immune response by mediating S1P secretion, which generates a gradient that enables activated T-cells to access lymph (PubMed:22406534, PubMed:34260944). Also required for the egress of immature B-cells from the bone marrow (PubMed:22406534). In contrast, it does not mediate S1P release from red blood cells (PubMed:23103166, PubMed:22406534). Involved in auditory function: S1P release in the inner ear is required for maintenance of the endocochlear potential in the cochlea (PubMed:25356849). In addition to export, also able to mediate S1P import (PubMed:33785361). {ECO:0000269|PubMed:22406534, ECO:0000269|PubMed:22664872, ECO:0000269|PubMed:23103166, ECO:0000269|PubMed:23180825, ECO:0000269|PubMed:25356849, ECO:0000269|PubMed:33785361, ECO:0000269|PubMed:34260944}. |
Alternative splicing;Cell membrane;Deafness;Endosome;Lipid transport;Membrane;Non-syndromic deafness;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:216892; |
endosome membrane [GO:0010008]; membrane [GO:0016020]; plasma membrane [GO:0005886]; sphingolipid transporter activity [GO:0046624]; transmembrane transporter activity [GO:0022857]; B cell homeostasis [GO:0001782]; bone development [GO:0060348]; lipid transport [GO:0006869]; lymph node development [GO:0048535]; lymphocyte homeostasis [GO:0002260]; lymphocyte migration [GO:0072676]; regulation of eye pigmentation [GO:0048073]; regulation of humoral immune response [GO:0002920]; regulation of T cell migration [GO:2000404]; sensory perception of sound [GO:0007605]; sphingolipid metabolic process [GO:0006665]; sphingosine-1-phosphate receptor signaling pathway [GO:0003376]; T cell homeostasis [GO:0043029] |
21084291_The sphingosine 1-phosphate transporter, SPNS2, functions as a transporter of the phosphorylated form of the immunomodulating agent FTY720. 22406534_Spns2 functions in endothelial cells to establish the S1P gradient required for T and B cells to egress from their respective primary lymphoid organs. 22664872_The changes in lymphocyte subpopulations in mutant Spns2 mice closely mimick the phenotypes of partial sphingosine-1-phosphate (S1P) deficiency, impaired S1P-dependent lymphocyte trafficking, and depletion of circulating lymphocytes. 22723910_SPNS2 is the first physiological S1P transporter in mammals and is a key determinant of lymphocyte egress from the thymus. 23103166_Spns2 expression by endothelial cells is essential for secretion of lymph S1P, while Spns2 is dispensable for RBC secretion of plasma S1P. 23180825_Spns2 is an S1P transporter in vivo that plays a role in regulation not only of blood S1P but also lymph node and lymph S1P levels and consequently influences lymphocyte trafficking and lymphatic vessel network organization. 26324848_results point to a broad role for Spns2-mediated S1P transport in the initiation and development of adaptive immune related disorders 28052056_genome-wide in vivo screen identifies novel host regulators of metastatic colonization, such as Spns2 29452570_SPNS2 is essential for proper postnatal retinal morphogenesis. 30484906_In summary, these results suggest that Spns2 promotes pro-inflammatory polarization of microglia and may play a crucial role in Alzheimer disease pathogenesis. 34240614_Endothelial Spns2 and ApoM Regulation of Vascular Tone and Hypertension Via Sphingosine-1-Phosphate. 34260944_SPNS2 enables T cell egress from lymph nodes during an immune response. 35977478_Mfsd2b and Spns2 are essential for maintenance of blood vessels during development and in anaphylactic shock. 35988880_Anomaly of cornea and ocular adnexa in spinster homolog 2 (Spns2) knockout mice. 36198832_Postnatal deletion of Spns2 prevents neuroinflammation without compromising blood vascular functions. |
ENSG00000183018 |
SPNS2 |
2851.899287 |
0.2759079379 |
-1.857741 |
0.08073275 |
507.310201 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000024402865020814029674751210260047360109618040816933447212578960656574490087581725505536719882109120539950687875732893538357524994038277819586810139973773 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001548768499987663705705912779132807373980890663863242281418332180066003202400914089350473575168486388614029080747134615060954858820453665931870628618019 |
Yes |
Yes |
1207.32716332837 |
67.5969042765619 |
4375.83337591802 |
177.87486896899 |
| ENSMUSG00000040483 |
327959 |
Xaf1 |
protein_coding |
Q5NBU8 |
FUNCTION: Seems to function as a negative regulator of members of the IAP (inhibitor of apoptosis protein) family. Inhibits anti-caspase activity of BIRC4. Induces cleavage and inactivation of BIRC4 independent of caspase activation. Mediates TNF-alpha-induced apoptosis and is involved in apoptosis in trophoblast cells. May inhibit BIRC4 indirectly by activating the mitochondrial apoptosis pathway. After translocation to mitochondria, promotes translocation of BAX to mitochondria and cytochrome c release from mitochondria. Seems to promote the redistribution of BIRC4 from the cytoplasm to the nucleus, probably independent of BIRC4 inactivation which seems to occur in the cytoplasm. The BIRC4-XAF1 complex mediates down-regulation of BIRC5/survivin; the process requires the E3 ligase activity of BIRC4. Seems to be involved in cellular sensitivity to the proapoptotic actions of TRAIL. May be a tumor suppressor by mediating apoptosis resistance of cancer cells (By similarity). {ECO:0000250}. |
Alternative splicing;Apoptosis;Cytoplasm;Metal-binding;Mitochondrion;Nucleus;Reference proteome;Tumor suppressor;Zinc;Zinc-finger |
|
|
mmu:327959; |
mitochondrion [GO:0005739]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; metal ion binding [GO:0046872]; apoptotic process [GO:0006915]; negative regulation of protein-containing complex assembly [GO:0031333]; response to interferon-beta [GO:0035456] |
18192275_Interferon gamma induces XAF1 and Noxa expression and potentiates apoptosis by STAT3 activation 18544999_XAF1 possesses a potential antiangiogenesis effect. Suppressed expression of Tie-1, Ang-1, Ang-2 and c-Myc may be mechanistically responsible for the observed antiangiogenesis effect. 27097110_XAF1 may be a candidate tumor suppressor in neuroblastoma 29132171_Macrophages activated by metabolic endotoxemia infiltrated into islets and produced IFNbeta, which induced beta-cell apoptosis by increasing the expression of Xaf1. 35829914_XAF1 overexpression exacerbates diabetes by promoting pancreatic beta-cell apoptosis. 35972291_XAF1 Protects Host against Emerging RNA Viruses by Stabilizing IRF1-Dependent Antiviral Immunity. 36394357_XAF1 prevents hyperproduction of type I interferon upon viral infection by targeting IRF7. |
ENSG00000132530 |
XAF1 |
2110.809689 |
2.5867612307 |
1.371147 |
0.07842765 |
300.088068 |
0.00000000000000000000000000000000000000000000000000000000000000000031519860812239670486957271583591554770895209768937404034153951045374632635537598218079641818902008610564596104735485466048823445888565819786765282889970060017158595168373924622073900536634027957916 |
0.0000000000000000000000000000000000000000000000000000000000000000813746643884804447031858306084448834853042669033877340862998163366101403442004826571391864359257000736946820143800747655091567886292879319752022036968649160832388333997045037904172204434871673584 |
Yes |
No |
3067.80311616278 |
166.853772334379 |
1185.96300260436 |
49.6456381481282 |
| ENSMUSG00000040488 |
108075 |
Ltbp4 |
protein_coding |
Q8K4G1 |
FUNCTION: Key regulator of transforming growth factor beta (TGFB1, TGFB2 and TGFB3) that controls TGF-beta activation by maintaining it in a latent state during storage in extracellular space. Associates specifically via disulfide bonds with the Latency-associated peptide (LAP), which is the regulatory chain of TGF-beta, and regulates integrin-dependent activation of TGF-beta. {ECO:0000250|UniProtKB:Q14766}. |
Alternative splicing;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Growth factor binding;Phosphoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:108075; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; microfibril [GO:0001527]; calcium ion binding [GO:0005509]; transforming growth factor beta binding [GO:0050431]; elastic fiber assembly [GO:0048251]; hormone secretion [GO:0046879]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
12208849_Mice homozygous for the disrupted allele develop severe pulmonary emphysema, cardiomyopathy, and colorectal cancer. 15466481_LTBP-4 has a role in targeting and activation of TGF-beta1 which regulates BMP-4 signaling in mouse lung 19016471_Results indicate taht there are two separate functions of LTBP-4, as a regulator of elastic fiber assembly and of TGF-beta levels in lungs. 19884661_an insertion/deletion polymorphism of 36 bp in the coding region of the Ltbp4 was found in an animal model of muscular dystrophy 20945348_These data indicate that LTBP-3 and -4 perform partially overlapping functions only in the lungs. 21468687_The lack of LTBP4-mediated targeting in malignant mammary tumor tissues may lead to a possible modification of TGF-ss1 and BMP bioavailability and function. 23382201_data suggest a unique function for LTBP-4 during elastic fibrogenesis, making it a potential therapeutic target for elastic fiber regeneration 24962333_These results are consistent with a role for TGFbeta2 in lung septation and for Ltbp4 in regulating fibulin-5 dependent elastic fiber assembly. 25713297_Ltbp-4L expression is essential for incorporation of fibulin-4 into the extracellular matrix. 27148972_we found that myostatin forms a complex with LTBP4 and that overexpression of LTBP4 led to a decrease in myostatin levels. LTBP4 also interacted with TGFbeta and GDF11, a protein highly related to myostatin. These data identify LTBP4 as a multi-TGFbeta family ligand binding protein with the capacity to modify muscle disease through overexpression 27585882_These data clearly establish a functional link between Ltbp-4L and fibulin-4 as a crucial molecular requirement for survival and elastogenesis in mice. 27645114_Ltbp4 regulates Pdgfrb expression via TGFbeta-dependent modulation of Nrf2 transcription factor function. 28252045_These data suggest that LTBP-2 and -4 have critical overlapping functions in forming the robust structure of microfibrils in vitro and in vivo. 28684544_Low LTBP4 expression is associated with lung fibrosis. 29065150_Anxa6 and Ltbp4 isoforms modify the extent of damage in acute muscle injury in an additive fashion, and the severe isoform of Ltbp4, characteristic of the DBA/2J genetic background, correlated with upregulation of Slug and Snail, and downregulation of Anxa1 and Anxa6. 30463013_an AMPK-LTBP4 axis in inflammatory macrophages controls the production of TGF-beta1, which is further activated by and acts on fibroblastic cells, leading to fibrosis in Duchenne muscular dystrophy. 34645813_LTBP4 affects renal fibrosis by influencing angiogenesis and altering mitochondrial structure. |
ENSG00000090006 |
LTBP4 |
1156.913558 |
0.3933005783 |
-1.346296 |
0.13475135 |
97.205868 |
0.00000000000000000000006248120447997946373414717234961197253675409463280274424894862508178539073355750588234513998031616210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000029741053332470224690434280005523798608745487556324279149752923268046345128823304548859596252441406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
641.355731713385 |
59.9913797167229 |
1630.70121708215 |
112.455405271927 |
| ENSMUSG00000040533 |
17180 |
Matn1 |
protein_coding |
P51942 |
FUNCTION: Cartilage matrix protein is a major component of the extracellular matrix of non-articular cartilage. It binds to collagen. |
Coiled coil;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:17180; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; matrilin complex [GO:0120216]; calcium ion binding [GO:0005509]; chondrocyte differentiation [GO:0002062]; extracellular matrix organization [GO:0030198]; growth plate cartilage chondrocyte morphogenesis [GO:0003429]; regulation of bone mineralization [GO:0030500] |
12009334_Expression of matrilin-1 during maturation of mouse skeletal tissues 17313942_The chondrocytes of the presumptive articular surface therefore appear to arise directly from a subpopulation of early chondrocytes that do not activate matrilin-1 expression rather than by redifferentiation from the flattened cells of the interzone. 17502381_Matrilin-1 and matrilin-3 modulate collagen fibrillogenesis in cartilage and show that biochemical compensation might exist between matrilins, evidenced by Matn3 deficiency. 17525268_Data show that the cartilage matrix protein subdomain of type VII collagen is pathogenic for epidermolysis bullosa acquisita. 17889519_Transgenic mice expressing mutant (D469Delta) cartilage oligomeric matrix protein showed a slight gender specific growth retardation and both, tibial growth plate and sternum abnormalities. 22083516_Secretion of matrilin 3 V194D mutant protein is not dependent on hetero-oligomerization with matrilin 1. 24692560_Matrilin-1 is an inhibitor of neovascularization 27270603_Results provide evidence that Matn1 plays a protective role in cartilage degeneration under altered mechanical environment. |
ENSG00000162510 |
MATN1 |
8.521942 |
0.0623415259 |
-4.003663 |
1.22910137 |
11.315244 |
0.00076873351106802932739686662344524847867432981729507446289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0036455008843674419939662190159879173734225332736968994140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.00205450536615 |
0.960782991048036 |
16.0736281525814 |
8.22761373865751 |
| ENSMUSG00000040543 |
327958 |
Pitpnm3 |
protein_coding |
Q3UHE1 |
FUNCTION: Catalyzes the transfer of phosphatidylinositol and phosphatidylcholine between membranes (in vitro). Binds calcium ions (By similarity). {ECO:0000250}. |
Alternative splicing;Calcium;Lipid-binding;Membrane;Metal-binding;Phosphoprotein;Reference proteome |
|
|
mmu:327958; |
cell body [GO:0044297]; cell projection [GO:0042995]; COPII-coated ER to Golgi transport vesicle [GO:0030134]; cytoplasm [GO:0005737]; endomembrane system [GO:0012505]; membrane [GO:0016020]; calcium ion binding [GO:0005509]; lipid binding [GO:0008289]; phospholipase activity [GO:0004620]; phospholipid transporter activity [GO:0005548]; receptor tyrosine kinase binding [GO:0030971] |
Human_homologues 17377520_Our finding on the first mutation in the human homologue of Drosophila rdgB indicates novel pathways and a potential important role of the PITPNM3 in mammalian phototransduction. 18188949_Mutation in the PYK2-binding domain of PITPNM3 causes autosomal dominant cone dystrophy (CORD5) in two Swedish families. 20590364_Thus, mutations in PITPNM3 do not appear to be a major cause of cone or cone-rod dystrophy. 21481794_CCL18 derived from Tumor-associated macrophages (TAMs) lays a critical role in promoting breast cancer metastasis via its receptor, PITPNM3. 22405330_Our observations of the PITPNM3 p.Q626H mutation carriers confirm that CORD5 is a disease not to mix with other retinal degenerations mapped to 17p13. 24001613_Data indicate that PYK2 N-terminal domain interacting receptor 1 (Nir1) could induce epithelial-mesenchymal transition by stabilising Snail via the PI3K/Akt/GSK3beta/Snail signalling pathway through binding to CCL18. 26449829_CCL18 enhances hepatocellular carcinoma (HCC) cell migration, invasion, and epithelial-mesenchymal transition (EMT) through the expression of PITPNM3 and the activation of the NF-kappaB signaling pathway. 26756176_CCL18 can increase the invasive ability of non-small cell lung cancer cells by binding to its receptor Nir1. 31955176_Mitofusin-2 (Mfn-2) Might Have Anti-Cancer Effect through Interaction with Transcriptional Factor SP1 and Consequent Regulation on Phosphatidylinositol Transfer Protein 3 (PITPNM3) Expression. 32641093_CCL18-NIR1 promotes oral cancer cell growth and metastasis by activating the JAK2/STAT3 signaling pathway. 35041514_Chemokine CCL18 Promotes Phagocytosis Through Its Receptor CCR8 Rather than PITPNM3 in Human Microglial Cells. |
ENSG00000091622 |
PITPNM3 |
31.821333 |
0.2499517881 |
-2.000278 |
0.53567332 |
13.643690 |
0.00022098269292056388324839433234814123352407477796077728271484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0012107943807791472085044759410266124177724123001098632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.6908867948016 |
4.06137111154963 |
42.7717956102297 |
11.1450746445547 |
| ENSMUSG00000040605 |
56175 |
Bace2 |
protein_coding |
A0A0R4J0I8 |
Human_homologues FUNCTION: Responsible for the proteolytic processing of the amyloid precursor protein (APP). Cleaves APP, between residues 690 and 691, leading to the generation and extracellular release of beta-cleaved soluble APP, and a corresponding cell-associated C-terminal fragment which is later released by gamma-secretase. It has also been shown that it can cleave APP between residues 671 and 672 (PubMed:10591213, PubMed:11083922, PubMed:11423558, PubMed:15857888, PubMed:16816112). Involved in the proteolytic shedding of PMEL at early stages of melanosome biogenesis. Cleaves PMEL within the M-beta fragment to release the amyloidogenic PMEL luminal fragment containing M-alpha and a small portion of M-beta N-terminus. This is a prerequisite step for subsequent processing and assembly of PMEL fibrils into amyloid sheets (PubMed:23754390). Responsible also for the proteolytic processing of CLTRN in pancreatic beta cells (PubMed:21907142). {ECO:0000269|PubMed:10591213, ECO:0000269|PubMed:11083922, ECO:0000269|PubMed:11423558, ECO:0000269|PubMed:15857888, ECO:0000269|PubMed:16816112, ECO:0000269|PubMed:21907142, ECO:0000269|PubMed:23754390}. |
Aspartyl protease;Disulfide bond;Endosome;Hydrolase;Membrane;Protease;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the peptidase A1 family of aspartic proteases. The encoded preproprotein undergoes proteolytic processing to generate an active endopeptidase enzyme. This transmembrane protease catalyzes the proteolysis of amyloid precursor protein to produce amyloid beta peptide. Mice lacking the encoded product exhibit increased pancreatic beta cell mass and improved glucose tolerance due to increased insulin secretion. [provided by RefSeq, Jul 2016]. |
mmu:56175; |
dense core granule [GO:0031045]; endosome [GO:0005768]; Golgi apparatus [GO:0005794]; melanosome membrane [GO:0033162]; plasma membrane [GO:0005886]; aspartic-type endopeptidase activity [GO:0004190]; astrocyte activation [GO:0048143]; glucose homeostasis [GO:0042593]; melanosome organization [GO:0032438]; membrane protein ectodomain proteolysis [GO:0006509]; negative regulation of amyloid precursor protein biosynthetic process [GO:0042985]; protein processing [GO:0016485] |
15987683_Data show that BACE1 knockout mice display a complex phenotype, and a combined deficiency of BACE2 and BACE1 enhanced the bace1-/- lethality. 17307738_BACE1 and BACE2 may act as alternative alpha-secretase-like proteases in proteolytic processing of IL-1R2 and APP 19117266_The presence of BACE2 in secretory granules of beta cells suggests that it may play a role in diabetes-associated amyloidogenesis. 20596738_The TgBACE2-APP mice showed deregulation of BACE2 expression levels that were significantly increased with respect to single TgBACE2 mice. 21907142_Bace2 is a beta cell-enriched protease that regulates pancreatic beta cell function and mass. 22628310_Tmem27 dimerization is a dynamic process involving Bace2 23430253_non-redundant roles of BACE1/2 in ectodomain shedding with BACE1 regulating a broader and BACE2 a more distinct set of beta-cell-enriched substrates including two proteins of the seizure 6 protein family (SEZ6L and SEZ6L2). 23754390_the BACE1 homologue BACE2 processes PMEL to generate functional amyloids. BACE2 is highly expressed in pigment cells and Bace2(-/-) but not Bace1(-/-) mice display coat color defects, implying a specific role for BACE2 during melanogenesis 28337562_The absence of BACE2 ameliorates glucose tolerance defects induced by IAPP overexpression in the beta-cell and promotes beta-cell survival. 34015524_BACE2 suppression in mice aggravates the adverse metabolic consequences of an obesogenic diet. |
ENSG00000182240 |
BACE2 |
25.190623 |
0.2188155546 |
-2.192213 |
0.77256699 |
6.210334 |
0.01270066674300004798403573857967785443179309368133544921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0412223643361126629303115009861357975751161575317382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.943442059385 |
6.14436892524904 |
50.0121761394767 |
19.8311464881485 |
| ENSMUSG00000040613 |
11810 |
Apobec1 |
protein_coding |
P51908 |
FUNCTION: Cytidine deaminase catalyzing the cytidine to uridine postranscriptional editing of a variety of mRNAs. Form complexes with cofactors that confer differential editing activity and selectivity. Responsible for the postranscriptional editing of a CAA codon for Gln to a UAA codon for stop in the apolipoprotein B mRNA. Also involved in CGA (Arg) to UGA (Stop) editing in the NF1 mRNA (By similarity). May also play a role in the epigenetic regulation of gene expression by participating in DNA demethylation (PubMed:21496894). {ECO:0000250|UniProtKB:P41238, ECO:0000269|PubMed:21496894}. |
Cytoplasm;Hydrolase;Metal-binding;mRNA processing;Nucleus;Reference proteome;Zinc |
|
|
mmu:11810; |
apolipoprotein B mRNA editing enzyme complex [GO:0030895]; cytoplasm [GO:0005737]; mRNA editing complex [GO:0045293]; nucleus [GO:0005634]; cytidine deaminase activity [GO:0004126]; cytosine deaminase activity [GO:0004131]; enzyme activator activity [GO:0008047]; mRNA 3'-UTR AU-rich region binding [GO:0035925]; mRNA binding [GO:0003729]; protein domain specific binding [GO:0019904]; ribonucleoprotein complex binding [GO:0043021]; RNA binding [GO:0003723]; zinc ion binding [GO:0008270]; cytidine to uridine editing [GO:0016554]; defense response to virus [GO:0051607]; DNA cytosine deamination [GO:0070383]; DNA demethylation [GO:0080111]; establishment of localization in cell [GO:0051649]; lipoprotein biosynthetic process [GO:0042158]; lipoprotein metabolic process [GO:0042157]; lipoprotein transport [GO:0042953]; mRNA modification [GO:0016556]; mRNA processing [GO:0006397]; mRNA stabilization [GO:0048255]; negative regulation of DNA methylation-dependent heterochromatin formation [GO:0090310]; negative regulation of nuclear-transcribed mRNA catabolic process, nonsense-mediated decay [GO:2000623]; negative regulation of triglyceride metabolic process [GO:0090209]; positive regulation of DNA demethylation [GO:1901537]; positive regulation of mRNA modification [GO:0090366]; regulation of cell population proliferation [GO:0042127]; response to calcium ion [GO:0051592]; response to ethanol [GO:0045471]; response to gamma radiation [GO:0010332]; response to osmotic stress [GO:0006970]; response to xenobiotic stimulus [GO:0009410]; triglyceride metabolic process [GO:0006641] |
12816761_studies establish the existence of preferential degradation of intestinal apolipoprotein B-100 and subtle defects in triglyceride secretion in apolipoprotein B editing complex 1-/- mice 12896982_apobec-1 complementation factor has a role in regulating nucleocytoplasmic import and shuttling 14559896_apobec-1-mediated apoB mRNA editing is regulated by BAG-4 14559972_RNA editing of this protein is unable to induce somatic hypermutation in mammalian cells. 15480992_Lipopolysaccharide increases intestinal stem cell survival through apobec-1-mediated regulation of cyclooxygenase-2 messenger RNA stability. 17875695_Deletion of apobec-1, by modulating expression of AU-rich RNA targets, provides an important mechanism for attenuating a dominant genetic restriction point in intestinal adenoma formation. 18983852_Murine APOBEC1 is a hypermutator of both RNA and single-stranded DNA in vivo, which could exert occasional side effects upon overexpression. 19386592_the AU-rich RNA binding-protein Apobec-1 mediates post-transcriptional regulation of Cyp7a1 expression and influences susceptibility to diet-induced gallstone formation 20667539_LDL receptor and the apolipoprotein B mRNA editing enzyme Apobec1 are regulated via calcium signaling in mechanistic response to genetic, mechanical, and environmental insults that trigger an imbalance of intracellular calcium homeostasis 21258325_The transcriptomics approach to RNA editing presented in this study dramatically expands the list of APOBEC1 mRNA editing targets and reveals a novel cellular mechanism for the modification of transcript 3' UTRs. 22923694_Results suggest that apo B mRNA editing protein (Apobec1 cytidine deaminase) plays a central role in controlling testicular germ cell tumors susceptibility in both a conventional and a transgenerational manner. 22993231_The transgenic rescue of intestinal apobec-1 expression restores C-to-U RNA editing of apoB mRNA in vivo, including the canonical site at position 6666 and also at approximately 20 other newly identified downstream sites present in WT mice. 24249727_Individual genetic variability at the Apobec1 locus results in differential rates of C-to-U(T) editing in murine macrophages; with mouse strains expressing mostly a truncated alternative transcript isoform of Apobec1 exhibiting lower rates of editing. 24916387_RBM47 and APOBEC1 constitute the basic machinery for C to U RNA editing. 24946870_Apobec-1-dependent C-to-U RNA editing exerts broad functional effects in a tissue-specific manner. 25303118_In contrast to in vitro results, APOBEC1 neither inhibited nor significantly drove the molecular evolution of Friend retrovirus in wild type or APOBEC1 knockout mice. 27165291_substantial rearrangement/duplication of transgene elements is present, and transgene integration was accompanied by the deletion of a 19,500 bp fragment of genomic DNA that contains the promoter, exon 1 and part of intron 1 of the APOBEC1 complementation factor (A1cf) gene, as well as several elements that are predicted to regulate chromosomal architecture 27582469_Here, we show that partial loss of either APOBEC1 complementation factor (A1CF), the RNA-binding cofactor of APOBEC1 in RNA editing, or Argonaute 2 (AGO2), a key factor in the biogenesis of certain noncoding RNAs, modulates risk for TGCTs and testicular abnormalities in both parent-of-origin and conventional genetic manners. 28069890_The observations demonstrate that A1CF does not act as the APOBEC1 complementation factor in vivo under normal physiological conditions and suggest new roles for A1CF, specifically within the male adult kidney. 29167375_APOBEC1-mediated RNA editing occurs within brain microglia cells and is key to maintaining their resting status. Apobec1-/- mice display age-related signs of CNS neurodegeneration. 31726973_At an experimental level mouse APOBEC1 is remarkable among 12 mammalian A1 enzymes in that it represents a source of somatic mutations in mouse genome, potentially fueling oncogenesis. |
ENSG00000111701 |
APOBEC1 |
330.318349 |
2.0087960726 |
1.006331 |
0.16118735 |
38.748898 |
0.00000000048198225408285350711831667902995852970082779620497603900730609893798828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000070254102336746651169869815620541708689472670812392607331275939941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
440.648057421209 |
49.3345810317394 |
219.359278645137 |
18.9006626239639 |
| ENSMUSG00000040624 |
213783 |
Plekhg1 |
protein_coding |
F7CES2 |
|
Reference proteome |
|
|
|
nucleoplasm [GO:0005654] |
Human_homologues 19165232_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20641033_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 29967039_PLEKHG1 rs9478812 variant substantially increases risk of preeclampsia. 30659137_Genetic variation in PLEKHG1 is associated with white matter hyperintensities and ischemic stroke, most strongly with the small vessel subtype, suggesting it acts by promoting small vessel arteriopathy. |
ENSG00000120278 |
PLEKHG1 |
357.993213 |
2.7276948526 |
1.447682 |
0.17606388 |
66.568608 |
0.00000000000000033792499429751494086832863993661087851461016545902282803837124447454698383808135986328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000095496725661219802568187910605401695477746637497062920374446548521518707275390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
533.507246126146 |
65.2699892122677 |
195.589050445529 |
18.5494785173378 |
| ENSMUSG00000040653 |
76142 |
Ppp1r14c |
protein_coding |
Q8R4S0 |
FUNCTION: Inhibitor of the PP1 regulatory subunit PPP1CA. {ECO:0000269|PubMed:11812771}. |
Acetylation;Membrane;Methylation;Phosphoprotein;Protein phosphatase inhibitor;Reference proteome |
|
|
mmu:76142; |
cytoplasm [GO:0005737]; endomembrane system [GO:0012505]; membrane [GO:0016020]; protein serine/threonine phosphatase inhibitor activity [GO:0004865]; regulation of phosphorylation [GO:0042325]; signal transduction [GO:0007165] |
11812771_PPP1R14C gene encodes a PKC-potentiated inhibitory protein, KEPI, for type-1 Ser/Thr protein phosphatase (PP1). KEPI mRNA and protein are enriched in brain, heart and skeletal muscle. 11812771_regulation by morphine 15837133_The expression patterns point to possible roles for KEPI in regulating protein dephosphorylation by inhibiting PP1 activities in a number of brain pathways. 27333042_the data presented in this manuscript demonstrate a critical function of VPS35 in regulating PTH1R trafficking. This event and VPS35-interaction with PPP1R14C appear to be essential for turning off PTH1R's endosomal signaling, and promoting PTH1R-mediated catabolic response and bone remodeling. |
ENSG00000198729 |
PPP1R14C |
103.261033 |
0.4456970930 |
-1.165865 |
0.26943199 |
18.666464 |
0.00001556969614685464563383007763341936424694722518324851989746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001113938993465899253768047749701963766710832715034484863281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
62.8962566073476 |
11.8146546723437 |
141.118839657085 |
19.0384029062128 |
| ENSMUSG00000040658 |
381101 |
Dnph1 |
protein_coding |
Q80VJ3 |
FUNCTION: Catalyzes the cleavage of the N-glycosidic bond of deoxyribonucleoside 5'-monophosphates to yield deoxyribose 5-phosphate and a purine or pyrimidine base. Deoxyribonucleoside 5'-monophosphates containing purine bases are preferred to those containing pyrimidine bases. {ECO:0000255|HAMAP-Rule:MF_03036}. |
Acetylation;Cytoplasm;Glycosidase;Hydrolase;Nucleotide metabolism;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:381101; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; deoxyribonucleoside 5'-monophosphate N-glycosidase activity [GO:0070694]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803]; allantoin metabolic process [GO:0000255]; amide catabolic process [GO:0043605]; deoxyribonucleoside monophosphate catabolic process [GO:0009159]; dGMP catabolic process [GO:0046055]; epithelial cell differentiation [GO:0030855]; nucleoside metabolic process [GO:0009116]; positive regulation of cell growth [GO:0030307] |
Human_homologues 18726892_characterization of the human Rcl gene, we cloned its promoter, Rcl is a bona fide target gene of ETV1. 20962348_analysis of human deoxynucleotide N-hydrolase Rcl and rat gene c6orf108 33833118_Targeting the nucleotide salvage factor DNPH1 sensitizes BRCA-deficient cells to PARP inhibitors. |
ENSG00000112667 |
DNPH1 |
223.332788 |
2.7048059205 |
1.435525 |
0.17281240 |
68.378702 |
0.00000000000000013492700417101886402475980971444546944965563834914479279802890232531353831291198730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000039675832577856357640561558850893206851215343038130622943526759627275168895721435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
335.584552839365 |
40.2940011721564 |
124.069734650853 |
11.6729240165567 |
| ENSMUSG00000040666 |
50795 |
Sh3bgr |
protein_coding |
Q9WUZ7 |
|
Reference proteome;SH3-binding |
|
|
mmu:50795; |
SH3 domain binding [GO:0017124] |
Human_homologues 20532202_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 27100087_RNA-seq evidence of biallelic expression of SH3BGR and 10 neighboring genes in at least one primary human tissue tested indicates that the expression of SH3BGR is uncoupled from the control of the maternally inherited 5mCpG imprints at the WRB differentially methylated region (DMR) in disomic controls or trisomy (Down syndrome) individuals. |
ENSG00000185437 |
SH3BGR |
45.691343 |
3.4773934250 |
1.798006 |
0.43973796 |
16.387332 |
0.00005162912603433548460818772185554337283974746242165565490722656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003300104270898019396054956509090061445022001862525939941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
71.6711385796132 |
22.1854222241392 |
20.6105924237204 |
5.04690508612988 |
| ENSMUSG00000040701 |
11766 |
Ap1g2 |
protein_coding |
O88512 |
FUNCTION: May function in protein sorting in late endosomes or multivesucular bodies (MVBs). Involved in MVB-assisted maturation of hepatitis B virus (HBV). {ECO:0000250|UniProtKB:O75843}. |
Cytoplasmic vesicle;Endosome;Golgi apparatus;Membrane;Protein transport;Reference proteome;Transport |
|
This gene encodes the gamma-2 subunit of the adaptor protein complex 1 (AP-1). AP-1 complex is a heterotetramer comprised of two heavy and one each of medium and small subunits. The encoded protein is a heavy subunit of AP-1 complex that regulates polarized sorting of cargo at the trans-Golgi network and endosomes. Alternative splicing of this gene results in multiple transcript variants. [provided by RefSeq, Jan 2015]. |
mmu:11766; |
AP-1 adaptor complex [GO:0030121]; endosome membrane [GO:0010008]; Golgi membrane [GO:0000139]; Golgi-associated vesicle [GO:0005798]; membrane [GO:0016020]; trans-Golgi network [GO:0005802]; transport vesicle [GO:0030133]; cargo adaptor activity [GO:0140312]; clathrin adaptor activity [GO:0035615]; Golgi to vacuole transport [GO:0006896]; intracellular protein transport [GO:0006886]; vesicle-mediated transport [GO:0016192] |
Human_homologues 17553870_These results demonstrate that HBV exploits the multivesicular bodies machinery with the aid of gamma 2-adaptin. 18772139_gamma2-adaptin's ubiquitin-interacting motif mediates a specific physical interaction with the ubiquitin ligase Nedd4 and promotes ubiquitination of gamma2-adaptin 20708039_Data show that gamma2-adaptin in MVB sorting specifically interacts with the ESCRT subunits Vps28 and CHMP2A. 27909244_Depletion of the gamma2 or mu1A (AP1M1) subunits of AP-1, but not of gamma1 (AP1G1), precludes Nef-mediated lysosomal degradation of CD4. |
ENSG00000213983 |
AP1G2 |
60.941854 |
5.2603872287 |
2.395169 |
0.39570680 |
35.490246 |
0.00000000256330988776619507302133555792875846401912554028967861086130142211914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000340107458279221972350337083266375026724404051492456346750259399414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
106.604104461741 |
29.6077039921246 |
20.2654481175901 |
4.42162398603423 |
| ENSMUSG00000040855 |
194590 |
Reps2 |
protein_coding |
Q80XA6 |
FUNCTION: Involved in ligand-dependent receptor mediated endocytosis of the EGF and insulin receptors as part of the Ral signaling pathway (By similarity). By controlling growth factor receptors endocytosis may regulate cell survival (By similarity). Through ASAP1 may regulate cell adhesion and migration (By similarity). {ECO:0000250|UniProtKB:Q8NFH8}. |
Calcium;Coiled coil;Cytoplasm;Metal-binding;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:194590; |
cytoplasm [GO:0005737]; calcium ion binding [GO:0005509]; endocytosis [GO:0006897]; endosomal transport [GO:0016197] |
Human_homologues 12149250_These results suggest that POB1 interacts with PAG2 through its proline-rich motif, thereby regulating cell migration. 12771942_POB1, through its influence on the Ral signalling pathway, is involved in growth factor signalling and consequently in control of cell proliferation 15184881_decreased expression of REPS2 might be a key factor, causing prostate cancer cells to become resistant to induction of apoptosis by androgen deprivation. 15455380_Decreased REPS2 expression is associated with androgen-independent state of advanced prostate cancer 15707977_These results show for the first time that POB1 can regulate the transport function of RLIP76 and are consistent with our previous studies showing that inhibition of RLIP76 induces apoptosis in cancer cells. 18474607_Hsf-1 causes specific and saturable inhibition of the transport activity of Ralbp1 and that the combination of Hsf-1 and POB1 causes nearly complete inhibition through specific bindings with Ralbp1. 19776672_REPS2 may be a useful tumor marker for favorable prognosis in breast cancer. 23803043_downregulation of REPS2 may contribute to malignant progression of esophageal squamous cell carcinoma and represent a novel prognostic marker and a potential therapeutic target for esophageal squamous cell carcinoma patients. 27120794_miR-675-5p might play an oncogenic role in esophageal squamous cell carcinoma (ESCC) through RalBP1/RAC1/CDC42 signaling pathway by inhibiting REPS2 and might serve as a valuable prognostic biomarker and therapeutic target for ESCC patients. 28579616_HIV-1 gp41 promotes viral endocytosis in a CD4-independent manner by interacting with the host protein POB1. 35690292_REPS2 downregulation facilitates FGF-induced adhesion and migration in human lens epithelial cells through FAK/Cdc42 signaling and contributes to posterior capsule opacification. |
ENSG00000169891 |
REPS2 |
154.281234 |
0.2409370825 |
-2.053272 |
0.30095088 |
44.948950 |
0.00000000002022385908383634527159326592408116179261368117181518755387514829635620117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000003484726488291800792803201430468309357513234658654255326837301254272460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
56.2828792992307 |
11.8261278158037 |
233.599903812794 |
34.8628662835046 |
| ENSMUSG00000040867 |
380785 |
Begain |
protein_coding |
Q68EF6 |
FUNCTION: May sustain the structure of the postsynaptic density (PSD). |
Acetylation;Cytoplasm;Membrane;Methylation;Phosphoprotein;Reference proteome |
|
|
mmu:380785; |
cytoplasm [GO:0005737]; dendrite [GO:0030425]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; postsynapse [GO:0098794]; presynapse [GO:0098793]; synapse [GO:0045202]; evoked excitatory postsynaptic potential [GO:0098817]; regulation of postsynaptic neurotransmitter receptor activity [GO:0098962] |
19641963_A Begain transcript variant is preferentially expressed from the paternal allele. 27785460_BEGAIN was highly localized at the synapse of inner lamina II in the spinal dorsal horn and that its expression was up-regulated after peripheral nerve injury in wild-type, but not in GluN2B Y1472F-KI, mice. Mechanical allodynia was significantly attenuated in BEGAIN-deficient mice, but no change was observed in terms of physiological threshold for mechanical stimuli. |
ENSG00000183092 |
BEGAIN |
103.306014 |
2.3129306716 |
1.209722 |
0.31208381 |
14.558356 |
0.00013588467126073863278437614532379029697040095925331115722656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007881931883638883641965411364083138323621824383735656738281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
145.437105234468 |
31.7068969986135 |
62.8800106366106 |
10.500361160607 |
| ENSMUSG00000040943 |
214133 |
Tet2 |
protein_coding |
Q4JK59 |
FUNCTION: Dioxygenase that catalyzes the conversion of the modified genomic base 5-methylcytosine (5mC) into 5-hydroxymethylcytosine (5hmC) and plays a key role in active DNA demethylation. Has a preference for 5-hydroxymethylcytosine in CpG motifs. Also mediates subsequent conversion of 5hmC into 5-formylcytosine (5fC), and conversion of 5fC to 5-carboxylcytosine (5caC). Conversion of 5mC into 5hmC, 5fC and 5caC probably constitutes the first step in cytosine demethylation. Methylation at the C5 position of cytosine bases is an epigenetic modification of the mammalian genome which plays an important role in transcriptional regulation. In addition to its role in DNA demethylation, also involved in the recruitment of the O-GlcNAc transferase OGT to CpG-rich transcription start sites of active genes, thereby promoting histone H2B GlcNAcylation by OGT. {ECO:0000269|PubMed:20639862, ECO:0000269|PubMed:21057493, ECO:0000269|PubMed:21778364, ECO:0000269|PubMed:21817016, ECO:0000269|PubMed:23352454, ECO:0000269|PubMed:23353889}. |
Alternative splicing;Cell cycle;Chromatin regulator;Dioxygenase;DNA-binding;Glycoprotein;Iron;Metal-binding;Oxidoreductase;Phosphoprotein;Reference proteome;Zinc |
|
|
mmu:214133; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; ferrous iron binding [GO:0008198]; methylcytosine dioxygenase activity [GO:0070579]; zinc ion binding [GO:0008270]; 5-methylcytosine catabolic process [GO:0006211]; 5-methylcytosine metabolic process [GO:0019857]; cell cycle [GO:0007049]; chromatin organization [GO:0006325]; cytosine metabolic process [GO:0019858]; DNA demethylation [GO:0080111]; hematopoietic stem cell homeostasis [GO:0061484]; hemoglobin metabolic process [GO:0020027]; hemopoiesis [GO:0030097]; homeostasis of number of cells [GO:0048872]; kidney development [GO:0001822]; leukocyte differentiation [GO:0002521]; liver morphogenesis [GO:0072576]; myeloid cell differentiation [GO:0030099]; myeloid progenitor cell differentiation [GO:0002318]; oxidative demethylation [GO:0070989]; oxidative DNA demethylation [GO:0035511]; positive regulation of transcription by RNA polymerase II [GO:0045944]; post-embryonic development [GO:0009791]; protein O-linked glycosylation [GO:0006493]; spleen development [GO:0048536] |
16722336_this gene may be involved in hormone secretion 20639862_all three mouse Tet proteins (Tet1, Tet2 and Tet3) can also catalyse conversion of 5-methylcytosine of DNA to 5-hydroxymethylcytosine 21057493_results demonstrate that Tet2 is important for normal myelopoiesis 21295276_Tet1 and Tet2 regulate 5-hydroxymethylcytosine production and cell lineage specification in mouse embryonic stem cells. 21723200_Tet2 haploinsufficiency contributes to hematopoietic transformation in vivo. 21723201_Impairment of TET2 function or/and expression predisposes to the development of hematological malignancies. 21778364_in addition to 5-hydroxymethylcytosine (5hmC), Tet proteins can generate 5-formylcytosine and 5-carboxylcytosine from 5mC in an enzymatic activity-dependent manner; DNA demethylation may occur through Tet-catalyzed oxidation followed by decarboxylation 21803851_Tet2 functions as a tumor suppressor to maintain hematopoietic cell homeostasis. 21863549_alterations in the expression of the Ayu17-449 gene affect the expression of other related genes 21873190_show here that Tet2 is a critical regulator of self-renewal and differentiation of hematopoietic stem cells 22467095_The Interaction With Bcr-Abl Protein Induces TET2 Cytoplasmatic Compartmentalization and Loss-of-Function. 22469782_Loss of TET2 results in an increase in the number of hematopoietic stem cells (HSCs)/progenitors in the fetal liver 22569366_Hydroxylation of 5-methylcytosine by TET2 maintains the active state of the HOXA cluster. 22902501_findings suggest that Parp1 and Tet2 contribute to an epigenetic program that directs subsequent transcriptional induction at pluripotency loci during somatic cell reprogramming 23028748_Aid is a regulator of the subcellular localization of Tet family proteins. 23074272_Data indicate that TET2-deficient bone marrow-derived mast cells (BMMC) infected with KITD816V could survive for > 3 weeks. 23254757_Our findings indicate that Tet2 are crucial downstream targets of the pluripotency factor Oct4, and highlight a role for Oct4 in the regulation of DNA methylation in ESCs. 23352454_Tet1 and Tet2 are stable partners of Ogt in the nucleus of embryonic stem cells. 23352810_combined Tet1 and Tet2 deficiency results in partially penetrant embryonic and neonatal abnormalities associated with perinatal lethality of about half of the mutants 23395962_find physical association and reprogramming synergy of TET2 with NANOG, and demonstrate that knockdown of TET2 abolishes the reprogramming synergy of NANOG with a catalytically deficient mutant of TET1 23415914_Tet1 and Tet2 have a locus-specific role in shaping the primordial germ cells epigenome during subsequent development. 23453809_Tet1 and Tet2 proteins have discrete roles in cell-fusion-mediated pluripotent reprogramming and imprint erasure in somatic cells. 23548903_Ascorbate enhances 5-hydroxymethylcytosine generation, most likely by acting as a co-factor for Tet methylcytosine dioxygenases. 23563267_IDAX depletion prevents TET2 downregulation in differentiating mouse embryonic stem cells 23926181_Aldosterone stimulates the dispersal of DNA methyltransferase (DNMT3b) and recruitment of Tet2 to demethylate the alphaENaC promoter in kidney collecting duct cells, inducing alpha-epithelial Na+ channel (alphaENaC) transcription. 24077167_We identify TET2 as a novel and necessary master epigenetic regulator of smooth muscle cell differentiation. 24218139_Deletion of Ezh2 alone was enough to induce MDS/MPN-like diseases. Furthermore, concurrent depletion of Ezh2 and Tet2 established more advanced myelodysplasia and markedly accelerated the development of myelodysplastic disorders. 24335252_PRDM14 physically interacts with TET1 and TET2 and enhances the recruitment of TET1 and TET2 at target loci. Knockdown of TET1 and TET2 impaired transcriptional regulation and DNA demethylation by PRDM14. 24474761_data point to a complex interplay between Tet1 and Tet2 in mESC, and to distinct roles for these two proteins in regulating promoter, exon, and polyadenylation site usage in cells 24645644_expression of Dnmt3a, Dnmt3b and Tet2 declines significantly with skin ageing 24672749_Coordination of engineered factors with TET1/2 promotes early-stage epigenetic modification during somatic cell reprogramming. 24788138_Data indicate the cooperation between receptor tyrosine kinase KIT D816V mutation and loss of function of tet oncogene 2 (TET2) in mast cell transformation in systemic mastocytosis (SM). 24838012_Tet oxidizes thymine to 5-hydroxymethyluracil in mouse embryonic stem cell DNA. 24844363_Findings indicate a novel type of hematological malignancy induced by tet oncogene 2 protein (Tet2) loss. 24984151_Collectively, our results suggest that up-regulation of Tet2 is required for Foxp3 stability and IL2 is required to maintain the high level of Tet2 during the thymic Treg development. 25263596_deletion of Tet2 causes extensive loss of 5-hydroxymethylcytosine at enhancers, accompanied by enhancer hypermethylation, reduction of enhancer activity, and delayed gene induction in the early steps of differentiation. 25281607_This work provides insight into the functional consequences of JAK2V617F-TET2 comutation in myeloproliferative neoplasms 25395421_loss of TET2 has two different roles in myeloproliferative neoplasms: disease accelerator and disease initiator and sustainer in combination with JAK2V617F. 25501021_These epigenetic changes, seen in Tet2 insufficiency-triggered lymphoma, possibly contributed to predated outgrowth of Tfh-like cells and subsequent lymphomagenesis. 25608811_TET1, TET2 and TET3 mediated DNA demethylation and played role in mouse development.[review] 25862091_Tet2 promotes DNA demethylation and activation of cytokine gene expression in T cells. 25886910_effect of Tet2 loss in preleukemic hematopoietic cells is progressive and widespread DNA hypermethylation affecting up to 25% of active enhancer elements. 25915124_SIRT6 functions as a chromatin regulator safeguarding the balance between pluripotency and differentiation through Tet-mediated production of 5-hydroxymethylcytosine. 26231219_Tet2 was responsible for increase in 5-hydroxymethylcytosine abundance after ischemic injury. 26275994_reduced expression leads to Foxp3 hypermethylation and impairment of Treg-cell-associated immune homeostasis 26287468_mechanistic evidence for the gene-specific transcription repression activity of Tet2 via histone deacetylation and for the prevention of constant transcription activation at the chromatin level for resolving inflammation 26325469_Both TET1 and TET2 are required for the repression of embryonic stem cells differentiation by PRDM14. 26328763_Reprogramming of spermatogonial stem cells from Tet1 and Tet2 double knockout mice however lacked demethylation of H19 imprinting control regions. 26378518_our results indicate that neuronal TET2 is positively involved in the regulation of cell survival. 26493943_the results showed that low shear stress downregulated endothelial cell autophagy by impaired TET2 expression, which might contribute to the atherogenic process. 26586431_The critical roles of TET1/2 individually. 26598602_These results uncover the hypermethylation of DNA methylation canyons as the genomic key feature of Tet1/Tet2 double-knockout mouse embryonic fibroblasts. 26607761_Acute elimination of TET function induces the rapid development of an aggressive, fully-penetrant and cell-autonomous myeloid leukaemia in mice, pointing to a causative role for TET loss-of-function in this myeloid malignancy. 26620559_UNG might be involved in Tet-mediated DNA demethylation. 26876596_confirm the transformation potential of DNMT3A(R882H) Tet2(-/-) progenitors and represent the first cooperative model in mice involving Tet2 inactivation driving lymphoid malignancies 26903244_the stability of Foxp3 expression is markedly compromised in T reg cells from Tet2/Tet3 double-deficient mice. 27003514_TET2 requires its catalytic activity in hematopoietic stem/progenitor cells to exert its myeloid malignancy-suppressive function 27160912_in the absence of TET2, mast cell differentiation is under the control of compensatory mechanisms mediated by other TET family members, while proliferation is strictly dependent on TET2 expression. 27184841_Tet enzymes play important roles in telomere maintenance and chromosomal stability of embryonic stem cells by modulating sub-telomeric methylation levels. 27288448_this study demonstrates the critical role of TET proteins in regulating the crosstalk between two key epigenetic mechanisms. 27289557_The data suggest that TET2 activity and the levels of 5-hydroxymethylcytosine and its derivatives should be tightly controlled to avoid genetic and chromosomal instabilities. 27345836_Zfp281 interacts with Tet1, but not Tet2, and its direct transcriptional target, miR-302/367, to negatively regulate Tet2 expression to establish and maintain primed pluripotency. 27362828_Iron(II)/alpha-Ketoglutarate-Dependent Catalytic Domains of the TET Enzymes 27384303_tet2 activity in GnRH neurons has influence over the neuroendocrine control of male reproductive function 27428748_Loss of TET2 expression is associated with development of malignancy. 27778125_TET1 and TET2 play an important role in the proliferation of neural stem cells in the adult mouse brain. 27821816_we identified that TET2 was downregulated during the pathogenesis of atherosclerosis. The downregulation of TET2 promotes the methylation of the Beclin 1 promoter, leading to endothelial cell autophagy, impaired autophagic flux, and inflammatory factor upregulation. 27863519_Our data implicate TET enzymes ( TET1 and TET2 )in the evolutionary dynamics of TEs, both in the context of exaptation processes and of retrotransposition control. The dual role of TET action on LINE-1s may reflect the evolutionary battle between TEs and the host 27869616_Here the authors show that concomitant loss of Tet2 and Tet3 in mice at early B cell stage blocked the pro- to pre-B cell transition in the bone marrow, decreased Irf4 expression and impaired the germline transcription and rearrangement of the Igkappa locus. 27869820_this study shows that simultaneous deletion of Tet2 and Tet3 in mouse CD4+CD8+ double-positive thymocytes results in dysregulated development and proliferation of invariant natural killer T cells 27930333_Tet1/2/3 proteins have a key role in modulating Wnt signaling and establishing the proper balance between neural and mesodermal cell fate determination in mouse embryos and embryonic stem cells 28077417_indicate that AID and TET2 share common effects on myeloid and erythroid lineage differentiation, however, their role is nonredundant in regulating HSC self-renewal and in myeloid transformation. 28100914_TET proteins, particularly TET2, were required for adipogenesis by modulating DNA methylation at the Ppargamma locus, subsequently by inducing Ppargamma gene expression. 28104796_These results support the hypothesis that somatic TET2 mutations in blood cells play a causal role in atherosclerosis. 28167288_TET2 plays a role in the iron and heme metabolism in erythroblasts. 28192139_the effects of vitamin C inhibiting the apo(a) expression were attenuated by ELK1siRNA and Tet2siRNA. These results suggested vitamin C down-regulate apo(a) expression via Tet2-dependent DNA demethylation in HepG2 cells 28272491_these results indicate that Tet2 is involved in myoblast differentiation through promoting DNA demethylation and myogenic gene expression. 28440315_Tet2-/- mice develop spontaneous myeloid, T- and B-cell malignancies after long latencies with higher mutation frequencies at genomic sites that gained 5-hydroxymethylcytosine, where Tet2 normally binds. 28538185_These findings suggest that the catalytic and scaffolding activities of TDG are essential for retinoic acid-dependent gene expression and provide important insights into the mechanisms underlying targeting of TET-TDG complexes. 28660881_Foxo3a physically interacts with Tet2 and regulates the expression of genes related to adult neural stem cells proliferation. 28697999_This work revealed a critical role of TET2-mediated DNA demethylation against oxidative stress, and provided the molecular mechanisms underlying the epigenetic regulation of iron homeostasis in response to stress erythropoiesis. 28813659_ablation of Tet2 in myeloid cells suppresses melanoma growth in vivo and shifted the immunosuppressive gene expression program in tumor-associated macrophages to a proinflammatory one 28826859_Tet methylcytosine dioxygenase 2 (TET2) was the most highly expressed Tet enzyme in murine macrophage (MPhi) differentiation. 29089070_TET2 has the effect of inhibiting LPS-induced macrophage activation and plays an inhibitory role in macrophage M1 and M2 polarization. 29150566_this article demonstrates that TET2 regulates CD8+ T cell differentiation 29195897_this is the first evidence to suggest that TET2 mutations promote clonal dominance with aging by conferring TNFalpha resistance to sensitive bone marrow progenitors while also propagating such an inflammatory environment. 29275866_Tet2/Nras double-mutant leukemia showed preferential sensitivity to MAPK kinase (MEK) inhibition in both mouse model and patient samples 29290626_These results highlight the critical role of TET2 in the maintenance of BMSC functions and osteoblast differentiation and provide evidence that dysregulation of epigenetic modifiers in BMSCs contributes to the progression of myeloid malignancies. 29351814_Results demonstrate a novel and unexpected role for SETDB1 in protecting IAPs from TET2-dependent histone arginine demethylation. 29364877_study reveals a previously unknown regulatory role of Tet2 at the epitranscriptomic level, promoting myelopoiesis during infection in the mammalian system by decreasing 5-mCs in mRNAs; moreover, the inhibitory function of cytosine methylation on double-stranded RNA formation and Adar1 binding in mRNA reveals its new physiological role in the mammalian system 29398449_RHOA G17V expression together with Tet2 loss resulted in development of angioimmunoblastic T cell lymphoma in mice 29466726_These data identify Tet2 as a key molecular mediator of neurogenic rejuvenation. 29467326_Study shows that the bone marrow-derived mast cells hyperproliferation caused by loss of Tet2 is significantly related to hyperactivation of the PI3K pathway and that activating KIT mutation-induced ligand-independent growth utilizes a similar pathway. Cells that express oncogenic KIT and lack TET2 are also dependent on the PI3K pathway for proliferation. 29471939_Tet2 deficiency in hematopoietic cells is associated with greater cardiac dysfunction in murine models of heart failure. 29542811_Suggest a role for TET2 in the pathogenesis of psoriasiform dermatitis. 29581109_our data establish that Tet2 deficiency predisposes to mature B-cell malignancies, which development might be attributed in part to AID-mediated accumulating mutations and BCR-mediated signaling. 29662175_Pluripotency transcription factors and Tet1/2 maintain Brd4-independent stem cell identity in mouse embryonic stem cells. 29705354_TET2 plays an important role in the pathogenesis of diabetic nephropathy by activating TGFbeta1 expression through demethylation of CpG islands in the TGFbeta1 regulatory region 29728415_Tet2 silencing promoted hematopoietic cell expansion and AngII-induced cardiac dysfunction in mice. 29764867_Tet2 epigenetically activated cell-cycle-inhibiting genes p21 and p27. 29769727_findings demonstrate the requirement for microbial-dependent inflammation in the development of pre-leukaemic myeloproliferation (PMP) and provide a mechanistic basis for the variation in PMP penetrance observed in Tet2(-/-) mice; this study will prompt new lines of investigation that may profoundly affect the prevention and management of haematopoietic malignancies 29858571_Results indicate that tet oncogene 1 protein (Tet1) and tet oncogene 2 protein (Tet2) play a critical role in maintaining bone marrow MSCs (BMMSCs) and bone homeostasis through demethylation of P2X7 purinoceptor (P2rX7) to control exosome and miRNA release. 29866713_Oncogenic N-Ras and Tet2 haploinsufficiency collaborate to dysregulate hematopoietic stem and progenitor cells. 30146412_results indicate that restoring TET2 function through SIRT1 activation represents a promising means to target Myelodysplastic syndrome (MDS) hematopoietic stem/progenitor cells (HSPCs) 30193945_Consistent with this, TET2 was required for Rosi-dependent gene activation of certain Rosiglitazone targets accompanied by changes in DNA demethylation at the promoter regions 30220521_Consistent with this finding, ablation of the DNA demethylase Tet2 almost completely abolishes cell reprogramming. 30343588_The melatonin synthesized by Aanat, upregulates Tet2 expression and maintain the normal genomic methylation status, and thereby promote embryonic development. 30538121_Genetic ablation of Tet2 in mice cooperated with oncogenic NRAS(Q61K) to promote melanoma initiation while suppressing specific gains in 5hmC. Authors conclude that TET2 acts as a barrier to melanoma initiation and progression, partly by promoting 5hmC gains in specific gene bodies. 30540950_loss of Tet1 (Tet1 KO) led to resistance to chronic restraint stress (CRS), whereas loss of Tet2 (Tet2 KO) increased the susceptibility of mice to CRS. 30726915_The data indicate that Tet2 and Tet3 are involved in TET on regulatory T cells (Treg cels) stability and immune homeostasis in mice. 30796038_TET2 localizes to regions of open chromatin and cell-type-specific enhancers. Loss of TET2 leads to attenuation of chromatin binding of the basic helix-loop-helix transcription factors. 31028100_Tet2 and Tet3 regulate class switch recombination in murine B cells by enhancing expression of Aicda. TET enzymes deposit 5hmC, facilitate DNA demethylation, and maintain chromatin accessibility at two TET-responsive enhancer elements, TetE1 and TetE2, located within a superenhancer in the Aicda locus. Study identified the BATF as a key transcription factor involved in TET-dependent Aicda expression. 31043609_Study in mice lacking Tet2 and Tet3 in T-reg cells indicate that Tet2 and Tet3 are guardians of T-reg cell stability and immune homeostasis. 31120187_TET2/TET3 double-deficiency does not prevent the generation and selection of high-affinity germinal centre B cells. Rather, combined TET2 and TET3 loss-of-function in germinal centre B cells favours C-to-T and G-to-A transition mutagenesis, a finding that may be of significance for understanding the aetiology of B-cell lymphomas evolving in conditions of reduced TET function. 31160465_identify two pathways of let-7 control of Tet2 and, in turn, of the key inflammatory cytokine, IL-6, thus characterizing a regulatory pathway in which a microRNA acts as a feedback inhibitor of inflammatory proce 31164154_Data demonstrated that AMPK is a critical regulator of myogenesis, partly through phosphorylating TET2. 31278362_Testes and duct deferens of mice during space flight: cytoskeleton structure, sperm-specific proteins and epigenetic events. 31286885_Loss of Tet2 has the most dramatic effect both on the phenotype of embryonic stem cells and the transcriptome compared to other genotypes. While loss of Tet proteins increased DNA hypermethylation, especially in gene promoters, these changes in DNA methylation did not correlate with gene expression changes. 31310587_These results suggest an IFNgamma-JAK-STAT-TET signaling pathway that mediates tumor response to anti-PD-L1/PD-1 therapy and is frequently disrupted in solid tumors. 31371502_data suggest that TET loss of function may at least partially underlie the characteristic pattern of global hypomethylation coupled to regional hypermethylation observed in diverse cancer genomes, and highlight the potential contribution of heterochromatin hypomethylation to oncogenesis. 31433977_Rinf Regulates Pluripotency Network Genes and Tet Enzymes in Embryonic Stem Cells. 31467227_Alternative pathways and key genes that connect Tet2, Bcl2, and Slc23a2 for their therapeutic applications with vitamin C were identified. 31484061_Non-catalytic Roles of Tet2 Are Essential to Regulate Hematopoietic Stem and Progenitor Cell Homeostasis. 31530896_TET2 catalyzes active DNA demethylation of the Sry promoter and enhances its expression. 31541101_Cardiac-specific deletion of Tet2 and Tet3 in mice (Tet2/3-DKO) leads to ventricular non-compaction cardiomyopathy (NCC) with embryonic lethality. 31582397_Data suggest that TET2 expression is tightly associated with na ive pluripotency marker expression and efficient embryonic stem cells (ESCs) self-renewal. In addition, TET2 and NANOG physically interact and co-localize on chromatin to regulate the pluripotency gene regulatory network. 31611558_Inhibition of Tet1- and Tet2-mediated DNA demethylation promotes immunomodulation of periodontal ligament stem cells. 31618637_TET2 Regulates the Neuroinflammatory Response in Microglia. 31669647_MiR-199a-5p may promote the osteoblast differentiation and prevent OVX-induced osteoporosis by targeting TET2. 31766102_Activation of endothelial cell CD137 signaling decreased TET2 expression and the TET2 content in exosomes. 31836704_Study shows that during islet autoimmunity an miR142-3p/Tet2/Foxp3 axis interferes with the efficient induction of Treg cells, resulting in impaired Treg stability in mouse and human and identified Tet2 as a direct target of miR142-3p. These findings offer a mechanistic model where during islet autoimmunity miR142-3p/Tet2-mediated Treg instability contributes to autoimmune activation and progression. 31943063_Reduction of Tet2 exacerbates early stage Alzheimer's pathology and cognitive impairments in 2xTg-AD mice. 32037456_TET2-mediated Cdkn2A DNA hydroxymethylation in midbrain dopaminergic neuron injury of Parkinson's disease. 32169654_Induction of DEAD Box helicase 5 in early adipogenesis is regulated by Ten-eleven translocation 2. 32220332_Divergent Effects of Dnmt3a and Tet2 Mutations on Hematopoietic Progenitor Cell Fitness. 32234487_TET-Mediated Hypermethylation Primes SDH-Deficient Cells for HIF2alpha-Driven Mesenchymal Transition. 32393787_Brain-specific Wt1 deletion leads to depressive-like behaviors in mice via the recruitment of Tet2 to modulate Epo expression. 32572241_Tet2 and Tet3 in B cells are required to repress CD86 and prevent autoimmunity. 32616016_Ten-eleven translocation methyl-cytosine dioxygenase 2 deficiency exacerbates renal ischemia-reperfusion injury. 32668244_The Zscan4-Tet2 Transcription Nexus Regulates Metabolic Rewiring and Enhances Proteostasis to Promote Reprogramming. 32770325_Paeonol suppresses the effect of ox-LDL on mice vascular endothelial cells by regulating miR-338-3p/TET2 axis in atherosclerosis. 32823238_TET2 promotes IL-1beta expression in J774.1 cell through TLR4/MAPK signaling pathway with demethylation of TAB2 promoter. 32934200_TET2 directs mammary luminal cell differentiation and endocrine response. 33046509_Bronchial Epithelial Tet2 Maintains Epithelial Integrity during Acute Pseudomonas aeruginosa Pneumonia. 33069358_Tet2 regulates Barx2 expression in undifferentiated and early differentiated mouse embryonic stem cells. 33090974_Hyperglycemia cooperates with Tet2 heterozygosity to induce leukemia driven by proinflammatory cytokine-induced lncRNA Morrbid. 33165916_Role of Ten eleven translocation-2 (Tet2) in modulating neuronal morphology and cognition in a mouse model of Alzheimer's disease. 33207953_Circular RNA CircMAP3K5 Acts as a MicroRNA-22-3p Sponge to Promote Resolution of Intimal Hyperplasia Via TET2-Mediated Smooth Muscle Cell Differentiation. 33216286_Roles of Tet2 in meiosis, fertility and reproductive aging. 33230319_TET2 chemically modifies tRNAs and regulates tRNA fragment levels. 33352108_Functionally distinct roles for TET-oxidized 5-methylcytosine bases in somatic reprogramming to pluripotency. 33402183_MicroRNA-210 downregulates TET2 and contributes to inflammatory response in neonatal hypoxic-ischemic brain injury. 33535102_Low shear stress-induced endothelial mesenchymal transformation via the down-regulation of TET2. 33589517_Tet2 Inactivation Enhances the Antitumor Activity of Tumor-Infiltrating Lymphocytes. 33791790_Loss of ten-eleven translocation 2 induces cardiac hypertrophy and fibrosis through modulating ERK signaling pathway. 33853366_LncRNA TUG1 Demethylated by TET2 Promotes NLRP3 Expression, Contributes to Cerebral Ischemia/Reperfusion Inflammatory Injury. 34035232_Muscle regeneration controlled by a designated DNA dioxygenase. 34086379_miR-29c-3p regulates TET2 expression and inhibits autophagy process in Parkinson's disease models. 34111946_TET2 Protects Against Vascular Smooth Muscle Cell Apoptosis and Intimal Thickening in Transplant Vasculopathy. 34158086_Roles of TET and TDG in DNA demethylation in proliferating and non-proliferating immune cells. 34165552_Ten-eleven translocation 2 modulates allergic inflammation by 5-hydroxymethylcytosine remodeling of immunologic pathways. 34218273_Stress modulates Ahi1-dependent nuclear localization of ten-eleven translocation protein 2. 34417463_Tet2 Controls the Responses of beta cells to Inflammation in Autoimmune Diabetes. 34657351_Tet2 deficiency in immune cells exacerbates tumor progression by increasing angiogenesis in a lung cancer model. 34848557_Mutant IDH Inhibits IFNgamma-TET2 Signaling to Promote Immunoevasion and Tumor Maintenance in Cholangiocarcinoma. 35011643_Role of Myeloid Tet Methylcytosine Dioxygenase 2 in Pulmonary and Peritoneal Inflammation Induced by Lipopolysaccharide and Peritonitis Induced by Escherichia coli. 35075236_Flanking sequences influence the activity of TET1 and TET2 methylcytosine dioxygenases and affect genomic 5hmC patterns. 35085104_Eltrombopag inhibits TET dioxygenase to contribute to hematopoietic stem cell expansion in aplastic anemia. 35255774_TET2 regulates osteoclastogenesis by modulating autophagy in OVX-induced bone loss. 35405484_Myeloid cell tet methylcytosine dioxygenase 2 does not affect the host response during gram-negative bacterial pneumonia and sepsis. 35658976_Tet2 deficiency drives liver microbiome dysbiosis triggering Tc1 cell autoimmune hepatitis. 35697791_Cooperation between KDM6B overexpression and TET2 deficiency in the pathogenesis of chronic myelomonocytic leukemia. 35714308_TET2-mutant clonal hematopoiesis and risk of gout. 35820129_PU.1-Dependent Enhancer Inhibition Separates Tet2-Deficient Hematopoiesis from Malignant Transformation. 35955629_Early Expression of Tet1 and Tet2 in Mouse Zygotes Altered DNA Methylation Status and Affected Embryonic Development. 35990681_TET proteins regulate T cell and iNKT cell lineage specification in a TET2 catalytic dependent manner. 36030305_Concurrent Zrsr2 mutation and Tet2 loss promote myelodysplastic neoplasm in mice. 36073548_Combined heterozygosity of FLT3 ITD, TET2, and DNMT3A results in aggressive leukemia. 36076297_ROS attenuates TET2-dependent ZO-1 epigenetic expression in cerebral vascular endothelial cells. 36271153_Srsf2[P95H/+] co-operates with loss of TET2 to promote myeloid bias and initiate a chronic myelomonocytic leukemia-like disease in mice. 36353202_Expression of the methylcytosine dioxygenase ten-eleven translocation-2 and connexin 43 in inflammatory bowel disease and colorectal cancer. 36527131_The upregulation of NLRP3 inflammasome in dorsal root ganglion by ten-eleven translocation methylcytosine dioxygenase 2 (TET2) contributed to diabetic neuropathic pain in mice. 36630508_DNA dioxygenases Tet2/3 regulate gene promoter accessibility and chromatin topology in lineage-specific loci to control epithelial differentiation. 36652477_Distinct and opposite effects of leukemogenic Idh and Tet2 mutations in hematopoietic stem and progenitor cells. 36736318_IDH2 and TET2 mutations synergize to modulate T Follicular Helper cell functional interaction with the AITL microenvironment. |
ENSG00000168769 |
TET2 |
417.225353 |
0.3636753662 |
-1.459277 |
0.20168575 |
49.523672 |
0.00000000000195992543328183477638390274086242689175266229462124556448543444275856018066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000377415729453209967124578514850383181844506452762288972735404968261718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
225.260657383648 |
31.593642557788 |
619.400372828556 |
63.6869898667598 |
| ENSMUSG00000041046 |
56089 |
Ramp3 |
protein_coding |
Q9WUP1 |
FUNCTION: Plays a role in cardioprotection by reducing cardiac hypertrophy and perivascular fibrosis in a GPER1-dependent manner. Transports the calcitonin gene-related peptide type 1 receptor (CALCRL) to the plasma membrane. Acts as a receptor for adrenomedullin (AM) together with CALCRL. {ECO:0000269|PubMed:10854696, ECO:0000269|PubMed:23674134}. |
Cell membrane;Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:56089; |
adrenomedullin receptor complex [GO:1903143]; amylin receptor complex 3 [GO:0150058]; cell surface [GO:0009986]; lysosome [GO:0005764]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; adrenomedullin receptor activity [GO:0001605]; amylin receptor activity [GO:0097643]; coreceptor activity [GO:0015026]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; adrenomedullin receptor signaling pathway [GO:1990410]; amylin receptor signaling pathway [GO:0097647]; calcium ion transport [GO:0006816]; cellular response to estradiol stimulus [GO:0071392]; cellular response to hormone stimulus [GO:0032870]; cross-receptor inhibition within G protein-coupled receptor heterodimer [GO:0038041]; G protein-coupled receptor signaling pathway [GO:0007186]; G protein-coupled receptor signaling pathway involved in heart process [GO:0086103]; intracellular protein transport [GO:0006886]; positive regulation of calcium ion import across plasma membrane [GO:1905665]; positive regulation of cell death [GO:0010942]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of gene expression [GO:0010628]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of protein kinase A signaling [GO:0010739]; positive regulation of protein kinase B signaling [GO:0051897]; positive regulation of protein localization to plasma membrane [GO:1903078]; positive regulation of receptor recycling [GO:0001921]; protein localization to plasma membrane [GO:0072659]; protein transport [GO:0015031]; receptor internalization [GO:0031623]; response to amyloid-beta [GO:1904645] |
11854283_role in cell surface expression of CRLR/RAMP heterodimeric receptors 17470425_RAMP2 and RAMP3 have distinct physiological functions throughout embryogenesis, adulthood, and old age 18255060_tight regulation of WAP gene expression parallels variations in the chromatin structure and DNA methylation profile throughout the Ramp3-WAP-Tbrg4 locus. 22100352_these data demonstrate a sex-dependant, cardioprotective role of RAMP3 in the setting of chronic hypertension. 22563471_Data show that mechanical ventilation reduced the expression of receptor activity-modifying protein RAMP3, but not of intermedin (IMD), calcitonin receptor-like receptor (CRLR), and RAMP1 and RAMP2. 23674134_G-protein-coupled receptor 30 interacts with receptor activity-modifying protein 3 and confers sex-dependent cardioprotection. 25264174_RAMP3 regulates drainage through lymphatic vessels. 27940069_the cardiovascular response of Ramp1(-/-), Ramp2(+/-), Ramp3(-/-), Ramp1(-/-)/Ramp3(-/-) double-knockout (dKO), and Calcrl(+/-) mice to AM and CGRP were compared to wildtype mice.These results suggest that the hypotensive effect of AM is primarily mediated through the CLR/RAMP1 heterodimer, but that AM signaling via CLR/RAMP2 and CLR/RAMP3 also contributes to some hypotensive action. 31754214_Deficiency of the adrenomedullin-RAMP3 system suppresses metastasis through the modification of cancer-associated fibroblasts. 31881259_RAMP1 and RAMP3 Differentially Control Amylin's Effects on Food Intake, Glucose and Energy Balance in Male and Female Mice. 33545715_Adrenomedullin-RAMP2 and -RAMP3 Systems Regulate Cardiac Homeostasis during Cardiovascular Stress. |
ENSG00000122679 |
RAMP3 |
14.253446 |
5.5729691683 |
2.478446 |
0.78577423 |
9.893181 |
0.00165892364587221104697822848805799367255531251430511474609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0072382483454384192664288200091959879500791430473327636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.003616639115 |
12.0138237960983 |
3.76883776040709 |
1.75704528053892 |
| ENSMUSG00000041064 |
208084 |
Pif1 |
protein_coding |
Q80SX8 |
FUNCTION: DNA-dependent ATPase and 5'-3' DNA helicase required for the maintenance of both mitochondrial and nuclear genome stability. Efficiently unwinds G-quadruplex (G4) DNA structures and forked RNA-DNA hybrids. Resolves G4 structures, preventing replication pausing and double-strand breaks (DSBs) at G4 motifs. Involved in the maintenance of telomeric DNA. Inhibits telomere elongation, de novo telomere formation and telomere addition to DSBs via catalytic inhibition of telomerase. Reduces the processivity of telomerase by displacing active telomerase from DNA ends. Releases telomerase by unwinding the short telomerase RNA/telomeric DNA hybrid that is the intermediate in the telomerase reaction. Possesses an intrinsic strand annealing activity. {ECO:0000255|HAMAP-Rule:MF_03176}. |
Alternative initiation;Alternative splicing;ATP-binding;DNA damage;DNA recombination;DNA repair;DNA-binding;Helicase;Hydrolase;Mitochondrion;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:208084; |
chromosome, telomeric region [GO:0000781]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; replication fork [GO:0005657]; 5'-3' DNA helicase activity [GO:0043139]; 5'-3' DNA/RNA helicase activity [GO:0033678]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; G-quadruplex DNA binding [GO:0051880]; magnesium ion binding [GO:0000287]; single-stranded DNA helicase activity [GO:0017116]; telomerase inhibitor activity [GO:0010521]; telomeric DNA binding [GO:0042162]; DNA duplex unwinding [GO:0032508]; DNA recombination [GO:0006310]; DNA repair [GO:0006281]; DNA replication [GO:0006260]; mitochondrial genome maintenance [GO:0000002]; negative regulation of telomerase activity [GO:0051974]; negative regulation of telomere maintenance via telomerase [GO:0032211]; regulation of telomere maintenance [GO:0032204]; telomere maintenance [GO:0000723] |
17130244_Murine telomere homeostasis or genetic stability does not depend on mPif1. 21945094_murine cells have evolved mechanisms to ensure the functional redundancy of Pif1 or Nbs1 in the regulation of chromosome healing. 26923168_pif1-/- animals develop a mitochondrial myopathy with respiratory chain deficiency. 31136580_They also support a role for Pif1 in weight gain resistance and liver metabolic dysregulation during nutrient stress. |
ENSG00000140451 |
PIF1 |
282.039360 |
2.6997526473 |
1.432827 |
0.16631549 |
73.618021 |
0.00000000000000000947952626552519802938751894395360788631455402206875751613956992969178827479481697082519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000003111899656820686042199655587926517562755317539667510029488539657904766499996185302734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
416.288059996337 |
48.0965164518548 |
154.194889079241 |
13.9078761243172 |
| ENSMUSG00000041112 |
140580 |
Elmo1 |
protein_coding |
Q8BPU7 |
FUNCTION: Involved in cytoskeletal rearrangements required for phagocytosis of apoptotic cells and cell motility. Acts in association with DOCK1 and CRK. Was initially proposed to be required in complex with DOCK1 to activate Rac Rho small GTPases. May enhance the guanine nucleotide exchange factor (GEF) activity of DOCK1 (By similarity). {ECO:0000250}. |
Acetylation;Alternative splicing;Apoptosis;Cell membrane;Cytoplasm;Membrane;Phagocytosis;Phosphoprotein;Reference proteome;SH3-binding |
|
|
mmu:140580; |
cytoplasm [GO:0005737]; guanyl-nucleotide exchange factor complex [GO:0032045]; plasma membrane [GO:0005886]; guanyl-nucleotide exchange factor activity [GO:0005085]; SH3 domain binding [GO:0017124]; actin cytoskeleton organization [GO:0030036]; actin filament organization [GO:0007015]; actin filament-based process [GO:0030029]; apoptotic process [GO:0006915]; cell motility [GO:0048870]; phagocytosis [GO:0006909]; phagocytosis, engulfment [GO:0006911]; Rac protein signal transduction [GO:0016601] |
17960134_functional studies suggest that BAI1 cooperates with ELMO/Dock180/Rac to promote maximal engulfment of apoptotic cells 20844538_findings uncover a selective requirement for ELMO1 in Sertoli-cell-mediated removal of apoptotic germ cells and make a compelling case for a relationship between engulfment and tissue homeostasis in vivo 21804544_Data show that intracellular engulfment protein ELMO1 was required for phagocytosis by DCX(+) cells. 23273896_regulator of Med31, revealing a previously unrecognized link between cytoplasmic signaling proteins and the Mediator complex 24821968_findings reveal a previously unknown, nonredundant role for Elmo1 in controlling Dock2 levels and Dock2-dependent T cell migration in primary lymphocytes. 26205662_Src contributed to tyrosine phosphorylation of ELMO1 at Y720 and Y724 residues, which was crucial for activation of Rac1, followed by the promoting cell adhesion, spreading, and migration. 26858454_Elmo1 hypermorphs have albuminuria, glomerulosclerosis, and changes in the ultrastructure of the glomerular basement membrane that increase in severity in parallel with the expression of Elmo 1. 28219480_The authors identify ELMO1 as a novel target of TGFbeta signaling and show that restoration of Tgfbr2 results in a complete block of ELMO1 in vivo. Knocking down Elmo1 impairs metastasis of carcinoma cells to the lung, thereby providing insights into the mechanisms of progression of Tgfbr2-deficient invasive transition zone squamous cell carcinoma. 29029244_ELMO1 is present in the phagosome and enhances bacterial clearance by differential regulation of lysosomal acidification and enzymatic activity. 30219846_ELMO1 protects against DSS-induced colonic injury in mice through its effect on epithelial migration via Rac1 activation. 30643265_Loss of the engulfment signaling protein Elmo1 alleviated disease severity in mouse models of arthritis through Elmo1's regulation of neutrophil recruitment to inflamed joints. 30787021_Platelet aggregation, granule secretion, integrin alphaIIbbeta3 activation, and thromboxane generation were enhanced in ELMO1(-/-) platelets in response to glycoprotein VI (GPVI) agonists but unaltered when a protease-activated receptor 4 agonist was used. These results indicate that ELMO1 plays an important role in hemostasis and thrombosis in vivo. 32003126_Host engulfment pathway controls inflammation in inflammatory bowel disease. 34404802_ELMO1 signaling is a promoter of osteoclast function and bone loss. 34719317_The interaction of enteric bacterial effectors with the host engulfment pathway control innate immune responses. 34919331_TNF-alpha/ENO1 signaling facilitates testicular phagocytosis by directly activating Elmo1 gene expression in mouse Sertoli cells. 36694274_The crosstalk between microbial sensors ELMO1 and NOD2 shape intestinal immune responses. |
ENSG00000155849 |
ELMO1 |
269.841160 |
0.1008135557 |
-3.310238 |
0.19347393 |
300.154853 |
0.00000000000000000000000000000000000000000000000000000000000000000030481342917091656184788169070839974146350266373924867193916289337791225822538271103881864377664355213514080969559364874661366383043722562031958552181107589948171651249991809251582708384376019239426 |
0.0000000000000000000000000000000000000000000000000000000000000000800503129850241672861863880465816952221813043151271764409484850697036641716346717419300524170932779507756386251296172672155919532194549117542655226404186004731701464809390245136455632746219635010 |
Yes |
No |
49.2468614690076 |
6.62410849546813 |
488.494440100728 |
40.5011921458033 |
| ENSMUSG00000041117 |
434130 |
Ccdc8 |
protein_coding |
D3YZV8 |
FUNCTION: Core component of the 3M complex, a complex required to regulate microtubule dynamics and genome integrity. It is unclear how the 3M complex regulates microtubules, it could act by controlling the level of a microtubule stabilizer. Required for localization of CUL7 to the centrosome. {ECO:0000250|UniProtKB:Q9H0W5}. |
Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:434130; |
3M complex [GO:1990393]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; microtubule cytoskeleton organization [GO:0000226]; regulation of mitotic nuclear division [GO:0007088] |
Human_homologues 21737058_We propose that CUL7, OBSL1, and CCDC8 are members of a pathway controlling mammalian growth. 22084066_p90 is a critical cofactor for p53-mediated apoptosis through promoting Tip60-mediated p53 acetylation. 22156540_discussion of roles of CCDC8, CUL7 (cullin 7), and OBSL1 (obscurin-like 1) in growth and development using findings from patients with Miller-McKusick-Malvaux syndrome and Silver-Russell syndrome [REVIEW] 23018678_Mutations in CUL7, OBSL1 and CCDC8 in 3-M syndrome lead to disordered growth factor signalling. 24711643_CUL7, OBSL1 and CCDC8 modulate the alternative splicing of the INSR 24793695_The CUL7, OBSL1, and CCDC8 proteins form a 3M complex that functions in maintaining microtubule and genome integrity and normal development. 27342910_CCDC8 may suppress the invasion and metastasis of non-small cell lung cancer cells. 28700999_Variants near TTN and CCDC8 were associated with KI67 expression, and rs2288563 and rs2562832 in TTN are potential biomarkers for the prediction of clinical outcomes in hepatitis B-related hepatocellular carcinoma patients. 32651437_Overexpressed coiled-coil domain containing protein 8 (CCDC8) mediates newly synthesized HIV-1 Gag lysosomal degradation. 33258289_A rare cause of syndromic short stature: 3M syndrome in three families. |
ENSG00000169515 |
CCDC8 |
160.208329 |
2.9984811738 |
1.584232 |
0.21054539 |
55.975286 |
0.00000000000007338777074216874666121529114217737699494333719618666123096772935241460800170898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000016784422281452166889075145937826006529557804070051929556939285248517990112304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
239.002490530471 |
35.0037154117731 |
79.7078509682898 |
9.21038856623219 |
| ENSMUSG00000041144 |
227058 |
Dnah7b |
protein_coding |
A0A087WPC9 |
Human_homologues FUNCTION: Force generating protein of respiratory cilia. Produces force towards the minus ends of microtubules. Dynein has ATPase activity; the force-producing power stroke is thought to occur on release of ADP (By similarity). {ECO:0000250}. |
Reference proteome |
|
|
Human_homologues hsa:56171; |
cytosol [GO:0005829]; dynein complex [GO:0030286]; inner dynein arm [GO:0036156]; dynein intermediate chain binding [GO:0045505]; dynein light intermediate chain binding [GO:0051959]; minus-end-directed microtubule motor activity [GO:0008569]; cilium movement [GO:0003341]; inner dynein arm assembly [GO:0036159]; microtubule-based movement [GO:0007018] |
Human_homologues 11877439_These studies identify DNAH7 as an inner arm component of human cilia that is synthesized but not assembled in a case of primary ciliary dyskinesia. 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 34476482_Bi-allelic mutations in DNAH7 cause asthenozoospermia by impairing the integrality of axoneme structure. 35543642_Loss of function mutation in DNAH7 induces male infertility associated with abnormalities of the sperm flagella and mitochondria in human. |
ENSG00000118997 |
DNAH7 |
55.005809 |
3.2249031313 |
1.689256 |
0.37120702 |
20.742986 |
0.00000525236023410729054505460822177553836809238418936729431152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000409595381739872051022645360607299380717449821531772613525390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
89.246336735228 |
23.2173960531899 |
27.6741139508435 |
5.88731876924949 |
| ENSMUSG00000041193 |
18784 |
Pla2g5 |
protein_coding |
Q6GTW1 |
Human_homologues FUNCTION: Secretory calcium-dependent phospholipase A2 that primarily targets extracellular phospholipids (PubMed:8300559). Hydrolyzes the ester bond of the fatty acyl group attached at sn-2 position of phospholipids (phospholipase A2 activity), preferentially releasing fatty acyl groups with a low degree of unsaturation such as oleoyl (C18:1) and linoleoyl (C18:2) groups (PubMed:8300559, PubMed:14998370, PubMed:23533611). Hydrolyzes low-density lipoprotein (LDL) phospholipids releasing unsaturated fatty acids that drive macrophage polarization toward an M2 phenotype (By similarity). May act in an autocrine and paracrine manner. Contributes to lipid remodeling of cellular membranes at different subcellular locations and generation of lipid mediators involved in pathogen clearance. Cleaves sn-2 fatty acyl chains of cardiolipin, a major component of the inner membrane of mitochondria and bacterial membranes (PubMed:23533611). Promotes phagocytosis of bacteria in macrophages through production of lysophosphatidylethanolamines (PubMed:25725101). Displays bactericidal activity against Gram-positive bacteria by directly hydrolyzing phospholipids of the bacterial membrane (PubMed:11694541). Promotes phagocytosis and killing of ingested fungi likely through controlling phagosome-lysosome fusion and phagosome maturation (By similarity). Plays a role in biosynthesis of cysteinyl leukotrienes (CysLTs) in myeloid cells (PubMed:12124392, PubMed:12796497). In eosinophils, triggers perinuclear arachidonate release and LTC4 synthesis in a PLA2G4A-independent way (PubMed:12796497). In neutrophils, amplifies CysLTs biosynthesis initiated by PLA2G4A (PubMed:12124392). Promotes immune complex clearance in macrophages via stimulating synthesis of CysLTs, which act through CYSLTR1 to trigger phagocytosis (By similarity). May regulate antigen processing in antigen-presenting cells (By similarity). In pulmonary macrophages regulates IL33 production required for activation of group 2 innate lymphoid cells (By similarity). May play a role in the biosynthesis of N-acyl ethanolamines that regulate energy metabolism. Hydrolyzes N-acyl phosphatidylethanolamines to N-acyl lysophosphatidylethanolamines, which are further cleaved by a lysophospholipase D to release N-acyl ethanolamines (PubMed:14998370). {ECO:0000250|UniProtKB:P97391, ECO:0000269|PubMed:11694541, ECO:0000269|PubMed:12124392, ECO:0000269|PubMed:12796497, ECO:0000269|PubMed:14998370, ECO:0000269|PubMed:23533611, ECO:0000269|PubMed:25725101, ECO:0000269|PubMed:8300559}. |
Calcium;Disulfide bond;Hydrolase;Lipid metabolism;Metal-binding;Reference proteome;Secreted;Signal |
Human_homologues PATHWAY: Lipid metabolism; phospholipid metabolism. {ECO:0000269|PubMed:25725101}.; PATHWAY: Lipid metabolism; leukotriene B4 biosynthesis. {ECO:0000269|PubMed:12124392}.; PATHWAY: Lipid metabolism; leukotriene C4 biosynthesis. {ECO:0000269|PubMed:12796497}. |
|
mmu:18784; |
extracellular region [GO:0005576]; calcium ion binding [GO:0005509]; calcium-independent phospholipase A2 activity [GO:0047499]; arachidonic acid secretion [GO:0050482]; cardiolipin acyl-chain remodeling [GO:0035965]; leukotriene biosynthetic process [GO:0019370]; phosphatidylcholine catabolic process [GO:0034638]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of immune complex clearance by monocytes and macrophages [GO:0090265]; positive regulation of phospholipase activity [GO:0010518] |
12676927_the effect of group IIa and V PLA2s on H2O2-induced AA release is dependent upon the presence of cPLA2alpha and the activation of PKC and ERK1/2. Group IIa and V PLA2s are regulatory and cPLA2alpha is responsible for AA release. 12963740_encapsulation of Group V PLA2 into granules brings the enzyme to the perinuclear envelope during cell activation 14761945_a role in innate immunity. 16150819_GIVA PLA2 is the dominant player involved in AA release, but it appears not to be involved in the regulation of LPS-induced expression of GV PLA2 or cyclooxygenase-2 16407308_Group V secretory PLA2 contributes to the innate immune response both through regulation of eicosanoid generation in response to a phagocytic stimulus and also as a component of the phagocytic machinery. 17008322_group V, but not group X, secreted phospholipase A2 has a role in lung dysfunction 17064958_Delayed-phase PGD(2) generation and COX-2 induction were reduced approximately 35% in C57BL/6 Pla2g5-/- bone marrow-derived mast cells. 17369491_In mast cells lacking group V secretory PLA(2), the course of phosphorylation of ERK1/2 and of cPLA(2)alpha was markedly truncated. 17878379_group v phospholipase A2 is a critical messenger enzyme in the development of airway hyperresponsiveness and regulation of cell migration during immunosensitization by a pathway that is independent of group IVa phospholipase A2 18825749_results demonstrate the critical role of cPLA2 for the duration of inflammation in collagen-induced arthritis 19056705_role for syndecan-4 in mediating the uptake of LDL modified by group V secretory phospholipase A2, a process implicated in atherosclerotic lesion progression 19115385_sPLA(2), either through pathways comprising Ras/Raf-1/MEK1/ERK1/2 or the classical PKC family, plays an essential role in Mtb-mediated ROS generation and inflammatory mediator release by microglial cells. 19164803_The capacity of group V sPLA2 to increase atherogenicity of ApoE-/- and LDLR-/- mouse LDL in vitro predicts its atherogenic role in vivo. 19286925_gVPLA(2) is an inducible protein in pla2g5(+/+) mice but not in pla2g5(-/-) mice within 24 h after lipopolysaccharide treatment 19342668_group V sPLA(2) regulates phagocytosis through major phagocytic receptors and contributes to the innate immune response against C. albicans by regulating phagocytosis and killing through a mechanism that is likely dependent on phagolysosome fusion 20138782_J-774 cells incubated with Group V secretory phospholipase A(2) hydrolyzed-LDL produced more TNF-alpha and IL-6 compared to cells incubated with control-LDL. 20232296_Demonstrate the role of sPLA(2)-V in lipopolysaccharide-induced ICAM-1 and VCAM-1 protein overexpression and leukocyte recruitment, supporting the contribution of sPLA(2)-V in the development of inflammatory innate immune responses. 20817863_expression of group V sPLA(2) in antigen-presenting cells regulates Ag processing and maturation of dendritc cells and contributes to pulmonary inflammation and immune response against D. farinae. 21169294_sPLA(2)-V plays an important role in the pathogenesis of myocardial ischemia reperfusion injury partly in concert with the activation of cPLA(2) 21262967_agonist-induced MAPK activation leads to Prdx6 phosphorylation and translocation to the cell membrane, where its PLA(2) activity facilitates assembly of the NOX2 complex and activation of the oxidase. 21530210_Data from cuprizone-induced model of multiple sclerosis suggest that sPLA2 down-regulation during remyelination can limit release of AA and consequent production of pro-inflammatory PGs/TXB2 (which are increased during demyelination). 21849511_GV sPLA(2) in bone marrow-derived myeloid cells as well as non-myeloid cells, which are likely bronchial epithelial cells, participate in the regulation of the innate immune response to pulmonary infection with E. coli. 22067043_The results indicate that the PLA(2) activity also plays a substantial role in protecting cells against oxidant stress caused by an exogenous hydroperoxide. 22813854_The impact of group V sPLA(2) deficiency on angiotensin (Ang) II-induced cardiac fibrosis in apoE deficient mice was investigated. 23451035_Key Data shows the role of group v secreted phospholipase A2 in Th2 cytokine and dendritic cell-driven airway hyperresponsiveness and remodeling. 23650617_Our studies identified a unique function of gV-sPLA2 in activation of macrophages 24910243_PLA2G5 hydrolyzed phosphatidylcholine in fat-overladen low-density lipoprotein to release unsaturated fatty acids, which prevented palmitate-induced M1 macrophage polarization. 25347447_AdPLA2 plays an important role in promoting tumorigenesis and disease progression by modulating the production of prostaglandins and may serve as a potential therapeutic target in TSC and LAM. 26820468_GV sPLA2 is involved in the maintenance of tubular cell function and integrity, promoting sodium retention through increased cortical (Na+ + K+)-ATPase expression and activity. 28825176_Deficiency of GV sPLA2 results in diminished glucose-stimulated insulin secretion in isolated pancreatic beta-cells 29346348_macrophage-associated Pla2g5 contributes significantly to type-2 immunity through regulation of IL-33 induction and free fatty acids-driven group 2 innate lymphoid cell activation 31730773_Lysosome-mediated autophagy pathway contributes to gVPLA2 clearance from lung endothelial cells. 33383652_Lipid Profile of Activated Macrophages and Contribution of Group V Phospholipase A2. 34359901_Group V Phospholipase A2 Mediates Endothelial Dysfunction and Acute Lung Injury Caused by Methicillin-Resistant Staphylococcus Aureus. |
ENSG00000127472 |
PLA2G5 |
49.069175 |
0.4124349725 |
-1.277761 |
0.38165899 |
11.062556 |
0.00088088616865796291657136896091628841531928628683090209960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0041259711319182315256148818605197448050603270530700683593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
22.9701127581788 |
6.14777502121213 |
55.6939015627432 |
11.1622371686253 |
| ENSMUSG00000041219 |
228482 |
Arhgap11a |
protein_coding |
Q80Y19 |
FUNCTION: GTPase activator for the Rho-type GTPases by converting them to an inactive GDP-bound state. {ECO:0000250|UniProtKB:Q6P4F7}. |
GTPase activation;Nucleus;Phosphoprotein;Reference proteome |
Human_homologues NA; + ;NA |
|
mmu:228482; |
mitochondrial matrix [GO:0005759]; nucleus [GO:0005634]; GTPase activator activity [GO:0005096]; positive regulation of GTPase activity [GO:0043547]; signal transduction [GO:0007165] |
Human_homologues 27216196_High ARHGAP11A expression is associated with Basal-like Breast Cancers. 34140070_[Rho GTPase-activating protein 11A (ARHGAP11A) is up-regulated in lung adenocarcinoma and positively associated with poor prognosis]. 35754342_MicroRNA-30c-2-3p represses malignant progression of gastric adenocarcinoma cells via targeting ARHGAP11A.; + ;25721503_ARHGAP11B may have contributed to evolutionary expansion of human neocortex. 27957544_The data also demonstrate that a single C-->G base substitution underlies the ARHGAP11B-mediated basal progenitor amplification implicated in neocortex expansion. 30484771_Human-specific ARHGAP11B induces hallmarks of neocortical expansion in developing ferret neocortex. 31883789_an ARHGAP11B-induced, mitochondria-based effect on basal progenitor metabolism that is a hallmark of highly mitotically active cells appears to underlie its role in neocortex expansion. 32554627_Human-specific ARHGAP11B increases size and folding of primate neocortex in the fetal marmoset. 33938018_Expression of human-specific ARHGAP11B in mice leads to neocortex expansion and increased memory flexibility. 36098218_Human-specific ARHGAP11B ensures human-like basal progenitor levels in hominid cerebral organoids. 36381990_Human-specific ARHGAP11B ensures human-like basal progenitor levels in hominid cerebral organoids. |
ENSG00000198826+ENSG00000285077 |
ARHGAP11A+ARHGAP11B |
767.983756 |
2.4929030716 |
1.317827 |
0.10055492 |
170.077669 |
0.00000000000000000000000000000000000000711545883192405863401694428907481861089819423547307170009069681607462709584719782261354954818401868411881228126958376378752291202545166015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000007911143717362574352591010404780138091347453172136395058651293372109356419116329484319033904689277836297378598828800022602081298828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1066.66213504978 |
74.4058746969774 |
427.879506108335 |
23.3095992711993 |
| ENSMUSG00000041235 |
320790 |
Chd7 |
protein_coding |
A2AJK6 |
FUNCTION: Probable transcription regulator. Maybe involved in the in 45S precursor rRNA production (By similarity). {ECO:0000250}. |
Alternative splicing;ATP-binding;Chromatin regulator;Coiled coil;DNA-binding;Helicase;Hydrolase;Methylation;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;rRNA processing;Transcription;Transcription regulation |
|
This gene encodes a protein containing two chromodomains and an ATP-binding helicase domain that functions as a regulator of transcription. Mutations in this gene result in an array of development defects, including inner ear problems. Mice defective for this gene exhibit many of the clinical features of the CHARGE syndrome caused by mutations in the homologous gene in human. [provided by RefSeq, Sep 2015]. |
mmu:320790; |
chromatin [GO:0000785]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; ATP-dependent chromatin remodeler activity [GO:0140658]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; helicase activity [GO:0004386]; histone binding [GO:0042393]; promoter-specific chromatin binding [GO:1990841]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; adult heart development [GO:0007512]; adult walking behavior [GO:0007628]; aorta development [GO:0035904]; aorta morphogenesis [GO:0035909]; artery morphogenesis [GO:0048844]; atrioventricular canal development [GO:0036302]; blood circulation [GO:0008015]; blood vessel development [GO:0001568]; blood vessel remodeling [GO:0001974]; camera-type eye development [GO:0043010]; cardiac septum morphogenesis [GO:0060411]; central nervous system development [GO:0007417]; chordate embryonic development [GO:0043009]; chromatin remodeling [GO:0006338]; cognition [GO:0050890]; cranial nerve development [GO:0021545]; ear morphogenesis [GO:0042471]; embryonic hindlimb morphogenesis [GO:0035116]; epithelium development [GO:0060429]; face development [GO:0060324]; female genitalia development [GO:0030540]; genitalia development [GO:0048806]; heart morphogenesis [GO:0003007]; in utero embryonic development [GO:0001701]; inner ear morphogenesis [GO:0042472]; innervation [GO:0060384]; limb development [GO:0060173]; locomotory behavior [GO:0007626]; nose development [GO:0043584]; olfactory behavior [GO:0042048]; olfactory bulb development [GO:0021772]; olfactory nerve development [GO:0021553]; positive regulation of multicellular organism growth [GO:0040018]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of gene expression [GO:0010468]; regulation of growth hormone secretion [GO:0060123]; regulation of neurogenesis [GO:0050767]; regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum [GO:0010880]; response to bacterium [GO:0009617]; retina development in camera-type eye [GO:0060041]; right ventricular compact myocardium morphogenesis [GO:0003226]; roof of mouth development [GO:0060021]; rRNA processing [GO:0006364]; secondary palate development [GO:0062009]; semicircular canal morphogenesis [GO:0048752]; sensory perception of sound [GO:0007605]; skeletal system development [GO:0001501]; T cell differentiation [GO:0030217]; tissue remodeling [GO:0048771]; transcription by RNA polymerase II [GO:0006366]; ventricular trabecula myocardium morphogenesis [GO:0003222] |
16207732_The large number of mouse mutants may be due to the combination of the Chd7 gene being a large target and the fact that many heterozygous carriers of the mutations are viable individuals with a readily detectable phenotype 17334657_Utility of Chd7 as a reporter-tagged loss-of-function allele for future studies exploring developmental mechanisms of Chd7 deficiency. 17701983_Chd7(Gt/+) mice display variable asymmetric lateral and posterior semicircular canal malformations, as well as defects in vestibular sensory epithelial innervation despite the presence of intact hair cells in the target organs. 19279158_Mammalian olfactory dysfunction due to Chd7 haploinsufficiency is linked to defects in olfactory neural stem cell proliferation. 19855134_biallelic expression of Chd7 and Tbx1 in the pharyngeal ectoderm was required for normal pharyngeal arch artery development 20507341_[review] recent and ongoing analyses of CHD7 function in mouse models and cell-based systems 20591827_CHD7 functions in the nucleolus as a positive regulator of ribosomal RNA biogenesis. 20657823_enhancer-mediated gene dysregulation contributes to disease pathogenesis and the critical CHD7 target genes may be subject to positive or negative regulation. 20736290_Conditional deletion of Chd7 in the developing otocyst using Foxg1-Cre resulted in cochlear hypoplasia and complete absence of the semicircular canals and cristae. 21532573_characterize gene regulation by Sox2 in neural stem cells. We 21875659_Chd7(Gt)(/+) mouse model of CHARGE syndrome demonstrates combined conductive and sensorineural hearing loss, correlating with changes in both middle and inner ears. 22539951_Otitis media in Chd7Ome/+ mice is characterized by Eustachian tube dysfunction, epithelial hyperplasia, middle ear effusion and associated hearing loss. 22658483_CHD7 may directly regulate Bmp4 expression by binding with an enhancer element downstream of the Bmp4 locus. 22705977_Chd7 may have critical selector gene functions during inner ear morphogenesis. 24026680_Findings demonstrate critical, cooperative roles for Retinoic Acid (RA) and CHD7 in subventricular zone neural stem cell function and inner ear development, suggesting that altered RA signaling may be an effective method for treating Chd7 deficiency. 24728844_CHD7 gene mutation is associated with CHARGE syndrome. 24975120_Conditional deletion of Chd7 in ectodermal and endodermal derivatives or migrating neural crest cells results in varied and severe craniofacial defects. 25183173_Findings directly link CHD7 to pathways involved in NSC quiescence and identify the first chromatin-remodeling factor with a role in NSC quiescence and maintenance. 26102480_This work reveals the importance of CHD7 in the cardiogenic mesoderm for multiple processes during cardiovascular development. 26670829_Chd7 mutant mice are models for determining the molecular etiology of ocular defects in CHARGE syndrome. 26928066_Chd7 coordinates with Sox10 to regulate the initiation of myelinogenesis and acts as a molecular nexus of regulatory networks that account for the development of a seemingly diverse array of lineages, including oligodendrocytes and osteoblasts, pointing to previously uncharacterized Chd7 functions. 27955690_CHD7 is an important factor in the proliferation and stemness maintenance of neural stem/progenitor cells. 28165338_CHD7 is necessary for maintaining an open, accessible chromatin state at the Reln locus. Taken together, this study shows that Reln gene expression is regulated by chromatin remodeling, identifies CHD7 as a previously unrecognized upstream regulator of Reln, and provides direct in vivo evidence that a mammalian CHD protein can control brain development 28317875_Data show that chromodomain helicase DNA-binding protein 7 (Chd7) is required for the maintenance of open chromatin and thus activation of genes essential for granule neuron differentiation. 28931573_Chd7 regulates the proliferation and identity of oligodendrocyte precursor cells after spinal cord injury. 29018080_Chd7 deficiency delays leukemia initiation induced by Cbfb-MYH11. 29168327_we have identified mild cerebellar hypoplasia and distinct cerebellar foliation anomalies in Chd7 gt/+ mice. Our findings imply a specific function for CHD7 in controlling the spatiotemporal initiation of cerebellar fissures and show that normal fissure formation requires bi-allelic Chd7 expression, consistent with the haploinsuffi- cient nature of CHARGE syndrome. 29212025_Findings extend current knowledge of the role of BMI1 and CHD7 in medulloblastoma pathogenesis, and they raise the possibility that pharmacological targeting of BMI1 or ERK may be particularly indicated in a subgroup of MB with low expression levels of CHD7. 29467333_Study using human SH-SY5Y neuroblastoma and 293T cells and transgenic mouse model show that CHD7 directly regulates expression of retinoic acid synthetic enzyme ALDH1A3. Results indicate that ALDH1A3 acts with CHD7 in a common genetic pathway to regulate inner ear development, providing insights for exploration of the pathogenic mechanisms underlying CHARGE syndrome. 30108144_CHD7 and CHD8 bind in Oligodendrocyte precursor cells to a majority of ASD risk-associated genes, suggesting an implication of oligodendrocyte lineage cells in ASD neurological defects. 32883883_CHD7 and Runx1 interaction provides a braking mechanism for hematopoietic differentiation. 33060836_CHD7 promotes neural progenitor differentiation in embryonic stem cells via altered chromatin accessibility and nascent gene expression. 33127760_CHD7 regulates cardiovascular development through ATP-dependent and -independent activities. 34732824_The chromatin remodelling factor Chd7 protects auditory neurons and sensory hair cells from stress-induced degeneration. 35129866_CHARGE syndrome-associated proteins FAM172A and CHD7 influence male sex determination and differentiation through transcriptional and alternative splicing mechanisms. 35418650_CHD7 regulates bone-fat balance by suppressing PPAR-gamma signaling. 36232804_Characterization of Two Mouse Chd7 Heterozygous Loss-of-Function Models Shows Dysgenesis of the Corpus Callosum and Previously Unreported Features of CHARGE Syndrome. 36288662_Loss of the chromatin remodeler CHD7 impacts glial cells and myelination in the mouse cochlear spiral ganglion. 36343670_Chromatin remodeler Chd7 regulates photoreceptor development and outer segment length. |
ENSG00000171316 |
CHD7 |
128.417796 |
0.3186016304 |
-1.650174 |
0.26886476 |
37.063042 |
0.00000000114371060217899366121489175313237496300899920242954976856708526611328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000160266788338458437829694411052780522730643042450537905097007751464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.3696194529982 |
11.1284486712144 |
186.344367970392 |
24.9633541145562 |
| ENSMUSG00000041268 |
235380 |
Dmxl2 |
protein_coding |
Q8BPN8 |
FUNCTION: May serve as a scaffold protein for MADD and RAB3GA on synaptic vesicles of neuronal and endocrine homeostatic processes (By similarity). Plays a role in the brain as a key controller of neuronal and endocrine homeostatic processes (PubMed:25248098). {ECO:0000250|UniProtKB:Q8TDJ6, ECO:0000269|PubMed:25248098}. |
Alternative splicing;Coiled coil;Cytoplasmic vesicle;Membrane;Phosphoprotein;Reference proteome;Repeat;Synapse;WD repeat |
|
|
|
neuronal dense core vesicle [GO:0098992]; RAVE complex [GO:0043291]; synaptic vesicle [GO:0008021]; synaptic vesicle membrane [GO:0030672]; small GTPase binding [GO:0031267]; brain development [GO:0007420]; chemical synaptic transmission [GO:0007268]; corpus callosum development [GO:0022038]; glucose homeostasis [GO:0042593]; sensory perception of smell [GO:0007608]; spermatogenesis [GO:0007283]; vacuolar acidification [GO:0007035] |
20810660_an important role for Rabconnectin-3 and V-ATPase activity in the Notch signaling pathway in mammalian cells. 22707207_The DMXL2 interactome, was studied. 25248098_Haploinsufficiency of Dmxl2, encoding a synaptic protein, causes infertility associated with a loss of GnRH neurons in humans and mice. 27657680_Data provide evidence that DMXL2 plays an important role in the inner ear function. 30735494_Dmxl2 exerts its principal function in the testes. 31253182_Co-immunoprecipitations from brain tissue is consistent with the formation of a protein complex between RB-3alpha and RB-3beta and both Cav2.2 and the related Cav2.1 calcium channel. |
ENSG00000104093 |
DMXL2 |
69.482236 |
2.1491974534 |
1.103798 |
0.32663377 |
11.205791 |
0.00081542478953753846134672178536106912360992282629013061523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0038465625253130339422646954972151434049010276794433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
93.3835361344978 |
21.3236145236937 |
43.4504219170868 |
7.62554704003517 |
| ENSMUSG00000041351 |
110351 |
Rap1gap |
protein_coding |
A2ALS5 |
FUNCTION: GTPase activator for the nuclear Ras-related regulatory protein RAP-1A (KREV-1), converting it to the putatively inactive GDP-bound state. {ECO:0000250}. |
Alternative splicing;Golgi apparatus;GTPase activation;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:110351; |
axon [GO:0030424]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; dendrite [GO:0030425]; early endosome [GO:0005769]; Golgi membrane [GO:0000139]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; GTPase activator activity [GO:0005096]; protein homodimerization activity [GO:0042803]; small GTPase binding [GO:0031267]; activation of GTPase activity [GO:0090630]; cell-cell adhesion [GO:0098609]; cellular response to glial cell derived neurotrophic factor [GO:1990792]; establishment of localization in cell [GO:0051649]; negative regulation of GTP binding [GO:1904425]; negative regulation of microvillus assembly [GO:1903697]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of thyroid gland epithelial cell proliferation [GO:1904442]; phagocytosis [GO:0006909]; positive regulation of cell-cell adhesion [GO:0022409]; positive regulation of GTPase activity [GO:0043547]; positive regulation of phagocytosis [GO:0050766]; regulation of GTPase activity [GO:0043087]; regulation of small GTPase mediated signal transduction [GO:0051056] |
12082090_ERK activation by cAMP does not require RAP1 12172908_evidence of differential Krit1 and Rap1A expression during mouse ontogenesis and suggest a more widespread functional significance of Krit1, not restricted to vascular endothelial cells. 12873387_Rap1 couples cAMP signaling to a distinct pool of p42/44MAPK regulating excitability, synaptic plasticity, learning, and memory. 14643017_Expression studies show that mr-gef is expressed by young neurons of the developing rodent CNS but not by progenitor cells in the ventricular zone (VZ). 15544853_down-regulation of Rap1 expression following somatostatin treatment is closely related to the diminished LFA-1 expression 16172130_forskolin-induced cellular proliferation in macrophages by protein kinase A/cAMP-response element-binding protein (CREB) and Epac1-Rap1 signaling 16182283_EGF-induced activation of GTPase Rap1 was sensitized in NIH3T3/EGFR-e3B1 cells. 16518411_Rap1 stimulates signal transduction pathways that lead to cell proliferation and cell survival, and may play crucial roles in leukemogenesis by BCR/ABL. 16802603_CTLA-4 deficient mice show altered RAP-1-mediated adhesion 16980616_A signaling module comprising the cytosolic adapter proteins ADAP and SKAP55 is involved in TCR-mediated inside-out signaling and, the interaction between ADAP and SKAP55 is mandatory for integrin activation. 17003042_Rap1 and MEKK2 are critical upstream signaling molecules mediating BDNF stimulation of ERK5 in central nervous system neurons 17254020_Ras plays a major role in ERK activation by cAMP, PACAP, and KCl, while Rap1 also mediates activation of a selective membrane-associated pool of ERKs. 17314400_functional role of Rap1 in male germ cell differentiation 17403904_Results describe a link between ADAP/SKAP-55 and the Rap1/RIAM complex and provide a potential new mechanism for T cell receptor-mediated integrin activation. 17532477_NF-kappaB activation could be regulated through Epac1-mediated Rap1 stimulation for LPS-induced inflammatory responses in murine macrophages 17538012_These findings proved a crucial role of the cross-talk between two Ras-family GTPases M-Ras and Rap1, mediated by RA-GEF-2, in adhesion signaling. 17827392_CD44 stimulation induces inside-out activation of CR3 through the GTPase Rap1 18049479_The transport of the signaling complex containing the p75 receptor and Rap1 may play a role in the effect of MAG (myelin-associated glycoprotein). 18056342_GTPase Rap1 exhibits an inverse pattern of expression to pERK in individual Ag-specific primed and tolerized T cells. 19218462_phosphorylation of Rap1GAP by protein kinase A plays an important role in striatal neurons by modulating Rap1 actions 21460216_Ca2+-dependent monomer and dimer formation switches CAPRI Protein between Ras GTPase-activating protein (GAP) and RapGAP activities 22253263_Plexins are GTPase-activating proteins for Rap and are activated by induced dimerization 24642466_increased podocyte expression of RAP1GAP contributes directly to podocyte dysfunction by a mechanism that involves loss of RAP1-mediated activation of beta1 integrin |
ENSG00000076864 |
RAP1GAP |
645.422965 |
0.4038614686 |
-1.308068 |
0.11683683 |
123.739158 |
0.00000000000000000000000000009607893392051373745522736122246349624641460733640580564488039344065735454666031534198822328107780776917934417724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000067441212971302540941402850279142284176551019156904429142706200912837661426424418209535360801964998245239257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
354.290907024144 |
28.723666106415 |
877.258502092492 |
50.918824535614 |
| ENSMUSG00000041361 |
102371 |
Myzap |
protein_coding |
Q3UIJ9 |
FUNCTION: Plays a role in cellular signaling via Rho-related GTP-binding proteins and activation of transcription factor SRF. Targets TJP1 to cell junctions (By similarity). In cortical neurons, may play a role in glutaminergic signal transduction through interaction with the NMDA receptor subunit GRIN1 (By similarity). {ECO:0000250, ECO:0000269|PubMed:20093627}. |
Cell junction;Cell membrane;Coiled coil;Cytoplasm;Cytoskeleton;Membrane;Reference proteome;Signal |
|
|
mmu:102371; |
anchoring junction [GO:0070161]; cytoskeleton [GO:0005856]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; I band [GO:0031674]; membrane [GO:0016020]; nuclear envelope [GO:0005635]; RNA polymerase II, core complex [GO:0005665]; RNA polymerase II, holoenzyme [GO:0016591]; Z disc [GO:0030018]; intracellular signal transduction [GO:0035556] |
20093627_Reveal myozap as a previously unrecognized component of a Rho-dependent signaling pathway that links the intercalated disc to cardiac gene regulation. 24698889_Cardiac overexpression of Myozap results in cardiomyopathy with hypertrophy and left ventricular dilation. |
ENSG00000263155 |
MYZAP |
115.280809 |
2.9583219763 |
1.564779 |
0.38340002 |
15.662911 |
0.00007569404309743730510304238201513271633302792906761169433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004621396283356402740448432009401358300237916409969329833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
173.062011002216 |
46.5349575890765 |
58.500059285818 |
12.0125045299697 |
| ENSMUSG00000041372 |
330406 |
B4galnt3 |
protein_coding |
Q6L8S8 |
FUNCTION: Transfers N-acetylgalactosamine (GalNAc) from UDP-GalNAc to N-acetylglucosamine-beta-benzyl with a beta-1,4-linkage to form N,N'-diacetyllactosediamine, GalNAc-beta-1,4-GlcNAc structures in N-linked glycans and probably O-linked glycans. Mediates the N,N'-diacetyllactosediamine formation on gastric mucosa (By similarity). {ECO:0000250}. |
Glycosyltransferase;Golgi apparatus;Membrane;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
|
|
mmu:330406; |
Golgi apparatus [GO:0005794]; Golgi cisterna membrane [GO:0032580]; intracellular membrane-bounded organelle [GO:0043231]; acetylgalactosaminyltransferase activity [GO:0008376]; glucuronosyl-N-acetylgalactosaminyl-proteoglycan 4-beta-N-acetylgalactosaminyltransferase activity [GO:0047238]; N-acetyl-beta-glucosaminyl-glycoprotein 4-beta-N-acetylgalactosaminyltransferase activity [GO:0033842]; chondroitin sulfate biosynthetic process [GO:0030206] |
17579116_up-regulation of beta4GalNAc-T3 may play a critical role in promoting tumor malignanc 22722937_Molecular basis for protein-specific transfer of N-acetylgalactosamine to N-linked glycans by the glycosyltransferases beta1,4-N-acetylgalactosaminyl transferase 3 (beta4GalNAc-T3) and beta4GalNAc-T4. 22722940_Peptide-specific transfer of N-acetylgalactosamine to O-linked glycans by the glycosyltransferases beta1,4-N-acetylgalactosaminyl transferase 3 (beta4GalNAc-T3) and beta4GalNAc-T4. |
ENSG00000139044 |
B4GALNT3 |
183.723847 |
0.2052888713 |
-2.284273 |
0.20979361 |
117.615665 |
0.00000000000000000000000000210461813736075031191971501697084758983900461310939154282198036397930789639237891819334436149802058935165405273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000001346955607910880199628617610861342457496962952390010587406067432946757053691122507643740391358733177185058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
61.9273097837068 |
9.03710821785132 |
301.659361160778 |
29.6642602747477 |
| ENSMUSG00000041498 |
381293 |
Kif14 |
protein_coding |
L0N7N1 |
FUNCTION: Microtubule motor protein that binds to microtubules with high affinity through each tubulin heterodimer and has an ATPase activity (PubMed:24949858). Plays a role in many processes like cell division, cytokinesis and also in cell proliferation and apoptosis (By similarity). During cytokinesis, targets to central spindle and midbody through its interaction with PRC1 and CIT respectively (By similarity). Regulates cell growth through regulation of cell cycle progression and cytokinesis. During cell cycle progression acts through SCF-dependent proteasomal ubiquitin-dependent protein catabolic process which controls CDKN1B degradation, resulting in positive regulation of cyclins, including CCNE1, CCND1 and CCNB1 (By similarity). During late neurogenesis, regulates the cerebellar and cerebral cortex development and olfactory bulb development through regulation of apoptosis, cell proliferation and cell division (PubMed:23308235, PubMed:24931760). Also is required for chromosome congression and alignment during mitotic cell cycle process (By similarity). Regulates cell spreading, focal adhesion dynamics, and cell migration through its interaction with RADIL resulting in regulation of RAP1A-mediated inside-out integrin activation by tethering RADIL on microtubules (By similarity). {ECO:0000250|UniProtKB:Q15058, ECO:0000269|PubMed:23308235, ECO:0000269|PubMed:24931760, ECO:0000269|PubMed:24949858}. |
3D-structure;ATP-binding;Coiled coil;Cytoplasm;Cytoskeleton;Microtubule;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:381293; |
cytosol [GO:0005829]; Flemming body [GO:0090543]; kinesin complex [GO:0005871]; membrane [GO:0016020]; microtubule [GO:0005874]; midbody [GO:0030496]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; spindle midzone [GO:0051233]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; PDZ domain binding [GO:0030165]; plus-end-directed microtubule motor activity [GO:0008574]; protein kinase binding [GO:0019901]; tubulin binding [GO:0015631]; activation of protein kinase activity [GO:0032147]; cell division [GO:0051301]; cell proliferation in forebrain [GO:0021846]; cerebellar cortex development [GO:0021695]; cerebellar granular layer structural organization [GO:0021685]; cerebellar Purkinje cell layer structural organization [GO:0021693]; cerebral cortex development [GO:0021987]; establishment of protein localization [GO:0045184]; hippocampus development [GO:0021766]; microtubule-based movement [GO:0007018]; mitotic metaphase plate congression [GO:0007080]; negative regulation of apoptotic process [GO:0043066]; negative regulation of integrin activation [GO:0033624]; negative regulation of neuron apoptotic process [GO:0043524]; olfactory bulb development [GO:0021772]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of cytokinesis [GO:0032467]; proteasome-mediated ubiquitin-dependent protein catabolic process [GO:0043161]; regulation of cell adhesion [GO:0030155]; regulation of cell growth [GO:0001558]; regulation of cell maturation [GO:1903429]; regulation of cell migration [GO:0030334]; regulation of G1/S transition of mitotic cell cycle [GO:2000045]; regulation of G2/M transition of mitotic cell cycle [GO:0010389]; regulation of myelination [GO:0031641]; regulation of neuron apoptotic process [GO:0043523]; regulation of Rap protein signal transduction [GO:0032487]; SCF-dependent proteasomal ubiquitin-dependent protein catabolic process [GO:0031146]; substrate adhesion-dependent cell spreading [GO:0034446] |
23308235_Kif14 mutation causes severe brain malformation and hypomyelination. 24949858_The ADP-bound form of the KIF14 motor domain reveals a dramatically opened ATP-binding pocket. 27270502_Data provide the first evidence that Kif14 can promote tumor formation in susceptible cells in vivo 30385851_this work reveals a novel association of Kif14 overexpression with lymphoma but suggests that Kif14 does not have as prominent a role in initiating cancer in other cell types as it does in accelerating tumour development in response to other oncogenic insults. 34131133_Structural basis of mechano-chemical coupling by the mitotic kinesin KIF14. |
ENSG00000118193 |
KIF14 |
479.694816 |
2.2851334481 |
1.192278 |
0.13650654 |
75.361281 |
0.00000000000000000392002292763786338847775257679265389246177905076175956855522031219152268022298812866210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000001329839403870377155793505912079747457461385699069994492660384821647312492132186889648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
664.459457358449 |
62.9644222795026 |
290.774903278757 |
21.2836514177813 |
| ENSMUSG00000041605 |
67313 |
Inava |
protein_coding |
G3X9Z8 |
Human_homologues FUNCTION: Expressed in peripheral macrophages and intestinal myeloid-derived cells, is required for optimal PRR (pattern recognition receptor)-induced signaling, cytokine secretion, and bacterial clearance. Upon stimulation of a broad range of PRRs (pattern recognition receptor) such as NOD2 or TLR2, TLR3, TLR4, TLR5, TLR7 and TLR9, associates with YWHAQ/14-3-3T, which in turn leads to the recruitment and activation of MAP kinases and NF-kappa-B signaling complexes that amplifies PRR-induced downstream signals and cytokine secretion (PubMed:28436939). In the intestine, regulates adherens junction stability by regulating the degradation of CYTH1 and CYTH2, probably acting as substrate cofactor for SCF E3 ubiquitin-protein ligase complexes. Stabilizes adherens junctions by limiting CYTH1-dependent ARF6 activation (PubMed:29420262). {ECO:0000269|PubMed:28436939, ECO:0000269|PubMed:29420262}. |
Cytoplasm;Proteomics identification;Reference proteome |
|
|
mmu:67313; |
cytoplasm [GO:0005737]; nuclear body [GO:0016604]; adherens junction maintenance [GO:0034334]; innate immune response [GO:0045087]; nucleotide-binding oligomerization domain containing 2 signaling pathway [GO:0070431]; positive regulation of cytokine production involved in immune response [GO:0002720]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of interleukin-1 beta production [GO:0032731]; positive regulation of interleukin-10 production [GO:0032733]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of protein ubiquitination [GO:0031398]; positive regulation of stress-activated MAPK cascade [GO:0032874]; reactive oxygen species biosynthetic process [GO:1903409]; response to muramyl dipeptide [GO:0032495]; response to peptidoglycan [GO:0032494] |
Human_homologues 27980695_Results found that CpG sites of C1orf106, DMBX1, and SIK3 mediate the genetic risk of psoriasis in Chinese Han population. 28436939_IBD-associated polymorphisms in INAVA modulate PRR-initiated signaling, cytokines, and intracellular bacterial clearance, likely contributing to intestinal immune homeostasis. 29420262_C1orf106 regulates adherens junction stability by regulating the degradation of cytohesin-1, a guanine nucleotide exchange factor that controls activation of ARF6. 30355448_INAVA-CUPID exhibits dual functions, coordinated directly by ARNO, that bridge epithelial barrier function with extracellular signals and inflammation. 31022698_Study identifies C1ORF106 as an epithelial cell junction protein, and the loss of C1ORF106 augments TNF-alpha-induced intestinal epithelial leakage and diarrhea that may play a critical role in the development of inflammatory bowel disease. 34251416_Small-molecule modulators of INAVA cytosolic condensate and cell-cell junction assemblies. |
ENSG00000163362 |
INAVA |
69.303716 |
42.3620677747 |
5.404701 |
0.57386077 |
113.332944 |
0.00000000000000000000000001824210610770595919657039390981205449773294507049218259498927768378208173760679500219339388422667980194091796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000010896618048336360361435351226057426367106148286010879040072901519000531100900275305320974439382553100585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
135.834693808668 |
55.4670817227376 |
3.20651707870385 |
1.19833985273926 |
| ENSMUSG00000041608 |
215446 |
Entpd3 |
protein_coding |
Q8BFW6 |
FUNCTION: Catalyzes the hydrolysis of nucleoside triphosphates and diphosphates (PubMed:15130768). Has a threefold preference for the hydrolysis of ATP and UTP over ADP and UDP (PubMed:15130768). {ECO:0000269|PubMed:15130768}. |
ATP-binding;Calcium;Cell membrane;Disulfide bond;Glycoprotein;Hydrolase;Magnesium;Membrane;Nucleotide-binding;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:215446; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; ATP binding [GO:0005524]; GDP phosphatase activity [GO:0004382]; nucleoside diphosphate phosphatase activity [GO:0017110]; ribonucleoside triphosphate phosphatase activity [GO:0017111]; UDP phosphatase activity [GO:0045134]; nucleoside diphosphate catabolic process [GO:0009134]; nucleoside triphosphate catabolic process [GO:0009143] |
21233276_the specific localization of NTPDase3 in the digestive system suggests functional roles of the enzyme, in association with NTPDase2 and ecto-5'-nucleotidase, in epithelial functions such as secretion and in enteric neurotransmission. 21807070_The results of this study proposeed that NTPDase3 is a key regulator of nociceptive signaling that also makes an unexpected contribution to innocuous tactile sensation. 24085034_NTPDase3 is the major ectonucleotidase in pancreatic beta-cells in multiple species and modulates insulin secretion by controlling activation of purinergic receptors. 24464643_In bladder cancer disease progression there was a weak immunostaining to NTPDase in bladder samples. 26924460_Mesenteric endothelial cells are primed by schistosomiasis to a pro-inflammatory phenotype characterized by an increased expression of NTPDases 2 and 3, favoring ADP accumulation and mononuclear cell adhesion, possibly contributing to mesenteric inflammation and schistosomiasis morbidity via P2Y1 receptor signaling. 27706204_Results identified the expression of ecto-NTPDase3 in trigeminal nociceptive neurons and demonstrated the presence of functional ecto-ATPase activity in trigeminal nerves and established that ecto-NTPDase3mediates extracellularATP degradation in the nociceptive lamina on the brainstem. 28472257_NTPDase2 and -3 are ecto-enzymes expressed in the enteric nervous system. Both enzymes confer protection against gut inflammation in experimental colitis and exhibit alterations in Crohn's disease. These observations suggest that purinergic signalling modulated by E-NTPDases governs neuro-immune interactions that are relevant in Crohn's disease. 33631144_Global deletion of NTPDase3 protects against diet-induced obesity by increasing basal energy metabolism. |
ENSG00000168032 |
ENTPD3 |
1106.954993 |
2.0586511439 |
1.041699 |
0.09083653 |
130.656410 |
0.00000000000000000000000000000294396829373099136426440565191201980009688913025739593700933045991662483958819628709902005425647075753659009933471679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000002292557777084842947057248275890297302303786258038556280495534464724573659984499518493805680918740108609199523925781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1484.87822776187 |
93.5542951027054 |
721.286961216188 |
34.9085296673365 |
| ENSMUSG00000041642 |
16565 |
Kif21b |
protein_coding |
E9Q0A4 |
Human_homologues FUNCTION: Plus-end directed microtubule-dependent motor protein which displays processive activity. Is involved in regulation of microtubule dynamics, synapse function and neuronal morphology, including dendritic tree branching and spine formation. Plays a role in lerning and memory. Involved in delivery of gamma-aminobutyric acid (GABA(A)) receptor to cell surface. {ECO:0000250|UniProtKB:Q9QXL1}. |
ATP-binding;Coiled coil;Motor protein;Nucleotide-binding;Proteomics identification;Reference proteome;Repeat;WD repeat |
|
|
|
cytoskeleton [GO:0005856]; ATP binding [GO:0005524]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; microtubule-based movement [GO:0007018] |
24086586_TRIM3, also known as BERP, interacts with KIF21B via its RBCC domain. 25172774_KIF21B participates in the delivery of GABAAR transport vesicles into dendrites. 27117409_This study provides insight into the cellular function of KIF21B and the basis for cognitive decline resulting from KIF21B dysregulation. 29847800_KIF21B constrains NBEA dynamics and is present in the NBEA-recycling endosome-NMDA receptor complex. 29949770_KIF21B-mediated Rac1 inactivation is a key molecular event in NMDAR-dependent long-term depression expression underlying cognitive flexibility in fear extinction. 30479371_The knockdown of post-mitotic Kif21B resulted in an increase in EPSCs. |
ENSG00000116852 |
KIF21B |
45.744395 |
0.4181177392 |
-1.258019 |
0.46828190 |
7.142794 |
0.00752657909555763671161088268490857444703578948974609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0263369751397964438344168058847571955993771553039550781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.4600711953889 |
8.7402383067232 |
63.2837804099621 |
15.0832830102323 |
| ENSMUSG00000041649 |
245671 |
Klf8 |
protein_coding |
Q8BLM0 |
FUNCTION: Transcriptional repressor and activator. Binds to CACCC-boxes promoter elements. Also binds the GT-box of cyclin D1 promoter and mediates cell cycle progression at G(1) phase as a downstream target of focal adhesion kinase (FAK) (By similarity). {ECO:0000250}. |
DNA-binding;Isopeptide bond;Metal-binding;Nucleus;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation;Ubl conjugation;Zinc;Zinc-finger |
|
|
mmu:245671; |
aggresome [GO:0016235]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; negative regulation of transcription by RNA polymerase II [GO:0000122]; regulation of transcription by RNA polymerase II [GO:0006357] |
16617055_sumoylation of KLF8 negatively regulates its transcriptional activity and cellular functions 18687676_Klf8 is repressed by Klf3 and activated by Klf1 in vivo. 19488069_KLF8 has two N terminal regulatory regions, one surrounding S165 and K171 and the other being two tandem ZFs, which are critical for the regulation of KLF8 nuclear localization and its cellular functions 20107328_These results identified the KLF8 activation domain located between residues 101-260 where the well-conserved Q118 and Q248 are essential for recruiting p300 and PCAF to activate target gene transcription. 21518760_PARP-1 as a novel KLF8-binding and -regulating protein and provided new insights into the mechanisms underlying the regulation of KLF8 nuclear localization, stability, and functions. 23285057_KLF8 is a key component of the transcription factor network that controls terminal differentiation during adipogenesis. 23716600_KLF3 and KLF8 have overlapping roles in vivo and participate in the silencing of embryonic globin expression during development. 26071188_Klf8 is highly expressed in various regions of the mouse brain and in particular in the neurons, where it was localized in the cell nuclei. |
ENSG00000102349 |
KLF8 |
31.749050 |
5.5896118840 |
2.482748 |
0.49216636 |
25.413826 |
0.00000046259079105774226157502784957664676568356298957951366901397705078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000043414559022745100142176392921111727218885789625346660614013671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
53.1047308535268 |
18.4697898252604 |
9.50061148347329 |
2.71383017239795 |
| ENSMUSG00000041695 |
16518 |
Kcnj2 |
protein_coding |
P35561 |
FUNCTION: Probably participates in establishing action potential waveform and excitability of neuronal and muscle tissues. Inward rectifier potassium channels are characterized by a greater tendency to allow potassium to flow into the cell rather than out of it. Their voltage dependence is regulated by the concentration of extracellular potassium; as external potassium is raised, the voltage range of the channel opening shifts to more positive voltages. The inward rectification is mainly due to the blockage of outward current by internal magnesium. Can be blocked by extracellular barium and cesium. |
3D-structure;Ion channel;Ion transport;Lipoprotein;Membrane;Myristate;Potassium;Potassium transport;Reference proteome;S-nitrosylation;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
|
mmu:16518; |
dendrite [GO:0030425]; dendritic spine [GO:0043197]; glutamatergic synapse [GO:0098978]; Golgi apparatus [GO:0005794]; intercalated disc [GO:0014704]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; postsynaptic membrane [GO:0045211]; rough endoplasmic reticulum [GO:0005791]; smooth endoplasmic reticulum [GO:0005790]; T-tubule [GO:0030315]; voltage-gated potassium channel complex [GO:0008076]; identical protein binding [GO:0042802]; inward rectifier potassium channel activity [GO:0005242]; phosphatidylinositol-4,5-bisphosphate binding [GO:0005546]; voltage-gated potassium channel activity involved in cardiac muscle cell action potential repolarization [GO:0086008]; cardiac muscle cell action potential [GO:0086001]; cardiac muscle cell action potential involved in contraction [GO:0086002]; cellular response to mechanical stimulus [GO:0071260]; magnesium ion transport [GO:0015693]; membrane repolarization during action potential [GO:0086011]; membrane repolarization during cardiac muscle cell action potential [GO:0086013]; positive regulation of potassium ion transmembrane transport [GO:1901381]; potassium ion import across plasma membrane [GO:1990573]; potassium ion transmembrane transport [GO:0071805]; potassium ion transport [GO:0006813]; protein homotetramerization [GO:0051289]; regulation of cardiac muscle cell contraction [GO:0086004]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of ion transmembrane transport [GO:0034765]; regulation of membrane repolarization [GO:0060306]; regulation of skeletal muscle contraction via regulation of action potential [GO:0014861]; relaxation of cardiac muscle [GO:0055119]; relaxation of skeletal muscle [GO:0090076] |
11809752_Modulation of the inward rectifier potassium channel IRK1 by the Ras signaling pathway 11906215_AMPA receptor-mediated modulation of inward rectifier K+ channels in astrocytes of mouse hippocampus. 12203395_K+ influxes from neuronal 'sources' into glial cells are mediated mainly by strongly rectifying Kir channels (Kir2.1), expressed in glial membrane sites where direct glia-neuron interactions occur. 12369836_Single and double mutations in the pore-forming M1-M2 segment of Kir2.1 characterize conductance states of the channel and demonstrate a novel functional role for M1-M2 as a critical structural determinant in adjustments of pore conformations. 12740427_These findings can be accounted for by a model in which the negative charges at E224, E299 and D172 act in a concerted manner to coordinate the surface charge reduction and open channel components of polyamine block. 12923194_macrophage cells expressed outward delayed and inwardly rectifying K+ currents mainly mediated by Kv1.3 and Kir2.1 channels 15271672_significant role of IK1 in cardiac excitability is consistent with adverse effects of IK1 upregulation on cardiac electrical activity. 15304346_Kv1.3 and Kir2.1 expression was impaired in brain during cancer cachexia 15311931_a short sequence located in the N-terminal part of the linker between the two transmembrane segments is essential for high-affinity inhibition by TPN and variability in the region underlies the variation of TPN affinities among eukaryotic Kir channels 15459242_These observations implicate E224 and E299 as allosteric modulators of a fast gate, located at the bundle crossing or below in Kir2.1 channels 15824191_E153 contributes to the conductance properties of Kir2.1 channels by acting as a surface charge 15880743_changes in these proteins and their modification may predispose striatal projection neurons to dysfunction and then degeneratation in Huntington disease 16996082_The data demonstrate novel transcriptional and/or post-transcriptional control of ion channel subunit expression (Kir6.2 and Kir2.1) and raise important questions about the control of regional expression of ion channels. 17437338_The molecular interfaces formed between PSD95 PDZ domains and Kir2.1 tail involve regions outside the previously identified binding motif and may be important for additional channel-specific interactions with associating PDZ-containing proteins. 17546530_mouse heart upregulation of IK1 is proarrhythmic 17640933_Kir2 channels exist in two states having different susceptibilities to internal cationic blockers 17699636_Functional Kir2.1 ion channel currents are present in mouse bone marrow mesenchymal stem cells. 18004877_We speculate that restricted dynamics & preferential Kir2.1 binding to PDZ2 are features that enable SAP97 to function as a scaffold protein, allowing other proteins to bind to other 2 PDZ domains in sufficient proximity to yield productive channelosomes. 18276733_The interaction of PIP(2) with only one Kir2.1 subunit is sufficient for the channel to become available and to open to its full conductance state. Interaction with additional subunits exerts positive cooperativity at multiple levels. 18319624_Results imply that Kir2.1 channels in astrocytes may be involved in buffering K(+) against accumulated extracellular K(+) caused by neuronal hyperexcitability under phathophysiological conditions. 19421833_we describe the transcription profiles of the four HCN genes from embryonic stage E9.5 dpc to postnatal day 120 in the mouse. 20045730_Data suggest that the earlier-than-normal expression of Kir2.1 in hair cells inhibits their spontaneous activity required for spiral ganglion neuron survival and neurite growth. 20568224_HL-1 cardiac myocytes express a current that is characteristic of cardiac inward rectifier K(+) channels, and express K(ir)2.1 mRNA. 20962011_These results suggested that the blocking kinetics depends on driving force to produce driving force-dependent inward rectification when the equilibrium potential for potassium is altered by changing external potassium. 21732368_These results demonstrate that three functional ion channel currents, I(KCa), I(Cl.vol), and I(Kir), are heterogeneously present in 3T3-L1 preadipocytes. I(KCa) and I(Cl.vol) participate in the regulation of cell proliferation. 22348965_Stronger expressions of SCN5a, SCN1b and Kir2.1 were observed in ventricular-like and atrial-like cells compared to that of pacemaker-like cardiomyocytes. 22496522_The data of this study suggested that Kir2.1 is required for I(K1) in type II utricle hair cells. 22949619_An inwardly rectifying K+ channel is required for patterning. 23543060_miR-26 controls the expression of KCNJ2 and may have a role in atrial fibrillation 23625347_Report pentamidine analogue which specifically blocks cardiac KIR2.1 channel, lengthening action potential duration. 23671115_The total Kcnj2 (gene for the Kir2.1 potassium channel) mRNA was reduced in Huntington disease skeletal muscle. 24249731_Kir2.1 mRNA levels were downregulated by LPS, and to a lesser extent by IL-4/IL-13. 24259419_the present work demonstrates that a functional Kir2.1-like channel is expressed in lactating mouse mammary secretory cells. 24633623_A184R mutation in the inner end of the bundle crossing region of Kir2.1 not only abolishes the inward rectifying features of spermine block but also tends to close the channel pore. 25472961_Mouse neutrophils express functional Kir2.1 channels from bone marrow and liver. 26520451_Cellular electrophysiology assays of mouse Kir2.1 and human Kir2.2 indicated that, consistent with simulations, the Leu residue increased the channel responses to phosphatidylinositol diphosphate (PIP2) through increased binding affinity and faster activation kinetics, and the deactivation kinetics decreased upon PIP2 inhibition. 26627720_Consistent with a role of the K(+) current in amplifying the sensory response, entry of protons through the Zn(2+)-sensitive conductance produces a transient block of the KIR2.1 current. 26840527_Our results support the concept that endothelial cell Kir2 channels boost vasodilatory signals that are generated by Ca(2+) -dependent activation of IK and SK channels. 26869275_Intracellular Mg(2+) and SPM therefore may have a synergistic action on the pore-blocking effect, presumably via prohibition of the outward exit of the higher-affinity blocking SPM by the lower-affinity Mg(2+). 27380411_Results suggest that a promyogenic cell adhesion molecule Cdo signaling is critical for Inward rectifier potassium channel Kir2.1 activities in the induction of myogenic differentiation. 27560040_This finding represents the first functional evidence for a significant role of the dystrophin-associated protein complex in the regulation of Kir2.x channels. 27598576_The data suggest that microglial Kir2.1 channels may represent novel therapeutic targets to inhibit excessive reactive oxygen species production by primed microglia in brain pathology. 27696527_Following differentiation with LPS or a combination of LPS and IFN-gamma microglia exhibited high KV 1.3 current densities ( approximately 50 pA/pF at 40 mV) and virtually no KCa 3.1 and Kir currents, while microglia differentiated with IL-4 exhibited large Kir 2.1 currents ( approximately 10 pA/pF at -120 mV). KCa 3.1 currents were generally low 27699887_Three pairs of weak interactions precisely regulate the G-loop gate of Kir2.1 channel. 28148533_Suggest that Kir2.1 channels, in part, account for hyperpolarization and associated absence of tone in urinary bladder arterioles. 28464250_histone H4 hyperacetylation induced by Class I HDACs inhibitors promoted the expression profiles of potassium channels (Kcnj2, Kcnj3, Kcnj5, Kcnj11, and Kcnh2) 29018970_KIR2.1 channels may be involved in the excitation-contraction coupling process required for proper skeletal muscle function. 29502106_Hypercholesterolemia-induced loss of flow-induced vasodilation and atherosclerotic plaque formation in ApoE-deficient mice depend on inwardly rectifying Kir2.1 channels. 30084828_These findings collectively support the concept that Gq-protein-coupled receptor activation functions as a molecular switch to favor capillary TRPV4 activity over Kir2.1 signaling, an observation with potentially profound significance for the control of cerebral blood flow. 30203408_Pretreatment with a selective Kir2.1 inhibitor preferentially inhibited dynamic, but not punctate, allodynia in mice with spared nerve injury. Low doses of a glycine receptor inhibitor selectively induced dynamic allodynia. GABAA antagonist induced only punctate allodynia. Results indicated the involvement of glycinergic transmission in the development of dynamic allodynia. 31349969_Apelin shorten QT interval by inhibiting Kir2.1/inward rectifier potassium currents via a PI3K way which is the mechanism of anti-acute myocardial infarction. 32220118_The inhibition of Kir2.1 potassium channels depolarizes spinal microglial cells, reduces their proliferation, and attenuates neuropathic pain. 32601236_The capillary Kir channel as sensor and amplifier of neuronal signals: Modeling insights on K(+)-mediated neurovascular communication. 32698687_Impairment of Flow-Sensitive Inwardly Rectifying K(+) Channels via Disruption of Glycocalyx Mediates Obesity-Induced Endothelial Dysfunction. 32810216_Impaired cytoplasmic domain interactions cause co-assembly defect and loss of function in the p.Glu293Lys KNCJ2 variant isolated from an Andersen-Tawil syndrome patient. 33669857_Potassium Channels Kv1.3 and Kir2.1 But Not Kv1.5 Contribute to BV2 Cell Line and Primary Microglial Migration. 33875602_PIP2 corrects cerebral blood flow deficits in small vessel disease by rescuing capillary Kir2.1 activity. 34784737_Cholesterol-Induced Suppression of Endothelial Kir Channels Is a Driver of Impairment of Arteriolar Flow-Induced Vasodilation in Humans. 34820829_Alzheimer's disease and cerebrovascular pathology alter inward rectifier potassium (KIR 2.1) channels in endothelium of mouse cerebral arteries. 35694964_Kir2.1 channel regulates macrophage polarization via the Ca2+/CaMK II/ERK/NF-kappaB signaling pathway. |
ENSG00000123700 |
KCNJ2 |
18.738803 |
0.0097508137 |
-6.680262 |
1.34610093 |
36.782921 |
0.00000000132041944119852707735920992280359564929881344141904264688491821289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000183676976514483699278472612643337003035526322491932660341262817382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.350903313323103 |
0.37702587570871 |
35.98708016718 |
13.1782981869889 |
| ENSMUSG00000041731 |
226041 |
Pgm5 |
protein_coding |
Q8BZF8 |
FUNCTION: Component of adherens-type cell-cell and cell-matrix junctions. Has no phosphoglucomutase activity in vitro. {ECO:0000250|UniProtKB:Q15124}. |
Cell adhesion;Cell junction;Cell membrane;Cytoplasm;Cytoskeleton;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:226041; |
adherens junction [GO:0005912]; cell-substrate junction [GO:0030055]; costamere [GO:0043034]; cytoplasmic side of plasma membrane [GO:0009898]; cytosol [GO:0005829]; dystrophin-associated glycoprotein complex [GO:0016010]; focal adhesion [GO:0005925]; intercalated disc [GO:0014704]; sarcolemma [GO:0042383]; spot adherens junction [GO:0005914]; stress fiber [GO:0001725]; Z disc [GO:0030018]; magnesium ion binding [GO:0000287]; carbohydrate metabolic process [GO:0005975]; cell adhesion [GO:0007155]; myofibril assembly [GO:0030239]; striated muscle tissue development [GO:0014706] |
24963132_Aciculin interacts with filamin C and Xin and is essential for myofibril assembly. |
ENSG00000154330 |
PGM5 |
824.278976 |
0.1165702810 |
-3.100728 |
0.16247721 |
327.336818 |
0.00000000000000000000000000000000000000000000000000000000000000000000000036545924770677018153161572191190549376779007548199452805345255644270939489927925838512055378378737033081126155246623997462415978432251763095794027782438070421556635814851288503158320553065863 |
0.0000000000000000000000000000000000000000000000000000000000000000000001210146795884678929418296217579683024520511916676449152138233134107027094886212314380146315751352086236025616193437885270659802628262114904305285740805810743882257166672389942993492972789227 |
Yes |
No |
177.667371699894 |
20.0513338688124 |
1524.12235881503 |
120.680094801683 |
| ENSMUSG00000042097 |
22685 |
Zfp239 |
protein_coding |
P24399 |
FUNCTION: May be involved in transcriptional regulation. |
DNA-binding;Metal-binding;Nucleus;Reference proteome;Repeat;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:22685; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; negative regulation of transcription by RNA polymerase II [GO:0000122]; regulation of transcription by RNA polymerase II [GO:0006357] |
Human_homologues 12409453_In this study, we identify a novel interaction between lamin A/C and hsMOK2 by using the yeast two-hybrid system 16385451_Observational study of gene-disease association. (HuGE Navigator) 17760566_Results indicate that pathogenic mutations in lamin A/C lead to sequestration of hsMOK2 into nuclear aggregates, which may deregulate MOK2 target genes. 19490114_Data identified Ser38 and Ser129 of hsMOK2 as phosphorylation sites of JNK3 kinase, and Ser46 as a phosphorylation site of Aurora A and protein kinase A. 19913121_Observational study of gene-disease association. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20641033_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) |
ENSG00000196793 |
ZNF239 |
146.048760 |
2.7875110259 |
1.478978 |
0.22915795 |
41.313603 |
0.00000000012966342533058032872527270969691256738420825911362044280394911766052246093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000020340198708912457642019728886037185922042169750056928023695945739746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
211.446578316504 |
33.727623133042 |
75.8549746901428 |
9.44003914341673 |
| ENSMUSG00000042182 |
320705 |
Bend6 |
protein_coding |
Q6PFX2 |
FUNCTION: Acts as a corepressor of recombining binding protein suppressor hairless (RBPJ) and inhibits Notch signaling in neural stem cells, thereby opposing their self-renewal and promoting neurogenesis (PubMed:23571214, PubMed:25561495). {ECO:0000269|PubMed:23571214, ECO:0000269|PubMed:25561495}. |
Alternative splicing;Coiled coil;Neurogenesis;Nucleus;Reference proteome;Repressor;Transcription;Transcription regulation |
|
|
mmu:320705; |
nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; transcription corepressor activity [GO:0003714]; negative regulation of Notch signaling pathway [GO:0045746]; nervous system development [GO:0007399]; positive regulation of neuron differentiation [GO:0045666] |
23571214_BEND6 knock down increases neural stem cell self-renewal in neocortex. |
ENSG00000151917 |
BEND6 |
310.459633 |
0.4789112303 |
-1.062170 |
0.33056427 |
10.168649 |
0.00142848896275835426927791615980822825804352760314941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0063487335736450295486932304811489302664995193481445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
187.100671942819 |
43.2464241057006 |
390.679232620756 |
66.7292080451823 |
| ENSMUSG00000042254 |
214425 |
Cilp |
protein_coding |
A0A0R4J0F8 |
Human_homologues FUNCTION: Probably plays a role in cartilage scaffolding. May act by antagonizing TGF-beta1 (TGFB1) and IGF1 functions. Has the ability to suppress IGF1-induced proliferation and sulfated proteoglycan synthesis, and inhibits ligand-induced IGF1R autophosphorylation. May inhibit TGFB1-mediated induction of cartilage matrix genes via its interaction with TGFB1. Overexpression may lead to impair chondrocyte growth and matrix repair and indirectly promote inorganic pyrophosphate (PPi) supersaturation in aging and osteoarthritis cartilage. {ECO:0000269|PubMed:12746903, ECO:0000269|PubMed:15864306}. |
Glycoprotein;Proteomics identification;Reference proteome;Secreted;Signal |
|
|
mmu:214425; |
collagen-containing extracellular matrix [GO:0062023]; extracellular space [GO:0005615]; negative regulation of gene expression [GO:0010629]; negative regulation of insulin-like growth factor receptor signaling pathway [GO:0043569]; negative regulation of SMAD protein signal transduction [GO:0060392]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512] |
24631904_Overexpression of CILP in the nucleus pulposus cells promotes disc degeneration, indicating that CILP plays a direct role in the pathogenesis of lumbar disc disease. 29438665_CILP-1 is a novel extracellular matrix protein possessing anti-fibrotic ability in pressure overload-induced fibrotic remodeling. 34610755_Matricellular Protein Cilp1 Promotes Myocardial Fibrosis in Response to Myocardial Infarction. |
ENSG00000138615 |
CILP |
30.370200 |
0.2967191358 |
-1.752830 |
0.51984364 |
11.229459 |
0.00080509090592449216589043814451542857568711042404174804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0038032223320501028912543617366281978320330381393432617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.1023805690295 |
5.19216105152448 |
47.5277084228246 |
12.1549016670401 |
| ENSMUSG00000042286 |
192187 |
Stab1 |
protein_coding |
G3X973 |
Human_homologues FUNCTION: Acts as a scavenger receptor for acetylated low density lipoprotein. Binds to both Gram-positive and Gram-negative bacteria and may play a role in defense against bacterial infection. When inhibited in endothelial tube formation assays, there is a marked decrease in cell-cell interactions, suggesting a role in angiogenesis. Involved in the delivery of newly synthesized CHID1/SI-CLP from the biosynthetic compartment to the endosomal/lysosomal system. {ECO:0000269|PubMed:12077138, ECO:0000269|PubMed:16357325}. |
Disulfide bond;EGF-like domain;Laminin EGF-like domain;Membrane;Proteomics identification;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:192187; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; hyaluronic acid binding [GO:0005540]; low-density lipoprotein particle binding [GO:0030169]; low-density lipoprotein particle receptor activity [GO:0005041]; scavenger receptor activity [GO:0005044]; cell adhesion [GO:0007155]; cell-cell signaling [GO:0007267]; defense response to bacterium [GO:0042742]; negative regulation of angiogenesis [GO:0016525] |
15907457_analysis of mouse fasciclin-containing genes (betaigH3, periostin, stabilin-1, and stabilin-2) expression patterns during valvuloseptal morphogenesis 19830743_Results indicate that anti-Clever-1/Stabilin-1 treatment can target two different arms of the vasculature--traffic via lymphatics and inflamed blood vessels. 21293057_Data suggest that stabilin-1 and -2 together guarantee proper hepatic clearance of potentially noxious agents in the blood and maintain tissue homeostasis not only in the liver but also distant organs. 21427291_we show that stabilin-1 and stabilin-2 in hepatic sinusoidal endothelial cells (HSECs) are critical in effectively clearing damaged RBCs in mouse liver 22334667_a low pH microenvironment up-regulates stabilin-1 expression in macrophage 22960114_We investigated stabilin-1 expressing immune cells in the tumor environment and showed a transient population of alternatively activated macrophages expressing it in the tumor environment and the disappearance of that population as the tumor progresses. 25320356_This work identifies Clever-1 in type II macrophages and in tumor vasculature as a new immunosuppressive molecule in cancer. 27105498_Results suggest stabilin-1 mediated silent clearance of extracellular tumor growth-inhibiting factors as a mechanism of stabilin-1 induced tumor growth. 27977633_Data show that stabilin-1 and stabilin-2 differentially expressed in isolated pancreatic Islets. 30235477_these findings indicated that STAB-1 regulated and maintained vascular integrity through the clearance of infected apoptotic endothelial cells 30312979_this study demonstrated the important role of macrophage STAB-1 in protecting against vascular barrier dysfunction in sepsis 30349531_Data show that the excessive inflammatory activity of monocytes/macrophages in the absence of Clever-1/Stabilin-1 results in augmented humoral immune responses in vivo. 30658191_Bone marrow-specific Stabilin-1 deletion does not affect the susceptibility for atherosclerosis in Ldl receptor knockout mice. 31025051_Ablation of Stabilin-1 Enhances Bone-Resorbing Activity in Osteoclasts In Vitro. 32595212_New tools to prevent cancer growth and spread: a 'Clever' approach. 33008099_Identifying Stabilin-1 and Stabilin-2 Double Knockouts in Reproduction and Placentation: A Descriptive Study. 33130055_Exploring the transcriptomic network of multi-ligand scavenger receptor Stabilin-1- and Stabilin-2-deficient liver sinusoidal endothelial cells. 33746947_Lymphatic Endothelial Cell Activation and Dendritic Cell Transmigration Is Modified by Genetic Deletion of Clever-1. 34374322_Stabilin-1 plays a protective role against Listeria monocytogenes infection through the regulation of cytokine and chemokine production and immune cell recruitment. |
ENSG00000010327 |
STAB1 |
240.971957 |
2.0879940757 |
1.062118 |
0.26586337 |
15.751817 |
0.00007221853044954658405827491174377996685507241636514663696289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004435615547610861127830750305633955576922744512557983398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
342.563298987497 |
63.4863169266262 |
164.063348157296 |
23.1695846915733 |
| ENSMUSG00000042340 |
13019 |
Ctf1 |
protein_coding |
Q60753 |
FUNCTION: Induces cardiac myocyte hypertrophy in vitro. Binds to and activates the ILST/gp130 receptor. |
Cytokine;Reference proteome;Secreted |
|
|
mmu:13019; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; cytokine activity [GO:0005125]; leukemia inhibitory factor receptor binding [GO:0005146]; cell surface receptor signaling pathway [GO:0007166]; leukemia inhibitory factor signaling pathway [GO:0048861]; nervous system development [GO:0007399]; neuron development [GO:0048666]; positive regulation of tyrosine phosphorylation of STAT protein [GO:0042531] |
15023522_Treatment of embryoid bodies grown from pluripotent murine embryonic stem (ES) cells with cardiotrophin 1 significantly stimulated cardiomyogenesis and increased nuclear expression of the proliferation marker Ki-67. 15716414_In cntf/lif/ct-1 triple-knock-out mice, various degrees of muscle fiber type grouping are found, showing that denervation & reinnervation had occurred. CNTF, LIF, & CT-1 have distinct functions for motoneuron survival and function. 16242406_The embryonic neurons of mice regulate the onset of cortical gliogenesis via cardiotrophin-1. 16467531_In embryonic deficient mice, preganglionic sympathetic neurons were reduced. 17150369_Induction of interleukin-6 mRNA after infarct was significantly abrogated in CT-1 null mice compared to wild-type mice 17178916_CT-1 is an essential endogenous defense of the liver against ischemia-reperfusion injury and is a key mediator of the protective effect induced by ischemic preconditioning. 17326158_Our data show that CT-1 is a natural defense of the liver against apoptosis. This cytokine may have therapeutic potential. 17940213_adipose tissue can be recognized as a source of CT-1, which could account for the high circulating levels of CT-1 in patients with metabolic syndrome X 18566129_aldosterone induces CT-1 expression in adult HL-1 cardiomyocytes via genomic and nongenomic mechanisms 18665789_Cardiotrophin-1 is an osteoclast-derived stimulus of bone formation required for normal bone remodeling 21771897_Hypoxia increased cardiotrophin-1 levels in cardiac cells through a direct regulation of CT-1 promoter by HIF-1alpha, protecting cells from apoptosis. 21803294_We conclude that CT-1 is a master regulator of fat and glucose metabolism with potential applications for treatment of obesity and insulin resistance. 21926338_Data suggest a key role for CT-1 in cardiac remodeling induced by aldosterone independent of changes in blood pressure levels. 23172930_CT-1 may be a major regulator of arterial stiffness with a major impact on the aging process. 23358882_CT-1 improves beta cell function and survival, and protects mice against STZ-induced diabetes 23541490_The transgenic expression of CTF1 brought about a marked improvement on cognitive functioning in the APPswe/PS1dE9 transgenic mouse model of Alzheimer's disease. 24362075_Cardiotrophin-1 (CT-1) is a hepatoprotective cytokine that modulates fat and glucose metabolism. Here we analyzed the changes in hepatic fat stores induced by recombinant CT-1 and its therapeutic potential in non-alcoholic fatty liver disease. 25351614_Data suggest that Ctf1 up-regulates lipolysis in white adipocytes via 1) induction of perilipin, 2) activation of hormone sensitive lipase (via phosphorylation by PKA), and 3) inactivation of adipose triglyceride lipase (via up-regulation of G0S2). 26005169_Nuclear translocation of CT-1 regulates cardiomyogenesis of ES cells and involves calcium, NO, ROS as well as CT-1 regulated signaling pathways. 27608275_Cardiotrophin-1 modulates the production of adipokines in vitro and in vivo, suggesting that the regulation of the secretory function of adipocytes could be involved in the metabolic actions of this cytokine. 28096235_CT-1 mRNA levels in adipose tissue showed significant circadian fluctuations in young WT mice. Clock genes displayed a circadian rhythm in adipose tissue of both wild-type (WT) and CT-1-/- mice. However, the pattern was altered in CT-1-/- mice toward a lower percentage of the rhythm or lower amplitude, especially for Bmal1 and Clock. 28731433_These results suggest that CT-1 may be a potent candidate for the early prevention and treatment of Alzheimer disease. 28754835_Identify cardiotrophin-1 as a powerful cytokine promoting mesenchymal stromal cell engraftment and thus improving cell therapy of the infarcted myocardium. 30589223_Cardiotrophin-1 participates in the endogenous response that opposes renal damage by counteracting the inflammatory, apoptotic and fibrotic processes 30892966_findings support the notion that cardiotrophin-1 (CT-1) is a critical regulator of inflammation and suggest that rCT-1 could be a molecule with potential therapeutic application in inflammatory conditions 32238841_Cardiotrophin-1 Deficiency Abrogates Atherosclerosis Progression. 33047392_Cardiotrophin-1 contributes to metabolic adaptations through the regulation of lipid metabolism and to the fasting-induced fatty acid mobilization. 35722965_Tumor-derived CTF1 (cardiotrophin 1) is a critical mediator of stroma-assisted and autophagy-dependent breast cancer cell migration, invasion and metastasis. |
ENSG00000150281 |
CTF1 |
37.959145 |
3.7190297978 |
1.894926 |
0.43395080 |
18.990907 |
0.00001313428988172316179612077180482998528532334603369235992431640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000952673826087653297664883944584346409101272001862525939941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
61.9329666075324 |
18.9111113046645 |
16.6529901547795 |
4.04168921965668 |
| ENSMUSG00000042379 |
71690 |
Esm1 |
protein_coding |
Q9QYY7 |
FUNCTION: Involved in angiogenesis; promotes angiogenic sprouting. May have potent implications in lung endothelial cell-leukocyte interactions (By similarity). {ECO:0000250}. |
Angiogenesis;Glycoprotein;Proteoglycan;Reference proteome;Secreted;Signal |
|
|
mmu:71690; |
extracellular region [GO:0005576]; hepatocyte growth factor receptor binding [GO:0005171]; integrin binding [GO:0005178]; angiogenesis [GO:0001525]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of hepatocyte growth factor receptor signaling pathway [GO:1902204]; sprouting angiogenesis [GO:0002040] |
16956626_Results suggest that endocan (ESM-1)is preferentially expressed in tumor endothelium in vivo and that its expression is regulated by tumor-derived factors. 20516397_A paracrine vasoregulatory pathway mediated by endothelial cell-derived ET-1 acting on the vascular smooth muscle ET(A) receptor. 22370430_Mouse endocan serum level was measured at a median of 0.96 ng/mL and 1.08 ng/mL in 129Sv mice and C57bl6, respectively. 24055127_endocan is normally present in the mouse brain and prior vascular perfusion with FITC may provide a useful tool for identify newly forming blood vessels. 24446198_This study shows endocan can elicit severe inflammatory responses and inhibiting endocan release offers a potential strategy for treating sepsis. 25057127_Esm1 modulates endothelial tip cell behavior and vascular permeability by enhancing VEGF bioavailability. 25575808_Results reveal a novel pathway for endocan in the control of tumor growth, which involves inflammatory cells of the innate immunity. 27683113_Data from murine model indicate that NGFR plays an important role in the pathogenesis and progression of oral squamous cell carcinoma via regulation of ESM1. 28934365_glomerular-derived Esm-1 as a potential non-invasive biomarker of diabetic nephropathy 36591362_Endothelial Cell-Specific Molecule-1 Inhibits Albuminuria in Diabetic Mice. |
ENSG00000164283 |
ESM1 |
253.979781 |
0.3553930376 |
-1.492513 |
0.18102270 |
67.251585 |
0.00000000000000023897854209396802270857520565202290234895399125697457165529158373828977346420288085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000068423329946904529022276745839848560152399756066621261396676345611922442913055419921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
132.088507619965 |
16.6173793151401 |
371.668810706117 |
33.3051172386888 |
| ENSMUSG00000042436 |
76293 |
Mfap4 |
protein_coding |
Q9D1H9 |
FUNCTION: Could be involved in calcium-dependent cell adhesion or intercellular interactions. May contribute to the elastic fiber assembly and/or maintenance. {ECO:0000250|UniProtKB:P55083}. |
Alternative splicing;Calcium;Cell adhesion;Extracellular matrix;Glycoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:76293; |
collagen-containing extracellular matrix [GO:0062023]; elastic fiber [GO:0071953]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; microfibril [GO:0001527]; antigen binding [GO:0003823]; carbohydrate derivative binding [GO:0097367]; signaling receptor binding [GO:0005102]; cell adhesion [GO:0007155]; cellular response to UV-B [GO:0071493]; complement activation, lectin pathway [GO:0001867]; elastic fiber assembly [GO:0048251]; regulation of collagen metabolic process [GO:0010712]; supramolecular fiber organization [GO:0097435]; UV protection [GO:0009650] |
26033354_Mfap4 knockout mice develop a spontaneous loss of lung function. 26038533_MFAP4 promoted the development of asthmatic airway disease in vivo and pro-asthmatic functions of bronchial smooth muscle cells in vitro 26564819_MFAP4 regulates integrin alphaVbeta3-induced VSMC proliferation and migration, as well as monocyte chemotaxis, and accelerates neointimal hyperplasia after vascular injury. 26899386_MFAP4 promotes follicular dendritic cell development in regenerating splenic tissue. 30677223_An integrative systems approach identifies IGFBP-2, Mfap4, and Spp1 as novel candidates in Marfan syndrome-related pathophysiology. 32856514_Deletion of Microfibrillar-Associated Protein 4 Attenuates Left Ventricular Remodeling and Dysfunction in Heart Failure. 32905637_MFAP4 deficiency alleviates renal fibrosis through inhibition of NF-kappaB and TGF-beta/Smad signaling pathways. 33530700_Microfibrillar-Associated Protein 4 Regulates Stress-Induced Cardiac Remodeling. |
ENSG00000166482 |
MFAP4 |
130.515746 |
0.4981166709 |
-1.005444 |
0.22341654 |
20.167665 |
0.00000709420364438967991041907340532546299982641357928514480590820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000539485321574356501252503481858013856253819540143013000488281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
87.0800221468911 |
13.5392244389095 |
174.818525920563 |
19.5380349497133 |
| ENSMUSG00000042500 |
76850 |
Ago4 |
protein_coding |
Q8CJF8 |
FUNCTION: Required for RNA-mediated gene silencing (RNAi). Binds to short RNAs such as microRNAs (miRNAs) and represses the translation of mRNAs which are complementary to them. Lacks endonuclease activity and does not appear to cleave target mRNAs. {ECO:0000255|HAMAP-Rule:MF_03033, ECO:0000269|PubMed:19174539}. |
Alternative splicing;Cytoplasm;Reference proteome;Ribonucleoprotein;RNA-binding;RNA-mediated gene silencing;Translation regulation;Ubl conjugation |
|
|
mmu:76850; |
cytoplasm [GO:0005737]; cytoplasmic ribonucleoprotein granule [GO:0036464]; cytosol [GO:0005829]; nucleus [GO:0005634]; P-body [GO:0000932]; RISC complex [GO:0016442]; RISC-loading complex [GO:0070578]; double-stranded RNA binding [GO:0003725]; endoribonuclease activity [GO:0004521]; miRNA binding [GO:0035198]; RNA binding [GO:0003723]; single-stranded RNA binding [GO:0003727]; male gonad development [GO:0008584]; male meiotic nuclear division [GO:0007140]; miRNA metabolic process [GO:0010586]; miRNA processing [GO:0035196]; miRNA-mediated gene silencing by inhibition of translation [GO:0035278]; mRNA catabolic process [GO:0006402]; negative regulation of apoptotic process [GO:0043066]; pre-miRNA processing [GO:0031054]; regulation of cell morphogenesis [GO:0022604]; RISC complex assembly [GO:0070922]; RNA secondary structure unwinding [GO:0010501]; synaptonemal complex assembly [GO:0007130] |
22863743_show that Ago4(-/-) spermatogonia initiate meiosis early, resulting from premature induction of retinoic acid-response genes 26995025_up-regulated in murine macrophage RAW264.7 cells transfected by Echinococcus multilocularis miR-71 |
ENSG00000134698 |
AGO4 |
195.409146 |
0.2426247382 |
-2.043201 |
0.74149353 |
6.561658 |
0.01041992566529401260944087681536984746344387531280517578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0346617837374445098008735044459172058850526809692382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
62.3462235625823 |
33.4732486696594 |
256.965650029161 |
100.788621509624 |
| ENSMUSG00000042501 |
329093 |
Cpa6 |
protein_coding |
Q5U901 |
FUNCTION: May be involved in the proteolytic inactivation of enkephalins and neurotensin in some brain areas. May convert inactive angiotensin I into the biologically active angiotensin II. Releases a C-terminal amino acid, with preference for large hydrophobic C-terminal amino acids and shows only very weak activity toward small amino acids and histidine. {ECO:0000250}. |
Alternative splicing;Carboxypeptidase;Cleavage on pair of basic residues;Disulfide bond;Extracellular matrix;Glycoprotein;Hydrolase;Metal-binding;Metalloprotease;Protease;Reference proteome;Secreted;Signal;Zinc;Zymogen |
|
This gene encodes a protein that belongs to the metallocarboxypeptidase family of proteins that catalyze the release of a C-terminal amino acid from the target protein. The encoded preproprotein undergoes proteolytic cleavage to yield the mature form which is thought to play a role in cell migration. In humans, this protein regulates neuropeptides in the brain and mutations in this gene are associated with a recessive familial form of febrile seizures and with temporal lobe epilepsy. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Jan 2014]. |
mmu:329093; |
extracellular space [GO:0005615]; metallocarboxypeptidase activity [GO:0004181]; zinc ion binding [GO:0008270]; proteolysis [GO:0006508] |
15950771_The distribution of Cpa6 suggests a specific role in a limited number of tissues, and it is possible that this role involves an aspect of cell migration. 18178555_CPA6 may have a role in the regulation of neuropeptides in the extracellular environment within the olfactory bulb and other parts of the brain |
ENSG00000165078 |
CPA6 |
129.096135 |
2.2478162685 |
1.168524 |
0.22576021 |
26.503469 |
0.00000026306529583526005438629652953552717775664859800599515438079833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000025661125825587643315762228363174912715294340159744024276733398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
172.569006887784 |
27.114817176349 |
76.7718471075961 |
9.40053503807918 |
| ENSMUSG00000042515 |
245631 |
Pwwp3b |
protein_coding |
Q4VA55 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:245631; |
Human_homologues extracellular exosome [GO:0070062] |
|
ENSG00000157502 |
PWWP3B |
114.181703 |
2.2005721047 |
1.137879 |
0.24105774 |
22.216845 |
0.00000243527875711668781170591256690638459758702083490788936614990234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000202258266239920315196325339979210866658831946551799774169921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
157.741884252156 |
26.4796654041883 |
71.6822156916943 |
9.35460583816014 |
| ENSMUSG00000042659 |
66412 |
Arrdc4 |
protein_coding |
A0A0B4J1F4 |
FUNCTION: Functions as an adapter recruiting ubiquitin-protein ligases to their specific substrates (PubMed:27462458). Plays a role in endocytosis of activated G protein-coupled receptors (GPCRs) (By similarity). Through an ubiquitination-dependent mechanism also plays a role in the incorporation of SLC11A2 into extracellular vesicles (PubMed:27462458). May play a role in glucose uptake (By similarity). {ECO:0000250|UniProtKB:Q8NCT1, ECO:0000269|PubMed:27462458}. |
Alternative splicing;Cell membrane;Cytoplasmic vesicle;Endosome;Membrane;Reference proteome;Repeat |
|
|
mmu:66412; |
cytoplasm [GO:0005737]; early endosome [GO:0005769]; endosome [GO:0005768]; extracellular vesicle [GO:1903561]; intracellular membrane-bounded organelle [GO:0043231]; plasma membrane [GO:0005886]; ubiquitin ligase-substrate adaptor activity [GO:1990756]; extracellular vesicle biogenesis [GO:0140112]; positive regulation of ubiquitin-protein transferase activity [GO:0051443]; protein transport [GO:0015031]; protein ubiquitination [GO:0016567] |
19605364_Txnip regulates cellular metabolism independent of its binding to thioredoxin and the arrestin domains are crucial structural elements in metabolic functions of alpha-arrestin proteins 34188787_Arrdc4-dependent extracellular vesicle biogenesis is required for sperm maturation. 34916487_TCF4 and HuR mediated-METTL14 suppresses dissemination of colorectal cancer via N6-methyladenosine-dependent silencing of ARRDC4. 35950500_Interaction of ARRDC4 With GLUT1 Mediates Metabolic Stress in the Ischemic Heart. |
ENSG00000140450 |
ARRDC4 |
60.292653 |
0.4472533603 |
-1.160836 |
0.37283618 |
9.313372 |
0.00227487372535398277989537874077541346196085214614868164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0095116323317573046869366493183406419120728969573974609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.4830126055221 |
10.0562488339452 |
86.0429815029246 |
16.2837500079893 |
| ENSMUSG00000042751 |
226518 |
Nmnat2 |
protein_coding |
Q8BNJ3 |
FUNCTION: Nicotinamide/nicotinate-nucleotide adenylyltransferase that acts as an axon maintenance factor (PubMed:20126265, PubMed:23082226). Axon survival factor required for the maintenance of healthy axons: acts by delaying Wallerian axon degeneration, an evolutionarily conserved process that drives the loss of damaged axons (PubMed:20126265, PubMed:23082226, PubMed:25818290). Catalyzes the formation of NAD(+) from nicotinamide mononucleotide (NMN) and ATP (By similarity). Can also use the deamidated form; nicotinic acid mononucleotide (NaMN) as substrate but with a lower efficiency. Cannot use triazofurin monophosphate (TrMP) as substrate (By similarity). Also catalyzes the reverse reaction, i.e. the pyrophosphorolytic cleavage of NAD(+). For the pyrophosphorolytic activity prefers NAD(+), NADH and NaAD as substrates and degrades nicotinic acid adenine dinucleotide phosphate (NHD) less effectively (By similarity). Fails to cleave phosphorylated dinucleotides NADP(+), NADPH and NaADP(+) (By similarity). Also acts as an activator of ADP-ribosylation by supporting the catalytic activity of PARP16 and promoting mono-ADP-ribosylation of ribosomes by PARP16 (By similarity). {ECO:0000250|UniProtKB:Q9BZQ4, ECO:0000269|PubMed:20126265, ECO:0000269|PubMed:23082226, ECO:0000269|PubMed:25818290}. |
ATP-binding;Cell projection;Cytoplasm;Cytoplasmic vesicle;Golgi apparatus;Lipoprotein;Membrane;NAD;Nucleotide-binding;Nucleotidyltransferase;Palmitate;Pyridine nucleotide biosynthesis;Reference proteome;Transferase;Ubl conjugation |
PATHWAY: Cofactor biosynthesis; NAD(+) biosynthesis; NAD(+) from nicotinamide D-ribonucleotide: step 1/1. {ECO:0000250|UniProtKB:Q9BZQ4}.; PATHWAY: Cofactor biosynthesis; NAD(+) biosynthesis; deamido-NAD(+) from nicotinate D-ribonucleotide: step 1/1. {ECO:0000250|UniProtKB:Q9BZQ4}. |
|
mmu:226518; |
axon [GO:0030424]; cytoplasm [GO:0005737]; cytoplasmic vesicle membrane [GO:0030659]; cytosol [GO:0005829]; Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; late endosome [GO:0005770]; synapse [GO:0045202]; trans-Golgi network [GO:0005802]; ATP binding [GO:0005524]; nicotinamide-nucleotide adenylyltransferase activity [GO:0000309]; nicotinate-nucleotide adenylyltransferase activity [GO:0004515]; protein ADP-ribosyltransferase-substrate adaptor activity [GO:0140768]; NAD biosynthetic process [GO:0009435] |
19778564_The brain-specific Nmnat2 delays injury-induced axon degeneration dependent on its NAD synthesis activity. These findings provide new clues to further study the molecular mechanisms of axon degeneration and the related neurodegenerative diseases. 20943658_Nmnat2 is a neuronal protein peripherally attached to membranes via palmitoylation and suggest that Nmnat2 is transported to synaptic terminals via an endosomal pathway. 22027994_Decreased endogenous NMNAT2 function caused by reduced CREB signaling during pathological insults may be one of underlying mechanisms for neuronal death in tauopathies 23082226_Nmnat2 is involved in axon development or survival in a mammal. 23371862_Complete loss of Nmnat2 leads to a mature but distended bladder in utero and is not compatible with survival. Moderate loss of Nmnat2 has no effect on bladder development, survival, and has only modest effects on bladder function later in life. 23610559_Nmnat2 activity supports axon survival through a site of action distinct from Nmnat2 transport vesicles. 23946398_results suggest an essential role for NMNAT2 during axon growth 23995269_Together, our results establish Nmnat2 localisation and turnover as a valuable target for modulating axon degeneration in vivo. 25271157_Data indicate that thioesterases APT1/APT2 depalmitoylate nicotinamide mononucleotide adenylyltransferase 2 (NMNAT2) and zDHHC17 is the strongest candidate palmitoyltransferase for NMNAT2. 25818290_nicotinamide mononucleotide adenylyltransferase 2-depletion-dependent degeneration of established axons and restricted extension of developing axons are thus both SARM1 dependent 26844829_Active nerve degeneration requires SARM1 and MAP kinases, including DLK, while the NAD+ synthetic enzyme NMNAT2 prevents degeneration. 27732853_Consistent with Skp1a functioning through regulation of Nmnat2, Skp1a knockdown fails to protect axons from Nmnat2 knockdown. 29549647_the deregulated expression of Nmnat2 appears to have a great influence on the cellular stress in the spinal cord of wobbler mice. 30278188_NMNAT2 depletion in the dorsal root ganglion neurons caused Golgi fragmentation and axon degeneration. 30304512_Data reveal that reducing NMNAT2 levels below a particular threshold compromises the development of peripheral axons and increases their vulnerability to stresses. 30909324_The data indicate a role for NMNAT2 in controlling redox homeostasis during oocyte maturation and uncover that NMNAT2- NAD(+) -SIRT1 is an important pathway mediating the effects of maternal age on oocyte developmental competence. 31740269_Mitochondrial impairment activates the Wallerian pathway through depletion of NMNAT2 leading to SARM1-dependent axon degeneration. 33248200_Upregulated Nmnat2 causes neuronal death and increases seizure susceptibility in temporal lobe epilepsy. 34644262_Nmnat2 attenuates amyloidogenesis and up-regulates ADAM10 in AMPK activity-dependent manner. 35114390_NMNAT2 is downregulated in glaucomatous RGCs, and RGC-specific gene therapy rescues neurodegeneration and visual function. 36322391_Loss-of-function approach using mouse retinal explants showed pivotal roles of Nmnat2 in early and middle stages of retinal development. |
ENSG00000157064 |
NMNAT2 |
72.512242 |
0.2635738368 |
-1.923721 |
0.32052483 |
35.567453 |
0.00000000246369680178497500349329170485270651402842645438795443624258041381835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000328320469683191058292284021571244556625401855853851884603500366210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
28.5665300761598 |
6.39899195627674 |
108.381508681984 |
16.6086653344209 |
| ENSMUSG00000042793 |
329252 |
Lgr6 |
protein_coding |
Q3UVD5 |
FUNCTION: Receptor for R-spondins that potentiates the canonical Wnt signaling pathway and acts as a marker of multipotent stem cells in the epidermis. Upon binding to R-spondins (RSPO1, RSPO2, RSPO3 or RSPO4), associates with phosphorylated LRP6 and frizzled receptors that are activated by extracellular Wnt receptors, triggering the canonical Wnt signaling pathway to increase expression of target genes. In contrast to classical G-protein coupled receptors, does not activate heterotrimeric G-proteins to transduce the signal (By similarity). {ECO:0000250}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Leucine-rich repeat;Membrane;Receptor;Reference proteome;Repeat;Signal;Transducer;Transmembrane;Transmembrane helix;Wnt signaling pathway |
|
|
mmu:329252; |
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; vesicle [GO:0031982]; G protein-coupled receptor activity [GO:0004930]; protein-hormone receptor activity [GO:0016500]; bone regeneration [GO:1990523]; positive regulation of cell migration [GO:0030335]; positive regulation of Wnt signaling pathway [GO:0030177]; tissue regeneration [GO:0042246]; Wnt signaling pathway [GO:0016055] |
20223988_study concludes that Lgr6 marks the most primitive epidermal stem cell 22002312_Lgr5 has been identified as a marker of Wnt-regulated adult stem cell populations in the hair-follicle, intestine and stomach. A closely-related protein, Lgr6 marks adult stem cells responsible for fueling the renewal of the sebaceous gland and skin.[Review] 24616052_The members of the R-spondin family are known as activators of Wnt signaling, and Lgr4, Lgr5, and Lgr6 have been identified as receptors for R-spondins. 25368147_Lgr6 is expressed in cells at the basal area of taste buds in fungiform and circumvallate papillae. Lgr6+ cells give rise to taste bud cells in taste papillae in both anterior and posterior tongue. 26362184_Lgr6 and Bmi1 have roles in stem cells in the acral epithelium that participate in the long-term maintenance of sweat glands, ducts, and interadnexal epidermis, while Lgr5 has a role in rapid cycling and maintance of sweat glands only 26460010_Lgr6 marks nail stem cells and is required for digit tip regeneration. 27798604_Lgr6 marks mammary gland progenitor cells that can initiate tumours, and cells of luminal breast tumours required for efficient tumour maintenance. 27880932_Leucine rich repeat containing G protein coupled receptor 6 (Lgr6) mRNA was found to be diminished in skin tumors. 28945253_Lgr6 is a stem cell marker in mouse skin squamous cell carcinoma whose expression is proportional with the disease progression. 31043491_Tumorigenesis was inhibited in Pot1b(-/-);p53(+/+) mice due to cellular senescence. Pot1b(-/-);p53(-/-) tumors also exhibited senescence, but proliferated and metastasized with expansion of Lgr6(+) stem cells indicative of senescence-associated secretory phenotype 31500806_LGR6 is highly expressed in osteoblastic progenitors, and promotes osteogenesis by enhancing beta-catenin stability to strengthen the Wnt signaling pathway. 31657786_LGR6 has a role in stimulating maresin 1's key proresolving functions of phagocytes 34856421_Mouse LGR6 regulates osteogenesis in vitro and in vivo through differential ligand use. 35476447_Mechanical tension mobilizes Lgr6(+) epidermal stem cells to drive skin growth. 36371908_LGR6-dependent conditional inactivation of E-cadherin and p53 leads to invasive skin and mammary carcinomas in mice. |
ENSG00000133067 |
LGR6 |
63.669717 |
0.0462712931 |
-4.433739 |
0.69140667 |
33.346150 |
0.00000000771303688703883203576532252322695004664865336962975561618804931640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000948990128137120198885233338451095086441000603372231125831604003906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.76066040044352 |
2.36987463152058 |
102.885830080972 |
33.6199510208953 |
| ENSMUSG00000042918 |
74490 |
Mamstr |
protein_coding |
Q0ZCJ7 |
FUNCTION: Transcriptional coactivator. Stimulates the transcriptional activity of MEF2C. Stimulates MYOD1 activity in part via MEF2, resulting in an enhancement of skeletal muscle differentiation. {ECO:0000269|PubMed:16818234}. |
Activator;Alternative splicing;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:74490; |
nucleus [GO:0005634]; transcription coregulator activity [GO:0003712]; positive regulation of myotube differentiation [GO:0010831]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
31348492_Pax7, Pax3 and Mamstr genes are involved in skeletal muscle impaired regeneration of dy2J/dy2J mouse model of Lama2-CMD |
ENSG00000176909 |
MAMSTR |
15.742539 |
4.5453356706 |
2.184387 |
0.70810348 |
9.333217 |
0.00225036894826364641114535558585885155480355024337768554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0094298816561078011022623002190812258049845695495605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.8205437880943 |
13.1882411494432 |
5.6806681969364 |
2.30789510290548 |
| ENSMUSG00000043333 |
230726 |
Rhbdl2 |
protein_coding |
A2AGA4 |
FUNCTION: Involved in regulated intramembrane proteolysis and the subsequent release of functional polypeptides from their membrane anchors. Known substrate: EFNB3 (By similarity). {ECO:0000250}. |
Cell membrane;Hydrolase;Membrane;Protease;Reference proteome;Serine protease;Transmembrane;Transmembrane helix |
|
|
mmu:230726; |
plasma membrane [GO:0005886]; serine-type endopeptidase activity [GO:0004252]; proteolysis [GO:0006508] |
21833011_RHBDL2 and soluble thrombomodulin were upregulated in ex vivo tissue culture of injured mouse skin. 3,4-Dichloroisocoumarin inhibited thrombomodulin production and wound healing; this was reversed by recombinant thrombomodulin in mice. |
ENSG00000158315 |
RHBDL2 |
1168.040150 |
0.3715114389 |
-1.428521 |
0.12575173 |
125.320987 |
0.00000000000000000000000000004329350185645761939604418446175228139114942673461310884418879106409588717458130496806845144419639836996793746948242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000031105972654602006590286272155567321014777681077500687040826347863019105859441748584970355295808985829353332519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
591.238616969267 |
51.6002744125366 |
1591.44121820618 |
102.498784932403 |
| ENSMUSG00000043336 |
78749 |
Filip1l |
protein_coding |
Q6P6L0 |
FUNCTION: Acts as a regulator of the antiangiogenic activity on endothelial cells. When overexpressed in endothelial cells, leads to inhibition of cell proliferation and migration and an increase in apoptosis. Inhibits melanoma growth When expressed in tumor-associated vasculature (By similarity). {ECO:0000250}. |
Alternative splicing;Coiled coil;Cytoplasm;Membrane;Nucleus;Phosphoprotein;Reference proteome |
|
|
|
cytoplasm [GO:0005737]; membrane [GO:0016020]; nucleus [GO:0005634] |
21832087_GPBP directs myofibril formation through interaction with intracellular downstream effector 130-kDa GPBP-interacting protein 34417201_FILIP1L Loss Is a Driver of Aggressive Mucinous Colorectal Adenocarcinoma and Mediates Cytokinesis Defects through PFDN1. |
ENSG00000168386 |
FILIP1L |
301.577660 |
0.3686011565 |
-1.439867 |
0.16368222 |
76.217244 |
0.00000000000000000254112986418674052263692496146644360088751220498202911673990556096214277204126119613647460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000885173492810834745997861609829955973000952145000701232202544588290038518607616424560546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
162.272786505679 |
18.4503236211272 |
440.239493682469 |
35.6893298274654 |
| ENSMUSG00000043458 |
93883 |
Pcdhb12 |
protein_coding |
Q91Y07 |
Human_homologues FUNCTION: Potential calcium-dependent cell-adhesion protein. May be involved in the establishment and maintenance of specific neuronal connections in the brain. |
Calcium;Cell adhesion;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:93883; |
cell-cell junction [GO:0005911]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell adhesion [GO:0007155]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
Human_homologues 34419445_Weighted genes associated with the progression of retinoblastoma: Evidence from bioinformatic analysis. |
ENSG00000113209 |
PCDHB5 |
103.691446 |
0.3877875544 |
-1.366662 |
0.28171555 |
23.200381 |
0.00000145968427015216362891227887266243357089479104615747928619384765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000126114071485863621152265223734900700947036966681480407714843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
57.6793302772736 |
11.3347768853975 |
148.739508604119 |
20.8838368278743 |
| ENSMUSG00000043633 |
242408 |
Fam221b |
protein_coding |
Q8C627 |
|
Alternative splicing;Phosphoprotein;Reference proteome |
|
|
mmu:242408; |
|
|
ENSG00000204930 |
FAM221B |
929.437746 |
2.6038292556 |
1.380635 |
0.16936459 |
63.381652 |
0.00000000000000170296849966969941760974663809458329845519569591821840148782030155416578054428100585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000453489793478476589566197300379713745557028019284029340951747144572436809539794921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1361.22401937787 |
160.167632327005 |
522.777757579916 |
46.5504471198438 |
| ENSMUSG00000043795 |
677289 |
Prr33 |
protein_coding |
A0A571BEK1 |
|
Reference proteome |
|
|
|
|
|
ENSG00000283787 |
PRR33 |
118.385027 |
0.2172256654 |
-2.202734 |
0.26752456 |
66.585321 |
0.00000000000000033507199660351620648219164409820371571487705058579875672108983053476549685001373291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000094866480525367258081680220419819086309165881432114986182568827643990516662597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.5628205254258 |
7.93789956545648 |
195.938267490945 |
24.9399429849726 |
| ENSMUSG00000044026 |
240660 |
Slc35g1 |
protein_coding |
Q8BY79 |
FUNCTION: May play a role in intracellular calcium sensing and homeostasis. May act as a negative regulator of plasma membrane calcium-transporting ATPases preventing calcium efflux from the cell (By similarity). {ECO:0000250}. |
Cell membrane;Endoplasmic reticulum;Membrane;Reference proteome;Repeat;Transmembrane;Transmembrane helix |
|
|
mmu:240660; |
endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; plasma membrane [GO:0005886]; calcium ion export across plasma membrane [GO:1990034]; regulation of cytosolic calcium ion concentration [GO:0051480] |
28572627_Study shows that microRNA-150 modulates intracellular Ca (2+) levels in naive CD8(+) T cells by targeting transmembrane protein 20. |
ENSG00000176273 |
SLC35G1 |
7.830215 |
0.0228184474 |
-5.453656 |
1.52278304 |
14.645393 |
0.00012975138768732508522986413002087147106067277491092681884765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007566512776620733498472715083948969549965113401412963867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.336884963698554 |
0.425390415293345 |
14.763711033063 |
8.17499636020601 |
| ENSMUSG00000044033 |
545428 |
Ccdc141 |
protein_coding |
A2AST1 |
Human_homologues FUNCTION: Plays a critical role in radial migration and centrosomal function. {ECO:0000250|UniProtKB:A2AST1}. |
Coiled coil;Proteomics identification;Reference proteome |
|
|
|
axon [GO:0030424]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; plasma membrane [GO:0005886]; cell-cell adhesion mediator activity [GO:0098632]; axon guidance [GO:0007411]; centrosome localization [GO:0051642]; cerebral cortex radially oriented cell migration [GO:0021799]; dendrite self-avoidance [GO:0070593]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
20956536_CAMDI is required for radial migration probably through DISC1 and myosin II-mediated centrosome positioning during neuronal development. 27014940_Ccdc141 is expressed in GnRH neurons and olfactory fibers during embryonic development and that knockdown of Ccdc141 reduces GnRH neuronal migration. 27737934_findings showed that CAMDI regulates neuronal migration through the modulation of HDAC6 and indicate that HDAC6 hyperactivation by CAMDI deletion causes psychiatric behaviors, at least in part, through delayed radial migration due to impaired centrosomes 31730661_KIBRA inhibited CAMDI-dependent Rab11 activation, thereby attenuating AMPAR cell surface expression. 34298015_Oscillation of Cdc20-APC/C-mediated CAMDI stability is critical for cortical neuron migration. |
ENSG00000163492 |
CCDC141 |
247.319988 |
2.2061181691 |
1.141510 |
0.28792995 |
15.484693 |
0.00008317608598556270943155666053314689634134992957115173339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005039531192251754318500722717999451560899615287780761718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
319.988956385045 |
64.2875027774856 |
145.04615431119 |
22.0066457119324 |
| ENSMUSG00000044092 |
319772 |
C130050O18Rik |
protein_coding |
F7AVJ9 |
|
Reference proteome |
|
|
|
|
|
|
|
165.736858 |
0.4802409410 |
-1.058170 |
0.20199837 |
27.375161 |
0.00000016756981364368293612770912780901566563329652126412838697433471679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000016970900275402782219599838800516700132448022486642003059387207031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
106.736916151444 |
14.9936040904528 |
222.257011113886 |
22.4915407605127 |
| ENSMUSG00000044177 |
278507 |
Wfikkn2 |
protein_coding |
Q7TQN3 |
FUNCTION: Protease-inhibitor that contains multiple distinct protease inhibitor domains. Probably has serine protease- and metalloprotease-inhibitor activity (By similarity). Inhibits the biological activity of mature myostatin, but not activin. {ECO:0000250}. |
3D-structure;Direct protein sequencing;Disulfide bond;Glycoprotein;Immunoglobulin domain;Metalloenzyme inhibitor;Metalloprotease inhibitor;Protease inhibitor;Reference proteome;Repeat;Secreted;Serine protease inhibitor;Signal |
|
|
mmu:278507; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; metalloendopeptidase inhibitor activity [GO:0008191]; receptor antagonist activity [GO:0048019]; serine-type endopeptidase inhibitor activity [GO:0004867]; transforming growth factor beta binding [GO:0050431]; muscle cell development [GO:0055001]; negative regulation of DNA binding [GO:0043392]; negative regulation of peptidase activity [GO:0010466]; negative regulation of protein binding [GO:0032091]; negative regulation of transforming growth factor beta receptor signaling pathway [GO:0030512]; roof of mouth development [GO:0060021]; skeletal system development [GO:0001501]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
12595574_Report of the identification of growth and differentiation factor-associated serum protein-1 (GASP-1) associated with endogenous myostatin in normal serum 20164830_Taken together, this study provides evidence that GASP1 regulates CB1 receptor downregulation in vivo, and that postendocytic receptor trafficking has a key role in the development of tolerance to WIN55,212-2. 30814254_results indicate that both WFIKKN and follistatin utilize their FSDs to block the type II receptor but do so via different binding interactions. |
ENSG00000173714 |
WFIKKN2 |
45.935205 |
4.2119046207 |
2.074473 |
0.39426247 |
27.496259 |
0.00000015739869928665459609777588223522570842760615050792694091796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000016058251758434846145418613966304732798562326934188604354858398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
73.2600395387229 |
20.2707767901829 |
17.3935656515325 |
3.886186114763 |
| ENSMUSG00000044201 |
12532 |
Cdc25c |
protein_coding |
P48967 |
FUNCTION: Functions as a dosage-dependent inducer in mitotic control. Tyrosine protein phosphatase required for progression of the cell cycle. Directly dephosphorylates CDK1 and activates its kinase activity. When phosphorylated, highly effective in activating G2 cells into prophase (By similarity). May be involved in regulating the proliferation of T-lymphocytes following cytokine stimulation. {ECO:0000250}. |
Acetylation;Cell cycle;Cell division;Hydrolase;Mitosis;Nucleus;Phosphoprotein;Protein phosphatase;Reference proteome |
|
This gene encodes a dual specificity phosphatase that dephosphorylates cyclin B-bound Cdk1 to trigger entry into mitosis. [provided by RefSeq, Dec 2014]. |
mmu:12532; |
cytoplasm [GO:0005737]; mitochondrial intermembrane space [GO:0005758]; nuclear speck [GO:0016607]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; protein kinase binding [GO:0019901]; protein tyrosine phosphatase activity [GO:0004725]; WW domain binding [GO:0050699]; cell division [GO:0051301]; G2/M transition of mitotic cell cycle [GO:0000086]; positive regulation of G2/M transition of mitotic cell cycle [GO:0010971]; positive regulation of G2/MI transition of meiotic cell cycle [GO:0110032]; protein dephosphorylation [GO:0006470]; spermatogenesis [GO:0007283] |
11972334_A single cell cycle genes homology region controls transcription of the cdc25C gene and is able to cooperate with E2F or Sp1/3 sites 15767688_Cdc25A, or possibly other phosphatases, is able to functionally compensate for the loss of Cdc25B and Cdc25C in mice 17160696_The present study was aimed to investigate the possibility that selenium (Se)-induced oxidative stress mediated alterations in Cdc25c and p21 may cause modulations in the CDC2/Cyclin B1 complex responsible for G2/M phase checkpoint in spermatogenesis. 17349584_In Lzts1(-/-) mouse embryo fibroblasts (MEFs), Cdc25C degradation was increased during M phase, resulting in decreased Cdk1 activity. 18056802_PP2A:B56delta as a key upstream regulator of Cdk1 activity upon exit from mitosis 21283624_CDC25A and CDC25B but not CDC25C compensate for each other to maintain the proliferative capacity of intestinal epithelial stem and progenitor cells 22071115_an asymmetrical distribution pattern for Cdc25c transcripts in 2-cell embryos. 25012723_The role of Cdc25c and Cdc25b in activating G2/M cell cycle checkpoint in zygote. 27117522_oxidative stress-induced DNA damage of mouse zygotes triggers the cell cycle checkpoint, which results in G2/M cell cycle arrest, and that phospho-Cdc25B (Ser323), phospho-Cdc25C (Ser216), and phospho-Cdc2 (Tyr15) participate in activating the G2/M checkpoint. |
ENSG00000158402 |
CDC25C |
258.767216 |
2.6053114196 |
1.381456 |
0.29456049 |
20.308334 |
0.00000659128472685593078567471356232765344884683145210146903991699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000505785637075413280379365676964198428322561085224151611328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
344.598496242259 |
70.8478295294567 |
132.267679652087 |
20.5053465100646 |
| ENSMUSG00000044217 |
11830 |
Aqp5 |
protein_coding |
Q9WTY4 |
FUNCTION: Forms a water-specific channel (By similarity). Plays an important role in fluid secretion in salivary glands (PubMed:10400615, PubMed:16571723, PubMed:18027168). Required for TRPV4 activation by hypotonicity. Together with TRPV4, controls regulatory volume decrease in salivary epithelial cells (PubMed:16571723). Seems to play a redundant role in water transport in the eye, lung and in sweat glands (PubMed:10619865, PubMed:12042359, PubMed:18027168). {ECO:0000250|UniProtKB:P55064, ECO:0000269|PubMed:10400615, ECO:0000269|PubMed:10619865, ECO:0000269|PubMed:12042359, ECO:0000269|PubMed:16571723, ECO:0000269|PubMed:18027168}. |
Cell membrane;Cytoplasmic vesicle;Glycoprotein;Membrane;Reference proteome;Repeat;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:11830; |
apical plasma membrane [GO:0016324]; basal plasma membrane [GO:0009925]; cytoplasmic vesicle membrane [GO:0030659]; endoplasmic reticulum [GO:0005783]; membrane [GO:0016020]; microvillus [GO:0005902]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; water channel activity [GO:0015250]; camera-type eye morphogenesis [GO:0048593]; carbon dioxide transport [GO:0015670]; cellular hypotonic response [GO:0071476]; odontogenesis [GO:0042476]; pancreatic juice secretion [GO:0030157]; protein homotetramerization [GO:0051289]; saliva secretion [GO:0046541]; water transport [GO:0006833] |
11773623_We conclude that the presence of AQP5 in plasma membranes of sweat glands is essential for secretion, providing potential insight into mechanisms underlying mammalian thermoregulation, tactile sensitivity, and the pathophysiology of hyperhidrosis 11989981_These data indicate that skeletal muscle cells express AQP5 protein and its expression is regulated by differentiation and hypertonic stress. 12042359_these results indicate the expression of AQP5 in sweat gland secretory epithelium, but provide direct evidence against its physiological involvement in sweat fluid secretion in mice 12943377_Dynamically expressed in developing mouse inner ear. Adult Aqp5 knockout mice show normal hearing and normal inner ear structural development. Redundant or alternative mechanisms likely regulate water homeostasis in developing and mature inner ear. 15536076_cAMP and beta-adrenergic agonists produce distinct short and long term effects on AQP5 distribution and abundance that may contribute to regulation of lung water homeostasis. 15557451_Aquaporin-1, -3, and -5 dependent water-transporting properties of cornea and conjunctiva. 17097063_atRA increases AQP5 expression through transactivation of Sp1, leading to an increase in plasma membrane water permeability. 17360692_There is a gender-influenced molecular mechanism involving AQP5 that allows transcellular and paracellular routes of water transport to act in conjunction. 17521680_AQP5 may be involved in topiramate-induced hypohidrosis. 17665453_Submandibular glands from 24-week-old NOD mice displayed inflammatory infiltrates, increased AQP5 protein expression, and impaired AQP5 distribution. 18155156_This is the first evidence demonstrating an association between AQP5 and a signaling pathway, namely the Ras signal transduction pathway, which may be the basis of the oncogenic properties seen in AQP-overexpressing cells. 18305162_AQP5 expression was needed for shear-induced barrier enhancement 19215235_Decreased expression of AQP-1 and AQP-5 in aged mice is sufficient to cause a significant decrease in alveolar water transport. 19237298_aquaporins are common targets of gene expression in both allergen and IL-13 induced mouse models of asthma 20522648_These results suggest that LPS-induced potential down-regulation of expression of AQP5 mRNA in the parotid gland is mediated via a complex(es) of these two classes of transcription factors, NF-kappaB and p-c-Jun/c-Fos. 20550619_The results implicate involvement of aquaporin 5 in the development of airway inflammation and mucous hyperproduction during chronic asthma. 21193966_These data provide more evidence that AQP5 plays important roles in the metastasis potential of lung cancer 21203896_AQP5 expression first occurs in a scattered pattern in the late canalicular stage and becomes more prominent and organized in the terminal tubuli/pro-acinar cells towards birth 21455588_results also showed that AQP5 expression increases MUC5AC and MUC5B mucin production 21633078_AQP5 is phosphorylated at its Thr259 by PKA through cAMP, but not Ca(2+), signaling pathways. This phosphorylation does not contribute to AQP5 trafficking in the salivary gland cells. 21745799_Aquaporin-5 was present at the apical face of the olfactory epithelium, completing a water transport pathway to the surface of the epithelium. 21818558_AQP5 plays a role during embryonic salivary gland development. 22226856_results indicate that AQP1 and AQP5 are closely related to pulmonary edema but not to eosinophil infiltration or mucus secretion during asthma. 22420587_AQP5-mediated high plasma membrane water permeability enhances the apoptosis rate of differentiating bone marrow-derived mesenchymal stem cells. 22550388_Phosphokinase A induces AQP5 internalization in the corneal epithelial cells. 23313152_AQP5 is a significant component of lens fiber cell membranes, representing the second most abundant water channel in these cells. 23326416_upregulated Aqp5 may contribute to polyuria, possibly by impairing Aqp2 membrane localization, in Dot1l(AC) mice and in patients with diabetic nephropathy. 23469202_Hypoxia decreases aquaporin 5 (AQP5) expression through both hypoxia inducible factor-1alpha and proteasome-mediated pathways. 23473857_The regulated AQP5 translocation may contribute to sweat secretion by increasing the water permeability of apical plasma membranes of sweat glands. 23925155_Administration of cevimeline maintains the proper localization of AQP-5 in the acinar cells of the salivary glands of mice with Sjogren's syndrome. 24148248_this is the first report providing evidence that AQP5 facilitates maintenance of lens transparency and homeostasis by regulating osmotic swelling caused by glucose transporters and cotransporters under hyperglycemic stressful conditions. 24274330_AQP5 plays an important role in high altitude pulmonary edema formation induced by high altitude simulation. 24429825_The co-regulation of pendrin and AQP5 membrane expression under chronic K(+)-deficiency indicates that these two molecules could cooperate as an osmosensor to rapidly detect and respond to alterations in luminal fluid osmolality. 24662389_GTP-dependent AQP5 expression could act as osmosensor 24747567_propose a new function of AQP5 as an inflammatory signal potentiator, which may be mediated by increased activation of ERK and NF-kappaB 24879973_Lung AQP1 and AQP5 expression were significantly decreased in mice with acute lung injury together with increased inflammatory reaction and apoptosis of alveolar epithelial and vascular endothelial cells 24941004_The activation of P2X7 receptor was connected with an increase of aquaporin-5, whereas the inhibition of the receptor with oxidized ATP resulted in down regulation of aquaporin-5. 25767807_AQP5 promoter methylation is not a universal mechanism for AQP5 regulation. 26272113_AQP5 immunoreactivity was found in the isthmic muscle and lamina propria beneath the epithelia. In cycling females, oviduct aqp5 mRNA levels were the highest at oestrus and the lowest at dioestrus. AQP5 immunoreactivity in non-ciliated cells was notable in the infundibulum, where AQP5 was relatively high at oestrus but low at dioestrus & pro-oestrus, indicating synchrony between aqp5 gene activation and the ovarian cycle. 27871297_The AQP5 genotype may influence survival following lipopolysaccharides by altering neutrophil cell migration. 28088675_Adjusting AQP5 protein levels could be considered a therapeutic strategy for the treatment of acute pulmonary edema induced by H2S and other hazardous gases 28484984_A daily rhythm was detected in the expression profiles of Aqp5 in submandibular glands in vivo. 28615712_Cooperative interactions among transcription factors and histone modifications regulate Aqp5 expression during alveolar epithelial cell transdifferentiation, suggesting that HDAC inhibitors may enhance repair by promoting acquisition of AT1 cell phenotype. 28877240_RUNX1 is essential for the development of the granular convoluted tubules in the submandibular glands; RUNX1 could also be involved in the membrane trafficking of the AQP5 protein of the acinar cells in the SMG in order to allow for the proper secretion of saliva 29118028_AQP5 forms functional water channels in the rodent lens, and suggest that dynamic membrane insertion of AQP5 may regulate water fluxes in the lens by modulating water permeability in the outer cortex. 29361536_Enriched salivary epithelial clusters that were grown in laminin-enriched basement membrane extract or laminin-111 together with exogenous FGF2, but not with EGF, underwent morphogenesis to form organoids that displayed robust expression of AQP5 in terminal buds 29951954_Results indicate that autophagy plays a crucial role in aquaporin 5 (AQP5) degradation in diabetic submandibular gland (SMG). 31953689_Distinct expression patterns of aquaporin 3 and 5 in ductal and alveolar epithelial cells in mouse mammary glands before and after parturition. 32025032_stem cells within the AQP5(+) compartment are a source of WNT-driven, invasive gastric cancer in vivo 32931478_TRPV4 channels are essential for alveolar epithelial barrier function as protection from lung edema. 33187367_Integrated Single-Cell RNA-Sequencing Analysis of Aquaporin 5-Expressing Mouse Lung Epithelial Cells Identifies GPRC5A as a Novel Validated Type I Cell Surface Marker. 33840298_FoxO1 as a Regulator of Aquaporin 5 Expression in the Salivary Gland. 34390680_Aquaporin 5 Facilitates Corneal Epithelial Wound Healing and Nerve Regeneration by Reactivating Akt Signaling Pathway. 34455107_Up-regulation of Aquaporin 5 Defines Spasmolytic Polypeptide-Expressing Metaplasia and Progression to Incomplete Intestinal Metaplasia. 34948308_Ezrin Regulates Ca(2+) Ionophore-Induced Plasma Membrane Translocation of Aquaporin-5. 35002213_Lacrimal gland homeostasis is maintained by the AQP5 pathway by attenuating endoplasmic reticulum stress inflammation in the lacrimal gland of AQP5 knockout mice. 36626177_Aquaporin5 Deficiency Aggravates ROS/NLRP3 Inflammasome-Mediated Pyroptosis in the Lacrimal Glands. 36688817_Loss of aquaporin 5 contributes to the corneal epithelial pathogenesis via Wnt/beta-catenin pathway. |
ENSG00000161798 |
AQP5 |
16.782706 |
0.1287991392 |
-2.956805 |
0.81008392 |
12.766572 |
0.00035286889564964635072896737000291977892629802227020263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0018369442988842832852058872816769508062861859798431396484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.38807779177923 |
2.00398828670669 |
26.3051276086824 |
10.0478036740838 |
| ENSMUSG00000044303 |
12578 |
Cdkn2a |
protein_coding |
Q64364 |
FUNCTION: Capable of inducing cell cycle arrest in G1 and G2 phases (PubMed:8521522, PubMed:9393858). Acts as a tumor suppressor (PubMed:8521522, PubMed:9393858, PubMed:15601844, PubMed:17936562). Binds to MDM2 and blocks its nucleocytoplasmic shuttling by sequestering it in the nucleolus (PubMed:9529248, PubMed:10359817). This inhibits the oncogenic action of MDM2 by blocking MDM2-induced degradation of p53 and enhancing p53-dependent transactivation and apoptosis (PubMed:10359817). Also induces G2 arrest and apoptosis in a p53-independent manner by preventing the activation of cyclin B1/CDC2 complexes (PubMed:15361884). Binds to BCL6 and down-regulates BCL6-induced transcriptional repression (PubMed:15567177). Binds to E2F1 and MYC and blocks their transcriptional activator activity but has no effect on MYC transcriptional repression (By similarity). Binds to TOP1/TOPOI and stimulates its activity (By similarity). This complex binds to rRNA gene promoters and may play a role in rRNA transcription and/or maturation (By similarity). Interacts with NPM1/B23 and promotes its polyubiquitination and degradation, thus inhibiting rRNA processing (By similarity). Plays a role in inhibiting ribosome biogenesis, perhaps by binding to the nucleolar localization sequence of transcription termination factor TTF1, and thereby preventing nucleolar localization of TTF1 (PubMed:20513429). Interacts with COMMD1 and promotes its 'Lys63'-linked polyubiquitination (By similarity). Interacts with UBE2I/UBC9 and enhances sumoylation of a number of its binding partners including MDM2 and E2F1 (By similarity). Binds to HUWE1 and represses its ubiquitin ligase activity (By similarity). May play a role in controlling cell proliferation and apoptosis during mammary gland development (By similarity). {ECO:0000250|UniProtKB:Q8N726, ECO:0000269|PubMed:10359817, ECO:0000269|PubMed:15361884, ECO:0000269|PubMed:15567177, ECO:0000269|PubMed:15601844, ECO:0000269|PubMed:17936562, ECO:0000269|PubMed:20513429, ECO:0000269|PubMed:8521522, ECO:0000269|PubMed:9393858, ECO:0000269|PubMed:9529248}.; FUNCTION: [Isoform smARF]: May be involved in regulation of autophagy and caspase-independent cell death; the short-lived mitochondrial isoform is stabilized by C1QBP. {ECO:0000269|PubMed:16713577}. |
3D-structure;Alternative splicing;Apoptosis;Cell cycle;DNA-binding;Mitochondrion;Nucleus;Reference proteome;rRNA processing;Transcription;Transcription regulation;Tumor suppressor;Ubl conjugation;Ubl conjugation pathway |
|
|
|
cytoplasm [GO:0005737]; granular component [GO:0001652]; mitochondrion [GO:0005739]; nuclear body [GO:0016604]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; protein-containing complex [GO:0032991]; senescence-associated heterochromatin focus [GO:0035985]; cyclin-dependent protein serine/threonine kinase inhibitor activity [GO:0004861]; disordered domain specific binding [GO:0097718]; DNA binding [GO:0003677]; MDM2/MDM4 family protein binding [GO:0097371]; NF-kappaB binding [GO:0051059]; p53 binding [GO:0002039]; protein kinase binding [GO:0019901]; protein N-terminus binding [GO:0047485]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; SUMO transferase activity [GO:0019789]; ubiquitin ligase inhibitor activity [GO:1990948]; ubiquitin-protein transferase inhibitor activity [GO:0055105]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; aging [GO:0007568]; amyloid fibril formation [GO:1990000]; apoptotic mitochondrial changes [GO:0008637]; apoptotic process involved in mammary gland involution [GO:0060057]; autophagy of mitochondrion [GO:0000422]; cell cycle [GO:0007049]; cellular response to hydrogen peroxide [GO:0070301]; cellular senescence [GO:0090398]; epidermis development [GO:0008544]; glucose homeostasis [GO:0042593]; mammary gland epithelial cell proliferation [GO:0033598]; mitochondrial depolarization [GO:0051882]; negative regulation of B cell proliferation [GO:0030889]; negative regulation of cell cycle [GO:0045786]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of cell-matrix adhesion [GO:0001953]; negative regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0045736]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of hepatocyte proliferation [GO:2000346]; negative regulation of immature T cell proliferation in thymus [GO:0033088]; negative regulation of mammary gland epithelial cell proliferation [GO:0033600]; negative regulation of NF-kappaB transcription factor activity [GO:0032088]; negative regulation of phosphorylation [GO:0042326]; negative regulation of protein binding [GO:0032091]; negative regulation of protein kinase activity [GO:0006469]; negative regulation of protein neddylation [GO:2000435]; negative regulation of proteolysis involved in protein catabolic process [GO:1903051]; negative regulation of ubiquitin protein ligase activity [GO:1904667]; negative regulation of ubiquitin-dependent protein catabolic process [GO:2000059]; negative regulation of ubiquitin-protein transferase activity [GO:0051444]; positive regulation of apoptotic process [GO:0043065]; positive regulation of apoptotic process involved in mammary gland involution [GO:0060058]; positive regulation of DNA damage response, signal transduction by p53 class mediator [GO:0043517]; positive regulation of DNA-binding transcription factor activity [GO:0051091]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of protein localization to nucleus [GO:1900182]; positive regulation of protein sumoylation [GO:0033235]; positive regulation of signal transduction by p53 class mediator [GO:1901798]; positive regulation of transcription by RNA polymerase II [GO:0045944]; protein destabilization [GO:0031648]; protein K63-linked ubiquitination [GO:0070534]; protein polyubiquitination [GO:0000209]; protein stabilization [GO:0050821]; regulation of apoptotic DNA fragmentation [GO:1902510]; regulation of cell cycle [GO:0051726]; regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0000079]; regulation of DNA-binding transcription factor activity [GO:0051090]; regulation of G1/S transition of mitotic cell cycle [GO:2000045]; regulation of G2/M transition of mitotic cell cycle [GO:0010389]; regulation of gene expression [GO:0010468]; regulation of nucleocytoplasmic transport [GO:0046822]; regulation of protein export from nucleus [GO:0046825]; regulation of protein stability [GO:0031647]; regulation of protein targeting to mitochondrion [GO:1903214]; replicative senescence [GO:0090399]; response to organic cyclic compound [GO:0014070]; response to organonitrogen compound [GO:0010243]; response to xenobiotic stimulus [GO:0009410]; rRNA processing [GO:0006364]; rRNA transcription [GO:0009303]; somatic stem cell division [GO:0048103]; somatic stem cell population maintenance [GO:0035019] |
11544531_Functions as a tumor suppressor in mice. 11718560_protein interaction mapping with Hdm2 11891301_The Arf tumor suppressor gene promotes hyaloid vascular regression during mouse eye development 11984000_Reactive oxygen-induced carcinogenesis causes hypermethylation of p16(Ink4a) and activation of MAP kinase. 12015983_Mutations of this gene promote tumorigenesis and drug resistance, in part, by disabling apoptosis. 12150825_Relief of transcriptional repression by E2F rendered mouse embryonic fibroblasts resistant to senescence induced by either p19(ARF), p53, or RAS(V12). 12154087_cooperates with CARF in activating p53 12185599_mice with a heterozygous deficiency for the A/J Cdkn2a allele were significantly more susceptible to lung tumor progression than mice with a heterozygous deficiency for a BALB/cJ Cdkn2a allele, when compared to their respective wild type mice 12446760_results on the differential regulation E2F1, DP1, and the E2F1/DP1 complex suggest the possibility that ARF regulates the function of these cell cycle factors by altering the dynamics of their heterodimerization during progression from G(1) to S phase 12538357_Perturbations of the Ink4a/Arf gene locus in aflatoxin B1-induced mouse lung tumors. 12547516_This protein inhibits the ability of cyclin D-dependent kinases to phosphorylate Rb and increases p53 stability by binding Mdm2. 12578823_p19(Arf) and p53 negatively regulate cell migration by suppression of PI 3-kinase and Rac1 activities 12589812_regions of p19ARF which are responsible for p53-independent apoptosis 12620229_p19(Arf) inhibits production of ribosomal RNA 12624188_absence of p16(INK4a) protein leads to higher glucocorticoid receptor transactivation activity during fibrosarcoma progression 12642863_is consistently inactivated in murine myeloid cells transformed in vivo by deregulated c-Myc 12700664_4 sequence variants were found between BALB/c & DBA/2 in the p16 promoter region.Most of the difference in activity was localized to a single nucleotide deletion in BALB/c. This deletion created a consensus binding site for RREB. 12740365_ARF seems to down-regulate Bcl-2 in a p53-dependent manner while up-regulating Bax/Bim via a p53-independent pathway 12777178_Quiescence of primordial follicles might be maintained by high expression of p16. The absence or decreased expression of p16 strongly coincides with the first measurable follicle growth. 12810676_The contribution of Ink4a/Arf heterozygosity to mammary tumor progression is tissue-specific in vivo. 14560011_Results provide genetic evidence that Cited2 controls the expression of INK4a/ARF and fibroblast proliferation, at least in part via the polycomb-group genes Bmi1 and Mel18. 14652011_Remarkable attenuation of p19(ARF)-induced apoptosis by disruption of the Bax gene thus leads to the conclusion that Bax plays a major role in p53-independent apoptosis induced by p19(ARF). 14665695_Inactivation of Arf led to the outgrowth of tumor cells, providing direct evidence that the Arf promoter monitors latent oncogenic signals in vivo 14724566_both p19(ARF)-/- and p16(INK4a)-/- animals were significantly more tumor prone than wild-type animals, but each less so than p53-/- or Ink4a/Arf-/- animals, and with differing tumor spectra 14724578_Cdk6-cyclin D3 complexes are not subject to inhibition by p16(INK4a) 14726696_the Arf gene responds to latent oncogenic signals in vivo to eliminate incipient cancer cells 14991053_reduced expression of p16 and p19 by methylation-induced silencing or inactivation of p38 MAPK correlated with tumor appearance 15016851_both products of the Ink4a/Arf locus influence Ab-MLV transformation and reveal that in addition to its well-recognized effects on the cell cycle, p16(Ink4a) can suppress transformation by inducing apoptosis 15044451_Cav-1 cooperates with the tumor suppressor INK4a genetic locus to prevent cell proliferation and oncogene-induced tumorigenesis 15057871_UV-induced mutations of INK4a-ARF occur in a small percentage in late stages skin tumors 15105443_p19ARF determines the balance between normal cell proliferation rate and apoptosis during mammary gland development. 15175242_E2f3 loss is sufficient to derepress Arf, triggering activation of p53 and expression of p21(Cip1); oncogenic activation of Arf is associated with recruitment of the endogenous activating E2Fs, E2F1, and E2F3a, to the Arf promoter 15199070_ARF attenuates c-Myc independently of the ARF-p53 axis 15273738_INK4a/Arf is required for suppression of EGFR/DeltaEGFR(2-7)-dependent ERK activation in mouse astrocytes and glioma. 15314658_p19Arf suppresses growth, progression, and metastasis of Hras-driven carcinomas through p53-dependent and -independent pathways 15361884_ARF can inhibit c-Myc by a unique and direct mechanism that is independent of p53 15370242_review of p15INK4b, p14ARF and p16INK4a function as cell cycle inhibitors where they are involved in the inhibition of G1 phase progression, and promoter methylation in hematologic malignancies and solid tumors 15377996_M33 deficient cells have a senescent phenotype, associated to an abnormal accumulation of the cyclin-dependent kinase inhibitor p16INK4a protein. 15467748_the stoichiometry of Mdm2 and ARF controls apoptosis and lymphoma development 15520862_Ink4a and Arf have roles in mammalian aging 15572761_INK4a/ARF mutations predispose to malignant melanoma, a UV-induced tumor. 15601844_a novel Jun-Dmp1 pathway directly links oncogenic Ras-Raf signaling and p19(Arf), independent of the classical cyclin D1/Cdk4-Rb-E2F pathway 15657897_although both BRAF and INK4A lesions promote cell growth and tumor formation, mutant BRAF may also induce dedifferentiation in melanoma cells 15662416_Pokemon can specifically repress the transcription of the tumour suppressor gene ARF through direct binding 15688012_AP-1 heterodimers may contribute to the regulation of ARF expression upon oncogenic signaling 15692066_mechanisms independent of the Ink4a-Arf gene locus play a dominant role in HSC loss during conditions of proliferative stress 15781613_Ink4a and Arf have independent and differential tumor suppressor functions in vivo in the glial cell compartment. 15897463_Arf's p53-independent effects on gene expression and tumor suppression might depend on Arf-induced sumoylation. 15964994_Bmi-1 prevents the premature senescence of neural stem cells by repressing Ink4a and Arf 15964995_Arf is a general target of Bmi1, however particularly in neural stem cells, derepression of Ink4a contributes to Bmi1(-/-) phenotypes 16004968_Efficient induction of apoptosis by oncogene activation depends on coordinated action of ARF and ATM/ATR pathways in the regulation of p53. 16037818_p19Arf blocks platelet-derived growth factor receptor beta-dependent signals and prevent the accumulation of perivascular cells selectively in a vascular bed destined to regress. 16199529_Arf is haploinsufficient for the induction of acute myeloid leukemia in the presence of the inv(16) 16199867_In the absence of nucleoplasmin, Arf protein is excluded from nucleoli and is markedly less stable. 16205116_p19(Arf) responds to specific developmental cues to disrupt platelet-derived growth factor receptor beta signaling in the developing eye extend existing paradigms for Arf tumor suppressor gene biology 16205640_E2F1 suppresses ras-driven skin carcinogenesis through a nonapoptotic mechanism involving ARF and p53 16206162_inhibition of Schwann cell proliferation is the overriding crucial function of p16 and p21 in peripheral nerves 16239970_In a carotid arterial-injury model, expression of p16 is required for activation of peroxisome proliferative activated receptor alpha (PPAR alpha) to inhibit vascular smooth muscle cell proliferation resulting in intimal hyperplasia 16407836_Loss of either p16(ink4a) or p19(arf) cooperates with Tal1 to induce leukemia in mice. Tal1 stimulates cell cycle entry independent of the ink4a/arf locus, but its ability to induce apoptosis is Ink4a/Arf-dependent. 16491120_p16Ink4a function is associated with T-cell apoptosis, and subsequently contributes to the control of T-cell population size in lymphoid organs 16491126_Mdm2 can function independently from ARF 16585505_Loss of either Ink4a/Arf (p16(Ink4a)/p19(Arf)) or p53 enables malignant progression of pancreatic adenocarcinoma. 16697958_These genetic and translational observations establish a cooperative role of Pten and Ink4a/Arf in the development of HS and provide mechanistic insights into the pathogenesis of human HS. 16760664_Both of these closely spaced ATG start codons can be utilized as a translational start codon to produce a nucleolar-localized ARF protein which can induce a p53-dependent inhibition of cell division 16809772_reduction in pRB renders cells resistant to p16(ink4a)-mediated G(1) arrest 16904610_Inactivation of p19 ARF enhances Myc induced apoptosis. 16912158_Altered expression of two proteins involves in immortalization and escape from senescence: low levels of p19(ARF) and high levels of TBX2 in mouse embryo fibroblasts. 16920973_nitric oxide-dependent apoptosis requires, in part, the activation of p19(ARF) 16957735_the cyclin-dependent kinase inhibitor p16INK4a, the level of which was previously noted to increase in other cell types with age, accumulates and modulates specific age-associated hematopoietic stem cells functions 16957737_These genetic data support the view that an age-induced increase of p16INK4a expression limits the regenerative capacity of beta-cells with ageing. 16957738_Declining subventricular zone progenitor function and olfactory bulb neurogenesis during ageing are thus caused partly by increasing p16INK4a expression. 16957780_role for the tumor suppressor ARF in viral infection surveillance 16971940_results indicate that the cancer-protective activity of p53 is abolished in the absence of ARF 17099725_Ink4c loss modulated the effects of Myc-induced transformation by decreasing the frequency of Arf loss, an ordinarily common event in Emu-Myc-induced lymphomas. 17219434_These results define downstream targets of p38 MAPK during preimplantation development and indicate that the p38 MAPK pathway regulates Trp53, Ppm1d, and Cdkn2a expression. 17251441_In the mouse model, loss of Arf in only a subset of cells causes a (persistent hyperplastic primary vitreous) PHPV-like disease. 17265068_This is the first direct evidence that the level of Tbx3 expression positively controls the proliferation of mammary epithelial cells via pathways alternative to Mdm2-p53. 17344414_Study shows that the ability of the oncogene BMI1 to repress the INK4A-ARF locus requires its direct association and is dependent on the continued presence of the EZH2-containing Polycomb-Repressive Complex 2 complex. 17360455_Ink4a/Arf tumor suppressor does not modulate the degenerative conditions or tumor spectrum of the telomerase-deficient mouse 17438529_PDGFB-induced activation of the mitogen-activated protein kinase pathway via extracellular signal-regulated kinase was involved in the initiation of low-grade oligodendrogliomas 17507663_expression of P16(INK4A) in developing TAL1xLMO1 thymocytes blocks leukemogenesis in the majority of the mice 17534149_Review. Induction of transfected murine ARF led to a rapid increase in ARF expression in human cells, resulting in p53 stabilization. The p19ARF/oncogene stress pathway induces the acetylation of p53 at lysine 120. 17546045_The Dmp1-Arf pathway is repressed by p65 in response to genotoxic stress. 17546053_It is demonstrated that NPMc+ blocks the p19(Arf) (Arf) induction elicited by E1A. 17576930_ARF and p53, although linked in a common pathway, suppress tumorigenesis through distinct, lineage-dependent mechanisms and suggest that ARF helps restrict melanoma progression by executing the oncogene-induced senescence program in benign nevi 17585020_The effects of aging and dietary folate on select features of DNA methylation in the colon that are relevant to carcinogenesis are reported. 17630509_Results show that p14ARF regulates E2F-1 ubiquitination and degradation via a p53-dependent mechanism. 17637672_observations extend the protective role of Arf/p53 to ageing, revealing a previously unknown anti-ageing mechanism and providing a rationale for the co-evolution of cancer resistance and longevity 17695724_ARF tumor suppressor has a role in mouse models of acute leukemias [review] 17699762_Concomitant loss of the Ink4a/Arf tumor suppressor locus accelerates tumor formation and metastasis, suggesting potential roles for the p16 and p19 tumor suppressors in this process. 17713536_data indicate that p15Ink4b can fulfil a critical backup function for p16Ink4a and provide an explanation for the frequent loss of the complete CDKN2b-CDKN2a locus in human tumours 17761812_A role for ARF in human Philadelphia chromosome-positive lymphoblastic leukemia progression. 17891181_Required for the elimination of cells with aberrant CD43 expression. 17936558_Demonstrate in an orthotopic transplantation model for glioma, that Bmi1 is also required for tumor development in an Ink4a/Arf-independent manner. 17952114_different strategies can be selected to collaborate with v-Ha-ras in tumorigenesis 18006687_Catnb induces immortalization of melanocytes by suppressing p16INK4a expression and cooperates with N-Ras in melanoma development. 18037485_lethally irradiated recipient mice reconstituted with RUNX1-ETO-expressing cells showed that neither p15(Ink4b) or p16(Ink4a) loss significantly accelerated disease progression over the time period of one year post-transplantation 18039844_inactivation of Ink4a rescued the defective proliferation of maturing cells but did not affect the hyperproliferative activity of progenitors and resulted in a shortening of the onset of lymphomas induced by Ink4a inactivation 18056257_expression leading to endothelial cell proliferation is suppressed by Rem2 18059331_p19/Arf and p53 suppress sentinel lymph node lymphangiogenesis and carcinoma metastasis. 18060046_Isolated p19 Arf deficiency mirrored p53 deficiency in that both promoted rapid, EMT-associated mammary tumor escape, whereas isolated p16 Ink4a deficiency failed to accelerate relapse 18070929_basal ARF proteins act as a monitor of steady-state ribosome biogenesis and growth independent of their ability to prevent unwarranted hyperproliferation. 18199542_A novel role for the Arf tumor suppressor protein in regulating phenotypes associated with tumor progression and metastasis in hepatocellular carcinoma cells. 18275833_findings show survival of activated pre-T cells induced by pre-TCR signaling required Bmi-1-mediated repression of Cdkn2A & that p19Arf expression resulted in thymocyte cell death & inhibited transition from DN to DP stage upstream of p53 pathway 18328429_Mesothelioma developed at high incidence in Nf2;Ink4a/Arf and Nf2;p53 conditional knockout mice which closely mimicked human malignant mesothelioma. 18356246_Tbx3 plays a crucial role in controlling hepatoblast proliferation and cell-fate determination by suppressing p19(ARF) expression and thereby promoting liver organogenesis. 18372918_p53 and p16 are critical in preventing mammary gland tumorigenesis in the human Aurora A overexpression mouse model 18381074_smARF has functions other than mitochondria-mediated autophagy, and induces p53 expression and cell death via a novel mechanism. 18418377_results demonstrate that p16Ink4a/p19Arf and Trp53 have a central role in limiting the expansion potential of multipotent progenitors 18442038_Inactivation of p19(ARF) in mice increases the number of cycling cells in the crypts of the colonic epithelium. 18458079_Cdc6 determines utilization of p21(WAF1/CIP1)-dependent damage checkpoint in S phase cells 18483220_The inherent instability of mutant p53 is alleviated by Mdm2 or p16INK4a loss. 18516091_Data show that skeletal muscle and fat, two tissues that develop early ageing-associated phenotypes in response to BubR1 insufficiency, have high levels of p16(Ink4a) and p19(Arf). 18538737_Mitochondrial p32/C1QBP is a critical mediator of p14ARF-induced apoptosis. 18567945_p16 was differentially expressed during distinct developmental stages and its transcriptional regulation was DNA methylation-independent 18579612_Changes in p19Arf localization accompany apoptotic crisis during pre-B cell transformation by Abelson murine leukemia. 18591425_These results reveal that loss of miRNA biogenesis activates a DNA damage checkpoint, up-regulates p19(Arf)-p53 signaling, and induces senescence in primary cells. 18636078_These results indicate that C/EBPbeta represses p53 to promote cell survival downstream of DNA damage and suggest that inhibition of C/EBPbeta may be a target for cancer cotherapy to increase the efficacy of alkylating chemotherapeutic agents. 18755985_activity of p19(ARF) profoundly influences the nature of tumor-initiating cells during BCR/ABL-mediated leukemogenesis. 18769141_Inactivation of p16(Ink4a) in BubR1 mutant mice delays both cellular senescence and aging specifically in these tissues. 18809412_These studies provide the first insights into the molecular basis through which Arf nullifies the p53-inhibiting activity of Hdm2, indirectly activating the tumor-suppressor function of p53 in mammalian cells. 18818403_PPARalpha trans-repression involved the recruitment of the retinoblastoma (RB) family proteins p107 and p130 to the TERT promoter resulting in impaired E2F-1 binding, an effect that was dependent on p16. 18948745_The p53-independent tumor suppressive functions of p19(Arf) may be mediated by its ability to antagonize Senp3, thereby inducing cell cycle arrest by abnormally elevating the cellular levels of SUMOylated proteins. 18957199_Hmga2 promotes fetal and young-adult stem cell self-renewal by decreasing p16(Ink4a)/p19(Arf) expression. 19056891_Data provide a molecular explanation for the preferential effects of aging on lymphopoiesis, suggest that inhibiting p16(Ink4a) and Arf expression can rejuvenate B lymphopoiesis, and link aging and cancer resistance. 19124561_Inhibition of the p38 mitogen-activated protein kinase pathway blocked the induction of p16(INK4a) and cellular senescence. 19196247_The increase in p16 INK4a in the vasculature with aging may modify VSMC's response to post-injury stress and therefore accelerate the development of age-related cardiovascular diseases. 19202064_combination of histone modifications driven by Ndy1 interferes with the binding of RNA Polymerase II, resulting in the transcriptional silencing of the Ink4a/Arf locus and contributing to the Ndy1 immortalization phenotype 19219070_Sox5 can suppress platelet-derived growth factor B-induced glioma development in Ink4a-deficient mice through induction of acute cellular senescence. 19239418_We conclude that the Ink4/Arf locus has a global anti-aging effect, probably by favouring quiescence and preventing unnecessary proliferation. 19240179_Findings suggest that p53 has a necessary but not sufficient role in repressing p16(INK4a) expression. 19308294_Loss of p53 was accompanied by a significant increase in the growth rate, gain of karyotypic instability, loss of p16 expression, and lack of senescence response. 19321450_Reactive oxygen species-induced up-regulation of p16(Ink4a) occurs correlatively with ERK1/2-dependent down-regulation and subsequent dissociation from chromatin of Bmi-1. 19380490_Results identify the dual capability of ELF4/MEF to promote transformation via effects on cellular senescence and tumor cell growth by regulating the p53/Mdm2/p19ARF and the p16/Rb pathways. 19390085_in aged islets, derepression of the Ink4a/Arf locus is associated with decreased Bmi-1 binding 19390090_Enhancer of zeste homolog 2 represses Ink4a/Arf in islet beta cells 19398954_Increased senescence as mediated by the p16(INK4A) tumor suppressor has only a minor function as an intrinsic regulator of steady-state HSC ageing in vivo. 19422607_gene amplification of Met, a receptor tyrosine kinase implicated in melanoma progression, was identified in Cdkn2a-deficient tumors. RNA interference targeting Met in tumor cells resulted in a delay in tumor growth in vivo compared with the control cells. 19451217_inhibition of Jmjd3 expression in mouse embryonic fibroblasts results in suppression of p16Ink4a and p19Arf expression and in their immortalization 19462008_In young proliferating embryonic fibroblasts, the Polycomb Repressive Complex 2 (PRC2) member EZH2 together with PRC1 members BMI1 and M33 are strongly expressed and localized at the INK4/ARF regulatory domain (RD) identified as a DNA replication origin. 19465598_p19(Arf) expression can be coupled to extracellular cues in normal cells; we suggest a new mechanism for Arf control in tumor cells. 19644500_provide evidence that ARF reduces the interaction of the E2F4-p130 repressor complex with the promoter of xeroderma pigmentosum, complementation group C (XPC) to ensure high-level expression of XPC. 19644500_provide evidence that alternative reading frame(ARF) reduces the interaction of the E2F4-p130 repressor complex with the promoter of xeroderma pigmentosum, complementation group C (XPC) to ensure high-level expression of XPC. 19649205_we provided evidence for a crucial role for direct inhibition of p53 by MDM2 and suppression of the p19(ARF)/p53 axis in neuroblastoma tumorigenesis 19690330_Data reveal differential consequences of p19(Arf) inactivation in prostate cancer and MEFs upon Pten loss that are independent of the p53 pathway. 19723773_Findings show that miR-290 might act as a physiological effector of NCZ induced as well as culture senescence via p16 regulation expanding the role of this miRNA from embryonic stem to differentiated cells. 19727727_Findings suggest methylation of p16 INK4A as early events in the pathogenesis and development of plasma cell disorders. 19777566_ta indicate that Zik1 and Gja9 demonstrated cancer-specific aberrant DNA methylation, whereas, Cdkn2a/p16, Igfbp3, Mgmt, Id4, and Cxcr4 were methylated in both the AOM tumors and normal colon mucosa. 19855433_Data show that activated BRAF generates gliomas with characteristics similar to activated KRas in the context of Akt but not Ink4a/Arf loss. 20118236_analysis of the p53/ARF pathway disruption in spontaneous escape from senescence in mice 20381282_Deficiency of the p19ARF protein, a tumor suppresor encoded by the CDKN2A gene, accelerates atheroma development in the atherosclerosis-prone aortic arch. 20382980_H3K27 trimethylation is an early epigenetic event of p16INK4a silencing for regaining tumorigenesis in fusion reprogrammed hepatoma cells 20479239_Lung tumors, which can arise in the presence of functional p19(Arf), rarely display p19(Arf) induction. In contrast, sarcomas always show robust activation, suggesting that loss of the p19(Arf)-p53 pathway is a requisite event for sarcomagenesis. 20501856_Results define a mechanism by which the ARF tumor suppressor targets the translational repression of specific oncogenes during neoplastic transformation. 20513429_The data delineate the pathway by which ARF regulates ribosomal RNA synthesis and provide a compelling explanation for the role of NPM. 20522552_GRIM-19/CDKN2A synergistically suppressed cell cycle progression via inhibiting E2F1-driven gene expression. 20569441_Nutlin-3 treatment and p19Arf gene transfer activated tumor p53 and reduced the cell viability and proliferation. 20648475_Repression of targets of Bmi1 other than the Ink4a/Arf locus plays a crucial role in the oncogenic transformation of hepatic stem/progenitor cells. 20682446_These results show that differentiation of mammalian cells is reversed by inactivation of Arf and Rb and support the hypothesis that Arf evolved at the expense of regeneration. 20697345_We found potent synergy between melanocyte-specific activation of K-Ras and loss of p16(INK4a) and/or p53 in melanomagenesis 20708155_Deletion of Ink4a/Arf in K-ras(G12D) expressing mice prevents senescence and leads to invasive, metastasizing carcinomas with morphological and molecular alterations comparable to human KRAS mutated serrated tumors 20718941_In animals carrying the Arf or p53 defects, no naevi were detected, with all tumours ostensibly skipping the benign precursor stage in progression. 20805995_Arf loss of function can promote tumorigenesis via facilitating angiogenesis, at least in part, through p53-independent mechanisms. 20808772_Zfp277 in the transcriptional regulation of the Ink4a/Arf locus and the recruitment of PRC1 to the Ink4a/Arf locus 20838381_found increased susceptibility of melanocytes to oxidative stress in the context of p16 depletion, which may explain why the compromise of p16 predisposes to melanoma over other cancers 20861074_results confirm that p16(INK4a) controls cell proliferation and matrix production and mitigates fibrosis following injury and suggest that the mechanism involves a role in limiting inflammation and cell proliferation. 20941790_The authors show that tumour progression is associated with down-regulation of p16(Ink4a) through enhanced CpG methylation of exon 1 and knockout of Cdkn2a confirms this gene is a barrier to tumour progression. 21054888_regulation of p16, p21, p27 and c-myc is increased by oridonin, which induces apoptosis and senescence in colorectal cancer cells 21057530_Activated MEK cooperates with Ink4a/Arf loss to induce gliomas in vivo. 21059868_Data show that Pcl2 differentially regulates Polycomb-mediated repression of Hox and Cdkn2a genes. 21062974_Data show a cooperative effect of increased benign skin tumor formation caused by p21Waf1/Cip1 loss, with increased malignant conversion of benign skin tumors caused by p16(INK4a) loss. 21187925_p38 MAPK and JNK antagonistically control senescence and cytoplasmic p16INK4A expression in doxorubicin-treated endothelial progenitor cells 21209895_ARF tumor suppressor regulates bone remodeling and osteosarcoma development in mice 21216978_These results suggest that a subpopulation of pulmonary artery smooth muscle cells are resistant to the signals that activate the p19(Arf) promoter. 21233453_Venous thrombosis is augmented by overexpression of the cellular senescence protein p16(INK4a). 21245485_Expression of p16(INK4a) can promote aging and prevent cancer in related lymphoid progeny of a common stem cell. 21393858_in glioblastomas with Ink4a/Arf deficiency, overexpressed PDGFRalpha promotes tumorigenesis through the PI3K/AKT/mTOR-mediated pathway regulated by SHP-2 activity 21427293_Notch1 mutation and Arf inactivation appear to independently cooperate in no requisite order with Lmo2 overexpression in inducing T-ALL, and all 3 events remained insufficient to guarantee immediate tumor development. 21452288_p19(ARF) /p14(ARF) controls oncogenic functions of signal transducer and activator of transcription 3 in hepatocellular carcinoma. 21526190_Asbestos-treated Ink4a(+/-) and Arf(+/-) mice showed increased incidence and shorter latency of malignant mesothelioma relative to wild-type littermates. 21622646_combined loss of p19Arf and Rag1 results in B-cell precursor leukemia in mice 21636855_p16(INK4a) is a modulator of macrophage activation and polarization via the JAK2-STAT1 pathway with possible roles in inflammatory diseases. 21681782_Nf1 mutant mice with p19ARF gene loss develop accelerated hematopoietic disease resembling acute leukemia with a variable phenotype. 21757616_Down-regulation of p19(ARF) induces accelerated onset of MLL-ENL leukemia, suggesting that p19(ARF) is a major target of AML1 in MLL-ENL leukemia. 21789189_DeltaNp63alpha has oncogenic activity and its overexpression in human squamous cell carcinomas contributes to the malignant phenotype, and implicate its ability to regulate p16(ink4a)/p19(arf) in the process. 21799732_p16( INK4a) is also a modulator of transcription and apoptosis through controlling the expression of two major transcription regulators, AUF1 and E2F1 21811412_Data show that inactivation of Arf triggers germ cell-autonomous, p53-dependent apoptosis of primary spermatocytes in late meiotic prophase, resulting in reduced sperm production. 21868699_Cdkn2a is an atherosclerosis modifier locus that regulates monocyte/macrophage proliferation. 21937682_ARF limited the nucleolar localization of the RNA helicase DDX5. ARF inhibited the interaction between DDX5 and nucleophosmin preventing association of DDX5 with the rDNA promoter and nuclear pre-ribosomes. 21957175_Knockout of the INK4a/ARF locus results in increased tubular cell proliferation, decreased apoptosis, and decreased capillary rarefaction. 21987724_Used a combination of somatic conditional overexpression and ligand-mediated chronic activation of EGFR in cooperation with Ink4a/Arf loss in the central nervous system of adult mice. 22048312_In tissues--such as adipose tissue, skeletal muscle and eye--in which p16(Ink4a) contributes to the acquisition of age-related pathologies, life-long removal of p16(Ink4a)-expressing cells delayed onset of these phenotypes. 22064482_Hyperactivation of mTORC1 through Tsc1 loss resulted in a significant increase in ARF expression, activation of the p53 pathway, and a dramatic cell cycle arrest, which were completely reversed upon Arf deletion. 22095712_Alternative reading frame (ARF) plays an important role in regulating inflammatory processes and as a regulator of inflammatory cell signaling. 22113502_p16 knockout mice developed the full spectrum of pancreatic intraepithelial neoplasia (mPanIN) lesions, pancreatic ductal adenocarcinoma (PDA), and metastases 22208613_The absence of mutations in genes such as KRAS, TP53, and CDKN2A, which are considered as key genes in the development of human pancreatic cancer suggests that SMAD4 might play a central and decisive role in mouse pancreatic cancer. 22226369_Increased gene dosage of the Ink4/Arf locus does not attenuate atherosclerosis development in hypercholesterolaemic mice. 22403661_Bone marrow p16(INK4a)-deficiency does not affect plasma lipids, obesity, glucose tolerance or atherosclerosis in mice 22430984_Inactivation of Arf is a causative event in vascular neoplasia and suggest that the ARF pathway may be a novel molecular target for therapeutic intervention in haemangiosarcoma patients. 22555975_Repression of Ink4a in aged (ETPs) Early T-cell progenitors results in their partial rejuvenation and this can be accomplished by in vivo fibroblast growth factor 7 administration. 22561347_data indicate that TRADD shuttles dynamically from the cytoplasm into the nucleus to modulate the interaction between p19(Arf) and its E3 ubiquitin ligase ULF, thereby promoting p19(Arf) protein stability and tumour suppression 22586120_Data show that that BRAF(V600E) expression in neural progenitors (NPs) is insufficient for tumorigenesis and increases NP cellular differentiation as well as apoptosis, but astrocytomas are readily generated from NPs with additional Ink4a-Arf deletion. 22629125_In conclusion, the current study demonstrates the potential of pseudotype VLPs with inserted target antigen as a new type of immunogens to generate antibodies of high diagnostic value. 22711607_High p16(INK4a)is associated with drug sensitivity in glioblastoma. 22729085_Identify c-Abl as a regulator of bone morphogenetic protein signalling pathways and uncover a role for c-Abl in p16(INK4a) expression and osteoprogenitor expansion. 22733434_Long-term arsenic exposure may induce down-regulation of p16(INK4a) by targeting recruitment of G9a and H3K9 dimethylation without altering DNA methylation before tumorigenesis in the liver. 22754337_Data suggest that, in the context of Wrn deficiency-related telomere dysfunction, loss of p16Ink4a function could prevent cells from senescence. 22813853_The mutations in factors regulating p16(Ink4a) methylation might be the driver mutation. These findings shed light on the strategy of anti-aging by regulating p16(Ink4a) expression. 22890319_HOPS acts as a functional bridge in the interaction between NPM and p19(Arf) 22907756_findings demonstrate a novel capacity for p19(Arf) to control Pdgfrbeta expression by p53-dependent and -independent mechanisms involving RNA transcription and protein synthesis, respectively, to promote the vascular remodeling needed for normal vision 22998186_[review] In addition to mediating apoptosis, ARF also plays a role in the induction of autophagy. 23029416_Mdm2 haploinsufficiency significantly delayed tumorigenesis in mice deficient in Arf and p53. 23045274_Meningioma progression in mice triggered by Nf2 and Cdkn2ab inactivation. 23066149_p16(INK4a) exe |
|
|
51.179791 |
0.0100772756 |
-6.632751 |
0.92609391 |
96.598765 |
0.00000000000000000000008490123651434735145322339613754836853378174526331691995637430524918043417414992291014641523361206054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000039791250294970424573994485443647518514068787868195048863621809953627916911500506103038787841796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.999619747300314 |
0.68666036292111 |
99.1954359280779 |
22.2751599364316 |
| ENSMUSG00000044337 |
12778 |
Ackr3 |
protein_coding |
P56485 |
FUNCTION: Atypical chemokine receptor that controls chemokine levels and localization via high-affinity chemokine binding that is uncoupled from classic ligand-driven signal transduction cascades, resulting instead in chemokine sequestration, degradation, or transcytosis. Also known as interceptor (internalizing receptor) or chemokine-scavenging receptor or chemokine decoy receptor. Acts as a receptor for chemokines CXCL11 and CXCL12/SDF1. Chemokine binding does not activate G-protein-mediated signal transduction but instead induces beta-arrestin recruitment, leading to ligand internalization and activation of MAPK signaling pathway. Required for regulation of CXCR4 protein levels in migrating interneurons, thereby adapting their chemokine responsiveness. In glioma cells, transduces signals via MEK/ERK pathway, mediating resistance to apoptosis. Promotes cell growth and survival. Not involved in cell migration, adhesion or proliferation of normal hematopoietic progenitors but activated by CXCL11 in malignant hemapoietic cells, leading to phosphorylation of ERK1/2 (MAPK3/MAPK1) and enhanced cell adhesion and migration. Plays a regulatory role in CXCR4-mediated activation of cell surface integrins by CXCL12. Required for heart valve development. Regulates axon guidance in the oculomotor system through the regulation of CXCL12 levels (PubMed:31211835). {ECO:0000269|PubMed:17804806, ECO:0000269|PubMed:18442043, ECO:0000269|PubMed:20161793, ECO:0000269|PubMed:21220100, ECO:0000269|PubMed:21246655, ECO:0000269|PubMed:31211835}. |
Cell adhesion;Cell membrane;Developmental protein;Disulfide bond;Endosome;G-protein coupled receptor;Glycoprotein;Membrane;Phosphoprotein;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:12778; |
cell surface [GO:0009986]; clathrin-coated pit [GO:0005905]; cytoplasm [GO:0005737]; early endosome [GO:0005769]; endosome [GO:0005768]; external side of plasma membrane [GO:0009897]; intracellular membrane-bounded organelle [GO:0043231]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; recycling endosome [GO:0055037]; C-C chemokine binding [GO:0019957]; C-C chemokine receptor activity [GO:0016493]; C-X-C chemokine binding [GO:0019958]; C-X-C chemokine receptor activity [GO:0016494]; coreceptor activity [GO:0015026]; scavenger receptor activity [GO:0005044]; angiogenesis [GO:0001525]; calcium-mediated signaling [GO:0019722]; cell adhesion [GO:0007155]; cell chemotaxis [GO:0060326]; chemokine-mediated signaling pathway [GO:0070098]; immune response [GO:0006955]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of intrinsic apoptotic signaling pathway in response to DNA damage [GO:1902230]; oculomotor nerve development [GO:0021557]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of mesenchymal stem cell migration [GO:1905322]; receptor internalization [GO:0031623]; vasculogenesis [GO:0001570] |
17804806_Disfrupted cardiac development but normal hematopoiesis in mice deficient in Cxcr7. 18442043_Seems to play an unimportant role in bone; however, an important role is indicated for CXCR7 in the cardiovascular system during embryonic and early postnatal development. 19302186_Data show that the SDF-1alphaCXCR7 signalling pathway regulates genes involved in cell cycle control, amino acid metabolism and ligase activity, which comprise targets that are distinct from those of CXCR4. 19794961_Data show that beta-arrestin 2 increased uptake of CXCL12 in cells expressing CXCR7, emphasizing the functional relevance of the interaction between CXCR7 and beta-arrestin 2. 19821487_Data show thatblockade of the SDF-1 receptors CXCR4 and CXCR7 significantly reduced tumor capillary density and tumor growth. 20151462_Stromal cell-derived factor-1 regulates myogenic differentiation via both CXCR4 and CXCR7 chemokine receptors in skeletal muscle. 20380740_CXCR7 regulates the invasion, angiogenesis and tumor growth of hepatocellular carcinoma cells 20681075_CXCR7 is involved in neuronal cell migration and may act independently of CXCR4 as a mediator of CXCL12 signaling. 20854168_These data indicate that SDF-1 is a key neuroprotective cytokine secreted by CD133-derived multipotent stromal cells that protects mouse neural progenitor cell through CXCR7. 20869949_exposure of mesenchymal stem cells (MSC) to hypoxia enhances the expression of CXCR4 and CXCR7 by upregulating HIF-1alpha through the PI3K/Akt pathways, and both of the two receptors are required for cell migration, adhesion and survival of MSCs in vitro. 20889540_whereas CXCR7 protein is expressed by primitive RBCs during murine embryonic development, in adult mammals CXCR7 protein is not expressed by normal peripheral blood cells. 21220099_Data show that CXCR4 and CXCR7 have distinct roles and signal transduction in regulating interneuron movement and laminar positioning. 21220100_Cxcr7 is necessary to regulate Cxcr4 protein levels, thereby adapting chemokine responsiveness in migrating cells. 21246655_Data suggest that CXCR7 is involved in semilunar valve development, possibly by regulating BMP signaling, and may contribute to aortic and pulmonary valve stenosis. 21300915_CXCR7 influences leukocyte entry into the CNS parenchyma by controlling abluminal CXCL12 abundance during autoimmunity. 21778049_The CXCL11/CXCR7 pathway is involved hepatocellular carcinoma progression. 22145790_targeting CXCR7 modifies both disease severity and recovery during experimental autoimmune encephalomyelitis, suggesting a role for this molecule in both phases of disease. 22232208_data implicate an important role for the CXCR4/CXCR7/SDF-1 pathway in the pathogenesis of hemolytic uremic syndrome 22736769_Regulatory pathways targeting CXCR7 C-terminal serine/threonine sites may control the CXCL12 scavenger activity of CXCR7. 22766125_Increase in CXCR7 expression is associated with muscular dystrophy. 22880115_CXCR7 fine-tunes CXCL12/CXCR4 mediated signalling between podocytes and glomerular capillaries. 23169289_Study suggests that the activation of CXCR7 on tumour blood vessels by its ligands may facilitate the progression of colorectal carcinoma (CRC) within lung but not within liver. 23452257_differential and hierarchically ordered roles for CXCR4/CXCL12- vs. CXCR7/CXCL12-dependent effects during experimental autoimmune neuritis 23599431_CXCR7 is induced during monocyte-to-macrophage differentiation, which is required for SDF-1 and I-TAC signaling to JNK and p38 pathways, leading to enhanced macrophage phagocytosis 24116850_endothelial CXCR7 regulates circulating CXCL12 levels and that CXCR7 inhibitors might be used to block CXCL12-mediated cell migration for therapeutic purposes. 24315101_Genetic and pharmacologic experiments demonstrated that intermediate opioid peptides secreted from SCH activate CXCR7, a beta-arrestin-biased G-protein-coupled receptor, to augment circadian oscillation of glucocorticoid levels in a paracrine manner and CXCR7 ligand is sufficient to induce anxiolytic-like behavior. 24374972_CXCR7 regulates blood cholesterol by promoting its uptake in adipose tissue. 24718993_Interfering with the CXCL12-scavenging activity of CXCR7 causes loss of CXCR4 function as a consequence of excessive CXCL12-mediated CXCR4 activation and degradation. 24733828_CXCR7 plays a role in oligodendrocyte progenitor cell-maturation during remyelination. 24886617_These findings identify CXCR7-mediated STAT3 activation and modulation of the tumor microenvironment as novel regulation of breast cancer growth and metastasis. 25051350_Findings indicate that upregulation of CXCR7 expression is associated with tumor invasion in colorectal cancer. 25063073_Studied the expression and function of chemokine receptor CXCR2 and CXCR7 in the rat with acute leukemia. 25085881_Sata demonstrate that CXCR7 has a role in the positioning of hem and pallium-subpallium boundary-derived Cajal-Retzius (CR)cells, CXCL12 regulating CR cell subpial localization through the combined action of CXCR4 and CXCR7. 25203207_CXCR7 has a role in modulating adrenomedullin-mediated cardiac and lymphatic vascular development 25268050_The inhibitory effect of corticosterone on mesenchymal stromal cells (MSC) migration might be mediated via decreasing the expression of CXCR4 and CXCR7 in MSCs. 25460567_the spermatogonial population may not only be simply controlled by interaction of Cxcl12 with Cxcr4 but may also involve Cxcr7 as an important player 25833331_Both CXCR7 and Rac1 are required for extracellular signal-regulated kinases (ERK) 1/2 activation and subsequent NPC migration, indicating that CXCR7 could serve as a functional receptor in CXCL12-mediated NPC migration independent of CXCR4. 25997895_expression of CXCR7 in normal and pathological nervous system suggests CXCR4-independent functions of SDF-1/CXCL12 mediated through its interaction with CXCR7 26119946_Endothelial CXCR7 limits breast cancer metastasis at multiple steps in the metastatic cascade, advancing understanding of CXCL12 pathways in tumor environments and informing ongoing drug development targeting CXCR7 in cancer. 26139098_Blockade of CXCR7 suppressed MIF-mediated ERK- and zeta-chain-associated protein kinase (ZAP)-70 activation 26265085_confirm a pivotal role of the SDF-1/CXCR4/CXCR7 axis for chronic allograft vasculopathy development 26303308_CXCR7 overexpression in MSCs reversed the inhibitory effect of high concentrations of SDF-1alpha. 26578388_our present study provided evidence that SDF-1 mediated CSCs migration through CXCR4 and CXCR7 via MEK/ERK and PI3K/Akt pathway 26607112_Collectively, our data demonstrate that CXCR7 suppression modulates microglial chemotaxis to ameliorate EAE. 26779814_CXCR7 acts to prevent epithelial damage and ameliorate fibrosis after a single lung injury, repeated injury leads to suppression of CXCR7 expression and recruitment of VEGFR1 expressing macrophages, Wnt and Notch signaling, and enhanced fibrosis. 27693695_CXCR7 expression in adipose tissue macrophages is upregulated in obesity. 27736296_the Sdf-1 receptors - Cxcr4 and Cxcr7, as well as signaling pathways induced by these molecules in primary myoblasts, as well as various stem cells, were studied. 28137917_Elevated expression of CXCR7 enhances endothelial progenitor cell resistance to diabetes mellitus-induced oxidative damage. 28188248_the data identified the pivotal role of the receptor CXCR7 in pulmonary inflammation with a predominant effect on the pulmonary epithelium and polymorphonuclear neutrophils 28542132_the pivotal role of CXCR4- and CXCR7-inhibition in acute pulmonary inflammation, which depended on A2B-receptor signaling, is reported. 28687617_High CXCR7 expression is associated with Prostate Cancer. 29117251_endothelial CXCR7+ cells regulate CXCL12 gradient direction by controlling concentrations near but not far from the vasculature. 29587613_Study shows that overexpression of CXCR7 in different cellular populations of endometriosis microenvironment may play a role in the pathogenesis and represent a novel target for treatment. 29980568_CXCR7 serves as Dkk3 receptor, which mediates Dkk3-induced vascular progenitor migration in vitro and in tissue-engineered vessels. 30726732_Absence and excess of CXCL12 cause similar interneuron motility defects. Phosphorylation regulates ACKR3-mediated scavenging independently of beta-arrestins. 30958662_CXCL14 is expressed by islet delta-cells where it may have paracrine effects to inhibit insulin secretion in a CXCR4/CXCR7-independent manner through reductions in beta-cell ATP levels. 31100368_Findings suggested that a single Cxcl12 or Cxcr7 allele might not be sufficient to maintain the hippocampal niche functionality throughout life, and that heterozygosity might represent a susceptibility factor for memory dysfunction progression. 31211835_ACKR3, through its regulation of CXCL12 levels, is an important regulator of axon guidance in the oculomotor system; complete loss causes oculomotor synkinesis in mice. 31505255_CXCR7: A key neuroprotective molecule against alarmin HMGB1 mediated CNS pathophysiology and subsequent memory impairment. 31672961_CXCR7 regulates epileptic seizures by controlling the synaptic activity of hippocampal granule cells. 31722287_analysis showed that interaction of SDF-1 or ITAC with CXC receptors is necessary for the regulation of crucial developmental regulators expression and that CXCR7 is involved in the control of ESC pluripotency and differentiation into mesodermal lineages. 31792315_CXCR7 promotes migration and invasion in head and neck squamous cell carcinoma by upregulating TGF-beta1/Smad2/3 signaling. 32170348_CXCR7 suppression modulates macrophage phenotype and function to ameliorate post-myocardial infarction injury. 32361166_Activation of CXCR7 alleviates cardiac insufficiency after myocardial infarction by promoting angiogenesis and reducing apoptosis. 32452398_Deletion of atypical chemokine receptor 3 (ACKR3) increases immune cells at the fetal-maternal interface. 32755592_Marginal Zone Formation Requires ACKR3 Expression on B Cells. 32978390_The atypical chemokine receptor 3 interacts with Connexin 43 inhibiting astrocytic gap junctional intercellular communication. 33564089_CXCR7 ameliorates myocardial infarction as a beta-arrestin-biased receptor. 33857173_Lymphatic endothelial-cell expressed ACKR3 is dispensable for postnatal lymphangiogenesis and lymphatic drainage function in mice. 34072818_Smooth Muscle Specific Ablation of CXCL12 in Mice Downregulates CXCR7 Associated with Defective Coronary Arteries and Cardiac Hypertrophy. 35365782_Atypical chemokine receptor 3 induces colorectal tumorigenesis in mice by promoting beta-arrestin-NOLC1-fibrillarin-dependent rRNA biogenesis. 35674847_Endothelial ACKR3 drives atherosclerosis by promoting immune cell adhesion to vascular endothelium. 36088505_CXCR4 and CXCR7 signaling promotes tumor progression and obesity-associated epithelial-mesenchymal transition in prostate cancer cells. 36179603_Histone deacetylase 2 regulates STAT1-dependent upregulation of atypical chemokine receptor 3 to induce M2 macrophage migration and immune escape in hepatocellular carcinoma. 36800350_An expression and function analysis of the CXCR4/SDF-1 signalling axis during pituitary gland development. |
ENSG00000144476 |
ACKR3 |
831.818721 |
0.2317258534 |
-2.109509 |
0.10412133 |
400.485385 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000004317899195378407419116123118930634303940641879551881014962283202966900903270861148622911701942035510363835273017190691571926809324787734687791331372428623001076201416305651870 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000219234135146679695910187343246381981983516181983352410933273428996737254460982880614035220335556549303744406963403847629109422085027171049527871619608208917422376418135355025 |
Yes |
No |
309.651640240566 |
22.3673993379234 |
1336.2843884834 |
66.8799356431693 |
| ENSMUSG00000044433 |
69697 |
Camsap3 |
protein_coding |
Q80VC9 |
FUNCTION: Key microtubule-organizing protein that specifically binds the minus-end of non-centrosomal microtubules and regulates their dynamics and organization (PubMed:23169647, PubMed:24706919, PubMed:26715742). Specifically recognizes growing microtubule minus-ends and autonomously decorates and stabilizes microtubule lattice formed by microtubule minus-end polymerization (PubMed:24706919). Acts on free microtubule minus-ends that are not capped by microtubule-nucleating proteins or other factors and protects microtubule minus-ends from depolymerization (PubMed:24706919). In addition, it also reduces the velocity of microtubule polymerization (PubMed:24706919). Required for the biogenesis and the maintenance of zonula adherens by anchoring the minus-end of microtubules to zonula adherens and by recruiting the kinesin KIFC3 to those junctional sites (By similarity). Required for orienting the apical-to-basal polarity of microtubules in epithelial cells: acts by tethering non-centrosomal microtubules to the apical cortex, leading to their longitudinal orientation (PubMed:26715742). Plays a key role in early embryos, which lack centrosomes: accumulates at the microtubule bridges that connect pairs of cells and enables the formation of a non-centrosomal microtubule-organizing center that directs intracellular transport in the early embryo (PubMed:28860385). Couples non-centrosomal microtubules with actin: interaction with MACF1 at the minus ends of non-centrosomal microtubules, tethers the microtubules to actin filaments, regulating focal adhesion size and cell migration (By similarity). Plays a key role in the generation of non-centrosomal microtubules by accumulating in the pericentrosomal region and cooperating with KATNA1 to release non-centrosomal microtubules from the centrosome (By similarity). Through the microtubule cytoskeleton, also regulates the organization of cellular organelles including the Golgi and the early endosomes (By similarity). Through the microtubule cytoskeleton, also regulates the organization of cellular organelles including the Golgi and the early endosomes (By similarity). Through interaction with AKAP9, involved in translocation of Golgi vesicles in epithelial cells, where microtubules are mainly non-centrosomal (By similarity). Plays an important role in motile cilia function by facilitatating proper orientation of basal bodies and formation of central microtubule pairs in motile cilia (PubMed:32482850). {ECO:0000250|UniProtKB:Q9P1Y5, ECO:0000269|PubMed:23169647, ECO:0000269|PubMed:24706919, ECO:0000269|PubMed:26715742, ECO:0000269|PubMed:28860385, ECO:0000269|PubMed:32482850}. |
3D-structure;Alternative splicing;Cell junction;Cell projection;Cilium biogenesis/degradation;Coiled coil;Cytoplasm;Cytoskeleton;Direct protein sequencing;Microtubule;Phosphoprotein;Reference proteome |
|
|
mmu:69697; |
axoneme [GO:0005930]; centrosome [GO:0005813]; ciliary basal body [GO:0036064]; cytoplasm [GO:0005737]; microtubule cytoskeleton [GO:0015630]; microtubule minus-end [GO:0036449]; motile cilium [GO:0031514]; nucleoplasm [GO:0005654]; zonula adherens [GO:0005915]; actin filament binding [GO:0051015]; calmodulin binding [GO:0005516]; microtubule minus-end binding [GO:0051011]; spectrin binding [GO:0030507]; cilium movement [GO:0003341]; cytoplasmic microtubule organization [GO:0031122]; embryo development ending in birth or egg hatching [GO:0009792]; epithelial cell-cell adhesion [GO:0090136]; establishment of epithelial cell apical/basal polarity [GO:0045198]; establishment or maintenance of microtubule cytoskeleton polarity [GO:0030951]; in utero embryonic development [GO:0001701]; microtubule anchoring [GO:0034453]; microtubule cytoskeleton organization [GO:0000226]; negative regulation of microtubule depolymerization [GO:0007026]; neuron projection development [GO:0031175]; protein transport along microtubule [GO:0098840]; regulation of cell migration [GO:0030334]; regulation of focal adhesion assembly [GO:0051893]; regulation of Golgi organization [GO:1903358]; regulation of microtubule cytoskeleton organization [GO:0070507]; regulation of microtubule polymerization [GO:0031113]; regulation of organelle organization [GO:0033043]; zonula adherens maintenance [GO:0045218] |
23169647_CAMSAP2 and -3 work together to maintain noncentrosomal microtubules in epithelial cells, suppressing the microtubule-organizing ability of the centrosome. 24706919_These results show that members of the CAMSAP/Patronin family all localize to and protect minus-ends but have evolved distinct effects on microtubule dynamics. 26715742_These findings demonstrate that apically localized CAMSAP3 determines the proper orientation of microtubules, and in turn that of organelles, in mature mammalian epithelial cells. 27349180_data suggest that CDH23-C is a CAMSAP3/Marshalin-binding protein that can modify MT networks indirectly through its interaction with CAMSAP3/Marshalin. 28386021_CAMSAP3 precisely coordinates with dynein and katanin to regulate the microtubule detachment process. 30190432_maintains neuronal polarity through regulation of microtubule stability 30282632_Findings suggest that CAMSAP3 functions to protect lung carcinoma cells against EMT by suppressing Akt activity via microtubule regulation and that CAMSAP3 loss promotes EMT in these cells. 32482850_CAMSAP3 facilitates basal body polarity and the formation of the central pair of microtubules in motile cilia. 33462112_CAMSAP3 is required for mTORC1-dependent ependymal cell growth and lateral ventricle shaping in mouse brains. 33468623_Intercellular and intracellular cilia orientation is coordinated by CELSR1 and CAMSAP3 in oviduct multi-ciliated cells. 33712686_Cyst formation in proximal renal tubules caused by dysfunction of the microtubule minus-end regulator CAMSAP3. 34319756_Tracheal motile cilia in mice require CAMSAP3 for the formation of central microtubule pair and coordinated beating. |
ENSG00000076826 |
CAMSAP3 |
42.908148 |
0.4776157037 |
-1.066078 |
0.37903004 |
7.886148 |
0.00498148559280658324160961925031188002321869134902954101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0185294233332429503224858535759267397224903106689453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
29.7790141003064 |
7.91396392424084 |
62.3493194859422 |
11.8163450096469 |
| ENSMUSG00000044468 |
74645 |
Tent5c |
protein_coding |
Q5SSF7 |
FUNCTION: Catalyzes the transfer of one adenosine molecule from an ATP to an mRNA poly(A) tail bearing a 3'-OH terminal group and enhances mRNA stability and gene expression (PubMed:34048638). Can also elongate RNA oligos ending with uridine molecule, provided that the sequence is adenosine-rich (By similarity). Mainly targets mRNAs encoding endoplasmic reticulum-targeted protein (PubMed:28931820). {ECO:0000250|UniProtKB:Q5VWP2, ECO:0000269|PubMed:28931820, ECO:0000269|PubMed:34048638}. |
3D-structure;Cytoplasm;Cytoskeleton;Nucleotidyltransferase;Nucleus;Reference proteome;RNA-binding;Transferase |
|
|
mmu:74645; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; polynucleotide adenylyltransferase activity [GO:0004652]; RNA adenylyltransferase activity [GO:1990817]; RNA binding [GO:0003723]; in utero embryonic development [GO:0001701]; mRNA stabilization [GO:0048255]; negative regulation of cell differentiation [GO:0045596] |
28931820_FAM46C protein is strongly induced during activation of primary splenocytes and that B lymphocytes isolated from newly generated FAM46C KO mice proliferate faster than those isolated from their WT littermates. 31087039_Non-canonical RNA polyadenylation polymerase FAM46C is essential for fastening sperm head and flagellum in micedagger. 32341344_TENT5C polyadenylates immunoglobulin mRNAs regulating their half-life and consequently steady-state levels and is upregulated in differentiating plasma cells by innate signaling |
ENSG00000183508 |
TENT5C |
138.438794 |
0.1757674651 |
-2.508260 |
0.32573045 |
56.237686 |
0.00000000000006421877409208540872706671490434773151470115165606600271530624013394117355346679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000014776138473876812332731657785242235930711629521283612120896577835083007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.7046576353144 |
9.04084043282517 |
225.893100321176 |
35.9009669532241 |
| ENSMUSG00000044469 |
66443 |
Tnfaip8l1 |
protein_coding |
Q8K288 |
FUNCTION: Acts as a negative regulator of mTOR activity. {ECO:0000269|PubMed:24444419}. |
Cytoplasm;Reference proteome |
|
|
mmu:66443; |
cytoplasm [GO:0005737]; identical protein binding [GO:0042802]; negative regulation of TOR signaling [GO:0032007]; regulation of apoptotic process [GO:0042981] |
21600655_TIPE1(TNFAIP8-like 1)protein was detected in a variety of tissues such as neurons, brain, hepatocytes, germ cells of female and male reproductive organs, muscular tissues and cells of epithelial origin it was not expressed in mature T or B lymphocytes 25043299_TIPE1 induced apoptosis in HCC cells by negatively regulating Rac1 pathway 33713358_TNF-alpha-Induce Protein 8-Like 1 Inhibits Hepatic Steatosis, Inflammation, and Fibrosis by Suppressing Polyubiquitination of Apoptosis Signal-Regulating Kinase 1. |
ENSG00000185361 |
TNFAIP8L1 |
98.119057 |
2.0018193903 |
1.001312 |
0.26810897 |
13.920044 |
0.00019075337657362587367830542017799189125071279704570770263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010670420242267606054931139425434594159014523029327392578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
134.700066768334 |
25.1768591548534 |
67.2888210708008 |
9.66299512768602 |
| ENSMUSG00000044471 |
232685 |
Lncpint |
lncRNA |
|
|
|
|
|
|
|
24070194_Pint lincRNA connects the p53 pathway with epigenetic silencing by the Polycomb repressive complex 2. 30201298_Inhibition of Lincpint expression affects insulin secretion and apoptosis in mouse pancreatic beta cells 32080970_A lncRNA survey finds increases in neuroprotective LINC-PINT in Parkinson's disease substantia nigra. |
|
|
17.868590 |
4.9767307250 |
2.315198 |
0.78263577 |
7.784892 |
0.00526849518860846629392247209011657105293124914169311523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0194074289511207177805207635401529842056334018707275390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.8386970587726 |
14.7147129344434 |
5.19190176978373 |
2.34303045988719 |
| ENSMUSG00000044562 |
69903 |
Rasip1 |
protein_coding |
Q3U0S6 |
FUNCTION: Required for the proper formation of vascular structures that develop via both vasculogenesis and angiogenesis. Acts as a critical and vascular-specific regulator of GTPase signaling, cell architecture, and adhesion, which is essential for endothelial cell morphogenesis and blood vessel tubulogenesis. Regulates the activity of Rho GTPases in part by recruiting ARHGAP29 and suppressing RhoA signaling and dampening ROCK and MYH9 activities in endothelial cells. May act as effector for Golgi-bound HRAS and other Ras-like proteins. May promote HRAS-mediated transformation. Negative regulator of amino acid starvation-induced autophagy (By similarity). {ECO:0000250, ECO:0000269|PubMed:19272373, ECO:0000269|PubMed:21396893}. |
Angiogenesis;Cytoplasm;Golgi apparatus;Methylation;Phosphoprotein;Reference proteome |
|
|
mmu:69903; |
cell-cell junction [GO:0005911]; Golgi apparatus [GO:0005794]; Golgi stack [GO:0005795]; perinuclear region of cytoplasm [GO:0048471]; protein-containing complex [GO:0032991]; GTPase binding [GO:0051020]; protein homodimerization activity [GO:0042803]; small GTPase binding [GO:0031267]; angiogenesis [GO:0001525]; branching morphogenesis of an epithelial tube [GO:0048754]; negative regulation of autophagy [GO:0010507]; negative regulation of membrane permeability [GO:1905709]; negative regulation of Rho protein signal transduction [GO:0035024]; negative regulation of Rho-dependent protein serine/threonine kinase activity [GO:2000299]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of integrin activation [GO:0033625]; regulation of GTPase activity [GO:0043087]; signal transduction [GO:0007165]; vasculogenesis [GO:0001570] |
15031288_Rain is a novel protein that can serve as an effector of endomembrane-localized Ras 19272373_Rasip1 expression is undetectable in VEGFR2 null embryos, which lack endothelial cells, suggesting that Rasip1 is endothelial specific. siRNA-mediated reduction of Rasip1 severely impairs angiogenesis and motility in endothelial cell cultures 21396893_Mice lacking Rasip1 fail to form patent lumens in all blood vessels, including the early endocardial tube. Rasipl null angioblasts fail to properly localize the polarity determinant Par3 and display defective cell polarity. 23886837_RASIP1 regulates the integrity of newly formed blood vessels as an effector of EPAC1-RAP1 signaling. 24622510_we summarize recent developments in understanding the small G protein RAP1 and its effector RASIP1 as critical mediators of endothelial junction stabilization. 26897025_Rasip1 is necessary in growing embryonic blood vessels. 27486147_Rasip1 controls different pools of GTPases, which in turn regulates blood vessel tubulogenesis. 30042182_Rasip1 regulates Cdc42 activity to regulate cell junctions and cytoskeleton organization, which are both activities required for lymphatic lumen maintenance |
ENSG00000105538 |
RASIP1 |
7.693136 |
0.0684198605 |
-3.869441 |
1.25038998 |
10.062670 |
0.00151303799458055838154935113237797850160859525203704833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0066685748650032019083089274147368996636942028999328613281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.00988562182443 |
0.989042925859091 |
14.7601239494691 |
7.78073172780105 |
| ENSMUSG00000044646 |
207259 |
Zbtb7c |
protein_coding |
Q8VCZ7 |
FUNCTION: May be a tumor suppressor gene. {ECO:0000250}. |
Metal-binding;Reference proteome;Repeat;Tumor suppressor;Zinc;Zinc-finger |
|
|
mmu:207259; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; negative regulation of cell population proliferation [GO:0008285]; positive regulation of fat cell differentiation [GO:0045600]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of RNA polymerase II regulatory region sequence-specific DNA binding [GO:1903025]; regulation of transcription by RNA polymerase II [GO:0006357] |
22331133_Kr-pok (kidney cancer-related POZ domain and Kruppel-like protein) activates transcription of the FASN gene. 27646874_findings show that Zbtb7c is a key molecule that recruits an NCoR/Hdac3 complex to inhibit phosphorylation of c-Jun, and thereby repress Mmp gene expression 30959128_Zbtb7c to be a crucial metabolic regulator of blood glucose homeostasis, during mammalian fasting. 31222731_Zbtb7c degradation or absence may contribute to the pathogenesis of emphysema 34017061_Proto-oncoprotein Zbtb7c and SIRT1 repression: implications in high-fat diet-induced and age-dependent obesity. |
ENSG00000184828 |
ZBTB7C |
774.655678 |
0.4888375527 |
-1.032573 |
0.13757252 |
55.266407 |
0.00000000000010525207145936282341880435011282145925609696895941524985573778394609689712524414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000023715969711080097833682442176340740406549623120469050263636745512485504150390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
503.022480161893 |
48.0399467876155 |
1029.01767131276 |
72.3146944727962 |
| ENSMUSG00000044702 |
233826 |
Palb2 |
protein_coding |
Q3U0P1 |
FUNCTION: Plays a critical role in homologous recombination repair (HRR) through its ability to recruit BRCA2 and RAD51 to DNA breaks. Strongly stimulates the DNA strand-invasion activity of RAD51, stabilizes the nucleoprotein filament against a disruptive BRC3-BRC4 polypeptide and helps RAD51 to overcome the suppressive effect of replication protein A (RPA). Functionally cooperates with RAD51AP1 in promoting of D-loop formation by RAD51. Serves as the molecular scaffold in the formation of the BRCA1-PALB2-BRCA2 complex which is essential for homologous recombination. Via its WD repeats is proposed to scaffold a HR complex containing RAD51C and BRCA2 which is thought to play a role in HR-mediated DNA repair. Essential partner of BRCA2 that promotes the localization and stability of BRCA2. Also enables its recombinational repair and checkpoint functions of BRCA2. May act by promoting stable association of BRCA2 with nuclear structures, allowing BRCA2 to escape the effects of proteasome-mediated degradation. Binds DNA with high affinity for D loop, which comprises single-stranded, double-stranded and branched DNA structures. May play a role in the extension step after strand invasion at replication-dependent DNA double-strand breaks; together with BRCA2 is involved in both POLH localization at collapsed replication forks and DNA polymerization activity (By similarity). {ECO:0000250}. |
3D-structure;Alternative splicing;Coiled coil;DNA damage;DNA recombination;DNA repair;DNA-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Tumor suppressor;WD repeat |
|
|
mmu:233826; |
DNA repair complex [GO:1990391]; nucleoplasm [GO:0005654]; protein-containing complex [GO:0032991]; DNA binding [GO:0003677]; animal organ morphogenesis [GO:0009887]; apoptotic process [GO:0006915]; double-strand break repair via homologous recombination [GO:0000724]; embryonic organ development [GO:0048568]; in utero embryonic development [GO:0001701]; inner cell mass cell proliferation [GO:0001833]; mesoderm development [GO:0007498]; multicellular organism growth [GO:0035264]; negative regulation of apoptotic process [GO:0043066]; post-anal tail morphogenesis [GO:0036342]; somitogenesis [GO:0001756] |
20484223_A key function for PALB2 is to interact with and to build up appropriate communication between BRCA1 and BRCA2. 23657012_Palb2 synergizes with Trp53 to suppress mammary tumor formation in a model of inherited breast cancer. 25016020_results underscore the in vivo importance of the PALB2-BRCA1 complex formation in DSB repair and male meiosis 27490902_we describe a genetic approach to examine the functional significance of the interaction between BRCA2 and PALB2 by generating a knock-in mouse model of Brca2 carrying a single amino acid change (Gly25Arg, Brca2G25R) that disrupts this interaction. In addition, we have combined Brca2G25R homozygosity as well as hemizygosity with Palb2 and Trp53 heterozygosity . 28240985_The findings demonstrate that RNF168 couples PALB2-dependent homologous recombination to H2A ubiquitylation to promote DNA repair and preserve genome integrity. 29739757_The tumor suppression mechanisms of the BRCA1-PALB2 DNA damage response pathway. 31877165_Individual deletion of Palb2, Brca1 or Brca2 genes in pancreas per se using Pdx1-Cre was insufficient to cause tumors, but it reduced pancreata size. Concurrent expression of mutant KrasG12D and p53R270H, with tumor suppressor inactivated strains in Palb2-KPC, Brca1-KPC or Brca2-KPC, accelerated pancreatic ductal adenocarcinoma (PDAC) development. 35006619_Disrupted BRCA1-PALB2 interaction induces tumor immunosuppression and T-lymphocyte infiltration in HCC through cGAS-STING pathway. 35404932_Tumor suppressor PALB2 maintains redox and mitochondrial homeostasis in the brain and cooperates with ATG7/autophagy to suppress neurodegeneration. |
ENSG00000083093 |
PALB2 |
118.113547 |
2.0911035102 |
1.064264 |
0.24927899 |
17.909205 |
0.00002316977713233481361442163415365058654060703702270984649658203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001597655252511199141726705441612921276828274130821228027343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
163.762600128895 |
28.4370612321514 |
78.3139616633345 |
10.3165350486012 |
| ENSMUSG00000045095 |
14924 |
Magi1 |
protein_coding |
Q6RHR9 |
FUNCTION: May play a role as scaffolding protein at cell-cell junctions. May regulate acid-induced ASIC3 currents by modulating its expression at the cell surface. {ECO:0000269|PubMed:15317815}. |
3D-structure;Alternative splicing;ATP-binding;Cell junction;Cell membrane;Cytoplasm;Membrane;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Tight junction |
|
|
mmu:14924; |
adherens junction [GO:0005912]; bicellular tight junction [GO:0005923]; cell junction [GO:0030054]; cell periphery [GO:0071944]; cell projection [GO:0042995]; cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; membrane [GO:0016020]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; alpha-actinin binding [GO:0051393]; ATP binding [GO:0005524]; positive regulation of cell-cell adhesion [GO:0022409]; signal transduction [GO:0007165] |
12773569_MAGI-1 together with JAM4 provides an adhesion machinery at tight junctions, which may regulate the permeability of kidney glomerulus and small intestinal epithelial cells 15383320_We found MAGI-1 to be associated with cell-cell contacts in human umbilical vein endothelial cells (HUVECs) and in mouse endothelium, where it colocalizes with ESAM. 15558322_Cx32 expression is in part related to induction of tight junctions through modulation of Magi1 expression in an immortalized hepatic cell line. 15908431_Dll1 is presented on the surface of AJs formed at the apical termini of processes through interaction with MAGI1 to activate Notch on neighboring cells in the developing central nervous system 17191119_MAGI-1 cleavage appears to be an important step in the disassembly of cell-cell contacts during apoptosis. 20219992_Depletion of MAGI-1 from photorecptors by RNA interference blocks synaptic localization of Sidekick-2 in photoreceptors without affecting localization of postsynaptic density (PSD)-95. 20562105_Data conclude that the up-regulation of sdk-1 in podocytes is an important pathogenic factor in glomerulosclerosis and that the mechanism involves disruption of the actin cytoskeleton possibly via alterations in MAGI-1 function. 23523645_ARIP1 and ARIP2 are co-expressed in some nerve cells and their biological activities are distinct. 27707879_the interaction between nephrin and MAGI-1 regulates Rap1 activation in podocytes to maintain long term slit diaphragm structure 30944250_Study in mouse model of atherosclerosis suggests that MAGI1 simultaneously regulates endothelial cells activation and endoplasmic reticulum stress-induced apoptosis, which are critical molecular events for atherogenesis, via MAGI1-S741 phosphorylation by p90RSK and sentrin/SUMO-specific protease 2 T368 phosphorylation-mediated MAGI1-K931 deSUMOylation. 33712709_MAGIs regulate aPKC to enable balanced distribution of intercellular tension for epithelial sheet homeostasis. |
ENSG00000151276 |
MAGI1 |
300.446940 |
0.4384436481 |
-1.189537 |
0.25361212 |
21.086408 |
0.00000439031145454404117908124918767676092556939693167805671691894531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000347935609134312389192911219026882463367655873298645019531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
169.474552633109 |
29.945698367609 |
386.53668117676 |
50.5527870799315 |
| ENSMUSG00000045102 |
272158 |
Poln |
protein_coding |
A0A0R4J0E3 |
Human_homologues FUNCTION: DNA polymerase with very low fidelity that catalyzes considerable misincorporation by inserting dTTP opposite a G template, and dGTP opposite a T template (PubMed:16787914, PubMed:17118716). Is the least accurate of the DNA polymerase A family (i.e. POLG, POLN and POLQ) (PubMed:17118716). Can perform accurate translesion DNA synthesis (TLS) past a 5S-thymine glycol. Can perform efficient strand displacement past a nick or a gap and gives rise to an amount of product similar to that on non-damaged template. Has no exonuclease activity (PubMed:16787914). Error-prone DNA polymerase that preferentially misincorporates dT regardless of template sequence (PubMed:25775266). May play a role in TLS during interstrand cross-link (ICL) repair (PubMed:19908865). May be involved in TLS when genomic replication is blocked by extremely large major groove DNA lesions. May function in the bypass of some DNA-protein and DNA-DNA cross-links. May have a role in cellular tolerance to DNA cross-linking agents (PubMed:20102227). Involved in the repair of DNA cross-links and double-strand break (DSB) resistance. Participates in FANCD2-mediated repair. Forms a complex with HELQ helicase that participates in homologous recombination (HR) repair and is essential for cellular protection against DNA cross-links (PubMed:19995904). {ECO:0000269|PubMed:16787914, ECO:0000269|PubMed:17118716, ECO:0000269|PubMed:19908865, ECO:0000269|PubMed:19995904, ECO:0000269|PubMed:20102227, ECO:0000269|PubMed:25775266}. |
Reference proteome |
|
|
mmu:272158; |
nucleoplasm [GO:0005654]; cyclin binding [GO:0030332]; DNA binding [GO:0003677]; DNA-directed DNA polymerase activity [GO:0003887]; DNA-templated DNA replication [GO:0006261]; double-strand break repair via homologous recombination [GO:0000724]; interstrand cross-link repair [GO:0036297]; translesion synthesis [GO:0019985] |
12794064_identification as homolog to Mus308 28570559_Inactivation of Poln in mouse embryonic fibroblasts did not alter cellular sensitivity to mitomycin C, cisplatin, or aldehydes. Pol nu-defective mice had no alteration in DNA end-joining during immunoglobulin class-switching. |
ENSG00000130997 |
POLN |
15.754021 |
0.2507941396 |
-1.995424 |
0.76021488 |
6.181314 |
0.01291063483513586220008395599734285497106611728668212890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0418058651804399383777877119428012520074844360351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.16069841225971 |
3.95032277995316 |
28.5520962465714 |
11.0057730897736 |
| ENSMUSG00000045165 |
101602 |
AI467606 |
protein_coding |
Q8C708 |
|
Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:101602; |
membrane [GO:0016020] |
21498670_these findings indicate that AI467606 is a novel transcriptional target of RUNX1/AML1 at the onset of hematopoietic development that is extensively expressed within the hematopoietic system |
ENSG00000185905 |
C16orf54 |
68.038180 |
2.4845471195 |
1.312983 |
0.31017118 |
17.831530 |
0.00002413506159278498182504599234299291765637462958693504333496093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001659707711879461965957111013736380300542805343866348266601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
90.7599907288493 |
19.6635491593741 |
36.5297924994908 |
6.24165771326638 |
| ENSMUSG00000045215 |
211961 |
Asxl3 |
protein_coding |
Q8C4A5 |
FUNCTION: Putative Polycomb group (PcG) protein. PcG proteins act by forming multiprotein complexes, which are required to maintain the transcriptionally repressive state of homeotic genes throughout development. PcG proteins are not required to initiate repression, but to maintain it during later stages of development. They probably act via methylation of histones, rendering chromatin heritably changed in its expressibility (By similarity). {ECO:0000250}. |
Alternative splicing;Metal-binding;Nucleus;Reference proteome;Repressor;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:211961; |
PR-DUB complex [GO:0035517]; chromatin binding [GO:0003682]; DNA binding [GO:0003677]; metal ion binding [GO:0046872]; peroxisome proliferator activated receptor binding [GO:0042975]; animal organ morphogenesis [GO:0009887]; negative regulation of lipid biosynthetic process [GO:0051055]; positive regulation of transcription by RNA polymerase II [GO:0045944] |
34559584_Alteration of long non-coding RNAs and mRNAs expression profiles by compound heterozygous ASXL3 mutations in the mouse brain. |
ENSG00000141431 |
ASXL3 |
41.576333 |
8.9152287267 |
3.156272 |
0.49569231 |
40.734691 |
0.00000000017436408584928797609380473499228513326042566689011437119916081428527832031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000026881718174659459186904043328202040807894945828593336045742034912109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
74.9327487032794 |
26.2555342229188 |
8.40502818274098 |
2.4584044156936 |
| ENSMUSG00000045287 |
237847 |
Rtn4rl1 |
protein_coding |
Q8K0S5 |
FUNCTION: Cell surface receptor that plays a functionally redundant role in postnatal brain development and in regulating axon regeneration in the adult central nervous system (PubMed:22406547, PubMed:27339102). Contributes to normal axon migration across the brain midline and normal formation of the corpus callosum (PubMed:27339102). Protects motoneurons against apoptosis; protection against apoptosis is probably mediated by MAG (PubMed:26335717). Plays a role in inhibiting neurite outgrowth and axon regeneration via its binding to neuronal chondroitin sulfate proteoglycans (PubMed:22406547). Binds heparin (PubMed:22406547). Like other family members, plays a role in restricting the number dendritic spines and the number of synapses that are formed during brain development (PubMed:22325200). Signaling mediates activation of Rho and downstream reorganization of the actin cytoskeleton (PubMed:22325200). {ECO:0000269|PubMed:22325200, ECO:0000269|PubMed:22406547, ECO:0000269|PubMed:26335717, ECO:0000269|PubMed:27339102}. |
Cell membrane;Cell projection;Glycoprotein;GPI-anchor;Heparin-binding;Leucine-rich repeat;Lipoprotein;Membrane;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:237847; |
cell projection [GO:0042995]; cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; membrane raft [GO:0045121]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; chondroitin sulfate binding [GO:0035374]; heparin binding [GO:0008201]; Roundabout binding [GO:0048495]; signaling receptor activity [GO:0038023]; axon guidance [GO:0007411]; corpus callosum development [GO:0022038]; negative chemotaxis [GO:0050919]; negative regulation of axon regeneration [GO:0048681]; negative regulation of neuron projection development [GO:0010977] |
14664809_predominantly expressed in the neurons of the embryonic and adult nervous systems 22406547_The results of this study identify NgR1 and NgR3 as CSPG receptors, suggest that there is functional redundancy among CSPG receptors, and provide evidence for shared mechanisms of MAI and CSPG inhibition. |
ENSG00000185924 |
RTN4RL1 |
333.496142 |
0.0877099617 |
-3.511115 |
0.18021494 |
392.220022 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000272006469568621898080488600560980568829919590708454816000655666952922399547125119952300368741480810055778452235106704269920169509868471972373028514557843060596326659286839419102 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000012185889836674260462512193663994438375688623511068296030874376468091592892148283760046738566249242452774863730046765526700896417355093428252040996310231993551694287631029784993 |
Yes |
No |
53.9891159590086 |
6.7616287410665 |
615.541437796781 |
46.8637318416432 |
| ENSMUSG00000045288 |
16470 |
Ush1g |
protein_coding |
Q80T11 |
FUNCTION: Plays a role in pre-mRNA splicing by regulating the release and transfer of U4/U6.U5 tri-small nuclear ribonucleoprotein (tri-snRNP) complexes from their assembly site in Cajal bodies to nuclear speckles, thereby contributing to the assembly of the pre-catalytic spliceosome on target pre-mRNAs (By similarity). May also participate in recycling of snRNPs back to Cajal bodies during splicing (By similarity). Plays a role in regulating MAGI2-mediated endocytosis (By similarity). Anchoring/scaffolding protein that is a part of the functional network formed by USH1C, USH1G, CDH23 and MYO7A that mediates mechanotransduction in cochlear hair cells. Required for normal development and maintenance of cochlear hair cell bundles. Required for normal hearing. {ECO:0000250|UniProtKB:Q495M9, ECO:0000269|PubMed:21436032, ECO:0000269|PubMed:21709241}. |
ANK repeat;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;Deafness;Hearing;Membrane;Nucleus;Phosphoprotein;Reference proteome;Repeat |
|
This gene encodes a protein that contains three ankyrin domains, a class I PDZ-binding motif and a sterile alpha motif. The encoded protein interacts with harmonin, which is associated with Usher syndrome type 1C. This protein plays a role in the development and maintenance of the auditory and visual systems and functions in the cohesion of hair bundles formed by inner ear sensory cells. Mutations in this gene are associated with combined auditory and vision loss. [provided by RefSeq, Sep 2015]. |
mmu:16470; |
actin cytoskeleton [GO:0015629]; Cajal body [GO:0015030]; ciliary basal body [GO:0036064]; ciliary base [GO:0097546]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; membrane [GO:0016020]; nuclear speck [GO:0016607]; photoreceptor cell cilium [GO:0097733]; photoreceptor connecting cilium [GO:0032391]; photoreceptor inner segment [GO:0001917]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; spectrin binding [GO:0030507]; equilibrioception [GO:0050957]; inner ear morphogenesis [GO:0042472]; inner ear receptor cell differentiation [GO:0060113]; inner ear receptor cell stereocilium organization [GO:0060122]; photoreceptor cell maintenance [GO:0045494]; regulation of clathrin-dependent endocytosis [GO:2000369]; sensory perception of light stimulus [GO:0050953]; sensory perception of sound [GO:0007605] |
12588793_Sans may have an important role in development of the stereocilia bundles 21156003_we examine the effects of null mutation of the Ush1c gene on subcellular localization of Myo7a, Pcdh15 and Sans in the inner ear. 21436032_Usher type 1G protein sans is a critical component of the tip-link complex, a structure controlling actin polymerization in stereocilia. 22381527_USH1G (Sans) form the upper tip-link complex in adult mice 24608321_In Usher syndrome 1G, mutations in SANS eliminate Magi2 binding and thereby deregulate endocytosis, lead to defective ciliary transport modules and ultimately disrupt photoreceptor cell function inducing retinal degeneration. 26936824_These results clearly show that the development of early-onset progressive hearing loss (ePHL) requires at least two mutant alleles of the Ush1g and Cdh23 genes. Our results also suggest that because the SANS and CDH23 proteins form a complex in the stereocilia, the interaction between these proteins may play key roles in the maintenance of stereocilia and the prevention of ePHL. 28966015_C-terminal hairpin extensions of the whirlin PDZ domains mediate the transient supramodular assembly, which improves the binding capacity of the first domain towards its partners, such as Sans. 29386551_under normal housing conditions, Ush1g(-/-) and Ush1c(-/-) albino mice have dysfunctional cone photoreceptors whereas pigmented knockout animals have normal photoreceptors. |
ENSG00000182040 |
USH1G |
26.476423 |
4.9041356967 |
2.293999 |
0.55022070 |
17.162796 |
0.00003430903074888676029330705774533782914659241214394569396972656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002286068050599488941064529878843814003630541265010833740234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.3365290333614 |
16.9314527720686 |
8.83673122305122 |
2.78887177582433 |
| ENSMUSG00000045294 |
231070 |
Insig1 |
protein_coding |
Q8BGI3 |
FUNCTION: Oxysterol-binding protein that mediates feedback control of cholesterol synthesis by controlling both endoplasmic reticulum to Golgi transport of SCAP and degradation of HMGCR (PubMed:16100574). Acts as a negative regulator of cholesterol biosynthesis by mediating the retention of the SCAP-SREBP complex in the endoplasmic reticulum, thereby blocking the processing of sterol regulatory element-binding proteins (SREBPs) SREBF1/SREBP1 and SREBF2/SREBP2 (By similarity). Binds oxysterol, including 25-hydroxycholesterol, regulating interaction with SCAP and retention of the SCAP-SREBP complex in the endoplasmic reticulum (PubMed:16100574). In presence of oxysterol, interacts with SCAP, retaining the SCAP-SREBP complex in the endoplasmic reticulum, thereby preventing SCAP from escorting SREBF1/SREBP1 and SREBF2/SREBP2 to the Golgi (By similarity). Sterol deprivation or phosphorylation by PCK1 reduce oxysterol-binding, disrupting the interaction between INSIG1 and SCAP, thereby promoting Golgi transport of the SCAP-SREBP complex, followed by processing and nuclear translocation of SREBF1/SREBP1 and SREBF2/SREBP2 (By similarity). Also regulates cholesterol synthesis by regulating degradation of HMGCR: initiates the sterol-mediated ubiquitin-mediated endoplasmic reticulum-associated degradation (ERAD) of HMGCR via recruitment of the reductase to the ubiquitin ligases AMFR/gp78 and/or RNF139 (By similarity). Also regulates degradation of SOAT2/ACAT2 when the lipid levels are low: initiates the ubiquitin-mediated degradation of SOAT2/ACAT2 via recruitment of the ubiquitin ligases AMFR/gp78 (By similarity). {ECO:0000250|UniProtKB:O15503, ECO:0000269|PubMed:16100574}. |
Cholesterol metabolism;Endoplasmic reticulum;Isopeptide bond;Lipid metabolism;Lipid-binding;Membrane;Phosphoprotein;Reference proteome;Steroid metabolism;Sterol metabolism;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:231070; |
endoplasmic reticulum [GO:0005783]; SREBP-SCAP-Insig complex [GO:0032937]; oxysterol binding [GO:0008142]; cellular response to insulin stimulus [GO:0032869]; cellular response to sterol [GO:0036315]; cholesterol biosynthetic process [GO:0006695]; cholesterol homeostasis [GO:0042632]; cholesterol metabolic process [GO:0008203]; cranial suture morphogenesis [GO:0060363]; inner ear morphogenesis [GO:0042472]; middle ear morphogenesis [GO:0042474]; negative regulation of cargo loading into COPII-coated vesicle [GO:1901303]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of fatty acid biosynthetic process [GO:0045717]; negative regulation of protein exit from endoplasmic reticulum [GO:0070862]; negative regulation of steroid biosynthetic process [GO:0010894]; response to sterol depletion [GO:0006991]; roof of mouth development [GO:0060021]; SREBP signaling pathway [GO:0032933]; SREBP-SCAP complex retention in endoplasmic reticulum [GO:0036316]; sterol biosynthetic process [GO:0016126]; triglyceride metabolic process [GO:0006641] |
12869692_insig-1 expression restricts lipogenesis in mature adipocytes and blocks differentiation in preadipocytes 15073165_the regulation of Insig-1 by PPARgamma agonists could in turn regulate SREBP processing and thus couple insulin sensitizers with the regulation of lipid homeostasis 16100574_Results define Insig proteins 1 and 2 as essential elements of the feedback inhibition system of cholesterol synthesis in mouse liver. 16327801_IRS2, HK2 and INSIG1 are direct targets of TFE3 16955138_Treatment of pregnant mice with the HMG-CoA reductase inhibitor lovastatin reduced sterol synthesis in Insig1-knockout embryos and reduced the pre-cholesterol intermediates ameliorating the clefting. 18187584_Activation of CAR and PXR in mouse liver results in activation of Insig-1 along with reduced protein levels of the active form of sterol regulatory element binding protein 1 (Srebp-1). 18627005_Insig-1 is a direct PPARdelta target gene in hepatocytes. PPARdelta inhibited SREBP-1 activation via induction of Insig-1. Overexpression of PPARdelta induced the expression of Insig-1, suppressed SREBP-1 activation, and ameliorated hepatic steatosis. 19360322_Silibinin inhibits adipocyte differentiation through a potential up-regulation of insig-1 and insig-2 at an early phase in adipocyte differentiation. 20090767_Insig deficiency in skin causes accumulation of cholesterol precursors, and this impairs normal hair development 20603810_analysis of mRNA abundance and expression of SLC27A, ACC, SCD, FADS, LPIN, INSIG, and PPARGC1 gene isoforms in mouse mammary glands during the lactation cycle 23087214_we consistently observe involvement of gp78 in Insig-1 degradation, we find no substantive evidence to support roles for either gp78 or TRC8 in the robust sterol-accelerated degradation of HMG-CoA reductase. 23919961_reducing Insig1 mRNA to a similar degree observed in morbidly obese human WAT accelerates adipocyte differentiation and enhances lipogenesis, and consequently, lipid accumulation 24337570_the essential role of Insig1,2 proteins in the sterol homeostasis of enterocytes. 25526307_myeloid-cell-specific Insig1-/- mice are more susceptible to HSV-1 infection 29899135_Intracellular biosynthesis of lipids and cholesterol by Scap and Insig in mesenchymal cells regulates long bone growth and chondrocyte homeostasis. 33722690_Suppression of insulin-induced gene 1 (INSIG1) function promotes hepatic lipid remodelling and restrains NASH progression. |
ENSG00000186480 |
INSIG1 |
1285.353602 |
0.4968479540 |
-1.009124 |
0.07922816 |
161.047294 |
0.00000000000000000000000000000000000066808509208326658251141739135380901791662549854487277142353158565198102660614483420510007635425599181733247178271994926035404205322265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000669491587013968222393624131496458854585883995543080270985024432118301466817864931207308107179165812539167745853774249553680419921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
865.201785517399 |
47.5379811829239 |
1741.38139974404 |
69.3929850948341 |
| ENSMUSG00000045319 |
227545 |
Proser2 |
protein_coding |
Q8C5R2 |
|
Methylation;Phosphoprotein;Reference proteome |
|
|
mmu:227545; |
|
|
ENSG00000148426 |
PROSER2 |
93.696481 |
0.2823020299 |
-1.824689 |
0.27853375 |
42.440246 |
0.00000000007287494295992848224277978311618484720535438015076579176820814609527587890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000011771273925404356528172049566874010628758639995794510468840599060058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.4524824462371 |
8.24709543275043 |
150.379657060361 |
20.2005160937437 |
| ENSMUSG00000045328 |
229841 |
Cenpe |
protein_coding |
E9QKK1 |
Human_homologues FUNCTION: Microtubule plus-end-directed kinetochore motor which plays an important role in chromosome congression, microtubule-kinetochore conjugation and spindle assembly checkpoint activation. Drives chromosome congression (alignment of chromosomes at the spindle equator resulting in the formation of the metaphase plate) by mediating the lateral sliding of polar chromosomes along spindle microtubules towards the spindle equator and by aiding the establishment and maintenance of connections between kinetochores and spindle microtubules (PubMed:7889940, PubMed:23891108, PubMed:25395579). The transport of pole-proximal chromosomes towards the spindle equator is favored by microtubule tracks that are detyrosinated (PubMed:25908662). Acts as a processive bi-directional tracker of dynamic microtubule tips; after chromosomes have congressed, continues to play an active role at kinetochores, enhancing their links with dynamic microtubule ends (PubMed:23955301). Suppresses chromosome congression in NDC80-depleted cells and contributes positively to congression only when microtubules are stabilized (PubMed:25743205). Plays an important role in the formation of stable attachments between kinetochores and spindle microtubules (PubMed:17535814) The stabilization of kinetochore-microtubule attachment also requires CENPE-dependent localization of other proteins to the kinetochore including BUB1B, MAD1 and MAD2. Plays a role in spindle assembly checkpoint activation (SAC) via its interaction with BUB1B resulting in the activation of its kinase activity, which is important for activating SAC. Necessary for the mitotic checkpoint signal at individual kinetochores to prevent aneuploidy due to single chromosome loss (By similarity). {ECO:0000250|UniProtKB:Q6RT24, ECO:0000269|PubMed:17535814, ECO:0000269|PubMed:23891108, ECO:0000269|PubMed:23955301, ECO:0000269|PubMed:25395579, ECO:0000269|PubMed:25743205, ECO:0000269|PubMed:25908662, ECO:0000269|PubMed:7889940}. |
ATP-binding;Coiled coil;Motor protein;Nucleotide-binding;Proteomics identification;Reference proteome |
|
|
mmu:229841; |
intercellular bridge [GO:0045171]; kinetochore [GO:0000776]; kinetochore microtubule [GO:0005828]; midbody [GO:0030496]; mitotic spindle midzone [GO:1990023]; nucleus [GO:0005634]; ATP binding [GO:0005524]; kinetochore binding [GO:0043515]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; kinetochore assembly [GO:0051382]; lateral attachment of mitotic spindle microtubules to kinetochore [GO:0099607]; microtubule plus-end directed mitotic chromosome migration [GO:0099606]; mitotic chromosome movement towards spindle pole [GO:0007079]; mitotic spindle organization [GO:0007052]; positive regulation of protein kinase activity [GO:0045860]; regulation of mitotic metaphase/anaphase transition [GO:0030071] |
12068923_results suggest that CENP-E is implicated in the spindle checkpoint, and in chromosome alignment, during the two meiotic divisions in vertebrate males 12361599_Deletion of the mouse gene CENP-E causes unstable kinetochore-microtubule capture and chromosomal instability. (CENP-E) 19283064_Shugoshin 2 is necessary for the loading of MCAK at the inner centromere, but is dispensable for the loading of the outer kinetochore proteins BubR1 and CENP-E. 22513372_CENP-E stabilises BubR1, thereby impacting meiosis I progression, and mediates bi-orientation by promoting kinetochore reorientation and preventing chromosomal drift towards the poles. 24133140_Exacerbating chromosome missegregation in CENP-E+/- mice by reducing levels of another mitotic checkpoint component, Mad2, results in elevated cell death and decreased tumor formation compared with reduction of either protein alone. 33862458_CENPE contributes to pulmonary vascular remodeling in pulmonary hypertension. 35037651_Functional Assessment of Kinesin-7 CENP-E in Spermatocytes using In Vivo Inhibition, Immunofluorescence and Flow Cytometry. 35680098_Kinesin-7 CENP-E is essential for chromosome alignment and spindle assembly of mouse spermatocytes. |
ENSG00000138778 |
CENPE |
1158.821772 |
3.3880156263 |
1.760441 |
0.11306717 |
232.252985 |
0.00000000000000000000000000000000000000000000000000019231549829732386216612564353853650480245898316768203964776626451195157647330198286312780181495638716843078832037412134501349917302913394845376160446903668344020843505859375000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000361648107415412021866713276490425596794003579305400041987180905394352816905455000606658046074293624435296672872454397450696752039367432018934778170660138130187988281250000000000000000000000000000000000000000000 |
Yes |
No |
1787.13081511851 |
140.204775750613 |
527.486001319529 |
31.8632323456674 |
| ENSMUSG00000045377 |
67020 |
Tmem88 |
protein_coding |
Q9D0N8 |
FUNCTION: Inhibits the Wnt/beta-catenin signaling pathway. Crucial for heart development and acts downstream of GATA factors in the pre-cardiac mesoderm to specify lineage commitment of cardiomyocyte development (By similarity). {ECO:0000250}. |
Cell membrane;Developmental protein;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Wnt signaling pathway |
|
|
mmu:67020; |
cytosol [GO:0005829]; plasma membrane [GO:0005886]; PDZ domain binding [GO:0030165]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; protein localization to plasma membrane [GO:0072659]; protein stabilization [GO:0050821]; Wnt signaling pathway [GO:0016055] |
21044957_TMEM88 associates with Dvl proteins and regulates Wnt signaling in a context-dependent manner 32451556_TMEM88 modulates the secretion of inflammatory factors by regulating YAP signaling pathway in alcoholic liver disease. |
ENSG00000167874 |
TMEM88 |
157.488848 |
2.0252553321 |
1.018104 |
0.20415545 |
24.666135 |
0.00000068171611217345114257248634564989586692718148697167634963989257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000062553613377265112802626256804927606935962103307247161865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
213.646343293448 |
30.334049326171 |
105.4910656973 |
11.571437706239 |
| ENSMUSG00000045392 |
258571 |
Or5m3b |
protein_coding |
Q8VFK5 |
Human_homologues FUNCTION: Odorant receptor. {ECO:0000305}. |
Cell membrane;G-protein coupled receptor;Membrane;Olfaction;Receptor;Reference proteome;Sensory transduction;Transducer;Transmembrane;Transmembrane helix |
|
Olfactory receptors interact with odorant molecules in the nose, to initiate a neuronal response that triggers the perception of a smell. The olfactory receptor proteins are members of a large family of G-protein-coupled receptors (GPCR) arising from single coding-exon genes. Olfactory receptors share a 7-transmembrane domain structure with many neurotransmitter and hormone receptors and are responsible for the recognition and G protein-mediated transduction of odorant signals. The olfactory receptor gene family is the largest in the genome. The nomenclature assigned to the olfactory receptor genes and proteins for this organism is independent of other organisms. [provided by RefSeq, Jul 2008]. |
mmu:258571; |
membrane [GO:0016020]; plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; odorant binding [GO:0005549]; olfactory receptor activity [GO:0004984]; G protein-coupled receptor signaling pathway [GO:0007186]; sensory perception of smell [GO:0007608] |
|
ENSG00000174937 |
OR5M3 |
15.541898 |
0.1352162243 |
-2.886660 |
0.94400135 |
8.268255 |
0.00403442535905668923229550060227666108403354883193969726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0154364147372900006177642850957454356830567121505737304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.61486718215161 |
2.5377587290056 |
26.7339751601724 |
13.1788513041749 |
| ENSMUSG00000045573 |
18619 |
Penk |
protein_coding |
P22005 |
FUNCTION: [Met-enkephalin]: Neuropeptide that competes with and mimic the effects of opiate drugs. They play a role in a number of physiologic functions, including pain perception and responses to stress. {ECO:0000250|UniProtKB:P01210}.; FUNCTION: [Leu-enkephalin]: Neuropeptide that competes with and mimic the effects of opiate drugs. They play a role in a number of physiologic functions, including pain perception and responses to stress. {ECO:0000250|UniProtKB:P01210}.; FUNCTION: [Met-enkephalin-Arg-Phe]: Met-enkephalin-Arg-Phe neuropeptide acts as a strong ligand of Mu-type opioid receptor OPRM1 (PubMed:6933569, PubMed:35201898). Met-enkephalin-Arg-Phe-binding to OPRM1 in the nucleus accumbens of the brain increases activation of OPRM1, leading to long-term synaptic depression of glutamate release (PubMed:35201898). {ECO:0000269|PubMed:35201898, ECO:0000269|PubMed:6933569}.; FUNCTION: [PENK(114-133)]: Increases glutamate release in the striatum and decreases GABA concentration in the striatum. {ECO:0000250|UniProtKB:P04094}.; FUNCTION: [PENK(238-259)]: Increases glutamate release in the striatum. {ECO:0000250|UniProtKB:P04094}. |
Cleavage on pair of basic residues;Cytoplasmic vesicle;Disulfide bond;Endorphin;Neuropeptide;Opioid peptide;Phosphoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:18619; |
axon [GO:0030424]; axon terminus [GO:0043679]; cell body fiber [GO:0070852]; chromaffin granule lumen [GO:0034466]; dendrite [GO:0030425]; extracellular region [GO:0005576]; neuronal cell body [GO:0043025]; neuronal dense core vesicle lumen [GO:0099013]; perikaryon [GO:0043204]; plasma membrane [GO:0005886]; symmetric synapse [GO:0032280]; synaptic vesicle lumen [GO:0034592]; opioid peptide activity [GO:0001515]; opioid receptor binding [GO:0031628]; aggressive behavior [GO:0002118]; aging [GO:0007568]; behavioral fear response [GO:0001662]; cellular response to cAMP [GO:0071320]; cellular response to oxidative stress [GO:0034599]; cellular response to transforming growth factor beta stimulus [GO:0071560]; cellular response to virus [GO:0098586]; cellular response to vitamin D [GO:0071305]; chemical synaptic transmission [GO:0007268]; general adaptation syndrome, behavioral process [GO:0051867]; glial cell proliferation [GO:0014009]; locomotory behavior [GO:0007626]; locomotory exploration behavior [GO:0035641]; neuropeptide signaling pathway [GO:0007218]; osteoblast differentiation [GO:0001649]; positive regulation of behavioral fear response [GO:2000987]; regulation of long-term synaptic depression [GO:1900452]; response to bacterium [GO:0009617]; response to calcium ion [GO:0051592]; response to epinephrine [GO:0071871]; response to estradiol [GO:0032355]; response to ethanol [GO:0045471]; response to hypoxia [GO:0001666]; response to lipopolysaccharide [GO:0032496]; response to morphine [GO:0043278]; response to nicotine [GO:0035094]; response to radiation [GO:0009314]; response to toxic substance [GO:0009636]; sensory perception [GO:0007600]; sensory perception of pain [GO:0019233]; startle response [GO:0001964]; synaptic signaling via neuropeptide [GO:0099538] |
12486185_Morphine dependence is examined in preproenkephalin knockout mice. 12864972_A defect in activation seen in preproenkephalin (Penk)-deficient T cells results in a reduced number of cells entering into cell cycle; proliferation can be rescued by increasing antigen or by co-incubation of Penk-deficient cells with wild-type cells. 15197532_mice with the Penk1 mutation in the DBA/2J, but not on other genetic backgrounds, showed an increased opioid-dependent stress-induced analgesia 15689546_Endogenous opioid peptides derived from preproenkephalin are involved in the antinociceptive and rewarding properties of nicotine and participate in the expression of physical nicotine dependence. 16095981_These data implicate enkephalin as a signaling molecule in the control of hypoxic depression of metabolism in mice. 17375141_Preproenkephalin knockout mice show no depression-related phenotype. 18280051_Increase in ACTH level after zero-maze test failed to reach the level of significance in Penk1(-/-) mice. 18463254_Met-enkephalin levels in neuronal projections from entorhinal cortex and dentate gyrus were increased, as were preproenkephalin mRNA levels. Elevations may contribute to cognitive impairments. (Met-enkephalin, preproenkephalin) 18780300_Preproenkephalin and GABA colocalized in a subpopulation of neurons in the spinal dorsal horn. They are likely to represent local inhibitory dorsal horn interneurons involved in the modulation of pain transmission. 19046386_As PPE-derived peptides are endogenous ligands for MOR in the neostriatum and few axon collaterals of matrix neurons enter the striosome compartment, the present results raised the question of the target of those peptides produced by matrix neurons. 19523041_Administration of the antagonist prevented the acquisition of MDMA-induced CPP, suggesting an implication of the delta opioid receptors in this behavior. 19997907_Inter-strain differences in PENK and PDYN genes expression in the nucleus accumbens parallel sensitivity of the selected mouse strains to rewarding effects of morphine. 20204609_Preproenkephalin deletion in Hyp mice (Phex-deficient) partially rescues their bone mineralization defect.A role for Penk in osteoblast differentiation. 20406487_c-fos expression in specific brain areas in preproenkephalin deficiency 21124002_ENKephalinergic neurons develop according to a rostrocaudal and ventrodorsal gradient. 22067363_Preproenkephalin-enhanced green fluorescent protein (GFP) transgenic mice were used to study ENKergic neuron distribution patterns. 24040390_upregulation of striatal enkephalin might have a role in early stage Huntington Disease in a mouse model 24804898_PENK KO mice do not display anxiety and depression-related behavioral changes after chronic mild stress. 25754760_PENK knockout exhibited fewer bouts of licking than wild types, even though the length of bouts was similar to that of wild types, a pattern that suggests diminished food motivation. 26634308_Nav1.7 deletion has profound effects on gene expression, leading to an upregulation of enkephalin precursor Penk mRNA and met-enkephalin protein in sensory neurons. 30040980_Deficiency of preproenkephalin (PENK) or proopioimelanocortin (POMC) gene blocks analgesic effects following systemic administration of N-methyl-kyotorphin, NMYR and arginine in mice. 31638440_In an ongoing expression screen, this study identified that Tfap2b, Casq2, Penk, Zic1, and Zic3 are expressed in unique cell types in and around the developing eye. 35201898_Angiotensin-converting enzyme gates brain circuit-specific plasticity via an endogenous opioid. 35937204_Cellular Profiles of Prodynorphin and Preproenkephalin mRNA-Expressing Neurons in the Anterior Olfactory Tubercle of Mice. |
ENSG00000181195 |
PENK |
76.204387 |
0.1059368127 |
-3.238724 |
0.38674184 |
69.489579 |
0.00000000000000007681929945637476112240673444464497172416979722288365994842251893715001642704010009765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000023079123655216970213323312958920260436386246577500358512224920559674501419067382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.7814438533866 |
4.01007543537329 |
139.530758769256 |
24.143695961098 |
| ENSMUSG00000045667 |
276829 |
Smtnl2 |
protein_coding |
Q8CI12 |
|
Coiled coil;Phosphoprotein;Reference proteome |
|
|
mmu:276829; |
filamentous actin [GO:0031941]; I band [GO:0031674]; M band [GO:0031430]; microtubule organizing center [GO:0005815]; protein phosphatase 1 binding [GO:0008157]; tropomyosin binding [GO:0005523]; actin cytoskeleton organization [GO:0030036]; positive regulation of vasoconstriction [GO:0045907] |
35551947_Smtnl2 regulates apoptotic germ cell clearance and lactate metabolism in mouse Sertoli cells. |
ENSG00000188176 |
SMTNL2 |
32.317951 |
0.1860327850 |
-2.426371 |
0.50542754 |
22.968642 |
0.00000164665588372414286336088348750017473776097176596522331237792968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000140909339443180586971685400166087731577135855332016944885253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
9.54127030932287 |
3.41144488725471 |
51.2881119887479 |
12.1420871539359 |
| ENSMUSG00000045763 |
70350 |
Basp1 |
protein_coding |
Q91XV3 |
|
Cell membrane;Cell projection;Direct protein sequencing;Isopeptide bond;Lipoprotein;Membrane;Myristate;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:70350; |
cell junction [GO:0030054]; chromatin [GO:0000785]; COP9 signalosome [GO:0008180]; cytoplasm [GO:0005737]; growth cone [GO:0030426]; nuclear matrix [GO:0016363]; nuclear speck [GO:0016607]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; PML body [GO:0016605]; protein domain specific binding [GO:0019904]; transcription cis-regulatory region binding [GO:0000976]; transcription corepressor activity [GO:0003714]; negative regulation of DNA-templated transcription [GO:0045892]; podocyte differentiation [GO:0072112] |
31167803_Our results show that the sustained presence of the WT1-BASP1 complex is required to maintain taste receptor cells in their differentiated 33692421_BASP1 labels neural stem cells in the neurogenic niches of mammalian brain. 35412917_A high-affinity cocaine binding site associated with the brain acid soluble protein 1. |
ENSG00000176788 |
BASP1 |
631.402116 |
2.2612452870 |
1.177117 |
0.12129373 |
92.700510 |
0.00000000000000000000060837905563619702614593263260918233688043821380438610515149766566622702157474122941493988037109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000269384586495655624634907986550878952516233238071333513409533200189116541878320276737213134765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
865.656871227222 |
72.8653607319162 |
382.823073728675 |
24.8433760336312 |
| ENSMUSG00000045777 |
320802 |
Ifitm10 |
protein_coding |
Q8BR26 |
|
Alternative splicing;Cell membrane;Lipoprotein;Membrane;Palmitate;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:320802; |
plasma membrane [GO:0005886] |
|
ENSG00000244242 |
IFITM10 |
93.510839 |
0.3487661906 |
-1.519668 |
0.46272859 |
10.439479 |
0.00123350050147063043312622987457416456891223788261413574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0055686661643155428366713799448461941210553050041198730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.2604090809194 |
15.4194945797354 |
135.507426914827 |
32.1937566411062 |
| ENSMUSG00000045903 |
225872 |
Npas4 |
protein_coding |
Q8BGD7 |
FUNCTION: Transcription factor expressed in neurons of the brain that regulates the excitatory-inhibitory balance within neural circuits and is required for contextual memory in the hippocampus (PubMed:18815592, PubMed:22194569, PubMed:23029555, PubMed:24201284, PubMed:24855953). Plays a key role in the structural and functional plasticity of neurons (PubMed:23172225). Acts as an early-response transcription factor in both excitatory and inhibitory neurons, where it induces distinct but overlapping sets of late-response genes in these two types of neurons, allowing the synapses that form on inhibitory and excitatory neurons to be modified by neuronal activity in a manner specific to their function within a circuit, thereby facilitating appropriate circuit responses to sensory experience (PubMed:24201284, PubMed:24855953). In excitatory neurons, activates transcription of BDNF, which in turn controls the number of GABA-releasing synapses that form on excitatory neurons, thereby promoting an increased number of inhibitory synapses on excitatory neurons (PubMed:18815592, PubMed:22194569, PubMed:24201284). In inhibitory neurons, regulates a distinct set of target genes that serve to increase excitatory input onto somatostatin neurons, probably resulting in enhanced feedback inhibition within cortical circuits (PubMed:24855953). The excitatory and inhibitory balance in neurons affects a number of processes, such as short-term and long-term memory, acquisition of experience, fear memory, response to stress and social behavior (PubMed:18815592, PubMed:22194569, PubMed:23029555, PubMed:24201284, PubMed:27238022). Acts as a regulator of dendritic spine development in olfactory bulb granule cells in a sensory-experience-dependent manner by regulating expression of MDM2 (PubMed:25088421). Efficient DNA binding requires dimerization with another bHLH protein, such as ARNT, ARNT2 or BMAL1 (PubMed:14701734, PubMed:15363889, PubMed:19284974). Can activate the CME (CNS midline enhancer) element (PubMed:14701734, PubMed:15363889). {ECO:0000269|PubMed:14701734, ECO:0000269|PubMed:15363889, ECO:0000269|PubMed:18815592, ECO:0000269|PubMed:22194569, ECO:0000269|PubMed:23029555, ECO:0000269|PubMed:23172225, ECO:0000269|PubMed:24201284, ECO:0000269|PubMed:24855953, ECO:0000269|PubMed:25088421, ECO:0000269|PubMed:27238022}. |
3D-structure;Activator;Coiled coil;Differentiation;DNA-binding;Neurogenesis;Nucleus;Reference proteome;Repeat;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:225872; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; postsynapse [GO:0098794]; transcription regulator complex [GO:0005667]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; protein heterodimerization activity [GO:0046982]; protein-containing complex binding [GO:0044877]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; cell differentiation [GO:0030154]; cellular response to corticosterone stimulus [GO:0071386]; excitatory postsynaptic potential [GO:0060079]; inhibitory postsynaptic potential [GO:0060080]; inhibitory synapse assembly [GO:1904862]; learning [GO:0007612]; long-term memory [GO:0007616]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of synaptic plasticity [GO:0048167]; regulation of synaptic transmission, GABAergic [GO:0032228]; regulation of transcription by RNA polymerase II [GO:0006357]; short-term memory [GO:0007614]; social behavior [GO:0035176] |
15380003_mRNA expression in the hippocampus 1 h following generalized convulsive seizure, in a pattern which is typical for other activity-dependant genes expressed in this structure. 18815592_identification of transcription factor Npas4, that plays a role in the development of inhibitory synapses by regulating the expression of activity-dependent genes, which in turn control the number of GABA-releasing synapses that form on excitatory neurons 19001414_NXF gene could be induced in response to several neurodegenerative stimuli/excitations for the cell protection, and thus provide an 'on demand' cell-protection system in nervous tissue 20626564_Corticosterone-induced down-regulation of Npas4 expression may play a role in stress-induced impairment of hippocampal function. 22194569_by recruiting RNA polymerase II to promoters and enhancers of target genes, Npas4 regulates a learning-specific transcriptional program in CA3, which suggests Npas4 is a master regulator of activity-regulated gene programs and central to memory formation 23020797_These results suggest that transcription of Npas4 is down-regulated by stress via the binding of agonist-bound glucocorticoid receptor to its promoter. 23029555_Npas4 may play a major role in the regulation of cognitive and social functions in the brain with possible implications for developmental disorders such as schizophrenia and autism. 23172225_NPAS4 plays an important role in the structural and functional plasticity of neurons 23300775_Optical activation of striatal medium spiny neurons induced immediate early gene expression evidenced by increased Npas4 proetin. 23637184_This study demonistrated that Npas4 is involved in activity-dependent changes to input connectivity to adult-born DG GCs but not to their output connectivity in CA3. 23656887_Npas4 is a key activity-dependent regulator that improves beta-cell efficiency in the face of stress. 24201284_sensory stimuli, by inducing NPAS4 and its target genes, differentially control spatial features of neuronal inhibition in a way that restricts the output of the neuron while creating a dendritic environment that is permissive for plasticity 24291638_Our results reveal REST and microRNA dependent mechanisms that restrict NPAS4 expression to the brain 24855953_Study describes a transcriptional program that is induced by neuronal activity in inhibitory neurons; while neuronal activity induces expression of early-response transcription factors such as Npas4 in both excitatory and inhibitory neurons, Npas4 activates distinct programs of late-response genes in inhibitory and excitatory neurons. 24887558_we provide the first evidence that Npas4 is expressed during embryonic development and that it may have a developmental role that is unrelated to its function in the adult brain 25088421_Npas4 regulates Mdm2 expression during dendritic spine development in newborn olfactory bulb after sensory experience. 25549857_Data showed that differential levels of Npas4 expression in the brain may regulate anxiety, depression and cognition related disorders. 25785202_Cortex activation by sensory conditioning was found to upregulate the expression of both Npas4 and c-Fos. 25912092_induction of Npas4 identifies the area influenced by cortical spreading depression propagation. 26222956_Results suggest that transcription of the Npas4 gene is downregulated by stress through DNA methylation of its promoter 26563879_These findings suggest that 11beta-HSD1 may contribute to the decline in Npas4 and Arc mRNA levels associated with memory impairment during ageing 26663079_beta cell cytotoxicity of the calcineurin inhibitor and immunosuppressant tacrolimus (FK-506) was prevented by Npas4 overexpression. 27189618_ectopic expression of miR-744 and miR-224 during neural differentiation resulted in downregulation of endogenous Npas4 expression. 27574128_The findings of this study suggest that Npas4 may be involved in post-stroke psychiatric changes related to anxiety ad sociability. 27993645_Using a series of behavioral, molecular, pharmacological and genetic approaches in mice, study shows that deficiency in Npas4 affects adolescent expression of multiple markers of GABAergic transmission in the PFC, including parvalbumin and GAD67, in a sex-specific manner. Abnormal pattern of expression of GABAergic markers is associated with sex-specific and age-dependent cognitive and emotional impairments. 28082451_NPAS4 is expressed in endothelial cells, regulates VE-cadherin expression and regulates sprouting angiogenesis. 29222951_the present results suggest that Npas4 controls neuronal homeostatic scaling during epileptogenesis through the induction of Homer1a as negative feedback machinery for the management of epilepsy 29298418_NPAS4 is critical for the coordination of metabolism during the stimulation of electrically excitable cells; its loss leads to the defects in cellular metabolism that underlie the cellular dysfunction that occurs in metabolic disease. 29345055_Results suggest that Npas4 mediates susceptibility to adolescent stress and subsequent cognitive functioning and inhibitory tone in adulthood. This shows a novel gene by environment interaction related to resilience vs vulnerability to stress, with implications for adolescent onset disorders like schizophrenia. 29429933_Npas4 is a critical regulator of experience-dependent, functional plasticity at mossy fiber-CA3 pyramidal synapses during contextual memory formation. 30052197_NPAS4 is an important regulator of plasticity mediated by endogenous cannabinoids. 30093305_The observed reduction of the infarct volume was comparable to the protection obtained by intracerebroventricular injection of recombinant Activin A or SerpinB2 or by stereotactic delivery 3 weeks prior to the injury of a recombinant adeno-associated virus containing an expression cassette for the potent neuroprotective transcription factor Npas4 30792384_Npas4 expression within PV+ neurons replicates deficits in short-term memory observed after neonatal ketamine, but does not reproduce disturbances in general activity in mice. 30798405_Hippocampal NPAS4 plays a key role in the beneficial effects of environmental enrichment on pain sensitivity, depression and memory deficit. 30962277_These findings highlight the significance of cell-type-specificity in mechanisms underlying reward regulation by NPAS2 and extend our knowledge of its function 31585079_Action potential and EPSPs induce NPAS4 in CA1 pyramidal neurons through distinct mechanisms. 31801086_Phosphorylation of Npas4 by MAPK Regulates Reward-Related Gene Expression and Behaviors. 32327470_Amyloid Precursor Protein (APP) Controls the Expression of the Transcriptional Activator Neuronal PAS Domain Protein 4 (NPAS4) and Synaptic GABA Release. 34289353_Npas4 regulates IQSEC3 expression in hippocampal somatostatin interneurons to mediate anxiety-like behavior. 34349016_Ras-like Gem GTPase induced by Npas4 promotes activity-dependent neuronal tolerance for ischemic stroke. 34416169_NPAS4 regulates the transcriptional response of the suprachiasmatic nucleus to light and circadian behavior. 34418347_NPAS4 suppresses propofol-induced neurotoxicity by inhibiting autophagy in hippocampal neuronal cells. 34661342_Npas4 regulates medium spiny neuron physiology and gates cocaine-induced hyperlocomotion. 35042829_Dysregulation of Npas4 and Inhba expression and an altered excitation-inhibition balance are associated with cognitive deficits in DBA/2 mice. 36099920_Functionally distinct NPAS4-expressing somatostatin interneuron ensembles critical for motor skill learning. 36780219_NPAS4 in the medial prefrontal cortex mediates chronic social defeat stress-induced anhedonia-like behavior and reductions in excitatory synapses. |
ENSG00000174576 |
NPAS4 |
1146.725531 |
2.3918936265 |
1.258153 |
0.09113835 |
188.411856 |
0.00000000000000000000000000000000000000000070631114107496611546688657915593326983519089424934804935606655924679770954276687902312711146475606293786603283660352481376776268007233738899230957031250000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000919532589816571203907343809199204568059604075322879631837203506967921789277305688254631991127872020334605568381292073354416061192750930786132812500000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1590.08151601185 |
100.515914186182 |
664.779360747978 |
32.4951092201891 |
| ENSMUSG00000046157 |
268567 |
Tmem229b |
protein_coding |
Q8BFQ2 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:268567; |
membrane [GO:0016020]; macrophage activation [GO:0042116]; response to bacterium [GO:0009617] |
31544253_Our data have uncovered the critical contribution of miR-505 in AMD, which is predominantly mediated by downregulation of TMEM229B. |
ENSG00000198133 |
TMEM229B |
320.995431 |
0.4489549280 |
-1.155357 |
0.15951588 |
51.940157 |
0.00000000000057218171772673969074433493381068722679130833519423049438046291470527648925781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000117617704782910916960856812919130058754657586206349151325412094593048095703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
198.471425363745 |
21.9893261171957 |
442.07427736309 |
35.3567820897313 |
| ENSMUSG00000046550 |
278240 |
Spin2c |
protein_coding |
Q6NVE3 |
FUNCTION: May be involved in the regulation of cell cycle progression. Exhibits H3K4me3-binding activity. {ECO:0000250|UniProtKB:Q99865}. |
Cell cycle;Nucleus;Reference proteome |
|
|
mmu:278240; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; methylated histone binding [GO:0035064]; cell cycle [GO:0007049]; gamete generation [GO:0007276]; regulation of DNA-templated transcription [GO:0006355] |
|
|
|
35.333585 |
2.3558255792 |
1.236233 |
0.41935417 |
8.673290 |
0.00322907726477144035176491954075572721194475889205932617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0127522180184077207204529358364197832997888326644897460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
50.2111594824962 |
14.8014835243228 |
21.3136150337136 |
4.88900759499638 |
| ENSMUSG00000046688 |
211550 |
Tifa |
protein_coding |
Q793I8 |
FUNCTION: Adapter molecule that plays a key role in the activation of pro-inflammatory NF-kappa-B signaling following detection of bacterial pathogen-associated molecular pattern metabolites (PAMPs) (PubMed:11798190). Promotes activation of an innate immune response by inducing the oligomerization and polyubiquitination of TRAF6, which leads to the activation of TAK1 and IKK through a proteasome-independent mechanism (By similarity). TIFA-dependent innate immune response is triggered by ADP-D-glycero-beta-D-manno-heptose (ADP-Heptose), a potent PAMP present in all Gram-negative and some Gram-positive bacteria: ADP-Heptose is recognized by ALPK1, which phosphorylates TIFA at Thr-9, leading to TIFA homooligomerization and subsequent activation of pro-inflammatory NF-kappa-B signaling (By similarity). {ECO:0000250|UniProtKB:Q96CG3, ECO:0000269|PubMed:11798190}. |
3D-structure;Cytoplasm;Immunity;Innate immunity;Phosphoprotein;Reference proteome |
|
|
mmu:211550; |
cytoplasm [GO:0005737]; cytosolic pattern recognition receptor signaling pathway [GO:0002753]; I-kappaB kinase/NF-kappaB signaling [GO:0007249]; innate immune response [GO:0045087]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; protein homooligomerization [GO:0051260] |
12566447_TIFA is likely to mediate IRAK-1/TRAF6 interaction upon IL-1 stimulation 16643855_We propose that ZCCHC11 is a unique TLR signal regulator, which interacts with TIFA after LPS treatment and suppresses the TRAF6-dependent activation of NF-kappaB. 23722163_The increase in TIFA level appears to be a feed-forward mechanism involved in TLR4/MyD88-dependent signaling, leading to NF-kappaB activation and HMGB1 release |
ENSG00000145365 |
TIFA |
92.958996 |
2.0491012443 |
1.034991 |
0.33608660 |
9.393904 |
0.00217708150541740539166135626203413266921415925025939941406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0091631128738651340670529066301241982728242874145507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
118.180609647927 |
27.7807478107829 |
57.6743633210014 |
10.2309908108541 |
| ENSMUSG00000046694 |
100342 |
Tent5b |
protein_coding |
Q8C152 |
FUNCTION: Catalyzes the transfer of one adenosine molecule from an ATP to an mRNA poly(A) tail bearing a 3'-OH terminal group in an ATP hydrolysis-dependent manner. May be involved in maintaining the translation efficiency of at least some genes through preventing degradation of their mRNAs. Prefers RNA molecules that are adenosine-rich close to 3'-end. In addition, may inhibit cell proliferation and cell cycle progression through ubiquitination of beta-catenin/CTNNB1. {ECO:0000250|UniProtKB:Q96A09}. |
Cytoplasm;Nucleotidyltransferase;Nucleus;Reference proteome;Transferase |
|
|
mmu:100342; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; polynucleotide adenylyltransferase activity [GO:0004652]; RNA adenylyltransferase activity [GO:1990817]; mRNA stabilization [GO:0048255]; negative regulation of apoptotic process [GO:0043066]; negative regulation of cell cycle [GO:0045786]; negative regulation of cell population proliferation [GO:0008285]; positive regulation of translation [GO:0045727] |
Human_homologues 30532005_these data show that FAM46B inhibits cell proliferation and cell cycle progression in PC through ubiquitination of beta-catenin 32009146_FAM46B is uniquely and highly expressed in human pre-implantation embryos and pluripotent stem cells, but sharply down-regulated following differentiation. |
ENSG00000158246 |
TENT5B |
67.885019 |
3.8313715147 |
1.937861 |
0.33878037 |
32.446792 |
0.00000001225010011151072709316842476853434695804878629132872447371482849121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001464204479923950146773444728337532083628502732608467340469360351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
110.545828057768 |
26.1981183956422 |
28.8528083568042 |
5.42562807726413 |
| ENSMUSG00000046818 |
73284 |
Ddit4l |
protein_coding |
Q8VHZ5 |
FUNCTION: Inhibits cell growth by regulating the TOR signaling pathway upstream of the TSC1-TSC2 complex and downstream of AKT1. {ECO:0000250}. |
Cytoplasm;Reference proteome |
|
|
mmu:73284; |
cytoplasm [GO:0005737]; negative regulation of signal transduction [GO:0009968] |
15607738_SMHS1 was found to be expressed in skeletal muscle, and comparisons of its expression in atrophied versus hypertrophied muscles and in oxidative versus glycolytic muscles suggested that SMHS1 contributes to muscle energy metabolism phenotypes. 15632201_RTP801 and RTP801L work downstream of AKT and upstream of TSC2 to inhibit mTOR functions 19129461_REDD2 is enriched in skeletal muscle and inhibits mTOR signaling in response to leucine and stretch. 24412152_IRF-1 physically interacts with REDD2 in the large cytoplasmic protein complex 25257176_Axonal injury-induced upregulation of the stress-responsive REDD2 leads to mTOR inhibition triggering early dendritic arbor retraction, neuronal dysfunction and subsequent death of adult retinal neurons. 28246202_Data showed that DDiT4L may be an important transducer of pathological stress to autophagy through mTOR signaling in the heart. |
ENSG00000145358 |
DDIT4L |
236.925501 |
0.4639178851 |
-1.108059 |
0.23485265 |
22.062837 |
0.00000263869044601703690543768356102649619288058602251112461090087890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000217727697040799094503529115263162907467631157487630844116210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
141.339005589428 |
23.1099697249677 |
304.663842752497 |
36.6542110605513 |
| ENSMUSG00000047033 |
93886 |
Pcdhb15 |
protein_coding |
Q91Y04 |
Human_homologues FUNCTION: Potential calcium-dependent cell-adhesion protein. May be involved in the establishment and maintenance of specific neuronal connections in the brain. |
Calcium;Cell adhesion;Membrane;Proteomics identification;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:93886; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell adhesion [GO:0007155]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
|
ENSG00000113212 |
PCDHB7 |
140.537942 |
2.0075837367 |
1.005460 |
0.21874398 |
21.005377 |
0.00000457996212046534296552470141850044171860645292326807975769042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000360713459249886804242231364003856697308947332203388214111328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
188.711417697731 |
28.7225163161212 |
93.9992759672194 |
11.0479582808927 |
| ENSMUSG00000047139 |
12484 |
Cd24a |
protein_coding |
P24807 |
FUNCTION: May have a pivotal role in cell differentiation of different cell types. May have a specific role in early thymocyte development. Signaling could be triggered by the binding of a lectin-like ligand to the CD24 carbohydrates, and transduced by the release of second messengers derived from the GPI-anchor. Modulates B-cell activation responses (By similarity). In association with SIGLEC10 may be involved in the selective suppression of the immune response to danger-associated molecular patterns (DAMPs) such as HMGB1, HSP70 and HSP90 (PubMed:19264983). Plays a role in the control of autoimmunity (PubMed:20200274). {ECO:0000250|UniProtKB:P25063, ECO:0000269|PubMed:19264983, ECO:0000269|PubMed:20200274}. |
Cell membrane;Direct protein sequencing;Glycoprotein;GPI-anchor;Immunity;Innate immunity;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:12484; |
cell surface [GO:0009986]; ciliary membrane [GO:0060170]; external side of plasma membrane [GO:0009897]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; membrane raft [GO:0045121]; microvillus membrane [GO:0031528]; motile cilium [GO:0031514]; plasma membrane [GO:0005886]; presynapse [GO:0098793]; carbohydrate binding [GO:0030246]; Hsp70 protein binding [GO:0030544]; Hsp90 protein binding [GO:0051879]; protein kinase binding [GO:0019901]; protein tyrosine kinase activator activity [GO:0030296]; apoptotic mitochondrial changes [GO:0008637]; apoptotic nuclear changes [GO:0030262]; apoptotic signaling pathway [GO:0097190]; B cell apoptotic process [GO:0001783]; B cell proliferation [GO:0042100]; B cell receptor transport into membrane raft [GO:0032597]; calcium-mediated signaling [GO:0019722]; CD4-positive, alpha-beta T cell proliferation [GO:0035739]; cell activation [GO:0001775]; cell migration [GO:0016477]; cell surface receptor signaling pathway [GO:0007166]; chemokine receptor transport out of membrane raft [GO:0032600]; cholesterol homeostasis [GO:0042632]; glomerular parietal epithelial cell differentiation [GO:0072139]; heterophilic cell-cell adhesion via plasma membrane cell adhesion molecules [GO:0007157]; homotypic cell-cell adhesion [GO:0034109]; innate immune response [GO:0045087]; integrin activation [GO:0033622]; leukocyte cell-cell adhesion [GO:0007159]; leukocyte migration involved in inflammatory response [GO:0002523]; negative regulation of B cell proliferation [GO:0030889]; negative regulation of erythrocyte aggregation [GO:0034119]; negative regulation of erythrocyte clearance [GO:0034107]; negative regulation of inflammatory response to wounding [GO:0106015]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of monocyte chemotactic protein-1 production [GO:0071638]; negative regulation of neuroblast proliferation [GO:0007406]; negative regulation of neurogenesis [GO:0050768]; negative regulation of neuron differentiation [GO:0045665]; negative regulation of T cell homeostatic proliferation [GO:0046014]; negative regulation of transforming growth factor beta3 production [GO:0032913]; negative regulation of tumor necrosis factor production [GO:0032720]; neuroblast proliferation [GO:0007405]; neuromuscular synaptic transmission [GO:0007274]; neuron projection development [GO:0031175]; peptidyl-tyrosine phosphorylation [GO:0018108]; podocyte differentiation [GO:0072112]; positive regulation of activated T cell proliferation [GO:0042104]; positive regulation of alpha-beta T cell proliferation [GO:0046641]; positive regulation of B cell apoptotic process [GO:0002904]; positive regulation of calcium-mediated signaling [GO:0050850]; positive regulation of CD4-positive, alpha-beta T cell proliferation [GO:2000563]; positive regulation of cell adhesion mediated by integrin [GO:0033630]; positive regulation of cell-cell adhesion [GO:0022409]; positive regulation of cell-cell adhesion mediated by integrin [GO:0033634]; positive regulation of cytosolic calcium ion concentration [GO:0007204]; positive regulation of inflammatory response to antigenic stimulus [GO:0002863]; positive regulation of inflammatory response to wounding [GO:0106016]; positive regulation of integrin activation [GO:0033625]; positive regulation of MAP kinase activity [GO:0043406]; positive regulation of nephron tubule epithelial cell differentiation [GO:2000768]; positive regulation of neuron projection development [GO:0010976]; positive regulation of peptidyl-tyrosine phosphorylation [GO:0050731]; positive regulation of T cell homeostatic proliferation [GO:0042103]; positive regulation of T cell mediated immune response to tumor cell [GO:0002842]; pre-B cell differentiation [GO:0002329]; regulation of B cell differentiation [GO:0045577]; regulation of cell-cell adhesion [GO:0022407]; regulation of cell-cell adhesion mediated by integrin [GO:0033632]; regulation of cytokine-mediated signaling pathway [GO:0001959]; regulation of MAPK cascade [GO:0043408]; regulation of phosphorylation [GO:0042325]; respiratory burst [GO:0045730]; response to estrogen [GO:0043627]; response to hypoxia [GO:0001666]; response to molecule of bacterial origin [GO:0002237]; synaptic vesicle endocytosis [GO:0048488]; T cell costimulation [GO:0031295]; T cell homeostatic proliferation [GO:0001777] |
12829373_N-glycans of the mouse glycoprotein HSA and its human analogue CD24 from lymphoblastoma, neuroblastoma and astrocytoma cell lines as well as from mouse brain homogenate were analysed and compared to each other 14657362_CD24 has a role in risk and progression of multiple sclerosis 15477346_CD24 expressed on T cells is essential for homeostatic proliferation in lymphopenic host. 15737737_Mouse CD24 acts cell nonautonomously to regulate transit-amplifying cells proliferation and/or differentiation in adult SVZ. 16390867_These results demonstrate a novel role for CD24 as a regulator of CXCR4 function that could be relevant for breast cancer growth and metastasis. 16606832_CD24 is required for normal presynaptic maturation and function. 17522896_These results and previous published data indicate that mCD24 contributes to the regulation of the production of differentiated cells by controlling the proliferation/differentiation balance between transit-amplifying and committed differentiated cells. 17700640_CD24 is not essential for exosome formation or release but may be a convenient exosome marker. Our studies suggest that exosomal secretion from the embryonic kidney could play a biological role at the fetal-maternal interphase. 18022325_elongation, collateralization, fasciculation and path finding of corticospinal axons in mice proceed normally in the absence of CD24. 18185529_CD24a expression levels discriminate Langerhans cells from dermal dendritic cells in murine skin and lymph nodes 18632604_CD44 but not CD24 may be involved in the breast cancer pathway and is a target for oncolytic virotherapy 19264983_results reveal that the CD24-Siglec G pathway protects the host against a lethal response to pathological cell death and discriminates danger- versus pathogen-associated molecular patterns 19284289_interaction of CD24 with the cell adhesion molecule L1 is mediated by O-linked glycans carrying alpha2,3-linked sialic acid 19725119_Dynamic changes in surface expression of the identified antigens CD15, CD24, and CD29 in combination can be exploited for the experimental separation of key neural cell populations. 19786366_mice with a targeted mutation of CD24 are resistant to induction of experimental autoimmune encephalomyelitis 20185687_CD24 is expressed in the Sox9(EGFPlo) population and marks intestinal epithelial stem cells that form organoids in culture. 20352469_antigen CD24 is not a major regulator of mammary stem/progenitor cell function, but rather plays a role in governing branching morphogenesis. 21037089_CD24 is expressed in an NFAT5-dependent manner during the mitogenic expansion of mature T lymphocytes under hypertonic conditions and is required to sustain T cell expansion under osmostress. 21183658_Report sorting mouse jejunal epithelial cells with CD24 yields a population with characteristics of intestinal stem cells. 21361791_The isolation and characterization of a rare, primary, nonhematopoietic, CD24+ progenitor cell population from normal, untreated mouse liver, is described. 22213356_CD24 expression on thymic antigen-presenting cells (mTECs, DCs) down-regulates autoantigen-mediated clonal deletion of autoreactive thymocytes. 22698401_Results suggest that LKB1 inactivation in the context of RAS activation facilitates metastasis by inducing an SFK-dependent expansion of a prometastatic, CD24(+) tumor subpopulation. 22723265_CD24-based sorting in wild-type mice isolates a colonic epithelial fraction highly enriched in actively cycling and quiescent Lgr5 positive putative colonic epithelial stem cells. 22899822_CD24 is expressed in gastric parietal cells, where it modulates gastric responses to H. felis and gamma-radiation. CD24 also regulates susceptibility to apoptosis in the distal murine gastrointestinal tract. 23012401_CD24 has a role in urothelial tumorigenesis and metastasis in male mice and is androgen regulated in mice and humans 23881416_CD24 expression negatively regulates the NF-kappaB pathway following experimental traumatic brain injury. 24500912_CD24 is shown in vitro and in vivo as being an important oncogene in the gut, and one that plays a critical role in the initiation and progression of carcinogenesis. 24631155_CD24 was shown to regulate CD8(+) T cell activation through HMGB1-mediated engagement of T cell RAGE. 24695850_Siglec-G-CD24 axis, controls the severity of GVHD and suggest that enhancing this interaction may represent a novel strategy for mitigating GVHD. 24808359_the deletion of CD24 in an HSP-driven model of autoimmunity led to the unexpected development of regulatory T cell and MDSC populations that augmented immune tolerance. 24956310_CD24 in non-immune cells might be crucialfor the guidance and recruitment of leukocytes. 25641732_CD24 controls breast cancer radiation response. Loss of CD24 expression leads to radiation resistance. 25969154_Findings indicate that CD24(+) antigen cells play a role in tumor migration and metastasis and support Janus kinase 2 protein (JAK2) as a therapeutic target in ovarian cancer. 26076835_CD24 is a conserved marker for tracking divergent states in both reprogramming and standard pluripotent culture. 26440795_Delayed wound-healing in the absence of HSA/CD24 suggests that CD24 plays an important role in this process 26536476_Loss of CD24 in Mice Leads to Metabolic Dysfunctions and a Reduction in White Adipocyte Tissue. 26978528_mice negative or positive for CD24 did not differ in terms of tumor initiation and burden in 3 mammary and prostate tumor models tested, except for Apc1572T/+ mice, in which lack of CD24 reduced the mammary tumor burden slightly but significantly. 27179633_CD24 cell surface expression may serve as a valuable biomarker in order to identify mammary tumors that will positively respond to targeted IGF1R therapies. 27203385_CD24 has a role in determining the epithelial phenotype of pancreatic cancer 28065940_this study shows that CD24 aggravates acute liver injury in autoimmune hepatitis by promoting IFN-gamma production by CD4(+) T cells 28182766_While no obvious role was found for CD24 in the normal development and maintenance of the dopaminergic nigrostriatal system in mice, it may have a role in mediating the neuroprotective aspects of GDNF in this system. 29620414_CD24 and CD49f expressions of E14.5 mouse mammary anlagen cells define putative distribution of earlier embryonic mammary stem cell activities. 30231671_CD24 regulates lipid accumulation but not glucose uptake in 3T3-L1 adipocytes. CD24 regulates gene expression in differentiating adipocytes in vitro in a depot specific manner. 30564864_Cancer-associated fibroblasts promoted self-renewal, chemotherapy resistance, metastasis, and tumorigenicity of CD24+ hepatocellular carcinoma cells. |
ENSG00000272398 |
CD24 |
26.190634 |
0.0486227730 |
-4.362224 |
0.98518277 |
17.040518 |
0.00003659063508592540474740478773085783359420020133256912231445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002424732225251914369702072038492701722134370356798171997070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.2498138906032 |
1.65856546285992 |
46.2707853252671 |
21.2655512440775 |
| ENSMUSG00000047250 |
19224 |
Ptgs1 |
protein_coding |
P22437 |
FUNCTION: Dual cyclooxygenase and peroxidase in the biosynthesis pathway of prostanoids, a class of C20 oxylipins mainly derived from arachidonate, with a particular role in the inflammatory response. The cyclooxygenase activity oxygenates arachidonate (AA, C20:4(n-6)) to the hydroperoxy endoperoxide prostaglandin G2 (PGG2), and the peroxidase activity reduces PGG2 to the hydroxy endoperoxide PGH2, the precursor of all 2-series prostaglandins and thromboxanes. This complex transformation is initiated by abstraction of hydrogen at carbon 13 (with S-stereochemistry), followed by insertion of molecular O2 to form the endoperoxide bridge between carbon 9 and 11 that defines prostaglandins. The insertion of a second molecule of O2 (bis-oxygenase activity) yields a hydroperoxy group in PGG2 that is then reduced to PGH2 by two electrons. Involved in the constitutive production of prostanoids in particular in the stomach and platelets. In gastric epithelial cells, it is a key step in the generation of prostaglandins, such as prostaglandin E2 (PGE2), which plays an important role in cytoprotection. In platelets, it is involved in the generation of thromboxane A2 (TXA2), which promotes platelet activation and aggregation, vasoconstriction and proliferation of vascular smooth muscle cells. {ECO:0000250|UniProtKB:P23219}. |
Dioxygenase;Disulfide bond;EGF-like domain;Endoplasmic reticulum;Fatty acid biosynthesis;Fatty acid metabolism;Glycoprotein;Heme;Iron;Lipid biosynthesis;Lipid metabolism;Membrane;Metal-binding;Microsome;Oxidoreductase;Peroxidase;Prostaglandin biosynthesis;Prostaglandin metabolism;Reference proteome;Signal |
PATHWAY: Lipid metabolism; prostaglandin biosynthesis. {ECO:0000250|UniProtKB:P23219}. |
This is one of two genes encoding similar enzymes that catalyze the conversion of arachinodate to prostaglandin. The encoded protein regulates angiogenesis in endothelial cells, and is inhibited by nonsteroidal anti-inflammatory drugs such as aspirin. Based on its ability to function as both a cyclooxygenase and as a peroxidase, the encoded protein has been identified as a moonlighting protein. [provided by RefSeq, Jan 2014]. |
mmu:19224; |
cytoplasm [GO:0005737]; endoplasmic reticulum membrane [GO:0005789]; Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; neuron projection [GO:0043005]; nuclear envelope [GO:0005635]; photoreceptor outer segment [GO:0001750]; heme binding [GO:0020037]; metal ion binding [GO:0046872]; oxidoreductase activity, acting on single donors with incorporation of molecular oxygen, incorporation of two atoms of oxygen [GO:0016702]; peroxidase activity [GO:0004601]; prostaglandin-endoperoxide synthase activity [GO:0004666]; cyclooxygenase pathway [GO:0019371]; keratinocyte differentiation [GO:0030216]; learning [GO:0007612]; maintenance of blood-brain barrier [GO:0035633]; memory [GO:0007613]; negative regulation of epinephrine secretion [GO:0032811]; negative regulation of norepinephrine secretion [GO:0010700]; positive regulation of smooth muscle contraction [GO:0045987]; positive regulation of vasoconstriction [GO:0045907]; prostaglandin biosynthetic process [GO:0001516]; prostaglandin metabolic process [GO:0006693]; regulation of blood pressure [GO:0008217]; regulation of cell population proliferation [GO:0042127]; response to oxidative stress [GO:0006979]; sensory perception of pain [GO:0019233] |
8121489_Prostaglandin G/H synthase is a moonlighting protein that functions both as a peroxidase as well as a cyclooxygenase. 11936618_The tissue-specific, compensatory expression of cyclooxygenase-1 and -2 in transgenic mice. 12093889_Opposite effects of cyclooxygenase-1 and -2 activity on the pressor response to angiotensin II. 12118094_constitutive and inducible enzymes affect dermal wound healing 12183660_after IL-1beta administration, COX1 is the major enzyme involved in the hypophagia 12184998_Prostaglandins that increase renin production in response to ACE inhibition are not derived from cyclooxygenase-1. 12213634_Results fail to confirm the harmful effect of losing COX-1 activity due to gene knockout in a permanent endovascular middle cerebral artery occlusion stroke model. 12411530_Pre-epithelial alkaline layer is regulated by endogenous COX activity. 12576525_Data show that both cyclooxygenase-1 and -2 isoforms are involved in the negative modulation of adipocyte differentiation. 12657565_Site-specific expression in mouse kidney and relationship to prostaglandin metabolism. 12664644_Knocking out this enzyme affects body temperature regulation. 12713596_cyclooxygenase-1-coupled prostaglandin E2/D2 biosynthesis has a central role in skin repair 12835322_mPGES-2 is a unique PGES that can be coupled with both COXs and may play a role in the production of the PGE2 involved in both tissue homeostasis and disease. 12958020_PGE2 derived from either COX-1 or -2 is involved in regulation of gastric mucosal inflammation and contributes to maintenance of mucosal integrity during H. pylori infection via inhibition of TNF-alpha expression 14625089_Like its counterpart COX-1, COX-3 does not generally appear to be induced by acute inflammatory stimulation of the CNS. 14638539_COX-1 plays an important role in the early stages of lesion development in the apoE-/- knockout model of atherosclerosis. 14701858_COX-1 can replace specific functions of COX-2 in ovulation, fertilization, and implantation 15181007_COX-1 may regulate tumor necrosis factor-alpha secretion and play a role in the regulation of the early response to endotoxemia 15263079_Acetaminophen reduces basal body temperature below normal in mice most likely by inhibiting COX-3. 15358864_CUGBP2 expression is higher in COX-1 deficient mice than wild-type controls at basal conditions, which is further increased after irradiation. 15863457_COX-1- or COX-2-derived prostanoids do not modulate responses to angiotensin II or other vasoactive agents 15883230_COX-1 mediates a suppressed sympathetic nervous activity and enhanced NO, which may contribute to renal vasodilatation and a reduced MAP while asleep or under anesthesia. COX-1 contributes to the normal nocturnal BP dipping phenomenon. 16189289_we conclude that the two COX isoforms in the renal medulla play a similar role in the stabilization of arterial blood pressure during salt loading. 16204198_prostaglandin E2 biosynthesis results from the coordinated activity of COX-1 and microsomal prostaglandin E2 synthase-1 16272346_COX-1 deficiency is detrimental whereas COX-2 deficiency is beneficial to the host during influenza viral infection. 16373427_In contrast, COX-1 has no significant role in healing when COX-2 is unimpaired but becomes important when COX-2 is impaired. 16380543_COX-1 deficiency in bone marrow-derived cells worsens early atherosclerosis in apoE(-/-) and LDLR(-/-) mice despite virtual elimination of platelet TX production 16493486_Of the aspirin non-responders detected by arachidonic acid, 3 of 5 (60%) carried the rare G allele for the -A842G polymorphism of COX-1 16518767_role in Helicobacter pylori induced gastric inflammation; COX-1 and COX-2 deficiency enhances H. pylori-induced gastritis, probably via TNF-alpha expression 16554545_Our results demonstrate that mPGES-1, which is strongly upregulated during inflammation in central structures involved in feeding control, is essential for immune anorexic behavior and thus may constitute a potential therapeutic target. 16556900_These data show that the gene products of both COX genes contribute to pain and local inflammation in inflammatory arthritis. There are sex differences in some of these effects, and this suggests that the effects of COX inhibitors may be sex dependent. 16627484_mPGES-1 mRNA levels in cardiomyocytes are augmented by stabilization of mPGES-1 mRNA, and JNK1 or JNK2 can participate in the regulation of mPGES-1 protein synthesis in these cells 16675300_results provided the evidence for the distinct gene expression of isoformic enzymes in the COX pathway leading to the synthesis of PGE2 and PGF2alpha and the specific action of these prostanoids at different cycle stages of adipocytes. 16787386_The substrate specificities of COX-1 and COX-2 in the synthesis of prostaglandins in peritoneal macrophages of mice with gene deletions at both loci are reported. 16815697_cutaneous PGD2 could be mainly produced by COX-1, and PGE2 and PGI2 could be produced by COX-1 and COX-2, respectively 16842596_COX-1-coupled skin PGD(2) biosynthesis plays a physiological role in inhibiting regulation of pruritus in NC mice with atopic dermatitis 16857891_Similarly, targeted deletion of COX-2 alone, or COX-1/COX-2 together, impaired postnatal ductus closure. Nitric oxide inhibition did not prevent the delay in ductus closure. 16955389_cyclooxygenase-1b (putative COX-3) cloning in mouse 17038629_This suggests that COX-1-derived PGE(2) may play a key role in the early phase of intestinal tumorigenesis in response to DNA damage and suggests that COX-1 may be a potential therapeutic target in this model of colon cancer. 17110378_Insertion of COX-1 under the regulatory sequences that drive COX-2 expression indicated that COX-1 can substitute for some COX-2 actions and rescue only some of the consequences of gene disruption 17145980_cyclooxygenase (COX)-1 participates in the angiotensin II-induced endothelial dysfunction in murine mesenteric small arteries 17266762_COX-1 and COX-2 differentially modulate brain gene expression. 17364940_Although, numerous free radical generating systems may contribute to the postanoxic microglial cell damage, the cyclooxygenase-mediated oxidant production seems to be of major importance. 17454003_Study explores Ptgs1 and Mrc1 genes as two new positional candidate genes for allergen-induced airway hyperresponsiveness through comparative sequence analysis and mRNA expression studies of mouse strains with genetically mediated airway responsiveness. 17505022_Regulation of the microsomal Ptgs1 in polarized mononuclear phagocytes and its constitutive expression in neutrophils were studied. 17641673_compromised vasomotor function in Marfan thoracic aortae is associated with an imbalanced synthesis of thromboxane A(2) and prostacyclin resulting from the differential protein expression of COX-1 and COX-2 17641732_Study suggests that in hypertensive mice, there are (a) significant microanatomic variations in the pulmonary, renal, and cardiac distribution and cellular localization of COX-1 and no differences in expression between genders. 17674023_prostaglandin F synthase and prostaglandin F(2alpha )receptor are involved in the control of testosterone release from Leydig cells 17916350_Lemnalol prevented iNOS/COX-2 elevation in LPS stimulated macrophages. 17921329_Demonstrate for the first time the involvement of COX-1 pathway in A(3)AR-mediated contraction via endothelium. 17933963_Platelet- and endothelial cell-associated COX-1 promote microvascular inflammation and thrombogenesis during hypercholesterolemia, yet nitric oxide-releasing aspirin directly inhibits platelets independent of COX-1. 17959725_Constitutive loss of Cox-1 leaves the gastric mucosa more prone to damage and slowed repair of microlesions. 17982098_peritoneal macrophages treated with HK-BCG in vivo express constitutive COX-1 and inducible COX-2 that are catalytically inactive, are localized subcellularly in the cytoplasm, and are not associated with the nuclear envelope (NE). 18162486_transcriptional down-regulation of cytokines and other inflammatory markers in COX-1(-/-) mice was mediated by a reduced activation of NF-kappaB and signal transducer and activator of transcription 3. 18292449_Indicate the synergistic nature of the interaction between NOS and COX inhibitors in formalin-induced nociceptive behavior in mice, and provide an alternative approach for controlling pain. 18419601_Consequences of altered eicosanoid patterns for nociceptive processing in Ptgs1-deficient mice are reported. 18514659_COX-1 contributes to the enhanced formation of both PGI(2) and TXA(2) in atherosclerosis, and to the development of the disease 18523303_Prostaglandin-endoperoxide synthase-1 genetic deletion results in a marked reduction in levels of type II collagen-specific IgG antibody production, correlating with reduction in collagen-induced arthritis incidence and severity. 18584335_Conclude that GSK-3 inhibitors block corticosterone-induced COX-1 gene expression via a mechanism beyond glucocorticoid receptors and Sp3 transcription factor. 18753249_despite the presence of COX-2, COX-1 is functionally predominant in the airways and explains clinical observations relating to drug specificity in patients with aspirin-sensitive asthma. 18772366_Either COX-1 or COX-2 can mediate elevated PGI(2) biosynthesis and the development of experimental prehepatic PHT. 19056763_COX-1 vs. COX-2 as a determinant of basal tone in the internal anal sphincter. 19114118_The selective up-regulation of mPGES-1 in COX-2 K/O cells suggests that mPGES-1 may be metabolically coupled with COX-1 for PGE(2) formation. 19523994_COX-1 as well as COX-2 may be involved in epileptogenesis, and that certain COX inhibitors can potentially prevent the occurrence of seizures. 19658194_our results demonstrate that endothelial cells control neuronal injury via mPGES-1-derived PGE2 19711010_COX-1 and COX-2 seem to play an important role in renal ischemia and reperfusion injury, involving the secretion of pro-inflammatory cytokines, activation of neutrophils, and ROS production. 19719848_The deficiency of COX-1 resulted in no detectable effect on cortical tissue loss or cognitive function following traumatic brain injury. 19767186_The absence of COX-1 or COX-2 did not appear to effect ocular development in KO mice. 19843949_Infection of mice lacking COX-1, but not COX-2, activity results in a defect in antibody (Ig) class-switching and a lack of Borrelia-specific IgG production. 19844242_In the hippocampus and cortex of LPS-treated mice, matrix metalloproteinase (MMP)-3 activity was significantly decreased in COX-1(-/-) mice 19885646_expressed in peritoneal macrophages and mast cells during acute peritonitis 20034541_These findings clearly demonstrate that the immediate corticosterone release seen after immune challenge with lipopolysaccharide is dependent on Cox-1 derived prostanoids 20065147_These data indicate that genetic background significantly modifies the blood pressure response to mPGES1 deficiency. 20157512_These results indicate that inhibition of COX-1 activity may be valid therapeutic strategy to reduce brain inflammatory response and neurodegeneration. 20304815_A novel mechanism was identified whereby chronic increases in oxidative stress, produced by mitochondrial superoxide dismutase deficiency, impair vascular function via a hydrogen peroxide-dependent, COX1-dependent, endothelium-derived contracting factor 20412387_A unique role for COX-1 in mediating chronic neuroinflammatory effects through PGE(2) production is suggested. 20585313_ErbB4-stimulated COX-2 induction is part of the molecular mechanisms responsible for ErbB4-induced cell survival 21357261_Cox-1-expressing cellular lineage is necessary to promote synovitis in vivo; Cox-1 activity is attributable to a bone marrow-derived lineage (hematopoietic) and not to a radioresistant mesenchymal (tissue) lineage. 21680698_COX-1 may play a crucial role in the pathology of neuroinflammation 21694498_Cyclooxygenase-1 gene deltion resulted in an incresed proliferation and differentiation of hippocampal progenitor cells in the adult mouse brain druing neuroinflammation. 21858200_Data suggest that inhibition of COX-1, COX-2, or both enzymes alone seems inadequate to explain the gastric toxicity of piroxicam in mast cell-deficient mice. 22001217_Novel selective COX-1 inhibitors suppress neuroinflammatory mediators in LPS-stimulated N13 microglial cells. 22022466_These findings suggest that purinergic signaling in microglia triggered by neuronal ATP modulates excitotoxic cortical lesion by regulating COX-1 dependent prostanoid production. 22080487_In the abdominal aorta, ACh evoked endothelium-dependent production of 6-keto-PGF(1alpha), which was abolished by COX-1(-/-), but not by COX-2(-/-). 22219191_Astrocytes respond to lipopolysaccharide by a COX-2-dependent production of prostanoids, mainly vasoactive PGE(2), and suggest that the coordinated down-regulation of COX-1 facilitates PGE(2) production after TLR-4 activation. 22406532_Niacin evoked platelet COX-1-derived PGD biosynthesis. 23178857_KLF10 induces COX-1 protein expression and mRNA expression in endothelial cells. 23305935_These observations suggest that COX-enzymes, most likely COX-1, are involved in cancer-elicited anorexia and weight loss 23742950_COX1/2-derived prostanoids appear to play an important role in the TLR response, with elevated cytokine levels following COX1/2 inhibition or genetic deletion. 23817787_role of COX-1 during maturation of adipocyte 23874970_The production of prostanoids found in the systemic circulation is driven overwhelmingly by COX-1 and not COX-2. 23966321_Data suggest deletion of Cox1 delays ovarian follicle development prior to reproductive maturation of mutant mouse strain (Wv); inhibition of Cox1 (but not Cox2) may prolong ovarian follicle lifespan and delay the onset of premature ovarian failure. 23987235_Hematopoietic COX-1 deletion results in impairments in metabolic homeostasis which may be partly due to increased adipose tissue inflammation and dysregulated adipokine profile. 24014678_Propose a mechanism whereby ANG II increases COX-1-derived PGE2 through the AT1R/PLA2 pathway, which promotes ROS production by EP1R/Nox2 signaling in the subfornical organ. 24878773_In arteries from non-insulin-dependent diabetic mice, COX-1 remains a major contributor to the endothelial PGI2 synthesis that evokes vasoconstrictor activity under the pathological condition. 24944008_specific inhibition of mmu-miR-100-5p significantly enhanced expression of Il6, Ptgs1/2 and Tlr4 mRNA 25432964_Expression of COX-1 is essential for the protection of liver against chemical-induced hepatotoxicity and required for hepatic homeostatic maintenance. 25757568_Data suggest that multitarget FAAH/Cox blockade may provide a transformative approach to inflammatory bowel disease (IBD) and other pathologies in which fatty acid amide hydrolase/cyclooxygenases (FAAH, Cox-1, and Cox-2) are overactive. 26017977_COX-1 inhibitor SC-560 has a protective effect on the thromboxane A2-mediated decrease in renal function in response to endotoxin. 26444418_role of cyclooxygenase-1 and -2 in endothelium-dependent contraction of atherosclerotic mouse abdominal aortas 27059285_Specific inhibition of PGE2 synthesis by targeting mPGES-1 may weaken host defense against bacterial infections. 27060751_Dimethyl ester of bilirubin exhibits anti-inflammatory activity through inhibition of secretory phospholipase A2, lipoxygenase and cyclooxygenase. 27386631_Suggest the expression of COX-1 and COX-2 in the urothelium protects bladder damage from radiation. 28684424_Data suggest that Il4 (usually released from helper T-cells) induces Cox1 in macrophages at post-transcriptional level; activation of Ampk (catalytic subunit Prkaa1) by metformin blocks Il4-dependent induction of Cox1 and blocks macrophage polarization/activation. (Il4 = interleukin-4; Cox1 = cyclooxygenase 1; Ampk = AMP-activated protein kinase) 28954232_IFNgamma upregulated microsomal prostaglandin E synthase-1 (mPGES-1) alongside cyclo-oxygenase (COX-1) within macrophage populations, resulting in sustained prostaglandin (PG)E2 biosynthesis. 29029054_Data (including data from studies in knockout mice) suggest that delayed parturition in Cox-1 knockout mice is result of impaired luteolysis and cervical dilation, despite the presence of strong uterine contractions. 29180445_Manipulation of COXs 1 & 2 revealed isoform-specific compensatory functions and variable degrees of interchangeability for PG biosynthesis in cells/tissues 29676940_the highly expressed knockin COX-2 enzyme barely makes any PGs or thromboxane in neonatal P8 or adult mice, demonstrating that prostanoid biosynthesis requires native COX-1 and cannot be rescued by the knockin COX-2. In summary, the 2 COX isoforms can preferentially compensate for some renal functions, which appears to be independent of the PG-synthetic capacity. 30108257_Transgenic mice study revealed that knock-in COX-1 failed to substitute for the loss of COX-2 in COX-1>COX-2 mice during implantation, indicating that COX-1 may be replaced by COX-2, but not vice versa. A panel of prostaglandins detected in uterus and ovary demonstrates that prostaglandin biosynthesis preferentially depends on native COX-1, but not COX-2. 30760327_PTGS1-modified cells were incubated with synthograft and transplanted into the dorsal space of mice. .. HE staining showed that the PTGS1sh group generated much more bone-like tissue compared with the NC group , and quantitative analysis verified that silencing of PTGS1 greatly promoted bone-like tissue formation in vivo 30811756_Expression of COX-1 and production of thromboxane A2 in platelets of STZ-induced diabetic mice were elevated. 31248980_Platelet-Specific Deletion of Cyclooxygenase-1 Ameliorates Dextran Sulfate Sodium-Induced Colitis in Mice 32259636_Exposure to growth hormone is associated with hepatic up-regulation of cPLA2alpha and COX. 33741600_Endothelial cyclooxygenase-1 paradoxically drives local vasoconstriction and atherogenesis despite underpinning prostacyclin generation. 33806238_Dynamics of Cyclooxygenase-1 Positive Microglia/Macrophage in the Retina of Pathological Model Mice as a Biomarker of the Retinal Inflammatory Diseases. 34348138_Astrocytes regulate ultra-slow arteriole oscillations via stretch-mediated TRPV4-COX-1 feedback. 36241001_The specific deletion of cyclooxygenase-1 in megakaryocytes/platelets reduces intestinal polyposis in ApcMin/+ mice. |
ENSG00000095303 |
PTGS1 |
805.660514 |
2.5134001061 |
1.329640 |
0.12995663 |
102.604340 |
0.00000000000000000000000409235767941571473255934250492634783642950831233944850260678095191929637408634334860835224390029907226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000002134753156604800226652904414502470170188233721164218189750684875893860237283661263063549995422363281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1178.45122571301 |
106.297407928483 |
468.867341443733 |
32.3417301597609 |
| ENSMUSG00000047330 |
57814 |
Kcne4 |
protein_coding |
Q9WTW3 |
FUNCTION: Ancillary protein that assembles as a beta subunit with a voltage-gated potassium channel complex of pore-forming alpha subunits. Modulates the gating kinetics and enhances stability of the channel complex. Associated with KCNQ1/KVLTQ1 inhibits potassium current (By similarity). {ECO:0000250}. |
Glycoprotein;Ion channel;Ion transport;Membrane;Potassium;Potassium channel;Potassium transport;Reference proteome;Transmembrane;Transmembrane helix;Transport;Voltage-gated channel |
|
|
mmu:57814; |
apical plasma membrane [GO:0016324]; voltage-gated potassium channel complex [GO:0008076]; potassium channel activity [GO:0005267]; potassium channel regulator activity [GO:0015459]; transmembrane transporter binding [GO:0044325]; voltage-gated potassium channel activity [GO:0005249]; membrane repolarization during action potential [GO:0086011]; membrane repolarization during ventricular cardiac muscle cell action potential [GO:0098915]; negative regulation of delayed rectifier potassium channel activity [GO:1902260]; potassium ion export across plasma membrane [GO:0097623]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of ventricular cardiac muscle cell membrane repolarization [GO:0060307]; ventricular cardiac muscle cell action potential [GO:0086005] |
12944270_KCNE4 beta-subunit has a drastic inhibitory effect on currents generated by Kv1.1 and Kv1.3 potassium channels 15707997_Based upon previous studies and the present results, it is concluded that both hKCNE4 and mKCNE4 have a drastic inhibitory impact on both hKCNQ1 and mKCNQ1 currents. 19773357_KCNE4 (potassium voltage-gated channel subfamily E member 4), but not KCNE2, functions as an inhibitory Kv1.3 partner in leukocytes. 26399785_Kcne4 expression and function are age-, sex- dependent and regulated by 5alpha-dihydrotestosterone. 27710966_Suggest role for KCNE4 in regulation of Kv7 channel activity to modulate vascular tone, and provide the first known molecular mechanism for sex-specificity of this modulation. 27802162_the tertiary structure of the C-terminal domain of Kv1.3 is necessary and sufficient for Kv1.3- KCNE4 interaction. 29844497_this study show that Kcne4 deletion sex-specifically predisposes to cardiac arrhythmia via testosterone-dependent impairment of RISK/SAFE pathway induction in aged mice 30758982_Kcne4 deletion sex dependently exacerbates hepatic ischemia-reperfusion injury, by RISK/SAFE pathway induction. 30969795_experimental data and the in silico structure suggest that the KCNE4 interaction hides a forward-trafficking motif within Kv1.3 in addition to adding a strong endoplasmic reticulum retention signature to the Kv1.3-KCNE4 complex. 34272451_KCNE4-dependent functional consequences of Kv1.3-related leukocyte physiology. 35915077_Induction of potassium channel regulator KCNE4 in a submandibular lymph node metastasis model. |
ENSG00000152049 |
KCNE4 |
286.371469 |
0.3090752909 |
-1.693970 |
0.16285903 |
106.979247 |
0.00000000000000000000000044985564450554593671990762926442853525258365816709804218709440844833208350550535215006675571203231811523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000245598608498511659169073063334900004303436687000144148851883490157488321159462429932318627834320068359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
133.29909240787 |
15.0794839349071 |
431.2835621332 |
33.9920343323776 |
| ENSMUSG00000047420 |
208164 |
Fam180a |
protein_coding |
Q8BR21 |
|
Reference proteome;Secreted;Signal |
|
|
mmu:208164; |
extracellular region [GO:0005576] |
|
ENSG00000189320 |
FAM180A |
1063.226954 |
3.2990455366 |
1.722049 |
0.10579379 |
257.594653 |
0.00000000000000000000000000000000000000000000000000000000057389638880206020148896878545724332552889688685225958673247419614908478552963798488742705426636404644136190573561222048204495303782004284612544911525766622162336716428399085998535156250000000000000000000000 |
0.0000000000000000000000000000000000000000000000000000001197478054004517918899505611445271641681338570243531837186085341501382967333581916040446920373747241178656919033655253877186292977331273906099751158649269200395792722702026367187500000000000000000000000000 |
Yes |
No |
1628.06904793651 |
119.494414208669 |
493.496991748158 |
28.1367751668852 |
| ENSMUSG00000047443 |
227358 |
Erfe |
protein_coding |
Q6PGN1 |
FUNCTION: Iron-regulatory hormone that acts as an erythroid regulator after hemorrhage: produced by erythroblasts following blood loss and mediates suppression of hepcidin (HAMP) expression in the liver, thereby promoting increased iron absorption and mobilization from stores (PubMed:24880340). Promotes lipid uptake into adipocytes and hepatocytes via transcriptional up-regulation of genes involved in fatty acid uptake (PubMed:22351773). {ECO:0000269|PubMed:22351773, ECO:0000269|PubMed:24880340}. |
Disulfide bond;Glycoprotein;Hormone;Reference proteome;Secreted;Signal |
|
|
mmu:227358; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; hormone activity [GO:0005179]; identical protein binding [GO:0042802]; cellular iron ion homeostasis [GO:0006879]; establishment of localization in cell [GO:0051649]; fatty acid transport [GO:0015908]; negative regulation of gluconeogenesis [GO:0045721]; positive regulation of fatty acid transport [GO:2000193]; positive regulation of glucose import [GO:0046326]; positive regulation of insulin receptor signaling pathway [GO:0046628]; regulation of fatty acid metabolic process [GO:0019217] |
24187137_myonectin is a novel regulator of cellular autophagy 25193872_Compared with wild-type mice, Fam132b-deficient mice exhibited a more severe anemia, had higher hepcidin levels and consequently lower serum iron concentration on days 14 and 21, and manifested impaired mobilization of iron from stores (liver and spleen). 26276665_Erythroferrone contributes to hepcidin suppression and iron overload in a mouse model of beta-thalassemia. 27067488_The here presented data unearthed EPO-dependent erythroferrone expression acts as an erythropoiesis-driven regulator of iron metabolism under phenylhydrazine-induced hemolytic anemia. 27658439_Erfe(-/-) mice failed to adequately suppress hepcidin expression after infection with Plasmodium berghei K173. Importantly, the sustained production of hepcidin allowed by erythroferrone ablation was associated with decreased parasitemia, providing further evidence that transient iron restriction could be beneficial in the treatment of malaria. 28187515_Erythroferrone and matriptase-2 independently regulate hepcidin expression. 29073189_The lack of effect of erythropoietin on hepcidin expression in mask mice can not be explained by changes in erythroferrone synthesis, as splenic erythroferrone content increased after erythropoietin administration in both C57BL/6 and mask mice. 29419424_serum ERFE levels acutely increase in response to EPO in the setting of normal or impaired kidney function. 30097509_ERFE specifically abrogated the induction of hepcidin by BMP5, BMP6, and BMP7 but had little or no effect on hepcidin induction by BMP2, BMP4, BMP9, or activin B. 30566056_Myonectin ameliorates acute myocardial ischemic injury by suppressing apoptosis and inflammation in the heart. 31002535_Female myonectin-deficient mice had larger gonadal fat pads. 31800957_ERFE bound directly to BMP2/6 heterodimers and prevented BMP2/6 binding to ALK3. 32602701_Protein Modifications Critical for Myonectin/Erythroferrone Secretion and Oligomer Assembly. 33407993_Myonectin inhibits adipogenesis in 3T3-L1 preadipocytes by regulating p38 MAPK pathway. 34614145_Erythroid overproduction of erythroferrone causes iron overload and developmental abnormalities in mice. 35286626_CTRP15 promotes macrophage cholesterol efflux and attenuates atherosclerosis by increasing the expression of ABCA1. |
ENSG00000178752 |
ERFE |
44.466192 |
2.7328537757 |
1.450408 |
0.41005264 |
12.376105 |
0.00043486346844711265720306858639787606080062687397003173828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0022123715268491717968191156984403278329409658908843994140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.7389182161091 |
19.2254259442489 |
24.4209620026551 |
5.45484981885294 |
| ENSMUSG00000047454 |
268566 |
Gphn |
protein_coding |
Q8BUV3 |
FUNCTION: Microtubule-associated protein involved in membrane protein-cytoskeleton interactions. It is thought to anchor the inhibitory glycine receptor (GLYR) to subsynaptic microtubules (By similarity). Acts as a major instructive molecule at inhibitory synapses, where it also clusters GABA type A receptors (PubMed:25025157). {ECO:0000250|UniProtKB:Q03555, ECO:0000269|PubMed:25025157}.; FUNCTION: Has also a catalytic activity and catalyzes two steps in the biosynthesis of the molybdenum cofactor. In the first step, molybdopterin is adenylated. Subsequently, molybdate is inserted into adenylated molybdopterin and AMP is released. {ECO:0000250|UniProtKB:Q03555}. |
ATP-binding;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;Lipoprotein;Magnesium;Membrane;Metal-binding;Molybdenum;Molybdenum cofactor biosynthesis;Multifunctional enzyme;Nucleotide-binding;Palmitate;Phosphoprotein;Postsynaptic cell membrane;Reference proteome;Synapse;Transferase |
PATHWAY: Cofactor biosynthesis; molybdopterin biosynthesis. {ECO:0000250|UniProtKB:Q9NQX3}. |
|
mmu:268566; |
cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; cytosol [GO:0005829]; dendrite [GO:0030425]; extrinsic component of cytoplasmic side of plasma membrane [GO:0031234]; extrinsic component of plasma membrane [GO:0019897]; GABA-ergic synapse [GO:0098982]; glycinergic synapse [GO:0098690]; inhibitory synapse [GO:0060077]; neuronal cell body [GO:0043025]; postsynapse [GO:0098794]; postsynaptic density [GO:0014069]; postsynaptic membrane [GO:0045211]; postsynaptic specialization [GO:0099572]; postsynaptic specialization membrane [GO:0099634]; postsynaptic specialization, intracellular component [GO:0099091]; synapse [GO:0045202]; synaptic membrane [GO:0097060]; ATP binding [GO:0005524]; cytoskeletal protein binding [GO:0008092]; identical protein binding [GO:0042802]; metal ion binding [GO:0046872]; molecular adaptor activity [GO:0060090]; molybdopterin adenylyltransferase activity [GO:0061598]; molybdopterin cofactor binding [GO:0043546]; molybdopterin molybdotransferase activity [GO:0061599]; nitrate reductase activity [GO:0008940]; signaling receptor binding [GO:0005102]; structural constituent of postsynaptic specialization [GO:0098879]; establishment of protein localization [GO:0045184]; establishment of synaptic specificity at neuromuscular junction [GO:0007529]; gamma-aminobutyric acid receptor clustering [GO:0097112]; glycine receptor clustering [GO:0072579]; maintenance of postsynaptic specialization structure [GO:0098880]; Mo-molybdopterin cofactor biosynthetic process [GO:0006777]; molybdenum incorporation into molybdenum-molybdopterin complex [GO:0018315]; molybdopterin cofactor biosynthetic process [GO:0032324]; neurotransmitter receptor localization to postsynaptic specialization membrane [GO:0099645]; postsynaptic neurotransmitter receptor diffusion trapping [GO:0098970]; response to metal ion [GO:0010038] |
12565882_Data suggest that the loss of neurotransmitter receptor clustering at inhibitory synapses causes the early lethality of gephyrin deficient mice. 14715953_Gephyrin is critical for glycine receptor clustering in hippocampal neurons. 15963957_gephyrin is required for the membrane insertion/stabilization of the GABAAR gamma2 subunit as well as for its subsequent localization in the postsynaptic membrane 17347650_Endogenous gephyrin undergoes proline-directed phosphorylation, which is followed by the recruitment of the peptidyl-prolyl isomerase Pin1. 18411266_gephyrin has splice-specific functions in molybdenum cofactor biosynthesis 19383528_Immunoelectron and confocal microscopy reveal that postsynaptic membrane specializations can be formed in the absence of gephyrin. 24096136_these findings suggest that gephyrin may be a key factor in BDNF-dependent GABAAR regulation in the amygdala. 25025157_study reveals that palmitoylation of gephyrin by DHHC-12 contributes to dynamic and functional modulation of GABAergic synapses 25082542_Within this network, collybistin can adopt open/active and closed/inactive conformations to act as a switchable adaptor that links gephyrin to plasma membrane phosphoinositides. 25263489_adult mice lacking interleukin-1 receptor 1 (IL-1R1) exhibit increased expression of both the excitatory scaffolding protein postsynaptic density-95 (PSD-95) and inhibitory scaffolding proteingephyrin, respectively, in the hippocampus. 25297980_Pin1-dependent signaling represents a mechanism to modulate GABAergic transmission by regulating NL2/gephyrin interaction. 25535349_These results reveal a homeostatic mechanism through which activity regulates the dynamics and function of perisomatic inhibitory synapses, and they identify a CaMKII-dependent phosphorylation site on gephyrin. 25701174_Data suggest that KIF5 regulates gephyrin sorting by a mechanism that involves GSK3 activity. 25772192_The results demonstrate that formation of GABAergic synapses containing a postsynaptic gephyrin scaffold is crucial for morphological differentiation, synaptic integration, and long-term survival of adult-born granule cells/ 26829712_Ablation of gephyrin result in the forebrain-specific loss of postsynaptic gamma2-GABAA receptors, reduces LTP and network excitability in the dentate gyrus. 27819299_Study unravels the critical role for cellular signal transduction pathways in determining gephyrin SUMOylation, phosphorylation and acetylation at identified residues to regulate its scaffolding properties at specific postsynaptic sites and in turn regulating GABAergic function and plasticity. 33878252_Artemisinin-treatment in pre-symptomatic APP-PS1 mice increases gephyrin phosphorylation at Ser270: a modification regulating postsynaptic GABAAR density. 36543537_Automated Image Analysis Reveals Different Localization of Synaptic Gephyrin C4 Splice Variants. |
ENSG00000171723 |
GPHN |
83.271370 |
0.0037822550 |
-8.046538 |
1.09360522 |
136.052914 |
0.00000000000000000000000000000019428342064179184846923100542510511539573986217768540241653808136194370448470217487696443114941757812630385160446166992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000158252677177314082384761021419659908817876893955946904949245775812944065627290921027814363242214312776923179626464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.625199916078971 |
0.520628258515921 |
165.29819298419 |
35.340895934591 |
| ENSMUSG00000047746 |
207215 |
Fbxo40 |
protein_coding |
P62932 |
FUNCTION: Probable substrate-recognition component of the SCF (SKP1-CUL1-F-box protein)-type E3 ubiquitin ligase complex that may function in myogenesis. {ECO:0000269|PubMed:17928169}. |
Cytoplasm;Metal-binding;Reference proteome;Ubl conjugation pathway;Zinc;Zinc-finger |
|
|
mmu:207215; |
cytoplasm [GO:0005737]; ubiquitin protein ligase activity [GO:0061630]; zinc ion binding [GO:0008270]; muscle cell differentiation [GO:0042692] |
17928169_FBXO40 may function as a regulator involved in the postnatal myogenesis. 22033112_The Fbxo40 is muscle specific in expression and is upregulated during differentiation. |
ENSG00000163833 |
FBXO40 |
153.414921 |
3.8891399941 |
1.959451 |
0.27620454 |
47.554503 |
0.00000000000534957352555244013452962360170180847358800679813839451526291668415069580078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000977034819439026040454673870058754665129940875090142071712762117385864257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
249.274476820501 |
48.015770997097 |
64.0950125724656 |
9.5598660615645 |
| ENSMUSG00000047747 |
330812 |
Rnf150 |
protein_coding |
Q5DTZ6 |
|
Alternative splicing;Glycoprotein;Membrane;Metal-binding;Reference proteome;Signal;Transmembrane;Transmembrane helix;Zinc;Zinc-finger |
|
|
mmu:330812; |
cytoplasm [GO:0005737]; membrane [GO:0016020]; metal ion binding [GO:0046872]; ubiquitin protein ligase activity [GO:0061630]; ubiquitin-dependent protein catabolic process [GO:0006511] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 25609945_rs10007052 in the RNF150 gene is significantly associated with the risk of COPD in Chinese populations of Hainan province. |
ENSG00000170153 |
RNF150 |
40.574353 |
0.3043751820 |
-1.716077 |
0.43005598 |
15.811078 |
0.00006999152780597782559906389066028964407450985163450241088867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004317986843016015504823079673712982184952124953269958496093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.5531562710278 |
5.6128201653939 |
60.9548917592413 |
12.8303336902053 |
| ENSMUSG00000047787 |
396184 |
Flrt1 |
protein_coding |
A0A452J8F8 |
|
Glycoprotein;Leucine-rich repeat;Membrane;Proteomics identification;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:396184; |
membrane [GO:0016020] |
16872596_FLRT1 is expressed at brain compartmental boundaries in embryo 28475893_Mice with deletions of FLRT1 and FLRT3 adhesion molecules develop macroscopic sulci with preserved layered organization and radial glial morphology. Cortex folding in these mutants does not require progenitor cell amplification but is dependent on changes in neuron migration. |
ENSG00000126500 |
FLRT1 |
10.695199 |
0.1186431090 |
-3.075300 |
0.94519896 |
10.647658 |
0.00110210352170763857190816104036912292940542101860046386718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0050336554250826840567478726029548852238804101943969726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.15432788437936 |
1.51459643617736 |
18.1580531988116 |
7.81502253563781 |
| ENSMUSG00000047904 |
20606 |
Sstr2 |
protein_coding |
P30875 |
FUNCTION: Receptor for somatostatin-14 and -28. This receptor is coupled via pertussis toxin sensitive G proteins to inhibition of adenylyl cyclase. In addition it stimulates phosphotyrosine phosphatase and PLC via pertussis toxin insensitive as well as sensitive G proteins. Inhibits calcium entry by suppressing voltage-dependent calcium channels. Acts as the functionally dominant somatostatin receptor in pancreatic alpha- and beta-cells where it mediates the inhibitory effect of somatostatin-14 on hormone secretion. Inhibits cell growth through enhancement of MAPK1 and MAPK2 phosphorylation and subsequent up-regulation of CDKN1B. Stimulates neuronal migration and axon outgrowth and may participate in neuron development and maturation during brain development. Mediates negative regulation of insulin receptor signaling through PTPN6. Inactivates SSTR3 receptor function following heterodimerization. {ECO:0000269|PubMed:8104154, ECO:0000269|PubMed:9507021}. |
Alternative splicing;Cell membrane;Cytoplasm;Disulfide bond;G-protein coupled receptor;Glycoprotein;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
The protein encoded by this gene is a receptor for somatostatin, which acts at many sites to inhibit the release of several hormones and other secretory proteins. The encoded protein is a member of the superfamily of receptors having seven transmembrane segments and is involved in many processes, including adenylyl cyclase inhibition, phosphotyrosine phosphatase stimulation, and inhibition of calcium entry and cell growth. Two transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Sep 2015]. |
mmu:20606; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; membrane [GO:0016020]; neuron projection [GO:0043005]; plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; neuropeptide binding [GO:0042923]; PDZ domain binding [GO:0030165]; peptide binding [GO:0042277]; somatostatin receptor activity [GO:0004994]; adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway [GO:0007193]; cellular response to estradiol stimulus [GO:0071392]; cellular response to glucocorticoid stimulus [GO:0071385]; cerebellum development [GO:0021549]; forebrain development [GO:0030900]; G protein-coupled receptor signaling pathway [GO:0007186]; neuropeptide signaling pathway [GO:0007218]; peristalsis [GO:0030432]; regulation of muscle contraction [GO:0006937]; response to starvation [GO:0042594]; spermatogenesis [GO:0007283] |
11897621_sst(2) receptors mediate inhibition of peristalsis in the jejunum 12145348_Brain somatostatin receptors 1,2,4 and 5 are up-regulated in somatostatin-deficient mice, and SSTR3 is down-regulated. 12441163_The somatostatin receptor subtype 2a (SSR(2(a))) was found in the cytoplasm and the plasma membrane of catecholaminergic CAD-cells. After serum deprivation, SSR(2(a)) was up-regulated in the cells. 12442323_Somatostatin 2 receptors are expressed in neurons and endocrine cells in the stomach and the small and large intestine. 12509482_Sst2 activation inhibits the AMPA component of glutamatergic synaptic responses. Decrease of AMPA currents induced by octreotide requires a concomitant activation of sst2 receptors with either NMDA and/or metabotropic glutamate receptors. 12676656_omatostatin regulates ductal bile formation in mice by inhibition of ductal fluid secretion, also by stimulation of ductal fluid absorption via interacting with SSTR2 on cholangiocytes, a process involving the intracellular cAMP/cGMP second messengers. 12752788_In the striatum, Sstr2 expression is identified in medium spiny projection neurons restricted to the matrix compartment and in cholinergic interneurons. 15093692_somatostatin receptors 1 and 2 are expressed in the adult mouse kidney 15464758_Evidence is provided that the overexpression of sst2 receptors has a profound impact on the sst2-mediated modulation of membrane excitability and neurotransmitter release in the mouse retina. 15544857_somatostatin receptor subtype-2 is responsible for somatostatin binding in cortical and amygdaloid regions 15601946_We provide direct evidence for activation of SSTR2 by an endogenous ligand after focal ischemia, which contributes to increased SSTR2 gene expression and postischemic neurodegeneration. 15857828_role of somatostatin receptor subtype 5 on somatostatin receptor subtype 2 action in pituitary tumor cells 17272399_the presence of sst(5) in the same cells modulates trafficking and cell surface regulation of sst(2A) and cellular desensitization to the effects of SRIF 17363388_sst2 deficiency is associated with exaggerated jejunal afferent sensitivity to mechanical/chemical stimulation, suggesting that somatostatin plays an inhibitory role to control visceral sensitivity by interacting with the sst2 receptor. 17408648_Decreased afferent responses to somatostatin receptor 2 stimulation correlated with lower expression of vagal somatostatin receptor 2 in stressed Nb-infected mice, confirming a link between molecular data and functional sequelae. 17525126_Increased glucagon concentration is associated with impaired glucose control in sst2(-/-) mice, resulting from decreased hepatic glucose storage, increased glycogen breakdown, and reduced lipid accumulation 17630220_Acute EAE disrupts the rat striatal SRIF receptor-effector system, providing insight into the molecular basis of EAE, leading to a better understanding of human multiple sclerosis. 17652715_sst(2) may be protective against retinal angiogenesis 17706924_The functional interactions between human somatostatin receptor 2 (hSSTR2) and human dopamine receptor 2 (hD2R) in both co-transfected CHO-K1 or HEK-293 cells as well as in cultured neuronal cells which express both the receptors endogenously. 17974627_there is a remodeling of the neuroendocrine mechanisms that regulate acid secretion in mutant mice lacking SSTR2 18298437_Data show that a reconsideration of the role of SSTR2 in the GI somatostatinergic effects is in order and further corroborate recent data on the role of other SSTR subtypes in the inflammatory effects of SOM during intestinal inflammation. 18434512_The somatostatin type 2A receptor is mobile and rapidly and laterally diffuses in hippocampal neuronal membranes. 18599562_SSTR2 is the predominant mediator of functional ocular antiangiogenic effects. 18624922_These results demonstrate that sst(2) activation protects against retinal ischaemia. However, in the presence of sst(2) over-expression sst(2) is functionally desensitised by agonists, possibly because of sustained RGS1 levels. 18752655_These results suggest that, in the adult mouse, testosterone is a major modulator of sst2A distribution on GHRH neurones. 19623609_sst(2a) and sst(4) were respectively detected in the dentate gyrus (DG) and the CA1 subfield suggesting that their functional interactions are not mediated by direct receptor coupling 20056831_Extraovarian somatostatin, acting through its receptors 2 and 5 present on granulosa cells, may be involved in mouse folliculogenesis by reducing recruitment of resting follicles. 20233796_Data indicate that OcF showing high-affinity binding to the sstr2. 20653396_SSTR2-based reporters can serve as reporters of gene transfer into non-small cell lung cancer. 20739383_Overexpressed in endomterium inendometriosis. 21896184_somatostatin receptors demonstrate specific expression in HCs and supporting cells of the mouse cochlea, and that absence of SST1 alters the expression of SST2. 22311350_sst2 may protect retinal cells from hypoxia, thus implementing the background to establish potential pharmacological targets. 22581439_SSTR2 was well expressed in most of the hypothalamic regions whereas it decreases significantly in ventromedial and arcuate nucleus of ApoD(-/-) mice. 22986312_SSTR2 was expressed in Kolliker's organ on E14, on cochlear duct hair cells & supporting cells on E17 and only on the organ of Corti at birth, increasing & peaking at P14 but dropping off at P21. 23831358_Findings suggest that somatostatin and its receptors (SSTR2 and SSTR5) are important markers in the regulation and development of Sertoli cell. 23898054_The aim of this study was to validate an animal model expressing SSTR2 and to correlate the immunohistochemical (IHC) analysis with (18)F-FDG and (68)Ga-DOTATOC uptake in vivo. 23913690_when activated by SST-14, SSTR2A internalizes and recycles via the Golgi, which requires ECE-1 degradation of SST-14 and receptor dissociation from beta-arrestins 24163059_The study encourages the use of liver tissue SSR2 protein and mRNA as a reliable tumor marker for liver cancer 24173662_OCT-P407 induces mRNA expression of SSTR-2 and caspase-3 and decreases that of VEGF in mice. 24685142_Inhibition of the Sstr2 receptor reversed the anticonvulsant effect mediated by cortistatin-14. 24998255_Knock-down of SSTR2 leads to loss of pluripotency in murine embryonic stem cells. 25149275_The expression and localization of the three receptors (SSTR3-SSTR5) in wild-type (WT), single-knockout (SSTR1 KO) and double-knockout SSTR1/SSTR2 (DKO) mice, are reported. 25268135_SST2 expression protects against gentamicin-induced auditory hair cell loss in the mammalian inner ear. 25683115_Loss of sst2 from pancreatic tissues activates PI3K signaling via AKT, leading to activation of NF-kappaB, amplification of oncogenic KRAS signaling, increased expression of CXCL16, and pancreatic tumor formation. 27986975_Both hippocampal sst2 and sst4 receptors selectively inhibit stress-induced HPA axis activation but mediate anxiolytic and antidepressive effects through distinct mechanisms. 29409919_Unpredictable chronic mild stress (UCMS) caused neuroendocrine changes, physical deteriorations, increased anxiety- and depressive-like behaviors and working memory impairments. sst2KO mice displayed behavioral alterations which were similar to stressed WT and exhibited exacerbated changes following UCMS exposure. The evaluations performed in the older mice showed significant long-term effects of UCMS exposure. 30261745_SSTR2 is mainly expressed in distal tubules and collecting ducts in murine kidneys, and the expression of SSTR2 decreases during cyst growth in two polycystic kidney disease mouse models 31381945_SSTR2 is primarily expressed and colocalized intracellularly. Activation of SSTR2, 5 and CB1R protected huntingtin-mutant knock-in striatal neurons from quinolinic-acid-induced toxicity. Co-activation of SSTRs and CB1R resulted in loss of protective effects, delayed ERK1/2 phosphorylation and altered receptor complex composition. 31686636_Somatostatin Type 2 Receptor Antibody Enhances Mechanical Hyperalgesia in the Dorsal Root Ganglion Neurons after Sciatic Nerve-pinch Injury: Evidence of Behavioral Studies and Bax Protein Expression. 33434183_Antagonizing somatostatin receptor subtype 2 and 5 reduces blood glucose in a gut- and GLP-1R-dependent manner. 34040115_A potential role for somatostatin signaling in regulating retinal neurogenesis. 35257878_Changes in dorsal root ganglion CGRP expression in mouse pinch nerve injury model: Modulation by Somatostatin type-2 receptor. |
ENSG00000180616 |
SSTR2 |
19.706485 |
0.1006145349 |
-3.313089 |
0.88811208 |
11.746104 |
0.00060970724088143493819835772029591680620796978473663330078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0029842740016407512046447969566997926449403166770935058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.66634550348624 |
3.05746401772887 |
46.3784433095449 |
19.5725473649879 |
| ENSMUSG00000047963 |
52331 |
Stbd1 |
protein_coding |
Q8C7E7 |
FUNCTION: Acts as a cargo receptor for glycogen. Delivers its cargo to an autophagic pathway called glycophagy, resulting in the transport of glycogen to lysosomes. {ECO:0000250|UniProtKB:O95210, ECO:0000269|PubMed:20810658}. |
Autophagy;Carbohydrate metabolism;Cell membrane;Endoplasmic reticulum;Glycogen metabolism;Membrane;Phosphoprotein;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix;Ubl conjugation |
|
|
mmu:52331; |
endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; perinuclear region of cytoplasm [GO:0048471]; phagophore assembly site membrane [GO:0034045]; plasma membrane [GO:0005886]; T-tubule [GO:0030315]; cargo receptor activity [GO:0038024]; enzyme binding [GO:0019899]; glycogen binding [GO:2001069]; molecular function activator activity [GO:0140677]; polysaccharide binding [GO:0030247]; starch binding [GO:2001070]; glycogen catabolic process [GO:0005980]; glycophagy [GO:0061723]; intracellular transport [GO:0046907]; substrate localization to autophagosome [GO:0061753] |
20810658_Stbd1 is involved in glycogen metabolism by binding to glycogen and anchoring it to membranes, thereby affecting its cellular localization and its intracellular trafficking to lysosomes. 23726947_Stbd1 is highly elevated in skeletal muscle of Pompe disease mice but suppression of its expression does not affect lysosomal glycogen accumulation. 27358407_results demonstrate that Stbd1 plays a dominant role in glycogen transport to lysosomes in liver and that the N-terminal transmembrane region and the C-terminal CBM20 domain are critical for this function. 28137759_mouse Stbd1 is a transmembrane endoplasmic reticulum (ER)-resident protein with the capacity to induce the formation of organized ER structures. 30772465_AMPK-dependent phosphorylation of Gapex-5 (also known as GTPase-activating protein and VPS9 domain-containing protein 1 (GAPVD1)) on Ser902 in hepatocytes and starch-binding domain 1 (STBD1) on Ser175 in multiple cells/tissues |
ENSG00000118804 |
STBD1 |
133.597305 |
2.4351432317 |
1.284007 |
0.26321275 |
23.558532 |
0.00000121170958468157460535103396581657619890393107198178768157958984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000106563281719802226924376925554227568682108540087938308715820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
192.015688561251 |
35.2270094500921 |
78.8519073778379 |
11.1130654735133 |
| ENSMUSG00000047992 |
240479 |
Dipk1c |
protein_coding |
Q8BQT2 |
|
Alternative splicing;Disulfide bond;Endoplasmic reticulum;Membrane;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:240479; |
endoplasmic reticulum membrane [GO:0005789] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000187773 |
DIPK1C |
19.640499 |
0.1507925706 |
-2.729363 |
0.68408459 |
15.995195 |
0.00006350346214356757373192818860729857988189905881881713867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003954557380910962314721213139989686169428750872611999511718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.72316859303479 |
2.32446390108344 |
31.3222897767785 |
9.71310369278762 |
| ENSMUSG00000048200 |
213573 |
Cracr2b |
protein_coding |
Q80ZJ8 |
FUNCTION: Plays a role in store-operated Ca(2+) entry (SOCE). {ECO:0000250}. |
Coiled coil;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:213573; |
cytoplasm [GO:0005737]; calcium ion binding [GO:0005509]; protein localization [GO:0008104]; regulation of store-operated calcium entry [GO:2001256]; store-operated calcium entry [GO:0002115] |
|
ENSG00000177685 |
CRACR2B |
35.805574 |
0.3729184593 |
-1.423068 |
0.54664767 |
6.471419 |
0.01096229495681172970422689871838883846066892147064208984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0362266524193133321318782691378146409988403320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.8545082463622 |
8.09236803654305 |
55.9224348477176 |
15.6339073380014 |
| ENSMUSG00000048347 |
93889 |
Pcdhb18 |
protein_coding |
Q91Y02 |
FUNCTION: Potential calcium-dependent cell-adhesion protein. {ECO:0000250}. |
Calcium;Cell adhesion;Cell membrane;Glycoprotein;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
Human_homologues NA; + ;NA |
|
mmu:93889; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell adhesion [GO:0007155]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
Human_homologues 28671736_Protocadherin B9 plays an important role in the progression rather than the pathogenesis of intestinal-type gastric cancers. 30324761_A functional analysis showed that PCDHB9 modulated cell migration and adhesion. 35390792_Protocadherin B9 Is Associated with Human Esophageal Squamous Cell Carcinoma Progression.; + ;NA |
ENSG00000177839+ENSG00000120324 |
PCDHB9+PCDHB10 |
41.238267 |
0.3935081387 |
-1.345535 |
0.43980808 |
9.295873 |
0.00229670714652827285054415007437000895151868462562561035156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0095923891570931316824522028241517546121031045913696289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
23.3699145160027 |
7.23522258422789 |
59.3886433776704 |
13.0651780559525 |
| ENSMUSG00000048368 |
27047 |
Omd |
protein_coding |
O35103 |
FUNCTION: May be implicated in biomineralization processes. Has a function in binding of osteoblasts via the alpha(V)beta(3)-integrin (By similarity). {ECO:0000250}. |
Cell adhesion;Disulfide bond;Extracellular matrix;Glycoprotein;Leucine-rich repeat;Proteoglycan;Reference proteome;Repeat;Secreted;Signal;Sulfation |
|
|
mmu:27047; |
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; cell adhesion [GO:0007155] |
16970923_found transforming growth factor beta1 to down regulate OSAD, while bone morphogenetic protein 2 up regulates OSAD 18496725_OSAD overexpression enhanced the differentiation and maturation of osteoblasts in vitro. 31638177_These results demonstrate that Omd expression is regulated during osteoblast differentiation, and that the protein product osteoadherin serves roles in the apoptosis and growth of osteoblast cells |
ENSG00000127083 |
OMD |
4144.406420 |
0.4554822466 |
-1.134533 |
0.08293742 |
183.669807 |
0.00000000000000000000000000000000000000000765942362790095646283505150334394715431607448391962998331978893623109246173471143515800529091231946476013830310547447766111872624605894088745117187500000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000009333467256014989572605325039900045511466336815690579869485804586823425507757814521123371373268978279207075399170889795641414821147918701171875000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2575.45310726595 |
148.138804774238 |
5654.34355951721 |
241.049694585827 |
| ENSMUSG00000048399 |
71338 |
Tprg |
protein_coding |
Q8CB49 |
|
Cytoplasm;Reference proteome |
|
|
mmu:71338; |
cytoplasm [GO:0005737] |
18256694_The Tprg gene is predominantly expressed in keratinocytes, is physically associated with the p63 gene during evolution, and is directly regulated by p63 through a long-distance enhancer located within the Tprg locus. 18256694_Tprg gene is predominantly expressed in skin, is physically associated with the p63 gene during evolution, and directly regulated by p63 through a long-distance enhancer located within the Tprg locus. |
ENSG00000188001 |
TPRG1 |
165.599182 |
0.4106428259 |
-1.284044 |
0.20788094 |
37.721359 |
0.00000000081606098521076398249212943250053588217785005554105737246572971343994140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000116062006785530870157275185484274659941661411721725016832351684570312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
93.9458933188928 |
13.5837515645763 |
228.777632038229 |
23.627574071089 |
| ENSMUSG00000048402 |
14633 |
Gli2 |
protein_coding |
Q0VGT2 |
FUNCTION: Functions as transcription regulator in the hedgehog (Hh) pathway (PubMed:9006072). Functions as transcriptional activator (PubMed:10806483). May also function as transcriptional repressor (PubMed:10433919). Requires STK36 for full transcriptional activator activity (PubMed:10806483). Binds to the DNA sequence 5'-GAACCACCCA-3' which is part of the TRE-2S regulatory element (By similarity). Is involved in the smoothened (SHH) signaling pathway (PubMed:10433919). Required for normal skeleton development (PubMed:9006072). {ECO:0000250|UniProtKB:P10070, ECO:0000269|PubMed:10806483, ECO:0000269|PubMed:9006072, ECO:0000305|PubMed:10433919}. |
Acetylation;Activator;Cell projection;Cilium;Cytoplasm;Developmental protein;DNA-binding;Metal-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Repressor;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:14633; |
axoneme [GO:0005930]; ciliary tip [GO:0097542]; cilium [GO:0005929]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; GLI-SUFU complex [GO:1990788]; membrane [GO:0016020]; motile cilium [GO:0031514]; nuclear speck [GO:0016607]; nucleolus [GO:0005730]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; chromatin binding [GO:0003682]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; promoter-specific chromatin binding [GO:1990841]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; transcription cis-regulatory region binding [GO:0000976]; zinc ion binding [GO:0008270]; anatomical structure development [GO:0048856]; anatomical structure formation involved in morphogenesis [GO:0048646]; anterior/posterior pattern specification [GO:0009952]; axon guidance [GO:0007411]; branching morphogenesis of an epithelial tube [GO:0048754]; cell differentiation [GO:0030154]; cell population proliferation [GO:0008283]; cellular response to organic cyclic compound [GO:0071407]; cellular response to virus [GO:0098586]; cerebellar cortex morphogenesis [GO:0021696]; chondrocyte differentiation [GO:0002062]; cochlea morphogenesis [GO:0090103]; developmental growth [GO:0048589]; dorsal/ventral neural tube patterning [GO:0021904]; dorsal/ventral pattern formation [GO:0009953]; embryonic digestive tract development [GO:0048566]; embryonic digit morphogenesis [GO:0042733]; epidermal cell differentiation [GO:0009913]; epithelial cell proliferation [GO:0050673]; floor plate formation [GO:0021508]; gene expression [GO:0010467]; hair follicle morphogenesis [GO:0031069]; head development [GO:0060322]; heart development [GO:0007507]; hindbrain development [GO:0030902]; hindgut morphogenesis [GO:0007442]; in utero embryonic development [GO:0001701]; kidney development [GO:0001822]; lung development [GO:0030324]; mammary gland development [GO:0030879]; mammary gland duct morphogenesis [GO:0060603]; morphogenesis of an epithelium [GO:0002009]; negative regulation of apoptotic process [GO:0043066]; negative regulation of chondrocyte differentiation [GO:0032331]; negative regulation of hh target transcription factor activity [GO:1990787]; negative regulation of smoothened signaling pathway [GO:0045879]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural tube development [GO:0021915]; neuron development [GO:0048666]; neuron differentiation [GO:0030182]; notochord regression [GO:0060032]; odontogenesis of dentin-containing tooth [GO:0042475]; osteoblast development [GO:0002076]; osteoblast differentiation [GO:0001649]; pattern specification process [GO:0007389]; pituitary gland development [GO:0021983]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA replication [GO:0045740]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of epithelial cell proliferation [GO:0050679]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of stem cell proliferation [GO:2000648]; positive regulation of transcription by RNA polymerase II [GO:0045944]; prostatic bud formation [GO:0060513]; proximal/distal pattern formation [GO:0009954]; regulation of smoothened signaling pathway involved in dorsal/ventral neural tube patterning [GO:1901620]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal system development [GO:0001501]; smoothened signaling pathway [GO:0007224]; smoothened signaling pathway involved in dorsal/ventral neural tube patterning [GO:0060831]; smoothened signaling pathway involved in regulation of cerebellar granule cell precursor cell proliferation [GO:0021938]; smoothened signaling pathway involved in spinal cord motor neuron cell fate specification [GO:0021776]; smoothened signaling pathway involved in ventral spinal cord interneuron specification [GO:0021775]; spinal cord dorsal/ventral patterning [GO:0021513]; spinal cord motor neuron differentiation [GO:0021522]; spinal cord ventral commissure morphogenesis [GO:0021965]; stem cell proliferation [GO:0072089]; tube development [GO:0035295]; ventral midline development [GO:0007418]; ventral spinal cord development [GO:0021517] |
12361967_Gli2 is required for mediating some of the inappropriate Shh signaling in Ptc mutants. Our studies demonstrate that, in mammals, Gli1 is not required for Shh signaling and that Gli2 mediates inappropriate activation of the pathway due to loss of Ptc. 12533516_Sonic hedgehog-dependent Gli2 activation plays a critical role in epithelial homeostasis by promoting proliferation through the transcriptional control of cell cycle regulators. 12812795_Gli2 and Gli3, mediators of Shh signaling, are required for the development of the ventral neural cell fates. 12837627_Gli2 plays an important role in regulating oligodendrocyte specification and differentiation in the caudal neural tube 14602680_Gli2 and Gli3 are required for Shh-dependent sclerotome induction. 14681189_role of the SHH-GLI2/3 signaling pathway in stem cells in the developing neocortex 14980515_Gli2 is necessary for normal endochondral bone development. 15039437_Meox and Gli have roles in skeletal myogenesis 15215207_Data show that Gli2 and Gli3 act together with Sonic hedgehog to regulate neuronal patterning by controlling progenitor cell segregation. 15604102_there are both positive and negative regulatory functions for Gli2 and Gli3 in the control of Myf5 activation in the epaxial muscle progenitor cells and in dorsoventral and mediolateral patterning of the somite 15614767_Fgf15 is directly regulated by Shh signaling through Gli proteins in the developing diencephalon and midbrain 15793308_Shh, functioning via Gli1/2, can specify mesodermal cells into the cardiac muscle lineage 15806168_Gli1 plays a central role in medulloblastoma formation in Ptc1+/- mice and that Gli2 may also contribute to oncogenesis. 16254602_cilia have a direct role in Gli2 and Gli3 processing and Shh signal transduction 16396903_Results demonstrate that SHH-SMO signaling controls renal morphogenesis via transcriptional control of Gli1 and 2, renal patterning and cell cycle regulator genes in a manner that is opposed by GLI3. 16571630_Shh regulates Gli2 activator functions. 16611981_Processing and degradation of Gli2 are suppressed by Shh signaling in vivo; this is the first demonstration of a molecular mechanism by which the Gli2 transcriptional activity is regulated by Shh signaling. 16880529_These results indicate that Gli2 mediates hedgehog signaling in osteoblasts and is a powerful activator of BMP-2 gene expression, which is required in turn for normal osteoblast differentiation. 17026983_Taken together, these observations indicate that Gli2 and Gli3 perform rhombomere-specific function during dorsoventral patterning of the hindbrain. 17283082_explain, at the molecular level, why Gli2 and Gli3 are differentially processed 17395647_A proper balance of Gli3 repressor proteins and Gli2/3 activator proteins is required along the length of the dorsoventral axis of the inner ear to mediate graded levels of Shh signaling, emanating from ventral midline tissues. 17442891_These results suggest that Ihh regulates osteoblast differentiation of mesenchymal cells through up-regulation of the expression and function of Runx2 by Gli2. 17638910_transforming growth factor-beta has a role in Smad3-dependent activation of Gli2 and Gli1 expression 17721996_Gene silencing of transcription factor Gli2 inhibits basal cell carcinomalike tumor growth 18279667_These findings suggest that the repressor form of Gli2 preferentially binds to the GliREs to control Fgf15 expression. 18590716_Disruption of FKBP8 function activates the Shh signaling pathway cell-autonomously dependent on the Gli2 transcription factor and kinesin-2 subunit Kif3a, a component of the intraflagellar transport machinery used to generate cilia. 18927476_These data reveal a novel transcriptional cascade, involving Gli2 --> Sox2 --> Hes5, which maintains the undifferentiated state of telencephalic neuroepithelial cells. 19056373_phosphorylation of Gli2 by PKA induces Gli2 processing and destabilization in vivo and plays an important role in the Hh-regulated mouse embryonic patterning. 19124651_Shh stimulation of progenitor proliferation and cell diversification requires Gli2 and Hes1 activity. 19273836_When hedgehog signaling was reduced by T lineage-specific transgenic expression of a transcriptional-repressor form of Gli2, differentiation to double-positive thymocyte after pre-TCR signal transduction was increased. 19574535_There is increased expression of cyclins D1, D2, and E1 associated with increased gli2 levels. 19647223_Gli2 overexpression and p53 deficiency have opposing effects on chondrocyte differentiation, but additive effects negatively regulating apoptosis. 19684112_Sufu regulates Gli protein levels by antagonizing the activity of Spop, a conserved Gli-degrading factor 19815519_found that mice deficient in Gli2 display aberrant temporo-mandibular joint development such that the condyle loses its growth-plate-like cellular organization and no disk is formed. 19906844_the Gli2 activator alone is sufficient to induce vascularization of the hypertrophic cartilage 19996169_changes in ciliary and nuclear trafficking of Gli2 are triggered by Hh-dependent accumulation of activated Smo in cilium, resulting in a shift from primarily cytoplasmic localization to accumulation at the distal tip of the cilium and within the nucleus 20081843_Results show that Gli1 and Gli2 are acetylated proteins and that their HDAC-mediated deacetylation promotes transcriptional activation and sustains a positive autoregulatory loop through Hedgehog-induced upregulation of HDAC1. 20479231_primary cilia regulate the level of Hh activation, and that ablation of these cellular organelles is sufficient to cause significant up-regulation of the Hh pathway in pancreata of mice overexpressing GLI2 20711444_Pias1-dependent SUMOylation influences Gli protein activity 20934421_Gli2, which encodes a major Gli activator, is required for proliferation of specific groups of pituitary progenitors but not for initial dorsoventral patterning. 20956384_The hedgehog signaling pathway at vertebrate cilia cause dissociation of inactive SuFu-Gli complexes, a process inhibited by PKA. 21151991_Pkd1 and Kif3a counterbalance the regulation of osteogenesis and adipogenesis through differential regulation of Runx2 and PPARgamma by Gli2 21209421_In fetal testes prior to Leydig cell differentiation, expression of Gli1 and Gli2 is abolished by Hedgehog inhibitor GANT61. At later stages, GANT61 inhibits Gli1 expression but has no effects on Gli2 expression and fetal Leydig cell appearance. 21209912_ciliary localization of Sufu is dependent on ciliary-localized Gli proteins, and is inhibited by PKA activation 21337666_The proper balance between the Gli2 full-length activator and repressor is essential for Hh signaling. 21454528_CCL5 signaling induces GLI2 through a PI3K-AKT-IkappaBalpha-p65 pathway and requires GLI2 transcriptional activity to modulate IL-6 expression and Ig secretion in vitro and in vivo 21559470_Ascl1/Mash1 is a novel target of Gli2 during Gli2-induced neurogenesis in P19 EC cells 21586556_Results suggest functions of Shh-Gli signaling in sexually dimorphic development during embryogenesis. Shh expressed in urethral plate epithelium regulates genital tubercle differentiation through Gli2 in mesenchyme. (Shh = sonic hedgehog protein) 22549777_SUMO modification inhibits GLI2 transcriptional activity by recruiting HDAC5. 22698544_Data show that Transmembrane protein 107 (Tmem107) is required for normal Sonic hedgehog (Shh) signaling in the neural tube and acts in combination with Gli2 and Gli3 to pattern ventral and intermediate neuronal cell types. 22841643_Establishment of a complete range of anterior-posterior positional identities in the embryonic limb requires integration of the spatial distribution, timing, and dosage of GLI2 and GLI3 activators and repressors. 23034632_These studies establish Sufu and Kif7 as crucial components in the regulation of Gli2 localization and activity, and illustrate their overlapping functions in skin development and tumor suppression. 23110168_Data indicate that epithelial Gli2 expression was sufficient to stimulate Il-1beta expression, repress Gast gene expression and increase proliferation, leading to antral hyperplasia. 23201751_we show that Hh signaling regulates Ndp expression in the developing retina and chromatin immunoprecipitation (ChIP) reveals Hh-dependent enrichment of Gli2 binding at the Ndp locus, suggesting that the relationship is direct 23383082_activation of Gli2 impairs bone formation in postnatal growing mice 23645682_Gli2, but not Gli3, is the major effector for hedgehog proteins signaling during intestine development. 23762415_Gli2 acetylation at lysine 757 regulates hedgehog-dependent transcriptional output by preventing its promoter occupancy. 23872071_PACAP blocks canonical Shh signaling by a mechanism that involves activation of protein kinase A (PKA) and inhibition of the translocation of the Shh-dependent transcription factor Gli2 into the primary cilium. 24036067_Association with DNA-bound Pax3 strengthens the ability of both Zic1 and Gli2 to transactivate Myf5 in the epaxial somite. 24121003_Data suggests with that Gli2 may play a novel role in the self-renewal of pluripotent stem cells. 24373970_Phosphorylation at P1-6 sites determines Gli2 transcriptional activity. 24463817_Systematic analysis in ESCs reveals that distinct regions of Gli2 regulate its nuclear and ciliary localization. 24586787_The teratogenic effects of prenatal ethanol exposure are exacerbated by Sonic Hedgehog or GLI2 haploinsufficiency in the mouse. 24617895_Suggest a novel miR-326-negative feedback loop in regulating the activity of Shh/Gli/Smo signaling. 24693967_The sonic hedgehog-induced type 3 deiodinase facilitates tumorigenesis of basal cell carcinoma by reducing Gli2 inactivation. 25695606_The results uncover that both Shh/Gli2 and Shh/Gli3 signals are required for proper development of murine placentas and are possibly essential for pregnant maintenance. 25833741_Gli2 protein expression is downregulated during neural tube patterning, regulating Shh signaling. Shh-induced upregulation of Gli2 transcription prevents Gli activity levels from adapting in a different cell type. 25834022_The ciliary localization of Gli2 is crucial for cilium-dependent activation of Hedgehog signaling. 25908851_Expression of Gli2A in T-cells altered gene expression profiles. 25910616_Study suggested that loss of Gli2 mRNA mediated the anterior-restricted defect in conditional Dicer knockdown mice and showed that miR-106b positively regulated Gli2 mRNA expression 26010061_the three GLI factors(GLI1, GLI2, and GLI3) in mature hepatocytes form an interactive transcriptional network that is involved in the control of target genes associated with metabolic zonation as well as with lipid and drug metabolism 26193634_While darinaparsin ameliorated fibrosis in WT and Gli1-KO mice, it was not effective in conditional Gli2-KO mice, supporting GLI2 as a direct darinaparsin target. 26238488_GLI2 binds to the IL-6Ralpha promoter and regulates its activity as well as the expression of this receptor. 26387942_Sufu deletion early in embryogenesis resulted in unstable Gli2 and Gli3 activity, leading to the ectopic activation of Shh signaling. 27543868_the GLI2 protein level could serve as a feasible marker of ligand-dependent hedgehog activation in pancreatic neoplasms. 27585885_mice with single-allele Gli2 mutations exhibit increased penetrance and severity of Holoprosencephaly in response to low-dose teratogen exposure. 27760825_Gli1 and Gli2 exhibited different functions in the regulation of p63 expression or proliferation of p63(+) cells in Kras-AR driven tumors. 27802276_Our findings suggest that ciliopathic facial phenotypes are generated via loss of both GLI3R and GLI2R and that this pathology occurs via a de-repression mechanism. Furthermore, these studies suggest a novel role for GLI2R in craniofacial development. 27814383_Gli2+/-;Gli3Delta699/+ mice can serve as a genetic mouse model for common DSD. 27849569_Sufu is upregulated in active Shh responding tissues and accompanies Gli activators translocating into and Gli repressors out of the nucleus. 28280246_Gli2, the major Hedgehog pathway transcriptional effector, acts within mouse mammary stromal cells to direct a hormone-responsive niche signaling program by activating expression of factors that regulate epithelial stem cells as well as receptors for the mammatrophic hormones estrogen and growth hormone. 28286175_Despite increased production of full-length GLI2 and GLI3 isoforms, previously identified GLI targets important for mandibular and glossal development (Foxf1, Foxf2, Foxd1 and Foxd2) were transcriptionally downregulated in Kif3a(f/f);Wnt1-Cre embryos. 28461568_studies identify a novel molecular mechanism of regulation of CD40L by the transcription factor GLI2 in the tumor microenvironment downstream of CCR3 signaling 28673820_Gli2 and Gli3 are dephosphorylated and activated in cilia and that impaired Gli2 and Gli3 processing in Ta3 mutant is at least in part due to a decrease in Gli2 and Gli3 phosphorylation. 29205155_Here by inducing expression of constitutively active Smoothened (SmoM2) or Gli2 (DeltaNGli2) in the adipocyte lineage of postnatal mice, the authors show that targeted activation of Hedgehog signaling suppresses high-fat-diet-induced obesity and improves whole-body glucose tolerance and insulin sensitivity. 29342244_Gli2 rescues neural stem cell proliferation, differentiation and migration in brain development. 29967343_knockdown inihibits tumor growth in cervical cancer xenograft 30001532_Gli2 upregulation changed the cytokine expression profile in mouse pancreatic acinar cells, mainly decreasing the pro-inflammatory cytokines interleukin (IL)-6, interferon-gamma, and FasL. 30463852_that Gli2 plays an unexpected role in cell cycle re-entry through an autophagy-mediated regulation on ciliary length in fibroblasts 30554998_establishes the dual role of SUFU inSonic Hedgehog medulloblastoma (MB) and provides mechanistic insights into transcriptional regulation underlying Gli2-mediated SHH MB tumorigenesis 31000813_PRMT7 methylates and suppresses GLI2 binding to SUFU thereby promoting its activation. 31167144_GLI2-mediated transcriptional program in gut mesenchymal cells induces key niche signals such as Wnt ligands and TGF-beta signaling genes that promote tumor initiation. 31264977_findings establish an important role for Shh upregulation in preventing atopic dermatitis, by increased Gli-driven Treg cell-mediated immune suppression, paving the way for a potential new therapeutic strategy. 33125874_Relative Levels of Gli1 and Gli2 Determine the Response of Ventral Neural Stem Cells to Demyelination. 33728992_Gli2-regulated activation of hepatic stellate cells and liver fibrosis by TGF-beta signaling. 34267186_PGE2-JNK signaling axis non-canonically promotes Gli activation by protecting Gli2 from ubiquitin-proteasomal degradation. 36272934_A study on etiology of incontinence in double knockout mouse model. |
ENSG00000074047 |
GLI2 |
17.113212 |
4.6581940297 |
2.219771 |
0.74381226 |
8.173025 |
0.00425179383677711745376237573168509698007255792617797851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0161908309304472641798611221020109951496124267578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
28.7483005280276 |
15.4872233929347 |
6.17155497276465 |
2.50657965729761 |
| ENSMUSG00000048424 |
223332 |
Ranbp3l |
protein_coding |
Q6PDH4 |
FUNCTION: Nuclear export factor for BMP-specific SMAD1/5/8 that plays a critical role in terminating BMP signaling and regulating mesenchymal stem cell differentiation by blocking osteoblast differentiation to promote myogenic differention. Directly recognizes dephosphorylated SMAD1/5/8 and mediates their nuclear export in a Ran-dependent manner. {ECO:0000250|UniProtKB:Q86VV4, ECO:0000269|PubMed:25755279}. |
Alternative splicing;Cytoplasm;Nucleus;Reference proteome |
|
|
mmu:223332; |
cytoplasm [GO:0005737]; nuclear pore [GO:0005643]; SMAD binding [GO:0046332]; mesenchymal cell differentiation involved in bone development [GO:1901706]; negative regulation of osteoblast differentiation [GO:0045668]; positive regulation of myoblast differentiation [GO:0045663]; protein export from nucleus [GO:0006611] |
25755279_Results demonstrate that RanBP3L, as a nuclear exporter for BMP-specific Smads, plays a critical role in terminating BMP signaling and regulating mesenchymal stem cell differentiation. 34233711_Loss of RANBP3L leads to transformation of renal epithelial cells towards a renal clear cell carcinoma like phenotype. |
ENSG00000164188 |
RANBP3L |
61.821778 |
0.2256783931 |
-2.147660 |
0.34361182 |
38.461317 |
0.00000000055849784204253638516278194950208380031853039326961152255535125732421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000080942332350065790983727887072593509820706003665691241621971130371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
22.4987063283763 |
5.40940354641375 |
99.693665913427 |
16.7490771071626 |
| ENSMUSG00000048442 |
66528 |
Smim5 |
protein_coding |
Q8BT42 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:66528; |
membrane [GO:0016020] |
|
ENSG00000204323 |
SMIM5 |
68.364245 |
0.3882962258 |
-1.364770 |
0.35255738 |
14.842582 |
0.00011686680034571501061221171502424454047286417335271835327148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006878342746777167844574041311034307000227272510528564453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.0314304799742 |
9.63349223217008 |
100.519726660974 |
17.6949850801875 |
| ENSMUSG00000048502 |
278672 |
Duxbl1 |
protein_coding |
L7MUF2 |
Human_homologues FUNCTION: [Isoform 1]: Transcription factor that is selectively and transiently expressed in cleavage-stage embryos (PubMed:28459457). Binds to double-stranded DNA elements with the consensus sequence 5'-TAATCTAATCA-3' (PubMed:28459457, PubMed:28459454, PubMed:29572508, PubMed:30540931, PubMed:30315230). Binds to chromatin containing histone H3 acetylated at 'Lys-27' (H3K27ac) and promotes deacetylation of H3K27ac. In parallel, binds to chromatin that lacks histone H3 acetylation at 'Lys-27' (H3K27ac) and recruits EP300 and CREBBP to promote acetylation of histone H3 at 'Lys-27' at new sites (PubMed:26951377). Involved in transcriptional regulation of numerous genes, primarily as transcriptional activator, but mediates also repression of a set of target genes (PubMed:17984056, PubMed:27378237, PubMed:26951377, PubMed:28459457, PubMed:28459454, PubMed:29618456, PubMed:30540931, PubMed:29572508). Promotes expression of ZSCAN4 and KDM4E, two proteins with essential roles during early embryogenesis (PubMed:27378237, PubMed:26951377, PubMed:28459457, PubMed:29618456). Heterologous expression in cultured embryonic stem cells mediates also transcription of HERVL retrotransposons and transcripts derived from ACRO1 and HSATII satellite repeats (PubMed:28459457). May activate expression of PITX1 (PubMed:17984056). May regulate microRNA (miRNA) expression (PubMed:24145033). Inappropriate expression can inhibit myogenesis and promote apoptosis (PubMed:26951377, PubMed:28935672, PubMed:29618456). {ECO:0000269|PubMed:17984056, ECO:0000269|PubMed:24145033, ECO:0000269|PubMed:26951377, ECO:0000269|PubMed:27378237, ECO:0000269|PubMed:28459454, ECO:0000269|PubMed:28459457, ECO:0000269|PubMed:28935672, ECO:0000269|PubMed:29572508, ECO:0000269|PubMed:29618456, ECO:0000269|PubMed:30315230, ECO:0000269|PubMed:30540931}.; FUNCTION: [Isoform 2]: Probably inactive as a transcriptional activator, due to the absence of the C-terminal region that is important for transcriptional activation. Can inhibit transcriptional activation mediated by isoform 1. Heterologous expression of isoform 2 has no deleterious effect on cell survival. {ECO:0000269|PubMed:29618456}. |
DNA-binding;Homeobox;Nucleus;Reference proteome |
|
|
|
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; regulation of T cell differentiation [GO:0045580]; regulation of transcription by RNA polymerase II [GO:0006357] |
17911620_Involved in thymocyte development. Knockdown by shRNA impaired CD4/8 double-negative thymocyte differentiation. Overexpression impaired TCR-beta expression and CD4/8 double-positive thymocyte production. Expressed also in B-lymphocytes. 20063414_Duxbl proteins play regulatory roles during myogenesis and reproductive developments 30765463_Duxbl is a regulator of beta-selection by inducing apoptosis in T cells with a nonfunctional rearrangement. |
ENSG00000260596 |
DUX4 |
212.503616 |
6.5835359656 |
2.718863 |
0.20223160 |
181.018156 |
0.00000000000000000000000000000000000000002904818043219283069377237749923892377464191288306664214709206173549210408899096541441035542705297416957339884380218109072302468121051788330078125000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000034567334714309465466852659849785947396373512616842637420118807495407534604038410341715552053121370251059785161373838491272181272506713867187500000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
379.706799470887 |
53.4003605504401 |
57.6752069787541 |
6.68166155610683 |
| ENSMUSG00000048572 |
226040 |
Tmem252 |
protein_coding |
Q8C353 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:226040; |
membrane [GO:0016020] |
|
ENSG00000181778 |
TMEM252 |
382.609990 |
0.1233617909 |
-3.019032 |
0.15523842 |
381.934808 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000047176515533707019948739457979795923462866957799539321967890258222194913649820994693649957581698507185423597643701863839723924771127603995280632733898898690561025043969867570271973 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000001996090790581736777993799370748702422261702753756659073078380030137599287201135086920804767352606204372122667910152535970600688263028599504380614387064068800404125932938487353976 |
Yes |
No |
83.0320386646639 |
8.95175356337949 |
673.077441827522 |
46.1079413742324 |
| ENSMUSG00000048763 |
15410 |
Hoxb3 |
protein_coding |
P09026 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15410; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; angiogenesis [GO:0001525]; anterior/posterior pattern specification [GO:0009952]; cartilage development [GO:0051216]; definitive hemopoiesis [GO:0060216]; embryonic skeletal system morphogenesis [GO:0048704]; face development [GO:0060324]; glossopharyngeal nerve morphogenesis [GO:0021615]; hematopoietic progenitor cell differentiation [GO:0002244]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of neurogenesis [GO:0050767]; regulation of transcription by RNA polymerase II [GO:0006357]; rhombomere development [GO:0021546]; thyroid gland development [GO:0030878] |
12748289_These data suggest a direct physiological role of Hoxb4 and Hoxb3 in regulating stem cell regeneration and that these genes are required for maximal proliferative response. 12954718_Results suggest that members of the Hox groups (Hoxa3, Hoxb3, Hoxd3) coordinate both suppression and activation mechanisms that ensure distinction between the multiple rhombomeres in the developing hindbrain. 15768390_b3IIIa in the Hoxb3 gene locus-lacZ transgene was expressed in a subset of enteric neuroblasts during early development of the gut 17309827_The Hoxb3 gene plays an essential role in regulating B lymphopoiesis in the BM of adult mice. 17761289_In this expt. mice with a compound deficiency in hoxa9, hoxb3 and hoxb4 (hoxa9/b3/b4) were investigated for evidence of synergy between these genes in hematopoiesis. 20515659_these results suggest that the multiple non-coding RNAs could be involved in the regulation of Hoxb3. 21320481_identified a novel Hoxb3 binding site S3 on the Hoxb1 locus and confirmed protein binding to this site in hindbrain tissues from the Hoxb3(Tg) mutant |
ENSG00000120093 |
HOXB3 |
88.204878 |
2.5062295213 |
1.325519 |
0.44238595 |
8.690851 |
0.00319811243746139211435730587140824354719370603561401367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0126397635307244227825806959231158543843775987625122070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
126.464078711679 |
39.3893381341118 |
50.4598950882439 |
11.8550052931853 |
| ENSMUSG00000048826 |
240025 |
Dact2 |
protein_coding |
Q7TN08 |
FUNCTION: Involved in regulation of intracellular signaling pathways during development. Negatively regulates the Nodal signaling pathway, possibly by promoting the lysosomal degradation of Nodal receptors, such as TGFBR1. May be involved in control of the morphogenetic behavior of kidney ureteric bud cells by keeping cells epithelial and restraining their mesenchymal character. May play an inhibitory role in the re-epithelialization of skin wounds by attenuating TGF-beta signaling. {ECO:0000269|PubMed:17197390, ECO:0000269|PubMed:18716284, ECO:0000269|PubMed:20685821}. |
Coiled coil;Developmental protein;Phosphoprotein;Reference proteome |
|
|
mmu:240025; |
cytoplasm [GO:0005737]; mitochondrion [GO:0005739]; beta-catenin binding [GO:0008013]; delta-catenin binding [GO:0070097]; protein kinase A binding [GO:0051018]; protein kinase C binding [GO:0005080]; transcription factor binding [GO:0008134]; epithelial cell morphogenesis [GO:0003382]; hematopoietic progenitor cell differentiation [GO:0002244]; inner medullary collecting duct development [GO:0072061]; negative regulation of cell adhesion [GO:0007162]; negative regulation of nodal signaling pathway [GO:1900108]; skin development [GO:0043588] |
16881060_Dact2 expression is most prominent during organogenesis of the thymus, kidneys, and salivary glands, with much lower levels in the somites and in the developing CNS. 18716284_Dpr2 plays an inhibitory role in the re-epithelialization of adult skin wounds by attenuating TGFbeta signaling. 20170752_Dact2 may play important signal-modulating roles in the adjacent epithelial cells including the enamel knot signaling centers and pre-ameloblasts. 20685821_Dact2 plays a role in regulating morphogenesis by renal collecting duct cells, probably by protecting cells from overly strong TGF-beta pathway activation. 21718540_conserved role for Dact2 protein in kinase-regulated biochemistry involving Vangl and Dvl. 23349981_we have identified a novel role for Dact2 as an inhibitor of the canonical Wnt pathway in embryonic tooth development through its regulation of cell proliferation and differentiation. 29199082_Our findings provide a novel DACT2 demethylating stimulator to verify and expand previously established link between DACT2 and colorectal cancer (CRC), suggesting DACT2 demethylation is strongly correlated with CRC prognosis. |
ENSG00000164488 |
DACT2 |
53.846697 |
2.5815532141 |
1.368239 |
0.43213785 |
9.836782 |
0.00171056485602647944006815539097487999242730438709259033203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0074316383020522919916617254898483224678784608840942382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
78.0929559342362 |
23.7429435278584 |
30.250376210915 |
7.08242775692235 |
| ENSMUSG00000048922 |
108912 |
Cdca2 |
protein_coding |
Q14B71 |
FUNCTION: Regulator of chromosome structure during mitosis required for condensin-depleted chromosomes to retain their compact architecture through anaphase. Acts by mediating the recruitment of phopsphatase PP1-gamma subunit (PPP1CC) to chromatin at anaphase and into the following interphase. At anaphase onset, its association with chromatin targets a pool of PPP1CC to dephosphorylate substrates (By similarity). {ECO:0000250}. |
Alternative splicing;Cell cycle;Cell division;Isopeptide bond;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation |
|
|
mmu:108912; |
chromosome [GO:0005694]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; cell division [GO:0051301]; chromosome segregation [GO:0007059]; positive regulation of protein dephosphorylation [GO:0035307]; regulation of chromosome segregation [GO:0051983]; regulation of mitotic nuclear division [GO:0007088] |
Human_homologues 16492807_Repo-Man forms an essential complex with protein phosphatase 1 (PP1) gamma and is required for the recruitment of PP1 to chromatin 20188555_This study reports essential DNA damage response regulation mediated by Repo-Man-protein phosphatase 1 and further delineate underlying mechanisms. 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 21514157_Repo-Man promotes H3T11ph dephosphorylation by an indirect mechanism but directly and specifically targets H3T3ph for dephosphorylation by associated PP1gamma. 21820363_This study demonistrated that identify Repo-Man as a key factor that coordinates chromatin remodeling and early events of nuclear envelope reformation during mitotic exit. 22801782_It was shown that Repo-Man and Sds22 contribute to timely Aurora B kinase substrate dephosphorylation on anaphase chromatin. 23362328_Proteomic identification of a large number of novel RepoMan interactors. 23418564_Data indicate that CDCA2 frequently is overexpressed in oral squamous cell carcinoma (OSCC) and might be associated with OSCC progression by preventing cell-cycle arrest and apoptosis. 23746640_Aurora B phosphorylates Repo-Man at S893, preventing its recruitment by histones. 27572260_Here the authors show how Ki-67 and RepoMan form mitotic exit phosphatases by recruiting PP1, how they distinguish between distinct PP1 isoforms and how the assembly of these two holoenzymes are dynamically regulated by Aurora B kinase during mitosis. 27998540_Results report that BubR1 and RepoMan bind directly to PP2A-B56 using an LSPIxE short linear motif (SLiM), where phosphorylation of the Ser residue enhances binding. RepoMan and BubR1 bind B56 using both hydrophobic and electrostatic interactions. 28091603_Repo-Man/PP1 regulates the formation of heterochromatin, dephosphorylates H3S28 and it is necessary and sufficient for heterochromatin protein 1 binding and H3K27me3 recruitment. 28423619_Inhibition of cell division cycle associated 2 (CDCA2) suppressed the proliferation of lung adenocarcinoma (LAC) cells via G1 phase arrest by downregulating cyclin E1(CCNE1), while overexpression of CDCA2 promoted LAC cells proliferation by upregulating CCNE1. 29222185_The p37 controls cortical NuMA levels via the phosphatase PP1 and its regulatory subunit Repo-Man, but it acts independently of Galphai, the kinase Aurora A, and the phosphatase PP2A. 31196027_this study shows that CDCA2 promotes the proliferation of colorectal cancer cells by activating the AKT/CCND1 pathway in vitro and in vivo 31967936_Co-regulation of the antagonistic RepoMan:Aurora-B pair in proliferating cells. 32633378_CDCA2 promotes proliferation and migration of melanoma by upregulating CCAD1. 32938714_Aurora B regulates PP1gamma-Repo-Man interactions to maintain the chromosome condensation state. 33054506_RepoMan stimulates the chromosome-dependent pathway of microtubule assembly. 34036376_CDCA2 promotes tumorigenesis and induces radioresistance in oesophageal squamous cell carcinoma cells. 34103686_CDCA2 protects against oxidative stress by promoting BRCA1-NRF2 signaling in hepatocellular carcinoma. 35386230_The Higher Expression of CDCA2 Associated with Poor Prognosis in Glioma. 35414260_Repo-Man/protein phosphatase 1 SUMOylation mediates binding to lamin A and serine 22 dephosphorylation. 35694386_Cell division cycle associated 2 (CDCA2) upregulation promotes the progression of hepatocellular carcinoma in a p53-dependant manner. 36588796_CDCA2 Promotes HCC Cells Development via AKT-mTOR Pathway. |
ENSG00000184661 |
CDCA2 |
499.608685 |
2.0502965549 |
1.035833 |
0.12891462 |
64.162748 |
0.00000000000000114554354279329200241760146673455059556512173127296172481237590545788407325744628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000308830429094290726485547351080470439188183605361270878120194538496434688568115234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
669.69133254214 |
59.9211470438216 |
326.63144798881 |
22.5198751254366 |
| ENSMUSG00000048965 |
244238 |
Mrgpre |
protein_coding |
Q91ZB7 |
FUNCTION: Orphan receptor. May regulate nociceptor function and/or development, including the sensation or modulation of pain. |
Cell membrane;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:244238; |
plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; G protein-coupled receptor signaling pathway [GO:0007186] |
18197975_The data suggests that MrgE may play a role in selective pain behavioural responses in mice. 21912971_The number of enteric neurons expressing MrgE and MrgF receptors was significantly reduced during intestinal schistosomiasis and ileitis. 34647371_Mas-related G protein-coupled receptor D is involved in modulation of murine gastrointestinal motility. |
ENSG00000184350 |
MRGPRE |
100.098604 |
0.4747027861 |
-1.074904 |
0.25104801 |
18.287489 |
0.00001899505852936240416634072758483853249344974756240844726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001334560569738229348236285831674763358023483306169509887695312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.4881204626429 |
11.2785051297039 |
135.849466969754 |
17.004156934505 |
| ENSMUSG00000048981 |
16660 |
Krt31 |
protein_coding |
Q61765 |
|
Coiled coil;Intermediate filament;Keratin;Reference proteome |
|
|
mmu:16660; |
cytoskeleton [GO:0005856]; intermediate filament [GO:0005882]; structural molecule activity [GO:0005198]; epithelial cell differentiation [GO:0030855]; intermediate filament organization [GO:0045109] |
|
ENSG00000006059 |
KRT33A |
52.563573 |
287.1429935889 |
8.165626 |
1.25182286 |
101.558206 |
0.00000000000000000000000693940829896284423921093852947998948355885437681885897454546995033464185098637244664132595062255859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000003523368906993401511081732566556640418784897647443264813671382970627909969607571838423609733581542968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
106.636197005994 |
104.583662369834 |
0.371369663850001 |
0.353893174065379 |
| ENSMUSG00000048988 |
243312 |
Elfn1 |
protein_coding |
Q8C8T7 |
FUNCTION: Postsynaptic protein that regulates circuit dynamics in the central nervous system by modulating the temporal dynamics of interneuron recruitment. Specifically present in excitatory synapses onto oriens-lacunosum molecular (OLM) interneurons and acts as a regulator of presynaptic release probability to direct the formation of highly facilitating pyramidal-OLM synapses. Inhibits phosphatase activity of protein phosphatase 1 (PP1) complexes. {ECO:0000269|PubMed:23042292}. |
Cell projection;Glycoprotein;Leucine-rich repeat;Membrane;Phosphoprotein;Protein phosphatase inhibitor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:243312; |
dendrite [GO:0030425]; excitatory synapse [GO:0060076]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; membrane [GO:0016020]; protein phosphatase inhibitor activity [GO:0004864]; synapse organization [GO:0050808] |
23042292_postsynaptic expression of Elfn1 in oriens-lacunosum moleculare interneurons regulates presynaptic release probability, which confers target-specific synaptic properties to pyramidal cell axons. 24312227_Mutation of Elfn1 in mice causes seizures and hyperactivity. 25047565_Elfn1 protein increases during postnatal development and localizes to postsynaptic sites of somatostatin-containing interneurons in the hippocampal CA1 stratum oriens and dentate gyrus hilus.Elfn1 recruits mGluR7 to synaptic sites. 30940718_A transsynaptic interaction between Elfn1 and mGluR7 constitutively reduces initial release probability onto mouse cortical SOM neurons. Elfn1 produces glutamate-independent activation of mGluR7 via presynaptic clustering, resulting in a divergence from the canonical 'autoreceptor' role of Type III mGluRs, and substantially altering synaptic pharmacology. |
ENSG00000225968 |
ELFN1 |
20.517567 |
8.3176113593 |
3.056169 |
0.76709735 |
14.588968 |
0.00013369492572153112612889924726289336831541731953620910644531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007763786155510340666885404914410173660144209861755371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.0284930902562 |
18.9580183971491 |
4.09113766205586 |
1.83460676199226 |
| ENSMUSG00000049001 |
68169 |
Ndnf |
protein_coding |
Q8C119 |
FUNCTION: Secretory protein that plays a role in various cellular processes. Acts as a chemorepellent acting on gonadotropin-releasing hormone (GnRH) expressing neurons regulating their migration to the hypothalamus (PubMed:31883645). Also promotes neuron migration, growth and survival as well as neurite outgrowth and is involved in the development of the olfactory system (PubMed:31883645). May also act through the regulation of growth factors activity and downstream signaling (By similarity). Also regulates extracellular matrix assembly and cell adhesiveness (PubMed:18757743). Promotes endothelial cell survival, vessel formation and plays an important role in the process of revascularization through NOS3-dependent mechanisms (PubMed:24706764). {ECO:0000250|UniProtKB:Q8TB73, ECO:0000269|PubMed:18757743, ECO:0000269|PubMed:24706764, ECO:0000269|PubMed:31883645}. |
Glycoprotein;Neurogenesis;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:68169; |
extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; glycosaminoglycan binding [GO:0005539]; heparin binding [GO:0008201]; angiogenesis [GO:0001525]; cellular response to fibroblast growth factor stimulus [GO:0044344]; cellular response to hypoxia [GO:0071456]; extracellular matrix organization [GO:0030198]; gonadotrophin-releasing hormone neuronal migration to the hypothalamus [GO:0021828]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of neuron apoptotic process [GO:0043524]; neuron migration [GO:0001764]; nitric oxide mediated signal transduction [GO:0007263]; peptide cross-linking via chondroitin 4-sulfate glycosaminoglycan [GO:0019800]; positive regulation of cell-substrate adhesion [GO:0010811]; positive regulation of neuron projection development [GO:0010976]; response to ischemia [GO:0002931]; vascular wound healing [GO:0061042] |
24706764_NDNF acts as an endogenous modulator that promotes endothelial cell function and ischemia-induced revascularization through eNOS-dependent mechanisms 25654972_Intramuscular administration of NDNF ameliorates post-MI remodeling and function in mice by its abilities to reduce myocyte apoptosis and promote neovascularization. 34038743_NDNF interneurons in layer 1 gain-modulate whole cortical columns according to an animal's behavioral state. 35051783_Neuron-derived neurotrophic factor protects against dexamethasone-induced skeletal muscle atrophy. |
ENSG00000173376 |
NDNF |
223.181898 |
7.4117252905 |
2.889809 |
0.19840932 |
213.969463 |
0.00000000000000000000000000000000000000000000000187016154994900619430522610934826494412035594434521667179403634676809831317209915455244906187194467870661920211365164502456681638875579665182158350944519042968750000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000002936732033899305601571476692353782554743472050173292993083468083901412080641065493439114171052776620973364542053232707427556924528744275448843836784362792968750000000000000000000000000000000000000000000000000000000 |
Yes |
No |
391.478169759581 |
54.0086394620341 |
52.8187641089478 |
6.09447739221492 |
| ENSMUSG00000049044 |
56508 |
Rapgef4 |
protein_coding |
Q9EQZ6 |
FUNCTION: Guanine nucleotide exchange factor (GEF) for RAP1A, RAP1B and RAP2A small GTPases that is activated by binding cAMP. Seems not to activate RAB3A. Involved in cAMP-dependent, PKA-independent exocytosis through interaction with RIMS2. {ECO:0000269|PubMed:11056535}. |
3D-structure;Alternative promoter usage;Alternative splicing;cAMP;cAMP-binding;Cytoplasm;Exocytosis;Guanine-nucleotide releasing factor;Membrane;Nucleotide-binding;Reference proteome;Repeat |
|
|
mmu:56508; |
apical plasma membrane [GO:0016324]; axon [GO:0030424]; basolateral plasma membrane [GO:0016323]; brush border [GO:0005903]; cilium [GO:0005929]; cone cell pedicle [GO:0044316]; cytosol [GO:0005829]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; excitatory synapse [GO:0060076]; glutamatergic synapse [GO:0098978]; growth cone [GO:0030426]; hippocampal mossy fiber to CA3 synapse [GO:0098686]; neuronal cell body [GO:0043025]; photoreceptor inner segment [GO:0001917]; photoreceptor outer segment [GO:0001750]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; protein-containing complex [GO:0032991]; cAMP binding [GO:0030552]; GTPase activator activity [GO:0005096]; guanyl-nucleotide exchange factor activity [GO:0005085]; molecular function activator activity [GO:0140677]; protein-containing complex binding [GO:0044877]; protein-macromolecule adaptor activity [GO:0030674]; small GTPase binding [GO:0031267]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; adenylate cyclase-modulating G protein-coupled receptor signaling pathway [GO:0007188]; calcium-ion regulated exocytosis [GO:0017156]; hormone secretion [GO:0046879]; insulin secretion [GO:0030073]; negative regulation of synaptic transmission [GO:0050805]; positive regulation of GTPase activity [GO:0043547]; positive regulation of insulin secretion [GO:0032024]; positive regulation of neuronal action potential [GO:1904457]; positive regulation of protein secretion [GO:0050714]; positive regulation of smooth muscle cell migration [GO:0014911]; Ras protein signal transduction [GO:0007265]; regulation of dendrite development [GO:0050773]; regulation of exocytosis [GO:0017157]; regulation of neurotransmitter receptor localization to postsynaptic specialization membrane [GO:0098696]; regulation of postsynapse organization [GO:0099175]; regulation of synaptic vesicle cycle [GO:0098693]; signal transduction [GO:0007165] |
12401793_Piccolo serves as a Ca(2+) sensor in exocytosis in pancreatic beta-cells and the formation of a cAMP-GEFII.Rim2.Piccolo complex is important in cAMP-induced insulin secretion 16452984_Mutational analysis suggests a model for cAMP-induced Epac activation with rigid body movement of the regulatory region, the features of which are universally conserved in cAMP-regulated proteins. 16644915_cAMP at least partially regulates proglucagon gene expression via the Epac-Ras/Rap-Raf-MEK-ERK signaling pathway 17077580_Northern blot analysis and in situ hybridization suggested that Epac1 and Epac2 mRNAs were transcribed in the seminiferous epithelia of the testis 18040047_These data indicate that Epac2/Rap1 signaling is essential in regulation of insulin granule dynamics by cAMP, most likely by controlling granule density near the plasma membrane. 18565730_AKAP150 acts as a key regulator in the two cAMP pathways to control PKB/Akt phosphorylation. 18660803_structure of Epac2 in complex with a cAMP analogue (Sp-cAMPS) and RAP1B by X-ray crystallography and single particle electron microscopy 18824540_Ras and cAMP regulate Epac2 function in a parallel fashion and the Ras-Epac2 interaction is required for the cAMP-dependent activation of endogenous Rap1 by Epac2. 18937323_Results are consistent with a cAMP/Epac2/Rap1-mediated signaling that could exert its action, among others, through RA-RhoGAP to promote the progression of spermatogenesis. 19167486_Cyclic AMP-dependent Epac2 signals to ERK1/2 and Akt upon activation of H-Ras, and is involved in BCR-induced growth arrest and apoptosis in WEHI-231 cells. 19170062_splicing variant Epac2B, which lacks the first cAMP-binding domain A in the N-terminus but has the second cAMP-binding domain B of Epac2A, possesses GEF activity towards Rap1, as was found for Epac2A 21041529_Phospholipase C-eta was found to be expressed in mouse beta-cells, and knockout of that gene disrupts the action of exendin-4 to facilitate calcium-induced calcium release in the beta-cells of these mice. 21478675_Phospholipase c-epsilon is a determinant of mouse islet insulin secretion that is under the control of Epac2. 21873431_analysis of autoinhibition and activation of the exchange protein directly activated by cyclic AMP (EPAC) and EPAC2 22915127_This study demonistrated that exhibited specific impairments in social and communication behavior displayed specific alterations in cortical columnar organization in the somatosensory cortex. 23363625_beta1-induced adrenergic-dependent arrhythmias are less inducible in Epac2-knockout versus wild-type mice. 23542788_there is a gut-heart GLP-1R-dependent and ANP-dependent axis that regulates blood pressure and involves Epac2 23578994_EPAC2A is required for potentiating the early dynamic increase in islet calcium levels after glucose stimulation, which is reflected in potentiated first-phase insulin secretion. 23633676_Epac2 localization is dynamically controlled by cAMP as well as by Ca(2+)-mediated activation of Ras. 23804752_data show the expression EPAC is altered in the cortex, striatum and midbrain of HPRT knockout mouse; propose that the alterations in EPAC/RAP1 signaling and cell migration in HPRT deficiency are crucial for neuro-developmental events that may contribute to neurological dysfunctions in Lesch-Nyhan syndrome 23861833_DNA methylation of alternative promoters directs tissue specific expression of Epac2 isoforms. 24150255_Data suggest that sulfonylureas stabilize Epac2A in its open, active state and provide insight for the development of drugs that target Epac2A. 25103224_Epac1 primarily is capable of inhibiting remodeling processes, whereas Epac2 primarily increases inflammatory processes in vivo 25122553_EPAC1 and EPAC2 are critical signaling intermediates in osteoclast differentiation that permit RANKL-stimulated NFkB nuclear translocation and actin rearrangements 25904804_data suggest that cAMP elevation uses an Epac2-dependent pathway to promote transmitter release, and that Epac2 is required to maintain the readily releasable pool at Mossy Fiber synapses in the hippocampus. 25985753_Epac2, but not Epac1 has a role in decreasing severity of ischemic retinopathy after retinal ischemia/reperfusion injury 26104314_cAMP-GEF II plays a key role in hippocampal functions including behavioral flexibility. 26645637_PACAP could trigger astrocytic differentiation of neural stem cells via Epac2 activation. 27598965_Epac2(-/-) mice exhibited a wide range of mood disorders, including anxiety and depression with learning and memory deficits in contextual and cued fear-conditioning tests without affecting Epac1 expression or PKA activity. 30048476_Deletion of exchange proteins directly activated by cAMP (Epac) causes defects in hippocampal signaling in female mice 30252533_Deletion of Epac1 and Epac2 decreases sodium-hydrogen exchanger type 3 expression in the proximal tubule, leading to polyuria and osmotic diuresis 31059774_EPAC2 is required for correct dendritic arborization and spine formation in vivo. Loss of EPAC2 results in an increase in inhibitory input onto neurons. 31184597_Barr1 can directly interact with Epac2; sulfonylureas such as glibenclamide promote Barr1/Epac2 complex formation, triggering enhanced Rap1 signaling and insulin secretion 31303487_When glucose metabolism induces insulin secretion, it also increases formation of Golgi-derived microtubules (GDMTs), notably with the same biphasic kinetics as insulin exocytosis. Furthermore, GDMT nucleation is controlled by a glucose signal-transduction pathway through cAMP and its effector EPAC2. 31551444_Mice depleted for Exchange Proteins Directly Activated by cAMP (Epac) exhibit irregular liver regeneration in response to partial hepatectomy. 31852500_Epac has a protecting effect on asthmatic airway inflammation and airway remodeling, and reduces airway smooth muscle cells proliferation by inhibiting SOCE. 31870404_The present data suggest that a PKA-independent cAMP signaling pathway via Epac2 may be party to adult neurogenesis. 33593853_Regulation of Mitochondrial Function by Epac2 Contributes to Acute Inflammatory Hyperalgesia. 34183057_Autophagy activity contributes to the impairment of social recognition in Epac2(-/-) mice. 34733289_Exchange Protein Directly Activated by cAMP 2 Enhances Respiratory Syncytial Virus-Induced Pulmonary Disease in Mice. 34862717_Epac-2 ameliorates spontaneous colitis in Il-10(-/-) mice by protecting the intestinal barrier and suppressing NF-kappaB/MAPK signalling. 34914636_Epac1-/- and Epac2-/- mice exhibit deficient epithelial Na+ channel regulation and impaired urinary Na+ conservation. |
ENSG00000091428 |
RAPGEF4 |
58.835046 |
0.1367394372 |
-2.870499 |
0.53984834 |
25.820836 |
0.00000037462246814928575330147183554407863681490198359824717044830322265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000035583669412376362917312892092391862775002664420753717422485351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.1280320644844 |
5.02788180272379 |
96.0076502507223 |
25.1889818338521 |
| ENSMUSG00000049134 |
18175 |
Nrap |
protein_coding |
Q80XB4 |
FUNCTION: May be involved in anchoring the terminal actin filaments in the myofibril to the membrane and in transmitting tension from the myofibrils to the extracellular matrix. {ECO:0000269|PubMed:10320340, ECO:0000269|PubMed:11732910, ECO:0000269|PubMed:9295142}. |
Actin-binding;Alternative splicing;LIM domain;Metal-binding;Phosphoprotein;Reference proteome;Repeat;Zinc |
|
|
mmu:18175; |
fascia adherens [GO:0005916]; muscle tendon junction [GO:0005927]; myofibril [GO:0030016]; Z disc [GO:0030018]; actin binding [GO:0003779]; actin filament binding [GO:0051015]; metal ion binding [GO:0046872]; muscle alpha-actinin binding [GO:0051371]; vinculin binding [GO:0017166]; actin cytoskeleton organization [GO:0030036]; cardiac muscle thin filament assembly [GO:0071691] |
12789664_Highly conserved and exclusively expressed in cardiac and skeletal muscle. Candidate cause for cardiac and skeletal myopathies 21276443_Results show that chronic NRAP overexpression in the mouse leads to right ventricular cardiomyopathy by 10 months of age. 21998026_Nrarp-deficient embryos and mice exhibit genetic background-dependent defects of the axial skeleton. In Nrarp mutant embryos, the posterior somite compartment was expanded. 30986853_Dysregulation of NRAP degradation by KLHL41 contributes to pathophysiology in nemaline myopathy. |
ENSG00000197893 |
NRAP |
24.821888 |
3.7190206133 |
1.894923 |
0.54260185 |
12.140360 |
0.00049342254429922340237812949936824225005693733692169189453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0024763796358371567042644389999850318417884409427642822265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.5042256752642 |
15.2104189970984 |
10.6222120776133 |
3.24053788039492 |
| ENSMUSG00000049420 |
77220 |
Tmem200a |
protein_coding |
Q8C817 |
|
Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:77220; |
membrane [GO:0016020] |
|
ENSG00000164484 |
TMEM200A |
28.121367 |
0.2183944999 |
-2.194992 |
0.79199256 |
7.061486 |
0.00787586478266799790148855464622101862914860248565673828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0273583305051061306811810425188014050945639610290527343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.70225921641795 |
5.02084822734088 |
39.8465127081355 |
16.5287368310844 |
| ENSMUSG00000049804 |
100503043 |
Armcx4 |
protein_coding |
E9PWM3 |
|
Membrane;Mitochondrion;Mitochondrion outer membrane;Proteomics identification;Reference proteome |
|
|
mmu:100503043; |
mitochondrial outer membrane [GO:0005741] |
32376790_Human X chromosome exome sequencing identifies BCORL1 as contributor to spermatogenesis. |
ENSG00000196440 |
ARMCX4 |
172.181559 |
0.4404573186 |
-1.182926 |
0.21180958 |
30.906113 |
0.00000002708173608163607506056047807656284032162830044399015605449676513671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003069263422585422237243548207463739174727379577234387397766113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
103.715644390723 |
15.2817956409873 |
235.472632675423 |
24.9814704321873 |
| ENSMUSG00000049939 |
192198 |
Lrrc4 |
protein_coding |
Q99PH1 |
FUNCTION: Synaptic adhesion protein. Regulates the formation of exitatory synapses through the recruitment of pre-and-postsynaptic proteins. Organize the lamina/pathway-specific differentiation of dendrites. Plays an important role for auditory synaptic responses. Involved in the suppression of glioma. {ECO:0000269|PubMed:15967442, ECO:0000269|PubMed:16980967, ECO:0000269|PubMed:17785411}. |
Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Leucine-rich repeat;Membrane;Postsynaptic cell membrane;Reference proteome;Repeat;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:192198; |
dendritic spine [GO:0043197]; excitatory synapse [GO:0060076]; glutamatergic synapse [GO:0098978]; neuron spine [GO:0044309]; plasma membrane [GO:0005886]; postsynaptic density membrane [GO:0098839]; postsynaptic membrane [GO:0045211]; Schaffer collateral - CA1 synapse [GO:0098685]; synapse [GO:0045202]; excitatory synapse assembly [GO:1904861]; modulation of chemical synaptic transmission [GO:0050804]; postsynaptic density protein 95 clustering [GO:0097119]; synapse organization [GO:0050808]; synaptic membrane adhesion [GO:0099560] |
15967442_LRRC4 may play an important role in maintaining normal function and suppressing tumorigenesis in the central nervous system. 16077900_Together, these findings provide clues to the function of LRRC4 as a negative regulator of cell growth and underscore a link between the above-mentioned cyclins, cyclin-associated molecules and tumorigenicity. 17928921_Promotes neurite extension of hippocampal neurons, and induces differentiation of glioblastoma U251 cells into astrocyte-like cells. 23177961_These results demonstrate that NGL-2 is critical for pathway-specific synapse development and functional integration of distinct inputs. 23864682_our results identify NGL-2 as a central component of pathway-specific development in the outer retina. 29553369_NGL2 protein (NGL2) promotes the formation, maintenance, and restoration of synapses in the developing and mature retina. 29949768_Mice lacking NGL-2 (Lrrc4(-/-) mice) show suppressed N-Methyl-D-aspartate receptor (NMDAR)-dependent synaptic plasticity in the hippocampus. NGL-2 associates with NMDARs through both PSD-95-dependent and -independent mechanisms. 32372061_Leucine-rich repeat containing 4 act as an autophagy inhibitor that restores sensitivity of glioblastoma to temozolomide. 33932995_LRRC4 functions as a neuron-protective role in experimental autoimmune encephalomyelitis. |
ENSG00000128594 |
LRRC4 |
11.562697 |
4.7533977332 |
2.248959 |
0.86590105 |
6.516290 |
0.01068907304039502792480931248064734973013401031494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0354255788840942276163303858993458561599254608154296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.2224205775211 |
12.2425093793223 |
4.04393271009742 |
2.08832165681605 |
| ENSMUSG00000049999 |
228966 |
Ppp1r3d |
protein_coding |
A2AJW4 |
Human_homologues FUNCTION: Seems to act as a glycogen-targeting subunit for PP1. PP1 is essential for cell division, and participates in the regulation of glycogen metabolism, muscle contractility and protein synthesis. |
Carbohydrate metabolism;Glycogen metabolism;Proteomics identification;Reference proteome |
|
|
mmu:228966; |
glycogen granule [GO:0042587]; intracellular membrane-bounded organelle [GO:0043231]; protein phosphatase type 1 complex [GO:0000164]; enzyme binding [GO:0019899]; glycogen binding [GO:2001069]; phosphoprotein phosphatase activity [GO:0004721]; protein phosphatase 1 binding [GO:0008157]; glycogen metabolic process [GO:0005977]; regulation of glycogen biosynthetic process [GO:0005979]; regulation of glycogen catabolic process [GO:0005981] |
32892347_Ppp1r3d deficiency preferentially inhibits neuronal and cardiac Lafora body formation in a mouse model of the fatal epilepsy Lafora disease. |
ENSG00000132825 |
PPP1R3D |
60.421894 |
0.3643593484 |
-1.456566 |
0.34237187 |
18.007819 |
0.00002199994749223547512080069454842856657705851830542087554931640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001522515659180674564218693767259082960663363337516784667968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.0789809599248 |
7.68445201082557 |
88.0421515197307 |
14.8645611834801 |
| ENSMUSG00000050014 |
328561 |
Apol10b |
protein_coding |
G3X9K7 |
Human_homologues FUNCTION: May affect the movement of lipids in the cytoplasm or allow the binding of lipids to organelles.; + ;FUNCTION: May play a role in lipid exchange and transport throughout the body. May participate in reverse cholesterol transport from peripheral cells to the liver (By similarity). {ECO:0000250}.; + ;FUNCTION: May affect the movement of lipids in the cytoplasm or allow the binding of lipids to organelles.; + ;FUNCTION: May play a role in lipid exchange and transport throughout the body. May participate in reverse cholesterol transport from peripheral cells to the liver. |
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
Human_homologues NA; + ;NA; + ;NA; + ;NA |
|
mmu:328561; |
extracellular space [GO:0005615]; high-density lipoprotein particle [GO:0034364]; membrane [GO:0016020]; very-low-density lipoprotein particle [GO:0034361]; chloride channel activity [GO:0005254]; lipid binding [GO:0008289]; lipid transport [GO:0006869]; lipoprotein metabolic process [GO:0042157] |
Human_homologues 11944986_APOL3 has been found only in humans and African green monkeys 20237496_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20602615_Observational study of gene-disease association. (HuGE Navigator) 20631155_An association between an single nucleotide polymorphisms within the APOL3 locus and prostate cancer risk, is revealed. 20631155_Observational study of gene-disease association. (HuGE Navigator) 28339911_A null variant in the apolipoprotein L3 gene is associated with non-diabetic nephropathy. 30423477_APOL3 regulates angiogenesis in vitro and functions as a modulator of MAPK and FAK signaling in endothelial cells. 32187552_APOL1 C-Terminal Variants May Trigger Kidney Disease through Interference with APOL3 Control of Actomyosin. 32530132_The function of apolipoproteins L (APOLs): relevance for kidney disease, neurotransmission disorders, cancer and viral infection. 34437126_A human apolipoprotein L with detergent-like activity kills intracellular pathogens.; + ;11944986_APOL4 has been found only in humans and African green monkeys 15949655_Observational study of gene-disease association. (HuGE Navigator) 18632255_An association of APOL1, 2 and 4 with schizophrenia was establised. 18632255_Observational study of gene-disease association. (HuGE Navigator); + ;11944986_APOL2 has been found only in humans and African green monkeys 15949655_Observational study of gene-disease association. (HuGE Navigator) 18632255_An association of APOL1, 2 and 4 with schizophrenia was establised. 18632255_Observational study of gene-disease association. (HuGE Navigator) 20602615_Observational study of gene-disease association. (HuGE Navigator) 20665705_Results reveal a novel function for ApoL2 in conferring anti-apoptotic ability of human bronchial epithelium to the cytotoxic effects of IFN-gamma, in maintaining airway epithelial layer integrity. 24901046_Apolipoprotein L2 contains a BH3-like domain but it does not behave as a BH3-only protein. 31466081_Epigenetic aging is accelerated in alcohol use disorder and regulated by genetic variation in APOL2. 34316015_Structures of the ApoL1 and ApoL2 N-terminal domains reveal a non-classical four-helix bundle motif.; + ;11944986_APOL1 has been found only in humans and African green monkeys 12621437_ApoL-I is the trypanosome lytic factor of normal human serum, and serum resistance associated protein confers resistance to lysis by interaction with apoL-I in the lysosome 12837283_APOL1 is a senescence-associated gene in normal human oral keratinocytes. 15500911_identified as component of trypanosome lytic factor 1 (TLF1); apoL-I alone is not sufficient for optimal trypanosome lytic activity in human TLF1. 15604524_The Apolipoprotein L-I (apoL-I) is present on a subset of HDL particles and is positively correlated with plasma triglycerides (TGs). 15949655_Observational study of gene-disease association. (HuGE Navigator) 16020735_contains a membrane pore-forming domain; APOL1 was targeted to the lysosomal membrane of Trypanosoma brucei & triggered depolarization of the membrane, continuous influx of chloride & subsequent osmotic swelling of the lysosome until the trypanosome lysed 17360487_apoL-I is responsible for the trypanolytic activity of normal human serum, whereas Hpr allows fast uptake of the carrier HDL particles in trypanolysis 17845074_Infection by Trypanosoma brucei brucei causes hemolysis that triggers activation of trypanosome lytic factor by formation of haptoglobin-related protein-hemoglobin complexes, enhancing binding, trypanolytic activity, and clearance of parasites 18505729_Apolipoprotein L1 is a novel Bcl-2 homology domain 3-only lipid-binding protein, induces autophagic cell death 18632255_An association of APOL1, 2 and 4 with schizophrenia was establised. 18632255_Observational study of gene-disease association. (HuGE Navigator) 18850207_Observational study of gene-disease association. (HuGE Navigator) 18927493_APOL1 is the first BH3-only protein with lipid binding activity that, when overproduced intracellularly, induces autophagic cell death. 19239905_Observational study of gene-disease association. (HuGE Navigator) 19239905_The Lys166Glu and Ile244Met polymorphisms in apoL-I gene are associated with TG levels in subjects with endogenous hypertriglyceridemia in Chinese. However, these polymorphisms were not associated with the risk of HTG in the population. 19324878_at low pH. Trypanosome lytic factor, apoL-1, and apoA-1 exhibit specificity for anionic membranes, whereas Hpr permeabilizes both anionic and zwitterionic membranes. 19913121_Observational study of gene-disease association. (HuGE Navigator) 20483474_Observational study of gene-disease association. (HuGE Navigator) 20483474_This study did not find DAOA significant associations with schizophrenia. Thus, APOL1 genes do not fit the antagonistic pleiotropy model. 20602615_Observational study of gene-disease association. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 20635188_Coding region mutations in the APOL1 gene are highly associated with end stage kidney disease in African and Hispanic Americans. 20635188_missense mutations with predicted functional effects in the APOL1 gene are significantly more associated with ESKD than all previously reported SNPs in MYH9 20647424_Observational study of gene-disease association. (HuGE Navigator) 20647424_in African Americans, focal segmental glomerulosclerosis and hypertension-attributed end-stage kidney disease are associated with 2 independent sequence variants in APOL1 gene; only kidney disease-associated ApoL1 variants lysed Trypanosoma rhodesiense 20668430_A risk allele for focal segmental glomerulosclerosis in African Americans is located within a region containing APOL1 and MYH9. 20668430_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 20805508_show that trypanosome lytic factor-1 resistance in Trypanosoma brucei brucei is caused by reduced expression of the Hp/Hb receptor gene. 20855565_Observational study of gene-disease association. (HuGE Navigator) 21080072_Susceptibility of African-Americans to nondiabetic chronic kidney disease is likely due to functional variants of APOL1 that have been selected for because of their ability to protect from parasital infection 21486385_APOL1 genotype has a role in failure of kidney after renal transplantation in African American deceased donors 21537348_The powerful evolutionary selection pressure of an infectious pathogen in West Africa favored the spread of APOL1 variants that protect against a lethal form of African sleeping sickness but are highly associated with an increased risk of kidney disease 21698141_type 2 diabetic nephropathy-associated FRMD3 SNPs were detectable in African Americans only after accounting for MYH9, with differential effects for APOL1 21910715_Data provide insight into the pathobiology of renal dysfunction in SCD, suggesting that MYH9 and APOL1 are both associated with risk. 21931123_Coding variants in the apolipoprotein L1 gene are strongly associated with non-diabetic nephropathy in African Americans. 21968148_The coinciding absence of HIVAN and the APOL1 risk variants among HIV-infected individuals of Ethiopian ancestry support a Western rather than Pan-African ancestry risk for end-stage kidney disease. 21997392_Comparing the renal distribution of APOL1 in nondiabetic kidney disease to normal kidney suggests that a previously unrecognized arteriopathy may contribute to disease pathogenesis in patients of African ancestry. 21997394_African Americans carrying two APOL1 risk alleles have a greatly increased risk for glomerular disease, and APOL1-associated focal segmental glomerulosclerosis occurs earlier and progresses to kidney failure more rapidly. 21997396_Our data suggest that more than 3 million African Americans likely have the high-risk genotype and are at markedly increased risk for nondiabetic chronic kidney disease. 21997397_These data further support the strong association of genetic variants in APOL1 with susceptibility to focal segmental glomerulosclerosis and HIV nephropathy among African Americans. 21997398_Genetic variations in APOL1 identify African Americans that initiate chronic hemodialysis at a younger age. 22119407_A genome-wide association study provides additional and independent evidence that APOL1 variants contribute to nondiabetic nephropathy in African Americans. 22135313_an association between APOL1 variants and renal outcomes in non-HIVAN kidney disease, suggesting a possible use for APOL1 genotyping to help guide the care of HIV-infected patients. 22239288_The current study has provided evidence that genetic variations of polymorphic sites in apoL-I gene might affect plasma TG variability in nonobese Chinese subjects, but are not associated with obesity in the population. 22357707_APOL1 allelic variants are associated with lower age of dialysis initiation and thereby increased dialysis vintage in African and Hispanic Americans with non-diabetic end-stage kidney disease. 22487534_conclude that APOL1 genotypes do not increase risk of allograft loss after kidney transplantations, and carrying 2 APOL1 risk alleles should not be an impediment to transplantation 22495294_There were no differences in the pathological findings of HIV-associated nephropathy and the number of APOL1 risk alleles. 22569246_discussion of role of ApoL1 in renal cell carcinoma and chronic kidney disease: Intracellularly, elevated ApoL1 can induce autophagy and autophagy-associated cell death, which may be critical in maintenance of cellular homeostasis in kidney. [REVIEW] 22691369_APOL1 expression is likely induced by bloodborne Trypanosoma brucei gambiense, but is not related to resistance/susceptibility in its human host. 22695330_Report association of APOL1 variants with mild kidney disease in the first-degree relatives of African American patients with non-diabetic end-stage renal disease. 22832513_Apolipoprotein L1 gene variants associate with hypertension-attributed nephropathy and the rate of kidney function decline in African Americans. 22878977_review focuses on the population genetics of non-diabetic chronic kidney disease with an emphasis on recent discoveries pertaining to the chromosome 22q12.3 region, containing the APOL1 gene as a major locus for chronic kidney disease risk 22956460_APOL1 risk variants are associated with non-diabetic forms of CKD among Nigerians of Yoruba ethnicity 23082501_evidence of natural selection of these apoL1 mutations despite their deleterious potential for kidneys highlights the importance of the resistance to trypanosomes in the evolution of Man. [lecture] 23107266_APOL1 protein genetic variants are risk for kidney transplantation living donors in african americans. 23300552_APOL1 null alleles do not correlate with glomerulosclerosis. 23438974_Arguing by exclusion, it is reasonable to conclude that the putative APOL1 causal variants are not proxies for any other variants with more direct roles in kidney disease. 23520206_APOL1 genotyping of African-American patients with systemic lupus erythematosus (SLE) might help identify patients at risk for collapsing glomerulopathy. 23677244_African Americans at increased risk for APOL1-associated nephropathy (two APOL1 risk variants) with JC viruria had a lower prevalence of kidney disease, suggesting that JCV interaction with APOL1 genotype may influence kidney disease risk. 23715117_Host APOL1 genotype is independently associated with proteinuria in HIV infection. 23748364_Variation in the donor apolipoprotein L1 (APOL1), caveolin 1 (CAV1), and multi-drug resistance 1 encoding P-glycoprotein genes (ABCB1) are all associated with graft survival after kidney transplantation. [Review] 23766536_APOL1 variants associate with increased risk of CKD among African Americans. 23768513_High levels of nonsynonymous polymorphism in the APOL1 regions encoding the functional domains that are required for lysing parasites are observed in humans with exposure to two Trypanosoma brucei subspecies that cause human African trypanosomiasis. 23860441_APOL1 risk variants are common in the Igbo population of south-eastern Nigeria, and are also highly associated with non-diabetic chronic kidney disease (CKD) in this area; APOL1 may explain the increased prevalence of CKD in this region 24011553_Possible genetic association link between keloids, non-diabetic kidney disease, and the APOL1-MYH9 haplotype amongst humans of African descent. 24119848_data support an important role for APOL1 in the progression of diverse etiologies of kidney disease, in concert with requisite environmental (gene*environment) and inherited (gene*gene) interactions [review] 24157943_Variants in NPHS2, SDCCAG8 and near BMP4 appear to interact with APOL1 to modulate the risk for non-diabetic end stage kidney disease in african americans. 24173214_finding supports the hypothesis that induction of APOL1 contributes to HIV-1 suppression in differentiated monocytes 24206458_Renal risk variants in APOL1 were associated with the higher rates of end-stage renal disease and progression of chronic kidney disease that were observed in black patients as compared with white patients, regardless of diabetes status. 24231663_The function of variant APOL1 proteins derived from circulation or synthesized in the kidney, but not the level of circulating APOL1, probably mediates APOL1-associated kidney disease in HIV-infected African Americans. 24233469_review of the biology of APOL1 present in the circulation and localized to the kidney as it may contribute to the pathogenesis of APOL1-associated kidney disease 24379297_APOL1 variants contribute to atherosclerotic CVD risk, indicating a genetic component to cardiovascular health disparities in individuals of African ancestry. 24504811_End-stage renal disease in African Americans with lupus nephritis is associated with APOL1. 24518129_APOL1 genotyping revealed that both donor and recipient were heterozygous for the G1 and G2 alleles. 24658608_APOL1 gene polymorphisms do not confer an increased risk of chronic kidney disease in lupus nephritis patients. 24731740_The presence of 2 APOL1 risk alleles confers a strong risk for the development of PLA2R-associated collapsing glomerulopathy in African Americans 24808134_analysis of genetic variants of human APOL1 that protect against T. brucei rhodesiense as well as evolution of this trypanolytic factor 24899058_APOL1 risk variants enhance podocyte necrosis through compromising lysosomal membrane permeability. 25029429_APOL1 risk variants were not significantly associated with prevalent cardiovascular disease 25054777_APOL1 nephropathy variants associate with lower levels of subclinical atherosclerosis and reduced risk of death in African Americans with type 2 diabetes mellitus 25081748_These findings support involvement of multiple cell types in subnephrotic forms of APOL1-associated nephropathy, particularly renal tubule cells with resultant tubulointerstitial disease 25100047_interferons and the molecular pattern recognition receptors that stimulate interferon production may contribute to APOL1-associated kidney disease 25168832_update on the spectrum of APOL1 kidney disease and on the worldwide distribution of these kidney risk variants[review] 25249559_study concludes that variation in APOL1 G3 makes a nominal, if any, contribution to end-stage renal disease in African Americans; G1 and G2 variants explain the vast majority of nondiabetic nephropathy susceptibility 25349204_African ancestry was significantly was associated with the genetic susceptibility of chronic kidney diseases. 25530085_Two APOL1 risk alleles in young hypertensive African Americans with a family history of ESRD are strongly associated with kidney disease. 25573908_APOL1 risk genotypes did not affect response to drug therapy with patients diagnosed FSGS. 25730870_These data are consistent with a model of lysis that involves endocytic recycling of APOL1 and the formation of cation-selective channels, at neutral pH, in the parasite plasma membrane. 25788523_HIV-positive, antiretroviral therapy-naive South-African blacks with two APOL1 risk alleles are at very high risk for developing HIV-associated nephropathy 25809272_Apolipoprotein L1 gene variants in deceased organ donors are associated with renal allograft failure. 25853332_This review provides an update on the biological functions for circulating (trypanosome resistance) and intracellular (emerging role for autophagy) APOL1. 25887069_Higher levels of factor VIIc, VIIIc, fibrinogen, vonWillebrand factor, and protein C were associated with ESRD risk, with a significantly stronger association of factor VIIIc and protein C in African Americans with two APOL1 risk alleles. 25921719_Among HIV-infected African American women, APOL1-associated kidney injury appears to localize to the glomerulus, rather than the tubules. 25924622_Identify exon 4 as a major determinant of APOL1 podocyte cytotoxicity. 25933006_The presence of APOL1 duplication was observed in 4.06%of cases with kidney diseases versus 0.78% in controls, indicating that this APOL1 duplication may alter susceptibility to kidney disease in African Americans. 25993319_Only APOL1 G1 and G2 confer renal risk, and other common and rare APOL1 missense variants, including the archaic G3 haplotype, do not contribute to sporadic FSGS and HIVAN in the US population. 26025194_The relationship between HDLC and eGFR is strongly influenced by the APOL1 rs73885319 kidney risk genotype. 26038529_These findings indicate that APOL1 risk alleles are associated with exaggerated age-related nephron loss. 26089538_basic biology and trafficking behavior of circulating APOL1 from the liver 26091559_Our study not only reveals the contribution of each domain of the APOL1 protein to cell injury, but also highlights some potential suggested targets for drug design to prevent or treat APOL1-associated nephropathy. 26108665_Expression of APOL1 increased ion permeability and caused profound morphological deterioration. 26112018_study does provide an indication that APOL1 variants may play a role in conferring an increased risk for renal and cardiovascular risk in this population. 26147622_concordant with the genetic interaction observed in sickle cell disease patients, APOL1 G2 reduces myh9 expression in vivo, suggesting a possible interaction between the altered APOL1 and myh9. 26150607_we have shown that individuals with the high risk APOL1 genotype enrolled in the NEPTUNE study present, from a clinical and histologic perspective, with more advanced disease and have less remission of proteinuria over time. 26152403_APOL1 single nucleotide polymorphisms are associated with nephropathy. 26180129_blacks with two APOL1 risk alleles had the highest risk for albuminuria and eGFRcys decline in young adulthood, whereas disparities between low-risk blacks and whites were related to differences in traditional risk factors. 26307671_Trypanolysis by APOL1 is linked to apoptosis-like mitochondrial membrane permabilization occurring together with TbKIFC1-mediated transport of APOL1 from endolysosomal membranes to the mitochondrion. 26430087_Sociodemographic factors and common risk factors for chronic kidney disease progression do not seem to alter APOL1-related CKD progression. 26566060_Shorter renal allograft survival is reproducibly observed after deceased donor kidney transplantation from APOL1 2-renal-risk-variant African American donors. 26586272_We examined whether APOL1 G1 and G2 renal-risk variant serum concentrations or lipoprotein distributions differed from nonrisk G0 APOL1 in African Americans without nephropathy. 26634651_APOL1 genotype is associated with albuminuria, subclinical atherosclerosis, incident myocardial infarction, and mortality in older African Americans. 26668025_APOL1 genotype may provide additional diagnostic information to traditional clinical variables in predicting underlying FSGS spectrum lesions in blacks who are HIV positive. 26699492_Expression of either G1 or G2 APOL1 variants increased cell swelling and death vs G0-expressing cells. This was preceded by G1 or G2 APOL1-induced net efflux of intracellular potassium, resulting in the activation of stress-activated protein kinases. 26940095_GSTM1 null and APOL1 high-risk alleles deleteriously affect chronic kidney disease progression among blacks with hypertension, and subjects with both GSTM1 null and APOL1 high-risk genotypes had highest risk of adverse renal outcomes 26966015_Among blacks, the APOL1 high-risk genotype associated only with higher risk of end-stage renal disease in a fully adjusted analysis. Black race and APOL1 high-risk status were associated with faster eGFR decline 27026370_Overall, our results suggest podocyte depletion could predispose individuals with APOL1 risk genotypes to kidney disease in response to a second stressor, and add to other published evidence associating APOL1 expression with preeclampsia 27035887_We report an unadjusted incidence of 1.2 CKD cases/100 person-years (95% CI: 0.5 to 2.5) in PHIV youth carrying APOL1 high-risk genotypes, with important implications for sub-Saharan Africa 27054572_APOL1 G1 and G2 genotypes in deceased African American liver donors have minimal effects on liver transplant outcomes. 27073096_The homozygous N264K ApoL1 variant may be at increased risk of contracting human African trypanosomiasis. 27112635_The proteomic profile of apoL1 is modified in HDLs of high cardiovascular risk patients, and apoL1 plasma levels are significantly lower in serum and in HDL3 of patients that will suffer an adverse cardiac event within 3 years. 27157405_We show that the levels of one member of the family, apolipoprotein L1 (apoL1) is higher in papillary thyroid carcinoma compared to normal tissue. 27157696_Two APOL1 renal-risk variants are associated with longer dialysis survival in African Americans with non-diabetic end-stage renal disease. 27230965_Among patients with CKD attributed to hypertension, those with the APOL1 high-risk genotype were more likely to experience a steady decline trajectory in eGFR than those without the APOL1 high-risk genotype. These findings suggest a persistent underlying pathophysiologic process in those patients with the APOL1 high-risk genotype. 27588375_APOL1 variant is associated with End-Stage Renal Disease. 27599995_Our findings indicate that the presence of risk disease risk variants of APOL1 is permissive of HIV-1 persistence in human podocytes in synergy with IL-1beta, a cytokine that characterizes the inflammatory milieu of acute and chronic phases of HIV-1 infection. 27633872_Letter: no APOL1 risk allele variants in Indian patients with chronic kidney disease. 27638911_Genetic variant in apolipoprotein L1 is not associated with preterm birth in African American population. 27658436_APOL1, alpha-thalassemia, and BCL11A variants as a genetic risk profile for progression of chronic kidney disease in sickle cell anemia. 27821631_Results suggest a pivotal role for mitochondrial dysfunction in APOL1-associated kidney disease 27862962_APOL1 Genetic Variations are associated with acute rejection. 27864430_This new Drosophila model uncovers a novel mechanism by which upregulated expression of APOL1-G1 could precipitate renal disease in humans. 27870653_Apolipoprotein L1 and apolipoprotein A-IV and their association with kidney function 27927600_Strict blood pressure control during chronic kidney disease associates with a lower risk of death in blacks with the high-risk CKD APOL1 genotype. 27997071_APOL1 Gene variation is associated with end-stage renal disease. 28143671_Relationships between APOL1 kidney risk variants and cardiovascular disease (CVD) susceptibility and CVD-related death remain controversial. Some studies detected an increased risk for CVD, whereas others support protection from death and subclinical CVD and cerebrovascular disease 28218918_mice with podocyte-specific expression of either APOL1 risk allele (G1 or G2), but not of the G0 allele, develop functional (albuminuria and azotemia), structural (foot-process effacement and glomerulosclerosis) and molecular (gene-expression) changes that closely resemble human kidney disease. Expression of the risk-variant APOL1 alleles interferes with endosomal trafficking and blocks autophagic flux. 28265848_Divergent intracellular biological pathways of ancestral and variant APOL1 may explain a worsened prognosis as demonstrated in Systemic Lupus Erythematosus. 28314584_Association of chronic kidney disease with APOL1 risk alleles was not identified in Aboriginal people in remote areas of Australia. 28335839_risk alleles are associated with higher systolic blood pressure and earlier hypertension diagnoses in young African Americans 28385815_The enhanced expression of GRP78 by podocytes expressing APOL1 variants would indicate endoplasmic reticulum (ER) stress. 28494221_APOL1 copy number variations may be not associated with susceptibility to focal segmental glomerulosclerosis in Chinese population. 28537557_These results implicate both forms of human African trypanosomiasis in the selection and persistence of otherwise detrimental APOL1 kidney disease variants. 28545715_APOL1 variants are not associated with longitudinal blood pressure in blacks. 28572159_Among African Americans with hypertension-attributed chronic kidney disease, APOL1 risk variants were not associated with an overall risk for cardiovascular disease although some signals for cardiovascular mortality were noted. 28650456_The synergy of circulating factor suPAR and APOL1 G1 or G2 on alphavbeta3 integrin activation is a mechanism for CKD. 28699644_Roles of APOL1 G1 and G2 variants in sickle cell disease patients: kidney is the main target. 28732083_The frequency of APOL1 risk variants ranged from 7.0% to 11.0%. 28918394_Data suggest that APOL1 confers chloride-selective permeability to preformed phospholipid vesicles; this selectivity is strongly pH-sensitive, with maximal activity at pH 5 and little activity above pH 7; APOL1 permease activity requires calcium ions; APOL1 stably associates with phospholipid vesicles, requiring low pH and presence of negatively charged phospholipids for maximal binding. 28975602_This is the first report of a specific association of APOL1 with small vessel disease (SVD) ischemic stroke. 29042080_APOL1 risk variants did not associate with subclinical markers of atherosclerosis or left ventricular hypertrophy in middle-aged black adults with preserved kidney function. 29051146_Among blacks with established moderate CKD, the APOL1 high-risk variants are associated with greater risk of incident proteinuria. After proteinuria onset, kidney function declines more rapidly but does not differ by APOL1 risk status. 29110756_In individuals at risk for nondiabetic kidney disease based on African ancestry, the majority of that risk can be explained by two variants in the APOL1 gene. 29110757_Considering APOL1 evolution also may help us understand how APOL1 risk variants cause kidney disease in modern humans. 29110758_APOL1 variants are associated with HIV-associated nephropathy(HIVAN), a podocyte disease,but not with HIV-immune complex disease, primarily a disease of the mesangium. 29110759_the associations of the APOL1 risk variants with microalbuminuria, incident CKD, and subsequent kidney function decline implicate a potential role in both the development and progression of CKD. Unlike the consistent association with CKD, the association of the APOL1 risk variants with cardiovascular disease is less clear. 29110760_The presence of two APOL1 renal risk variants in deceased donors shortens survival of their renal allografts. No study has examined the potential interaction of APOL1 genotype of the donors, deceased or living, and recipients. 29110762_expression of G1 or G2 APOL1 results in significantly more cell death compared with wild-type APOL1 (G0) in various human cells in culture 29113983_Novel relationships were identified between higher plasma FGF23 concentrations and absence of APOL1 renal-risk genotypes with higher mortality in African Americans with diabetes. 29180397_Our results indicate that APOL1-mediated pore formation is critical for the trypanolytic activity of APOL1 and drives APOL1-mediated cytotoxicity in overexpression systems. 29269352_Among blacks without baseline CVD, the APOL1 high-risk variants may be associated with increased risk for incident heart failure but not subclinical CVD or incident clinical atherosclerotic CVD. 29357411_ApoL1 variants regulate monocyte differentiation into macrophages and eicosanoid metabolism via enhanced COX-2 expression. 29357419_Modulation of the APOL1-miR193a axis carries a potential to preserve podocyte molecular phenotype in high glucose milieu. 29406442_Review of the role of APOL1 in kidney disease in children and young adults of African ancestry. APOL1 explains almost 70% of the excess risk of kidney disease in those of African descent, and is common in children with glomerular disease. 29420808_lacking APOL1 renal-risk genotypes, JC polyomavirus was detected in 12.2% of cases and 48.8% of controls 29470556_study found strong evidence for no association with Trypanosoma brucei rhodesiense Human African trypanosomiasis and APOL1 G2 in two Ugandan populations 29531077_APOL1 Risk Variant is associated with focal segmental glomerulosclerosis. 29685497_Plasma concentrations of TNFR1, TNFR2, and KIM1 are independently associated with renal outcome and improve discrimination or reclassification of African ancestry individuals with a high-risk APOL1 genotype and preserve renal function. 29880816_data showing no cytotoxicity of overexpressed APOL1 isoform lacking exon 4, we propose that morpholino-induced APOL1 isoform switch may provide a new tool to identify in vivo molecular mechanism(s) by which risk alleles promote or mediate the kidney disease phenotype. 29885931_Individual genome-wide association studies and meta-analysis results of 2650 end-stage renal disease (ESRD) cases and 1656 controls African American patients did not detect significant genome-wide associations with ESRD beyond APOL1. Similarly, no single nucleotide polymorphism showed significant genome-wide evidence of an interaction with APOL1 risk variants. [meta-analysis] 29899045_APOL1 high-risk status is associated with CVD events in community-dwelling Black adults without diabetes mellitus. 29967295_APOL1 risk variants contribute to podocyte injury through enhanced endoplasmic reticulum stress. The study provides insight into new mechanisms involved in APOL1 risk variants-induced podocyte damage, and highlights some new therapeutic targets for APOL1-associated nephropathy. 29971324_Status as a carrier of a high-risk APOL1 genotype was associated with HFpEF hospitalization among postmenopausal women, which is partly accounted for by baseline kidney function. These findings do not support an association of high-risk APOL1 genotypes with coronary heart disease, stroke, or mortality in postmenopausal African American women. 29992269_APOL1 mutation is associated with steroid-resistant nephrotic syndrome and/or focal segmental glomerulosclerosis patients of different African ancestries. 30052698_There are striking differences in chronic kidney disease between Caucasians and African descendants. It was widely accepted that this occurred due to socioeconomic factors, but recent studies show that apolipoprotein L-1 (APOL1) gene variants are strongly associated with focal segmental glomerulosclerosis, HIV-associated nephropathy, hypertensive nephrosclerosis, and lupus nephritis in the African American population 30173819_Fetal APOL1 high-risk genotype increases the risk for preeclampsia, likely by adversely affecting placental function. 30247453_APOL1 genotype may not be sufficient to explain discrepancies in postdonation kidney function between caucasian donors and low-risk APOL1 genotype living kidney donors of African ancestry. 30287079_Donor APOL1 high-risk genotypes are associated with inferior graft survival and de novo collapsing focal segmental glomerulosclerosis with high proteinuria after kidney transplantation. 30369398_Apolipoprotein L1 risk variants are present in black South Africans with similar frequencies between chronic kidney disease patients, first-degree relatives, |
ENSG00000128284+ENSG00000100336+ENSG00000128335+ENSG00000100342 |
APOL3+APOL4+APOL2+APOL1 |
39.226649 |
4.2729387382 |
2.095229 |
0.45911432 |
20.623411 |
0.00000559082602807457180831268023224467356158129405230283737182617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000433602148979795703727727063103714044700609520077705383300781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.3424870712094 |
19.2051554158625 |
13.8879798439845 |
3.62025635466099 |
| ENSMUSG00000050107 |
14841 |
Haspin |
protein_coding |
A0A0R4J0P2 |
Human_homologues FUNCTION: Serine/threonine-protein kinase that phosphorylates histone H3 at 'Thr-3' (H3T3ph) during mitosis. May act through H3T3ph to both position and modulate activation of AURKB and other components of the chromosomal passenger complex (CPC) at centromeres to ensure proper chromatid cohesion, metaphase alignment and normal progression through the cell cycle. {ECO:0000269|PubMed:11228240, ECO:0000269|PubMed:15681610, ECO:0000269|PubMed:17084365, ECO:0000269|PubMed:20705812, ECO:0000269|PubMed:20929775}. |
ATP-binding;Kinase;Nucleotide-binding;Proteomics identification;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:14841; |
centrosome [GO:0005813]; nucleoplasm [GO:0005654]; spindle [GO:0005819]; ATP binding [GO:0005524]; histone H3T3 kinase activity [GO:0072354]; mitotic sister chromatid cohesion [GO:0007064]; mitotic spindle assembly checkpoint signaling [GO:0007094]; protein localization to chromosome, centromeric region [GO:0071459]; protein phosphorylation [GO:0006468] |
17123944_the 193-bp DNA sequence in Haspin promotor is sufficient for the specific, bidirectional, and synchronous expression in germ cells in the testis. 21606629_the tissue-dependent and differentially methylated region functions as a modulator in a DNA methylation-dependent manner; Gsg2 is under epigenetic control 25315835_Data implicate haspin as a regulator of the chromosomal passenger complex and chromosome segregation during meiosis I. 27562071_These data uncover a role for haspin as a regulator of bipolar spindle assembly by regulating AURKC function at acentriolar microtubule-organizing centers in oocytes. 34615946_Distinct roles of haspin in stem cell division and male gametogenesis. 35694956_Haspin participates in AURKB recruitment to centromeres and contributes to chromosome congression in male mouse meiosis. |
ENSG00000177602 |
HASPIN |
137.676166 |
2.0660054133 |
1.046844 |
0.22777627 |
21.061555 |
0.00000444762708983353452257998583707632178629864938557147979736328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000351563341112321774075029356954047443650779314339160919189453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
188.296496807132 |
29.8523794913571 |
91.1403695222269 |
11.1399223660497 |
| ENSMUSG00000050222 |
239114 |
Il17d |
protein_coding |
A0A0B4J1G4 |
Human_homologues FUNCTION: Induces expression of IL6, CXCL8/IL8, and CSF2/GM-CSF from endothelial cells. {ECO:0000269|PubMed:12097364}. |
Cytokine;Disulfide bond;Reference proteome;Secreted;Signal |
|
|
mmu:239114; |
extracellular space [GO:0005615]; cytokine activity [GO:0005125]; protein homodimerization activity [GO:0042803]; receptor ligand activity [GO:0048018]; inflammatory response [GO:0006954]; negative regulation of hemopoiesis [GO:1903707]; positive regulation of cytokine production involved in inflammatory response [GO:1900017]; positive regulation of granulocyte macrophage colony-stimulating factor production [GO:0032725]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of interleukin-8 production [GO:0032757] |
27545889_Nrf2 Induces IL-17D to mediate tumor and virus surveillance. 27940089_a new role for nuclear factor erythroid-derived 2-like 2 (Nrf2) in regulating early anti-cancer immune responses by inducing IL-17D and recruiting natural killer (NK) cells. 30209334_Mice deficient in IL-17D or the transcription factor Nuclear factor (erythroid-derived 2)-like 2 (Nrf2), an inducer of IL-17D, featured an early decreased number of innate immune cells at the point of viral entry and were more susceptible to mouse cytomegalovirus infection. |
ENSG00000172458 |
IL17D |
43.534952 |
0.3447976812 |
-1.536178 |
0.42928991 |
12.696460 |
0.00036634851428447569266985683889004121738253161311149597167968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0018973888369878048713867979557790022226981818675994873046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
23.1468031393987 |
6.98967982570975 |
67.1315510535894 |
14.2408935365176 |
| ENSMUSG00000050295 |
17300 |
Foxc1 |
protein_coding |
Q61572 |
FUNCTION: DNA-binding transcriptional factor that plays a role in a broad range of cellular and developmental processes such as eye, bones, cardiovascular, kidney and skin development (PubMed:9635428, PubMed:9106663, PubMed:10479458, PubMed:10395790, PubMed:11562355, PubMed:18187037, PubMed:19668217, PubMed:22493429, PubMed:24590069, PubMed:25808752, PubMed:28223138). Acts either as a transcriptional activator or repressor (PubMed:28223138). Binds to the consensus binding site 5'-[G/C][A/T]AAA[T/C]AA[A/C]-3' in promoter of target genes (PubMed:25808752). Upon DNA-binding, promotes DNA bending. Acts as a transcriptional coactivator (PubMed:25808752). Stimulates Indian hedgehog (Ihh)-induced target gene expression mediated by the transcription factor GLI2, and hence regulates endochondral ossification (PubMed:25808752). Acts also as a transcriptional coregulator by increasing DNA-binding capacity of GLI2 in breast cancer cells. Regulates FOXO1 through binding to a conserved element, 5'-GTAAACAAA-3' in its promoter region, implicating FOXC1 as an important regulator of cell viability and resistance to oxidative stress in the eye (By similarity). Cooperates with transcription factor FOXC2 in regulating expression of genes that maintain podocyte integrity (PubMed:28223138). Promotes cell growth inhibition by stopping the cell cycle in the G1 phase through TGFB1-mediated signals. Involved in epithelial-mesenchymal transition (EMT) induction by increasing cell proliferation, migration and invasion (By similarity). Involved in chemokine CXCL12-induced endothelial cell migration through the control of CXCR4 expression (PubMed:18187037). Plays a role in the gene regulatory network essential for epidermal keratinocyte terminal differentiation (By similarity). Essential developmental transcriptional factor required for mesoderm-derived tissues formation, such as the somites, skin, bone and cartilage (PubMed:9106663, PubMed:10479458, PubMed:10395790, PubMed:10704385, PubMed:11562355, PubMed:15196959). Positively regulates CXCL12 and stem cell factor expression in bone marrow mesenchymal progenitor cells, and hence plays a role in the development and maintenance of mesenchymal niches for haematopoietic stem and progenitor cells (HSPC) (PubMed:24590069). Plays a role in corneal transparency by preventing both blood vessel and lymphatic vessel growth during embryonic development in a VEGF-dependent manner (PubMed:22171010). May function as a tumor suppressor (By similarity). {ECO:0000250|UniProtKB:Q12948, ECO:0000269|PubMed:10395790, ECO:0000269|PubMed:10479458, ECO:0000269|PubMed:10704385, ECO:0000269|PubMed:11562355, ECO:0000269|PubMed:15196959, ECO:0000269|PubMed:18187037, ECO:0000269|PubMed:19668217, ECO:0000269|PubMed:22171010, ECO:0000269|PubMed:22493429, ECO:0000269|PubMed:24590069, ECO:0000269|PubMed:25808752, ECO:0000269|PubMed:28223138, ECO:0000269|PubMed:9106663, ECO:0000269|PubMed:9635428}. |
Activator;Angiogenesis;Developmental protein;DNA-binding;Nucleus;Phosphoprotein;Reference proteome;Repressor;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:17300; |
cytosol [GO:0005829]; heterochromatin [GO:0000792]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA binding, bending [GO:0008301]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; promoter-specific chromatin binding [GO:1990841]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; RNA polymerase II-specific DNA-binding transcription factor binding [GO:0061629]; sequence-specific DNA binding [GO:0043565]; transcription cis-regulatory region binding [GO:0000976]; anatomical structure morphogenesis [GO:0009653]; angiogenesis [GO:0001525]; apoptotic process involved in outflow tract morphogenesis [GO:0003275]; artery morphogenesis [GO:0048844]; blood vessel development [GO:0001568]; blood vessel diameter maintenance [GO:0097746]; blood vessel remodeling [GO:0001974]; brain development [GO:0007420]; camera-type eye development [GO:0043010]; cardiac muscle cell proliferation [GO:0060038]; cell differentiation [GO:0030154]; cell migration [GO:0016477]; cell population proliferation [GO:0008283]; cellular response to chemokine [GO:1990869]; cellular response to epidermal growth factor stimulus [GO:0071364]; cerebellum development [GO:0021549]; chemokine-mediated signaling pathway [GO:0070098]; collagen fibril organization [GO:0030199]; embryonic heart tube development [GO:0035050]; endochondral ossification [GO:0001958]; eye development [GO:0001654]; germ cell migration [GO:0008354]; glomerular epithelium development [GO:0072010]; glycosaminoglycan metabolic process [GO:0030203]; heart development [GO:0007507]; heart morphogenesis [GO:0003007]; in utero embryonic development [GO:0001701]; kidney development [GO:0001822]; lacrimal gland development [GO:0032808]; lymph vessel development [GO:0001945]; maintenance of lens transparency [GO:0036438]; mesenchymal cell development [GO:0014031]; mesenchymal cell differentiation [GO:0048762]; negative regulation of angiogenesis [GO:0016525]; negative regulation of apoptotic process involved in outflow tract morphogenesis [GO:1902257]; negative regulation of lymphangiogenesis [GO:1901491]; negative regulation of mitotic cell cycle [GO:0045930]; negative regulation of transcription by RNA polymerase II [GO:0000122]; neural crest cell development [GO:0014032]; Notch signaling pathway [GO:0007219]; odontogenesis of dentin-containing tooth [GO:0042475]; ossification [GO:0001503]; ovarian follicle development [GO:0001541]; paraxial mesoderm formation [GO:0048341]; positive regulation of core promoter binding [GO:1904798]; positive regulation of DNA binding [GO:0043388]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of epithelial to mesenchymal transition [GO:0010718]; positive regulation of gene expression [GO:0010628]; positive regulation of hematopoietic progenitor cell differentiation [GO:1901534]; positive regulation of hematopoietic stem cell differentiation [GO:1902038]; positive regulation of keratinocyte differentiation [GO:0045618]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of organ growth [GO:0046620]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal system development [GO:0001501]; somitogenesis [GO:0001756]; ureteric bud development [GO:0001657]; vascular endothelial growth factor receptor signaling pathway [GO:0048010]; vascular endothelial growth factor signaling pathway [GO:0038084]; ventricular cardiac muscle tissue morphogenesis [GO:0055010] |
14512019_Progression of calvarial bone development requires Foxc1 regulation of Msx2 and Alx4. 15906377_FGF2 regulates the expression of Foxc1, indicating that Foxc1 integrates Bmp and Fgf signalling pathways 16412416_alongside its previously known roles in kidney, cardiovascular and eye development, Foxc1 has essential functions during at least two stages of gonad development-germ cell migration and folliculogenesis 16470615_Foxc1 mediates the bone morphogenetic protein signaling required for lacrimal gland development. 16678147_Results demonstrate that Foxc1 and 2 transcription factors are novel regulators of arterial cell specification upstream of Notch signaling and lymphatic sprouting during embryonic development. 16759280_Analysis of the regulation of Tgf beta1i4 by Foxc1 in mesenchymal cells isolated from the periocular region revealed that Foxc1 negatively regulated Tgf beta1i4 expression, presumably via secreted factors such as TGF-beta1. 16839542_Foxc1 is required for the development of the second heart field where the outflow tract, right ventricle, and part of the atrium and inflow tract are derived from a second cell lineage encompassing the secondary heart field. 17715063_defects in meningeal differentiation caused by mutations in Foxc1 can lead to severe cortical dysplasia. 18187037_These results provide novel evidence that Foxc transcription factors are important regulators of the chemotactic motility of endothelial cells through the induction of CXCR4 expression. 20976766_Cortical dysplasia resembling cobblestone cortex, with basement membrane breakdown and lamination defects, is seen in Foxc1 mutants 22022403_These data provide evidence that the Foxf2 gene, separated from Foxc1 by less than 70 kb of genomic sequence (250 kb in human DNA), may explain human abnormalities in some cases of ASD where FOXC1 has been excluded genetically. 22171010_These findings identify a role for FoxC1 in inhibiting corneal angiogenesis. 22248045_A territorial correlation is identified between meningeal defects and disruption of interneuron migration along the adjacent marginal zone in the cerebral cortex of Foxc1-deficient mice. 23145080_These data indicate that Foxc1 expression leads to a similar enhanced osteogenic differentiation phenotype as observed with Msx2 overexpression 23344708_Foxc1 sets a threshold for the Bmp-dependent activation of Msx2, thus controlling the differentiation of osteogenic precursor cells and the rate and pattern of calvarial bone development. 23677979_FOXC1 is unnecessary for mammary morphogenesis; role in basal-like breast cancer likely involves processes that are unrelated to cell lineage specification. 23838094_Upper pole kidneys of newborn Foxc1(ch/ch) mice were significantly more hypoplastic and contained significantly fewer glomeruli than their lower pole counterparts. 24385915_Together our data demonstrates that Foxc1 - Fgf8 signaling regulates mammalian jaw patterning and provides a mechanistic basis for the pathogenesis of syngnathia. 24417671_A loss of Foxc1 function affects skull bone formation of the apical region. 24590069_identification of Foxc1 as a specific transcriptional regulator essential for development and maintenance of the mesenchymal niches for haematopoietic stem and progenitor cells 25513817_loss of Foxc1 non-autonomously induces a rapid and devastating decrease in embryonic cerebellar ventricular zone radial glial proliferation and concurrent increase in cerebellar neuronal differentiation 25733312_These observations reveal an essential role for Foxc1 in the early stage of vascular formation in the telencephalon. 25808752_Foxc1 is an important transcriptional partner of Ihh-Gli2 signalling during endochondral ossification, and that disruption of the Foxc1-Gli2 interaction causes skeletal abnormalities observed in the Axenfeld-Rieger syndrome. 26666591_These data indicate that Foxc1 expression is regulated by BMP4 and FOXC1 functions in the commitment of progenitor cells to the osteoblast fate and its expression is reduced when differentiation proceeds. 26824887_Foxc1 regulates both early cardiomyogenesis and the functional properties of embryonic stem cell-derived cardiomyocytes. 26839363_deletion of Foxc1 and Foxc2 specifically in Pax3-positive cells affects cell fate choices in the dermomyotome of somites at forelimb level, promoting the myogenic cell fate at the expense of endothelial cells that migrate to the limb 26912458_FOXC1 maintains the hair follicle stem cell niche and governs stem cell quiescence to preserve long-term tissue-regenerating potential.FOXC1 is necessary to establish a multiple-bulge hair follicle architecture. 26912704_In self-renewing stem cells (SCs), Foxc1 activates Nfatc1 and bone morphogenetic protein (BMP) signaling, two key mechanisms that govern quiescence. These findings reveal a dynamic, cell-intrinsic mechanism used by hair follicle SCs to reinforce quiescence upon self-renewal and suggest a unique ability of SCs to maintain cell identity. 27193493_Foxc1 and Foxc2 have a role in kidney and axial skeleton development. 27214551_FOXC1 and FOXC2 are essential regulators of lymphangiogenesis and may have roles in lymphatic-associated diseases 27592801_Foxc1 regulates sweat duct luminal cell differentiation and mimic apocrine miliaria. 27671872_These findings offer the first evidence for a role of the meninges in brain vascular development and provide new insight into potential causes of cerebrovascular defects in patients with FOXC1 mutations. 28092268_Lineage tracing experiments in Foxc1 mutant mouse cerebella indicate that aberrant migration of granule cell progenitors destined to form the posterior-most lobule causes this cerebellar malformation phenotype. 28223138_Foxc1 and Foxc2 maintain glomerular podocyte integrity by regulating the gene expression. 28253399_Compound, NC-specific Foxc1; Foxc2 homozygous mutant mice have more severe defects in structures of the ocular surface, such as the cornea and eyelids, accompanied by significant declines in the expression of another key developmental factor, Pitx2, and its downstream effector Dkk2, which antagonizes canonical Wnt signaling. 29070831_FOXC1 is a regulator of mammary gland development. 29322554_These findings suggest that Foxc1 is an important regulator to further chondrogenesis and initiate the ossification of the presphenoid and basisphenoid bones. 29511313_siRNA-mediated knockdown of FOXC1 was confirmed to decrease the proliferation rate, migration, and invasion in a model Basal-like breast cancer-like cell line (4T1). 29959321_Targeting EZH2 reactivates a breast cancer subtype-specific anti-metastatic transcriptional program driven by FOXC1. 31089719_Overexpression of miR-103-3p inhibited forkhead transcription factor C1 (Foxc1)-promoted osteogenic differentiation and special AT-rich sequence-binding protein 2 (Satb2) expression. 31422096_High FOXC1 expression is associated with pulmonary hypertension. 31517441_FOXC1 up-regulates the expression of toll-like receptors in myocardial ischaemia. 31588228_Role of FOXC1 in regulating APSCs self-renewal via STI-1/PrP(C) signaling. 31916318_FOXC1 negatively regulates BMP-SMAD activity and Id1 expression during osteoblast differentiation. 32259372_Conditional inactivation of Foxc1 and Foxc2 in neural crest cells leads to cardiac abnormalities. 34331943_Loss of Foxc1 and Foxc2 function in chondroprogenitor cells disrupts endochondral ossification. |
ENSG00000054598 |
FOXC1 |
349.969780 |
0.4392833347 |
-1.186776 |
0.14037494 |
71.037434 |
0.00000000000000003505108742105664214262480552824584042120901279573214059936958619800861924886703491210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000010918162854755312091688087106156719020857407027869090399008200620301067829132080078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
210.843260048516 |
20.547539593042 |
479.970996792562 |
33.520678428537 |
| ENSMUSG00000050315 |
118449 |
Synpo2 |
protein_coding |
Q91YE8 |
FUNCTION: Has an actin-binding and actin-bundling activity (PubMed:11673475). Can induce the formation of F-actin networks. At the sarcomeric Z lines is proposed to act as adapter protein that links nascent myofibers to the sarcolemma via ZYX and may play a role in early assembly and stabilization of the Z lines. Involved in autophagosome formation. May play a role in chaperone-assisted selective autophagy (CASA) involved in Z lines maintenance in striated muscle under mechanical tension; may link the client-processing CASA chaperone machinery to a membrane-tethering and fusion complex providing autophagosome membranes. Involved in regulation of cell migration. May be a tumor suppressor (By similarity). {ECO:0000250|UniProtKB:D4A702, ECO:0000250|UniProtKB:Q9UMS6, ECO:0000269|PubMed:11673475}. |
Actin-binding;Alternative splicing;Cell junction;Cytoplasm;Muscle protein;Nucleus;Phosphoprotein;Reference proteome;Tumor suppressor |
|
|
Human_homologues hsa:171024; |
actin cytoskeleton [GO:0015629]; cytosol [GO:0005829]; focal adhesion [GO:0005925]; intracellular membrane-bounded organelle [GO:0043231]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; stress fiber [GO:0001725]; Z disc [GO:0030018]; 14-3-3 protein binding [GO:0071889]; actin binding [GO:0003779]; alpha-actinin binding [GO:0051393]; filamin binding [GO:0031005]; muscle alpha-actinin binding [GO:0051371]; protein-macromolecule adaptor activity [GO:0030674]; autophagosome assembly [GO:0000045]; chaperone-mediated autophagy [GO:0061684]; positive regulation of actin filament bundle assembly [GO:0032233]; positive regulation of cell migration [GO:0030335]; regulation of Rho-dependent protein serine/threonine kinase activity [GO:2000298] |
17923693_The identification of myopodin as a direct target of PKA, CaMKII, and calcineurin defines a novel intracellular signaling pathway whereby changes in Z-disc dynamics may translate into compartmentalized signal transduction in the heart. 34637763_Isoform-specific functions of synaptopodin-2 variants in cytoskeleton stabilization and autophagy regulation in muscle under mechanical stress. |
ENSG00000172403 |
SYNPO2 |
175.155917 |
2.1713928815 |
1.118621 |
0.22677206 |
23.845771 |
0.00000104370933038044063780156074650040665119377081282436847686767578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000093135873100549529466762183060879465301695745438337326049804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
229.711292962572 |
36.2563660097471 |
105.789834219566 |
12.8969307735474 |
| ENSMUSG00000050395 |
326623 |
Tnfsf15 |
protein_coding |
A0A0U5J7F3 |
Human_homologues FUNCTION: Receptor for TNFRSF25 and TNFRSF6B. Mediates activation of NF-kappa-B. Inhibits vascular endothelial growth and angiogenesis (in vitro). Promotes activation of caspases and apoptosis. {ECO:0000269|PubMed:10597252, ECO:0000269|PubMed:11911831, ECO:0000269|PubMed:11923219, ECO:0000269|PubMed:9872942}. |
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:326623; |
plasma membrane [GO:0005886]; tumor necrosis factor receptor binding [GO:0005164]; activation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0006919]; activation of NF-kappaB-inducing kinase activity [GO:0007250]; immune response [GO:0006955] |
15702971_findings suggest that the transcription factor NF-kappaB plays an important role in the regulation of VEGI expression 16698931_induction of intestinal inflammation is associated with significant up-regulation of TL1A and tm DR3 in the inflamed mucosa 16920944_neonatal CD4+ CD3- cells up-regulate both CD30L and OX40L after adoptive transfer into an adult recipient 17785811_VEGI may have a key role in both the termination of angiogenesis and facilitation of dendritic cell maturation, which is the initial step in immune responses toward inflammatory stimuli. 18287561_TL1A may contribute to renal inflammation and injury through DR3-mediated activation of NF-kappaB and caspase-3 18598698_Results reveal that TL1A is an important modulator in the development of chronic mucosal inflammation by enhancing T(H)1 and T(H)17 effector functions. 20980995_These results establish a novel link between tumor necrosis factor (TNF)-family cytokine TL1A (TNFSF15) and interleukin 13 (IL-13) responses that results in small intestinal inflammation. 21264313_role of TL1A in mucosal T cells and antigen presenting cells function and showed that up-regulation of TL1A expression can promote mucosal inflammation and gut fibrosis. 23319737_Both in supernatant and as purified fusion protein, TL1A-Ig binds to TNFRSF25 transfected--but not untransfected--P815 tumor cells as determined by flow cytometry. 23918400_TNFSF15 inhibits vasculogenesis by regulating relative levels of membrane-bound and soluble isoforms of VEGF receptor 1. 24140642_delivers survival signals to plasma cells and promotes pathogenic Ab production in collagen arthritis 24220298_Our data demonstrate a key role for TL1A in promoting ILC2s at mucosal barriers. 24850426_These data demonstrated a direct role for TL1A-DR3 signaling in tissue fibrosis and that modulation of TL1A-DR3 signaling could inhibit gut fibrosis. 25399326_TL1A induces NF-kappaB activation in EC in renal and cardiac tissue from wild type but not DR3 knock-out mice. 25404161_TL1A deficiency impacts on the gut microbial composition and the mucosal immune system, especially the intraepithelial TCRgammadelta(+) T-cell subset, and that TL1A is involved in the establishment of adipose tissue. 25786692_these data identify TL1A-DR3 interactions as a novel pathway that promotes Th9 differentiation and pathogenicity. TL1A may be a potential therapeutic target in diseases dependent on IL-9. 27233964_this study shows that TL1A-/- mice are more susceptible to dextran sodium sulfate colitis 27664471_This work describes both a novel function and essential requirement for the DR3/TL1A pathway in acute, resolving, and chronic inflammation in the peritoneal cavity. 28177121_ATF3 protects against LPS-induced acute lung injury by inhibiting TL1A expression. 29254800_TL1A blocking ameliorates intestinal fibrosis in the T cell transfer model of chronic colitis 29335258_These data suggest differential roles for membrane and soluble TL1A on adaptive and innate immune cells 29988118_Tl1a-mediated intestinal fibrosis and fibroblast activation are dependent on specific microbial populations. 30552020_Results indicate a role for group 3 innate lymphoid cell (ILC3) in activating intestinal T cells and a central role for TNF-like ligand 1A (TL1A) in promoting ILC3 barrier immunity during colitis. 30906791_These results indicated that constitutive TL1A expression in myeloid cells exacerbated liver fibrosis, probably through macrophage recruitment and secretion of proinflammatory and profibrotic cytokines. 30949903_TL1A could impair intestinal epithelial barrier in the mouse model of colitis and might regulate tight junction expression via MLCK/p-MLC pathway and LPS-mediated MyD88/TRAF6 pathway. 31176785_Authors presented a possible mechanism of TL1A in UC development that TL1A may promote the differentiation of Th9 cells and enhanced IL-9 secretion by up-regulating the expression of TGF-beta, IL-4 and PU.1. 31299941_In contrast to pre-menopausal females, post-menopausal serum levels of TL1A are not significantly elevated and that DR3 expression is not induced on CD14+ monocytes. Murine ovariectomy (OVX) model of estrogen-deficiency revealed that early post-OVX expression of DR3 on CD4+ T cells is significantly elevated suggesting that DR3 and TL1A could play a potentially indirect role in early post-menopausal bone loss. 31379829_Activation of the DR3-TL1A Axis in Donor Mice Leads to Regulatory T Cell Expansion and Activation With Reduction in Graft-Versus-Host Disease. 31953572_Effects of tumor necrosis factor-like ligand 1A (TL1A) on imiquimod-induced psoriasiform skin inflammation in mice. 32001795_TL1A regulates adipose-resident innate lymphoid immune responses and enables diet-induced obesity in mice. 32958689_TL1A Promotes Lung Tissue Fibrosis and Airway Remodeling. 32963108_TL1A inhibits atherosclerosis in apoE-deficient mice by regulating the phenotype of vascular smooth muscle cells. 33097818_Direct signaling of TL1A-DR3 on fibroblasts induces intestinal fibrosis in vivo. 34257516_Effect and Mechanism of TL1A Expression on Epithelial-Mesenchymal Transition during Chronic Colitis-Related Intestinal Fibrosis. 34305924_Targeting TL1A/DR3 Signaling Offers a Therapeutic Advantage to Neutralizing IL13/IL4Ralpha in Muco-Secretory Fibrotic Disorders. 34596444_A Protective Role of Tumor Necrosis Factor Superfamily-15 in Intracerebral Hemorrhage-Induced Secondary Brain Injury. 34679191_Tumor necrosis factor ligand-related molecule 1A maintains blood-retinal barrier via modulating SHP-1-Src-VE-cadherin signaling in diabetic retinopathy. 35127254_TNFSF15 facilitates differentiation and polarization of macrophages toward M1 phenotype to inhibit tumor growth. |
ENSG00000181634 |
TNFSF15 |
16.968626 |
3.3841595576 |
1.758798 |
0.62607059 |
7.849978 |
0.00508213191930039550475717291533328534569591283798217773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0188485593851433208545387287813355214893817901611328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.7442135831483 |
11.9736106154298 |
7.90276378170136 |
2.78869221218474 |
| ENSMUSG00000050397 |
26927 |
Foxl2 |
protein_coding |
O88470 |
FUNCTION: Transcriptional regulator. Critical factor essential for ovary differentiation and maintenance, and repression of the genetic program for somatic testis determination. Prevents trans-differentiation of ovary to testis through transcriptional repression of the Sertoli cell-promoting gene SOX9. Has apoptotic activity in ovarian cells. Suppresses ESR1-mediated transcription of PTGS2/COX2 stimulated by tamoxifen. Activates SIRT1 transcription under cellular stress conditions. Activates transcription of OSR2. Is a regulator of CYP19 expression. Is a transcriptional repressor of STAR. Participates in SMAD3-dependent transcription of FST via the intronic SMAD-binding element. {ECO:0000269|PubMed:15059956, ECO:0000269|PubMed:15944199, ECO:0000269|PubMed:19106105, ECO:0000269|PubMed:19797124, ECO:0000269|PubMed:20005806}. |
Differentiation;DNA-binding;Isopeptide bond;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation;Ubl conjugation |
|
|
mmu:26927; |
Flemming body [GO:0090543]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; cysteine-type endopeptidase regulator activity involved in apoptotic process [GO:0043028]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; nuclear estrogen receptor binding [GO:0030331]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; ubiquitin conjugating enzyme binding [GO:0031624]; anatomical structure morphogenesis [GO:0009653]; apoptotic DNA fragmentation [GO:0006309]; cell differentiation [GO:0030154]; embryonic eye morphogenesis [GO:0048048]; extraocular skeletal muscle development [GO:0002074]; female gonad development [GO:0008585]; female somatic sex determination [GO:0019101]; granulosa cell differentiation [GO:0060014]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of gene expression [GO:0010629]; negative regulation of transcription by RNA polymerase II [GO:0000122]; ovarian follicle development [GO:0001541]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of follicle-stimulating hormone secretion [GO:0046881]; positive regulation of luteinizing hormone secretion [GO:0033686]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; response to human chorionic gonadotropin [GO:0044752]; response to organic cyclic compound [GO:0014070]; single fertilization [GO:0007338]; uterus development [GO:0060065] |
12471206_Molecular evolution and expression of FOXL2 12810580_FOXL2 is a highly conserved early regulator of vertebrate ovarian development. 15059956_mutations of FOXL2 could increase expression of StAR in small and medium follicles to accelerate follicle development, resulting in increased initial recruitment of dormant follicles and premature ovarian failure phenotype. 15944199_Foxl2 action thus represses the male gene pathway at several stages of female gonadal differentiation 16840539_FOXL2 plays a role in pituitary developmental control and in regulation of Cga gene expression. 17728319_inactivation of two ovarian somatic factors, Wnt4 and Foxl2, produces testis differentiation in XX mice, resulting in the formation of testis tubules and spermatogonia 19106105_FoxL2 and Smad3 coordinately regulate follistatin gene transcription.( 19538736_The results support the proposal of dose-dependent Foxl2 function and anti-testis action throughout ovary differentiation. 19875493_Following Foxl2 loss, early testis genes and several novel ovarian genes were consistently dysregulated during embryo-fetal development. 20005806_Study demonstrates in the mouse that a single factor, the forkhead transcriptional regulator FOXL2, is required to prevent transdifferentiation of an adult ovary to a testis. 20209145_FOXL2 interacts with PIAS1 and UBC9, and sumoylated in both human and mouse 20233786_FoxL2 plays a key role in activin induction of the FSHbeta gene, by binding to sites conserved across multiple species. 20650879_FOXL2 is expressed in ovary and adrenal gland; FOXL2 represses the promoter activity of Star; FOXL2 and NR5A1 synergistically up-regulate the transcriptional activity of Mc2r 21084449_These data support a model in which FOXL2 and BMP2 cooperate to ensure correct expression of Fst in the developing ovary. 21622537_SMADs and FOXL2 synergistically regulate murine follicle stimulating hormone beta transcription via a conserved proximal promoter element 21700720_These data indicate that, in addition to its role in the ovary, FoxL2 function in the pituitary is required for normal expression of FSH. 22116255_Data suggest that Foxl2 is involved in preventing ectopic steroidogenesis in foetal ovaries. 22294749_Protein-protein interactions among FOXL2, SMAD3, and progesterone receptor (PR) recruited to the Fshb promoter play a key role in facilitating Fshb transcription before the secondary FSH surge in rodents. 22837482_Increasing expression of the ovarian marker FOXL2 in mutant cords starting at E15.5, indicating progressive transdifferentiation of mutant Sertoli cells. 22847492_Data suggest that forkhead L2 plays a role in down-regulation of Star, Cyp19a1, Cyp11a1, and Ccnd2 genes in granulosa cells under influence of follicle stimulating hormone (or testosterone). 23340250_FOXL2 is required for FSH synthesis in vivo. 23810756_results demonstrate that miR-133b down-regulates Foxl2 expression in granulosa cells by directly targeting the 3'UTR 24098544_Differential proximal promoter DNA methylation may contribute to cell- specific Foxl2 expression in some cellular contexts. 24099863_In this review, we focus on the role of four specific FOX factors (FOXD1, FOXL2, FOXO1 and FOXP3) in gonadotropin hormone production 24451388_FOXL2 negatively regulates Sf1 expression by antagonizing WT1-KTS during early ovarian development in mice 24565867_Results show that a piggyBac insertion ~160 kb upstream of the transcription start site of Foxl2 partially disrupted its expression resulting in BPES (Blepharophimosis, ptosis, epicanthus inversus syndrome)-like conditions. 24739304_When the Smad4 gene is ablated in combination with its DNA binding cofactor Foxl2 in gonadotrope cells, mice make essentially no FSH and females are sterile. 24973035_AMH is an endogenous target gene of FOXL2. AMH and FOXL2 collaboratively work to reserve ovarian follicles. 25105693_our results demonstrate the necessity of FOXL2 for proper FSH production in mice and implicate FOXL2 in integration of transcription factors at the level of the FSHbeta promoter. 25289806_Microarray expression profiling of whole adrenal mRNA from ovariectomized vs. intact mice demonstrated selective upregulation of gonadal-like genes including Spinlw1 and Insl3 in GDX-induced adrenocortical tumors of the mouse. 25369636_FOXL2 mobilizes estrogen signaling to establish a coherent feed-forward loop repressing Sox9. 25687138_Foxl2 has a crucial role in postnatal uterine maturation and could help to understand sub-fertility predisposition in women. 25955311_Data indicate that the hypothalamic-pituitary-gonadal axis controls expression of Foxl2 in pituitary gonadotropes primarily through positive feedback from ovarian hormones. 26134413_Results support FOXL2 as a master transcription factor in a spectrum of developmental processes, including growth, cartilage and bone formation. Its action overlaps that of SOX9, though they are antagonistic in female vs male gonadal sex determination but conjoint in cartilage and skeletal development. 26939755_Data indicate that a balance between supporting cell self-renewal and differentiation is maintained in the developing ovary by relative Wnt4 protein/beta-catenin and p27Kip1 protein (p27)/Forkhead box L2 (FOXL2) activities. 27212026_evidence that FOXL2 modulates Col1a2 transcription through interaction with a response element 65 Kb upstream of the transcription start site 28900034_direct overexpression of Foxl2 decreased the expression of Sertoli cell-specific genes in primary Sertoli cells. Taken together, these results demonstrate that repression of beta-catenin (CTNNB1) signaling is required for lineage maintenance of Sertoli cells. 28938467_Testis determination involves FGFR2c-mediated repression of both the WNT4- and FOXL2-driven ovarian-determining pathways. 29415736_We found that endometrial cells expressing low FOXL2 levels, either endogenous or genetically manipulated, were associated with a higher attachment rate of mouse blastocysts. low-FOXL2 levels were associated with changes in the expression level of components of the Wnt/Fzd and apoptotic pathways, both of which are involved in uterine receptivity. 30212841_comparison of FOXL2 genome-wide occupancy in the fetal ovary with testis-determining factor SOX9 genome-wide occupancy in the fetal testis revealed extensive overlaps, implying that antagonistic signals between FOXL2 and SOX9 occur at the chromatin level. 31712577_RUNX1 plays complementary/redundant roles with FOXL2 to maintain fetal granulosa cell identity and combined loss of RUNX1 and FOXL2 results in masculinization of fetal ovaries. 31745296_Aberrant granulosa cell-fate related to inactivated p53/Rb signaling contributes to granulosa cell tumors and to FOXL2 downregulation in the mouse ovary. 32191302_Human Follicle-Stimulating Hormone ss Subunit Expression Depends on FOXL2 and SMAD4. 32307529_Oocytes suppress FOXL2 expression in cumulus cells in micedagger. 32945847_Aberrant and constitutive expression of FOXL2 impairs ovarian development and functions in mice. 32948877_Increased FOXL2 expression alters uterine structures and functionsdagger. 33215742_An alternative miRISC targets a cancer-associated coding sequence mutation in FOXL2. 33749886_Genomic exploration of the targets of FOXL2 and ESR2 unveils their implication in cell migration, invasion, and adhesion. 36409821_The Oncogenic FOXL2 C134W Mutation Is a Key Driver of Granulosa Cell Tumors. |
ENSG00000183770 |
FOXL2 |
196.622876 |
0.3548526457 |
-1.494708 |
0.20968958 |
50.216168 |
0.00000000000137708535847605903458345388079725083807849927985955673648277297616004943847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000268920053593683757715140280755103949564266407890045229578390717506408691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.573481170367 |
14.6694494831935 |
283.423224800311 |
29.5110054341047 |
| ENSMUSG00000050541 |
11548 |
Adra1b |
protein_coding |
B1AU41 |
FUNCTION: This alpha-adrenergic receptor mediates its action by association with G proteins that activate a phosphatidylinositol-calcium second messenger system. Its effect is mediated by G(q) and G(11) proteins. Nuclear ADRA1A-ADRA1B heterooligomers regulate phenylephrine (PE)-stimulated ERK signaling in cardiac myocytes. {ECO:0000256|ARBA:ARBA00025681}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Lipoprotein;Membrane;Palmitate;Phosphoprotein;Proteomics identification;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
|
caveola [GO:0005901]; cytoplasm [GO:0005737]; nuclear membrane [GO:0031965]; alpha1-adrenergic receptor activity [GO:0004937]; regulation of cardiac muscle contraction [GO:0055117]; regulation of vasoconstriction [GO:0019229] |
12390524_Overexpression of the alpha(1B)-AR in mice can cause a synucleinopathy with excessive tyrosine nitration but decreased phoshorylation similar to parkinsonian syndromes 12782680_alpha(1)-ARs in male mice are required for the physiological hypertrophy of normal postnatal cardiac development and for an adaptive response to cardiac stress 14519431_alpha(1A)- and alpha(1B)-adrenoceptors have roles in myocardial contractions 14581480_ADRA1b has a role in regulating glucose homeostasis in mice 15466664_These results demonstrate a critical role for the alpha(1B)AR in baroreceptor-mediated adrenergic signaling at the vascular neuroeffector junction. Moreover, alpha(1B)ARs modulate inotropic responses to baroreceptor activation. 15719155_changes at the level of central alpha1-adrenoceptors' binding during the formalin-induced pain in mice 17365508_alpha1B-ARs are the major alpha1-AR subtype expressed in DU145, PC3, and all TRAMP cell lines, but most of the receptor is localized in intracellular compartments in a nonfunctional state, which can be rescued upon prolonged incubation with any ligand. 17384126_These results suggest that lack of both alpha(1A)- and alpha(1B)-ARs could be necessary to inhibit neointimal formation in the mouse femoral artery. 17603545_These results demonstrate that alpha(1)-adrenoceptors, particularly alpha(1A)-adrenoceptors, are required for normal contractility of the vas deferens and consequent sperm ejaculation as well as having a function in fertility 17951539_Alpha 1b-AR signaling plays a critical role in the control of male fertility, spermatogenesis, and steroidogenic capacityof Leydig cells. 18802028_Nuclear alpha1B-adrenergic receptors signal activated ERK localization to caveolae in adult cardiac myocytes. 19112097_Data provide strong evidence for a pathological role of chronic alpha(1B)-AR activity in the development of repolarization defects and ventricular arrhythmias. 19125850_Alpha 1b-adrenergic receptor (alpha 1b-AR) plays a fundamental role in the mechanisms responsible for seizure onset, severity, and duration, whereas the brain damage observed in alpha 1b-AR-overexpressing mice is likely to be a secondary phenomenon. 19259685_in mouse ureters: the mRNA for the alpha1A-adrenoceptor was more prevalent than those for the alpha1B- and alpha1D-adrenoceptors 19540213_This is the first study to demonstrate that alpha(1A)- and alpha(1B)-ARs differentially modulate antidepressant-like behavior in the mouse. 24749494_The adra1b mediates adrenergic vasoconstriction in mouse retinal arterioles with damaged endothelium 26497985_eIF3f/alpha adrenergic receptor interaction 26750808_Taken together, these results suggested that alpha1B-adrenoceptor signaling is required for bone formation and regulated cellular proliferation through a mechanism relevant to the up-regulation of CCAAT/enhancer-binding protein delta in osteoblasts and, thus, provide new evidence for the physiological importance of alpha1B-adrenoceptor signalling in bone homeostasis |
ENSG00000170214 |
ADRA1B |
73.039580 |
2.1394333549 |
1.097229 |
0.30258753 |
13.113131 |
0.00029323325378633564516905885000142006902024149894714355468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0015611775329162757425249807852196681778877973556518554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
101.556949669295 |
21.4568308358913 |
47.4690877540477 |
7.74919360814232 |
| ENSMUSG00000050549 |
70617 |
Fam241a |
protein_coding |
Q9CZL2 |
|
Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:70617; |
Golgi apparatus [GO:0005794]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020] |
Human_homologues 18437351_Observational study of gene-disease association. (HuGE Navigator) 19380854_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 20332099_Observational study of gene-disease association. (HuGE Navigator) 23349771_Data indicate associations of SNPs in eight loci CXCR4, HHEX, FOXA2, NGN3, TCF7L2, FLJ39370 (C4orf32), LOC646279 (RPL21P7) and THADA with body mass index (BMI) and weight. |
ENSG00000174749 |
FAM241A |
39.060230 |
0.4563371400 |
-1.131828 |
0.39664850 |
8.105732 |
0.00441255041157480321079020768593181855976581573486328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0167069271362434504379912425520160468295216560363769531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.5599731431858 |
6.83780650340056 |
53.819798981086 |
10.6309762744739 |
| ENSMUSG00000050711 |
20254 |
Scg2 |
protein_coding |
Q03517 |
FUNCTION: Neuroendocrine protein of the granin family that regulates the biogenesis of secretory granules. {ECO:0000250|UniProtKB:P13521}. |
Calcium;Cleavage on pair of basic residues;Direct protein sequencing;Phosphoprotein;Reference proteome;Secreted;Signal;Sulfation |
|
|
mmu:20254; |
dense core granule [GO:0031045]; extracellular space [GO:0005615]; neuronal dense core vesicle [GO:0098992]; secretory granule [GO:0030141]; chemoattractant activity [GO:0042056]; cytokine activity [GO:0005125]; angiogenesis [GO:0001525]; eosinophil chemotaxis [GO:0048245]; induction of positive chemotaxis [GO:0050930]; intracellular signal transduction [GO:0035556]; MAPK cascade [GO:0000165]; negative regulation of endothelial cell apoptotic process [GO:2000352]; negative regulation of extrinsic apoptotic signaling pathway [GO:2001237]; positive chemotaxis [GO:0050918]; positive regulation of endothelial cell proliferation [GO:0001938] |
19357184_SCG2, as well as SCG3, may be a potential regulator of food intake based on its capacity to accumulate appetite-related hormones into secretory granules. 21044184_The wide subcellular localization of secretogranin II in mouse brain may reflect diverse release sites of neuropeptides and secretorneurin, or suggests its role in the sorting and packaging of molecules other than neuropeptides in non-LDCV compartments. 21521715_SN activates the protein kinase A (PKA) and cAMP-induced ERK signaling pathways in the LH-secreting mouse LbetaT2 pituitary cell line 21945639_it was proven that ScgII cannot be categorized as an activity marker, but more likely plays a role in visual system plasticity by modulating a range of neurotransmitters and neuropeptides 22655045_SgII mRNA levels were 10.5 fold upregulated in the left ventricle of animals with myocardial infarction and heart failure. 23171199_SgII binds directly to the secretory granule membrane and controls intracellular retention of proopiomelanocortin in AtT-20 cells. 23918206_Topical secretoneurin gene therapy accelerates diabetic wound healing by interaction between heparan-sulfate proteoglycans and basic FGF. 24111984_Scg2 is a good candidate for the signal integrator required for activity-dependent plasticity such as associative learning and memory. 26538656_Scg2 is a major secretory target of REST with a cell differentiation-enhancing activity in neural stem cells. 31121260_Endogenous secretoneurin plasma levels are lower in newborn animals subjected to an excitotoxic insult than in healthy controls. 32206122_Fasting before or after wound injury accelerates wound healing through the activation of pro-angiogenic SMOC1 and SCG2. |
ENSG00000171951 |
SCG2 |
784.605801 |
0.0238999100 |
-5.386851 |
0.17569834 |
1161.358880 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001524779809 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000387090768 |
Yes |
No |
35.5689125136782 |
4.34246897553372 |
1488.24462123769 |
88.8373565539143 |
| ENSMUSG00000050761 |
14724 |
Gp1bb |
protein_coding |
P56400 |
FUNCTION: Gp-Ib, a surface membrane protein of platelets, participates in the formation of platelet plugs by binding to von Willebrand factor, which is already bound to the subendothelium. {ECO:0000250}. |
Blood coagulation;Cell adhesion;Disulfide bond;Glycoprotein;Hemostasis;Leucine-rich repeat;Membrane;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:14724; |
glycoprotein Ib-IX-V complex [GO:1990779]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; blood coagulation [GO:0007596]; blood coagulation, intrinsic pathway [GO:0007597]; cell adhesion [GO:0007155]; megakaryocyte development [GO:0035855]; positive regulation of platelet activation [GO:0010572]; release of sequestered calcium ion into cytosol [GO:0051209] |
11816713_Role of GPIbbeta in modulating vWF mediated platelet adhesion. 19808696_Data show that the surface-bound VWF appears as a large, linear structure on the surface of 50% of the PT-VWD platelets. 24051142_These data indicate an important role for the platelet adhesion receptor GPIb-IX in endotoxin-induced thrombosis and thrombocytopenia. 27148783_GPIbbeta has a repressor function in thrombus formation 28943410_These results indicate that Reelin is an important regulator of GPIb-mediated platelet activation and may represent a new therapeutic target for the prevention and treatment of cardio- and cerebrovascular diseases 34732055_Early Endosomal GTPase Rab5 (Ras-Related Protein in Brain 5) Regulates GPIbbeta (Glycoprotein Ib Subunit beta) Trafficking and Platelet Production In Vitro. |
ENSG00000203618 |
GP1BB |
51.049174 |
0.3853661514 |
-1.375698 |
0.36804506 |
13.849163 |
0.00019808498856301726262149620616526135563617572188377380371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011012729910623935379587745586604796699248254299163818359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
31.2024012914607 |
8.04664803168927 |
80.9681939680559 |
14.572692635105 |
| ENSMUSG00000050994 |
215772 |
Adgb |
protein_coding |
G3UZ78 |
FUNCTION: Required for sperm flagellum formation and maturation of elongating spermatids, thus playing an essential role in male fertility (PubMed:35700329). Contributes to in vitro proteolytic cleavage of SEPT10 in a calmodulin-dependent manner (PubMed:35700329). {ECO:0000269|PubMed:35700329}. |
Alternative splicing;Cell projection;Cilium;Coiled coil;Differentiation;Flagellum;Reference proteome;Spermatogenesis |
|
|
Human_homologues hsa:79747; |
sperm annulus [GO:0097227]; sperm flagellum [GO:0036126]; sperm midpiece [GO:0097225]; calcium-dependent cysteine-type endopeptidase activity [GO:0004198]; heme binding [GO:0020037]; oxygen binding [GO:0019825]; cell differentiation [GO:0030154]; proteolysis [GO:0006508]; spermatogenesis [GO:0007283] |
Human_homologues 22115833_Molecular evolutionary analyses indicate that this chimeric globin family is phylogenetically ancient and originated in the common ancestor to animals and choanoflagellates. In humans and mice, the gene is predominantly expressed in testis tissue. 24966926_Suggest ADGB is involved in the progression of glioma in vitro. 33453283_Androglobin gene expression patterns and FOXJ1-dependent regulation indicate its functional association with ciliogenesis. |
ENSG00000118492 |
ADGB |
7.214336 |
0.0126244476 |
-6.307636 |
1.61012734 |
15.066854 |
0.00010376953152106402138935697943011859933903906494379043579101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0006162251478085174630563991549081492848927155137062072753906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.189987731330952 |
0.258879033287959 |
15.0491916977548 |
9.45702945500554 |
| ENSMUSG00000051177 |
18795 |
Plcb1 |
protein_coding |
Q9Z1B3 |
FUNCTION: Catalyzes the hydrolysis of 1-phosphatidylinositol 4,5-bisphosphate into diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3) and mediates intracellular signaling downstream of G protein-coupled receptors (PubMed:27653213). Regulates the function of the endothelial barrier (PubMed:27653213). {ECO:0000269|PubMed:27653213, ECO:0000303|PubMed:27653213}. |
Alternative splicing;Calcium;Cytoplasm;Hydrolase;Lipid degradation;Lipid metabolism;Lipoprotein;Membrane;Nucleus;Palmitate;Phosphoprotein;Reference proteome;Transducer |
|
|
mmu:18795; |
chromatin [GO:0000785]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; membrane [GO:0016020]; myelin sheath [GO:0043209]; nuclear membrane [GO:0031965]; nuclear speck [GO:0016607]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; postsynaptic cytosol [GO:0099524]; protein-containing complex [GO:0032991]; calcium channel regulator activity [GO:0005246]; calcium ion binding [GO:0005509]; calmodulin binding [GO:0005516]; channel activator activity [GO:0099103]; enzyme binding [GO:0019899]; GTPase activator activity [GO:0005096]; identical protein binding [GO:0042802]; ion channel regulator activity involved in G protein-coupled receptor signaling pathway [GO:0099107]; lamin binding [GO:0005521]; phosphatidylinositol phospholipase C activity [GO:0004435]; phosphatidylinositol-4,5-bisphosphate binding [GO:0005546]; protein serine/threonine kinase activator activity [GO:0043539]; activation of meiosis involved in egg activation [GO:0060466]; brain development [GO:0007420]; cell adhesion [GO:0007155]; cellular response to fluoride [GO:1902618]; cellular response to glyceraldehyde [GO:1905631]; cellular response to ionomycin [GO:1904637]; cellular response to vasopressin [GO:1904117]; cerebral cortex development [GO:0021987]; erythrocyte differentiation [GO:0030218]; fat cell differentiation [GO:0045444]; G protein-coupled acetylcholine receptor signaling pathway [GO:0007213]; G2/M transition of mitotic cell cycle [GO:0000086]; glutamate receptor signaling pathway [GO:0007215]; inositol trisphosphate metabolic process [GO:0032957]; insulin receptor signaling pathway [GO:0008286]; insulin-like growth factor receptor signaling pathway [GO:0048009]; interleukin-1-mediated signaling pathway [GO:0070498]; interleukin-12-mediated signaling pathway [GO:0035722]; interleukin-15-mediated signaling pathway [GO:0035723]; ion channel modulating, G protein-coupled receptor signaling pathway [GO:0099105]; learning [GO:0007612]; ligand-gated ion channel signaling pathway [GO:1990806]; macrophage differentiation [GO:0030225]; memory [GO:0007613]; mRNA processing [GO:0006397]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of monocyte extravasation [GO:2000438]; oocyte maturation [GO:0001556]; phosphatidylinositol catabolic process [GO:0031161]; phosphatidylinositol metabolic process [GO:0046488]; phosphatidylinositol-mediated signaling [GO:0048015]; phospholipase C-activating G protein-coupled acetylcholine receptor signaling pathway [GO:0007207]; phospholipase C-activating G protein-coupled receptor signaling pathway [GO:0007200]; positive regulation of acrosome reaction [GO:2000344]; positive regulation of CD24 production [GO:2000560]; positive regulation of developmental growth [GO:0048639]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of embryonic development [GO:0040019]; positive regulation of G1/S transition of mitotic cell cycle [GO:1900087]; positive regulation of insulin secretion [GO:0032024]; positive regulation of interleukin-12 production [GO:0032735]; positive regulation of JNK cascade [GO:0046330]; positive regulation of myoblast differentiation [GO:0045663]; positive regulation of sodium:proton antiporter activity [GO:0032417]; postsynaptic modulation of chemical synaptic transmission [GO:0099170]; regulation of establishment of endothelial barrier [GO:1903140]; regulation of fertilization [GO:0080154]; regulation of G protein-coupled receptor signaling pathway [GO:0008277]; regulation of retrograde trans-synaptic signaling by endocanabinoid [GO:0099178]; response to monosaccharide [GO:0034284]; response to peptide hormone [GO:0043434]; signal transduction [GO:0007165] |
12123715_Regulation of nuclear phospholipase C, beta 1 (review) 15174099_Expression of Plcb1 was studied in a cell line during myoblast differentiation. 15664177_PLCbeta1 serves as a coincidence detector through its Ca(2+) dependency for endocannabinoid release in hippocampal neurons. 15749982_phospholipase C, beta 1 signalling pathway plays a role in the development of normal cortical circuitry 15849202_nuclear PLCbeta1 up-regulated the expression of CD24. The correlation was strengthened by the observation that when PLCbeta1 expression was silenced by means of small interfering RNA, CD24 expression was down-regulated 16210321_activation of Galpha q in cardiac myocytes of adult mice causes a dilated cardiomyopathy that requires the activation of PLCbeta 16538662_beta-N-acetylglucosamine modification negatively modulates the activity of PLC-beta1 16951552_desensitization of inward cationic current following carbachol in murine gastric myocytes is not due to a pathway dependent on intracellular Ca2+ but to the PLC beta1 pathway. 17620288_Spontaneous calcium oscillations and nuclear translocation of PLC-beta1 may reflect some degree of oocyte maturity. 17667964_Knockout mice are a model for schizophrenia which can be treated by environemntal enrichment and clozapine. 17696993_The present results show that the PLCbeta1 mutant mice share some of the behavioral abnormalities that have been reported in patients with schizophrenia. Thus, the PLCbeta1-linked signaling pathways may be involved. 17925404_that IP3R3 is a principal mediator of sweet, bitter, and umami taste perception and would be a missing molecule linking phospholipase C beta2 to TRPM5 activation. 17961538_These results provide the first evidence for a role of an egg-derived PLC acting in conjunction with a sperm-derived PLCzeta in egg activation. 18493969_dramatic changes in expression of RGS4 mRNA in selective regions of the PLC-beta1 knockout mouse brain, particularly the CA1 region of the hippocampus 18690108_NDMC/clozapine ratio and treatment response. Administration of NMDC reversed a striking hyperactive phenotype in the phospholipase C-beta1 knockout mouse, whereas no significant effects were observed in wild-type animals. 18692062_Ins(1,4,5)P(3) and IP(3)-receptor (type 2) regulate PLCbeta1 and thereby maintain levels of Ins(1,4,5)P(3), which implies some functional significance for Ins(1,4,5)P(3) in the heart 18836131_PLCbeta1 localization is a key factor that determines progressive aquisition of meiotic competence and meiosis during folliculogenesis. 18957514_PLCb1 localization is regulated by its own substrate, phosphatidylinositol (4,5)-bisphosphate. 19028838_PLCbeta1 colocalizes and physically interacts with lamin B1. 19236734_Demonstrate critical role of the glutamatergic system in the phenotype of the PLC-beta1 knockout mouse and highlight the role of these interconnected signalling pathways in schizophrenia-like behavioural disruption. 19342451_female Plc-beta1-/- mice have a range of reproductive defects, affecting ovulation, support of preimplantation embryo development, implantation & behavior but reduced fertility of Plc-beta1-/- males may be due to altered reproductive behavior. 19385066_Inhibition of PLCbeta1 activity prevents the upregulation of cyclinD3 and cdk4 protein, suggesting that PLCbeta1 plays a role in the control of the cell cycle during differentiation. 19687358_thrombin-induced signaling via protease-activated receptor-1, phospholipase-Cbeta, phosphoinositide 3-kinase, and nitric oxide in monocytes impairs monocyte transendothelial migration 19729020_Dilated atria from human and mouse showed heightened expression of PLCbeta1b. 21974932_These findings highlight a novel pathway by which nuclear PLCs affect insulin secretion and identify PPARgamma as a novel molecular target of nuclear PLCs. 23295235_Defective PLCbeta1-mediated signalling during implantation is associated with the implantation failure, aberrant ovarian steroid signalling and endocannabinoid metabolism. 23478264_Galphaq-coupled Lpar1 regulates cell proliferation and migration by activating two distinct PLC-beta isozymes, PLC-beta1 and PLC-beta2, respectively. 23503970_Tnis study demonistrated that plc beta1 distribuate in mice cerebellum. 23665500_Described is the interaction of nuclear PI-PLCbeta1b with a number of proteins involved in nuclear import, differentiation, mRNA processing, and apoptosis, thereby hinting at novel targets for therapeutic intervention in hematological malignancies. 24227722_PLCbeta coupling to D1 dopamine receptor regulates forward locomotion. 25268789_Results indicate that PLC-beta1 signalling in the medial prefrontal cortex is required for working memory 25550457_In pro-B-lymphoblastic cells (Ba/F3), treated with H2O2, PI-PLCbeta1b conferred resistance to cell death, promoting cell cycle progression and cell proliferation and influencing the expression of cyclin A and E. 25916683_Unpredictable chronic mild stress mice exhibited lower nucleus accumbens (NAC) shell levels of phospholipase Cbeta. 25918049_Heightened PLCbeta1b activity observed in diseased myocardium contributes to pathology by PKCalpha-mediated contractile dysfunction. 26217938_inhibition of miR-214 in C2C12 cells significantly enhances the protein level of PLCbeta1 and promotes C2C12 BMP-2-induced osteogenesis by targeting PLCbeta1 27342861_functions in plasma membrane remodeling, particularly caveolae formation 27563828_Data show that phospholipase C (PLC)-beta1 (PLC-beta1) overexpression determines an increase in beta-catenin translocation and that PLC-beta1, inositol polyphosphate multikinase (IPMK) and beta-catenin are mediators of the same signaling pathway. 27776973_Plcb1 plays a crucial role in the initiation of the genetic program responsible for muscle differentiation and osteogenesis. (Review) 30517689_Phospholipase C-Beta1 reduction leads to disturbances to beta and gamma oscillatory dynamics and pre-pulse inhibition behavior in an animal model of schizophrenia. 31268747_The present study showed that PLC-beta1 was crucial for beta-cell function by generation of each PLC-beta conditional knockout mouse. Mice lacking PLC-beta1 in beta cells exhibited a marked defect in glucose-stimulated insulin secretion, leading to glucose intolerance. 34625112_Activation of PLCbeta1 enhances endocannabinoid mobilization to restore hippocampal spike-timing-dependent potentiation and contextual fear memory impaired by Alzheimer's amyloidosis. 36202196_Cell-autonomous PLCbeta1 modulation of neural stem/progenitor cell proliferation during adult hippocampal neurogenesis. 36517806_Conformational alteration in glycan induces phospholipase Cbeta1 activation and angiogenesis. |
ENSG00000182621 |
PLCB1 |
132.699123 |
0.4763520280 |
-1.069900 |
0.24898243 |
18.402341 |
0.00001788381340335941916655677907943555737801943905651569366455078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001263479804081496450825217170432779312250204384326934814453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
85.1759810315894 |
14.7728750917964 |
178.808897689592 |
22.5115838327735 |
| ENSMUSG00000051335 |
328232 |
Gfod1 |
protein_coding |
Q3UHD2 |
|
Oxidoreductase;Reference proteome;Secreted;Signal |
|
|
mmu:328232; |
extracellular region [GO:0005576]; nucleotide binding [GO:0000166]; oxidoreductase activity [GO:0016491] |
Human_homologues 18821565_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 27191742_Authors found the expression of peejar was positively correlated with the expression of GFOD1 in ccRCC tissue, with Pearson correlation coefficiency reaching 0.939 (p |
ENSG00000145990 |
GFOD1 |
55.739189 |
0.4899278056 |
-1.029359 |
0.33732356 |
9.251110 |
0.00235352705597439591442276984878390067024156451225280761718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0097947880100005477177482759998383698984980583190917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
36.3204738588898 |
8.56985659427979 |
74.1343386589001 |
12.5348824602532 |
| ENSMUSG00000051359 |
52589 |
Ncald |
protein_coding |
Q91X97 |
FUNCTION: May be involved in the calcium-dependent regulation of rhodopsin phosphorylation. Binds three calcium ions (By similarity). {ECO:0000250}. |
Calcium;Direct protein sequencing;Lipoprotein;Metal-binding;Myristate;Reference proteome;Repeat |
|
|
mmu:52589; |
cytosol [GO:0005829]; actin binding [GO:0003779]; alpha-tubulin binding [GO:0043014]; calcium ion binding [GO:0005509]; clathrin binding [GO:0030276]; tubulin binding [GO:0015631]; calcium-mediated signaling [GO:0019722]; regulation of systemic arterial blood pressure [GO:0003073] |
16101019_Neurocalcin-containing DRG neurons include all proprioceptive, and a subset of mechanoreceptive and proprioceptive. Furthermore, NT-3 is involved in the maturation of motor end-plates. 17548529_According to their distribution in the olfactory bulb, neurocalcin-immunoreactive interneurons could reflect a sexually dimorphic regulation of specific odorants. |
ENSG00000104490 |
NCALD |
31.509378 |
2.5889305094 |
1.372356 |
0.47361579 |
8.358802 |
0.00383823514653794861314906050608897203346714377403259277343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0147823003165780102879933721737870655488222837448120117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.2887004740519 |
14.133472992764 |
16.3344285685318 |
4.26971704565476 |
| ENSMUSG00000051373 |
227721 |
Plpp7 |
protein_coding |
Q91WB2 |
FUNCTION: Plays a role as negative regulator of myoblast differentiation, in part through effects on MTOR signaling. Has no detectable enzymatic activity. Knockdown in myoblasts strongly promotes differentiation, whereas overexpression represses myogenesis. {ECO:0000269|PubMed:19704009}. |
Endoplasmic reticulum;Membrane;Nucleus;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:227721; |
endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; nuclear envelope [GO:0005635]; sphingosine-1-phosphate phosphatase activity [GO:0042392]; negative regulation of myotube differentiation [GO:0010832] |
19704009_The nuclear lamina-associated nuclear envelope transmembrane protein NET39 (Ppapdc3) functions as a negative regulator of myoblast differentiation. 27264872_Three muscle-specific nuclear membrane proteins, NET39, Tmem38A, and WFS1, direct specific myogenic genes to the nuclear periphery to facilitate their repression during myogenesis. 33514739_The nuclear envelope protein Net39 is essential for muscle nuclear integrity and chromatin organization. |
ENSG00000160539 |
PLPP7 |
23.078629 |
0.2037263518 |
-2.295295 |
0.58627326 |
15.197997 |
0.00009680602553668842663852495133980369246273767203092575073242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005782546592058188886925629290658434911165386438369750976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.66962239723875 |
3.20322625547711 |
37.6466879649169 |
10.4939541637636 |
| ENSMUSG00000051378 |
70218 |
Kif18b |
protein_coding |
Q6PFD6 |
FUNCTION: In complex with KIF2C, constitutes the major microtubule plus-end depolymerizing activity in mitotic cells. Its major role may be to transport KIF2C and/or MAPRE1 along microtubules (By similarity). {ECO:0000250}. |
ATP-binding;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Microtubule;Mitosis;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:70218; |
astral microtubule [GO:0000235]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; kinesin complex [GO:0005871]; microtubule [GO:0005874]; microtubule end [GO:1990752]; microtubule plus-end [GO:0035371]; mitotic spindle astral microtubule [GO:0061673]; mitotic spindle midzone [GO:1990023]; nuclear body [GO:0016604]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; cytoskeletal motor activity [GO:0003774]; kinesin binding [GO:0019894]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; plus-end-directed microtubule motor activity [GO:0008574]; cell division [GO:0051301]; microtubule depolymerization [GO:0007019]; microtubule-based movement [GO:0007018]; mitotic cell cycle [GO:0000278]; mitotic sister chromatid segregation [GO:0000070]; regulation of cell division [GO:0051302] |
34432485_KIF18B is a cell type-specific regulator of spindle orientation in the epidermis. |
ENSG00000186185 |
KIF18B |
557.839243 |
2.0485311309 |
1.034590 |
0.11162783 |
85.491205 |
0.00000000000000000002327344451058872559067109758993359261243098013948538214500788567917766158643644303083419799804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000009183966497028171453961751409889541534122336328407540384510809872153913602232933044433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
746.736185430663 |
57.8360218450263 |
364.522742254948 |
21.8908497692106 |
| ENSMUSG00000051379 |
71436 |
Flrt3 |
protein_coding |
Q8BGT1 |
FUNCTION: Functions in cell-cell adhesion, cell migration and axon guidance, exerting an attractive or repulsive role depending on its interaction partners (PubMed:19056886, PubMed:25374360). Plays a role in the spatial organization of brain neurons (PubMed:25374360). Plays a role in vascular development in the retina (PubMed:25374360). Plays a role in cell-cell adhesion via its interaction with ADGRL3 and probably also other latrophilins that are expressed at the surface of adjacent cells (PubMed:22405201, PubMed:25374360). Interaction with the intracellular domain of ROBO1 mediates axon attraction towards cells expressing NTN1 (PubMed:24560577). Mediates axon growth cone collapse and plays a repulsive role in neuron guidance via its interaction with UNC5B, and possibly also other UNC-5 family members (PubMed:21673655, PubMed:25374360). Promotes neurite outgrowth (in vitro) (By similarity). Mediates cell-cell contacts that promote an increase both in neurite number and in neurite length (By similarity). Plays a role in the regulation of the density of glutamaergic synapses (PubMed:22405201). Plays a role in fibroblast growth factor-mediated signaling cascades (PubMed:16872596). Required for normal morphogenesis during embryonic development, but not for normal embryonic patterning (PubMed:19056886). Required for normal ventral closure, headfold fusion and definitive endoderm migration during embryonic development (PubMed:18448090). Required for the formation of a normal basement membrane and the maintenance of a normal anterior visceral endoderm during embryonic development (PubMed:19056886). {ECO:0000250|UniProtKB:B1H234, ECO:0000269|PubMed:16872596, ECO:0000269|PubMed:18448090, ECO:0000269|PubMed:19056886, ECO:0000269|PubMed:21673655, ECO:0000269|PubMed:22405201, ECO:0000269|PubMed:24560577, ECO:0000269|PubMed:25374360}. |
3D-structure;Cell adhesion;Cell junction;Cell membrane;Cell projection;Developmental protein;Disulfide bond;Endoplasmic reticulum;Glycoprotein;Leucine-rich repeat;Membrane;Reference proteome;Repeat;Secreted;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:71436; |
axon terminus [GO:0043679]; axonal growth cone [GO:0044295]; cell junction [GO:0030054]; cell-cell junction [GO:0005911]; cytosol [GO:0005829]; endoplasmic reticulum membrane [GO:0005789]; extracellular space [GO:0005615]; focal adhesion [GO:0005925]; glutamatergic synapse [GO:0098978]; growth cone membrane [GO:0032584]; plasma membrane [GO:0005886]; postsynaptic density [GO:0014069]; postsynaptic membrane [GO:0045211]; synaptic membrane [GO:0097060]; chemorepellent activity [GO:0045499]; fibroblast growth factor receptor binding [GO:0005104]; protein homodimerization activity [GO:0042803]; axon guidance [GO:0007411]; cell-cell adhesion via plasma-membrane adhesion molecules [GO:0098742]; embryonic morphogenesis [GO:0048598]; fibroblast growth factor receptor signaling pathway [GO:0008543]; head development [GO:0060322]; heart development [GO:0007507]; neuron projection development [GO:0031175]; neuron projection extension [GO:1990138]; positive regulation of synapse assembly [GO:0051965]; proepicardium cell migration involved in pericardium morphogenesis [GO:0003345]; response to axon injury [GO:0048678]; synapse assembly [GO:0007416]; synapse organization [GO:0050808]; synaptic membrane adhesion [GO:0099560] |
16872596_FLRT3 has an expression pattern during somite development 18448090_Data demonstrate that FLRT3 plays a key role in controlling cell adhesion and tissue morphogenesis in the developing mouse embryo. 19635589_Flrt3 was highly expressed initially in the midbrain. 24560577_FLRT3 is a modulator of Netrin-1 response in axons.FLRT3 is a Robo1 coreceptor.FLRT3 is a Robo1-interacting protein that determines Netrin-1 attraction in developing axons. 26235031_The crystal structure of the OLF/FLRT3 complex shows that LPHN3-OLF in the closed state binds with high affinity to the concave face of FLRT3-LRR with a combination of hydrophobic and charged residues. 28475893_Mice with deletions of FLRT1 and FLRT3 adhesion molecules develop macroscopic sulci with preserved layered organization and radial glial morphology. Cortex folding in these mutants does not require progenitor cell amplification but is dependent on changes in neuron migration. 34301831_FLRT2 and FLRT3 Cooperate in Maintaining the Tangential Migratory Streams of Cortical Interneurons during Development. |
ENSG00000125848 |
FLRT3 |
209.753829 |
3.2327291900 |
1.692753 |
0.20141854 |
69.506026 |
0.00000000000000007618140383752201566581809022465419793978158076062576209430687868007225915789604187011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000022932710340971054046109330001814519543507260579406903389099170453846454620361328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
319.709386526647 |
44.7806325998512 |
98.8976705858468 |
10.8891017370359 |
| ENSMUSG00000051439 |
12475 |
Cd14 |
protein_coding |
P10810 |
FUNCTION: Coreceptor for bacterial lipopolysaccharide. In concert with LBP, binds to monomeric lipopolysaccharide and delivers it to the LY96/TLR4 complex, thereby mediating the innate immune response to bacterial lipopolysaccharide (LPS) (PubMed:16148141). Acts via MyD88, TIRAP and TRAF6, leading to NF-kappa-B activation, cytokine secretion and the inflammatory response (PubMed:8612135, PubMed:15895089). Acts as a coreceptor for TLR2:TLR6 heterodimer in response to diacylated lipopeptides and for TLR2:TLR1 heterodimer in response to triacylated lipopeptides, these clusters trigger signaling from the cell surface and subsequently are targeted to the Golgi in a lipid-raft dependent pathway (By similarity). Acts as an accessory receptor for M.tuberculosis lipoproteins LprA, LprG and LpqH, in conjunction with coreceptors TLR2 and TLR1. The lipoproteins act as agonists to modulate antigen presenting cell functions in response to the pathogen (PubMed:19362712). Binds electronegative LDL (LDL(-)) and mediates the cytokine release induced by LDL(-) (By similarity). {ECO:0000250|UniProtKB:P08571, ECO:0000269|PubMed:15895089, ECO:0000269|PubMed:16148141, ECO:0000269|PubMed:19362712, ECO:0000269|PubMed:21821728, ECO:0000269|PubMed:8612135}. |
3D-structure;Cell membrane;Disulfide bond;Glycoprotein;Golgi apparatus;GPI-anchor;Immunity;Inflammatory response;Innate immunity;Leucine-rich repeat;Lipoprotein;Membrane;Reference proteome;Repeat;Secreted;Signal |
|
This gene encodes a protein that plays an important role in the innate immune response and is expressed in monocyte/macrophage cells. This gene product acts as a co-receptor that binds several microbial and fungal molecules, including lipopolysaccharides (LPS). This proteins LPS-binding activity is enhanced by the LPS binding protein (LBP) to allow binding to the TLR4-MD-2 co-receptor complex. The product of this gene is found in two forms, either as a soluble protein or attached to the cell surface by a glycosylphosphatidylinositol anchor. [provided by RefSeq, Jul 2014]. |
mmu:12475; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; lipopolysaccharide receptor complex [GO:0046696]; membrane raft [GO:0045121]; plasma membrane [GO:0005886]; lipopeptide binding [GO:0071723]; lipopolysaccharide binding [GO:0001530]; lipopolysaccharide immune receptor activity [GO:0001875]; lipoteichoic acid binding [GO:0070891]; cellular response to diacyl bacterial lipopeptide [GO:0071726]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to lipoteichoic acid [GO:0071223]; cellular response to molecule of bacterial origin [GO:0071219]; cellular response to triacyl bacterial lipopeptide [GO:0071727]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; lipopolysaccharide-mediated signaling pathway [GO:0031663]; positive regulation of cytokine production [GO:0001819]; positive regulation of endocytosis [GO:0045807]; positive regulation of interleukin-8 production [GO:0032757]; positive regulation of lipopolysaccharide-mediated signaling pathway [GO:0031666]; positive regulation of NIK/NF-kappaB signaling [GO:1901224]; positive regulation of toll-like receptor 4 signaling pathway [GO:0034145]; positive regulation of tumor necrosis factor production [GO:0032760]; positive regulation of type I interferon production [GO:0032481]; positive regulation of type II interferon production [GO:0032729]; receptor-mediated endocytosis [GO:0006898]; response to bacterium [GO:0009617]; response to electrical stimulus [GO:0051602]; response to ethanol [GO:0045471]; response to heat [GO:0009408]; response to magnesium ion [GO:0032026]; response to molecule of bacterial origin [GO:0002237]; response to tumor necrosis factor [GO:0034612]; toll-like receptor 4 signaling pathway [GO:0034142] |
11779220_CD14-dependent activation of NF-kappaB by filarial parasitic sheath proteins 11870620_Hyperreactivity to LPS is characterized by an up-regulation of CD14, but the sensitization by P. acnes is not CD14 dependent. 12146799_expression of toll-like receptor 2 and CD14 transcripts occurred in barrier-associated structures, as well as parenchymal elements of the spinal cord, and within numerous regions of the brain in experimental autoimmune encephalomyelitis 12165539_leukocyte-endothelial cell interactions in vivo in response to LPS in CD14-deficient (CD14(-/-)) mice 12171914_CD14 has a role in mediating signaling responses to Aspergillus fumigatus 12370383_In the case of intraperitoneal infection of CD14-deficient mice with Salmonella typhimurium, no local inflammatory response is evident, neutrophil influx is delayed, and the mice succumb to the infection. 12391013_role in interleukin-10-mediated immunosuppression by Yersinia V antigen 12424240_binding to minimally modified LDL induces macrophage spreading via TLR4/MD-2, and inhibits phagocytosis of apoptotic cells 12427659_CD14 is necessary for the development of left ventricular dysfunction during lipopolysaccharide induced shock in vivo. 12482856_upregulation caused by acute ethanol involves oxidant-dependent AP-1 pathway 12496149_pulmonary surfactant protein C binds with CD14 and lipopolysaccharide 12639839_CD14 has a role in cytokine production and nitric oxide release of macrophages which might contribute to the attenuation of lung injury 12935356_We now report the existence of a new locus, Lps2, which is required for tumor necrosis factor(TNF) production in response to lipopolysaccharide(LPS). 14517279_direct LPS interaction with cell surface TLR4-MD-2 that is distinct from that with MD-2 or CD14. 14614560_iIvolves the immune system in triggering type I diabetes onset in NOD mice. 14752288_CD14 and Tlr4 have roles in a cascade of signaling events that control the immediate-early and differential induction of serum amyloid A proteins and serum amyloid P component in the liver after burn injury 15194867_important role for CD14 in the recognition and subsequent killing of amyloid-beta damaged neurons by microglia. 15208271_Mice with mutations in CD14 have an 'ideal' body plan: increased bone mineral content& density, size, decreased body fat,longevity, normal activity and fertility, and no evidence of infection. They do not become obese. 15271679_CD14 is involved in mediating the anorectic effects of both lipopolysaccharides and muramyl dipeptide. 15322035_Plays relatively minor role in the response to Cryptococcus neoformans infection in mice. 15459483_CD14 plays a role in the Propionibacterium acnes-LPS system in both priming and induction phases. 15522312_Injury elicits CD14 induction and hyperphosphorylation of c-Jun N-terminus activation domain, and CD14 is involved in modulation of c-Jun's transactivation potential via phosphorylation. May be associated with hepatic pathogenesis after injury. 15541715_the expression of CD14 and TLR4 on SMphi and PMphi from female and male mice was similar in sham-operated animals and after (trauma-hemorrhage) 15644310_Ligand diversity of CD14 could be explained by the generous size of the pocket, the considerable flexibility of the rim of the pocket, and the multiplicity of grooves available for ligand binding. 15661914_Engagement of the CD14 signaling pathway plays a critical role in attenuating the innate inflammatory response to Borrelia burgdorferi and facilitating disease resolution. 15661914_The absence of CD14 leads to higher cytokine production in Lyme Arthritis model 15731076_LPS-induced acute lung injury is partially dependent on CD14 in an in vivo mouse model 15814723_SP-D influences innate host defense, in part, by regulating sCD14 in a process mediated by MMP-9 and MMP-12. 15895089_The Heedless phenotype was positionally ascribed to a premature stop codon in Cd14. 15896296_Cell fractionation analyses show that acute EtOH altered LPS-related partition of CD14, a critical component of the LPS receptor. complex. 15941778_CD14 deficiency leads to increased MIP-2 production, CXCR2 expression, neutrophil transmigration, and early death in pneumococcal infection. 16148141_CD14(+/+), but not CD14(-/-), macrophages are highly sensitive to stimulation by natural forms of LPS and show the ability to distinguish between various LPS ligands, consistent with CD14 being a highly specific receptor. 16163057_CD14 plays a crucial intermediate role in the progression of acute necrotizing pancreatitis 16210672_CD14 and Toll-like receptor 4 play a key role in innate sensing of A. baumannii via the LPS moiety, resulting in effective elimination of the bacteria from the lung. 16225933_The survival of HuPrP106-126-damaged neurons was increased by pre-treating microglial cells with anti-CD14 antibodies, while microglial cells from CD14 knockout mice failed to kill HuPrP106-126-damaged neurons. 16229899_Microglial responses to intact S. aureus occurred primarily via a CD14-independent manner. Complex nature of gram-positive bacterial recognition by microglia, which occurs, in part, via CD14. 16473828_TLR3 was localized in the lysosomes as was CD14 once the cells took up polyinosine-polycytidylic acid (pIpC). We also observed that internalized pIpCsf colocalized with CD14 and TLR3. Consequently, CD14 mediates pIpC uptake and enhances TLR3 signaling. 16480837_CD14-mediated LPS signaling pathway may play a role in the regulation of NF-kappaB alternative splicing in the lungs after injury 16574244_Elevated circulating levels of LBP and constitutively higher cell surface expression of toll-like receptor 4 and CD14 on macrophages in males could result in the observed sexual dimorphism in lipopolysaccharides-induced inflammatory mediator production 16995947_Involvement of CD14 during chlamydial infection impedes infection resolution but this does not affect the sequela of infertility as assessed by hydrosalpinx formation 17384086_These findings demonstrate that CD14 is critical to lipopolysaccharides-induced airway disease and that macrophage CD14 is sufficient to initiate neutrophil recruitment into the airways. 17474083_Ganoderma lucidum polysaccharides increased macrophage surface expression of CD14 to promote lipopolysaccharide binding/clearance. 17825924_Plays a limited role in host defense against infection with influenza A virus. 17950615_The lower expression of the CD14 and TLR-4/MD-2 receptors may be partly responsible for the immunodeficiency observed in the malnourished mice. 18027846_Our findings suggest that LPS regulates N/OFN expression in sensory neurons via a novel combination of LPS receptor components and demonstrate for the first time a direct action of a key initiator of innate immune responses on neurons. 18057002_structural analysis of CD14 and discussion of the role of disulfide bonds and N-linked glycosylation 18156186_Migration of neutrophils in the lymphoid organs and in the periphery is regulated differently by a signal transduced by CD14. 18339426_PI3K-Akt axis is an important component of the TLR4-dependent antiviral mechanism. 18383566_reduced nitric oxide production in response to lipopolysaccharide (LPS) may depend on low expression of CD14 on the cell surface of skeletal muscle, and that it may enhance LPS-induced MCP-1 production 18461018_Our data suggest that signaling through the CD14 pathway plays an obligate role in cardiac inflammation/dysfunction which occurs after major burn injury. 18462086_The present study demonstrated the role of CD14 and TLR2 in the recognition and control of Listeria infection and host resistance. 18686098_There is an augmented expression of CD14 in the substantia nigra and caudate putamen in Parkinson Disease animals compared to controls. 18701460_Beta2-integrin-induced p38 MAPK activation is a key mediator in the CD14/TLR4/MD2-dependent uptake of lipopolysaccharide by hepatocytes 18761356_These results suggest that CD14-mediated toll-like receptor activation might contribute to the cardiovascular and metabolic complications of obesity. 18855560_This study found that CD14 is crucially involved in the recognition of B. pseudomallei by innate immune cells but plays a remarkable detrimental role in the host response against B. pseudomallei. 18976692_CD14 plays a contributing role in TLR4-dependent nerve injury-induced neuropathic pain 18988922_Myeloid differentiation protein-2 has an important role in the CD14-independent LPS-mediated cascade of neutrophil influx 19160083_Highest expressing lines had the least edema, suggesting the presence of sCD14 had an effect on reducing the inflammatory response to E. coli, thus, possibly protecting against gland tissue damage. 19521507_results show MARCO-expressing macrophages secrete pro-inflammatory cytokines in response to Mycobacterium tuberculosis trehalose 6,6'-dimycolate(TDM) by cooperation between MARCO & TLR2/CD14; MARCO identified as novel component required for TLR signaling 19525933_findings reveal novel aspects of molecular signalling triggered by LPS in dendritic cells, and identify a new role for CD14: the regulation of the dendritic cell life cycle through NFAT activation 19540594_Results show that stimulation of murine bone marrow-derived DC with rHagB leads to upregulation of the costimulatory molecules and ERK MAP kinases are absolutely dependent on TLR4 and CD14. 19637338_this study shows that Cd14-promoter polymorphisms affect CD14 expression and confirms the protective effect of CD14 against experimental inflammatory bowel disease. 19666536_CD14-Mac-1 interactions have roles in Bacillus anthracis spore internalization by macrophages 19710907_deletion of the CD14 gene not only prevented the onset of disease in a majority of susceptible mice (only 21% of CD14-deficient compared to 80% of wildtype mice developed ECM, pCD14->NF-kappaB mediated inflammatory response during stroke. 23508573_Signaling of HMGB1 through toll-like receptor 4 in macrophages requires CD14. 23537804_Findings link CD14 innate immune signaling via a novel IL-6 amplification loop to adventitial macrophage precursor recruitment in the pathogenesis of abdominal aortic aneurysm. 23548899_CD14 protein has a role in immune recognition of Salmonella curli fibers 23589304_Lgr4/Gpr48 negatively regulates TLR2/4-associated pattern recognition and innate immunity by targeting CD14 expression. 23656919_CD14 is actively involved in hepatic I/R injury. 24062059_CD14 knockdown significantly changed the expression of 199a-3p, miR-199a-5p, and miR-21-5p in RAW264.7 cells 24089559_CD14 plays a role in the disease progression by suppressing anti-inflammatory responses in the brain in the early stage of infection. 24116867_VB-201 may counter inflammation where TLR-2 and/or CD14 complicity is essential, and is therefore beneficial for the treatment of atherosclerosis. 24489448_CD14 together with SR participates in the binding of high doses of sLPS 24516606_We demonstrated that TLR2, TLR4 and CD14 receptors sense Tityus serrulatus venom (TsV) and its major component, toxin 1 (Ts1), to mediate cytokine and lipid mediator production. 24659539_Mouse astrocytes expressed CD14 diffently than human expression of CD14 in astrocytes. 24703105_Data show that ingestion of sCD14 resulted in blood sCD14 levels up. 24790148_specific blockade of CD14 and C5 might represent a promising new therapeutic strategy for treatment of polymicrobial sepsis. 24866794_Collectively, these data identify a previously unrecognized role for CD14 in regulating macrophage plasticity and CD4(+) T cell biasing during Schistosoma mansoni infection. 24891322_Mechanistically, mDia1 deficiency led to a downregulation of membrane-associated genes and a specific upregulation of CD14 messenger RNA in granulocytes, but not in other lineages. 25431457_fucosyllactose directly inhibits lipopolysaccharide-mediated inflammation during E. coli infection of intestinal epithelial cells through attenuation of CD14 induction. 25450368_Gene expression of the anti-inflammatory cytokine IL-13 in prion-infected CD14(-/-) mice was temporarily upregulated at 75dpi, whereas IL-13 gene expression was not upregulated in prion-infected WT mice. 25497142_27OHChol can prime monocytes/macrophages by up-regulation of CD14 such that LPS-mediated inflammatory reaction is accelerated, thereby contributing to aggravated development of atherosclerotic lesions. 25633197_CD14-mediated lipid signaling induced epithelial apoptosis, whereas TLR4 antagonistically promoted cell survival and cancer development. 25825741_Data show that guanine nucleotide-binding protein G(i) subunit alpha-1 and alpha-3 (Galphai1/3) can interact with CD14 antigen/Grb2-associated binding protein Gab1, which modulates macrophage polarization in vitro and in vivo. 25862090_In acute lung injury, lipopolysaccharide induced alveolar macrophage necrosis via CD14 and the P2X7 receptor leading to interleukin-1alpha release. 25869127_microglial HSPGs facilitate CD14-dependent TLR4 activation and that heparanase can modulate this mechanism 25965346_studies revealed a novel physical association between SP-R210S, CD14, and SR-A leading to an enhanced response to LPS, and found that SP-R210L and SP-R210S regulate internalization of CD14 via distinct macropinocytosis-like mechanisms 26106158_Data indicate that Toll-like receptor 4 (TLR4) endocytosis and the TIR-domain-containing adapter-inducing IFN-beta (TRIF)-signaling pathway in macrophages during endotoxin tolerance in the absence of cluster of differentiation 14 (CD14). 26683584_Results demonstrate remarkable sophistication of microglial CD14 in enabling, facilitating and moderating innate immune responses to infectious and non-infectious CNS threats of diverse kinds 27228163_these results indicate that CD14 is a co-receptor of TLR4 in the S100A9-induced cytokine response. 27418354_Stimulation of murine peritoneal macrophages with LPS induces biphasic accumulation of PI(4,5)P2 with peaks at 10 and 60-90 min that was abrogated when CD14 was removed from the cell surface. 27510408_These findings suggested that the degree of lung injury was reduced during the acute inflammatory reaction when NFkappaB was inhibited, and that the expression of sphingomyelin synthase 2 may affect the induction of the NFkappaB pathway by lipopolysaccharide through CD14. 27908440_this study shows that lipid rafts may serve as sites in which LPS receptors (CD14) are sorted for endocytosis, rather than being platforms for the assembly of TLR4-centered signaling complexes 28443393_this paper shows that CD14 gene silencing alters the microRNA expression profile of RAW264.7 cells stimulated by Brucella melitensis infection 28705936_Data (including data from studies conducted in cells from knockout mice) suggest that signaling via Lpar1, Cd14, and Scara1 mediates uptake of oxidized LDL by macrophages leading to foam cell formation; lysophosphatidic acid (LPA) induces expression of Cd14 and Scara1 in macrophages. (Lpar1 = LPA receptor 1; Cd14 = monocyte differentiation antigen CD14; Scara1 = scavenger receptor class A type I) 28765651_CD14 is critical for ArtinM-induced macrophage activation, providing fundamental insight into the design of anti-infective therapies based on carbohydrate recognition 28963650_Investigations on the variant of CD14 gene revealed negative association among ischemic stroke patients; however, a significant association was observed for hemorrhagic stroke following dominant and recessive genotypic model. 29045901_Data show that multiple phagocytes are capable of hyperactivation in response to oxPAPC, with CD14 antigen acting as the earliest regulator in this process, serving to capture and transport these lipids to promote inflammatory cell fate decisions. 29128406_the functional activity of TLR2, cluster of differentiation 14 (CD14), and myeloid differentiation primary response gene 88 (MyD88) molecules in the recognition of C. albicans by gingival fibroblast, was investigated. 29335601_Overall, CD14/TLR4 signalling seems to be critical for intestinal barrier function and for the crosstalk between B cells and the epithelium, underlining that CD14 serves as a protective modulator of intestinal homeostasis. 29755470_this study provides major insights into molecular mechanisms connecting CD14 and CD36 with differential eicosanoid metabolism and inflammation mediated by IL-1beta. 30485272_CD14 plays an important role in progression of structural and functional features of osteoarthritis in the destabilization of the medial meniscus model. 30529429_CD14 expression on macrophages is necessary for airway inflammation but not for viral pathogenesis in allergic hosts. 31142629_CD14 dictates differential activation of mesenchymal stromal cells through AKT, NF-kappaB and P38 signals. 31955888_Complement component C3 and the TLR co-receptor CD14 are not involved in angiotensin II induced cardiac remodelling. 33168941_Lateral diffusion of CD14 and TLR2 in macrophage plasma membrane assessed by raster image correlation spectroscopy and single particle tracking. 36280620_CD14 signaling mediates lung immunopathology and mice mortality induced by Achromobacter xylosoxidans. |
ENSG00000170458 |
CD14 |
45.578266 |
0.3126339213 |
-1.677454 |
0.38517101 |
18.821227 |
0.00001435604299912228114916544485790694807292311452329158782958984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001032442148076631555997720313122556490270653739571571350097656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.4627557149499 |
6.06860944951117 |
71.8500270942394 |
13.3932545017429 |
| ENSMUSG00000051483 |
12408 |
Cbr1 |
protein_coding |
P48758 |
FUNCTION: NADPH-dependent reductase with broad substrate specificity. Catalyzes the reduction of a wide variety of carbonyl compounds including quinones, prostaglandins, menadione, plus various xenobiotics. Catalyzes the reduction of the antitumor anthracyclines doxorubicin and daunorubicin to the cardiotoxic compounds doxorubicinol and daunorubicinol (By similarity). Can convert prostaglandin E to prostaglandin F2-alpha (By similarity). Can bind glutathione, which explains its higher affinity for glutathione-conjugated substrates. Catalyzes the reduction of S-nitrosoglutathione. In addition, participates in the glucocorticoid metabolism by catalyzing the NADPH-dependent cortisol/corticosterone into 20beta-dihydrocortisol (20b-DHF) or 20beta-corticosterone (20b-DHB), which are weak agonists of NR3C1 and NR3C2 in adipose tissue (PubMed:28878267, PubMed:33785425). {ECO:0000250|UniProtKB:P16152, ECO:0000250|UniProtKB:Q28960, ECO:0000269|PubMed:28878267, ECO:0000269|PubMed:33785425}. |
Acetylation;Cytoplasm;Direct protein sequencing;Lipid metabolism;NADP;Oxidoreductase;Phosphoprotein;Reference proteome |
|
|
mmu:12408; |
cytoplasm [GO:0005737]; intracellular membrane-bounded organelle [GO:0043231]; microvillus [GO:0005902]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; subapical complex [GO:0035003]; 15-hydroxyprostaglandin dehydrogenase (NADP+) activity [GO:0047021]; 15-hydroxyprostaglandin-D dehydrogenase (NADP+) activity [GO:0047020]; alcohol dehydrogenase (NADP+) activity [GO:0008106]; carbonyl reductase (NADPH) activity [GO:0004090]; oxidoreductase activity [GO:0016491]; oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor [GO:0016655]; oxidoreductase activity, acting on the CH-OH group of donors, NAD or NADP as acceptor [GO:0016616]; prostaglandin-E2 9-reductase activity [GO:0050221]; glucocorticoid metabolic process [GO:0008211]; positive regulation of reactive oxygen species metabolic process [GO:2000379]; vitamin K metabolic process [GO:0042373]; xenobiotic metabolic process [GO:0006805] |
20728534_CBR1 attenuates apoptosis and promotes cell survival in pancreatic beta-cell lines under glucotoxic and glucolipotoxic conditions via reducing ROS generation 23732109_CBR1 plays a critical role in controlling redox balance and detoxifying lipid peroxidation during muscle differentiation and regeneration. 28878267_These findings suggest that CBR1 generating 20beta-dihydrocortisol is a novel pathway modulating GR activation and providing enzymatic protection against excessive GR activation in obesity. 31955137_Carbonyl Reductase 1 Plays a Significant Role in Converting Doxorubicin to Cardiotoxic Doxorubicinol in Mouse Liver, but the Majority of the Doxorubicinol-Forming Activity Remains Unidentified. 32843708_Impairment of spatial memory accuracy improved by Cbr1 copy number resumption and GABAB receptor-dependent enhancement of synaptic inhibition in Down syndrome model mice. 33785425_Carbonyl reductase 1 amplifies glucocorticoid action in adipose tissue and impairs glucose tolerance in lean mice. |
ENSG00000159228 |
CBR1 |
179.048709 |
2.2153419641 |
1.147529 |
0.20480703 |
31.055381 |
0.00000002507701865712385841490625402842862268215640142443589866161346435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000002859080450488851824495176168206000966165447607636451721191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
249.235188365551 |
35.5007419465579 |
112.504160716331 |
12.3184129683593 |
| ENSMUSG00000051486 |
93882 |
Pcdhb11 |
protein_coding |
Q91UZ8 |
Human_homologues FUNCTION: Potential calcium-dependent cell-adhesion protein. May be involved in the establishment and maintenance of specific neuronal connections in the brain. |
Calcium;Cell adhesion;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:93882; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell adhesion [GO:0007155]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
Human_homologues 34419445_Weighted genes associated with the progression of retinoblastoma: Evidence from bioinformatic analysis. |
ENSG00000113209 |
PCDHB5 |
27.542961 |
0.3653199792 |
-1.452767 |
0.48918213 |
8.792896 |
0.00302405814346524319871645225532574841054156422615051269531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0120930568761518988929459794690046692267060279846191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.7059805969809 |
4.73692429835937 |
37.5177416487566 |
9.09512180032236 |
| ENSMUSG00000051517 |
230098 |
Arhgef39 |
protein_coding |
Q66JY6 |
FUNCTION: Promotes cell proliferation. {ECO:0000250}. |
Alternative splicing;Cell membrane;Guanine-nucleotide releasing factor;Membrane;Reference proteome |
|
|
mmu:230098; |
plasma membrane [GO:0005886]; guanyl-nucleotide exchange factor activity [GO:0005085]; positive regulation of cell migration [GO:0030335] |
Human_homologues 22327280_C9orf100 plays a potential oncogenic role in the development and metastasis of HCC. 28871449_findings suggest that ARHGEF39 may contribute to cell proliferation and migration in gastric cancer via a possible mechanism involving Akt signaling. 29382922_ARHGEF39 promotes tumor growth and invasion by activating the Rac1-P38-ATF2 signaling pathway, as well as increasing the expression of Cyclin A2, Cyclin D1, and MMP2 in NSCLC cells. 34731623_FARP1, ARHGEF39, and TIAM2 are essential receptor tyrosine kinase effectors for Rac1-dependent cell motility in human lung adenocarcinoma. |
ENSG00000137135 |
ARHGEF39 |
525.557037 |
2.6580811210 |
1.410385 |
0.12724914 |
120.712571 |
0.00000000000000000000000000044170858333392735102573677941070691823030981270421616551358942361766424635274863597089733957545831799507141113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000297703767316034579255755830186999906793558773950777489612315703341202752923999597101101244334131479263305664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
763.441141919058 |
67.4246709764263 |
287.215140228068 |
19.7706725724758 |
| ENSMUSG00000051590 |
22625 |
Map3k19 |
protein_coding |
E9Q3S4 |
|
ATP-binding;Kinase;Nucleotide-binding;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:22625; |
ATP binding [GO:0005524]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; protein phosphorylation [GO:0006468]; signal transduction [GO:0007165] |
Human_homologues 27935962_Mitogen-activated protein kinase kinase kinase 19 (MAP3K19) is overexpressed in the lungs of chronic obstructive pulmonary disease (COPD) patients. 32358059_The protein kinase MAP3K19 phosphorylates MAP2Ks and thereby activates ERK and JNK kinases and increases viability of KRAS-mutant lung cancer cells. |
ENSG00000176601 |
MAP3K19 |
53.882586 |
7.7375518738 |
2.951877 |
0.40113175 |
54.872606 |
0.00000000000012860106176362127595872881139671766278142692185859630171762546524405479431152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000028554684734453052310414819645467286637202652954670156759675592184066772460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
95.3764436406815 |
26.8617944388676 |
12.3264367329877 |
2.91363446067387 |
| ENSMUSG00000051920 |
239405 |
Rspo2 |
protein_coding |
Q8BFU0 |
FUNCTION: Activator of the canonical Wnt signaling pathway by acting as a ligand for LGR4-6 receptors. Upon binding to LGR4-6 (LGR4, LGR5 or LGR6), LGR4-6 associate with phosphorylated LRP6 and frizzled receptors that are activated by extracellular Wnt receptors, triggering the canonical Wnt signaling pathway to increase expression of target genes. Also regulates the canonical Wnt/beta-catenin-dependent pathway and non-canonical Wnt signaling by acting as an inhibitor of ZNRF3, an important regulator of the Wnt signaling pathway. Probably also acts as a ligand for frizzled and LRP receptors (PubMed:21693646). During embryonic development, plays a crucial role in limb specification, amplifying the Wnt signaling pathway independently of LGR4-6 receptors, possibly by acting as a direct antagonistic ligand to RNF43 and ZNRF3, hence governing the number of limbs an embryo should form (By similarity). {ECO:0000250|UniProtKB:Q6UXX9, ECO:0000269|PubMed:21693646}. |
3D-structure;Developmental protein;Disulfide bond;Glycoprotein;Heparin-binding;Reference proteome;Secreted;Sensory transduction;Signal;Wnt signaling pathway |
|
|
mmu:239405; |
cell surface [GO:0009986]; extracellular region [GO:0005576]; heparin binding [GO:0008201]; signaling receptor binding [GO:0005102]; bone mineralization [GO:0030282]; canonical Wnt signaling pathway [GO:0060070]; dopaminergic neuron differentiation [GO:0071542]; embryonic forelimb morphogenesis [GO:0035115]; embryonic hindlimb morphogenesis [GO:0035116]; epithelial tube branching involved in lung morphogenesis [GO:0060441]; limb development [GO:0060173]; lung growth [GO:0060437]; negative regulation of odontogenesis of dentin-containing tooth [GO:0042489]; osteoblast differentiation [GO:0001649]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; positive regulation of Wnt signaling pathway [GO:0030177]; trachea cartilage morphogenesis [GO:0060535] |
17904116_The findings suggest that Rspo2 expression in the apical ectodermal ridge (AER) is required for AER maintenance likely by regulating Wnt/beta-catenin signaling. 18067586_This study shows that Rspo2 is critical for maintenance of the AER and for growth and patterning in limb development. 18256198_Lrp6-mediated R-spondin 2 signaling through the canonical Wnt pathway is required for normal morphogenesis of the respiratory tract and limbs. 19213727_Wnt11 signals through beta-catenin, activating Rspo2 expression, which is then required for Wnt11-mediated osteoblast maturation. 19233133_These findings indicate that Rspo2 regulates midfacial, limb, and lung morphogenesis during development through the Wnt/beta-catenin signaling. 19385064_The Rspo2 mutants had markedly reduced potency relative to the wild-type protein,demonstrating for the first time specific amino acid residues in Rspos that are critical for beta-catenin signaling. 21237142_Rspo2 is a mesenchyme-derived factor that plays critical roles in regulating branchial arch 1 patterning and morphogenesis through ectodermal-mesenchymal interaction and a novel genetic factor for cleft palate. 21732367_Results indicate that Rspo2 and Wnt1 have mutually distinct effects on mammary epithelial cell growth and these effects are context-dependent. 22238613_Recombinant R-spondin2 and Wnt3a up- and down-regulate novel target genes in C57MG mouse mammary epithelial cells. 22610508_Rspo2 mutation in combination with Rspo3 mutation causes severe limb truncation. 23192966_Rspo2 is required to limit the number of outer hair cells to three rows and for optimal arrangement of peripheral nerve fibers in the developing cochlea. 23407710_Oocyte-derived R-spondin2 is a paracrine factor essential for primary ovarian follicle development 23695692_R-spondin2 is a key molecular link between infection and intestinal homoeostasis. 24005619_R-spondin 2 homolog (Xenopus laevis) is identified as an essential regulator of Citrobacter rodentium infection susceptibility in mice 24344199_identify the transforming growth factor beta antagonist follistatin as a key mediator of RSPO-LGR4 signaling in myogenic differentiation 24852883_These findings suggest that R-Spondin2 may promote hepatic stellate cell activation by enhancing the canonical Wnt pathway. 25188337_The endogenously expressed Lgr4 may act as an antagonist molecule that helps to fine-tune the R-spondin/norrin-mediated Lgr4-Wnt signaling during gonadal development. 26123262_High resolution crystal structure of an ectodomain variant of human LGR5 (hLGR5ecto) complexed with a signalling competent fragment of mouse Rspo2. 27012200_Rspo2 activates Wnt/beta-catenin signaling to reduce Col2a1 and Sox9 and to facilitate differentiation of proliferating chondrocytes into hypertrophic chondrocytes in growth cartilage 27140682_LGR4 acted as a key receptor for Rspo2 to promote osteogenesis. 27328992_Data report that Rspo2, is highly expressed in the spinal motor neuron (SMN) and binds to its receptor Lgr5, at the neuromuscular junction (NMJ). Rspo2 enhances the LRP4/MuSK signaling via Lgr5 in an agrin-independent manner and promotes AChR clustering. In addition, the loss of Rspo2 in mice compromises AChR clustering, the ultrastructure of the NMJ, and neuromuscular signal transduction. 28695896_Rspo2 and Rspo3 fusion events are sufficient to initiate hyperplasia and tumour development. 30146491_RSPO2, but not SPON1, specifically promoted the differentiation of midbrain dopaminergic (mDA) neuroblasts into mDA neurons in mouse primary cultures and embryonic stem cells (ESCs). 30206360_These results suggest that SMN-derived Rspo2 plays a major role in AChR clustering and NMJ formation in the postsynaptic region, and muscle-derived Rspo2 also plays a substantial role in juxtaposition of the active zones and synaptic folds. 32019591_R-spondin 2 mediates neutrophil egress into the alveolar space through increased lung permeability. 32341451_R-spondin2 signaling is required for oocyte-driven intercellular communication and follicular growth. 32750106_Role of R-spondin 2 in arterial lymphangiogenesis and atherosclerosis. 33443181_R-spondin substitutes for neuronal input for taste cell regeneration in adult mice. 33712657_Synergistic roles of Wnt modulators R-spondin2 and R-spondin3 in craniofacial morphogenesis and dental development. 34175894_DNA methylation mediated RSPO2 to promote follicular development in mammals. 35027768_Identification of a regulatory pathway inhibiting adipogenesis via RSPO2. 36175067_Synovial fibroblasts assume distinct functional identities and secrete R-spondin 2 in osteoarthritis. |
ENSG00000147655 |
RSPO2 |
76.427694 |
5.0952212525 |
2.349145 |
0.32470735 |
52.132621 |
0.00000000000051875948601398256032560658521369300407062585822615119468537159264087677001953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000107360658844632914943279870950404561360386201940286809985991567373275756835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
135.929323416037 |
30.8525594849222 |
26.6778058655823 |
4.87323125621653 |
| ENSMUSG00000052013 |
208154 |
Btla |
protein_coding |
A0A0R4J1A0 |
Human_homologues FUNCTION: Inhibitory receptor on lymphocytes that negatively regulates antigen receptor signaling via PTPN6/SHP-1 and PTPN11/SHP-2 (PubMed:12796776, PubMed:14652006, PubMed:15568026, PubMed:18193050). May interact in cis (on the same cell) or in trans (on other cells) with TNFRSF14 (PubMed:19915044). In cis interactions, appears to play an immune regulatory role inhibiting in trans interactions in naive T cells to maintain a resting state. In trans interactions, can predominate during adaptive immune response to provide survival signals to effector T cells (PubMed:19915044). {ECO:0000269|PubMed:12796776, ECO:0000269|PubMed:14652006, ECO:0000269|PubMed:15568026, ECO:0000269|PubMed:18193050, ECO:0000269|PubMed:19915044}. |
Membrane;Proteomics identification;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
|
plasma membrane [GO:0005886]; signaling receptor activity [GO:0038023]; immune response-regulating cell surface receptor signaling pathway [GO:0002768] |
12796776_BTLA is a third inhibitory receptor on T lymphocytes with similarities to cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and programmed death 1 (PD-1). 15128774_BTLA is expressed by all mature lymphocytes, splenic macrophages, and mature, but not immature bone marrow-derived dendritic cells. BTLA is implicated as a negative regulator of the activation and/or function of various hemopoietic cell types. 15647361_Binding of HVEM to BTLA attenuates T cell activation, identifying HVEM/BTLA as a coinhibitory receptor pair. 15749870_existence of three distinct BTLA alleles among 23 murine strains, differing both in Ig domain structure and cellular distribution of expression on lymphoid subsets 16272294_BTLA adds to the growing list of cell surface proteins that are potential targets to down-modulate T cell function. 16547224_these receptors are critical determinants of the duration of allergic airway inflammation 16805995_BTLA had a regulatory effect on the expression of B7 on dendritic cells. 17206146_mice deficient in full-length BTLA or its ligand, herpesvirus entry mediator, had increased number of memory CD8(+) T cells 17541277_BTLA inhibits antigen-induced eosinophil recruitment into the airways by preventing IL-5 production from Th2 cells. 17607320_These results suggest that BTLA functions to regulate T-cell signaling by controlling the phosphorylated form of TCRzeta accumulation in the lipid raft. 17785848_BTLA-herpesvirus entry mediator (HVEM) interactions are functionally involved in T cell regulation during Plasmodium berghei strain ANKA blood-stage malaria and regulate sequestration of T cells in brain capillaries. 18097025_The LTbetaR and HVEM-BTLA pathways form an integrated signaling network regulating DC homeostasis. 18178834_BTLA employs a conserved binding mode for herpesvirus entry mediator recognition. 18519647_HVEM triggers inhibitory signals by acting as a ligand that binds to B and T lymphocyte attenuator (BTLA), an immunoglobulin super family member. 18668554_BTLA plays an important role in the maintenance of immune tolerance and the prevention of autoimmune diseases. 18713967_role of BTLA on the proliferation, recruitment, and survival of T cells in response to inhaled allergen 19342624_demonstrate an important role for BTLA in the induction of peripheral tolerance of both CD4(+) and CD8(+) T cells in vivo 19535622_B and T lymphocyte attenuator (BTLA) coinhibitory signaling of natural killer T (NKT) cells is required to temper early inflammation in a model of acute hepatitis. 19567411_role in down-modulating immune responses[REVIEW] 19587015_engagement of the newly discovered coinhibitory receptor B and T lymphocyte attenuator (BTLA) by herpesvirus entry mediator (HVEM) is critical for negatively regulating early host immunity against intracellular bacteria. 19892849_The subcellular localization of BTLA in mouse T cells in a steady state, as well as upon activation, was examined. 19915044_the HVEM-BTLA cis-complex competitively inhibits HVEM activation by ligands expressed in the surrounding microenvironment, thus helping maintain T cells in the naive state. 19923459_The extracellular domain of BTLA binds herpesvirus entry mediator (HVEM), blocks BTLA-HVEM interactions and significantly improves antitumor immunity in cervical cancer when combined with heat shock protein (HSP)70 vaccine. 19949073_BTLA functions as the inhibitory coreceptor of NKT cells 20660710_BTLA suppresses IgG2a and IgG2b production in vivo by inhibiting interleukin (IL)-21 production from follicular T helper (Th) cells. 21042564_The effect of the B and T lymphocyte attenuator (BTLA; CD272) on cluster of differentiation (CD)4(+) T cell-mediated corneal immunopathology during murine herpetic stromal keratitis, was investigated. 21078889_Targeting of B and T lymphocyte associated (BTLA) prevents graft-versus-host disease without global immunosuppression. 21220749_dichotomous functions of BTLA in GVHD to serve as a costimulatory ligand of HVEM and to transmit inhibitory signal as a receptor 21978997_BTLA receptor is a potential immunoregulatory target for the modulation of cytotoxic T-lymphocyte-mediated 21998455_During increased resistance to malaria infection, BTLA regulates production of proinflammatory cytokines in a T cell-intrinsic way, while B cells intrinsically regulate the production of nonlethal Plasmodium yoelii 17NL-specific antibodies. 22459947_These findings support role for BTLA and/or HVEM as potential, novel diagnostic markers of innate immune response/status and as therapeutic targets of sepsis. 22490863_Combined blockade of BTLA and herpesvirus entry mediator (HVEM) does not inhibit donor T cell infiltration into graft-versus-host reaction organs; instead, it decreases the functional activity of the alloreactive T cells. 22637698_BTLA promotes the pathogenesis of virus-induced fulminant hepatitis by enhancing macrophage viability and function. Targeting BTLA may be a novel strategy for the treatment of FH. 23479601_B and T lymphocyte attenuator inhibits LPS-induced endotoxic shock by suppressing Toll-like receptor 4 signaling in innate immune cells. 24315996_by coordinating expression of BTLA, RORgammat and IL-7 balance suppressive and activation stimuli to regulate gammadelta T cell homeostasis and inflammatory responses 25011109_This study uncovers a BTLA-mediated strategy used by the host that permits Listeria proliferation to enable increasing T cell responses for long-term protection. 25595777_results support a model whereby BTLA on innate leukocytes is triggered by HVEM and delivers negative signals into BTLA(+) cells, thereby interfering with the protective immune response to this intestinal parasite. 25973010_Our findings indicate that BTLA may be involved in the control of inflammatory responses through increasing Foxp3 expression, rather than attenuating IL-17 production, in dextran sulfate sodium-induced colitis. 26924526_these data indicate that HVEM/BTLA interactions are dispensable for the formation of de novo host antidonor isotype-specific antibodies following skin transplantation 27350630_this study shows that miR-155 is involved in the inhibition of BTLA during CD4+ T cell activation 27793593_Dendritic cells require BTLA and HVEM to actively adjust tolerizing T cell responses under steady-state conditions. 28164546_The percentage of circulating BTLA+CD4+ lymphocytes was significantly higher in mice with LPS-induced acute lung inflammation. 28393074_these data indicate that CCR7 and BTLA cooverexpression imparts an intermediate immune phenotype in mmature dendritic cells when compared to that in CCR7- or BTLA-expressing counterparts that show a more immunocompetent or immunotolerant phenotype 29518903_these data support a role of BTLA on type I NKT cells in limiting anti-tumor immunity. 30134817_Soluble BTLA is produced as a result of alternative RNA splicing. This isoform of BTLA has biological significance through changes in cellular proliferation and can predict the diagnosis of sepsis. 31204070_these results suggest that BTLA functions as a cell-extrinsic suppressor of germinal center B cell lymphomagenesis 33348294_B and T lymphocyte attenuator regulates autophagy in mycobacterial infection via the AKT/mTOR signal pathway. 34315113_Simultaneous transduction of dendritic cells with A20 and BTLA genes stimulates the development of stable and efficient tolerogenic dendritic cells and induces regulatory T cells. 35185877_The HVEM-BTLA Immune Checkpoint Restrains Murine Chronic Cholestatic Liver Injury by Regulating the Gut Microbiota. |
ENSG00000186265 |
BTLA |
518.403698 |
0.2208561349 |
-2.178821 |
0.14693685 |
211.806839 |
0.00000000000000000000000000000000000000000000000554207789429640643301998806550739624787725678819837674025724408391906083556809231700244421065837362322151610931066125485290607688426689492189325392246246337890625000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000008441693048592286667714500687702222661270640901041532487650594733698459283557106641614300148886299776887564783059100283202358383505270467139780521392822265625000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
183.015200132602 |
18.6721324003862 |
828.662514510698 |
59.3910092977691 |
| ENSMUSG00000052062 |
72823 |
Pard3b |
protein_coding |
Q9CSB4 |
FUNCTION: Putative adapter protein involved in asymmetrical cell division and cell polarization processes. May play a role in the formation of epithelial tight junctions (By similarity). {ECO:0000250}. |
3D-structure;Alternative splicing;Cell cycle;Cell division;Cell junction;Membrane;Phosphoprotein;Reference proteome;Repeat;Tight junction |
|
|
mmu:72823; |
adherens junction [GO:0005912]; apical junction complex [GO:0043296]; apical plasma membrane [GO:0016324]; bicellular tight junction [GO:0005923]; cell cortex [GO:0005938]; cell junction [GO:0030054]; endomembrane system [GO:0012505]; protein-containing complex [GO:0032991]; phosphatidylinositol binding [GO:0035091]; cell adhesion [GO:0007155]; cell cycle [GO:0007049]; cell division [GO:0051301]; establishment of cell polarity [GO:0030010]; establishment of centrosome localization [GO:0051660]; establishment or maintenance of epithelial cell apical/basal polarity [GO:0045197]; microtubule cytoskeleton organization [GO:0000226]; protein localization [GO:0008104] |
24305829_Atp6ap2/(P)RR interacted with partitioning defective 3 homolog (PAR3) protein, which is known to function in the Par-atypical protein kinase C 24859006_Par3L is expressed by multipotent stem cells in the terminal end buds of murine mammary glands. Ablation of Par3L resulted in rapid and profound stem cell loss. |
ENSG00000116117 |
PARD3B |
111.319811 |
0.4145680940 |
-1.270319 |
0.28020006 |
20.425007 |
0.00000620141461848134078360022039544396932342351647093892097473144531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000477552818345337645321475872339789248144370503723621368408203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.2887350574839 |
12.5647978534358 |
155.074005915037 |
21.7005936504491 |
| ENSMUSG00000052105 |
68617 |
Mtcl1 |
protein_coding |
Q3UHU5 |
FUNCTION: Microtubule-associated factor involved in the late phase of epithelial polarization and microtubule dynamics regulation. Plays a role in the development and maintenance of non-centrosomal microtubule bundles at the lateral membrane in polarized epithelial cells. {ECO:0000269|PubMed:23902687}. |
Alternative splicing;Cell membrane;Coiled coil;Cytoplasm;Cytoskeleton;Membrane;Phosphoprotein;Reference proteome |
|
|
mmu:68617; |
apical plasma membrane [GO:0016324]; apicolateral plasma membrane [GO:0016327]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; extracellular space [GO:0005615]; lateral plasma membrane [GO:0016328]; microtubule bundle [GO:0097427]; midbody [GO:0030496]; spindle pole [GO:0000922]; microtubule binding [GO:0008017]; protein homodimerization activity [GO:0042803]; establishment or maintenance of epithelial cell apical/basal polarity [GO:0045197]; microtubule bundle formation [GO:0001578]; positive regulation of protein targeting to membrane [GO:0090314]; regulation of autophagy [GO:0010506] |
28283581_These results indicate that MTCL1-mediated formation of stable microtubule bundles is crucial for maintenance of AnkG localization. |
ENSG00000168502 |
MTCL1 |
221.470245 |
3.3860751960 |
1.759614 |
0.20348281 |
73.442862 |
0.00000000000000001035919215200998236681497001728393883328426016890823693761269908009126083925366401672363281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000003378826870651307024625140066860417604870091572068918628346523291838821023702621459960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
329.356859892024 |
46.6078283301525 |
97.2680288607941 |
10.7614635236625 |
| ENSMUSG00000052387 |
226025 |
Trpm3 |
protein_coding |
F7CJ77 |
Human_homologues FUNCTION: Calcium channel mediating constitutive calcium ion entry. Its activity is increased by reduction in extracellular osmolarity, by store depletion and muscarinic receptor activation. In addition, forms heteromultimeric ion channels with TRPM1 which are permeable for calcium and zinc ions (PubMed:21278253). {ECO:0000269|PubMed:12672799, ECO:0000269|PubMed:12672827, ECO:0000269|PubMed:21278253}. |
Coiled coil;Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
|
plasma membrane [GO:0005886]; cation channel activity [GO:0005261]; cation transmembrane transport [GO:0098655]; cation transport [GO:0006812]; protein tetramerization [GO:0051262] |
15824111_the divalent cation selectivity of TRPM3 channels is regulated by altenrative splicing 18978782_TRPM3 is an essential component of an ionotropic steroid receptor enabling unanticipated crosstalk between steroidal and insulin-signalling endocrine systems. 20360246_data suggest functional relevance of TRPM3 in contractile and proliferating phenotypes of vascular smooth muscle cells 20360254_These results demonstrate that TRB3 induction is a significant aspect of the ER stress response in cardiac myocytes and that TRB3 antagonizes cardiac glucose metabolism and cardiac myocyte survival. 21257751_Signal transduction of pregnenolone sulfate in insulinoma cells: activation of Egr-1 expression involving TRPM3, voltage-gated calcium channels, ERK, and ternary complex factors. 21555074_These experiments reveal an unanticipated role for TRPM3 as a thermosensitive nociceptor channel implicated in the detection of noxious heat. 22211741_data support an indirect role for TRPM3 in pupillary light responses 24623969_transcriptional control of Trpm3/miR-204 is reported. 25733887_TRPM3 activation in sensory nerve endings can contribute to neurogenic inflammation. 28122715_Primary cilia are required for the maximal osmotic response in renal epithelial cells and TRPM3 is involved in this osmoregulatory mechanism. 29539642_findings indicate that the initiation of the acute heat-evoked pain response in sensory nerve endings relies on three functionally redundant TRP channels, representing a fault-tolerant mechanism to avoid burn injury 29604058_Site-directed mutagenesis studies and patch clamp experiments led to the identification of four residues in S4 (W982, R985, D988 and G991) as crucial determinants of the properties of the alternative ion permeation pathway involved in the formation of the non-canonical pore in TRPM3. 30853321_TRPM3 is implicated in the regulation of the tone of resistance arteries. 30883612_Knocking out TRPM3 did not change the level of TRPP2 protein in the cilia of primary renal epithelial cells, so it is unlikely that the absence of functional ciliary channels results from a failure of trafficking. 31011981_TRPM3gamma variants have low channel activity compared to TRPM3alpha. 32238415_The Impact of Steroid Activation of TRPM3 on Spontaneous Activity in the Developing Retina. 32880575_Upregulation of TRPM3 in nociceptors innervating inflamed tissue. 33130130_The TRPM3 ion channel mediates nociception but not itch evoked by endogenous pruritogenic mediators. 33166100_Control of Insulin Release by Transient Receptor Potential Melastatin 3 (TRPM3) Ion Channels. 33478988_TRPM3 Channels Play Roles in Heat Hypersensitivity and Spontaneous Pain after Nerve Injury. 33484482_Mutation of the TRPM3 cation channel underlies progressive cataract development and lens calcification associated with pro-fibrotic and immune cell responses. 33658516_ATF4 selectively regulates heat nociception and contributes to kinesin-mediated TRPM3 trafficking. 35008533_TRPM3 Is Expressed in Afferent Bladder Neurons and Is Upregulated during Bladder Inflammation. 35240732_TRPM3 in the eye and in the nervous system - from new findings to novel mechanisms. 36283409_Structural and functional analyses of a GPCR-inhibited ion channel TRPM3. |
ENSG00000083067 |
TRPM3 |
225.710783 |
0.3210101436 |
-1.639309 |
0.22342294 |
52.206093 |
0.00000000000049970844334182888648752674390807457834983579259002794970001559704542160034179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000103699714018838393114081459413216158415432799344557679432909935712814331054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
105.024882022669 |
16.3297677273026 |
327.169979213209 |
36.2634661719645 |
| ENSMUSG00000052477 |
620078 |
C130026I21Rik |
protein_coding |
F7D3Q7 |
|
Reference proteome |
|
|
|
fibrillar center [GO:0001650]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
|
|
183.253463 |
4.1716275945 |
2.060610 |
0.29456702 |
46.806582 |
0.00000000000783498178948939443995629934795445420761539345733126538107171654701232910156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001410667170419650910987676926662635207287976868428813759237527847290039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
309.820503824298 |
63.6990700660631 |
74.2684951625581 |
11.7767464042347 |
| ENSMUSG00000052516 |
268902 |
Robo2 |
protein_coding |
A0A2I3BR72 |
Human_homologues FUNCTION: Receptor for SLIT2, and probably SLIT1, which are thought to act as molecular guidance cue in cellular migration, including axonal navigation at the ventral midline of the neural tube and projection of axons to different regions during neuronal development. |
Immunoglobulin domain;Membrane;Proteomics identification;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
Human_homologues hsa:6092; |
membrane [GO:0016020]; axon guidance receptor activity [GO:0008046]; brain development [GO:0007420]; spinal cord development [GO:0021510] |
12954717_Data suggest that the Slit family of axon guidance molecules (Slit 1-3) and their Robo 1 and 2 receptors contribute to the topographic targeting of basal vomeronasal axons. 14960623_The Slit receptors Robo2 and Robo3/Rig-1 are expressed in the subventricular zone and the rostral migratory stream and Slit1 and Slit2 are present in the adult septum and are responsible for both the septum and the CP repulsive activity in vitro 15091338_Robo2 single mutants show guidance defects that reveal a role for this receptor in guiding commissural axons to different positions within the ventral and lateral funiculi. 15130495_Mutants lacking ROBO2 develop supernumerary ureteric buds that remain inappropriately connected to the nephric duct; the ROBO2 signal is transduced in the nephrogenic mesenchyme. 15162513_Slit-3 and Robo-2 expression is restricted to specific, complementary subsets of mesenchyme 16829019_two differentially expressed isoforms were isolated and identified. The 5' end of mouse Robo2a lies in a |
ENSG00000185008 |
ROBO2 |
519.308973 |
4.0805926582 |
2.028779 |
0.14735129 |
183.472376 |
0.00000000000000000000000000000000000000000845860431699512233916655171168250988410066598326153058050439198649371318171088487295704759153419078833598381689240497394166595768183469772338867187500000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000010225512774322992429532793458102363708644667728328210797614813007963601987427109372387564818953086019995279976058100146474316716194152832031250000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
822.139923466016 |
84.116262300914 |
201.475616981353 |
16.257339841471 |
| ENSMUSG00000052760 |
319997 |
A630001G21Rik |
protein_coding |
Q8C9T1 |
|
Proteomics identification;Reference proteome |
|
|
|
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
|
|
222.100273 |
2.1245497963 |
1.087157 |
0.20882738 |
26.947141 |
0.00000020909622204963085630604442081287297483527254371438175439834594726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000020748883741107345206040966228977495688923227135092020034790039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
297.272273227209 |
43.1800161698155 |
139.922478515376 |
15.5390460874098 |
| ENSMUSG00000052920 |
19091 |
Prkg1 |
protein_coding |
P0C605 |
FUNCTION: Serine/threonine protein kinase that acts as key mediator of the nitric oxide (NO)/cGMP signaling pathway. GMP binding activates PRKG1, which phosphorylates serines and threonines on many cellular proteins. Numerous protein targets for PRKG1 phosphorylation are implicated in modulating cellular calcium, but the contribution of each of these targets may vary substantially among cell types. Proteins that are phosphorylated by PRKG1 regulate platelet activation and adhesion, smooth muscle contraction, cardiac function, gene expression, feedback of the NO-signaling pathway, and other processes involved in several aspects of the CNS like axon guidance, hippocampal and cerebellar learning, circadian rhythm and nociception. Smooth muscle relaxation is mediated through lowering of intracellular free calcium, by desensitization of contractile proteins to calcium, and by decrease in the contractile state of smooth muscle or in platelet activation. Regulates intracellular calcium levels via several pathways: phosphorylates IRAG1 and inhibits IP3-induced Ca(2+) release from intracellular stores, phosphorylation of KCNMA1 (BKCa) channels decreases intracellular Ca(2+) levels, which leads to increased opening of this channel. PRKG1 phosphorylates the canonical transient receptor potential channel (TRPC) family which inactivates the associated inward calcium current. Another mode of action of NO/cGMP/PKGI signaling involves PKGI-mediated inactivation of the Ras homolog gene family member A (RhoA). Phosphorylation of RHOA by PRKG1 blocks the action of this protein in myriad processes: regulation of RHOA translocation; decreasing contraction; controlling vesicle trafficking, reduction of myosin light chain phosphorylation resulting in vasorelaxation. Activation of PRKG1 by NO signaling alters also gene expression in a number of tissues. In smooth muscle cells, increased cGMP and PRKG1 activity influence expression of smooth muscle-specific contractile proteins, levels of proteins in the NO/cGMP signaling pathway, down-regulation of the matrix proteins osteopontin and thrombospondin-1 to limit smooth muscle cell migration and phenotype. Regulates vasodilator-stimulated phosphoprotein (VASP) functions in platelets and smooth muscle. {ECO:0000269|PubMed:10209042, ECO:0000269|PubMed:11055988, ECO:0000269|PubMed:19156199, ECO:0000269|PubMed:9606187, ECO:0000269|PubMed:9920894}. |
Acetylation;Allosteric enzyme;Alternative splicing;ATP-binding;cGMP;cGMP-binding;Coiled coil;Cytoplasm;Disulfide bond;Kinase;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:19091; |
cytoplasm [GO:0005737]; Golgi apparatus [GO:0005794]; plasma membrane [GO:0005886]; ATP binding [GO:0005524]; calcium channel regulator activity [GO:0005246]; cGMP binding [GO:0030553]; cGMP-dependent protein kinase activity [GO:0004692]; identical protein binding [GO:0042802]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; cGMP-mediated signaling [GO:0019934]; collateral sprouting [GO:0048668]; dendrite development [GO:0016358]; forebrain development [GO:0030900]; negative regulation of platelet aggregation [GO:0090331]; negative regulation of smooth muscle contraction [GO:0045986]; negative regulation of vascular associated smooth muscle cell migration [GO:1904753]; negative regulation of vascular associated smooth muscle cell proliferation [GO:1904706]; neuron migration [GO:0001764]; protein phosphorylation [GO:0006468]; regulation of GTPase activity [GO:0043087]; relaxation of vascular associated smooth muscle [GO:0060087]; signal transduction [GO:0007165] |
12371625_Elevated cGMP concentration may activate PKG1beta in thymus, lymph node, and spleen cells and also PKG1alpha in spleen cells 12417579_Data show that the alpha isoform of cGMP-dependent protein kinase I (cGKI) is expressed by sensory axons but not spinal cord neurons during development, and that cGMP signaling is important for axonal growth in vivo and in vitro. 12853418_Hippocampal cGKI supports an age- and protein synthesis-dependent form of hippocampal long term potentiation, whereas it is dispensable for hippocampus-dependent spatial reference and contextual memory 14568994_Ablation of CGKI in Purkinje cells of the cerebellum leads to impairment of long-term depression and cerebellar learning in knockout mice. 14597716_activation of smooth muscle cGKI contributes to the proatherogenic effect of nitric oxide 14973199_reduction of nociception in PKG-I(-/-) mice and a possible role of PKG-I in substance P synthesis. 15280395_PKG has a role in platelet secretion 15388327_These results suggest that IRAG may be involved in cGMP kinase I-dependent and -independent pathways. 15483626_Data show that the targeted deletion of exon 12 of IRAG disrupted the IRAG-IP(3)RI interaction and resulted in hypomorphic IRAG(Delta12/Delta12) mice. 15505114_We conclude that cGMP and cGMP-dependent protein kinase type I are the major effectors of nitric oxide to induce acute dilations of murine resistance vessels. 16034136_Type I cGMP-dependent protein kinase (cGKIalpha) down-regulates calcium-ion transients and chemotaxis in murine neutrophils; the regulatory influences of cGKIalpha on secretagogue responses are complex, depending on the granule subtype 16107711_Forkhead box O1a (Foxo1) and cyclic GMP-dependent kinase I (Prkg1) interact to create a novel feedback loop that allows the precise tuning of myoblast fusion. 16154207_These results demonstrate a role for cGKI signaling in cortical development related to neuronal migration/positioning that is important for dendritic orientation and connectivity. 16154279_appears that distinct brain regions express distinct cGMP-dependent protein kinase type I isoforms that signal via distinct pathways 16167338_NO stimulates prolidase activity by increasing serine/threonine phosphorylation through PKG-cGMP pathway 16533816_in Th2 cells, cGMP is produced upon TCR engagement and activates PKG, which controls dihydropyridine-sensitive calcium inflow and Th2 cytokine production 16920709_Protein kinase G is a critical downstream effector of the noncanonical Wnt-Frizzled-2/cGMP/Ca2+ pathway 17105400_The specific, membrane-permeable cGMP-dependent protein kinase G-Ialpha (PKG-Ialpha) inhibitor DT-2 prevents an efficient Shh response, indicating that the effects of cGMP on the Shh response are mediated via PKG.[PKG-Ialpha] 17420342_Regulation of cardiac beta-adrenergic response by cGMP is specifically linked to a nitric oxide-synthesis/PDE-5-hydrolyzed pool signaling via protein kinase G. 17878170_CRP4 mediates cGMP/PKG stimulation of SM-specific gene expression, and PKG plays an important role in regulating the phenotype of VSMCs 17991884_phosphorylation of Smad3 by protein kinase G is a potential molecular mechanism by which activation of ANP/cGMP/protein kinase G signaling disrupts TGF-beta1-induced nuclear translocation of pSmad3 and downstream events 18256252_CRP2 has been confirmed as a target of cyclic GMP/cGMP-dependent protein kinase I signaling in the spinal cord and in dorsal root ganglia. 18420996_The absence of cGKI in vascular smooth muscle did not affect vascular remodeling after carotid ligation or removal of the endothelium. 18443297_Tese results identify a role of cGKI as mediator of erythrocyte survival and extend the emerging concept that cGMP/cGKI signaling has an antiapoptotic/prosurvival function in a number of cell types in vivo. 18535260_posttranslational modification of PKGI critically influences the nuclear translocation of PKGI and activities of cGMP in SMCs. 18685080_Cyclic guanosine monophosphate-dependent protein kinase I promotes adhesion of primary vascular smooth muscle cells 18723505_sildenafil activates a PKG-dependent novel signaling cascade that involves activation of ERK and inhibition of glycogen synthase kinase 3beta leading to cytoprotection 19008225_The commonly used cGMP-dependent protein kinase type I (cGKI) inhibitor Rp-8-Br-PET-cGMPS can activate cGKI in vitro and in intact cells 19156199_PRKG1 might be involved in the stabilization and output strength of the circadian oscillator in mice 19168131_The results suggest that chronic elevation of cGMP, as seen in inflammatory conditions, triggers ubiquitination and degradation of PKG-Ialpha in smooth muscle. 19287493_findings demonstrate that the cGMP-cGKI pathway is critical for postnatal neovascularization and establish a new role for cGKI in vasculogenesis, which is mediated by bone marrow-derived progenitors 19457107_The neuronal NOS/cGMP/PKG pathway stimulates cPLA(2) phosphorylation at Ser505,suggesting that up-regulation of this pathway in Parkinson's disease may mediate dopaminergic neuron degeneration and death through activation of cPLA(2). 19487814_loss of caveolin-1 leads to hyperactive eNOS and subsequent tyrosine nitration-dependent impairment of PKG activity, which results in pulmonary hypertension. 19628652_cGMP/cGKI signaling differentially inhibits contraction: in jejunum, inhibition is performed without changing [Ca(2+)](i) and is dependent on phosphatase activity, whereas in colon, inhibition is mediated by inhibition of [Ca(2+)](i) signals. 19737918_cGK I-mediated Ser188 phosphorylation of RhoA is identified as a converging node for pro- and antifibrotic signals. 19952371_PKGI links NO and cGMP signaling with the RhoA-ROCK and the insulin pathways, thereby controlling induction of adipogenic and thermogenic programs during brown fat cell differentiation 19955367_This study identified p53 as a transcription factor that regulates the expression of cGKI during neuronal maturation and cGMP-dependent inhibition of growth cone collapse. 19961855_PDE5-inhibition blocks TRPC6 channel activation and associated Cn/NFAT activation signaling by PKG-dependent channel phosphorylation 20018259_(D)-Amino acid analogues of DT-2 as highly selective and superior inhibitors of cGMP-dependent protein kinase Ialpha. 20023176_TGF-beta(1) decreased sGC and PKGI expression in pulmonary artery and aortic smooth muscle cells from adult rats and mice. 20060325_Basal/moderately elevated PKG activity protects against high/pathological-level nitric oxide-induced apoptosis and promotes DNA synthesis/proliferation in vascular smooth muscle cells. 20164378_These data establish that glucose-mediated downregulation of PKG levels stimulates TSP1 expression and enhances TGF-beta activity and matrix protein expression, which can contribute to vascular remodeling in diabetes. 20171263_Cytosolic cGKI can support fear memory consolidation and LTP in neurons of the lateral amygdala via activation of CREB and CRE-dependent transcription. 20212138_cardiac myocyte cGKI does not affect the development of heart hypertrophy induced by pressure overload or chronic isoproterenol infusion 20352235_Ang II suppression by ANP was abolished in cardiomyocytes of mice deficient in GC-A, in cyclic GMP-dependent protein kinase I (PKG I) or in the regulator of G protein signalling (RGS) 2, a target of PKG I 20410120_Activation and inhibition of protein kinase G (PKG) respectively upregulates and downregulates recombinant potassium channel subfamily K (TASK1) channels heterologously expressed in PKG-loaded human embryonic kidney cells in vitro. 20453163_Cyclis GMP, through PKGI, attenuated H(2)O(2)-induced cytotoxicity in lung microvascular endothelial cells by increasing catalase and Gpx-1 expression through an unknown posttranscriptional effect. 20456015_evidence for PKGIalpha-dependent phosphorylation and activation of neuronal AAAD in vitro, and introduce AAAD as a putative PKGIalpha substrate. 20930715_There are sex differences in the effects of PKG-I activation on the regulation of adipose tissue function and diet-induced obesity. 20978093_cGKI modulates glucagon release by suppression of calcium in alpha cells in mice. 21328456_protein kinase G type-I is essential for promoting proliferation and cell survival of mouse bone marrow stromal cells 21464444_Genetic deletion of cGKI in non-neuronal cells results in a complex metabolic phenotype, including liver inflammation and fasting hyperglycemia. 21914444_different signaling pathways exist for each cGKI isoform in vivo in fundus muscles 22203739_Sodium depletion enhances renal expression of (pro)renin receptor via cyclic GMP-protein kinase G signaling pathway. 22253479_Data show that protection of the duodenum from acid injury requires neuronal cGMP kinase I. 22270721_role in noise-induced hearing loss and endogenous signaling pathway and activation of poly (ADP-ribose) polymerase 22427743_PKG-I-mediated presynaptic facilitation and synaptic long-term potentiation in spinal projection neurons is functionally involved in activity-dependent centrally mediated nociceptive hypersensitivity. 22503686_these findings suggest that cGK-Ialpha interacts with and phosphorylates rhotekin, thereby contributing to neurite outgrowth regulation. 22632818_PKG-I deficiency induces pulmonary hypertension through Rho A/Rho kinase activation-mediated vasoconstriction and pulmonary vascular remodeling. 23006734_Data indicate that the 2 mechanisms (cGMP binding and the disulfide homodimer) of activating PKG1alpha are intricately linked with the binding of cGMP preventing oxidation to the disulfide state. 23066013_RhoA is a PKGIalpha target and direct binding of activated PKGIalpha to RhoA is central to cGMP-mediated inhibition of the VSMC Rho kinase contractile pathway 23407708_our study demonstrates a direct connection between stimulus-induced, cGKIalpha-mediated phosphorylation of Ser residues identified in the BKCa channel and increases in channel activity in a VSM cell system 23657971_mutation of the PKGIalpha LZ domain produces a clinically relevant model for hypertensive heart disease of aging. 23686857_overexpression of PCs, furin and PC5, but not PC7, which are all expressed in SMC, increase PKGI cleavage in a dose-dependent manner 23716652_Data indicate that oxidative activation of PKG Ialpha is a key mediator of hypotension and consequential organ injury during sepsis. 23755100_These findings characterize PKG as a novel regulator of AR-mediated transcription by enhancing AR cofactor p44/WDR77's function. 23760283_Results indicate that cGMP, acting primarily through cGKIalpha, is an important suppressor of kidney fibrosis. 23770744_PKG positively regulates proteasome activities and proteasome-mediated degradation of misfolded proteins, likely through posttranslational modifications to proteasome subunits. 23788765_Abnormal cGMP/cGKI signaling in nonhematopoietic cells affects thrombopoiesis via elevated interleukin-6 production and results in thrombocytosis in vivo. 23814119_ANP attenuates the inflammatory actions of histamine via endothelial GC-A/cGMP/cGKI signaling and inhibitory phosphorylation of TRPC6 channels. 23832809_cGKI and Trpc1,3,6 channels are not functionally coupled in vascular smooth cell. 24375799_PKG-1alpha dimerization is a major contributing factor to the vasodilator actions of DHEA. 24401847_cGMP, through activation of PKGI, inhibits c-Abl, leading to increased key antioxidant enzymes and resistance to lung endothelial oxidant injury. 24449818_In dystrophic hearts, excess contractility and arrhythmia are coupled to TRPC6 and are ameliorated by its targeted suppression or PKG activation. 24573388_Increasing PKG activity ameliorates renal fibrosis in part through regulation of macrophage and tubular cell function, leading to reduced TGF-beta-induced fibrosis. 24872318_the key role of PKG-I in transforming growth factor beta1 induction by like weak inducer of apoptosis in kidney cells 25128522_A functional leucine zipper domain in PKG-1alpha is essential for maintenance of a low pulmonary vascular tone in normoxia. 25139994_For the initial phase of angiotenin II-induced cardiac hypertrophy, lack of cardiomyocyte cGKI activity does not worsen hypertrophic growth. 25447536_miR-20a expression is up-regulated in response to hypoxia in both mouse and human pulmonary arterial smooth muscle cells 25586176_Data show that cGMP-dependent kinase (cGK-Ialpha) knockdown partially rescued TNF-alpha-induced decrease in 42-kDa cGMP-dependent protein kinase-anchoring protein (GKAP42) and impairment of insulin signals. 25900831_Protein kinase G has a role in regulating production of H2S, which governs oxygen sensing 25938783_these results demonstrate that myocardial PKG1alpha oxidation prevents a beneficial response to pathological stress, may explain variable responses to PKG1alpha pathway stimulation in heart disease 26124274_cGMP/protein kinase G signaling suppresses Itpr1 phosphorylation and promotes endoplasmic reticulum stress in photoreceptors of Cnga3-deficient mice. 26830212_Cyclic GMP kinase deletion causes iron deficiency anemia, duodenal ulcers, and bleeding in the knockout mice. 27342776_PKG Ialpha disulfide formation triggers cardiac injury, and this initiation of maladaptive signaling can be blocked by pharmacological therapies that elevate cGMP, which binds kinase to limit its oxidation. 28062507_Brown adipose tissue, via Nox4-derived hydrogen peroxide, induces cyclic GMP-dependent protein kinase G type-1alpha activation, resulting in reduced vascular contractility. 28280239_analysis of the role of binding interface with protein kinase G-Ialpha leucine zipper in vascular function 28326941_that PKG-I expressed in nociceptors is not only a key determinant of dorsal root ganglion hyperexcitability and spinal synaptic plasticity but also an important modulator of cortical neuronal activity in pathological pain states 28360102_disulfide formation at Cys(43) does not directly activate PKGIalpha, and the C43S-mutant PKGIalpha has a higher Ka for cGMP. Results highlight that mutant enzymes should be carefully biochemically characterized before making in vivo inferences. 28690194_Despite robust evidence for PKG Ialpha oxidation during NOS uncoupling in cell models, it is unlikely that PKG Ialpha oxidation occurs to a significant extent in vivo during diet-induced obesity and so is unlikely to mediate the associated cardiovascular dysfunction. 29054855_This study demonstrates that H- ras deletion protects against AngII-induced cardiac remodeling, possibly via a mechanism in which PKG-Ibeta overexpression could play a partial role, and points to H-Ras and/or downstream proteins as potential therapeutic targets in cardiovascular disease. 30104414_the NF-kappaB/miR-155-5p/PKG1 axis may be crucial in the pathogenesis of inflammatory vascular diseases, such as atherosclerotic intimal hyperplasia and preeclamptic hypertension. 30152261_These data thus demonstrate the existence of two differently distributed cGKI isoforms in the dorsal root ganglion, and may provide insight into the cellular and molecular mechanisms of pain. 30327468_Prkg1 cascade role in platelet activation and thrombosis. 31186362_Increased oxidation of PKGIalpha to its disulfide-activated state found in pulmonary arteries from mice with hypoxic pulmonary arterial hypertension (PH) or lungs from patients. It caused by chronic hypoxia, and it limits progression of PH by pulmonary vasodilation that counters and offsets maladaptive vasoconstriction, and also disulfide PKGIalpha is protective by preventing maladaptive growth and fibrosis signaling. 31387997_Study shows that mice carrying gain-of-function mutation in PRKG1 recapitulate the human early-onset thoracic aortic disease, and that pathological changes in the aorta are, at least in part, caused by oxidative stress, revealing that increased basal PKG activity induces oxidative stress in the aorta, raising concern about the widespread clinical use of PKG-activating drugs. 31423931_These results suggest that cGKI in vascular smooth muscle cells should be targeted by therapies designed to protect brain tissue against ischemic stroke. 32315291_Protein kinase G1 regulates bone regeneration and rescues diabetic fracture healing. 33291075_The endothelial nitric oxide synthase/cyclic guanosine monophosphate/protein kinase G pathway activates primordial follicles. 34863181_Ferulic acid ameliorates lipopolysaccharide-induced tracheal injury via cGMP/PKGII signaling pathway. |
ENSG00000185532 |
PRKG1 |
33.435381 |
0.1198982278 |
-3.060118 |
0.51200500 |
36.293642 |
0.00000000169716627124848476361591030940004914961960480468405876308679580688476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000231641905409112158974438536217579920339915133808972314000129699707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.78600236294863 |
2.4591911756786 |
56.5980205692745 |
12.9237576186793 |
| ENSMUSG00000053007 |
231991 |
Creb5 |
protein_coding |
A0A0N4SUK5 |
FUNCTION: Binds to the cAMP response element and activates transcription. {ECO:0000256|PIRNR:PIRNR003153}. |
Activator;Coiled coil;DNA-binding;Nucleus;Proteomics identification;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:231991; |
nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity [GO:0003700] |
36542062_TGF-beta signaling and Creb5 cooperatively regulate Fgf18 to control pharyngeal muscle development. |
ENSG00000146592 |
CREB5 |
43.231332 |
2.1556744496 |
1.108139 |
0.39582389 |
7.846593 |
0.00509165629148387258867680671414746029768139123916625976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0188752914298025535611014191772483172826468944549560546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
56.5968569304621 |
15.7237021691789 |
26.2548256960033 |
5.67381096209397 |
| ENSMUSG00000053062 |
67374 |
Jam2 |
protein_coding |
Q9JI59 |
FUNCTION: Junctional adhesion protein that mediates heterotypic cell-cell interactions with its cognate receptor JAM3 to regulate different cellular processes (PubMed:16093349, PubMed:21868569, PubMed:24357068). Plays a role in homing and mobilization of hematopoietic stem and progenitor cells within the bone marrow (PubMed:21868569, PubMed:24357068). At the surface of bone marrow stromal cells, it contributes to the retention of the hematopoietic stem and progenitor cells expressing JAM3 (PubMed:21868569, PubMed:24357068). Plays a central role in leukocytes extravasation by facilitating not only transmigration but also tethering and rolling of leukocytes along the endothelium (PubMed:16297198, PubMed:19740376). Tethering and rolling of leukocytes are dependent on the binding by JAM2 of the integrin alpha-4/beta-1 (PubMed:19740376). Plays a role in spermatogenesis where JAM2 and JAM3, which are respectively expressed by Sertoli and germ cells, mediate an interaction between both cell types and play an essential role in the anchorage of germ cells onto Sertoli cells and the assembly of cell polarity complexes during spermatid differentiation (Probable). Also functions as an inhibitory somatodendritic cue that prevents the myelination of non-axonal parts of neurons (PubMed:27499083). During myogenesis, it is involved in myocyte fusion (By similarity). May also play a role in angiogenesis (PubMed:25911611). {ECO:0000250|UniProtKB:A0A0R4IGV4, ECO:0000269|PubMed:16093349, ECO:0000269|PubMed:16297198, ECO:0000269|PubMed:19740376, ECO:0000269|PubMed:21868569, ECO:0000269|PubMed:24357068, ECO:0000269|PubMed:25911611, ECO:0000269|PubMed:27499083, ECO:0000305|PubMed:15372036, ECO:0000305|PubMed:25817991}. |
3D-structure;Cell junction;Cell membrane;Direct protein sequencing;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Reference proteome;Signal;Tight junction;Transmembrane;Transmembrane helix |
|
|
mmu:67374; |
bicellular tight junction [GO:0005923]; cell surface [GO:0009986]; cell-cell contact zone [GO:0044291]; plasma membrane [GO:0005886]; protein complex involved in cell adhesion [GO:0098636]; somatodendritic compartment [GO:0036477]; tight junction [GO:0070160]; integrin binding [GO:0005178]; cell-cell adhesion [GO:0098609]; cellular extravasation [GO:0045123]; hematopoietic stem cell migration to bone marrow [GO:0097241]; leukocyte cell-cell adhesion [GO:0007159]; leukocyte tethering or rolling [GO:0050901]; lymphocyte aggregation [GO:0071593]; myoblast fusion [GO:0007520]; negative regulation of cell adhesion [GO:0007162]; negative regulation of myelination [GO:0031642]; positive regulation of lymphocyte migration [GO:2000403]; spermatid development [GO:0007286] |
12239159_promotes lymphocyte transendothelial migration 12476045_Results suggest a role for junctional adhesion molecule-2 (JAM-2) in facilitating transmigration in endothelial cells. 16297198_Interactions with JAM-B and -C are essential for development of cutaneous inflammation. 16914739_These results demonstrate that Jam-B is dispensable for normal mouse development and stem cell identity in embryonic, neural, and hematopoietic stem cells. 18368118_JAM-B identifies a unique population of RGCs in which structure corresponds remarkably to function 19164472_IL-1alpha and TGF-beta2 regulate JAM-B expression in an opposite manner in sertoli cells. 21868569_JAM-B is an active player in the maintenance of the bone marrow stromal microenvironment. 22511936_Jam2 has a role in the interaction between hatched blastocyst and receptive uterus. 23068611_Data show that junctional adhesion molecule-B (JAM-B) expressed by endothelial cells contributes to murine B16 melanoma cells metastasis through its interaction with junctional adhesion molecule-C (JAM-C) on tumor cells. 24357068_Function of Jam-B/Jam-C interaction in homing and mobilization of human and mouse hematopoietic stem and progenitor cells. 25817991_TGF-beta3 significantly downregulates JAM-B expression via post-transcriptional and post-translational modulation and results in the disruption of BTB and apical ES. 25911611_results provided evidence for JAM-B as a novel antiangiogenic factor through regulation of VEGF signaling in vitro, but this effect did not extend to in vivo studies. 26914234_JAM-B is an as yet underappreciated trophoblast lineage-specific protein, which is modulated via the progesterone receptor and shows unique strain-specific kinetics. 27111582_JAM-B/JAM-C mediated interaction between endothelial cells and stellate cells stabilizes vessel walls and may control the sinusoidal diameter. 27499083_identify neuronal junction adhesion molecule 2 (JAM2) as an inhibitory myelin-guidance molecule. 29753567_this study shows that JAM-B promotes autoimmune-mediated liver fibrosis in mice 31167130_Frequency of Jam2(high) hematopoietic stem cell (HCSs) changes upon T cell depletion in vivo, potentially suggesting that Jam2 expression may reflect scarcity of T cells and requirement of T cell replenishment. These findings highlight Jam2 as a potential marker for a subfraction of HSCs with an extensive lymphopoietic capacity, mainly in T lymphopoiesis. |
ENSG00000154721 |
JAM2 |
251.406612 |
0.1036450555 |
-3.270277 |
0.20271452 |
268.248903 |
0.00000000000000000000000000000000000000000000000000000000000273248453833124172506721851058268493796344099533485594876448960227678009230134044804167796658689900902594248172518275813084812667146773261086138771380066314620194134477060288190841674804687500000000000000 |
0.0000000000000000000000000000000000000000000000000000000006306243104221435426474073749473950944352900654669512005820629644084224265712978795352822830914915270860299537083032343618344212340315214356889841895714710062748054042458534240722656250000000000000000000 |
Yes |
No |
47.1740559568111 |
6.65030650114575 |
455.150086318607 |
39.3192649004099 |
| ENSMUSG00000053101 |
353346 |
Gpr141 |
protein_coding |
Q7TQP0 |
FUNCTION: Orphan receptor. |
Cell membrane;G-protein coupled receptor;Glycoprotein;Membrane;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:353346; |
plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930] |
Human_homologues 25672891_GPR141-NME8 locus had strong genetic effect on susceptibility to generalized periodontitis in Japanese individuals with history of smoking. identified suggestive loci for periodontitis in Japanese population. |
ENSG00000187037 |
GPR141 |
181.441775 |
2.1272027700 |
1.088958 |
0.27605046 |
15.366706 |
0.00008853464434096823879299237303186487224593292921781539916992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005332576296697480715963690656167273118626326322555541992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
239.691022437111 |
46.1437161600153 |
112.678972509296 |
16.5271381831382 |
| ENSMUSG00000053117 |
107376 |
E330013P04Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
53.274106 |
2.2326369386 |
1.158749 |
0.38486835 |
9.063382 |
0.00260779121367284974150613230392536934232339262962341308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0106807947745804904249311562125512864440679550170898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
78.9716266021394 |
21.3180645633614 |
35.3714592979063 |
7.48244874418974 |
| ENSMUSG00000053166 |
104010 |
Cdh22 |
protein_coding |
Q9WTP5 |
FUNCTION: Cadherins are calcium-dependent cell adhesion proteins. They preferentially interact with themselves in a homophilic manner in connecting cells; cadherins may thus contribute to the sorting of heterogeneous cell types. PB-cadherins may have a role in the morphological organization of pituitary gland and brain tissues. |
3D-structure;Calcium;Cell adhesion;Cell membrane;Glycoprotein;Membrane;Metal-binding;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:104010; |
adherens junction [GO:0005912]; catenin complex [GO:0016342]; cadherin binding [GO:0045296]; calcium ion binding [GO:0005509]; adherens junction organization [GO:0034332]; brain development [GO:0007420]; calcium-dependent cell-cell adhesion via plasma membrane cell adhesion molecules [GO:0016339]; cell morphogenesis [GO:0000902]; cell-cell adhesion via plasma-membrane adhesion molecules [GO:0098742]; cell-cell junction assembly [GO:0007043]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
20723620_spatio-temporal mRNA distribution pattern of Cdh12 and Cdh22 during CNS development; demonstrate presence of Cdh12 and Cdh22 mRNA in both excitatory and inhibitory neurons in adult CNS including cerebellum, neocortex, hippocampus and subcortical nuclei 21761482_Results describe the expression of Cdh22 in developing mouse brain and compare it to expression of related family members. 29063123_Janus kinase 2 protein (JAK2) and beta-catenin were found to interact with cadherin-22 (Cdh22) and involved in CDH22 signaling in female germ line stem cells (FGSCs). 31041846_glial cell line-derived neurotrophic factor (GDNF) was identified as an essential factor for FGSCs self-renewal with a more complicated mechanism: GDNF-GFRA1 activates AKT3 via PI3K or Src family kinase (SFK), and SFK upregulates its target genes, Bcl6b, Etv5, and Lhx1, to promote self-renewal of Female germline stem cells. |
ENSG00000149654 |
CDH22 |
94.194151 |
24.9924884554 |
4.643423 |
1.47039171 |
6.968669 |
0.00829491788863929296882204766916402149945497512817382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0285532631140686353476976222509620129130780696868896484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
184.915620159539 |
222.831355982978 |
7.39884787741693 |
6.2533881724713 |
| ENSMUSG00000053219 |
379043 |
Raet1e |
protein_coding |
Q9CZQ6 |
FUNCTION: Acts as a ligand for KLRK1. {ECO:0000269|PubMed:11567106, ECO:0000269|PubMed:16086018}. |
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:379043; |
external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; intracellular membrane-bounded organelle [GO:0043231]; plasma membrane [GO:0005886]; natural killer cell lectin-like receptor binding [GO:0046703]; positive regulation of natural killer cell mediated cytotoxicity [GO:0045954]; T cell mediated cytotoxicity [GO:0001913] |
22244846_RAE1 expression was sufficient to induce the recruitment of adoptively transferred CTLs to islets. 23166357_Raet1e genes are directly transcriptionally activated by E2F family transcription factors, which play a central role in regulating cell cycle entry. 23948654_Reveal Raet1e, a major histocompatibility complex class 1-like molecule expressed in lesional aortic endothelial cells and macrophage-rich regions, as a novel atherosclerosis gene. 24708417_CD4(+) NKG2D(+) T cells induce NKG2D down-regulation in natural killer cells in CD86-RAE-1epsilon transgenic mice. 27444966_Study reports that retinoic acid early induced transcript-1-d and retinoic acid early induced transcript-1-e (Raet1) genes are induced early upon experimental autoimmune encephalomyelitis onset and reach a maximal expression at the peak of the pathology. Study also shows that macrophages as well as microglia, are cellular sources of Raet1 transcripts. |
|
|
358.648549 |
2.1019926381 |
1.071758 |
0.19205284 |
30.857637 |
0.00000002776673291724320089385732665283185172100388626859057694673538208007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003142220473963212943294034517477397727702737029176205396652221679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
488.467140500136 |
65.217401077025 |
232.382897850671 |
23.5832759697633 |
| ENSMUSG00000053522 |
16858 |
Lgals7 |
protein_coding |
Q9CRB1 |
Human_homologues FUNCTION: Could be involved in cell-cell and/or cell-matrix interactions necessary for normal growth control. Pro-apoptotic protein that functions intracellularly upstream of JNK activation and cytochrome c release. {ECO:0000269|PubMed:11706006}.; + ;FUNCTION: Could be involved in cell-cell and/or cell-matrix interactions necessary for normal growth control. Pro-apoptotic protein that functions intracellularly upstream of JNK activation and cytochrome c release. {ECO:0000269|PubMed:11706006}. |
Lectin;Proteomics identification;Reference proteome |
Human_homologues NA; + ;NA |
|
Human_homologues hsa:3963;hsa:653499;;hsa:3963;hsa:653499; |
carbohydrate binding [GO:0030246] |
12194966_Galectin-3 and galectin-7 play a role in corneal epithelial wound healing. 12682633_Upregulation of galectin-7 in murine lymphoma cells is associated with progression toward an aggressive phenotype. 15958565_Galectin-7 promotes maliganacy by upregulating MMP-9 gene expression. 17706016_A comparative profiling has been started in the mouse as a model organism, combining sequence analysis, expression patterns and structural features in the cases of the homodimeric galectins-1, -2 and -7. 18395096_Hepatocyte growth factor suppresses profibrogenic signal transduction via nuclear export of Smad3 with galectin-7. 18597104_demonstrates cell-type specificity and cycle-associated regulation for galectin-3 with increased presence in atretic preantral follicles and in late stages of luteolysis 18829868_These studies provide the first genetic evidence showing that galectin-7 can modulate keratinocyte apoptosis, proliferation, and migration during skin repair. 20382700_Data show that high levels of galectin-7 expression in breast cancer cells drastically increased their ability to metastasize to lungs and bones, and were restricted to high-grade breast carcinomas. 21677144_Galectin-7 modulates the length of the primary cilia and wound repair in polarized kidney epithelial cells. 22251572_Extracellular superoxide dismutase plays a role not only as a reactive oxygen species scavenger, but also as a pro-apoptotic factor via COX-2/galectin-7 pathways in the epidermis. 23498801_In cardiac transplantation galectin-7 relates to acute allograft rejection and T-cell responses possibly as an accelerant. 23658821_Overexpression of galectin-7 increased the resistance of melanoma cells to apoptosis while inducing de novo egr-1 expression. 25741714_results indicate that an excess of galectin-7 leads to a destabilisation of adherens junctions associated with defects in epidermal repair 27902734_The data suggest that gal-7 could potentiate the phenotype of HER-2 positive primary breast cancer. 28526214_Diabetic mice model shows that increased O-linked N-Acetyl-glucosamine (O-GlcNAc) is responsible for reduced Gal-7 expression in keratinocytes exposed to high glucose environment. 29693486_Serum galectin-7 was not useful for predicting prognosis in threatened abortion 32627954_Galectin-7 overexpression destroys airway epithelial barrier in transgenic mice. 33055419_Galectin-7 downregulation in lesional keratinocytes contributes to enhanced IL-17A signaling and skin pathology in psoriasis. 34318956_Enhanced galectin-7 expression favors wound healing. |
ENSG00000178934+ENSG00000205076 |
LGALS7B+LGALS7 |
58.336550 |
9.7878603801 |
3.290994 |
0.40860495 |
66.933053 |
0.00000000000000028088563396490120374744314285821803929520926092905375170971637999173253774642944335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000080120786077778562125891162827745410812067382994028363896177324932068586349487304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
108.095590764811 |
31.0261443849952 |
11.0438427364819 |
2.69442581535001 |
| ENSMUSG00000053552 |
228598 |
Ebf4 |
protein_coding |
Q8K4J2 |
FUNCTION: Transcription factor (PubMed:12139918). Positively modulates transcription, perhaps less strongly than other early B cell factor/EBF family proteins (PubMed:12139918). Binds an EBF1/Olf-1 consensus site in vitro (PubMed:12139918). {ECO:0000269|PubMed:12139918}. |
Activator;Alternative splicing;Developmental protein;DNA-binding;Metal-binding;Nucleus;Reference proteome;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:228598; |
chromatin [GO:0000785]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription factor binding [GO:0140297]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of transcription by RNA polymerase II [GO:0006357]; T cell apoptotic process [GO:0070231] |
12139918_The fourth member of the Olf-1/Ebf-like gene family, O/E-4, is expressed in the neuronal and basal cell layers of olfactory epithelium and may interact with other O/E family members to regulate gene expression in the olfactory sensory neurons. |
ENSG00000088881 |
EBF4 |
29.501652 |
0.2330731024 |
-2.101146 |
0.52779268 |
15.693000 |
0.00007449936289487677962793676922359509262605570256710052490234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004555496971556656331248102986819503712467849254608154296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.1059109450209 |
4.15420222045313 |
47.649903953368 |
12.0962537904183 |
| ENSMUSG00000053559 |
207818 |
Smagp |
protein_coding |
Q99KC7 |
FUNCTION: May play a role in epithelial cell-cell contacts. May play a role in tumor invasiveness and metastasis formation (By similarity). {ECO:0000250}. |
Cell membrane;Cytoplasmic vesicle;Glycoprotein;Membrane;Reference proteome;Sialic acid;Transmembrane;Transmembrane helix |
|
|
mmu:207818; |
cell junction [GO:0030054]; cytoplasmic vesicle membrane [GO:0030659]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886] |
Human_homologues 33049294_SMAGP knockdown inhibits the malignant phenotypes of glioblastoma cells by inactivating the PI3K/Akt pathway. 36436062_SMAGP regulates doxorubicin sensitivity in triple-negative breast cancer cells via modulating mitochondrial respiration. |
ENSG00000170545 |
SMAGP |
51.554139 |
0.4110382302 |
-1.282656 |
0.35100198 |
13.255084 |
0.00027184158509829114839032038197785823285812512040138244628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0014593022362451031192359662824742372322361916303634643554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
29.5111543154474 |
7.2509905119723 |
71.7966168334872 |
12.504529818715 |
| ENSMUSG00000053687 |
319446 |
Dpep2 |
protein_coding |
Q8C255 |
FUNCTION: Dipeptidase that hydrolyzes leukotriene D4 (LTD4) into leukotriene E4 (LTE4) (PubMed:12738806). Hydrolyzes cystinyl-bis-glycine (By similarity). {ECO:0000250|UniProtKB:Q9H4A9, ECO:0000269|PubMed:12738806}.; FUNCTION: Independently of its dipeptidase activity can also modulate macrophage inflammatory response by acting as a regulator of NF-kappa-B inflammatory signaling pathway. {ECO:0000269|PubMed:30899700}. |
Alternative splicing;Dipeptidase;Disulfide bond;Glycoprotein;GPI-anchor;Hydrolase;Lipid metabolism;Lipoprotein;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Signal;Zinc |
|
|
mmu:319446; |
membrane [GO:0016020]; dipeptidase activity [GO:0016805]; exopeptidase activity [GO:0008238]; metal ion binding [GO:0046872]; metallodipeptidase activity [GO:0070573]; leukotriene D4 catabolic process [GO:1901749]; leukotriene metabolic process [GO:0006691]; proteolysis [GO:0006508] |
12738806_cloning and characterization of membrane-bound dipeptidase-2 and membrane-bound dipeptidase-3 30899700_Taken together, this study revealed that Dpep2 could protect against Coxsackievirus B3-induced viral myocarditis by acting as a suppressor of macrophage inflammation. |
ENSG00000167261 |
DPEP2 |
108.885113 |
0.3309734038 |
-1.595213 |
0.40787793 |
14.654743 |
0.00012910933091197097487973222129653549927752465009689331054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007531954532558950707668565804908666905248537659645080566406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
53.0394712522835 |
15.1958522735043 |
160.252970918231 |
33.6904060904882 |
| ENSMUSG00000053863 |
94111 |
Mepe |
protein_coding |
Q8K4L6 |
FUNCTION: Regulates renal phosphate and uric acid excretion (PubMed:26051469). Regulates bone mineralization by osteoblasts and cartilage mineralization by chondrocytes (PubMed:11414762, PubMed:12421822, PubMed:15843468, PubMed:22766095). Regulates the mineralization of the extracellular matrix of the craniofacial complex, such as teeth, bone and cartilage (PubMed:26927967). Increases dental pulp stem cell proliferation (By similarity). {ECO:0000250|UniProtKB:Q9NQ76, ECO:0000269|PubMed:11414762, ECO:0000269|PubMed:12421822, ECO:0000269|PubMed:15843468, ECO:0000269|PubMed:22766095, ECO:0000269|PubMed:26051469, ECO:0000269|PubMed:26927967}. |
Biomineralization;Extracellular matrix;Glycoprotein;Phosphoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:94111; |
extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular matrix protein binding [GO:1990430]; biomineral tissue development [GO:0031214]; bone mineralization [GO:0030282]; negative regulation of bone mineralization [GO:0030502]; skeletal system development [GO:0001501] |
12220505_MEPE may be involved in the pathogenesis defective mineralization due to Phex deficiency in X-linked hypophosphatemia (XLH) and the Hyp-mouse. 15221418_Mepe appears to play a role in both long bone regeneration and the latter stages of skeletogenesis. 18162525_degradation of MEPE and DMP-1 and release of ASARM peptides are chiefly responsible for the HYP mineralization defect and changes in osteoblast-osteoclast differentiation. 18597632_results, showing that pASARM inhibits mineralization provide a mechanism explaining how loss of PHEX activity can lead to extracellular matrix accumulation of ASARM resulting in the osteomalacia of X-linked hypophosphatemia 19520780_MEPE and MEPE-phosphate-regulating gene with homologies to endopeptidases on the X chromosome (MEPE-PHEX) interactions are components to an age-diet-dependent pathway that regulates bone turnover and mineralization and suppresses renal calcification. 19617624_MEPE expression is bone cell-specific and induced by the BMP-2 signaling pathway 20599717_the C-terminal fragment of MEPE containing an RGD sequence, cleaved in odontoblasts, appeared to be the active form of MEPE, which may play important roles in dentinogenesis and pulpal homeostasis by keeping the odontoblasts in immature condition. 20725825_MEPE might be an important signaling molecule involved in the regulation of osteoblast and osteoclast activity during bone remodeling. 22042093_The temporal-spatial specific pattern and unique co-localization of Dspp, Mepe, Mimecan and Versican suggest they play complementary roles during odontogenesis. 22213482_Wnt3a stimulates Mepe transcription directly by a canonical Wnt signaling pathway through beta-catenin and Lef-1 and indirectly through the activation of a Bmp-2 autocrine loop. 22766095_MEPE is an important regulator of growth plate chondrocyte matrix mineralization through its cleavage to an ASARM peptide. 26051469_The C-terminal ASARM-motif plays a major role in regulating bone-mass and cancellous structure as mice age 26428891_An in situ hybridization study of perlecan, DMP1, and MEPE in developing condylar cartilage of the fetal mouse mandible and limb bud cartilage. 26927967_Mepe(-/-) molars exhibited increased thickness of predentin, dentin, and enamel over controls and decreased gene expression of Enam, Bsp, Dmp1, Dspp, and Opn by RT-PCR. |
ENSG00000152595 |
MEPE |
23.386165 |
0.0575451856 |
-4.119161 |
0.74394163 |
33.362034 |
0.00000000765029622264574758529288097015641501030813742545433342456817626953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000942793786920226635367368850891567344518762183724902570247650146484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.74627145826111 |
1.47974525826655 |
47.7237397101618 |
14.0124138180421 |
| ENSMUSG00000053963 |
381310 |
Stum |
protein_coding |
Q0VBF8 |
|
Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:381310; |
membrane [GO:0016020] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000203685 |
STUM |
15.490559 |
0.0215287004 |
-5.537595 |
1.09959405 |
31.157194 |
0.00000002379558013933732870114541341644703864588450414885301142930984497070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000002723172627215523879511149298038308330660584033466875553131103515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.696036155670065 |
0.583381337313672 |
32.3306164720709 |
12.9904986849136 |
| ENSMUSG00000054256 |
17690 |
Msi1 |
protein_coding |
Q61474 |
FUNCTION: RNA binding protein that regulates the expression of target mRNAs at the translation level. Regulates expression of the NOTCH1 antagonist NUMB. Binds RNA containing the sequence 5'-GUUAGUUAGUUAGUU-3' and other sequences containing the pattern 5'-[GA]U(1-3)AGU-3'. May play a role in the proliferation and maintenance of stem cells in the central nervous system. {ECO:0000269|PubMed:11359897, ECO:0000269|PubMed:12407178}. |
3D-structure;Acetylation;Alternative splicing;Cytoplasm;Nucleus;Phosphoprotein;Reference proteome;Repeat;RNA-binding |
|
|
mmu:17690; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleus [GO:0005634]; polysome [GO:0005844]; identical protein binding [GO:0042802]; mRNA binding [GO:0003729]; poly(U) RNA binding [GO:0008266]; single-stranded RNA binding [GO:0003727]; central nervous system development [GO:0007417]; epithelial cell differentiation [GO:0030855]; regulation of translation [GO:0006417]; response to hormone [GO:0009725] |
12560091_Musashi-1 is present in a few epithelial cells just above the Paneth cells in the small intestinal crypt; Musashi-1 and Hes1 were coexpressed in the crypt base columnar cells located between the Paneth cells 12924647_Distribution of Musashi-1-positive cells corresponded with that of stem cells, as outlined in previous reports, implying that Musashi-1 is a key control element of asymmetrical division within the colon crypt. 14700731_Msi1 immunostaining was found in a variety of supporting cells but not in outer hair cells in the organ of Corti. Although an immunoreactive ring was found around the inner hair cells, it also seemed to originate from the supporting cells. 15076722_By immunohistochemistry and RT-PCR, researchers demonstrate for the first time the expression of Musashi1 during development of the inner ear. 16287482_Important for digestive organ development, particularly for epithelial differentiation. 16554442_It is proposed that ELAV RNA-binding proteins exert an important post-transcriptional control on Msi1 expression in the transition from proliferation to neural differentiation of stem/progenitor cells. 16717192_the complementary expression patterns of the murine Msi paralogues Msi1 and Msi2 during spermatogenesis support the idea of distinct, evolutionarily conserved roles of Msi 17768378_The RNA-binding protein, Msi1, is produced in mouse eyes from embryonic stages until adulthood. 18098125_Msi1 regulates oligodendrocyte lineage cell differentiation and survival. 18362162_These results identify Msi1 as a key determinant of the mammary lineage through its ability to coordinate cell cycle entry and activate the Notch and Wnt pathways by a novel autocrine process involving PLF1 and DKK3. 18471519_Msi-1 involved in cancer cell proliferation, inhibition of apoptosis, and mitotic catastrophe, suggesting an important potential mechanism for its role in tumorigenesis. 18490513_Poly(A) binding protein (PABP) is identified as an Msi1-binding protein, and Msi1 competes with eIF4G for PABP binding. 19573529_Data show that doublecortin (dcx) mRNA is a specific binding target of Musashi1 in vitro. 19645571_Positional specification in a neural stem cell line gene expression is modulated by the RNA-binding protein Musashi1 20036290_Msi1-expression is induced after brain ischemia and may be involved in the reactivation of astrocytes, including their proliferation. 20110912_Expression of the Msi1 and Hes1 small intestinal stem cell markers in the small intestine was not homogenous and the markers were more highly expressed in jejunal tissues. 20477901_data show that ectopic MSI1 expression may contribute to tumorigenesis in selected bladder cancers through multiple mechanisms and reveal a previously unrecognized function of Musashi1 in the regulation of SG formation 20670215_The Msi1-positive cells can differentiate into neural and intestinal epithelial-like cells in vivo. 20696379_Msi1-mediated posttranscriptional regulation of Robo3 controls midline crossing of precerebellar neurons. 20826465_overexpression induces tumorigenesis through Wnt and Notch activation 21221806_Musash1 and Hes1 high expression cells locate predominantly in impaired small-intestinal epithelium after transplantation and contribute to epithelial regeneration. 21320487_These data provide evidence that nuclear, and cytoplasmic sequestering of Musashi-1 in retinal cells is context-specific, and may contribute to downstream functioning of Musashi-1. 21378162_Musashi1 cooperates in abnormal cell lineage protein 28 (Lin28)-mediated let-7 family microRNA biogenesis in early neural differentiation. 21486496_A regulatory element for Msi1 transcription in neural stem/progenitor cells is located in the sixth intron of the Msi1 gene. 22428049_RNA-binding protein Musashi1 modulates glioma cell growth through the post-transcriptional regulation of Notch and PI3 kinase/Akt signaling pathways. 23308249_Msi1-dependent post-transcriptional enhancement of m-Numb is crucial in gastric epithelial regeneration. 24586322_Musashi1 binds to the Musashi binding element and that this element is required for polyadenylation in oocytes. 24935936_MSI1 is specifically inhibited by 18-22 carbon omega-9 monounsaturated fatty acids; MSI1 regulates stearoyl-CoA desaturase 25058468_This study suggests that ciliogenic Rfx transcription factors regulate Msi1 expression in neural stem/progenitor cells. 25380226_Msi proteins contribute to an epithelial gene expression program in neural and mammary cell types. 25782991_Through expression studies and utilizing a transgenic Msi1 testis-specific overexpression model, we have identified 2 unique RNA-binding targets of MSI1 in spermatogonia, Msi2 and Erh, and have demonstrated a role for MSI1 in translational regulation. 26064941_Osteoarticular expression of Msi1 protein is increased in joints with CIA-induced lesion and absent in nonlesioned joints, suggesting that this protein is expressed when the lesion is produced in order to favor tissue repair. 26131972_MSI1 and MSI2 display distinct expression profiles during mammalian folliculogenesis and that MSI2 is required for pre-antral follicle development 26303183_A Mouse Model of Targeted Musashi1 Expression in Whole Intestinal Epithelium Suggests Regulatory Roles in Cell Cycle and Stemness. 26373800_the RNA-binding protein Musashi 1 competes with miR130a and -206 for interaction with tachykinin mRNA 27129280_Given that deregulated fatty acid metabolism plays a key role in kidney fibrosis, these results demonstrate a novel connection between fatty acid and Msi1, an RNA-binding protein, in kidney fibrosis 27281208_studies highlight Msi reporters as a unique tool to identify therapy resistance, and define Msi signalling as a central regulator of pancreatic cancer 27541351_photoreceptors lack prototypical neuronal splicing factors and their splicing profile is driven to a significant degree by the Musashi 1 (MSI1) protein. A striking feature of the photoreceptor splicing program are exons that display a 'switch-like' pattern of high inclusion levels in photoreceptors and near complete exclusion outside of the retina. 27799368_Msi proteins are dispensable for normal homeostasis and self-renewal of the active intestinal stem cell. 28753936_Msi1 has two RNA-binding domains. 29228137_leptin's direct stimulatory actions on gonadotrope GnRHR correlate with a direct inhibition of expression of the posttranscriptional regulator MSI1. There also is a direct MSI1 interaction with 3'-UTR of Gnrhr mRNA. 33110120_Interplay among p21(Waf1/Cip1), MUSASHI-1 and Kruppel-like factor 4 in activation of Bmi1-Cre(ER) reserve intestinal stem cells after gamma radiation-induced injury. 33168629_The Musashi proteins MSI1 and MSI2 are required for photoreceptor morphogenesis and vision in mice. 33175598_Constitutive Musashi1 expression impairs mouse postnatal development and intestinal homeostasis. 33373440_Control of the Anterior Pituitary Cell Lineage Regulator POU1F1 by the Stem Cell Determinant Musashi. 34309875_Assessment of MUSASHI 1 and MUSASHI 2 expression in spermatozoa and testicular tissue. |
ENSG00000135097 |
MSI1 |
220.508021 |
0.1810085408 |
-2.465870 |
0.19510370 |
158.102906 |
0.00000000000000000000000000000000000293870240597921881627905199422499188040748285559003462014925394539846967757036168243601102973480004854067715314158704131841659545898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000002869379169735606450300282161715314312700610742726539537482377266742466514245689120241533057870109146847426018211990594863891601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.4532307705755 |
9.01425906139223 |
367.127597754512 |
33.3320201385883 |
| ENSMUSG00000054280 |
215476 |
Prr14l |
protein_coding |
H3BKZ4 |
|
Reference proteome |
|
|
|
|
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 30573780_we report for the first time the finding of loss of function PRR14L mutations in myeloid neoplasia and ARCH. Knockdown of PRR14L results in altered myeloid differentiation and cell growth in vitro and our data suggest that PRR14L may play a role in cell division. |
ENSG00000183530 |
PRR14L |
670.327435 |
0.4762031728 |
-1.070351 |
0.28944086 |
13.439576 |
0.00024637134156324643971536225706131517654284834861755371093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0013354904892140106054815307246030897658783942461013793945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
446.384334058874 |
90.1578971599848 |
937.382108195738 |
141.246999462705 |
| ENSMUSG00000054459 |
26950 |
Vsnl1 |
protein_coding |
P62761 |
FUNCTION: Regulates (in vitro) the inhibition of rhodopsin phosphorylation in a calcium-dependent manner. {ECO:0000250}. |
Calcium;Direct protein sequencing;Lipoprotein;Metal-binding;Myristate;Reference proteome;Repeat |
|
|
mmu:26950; |
cytosol [GO:0005829]; membrane [GO:0016020]; calcium ion binding [GO:0005509]; negative regulation of insulin secretion [GO:0046676]; positive regulation of exocytosis [GO:0045921]; positive regulation of insulin secretion involved in cellular response to glucose stimulus [GO:0035774] |
15735716_Decreased or absent VILIP-1 expression in SCC cell subpopulations may lead to a more advanced malignant phenotype characterized by changes in adhesive ability and increased cell motility, suggestive of a tumor suppressor function 18922787_VILIP1 constitutively binds to P2X2 receptors and displays enhanced interactions in an activation- and calcium-dependent manner owing to exposure of its binding segment in P2X2 receptors 20419170_VILIP-1 overexpression decreases the susceptibility to skin carcinogenesis in experimental mouse cancer models, thus supporting its role as a tumor suppressor gene 21289216_Vsnl1 marks ureteric bud tips in embryonic kidneys, and its mosaic pattern demonstrates a heterogeneity of cell types that may be critical for normal ureteric branching. 21480386_Data show that VILIP-1 led to reduced migration of aggressive SCC cells depending on cAMP levels. 22138150_VSNL1 expresses exclusively in atrial cardiomyocytes during embryogenesis. 22479362_Findings indicate that VILIP-1 is involved in epithelial-mesenchymal transition (EMT) of squamous cell carcinoma (SCC) cells by regulating the transcription factor Snail1 in a cAMP-dependent manner. 31712124_Our results demonstrated that...Helios has a specific and important role for a correct function of hippocampal calbindin-positive CA1 pyramidal neurons regulating spinogenesis...and that Visinin-like protein 1 (VSNL1) could play an important role in such processes |
ENSG00000163032 |
VSNL1 |
5.981776 |
17.4415685029 |
4.124458 |
1.44340227 |
8.960598 |
0.00275864515544551767642356132625991449458524584770202636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0111962917686507130998752046480149147100746631622314453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.7957409064854 |
12.6953353981893 |
0.6189661729485 |
0.634215198636442 |
| ENSMUSG00000054556 |
232599 |
Gm4876 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
72.646555 |
3.6848595193 |
1.881610 |
0.61912675 |
8.574648 |
0.00340875832803420788369441751797239703591912984848022460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0133339000648734079534918350873340386897325515747070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
96.7168812390375 |
42.7914318873559 |
26.2471013433296 |
8.62017812586738 |
| ENSMUSG00000054728 |
218194 |
Phactr1 |
protein_coding |
Q2M3X8 |
FUNCTION: Binds actin monomers (G actin) and plays a role in multiple processes including the regulation of actin cytoskeleton dynamics, actin stress fibers formation, cell motility and survival, formation of tubules by endothelial cells, and regulation of PPP1CA activity. Involved in the regulation of cortical neuron migration and dendrite arborization (PubMed:30256902). {ECO:0000250, ECO:0000269|PubMed:22976292, ECO:0000269|PubMed:23041370, ECO:0000269|PubMed:30256902}. |
3D-structure;Actin-binding;Alternative splicing;Cytoplasm;Nucleus;Phosphoprotein;Protein phosphatase inhibitor;Reference proteome;Repeat;Synapse |
|
|
mmu:218194; |
cytosol [GO:0005829]; nucleus [GO:0005634]; synapse [GO:0045202]; actin binding [GO:0003779]; protein phosphatase 1 binding [GO:0008157]; protein phosphatase inhibitor activity [GO:0004864]; actin cytoskeleton organization [GO:0030036]; actin cytoskeleton reorganization [GO:0031532]; actomyosin structure organization [GO:0031032]; cell motility [GO:0048870]; cerebral cortex development [GO:0021987]; dendrite arborization [GO:0140059]; regulation of neuron migration [GO:2001222]; regulation of phosphorylation [GO:0042325]; stress fiber assembly [GO:0043149] |
22766235_This study demonistrated that Phactr1 appeared to be expressed in post-mitotic neurons in developmental brain. 28720499_It has been demonstrated that PHACTR1 gene expression increases with the progression of vascular calcification and that regulation of PHACTR1 in smooth muscle cells modulates the severity of vascular calcification. 28803787_that Phactr1 takes part in neuronal functions regulated in a spatiotemporal manner 32857129_Genetic deficiency of Phactr1 promotes atherosclerosis development via facilitating M1 macrophage polarization and foam cell formation. 32975518_Molecular basis for substrate specificity of the Phactr1/PP1 phosphatase holoenzyme. 33630758_Deficiency of macrophage PHACTR1 impairs efferocytosis and promotes atherosclerotic plaque necrosis. 33727568_PHACTR1 genetic variability is not critical in small vessel ischemic disease patients and PcomA recruitment in C57BL/6J mice. 35387477_PHACTR-1 (Phosphatase and Actin Regulator 1) Deficiency in Either Endothelial or Smooth Muscle Cells Does Not Predispose Mice to Nonatherosclerotic Arteriopathies in 3 Transgenic Mice. 36091033_PHACTR1, a coronary artery disease risk gene, mediates endothelial dysfunction. |
ENSG00000112137 |
PHACTR1 |
41.892400 |
0.0325128594 |
-4.942846 |
0.64526891 |
66.947111 |
0.00000000000000027888983934768190570979641454082330966916919413010889083892607231973670423030853271484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000079700751087127403466339380394204161301308780851471169626165647059679031372070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.46188626934312 |
1.13820041111565 |
75.7203861225307 |
18.0589069664577 |
| ENSMUSG00000054999 |
381204 |
Naaladl1 |
protein_coding |
Q7M758 |
FUNCTION: Aminopeptidase with broad substrate specificity. Has lower activity with substrates that have Asp or Glu in the P2' position, or Pro in the P3' position. Lacks activity with substrates that have both Pro in the P3' position and Asp or Glu in the P2' position. Lacks carboxypeptidase activity. Lacks dipeptidyl-peptidase IV type activity. {ECO:0000250|UniProtKB:Q9UQQ1}. |
Aminopeptidase;Calcium;Cell membrane;Disulfide bond;Glycoprotein;Hydrolase;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix;Zinc |
|
|
mmu:381204; |
apical plasma membrane [GO:0016324]; membrane [GO:0016020]; aminopeptidase activity [GO:0004177]; calcium ion binding [GO:0005509]; carboxypeptidase activity [GO:0004180]; metallopeptidase activity [GO:0008237]; protein homodimerization activity [GO:0042803]; zinc ion binding [GO:0008270]; peptide catabolic process [GO:0043171]; proteolysis [GO:0006508] |
Human_homologues 25752612_characterization of the protein product of the human NAALADL1 gene |
ENSG00000168060 |
NAALADL1 |
47.431617 |
2.0490636865 |
1.034965 |
0.40109645 |
6.597994 |
0.01020937325675054865858903951902902917936444282531738281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0340655363519878073841340437866165302693843841552734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.8358839304058 |
16.8506401956282 |
29.2015735403023 |
6.3356829850862 |
| ENSMUSG00000055782 |
26874 |
Abcd2 |
protein_coding |
Q61285 |
FUNCTION: ATP-dependent transporter of the ATP-binding cassette (ABC) family involved in the transport of very long chain fatty acid (VLCFA)-CoA from the cytosol to the peroxisome lumen (By similarity). Like ABCD1 seems to have fatty acyl-CoA thioesterase (ACOT) and ATPase activities, according to this model, VLCFA-CoA as free VLCFA is transpoted in an ATP-dependent manner into peroxisomes after the hydrolysis of VLCFA-CoA mediated by the ACOT activity of ABCD2 (By similarity). Shows overlapping substrate specificities with ABCD1 toward saturated fatty acids (FA) and monounsaturated FA (MUFA) but has a distinct substrate preference for shorter VLCFA (C22:0) and polyunsaturated fatty acid (PUFA) such as C22:6-CoA and C24:6-CoA (in vitro) (By similarity). Thus, may play a role in regulation of VLCFAs and energy metabolism namely, in the degradation and biosynthesis of fatty acids by beta-oxidation (PubMed:18854420, PubMed:16223892). {ECO:0000250|UniProtKB:P33897, ECO:0000250|UniProtKB:Q9UBJ2, ECO:0000269|PubMed:16223892, ECO:0000269|PubMed:18854420}. |
ATP-binding;Glycoprotein;Hydrolase;Lipid metabolism;Membrane;Nucleotide-binding;Peroxisome;Phosphoprotein;Reference proteome;Translocase;Transmembrane;Transmembrane helix;Transport |
|
The membrane-associated protein encoded by this gene is a member of the superfamily of ATP-binding cassette (ABC) transporters. ABC proteins transport various molecules across extra- and intra-cellular membranes. ABC genes are divided into seven distinct subfamilies (ABC1, MDR/TAP, MRP, ALD, OABP, GCN20, White). This protein is a member of the ALD subfamily, which is involved in peroxisomal import of fatty acids and/or fatty acyl-CoAs in the organelle. All known peroxisomal ABC transporters are half transporters which require a partner half transporter molecule to form a functional homodimeric or heterodimeric transporter. The function of this peroxisomal membrane protein is unknown; however this protein is speculated to function as a dimerization partner of Abcd1 and/or other peroxisomal ABC transporters. Mutations in the human gene have been observed in patients with adrenoleukodystrophy, a severe demyelinating disease. This gene has been identified as a candidate for a modifier gene, accounting for the extreme variation among adrenoleukodystrophy phenotypes. This gene is also a candidate for a complement group of Zellweger syndrome, a genetically heterogeneous disorder of peroxisomal biogenesis. [provided by RefSeq, Jul 2008]. |
mmu:26874; |
mitochondrion [GO:0005739]; peroxisomal membrane [GO:0005778]; peroxisome [GO:0005777]; ABC-type transporter activity [GO:0140359]; acyl-CoA hydrolase activity [GO:0047617]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; ATPase-coupled transmembrane transporter activity [GO:0042626]; long-chain fatty acid transporter activity [GO:0005324]; protein heterodimerization activity [GO:0046982]; protein homodimerization activity [GO:0042803]; fatty acid beta-oxidation [GO:0006635]; long-chain fatty acid import into peroxisome [GO:0015910]; myelin maintenance [GO:0043217]; negative regulation of cytokine production involved in inflammatory response [GO:1900016]; negative regulation of reactive oxygen species biosynthetic process [GO:1903427]; neuron projection maintenance [GO:1990535]; peroxisome organization [GO:0007031]; positive regulation of fatty acid beta-oxidation [GO:0032000]; positive regulation of unsaturated fatty acid biosynthetic process [GO:2001280]; response to bacterium [GO:0009617]; very long-chain fatty acid catabolic process [GO:0042760]; very long-chain fatty acid metabolic process [GO:0000038] |
14713219_Evidence against the adrenoleukodystrophy-related gene acting as a modifier of X-adrenoleukodystrophy. 16249184_LXRalpha is a negative modulator of Abcd2, acting through a novel regulatory mechanism involving overlapping SREBP and LXRalpha binding sites 18723473_Abcd1 and Abcd2 gene silencing sensitizes astrocytes for inflammation and may have a role in X-adrenoleukodystrophy 18854420_ABCD2 plays a role in th degradation of long-chain saturated and omega9-monounsaturated fatty acids and in the synthesis of docosahexanoic acid. 19556607_ABCD2 is a sterol regulatory element binding protein target gene that is highly abundant in fat and opposes the accumulation of dietary lipids generally absent from the triglyceride storage pool within adipose tissue. 22493092_an essential role for D2 in the protection from dietary erucic acid 25123288_The absence of ABCD2 altered expression of gene clusters associated with lipid metabolism, including PPARalpha signaling. 25255441_Abcd2 is a strong modifier of the metabolic impairments in peritoneal macrophages of ABCD1-deficient mice 25446110_analysis of D2 localization and the proteome of D2-containing organelles, in adipose tissue 28258215_ABCD1 and its homolog ABCD2 exist mainly as homotetramers in the peroxisomal membrane 30769094_Combined absence of ABCD1 and ABCD2 proteins resulted in the accumulation of very long-chain fatty acids in murine BV-2 cells. |
ENSG00000173208 |
ABCD2 |
33.718193 |
2.6650794833 |
1.414179 |
0.46498688 |
9.076712 |
0.00258885103826007227992289649876056500943377614021301269531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0106146376890383364877123995029251091182231903076171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.2555527582039 |
15.495736746187 |
17.7313858945445 |
4.53247150833163 |
| ENSMUSG00000056019 |
236193 |
Zfp709 |
protein_coding |
Q3THF1 |
Human_homologues FUNCTION: May be involved in transcriptional regulation. {ECO:0000250}. |
Reference proteome |
|
|
|
DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; negative regulation of transcription by RNA polymerase II [GO:0000122]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
ENSG00000257446 |
ZNF878 |
36.673900 |
0.2214481412 |
-2.174959 |
0.48568493 |
19.107744 |
0.00001235425640870829051538946213861791534327494446188211441040039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000900382935968634898768817031999844857637071982026100158691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.438577253064 |
4.26700757177245 |
56.1692556274567 |
13.3154119669076 |
| ENSMUSG00000056427 |
20564 |
Slit3 |
protein_coding |
Q9WVB4 |
FUNCTION: May act as molecular guidance cue in cellular migration, and function may be mediated by interaction with roundabout homolog receptors. |
Developmental protein;Differentiation;Disulfide bond;EGF-like domain;Glycoprotein;Leucine-rich repeat;Neurogenesis;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:20564; |
extracellular space [GO:0005615]; membrane [GO:0016020]; mitochondrion [GO:0005739]; plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; heparin binding [GO:0008201]; Roundabout binding [GO:0048495]; signaling receptor binding [GO:0005102]; animal organ morphogenesis [GO:0009887]; aortic valve morphogenesis [GO:0003180]; apoptotic process involved in luteolysis [GO:0061364]; atrioventricular valve morphogenesis [GO:0003181]; axon extension involved in axon guidance [GO:0048846]; axon guidance [GO:0007411]; cellular response to hormone stimulus [GO:0032870]; negative chemotaxis [GO:0050919]; negative regulation of cell growth [GO:0030308]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of chemokine-mediated signaling pathway [GO:0070100]; negative regulation of gene expression [GO:0010629]; response to cortisol [GO:0051414]; Roundabout signaling pathway [GO:0035385]; ventricular septum morphogenesis [GO:0060412] |
12141424_Analysis of alternative splicing and conserved domains in human and mouse slit genes 12702769_role in regulating development of the diaphragm 12954717_Data suggest that the Slit family of axon guidance molecules (Slit 1-3) and their Robo 1 and 2 receptors contribute to the topographic targeting of basal vomeronasal axons. 15091338_Experiments with triple mutant mice demonstrate a key role for Slit3 signaling in vertebrate midline commissural axon guidance. 15162513_Slit-3 and Robo-2 expression is restricted to specific, complementary subsets of mesenchyme 19741192_functions to promote angiogenesis in coordinating organogenesis during embryonic development 20941780_Study identify that the homeobox gene Nkx2-5 is required for early ventral restriction of Slit3 and that the T-box transcription factor Tbx2 mediates repression of Slit3 in nonchamber myocardium. 20947829_During blood vessel development, Slit3 gene expression (and that of Slit2) are linked to posttranscriptional regulation of Robo receptors. 21078908_The chemorepellent Slit3 promotes monocyte migration. 22981605_Robo-2-mediated targeting of P2 axons along the dorsoventral axis of the OB is controlled by Slit-3 expression 23255421_Report role of Slit3 in development of the caval veins and pericardium. 23536828_FREM1-deficient mice faithfully recapitulate many of the phenotypes seen in individuals with FREM1 deficiency and that variations in GATA4 and SLIT3 expression modulate some FREM1-related phenotypes in mice. 25325980_Heparan sulfate-deficient mouse endothelial cells was used to determine the co-reception function of heparan sulfate in Slit3-induced endothelial cell migration. 25490618_These observations from mutant Slit and Robo mice give great support for Slit/Robo role in neural crest cell migration during Enteric Nervous System development. 25691540_Cardiac defects in mutants for Robo or Slit range from membranous ventricular septum defects to bicuspid aortic valves. 28234971_While Slit1 and Robo2 are only expressed in peripheral axons and their cell bodies, Slit2, Slit3 and Robo1 are also expressed in satellite cells of the dorsal root ganglion, Schwann cells and fibroblasts of peripheral nerves. 29504949_these results indicate that SLIT3 plays an osteoprotective role by synchronously stimulating bone formation and inhibiting bone resorption, making it a potential therapeutic target for metabolic bone diseases. 29764562_A small truncated recombinant SLIT3 protein increased bone mass in an osteopenic mouse model. 30389141_these indicate that SLIT3/ROBO2 promotes chondrocyte maturation via the inhibition of beta-catenin signaling. 30726731_Slit3 is required for interaction with Robo1 on Schwann cells and fibroblasts in the nerve bridge to control cell migration and axon guidance. 32470195_Tissue-specific angiogenic and invasive properties of human neonatal thymus and bone MSCs: Role of SLIT3-ROBO1. 32644051_SLIT3 deficiency attenuates pressure overload-induced cardiac fibrosis and remodeling. 34423586_SLIT3 promotes myogenic differentiation as a novel therapeutic factor against muscle loss. 34782792_Slit3 secreted from M2-like macrophages increases sympathetic activity and thermogenesis in adipose tissue. |
ENSG00000184347 |
SLIT3 |
149.167127 |
2.4717671738 |
1.305543 |
0.28079829 |
21.292138 |
0.00000394345602791828733378262650255408061639172956347465515136718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000315003520180076387467889287297140299415332265198230743408203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
200.454273782308 |
39.2621259914741 |
81.0975547813021 |
12.1832189113426 |
| ENSMUSG00000056458 |
26448 |
Mok |
protein_coding |
Q9WVS4 |
FUNCTION: Able to phosphorylate several exogenous substrates and to undergo autophosphorylation (PubMed:10421840). Negatively regulates cilium length in a cAMP and mTORC1 signaling-dependent manner (PubMed:25243405). {ECO:0000269|PubMed:10421840, ECO:0000269|PubMed:25243405}. |
Alternative splicing;ATP-binding;Cell projection;Cytoplasm;Kinase;Magnesium;Metal-binding;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Transferase |
|
|
mmu:26448; |
ciliary base [GO:0097546]; cilium [GO:0005929]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; ATP binding [GO:0005524]; cyclin-dependent protein serine/threonine kinase activity [GO:0004693]; metal ion binding [GO:0046872]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; intracellular signal transduction [GO:0035556]; protein phosphorylation [GO:0006468] |
15327990_identification of MOK, a member of the mitogen-activated protein kinase superfamily, as one of the genes induced by a caudal-related homeobox transcription factor, Cdx2 19176597_RAGE inactivation inhibits the atherosclerosis through reducing oxLDL-induced pro-inflammatory responses and oxidative stress in hyperlipidaemia. 19303658_Suppression of Egr-1 may contribute to the protective mechanisms underlying the beneficial impact of RAGE blockade or deletion in hepatic ischemia/reperfusion injury. 24244486_the expressions of ICK/MAK/MOK proteins in the intestinal tract can be differentially and dynamically regulated, implicating a significant functional diversity within this group of protein kinases. 28336525_This provides new insight into the immune phenotype, mechanisms, and signaling pathways that operate in microglial neurotoxic activation in amyotrophic lateral sclerosis. |
ENSG00000080823 |
MOK |
49.316094 |
6.2168393326 |
2.636181 |
0.66263574 |
12.930062 |
0.00032334699078832423177823351956305941712344065308570861816406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0017012854451425750076931642240651854081079363822937011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
85.9004904385588 |
40.8563731584975 |
13.8173894872483 |
4.88211716207661 |
| ENSMUSG00000056492 |
224792 |
Adgrf5 |
protein_coding |
G5E8Q8 |
FUNCTION: Receptor that plays a critical role in lung surfactant homeostasis (PubMed:23590306, PubMed:23922714, PubMed:23684610). May play a role in controlling adipocyte function (PubMed:22971422). {ECO:0000269|PubMed:22971422, ECO:0000269|PubMed:23590306, ECO:0000269|PubMed:23684610, ECO:0000269|PubMed:23922714}. |
Autocatalytic cleavage;Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Phosphoprotein;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:224792; |
apical part of cell [GO:0045177]; cell surface [GO:0009986]; cytoplasmic vesicle [GO:0031410]; plasma membrane [GO:0005886]; G protein-coupled receptor activity [GO:0004930]; adenylate cyclase-activating G protein-coupled receptor signaling pathway [GO:0007189]; cell surface receptor signaling pathway [GO:0007166]; energy reserve metabolic process [GO:0006112]; erythrocyte development [GO:0048821]; fat cell differentiation [GO:0045444]; glomerular filtration [GO:0003094]; glucose homeostasis [GO:0042593]; macrophage activation [GO:0042116]; negative regulation of macrophage activation [GO:0043031]; pharyngeal arch artery morphogenesis [GO:0061626]; phospholipid biosynthetic process [GO:0008654]; positive regulation of phospholipid biosynthetic process [GO:0071073]; regulation of lipid metabolic process [GO:0019216]; surfactant homeostasis [GO:0043129] |
22971422_GPR116 may play a critical role in controlling adipocyte biology and systemic energy homeostasis 23590306_GPR116 functions as a molecular sensor of alveolar surfactant lipid pool sizes by regulating surfactant secretion. 23684610_GPR116 plays an indispensable role in lung surfactant homeostasis with important ramifications for the understanding and treatment of lung surfactant disorders. 23922714_Sp-D was identified as the ligand of Ig-Hepta/GPR116. 25778400_These results suggest that Ig-Hepta plays an important role in regulating macrophage immune responses, and its deficiency leads to local inflammation in the lung 26394398_Results indicate the function of adhesion receptor Gpr116 in lung and in the central nervous system vasculature. 28806758_loss of GPR116 and ELTD1 expressed by other cells accounts for the observed cardiovascular and renal defects. 30654796_Targeted disruption of ADGRF5 results in the development of airway inflammation. 31256320_these results reveal a previously unobserved vascular morphogenetic defect in Adgrf5 knockout mice, implicating a role for ADGRF5 in the initiation of subretinal vascularization. 33004624_Adhesion-GPCR Gpr116 (ADGRF5) expression inhibits renal acid secretion. 34016966_Orphan GPR116 mediates the insulin sensitizing effects of the hepatokine FNDC4 in adipose tissue. |
ENSG00000069122 |
ADGRF5 |
706.027222 |
0.4245004014 |
-1.236162 |
0.10320174 |
141.689514 |
0.00000000000000000000000000000001137028619961396123686767715243890989742248461170133001539753019685110043010503728629686376327967423094378318637609481811523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000009896697108143993331933013734540751644632593060436775489677049253680758159285122538273427039712260011583566665649414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
419.610368676376 |
30.0420134780697 |
988.480499280308 |
51.5750880551462 |
| ENSMUSG00000056602 |
320365 |
Fry |
protein_coding |
E9Q8I9 |
FUNCTION: Plays a crucial role in the structural integrity of mitotic centrosomes and in the maintenance of spindle bipolarity by promoting PLK1 activity at the spindle poles in early mitosis. May function as a scaffold promoting the interaction between AURKA and PLK1, thereby enhancing AURKA-mediated PLK1 phosphorylation. {ECO:0000269|PubMed:22753416}. |
Alternative splicing;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:320365; |
cell cortex [GO:0005938]; microtubule organizing center [GO:0005815]; site of polarized growth [GO:0030427]; spindle pole [GO:0000922]; enzyme inhibitor activity [GO:0004857]; cell morphogenesis [GO:0000902]; negative regulation of tubulin deacetylation [GO:1904428]; neuron projection development [GO:0031175] |
Human_homologues 19327996_Results suggest that MST2-, Fry-, and MOB2-mediated activation of NDR1 is crucial for the fidelity of mitotic chromosome alignment in mammalian cells. 19773279_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 22753416_Fry also binds to Aurora A and promotes Plk1 activity by binding to the polo-box domain of Plk1 and by facilitating Aurora A-mediated Plk1 phosphorylation at Thr-210. 23886946_Results suggest that Fry plays a crucial role in promoting the level of microtubule acetylation in the mitotic spindle by inhibiting the tubulin-deacetylase activity of SIRT2. |
ENSG00000073910 |
FRY |
18.190525 |
0.3027521061 |
-1.723791 |
0.61266914 |
7.930312 |
0.00486134158182970413375478813122754218056797981262207031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0181445613757486058637002912519164965488016605377197265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
9.30983633983965 |
4.07351857503918 |
30.750690590183 |
9.11038746811631 |
| ENSMUSG00000056771 |
|
Gm10010 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
35.803253 |
3.9892282981 |
1.996110 |
0.47330687 |
17.624667 |
0.00002690755445586544844852050495376261096680536866188049316406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001833807022244932853193705168948213213298004120588302612304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
56.9785538722204 |
19.0301630669585 |
14.2831017968019 |
3.82665957856771 |
| ENSMUSG00000056966 |
118446 |
Gjc3 |
protein_coding |
Q921C1 |
FUNCTION: One gap junction consists of a cluster of closely packed pairs of transmembrane channels, the connexons, through which materials of low MW diffuse from one cell to a neighboring cell. {ECO:0000250}. |
Cell junction;Cell membrane;Gap junction;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:118446; |
connexin complex [GO:0005922]; gap junction [GO:0005921]; myelin sheath [GO:0043209]; gap junction channel activity [GO:0005243]; identical protein binding [GO:0042802]; cell-cell signaling [GO:0007267]; myelination [GO:0042552]; sensory perception of sound [GO:0007605] |
12372015_Cx29 is a second connexin expressed in Schwann cells of sciatic nerve. 12900929_Comparative mapping of Y1 and Y5 receptor subtypes within cell bodies and nerve fibers in the brain. Together with physiological and electrophysiological studies, provides better understanding of NPY neural circuitries. 15293232_Cx29 is localized to the inner membrane of small myelin sheaths 16236250_Our data first provide a comprehensive and detailed pattern of Cx29 gene expression in the mouse and rat cochlea. 16435366_Thus, in contrast to connexin32 and connexin47, which are also expressed in myelinating cells, Cx29 does not contribute to the function of myelin in adult mice. 16481432_Findings demonstrated the requirement of Cx29 for normal cochlear functions and suggest that Cx29 is a new candidate gene for studying the auditory neuropathy. 17024657_Cx29 protein expression was absent from neural crest cells but appeared as neural crest cells generated precursors (embryonic day 12) both in vivo and in vitro. This identifies Cx29 as a novel marker for cells of the defined Schwann cell lineage. 17972320_Homomeric interactions of Cx29 and Cx32 require other domains: the N-terminus, transmembrane domains, and extracellular loops. Substituting the intracellular loop and/or tail of Cx32 with those of Cx29 prevents Cx32 from forming functional gap junctions. 18849090_Cx23 seems to share functional properties with pannexin (hemi) channels rather than gap junction channels of other connexins. 20926974_Induction of Cxs 29 and 32 in the injury border suggests that altered Cx expression may contribute to the propagation of injury-related and/or regeneration signals after acute brain injury. |
ENSG00000176402 |
GJC3 |
23.273651 |
2.8184688374 |
1.494912 |
0.55706734 |
7.084043 |
0.00777733297952891856202883857918095600325614213943481445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0270837530736590080537862235132706700824201107025146484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.0902774308509 |
13.4928130009163 |
12.095318201813 |
3.69925570034268 |
| ENSMUSG00000057123 |
14613 |
Gja5 |
protein_coding |
Q01231 |
FUNCTION: One gap junction consists of a cluster of closely packed pairs of transmembrane channels, the connexons, through which materials of low MW diffuse from one cell to a neighboring cell. |
Cell junction;Cell membrane;Gap junction;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:14613; |
cell projection [GO:0042995]; connexin complex [GO:0005922]; gap junction [GO:0005921]; intercalated disc [GO:0014704]; plasma membrane [GO:0005886]; connexin binding [GO:0071253]; disordered domain specific binding [GO:0097718]; gap junction channel activity [GO:0005243]; gap junction channel activity involved in atrial cardiac muscle cell-AV node cell electrical coupling [GO:0086076]; gap junction channel activity involved in AV node cell-bundle of His cell electrical coupling [GO:0086077]; gap junction channel activity involved in bundle of His cell-Purkinje myocyte electrical coupling [GO:0086078]; gap junction channel activity involved in cardiac conduction electrical coupling [GO:0086075]; gap junction hemi-channel activity [GO:0055077]; artery morphogenesis [GO:0048844]; atrial cardiac muscle cell to AV node cell communication by electrical coupling [GO:0086044]; atrial septum development [GO:0003283]; atrial septum morphogenesis [GO:0060413]; atrial ventricular junction remodeling [GO:0003294]; AV node cell to bundle of His cell communication [GO:0086067]; AV node cell to bundle of His cell communication by electrical coupling [GO:0086053]; blood vessel development [GO:0001568]; blood vessel diameter maintenance [GO:0097746]; bundle of His cell to Purkinje myocyte communication by electrical coupling [GO:0086054]; cardiac conduction [GO:0061337]; cardiac conduction system development [GO:0003161]; cell communication by chemical coupling [GO:0010643]; cell communication by electrical coupling [GO:0010644]; cell communication by electrical coupling involved in cardiac conduction [GO:0086064]; cell communication involved in cardiac conduction [GO:0086065]; cell-cell signaling [GO:0007267]; embryonic heart tube development [GO:0035050]; embryonic limb morphogenesis [GO:0030326]; foramen ovale closure [GO:0035922]; gap junction assembly [GO:0016264]; mitral valve development [GO:0003174]; negative regulation of blood pressure [GO:0045776]; negative regulation of glomerular filtration [GO:0003105]; outflow tract morphogenesis [GO:0003151]; positive regulation of cell communication by chemical coupling [GO:0010652]; positive regulation of vasoconstriction [GO:0045907]; potassium ion transport [GO:0006813]; pulmonary valve formation [GO:0003193]; regulation of atrial cardiac muscle cell action potential [GO:0098910]; regulation of atrial cardiac muscle cell membrane depolarization [GO:0060371]; regulation of AV node cell action potential [GO:0098904]; regulation of bundle of His cell action potential [GO:0098905]; regulation of cardiac muscle contraction [GO:0055117]; regulation of cell communication by electrical coupling [GO:0010649]; regulation of heart rate by cardiac conduction [GO:0086091]; regulation of membrane depolarization during cardiac muscle cell action potential [GO:1900825]; regulation of Purkinje myocyte action potential [GO:0098906]; regulation of renin secretion into blood stream [GO:1900133]; regulation of systemic arterial blood pressure [GO:0003073]; regulation of ventricular cardiac muscle cell membrane depolarization [GO:0060373]; regulation of ventricular cardiac muscle cell membrane repolarization [GO:0060307]; renal system process involved in regulation of systemic arterial blood pressure [GO:0003071]; SA node cell action potential [GO:0086015]; septum primum development [GO:0003284]; skeletal system development [GO:0001501]; vasodilation [GO:0042311]; vasomotion [GO:1990029]; ventricular cardiac muscle cell action potential [GO:0086005]; ventricular septum development [GO:0003281]; ventricular septum morphogenesis [GO:0060412] |
12011587_Cx40-deficient endothelial cells upregulate and redistribute Cx37 as a molecular adaptation to the lack of Cx40 12064615_Data suggest that the dynamic changes of connexins 43, 40 and 45 during mouse cardiac development appear to be mirrored in the human. 12697838_Cx37 and Cx40 are the only functional connexins expressed in mouse aortic endothelium and are collectively crucial for endothelial communication and are codependent on each other for optimal expression in vascular endothelium. 12700362_Cx40-deficient mice display an irregular vasomotion, which leads temporarily to a complete flow stop and suggests that Cx40-dependent cellular coupling, presumably within the endothelial cell layer, is involved in the regulation of vascular resistance. 12842919_A high incidence of cardiac malformations in connexin40-deficient mice suggests Cx40 participates in cardiac morphogenesis, likely in association with other (unknown) products whose expression may vary with the genetic background of the mice 14681045_Cx37 and Cx40 are collectively critical for endothelial communication and provide evidence of an important role for gap junctions in vascular development. 15923624_Results suggest that T-box transcription factor 5 regulation of connexin 40 plays a critical role in the exquisite developmental patterning of the forelimbs and sternum. 16037099_abrupt reoxygenation or mouse cells reduces interendothelial electrical coupling via oxidant- and PKA-dependent signaling that targets Cx40 16105048_Connexins 37, 40, and 43 were expressed in endothelial cells of the renal, lobar, arcuate, and interlobular arteries and afferent arterioles, although connexin 43 was weak in the renal and arcuate arteries 16203917_demonstration of intra-atrial block, ectopic rhythms, and altered atrial propagation in the right atrium of Cx40 deficient mice. 16284228_The findings suggest that interactions between Cx40 and Cx43 regulate communication between endothelial cells and perhaps between smooth muscle and endothelial cells as well. 16919680_Cx40 and Cx45 have overlapping and partially compensatory functions with regard to heart morphogenesis and cardiac conduction. Cx45 might be one of the genetic modifiers that can cause variations in the phenotype of connexin40-deficient animals. 17053190_Relative abundance of Cx43 and Cx40 is important determinant of atrial impulse propagation in neonatal hearts, whereby dominance of Cx40 decreases and dominance of Cx43 increases local propagation velocity. 17122196_Connexin40 is significantly downregulated in transgenic mice with targeted overexpression of tumor necrosis factor in the cardiac compartment. 17149706_Lipopolysaccharides reduces inter-endothelial electrical coupling via tyrosine-, ERK1/2-, PKA-, and PKC-dependent signaling that targets Cx40. 17255527_Cx40 connexins mediate the calcium-dependent inhibitor effects of angiotensin II and of intrarenal pressure on renin secretion and synthesis. 17483738_pulmonary expression of the Cx40 protein is progressively and markedly decreased in two different models of acute lung inflammation, but there is no causal relationship between this process and the pulmonary transmigration of neutrophils 17546509_a cross-talk between CFTR and a variety of gap junction channels. Cytoskeletal scaffolding proteins and/or other intermediate cytoplasmic proteins are likely to play a role in CFTR-connexins interaction. 17622273_Renin secretion is dependent on Cx40; Cx40-/- mice may be a genetic model of renin-dependent hypertension. 17855490_aberrant renin-producing cells in Cx40-deficient kidneys express significant amounts of COX-2 18401004_Connexin 40 hemichannels and extracellular ATP are key molecular elements of the glomerular endothelial calcium wave, which may serve important juxtaglomerular functions. 18463230_downregulation of Cx40 protein expression and resultant inhibition of GJIC contribute to coronary vascular dysfunction in diabetes. 18482576_These data show that cardiac fibroblasts express connexin40 and connexin43 which are able to establish functional communications through homo and/or heterotypic junctions to form an extensive coupled cell network. 18521823_Lipopolysaccharide plus hypoxia and reoxygenation synergistically reduce electrical coupling between microvascular endothelial cells by dephosphorylating connexin40. 18599871_Connexin40 imparts conduction heterogeneity to atrial tissue. 18662696_In calcineurin-induced cardiac hypertrophy, connexin 40 downregulation originates at the transcriptional level. 18787025_Cx40 mRNA is up-regulated by thyroid hormone (TH) acting in cardiac atria via the TH receptor. 18790841_Dissection of two Cx37-independent conducted vasodilator mechanisms by deletion of Cx40: electrotonic versus regenerative conduction. 18812700_renal COX-2 and nNOS are differentially regulated due to the elevation of renin-dependent blood pressure in mice lacking Cx40. 19109587_Cx40 can be replaced by Cx45, a connexin with a significantly lower conductivity, suggesting that the regulation of renin release is not dependent on the unique electrical properties of these channel proteins 19151513_Vitamin B(12) promotes Cx40 and HCN4 gene expression at an early stage of cardio-myocyte differentiation. 19248787_Cx30.2 and Cx40 act as counterparts in the AV-node and His-bundle, decreasing or increasing, respectively, electrical coupling and conduction velocity in these areas. 19443640_Cx40 contributes to renal blood flow autoregulation by transducing TGF-mediated signals to the afferent arteriole, a function that is independent of nitric oxide (NO). 19474190_Replacement of connexin 40 by connexin 45 causes ectopic localization of renin-producing cells in the kidney but maintains in vivo control of renin gene expression. 19486903_Neonatal murine atrial myocytes showed similar abundances of Cx40 and Cx43 proteins, while ventricular myocytes contained at least 20 times more Cx43 than Cx40. 19672618_it appears unlikely that Cx40 exerts its important function in renin-producing cells via Cx37/Cx40 heteromeric gap junctions. 20026782_Cx40-mediated gap junctional communication contributes to a quiescent nonactivated endothelium by propagating adenosine-evoked antiinflammatory signals between endothelial cells 20427707_Data support the concept that endothelial hyperpolarization through activation of SK3 contributes to exercise hyperemia and the hyperpolarization ascends the vascular tree through gap junctions formed by Cx40 to orchestrate dilation. 20530546_In situ hybridization in normal and ligation embryos confirmed that Gja5 expression is confined to arteries and regulated by flow; in mice, Gja5 (connexin 40) was also expressed in arteries. 20541007_required for hypoxia and reoxygenation-induced oxidant production increase in microvascular endothelial cells 20686449_endothelial Cx40 is not essential for the control of renin expression and/or release. Cx40 in renin-producing cells is required for their correct positioning in the juxtaglomerular area and the control of renin secretion by pressure. 20802140_Endothelial connexin 40 may participate in the control of eNOS expression levels and function in the mouse aorta. 20818502_In this study, the entire coding region of the connexin 40 gene, encoding the cardiac gap junction membrane channel protein alpha5, was sequenced in 126 unrelated probands with familial atrial fibrillation. 21239638_While Cx43 G60S/+ mutant mice had severe atrial tachycardia/fibrillation, Cx40-/- mice were resistant to carbachol-induced atrial tachycardia/fibrillation. 21597036_The connexin40 A96S mutation causes renin-dependent hypertension in mice. 21641379_Expression of caveolin-1 may be linked to expression of Cx40 and endothelial Cx43. 21986401_Cx40 is required for, and cx37 limits, postischemic hindlimb perfusion, survival and recovery. 22739121_The switch of function from the early primary interventricular ring conduction pathway to the mature apex to base activation is dependent upon upregulation of connexin40 expression in the ventricular trabeculae. 22811484_The results suggested that connexin40 plays a significant role in the renal autoregulatory response of preglomerular resistance vessels. 23090768_endothelial Cx40 is necessary to support conducted dilations initiated by endothelial agonists and to locate Cx37 into the plasma membrane. 23093775_Hypoxic pulmonary vasoconstriction requires connexin 40-mediated endothelial signal conduction. 23407022_connecxin 40 contributes to the propagation Ca2+ signaling triggered by Acetylcholine in endothelial cells expressing the M3 muscarinic receptors. 23471232_genetic polymorphisms in endothelial Cx40 can contribute to the pathogenesis of arterial disease. 24060583_heterozygous Cx40A96S mice exhibit prolonged episodes of induced atrial fibrillation and severely reduced atrial conduction velocities similar to the corresponding human patient. 24096827_Connexin40 regulates platelet function. 24614215_Data indicate the role of renin in hypertension linked to loss of connexin 40 (Cx40). 24639196_Endothelial connexin 40, but not connexin 37, is implicated in resistance of the heart to ischaemia/reperfusion injury by activation of the CD73 pathway 25241776_Cx40 is a requirement for the pressure control of renin secretion irrespective of the localization of renin secreting cells. 25369777_We conclude that endothelial expression of the chemically dysfunctional Cx40T202S reduces peripheral vasodilator capacity mediated by SKCa-dependent hyperpolarisation and also increases blood pressure. 25547341_Loss of functional endothelial connexin40 results in exercise-induced hypertension in mice. 25690864_This study showed the progressive changes of Cx 40 at protein and mRNA levels in the mouse hippocampus during and after pilocarpine-induced status epilepticus. 25712722_Cx40 and Cx37 play an in vivo role in the regulation of eNOS. 26198171_Cx37 is more markedly involved in basal NO release, release of cyclooxygenase products and the regulation of the sensitivity for Acetylcholine as compared to Cx40. 26550940_These data indicate a fine regulation of the gap junction channel make-up in function of the type and the ratio of co-expressed connexins that specifically regulates chemical and electrical coupling. 26654778_High-fat diet increases vulnerability to atrial arrhythmia by down-regulation of Cx40 via miR-27b. 26821948_Report interaction between ALK1 signaling and connexin40 in the development of arteriovenous malformations. 26883111_High CX40 expression is associated with neoplasms. 27194161_Cx40 may participate in compromised endothelial electrical couplilng response in the microvasculature during sepsis. 28122731_We also detected a significant decrease in the concentration of mitochondrial reactive oxygen species (ROS) in Cx40 KO MCECs. Cx40 overexpression in ECs significantly increased resting [Ca(2+)]mito level and calcium uptake by mitochondria in response to increased [Ca(2+)]cyto and augmented mitochondrial ROS production. These data suggest that mitochondrial Cx40 contributes to the regulation of mitochondrial calcium homeos 28124622_intermittent hypoxia (but not sleep fragmentation) causes reductions and remodeling of atrial Cx40 and Cx43. 28982669_Endothelial Cx40 contributes to the early stages of physiological angiogenesis in the developing retina, by regulating vessel growth and maturation. 29427253_Deletion of Cx40 decreases the calcium sensitivity of renin-secreting juxtaglomerular cells. 30234377_findings reveal a role of Cx40 in regulation of ROCK1 and MLC20 that contributes critically to lung vascular barrier failure in acute lung injury. 30745457_Thus, while gestation and gross development were conserved in Cx40(-/-)Panx1(-/-) mice, they exhibit cardiac hypertrophy, hypertension, and impaired endothelial-mediated vasodilation that phenocopies Cx40(-/-) mice. 31778152_Molecular basis of arrhythmic substrate in ageing murine peroxisome proliferator-activated receptor gamma co-activator deficient hearts modelling mitochondrial dysfunction. 32242217_Systemic therapy in an RNA toxicity mouse model with an antisense oligonucleotide therapy targeting a non-CUG sequence within the DMPK 3'UTR RNA. 32687896_Conjugated activation of myocardial-specific transcription of Gja5 by a pair of Nkx2-5-Shox2 co-responsive elements. 33706307_Calcium Signal Profiles in Vascular Endothelium from Cdh5-GCaMP8 and Cx40-GCaMP2 Mice. 34198942_Characteristics of Ventricular Electrophysiological Substrates in Metabolic Mice Treated with Empagliflozin. 36142603_Connexin Lateralization Contributes to Male Susceptibility to Atrial Fibrillation. |
ENSG00000265107 |
GJA5 |
8.454044 |
8.2409165122 |
3.042805 |
1.22021677 |
5.834460 |
0.01571523024066211887683763848144735675305128097534179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0492945607548940303854756450618879171088337898254394531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.5333070086966 |
13.810994344036 |
1.76355469529753 |
1.34910804775825 |
| ENSMUSG00000057286 |
20446 |
St6galnac2 |
protein_coding |
P70277 |
FUNCTION: Catalyzes the transfer of N-acetylneuraminyl groups onto glycan chains in glycoproteins (PubMed:8662927). Shows a preference for N-acetylgalactosamine (GalNAc) residues already modified by the addition of galactose or galactose followed by sialic acid in alpha-2,3 linkage (PubMed:8662927). {ECO:0000269|PubMed:8662927}. |
Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Membrane;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000269|PubMed:8662927}. |
|
mmu:20446; |
Golgi membrane [GO:0000139]; alpha-N-acetylgalactosaminide alpha-2,6-sialyltransferase activity [GO:0001665]; transferase activity [GO:0016740]; protein glycosylation [GO:0006486]; protein O-linked glycosylation [GO:0006493]; protein sialylation [GO:1990743] |
Human_homologues 17418236_Expression of the ST6-GalNAcII gene and activity of the CMP-NeuAc:GalNAc-IgA1 alpha2,6-sialyltransferase were higher in immunoglobulinA1-producing cell lines. 17480010_Observational study of gene-disease association. (HuGE Navigator) 17480010_the ADG haplotype in the ST6GALNAC2 gene is a functional regulatory variant that may contribute to the genetic susceptibility in a subset of patients in whom the desialylation of IgA1 molecules was the main causative pathogenesis of IgAN 19170967_reduced sialylation of serum IgA1 may result from decreased expression of ST6GALNAC2. 19357720_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 19357720_potential genetic interactions of C1GALT1 and ST6GALNAC2 variants influence IgA1 O-glycosylation, disease predisposition, and disease severity, and may contribute to the polygenic nature of IgA nephropathy 23548905_ST6GalNAc2 is a novel enzyme that regulates leukocyte adhesion under fluid shear 24398680_Il-6 and Il-4 reduced galactosylation of the O-glycan substrate directly via decreased expression of the galactosyltransferase C1GalT1 and, indirectly, via increased expression of the sialyltransferase ST6GalNAc-II 24520024_Results identified the sialyltransferase ST6GalNAc2 as a novel breast cancer metastasis suppressor. 24756995_High expression level of ST6GalNAcII is associated with invasive phenotype of breast cancer. 26820593_Results provide evidence that ST6GalNAcII activated the invasion in follicular thyroid cancer cells through regulating the activity of PI3K/Akt pathway. 27004849_Study provides evidence that mutations in B3GNT2, B4GALT2, and ST6GALNAC2 underlie aberrant glycosylation, and contribute to the pathogenesis of molecular subsets of colon and other gastrointestinal malignancies. 28767179_MicroRNAs miR-135b and miR-182 may reverse the resistance to 5-fluorouracil (5-FU) in colorectal cancer (CRC) cells by targeting ST6GALNAC2 via PI3K/AKT pathway, which render potential chemotherapy targets for the treatment of CRC. 29030743_miR-182/-135b/ST6GALNAC2/PI3K/AKT axis could function as regulator of progression in colorectal cancer |
ENSG00000070731 |
ST6GALNAC2 |
170.364441 |
3.0646511472 |
1.615723 |
0.28642812 |
29.748911 |
0.00000004917825069273297104032824519047328859500112230307422578334808349609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000005385212901162535266311008619322109325366909615695476531982421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
248.352049204003 |
49.6315918477277 |
81.0376246025709 |
12.4762098558056 |
| ENSMUSG00000057315 |
231532 |
Arhgap24 |
protein_coding |
Q8C4V1 |
FUNCTION: Rho GTPase-activating protein involved in cell polarity, cell morphology and cytoskeletal organization. Acts as a GTPase activator for the Rac-type GTPase by converting it to an inactive GDP-bound state. Controls actin remodeling by inactivating Rac downstream of Rho leading to suppress leading edge protrusion and promotes cell retraction to achieve cellular polarity. Able to suppress RAC1 and CDC42 activity in vitro. Overexpression induces cell rounding with partial or complete disruption of actin stress fibers and formation of membrane ruffles, lamellipodia, and filopodia. Isoform 2 is a vascular cell-specific GAP involved in modulation of angiogenesis (By similarity). {ECO:0000250}. |
Alternative splicing;Angiogenesis;Cell junction;Cell projection;Coiled coil;Cytoplasm;Cytoskeleton;Developmental protein;Differentiation;GTPase activation;Phosphoprotein;Reference proteome |
|
|
mmu:231532; |
adherens junction [GO:0005912]; cell projection [GO:0042995]; cytoplasm [GO:0005737]; cytoskeleton [GO:0005856]; focal adhesion [GO:0005925]; GTPase activator activity [GO:0005096]; activation of GTPase activity [GO:0090630]; angiogenesis [GO:0001525]; cell differentiation [GO:0030154]; negative regulation of Rac protein signal transduction [GO:0035021]; negative regulation of ruffle assembly [GO:1900028]; Rac protein signal transduction [GO:0016601]; wound healing, spreading of epidermal cells [GO:0035313] |
21911940_Increased levels of active Rac1 and Cdc42 were measured in Arhgap24 knockdown experiments, which influenced podocyte cell shape and membrane dynamics. |
ENSG00000138639 |
ARHGAP24 |
598.148078 |
0.2805239098 |
-1.833804 |
0.17307023 |
107.760876 |
0.00000000000000000000000030324376925890719961333492077572223944800494316499121164865299904943828079861845026243827305734157562255859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000167963967030969986511982786486401900614368236560128764743471288349574432885447095031850039958953857421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
259.541951832938 |
31.2102010430697 |
925.204386431286 |
80.3450820241915 |
| ENSMUSG00000057346 |
223672 |
Apol9a |
protein_coding |
A0A2R8W6T2 |
Human_homologues FUNCTION: May affect the movement of lipids in the cytoplasm or allow the binding of lipids to organelles.; + ;FUNCTION: May play a role in lipid exchange and transport throughout the body. May participate in reverse cholesterol transport from peripheral cells to the liver (By similarity). {ECO:0000250}.; + ;FUNCTION: May affect the movement of lipids in the cytoplasm or allow the binding of lipids to organelles.; + ;FUNCTION: May play a role in lipid exchange and transport throughout the body. May participate in reverse cholesterol transport from peripheral cells to the liver. |
Reference proteome |
Human_homologues NA; + ;NA; + ;NA; + ;NA |
|
|
extracellular space [GO:0005615]; high-density lipoprotein particle [GO:0034364]; very-low-density lipoprotein particle [GO:0034361]; chloride channel activity [GO:0005254]; lipid binding [GO:0008289]; lipid transport [GO:0006869]; lipoprotein metabolic process [GO:0042157] |
27697524_ApoL9 is a phosphatidylethanolamine -binding protein that may have important roles in several cellular processes that involve this phospholipid. |
ENSG00000128284+ENSG00000100336+ENSG00000128335+ENSG00000100342 |
APOL3+APOL4+APOL2+APOL1 |
71.802150 |
2.2303054487 |
1.157241 |
0.33088840 |
11.996042 |
0.00053313667660978385220982289638413931243121623992919921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0026391738245434602852523831018061173381283879280090332031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.805967740134 |
23.3235235267457 |
45.1982789165819 |
8.01921879051378 |
| ENSMUSG00000057457 |
18675 |
Phex |
protein_coding |
P70669 |
FUNCTION: Peptidase that cleaves SIBLING (small integrin-binding ligand, N-linked glycoprotein)-derived ASARM peptides, thus regulating their biological activity (By similarity). Cleaves ASARM peptides between Ser and Glu or Asp residues (By similarity). Regulates osteogenic cell differentiation and bone mineralization through the cleavage of the MEPE-derived ASARM peptide (PubMed:11159866, PubMed:18597632, PubMed:26051469). Promotes dentin mineralization and renal phosphate reabsorption by cleaving DMP1- and MEPE-derived ASARM peptides (PubMed:26051469). Inhibits the cleavage of MEPE by CTSB/cathepsin B thus preventing MEPE degradation (By similarity). {ECO:0000250|UniProtKB:P78562, ECO:0000269|PubMed:11159866, ECO:0000269|PubMed:26051469, ECO:0000305|PubMed:18597632}. |
Biomineralization;Cell membrane;Disulfide bond;Glycoprotein;Hydrolase;Membrane;Metal-binding;Metalloprotease;Protease;Reference proteome;Signal-anchor;Transmembrane;Transmembrane helix;Zinc |
|
|
mmu:18675; |
endoplasmic reticulum [GO:0005783]; Golgi apparatus [GO:0005794]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; metal ion binding [GO:0046872]; metalloendopeptidase activity [GO:0004222]; bone development [GO:0060348]; bone mineralization [GO:0030282]; cellular response to parathyroid hormone stimulus [GO:0071374]; cellular response to vitamin D [GO:0071305]; lung development [GO:0030324]; odontogenesis [GO:0042476]; organophosphate metabolic process [GO:0019637]; protein processing [GO:0016485]; proteolysis [GO:0006508]; response to growth hormone [GO:0060416]; response to insulin-like growth factor stimulus [GO:1990418]; response to sodium phosphate [GO:1904383] |
11811562_Phex protein is expressed in osteoblasts and osteocytes during the embryonic and postnatal periods 12110521_Glucocorticoid regulation of the murine PHEX gene. 12220505_MEPE may be involved in the pathogenesis defective mineralization due to Phex deficiency in X-linked hypophosphatemia (XLH) and the Hyp-mouse. 12414538_mutagen-induced point mutation in phex causes exon skipping, x-linked hypophosphatemia, and rickets 12456809_findings suggest that hypophosphatemia bone and teeth abnormalities partially correct after phosphate-regulating gene with homologies to endopeptidases on the X chromosome (PHEX) gene transfer but additional factors of PHEX expression are likely critical 14751570_rickets and osteomalacia are not the only causes of decreased vertebral growth in Hyp mice and Phex protein may influence bone growth and mineralization by distinct pathways 15050894_Phex may control mineralization and removal of hypertrophic chondrocytes and cartilage matrix in growth plate by regulating the synthesis and deposition of certain bone matrix proteins and proteases such as matrix metalloproteinase 9 16084134_downregulation of Phex expression by PTH in vitro is associated with inhibition of matrix mineralization, consistent with a role for Phex in bone mineralization 16890604_TNF-alpha-mediated reduction in Phex protein is at least in part responsible for inhibition of osteoblast mineralization, and the described mechanism may contribute to the abnormal bone metabolism associated with IBD. 17251516_loss of function is related to defect of type II sodium-dependent phosphate co-transporter in [developing] teeth 17710565_A new mutation was mapped to X-chromosome between 65.4 cM and 66.6 cM, where Phex gene resides. Sequence analysis revealed a unique T-to-C transition mutation resulting in Phe-to-Ser substitution at amino acid 80 of PHEX protein. 17848631_PHEX functions in bone to coordinate bone mineralization and systemic phosphate homeostasis by directly regulating the mineralization process and producing FGF23. 17942069_findings suggest that loss of Phex function is related to overexpression of FGF23 in teeth, which is an intrinsic defect of Hyp mouse teeth. 18289812_Phex Hyp-Duk/Y mice develop endolhympattic hydrops without evidence of endolymphatic duct obstruction 18597632_PHEX rescued the inhibition of osteoblast culture mineralization by pASARM, and mass spectrometry of cleaved peptides obtained after pASARM-PHEX incubations identified pASARM as a substrate for PHEX 19082853_The PHEX transgene corrects mineralization defects in 9-month-old hypophosphatemic mice. 19584304_Our in vivo studies provide genetic evidence for a pathological role of increased FGF23 activities in regulating abnormal phosphate homeostasis in Hyp (PHEX-deficient) mice. 19775205_OPN ASARM inhibits mineralization by binding to hydroxyapatite in a phosphorylation-dependent manner and can be cleaved by PHEX. 20204609_Preproenkephalin deletion in Hyp mice (Phex-deficient) partially rescues their bone mineralization defect. 20817730_Cooperative role of NF-{kappa}B and poly(ADP-ribose) polymerase 1 (PARP-1) in the TNF-induced inhibition of PHEX expression in osteoblasts. 21177780_ASARM peptides likely play a bone PHEX-dependent role in renal phosphate regulation and FGF23 expression. 21507898_PHEX and DMP1 control a common pathway regulating bone mineralization and FGF23 production, the latter involving activation of the FGFR signaling in osteocytes. 22006791_These data suggest that Phex mutations alter the responsiveness of bone cells to extracellular phosphate concentrations and may create a lower set point for 'normal' phosphate levels. 22991293_results identify full-length OPN as novel, physiologically relevant substrates for PHEX, suggesting accumulation of mineralization-inhibiting OPN fragments may contribute to the mineralization defect seen in the osteomalacic bone characteristic of XLH/HYP 23028440_Mutation in Phex gene predisposes BALB/c-Phex(Hyp-Duk)/Y mice to otitis media. 23266491_demonstrates a previously unknown impact of retinol on the cell-autonomous mineralization defect of Phex-deficient osteoblasts 23652555_Hyp/Lrp6 double mutants developed the same reduced bone mineralization,hypophosphatemia & high FGF23 levels as Hyp(-) mice, showing that WNT signaling does not affect the FGF23-induced phosphate wasting. 23700148_the phex mutation may create the lower set point for extracellular phosphate concentrations in a murine model of X-linked hypophosphatemia 34326685_Osteocytes but not osteoblasts directly build mineralized bone structures. 35654784_Function of PHEX mutations p.Glu145* and p.Trp749Arg in families with X-linked hypophosphatemic rickets by the negative regulation mechanism on FGF23 promoter transcription. 36011266_PHEX(L222P) Mutation Increases Phex Expression in a New ENU Mouse Model for XLH Disease. |
ENSG00000102174 |
PHEX |
1907.887184 |
0.1512017419 |
-2.725453 |
0.08699966 |
927.662093 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000951730888364226396813558470282124006927517165174950796040009 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000144967648915638962768316806289927116365490736908357520725838 |
Yes |
No |
481.258635079989 |
29.0392061707882 |
3182.89081115968 |
133.130068562586 |
| ENSMUSG00000057614 |
14677 |
Gnai1 |
protein_coding |
B2RSH2 |
FUNCTION: Guanine nucleotide-binding proteins (G proteins) function as transducers downstream of G protein-coupled receptors (GPCRs) in numerous signaling cascades. The alpha chain contains the guanine nucleotide binding site and alternates between an active, GTP-bound state and an inactive, GDP-bound state. Signaling by an activated GPCR promotes GDP release and GTP binding. The alpha subunit has a low GTPase activity that converts bound GTP to GDP, thereby terminating the signal. Both GDP release and GTP hydrolysis are modulated by numerous regulatory proteins (By similarity). Signaling is mediated via effector proteins, such as adenylate cyclase. Inhibits adenylate cyclase activity, leading to decreased intracellular cAMP levels (By similarity). The inactive GDP-bound form prevents the association of RGS14 with centrosomes and is required for the translocation of RGS14 from the cytoplasm to the plasma membrane. Required for normal cytokinesis during mitosis. Required for cortical dynein-dynactin complex recruitment during metaphase (By similarity). {ECO:0000250|UniProtKB:P10824, ECO:0000250|UniProtKB:P63096}. |
3D-structure;Cell cycle;Cell division;Cell membrane;Cytoplasm;Cytoskeleton;GTP-binding;Lipoprotein;Magnesium;Membrane;Metal-binding;Mitosis;Myristate;Nucleotide-binding;Nucleus;Palmitate;Reference proteome;Transducer;Transport |
|
|
mmu:14677; |
cell cortex [GO:0005938]; cell cortex region [GO:0099738]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; heterotrimeric G-protein complex [GO:0005834]; membrane [GO:0016020]; membrane raft [GO:0045121]; midbody [GO:0030496]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; protein-containing complex [GO:0032991]; G protein-coupled receptor binding [GO:0001664]; G protein-coupled serotonin receptor binding [GO:0031821]; G-protein beta/gamma-subunit complex binding [GO:0031683]; GDP binding [GO:0019003]; GTP binding [GO:0005525]; GTPase activating protein binding [GO:0032794]; GTPase activity [GO:0003924]; magnesium ion binding [GO:0000287]; adenylate cyclase-modulating G protein-coupled receptor signaling pathway [GO:0007188]; cell cycle [GO:0007049]; cell division [GO:0051301]; cellular response to forskolin [GO:1904322]; G protein-coupled receptor signaling pathway [GO:0007186]; negative regulation of synaptic transmission [GO:0050805]; positive regulation of protein localization to cell cortex [GO:1904778]; regulation of cAMP-mediated signaling [GO:0043949]; regulation of mitotic spindle organization [GO:0060236] |
12183468_Gi-mediated signaling pathways are activated by G12/G13 and are sufficient to induce integrin alpha(IIb)beta3 activation 14623889_Gi, but not Gq or G12/13, signaling pathways are required for activation of Akt in platelets 14715142_Loss of G(ialpha1) amplifies the responsiveness of CA1 postsynaptic neurons to stimuli that strengthen synaptic efficacy, thereby diminishing synapse-specific plasticity required for new memory formation. 17023430_lysophospholipids regulate integrin-mediated adhesion of splenic B cells to ICAM-1 through G(i) and G(12)/G(13) family G-proteins but not through G(q)/G(11) 18583341_Galpha (i) is activated and subsequently uncoupled from receptor-Galpha(i) signaling by Pasteurella multocida toxin 18832081_Report role of Gnai1 in the control of in vivo heart rate dynamics. 19706594_PKA and GRK phosphorylation of the beta(2)AR has distinct roles for agonist dose-dependent coupling to G(i) proteins in cardiac myocytes 20097574_CKbeta8- and CKbeta8-1-induced activation of ERK1/2 is mediated by the G(i)/G(o) protein, PLC, and PKCdelta. 20586915_analysis of a novel Gi, P2Y-independent signaling pathway mediating Akt phosphorylation in response to thrombin receptors 20803555_in osteoblasts CB2 targets a Gi protein-cyclin D1 mitogenic axis 21131397_The decrease in beta(2)-AR may account for additional relaxation impairment, given that there is no enhancement over nontransgenic after pertussis toxin, despite AC5 overexpression. 21158412_Activation of the Rsg14-Galphai1-GDP signaling complex is regulated by Ric8. 21220323_Data show that mice lacking G(o2)alpha, but not those lacking alpha subunits of either G(o1) or any G(i) proteins, handle glucose loads more efficiently than wild-type (WT) mice. 21880739_RGS14 can form complexes with GPCRs in cells that are dependent on Galpha(i/o) and these RGS14.Galpha(i1).GPCR complexes may be substrates for other signaling partners such as Ric-8A 22586107_Results suggest a model in which the Gbetagamma dimer that is released as a result of the dissociation from Galpha(o) upon activation of mGluR6 closes the TRPM1 channel, perhaps via a direct interaction. 23236180_Mice with mutations of Gnai1 or Gnai2 have neither fusions of ribs nor lumbar vertebrae, but loss of both Gnai3 and one of the other two genes increases the number and severity of rib fusions without affecting the lumbar fusions. 23250758_Inactive Galpha(i1)-GDP enhances the affinity of RGS14 for H-Ras-GTP in live cells, resulting in a ternary signaling complex that is further regulated by G protein-coupled receptors. 23404499_leucine can directly facilitate insulin signaling through a Galphai protein-dependent intracellular signaling pathway 24478091_By using mice deficient in individual Galphai/o G-protein subunits, authors demonstrate that Galphai1 and Galphai3 are the critical in vivo targets of ADP-ribosylation underlying vasoactive amine sensitization elicited by pertussis toxin exposure. 25825741_Data show that guanine nucleotide-binding protein G(i) subunit alpha-1 and alpha-3 (Galphai1/3) can interact with CD14 antigen/Grb2-associated binding protein Gab1, which modulates macrophage polarization in vitro and in vivo. 25933021_acidosis in inflamed tissues may be a decisive factor to regulate switching of PKA and PKCepsilon dependence via proton-sensing G-protein-coupled receptors. 26620557_stimulation of GPR17 by the small molecule agonist MDL29,951 (2-carboxy-4,6-dichloro-1H-indole-3-propionic acid) decreases myelin basic protein expression levels mainly by triggering the Galphai/o signaling pathway. 26763459_Gnai1 missense mutation is responsible of hyperpigmentation in mouse model. 27534535_Gnai1 function is impaired in the spinal cord of Ews/Ewsr1 KO mice 27660326_LGN and Galphai participate in a long-inferred signal that originates outside the bundle to model its staircase-like architecture, a property that is essential for direction sensitivity to mechanical deflection and hearing. 29022064_This study presents an unexpected proinflammatory switch from Galphas to GalphaI glp1r signaling in burn monocytes which promotes ERK1/2 and NF-kappaB activation 31173603_AC6 is tightly regulated by intrinsic receptor-independent Gi activity. 32532984_Adipocyte Gi signaling is essential for maintaining whole-body glucose homeostasis and insulin sensitivity. 32957635_Allosteric Inhibition of Adenylyl Cyclase Type 5 by G-Protein: A Molecular Dynamics Study. 33754034_Requirement of Galphai1 and Galphai3 in interleukin-4-induced signaling, macrophage M2 polarization and allergic asthma response. 34871353_In Vivo Metabolic Roles of G Proteins of the Gi Family Studied With Novel Mouse Models. |
ENSG00000127955 |
GNAI1 |
20.558804 |
0.0160988998 |
-5.956894 |
1.22199662 |
34.475453 |
0.00000000431660912095523133041642343632297851296897306383471004664897918701171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000555325930155321665651095759154431963366960189887322485446929931640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.631797627893991 |
0.601472683577264 |
39.2447705204493 |
14.5218363510131 |
| ENSMUSG00000057722 |
16847 |
Lepr |
protein_coding |
P48356 |
FUNCTION: Receptor for hormone LEP/leptin (Probable) (PubMed:11861497). On ligand binding, mediates LEP central and peripheral effects through the activation of different signaling pathways such as JAK2/STAT3 and MAPK cascade/FOS (PubMed:10799542, PubMed:25383904, PubMed:11923481, PubMed:11861497). In the hypothalamus, LEP acts as an appetite-regulating factor that induces a decrease in food intake and an increase in energy consumption by inducing anorexinogenic factors and suppressing orexigenic neuropeptides, also regulates bone mass and secretion of hypothalamo-pituitary-adrenal hormones (PubMed:10660043, PubMed:12594516). In the periphery, increases basal metabolism, influences reproductive function, regulates pancreatic beta-cell function and insulin secretion, is pro-angiogenic and affects innate and adaptive immunity (PubMed:25383904, PubMed:11923481). Control of energy homeostasis and melanocortin production (stimulation of POMC and full repression of AgRP transcription) is mediated by STAT3 signaling, whereas distinct signals regulate NPY and the control of fertility, growth and glucose homeostasis (PubMed:12594516). Involved in the regulation of counter-regulatory response to hypoglycemia by inhibiting neurons of the parabrachial nucleus (PubMed:25383904). Has a specific effect on T lymphocyte responses, differentially regulating the proliferation of naive and memory T-cells. Leptin increases Th1 and suppresses Th2 cytokine production (PubMed:9732873). {ECO:0000269|PubMed:10660043, ECO:0000269|PubMed:10799542, ECO:0000269|PubMed:11861497, ECO:0000269|PubMed:11923481, ECO:0000269|PubMed:12594516, ECO:0000269|PubMed:25383904, ECO:0000269|PubMed:9732873, ECO:0000305|PubMed:25232147}.; FUNCTION: [Isoform A]: May transport LEP across the blood-brain barrier. Binds LEP and mediates LEP endocytosis (PubMed:17620316, PubMed:20223942). Does not induce phosphorylation of and activate STAT3 (PubMed:11923481, PubMed:20223942). {ECO:0000269|PubMed:11923481, ECO:0000269|PubMed:17620316, ECO:0000269|PubMed:20223942}.; FUNCTION: [Isoform E]: Antagonizes Isoform A and isoform B-mediated LEP binding and endocytosis. {ECO:0000269|PubMed:17620316}. |
Alternative splicing;Cell membrane;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Obesity;Phosphoprotein;Receptor;Reference proteome;Repeat;Secreted;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:16847; |
basolateral plasma membrane [GO:0016323]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; receptor complex [GO:0043235]; cytokine binding [GO:0019955]; cytokine receptor activity [GO:0004896]; identical protein binding [GO:0042802]; leptin receptor activity [GO:0038021]; peptide hormone binding [GO:0017046]; protein-hormone receptor activity [GO:0016500]; transmembrane signaling receptor activity [GO:0004888]; angiogenesis [GO:0001525]; bone growth [GO:0098868]; cholesterol metabolic process [GO:0008203]; cytokine-mediated signaling pathway [GO:0019221]; eating behavior [GO:0042755]; energy homeostasis [GO:0097009]; glial cell proliferation [GO:0014009]; gluconeogenesis [GO:0006094]; glucose homeostasis [GO:0042593]; glycogen metabolic process [GO:0005977]; leptin-mediated signaling pathway [GO:0033210]; negative regulation of autophagy [GO:0010507]; negative regulation of eating behavior [GO:1903999]; negative regulation of gluconeogenesis [GO:0045721]; negative regulation of hydrolase activity [GO:0051346]; negative regulation of locomotor rhythm [GO:1904060]; phagocytosis [GO:0006909]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of insulin secretion involved in cellular response to glucose stimulus [GO:0035774]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of protein phosphorylation [GO:0001934]; regulation of bone remodeling [GO:0046850]; regulation of feeding behavior [GO:0060259]; regulation of metabolic process [GO:0019222]; regulation of transport [GO:0051049]; response to leptin [GO:0044321]; sexual reproduction [GO:0019953]; signal transduction [GO:0007165]; T cell differentiation [GO:0030217] |
11801369_Vagal afferent neurones that express the cholecystokinin type A receptor and cocaine- and amphetamine-related transcript, may also express the long form of the leptin receptor. 11967222_possible role in the regulation of oocyte maturation 11980179_Expression of the Lepr gene revealed transcripts in the yok sac and various CNS and mesoderm-derived tissues. Later, it is made near the eye of embryos and adults. 12006366_effects of changing type and amount of dietary fats on leptin receptor mRNA expression 12010881_presence of the long leptin receptor isoform in the absorptive cells of the small intestine, suggesting that leptin could have a physiological role in the regulation of nutrient absorption 12196522_Intracellular amino acids 31-36 of LRb play a critical role in Jak2 activation and contain a loose homology motif found in other Jak2-activating cytokine receptors. 12413939_role in natural killer (NK) cell development evidenced by decline in NK cells in the liver, spleen, lung, and peripheral blood 12419326_non-pregnant and pregnant states are accompanied by differently glycosylated isoforms of sOB-R whose physiological relevance remains to be determined 12424222_Divergent selection on fatness in mice indicates a regulation system independent of the leptin receptor. 12594516_STAT3 signalling is required for leptin regulation of energy balance but not reproduction 12730408_The effect of spontaneous recessive mutations in this receptor is studied, using genetic crossing. 12770749_expression of leptin and its functional receptor (OB-Rb) in mouse oocyte and preimplantation embryo 12925533_leptin receptors have roles in adiposity, dyslipidemia, and insulin resistance 12951635_Mutation in the C2 domain of Leptin receptor alone might not result in impaired leptin binding or signaling. 14525952_Nonredundant function for the individual receptor chains within the homomeric Leptin receptor complex. Based on these data, we propose a possible model for LR clustering. 14527166_Leptin receptor isoforms were found in the fungiform and circumvallate papillae taste buds. 14592964_results suggest that leptin suppresses behavioral responses to sweet substances through its action on leptin receptor in taste cells 14600075_Now, we show that the transgene, in the homozygous state, almost fully corrects the excess adiposity of LEPR-deficient (db/db) mice. 14615386_The effects of leptin on vascular lesion growth are specifically mediated by the leptin receptor. Strong expression of this receptor was demonstrated in endothelial cells, SMCs, & macrophages within lesions developing in WT mouse vessels after injury. 15166788_STAT3-independent signals triggered by the leptin receptor are clearly important in fertility [review]. 15256362_Important role for leptin receptors and osteopontin in the pathogenesis of nonalcoholic steatohepatitis. 15358114_in both liver and kidney, expression of apoM was significantly lower in leptin deficient ob/ob mice and in leptin-receptor deficient db/db mice than in control mice 15389315_Suppression of Lepr gene expression by PGK-neo is phenotypically equivalent to deletion of the Lepr signaling motifs, and therefore Lepr(neo/neo) mouse may be used to investigate conditional gene repair of Lepr signaling deficiency. 15561935_Leptin receptor-STAT3 signal is central to both the control of energy expenditure by leptin and the neuroendocrine regulation of the sympathetic nervous system and thyroid. 15604114_Tyr1138 of leptin receptor and SOCS3 represent major effector pathways for the feedback inhibition of LRb signaling. 15616027_Deficient mice were highly susceptible to Listeria monocytogenes, and the elimination of bacteria from the liver was inhibited. 15644325_leptin receptor expression in the liver is regulated by leptin and food deprivation 15654653_Study suggests that the severity of the genomic db/db-mutation expression may be modified by etradiol treatment. 15737466_differential expression of leptin receptors in implantation sites compared to interimplantation sites suggests that the leptin/leptin receptor system may be a delicate regulator of the implantation process 15870101_body weight, adiposity, serum leptin concentrations, and glucose intolerance are proportional to hypothalamic LEPR deficiency, however, fertility and cold tolerance remain intact unless hypothalamic LEPR deficiency is complete 16054060_in addition to leptin receptor/STAT3-mediated adiposity signals, non-leptin receptor/STAT3 leptin signals mediate an important adiposity-independent role in promoting glycemic control 16099070_Levels of expression altered in fetuses from pregnant dams on a protein-restricted diet. 16284652_LEPR-B has a role in obesity, diabetes, and infertility in a transgenic mouse model 16339386_SNGFR is elevated in this mouse model of DM2 (B6.Cg-m(+/+)Lepr(db)/J (db/db)) , is suppressed by nNOS inhibition, and is modulated by TGF influences that are altered by the diabetic state and responsive to changes in extracellular fluid volume. 16380490_metabolic disturbances elicited by a type 2 diabetes syndrome (insulin and/or leptin resistance, but not hypercorticism) appear to suppress type 1 diabetes development in NOD-Lepr(db-5J)/Lt by inhibiting activation of T-effector cells 16484328_in leptin-receptor deficient diabetes mice, the haplotype at chromosome 5 is associated with beta-cell mass and circulating insulin concentrations 16540470_a conserved cluster of residues Leu370, Ala407, Tyr409, His417, and His418 forms the center of binding site III of the LR 16705160_Ser523 was highly phosphorylated in HEK293 cells independently of LRb-Jak2 activation, suggesting a potential role for the phosphorylation of Ser523 in the regulation of LRb by other pathways. 16751422_Leptin receptor (ObR) is expressed on resting normal mouse CD4+ and CD8+ T cells and on B cells and monocyte/macrophages, plays a role in lymphocyte survival, and alters ObR/STAT-3-mediated signaling in T cells. 16825198_Results suggest that leptin signaling promotes the growth of mammary tumors and increases the expression of vascular endothelial growth factor and its receptor type two (VEGF-R2). 16920925_leptin receptor deficiency affects the immune system indirectly via changes in the systemic environment 17095713_The molecular mechanism by which the leptin receptor mediates neointima formation and vascular smooth muscle cell proliferation is largely independent of the STAT3-dependent signaling pathways involved in energy balance. 17202473_Thus, these data suggest that, although LRb-->STAT3 signaling is crucial for the regulation of feeding, it is not required for the acute or chronic regulation of orexigenic ARC neurons. 17323382_all receptor subtypes could mediate the binding and endocytosis of leptin, although ObRb was expressed at a lower level than ObRa, ObRc, and ObRd after transient transfection 17400175_leptin attenuates renal 1alpha-hydroxylase gene expression through activated leptin receptor. Furthermore, leptin appears to act indirectly on renal proximal tubules to regulate 1alpha-hydroxylase gene expression. 17415414_Leptin activates autoinhibitory signals via leptin receptor site tyrosine985 to attenuate the anti-adiposity effects of leptin, especially in females, potentially contributing to leptin insensitivity in obesity. 17495001_the relative contribution to whole-body energy metabolism from peripheral leptin signaling is insignificant in vivo; leptin signaling in adipocyte is a distinct short-loop negative feedback regulation of leptin production independent of metabolic status 17620316_Soluble leptin receptor inhibits leptin transport. 17680264_support a possible action of leptin on the cardiovascular system by demonstrating expression of the leptin receptor in different types of vascular lesions 17690315_Identify a critical role for leptin/leptin receptor pathway in the modulation of the regulatory immune response in atherosclerosis, and suggest that alteration in regulatory immunity may predispose obese individuals to atherosclerosis. 17726024_the phosphorylation of Tyr(1077) on LepRb during receptor activation, substantiate the hypothalamic regulation of STAT5 and S6 by leptin, and define the alternate LepRb signaling pathways 17879067_analysis of leptin and its receptor in mouse mammary gland 17909627_Together, these data provide genetic evidence for cross-talk between leptin and insulin signaling pathways in the regulation of islet biology, with implications for beta cell dysfunction in obesity. 18039787_The developmental changes observed for leptin receptors unexpectedly failed to correlate with the entry of leptin into brain, and this may indicate different functions of the receptors in neonates and adults. 18042135_Absence of leptin receptor signaling leads to obesity and chronic hyperglycemia by 4 weeks of age in leptin receptor (lepr)-null mutant (knockout) mice, a type 2 model of diabetes mice. 18068663_In WBN/Kob rats, this mutation might promote the onset of severe pancreatitis, leading to the rapid development of diabetes mellitus. 18162515_Collective and individual functions of leptin receptor modulated neurons controlling metabolism and ingestion. 18182468_in these studies macrophage leptin receptor expression did not have an effect on aortic root atherosclerotic lesion formation 18258679_Nonredundant roles of the Lepr in ventromedial hypothalamic nucleus neurons in energy homeostasis. 18583419_acts in the periphery to reduce the sensitivity of thymocytes to glucocorticoid-mediated apoptosis in vivo in obesity 18718905_leptin stimulates non-JAK2 tyrosine kinase(s), including the Src family members, which phosphorylate JAK2, STAT3, and other molecules downstream of LEPRb 18753291_Afferent arteriole responses to increases in renal perfusion pressure and angiotensin II remain intact in db/db mice. Diabetic renal disease occurs in db/db mice independently of elevated blood pressure. 19052144_lack of lepr-induced STAT3 signaling is associated with reduced cytokine production following DSS and Con A administration, but it appears to sensitize mice to the effects of proinflammatory mediators 19150989_Bardet-Biedl syndrome (BBS) proteins mediate LepR trafficking and that impaired LepR signaling underlies energy imbalance in BBS. 19156569_oncogenic transformation of nonmalignant mammary cell lines increased expression of leptin receptor 19177596_In leptin receptor-deficient db/db mice, the expression of ATP-citrate lyase was selectively elevated in the liver but not in the white adipose tissue. 19182604_aging and type 1 and type 2 diabetes impair intrinsic adipose-derived stromal cell function in db/db diabetic mice 19196818_Leptin receptor polymorphisms have a role in lung function decline in COPD 19223399_Variations in leptin signalling through the short splice variant of the Ob receptor do not contribute to dysregulation of glucocorticoid metabolism. 19350671_LepRb distribution in the brain; found LepRb mRNA expression in hypothalamic & extrahypothalamic sites described before & expression in insular cortex, lateral septal nucleus, medial preoptic area, rostral linear nucleus, & Edinger-Westphal nucleus 19524014_Leptin has a positive short-term effect on the regulation of OB-Rb mRNA in C2C12 cells, involving both Sam68 and ERKs. 19582570_Leptin receptor mice show a subtle trend toward higher body weight and insulin levels, lower oxygen, carbon dioxide production, respiratory exchange ratio and temperature suggesting the short isoforms may play an additional role in energy homeostasis. 19628573_activation of mTORC1-dependent arcuate nucleus pS6 in response to fasting and leptin deficiency in leptin receptor-expressing Agouti-related protein neurons 19656487_The neuronal population of leptin receptor in lateral hypothalamic area represents a major link between anorectic leptin action and the mesolimbic dopamine system. 19706988_The db gene heterozygote, but not homozygote, acquires a compensatory mechanism suppressing beta-cell apoptosis and augmenting the capacity of beta-cell function. 19766595_A novel proximal promoter of OB-RGRP was identified that showed robust activity in neuron-derived, but not non-neuronal cell lines. 19961738_Mice lacking leptin or the leptin receptor were protected from neointimal formation following vascular injury. 20086094_Results unmask distinct binary roles for leptin receptor Tyr985-mediated signaling in energy metabolism, acting as an age/diet-dependent regulatory switch to counteract age-associated or diet-induced obesity. 20150284_VIP knockout mouse taste cells show a significant decrease in leptin receptor expression and elevated expression of glucagon-like peptide 1, which may explain sweet taste preference of VIP knockout mice. 20151325_These results indicate that leucine affects leptin receptor expression in muscle cells via the mTOR signaling pathway. 20193700_This study provided evidence that Lepr in POMC neurons, although important for leptin's effect on the regulation of body fat accumulation and glucose homeostasis. 20223942_leptin-induced internalization of the ligand-receptor complex can occur without specific sorting signals in the cytoplasmic region of ObR 20506495_Knockout of leptin receptor in vitro Decreases Mineralization and Increases Adipogenesis of Primary Stromal Cells 20601111_results suggest that leptin induces lipid oxidation in skeletal muscle through the JAK2/p38 MAPK/STAT3 signaling pathway via not only Ob-R(L) but also Ob-R(S) 20691166_Co-localization of LepRb and TRHR1 is not observed on individual fibers in the hindbrain but these two fiber types co-mingle in vagal and reticular nuclei. 20833647_After MI, ObRKO mice displayed a loss of cardiac STAT3 and AMPK signalling. Worse survival and cardiac morbidity were also seen in the ObRKO mouse post-MI. 20851886_Lepr signaling is a negative modulator of bone mechanosensitivity. 21037323_Cut-like homeobox 1 (CUX1) regulates expression of the fat mass and obesity-associated and retinitis pigmentosa GTPase regulator-interacting protein-1-like (RPGRIP1L) genes and coordinates leptin receptor signaling. 21056997_Local leptin action within the mammary gland is a critical mediator, linking obesity and dysfunctional adipose tissue with aggressive tumor growth. 21067751_leptin receptor-mediated STAT3-independent signaling pathways confer protection against atherosclerosis 21084451_The specific ablation of the Lepr exon 17 gene in somatotropes resulted in GH deficiency with a consequential reduction in lipolytic activity normally maintained by growth hormone and increased adiposity. 21148797_The role of leptin receptor-->STAT3 signaling in the innate immune response against bacterial infection of the lung is compared to the responses of wild-type and mutant s/s mice in a murine model of pneumococcal pneumonia. 21160171_Leptin receptor-deficient mice exhibit reduced BDNF expression in the hippocampus in a glucocorticoid-dependent fashion, while hypothalamic BDNF expression is reduced via glucocorticoid-independent mechanisms. 21263121_selective loss of leptin receptor signaling emanating from tyrosine985 enhances the cardiovascular and renal sympathetic effects of leptin. 21285324_This study evaluated whether neuropeptide Y or agouti-related protein affect hyperphagia and energy metabolism in leptin receptor-deficient mice. 21289197_Data present strong evidence that LepRb RECPRb neurons in the dorsomedial hypothalamus/dorsal hypothalamic area and median preoptic area mediate thermoregulatory leptin action. 21311043_There is a critical role for ObR in the arcuate nucleus in mediating the sympathetic nerve responses to leptin and in the adverse sympathoexcitatory effects of leptin in obesity. 21328476_The bone phenotype of leptin receptor-deficient (db/db) and wild-type mice was compared using micro-computed tomographic (microCT) analysis of the proximal tibias and vertebrae. 21406387_leptin is important for CRC growth in obesity, and acts as a growth factor for CRC at stages subsequent to tumour initiation in colorectal carcinogenesis. 21483433_Lepr deficient mice display an anxiogenic-like phenotype in the elevated plus-maze, light-dark box, social interaction and novelty-suppressed feeding tests. Dopamine neurons in the ventral tegmental area revealed an increase in burst firing. 21521753_Lack of LepRb-Tyr985 signaling enhances insulin sensitivity partly through increased suppression of hepatic glucose production. 21558549_the Rho kinase-dependent component of myometriual contractions was greater in leptin receptor deficient db/+ mice, along with an increased expression of Rho kinase. 21606595_When Lepr was deleted from glucagon-like 1 peptide-expressing neurons in the nucleus tractus solitarius, mice became hyperphagic, gained weight, had a higher metabolic rate, but had normal glucose homeostasis. 21616110_The absence of leptin signaling at the blood brain barrier appears to enhance the availability of leptin to CNS parenchyma. 21636700_Using a transplantable tumor that develops spontaneously in the murine mammary tumor virus-Wnt-1 transgenic mice, we show that tumors transplanted into obese leptin receptor-deficient mice grow to eight times the volume of tumors in wild-type mice. 21674492_Data suggest that ghrelin and leptin act largely on distinct neuronal populations and that ghrelin receptor deficiency does not affect sensitivity to the anorexigenic and body weight-lowering actions of leptin/leptin receptors. 21726645_the level of the leptin receptor in the hypothalamus of metallothionein-3-null mice was significantly reduced 21859958_we studied the role of the leptin receptor in regulating distinct immune cells during chronic infection 22207628_Robust and selective expression of the receptor for the adipocyte-derived peptide hormone leptin (OB-R) is a characteristic feature of tumor-initiating stem cells. 22276206_Data show that over-expression of LepRb selectively in POMC neurons increases susceptibility to the development of -induced obesity. 22287654_CAPD-induced hyperleptinemia amplifies the inflammatory response of adipose tissue through short-form leptin receptor when the long-form isotype is defective. 22291999_In IUGR rats with no catch-up, leptin resistance was associated with low hypothalamic expression of LepRa and LepRb. 22323652_Endothelial leptin receptor mutation provides partial resistance to diet-induced obesity. 22326958_direct leptin action on POMC neurons does not reduce food intake, but is sufficient to normalize glucose and glucagon levels in mice otherwise lacking LEPR 22341640_Impairment of reproduction in intact db/db mice is not mediated by intraovary intact/defective leptin signaling even in face of a divergent modulation by gonadotropins. 22560222_Deletion of Foxo1 from leptin receptor neurons in Lepr(DeltaIrs2) mice normalized energy balance. 22684620_ObR increased along GFAP+ intermediate filaments in reactive astrocytes in the hippocampus & hypothalamus of mice with EAE. ObRa mRNA was elevated only after resolution of EAE symptoms; it was even decreased at the peak time of EAE symptoms. 22685316_Substitution of LepR Tyr985 with Leu985 abrogates LepR-mediated ERK MAP kinase 1/2 activation in mutant mouse lung following infection with Klebsiella pneumoniae. 22815920_significant changes in fasting-induced adipose factor (FIAF), resistin (Rstn), long-form leptin receptor (LepRb) and suppressor of cytokine-3 (SOCS3) gene expression following chronic SCI and with advanced age 22932068_Leptin receptors in the mouse hippocampus are essential for positive mood states and coping with stress. 23045458_Data indicate that obese insulin-resistant mice with leptin receptor/protein tyrosine phosphatase 1B mutations (KdbHPTP1B) not only develop hypertension but also present an increase in blood oressure variability. 23070544_TNF-alpha up-regulates protein level and cell surface expression of the leptin receptor by stimulating its export via a PKC-dependent mechanism 23115124_Leptin plays an important role in regulating cardiac function in the setting of cardiac stress caused by Cre-recombinase expression, likely through actions on cardiomyocyte energy metabolism. 23119079_Data indicate that insulin receptors in pro-opiomelanocortin neurons (IR/LepR(POMC) mice may serve as a new mouse model to clarify the involvement of adipose and liver tissue in the pathogenesis and etiology of polycystic ovary syndrome (PCOS). 23178499_SOCS3-deficient epithelial cells in the stomach induce activation of leptin receptor signaling via STAT3 and initiates hyperproliferation, mimics intestinal metaplasia and eventually promotes adenocarcinoma formation. 23251447_results suggested that some alleles in GPR37 were related to the deleterious effect of ASD. GPR37 is associated with the dopamine transporter to modulate dopamine uptake, and regulates behavioral responses to dopaminergic drugs 23329815_In response to a high-fat diet astrocytic leptin receptor KO mice had attenuated hyperleptinemia and sObR, a lower level of leptin mRNA in subcutaneous fat, and a paradoxical increase in UCP1 mRNA. 23359322_determined the kinetics of leptin transfer in groups of leptin receptor knockout and wildtype mice 23417423_Mice with mutant Lepr reduced the percentage of immunolabeled GH cells and serum GH. 23482448_extended perifornical area galanin-LepRb neurons galanin acts inhibitory rather than orexigenic 23916894_The worsening of experimental autoimmune encephalomyelitis in the astrocyte specific leptin receptor knockout mouse suggests that astrocytic leptin signaling helps to clear infiltrating leukocytes and reduce autoimmune destruction of the CNS. 23963458_Data indicate that conditional deletion of Pten (PtenDelta/Delta) activated adipocyte signaling in the respiratory epithelium that was associated with increased expression of LEP and its receptor. 23967295_leptin can act directly on its receptors in peripheral tissues to regulate cell proliferation and differentiation 24013029_Genistein stimulates fatty acid oxidation in a leptin receptor-independent manner through the JAK2-mediated phosphorylation and activation of AMPK in skeletal muscle. 24055508_40% of Lepr-deficient (C57BL/KsJ-db/db) mice developed liver tumors after administration of thioacetamide compared with none of the control mice. 24169048_Agrp(-/-) Lepr(db/db) females are fertile and sustain adequate nutrition of pups with lactation to weaning age. 24256641_results demonstrate that leptin receptor is dramatically induced in the granulosa cells of ovulating follicles and this induction of Lepr expression requires the transcription factor CCAAT/enhancer-binding protein beta 24343628_LEPR223Arg allele (rs1137101) was associated with higher daily energy intake at 4 years of age. 24343796_Obese diabetic mice lacking leptin receptor (db/db) are deficient in postnatal regenerative osteogenesis. 24424041_the role of leptin and LepRb signaling is extrinsic to the intestinal epithelium and independent of food intake in the control of the gut microbiome 24464605_Lepr(db/db) mice fed a diet high in unsaturated fat develop weight gain and NASH through adiponectin depletion, which is associated with adipose tissue inflammation and hepatic mitochondrial dysfunction. 24473435_Loss of leptin receptor signaling in close to one-half of alpha-cells does not alter glucose metabolism in vivo, nor is it sufficient to prevent the therapeutic action of leptin in type 1 diabetes. 24694605_data suggest that both adipose and placenta-derived leptin system are involved in the regulation of energy metabolism during fetal growth. 24743494_leptin binding to the Q223R leptin receptor 24784449_Results suggest that endospanin 1 (also named OB-RGRP) is implicated in obesity development and/or the installation of leptin resistance. 24928195_High LEPR expression is associated with onset of obesity. 24939734_Data (including data from studies in mutant/knockout mice) suggest that signal transduction via leptin/leptin receptor is involved in regulation of triglyceride accumulation in the myocardium as seen in obesity. 24953181_LepR(+) cells are the major source of bone and adipocytes in adult bone marrow 24958593_STAT3 is one of the important molecules downstream of LEPR, and LEPR/STAT3 signaling controls tumor cell proliferation. 25004093_Expression of Lepr in the liver, but not in the hypothalamus, was up-regulated in Helz2 knockout mice. 25057200_Arcuate nucleus neurons express a functional leptin receptor throughout early postnatal development. 25057790_Collectively, these studies highlight the importance of LEP/LEPR to gonadotropes with GnRH-binding sites and activin as potential targets. LEP/LEPR may modulate population growth, adjusting the number of offspring to the availability of food supplies. 25098352_Leptin receptor deficiency or antagonism profoundly affects metabolism, with little concomitant effects on immune functions. 25123537_A markedly elevated leptin production from inflamed visceral fat could deteriorate beta-cell function via leptin receptor-mediated oxidative stress and cyclooxygenase-2 activation in the development of obesity 25125486_Leptin knockouts are hyperphagic and obese, whereas insulin receptor knockouts are similar to controls but double knockouts exhibit higher body weight and adiposity solely due to reduced energy expenditure. 25232724_Global loss of the leptin receptor results in reduced viral clearance and worse outcomes following influenza A infection. 25313970_leptin of dermal white adipose tissue might regulate hair growth and, therefore, hair cycle progression via leptin receptor on the hair follicle epithelium 25353183_This study investigated the importance of fetal genotype (db/+) relative to abnormal maternal metabolism for placental function and therefore fetal growth and offspring health. 25383904_Hypoglycemia activates neurons of the parabrachial nucleus that express leptin receptors. 25516614_The newly recognized effect of the leptin receptor Q223R mutation on diminished neutrophil chemotaxis and the impact of this mutation on multiple infectious diseases suggest a broader impact of this mutation on susceptibility to disease. 25919692_Human and murine pancreatic cancer cell lines were found to express the short as well as the long form of the leptin receptor and functionally responded to leptin induced activation through an increased phosphorylation of AKT473. 26059465_absence of leptin receptor on thymic epithelial cells or T cells does not lead to the loss of thymic function, demonstrating the thymoprotective effect of leptin is mediated by obesity suppression rather than signalling to the thymus. 26168341_vital role in the optimization of somatotrope lipolytic function, especially in the young male 26284788_Diet- and Genetically-Induced Obesity Differentially Affect the Fecal Microbiome and Metabolome in Apc1638N Mice 26297880_Leptin receptor expressing neurons express phosphodiesterase-3B (PDE3B) and leptin induces STAT3 activation in PDE3B neurons in the mouse hypothalamus. 26529315_JAK-STAT and hormone biosynthesis pathways were involved in the follicular development and ovulation disorders caused by LepR deficiency in ovaries 26936780_These data show that leptin directly impairs vascular relaxation via a vascular skeletal muscle-LepR-mediated mechanism, suggesting a potential pathogenic role for leptin to increase cardiovascular risk during obesity. 26937712_we report that estradiol has minimal direct actions on LepRb cells in the mediodasal hypothalamus and that its anorexigenic effects can occur entirely independently of LepRb-STAT3 signaling in female mice. 26990203_This study reports about bone quality and bone turnover mechanisms in leptin receptor-deficient animals. 27053299_Leptin/LepR signaling regulates adipogenesis and osteogenesis by mesenchymal stromal cells in the bone marrow in response to diet and adiposity. 27101301_These mice showed an increase in FSH levels, but they remained in a prepubertal state. Together with previous findings, our data indicate that leptin-selective action in gonadotropes serves a role in adult reproductive physiology but is not sufficient to allow pubertal maturation in mice. 27147656_LepRb preoptic area neurons are stimulatory glutamatergic neurons, contrary to prevalent models 27283954_The leptin-receptor mice is not a proper model for secondary osteoporosis associated with type 2 diabetes mellitus. 27494408_Leptin expression is regulated by DNA methylation in adipocytes. 27613336_Data suggest that hypothalamic leptin/leptin receptor signaling plays a critical role in neurodevelopment, sympathetic activation, and prevention/development of hypertension. [REVIEW] 27644882_leptin/ObRb-mediated signals were enhanced in hypothalamus of GD3S KO mice due to increased a-series gangliosides, leading to the apparently similar features of energy expenditure between the KO and wild type mice 27693038_HFD-induced lipotoxicity and deregulated organelle biosynthesis confer cancer stem cell-like properties to the gastric mucosa via signaling pathway mediated by the leptin receptor, leptin, PI3K and beta-catenin 27889607_This study found that the expression of leptin and its receptor OB-R in mouse models of Sjogren's syndrome are elevated both locally and systemically during Sjogren's syndrome progression. 27941793_Attenuated leptin-receptor (LEPR) expression is essential for the development and maintenance of acute lymphoblastic leukemia (ALL), and that fasting inhibits ALL development by upregulation of LEPR and its downstream signaling through the protein PR/SET domain 1 (PRDM1). 28275162_Leptin receptors on agouti-related peptide neurons modulate the onset of puberty and adult fertility. 28323938_Neurotensin-leptin receptor neurons are important neuronal hubs within the lateral hypothalamic area for hormone-mediated control of ingestive and locomotor behaviors. 28481328_Identify Lepr(+) stromal lineage cells as the origin of myofibroblasts in primary myelofibrosis and suggest that targeting PDGFRA signalling could be an effective way to treat bone marrow fibrosis. 28580288_Knockin of Lepr in beta cells of Lepr null mice does not prevent hyperinsulinemia and hyperglycemia. 28705795_Suggest that intact endothelial leptin signaling limits neointima formation and that obesity represents a state of endothelial leptin resistance. 28893589_Study shows that compared to the other mouse lines, db/db mice with dysfunctional leptin receptors had a significantly longer tail flick latency after saline and buprenorphine. The results provide novel support for the interpretation that acute thermal nociception is associated with altered leptin signaling. 28938472_Data show that leptin receptor deficient (LeprNULL) mice exhibited increased body weight and food intake. 29174000_Deficiency of leptin receptor in myeloid cells disrupts hypothalamic metabolic circuits and causes hyperphagia with body weight gain. 29430464_Expression of IGF-1 and LEPR indicates a relevant role in androgenic features reversion present in PCOS, hormonal equilibrium, body weight regulation, and glucose metabolism, therefore, under phenotype obesity and infertility regulation in this model. 29535089_Data (including data from studies using mutant, transgenic, and knockout mice) suggest that gene targets of leptin/leptin-receptor (Lep/Lepr) signaling in hypothalamic neurons regulate energy metabolism; Lep/Lepr signaling appears to up-regulate expression of Atf3 (activating transcription factor-3) in hypothalamic neurons. 29565180_Ablation of the leptin receptor in myeloid cells impairs pulmonary clearance of Streptococcus pneumoniae and alveolar macrophage bactericidal function. 29728128_Abnormal sex hormone levels of Y123F mice were due to not only decreased gonadotropin levels in the central nervous system, but also ovarian hormone synthase abnormalities in the peripheral gonads. 29761783_The authors show that the preoptic area leptin-Lepr signaling modulates energy expenditure in response to internal energy state, and thus contributes to body weight homeostasis. 29929987_The dysfunction caused by LEPR deletion in somatotropes begins as early as neonatal development and involves developing GH and prolactin cells (somatolactotropes). 29948931_These lines of evidence suggest that downregulation of Ob-R and kisspeptin/GPR54 in the murine hypothalamus may contribute to male hypogonadism caused by high-fat diet-induced obesity. 30133304_LepR expression is required for the full anorexic effect of amylin; however, the n |
ENSG00000116678 |
LEPR |
141.342865 |
3.6407504182 |
1.864236 |
0.40283699 |
20.294625 |
0.00000663868230078119161779136303547232955679646693170070648193359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000508653967834502547073864675564180970468441955745220184326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
221.241514358191 |
62.5821172502569 |
60.7681079290015 |
13.0397882629603 |
| ENSMUSG00000057723 |
16671 |
Krt33b |
protein_coding |
Q61897 |
|
Coiled coil;Intermediate filament;Keratin;Reference proteome |
|
|
mmu:16671; |
cytoskeleton [GO:0005856]; intermediate filament [GO:0005882]; structural molecule activity [GO:0005198]; epithelial cell differentiation [GO:0030855]; intermediate filament organization [GO:0045109] |
|
ENSG00000006059 |
KRT33A |
10.483604 |
35.9871303992 |
5.169409 |
1.36182038 |
16.467188 |
0.00004949936313319576957183223986369569047383265569806098937988281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003181326157151215163615409764474861731287091970443725585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.5265920280392 |
20.203263787045 |
0.514811595771649 |
0.505164763665216 |
| ENSMUSG00000057766 |
225187 |
Ankrd29 |
protein_coding |
A0A3Q4EHJ7 |
|
ANK repeat;Proteomics identification;Reference proteome;Repeat |
|
|
|
Human_homologues |
|
ENSG00000154065 |
ANKRD29 |
370.531598 |
0.4591731623 |
-1.122890 |
0.13920422 |
64.384700 |
0.00000000000000102349885941428948787885110863439457871213041865315895861954231804702430963516235351562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000278391689760686731236716638896413660468956690480224480666038289200514554977416992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
231.079098290829 |
22.331206923791 |
503.250445091024 |
35.096761074322 |
| ENSMUSG00000057895 |
22646 |
Zfp105 |
protein_coding |
D3Z140 |
Human_homologues FUNCTION: May be involved in transcriptional regulation. Involved in cell differentiation and/or proliferation. |
Proteomics identification;Reference proteome |
|
|
|
nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II transcription regulatory region sequence-specific DNA binding [GO:0000977]; sequence-specific DNA binding [GO:0043565]; regulation of transcription by RNA polymerase II [GO:0006357] |
20186958_Results suggest that ZFP105 is a male germ-cell factor and plays a role in male reproduction. |
ENSG00000169981 |
ZNF35 |
9.191761 |
0.0099199123 |
-6.655457 |
1.52500327 |
19.550522 |
0.00000979739694063224732245576120703844935633242130279541015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000727694112830979316980037352458055011084070429205894470214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.186408529626749 |
0.235843597680341 |
18.7913485902164 |
10.356255216048 |
| ENSMUSG00000058153 |
56747 |
Sez6l |
protein_coding |
Q6P1D5 |
FUNCTION: Candidate tumor suppressor gene. May contribute to specialized endoplasmic reticulum functions in neurons. {ECO:0000250, ECO:0000269|PubMed:16814779}. |
Alternative splicing;Cell membrane;Disulfide bond;Endoplasmic reticulum;Glycoprotein;Membrane;Reference proteome;Repeat;Signal;Sushi;Transmembrane;Transmembrane helix |
|
|
mmu:56747; |
endoplasmic reticulum [GO:0005783]; endoplasmic reticulum membrane [GO:0005789]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; adult locomotory behavior [GO:0008344]; cerebellar Purkinje cell layer development [GO:0021680]; regulation of protein kinase C signaling [GO:0090036]; synapse maturation [GO:0060074] |
27716410_study demonstrates that SEZ6 and SEZ6L are physiological BACE1 substrates in the murine brain and suggests that sSEZ6 and sSEZ6L levels in CSF are suitable markers to monitor BACE1 inhibition in mice 34958451_The beta-Secretase Substrate Seizure 6-Like Protein (SEZ6L) Controls Motor Functions in Mice. |
ENSG00000100095 |
SEZ6L |
33.365640 |
2.4390599487 |
1.286325 |
0.48224318 |
6.822627 |
0.00900100785441683773346888131072773830965161323547363281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0306404029531394325736037842489167815074324607849121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
48.6199526921978 |
16.5563161911941 |
19.9338899879948 |
5.22697727135504 |
| ENSMUSG00000058183 |
27390 |
Mmel1 |
protein_coding |
B1AS18 |
Human_homologues FUNCTION: Metalloprotease involved in sperm function, possibly by modulating the processes of fertilization and early embryonic development. Degrades a broad variety of small peptides with a preference for peptides shorter than 3 kDa containing neutral bulky aliphatic or aromatic amino acid residues. Shares the same substrate specificity with MME and cleaves peptides at the same amide bond (By similarity). {ECO:0000250}. |
Reference proteome;Signal-anchor;Transmembrane |
|
|
|
membrane [GO:0016020]; metalloendopeptidase activity [GO:0004222]; proteolysis [GO:0006508] |
15121861_One of the roles of NL1 in mice is related to sperm function. NL1 modulates the processes of fertilization and early embryonic development in vivo. 16081046_Modification of the structure of the C-terminal region will either impair substrate hydrolysis or completely abolish the secretion and enzymatic activity of the secreted isoform of rat NEP2. 18425424_Intracellular transport and secretion of NEP2 is regulated by processes such as glycosylation, endoplasmic reticulum-Golgi transport, and intracellular calcium levels. 20941644_Data show that intranasal delivery of drugs can be used to model Alzheimer disease and suggest that other phosphoramidon-sensitive peptidases are degrading amyloid beta in neprilysin/NEP2-deficient mice. 21224067_In NEP/NEP2 double-knockout mice, Abeta levels were marginally increased ( approximately 1.5- to 2-fold), compared with NEP(-/-)/NEP2(+/+) controls. 25510509_This study demonstrated that Cortical fast-spiking parvalbumin interneurons enwrapped in the perineuronal net express the Neprilysin in mice. 25991605_neprilysin x ABCC1 double-deficient mice present a new model for early effects of amyloid-beta-related mild cognitive impairment that allows investigations without artificial overexpression of inherited Alzheimer's disease genes 27369050_These studies help to characterize our complex transgenic model of NEP overexpression and further demonstrate NEP's protective effects against plasma extravasation. 29191928_Sustained Rho activation in NEP(-/-) pulmonary artery smooth muscle cells is associated with a decrease in SM-protein levels and increased migration and proliferation. 33548010_Rho-kinase inhibitor hydroxyfasudil protects against HIV-1 Tat-induced dysfunction of tight junction and neprilysin/Abeta transfer receptor expression in mouse brain microvessels. |
ENSG00000142606 |
MMEL1 |
31.696388 |
26.9453579009 |
4.751965 |
0.81009579 |
35.324622 |
0.00000000279081482240882952715322723154581230353699083934770897030830383300781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000368687696226637397628365596952831895194435674056876450777053833007812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
60.1086198663561 |
35.5537638232217 |
2.23075975043491 |
1.14971887256013 |
| ENSMUSG00000058254 |
21912 |
Tspan7 |
protein_coding |
Q62283 |
FUNCTION: May be involved in cell proliferation and cell motility. |
Glycoprotein;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:21912; |
plasma membrane [GO:0005886]; regulation of postsynaptic neurotransmitter receptor internalization [GO:0099149] |
23945674_DC-STAMP, Siglec-15, Tspan7 and Mst1r expression was downregulated by docosahexaenoic acid, but not eicosapentaenoic acid during osteoclastogenesis in vitro. 27416754_Our results suggest that Tspan7 plays an important role in cytoskeletal organization required for the bone-resorbing function of osteoclasts by regulating signaling to Src, Pyk2, and microtubules. 27416759_Our results suggest that Tspan7 plays an important role in cytoskeletal organization required for the bone-resorbing function of osteoclasts by regulating signaling to Src, Pyk2, and microtubules. 34407208_Tetraspanin 7 regulates osteoclast function through association with the RANK/alphavbeta3 integrin complex. |
ENSG00000156298 |
TSPAN7 |
58.598014 |
0.0481372239 |
-4.376703 |
0.54971030 |
58.835375 |
0.00000000000001714312957263762569737787469837511588772733898966560417420623707585036754608154296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000004177986394406661540445923073830583884219307122531716913726995699107646942138671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.99906600481488 |
1.95122153335972 |
103.85031799152 |
25.3391760823346 |
| ENSMUSG00000058290 |
105988 |
Espl1 |
protein_coding |
P60330 |
FUNCTION: Caspase-like protease, which plays a central role in the chromosome segregation by cleaving the SCC1/RAD21 subunit of the cohesin complex at the onset of anaphase. During most of the cell cycle, it is inactivated by different mechanisms (By similarity). {ECO:0000250}. |
Autocatalytic cleavage;Chromosome partition;Cytoplasm;Direct protein sequencing;Hydrolase;Nucleus;Phosphoprotein;Protease;Reference proteome;Thiol protease |
|
|
mmu:105988; |
centrosome [GO:0005813]; cytoplasm [GO:0005737]; mitotic spindle [GO:0072686]; nucleus [GO:0005634]; cysteine-type endopeptidase activity [GO:0004197]; cysteine-type peptidase activity [GO:0008234]; peptidase activity [GO:0008233]; chromosome segregation [GO:0007059]; homologous chromosome segregation [GO:0045143]; meiosis I [GO:0007127]; meiotic chromosome separation [GO:0051307]; meiotic spindle organization [GO:0000212]; mitotic cell cycle [GO:0000278]; mitotic sister chromatid segregation [GO:0000070]; proteolysis [GO:0006508] |
16030258_Separase phosphorylation act redundantly to prevent sister chromatid separation. 16533945_In embryonic fibroblasts, Separase depletion blocks sister chromatid separation but does not prevent other aspects of mitosis, cytokinesis, or chromosome replication. 16839882_Proteolytic activity of separase is therefore essential for Rec8's removal from chromosome arms and for chiasma resolution but not for PBE. 18232736_inhibitory phosphorylation of separase plays a critical role in the maintenance of sister chromatid cohesion and genome stability in proliferating postmigratory primordial germ cells 18728194_These results collectively suggest that Separase is an oncogene, whose overexpression alone in mammary epithelial cells is sufficient to induce aneuploidy and tumorigenesis in a p53 mutant background. 18843049_Results describe a new function for the TGF-beta pathway in the regulation of APC to mediate chromatid separation during mitosis. 19124608_deregulation of separase may be a significant contributor to infertility in humans. 21494564_Separase phospho-regulation is critical for genome stability in oogenesis. 21799785_Separase act synergistically with loss of p53 in the initiation and progression of B- and T- cell lymphomas 21865557_With the continual loss of cohesins from chromosomes that occurs throughout the natural reproductive lifespan, tight regulation of separase in oocytes may be particularly important to maintain cohesion and prevent aneuploidy. 24276237_Separase-overexpressing mammary cells are not only susceptible to chromosomal missegregation-induced aneuploidy but also other genetic instabilities 26858254_Degradation of the Separase-cleaved Rec8, a Meiotic Cohesin Subunit, by the N-end Rule Pathway. 30305303_Thus, tethering of separase to DSBs and confined cohesin cleavage promote DSB repair in G2 cells. Importantly, this conserved interphase function of separase protects mammalian cells from oncogenic transformation. 31704793_results indicate that cyclin B2/CDK1 is also responsible for separase inhibition via inhibitory phosphorylation to regulate chromosome separation in oocyte meiosis, which may not occur in other cell types. 35712867_MicroRNA-202 safeguards meiotic progression by preventing premature SEPARASE-mediated REC8 cleavage. |
ENSG00000135476 |
ESPL1 |
954.930354 |
3.5491765456 |
1.827484 |
0.11094121 |
262.640450 |
0.00000000000000000000000000000000000000000000000000000000004560075438994052337836838112015332716974258659310516244578587810028903749087254508052031605729927983415669495987637160729121147230448365431733142791315138708796439459547400474548339843750000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000097829674770080855278110578673560324837329223025874555057733195920656837411528747749487805777279456140538104864928537477979256558326339682858836799272950202066567726433277130126953125000000000000000000000 |
Yes |
No |
1501.05355525006 |
115.54269124212 |
422.930089827888 |
25.3176723840592 |
| ENSMUSG00000058354 |
16687 |
Krt6a |
protein_coding |
P50446 |
FUNCTION: Epidermis-specific type I keratin involved in wound healing (PubMed:10866680). Involved in the activation of follicular keratinocytes after wounding, while it does not play a major role in keratinocyte proliferation or migration (PubMed:10866680). Participates in the regulation of epithelial migration by inhibiting the activity of SRC during wound repair (PubMed:22529101). {ECO:0000269|PubMed:10866680, ECO:0000269|PubMed:22529101}. |
Coiled coil;Intermediate filament;Keratin;Reference proteome |
Human_homologues NA; + ;NA; + ;NA |
|
mmu:16687; |
keratin filament [GO:0045095]; structural constituent of skin epidermis [GO:0030280]; antimicrobial humoral immune response mediated by antimicrobial peptide [GO:0061844]; defense response to Gram-positive bacterium [GO:0050830]; intermediate filament organization [GO:0045109]; keratinization [GO:0031424]; morphogenesis of an epithelium [GO:0002009]; Wnt signaling pathway [GO:0016055]; wound healing [GO:0042060] |
16790075_Although K6a was not essential for mammary gland development, loss of both K6a and K6b resulted in an increase in PR-positive mammary epithelial cells and decreased proliferation after exposure to steroid hormones 17170733_mK6a and mK75 thus exhibit temporally distinct, and spatially opposed, expression patterns in the companion layer (Cl) during postnatal anagen. 30389720_These findings reveal that K6a/K6b modulate keratinocyte migration by regulating cell-matrix and cell-cell adhesion and highlight a role for keratins in collective cell migration. |
ENSG00000185479+ENSG00000170465+ENSG00000205420 |
KRT6B+KRT6C+KRT6A |
27.095293 |
271.9276857754 |
8.087079 |
1.30491078 |
58.074308 |
0.00000000000002524012139377642561806405649387937204839826681179726364234738866798579692840576171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000006063998881230323532888420070442985209150862924154523625475121662020683288574218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
57.1004475669 |
58.9830016101241 |
0.209983942620886 |
0.206570060580343 |
| ENSMUSG00000058441 |
406218 |
Panx2 |
protein_coding |
Q6IMP4 |
FUNCTION: Structural component of the gap junctions and the hemichannels. |
Alternative splicing;Cell junction;Cell membrane;Gap junction;Glycoprotein;Ion channel;Ion transport;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:406218; |
cytoplasm [GO:0005737]; gap junction [GO:0005921]; plasma membrane [GO:0005886]; gap junction channel activity [GO:0005243]; protein-containing complex binding [GO:0044877]; structural molecule activity [GO:0005198]; wide pore channel activity [GO:0022829]; cation transport [GO:0006812]; cell-cell signaling [GO:0007267]; positive regulation of interleukin-1 production [GO:0032732]; response to ischemia [GO:0002931] |
16143426_Results reveal a widespread and similar mRNA distribution for both pannexin 1 and 2, but indicate that they are inversely regulated during the development of the rat brain. 16616526_Panx1 and Panx2 are abundantly expressed in the retina, and may therefore contribute to the electrical and metabolic coupling, or to signaling between retinal neurons 17149368_Despite similarity in detergent solubility of pannexins and connexins, the lack of spatial co-localization in the lens membranes suggested a distinct, non-redundant to connexin function for these proteins in the lens cell membrane. 18695503_Data show that Panx2 is expressed in the spiral and Scarpa's ganglion neurons. 19009624_Results show that pannexins 1, 2, and 3 are abundantly expressed in the mouse and rat cochlea and demonstrate distinct cellular distributions. 19692571_Panx2 strongly interacts with the core and high-mannose species of Panx1 but not with Panx3. The combination of Panx1 & Panx2 results in compromised channel function. 20529862_Pannexin 2 is expressed by postnatal hippocampal neural progenitors and modulates neuronal commitment 22147915_Px1 and Px2 underlie channel function in neurons and contribute to ischemic brain damage 22945868_In situ hybridization revealed that mRNAs for Panx1 and Panx2 were both expressed in the olfactory epithelium and olfactory bulb. 23111424_). Pannexin double-knockout mice (Px1(-/-) Px2(-/-)) were less impaired in parameters such as exploration, anxiety, sensorimotor function and behavioral symmetry. 23659289_It used overlap-extension cloning to create the first chimeric Panx, consisting of Panx2 with the Panx1 C-terminus (Panx2(Panx1CT)), to determine whether the Panx1 C-terminus influences the trafficking of Panx2. 28390953_Panx2 is an important regulator of the insulin secretory capacity and apoptosis in pancreatic beta-cells. 34985913_Pannexin 2 is expressed in murine skin and promotes UVB-induced apoptosis of keratinocytes. |
ENSG00000073150 |
PANX2 |
34.030202 |
2.8322174298 |
1.501932 |
0.52813744 |
7.871102 |
0.00502310229066161521155331470822602568659931421279907226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0186477928567774127455169974609816563315689563751220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
53.1662717418951 |
19.9005827657506 |
18.7719598017996 |
5.22103533528777 |
| ENSMUSG00000058914 |
81799 |
C1qtnf3 |
protein_coding |
Q9ES30 |
|
Collagen;Reference proteome;Secreted;Signal |
|
|
mmu:81799; |
collagen trimer [GO:0005581]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; protein-containing complex [GO:0032991]; identical protein binding [GO:0042802]; cellular triglyceride homeostasis [GO:0035356]; ERK1 and ERK2 cascade [GO:0070371]; glucose homeostasis [GO:0042593]; negative regulation of gene expression [GO:0010629]; negative regulation of gluconeogenesis [GO:0045721]; negative regulation of inflammatory response [GO:0050728]; negative regulation of interleukin-6 production [GO:0032715]; negative regulation of monocyte chemotactic protein-1 production [GO:0071638]; negative regulation of NIK/NF-kappaB signaling [GO:1901223]; positive regulation of adiponectin secretion [GO:0070165]; positive regulation of cytokine production [GO:0001819]; positive regulation of ERK1 and ERK2 cascade [GO:0070374]; positive regulation of protein kinase B signaling [GO:0051897]; protein kinase B signaling [GO:0043491] |
12850274_Genomic organization, chromosomal localization and adipocytic expression of the murine gene for CORS-26 15157741_the murine CORS-26 promoter is transcriptionally regulated by specificity protein-1, PPARgamma, and pituitary protein transcription factor-1 16155912_Cartducin is a novel growth factor and plays important roles in regulating both chondrogenesis and cartilage development by its direct stimulatory action on the proliferation of chondrogenic precursors and chondrocytes. 17299102_CORS-26 up-regulates adipokine secretion and might be involved in metabolic and immunologic pathways. 19424626_Elevated expression of CTRP3/cartducin contributes to promotion of osteosarcoma cell proliferation 19947921_Exogenous CTRP3/cartducin promoted the proliferation of p53LMAC01 cells in a dose-dependent manner via ERK1/2 (extracellular signal-regulated kinase 1/2)- and MAPK (p38 mitogen-activated protein kinase)-signalling pathways 20952387_This study provides the first functional evidence linking CTRP3 to hepatic glucose metabolism and establishes CTRP3 as a novel adipokine. 22644487_c-Jun is a cis-acting element for CTRP3 regulation in chondrogenic cells. 22653084_CTRP3 is a novel antiapoptotic, proangiogenic, and cardioprotective adipokine, the expression of which is significantly inhibited after myocardial infarction. 23744740_results establish a novel role for C1q tumor necrosis factor-related protein 3 (CTRP3) in regulating hepatic lipid metabolism and highlight its protective function and therapeutic potential in attenuating hepatic steatosis 24269820_These observations indicate that CTRP3 plays an important role in the development of autoimmune arthritis, suggesting CTRP3 as a possible medicine to treat rheumatoid arthritis. 24578384_CTRP3 promotes vascular calcification by enhancing phosphate-induced osteogenic transition of VSMC through reactive oxygen species-extracellular signal-regulated kinase 1/2-Runx2 pathway. 24996172_present study shows the proof of principle that the novel adipokine CTRP-3 is a potent inhibitor of LPS-induced systemic inflammation and LPS-induced signaling in adipose tissue in vivo 25168658_Data suggest that CTRP3 up-regulates expression and secretion of adipokines (adiponectin, leptin, visfatin, and apelin) in adipocytes; these responses to CTRP3 are down-regulated by insulin resistance or inhibition of AMPK signaling pathway. 26103094_Our study suggests that CTRP3 prevents osteoclast differentiation 26272338_CTRP3 was expressed in developing skeletal muscle tissues, and the expression level of CTRP3 was increased during myogenic differentiation of C2C12 cells. 26670485_Although it is dispensable for physiological control of energy balance, CTRP3 plays a previously unsuspected role in modulating liver size and circulating cytokine levels in response to obesity. 29061338_CTRP3 protected against doxorubicin-induced heart injury via activation of Sirt1. 29438034_Low C1QTNF3 expression is associated with polycystic ovarian syndrome. 29572383_CTRP3 expression protected against high-fat diet-induced spermatogenic deficiency through the SIRT1/endoplasmic reticulum stress pathway. 29655602_The cardioprotective effects of CTRP3 might be mediated by activating AMPKalpha signaling pathway and blunting inflammatory response and apoptosis. 30407523_CTRP3 promotes pressure overload-induced cardiac hypertrophy via activation of the TAK1-JNK axis. 30887395_CTRP3 can efficiently inhibit the inflammatory response and endothelial dysfunction induced by ox-LDL in mouse aortic endothelial cells, perhaps by activating the PI3K/Akt/eNOS pathwaf 31285424_CTRP3 protects the host against pathological cardiac remodeling and left ventricular dysfunction induced by pressure overload largely by inhibiting the p38/CREB pathway and alleviating p38-induced endoplasmic reticulum stress. 31615574_Study found lower levels of C1q/TNF-related protein 3 (CTRP3) in the spinal muscular atrophy muscle (SMA) secretome and confirmed that CTRP3 levels are indeed reduced in muscle tissues and serum of an SMA mouse model. 31629950_CTRP3 may be an innovative therapeutic target for treating patients with depression through regulating p38 and JNK signaling. 31808575_CTRP3 Regulates Endochondral Ossification and Bone Remodeling During Fracture Healing. 32219332_CTRP3 ameliorates cerulein-induced severe acute pancreatitis in mice via SIRT1/NF-kappaB/p53 axis. 33057992_Deficiency of C1q/TNF-related protein 3 (CTRP3) decreases adipose tissue weight in diet-induced obesity mice. 33991612_CTRP3 promotes TNF-alpha-induced apoptosis and barrier dysfunction in salivary epithelial cells. 34165676_CTRP-3 Regulates NOD1-mediated Inflammation and NOD1 Expression in Adipocytes and Adipose Tissue. 34388476_The adipokine C1q/TNF-related protein-3 (CTRP-3) inhibits Toll-like receptor (TLR)-induced expression of Cathelicidin antimicrobial peptide (CAMP) in adipocytes. 34440913_Anti-Inflammatory Effects of C1q/Tumor Necrosis Factor-Related Protein 3 (CTRP3) in Endothelial Cells. 34533812_C1q/tumor necrosis factor-related protein-3 acts as a target treating hepatic fibrosis. 34687744_Adipocyte-Derived CTRP3 Exhibits Anti-Inflammatory Effects via LAMP1-STAT3 Axis in Psoriasis. 34925309_The CTRP3-AdipoR2 Axis Regulates the Development of Experimental Autoimmune Encephalomyelitis by Suppressing Th17 Cell Differentiation. 34995284_Transgenic overexpression of CTRP3 does not prevent alcohol induced hepatic steatosis in female mice. 35278832_CTRP3 alleviates myocardial ischemia/reperfusion injury in mice through activating LAMP1/JIP2/JNK pathway. |
ENSG00000082196 |
C1QTNF3 |
528.925522 |
4.1400620408 |
2.049652 |
0.13021826 |
241.970313 |
0.00000000000000000000000000000000000000000000000000000146250249270254135912231444767601483816002976031457152799006047272515009693715763288981434888591575068744353686334917165094066095596032645725914633771935768891125917434692382812500000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000002931162890637514710481856250504939082026822561507225666375176672614089141912621570235856422248507187257113222088772233604368226884441528401481491528102196753025054931640625000000000000000000000000000000000000 |
Yes |
No |
842.19584049264 |
76.1202114947309 |
203.425898497169 |
14.667336526192 |
| ENSMUSG00000059022 |
333088 |
Kcp |
protein_coding |
Q3U492 |
FUNCTION: Enhances bone morphogenetic protein (BMP) signaling in a paracrine manner. In contrast, it inhibits both the activin-A and TGFB1-mediated signaling pathways. {ECO:0000269|PubMed:15793581}. |
Alternative splicing;Coiled coil;Disulfide bond;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:333088; |
extracellular region [GO:0005576]; extracellular space [GO:0005615]; hematopoietic progenitor cell differentiation [GO:0002244]; positive regulation of BMP signaling pathway [GO:0030513] |
15793581_The data indicate an important role for KCP in attenuating the pathology of renal fibrotic disease. 16738323_role for KCP in modulating the responses between these anti- and profibrotic cytokines in the initiation and progression of renal interstitial fibrosis 23539757_These data suggest that extracellular regulation of the TGF-beta/BMP signaling axis by KCP, and by extension possibly other cysteine-rich domain proteins, can attenuate both acute and chronic renal injury. 28424263_Metabolic profiling indicated increased energy expenditure in KCP-overexpressing mice and reduced expenditure in the KCP mutants with no effect on food intake or activity. These findings demonstrate that shifting the TGF-beta superfamily signaling with a secreted protein can alter the physiology and thermogenic properties of adipose tissue to reduce obesity even when mice are fed a high-fat diet. |
ENSG00000135253 |
KCP |
12.426446 |
43.6122571432 |
5.446662 |
1.34678083 |
18.935865 |
0.00001351871568795007248796943150059846061594726052135229110717773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000977763900089532268572120599081642922101309522986412048339843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.8099365526691 |
23.4486394623594 |
0.500087314468631 |
0.4874633198572 |
| ENSMUSG00000059049 |
329872 |
Frem1 |
protein_coding |
Q684R7 |
FUNCTION: Extracellular matrix protein that plays a role in epidermal differentiation and is required for epidermal adhesion during embryonic development. {ECO:0000269|PubMed:15345741}. |
Alternative splicing;Basement membrane;Calcium;Cell adhesion;Developmental protein;Disulfide bond;Extracellular matrix;Glycoprotein;Lectin;Metal-binding;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:329872; |
basement membrane [GO:0005604]; collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; membrane [GO:0016020]; carbohydrate binding [GO:0030246]; metal ion binding [GO:0046872]; anatomical structure morphogenesis [GO:0009653]; cell communication [GO:0007154]; cell-matrix adhesion [GO:0007160]; craniofacial suture morphogenesis [GO:0097094] |
15345741_Frem1 appears to act as a dermal mediator of basement membrane adhesion and is required for epidermal adhesion during embryonic development 15878328_QBRICK is an adhesive ligand of basement membrane distinctively recognized by cells in the embryonic skin and hair follicles through different types of integrins directed to the Arg-Gly-Asp motif. 16880404_QBRICK/Frem1, Fras1, and Frem2 interactions at the basement membrane have roles in preventing Fraser syndrome-like defects 17240369_Taken together, our findings indicate that besides a cooperative function with Fras1 in embryonic basement membranes, Frem1 can also act independently in processes related to epidermal differentiation. 17251066_Study reports a comparative analysis of the spatiotemporal localization of Fras1, Frem1, Frem2 and Frem3 proteins during mouse embryonic development. 17596926_in the absence of Fras1 the basement membrane localization of Frem3 remains unaffected in contrast to Frem1 and Frem2 which are completely abolished from the basement membrane. 18563433_The localization pattern of Fras1 and Frem2 was indistinguishable, while both proteins along with Frem3 could be detected even in the absence of Frem1. 19732862_The phenotypic variability reported for different Frem1 mouse mutants suggests that the apparently distinct phenotype of bifid nose and anorectal and renal anomalies syndrome in humans may represent a previously unrecognized variant of Fraser syndrome. 19940113_TILRR, an isoform encoded by an alternatively spliced FREM1 mRNA, is an IL-1RI co-receptor that associates with the signaling receptor complex to enhance recruitment of MyD88 and control Ras-dependent amplification of NF-kappaB and inflammatory responses. 21931569_Frem1 mutant mice is the first bona fide mouse model of human metopic craniosynostosis and a new model for midfacial hypoplasia. 22613833_the loss of QBRICK significantly diminished The expression of nephronectin, an integrin alpha8beta1 ligand necessary for renal development. 23221805_We conclude that FREM1 plays a critical role in the development of the diaphragm and that FREM1 deficiency can cause CDH in both humans and mice. 23536828_FREM1-deficient mice faithfully recapitulate many of the phenotypes seen in individuals with FREM1 deficiency and that variations in GATA4 and SLIT3 expression modulate some FREM1-related phenotypes in mice. 24046351_loss of FREM1 function promotes epidermal blistering in Fraser syndrome as a consequence of reduced PDGFC activity, in addition to its stabilising role in the basement membrane |
ENSG00000164946 |
FREM1 |
73.293110 |
0.0115578171 |
-6.434987 |
0.68661363 |
129.846603 |
0.00000000000000000000000000000442701027080501199772035320305778085063057483938211765850668132302145667494961550625376389689336065202951431274414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000003371611022245096868414515849083928278484165806009523602908395655322113226284516662722978708188747987151145935058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.76070934575731 |
0.869958271691196 |
152.339263587104 |
30.2778612993461 |
| ENSMUSG00000059173 |
18573 |
Pde1a |
protein_coding |
Q61481 |
FUNCTION: Calcium/calmodulin-dependent cyclic nucleotide phosphodiesterase with a dual specificity for the second messengers cGMP and cAMP, which are key regulators of many important physiological processes. Has a higher efficiency with cGMP compared to cAMP. {ECO:0000269|PubMed:15901640}. |
Alternative splicing;Calmodulin-binding;cAMP;Cell projection;cGMP;Cilium;Flagellum;Hydrolase;Magnesium;Metal-binding;Reference proteome;Zinc |
|
|
mmu:18573; |
cytoplasm [GO:0005737]; motile cilium [GO:0031514]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; sperm flagellum [GO:0036126]; 3',5'-cyclic-AMP phosphodiesterase activity [GO:0004115]; 3',5'-cyclic-GMP phosphodiesterase activity [GO:0047555]; 3',5'-cyclic-nucleotide phosphodiesterase activity [GO:0004114]; calmodulin binding [GO:0005516]; calmodulin-activated 3',5'-cyclic-GMP phosphodiesterase activity [GO:0048101]; calmodulin-activated dual specificity 3',5'-cyclic-GMP, 3',5'-cyclic-AMP phosphodiesterase activity [GO:0004117]; metal ion binding [GO:0046872]; cGMP catabolic process [GO:0046069]; regulation of smooth muscle cell apoptotic process [GO:0034391]; regulation of smooth muscle cell proliferation [GO:0048660]; signal transduction [GO:0007165] |
15901640_new variant PDE1A_v7 is the major form of cyclic nucleotide phosphodiesterase 1A expressed in mature sperm and is therefore likely to play an important role in cyclic nucleotide regulation of mature sperm function. 16514069_PDE1A is important in VSMC growth and survival and may contribute to the neointima formation in atherosclerosis and restenosis. 18335582_These results prove that the kinetic properties of PDE isoforms play a major role in determining intracellular cAMP signals in response to physiological elevation of [Ca2+]i. 19797176_a novel role for Ca(2+)/CaM-stimulated PDE1, particularly PDE1A, in regulating pathological cardiomyocyte hypertrophy via a cGMP/PKG-dependent mechanism, thereby demonstrating Ca(2+) and cGMP signaling cross-talk during cardiac hypertrophy. 22012077_These results suggest that induction of PDE1A plays a critical role in cardiac fibroblast activation and cardiac fibrosis 28750036_These results support an important role of PDE1A in the renal pathogenesis of autosomal dominant polycystic kidney disease and in the regulation of blood pressure. 31131329_PDE1 inhibition induces PKA- and PKG-mediated promotion of proteasomal degradation of misfolded proteins and treats heart failure with preserved ejection fraction caused by CryAB(R120G) in a mouse model of cardiac proteinopathy |
ENSG00000115252 |
PDE1A |
40.404462 |
5.9589897755 |
2.575068 |
0.44917404 |
32.563600 |
0.00000001153550796065662343367082655690158965455793804721906781196594238281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001383534309108044891061461371256635644044763466808944940567016601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
70.3669683713568 |
22.2639798483213 |
11.8085398738464 |
3.04958074736619 |
| ENSMUSG00000059187 |
320265 |
Tafa1 |
protein_coding |
Q7TPG8 |
FUNCTION: Regulatory factor which is ligand for CMKLR2 and is involved in the modulation of neural stem-cell proliferation and differentiation. {ECO:0000269|PubMed:29799787}. |
Reference proteome;Secreted;Signal |
|
This gene is a member of the TAFA family which is composed of five highly homologous genes that encode small secreted proteins. These proteins contain conserved cysteine residues at fixed positions, and are distantly related to MIP-1alpha, a member of the CC-chemokine family. The TAFA proteins are predominantly expressed in specific regions of the brain, and are postulated to function as brain-specific chemokines or neurokines that act as regulators of immune and nervous cells. [provided by RefSeq, Jul 2008]. |
mmu:320265; |
endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; receptor ligand activity [GO:0048018]; neuroblast differentiation [GO:0014016]; regulation of neuroblast proliferation [GO:1902692]; regulation of signaling receptor activity [GO:0010469] |
31689372_In male mice, FAM19A1 deficiency alters food intake patterns during the light and dark cycle. Fam19a1 KO mice are hyperactive, and locomotor hyperactivity is more pronounced in female KO mice. 32706374_Deorphanizing FAM19A proteins as pan-neurexin ligands with an unusual biosynthetic binding mechanism. 34440636_Alterations in Dendritic Spine Maturation and Neurite Development Mediated by FAM19A1. |
ENSG00000183662 |
TAFA1 |
8.360164 |
0.0211398940 |
-5.563888 |
1.50775077 |
15.696672 |
0.00007435486971947680265478120009703388859634287655353546142578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004548487452076589200589129546870026388205587863922119140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.390753859213107 |
0.486880183952366 |
18.4841920210915 |
9.86496774880893 |
| ENSMUSG00000059325 |
74318 |
Hopx |
protein_coding |
Q8R1H0 |
FUNCTION: Atypical homeodomain protein which does not bind DNA and is required to modulate cardiac growth and development. Acts via its interaction with SRF, thereby modulating the expression of SRF-dependent cardiac-specific genes and cardiac development. Prevents SRF-dependent transcription either by inhibiting SRF binding to DNA or by recruiting histone deacetylase (HDAC) proteins that prevent transcription by SRF. Overexpression causes cardiac hypertrophy (PubMed:12297045, PubMed:12297046). Acts as a co-chaperone for HSPA1A and HSPA1B chaperone proteins and assists in chaperone-mediated protein refolding (By similarity). {ECO:0000250|UniProtKB:Q9BPY8, ECO:0000269|PubMed:12297045, ECO:0000269|PubMed:12297046}. |
3D-structure;Cytoplasm;Developmental protein;Homeobox;Nucleus;Reference proteome;Repressor;Transcription;Transcription regulation |
|
|
mmu:74318; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; DNA binding [GO:0003677]; chaperone-mediated protein complex assembly [GO:0051131]; heart development [GO:0007507]; histone deacetylation [GO:0016575]; lung alveolus development [GO:0048286]; negative regulation of cell differentiation [GO:0045596]; negative regulation of transcription by RNA polymerase II [GO:0000122]; positive regulation of gap junction assembly [GO:1903598]; positive regulation of skeletal muscle tissue regeneration [GO:0043415]; positive regulation of striated muscle cell differentiation [GO:0051155]; regulation of heart contraction [GO:0008016]; regulation of protein binding [GO:0043393]; regulation of transcription by RNA polymerase II [GO:0006357]; trophectodermal cell differentiation [GO:0001829] |
12297045_Inactivation of Hop in mice by homologous recombination results in a partially penetrant embryonic lethal phenotype with severe developmental cardiac defects involving the myocardium. 12297046_HOP is highly expressed in the developing heart, dependent on the cardiac-restricted homeodomain protein Nkx2.5. Mice homozygous for a HOP null allele segregate into two phenotypic classes characterized by an excess or deficiency of cardiac myocytes. 12617835_Expressed in embryonic myocardium and other mesoderm, but not in endocardium or great vessels. 12975471_data show that Hop can inhibit serum response factor-dependent transcriptional activation by recruiting histone deacetylase (HDAC) activity and can form a complex that includes HDAC2 15790958_Hop functions in the adult cardiac conduction system and demonstrate conservation of molecular hierarchies between embryonic myocardium and the specialized conduction tissue of the mature heart. 15967424_Hop is regulated in the forebrain by a so far unidentified paracrine signaling factor from the roof plate 17277742_Expression pattern of Hop provides first evidence that new transcription is initiated in lens fiber cells after they detach from the capsule. Hop may be first of class of genes with this pattern of expression. 17576768_HOP/NECC1 (-/-) placenta exhibited marked propagation of giant cell layers and, in turn reduction of spongiotrophoblast formation 18507846_HOP participates in the regulation of the adult mouse hippocampal stem cell niche by negatively affecting cell survival 20802482_Hopx is required for the function of regulatory T cells induced by DCs and the promotion of DC-mediated T cell unresponsiveness in vivo. 20833366_Hdac2, Hopx, and Gata4 coordinately regulate cardiac myocyte proliferation during embryonic development. 21061432_Hopx regulates the expression of genes involved in regulation of apoptosis and survival and makes them refractory to Fas-induced apoptosis. 21382376_HOPX/Hopx expression is reduced in multiple examples of human and murine cardiac hypertrophy and failure. 23487314_Hopx expression defines a subset of multipotent hair follicle stem cells and a progenitor population primed to give rise to K6+ niche cells. 25865356_Type I alveolar cells expressing Hopx manifest plasticity to regenerate type II alveolar cells in the lung. 26050929_Analysis of the hematopoietic stem/progenitor cell pool in Hopx-/- mice demonstrated significantly reduced cell frequencies and impaired engraftment in competitive repopulation assays, thus providing functional validation of this positional candidate gene 26113728_Hopx integrates Bmp and Wnt signaling by physically interacting with activated Smads and repressing Wnt genes. 26170384_Peripherally Induced Tolerance Depends on Peripheral Regulatory T Cells That Require Hopx To Inhibit Intrinsic IL-2 Expression. 30154568_HOPX expression in cultured ATII cells increased over culture time. HOPX expression was increased in AECs from bleomycin-instilled mouse lungs in vivo. Small interfering RNA-based knockdown of Hopx resulted in suppressing ATII-ATI trans-differentiation and activating cellular proliferation in vitro. 30174314_These results support an early priming of neural stem cells (NSCs) for the genesis of defined cell types depending on their spatial location in the subventricular zone and identify HOPX as a marker of a subpopulation primed toward astrocytic fates. 30266827_Hopx is required and sufficient, respectively, for basal radial glial cells abundance as found in the developing gyrencephalic neocortex. 32533098_Knock-out of Hopx disrupts stemness and quiescence of hematopoietic stem cells in mice. |
ENSG00000171476 |
HOPX |
282.499904 |
0.4756474115 |
-1.072036 |
0.15118864 |
50.039290 |
0.00000000000150698079059797128648445903969152188765778799250938391196541488170623779296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000293533649646909212192967797541752819623361858702992321923375129699707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
186.225435266237 |
19.5514174529379 |
391.519917411613 |
29.4236212151365 |
| ENSMUSG00000059326 |
12982 |
Csf2ra |
protein_coding |
Q00941 |
FUNCTION: Low affinity receptor for granulocyte-macrophage colony-stimulating factor. Transduces a signal that results in the proliferation, differentiation, and functional activation of hematopoietic cells. |
Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:12982; |
external side of plasma membrane [GO:0009897]; granulocyte macrophage colony-stimulating factor receptor complex [GO:0030526]; receptor complex [GO:0043235]; cytokine binding [GO:0019955]; cytokine receptor activity [GO:0004896]; cytokine-mediated signaling pathway [GO:0019221]; granulocyte-macrophage colony-stimulating factor signaling pathway [GO:0038157]; positive regulation of leukocyte proliferation [GO:0070665]; protein phosphorylation [GO:0006468]; receptor signaling pathway via JAK-STAT [GO:0007259] |
17694571_ability to detect GM-CSF receptor expression in experimental studies should greatly facilitate the analysis of its role in immune pathologies[GM-CSF receptor alpha] 22103458_GM-CSFRalpha levels peaked between E13 and E15 in the brain. 22223820_Loss of the CSF2RA gene may be a critical mutation explaining the high incidence of loss of sex chromosomes associated with the t(8;21)(q22;q22) translocation. 27468760_a STOP mutation in the GM-CSFRalpha chain, leading to a complete and specific deficiency in GM-CSF signaling, is reported. 28641926_GM-CSF Ralpha blockade in relapsing-remitting experimental autoimmune encephalomyelitis prevented disease relapses and inhibited T cell responses specific for both the inducing and spread myelin peptides 30154795_we have demonstrated that CFP1 plays a crucial role in GM-CSF-derived macrophages, in which it positively regulates phagocytic and bactericidal activity mainly through promoting Csf2ralpha expression and the subsequent activation of downstream pathways. 30942918_These data show that loss of membrane GM-CSFRalpha following GM-CSF exposure does not preclude sustained GM-CSF/GM-CSFRalpha signaling and that this receptor plays a key role in ligand clearance. Hence neutrophilic activation via GM-CSFR may play an important role in neutrophilic lung inflammation even in the absence of high GM-CSF levels or GM-CSFRalpha expression. 35043685_A murine model of hereditary pulmonary alveolar proteinosis caused by homozygous Csf2ra gene disruption. |
ENSG00000198223 |
CSF2RA |
327.206884 |
0.3817248817 |
-1.389395 |
0.26870664 |
26.002813 |
0.00000034092022045778590676239289472548854575961740920320153236389160156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000032639200490339380550158712018804507692948391195386648178100585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
190.000629535094 |
35.5931943574611 |
497.742323446349 |
68.4281211815973 |
| ENSMUSG00000059751 |
668144 |
Rps3a3 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
64.781561 |
0.2539955799 |
-1.977125 |
0.35367120 |
30.925222 |
0.00000002681639638019780965919244277665539621935408831632230430841445922851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003043720936387280510908152388049874303987962775863707065582275390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.9453591716055 |
6.17669539757537 |
98.2117845504984 |
16.8026885245188 |
| ENSMUSG00000060180 |
544791 |
Myh13 |
protein_coding |
M0QWL0 |
Human_homologues FUNCTION: Fast twitching myosin mediating the high-velocity and low-tension contractions of specific striated muscles. {ECO:0000269|PubMed:23908353}. |
Coiled coil;Reference proteome |
|
|
|
myofibril [GO:0030016]; myosin filament [GO:0032982]; myosin II complex [GO:0016460]; actin filament binding [GO:0051015]; microfilament motor activity [GO:0000146]; cellular response to starvation [GO:0009267]; muscle contraction [GO:0006936] |
Human_homologues 12110653_phylogenic imlications in extraocular muscles (REVIEW) 18830724_Meta-analysis of gene-disease association. (HuGE Navigator) 19240061_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 21426302_Domain-mapping experiments indicated that muscle-type creatine kinase binds to the C-terminal domains of MyBPC1, which is also the binding site of myosin. |
ENSG00000006788 |
MYH13 |
12.597739 |
0.0894915583 |
-3.482105 |
1.12459611 |
8.295253 |
0.00397488408410527280234525449031934840604662895202636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0152392233498846001171767028381509589962661266326904296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.24303636557404 |
1.93100148911184 |
25.0642229053198 |
14.2955703535437 |
| ENSMUSG00000060487 |
320825 |
Samd5 |
protein_coding |
A0A0R4J186 |
|
Reference proteome |
|
|
mmu:320825; |
cytoplasm [GO:0005737] |
|
ENSG00000203727 |
SAMD5 |
97.486379 |
0.3000174492 |
-1.736882 |
0.29447775 |
34.299947 |
0.00000000472394598422901991007668534745811417252525643561966717243194580078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000603650547246446493781919209915820534462227442418225109577178955078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.8432884961863 |
9.01140682666524 |
146.135795158197 |
21.1278020810946 |
| ENSMUSG00000060988 |
271786 |
Galnt13 |
protein_coding |
Q8CF93 |
FUNCTION: Catalyzes the initial reaction in O-linked oligosaccharide biosynthesis, the transfer of an N-acetyl-D-galactosamine (GalNAc) residue from UDP-GalNAc to a serine or threonine residue on the protein receptor (PubMed:12407114, PubMed:8618846, PubMed:27629416). Generates GalNAc-O-Ser/Thr structure also known as Tn antigen, which itself is immunogenic but also serves as a precursor for the synthesis of different mucin-type O-glycan core structures (PubMed:12407114). Contributes to the synthesis of O-linked glycans on mucins and proteoglycans of the central nervous system (PubMed:12407114, PubMed:27629416). Can glycosylate both unmodified peptides and glycopeptides that already contain an O-linked GalNAc sugar. Transfers GalNAc to Thr-/Ser-rich tandem repeats GTTPSPVPTTSTTSAP of MUC5AC. Transfers GalNAc to three consecutive serine/threonine residues on SDC3 forming a triplet-Tn epitope expressed in Purkinje cells of the developing brain (By similarity). May promote neurogenesis through glycosylation and stabilization of PDPN (PubMed:27629416). {ECO:0000250|UniProtKB:Q8IUC8, ECO:0000269|PubMed:12407114, ECO:0000269|PubMed:27629416, ECO:0000269|PubMed:8618846}. |
Alternative splicing;Disulfide bond;Glycoprotein;Glycosyltransferase;Golgi apparatus;Lectin;Manganese;Membrane;Metal-binding;Reference proteome;Signal-anchor;Transferase;Transmembrane;Transmembrane helix |
PATHWAY: Protein modification; protein glycosylation. {ECO:0000305|PubMed:12407114}. |
|
mmu:271786; |
Golgi apparatus [GO:0005794]; Golgi membrane [GO:0000139]; carbohydrate binding [GO:0030246]; metal ion binding [GO:0046872]; polypeptide N-acetylgalactosaminyltransferase activity [GO:0004653]; protein O-linked glycosylation [GO:0006493]; protein O-linked glycosylation via serine [GO:0018242]; protein O-linked glycosylation via threonine [GO:0018243] |
12407114_cloning and characterization of pp-GalNAc-T13; expressed in all neuroblastoma cells examined and primary cultured neurons but not in glioblastoma cells and primary cultured astrocytes 22306014_These data suggested that high expression of pp-GalNAc-T13 gene generated trimeric Tn antigen on Syndecan-1, leading to the enhanced metastasis. 23814067_high expression of the ppGalNAc-T13 gene generates tTn antigen on Syndecan 1, leading to enhanced invasion and metastasis via the formation of a complex consisting of integrin alpha5beta1, Syndecan 1, and MMP-9 in the glycolipid-enriched microdomain/rafts. 27629416_This study suggests that ppGalNAc-T13 contributes to neuronal differentiation through glycosylating and stabilizing PDPN, which provides insights into the regulatory roles of O-glycosylation in mammalian neural development. |
ENSG00000144278 |
GALNT13 |
64.582289 |
0.4328593374 |
-1.208030 |
0.36411658 |
10.789207 |
0.00102093626604252444775577934876764629734680056571960449218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0047066892265011294957388621185145893832668662071228027343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
40.7744004649738 |
10.4005050644829 |
94.1978073267823 |
16.9449439140941 |
| ENSMUSG00000061048 |
12560 |
Cdh3 |
protein_coding |
P10287 |
FUNCTION: Cadherins are calcium-dependent cell adhesion proteins. They preferentially interact with themselves in a homophilic manner in connecting cells; cadherins may thus contribute to the sorting of heterogeneous cell types. |
3D-structure;Calcium;Cell adhesion;Cell membrane;Cleavage on pair of basic residues;Glycoprotein;Membrane;Metal-binding;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
This gene encodes a calcium-dependent cell-cell adhesion protein containing five cadherin domains. The encoded protein plays a role in epithelial outgrowth, such as that which occurs during the development of hair follicles and limb buds. Loss of function of the related gene in humans results in ectodermal dysplasia, ectrodactyly, and macular dystrophy and congential hypotrichosis with juvenile macular dystrophy. This gene is located in the vicinity of similar cadherin genes on chromosome 8. The proprotein is further cleaved into a functional chain. Alternative splicing results in multiple transcript variants. [provided by RefSeq, Mar 2013]. |
mmu:12560; |
adherens junction [GO:0005912]; catenin complex [GO:0016342]; cell junction [GO:0030054]; cytoplasm [GO:0005737]; plasma membrane [GO:0005886]; cadherin binding [GO:0045296]; calcium ion binding [GO:0005509]; cell-cell adhesion [GO:0098609]; cell-cell adhesion via plasma-membrane adhesion molecules [GO:0098742]; hair cycle process [GO:0022405]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156]; keratinization [GO:0031424]; negative regulation of timing of catagen [GO:0051796]; negative regulation of transforming growth factor beta2 production [GO:0032912]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; positive regulation of gene expression [GO:0010628]; positive regulation of insulin-like growth factor receptor signaling pathway [GO:0043568]; positive regulation of keratinocyte proliferation [GO:0010838]; positive regulation of melanin biosynthetic process [GO:0048023]; positive regulation of melanosome transport [GO:1902910]; positive regulation of tyrosinase activity [GO:0032773]; regulation of hair cycle by canonical Wnt signaling pathway [GO:0060901]; response to xenobiotic stimulus [GO:0009410]; retina homeostasis [GO:0001895]; wound healing [GO:0042060] |
14997319_nuclear localization of beta-catenin in the enamel knot suggests a specific activation of Wnt signaling; upregulation of P-cadherin was observed in this area; these observations suggest a linkage between cell adhesion and Wnt signaling in the enamel knot 16919267_Truncated P-cadherin promotes melanoma cell invasion and migration and therefore has an important role in the progression of malignant melanoma. 17292359_in skin and mammary, activation of beta-catenin signalling correlates with up-regulation of P-cadherin expression 18199584_P-cadherin is a newly defined transcriptional target gene of p63, with a crucial role in hair follicle morphogenesis as well as the apical ectodermal ridge during limb bud outgrowth in humans whereas it is not required for either in mice. 20212509_UVB exposure resulted in the overexpression of the cytokines, tumour necrosis factor (TNF)-alpha, interleukin (IL)-6 and the adhesion molecule P-cadherin. 21445019_Loss of P-cadherin is associated with melanoma. 22710718_P-cadherin is a direct PAX3-FOXO1A transcriptional target involved in Alveolar rhabdomyosarcoma aggressiveness. 23277553_importance of cadherin-based cell-cell adhesions in coordinating mechanical activity of epithelial cells 26783302_role of P-cadherin through beta-PIX-mediated Cdc42 activation in the regulation of cell polarity 28494434_P-Cadherin is not required for maintenance of retinal stem cells in vivo. 29338041_P-cadherin (CDH3) is highly dominant in both mouse and human retinal pigment epithelium in situ. The degree of dominance of P-cadherin is surprisingly large, with mouse Cdh3 and human CDH3 accounting for 82-85% and 92-93% of the total of the three cadherin mRNAs, respectively. 36468795_The transcription factors Junb and Fosl2 cooperate to regulate Cdh3 expression in 15P-1 Sertoli cells. |
ENSG00000062038 |
CDH3 |
16.372967 |
0.1756029633 |
-2.509611 |
0.70644210 |
12.622381 |
0.00038115542574306969687836232907329758745618164539337158203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0019640593521374960159220357525100553175434470176696777343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.22078826534614 |
2.65984896662714 |
29.7306387496304 |
9.75326300799674 |
| ENSMUSG00000061186 |
353282 |
Sfmbt2 |
protein_coding |
Q3UH63 |
Human_homologues FUNCTION: Transcriptional repressor of HOXB13 gene. {ECO:0000269|PubMed:23385818}. |
Nucleus;Proteomics identification;Reference proteome;Repressor |
|
|
mmu:353282; |
aggresome [GO:0016235]; cytosol [GO:0005829]; nuclear speck [GO:0016607]; histone binding [GO:0042393]; transcription corepressor activity [GO:0003714]; negative regulation of gene expression [GO:0010629] |
21605348_a large miRNA gene cluster in intron 10 of the Sfmbt2 gene 21911408_Data show that the expression of Sfmbt2 miRNAs is dependent on Dicer. 24154523_loss of SFMBT2 results in defects in maintenance of trophoblast cell types necessary for development of the extra-embryonic tissues, the placenta in particular. 24389345_Data indicate that in transgenic mice overexpressing Sfmbt2 10th intron-hosted miR-466a-3p, significant down-regulation of Nfat5 and many other osmoregulation-related genes was observed in both the renal cortex and medulla. |
ENSG00000198879 |
SFMBT2 |
168.498110 |
0.3952280880 |
-1.339243 |
0.20185863 |
43.661892 |
0.00000000003902982901921436977225904812979523795385405904312392522115260362625122070312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000006505287180329283263597454774705957508684761592121503781527280807495117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
91.6786274510095 |
12.8693610720941 |
231.963846293302 |
23.210427200693 |
| ENSMUSG00000061232 |
14972 |
H2-K1 |
protein_coding |
P01901 |
FUNCTION: Involved in the presentation of foreign antigens to the immune system. |
3D-structure;Direct protein sequencing;Disulfide bond;Glycoprotein;Immunity;Membrane;MHC I;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:14972; |
cell surface [GO:0009986]; cis-Golgi network membrane [GO:0033106]; early endosome [GO:0005769]; early endosome membrane [GO:0031901]; endoplasmic reticulum [GO:0005783]; endoplasmic reticulum exit site [GO:0070971]; external side of plasma membrane [GO:0009897]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; Golgi medial cisterna [GO:0005797]; Golgi membrane [GO:0000139]; lumenal side of endoplasmic reticulum membrane [GO:0098553]; lysosomal membrane [GO:0005765]; MHC class I peptide loading complex [GO:0042824]; MHC class I protein complex [GO:0042612]; MHC class Ib protein complex [GO:0032398]; phagocytic vesicle membrane [GO:0030670]; plasma membrane [GO:0005886]; 14-3-3 protein binding [GO:0071889]; beta-2-microglobulin binding [GO:0030881]; CD8 receptor binding [GO:0042610]; chaperone binding [GO:0051087]; identical protein binding [GO:0042802]; MHC class I protein binding [GO:0042288]; natural killer cell lectin-like receptor binding [GO:0046703]; peptide antigen binding [GO:0042605]; peptide binding [GO:0042277]; protein homodimerization activity [GO:0042803]; protein-containing complex binding [GO:0044877]; signaling receptor binding [GO:0005102]; T cell receptor binding [GO:0042608]; TAP binding [GO:0046977]; TAP complex binding [GO:0062061]; TAP1 binding [GO:0046978]; TAP2 binding [GO:0046979]; antigen processing and presentation of endogenous peptide antigen via MHC class I via ER pathway, TAP-dependent [GO:0002485]; antigen processing and presentation of endogenous peptide antigen via MHC class Ib [GO:0002476]; antigen processing and presentation of exogenous peptide antigen via MHC class I [GO:0042590]; defense response to bacterium [GO:0042742]; inner ear development [GO:0048839]; negative regulation of neuron projection development [GO:0010977]; positive regulation of T cell mediated cytotoxicity [GO:0001916]; T cell mediated cytotoxicity [GO:0001913] |
11943852_Analysis of structure and function relationships of an autoantigenic peptide of insulin bound to H-2K(d) that stimulates CD8 T cells in insulin-dependent diabetes mellitus. 12006653_intramembrane mechanism by which unassembled H2Kb molecules are retained in the endoplasmic reticulum of fibroblasts 12184911_The H2-K(b) gene belongs to target genes of the NF-IL6 (C/EBPbeta) in the course of the cellular response to TNF-alpha. Some consequences of this are discussed in a general framework of inducible expression of the H2-K(b) gene. 12459592_linked mhc polymorphism-driven diversification of the cytotoxic T lymphocyte repertoire to the generation of high-avidity, protective antiviral T cells and to superior antiviral defense 15272181_crystal structure complexed with an HBV-core nonapeptide 16148097_signaling through the mouse NK inhibitory receptor Ly49C requires the presence of H-2Kb and this signaling is prevented when H-2Kb is ablated by pulsing with a peptide that can bind to it with high affinity 16148128_two H-2K(k)-peptide complexes provide insights into the mechanisms through which MHC polymorphism outside primary peptide pockets influences the conformation of the bound peptides and have implications for TCR recognition 16148132_Linkage analysis performed in F(2) backcross ([BALB/c x C3H/HeN] x BALB/c) mice carrying the H2(k) region confirmed the presence of a susceptibility locus within the H2 region on proximal chromosome 17. 16473882_analysis of K(d)-restricted antigen presentation and recognition events 17240452_These results demonstrate that Ly49Q efficiently binds H-2K(b) ligand. 17418792_Study presents the 2.35 A structure of the 2C TCR complexed with its foreign ligand H-2L(d)-QL9; surprisingly, TCR utilizes a different strategy to engage the foreign pMHC in comparison to the manner in which it recognizes a self ligand H-2K(b)-dEV8. 18292501_Naive polyclonal class Ib-restricted CD8-positive T cells have the ability to homeostatically expand in the periphery in a largely class Ib-dependent manner. 18316618_the mHpa398 and mHpa519 peptides are novel H-2K(b)-restricted CTL epitopes capable of inducing heparanase-specific CTLs in vitro and in vivo 18418877_Data show that, despite the absence of H2-D1 and H2-K1 moecules, adult Purkinje cell in these transgenic mice are monoinnervated as in wild-type animals. 18453559_an unsuspected role for the MHC-I-Ly49 interaction in the development and function of the brain. 18641335_data suggest that APLP2 modulates the stability and endocytosis of K(d) molecules 19195028_Ethanol-elicited reduction in proteasome activity contributes to the suppression of SIINFEKL-H2Kb presentation on the surface of liver cells. 19346486_These physiological and behavioral phenotypes in K(b)D(b-/-) mice reveal a surprising role for classical MHCI molecules in synaptic plasticity and motor learning. 19454713_the MHC class I D locus prompts expansion and activation of Ly49G2(+) NK cells that are needed in H-2(k) MCMV resistance. 19687800_showed that a large disulfide-bonded complex was present in the mouse cells that included ERp57, tapasin, and K(d). 19700752_comparison of the consequences of MHC overexpression in muscle at different ages;a markedly more severe disease phenotype in young mice, with rapid onset of muscle weakness and pathology 19820922_Data provide biological confirmation that the chromosome 6 QTL provided MCMV control independent of H-2(k) via NK cells. 19864563_results show that neuronal MHC class Ia (H2-K(b)/D(b)) molecules are present in motor axons, are important for organization of neuromuscular junctions and motor recovery after nerve lesion, and that Schwann cells may mediate their actions 19945389_H2-Kb and H2-Db ligands, signaling via neuronal class I histocompatibility antigen receptors, may enable activity-dependent remodeling of brain circuits during developmental critical periods. 19950185_Data show that At early stages, tumors demonstrated reduced MHC class I expression and increased expression of natural killer group 2D ligands (NKG2D-L). 20124105_Data imply that a cognate linkage between TLR3 and MHC class I is required for efficient CTL priming to HSV-1. 20173027_Endoplasmic reticulum aminopeptidase (ERAAP)-deficiency changes the antigen processing mechanism and causes a marked increase in the length of peptides normally presented by the histocompatibility class I-peptide binding groove. 20363967_The interaction of SHIP, 2B4 activating receptor, and the major histocompatibility complex influence the regulation of the natural killer (NK) cell receptor repertoire, cytolytic function, and interferon-gamma production. 21098224_Major histocompatibility class Ib-restricted CD8-positive T cells, in addition to H2-M3-restricted T cells, contribute to antilisterial immunity and may contribute to immune responses against other intracellular bacteria. 21178009_Adult mice exposed to a noninherited maternal antigen accepts permanently H2-Kb-positive heart allografts despite the presence of normal levels of anti-K(b) T cell receptor transgenic T cells. 21690324_Analysis of CD8-positive T lymphocytes responsive to an H-2Kb-restricted immunodominant peptide displays T cell-homing patterns that support surveillance of both upper- and lower respiratory tract tissues by clonally related cells. 22084437_Mice lacking conventional class Ia and class II MHC molecules H2-Kb, H-2Db, and CIITA mediate partial but incomplete virus clearance during acute lymphocytic choriomeningitis virus infection. Clearance is incomplete and chronic infection follows. 23142461_tissue-specific h-2K(d) transgenic mice may be valuable tools for determining the mode of induction of CD8(+) T-cell responses. 25316337_Major histocompatibility class I molecules H2-Kb and H2-Db proteins regulate sculpting of local cortical circuits and in their absence, the excess connectivity can function as a substrate for cortical plasticity throughout life. 25782992_Because all forms of H-2K(b) are internalized but little beta2m-receptive heavy chain is present at the cell surface, it is likely that beta2m dissociation and recognition of the heavy chain for lysosomal degradation take place in an endocytic compartment. 26714929_H-2Kb in an unfolded form to a post-endoplasmic reticulum compartment from where they can cycle back to the endoplasmic reticulum. |
|
|
758.339121 |
0.4112864417 |
-1.281785 |
0.09932684 |
165.006780 |
0.00000000000000000000000000000000000009116515853305479046011153069786937597764533766589396413265051780144567410185229984897165036002641622103759289075242122635245323181152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000094464469032346302889830422006401985599890646163168438390968327242221394130642532949121997916906712244866639593965373933315277099609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
430.261910490841 |
29.6461308692004 |
1046.13686925026 |
51.7383196966408 |
| ENSMUSG00000061414 |
381812 |
Cracr2a |
protein_coding |
Q3UP38 |
FUNCTION: [Isoform 1]: Ca(2+)-binding protein that plays a key role in store-operated Ca(2+) entry (SOCE) in T-cells by regulating CRAC channel activation. Acts as a cytoplasmic calcium-sensor that facilitates the clustering of ORAI1 and STIM1 at the junctional regions between the plasma membrane and the endoplasmic reticulum upon low Ca(2+) concentration. It thereby regulates CRAC channel activation, including translocation and clustering of ORAI1 and STIM1. Upon increase of cytoplasmic Ca(2+) resulting from opening of CRAC channels, dissociates from ORAI1 and STIM1, thereby destabilizing the ORAI1-STIM1 complex (By similarity). {ECO:0000250|UniProtKB:Q9BSW2}.; FUNCTION: [Isoform 2]: Rab GTPase that mediates the trafficking of Weibel-Palade bodies (WPBs) to microtubule organizing center (MTOC) in endothelial cells in response to acute inflammatory stimuli (By similarity). During histamine (but not thrombin) stimulation of endothelial cells, the dynein-bound form induces retrograde transport of a subset of WPBs along microtubules to the MTOC in a Ca(2+)-independent manner and its GTPase activity is essential for this function (By similarity). Ca(2+)-regulated dynein adapter protein that activates dynein-mediated transport and dynein-dynactin motility on microtubules and regulates endosomal trafficking of CD47 (By similarity). Acts as an intracellular signaling module bridging two important T-cell receptor (TCR) signaling pathways, Ca(2+)-NFAT and JNK, to affect T-cell activation (PubMed:27016526). In resting T-cells, is predominantly localized near TGN network in a GTP-bound form, upon TCR stimulation, localizes at the immunological synapse via interaction with VAV1 to activate downstream Ca(2+)-NFAT and JNK signaling pathways (By similarity). Plays a role in T-helper 1 (Th1) cell differentiation and T-helper 17 (Th17) cell effector function (PubMed:29987160). Plays a role in store-operated Ca(2+) entry (SOCE) in T-cells by regulating CRAC channel activation (By similarity). {ECO:0000250|UniProtKB:Q9BSW2, ECO:0000269|PubMed:27016526, ECO:0000269|PubMed:29987160}. |
Adaptive immunity;Alternative splicing;Calcium;Calcium transport;Cell membrane;Coiled coil;Cytoplasm;Cytoskeleton;Golgi apparatus;GTP-binding;Immunity;Ion transport;Lipoprotein;Membrane;Metal-binding;Nucleotide-binding;Prenylation;Reference proteome;Repeat;Transport |
|
|
mmu:381812; |
cytoplasm [GO:0005737]; Golgi membrane [GO:0000139]; immunological synapse [GO:0001772]; microtubule organizing center [GO:0005815]; plasma membrane [GO:0005886]; trans-Golgi network membrane [GO:0032588]; vesicle [GO:0031982]; Weibel-Palade body [GO:0033093]; calcium ion binding [GO:0005509]; GTP binding [GO:0005525]; GTPase activity [GO:0003924]; activation of store-operated calcium channel activity [GO:0032237]; endosomal transport [GO:0016197]; positive regulation of calcium ion transport [GO:0051928]; positive regulation of JNK cascade [GO:0046330]; response to histamine [GO:0034776]; store-operated calcium entry [GO:0002115]; T-helper 1 cell differentiation [GO:0045063] |
27016526_GTP binding and prenylation of CRACR2A were associated with its localization near the Golgi and its stability 29987160_this study demonstrates the requirement of CRACR2A-mediated TCR signaling in Th1 responses as well as pathogenic conversion of Th17 cells, which occurs at the site of inflammation |
ENSG00000130038 |
CRACR2A |
837.073563 |
3.0048819572 |
1.587308 |
0.09656581 |
267.345426 |
0.00000000000000000000000000000000000000000000000000000000000430003942384993513469274406562954072594905380308232028643758847257678037590755531645964486237728911004057071721368179486095865399333185428492722302473358353402943521359702572226524353027343750000000000000 |
0.0000000000000000000000000000000000000000000000000000000009775850821504808189893594409604547694838919461350105575058301793766002990523166345190832336642025155096140503110924228457651183568300091957902885758668887206113140564411878585815429687500000000000000000 |
Yes |
Yes |
1218.5853564215 |
81.6260976450752 |
405.535183665643 |
21.4679233907032 |
| ENSMUSG00000061461 |
66278 |
Smim20 |
protein_coding |
D3Z7Q2 |
FUNCTION: [Small integral membrane protein 20]: Component of the MITRAC (mitochondrial translation regulation assembly intermediate of cytochrome c oxidase complex) complex, that regulates cytochrome c oxidase assembly (By similarity). Promotes the progression of complex assembly after the association of MT-CO1/COX1 with COX4I1 and COX6C (By similarity). Chaperone-like assembly factor required to stabilize newly synthesized MT-CO1/COX1 and to prevent its premature turnover (By similarity). {ECO:0000250|UniProtKB:Q8N5G0}.; FUNCTION: [Phoenixin-14]: Peptide involved in a broad spectrum of regulatory functions (PubMed:25687846, PubMed:26505917, PubMed:27268078). Is a ligand for GPR173 (By similarity). As part of the reproductive cycle, it regulates gonadotropin-releasing hormone (GnRH) signaling in the hypothalamus and pituitary gland which augments the release of luteinizing hormone (PubMed:27268078). More specifically, it regulates the expression of transcription factors CEBPB and POU2F1/OCT1 through the cAMP-PKA signaling pathway, which subsequently regulate the expression of GNRHR and KISS1 (PubMed:27268078). Plays a protective role in memory retention through activation of GNRHR (PubMed:26505917). Regulates the secretion of AVP by hypothalamic neurons (By similarity). Plays a role in the transduction of the itch sensation (PubMed:26415767). Induces anxiolytic effects, reducing behavior associated with anxiety (PubMed:25687846). Regulates food intake as well as satiation and satiety by increasing Nucb2 expression in neurons (By similarity). In the ovary, it regulates follicular growth by stimulating granulosa cell proliferation by increasing the expression of GPR173, CREB1, CYP19A1, KITLG, FSHR, and LHCGR (PubMed:30933929). It also increases the production of estradiol (E2) (PubMed:30933929). In the heart, it regulates contractility and relaxation by activating the AKT1-NOS3 and MAPK1-MAPK3 signaling pathways (By similarity). It also plays a cardioprotective role during ischemia, where it activates the SAFE and RISK pathways (By similarity). Stimulates the proliferation and differentiation of preadipocytes (PubMed:30251651). In pancreatic islet cells, it induces proliferation of islet cells as well as the production of INS through activation of the MAPK1-MAPK3 signaling pathways (PubMed:31422055). {ECO:0000250|UniProtKB:C0HLM6, ECO:0000269|PubMed:25687846, ECO:0000269|PubMed:26415767, ECO:0000269|PubMed:26505917, ECO:0000269|PubMed:27268078, ECO:0000269|PubMed:30251651, ECO:0000269|PubMed:30933929, ECO:0000269|PubMed:31422055}.; FUNCTION: [Phoenixin-20]: Peptide involved in a broad spectrum of regulatory functions (PubMed:25687846, PubMed:26505917, PubMed:27268078). Is a ligand for GPR173 (By similarity). As part of the reproductive cycle, it regulates gonadotropin-releasing hormone (GnRH) signaling in the hypothalamus and pituitary gland which augments the release of luteinizing hormone (PubMed:27268078). More specifically, it regulates the expression of transcription factors CEBPB and POU2F1/OCT1 through the cAMP-PKA signaling pathway, which subsequently regulate the expression of GNRHR and KISS1 (PubMed:27268078). Plays a protective role in memory retention through activation of GNRHR (PubMed:26505917). Regulates the secretion of AVP by hypothalamic neurons (By similarity). Plays a role in the transduction of the itch sensation (PubMed:26415767). Induces anxiolytic effects, reducing behavior associated with anxiety (PubMed:25687846). Regulates food intake as well as satiation and satiety by increasing Nucb2 expression in neurons (By similarity). In the ovary, it regulates follicular growth by stimulating granulosa cell proliferation by increasing the expression of GPR173, CREB1, CYP19A1, KITLG, FSHR, and LHCGR (PubMed:30933929). It also increases the production of estradiol (E2) (PubMed:30933929). In the heart, it regulates contractility and relaxation by activating the AKT1-NOS3 and MAPK1-MAPK3 signaling pathways (By similarity). It also plays a cardioprotective role during ischemia, where it activates the SAFE and RISK pathways (By similarity). Stimulates the proliferation and differentiation of preadipocytes (PubMed:30251651). In pancreatic islet cells, it induces proliferation of islet cells as well as the production of INS through activation of the MAPK1-MAPK3 signaling pathways (PubMed:31422055). {ECO:0000250|UniProtKB:C0HLM6, ECO:0000269|PubMed:25687846, ECO:0000269|PubMed:26415767, ECO:0000269|PubMed:26505917, ECO:0000269|PubMed:27268078, ECO:0000269|PubMed:30251651, ECO:0000269|PubMed:30933929, ECO:0000269|PubMed:31422055}. |
Amidation;Membrane;Mitochondrion;Mitochondrion inner membrane;Reference proteome;Secreted;Transmembrane;Transmembrane helix |
|
|
mmu:66278; |
extracellular region [GO:0005576]; mitochondrial inner membrane [GO:0005743]; mitochondrial cytochrome c oxidase assembly [GO:0033617] |
Human_homologues 26321642_MITRAC7 affects the biogenesis pathway by stabilizing newly synthesized COX1 in assembly intermediates, concomitantly preventing turnover. 26321642_SMIM20 (MITRAC7) is a COX1 specific chaperone which is necessary for cytochrome c oxidase biogenesis. |
ENSG00000250317 |
SMIM20 |
15.418761 |
0.0058959441 |
-7.406061 |
1.39835332 |
33.203717 |
0.00000000829917360636956648170800213007783363572400503471726551651954650878906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000001016999295029937451124109662990713243146956301643513143062591552734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.188734871905408 |
0.212953659551253 |
32.0109672494848 |
13.5337852841249 |
| ENSMUSG00000061462 |
380698 |
Obscn |
protein_coding |
A2AAJ9 |
FUNCTION: Structural component of striated muscles which plays a role in myofibrillogenesis. Probably involved in the assembly of myosin into sarcomeric A bands in striated muscle (By similarity). Has serine/threonine protein kinase activity and phosphorylates N-cadherin CDH2 and sodium/potassium-transporting ATPase subunit ATP1B1 (PubMed:23392350). Binds (via the PH domain) strongly to phosphatidylinositol 3,4-bisphosphate (PtdIns(3,4)P2) and phosphatidylinositol 4,5-bisphosphate (PtdIns(4,5)P2), and to a lesser extent to phosphatidylinositol 3-phosphate (PtdIns(3)P), phosphatidylinositol 4-phosphate (PtdIns(4)P), phosphatidylinositol 5-phosphate (PtdIns(5)P) and phosphatidylinositol 3,4,5-trisphosphate (PtdIns(3,4,5)P3) (By similarity). {ECO:0000250|UniProtKB:Q5VST9, ECO:0000269|PubMed:23392350}.; FUNCTION: Isoform 2 and isoform 3: bind phosphatidylinositol bisphosphates (PIP2s) via their PH domains and negatively regulate the PI3K/AKT/mTOR signaling pathway, thus contributing to the regulation of cardiomyocyte size and adhesion. {ECO:0000269|PubMed:28826662}. |
Alternative splicing;ATP-binding;Calmodulin-binding;Cell membrane;Cytoplasm;Developmental protein;Differentiation;Disulfide bond;Glycoprotein;Immunoglobulin domain;Kinase;Lipid-binding;Magnesium;Membrane;Metal-binding;Muscle protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome;Repeat;Secreted;Serine/threonine-protein kinase;SH3 domain;Transferase |
|
The obscurin gene spans more than 150 kb, contains over 80 exons and encodes a protein of approximately 800 kDa. The encoded protein contains 68 Ig domains, 2 fibronectin domains, 1 calcium/calmodulin-binding domain, 1 RhoGEF domain with an associated PH domain, and 2 serine-threonine kinase domains. This protein is one of three giant sacromeric signaling proteins that includes titin and nebulin. It may have a role in the organization of myofibrils during assembly and also may mediate interactions between the sarcoplasmic reticulum and myofibrils. Alternatively spliced transcript variants encoding different isoforms have been described although the full-length nature is not known for all splicing variants. [provided by RefSeq, Jan 2010]. |
|
cytosol [GO:0005829]; extracellular space [GO:0005615]; intercalated disc [GO:0014704]; M band [GO:0031430]; nuclear body [GO:0016604]; plasma membrane [GO:0005886]; sarcolemma [GO:0042383]; sarcomere [GO:0030017]; striated muscle myosin thick filament [GO:0005863]; Z disc [GO:0030018]; ankyrin binding [GO:0030506]; ATP binding [GO:0005524]; cadherin binding [GO:0045296]; calmodulin binding [GO:0005516]; guanyl-nucleotide exchange factor activity [GO:0005085]; metal ion binding [GO:0046872]; phosphatidylinositol bisphosphate binding [GO:1902936]; phosphatidylinositol-3,4,5-trisphosphate binding [GO:0005547]; phosphatidylinositol-3,4-bisphosphate binding [GO:0043325]; phosphatidylinositol-3-phosphate binding [GO:0032266]; phosphatidylinositol-4,5-bisphosphate binding [GO:0005546]; phosphatidylinositol-4-phosphate binding [GO:0070273]; phosphatidylinositol-5-phosphate binding [GO:0010314]; protein kinase activity [GO:0004672]; protein serine kinase activity [GO:0106310]; protein serine/threonine kinase activity [GO:0004674]; titin binding [GO:0031432]; cell differentiation [GO:0030154]; negative regulation of phosphatidylinositol 3-kinase signaling [GO:0014067]; protein autophosphorylation [GO:0046777]; protein phosphorylation [GO:0006468] |
16962094_Results support a role of obscurin in mediating the subcellular localization of small ankyrin1 isoforms in striated muscle cells. 18579686_the Rho-GEF domain of obscurin interacts with RanBP9 and that both can interact with the N-terminal region of titin to influence the formation of the Z-disk and A/I junction 18782775_ANK2 is subject to alternative splicing that gives rise to unique polypeptides with diverse roles in cardiac function. 19002483_the organization of obscurin at different locations in the sarcomere changes during muscle development and that this might affect the interaction with ank1.5. 23392350_the obscurin kinase domains, SK1 and SK2, are active enzymes with distinct substrate specificities. 23420875_Obscurin, by targeting ankB at the M band, contributes to the organization of subsarcolemma microtubules, localization of dystrophin at costameres, and maintenance of sarcolemmal integrity. 24516603_obscurins are not restricted to striated muscles, but are abundantly expressed in several tissues and organs including brain, skin, kidney, liver, spleen, and lung. 27784675_Altogether, these results suggest that obscurin is required for the maintenance of morphological and ultrastructural integrity of skeletal muscle fibers against damage induced by intense mechanical stress and point to the diaphragm as the skeletal muscle most severely affected in obscurin-deficient mice. 28826662_These findings implicate small obscurins in the maintenance of cardiomyocyte size and coupling. 31098411_Skeletal muscle-specific Obsl1 knockouts show a benign phenotype similar to obscurin knockouts. Only deletion of both proteins and removal of their functional redundancy revealed their roles for sarcolemmal stability and sarcoplasmic reticulum organization. 32910221_Deletion of obscurin immunoglobulin domains Ig58/59 leads to age-dependent cardiac remodeling and arrhythmia. 35163243_Impaired Intracellular Ca(2+) Dynamics, M-Band and Sarcomere Fragility in Skeletal Muscles of Obscurin KO Mice. |
ENSG00000154358 |
OBSCN |
101.988322 |
0.1545853921 |
-2.693524 |
0.43776820 |
34.714958 |
0.00000000381687524671001431987630664278629555319355404208181425929069519042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000494376222431011369120962240745337235026113376079592853784561157226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.6833609907587 |
7.91344831743471 |
166.143518774637 |
37.028432079982 |
| ENSMUSG00000061533 |
75216 |
Cep128 |
protein_coding |
Q8BI22 |
|
Alternative splicing;Coiled coil;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:75216; |
centriolar subdistal appendage [GO:0120103]; centriole [GO:0005814]; centrosome [GO:0005813]; cytoplasm [GO:0005737]; spindle pole [GO:0000922]; protein localization [GO:0008104] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20966902_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 29514088_CEP128 loss affects TGF-beta1-induced phosphorylation of multiple proteins that regulate cilium-associated vesicle trafficking. 30393005_This study highlight the crucial role of CEP128 in the pathogenesis of Grave's disease. 30623524_Cep128 deletion decreased the stability of centriolar microtubules. Cep128 associates with Odf2. 30939216_Overexpression of circCEP128 promoted bladder cancer progression through modulating miR-145-5p and MYD88 via MAKP signaling pathway. 35391656_Downregulation of Circ-CEP128 Enhances the Paclitaxel Sensitivity of Cervical Cancer Through Regulating miR-432-5p/MCL1. |
ENSG00000100629 |
CEP128 |
252.497334 |
2.1026334485 |
1.072197 |
0.19879941 |
28.603597 |
0.00000008881704910555741750471567784452897598157505854032933712005615234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000009434179163011509415870978027818249955771534587256610393524169921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
347.496929682615 |
48.0358032748611 |
165.267479182979 |
17.3854216435904 |
| ENSMUSG00000061535 |
109323 |
C1qtnf7 |
protein_coding |
Q8BVD7 |
|
Collagen;Reference proteome;Secreted;Signal |
|
|
mmu:109323; |
collagen trimer [GO:0005581]; extracellular space [GO:0005615]; identical protein binding [GO:0042802] |
28223291_In obese male mice consuming a high-fat diet, CTRP7 deficiency attenuated insulin resistance and enhanced glucose tolerance, independent of body weight, metabolic rate, and physical activity level. Improved glucose metabolism in CTRP7-deficient mice was associated with reduced adipose tissue inflammation, as well as decreased liver fibrosis and cellular oxidative and endoplasmic reticulum stress. |
ENSG00000163145 |
C1QTNF7 |
51.050084 |
0.0936038068 |
-3.417289 |
0.64018648 |
25.476647 |
0.00000044777025533400146655821317739909481758786569116637110710144042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000042101460057083402526941807997307165578604326583445072174072265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.79349349033729 |
3.57292941304436 |
83.2604330796593 |
25.7461763085771 |
| ENSMUSG00000061718 |
19049 |
Ppp1r1b |
protein_coding |
Q60829 |
FUNCTION: Inhibitor of protein-phosphatase 1. |
Acetylation;Alternative splicing;Cytoplasm;Phosphoprotein;Protein phosphatase inhibitor;Reference proteome |
|
This gene encodes a bifunctional signal transduction molecule. Dopaminergic and glutamatergic receptor stimulation regulates its phosphorylation and function as a kinase or phosphatase inhibitor. As a target for dopamine, this gene may serve as a therapeutic target for neurologic and psychiatric disorders in humans. Two transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Sep 2015]. |
mmu:19049; |
cytoplasm [GO:0005737]; dendritic spine head [GO:0044327]; dendritic spine neck [GO:0044326]; glutamatergic synapse [GO:0098978]; neuronal cell body [GO:0043025]; nucleus [GO:0005634]; postsynapse [GO:0098794]; D1 dopamine receptor binding [GO:0031748]; D2 dopamine receptor binding [GO:0031749]; D3 dopamine receptor binding [GO:0031750]; D4 dopamine receptor binding [GO:0031751]; D5 dopamine receptor binding [GO:0031752]; protein phosphatase inhibitor activity [GO:0004864]; protein serine/threonine phosphatase inhibitor activity [GO:0004865]; behavioral response to cocaine [GO:0048148]; cellular response to cocaine [GO:0071314]; DNA-templated transcription [GO:0006351]; intracellular signal transduction [GO:0035556]; locomotory behavior [GO:0007626]; memory [GO:0007613]; negative regulation of female receptivity [GO:0007621]; negative regulation of phosphoprotein phosphatase activity [GO:0032515]; response to amphetamine [GO:0001975]; response to cocaine [GO:0042220]; response to morphine [GO:0043278]; response to nicotine [GO:0035094]; visual learning [GO:0008542] |
11880652_DARPP-32 is essential not only for dopaminergic but also for serotonergic neurotransmission. 12181566_stimulatory effect of caffeine on motor activity in mice was greatly reduced following genetic deletion of DARPP-32 (dopamine- and cyclic AMP-regulated phosphoprotein of relative molecular mass 32,000) 12538871_adenosine A(2A)DARPP-32 signaling is stimulated by mGlu receptors by stimulating the adenosine A(2A) receptor-mediated formation of cAMP in an extracellular signal-regulated kinase-dependent manner 14631045_data demonstrate that the effects of psychotomimetics on sensorimotor gating and repetitive movements depend on DARPP-32; they regulate, through distinct mechanisms, phosphorylation of DARPP-32 as well as downstream effectors in the cortex and striatum 15608059_Blockade of the ERK pathway or mutation of DARPP-32 altered locomotor sensitization induced by a single injection of psychostimulants, demonstrating the functional relevance of this regulation. 15737638_These data indicate a potential role for DARPP-32 and inhibitor-1 in the regulation of PP1 activity in pancreatic beta-cell stimulus-secretion coupling. 16123776_these findings highlight distinct roles of the Thr-34 and Thr-75 phosphorylation sites of DARPP-32 in mediating short- and long-term behavioral and biochemical actions of cocaine 16162925_Psychomotor effect of cannabinoids can be accounted for by striatal projection neurons involving protein kinase A-dependent phosphorylation of DARPP-32. 16189209_DARPP-32 plays a key role for development of D1R motor stimulatory effects in mice 16971527_The results indicate that DARPP-32 is involved in the retinal pathway transmitting photic information that resets the circadian clock. 17010100_Data suggest a role for PDE1B in locomotor responses to psychostimulants through modulation of DARPP32-dependent pathways; however, this modulation does not necessarily impact other behaviors, such as anxiety or learning. 17209049_multiple signal transduction pathways mediating expression of DARPP-32 in vitro, including a novel, important pathway via by which PI3K regulates the contribution of cdk5/p35. 17552992_These data indicate that DARPP-32 participates in dopamine-mediated modifications of retinal function. 17596448_The abnormal involuntary movements developed in response to chronic L-DOPA are due to hyperactivation in striatal medium spiny neurons of a signaling pathway including sequential phosphorylation of DARPP-32, ERK1/2, MSK-1, and histone H3. 17693396_although DARPP-32 impacts cortical inhibition through a previously demonstrated D2-dependent regulation of GABAergic currents in pyramidal cells, it is not involved in the direct D1-mediated regulation of fast-spiking interneurons 18155847_DARPP-32 plays a central role in 1) modulating intercellular coupling, 2) cortical excitatory drive of spiny and aspiny GABAergic neurons, and 3) local feedforward inhibitory drive of cholinergic-like interneurons within accumbens circuits. 18496528_The nuclear accumulation of DARPP-32, a potent inhibitor of protein phosphatase-1, increases the phosphorylation of histone H3, an important component of nucleosomal response. 19050854_Csnk1-epsilon is crucial for the locomotor stimulant response to a moderate dose of MA and suggest that genetic polymorphisms affecting Csnk1-epsilon expression or function could influence sensitivity to amphetamines in both mice and humans. 19737990_Data confirmed that CR specifically increased dopamine- and cAMP-regulated phosphoprotein (Darpp-32), a key regulator of dopamine receptor signaling, in the amygdala. 19852993_results indicate that under chronic dopamine deficiency (a) the A(2A)/cAMP/PKA/DARPP-32 cascade is overactive due to the elevated endogenous phospho-DARPP-32 levels and (b) the A(2A) receptor modulatory effect on ERK1/2 signaling is dysregulated 20236221_Our data demonstrate that beta(1)- and alpha(2)-adrenoceptors regulate DARPP-32 phosphorylation in neostriatal neurons. 20682746_Data show that the loss of DARPP-32 in striatopallidal neurons produced a robust increase in locomotor activity. 21741919_DARPP-32 increases interactions between epidermal growth factor receptor and ERBB3 to promote tumor resistance to gefitinib. 22753408_l-DOPA-induced activation of ERK and mTORC1 requires DARPP-32 and indicates the importance of the cAMP/DARPP-32 signaling cascade in dyskinesia. 23250204_DARPP32 has a role in pluripotent stem cell differentiation toward medium-sized spiny neurons 23558143_This study demonistrated that environment enhanced Darpp32 expression in brain and enhanced totaroad perfermance and short term working menory. 24058659_In this work, the dissected postsynaptic topology of the DARPP-32 phosphoprotein provides strong evidence for a compartmentalized and confined distribution in dendritic spines. 24204683_Data suggest that histone deacetylase inhibitors inhibit the induction of NGF1A-binding protein Nab2 by brain derived neurotrophic factor (BDNF), and thereby the relative induction of dopamine and cyclic AMP-regulated phosphoprotein, 32 kDa (DARPP-32). 24312250_Adenosine AA receptors in striatal glutamatergic terminals and GABAergic neurons oppositely modulate psychostimulant action and DARPP-32 phosphorylation. 25128420_Results show that Darpp-32 was expressed in normal mouse mammary tissue and in some breast tumors, whereas t-Darpp was found exclusively in mammry tumors. 25548102_Results describe the development of new vascular mouse model with a conditional knockout allele of Tsc1 with a Darpp32-Cre allele. This mouse displayed accelerated formation of both kidney cystadenomas and paw hemangiosarcomas. 25692798_analysis of cAMP-activated DARPP-32 signalling to the nucleus 26099530_Cannabinoid-stimulated G-protein activity did not differ between DARPP-32 KO and WT mice treated with vehicle or repeated THC. 26639316_Darpp32 interaction with beta-adducin may be the molecular pathway by which environmental changes may rapidly alter responsiveness of striatal neurons involved in the reward system. 27590997_This study strongly suggests that inflammation-regulated DARPP-32 constitutes a key component of H. pylori-mediated gastric tumourigenesis. 27998980_the results suggest that by inducing dephosphorylation of DARPP-32 at Ser-97 and altering its cytonuclear distribution, glutamate may counteract dopamine/D1 receptor/PKA signaling at multiple cellular levels. 31060804_These results point to Cdk5 in the nucleus accumbens as a critical contributor to depressive-like behaviors in Huntington's disease (HD) mice by altering DARPP-32/beta-adducin signaling and disrupting the dendritic spine cytoskeleton. 32242231_Nemo-like kinase reduces mutant huntingtin levels and mitigates Huntington's disease. 33096475_Epistatic evidence for gender-dependant slow neurotransmission signalling in substance use disorders: PPP1R12B versus PPP1R1B. 35939707_DARPP32, a target of hyperactive mTORC1 in the retinal pigment epithelium. 36055413_PLPP/CIN-mediated DARPP-32 serine 97 dephosphorylation delays the seizure onset in response to kainic acid in the mouse hippocampus. |
ENSG00000131771 |
PPP1R1B |
20.597132 |
7.2433230315 |
2.856652 |
0.66200336 |
18.131147 |
0.00002062008111128483254289450732343880190455820411443710327148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001439436642928920977602819109009146814059931784868240356445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
30.7754345001082 |
14.622612445975 |
4.24880049752224 |
1.68296144506778 |
| ENSMUSG00000061859 |
12695 |
Patj |
protein_coding |
Q63ZW7 |
FUNCTION: Scaffolding protein that facilitates the localization of proteins to the cell membrane (PubMed:11872753). Required for the correct formation of tight junctions and epithelial apico-basal polarity (By similarity). Positively regulates epithelial cell microtubule elongation and cell migration, possibly via facilitating localization of PRKCI/aPKC and PAR3D/PAR3 at the leading edge of migrating cells (By similarity). Plays a role in the correct reorientation of the microtubule-organizing center during epithelial migration (By similarity). May regulate the surface expression and/or function of ASIC3 in sensory neurons (PubMed:11872753). May recruit ARHGEF18 to apical cell-cell boundaries (By similarity). {ECO:0000250|UniProtKB:E2QYC9, ECO:0000250|UniProtKB:Q8NI35, ECO:0000269|PubMed:11872753}. |
Alternative splicing;Cell junction;Cell membrane;Cytoplasm;Membrane;Phosphoprotein;Reference proteome;Repeat;Tight junction |
|
This gene encodes a multivalent PDZ domain protein, which is expressed exclusively in brain and kidney. This protein selectively interacts with inward rectifier K+ (Kir) family members, N-methyl-D-aspartate receptor subunits, neurexins and neuroligins, as well as cell surface molecules enriched in synaptic membranes. Thus, this protein may serve as a scaffold that brings structurally diverse but functionally connected proteins into close proximity at the synapse. Multiple alternatively spliced transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Jul 2008]. |
mmu:12695; |
apical part of cell [GO:0045177]; apical plasma membrane [GO:0016324]; bicellular tight junction [GO:0005923]; cell junction [GO:0030054]; centriolar satellite [GO:0034451]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; perinuclear region of cytoplasm [GO:0048471]; protein-containing complex [GO:0032991]; subapical complex [GO:0035003]; tight junction [GO:0070160]; establishment of apical/basal cell polarity [GO:0035089]; tight junction assembly [GO:0120192] |
15863617_analysis of structures of tetrameric L27 domain complexes formed by mLin-2/mLin-7 and Patj/Pals1 scaffold proteins 17397395_Data show that MUPP1 interacts with angiomotin (Amot), JEAP/Amot-like 1 and MASCOT/Amot-like 2, and that all the Amot/JEAP family proteins also interacted with Patj, a close relative of MUPP1. 19138174_Data show taht channel-interacting PDZ protein, 'CIPP', interacts with proteins involved in cytoskeletal dynamics, in particular, with the small G-protein effector IRSp53 |
ENSG00000132849 |
PATJ |
55.154979 |
3.4706626082 |
1.795211 |
0.49317199 |
12.600928 |
0.00038555534014074184338827233631263879942707717418670654296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0019847174521878271045982611298086339957080781459808349609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
76.3764944982562 |
26.6200663556998 |
22.0063149662618 |
5.7355876010592 |
| ENSMUSG00000062074 |
327743 |
Ccn6 |
protein_coding |
D3Z5L9 |
FUNCTION: Plays a role in mitochondrial electron transport and mitochondrial respiration. {ECO:0000250|UniProtKB:O95389}. |
Disulfide bond;Glycoprotein;Growth factor;Mitochondrion;Reference proteome;Secreted;Signal |
|
|
mmu:327743; |
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; mitochondrion [GO:0005739]; growth factor activity [GO:0008083]; heparin binding [GO:0008201]; integrin binding [GO:0005178]; cell adhesion [GO:0007155]; negative regulation of angiogenesis [GO:0016525]; negative regulation of cell death [GO:0060548]; negative regulation of cell population proliferation [GO:0008285]; regulation of mitochondrial membrane potential [GO:0051881]; regulation of reactive oxygen species biosynthetic process [GO:1903426]; signal transduction [GO:0007165] |
15601861_the WISP3 gene is essential to skeletal growth or homeostasis in humans but not in mice [WISP3] 21921419_demonstrated that CCN6 induced the proliferation of lung fibroblasts by binding to integrin beta1, leading to the phosphorylation of FAK(Y397). 29920361_WISP-3/CCN6 has a role in inhibiting apoptosis by regulating caspase pathway after hyperoxia in lung epithelial cells |
ENSG00000112761 |
CCN6 |
14.984730 |
4.0074080602 |
2.002669 |
0.69459771 |
8.182173 |
0.00423040497872743340851409143965611292514950037002563476562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0161255076666607273516707010685422574169933795928955078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.4869887843416 |
10.7494192586541 |
5.36181703019388 |
2.17119928580984 |
| ENSMUSG00000062082 |
239849 |
Cd200r4 |
protein_coding |
Q6XJV4 |
FUNCTION: Involved in the recruitment or surface expression of the TYROBP receptor. {ECO:0000269|PubMed:12960329, ECO:0000269|PubMed:15187158, ECO:0000269|PubMed:15471863, ECO:0000269|PubMed:16081818}. |
3D-structure;Disulfide bond;Glycoprotein;Immunoglobulin domain;Membrane;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
Human_homologues NA; + ;NA |
|
mmu:239849; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; plasma membrane [GO:0005886]; signaling receptor activity [GO:0038023]; regulation of neuroinflammatory response [GO:0150077] |
Human_homologues 12960329_The CD200 receptor gene family resembles the signal regulatory proteins and killer Ig-related receptors in having receptor family members with potential activatory and inhibitory functions that may play important roles in immune regulation and balance. 15220441_Human Herpesvirus-8 K14 protein interacts with human CD200R during the lytic cycle. Despite sharing only 40% primary sequence identity, K14 and CD200 interacts with CD200R with an almost identical and low affinity (K(D) = 0.5 microM) 15661892_CD200R represents a novel and potent inhibitory receptor that can be targeted in vivo to regulate mast cell-dependent pathologies. 16177086_Human CD200R is an important regulatory molecule of basophil activation. 16354172_Reactions with CD200 antigen modulate immunomodulation for prevention of undesired immune responses of the skin. 17714785_In human tonsils, CD200R is differentially expressed on B cells, with high expression on memory cells and plasmablasts. 18040859_Review highlights the close correlation between CD200-CD200 receptor (CD200R), microglia activation, and Parkinson's disease. 18164423_The CD200-CD200R pathway seems of critical relevance for macrophage-mediated damage in autoimmune inflammation of the CNS. 19151626_CD200-mediated immune suppression may occur not only via neuron-microglia interactions, but also via glia-glia interactions, especially in inflammatory conditions in which an immune-suppressive environment needs to be restored. 19786546_CD200R inhibits the activation of human myeloid cells through direct recruitment of Dok2 and subsequent activation of RAS p21 protein activator 1. 19924532_results suggest an intrinsic abnormality in the CD200-CD200R signaling in monocyte-derived macrophages during aging and, especially, in Parkinson disease 21078907_Dok1 negatively regulates Dok2-mediated CD200R signaling through the recruitment of CrkL. 22363701_The role of the CD200-CD200R axis in bone marrow-derived mesenchymal stem cell mediated immunosuppression was studied using THP-1 human macrophages. 22491458_Mechanistically, Kaposi's sarcoma-associated herpesvirus viral OX2 protein and CD200 expression on antigen presenting cells suppressed the phosphorylation of ERK1/2 mitogen-activated protein kinase in responding T cells. 22496920_these results indicate a role of CD200R:CD200 in T cell responses to helminths which has diagnostic and prognostic relevance as a marker of infection for chronic schistosomiasis in mouse and man. 22575528_Mesenchymal stromal cells are able to modulate the expression of both CD200 and CD200R on some T-cells. 22621248_CD200 and CD200R1 expression and function are abnormal in systemic lupus erythematosus 23066977_The expression of CD200R as well as B7-H4 co-stimulatory molecules on CD14(+) cells were significantly higher in cord blood when compared with peripheral blood. 23541619_blocking CD200 on tumor cells may have opposite effects on tumor proliferation depending on the 'affinity' of the macrophages to form the CD200-CD200R-complex with tumor cells. 23560298_in cutaneous squamous cell carcinoma, CD200 expression on local blood vessels may promote tumor progression by suppressing CD200R myeloid cells during diapedesis 23690219_The expression of CD200 and its receptor, CD200R, on CD83+ monocyte-derived dendritic cells (Mo-DCs), pulsed or not with autologous tumor cell lysates (aTCL) in patients who suffer from laryngeal carcinoma, were examined. 23940819_Mesenchymal stem cells, through the expression of CD200, play a major role in the regulation of bone resorption and bone physiology and the CD200-CD200R couple could be a new target to control bone diseases. 24333170_Measurement of sCD200 and/or sCD200R1 may prove a useful and rapid means of monitoring subjects at risk of bone loss. 24388216_[review] CD200R1 is comparable to other pathogen-targeted inhibitory receptors; its signaling pathway is utilized by a diverse number of pathogens and represents a novel targeting strategy for infectiouis diseases. 24496593_We demonstrate a significant correlation between CD200R1 positive cells and disease severity in rheumatoid arthritis (RA) patients, thus indicating the relevance of the CD200/CD200R1 signaling pathway potential involvement in the pathogenesis of RA. 25145957_The mechanism underlying ESA might be associated with enhanced expression of CD200 and CD200R in the trophoblast, leading to an upregulation of the immune response during the first trimester of pregnancy. 25261692_The proportion of CD200R1+ cells in PBMCs, peripheral CD14+ cells and CD4+ T cells was significantly lower in rheumatoid arthritis patients compared to controls. 25404054_data show that the expression of CD200R, CD95 and CD95L was influenced by cardiac surgery and imply the role of these membrane molecules in cell regulation-inhibition and apoptosis following cardiac surgery. 26011475_This is the first report of CD200R1 expression by human epithelial tumor cells, and specifically, early-stage human breast cancer cells. It is also the first report of CD200R1 expression by term placental villous trophoblasts. 26123445_CD200-CD200R1 signaling may be required for human pregnancy success. 26538068_The authors showed that the viral orthologues e127 from rat cytomegalovirus and K14 from human herpesvirus 8 do not bind the activating CD200R-like proteins from their respective species, although they do bind the inhibitory receptors. 26690123_Inflammatory Bowel Disease patients showed a significant decrease in the percentage of plasmacytoid dendritic cells and myeloid dendritic cells expressing CD200R1 compared to that of the control group. 26923638_Constitutively expressed Siglec-9 inhibits LPS-induced CCR7, but enhances IL-4-induced CD200R expression in human macrophages. 27385779_we revealed a critical role for CD200R signaling in limiting the growth and metastasis of CD200(+) tumors. 27562325_Our findings suggest a promising role of CD200R as a prognostic marker in predicting elevated recurrence and reduced survival, and a potential therapeutic target in treating hepatocellular carcinoma. 27929051_Reduced expression of monocyte CD200R is associated with enhanced proinflammatory cytokine production in sarcoidosis 28106273_we demonstrated for the first time that CD200R is highly expressed on cells involved in type 2 immune responses, namely on Th2, Tc2, ILC2, and basophils. All these cell types play a role in type 2 inflammation. 28326648_Residual CD200 activity may prevent completion of abortions via induction of Treg cells. In chronic histiocytic intervillositis, infiltrating maternal effector T cells may block Treg induction. An autocrine role for CD200-CD200R interaction versus inhibition of soluble CD200 by soluble CD200R is discussed. 29042364_Coexpression of CD200R-CD28 enhances function in WT1-specific T-cell receptor - transduced human primary T cells. 29101312_MMP12-producing monocyte-derived cells in gingival tissue of periodontitis patients also had reduced CD200R surface levels. Treatment with a CD200R ligand reduced CSF2-induced MMP12 production in these cells. This novel association of the CD200/CD200R pathway with MMP12 production by monocyte-derived cells may play a key role in PD progression. 32030689_Review of the CD200-CD200R axis, including the biology behind CD200-CD200R interaction and the role(s) it plays in tumor microenvironment and tumor growth, and activation/effector functions of T cells; discussion of CD200-CD200R's role as potential checkpoint molecules for cancer immunotherapy 32190938_Identification of the CD200R1 promoter and the association of its polymorphisms with the risk of Parkinson's disease. 33321503_Tumor-Infiltrating T Cells Concurrently Overexpress CD200R with Immune Checkpoints PD-1, CTLA-4, and TIM-3 in Non-Small-Cell Lung Cancer. 33463116_Up-regulation of CD200/CD200R1 Immunomodulatory Axis of Allogenic Peripheral Blood Mononuclear Cells in a Co-culture with Adipose-derived Mesenchymal Stem Cells. 33780466_Identification of a novel conserved signaling motif in CD200 receptor required for its inhibitory function. 35326506_The Role of CD200-CD200 Receptor in Human Blood and Lymphatic Endothelial Cells in the Regulation of Skin Tissue Inflammation.; + ;NA |
ENSG00000163606+ENSG00000206531 |
CD200R1+CD200R1L |
135.099485 |
9.4685629333 |
3.243145 |
0.27825086 |
136.273076 |
0.00000000000000000000000000000017389301758682825548232611503968231048982344537187996906245193941646795205811730608731757730112121862475760281085968017578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000143175051020679344010406839118948038491502021102262493843038759786932536817688314811913841140267322771251201629638671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
238.450924955878 |
46.2753983050519 |
25.1834334984923 |
4.09775337096982 |
| ENSMUSG00000062168 |
237178 |
Ppef1 |
protein_coding |
O35655 |
FUNCTION: May have a role in the recovery or adaptation response of photoreceptors. May have a role in diverse sensory neurons and in development. |
Calcium;Hydrolase;Magnesium;Manganese;Metal-binding;Protein phosphatase;Reference proteome;Repeat |
|
|
mmu:237178; |
cytosol [GO:0005829]; nucleus [GO:0005634]; calcium ion binding [GO:0005509]; iron ion binding [GO:0005506]; manganese ion binding [GO:0030145]; myosin phosphatase activity [GO:0017018]; protein serine/threonine phosphatase activity [GO:0004722]; detection of stimulus involved in sensory perception [GO:0050906] |
33783058_The protein phosphatase with EF-hand domain 1 is a calmodulin-binding protein that interacts with proteins involved in sperm capacitation, binding to the zona pellucida, and motility. |
ENSG00000086717 |
PPEF1 |
28.546907 |
5.0307107666 |
2.330762 |
0.87329897 |
6.475128 |
0.01093944454506993145537219191965050413273274898529052734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0361687908206002189936967283756530378013849258422851562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.2684723293856 |
27.8205419432975 |
8.60086662443329 |
4.58172510093477 |
| ENSMUSG00000062310 |
14659 |
Glrp1 |
protein_coding |
E9Q9S8 |
|
Reference proteome |
|
|
mmu:14659; |
nucleus [GO:0005634]; negative regulation of transcription by RNA polymerase II [GO:0000122] |
|
|
|
25.800143 |
3.2585821359 |
1.704244 |
0.51278377 |
10.892854 |
0.00096535967534218381207872239357925536751281470060348510742187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0044775756926955373460330633861303795129060745239257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.7467367362311 |
13.7007797317058 |
11.5837917111337 |
3.34783219361006 |
| ENSMUSG00000062611 |
100043780 |
Rps3a2 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
1360.993824 |
0.2283864938 |
-2.130451 |
0.50495446 |
16.253335 |
0.00005541196936729533861244745285290491665364243090152740478515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003513884751884440446959012582794912304962053894996643066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
475.222179050284 |
169.74839879835 |
2080.78057163585 |
550.246775341162 |
| ENSMUSG00000062937 |
66902 |
Mtap |
protein_coding |
Q9CQ65 |
FUNCTION: Catalyzes the reversible phosphorylation of S-methyl-5'-thioadenosine (MTA) to adenine and 5-methylthioribose-1-phosphate. Involved in the breakdown of MTA, a major by-product of polyamine biosynthesis. Responsible for the first step in the methionine salvage pathway after MTA has been generated from S-adenosylmethionine. Has broad substrate specificity with 6-aminopurine nucleosides as preferred substrates. {ECO:0000255|HAMAP-Rule:MF_03155}. |
Acetylation;Cytoplasm;Glycosyltransferase;Nucleus;Purine salvage;Reference proteome;Transferase |
PATHWAY: Amino-acid biosynthesis; L-methionine biosynthesis via salvage pathway; S-methyl-5-thio-alpha-D-ribose 1-phosphate from S-methyl-5'-thioadenosine (phosphorylase route): step 1/1. {ECO:0000255|HAMAP-Rule:MF_03155}. |
|
mmu:66902; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; S-methyl-5-thioadenosine phosphorylase activity [GO:0017061]; L-methionine salvage from methylthioadenosine [GO:0019509]; methylation [GO:0032259]; purine ribonucleoside salvage [GO:0006166] |
18237276_Regulation of MTAP by reactive oxygen species might participate in the redox regulation of the methionine catabolic pathway in the liver 19567676_Studies show that Mtap is a tumor suppressor gene independent of CDKN2A and ARF. 23840755_Germline Mutations in Mtap is associated with lymphomagenesis. 24324622_MTAP-mediated regulation of methylthioadenosine links polyamine metabolism with NF-kappaB activation and apoptosis in hepatic stellate cells. 26819315_Data demonstrate that MTAP deficiency leads to accumulation of MTA inducing deregulation of central cellular pathways that control cell fate and contributes to the onset of a proliferative and antiapoptotic phenotype in the liver. |
ENSG00000099810 |
MTAP |
29.419395 |
0.0030921614 |
-8.337169 |
1.29292295 |
66.215934 |
0.00000000000000040413241623408564869332140108339684726671732777243672973810362236690707504749298095703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000112949448882157666223044964397237016628678524243767355983436573296785354614257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.188135391338368 |
0.19209719205797 |
60.8426817001755 |
18.0707717158323 |
| ENSMUSG00000063296 |
320709 |
Tmem117 |
protein_coding |
Q8BH18 |
FUNCTION: Involved in endoplasmic reticulum (ER) stress-induced cell death pathway. {ECO:0000250|UniProtKB:Q9H0C3}. |
Cell membrane;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:320709; |
endoplasmic reticulum [GO:0005783]; plasma membrane [GO:0005886]; intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress [GO:0070059] |
Human_homologues 19913121_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 28285135_Data suggest that transmembrane protein 117 (TMEM117) probably mediates the signalling of mitochondrial membrane potential (DeltaPsim) loss in endoplasmic reticulum (ER) stress-mediated mitochondria cell death. |
ENSG00000139173 |
TMEM117 |
9.361273 |
0.1421496461 |
-2.814518 |
0.95405337 |
8.791092 |
0.00302704998409773122849952819990448915632441639900207519531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0120986684224026874029389588827143597882241010665893554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.33076550950942 |
1.6561579096866 |
16.3965621634106 |
7.19691264179167 |
| ENSMUSG00000063535 |
76373 |
Zfp773 |
protein_coding |
A0A1B0GRS1 |
|
Reference proteome |
|
|
|
DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of DNA-templated transcription [GO:0006355] |
|
|
|
27.407525 |
3.6183149957 |
1.855318 |
0.58375344 |
9.912968 |
0.00164118253990404609418685133448434498859569430351257324218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0071711108570907721240783594396361877443268895149230957031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
44.5379046370089 |
18.5170309070686 |
12.3090180623137 |
3.97349361159562 |
| ENSMUSG00000063564 |
237759 |
Col23a1 |
protein_coding |
Q8K4G2 |
|
Cell membrane;Collagen;Membrane;Reference proteome;Repeat;Signal-anchor;Transmembrane;Transmembrane helix |
|
|
mmu:237759; |
cell surface [GO:0009986]; collagen trimer [GO:0005581]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; plasma membrane [GO:0005886]; extracellular matrix structural constituent [GO:0005201]; heparin binding [GO:0008201]; identical protein binding [GO:0042802]; extracellular matrix organization [GO:0030198] |
16728390_analysis of collagen XXIII mRNA and protein |
ENSG00000050767 |
COL23A1 |
92.710695 |
5.2746687179 |
2.399080 |
0.31189112 |
58.167218 |
0.00000000000002407577256403376832475748878043263177264020347134731991900480352342128753662109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000005802565944546872464171842759366514515224910741864050578442402184009552001953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
154.518915060332 |
33.6657098613449 |
29.2945250829179 |
5.16124185161942 |
| ENSMUSG00000063611 |
|
Gm10134 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
16.080826 |
0.1587465239 |
-2.655203 |
0.90496689 |
7.977600 |
0.00473596932045780832226666845485851808916777372360229492187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0177680504160623968512577874889757367782294750213623046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.53904525410413 |
3.03765744485767 |
28.5930371511264 |
13.4571558188694 |
| ENSMUSG00000063632 |
20666 |
Sox11 |
protein_coding |
Q7M6Y2 |
FUNCTION: Transcription factor that acts as a transcriptional activator (PubMed:18505825, PubMed:18403418). Binds cooperatively with POU3F2/BRN2 or POU3F1/OCT6 to gene promoters, which enhances transcriptional activation (PubMed:18505825, PubMed:18403418). Acts as a transcriptional activator of TEAD2 by binding to its gene promoter and first intron (PubMed:20596238). Plays a redundant role with SOX4 and SOX12 in cell survival of developing tissues such as the neural tube, branchial arches and somites, thereby contributing to organogenesis (PubMed:20596238). {ECO:0000269|PubMed:18403418, ECO:0000269|PubMed:18505825, ECO:0000269|PubMed:20596238}. |
Activator;Developmental protein;Differentiation;DNA-binding;Neurogenesis;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:20666; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; plasma membrane [GO:0005886]; cis-regulatory region sequence-specific DNA binding [GO:0000987]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; transcription cis-regulatory region binding [GO:0000976]; anatomical structure morphogenesis [GO:0009653]; cardiac ventricle formation [GO:0003211]; cell differentiation [GO:0030154]; closure of optic fissure [GO:0061386]; cornea development in camera-type eye [GO:0061303]; embryonic digestive tract morphogenesis [GO:0048557]; embryonic skeletal system morphogenesis [GO:0048704]; eyelid development in camera-type eye [GO:0061029]; glial cell development [GO:0021782]; glial cell proliferation [GO:0014009]; hard palate development [GO:0060022]; kidney development [GO:0001822]; lens morphogenesis in camera-type eye [GO:0002089]; limb bud formation [GO:0060174]; lung morphogenesis [GO:0060425]; negative regulation of cell death [GO:0060548]; negative regulation of gene expression [GO:0010629]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of lymphocyte proliferation [GO:0050672]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of transcription regulatory region DNA binding [GO:2000678]; neural crest cell development [GO:0014032]; neural tube formation [GO:0001841]; neuroepithelial cell differentiation [GO:0060563]; neuron differentiation [GO:0030182]; noradrenergic neuron differentiation [GO:0003357]; oligodendrocyte development [GO:0014003]; outflow tract morphogenesis [GO:0003151]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of gene expression [GO:0010628]; positive regulation of hippo signaling [GO:0035332]; positive regulation of hormone secretion [GO:0046887]; positive regulation of lens epithelial cell proliferation [GO:2001111]; positive regulation of neurogenesis [GO:0050769]; positive regulation of neuron differentiation [GO:0045666]; positive regulation of ossification [GO:0045778]; positive regulation of osteoblast differentiation [GO:0045669]; positive regulation of stem cell proliferation [GO:2000648]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of DNA-templated transcription [GO:0006355]; regulation of transforming growth factor beta receptor signaling pathway [GO:0017015]; skeletal muscle cell differentiation [GO:0035914]; skeletal system development [GO:0001501]; soft palate development [GO:0060023]; somite development [GO:0061053]; spinal cord development [GO:0021510]; sympathetic nervous system development [GO:0048485]; ventricular septum morphogenesis [GO:0060412] |
15254231_SOX11 has a role in in tissue remodeling 15456859_Data support the model that synergistic interactions between group B1/C SOX (SOX1/2/3, SOX11) and class III POU (Brn2)within the nucleus determine Nestin expression. 18505825_Sox4 and Sox11 function redundantly with Sox12 and can compensate its loss during mouse development. 19133245_These anatomical and expression data support an intrinsic role for Sox11 in events that underlie successful regeneration following peripheral nerve injury. 19490090_Sox11 is involved in the transcriptional regulation of specific gene expression in adult neurogenesis at the stage of the immature neuron. 19799774_The study reports spatio-temporally regulated Sox4 and Sox11 antisense transcripts in the brain, neural stem/progenitor cells and P19 cells, suggesting they have an important role in cerebral corticogenesis and neuronal/glial cell differentiation. 20147379_In the absence of both Sox4 and Sox11, sympathetic ganglia remained hypoplastic throughout embryogenesis because of consecutive proliferation and survival defects. 20596238_Study show that the survival of neural and mesenchymal progenitor cells critically relies on the redundantly acting SoxC transcription factors Sox4, Sox11 and Sox12. 20646169_Sox11 protein directly function as pro-survival factors during spinal cord development in neural cell types 21117150_Sox11 is a key regulator of sensory neuron development. 21880559_The results clearly showed a correlation between SOX11 level and cellular growth rate, which was dependent on autotaxin, as well as a direct relation between the level of SOX11 in tumorigenic cells and the growth rate of these tumors in NOD-SCID mice. 22024412_The results of this study revealed a new functional linkage between Sox11 and Sprr1a in adult peripheral neuron regeneration. 22085726_genome-wide binding for Sox11, which have vital functions in ES cells, neural precursor cells (NPCs), and maturing neurons, respectively. (Sox11) 22331573_The results of this study indicted that cell type and contextual specificity of Sox11 in modulation of BDNF transcription. 22378879_SoxC transcription factors Sox11 and Sox4 target the promoters of genes that are induced upon neuronal differentiation of adult neural stem cells (NSCs). 22465478_Sox11 represents a synergistic factor for WT1 in regulating the Wnt4 gene expression that is critical for nephrogenesis during kidney ontogeny. 22687574_Sox11 modulates neocortical development by regulating the proliferation and neuronal differentiation of cortical intermediate precursors 23201825_TANK is expressed in DRG neurons. TANK is increased by peripheral nerve injury. the regulation of TANK expression is, at least in part, controlled by the injury-associated transcription factor Sox11. 23313252_Data suggest that levels of Sox11 decline in developing retina; levels of histone H3-acetylation and H3-lysine 4 trimethylation at Sox11 gene locus decline during retinal development but increase markedly in adult retina. 23318640_The sequential expression of Sox11 and Sox4 during retinogenesis leads to the fine adjustment of retinal differentiation by helping to establish the competency of retinal progenitors. 23356643_This work implicates SOX11 as a potential regulator of GDF5 expression in joint maintenance and suggests a possible role in the pathogenesis of osteoarthritis 23483698_Sox11 is required for both embryonic and adult neurogenesis 23649630_Data indicate that a complete loss of retinal ganglion cells (RGCs) was observed in Sox4/Sox11-null retinas, suggesting the two genes play similar roles in the development of RGCs. 23888050_Sox11 plays a crucial role in the proliferation and survival of mesenchymal and osteoblast precursors by Tead2, and osteogenic differentiation by regulating Runx2 and Osterix. 24310815_Sox4 and Sox11 are required for proper development of cardiac neural crest cells. 24589730_The expression of Sox11 is increased in the injured spinal cord. Sox11 expression is located in ependymal cells lining the central canal and in newly-generated neurons in the injured spinal cord. 24722414_regulates osteogenesis 24886874_Fetal Sox11 disruption lead to hypoplastic lungs, ventricular septation defects, abdominal defects, skeletal malformations, decreased birth weight and postnatal lethality. 25234735_Sox11 expression was high in fetal. 25698749_These data identify Sox11 as a key transcription factor that can confer an elevated innate regenerative capacity to CNS neurons. 25761772_SOXC proteins act cell- and non-cell-autonomously in perichondrocytes and chondrocytes to establish noncanonical WNT signaling crosstalk essential for growth plate induction and control 26203155_Sox11 and Sox4 are not redundant in promoting neuronal differentiation in the cortex, but rather act in overlapping and discrete populations of neurons 26826126_Sox11 directly regulates the expression of Fgf9; ablation of the Sox11 gene results in clefting of the secondary palate resembling the Pierre Robin sequence. 27225767_These findings indicate that suppression of dendritic morphogenesis by Sox11 during radial migration is crucial for the formation of the cerebral cortex. 27241892_Following elevation of the palatal shelves (E14.5), Sox2, Sox11 and Sox21 expression was present in the midline epithelial seam. We thus identify dynamic spatio-temporal expression of Sox gene family during the process of palatogenesis 28429312_SOX11 represents a useful prognostic marker in mantle cell lymphoma. 28641110_Sox11 can induce substantial axon regeneration. Transcriptome profiling indicated that Sox11 activates genes involved in cytoskeletal remodeling and axon growth. Remarkably, alpha-RGCs, which preferentially regenerate following treatments such as Pten deletion, were killed by Sox11 overexpression. Thus, Sox11 promotes regeneration of non-alpha-RGCs, which are refractory to Pten deletion-induced regeneration. 29459093_Data demonstrate a key role for SOX11 in normal kidney development and may suggest that variants in this gene predispose to CAKUT. 29746989_These data support the role of Sox11 to promote neurite growth by altering responsiveness of neurotrophic factors 29954934_The global gene expression profile for SRY-box containing gene 11 protein (Sox11)-ON cells following 72 h of Sox11 expression was distinct from both Sox11-OFF and non-transduced Ba/F3 cells. 30385877_These data provide novel insight into the control of the critical neurodevelopmental regulator SOX11 and imply SOX11 as a mediator of PKA-regulated neuronal development. 30879763_The dysregulation in the Sox11 and FAK after High volume mechanical ventilation play an important role in the pathogenesis of Ventilator-induced lung injury (VILI), and facilitating the activity of Sox11and FAK might be an effective target and potential option in the prevention and treatment of VILI in clinic. 31492871_Data indicate that the reactivated embryonic gene program during wound repair and that SRY-box containing gene 11 protein (SOX11) and SRY-box containing gene 4 protein (SOX4) play a central role in this process. 32080705_Sox11 is an Activity-Regulated Gene with Dentate-Gyrus-Specific Expression Upon General Neural Activation. 32346881_Wnt7b-induced Sox11 functions enhance self-renewal and osteogenic commitment of bone marrow mesenchymal stem cells. 33236128_SOX11 and FAK participate in the stretchinduced mechanical injury to alveolar type 2 epithelial cells. 33441400_Posttranslational Modification of Sox11 Regulates RGC Survival and Axon Regeneration. 33662665_Epithelial SOX11 regulates eyelid closure during embryonic eye development. 33969421_Sox11 regulates mammary tumour-initiating and metastatic capacity in Brca1-deficient mouse mammary tumour cells. 34374250_[Effects of Sox11 gene on neuronal migration in the development mouse cerebral cortex]. 35254515_Disrupted mossy fiber connections from defective embryonic neurogenesis contribute to SOX11-associated schizophrenia. 35642566_Cochlear nerve deficiency in SOX11-related Coffin-Siris syndrome. 36571426_Silencing SOX11 Alleviates Allergic Rhinitis by Inhibiting Epithelial-Derived Cytokines. |
ENSG00000176887 |
SOX11 |
172.571272 |
4.9284623669 |
2.301138 |
0.21636442 |
111.989893 |
0.00000000000000000000000003591362748195557721207865191618108527643797159031087335312994955730013860173421846866403939202427864074707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000021121095513712252579490596768040718000164420176407935984615898451777216138225412578321993350982666015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
287.519671958501 |
43.2818339460038 |
58.3386156880586 |
7.12187861212257 |
| ENSMUSG00000063687 |
93876 |
Pcdhb5 |
protein_coding |
Q91XZ5 |
Human_homologues FUNCTION: Potential calcium-dependent cell-adhesion protein. May be involved in the establishment and maintenance of specific neuronal connections in the brain. |
Calcium;Cell adhesion;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:93876; |
plasma membrane [GO:0005886]; calcium ion binding [GO:0005509]; cell adhesion [GO:0007155]; homophilic cell adhesion via plasma membrane adhesion molecules [GO:0007156] |
|
ENSG00000081818 |
PCDHB4 |
156.797233 |
2.3994025991 |
1.262675 |
0.20885731 |
36.369179 |
0.00000000163264693265330497767847693960617527775269763878895901143550872802734375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000223235889391159268092952019848368427901164068316575139760971069335937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
225.15008821627 |
32.7086928681047 |
93.8358941115391 |
10.6097089622551 |
| ENSMUSG00000063696 |
667618 |
Gm8730 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
165.443969 |
0.1597009540 |
-2.646555 |
0.37403836 |
46.136285 |
0.00000000001103065115221913081838963210200554041287568374229977052891626954078674316406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000001951438772945433188729767401233179159592001639111913391388952732086181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
42.5291185474907 |
11.1501962276243 |
266.304724425743 |
49.8274054578527 |
| ENSMUSG00000063727 |
18383 |
Tnfrsf11b |
protein_coding |
O08712 |
FUNCTION: Acts as decoy receptor for TNFSF11/RANKL and thereby neutralizes its function in osteoclastogenesis. Inhibits the activation of osteoclasts and promotes osteoclast apoptosis in vitro. Bone homeostasis seems to depend on the local ratio between TNFSF11 and TNFRSF11B. May also play a role in preventing arterial calcification. May act as decoy receptor for TNFSF10/TRAIL and protect against apoptosis. TNFSF10/TRAIL binding blocks the inhibition of osteoclastogenesis. {ECO:0000269|PubMed:10952716}. |
3D-structure;Apoptosis;Disulfide bond;Glycoprotein;Receptor;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:18383; |
extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; receptor complex [GO:0043235]; apoptotic process [GO:0006915]; extracellular matrix organization [GO:0030198]; negative regulation of bone resorption [GO:0045779]; negative regulation of odontogenesis of dentin-containing tooth [GO:0042489]; negative regulation of osteoclast differentiation [GO:0045671]; response to arsenic-containing substance [GO:0046685]; response to estrogen [GO:0043627]; response to magnesium ion [GO:0032026]; response to nutrient [GO:0007584]; response to xenobiotic stimulus [GO:0009410]; signal transduction [GO:0007165] |
11918293_Osteoprotegerin(OPG) ex vivo gene therapy may be useful to treat patients for aseptic loosening of prosthetic implants. 12211438_expression of RANKL and osteoprotegerin mRNAs by marrow stromal cells 12446599_elevated levels of osteoprotegerin in mutant mice contribute at least in part to reveal the osteopetrotic phenotype in klotho mutant mice 12506018_Osteosclerosis in mice with thrombopoietin overexpression occurs predominantly via up-regulation of osteoprotegerin in host stromal cells leading to disruption of osteoclastogenesis. 12526098_The presence of OPG on the membrane of these cells was confirmed by western blotting and immunostaining, sRANKL was found to be bound to the OPG on the osteoclast precursors. 12568404_expression of RANKL and OPG change with age in whole bone and in cultured osteoblast-like cells 12697767_Runx2 promotes osteoclast differentiation by inducing RANKL and inhibiting OPG 12923331_osteoprotegerin and RANK ligand have roles in breast cancer bone metastasis 14500574_bone formation is coupled with bone resorption at local sites in OPG-/- mice, and that serum RANKL levels do not reflect this coupling. 14525906_OPG secretion is regulated through calmodulin-sensitive protein kinase signaling that depends on the activity of the L-type VSCC and is mediated through the cAMP response element-binding protein. 14969393_suppression of gene expression affects osteoclastogenic activity of parathyroid hormone 15340228_Expression in mice with collagen-induced arthritis treated with Kanzo-bushi-to, a Kampo medicine. 15474477_expression of RANKL and OPG and the formation of osteoclast-like cells do not require Runx2 15845617_RANKL inhibitor osteoprotegerin is used to evaluate the role of osteoclast-mediated hypercalcemia in two murine models of hypercalcemia of malignancy. 15855336_Microorganism-reactive CD4+ T-cells and the RANKL-osteoprotegerin axis provide the molecular basis of the advanced periodontal breakdown in diabetes. 16251181_IL-4 and IL-13, via a STAT6-dependent pathway, inhibit osteoclast differentiation and bone resorption by activating receptors on osteoblasts and osteoclasts that affect the RANKL/RANK/OPG system 16467704_findings in OPG knockout mice support the hypothesis that OPG is important in the inhibition of bone remodeling within the otic capsule and the maintenance of normal auditory function 16614077_Glucocorticoids regulate osteoclast differentiation and expression of receptor activator of nuclear factor-kappaB (NF-kappaB) ligand, osteoprotegerin, and receptor activator of NF-kappaB in mouse calvarial bones 16840715_OPG inhibits advanced plaque progression by preventing an increase in lesion size and lesion calcification. OPG may act as a survival factor and may modulate MMP9 production in vascular cells. 16927271_microgravity is capable to indirectly stimulate osteoclast formation and activity by regulating osteoblast secretion of crucial regulatory factors such as RANKL and OPG 17148684_Altogether, these findings suggest that increased OPG production represents an early event in the natural history of diabetes mellitus, possibly contributing to disease-associated endothelial cell dysfunction. 17182555_OPG suppresses the shedding of RANKL from osteoblasts and the serum RANKL in OPG(-/-) mice exactly reflects the state of bone resorption. 17516544_bone resorptive action of Dexamethasone is mediated mainly by the inhibition of OPG by transrepressing the OPG gene through the AP-1 site 17560185_These results demonstrate that heparin enhances osteoclastic bone resorption by inhibiting osteoprotegerin activity. 17615383_results demonstrated that OPG had anticalcification activity in the aorta, probably through the downregulation of alkaline phosphatase activity 18172035_data support a role for osteoprotegerin in the vasculature as an inhibitor of calcification and a marker, rather than a mediator, of atherosclerosis 18242177_R-sponin1 (Rspo1) acted synergistically with Wnt3A to activate Wnt/beta-catenin signaling in the uncommitted mesenchymal C2C12 cells. 18668542_synovial fibroblasts may significantly contribute to bone resorption through modulation of RANKL and OPG production in a cytokine-rich milieu of inflamed joints. 18668550_in an osteoarthritis model, disruption of RANKL/OPG balance might result in the degradation of cartilage subjected to mechanical loading. 18788552_RANKL and OPG could have a close relationship with the osteoclast activity during mandibular first molar eruption. 19170762_BMP-2 enhanced Wnt/beta-catenin-dependent transcriptional activation of the OPG promoter. 19633200_ADA-deficient SCID is associated with a specific microenvironment and bone phenotype characterized by RANKL/OPG imbalance and osteoblast insufficiency. 19641036_OPG regulates not only DC survival but also the nature of DC-dependent inflammatory responses. 20200998_mechanical stretching may play an important role in bone remodeling through the upregulation of OPG and the mechanical signaling leading to OPG induction involves the noncanonical Wnt pathway. 20369527_Co2+ and Cr3+ can stimulate the mRNA expressions of RANKL and osteoprotegerin in osteoblasts. 20936961_overexpression in preosteoblasts promotes their differentiation into mature osteoblasts 21079361_distribution of the osteocytic lacunar-canalicular system and osteocyte-secreting molecules--dentin matrix protein 1 and sclerostin--in the epiphyses and cortical bones in osteoprotegerin deficiency 21332025_Estrogens modulate RANKL-RANK/osteoprotegerin mediated interleukin-6 effect on thyrotoxicosis-related bone turnover. 21346198_Increased bone resorption is a noncell autonomous defect, caused by exuberant stimulation of osteo-clastogenesis by Cx43-deficient bone marrow stromal cells, via decreased Opg production. 21426505_TRAIL and osteoprotegerin are expressed in atherosclerotic plaques in a calcification-prone mouse model 21439958_Results suggest that ERalpha regulates osteoprotegerin and IL-6 production in osteoblasts and modulates BMD. 21478097_Osteoprotegerin serum levels were decreased in all IL-6 deficient mice and in a highest degree in sham-operated hypothyroid mice. 21656335_Coptisine inhibits RANKL-induced NF-kappaB phosphorylation in osteoclast precursors and suppresses function through the regulation of RANKL and OPG gene expression in osteoblastic cells. 21750093_Omentin-1 ameliorates arterial calcification and bone loss in vivo through the regulation of the RANK signalling pathway in osteoprotegerin-deficient mice. 22196183_OPG gene transfer to periodontal tissues could inhibit relapse after orthodontic tooth movement, through the inhibition of osteoclastogenesis 23071670_Data indicate in TLR4(-/-) mice, higher expression levels of IL-1beta and lower expression levels of VEGF, RANK, RANKL and OPG were detected at late time points (> postoperative 4 days). 23268391_High concentrations of alendronate decrease the expressions of OPG and RANKL in cultured osteoblasts. 23331792_In a xenograft model, increased osteoprotegerin expression correlated with a delay to pathologic fracture suggesting a potential role for mTOR inhibitors in the treatment of neuroblastoma bone metastases. 23990207_These results indicate that OPG derived either from the bone marrow or from the vessel wall is sufficient to slow down lesion progression and vascular calcification independent of bone turnover. 24642764_Osteoprotegerin inhibits vascular calcification by regulating the procalcific effects of RANKL and Il6 on vascular smooth muscle cells. 24998520_Osteoprotegerin is increased in metabolic syndrome and promotes adipose tissue proinflammatory changes. 25846796_identify osteoprotegerin as a HIF target gene capable of directing osteoblast-mediated osteoclastogenesis to regulate bone homeostasis 26254339_the generation of Rag2GFP+ regulatory T cells is unaltered in Tnfrsf11b(-/-) mice 26329493_OPG expression in chondrocyte increases bone mass in the proximal metaphysis of tibiae through negative regulation of osteoclast formation. 26825871_findings demonstrate that mTORC1 activation-stimulated RANKL expression in B cells is sufficient to induce bone loss and osteoporosis. The study also established a link between mTORC1 and the RANKL/OPG axis via negative regulation of beta-catenin. 26947973_Data show that p38alpha mitogen-activated protein kinase (MAPK) in paired related homeobox 1 protein (Prx1+) bone marrow-derived mesenchymal stromal cells acts to preserve the bone by sustaining osteoprotegerin (OPG) production. 27460899_endocortical resorption is driven by reduced OPG rather than elevated RANKL expression 27541035_These data suggested that OPG played an important role in maintaining the homeostasis of articular cartilage of femoral head. 27697839_These discoveries strongly suggest that Heparan Sulfate plays an active role in regulating OPG-RANKL interaction and osteoclastogenesis. 27898403_OPG plays an important role in regulating bone marrow mesenchymal stem cells differentiation and bone marrow adipogenesis. 28070143_Diabetes significantly increased OPG and the OPG/TRAIL ratio expression in the aorta, while dyslipidemia was the major determinant of the changes observed in the heart, where it significantly increased OPG and reduced TRAIL expression, thus increasing cardiac OPG/TRAIL ratio. 28373003_RANK/RANKL signaling is involved in the androgen deprivation therapy-induced acceleration of bone metastasis in castration-insensitive prostate cancer and is inhibited by osteoprotegerin to prevent bone metastasis. 28840938_Hdac3-depletion in osteoblasts increases expression of Opg, subsequently preserving insulin sensitivity. 29382610_lactation increases physiological maxillary bone remodeling and orthodontic tooth movement, and both require activation of RANK/RANKL/OPG system 29415886_Removal of heparan sulfate HS from osteoblasts leads to severe osteopenia and this phenotype is not due to impairment in bone formation but due to enhancement in bone resorption resulting from dysfunction of osteoprotegerin (OPG). OPG associates with the surface of osteoblasts via interaction with HS and that, in this form, OPG efficiently inhibits RANKL/RANK signaling and osteoclastogenesis. 29644561_Epithelial rests of Malassez may be associated with root resorption via OPG, thus helping to explain the mechanism underlying root resorption. 30254073_intact dimerization interface is essential for the biological activity of OPG. 30422320_our findings highlight that PEMF with 5 G can inhibit cellular apoptosis, modulate cytoskeletal distribution, and decrease RANKL/OPG expression in osteocytes, and also inhibit osteocyte-mediated osteoclastogenesis, which requires the existence of primary cilia in osteocytes. 30623241_OPG is Required for the Postnatal Maintenance of Condylar Cartilage. 30626205_OPG is suppressed via NF-kappaB-dependent signaling mediated by ERK1/2. On ligand binding, TLR2 activates p38 and ERK1/2 signaling to selectively modulate the upregulation of IL-6-mediated RANKL and downregulation of OPG.These signaling pathways act in concert to induce chondrogenic transdifferentiation of VSMCs, which in turn leads to vascular calcification during the pathogenesis of atherosclerosis. 30626485_These data show that connexin43-dependent gap junctional communication among osteoblast cells permits efficient ERK1/2 activation. ERK1/2 signaling promotes the recruitment of the potent transcriptional activator, Sp1, to the osteoprotegerin proximal promoter, resulting in robust transcription of anti-osteoclastogenic factor, osteoprotegerin. 31081236_We hypothesize that RANKL can stimulate RANK on macrophages and initiate the process of ECM degradation. This process may be inhibited by highly expressed OPG in fibrotic conditions. In this case, RANKL mutants that can bind to RANK without binding to OPG might become promising therapeutic candidates 31318102_Oleanolic acid exerts inhibitory effects on the late stage of osteoclastogenesis and prevents bone loss in osteoprotegerin knockout mice. 31356686_MiR-146a Deletion Protects From Bone Loss in OVX Mice by Suppressing RANKL/OPG and M-CSF in Bone Microenvironment. 31696568_These results demonstrated that the AMPK signalling pathway functions as a critical regulator in the OPG-mediated inhibition of Osteoclasts differentiation, by inducing autophagy 31932605_Microfold cells (M cells) express osteoprotegerin (OPG), which suppresses the differentiation of adjacent follicle-associated epithelium (FAE) into M cells. OPG deficiency increases M cell number in the GALT and enhances commensal bacterium-specific immunoglobulin production, resulting in the amelioration of disease symptoms in mice with experimental colitis. OPG-deficient mice are susceptible to Salmonella infection. 31943048_Muscle weakness and selective muscle atrophy in osteoprotegerin-deficient mice. 31958462_Osteoprotegerin promotes intimal hyperplasia and contributes to in-stent restenosis: Role of an alphaVbeta3/FAK dependent YAP pathway. 32053272_Quercetin inhibits macrophage polarization through the p-38alpha/beta signalling pathway and regulates OPG/RANKL balance in a mouse skull model. 32307402_Effect of continuous compressive force on the expression of RANKL, OPG, and VEGF in osteocytes. 32315212_RANKL induces beige adipocyte differentiation in preadipocytes. 33141273_Effect of periostin silencing on Runx2, RANKL and OPG expression in osteoblasts.', trans 'Wirkung von Periostin-Silencing auf die Expression von Runx2, RANKL und OPG in Osteoblasten. 33277741_Overexpression of c-Fos reverses osteoprotegerin-mediated suppression of osteoclastogenesis by increasing the Beclin1-induced autophagy. 33615520_H19 Promotes HCC Bone Metastasis Through Reducing Osteoprotegerin Expression in a Protein Phosphatase 1 Catalytic Subunit Alpha/p38 Mitogen-Activated Protein Kinase-Dependent Manner and Sponging microRNA 200b-3p. 33633362_RANKL/OPG ratio regulates odontoclastogenesis in damaged dental pulp. 34854215_LncRNA SNHG1 modulates adipogenic differentiation of BMSCs by promoting DNMT1 mediated Opg hypermethylation via interacting with PTBP1. 34986499_Incretins Enhance PGF2alpha-Induced Synthesis of IL-6 and Osteoprotegerin in Osteoblasts. 35060690_Osteoprotegerin Expression in Liver is Induced by IL13 through TGFbeta. 35266010_Potential mechanisms of osteoprotegerin-induced damage to osteoclast adhesion structures via P2X7R-mediated MAPK signaling. 35691140_Genetic deletion of osteoprotegerin attenuates asthma development through suppression of inflammatory response in mice. |
ENSG00000164761 |
TNFRSF11B |
8576.944921 |
3.7426824165 |
1.904073 |
0.06849486 |
732.477461 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000002591148775457160880415376965551126895679053600781855047425709213572931501296323177152829425190596384902 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000232166930280961630047122570934111869717591453022276835411808252617159316775722329768512969397883901880 |
Yes |
No |
13488.8028689148 |
640.648760322559 |
3604.04687545726 |
129.799721316612 |
| ENSMUSG00000063810 |
236266 |
Alms1 |
protein_coding |
Q8K4E0 |
FUNCTION: Involved in PCM1-dependent intracellular transport. Required, directly or indirectly, for the localization of NCAPD2 to the proximal ends of centrioles. Required for proper formation and/or maintenance of primary cilia (PC), microtubule-based structures that protrude from the surface of epithelial cells (By similarity). {ECO:0000250}. |
Alternative splicing;Cell projection;Cilium;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:236266; |
centriole [GO:0005814]; centrosome [GO:0005813]; ciliary basal body [GO:0036064]; cytosol [GO:0005829]; spindle pole [GO:0000922]; alpha-actinin binding [GO:0051393]; microtubule binding [GO:0008017]; protein C-terminus binding [GO:0008022]; apoptotic process [GO:0006915]; calcium-mediated signaling [GO:0019722]; cellular glucose homeostasis [GO:0001678]; cholesterol homeostasis [GO:0042632]; cilium assembly [GO:0060271]; endosomal transport [GO:0016197]; epithelial cell apoptotic process [GO:1904019]; epithelial cell proliferation [GO:0050673]; establishment of planar polarity [GO:0001736]; glucose homeostasis [GO:0042593]; inner ear receptor cell stereocilium organization [GO:0060122]; lipid metabolic process [GO:0006629]; negative regulation of epithelial cell apoptotic process [GO:1904036]; negative regulation of multicellular organism growth [GO:0040015]; ovulation [GO:0030728]; positive regulation of cold-induced thermogenesis [GO:0120162]; positive regulation of endocytosis [GO:0045807]; regulation of centriole replication [GO:0046599]; regulation of fat cell differentiation [GO:0045598]; regulation of stress fiber assembly [GO:0051492]; retinal rod cell development [GO:0046548]; sensory perception of sound [GO:0007605]; spermatid development [GO:0007286] |
16000322_Disruption in ALMS1 manifests human Alstrom syndrome 16516152_In this model of metabolic syndrome, transition of steatosis to steatohepatitis was associated with hypoadiponectinemia, a mediator of hepatic fatty acid disposal pathways. 17206865_In vitro knockdown of Alms1 in mice causes stunted cilia on kidney epithelial cells and prevents these cells from increasing calcium influx in response to mechanical stimuli. 21071598_investigated the role of ALMS1 in the cochlea and the pathogenesis of hearing loss in Alstrom Syndrome 22581473_This study demonitrated that Alms1 localizes at the base of cilia in hypothalamic neurons and Alms1 is lost from this location in foz/foz mice. 22693585_a role for ALMS1 variants in the recycling endosome pathway 24595103_Data conclude that deficiency of Alstrom protein impairs postnatal cardiomyocyte cell cycle arrest. 25299671_GLUT4 defects in adipose tissue are early signs of metabolic alterations in Alms1-deficient mice, a mouse model for obesity and insulin resistance. 34147365_LHCGR and ALMS1 defects likely cooperate in the development of polycystic ovary syndrome indicated by double-mutant mice. |
ENSG00000116127 |
ALMS1 |
581.689131 |
2.1261266874 |
1.088228 |
0.12481998 |
75.501588 |
0.00000000000000000365113104914891101542823707399058525749456175312867949045436688493282417766749858856201171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000001241384556710629822282798380646233109787268699460843768278550669492688030004501342773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
801.767495648495 |
69.4543884353994 |
377.102408987533 |
25.0939557007801 |
| ENSMUSG00000063975 |
108096 |
Slco1a5 |
protein_coding |
A0A0G2JDD1 |
Human_homologues FUNCTION: Na(+)-independent transporter that mediates the cellular uptake of a broad range of organic anions such as the endogenous bile salts cholate and deoxycholate, either in their unconjugated or conjugated forms (taurocholate and glycocholate), at the plasmam membrane (PubMed:7557095, PubMed:19129463). Responsible for intestinal absorption of bile acids (By similarity). Transports dehydroepiandrosterone 3-sulfate (DHEAS), a major circulating steroid secreted by the adrenal cortex, as well as estrone 3-sulfate and 17beta-estradiol 17-O-(beta-D-glucuronate) (PubMed:9539145, PubMed:23918469, PubMed:25560245, PubMed:12568656, PubMed:11159893, PubMed:19129463). Mediates apical uptake of all-trans-retinol (atROL) across human retinal pigment epithelium, which is essential to maintaining the integrity of the visual cycle and thus vision (PubMed:25560245). Involved in the uptake of clinically used drugs (PubMed:17301733, PubMed:20686826, PubMed:27777271). Capable of thyroid hormone transport (both T3 or 3,3',5'-triiodo-L-thyronine, and T4 or L-tyroxine) (PubMed:20358049, PubMed:19129463). Also transports prostaglandin E2 (PubMed:19129463). Plays roles in blood-brain and -cerebrospinal fluid barrier transport of organic anions and signal mediators, and in hormone uptake by neural cells (By similarity). May also play a role in the reuptake of neuropeptides such as substance P/TAC1 and vasoactive intestinal peptide/VIP released from retinal neurons (PubMed:25132355). May play an important role in plasma and tissue distribution of the structurally diverse chemotherapeutic drugs methotrexate and paclitaxel (PubMed:23243220). Shows a pH-sensitive substrate specificity which may be ascribed to the protonation state of the binding site and leads to a stimulation of substrate transport in an acidic microenvironment (PubMed:19129463). Hydrogencarbonate/HCO3(-) acts as the probable counteranion that exchanges for organic anions (PubMed:19129463). May contribute to regulate the transport of organic compounds in testis across the blood-testis-barrier (Probable). {ECO:0000250|UniProtKB:O88397, ECO:0000250|UniProtKB:Q91YY5, ECO:0000269|PubMed:11159893, ECO:0000269|PubMed:12568656, ECO:0000269|PubMed:17301733, ECO:0000269|PubMed:19129463, ECO:0000269|PubMed:20358049, ECO:0000269|PubMed:20686826, ECO:0000269|PubMed:23243220, ECO:0000269|PubMed:23918469, ECO:0000269|PubMed:25132355, ECO:0000269|PubMed:25560245, ECO:0000269|PubMed:27777271, ECO:0000269|PubMed:7557095, ECO:0000269|PubMed:9539145, ECO:0000305|PubMed:35307651}. |
Coiled coil;Ion transport;Membrane;Reference proteome;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:108096; |
plasma membrane [GO:0005886]; transmembrane transporter activity [GO:0022857]; ion transport [GO:0006811] |
21084765_Immunoreactivity to anti-Oatp1a antibody was colocalized with PDZK1 in the small intestine of wild-type mice, whereas apical localization of Oatp1a protein was reduced in pdzk1(-/-) mice. PDZK1 appears to act as an adaptor for Oatp1a[Oatp1a] |
ENSG00000084453 |
SLCO1A2 |
37.172700 |
0.2115036598 |
-2.241245 |
0.44610358 |
25.153754 |
0.00000052936501561197929658584551346200974819566908990964293479919433593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000049437694161874119852767388294534356418807874433696269989013671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.5940584366905 |
3.95663269901381 |
59.5453452002892 |
12.4412311149589 |
| ENSMUSG00000064032 |
|
Gm10143 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
64.789796 |
2.0465902274 |
1.033222 |
0.39052139 |
6.897225 |
0.00863296546832175923369412373631348600611090660095214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0296031810025837553712602101541051524691283702850341796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
89.1221037553105 |
24.4200450902198 |
43.5466282226011 |
9.078940359533 |
| ENSMUSG00000064080 |
14115 |
Fbln2 |
protein_coding |
P37889 |
FUNCTION: Its binding to fibronectin and some other ligands is calcium dependent. May act as an adapter that mediates the interaction between FBN1 and ELN. {ECO:0000250|UniProtKB:P98095}. |
Alternative splicing;Calcium;Direct protein sequencing;Disulfide bond;EGF-like domain;Extracellular matrix;Glycoprotein;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:14115; |
collagen-containing extracellular matrix [GO:0062023]; extracellular region [GO:0005576]; calcium ion binding [GO:0005509]; extracellular matrix binding [GO:0050840]; positive regulation of cell-substrate adhesion [GO:0010811] |
11733994_short arm region of laminin-5 gamma2 chain: structure, mechanism of processing and binding to heparin, nidogen-1, fibulin-1 and fibulin-2 15610515_The alterations in elastogenesis associated with increased fibulin-2 in hypodermal connective tissues and decreased fibulin-5 at the hypodermal M-CT interface suggest that these proteins mediate the effect of Tsk-fibrillin-1 on elastogenesis. 18070922_fibulin-2 is not required for mouse development and elastic fiber formation and suggest possible functional redundancy between fibulin-1 and fibulin-2. 19893004_Fibulin-2 and fibulin-5 cooperatively function to form the internal elastic lamina during postnatal development by directing the assembly of elastic fibers, and are responsible for maintenance of the adult vessel wall after injury 22100229_absence of fibulin-2 results in decreased up-regulation of TGF-beta signaling after MI and protects against ventricular dysfunction. 23785517_Data conclude that fibulin-2 is a driver of malignant progression in lung adenocarcinoma and plays an unexpected role in collagen cross-linking and tumor cell adherence to collagen. 23841699_Fibulin-2 is essential for angiotensin II-induced transforming growth factor-beta-mediated cardiac hypertrophy via enhanced TGF-beta activation. 24390135_integrin alpha3beta1 has a role in basement membrane stability through fibulin-2 induction, both in neonatal skin and in adult wounds 24522256_FBLN2 and VCAN were both preferentially expressed in the mammary gland during times of epithelial expansion in TEB-enriched tissue and co-localised specifically in cap cell. 24692557_levels of fibulin 2 dramatically increased in the retinal pigment epithelium following retinal detachment, suggesting a direct role for fibulin 2 in the re-attachment of the retina to the retinal pigment epithelium. 24694902_These findings demonstrate a prominent role for alpha3beta1 in immortalized/transformed keratinocytes in regulating fibulin-2 and other genes that promote matrix remodeling and invasion. 27111286_Our study indicates that Ang II cannot induce TGF-beta activation without fibulin-2 and that fibulin-2 has an essential role in Ang II-induced TGF-beta signaling and subsequent myocardial fibrosis. Fibulin-2 can be considered as a critical regulator of TGF-beta that induces myocardial fibrosis. 29721953_Fibulin 2 localized to the RPE, choroid, and Bruch's membrane. Knockout mice had a significantly reduced scotopic A response by 1 month of age, when compared to their wild-type counterparts, which remained throughout life and only declined as a result of normal aging. The functional decline was associated with reduced number of photoreceptor cells. 30118321_High FBLN2 expression is associated with cognitive impairment. 34769264_Fibulin 2 Is Hypermethylated and Suppresses Tumor Cell Proliferation through Inhibition of Cell Adhesion and Extracellular Matrix Genes in Non-Small Cell Lung Cancer. |
ENSG00000163520 |
FBLN2 |
7769.629694 |
3.3200495151 |
1.731205 |
0.09287461 |
330.749186 |
0.00000000000000000000000000000000000000000000000000000000000000000000000006601069359619066710442066005303749527748020862411083456956007630740531307834172564435118546314767617669998065635632944982249204129185312407330403869645416845558066134145686863359024990072221 |
0.0000000000000000000000000000000000000000000000000000000000000000000000223438863301594696614223838771684651604685404745865678654040125459188224381267486627405934060417826400560257859552827685505217862182796115383301241652500483699472626652534495723251284360344 |
Yes |
No |
11899.0430840923 |
766.539309270327 |
3583.99566934543 |
173.928746065507 |
| ENSMUSG00000066058 |
242653 |
Cldn19 |
protein_coding |
Q9ET38 |
FUNCTION: Plays a major role in tight junction-specific obliteration of the intercellular space, through calcium-independent cell-adhesion activity. {ECO:0000305|PubMed:25678664}. |
3D-structure;Alternative splicing;Cell junction;Cell membrane;Disulfide bond;Membrane;Reference proteome;Tight junction;Transmembrane;Transmembrane helix |
|
This gene encodes a member of the claudin family. Claudins are integral membrane proteins and components of tight junction strands. Tight junction strands serve as a physical barrier to prevent solutes and water from passing freely through the paracellular space between epithelial or endothelial cell sheets, and also play critical roles in maintaining cell polarity and signal transductions. siRNA knockdown of this gene in mice develops the FHHNC (familial hypomagnesemia with hypercalciuria and nephrocalcinosis) symptoms of chronic renal wasting of magnesium and calcium together with defective renal salt handling. The protein encoded by this gene interacts with another family member, Claudin 16, and their interaction is required for their assembly into tight junctions and for renal reabsorption of magnesium. This protein is a constituent of tight junctions in the Schwann cells of peripheral myelinated nerves and the gene deficiency affects the nerve conduction of peripheral myelinated fibers. Alternatively spliced transcript variants encoding different isoforms have been found for this gene. [provided by RefSeq, Aug 2010]. |
mmu:242653; |
apical junction complex [GO:0043296]; basolateral plasma membrane [GO:0016323]; bicellular tight junction [GO:0005923]; cell junction [GO:0030054]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; structural molecule activity [GO:0005198]; actin cytoskeleton reorganization [GO:0031532]; apical junction assembly [GO:0043297]; bicellular tight junction assembly [GO:0070830]; calcium-independent cell-cell adhesion via plasma membrane cell-adhesion molecules [GO:0016338]; cell adhesion [GO:0007155]; negative regulation of cell migration [GO:0030336]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of gene expression [GO:0010629]; negative regulation of wound healing [GO:0061045]; neuronal action potential propagation [GO:0019227]; positive regulation of cell junction assembly [GO:1901890]; positive regulation of gene expression [GO:0010628]; positive regulation of protein phosphorylation [GO:0001934]; regulation of transepithelial transport [GO:0150111]; tight junction organization [GO:0120193] |
15589828_Sp1 site is crucial for Cldn19 gene expression in kidney cells. 19706394_Claudin-16 and claudin-19 interaction is required for their assembly into tight junctions and for renal reabsorption of magnesium. 25678664_this study presents the structure of mammalian claudin-19 in complex with C-CPE at 3.7 A resolution. |
ENSG00000164007 |
CLDN19 |
35.156114 |
7.4630028407 |
2.899756 |
0.77564326 |
12.585182 |
0.00038881717298972141199156493662769662478240206837654113769531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0019982983385382862943913107045545984874479472637176513671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
60.9252708072466 |
34.3565241131929 |
8.16364030773155 |
3.77909153547834 |
| ENSMUSG00000066113 |
77739 |
Adamtsl1 |
protein_coding |
Q8BLI0 |
|
Alternative splicing;Disulfide bond;Extracellular matrix;Glycoprotein;Immunoglobulin domain;Reference proteome;Repeat;Secreted;Signal |
|
|
mmu:77739; |
collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular region [GO:0005576]; extracellular matrix organization [GO:0030198] |
Human_homologues 11805097_Punctin, a novel ADAMTS-like molecule, ADAMTSL-1, in extracellular matrix 17395588_data define a critical role for N-glycosylation and O-fucosylation in the biosynthesis of punctin-1 19671700_thrombospondin type-1 repeats from punctin-1 carries C-mannosylation in close proximity to O-linked fucose 19824886_Observational study of gene-disease association, gene-gene interaction, and gene-environment interaction. (HuGE Navigator) 19913121_Observational study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) 28722276_These data imply a multisystem role for ADAMTSL1 and present the first disease-associated variant affecting a C-mannosylation motif. 29158497_ADAMTSL1 are associated with breast cancer prognosis and may involve an interaction with AREG expression. 30714143_This explains why the patients with ADAMTSL1 mutations had abnormal mandibles but normal long bones. This is the first report that mutations in ADAMTSL1 are responsible for the pathogenesis of mandibular prognathism. |
ENSG00000178031 |
ADAMTSL1 |
472.952505 |
0.2280918165 |
-2.132313 |
0.31515834 |
41.029093 |
0.00000000014997999364956412414973071637106210446388487866897776257246732711791992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000023358847272701027471969442158390359298714145097619621083140373229980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
185.016484515071 |
40.7392812564406 |
811.149156356638 |
131.456286484771 |
| ENSMUSG00000066878 |
100043906 |
Gm10184 |
protein_coding |
P52293 |
FUNCTION: Functions in nuclear protein import as an adapter protein for nuclear receptor KPNB1. Binds specifically and directly to substrates containing either a simple or bipartite NLS motif. Docking of the importin/substrate complex to the nuclear pore complex (NPC) is mediated by KPNB1 through binding to nucleoporin FxFG repeats and the complex is subsequently translocated through the pore by an energy requiring, Ran-dependent mechanism. At the nucleoplasmic side of the NPC, Ran binds to importin-beta and the three components separate and importin-alpha and -beta are re-exported from the nucleus to the cytoplasm where GTP hydrolysis releases Ran from importin. The directionality of nuclear import is thought to be conferred by an asymmetric distribution of the GTP- and GDP-bound forms of Ran between the cytoplasm and nucleus. |
3D-structure;Acetylation;Cytoplasm;Nucleus;Phosphoprotein;Protein transport;Reference proteome;Repeat;Transport |
|
|
mmu:16647; |
cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; glutamatergic synapse [GO:0098978]; host cell [GO:0043657]; NLS-dependent protein nuclear import complex [GO:0042564]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; postsynaptic density [GO:0014069]; DNA-binding transcription factor binding [GO:0140297]; histone deacetylase binding [GO:0042826]; nuclear import signal receptor activity [GO:0061608]; nuclear localization sequence binding [GO:0008139]; entry of viral genome into host nucleus through nuclear pore complex via importin [GO:0075506]; NLS-bearing protein import into nucleus [GO:0006607]; positive regulation of viral life cycle [GO:1903902]; postsynapse to nucleus signaling pathway [GO:0099527]; protein import into nucleus [GO:0006606] |
Human_homologues 11796712_Characterization of an unusual importin alpha binding motif in the borna disease virus p10 protein that directs nuclear import 11882654_Identification of a karyopherin alpha 2 recognition site in PLAG1, which functions as a nuclear localization signal 12551970_HPV16 E6 interacts with the karyopherin alpha2 adapter and can enter the nucleus of hela cells via a classical Kap alpha2beta1-mediated pathway 12883947_IPOA1 was localized at the lymphocyte plasma membrane. Its subcellular distribution was also studied. 12909615_confirmed the role of KPNA-2 in nuclear import of Chk2 15194443_Importin alpha/beta-mediated nuclear import machinery is regulated in a cell cycle-dependent manner through the modulation of interaction modes between importins alpha and beta. 15234975_interaction with importin system is required for thioredoxin-binding protein-2 nuclear translocation and growth control tightly associated with TRX-dependent redox regulation of transcription factors 15342649_Results point to importin alpha1 as a critical downstream target of AMP-activated protein kinase (AMPK) and key mediator of AMPK-triggered HuR nuclear import. 15731250_These observations indicate that importin-alpha functions as a mediator for the nuclear entry of Vpr. 15795315_The model reflects experimentally determined rates for cargo import and correctly predicts that import is limited principally by Impalpha and Ran, but is also sensitive to NTF2 16188882_an interaction with KPNA2 contributes to nuclear localization and multiple tumor suppression functions of the NBS1 complex 16552725_These data suggest the importance of receptor endocytosis, endosomal sorting machinery, interaction with importins alpha1/beta1, and exportin CRM1 in EGFR nuclear-cytoplasmic trafficking. 16752129_This review focuses on recent experimental evidences demonstrating how NBS1 is translocated into the nucleus by an importin KPNA2 which mediates NBS1 subcellular localization and the functions of the NBS1 complex in tumorigenesis. 16818692_The gene expression profiling of laser-microdissected breast cancer tissue revealed KPNA2 genes that may represent potential molecular targets for breast cancer therapy and prediction of outcome. 17070506_importin alpha1, an essential component of cytoplasmic-nuclear transport, is abnormally accumulated in Hirano bodies in vulnerable hippocampal neurons in AD. 17255955_KPNA2 may play an important role in the signal-transduction pathways that regulate epidermal proliferation and differentiation 17344301_interaction between importin alpha and the N-terminal alpha-helical domain of Vpr is indispensable, not only for the nuclear import of Vpr but also for HIV-1 replication in macrophages. 17537211_dengue virus nonstructural protein 5 nuclear localization through its importin alpha/beta-recognized nuclear localization sequences is integral to viral infection 17551513_The data, together with previous analyses of nuclear protein import, suggest that the use of adapters such as importin-alpha provides the cell with increased dynamic range for control of nuclear import rates, but at the expense of efficiency. 17596301_SARS-COV ORF6 protein is localized to the endoplasmic reticulum (ER)/Golgi membrane in infected cells, where it binds to and disrupts nuclear import complex formation by tethering karyopherin alpha 2 and karyopherin beta 1 to the membrane. 17899179_Expression of KPNA2 in invasive breast cancer correlates with conventional prognostic parameters & shorter disease-free survival. KPNA2 is overexpressed in DCIS, which emphasizes its potential role in carcinogenesis of invasive breast carcinomas. 18068677_The formation of high molecular mass complexes containing importin-alpha, Nup153 and Nup88 is increased upon oxidant treatment. 18561322_KPNA2 nuclear expression as novel prognostic marker in node-positive breast cancer patients 19103427_Oct4 nuclear localization may be mediated by its interaction with KPNA-2. 19117056_Crm1, Kpnbeta1 and Kpnalpha2 are overexpressed in cervical cancer and inhibiting the expression of Crm1 and Kpnbeta1, not Kpnalpha2, induces cancer cell death 19883659_The tripartite protein-RNA complex formation between Hexim, Cyclin T and 7SK snRNA, was analyzed. 19950226_Observational study of gene-disease association and gene-gene interaction. (HuGE Navigator) 19961612_nuclear import of HIV-1 integrase(IN) occurs via importin alpha pathway and promoted by a specific nuclear localization signal(NLS).Import could be blocked by NLS-IN peptide confirming that nuclear import of viral preintegration complex is mediated by IN. 20362631_Data reveal the involvement of importin alpha1 in the assembly of RNA granules and its pro-survival role during stress 20393006_KPNA2 expression is associated with poor differentiation, tumor invasiveness, and tumor proliferation in esophageal squamous cell carcinoma. 20658535_protein levels of KPNA2 in pleural effusion from NSCLC patients were also significantly higher than those from non-lung cancer. Moreover, knockdown of KPNA2 inhibited the migration ability and viability of lung cancer cells 20859152_Overexpression of KPNA2 in patients with epithelial ovarian cancer is positively associated with an epithelial ovarian cancer-specific histologic type and a poor prognosis. 21220479_KPNA2 expression was significantly upregulated in carcinomas of the prostate, especially in metastatic and castration-resistant prostate cancer samples; it is a novel independent prognostic marker for disease progression after radical prostatectomy 21286940_Overexpression of importin alpha1 is associated with hepatocellular carcinoma. 21326825_Transportin 3 and importin alpha act as receptors and are required for effective nuclear import of HIV-1 integrase in virus-infected cells. 21330047_KPNA2 expression is a marker for progression of non-muscle-invasive bladder cancer and a prognostic marker in patients undergoing radical cystectomy. 21418451_Taspase1 appears to exploit the nuclear export activity of importin-alpha/nucleophosmin to gain transient access to the cytoplasm required to also cleave its cytoplasmic substrates. 21454664_importin alpha interacted with the zinc finger domain of Snail to compete with the binding of importin beta1 and Snail did not form a ternary complex with importin alpha/importin beta1. 21690087_Crystallographic analysis of mammalian importin alpha1 in complex with the hPLSCR4-NLS reveals this minimal NLS binds specifically and exclusively to the minor binding site of importin alpha 21909132_This study gives clear evidence that KPNA2 acts as a novel oncogenic factor in human breast cancer, in vitro. 22125623_findings suggest that the deregulated activity of E2F in cancer cells causes increased activation of the Kpnbeta1 and Kpnalpha2 promoters, leading to elevated levels of these proteins, and ultimately impacting the cancer phenotype. 22275867_Importin alpha1 and alpha7 are positive regulators of human-like polymerase activity and pathogenicity beyond their role in nuclear transport. 22415091_Nuclear translocation of Wilms' tumour protein involves importins alpha and beta, and a nuclear localisation signal in the third zinc finger 22772608_KPNA2 expression may have potential as a novel diagnostic and prognostic biomarker for astrocytic gliomas. 22843992_The KPNA2-regulating protein profiles in an adenocarcinoma cell line, were analyzed. 22962582_Overexpression of karyopherin 2 (KPNA2) in human ovarian malignant germ cell tumor correlates with poor prognosis. 23283818_Overexpression of KPNA2 correlates with gastric adenocarcinoma. 23435424_IGFBP-2 possesses a functional nuclear localization signal sequence and IGFBP-2 actively translocates into the nucleus by a classical nuclear import mechanism, involving formation of IGFBP-2 complexes with importin-alpha. 23536776_This work defines an inflammatory signature shared by seven epithelial cancer types and KPNA2 as a consistently up-regulated protein in cancer. 23649804_Tpr import is mediated by the most abundant import receptor, KPNA2, which binds the bipartite NLS in Tpr with nanomolar affinity 23749771_Nuclear karyopherin-alpha2 expression in primary lesions and metastatic lymph nodes was associated with poor prognosis and progression in gastric cancer. 23887301_Identified a strong link between high KPNA2 expression and early PSA recurrence in prostate cancer patients treated with radical prostatectomy. 23907459_KPNA2 has an important role in promoting proliferation and tumorigenicity of epithelial ovarian carcinoma through upregulation of c-myc and suppression of FOXO3A. 24067372_SEPT9 isoform 1 is required for the association between HIF-1alpha and importin-alpha to promote efficient nuclear translocation. 24070213_Oct4 and KPNA2 play an important role in non-small-cell lung cancer progression. 24098495_Karyopherin alpha2 is essential for rRNA transcription and protein synthesis in proliferative keratinocytes. 24510842_High NBS1 expression was linked to biochemical recurrence in ERG-negative and PTEN non-deleted cancers, which was largely driven by high KPNA2 karyopherin alpha 2 expression. 24664371_study analyzed nuclear expression of karyopherin a2 in meningiomas; expression correlated with histological grade and proliferative activity; recurrent tumors expressed significantly higher levels of karyopherin a2 when compared to primary growths 24712655_This work extends published observations on SAMHD1 nuclear localization to a natural cell type for HIV-1 infection and identifies KPNA2/KPNB1 as cellular proteins important for SAMHD1 nuclear import. 24799195_The cellular apoptosis susceptibility/importin-alpha1 transport cycle is linked to X-linked inhibitor (XIAP) and is required to maintain tumor cell survival in hepatocellular carcinoma. 24799281_KPNA2 and CDK1 expression levels may discriminate between estrogen receptor positive patients of high and low risk of disease recurrence. 24814927_These findings demonstrate that rotavirus probably employs a novel strategy to inhibit interferon-induced STAT signalling, which acts after STAT1/STAT2 activation and binding to importin-alpha. 24930886_a significant correlation of KPNA2 expression and tumour aggressiveness in a large variety of other solid tumour entities 25031071_Aberrant expression of nuclear KPNA2 is correlated with early recurrence and poor prognosis in patients with small hepatocellular carcinoma after hepatectomy. 25060425_The nuclear import of PLAG1 by KPNA2 is essential for the role of KPNA2 in HCC cells. 25109899_KPNA2 was overexpressed in esophageal squamous cell carcinoma tissue and cell lines. Serum KPNA2 was higher in ESCC patients than in controls. KPNA2 knockdown inhibited cell proliferation and colony formation ability and induced a G2/M phase arrest. 25658636_Crystal structure of human importin-alpha1 (Rch1), revealing a potential autoinhibition mode involving homodimerization. 25728791_A novel role of karyopherin alpha 2 in cell migration through the regulation of vimentin-pErk protein complex levels in lung cancer. 25862856_OPN, SPINK1, GPC3 and KNPA2 were significantly over-expressed in HCC tissues. These genes may be useful in developing future biomarkers and therapeutic strategies for HCC 25956057_High KPNA2 immunoreactivity is a predictor of bladder recurrence and poor survival in patients with upper tract urothelial carcinoma treated with radical nephroureterectomy. 25989275_This study provides further evidence for the complexity of DDR mechanism in BC, and that KNPA2 has a role in the aberrant subcellular localisation of DDR proteins with subsequent impaired function. 26135850_Suggest that KPNA2 may play a key role in the inflammation process of rheumatoid arthritis via NF-kappaB P65 signal transduction pathway. 26209501_High KPNA2 expression is associated with osteoarthritis. 26491019_RBBP4 functions as a novel regulatory factor to increase the efficiency of importin alpha/beta-mediated nuclear import 26553592_provided support for a link between autophagy and epithelial-to-mesenchymal (-like) transition status in WT TP53 glioblastoma cells and provided evidence for the signaling pathway (MIR517C-KPNA2-cytoplasmic TP53) involved in attenuating autophagy 26626145_KPNA2 might play an important role in colorectal carcinogenesis and functions as a novel prognostic indicator and a potential therapeutic target for colorectal cancer. 26663089_High KPNA2 expression was found to be associated with poor prognosis and resistance to hyperthermochemoradiation therapy (HCRT). 26884852_The combination of low nuclear and cytoplasmic KPNA2 expression is associated with adverse outcome in head and neck squamous cell carcinoma treated with radio(chemo)therapy. 26887791_anti-importin alpha1 antibody treatment suppressed the importin alpha1-FGF1 complex formation and ERK1/2 activation, resulting in decreased cell growth. This study provides novel evidence that functional importin alpha1 is located at the cell surface, where it accelerates the proliferation of cancer cells. 27009856_our results show for the first time that KPNA2 is transcriptionally and post-translationally regulated by the mTOR pathway and provide new insights into targeted therapy for non-small cell lung cancer 27078844_miR-26b plays an anti-metastatic role and is downregulated in gastric cancer tissues via the KPNA2/c-jun pathway 27107455_Results demonstrated that radiation-induced dying colorectal cancer cells released considerable amounts of KPNA2 that induce the maturation and activation of DCs for synergistic antitumor effect of radiation. 27109484_p53/p21Cip1/Waf1/p16INK4a may be an important pathway involved in the function of KPNA2 in tongue squamous cell carcinoma (TSCC) CAL-27 cells. 27154959_These results suggest that the selected aptamers (76 and 72) warrant further study to explore not only their application in cancer diagnosis but also their use as a specific reagent to potentially block KPNA2-dependent nuclear transport of macromolecules across the nuclear membrane. 27328359_The authors identified Importin-alpha1 to bind to Coxiella burnetii AnkG and concluded that binding of AnkG to p32 and Importin-alpha1 is essential for its migration into the nucleus. 27611951_we confirmed that up-regulated KPNA2 and OCT4 expression is a common feature of bladder cancer that is correlated with increased aggressive tumor behavior. Also, we propose that KPNA2 regulates the process of OCT4 nuclear transportation in bladder cancer. 27974678_KPNA2 overexpression was associated with poor OS in East-Asian patients and European patients, as well as patients with gastric and colorectal cancer.[meta-analysis] 28166455_We have therefore modeled the structure of the complex of the whole Oct4 POU domain and importin alpha2 using protein-protein docking and molecular dynamics. The model explains how the Ebola virus VP24 protein has a negative effect on the nuclear import of STAT1 by importin alpha but not on Oct4, and how Nup 50 facilitates cargo release from importin alpha 28178675_KPNA2 expression may be a useful prognostic and predictive marker of gemcitabine sensitivity and survival. 28240251_Development of a pipeline for automated, high-throughput analysis of PSPC1 paraspeckle protein isoforms reveals specific roles for KPNA2, KPNA4 and KPNA6 proteins. 28383161_the crystal structure of the nuclear import adaptor importin-alpha1 bound to the nuclear localization signal (NLS) of EBNA-LP that shows EBNA-LP residues 44-RRVRRR-49 binding to the major NLS-binding site at the P0-P5 positions. 28422734_KPNA2 was an essential factor promoting hepatocellular carcinoma and unraveled potential molecular pathways and networks underlying KPNA2-induced hepatocellular carcinogenesis. 28651492_High serum karyopherin alpha 2 levels (>485 microg/mL) correlated with International Federation of Gynecology and Obstetrics stage ( p |
ENSG00000182481 |
KPNA2 |
31.635340 |
0.0029440816 |
-8.407967 |
1.32662911 |
56.606419 |
0.00000000000005323777458048672920635947651787541392290814198928750045070046326145529747009277343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000012323978456078630509787878258986951532372167150697350734844803810119628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.178542682464184 |
0.188314395680588 |
60.6446114030841 |
20.621848425623 |
| ENSMUSG00000067017 |
|
Capza1-ps1 |
pseudogene |
|
|
|
|
|
|
|
|
|
|
24.390038 |
0.1976297623 |
-2.339128 |
0.72641356 |
9.585004 |
0.00196172969528623109686238379367750894743949174880981445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0083700466998879192309646057879035652149468660354614257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
8.0917291407543 |
4.24862874128178 |
40.943879342643 |
14.7751342956236 |
| ENSMUSG00000067101 |
|
1700010H22Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
415.960557 |
0.4179386349 |
-1.258637 |
0.13873173 |
81.441312 |
0.00000000000000000018054416254877416500345532892666522277672967557829610115924245938856529392069205641746520996093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000068239421437789779018088218164867704072352268684518907665426112885143083985894918441772460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
242.41504909574 |
23.3469393834803 |
580.025460328779 |
40.1616520271292 |
| ENSMUSG00000067276 |
12338 |
Capn6 |
protein_coding |
O35646 |
FUNCTION: Microtubule-stabilizing protein that may be involved in the regulation of microtubule dynamics and cytoskeletal organization. May act as a regulator of RAC1 activity through interaction with ARHGEF2 to control lamellipodial formation and cell mobility. Does not seem to have protease activity as it has lost the active site residues. {ECO:0000269|PubMed:17210638, ECO:0000269|PubMed:21406564}. |
Cytoplasm;Cytoskeleton;Microtubule;Reference proteome |
|
|
mmu:12338; |
cytoplasm [GO:0005737]; microtubule [GO:0005874]; perinuclear region of cytoplasm [GO:0048471]; spindle microtubule [GO:0005876]; microtubule binding [GO:0008017]; microtubule bundle formation [GO:0001578]; regulation of cytoskeleton organization [GO:0051493] |
17210638_Capn6 is a microtubule-stabilizing protein expressed in embryonic tissues that may be involved in the regulation of microtubule dynamics and cytoskeletal organization. 20814968_Capn6 promotes cytoskeletal organization and microtubule stability in osteoclasts. 21406564_CAPN6 acts as a regulator of Rac1 and cell motility through interaction with GEF-H1. 23935533_These results collectively showed that a loss of CAPN6 promotes skeletal muscle differentiation during both development and regeneration, suggesting a novel physiological function of CAPN6 as a suppressor of skeletal muscle differentiation. 27525442_CAPN6 promotes atherogenicity in inflamed macrophages by disturbing CWC22/EJC systems. 31619317_Calpain-6 deficiency reduced the percentage of ciliated cells and impaired sonic hedgehog signaling. It has been speculated that this defect might be associated with decreased levels of alpha-tubulin acetylation at lysine 40. |
ENSG00000077274 |
CAPN6 |
9620.006495 |
2.6257339632 |
1.392721 |
0.04660350 |
868.769382 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000006041085194747170112594943758180236800817595438533416501917506635093760830 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000707829305279914522912813988725790318325750668033263997810228744047687925 |
Yes |
No |
13974.4367689572 |
451.495891899572 |
5322.10687176172 |
130.77970671868 |
| ENSMUSG00000067878 |
320923 |
Map7d3 |
protein_coding |
A2AEY4 |
FUNCTION: Promotes the assembly and stability of microtubules. {ECO:0000250|UniProtKB:Q8IWC1}. |
Alternative splicing;Coiled coil;Cytoplasm;Cytoskeleton;Phosphoprotein;Reference proteome |
|
|
mmu:320923; |
cytoplasm [GO:0005737]; microtubule cytoskeleton [GO:0015630]; spindle [GO:0005819]; microtubule binding [GO:0008017]; tubulin binding [GO:0015631]; microtubule cytoskeleton organization [GO:0000226]; microtubule polymerization [GO:0046785] |
32376790_Human X chromosome exome sequencing identifies BCORL1 as contributor to spermatogenesis. |
ENSG00000129680 |
MAP7D3 |
6.228169 |
0.0152414461 |
-6.035856 |
1.68588505 |
12.254492 |
0.00046413941541596055121582375946331922023091465234756469726562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0023440887187055406415403524533758172765374183654785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.175341643528004 |
0.254820936266419 |
11.5042655660926 |
8.04663869725959 |
| ENSMUSG00000067916 |
666532 |
Zfp991 |
protein_coding |
B1AVX1 |
Human_homologues FUNCTION: May be involved in transcriptional regulation. |
Reference proteome |
|
|
|
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of transcription by RNA polymerase II [GO:0006357] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 26823146_genes encoding transcription factors expressed in the brain might be an important group of major depressive disorder candidate genes. |
ENSG00000196378 |
ZNF34 |
16.293796 |
176.5960639741 |
7.464309 |
1.38350573 |
35.396755 |
0.00000000268934532472835738365705034451803162598437779706728179007768630981445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000356209634663150796868371923253188127134194473910611122846603393554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.5883878181585 |
36.2664698052262 |
0.184536320259878 |
0.19029220811492 |
| ENSMUSG00000068227 |
16185 |
Il2rb |
protein_coding |
P16297 |
FUNCTION: Receptor for interleukin-2. This beta subunit is involved in receptor mediated endocytosis and transduces the mitogenic signals of IL2. Probably in association with IL15RA, involved in the stimulation of neutrophil phagocytosis by IL15 (By similarity). {ECO:0000250|UniProtKB:P14784}. |
3D-structure;Cell membrane;Disulfide bond;Glycoprotein;Membrane;Receptor;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
The interleukin 2 receptor is composed of alpha and beta subunits. The beta subunit encoded by this gene is very homologous to the human beta subunit and also shows structural similarity to other cytokine receptors. [provided by RefSeq, Jul 2008]. |
mmu:16185; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; plasma membrane [GO:0005886]; cytokine receptor activity [GO:0004896]; interleukin-15 receptor activity [GO:0042010]; interleukin-2 binding [GO:0019976]; interleukin-2 receptor activity [GO:0004911]; cytokine-mediated signaling pathway [GO:0019221]; interleukin-15-mediated signaling pathway [GO:0035723]; interleukin-2-mediated signaling pathway [GO:0038110]; natural killer cell activation [GO:0030101]; negative regulation of apoptotic process [GO:0043066]; positive regulation of phagocytosis [GO:0050766] |
11864627_Knockout mice exhibited deficit in prepulse inhibition of acoustic startle reflex, significant reductions in acoustic startle reactivity, and modest differences in behavior in elevated plus-maze test. (IL-2/15 receptor-beta) 12196288_CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rbeta-deficient mice: implications for the nonredundant function of IL-2 12231477_role of the interleukin-2 receptor beta chain region between the two Stat5 docking sites in induction of interleukin-2 receptor alpha expression by interleukin-2 15528339_Depletion of IL-2 in vivo induces memory CD8+ T cell division by an IL-15-independent but by an IL-2/15 receptor beta-dependent mechanism, implying a novel process for antigen-independent homeostasis of memory CD8+ T cells in vivo. 15752766_This study for the first time delineates the molecular mechanisms underlying regulation of mouse IL-2Rbeta gene transcription by Ets family proteins, partially with Egr-1, and thereby further elucidates the molecular basis of lymphocyte activation. 16127247_The multivesicular regions were present in the cap structure of these granules, suggesting that the uNK cells of the Tg2Rbeta mice had cytotoxic activity. 16920913_these cells make up a specialized subset of central memory T cells with distinguishable phenotypic characteristics, most notably the higher expression of CD122 and CD127 17182565_STAT5 binds to the promoter of the foxp3 gene suggesting that IL-2Rbeta-dependent STAT5 activation promotes Treg differentiation by regulating expression of foxp3. 17559173_a signal mediated by IL-2Rbeta is essential for the development and homeostasis of Foxp3(+) Treg in vivo 18641329_local blockade of the beta-chain of the IL-2R restored an immunosuppressive cytokine milieu in the lung that ameliorated both inflammation and airway hyperresponsiveness in experimental allergic asthma 19050240_Common gamma-chain binding-dependent activation of the shared IL-15/IL-2 receptor beta/common gamma signaling pathway may play an important role in the activation of natural killer (NK) cells and CD8-positive T cells, resulting in tumor growth inhibition. 19139102_IL-9Ralpha and IL-2Rbeta homodimers efficiently mediate constitutive activation of ALL-associated JAK1 mutants. 19185518_subset of IL-2-dependent targets is indexed to a low IL-2R signaling threshold 19265164_A population of memory CD8 T cells generated under sterile inflammatory conditions is identified by intermediate expression levels of CD122/CD44 and is involved in recall contact hypersensitivity reactions responsible for allergic contact dermatitis. 19293560_IL2R beta-chain induces growth potential for glandular epithelial cells and an immune-privileged condition mediated by CD25+regulatory-T cells. 19329337_Results indicate that potential nuclear function of cleaved interleukin-2 receptor beta-chain and IL-15 receptor alpha-chain subunits is not plausible. 21541329_Continuous activation of the CD122/STAT-5 signaling pathway characterize regulatory lineage differentiation in the murine thymus. 22522341_This study demonstrated that IL-12 signaling plays a complex role during the induction of tolerance as the presence of exogenous IL-12 and the lack of IL-12 signaling both prevent complete induction of tolerance. 23018460_CDCD11c+B220+CD122+ cells play an important role in xenograft rejection. Their absence in NOG mice may be critical in supporting the successful engraftment of xenotransplants. 23315074_Thymic development of CD4+Foxp3+ regulatory T cells (Tregs) is highly dysregulated in IL-2 receptor (2Rbeta)-deficient mice. 25527788_IL-2Rbeta-dependent signaling and CD103 functionally cooperate to maintain tolerance in the gut mucosa. 26569069_Intratesticular inoculation with donor antigens promotes long-term skin allograft survival induced by conventional costimulatory blockade via the induction of both CD8 + CD122+ and CD4 + CD25+ Treg cells. 27573835_Genetic ablation of the IL2Rbeta chain on CD8(+) T cells restrains inhibitory receptor induction, in particular 2B4 and Tim-3; precludes terminal differentiation of highly defective PD-1(hi) effectors; and rescues memory T-cell development and responsiveness to IL-7-dependent signals. Together, we ascribe a previously unexpected role to IL-2 and IL-15 as instigators of CD8(+) T-cell exhaustion during chronic viral infe... 27799311_CBP physically associates with the IL-2 receptor beta-chain. CBP, found in the nucleus in resting CTLL-2 cells, relocates to the cytoplasm after IL-2 stimulation in an MEK/ERK pathway-dependent manner. Thus, IL-2-mediated acetylation plays an important role in the modulation of cytokine signaling and T cell fate. 28111009_Renal lesions are indeed modulated by intrinsic glomerular cells through the gamma-C/IL-2-beta receptor response, to date classically described only in immune cells. 28652399_c-REL, but not IkappaBNS, controlled the generation of classical CD25(+)Foxp3(-) precursors via direct binding to the Cd25 locus; propose that CD4(+)GITR(+)CD122(+)CD25(-)Foxp3(-) cells represent a Treg pre-precursor population, whose transition into Treg precursors is mediated via c-REL 28678791_Results indicate that conjugated-interleukin-2 (IL2) protein derived from NKTR-214 occupy interleukin-2 (IL2) receptor complex (IL2Ralphabetagamma) to a greater extent compared to free-IL2 protein. 29259099_The larger pool of IL-2Rbeta chains in CD8+ T cells is required to sustain IL-2 signaling and contributes to the quantitatively greater proliferative response to IL-2 relative to that of CD4+ mouse primary T cell cultures. 29259102_Diabetes was accelerated in male and female NOD mice in which IL-2Rbeta signaling was modestly and selectively reduced in T cells. 30222140_Combined costimulatory and CD122 blockade improved survival of transplanted tissue in mice and nonhuman primates by controlling proliferation and effector function of CD8+ T cells. 32393513_The Abundance and Availability of Cytokine Receptor IL-2Rbeta (CD122) Constrain the Lymphopenia-Induced Homeostatic Proliferation of Naive CD4 T Cells. 33185980_Chronic shift-lag promotes NK cell ageing and impairs immunosurveillance in mice by decreasing the expression of CD122. 33954185_Downregulation of miR-497-5p Improves Sepsis-Induced Acute Lung Injury by Targeting IL2RB. |
ENSG00000100385 |
IL2RB |
81.443877 |
3.1082995021 |
1.636126 |
0.29635399 |
30.095313 |
0.00000004113241576026947377785656051289719936647770737181417644023895263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000004540064904785685327859724291083010072611614305060356855392456054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
118.676190340245 |
24.5499046847239 |
38.1804231731883 |
6.31065878798658 |
| ENSMUSG00000068231 |
113847 |
Vmn1r43 |
protein_coding |
Q8VIC9 |
FUNCTION: Putative pheromone receptor implicated in the regulation of social and reproductive behavior. {ECO:0000269|PubMed:12214233}. |
Cell membrane;Disulfide bond;G-protein coupled receptor;Glycoprotein;Membrane;Pheromone response;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:113847; |
plasma membrane [GO:0005886]; pheromone receptor activity [GO:0016503]; response to pheromone [GO:0019236]; sensory perception of chemical stimulus [GO:0007606] |
|
ENSG00000197617 |
VN1R5 |
13.791056 |
0.0996735130 |
-3.326646 |
1.09659446 |
7.939682 |
0.00483623351491744429830887952448392752557992935180664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0180774254967417218054048788644649903289973735809326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.55169481915674 |
1.29634321357054 |
15.567774957941 |
8.13363350708468 |
| ENSMUSG00000068327 |
21909 |
Tlx2 |
protein_coding |
Q61663 |
FUNCTION: Transcription activator that binds DNA elements with the consensus sequence 5'-CGGTAATTGG-3'. Binds DNA via its homeobox. Required for normal cell death of enteric neurons in the gastrointestinal tract. Required for normal development of the enteric nervous system, and for proper development of normal motility of the gastrointestinal tract. {ECO:0000269|PubMed:10869550, ECO:0000269|PubMed:17560225, ECO:0000269|PubMed:19697050}. |
Activator;Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:21909; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity [GO:0003700]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; sequence-specific double-stranded DNA binding [GO:1990837]; animal organ development [GO:0048513]; enteric nervous system development [GO:0048484]; mesoderm formation [GO:0001707]; negative regulation of dendrite morphogenesis [GO:0050774]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
14598259_Expression of the transgene by enteric neurons and penetrance of pseudo-obstruction in Hox11L1-null animals are influenced by one or more modifier genes, counterparts of which may play a similar role in human disease. 15803212_Intestinal dysmotility in Hox11L1-null animals may not be a primary neural disorder. 16291166_Alignment of sequences obtained from rat, human, and mouse indicates that putative regulatory elements of the Hox11L1 gene are conserved in rat, mice, and humans. 17035299_Our study suggests that NKCC1 acting in concert with NCX causes a perturbation of Ca(m)(2+) homeostasis and mitochondrial dysfunction and cell death following in vitro ischemia. 17560225_Ncx participates in normal developmental cell death of enteric neurons. Ncx may modulate, if not directly target, expression of the caspase-3 at the transcriptional level to participate in neuronal cell death in the enteric nervous system. 20682252_These results suggest that high subsarcolemmal Ca2+ is required to ensure synchronous triggering with short spark latency in the absence of NCX. |
ENSG00000115297 |
TLX2 |
19.858747 |
25.1871299111 |
4.654615 |
0.90336176 |
29.717777 |
0.00000004997443633922178763262709447375009563785397403989918529987335205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000005460621336578380955660517656613528458819928346201777458190917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
36.628419123434 |
24.463752735635 |
1.45425140747589 |
0.867628943861371 |
| ENSMUSG00000068587 |
232714 |
Mgam |
protein_coding |
Q6XK23 |
Human_homologues FUNCTION: Alpha-(1,4) exo-glucosidase involved in breakdown of dietary starch oligosaccharides in small intestine. Cleaves the non-reducing alpha-(1,4)-linked glucose residue in linear dextrins with retention of anomeric center stereochemistry (PubMed:12547908, PubMed:18356321, PubMed:27480812, PubMed:18036614, PubMed:22058037). Mainly hydrolyzes short length oligomaltoses having two to seven glucose residues (PubMed:12547908, PubMed:18356321, PubMed:27480812, PubMed:18036614, PubMed:22058037). Can cleave alpha-(1,2), alpha-(1,3) and alpha-(1,6) glycosidic linkages with lower efficiency, whereas beta glycosidic linkages are usually not hydrolyzed (PubMed:27480812). {ECO:0000269|PubMed:12547908, ECO:0000269|PubMed:18036614, ECO:0000269|PubMed:18356321, ECO:0000269|PubMed:22058037, ECO:0000269|PubMed:27480812}. |
Disulfide bond;Glycosidase;Hydrolase;Proteomics identification;Reference proteome |
Human_homologues PATHWAY: Carbohydrate degradation. {ECO:0000305|PubMed:18036614, ECO:0000305|PubMed:22058037, ECO:0000305|PubMed:27480812}. |
|
|
apical plasma membrane [GO:0016324]; alpha-1,4-glucosidase activity [GO:0004558]; amylase activity [GO:0016160]; carbohydrate binding [GO:0030246]; glucan 1,4-alpha-glucosidase activity [GO:0004339]; hydrolase activity, hydrolyzing O-glycosyl compounds [GO:0004553]; maltose alpha-glucosidase activity [GO:0032450]; dextrin catabolic process [GO:1901027]; maltose catabolic process [GO:0000025] |
17585022_The alpha-glucogenic activities of the jejunal mucosa with and without added recombinant pancreatic alpha-amylase, using a range of food starch substrates are reported. 19193815_mucosal alpha-glucogenic activity of Mgam plays a crucial role in the regulation of prandial glucose homeostasis 20425755_investigation of dietary control of Mgam expression in jejunum: high starch diet induces Mgam through mechanism involving increased acetylation of histones and increased binding of CREBBP, CDX2, and HNF1 to Mgam gene |
ENSG00000257335 |
MGAM |
195.034038 |
7.5091740371 |
2.908654 |
0.29511869 |
91.056537 |
0.00000000000000000000139625141030475201170245733985687561689239687082728826123998421004923642385620041750371456146240234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000600782527733389378259965855134605269369761269657920276041768170571799601020757108926773071289062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
339.542166141072 |
69.9423983772776 |
45.2169791863991 |
7.44086625298818 |
| ENSMUSG00000068744 |
56742 |
Psrc1 |
protein_coding |
Q9D0P7 |
FUNCTION: Required for normal progression through mitosis. Required for normal congress of chromosomes at the metaphase plate, and for normal rate of chromosomal segregation during anaphase. Plays a role in the regulation of mitotic spindle dynamics. Increases the rate of turnover of microtubules on metaphase spindles, and contributes to the generation of normal tension across sister kinetochores. Recruits KIF2A and ANKRD53 to the mitotic spindle and spindle poles. May participate in p53/TP53-regulated growth suppression (By similarity). {ECO:0000250|UniProtKB:Q6PGN9}. |
Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Mitosis;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:56742; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; microtubule cytoskeleton [GO:0015630]; midbody [GO:0030496]; nucleoplasm [GO:0005654]; spindle [GO:0005819]; spindle microtubule [GO:0005876]; spindle pole [GO:0000922]; microtubule binding [GO:0008017]; cell division [GO:0051301]; microtubule bundle formation [GO:0001578]; mitotic metaphase plate congression [GO:0007080]; negative regulation of cell growth [GO:0030308]; positive regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0045737]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of microtubule polymerization [GO:0031116]; regulation of mitotic spindle organization [GO:0060236] |
12082536_A p53 response element mediates transcriptional activation 12427559_molecular cloning, sequence analysis and gene expression 17310996_DDA3 is a novel microtubule-associated protein that binds to EB3, and results implicate DDA3 and EB3 in the beta-catenin-mediated growth signaling. 29378206_PSRC1 protects against the development of atherosclerosis and enhances the stability of plaques by modulating cholesterol transportation and inflammation in macrophages and the liver of apoE(-/-) mice. 31838625_findings reveal that PSRC1 may affect liver actin polymerization and cholesterol metabolism-related genes or pathways. 35613310_Deficiency of PSRC1 accelerates atherosclerosis by increasing TMAO production via manipulating gut microbiota and flavin monooxygenase 3. 35690006_Deficiency of proline/serine-rich coiled-coil protein 1 (PSRC1) accelerates trimethylamine N-oxide-induced atherosclerosis in ApoE(-/-) mice. |
ENSG00000134222 |
PSRC1 |
1730.838191 |
2.0074968146 |
1.005398 |
0.07271681 |
189.559833 |
0.00000000000000000000000000000000000000000039665283422922497512820899439901113200637135322614095425230058142621138805961070659939640313085495405535403764737956144870167918270453810691833496093750000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000525375301824309125521942450231136733786005478339203972571119330006710837686819462867766682494960819182648267822166587848187191411852836608886718750000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2320.43684206962 |
117.007551388109 |
1155.88569070066 |
44.7052224542638 |
| ENSMUSG00000068747 |
20661 |
Sort1 |
protein_coding |
Q6PHU5 |
FUNCTION: Functions as a sorting receptor in the Golgi compartment and as a clearance receptor on the cell surface. Required for protein transport from the Golgi apparatus to the lysosomes by a pathway that is independent of the mannose-6-phosphate receptor (M6PR). Lysosomal proteins bind specifically to the receptor in the Golgi apparatus and the resulting receptor-ligand complex is transported to an acidic prelysosomal compartment where the low pH mediates the dissociation of the complex. The receptor is then recycled back to the Golgi for another round of trafficking through its binding to the retromer. Also required for protein transport from the Golgi apparatus to the endosomes. Promotes neuronal apoptosis by mediating endocytosis of the proapoptotic precursor forms of BDNF (proBDNF) and NGFB (proNGFB). Also acts as a receptor for neurotensin. May promote mineralization of the extracellular matrix during osteogenic differentiation by scavenging extracellular LPL. Probably required in adipocytes for the formation of specialized storage vesicles containing the glucose transporter SLC2A4/GLUT4 (GLUT4 storage vesicles, or GSVs). These vesicles provide a stable pool of SLC2A4 and confer increased responsiveness to insulin. May also mediate transport from the endoplasmic reticulum to the Golgi. {ECO:0000269|PubMed:10594043, ECO:0000269|PubMed:15236332, ECO:0000269|PubMed:15236333, ECO:0000269|PubMed:15372498, ECO:0000269|PubMed:15992544, ECO:0000269|PubMed:19407813, ECO:0000269|PubMed:21102451}. |
3D-structure;Alternative splicing;Cell membrane;Cleavage on pair of basic residues;Developmental protein;Differentiation;Disulfide bond;Endocytosis;Endoplasmic reticulum;Endosome;Glycoprotein;Golgi apparatus;Lipoprotein;Lysosome;Membrane;Nucleus;Osteogenesis;Palmitate;Phosphoprotein;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:20661; |
cell surface [GO:0009986]; cerebellar climbing fiber to Purkinje cell synapse [GO:0150053]; clathrin-coated pit [GO:0005905]; clathrin-coated vesicle [GO:0030136]; cytoplasmic vesicle [GO:0031410]; cytoplasmic vesicle membrane [GO:0030659]; cytosol [GO:0005829]; dendrite [GO:0030425]; early endosome [GO:0005769]; endoplasmic reticulum membrane [GO:0005789]; endosome membrane [GO:0010008]; Golgi apparatus [GO:0005794]; Golgi cisterna membrane [GO:0032580]; intracellular membrane-bounded organelle [GO:0043231]; lysosomal membrane [GO:0005765]; lysosome [GO:0005764]; membrane [GO:0016020]; neuronal cell body [GO:0043025]; nuclear membrane [GO:0031965]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; trans-Golgi network transport vesicle [GO:0030140]; enzyme binding [GO:0019899]; nerve growth factor binding [GO:0048406]; nerve growth factor receptor activity [GO:0010465]; neurotensin receptor activity, non-G protein-coupled [GO:0030379]; retromer complex binding [GO:1905394]; endocytosis [GO:0006897]; endosome to lysosome transport [GO:0008333]; endosome transport via multivesicular body sorting pathway [GO:0032509]; extrinsic apoptotic signaling pathway via death domain receptors [GO:0008625]; G protein-coupled receptor signaling pathway [GO:0007186]; glucose import [GO:0046323]; Golgi to endosome transport [GO:0006895]; Golgi to lysosome transport [GO:0090160]; maintenance of synapse structure [GO:0099558]; myotube differentiation [GO:0014902]; negative regulation of fat cell differentiation [GO:0045599]; negative regulation of lipoprotein lipase activity [GO:0051005]; neuropeptide signaling pathway [GO:0007218]; neurotrophin TRK receptor signaling pathway [GO:0048011]; ossification [GO:0001503]; plasma membrane to endosome transport [GO:0048227]; positive regulation of epithelial cell apoptotic process [GO:1904037]; post-Golgi vesicle-mediated transport [GO:0006892]; protein targeting to lysosome [GO:0006622]; regulation of gene expression [GO:0010468]; response to insulin [GO:0032868]; vesicle organization [GO:0016050] |
15236333_The trafficking of prosaposin and GM2 activator protein to the lysosome in sertoli cells is dependent on sortilin. 15372498_Activation of type I neurotensin receptor-3 in microglial cells mediates the induction of chemokine and cytokine expression by neurotensin. 15992544_Sortilin is not only essential, but also sufficient for biogenesis of GSVs and acquisition of insulin responsiveness in adipose cells. 17220298_the luminal Vps10p domain of sortilin plays the predominant role in targeting to insulin-responsive Glut4-containing vesicles 17934455_The sortilin pathway has distinct roles in pro-neurotrophin-induced apoptotic signaling in pathological conditions, but also in specific stages of neuronal development and aging. 19219422_Sortilin, an essential player in adipocyte and muscle glucose metabolism through the control of GLUT4 localisation, is downregulated in obesity and TNF-alpha is likely to be involved in this defect. 19407813_The authors show that the mammalian p75NTR homologue NRH2 critically regulates the expression of sortilin on the neuronal cell surface and promotes p75NTR and sortilin receptor complex formation, rendering cells responsive to proneurotrophins 19732768_inactivation of the sortilin gene produces a significant decrease of prosaposin in the lysosomes 20405001_Results describe the antidepressant effects of spadin, a secreted peptide derived from the propeptide generated by the maturation of the neurotensin receptor 3 (NTSR3/Sortilin) and acting through TREK-1 inhibition. 20686566_Sort1 alters plasma LDL-C and very low-density lipoprotein (VLDL) particle levels by modulating hepatic VLDL secretion 20805226_insulin-sensitizing action of peroxisome proliferator-activated receptor gamma agonists to be at least partially mediated through the restoration of proper GLUT4 trafficking/sorting events governed by sortilin. 20816088_Data show that sortilin, encoded by SORT1, is an intracellular sorting receptor for apolipoprotein (apo) B100, facilitates the formation and hepatic export of apoB100-containing lipoproteins, and regulates plasma low-density lipoprotein cholesterol. 21040701_These results suggest that mouse Vps26b-Vps29-Vps35 retromer complex is implicated in the transport of sortilin from endosomes to the trans-Golgi network. 21092856_Sortilin-mediated progranulin endocytosis is likely to play a central role in frontotemporal lobar degeneration. pathophysiology. 21102451_findings demonstrate a role for sortilin as an anterograde trafficking receptor for Trk and a positive modulator of neurotrophin-induced neuronal survival. 21261755_Results suggest that proneurotrophin-3 and proBDNF may play important roles in the response to noise-induced injuries or ototoxic damage via the Sortilin:p75(NTR) death-signalling complex. 21730062_the regulation of sortilin shedding and identify a novel mechanism by which sortilin ectodomain shedding acts as a regulatory switch for delivery of BDNF to the secretory pathway or to the lysosome, thus modulating the bioavailability of endogenous BDNF. 22466652_Endoplasmic reticulum stress target Atf3 acts at the SORT1 promoter region as a transcriptional repressor. 22558402_role of sortilin in light-dependent photoreceptor degeneration 22579764_Sortilin is required for toxic action of Abeta oligomers (AbetaOs): extracellular AbetaOs trigger apoptosis, and intraneuronal AbetaOs impair degradation pathways. 22751103_model in which increased hepatic sortilin binds intracellular APOB-containing particles in the Golgi apparatus as well as extracellular LDL at the plasma membrane and traffics them to the lysosome for degradation 22781549_We demonstrate that loss of PGRN impairs proper neurite outgrowth and branching independent of SORT1 23084031_Loss of Sortilin reduced the release of interferon-gamma upon infections and in autoimmune colitis. 23283348_our findings identified sortilin as an essential neuronal pathway for APOE-containing lipoproteins in vivo and suggest an intriguing link between amyloid-beta catabolism and pro-neurotrophin signaling converging on this receptor. 23313508_this study suggests that sortilin might be involved in different aspects of neurodegeneration in a complex way, supporting the view that sortilin functions in the CNS are broader than being a co-receptor in proneurotrophin and neurotrophin signaling. 23704887_sortilin is implicated in APP lysosomal and lipid raft targeting via its carboxyl-terminal F/YXXXXF/Y motif. 23904453_Increased saturated fatty acids downregulate liver Sort1 protein, which may contribute to the development of dyslipidemia in obesity and diabetes. 23966466_Sortilin ectopically expressed in undifferentiated cells was translocated to the plasma membrane regardless of the presence or absence of myc7-Glut4 24416154_endogenous changes occur in NT and its receptors across multiple brain regions, and these likely support the emergence of some maternal behaviors. 24928897_sigma1B adaptin regulates adipogenesis by mediating the sorting of sortilin in adipose tissue. 25173968_Sortilin deficiency induces a beneficial metabolic phenotype in liver and adipose tissue upon DIO, mediated in part by reduced aSMase activity. 25401472_sortilin influences cytokine secretion and that targeting sortilin in immune cells attenuates inflammation and reduces atherosclerosis. 25593281_Sortilin-deficient macrophages had significantly reduced uptake of native LDL ex vivo and reduced foam cell formation in vivo, whereas sortilin overexpression in macrophages resulted in increased LDL uptake and foam cell formation. 25805502_Sort1 may play a role in altered hepatic apoB100 metabolism in insulin-resistant conditions 26370502_Prosaposin facilitates sortilin-independent lysosomal trafficking of progranulin. 26477595_Overexpression of SORT1 was shown to reduce Very-Low-Density Lipoproteins , Triglyceride and Apolipoprotein B secretion in mice. 26566674_plays a role in innate immune processes and, thus, not in autoimmune neuroinflammation. 26566675_promotes neutrophil adhesion and recruitment via the promotion of cytoskeletal function downstream of integrin-mediated adhesion 26614389_TDP-43 functions within a network of hnRNP proteins to inhibit the production of a truncated human SORT1 receptor. 26950419_Sortilin mediates vascular calcification via its recruitment into extracellular vesicles. 26964886_Sortilin is highly expressed in epithelial tissues of the developing lung, nasal cavity, kidney, pancreas and salivary gland. 27118371_This study demonstrated that Mice with Sort1 deficiency display normal cognition but elevated anxiety-like behavior. 27220277_findings provide insights into the posttranslational regulation of cytokine production through the posttranscriptional control of sortilin expression by TLR signals. 27389464_Altogether, sortilin defines a pathway required for optimal intracellular mycobacteria control and lung inflammation in vivo. 27632999_Shh and Sort1 can interact at the level of the Golgi and that Sort1 directs Shh away from the pathways that promote its secretion. 27816451_By contrast, treatment with dexamethasone, which also induced insulin resistance, was without effect on sortilin expression and PE production. As a putative bioactive peptide, we have evaluated its autocrine effect by the use of spadin on 3T3-L1 adipocytes by performing glucose uptake and signalling experiments. Any effect was measured on adipocytes indicating that the use of spadin as an antidepressant would have no side 27842214_sortilin regulates cholestatic liver damage and fibrosis via effects on both aSMase activity and serum IL-6. 27980238_Data suggest that sortilin expression in both gastrocnemius and extensor digitorum longus muscle is down-regulated by 17-18h of fasting/food deprivation; pathological levels of high blood glucose, as in streptozotocin-induced diabetes, do not alter sortilin expression in skeletal muscle; sortilin expression is elevated during differentiation of myocyte cell line or in presence of high concentration of glucose. 28236654_Sortilin 1 (Sort1) knockout (KO) does not affect Western diet-induced obesity nor adipose fatty acid and ceramide concentrations. 28498396_We showed that expression levels for NTS and NTSR1 varied, that NTSR2 expression was not detectable and that NTSR3 was consistently expressed in all CRC cell lines examined. 28742217_we identify interferon-gamma (IFN-gamma) as the key inflammatory mediator controlling sortilin-1 levels 28844948_this study revealed an important role of insulin signaling in regulating adipose Sort1 protein stability, and further suggests that impaired insulin signaling may underlie reduced adipose Sort1 in obesity. 29056359_our findings indicate that sortilin is a beneficial protein for the reduction of amyloid pathology in APP/PS1 mice by promoting APP degradation 29167428_Present crystal structures that show the sortilin luminal segment (s-sortilin) undergoes a conformational change and dimerizes at low pH. The conformational change, within all three sortilin luminal domains, provides an altered surface and the dimers sterically shield a large interface while bringing the two s-sortilin C-termini into close proximity. 29398357_Study shows that retrograde signaling from progranulin to sort1 counteracts synapse elimination in the developing cerebellum. 29405286_The sortilin and SorCS2 are extensively present in several distinct anatomical areas in the developing nervous system and are rarely co-expressed. 29899496_Findings suggest a role of Sortilin (Sort1) in energy regulation and lipid homeostasis in female mice, which may be a potential therapeutic target for obesity and cardiovascular disease. 30670473_Hepatocyte sortilin 1 knockout and treatment with a sortilin 1 inhibitor reduced plasma cholesterol in Western diet-fed mice 31767632_The Rac-p21-activated kinases 1 to 3 (PAK1-3) have a direct impact on sortilin and each of the three group A kinases binds and phosphorylates a serine residue positioned in a sorting motif within the sortilin cytoplasmic domain. 31961070_PCBP2 post-transcriptionally regulates sortilin expression by binding to a C-rich element in its 3' UTR. 32588544_Apolipoprotein E4 disrupts the neuroprotective action of sortilin in neuronal lipid metabolism and endocannabinoid signaling. 33498179_Identification of Sortilin Alternatively Spliced Variants in Mouse 3T3L1 Adipocytes. 34004257_Sortilin regulates keratinocyte proliferation and apoptosis through the PI3K-AKT signaling pathway. 34557909_ApoE4 disrupts interaction of sortilin with fatty acid-binding protein 7 essential to promote lipid signaling. 34931016_Sortilin deletion in the prefrontal cortex and hippocampus ameliorates depressive-like behaviors in mice via regulating ASM/ceramide signaling. 35104805_Targeting the ASMase/S1P pathway protects from sortilin-evoked vascular damage in hypertension. |
ENSG00000134243 |
SORT1 |
256.727684 |
0.4881197094 |
-1.034693 |
0.16470684 |
39.042821 |
0.00000000041461104320800029516864847465077142246725472318757965695112943649291992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000061136063989779865430417807748569858361520346079487353563308715820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
167.390459877751 |
19.151853106393 |
342.929114861542 |
28.3327396766234 |
| ENSMUSG00000068794 |
213945 |
Col28a1 |
protein_coding |
Q2UY11 |
FUNCTION: May act as a cell-binding protein. |
Alternative splicing;Basement membrane;Cell adhesion;Collagen;Disulfide bond;Extracellular matrix;Protease inhibitor;Reference proteome;Repeat;Secreted;Serine protease inhibitor;Signal |
|
|
mmu:213945; |
basement membrane [GO:0005604]; collagen trimer [GO:0005581]; collagen-containing extracellular matrix [GO:0062023]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; extracellular matrix structural constituent [GO:0005201]; serine-type endopeptidase inhibitor activity [GO:0004867]; cell adhesion [GO:0007155]; extracellular matrix organization [GO:0030198]; negative regulation of peptidase activity [GO:0010466] |
16330543_Collagen XXVIII is a novel von Willebrand factor A domain-containing protein with many imperfections in the collagenous domain 20830809_The correlation between the absence of myelin and the presence of collagen XXVIII is confirmed in a mouse model of Charcot-Marie-Tooth characterized by dysmyelinated nerve fibers, in which enhancement of collagen XXVIII labeling is observed 36518326_Type XXVIII Collagen Regulates Renal Interstitial Fibrosis and Epithelial-Mesenchymal Transition by SREBP1-Mediated HKDC1 Expression. |
ENSG00000215018 |
COL28A1 |
29.046574 |
0.2789699992 |
-1.841818 |
0.49188913 |
13.768559 |
0.00020676787803760964109742181982198871992295607924461364746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011415325546462015866844286193781954352743923664093017578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.550742587431 |
4.36258646013137 |
44.9895781748881 |
10.6965534881759 |
| ENSMUSG00000069053 |
22202 |
Uba1y |
protein_coding |
P31254 |
FUNCTION: Activates ubiquitin by first adenylating its C-terminal glycine residue with ATP, and thereafter linking this residue to the side chain of a cysteine residue in E1, yielding a ubiquitin-E1 thioester and free AMP (By similarity). The Y chromosome form could be involved in the survival and proliferation of differentiating spermatogonia. {ECO:0000250|UniProtKB:P22314, ECO:0000305}. |
ATP-binding;Ligase;Nucleotide-binding;Reference proteome;Ubl conjugation pathway |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000250|UniProtKB:P22314}. |
|
|
cytoplasm [GO:0005737]; nucleus [GO:0005634]; ATP binding [GO:0005524]; ubiquitin activating enzyme activity [GO:0004839]; cellular response to DNA damage stimulus [GO:0006974]; protein modification by small protein conjugation [GO:0032446]; protein ubiquitination [GO:0016567]; ubiquitin-dependent protein catabolic process [GO:0006511] |
|
|
|
34.092913 |
16.2183872045 |
4.019558 |
0.66761247 |
36.275086 |
0.00000000171340284907125709334868843199692692302882335297908866778016090393066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000233648632023754580865654187186997159386692146654240787029266357421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
63.3109154689344 |
30.3542408305761 |
3.90365050917021 |
1.59661591471863 |
| ENSMUSG00000069171 |
13865 |
Nr2f1 |
protein_coding |
Q32NY6 |
Human_homologues FUNCTION: Coup (chicken ovalbumin upstream promoter) transcription factor binds to the ovalbumin promoter and, in conjunction with another protein (S300-II) stimulates initiation of transcription. Binds to both direct repeats and palindromes of the 5'-AGGTCA-3' motif. Represses transcriptional activity of LHCG. {ECO:0000269|PubMed:10644740, ECO:0000269|PubMed:11682620}. |
DNA-binding;Metal-binding;Nucleus;Proteomics identification;Receptor;Reference proteome;Transcription;Transcription regulation;Zinc;Zinc-finger |
|
|
mmu:13865; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654]; DNA-binding transcription repressor activity, RNA polymerase II-specific [GO:0001227]; retinoic acid-responsive element binding [GO:0044323]; zinc ion binding [GO:0008270]; negative regulation of neuron projection development [GO:0010977]; nervous system development [GO:0007399]; positive regulation of transcription by RNA polymerase II [GO:0045944] |
14738874_Results suggest COUP-TFI is an important regulator of oligodendrocyte differentiation. 15907456_analysis of COUP-TFI and COUP-TFII expression during development and functional maturation of the mouse inner ear 16144621_In the marginal zone (MZ) of the cortex , the nuclear receptor COUP-TFI is localized in calbindin-positive cells and not in reelin-positive cells and suggested represses differentiation of Cajal-Retzius cells. 17021036_Results show that COUP-TFI is required for proper axonal growth and guidance of all major forebrain commissures. 18165280_COUP-TFI controls cerebral cortical development processes by repressing Mapk/Erk, Akt, and beta-catenin signaling 18177425_COUP-TF1 is an important signaling molecule during vitamin A (Rol)-mediated very early stage of embryonic development. 19766631_we found that COUP-TFI and -TFII play roles in amacrine cell differentiation, and COUP-TFgamma has distinct expression pattern and roles during retinal development. 20007910_Transcriptional and posttranscriptional mechanisms involving NR2F1 and IRE1beta ensure low microsomal triglyceride transfer protein expression in undifferentiated intestinal cells and avoid apolipoprotein B lipoprotein biosynthesis. 20133588_COUP-TFI exerts critical areal and temporal control over the precise differentiation of CSMN during corticogenesis, thereby enabling the area-specific functional features of motor and sensory areas to arise. 20147377_Novel and distinct cell-intrinsic mechanisms mediated by COUP-TF genes to direct the specification and differentiation of progenitor cells; COUP-TFs are crucial for dorsalization of the eye. 20797425_LHbeta promoter activity is further regulated by the orphan nuclear receptors, chicken ovalbumin upstream promoter-transcription factors (COUP-TFI and COUP-TFII). 21266512_This study demonstrated that kallistatin expression is repressed by triiodothyronine and modulated by the orphan nuclear receptor, chicken ovalbumin upstream promoter transcription factor 1 (COUP-TF1). 21430164_COUP-TFI controls the delicate balance between medial ganglionic eminence and caudal ganglionic eminence derived cortical interneurons by regulating intermediate progenitor divisions 21965613_Data demonstrate that COUP-TFI modulates late-born neuron migration and favours proper differentiation of callosal projection neurons by finely regulating Rnd2 expression levels. 23018522_Exogenous expression of COUP-TFI in AS cells, in a dose-dependent fashion, leads to growth inhibition. 23056316_study extends the pool of recognized putative AR targets and identifies a negatively regulated target of AR - COUP-TF1 - which could possibly play a role in human prostate cancer 23307639_Findings provide evidence that Sp8 and another transcription factor, COUP-TF1, mutually repress each other's cortical neuroepithelial expression along the anterior-posterior and dorsoventral axes 23472160_CoupTFI collaborates with retinoic acid signaling to regulate both cortical ventricular zone progenitor cell behavior and cortical neurogenesis 23785296_Our data suggest that the non-protein coding locus Mcs1a regulates Nr2f1, which is a candidate modifier of differentiation, proliferation, and mammary cancer risk. 23975195_COUP-TFI and related NRs such as the COUPTFs and PNR can selectively associate with the developmental corepressor BCL11A via a conserved motif F/YSXXLXXL/Y within the RID1 and RID2 domains. The interaction with BCL11A facilitates COUP-TFII-mediated repression of the RARb2 gene. 23975195_Interaction of NSD1 with the NR2E/F subfamily including COUP-TFI, COUP-TFII, EAR2 and TLX requires a F/YSXXLXXL/Y motif. NSD1 interaction with liganded NRs is mediated by an overlapping LXXLL motif. 24227652_odour deprivation, which is known to downregulate tyrosine hydroxylase expression in the olfactory bulb, also downregulates COUP-TFI in dopaminergic cells, indicating a correlation between tyrosine hydroxylase- and COUP-TFI-activity-dependent action. 24349493_a novel tissue-dependent coregulatory network for NR2F1, miR-140, and Klf9 in the inner ear 25922524_Transcription factors COUP-TFI and COUP-TFII are required for the production of granule cells in the mouse olfactory bulb. 26813976_COUP-TFI expression is required for functional organization of the hippocampal septo-temporal longitudinal axis 27585883_These data identify silencer elements of the Nr2f1-A830082K12Rik gene pair as new candidate loci for Waardenburg syndrome type 4. 28506990_COUP-TFI is required predominantly in Dentate gyrus progenitors for modulating expression of the Cxcr4 receptor during granule cell neurogenesis and migration. 28694260_Coup-TF1 and Coup-TF2 control subtype and laminar identity of MGE-derived neocortical interneurons 29205104_These results indicate that COUP-TF1 plays an important role in modulating the timing and magnitude of T3-stimulated gene expression required for normal corticogenesis. 29996095_COUP-TFI as a central regulator of the neuron-astroglia cell fate decision and a key modulator during neuroinflammation in the adult hippocampus. 30174317_These data support a translational control of COUP-TFI gradient expression by FGF8 via miR-21 and contribute to our understanding of how regionalized expression is established during neocortical area mapping. 30653836_Loss of Nr2f1 in mouse cortex leads to increased locomotor activity but less anxiety. 31318166_Mouse Nr2f1 haploinsufficiency results in model of Bosch-Boonstra-Schaaf optic atrophy. 31600777_Nr2f1 heterozygous knockout mice recapitulate neurological phenotypes of Bosch-Boonstra-Schaaf optic atrophy syndrome and show impaired hippocampal synaptic plasticity. 32484994_NR2F1 regulates regional progenitor dynamics in the mouse neocortex and cortical gyrification in BBSOAS patients. 32572460_COUP-TFI/Nr2f1 Orchestrates Intrinsic Neuronal Activity during Development of the Somatosensory Cortex. 35163483_Genome-Wide ChIPseq Analysis of AhR, COUP-TF, and HNF4 Enrichment in TCDD-Treated Mouse Liver. |
ENSG00000175745 |
NR2F1 |
288.524643 |
0.4485435461 |
-1.156680 |
0.24209473 |
22.432912 |
0.00000217612862360050525153184336479839089406596031039953231811523437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000181746752293262343793539548286730678228195756673812866210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
172.505177235015 |
29.0836737165098 |
384.589587211945 |
47.5726028679454 |
| ENSMUSG00000069372 |
629147 |
Ctxn3 |
protein_coding |
Q8BXZ0 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:629147; |
membrane [GO:0016020] |
Human_homologues 17786280_Selective expression of CTXN3 in the kidney and brain, the amino acid identity to cortexin, and its high conservation among different species indicate that CTXN3 may be involved in a process specifically restricted to kidney and brain tissue function. 19023125_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 22643131_Two single nucleotide polymophisms of the CTXN3 and SLC12A2 genes are associated with risk of schizophrenia in a Thai population. 25889058_altered expression of cortexin 3, either alone, or in parallel with changes in disrupted-in-schizophrenia 1, could subtly perturb GABAergic neurotransmission |
ENSG00000205279 |
CTXN3 |
10.629786 |
0.1311204147 |
-2.931036 |
0.89650883 |
10.925011 |
0.00094874660277413343581476290466980572091415524482727050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0044112662556335781663396922169795288937166333198547363281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.67946972829132 |
1.77430555385033 |
20.4351834436545 |
8.20006091280072 |
| ENSMUSG00000069516 |
17105 |
Lyz2 |
protein_coding |
P08905 |
FUNCTION: Lysozymes have primarily a bacteriolytic function; those in tissues and body fluids are associated with the monocyte-macrophage system and enhance the activity of immunoagents. Lyz2 is active against a range of Gram-positive and Gram-negative bacteria. More effective than Lyz1 in killing Gram-negative bacteria. Lyz1 and Lyz2 are equally effective in killing Gram-positive bacteria. {ECO:0000255|PROSITE-ProRule:PRU00680, ECO:0000269|PubMed:14977423}. |
3D-structure;Antimicrobial;Bacteriolytic enzyme;Direct protein sequencing;Disulfide bond;Glycosidase;Hydrolase;Milk protein;Reference proteome;Secreted;Signal |
|
|
mmu:17105; |
cytoplasm [GO:0005737]; endoplasmic reticulum lumen [GO:0005788]; extracellular space [GO:0005615]; Golgi cis cisterna [GO:0000137]; Golgi stack [GO:0005795]; microvillus [GO:0005902]; rough endoplasmic reticulum lumen [GO:0048237]; secretory granule [GO:0030141]; trans-Golgi network transport vesicle [GO:0030140]; hydrolase activity, acting on glycosyl bonds [GO:0016798]; identical protein binding [GO:0042802]; lysozyme activity [GO:0003796]; cytolysis [GO:0019835]; defense response to bacterium [GO:0042742]; defense response to Gram-negative bacterium [GO:0050829]; defense response to Gram-positive bacterium [GO:0050830]; killing of cells of another organism [GO:0031640]; metabolic process [GO:0008152] |
18508059_ApoE and lysozyme M have roles in monocyte and neutrophil trafficking in atherosclerosis in a mouse model 19416848_P-tau was found in medial entorhinal cortex of Naglu(-/-) mice, in the same neurons as lysozyme 21875910_Depletion of LysM(+) cells attenuated ATII-induced blood pressure increase. 26471282_Data show that receptor-interacting serine-threonine kinase 3 (RIPK3) deletion prevents inflammatory phenotypes in CreLysM (lysozyme M) Casp8fl/fl (caspase 8) mice. 26627460_MyD88 signaling in lysozyme M and CD11c-expressing myeloid cells, as well as in pulmonary epithelial cells, is critical to restore inflammatory cytokine and antimicrobial peptide production, leading to efficient neutrophil recruitment and enhanced bacterial clearance. 28184027_Results show that lysozyme (LYSO) - induced Staphylococcus aureus bacteriostasis increased with increasing concentrations of baicalin, and that baicalin binding to LYSO synergistically increased the antibacterial activity of LYSO. 31705272_These findings indicate that loss of LysM is non-contributory in sterile acute lung injury. 33129690_Toll-like receptor mediated lysozyme expression in Niemann-pick disease, type C1. 33556481_Adipose tissue knockdown of lysozyme reduces local inflammation and improves adipogenesis in high-fat diet-fed mice. 33929610_Ablation of lysozyme M-positive cells prevents aircraft noise-induced vascular damage without improving cerebral side effects. |
|
|
172.471365 |
2.0911204449 |
1.064276 |
0.22807509 |
21.212251 |
0.00000411128204774616542457139553579104074287897674366831779479980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000327526402464799112077345477622714042809093371033668518066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
230.322220064934 |
36.5634066097678 |
110.142971741104 |
13.4404987824503 |
| ENSMUSG00000069763 |
67888 |
Tmem100 |
protein_coding |
Q9CQG9 |
FUNCTION: Plays a role during embryonic arterial endothelium differentiation and vascular morphogenesis through the ACVRL1 receptor-dependent signaling pathway upon stimulation by bone morphogenetic proteins, such as GDF2/BMP9 and BMP10 (PubMed:22783020). Involved in the regulation of nociception, acting as a modulator of the interaction between TRPA1 and TRPV1, two molecular sensors and mediators of pain signals in dorsal root ganglia (DRG) neurons (PubMed:25640077). Mechanistically, it weakens their interaction, thereby releasing the inhibition of TRPA1 by TRPV1 and increasing the single-channel open probability of the TRPA1-TRPV1 complex (PubMed:25640077). {ECO:0000269|PubMed:22783020, ECO:0000269|PubMed:25640077}. |
Cell membrane;Cytoplasm;Developmental protein;Differentiation;Direct protein sequencing;Endoplasmic reticulum;Membrane;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:67888; |
endoplasmic reticulum [GO:0005783]; perikaryon [GO:0043204]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; angiogenesis [GO:0001525]; arterial endothelial cell differentiation [GO:0060842]; BMP signaling pathway [GO:0030509]; cellular response to BMP stimulus [GO:0071773]; endocardial cushion development [GO:0003197]; epithelial to mesenchymal transition involved in endocardial cushion formation [GO:0003198]; in utero embryonic development [GO:0001701]; Notch signaling pathway [GO:0007219]; positive regulation of endothelial cell differentiation [GO:0045603]; positive regulation of vasculogenesis [GO:2001214]; protein kinase B signaling [GO:0043491]; regulation of calcium-mediated signaling [GO:0050848]; regulation of sensory perception of pain [GO:0051930]; vasculogenesis [GO:0001570] |
20848592_Tmem100 was predominantly expressed in arterial endothelial cells of developing embryos. 22783020_TMEM100 may play indispensable roles downstream of BMP9/BMP10-ALK1 signaling during endothelial differentiation and vascular morphogenesis. 23485812_This study demonistrated that TMEM100 is a novel, membrane-associated marker for enteric nerves and is as effective as PGP9.5 for identifying neuronal structures in the gastrointestinal tract. 25318679_The Tmem100 deficiency causes EndMT defect during AVC cushion formation possibly via disturbance of multiple calcium-related signaling events. 25538155_TMEM100 has essential functions for the maintenance of vascular integrity as well as the formation of blood vessels. 25640077_This study demonistrated that Tmem100 Is a Regulator of TRPA1-TRPV1 Complex and Contributes to Persistent Pain in mice. 29247399_Tmem100 is a potential regulator of kidney development by related to BMP signal transduction in the process of metanephric mesenchymal-cell-to-epithelial transition.BMP 7 is a key regulator of Tmem100-mediated cell proliferation in metanephric mesenchymal cells.There is a complicated regulation network among Tmem100, BMP7, and BMPR-II in mouse embryonic kidney-derived cells. 32112176_TMEM100 is a key factor for specification of lymphatic endothelial progenitors. 33651473_Tmem100-BAC-EGFP mice to selectively mark and purify embryonic endothelial cells of large caliber arteries in mid-gestational vascular formation. |
ENSG00000166292 |
TMEM100 |
61.874981 |
3.0193652699 |
1.594245 |
0.34118531 |
21.374991 |
0.00000377665608926123686395377732072375920324702747166156768798828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000303247367167249104360614991859890210434969048947095870971679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
96.5613554432574 |
23.0494117714738 |
31.9806803119587 |
5.97436666058264 |
| ENSMUSG00000070003 |
76900 |
Ssbp4 |
protein_coding |
A0A1B0GRX5 |
|
Proteomics identification;Reference proteome |
|
|
Human_homologues hsa:170463; |
nucleus [GO:0005634]; positive regulation of transcription by RNA polymerase II [GO:0045944] |
Human_homologues 19913121_Observational study of gene-disease association. (HuGE Navigator) 20628086_Observational study of gene-disease association, gene-environment interaction, and pharmacogenomic / toxicogenomic. (HuGE Navigator) |
ENSG00000130511 |
SSBP4 |
274.230173 |
0.2816220922 |
-1.828168 |
0.20864248 |
74.113708 |
0.00000000000000000737444412010008992045889804737016452332438779627499583768646118642209330573678016662597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000002436605918381010224398250729982482267227771716676387736555398078053258359432220458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
116.060090914405 |
16.8431738297249 |
412.11287791911 |
42.9000428154777 |
| ENSMUSG00000070031 |
434484 |
Sp140 |
protein_coding |
D3Z054 |
|
Reference proteome |
|
|
|
fibrillar center [GO:0001650]; mitochondrion [GO:0005739] |
34156030_The role of Sp140 revealed in IgE and mast cell responses in Collaborative Cross mice. 35952671_Epigenetic reader SP140 loss of function drives Crohn's disease due to uncontrolled macrophage topoisomerases. |
|
|
137.388348 |
4.5183352810 |
2.175791 |
0.25025361 |
74.232398 |
0.00000000000000000694412419897453762310313876384194953176027699103152468484978498963755555450916290283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000002309451960672055581585674321968764122220706168711612527033594233216717839241027832031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
221.197790662371 |
38.5622481951499 |
48.9555946835826 |
6.87596054184204 |
| ENSMUSG00000070034 |
109032 |
Sp110 |
protein_coding |
Q8BVK9 |
FUNCTION: May act as a transcription factor. Plays a role in the innate immunity against intracellular pathogens. Required for resistance to M.tuberculosis and L.monocytogenes. Promotes apoptosis of infected cells. {ECO:0000269|PubMed:15815631}. |
3D-structure;Apoptosis;DNA-binding;Immunity;Innate immunity;Nucleus;Phosphoprotein;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:109032; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; apoptotic process [GO:0006915]; innate immune response [GO:0045087]; positive regulation of apoptotic process [GO:0043065]; regulation of transcription by RNA polymerase II [GO:0006357]; response to bacterium [GO:0009617] |
11471051_identification and analysis of the genes Sp100, Csprs, and Ifi75 in two members of the genus Mus, M. musculus and M. caroli 15815631_data indicate that the Ipr1 gene product might have a previously undocumented function in integrating signals generated by intracellular pathogens with mechanisms controlling innate immunity, cell death and pathogenesis 20221700_Ipr1 might be involved in apoptosis pathway via MYBBP1A. 21790702_mediates RAW 264.7 macrophage cell line resistance to Mycobacterium bovis 26590945_strain-specific promoter region polymorphism is associated with resistance to tuberculosis 26912204_These findings not only reveal the transcriptional basis of Sp110-mediated macrophage resistance to Mtb, but also suggest potential regulatory roles for Sp110 related to inflammatory responses, miRNA profiles, and the intracellular growth of Mtb. 27622275_mechanistic studies revealed that an arginine/lysine-rich element within the DNA-binding domain (SAND domain) is critical for Ipr1 binding to the importin protein receptor NPI-1, demonstrating that this element plays an essential role in mediating the nuclear localization of Ipr1 protein 27858493_Using mouse models and genetic characteristics of human patients the role of SP110b was studied to determine its role in controlling host immunity and susceptibility to TB by modulating nuclear factor-kappaB (NF-kappaB) activity resulting in down regulation of TNF-alpha production and upregulation of NF-kappaB-induced antipoptotic gene expression suppressing IFN-gamma-mediated monocyte and/or macrophage cell death. |
ENSG00000135899 |
SP110 |
99.627180 |
3.7967991612 |
1.924784 |
0.26801492 |
51.245425 |
0.00000000000081510496255897788415685820896207841373612423829797535290708765387535095214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000164228555419290360078705604985420374829219181123107773601077497005462646484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
148.841481904351 |
27.8101679025577 |
39.2018317499609 |
5.95044759220817 |
| ENSMUSG00000070047 |
14107 |
Fat1 |
protein_coding |
A0A087WPU4 |
Human_homologues FUNCTION: [Protocadherin Fat 1]: Plays an essential role for cellular polarization, directed cell migration and modulating cell-cell contact. {ECO:0000250}. |
Proteomics identification;Reference proteome |
|
|
|
apical plasma membrane [GO:0016324]; cell junction [GO:0030054]; cell-cell junction [GO:0005911]; filopodium [GO:0030175]; lamellipodium [GO:0030027]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; actin filament organization [GO:0007015]; camera-type eye development [GO:0043010]; camera-type eye morphogenesis [GO:0048593]; cell-cell adhesion [GO:0098609]; epithelial cell morphogenesis [GO:0003382]; establishment of epithelial cell apical/basal polarity involved in camera-type eye morphogenesis [GO:0003412]; establishment or maintenance of cell polarity [GO:0007163]; establishment or maintenance of epithelial cell apical/basal polarity [GO:0045197]; lens development in camera-type eye [GO:0002088] |
12724416_results confirm a necessary role for FAT1 in the modified adhesion junctions of the renal glomerular epithelial cell and reveal hitherto unsuspected roles for FAT1 in CNS development 15148305_Fat1 regulates actin cytoskeletal organization at cell peripheries, thereby modulating cell contacts and polarity. 16059920_data are compatible with conservation and sub-functionalization of dachsous 1, Fj, and Fat signaling in higher vertebrates. 17500054_FAT1(WT) is up-regulated in migration, induces cellular process formation when overexpressed, and is necessary for efficient wound healing 19131340_Fat1 and Atrs 1,2 act in concert after vascular injury but show further that distinct Atr isoforms have disparate effects on VSMC directional migration. 22510986_Data find that Fat1 and Fat4 cooperate during mouse development to control renal tubular elongation, cochlear extension, cranial neural tube formation and patterning of outer hair cells in the cochlea. 22959770_FAT1 suppression in activated hepatic stellate cells caused a downregulation of NFkappaB activity 26209645_Fat1 interacts with Fat4 to regulate neural tube closure, neural progenitor proliferation and apical constriction during mouse brain development. 26905694_Fat1 deletion in podocytes induces abnormal glomerular filtration barrier development leading to podocyte foot process effacement. Fat1 knockdown in renal tubular cells reduces migration, decreases active RAC1/CDC42 and induces defects in lumen formation. 27828948_studies suggest that Fat1 controls mitochondrial activity to restrain cell growth during the reparative, proliferative state induced by vascular injury. Given recent reports linking Fat1 to cancer, abnormal kidney and muscle development, and neuropsychiatric disease, this Fat1 function may have importance in other settings of altered cell growth and metabolism 29768404_coordinates the coupling of muscular and neuronal morphogenesis by playing distinct but complementary actions in several cell types 33328637_Fat1 deletion promotes hybrid EMT state, tumour stemness and metastasis. 34100899_Postnatal expression profiles of atypical cadherin FAT1 suggest its role in autism. 34627073_Effects of cyclic tensile strain and microgravity on the distribution of actin fiber and Fat1 cadherin in murine articular chondrocytes. 35050341_Astrocyte-intrinsic and -extrinsic Fat1 activities regulate astrocyte development and angiogenesis in the retina. |
ENSG00000083857 |
FAT1 |
6296.751900 |
0.4358018617 |
-1.198256 |
0.06736273 |
308.811059 |
0.00000000000000000000000000000000000000000000000000000000000000000000396487053446700296364184177887194384311341121883510042451420100409558922263809878628953930532998005180442706432546817715789891538799171510715314718229336286376196759245532294513303384064784040675 |
0.0000000000000000000000000000000000000000000000000000000000000000010784447853750247484579359618112448421705835086402308670540113632862246040697963263105905998088818039715638543504381852477480487257608304983217456753230421587639773460531866788869592710398137569 |
Yes |
No |
3733.42309510067 |
174.385408694277 |
8566.7901484754 |
296.302218700878 |
| ENSMUSG00000070476 |
71532 |
Fam217b |
protein_coding |
A2AJW5 |
|
Proteomics identification;Reference proteome |
|
|
mmu:71532; |
cytosol [GO:0005829]; nucleoplasm [GO:0005654] |
|
ENSG00000196227 |
FAM217B |
152.095412 |
0.4601366370 |
-1.119866 |
0.23148380 |
23.272953 |
0.00000140562886097767339456095312538730368601136433426290750503540039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000121781299244070358026633543735428588661307003349065780639648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
94.7703602542865 |
15.2714574259108 |
205.96134414337 |
23.9768293489956 |
| ENSMUSG00000070526 |
27412 |
Peg12 |
protein_coding |
Q9WVA7 |
Human_homologues FUNCTION: Positively regulates the Wnt signaling pathway by stabilizing beta-catenin through the association with GSK-3. May play a role in tumor progression and collaborate with PIM1 and MYC in lymphomagenesis. {ECO:0000269|PubMed:12556519}. |
Proteomics identification;Reference proteome |
|
|
mmu:27412; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; nucleus [GO:0005634]; molecular function inhibitor activity [GO:0140678]; beta-catenin destruction complex disassembly [GO:1904886]; canonical Wnt signaling pathway [GO:0060070]; embryonic axis specification [GO:0000578]; positive regulation of canonical Wnt signaling pathway [GO:0090263]; regulation of protein export from nucleus [GO:0046825] |
11845283_Loss of Frat3 expression may contribute to the phenotype of mouse models of Prader-Willi syndrome |
ENSG00000165879 |
FRAT1 |
20.060890 |
0.1563381178 |
-2.677259 |
0.69506256 |
14.508830 |
0.00013950407765825088087126293334705451343324966728687286376953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0008067297307860582479197009142524166236398741602897644042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.12749930242985 |
2.5670109880943 |
32.7974992514353 |
10.7379205304555 |
| ENSMUSG00000070605 |
433791 |
Zfp992 |
protein_coding |
B1ASD8 |
Human_homologues FUNCTION: May be involved in transcriptional regulation. |
Metal-binding;Reference proteome;Repeat;Zinc;Zinc-finger |
|
|
mmu:433791; |
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of transcription by RNA polymerase II [GO:0006357] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 26823146_genes encoding transcription factors expressed in the brain might be an important group of major depressive disorder candidate genes. |
ENSG00000196378 |
ZNF34 |
20.438385 |
4.4870964544 |
2.165782 |
0.61721237 |
12.098749 |
0.00050455671562297643023553561647531751077622175216674804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0025231148694580358624039995163457206217572093009948730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
31.5163235008939 |
13.8983684589221 |
7.02376777979053 |
2.5024227817667 |
| ENSMUSG00000070858 |
381633 |
Gm1673 |
protein_coding |
Q3UR78 |
|
Alternative splicing;Reference proteome;Secreted;Signal |
|
|
mmu:381633; |
extracellular region [GO:0005576] |
Human_homologues 21287218_C4Orf48 probably encodes a novel neuropeptide, which, if hemizygously deleted, may be involved in the observed intellectual and fine motor disabilities and thus in the overall neurological aspects of Wolf-Hirschhorn syndrome. |
ENSG00000243449 |
C4orf48 |
40.999700 |
0.4070539717 |
-1.296708 |
0.40379417 |
10.154054 |
0.00143984223313475454518461926767258773907087743282318115234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0063940748965331138317647052815573260886594653129577636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.3530674829255 |
7.18905083108544 |
62.2842896649627 |
12.4836842476125 |
| ENSMUSG00000070942 |
107527 |
Il1rl2 |
protein_coding |
Q9ERS7 |
FUNCTION: Receptor for interleukin-36 (IL36A, IL36B and IL36G). After binding to interleukin-36 associates with the coreceptor IL1RAP to form the interleukin-36 receptor complex which mediates interleukin-36-dependent activation of NF-kappa-B, MAPK and other pathways. The IL-36 signaling system is thought to be present in epithelial barriers and to take part in local inflammatory response; it is similar to the IL-1 system. Seems to be involved in skin inflammatory response by induction of the IL-23/IL-17/IL-22 pathway. {ECO:0000269|PubMed:21860022, ECO:0000269|PubMed:23064362}. |
Disulfide bond;Glycoprotein;Hydrolase;Immunity;Immunoglobulin domain;Inflammatory response;Innate immunity;Membrane;NAD;Receptor;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:107527; |
membrane [GO:0016020]; cytokine receptor activity [GO:0004896]; interleukin-1, type I, activating receptor activity [GO:0004909]; NAD(P)+ nucleosidase activity [GO:0050135]; NAD+ nucleotidase, cyclic ADP-ribose generating [GO:0061809]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; positive regulation of interleukin-6 production [GO:0032755]; positive regulation of T cell differentiation [GO:0045582]; regulation of inflammatory response [GO:0050727] |
27966554_IL-36R-deficient mice were protected from influenza virus-induced lung injury and mortality 28327619_these findings reveal a fundamental contribution for the IL-36/IL-36R axis in regulating the Treg-Th9 cell balance with broad implications for Th cell-mediated disorders, such as inflammatory bowel diseases and particularly ulcerative colitis 28857172_Data show that Interleukin-36 receptor (IL-36R)-deficient fibroblast-like synoviocytes (FLS) had a limited capacity to support plasma cells (PC) survival compared to wild-type FLS. 29241623_IL-36R likely contributes to acute kidney injury pathogenesis through NF-kB activity and IL-6 and TNF-a upregulation. 32345660_IL-36 signaling in keratinocytes controls early IL-23 production in psoriasis-like dermatitis. 33087566_IL-36R signaling integrates innate and adaptive immune-mediated protection against enteropathogenic bacteria. 35578812_Interleukin-38 promotes skin tumorigenesis in an IL-1Rrp2-dependent manner. |
ENSG00000115598 |
IL1RL2 |
82.644551 |
3.3676200677 |
1.751729 |
0.51825515 |
9.925006 |
0.00163048302846600478947791668105082862894050776958465576171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0071366429567799385957904689803399378433823585510253906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
131.159692392175 |
48.1360162822152 |
38.9472950499376 |
10.7680753156926 |
| ENSMUSG00000070960 |
100503415 |
Gm19680 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
23.785984 |
3.5611741592 |
1.832353 |
0.57866271 |
9.633481 |
0.00191062156544438178917832971137613640166819095611572265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0081863819085369413791619663811616192106157541275024414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.4556398084484 |
14.193658149005 |
9.67535938090638 |
3.17073108860995 |
| ENSMUSG00000071042 |
240168 |
Rasgrp3 |
protein_coding |
A0A3Q4EG32 |
Human_homologues FUNCTION: Guanine nucleotide exchange factor (GEF) for Ras and Rap1. {ECO:0000269|PubMed:10934204}. |
Guanine-nucleotide releasing factor;Reference proteome |
|
|
|
cytoplasm [GO:0005737]; guanyl-nucleotide exchange factor complex [GO:0032045]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; GTPase activator activity [GO:0005096]; guanyl-nucleotide exchange factor activity [GO:0005085]; kinase binding [GO:0019900]; small GTPase binding [GO:0031267]; positive regulation of GTPase activity [GO:0043547]; Ras protein signal transduction [GO:0007265]; regulation of GTPase activity [GO:0043087] |
21474816_Expressed in developing blood vessels, Rasgrp3 contributes to the incidence of cardiovascular defects found in embryos from diabetic mothers. 23308188_RasGRP1, but not RasGRP3, is required for thymocyte positive selection and invariant natural killer T cell selection. 25118589_RasGRP3 limits inflammatory response by activating Rap1 on low-intensity pathogen infection, setting a threshold for preventing excessive inflammatory response. 27465532_RasGRP1/3-deficient progenitors show impaired migration toward the CCR9 ligand, CCL25, suggesting that RasGRP1 and RasGRP3 may regulate progenitor entry into the thymus through a CCR9-dependent mechanism. 29490280_RasGRP3 is specifically required for GNAQ/GNA11-driven Ras activation and tumorigenesis. |
ENSG00000152689 |
RASGRP3 |
40.717771 |
2.4909237611 |
1.316681 |
0.48722201 |
7.221624 |
0.00720305296896563140851554862820194102823734283447265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0254033116052985626087679094098348286934196949005126953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
59.2085774662884 |
20.3779841671338 |
23.7697268744152 |
6.35603242861156 |
| ENSMUSG00000071369 |
26408 |
Map3k5 |
protein_coding |
O35099 |
FUNCTION: Serine/threonine kinase which acts as an essential component of the MAP kinase signal transduction pathway. Plays an important role in the cascades of cellular responses evoked by changes in the environment. Mediates signaling for determination of cell fate such as differentiation and survival. Plays a crucial role in the apoptosis signal transduction pathway through mitochondria-dependent caspase activation. MAP3K5/ASK1 is required for the innate immune response, which is essential for host defense against a wide range of pathogens. Mediates signal transduction of various stressors like oxidative stress as well as by receptor-mediated inflammatory signals, such as the tumor necrosis factor (TNF) or lipopolysaccharide (LPS). Once activated, acts as an upstream activator of the MKK/JNK signal transduction cascade and the p38 MAPK signal transduction cascade through the phosphorylation and activation of several MAP kinase kinases like MAP2K4/SEK1, MAP2K3/MKK3, MAP2K6/MKK6 and MAP2K7/MKK7. These MAP2Ks in turn activate p38 MAPKs and c-jun N-terminal kinases (JNKs). Both p38 MAPK and JNKs control the transcription factors activator protein-1 (AP-1). {ECO:0000269|PubMed:11266364, ECO:0000269|PubMed:14749717, ECO:0000269|PubMed:15864310, ECO:0000269|PubMed:16527894, ECO:0000269|PubMed:16648474}. |
Apoptosis;ATP-binding;Coiled coil;Cytoplasm;Endoplasmic reticulum;Immunity;Innate immunity;Kinase;Magnesium;Metal-binding;Methylation;Nucleotide-binding;Phosphoprotein;Reference proteome;Serine/threonine-protein kinase;Stress response;Transferase;Ubl conjugation |
|
|
mmu:26408; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; external side of plasma membrane [GO:0009897]; IRE1-TRAF2-ASK1 complex [GO:1990604]; protein kinase complex [GO:1902911]; protein-containing complex [GO:0032991]; ATP binding [GO:0005524]; identical protein binding [GO:0042802]; JUN kinase kinase kinase activity [GO:0004706]; magnesium ion binding [GO:0000287]; MAP kinase kinase kinase activity [GO:0004709]; protein domain specific binding [GO:0019904]; protein homodimerization activity [GO:0042803]; protein kinase activity [GO:0004672]; protein kinase binding [GO:0019901]; protein phosphatase binding [GO:0019903]; protein serine kinase activity [GO:0106310]; cellular response to amino acid starvation [GO:0034198]; cellular response to hydrogen peroxide [GO:0070301]; cellular response to reactive nitrogen species [GO:1902170]; cellular response to tumor necrosis factor [GO:0071356]; endothelial cell apoptotic process [GO:0072577]; innate immune response [GO:0045087]; intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress [GO:0070059]; intrinsic apoptotic signaling pathway in response to oxidative stress [GO:0008631]; JNK cascade [GO:0007254]; MAPK cascade [GO:0000165]; p38MAPK cascade [GO:0038066]; positive regulation of apoptotic process [GO:0043065]; positive regulation of cardiac muscle cell apoptotic process [GO:0010666]; positive regulation of cysteine-type endopeptidase activity involved in apoptotic process [GO:0043280]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of JNK cascade [GO:0046330]; positive regulation of JUN kinase activity [GO:0043507]; positive regulation of MAPK cascade [GO:0043410]; positive regulation of myoblast differentiation [GO:0045663]; positive regulation of neuron death [GO:1901216]; positive regulation of p38MAPK cascade [GO:1900745]; positive regulation of protein kinase activity [GO:0045860]; positive regulation of protein phosphorylation [GO:0001934]; positive regulation of vascular associated smooth muscle cell proliferation [GO:1904707]; programmed necrotic cell death [GO:0097300]; protein phosphorylation [GO:0006468]; regulation of cell death [GO:0010941]; response to endoplasmic reticulum stress [GO:0034976]; response to ischemia [GO:0002931]; stress-activated MAPK cascade [GO:0051403]; wound healing [GO:0042060] |
12215209_roles of ASK1-mediated signal transduction in stress responses and the molecular mechanisms by which ASK1 determines cell fate such as survival, differentiation, or apoptosis [review] 12391142_These observations suggest that heat shock protein 72 functions as an endogenous inhibitor of ASK1. 12724324_ASK1/p38 signaling is essential for caspase 8 activation linked to M. avium-induced death signaling 14551246_These results provide the first in vivo evidence that ASK1 is the critical signaling molecule for Ang II-induced cardiac hypertrophy and remodeling. Thus, ASK1 is proposed to be a potential therapeutic target for cardiac diseases. 14665690_ASK1 deleted mice had significantly smaller increases in left ventricular end-diastolic and end-systolic ventricular dimensions. 14668338_results suggest that nitric oxide mediates the interferon-gamma-induced inhibition of apoptosis signal regulating kinase 1(ASK1) in L929 cells through a thiolredox mechanism 15864310_complex of the adaptor molecule TRAF6 and ASK1 and subsequent activation of the ASK1-p38 pathway 15938714_A functional analysis of ASK1 was performed in ASK knockout stem cells. 15953587_These data suggest that ASK1 is involved in necrosis as well as apoptosis and that ASK1-dependent necrosis is likely to contribute to myocardial cell death in the ischemia-reperfused heart. 15976330_ASK1 pathway promotes early angiogenesis by inducing inflammatory cell infiltration and VEGF and MCP-1 expression 16109778_predisposition of Fanconi anemia type C-/- hematopoietic progenitors to apoptosis is mediated in part through altered redox regulation and Ask1 hyperactivation 16449798_increased ROS production in aged liver may alter the ratio of ASK1 and Trx-ASK1, thereby increasing the age-associated basal level of p38 MAPK pathway activity 17068291_Beta-agonist-induced cardiac injury is associated with activation of the ASK1-JNK/p38 cascade. Beneficial effects of Hsp20 are at least partially attributable to inhibition of the ASK1-signaling cascade. 17210579_Apoptosis signal-regulating kinase (ASK) 2 functions as a mitogen-activated protein kinase kinase kinase in a heteromeric complex with ASK1. 17331470_ASK1 is activated by arsenic trioxide through reactive oxygen species accumulation and may negatively regulate apoptosis in leukemic cells without activating p38 and JNK 17389227_These findings indicate that ASK1 is required for wounding-induced infiltration and activation of macrophages, which play central roles in inflammation-dependent hair regrowth in skin. 17522316_Following release from 14-3-3 inhibition, ASK1 activates p38 MAP kinase leading to p53 phosphorylation and the Bax cascade to cause cerebral endothelial cell death. 17602894_ASK1 has a spatially restricted and temporally dynamic expression pattern in both chick and mouse embryos, which includes conserved as well as species-specific expression domains 17646574_Implicated in vascular endothelial dysfunction and cardiovascular remodeling induced by NO deficiency by regulating eNOS and reduced nicotinamide-adenine dinucleotide phosphate oxidase. 17724081_These findings demonstrate that Trx, TRAF2, and TRAF6 regulate ASK1 activity by modulating N-terminal homophilic interaction of ASK1. 18195174_When exercised by swimming for 4 weeks, ASK1-/- mice showed exaggerated growth of the heart accompanied by typical characteristics of physiological hypertrophy 18211888_ROS-mediated activation of the ASK1-p38 MAPK pathway downstream of P2X(7) receptor is required for ATP-induced apoptosis in macrophages. 18519638_These findings demonstrate that ER stress-induced ASK1 activation, which is triggered by the specific interaction of Derlin-1 with SOD1(mut), is crucial for disease progression of familial ALS. 18676690_Cardiac hypertrophy is associated with altered thioredoxin and ASK-1 signaling in a mouse model of menopause. 18700144_ASK1 is activated by acetaminophin overdose, most likely via a mechanism involving thioredoxin-ASK1 dissociation, and it plays a role in induced liver injury through JNK activation 18708926_Expression of apoptosis signal-regulating kinase 1 is increased in the spinal cord of an animal model of spinal cord compression. 18718527_the selectivity of Bz-423 observed in vivo results from cell-type specific differences in redox balance and signaling by ASK1 and Bcl-2 proteins 18776134_Data show that SOCS-1 is an important regulator in IL-6-induced cytoprotection against hyperoxic acute lung injury, and that the interaction between SOCS-1 and ASK-1 promotes ubiquitin-mediated degradation both in vivo and in vitro. 18852704_ASK1 activation is an essential requirement for downstream inflammatory signaling activation and the intracellular control of mycobacteria. 18948261_MPK38 physically interacts with ASK1 in vivo and acts as a positive upstream regulator of ASK1 19004820_an essential role for ASK1 in cell death induced by ER stress. 19287004_JAK2-SOCS1 and SHP2 reciprocally regulate ASK1 phosphorylation and stability in response to cytokines. 19451227_Findings identify a novel pathway that regulates ASK1 activation and oxidant stress-induced cell death. 19581502_Results demonstrate that ASK1 is implicated in aldosterone/salt-induced cardiac inflammation and fibrosis through the enhancement of NADPH oxidase-mediated oxidative stress and the upregulation of the cardiac renin-angiotensin system. 19646509_Ask1 may interact with htt fragments and subsequently induce endoplasmic reticulum stress; regulating the amounts and activity of the Ask1 protein is a novel strategy for treatment of Huntington's disease 19695249_Ask1 regulates the expression of caspase-11 following LPS stimulation 19727340_RNA interference against ASK-1 may provide a benefit by inhibiting photoreceptor apoptosis in rd1 mice. 19766627_mIg engagement induces H2O2 production in WEHI-231 B lymphoma cells, leading to activation of ASK1-JNK1 pathway, creating a feedback amplification loop of ROS-ASK/JNK that leads to loss of DeltaPsim and finally apoptosis. 19788501_Activation of ASK1 largely co-localizes with TNFR1, and activation of Etk largely co-localizes with TNFR2 in vascular endothelium cells and cardiomyocytes. 19815822_ASK1 does not directly regulate the cardiac hypertrophic response in vivo, but it does alter cell death and propensity to cardiomyopathy, in part, through a calcineurin-dependent mechanism 19931259_ASK1 controls the development of intestinal inflammation and colitis-associated tumorigenesis through the regulation of innate immunity. 20006657_Our findings thus demonstrate novel behavioral functions of ASK1, including regulation of locomotor activity, novelty preference, and motor coordination with dopaminergic transmission. 20385180_DJ-1 has anti-oxidant and cytoprotective properties by regulating the thioredoxin 1 ASK1 complex and controlling the availability of ASK1 to effect apoptosis 20489729_results suggest that ASK1 activation is involved in normal tension glaucoma-like pathology in both neural and glial cells 20568119_the induction of myostatin by trichostatin A treatment in myocytes is in part through ASK1-MKK3/6-p38 MAPK and ASK1-MKK4-JNK signaling pathways. Activation of p38 MAPK and JNK axis is necessary, but not sufficient for TSA-induced myostatin expression. 20609399_ASK1 may be critical for the development of rheumatoid arthritis (RA) and ASK1 may be involved in the production of proinflammatory mediators in response to TNF-alpha stimulation in the RA joint. 20844314_The ASK1-Signalosome regulates p38 MAPK activity in response to levels of endogenous oxidative stress in the Klotho mouse models of aging. 20935579_ASK1 is implicated in cardiac inflammation and fibrosis, and vascular endothelial dysfunction caused by high-salt diet, through the enhancement of oxidative stress 21187402_Apoptosis signal-regulating kinase 1 and cyclin D1 compose a positive feedback loop contributing to tumor growth in gastric cancer 21488081_Apoptosis signal-regulating kinase 1 inhibits hepatocarcinogenesis by controlling the tumor-suppressing function of stress-activated mitogen-activated protein kinase. 21527753_Suggest that ASK1 suppresses hyperlipidemia-induced atherosclerosis via increased macrophage apoptosis and that ASK1 may cause pronounced plaque vulnerability via necrotic core development. 21803338_Our results suggest the increased Ask1 expression induce apoptotic cell death after ischemia/reperfusion. 22052879_ASK-1 is an important regulator of lung injury and apoptosis 22167179_TAK1 and its adapter protein, TAB2, reciprocally regulate both TAK1- and ASK1-mediated signaling pathways to direct the activations of NF-kappaB and AP-1. 22253830_ASK1 is an important effector of MPTP-induced toxicity. 22337877_ASK1 and TAK1 function as MAP3 kinases in Cdo-mediated p38MAPK activation to promote myogenic differentiation. 22720096_Results indicate that the anti-inflammatory properties of DCO-6 was through inhibition of reactive oxygen species-dependent activation of TRAF6-ASK1-p38 pathway. 23087182_Injection of vectorised miR-20a mimics to mice led to a global decrease in ASK1 protein expression. 23178077_ASK1 accelerates mechanical injury-induced vascular remodeling with activated smooth muscle cell migration. 23416061_ER stress-induced ASK1-p38 activation, which is triggered by the accumulation of Ins(C96Y), plays an important role in the pathogenesis of diabetes. 23747528_These results indicate that Trx functions as a physiological inhibitor of MPK38, which plays an important role in inducing ASK1-, TGF-beta-, and p53-mediated activity 23914844_OXO can potentially inhibit several biomarkers related to inflammation through inhibition of ROS-mediated activation of TRAF6-ASK1-p38 pathway. 23921222_The ASK1 signaling pathway regulates the OVA-induced asthmatic phenotype, specifically airway hyperreactivity sensitivity and cytokine production 23982205_Data indicate that ASK1 activation stimulated the activity of the transcription factor FoxO3a, which increased the abundance of the apoptosis-promoting adaptor protein TRADD, leading to activation of caspase 8. 24021285_The present study suggests that ROS-inducing ASK1 may be an important step in the pathogenesis of 3-NP infused striatal lesions in murine brains. 24357833_ASK1 regulates neurogenesis after ischemic brain injury through the p38 MAP kinase pathway. 24371084_ASK1 knockout and ASK1-inhibited mice were protected from cognitive decline due to bilateral common carotid stenosis. The oxidative stress-ASK1-p38 cascade impairs cognition by disrupting endothelial tight junctions in the blood-brain barrier. 24523853_Chop and Ask1 do not regulate rod survival or retinal degeneration directly but may be involved in regulating secondary pathological responses such as inflammation that are upregulated during later stages of disease progression 24736726_ASK1 in sensitized CD41 T cells appears to play a major role in CHS by facilitating IL-17 production. 24938188_The neurotoxicity of 5-S-cysteinyldopamine is mediated by the early activation of ERK1/2 followed by the subsequent activation of ASK1/JNK1/2 pro-apoptotic signalling 25087120_Flip preserves cardiac functions and inhibits cardiac hypertrophy partially by blocking ASK1/P38 signaling. 25249581_Data show that ASK1 plays a causal role in diabetes-induced ER stress and apoptosis. 25298527_ASK1 is an important upstream activator of p38 and JNK signaling in the obstructed kidney 25383781_Study demonstrates that ASK1 is not involved in beta-cell function and dysfunction but controls stress-induced beta-cell death. 25393478_Inhibition of ASK1 enhances endochondral bone formation by increasing chondrocyte survival. 25628390_ASK1 inhibition reduced apoptosis and reduced mitochondrial reactive oxygen species generation in cardiac hypertrophy. 25713304_CTSB protein functions as a necessary modulator of hypertrophic response by regulating TNF-alpha/ASK1/JNK signaling pathway involved in cardiac remodeling. 25752609_Because the phosphorylation site mutants of NR4A2 cannot rescue the cell death-promoting activity, ASK1-p38 pathway-dependent phosphorylation and subsequent cytoplasmic translocation of NR4A2 may be required for oxidative stress-induced cell death. 26044555_Our results provided the evidence that high-fat diet itself causes cognitive impairment and ASK1 participates in such cognitive impairment. 26164633_blocking MPTP-mediated TNF signaling through intrathecal administration of TNF-neutralizing antibody prevented Trx1 oxidation and downstream ASK1-p38 MAPK activation 26173459_ASK1 may mediate the teratogenicity of diabetes through activating the JNK1/2-ER stress pathway and inhibiting cell cycle progression, thereby impeding the cardiogenesis pathways essential for ventricular septation and outflow tract development. 26243192_Data show that deletion of either MAPKKK5 ASK1 or MAPKKK6 ASK2 in mice promoted the replication of influenza A virus in the lung. 26405834_ASK1 MAP kinase signaling cascade is an important regulator of chondrocyte terminal differentiation. 26452228_data highlight the importance of ASK1/p38 MAPK pathway in the brain and identify Klotho as a possible anti-oxidant effector 26728245_ASK1 mediates 3-NP toxicity and regulates C1q level through the astrocyte TGF-beta. 26797817_ASK1 mediates astrocyte activation and leads to glial scar formation after cerebral ischaemia. 26807721_ASK1 deletion in vivo significantly suppresses hyperoxia-induced elevation of inflammatory cytokines (i.e. IL-1beta and TNF-alpha), cell apoptosis in the lung, and recruitment of immune cells 27045525_ASK1 signalling regulates brown and beige adipocyte function. 27063938_Lysine 29-linkage of ASK1 by Skp1-Cullin 1-Fbxo21 ubiquitin ligase complex is required for antiviral innate response. 27450812_Conversely, treatment with LY294002 (a selective inhibitor of Akt1) reversed the effects of quercetin. In conclusion, these findings highlight the important role of quercetin in protecting against cognitive deficits and inhibiting neuronal apoptosis via the Akt signaling pathway. We believe that quercetin might prove to be a useful therapeutic component in treating cerebral I/R diseases in the near future. 28028021_These results suggest that the platelet Ask1 plays an important role in regulation of hemostasis and thrombosis. 28360100_These results demonstrate that trans-fatty acids promote extracellular ATP-induced apoptosis by targeting ASK1 and indicate novel TFA-associated pathways leading to inflammatory signal transduction and cell death that underlie the pathogenesis and progression of trans-fatty acids-induced atherosclerosis. 28739531_Oxidative and endoplasmic reticulum stress-dependent ASK1 activation in steatotic hepatocytes and Kupffer cells sensitizes mice fatty liver to ischemia/reperfusion injury. 28753204_Findings offer insight into positive regulation of Akt signaling through P2Y12 phosphorylation as well as MAPK signaling in platelets by ASK1. 28931753_study provides ASK1 as a therapeutic target for the treatment of ovarian cancer and other transcoelomic metastasis cancers 28939911_Study shows that Cib1 negatively regulates degeneration of dopaminergic neurons in a mouse model of Parkinson's disease using 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Cib1 physically associated with apoptosis signal-regulating kinase 1 (Ask1) and thereby inhibited the 1-methyl-4-phenylpyrinidium-induced stimulation of the Ask1-mediated signaling cascade. 29045420_The results of this investigation indicate ASK1 inhibition prolongs keratinocyte and blastemal cell activation leading to ear regeneration. 29154282_This study demonstrated that the first evidence suggesting that ASK1 may play some role in the pathophysiology of Alzheimer's Disease and brain aging. 29353059_the ASK1/2 complex is a regulator of NLRP3 activation 29629899_the present study identifies SHP-1 and ASK signaling crosstalk as a critical regulator of IL-1alpha-driven inflammation and opens future avenues for finding novel drug targets to treat neutrophilic dermatosis 29729191_interaction between Ask1 and Card6 absolutely required for Card6 function in vivo 29958938_CARD6 is a novel protective factor against hepatic I/R injury that suppresses inflammation and liver cell death by inhibiting the ASK1 signaling pathway. 30024858_ASK1 contributes to fibrosis and dysfunction in models of kidney disease. 30031111_beta-TrCP was identified as a novel interacting partner of ASK1 that is capable of ubiquitinating and degrading ASK1 through the ubiquitin-proteasome system. These findings suggest that beta-TrCP is capable of suppressing oxidative stress-induced caspase 3-dependent apoptosis through suppression of ASK1. 30172704_Study shownd that apoptosis signal-regulating kinase 1 (ASK1) silencing and ASK1 inhibition reduced ischemic injury-induced nucleotide-binding oligomerization domain-like receptor pyrin domain-containing 2 inflammasomes in the brain and astrocyte cell line, respectively. 30188966_Data show that miR-23b could inhibit inflammation to promote wound healing by targeting apoptotic signal-regulating kinase 1 (ASK1). 30371330_RGS 6 mediated cardiac hypertrophy by directly interacting with apoptosis signal-regulating kinase 1, which further activates the P38-c- JUN N-terminal kinase 1/2 signaling pathway. Conclusions Based on our findings, RGS 6 aggravates cardiac hypertrophy, and the RGS 6-apoptosis signal-regulating kinase 1 pathway represents a potential therapeutic target to attenuate pressure overload-driven cardiac remodeling 30703849_FBXW5 facilitated the addition of Lys63-linked ubiquitin to ASK1 and thus exacerbated ASK1-c-Jun N-terminal kinase/p38 MAPK signaling, inflammation, and lipid accumulation. 30925081_ASK1 and NADPH oxidase 1 physically associate in a multiprotein signaling complex. Nox1-derived oxidants are required for TNFalpha receptor endocytosis 30961932_ASK1 can be dephosphorylated in tyrosine residue(s) by SHP-1. SHP-1 could bind to and dephosphorylate ASK1 to inhibit its activation, thus repressing apoptosis. 31178136_Trim69 might be a functionally essential inhibitor of ASK1 activation during the pathogenesis of hippocampal inflammation and apoptosis 31541591_results for the first time demonstrate that melatonin safeguards against nonalcoholic fatty liver disease by eliminating ASK1 activation via inhibiting TRAFs-mediated ASK1 deubiquitination and stabilization in a beta-arrestin-1 dependent manner. 31996485_ASK1 inhibition reduces cell death and hepatic fibrosis in an Nlrp3 mutant liver injury model. 32024889_ASK1 promotes uterine inflammation leading to pathological preterm birth. 32121597_ASK1 Mediates Nur77 Expression in T-Cell Receptor Mediated Thymocyte Apoptosis. 32242025_High fat diet-fed adipocyte-specific ASK1 knockout mice reveal increased UCP1 protein levels in inguinal adipose tissue concomitant with elevated energy expenditure, reduced obesity and ameliorated glucose tolerance. 32297339_Milk Fat Globule-Epidermal Growth Factor-Factor 8 Improves Hepatic Steatosis and Inflammation. 32445435_ARRB1 ameliorates liver ischaemia/reperfusion injury via antagonizing TRAF6-mediated Lysine 6-linked polyubiquitination of ASK1 in hepatocytes. 32544093_Chronic-plus-binge alcohol intake induces production of proinflammatory mtDNA-enriched extracellular vesicles and steatohepatitis via ASK1/p38MAPKalpha-dependent mechanisms. 32767736_Apoptosis signal-regulating kinase 1 regulates immune-mediated thrombocytopenia, thrombosis, and systemic shock. 32930631_Depletion of ASK1 blunts stress-induced senescence in adipocytes. 33166004_MicroRNA-146b-5p overexpression attenuates premature ovarian failure in mice by inhibiting the Dab2ip/Ask1/p38-Mapk pathway and gammaH2A.X phosphorylation. 33416127_ASK1/p38mediated NLRP3 inflammasome signaling pathway contributes to aberrant retinal angiogenesis in diabetic retinopathy. 33460648_Interaction of RIPK1 and A20 modulates MAPK signaling in murine acetaminophen toxicity. 33565179_ASK1 suppresses NK cell-mediated intravascular tumor cell clearance in lung metastasis. 33640602_FBXW11 deletion alleviates Alzheimer's disease by reducing neuroinflammation and amyloid-beta plaque formation via repression of ASK1 signaling. 34662438_N-acetylgalactosaminyltransferase-4 protects against hepatic ischemia/reperfusion injury by blocking apoptosis signal-regulating kinase 1 N-terminal dimerization. 34759307_A novel chemical pull-down method for endogenous kinases using analog sensitive kinase allele (ASKA) technology identified an ASK1 interactor in brown adipocytes, receptor-interacting serine/threonine-protein kinase 2 (RIPK2). 35101972_ASK1 signaling regulates phase-specific glial interactions during neuroinflammation. 35104016_Activation of osteoblast ferroptosis via the METTL3/ASK1-p38 signaling pathway in high glucose and high fat (HGHF)-induced diabetic bone loss. 35438255_Phosphoproteomics identifies pathways underlying the role of receptor-interaction protein kinase 3 in alcohol-associated liver disease and uncovers apoptosis signal-regulating kinase 1 as a target. 35471608_ASK1 is a novel molecular target for preventing aminoglycoside-induced hair cell death. 36316322_Thioredoxin-interacting protein deficiency protects against severe acute pancreatitis by suppressing apoptosis signal-regulating kinase 1. 36473935_ASK1-ER stress pathway-mediated fibrotic-EV release contributes to the interaction of alveolar epithelial cells and lung fibroblasts to promote mechanical ventilation-induced pulmonary fibrosis. |
ENSG00000197442 |
MAP3K5 |
162.267229 |
0.1356968172 |
-2.881541 |
0.26114196 |
116.733104 |
0.00000000000000000000000000328418590100755533325564242547840974356334973013916591498146296898241161717202363234946460579521954059600830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000002058630438030744059878885254016290644551042028411157004193231960970404334498873311076749814674258232116699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
38.6810994623744 |
7.03995574993505 |
285.055318699199 |
34.3635813331302 |
| ENSMUSG00000071540 |
100504518 |
3425401B19Rik |
protein_coding |
D3Z1D3 |
FUNCTION: Plays an important role in cardiomyocyte hypertrophy via activation of the calcineurin/NFAT signaling pathway. {ECO:0000269|PubMed:28717008}. |
Cytoplasm;Phosphoprotein;Reference proteome |
|
|
mmu:100504518; |
Z disc [GO:0030018]; positive regulation of calcineurin-NFAT signaling cascade [GO:0070886] |
28717008_Data suggest that Cefip (cardiac-enriched Fhl2-interacting protein) interacts with Fhl2 (four and a half LIM domains protein-2) and modulates calcineurin signaling in cardiomyocytes; Cefip is located at sarcomeric z-disc and is up-regulated in several models of cardiomegaly. |
ENSG00000177354 |
C10orf71 |
104.055692 |
2.1162570925 |
1.081515 |
0.36544344 |
8.561105 |
0.00343420858049576843348815735623702494194731116294860839843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0134196678035175855081551077319090836681425571441650390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
139.025971861216 |
35.5939367945139 |
65.6942733258988 |
12.8282936889583 |
| ENSMUSG00000071648 |
19881 |
Rom1 |
protein_coding |
P32958 |
FUNCTION: Plays a role in rod outer segment (ROS) morphogenesis (PubMed:10802659). May play a role with PRPH2 in the maintenance of the structure of ROS curved disks (By similarity). Plays a role in the organization of the ROS and maintenance of ROS disk diameter (PubMed:10802659). Involved in the maintenance of the retina outer nuclear layer (PubMed:10802659). {ECO:0000250|UniProtKB:P52205, ECO:0000269|PubMed:10802659}. |
Cell adhesion;Cell projection;Disulfide bond;Membrane;Reference proteome;Sensory transduction;Transmembrane;Transmembrane helix;Vision |
|
|
mmu:19881; |
photoreceptor outer segment [GO:0001750]; photoreceptor outer segment membrane [GO:0042622]; plasma membrane [GO:0005886]; protein homodimerization activity [GO:0042803]; camera-type eye photoreceptor cell differentiation [GO:0060219]; cell adhesion [GO:0007155]; detection of light stimulus involved in visual perception [GO:0050908]; photoreceptor cell outer segment organization [GO:0035845]; protein heterooligomerization [GO:0051291]; protein homooligomerization [GO:0051260]; protein localization to photoreceptor outer segment [GO:1903546]; regulation of gene expression [GO:0010468]; retina development in camera-type eye [GO:0060041]; retina morphogenesis in camera-type eye [GO:0060042]; retina vasculature development in camera-type eye [GO:0061298] |
20300562_The results suggested that Trp182Arg mutant Rom1 causes a decrease in the levels of wild-type Prph2 and Rom1. 22183387_ROM1 overexpression leads to negative effects on cone outer segments ultrastructure and function. 28053051_ablation of Rom1 results in the conversion of an MD/PD phenotype characterized by cone functional defects and the formation of abnormal Prph2/Rom1 complexes to an RP phenotype characterized by rod-dominant functional defects and reductions in total Prph2 protein. Thus one method by which ROM1 may act as a disease modifier is by contributing to the large variability in PRPH2-associated disease phenotype 30307502_Experiments evaluating Prph2 trafficking show that Rom1 is a key determinant of whether Prph2 complexes utilize conventional versus unconventional (Golgi bypass) secretory pathways to reach the OS. 32716032_ROM1 contributes to phenotypic heterogeneity in PRPH2-associated retinal disease. |
ENSG00000149489 |
ROM1 |
256.620639 |
0.2535080772 |
-1.979896 |
0.16853459 |
136.873732 |
0.00000000000000000000000000000012850193995932062926086623763094088957862652533981866600346593556106444989847815475215997427227421212592162191867828369140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000107546238981339105868715496091060256340690571630878501700354791159891740541694471477995875829947181046009063720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
104.612363308728 |
12.2485548410372 |
412.65889611174 |
33.0782571389399 |
| ENSMUSG00000071847 |
494504 |
Apcdd1 |
protein_coding |
Q3U128 |
FUNCTION: Negative regulator of the Wnt signaling pathway. Inhibits Wnt signaling in a cell-autonomous manner and functions upstream of beta-catenin. May act via its interaction with Wnt and LRP proteins (By similarity). {ECO:0000250}. |
Cell membrane;Glycoprotein;Membrane;Reference proteome;Signal;Transmembrane;Transmembrane helix;Wnt signaling pathway |
|
|
mmu:494504; |
plasma membrane [GO:0005886]; identical protein binding [GO:0042802]; Wnt-protein binding [GO:0017147]; astrocyte cell migration [GO:0043615]; hair follicle development [GO:0001942]; negative regulation of Wnt signaling pathway [GO:0030178]; regulation of odontogenesis of dentin-containing tooth [GO:0042487]; Wnt signaling pathway [GO:0016055] |
15465500_mouse Drapc1 is a novel in vivo target gene of Wnt/beta-catenin signaling pathway 26170146_Suggest that APCDD1 modulates the gene expression of Wnt- and EK-related signaling molecules at the cap stage of tooth development, and is involved in tooth cusp patterning by modulating epithelial rearrangement. 28242765_these novel findings suggest that APCDD1 positively regulates adipogenic differentiation and that its down-regulation by miR-130 during diet-induced obesity may contribute to impaired adipogenic differentiation and obesity-related metabolic disease. |
ENSG00000154856 |
APCDD1 |
31.893590 |
3.3129552160 |
1.728119 |
0.47596118 |
13.131611 |
0.00029035499575448130323007678299518374842591583728790283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0015485599773572334726667865112403887906111776828765869140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
47.7839204041977 |
16.0519790671947 |
14.4233523513851 |
3.84345926620711 |
| ENSMUSG00000071862 |
107065 |
Lrrtm2 |
protein_coding |
Q8BGA3 |
FUNCTION: Involved in the development and maintenance of excitatory synapse in the vertebrate nervous system. Regulates surface expression of AMPA receptors and instructs the development of functional glutamate release sites. Acts as a ligand for the presynaptic receptors NRXN1-A and NRXN1-B (By similarity). {ECO:0000250}. |
Cell membrane;Glycoprotein;Leucine-rich repeat;Membrane;Postsynaptic cell membrane;Reference proteome;Repeat;Signal;Synapse;Transmembrane;Transmembrane helix |
|
|
mmu:107065; |
excitatory synapse [GO:0060076]; extracellular space [GO:0005615]; GABA-ergic synapse [GO:0098982]; glutamatergic synapse [GO:0098978]; hippocampal mossy fiber to CA3 synapse [GO:0098686]; membrane [GO:0016020]; postsynaptic density membrane [GO:0098839]; postsynaptic specialization membrane [GO:0099634]; Schaffer collateral - CA1 synapse [GO:0098685]; neurexin family protein binding [GO:0042043]; long-term synaptic potentiation [GO:0060291]; negative regulation of receptor internalization [GO:0002091]; positive regulation of synapse assembly [GO:0051965]; regulation of postsynaptic density assembly [GO:0099151]; synapse organization [GO:0050808] |
21788371_The LRRTM2 acts redundantly to maintain excitatory synapses and that synapse elimination caused by the absence of LRRTM2 is promoted by synaptic activity and mediated by a postsynaptic Ca(2+)/CaM-dependent signaling pathway. 23931994_This study demonistrated that LRRTM2 is essential for maintenance of long-term potentiation in mice hippocampus. 25527504_Data indicate that neural Cell Adhesion Molecules Lrrtm1 and Lrrtm2 mRNA expression is controlled by synaptic activity and nuclear calcium signaling. 26785044_The crystal structure of a thermostabilized LRRTM2 protein was determined. 29784826_Genetic deletion of LRRTM1,2 in vivo in CA1 neurons using Cre recombinase-expressing lentiviruses dramatically impaired long-term potentiation (LTP), an impairment that was rescued by simultaneous expression of LRRTM2, but not LRRTM4. 30262834_The Nrxn1beta-LRRTM2 interface involves Ca(2+)-mediated interactions and overlaps with the Nrxn-neuroligin interface. 35662394_Distinct but overlapping roles of LRRTM1 and LRRTM2 in developing and mature hippocampal circuits. |
ENSG00000146006 |
LRRTM2 |
137.067398 |
2.3948920394 |
1.259961 |
0.21752934 |
33.376872 |
0.00000000759214986271806574397596502606216928299431856430601328611373901367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000938240690652298806291459480795336123293282071244902908802032470703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
196.540339923974 |
29.7467375048248 |
82.0664717635065 |
9.66728654326644 |
| ENSMUSG00000071984 |
68655 |
Fndc1 |
protein_coding |
A0A8V5KYN9 |
Human_homologues FUNCTION: May be an activator of G protein signaling. {ECO:0000250}. |
Proteomics identification;Reference proteome;Signal |
|
|
|
cell-cell junction [GO:0005911]; cytoplasm [GO:0005737]; mitochondrial membrane [GO:0031966]; nuclear speck [GO:0016607]; plasma membrane [GO:0005886]; cellular response to hypoxia [GO:0071456]; positive regulation of cardiac muscle cell apoptotic process [GO:0010666]; positive regulation of protein phosphorylation [GO:0001934]; regulation of protein transport [GO:0051223] |
Human_homologues 14657397_mechanical stretching of FN-III1 may induce fibrillogenesis through a partially unfolded intermediate 16098131_MEL4B3 is a novel mRNA induced in skin tumors and regulated by TGF-beta and pro-inflammatory cytokines 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 26826188_Knockdown of AGS8 inhibited vascular endothelial growth factor (VEGF)-induced tube formation, as well as VEGF-stimulated cell growth and migration. 27693493_these studies have revealed a novel microRNA-1207-3p/FNDC1/FN1/AR regulatory pathway in prostate cancer. 29649427_activator of G-protein signaling 8 regulated the trafficking of VEGFR-3 to the plasma membrane 31530096_The findings elaborated the biological role of fibronectin type III domain containing 1 in gastric cancer and potential mechanism of action, possibly providing a new insight for future clinical diagnosis or even molecular therapy 33899120_The silencing of FNDC1 inhibits the tumorigenesis of breast cancer cells via modulation of the PI3K/Akt signaling pathway. 35255438_Up-regulated FNDC1 accelerates stemness and chemoradiation resistance in colorectal cancer cells. 36073217_[FNDC1 is highly expressed in lung adenocarcinoma and closely related with poor prognosis]. |
ENSG00000164694 |
FNDC1 |
2897.799331 |
6.1520200581 |
2.621060 |
0.08477087 |
887.864313 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000426572744228789918512381938237601007944351495506662658515268588668281 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000059068691273572071133105887779530327566096419875377012608267985969837 |
Yes |
No |
4955.45067960019 |
291.343362746974 |
805.499759878233 |
36.9809778308286 |
| ENSMUSG00000072082 |
12449 |
Ccnf |
protein_coding |
P51944 |
FUNCTION: Substrate recognition component of a SCF (SKP1-CUL1-F-box protein) E3 ubiquitin-protein ligase complex which mediates the ubiquitination and subsequent proteasomal degradation of target proteins (By similarity). The SCF(CCNF) E3 ubiquitin-protein ligase complex is an integral component of the ubiquitin proteasome system (UPS) and links proteasome degradation to the cell cycle (By similarity). Mediates the substrate recognition and the proteasomal degradation of various target proteins involved in the regulation of cell cycle progression and in the maintenance of genome stability (By similarity). Mediates the ubiquitination and subsequent proteasomal degradation of CP110 during G2 phase, thereby acting as an inhibitor of centrosome reduplication (By similarity). In G2, mediates the ubiquitination and proteasomal degradation of CDC6, thereby suppressing DNA re-replication and preventing genome instability (By similarity). Involved in the ubiquitination and degradation of the substrate adapter CDH1 of the anaphase-promoting complex (APC/C), thereby acting as an antagonist of APC/C in regulating G1 progression and S phase entry (By similarity). May play a role in the G2 cell cycle checkpoint control after DNA damage, possibly by promoting the ubiquitination of MYBL2/BMYB (By similarity). {ECO:0000250|UniProtKB:P41002}. |
Alternative splicing;Cell cycle;Cell division;Cyclin;Cytoplasm;Cytoskeleton;Mitosis;Nucleus;Phosphoprotein;Reference proteome;Ubl conjugation;Ubl conjugation pathway |
|
|
mmu:12449; |
cell junction [GO:0030054]; centriole [GO:0005814]; centrosome [GO:0005813]; cyclin-dependent protein kinase holoenzyme complex [GO:0000307]; cytoplasm [GO:0005737]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; SCF ubiquitin ligase complex [GO:0019005]; anaphase-promoting complex binding [GO:0010997]; cyclin-dependent protein serine/threonine kinase regulator activity [GO:0016538]; cell division [GO:0051301]; mitotic cell cycle phase transition [GO:0044772]; negative regulation of centrosome duplication [GO:0010826]; placenta development [GO:0001890]; protein ubiquitination [GO:0016567]; re-entry into mitotic cell cycle [GO:0000320]; regulation of cell cycle [GO:0051726]; regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0000079]; SCF-dependent proteasomal ubiquitin-dependent protein catabolic process [GO:0031146] |
14993286_CycF(-/-) animals, with a myriad of developmental anomalies due in large part to failures in yolk sac and chorioallantoic placentation, die around embryonic day 10.5. 20596027_SCF(Cyclin F)-mediated degradation of CP110 is required for the fidelity of mitosis and genome integrity 31577344_Pathogenic mutations in the ALS gene CCNF cause cytoplasmic mislocalization of Cyclin F and elevated VCP ATPase activity |
ENSG00000162063 |
CCNF |
791.314281 |
2.9211744801 |
1.546549 |
0.22640503 |
43.780803 |
0.00000000003672899715813345035061324447740206938090024024745616770815104246139526367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000006134386893779481432473182037242147995881325073241896461695432662963867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1210.48390014727 |
190.744359100923 |
414.382608231739 |
49.1070513432136 |
| ENSMUSG00000072109 |
621875 |
A530040E14Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
47.184654 |
6.2432706743 |
2.642302 |
0.54498174 |
20.974596 |
0.00000465414058711609348461960347642651925070822471752762794494628906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000365610466338072871293925214519759947506827302277088165283203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
76.6483005792002 |
29.6476319086033 |
12.2769465841323 |
3.78300883171924 |
| ENSMUSG00000072244 |
94088 |
Trim6 |
protein_coding |
Q8BGE7 |
FUNCTION: E3 ubiquitin ligase that plays a crucial role in the activation of the IKBKE-dependent branch of the type I interferon signaling pathway (PubMed:24882218). In concert with the ubiquitin-conjugating E2 enzyme UBE2K, synthesizes unanchored 'Lys-48'-linked polyubiquitin chains that promote the oligomerization and autophosphorylation of IKBKE leading to stimulation of an antiviral response (PubMed:24882218). Ubiquitinates also MYC and inhibits its transcription activation activity, maintaining the pluripotency of embryonic stem cells (PubMed:22328504). {ECO:0000269|PubMed:22328504, ECO:0000269|PubMed:24882218}. |
Coiled coil;Cytoplasm;Metal-binding;Reference proteome;Transferase;Ubl conjugation pathway;Zinc;Zinc-finger |
PATHWAY: Protein modification; protein ubiquitination. {ECO:0000269|PubMed:22328504}. |
|
mmu:94088; |
cytoplasm [GO:0005737]; cytosol [GO:0005829]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA-binding transcription factor binding [GO:0140297]; identical protein binding [GO:0042802]; protein homodimerization activity [GO:0042803]; protein kinase binding [GO:0019901]; protein tyrosine kinase binding [GO:1990782]; protein-macromolecule adaptor activity [GO:0030674]; ubiquitin protein ligase activity [GO:0061630]; zinc ion binding [GO:0008270]; cellular response to interferon-beta [GO:0035458]; cellular response to virus [GO:0098586]; free ubiquitin chain polymerization [GO:0010994]; innate immune response [GO:0045087]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of gene expression [GO:0010629]; negative regulation of stem cell differentiation [GO:2000737]; negative regulation of viral genome replication [GO:0045071]; positive regulation of autophagy [GO:0010508]; positive regulation of cytokine production involved in immune response [GO:0002720]; positive regulation of defense response to virus by host [GO:0002230]; positive regulation of gene expression [GO:0010628]; positive regulation of I-kappaB kinase/NF-kappaB signaling [GO:0043123]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of peptidyl-serine phosphorylation [GO:0033138]; positive regulation of peptidyl-threonine phosphorylation [GO:0010800]; positive regulation of transcription regulatory region DNA binding [GO:2000679]; positive regulation of type I interferon-mediated signaling pathway [GO:0060340]; protein polyubiquitination [GO:0000209]; protein ubiquitination [GO:0016567]; regulation of gene expression [GO:0010468]; regulation of protein localization [GO:0032880]; regulation of viral entry into host cell [GO:0046596]; response to lipopolysaccharide [GO:0032496] |
22328504_TRIM6 regulates the transcriptional activity of Myc during the maintenance of cell pluripotency, suggesting that TRIM6 functions as a novel regulator for Myc-mediated transcription in embryonic stem cells. 31171760_these results suggest that TRIM6 aggravates MI/R injury through promoting IKKepsilon/STAT1 activation-dependent cardiomyocyte apoptosis 36654781_TRIM6 Reduces Ferroptosis and Chemosensitivity by Targeting SLC1A5 in Lung Cancer. |
ENSG00000121236 |
TRIM6 |
13.732774 |
9.6628381823 |
3.272447 |
1.10438965 |
7.450992 |
0.00634014867425502105097168126235374074894934892654418945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0227230928485299965569055302694323472678661346435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
24.8440839967873 |
20.9308894473481 |
2.57109593765113 |
1.71699098460176 |
| ENSMUSG00000072324 |
667014 |
Gm8420 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
7.349583 |
0.1273496622 |
-2.973133 |
1.06542399 |
8.011590 |
0.00464789081477315433460484328520578856114298105239868164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0175046154980580022375580284688112442381680011749267578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.6751785795855 |
1.35266701446926 |
13.1541658640756 |
6.30687231791553 |
| ENSMUSG00000072676 |
66039 |
Tmem254 |
protein_coding |
P0DN89 |
|
Alternative splicing;Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:66039; |
membrane [GO:0016020] |
Human_homologues 16385451_Observational study of gene-disease association. (HuGE Navigator) |
ENSG00000133678 |
TMEM254 |
532.701131 |
2.3159528516 |
1.211606 |
0.12334387 |
95.086263 |
0.00000000000000000000018226717594311177161364303113076424591197825494501193735862195309527233888502451009117066860198974609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000083372180899864219195209596816026695283222874136472190973422280446669674347504042088985443115234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
741.374522137167 |
63.461399948084 |
320.116414130723 |
21.2286000870673 |
| ENSMUSG00000073002 |
53620 |
Vamp5 |
protein_coding |
Q9Z2P8 |
FUNCTION: May participate in trafficking events that are associated with myogenesis, such as myoblast fusion and/or GLUT4 trafficking. |
Cell membrane;Coiled coil;Developmental protein;Differentiation;Golgi apparatus;Membrane;Myogenesis;Phosphoprotein;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:53620; |
cell surface [GO:0009986]; cytoplasmic vesicle membrane [GO:0030659]; intercalated disc [GO:0014704]; late endosome [GO:0005770]; organelle membrane [GO:0031090]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; trans-Golgi network [GO:0005802]; cell differentiation [GO:0030154]; Golgi to plasma membrane protein transport [GO:0043001]; skeletal muscle tissue development [GO:0007519] |
12682051_The cytoplasmic domain of this protein is responsible for its correct subcellular targeting. 23180306_study found that VAMP5 was expressed in the type IIa fibers of skeletal muscle and that in cardiomyocytes, VAMP5 expression was localized to the vicinity of intercalated discs 29330887_Loss of VAMP5 in mice results in duplication of the ureter and insufficient expansion of the lung |
ENSG00000168899 |
VAMP5 |
355.396440 |
2.4103645644 |
1.269251 |
0.14888163 |
71.930908 |
0.00000000000000002228656560511536780267173659494045686362056111985609913039851903704402502626180648803710937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000007161792559010912289721944686772929655155608442973491634120364324189722537994384765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
503.677400085324 |
52.0702446858034 |
208.96316164021 |
16.7946723202233 |
| ENSMUSG00000073164 |
69732 |
2410018L13Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
88.143249 |
3.1963058888 |
1.676405 |
0.41522650 |
15.645292 |
0.00007640253045758332677266783106162506555847357958555221557617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004658780400039669094755523381934381177416071295738220214843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
138.62260519504 |
40.4505104044073 |
43.3696304478844 |
9.59844481161768 |
| ENSMUSG00000073418 |
12268 |
C4b |
protein_coding |
P01029 |
FUNCTION: Non-enzymatic component of C3 and C5 convertases and thus essential for the propagation of the classical complement pathway. Covalently binds to immunoglobulins and immune complexes and enhances the solubilization of immune aggregates and the clearance of IC through CR1 on erythrocytes. Catalyzes the transacylation of the thioester carbonyl group to form ester bonds with carbohydrate antigens (By similarity). {ECO:0000250}. |
Cell projection;Cleavage on pair of basic residues;Complement pathway;Disulfide bond;Glycoprotein;Immunity;Inflammatory response;Innate immunity;Reference proteome;Secreted;Signal;Sulfation;Synapse;Thioester bond |
Human_homologues NA; + ;NA |
|
mmu:12268; |
axon [GO:0030424]; classical-complement-pathway C3/C5 convertase complex [GO:0005601]; dendrite [GO:0030425]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; neuronal cell body [GO:0043025]; plasma membrane [GO:0005886]; symbiont cell surface [GO:0106139]; synapse [GO:0045202]; carbohydrate binding [GO:0030246]; complement binding [GO:0001848]; complement component C1q complex binding [GO:0001849]; endopeptidase inhibitor activity [GO:0004866]; complement activation [GO:0006956]; complement activation, classical pathway [GO:0006958]; immunoglobulin mediated immune response [GO:0016064]; inflammatory response [GO:0006954]; innate immune response [GO:0045087]; positive regulation of immune response [GO:0050778]; positive regulation of opsonization [GO:1903028] |
11726230_C4 modulates interstitial inflammation in experimental glomerulonephritis. 11801636_Complement C4 provides an important protective role against the development of systemic lupus erythematosus. 12115608_identified the murine follicular dendritic cell marker, FDC-M2, as complement c4. FDC-M2 was detectable at sites of immune complex-mediated inflammatory disease 12207352_C4 normally helps prevent early stages of autoimmune disease and that C4 deficiency predisposes to abnormal regulation of autoreactive B cells. 12421924_Macrophages derived from bone marrow produce sufficient C4 protein to restore the humoral response to NP-KLH hapten in C4-deficient animals when administered along with thymus-dependent antigen. 15192652_effects of complement depletion and deficiencies of C4, C5, and complement receptors 1 and 2 on mouse survival after intravenous exposure to S aureus 15390104_We found that deletion of the C4 gene does not significantly change either the time of onset or the severity and tempo of myelin oligodendrocyte-induced EAE compared with controls with a fully intact complement system. 17971229_In comparison with C57BL/6 mice, MRL/1pr mice had depressed C4 levels as early as 3 weeks of age. 19112490_The increased mortality of C4(-/-) or Factor B(-/-) mice indicates that these two pathways of complement activation are required for survival in mousepox. 19200601_Increased transcription of the complement C4 gene in the Rag1(-/-) intestine. 21707943_C3 and C4 bind to collagen and elastin in the vascular wall and have a potential role in vascular stiffness and atherosclerosis 23182714_Data indicate complement C3 deposition on peanut extract (PE) was abolished in immunoglobulin- and C4-deficient sera. 23259761_a deficiency in C4b does not alter radiation-induced lung disease in the C57BL/6 mouse model 25339671_role in suppressing autoantibody production elicited by S. pneumoniae antigens 26814963_C4 mediated synapse elimination during postnatal development 28873507_These data show that Borrelia burgdorferi OspC inhibits the classical and lectin complement pathways and competes with complement protein C2 for C4b binding. |
ENSG00000224389+ENSG00000244731 |
C4B+C4A |
59.826190 |
3.0340770726 |
1.601258 |
0.47833179 |
10.965754 |
0.00092811170349316792632654582462237158324569463729858398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0043272107338867263001680463219145167386159300804138183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
80.1668045254762 |
27.0693105663204 |
26.4221384651898 |
6.88997430885844 |
| ENSMUSG00000073599 |
68545 |
Ecscr |
protein_coding |
Q3TZW0 |
FUNCTION: Regulates endothelial chemotaxis and tube formation (By similarity). Has a role in angiogenesis and apoptosis via modulation of the actin cytoskeleton and facilitation of proteasomal degradation of the apoptosis inhibitors BIRC3/IAP1 and BIRC2/IAP2. {ECO:0000250, ECO:0000269|PubMed:19416853}. |
Alternative splicing;Angiogenesis;Apoptosis;Cell membrane;Chemotaxis;Cytoplasm;Developmental protein;Differentiation;Glycoprotein;Membrane;Phosphoprotein;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:68545; |
cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; angiogenesis [GO:0001525]; apoptotic process [GO:0006915]; cell differentiation [GO:0030154]; chemotaxis [GO:0006935]; negative regulation of angiogenesis [GO:0016525]; positive regulation of endothelial cell apoptotic process [GO:2000353]; positive regulation of proteasomal protein catabolic process [GO:1901800] |
19416853_ARIA was expressed in blood vessels during mouse embryogenesis, as well as in endothelial cells both in vitro and in vivo. 22606020_two mouse ECSCR splicing variants/isoform precursors exist and are differentially expressed in a variety of tissue types and likely involved in modulation of vascular Endothelial cell migration. 24022200_Inactivation of Ecscr enhances insulin sensitivity and may represent a new therapeutic strategy for treating metabolic syndrome. 24338479_These studies, revealed a previously undescribed mode of manipulating cardiac PI3K/Akt signaling by ARIA, thus identifying it as an attractive new target for the prevention of stress-induced myocardial dysfunction. 26692198_these results suggest that ECSCR plays a critical function in regulating lipolysis in white adipose tissue. |
ENSG00000249751 |
ECSCR |
28.578634 |
10.5880018866 |
3.404358 |
0.61852516 |
30.871814 |
0.00000002756462030014770672528014693047904160039252019487321376800537109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003121667631314868625581036582633531750730071507859975099563598632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.5726226819221 |
23.2362695057395 |
4.9653015974943 |
1.8411121458245 |
| ENSMUSG00000073787 |
100126795 |
Gm10575 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
88.521162 |
0.3473417033 |
-1.525572 |
0.41945823 |
12.837892 |
0.00033967007569387887766163003355757155077299103140830993652343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0017785680965861680024459046123297412123065441846847534179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
30.6039491738719 |
9.02387833680279 |
88.1090547974986 |
19.0276308733323 |
| ENSMUSG00000073802 |
12579 |
Cdkn2b |
protein_coding |
P55271 |
FUNCTION: Interacts strongly with CDK4 and CDK6. Potent inhibitor. Potential effector of TGF-beta induced cell cycle arrest (By similarity). {ECO:0000250}. |
3D-structure;ANK repeat;Cell cycle;Phosphoprotein;Reference proteome;Repeat;Tumor suppressor |
|
|
mmu:12579; |
cytoplasm [GO:0005737]; nucleus [GO:0005634]; cyclin-dependent protein serine/threonine kinase inhibitor activity [GO:0004861]; protein kinase binding [GO:0019901]; aging [GO:0007568]; cell cycle [GO:0007049]; cellular response to extracellular stimulus [GO:0031668]; cellular response to nutrient [GO:0031670]; cellular response to transforming growth factor beta stimulus [GO:0071560]; cellular senescence [GO:0090398]; liver development [GO:0001889]; megakaryocyte differentiation [GO:0030219]; negative regulation of cell cycle G1/S phase transition [GO:1902807]; negative regulation of cell population proliferation [GO:0008285]; negative regulation of epithelial cell proliferation [GO:0050680]; negative regulation of G1/S transition of mitotic cell cycle [GO:2000134]; negative regulation of glial cell proliferation [GO:0060253]; negative regulation of phosphorylation [GO:0042326]; positive regulation of epithelial cell differentiation [GO:0030858]; positive regulation of transforming growth factor beta receptor signaling pathway [GO:0030511]; regulation of cyclin-dependent protein serine/threonine kinase activity [GO:0000079]; regulation of G0 to G1 transition [GO:0070316]; regulation of G1/S transition of mitotic cell cycle [GO:2000045]; response to cytokine [GO:0034097]; response to organic cyclic compound [GO:0014070]; spleen development [GO:0048536] |
12642863_is not consistently inactivated in murine myeloid cells transformed in vivo by deregulated c-Myc 14681685_Ink4b is hypermethylated in murine myeloid leukemia 14757439_review experiments and present examples of new models of myeloid leukemia where retroviruses have collaborated with a transgene [Cbfbeta-MYH111 from Inv(16)] and with loss of a tumor suppressor (Ink4b) mice to induce leukemia. 14976051_regulation of the Ink4b promoter is apparently myeloid specific because both ICSBP and PU.1 are myeloid commitment factors. 15370242_review of p15INK4b, p14ARF and p16INK4a function as cell cycle inhibitors where they are involved in the inhibition of G1 phase progression, and promoter methylation in hematologic malignancies and solid tumors 17713536_data indicate that p15Ink4b can fulfil a critical backup function for p16Ink4a and provide an explanation for the frequent loss of the complete CDKN2b-CDKN2a locus in human tumours 18037485_results strongly suggest that p15(Ink4b) loss must be accompanied by additional oncogenic changes for RUNX1-ETO-associated acute myeloid leukemia to develop 18185590_Expression of exogenous p15AS in mouse embryonic stem cells caused p15 silencing and increased growth, through heterochromatin formation, as well as DNA methylation after differentiation of the embryonic stem cells 18625706_Hdac3 overexpression inhibits several cyclin-dependent kinase inhibitors, including Cdkn1a, Cdkn1b, Cdkn1c, Cdkn2b, and Cdkn2c. Hdac3-Tg mice did not develop cardiac hypertrophy at 3 months of age, unlike previously reported Hdac2-Tg mice 18836456_Results indicate that Jhdm1b is an H3K36 demethylase that regulates cell proliferation and senescence through p15(Ink4b). 20457873_new animal model demonstrates experimentally that p15Ink4b is a tumor suppressor for myeloid leukemia, and its loss may play an active role in the establishment of preleukemic conditions 21619792_mRNA expression of p15 gene in mouse bone marrow cells decreases after exposure to 1,4-benzoquinone, but the CpG islands methylation status in promoter is not affected. 22226369_Increased gene dosage of the Ink4/Arf locus does not attenuate atherosclerosis development in hypercholesterolaemic mice. 22227036_findings indicate that in lymphoid tissue the inactivation of both p15 alleles is unlikely to be the first event in tumor development 22461492_p15Ink4b is an important modulator of cDC development and has a novel function for this tumor suppressor in the regulation of adaptive immune responses. 23045274_Meningioma progression in mice triggered by Nf2 and Cdkn2ab inactivation. 23162013_Reduced CDKN2B expression and increased p53-dependent smooth muscle cell apoptosis may be one mechanism underlying the 9p21.3 association with aneurysmal disease. 23699408_data demonstrate in vivo that Mule suppresses Ras-mediated tumorigenesis by preventing an accumulation of c-Myc/Miz1 complexes that mediates p21 and p15 down-regulation 24449168_Data indicate that loss of cyclin-dependent kinase inhibitor p15 (p15Ink4b) collaborates with oncogene fusion protein Nup98-HoxD13 transgene in the development of predominantly myeloid neoplasms. 24531546_Loss of CDKN2B promotes atherosclerosis by increasing the size and complexity of the lipid-laden necrotic core through impaired efferocytosis. 24632607_Radiation-induced double strand breaks cooperate with loss of Ink4 and Arf tumor suppressors to generate high-grade gliomas that are commonly driven by Met amplification and activation. 25157181_Cdk4 and Cdk6 cooperate in hematopoietic tumor development and suggest a role for Cdk6 in sequestering INK4 proteins away from Cdk4. 25550505_When overexpressed in naked mole rat or human cells, pALT(INK4a/b) has stronger ability to induce cell-cycle arrest than either p15(INK4b) or p16(INK4a). 25882552_Control of CD8 T cell proliferation and terminal differentiation by active STAT5 and CDKN2A/CDKN2B. 26418719_Loss of Nf2 and Cdkn2a/b have synergistic effects with PDGF-B overexpression promoting meningioma malignant transformation. 26596284_Loss of CDKN2B may not only promote cardiovascular disease through the development of atherosclerosis but may also impair TGFbeta signaling and hypoxic neovessel maturation. 27608041_miR-541 contributes to microcystin-LR-induced testicular toxicity by regulating the expression of p15 and promoting apoptosis. 28074012_The expression of three tumor suppressor genes encoded in the INK4/ARF locus (p15(INK4b), p16(INK4a), and p19(ARF)) was decreased in E6AP(-/-) embryo fibroblasts. 28346462_Data show that the Wnt-effector hepatocyte nuclear factor 1-alpha (Tcf1) is recruited to and triggers transcription of the Ink4/Arf tumor suppressor locus, such as as p15Ink4b, p16Ink4a and p19Arf. 28892048_Pancreatic cancer was induced in adult mice by the combination of KRASG12D overexpression and loss of Tp53 and Cdkn2a only if Cdkn2b was concomitantly inactivated. inactivation of both Cdkn2b and Cdkn2a was necessary for Rb phosphorylation and to encompass oncogene-induced cellular senescence. 30785991_DNMT1-mediated DNA hypermethylation can enhance the process of corneal epithelial wound healing by directly targeting miR-200a and CDKN2B. 30926782_These results point to a critical role of the Cdkn2ab locus in keeping the oncogenic potential of physiological levels of WNT signaling in check. 32271879_Combined deletion of Bap1, Nf2, and Cdkn2ab causes rapid onset of malignant mesothelioma in mice. 33824349_Dominant role of CDKN2B/p15INK4B of 9p21.3 tumor suppressor hub in inhibition of cell-cycle and glycolysis. 33900379_B-cell receptor signaling and genetic lesions in TP53 and CDKN2A/CDKN2B cooperate in Richter transformation. |
ENSG00000147883 |
CDKN2B |
17.385444 |
0.0053853901 |
-7.536733 |
1.37342563 |
37.347818 |
0.00000000098831411619889707261699914378743486720324540328874718397855758666992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000139388894610570376727390405451056931696030005696229636669158935546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.177138516697941 |
0.195284282573493 |
32.8924208094087 |
13.0762028956095 |
| ENSMUSG00000074466 |
545539 |
Gm15417 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
139.448195 |
0.4935545948 |
-1.018718 |
0.30910616 |
10.749777 |
0.00104291857931788477936718706473584461491554975509643554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0047964178140609964318130664651107508689165115356445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
90.2902968283238 |
19.493593107744 |
182.938823338843 |
29.1808603135358 |
| ENSMUSG00000074479 |
|
Rtraf-ps |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
10.035916 |
0.0091241320 |
-6.776097 |
1.50722448 |
21.002385 |
0.00000458712067525261002395040380030799553878750884905457496643066406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000361080952237662438611583148873052095950697548687458038330078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.18395422235573 |
0.229100175993865 |
20.1612847151318 |
10.7703979273906 |
| ENSMUSG00000074483 |
12096 |
Bglap |
protein_coding |
P86546 |
FUNCTION: Constitutes 1-2% of the total bone protein. It binds strongly to apatite and calcium. |
Biomineralization;Calcium;Cleavage on pair of basic residues;Disulfide bond;Gamma-carboxyglutamic acid;Metal-binding;Reference proteome;Secreted;Signal |
|
This gene encodes one of the most abundant non-collagenous proteins in bone tissue that is localized to the mineralized matrix of bone. The encoded preproprotein undergoes proteolytic processing and post-translational gamma carboxylation to generate a mature, calcium-binding protein. Mice lacking the encoded protein develop abnormalities of bone remodelling. This gene is located adjacent to two other osteocalcin-related genes on chromosome 3. [provided by RefSeq, Oct 2015]. |
mmu:12096; |
cell projection [GO:0042995]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; perikaryon [GO:0043204]; rough endoplasmic reticulum [GO:0005791]; vesicle [GO:0031982]; calcium ion binding [GO:0005509]; hydroxyapatite binding [GO:0046848]; structural constituent of bone [GO:0008147]; biomineral tissue development [GO:0031214]; bone development [GO:0060348]; cellular response to BMP stimulus [GO:0071773]; dephosphorylation [GO:0016311]; osteoblast differentiation [GO:0001649]; regulation of bone mineralization [GO:0030500]; regulation of cellular response to insulin stimulus [GO:1900076]; response to vitamin K [GO:0032571] |
12704187_the activation of PPARgamma inhibits osteocalcin expression both by suppressing the expression of Runx2 and by interfering with the transactivation ability of Runx2 12798773_1,25-(OH)(2)D(3)and T3 upregulated osteocalcin, depending on the stage of cell differentiation. 15131132_osteocalcin fibroblast growth factor response element activation is enhanced by MINT, the Msx2 interacting nuclear matrix target 16936265_Nell-1 reduced osterix-producing cells and increased bone sialoprotein, osteocalcin, and BMP-7 expression. 17705049_Expression of the osteoblast-specific gene OC was examined in marrow stromal cell cultures at various time points. 18821584_during osteoblast differentiation, early on, when Runx2 protein and DNA-binding are maximal, Runx2 is nearly absent from the OC promoter. Later, Runx2 is recruited to the OC promoter as Runx2 mRNA, protein, and in vitro DNA binding progressively decrease. 19162043_Results show that induction of alkaline phosphatase, type I collagen, osteocalcin, CD44, CD47 and CD51 by mevinolin is responsible for the osteoblastic differentiation of D1 cells. 21333348_Findings expand the biological importance of osteocalcin, begin to unravel its molecular mode of action, and provide the first evidence that the skeleton is an endocrine regulator of fertility. 21427224_Data show that osteocalcin synthesis was significantly reduced in the stable HSP27-transfected MC3T3-E1 cells and normal human osteoblasts. 21471200_Foxo1 is a novel negative regulator of osteoblast-specific transcription factor Runx2 and modulates IGF1/insulin-dependent regulation of osteocalcin expression in osteoblasts. 21757657_Both BGP overexpression and treatment with purified Bone Gla Protein (BGP) resulted in stabilization of hypoxia-inducible factor 1alpha (HIF-1alpha) in chondrocytes and vascular smooth muscle cells 21852324_Data show that Mkp1 KO mice displayed delayed differentiation and reduced expressions of osteocalcin (OCN) and Runx2 genes associated with osteoblast maturation and function. 22102412_BMP2 induces osteoblast differentiation through Runx2-dependent ATF6 expression, which directly regulates Oc transcription. 22350212_Alterations of responses in knockout mice that suggest participation of osteocalcin in transmission of information about those sensory stimuli. 22676849_Data from TR4 (steroid/thyroid hormone receptor 4) knockout mice suggest that TR4 binds to promoter region of osteocalcin gene and induces osteocalcin expression; this interaction maintains osteoblast activity during bone development/remodeling. 23042641_These results indicate that PP2A Calpha and its activity play a negative role in osteoblast differentiation and Osterix is a key factor responsible for regulating the expressions of Bsp and OCN during PP2A Calpha-mediated osteoblast differentiation. 23092676_alphaNAC interacts with histone deacetylase corepressors to control Myogenin and Osteocalcin gene expression. 23123502_T3 stimulates the activation of Rho-kinase in osteoblasts, which functions as a negative regulator of T3-stimulated osteocalcin synthesis 23437377_Uncarboxylated osteocalcin acts via Gprc6a to induce GLP-1 release from the gut; the stimulatory effect of uncarboxylated osteocalcin on insulin secretion is largely mediated by GLP-1. 24009262_loss of osteocalcin/Gprc6a signaling has a profound effect on beta-cell mass accrual during late pancreas morphogenesis 24046454_Osteoblast-specific expression of Fra-2/AP-1 controls adiponectin and osteocalcin expression and affects metabolism. 24074871_This study reveals that the skeleton via osteocalcin influences cognition and contributes to the maternal influence on fetal brain development. 24128197_contribution of OC-OPN to fracture toughness is related to their presence at the extrafibrillar organic-mineral interfaces 24389415_Taken together, these findings demonstrate that SMILE negatively regulates osteocalcin via a direct interaction with RUNX2 24554534_In primary adipocytes, both carboxylated and uncarboxylated osteocalcin increased basal and insulin-stimulated glucose transport. 24642469_Insulin resistance in osteoblasts led to a decrease in circulating levels of the active form of osteocalcin, thereby decreasing insulin sensitivity in skeletal muscle. 24680482_results showed an increase in the expression of Cbfa1 and OCN in fibroblasts and osteoblasts exposed to fluoride. 24896339_Data indicate that Ca2+ binding triggered a similar conformational transition in both gamma-carboxyglutamic acid-containing protein osteocalcin (Gla-OCN) and Glu-OCN from a disordered structure to a more compact/stable form. 25183373_The Twist2-Cre, Osterix-Cre and osteocalcin-Cre lines to generate conditional beta1 integrin deletions, were used to investigate the role of beta1 integrins on skeletal phenotype. 25279792_expression in offspring bone not changed by maternal high fat diet 25735975_the uncarboxylated form of osteocalcin does not trigger Akt phosphorylation and glucose uptake by itself but promotes insulin-induced glucose uptake in myotubes, probably by up-regulating Akt signaling through ERK activation 25753038_OCN is gamma-carboxylated by the gamma-carboxylase (GGCX) on three glutamic acid residues, a cellular process requiring reduction of vitamin K by a second enzyme, VKORC1. 30355501_activation of the OCN/GPR158 pathway increases the expression of RbAp48 in the aged dentate gyrus and rescues age-related memory loss. 32463812_Absence of endocrine effects in mice with this new Bglap knockout allele. 32463816_Ocn is required for bone quality and strength by adjusting the alignment of Biological apatite crystallites parallel to collagen fibrils; but it does not function as a hormone. |
ENSG00000242252 |
BGLAP |
12756.652985 |
0.1358415354 |
-2.880003 |
0.08614297 |
984.253314 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000475395484844321820616716631715487697800855715516 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000103446057502124425692796411535990932682284186403 |
Yes |
No |
2994.41788843388 |
178.902223006675 |
22043.4632083683 |
973.623522437587 |
| ENSMUSG00000074486 |
12097 |
Bglap2 |
protein_coding |
P86547 |
FUNCTION: Constitutes 1-2% of the total bone protein. It binds strongly to apatite and calcium. |
Biomineralization;Calcium;Cleavage on pair of basic residues;Disulfide bond;Gamma-carboxyglutamic acid;Metal-binding;Reference proteome;Secreted;Signal |
|
This gene encodes a preproprotein that is proteolytically processed to generate a mature protein product. This protein product is a hormone that is secreted by osteoblasts and may function in bone remodeling and energy metabolism. Homozygous knockout mice for this gene exhibit a gradual increase in bone size, density and strength, as well as elevated adiposity and impaired glucose tolerance. This gene is present in a gene cluster with other related genes on chromosome 3. [provided by RefSeq, Aug 2015]. |
mmu:12097; |
cell projection [GO:0042995]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; perikaryon [GO:0043204]; rough endoplasmic reticulum [GO:0005791]; vesicle [GO:0031982]; calcium ion binding [GO:0005509]; hydroxyapatite binding [GO:0046848]; structural constituent of bone [GO:0008147]; biomineral tissue development [GO:0031214]; bone development [GO:0060348]; cellular lipid catabolic process [GO:0044242]; cellular response to insulin stimulus [GO:0032869]; dephosphorylation [GO:0016311]; glucose homeostasis [GO:0042593]; osteoblast differentiation [GO:0001649]; pancreas development [GO:0031016]; positive regulation of insulin secretion [GO:0032024]; regulation of bone mineralization [GO:0030500]; regulation of cellular response to insulin stimulus [GO:1900076]; response to vitamin K [GO:0032571]; type B pancreatic cell proliferation [GO:0044342] |
12551949_LEF1 repressed Runx2-induced activation of the mouse osteocalcin 2 promoter in several osteoblast lineage cell lines 14634012_osteocalcin gene induction is regulated by parathyroid hormone and is mediated by multiple signaling pathways requiring OSE1 and associated nuclear proteins 15292260_HDAC3 repressed Runx2-mediated activation of the osteocalcin (mOG2) promoter. 15485875_Ocn is a Nurr1 target gene, which positions Nurr1 in the core of transcriptional factors regulating osteoblastic gene expression 15647836_expressions of osteocalcin, OPG, RANKL and RANK are regulated by menaquinone-7 in osteoblastic MC3T3E1 cells 16000305_Results describe the cooperative interactions between activating transcription factor 4 and Runx2/Cbfa1 that stimulate osteoblast-specific osteocalcin expression. 16720721_Data show that CCAAT/enhancer-binding protein delta regulates mouse osteocalcin OG2 promoter activity indirectly by interacting with Runx2 in the context of the OSE2 element, and resulting in the cooperative activation of the OG2 promoter. 16828750_PPARgamma-siRNA downregulated the expression of aP2 mRNA and lipid accumulation, whereas it upregulated the expression of osteocalcin and calcium deposition. 16936265_Nell-1 reduced osterix-producing cells and increased bone sialoprotein, osteocalcin, and BMP-7 expression. 17308994_c-Jun and JunD family members utilize distinct mechanisms that implicate differential interaction with transcriptional coactivators to regulate osteocalcin expression. 18171674_TFIIA gamma together with ATF4 and Runx2 stimulates osteocalcin promoter activity and endogenous mRNA expression. 18187540_PTH increases ATF4 expression and activity and that ATF4 is required for PTH induction of Ocn expression in osteoblasts. 18362359_results establish in WT mice the importance of osteocalcin in the regulation of glucose metabolism and fat mass 18445883_is an osteoblast-specific gene and regulates energy metabolism. 19225217_findings suggest an interrelationship between osteocalcin and MMP-13 expression in osteoblasts 19232401_Data support the idea that a stoichiometric excess of ATF4 over FIAT in mature osteoblasts releases ATF4 from sequestration by FIAT, thereby allowing ATF4 homodimerization and subsequent transactivation of the osteocalcin gene. 19463978_NFATc1 acts as a transcriptional co-repressor of osteocalcin promoter, possibly in an HDAC-dependent manner. 19563782_The present study demonstrates a novel, non-genomic action of T3 in primary mouse osteoblasts, by which T3 suppresses Src thereby stimulating osteocalcin expression. 19627841_Osteocalcin plays a role in the growth of apatite crystals in bone by increasing the degree of carbonate substitutions. 20038793_FoxO1 expression in osteoblasts regulates glucose homeostasis through regulation of osteocalcin in mice. 20534727_When the conditional deletion of Ctgf was achieved by Oc-Cre, an osteopenic phenotype was observed only in 6-month-old male mice. 21852324_Data show that Mkp1 KO mice displayed delayed differentiation and reduced expressions of osteocalcin (OCN) and Runx2 genes associated with osteoblast maturation and function. 22682502_In mouse osteoblasts,mRNA and protein expressions of OCN and Cbfalpha1 were markedly increased after treatment with low dose X-irradiation. 24956112_expression of MGP and OCN increased gradually in the murine developing tibial epiphysis, and the two mineral-associated proteins may occur at the same location during a particular period, but at different levels. 25918236_Undercarboxylated osteocalcin (Glu-OCN) controlled production of DLK1 by pancreatic beta-cells acts as a negative feedback mechanism to counteract the stimulatory effects of insulin on osteoblast production of Glu-OCN. 26719856_Uncarboxylated osteocalcin inhibits high glucose-induced reactive oxygen species production and stimulates osteoblastic differentiation by inhibiting the activation of PI3K/Akt in MC3T3-E1 cells. 27304508_Osteocalcin signaling in myofibers also accounts for most of the exercise-induced release of interleukin-6. 27564111_leptin regulates the expression of osteocalcin in growth plate chondrocytes via the ERK1/2 signaling pathway, while there is no effect on the phosphorylation of either p38 or AKT 28972540_The proprotein convertase furin is responsible for pro-OCN maturation in vitro and in vivo. 29044594_Data show that, together, osteocalcin (OC) and osteopontin (OPN) play important roles in determining bone size, shape, and strength. 30205977_OC and OPN rely on ionic interactions of their charged side chains and on hydrogen bonding to dissipate energy in bone 30302795_osteocalcin acts as a positive inducer of SCs in coculture with Leydig cells probably through stimulation of testosterone release from Leydig cells and associated signaling. 30908701_Undercarboxylated Osteocalcin Improves Insulin-Stimulated Glucose Uptake in Muscles of Corticosterone-Treated Mice. 31216497_Undercarboxylated osteocalcin downregulates pancreatic lipase expression in an ATF4-dependent manner in pancreatic acinar cells. 31401301_Osteocalcin affects bone mineral and mechanical properties in female mice. 31519471_Altered chondrocyte differentiation, matrix mineralization and MEK-Erk1/2 signaling in an INPPL1 catalytic knock-out mouse model of opsismodysplasia. 31693486_GLP-1 signaling is required for improvement of glucose tolerance by osteocalcin. 32927845_Effects of Early Life Stress on Bone Homeostasis in Mice and Humans. 33428938_Adipocyte-specific GPRC6A ablation promotes diet-induced obesity by inhibiting lipolysis. 33814269_Male but not female mice with severe osteogenesis imperfecta are partially protected from high-fat diet-induced obesity. 33851585_THE EFFECT OF DIET ENRICHED WITH PYROPHOSPHATE (E450) ON EXPRESSION OF GENES ENCODING BONE MORPHOGENETIC PROTEIN AND OSTEOCALCIN IN MOUSE EMBRYONIC MANDIBLE TISSUES. 34489478_Endothelium-specific depletion of LRP1 improves glucose homeostasis through inducing osteocalcin. 34905510_Embryonic osteocalcin signaling determines lifelong adrenal steroidogenesis and homeostasis in the mouse. |
ENSG00000242252 |
BGLAP |
9030.817783 |
0.1797385755 |
-2.476028 |
0.05832373 |
1657.009720 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2735.68682798302 |
110.625541286543 |
15220.3655815253 |
447.776175796533 |
| ENSMUSG00000074489 |
12095 |
Bglap3 |
protein_coding |
P54615 |
FUNCTION: Binds strongly to apatite and calcium. {ECO:0000250|UniProtKB:P02820}. |
Biomineralization;Calcium;Cleavage on pair of basic residues;Disulfide bond;Gamma-carboxyglutamic acid;Metal-binding;Reference proteome;Secreted;Signal |
|
|
mmu:12095; |
cell projection [GO:0042995]; cytoplasm [GO:0005737]; dendrite [GO:0030425]; extracellular region [GO:0005576]; extracellular space [GO:0005615]; Golgi apparatus [GO:0005794]; perikaryon [GO:0043204]; rough endoplasmic reticulum [GO:0005791]; vesicle [GO:0031982]; calcium ion binding [GO:0005509]; hydroxyapatite binding [GO:0046848]; structural constituent of bone [GO:0008147]; biomineral tissue development [GO:0031214]; bone development [GO:0060348]; dephosphorylation [GO:0016311]; osteoblast differentiation [GO:0001649]; regulation of bone mineralization [GO:0030500]; regulation of cellular response to insulin stimulus [GO:1900076]; response to vitamin K [GO:0032571] |
16902318_we conclude that ORG, the third gene of the mouse osteocalcin gene cluster is silent and unlikely to play a major role in mouse renal physiology |
ENSG00000242252 |
BGLAP |
196.642465 |
0.1235347461 |
-3.017011 |
0.28929429 |
99.613872 |
0.00000000000000000000001852026992933106638959629866033099416331819949216256569394148238406894169116867487900890409946441650390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000009100024243986154178979506729575662659546265298843462445994162424156570523336995393037796020507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.9232710557585 |
8.86679318444386 |
355.553983338 |
49.3159951213743 |
| ENSMUSG00000074577 |
69553 |
Ripor3 |
protein_coding |
A1L3T7 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:69553; |
|
Human_homologues 20877624_Observational study of gene-disease association. (HuGE Navigator) 35277469_Downregulated Expression of RIPOR3 Correlated with Immune Infiltrates Predicts Poor Prognosis in Oral Tongue Cancer. |
ENSG00000042062 |
RIPOR3 |
379.133779 |
0.2324015060 |
-2.105309 |
0.14663916 |
202.654622 |
0.00000000000000000000000000000000000000000000055024059353145620533140815531616783973973685383465950039869103277995422828171797635010790334543994641817357886499429212023848378976254025474190711975097656250000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000000805890838526071182912008986341404921752357463713577773415369872515815692457342310444876850716125374845605144153923293792729509732453152537345886230468750000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
140.046519311256 |
14.2592042600007 |
602.605902678512 |
42.4478238816757 |
| ENSMUSG00000074766 |
319909 |
Ism1 |
protein_coding |
A2ATD1 |
FUNCTION: Acts as an angiogenesis inhibitor. {ECO:0000269|PubMed:19874420}. |
Disulfide bond;Reference proteome;Secreted;Signal |
|
|
|
extracellular region [GO:0005576]; negative regulation of angiogenesis [GO:0016525] |
19874420_results demonstrate that Isthmin is a novel endogenous angiogenesis inhibitor with functions likely in physiological as well as pathological angiogenesis. 24675886_ISM1 transcripts are found in tissues as the anterior mesendoderm, paraxial and lateral plate mesoderm, MHB and trunk neural tube, as well as in the somites and dermomyotome. In the newborn and adult, ISM1 is prominently expressed in the lung and brain. 24956034_Given that ISM1 has been reported to have anti-angiogenic properties, these observations suggest that ISM1 is a mediator of lymphocyte effector functions and may participate in both innate and acquired immune responses. 25952901_ISM is a novel vascular permeability inducer that functions through cell-surface GRP78-mediated Src activation as well as induction of apoptosis 34348115_Isthmin-1 is an adipokine that promotes glucose uptake and improves glucose tolerance and hepatic steatosis. 34469202_Novel HIF-1-target gene isthmin1 contributes to hypoxia-induced hyperpermeability of pulmonary microvascular endothelial cells monolayers. 35046017_ISM1 protects lung homeostasis via cell-surface GRP78-mediated alveolar macrophage apoptosis. 35752760_ISM1 suppresses LPS-induced acute lung injury and post-injury lung fibrosis in mice. 36169399_Phosphoproteomic mapping reveals distinct signaling actions and activation of muscle protein synthesis by Isthmin-1. |
ENSG00000101230 |
ISM1 |
13.198556 |
0.0391509274 |
-4.674810 |
1.11132779 |
20.936071 |
0.00000474867982974882492711293097142899455320730339735746383666992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000372652710802339536357886962125718355309800244867801666259765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.948341187085892 |
0.804940665585113 |
24.2227004462164 |
9.9771819389271 |
| ENSMUSG00000074807 |
|
Gm10762 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
66.173928 |
0.4260946619 |
-1.230754 |
0.31540373 |
15.132078 |
0.00010024537594832874231199487669030645520251709967851638793945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0005971597835138613141747754298194195143878459930419921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.512646344927 |
8.70727857799018 |
92.7320848671693 |
14.6594425155464 |
| ENSMUSG00000074934 |
23892 |
Grem1 |
protein_coding |
O70326 |
FUNCTION: Cytokine that may play an important role during carcinogenesis and metanephric kidney organogenesis, as BMP a antagonist required for early limb outgrowth and patterning in maintaining the FGF4-SHH feedback loop (PubMed:12808456, PubMed:15201225). Down-regulates the BMP4 signaling in a dose-dependent manner (PubMed:15133038). Antagonist of BMP2; inhibits BMP2-mediated differentiation of osteoblasts (in vitro) (By similarity). Acts as inhibitor of monocyte chemotaxis (By similarity). {ECO:0000250|UniProtKB:O35793, ECO:0000250|UniProtKB:O60565, ECO:0000269|PubMed:12808456, ECO:0000269|PubMed:15133038, ECO:0000269|PubMed:15201225}. |
Cytokine;Disulfide bond;Glycoprotein;Reference proteome;Secreted;Signal |
|
|
mmu:23892; |
cell surface [GO:0009986]; extracellular space [GO:0005615]; BMP binding [GO:0036122]; cytokine activity [GO:0005125]; heparan sulfate proteoglycan binding [GO:0043395]; protein homodimerization activity [GO:0042803]; receptor ligand activity [GO:0048018]; transmembrane receptor protein tyrosine kinase activator activity [GO:0030297]; vascular endothelial growth factor receptor 2 binding [GO:0043184]; angiogenesis [GO:0001525]; animal organ morphogenesis [GO:0009887]; apoptotic process [GO:0006915]; cardiac muscle cell differentiation [GO:0055007]; cardiac muscle cell myoblast differentiation [GO:0060379]; cell migration involved in sprouting angiogenesis [GO:0002042]; cell morphogenesis [GO:0000902]; cell-cell signaling [GO:0007267]; collagen fibril organization [GO:0030199]; determination of dorsal identity [GO:0048263]; embryonic limb morphogenesis [GO:0030326]; endothelial cell migration [GO:0043542]; limb development [GO:0060173]; mesenchymal to epithelial transition involved in metanephros morphogenesis [GO:0003337]; negative regulation of apoptotic process [GO:0043066]; negative regulation of BMP signaling pathway [GO:0030514]; negative regulation of bone mineralization [GO:0030502]; negative regulation of bone mineralization involved in bone maturation [GO:1900158]; negative regulation of bone remodeling [GO:0046851]; negative regulation of bone trabecula formation [GO:1900155]; negative regulation of canonical Wnt signaling pathway [GO:0090090]; negative regulation of cell growth [GO:0030308]; negative regulation of chondrocyte differentiation [GO:0032331]; negative regulation of DNA-templated transcription [GO:0045892]; negative regulation of leukocyte chemotaxis [GO:0002689]; negative regulation of monocyte chemotaxis [GO:0090027]; negative regulation of osteoblast differentiation [GO:0045668]; negative regulation of osteoblast proliferation [GO:0033689]; negative regulation of osteoclast proliferation [GO:0090291]; negative regulation of pathway-restricted SMAD protein phosphorylation [GO:0060394]; positive regulation of angiogenesis [GO:0045766]; positive regulation of branching involved in ureteric bud morphogenesis [GO:0090190]; positive regulation of cell population proliferation [GO:0008284]; positive regulation of DNA-templated transcription [GO:0045893]; positive regulation of NF-kappaB transcription factor activity [GO:0051092]; positive regulation of NIK/NF-kappaB signaling [GO:1901224]; positive regulation of peptidyl-tyrosine autophosphorylation [GO:1900086]; positive regulation of protein tyrosine kinase activity [GO:0061098]; positive regulation of receptor internalization [GO:0002092]; positive regulation of signaling receptor activity [GO:2000273]; positive regulation of telomerase activity [GO:0051973]; positive regulation of transcription by RNA polymerase II [GO:0045944]; proximal/distal pattern formation [GO:0009954]; regulation of epithelial to mesenchymal transition [GO:0010717]; regulation of focal adhesion assembly [GO:0051893]; sequestering of BMP from receptor via BMP binding [GO:0038098]; signal transduction [GO:0007165]; ureteric bud formation [GO:0060676] |
15133038_gremlin modulates the cross-talk between GDF9 and BMP signaling that is necessary during follicle development 15201225_Gremlin-mediated BMP antagonism induces the epithelial-mesenchymal feedback signaling controlling metanephric kidney and limb organogenesis 15256670_Shh-expressing cells and their descendants cannot express Gremlin; proliferation of these descendants forms a barrier separating the Shh signal from Gremlin-expressing cells which breaks down the Shh-Fgf4 loop and affects limb size 15919833_study is the first to show that peripheral benzodiazepine receptor and DRM/gremlin are expressed in preadipocyte cell lines and that they are differentially regulated during adipogenesis 16606614_gremlin and insulin-like growth factor-binding proteins have roles in liver fibrogenesis 16880207_an intracellular gremlin-BMP4 interaction negatively regulates BMP4 activation and secretion 16908629_The BMP antagonist gremlin 1 (GREM1) controls gene expression in the digit-forming mesenchyme of mouse limb buds 17077323_point capacity of Drm/gremlin to interact directly with target endothelial cells and to modulate angiogenesis 17522159_initiation of metanephric kidney development requires the reduction of BMP4 activity by the antagonist gremlin 1 in the mesenchyme, which in turn enables ureteric bud outgrowth and establishment of autoregulatory GDNF/WNT11 feedback signalling 17554679_Limb deformity phenotype is characterized by developmental failure of distal limb structures and is caused either through mutation of Grem1, or by loss of a transcriptional control region. 17785465_Conditional deletion of Grem1 causes a transient increase in bone formation and bone mass. 17975199_Gremlin mRNA levels were up-regulated in the asbestos-exposed mouse lungs 17980714_Notch pathway gene expression is elevated in diabetic nephropathy, co-incident with Gremlin, and may contribute to the pathogenesis of this dise 18469115_the mechanisms of pulmonary hypertension and vascular loss in chronic lung disease and identifies gremlin 1 as a potentially important mediator of vascular changes in hypoxic pulmonary hypertension. 18505784_the proangiogenic activity of Drm is mediated by the activation of an Ang-1-dependent autocrine loop of stimulation in EC. 18594511_analysis of compound mouse mutants defective in genes comprising the positive Fgf/Shh loop provided genetic evidence that FGF signalling can repress Grem1 expression, revealing a novel Fgf/Grem1 inhibitory loop 19085239_In vitro studies using murine pulp cells revealed that gremlin inhibited BMP-4 mediated induction of Dspp. Balanced interactions between BMP agonists/antagonists are required for proper development of teeth and surrounding tissues. 19401426_Knockout mice heterozygous for grem1 gene deletion exhibit protection from the progression of diabetic kidney disease in a streptozotocin-induced model of type 1 diabetes. 19818739_This study indicates a prominent role for gremlin in central epiphyseal development compared to its role in physeal development. 20661431_Study conclude that inhibition of Gremlin exerts beneficial effects on the diabetic kidney mainly through maintenance of BMP-7 activity and that Gremlin may serve as a novel therapeutic target in the management of diabetic nephropathy. 20705941_Gremlin-mediated BMP inhibition results in activation of epithelial cells and transient fibrosis, but also induction of epithelium-protective FGF10 21268093_Nov stabilizes gremlin transcripts, and this effect is possibly mediated by AU-rich elements present in the 3'-UTR of gremlin. 21281623_This study uncovers an essential function for Six1 in the metanephric mesenchyme as an upstream regulator of Grem1 in initiating branching morphogenesis. 21303853_Nog and Grem1 cooperate to maintain a BMP signaling-free zone that is a crucial prerequisite for Hh-mediated sclerotome induction. 21412775_Grem1 is not only necessary for skeletal development, but also for postnatal skeletal homeostasis 21832168_role in the regulation of oocyte numbers and the timing of primordial follicle development 22182508_Grem1 is an inhibitor of embryonic fibroblast proliferation in vitro. 22247494_gremlin 1 was further increased in the walls of small intrapulmonary vessels of mice during the development of hypoxic pulmonary hypertension 22644325_Gremlin1 was down-regulated by BMP2 22888807_This study establishes that distant cis-regulatory regions scattered through a larger genomic landscape control the highly dynamic expression of Grem1, which is key to normal progression of mouse limb bud development. 23053782_The results demonstrate a non-redundant role for alpha(v)beta(3) in gremlin-induced angiogenesis. 23548835_endogenous Gremlin blockade inhibited TGF-beta-mediated matrix production and epithelial mesenchymal transition in kidney fibroblasts 23553804_gremlin was clearly elevated in high glucose cultured mouse podocytes, and likely employed endogenous canonical TGFbeta1/Smad signaling to induce podocyte injury. Knockdown gremlin1 by siRNA may be clinically useful in the attenuation of podocyte injury. 24003215_The present data disclose that Gremlin-1 is an endogenous antagonist of MIF and define a role for Gremlin-1/MIF interaction in atherosclerosis. 24233491_Suggest cross-talk between angiogenesis and inflammation and demonstrate a crucial role of CREB activation in the modulation of the VEGFR2-mediated proinflammatory/proangiogenic response of endothelial cells to gremlin. 24700818_A Gli-dependent cis-regulatory module (CRM), regulation of Grem1 in the mouse limb bud, is reported. 25194662_GREM1 induces fibrosis and angiogenesis in mouse peritoneum and is associated with increased solute transport in peritoneal dialysis patients. 25594183_Grem1 expression identifies distinct connective tissue stem cells in both the bone (osteochondroreticular stem cells) and the intestine (reticular stem cells). 25810250_Gremlin/VEGFR2 axis participates in renal inflammation and could be a novel target for kidney disease. 26141517_Gremlin is a key pro-fibrogenic factor in chronic pancreatitis 26495794_Data show that parathyroid hormone-related protein (PTHrP) regulates gremlin levels in pancreatic acinar and stellate cells. 27428020_Gremlin-1 protein expression was detected specifically in the lung after birth and did not result in any signs of respiratory insufficiency. CXCL10 chemokine levels correlate negatively with gremlin-1 levels in mouse and human lung. 27769098_involved in regulation of bone morphogenetic protein activities in cartilage tissues in the adult skeleton 27894090_We conclude that gremlin promotes RPE cell proliferation, migration and VEGF production possibly via activating VEGFR2-Akt-mTORC2 signaling. Gremlin could be a novel therapeutic target of ROP or other retinal vasoproliferation diseases. 27979781_Wild-type gremlin is able to bind broadly across the various regions of kidney in an heparin sulfate-dependent manner, with particularly intense binding to tubular structures in the renal cortex. In a model of chronic kidney disease, fibrotic changes in the kidney result in a loss of gremlin binding sites. Gremlin mutants with reduced affinity for heparin showed negligible binding under the same conditions. 28100499_Grem1 expression in the tubular epithelial compartment plays a significant role in the fibrotic response to renal injury in vivo. 28467108_Gremlin1 accelerates hepatic stellate cell activation through upregulation of TGF-B1, alpha-SMA, and COL1a1 expression in a liver fibrosis disease model. 29315280_remlin can aggravate podocyte lesions through the TGF-beta signaling pathway. 31694641_GREM1 is associated with metastasis and predicts poor prognosis in ER-negative breast cancer patients 32297672_Gremlin 1 depletion in vivo causes severe enteropathy and bone marrow failure. 32580008_Down-regulation of Gremlin1 inhibits inflammatory response and vascular permeability in chronic idiopathic urticaria through suppression of TGF-beta signaling pathway. 33729579_Protective role of Gremlin-1 in myocardial function. 33819486_Bone Morphogenetic Protein Pathway Antagonism by Grem1 Regulates Epithelial Cell Fate in Intestinal Regeneration. 34184027_The BMP antagonist gremlin 1 contributes to the development of cortical excitatory neurons, motor balance and fear responses. 34548488_Spatial regulation by multiple Gremlin1 enhancers provides digit development with cis-regulatory robustness and evolutionary plasticity. 34633599_Gremlin1 and TGF-beta1 protect kidney tubular epithelial cells from ischemia-reperfusion injury through different pathways. 35768509_GREM1 is required to maintain cellular heterogeneity in pancreatic cancer. |
ENSG00000166923 |
GREM1 |
1432.782267 |
0.4302630897 |
-1.216709 |
0.09255657 |
170.459054 |
0.00000000000000000000000000000000000000587361267811116841730658944707873454784505654739414230933511544985923171378590539091950003428831150482231082676776168227661401033401489257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000006578446199484508314168765716799005411492346088767156712799444916531945227025664175283124297723569978701974036994215566664934158325195312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
843.605876388914 |
54.1589437791803 |
1960.67451871762 |
91.9514840856216 |
| ENSMUSG00000074971 |
67606 |
Fibin |
protein_coding |
Q9CQS3 |
|
Disulfide bond;Endoplasmic reticulum;Glycoprotein;Golgi apparatus;Reference proteome;Secreted;Signal |
|
|
mmu:67606; |
endoplasmic reticulum [GO:0005783]; extracellular region [GO:0005576]; Golgi apparatus [GO:0005794]; protein homodimerization activity [GO:0042803]; protein kinase C signaling [GO:0070528]; response to dexamethasone [GO:0071548]; response to manganese ion [GO:0010042] |
21615908_Fibin is expressed in cerebellum, skeletal muscle and many other embryonic and adult mouse tissues suggesting not only a role during embryonic development but also in adult functions |
ENSG00000176971 |
FIBIN |
13.871299 |
0.0462216879 |
-4.435286 |
1.02477227 |
20.785924 |
0.00000513590500157663722749197890427019785875017987564206123352050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000401180025559053006793328033374734786775661632418632507324218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.35961527581137 |
1.04904536519003 |
29.4150936070054 |
11.5705060042484 |
| ENSMUSG00000075033 |
385658 |
Nxpe3 |
protein_coding |
B9EKK6 |
|
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:385658; |
membrane [GO:0016020] |
|
ENSG00000144815 |
NXPE3 |
69.052678 |
2.8905562786 |
1.531347 |
0.46266496 |
10.568756 |
0.00115015111209969766135896573899799477658234536647796630859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0052295826088067454487373630911406507948413491249084472656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
100.527448483486 |
32.7940883600116 |
34.7778900646248 |
8.48354767998996 |
| ENSMUSG00000075225 |
75973 |
Ccdc162 |
protein_coding |
Q0VG85 |
|
Alternative splicing;Coiled coil;Direct protein sequencing;Reference proteome |
|
|
|
|
|
|
|
46.699425 |
0.1453209042 |
-2.782686 |
0.54066970 |
23.567846 |
0.00000120585820646975640260093083633297084134028409607708454132080078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000106171284398539471653579585597171330846322234719991683959960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.8175539683037 |
4.91678738256313 |
88.2017218257573 |
23.8156578094683 |
| ENSMUSG00000075394 |
15423 |
Hoxc4 |
protein_coding |
Q08624 |
FUNCTION: Sequence-specific transcription factor which is part of a developmental regulatory system that provides cells with specific positional identities on the anterior-posterior axis. |
Developmental protein;DNA-binding;Homeobox;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15423; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; HMG box domain binding [GO:0071837]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific double-stranded DNA binding [GO:1990837]; anterior/posterior pattern specification [GO:0009952]; cartilage development [GO:0051216]; embryonic organ morphogenesis [GO:0048562]; embryonic skeletal system morphogenesis [GO:0048704]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357]; skeletal system development [GO:0001501] |
19037915_Quantitative RNA analysis for the genes flanking the breakpoint indicated no changes in expression levels except for some homeobox C (Hoxc) genes whose expression was elevated in developing and mature skin of the ears but not of other body regions. 19363484_HoxC4 directly activates the Aicda promoter 20855884_Esr1 bind to and activate the HOXC4 promoter to potentiate HoxC4-mediated AID induction, immunoglobulin class switch and somatic hypermutation. 21585311_These findings suggest that in lupus B cells, upregulation of HoxC4 plays a major role in dysregulation of AID expression. 23178148_NF-kappaB mediates APRIL-induced HoxC4 transcription. 26820751_In presence of these inducing agents, lipid accumulation as well as expression of HoxA1, HoxA5, HoxC4 &HoxC8 markedly enhanced. Irrespective of presence or absence of T3, insulin down regulates HoxA10. T3 results in over expression of HoxA5, HoxC4 and HoxC8 genes, whereas insulin up regulates expression of only HoxC8 |
ENSG00000198353 |
HOXC4 |
88.908689 |
2.5473821533 |
1.349015 |
0.35742388 |
13.673903 |
0.00021745545720572393441974434757923972938442602753639221191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011940452502370536499998188162408041534945368766784667968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
126.294882189524 |
31.6102886544034 |
49.5783021895207 |
9.4915054390004 |
| ENSMUSG00000075585 |
|
6330403L08Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
267.023329 |
0.4020627928 |
-1.314507 |
0.16588554 |
62.373665 |
0.00000000000000284097014422843730308078674844317097391221352969958857670462748501449823379516601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000739720636527992367810543355492277736349358840950429794247611425817012786865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
151.595417930369 |
17.4693444624894 |
377.044134993189 |
31.025727682247 |
| ENSMUSG00000076441 |
11898 |
Ass1 |
protein_coding |
P16460 |
FUNCTION: One of the enzymes of the urea cycle, the metabolic pathway transforming neurotoxic amonia produced by protein catabolism into inocuous urea in the liver of ureotelic animals. Catalyzes the formation of arginosuccinate from aspartate, citrulline and ATP and together with ASL it is responsible for the biosynthesis of arginine in most body tissues. {ECO:0000250|UniProtKB:P00966}. |
Acetylation;Amino-acid biosynthesis;Arginine biosynthesis;ATP-binding;Cytoplasm;Direct protein sequencing;Ligase;Nucleotide-binding;Phosphoprotein;Reference proteome;Urea cycle |
PATHWAY: Amino-acid biosynthesis; L-arginine biosynthesis; L-arginine from L-ornithine and carbamoyl phosphate: step 2/3. {ECO:0000250|UniProtKB:P00966}.; PATHWAY: Nitrogen metabolism; urea cycle; (N(omega)-L-arginino)succinate from L-aspartate and L-citrulline: step 1/1. {ECO:0000250|UniProtKB:P00966}. |
|
mmu:11898; |
cell body fiber [GO:0070852]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; endoplasmic reticulum [GO:0005783]; lysosome [GO:0005764]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; myelin sheath [GO:0043209]; neuron projection [GO:0043005]; neuronal cell body [GO:0043025]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; perikaryon [GO:0043204]; amino acid binding [GO:0016597]; argininosuccinate synthase activity [GO:0004055]; ATP binding [GO:0005524]; identical protein binding [GO:0042802]; toxic substance binding [GO:0015643]; acute-phase response [GO:0006953]; aging [GO:0007568]; arginine biosynthetic process [GO:0006526]; argininosuccinate metabolic process [GO:0000053]; aspartate metabolic process [GO:0006531]; cellular response to amine stimulus [GO:0071418]; cellular response to amino acid stimulus [GO:0071230]; cellular response to ammonium ion [GO:0071242]; cellular response to cAMP [GO:0071320]; cellular response to dexamethasone stimulus [GO:0071549]; cellular response to glucagon stimulus [GO:0071377]; cellular response to laminar fluid shear stress [GO:0071499]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to oleic acid [GO:0071400]; cellular response to tumor necrosis factor [GO:0071356]; cellular response to type II interferon [GO:0071346]; circadian rhythm [GO:0007623]; citrulline metabolic process [GO:0000052]; diaphragm development [GO:0060539]; kidney development [GO:0001822]; liver development [GO:0001889]; midgut development [GO:0007494]; negative regulation of leukocyte cell-cell adhesion [GO:1903038]; positive regulation of nitric oxide biosynthetic process [GO:0045429]; response to estradiol [GO:0032355]; response to glucocorticoid [GO:0051384]; response to growth hormone [GO:0060416]; response to mycotoxin [GO:0010046]; response to nutrient [GO:0007584]; response to xenobiotic stimulus [GO:0009410]; response to zinc ion [GO:0010043]; urea cycle [GO:0000050] |
15192091_Argininosuccinate synthetase has a role in preventing autotoxicity from nitric oxide overproduction 20724589_Loss of Ass1 is associated with citrullinemia type I and other hyperammonemic syndromes. 22213272_Partial argininosuccinate synthase ablation protects only in acute ethanol-induced liver injury by decreasing nitrosative stress but not in a more chronic scenario where oxidative stress and impaired fatty acid beta-oxidation are key events. 22980328_Thus, extracellular arginine fuels rapid NO production in activated macrophages, and citrulline recycling via Ass1 and Asl is a fail-safe system that sustains optimum NO production. 24224890_may contribute to liver injury by enhancing nitrosative stress 24556046_cAMP-induced Ass1 expression is important in controlling the magnitude of decidualization through regulating L-Arg level. 24884799_We demonstrate that the transgenic mouse system reported here has the merit of sensitivity and direct visualization advantage, and is ideal for annotating temporal and spatial expression profiles and the regulation mode of the ASS gene. 25033204_In diabetic mice, ablation of Ass resulted in diminished endothelium-derived nitric oxide-mediated vascular relaxation responses. 28560349_findings uncover a new function of p53 in the regulation of Akt signaling and reveal how p53, ASS1, and Akt are interrelated to each other. 28985504_ASS1 acetylation by CLOCK exhibits circadian oscillation in human cells and mouse liver, possibly caused by rhythmic interaction between CLOCK and ASS1, leading to the circadian regulation of ASS1 and ureagenesis. 34599156_Epithelial argininosuccinate synthetase is dispensable for intestinal regeneration and tumorigenesis. 35016033_Citrulline depletion by ASS1 is required for proinflammatory macrophage activation and immune responses. |
ENSG00000130707 |
ASS1 |
291.570578 |
6.6924593624 |
2.742536 |
0.20070840 |
179.047691 |
0.00000000000000000000000000000000000000007822597729845195688013386549781920479920693790324668256160374635656452522019742868631882212096738903311238244597802804491948336362838745117187500000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000091656775862309249518454158465732267467628237690686136977297546899184885815570106490804497560961395553891239984523053863085806369781494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
496.480421046548 |
69.2936038218962 |
74.185048299616 |
8.39423073579847 |
| ENSMUSG00000076617 |
16019 |
Ighm |
IG_C_gene |
A0A075B6A0 |
Human_homologues FUNCTION: Constant region of immunoglobulin heavy chains. Immunoglobulins, also known as antibodies, are membrane-bound or secreted glycoproteins produced by B lymphocytes. In the recognition phase of humoral immunity, the membrane-bound immunoglobulins serve as receptors which, upon binding of a specific antigen, trigger the clonal expansion and differentiation of B lymphocytes into immunoglobulins-secreting plasma cells. Secreted immunoglobulins mediate the effector phase of humoral immunity, which results in the elimination of bound antigens (PubMed:22158414, PubMed:20176268). The antigen binding site is formed by the variable domain of one heavy chain, together with that of its associated light chain. Thus, each immunoglobulin has two antigen binding sites with remarkable affinity for a particular antigen. The variable domains are assembled by a process called V-(D)-J rearrangement and can then be subjected to somatic hypermutations which, after exposure to antigen and selection, allow affinity maturation for a particular antigen (PubMed:17576170, PubMed:20176268). IgM antibodies play an important role in primary defense mechanisms. They have been shown to be involved in early recognition of external invaders like bacteria and viruses, cellular waste and modified self, as well as in recognition and elimination of precancerous and cancerous lesions. The membrane-bound form is found in the majority of normal B-cells alongside with IgD. Membrane-bound IgM induces the phosphorylation of CD79A and CD79B by the Src family of protein tyrosine kinases. It may cause death of cells by apoptosis. It is also found in soluble form, which represents about 30% of the total serum immunoglobulins where it is found almost exclusively as a homopentamer. After the antigen binds to the B-cell receptor, the secreted form is secreted in large amounts (PubMed:3137579, PubMed:16895553). {ECO:0000269|PubMed:3137579, ECO:0000303|PubMed:16895553, ECO:0000303|PubMed:17576170, ECO:0000303|PubMed:20176268, ECO:0000303|PubMed:22158414}. |
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
|
cell surface [GO:0009986]; hexameric IgM immunoglobulin complex [GO:0071757]; membrane [GO:0016020]; pentameric IgM immunoglobulin complex [GO:0071756]; immunoglobulin receptor binding [GO:0034987]; peptidoglycan binding [GO:0042834]; phosphatidylcholine binding [GO:0031210]; single-stranded DNA binding [GO:0003697]; adaptive immune response [GO:0002250]; antibacterial humoral response [GO:0019731]; defense response to Gram-negative bacterium [GO:0050829]; innate immune response [GO:0045087] |
11717190_Truncation of the mu heavy chain leads to compromised antigenic stimulation of developing B cell populations with reduced levels of B-2 cells and dramatically increased numbers of the IgM+ CD5+ B-1 subset. 12686505_Somatic translocation & differential expression of Ig mu transgenes indicate a role in memory B cell development. Elements in the Igh locus may promote memory-B-cell selection by regulating Ig expression at some stages of antigen-driven differentiation. 12754269_conventional light chains, a hallmark of B cell differentiation, are necessary and sufficient to divert mu from a vesicular transport-independent to a vesicular transport-dependent degradative route 14500626_In a mouse model it is shown that the transgenic immunoglobulin (Ig) mu heavy chain promotes in vivo differentiation of pro-B cells in the absence of surrogate light chain Ig lambda 5. 14568941_Two specific structural traits common to functionally successful Ig mu heavy chains that have passed the pre-B cell receptor checkpoint are: a complementarity-determining region 3 length of nine amino acids, and the presence of histidine in position 1. 20116104_lambda5 differentially relies on the lambda-like Ig and unique region to probe H chain structure to best accommodate diversity among H chains. 21149550_Data suggest a novel role for the IgM isotype in shaping the earliest steps in recognition and clearance of fungus. 21623346_The critical importance of pre-BCR and BCR receptor levels for the normal development of B-lymphocyte subpopulations in the context of intact VDJ recombination and a diverse antibody repertoire. 21727177_deficiencies in B-cell development increase with increased transgene copy number 22084235_Core fucosylation of mu heavy chains regulates assembly and intracellular signaling of precursor B cell receptors. 22232667_Coronavirus-induced demyelinating CNS disease can be efficiently prevented by a long-lasting germline-encoded IgM response. 23349395_Emu-myc mice provide a model to study spontaneous regression and possible mechanisms of immune evasion or suppression by cancer cells. 24175013_Radiation combined injury-induced alterations of corticosterone, CRP, C3, IgM, and PGE2 cause homeostatic imbalance and may contribute to reduced survival. 24965776_Full-length Emu region does not influence variable heavy (VH) segment usage but ensures efficient Igmu-chain expression required for strong signaling through pre-B cells and newly formed B cell receptors, thus participating in B cell inflow and fate. 27701733_this study shows that in muMT-/- mice, induction of memory 'precursor' effector CD4+ T cells is strongly impaired resulting in a deficiency of the memory TH1 response 27924814_IgM-expressing plasma cells can sense antigen and acquire competence for cytokine production upon antigenic challenge. 30336975_The circulating immunoglobulins negatively impact on the parasite clearance in the liver of Leishmania donovani-infected mice via dampening reactive oxygen species activity. |
ENSG00000211899 |
IGHM |
304.425020 |
0.2702716239 |
-1.887518 |
0.22883863 |
65.574164 |
0.00000000000000055967685824253359767411568678043143719357059009608668098678663227474316954612731933593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000155282293346999476927142866804160312980845424363218754137960786465555429458618164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
121.268164173564 |
19.3161783847475 |
448.689960244471 |
51.5577406168959 |
| ENSMUSG00000078302 |
15229 |
Foxd1 |
protein_coding |
Q61345 |
FUNCTION: Transcription factor involved in regulation of gene expression in a variety of processes including formation of positional identity in the developing retina, regionalization of the optic chiasm, morphogenesis of the kidney, and neuralization of ectodermal cells (PubMed:15509772, PubMed:15634693, PubMed:9811586). Involved in transcriptional activation of PGF and C3 genes (By similarity). {ECO:0000250|UniProtKB:Q16676, ECO:0000269|PubMed:15509772, ECO:0000269|PubMed:15634693, ECO:0000269|PubMed:9811586}. |
Developmental protein;DNA-binding;Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
mmu:15229; |
nucleus [GO:0005634]; DNA binding [GO:0003677]; DNA binding, bending [GO:0008301]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; sequence-specific DNA binding [GO:0043565]; anatomical structure morphogenesis [GO:0009653]; axon guidance [GO:0007411]; branching involved in ureteric bud morphogenesis [GO:0001658]; canonical Wnt signaling pathway [GO:0060070]; cell differentiation [GO:0030154]; dichotomous subdivision of terminal units involved in ureteric bud branching [GO:0060678]; kidney development [GO:0001822]; luteinizing hormone secretion [GO:0032275]; metanephric capsule development [GO:0072213]; metanephric capsule specification [GO:0072267]; metanephric nephron development [GO:0072210]; negative regulation of DNA-templated transcription [GO:0045892]; nephrogenic mesenchyme development [GO:0072076]; pattern specification involved in metanephros development [GO:0072268]; positive regulation of BMP signaling pathway [GO:0030513]; positive regulation of gene expression [GO:0010628]; positive regulation of kidney development [GO:0090184]; positive regulation of transcription by RNA polymerase II [GO:0045944]; regulation of transcription by RNA polymerase II [GO:0006357] |
11943768_Mechanisms of FOXC2- and FOXD1-mediated regulation of the RI alpha subunit of cAMP-dependent protein kinase include release of transcriptional repression and activation by protein kinase B alpha and cAMP. 13678594_inducible expression of BF-2, combined with microarray analysis, to identify Placental Growth Factor (PlGF), a Vascular Endothelial Growth Factor (VEGF) family member previously implicated in angiogenesis, as a downstream target of BF-2 15509772_even though the ipsilateral program is lost in the retina, a larger than normal uncrossed component develops in Foxd1 deficient embryos 15634693_Foxd1-null embryo phenotypes are caused by a failure in the correct formation of the renal capsule 21490208_in the mouse, Foxd1 imprints temporal features in the retina such as axonal ipsilaterality and rostral targeting in collicular areas 23284914_The studies suggest that Foxd1 is indirectly required for normal Lhb expression and cartilage formation. 23924232_In a study to map mice Foxd1-expressing embryonic progenitors cells after bleomycin-induced lung injury, it was found they enter lung buds by 13.5 days post-conception. Collagen-I(alpha)1-expressing mesenchymal cells are also found in adult lung. 24099863_In this review, we focus on the role of four specific FOX factors (FOXD1, FOXL2, FOXO1 and FOXP3) in gonadotropin hormone production 24226518_Glomeruli displayed striking abnormalities, in Rbpj knockout mice including a failure of FOXD1-descendent cells to populate the glomerulus 24284212_the gene encoding the small leucine-rich proteoglycan decorin is repressed by FOXD1 in cortical interstitial cells, compound genetic inactivation of DCN partially rescues the failure of progenitor cell differentiation in the Foxd1 null. 24496101_mediator and indicator of successful progression of cell reprogramming 27088801_FOXD1 lineage renal interstitial cells consist of distinct subpopulations that differ in their responsiveness to Phd2 inactivation and thus regulation of HIF-2 activity and EPO production under hypoxia or conditions of pharmacologic or genetic PHD inactivation. 27805902_Results showed that FOXD1 is a major actor in mammalian reproduction as sequence variants generated recurrent spontaneous abortion (RSA) and embryonic resorption (ER). 28665931_Foxd1 is an upstream positive regulator of renin angiotensin system during early metanephric development and propose that the cross-talk between Foxd1 and RAS is essential in ureter bud-branching morphogenesis. 29284139_The results reveal a novel Shh-Foxd1-Cdkn1c regulatory circuit that drives the mitogenic action of Shh signaling. Forkhead box gene Foxd1 is transcriptionally regulated by canonical Shh signaling. 29667914_Inactivation of MAP3K7 in FOXD1-expressing cells results in loss of mesangial PDGFRBeta and juvenile kidney scarring. 30124871_Here we report that loss of the tumor suppressor gene Tsc2 in pericytes using a FoxD1 promoter driven cre allele (Foxd1tm1(GFP/cre) Amc, FoxD1GC) leads to the formation of Hemangiopericytoma in multiple sites 30158529_FOXD1-dependent MICU1 expression regulates mitochondrial activity and cell differentiation 30810761_It is viable to isolate highly purified metanephric mesenchyme-derived Foxd1(+) mesangial precursor cells using transgenic mice. Foxd1(+) cells could alleviate experimental mesangial proliferative glomerulonephritis and PDGF-induced mesangial cell proliferation through a variety of mechanisms including inhibiting the sonic hedgehog pathway and secreting multiple cytokines. 31102349_LncRNA FOXD1-AS1 acts as a potential oncogenic biomarker in glioma. |
ENSG00000251493 |
FOXD1 |
22.408007 |
0.0294002318 |
-5.088029 |
0.94468793 |
36.272069 |
0.00000000171605683612021558046196412715932166914711842764518223702907562255859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000233801231912192539403877054784292033318138237518724054098129272460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.12746052918211 |
0.792171006241023 |
38.3486951853614 |
12.6343263628178 |
| ENSMUSG00000079008 |
100216455 |
Gm14124 |
protein_coding |
A2AU83 |
|
Metal-binding;Reference proteome;Repeat;Zinc;Zinc-finger |
|
|
mmu:100216455; |
nucleus [GO:0005634]; DNA-binding transcription activator activity, RNA polymerase II-specific [GO:0001228]; metal ion binding [GO:0046872]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
|
|
84.441652 |
4.1842648146 |
2.064974 |
0.31818569 |
41.491801 |
0.00000000011836631170969469986218928648228276498399758054347330471500754356384277343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000018683478341575852410739620387825077330035128397867083549499511718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
140.499219821295 |
31.2388587840689 |
33.5779942345622 |
5.9254110084756 |
| ENSMUSG00000079009 |
100271882 |
Gm14139 |
protein_coding |
A2ANU6 |
|
Metal-binding;Reference proteome;Zinc;Zinc-finger |
|
|
|
nucleus [GO:0005634]; DNA-binding transcription factor activity, RNA polymerase II-specific [GO:0000981]; RNA polymerase II cis-regulatory region sequence-specific DNA binding [GO:0000978]; regulation of transcription by RNA polymerase II [GO:0006357] |
|
|
|
71.040618 |
2.5666524782 |
1.359888 |
0.30398955 |
19.875893 |
0.00000826358033058105228977393424960595780248695518821477890014648437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000622814723381546683778076101134502096101641654968261718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
102.691038910969 |
21.7984491856015 |
40.0097168518327 |
6.64976529173095 |
| ENSMUSG00000079018 |
17067 |
Ly6c1 |
protein_coding |
P0CW02 |
|
Cell membrane;Disulfide bond;Glycoprotein;GPI-anchor;Lipoprotein;Membrane;Reference proteome;Signal |
|
|
mmu:17067; |
cell surface [GO:0009986]; external side of plasma membrane [GO:0009897]; plasma membrane [GO:0005886]; synapse [GO:0045202]; acetylcholine receptor binding [GO:0033130]; acetylcholine receptor inhibitor activity [GO:0030550]; acetylcholine receptor signaling pathway [GO:0095500] |
12184918_Th1 but not Th2 cytokine environments transiently induce the expression of Ly-6C on B cells. There are differences in the regulation of Ly-6C expression in Ly6.1 and Ly6.2 strains. 12538687_Ly6C when cross-linked is able to regulate endothelial adhesion of CD8 T cells via activation of LFA-1 in a manner that involves Ly6C down-regulation on the cell surface. 15034057_During systemic listeria infection, a subset of Ly-6C-expressing monocytes transports L. monocytogenes into the brain and establishes its role of bacterial spreading in vivo. 17084826_plasmacytoid dendritic cells can only be derived from the CD31(high)/Ly6C(-) subset, the subset that reportedly contains the highest frequency of early and late cobblestone area forming cells 17200719_Ly-6C(hi) monocytes represent a newly recognized component of the inflammatory response in experimental atherosclerosis. 18292524_In resting lung, Ly-6C(high)CCR2(high) monocytes repopulate CD103-positive dendritic cells (DCs) using a CCR2-dependent mechanism, and these DCs preferentially retain residual CCR2 in the lung. 18566419_DeltaactA-infected IFN-gamma-/- mice had no brain influx of Ly-6Chigh monocytes despite normal monocyte trafficking from bone marrow to blood and spleen 18779347_Data suggest that CCL2-dependent, Ly6C+ inflammatory monocyte migration is critical for increases in microglia during West Nile Virus infection and may also play a pathogenic role during WNV encephalitis. 19196868_Granulocyte-macrophage colony-stimulating factor-driven release of Ly6C(hi) precursors from the bone marrow prevents exhaustion of central nervous system myeloid populations during relapsing or chronic autoimmune demyelination. 19234172_Ly6C(low-expressing) mature monocytes can promote tolerance to self antigen contained in apoptotic cells through a programmed death ligand (PDL-1)-dependent mechanism. 19830733_Gr-1 Ab induces signals leading to myelopoiesis and affects myeloid-derived suppressor cell activity, suggesting functional roles for Ly-6C/G molecules in macrophage differentiation and neutrophil apoptosis. 19837977_Data show that splenic CD11b(high)Ly6C(+) cells from P chabaudi-infected wild-type mice significantly reduce acute-stage parasitemia in CCR2(-/-) mice. 19864592_Ly6C is a marker of macrophage subsets and support a model of selective recruitment of Ly6C(high) bone marrow monocytes to the kidney that differentiate into three populations of kidney macrophages, including a profibrotic Ly6C(low) population. 20570887_The tumor-monocyte pool almost exclusively consisted of Ly6C(hi)CX(3)CR1(low) monocytes, which continuously seeded tumors and renewed all nonproliferating tumor associated macrophages subsets. 21248254_Ly6C(high-expressing) and Ly6C(low-expressing) bone marrow (BM)-derived monocytes have different fates, demonstrating that BM may be a reservoir of preinflammatory monocytes for rapid deployment as inflammatory dendritic cells during virus infection. 21308682_in vitro and in vivo differentiation of naive CD8(+) T cells into central (Tcm) but not effector (Tem) memory T cells enhances Ly6C expression, and its crosslinking induces strong LFA-1 clustering on Tcm 21343291_TNF-alpha-dependent regulation of acute pancreatitis severity by Ly-6C(hi) monocytes in mice. 22018471_decreased Ly6C and T-bet (Tbx21) expression distinguished a subset of Th1 cells that displayed greater longevity and proliferative responses to secondary infection 22704806_Identify for the involvement of inflammatory Ly-6C(hi) monocytes in the pathogenesis of transplant arteriosclerosis, particularly in conditions of hyperlipidemia. 23100531_Differential Ly-6C expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. 23378430_MPYS-deficient Ly6C(hi) monocytes are intrinsically defective in migration to the liver, but not the spleen. 24625784_Monocytes expressing Ly-6C orchestrate both inflammatory and reparative phases during myocardial infarction. 24995967_in vitro, Ly6C(high) NK cells become Ly6C(low) NK cells with strong effector functions upon stimulation with IL-15. 25473946_Chronic parasitic infection maintains high frequencies of short-lived Ly6C+CD4+ effector T cells that are required for protection against re-infection. 26626303_data demonstrate that CD11c(hi) DCs provide a critical and unexpected role to preserve the immune-privileged CNS in lethal neuroinflammation via regulating the differentiation, function, and trafficking of CD11b(+)Ly-6C(hi) monocytes. 26927544_Data show that Ly6C(high) monocyte subset after middle cerebral artery occlusion/reperfusion (MCAO/R) peaked on day 2 after the operation and was correlated with cerebral infarct size. 27646001_The adapter Mal (encoded by TIRAP) has appeared crucial for the cytokine production by Ly6C(lo) but not by Ly6C(hi) monocytes. The protein Mal was necessary to induce cytokine synthesis by Ly6C(lo) monocytes after triggering TLR2 or TLR9. 28011265_Ly6C, 4-1BB, and KLRG1 have roles in the activation of lamina propria lymphocytes in the small intestine in a mouse model of Crohn's disease 28514690_monocyte differentiation occurred through de novo enhancer establishment and activation of pre-established (poised) enhancers. Generation of Ly6C(-) monocytes involved induction of the transcription factor C/EBPbeta and C/EBPbeta-deficient mice lacked Ly6C(-) monocytes. 28534119_Splenic Ly6C(hi) monocytes contribute to adverse late post-ischemic left ventricular remodeling in heme oxygenase-1 deficient mice. 28663258_surface expression on Ly6C(high) monocytes changes in a time-of-day-dependent manner, which crucially affects cardiac monocyte recruitment after an acute ischemic event. 28691930_CX3CL1 is upregulated in both human and murine tumors following VEGF signaling blockade, resulting in recruitment of CX3CR1+Ly6Clo monocytes into the tumor 29358280_TCR-dependent and cytokine-mediated signals that modulate T cell expression of Ly6C and Sca-1 in vitro and in vivo during infection. 30291130_The Ym1(+)Ly6C(hi) monocytes greatly expanded in bone marrow during the recovery phase of systemic inflammation or tissue injury. 31874104_Splenic Ly6Chi monocytes are critical players in dystrophic muscle injury and repair. 31887775_Ly6CLo Monocyte/Macrophages are Essential for Thrombus Resolution in a Murine Model of Venous Thrombosis. 32346042_Ly6c(Lo) non-classical monocytes promote resolution of rhesus rotavirus-mediated perinatal hepatic inflammation. 34489451_Infection-induced type I interferons critically modulate the homeostasis and function of CD8(+) naive T cells. 35008441_Ly6c as a New Marker of Mouse Blood Vessels: Qualitative and Quantitative Analyses on Intact and Ischemic Retinas. |
ENSG00000291309 |
LY6S |
400.733805 |
2.4117225768 |
1.270064 |
0.15061528 |
70.144714 |
0.00000000000000005510952706701678310113874753416873158902198907220341961288312404576572589576244354248046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000016788566325695992693490381317867509633637659611365977596619813994038850069046020507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
571.290480077131 |
59.750302437067 |
236.88067839132 |
19.0926035440158 |
| ENSMUSG00000079022 |
69700 |
Col22a1 |
protein_coding |
F6TGL6 |
|
Proteomics identification;Reference proteome |
|
|
|
extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; extracellular matrix structural constituent [GO:0005201]; angiogenesis [GO:0001525]; endothelial cell morphogenesis [GO:0001886]; extracellular matrix organization [GO:0030198] |
34831244_Decreased Trabecular Bone Mass in Col22a1-Deficient Mice. |
|
|
995.284158 |
0.3331581832 |
-1.585721 |
0.11355112 |
188.949827 |
0.00000000000000000000000000000000000000000053896970550682181712166748222352886844126372929673011132659467189425956657956978181245165300676595720578862481683901641815737093565985560417175292968750000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000707722978817233679575979002828093508444820694237022391332489356443998192552173839821160026281635889038078637836015616358054103329777717590332031250000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
483.345404440243 |
38.0822662915831 |
1450.79853596397 |
81.5116815611442 |
| ENSMUSG00000079055 |
110893 |
Slc8a3 |
protein_coding |
S4R2P9 |
FUNCTION: Mediates the electrogenic exchange of Ca(2+) against Na(+) ions across the cell membrane, and thereby contributes to the regulation of cytoplasmic Ca(2+) levels and Ca(2+)-dependent cellular processes. Contributes to cellular Ca(2+) homeostasis in excitable cells, both in muscle and in brain (PubMed:14722618, PubMed:21593315). In a first phase, voltage-gated channels mediate the rapid increase of cytoplasmic Ca(2+) levels due to release of Ca(2+) stores from the endoplasmic reticulum. SLC8A3 mediates the export of Ca(2+) from the cell during the next phase, so that cytoplasmic Ca(2+) levels rapidly return to baseline (PubMed:14722618, PubMed:21593315). Contributes to Ca(2+) transport during excitation-contraction coupling in muscle (PubMed:14722618). In neurons, contributes to the rapid decrease of cytoplasmic Ca(2+) levels back to baseline after neuronal activation, and thereby contributes to modulate synaptic plasticity, learning and memory (PubMed:21593315). Required for normal oligodendrocyte differentiation and for normal myelination (PubMed:21959935). Mediates Ca(2+) efflux from mitochondria and contributes to mitochondrial Ca(2+) ion homeostasis (PubMed:24616101). Isoform 1 displays higher calcium exchanger activity than isoform 2, probably because isoform 1 has a lower threshold for activation by cytoplasmic Ca(2+) (PubMed:24616101). {ECO:0000269|PubMed:14722618, ECO:0000269|PubMed:21593315, ECO:0000269|PubMed:21959935, ECO:0000269|PubMed:24616101}. |
3D-structure;Alternative splicing;Antiport;Calcium;Calcium transport;Calmodulin-binding;Cell junction;Cell membrane;Cell projection;Cytoplasm;Endoplasmic reticulum;Glycoprotein;Ion transport;Membrane;Metal-binding;Mitochondrion;Mitochondrion outer membrane;Reference proteome;Repeat;Signal;Sodium;Sodium transport;Synapse;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:110893; |
anchoring junction [GO:0070161]; axon [GO:0030424]; axon terminus [GO:0043679]; cell projection [GO:0042995]; dendrite [GO:0030425]; dendritic spine [GO:0043197]; endoplasmic reticulum membrane [GO:0005789]; membrane [GO:0016020]; microtubule [GO:0005874]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; neuromuscular junction [GO:0031594]; neuronal cell body [GO:0043025]; perikaryon [GO:0043204]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; postsynapse [GO:0098794]; postsynaptic density [GO:0014069]; postsynaptic membrane [GO:0045211]; sarcolemma [GO:0042383]; sarcoplasm [GO:0016528]; synapse [GO:0045202]; calcium:cation antiporter activity [GO:0015368]; calcium:cation antiporter activity involved in regulation of postsynaptic cytosolic calcium ion concentration [GO:1905060]; calcium:sodium antiporter activity [GO:0005432]; calmodulin binding [GO:0005516]; ion antiporter activity involved in regulation of postsynaptic membrane potential [GO:0099580]; metal ion binding [GO:0046872]; calcium ion export across plasma membrane [GO:1990034]; calcium ion import across plasma membrane [GO:0098703]; calcium ion transmembrane transport [GO:0070588]; calcium ion transport into cytosol [GO:0060402]; cell communication [GO:0007154]; cellular calcium ion homeostasis [GO:0006874]; cellular response to cAMP [GO:0071320]; cellular response to hypoxia [GO:0071456]; hematopoietic progenitor cell differentiation [GO:0002244]; learning [GO:0007612]; learning or memory [GO:0007611]; long-term synaptic potentiation [GO:0060291]; memory [GO:0007613]; metal ion transport [GO:0030001]; mitochondrial calcium ion homeostasis [GO:0051560]; mitochondrial calcium ion transmembrane transport [GO:0006851]; modulation of excitatory postsynaptic potential [GO:0098815]; myelination [GO:0042552]; negative regulation of protein serine/threonine kinase activity [GO:0071901]; oligodendrocyte differentiation [GO:0048709]; regulation of skeletal muscle contraction [GO:0014819]; sodium ion transmembrane transport [GO:0035725]; sodium ion transport [GO:0006814]; synapse organization [GO:0050808] |
14722618_NCX3 plays an important role in vivo in the control of Ca(2+) concentrations in the skeletal muscle fibers and at the neuromuscular junction 17317768_three NCX variants are expressed in mouse osteoclasts and play an important role for Ca(2+) transport and regulation during osteoclastic bone resorption 17668452_NCX3 was found to serve as a major contributor of Ca++ translocation out of osteoblasts into calcifying bone matrix. NCX1 had little to no involvement. 19667209_Data suggest that the electrostatic switch in CBD1 and the associated conformational change are necessary for Na+/Ca2+ exchanger activation. 20118617_These results showed significant expression of NCX1 and NCX3 in ameloblasts, indicating their involvement in the directional Ca(2+) extrusion pathway from cells to the enamel mineralizing front 21307349_Results show that glucocorticoid treatment blunts LPS-induced increase in [Ca(2+)](i) in dendritic cells by increasing expression and activity of Na(+)/Ca(2+) exchanger NCX3. 21593315_The deletion of the NCX3 gene has detrimental consequences on basal synaptic transmission, long term potentiation regulation, spatial learning, and memory performance. 21959935_calcium signaling mediated by NCX3 has a crucial role in oligodendrocyte maturation and myelin formation. 22806131_the solution structure of NCX3 calcium binding domain 2B 23224882_the use of genetic-modified mice for NCX1, NCX2, and NCX3 represents a fruitful strategy to characterize the physiological role exerted by NCX in CNS 23224890_Functional studies, as well as mRNA and protein expression analyses, revealed that NCX1 and NCX3, but not NCX2, were divergently modulated during OPC differentiation into oligodendrocyte. 24101730_NCX3 regulates mitochondrial calcium handling from the outer mitochondrial membrane through an AKAP121-anchored signaling complex. 24616101_Function and regulation of the Na+-Ca2+ exchanger NCX3 splice variants in brain and skeletal muscle. 25284465_Demonstrate that NCX3 genetic deletion protects against ziram-induced neurotoxicity and suggest NCX3 and its downstream molecular pathways as key factors involved in ziram toxicity. 27120265_This study demonstrate that knocking-out NCX3 impairs oligodendrocyte response and worsens clinical symptoms in EAE without altering the immune T-cell population. 29274751_NCX2 and NCX3 protein and mRNA levels are relatively reduced in hippocampal CA1 of APP23 and APP-KI mice. Likewise, NCX2(+/-) or NCX3(+/-) mice exhibited impaired hippocampal LTP and memory-related behaviors. Moreover, relative to controls, calcium/calmodulin-dependent protein kinase II (CaMKII) autophosphorylation significantly decreased in NCX2(+/-) mouse hippocampus but increased in hippocampus of NCX3(+/-) mice. 29434186_preconditioning prevented NCX3 downregulation in SOD1 G93A mice spinal cord, leading to an increased number of motor neurons associated to a reduced astrogliosis, and reduced the denervation of neuromuscular junctions observed in SOD1 G93A mice. 29941946_NCX1 was exclusively co-expressed in IBA-1-positive microglial cells in the striatum, whereas NCX3 was solely co-expressed in tyrosine hydroxylase-positive neurons in substantia nigra. 32326436_TGF-beta Signaling Regulates SLC8A3 Expression and Prevents Oxidative Stress in Developing Midbrain Dopaminergic and Dorsal Raphe Serotonergic Neurons. 34360942_Ncx3-Induced Mitochondrial Dysfunction in Midbrain Leads to Neuroinflammation in Striatum of A53t-alpha-Synuclein Transgenic Old Mice. 35705078_Sodium-calcium exchanger-3 regulates pain ''wind-up'': From human psychophysics to spinal mechanisms. |
ENSG00000100678 |
SLC8A3 |
1191.431010 |
0.3225479771 |
-1.632414 |
0.11554701 |
193.455397 |
0.00000000000000000000000000000000000000000005599217553037348777287064027206529565303396999097749068501054787356289819468521968832296424908577400457663884739481496843893637560540810227394104003906250000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000000000075448165336493015402382346519571248265974945529541210298629816368624805799303320179504875854987894003440265441873258112082112347707152366638183593750000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
565.279011362385 |
45.3222281179859 |
1752.54241666755 |
102.266310424291 |
| ENSMUSG00000079190 |
100041057 |
|
protein_coding |
|
|
|
|
|
|
|
|
|
|
58.703396 |
4.8082243160 |
2.265504 |
0.37361304 |
36.442281 |
0.00000000157254691009330039005694164229226181261900308072654297575354576110839843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000215404986821413242330925898919760652638899500743718817830085754394531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
94.1107795855827 |
24.6450879089238 |
19.5728762638929 |
4.16362255861142 |
| ENSMUSG00000079192 |
|
|
protein_coding |
|
|
|
|
|
|
|
|
|
|
29.560394 |
5.5846121198 |
2.481457 |
0.54653321 |
20.382916 |
0.00000633932048751275214559883913745252925764361862093210220336914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000487679442756536570468747782136631485627731308341026306152343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
49.1671896878905 |
19.0746162000839 |
8.80404737757982 |
2.77042308117921 |
| ENSMUSG00000079355 |
252837 |
Ackr4 |
protein_coding |
Q924I3 |
FUNCTION: Atypical chemokine receptor that controls chemokine levels and localization via high-affinity chemokine binding that is uncoupled from classic ligand-driven signal transduction cascades, resulting instead in chemokine sequestration, degradation, or transcytosis. Also known as interceptor (internalizing receptor) or chemokine-scavenging receptor or chemokine decoy receptor. Acts as a receptor for chemokines CCL2, CCL8, CCL13, CCL19, CCL21 and CCL25. Chemokine-binding does not activate G-protein-mediated signal transduction but instead induces beta-arrestin recruitment, leading to ligand internalization. Plays an important role in controlling the migration of immune and cancer cells that express chemokine receptors CCR7 and CCR9, by reducing the availability of CCL19, CCL21, and CCL25 through internalization. Negatively regulates CXCR3-induced chemotaxis. Regulates T-cell development in the thymus and inhibits spontaneous autoimmunity. {ECO:0000269|PubMed:11981810, ECO:0000269|PubMed:23152546}. |
Cell membrane;Endosome;G-protein coupled receptor;Glycoprotein;Membrane;Phosphoprotein;Receptor;Reference proteome;Transducer;Transmembrane;Transmembrane helix |
|
|
mmu:252837; |
early endosome [GO:0005769]; external side of plasma membrane [GO:0009897]; recycling endosome [GO:0055037]; C-C chemokine binding [GO:0019957]; C-C chemokine receptor activity [GO:0016493]; chemokine receptor activity [GO:0004950]; scavenger receptor activity [GO:0005044]; calcium-mediated signaling [GO:0019722]; cell chemotaxis [GO:0060326]; immune response [GO:0006955]; positive regulation of cytosolic calcium ion concentration [GO:0007204] |
11981810_Characterization of mouse CCX-CKR, a receptor for the lymphocyte-attracting chemokines TECK/mCCL25, SLC/mCCL21 and MIP-3beta/mCCL19: comparison to human CCX-CKR. (CCX-CKR) 17485674_These observations indicate that the silent chemokine receptor CCX-CKR1, regulates homeostatic leukocyte migration by controlling the availability of chemokines in the extracellular space. 20562329_CCX-CKR(-/-) have a 5-fold increase in the level of CCL21 protein in blood, and 2- to 3-fold increases in CCL19 and CCL21 in peripheral lymph nodes. 23152546_CCX-CKR deletion increases incidence of a spontaneous Sjogren's syndrome-like pathology, suggestive of a defect in self-tolerance. CCX-CKR(-/-) mice have fewer thymic epithelial cells per thymocyte, with defects in thymocyte distribution & frequency. 24813163_found that lymph node fringes indeed contained physiological gradients of the chemokine CCL21, which depended on the expression of CCRL1, the atypical receptor for the CCR7 ligands CCL19 and CCL21 25027038_The data shows a novel function for the chemokine receptor CCX-CKR as a regulator of TGF-beta1 expression and epithelial-mesenchymal transition in breast cancer cells. 25070355_stepwise acquisition of chemokine (C-C motif) receptor-like 1 (CCRL1) is a late determinant of cortical thymic epithelial cell differentiation 25521433_CCRL1 is expressed in key thymic microenvironments but is dispensable for T lymphopoiesis at steady state in adult mice. 26976955_ACKR4 on stromal cells aids the egress of antigen presenting cells from mouse skin, and, during inflammation, facilitates CCR7-dependent cell trafficking by scavenging CCL19. 28807994_a comprehensive model of CCL19 and CCL21 transport and gradient formation in the lymph nodes (LNs) was built; predicts that ACKR4 in LNs prevents CCL19/CCL21 accumulation in efferent lymph, but does not control intranodal gradients; instead, it attributes the disrupted interfollicular CCL21 gradients observed in Ackr4-deficient LNs to ACKR4 loss upstream 29386231_atypical chemokine receptor 4 (ACKR4), is a identified a decoy receptor that binds and degrades CCR7 ligands CCL19/CCL21, as a regulator of early activated B cell differentiation. 31841228_B cell hyperactivation in an Ackr4-deficient mouse strain is not caused by lack of ACKR4 expression. 32289156_ACKR4 restrains antitumor immunity by regulating CCL21. 32533638_CCL20 is a novel ligand for the scavenging atypical chemokine receptor 4. 33610913_Inhibition of fibroblast IL-6 production by ACKR4 deletion alleviates cardiac remodeling after myocardial infarction. 33875601_Scavenging of soluble and immobilized CCL21 by ACKR4 regulates peripheral dendritic cell emigration. 34260918_Expression of ACKR4 demarcates the ''peri-marginal sinus,'' a specialized vascular compartment of the splenic red pulp. 35108538_Mechanosensitive ACKR4 scavenges CCR7 chemokines to facilitate T cell de-adhesion and passive transport by flow in inflamed afferent lymphatics. |
ENSG00000129048 |
ACKR4 |
1393.188746 |
0.3812042313 |
-1.391364 |
0.07818202 |
312.547481 |
0.00000000000000000000000000000000000000000000000000000000000000000000060852758337228531375304929739394655430320949760780339574941695565001003527670208351763910120465800655685827250852361544067176114357812199532707175115862136002122458562960637118433027126229717396 |
0.0000000000000000000000000000000000000000000000000000000000000000001782517721139740333531654622101474718211541830090002755497494705567888920648015167081908508297472524415759623764479087746213646565313608775089986022811038271629601468029613364763008576119318604 |
Yes |
No |
769.854575295505 |
41.7401106370165 |
2019.53313226586 |
78.4698417396748 |
| ENSMUSG00000079429 |
100040766 |
Mroh2a |
protein_coding |
D3Z750 |
|
Reference proteome;Repeat |
|
|
|
cytoplasm [GO:0005737] |
Human_homologues 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) |
ENSG00000185038 |
MROH2A |
159.764007 |
2.3179707698 |
1.212862 |
0.24010710 |
25.218941 |
0.00000051177117939135163956018313366480576576122984988614916801452636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000047882669560743656319537660259744171753482078202068805694580078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
212.808313536182 |
35.5813251364237 |
91.8080229075168 |
11.6799249224589 |
| ENSMUSG00000079434 |
23956 |
Neu2 |
protein_coding |
Q9JMH3 |
FUNCTION: Exo-alpha-sialidase that catalyzes the hydrolytic cleavage of the terminal sialic acid (N-acetylneuraminic acid, Neu5Ac) of a glycan moiety in the catabolism of glycolipids, glycoproteins and oligosacharides (PubMed:10713120). Recognizes sialyl linkage positions of the glycan moiety as well as the supramolecular organization of the sialoglycoconjugate (By similarity). Displays preference for alpha-(2->3)-sialylated GD1a and GT1B gangliosides over alpha-(2->8)-sialylated GD1b, in both monomeric forms and micelles (PubMed:10713120). Hydrolyzes exclusively monomeric GM1 ganglioside, but has no activity toward the miscellar form (By similarity). Has lower sialidase activity for glycoproteins such as fetuin and TF/transferrin that carry a mixture of alpha-(2->3) and alpha-(2->6)-sialyl linkages (By similarity). Cleaves milk oligosaccharide alpha-(2->3)-sialyllactose, but is inactive toward isomer alpha-(2->6)-sialyllactose isomer. Has no activity toward colominic acid, a homomer of alpha-(2->8)-linked Neu5Ac residues (By similarity). {ECO:0000250|UniProtKB:Q9Y3R4, ECO:0000269|PubMed:10713120}. |
Carbohydrate metabolism;Cytoplasm;Glycosidase;Hydrolase;Lipid degradation;Lipid metabolism;Reference proteome;Repeat |
|
|
mmu:23956; |
catalytic complex [GO:1902494]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; intracellular membrane-bounded organelle [GO:0043231]; membrane [GO:0016020]; exo-alpha-(2->3)-sialidase activity [GO:0052794]; exo-alpha-(2->6)-sialidase activity [GO:0052795]; exo-alpha-(2->8)-sialidase activity [GO:0052796]; exo-alpha-sialidase activity [GO:0004308]; cellular oligosaccharide catabolic process [GO:0051692]; ganglioside catabolic process [GO:0006689]; glycoprotein catabolic process [GO:0006516]; oligosaccharide catabolic process [GO:0009313]; positive regulation of myoblast differentiation [GO:0045663]; positive regulation of myotube differentiation [GO:0010831] |
16869890_Our data demonstrate that IGF-1-induced myoblast differentiation and hypertrophy are driven, at least in part, by Neu2 upregulation and further support the significant role of cytosolic sialidase in myoblasts. 18553168_the lack of soluble sialidase activity in the SM/J mouse thymus is due to the thymus-specific low expression level of the Neu2 gene 19632200_30-40% of B and C forms of Neu2 activity was located in the crude membrane-fraction, and hydrolyzed ganglioside effectively, while both soluble fraction showed particular behavior with substrate specificity. 23750721_Neu2 from thymus exhibited sialidase activity with fetuin at pH 7.0. 35217678_Defect in cytosolic Neu2 sialidase abrogates lipid metabolism and impairs muscle function in vivo. |
ENSG00000115488 |
NEU2 |
40.669804 |
0.2570460582 |
-1.959901 |
0.43879943 |
19.818399 |
0.00000851589812336062696048431808204526305416948162019252777099609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000639300937481661237914848383567800738092046231031417846679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.8236276231489 |
5.19620947308207 |
65.4498565033667 |
13.7711695608944 |
| ENSMUSG00000079455 |
|
Gm16026 |
transcribed_unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
15.410461 |
10.3917614804 |
3.377368 |
0.88185884 |
13.875934 |
0.00019528307925719076980526633935397740060579963028430938720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010883834113595059790713381886462229886092245578765869140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.2631661559699 |
17.7221524009047 |
2.62353655896738 |
1.38917173993962 |
| ENSMUSG00000079481 |
100042480 |
Nhsl2 |
protein_coding |
B1AXH1 |
|
Phosphoprotein;Reference proteome |
|
|
mmu:100042480; |
cell differentiation [GO:0030154] |
|
ENSG00000204131 |
NHSL2 |
104.155089 |
2.4669601421 |
1.302734 |
0.31922216 |
16.433634 |
0.00005038326484009686965278435044623961402976419776678085327148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003227240916923278116548723382095431588822975754737854003906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
154.259737468218 |
34.4119369105461 |
62.5302917683057 |
10.7101606928036 |
| ENSMUSG00000079553 |
100502766 |
Kifc1 |
protein_coding |
Q9QWT9 |
FUNCTION: Minus end-directed microtubule-dependent motor required for bipolar spindle formation (PubMed:16638812). May contribute to movement of early endocytic vesicles (PubMed:17360972). Regulates cilium formation and structure (PubMed:23807208). {ECO:0000269|PubMed:16638812, ECO:0000269|PubMed:17360972, ECO:0000269|PubMed:23807208}. |
Alternative splicing;ATP-binding;Cell cycle;Cell division;Coiled coil;Cytoplasm;Cytoskeleton;Direct protein sequencing;Endosome;Microtubule;Mitosis;Motor protein;Nucleotide-binding;Nucleus;Phosphoprotein;Reference proteome |
|
|
mmu:100502766;mmu:16580; |
early endosome [GO:0005769]; endocytic vesicle [GO:0030139]; kinesin complex [GO:0005871]; microtubule [GO:0005874]; microtubule organizing center [GO:0005815]; mitotic spindle [GO:0072686]; nucleus [GO:0005634]; spindle pole centrosome [GO:0031616]; ATP binding [GO:0005524]; ATP hydrolysis activity [GO:0016887]; microtubule binding [GO:0008017]; microtubule motor activity [GO:0003777]; minus-end-directed microtubule motor activity [GO:0008569]; cell division [GO:0051301]; microtubule-based movement [GO:0007018]; minus-end-directed vesicle transport along microtubule [GO:0072382]; mitotic metaphase plate congression [GO:0007080]; mitotic spindle assembly [GO:0090307]; negative regulation of centrosome duplication [GO:0010826]; vesicle transport along microtubule [GO:0047496] |
23807208_Knockdown of KIFC5A dramatically reduces ciliogenesis. 31904480_TRIM8 interacts with KIF11 and KIFC1 and controls bipolar spindle formation and chromosomal stability. 35142352_Kinesin motor KIFC1 is required for tubulin acetylation and actin-dependent spindle migration in mouse oocyte meiosis. 35259401_Kinesin-14 KIFC1 modulates spindle assembly and chromosome segregation in mouse spermatocytes. |
ENSG00000237649 |
KIFC1 |
1052.468840 |
2.1471433907 |
1.102419 |
0.11414455 |
92.111400 |
0.00000000000000000000081932014242382407483204760773644436330186930174739267745921342256387731595168588683009147644042968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000359650847533132268304308066801084324155562324174826774455734912372406597569352015852928161621093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1447.25210152874 |
114.624597392816 |
674.036074075706 |
40.7202426110327 |
| ENSMUSG00000079652 |
245884 |
Garin1a |
protein_coding |
B2RXB0 |
FUNCTION: RAB2B effector protein required for accurate acrosome formation and normal male fertility. {ECO:0000269|PubMed:34714330}. |
Golgi apparatus;Reference proteome |
|
|
mmu:245884; |
Golgi apparatus [GO:0005794]; acrosome assembly [GO:0001675] |
Human_homologues 17672918_validated occurrence of an unusual TG 3' splice site in intron 1 |
ENSG00000205085 |
GARIN1A |
110.930583 |
2.2984218371 |
1.200644 |
0.25242974 |
22.548585 |
0.00000204895387819867324450924875312018968998017953708767890930175781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000172048872506737557668173022040747355276835151016712188720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
155.041183183631 |
27.2663358790447 |
67.4554951932338 |
9.16883419642017 |
| ENSMUSG00000080316 |
75202 |
Spaca6 |
protein_coding |
E9Q8Q8 |
FUNCTION: Sperm protein required for fusion of sperm with the egg membrane during fertilization. {ECO:0000269|PubMed:24275887, ECO:0000269|PubMed:32210282, ECO:0000269|PubMed:32393636}. |
Alternative splicing;Cytoplasmic vesicle;Disulfide bond;Fertilization;Glycoprotein;Immunoglobulin domain;Membrane;Reference proteome;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:75202; |
acrosomal membrane [GO:0002080]; acrosomal vesicle [GO:0001669]; membrane [GO:0016020]; fusion of sperm to egg plasma membrane involved in single fertilization [GO:0007342] |
24275887_The Spaca6 gene product may, together with Izumo1, mediate sperm fusion by binding an as yet unidentified egg membrane receptor. 25059272_Dual-luciferase reporter gene analysis showed that RTEF-1 is a direct target of mir-125a-5p, which regulates angiogenesis by repressing RTEF-1 expression and modulating eNOS and VEGF expression. 28533542_Our findings suggest a mechanism, by which a decrease in miR-125a-3p during oocyte maturation facilitates GVBD by allowing Fyn up-regulation and the resulting stabilization of the interaction between actin and A-type lamins. 32210282_Sperm SPACA6 protein is required for mammalian Sperm-Egg Adhesion/Fusion. 32393636_Sperm proteins SOF1, TMEM95, and SPACA6 are required for sperm-oocyte fusion in mice. 33599498_MiR-99b-5p Attenuates Adipogenesis by Targeting SCD1 and Lpin1 in 3T3-L1 Cells. 34775219_Megakaryocyte-specific knockout of the Mir-99b/let7e/125a cluster lowers platelet count without altering platelet function. |
ENSG00000182310 |
SPACA6 |
1373.741110 |
0.3514887052 |
-1.508450 |
0.11506905 |
166.907977 |
0.00000000000000000000000000000000000003503729578639289739894220595740122918260308672873875245899660278607994483732901513959982017148780345655589840703214576933532953262329101562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000037061672876273373548815113018586182959553490338246855733449724465152695885242537052868179740707649250097688309324439615011215209960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
687.939659089957 |
54.9281169690942 |
1957.21697152823 |
117.203824913697 |
| ENSMUSG00000081434 |
628060 |
Gm14165 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
31.537537 |
0.3039659584 |
-1.718018 |
0.51946286 |
10.434692 |
0.00123670187685164228302836519901575229596346616744995117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0055798113116718650247860544766353996237739920616149902343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.0978054560211 |
5.5544067021927 |
49.6693956705172 |
12.7451567623566 |
| ENSMUSG00000081974 |
100041006 |
Gm11960 |
transcribed_processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
38.178755 |
2.5521606074 |
1.351719 |
0.50166831 |
7.156134 |
0.00747080276613095069226933020445358124561607837677001953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0261598316629210667083604846538946731016039848327636718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
52.8543477400698 |
18.7516739349916 |
20.7096479689113 |
5.59256646621897 |
| ENSMUSG00000082450 |
637027 |
Mageb7-ps |
unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
16.768933 |
31.5272943760 |
4.978529 |
1.07683173 |
25.732438 |
0.00000039217949116020787956346342617575118083550478331744670867919921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000037219177628363155978751235924972817770139954518526792526245117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.3017565011768 |
26.4120127562053 |
1.02456481409167 |
0.750795232922548 |
| ENSMUSG00000082978 |
433712 |
Rpsa-ps11 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
15.488213 |
3.5146856497 |
1.813396 |
0.67315132 |
7.234740 |
0.00715061707048034872480357648782955948263406753540039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0252537188651617526546910852403016178868710994720458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.7784459728111 |
10.5344181188627 |
6.19641360375595 |
2.40183612138919 |
| ENSMUSG00000083282 |
56464 |
Ctsf |
protein_coding |
Q9R013 |
FUNCTION: Thiol protease which is believed to participate in intracellular degradation and turnover of proteins. Has also been implicated in tumor invasion and metastasis. |
Disulfide bond;Glycoprotein;Hydrolase;Lysosome;Protease;Reference proteome;Signal;Thiol protease;Zymogen |
|
|
mmu:56464; |
endoplasmic reticulum [GO:0005783]; extracellular space [GO:0005615]; intracellular membrane-bounded organelle [GO:0043231]; lysosome [GO:0005764]; plasma membrane [GO:0005886]; cysteine-type endopeptidase activity [GO:0004197]; cysteine-type peptidase activity [GO:0008234]; proteolysis involved in protein catabolic process [GO:0051603] |
16508006_cathepsin F inactivation causes a lysosomal storage defect and progressive neurological features in mice 21309947_This gene is known to be important for immune responses and may potentially regulate alcohol consumption. |
ENSG00000174080 |
CTSF |
21.283562 |
0.0158148393 |
-5.982577 |
1.22094731 |
33.833252 |
0.00000000600437744254111049626493050337776302693981733682448975741863250732421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000751509262159294955396254367223363157535231948713771998882293701171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.700854406118986 |
0.666511253098546 |
44.316252083918 |
16.3771697112084 |
| ENSMUSG00000083307 |
245350 |
AA414768 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
54.289389 |
2.4864789363 |
1.314104 |
0.36335569 |
13.025791 |
0.00030723020189611215807756749995860445778816938400268554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016271663544094505573361919559260968526359647512435913085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
78.8058207261713 |
20.0584550052631 |
31.6937415302967 |
6.26210539089011 |
| ENSMUSG00000083833 |
|
Gm13841 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
13.716799 |
0.0323525734 |
-4.949976 |
1.21365145 |
18.933802 |
0.00001353333990992943434202716579406455821299459785223007202148437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000978357064584931869228451306597094117023516446352005004882812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.722649770831169 |
0.682204142352838 |
22.3367014114533 |
10.3601675008259 |
| ENSMUSG00000084349 |
100043000 |
Rpl3-ps1 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
40.443942 |
0.0022680592 |
-8.784326 |
1.26557875 |
89.262689 |
0.00000000000000000000345718269539827697268257197209372962738374145318550897476670563657208390395680908113718032836914062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001438792535964659833022713920267742296862457586304202936285101532831731674377806484699249267578125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.200329642901385 |
0.199158106172698 |
88.3264594512498 |
22.637742627549 |
| ENSMUSG00000084762 |
102642414 |
Platr3 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
435.832086 |
10.4236547847 |
3.381789 |
0.18648618 |
315.124826 |
0.00000000000000000000000000000000000000000000000000000000000000000000016704933793765686172814955222396274071972348520920907493721053789616785758023547340126222260633456604212511253326175773383252435430843898569843536783906556696106981840194778861363733568623501924 |
0.0000000000000000000000000000000000000000000000000000000000000000000508899103093277821564564865835964121314715889812352542044340799772363491491342293004342825615301746618140576186320802961787069775130675432431435957852095349631014954867869182209005884942598641 |
Yes |
Yes |
807.428015692402 |
104.660951770574 |
77.461123988901 |
8.27190690802723 |
| ENSMUSG00000085101 |
|
Platr16 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
694.796171 |
5.9808990441 |
2.580362 |
0.19381518 |
164.023062 |
0.00000000000000000000000000000000000014952843063325038447215287763217249756380128597342236367996607767773901578776407282491712702829226092071657205906376475468277931213378906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000153893044284166870081391692967306972397018776270127267767233265415476886826311512332596535997109032933849448454566299915313720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
1214.63809810497 |
163.668729322328 |
203.086206461964 |
21.2692735100478 |
| ENSMUSG00000085146 |
|
Eif2c5 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
8.966637 |
97.0680992741 |
6.600925 |
1.53045484 |
19.025429 |
0.00001289880017678331559948575607554843713842274155467748641967773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000937825891612236096266455587233679125347407534718513488769531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
17.9853709539338 |
22.8648091008742 |
0.185286114474598 |
0.208115413179831 |
| ENSMUSG00000085402 |
|
Gm12111 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
10.579387 |
11.5883730599 |
3.534606 |
1.02737040 |
10.666915 |
0.00109068864536913403238960196262041790760122239589691162109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0049919980307279593345626267364423256367444992065429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.384243253223 |
15.0005168179895 |
1.67273206971903 |
1.04268403893217 |
| ENSMUSG00000085416 |
72249 |
1700019G24Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
14.059578 |
6.0040499460 |
2.585936 |
0.96243077 |
6.672306 |
0.00979223948367464493147682702556267031468451023101806640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0328970868582558889636757726293581072241067886352539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.8674507537506 |
16.4119552781027 |
3.80867097367426 |
2.04467773904765 |
| ENSMUSG00000085492 |
68789 |
Trmt61b |
lncRNA |
|
|
|
|
|
|
|
|
|
|
146.150290 |
0.4840257026 |
-1.046844 |
0.22930809 |
20.650087 |
0.00000551346782441061529955912690059705028033931739628314971923828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000428348611792473085734846149907895096475840546190738677978515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
92.6607795134668 |
14.7899897808224 |
191.437725337083 |
22.0592410700998 |
| ENSMUSG00000085643 |
102637828 |
Gm12519 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
179.348035 |
0.2842564127 |
-1.814735 |
0.29268156 |
37.359185 |
0.00000000098257050794459250407603955758915892371163636198616586625576019287109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000138707265773976201025772611737617301397307301158434711396694183349609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
74.5548759365697 |
15.2290153106103 |
262.280365918235 |
38.3349947375147 |
| ENSMUSG00000085649 |
102639814 |
A730032A03Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
14.694278 |
4.1786012974 |
2.063020 |
0.71564238 |
8.253150 |
0.00406813445432854109418130406083946581929922103881835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0155575756988029969413167563629940559621900320053100585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
23.3829974234114 |
12.0805958079971 |
5.59589100733274 |
2.29873052490912 |
| ENSMUSG00000085867 |
|
Gm5834 |
unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
21.549396 |
0.2268868065 |
-2.139955 |
0.58051880 |
13.533925 |
0.00023428954575523507747845941562303551108925603330135345458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0012749904826522833085900021643510626745410263538360595703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.9914789037129 |
3.30313251472301 |
35.2223164850023 |
9.7061350937213 |
| ENSMUSG00000085921 |
|
Gm13752 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
27.047141 |
0.3179615340 |
-1.653076 |
0.52356321 |
9.903270 |
0.00164985322714917875992768347970240938593633472919464111328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0072069298411059053502403237700946192489936947822570800781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.5401310045256 |
4.6514461067946 |
39.4391448702316 |
10.306746846724 |
| ENSMUSG00000086389 |
|
Gm15998 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
33.236717 |
0.2012696297 |
-2.312799 |
0.51335911 |
19.859522 |
0.00000833465475146620511431513889100131109444191679358482360839843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000627306337149345362132549230871347845095442607998847961425781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.8421363060203 |
3.93992631049185 |
53.8687149326395 |
13.2855575190789 |
| ENSMUSG00000086411 |
|
Gm14236 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
8.356415 |
0.1550935015 |
-2.688790 |
1.06096844 |
6.156862 |
0.01309032697751429281940449556032035616226494312286376953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0422619458502538625221944812437868677079677581787109375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.63459229280695 |
2.11692303275172 |
16.987122398744 |
8.5013679836465 |
| ENSMUSG00000086421 |
|
Gm14091 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
41.411479 |
3.5830522068 |
1.841189 |
0.40690403 |
20.375232 |
0.00000636482845539781158343550782063857695902697741985321044921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000489394583708326435362266637607575603396981023252010345458984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.965030732171 |
18.5669198121781 |
18.1311984819079 |
4.13224958287771 |
| ENSMUSG00000086607 |
75084 |
4930511M06Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
70.589665 |
2.2417825550 |
1.164646 |
0.31179817 |
13.911031 |
0.00019167028616639704267400612369698365000658668577671051025390625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010713841463803890248496220038987303269095718860626220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
91.3370133818705 |
19.8939706773222 |
40.7430297721577 |
6.88831615914858 |
| ENSMUSG00000086748 |
|
Gm13261 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
8.358970 |
0.0422064099 |
-4.566394 |
1.34207386 |
13.798029 |
0.00020354965282181147301181212139198350996593944728374481201171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011262144249116717679981558575263989041559398174285888671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.635995716473584 |
0.680738524478251 |
15.0686996966415 |
7.52573538737825 |
| ENSMUSG00000086854 |
|
Gm13781 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
8.816683 |
8.3706969783 |
3.065348 |
1.19407018 |
6.444327 |
0.01113071861621236190165173951527322060428559780120849609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0366738278092465300184166210328839952126145362854003906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
15.1570226252392 |
14.0271059079353 |
1.8107240848084 |
1.35234477407097 |
| ENSMUSG00000087013 |
69909 |
2610027K06Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
35.789911 |
0.4100184288 |
-1.286239 |
0.50995425 |
6.140217 |
0.01321411144857232919380685132182406960055232048034667968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0425516571001228957604745062326401239261031150817871093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.8636013821959 |
7.52925987985228 |
50.8845454675911 |
13.1574334431232 |
| ENSMUSG00000087203 |
329496 |
Gm13986 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
23.134547 |
0.3204444315 |
-1.641854 |
0.59276021 |
7.290231 |
0.00693305471631984124303293981483875541016459465026855468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0245591370788334449615231136476722895167768001556396484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
12.4316201471803 |
5.25270951425776 |
38.7949326746382 |
11.5114974741027 |
| ENSMUSG00000087362 |
672763 |
Gm13710 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
71.499885 |
0.3561926196 |
-1.489270 |
0.33891744 |
19.022646 |
0.00001291762256837140649540218717561401717830449342727661132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000938746311838899063877128536859117957646958529949188232421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
36.1570369860896 |
8.57234181709008 |
101.509787103602 |
17.1329382606716 |
| ENSMUSG00000087367 |
|
Gm15491 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
31.208865 |
9.0043227845 |
3.170618 |
0.94202169 |
7.720493 |
0.00545974971012060453223746492312784539535641670227050781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0200344272668169232076706975931301712989807128906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
46.6851858544344 |
32.6963349915505 |
5.1847525873642 |
2.86583883633524 |
| ENSMUSG00000087400 |
|
Gm15270 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
68.691477 |
0.2187719831 |
-2.192500 |
0.41178019 |
27.405927 |
0.00000016492494982633503851056572600541905160298483679071068763732910156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000016734724483364979751378893296775984822488680947571992874145507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.1985166051576 |
7.57963794107535 |
119.752612900426 |
24.5970332790379 |
| ENSMUSG00000087674 |
|
4930447M23Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
16.481111 |
4.5240838804 |
2.177626 |
0.66181701 |
10.791303 |
0.00101978069815888234758705088722763321129605174064636230468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0047029473903107904889098200840180652448907494544982910156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.8849925302659 |
13.2452627527258 |
6.16367717030461 |
2.337083039145 |
| ENSMUSG00000089774 |
53881 |
Slc5a3 |
protein_coding |
Q9JKZ2 |
FUNCTION: Electrogenic Na(+)-coupled sugar symporter that actively transports myo-inositol and its stereoisomer scyllo-inositol across the plasma membrane, with a Na(+) to sugar coupling ratio of 2:1 (By similarity). Maintains myo-inositol concentration gradient that defines cell volume and fluid balance during osmotic stress, in particular in the fetoplacental unit and central nervous system (PubMed:12582158, PubMed:24595108). Forms coregulatory complexes with voltage-gated K(+) ion channels, allosterically altering ion selectivity, voltage dependence and gating kinetics of the channel. In turn, K(+) efflux through the channel forms a local electrical gradient that modulates electrogenic Na(+)-coupled myo-inositol influx through the transporter (PubMed:24595108) (By similarity). Associates with KCNQ1-KCNE2 channel in the apical membrane of choroid plexus epithelium and regulates the myo-inositol gradient between blood and cerebrospinal fluid with an impact on neuron excitability (PubMed:24595108). Associates with KCNQ2-KCNQ3 channel altering ion selectivity, increasing Na(+) and Cs(+) permeation relative to K(+) permeation (By similarity). Provides myo-inositol precursor for biosynthesis of phosphoinositides such as PI(4,5)P2, thus indirectly affecting the activity of phosphoinositide-dependent ion channels and Ca(2+) signaling upon osmotic stress (By similarity). {ECO:0000250|UniProtKB:P31637, ECO:0000250|UniProtKB:P53794, ECO:0000269|PubMed:12582158, ECO:0000269|PubMed:24595108}.; FUNCTION: (Microbial infection) Functions as a retroviral receptor for M813 murine leukemia virus (MuLV) entry. {ECO:0000269|PubMed:12719585}. |
Cell membrane;Glycoprotein;Ion transport;Membrane;Phosphoprotein;Reference proteome;Sodium;Sodium transport;Symport;Transmembrane;Transmembrane helix;Transport |
|
|
mmu:53881; |
apical plasma membrane [GO:0016324]; basolateral plasma membrane [GO:0016323]; membrane [GO:0016020]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; voltage-gated potassium channel complex [GO:0008076]; glucose:sodium symporter activity [GO:0005412]; myo-inositol:sodium symporter activity [GO:0005367]; potassium channel regulator activity [GO:0015459]; transmembrane transporter binding [GO:0044325]; inositol metabolic process [GO:0006020]; myo-inositol transport [GO:0015798]; peripheral nervous system development [GO:0007422]; positive regulation of catalytic activity [GO:0043085]; positive regulation of protein localization to membrane [GO:1905477]; positive regulation of reactive oxygen species biosynthetic process [GO:1903428]; regulation of respiratory gaseous exchange [GO:0043576]; regulation of transporter activity [GO:0032409] |
12582158_SLC5a3 is responsible for the Ins concentration gradient in the murine fetal-placental unit 12719585_used by Mus cervicolor Murine Leukemia Virus for cell entry 14613899_When Sertoli cells were cultured in a hypertonic medium, there was an increase in the expression of Isyna1 and Slc5a3 16174787_SMIT-1 has a role in the development and function of the peripheral nerves 18675571_Mrps6 gene expression may not be contingent on function of Slc5a3 gene, while inositide deficiency may not be the cause of lethal apnea in null Slc5a3 mice. 20818642_Sodium/MI cotransporter 1 (SMIT1, the major cotransporter of MI) knockout (SMIT1(-/-)) mice with markedly reduced tissue myoinositol (MI) levels were used to characterize the essential roles of MI and SMIT1 in osteogenesis. 21887366_brain SMIT1 levels were higher than SMIT2, however, regional differences were observed 22457525_These findings add Mus spicilegus endogenous retrovirus HEMV as a second member to the murine leukemia virus subgroup that uses mSMIT1 to gain entry into cells. 22564531_At low concentration, where SMIT1/2 activity gains importance, myo-inositol uptake is reduced by ammonia-induced intracellular acidification 23172129_activation of NFAT5 and its downstream SMIT induction is important in protecting neurons from ischemia-induced oxidative stress 24595108_KCNQ1, KCNE2, and SMIT1 form reciprocally regulating complexes that affect neuronal excitability. 27922641_Lithium affects the phosphatidylinositol signaling system in two ways: depleting inositol, consequently decreasing phosphoinositides; elevating inositol monophosphate levels followed by phosphoinositols accumulation. Each or both may mediate lithium-induced behavior 28695546_SMIT1 overexpression contributes to abnormal cholinergic and glutamatergic Ca(2+) signals in the trisomic condition |
ENSG00000198743 |
SLC5A3 |
334.702613 |
0.3773878549 |
-1.405880 |
0.19448962 |
50.812389 |
0.00000000000101630178745181212812887340263629318152030500677085456118220463395118713378906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000202887402705976438041519007817454329188622264013019957928918302059173583984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
175.659824836542 |
23.7524821217415 |
465.462315657463 |
45.8117558098894 |
| ENSMUSG00000089809 |
320292 |
Rasgef1b |
protein_coding |
Q8JZL7 |
FUNCTION: Guanine nucleotide exchange factor (GEF) with specificity for RAP2A, it doesn't seems to activate other Ras family proteins (in vitro). {ECO:0000269|PubMed:20090772}. |
Alternative splicing;Endosome;Guanine-nucleotide releasing factor;Reference proteome |
|
|
mmu:320292; |
early endosome [GO:0005769]; late endosome [GO:0005770]; midbody [GO:0030496]; plasma membrane [GO:0005886]; guanyl-nucleotide exchange factor activity [GO:0005085]; positive regulation of GTPase activity [GO:0043547]; Ras protein signal transduction [GO:0007265] |
20090772_Upon toll-like receptor activation, RasGEF1b may trigger Ras-like proteins and regulate specific biological activities of macrophages. 28947785_The authors show that knockdown of the lipopolysaccharide (LPS)-induced cytoplasmic circular RNA, mcircRasGEF1B, results in altered expression of a wide array of genes. Pathway analysis revealed an overall enrichment of genes involved in cell cycle progression, mitotic division, active metabolism, and of particular interest, NF-kappaB, LPS signaling pathways, and macrophage activation. 32866686_Toll-like Receptor (TLR)-induced Rasgef1b expression in macrophages is regulated by NF-kappaB through its proximal promoter. 34438395_Circ_RASGEF1B Promotes LPS-Induced Apoptosis and Inflammatory Response by Targeting MicroRNA-146a-5p/Pdk1 Axis in Septic Acute Kidney Injury Cell Model. |
ENSG00000138670 |
RASGEF1B |
26.652794 |
3.0980008055 |
1.631338 |
0.53237535 |
9.380444 |
0.00219312484339535080854477477885211555985733866691589355468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0092185962792918543090570082654267025645822286605834960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.9889644804449 |
15.0936972925601 |
12.9079903431269 |
3.89928394835804 |
| ENSMUSG00000090098 |
|
Alms1-ps2 |
transcribed_unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
20.738157 |
26.0317560128 |
4.702201 |
1.36926201 |
7.244024 |
0.00711373869171179167014695821080749738030135631561279296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0251348800167371863589771407987427664920687675476074218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
34.5796728506878 |
37.9735747101254 |
1.32836497213948 |
1.20840498538054 |
| ENSMUSG00000090257 |
|
Gm4524 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
23.860664 |
0.2334759249 |
-2.098654 |
0.58058231 |
12.865694 |
0.00033466084550857561280903795797314614901551976799964904785156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0017541479693002834383541710394638357684016227722167968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
9.70721621774898 |
4.01276373009648 |
41.5769472733089 |
11.5574254663069 |
| ENSMUSG00000090273 |
100504446 |
Prr22 |
protein_coding |
F6TNE3 |
|
Reference proteome |
|
|
mmu:100504446; |
|
|
ENSG00000212123 |
PRR22 |
34.336206 |
2.2072138799 |
1.142226 |
0.43541330 |
6.866138 |
0.00878442773802995749421462789996439823880791664123535156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0300279181565691923039285171626033843494951725006103515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
49.7241473137599 |
15.2358841045697 |
22.5280149629423 |
5.28780735095201 |
| ENSMUSG00000090622 |
320700 |
A930033H14Rik |
lncRNA |
G5E8X6 |
|
Reference proteome |
|
|
mmu:320700; |
|
|
|
|
362.776248 |
0.4335481262 |
-1.205736 |
0.13690287 |
76.917499 |
0.00000000000000000178250755805593727666779276021263897632015949308483936802671721011392946820706129074096679687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000628498961210834211328856409352230058324088502624306384891639254419715143740177154541015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
216.245842466853 |
20.5512084017795 |
498.781633275414 |
33.9214621214865 |
| ENSMUSG00000090638 |
|
Gm17028 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
116.236685 |
0.3583977214 |
-1.480367 |
0.29282520 |
25.146351 |
0.00000053140095542270562866559889189654874996904254658147692680358886718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000049536715746625772414739839744068916616015485487878322601318359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.5942195855523 |
13.2009576341411 |
180.230553188111 |
26.3162659952217 |
| ENSMUSG00000090799 |
546611 |
Klhl33 |
protein_coding |
B7ZMY3 |
|
Reference proteome |
|
|
|
|
|
ENSG00000185271 |
KLHL33 |
348.452000 |
0.4900005882 |
-1.029145 |
0.15235178 |
45.304269 |
0.00000000001686810245674240146607638788103057181713695378277861891547217965126037597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000002929702812099204722556349656621341170925632013677386566996574401855468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
226.238260872952 |
23.9356899310134 |
461.710182231573 |
35.4855466622022 |
| ENSMUSG00000090919 |
241989 |
Pabpc4l |
protein_coding |
A0A0A6YWT2 |
Human_homologues FUNCTION: May bind RNA. {ECO:0000305}. |
Reference proteome;Repeat;RNA-binding |
|
|
|
cytoplasmic stress granule [GO:0010494]; cytosol [GO:0005829]; nucleus [GO:0005634]; ribonucleoprotein complex [GO:1990904]; mRNA 3'-UTR binding [GO:0003730]; poly(A) binding [GO:0008143]; poly(U) RNA binding [GO:0008266]; RNA binding [GO:0003723] |
Human_homologues 31537871_Segregation and potential functional impact of a rare stop-gain PABPC4L variant in familial atypical Parkinsonism. |
ENSG00000254535 |
PABPC4L |
14.787686 |
4.9160361591 |
2.297496 |
0.91421840 |
5.839811 |
0.01566750611646602203697042909880110528320074081420898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0491549852041216137066292901636188616976141929626464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
18.4049632560618 |
12.4593922095028 |
3.74386246570729 |
1.94882748350617 |
| ENSMUSG00000090958 |
434215 |
Lrrc32 |
protein_coding |
G3XA59 |
FUNCTION: Key regulator of transforming growth factor beta (TGFB1, TGFB2 and TGFB3) that controls TGF-beta activation by maintaining it in a latent state during storage in extracellular space (PubMed:25127859). Associates specifically via disulfide bonds with the Latency-associated peptide (LAP), which is the regulatory chain of TGF-beta, and regulates integrin-dependent activation of TGF-beta (PubMed:25127859, PubMed:28912269). Able to outcompete LTBP1 for binding to LAP regulatory chain of TGF-beta (By similarity). Controls activation of TGF-beta-1 (TGFB1) on the surface of activated regulatory T-cells (Tregs) (PubMed:25127859). Required for epithelial fusion during palate development by regulating activation of TGF-beta-3 (TGFB3) (PubMed:28912269). {ECO:0000250|UniProtKB:Q14392, ECO:0000269|PubMed:25127859, ECO:0000269|PubMed:28912269}. |
Cell membrane;Disulfide bond;Glycoprotein;Growth factor binding;Leucine-rich repeat;Membrane;Reference proteome;Repeat;Signal;Transmembrane;Transmembrane helix |
|
|
mmu:434215; |
cell surface [GO:0009986]; extracellular matrix [GO:0031012]; extracellular space [GO:0005615]; nucleoplasm [GO:0005654]; plasma membrane [GO:0005886]; transforming growth factor beta binding [GO:0050431]; negative regulation of activated T cell proliferation [GO:0046007]; negative regulation of cytokine production [GO:0001818]; positive regulation of gene expression [GO:0010628]; regulation of transforming growth factor beta activation [GO:1901388]; regulation of transforming growth factor beta3 activation [GO:1901398]; secondary palate development [GO:0062009]; transforming growth factor beta receptor signaling pathway [GO:0007179] |
18628982_GARP is a regulatory T cell specific cell surface molecule that mediates suppressive signals and induces Foxp3 expression 23576681_These findings suggest a role for GARP in natural and induced Treg development through activation of bound latent TGF-beta and signaling, which negatively regulates GARP expression on Tregs. 24115398_We conclude that a missense mutation in VPS54, an essential component of the Golgi-associated retrograde protein complex, not only prevents the formation of an acrosome but also initiates a cascade of metabolic abnormalities and a stress reaction. 25182959_GARP expression on MSCs contributed to their ability to inhibit T-cell responses in vitro. 26246140_These results demonstrate the unexpected presence of GARP on Hepatic stellate cells and its significance in regard to the ability of Hepatic stellate cells to activate latent TGF-beta1 and thereby inhibit T cells. 28912269_Data (including data from studies using knockout mice) suggest that Garp/Lrrc32 is involved in up-regulation of Tgfb3 and is essential for normal embryogenesis of palate; knockout of Garp causes postnatal lethality, cleft palate, and decreased apoptosis and Smad2 phosphorylation in medial edge epithelial cells of palatal shelf of embryos. (Tgfb3 = transforming growth factor beta 3; Smad2 = MAD homolog protein 2) 29618665_Study demonstrated that B cells are required for the induction of oral tolerance of T cell-dependent antigens via GARP and revealed for the first time that cell surface GARP-TGF-beta is an important checkpoint for regulating B cell peripheral tolerance, highlighting a mechanism of autoimmune disease pathogenesis. 31915300_Thrombin contributes to cancer immune evasion via proteolysis of platelet-bound GARP to activate LTGF-beta. |
ENSG00000137507 |
LRRC32 |
97.608101 |
0.4435804830 |
-1.172732 |
0.28622158 |
16.687891 |
0.00004406133395097937505233837152118780977616552263498306274414062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002875502308231867596224895500256479863310232758522033691406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
57.3061940266328 |
11.4439154972238 |
129.190070847487 |
18.6187758121736 |
| ENSMUSG00000091243 |
73569 |
Vgll3 |
protein_coding |
E9Q1Y1 |
FUNCTION: May act as a specific coactivator for the mammalian TEFs. {ECO:0000256|ARBA:ARBA00002229}. |
Nucleus;Reference proteome;Transcription;Transcription regulation |
|
|
Human_homologues hsa:389136; |
nucleus [GO:0005634]; regulation of DNA-templated transcription [GO:0006355] |
19118645_In embryonic and fetal development VITO-2 is mainly expressed in the myogenic lineage.Later developmental stages it is found in the nervous system.In adult mice it was detected in skeletal muscle, heart, kidney, liver, brain. 23152581_vestigial-like 3 (Vgll3) is an inhibitor of adipogenesis 23415753_Loss of VGLL3 expression was observed in tumors derived from mouse xenograft models. 30996136_Study demonstrated that skin-directed overexpression of murine female-biased factor VGLL3 causes a severe lupus-like rash and systemic autoimmune disease that involves B cell expansion, autoantibody production, immune complex deposition, and end-organ damage. Results strongly implicate VGLL3 as a pivotal orchestrator of sex-biased autoimmunity. 35750933_Vestigial-Like 3 Plays an Important Role in Osteoblast Differentiation by Regulating the Expression of Osteogenic Transcription Factors and BMP Signaling. 36046460_lncRNA Vgll3 Regulates the Activated Proliferation of Mouse Myocardial Fibroblasts through TGF-beta3-Related Pathway. 36754961_VGLL3 is a mechanosensitive protein that promotes cardiac fibrosis through liquid-liquid phase separation. |
ENSG00000206538 |
VGLL3 |
1369.770381 |
0.3258535000 |
-1.617705 |
0.08087101 |
392.757800 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000207733297841617187719142875218028363453560023643854121833867434001104972561881404526008304202726256095629551191742560485452415672252105883419281557172504019741729269059728663552 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000009588465432495493932191052481544342791047601632171155110648238729063144351475601023036349424491622286276937013490189494146957344705678592894362999598278749918411541694750244828 |
Yes |
No |
670.502382234643 |
37.6050412943794 |
2057.68046759884 |
82.0804609885386 |
| ENSMUSG00000091358 |
70365 |
Speer5-ps1 |
transcribed_processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
57.980505 |
178.2197032807 |
7.477513 |
0.82396785 |
72.032477 |
0.00000000000000002116846246276925975818316489262689975053314273283240620759926287064445205032825469970703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000006831313987985198403318770500942646970341543010731000240554067204357124865055084228515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
114.630487002213 |
69.0867559196114 |
0.643197608861957 |
0.327375872563773 |
| ENSMUSG00000091625 |
66373 |
Lsm5 |
protein_coding |
P62322 |
FUNCTION: Plays role in pre-mRNA splicing as component of the U4/U6-U5 tri-snRNP complex that is involved in spliceosome assembly, and as component of the precatalytic spliceosome (spliceosome B complex). The heptameric LSM2-8 complex binds specifically to the 3'-terminal U-tract of U6 snRNA. {ECO:0000250|UniProtKB:Q9Y4Y9}. |
Acetylation;mRNA processing;mRNA splicing;Nucleus;Reference proteome;Ribonucleoprotein;RNA-binding;Spliceosome |
|
|
mmu:66373; |
cytoplasm [GO:0005737]; Lsm1-7-Pat1 complex [GO:1990726]; Lsm2-8 complex [GO:0120115]; nucleus [GO:0005634]; U2-type precatalytic spliceosome [GO:0071005]; U4/U6 x U5 tri-snRNP complex [GO:0046540]; U6 snRNP [GO:0005688]; protein heterodimerization activity [GO:0046982]; RNA binding [GO:0003723]; mRNA catabolic process [GO:0006402]; mRNA splicing, via spliceosome [GO:0000398]; response to bacterium [GO:0009617] |
Human_homologues 12515382_LSm1-7 proteins colocalize with DCP1,DCP2 and Xrn1 in cytoplasmic foci |
ENSG00000106355 |
LSM5 |
328.832639 |
2.0973335518 |
1.068556 |
0.25979842 |
16.622926 |
0.00004559652716405233506200000981323228188557550311088562011718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002956689236964006877068922296558639573049731552600860595703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
458.832277425538 |
83.073142665952 |
218.769340257882 |
29.847496280403 |
| ENSMUSG00000091845 |
100043718 |
Rpl36-ps12 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
324.681169 |
2.0080358968 |
1.005785 |
0.20726855 |
22.709823 |
0.00000188402340024080347677881090012741438499688229057937860488891601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000159607588612168627638897289156005854238173924386501312255859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
457.974768727603 |
66.022717315414 |
228.071006823217 |
24.8672431746088 |
| ENSMUSG00000091898 |
21924 |
Tnnc1 |
protein_coding |
P19123 |
FUNCTION: Troponin is the central regulatory protein of striated muscle contraction. Tn consists of three components: Tn-I which is the inhibitor of actomyosin ATPase, Tn-T which contains the binding site for tropomyosin and Tn-C. The binding of calcium to Tn-C abolishes the inhibitory action of Tn on actin filaments. |
3D-structure;Acetylation;Calcium;Metal-binding;Muscle protein;Phosphoprotein;Reference proteome;Repeat |
|
|
mmu:21924; |
cardiac Troponin complex [GO:1990584]; contractile fiber [GO:0043292]; troponin complex [GO:0005861]; actin filament binding [GO:0051015]; calcium ion binding [GO:0005509]; calcium-dependent protein binding [GO:0048306]; protein homodimerization activity [GO:0042803]; troponin I binding [GO:0031013]; troponin T binding [GO:0031014]; cardiac muscle contraction [GO:0060048]; diaphragm contraction [GO:0002086]; regulation of ATP-dependent activity [GO:0043462]; regulation of muscle contraction [GO:0006937]; regulation of muscle filament sliding speed [GO:0032972]; response to metal ion [GO:0010038]; skeletal muscle contraction [GO:0003009]; transition between fast and slow fiber [GO:0014883]; ventricular cardiac muscle tissue morphogenesis [GO:0055010] |
12062407_Overexpression of sTnC polypeptide in muscle cells is controlled by its rapid degradation. 18285522_These results demonstrate that the L29Q mutation enhances the Ca(2+)-binding characteristics of cTnC and that when incorporated into cardiac myocytes, this mutant alters myocyte contractility. 20566645_A region in cTnC associated with increased Ca(2+) sensitivity in skinned fibers was identified, an the F27W reporter mutation affected Ca(2+) sensitivity, maximal force, and ATPase activation of some mutants. 21035455_Calcium binding kinetics of troponin C strongly modulate cooperative activation and tension kinetics in cardiac muscle. 32189431_Sexual dimorphism in cardiac transcriptome associated with a troponin C murine model of hypertrophic cardiomyopathy. 32931484_Modulating the tension-time integral of the cardiac twitch prevents dilated cardiomyopathy in murine hearts. |
ENSG00000114854 |
TNNC1 |
145.259281 |
2.3445636612 |
1.229319 |
0.27467194 |
19.231074 |
0.00001158128125553450897707290917493239135183102916926145553588867187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000848106135020680997407827028666815749602392315864562988281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
204.96524434816 |
39.2591149626687 |
87.4214881588766 |
12.8279522425938 |
| ENSMUSG00000092178 |
72716 |
Gm45351 |
unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
103.703599 |
0.4182688737 |
-1.257497 |
0.31577578 |
15.552717 |
0.00008023626084497204166614409137636698687856551259756088256835937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004869158267691690053328990028802536471630446612834930419921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
64.651926111164 |
14.2642164987009 |
154.570254134402 |
24.577615341684 |
| ENSMUSG00000094796 |
625360 |
BC147527 |
protein_coding |
Q3V185 |
|
Reference proteome |
|
|
mmu:625360; |
|
|
|
|
69.026211 |
0.4859287899 |
-1.041183 |
0.31923817 |
10.413835 |
0.00125074725637260106975157114561625348869711160659790039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0056381717102892752943588128289320593466982245445251464843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
44.5661450843712 |
9.94222852898325 |
91.7133250976304 |
14.8524509477197 |
| ENSMUSG00000095304 |
211623 |
Plac9 |
protein_coding |
Q8K262 |
|
Coiled coil;Reference proteome;Secreted;Signal |
|
|
mmu:211623; |
extracellular region [GO:0005576] |
Human_homologues 30291214_Plac9 is a novel protein that is localized to the cytoplasm and may be secreted through the classic endoplasmic reticulum-Golgi route. The overexpression of Plac9 inhibits cell growth and induces G2/M phase arrest. Conclusion: Our findings reveal a novel role for Plac9 in regulating cell growth. |
ENSG00000189129 |
PLAC9 |
381.037797 |
4.3709142599 |
2.127935 |
0.17819261 |
136.486985 |
0.00000000000000000000000000000015613369815341226035937899206085758191849975345778854871155341511876658333967093352998012356636081676697358489036560058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000129251548384389969135053793684040691858114306973700007958281440296833030969354989825337298725571599788963794708251953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
637.665454678777 |
78.9608319950505 |
145.888346639351 |
14.1973570975813 |
| ENSMUSG00000095546 |
100042175 |
Gm10230 |
protein_coding |
Q62478 |
Human_homologues FUNCTION: Component of the synaptonemal complexes (SCS), formed between homologous chromosomes during meiotic prophase. Required for centromere pairing during meiosis in male germ cells (By similarity). Required for normal meiosis during spermatogenesis and male fertility (PubMed:14643120). Plays a lesser role in female fertility. Required for efficient phosphorylation of HORMAD1 and HORMAD2 (By similarity). {ECO:0000250|UniProtKB:P70281, ECO:0000269|PubMed:14643120}. |
Coiled coil;Nucleus;Reference proteome |
|
|
mmu:100039240;mmu:100039324;mmu:100039377;mmu:100039467;mmu:100039550;mmu:100039585;mmu:100042144;mmu:100042175;mmu:22526; |
synaptonemal complex [GO:0000795]; meiotic cell cycle [GO:0051321]; spermatid development [GO:0007286] |
Human_homologues 14643120_Observational study of gene-disease association. (HuGE Navigator) 14643120_We suggest that SYCP3 has an essential meiotic function in human spermatogenesis that is compromised by the mutant protein via dominant negative interference. 16213863_In contrast to previously reported high frequency of SYCP3 mutations in patients with azoospermia, only polymorphisms are found in the present study 16824523_SYCP3 is expressed in human testis and is restricted to germ cells. Lack of SYCP3 expression in human testis may have a negative effect on spermatogenesis and male fertility. 17434513_No functional SYCP3 exonic SNP was found in infertile Spanish patients with meiosis arrest, suggesting that SYCP3 mutations very likely are not relevant in Spain in genetic susceptibility to meiosis arrest. 18256545_These observations suggest that SCP3 is expressed in adult human and monkey ovaries. 19110213_Observational study of gene-disease association. (HuGE Navigator) 19110213_mutations in SYPR3 are likely to generate an aberrant synaptonemal complex in a dominant-negative manner and contribute to abnormal chromosomal behavior that might lead to recurrent miscarriage. 20395201_X-linked lymphocyte-regulated protein pM1 (XMR), XLR, and SCP3 render tumor cells resistant to antitumor immunity 21126912_mutation analysis of SYCP3, DNMT3L and MSH4 in patients with maturation arrest of spermatogenesis and couples with recurrent miscarriages; heterozygous change, detected in a conserved functional domain of the SYCP3 gene, was absent in >200 controls 21357605_The 657T>C mutation of SYCP3 may not be associated with recurrent miscarriage caused by aneuploidy. 22116401_Expression of SYCP3 inhibits the homologous recombination (HR) pathway mediated by RAD51. 22197129_mutations in SYCP3 do not contribute significantly to risk for recurrent miscarriage through maternal meiotic nondisjunction. 22670862_SYCP3 mutations are not associated with the genetic susceptibility for meiotic arrest in infertile male patients with nonobstructive azoospermia in the Turkish population 23069255_Positive synaptonemal complex protein 3 expression is a portent of poor outcome and may be a potential biomarker in the early stages of the non-small cell lung cancer for survival. 23100464_screening for genetic variants in AURKB and SYCP3 among these patients with reproductive problems using Sanger sequencing. Only one apparently non-pathogenic deletion was found in SYCP3. 24905095_SCP3 plays an important role in the progression of cervical cancer 24950965_SYCP3 binding and assembly on meiotic chromosomes leads to their organisation into compact structures compatible with recombination and crossover formation 25059562_The T657C polymorphism of the SYCP3 gene is possibly associated with recurrent pregnancy loss of unknown cause in human. 28287952_In the model, the reported compaction of chromosomal DNA caused by SYCP3 would result from its ability to bridge distant sites on a DNA molecule with the DNA-binding domains located at each end of its strut-like structure. 28623914_SCP3 is associated with lymphangiogenesis and provides insight into the SCP3-VEGF-C/VEGF-D axis based cancer therapy strategy. 28745000_SYCP3 binds to RAD51 and attenuates RAD51 activity during meiosis. |
ENSG00000139351 |
SYCP3 |
17.059794 |
0.0489688192 |
-4.351993 |
1.32724219 |
7.065064 |
0.00786014953060416333985305215037442394532263278961181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0273284176329976306873970059996281634084880352020263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.43770153572381 |
2.57262378070937 |
49.7806885233274 |
32.8012835577606 |
| ENSMUSG00000096727 |
16912 |
Psmb9 |
protein_coding |
A0A0R4J256 |
FUNCTION: Component of the proteasome, a multicatalytic proteinase complex which is characterized by its ability to cleave peptides with Arg, Phe, Tyr, Leu, and Glu adjacent to the leaving group at neutral or slightly basic pH. The proteasome has an ATP-dependent proteolytic activity. {ECO:0000256|RuleBase:RU004203}. |
Cytoplasm;Nucleus;Proteasome;Proteomics identification;Reference proteome |
|
|
mmu:16912; |
cytosol [GO:0005829]; nucleus [GO:0005634]; proteasome core complex [GO:0005839]; threonine-type endopeptidase activity [GO:0004298]; immune system process [GO:0002376]; proteolysis involved in protein catabolic process [GO:0051603]; regulation of cysteine-type endopeptidase activity [GO:2000116] |
11782352_Development of spontaneous uterine tumors in low molecular mass polypeptide-2 knockout mice. 12220526_The presence of immuno subunits in nonimmune tissues such as lens suggests that the immunoproteasome may also have nonimmune functions, such as that in lens differentiation. 15036617_Comparison of LMP2 KO mice and non-transgenic mice of the same background show that LMP2 KO mice exhibit a higher degree of motor function, but possess a similar level of cognitive function and ambulation. 15356141_Immunoproteasome LMP2 can at least in part determine subdominance and shape the epitope hierarchy of cytotoxic T cell responses in vivo. 16002717_Immunoproteasome LMP2 may affect CD8+ T cell responses to only a limited number of viral epitopes; its main biological function may lie elsewhere. 17079320_These results demonstrate the ability of the IFN-induced proteasome catalytic subunits to shape the HBV-specific CD8 T-cell response and thus potentially influence the progression of infection to acute or chronic disease. 17142736_two inducible subunits of the proteasome, lmp2 and lmp7, are transcriptionally up-regulated by heat shock; heat-shocked cells show enhanced presentation of immunoproteasome-dependent MHC I antigenic epitopes, but not immunoproteasome-independent epitopes 17482163_SOD1/LMP2-/- mice show no change in motor function decline, or survival compared to G93A SOD1 mice, suggesting that the loss of immuno-proteasome function in vivo does not significantly alter mutant SOD1-induced disease. 18728217_LMP-2 is required for normal proteasomal function and ischemic preconditioning induction in the heart. Its action may be related to PTEN protein degradation. 20228196_Mixed proteasomes present in LMP2-deficient mice compromise virtually every parameter of the immune response against influenza A virus, potentially through dysregulation of signaling pathways, such as the nuclear factor (NF)-kappaB pathway. 22355695_LMP2 has a role as a tumor-suppressor and may have a role in uterine leiomyosarcoma therapy 22772448_In this study, we have identified how LMP2 can exert its function in Ag processing 24164898_Data show that knockdown of i-proteasome catalytic subunit PSMB9 by short hairpin RNA (shRNA) decreased the expression of both PSMB9 and PSMB8 without affecting other catalytic subunits of the proteasome. 24619410_down-regulation of IL-33 protein levels by IFN-gamma in pulmonary fibroblasts and in the lungs in vivo occurs through STAT1 and non-canonical use of the LMP2 proteasome 26254356_Although treatment with IFN-gamma strongly induced the activation of STAT1 as a transcriptional activator, its target molecule, IRF1, was not clearly produced in Lmp2-deficient uterine smooth muscle cells. 29738610_LMP2 is crucial for lymphocyte development and survival in the periphery. LMP2-deficient lymphocytes show impaired degradation of key BH3-only proteins, resulting in elevated levels of pro-apoptotic BIM and increased cell death. 30279279_co-inhibition of LMP2 and LMP7 appears to be synergistic and advantageous for the treatment of autoimmune diseases. 34132031_Immunoproteasome impairment via beta5i/LMP7-deletion leads to sustained pancreatic injury from experimental pancreatitis. 34547302_Expression of immunoproteasome subunits in the brains of Toxoplasma gondii-infected mice. 34819510_Heterozygous missense variant of the proteasome subunit beta-type 9 causes neonatal-onset autoinflammation and immunodeficiency. |
ENSG00000240065 |
PSMB9 |
177.390288 |
4.1738941524 |
2.061394 |
0.21622097 |
89.697177 |
0.00000000000000000000277550783075677047282636979904736717680105230347138751690596811250166808804351603612303733825683593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000001167860090554892953178124298026376268692465918355304251019521899479514104314148426055908203125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
302.965253326261 |
45.5764749114312 |
72.5857537976314 |
8.7111404624826 |
| ENSMUSG00000097072 |
768252 |
Foxl2os |
lncRNA |
|
|
|
|
|
|
|
|
|
|
427.893609 |
0.3521772975 |
-1.505626 |
0.13094925 |
130.872915 |
0.00000000000000000000000000000263976659396867232019334694153315543015292207937237349904507776960595200394150451617125696657240041531622409820556640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000002086271812056917251406593241048493505001896585630159679179203106790473597198509736472260556183755397796630859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
221.028606963959 |
20.0896845474461 |
627.606062376293 |
40.4528697695327 |
| ENSMUSG00000097113 |
100503460 |
Gm19705 |
lncRNA |
|
|
|
|
|
|
|
34002693_PHAROH lncRNA regulates Myc translation in hepatocellular carcinoma via sequestering TIAR. |
|
|
89.506456 |
0.4487197462 |
-1.156113 |
0.28648058 |
16.249184 |
0.00005553351524510849267614667756909341278515057638287544250488281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003517199601719304198794990856669073764351196587085723876953125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
57.490529741945 |
11.4912521519603 |
128.121238776496 |
18.3941374935303 |
| ENSMUSG00000097124 |
105242407 |
A530020G20Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
19.087356 |
3.2418508091 |
1.696818 |
0.62182427 |
7.378253 |
0.00660172568473072710754090408613592444453388452529907226562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0235607979451308430229872215022624004632234573364257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
29.8907532010085 |
13.2860076276959 |
9.22027414627039 |
3.19688737296736 |
| ENSMUSG00000097184 |
100043102 |
4632428C04Rik |
lncRNA |
|
|
|
|
|
|
|
32643756_A long noncoding RNA CHAIR protects the heart from pathological stress. |
|
|
8.555359 |
0.0991369273 |
-3.334434 |
1.13737328 |
8.127892 |
0.00435894089627185312568746766714866680558770895004272460937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0165368338062298544932637867077573901042342185974121093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.71044310292399 |
1.49254539311577 |
17.2533399014208 |
8.94027250643381 |
| ENSMUSG00000097248 |
100040294 |
Gm2694 |
lncRNA |
|
|
|
|
|
|
|
36459549_Long noncoding RNA Gm2694 drives depressive-like behaviors in male mice by interacting with GRP78 to disrupt endoplasmic reticulum homeostasis. |
|
|
12.944027 |
0.2286761176 |
-2.128622 |
0.79976565 |
6.882293 |
0.00870537983758642129605842541195670492015779018402099609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0298112287963391113521094410998557577840983867645263671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.2774150268976 |
2.49453679531568 |
18.7051235287872 |
7.7867164267169 |
| ENSMUSG00000097254 |
320021 |
C430042M11Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
13.554428 |
6.2397328814 |
2.641484 |
0.76820523 |
11.989820 |
0.00053491969690925666829073303887298607151024043560028076171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0026471399685905772775074140668039035517722368240356445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.9928808574014 |
12.8300530192824 |
3.68491428949146 |
1.69620980121487 |
| ENSMUSG00000097462 |
330385 |
9530026P05Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
15.862131 |
4.0566924630 |
2.020304 |
0.80989083 |
5.914623 |
0.01501571287892959453980079587154250475578010082244873046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0473735166884539302123435788871574914082884788513183593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
26.0626339488903 |
15.4115360168799 |
6.42460186135471 |
2.96141214171371 |
| ENSMUSG00000097487 |
73635 |
Ptges3l |
protein_coding |
Q9D9A7 |
|
Alternative splicing;Reference proteome |
|
|
mmu:73635; |
cytosol [GO:0005829]; nucleus [GO:0005634]; chaperone binding [GO:0051087]; Hsp90 protein binding [GO:0051879]; chaperone-mediated protein complex assembly [GO:0051131]; protein folding [GO:0006457] |
|
ENSG00000267060 |
PTGES3L |
537.663094 |
0.2621687583 |
-1.931432 |
0.28997858 |
40.940715 |
0.00000000015691778464697318953174695446478505850285145584166457410901784896850585937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000024372052215050637164998457333871945129644132066459860652685165405273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
247.01098804755 |
49.9836516170421 |
942.183155673628 |
141.108840696778 |
| ENSMUSG00000097616 |
76311 |
1110019D14Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
113.883111 |
2.0715702582 |
1.050725 |
0.33781143 |
9.576947 |
0.00197035805001067676825443619748057244578376412391662597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0083951031658077279046237251236561860423535108566284179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
158.495129921022 |
37.4521762305169 |
76.5096570062849 |
13.636466331945 |
| ENSMUSG00000097697 |
73904 |
4833412C05Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
21.678292 |
8.1651888562 |
3.029486 |
0.75336620 |
15.952268 |
0.00006495989039392894247564730347832551160536240786314010620117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004035355018272127102310631308057509158970788121223449707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.8329630298787 |
20.6663258923297 |
4.63344616961111 |
2.10332333364087 |
| ENSMUSG00000097767 |
330166 |
Miat |
lncRNA |
|
|
|
|
This gene encodes a spliced long non-coding RNA that may constitute a component of the nuclear matrix. This transcript has been associated with retinal cell fate determination. Altered expression of a similar gene in human has been associated with a susceptibility to myocardial infarction, and is involved in pathways that may contribute to the pathophysiology of schizophrenia. Alternatively spliced transcript variants have been identified. [provided by RefSeq, Dec 2014]. |
|
|
17623775_it is proposed that Gomafu RNA represents a novel family of mRNA-like noncoding RNA that constitutes a cell-type-specific component of the nuclear matrix 20026622_implicated in the regulation of Oct4 in mouse ES cells 20459797_These data demonstrate that the lncRNA RNCR2 plays a critical role in regulating mammalian retinal cell fate specification. 24240175_Miat controls the differentiation of neural progenitor cells. 25587098_qRT-PCRs detected higher levels of MIAT in the retinas of db/db mice than in their nondiabetic controls. 27251103_Observations suggest that Gomafu modifies mouse behavior thorough a mild modulation of gene expression and/or alternative splicing of target genes. 27391072_MIAT knockdown leads to cerebral microvascular degeneration, progressive neuronal loss and neurodegeneration, and behavioral deficits in a CNS neurovascular disorder, Alzheimer's disease model 28198439_Our study therefore has identified MIAT as the first pro-fibrotic lncRNA in heart and unraveled the role of MIAT in the pathogenesis of MI. 28246353_MiR-29b controled MIAT to regulate its expression and MIAT overexpression suppressed miR-29b, but promoted Sp1. High glucose stimulation increased the cell apoptosis and decreased the cell activity, while MIAT suppression reversed the effect induced by high glucose, however, miR-29b knockdown reversed the effects induced by MIAT suppression. 29459686_Knockdown of hepatic Gomafu inhibited hepatic glucose production (HGP) and improved insulin sensitivity in obese mice. 29914974_MIAT transcript levels were significantly elevated in breast cancer samples. Such increase was specific to the early stages of the disease. Silencing of MIAT induced growth arrest and increased basal apoptosis. 30556210_MIAT could elevate the expression of SIRT1/PGC-1alpha via downregulating miR-29b. And the downregulated SIRT/PGC-1alpha increased the incidence of age-related hearing loss via promoting the apoptosis of cochlear hair cells. 30755588_MIAT was consistently increased in serum and macrophages of necrotic cores in an advanced atherosclerosis mouse model. These data also demonstrated how MIAT inhibited efferocytosis by targeting miR-149-5p/CD47 axis. 30971184_The study suggests that targeting MIAT may protect against cardiomyocytes against hypoxia-reperfusion injury or myocardial ischemia-reperfusion injury via inhibition of cell apoptosis, mediated by NF-kappaB and PUMA signaling pathway. 31204523_silence of MIAT protected ATDC5 cells against LPS challenge 31237148_Upregulation of MIAT can aggravate AS injury in AS mice. 32556678_LncRNA MIAT Promotes Inflammation and Oxidative Stress in Sepsis-Induced Cardiac Injury by Targeting miR-330-5p/TRAF6/NF-kappaB Axis. 32972755_Ablation of lncRNA MIAT mitigates high glucose-stimulated inflammation and apoptosis of podocyte via miR-130a-3p/TLR4 signaling axis. 33819189_Silencing long non-coding RNA MIAT ameliorates myocardial dysfunction induced by myocardial infarction via MIAT/miR-10a-5p/EGR2 axis. 33941765_LncRNA MIAT impairs cardiac contractile function by acting on mitochondrial translocator protein TSPO in a mouse model of myocardial infarction. 33942232_Overexpressing microRNA-203 alleviates myocardial infarction via interacting with long non-coding RNA MIAT and mitochondrial coupling factor 6. 34265390_Long non-coding RNA MIAT impairs neurological function in ischemic stroke via up-regulating microRNA-874-3p-targeted IL1B. 34335976_Ablation of lncRNA Miat attenuates pathological hypertrophy and heart failure. 34459123_Serum extracellular vesicles containing MIAT induces atrial fibrosis, inflammation and oxidative stress to promote atrial remodeling and atrial fibrillation via blockade of miR-485-5p-mediated CXCL10 inhibition. 34489160_LncRNA MIAT knockdown alleviates oxygen-glucose deprivationinduced cardiomyocyte injury by regulating JAK2/STAT3 pathway via miR-181a-5p. 34709954_LncRNA MIAT Promotes Allergic Inflammation and Symptoms by Targeting MiR-10b-5p in Allergic Rhinitis Mice. 34953943_Knockdown of long noncoding RNA MIAT attenuates cigarette smoke-induced airway remodeling by downregulating miR-29c-3p-HIF3A axis. 34967706_Long non-coding RNA myocardial infarction-associated transcript promotes 1-Methyl-4-phenylpyridinium ion-induced neuronal inflammation and oxidative stress in Parkinson's disease through regulating microRNA-221-3p/ transforming growth factor /nuclear factor E2-related factor 2 axis. 36122770_LncRNA Miat promotes neuropathic pain through miR-362-3p/BAMBI signaling axis. |
|
|
38.557789 |
0.0362482977 |
-4.785943 |
0.63997862 |
62.871009 |
0.00000000000000220696867046048718883486364613554566966398175903441014611416903790086507797241210937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000580596663013024859602128739217035285370954039718682793136395048350095748901367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.92500705507266 |
1.34050676327269 |
80.6936392408127 |
19.5982115498813 |
| ENSMUSG00000097789 |
100039239 |
Ctxnd1 |
protein_coding |
A0A1B0GST9 |
|
Membrane;Reference proteome;Transmembrane;Transmembrane helix |
|
|
|
membrane [GO:0016020] |
Human_homologues 36246563_Analysis of LINC01314 and miR-96 Expression in Colorectal Cancer Patients via Tissue Microarray-Based Fluorescence In Situ Hybridization. |
ENSG00000259417 |
CTXND1 |
35.789146 |
0.0426124537 |
-4.552581 |
0.67471769 |
50.328242 |
0.00000000000130064206322721007850918591844541091990042347426026481116423383355140686035156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000255630708478411140672196275250367312158183974446501451893709599971771240234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.0419421507368 |
1.4750827425354 |
71.386223665277 |
18.4005458850192 |
| ENSMUSG00000097906 |
674597 |
Gm9625 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
51.464790 |
0.2622434042 |
-1.931022 |
0.50952467 |
13.783580 |
0.00020512113344825752049051248437905314858653582632541656494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011340853374533061380391441019810372381471097469329833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.7381305498673 |
7.11685182023625 |
75.2664518394207 |
19.2385637762446 |
| ENSMUSG00000098134 |
66381 |
Rnf113a2 |
protein_coding |
Q14B01 |
Human_homologues FUNCTION: Required for pre-mRNA splicing as component of the spliceosome (PubMed:29361316, PubMed:29360106). E3 ubiquitin-protein ligase that catalyzes the transfer of ubiquitin onto target proteins (PubMed:28978524, PubMed:29144457). Catalyzes polyubiquitination of SNRNP200/BRR2 with non-canonical 'Lys-63'-linked polyubiquitin chains (PubMed:29144457). Plays a role in DNA repair via its role in the synthesis of 'Lys-63'-linked polyubiquitin chains that recruit ALKBH3 and the ASCC complex to sites of DNA damage by alkylating agents (PubMed:29144457). Ubiquitinates CXCR4, leading to its degradation, and thereby contributes to the termination of CXCR4 signaling (PubMed:28978524). {ECO:0000269|PubMed:28978524, ECO:0000269|PubMed:29144457, ECO:0000269|PubMed:29360106, ECO:0000269|PubMed:29361316}. |
Metal-binding;Proteomics identification;Reference proteome;Zinc;Zinc-finger |
Human_homologues PATHWAY: Protein modification; protein ubiquitination. {ECO:0000269|PubMed:28978524, ECO:0000269|PubMed:29144457}. |
|
mmu:66381; |
nucleoplasm [GO:0005654]; nucleus [GO:0005634]; U2-type precatalytic spliceosome [GO:0071005]; U2-type spliceosomal complex [GO:0005684]; metal ion binding [GO:0046872]; ubiquitin protein ligase activity [GO:0061630]; isopeptide cross-linking via N6-glycyl-L-lysine [GO:0018276]; mRNA splicing, via spliceosome [GO:0000398]; negative regulation of chemokine-mediated signaling pathway [GO:0070100]; protein ubiquitination [GO:0016567]; snoRNA splicing [GO:0034247] |
Human_homologues 25612912_A mutation that resulted in significant reduction of RNF113A protein was implicated in a non-photosensitive form of trichothiodystrophy. Lymphoblastoid cell lines from patients with the mutation in RNF113A had normal DNA damage responses on exposure to mitomycin C, diepoxybutane (DEB), bromodeoxyuridine (BrdU) and irradiation with ultraviolet light. 29393393_RNF113A promotes the proliferation, migration and invasiveness of esophageal squamous cell carcinoma (ESCC) cell lines. RNF113A expression in ESCC is associated with a poor prognosis of affected patients. 31793730_Second report of RING finger protein 113A (RNF113A) involvement in a Mendelian disorder. 31880405_A novel truncating variant in ring finger protein 113A (RNF113A) confirms the association of this gene with X-linked trichothiodystrophy. 31985874_Ring Finger Protein 11 acts on ligand-activated EGFR via the direct interaction with the UIM region of ANKRD13 protein family. 32152280_RNF113A deficiency circumvents the resistance to Cisplatin and to BCL-2 inhibitors through the destabilization of MCL-1. 35819319_SMYD3 Impedes Small Cell Lung Cancer Sensitivity to Alkylation Damage through RNF113A Methylation-Phosphorylation Cross-talk. |
ENSG00000125352 |
RNF113A |
13.706428 |
0.2057006983 |
-2.281381 |
0.75483348 |
9.100890 |
0.00255485198355484381671498539390086079947650432586669921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0104893545588968673670793307906023983377963304519653320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
4.45871482344627 |
2.44074959582397 |
21.6757398465451 |
7.82065194889513 |
| ENSMUSG00000098206 |
|
Mir181a-1hg |
lncRNA |
|
|
|
|
|
|
|
|
|
|
36.360063 |
0.4050856900 |
-1.303701 |
0.44391858 |
8.456930 |
0.00363655125916549160428048459436922712484374642372131347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0141161948979634972733965270208500442095100879669189453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
21.6586158549901 |
6.77003846461576 |
53.4667513271612 |
11.8101460123989 |
| ENSMUSG00000098318 |
381822 |
Lockd |
lncRNA |
|
|
|
|
|
|
|
27041223_Findings indicate that the Cdkn1b gene is positively regulated by a cis element at the promoter of the adjacent Lockd locus, whereas the transcribed Lockd lncRNA is dispensable for this function. 35675771_Lockd promotes myoblast proliferation and muscle regeneration via binding with DHX36 to facilitate 5' UTR rG4 unwinding and Anp32e translation. |
|
|
540.693358 |
2.0265331300 |
1.019014 |
0.16349077 |
38.415671 |
0.00000000057171458345215610156913698034733713193578097389035974629223346710205078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000082779054516570729340608599997573813222828675861819647252559661865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
733.124348560289 |
83.2579067131854 |
361.762824251771 |
31.2731092985969 |
| ENSMUSG00000098557 |
239217 |
Kctd12 |
protein_coding |
A0A0R4J2B2 |
Human_homologues FUNCTION: Auxiliary subunit of GABA-B receptors that determine the pharmacology and kinetics of the receptor response. Increases agonist potency and markedly alter the G-protein signaling of the receptors by accelerating onset and promoting desensitization (By similarity). {ECO:0000250}. |
Proteomics identification;Reference proteome |
|
|
mmu:239217; |
identical protein binding [GO:0042802]; protein homooligomerization [GO:0051260] |
15357420_new gene encoding 327 amino acids in mouse contains a voltage-gated potassium (K+) channel tetramerization (T1) domain. 21452234_Data suggest that the distinct spatial and temporal distribution patterns of KCTD8, 12, 12b, and 16 underlie functional differences in native GABA(B) responses. 23843457_the up-regulation of functional GABA(B) receptors at the neuronal plasma membrane is an additional physiological role of the auxiliary subunit KCTD12. 25065880_Serine-892 phosphorylation in GABAB receptor rearranges KCTD12 at the receptor and slows KCTD12-induced desensitization in neurons. 25689571_KCTD12 is involved in determining phenotypes of emotionality, behavioral activity and neuronal excitability. 33285230_The effects of Kctd12, an auxiliary subunit of GABAB receptor in dentate gyrus on behavioral response to chronic social defeat stress in mice. 35017224_KCTD8 and KCTD12 Facilitate Axonal Expression of GABAB Receptors in Habenula Cholinergic Neurons. |
ENSG00000178695 |
KCTD12 |
304.045411 |
0.4367073649 |
-1.195261 |
0.14734967 |
65.349351 |
0.00000000000000062730579882388658434667491131692418343755973230563283138394581328611820936203002929687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000172475125048473660989169896601604095183447006023325087653574883006513118743896484375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
180.143816811895 |
18.4309948517965 |
412.50464569801 |
30.1953684153272 |
| ENSMUSG00000100162 |
|
Gm20687 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
18.029943 |
0.0451928104 |
-4.467763 |
1.07713625 |
14.632907 |
0.00013061385978236904358874836962201015921891666948795318603515625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0007608070027552753461855261640778280707309022545814514160156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.36777331081532 |
1.11875319079185 |
30.2652855278731 |
15.3209475425518 |
| ENSMUSG00000100546 |
102641120 |
Gm29483 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
78.234869 |
0.3233946321 |
-1.628632 |
0.34668388 |
21.511676 |
0.00000351680881628112742594153075637031236055918270722031593322753906250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000284180540528350851763473433608808704775583464652299880981445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
37.7647640762499 |
9.1625767905033 |
116.776100560466 |
20.1046688817318 |
| ENSMUSG00000100801 |
727711 |
Gm15459 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
38.478245 |
0.0145114855 |
-6.106661 |
1.78300529 |
7.060521 |
0.00788010946973966182327320240119661320932209491729736328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0273665817243671956326966920869381283409893512725830078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.93460006653194 |
3.04781313154969 |
133.315094618694 |
127.276155533133 |
| ENSMUSG00000101429 |
407809 |
BC055402 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
45.647575 |
0.0463682097 |
-4.430720 |
0.57012319 |
69.186777 |
0.00000000000000008956655433549206907635960366486573193158751032046799300800898890884127467870712280273437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000026698194826579553524991327004482770117764012587913136087536258855834603309631347656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.6936461010996 |
1.49794168478832 |
79.659019033752 |
17.0942642663736 |
| ENSMUSG00000101445 |
|
Gm28932 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
35.811857 |
0.1026348951 |
-3.284407 |
0.53521519 |
37.896249 |
0.00000000074608553564537419145489224146122232694811060582651407457888126373291015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000106507730824276854412724867531697681144464695535134524106979370117187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.5358519918259 |
2.48069094675169 |
63.6806028258079 |
14.9048465943233 |
| ENSMUSG00000102252 |
20646 |
Snrpn |
protein_coding |
P63163 |
FUNCTION: May be involved in tissue-specific alternative RNA processing events. |
Methylation;Nucleus;Reference proteome;Repeat;Ribonucleoprotein;RNA-binding |
|
This locus represents a paternally-expressed imprinted gene that encodes a component of the small nuclear ribonucleoprotein complex, which functions in pre-mRNA processing. Genomic and genetic changes in this region result in growth defects and lethality; the corresponding region in human is the critical region for Prader-Willi Syndrome. Alternative promoter use and alternative splicing result in a multitude of transcript variants encoding the same protein. Transcript variants may be bicistronic and also encode the SNRPN upstream reading frame protein (Snurf) from an upstream open reading frame. In addition, long spliced transcripts for small nucleolar RNA host gene 14 (Snhg14) may originate from the promoters at this locus and incorporate exons shared with this gene. [provided by RefSeq, Mar 2017]. |
mmu:20646; |
catalytic step 2 spliceosome [GO:0071013]; cytoplasm [GO:0005737]; nucleoplasm [GO:0005654]; nucleus [GO:0005634]; U1 snRNP [GO:0005685]; U2 snRNP [GO:0005686]; U2-type prespliceosome [GO:0071004]; U4 snRNP [GO:0005687]; U4/U6 x U5 tri-snRNP complex [GO:0046540]; U5 snRNP [GO:0005682]; RNA binding [GO:0003723]; mRNA splicing, via spliceosome [GO:0000398]; response to hormone [GO:0009725] |
9294199_Snrpn gene is imprinted, with monoallelic expression from the paternal allele in brain and heart. 9590284_Mouse model for Prader-Willi syndrome. Deletion of Snrpn gene and putative imprinting-centre is associated with absent expression of the imprinted genes Zfp127, Ndn and Ipw, and phenotypes similar to those found in Prader-Willi infants. 12075010_the ATG-to-AAG mutation causes a 15-fold or more increase in translation of the downstream ORF in two fusion constructs, and it is likely that similar translational control affects the normal Snurf-Snrpn transcript as well 18381202_These data indicate that oocytes undergo time-dependent demethylation of Snrpn differentially methylated regions during the process of postovulatory aging. 19095049_The tissue-specific methylation of the mouse conserved activator sequence and its methylation-sensitive enhancer activity may control expression of imprinting center transcripts, establishing and/or maintaining imprinting in the Snrpn locus. 22242001_These results support a model in which transcription from the Snrpn upstream exons directs the maternal imprint at the PWS-IC 25064401_Oocytes vitrification could lead to the loss of DNA methylation of imprinted genes (H19, Peg3, and Snrpn) in mouse blastocysts, which is mainly caused by the reductions of DNMTs after vitrification of oocytes. 26962997_The androgenetic patterns of H19, Snrpn, and Mest were maintained even after differentiation of germline-derived pluripotent stem cells(gPS) into neural stem cells(NSC), whereas the fully unmethylated status of Ndn in SSCs was altered to somatic patterns in gPS cells and gPS-NSCs. 27430727_we found that either knockdown of Nr4a1 or 3, 3'- Diindolylmethane (DIM), an Nr4a1 antagonist, were able to rescue the effects of SNRPN knockdown on neurite outgrowth of embryonic cortical neurons, providing the potential therapeutic methods for SNRPN deletion disorders. We thus concluded that maintaining the proper level of SNRPN is critical in cortical neurodevelopment. |
ENSG00000128739 |
SNRPN |
9.765005 |
0.0093551935 |
-6.740017 |
1.56261012 |
18.515616 |
0.00001685178851487053282363730299930892897464218549430370330810546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0001197790213058833334782565804843557089043315500020980834960937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.185515914280876 |
0.242593568368999 |
19.8302594814748 |
11.6089010781729 |
| ENSMUSG00000102758 |
635702 |
Naaladl2 |
protein_coding |
A0A0N4SUJ3 |
Human_homologues FUNCTION: May be catalytically inactive. |
Membrane;Proteomics identification;Reference proteome;Transmembrane;Transmembrane helix |
|
|
mmu:635702; |
membrane [GO:0016020]; nucleoplasm [GO:0005654]; response to bacterium [GO:0009617] |
Human_homologues 19132087_Observational study and genome-wide association study of gene-disease association. (HuGE Navigator) 20379614_Clinical trial of gene-disease association and gene-environment interaction. (HuGE Navigator) 24240687_Data suggest that changes in expression of N-acetyl-L-aspartyl-L-glutamate peptidase-like 2 (NAALADL2) can impact upon a number of pro-oncogenic pathways and processes, making it a useful biomarker for both diagnosis and prognosis. 28045058_GWAS showed intestinal Behcet's disease-specific associations with YIPF7 gene locus rs6838327. Down-regulation of YIPF7 expression was associated with exacerbating intestinal inflammatory responses both in vitro and in vivo. |
ENSG00000177694 |
NAALADL2 |
39.177517 |
0.0496434063 |
-4.332254 |
0.80767671 |
26.502863 |
0.00000026314783705073463906869515351039101602736991480924189090728759765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000025661125825587643315762228363174912715294340159744024276733398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.05918979925244 |
1.80352889084498 |
61.6232855447591 |
21.3029925033507 |
| ENSMUSG00000103189 |
|
Gm37092 |
TEC |
|
|
|
|
|
|
|
|
|
|
7.984248 |
8.8571081976 |
3.146836 |
1.03999388 |
9.482049 |
0.00207492080422597841016241204670222941786050796508789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0087862920483836662310928034003154607489705085754394531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.6281733900764 |
10.6973624710022 |
1.53867075867417 |
1.01872280086459 |
| ENSMUSG00000103308 |
|
Gm37800 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
53.401951 |
0.4355614908 |
-1.199052 |
0.39196656 |
9.274211 |
0.00232402784859923145985294290483125223545357584953308105468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0096932070618465197842095193436762201599776744842529296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.4410281904373 |
8.92275493383478 |
74.4809375453882 |
14.7144096547026 |
| ENSMUSG00000104164 |
|
Gm38248 |
TEC |
|
|
|
|
|
|
|
|
|
|
55.786034 |
2.0961250748 |
1.067725 |
0.34431918 |
9.610093 |
0.00193510887263648612838240481437424023170024156570434570312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0082808190982225724635634733772349136415868997573852539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
73.9903210052263 |
17.8269369576585 |
35.2986192920255 |
6.59360867975099 |
| ENSMUSG00000104213 |
380797 |
Ighd |
IG_C_gene |
A0A0A6YXD7 |
|
Proteomics identification;Reference proteome |
|
|
|
B cell receptor complex [GO:0019815]; external side of plasma membrane [GO:0009897]; immunoglobulin complex, circulating [GO:0042571]; antigen binding [GO:0003823]; immunoglobulin receptor binding [GO:0034987]; B cell proliferation [GO:0042100]; B cell receptor signaling pathway [GO:0050853]; complement activation, classical pathway [GO:0006958]; defense response to bacterium [GO:0042742]; immunoglobulin mediated immune response [GO:0016064]; innate immune response [GO:0045087]; phagocytosis, engulfment [GO:0006911]; phagocytosis, recognition [GO:0006910]; positive regulation of B cell activation [GO:0050871]; positive regulation of B cell proliferation [GO:0030890]; positive regulation of interleukin-1 production [GO:0032732]; regulation of immune response [GO:0050776]; somatic hypermutation of immunoglobulin genes [GO:0016446] |
1838007_Mouse B cells develop and function without expressing IgD due to targeted gene inactivation 8418208_IgD is an auxiliary antigen receptor 22527518_analysis of a transgenic mouse model with liver-specific expression of secretory immunoglobulin D |
|
|
27.995023 |
0.1208515051 |
-3.048693 |
0.65195060 |
21.377815 |
0.00000377109549997334722910174477361611167225419194437563419342041015625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000302960583626550739716736665441132458909123670309782028198242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.2605610283789 |
2.92641150654437 |
51.803748927203 |
15.468755251837 |
| ENSMUSG00000104559 |
|
Gm43118 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
135.558196 |
0.4683368877 |
-1.094381 |
0.31311455 |
12.046520 |
0.00051889152641511739148111326258572262304369360208511352539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0025846160007701331727536508964249151176773011684417724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
85.2309147653412 |
18.6436107137118 |
181.986337212702 |
29.1130625622163 |
| ENSMUSG00000104784 |
|
Gm42984 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
27.875392 |
0.1702505779 |
-2.554268 |
0.81627561 |
8.629082 |
0.00330838276958482328804489469575855764560401439666748046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0130248187271427371541898665441294724587351083755493164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.21422213688146 |
4.30309684988105 |
42.3741418397158 |
18.1256387928441 |
| ENSMUSG00000105440 |
102634003 |
Gm31693 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
10.021279 |
0.1992367040 |
-2.327445 |
0.86703241 |
7.226377 |
0.00718400354513205711970424971468673902563750743865966796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0253537400369442771086436749783388222567737102508544921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.45644468811487 |
2.20457889492461 |
17.3484333876179 |
7.11907375878822 |
| ENSMUSG00000105826 |
|
Gm42141 |
TEC |
|
|
|
|
|
|
|
|
|
|
19.865315 |
0.3243362240 |
-1.624438 |
0.58760540 |
7.437595 |
0.00638752053223085643901546504253019520547240972518920898437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0228659724434642566692765797142783412709832191467285156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.1723066100616 |
4.25865598345726 |
31.3634613022708 |
9.02616560499717 |
| ENSMUSG00000106164 |
|
9430085M18Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
25.146714 |
5.2988337154 |
2.405675 |
0.57840421 |
17.166630 |
0.00003423985774033367696566818039194401990243932232260704040527343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002282457387749507954702310463801495643565431237220764160156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
43.1858378567413 |
17.7816076813965 |
8.15006474562087 |
2.70106410600283 |
| ENSMUSG00000106547 |
|
B230303O12Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
46.784248 |
2.7696472507 |
1.469702 |
0.43598295 |
11.044778 |
0.00088937357220111607630785854894384101498872041702270507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0041618857916336099433962658622476737946271896362304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
69.0088522879188 |
21.1733750326249 |
24.9161160402489 |
5.91731482660359 |
| ENSMUSG00000106674 |
|
Gm20005 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
45.253145 |
4.8319279490 |
2.272599 |
0.40413934 |
31.533344 |
0.00000001960447237670313940292884480158519533432581738452427089214324951171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000002257107507497673548689087058560787113492551725357770919799804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
72.5540222714482 |
20.5912765170731 |
15.0155430785859 |
3.48335614757772 |
| ENSMUSG00000106924 |
|
Gm42857 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
24.877933 |
0.3640548258 |
-1.457772 |
0.54812696 |
7.023686 |
0.00804384761073056314684137646509043406695127487182617187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0278666566947653807084783750269707525148987770080566406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
13.0813614625343 |
5.09047215384814 |
35.9323940633361 |
9.82156706460341 |
| ENSMUSG00000107233 |
|
Gm42959 |
TEC |
|
|
|
|
|
|
|
|
|
|
34.539644 |
0.4157585175 |
-1.266182 |
0.43217916 |
8.572178 |
0.00341338571639642493829591707310555648291483521461486816406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0133417221534899536894425153832344221882522106170654296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.0530634353497 |
6.09742096690532 |
48.232477729759 |
10.3651131879569 |
| ENSMUSG00000107479 |
72484 |
2610300M13Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
494.156254 |
4.6535708897 |
2.218338 |
0.16587208 |
171.406111 |
0.00000000000000000000000000000000000000364808664947578471640647350114347824373974198746313706174889520820526405226206938249633902188614434803498598469673197541851550340652465820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000000004116122655171492775274653007992074432672347522896538435011819392463073688801011430676622656146552287070861098072782624512910842895507812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
801.838001518991 |
92.393577404998 |
172.305960415336 |
15.5554866578296 |
| ENSMUSG00000107567 |
|
4930480K02Rik |
TEC |
|
|
|
|
|
|
|
|
|
|
23.880539 |
0.2995301463 |
-1.739227 |
0.53487087 |
10.550872 |
0.00116133294151128207798151503737926759640686213970184326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0052772742735978069375524235340435552643612027168273925781250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.1359540749644 |
3.84452694217434 |
33.8395123166603 |
8.84629377957593 |
| ENSMUSG00000107655 |
|
Gm44220 |
TEC |
|
|
|
|
|
|
|
|
|
|
9.638818 |
7.4054157076 |
2.888581 |
0.91744478 |
10.146793 |
0.00144552474140814874760396779862503535696305334568023681640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0064174388986094205167276527390640694648027420043945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
16.1964205259594 |
11.0080683740373 |
2.18710483861194 |
1.24074933119151 |
| ENSMUSG00000107709 |
319812 |
9430018G01Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
176.707593 |
7.6303356371 |
2.931747 |
0.23288875 |
158.460461 |
0.00000000000000000000000000000000000245487305956770339249160808185141297183394725278668953003282241148939573154570966484172607892204343027486856954055838286876678466796875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000000000000002428092626190601173664426902776670284868486010029016553341555257909511414474301923043452703515621138308233639691025018692016601562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
312.013723850243 |
50.5862168844005 |
40.8912187734103 |
5.50739192225472 |
| ENSMUSG00000107789 |
|
Gm44965 |
protein_coding |
A0A0N4SVR3 |
|
Reference proteome |
|
|
|
|
|
|
|
33.443802 |
0.1240888704 |
-3.010554 |
0.96689102 |
8.623795 |
0.00331799767297377180830242338060998008586466312408447265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0130458803703501526050079206697773770429193973541259765625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
7.64407822514971 |
5.51526550245016 |
61.601642431154 |
30.6651985898079 |
| ENSMUSG00000107810 |
100417431 |
Gm18609 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
56.392148 |
3.0361101277 |
1.602224 |
0.35978598 |
19.539387 |
0.00000985467038890884561037203370759129938960541039705276489257812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000731512375067541503921575429814083690871484577655792236328125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
83.7058310098734 |
21.0920003364716 |
27.5700905068798 |
5.47659334554478 |
| ENSMUSG00000107879 |
330427 |
9330102E08Rik |
translated_unprocessed_pseudogene |
|
|
|
|
|
|
|
|
|
|
116.473699 |
3.0820073178 |
1.623870 |
0.24507540 |
43.363300 |
0.00000000004546356011365622266368260492701031810769407570660405326634645462036132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000007527184213600125764981778667388895298628881391778122633695602416992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
169.814464127953 |
28.9858863575419 |
55.0986570177561 |
7.47704196616891 |
| ENSMUSG00000107932 |
|
Gm44432 |
TEC |
|
|
|
|
|
|
|
|
|
|
21.967480 |
2.8559436430 |
1.513968 |
0.54285490 |
7.768606 |
0.00531620564692824194158449202518568199593573808670043945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0195664202369934953074093186842219438403844833374023437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.6572936126119 |
12.9654850416358 |
11.7849992227567 |
3.54425924174184 |
| ENSMUSG00000108015 |
102635189 |
Gm32591 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
30.355821 |
2.8623743527 |
1.517212 |
0.50785623 |
8.646055 |
0.00327770299126259280139694674005568231223151087760925292968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0129208001974409469186921839423121127765625715255737304687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.5194497273182 |
14.2007021263678 |
13.8065273292955 |
3.81438738203415 |
| ENSMUSG00000108398 |
115489946 |
Gm30191 |
protein_coding |
A0A0U1RNT7 |
|
Reference proteome |
|
|
|
|
|
ENSG00000282872 |
C1orf232 |
9.770232 |
0.2126340900 |
-2.233555 |
0.87633210 |
6.415740 |
0.01131131764802786703050241357004779274575412273406982421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0372125249275940539472884438509936444461345672607421875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.52861738268943 |
2.27763718583418 |
16.5947867696606 |
6.99398901050286 |
| ENSMUSG00000108900 |
619297 |
Ccdc194 |
protein_coding |
A0A140LIT1 |
|
Coiled coil;Reference proteome;Signal |
|
|
|
|
|
ENSG00000269720 |
CCDC194 |
321.538160 |
0.0184682529 |
-5.758809 |
0.30290025 |
471.375904 |
0.00000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000001608284499931729016948939206132905819590275432607979251278906891483736741988928174554114450503906584247580376568077988437938166401428154547271266893858569247241 |
0.0000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000090731072233185543786574056579420858126748318232812637280702961346133633861173969140160476888143896938487670980918104131889910143249958791476914627073155801164 |
Yes |
No |
11.5130725083913 |
2.43501890337571 |
623.398033206332 |
61.0604515815471 |
| ENSMUSG00000109155 |
|
Gm18757 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
7.857704 |
0.0120012178 |
-6.380675 |
1.57962561 |
16.205720 |
0.00005682228196529736898500936859157661729113897308707237243652343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0003588378934060570133249279400899922620737925171852111816406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.175948219734695 |
0.233514434122153 |
14.6608638482685 |
8.87099956850268 |
| ENSMUSG00000109394 |
319893 |
A230057D06Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
116.433375 |
2.2344187275 |
1.159900 |
0.33070072 |
11.709331 |
0.00062187472707879725038193630126670541358180344104766845703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0030263245504358591599081140799398781382478773593902587890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
160.22349830916 |
37.0495913453512 |
71.7070154921839 |
12.6090685672451 |
| ENSMUSG00000109953 |
|
5430430B14Rik |
TEC |
|
|
|
|
|
|
|
|
|
|
16.698267 |
0.2641883328 |
-1.920361 |
0.64515974 |
8.849203 |
0.00293216889928901930720273050212654197821393609046936035156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0117688528785165578971483313353019184432923793792724609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.8321830584517 |
3.1581439904784 |
25.8610324926982 |
8.06966262076479 |
| ENSMUSG00000110170 |
|
St6galnac2 |
protein_coding |
C0J8I2 |
|
Reference proteome |
|
|
|
|
|
ENSG00000284526 |
|
43.943617 |
3.4927409954 |
1.804360 |
0.43035525 |
17.245366 |
0.00003284978006786611314784737891692145694833016023039817810058593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002198452767986540455202176813642722663644235581159591674804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.9757053910937 |
20.2764370320657 |
19.1756862243606 |
4.58744316619811 |
| ENSMUSG00000110195 |
207728 |
Pde2a |
protein_coding |
Q922S4 |
FUNCTION: cGMP-activated cyclic nucleotide phosphodiesterase with a dual-specificity for the second messengers cAMP and cGMP, which are key regulators of many important physiological processes (PubMed:21724846). Has a higher efficiency with cGMP compared to cAMP (By similarity). Plays a role in cell growth and migration (By similarity). {ECO:0000250|UniProtKB:O00408, ECO:0000269|PubMed:21724846}.; FUNCTION: [Isoform PDE2A2]: Regulates mitochondrial cAMP levels and respiration (PubMed:21724846). Involved in the regulation of mitochondria morphology/dynamics and apoptotic cell death via local modulation of cAMP/PKA signaling in the mitochondrion, including the monitoring of local cAMP levels at the outer mitochondrial membrane and of PKA-dependent phosphorylation of DNM1L (PubMed:28463107). {ECO:0000269|PubMed:21724846, ECO:0000269|PubMed:28463107}. |
3D-structure;Alternative splicing;cAMP;Cell membrane;cGMP;cGMP-binding;Cytoplasm;Direct protein sequencing;Hydrolase;Lipoprotein;Magnesium;Membrane;Metal-binding;Mitochondrion;Mitochondrion inner membrane;Mitochondrion outer membrane;Myristate;Nucleotide-binding;Palmitate;Phosphoprotein;Reference proteome;Repeat;Zinc |
|
|
mmu:207728; |
axon [GO:0030424]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; dendrite [GO:0030425]; endoplasmic reticulum [GO:0005783]; Golgi apparatus [GO:0005794]; hippocampal mossy fiber [GO:0097457]; membrane raft [GO:0045121]; mitochondrial inner membrane [GO:0005743]; mitochondrial matrix [GO:0005759]; mitochondrial outer membrane [GO:0005741]; mitochondrion [GO:0005739]; nucleus [GO:0005634]; perinuclear region of cytoplasm [GO:0048471]; plasma membrane [GO:0005886]; presynaptic membrane [GO:0042734]; synaptic membrane [GO:0097060]; 3',5'-cyclic-AMP phosphodiesterase activity [GO:0004115]; 3',5'-cyclic-GMP phosphodiesterase activity [GO:0047555]; 3',5'-cyclic-nucleotide phosphodiesterase activity [GO:0004114]; cAMP binding [GO:0030552]; cGMP binding [GO:0030553]; cGMP-stimulated cyclic-nucleotide phosphodiesterase activity [GO:0004118]; GAF domain binding [GO:0036004]; identical protein binding [GO:0042802]; magnesium ion binding [GO:0000287]; phosphate ion binding [GO:0042301]; protein homodimerization activity [GO:0042803]; TPR domain binding [GO:0030911]; zinc ion binding [GO:0008270]; adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway [GO:0007193]; aorta development [GO:0035904]; cAMP-mediated signaling [GO:0019933]; cardiac septum development [GO:0003279]; cellular response to 2,3,7,8-tetrachlorodibenzodioxine [GO:1904613]; cellular response to cAMP [GO:0071320]; cellular response to cGMP [GO:0071321]; cellular response to granulocyte macrophage colony-stimulating factor stimulus [GO:0097011]; cellular response to lipopolysaccharide [GO:0071222]; cellular response to macrophage colony-stimulating factor stimulus [GO:0036006]; cellular response to mechanical stimulus [GO:0071260]; cGMP catabolic process [GO:0046069]; cGMP-mediated signaling [GO:0019934]; coronary vasculature development [GO:0060976]; establishment of endothelial barrier [GO:0061028]; heart development [GO:0007507]; heart valve development [GO:0003170]; negative regulation of cAMP-mediated signaling [GO:0043951]; negative regulation of cell death [GO:0060548]; negative regulation of cGMP-mediated signaling [GO:0010754]; negative regulation of oxidative phosphorylation [GO:0090324]; negative regulation of transcription by RNA polymerase II [GO:0000122]; negative regulation of vascular permeability [GO:0043116]; positive regulation of gene expression [GO:0010628]; positive regulation of inflammatory response [GO:0050729]; positive regulation of vascular permeability [GO:0043117]; regulation of cAMP-mediated signaling [GO:0043949]; regulation of cGMP-mediated signaling [GO:0010752]; regulation of mitochondrion organization [GO:0010821]; regulation of nitric oxide mediated signal transduction [GO:0010749]; signal transduction [GO:0007165]; ventricular septum development [GO:0003281] |
15210692_cGMP binding to PDE2 GAF-B activates the enzyme rapidly, stoichiometrically, and in an all or none fashion, rather than variably over a large range of cyclic nucleotide concentrations 18849438_HV(T)-induced endothelial barrier dysfunction resulted from a simultaneous increase in NO/sGC-derived cGMP and PDE2A expression causing decreased cAMP. 19390057_Phosphodiesterase 2 mediates redox-sensitive endothelial cell proliferation and angiogenesis by thrombin via Rac1 and NADPH oxidase 2. 19632989_dual acylation as mechanism targeting neuronal PDE2A3 to synapses thereby ensuring local control of cyclic nucleotides. 23045693_results demonstrate strong presynaptic inhibition by natriuretic peptides in the brain and suggest important physiological and behavioral roles of PDE2A in modulating neurotransmitter release 25063878_We conclude that increased AM PDE2A is an important negative regulator of macrophage iNOS expression. 25442113_Effect of PDE2 inhibition on stress-induced memory impairment is potentially mediated via modulation of neuroplasticity-related NMDAR-CaMKII-cGMP/cAMP signaling. 26243800_Cardiac hypertrophy is inhibited by a local pool of cAMP regulated by PDE2A. 29409032_Pde2A is indispensable for heart development because Pde2A knockout embryos show severe atria and ventricular defects. 31100470_PDE2 regulates membrane potential, respiration and permeability transition of rodent subsarcolemmal cardiac mitochondria. 34062838_Cellular Mechanisms of the Anti-Arrhythmic Effect of Cardiac PDE2 Overexpression. |
ENSG00000186642 |
PDE2A |
11.554016 |
5.4680318454 |
2.451022 |
0.91495754 |
7.522103 |
0.00609464785477788466988569027193989313673228025436401367187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0220193728946813903346590990395270637236535549163818359375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
17.4998135748305 |
11.857472049421 |
3.20038618459147 |
1.95405068609592 |
| ENSMUSG00000110290 |
|
Gm45336 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
120.174097 |
0.1674122461 |
-2.578523 |
0.32075661 |
61.931115 |
0.00000000000000355684633424563210865784043467890380546661587349288602410979365231469273567199707031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000916715454538569681951701837560269533762124377629376681397843640297651290893554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
32.8562693909437 |
7.36531628544621 |
196.25965339534 |
30.3707751187964 |
| ENSMUSG00000110409 |
|
Gm9908 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
18.536346 |
0.2508875706 |
-1.994887 |
0.73864410 |
7.011212 |
0.00810008314077776889194915099778881995007395744323730468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0280346435810786137610151058652263600379228591918945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
6.49239880212686 |
3.47117577353407 |
25.8777219913543 |
9.57918247695763 |
| ENSMUSG00000110652 |
102635769 |
Gm33023 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
39.784109 |
0.0531383637 |
-4.234102 |
0.90297927 |
17.394136 |
0.00003037617513692026087902126052497919772577006369829177856445312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0002043683302498098045257152177711645890667568892240524291992187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.94027277372641 |
2.63041242815215 |
74.1511875345058 |
35.2928256376565 |
| ENSMUSG00000110866 |
|
Gm48362 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
21.275279 |
4.1969941546 |
2.069356 |
0.68908038 |
8.386288 |
0.00378062137185034095182567526194361562374979257583618164062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0145862271367842941810488710530080425087362527847290039062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
28.5414342051133 |
14.1566427193537 |
6.80044649896857 |
2.70705777075525 |
| ENSMUSG00000110998 |
619328 |
I730028E13Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
12.041068 |
0.0572978161 |
-4.125376 |
1.07680444 |
15.744136 |
0.00007251237274899486424592498501340287475613877177238464355468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004451868043985045416376100657629422130412422120571136474609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.20035842608805 |
0.981461629198656 |
20.9494620851164 |
9.31962953695386 |
| ENSMUSG00000111059 |
|
Gm33838 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
26.097741 |
3.5515858463 |
1.828463 |
0.56311917 |
10.141037 |
0.00145004576477099291105521405853551186737604439258575439453125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0064318861645287607811671826141264318721368908882141113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
39.286023315973 |
15.7266444058969 |
11.061544058297 |
3.40889238952951 |
| ENSMUSG00000111765 |
100038575 |
Gm10635 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
212.758090 |
0.0075681349 |
-7.045846 |
1.89190098 |
7.284522 |
0.00695512457040863181501055834132785093970596790313720703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0246258617983413016783167392986797494813799858093261718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.80650388461334 |
4.82969484616264 |
370.831643078784 |
419.064850712363 |
| ENSMUSG00000112035 |
102632477 |
Gm30539 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
65.743748 |
5.9086669898 |
2.562833 |
0.79972115 |
8.908833 |
0.00283794632457492123842057729632415430387482047080993652343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0114570894290816870098037583147743134759366512298583984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
111.453186489695 |
64.9941723404691 |
18.8626616937052 |
8.02406013300159 |
| ENSMUSG00000112280 |
414093 |
A830082N09Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
63.382159 |
0.4238747592 |
-1.238290 |
0.38448581 |
10.214753 |
0.00139321668562485253758942427282363496487960219383239746093750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0062032962746091059244579746234649064717814326286315917968750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
38.0967152605416 |
10.2735882861482 |
89.8772914213065 |
17.9746196628913 |
| ENSMUSG00000112302 |
|
Gm48226 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
11.204302 |
0.0163192140 |
-5.937285 |
1.43589314 |
21.606681 |
0.00000334684057061020875608638169840247655884013511240482330322265625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000271598697770563155635611951588259671552805230021476745605468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.343197664300114 |
0.400833950897604 |
21.0302814623695 |
9.81217116310185 |
| ENSMUSG00000112466 |
432500 |
Gm29674 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
44.725961 |
0.2264739446 |
-2.142583 |
0.45303850 |
21.955068 |
0.00000279108801047145067185601199910571779128076741471886634826660156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000229185189086259510102359243655101295189524535089731216430664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
14.4509318551992 |
4.61286498188576 |
63.8083638297172 |
14.0857636093507 |
| ENSMUSG00000112481 |
628062 |
Gm29684 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
35.971915 |
0.3816017497 |
-1.389860 |
0.42429649 |
10.665623 |
0.00109145110271155541641074915304443493369035422801971435546875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0049939871422356296032374345372772950213402509689331054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
19.0455072813719 |
5.68237890835034 |
49.9093814377472 |
10.5237964721554 |
| ENSMUSG00000112540 |
|
Gm47461 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
11.031434 |
0.0698546348 |
-3.839500 |
1.10683053 |
12.031316 |
0.00052314117345979792815524600513299446902237832546234130859375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0025998324157062453060307305463538796175271272659301757812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.25347910783041 |
1.05881805358537 |
17.9441079584025 |
8.76835617250215 |
| ENSMUSG00000112596 |
|
Gm48804 |
TEC |
|
|
|
|
|
|
|
|
|
|
9.195568 |
0.1254109186 |
-2.995265 |
1.00207966 |
8.743486 |
0.00310710004144405631254510780081545817665755748748779296875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0123408990433574606432332032568410795647650957107543945312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.95744297042389 |
1.47161085062325 |
15.6082340488558 |
7.14305121208544 |
| ENSMUSG00000112778 |
102636674 |
Gm33677 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
9.350597 |
0.0564114855 |
-4.147867 |
1.20115249 |
13.015370 |
0.00030894475967479286806702254430945231433724984526634216308593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0016351099997798626724254678066472479258663952350616455078125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1.01831580957904 |
0.949225628473878 |
18.0515687745807 |
8.80302872676044 |
| ENSMUSG00000112903 |
546472 |
Gm5948 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
11.363603 |
0.2135704924 |
-2.227216 |
0.82149599 |
7.328742 |
0.00678605170151511897375096182827292068395763635635375976562500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0241099046322877687276164948571022250689566135406494140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
3.77461388855763 |
2.26737789334699 |
17.6738548732487 |
6.99121886151721 |
| ENSMUSG00000113063 |
|
Gm34667 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
16.132615 |
3.5894663410 |
1.843769 |
0.68932914 |
7.091274 |
0.00774601632984922225388935146384028485044836997985839843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0269932099602524233628031424814253114163875579833984375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
22.8081958482607 |
11.3173281397938 |
6.35420245840699 |
2.52991980236112 |
| ENSMUSG00000114161 |
|
Gm48662 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
106.094705 |
0.1056837975 |
-3.242174 |
0.30472331 |
115.719681 |
0.00000000000000000000000000547459303018224450670193922978168947738906921208396377175752848108055879060296344817970748408697545528411865234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000000003376072916426556675598679070197785046176862985293845223656917668491814210618429115129401907324790954589843750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.1835078726508 |
4.29489151573485 |
190.980153551287 |
25.2263947699377 |
| ENSMUSG00000115099 |
|
1700087I21Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
20.747404 |
0.3742517151 |
-1.417919 |
0.55891615 |
6.337298 |
0.01182248162702568411441816920159908477216958999633789062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0387102407873721465758620752239949069917201995849609375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
11.1157764540327 |
4.41490915605032 |
29.7013373760577 |
8.37356249016738 |
| ENSMUSG00000115437 |
|
Gm49314 |
TEC |
|
|
|
|
|
|
|
|
|
|
18.630043 |
3.0231330332 |
1.596044 |
0.63141738 |
6.158843 |
0.01307567014971760047115090230818168492987751960754394531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0422235759424419085372726101468288106843829154968261718750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
27.5852917834785 |
12.4622659717638 |
9.12473631850696 |
3.22126085498218 |
| ENSMUSG00000115483 |
108168154 |
Gm9732 |
protein_coding |
A0A2I3BPB7 |
|
Reference proteome |
|
|
|
|
|
|
|
24.327964 |
2.7566811038 |
1.462932 |
0.54451101 |
7.167953 |
0.00742174139954480775738243636396873625926673412322998046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0260171861299851800430893433713208651170134544372558593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
34.6823366731899 |
13.4030263810759 |
12.5811928789155 |
3.82130310794771 |
| ENSMUSG00000115600 |
|
Gm49299 |
unitary_pseudogene |
|
|
|
|
|
|
|
|
|
|
15.356004 |
0.1920915076 |
-2.380134 |
0.80437767 |
8.592936 |
0.00337469483614860447928673892192819039337337017059326171875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0132414610366346063158315971008960332255810499191284179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
5.29615025988728 |
3.10826624661003 |
27.5709755589017 |
10.7236848266801 |
| ENSMUSG00000115816 |
102637893 |
Gm34589 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
26.146997 |
3.3156964183 |
1.729312 |
0.53587748 |
10.095108 |
0.00148663482324690480670947856367547501577064394950866699218750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0065761476409351600491692835248613846488296985626220703125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
40.5199417844202 |
15.3992932475982 |
12.2206428674076 |
3.64722377228327 |
| ENSMUSG00000115855 |
102637961 |
Gm34643 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
21.933862 |
10.6973095241 |
3.419176 |
0.86789203 |
12.333647 |
0.00044486637372846216456623302804018749156966805458068847656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0022587348682106453932882939028559121652506291866302490234375000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
36.4546691437729 |
23.277172076379 |
3.40783531239891 |
1.65945367377911 |
| ENSMUSG00000116165 |
57028 |
Pdxp |
protein_coding |
P60487 |
FUNCTION: Functions as a pyridoxal phosphate (PLP) phosphatase, which also catalyzes the dephosphorylation of pyridoxine 5'-phosphate (PNP) and pyridoxamine 5'-phosphate (PMP), with order of substrate preference PLP > PNP > PMP and therefore plays a role in vitamin B6 metabolism (PubMed:24338473, PubMed:24338687, PubMed:25783190). Also functions as a protein serine phosphatase that specifically dephosphorylates 'Ser-3' in proteins of the actin-depolymerizing factor (ADF)/cofilin family like CFL1 and DSTN. Thereby, regulates cofilin-dependent actin cytoskeleton reorganization, being required for normal progress through mitosis and normal cytokinesis. Does not dephosphorylate phosphothreonines in LIMK1. Does not dephosphorylate peptides containing phosphotyrosine (By similarity). {ECO:0000250|UniProtKB:Q96GD0, ECO:0000269|PubMed:24338473, ECO:0000269|PubMed:24338687, ECO:0000269|PubMed:25783190}. |
3D-structure;Cell membrane;Cell projection;Cytoplasm;Cytoskeleton;Direct protein sequencing;Hydrolase;Magnesium;Membrane;Metal-binding;Pyridoxal phosphate;Reference proteome |
|
|
mmu:57028; |
actin cytoskeleton [GO:0015629]; cell-cell junction [GO:0005911]; cleavage furrow [GO:0032154]; contractile ring [GO:0070938]; cytoplasm [GO:0005737]; cytosol [GO:0005829]; lamellipodium [GO:0030027]; lamellipodium membrane [GO:0031258]; midbody [GO:0030496]; plasma membrane [GO:0005886]; ruffle membrane [GO:0032587]; glycerol-3-phosphatase activity [GO:0043136]; growth factor binding [GO:0019838]; heat shock protein binding [GO:0031072]; magnesium ion binding [GO:0000287]; myosin phosphatase activity [GO:0017018]; phosphatase activity [GO:0016791]; phosphoprotein phosphatase activity [GO:0004721]; protein homodimerization activity [GO:0042803]; pyridoxal phosphatase activity [GO:0033883]; actin rod assembly [GO:0031247]; cellular response to ATP [GO:0071318]; dephosphorylation [GO:0016311]; positive regulation of actin filament depolymerization [GO:0030836]; protein dephosphorylation [GO:0006470]; pyridoxal phosphate catabolic process [GO:0032361]; regulation of cytokinesis [GO:0032465]; regulation of mitotic nuclear division [GO:0007088] |
14522954_analysis of pyridoxal phosphatase cloning, expression and tissue distribution 24338473_Evolutionary and structural analyses of mammalian haloacid dehalogenase-type phosphatases AUM and chronophin provide insight into the basis of their different substrate specificities. 24338687_Dimer formation is essential for an intermolecular arginine-arginine-tryptophan stacking interaction that positions a critical histidine residue in the substrate specificity loop of chronophin for PLP coordination. 27212638_Results provided novel evidence that PLPP is involved in controlling dendritic spine as well as synaptic plasticity. It showed also for the first time the role of PLPP in GluN functionality via regulation of GluN2A interaction with postsynaptic proteins. These findings suggest that PLPP/CIN may play an important role in information storage and recall capacity, which manifests as a learning memory. 31320629_PLPP/CIN deletion inhibited NEDD4-2 ubiquitination, and diminished the responsiveness of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor by facilitating NEDD4-2-mediated ubiquitination of GluA1 subunit under physiological condition. PLPP/CIN overexpression reversed these effects. These PLPP/CIN-mediated processes were required for the increased seizure severity and its progression in response to kaini... 33414453_PLPP/CIN-mediated NF2-serine 10 dephosphorylation regulates F-actin stability and Mdm2 degradation in an activity-dependent manner. |
ENSG00000241360 |
PDXP |
77.183376 |
0.4895172720 |
-1.030568 |
0.33297160 |
9.497649 |
0.00205735389400231449830158680924796499311923980712890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0087267096946374977245453052887569356244057416915893554687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.2676480144379 |
12.1706688356 |
106.773858666244 |
17.9613590826514 |
| ENSMUSG00000116237 |
|
Gm30339 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
171.692999 |
2.0119233589 |
1.008575 |
0.33526437 |
8.857033 |
0.00291961833784723946011441242376349691767245531082153320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0117370352394006744606302916622553311754018068313598632812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
215.588452898962 |
50.5522264825268 |
107.155400301987 |
18.927852719862 |
| ENSMUSG00000116597 |
224098 |
Gm536 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
163.817650 |
2.0010641609 |
1.000767 |
0.22784950 |
18.910946 |
0.00001369645566146602149489985150720983142491604667156934738159179687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000989679376828512494691098000387796673749107867479324340820312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
219.393037725765 |
34.7936760390431 |
109.63818252965 |
13.3014502790313 |
| ENSMUSG00000116815 |
|
Gm49760 |
processed_pseudogene |
|
|
|
|
|
|
|
|
|
|
11.130905 |
10.2363243131 |
3.355626 |
1.05259953 |
9.629857 |
0.00191439564097768428335699653075607784558087587356567382812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0082002458952114987839054904839031223673373460769653320312500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
20.6276469571211 |
16.4213119104571 |
2.01514199102918 |
1.30321164696854 |
| ENSMUSG00000117106 |
|
Gm49872 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
25.722126 |
0.2631045912 |
-1.926292 |
0.54693093 |
11.933036 |
0.00055147227994359739131763342712133635359350591897964477539062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0027255112810191032927942256236519824597053229808807373046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.5599247313985 |
4.0998894765082 |
40.1358436308002 |
10.6553749349629 |
| ENSMUSG00000117916 |
319720 |
9630028I04Rik |
lncRNA |
|
|
|
|
|
|
|
|
|
|
120.197409 |
5.6939217181 |
2.509423 |
0.36220817 |
45.250619 |
0.00000000001733657077506092737857151531477480901552445935465129878139123320579528808593750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000003004216678563459096919173261098622712550465507774788420647382736206054687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
208.612589525059 |
52.9269840712506 |
36.6377691603613 |
7.23797978482222 |
| ENSMUSG00000118364 |
|
Gm54420 |
TEC |
|
|
|
|
|
|
|
|
|
|
850.674490 |
2.4567829178 |
1.296770 |
0.13214962 |
94.667003 |
0.00000000000000000000022526306012390161744213413872829155941046545375980895560791110454612606872615288011729717254638671875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000000000000000000102730746461295487887518170963224672878497893206251798104127609390090469787537585943937301635742187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
1163.39092833877 |
106.714691532976 |
473.542420005302 |
33.2601539456561 |
| ENSMUSG00000118365 |
|
Gm41724 |
lncRNA |
|
|
|
|
|
|
|
|
|
|
180.798890 |
0.4271169524 |
-1.227297 |
0.23726645 |
26.275812 |
0.00000029597237443750337485649315807934023325742600718513131141662597656250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000028623817190044771790536411865124577502683678176254034042358398437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
105.957218346415 |
17.5044443644837 |
248.075422313962 |
29.6407360537701 |
| ENSMUSG00000120064 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
31.761407 |
6.5135938139 |
2.703454 |
0.67057479 |
14.258547 |
0.00015933556453599995650013965153846129396697506308555603027343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0009062730840225359735598531507605457591125741600990295410156250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
52.2933436368665 |
25.1910284785125 |
8.02833967402829 |
3.07236403233796 |
| ENSMUSG00000120130 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
55.913397 |
2.0900329013 |
1.063526 |
0.34401817 |
9.546962 |
0.00200280650805240330314394014976642210967838764190673828125000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0085190585676219506700013184286035539116710424423217773437500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
77.2394571188543 |
18.5931946407222 |
36.9560962760077 |
6.85942543768778 |
| ENSMUSG00000120156 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
16.964459 |
0.0217784824 |
-5.520953 |
1.20300527 |
30.917732 |
0.00000002692009142978420450254022815508858634814259858103469014167785644531250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000003053215433048942962632654653937214206393946369644254446029663085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
0.715579800147773 |
0.668286375140449 |
32.8571930633503 |
11.8515233031899 |
| ENSMUSG00000120268 |
105245601 |
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
8.682119 |
0.1481898892 |
-2.754481 |
0.98659602 |
7.807185 |
0.00520389151769648592577022228056193853262811899185180664062500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0192024753542905692038633702622973942197859287261962890625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
2.41250665375776 |
1.78143965017666 |
16.2798330399073 |
7.4132930167108 |
| ENSMUSG00000120364 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
44.215119 |
2.8923675435 |
1.532251 |
0.38306853 |
15.865505 |
0.00006800705474313708089398322620411363459425047039985656738281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0004207487643572153023993620557519079738995060324668884277343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
66.3113666916819 |
17.8148065349654 |
22.9263278934809 |
4.78040358866999 |
| ENSMUSG00000120798 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
51.025851 |
0.4703061967 |
-1.088328 |
0.37309605 |
8.446560 |
0.00365734553986618495663440953080680628772825002670288085937500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0141824560242468748760824581722772563807666301727294921875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
33.4780368397257 |
8.75459870542854 |
71.1834908237497 |
13.3158393643736 |
| ENSMUSG00000120903 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
63.473235 |
0.2643469964 |
-1.919495 |
0.38233601 |
24.718972 |
0.00000066328017487915015033280536835280649654578155605122447013854980468750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0000060972140155456937309552331583084594512911280617117881774902343750000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
25.6320599319505 |
6.87267747986884 |
96.9636889261728 |
18.2239660580939 |
| ENSMUSG00000121092 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
22.377857 |
0.2873648992 |
-1.799044 |
0.66518012 |
7.177912 |
0.00738065647530688345723248744434386026114225387573242187500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0259144405022137848393359860210694023407995700836181640625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.4032140870329 |
4.96834760581003 |
36.2021044189244 |
12.0160606917372 |
| ENSMUSG00000121179 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
26.049217 |
0.2466275694 |
-2.019594 |
0.53886120 |
13.959928 |
0.00018674884604605405122659811389951300952816382050514221191406250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0010473337345263237802700517775633670680690556764602661132812500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
10.3183866762923 |
3.94426135565363 |
41.837928744188 |
10.839675769585 |
| ENSMUSG00000121184 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
102.386770 |
2.0694944823 |
1.049278 |
0.28184111 |
13.704893 |
0.00021389648223389757576186342991064748275675810873508453369140625000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0011761989954464722573507229341771562758367508649826049804687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
No |
138.971869003715 |
27.3221981266123 |
67.152568026349 |
10.1410861154645 |
| ENSMUSG00000121318 |
|
|
lncRNA |
|
|
|
|
|
|
|
|
|
|
31.564584 |
2.6311821829 |
1.395711 |
0.50902478 |
7.164107 |
0.00743766943427981008091220971323309640865772962570190429687500000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0260557913576242097808410846937476890161633491516113281250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
37.3493540075922 |
13.4530622843649 |
14.1948946941707 |
3.89119424933595 |
| ENSMUSG00002076083 |
231003 |
Klhl17 |
protein_coding |
Q6TDP3 |
FUNCTION: Substrate-recognition component of some cullin-RING-based BCR (BTB-CUL3-RBX1) E3 ubiquitin-protein ligase complexes. The BCR(KLHL17) complex mediates the ubiquitination and subsequent degradation of GLUR6. May play a role in the actin-based neuronal function (By similarity). {ECO:0000250}. |
Actin-binding;Kelch repeat;Reference proteome;Repeat;Synapse;Ubl conjugation pathway |
PATHWAY: Protein modification; protein ubiquitination. |
|
mmu:231003; |
actin cytoskeleton [GO:0015629]; dendrite cytoplasm [GO:0032839]; glutamatergic synapse [GO:0098978]; neuronal cell body [GO:0043025]; postsynapse [GO:0098794]; postsynaptic density [GO:0014069]; actin filament binding [GO:0051015]; molecular adaptor activity [GO:0060090]; POZ domain binding [GO:0031208]; actin cytoskeleton organization [GO:0030036]; brain development [GO:0007420]; protein ubiquitination [GO:0016567]; regulation protein catabolic process at postsynapse [GO:0140252] |
33256713_KLHL17/Actinfilin, a brain-specific gene associated with infantile spasms and autism, regulates dendritic spine enlargement. |
ENSG00000187961 |
KLHL17 |
765.149074 |
5.7492913387 |
2.523384 |
0.90004981 |
6.989206 |
0.00820027121295145537682724778960619005374610424041748046875000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
0.0283042218707628759344352431526203872635960578918457031250000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000000 |
Yes |
Yes |
1148.72038847708 |
764.049646913227 |
199.802083562491 |
96.7225048466654 |